Written by Dr. Michael Torres, MD, Board-Certified Obesity Medicine Physician
Medically reviewed by Dr. Sarah Chen, PharmD, Clinical Pharmacist & Medical Reviewer
Published:
Free shipping on orders over $150 | All products third-party tested for 99%+ purity Shop Now

Compare Ozempic and Trulicity for diabetes management. Clinical trial data shows semaglutide produces greater weight loss while dulaglutide offers...
Written by Dr. Michael Torres, MD, Board-Certified Obesity Medicine Physician
Medically reviewed by Dr. Sarah Chen, PharmD, Clinical Pharmacist & Medical Reviewer
Published:
Ozempic (semaglutide) and Trulicity (dulaglutide) are both GLP-1 receptor agonists that effectively manage type 2 diabetes, but Ozempic typically produces greater weight loss while Trulicity offers more convenient weekly dosing with potentially fewer gastrointestinal side effects. Both medications work through similar mechanisms but have distinct pharmacological profiles that make them suitable for different patient populations.
According to the landmark SUSTAIN-7 trial published in Diabetes Care (Pratley et al., 2018), semaglutide demonstrated superior glycemic control and weight reduction compared to dulaglutide when directly compared head-to-head. However, the choice between these medications depends on individual patient factors including weight loss goals, side effect tolerance, and injection preferences.
| Factor | Ozempic (Semaglutide) | Trulicity (Dulaglutide) |
|---|---|---|
| Mechanism | GLP-1 receptor agonist | GLP-1 receptor agonist |
| FDA Status | Approved 2017 | Approved 2014 |
| Typical Dose | 0.5-2.0 mg weekly | 0.75-4.5 mg weekly |
| Key Benefit | Greater weight loss | Convenient dosing |
| Common Side Effects | Nausea (16-20%) | Nausea (12-21%) |
| Monthly Cost Range | $900-1,000 (brand) | $800-900 (brand) |
Both Ozempic and Trulicity belong to the GLP-1 receptor agonist class, but they have distinct molecular structures that affect their pharmacokinetic properties and clinical effects. Understanding these differences helps explain why patients may respond differently to each medication.
Semaglutide, the active ingredient in Ozempic, is a modified human GLP-1 analog with 94% amino acid sequence homology to native GLP-1. The key modification involves fatty acid acylation that allows the molecule to bind to albumin, significantly extending its half-life to approximately 165 hours (Lau et al., Journal of Clinical Pharmacology, 2015). This extended half-life enables once-weekly dosing while maintaining consistent therapeutic levels.
Think of GLP-1 receptors as volume controls throughout your body. When activated, they turn down appetite signals in your brain, slow gastric emptying to help you feel full longer, and increase insulin sensitivity in your pancreas. Semaglutide's albumin-binding design means it stays in your system longer, providing more sustained receptor activation.
Dulaglutide, found in Trulicity, uses a different approach to achieve weekly dosing. Rather than albumin binding, dulaglutide is fused to a modified human IgG4 Fc fragment, creating a larger molecule with an elimination half-life of approximately 112 hours (Glaesner et al., Diabetes, Obesity and Metabolism, 2010). This Fc fusion technology provides stability and extended circulation time through a different mechanism than semaglutide.
The structural differences translate to clinical variations. Semaglutide demonstrates higher receptor binding affinity and potency in preclinical studies, which may contribute to its superior weight loss effects observed in clinical trials. Dulaglutide's Fc fusion design may result in different immunogenicity profiles and injection site reactions compared to semaglutide.
Both medications work by activating GLP-1 receptors in multiple tissues. In the pancreas, they enhance glucose-dependent insulin secretion and suppress glucagon release. In the stomach, they slow gastric emptying, promoting satiety. In the brain, they activate hypothalamic GLP-1 receptors that regulate appetite and food intake. The central nervous system effects are particularly important for weight management, an area where semaglutide shows clinical advantages.
Bioavailability differs between the two drugs due to their distinct formulations. Semaglutide achieves peak plasma concentrations 1-3 days after injection, while dulaglutide reaches peak levels within 12-72 hours. These pharmacokinetic differences may influence side effect profiles and patient tolerance during dose titration.
Clinical trial data consistently demonstrates that semaglutide produces superior weight loss compared to dulaglutide, making this a key differentiating factor for patients whose primary goal includes weight management alongside glycemic control.
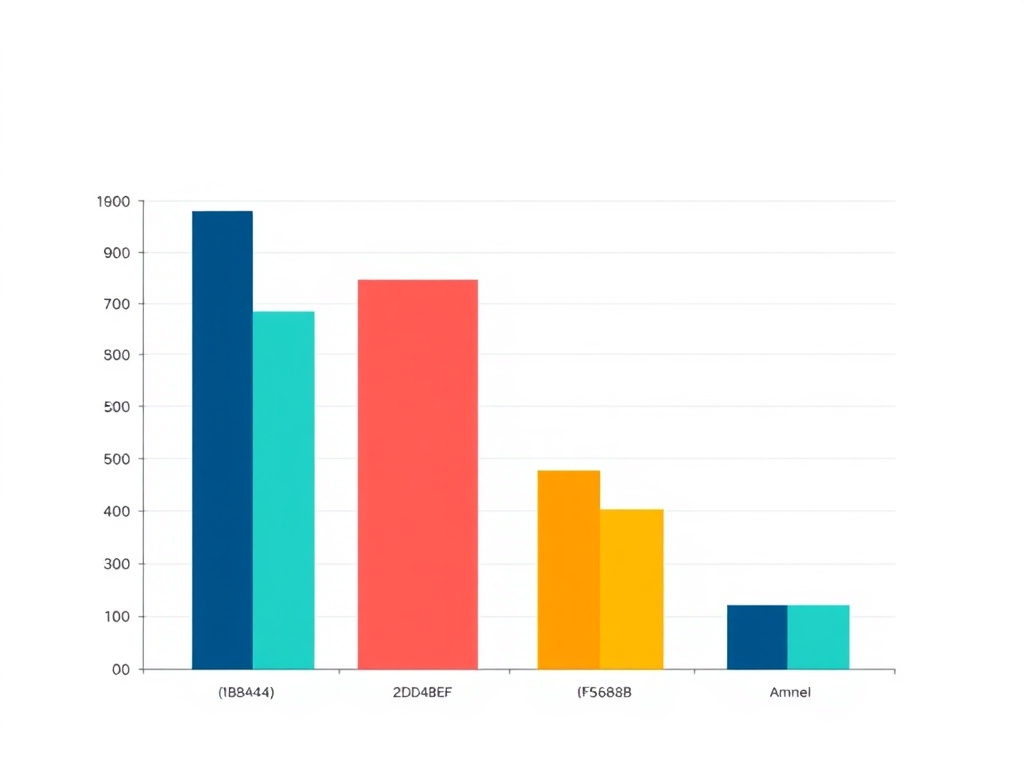
The SUSTAIN-7 trial represents the most direct comparison between these medications. This 40-week, randomized, double-blind study enrolled 1,201 adults with type 2 diabetes and compared semaglutide 0.5 mg and 1.0 mg weekly against dulaglutide 0.75 mg and 1.5 mg weekly (Pratley et al., Diabetes Care, 2018). The results showed clear superiority for semaglutide across multiple endpoints.
Weight loss results from SUSTAIN-7 were particularly striking. Patients receiving semaglutide 1.0 mg lost an average of 4.6 kg compared to 2.3 kg with dulaglutide 1.5 mg. Even the lower dose of semaglutide (0.5 mg) produced greater weight loss (3.5 kg) than the higher dose of dulaglutide. These differences were statistically significant and clinically meaningful for most patients.
| Trial/Medication | Dose | Duration | Weight Loss (kg) | Sample Size |
|---|---|---|---|---|
| SUSTAIN-7 | 40 weeks | 1,201 | ||
| Semaglutide | 0.5 mg | 40 weeks | -3.5 kg | 300 |
| Semaglutide | 1.0 mg | 40 weeks | -4.6 kg | 300 |
| Dulaglutide | 0.75 mg | 40 weeks | -1.9 kg | 299 |
| Dulaglutide | 1.5 mg | 40 weeks | -2.3 kg | 302 |
Individual SUSTAIN program trials for semaglutide consistently showed strong weight loss across diverse patient populations. SUSTAIN-1 demonstrated 3.5-5.6 kg weight loss with semaglutide monotherapy over 30 weeks (Sorli et al., Diabetes Care, 2017). SUSTAIN-6, the cardiovascular outcomes trial, showed sustained weight loss of 3.6-4.9 kg over 104 weeks, indicating durability of the weight loss effect (Marso et al., NEJM, 2016).
The REWIND trial, dulaglutide's major cardiovascular outcomes study, showed more modest weight loss of 1.46 kg compared to placebo over a median follow-up of 5.4 years (Gerstein et al., Lancet, 2019). While this demonstrates long-term weight maintenance, the magnitude is substantially less than what has been observed with semaglutide.
Glycemic control also favored semaglutide in head-to-head comparisons. In SUSTAIN-7, semaglutide 1.0 mg reduced HbA1c by 1.8% compared to 1.4% with dulaglutide 1.5 mg. More patients achieved HbA1c targets below 7% with semaglutide (84% vs 67% for the highest doses). These differences remained statistically significant throughout the study period.
Real-world evidence supports the clinical trial findings. A large retrospective analysis of electronic health records from over 20,000 patients showed that semaglutide users achieved greater weight loss (4.2% vs 2.8% body weight reduction) and better glycemic control compared to dulaglutide users over 12 months (Blonde et al., Diabetes, Obesity and Metabolism, 2022).
Both medications share similar side effect profiles typical of GLP-1 receptor agonists, with gastrointestinal effects being the most common. However, important differences exist in the frequency and severity of certain adverse events that can influence treatment selection and patient adherence.
Nausea represents the most frequently reported side effect for both drugs, but the incidence and severity patterns differ. In the SUSTAIN-7 direct comparison trial, nausea occurred in 18-20% of semaglutide patients compared to 12-16% of dulaglutide patients. However, the temporal pattern of nausea differs significantly between the medications.
Semaglutide-associated nausea typically peaks during the first 4-8 weeks of treatment and during dose escalations, then tends to diminish as patients develop tolerance. The prescribing information indicates that most nausea events are mild to moderate and resolve within several weeks (Novo Nordisk, 2023). Dulaglutide shows a more consistent but potentially longer-lasting nausea profile, with some patients experiencing persistent mild nausea throughout treatment.
| Side Effect | Semaglutide (SUSTAIN trials) | Dulaglutide (AWARD trials) |
|---|---|---|
| Nausea | 16-20% | 12-21% |
| Vomiting | 5-9% | 6-13% |
| Diarrhea | 8-12% | 8-15% |
| Constipation | 5-7% | 4-10% |
| Injection site reactions | 1-2% | 2-4% |
| Hypoglycemia (with insulin) | 15-25% | 10-40% |
Injection site reactions show notable differences between the medications. Dulaglutide's Fc fusion protein structure may increase immunogenicity risk, leading to slightly higher rates of injection site reactions including redness, swelling, and itching. The AWARD program trials reported injection site reactions in 2-4% of dulaglutide patients compared to 1-2% with semaglutide (Wysham et al., Diabetes Care, 2014).
Pancreatitis represents a rare but serious potential adverse event for both medications. Post-marketing surveillance data suggests similar low incidence rates, with approximately 2-4 cases per 10,000 patient-years for both drugs. The FDA requires warnings about pancreatitis risk for all GLP-1 receptor agonists, and patients should be counseled about symptoms such as severe abdominal pain.
Tolerability during dose titration varies between the medications. Semaglutide follows a standard escalation from 0.25 mg to 0.5 mg to 1.0 mg (and potentially 2.0 mg) over 8-16 weeks. This gradual titration helps minimize gastrointestinal side effects. Dulaglutide starts at 0.75 mg weekly and can be increased to 1.5 mg, 3.0 mg, or 4.5 mg based on glycemic response and tolerance.
Discontinuation rates due to adverse events provide insight into real-world tolerability. Meta-analyses suggest slightly higher discontinuation rates with semaglutide (8-12%) compared to dulaglutide (6-10%), primarily driven by gastrointestinal intolerance during the initial treatment period (Kristensen et al., Diabetes, Obesity and Metabolism, 2019).
Both medications carry theoretical risks of thyroid C-cell tumors based on rodent studies, though no causal relationship has been established in humans. Patients with personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2 should not use either medication.
Cost considerations significantly impact medication access and long-term adherence for diabetes medications. Both Ozempic and Trulicity carry substantial price tags as brand-name biologics, but various options exist to improve affordability including manufacturer programs, insurance coverage, and compounded alternatives.
Brand-name pricing for both medications remains high without insurance coverage. Ozempic typically costs $900-1,000 per month for a 1.5 mL pen containing four weekly doses. Trulicity pricing ranges from $800-900 monthly for the standard 0.75 mg or 1.5 mg pens. These prices can vary significantly based on pharmacy and geographic location, but both represent substantial monthly expenses for uninsured patients.
Insurance coverage varies widely between plans and often requires prior authorization. Most commercial insurance plans cover both medications on their formularies, but patients may face high copayments ranging from $25-100 monthly even with good coverage. Medicare Part D coverage depends on the specific plan, with many requiring step therapy or quantity limits.
Manufacturer assistance programs provide some relief for eligible patients. Novo Nordisk offers the Ozempic Savings Card, which can reduce copayments to as low as $25 per month for commercially insured patients. Eli Lilly provides the Trulicity Savings Card with similar benefits. However, these programs exclude patients with government insurance like Medicare or Medicaid.
| Option | Ozempic Cost | Trulicity Cost | Notes |
|---|---|---|---|
| Brand (No Insurance) | $900-1,000/month | $800-900/month | Varies by pharmacy |
| With Commercial Insurance | $25-100/month | $25-100/month | Depends on plan |
| Manufacturer Savings Card | $25/month | $25/month | Commercial insurance only |
| Compounded (Telehealth) | $200-400/month | $200-400/month | Requires physician oversight |
Compounded versions of both medications have emerged as cost-effective alternatives through physician-supervised telehealth providers. Compounded semaglutide from FormBlends and similar providers typically costs $200-400 monthly, representing significant savings compared to brand pricing. These compounded formulations use the same active pharmaceutical ingredients but are prepared by specialized compounding pharmacies.
The FDA has clarified that compounding of GLP-1 medications is permissible when done by licensed pharmacies under physician supervision. However, patients should ensure their provider sources from FDA-registered facilities that follow proper quality standards. FormBlends works exclusively with FDA-registered 503B compounding facilities that undergo regular inspections and testing.
International pharmacy options exist but carry risks including counterfeit medications, lack of FDA oversight, and potential customs issues. While some patients report significant cost savings through international sources, the safety and efficacy of these products cannot be guaranteed.
Long-term cost considerations favor finding sustainable access solutions. Both medications typically require ongoing use to maintain benefits, making affordability important for treatment success. Patients should discuss all available options with their healthcare providers, including manufacturer programs, insurance appeals, and legitimate compounded alternatives.
Both Ozempic and Trulicity offer convenient weekly dosing, but their titration schedules and maximum doses differ in ways that can impact treatment individualization and optimization. Understanding these dosing differences helps physicians and patients select the most appropriate medication based on glycemic goals and tolerance.
Semaglutide follows a standardized dose escalation designed to minimize gastrointestinal side effects while achieving therapeutic levels. The FDA-approved titration schedule begins with 0.25 mg weekly for four weeks, increases to 0.5 mg weekly for at least four weeks, then can advance to 1.0 mg weekly based on glycemic response. A 2.0 mg weekly dose was approved in 2022 for patients requiring additional glycemic control.
The 0.25 mg starting dose for semaglutide is considered a "run-in" dose that helps patients adapt to GLP-1 effects without providing substantial therapeutic benefit. Most patients require advancement to 0.5 mg or higher for meaningful HbA1c reduction. Clinical trials suggest that 1.0 mg weekly provides optimal balance of efficacy and tolerability for most patients.
| Week | Semaglutide (Ozempic) | Dulaglutide (Trulicity) |
|---|---|---|
| 1-4 | 0.25 mg weekly | 0.75 mg weekly |
| 5-8 | 0.5 mg weekly | 0.75 mg or increase to 1.5 mg |
| 9+ | 1.0 mg weekly (can increase to 2.0 mg) | 1.5 mg weekly (can increase to 3.0 or 4.5 mg) |
| Maximum Dose | 2.0 mg weekly | 4.5 mg weekly |
Dulaglutide starts at a higher therapeutic dose of 0.75 mg weekly, which provides immediate clinical benefit but may increase initial side effects. This starting dose is already within the therapeutic range, unlike semaglutide's sub-therapeutic starting dose. Patients can remain at 0.75 mg if achieving glycemic targets, or increase to 1.5 mg after four weeks for additional benefit.
Higher dose options give dulaglutide more flexibility for patients requiring intensive glycemic control. The 3.0 mg and 4.5 mg doses were approved based on AWARD-11 trial data showing superior HbA1c reduction compared to lower doses (Frias et al., Lancet, 2021). These higher doses particularly benefit patients with baseline HbA1c above 9% who need substantial glycemic improvement.
Injection administration differs between the medications in clinically relevant ways. Ozempic uses a dial pen that allows dose adjustment and requires patients to attach needles before each injection. This system provides dosing flexibility but requires more steps and dexterity. Trulicity comes as a pre-filled, single-use pen with a hidden needle that activates with a simple button press.
Storage requirements are similar for both medications. Unopened pens should be refrigerated at 36-46°F but can be stored at room temperature for up to 56 days for Ozempic and 14 days for Trulicity once removed from refrigeration. Both can be kept at room temperature for up to 56 days after first use, making travel and daily use convenient.
Missed dose instructions differ slightly between medications. If a weekly Ozempic dose is missed, it should be administered as soon as possible within 5 days, then resume the regular weekly schedule. For Trulicity, missed doses can be taken within 3 days of the scheduled dose. If more time has passed for either medication, patients should skip the missed dose and resume their regular schedule.
Dose adjustments for special populations require consideration. Both medications should be used cautiously in patients with severe renal impairment, though no dose adjustment is typically required. Neither medication has been studied in patients with severe hepatic impairment. Age alone does not require dose modification for either drug, though elderly patients may need more gradual titration due to increased sensitivity to side effects.
The choice between Ozempic and Trulicity depends on individual patient factors including weight loss goals, side effect tolerance, injection preferences, and cost considerations. Both medications effectively manage type 2 diabetes, but their distinct profiles make them suitable for different clinical scenarios.
Ozempic emerges as the preferred choice for patients whose primary goals include substantial weight loss alongside glycemic control. The superior weight loss demonstrated in clinical trials makes semaglutide particularly valuable for overweight or obese patients with type 2 diabetes. Patients seeking 10+ pounds of weight loss over 6-12 months may find Ozempic's efficacy profile more aligned with their goals.
Trulicity may be preferable for patients who prioritize injection convenience and potentially better initial tolerability. The single-use, hidden-needle design appeals to patients with needle anxiety or dexterity limitations. Additionally, patients who experience significant gastrointestinal side effects with other GLP-1 medications might tolerate Trulicity's different pharmacokinetic profile better.
Cost considerations can significantly influence medication selection. Patients with excellent insurance coverage and low copayments can choose based purely on clinical factors. However, those facing high out-of-pocket costs should explore compounded alternatives or manufacturer assistance programs. A free physician assessment can help determine eligibility for cost-effective compounded options.
Clinical scenarios favoring Ozempic include patients with BMI above 30, those who have struggled with weight loss on other diabetes medications, and individuals willing to tolerate potentially higher initial nausea for greater long-term weight benefits. The medication's proven cardiovascular benefits also make it suitable for patients with established cardiovascular disease.
Trulicity works well for elderly patients who may benefit from simpler injection procedures, those with a history of gastrointestinal intolerance to other GLP-1 medications, and patients requiring higher maximum doses for glycemic control. The medication's longer track record and extensive real-world experience provide confidence for conservative prescribers.
Switching between medications is possible and sometimes beneficial. Patients experiencing inadequate weight loss on Trulicity might benefit from switching to Ozempic, while those with persistent nausea on Ozempic could try Trulicity. Any medication switches should be done under physician supervision with appropriate monitoring.
Ultimately, the decision should involve shared decision-making between patient and physician, considering individual health goals, medical history, and personal preferences. Both medications represent effective options for type 2 diabetes management, and the "best" choice varies by individual circumstances.

Yes, you can switch between these medications, but it should be done under physician supervision. Most doctors recommend stopping one medication and starting the other at its recommended starting dose rather than attempting dose conversions. Monitor for side effects during the transition period, as your body may respond differently to the new medication.
Clinical trial data consistently shows Ozempic (semaglutide) produces greater weight loss than Trulicity (dulaglutide). In the direct comparison SUSTAIN-7 trial, semaglutide 1.0 mg resulted in 4.6 kg weight loss compared to 2.3 kg with dulaglutide 1.5 mg over 40 weeks. However, individual responses vary, and both medications can support meaningful weight loss when combined with lifestyle modifications.
Both medications have demonstrated cardiovascular benefits in dedicated outcome trials. Ozempic showed 26% reduction in major adverse cardiovascular events in the SUSTAIN-6 trial, while Trulicity demonstrated 12% reduction in the REWIND trial. Both are considered appropriate for patients with established cardiovascular disease, though the magnitude of benefit may differ.
Both medications have similar side effect profiles, with nausea being the most common issue for both. Some patients find Trulicity slightly better tolerated initially, while others prefer Ozempic's side effect pattern. The best way to determine individual tolerance is through careful trial under medical supervision, starting with the lowest effective dose.
Compounded versions use the same active pharmaceutical ingredients as brand-name medications and can be equally effective when sourced from reputable, FDA-registered compounding pharmacies. However, compounded medications undergo different quality control processes than brand-name drugs. Patients should work with providers who source from established compounding facilities with proper oversight and testing protocols.
FormBlends offers physician-supervised access to compounded semaglutide and dulaglutide at competitive pricing. Our medical team provides personalized treatment plans and ongoing support to help you achieve your health goals safely and effectively.
Start your free consultation today to learn which medication might be right for your specific needs and circumstances.
This article is for educational purposes only and does not constitute medical advice. The information provided should not replace consultation with a qualified healthcare provider. Individual responses to medications vary, and treatment decisions should always be made in consultation with a physician who can evaluate your specific medical history, current medications, and health status.
Both Ozempic and Trulicity are prescription medications that require medical supervision. Do not start, stop, or change dosing without consulting your healthcare provider. If you experience severe side effects or signs of pancreatitis, seek immediate medical attention.
FormBlends provides physician-supervised care and does not recommend self-medication or unsupervised use of any prescription medications.
This article is for informational purposes only and does not constitute medical advice, diagnosis, or treatment. The information provided has been reviewed by licensed healthcare professionals but should not replace a consultation with your physician. Individual results vary. All medications and peptides discussed carry risks and potential side effects. Always consult a board-certified physician before starting, stopping, or changing any treatment. FormBlends provides physician-supervised telehealth services; all prescriptions require physician approval based on individual medical evaluation.
Medical Disclaimer: This content is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before starting, stopping, or changing any medication or treatment. FormBlends articles are reviewed by licensed physicians but are not a substitute for a personal medical consultation.
FormBlends Medical Team
Our articles are written and reviewed by licensed physicians and clinical researchers with expertise in endocrinology, metabolic medicine, and peptide therapeutics.
Ready to get started?
Physician-supervised GLP-1 and peptide therapy, delivered to your door.
Start Your ConsultationCompare Ozempic, Mounjaro, and Trulicity for diabetes. Expert analysis of efficacy, pricing, and side effects. Find the best GLP-1 medication for your needs.
Provider ComparisonsOzempic and Wegovy both contain semaglutide, but they're approved for different purposes. Learn which one is right for your weight loss goals in this complete comparison.
Provider ComparisonsOzempic and Mounjaro are both injectable medications used for weight loss, but they work differently. Compare semaglutide vs tirzepatide to find your best fit.
Provider ComparisonsComparing Ozempic (semaglutide) and Zepbound (tirzepatide) for weight loss. Learn about differences in mechanism, efficacy, side effects, and cost.
Provider ComparisonsCompounded semaglutide and Ozempic use the same active ingredient at vastly different prices. Learn how they compare in quality, efficacy, safety, and cost.
Provider ComparisonsOzempic and Saxenda are both GLP-1 injections used for weight loss. Compare their efficacy, dosing schedules, side effects, costs, and which is best for you.
GLP-1 & Peptide Expert
Hi! I'm your GLP-1 & peptide expert.
Ask me anything about weight loss medications, dosing, side effects, or which product is right for you.