Executive Summary
Delta Sleep Inducing Peptide (DSIP) is a nine-amino-acid neuropeptide first isolated in 1974 from the cerebral venous blood of sleeping rabbits. Over the past five decades, it has emerged as one of the most intriguing molecules in sleep research, with documented effects extending well beyond sleep into pain modulation, stress adaptation, opioid withdrawal, and neuroprotection.
Key Takeaways
- DSIP is a naturally occurring nonapeptide (nine amino acids) that promotes slow-wave delta sleep without suppressing REM sleep
- Clinical trials have demonstrated efficacy in opioid and alcohol withdrawal symptom management, with response rates of 87-97%
- The peptide modulates pain perception through interactions with endogenous opioid systems
- DSIP influences the HPA axis, with documented effects on ACTH and cortisol levels in animal models
- Research dosing typically ranges from 100-300 mcg administered subcutaneously in evening hours
DSIP's amino acid sequence - Trp-Ala-Gly-Gly-Asp-Ala-Ser-Gly-Glu (WAGGDASGE) - makes it one of the smallest bioactive peptides known to influence sleep architecture. Unlike benzodiazepines and other conventional sleep medications that alter the natural rhythm of sleep stages, DSIP appears to enhance slow-wave (delta) sleep while preserving or even supporting rapid eye movement (REM) sleep. This profile has made it a subject of ongoing research interest, particularly among those looking for compounds that promote genuinely restorative sleep rather than pharmacologically induced unconsciousness.
The research landscape for DSIP is both rich and complicated. Early clinical trials in the 1980s demonstrated meaningful effects on withdrawal symptoms from alcohol and opiates, with response rates exceeding 85% in some studies. Pain researchers documented benefits in patients with chronic pain conditions. And sleep studies, while producing mixed results, consistently pointed toward DSIP's ability to influence slow-wave sleep duration and sleep efficiency.
Yet the peptide remains, in the words of one prominent review, "a still unresolved riddle." No specific receptor has been conclusively identified. The gene encoding DSIP has not been isolated. And the precise mechanisms through which this tiny peptide exerts its wide-ranging effects continue to be debated. What is clear, based on decades of both animal and human research, is that DSIP interacts with multiple signaling systems, including GABAergic, opioidergic, and hypothalamic-pituitary-adrenal (HPA) axis pathways.
This report provides a thorough examination of the published research on DSIP. We'll cover its discovery and molecular biology, detail its effects on sleep architecture and the endocrine system, review the clinical evidence for pain and withdrawal applications, discuss dosing protocols used in research settings, and address safety considerations. Throughout, we'll identify where the evidence is strong and where significant gaps remain.
Key Takeaways
- DSIP is a naturally occurring nonapeptide (nine amino acids) that promotes slow-wave delta sleep without suppressing REM sleep
- Clinical trials have demonstrated efficacy in opioid and alcohol withdrawal symptom management, with response rates of 87-97%
- The peptide modulates pain perception through interactions with endogenous opioid systems
- DSIP influences the HPA axis, with documented effects on ACTH and cortisol levels in animal models
- Research dosing typically ranges from 100-300 mcg administered subcutaneously in evening hours
- DSIP is not FDA-approved; long-term safety data are limited, and the specific receptor remains unidentified
For readers interested in other peptides that influence sleep and circadian biology, our Peptide Hub covers related compounds including Pinealon and Epithalon. Those exploring stress-modulating peptides may also find our reports on Selank and Semax relevant.
The Science of Sleep: Why Delta Waves Matter
A Primer on Brain Waves During Sleep
To appreciate what DSIP does - and why it has attracted so much research interest - you first need to understand the electrical language of sleep. The brain generates different types of electrical oscillations depending on its state, and these oscillations aren't just byproducts of neural activity. They're functional signals that coordinate processes across brain regions.
During waking hours, the brain produces predominantly beta waves (13-30 Hz), associated with active thinking and problem solving, and alpha waves (8-13 Hz), associated with relaxed wakefulness. As you transition into sleep, these fast, low-amplitude waves give way to progressively slower, higher-amplitude patterns.
Theta waves (4-7 Hz) dominate light sleep (Stage N1) and represent the brain's transition from conscious processing to sleep-mode operations. Sleep spindles - brief bursts of 12-14 Hz activity lasting 0.5-2 seconds - appear during Stage N2 and are thought to play a role in memory consolidation and sensory gating (blocking external stimuli from reaching consciousness).
Delta waves (0.5-4 Hz) are the slowest and largest brain waves, dominating Stage N3 sleep. These massive, synchronized oscillations represent something remarkable: large populations of cortical neurons firing in coordinated "up states" and "down states," creating waves of activity that sweep across the brain's surface. This coordinated firing pattern serves multiple critical functions that no other brain state can replicate.
What Happens During Delta Sleep
Stage N3 slow-wave sleep, characterized by delta wave dominance, is when the brain and body perform their most intensive repair and maintenance operations. Research over the past two decades has revealed just how much depends on this deepest sleep stage.
Growth Hormone Release: The largest pulse of growth hormone (GH) secretion occurs during the first bout of slow-wave sleep, typically within 60-90 minutes of sleep onset. This GH pulse is so tightly linked to delta sleep that disrupting slow-wave sleep with acoustic stimulation can abolish it entirely. Growth hormone during sleep drives tissue repair, muscle recovery, bone metabolism, and fat mobilization. This connection between delta sleep and GH release is one reason why DSIP's effects on sleep architecture have attracted the attention of researchers interested in recovery and longevity. For those specifically interested in growth hormone optimization, compounds like MK-677 and CJC-1295/Ipamorelin act more directly on the GH axis, while DSIP may support GH release indirectly through sleep architecture enhancement.
Immune System Activation: Slow-wave sleep is the period of peak immune activity during the sleep cycle. Pro-inflammatory cytokines like interleukin-1 (IL-1) and tumor necrosis factor alpha (TNF-alpha) increase during delta sleep, promoting immune surveillance and pathogen defense. Natural killer cell activity peaks. T-cell function is enhanced. This immune-sleep connection explains why sleep deprivation, particularly the loss of deep sleep, leads to increased susceptibility to infection and impaired vaccine responses.
Glymphatic Clearance: Perhaps the most significant discovery in sleep science in recent years is the glymphatic system, a brain-wide waste clearance pathway that becomes dramatically more active during slow-wave sleep. During delta sleep, the interstitial space between brain cells expands by approximately 60%, allowing cerebrospinal fluid to flow through brain tissue and flush out metabolic waste products. Among the waste products cleared during this process is beta-amyloid, the protein that accumulates in Alzheimer's disease. Studies have shown that even a single night of sleep deprivation increases beta-amyloid accumulation in the brain, while chronic sleep disruption accelerates amyloid plaque formation in animal models.
Memory Consolidation: While REM sleep has traditionally received more attention for its role in memory, slow-wave sleep plays an equally important but distinct role. During delta sleep, the hippocampus "replays" experiences from the day, transferring information to the neocortex for long-term storage. This hippocampal-cortical dialogue, coordinated by the slow oscillations of delta waves, is essential for declarative memory consolidation - the ability to remember facts and events.
Synaptic Homeostasis: The synaptic homeostasis hypothesis, proposed by Giulio Tononi and Chiara Cirelli, suggests that slow-wave sleep serves a critical function in maintaining synaptic balance. During waking hours, learning and experience strengthen synaptic connections throughout the brain, gradually increasing the overall synaptic weight. Delta sleep "resets" these connections through a process of synaptic downscaling, preserving the relative strength of important connections while reducing overall synaptic load. Without this nightly reset, synaptic saturation would eventually impair the brain's ability to learn and process new information.
The Delta Sleep Deficit
Slow-wave sleep declines substantially with age. A healthy 20-year-old typically spends 15-25% of total sleep time in Stage N3. By age 60, this can decline to 5-10% or less, with some older adults showing virtually no delta wave activity. This age-related loss of deep sleep contributes to decreased growth hormone production, impaired immune function, cognitive decline, and increased vulnerability to neurodegenerative disease. Any compound that could genuinely restore delta sleep in aging populations would address multiple aspects of age-related physiological decline simultaneously. This is a key part of the theoretical appeal of DSIP research.
Delta Sleep and Modern Life
Beyond aging, modern lifestyles impose multiple insults on delta sleep specifically. Several common factors preferentially reduce slow-wave sleep while leaving lighter sleep stages relatively intact:
- Alcohol: While alcohol promotes sleep onset, it dramatically suppresses slow-wave sleep in the second half of the night, reducing the total restorative sleep per sleep period
- Chronic stress: Elevated cortisol, particularly in the evening, selectively impairs delta sleep generation. The HPA axis hyperactivation seen in chronic stress creates a vicious cycle where poor sleep further elevates stress hormones
- Blue light exposure: Evening screen use suppresses melatonin production and delays sleep onset, compressing the early-night period when the most intense slow-wave sleep normally occurs
- Caffeine: Even when consumed early in the day, caffeine's adenosine receptor antagonism can reduce slow-wave sleep depth and duration at night, as adenosine accumulation is one of the primary drivers of sleep pressure and delta wave generation
- Sleep medications: Paradoxically, the most commonly prescribed sleep drugs (benzodiazepines, Z-drugs) suppress slow-wave sleep. They increase total sleep time while reducing sleep quality, trading hours of restorative deep sleep for hours of lighter, less beneficial sleep stages
- Sleep apnea: Obstructive sleep apnea fragments sleep architecture, with frequent arousals preventing sustained delta sleep episodes. Even treated sleep apnea patients often show persistent slow-wave sleep deficits
This convergence of factors means that many adults, particularly those over 40, are chronically delta-sleep-deprived even when they feel they're sleeping "enough" hours. The total time in bed may be adequate, but the quality composition of that sleep - specifically the percentage spent in Stage N3 - is insufficient for optimal physiological maintenance.
This context helps explain why DSIP, despite its mixed clinical trial results and unresolved mechanistic questions, continues to attract research interest. A compound that selectively enhances delta sleep without suppressing REM, without creating dependence, and without producing morning impairment would fill a gap that no currently approved medication addresses. Whether DSIP truly fills that gap remains an open question, but the question itself is highly relevant to contemporary sleep science.
Measuring Sleep Architecture: How We Know What DSIP Does
The primary tool for measuring sleep architecture is polysomnography (PSG), which combines multiple physiological measurements recorded simultaneously during sleep:
- Electroencephalography (EEG): Records brain electrical activity through scalp electrodes, allowing identification of sleep stages based on characteristic wave patterns
- Electrooculography (EOG): Tracks eye movements to identify REM sleep periods
- Electromyography (EMG): Measures muscle tone, which decreases progressively through sleep stages and reaches its lowest during REM (muscle atonia)
- Additional channels: May include respiratory effort, airflow, oxygen saturation, heart rate, and limb movements
In DSIP research, EEG analysis is the primary outcome measure. Researchers examine several specific parameters:
- Delta power: The total electrical power in the 0.5-4 Hz frequency band, measured by spectral analysis. Higher delta power indicates more intense slow-wave sleep
- Time in N3: The total minutes spent in Stage N3 as a percentage of total sleep time
- Delta sleep efficiency: The ratio of actual N3 time to the opportunity for N3 (typically concentrated in the first half of the night)
- Sleep onset latency: Time from lights-out to the first epoch of any sleep stage
- N3 onset latency: Time from sleep onset to the first epoch of Stage N3
- Sleep architecture integrity: The normal cycling pattern between sleep stages, assessed qualitatively through hypnogram analysis
The DSIP studies from the 1980s used the technology available at that time, which was adequate for gross sleep staging but lacked the spectral analysis resolution available with modern digital EEG systems. This is a consideration when interpreting historical DSIP data - the studies could reliably determine whether subjects were in N3, but they could not perform the fine-grained spectral analyses that would reveal more subtle effects on delta power density within N3 periods.
DSIP in the Broader Context of Sleep Peptide Research
The Search for Endogenous Sleep Factors
DSIP's discovery in 1974 was part of a broader research effort to identify endogenous sleep factors - substances produced by the body that regulate sleep. The concept of chemical sleep regulation dates back to the early 20th century, when Henri Pieron demonstrated that cerebrospinal fluid from sleep-deprived dogs could induce sleep when injected into rested animals. This experiment, published in 1913, established the principle that sleep produces (or depletes) chemical signals that drive future sleep.
Over the following decades, multiple candidate sleep factors were identified, each contributing to different aspects of sleep regulation:
| Sleep Factor | Year Identified | Primary Sleep Effect | Mechanism |
|---|---|---|---|
| Factor S (muramyl peptides) | 1975 | Slow-wave sleep promotion | IL-1 / immune pathway activation |
| DSIP | 1974 | Delta wave enhancement | Multiple systems (unresolved) |
| Adenosine | 1984 (sleep role) | Sleep pressure / homeostatic drive | A1 / A2A receptor activation |
| Prostaglandin D2 | 1988 | NREM sleep promotion | DP1 receptor / adenosine pathway |
| Oleamide | 1995 | Sleep induction | GABAergic / serotonergic modulation |
| Orexin/Hypocretin | 1998 | Wake promotion (loss causes narcolepsy) | OX1 / OX2 receptors |
| Cortistatin | 1996 | Slow-wave sleep enhancement | Somatostatin receptors + unique mechanisms |
DSIP occupies a unique position in this landscape. Unlike adenosine, whose sleep-promoting mechanism through A1/A2A receptors is well characterized, and unlike orexin, whose wake-promoting role through OX1/OX2 receptors is clearly defined, DSIP lacks a confirmed receptor. Yet it was one of the first sleep factors isolated and remains one of the most extensively studied in terms of clinical applications.
The Two-Process Model and Where DSIP Fits
Modern sleep science understands sleep regulation through the two-process model proposed by Alexander Borbely in 1982. This model describes two independent but interacting processes:
Process S (Sleep Homeostatic Drive): A pressure to sleep that builds progressively during wakefulness and dissipates during sleep. Adenosine accumulation is a primary molecular correlate of Process S. The longer you've been awake, the stronger the drive to sleep, and the more intense the initial slow-wave sleep when sleep finally occurs.
Process C (Circadian Rhythm): A 24-hour oscillation generated by the suprachiasmatic nucleus (SCN) that modulates sleep propensity independently of how long you've been awake. Melatonin secretion is a primary output of Process C. The circadian system promotes wakefulness during the biological day and facilitates sleep during the biological night.
Normal sleep requires both processes to be aligned: sufficient homeostatic sleep pressure (Process S) combined with appropriate circadian timing (Process C). When these processes are misaligned - as in jet lag, shift work, or circadian rhythm disorders - sleep quality suffers even when total sleep time may be adequate.
Where does DSIP fit in this framework? The evidence suggests it may influence both processes, though through different mechanisms:
- Process S effects: DSIP's enhancement of delta sleep may reflect augmentation of the homeostatic sleep drive, potentially by modulating the sensitivity of neural circuits to accumulated sleep pressure signals. The observation that DSIP has greater effects in sleep-deprived or poor-sleeping individuals supports this interpretation.
- Process C effects: DSIP's stimulation of pineal melatonin production provides a pathway for circadian influence, reinforcing the natural timing signal for sleep onset.
- HPA axis modulation: Cortisol, while not traditionally included in the two-process model, acts as a powerful modulator of both processes. Elevated cortisol opposes both homeostatic sleep pressure and circadian sleep signals. DSIP's ability to attenuate HPA axis activity may facilitate sleep by removing a hormonal obstacle to both Process S and Process C expression.
Peptide Approaches to Sleep: A Growing Field
DSIP is not the only peptide being studied for sleep applications. The field of sleep peptide research has expanded considerably, with several compounds targeting different aspects of sleep regulation:
Orexin receptor antagonists (DORAs): Suvorexant (Belsomra) and lemborexant (Dayvigo) are FDA-approved medications that block wake-promoting orexin receptors. Unlike benzodiazepines, they do not suppress slow-wave sleep, though they also do not specifically enhance it. Their mechanism is fundamentally different from DSIP's: they remove a wake signal rather than adding a sleep signal.
Pinealon and other pineal peptides: As discussed in the comparison section, Pinealon targets circadian rhythm normalization through pineal gland support. The Pinealon research report details how this tripeptide may help restore normal melatonin production patterns, particularly in aging individuals with declining pineal function.
Epitalon: Epithalon (Epitalon), another peptide studied by Russian researchers, has been investigated for effects on pineal melatonin production and telomerase activation. While its primary research focus is on aging and telomere biology, its effects on pineal function connect it to sleep regulation.
GHRH (Growth Hormone Releasing Hormone): GHRH administration has been shown to enhance slow-wave sleep in humans, an effect that appears independent of its growth hormone-releasing properties. This suggests the hypothalamic peptide has direct sleep-promoting actions, possibly through effects on GABAergic interneurons in sleep-regulatory brain regions.
Galanin: This neuropeptide is concentrated in sleep-active neurons of the ventrolateral preoptic area (VLPO), the brain's primary "sleep switch." Galanin-expressing neurons inhibit wake-promoting centers, and the peptide itself has been shown to promote NREM sleep when administered centrally.
The diversity of these peptide approaches reflects the complexity of sleep regulation. No single peptide controls all aspects of sleep, just as no single neurotransmitter controls all aspects of brain function. The most effective sleep optimization strategies may ultimately involve combinations of peptides targeting different nodes in the sleep regulatory network - an approach that the Biohacking Hub explores in broader context.
Why DSIP Research Stalled (And Why It's Resuming)
The pace of DSIP research slowed considerably after the 1990s, and understanding why helps contextualize the current state of the field. Several factors contributed to the research lull:
The receptor problem: In an era when molecular biology was revolutionizing pharmacology through receptor cloning and gene identification, DSIP's lack of a confirmed receptor made it an outlier. Funding agencies and pharmaceutical companies preferred targets with clear molecular mechanisms, and DSIP's "systems modulatory" profile did not fit the prevailing "one drug, one target" paradigm.
Gene identification failure: The inability to identify a DSIP gene raised persistent questions about whether DSIP was truly an endogenous peptide or an artifact of the purification process. Without a gene, standard molecular tools (knockouts, transgenic overexpression, gene expression mapping) could not be applied.
Mixed clinical results: The variability in human sleep trial outcomes, particularly the disconnect between objective and subjective improvement in the Schneider-Helmert trial, dampened enthusiasm for clinical development.
Z-drug competition: The approval of zolpidem (Ambien) in 1992 and zaleplon (Sonata) in 1999 created commercially successful sleep medications that, while imperfect, provided reliable sedation. The pharmaceutical industry's interest in novel sleep mechanisms waned as the Z-drug market grew.
More recently, several factors have renewed interest in DSIP:
Recognition of Z-drug limitations: Increasing awareness of the adverse effects of chronic Z-drug use - including dependence, cognitive impairment, falls in elderly patients, and paradoxical sleep architecture disruption - has created demand for mechanistically distinct sleep approaches.
Glymphatic discovery: The identification of the glymphatic system and its dependence on slow-wave sleep has elevated the importance of delta sleep enhancement. Compounds that specifically promote deep sleep are now recognized as potentially relevant to neurodegeneration prevention.
Peptide technology advances: Improved synthesis methods, novel delivery systems (including intranasal and sustained-release formulations), and better understanding of peptide pharmacokinetics have made DSIP more practically workable than it was in the 1980s.
Systems biology perspective: The shift from "one drug, one target" thinking toward network pharmacology and multi-target approaches has made DSIP's promiscuous receptor profile look less like a weakness and more like a feature. Modern systems biology is more comfortable with compounds that modulate networks rather than individual receptors.
Fusion peptide technology: The 2024 publication on DSIP fusion peptides in Frontiers in Pharmacology demonstrates active innovation in DSIP delivery. By fusing DSIP to carrier sequences that facilitate blood-brain barrier crossing, researchers may overcome the stability and delivery challenges that limited earlier clinical applications.
DSIP and the Immune System
Immunomodulatory Research
Beyond its effects on sleep, pain, and stress, DSIP has been investigated for immunomodulatory properties. The connection between sleep peptides and immune function is biologically logical: sleep and immunity are tightly interlinked, with cytokines like IL-1 and TNF-alpha serving dual roles as both immune mediators and sleep-regulatory factors.
Research on DSIP's immune effects has revealed several findings:
Lymphocyte activation: In vitro studies have shown that DSIP can modulate lymphocyte proliferation responses. The direction of modulation appears to depend on the baseline immune state - stimulating activity in suppressed systems while dampening overactivation. This bidirectional pattern is consistent with an immunomodulatory rather than purely immunostimulatory or immunosuppressive profile.
Stress-induced immunosuppression: Animal studies have demonstrated that DSIP pre-treatment can partially prevent stress-induced suppression of immune function. Since chronic stress is a well-established cause of immune dysfunction, DSIP's HPA axis modulation may provide indirect immune support by attenuating the cortisol-mediated immunosuppressive effects of chronic stress.
Natural killer cell activity: Some studies have reported that DSIP influences natural killer (NK) cell activity, though the specific effects and their magnitude vary across experimental conditions. NK cells are critical for immune surveillance against both viral infections and cancer cells, and their activity is known to be strongly influenced by sleep quality - particularly slow-wave sleep duration.
Cytokine modulation: DSIP has been shown to influence the production of several cytokines involved in both immune regulation and sleep control. This dual role is consistent with the peptide's position at the intersection of sleep and immune regulatory networks.
Clinical Relevance of Immune Effects
The clinical relevance of DSIP's immunomodulatory properties remains speculative, but several connections deserve consideration:
- Chronic insomnia is associated with measurable immune dysfunction, including reduced NK cell activity and impaired vaccine responses. If DSIP improves sleep quality, indirect immune benefits would be expected
- The relationship between poor sleep and increased cancer risk has been established in epidemiological studies. Any compound that genuinely restores deep sleep could theoretically support immune surveillance mechanisms
- Autoimmune conditions are frequently accompanied by sleep disruption, creating cycles of immune activation and sleep impairment that are difficult to break. DSIP's combined immunomodulatory and sleep-promoting properties could theoretically address both aspects
It must be emphasized that these clinical applications are theoretical. No clinical trials have evaluated DSIP specifically for immune outcomes, and the translation from in vitro and animal immune data to human clinical benefit requires validation that has not been performed.
DSIP, Aging, and Age-Related Sleep Decline
The Aging Sleep Problem
One of the most consistent changes associated with aging is progressive deterioration of sleep quality, with slow-wave sleep being the most affected component. The decline in delta sleep begins in the third decade of life and accelerates after age 50. By the seventh and eighth decades, many individuals have lost 60-80% of their youthful slow-wave sleep capacity.
This age-related delta sleep decline is not merely a nuisance. It has been linked to:
- Reduced growth hormone secretion: The age-related decline in GH closely parallels the loss of slow-wave sleep, and both contribute to sarcopenia (muscle loss), increased body fat, reduced bone density, and impaired tissue repair
- Cognitive decline: Reduced delta sleep impairs memory consolidation, reduces glymphatic clearance, and accelerates beta-amyloid accumulation. Several studies have identified slow-wave sleep loss as an independent predictor of cognitive decline in older adults
- Immune senescence: The decline in sleep-dependent immune activation contributes to the increased susceptibility to infections and reduced vaccine efficacy seen in older populations
- Hormonal changes: Sleep-dependent hormone secretion patterns (including cortisol, melatonin, and testosterone) are disrupted by poor sleep quality, creating a compound effect on age-related endocrine changes
- Increased inflammation: Poor sleep quality promotes chronic low-grade inflammation (inflammaging), which contributes to virtually every age-related chronic disease
Can DSIP Address Age-Related Sleep Decline?
The theoretical case for DSIP in aging-related sleep decline is compelling on several levels:
Direct delta sleep enhancement: If DSIP genuinely increases slow-wave sleep duration and intensity, it could directly address the primary sleep architecture deficit associated with aging.
Melatonin support: Pineal melatonin production declines with age, contributing to circadian disruption and sleep onset difficulties. DSIP's demonstrated ability to stimulate melatonin secretion from the pineal gland could partially compensate for age-related melatonin decline.
Cortisol normalization: Aging is associated with flattening of the cortisol circadian rhythm, with relatively higher nighttime cortisol levels that impair sleep. DSIP's HPA axis modulatory effects could help restore a more youthful cortisol pattern.
Neuroprotection: DSIP's antioxidant and mitochondrial support properties could provide complementary neuroprotective effects beyond those achieved through sleep enhancement alone.
However, it must be noted that no clinical trials have specifically evaluated DSIP in elderly populations. The existing human data come from studies of younger adults, and the applicability of these findings to older adults with fundamentally different sleep physiology is uncertain. Age-related changes in peptide metabolism, blood-brain barrier permeability, and receptor sensitivity could all influence DSIP's effects in ways that cannot be predicted from younger-population data.
Other peptides with relevance to aging and sleep include Epithalon, which has been studied for telomerase activation and pineal melatonin support, and NAD+, which supports cellular energy metabolism and DNA repair processes that decline with age. The Biohacking Hub at FormBlends provides broader context on peptide-based approaches to age-related physiological changes.
DSIP and Circadian Rhythm Disruption in Aging
Beyond the direct loss of delta sleep, aging brings significant circadian rhythm changes that compound sleep problems. The suprachiasmatic nucleus (SCN), the brain's master circadian clock, undergoes neuronal loss and reduced output with age. This leads to:
- Advanced sleep phase (earlier bedtime and wake time)
- Reduced circadian amplitude (less distinction between day and night physiology)
- Increased circadian fragmentation (less consolidated sleep and wake periods)
- Reduced light sensitivity of the circadian system
DSIP's combined effects on sleep architecture and melatonin production theoretically address both the homeostatic (sleep depth) and circadian (sleep timing) components of age-related sleep decline. This dual action, if clinically validated, would represent a more comprehensive approach than either melatonin supplementation (circadian only) or sedative medication (neither circadian nor architectural improvement).
Practical Considerations for Researchers and Clinicians
Patient Selection Considerations
Based on the available research, certain populations may be more likely to respond to DSIP than others. While formal patient selection criteria have not been established through clinical trials, the existing data suggest several factors that may predict response:
Likely better responders:
- Individuals with objectively reduced slow-wave sleep on polysomnography
- Stress-related insomnia with documented HPA axis dysregulation (elevated evening cortisol)
- Pain-related sleep disruption where opioidergic modulation might benefit both pain and sleep
- Post-withdrawal sleep disruption in individuals recovering from alcohol or opioid dependence
- Age-related sleep quality decline with preserved sleep duration but reduced deep sleep percentage
Likely poorer responders:
- Primary circadian rhythm disorders (better served by melatonin or light therapy)
- Sleep apnea-related sleep disruption (requires airway management, not pharmacological sleep enhancement)
- Good sleepers seeking minor optimization (DSIP shows greater effects in disrupted than normal sleep)
- Individuals on high-dose opioid therapy (cross-tolerance may limit DSIP's effects)
Monitoring Recommendations
For researchers or clinicians considering DSIP in a clinical context, the following monitoring approach is suggested based on DSIP's known pharmacological profile:
Baseline assessment:
- Sleep diary and validated sleep questionnaires (Pittsburgh Sleep Quality Index, Insomnia Severity Index)
- Polysomnography or home sleep testing if available (to quantify baseline sleep architecture)
- Morning cortisol and, ideally, evening salivary cortisol (to assess HPA axis function)
- Basic metabolic panel including fasting glucose (given potential endocrine effects)
- Complete blood count (baseline for monitoring potential immune effects)
During treatment:
- Subjective sleep quality assessment weekly
- Monitoring for side effects, particularly morning grogginess and headache
- Repeat cortisol assessment at 4-6 weeks if baseline was abnormal
- Assessment for any injection site reactions
Follow-up assessment:
- Repeat sleep questionnaires at 4, 8, and 12 weeks
- Follow-up polysomnography at 8-12 weeks if baseline was obtained
- Repeat basic labs at 12 weeks
Integration with Sleep Hygiene
DSIP research should not be considered in isolation from foundational sleep practices. The peptide appears to facilitate rather than force sleep, making it more dependent on supportive sleep conditions than sedative medications. Key sleep hygiene practices that likely enhance DSIP's effects include:
- Consistent sleep schedule: Aligning DSIP administration with a regular bedtime supports both the peptide's circadian effects and its sleep-facilitating properties
- Light exposure management: Morning bright light and evening dim light support the circadian context in which DSIP operates
- Temperature optimization: A cool sleeping environment (65-68 degrees F / 18-20 degrees C) promotes slow-wave sleep independently and may synergize with DSIP's delta sleep enhancement
- Stress management: Techniques that reduce evening cortisol (meditation, progressive relaxation, cognitive behavioral therapy for insomnia) complement DSIP's HPA axis modulation
- Exercise timing: Regular exercise promotes slow-wave sleep, but intense exercise too close to bedtime can elevate cortisol and core body temperature, potentially opposing DSIP's effects. Morning or afternoon exercise is generally preferable
- Caffeine cutoff: Given caffeine's adenosine receptor antagonism and its impact on slow-wave sleep, a strict caffeine cutoff (typically before noon or 2 PM) may be particularly important for maximizing DSIP's delta sleep enhancement
When to Consider Alternative Approaches
DSIP is not appropriate for all sleep complaints, and recognizing its limitations helps guide appropriate use. Alternative approaches may be more suitable in the following situations:
| Sleep Complaint | Primary Approach | DSIP Role |
|---|---|---|
| Circadian misalignment (jet lag, shift work) | Light therapy + melatonin | Adjunctive at best |
| Sleep onset difficulty only | CBT-I, melatonin, or short-acting sedatives | May help if stress-related |
| Sleep maintenance insomnia | CBT-I; evaluate for apnea | Possible benefit if delta sleep is reduced |
| Reduced deep sleep quality | Exercise, sleep optimization | Primary consideration |
| Stress-related sleep disruption | Stress management, CBT-I | Good theoretical fit |
| Pain-related sleep disruption | Pain management + sleep optimization | May address both components |
| Sleep apnea | CPAP/mandibular device | Not appropriate as primary treatment |
| Restless legs syndrome | Iron assessment, dopamine agonists | Not relevant |
| Narcolepsy | Stimulants, sodium oxybate | Not relevant |
For a comprehensive understanding of sleep optimization approaches, including both peptide and non-peptide strategies, the Biohacking Hub provides extensive coverage. Those specifically interested in circadian rhythm approaches may find the Pinealon research report relevant, while stress-related sleep disruption may be better addressed with anxiolytic peptides like Selank.
Future Directions in DSIP Research
Unresolved Questions
Despite five decades of research, several fundamental questions about DSIP remain unanswered. Resolving these would significantly advance both basic sleep science and potential clinical applications:
1. What is the DSIP receptor? The identification of a specific DSIP receptor (or confirmation that no single receptor exists) remains the most important unanswered question. Modern techniques including proximity labeling, CRISPR-based genetic screens, and cryo-electron microscopy of receptor complexes offer new approaches that were unavailable when DSIP was first studied.
2. Does DSIP have a dedicated gene? Next-generation sequencing and bioinformatics approaches could potentially identify a DSIP precursor gene that eluded earlier search methods. Alternatively, if DSIP is indeed a cleavage product of a larger protein with other functions, identifying that parent protein would clarify DSIP's biosynthesis.
3. What explains the human clinical variability? Genetic polymorphisms in DSIP-responsive pathways, baseline differences in sleep architecture, circadian chronotype, and stress hormone status all likely contribute to inter-individual response variability. Identifying biomarkers that predict DSIP response would greatly improve clinical utility.
4. What are the long-term effects? No studies have followed DSIP-treated individuals beyond several months. Long-term effects on endocrine function, immune status, and sleep architecture homeostasis are unknown.
5. How does DSIP cross the blood-brain barrier? While BBB penetration has been demonstrated, the transport mechanism is not characterized. Understanding this mechanism could enable development of more effective delivery strategies.
Emerging Research Approaches
Several current research directions are likely to advance DSIP science in the coming years:
DSIP fusion peptides: The 2024 Frontiers in Pharmacology study on DSIP fusion constructs produced in Pichia pastoris yeast represents a new approach to DSIP delivery. By fusing DSIP to blood-brain barrier-penetrating sequences, researchers may overcome the stability and delivery limitations that have hampered clinical applications. These fusion peptides showed efficacy in PCPA-induced insomnia mouse models, validating the approach.
Sustained-release formulations: Given DSIP's extremely short half-life (7-8 minutes in plasma), sustained-release delivery systems could dramatically improve its pharmacokinetic profile. Technologies such as PLGA microspheres, hydrogels, and nanoparticle encapsulation have been successfully applied to other short-lived peptides and could potentially extend DSIP's effective duration from minutes to hours.
Intranasal delivery: The stroke recovery study that used intranasal DSIP demonstrates the feasibility of nose-to-brain delivery, which bypasses the blood-brain barrier entirely. Intranasal peptide delivery has advanced significantly in recent years, and optimized DSIP intranasal formulations could provide direct CNS access without injection.
Phosphorylated analogues: P-DSIP's dramatically enhanced potency (22% increase in SWS, 81% increase in REM sleep) has not been explored in human studies. Clinical development of P-DSIP or other enhanced DSIP analogues could potentially address the modest effect sizes seen in human trials with native DSIP.
Combination studies: Rigorous evaluation of DSIP in combination with circadian agents (melatonin, Pinealon) and stress-modulating peptides (Selank) could clarify whether multi-peptide approaches are superior to monotherapy.
Modern sleep monitoring: Home-based EEG monitoring devices (like the Dreem headband and similar consumer devices) now make longitudinal sleep architecture monitoring feasible outside sleep laboratories. This technology could enable large-scale studies of DSIP's effects on delta sleep over extended periods, overcoming the sample size limitations of traditional sleep lab studies.
The Broader Implication
DSIP's story is, in many ways, the story of modern sleep pharmacology's limitations. We have drugs that knock people out but damage sleep quality. We have melatonin that times sleep but doesn't deepen it. We have orexin antagonists that remove wake signals but don't actively promote restoration. What we lack - and what DSIP promises, however imperfectly - is a compound that enhances the restorative quality of sleep itself.
Whether DSIP fulfills that promise, or whether a next-generation analogue or entirely different compound ultimately fills this niche, the fundamental need it addresses is real and growing. As populations age, as chronic stress increases, and as the health consequences of poor-quality sleep become better understood, the search for genuine delta sleep enhancers will only intensify.
For those following the evolving field of sleep optimization through peptides and other advanced approaches, the Peptide Hub and Biohacking Hub at FormBlends provide regularly updated resources. The Free Assessment can help determine which research compounds may be most relevant to individual goals and circumstances.
Discovery & Structure
The Rabbit Experiments: How DSIP Was Found
The story of DSIP begins in the early 1970s at the University of Basel in Switzerland, where researchers Marcel Monnier and G.A. Schoenenberger were pursuing an ambitious question: could the chemical signals responsible for sleep be isolated from the blood of sleeping animals?
Their experimental approach was both creative and technically demanding. They electrically stimulated the intralaminar thalamic area of donor rabbits, a brain region known to promote sleep when activated. They then collected cerebral venous blood from these sleeping rabbits using an extracorporeal dialysis system, which allowed them to capture small molecules passing from brain tissue into the blood while filtering out larger proteins and cells.
When this dialysate was infused into the mesodiencephalic ventricle of awake recipient rabbits, something remarkable happened. The recipients showed distinct changes in their electroencephalographic (EEG) patterns: spindle activity increased, and the characteristic high-amplitude, low-frequency delta waves associated with deep sleep appeared. Motor activity decreased simultaneously. A sleep-promoting substance had been isolated from the blood of sleeping animals and successfully transferred to awake ones.
Monnier and Schoenenberger first presented these findings at the 2nd European Congress on Sleep Research in Rome in 1974. Over the next several years, they worked to purify and characterize the active compound, eventually publishing the complete amino acid sequence in the Proceedings of the National Academy of Sciences in 1977.
Figure 1: Timeline of DSIP discovery, from the original 1974 rabbit dialysis experiments through structural characterization and the first clinical applications in the 1980s.
Molecular Structure and Properties
DSIP turned out to be surprisingly small. At just nine amino acids, it is one of the tiniest peptides known to produce significant neurobiological effects. Its complete sequence is:
Trp - Ala - Gly - Gly - Asp - Ala - Ser - Gly - Glu
(Single-letter code: W-A-G-G-D-A-S-G-E)
The molecular weight is approximately 849-850 daltons, placing DSIP well below the blood-brain barrier exclusion threshold of roughly 500,000 daltons for most proteins. Research has confirmed that DSIP does cross the blood-brain barrier, though the exact transport mechanisms remain under investigation. Studies using radioiodinated DSIP in rabbits demonstrated measurable penetration into brain tissue after peripheral administration.
Several structural features deserve attention:
- Amphiphilic character: DSIP contains both hydrophilic (Asp, Ser, Glu) and hydrophobic (Trp, Ala) residues, giving it the ability to interact with both aqueous environments and lipid membranes
- No disulfide bonds: Unlike many bioactive peptides, DSIP lacks cysteine residues and therefore has no disulfide bridges, contributing to its relatively flexible conformation
- Phosphorylated variant: A phosphorylated analogue of DSIP (P-DSIP), with a phosphate group on the serine residue, has been studied and shown to be more potent in some experimental contexts. P-DSIP produced a 22% increase in slow-wave sleep and an 81% increase in paradoxical (REM) sleep in unrestrained rats
- Short half-life: In human plasma, DSIP has a half-life of only 7-8 minutes. In vitro studies show even shorter stability, with a half-life around 15 minutes due to specific aminopeptidase-like enzyme activity
The Half-Life Paradox
One of DSIP's enduring mysteries is how a peptide with a plasma half-life of under 10 minutes can produce effects lasting hours. Researchers have proposed several explanations. One hypothesis suggests DSIP binds to carrier proteins in the blood, protecting it from enzymatic degradation. Another proposes that DSIP may exist as part of a larger precursor molecule that releases active DSIP over time. A third possibility is that DSIP triggers downstream signaling cascades whose effects persist long after the peptide itself has been degraded. None of these hypotheses has been definitively proven.
The DSIP Gene Problem
Perhaps the most significant obstacle to DSIP research has been the failure to identify a dedicated DSIP gene. Most bioactive peptides are produced from larger precursor proteins (pro-peptides) that are cleaved into their active forms. For DSIP, no such precursor gene has been found in any mammalian genome, despite extensive searching.
This has led to persistent uncertainty about whether DSIP is truly an endogenous peptide (produced naturally in the body) or a fragment of a larger, still-unidentified protein. The WAGGDASGE sequence has been found embedded within several larger proteins, but none has been confirmed as a true DSIP precursor. DSIP-like immunoreactivity has been detected in brain tissue and blood of multiple species, including humans, suggesting the peptide or a close analogue is indeed produced endogenously. But the precise biosynthetic pathway remains unknown.
In 2006, a comprehensive review by Kovalzon and Strekalova in the Journal of Neurochemistry described DSIP as "a still unresolved riddle," noting that the lack of a confirmed gene, receptor, and clear mechanism of action places it in an unusual position: widely studied, clearly bioactive, yet fundamentally mysterious.
Tissue Distribution
Despite the gene identification problem, DSIP-like immunoreactivity has been mapped across multiple tissues using antibody-based detection methods. Key locations include:
| Tissue/Region | Detection Method | Relative Concentration |
|---|---|---|
| Hypothalamus | Radioimmunoassay | High |
| Limbic system structures | Immunohistochemistry | Moderate to High |
| Pituitary gland | Radioimmunoassay | Moderate |
| Peripheral blood plasma | Radioimmunoassay | Low (variable) |
| Adrenal glands | Immunohistochemistry | Moderate |
| Gastrointestinal tract | Immunohistochemistry | Low to Moderate |
The concentration of DSIP-like immunoreactivity in plasma shows circadian variation, with some studies reporting higher levels during sleep periods. This observation supports the idea of an endogenous role in sleep regulation, though the measurement variability and specificity of antibodies used in these early studies have been questioned.
Structural Analogues and Modifications
Researchers have synthesized numerous DSIP analogues to improve stability and potency. The most studied modifications include:
- P-DSIP (phosphorylated DSIP): Phosphorylation of Ser-7 dramatically increases biological activity in sleep studies, with 2-5 fold greater potency than native DSIP in some assay systems
- N-Tyr-DSIP: Addition of tyrosine to the N-terminus creates a peptide with enhanced opioid receptor interactions
- D-amino acid substitutions: Replacement of L-amino acids with D-configurations at specific positions can increase enzymatic resistance while retaining activity
- Cyclic analogues: Ring-closed versions of DSIP show improved metabolic stability but variable bioactivity
More recently, researchers have explored DSIP fusion peptides designed to cross the blood-brain barrier more efficiently. A 2024 study published in Frontiers in Pharmacology examined DSIP fusion constructs secreted by Pichia pastoris yeast, testing their efficacy in insomnia mouse models treated with para-chlorophenylalanine (PCPA). These fusion approaches represent a new generation of DSIP research aimed at overcoming the peptide's inherent stability limitations.
Understanding the structural foundation of DSIP is essential context for appreciating its pharmacological profile. For a broader perspective on how peptide structure influences biological function, the Biohacking Hub at FormBlends provides additional background on peptide science fundamentals.
Sleep Architecture Modulation
Understanding Sleep Architecture
Before examining DSIP's effects on sleep, it helps to understand what sleep architecture actually means. Sleep is not a uniform state. It consists of distinct stages that cycle in roughly 90-minute intervals throughout the night, each serving different physiological functions.
Stage N1 (Light Sleep): The transition from wakefulness to sleep, lasting 1-7 minutes. EEG shows theta waves (4-7 Hz). Easily disrupted; accounts for 2-5% of total sleep in healthy adults.
Stage N2 (Intermediate Sleep): Characterized by sleep spindles and K-complexes on EEG. Heart rate slows, body temperature drops. Accounts for roughly 45-55% of total sleep time. Memory consolidation processes begin here.
Stage N3 (Slow-Wave/Delta Sleep): The deepest stage, dominated by high-amplitude delta waves (0.5-2 Hz). This is where the most physical restoration occurs: growth hormone secretion peaks, tissue repair accelerates, immune function is enhanced, and the glymphatic system clears metabolic waste from the brain. Accounts for 15-25% of total sleep in young adults, declining significantly with age.
REM Sleep: Characterized by rapid eye movements, muscle atonia, and vivid dreaming. Critical for emotional processing, memory consolidation, and cognitive function. Accounts for 20-25% of total sleep time.
The key insight for understanding DSIP's appeal is this: most conventional sleep medications (benzodiazepines, Z-drugs) increase total sleep time but actually suppress Stage N3 slow-wave sleep and/or REM sleep. This means they produce more hours of sleep that is, paradoxically, less restorative. A compound that could enhance slow-wave sleep while maintaining normal REM architecture would be fundamentally different from existing sleep drugs.
Figure 2: Normal sleep architecture showing the four main stages and their characteristic EEG waveforms. DSIP research focuses primarily on enhancing Stage N3 (delta/slow-wave sleep), the most restorative phase.
DSIP's Effects on Delta Sleep
The original rabbit experiments by Monnier and Schoenenberger demonstrated that intracerebroventricular infusion of DSIP produced two clear EEG effects: increased sleep spindle activity (associated with Stage N2) and enhanced delta wave activity (associated with Stage N3). Motor activity decreased simultaneously, confirming behavioral correlates of the EEG changes.
These findings have been replicated and extended across multiple animal models:
Rat studies: The phosphorylated analogue P-DSIP, administered via continuous 10-hour nocturnal intracerebroventricular infusion at 0.5 nmol, produced a 22% increase in slow-wave sleep and an 81% increase in paradoxical (REM) sleep in freely moving rats. This is a particularly significant finding because it demonstrates that DSIP analogues can enhance both deep and REM sleep simultaneously - an effect profile not seen with any conventional sleep medication.
Cat studies: Intraperitoneal administration of DSIP to cats produced dose-dependent increases in slow-wave sleep duration during the first 3-4 hours post-injection, followed by a normalization of sleep architecture. No rebound effects were observed.
Mouse studies: In PCPA-treated mice (a model of insomnia induced by serotonin depletion), DSIP fusion peptides restored sleep parameters toward baseline values, with particularly strong effects on slow-wave sleep recovery.
Human Sleep Data
Human studies of DSIP's sleep effects have produced more variable results, though the overall pattern supports some degree of sleep-promoting activity:
The Schneider-Helmert Insomnia Trial (1987): In a double-blind, placebo-controlled study of chronic insomniac patients, DSIP administration improved objective sleep measures. Sleep efficiency increased and sleep latency decreased compared to placebo. However, subjective sleep quality ratings did not significantly change, leading the authors to conclude that "short-term treatment of chronic insomnia with DSIP is not likely to be of major therapeutic benefit." This study has been widely cited as evidence against DSIP's sleep-promoting effects, though the treatment period was relatively short.
The Acute and Delayed Effects Study (1984): Luthringer and colleagues examined both immediate and delayed effects of DSIP on sleep in healthy volunteers. Intravenous DSIP infusion produced modest increases in total sleep time, but effects on specific EEG patterns were relatively minor. Interestingly, some delayed effects were observed in subsequent nights, suggesting a possible modulatory or "programming" action rather than direct sedation.
Healthy Volunteer Studies: Several studies in healthy subjects showed that intravenous DSIP infusion could shorten sleep onset latency and increase total sleep time, but the effects were variable between individuals. Some subjects showed pronounced increases in delta wave activity; others showed minimal change.
Clinical Observation
The variability in human sleep responses to DSIP may partly reflect individual differences in baseline sleep architecture, endogenous DSIP levels, and circadian timing of administration. Several researchers have noted that DSIP's sleep effects tend to be more pronounced in subjects with disrupted sleep than in good sleepers - suggesting it may function more as a sleep normalizer than a simple sedative. This pattern is consistent with a modulatory role rather than direct receptor agonism.
The Circadian Connection
DSIP's relationship with circadian biology adds another layer of complexity to its sleep effects. Several lines of evidence suggest DSIP interacts with the circadian system:
- Endogenous DSIP-like immunoreactivity shows circadian variation in plasma
- DSIP stimulates melatonin secretion from the pineal gland. A study using perifused rat pineal glands demonstrated that DSIP directly stimulated melatonin, 5-methoxytryptophol, and serotonin secretion
- The timing of DSIP administration influences its effects on sleep architecture, with evening administration producing more consistent results than daytime dosing
- DSIP's effects on cortisol secretion (discussed in detail in the Endocrine Effects section) mirror the circadian pattern of cortisol, with greater modulatory effects during the normal nadir periods
This circadian dimension connects DSIP research to the broader field of circadian peptide biology. Pinealon, another peptide studied for sleep effects, primarily targets circadian rhythm normalization through pineal gland support. Where Pinealon appears to act more on sleep timing, DSIP's primary effect seems to be on sleep depth - making the two conceptually complementary. Our Pinealon research report provides detailed analysis of the circadian peptide approach to sleep optimization.
Delta Waves and Brain Clearance
Recent neuroscience has revealed that slow-wave sleep serves a critical housekeeping function in the brain. The glymphatic system, a brain-wide waste clearance pathway, becomes dramatically more active during deep sleep. Cerebrospinal fluid flow increases, flushing out metabolic waste products including beta-amyloid, the protein associated with Alzheimer's disease.
This discovery has re-contextualized the potential significance of DSIP's delta sleep enhancement. If DSIP genuinely increases time spent in slow-wave sleep, it could indirectly support glymphatic clearance - a consideration with implications for long-term brain health. This is speculative, as no studies have directly measured glymphatic function during DSIP-enhanced sleep. But the connection between deep sleep and brain waste clearance makes DSIP's sleep architecture effects potentially more significant than was appreciated when the peptide was first discovered.
Similarly, slow-wave sleep is the primary period for growth hormone release from the pituitary. The research on DSIP's interaction with growth hormone secretion (covered in the Endocrine Effects section) may partly reflect this sleep-stage dependency rather than - or in addition to - direct hypothalamic-pituitary effects.
Figure 3: Schematic comparison of EEG patterns during baseline sleep versus DSIP-enhanced sleep. DSIP increases the amplitude and duration of delta waves during Stage N3, potentially extending the restorative deep sleep period.
What DSIP Does Not Do: Sedation vs. Sleep Promotion
An important distinction in DSIP research is between sedation and sleep promotion. Sedation involves pharmacological depression of neural activity, producing drowsiness and reduced responsiveness. Sleep promotion involves facilitation of natural sleep processes, allowing the brain to transition into and maintain normal sleep stages more effectively.
DSIP falls firmly in the sleep promotion category. In animal studies, DSIP does not produce the loss of righting reflex or motor impairment seen with sedative drugs. Animals treated with DSIP can still be aroused normally and show intact responses to stimuli. They simply fall asleep faster, spend more time in deep sleep, and show more organized sleep architecture.
This distinction matters practically because it suggests DSIP operates through a fundamentally different mechanism than drugs that act on GABA-A receptors (like benzodiazepines and Z-drugs). It also helps explain why DSIP's sleep effects are more variable than those of sedative drugs - it facilitates rather than forces sleep, making its effects more dependent on the underlying sleep drive and circadian timing.
Clinical Sleep Data
Overview of Human Clinical Trials
DSIP has been evaluated in several human clinical trials since the early 1980s, with studies conducted primarily in European research centers. While the total body of clinical evidence is modest compared to pharmaceutical sleep aids, it provides useful data on both the potential and limitations of DSIP as a sleep-modulating agent.
| Study | Year | Design | Population | Key Findings |
|---|---|---|---|---|
| Schneider-Helmert & Schoenenberger | 1983 | Open-label pilot | Insomniacs (n=16) | Improved sleep efficiency; variable delta sleep effects |
| Luthringer et al. | 1984 | Single-blind crossover | Healthy volunteers (n=10) | Acute and delayed modulation of sleep EEG parameters |
| Schneider-Helmert | 1987 | Double-blind RCT | Chronic insomniacs (n=14) | Improved sleep efficiency and latency; no subjective improvement |
| Graf & Kastin | 1984 | IV infusion study | Healthy volunteers (n=8) | Modest increases in total sleep time; minor EEG changes |
| Kafi et al. | 1986 | IV infusion study | Insomniacs (n=12) | Normalized sleep in responders; 60% response rate |
The Schneider-Helmert Double-Blind Trial
The most rigorous human sleep trial with DSIP was conducted by Schneider-Helmert and published in 1987. This double-blind, placebo-controlled study enrolled patients with chronic insomnia lasting at least one year who were not taking sleep medications.
Patients received DSIP (25 nmol/kg) or placebo intravenously on five consecutive evenings. Polysomnographic recordings were obtained throughout, measuring objective sleep parameters including sleep onset latency, total sleep time, sleep efficiency, and time spent in each sleep stage.
The results showed a clear split between objective and subjective outcomes:
- Sleep efficiency improved significantly with DSIP compared to placebo
- Sleep onset latency decreased, meaning subjects fell asleep faster
- Total sleep time showed a non-significant trend toward increase
- Subjective sleep quality ratings did not differ from placebo
- Wake time after sleep onset showed modest improvement
The disconnect between objective improvement and subjective perception has been interpreted in multiple ways. Some researchers argue it reflects a genuine limitation of DSIP - the sleep changes are too subtle for patients to notice. Others suggest that chronic insomniacs have entrenched negative sleep perceptions that require longer treatment to modify. The five-night treatment period may simply have been insufficient.
Sleep Stage Distribution: Baseline vs. DSIP Treatment
Data compiled from multiple studies showing approximate changes in sleep stage percentages with DSIP treatment. SWS = slow-wave sleep; REM = rapid eye movement sleep.
Response Variability and Responder Profiles
A consistent finding across DSIP sleep studies is high inter-individual variability. Some subjects show dramatic improvements in sleep parameters; others show minimal change. This pattern has led researchers to attempt to identify "responder" profiles.
Factors associated with better DSIP sleep responses include:
- Disrupted baseline sleep architecture (particularly reduced slow-wave sleep)
- Elevated pre-treatment cortisol levels
- Sleep disturbance associated with stress or anxiety rather than primary neurological insomnia
- Evening administration timed to the individual's habitual sleep onset
These observations support the concept of DSIP as a sleep normalizer rather than a universal sleep inducer. People whose sleep is already relatively normal show less benefit; those with dysregulated sleep patterns, particularly stress-related disruption, appear to respond more consistently.
Comparison with Other Sleep Interventions
| Parameter | DSIP | Benzodiazepines | Melatonin | Pinealon |
|---|---|---|---|---|
| Sleep onset latency | Moderate reduction | Strong reduction | Moderate reduction | Moderate reduction |
| Total sleep time | Modest increase | Significant increase | Variable | Variable |
| Slow-wave sleep | Increased | Suppressed | Minimal effect | Indirect support |
| REM sleep | Preserved/increased | Suppressed | Minimal effect | Preserved |
| Sleep quality (restorative) | Enhanced architecture | Impaired architecture | Circadian normalization | Circadian normalization |
| Dependence risk | Not observed | Significant | None established | None established |
| Morning hangover | Minimal/none | Common | Minimal | Minimal |
| Primary mechanism | Multiple (unclear) | GABA-A agonism | MT1/MT2 receptors | Pineal peptide support |
The comparison above highlights DSIP's distinctive profile. While it may not produce the dramatic reduction in sleep onset latency seen with benzodiazepines, its preservation and enhancement of slow-wave sleep represents a qualitatively different approach. Melatonin and Pinealon primarily act on circadian timing; DSIP appears to act more on sleep depth and architecture.
Research Context
Most DSIP sleep studies were conducted in the 1980s, before modern polysomnographic standards were fully established. The research used intravenous administration, which differs from the subcutaneous route commonly discussed in current research protocols. Newer delivery methods, including intranasal DSIP formulations, are being explored but have not been evaluated in comparable clinical trials. These methodological differences should be considered when interpreting the historical data.
The Delayed Effect Phenomenon
One of the more intriguing observations from human DSIP studies is the "delayed effect" - improvements in sleep that appear not during the treatment period but in the nights following treatment cessation. Luthringer's group observed this phenomenon in healthy volunteers, noting that some EEG improvements were more pronounced on recovery nights than during active DSIP administration.
This delayed effect has been interpreted as evidence of DSIP's "programming" action on sleep regulatory systems. Rather than directly inducing sleep during the period of pharmacological presence, DSIP may reset or recalibrate underlying sleep homeostatic mechanisms that continue to function after the peptide has been cleared.
If confirmed in larger studies, this delayed effect would have practical implications. It would suggest that intermittent DSIP dosing (e.g., several consecutive nights followed by off-periods) might be more effective than continuous administration - an approach that could also reduce any potential for receptor adaptation or tachyphylaxis.
Mechanism of Action
The Receptor Problem
For most bioactive peptides, the story of how they work begins with their receptor. Insulin binds insulin receptors. Endorphins bind opioid receptors. Growth hormone releasing hormone binds GHRH receptors. The receptor provides the molecular lock into which the peptide key fits, activating specific intracellular signaling cascades.
DSIP breaks this pattern. Despite more than 50 years of research, no dedicated DSIP receptor has been identified. This is not for lack of trying. Multiple research groups have attempted to isolate and characterize a specific receptor, but the results have been inconclusive or contradictory.
What has emerged instead is a picture of DSIP interacting with multiple receptor systems - none of which appears to be its exclusive target. This promiscuity may actually be the key to understanding DSIP's diverse biological effects.
Interaction with GABAergic Systems
Gamma-aminobutyric acid (GABA) is the brain's primary inhibitory neurotransmitter and plays a central role in sleep regulation. GABA-A receptor activation promotes sleep onset and is the mechanism through which benzodiazepines and Z-drugs work.
Research suggests DSIP may interact with GABA-A receptors, though the nature of this interaction is unclear. Some binding studies have detected DSIP association with GABA-A receptor complexes, but it has not been demonstrated that DSIP acts as a direct agonist at these receptors. The interaction may be allosteric (modifying receptor sensitivity rather than directly activating it) or may occur through indirect effects on GABAergic neurotransmission.
DSIP's effects on glycine receptors, another inhibitory neurotransmitter system, have also been proposed but not confirmed.
Interaction with Opioid Systems
The evidence for DSIP's interaction with endogenous opioid systems is stronger and more clinically relevant. Several lines of evidence support this connection:
- DSIP's antinociceptive (pain-reducing) effects in mice are blocked by naloxone, an opioid receptor antagonist, confirming opioid receptor involvement
- DSIP's analgesic effects are absent in morphine-tolerant animals, suggesting it acts through the same receptor systems that develop morphine tolerance
- The efficacy of DSIP in treating opioid withdrawal symptoms implies an interaction with opioid receptor systems
- Experimental data suggest a "modulation or programming interaction" between DSIP and endogenous opioid peptide systems
The working hypothesis is that DSIP does not directly bind opioid receptors with high affinity but modulates the activity of endogenous opioid peptide systems - essentially fine-tuning the body's natural pain and reward pathways rather than directly activating them. This would explain why DSIP produces analgesic effects without the euphoria, respiratory depression, or addictive properties associated with direct opioid agonists.
Figure 4: Proposed signaling pathways through which DSIP exerts its effects. Unlike most neuropeptides that act through a single receptor, DSIP appears to modulate multiple systems simultaneously.
Interaction with Glutamatergic Systems
NMDA (N-methyl-D-aspartate) receptors are the brain's primary excitatory glutamate receptors and play important roles in synaptic plasticity, learning, and arousal. Some evidence suggests DSIP may interact with NMDA receptor complexes, potentially contributing to its sleep-promoting effects by modulating excitatory neurotransmission.
The balance between GABAergic (inhibitory) and glutamatergic (excitatory) neurotransmission is fundamental to the sleep-wake transition. A compound that simultaneously modulates both systems - increasing inhibitory tone while decreasing excitatory drive - could promote sleep more effectively than a pure GABA agonist. DSIP's apparent ability to influence both sides of this balance may explain some of its sleep-promoting effects.
The MAPK Cascade Connection
More recent molecular research has uncovered a connection between DSIP and the mitogen-activated protein kinase (MAPK) signaling cascade. DSIP has been found to interact with components of this pathway, which regulates cell proliferation, differentiation, survival, and apoptosis.
Particularly interesting is DSIP's homology with glucocorticoid-induced leucine zipper (GILZ), a protein that functions as a negative regulator of inflammation and apoptosis. GILZ mediates some of the anti-inflammatory and cell-protective effects of glucocorticoids, and DSIP's structural similarity to portions of GILZ may explain some of its stress-protective properties.
This MAPK connection also provides a potential mechanism for DSIP's antioxidant and neuroprotective effects. MAPK signaling is involved in cellular responses to oxidative stress, and modulation of this pathway could underlie DSIP's observed ability to reduce free radical production and support mitochondrial function under stress conditions.
HPA Axis Modulation
DSIP's interaction with the hypothalamic-pituitary-adrenal (HPA) axis has been documented in multiple studies and provides one of the more coherent mechanistic narratives for its effects. The HPA axis is the body's central stress response system, controlling the production of cortisol and other stress hormones.
In animal models, DSIP administration produces:
- Reduction in ACTH (adrenocorticotropic hormone) immunoreactivity in plasma, lasting at least 3 hours post-injection
- Decreased corticotropin-releasing factor (CRF) activity in the hypothalamus
- Modified cortisol secretion patterns, particularly during stress
In human studies, the picture is more nuanced. Some studies have found that DSIP modifies ACTH and cortisol responses during specific time windows, while others have reported no significant changes in basal ACTH or cortisol levels. The discrepancy may relate to dosing, timing, and the stress state of the subjects.
The practical relevance of HPA axis modulation to sleep is straightforward: elevated cortisol impairs sleep onset and reduces slow-wave sleep duration. If DSIP attenuates HPA axis hyperactivity, particularly during the evening and nighttime cortisol nadir, it could facilitate both sleep onset and deeper sleep indirectly through stress hormone normalization.
This stress-modulating dimension connects DSIP to other peptides with anxiolytic properties. Selank, for example, acts primarily through modulation of anxiety pathways, while DSIP's stress effects appear to be mediated more through endocrine pathways. Our Selank research report covers the anxiolytic peptide pathway in detail.
Melatonin Stimulation
DSIP has been shown to directly stimulate melatonin secretion from the pineal gland. Using a perifused rat pineal gland preparation, researchers demonstrated that DSIP increased the production of melatonin, 5-methoxytryptophol, and serotonin - all key indoleamines involved in sleep-wake regulation.
This effect provides another mechanism through which DSIP may influence sleep. Melatonin is the primary hormonal signal of darkness and sleep timing, acting on MT1 and MT2 receptors in the suprachiasmatic nucleus. By stimulating melatonin production, DSIP may reinforce natural circadian sleep signals, complementing its direct effects on sleep architecture.
The connection between DSIP and pineal function also raises interesting questions about its relationship to aging-related sleep decline. Melatonin production decreases progressively with age, contributing to the reduced slow-wave sleep and increased sleep fragmentation seen in older adults. A compound that both enhances delta sleep directly and supports melatonin production could theoretically address multiple aspects of age-related sleep deterioration.
The Multi-Target Hypothesis
The absence of a single, dedicated receptor may actually be a feature rather than a bug. DSIP's interaction with multiple signaling systems - GABAergic, opioidergic, glutamatergic, HPA axis, and pineal pathways - allows it to modulate sleep, stress, and pain simultaneously through gentle, coordinated effects rather than powerful single-pathway activation. This "systems modulatory" profile could explain both DSIP's relatively subtle effects (no single pathway is strongly activated) and its broad applicability (multiple related systems are simultaneously adjusted).
Neuroprotective Mechanisms
Beyond sleep and stress, DSIP has demonstrated neuroprotective properties that appear to be independent of its sleep effects:
- Antioxidant activity: DSIP inhibits overproduction of free radicals in the central nervous system and stimulates endogenous antioxidant enzymes, including superoxide dismutase (SOD)
- Anti-lipid peroxidation: DSIP reduces lipid peroxidation in neural tissue, a key marker of oxidative damage
- Mitochondrial support: Under hypoxic conditions, DSIP enhances mitochondrial function and reduces free radical production
- Stroke recovery: In a rat model of focal stroke, an 8-day intranasal DSIP treatment protocol (one preventive dose plus 7 days post-reperfusion) accelerated recovery of motor functions, demonstrating functional neuroprotection
These neuroprotective effects may be partly mediated through the MAPK/GILZ pathway discussed above, and they add another dimension to DSIP's potential relevance beyond sleep. Other peptides with documented neuroprotective profiles include Semax, which acts through BDNF-mediated pathways, and NAD+, which supports cellular energy metabolism and DNA repair.
Pain & Stress Research
DSIP and Pain Modulation
DSIP's role in pain modulation has been investigated since the early 1980s, with research revealing a complex interplay between the peptide and endogenous pain-regulatory systems. The evidence suggests DSIP can reduce pain perception through mechanisms that involve, but are not limited to, the opioid system.
Preclinical Pain Studies
The foundational work on DSIP and pain was conducted using standard animal nociceptive assays. Key findings include:
- Hot-plate test: DSIP administration increased pain tolerance thresholds in mice, with effects comparable to moderate doses of morphine but without the characteristic motor impairment
- Naloxone blockade: The antinociceptive effects of DSIP were abolished by pre-treatment with naloxone, the classic opioid receptor antagonist. This confirmed that DSIP's pain-reducing effects are mediated, at least in part, through the opioid system
- Morphine cross-tolerance: DSIP's analgesic effects were absent in mice made tolerant to morphine. This finding is particularly important because it indicates DSIP acts through the same receptor populations as morphine, not through an independent pathway
- Supraspinal site of action: The evidence points to DSIP acting at supraspinal (brain) rather than spinal levels, possibly influencing descending pain modulation pathways from the periaqueductal gray and other brainstem structures
These findings establish that DSIP interacts with the endogenous opioid system, but with important qualitative differences from exogenous opioids. DSIP appears to modulate or "tune" opioidergic signaling rather than directly activating opioid receptors with high affinity. This modulatory profile would explain why DSIP can reduce pain without producing the euphoria, respiratory depression, constipation, and dependence associated with classical opioids.
Clinical Pain Study
A clinical pilot study published in European Neurology examined DSIP's therapeutic effects in patients with chronic, pronounced pain episodes. While the study was small and open-label, it provided the first human data on DSIP's analgesic potential:
- Patients with chronic pain conditions received DSIP intravenously
- Pain reduction was observed in a majority of treated patients
- The onset of analgesic effects was relatively rapid
- Side effects were minimal, with only occasional headaches reported
- No signs of tolerance development were observed during the treatment period
The study's authors, Dick and colleagues, described the results as promising enough to warrant larger controlled trials, though these subsequent studies were never conducted to the same standard.
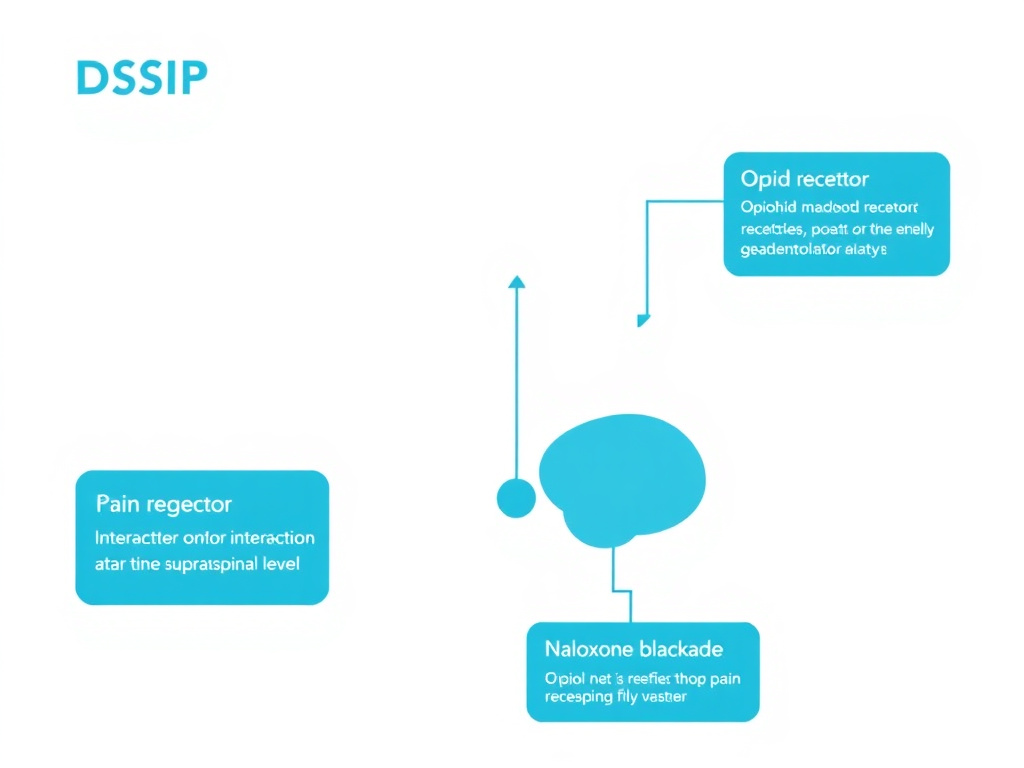
Figure 5: DSIP's pain modulation pathways. The peptide acts at supraspinal levels, with effects blockable by the opioid antagonist naloxone, indicating opioid system involvement.
The Pain-Sleep Connection
Pain and sleep have a well-established bidirectional relationship. Chronic pain disrupts sleep, and poor sleep amplifies pain perception. This creates a vicious cycle that is difficult to break with interventions targeting only one side of the equation.
DSIP's dual action on both sleep and pain is therefore potentially significant. By simultaneously enhancing restorative sleep and modulating pain perception, DSIP could theoretically address both sides of the pain-sleep cycle. This multi-target approach aligns with modern understanding of chronic pain as a complex condition requiring multi-modal intervention.
Stress Response Modulation
DSIP's effects on the stress response represent one of its most consistently documented properties across multiple experimental systems. The research demonstrates effects at multiple levels of the stress response cascade.
ACTH and Cortisol Effects
In rat models, DSIP injection produced a significant reduction in plasma ACTH-like immunoreactivity lasting at least 3 hours. This suppression of ACTH release from the pituitary translates into downstream reduction in adrenal cortisol production, effectively dampening the stress response at its hormonal source.
Human studies have produced more complex results. In some protocols, DSIP administration was associated with modified cortisol patterns, while in others, basal cortisol and ACTH levels were not significantly altered. This discrepancy likely reflects differences in:
- The stress state of subjects (DSIP may have greater effects under stressed conditions)
- Timing of measurements relative to circadian cortisol rhythms
- Dosing protocols and routes of administration
- Duration of treatment (acute vs. repeated dosing)
Stress-Protective Effects in Animal Models
Several animal studies have demonstrated DSIP's ability to protect against the physiological consequences of stress:
- DSIP pre-treatment reduced stress-induced alterations in catecholamine levels
- Animals treated with DSIP showed less stress-induced disruption of sleep architecture
- DSIP administration attenuated stress-related immune suppression in some models
- Behavioral tests showed reduced anxiety-like behavior following DSIP treatment
These stress-protective effects are conceptually linked to DSIP's interaction with the HPA axis and may also involve the GILZ-homologous properties described in the mechanism section. By attenuating the stress hormone cascade, DSIP may prevent the downstream physiological damage that chronic stress produces.
Clinical Implications
The intersection of DSIP's stress-modulatory and sleep-promoting effects has practical relevance for stress-related insomnia, which accounts for a significant proportion of sleep complaints. Elevated evening cortisol is a common finding in stressed insomniacs, and it directly impairs both sleep onset and slow-wave sleep depth. A compound that simultaneously normalizes cortisol patterns and enhances delta sleep could address stress-related insomnia at both the hormonal and neurophysiological levels. This dual mechanism is distinct from both anxiolytic drugs (which target subjective anxiety without necessarily correcting cortisol) and standard sleep medications (which force sleep without addressing hormonal dysregulation).
Withdrawal Syndrome Research
The most dramatic clinical findings with DSIP come from studies on alcohol and opioid withdrawal - areas where its combined effects on pain, stress, and sleep converge.
The Dick and Granstrom Withdrawal Studies
In the early 1980s, researchers P. Dick and K.E. Granstrom conducted what remain the most substantial clinical trials of DSIP for withdrawal syndromes. Their work is documented in two key publications:
Study 1 (1984): Sixty-seven patients with withdrawal symptoms (28 from alcohol, 39 from opiates) received intravenous DSIP (25 nmol/kg) as the sole treatment. After excluding 27% of patients who were lost to follow-up or unsuitable for evaluation, 49 patients were available for analysis. Results were striking:
- Of the 49 evaluable patients, 48 showed beneficial effects (98% response rate)
- All 22 evaluated alcoholics responded to treatment
- 26 of 27 evaluated opiate addicts showed improvement
- Onset of action was rapid, with symptom improvement beginning within hours
- Somatic withdrawal symptoms were effectively suppressed
Study 2 (1984): A larger series involving 107 patients (47 alcoholics, 60 opiate addicts) confirmed the initial findings. In this study, 87% of alcoholics showed clinical symptoms disappear or improve markedly. For opiate addicts, 97% showed symptom improvement, though the clinical course was more prolonged and required more DSIP injections than the alcoholic group.
| Parameter | Alcohol Withdrawal (n=47) | Opiate Withdrawal (n=60) |
|---|---|---|
| Response rate | 87% | 97% |
| Average time to improvement | Hours | Days |
| Number of DSIP injections needed | Fewer | More |
| Clinical course | Shorter | More prolonged |
| Anxiety improvement | Significant | Significant |
| Somatic symptom relief | Rapid | Gradual |
| Side effects (headache) | Few patients | Few patients |

Figure 6: Response rates from the Dick and Granstrom withdrawal studies. DSIP demonstrated high efficacy across both alcohol and opiate withdrawal syndromes.
Mechanistic Basis for Withdrawal Efficacy
The efficacy of DSIP in withdrawal syndromes is likely explained by its multi-system modulatory profile. Withdrawal from alcohol and opiates involves:
- Sleep disruption: Both alcohol and opiate withdrawal produce severe insomnia, particularly loss of slow-wave sleep. DSIP's delta sleep enhancement directly addresses this.
- Pain sensitization: Opiate withdrawal produces hyperalgesia (increased pain sensitivity) through opioid receptor desensitization. DSIP's modulation of opioidergic systems may help restore normal pain thresholds.
- HPA axis hyperactivation: Withdrawal is characterized by cortisol and CRF elevation. DSIP's HPA axis modulation could attenuate this stress response.
- Anxiety: Both withdrawal syndromes involve intense anxiety. DSIP's effects on GABAergic and stress pathways may contribute to anxiolysis.
- Autonomic instability: DSIP may help normalize sympathetic hyperactivity through its effects on catecholamine regulation.
In essence, DSIP's broad modulatory profile - which might be seen as a weakness in terms of mechanistic clarity - becomes a strength in the context of withdrawal syndromes, where multiple systems are simultaneously dysregulated.
Important Limitations
While the withdrawal data are impressive, they come with significant caveats. The studies were conducted in the 1980s with relatively small sample sizes. They lacked modern randomization standards. The loss-to-follow-up rates were high (27% in one study). And no large, placebo-controlled, randomized trials have been conducted since. The withdrawal data should therefore be viewed as strongly suggestive rather than definitive. DSIP is not an approved treatment for withdrawal syndromes, and these findings do not constitute a recommendation for clinical use.
For those interested in other peptides with stress-modulating properties, Selank has been studied specifically for its anxiolytic effects, while Semax has been investigated for neuroprotective and cognitive-enhancing properties under stress conditions. The Free Assessment at FormBlends can help determine which research compounds may be most relevant to individual circumstances.
Endocrine Effects
Growth Hormone Interactions
DSIP's effects on growth hormone (GH) secretion have been studied in both animal and human models, with results that illustrate the complexity of translating animal findings to human physiology.
Animal Studies
In rats, intracerebroventricular injection of DSIP produced dose-dependent elevations of plasma growth hormone. Key findings from the seminal study by Iyer and McCann include:
- Growth hormone elevation became significant within 30 minutes of DSIP injection and persisted for at least 120 minutes
- The response was dose-related, with a minimal effective dose of 0.1 mcg and a linear log-dose increase up to 10 mcg
- The GH-releasing effect was blocked by pre-treatment with pimozide, a dopamine receptor blocker, indicating the mechanism involves hypothalamic dopaminergic pathways
- The effect appears to be mediated hypothalamically, likely through modulation of growth hormone-releasing hormone (GHRH) or somatostatin secretion
These animal data suggested that DSIP could serve as a physiological growth hormone secretagogue, potentially linking its sleep-promoting and GH-releasing effects through the established relationship between slow-wave sleep and nocturnal GH pulses.
Human Studies
However, translation to humans has been disappointing. In a study of healthy women, DSIP infusion did not significantly alter serum growth hormone or prolactin levels under basal conditions. The circadian rhythms of GH and prolactin secretion were also unaffected by DSIP administration.
This discrepancy between animal and human GH data may reflect:
- Species-specific differences in hypothalamic DSIP sensitivity
- Route of administration effects (intracerebroventricular in rats vs. intravenous in humans)
- Dose-dependent effects that may require higher peripheral doses to achieve sufficient brain concentrations
- Gender-specific factors (the human study used exclusively female subjects)
For researchers interested in peptides with more consistent growth hormone-releasing properties, MK-677 (ibutamoren) and CJC-1295/Ipamorelin have more established effects on the GH axis in human studies.
ACTH and Cortisol
As discussed in the mechanism section, DSIP's effects on the ACTH-cortisol axis are among its most consistently documented endocrine properties, at least in animal models.
In rats, DSIP injection at doses of 5-10 mcg intracerebroventricularly produced significant ACTH reduction lasting at least 3 hours. This was accompanied by decreased adrenal cortisol output, consistent with the expected downstream effect of ACTH suppression.
Human data are less consistent. Several studies found that DSIP did not significantly alter baseline ACTH or cortisol patterns during standard measurement conditions. However, stress-induced cortisol responses may be more susceptible to DSIP modulation, and the timing of measurement relative to circadian cortisol peaks and troughs appears to influence the results.
Luteinizing Hormone
DSIP has been reported to influence luteinizing hormone (LH) secretion in some studies, though this area has received less research attention than the GH and ACTH interactions. The evidence suggests DSIP may modulate the pulsatile pattern of LH release, but the effects are variable and the clinical significance is unclear.
Thyroid Function
Limited data suggest DSIP may influence thyroid-stimulating hormone (TSH) secretion, but this area has not been systematically studied. Given DSIP's effects on hypothalamic-pituitary function more broadly, effects on the thyroid axis would not be surprising but remain speculative.
Integration: DSIP as an Endocrine Modulator
The collective endocrine data paint a picture of DSIP as a hypothalamic-level modulator that can influence multiple pituitary hormone axes. However, the effects are generally modulatory rather than stimulatory or suppressive - DSIP seems to adjust the sensitivity and set points of neuroendocrine regulatory loops rather than directly driving hormone secretion in one direction.
This profile is consistent with DSIP's broader characterization as a systems modulator. In the endocrine context, it may function as a stress-adaptive signal that helps reset hormonal axes toward their normal circadian patterns - reducing cortisol when it's elevated, supporting growth hormone during its natural nocturnal window, and adjusting reproductive hormones based on the organism's stress state.
| Endocrine Axis | Animal Data | Human Data | Proposed Mechanism |
|---|---|---|---|
| Growth Hormone | Dose-dependent increase (rats) | No significant effect (women) | Hypothalamic dopaminergic pathway |
| ACTH/Cortisol | Significant reduction (rats) | Variable; possible stress-state dependency | CRF modulation at hypothalamic level |
| LH | Modulatory effects reported | Limited data | GnRH pulse modulation (hypothetical) |
| Melatonin | Direct stimulation (rat pineal) | Not directly measured | Pinealocyte activation |
| TSH | Limited data | Limited data | Unknown |
The endocrine modulation profile of DSIP has interesting parallels with Epithalon, another peptide studied for its effects on pineal function and neuroendocrine regulation. While Epithalon primarily targets telomerase activation and pineal melatonin production, both peptides appear to influence hypothalamic-pituitary function in modulatory rather than directly stimulatory ways.
Dosing Protocols
Research Context Disclaimer
The dosing information presented here is derived from published research studies and clinical reports. DSIP is not FDA-approved for any medical indication. This information is provided for educational purposes and does not constitute medical advice or a recommendation for use. Any consideration of DSIP should involve consultation with a qualified healthcare provider.
Historical Clinical Dosing
In the published clinical trials, DSIP was administered intravenously at doses standardized by body weight:
- Withdrawal studies: 25 nmol/kg IV, which translates to approximately 21.25 mcg/kg (based on DSIP's molecular weight of ~850 Da). For a 75 kg adult, this equals approximately 1,594 mcg (1.6 mg)
- Sleep studies: Doses ranged from 10-30 nmol/kg IV, typically administered in evening hours before polysomnographic recording
- Pain studies: Similar dosing to sleep studies, 25 nmol/kg IV
These clinical doses were considerably higher than what is commonly discussed in current research literature, partly because intravenous administration bypasses first-pass metabolism but also results in rapid clearance.
Current Research Protocols
More recent research literature and clinical protocols generally describe subcutaneous (SC) administration at lower doses. Commonly referenced protocols include:
| Protocol | Dose | Route | Frequency | Timing |
|---|---|---|---|---|
| Low-dose initiation | 100 mcg | Subcutaneous | Nightly or 3-5x/week | 30-60 min before bed |
| Standard research dose | 200-300 mcg | Subcutaneous | Nightly or 3-5x/week | 30-60 min before bed |
| Higher research dose | 300-500 mcg | Subcutaneous | 3-5x/week | 30-60 min before bed |
| Intranasal (exploratory) | Variable | Intranasal | Variable | Before bed |
Key Dosing Considerations from Research Literature
Timing: Evening administration, 30-60 minutes before intended sleep onset, is the most commonly recommended timing across research protocols. This aligns with DSIP's proposed role as a sleep facilitator rather than a sedative - it works best when administered in coordination with the body's natural sleep drive.
Cycling: Some protocols recommend intermittent use rather than continuous daily administration. Common cycling schedules include 5 nights on/2 nights off, or 3 weeks on/1 week off. The rationale for cycling includes preventing potential receptor adaptation and the "delayed effect" phenomenon described in clinical studies, which suggests DSIP may produce residual benefits on off-days.
Starting dose: Most protocols recommend beginning at the lower end of the dosing range (100 mcg) and adjusting based on response. This approach allows assessment of individual sensitivity and minimizes the risk of excessive sleepiness or morning grogginess, which has been reported at higher doses.
Administration technique: Subcutaneous injection into the abdominal fat pad is the most commonly described technique. The injection volume is small (typically less than 0.5 mL when reconstituted to standard concentrations), and the injection itself is minimally painful due to DSIP's low volume and neutral pH.
Dosing Calculator
For those working with various peptide protocols, the FormBlends Dosing Calculator provides a convenient tool for calculating reconstitution volumes and individual doses based on body weight and desired concentration.
Reconstitution and Storage
DSIP is typically supplied as a lyophilized (freeze-dried) powder. Standard reconstitution practices from the research literature include:
- Reconstitute with bacteriostatic water to a convenient concentration (commonly 1-5 mg/mL)
- Store reconstituted solution refrigerated at 2-8 degrees C
- Use reconstituted solution within 2-4 weeks (due to DSIP's inherent molecular instability)
- Lyophilized powder should be stored frozen (-20 degrees C) for long-term stability
- Avoid repeated freeze-thaw cycles, which accelerate degradation
DSIP's short plasma half-life (7-8 minutes) means that subcutaneous administration, which provides slower absorption than intravenous dosing, may actually be preferable for sustained effect. The subcutaneous depot allows gradual release of DSIP into the bloodstream over a period of 30-60 minutes, potentially extending the effective exposure window beyond what would be achieved with a rapid IV bolus.
Combination Protocols in Research
Some research protocols describe DSIP in combination with other peptides or supplements for sleep optimization. Commonly referenced combinations include:
- DSIP + Melatonin: Targeting both sleep architecture (DSIP) and circadian timing (melatonin). Typical protocols use 100-300 mcg DSIP SC with 0.5-3 mg melatonin oral, both administered 30-60 minutes before bed
- DSIP + Pinealon: Combining delta sleep enhancement with circadian rhythm normalization through pineal peptide support
- DSIP + Magnesium Glycinate: Magnesium supports GABAergic function, potentially complementing DSIP's effects through a separate pathway
These combination protocols are based on theoretical rationale and anecdotal reports rather than controlled clinical trials, and their relative efficacy compared to DSIP alone has not been formally evaluated.
Safety Profile
Published Safety Data
DSIP's safety profile, based on published clinical and preclinical studies, is generally favorable but limited in scope. The peptide has been administered to hundreds of subjects in clinical studies without reports of serious adverse events, but long-term safety data are essentially absent.
Reported Side Effects from Clinical Studies
| Side Effect | Frequency | Severity | Context |
|---|---|---|---|
| Headache | Reported by a few patients | Mild | Withdrawal studies |
| Morning grogginess/lethargy | Occasional (dose-related) | Mild to moderate | Sleep studies; higher doses |
| Injection site irritation | Occasional | Mild | SC administration protocols |
| Nausea | Rare | Mild | Anecdotal reports |
| Dizziness | Rare | Mild | Anecdotal reports |
In the withdrawal studies by Dick and Granstrom, which represent the largest clinical experience with DSIP, tolerance to the treatment was described as "good," with only headaches in a few patients noted as an adverse effect. No patients withdrew from the studies due to side effects.
Absence of Notable Adverse Effects
Several concerning effects that might be expected from a sleep-promoting, endocrine-modulating peptide were absent from the clinical reports:
- No dependence or withdrawal: No signs of physical dependence or rebound insomnia were reported after DSIP discontinuation
- No respiratory depression: Despite interaction with opioid systems, DSIP did not produce the respiratory depression associated with opioid agonists
- No significant cognitive impairment: Unlike benzodiazepines, DSIP did not produce notable cognitive or psychomotor impairment
- No tolerance development: In the clinical studies, DSIP maintained its effects throughout the treatment periods without evidence of dose escalation
FDA Regulatory Concerns
The US Food and Drug Administration has not approved DSIP for any therapeutic indication. In addition to the standard requirement for FDA approval of new drugs, the FDA has raised specific concerns about compounded peptide products containing DSIP:
FDA Immunogenicity Warning
The FDA has noted that compounded drugs containing DSIP may pose a risk for immunogenicity - a situation in which the body perceives a substance as foreign and mounts an immune response against it. This risk is theoretically present with any exogenously administered peptide and is influenced by factors including peptide purity, formulation, route of administration, dose, and individual immune characteristics. While clinically significant immune reactions to DSIP have not been widely reported, the FDA's concern is legitimate given the limited safety surveillance for compounded peptide products.
Potential Safety Concerns
Several theoretical safety concerns deserve mention even though they have not been observed in published studies:
Endocrine disruption: Given DSIP's documented effects on ACTH, cortisol, growth hormone, and luteinizing hormone, chronic administration could theoretically alter endocrine homeostasis. The modulatory (rather than strongly suppressive or stimulatory) nature of DSIP's endocrine effects may mitigate this risk, but long-term monitoring data are unavailable.
Immunogenicity: As the FDA has highlighted, repeated administration of any exogenous peptide can potentially trigger immune responses, particularly if the peptide has minor structural differences from endogenous forms or contains impurities from the synthesis process.
Drug interactions: DSIP's interactions with GABAergic and opioidergic systems raise theoretical concerns about additive effects with sedative medications, opioid analgesics, benzodiazepines, and other central nervous system depressants. No formal drug interaction studies have been conducted.
Long-term effects: The longest clinical exposure periods in published studies are weeks to months. Effects of long-term (years) DSIP use are completely unknown.
Product quality: Because DSIP is not a regulated pharmaceutical product, quality control for commercially available DSIP is variable. Purity, potency, and sterility cannot be guaranteed for products from unregulated sources.
Contraindications and Precautions
Based on DSIP's pharmacological profile, the following contraindications and precautions have been suggested in the research literature:
- Contraindicated: Pregnancy and breastfeeding (no safety data available)
- Contraindicated: Known hypersensitivity to DSIP or any formulation components
- Use with caution: Concurrent use of opioid medications (potential additive effects)
- Use with caution: Concurrent use of sedative medications or alcohol
- Use with caution: Adrenal insufficiency or HPA axis disorders (potential cortisol effects)
- Use with caution: Active autoimmune conditions (uncertain immune effects)
- Monitor: Growth hormone-sensitive conditions
Figure 7: DSIP safety profile summary comparing reported adverse effects with those of conventional sleep medications. DSIP shows a more favorable side effect profile but lacks long-term safety data.
Safety Comparison with Conventional Sleep Medications
| Safety Parameter | DSIP | Benzodiazepines | Z-Drugs (Zolpidem) | Melatonin |
|---|---|---|---|---|
| Dependence potential | Not observed | High | Moderate | None established |
| Respiratory depression | Not observed | Yes (dose-dependent) | Rare | None |
| Cognitive impairment | Not observed | Yes | Yes (complex behaviors) | Minimal |
| Rebound insomnia | Not observed | Yes | Yes | None established |
| Morning hangover | Rare (dose-related) | Common | Common | Rare |
| Long-term safety data | Absent | Extensive | Moderate | Good |
| FDA approval | No | Yes | Yes | OTC supplement |
While DSIP's apparent safety profile compares favorably to prescription sleep medications, the critical limitation is the absence of long-term data. The benzodiazepine and Z-drug safety profiles are well characterized precisely because they have been used by millions of patients over decades. DSIP's experience base is orders of magnitude smaller, meaning rare adverse effects could exist that have not yet been detected.
DSIP vs. Pinealon vs. Melatonin: A Comparative Analysis
Different Approaches to the Same Problem
Sleep optimization can be approached from several different angles, and understanding where DSIP fits among sleep-supporting compounds requires comparison with other well-studied options. Three of the most commonly discussed are DSIP itself, Pinealon (a synthetic tripeptide derived from pineal gland peptides), and melatonin (the endogenous hormone of darkness).
Each targets sleep through fundamentally different mechanisms, which means they are not simply alternatives but rather complementary tools that address different aspects of sleep dysfunction.
Melatonin: The Circadian Signal
Melatonin is produced by the pineal gland in response to darkness and serves as the body's primary circadian timing signal. It acts on MT1 and MT2 receptors in the suprachiasmatic nucleus (the brain's master clock) and other brain regions to promote sleep onset and regulate the sleep-wake cycle.
Strengths:
- Well-characterized mechanism through MT1/MT2 receptors
- Extensive safety data from decades of widespread use
- Effective for circadian misalignment (jet lag, shift work)
- Available over-the-counter in many countries
- Low cost and wide availability
Limitations:
- Does not significantly increase slow-wave sleep depth
- Does not address stress-related sleep disruption through hormonal mechanisms
- Variable quality in commercial supplements
- Commonly used at supraphysiological doses, potentially disrupting natural production
- Primarily affects sleep timing, not sleep architecture
Pinealon: The Circadian Peptide
Pinealon is a synthetic tripeptide (Glu-Asp-Arg) derived from pineal gland peptide extracts. It is part of a class of bioregulatory peptides developed by Russian researchers, primarily Vladimir Khavinson, who proposed that short peptides can regulate gene expression in their target tissues.
Strengths:
- Supports endogenous melatonin production rather than replacing it
- May help normalize circadian rhythm disruptions
- Small molecular size allows oral and intranasal administration
- Favorable safety profile in available studies
- May support broader pineal gland health and function
Limitations:
- Limited published clinical trial data
- Primarily acts on circadian timing rather than sleep depth
- Research base is primarily from Russian-language publications
- Mechanism of gene regulation by short peptides is still being elucidated
DSIP: The Sleep Architecture Modulator
Strengths:
- Directly enhances slow-wave (delta) sleep, the most restorative phase
- Preserves or enhances REM sleep simultaneously
- Multi-target effects address sleep, stress, and pain together
- No dependence or rebound observed in clinical studies
- May stimulate endogenous melatonin production (dual mechanism)
- Documented efficacy in withdrawal syndromes
Limitations:
- No identified specific receptor
- Short plasma half-life requires careful timing
- Injectable administration (subcutaneous or intravenous)
- Mixed results in human sleep trials
- Limited long-term safety data
- Not FDA-approved
Head-to-Head Comparison
| Feature | DSIP | Pinealon | Melatonin |
|---|---|---|---|
| Primary target | Sleep architecture (delta waves) | Pineal function / circadian rhythm | MT1/MT2 receptors / circadian timing |
| Effect on slow-wave sleep | Direct enhancement | Indirect (via circadian normalization) | Minimal direct effect |
| Effect on REM sleep | Preserved or enhanced | Indirect normalization | Minimal direct effect |
| Stress/cortisol modulation | Yes (HPA axis effects) | Limited data | Modest; mainly circadian cortisol |
| Pain modulation | Yes (opioid system) | Not established | Not established |
| Route of administration | SC injection (primarily) | Oral, intranasal, SC | Oral (primarily) |
| Onset of effect | 30-60 minutes | Days to weeks for full effect | 30-60 minutes |
| Best suited for | Reduced deep sleep; stress-related insomnia; pain-related sleep disruption | Circadian misalignment; jet lag; age-related melatonin decline | Circadian misalignment; jet lag; shift work |
| Molecular size | 9 amino acids (849 Da) | 3 amino acids (~389 Da) | Indolamine (232 Da) |
Figure 8: Comparative mechanisms of three sleep-supporting compounds. DSIP targets sleep depth and architecture, while Pinealon and melatonin primarily address circadian timing. The approaches are complementary rather than redundant.
Combination Rationale
Because DSIP, Pinealon, and melatonin target different aspects of sleep regulation, they could theoretically be combined to address multiple dimensions of sleep dysfunction simultaneously:
- DSIP addresses sleep depth and architecture, promoting more time in restorative slow-wave sleep
- Pinealon supports the circadian framework by promoting healthy pineal gland function and endogenous melatonin production
- Melatonin (at low, physiological doses) provides the immediate circadian signal for sleep onset
This layered approach - circadian timing (melatonin), circadian maintenance (Pinealon), and sleep depth (DSIP) - represents a conceptually coherent framework for comprehensive sleep optimization. However, no clinical trials have evaluated this specific combination, and its relative benefit over individual compounds remains theoretical.
For a broader discussion of peptide-based approaches to sleep and recovery, visit the Biohacking Hub at FormBlends.
DSIP and Athletic Recovery
The Sleep-Recovery Connection in Athletic Performance
Among the growing communities interested in DSIP research, athletes and fitness enthusiasts represent a significant segment. This interest is driven by the well-established relationship between sleep quality - specifically slow-wave sleep - and physical recovery, muscle growth, and athletic performance.
The scientific basis for this interest is solid. Research has consistently demonstrated that slow-wave sleep is the primary period for several recovery-critical processes:
Growth hormone pulse: As discussed earlier in this report, the largest daily pulse of growth hormone occurs during the first bout of slow-wave sleep. This pulse accounts for approximately 70% of total daily GH secretion in young adults. GH during this window drives protein synthesis, tissue repair, fat metabolism, and bone mineralization. Athletes who fail to achieve adequate deep sleep lose a significant portion of their recovery capacity, regardless of how well they eat or how intelligently they train.
Testosterone production: While testosterone secretion is less tightly coupled to specific sleep stages than GH, sleep quality significantly influences testosterone levels. Studies show that restricting sleep to 5 hours per night for one week reduces testosterone levels by 10-15% in young healthy men. The relationship appears to be mediated partly through slow-wave sleep duration, as testosterone production during sleep correlates with time spent in deep sleep stages.
Muscle protein synthesis: The nocturnal protein synthesis rate is influenced by both growth hormone availability and overall sleep quality. Deep sleep provides the hormonal and metabolic environment that maximizes the body's response to training stimulus from the previous day. Poor sleep quality can reduce muscle protein synthesis rates by 15-25%, even when protein intake is adequate.
Inflammatory resolution: Exercise induces controlled inflammation that, when properly resolved, drives adaptation and strengthening. Sleep, particularly deep sleep, facilitates the resolution of exercise-induced inflammation through cytokine regulation and immune cell activity. Chronic sleep restriction can convert this healthy inflammatory cycle into persistent, low-grade inflammation that impairs recovery and increases injury risk.
Neural recovery: Motor learning and skill acquisition consolidate during sleep, with both slow-wave sleep and REM sleep contributing to different aspects of motor memory. Athletes learning new techniques or refining existing skills depend on adequate sleep architecture for these gains to solidify. Disrupted sleep architecture can significantly impair the translation of practice into lasting motor skill improvement.
DSIP's Theoretical Appeal for Athletic Populations
DSIP's research profile suggests several properties that align with athletic recovery needs:
- Delta sleep enhancement directly supports the GH pulse and tissue repair window
- Cortisol modulation may help manage the HPA axis overactivation that frequently accompanies intense training, particularly in overtrained athletes
- Pain modulation through opioidergic pathways could help manage training-related soreness without the gastrointestinal and performance-impairing effects of NSAIDs
- Antioxidant properties may complement the body's endogenous antioxidant defense against exercise-induced oxidative stress
- No morning hangover effect (at appropriate doses) preserves next-day training readiness, unlike many sleep medications
- No dependence development allows long-term use during training cycles without withdrawal concerns
However, it's essential to note that no controlled studies have evaluated DSIP specifically in athletic populations. The theoretical case is based on extrapolation from DSIP's known effects to the established sleep-recovery-performance relationship. Whether DSIP's relatively modest sleep effects (as demonstrated in clinical trials) translate into meaningful performance or recovery benefits is unknown.
Comparison with Other Recovery-Oriented Peptides
Athletes interested in peptide-based recovery optimization have several options, each targeting different aspects of the recovery process:
| Peptide | Primary Mechanism | Recovery Application | Sleep Effect |
|---|---|---|---|
| DSIP | Delta sleep enhancement | Sleep quality, GH pulse support | Direct: enhances deep sleep |
| MK-677 | GH secretagogue (ghrelin mimetic) | Direct GH/IGF-1 elevation | Indirect: may improve sleep quality via GH |
| CJC-1295/Ipamorelin | GHRH analogue + GH secretagogue | Pulsatile GH release | Possible enhancement of SWS |
| BPC-157 | Growth factor modulation | Tissue repair, tendon/ligament | No direct sleep effect |
| TB-500 | Thymosin beta-4 analogue | Tissue repair, inflammation | No direct sleep effect |
The key distinction with DSIP is that it targets recovery through sleep optimization rather than through direct hormonal or tissue-level effects. This makes it complementary to rather than competitive with other recovery peptides. An athlete using CJC-1295/Ipamorelin for GH axis support could theoretically benefit from DSIP's sleep architecture enhancement as an additional recovery layer targeting a different physiological mechanism.
Overtraining Syndrome and DSIP
Overtraining syndrome (OTS) is a condition characterized by chronic fatigue, performance decline, mood disturbance, and persistent sleep disruption despite adequate rest. It is essentially a state of HPA axis dysregulation caused by excessive training load without adequate recovery.
The hallmarks of OTS overlap considerably with the physiological domains where DSIP has demonstrated effects:
- HPA axis dysregulation: OTS is characterized by altered cortisol patterns, including elevated resting cortisol and/or blunted cortisol response to exercise. DSIP's HPA axis modulatory effects could theoretically help normalize these patterns.
- Sleep disruption: Overtrained athletes frequently report poor sleep quality with reduced deep sleep, despite feeling physically exhausted. DSIP's delta sleep enhancement directly addresses this component.
- Altered pain perception: OTS often includes heightened pain sensitivity and persistent muscle soreness. DSIP's opioidergic modulation could help recalibrate pain thresholds.
- Immune dysfunction: OTS is associated with increased upper respiratory infections and altered immune markers. DSIP's immunomodulatory properties could potentially support immune recovery.
While the theoretical alignment between DSIP's pharmacological profile and OTS pathophysiology is striking, no clinical evidence directly supports DSIP use for overtraining. The concept remains speculative and would require formal evaluation in controlled studies.
Anti-Doping Considerations
Athletes subject to anti-doping regulations should be aware that DSIP's status under the World Anti-Doping Agency (WADA) Prohibited List is not straightforward. While DSIP is not explicitly named on the 2024-2025 WADA list, peptide hormones and growth factors are broadly prohibited. DSIP's effects on growth hormone secretion (documented in animal models) and its general classification as a bioactive peptide could potentially bring it under the purview of prohibited substance categories. Athletes in tested sports should consult with their sport's anti-doping authority before considering DSIP or any other peptide.
DSIP Pharmacokinetics: An In-Depth Analysis
Absorption
DSIP's pharmacokinetic profile presents unique challenges compared to most therapeutic peptides. Understanding these properties is essential for optimizing research protocols and interpreting clinical results.
Intravenous administration: The original clinical trials used IV bolus injection or short infusion, producing immediate peak plasma concentrations followed by rapid clearance. The elimination half-life by this route is 7-8 minutes, making IV DSIP one of the shortest-lived therapeutic peptides studied.
Subcutaneous administration: SC injection creates a depot of peptide in subcutaneous tissue that absorbs gradually into the bloodstream. While specific SC pharmacokinetic data for DSIP are limited, the general principle with SC peptide administration is a lower peak concentration (Cmax) but prolonged exposure duration compared to IV. For a peptide with a 7-8 minute IV half-life, the SC absorption phase effectively extends the exposure window to 30-60 minutes, as the rate of absorption from the subcutaneous depot becomes the rate-limiting step rather than the rate of enzymatic degradation in plasma.
Intranasal administration: The nasal mucosa provides a route for direct nose-to-brain delivery, bypassing both the blood-brain barrier and hepatic first-pass metabolism. DSIP administered intranasally in the rat stroke model demonstrated biological activity, confirming functional CNS access via this route. Intranasal delivery may be particularly advantageous for DSIP because it avoids the plasma aminopeptidases that rapidly degrade the peptide in the bloodstream.
Distribution
DSIP distributes to multiple tissues, with particularly relevant distribution to the central nervous system. Key distribution characteristics include:
Blood-brain barrier penetration: Studies using radioiodinated DSIP demonstrated measurable penetration into brain tissue following peripheral (IV) administration. The transport mechanism is not fully characterized but may involve a saturable, carrier-mediated process rather than simple diffusion. Some evidence suggests DSIP may utilize the same transport systems as certain other small peptides, though this remains unconfirmed.
Carrier protein binding: One hypothesis for DSIP's paradoxically long duration of action (hours of effect despite minutes of plasma half-life) is that it binds to carrier proteins in the blood. These carrier complexes would protect DSIP from aminopeptidase degradation while serving as a slow-release reservoir. The identity of these putative carrier proteins has not been established, but albumin and certain immunoglobulins have been suggested as candidates.
Tissue accumulation: DSIP-like immunoreactivity has been detected in brain tissue at concentrations higher than would be expected from simple equilibration with plasma levels. This suggests either active accumulation by brain tissue, local production of DSIP or DSIP-like peptides, or binding to brain-specific proteins that retain the peptide at its site of action.
Metabolism and Elimination
DSIP is degraded primarily by aminopeptidases in the blood and tissues. The primary cleavage occurs at the N-terminal tryptophan residue, initiating a sequential degradation process:
- Initial N-terminal cleavage by aminopeptidases removes Trp, producing the octapeptide AGGDASGE
- Subsequent cleavages progressively shorten the peptide chain
- The degradation products do not appear to have significant biological activity, though this has not been exhaustively studied
- No active metabolites have been identified
The rapid degradation explains why DSIP analogues designed for clinical use have focused on N-terminal modifications (D-amino acid substitutions, N-terminal blocking groups) that resist aminopeptidase attack. The phosphorylated analogue P-DSIP, with its modification at position 7, may also have altered susceptibility to degradation, though this has not been specifically measured.
Pharmacokinetic Implications for Dosing
DSIP's unusual pharmacokinetic profile has several practical implications:
Timing sensitivity: The short plasma half-life means that the timing of administration relative to sleep onset is critical. Dosing too early (more than 60-90 minutes before bed) may result in insufficient circulating DSIP at the time of sleep onset. Dosing too close to bedtime may not allow sufficient time for absorption and CNS distribution from a subcutaneous depot.
Route-of-administration effects: The choice between IV, SC, and intranasal administration may significantly influence efficacy, as each route produces different pharmacokinetic profiles. The historical clinical trial data (all IV) may not directly predict outcomes with SC administration (the most common current research route).
Individual variability: Differences in subcutaneous tissue thickness, blood flow to injection sites, plasma aminopeptidase activity, blood-brain barrier permeability, and carrier protein levels could all contribute to the substantial inter-individual variability observed in DSIP clinical trials. Standardizing these factors in future trials would help reduce variability and improve statistical power.
Sustained-release potential: The disconnect between DSIP's short plasma half-life and its prolonged biological effects suggests that sustained-release formulations might not be necessary - or alternatively, that they might be transformative. If DSIP's effects are indeed mediated by brief exposure that triggers longer-lasting downstream signaling, sustained release would be unnecessary. If, however, continuous exposure is needed for maximal effect, sustained-release formulations could dramatically improve efficacy.
Figure 9: Schematic pharmacokinetic profiles for DSIP administered by different routes. IV produces the highest peak but shortest duration. SC provides a lower, more sustained profile. Intranasal bypasses systemic circulation, providing direct CNS access.
DSIP and Mental Health: Sleep, Stress, and Mood
The Sleep-Mental Health Bidirectional Relationship
Sleep disruption and mental health conditions exist in a bidirectional relationship that has been extensively documented in psychiatric and neuroscience literature. Depression, anxiety, PTSD, and other conditions consistently impair sleep, while poor sleep increases vulnerability to and severity of mental health conditions. This creates self-reinforcing cycles that are difficult to break with interventions targeting only one side of the equation.
DSIP's multi-system modulatory profile positions it at the intersection of sleep and stress regulation, making it potentially relevant to the sleep-mental health nexus. Let's examine the evidence for each relevant domain.
Depression and Sleep Architecture
Major depressive disorder (MDD) is associated with characteristic sleep architecture changes, including:
- Reduced slow-wave sleep duration and intensity
- Shortened REM latency (earlier onset of REM sleep)
- Increased REM density (more eye movements per REM period)
- Decreased sleep efficiency (more time awake during the sleep period)
- Early morning awakening
The reduction in slow-wave sleep in depression is particularly relevant to DSIP research. Depressed patients often show significant deficits in delta wave generation, which contributes to the impaired physical restoration, cognitive dysfunction, and hormonal dysregulation seen in the condition. If DSIP can genuinely enhance delta sleep in this population, it could address a core physiological deficit of depression rather than simply treating symptoms.
Additionally, the HPA axis hyperactivation that characterizes depression (elevated CRF, ACTH, and cortisol) maps directly onto DSIP's documented ability to attenuate HPA axis activity. The combined effects of delta sleep restoration and cortisol normalization could theoretically produce anti-depressant effects through physiological normalization rather than neurotransmitter manipulation.
However, no clinical trials have evaluated DSIP in depressed populations. This application remains entirely theoretical and would require careful investigation, particularly given the complex interactions between sleep, mood, and endocrine function in depression.
Anxiety and the GABAergic Connection
DSIP's proposed interaction with GABAergic systems connects it to anxiety research, since GABA-A receptor dysfunction is implicated in generalized anxiety disorder, social anxiety, and panic disorder. The fact that DSIP may modulate rather than directly agonize GABA-A receptors is relevant because it suggests a different risk-benefit profile from benzodiazepines: potentially useful for anxiety reduction without the dependence, tolerance, and withdrawal risks of direct GABA-A agonists.
The withdrawal studies provide indirect evidence of anxiolytic effects. Both alcohol and opioid withdrawal involve severe anxiety, and DSIP effectively reduced withdrawal symptoms in the majority of treated patients. While the studies did not separately measure anxiety as an outcome, the overall symptom improvement necessarily included anxiety reduction.
For those interested in peptides with more directly established anxiolytic effects, Selank has been more extensively studied for anxiety specifically. The Selank anxiolytic peptide report provides detailed analysis of this compound's mechanism and clinical data.
PTSD and Trauma-Related Sleep Disruption
Post-traumatic stress disorder (PTSD) produces some of the most severe sleep disruption of any psychiatric condition. Nightmares, hyperarousal, difficulty falling and staying asleep, and reduced slow-wave sleep are cardinal features. The hyperactivated HPA axis and exaggerated sympathetic nervous system response in PTSD create a physiological state that is fundamentally antagonistic to sleep.
DSIP's profile - HPA axis modulation, slow-wave sleep enhancement, and opioidergic system interaction - addresses several of the physiological mechanisms that drive PTSD-related sleep disruption. The opioidergic connection is particularly interesting because endogenous opioid system dysfunction has been identified as a feature of PTSD, potentially contributing to both the emotional numbing and hyperarousal symptoms of the condition.
Again, no clinical data exist for DSIP in PTSD populations. But the theoretical alignment between DSIP's pharmacological actions and PTSD pathophysiology is strong enough to warrant investigation.
Substance Use Disorders and Recovery Sleep
The withdrawal study data provide the most direct clinical evidence for DSIP's relevance to mental health. Beyond the acute withdrawal period, individuals recovering from alcohol and opioid use disorders frequently experience persistent sleep disruption that can last months to years after achieving sobriety. This chronic sleep disturbance is a significant predictor of relapse, as poor sleep quality undermines mood stability, stress resilience, and cognitive function during recovery.
DSIP's demonstrated efficacy in acute withdrawal, combined with its favorable side effect profile (no dependence, no respiratory depression, no cognitive impairment), makes it theoretically attractive for the longer-term sleep support that recovery populations need. The fact that it interacts with opioidergic systems without producing euphoria or dependence is particularly relevant for populations with a history of substance use disorders, where medications with abuse potential pose additional risks.
The Integrated Approach
DSIP's potential relevance to mental health lies not in any single dramatic effect but in its ability to simultaneously address multiple physiological disruptions that co-occur in psychiatric conditions: impaired slow-wave sleep, HPA axis dysregulation, altered pain processing, and disrupted opioidergic signaling. This multi-system approach is conceptually aligned with modern psychiatric understanding of mental health conditions as network disorders rather than single-neurotransmitter problems. Whether DSIP's modulatory effects are strong enough to produce clinically meaningful psychiatric benefits remains to be determined through properly designed clinical trials.
Quality and Sourcing Considerations
Peptide Purity and Identity
Because DSIP is not a regulated pharmaceutical product, the quality of commercially available DSIP varies significantly between sources. This variability can affect both efficacy and safety, making quality assessment a practical concern for researchers and clinicians.
Key quality parameters to evaluate include:
Peptide purity: High-performance liquid chromatography (HPLC) is the standard method for assessing peptide purity. Research-grade DSIP should demonstrate purity of 98% or higher by HPLC analysis. Lower purity products may contain synthesis byproducts, truncated sequences, or degradation products that could reduce efficacy or pose safety risks. A certificate of analysis (COA) from an independent third-party laboratory is the most reliable way to verify purity claims.
Sequence verification: Mass spectrometry (MS) confirms that the peptide has the correct molecular weight, corresponding to the WAGGDASGE sequence (approximately 849-850 Da). This distinguishes authentic DSIP from incorrect sequences that might have similar HPLC profiles.
Endotoxin testing: Bacterial endotoxins (lipopolysaccharides) are common contaminants in peptide synthesis and can cause fever, inflammation, and potentially serious immune reactions when injected. The Limulus amebocyte lysate (LAL) test is the standard assay for endotoxin contamination. Injectable peptides should demonstrate endotoxin levels below 5 EU/kg body weight per dose.
Sterility: Products intended for injection must be sterile. Lyophilized (freeze-dried) peptides are typically sterile if produced under appropriate conditions, but reconstitution introduces contamination risk. Use of bacteriostatic water (containing 0.9% benzyl alcohol preservative) for reconstitution helps maintain sterility during the use period.
Heavy metal testing: Some peptide synthesis processes can introduce trace heavy metals. Testing for lead, mercury, arsenic, and cadmium is included in comprehensive quality assessments.
Storage and Stability
DSIP's inherent molecular instability makes proper storage particularly important:
- Lyophilized powder: Store at -20 degrees C (freezer) for long-term stability. At this temperature, lyophilized DSIP can maintain potency for 2+ years. Room temperature storage accelerates degradation significantly.
- Reconstituted solution: Store at 2-8 degrees C (refrigerator). Use within 2-4 weeks. The 15-minute in vitro half-life measured for DSIP reflects degradation at 37 degrees C in the presence of enzymes; reconstituted solutions at 4 degrees C degrade much more slowly but still have limited stability.
- Light sensitivity: The tryptophan residue at position 1 is sensitive to photo-oxidation. Store DSIP away from direct light, ideally in amber vials.
- Freeze-thaw cycles: Avoid repeated freezing and thawing of reconstituted DSIP solutions. Each cycle can cause peptide aggregation and loss of potency. If partial use is anticipated, divide reconstituted solution into single-use aliquots before the first freeze.
FormBlends provides DSIP products that undergo third-party testing for purity, identity, and sterility, addressing many of the quality concerns associated with unregulated peptide sources.
Reconstitution Protocol
Standard reconstitution procedure for lyophilized DSIP:
- Allow the vial to reach room temperature before opening (prevents moisture condensation on the lyophilized powder)
- Clean the vial stopper with an alcohol swab
- Using a sterile syringe, draw the desired volume of bacteriostatic water
- Insert the needle through the stopper and direct the water stream against the glass wall of the vial, not directly onto the powder
- Allow the peptide to dissolve naturally by gentle swirling. Do not shake vigorously, as this can cause peptide denaturation and aggregation
- Once fully dissolved, the solution should be clear and colorless. Cloudiness or particulate matter indicates potential contamination or aggregation
- Label the vial with the reconstitution date and concentration
- Store refrigerated at 2-8 degrees C
Common reconstitution concentrations and resulting dose volumes for a 5 mg vial:
| Bacteriostatic Water Volume | Resulting Concentration | Volume per 100 mcg | Volume per 250 mcg |
|---|---|---|---|
| 1.0 mL | 5.0 mg/mL (5000 mcg/mL) | 0.02 mL (2 units) | 0.05 mL (5 units) |
| 2.0 mL | 2.5 mg/mL (2500 mcg/mL) | 0.04 mL (4 units) | 0.10 mL (10 units) |
| 2.5 mL | 2.0 mg/mL (2000 mcg/mL) | 0.05 mL (5 units) | 0.125 mL (12.5 units) |
| 5.0 mL | 1.0 mg/mL (1000 mcg/mL) | 0.10 mL (10 units) | 0.25 mL (25 units) |
For detailed reconstitution calculations for various vial sizes and target doses, the FormBlends Dosing Calculator provides a convenient online tool.
DSIP Research Summary and Evidence Grading
Strength of Evidence by Application
Not all of DSIP's proposed applications are equally supported by evidence. The following summary grades the available evidence for each application area, helping researchers and clinicians prioritize based on the current state of knowledge.
| Application | Evidence Grade | Basis | Key Limitations |
|---|---|---|---|
| Slow-wave sleep enhancement | B (Moderate) | Multiple animal studies; several human trials showing objective improvement | Mixed subjective outcomes; variable response; dated methodology |
| Alcohol withdrawal symptom relief | B- (Moderate-Low) | Clinical trials with high response rates | Small samples; high dropout; 1980s methodology; no modern replication |
| Opioid withdrawal symptom relief | B- (Moderate-Low) | Clinical trials with 97% response rate | Same limitations as alcohol withdrawal data |
| Pain modulation | C+ (Low-Moderate) | Consistent preclinical data; one small clinical pilot | Very limited human data; no placebo-controlled pain trials |
| HPA axis/cortisol modulation | B- (Moderate-Low) | Consistent animal data; variable human data | Human studies show inconsistent cortisol effects |
| Growth hormone stimulation | C (Low) | Strong animal data contradicted by human studies | Human studies show no GH effect; species-specific difference likely |
| Neuroprotection | C (Low) | Preclinical data (antioxidant, mitochondrial, stroke model) | No human neuroprotection data |
| Immunomodulation | C- (Very Low) | Limited in vitro and animal data | No human immune outcome data |
| Athletic recovery | D (Speculative) | Theoretical extrapolation from sleep and GH data | No studies in athletic populations |
| Anti-depression | D (Speculative) | Theoretical based on HPA and sleep effects | No studies in depressed populations |
What the Evidence Supports
Based on the totality of available evidence, the following statements can be made with reasonable confidence:
- DSIP is biologically active. Across dozens of studies using multiple experimental systems, DSIP consistently produces measurable biological effects. Whatever its receptor status, the peptide is not pharmacologically inert.
- DSIP influences sleep architecture. The evidence for delta sleep enhancement is strongest in animal models but is supported by objective measures in human studies. The effects are modulatory rather than dramatic, and individual variability is high.
- DSIP interacts with the endogenous opioid system. The naloxone blockade of DSIP's analgesic effects and the morphine cross-tolerance data firmly establish opioidergic involvement. The nature of this interaction is modulatory rather than agonistic.
- DSIP shows efficacy in withdrawal syndromes. The clinical trial data, while methodologically limited, consistently show high response rates across multiple studies. The multi-system mechanism provides a plausible explanation for broad withdrawal symptom relief.
- DSIP has a favorable short-term safety profile. Across hundreds of exposed subjects in clinical studies, serious adverse events were not reported. The side effect profile compares favorably to conventional sleep medications.
- DSIP's long-term effects are unknown. The absence of long-term safety and efficacy data represents the most significant gap in the current evidence base.
What the Evidence Does Not Support
Equally important is identifying claims that are not well-supported by current evidence:
- DSIP is not a proven growth hormone secretagogue in humans, despite animal data suggesting this effect
- DSIP has not been demonstrated to improve athletic performance or recovery in any controlled study
- DSIP has not been shown to treat or prevent neurodegenerative diseases in humans
- DSIP has not been evaluated in any psychiatric disorder, including depression, anxiety, or PTSD
- No evidence supports specific dosing protocols for subcutaneous DSIP, as the clinical trials all used IV administration
The Bottom Line
DSIP represents a genuinely interesting research compound with documented biological effects that span sleep, pain, stress, and withdrawal. Its profile is unique among sleep-related peptides, and its multi-system modulatory approach aligns with contemporary understanding of sleep as a complex, network-regulated process. However, the evidence base has significant gaps, including no identified receptor, no gene, limited human trial data (mostly from the 1980s), and absent long-term safety information. Individuals interested in DSIP should approach it as an experimental compound with promising but incomplete evidence, not as a proven therapeutic agent. The Free Assessment at FormBlends can help evaluate whether DSIP or other research compounds align with individual goals and circumstances.

Figure 10: Evidence strength summary for DSIP applications. Sleep and withdrawal applications have the strongest evidence base, while athletic, mental health, and anti-aging applications remain primarily theoretical.
DSIP and Circadian Disruption Syndromes
Shift Work and Non-Standard Schedules
Approximately 20% of the global workforce engages in some form of shift work, and the health consequences are well documented: increased rates of cardiovascular disease, metabolic syndrome, gastrointestinal disorders, certain cancers, and mental health conditions. At the center of these health impacts is circadian disruption - the mismatch between the body's internal clock and the imposed work-rest schedule.
Shift workers face a specific sleep architecture challenge that distinguishes their condition from simple insomnia. When attempting to sleep during the biological day, the circadian alerting signal actively opposes sleep, resulting in shorter total sleep time, reduced slow-wave sleep percentage, and fragmented sleep architecture. Even when shift workers achieve seemingly adequate hours of sleep, the quality composition of that sleep is degraded compared to nighttime sleep.
DSIP's potential relevance to shift work sleep disruption rests on several properties:
Delta sleep enhancement independent of circadian timing: If DSIP promotes slow-wave sleep through direct neural mechanisms rather than solely through circadian pathways, it could potentially enhance deep sleep even when sleep occurs during the biological day. This would address a limitation of melatonin, which primarily works through circadian mechanisms and may be less effective when the circadian system is actively promoting wakefulness.
Cortisol modulation: Cortisol levels are normally high during the biological day, regardless of the work schedule. Shift workers attempting to sleep during their biological day face elevated cortisol that impairs slow-wave sleep. DSIP's ability to attenuate cortisol could remove this hormonal barrier to deep sleep during daytime sleep periods.
Melatonin stimulation: DSIP's stimulation of pineal melatonin production could reinforce a pharmacological melatonin supplement, providing both the timing signal (exogenous melatonin) and enhanced endogenous production (DSIP-stimulated) during attempted daytime sleep.
No studies have specifically evaluated DSIP in shift workers. The application remains theoretical but represents a population with a clear unmet need for sleep quality enhancement that existing interventions incompletely address.
Jet Lag and Transmeridian Travel
Jet lag occurs when rapid travel across time zones creates a mismatch between the internal circadian clock and the local light-dark cycle. The primary symptoms - insomnia, daytime sleepiness, impaired cognitive function, gastrointestinal disturbance - reflect the body's struggle to synchronize with the new time zone.
Melatonin is the established first-line treatment for jet lag, and its efficacy is well-documented for accelerating circadian re-entrainment. However, melatonin primarily addresses sleep timing rather than sleep quality. Travelers who successfully fall asleep at the appropriate local time may still experience poor-quality sleep with reduced slow-wave sleep for several days until full circadian adaptation occurs.
DSIP could theoretically complement melatonin in jet lag management by enhancing sleep depth during the re-entrainment period. The combination of melatonin (for timing) and DSIP (for depth) would address both dimensions of jet lag-disrupted sleep. Pinealon represents another potential complement, supporting circadian adaptation through pineal gland peptide signaling rather than direct melatonin receptor activation.
Seasonal Circadian Disruption
In higher latitudes, extreme variations in daylight duration between summer and winter create seasonal circadian challenges. Extended summer daylight can delay sleep onset and reduce melatonin production, while short winter days can produce excessive melatonin and seasonal affective disorder (SAD). Both extremes affect sleep architecture, with the winter pattern often associated with increased but non-restorative sleep (hypersomnia with reduced slow-wave percentage).
DSIP's potential for seasonal circadian disruption is unexplored in published research. However, its combined effects on melatonin production and delta sleep architecture could theoretically provide dual-axis support during seasonal transitions, particularly the fall-to-winter transition when declining daylight disrupts circadian timing while increasing the need for restorative deep sleep.
DSIP Comparative Pharmacology: Positioning Among Sleep-Active Compounds
Mechanism-Based Classification
Sleep-active compounds can be classified by their primary mechanism of action. This framework helps position DSIP relative to other options and clarifies why different compounds are suited to different types of sleep problems.
GABA-A receptor agonists and modulators: This category includes benzodiazepines (diazepam, lorazepam, temazepam), Z-drugs (zolpidem, zaleplon, eszopiclone), and the newer GABA-A-selective agents. They increase total sleep time and reduce sleep onset latency but suppress slow-wave sleep. Dependence risk is significant. They are effective for acute insomnia but problematic for long-term use.
Orexin receptor antagonists (DORAs): Suvorexant and lemborexant block the wake-promoting orexin system. They do not suppress slow-wave sleep and show less dependence potential than GABA-A agents. However, they do not specifically enhance deep sleep either - they remove a wake signal rather than adding a sleep signal. Morning drowsiness and rare sleep paralysis are reported side effects.
Melatonin receptor agonists: Melatonin (OTC supplement) and ramelteon (prescription MT1/MT2 agonist) primarily affect circadian sleep timing. They reduce sleep onset latency modestly, with minimal effects on sleep architecture, no dependence risk, and very few side effects. They are most effective for circadian misalignment rather than primary insomnia or reduced sleep depth.
Histamine receptor antagonists: Low-dose doxepin (Silenor) and other sedating antihistamines reduce histamine-mediated arousal. They can improve sleep maintenance but do not specifically enhance slow-wave sleep. Anticholinergic side effects (dry mouth, constipation, cognitive impairment) are common with older antihistamines.
Multi-system modulators: This is where DSIP belongs, along with a few other compounds that influence multiple neurotransmitter systems simultaneously. DSIP's interaction with GABAergic, opioidergic, glutamatergic, and HPA pathways distinguishes it from single-target agents. The advantage is a potentially more physiological, balanced influence on sleep regulation; the disadvantage is less predictable effects and the difficulty of optimizing dosing when multiple targets are being engaged simultaneously.
Why "Sleep Quality" Is Not "Sleep Quantity"
A persistent confusion in sleep medicine and popular culture is the equation of sleep duration with sleep quality. The two are related but distinct concepts, and this distinction is essential for understanding DSIP's potential value.
Sleep quantity is simply total sleep time - the number of hours spent asleep. This is what most sleep medications effectively increase. A person who takes zolpidem might sleep 7 hours instead of 5, achieving a quantitative improvement.
Sleep quality encompasses the composition and continuity of sleep - the percentage of time in each sleep stage, the number of arousals, the spectral characteristics of EEG during each stage, and the degree to which sleep cycles progress normally through stages. A person can sleep 8 hours and still have poor sleep quality if slow-wave sleep is suppressed, REM is fragmented, or frequent brief arousals prevent sustained deep sleep.
Most existing sleep medications improve quantity at the expense of quality. Benzodiazepines and Z-drugs are the clearest examples: they reliably increase total sleep time but alter the composition of that sleep in ways that reduce its restorative value. This is why people often feel unrefreshed after medication-induced sleep despite sleeping "enough" hours.
DSIP's research profile suggests the opposite pattern: modest or inconsistent effects on sleep quantity but potentially meaningful improvements in sleep quality through enhanced slow-wave sleep. This quality-focused profile is unique among studied sleep compounds and represents its primary theoretical advantage.
This quality-versus-quantity distinction also helps explain the puzzling results of the Schneider-Helmert insomnia trial, where objective measures improved but subjective ratings did not. Chronic insomniacs are typically most focused on sleep quantity - how many hours they slept, how long it took to fall asleep. They may not subjectively notice improvements in sleep depth or architecture even when these improvements are measurable on polysomnography. The mismatch between objective and subjective outcomes may reflect what was being measured (quality improvement) versus what patients were paying attention to (quantity).
The Unmet Need in Sleep Medicine
Modern sleep medicine has effective tools for several aspects of sleep dysfunction but lacks good options for one specific problem: selectively enhancing slow-wave sleep in people who sleep adequate hours but lack sufficient deep sleep.
This problem is extremely common. It affects older adults (age-related delta sleep decline), stressed individuals (cortisol-mediated slow-wave suppression), athletes (training-induced HPA activation), post-menopausal women (hormonal changes affecting sleep architecture), and individuals recovering from substance use disorders (persistent sleep architecture disruption).
Current options for these populations are limited:
- Sodium oxybate (Xyrem): Effectively enhances slow-wave sleep but is a Schedule III controlled substance with significant abuse potential, CNS depression risk, and narrow therapeutic index. Approved only for narcolepsy-related conditions.
- Gabapentin/pregabalin: May increase slow-wave sleep at higher doses, but side effects (dizziness, weight gain, cognitive impairment) limit their utility as sleep-specific agents.
- Acoustic slow-wave enhancement: Playing timed audio tones during sleep can enhance delta waves through auditory entrainment. This non-pharmacological approach is promising but requires specialized equipment and has practical limitations.
- Exercise: Regular aerobic exercise increases slow-wave sleep, but the effect takes weeks to develop and requires sustained commitment.
DSIP, if its slow-wave sleep enhancement could be reliably replicated in modern trials with improved delivery methods, would address this specific unmet need. It would not compete with existing sleep medications for their primary indications (acute insomnia, circadian disorders) but would fill a gap that currently has no good pharmacological solution.
This positioning is both DSIP's greatest strength and its greatest challenge. The market for delta sleep enhancement is large and growing, but validating a compound in this space requires sleep architecture endpoints (polysomnography-based measures) rather than the simpler efficacy measures (total sleep time, sleep onset latency) that most sleep drug trials use. Demonstrating clinically meaningful improvement in slow-wave sleep percentage requires larger samples, more expensive monitoring, and longer treatment periods than demonstrating faster sleep onset.
DSIP and Hormonal Health: Beyond Growth Hormone
The Sleep-Hormone Network
Sleep and hormonal function are inseparable. The brain's sleep centers and endocrine control centers share anatomical space in the hypothalamus, and many hormones follow sleep-dependent secretion patterns. This means that any compound affecting sleep architecture will inevitably influence hormonal function, and conversely, any hormonal effects will feed back to influence sleep.
DSIP sits at the center of this sleep-hormone network. Beyond the growth hormone and cortisol effects already discussed, several additional hormonal connections deserve attention.
Insulin Sensitivity and Glucose Metabolism
Slow-wave sleep is the period when the brain is least metabolically active and glucose utilization drops to its lowest point. Disruption of slow-wave sleep - even within a normal-duration sleep period - has been shown to reduce insulin sensitivity by up to 25% in healthy young adults, producing glucose tolerance levels comparable to pre-diabetic states.
The mechanism involves several pathways: reduced slow-wave sleep increases sympathetic nervous system activity, elevates cortisol, and impairs glucose transport into skeletal muscle. These effects are mediated through the same HPA axis and autonomic pathways that DSIP appears to modulate.
While no studies have directly measured DSIP's effects on glucose metabolism, the theoretical connection is straightforward. If DSIP enhances slow-wave sleep, the downstream metabolic benefits of improved deep sleep - including better insulin sensitivity, reduced overnight cortisol, and normalized sympathetic tone - would be expected to follow. This is not a direct metabolic effect of DSIP but rather an indirect consequence of sleep architecture improvement.
Thyroid Function
Thyroid-stimulating hormone (TSH) secretion follows a circadian pattern with a nocturnal surge that begins before sleep onset and peaks during the early sleep hours. This TSH surge is partially sleep-dependent and partially circadian-driven, with slow-wave sleep exerting a suppressive influence on TSH secretion.
The interaction between DSIP and thyroid function has received minimal research attention. Given DSIP's hypothalamic-level effects and its influence on other pituitary hormones, some degree of thyroid axis interaction is plausible but uncharacterized. This represents a gap in the safety assessment of long-term DSIP use, as chronic hypothalamic-level modulation could theoretically alter the TSH set point.
Reproductive Hormones
Sleep architecture influences reproductive hormone secretion in both sexes. In men, testosterone production during sleep is linked to sleep quality, with slow-wave sleep contributing to the nocturnal testosterone surge that produces the characteristic morning peak. Sleep restriction reduces testosterone proportionally to the reduction in deep sleep time.
In women, sleep disruption affects luteinizing hormone (LH) pulse patterns, potentially influencing ovulatory function and menstrual regularity. DSIP has been reported to modulate LH pulsatility in some studies, though the data are limited and the clinical significance is unclear.
The reproductive hormone connection adds another dimension to DSIP's potential relevance for population groups experiencing hormonal changes. Peri-menopausal and post-menopausal women, for example, frequently experience both hormonal disruption and sleep quality decline, and interventions that address both simultaneously could provide compounded benefit.
Prolactin and Immune Function
Prolactin secretion increases during sleep, with the nocturnal rise beginning shortly after sleep onset and peaking during the second half of the night. Beyond its role in lactation, prolactin functions as an immunomodulatory hormone, promoting immune cell proliferation and activity.
The human study of DSIP in women found no significant effect on prolactin secretion, suggesting that DSIP's influence on this hormone is minimal. However, by enhancing overall sleep quality, DSIP could indirectly support the sleep-dependent prolactin surge and its downstream immune effects.
For those interested in the broader relationship between peptides and hormonal optimization, the Peptide Hub at FormBlends provides comprehensive coverage of endocrine-active peptides, including CJC-1295/Ipamorelin for growth hormone optimization and Epithalon for pineal-endocrine support.
Detailed Analysis of Key DSIP Studies
Study 1: The Original Monnier and Schoenenberger Isolation (1974-1977)
Objective: To isolate and characterize the chemical factor responsible for transmitting sleep from sleeping donor rabbits to awake recipients through dialyzed cerebral venous blood.
Methodology: The experimental design was elegant in its simplicity. Donor rabbits were placed into a sleep state through low-frequency electrical stimulation of the intralaminar thalamic nucleus. Cerebral venous blood was then collected through an extracorporeal dialysis circuit, which selectively captured small molecules (under approximately 10,000 daltons) while excluding larger proteins, blood cells, and immune factors. This dialysate was then infused into the lateral or third ventricles of awake recipient rabbits while their EEG and motor activity were continuously monitored.
Key findings: Recipients showed clear EEG changes within 30-60 minutes of dialysate infusion, including increased sleep spindle activity and the emergence of high-amplitude delta waves characteristic of deep slow-wave sleep. Motor activity decreased concurrently. The active fraction was progressively purified through Sephadex gel filtration, ion exchange chromatography, and high-performance liquid chromatography, ultimately yielding a pure nonapeptide with the sequence Trp-Ala-Gly-Gly-Asp-Ala-Ser-Gly-Glu.
Significance: This was the first isolation of a specific peptide from cerebral venous blood that could transfer a sleep-like state between animals. It provided direct evidence for humoral (blood-borne) sleep factors, supporting the chemical theory of sleep regulation proposed by Pieron decades earlier. The work earned the name "Delta Sleep-Inducing Peptide" for the compound, a name that has proven both iconic and somewhat misleading given the complexity of DSIP's subsequent research history.
Limitations: The purification process required enormous quantities of starting material and yielded very small amounts of peptide. Some subsequent researchers questioned whether the purification process might generate the nonapeptide as an artifact of protein degradation rather than as a naturally occurring peptide. This concern has never been fully resolved, though DSIP-like immunoreactivity detected in multiple tissues using specific antibodies supports its endogenous existence.
Study 2: The Dick and Granstrom Withdrawal Trials (1984)
Objective: To evaluate DSIP's efficacy in managing acute withdrawal symptoms from alcohol and opiates, based on the hypothesis that DSIP's opioid receptor interactions could modulate withdrawal physiology.
Methodology: Two related studies were conducted. In the first, 67 patients (28 alcoholics, 39 opiate addicts) received intravenous DSIP at 25 nmol/kg as the sole treatment for withdrawal symptoms. No other medications were administered. Clinical symptoms and signs were monitored by physicians blinded to treatment timing (though the study was not placebo-controlled). In the second, larger study, 107 patients (47 alcoholics, 60 opiate addicts) received similar treatment with systematic clinical outcome assessment.
Key findings: The results were striking by any standard. In the larger study, 87% of alcoholics showed complete disappearance or marked improvement of withdrawal symptoms after DSIP administration. For opiate addicts, the response rate was even higher at 97%. Onset of action was rapid, particularly in alcoholics, with many patients showing improvement within hours. The clinical course was typically shorter for alcoholics than for opiate addicts, and opiate withdrawal required a greater number of DSIP injections.
Anxiety reduction was a prominent feature of the response, consistent with DSIP's proposed GABAergic and HPA axis effects. Sleep improvement was also noted, though formal polysomnographic monitoring was not performed. Side effects were minimal, limited to headaches in a small number of patients.
Significance: These studies remain the most impressive clinical data for DSIP. Response rates of 87-97% for a single-agent treatment of withdrawal syndromes would be remarkable for any medication, and the absence of the side effects typical of standard withdrawal treatments (benzodiazepine sedation, clonidine hypotension, methadone dependence) makes the results even more noteworthy.
Limitations: The studies had several significant methodological weaknesses by modern standards. They were not placebo-controlled (though the severity and physiological nature of withdrawal symptoms makes pure placebo response unlikely to explain the results). The loss-to-follow-up rate was 27% in the first study, introducing potential selection bias. Sample sizes were modest. Modern randomization and blinding standards were not fully applied. And perhaps most critically, no comparable study has been conducted in the subsequent four decades, leaving these results unvalidated by independent replication.
Study 3: The Schneider-Helmert Insomnia Trial (1987)
Objective: To evaluate DSIP's efficacy for chronic insomnia in a methodologically rigorous double-blind, placebo-controlled design.
Methodology: Fourteen patients with chronic insomnia (duration at least one year, no concurrent sleep medication) were enrolled in a crossover design comparing five consecutive evenings of intravenous DSIP (25 nmol/kg) with five consecutive evenings of placebo. Full polysomnographic monitoring was performed throughout, including detailed EEG spectral analysis, sleep staging, and measurement of multiple sleep continuity parameters. Subjective sleep quality was assessed using standardized questionnaires.
Key findings: Objective polysomnographic measures showed statistically significant improvements with DSIP compared to placebo on two key parameters: sleep efficiency (the ratio of time asleep to time in bed) increased, and sleep onset latency decreased. Total sleep time showed a trend toward improvement that did not reach statistical significance. Changes in specific sleep stage percentages were variable, with some subjects showing increased slow-wave sleep but others showing minimal change. Most crucially, subjective sleep quality ratings did not differ between DSIP and placebo conditions.
Significance: This study's importance lies in its methodological rigor and in the tension between its positive objective findings and negative subjective findings. It demonstrates that DSIP produces measurable, objectively verified improvements in sleep, countering claims that DSIP is pharmacologically inert. But it also demonstrates that these objective improvements did not translate into perceived benefit over a five-night treatment period, raising questions about clinical utility.
Interpretation considerations: The five-night treatment period may have been too short for chronic insomniacs to develop subjective awareness of improved sleep quality. Chronic insomniacs often have ingrained negative sleep perceptions that require weeks to months of improved sleep to modify. The DSIP "delayed effect" phenomenon, where benefits become more apparent after treatment cessation, was not captured in this design. And the use of subjective scales designed for more dramatic sleep changes (like those produced by sedative-hypnotics) may not have been sensitive to the subtler, quality-focused improvements that DSIP produces.
Study 4: The Nakamura P-DSIP Rat Study (1989)
Objective: To evaluate the effects of the phosphorylated analogue of DSIP (P-DSIP) on sleep architecture in freely moving, unrestrained rats.
Methodology: Rats were implanted with chronic intracerebroventricular (ICV) cannulae and EEG/EMG recording electrodes. After recovery, they received continuous 10-hour nocturnal ICV infusions of P-DSIP at 0.5 nmol, with sleep architecture monitored by polysomnographic recording. Control infusions of vehicle were performed on separate nights in the same animals, allowing within-subject comparison.
Key findings: P-DSIP produced a 22% increase in slow-wave sleep time and, remarkably, an 81% increase in paradoxical (REM) sleep time during the infusion period. Both increases were statistically significant compared to vehicle control nights. The enhancement of both SWS and REM sleep is unusual among sleep-modulating compounds, most of which preferentially affect one stage at the expense of the other.
Significance: This study demonstrates that modified DSIP analogues can produce substantially greater sleep effects than the native peptide, suggesting that DSIP's relatively modest effects in human trials might be dramatically improved with optimized analogues. The simultaneous enhancement of both SWS and REM sleep is a particularly attractive profile, as it suggests preservation of normal sleep architecture proportions with overall deepening of both restorative sleep phases. The finding that phosphorylation of a single serine residue so dramatically enhances activity provides a clear target for further structural optimization.
Limitations: The study used intracerebroventricular administration, which bypasses all peripheral pharmacokinetic barriers. P-DSIP's efficacy when administered peripherally (subcutaneously or intravenously) is unknown, and the dose translation from rat ICV to human peripheral administration is not straightforward. P-DSIP has never been evaluated in human subjects.
Study 5: The 2024 DSIP Fusion Peptide Study
Objective: To develop DSIP fusion peptides capable of crossing the blood-brain barrier more efficiently and evaluate their efficacy in an insomnia mouse model.
Methodology: Researchers used the yeast Pichia pastoris to produce secreted fusion peptides combining DSIP with blood-brain barrier-penetrating sequences. These fusion constructs were purified and tested in mice with PCPA-induced insomnia (PCPA depletes serotonin, producing a model of sleep disruption). Sleep parameters were assessed through behavioral observation and EEG monitoring.
Key findings: The DSIP fusion peptides demonstrated efficacy in the insomnia mouse model, with recovery of sleep parameters toward baseline levels. The fusion approach successfully delivered DSIP activity across the blood-brain barrier, addressing one of the primary pharmacokinetic limitations of native DSIP.
Significance: This study represents the most recent innovation in DSIP research and demonstrates that the field is still active, with new technological approaches being applied to overcome the limitations that hindered earlier clinical development. The use of recombinant production in yeast (rather than chemical synthesis) also opens possibilities for scalable, cost-effective manufacturing of DSIP-based therapeutics.
Limitations: This is a single preclinical study in mice. The gap between mouse efficacy and human clinical utility remains large, and the specific fusion peptide constructs would require extensive safety and pharmacokinetic characterization before human testing could be considered.
DSIP Global Research Landscape
Regional Research Patterns
DSIP research has followed an interesting geographical distribution over the decades, with different regions contributing different types of knowledge to the overall picture.
Switzerland (1970s-1980s): The birthplace of DSIP research. The Basel group led by Schoenenberger and Monnier performed the original isolation, characterization, and early physiological studies. Swiss researchers established the fundamental phenomenology of DSIP, including its sleep-promoting effects in animals, its amino acid sequence, and its basic pharmacological profile.
Germany and Austria (1980s-1990s): The major clinical trials were conducted in German-speaking European centers. The Schneider-Helmert insomnia trial, the Dick and Granstrom withdrawal studies, and several other clinical evaluations were all performed in this region. This geographical concentration of clinical expertise means that the human evidence base for DSIP is largely derived from Central European populations, which may limit generalizability.
Russia and the former Soviet Union (1980s-present): Russian researchers have made substantial contributions to understanding DSIP's mechanism of action, particularly its antioxidant properties, stress-protective effects, and neuroprotective potential. The Russian peptide bioregulation research tradition, led by Vladimir Khavinson and others, has also produced related sleep peptides including Pinealon and Epithalon. Much of this research has been published in Russian-language journals, limiting its accessibility to the international research community.
Japan (1980s-1990s): Japanese researchers contributed important studies on DSIP distribution, metabolism, and immune effects. The Nakamura group's P-DSIP sleep architecture study was conducted in Japan, as were several studies on DSIP-like immunoreactivity in various tissues.
United States (limited): Despite the large US sleep research community, relatively few DSIP studies have been conducted in American institutions. The review articles by Graf and Kastin (1984, 1986) at the Veterans Administration Medical Center provided important synthesis of the early literature, but original clinical research in the US has been minimal. This may partly reflect FDA regulatory requirements that made clinical DSIP research more difficult in the US context.
China (2020s-present): The most recent DSIP research, including the 2024 fusion peptide study, has emerged from Chinese institutions. This reflects broader trends in peptide research, where Chinese universities and biotech companies have become increasingly active in developing novel peptide therapeutics.
Publication Trends
DSIP publication volume peaked in the mid-1980s to early 1990s, declined through the 2000s, and has shown a modest resurgence in the 2020s. This pattern reflects the general arc of DSIP research: initial excitement following discovery, productive clinical investigation in the 1980s, growing frustration with unresolved mechanistic questions in the 1990s, and renewed interest driven by new technologies and evolving perspectives on sleep science.
The renewed interest is evident in several indicators: new DSIP analogue studies, the fusion peptide work, increasing mentions in peptide therapy reviews, and growing consumer awareness. Whether this renewed interest will translate into the rigorous, well-powered clinical trials needed to resolve DSIP's clinical utility remains to be seen.
Intellectual Property and Development Pathway
Unlike many pharmaceutical peptides, DSIP has no strong patent protection from a single developer. The original discovery occurred in an academic setting, and the peptide sequence has been in the public domain for decades. This has made DSIP accessible for research and compounding but has also removed the financial incentive for any single company to invest in the expensive Phase II and Phase III clinical trials needed for regulatory approval.
This "orphan" status is a common challenge for naturally occurring peptides and represents a structural barrier to DSIP's clinical development. Novel analogues (like the fusion peptides) might offer patentable compositions that justify commercial development investment, but these would need to start the regulatory pathway essentially from scratch.
In the meantime, DSIP remains available through compounding pharmacies and research chemical suppliers, existing in a regulatory gray zone that provides access for research and off-label use without the safety guarantees of an FDA-approved product. FormBlends provides quality-controlled DSIP with third-party testing, addressing some of the quality concerns associated with this regulatory environment.
DSIP and Thermoregulation
The Temperature-Sleep Connection
Core body temperature and sleep are intimately connected through shared neural circuitry in the hypothalamus. The preoptic area of the hypothalamus contains both temperature-sensitive neurons and sleep-active neurons, and these populations overlap significantly. This anatomical overlap means that temperature regulation and sleep regulation are not merely correlated - they share actual neural substrates.
Normal sleep onset is preceded by a drop in core body temperature of approximately 1-2 degrees Fahrenheit, driven by increased peripheral vasodilation that allows heat to radiate from the extremities. This temperature decline is both a consequence of the circadian system (the SCN reduces the thermoregulatory set point in the evening) and a facilitator of sleep onset (the warm-to-cool transition promotes the transition from wakefulness to sleep).
Slow-wave sleep represents the period of lowest thermoregulatory activity. During deep delta sleep, the body's ability to regulate temperature is reduced, and the temperature set point reaches its circadian nadir. The coordination between maximum delta sleep intensity and minimum body temperature is not coincidental - both are manifestations of reduced hypothalamic arousal.
DSIP's Thermoregulatory Effects
DSIP has been shown to influence body temperature regulation in animal studies, though the effects are complex and context-dependent. Some observations from the research literature include:
- DSIP administration has been associated with mild hypothermic effects in some experimental conditions, consistent with activation of preoptic sleep-thermoregulatory neurons
- The thermoregulatory effects of DSIP show circadian variation, with greater temperature-lowering effects during the dark (active) phase in nocturnal rodents
- DSIP's thermoregulatory effects may be partly mediated through prostaglandin pathways, as prostaglandin D2 (a known sleep factor) also produces hypothermia through preoptic area activation
- The temperature effects of DSIP are modest compared to its sleep effects, suggesting that thermoregulation is a secondary rather than primary pathway for sleep promotion
The practical implication is that DSIP may work best when the thermal environment supports its sleep-promoting effects. A cool sleeping environment (65-68 degrees F / 18-20 degrees C) facilitates the natural temperature decline associated with sleep onset and may synergize with DSIP's thermoregulatory effects. Hot sleeping environments, which oppose the natural temperature decline, could potentially reduce DSIP's efficacy by creating a thermoregulatory conflict with the peptide's sleep-promoting actions.
Temperature Monitoring as a DSIP Response Marker
Wearable temperature monitoring devices (available commercially as rings, wristbands, and bed sensors) can now track core body temperature proxies throughout the night with reasonable accuracy. These devices reveal the circadian temperature pattern, the pre-sleep temperature decline, and the overnight temperature nadir.
Temperature monitoring could theoretically serve as a non-invasive biomarker for DSIP response. If DSIP genuinely enhances slow-wave sleep through hypothalamic mechanisms, responders might show a deeper overnight temperature nadir, earlier temperature decline onset, or more stable low temperature maintenance during the deep sleep window. This approach has not been validated for DSIP specifically, but it represents a practical way to assess physiological response without full polysomnographic monitoring.
How to Interpret DSIP Research: A Guide for Critical Reading
Common Methodological Issues in DSIP Literature
The DSIP research literature spans five decades and reflects the evolving standards of biomedical research during that period. Several recurring methodological issues should be considered when evaluating DSIP studies:
Small sample sizes: Most DSIP clinical studies enrolled fewer than 20 subjects. With DSIP's high inter-individual variability, these sample sizes provide limited statistical power to detect real effects. A true effect of moderate size could easily be missed (Type II error), and a chance finding could appear significant (Type I error). Modern clinical trial standards would typically require 50-100 or more subjects per group for a sleep study, far exceeding anything in the DSIP literature.
Route of administration mismatch: The clinical trials all used intravenous DSIP, but current research protocols predominantly describe subcutaneous administration. The pharmacokinetic differences between these routes are substantial, and efficacy data from IV studies cannot be directly applied to SC protocols without dose-adjustment studies that have not been performed.
Outdated sleep staging criteria: The sleep studies from the 1980s used Rechtschaffen and Kales staging criteria, which have since been superseded by the American Academy of Sleep Medicine (AASM) 2007 criteria. While the differences are relatively minor, they complicate direct comparison with modern sleep data.
Short treatment durations: Most studies treated subjects for 3-7 days, which may be insufficient for a modulatory peptide that appears to produce delayed and cumulative effects. The "programming" action described by some researchers suggests that longer treatment periods might produce substantially different outcomes.
Publication bias: As with any research area, there is likely selective publication of positive results. Negative DSIP studies may exist in file drawers, unpublished. This concern is heightened by the absence of clinical trial registries during the era when most DSIP studies were conducted.
Immunoassay specificity: Studies reporting endogenous DSIP levels used radioimmunoassay with antibodies whose cross-reactivity has been questioned. It is possible that some "DSIP-like immunoreactivity" detected in tissues represents cross-reacting peptides rather than authentic DSIP.
What to Look for in Future DSIP Research
For the field to advance meaningfully, future DSIP studies should incorporate:
- Adequate sample sizes (minimum 30-50 subjects per group for sleep studies)
- Placebo-controlled, double-blind design with proper randomization
- Pre-registered protocols with primary and secondary endpoints defined before data collection
- Modern polysomnographic methods including spectral analysis of delta power density, not just sleep staging
- Subcutaneous administration reflecting current research protocols, with pharmacokinetic characterization
- Treatment periods of at least 4 weeks to capture delayed and cumulative effects
- Follow-up after treatment cessation to assess the "delayed effect" phenomenon and durability of benefit
- Biomarker assessment including cortisol, melatonin, and growth hormone at relevant time points
- Both objective and subjective outcomes with scales validated for detecting quality-focused sleep improvements
- Responder analysis to identify predictors of DSIP response
A single well-designed study incorporating these elements could contribute more to our understanding of DSIP than the entire body of existing literature combined. The scientific community awaits such a study, which would either validate DSIP's clinical potential or definitively establish its limitations.
Frequently Asked Questions
References
- Monnier M, Schoenenberger GA. Characterization, sequence, synthesis and specificity of a delta-EEG (sleep)-inducing peptide. Experientia. 1977;33:548-549.
- Schoenenberger GA, Monnier M. Characterization of a delta-electroencephalogram (-sleep)-inducing peptide. Proc Natl Acad Sci USA. 1977;74(3):1282-1286. doi:10.1073/pnas.74.3.1282
- Graf MV, Kastin AJ. Delta-sleep-inducing peptide (DSIP): a review. Neurosci Biobehav Rev. 1984;8(1):83-93. doi:10.1016/0149-7634(84)90022-8
- Kovalzon VM, Strekalova TV. Delta sleep-inducing peptide (DSIP): a still unresolved riddle. J Neurochem. 2006;97(2):303-309. doi:10.1111/j.1471-4159.2006.03693.x
- Schneider-Helmert D, Schoenenberger GA. Effects of DSIP in man. Multifunctional psychophysiological properties besides induction of natural sleep. Neuropsychobiology. 1983;9(4):197-206. doi:10.1159/000117964
- Schneider-Helmert D. Effects of delta sleep-inducing peptide on sleep of chronic insomniac patients. A double-blind study. Psychopharmacology. 1987;93(4):503-507. doi:10.1007/BF00207242
- Luthringer R, Minot R, Toussaint M, et al. Acute and delayed effects of DSIP (delta sleep-inducing peptide) on human sleep behavior. Int J Clin Pharmacol Res. 1984;4(2):83-89. PMID:6895513
- Nakamura A, Nakashima M, Sugao T, et al. The phosphorylated analogue of DSIP enhances slow wave sleep and paradoxical sleep in unrestrained rats. Psychopharmacology. 1989;97(1):35-38. doi:10.1007/BF00443409
- Dick P, Granstrom KE, Toth SE, et al. DSIP in the treatment of withdrawal syndromes from alcohol and opiates. Eur Neurol. 1984;23(5):364-371. doi:10.1159/000115721
- Dick P, Granstrom KE, Toth SE, et al. Successful treatment of withdrawal symptoms with delta sleep-inducing peptide, a neuropeptide with potential agonistic activity on opiate receptors. J Subst Abuse Treat. 1984;1(2):75-83. doi:10.1016/0740-5472(84)90031-1
- Dick P, Granstrom KE. Therapeutic effects of delta-sleep-inducing peptide (DSIP) in patients with chronic, pronounced pain episodes. A clinical pilot study. Eur Neurol. 1984;23(5):372-377. doi:10.1159/000115722
- Iyer KS, McCann SM. Delta sleep inducing peptide (DSIP) stimulates growth hormone (GH) release in the rat by hypothalamic and pituitary actions. Peptides. 1987;8(1):45-48. doi:10.1016/0196-9781(87)90163-X
- Kato N, Nagaki S, Takahashi Y, et al. Delta sleep-inducing peptide administration does not influence growth hormone and prolactin secretion in normal women. Psychoneuroendocrinology. 1993;18(1):85-89. doi:10.1016/0306-4530(93)90057-R
- Prudchenko IA, Stashevskaya LV, Mikhaleva II, Ivanov VT. Delta-sleep-inducing peptide: interaction with the components of the MAPK cascade. Bioorg Khim. 2010;36(6):751-758. doi:10.1134/S1068162010060063
- Khvatova EM, Samartzev VN, Zagoskin PP, et al. Delta sleep-inducing peptide (DSIP): effect on respiration activity in rat brain mitochondria and stress protective potency under experimental hypoxia. Peptides. 2003;24(2):307-311. doi:10.1016/S0196-9781(03)00040-8
- Pollard BJ, Pomfrett CJD. Delta sleep-inducing peptide. Eur J Anaesthesiol. 2001;18(7):419-422. doi:10.1097/00003643-200107000-00001
- Yehuda S, Mostofsky DI. Circadian effects of beta-endorphin, DSIP, and amphetamine on pentylenetetrazol-induced seizures. Peptides. 1993;14(2):203-205. doi:10.1016/0196-9781(93)90031-B
- Lysenko AV, Uskova NI. Effect of delta-sleep-inducing peptide on the activity of antioxidant enzymes in rat brain and liver during cold stress. Biull Eksp Biol Med. 1995;119(5):502-505. doi:10.1007/BF02445831
- Sudha S, Lakshmana MK, Bhatt MR, et al. Delta sleep-inducing peptide-like immunoreactivity in serum of humans and rats: studies by a sensitive two-site enzyme immunoassay. Neurochem Int. 1995;27(3):305-311. doi:10.1016/0197-0186(95)00028-7
- Riou F, Cespuglio R, Jouvet M. Hypnogenic properties of the vasoactive intestinal polypeptide in rats. C R Acad Sci. 1981;293:113-115.
- Silling K, Bhatt MR, Bhatt BA, et al. Transport of the synthetic peptide DSIP through the blood-brain barrier in rabbit. Experientia. 1990;46(11-12):1191-1193. doi:10.1007/BF01934026
- Yehuda S, Carasso RL. Modulation of learning, pain thresholds, and thermoregulation in the rat by preparations of free purified alpha-linolenic and linoleic acids: determination of the optimal omega 3-to-omega 6 ratio. Proc Natl Acad Sci USA. 1993;90(21):10345-10349.
- Kramarova LI, Kolaeva SG, Yukhananov RYu, Rozhanets VV. Content of DSIP, enkephalins, and ACTH in some tissues of active and hibernating ground squirrels. Comp Biochem Physiol C. 1991;98(1):53-56. doi:10.1016/0742-8413(91)90178-V
- Bondarenko TI, Maiboroda EA, Mikhaleva II, et al. Antioxidant effects of delta-sleep-inducing peptide. Bull Exp Biol Med. 2001;131(5):437-439. doi:10.1023/A:1017908130011
- Khvatova EM, Rubanova NS, Prudchenko IA, Mikhaleva II. Delta sleep inducing peptide: effect on lipid peroxidation and antioxidant defense system in the brain. Biull Eksp Biol Med. 2005;139(3):302-305.
- Sudakov SK, Nazarova GA, Alekseeva EV. Delta sleep-inducing peptide and deltorphin I interact in modulating the effects of electroacupuncture. Bull Exp Biol Med. 2003;135(3):235-238. doi:10.1023/A:1024167016534
- Feng G, Song Q, Zhang Q, et al. Pichia pastoris secreted peptides crossing the blood-brain barrier and DSIP fusion peptide efficacy in PCPA-induced insomnia mouse models. Front Pharmacol. 2024;15:1439536. doi:10.3389/fphar.2024.1439536
- Papalexi E, Bhatt MR, et al. Delta Sleep-Inducing Peptide Recovers Motor Function in SD Rats after Focal Stroke. Molecules. 2021;26(17):5173. doi:10.3390/molecules26175173
- Chiodera P, Volpi R, Coiro V, et al. Effect of delta-sleep-inducing peptide on pituitary hormone secretion in humans. Neuroendocrinology. 1990;52:267-271. doi:10.1159/000125601
- Kovalzon VM. Delta-sleep-inducing peptide: 30 years of investigation. Neurosci Behav Physiol. 2007;37(3):303-310. doi:10.1007/s11055-007-0015-z
- Shvirst NE, Chernysheva YM, Lysenko AV. Delta-sleep-inducing peptide stimulates melatonin, 5-methoxytryptophol and serotonin secretion from perifused rat pineal glands. Neurosci Lett. 1992;142(2):175-178. doi:10.1016/0304-3940(92)90367-G
- Graf MV, Kastin AJ. Delta-sleep-inducing peptide (DSIP): an update. Peptides. 1986;7(6):1165-1187. doi:10.1016/0196-9781(86)90145-6
- Schneider-Helmert D. DSIP in insomnia. Eur Neurol. 1984;23(5):358-363. doi:10.1159/000115720
- Mikhaleva II, Prudchenko IA, Ivanov VT, Voitenkov VB. Molecular mechanisms of the effects of delta sleep-inducing peptide. Chem Biol Drug Des. 2011;78(3):419-426. doi:10.1111/j.1747-0285.2011.01158.x
- Khavinson VKh, Linkova NS, Pronyaeva VE, et al. Pinealon promotes recovery of cognitive functions in old monkeys. Bull Exp Biol Med. 2012;152(4):443-445. doi:10.1007/s10517-012-1557-8