Executive Summary
Semax is a synthetic heptapeptide analog of the adrenocorticotropic hormone fragment ACTH(4-10), developed at the Institute of Molecular Genetics of the Russian Academy of Sciences. With over three decades of clinical use in Russia and Ukraine, it stands as one of the most thoroughly studied nootropic peptides in the world. Its primary actions center on upregulating brain-derived neurotrophic factor (BDNF), modulating dopaminergic and serotonergic neurotransmission, and providing broad-spectrum neuroprotection following ischemic injury.
Key Takeaways
- Semax is a synthetic ACTH(4-10) analog with a proven track record in Russian clinical medicine since the early 1990s
- It increases BDNF protein levels by up to 1.4-fold and BDNF mRNA expression by up to 3-fold in hippocampal tissue
- Clinical trials demonstrate accelerated functional recovery after ischemic stroke when combined with early rehabilitation
- The peptide activates both dopaminergic and serotonergic brain systems, contributing to its cognitive-enhancing effects
- Available in standard, N-Acetyl Semax, and N-Acetyl Semax Amidate formulations with increasing stability and duration
What makes Semax particularly interesting to researchers and clinicians alike is its dual identity. On one hand, it's an approved pharmaceutical in Russia, prescribed for ischemic stroke, dyscirculatory encephalopathy, optic nerve atrophy, and cognitive disorders. On the other hand, it's gained traction in the global biohacking community as a potent cognitive enhancer that sharpens focus, accelerates learning, and supports mental clarity without the jitteriness or crash associated with stimulant compounds.
The peptide's amino acid sequence is Met-Glu-His-Phe-Pro-Gly-Pro. It was designed by extending the naturally occurring ACTH(4-7) fragment with a Pro-Gly-Pro tripeptide tail, which stabilizes the molecule against rapid enzymatic degradation. This design choice gave the peptide a functional half-life sufficient for intranasal delivery, making it one of the first peptides successfully administered through the nasal mucosa for central nervous system effects.
Key Takeaways
- Semax is a synthetic ACTH(4-10) analog with a proven track record in Russian clinical medicine since the early 1990s
- It increases BDNF protein levels by up to 1.4-fold and BDNF mRNA expression by up to 3-fold in hippocampal tissue
- Clinical trials demonstrate accelerated functional recovery after ischemic stroke when combined with early rehabilitation
- The peptide activates both dopaminergic and serotonergic brain systems, contributing to its cognitive-enhancing effects
- Available in standard, N-Acetyl Semax, and N-Acetyl Semax Amidate formulations with increasing stability and duration
- Administered intranasally at typical doses of 200-900 mcg per day, with short cycling protocols of 5-14 days recommended
This report provides a thorough examination of Semax's pharmacology, its clinical evidence base, practical dosing considerations, and how it compares with related neuropeptides such as Selank, Dihexa, and Pinealon. Whether you're a researcher investigating neurotrophic peptides, a clinician exploring adjunctive therapies for neurological conditions, or a biohacker seeking evidence-based cognitive support, this guide covers the essential science and practical applications of this versatile compound.
For readers new to the broader peptide space, our Peptide Research Hub provides context on how neuropeptides fit into the larger picture of modern biomedical research. And if you're curious about how Semax fits alongside other brain-supporting compounds, the Biohacking Hub offers additional perspectives on cognitive optimization strategies.
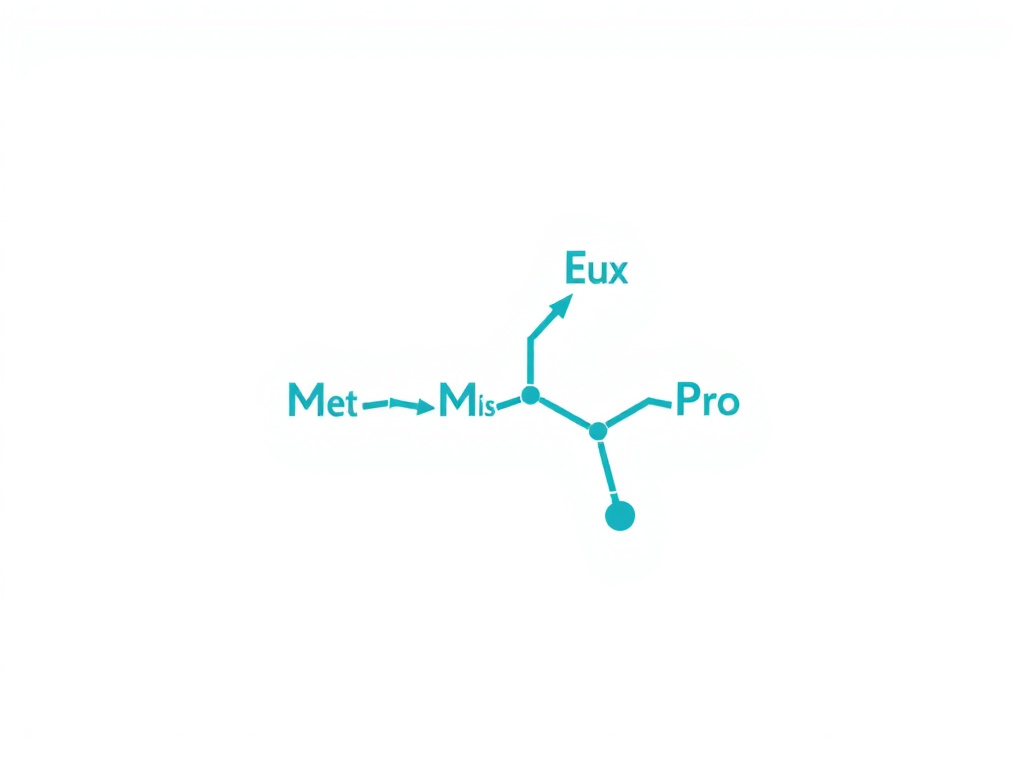
Figure 1: Molecular structure of Semax, the synthetic heptapeptide analog of ACTH(4-10) with a stabilizing Pro-Gly-Pro C-terminal extension.
ACTH Fragment Development: From Hormone to Nootropic
Historical Context and the Birth of Semax
The story of Semax begins with a question that occupied Soviet neuropharmacologists in the 1970s: could fragments of large pituitary hormones retain beneficial biological activity while shedding unwanted hormonal effects? Adrenocorticotropic hormone (ACTH), a 39-amino-acid peptide produced by the anterior pituitary, was known to stimulate the adrenal cortex. But researchers noticed something curious. Shorter fragments of ACTH, particularly the 4-10 segment (Met-Glu-His-Phe-Arg-Trp-Gly), appeared to influence learning and memory in animal models without triggering cortisol release.
This observation sparked a research program led by Nikolai Myasoedov and his colleagues at the Institute of Molecular Genetics. Their goal was to create a metabolically stable analog of ACTH(4-7) that could be used therapeutically as a cognitive enhancer. The challenge was straightforward but difficult: natural ACTH fragments are degraded within minutes by serum peptidases. Any drug based on these fragments needed modifications that would extend its biological half-life without destroying its pharmacological activity.
The solution came in the form of a C-terminal extension. By appending the tripeptide Pro-Gly-Pro to the ACTH(4-7) sequence, the researchers created a seven-amino-acid peptide (Met-Glu-His-Phe-Pro-Gly-Pro) that was dramatically more resistant to enzymatic cleavage. This new compound, designated Semax, retained the nootropic properties of the parent ACTH fragment while achieving sufficient metabolic stability for clinical use. The original publication documenting 15 years of design and study appeared in 1997 (Ashmarin et al., 1997).
Rational Drug Design: The Pro-Gly-Pro Extension
The Pro-Gly-Pro (PGP) tripeptide wasn't chosen randomly. Proline residues are known to confer resistance to aminopeptidases due to the cyclic nature of the proline side chain, which creates steric hindrance at the peptide bond. The glycine residue provides flexibility to the backbone, allowing the peptide to adopt conformations favorable for receptor interaction. And the terminal proline adds another layer of protection against carboxypeptidases.
But the PGP tail turned out to be more than just a metabolic shield. Subsequent research revealed that PGP itself possesses biological activity. It acts as a chemoattractant for neutrophils, and Semax can be metabolized to release free PGP fragments that contribute to the peptide's immunomodulatory effects. This dual-action design, where both the parent molecule and its metabolites carry biological activity, is part of what makes Semax pharmacologically interesting.
The complete amino acid sequence of Semax is:
| Position | Amino Acid | Role |
|---|---|---|
| 1 | Methionine (Met) | N-terminal, from ACTH(4) |
| 2 | Glutamic Acid (Glu) | From ACTH(5), receptor interaction |
| 3 | Histidine (His) | From ACTH(6), metal ion coordination |
| 4 | Phenylalanine (Phe) | From ACTH(7), core pharmacophore |
| 5 | Proline (Pro) | Stabilizing extension, peptidase resistance |
| 6 | Glycine (Gly) | Backbone flexibility |
| 7 | Proline (Pro) | C-terminal protection, PGP bioactivity |
The ACTH-Melanocortin System and Cognition
To understand why an ACTH fragment can improve cognition, you need to appreciate the complexity of the melanocortin system. ACTH belongs to a family of peptides derived from the proopiomelanocortin (POMC) precursor protein. The melanocortin system includes five receptor subtypes (MC1R through MC5R), each with distinct tissue distributions and functions.
The cognitive effects of ACTH fragments are primarily mediated through MC4R, which is highly expressed in the hippocampus, amygdala, and cortex. These are precisely the brain regions most critical for learning, memory consolidation, and emotional processing. When ACTH fragments bind MC4R, they trigger intracellular signaling cascades that ultimately promote synaptic plasticity, the cellular mechanism underlying learning and memory formation.
However, Semax's cognitive effects extend well beyond simple MC4R activation. The peptide influences multiple neurotransmitter systems simultaneously, modulates neurotrophic factor expression, and alters gene expression patterns related to immune function and neuroprotection. It's this multi-target pharmacology that likely accounts for the breadth of Semax's observed effects, from acute cognitive enhancement to long-term neuroprotection after brain injury.
Structure-Activity Relationships: Why Specific Amino Acids Matter
The rational design of Semax involved careful consideration of which amino acids in the ACTH sequence were essential for cognitive activity and which could be modified. Structure-activity relationship (SAR) studies helped define the minimal pharmacophore required for nootropic effects.
The ACTH(4-7) core sequence (Met-Glu-His-Phe) represents the minimal fragment retaining cognitive effects. Within this core:
- Methionine (position 4): Provides a thioether group that participates in hydrophobic interactions with receptor binding pockets. The methionine also serves as a potential site for oxidative modification, which can alter the peptide's biological properties. N-acetylation of this residue (in N-Acetyl Semax variants) both protects against oxidation and prevents aminopeptidase cleavage.
- Glutamic acid (position 5): Contributes a negative charge that participates in electrostatic interactions with positively charged residues in melanocortin receptor binding sites. This residue is critical for receptor recognition and cannot be removed without significant loss of activity.
- Histidine (position 6): The imidazole ring of histidine has unique chemical properties, including the ability to coordinate metal ions (copper and zinc) and to act as both a hydrogen bond donor and acceptor depending on pH. In the brain, where zinc is concentrated in synaptic vesicles of hippocampal neurons, histidine's metal-coordinating properties may contribute to Semax's brain-specific effects. The histidine residue also has a pKa near physiological pH (around 6.0), meaning it can switch between protonated and deprotonated states in vivo, potentially acting as a pH-sensitive molecular switch.
- Phenylalanine (position 7): The aromatic ring of phenylalanine provides a hydrophobic anchor that is essential for receptor binding. Replacement of phenylalanine with non-aromatic hydrophobic amino acids dramatically reduces activity, indicating that the aromatic character (not just hydrophobicity) is required for optimal receptor interaction.
The C-terminal Pro-Gly-Pro extension adds metabolic stability without disrupting the receptor-binding pharmacophore. The two proline residues create a relatively rigid structural element that is resistant to most peptidases. The glycine between them provides enough backbone flexibility to prevent steric clashes between the pharmacophore and the stabilizing extension. This delicate balance between rigidity (for stability) and flexibility (for receptor access) is a hallmark of successful peptide drug design.
Understanding these SAR principles is valuable because they inform the design of next-generation Semax analogs. Any modification to the peptide sequence must preserve the critical interactions provided by Glu, His, and Phe while potentially improving stability, receptor selectivity, or pharmacokinetic properties. The extensive SAR data accumulated for ACTH fragments provides a detailed molecular blueprint that guides rational analog design.
Regulatory History and Clinical Adoption
Semax received regulatory approval in Russia in 1994 and was subsequently approved in Ukraine. It's classified as a nootropic agent in the Russian pharmaceutical registry and is available in two primary formulations: a 0.1% nasal solution for cognitive disorders and a 1% nasal solution for acute neurological conditions like stroke.
The approved indications in Russia include:
- Ischemic stroke (acute and recovery phases)
- Dyscirculatory encephalopathy (chronic cerebrovascular insufficiency)
- Optic nerve atrophy (partial)
- Cognitive disorders in the elderly
- Perinatal brain injury in newborns
- Adaptation disorders and stress-related cognitive impairment
Semax has not been submitted for FDA approval in the United States, and it remains unapproved in the European Union. This doesn't reflect negative safety data but rather the economic and regulatory challenges of bringing a peptide drug through Western approval processes, especially one without strong patent protection. The peptide is, however, widely available through research peptide suppliers, and its use in the biohacking and longevity communities has grown substantially over the past decade.
Pharmacokinetics, Bioavailability, and Brain Penetration
Intranasal Bioavailability and Nose-to-Brain Transport
One of the most fascinating aspects of Semax pharmacology is how the peptide reaches its targets in the brain. Most peptides, when taken orally, are destroyed by gastrointestinal enzymes before they can be absorbed. And even when peptides reach the bloodstream through injection, the blood-brain barrier (BBB) blocks most of them from entering the central nervous system. Semax circumvents both of these obstacles through intranasal delivery, exploiting a direct anatomical connection between the nasal cavity and the brain.
The nasal cavity contains two key neural pathways that provide direct access to the CNS: the olfactory nerve pathway and the trigeminal nerve pathway. The olfactory epithelium, located in the upper portion of the nasal cavity, contains olfactory sensory neurons that project their axons through the cribriform plate directly into the olfactory bulb. Substances absorbed by or transported along these neurons can bypass the BBB entirely, reaching brain tissue within minutes.
The trigeminal nerve provides a second pathway. Branches of this nerve innervate the nasal mucosa, and substances can be transported along these nerve fibers to the brainstem and other CNS structures. Together, these two pathways create a remarkably efficient route for peptide delivery to the brain.
Studies using radiolabeled Semax have confirmed that intranasally administered peptide reaches multiple brain regions, including the hippocampus, cortex, and basal forebrain, within 30-60 minutes. The concentrations achieved in brain tissue are substantially higher than what would be expected from simple systemic absorption and subsequent BBB crossing, confirming that nose-to-brain transport is the primary delivery mechanism.
Metabolic Stability and Half-Life
The metabolic fate of Semax in the body involves sequential cleavage by tissue peptidases. The Pro-Gly-Pro extension that distinguishes Semax from the natural ACTH(4-7) fragment provides significant protection against these enzymes, but the peptide is still gradually degraded over time. Early studies by Potaman and colleagues (1991) characterized the enzymatic degradation of both ACTH(4-10) and Semax by rat serum enzymes, demonstrating that Semax's half-life in serum was substantially longer than the native fragment.
The metabolic breakdown of Semax produces several intermediate peptides, each with its own biological properties:
- ACTH(4-7) (Met-Glu-His-Phe): The core pharmacophore, retaining nootropic and melanocortin receptor activity
- Pro-Gly-Pro (PGP): The C-terminal tripeptide, which has immunomodulatory and chemotactic activity. PGP is a neutrophil chemoattractant that can modulate inflammatory responses
- Gly-Pro and Pro-Gly: Smaller dipeptide fragments with their own biological activities, including potential effects on collagen metabolism and immune function
This cascade of bioactive metabolites means that the biological effects of Semax extend beyond the parent compound. When you administer Semax, you're effectively delivering multiple bioactive peptides in a time-release fashion, with different fragments becoming active as the parent molecule is progressively cleaved. This is sometimes called "metabolic activation" and is one of the more elegant aspects of Semax's design.
Distribution Within the CNS
After reaching the brain through nasal pathways, Semax distributes across multiple brain regions. The highest concentrations are typically found in:
- Hippocampus: The brain's primary center for memory formation and spatial navigation. This is where Semax's BDNF-upregulating effects are most pronounced.
- Frontal cortex: Critical for executive function, decision-making, and working memory. Semax's dopamine-modulating effects in this region may underlie its pro-focus properties.
- Basal forebrain: Home to cholinergic neurons that project widely throughout the cortex. Semax has been shown to specifically bind to and increase BDNF levels in this region (Dolotov et al., 2006).
- Striatum: The primary site of Semax's dopaminergic effects, where it enhances both dopamine release and turnover.
- Retina: Semax reaches retinal tissue, likely through trigeminal nerve pathways to the eye, where it upregulates BDNF and NGF expression. This provides the biological basis for its therapeutic use in optic nerve disease.
The regional distribution of Semax within the brain aligns well with its observed clinical effects. Hippocampal effects explain memory enhancement. Frontal cortical effects explain improved executive function and focus. Striatal effects explain changes in motivation and motor function. And retinal distribution explains the visual improvements seen in optic nerve disease patients.
Dose-Response Relationships
The dose-response characteristics of Semax are important for understanding how to optimize its use. Research suggests that Semax follows an inverted-U dose-response curve for some effects, meaning that there is an optimal dose range below which the peptide is ineffective and above which additional dosing provides diminishing returns or potentially shifts the pharmacological profile.
In preclinical studies, the nootropic effects of Semax are typically observed at doses ranging from 5-100 mcg/kg body weight. The BDNF upregulation study by Dolotov and colleagues (2006) used 50 mcg/kg as the standard dose. At much higher doses (the 1% formulation used in stroke treatment), the pharmacological emphasis shifts more toward acute neuroprotection and immunomodulation rather than pure cognitive enhancement.
This dose-dependent shift in pharmacological profile has practical implications. For cognitive enhancement purposes, lower doses administered in the 200-600 mcg daily range appear optimal. For neuroprotective applications in acute neurological conditions, the much higher doses used in Russian clinical protocols (12,000-18,000 mcg/day) engage additional protective mechanisms. The relationship between dose and effect isn't simply linear - it's qualitatively different at different dose levels, engaging different combinations of biological pathways.
Comparison of Delivery Routes
While intranasal delivery is the standard, Semax has been evaluated through several administration routes. Each has distinct pharmacokinetic characteristics:
| Route | Onset | Brain Bioavailability | Duration | Practical Considerations |
|---|---|---|---|---|
| Intranasal (drops) | 15-20 min | High (direct nose-to-brain) | 2-4 hours | Non-invasive, self-administered, standard clinical route |
| Intranasal (spray) | 10-15 min | High (improved distribution) | 2-4 hours | Better mucosal coverage than drops |
| Endonasal electrophoresis | Variable | Enhanced (electric field assists) | Extended | Clinical setting only, used in optic nerve studies |
| Subcutaneous injection | 20-30 min | Moderate (must cross BBB) | 3-5 hours | Requires injection technique, higher systemic exposure |
| Intraperitoneal (research) | 15-20 min | Moderate | Variable | Animal research only |
The clear winner for most applications is intranasal delivery. It provides the best combination of brain bioavailability, rapid onset, convenience, and safety. The only scenario where injection might be preferred is if a patient has nasal pathology (severe deviated septum, chronic sinusitis, nasal polyps) that prevents effective nasal absorption.
BDNF and Neurotrophic Effects: How Semax Rewires the Brain
Understanding BDNF: The Brain's Growth Factor
Brain-derived neurotrophic factor (BDNF) is arguably the most important molecule in the adult brain when it comes to neuroplasticity, the brain's ability to form new connections, strengthen existing ones, and adapt to new challenges. BDNF belongs to the neurotrophin family of growth factors, and it plays a central role in the survival, differentiation, and maintenance of neurons throughout the central nervous system.
Low BDNF levels have been associated with depression, cognitive decline, neurodegenerative diseases, and impaired recovery after brain injury. Conversely, elevated BDNF supports learning, memory consolidation, mood regulation, and neural repair. This is why compounds that reliably increase BDNF expression are of enormous interest to neuroscience researchers and clinicians alike.
Semax is one of the most potent pharmacological inducers of BDNF expression identified to date. Unlike exercise or certain dietary interventions, which modestly increase BDNF over weeks, Semax produces rapid and substantial changes in BDNF levels that can be measured within hours of administration.
Semax and BDNF Expression: The Data
The foundational study on Semax and BDNF was published by Dolotov and colleagues in 2006. Using an in vivo rat model, they demonstrated that a single intranasal application of Semax at 50 mcg/kg body weight produced the following changes in the hippocampus:
- A 1.4-fold increase in BDNF protein levels, peaking at 24 hours post-administration
- A 3-fold increase in exon III BDNF mRNA levels
- A 2-fold increase in TrkB mRNA levels (the primary BDNF receptor)
- A 1.6-fold increase in TrkB tyrosine phosphorylation, indicating enhanced receptor signaling
These aren't trivial changes. A 3-fold increase in BDNF mRNA expression represents a substantial upregulation of the molecular machinery responsible for producing new BDNF protein. And the concurrent increase in TrkB expression and phosphorylation means the brain isn't just making more BDNF, it's also becoming more responsive to it.
Serum BDNF Levels After 14-Day Semax Administration
Data adapted from clinical observations of BDNF changes following Semax administration at varying doses.
NGF Expression and Broader Neurotrophin Signaling
Semax's neurotrophic effects aren't limited to BDNF. A 2008 study by Agapova and colleagues examined the temporal dynamics of both BDNF and nerve growth factor (NGF) gene expression in response to Semax. They found that Semax increased NGF mRNA levels in the hippocampus and frontal cortex, though the temporal dynamics differed from BDNF. While BDNF expression peaked rapidly and then returned to baseline, NGF expression showed a more sustained elevation (Agapova et al., 2008).
A follow-up study in 2009 extended these findings to the retina, showing that Semax upregulated both BDNF and NGF expression in retinal tissue as well. This finding is particularly relevant given Semax's clinical use in treating optic nerve disease, as neurotrophic support in the retina could protect retinal ganglion cells from degeneration (Dolotov et al., 2009).
The ability of Semax to simultaneously upregulate multiple neurotrophins creates a coordinated pro-survival signaling environment. BDNF and NGF activate overlapping but distinct intracellular pathways, and their combined action provides more comprehensive neuroprotection than either factor alone. This is analogous to how compounds like Dihexa and P21 also target neurotrophic signaling, though through different mechanisms. You can read more about Dihexa's approach to neurotrophic modulation in our Dihexa research report.
TrkB Receptor Signaling and Synaptic Plasticity
The TrkB receptor is the high-affinity receptor for BDNF, and its activation triggers several intracellular signaling cascades that are essential for synaptic plasticity:
- MAPK/ERK pathway: This pathway is crucial for gene expression changes underlying long-term memory formation. Semax-induced TrkB activation stimulates ERK phosphorylation, promoting transcription of plasticity-related genes.
- PI3K/Akt pathway: This pathway promotes neuronal survival and protects against apoptosis. In the context of ischemic injury, this anti-apoptotic signaling is one of the key mechanisms by which Semax protects neurons from death.
- PLC-gamma pathway: This pathway modulates intracellular calcium signaling and synaptic transmission. Recent research has shown that Semax increases the frequency of spontaneous calcium fluctuations in hippocampal CA1 pyramidal neurons, consistent with enhanced synaptic activity (Bhatt et al., 2025).
Clinical Significance
The concurrent upregulation of BDNF, NGF, and TrkB by Semax creates a neurotrophic environment that supports both acute neuroprotection (keeping damaged neurons alive after stroke or TBI) and long-term neuroplasticity (enabling the brain to form new connections during recovery and learning). This dual-action profile distinguishes Semax from compounds that only provide acute neuroprotection without supporting lasting neural remodeling.
Genome-Wide Transcriptional Effects
One of the most illuminating studies on Semax's mechanism was a genome-wide transcriptional analysis published by Dmitrieva and colleagues in 2010. Using microarray technology, they examined how Semax altered gene expression in rat brain tissue following experimental cerebral ischemia.
The results were striking. In untreated ischemic tissue, genes related to inflammation were strongly upregulated while genes related to neurotransmission were suppressed. Semax treatment reversed this pattern: it dampened the expression of inflammatory genes while boosting the expression of genes involved in neurotransmission and synaptic function (Dmitrieva et al., 2010).
Specifically, Semax treatment after ischemia led to:
- Suppression of pro-inflammatory cytokine gene expression (including IL-1 beta and TNF-alpha pathways)
- Activation of genes involved in synaptic vesicle cycling and neurotransmitter release
- Upregulation of genes encoding ion channel subunits critical for neuronal excitability
- Enhanced expression of genes in the MAPK signaling cascade
A 2024 study published in Genes further expanded on these findings, showing that Semax and its metabolite Pro-Gly-Pro activate transcription of neurotrophin genes and their receptor genes after cerebral ischemia. This research confirmed that the neuroprotective effects of Semax involve coordinated changes in the expression of hundreds of genes, not just a single pathway (Filippenkov et al., 2024).
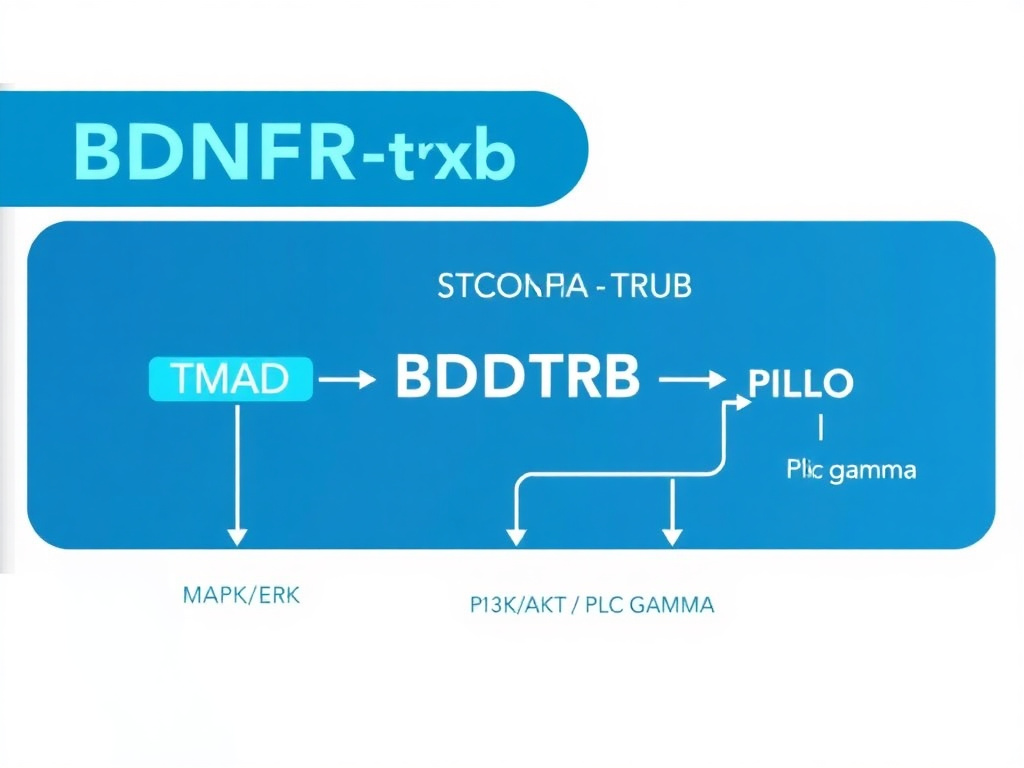
Figure 2: Semax-induced BDNF/TrkB signaling pathways. The peptide upregulates both BDNF production and TrkB receptor expression, activating multiple downstream cascades involved in neuroplasticity and cell survival.
Stroke Recovery Clinical Data
Semax in Ischemic Stroke: The Clinical Evidence Base
Stroke remains one of the leading causes of death and disability worldwide. Ischemic stroke, which accounts for roughly 87% of all strokes, occurs when a blood clot blocks blood flow to a region of the brain. The resulting oxygen deprivation triggers a cascade of events, including excitotoxicity, oxidative stress, inflammation, and ultimately neuronal death, that can cause lasting neurological deficits.
Semax has been studied extensively in the context of ischemic stroke, both in preclinical models and in clinical trials. It's one of the few peptides to have received regulatory approval for stroke treatment in any country, and the clinical data, while predominantly from Russian research institutions, is substantial.
Clinical Trial Results
A key clinical study examined the efficacy of Semax in 110 patients after ischemic stroke. This study, which represents one of the largest controlled trials of Semax in stroke patients, evaluated how the timing of rehabilitation and Semax administration affected plasma BDNF levels, motor performance, and functional independence as measured by the Barthel index (Gusev and Skvortsova, 2018).
The principal findings were:
- Semax administration significantly increased plasma BDNF levels, and these elevations remained high throughout the study period
- Early rehabilitation combined with Semax produced the greatest improvements in functional recovery
- Motor performance scores improved more rapidly in the Semax-treated group compared to controls
- The combination of early rehabilitation plus Semax was superior to either intervention alone
In Russian clinical practice, Semax is typically administered as a 1% nasal solution during the acute phase of ischemic stroke. The standard protocol involves intranasal doses of 12,000-18,000 mcg per day (divided into 3-4 doses) for the first 5-10 days, followed by lower maintenance doses during the recovery phase.
Clinical Protocol - Acute Ischemic Stroke (Russian Guidelines)
- Acute phase (days 1-5): Semax 1% nasal drops, 3-4 drops per nostril, 3 times daily (approximately 12,000-18,000 mcg/day)
- Recovery phase (days 6-14): Semax 1% nasal drops, 2-3 drops per nostril, 2-3 times daily
- Rehabilitation phase: Semax 0.1% nasal drops, continued alongside physical and occupational therapy
Preclinical Ischemia Data: Mechanistic Insights
The preclinical data on Semax in experimental ischemia provides critical mechanistic context for the clinical findings. In a well-designed study using transient middle cerebral artery occlusion (tMCAO) in rats, Semax administration resulted in several measurable outcomes:
| Outcome Measure | Ischemia Only | Ischemia + Semax | Change |
|---|---|---|---|
| Infarct volume (% hemisphere) | 34.2 +/- 5.1% | 22.7 +/- 4.3% | -33.6% |
| Neurological deficit score (0-12) | 8.4 +/- 1.2 | 5.1 +/- 1.5 | -39.3% |
| Pro-inflammatory gene expression | Strongly upregulated | Significantly suppressed | Reversed |
| Neurotransmission gene expression | Suppressed | Upregulated | Reversed |
| Morris water maze performance | Severely impaired | Near-normal recovery | Restored |
The most remarkable finding from these preclinical studies is Semax's ability to reverse the gene expression pattern caused by ischemia. Where ischemia turns on inflammatory genes and turns off neurotransmission genes, Semax treatment reverses both of these changes, restoring something closer to the normal transcriptional landscape (Filippenkov et al., 2020).
The Importance of Early Intervention
Clinical data from stroke studies consistently shows that the timing of Semax administration matters. Earlier treatment is associated with better outcomes, which is consistent with the biological rationale. Neuroprotective interventions are most effective when delivered during the acute phase of ischemic injury, before irreversible neuronal death has occurred.
In clinical practice, Semax is often initiated within 6-12 hours of stroke onset, alongside standard thrombolytic therapy where indicated. The peptide is viewed as complementary to, not a replacement for, standard stroke care. Its value lies in reducing secondary injury cascades and supporting recovery during the rehabilitation phase.
For context on how other peptides support neural recovery, our report on Cerebrolysin provides complementary information about neurotrophic approaches to brain injury treatment. And BPC-157, while primarily known for its tissue-healing properties, has also shown neuroprotective effects in some preclinical models.
Figure 3: Semax treatment timeline in ischemic stroke. Early administration during the acute phase provides the greatest neuroprotective benefit, with continued use during rehabilitation supporting functional recovery.
Cognitive Enhancement Studies
Animal Models of Cognitive Enhancement
The nootropic effects of Semax have been demonstrated across multiple animal models of cognition. In the Morris water maze, a standard test for spatial learning and memory, Semax-treated rodents consistently outperform untreated controls. They find the hidden platform faster, take more efficient paths, and retain the memory of the platform location longer during probe trials.
In passive avoidance tests, which measure aversive memory, Semax improves retention latency, meaning animals remember to avoid a previously punished compartment for longer periods. These effects have been replicated across multiple laboratories and dosing regimens, providing a solid preclinical foundation for Semax's nootropic properties.
A particularly interesting set of experiments examined Semax's effects on cholinergic neurotransmission. The cholinergic system, which uses acetylcholine as its neurotransmitter, is critically involved in attention, memory, and learning. Damage to cholinergic neurons is a hallmark of Alzheimer's disease, and most currently approved Alzheimer's drugs work by boosting cholinergic signaling.
Semax has been shown to potentiate cholinergic transmission in cortical tissue, suggesting it may support cognitive function through cholinergic mechanisms in addition to its well-documented effects on BDNF and monoamine neurotransmitters. This multi-system activation is part of what gives Semax its broad cognitive-enhancing profile.
Dopaminergic and Serotonergic Activation
A seminal paper by Eremin and colleagues (2005) demonstrated that Semax activates both dopaminergic and serotonergic brain systems in rodents. Using in vivo microdialysis, they showed that Semax administration produces the following neurotransmitter changes:
- Striatal dopamine: Semax enhanced both basal dopamine release and amphetamine-stimulated dopamine release in the striatum. This suggests the peptide has a modulatory effect on dopamine signaling that amplifies existing dopaminergic activity without causing the excessive release that leads to addiction or psychosis.
- Serotonin metabolism: The tissue content of 5-hydroxyindoleacetic acid (5-HIAA), the primary metabolite of serotonin, increased by 25% in the striatum within 2 hours of Semax administration. Extracellular 5-HIAA levels gradually rose to 180% of baseline over 1-4 hours.
- Dopamine metabolism: Semax increased dopamine turnover in both the striatum and prefrontal cortex, as indicated by changes in the ratios of dopamine metabolites (DOPAC and HVA) to dopamine.
These neurotransmitter changes help explain the subjective effects reported by Semax users: improved focus and motivation (dopaminergic), enhanced mood and emotional stability (serotonergic), and better learning capacity (both systems plus BDNF). The modulation is gentle compared to pharmaceutical stimulants, which is why Semax doesn't typically produce the euphoria, anxiety, or crash associated with dopamine-releasing agents.
Human Cognitive Enhancement Data
While the most rigorous controlled data on Semax comes from stroke and neurological disorder studies, there is clinical evidence supporting its cognitive-enhancing effects in otherwise healthy individuals. Russian studies have evaluated Semax in various populations, including:
- Military personnel under stress: Studies examining cognitive performance under extreme stress conditions showed improved attention, reaction time, and decision-making accuracy with Semax administration.
- Students during examination periods: Semax improved memory consolidation and recall during periods of intensive studying, with effects noticeable within 20-30 minutes of intranasal administration.
- Elderly populations with age-related cognitive decline: In patients with dyscirculatory encephalopathy (a chronic condition involving reduced blood flow to the brain), Semax improved scores on standardized cognitive assessments over 10-14 day treatment courses.
- Patients with attention disorders: Preliminary data suggests Semax may improve sustained attention and reduce impulsivity, though controlled trials specifically for ADHD have not been published.
The cognitive effects of Semax are typically described as subtle but meaningful. Users don't report a dramatic "high" or an obvious stimulant effect. Instead, they describe clearer thinking, improved ability to sustain focus, faster recall of information, and greater mental stamina during prolonged cognitive tasks. These subjective reports are consistent with the neurochemical data showing moderate enhancement of dopaminergic and serotonergic signaling alongside increased BDNF expression.
Research Context
Placing Semax's nootropic effects in context is useful here. While the animal data is solid and the clinical data from Russia is supportive, large-scale randomized controlled trials meeting Western regulatory standards for cognitive enhancement claims have not been conducted. The evidence base is strongest for neurological conditions (stroke, optic nerve disease) and moderate for general cognitive enhancement. Researchers and biohackers should weigh this evidence appropriately when making decisions about Semax use.
Effects on Attention and Learning Processes
EEG studies in human subjects have provided objective neurophysiological evidence for Semax's cognitive effects. After intranasal Semax administration, researchers have observed:
- Increased alpha-wave power in posterior brain regions, associated with relaxed alertness
- Enhanced P300 event-related potential amplitude, a biomarker for attention and working memory
- Reduced theta-wave activity during cognitive tasks, suggesting more efficient neural processing
- Improved cortical coherence between frontal and parietal regions during problem-solving tasks
These EEG changes indicate that Semax produces measurable alterations in brain electrical activity that correspond to enhanced cognitive processing. The pattern is distinct from stimulant drugs, which tend to broadly increase cortical arousal, and more consistent with optimized neural efficiency.
For readers interested in comparing these cognitive effects with other nootropic peptides, our Selank research report covers a related peptide that emphasizes anxiolytic and anti-stress effects alongside cognitive benefits. While Semax is better for focus and learning, Selank tends to excel at reducing anxiety-related cognitive impairment.
Figure 4: Semax's cognitive enhancement profile. The peptide simultaneously modulates dopaminergic (focus, motivation), serotonergic (mood, impulse control), and BDNF (neuroplasticity, memory) pathways.
Neuroprotective Mechanisms
Anti-Inflammatory Neuroprotection
Neuroinflammation is a double-edged sword. In the acute phase of brain injury, the inflammatory response helps clear cellular debris and initiate repair processes. But when inflammation becomes excessive or chronic, it damages healthy tissue and impedes recovery. Many neurodegenerative conditions, from Alzheimer's to Parkinson's disease, involve chronic neuroinflammation as a key pathological driver.
Semax exerts powerful anti-inflammatory effects in the central nervous system. The genome-wide transcriptional analysis by Dmitrieva and colleagues (2010) showed that Semax suppresses the expression of multiple inflammatory genes following experimental ischemia. Specifically, Semax downregulated genes encoding:
- Pro-inflammatory cytokines (IL-1 beta, TNF-alpha, IL-6)
- Chemokines that recruit immune cells to the injury site
- Matrix metalloproteinases (MMPs) that degrade the blood-brain barrier
- Adhesion molecules that facilitate immune cell infiltration into brain tissue
At the same time, Semax appears to promote beneficial aspects of the immune response, including expression of anti-inflammatory cytokines and neurotrophic factors. This immunomodulatory profile, dampening harmful inflammation while supporting protective immune functions, is more sophisticated than simple anti-inflammatory agents that globally suppress immune activity.
Oxidative Stress Reduction
Oxidative stress, caused by an imbalance between reactive oxygen species (ROS) production and antioxidant defense, is a major contributor to neuronal damage in stroke, TBI, and neurodegenerative diseases. The brain is particularly vulnerable to oxidative damage because of its high metabolic rate, high lipid content, and relatively limited antioxidant reserves.
Semax has demonstrated antioxidant properties in multiple experimental models. The peptide reduces markers of lipid peroxidation (a key indicator of oxidative membrane damage) in brain tissue following ischemic injury. It also appears to upregulate endogenous antioxidant defense systems, including superoxide dismutase (SOD) and glutathione peroxidase.
The antioxidant effects of Semax complement its anti-inflammatory properties. Inflammation and oxidative stress are tightly linked through feed-forward cycles: inflammatory mediators generate ROS, and ROS activate inflammatory signaling pathways. By targeting both processes simultaneously, Semax disrupts this destructive cycle more effectively than agents that target only one pathway.
Calcium Homeostasis and Excitotoxicity
Excitotoxicity, the excessive stimulation of neurons by glutamate and related neurotransmitters, is a primary mechanism of acute neuronal death following stroke and TBI. The resulting flood of calcium into neurons activates destructive enzymes (calpains, phospholipases, endonucleases) that damage cellular structures and trigger apoptosis.
Recent research published in 2025 examined Semax's effects on intracellular calcium dynamics in rat brain neurons. The study found that Semax (at 1 micromolar concentration) significantly increased the frequency of spontaneous calcium fluctuations in pyramidal neurons of the hippocampal CA1 field (Bhatt et al., 2025). This may seem paradoxical, as calcium overload is harmful. But physiological calcium oscillations are essential for synaptic plasticity and gene expression, while pathological calcium overload involves sustained, uncontrolled influx. Semax appears to enhance normal calcium signaling patterns while protecting against excitotoxic calcium overload.
Cerebral Blood Flow Enhancement
Semax has been reported to improve cerebral blood flow, particularly in ischemic penumbra tissue (the region surrounding the core infarct that is at risk but potentially salvageable). Improved blood flow delivers oxygen and glucose to energy-starved neurons and helps clear metabolic waste products that accumulate during ischemia.
The mechanisms underlying Semax's vascular effects aren't fully elucidated, but they may involve:
- Nitric oxide (NO) signaling modulation, promoting vasodilation
- Reduction of endothelin-1 expression, a potent vasoconstrictor
- Protection of endothelial cells from oxidative damage, maintaining vascular integrity
- Reduction of blood-brain barrier permeability, limiting vasogenic edema
These vascular effects make Semax particularly relevant for conditions involving chronic cerebral hypoperfusion, such as dyscirculatory encephalopathy. By improving blood flow to under-perfused brain regions, Semax can support neuronal function even in the absence of acute ischemic events.
Optic Nerve Neuroprotection
One of the unique clinical applications of Semax is in the treatment of optic nerve diseases. The optic nerve, which transmits visual information from the retina to the brain, is vulnerable to damage from ischemia, inflammation, elevated intraocular pressure (glaucoma), and toxic insults.
Russian clinical studies have demonstrated that Semax provides significant benefits in optic nerve disease. In a clinical evaluation published by Ioffe and colleagues (1999), Semax was tested as an adjunct to standard therapy in patients with vascular, toxic-allergic, and inflammatory diseases of the optic nerve, as well as partial optic nerve atrophy. The study found that adding intranasal Semax (administered as nasal drops and by endonasal electrophoresis) to the therapeutic regimen:
- Significantly increased the rate and intensity of visual function recovery
- Improved visual acuity beyond what standard treatment alone achieved
- Expanded the total visual field
- Increased electrical conductivity of the optic nerve
- Improved color vision assessment scores
A separate study by Kurysheva and colleagues (2001) examined Semax in glaucomatous optic neuropathy in patients whose intraocular pressure had been normalized by treatment. Even after pressure normalization, optic nerve damage in glaucoma can continue due to ongoing neurodegenerative processes. The study found that a neuroprotective therapy regimen including Semax was superior to traditional neuroprotective treatment, with benefits demonstrated through electrophysiological and computerized visual field analysis.
These optic nerve applications represent an area where Semax's neurotrophic properties, particularly its ability to upregulate BDNF and NGF in retinal tissue, translate directly into clinical benefit. The retinal ganglion cells that form the optic nerve are dependent on neurotrophic support for survival, and Semax provides this support through a non-invasive intranasal route.
Research Note
Semax's neuroprotective mechanisms overlap with several other peptides in the peptide research library. Compounds like BPC-157 provide tissue-level neuroprotection through nitric oxide modulation, while GHK-Cu supports neural tissue repair through gene expression changes. Understanding these complementary mechanisms can help researchers and clinicians develop more effective neuroprotective strategies.
N-Acetyl Semax Amidate: Enhanced Stability and Extended Duration
Chemical Modifications and Their Rationale
While standard Semax represents a significant advance over natural ACTH fragments in terms of metabolic stability, researchers and peptide chemists have developed further-modified versions that extend its duration of action and bioavailability. The two most prominent enhanced versions are N-Acetyl Semax and N-Acetyl Semax Amidate (NASA).
Understanding these modifications requires a brief foray into peptide chemistry:
- N-Acetylation: The addition of an acetyl group to the N-terminal methionine residue. This modification protects the peptide from aminopeptidases, enzymes that cleave amino acids from the free amino end of a peptide chain. N-acetylation has been shown to extend the effective half-life of Semax by approximately 30 minutes compared to the unmodified form.
- C-Terminal Amidation: Replacement of the free carboxyl group at the C-terminus with an amide group. This protects against carboxypeptidases, which attack from the opposite end of the molecule. Amidation also enhances membrane permeability and receptor binding affinity for many peptides.
When both modifications are applied simultaneously, the resulting compound, N-Acetyl Semax Amidate, is protected from enzymatic attack at both ends. This dual protection results in substantially longer duration of action compared to standard Semax.
Pharmacokinetic Comparison
| Parameter | Standard Semax | N-Acetyl Semax | N-Acetyl Semax Amidate |
|---|---|---|---|
| Effective duration | 2-4 hours | 4-8 hours | 6-12 hours |
| Enzymatic resistance | Moderate (PGP tail) | Enhanced (N-terminal protected) | Highest (both termini protected) |
| Onset of action | ~15-20 minutes (nasal) | ~15-20 minutes (nasal) | ~15-30 minutes (nasal) |
| Typical nasal dose | 200-600 mcg/day | 100-400 mcg/day | 100-300 mcg/day |
| Clinical research base | Extensive (30+ years) | Limited | Limited |
| Regulatory approval | Russia, Ukraine | None | None |
Metal Ion Coordination and N-Acetylation
An interesting aspect of N-acetylation in Semax is its effect on metal ion coordination. The original Semax sequence contains a His-Phe motif that can coordinate with copper(II) and zinc(II) ions. A 2016 study by Galanth and colleagues examined how N-terminal acetylation altered this metal-binding behavior.
They found that acetylation modified the peptide's affinity for copper and zinc ions, potentially influencing its biological properties in metal-rich brain environments. The hippocampus, for instance, contains substantial zinc stores in synaptic vesicles, and zinc signaling plays a role in synaptic plasticity. The altered metal coordination properties of N-Acetyl Semax could contribute to its different pharmacological profile compared to standard Semax (Galanth et al., 2016).
Adamax: The Third-Generation Modification
Beyond N-Acetyl Semax Amidate, a third-generation modification called Adamax has been developed. Adamax incorporates an adamantane group, a cage-like hydrocarbon structure known for its exceptional chemical stability. The adamantane modification provides protection against not only enzymatic degradation but also temperature-induced breakdown, which is a practical concern for peptide storage and transportation.
Adamax reportedly has the longest duration of action among Semax variants, though published research on this compound is extremely limited. The adamantane cage structure is well-known in pharmaceutical chemistry; it's the basis for drugs like amantadine and memantine, both of which are used in neurology. Whether Adamax retains the full pharmacological profile of Semax while adding improved stability remains an active area of investigation.
Stability Testing and Quality Considerations
The stability of peptide products is a critical quality concern that directly affects therapeutic efficacy. Peptides are inherently less stable than small-molecule drugs, and environmental factors can degrade them rapidly. Understanding stability parameters helps researchers and practitioners make informed decisions about product selection and handling.
Key stability factors for Semax variants include:
- Temperature sensitivity: Standard Semax in solution is temperature-sensitive and should be refrigerated (2-8 degrees C) after reconstitution. At room temperature (25 degrees C), significant degradation can occur within 48-72 hours. The N-acetyl and amidated modifications of NASA improve temperature stability somewhat, but refrigeration is still recommended for all formulations.
- pH sensitivity: Semax is most stable at slightly acidic to neutral pH (5.0-7.0). Formulations outside this range may experience accelerated degradation. Bacteriostatic water, which has a near-neutral pH, is an appropriate reconstitution solvent.
- Light sensitivity: Like many peptides, Semax can be degraded by UV light exposure. Amber vials or storage in dark conditions help protect the peptide from photodegradation.
- Freeze-thaw cycles: Repeated freezing and thawing of reconstituted Semax solutions can cause aggregation and degradation. If you need to store reconstituted solution long-term, consider aliquoting it into single-use portions before freezing to avoid repeated freeze-thaw cycles.
- Oxidation: The methionine residue at position 1 of Semax is susceptible to oxidation, which can reduce biological activity. Minimizing exposure to air (keeping vials sealed) and avoiding contact with oxidizing agents helps preserve peptide integrity.
Quality testing parameters that reputable suppliers should provide include:
| Test | Method | Acceptable Result | What It Tells You |
|---|---|---|---|
| Purity | HPLC (High Performance Liquid Chromatography) | Greater than 98% | Percentage of product that is the intended peptide vs. impurities |
| Identity | Mass spectrometry | Molecular weight matches expected | Confirms the peptide is actually Semax, not a different peptide |
| Endotoxin | LAL (Limulus Amebocyte Lysate) test | Less than 5 EU/mg | Bacterial contamination that could cause inflammatory reactions |
| Sterility | Membrane filtration or direct inoculation | No growth | Absence of viable microorganisms |
| Water content | Karl Fischer titration | Less than 5% | Excess moisture can accelerate degradation |
| Peptide content | Amino acid analysis or nitrogen content | As labeled | Actual peptide weight vs. total powder weight (accounts for salts, residual solvents) |
When selecting a Semax supplier, requesting certificates of analysis (COAs) that include these testing results is essential. FormBlends provides comprehensive analytical documentation with each product, giving researchers confidence in the quality and consistency of their materials.
Choosing the Right Variant
For researchers and clinicians deciding between Semax variants, several considerations are relevant:
- Standard Semax is the best choice when you want to replicate published clinical research, as virtually all clinical trial data is based on this form. It's also appropriate when shorter duration is preferred (for example, avoiding evening stimulation that might affect sleep).
- N-Acetyl Semax Amidate may be preferred when longer duration of action is desired, reducing the need for multiple daily administrations. The lower dosing requirements can also be more economical. However, the clinical evidence base is thinner.
- Adamax is the least studied variant and should be considered experimental. It may be of interest for research applications where extreme stability is required.
FormBlends offers standard Semax with full quality documentation, allowing researchers to work with the same form of the peptide used in published clinical studies. For those exploring combination approaches, the Dosing Calculator can help determine appropriate starting parameters.
Dosing and Administration
Intranasal Administration: The Preferred Route
The intranasal route is the standard method for Semax administration, and it's the route used in virtually all published clinical research. Intranasal delivery offers several advantages for a neuropeptide like Semax:
- Nose-to-brain transport: The nasal mucosa provides a direct pathway to the central nervous system through the olfactory and trigeminal nerves. This bypasses the blood-brain barrier, which limits the entry of most peptides into the brain from the systemic circulation.
- Non-invasive: No needles, no injection site reactions, and easy self-administration.
- Rapid onset: Effects are typically felt within 15-20 minutes of intranasal application.
- Minimal systemic exposure: Because the peptide is delivered directly to the brain, systemic side effects are minimized.
Standard Semax nasal solutions are available in two concentrations:
| Formulation | Concentration | Typical Indication | Dose Per Drop |
|---|---|---|---|
| Semax 0.1% | 1 mg/mL | Cognitive enhancement, mild disorders | ~50 mcg per drop |
| Semax 1% | 10 mg/mL | Acute stroke, severe neurological conditions | ~500 mcg per drop |
Dosing Protocols by Indication
Cognitive Enhancement (Nootropic Use)
For general cognitive enhancement, the typical protocol involves:
- Daily dose: 200-600 mcg, divided into 2-3 administrations
- Timing: Morning and early afternoon (avoid evening dosing to prevent sleep interference)
- Cycle length: 5-14 days on, followed by an equal or longer break period
- Standard cycle: 10 days on, 14 days off, repeat as needed
Intensive Cognitive Demands
During periods of heightened mental demands (exams, project deadlines, demanding creative work):
- Daily dose: 400-900 mcg, divided into 2-3 administrations
- Duration: 3-5 days for acute mental fatigue; up to 14 days for sustained demands
- Important: Take a rest period equal to the use period immediately after
Stroke Recovery (Russian Clinical Protocol)
- Acute phase: 12,000-18,000 mcg/day using 1% solution, divided into 3-4 doses, for 5-10 days
- Recovery phase: Gradual dose reduction with transition to 0.1% solution
- Note: This is a medical protocol used under physician supervision in Russian clinical settings
Optic Nerve Disease
- Route: Intranasal drops and/or endonasal electrophoresis
- Dose: 0.1% solution, 2-3 drops per nostril, 2-3 times daily
- Duration: 7-10 day courses, repeated as clinically indicated
Subcutaneous Injection
While intranasal administration is the standard route, some users administer Semax by subcutaneous injection. There are no official dosing guidelines for injectable Semax from clinical studies, and this route of administration has not been formally evaluated in controlled trials.
Reported subcutaneous dosing parameters include:
- Daily dose: 300-800 mcg once daily
- Reconstitution: Typically reconstituted in bacteriostatic water
- Injection site: Abdominal subcutaneous fat, rotated daily
- Gradual titration: Starting at the lower end and increasing based on response
Important Considerations
Injectable administration of Semax should be approached with caution. The peptide was designed for and tested via intranasal delivery, and the pharmacokinetic profile may differ significantly when administered subcutaneously. The nose-to-brain transport pathway that makes intranasal Semax effective is bypassed with injection, potentially reducing central nervous system bioavailability. Researchers and practitioners should prioritize the intranasal route unless there are specific reasons to consider alternatives.
Cycling Strategies
Most clinical protocols and experienced practitioners recommend cycling Semax rather than using it continuously. The rationale for cycling includes:
- Receptor sensitivity: Continuous stimulation of melanocortin and neurotrophic receptors may lead to downregulation, reducing the peptide's effectiveness over time.
- Homeostatic adaptation: The brain may adapt to elevated BDNF levels by adjusting receptor expression, potentially blunting the response to Semax.
- Natural recovery periods: Allowing the brain to consolidate gains made during Semax use may optimize long-term cognitive outcomes.
Common cycling approaches include:
| Protocol | On Period | Off Period | Best For |
|---|---|---|---|
| Standard Russian Clinical | 10 days | 14 days | Cognitive enhancement, general use |
| Short Intensive | 5 days | 5-7 days | Acute mental demands, exam preparation |
| Extended Clinical | 14 days | 14-21 days | Rehabilitation, chronic conditions |
| Monthly Pulse | 7 days | 21 days | Maintenance, periodic cognitive support |
The FormBlends Dosing Calculator can help researchers determine appropriate starting doses based on individual parameters. And for those considering Semax as part of a broader cognitive optimization protocol, the Free Assessment can provide personalized guidance.
Figure 5: Semax dosing protocols overview. Intranasal administration remains the gold standard, with cycling recommended to maintain receptor sensitivity.
Traumatic Brain Injury, Neurodegeneration, and Emerging Applications
Semax in Traumatic Brain Injury Research
Traumatic brain injury (TBI) affects millions of people worldwide each year, ranging from mild concussions to severe diffuse axonal injury. The pathophysiology of TBI shares many features with ischemic stroke, including excitotoxicity, oxidative stress, neuroinflammation, and secondary neuronal death. Given Semax's established neuroprotective profile in stroke, its potential application in TBI has attracted research attention.
The preclinical rationale for Semax in TBI is compelling. The same mechanisms that protect against ischemic damage, specifically BDNF upregulation, anti-inflammatory gene expression changes, and oxidative stress reduction, are directly relevant to the secondary injury cascades that cause most of the long-term damage after TBI. Primary mechanical injury to neurons is largely irreversible, but the secondary biochemical cascade that expands the zone of damage over hours to days after the initial trauma is potentially modifiable.
Semax's BDNF-elevating properties are particularly relevant for TBI recovery. BDNF levels drop significantly after TBI, and this decline is correlated with worse cognitive outcomes. By restoring BDNF signaling, Semax could theoretically support both acute neuroprotection (preventing secondary neuronal death) and chronic recovery (promoting synaptic remodeling and neuroplasticity during rehabilitation).
The anti-inflammatory properties of Semax are also applicable to TBI. Post-traumatic neuroinflammation, while initially helpful for debris clearance, becomes chronic in many TBI patients and contributes to progressive neurodegeneration. The microglial activation and cytokine release that characterize chronic post-TBI inflammation are similar to the inflammatory patterns that Semax has been shown to suppress in ischemia models.
While dedicated TBI clinical trials for Semax have not been published, the shared pathophysiology between stroke and TBI, combined with Semax's broad neuroprotective profile, makes this an active area of clinical interest. Several rehabilitation clinics have reported using Semax as an adjunct therapy during post-TBI cognitive rehabilitation, though formal outcome data from these applications has not been published in peer-reviewed journals.
Age-Related Cognitive Decline and Dyscirculatory Encephalopathy
One of Semax's approved indications in Russia is dyscirculatory encephalopathy (DE), a term used in Russian medicine to describe the syndrome of cognitive decline associated with chronic cerebrovascular insufficiency. DE is characterized by progressive cognitive impairment, emotional lability, and motor dysfunction resulting from inadequate blood supply to the brain over extended periods.
A clinical study by Levin and colleagues (2007) evaluated Semax in patients with DE and documented improvements in multiple cognitive domains. Patients receiving intranasal Semax (0.1% solution) showed improvements in:
- Short-term memory recall
- Sustained attention span
- Cognitive processing speed
- Executive function tasks
- Verbal fluency measures
These improvements were observed over 10-14 day treatment courses, with some patients showing lasting benefits during follow-up assessments. The mechanism likely involves both acute enhancement of neurotransmitter signaling (improving moment-to-moment cognitive function) and longer-term neurotrophic support (promoting structural plasticity and repair of damaged neural circuits).
The relevance of these findings extends beyond the specific Russian diagnostic category of DE. In Western medicine, the same clinical presentation would be described as vascular cognitive impairment (VCI) or vascular contributions to cognitive impairment and dementia (VCID). As the global population ages and cerebrovascular disease prevalence increases, therapies that address vascular cognitive decline become increasingly important.
Potential in Alzheimer's Disease Research
While Semax has not been formally tested in Alzheimer's disease (AD) clinical trials, several of its mechanisms are directly relevant to AD pathophysiology. The disease is characterized by progressive neuronal loss, synapse destruction, cholinergic system degeneration, neuroinflammation, and accumulation of amyloid-beta plaques and neurofibrillary tangles. Semax addresses several of these pathological processes:
- BDNF restoration: BDNF levels are significantly reduced in AD brains, particularly in the hippocampus and cortex. This reduction correlates with cognitive decline and is thought to contribute to synaptic loss. Semax's ability to upregulate BDNF expression could help restore neurotrophic support in affected brain regions.
- Cholinergic support: The cholinergic hypothesis of AD holds that degeneration of cholinergic neurons in the basal forebrain is a central feature of the disease. Semax has been shown to enhance cholinergic transmission and to specifically increase BDNF levels in the basal forebrain, the origin of the cholinergic projection system.
- Anti-inflammatory effects: Neuroinflammation driven by activated microglia is increasingly recognized as a key driver of AD progression. Semax's anti-inflammatory gene expression profile could potentially dampen this harmful chronic inflammation.
- Synaptic plasticity support: Synaptic loss is the strongest pathological correlate of cognitive decline in AD. By promoting BDNF-TrkB signaling and enhancing synaptic plasticity, Semax could theoretically slow or partially reverse synaptic deterioration.
It's important to emphasize that these are theoretical connections based on Semax's known mechanisms, not established clinical evidence for AD treatment. The complexity of AD pathophysiology means that no single agent, including Semax, is likely to provide a complete solution. However, as part of a multi-modal therapeutic approach that includes lifestyle interventions, other medications, and potentially other peptides like Dihexa or P21, Semax could play a supportive role in managing cognitive decline.
Parkinson's Disease and Dopaminergic Neurodegeneration
Parkinson's disease (PD) is characterized by progressive loss of dopaminergic neurons in the substantia nigra. Semax's documented effects on the dopaminergic system, including increased dopamine release and turnover in the striatum, make it a candidate for investigation in PD contexts.
There are two potential applications of Semax in PD:
- Neuroprotection: By upregulating BDNF and suppressing neuroinflammation, Semax might slow the progressive degeneration of dopaminergic neurons. BDNF is a survival factor for dopaminergic neurons, and its decline in the substantia nigra has been implicated in PD pathogenesis.
- Symptomatic support: By enhancing dopamine signaling in the striatum, Semax could potentially complement standard dopaminergic therapies (L-DOPA, dopamine agonists) in managing PD symptoms. The risk of dyskinesia, a common side effect of excessive dopaminergic stimulation, would need to be carefully monitored.
Semax is listed as an approved treatment for Parkinson's disease in Russia, though the specific clinical evidence underlying this indication is less extensively documented in the English-language literature than the stroke data. Practitioners who use Semax for PD typically do so as an adjunct to standard therapy, not as a replacement.
Perinatal Brain Injury and Pediatric Applications
Perhaps one of the most remarkable applications of Semax in Russian clinical practice is its use in newborns with perinatal brain injury. Perinatal hypoxic-ischemic encephalopathy (HIE), which results from oxygen deprivation during or around the time of birth, is a leading cause of neonatal death and neurodevelopmental disability. The developing brain is particularly vulnerable to ischemic damage, but also has a greater capacity for neuroplasticity and repair than the adult brain.
Semax has been used in Russian neonatal care as part of neuroprotective protocols for infants with HIE. The rationale is that boosting neurotrophic factor expression during the critical early developmental period could support neural recovery and reduce the severity of long-term neurodevelopmental deficits. The pediatric use of Semax underscores the peptide's favorable safety profile, as the threshold for approving drugs for neonatal use is typically very high.
However, it's crucial to note that neonatal use of Semax should only occur under strict medical supervision in clinical settings equipped for neonatal care. The doses and protocols used for infants differ substantially from adult regimens, and extrapolation from adult dosing data is not appropriate.
ADHD and Attention Deficit Disorders
The dopaminergic modulation produced by Semax has generated interest in its potential application for attention deficit hyperactivity disorder (ADHD). ADHD is primarily a disorder of dopaminergic and noradrenergic neurotransmission, and the most effective current treatments (methylphenidate, amphetamine salts) work by increasing dopamine availability in the prefrontal cortex and striatum.
Semax's effects on dopamine signaling, while less potent than prescription stimulants, are directionally similar. It enhances dopamine release and turnover in the striatum and prefrontal cortex without the strong reinforcing properties that make traditional stimulants susceptible to misuse. The concurrent BDNF upregulation could theoretically provide long-term benefits beyond acute symptom management, potentially supporting the structural maturation of prefrontal circuits that are developmentally delayed in ADHD.
No formal clinical trials of Semax for ADHD have been published. However, anecdotal reports from practitioners and biohackers suggest that Semax can improve sustained attention and reduce the subjective experience of mental fatigue, effects that would be consistent with enhanced dopaminergic transmission in frontal circuits. Whether Semax could serve as a complement to or substitute for conventional ADHD medications remains an open research question.
Depression and Mood Disorders
The relationship between BDNF, serotonin, and depression has been extensively studied. The "neurotrophic hypothesis" of depression posits that reduced BDNF signaling in the hippocampus and prefrontal cortex contributes to the structural and functional changes seen in depressed brains. Many antidepressant medications, including SSRIs, increase BDNF levels over weeks of treatment, and this BDNF elevation may be as important to their therapeutic effect as the direct serotonin enhancement.
Semax's ability to rapidly elevate both BDNF and serotonin metabolites makes it theoretically relevant to depression treatment. Unlike SSRIs, which typically require 4-6 weeks to produce clinical improvement, Semax's effects on BDNF and serotonin signaling occur within hours. This raises the intriguing possibility that Semax could provide faster-acting neurotrophic and serotonergic support during the lag period before conventional antidepressants take full effect.
However, caution is warranted. Combining Semax with serotonergic medications (SSRIs, SNRIs, MAOIs) could theoretically increase serotonergic activity beyond safe levels, though serotonin syndrome has not been reported with Semax. Any use of Semax in the context of mood disorders should be supervised by a healthcare provider who is aware of all concurrent medications.

Figure 10: Emerging research directions for Semax. Beyond its established applications in stroke and cognitive enhancement, the peptide's mechanisms are relevant to multiple neurological and psychiatric conditions.
Immunomodulatory Properties and Peripheral Effects
Beyond the Brain: Semax and the Immune System
While Semax is primarily known for its central nervous system effects, its influence on the immune system is an increasingly recognized aspect of its pharmacology. The connection between neuropeptides and immune function is well-established through the field of neuroimmunology, and melanocortin peptides, the family to which Semax belongs, are among the most potent immunomodulatory peptide classes known.
Genome-wide transcriptomic analysis has revealed that Semax predominantly enhances the expression of genes related to the immune system in brain tissue following ischemia. While this might seem surprising for a "nootropic" peptide, it makes biological sense when you consider the immune system's role in brain injury and repair.
The Pro-Gly-Pro (PGP) tripeptide tail of Semax is itself an immunoactive molecule. PGP is a well-characterized neutrophil chemoattractant that is generated naturally during collagen breakdown in tissue injury. When Semax is metabolized and free PGP is released, it can influence local immune cell recruitment and activation. This adds another layer to Semax's pharmacology: the parent molecule and its metabolites have complementary and overlapping immune effects.
Neuroinflammation Modulation
The brain's immune system differs from the peripheral immune system. The primary immune cells in the brain are microglia, which serve as resident macrophages. Under normal conditions, microglia surveil the brain environment and support neuronal health. But following injury or during chronic disease, microglia can become activated and adopt a pro-inflammatory phenotype that produces cytokines, reactive oxygen species, and other mediators that damage surrounding neurons.
Semax appears to modulate microglial activation states. Rather than simply suppressing all microglial activity (which would impair the beneficial surveillance and repair functions), Semax shifts the balance toward a more neuroprotective microglial phenotype. This selective immunomodulation, dampening harmful inflammation while preserving or enhancing protective immune functions, is a more sophisticated approach than broad-spectrum anti-inflammatory drugs.
The specific gene expression changes induced by Semax in the immune system context include upregulation of anti-inflammatory cytokines (like IL-10), downregulation of pro-inflammatory mediators (like TNF-alpha and IL-1 beta), and modulation of complement system components. These changes create a local immune environment that favors tissue repair and neuronal survival rather than further damage.
Melanocortin System and Immune Regulation
The melanocortin system plays a well-characterized role in immune regulation throughout the body. Melanocortin receptors, particularly MC1R and MC3R, are expressed on immune cells including macrophages, monocytes, and lymphocytes. Activation of these receptors by melanocortin peptides, including ACTH and its fragments, generally produces anti-inflammatory effects.
Alpha-MSH, another melanocortin peptide derived from the same POMC precursor as ACTH, is one of the most potent endogenous anti-inflammatory molecules known. Semax, as an ACTH fragment analog, likely engages some of the same immunomodulatory pathways, though its specific receptor binding profile differs from alpha-MSH.
The melanocortin system's immune regulatory role is relevant to brain health because peripheral inflammation can drive central neuroinflammation through several pathways. Circulating cytokines can cross the blood-brain barrier at certain sites, vagal nerve signaling can transmit inflammatory status from the periphery to the brain, and inflammatory mediators can increase BBB permeability, allowing additional immune factors to enter the CNS. By modulating peripheral immune activity through melanocortin pathways, Semax may reduce the inflammatory burden on the brain from systemic sources.
Stress Response Modulation
As a fragment of ACTH, a hormone intimately involved in the hypothalamic-pituitary-adrenal (HPA) axis stress response, Semax has predictable effects on stress physiology. However, these effects are more nuanced than simple ACTH-like cortisol stimulation.
Semax, unlike full-length ACTH, does not significantly stimulate adrenal cortisol release at nootropic doses. The cortisol-stimulating activity of ACTH is primarily mediated by the C-terminal portion of the molecule (ACTH 18-39), which is not present in Semax. At the doses used for cognitive enhancement, Semax appears to modulate the stress response through central mechanisms rather than through direct adrenal stimulation.
Research has shown that Semax can influence the expression of stress-related genes in the brain, potentially improving stress resilience without the negative consequences of elevated cortisol. This central stress modulation may explain why some users report improved performance under pressure: Semax may help maintain cognitive function during stress by preventing the cognitive impairment that typically accompanies HPA axis hyperactivation.
The stress-modulating effects of Semax are distinct from the anxiolytic effects of Selank. While Selank directly reduces anxiety through GABAergic mechanisms, Semax works upstream by modulating the brain's response to stressors. The combination of both peptides, addressing both the upstream stress response and the downstream anxiety experience, provides a comprehensive approach to stress-related cognitive impairment.
Other Peripheral Effects
While the central nervous system effects of Semax receive the most attention, the peptide does have documented peripheral effects worth noting:
- Cardiovascular effects: Semax has been reported to have mild vasodilatory properties and may improve microcirculation. These effects are modest compared to dedicated cardiovascular drugs but may contribute to the peptide's benefits in cerebrovascular conditions.
- Metabolic effects: The glucose elevation noted in some diabetic patients suggests Semax may have minor effects on glucose metabolism. The mechanism isn't fully characterized but may involve melanocortin receptor activation in metabolic tissues.
- Wound healing: The PGP metabolite of Semax has roles in tissue repair through neutrophil recruitment and collagen metabolism modulation. Some anecdotal reports suggest accelerated wound healing during Semax use, though this hasn't been formally studied.
- Thymus and adrenal morphology: A study by Bobyntsev and colleagues (2015) examined the effects of Semax on thymus and adrenal morphology in rats, finding that the peptide influenced the structure of these immune and endocrine organs. These findings underscore the broader systemic effects of Semax beyond its CNS actions.
Practical User Guide: Optimizing Your Semax Protocol
Preparation and Reconstitution
For researchers and practitioners working with lyophilized Semax powder, proper reconstitution is essential for maintaining peptide integrity and achieving accurate dosing. The reconstitution process is straightforward but requires attention to detail.
Standard reconstitution protocol:
- Gather materials: Lyophilized Semax vial, bacteriostatic water (BAC water, containing 0.9% benzyl alcohol as preservative), sterile syringe, alcohol swabs.
- Clean the vial tops: Wipe the rubber stoppers of both the peptide vial and BAC water with alcohol swabs and allow to air dry.
- Calculate the volume: For a 10mg Semax vial, adding 2mL of BAC water gives a concentration of 5mg/mL (5,000 mcg/mL). For a nasal spray application targeting 200 mcg per spray, you'd want a concentration that delivers this dose in the spray volume of your device (typically 0.1 mL per spray, so 2mg/mL would give 200 mcg per spray).
- Add the water slowly: Draw up the BAC water with a sterile syringe and inject it gently down the inside wall of the peptide vial. Do NOT spray the water directly onto the lyophilized powder, as this can damage the peptide.
- Mix gently: Swirl the vial very gently to dissolve the powder. Do not shake vigorously, as this can create foam and denature the peptide. If the powder doesn't dissolve immediately, let the vial sit at room temperature for 5-10 minutes and gently swirl again.
- Transfer to nasal spray device: If using as a nasal spray, transfer the reconstituted solution to a sterile nasal spray bottle using a sterile syringe. Most commercially available peptide nasal spray bottles deliver approximately 0.1 mL per pump.
Proper Nasal Administration Technique
The effectiveness of intranasal Semax depends partly on proper administration technique. These steps help maximize absorption through the nasal mucosa:
- Clear the nasal passages: Gently blow your nose or use a saline rinse 5-10 minutes before application. A clear nasal passage improves peptide contact with the absorptive mucosa.
- Tilt the head slightly forward: This prevents the solution from draining into the throat (posterior drip), which would reduce nasal absorption and route the peptide to the gastrointestinal tract where it would be degraded.
- Aim toward the outer wall: When inserting the spray nozzle, angle it slightly toward the outer wall of the nostril (toward the ear), not toward the septum. This directs the spray toward the more absorptive lateral nasal mucosa.
- Spray and sniff gently: Actuate the spray while inhaling gently through the nose. A soft sniff helps distribute the solution over a larger mucosal surface area. Avoid hard sniffing, which can pull the solution past the nasal cavity into the throat.
- Alternate nostrils: If your protocol calls for multiple sprays per dose, alternate between nostrils.
- Don't blow your nose: Avoid blowing your nose for at least 15-20 minutes after administration to allow adequate absorption time.
Timing Optimization
The timing of Semax administration can significantly influence its effects. Consider the following guidelines:
- Morning administration (recommended): Taking Semax first thing in the morning aligns with the brain's natural dopamine and cortisol rhythms. Dopamine sensitivity is typically highest in the morning, which may amplify Semax's pro-focus effects. Many users report that morning administration provides a clear, productive start to the day.
- Pre-study or pre-work: Administering Semax 15-20 minutes before engaging in cognitively demanding tasks takes advantage of the rapid onset and maximizes the overlap between peak peptide effect and peak cognitive demand.
- Early afternoon redose: If your protocol includes a second dose, early afternoon (1-2 PM) is generally optimal. This avoids the natural post-lunch dip in alertness while leaving enough time before bedtime for any stimulatory effects to dissipate.
- Avoid evening dosing: Semax's dopaminergic and alerting effects can interfere with sleep onset if taken too late in the day. Most practitioners recommend that the last dose be administered no later than 3-4 PM.
- Fasted vs. fed state: For intranasal administration, food intake doesn't directly affect absorption since the peptide doesn't pass through the GI tract. However, a light meal after taking Semax may help prevent any mild dizziness that some users experience, particularly when starting.
Tracking and Measuring Your Response
Because Semax's cognitive effects are often described as subtle, having objective methods to track response can be valuable. Consider using:
- Cognitive testing apps: Applications like Cambridge Brain Sciences, Lumosity, or n-back training apps can provide standardized measures of working memory, processing speed, and attention that you can track over time. Test yourself at the same time each day to control for circadian variation.
- Subjective rating scales: A simple daily rating (1-10) of focus, mental clarity, motivation, and mood can reveal patterns that might not be apparent from day-to-day experience.
- Productivity metrics: If you're using Semax for work performance, tracking objective output (words written, tasks completed, code commits, etc.) during on-cycle vs. off-cycle periods can provide concrete data on effectiveness.
- Sleep quality: Monitor sleep quality using a wearable device or sleep diary. Any negative impact on sleep should prompt adjustment of dosing timing or amount.
- Journaling: A brief daily journal noting cognitive state, energy levels, and any side effects provides qualitative data that complements quantitative measures.
Tracking becomes especially important during the first cycle, as it helps establish whether Semax is providing meaningful benefit at the chosen dose and allows for informed adjustments during subsequent cycles.
Common Mistakes to Avoid
Based on common patterns reported in research communities and clinical settings, these are the most frequent mistakes made when using Semax:
- Skipping the cycling protocol: Continuous use without breaks may lead to diminished response. The standard cycling protocol exists for good reason - stick to it.
- Dosing too high at the start: Start at the lower end of the recommended range and increase gradually. More is not always better with nootropic peptides.
- Poor storage: Leaving reconstituted Semax at room temperature drastically reduces its potency. Refrigerate reconstituted solution and use within 30 days.
- Evening dosing: Taking Semax too late in the day disrupts sleep, which negates the cognitive benefits. Morning and early afternoon only.
- Expecting dramatic effects: Semax is not a stimulant. Its effects are more like "removing fog" than "adding fire." If you're expecting a caffeine-like buzz, you'll be disappointed.
- Combining with too many compounds: Using Semax alongside five other nootropics makes it impossible to determine what's working and what isn't. Keep your stack simple, especially when starting.
- Ignoring nasal technique: Spraying directly up the nose and sniffing hard can actually reduce absorption by pulling the peptide past the absorptive nasal mucosa. Gentle, directional application is key.
- Not controlling for other variables: Sleep, diet, exercise, stress, and caffeine intake all affect cognition. If you're evaluating Semax, try to keep these variables consistent during your trial period.
Semax Compared with Other Nootropic Peptides
Semax vs. Selank: Focus vs. Calm
Semax and Selank are often discussed together because they share a common developmental origin (both emerged from Russian peptide research programs) and are frequently used in combination. However, their pharmacological profiles are distinct and complementary.
| Parameter | Semax | Selank |
|---|---|---|
| Origin | ACTH(4-10) analog | Tuftsin analog (immunomodulatory tetrapeptide) |
| Primary classification | Nootropic | Anxiolytic |
| BDNF effects | Strong upregulation | Moderate upregulation |
| Dopamine modulation | Significant enhancement | Minimal direct effect |
| Serotonin modulation | Moderate enhancement | Moderate enhancement |
| GABA modulation | Minimal | Significant (benzodiazepine-like) |
| Best for | Focus, learning, motivation | Anxiety reduction, emotional stability |
| Typical user experience | "Sharper, more focused" | "Calmer, less stressed" |
| Administration | Intranasal | Intranasal |
| Cycling | 10 on / 14 off | 14 on / 14 off |
The combination of Semax and Selank has been used in Russian clinical practice without reported adverse interactions. They target different receptor systems: Semax works primarily through melanocortin receptors, BDNF, and monoamine pathways, while Selank modulates GABAergic and enkephalin systems. This pharmacological separation makes them well-suited for combination use.
For users who experience anxiety alongside cognitive deficits, the Semax-Selank combination offers a compelling solution. Semax sharpens cognitive performance while Selank reduces the anxiety that often impairs it. FormBlends offers both Selank and Selank Nasal formulations for researchers exploring this combination. Detailed information on Selank's pharmacology is available in our Selank research report.
The Semax-Selank Combination: Detailed Pharmacological Analysis
The Semax-Selank combination deserves special attention because it represents the most established peptide nootropic stack in clinical practice. Understanding why these two peptides complement each other requires examining their mechanisms in detail.
Semax's primary cognitive effects derive from enhanced dopaminergic transmission in the prefrontal cortex and striatum, combined with BDNF-driven neuroplasticity in the hippocampus. These changes promote focused attention, faster information processing, enhanced working memory, and improved motivation. However, dopaminergic enhancement can sometimes produce restlessness or an "edge" in sensitive individuals, particularly during high-stress periods.
Selank addresses this potential limitation through a completely different pharmacological mechanism. By modulating GABA-A receptor function (similar to benzodiazepines but without the sedation, dependence, or memory impairment), Selank reduces anxiety and promotes emotional stability. It also influences enkephalin metabolism, which affects the brain's endogenous opioid system and contributes to stress resilience and emotional regulation.
When combined, the two peptides create what practitioners describe as "calm focus" - a state of enhanced cognitive performance without the anxiety or tension that can accompany dopaminergic stimulation. The Selank component removes the emotional friction that often impedes cognitive performance, while the Semax component provides the neurochemical substrate for enhanced processing, attention, and memory.
From a receptor pharmacology perspective, there is minimal overlap between the two peptides' mechanisms. Semax works through MC4R, TrkB, and dopamine/serotonin modulation. Selank works through GABA-A receptors and enkephalin metabolism. This separation of mechanisms is important because it means the two peptides are unlikely to compete for the same receptors, and their effects should be additive rather than interfering with each other.
The practical implementation of this combination is straightforward: both peptides are administered intranasally, and they can be applied sequentially (Semax first, followed by Selank 5-10 minutes later) or concurrently (one in each nostril). Some practitioners prepare combined solutions containing both peptides at appropriate concentrations, though stability data for combined formulations is limited.
Russian clinical practice has accumulated years of experience with this combination, and no adverse interactions have been reported. The combination is used for conditions ranging from post-stroke cognitive recovery to examination stress in students to age-related cognitive decline. For those interested in exploring this combination, FormBlends offers both Semax and Selank Nasal formulations, with detailed information on each compound available in the respective research reports.
Semax vs. Cerebrolysin
Cerebrolysin is a complex mixture of low-molecular-weight neuropeptides and free amino acids derived from porcine brain tissue. While it's not a single peptide like Semax, it's the most directly comparable neuroprotective agent in terms of clinical applications and evidence base. Both are approved in multiple countries for neurological conditions, and both target neurotrophic signaling.
| Parameter | Semax | Cerebrolysin |
|---|---|---|
| Composition | Single synthetic heptapeptide | Complex peptide mixture (porcine brain-derived) |
| Administration | Intranasal | Intravenous or intramuscular injection |
| Mechanism | BDNF/NGF upregulation, monoamine modulation | Mimics endogenous neurotrophic factors |
| Approved indications | Stroke, encephalopathy, optic nerve disease | Stroke, TBI, Alzheimer's disease, dementia |
| Ease of use | Simple nasal spray | Requires injection (typically IV infusion) |
| Cost | Moderate | Higher (complex manufacturing) |
| Safety profile | Minimal side effects reported | Generally well-tolerated; injection-site reactions possible |
For more on Cerebrolysin's specific mechanisms and clinical evidence, see our detailed Cerebrolysin report.
Semax vs. Dihexa and P21
Dihexa and P21 represent different approaches to nootropic peptide design. Dihexa is an angiotensin IV analog that targets hepatocyte growth factor (HGF) signaling, while P21 is a synthetic peptide fragment designed to mimic the neurotrophic effects of the ciliary neurotrophic factor (CNTF).
- Dihexa is often described as one of the most potent nootropic peptides in preclinical research. It crosses the blood-brain barrier and promotes dendritic spine formation and synaptic connectivity. Unlike Semax, which has decades of clinical use, Dihexa's evidence base is primarily preclinical. See the Dihexa research report for the full evidence review.
- P21 targets the CNTF pathway, which is involved in neuronal differentiation and survival. It has shown promise in animal models of Alzheimer's disease, where it improved cognitive function and reduced tau pathology. Like Dihexa, its clinical evidence is more limited than Semax's.
Semax vs. Noopept
Noopept (N-phenylacetyl-L-prolylglycine ethyl ester) is another Russian-developed nootropic that's frequently compared with Semax. While both are used for cognitive enhancement, their pharmacological profiles differ substantially:
| Parameter | Semax | Noopept |
|---|---|---|
| Chemical class | Heptapeptide (ACTH analog) | Dipeptide derivative (piracetam-related) |
| Administration | Intranasal | Sublingual, oral, or intranasal |
| Primary mechanism | BDNF/TrkB upregulation, melanocortin signaling | Cycloprolylglycine release, AMPA receptor modulation |
| BDNF effects | Strong, rapid upregulation | Moderate, with NGF emphasis |
| Dopamine effects | Significant enhancement | Minimal direct dopamine effects |
| Anxiolytic properties | Mild to none | Moderate (via HIF-1alpha pathway) |
| Onset | 15-20 minutes (nasal) | 15-20 minutes (sublingual) |
| Duration | 2-4 hours | 3-6 hours |
| Typical daily dose | 200-600 mcg | 10-30 mg |
| Clinical evidence | Extensive (30+ years Russian clinical use) | Limited clinical data |
Noopept is often preferred by users who want both cognitive enhancement and mild anxiety reduction, as it provides a more balanced profile in that regard. Semax is typically chosen by users who prioritize focus, motivation, and BDNF upregulation, and who are willing to address anxiety separately (for example, with Selank). The two compounds can be combined, as they work through largely different mechanisms, though this combination has not been formally studied.
One practical difference is that Noopept is available in oral form (tablets or sublingual powder), while Semax is primarily administered intranasally. For users who find nasal spray inconvenient or uncomfortable, Noopept offers a simpler administration route, though the nose-to-brain delivery advantage of intranasal Semax may result in more efficient brain penetration at lower doses.
Comprehensive Nootropic Peptide Comparison
To help researchers and practitioners compare all the major nootropic peptides discussed in this report, here is a comprehensive comparison matrix covering the key differentiating factors:
| Feature | Semax | Selank | Dihexa | P21 | Pinealon | Noopept |
|---|---|---|---|---|---|---|
| Primary target | BDNF/MC4R | GABA/Enkephalin | HGF/c-Met | CNTF pathway | Pineal gland | AMPA/NGF |
| Main benefit | Focus, learning | Anxiety relief | Synaptogenesis | Neuroprotection | Sleep, circadian | Memory, focus |
| Clinical approval | Russia/Ukraine | Russia/Ukraine | None | None | Russia | Russia |
| Route | Nasal | Nasal | Oral/nasal | Injection/nasal | Sublingual | Oral/sublingual |
| Research depth | Deep | Moderate | Preclinical | Preclinical | Moderate | Moderate |
| Best time | Morning | Any time | Morning | Morning | Evening | Morning |
| Stacks well with | Selank, BPC-157 | Semax | Semax | Semax, NAD+ | Semax (night) | Semax |
This comparison highlights an important point: no single nootropic peptide does everything. Each compound has strengths in specific domains, and the "best" choice depends on the individual's specific needs, whether that's pure cognitive enhancement (Semax), anxiety management (Selank), maximal synaptogenesis (Dihexa), circadian optimization (Pinealon), or some combination thereof.
The Peptide Research Hub at FormBlends provides detailed research reports on each of these compounds, allowing researchers to make informed decisions about which peptides best match their specific research or clinical objectives.
Semax vs. Pinealon
Pinealon (Glu-Asp-Arg) is a short peptide bioregulator that targets the pineal gland and central nervous system. While Semax is primarily a cognitive enhancer and neuroprotectant, Pinealon focuses on normalizing circadian rhythms, improving sleep quality, and supporting melatonin synthesis. Our Pinealon research report covers this complementary peptide in detail.
For researchers interested in comprehensive neuroprotective strategies, the combination of daytime Semax (for cognitive support and BDNF upregulation) with evening Pinealon (for sleep optimization and circadian regulation) represents a logical day-night protocol that addresses both active cognition and restorative sleep.
Figure 6: Comparative overview of nootropic peptides. Each compound targets distinct but complementary pathways in brain health and cognitive enhancement.
Stacking Strategies: Combining Semax with Other Compounds
Principles of Peptide Stacking
Peptide stacking refers to the concurrent use of multiple peptides to achieve complementary or additive effects. While the concept is appealing, it requires careful consideration of pharmacological interactions, timing, and individual response. The general principles for stacking with Semax include:
- Target different pathways: Combine compounds that work through distinct mechanisms rather than doubling up on the same pathway. This reduces the risk of receptor desensitization and maximizes the breadth of benefit.
- Start with one compound: Establish your baseline response to Semax before adding other peptides. This allows you to attribute any effects (positive or negative) to specific compounds.
- Limit complexity: Most experienced practitioners recommend no more than 2-3 peptides simultaneously. Beyond that, it becomes extremely difficult to determine which compounds are contributing to the overall effect.
- Consider timing: Some peptides are better suited for morning use (stimulating, focus-enhancing) while others work best in the evening (calming, sleep-supportive). Design your stack to align with the body's natural circadian rhythms.
The Cognitive Performance Stack
For pure cognitive enhancement, a well-regarded combination is:
Cognitive Stack Protocol
- Morning: Semax 200-400 mcg intranasal - for BDNF upregulation and dopamine/serotonin modulation
- Morning: Selank 200-400 mcg intranasal - for anxiety reduction and calm focus
- Evening: Pinealon - for sleep optimization and circadian support
Cycle: 10-14 days on, followed by 14 days off. Semax and Selank can be administered simultaneously through the nasal route without known interactions.
The Neuroprotection and Recovery Stack
For individuals recovering from brain injury, dealing with chronic neurodegenerative conditions, or seeking to maximize neural resilience:
- Semax (200-600 mcg/day nasal) - BDNF upregulation, anti-inflammatory neuroprotection
- BPC-157 (250-500 mcg/day) - tissue repair, nitric oxide modulation, gut-brain axis support
- NAD+ - cellular energy support, DNA repair, mitochondrial function
This combination targets neuroprotection from three complementary angles: neurotrophic support (Semax), tissue repair and vascular protection (BPC-157), and cellular energy and mitochondrial health (NAD+). Each compound addresses a different aspect of the recovery process, reducing redundancy while maximizing coverage.
The Longevity and Brain Aging Stack
For age-related cognitive decline prevention and neural longevity:
- Semax (200-400 mcg/day nasal) - maintains BDNF levels and synaptic plasticity
- Epithalon - telomere support and cellular aging protection
- GHK-Cu - gene expression modulation, anti-aging signaling, tissue repair
- NAD+ - metabolic support for aging neurons
This stack combines Semax's brain-specific neurotrophic effects with broader anti-aging compounds. Epithalon supports telomere maintenance, GHK-Cu modulates hundreds of genes involved in aging and repair, and NAD+ addresses the metabolic decline that accompanies brain aging. The full Science & Research section on FormBlends provides additional context on how these compounds interact at the molecular level.
Lifestyle Factors That Amplify Semax's Effects
Beyond peptide stacking, certain lifestyle interventions can amplify Semax's cognitive and neuroprotective benefits through complementary biological mechanisms. These lifestyle factors don't just add to Semax's effects; they create synergies by activating overlapping but distinct signaling pathways.
Aerobic Exercise
Regular aerobic exercise is one of the most reliable natural methods for increasing BDNF levels. Studies have shown that even a single bout of moderate-intensity exercise (30 minutes of running, cycling, or swimming) can increase serum BDNF by 20-30%. Chronic exercise training further amplifies this effect and produces additional benefits including improved cerebral blood flow, enhanced neurogenesis in the hippocampal dentate gyrus, and increased synaptic density.
When combined with Semax, exercise and the peptide activate BDNF expression through different upstream mechanisms. Exercise increases BDNF partly through lactate signaling (the ketone body beta-hydroxybutyrate inhibits histone deacetylases that suppress BDNF transcription) and through increased neuronal activity in the hippocampus during movement. Semax activates BDNF through melanocortin receptor signaling and CREB phosphorylation. Because these upstream mechanisms are distinct, the combined effect on BDNF could theoretically be additive or even greater than additive.
Practical recommendation: Time your Semax dose 15-20 minutes before exercise to allow the peptide to reach peak brain levels during the exercise session. Morning exercise combined with morning Semax provides the dual benefit of circadian alignment (both exercise and dopamine sensitivity peak in the morning) and maximum overlap of their BDNF-enhancing effects.
Sleep Optimization
Sleep is when the brain consolidates memories formed during the day, clears metabolic waste through the glymphatic system, and performs essential maintenance functions. BDNF, which Semax upregulates during waking hours, plays a critical role in sleep-dependent memory consolidation. During slow-wave sleep, BDNF-dependent synaptic plasticity mechanisms convert short-term memories into long-term storage.
Poor sleep undermines the cognitive benefits of any nootropic intervention, including Semax. If you're not sleeping well, the BDNF you're producing during the day can't be effectively utilized for memory consolidation at night. Optimizing sleep quality through consistent sleep-wake schedules, light hygiene (bright light in the morning, dim light in the evening), temperature regulation (a cool sleeping environment), and avoidance of late-day stimulants should be considered a prerequisite for maximizing Semax's cognitive benefits.
For those seeking peptide support for sleep quality, Pinealon targets the pineal gland and melatonin synthesis, providing a complementary night-time approach to Semax's day-time cognitive enhancement. See our Pinealon research report for details.
Meditation and Mindfulness
Regular meditation practice has been associated with increased BDNF levels, enhanced dopamine signaling, and improved cortical connectivity, the same biological processes that Semax modulates. Neuroimaging studies of experienced meditators show increased gray matter density in the hippocampus, prefrontal cortex, and insula, regions that are also targets of Semax's neurotrophic effects.
The convergence of meditation and Semax effects on attention networks is particularly interesting. Both interventions enhance the brain's ability to maintain focused attention while reducing distractibility. Meditators using Semax may find that the peptide accelerates the development of meditation-related attentional skills, while the meditation practice amplifies Semax's pro-focus effects.
Nutritional Support
Several nutritional factors interact with Semax's mechanisms and should be considered when designing a comprehensive cognitive optimization protocol:
- Omega-3 fatty acids (DHA/EPA): DHA is a major structural component of neuronal membranes and is required for BDNF signaling efficiency. BDNF-TrkB signaling is less effective in DHA-deficient neural membranes because TrkB receptors need a fluid, DHA-rich lipid environment to function optimally. Ensuring adequate omega-3 intake (through fatty fish or supplementation) supports the biological infrastructure that Semax's BDNF upregulation depends on.
- Zinc: The hippocampus has the highest zinc concentrations in the brain, and zinc is released from synaptic vesicles during neurotransmission. Zinc interacts with BDNF signaling through matrix metalloproteinase activation (which converts pro-BDNF to mature BDNF) and through direct modulation of TrkB receptor function. Semax itself interacts with zinc ions through its histidine residue, and adequate zinc status may be important for optimal peptide-receptor interactions.
- Magnesium: Magnesium is a cofactor for hundreds of enzymatic reactions, including many involved in neurotransmitter synthesis and synaptic plasticity. Magnesium L-threonate, which crosses the BBB more effectively than other magnesium forms, has been shown to enhance synaptic density and BDNF-dependent plasticity in animal models. Combining adequate magnesium with Semax provides the enzymatic cofactor support needed for the biological changes Semax initiates.
- B-vitamins: The B-vitamin complex, particularly B6 (pyridoxine), B9 (folate), and B12 (cobalamin), is essential for neurotransmitter synthesis and methylation reactions. Since Semax enhances dopamine and serotonin signaling, ensuring adequate substrate availability through B-vitamin sufficiency supports the neurotransmitter changes the peptide produces.
- Creatine: Brain creatine supports cellular energy (ATP) reserves and has been associated with improved cognitive function, particularly under conditions of metabolic stress. Since many of Semax's downstream signaling pathways are ATP-dependent, adequate cellular energy reserves may amplify the peptide's effects.
Cognitive Training
The principle of "neurons that fire together wire together" suggests that combining Semax's neuroplasticity-enhancing effects with structured cognitive training could produce greater lasting cognitive improvements than either approach alone. When Semax upregulates BDNF and enhances synaptic plasticity, the brain is in a state of heightened readiness for structural change. Providing specific cognitive challenges during this window (learning a new language, practicing a musical instrument, engaging in working memory training) directs the enhanced plasticity toward functionally useful neural circuit modifications.
This is analogous to how physical therapy is most effective during the window of enhanced neuroplasticity that follows stroke, the same principle that underlies Semax's clinical use in combination with early rehabilitation. The brain's potential for change is increased by Semax, but the direction and specificity of that change is determined by the experiences and challenges encountered during the enhanced plasticity window.
Stacking Disclaimer
Peptide stacking protocols described here are based on theoretical pharmacological rationale and practitioner experience, not controlled clinical trials evaluating specific combinations. Individual responses vary, and the safety of specific multi-peptide regimens has not been formally established. Always consult a qualified healthcare professional before implementing any combination protocol, particularly if you have existing medical conditions or are taking prescription medications.
Figure 7: Semax stacking strategies. Each protocol targets distinct aspects of brain health using complementary mechanisms of action.
Molecular Biology of Semax: Gene Expression, Epigenetics, and Proteomics
Transcriptomic Analysis: What Semax Does to Gene Expression
Modern molecular biology has provided unprecedented insight into how Semax works at the gene expression level. Using techniques like RNA-sequencing (RNA-Seq) and microarray analysis, researchers have cataloged the specific genes whose expression is altered by Semax treatment in various brain regions and under different conditions (normal brain vs. ischemic brain).
The most comprehensive transcriptomic study of Semax was published by Filippenkov and colleagues (2020), who used RNA-Seq to analyze genome-wide expression changes in rat brain tissue following experimental cerebral ischemia-reperfusion with and without Semax treatment. This study identified thousands of differentially expressed genes (DEGs) and organized them into functional categories that reveal the biological processes most affected by Semax.
The key transcriptomic findings can be organized into functional clusters:
Neurotrophic Factor Genes
Semax upregulates the expression of multiple neurotrophic factor genes, not just BDNF. The transcriptomic data reveals increased expression of:
- Bdnf (brain-derived neurotrophic factor) - the most consistently upregulated neurotrophin
- Ngf (nerve growth factor) - supporting cholinergic neuron survival
- Ntrk2 (TrkB receptor) - amplifying BDNF signaling capacity
- Ntrk1 (TrkA receptor) - amplifying NGF signaling
- Various growth factor-associated signaling molecules in the MAPK cascade
The coordinated upregulation of both neurotrophins and their receptors creates a positive feedback loop: more growth factors are produced, and the brain becomes more sensitive to them. This double action is more effective than simply increasing growth factor levels alone, as it avoids the bottleneck of limited receptor availability.
Inflammatory Response Genes
In the context of ischemia, Semax produces a dramatic shift in inflammatory gene expression. The peptide suppresses genes encoding:
- Il1b (interleukin-1 beta) - a master regulator of neuroinflammation
- Tnf (tumor necrosis factor) - a pro-inflammatory cytokine that promotes neuronal death
- Il6 (interleukin-6) - involved in acute-phase inflammatory response
- Ccl2 and Ccl3 (chemokines) - recruit inflammatory cells to the injury site
- Mmp9 (matrix metalloproteinase 9) - degrades the blood-brain barrier
Simultaneously, Semax upregulates anti-inflammatory genes including Il10 (interleukin-10), which promotes resolution of inflammation and tissue repair. This selective immunomodulation is more therapeutically desirable than blanket immunosuppression, which would impair the beneficial aspects of the immune response.
Neurotransmission-Related Genes
Ischemia dramatically suppresses the expression of genes involved in synaptic transmission. Semax treatment partially or fully reverses this suppression, restoring expression of genes encoding:
- Synaptic vesicle proteins (involved in neurotransmitter packaging and release)
- Neurotransmitter receptor subunits (especially glutamate and GABA receptors)
- Ion channel subunits (required for action potential generation and propagation)
- Synaptic scaffolding proteins (organizing the post-synaptic density)
- Axon guidance molecules (relevant for neural circuit repair)
The restoration of neurotransmission-related gene expression after ischemia is particularly significant because synaptic function recovery is what ultimately determines clinical outcome. A neuron that survives ischemia but can't form functional synapses provides little clinical benefit. By supporting both neuronal survival and synaptic function recovery, Semax addresses both sides of the neuroprotection equation.
Proteomic Validation
Transcriptomic data tells you what genes are being expressed, but protein-level analysis is needed to confirm that these gene expression changes actually translate into functional protein changes. A 2021 study by Sudarkina and colleagues performed proteomic analysis of rat brain tissue following ischemia-reperfusion with and without Semax treatment.
The proteomic data confirmed the transcriptomic findings: Semax treatment after ischemia led to increased levels of proteins involved in synaptic transmission, energy metabolism, and cellular survival, while reducing levels of proteins associated with inflammation and cell death. Specific proteins elevated by Semax included synaptic vesicle glycoprotein 2A, synaptophysin (a marker of synaptic integrity), and several mitochondrial enzymes involved in energy production.
The convergence of transcriptomic and proteomic data strengthens the mechanistic case for Semax. When gene expression changes are confirmed at the protein level, it provides high confidence that the molecular effects are biologically meaningful and likely to translate into functional outcomes.
Potential Epigenetic Effects
An emerging area of interest is whether Semax's effects on gene expression involve epigenetic mechanisms. Epigenetic modifications, including DNA methylation, histone acetylation, and chromatin remodeling, regulate gene expression without altering the DNA sequence itself. Many neuropeptides and neurotrophic factors influence gene expression partly through epigenetic mechanisms.
BDNF itself is subject to complex epigenetic regulation. The BDNF gene has multiple promoters, each associated with different exons, and the activity of these promoters is controlled by DNA methylation and histone modifications. When Semax upregulates BDNF exon III expression specifically, this suggests involvement of promoter-specific regulatory mechanisms that may include epigenetic changes.
Furthermore, BDNF signaling through TrkB activates intracellular cascades (particularly the MAPK/ERK pathway) that can modify histone acetyltransferase and histone deacetylase activity, leading to chromatin remodeling and broad changes in gene expression. This creates the possibility that Semax's initial effects on BDNF expression could cascade into wider epigenetic changes that persist beyond the acute presence of the peptide.
While direct evidence for Semax-induced epigenetic modifications is still limited, the circumstantial evidence is suggestive. The lasting effects of Semax on cognitive function, which can persist beyond the acute pharmacokinetic window, are consistent with epigenetic mechanisms that create more stable changes in gene expression than simple receptor activation alone.
Melanocortin Receptor Signaling in Detail
Understanding Semax's mechanism requires a closer look at melanocortin receptor signaling. The five melanocortin receptor subtypes (MC1R through MC5R) are G protein-coupled receptors that share structural features but have distinct tissue distributions and signaling properties:
| Receptor | Primary Location | Key Function | Semax Relevance |
|---|---|---|---|
| MC1R | Skin, immune cells | Melanin production, anti-inflammation | May contribute to immunomodulatory effects |
| MC2R | Adrenal cortex | Cortisol production (ACTH receptor) | Semax does NOT significantly activate (no cortisol stimulation) |
| MC3R | Brain (hypothalamus), immune cells | Energy homeostasis, immune regulation | May contribute to metabolic and immune effects |
| MC4R | Brain (hippocampus, cortex, amygdala) | Appetite, cognition, synaptic plasticity | Primary receptor for cognitive effects |
| MC5R | Sebaceous glands, brain | Exocrine function, immune modulation | Limited relevance to primary effects |
The critical point is that Semax, as a modified ACTH(4-10) fragment, has a different receptor binding profile than full-length ACTH. Full ACTH binds strongly to MC2R (the "classical" ACTH receptor on adrenal cells), triggering cortisol release. Semax, lacking the C-terminal portion of ACTH responsible for MC2R selectivity, has weak affinity for MC2R but retains affinity for MC4R and potentially MC3R and MC1R. This is why Semax doesn't raise cortisol levels but does enhance cognition and modulate immunity.
MC4R signaling in the hippocampus and cortex activates adenylyl cyclase, increasing intracellular cyclic AMP (cAMP) levels. cAMP activates protein kinase A (PKA), which phosphorylates the transcription factor CREB (cAMP response element-binding protein). Phosphorylated CREB enters the nucleus and binds to CRE elements in the promoter regions of target genes, including the BDNF gene. This CREB-mediated transcription is one of the most important molecular pathways for long-term memory formation, and Semax's engagement of this pathway provides a direct molecular link between melanocortin receptor activation and cognitive enhancement.
Intracellular Calcium Dynamics
The most recent addition to our understanding of Semax's cellular mechanisms comes from the 2025 study by Bhatt and colleagues examining intracellular calcium dynamics. Using calcium imaging in hippocampal brain slices, they showed that Semax at 1 micromolar concentration significantly increased the frequency of spontaneous calcium oscillations in CA1 pyramidal neurons.
Calcium oscillations in neurons serve as a critical intracellular signaling mechanism. Different patterns of calcium fluctuation activate different transcription factors and signaling cascades. Low-frequency, high-amplitude calcium transients tend to activate NFAT (nuclear factor of activated T-cells), while high-frequency calcium oscillations preferentially activate NF-kB and CREB. By increasing the frequency of calcium oscillations, Semax may bias intracellular signaling toward CREB activation, which as discussed above, promotes BDNF expression and synaptic plasticity.
This finding also provides a potential mechanism for Semax's effects on synaptic transmission. Presynaptic calcium dynamics directly control neurotransmitter release probability, and changes in calcium oscillation frequency could alter the timing and efficiency of synaptic transmission. Enhanced calcium signaling in postsynaptic neurons could also affect the induction of long-term potentiation (LTP), the synaptic mechanism underlying learning and memory.
The calcium signaling data adds a new dimension to our understanding of how Semax works at the cellular level. Rather than simply increasing the total amount of BDNF or dopamine, the peptide appears to modulate the fundamental electrophysiological properties of neurons in ways that favor enhanced information processing and plasticity.
Historical and Cultural Context: Nootropics in Russian Medicine
The Russian Nootropic Tradition
To fully appreciate Semax's significance, it helps to understand the broader context of nootropic research in Russia and the former Soviet Union. The concept of "nootropics," compounds that enhance cognitive function without significant side effects, was formally coined by Romanian psychologist and chemist Corneliu Giurgea in 1972 when he developed piracetam. But the Soviet research establishment embraced the nootropic concept with particular enthusiasm, investing significant resources into developing cognitive-enhancing compounds for both military and civilian applications.
The Soviet and later Russian approach to nootropic development differed from Western pharmaceutical practices in several ways. Russian researchers tended to favor peptide-based compounds, drawing on the rich biology of endogenous neuropeptides. They also maintained a more holistic view of brain function, looking for compounds that modulated multiple neurotransmitter systems simultaneously rather than targeting single receptors. And they placed significant emphasis on "adaptogenic" properties, the ability of a compound to help the brain adapt to stress and maintain performance under adverse conditions.
This research tradition produced several notable compounds beyond Semax:
- Selank (1990s) - the anxiolytic complement to Semax, derived from the immunomodulatory peptide tuftsin
- Noopept (1996) - a dipeptide nootropic with a distinct mechanism involving cycloprolylglycine and NGF/BDNF modulation
- Cortexin - a complex of polypeptides derived from porcine cerebral cortex, used for cognitive disorders and brain injury
- Cerebrolysin - while developed in Austria, it was widely adopted and extensively studied in Russian clinical practice for neurodegenerative diseases
Semax emerged from this tradition as one of the most successful examples of rational peptide drug design. Unlike the complex peptide mixtures (Cerebrolysin, Cortexin) that contain multiple undefined active components, Semax is a single, well-characterized molecule with a defined mechanism of action. This makes it more amenable to rigorous scientific study and quality control.
Military and Space Research Applications
Like many cognitive-enhancing compounds developed in the Soviet/Russian research establishment, Semax has connections to military and space research programs. The Soviet military was particularly interested in compounds that could maintain cognitive performance under extreme stress, including sleep deprivation, high-G forces, hypoxia, and combat stress.
Semax was reportedly tested in these contexts, though much of the military research remains classified or published only in restricted military journals. What has been publicly reported suggests that Semax improved cognitive performance under stress conditions, maintained attention during extended periods of sleep deprivation, and enhanced the ability to perform complex tasks under adverse environmental conditions.
The space program's interest in Semax was related to the cognitive challenges of spaceflight. Astronauts face a unique combination of stressors, including microgravity (which can cause cognitive changes through intracranial pressure alterations), radiation exposure, isolation, and disrupted circadian rhythms. A neuroprotective compound that could maintain cognitive function under these conditions would be valuable for long-duration space missions.
While the specific military and space research data is not publicly available in detail, the fact that Semax was investigated in these demanding contexts suggests the compound was taken seriously by researchers working at the highest levels of Soviet and Russian science.
Key Research Institutions and Contributors
The development and ongoing study of Semax has involved several prominent Russian research institutions, each contributing distinct aspects to our understanding of the peptide:
- Institute of Molecular Genetics, Russian Academy of Sciences (Moscow): This is the birthplace of Semax. Under the leadership of Nikolai Myasoedov, this institute designed the original peptide, characterized its structure-activity relationships, and conducted the foundational biochemical studies that established Semax's mechanism of action. The institute continues to publish research on Semax analogs and metabolites.
- Research Center of Neurology, Russian Academy of Medical Sciences (Moscow): This center conducted many of the key clinical trials evaluating Semax in stroke patients. Under the direction of prominent Russian neurologists including Evgeny Gusev and Veronika Skvortsova (who later became Russia's Health Minister), the center established the clinical protocols for Semax use in ischemic stroke that remain the standard of care in Russian neurology.
- Moscow State University, Department of Physiology: Numerous preclinical studies on Semax's behavioral and cognitive effects were conducted here, including the Morris water maze and passive avoidance studies that established Semax's nootropic properties.
- Institute of Gene Biology, Russian Academy of Sciences: This institute has been at the forefront of transcriptomic research on Semax, producing the genome-wide gene expression studies that revealed the peptide's broad effects on inflammatory, neurotrophic, and neurotransmission-related gene networks.
- Helmholtz Moscow Research Institute of Eye Diseases: The clinical studies evaluating Semax in optic nerve disease and glaucomatous neuropathy were conducted here, establishing the ophthalmological applications of the peptide.
The collaborative nature of Russian Semax research, spanning molecular biology, pharmacology, neurology, and ophthalmology, reflects the multidisciplinary approach needed to fully characterize a compound with such wide-ranging effects. The depth and breadth of this research program is unusual for a peptide drug, and it provides a rich foundation of knowledge that subsequent international research can build upon.
Publication Landscape and Literature Access
One of the practical challenges for international researchers interested in Semax is navigating the publication landscape. The Semax literature is distributed across several categories:
- English-language international journals: The highest-impact Semax research has been published in journals like Brain Research, Neurochemical Research, BMC Genomics, Genes, and the International Journal of Molecular Sciences. These papers are accessible through standard databases like PubMed and are written by Russian research groups who chose to publish in English for maximum international visibility.
- Russian-language journals: A substantial body of clinical and pharmacological research on Semax exists in Russian journals such as Zhurnal Nevrologii i Psikhiatrii im. S.S. Korsakova (Journal of Neurology and Psychiatry), Vestnik Oftalmologii (Ophthalmology Herald), Bulletin of Experimental Biology and Medicine, and others. Many of these articles have English abstracts available through PubMed, but the full texts are often only in Russian.
- Conference proceedings and dissertations: Additional data on Semax exists in Russian academic conference proceedings and doctoral dissertations that are generally not indexed in international databases.
- Regulatory documents: The pharmacological documentation submitted for Semax's regulatory approval in Russia contains safety and efficacy data that may not be fully represented in published journal articles.
For researchers conducting systematic reviews or meta-analyses of Semax, this fragmented publication landscape poses challenges. A complete assessment of the evidence requires accessing both English and Russian-language sources, which may introduce language bias into review processes. This is an important consideration when evaluating the overall strength of the evidence base.
From Russian Medicine to Global Biohacking
Semax remained largely unknown outside of Russia and former Soviet states until the 2010s, when the growing global interest in nootropics and biohacking brought it to wider attention. Early adopters in the Western biohacking community discovered Semax through English translations of Russian medical literature and began experimenting with it for cognitive enhancement.
The peptide quickly gained a reputation in nootropic communities as a "clean" cognitive enhancer. Users reported improved focus, mental clarity, and motivation without the side effects common to stimulant nootropics (jitteriness, anxiety, crash, insomnia). The nasal spray formulation made it easy to use, and the rapid onset of effects (15-20 minutes) provided quick feedback that encouraged continued experimentation.
By the mid-2010s, Semax was among the most discussed peptide nootropics in online communities like Reddit's r/nootropics, Longecity, and various biohacking forums. User reports, while not constituting scientific evidence, were remarkably consistent in describing subtle but meaningful improvements in cognitive function, particularly in areas of sustained attention, verbal fluency, and creative problem-solving.
The transition from Russian clinical medicine to global biohacking use brought both opportunities and challenges. On the positive side, it expanded access to a potentially valuable cognitive tool and generated grassroots interest that may eventually drive more rigorous Western clinical research. On the negative side, the lack of regulatory oversight in the research peptide market means that product quality varies, dosing guidance is often based on anecdote rather than evidence, and users lack the medical supervision that accompanies clinical use in Russia.
This context underscores the importance of working with reputable peptide suppliers who provide quality documentation and testing results. FormBlends Semax is manufactured to research-grade specifications with full analytical documentation, providing researchers and practitioners with confidence in product quality.
Safety Profile
Clinical Safety Data
Semax has been used clinically in Russia and Ukraine since 1994, providing nearly three decades of real-world safety data. This extended period of clinical use, while not equivalent to the double-blind, placebo-controlled trials required by Western regulatory agencies, provides meaningful reassurance about the peptide's safety profile in real-world populations.
The overall safety assessment based on available data is favorable. No clinical or preclinical trials have reported serious adverse events following intranasal or subcutaneous Semax administration. The peptide does not appear to be addictive, does not produce tolerance requiring dose escalation, and does not cause withdrawal symptoms upon discontinuation.
Reported Side Effects
The side effect profile of Semax is mild and primarily related to the nasal route of administration:
| Side Effect | Frequency | Severity | Management |
|---|---|---|---|
| Nasal irritation (dryness, burning) | Common (~15-20%) | Mild | Self-limiting; use saline rinse before application |
| Nasal mucosa discoloration | Uncommon (~10%) | Mild | Reversible upon discontinuation |
| Headache | Uncommon (~5-8%) | Mild to moderate | Usually resolves with dose reduction |
| Dizziness | Rare (~2-3%) | Mild | Transient; associated with initial use |
| Sleep disturbance | Uncommon | Mild | Avoid evening dosing |
| Mild blood glucose elevation (diabetics) | ~7.4% of diabetic patients | Mild | Monitor blood glucose; consult physician |
| Injection site reaction (SC route) | Variable | Mild | Site rotation, proper technique |
Special Populations and Precautions
- Diabetes: Clinical observations have noted mild blood glucose increases in approximately 7.4% of diabetic patients using Semax. While this is a small percentage and the increases were mild, diabetic patients should monitor blood glucose levels during Semax use and consult their physician.
- Pregnancy and lactation: Semax has not been evaluated for safety during pregnancy or breastfeeding. Due to the absence of safety data, use during pregnancy is not recommended.
- Hepatic and renal impairment: The peptide has not been specifically studied in patients with liver or kidney disease. Given that peptides are generally metabolized by tissue peptidases rather than hepatic enzymes, hepatic impairment may be less of a concern than with small-molecule drugs, but formal data is lacking.
- Children: Semax has been used in Russia for perinatal brain injury in newborns under medical supervision. However, use in children should only occur under strict medical guidance.
- Drug interactions: No significant drug interactions have been reported in clinical use. However, given Semax's effects on dopaminergic and serotonergic systems, caution may be warranted when combining it with dopaminergic drugs (L-DOPA, dopamine agonists), serotonergic medications (SSRIs, MAOIs), or other nootropics that affect the same neurotransmitter systems.
Contraindications and Drug Interactions
While Semax has a favorable safety profile, certain situations warrant caution or may represent contraindications based on its pharmacological properties:
Potential Contraindications
- Active seizure disorders: Because Semax increases neuronal excitability and enhances synaptic transmission, it could theoretically lower the seizure threshold in individuals with epilepsy or other seizure disorders. While seizures have not been reported in clinical use, this remains a theoretical concern that warrants caution.
- Manic episodes in bipolar disorder: Semax's dopaminergic activation could theoretically exacerbate manic symptoms in individuals with bipolar disorder. Dopamine dysregulation is implicated in the pathophysiology of mania, and compounds that increase dopaminergic activity should be used cautiously in this population.
- Active bleeding or hemorrhagic stroke: Semax's approved use is for ischemic (not hemorrhagic) stroke. The peptide's vascular effects and immunomodulatory properties make its safety in the context of active intracranial hemorrhage uncertain.
- Known hypersensitivity: While allergic reactions to Semax are extremely rare, individuals with known sensitivity to ACTH-related peptides or any components of the formulation should avoid use.
Drug Interactions to Consider
No formal drug interaction studies have been conducted for Semax, but theoretical interactions based on its pharmacological profile include:
- Serotonergic medications (SSRIs, SNRIs, triptans, MAOIs): Semax increases serotonin metabolite levels. Combined with medications that also increase serotonergic activity, there is a theoretical risk of serotonin syndrome, though this has not been reported. Monitor for symptoms including agitation, confusion, rapid heart rate, and hyperthermia.
- Dopaminergic medications (L-DOPA, dopamine agonists, stimulants): Semax enhances dopamine release and turnover. Combined with other dopaminergic agents, there could be additive effects that increase the risk of dopamine-related side effects (agitation, insomnia, psychotic symptoms in susceptible individuals).
- Anticoagulants: While Semax doesn't have direct anticoagulant properties, its immunomodulatory effects could theoretically influence inflammatory mediators that interact with the coagulation cascade. Use caution in patients on warfarin, heparin, or novel oral anticoagulants.
- Antidiabetic medications: Given the mild blood glucose elevation reported in some diabetic patients, Semax could potentially affect glycemic control. Patients on insulin or oral hypoglycemics should monitor blood glucose more frequently when starting Semax.
- Other nootropics: Combining Semax with other nootropic compounds that affect similar neurotransmitter systems (racetams, modafinil, phenylpiracetam) could produce additive effects that may be desirable or excessive depending on individual sensitivity.
Monitoring Recommendations During Semax Use
For practitioners supervising Semax use, the following monitoring recommendations can help ensure safety and optimize outcomes:
| Parameter | Baseline | During Use | Rationale |
|---|---|---|---|
| Blood glucose | Before starting | Weekly (diabetics) | Mild glucose elevation reported in ~7.4% of diabetics |
| Blood pressure | Before starting | Weekly | Vascular effects possible |
| Nasal mucosa inspection | Before starting | Monthly | Discoloration reported in ~10% of users |
| Mood and sleep assessment | Before starting | Daily (self-report) | Dopaminergic effects may affect mood/sleep |
| Cognitive testing (optional) | Before starting | Before/after each cycle | Objective tracking of nootropic effects |
| Serum BDNF (optional) | Before starting | During/after cycle | Confirm biological response |
Long-Term Safety Considerations
The long-term safety of continuous Semax use has not been formally studied through the multi-year follow-up studies that Western regulatory agencies typically require. The cycling protocols recommended in clinical practice (10-14 day courses with intervening breaks) may mitigate potential long-term concerns by limiting cumulative exposure.
Several theoretical concerns have been raised about long-term use, though none has been confirmed by clinical evidence:
- Melanocortin system desensitization: Chronic stimulation of melanocortin receptors could theoretically lead to receptor downregulation. Cycling is the primary strategy to prevent this.
- BDNF overstimulation: While BDNF is generally protective, some preclinical research suggests that chronically elevated BDNF could potentially promote certain types of neural plasticity that aren't always beneficial (for example, in epilepsy-prone individuals). This remains speculative and hasn't been observed with Semax in clinical populations.
- Hair color changes: There have been scattered anecdotal reports (unconfirmed by clinical studies) of hair darkening with long-term melanocortin receptor activation. This is biologically plausible given the role of MC1R in melanin production but has not been documented in the medical literature for Semax specifically.
Safety Summary
Based on nearly three decades of clinical use in Russia and Ukraine, plus extensive preclinical research, Semax appears to have a favorable safety profile with minimal reported side effects. The most common issues are local nasal irritation and mild, self-limiting systemic effects. However, formal long-term safety studies meeting Western regulatory standards are lacking, and the peptide should be used with appropriate caution, particularly in special populations. As with any bioactive compound, consulting a qualified healthcare professional before use is strongly recommended.
Figure 8: Semax safety profile overview. Most reported side effects are mild and related to intranasal delivery. No serious adverse events have been documented in clinical or preclinical research.
Research Methodology and Evidence Quality Assessment
Understanding the Evidence Base
Any comprehensive review of Semax must address the quality and limitations of its evidence base with intellectual honesty. While the totality of the data paints a consistent picture of a safe and effective neuropeptide, the evidence varies significantly in quality across different claims and applications. Understanding these distinctions helps researchers, clinicians, and informed consumers make better decisions about Semax use.
The evidence supporting Semax can be organized into several tiers based on methodological rigor:
Tier 1: Strong Evidence (Well-Replicated Preclinical Data)
The strongest evidence for Semax relates to its molecular mechanisms. The upregulation of BDNF and TrkB in the hippocampus has been demonstrated across multiple independent laboratories using different animal models and experimental designs. The dopaminergic and serotonergic activation data comes from rigorous microdialysis studies with appropriate controls. And the gene expression changes following ischemia have been documented through multiple genome-wide transcriptomic analyses using modern RNA-sequencing technology. These findings are well-replicated, internally consistent, and published in reputable peer-reviewed journals.
Tier 2: Moderate Evidence (Clinical Trials, Primarily Russian)
The clinical trial data for Semax in stroke treatment, optic nerve disease, and cognitive disorders constitutes moderate-quality evidence. These studies generally include control groups and standardized outcome measures, but they often fall short of the methodological standards expected by Western regulatory agencies. Common limitations include relatively small sample sizes (typically 50-200 patients), single-center design, potential for language-related publication bias (many studies are published only in Russian journals), and absence of double-blind placebo-controlled designs in some studies.
That said, the clinical data is consistent across multiple studies and aligns with the strong preclinical mechanistic data. The fact that Semax has maintained its approved status in Russia for over 30 years, with continued clinical use and positive outcomes, provides a form of real-world evidence that complements the formal trial data.
Tier 3: Limited Evidence (General Cognitive Enhancement)
The evidence for Semax as a general cognitive enhancer in healthy individuals is more limited. While the mechanistic rationale is sound (BDNF upregulation, monoamine modulation) and user reports are generally positive, large-scale placebo-controlled trials in healthy populations have not been conducted. The cognitive enhancement claims are supported by extrapolation from mechanism-of-action data, small clinical studies in specific populations (students, military personnel, elderly patients), and accumulated clinical experience in Russian practice.
Tier 4: Preliminary/Theoretical (Emerging Applications)
Evidence for newer proposed applications, including Alzheimer's disease, Parkinson's disease, ADHD, and depression, is primarily theoretical. These applications are supported by mechanistic overlap between Semax's known actions and the pathophysiology of these conditions, but dedicated clinical trials have not been conducted for most of them.
Key Limitations in Current Research
Several limitations in the current Semax evidence base should be acknowledged:
- Western regulatory-grade trials absent: No Phase III clinical trials meeting FDA or EMA standards have been conducted for any indication. The clinical data comes primarily from Russian and Ukrainian institutions, which operate under different regulatory frameworks.
- Publication bias concerns: Studies with positive results are more likely to be published, and this bias may be amplified in the Russian medical literature, where negative results may face additional barriers to publication.
- Translation gaps: Many relevant studies are published exclusively in Russian-language journals, limiting accessibility for international researchers and making independent verification more difficult.
- Placebo controls: While some clinical studies included control groups, rigorous double-blind placebo-controlled designs are not universal in the published literature.
- Long-term data: Despite decades of clinical use, formal long-term safety and efficacy follow-up studies are limited. Most clinical trials evaluated outcomes over weeks to months, not years.
- Dose optimization: Systematic dose-finding studies using modern pharmacokinetic/pharmacodynamic methodology are lacking for most indications.
Future Research Priorities
To advance the evidence base for Semax and potentially support broader regulatory approval, several types of studies would be valuable:
- Multi-center randomized controlled trials: Well-powered, double-blind, placebo-controlled trials for stroke recovery and cognitive enhancement conducted at multiple centers across different countries would provide the strongest possible evidence for Semax's clinical utility.
- Pharmacokinetic studies in humans: Detailed characterization of Semax absorption, distribution, metabolism, and excretion in humans using modern analytical techniques would improve dosing optimization and safety assessment.
- Head-to-head comparisons: Trials comparing Semax with established therapies (e.g., cholinesterase inhibitors for cognitive decline, established neuroprotective agents for stroke) would clarify its relative efficacy and position in treatment algorithms.
- Biomarker studies: Studies measuring BDNF levels, neurotransmitter metabolites, and other biomarkers in humans receiving Semax would bridge the gap between preclinical mechanism data and clinical outcomes.
- Long-term safety registries: Prospective registries tracking long-term outcomes in Semax users would address the most significant remaining safety questions.
- Combination therapy studies: Formal evaluation of Semax in combination with other peptides (particularly Selank) and conventional medications would establish evidence-based stacking protocols.
The field of peptide research is evolving rapidly, and increased interest in neuroprotective peptides from Western research institutions may lead to the kind of rigorous studies needed to fill these evidence gaps. For the latest developments in peptide science, the FormBlends Science & Research section provides ongoing updates.
Figure 11: Evidence quality tiers for Semax research. The strongest evidence supports molecular mechanisms, while clinical applications have moderate evidence, and emerging applications remain largely theoretical.
Frequently Asked Questions
What exactly is Semax, and how was it developed?
Semax is a synthetic peptide consisting of seven amino acids (Met-Glu-His-Phe-Pro-Gly-Pro) designed as an analog of the ACTH(4-10) hormone fragment. It was developed at the Institute of Molecular Genetics of the Russian Academy of Sciences beginning in the 1980s. The researchers took the biologically active core of ACTH, which had demonstrated cognitive effects in animal studies, and added a Pro-Gly-Pro tripeptide tail to protect the molecule from rapid enzymatic breakdown. This gave the peptide enough metabolic stability for intranasal delivery while preserving its ability to enhance learning, memory, and neuroprotection. Semax received regulatory approval in Russia in 1994 and has been used clinically there for over 30 years.
How does Semax work in the brain?
Semax works through multiple simultaneous mechanisms. Its most well-documented effect is upregulating brain-derived neurotrophic factor (BDNF) and its receptor TrkB in the hippocampus, a brain region critical for memory formation. It also increases nerve growth factor (NGF) expression. Beyond neurotrophic effects, Semax activates dopaminergic systems (enhancing focus and motivation) and serotonergic systems (supporting mood and impulse control). In the context of brain injury, it suppresses pro-inflammatory gene expression while activating genes involved in neurotransmission and synaptic function. This multi-target pharmacology, affecting neurotrophins, monoamines, inflammation, and gene expression simultaneously, gives Semax a broader profile than most single-mechanism nootropics.
How much does Semax increase BDNF levels?
In preclinical studies, a single intranasal application of Semax at 50 mcg/kg body weight produced a 1.4-fold increase in BDNF protein levels and a 3-fold increase in exon III BDNF mRNA levels in the rat hippocampus. It also increased TrkB (the BDNF receptor) mRNA by 2-fold and TrkB tyrosine phosphorylation by 1.6-fold. In clinical settings, Semax administration to stroke patients significantly elevated plasma BDNF levels, with the elevation sustained throughout the treatment period. These are substantial increases. For comparison, regular aerobic exercise, one of the most reliable natural BDNF boosters, typically increases serum BDNF by 20-30% over weeks of consistent training. Semax produces comparable or greater changes within hours.
Can Semax help with stroke recovery?
Semax is approved in Russia for the treatment of ischemic stroke, and clinical trial data supports its use in this context. In a study of 110 stroke patients, Semax administration increased plasma BDNF levels, and when combined with early rehabilitation, it produced faster functional recovery and improved motor performance compared to controls. The Russian clinical protocol uses high-dose Semax (1% nasal solution, 12,000-18,000 mcg/day) during the acute phase, tapering to lower doses during rehabilitation. Preclinical studies show Semax reduces infarct volume by approximately 33%, improves neurological deficit scores, and reverses the gene expression changes caused by ischemia. It's considered complementary to standard stroke treatments, not a replacement for them.
What's the difference between Semax and Selank?
While both are Russian-developed peptides administered intranasally, they target different aspects of brain function. Semax is classified as a nootropic (cognitive enhancer) that works primarily through BDNF upregulation and dopamine modulation. It sharpens focus, speeds learning, and enhances motivation. Selank, derived from the immunomodulatory peptide tuftsin, is classified as an anxiolytic that modulates GABA receptors and serotonin signaling. It reduces anxiety and promotes emotional stability without the sedation or dependence risks of benzodiazepines. They complement each other well: Semax provides the focus, while Selank removes the anxiety that can impair cognitive performance. Russian clinics have prescribed them together without adverse interactions.
Is nasal spray or injection better for Semax?
Intranasal administration is the preferred and most-studied route for Semax. It offers several advantages: direct nose-to-brain transport via olfactory and trigeminal nerve pathways, rapid onset (15-20 minutes), non-invasive administration, and minimal systemic exposure. All clinical trial data is based on intranasal delivery. Subcutaneous injection is sometimes used but lacks formal clinical evaluation. When injected, Semax enters the systemic circulation and must cross the blood-brain barrier to reach the brain, which may reduce central nervous system bioavailability compared to intranasal delivery. Unless there are specific reasons to use injection (such as nasal pathology preventing intranasal use), the nasal route should be prioritized.
What is the recommended dosing protocol for cognitive enhancement?
For general cognitive enhancement, the standard protocol is 200-600 mcg per day administered intranasally, divided into 2-3 doses. Morning and early afternoon timing is recommended to avoid potential interference with sleep. The most common cycling protocol is 10 days on, 14 days off. For periods of intense mental demand, doses up to 900 mcg/day for 3-5 days have been used, followed by an equal rest period. If using N-Acetyl Semax Amidate, lower doses (100-300 mcg/day) are appropriate due to its enhanced stability and longer duration. Always start at the lower end of the dosing range and adjust based on individual response. The FormBlends Dosing Calculator can help determine an appropriate starting point.
What are the side effects of Semax?
Semax is considered well-tolerated based on nearly 30 years of clinical use. The most common side effects are related to nasal administration: dryness, mild burning, or irritation in the nasal cavity, affecting roughly 15-20% of users. These are typically mild and self-limiting. Approximately 10% of users report reversible nasal mucosa discoloration. Uncommon side effects include headache (5-8%), dizziness (2-3%), and occasional sleep disturbance when used late in the day. About 7.4% of diabetic patients in clinical observations showed mild blood glucose increases. No serious adverse events, organ toxicity, addiction, tolerance, or withdrawal symptoms have been reported in clinical or preclinical studies. The compound has not been evaluated during pregnancy.
What is N-Acetyl Semax Amidate, and how does it differ from standard Semax?
N-Acetyl Semax Amidate (NASA) is a chemically modified version of Semax with two protective modifications: an acetyl group added to the N-terminus and an amide group replacing the C-terminal carboxyl group. These modifications protect the peptide from enzymatic degradation at both ends, extending its effective duration from 2-4 hours (standard Semax) to approximately 6-12 hours. This means fewer daily doses are needed, and effects are more sustained. The trade-off is that virtually all clinical research has been conducted with standard Semax, so the clinical evidence base for NASA is more limited. Dosing is typically lower (100-300 mcg/day) due to the enhanced bioavailability.
Can Semax be combined with other peptides?
Yes, Semax is commonly combined with other peptides in clinical and research settings. The most established combination is Semax plus Selank, which has been used in Russian clinics without reported adverse interactions. Other popular research combinations include Semax with BPC-157 for neuroprotection and tissue repair, or with NAD+ for cellular energy support. When stacking, the general principle is to combine compounds that work through different mechanisms rather than redundant pathways. Start with one compound, establish your baseline response, and add others individually. Most practitioners recommend limiting simultaneous peptides to 2-3 to maintain the ability to identify which compounds are providing benefit.
Is Semax legal and is it FDA-approved?
Semax is not FDA-approved and is not regulated as a pharmaceutical drug in the United States, European Union, or most Western countries. It is, however, approved in Russia and Ukraine for several neurological indications including ischemic stroke, optic nerve atrophy, and cognitive disorders. In the US, Semax is available as a research peptide. It is not a controlled substance and is not scheduled under the Controlled Substances Act. The regulatory status varies by country, and researchers should confirm the applicable regulations in their jurisdiction. The fact that Semax lacks FDA approval reflects the challenges of international pharmaceutical regulation rather than specific safety concerns, as the peptide has maintained a favorable safety record through decades of clinical use in Russia.
How quickly does Semax take effect?
When administered intranasally, Semax typically produces noticeable cognitive effects within 15-20 minutes. This rapid onset is due to nose-to-brain transport, where the peptide reaches central nervous system targets through olfactory and trigeminal nerve pathways without needing to cross the blood-brain barrier from the bloodstream. The acute cognitive effects (improved focus, mental clarity) last approximately 2-4 hours for standard Semax and 6-12 hours for N-Acetyl Semax Amidate. However, the neurotrophic effects, particularly BDNF upregulation, occur on a longer timescale: BDNF mRNA increases within hours but protein levels peak at around 24 hours and may have lasting effects on synaptic plasticity that extend well beyond the acute dosing period.
How should Semax be stored?
Proper storage is essential for maintaining Semax's potency. Lyophilized (freeze-dried) Semax powder should be stored at -20 degrees Celsius (standard freezer temperature) and can remain stable for years under these conditions. Once reconstituted into a nasal solution, it should be refrigerated at 2-8 degrees Celsius (standard refrigerator temperature) and used within 30 days. Avoid exposure to heat, direct sunlight, and repeated freeze-thaw cycles, which can degrade the peptide. Premade nasal spray solutions from compounding pharmacies typically include preservatives (like benzalkonium chloride) that extend shelf life somewhat, but refrigeration is still recommended. N-Acetyl Semax Amidate and Adamax have improved temperature stability compared to standard Semax but should still be stored properly for optimal potency.
Does Semax cause tolerance or dependence?
Based on available clinical data and decades of use in Russia, Semax does not appear to cause physical dependence or classic pharmacological tolerance. Users don't report needing progressively higher doses to achieve the same effect, and no withdrawal symptoms have been documented upon discontinuation. However, the cycling protocols commonly recommended (10-14 days on, followed by 14+ days off) are designed in part to prevent potential receptor desensitization. The underlying concern is that sustained melanocortin receptor stimulation and chronic BDNF elevation could theoretically lead to compensatory downregulation over time. While this hasn't been formally documented with Semax, cycling is considered a prudent precaution that aligns with how other receptor-active compounds are used clinically.
Can I combine Semax with exercise for enhanced BDNF benefits?
Yes, combining Semax with exercise is a strategy supported by the underlying biology. Both Semax and aerobic exercise independently increase BDNF levels, and their mechanisms are partially distinct. Exercise elevates BDNF through increased neuronal activity, lactate signaling, and FNDC5/irisin pathways, while Semax works through melanocortin receptor activation and direct gene expression changes. Administering Semax 15-20 minutes before aerobic exercise could theoretically produce additive or even greater-than-additive BDNF elevation, though this specific combination hasn't been tested in controlled studies. From a practical standpoint, morning exercise combined with morning Semax dosing aligns both interventions with the brain's peak plasticity window.
Is Semax safe for women? Are there any gender-specific considerations?
Semax has been used by both men and women in clinical practice without documented gender-specific adverse effects. The peptide does not have direct hormonal activity at nootropic doses, as it lacks the C-terminal portion of ACTH responsible for adrenal cortisol stimulation. However, women should be aware of a few considerations. The melanocortin system interacts with reproductive hormones, and some women report that Semax's effects feel different at different points in their menstrual cycle, possibly due to estrogen's influence on dopamine sensitivity. During pregnancy and breastfeeding, Semax should be avoided due to the absence of safety data in these populations. There is no evidence suggesting different dosing requirements for women, though starting at the lower end of the range is always a reasonable approach for any new user regardless of gender.
Can I take Semax with coffee or caffeine?
Yes, Semax and caffeine can generally be combined, and many users do so without issues. Both compounds enhance alertness and focus, but through different mechanisms. Caffeine blocks adenosine receptors (reducing the signal for drowsiness) and increases catecholamine release, while Semax modulates BDNF expression and enhances dopaminergic transmission through melanocortin receptor signaling. The combination may produce additive focus-enhancing effects. However, some individuals may find the combination overstimulating, particularly at higher doses of either compound. If you experience jitteriness, anxiety, or racing thoughts when combining Semax with your usual caffeine intake, try reducing the caffeine dose. Starting with a lower dose of Semax on your first combination day is prudent. Timing-wise, taking both in the morning aligns with circadian physiology.
Can Semax affect hair color or hair growth?
There have been scattered anecdotal reports of hair darkening among Semax users, which is biologically plausible given the melanocortin system's role in melanin production. The MC1R receptor, which is present in hair follicles, regulates melanin synthesis. ACTH-derived peptides can activate MC1R, potentially increasing melanin production in hair. However, this effect has not been documented in the medical literature for Semax specifically, and the doses used for cognitive enhancement may be too low to produce significant melanogenic effects. Hair loss has not been reported as a side effect. If anything, the neurotrophic effects of BDNF, which supports hair follicle health, could theoretically have mild positive effects on hair, though this remains speculative.
Advanced Topics: Semax in the Future of Neurological Medicine
Personalized Dosing and Pharmacogenomics
One of the frontiers in Semax research is the concept of personalized dosing based on individual genetic and biochemical profiles. The field of pharmacogenomics studies how genetic variation affects drug response, and while specific pharmacogenomic data for Semax is limited, the general principles can be applied to optimize its use.
Several factors likely influence individual response to Semax:
- Baseline BDNF levels: Individuals with naturally lower BDNF levels (due to genetics, age, depression, sedentary lifestyle, or chronic stress) may experience more dramatic benefits from Semax. Conversely, individuals with already-optimized BDNF signaling may notice more subtle effects. Measuring baseline serum BDNF before starting Semax and tracking changes over time could help calibrate expectations and dosing.
- BDNF Val66Met polymorphism: The most studied genetic variant affecting BDNF function is the Val66Met polymorphism (rs6265). The Met allele, carried by approximately 20-30% of the population, results in reduced activity-dependent BDNF secretion and has been associated with impaired cognitive function and increased susceptibility to mood disorders. Individuals carrying the Met allele might theoretically benefit more from Semax's BDNF-enhancing effects, though this hypothesis hasn't been directly tested.
- Melanocortin receptor variants: Genetic polymorphisms in MC4R and other melanocortin receptors could affect Semax's binding affinity and downstream signaling. Individuals with certain MC4R variants may be more or less responsive to melanocortin-based drugs.
- Dopamine system genetics: Variations in dopamine-related genes (COMT Val158Met, DAT1, DRD2, DRD4) significantly affect baseline dopamine signaling. Since Semax modulates dopamine release and turnover, these genetic variants could influence the cognitive enhancement response. For example, individuals with the COMT Val/Val genotype (associated with faster dopamine breakdown in the prefrontal cortex) might experience different effects than those with the Met/Met genotype.
- Peptidase activity: Individual variation in tissue peptidase activity affects how quickly Semax is degraded, influencing both the effective half-life and the rate of metabolite generation. Some individuals may metabolize Semax faster than others, requiring higher doses or more frequent administration.
While we're not yet at the point where pharmacogenomic testing can guide Semax dosing decisions, the principle of individual variation in response is well-established. This is why starting at the lower end of the dosing range and titrating based on personal response remains the most practical approach to optimization. The FormBlends Free Assessment can help individuals understand their baseline profile and identify starting parameters that account for individual variation.
Drug Delivery Innovations
While the current intranasal delivery method for Semax is effective, ongoing research into advanced drug delivery systems could further improve its pharmacokinetics and clinical utility. Several approaches are being explored in the broader peptide delivery field that could be applied to Semax:
- Nanoparticle encapsulation: Encapsulating Semax in polymeric or lipid nanoparticles could protect the peptide from enzymatic degradation, extend its release profile, and improve targeting to specific brain regions. Chitosan nanoparticles, in particular, have shown promise for intranasal peptide delivery due to their mucoadhesive properties and ability to transiently open tight junctions in the nasal epithelium, enhancing absorption.
- Mucoadhesive formulations: Adding mucoadhesive polymers (like carbopol, hydroxypropyl methylcellulose, or chitosan) to Semax nasal formulations could increase the contact time between the peptide and the absorptive nasal mucosa, improving bioavailability. This is particularly relevant for standard Semax, which has a relatively short half-life.
- Controlled-release implants: For chronic neurological conditions requiring sustained Semax delivery, implantable controlled-release devices could provide steady peptide levels without the need for multiple daily nasal administrations. This approach is highly theoretical for Semax but has been explored for other peptides.
- Dry powder inhalers: Nasal dry powder formulations could offer improved stability compared to liquid solutions, eliminating the need for refrigeration and extending shelf life. This would make Semax more practical for use in settings without reliable cold chain access.
- Transdermal patches: While peptide delivery through the skin is challenging due to the skin's barrier function, advances in microneedle patch technology and chemical permeation enhancers are making transdermal peptide delivery increasingly feasible. A Semax patch could provide sustained, controlled delivery without the need for nasal or injectable administration.
These delivery innovations are at various stages of development for the broader peptide therapeutic class. Their application to Semax specifically would require dedicated formulation studies and stability testing. For now, the intranasal spray remains the most practical and well-characterized delivery method.
Formal Combination Therapy Research
The informal stacking protocols discussed earlier in this report are based on pharmacological rationale and clinical experience, but formal research into optimized combination therapies could significantly advance the field. Several combinations warrant formal clinical evaluation:
Semax + Selank: The Russian Clinical Standard
This is the combination with the most real-world clinical experience, having been prescribed together in Russian medical practice for years. A formal randomized controlled trial comparing Semax alone, Selank alone, and the combination against placebo for conditions like post-stroke cognitive recovery or vascular cognitive impairment would provide definitive evidence for the value of this combination. The distinct mechanisms (melanocortin/BDNF vs. GABA/enkephalin) suggest truly additive benefits rather than redundancy.
Semax + Physical Rehabilitation
The clinical data showing that early rehabilitation combined with Semax produced superior outcomes in stroke patients deserves further investigation. Specific questions include: What types of rehabilitation exercises (motor, cognitive, combined) pair best with Semax? What is the optimal timing of Semax administration relative to rehabilitation sessions? Can Semax enhance the neuroplastic changes driven by rehabilitation training?
Semax + Conventional Nootropics
How Semax interacts with established cognitive-enhancing medications, such as racetams, modafinil, cholinesterase inhibitors, and memantine, is largely unexplored. Given the growing trend toward polypharmacy in cognitive optimization, evidence-based guidance on these combinations would be clinically valuable. For instance, combining Semax (which upregulates BDNF and enhances monoamine signaling) with a cholinesterase inhibitor (which enhances cholinergic transmission) could theoretically provide complementary cognitive benefits through distinct neurotransmitter systems.
Semax + Other Neuroprotective Peptides
Combinations of Semax with other neuroprotective peptides like BPC-157, Dihexa, and GHK-Cu target different aspects of neural repair and could produce additive neuroprotection. BPC-157 works through nitric oxide modulation and growth factor upregulation, Dihexa targets the HGF/c-Met pathway, and GHK-Cu modulates gene expression patterns related to tissue repair. Each of these mechanisms is distinct from Semax's primary BDNF/melanocortin pathway, suggesting minimal pharmacological redundancy.
Biomarker-Guided Therapy
The future of Semax therapy may involve biomarker-guided treatment decisions, where objective measurements guide dosing, cycling, and combination choices. Several biomarkers are relevant:
- Serum BDNF: Blood BDNF levels can be measured through standard clinical laboratory tests. Tracking BDNF before, during, and after Semax cycles could confirm biological response and help optimize dosing. Target levels and the relationship between serum BDNF and brain BDNF are still being characterized, but the direction of change (increase from baseline) is a useful indicator of biological activity.
- Dopamine and serotonin metabolites: Urinary and blood levels of dopamine metabolites (homovanillic acid/HVA) and serotonin metabolites (5-HIAA) could provide indirect evidence of Semax's neurotransmitter-modulating effects in individual patients.
- Inflammatory markers: For neuroprotective applications, measuring inflammatory markers like C-reactive protein (CRP), IL-6, and TNF-alpha could help assess Semax's anti-inflammatory effects at the systemic level.
- Neuroimaging: Functional MRI (fMRI) and PET scanning could provide direct evidence of Semax's effects on brain activity, connectivity, and neurotransmitter system function. While these tools are too expensive for routine clinical use, they could be invaluable in research settings for understanding how Semax changes brain function in real-time.
- EEG biomarkers: As discussed earlier, Semax produces measurable changes in EEG patterns, including increased alpha power and enhanced P300 amplitude. Quantitative EEG (qEEG) could serve as a relatively affordable and accessible biomarker for tracking cognitive response to Semax.
The integration of biomarker data with clinical assessment would represent a significant advance in how Semax and similar neuropeptides are used therapeutically. Rather than applying one-size-fits-all dosing protocols, clinicians could tailor treatment to individual biological responses, adjusting doses, cycle lengths, and combinations based on objective measurements.
Regulatory Future: Could Semax Get FDA Approval?
The question of whether Semax could ever receive FDA approval in the United States is frequently asked in the nootropic community. The answer involves both scientific and economic considerations.
From a scientific perspective, the existing evidence base for Semax is substantial but doesn't meet the specific requirements of the FDA approval process. An NDA (New Drug Application) would require at least two adequate and well-controlled Phase III clinical trials demonstrating safety and efficacy for a specific indication. These trials would need to be conducted under Good Clinical Practice (GCP) guidelines, with proper randomization, blinding, and predetermined endpoints. While the Russian clinical data is supportive, it would likely need to be supplemented with new trials meeting FDA standards.
From an economic perspective, the barrier is even higher. FDA approval costs typically run into hundreds of millions of dollars, and the return on investment depends on the ability to protect the drug with patents. Semax, as a simple heptapeptide with a structure published in the 1990s, is difficult to patent in its current form. Without patent protection, any company that invested in FDA approval would face generic competition shortly after approval, making it difficult to recoup development costs.
There are potential workarounds. Modified versions of Semax (like N-Acetyl Semax Amidate or novel formulations) might be patentable, providing a commercial incentive for development. Alternatively, the 505(b)(2) regulatory pathway, which allows reference to published literature as part of the approval package, could reduce the clinical development burden. Orphan drug designation for rare neurological conditions could provide additional incentives.
The most likely near-term regulatory path for Semax in Western markets may be through compounding pharmacies, which can prepare custom formulations of unapproved substances under specific regulatory frameworks. Some US compounding pharmacies already offer Semax as a compounded prescription medication, though the regulatory landscape for compounded peptides is evolving and varies by state.
Next-Generation Semax Analogs in Development
Research into improved analogs of Semax continues, driven by the desire to enhance its pharmacological properties while maintaining its favorable safety profile. Several design strategies are being explored:
- D-amino acid substitutions: Replacing specific L-amino acids with their D-isomers can dramatically increase peptide stability without always disrupting receptor binding. D-amino acids are resistant to most peptidases because they don't fit the enzyme's active site. However, D-amino acid-containing peptides can have altered receptor pharmacology, so each substitution needs to be individually optimized.
- PEGylation: Attaching polyethylene glycol (PEG) chains to peptides increases their molecular weight and hydrodynamic radius, reducing renal clearance and increasing half-life. PEGylated Semax analogs could potentially be administered less frequently while maintaining therapeutic levels.
- Cyclization: Converting linear peptides into cyclic structures through head-to-tail cyclization or side-chain crosslinks increases stability and can improve receptor selectivity. Cyclic Semax analogs might retain nootropic activity while gaining resistance to enzymatic degradation.
- Lipidation: Adding fatty acid chains to Semax could enhance its ability to cross the blood-brain barrier from the bloodstream, potentially making subcutaneous or oral administration more effective. Lipidated peptides also tend to have extended half-lives due to albumin binding in the bloodstream.
- Fusion peptides: Combining the active elements of Semax with those of complementary peptides (like Selank) into a single chimeric molecule could create dual-action compounds that provide both nootropic and anxiolytic effects from a single agent.
These design approaches represent the cutting edge of peptide medicinal chemistry. While none has yet produced a successor that clearly surpasses standard Semax in clinical utility, the tools and knowledge base for rational peptide design continue to advance rapidly. The next generation of ACTH-derived nootropic peptides may combine the established efficacy of Semax with improved pharmacokinetic properties that make them more practical for clinical use.
AI-Driven Peptide Design
The application of artificial intelligence and machine learning to peptide drug design is a transformative development that could accelerate the discovery of improved Semax analogs. AI tools can predict peptide-receptor binding affinity, metabolic stability, blood-brain barrier permeability, and toxicity from sequence data alone, dramatically reducing the need for costly trial-and-error experimental screening.
Several AI-driven approaches are relevant to Semax analog development:
- Structure-activity relationship (SAR) modeling: Machine learning models trained on libraries of ACTH fragment analogs with known biological activities can predict which sequence modifications will preserve nootropic activity while enhancing stability or potency.
- Molecular dynamics simulations: AI-enhanced molecular dynamics can predict how modified Semax analogs interact with melanocortin receptors at the atomic level, identifying binding poses and key interactions that drive biological activity.
- Generative models: AI models can generate novel peptide sequences optimized for specific properties (e.g., high MC4R affinity, high metabolic stability, low MC2R affinity), exploring design space that would be impractical to search experimentally.
- Pharmacokinetic prediction: AI models can estimate absorption, distribution, metabolism, and excretion properties from peptide structure, helping prioritize candidates for experimental validation.
The convergence of established Semax research data with modern AI tools creates a unique opportunity to design optimized next-generation nootropic peptides. Machine learning models trained on the extensive ACTH fragment SAR database can identify promising candidate sequences that human intuition alone might miss. And the rapid advancement of protein structure prediction tools (like AlphaFold and its successors) enables accurate modeling of peptide-receptor interactions without the need for expensive experimental crystallography. These computational approaches can screen millions of potential analog sequences in silico, identifying a shortlist of candidates that merit experimental validation. The result is a dramatically accelerated design cycle that could compress years of traditional medicinal chemistry into months. The established mechanism-of-action data for Semax provides clear design targets, and AI can efficiently explore the vast chemical space of possible modifications to identify candidates most likely to improve on the original.
Comprehensive Dosing Reference Guide
Dosing Protocols by Application
This section consolidates all dosing information into a single reference, organized by intended application. All doses refer to standard Semax unless otherwise noted. These protocols are based on published clinical data, manufacturer guidelines, and clinical experience. They should not be considered medical advice - always consult with a qualified healthcare provider before implementing any peptide protocol.
| Application | Daily Dose | Route | Frequency | Cycle Length | Evidence Level |
|---|---|---|---|---|---|
| General cognitive enhancement | 200-600 mcg | Intranasal | 2-3x daily | 10 on / 14 off | Moderate |
| Intensive cognitive demand | 400-900 mcg | Intranasal | 2-3x daily | 3-5 days | Moderate |
| Ischemic stroke (acute) | 12,000-18,000 mcg | Intranasal (1%) | 3-4x daily | 5-10 days | Strong (Russian data) |
| Stroke recovery | 600-3,000 mcg | Intranasal | 2-3x daily | 14-21 days | Moderate |
| Dyscirculatory encephalopathy | 200-600 mcg | Intranasal (0.1%) | 3x daily | 10-14 days | Moderate |
| Optic nerve disease | 200-600 mcg | Intranasal/electrophoresis | 2-3x daily | 7-10 days | Moderate |
| Age-related cognitive decline | 200-400 mcg | Intranasal | 2-3x daily | 10-14 days, repeating | Limited |
| Stress adaptation | 200-600 mcg | Intranasal | 2-3x daily | 5-10 days | Limited |
N-Acetyl Semax Amidate Dosing Adjustments
When using N-Acetyl Semax Amidate instead of standard Semax, dosing adjustments are necessary due to the enhanced stability and longer duration of action:
| Standard Semax Dose | NA-Semax Amidate Equivalent | Rationale |
|---|---|---|
| 200 mcg 3x daily | 100-150 mcg 1-2x daily | Extended half-life reduces dosing frequency |
| 400 mcg 3x daily | 200-300 mcg 1-2x daily | Enhanced bioavailability allows lower doses |
| 600 mcg 3x daily | 300-400 mcg 1-2x daily | Higher doses may not be needed with better stability |
These equivalences are approximate and based on the estimated 2-3x difference in effective half-life between the formulations. Individual response may vary, and starting at the lower end of the range is always recommended when transitioning between formulations.
Real-World Use Patterns and Practitioner Observations
While controlled clinical trials provide the most rigorous evidence, observations from practitioners who prescribe Semax in clinical settings add practical context that helps bridge the gap between research data and real-world application. These patterns, while not constituting formal evidence, can help guide expectations and protocol design.
Common Response Patterns
Practitioners who work with Semax regularly have noted several consistent patterns in patient responses:
- The "clarity window": Many patients report a distinctive window of enhanced mental clarity that begins approximately 15-20 minutes after nasal administration and persists for 2-4 hours. During this period, patients describe feeling more organized in their thinking, better able to articulate ideas, and more efficient at complex tasks. This window aligns with the expected pharmacokinetic profile of standard Semax.
- Cumulative vs. acute effects: Some cognitive benefits appear immediately (within the first dose), while others develop over the course of a 10-14 day cycle. Acute effects tend to be more noticeable for focus and processing speed, while memory improvements and emotional regulation changes often become apparent only after several days of consistent use. This distinction aligns with the different timescales of Semax's mechanisms: dopamine modulation occurs within minutes, while BDNF-driven neuroplastic changes require days to weeks.
- Individual variation in response: Practitioners consistently note significant individual variation in both the magnitude and character of Semax's effects. Some patients report dramatic improvements in focus and productivity, while others describe more subtle enhancements. A smaller percentage (estimated at 10-15% in clinical observation) report minimal noticeable benefit. Factors that appear to predict stronger response include higher baseline levels of cognitive impairment, chronic stress or burnout, age-related cognitive decline, and sleep deprivation.
- Return to baseline after cycling off: Most patients report that cognitive performance returns to baseline within 3-7 days after discontinuing Semax at the end of a cycle. However, some patients report lasting improvements that persist beyond the immediate pharmacological effect, possibly reflecting BDNF-driven structural changes that outlast the acute peptide exposure.
Practitioner Dosing Preferences
Among practitioners who prescribe Semax regularly, several dosing preferences have emerged:
- Most practitioners start patients at the lower end of the dosing range (200 mcg/day) and titrate upward based on response, rather than starting at the standard 600 mcg dose
- Morning-only dosing is preferred by most practitioners over split dosing, as it simplifies the protocol and reduces the risk of sleep interference
- Practitioners working with brain injury rehabilitation patients tend to use longer cycles (14-21 days) and higher doses than those working with healthy individuals seeking cognitive optimization
- Many practitioners recommend a "loading" protocol for the first cycle (slightly higher dose for the first 3 days followed by standard dosing) to establish initial neurotrophic changes, though this approach is not supported by specific clinical evidence
- Some practitioners have patients take weekends off even during their on-cycle (dosing Monday through Friday only), providing micro-breaks that may help maintain receptor sensitivity
Common Combination Patterns in Clinical Practice
In clinical settings, Semax is rarely used in isolation. Practitioners typically combine it with lifestyle modifications and, in some cases, other therapeutic agents. The most common clinical combinations observed include:
- Semax + targeted cognitive rehabilitation exercises (for post-stroke or post-TBI patients)
- Semax + Selank (for patients with combined cognitive deficits and anxiety)
- Semax + supervised exercise programs (for age-related cognitive decline)
- Semax + micronutrient optimization (magnesium, zinc, omega-3s, B-vitamins)
- Semax + meditation or neurofeedback training (for executive function enhancement)
These combination patterns reflect the understanding that cognitive optimization is inherently multi-modal. No single intervention, whether pharmaceutical, nutraceutical, or behavioral, addresses all the factors that influence brain performance. Semax serves as a pharmacological catalyst within a broader protocol that includes lifestyle, nutritional, and behavioral components.
Reconstitution Calculator
Proper reconstitution is essential for accurate dosing. Here are the most common reconstitution scenarios:
| Vial Size | BAC Water Added | Concentration | Volume for 200 mcg | Volume for 400 mcg | Volume for 600 mcg |
|---|---|---|---|---|---|
| 5 mg | 1.0 mL | 5,000 mcg/mL | 0.04 mL (4 units) | 0.08 mL (8 units) | 0.12 mL (12 units) |
| 5 mg | 2.0 mL | 2,500 mcg/mL | 0.08 mL (8 units) | 0.16 mL (16 units) | 0.24 mL (24 units) |
| 10 mg | 2.0 mL | 5,000 mcg/mL | 0.04 mL (4 units) | 0.08 mL (8 units) | 0.12 mL (12 units) |
| 10 mg | 5.0 mL | 2,000 mcg/mL | 0.10 mL (10 units) | 0.20 mL (20 units) | 0.30 mL (30 units) |
Note: "Units" in this table refer to insulin syringe units (1 unit = 0.01 mL on a standard 100-unit insulin syringe). For nasal spray use, the concentration should be adjusted to deliver the desired dose in the spray volume of your device (typically 0.1 mL per pump).
For personalized dosing calculations, the FormBlends Dosing Calculator provides an interactive tool that accounts for vial size, desired concentration, and target dose per administration.
References
- Ashmarin IP, Nezavibatko VN, Levitskaya NG, et al. Design and investigation of an ACTH(4-10) analogue lacking D-amino acids and possessing nootropic properties. Neurosci Res Commun. 1995;16(2):105-112.
- Ashmarin IP, Myasoedov NF, Lermontova NN, Ponomareva-Stepnaya MA. A nootropic adrenocorticotropin analog 4-10-semax (15 years experience in its design and study). Vestn Ross Akad Med Nauk. 1997;(11):28-33. PMID: 9173745.
- Dolotov OV, Karpenko EA, Inozemtseva LS, et al. Semax, an analogue of adrenocorticotropin (4-10), binds specifically and increases levels of brain-derived neurotrophic factor protein in rat basal forebrain. J Neurochem. 2006;97 Suppl 1:82-86. doi:10.1111/j.1471-4159.2006.03658.x. PMID: 16635254.
- Dolotov OV, Karpenko EA, Grivennikov IA, et al. Semax, an analog of ACTH(4-10) with cognitive effects, regulates BDNF and trkB expression in the rat hippocampus. Brain Res. 2006;1117(1):54-60. doi:10.1016/j.brainres.2006.07.108. PMID: 16996037.
- Eremin KO, Kudrin VS, Grivennikov IA, Myasoedov NF, Rayevsky KS. Semax, an ACTH(4-10) analogue with nootropic properties, activates dopaminergic and serotoninergic brain systems in rodents. Neurochem Res. 2005;30(12):1493-1500. doi:10.1007/s11064-005-8826-8. PMID: 16362768.
- Agapova TYu, Agniullin YV, Silachev DN, et al. Effect of semax on the temporal dynamics of brain-derived neurotrophic factor and nerve growth factor gene expression in the rat hippocampus and frontal cortex. Mol Biol. 2008;42(4):652-657. PMID: 18756821.
- Dolotov OV, Inozemtseva LS, Karpenko EA, et al. Comparison of the temporary dynamics of NGF and BDNF gene expression in rat hippocampus, frontal cortex, and retina under Semax action. Dokl Biol Sci. 2009;427(1):329-332. PMID: 19662538.
- Dmitrieva VG, Povarova OV, Skvortsova VI, et al. The peptide semax affects the expression of genes related to the immune and vascular systems in rat brain focal ischemia: genome-wide transcriptional analysis. BMC Genomics. 2010;11:671. doi:10.1186/1471-2164-11-671.
- Filippenkov IB, Stavchansky VV, Denisova AE, et al. Novel insights into the protective properties of ACTH(4-7)PGP (Semax) peptide at the transcriptome level following cerebral ischaemia-reperfusion in rats. Genes (Basel). 2020;11(6):681. doi:10.3390/genes11060681.
- Filippenkov IB, Remizova JA, Denisova AE, et al. Semax and Pro-Gly-Pro activate the transcription of neurotrophins and their receptor genes after cerebral ischemia. Int J Mol Sci. 2024;25(21):11530. doi:10.3390/ijms252111530.
- Gusev EI, Skvortsova VI. The efficacy of semax in the treatment of patients at different stages of ischemic stroke. Zh Nevrol Psikhiatr Im S S Korsakova. 2018;118(3):61-68. doi:10.17116/jnevro20181183161. PMID: 29798983.
- Ioffe DI, Orekhova OV, Salichov AI. Evaluation of therapeutic effect of new Russian drug semax in optic nerve disease. Vestn Oftalmol. 1999;115(1):15-18. PMID: 10741256.
- Kurysheva NI, Shpak AA, Ioileva EE, et al. Semax in the treatment of glaucomatous optic neuropathy in patients with normalized ophthalmic tone. Vestn Oftalmol. 2001;117(4):5-8. PMID: 11569188.
- Galanth C, Gonzalez K, Bhatt P, et al. Influence of the N-terminus acetylation of Semax, a synthetic analog of ACTH(4-10), on copper(II) and zinc(II) coordination and biological properties. J Inorg Biochem. 2016;165:47-58. doi:10.1016/j.jinorgbio.2016.08.011. PMID: 27586814.
- Bhatt P, Bhatt DK, Filippenkov IB, et al. The effect of peptide Semax, an ACTH(4-10) analogue, on intracellular calcium dynamics in rat brain neurons. Bull Exp Biol Med. 2025;178(3):340-344. doi:10.1007/s10517-025-06501-z.
- Dmitrieva VG, Torshina EV, Yuzhakov VV, et al. Semax, an analog of ACTH(4-7), regulates expression of immune response genes during ischemic brain injury in rats. Mol Genet Genomics. 2017;292(3):635-653. doi:10.1007/s00438-017-1297-1. PMID: 28255762.
- Medvedeva EV, Dmitrieva VG, Povarova OV, et al. Genome-wide transcriptomic analysis of the neuroprotective peptide ACTH(4-7)PGP (Semax) action on the rat brain under conditions of cerebral ischemia-reperfusion. BMC Neurosci. 2014;15:45. doi:10.1186/1471-2202-15-45.
- Grivennikov IA, Dolotov OV, Zolotarev YA, et al. Effects of behaviorally active ACTH(4-10) analogue (Semax) on rat basal forebrain cholinergic neurons. Restor Neurol Neurosci. 2008;26(1):35-43.
- Myasoedov NF, Lyapina LA, Obergan TY, et al. Mechanism for glyproline protection in brain ischemia. J Evol Biochem Physiol. 2016;52(4):329-337.
- Levitskaya NG, Glazova NY, Andreeva LA, et al. Nootropic and anxiolytic effects of heptapeptide selank and its fragments. Bull Exp Biol Med. 2008;146(1):73-76.
- Silachev DN, Shram SI, Shakova FM, et al. Neuroprotective effect of Semax in conditions of global cerebral ischemia. Bull Exp Biol Med. 2008;146(1):123-126.
- Potaman VN, Alfeeva LYu, Kamensky AA, Levitskaya NG. Degradation of ACTH(4-10) and its synthetic analog semax by rat serum enzymes: an inhibitor study. Peptides. 1991;12(6):1297-1302.
- Manchenko DM, Glazova NY, Levitskaya NG, et al. Nootropic and analgesic effects of Semax following different routes of administration. Regul Pept. 2010;160(1-3):105-109.
- Agapova TY, Agniullin YV, Silachev DN, et al. Temporal and regional dynamics of neurotrophic factor gene expression evoked by a single intranasal administration of Semax. Dokl Biochem Biophys. 2009;424(1):18-21.
- Vakhitova YV, Sadovnikov SV, Borisevich SS, et al. Analysis of molecular mechanisms underlying the nootropic action of Semax. Bull Exp Biol Med. 2016;160(4):461-465.
- Glazova NY, Manchenko DM, Volodina MA, et al. Semax, synthetic ACTH(4-10) analogue, attenuates behavioural and neurochemical alterations following early-life fluvoxamine exposure in white rats. Neuropeptides. 2021;86:102114.
- Iasnetsov VV, Voronina TA, Saribaeva TG. Effects of semax on learning and memory in conditions of acute hypoxia. Eksp Klin Farmakol. 2005;68(2):5-7.
- Kozlov AP, Korobov VG, Galaeva IP, et al. The role of neurotrophin and melanocortin systems in the mechanism of action of Semax. Neurosci Behav Physiol. 2006;36(2):203-208.
- Stavchansky VV, Tvorogova TV, Botsina AY, et al. Analysis of the whole-transcriptome profiles of the neuroprotective action of Semax peptide in the acute period of rat cerebral ischemia. Dokl Biochem Biophys. 2020;492(1):91-95.
- Polunin GS, Nurieva SM, Baiandin DL, et al. Evaluation of therapeutic effect of new Russian drug semax in optic nerve disease. Vestn Oftalmol. 2000;116(1):15-18.
- Shadrina MI, Dolotov OV, Grivennikov IA, et al. Semax: an analog of ACTH(4-10) with cognitive effects, regulates neuropeptide gene expression in the rat hippocampus. Bull Exp Biol Med. 2010;149(4):446-449.
- Gusev EI, Martynov MYu, Kostenko EV, et al. The efficacy of neuroprotective therapy in the treatment of patients with ischemic stroke. Zh Nevrol Psikhiatr Im S S Korsakova. 2017;117(2):28-34.
- Dergunova LV, Filippenkov IB, Stavchansky VV, et al. Genome-wide transcriptome analysis using RNA-Seq reveals a large number of differentially expressed genes in a transient MCAO rat model. BMC Genomics. 2018;19(1):655. doi:10.1186/s12864-018-5039-5.
- Bobyntsev II, Kryukov AA, Shepeleva OM, Ivanov AV. Effect of Semax on the morphology of the thymus and adrenals in neonatal monosodium glutamate-treated rats. Bull Exp Biol Med. 2015;159(1):69-72.
- Levin OS, Yunishchenko NA, Dudarova MA. Efficacy of Semax in the treatment of cognitive impairment in patients with dyscirculatory encephalopathy. Zh Nevrol Psikhiatr Im S S Korsakova. 2007;107(7):20-24.
- Romanova GA, Shakova FM, Barskov IV, Stelmashuk EV. Effect of Semax on passive avoidance retention and morphological changes in the rat hippocampus after global cerebral ischemia. Bull Exp Biol Med. 2006;142(6):663-666.
- Myasoedov NF, Gavrilova SA, Evdokimova NR, et al. The effect of Semax on the stress response system and inflammatory markers in rats with experimental ischemic stroke. Dokl Biol Sci. 2014;456(1):186-189.
- Inozemtseva LS, Karpenko EA, Dolotov OV, et al. Intranasal administration of the heptapeptide Semax increases the expression of BDNF mRNA in the rat hippocampus in vivo. Dokl Biol Sci. 2008;423(1):402-404.
- Kaplan AYa, Kochetova AG, Nezavibatko VN, et al. Synthetic ACTH analogue Semax displays nootropic-like activity in humans. Neurosci Res Commun. 1996;19(2):115-123.
- Sudarkina OY, Filippenkov IB, Stavchansky VV, et al. Brain protein expression profile confirms the neuroprotective effect of the ACTH(4-7)PGP peptide (Semax) in a rat model of cerebral ischemia-reperfusion. Int J Mol Sci. 2021;22(12):6179. doi:10.3390/ijms22126179.

Figure 9: Semax research timeline spanning over three decades, from initial ACTH fragment studies through modern genome-wide transcriptomic analyses.