Executive Summary

Figure 1: CagriSema combines two distinct peptide therapies into a single weekly injection for enhanced weight management.
Key Takeaways
- Figure 1: CagriSema combines two distinct peptide therapies into a single weekly injection for enhanced weight management.
- CagriSema is Novo Nordisk's fixed-dose combination of cagrilintide (a long-acting amylin analog) and semaglutide (a GLP-1 receptor agonist), administered as a single once-weekly subcutaneous injection.
- By engaging two independent hormonal pathways that govern satiety and energy balance, CagriSema produces weight loss substantially greater than either agent alone.
- Phase 3 data from the REDEFINE program showed mean body weight reductions of up to 22.7% at 68 weeks in adults without diabetes, and 13.7% in adults with type 2 diabetes.
- Novo Nordisk filed for FDA approval in December 2025, with a regulatory decision anticipated by late 2026.
CagriSema is Novo Nordisk's fixed-dose combination of cagrilintide (a long-acting amylin analog) and semaglutide (a GLP-1 receptor agonist), administered as a single once-weekly subcutaneous injection. By engaging two independent hormonal pathways that govern satiety and energy balance, CagriSema produces weight loss substantially greater than either agent alone. Phase 3 data from the REDEFINE program showed mean body weight reductions of up to 22.7% at 68 weeks in adults without diabetes, and 13.7% in adults with type 2 diabetes. Novo Nordisk filed for FDA approval in December 2025, with a regulatory decision anticipated by late 2026.
The logic behind CagriSema is disarmingly simple: if one appetite-suppressing hormone works well, two hormones acting through separate brain circuits should work better. That hypothesis has now been tested in thousands of patients across multiple Phase 3 trials. The results confirm that combining amylin and GLP-1 receptor agonism produces additive, and possibly complementary, reductions in body weight.
Amylin is a 37-amino-acid peptide co-secreted with insulin from pancreatic beta cells after meals. It slows gastric emptying, suppresses glucagon release, and signals satiety through the area postrema and other hindbrain structures. GLP-1, by contrast, acts primarily on hypothalamic and brainstem circuits and through vagal afferent pathways to reduce hunger. These two systems share some downstream effects but reach the brain through distinct receptor populations and anatomical routes. When both are activated simultaneously, the cumulative effect on appetite suppression and caloric intake exceeds what either pathway achieves in isolation.
Cagrilintide was engineered specifically to overcome the limitations of pramlintide (Symlin), an earlier amylin analog that required multiple daily injections and never gained traction in obesity treatment. Through fatty acid acylation, cagrilintide achieves a half-life of roughly seven days, making weekly dosing feasible. Paired with semaglutide 2.4 mg, which already represented the standard of care for pharmacological weight management, the combination creates a dual-hormone therapy that pushes weight loss toward levels previously seen only with bariatric surgery.
Key Takeaway
CagriSema delivered 22.7% mean weight loss at 68 weeks in the REDEFINE 1 trial (efficacy estimand), with over 40% of participants losing at least 25% of their body weight. This positions it among the most effective non-surgical obesity treatments ever tested in a Phase 3 program.
The clinical development program for CagriSema, known as REDEFINE, spans multiple trials examining obesity with and without type 2 diabetes, cardiovascular outcomes, and head-to-head comparison with tirzepatide. The REDEFINE 4 trial, which compared CagriSema directly against tirzepatide 15 mg, showed CagriSema achieving 23.0% weight loss versus 25.5% with tirzepatide at 84 weeks. While CagriSema did not meet its prespecified margin for non-inferiority in that trial, both agents produced clinically meaningful results that would have seemed extraordinary just a decade ago.
This report examines CagriSema from every angle: the scientific rationale for combining amylin and GLP-1 agonism, the molecular pharmacology of cagrilintide, the full results from Phase 2 and Phase 3 clinical trials, safety and tolerability data, and the regulatory path forward. For those tracking the rapidly evolving GLP-1 research hub, CagriSema represents one of the most consequential developments in the next wave of anti-obesity therapeutics.
The broader implications reach beyond individual weight loss. If approved, CagriSema would be the first combination amylin/GLP-1 therapy on the market, establishing a new therapeutic class. It would also intensify competition with Eli Lilly's tirzepatide (Zepbound/Mounjaro) and Amgen's MariTide, among other pipeline agents. For patients and clinicians, more options mean more personalized treatment. For the field of obesity medicine, CagriSema underscores a central truth: obesity is a disease of multiple disrupted signaling pathways, and the most effective treatments will likely target more than one of them.
Whether you're a clinician evaluating next-generation options for your patients, a researcher following the incretin and amylin literature, or someone seeking evidence-based information about emerging weight management therapies, this guide provides a thorough analysis grounded in published trial data and peer-reviewed science.
The Amylin-GLP-1 Hypothesis
Figure 2: The path from amylin's discovery to CagriSema spans over three decades of research and pharmaceutical development.
Discovery of Amylin: A Forgotten Hormone
Amylin's story begins in 1987, when researchers at Oxford identified a previously unknown peptide deposited in the pancreatic islets of patients with type 2 diabetes. Garth Cooper and colleagues characterized this 37-amino-acid peptide and named it islet amyloid polypeptide (IAPP), later known simply as amylin. It was co-stored and co-secreted with insulin from beta cell granules, yet for years it attracted far less attention than its better-known companion.
Early research focused primarily on amylin's role in amyloid formation and beta cell destruction in type 2 diabetes. But by the early 1990s, physiologists began to appreciate that amylin had important metabolic functions of its own. Studies in rodents showed that amylin slowed gastric emptying, suppressed postprandial glucagon secretion, and reduced food intake. These effects were mediated through receptors concentrated in the area postrema, a circumventricular organ in the brainstem that sits outside the blood-brain barrier and serves as a critical relay for satiety signals.
The area postrema connection was significant. Unlike many CNS targets, the area postrema is accessible to circulating peptides. Amylin didn't need to cross the blood-brain barrier to exert its central effects. It could reach its target receptors directly from the bloodstream. This made amylin a viable therapeutic candidate, something that couldn't be said for many other appetite-regulating neuropeptides.
Pramlintide: The First (and Only) Amylin Therapy
Amylin Pharmaceuticals, founded in San Diego in 1987, set out to develop an amylin-based drug. Native human amylin was unsuitable for clinical use because it aggregated rapidly into amyloid fibrils. The company turned to rat amylin, which differs at several key residues and resists aggregation. Using rat amylin as a template, they engineered pramlintide (Symlin) - a synthetic amylin analog with three proline substitutions (A25P, S28P, S29P) that prevented fibril formation while preserving receptor activity.
The FDA approved pramlintide in 2005 as an adjunct to insulin therapy in patients with type 1 or type 2 diabetes. It was administered by subcutaneous injection before each major meal - three times daily, separate from insulin injections. This dosing burden, combined with a requirement for careful insulin dose adjustment to avoid hypoglycemia, limited its uptake. Pramlintide modestly reduced HbA1c (by about 0.3-0.5%) and body weight (by roughly 1-2 kg), but the clinical effect was underwhelming compared to the injection burden.
The weight loss signal, however small, caught attention. If a short-acting amylin analog given three times daily could produce modest weight reduction, what might a long-acting version achieve? And what if that long-acting amylin analog were combined with a potent GLP-1 receptor agonist?
The Incretin Revolution and GLP-1 Agonism
While amylin research progressed slowly, the GLP-1 field accelerated dramatically. Exenatide (Byetta) became the first GLP-1 receptor agonist approved by the FDA in 2005, the same year as pramlintide. Liraglutide followed in 2010 for diabetes and 2014 for obesity (as Saxenda). Then came semaglutide, which demonstrated unprecedented weight loss in the STEP program - roughly 15-17% mean body weight reduction at 68 weeks in people with obesity.
Semaglutide's success with Wegovy and Ozempic transformed the obesity treatment landscape. But researchers noticed something: even with potent GLP-1 agonism, weight loss plateaued in most patients after 60-70 weeks. The body's counter-regulatory mechanisms - metabolic adaptation, altered appetite hormone profiles, reduced energy expenditure - created resistance to further weight loss. Breaking through that plateau might require engaging additional hormonal pathways.
This is exactly where amylin entered the conversation again. GLP-1 receptors are densely expressed in the hypothalamic arcuate nucleus, the nucleus tractus solitarius, and on vagal afferent neurons. Amylin receptors, by contrast, are concentrated in the area postrema and the dorsal vagal complex. The two systems converge on appetite and satiety regulation but approach the problem through different anatomical and molecular routes. Combining them could, in theory, produce additive or even complementary effects.
Novo Nordisk's Strategic Bet
Novo Nordisk had been developing GLP-1 therapies since the late 1990s. The company launched liraglutide (Victoza/Saxenda) and then semaglutide (Ozempic/Wegovy/Rybelsus), building an enormous commercial and scientific franchise around incretin-based therapies. By the mid-2010s, the company recognized that the next competitive frontier would require something beyond semaglutide alone.
In 2016, Novo Nordisk began clinical development of cagrilintide (originally designated AM833 or NN0174-0833), a long-acting amylin analog designed from the ground up for once-weekly dosing. The goal was clear: create an amylin analog that could be combined with semaglutide in a single injection pen, doubling the hormonal firepower against obesity.
The strategic rationale went beyond basic science. Novo Nordisk needed a successor to semaglutide that would maintain its market leadership as competitors developed dual and triple agonists. Tirzepatide, Eli Lilly's dual GIP/GLP-1 agonist, was showing weight loss of 20-25% in its own Phase 3 program. Amgen was developing MariTide, a long-acting amylin/calcitonin receptor agonist combined with an anti-GIP antibody. The competitive pressure was intense.
CagriSema was Novo Nordisk's answer: a combination that could potentially match or exceed the weight loss achieved by competitors' novel mechanisms, while building on the company's deep experience with semaglutide. The concept was elegant in its simplicity - two proven hormone analogs, each well-characterized individually, combined into one weekly injection.
Historical Context
The idea of combining amylin and GLP-1 agonism dates back to early 2000s academic research. Roth and colleagues at Novo Nordisk published preclinical data as early as 2008 showing additive weight loss effects when amylin and GLP-1 agonists were co-administered in rodent models. But it took another decade to develop a long-acting amylin analog suitable for weekly dosing, which was the missing piece that made the combination clinically practical.
Preclinical Foundations
Before entering human trials, Novo Nordisk conducted extensive preclinical work characterizing the cagrilintide/semaglutide combination. Studies in diet-induced obese rats and non-human primates consistently showed that the combination produced greater reductions in food intake and body weight than either agent alone. The effects appeared additive rather than merely redundant.
Mechanistic studies revealed that the two drugs activated distinct neuronal populations. c-Fos mapping (a marker of neuronal activation) in rodent brains showed that semaglutide primarily activated neurons in the hypothalamic arcuate nucleus and the nucleus tractus solitarius, while cagrilintide activated area postrema neurons. When given together, both populations lit up. The sum was clearly greater than either part.
These preclinical findings provided the scientific justification for moving into human trials with the combination. They also offered a neuroanatomical explanation for why the combination worked: two distinct brain regions, each governing different aspects of appetite and energy balance, were being simultaneously engaged. It was not simply doubling the same signal. It was activating complementary circuits.
From Hypothesis to Clinical Reality
The amylin-GLP-1 combination hypothesis matured through several phases. First came the academic observation that amylin and GLP-1 acted through different receptor systems. Then came preclinical proof-of-concept showing additive effects in animal models. Then came the engineering challenge of creating a long-acting amylin analog. And finally came the clinical development program that would test whether the combination hypothesis held true in humans.
Each step took years. The total journey from amylin's discovery in 1987 to CagriSema's FDA filing in December 2025 spans nearly four decades. That timeline reflects the complexity of translating a basic science observation into a viable therapeutic product, but it also reflects something else: the obesity treatment field's slow recognition that effective pharmacotherapy would require targeting multiple hormonal systems simultaneously.
For a broader view of GLP-1 receptor agonist development, see the GLP-1 research hub. For detailed information on semaglutide monotherapy, consult our semaglutide guide.
Cagrilintide: Long-Acting Amylin Analog
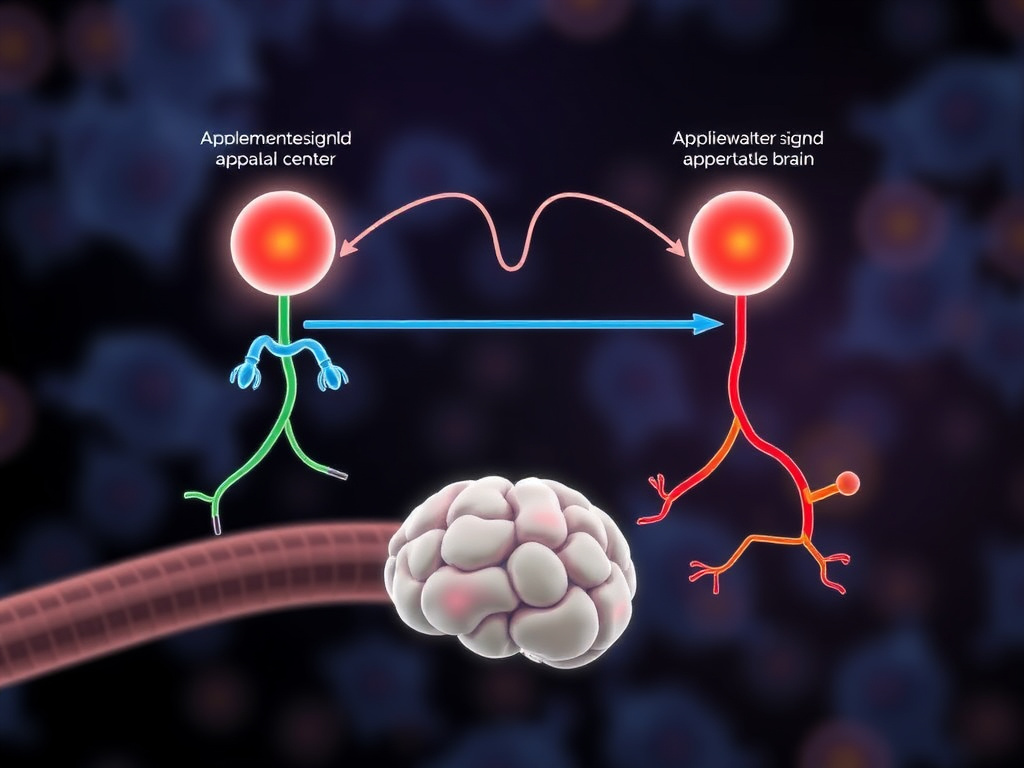
Figure 3: Cagrilintide's molecular design enables potent, long-acting amylin receptor engagement through strategic acylation and sequence optimization.
The Engineering Challenge
Native human amylin is a poor drug candidate for three reasons. First, it aggregates into amyloid fibrils within hours of being dissolved, making it unstable in solution. Second, its plasma half-life is roughly 13 minutes, requiring frequent injections. Third, its potency at amylin receptors, while adequate for physiological signaling, is insufficient for the supraphysiological exposure needed for meaningful weight loss.
Pramlintide solved the first problem with proline substitutions that disrupted beta-sheet formation and prevented aggregation. But it didn't address the other two. Cagrilintide was designed to solve all three simultaneously.
Molecular Design Strategy
Cagrilintide is a 32-amino-acid synthetic peptide derived from human amylin but with several critical modifications. The Novo Nordisk team used an iterative structure-activity relationship (SAR) approach, systematically testing thousands of analogs to optimize potency, stability, and pharmacokinetics.
The key modifications include:
- Sequence optimization: Multiple amino acid substitutions were introduced to increase receptor potency and reduce aggregation propensity. The peptide incorporates elements from both human amylin and the more stable rat amylin sequence, along with novel substitutions identified through SAR screening.
- Intra-peptide ionic lock: An engineered salt bridge between residues at positions analogous to R17 and E14 stabilizes a specific peptide conformation (the "bypass" conformation) that favors high-affinity binding to calcitonin receptors and amylin receptors.
- N-terminal acetylation and C-terminal amidation: These modifications cap both ends of the peptide, protecting it from exopeptidase degradation.
- C18 fatty acid acylation at Lys-1: This is the single most important modification for pharmacokinetics. A fatty acid side chain (specifically a C18 fatty diacid) is conjugated to the lysine at position 1 through a glutamic acid-based linker.
Acylation: The Key to Weekly Dosing
Fatty acid acylation is the technology that transformed cagrilintide from a short-acting peptide into a once-weekly drug. Novo Nordisk pioneered this approach with liraglutide (which uses a C16 fatty acid) and refined it further with semaglutide (which uses a C18 fatty diacid). The same principle applies to cagrilintide.
Here's how it works: the fatty acid chain, once conjugated to the peptide, binds non-covalently to serum albumin in the bloodstream. Albumin is the most abundant plasma protein, with a half-life of approximately 19 days. When cagrilintide binds to albumin, it effectively hitches a ride on this long-lived carrier protein. The bound fraction is protected from renal filtration (albumin is too large to pass through the glomerulus) and from enzymatic degradation (the albumin-peptide complex shields the peptide from proteases).
The result is dramatic. Native amylin has a half-life of about 13 minutes. Cagrilintide has a half-life of approximately 160-170 hours (roughly 7 days). That's a greater than 700-fold extension. This pharmacokinetic profile supports once-weekly dosing with stable plasma concentrations and minimal peak-to-trough fluctuation.
| Property | Native Amylin | Pramlintide | Cagrilintide |
|---|---|---|---|
| Amino acid length | 37 | 37 | 32 |
| Aggregation | High (amyloidogenic) | Low (proline substitutions) | Low (sequence design) |
| Half-life | ~13 minutes | ~48 minutes | ~160-170 hours |
| Dosing frequency | N/A | 3x daily (before meals) | Once weekly |
| Acylation | None | None | C18 fatty diacid at Lys-1 |
| Albumin binding | No | No | Yes (high affinity) |
| Route | N/A | Subcutaneous | Subcutaneous |
Amylin Receptor Pharmacology
Amylin doesn't have a single dedicated receptor. Instead, it signals through a family of heteromeric receptors formed by the calcitonin receptor (CTR) complexed with receptor activity-modifying proteins (RAMPs). There are three RAMPs (RAMP1, RAMP2, RAMP3), and each forms a distinct amylin receptor subtype when paired with CTR:
- AMY1 receptor = CTR + RAMP1
- AMY2 receptor = CTR + RAMP2
- AMY3 receptor = CTR + RAMP3
Cagrilintide binds with high affinity to all three amylin receptor subtypes, with particularly strong potency at AMY1 and AMY3. It also retains activity at the calcitonin receptor itself, though its selectivity profile favors the RAMP-complexed forms. This broad amylin receptor engagement is important because the different subtypes are expressed in different brain regions, and maximal satiety signaling likely requires activation of multiple subtypes.
Cryo-EM structural studies published in 2025 revealed how cagrilintide binds to these receptors at the molecular level. The peptide adopts a characteristic "bypass" conformation when engaging the receptor's transmembrane domain, with the engineered ionic lock stabilizing this conformation. The fatty acid chain, while primarily serving a pharmacokinetic function, also makes contacts with the receptor extracellular domain that may contribute to binding affinity.
Cagrilintide as Monotherapy
Before being combined with semaglutide, cagrilintide was tested as a standalone agent. A Phase 2 trial published in The Lancet in 2021 evaluated cagrilintide at doses ranging from 0.3 mg to 4.5 mg once weekly in 706 adults with overweight or obesity. At 26 weeks, the highest dose (4.5 mg) produced a mean weight loss of 10.8% compared to 3.0% with placebo.
This was an impressive result for a standalone amylin analog and validated the concept that long-acting amylin agonism could produce clinically meaningful weight loss. But 10.8% was not competitive with semaglutide 2.4 mg monotherapy, which had already shown 15-17% weight loss in the STEP trials. Cagrilintide as monotherapy was never going to be the commercial product. Its purpose was always to be part of a combination.
Clinical Insight
The Phase 2 monotherapy data for cagrilintide served a critical purpose: establishing the dose-response relationship and safety profile for the amylin component independently, before combining it with semaglutide. The 2.4 mg dose was selected for CagriSema because it offered a strong efficacy signal with an acceptable side effect profile, while matching the standard weekly dosing of semaglutide 2.4 mg.
Pharmacokinetic Profile
Detailed pharmacokinetic studies of cagrilintide reveal several features relevant to its clinical performance:
- Absorption: After subcutaneous injection, cagrilintide is absorbed slowly, reaching peak plasma concentrations (Tmax) at approximately 24-72 hours post-dose.
- Distribution: Greater than 99% bound to plasma albumin. The volume of distribution is small, consistent with distribution primarily within the vascular compartment.
- Metabolism: Eliminated primarily through proteolytic degradation, not hepatic or renal clearance. This means dose adjustment is generally not required for mild to moderate hepatic or renal impairment.
- Steady state: Achieved after approximately 4-5 weekly doses, with an accumulation ratio of roughly 3-fold.
- Drug interaction with semaglutide: Pharmacokinetic studies showed no clinically relevant drug-drug interaction between cagrilintide and semaglutide when co-administered. Neither agent altered the exposure of the other.
This last point was critical for CagriSema development. If semaglutide and cagrilintide had interfered with each other's absorption or clearance, the combination approach would have been far more complex. The fact that their pharmacokinetics are essentially independent simplified the fixed-dose combination design.
For researchers interested in understanding how cagrilintide fits into the broader peptide pharmacology landscape, a useful comparison can be drawn to the acylation strategies used for other long-acting peptides including semaglutide and insulin degludec.
Dual Mechanism of CagriSema
Figure 4: CagriSema engages two distinct brain pathways - amylin signaling through the area postrema and GLP-1 signaling through the hypothalamus and brainstem.
Two Hormones, Two Pathways, One Goal
Understanding why CagriSema works better than either of its components requires understanding how amylin and GLP-1 signaling differ at the neuroanatomical level. Both hormones reduce appetite and food intake. But they do so through circuits that are more complementary than redundant.
Semaglutide activates GLP-1 receptors (GLP-1R) found in high density across several key brain regions: the hypothalamic arcuate nucleus (ARC), the paraventricular nucleus (PVN), the dorsomedial hypothalamus (DMH), the nucleus tractus solitarius (NTS) in the brainstem, and on vagal afferent neurons innervating the gastrointestinal tract. Through these pathways, GLP-1 agonism reduces hunger, promotes satiety, slows gastric emptying, and enhances insulin secretion in a glucose-dependent manner.
Cagrilintide activates amylin receptors (AMY1R, AMY2R, AMY3R) concentrated in the area postrema (AP), the dorsal vagal complex, and to a lesser extent in the hypothalamus. The area postrema is a circumventricular organ that lacks a blood-brain barrier, allowing circulating peptides direct access to its neurons. Amylin receptor activation in the AP triggers downstream signaling that inhibits food intake, slows gastric emptying, and suppresses postprandial glucagon secretion.
The Area Postrema: Amylin's Primary Target
The area postrema deserves special attention because it's the critical differentiator between amylin and GLP-1 signaling. Situated at the floor of the fourth ventricle, the AP is one of the brain's sensory windows to the bloodstream. It detects circulating toxins (which is why it's involved in the vomiting reflex), but it also monitors metabolic hormones and nutrients.
Amylin receptors in the AP trigger a cascade that includes activation of extracellular signal-regulated kinase (ERK1/2), phosphorylation of CREB, and downstream expression of genes involved in satiety signaling. Lesion studies in rodents have been definitive: when the area postrema is destroyed, amylin loses its ability to reduce food intake. The same lesion does not abolish the anorectic effects of GLP-1 agonists, confirming that the two systems operate through anatomically distinct circuits.
This is the core neurobiological rationale for CagriSema. By simultaneously activating GLP-1 receptors in the hypothalamus and NTS, and amylin receptors in the area postrema, the combination engages two parallel satiety circuits. The signals converge on common downstream effectors (such as reduced meal size and increased inter-meal intervals), but because they originate from different receptor populations in different brain regions, their effects are additive rather than ceiling-limited.
Complementary Effects on Gastric Emptying
Both amylin and GLP-1 slow gastric emptying, but they appear to do so through partially distinct mechanisms. GLP-1 receptor agonism delays gastric emptying primarily through vagal efferent pathways and possibly direct effects on gastric smooth muscle GLP-1 receptors. Amylin slows gastric emptying through area postrema-mediated vagal circuits.
Clinical studies suggest that the gastric emptying effects of these two hormones are not fully redundant. In the CagriSema Phase 2 trials, participants reported greater and more sustained reductions in appetite and food intake compared to either monotherapy, consistent with enhanced delay in gastric emptying from dual pathway engagement.
Slower gastric emptying contributes to weight loss by extending the period of postprandial satiety and reducing overall caloric intake. It also contributes to improved glycemic control by slowing the rate at which ingested carbohydrates enter the small intestine and are absorbed.
Glucose Regulation: Beyond Weight Loss
CagriSema's dual mechanism provides glucose-lowering effects through multiple routes:
| Mechanism | Semaglutide (GLP-1R) | Cagrilintide (AMYR) | Combined Effect |
|---|---|---|---|
| Insulin secretion (glucose-dependent) | Strong enhancement | Modest enhancement | Additive |
| Glucagon suppression | Moderate (post-meal) | Strong (post-meal) | Additive |
| Gastric emptying delay | Moderate | Moderate | Additive |
| Appetite suppression | Strong (hypothalamic) | Strong (area postrema) | Additive/complementary |
| Weight loss | 15-17% (monotherapy) | 10-11% (monotherapy) | 20-23% (combination) |
The REDEFINE 2 trial demonstrated that in patients with type 2 diabetes, CagriSema produced an HbA1c reduction of 2.2 percentage points (from a baseline of approximately 8.1%), with 73.5% of participants achieving an HbA1c of 6.5% or below. These glycemic improvements reflect the convergent effects of both components on insulin secretion, glucagon suppression, and weight loss-mediated improvements in insulin sensitivity.
Reward Circuitry and Food Preference
Emerging evidence suggests that both amylin and GLP-1 signaling modulate reward-related brain regions, including the ventral tegmental area (VTA) and nucleus accumbens. GLP-1 receptor agonists reduce the hedonic value of food, particularly high-fat and high-sugar foods, as demonstrated in neuroimaging studies with semaglutide. Amylin receptor activation in the VTA has been shown in rodent studies to similarly reduce the motivation to seek palatable food.
While human neuroimaging data for the CagriSema combination are not yet published, the preclinical evidence suggests that dual pathway engagement may produce greater reductions in hedonic eating than either agent alone. This could be particularly relevant for long-term weight maintenance, where food reward sensitivity often drives regain.
Key Mechanism Summary
CagriSema's superiority over monotherapy stems from engaging two neuroanatomically distinct appetite circuits: GLP-1 agonism in the hypothalamus/NTS and amylin agonism in the area postrema. These pathways are additive because they use different receptors in different brain regions, converging on shared downstream outcomes (reduced hunger, slower gastric emptying, improved glucose regulation) without competing for the same receptors.
Why Not Just Increase the Semaglutide Dose?
A reasonable question arises: if the goal is more weight loss, why not simply increase the dose of semaglutide beyond 2.4 mg? Novo Nordisk is in fact testing higher semaglutide doses in separate trials. But dose escalation hits a wall: gastrointestinal side effects (nausea, vomiting, diarrhea) increase sharply at higher doses, and the dose-response curve for weight loss flattens. There's a ceiling effect for any single pathway.
By adding cagrilintide instead of increasing the semaglutide dose, CagriSema achieves greater efficacy without proportionally increasing GI side effects. The amylin pathway provides additional appetite suppression through different receptors, bypassing the ceiling on GLP-1 receptor-mediated effects. This is the fundamental advantage of combination therapy over dose escalation.
For additional reading on how GLP-1 receptor agonists work as monotherapies, see our semaglutide guide. For an understanding of the dual GIP/GLP-1 approach taken by competitors, see our tirzepatide guide.
Phase 2 Trial Results
Figure 5: Phase 2 clinical data demonstrated that CagriSema produced significantly greater weight loss than either component alone.
Trial Design
The Phase 2 proof-of-concept trial for the cagrilintide/semaglutide combination was a multicenter, randomized, double-blind, active-controlled study conducted in adults with type 2 diabetes. Published in The Lancet in 2023 by Frias and colleagues, this trial enrolled 92 participants and evaluated the combination of cagrilintide 2.4 mg and semaglutide 2.4 mg (both administered once weekly) against cagrilintide 2.4 mg alone and semaglutide 2.4 mg alone over 32 weeks.
The trial was relatively small but designed to answer a specific question: does combining the two agents produce greater weight loss than either monotherapy? Key inclusion criteria included adults aged 18-74 with type 2 diabetes, HbA1c 7.0-10.0%, BMI of 27 kg/m2 or higher, and stable metformin therapy.
Primary Results
% Weight Loss at 32 Weeks: Mono vs Combination
The results were striking. At 32 weeks, the combination group achieved a mean body weight reduction of 15.6% (in the primary analysis population). When broken down by group:
| Treatment Arm | Mean Weight Loss (%) | Mean Weight Loss (kg) | Participants Achieving ≥10% Loss |
|---|---|---|---|
| Placebo | 1.1% | ~1.0 kg | <5% |
| Cagrilintide 2.4 mg alone | 10.8% | ~10.5 kg | ~60% |
| Semaglutide 2.4 mg alone | 15.5% | ~15.0 kg | ~75% |
| CagriSema (combination) | 22.7% | ~22.0 kg | ~90% |
The 22.7% weight loss with the combination exceeded the sum of the individual components over placebo (cagrilintide added 9.7% and semaglutide added 14.4%, for a theoretical additive total of 24.1%). The actual combination result of 22.7% fell slightly below perfect additivity, suggesting some overlap in the mechanisms, but the clinical magnitude was remarkable for a 32-week trial in patients with type 2 diabetes, a population where weight loss is typically harder to achieve than in non-diabetic individuals.
Glycemic Control
The combination also produced superior glycemic outcomes. HbA1c reductions at 32 weeks were:
| Treatment Arm | Baseline HbA1c | HbA1c Change at 32 Weeks | % Achieving HbA1c <7.0% |
|---|---|---|---|
| Placebo | ~8.0% | -0.1% | ~15% |
| Cagrilintide 2.4 mg alone | ~8.1% | -0.9% | ~45% |
| Semaglutide 2.4 mg alone | ~8.0% | -1.8% | ~70% |
| CagriSema (combination) | ~8.0% | -2.2% | ~85% |
The combination's HbA1c-lowering effect exceeded both monotherapies, driven by the additive effects of amylin and GLP-1 agonism on glucagon suppression, gastric emptying, and weight loss-mediated improvements in insulin sensitivity.
Body Composition and Metabolic Parameters
Secondary endpoints from the Phase 2 trial revealed improvements across a broad range of metabolic parameters:
- Waist circumference: CagriSema reduced waist circumference by approximately 14 cm, compared to ~10 cm with semaglutide alone and ~8 cm with cagrilintide alone.
- Fasting plasma glucose: Greater reductions with the combination than with either monotherapy.
- Lipid profile: Improvements in triglycerides, total cholesterol, and LDL cholesterol that exceeded the improvements seen with either drug alone.
- Blood pressure: Modest reductions in systolic blood pressure in the combination group, consistent with the weight loss achieved.
Dose-Finding Insights
A separate Phase 2 dose-finding study evaluated multiple dose combinations of cagrilintide and semaglutide to identify the optimal doses for Phase 3. The study tested cagrilintide at 1.0 mg, 2.4 mg, and 4.5 mg combined with semaglutide at 2.4 mg. The cagrilintide 2.4 mg / semaglutide 2.4 mg combination was selected for Phase 3 development because:
- It provided near-maximal weight loss efficacy
- It had a manageable side effect profile
- The 2.4 mg dose of cagrilintide matched well with the established 2.4 mg semaglutide dose for pen device engineering
- Higher cagrilintide doses (4.5 mg) added modest additional weight loss but increased gastrointestinal side effects disproportionately
Clinical Significance
The Phase 2 results were remarkable for two reasons. First, 22.7% weight loss at just 32 weeks in a diabetic population surpassed what most existing monotherapies achieve at 68 weeks. Second, the weight loss curve had not yet fully plateaued at week 32, suggesting that longer treatment duration could produce even greater reductions, a prediction that was subsequently confirmed in the Phase 3 REDEFINE program.
Limitations of the Phase 2 Data
The Phase 2 trial had important limitations. The sample size was small (N=92), limiting statistical power for safety analyses and subgroup comparisons. The 32-week duration was shorter than the standard 68-week timeframe used in registrational obesity trials. And the study population included only adults with type 2 diabetes, meaning the results could not be directly extrapolated to the broader obesity population without diabetes.
These limitations were addressed by the Phase 3 REDEFINE program, which enrolled thousands of participants across multiple populations and treatment durations. Still, the Phase 2 data provided the foundational evidence that CagriSema's dual mechanism translated into clinically meaningful superiority over each component, setting the stage for the large-scale trials that would follow.
Phase 3 REDEFINE Program

Figure 6: The REDEFINE Phase 3 program spans multiple trials enrolling thousands of participants across different populations and comparators.
Program Overview
The REDEFINE clinical trial program is one of the largest Phase 3 programs ever conducted for an anti-obesity medication. It includes multiple trials designed to evaluate CagriSema across different patient populations, treatment comparisons, and clinical endpoints. The program's name reflects Novo Nordisk's ambition: to redefine the standard of care in obesity pharmacotherapy.
| Trial | Population | Comparator | Duration | N (approx) | Status |
|---|---|---|---|---|---|
| REDEFINE 1 | Obesity/overweight without T2D | Placebo | 68 weeks | 3,417 | Completed; published in NEJM |
| REDEFINE 2 | Obesity/overweight with T2D | Placebo | 68 weeks | 1,206 | Completed; published in NEJM |
| REDEFINE 3 | Obesity with cardiovascular disease | Placebo | Event-driven (MACE) | ~20,000 | Ongoing (cardiovascular outcomes trial) |
| REDEFINE 4 | Obesity without T2D | Tirzepatide 15 mg | 84 weeks | 809 | Completed; headline results reported |
| REDEFINE 6 | Obesity with heart failure (HFpEF) | Placebo | 52 weeks | ~600 | Ongoing |
| REDEFINE 11 | Obesity without T2D | Higher-dose CagriSema | 68 weeks | TBD | Planned |
REDEFINE 1: The Registrational Obesity Trial
REDEFINE 1 was the registrational trial that formed the primary basis for the FDA submission. Published in the New England Journal of Medicine in 2025 (Wilding et al.), it enrolled 3,417 adults with a BMI of 30 kg/m2 or greater (or 27+ with at least one weight-related comorbidity) but without type 2 diabetes.
Participants were randomized 1:1 to CagriSema (cagrilintide 2.4 mg + semaglutide 2.4 mg) or matching placebo, both administered once weekly by subcutaneous injection. The treatment period was 68 weeks, following a dose-escalation phase.
Primary Endpoints and Key Results
REDEFINE 1 used two co-primary endpoints, each assessed under a different statistical estimand:
- Treatment policy estimand (all randomized participants, including those who discontinued): CagriSema achieved a mean weight loss of 20.4% versus 3.0% with placebo.
- Trial product estimand (efficacy if all participants remained on treatment): CagriSema achieved 22.7% weight loss versus 2.3% with placebo.
Both co-primary endpoints were met with high statistical significance (p < 0.0001).
| Endpoint | CagriSema | Placebo | Difference |
|---|---|---|---|
| Mean weight loss (treatment policy) | 20.4% | 3.0% | 17.4 percentage points |
| Mean weight loss (trial product) | 22.7% | 2.3% | 20.4 percentage points |
| Participants achieving ≥5% loss | 89.7% | 31.7% | -- |
| Participants achieving ≥10% loss | 79.3% | 14.1% | -- |
| Participants achieving ≥15% loss | 66.8% | 7.0% | -- |
| Participants achieving ≥20% loss | 54.0% | 3.8% | -- |
| Participants achieving ≥25% loss | 40.4% | 1.5% | -- |
| Participants achieving BMI <30 | >50% | 10.2% | -- |
These results placed CagriSema among the most effective pharmacological weight management therapies ever tested. Over half of participants in the CagriSema group lost more than 20% of their body weight, and 40% lost at least 25%. More than half achieved a BMI below the obesity threshold of 30 kg/m2.
REDEFINE 2: Obesity with Type 2 Diabetes
REDEFINE 2, also published in the NEJM, enrolled 1,206 adults with both obesity/overweight and type 2 diabetes. This population traditionally shows lower weight loss with GLP-1 agonists compared to non-diabetic individuals, possibly due to insulin's anabolic effects and the metabolic adaptations inherent in type 2 diabetes.
Key results from REDEFINE 2:
| Endpoint | CagriSema | Placebo |
|---|---|---|
| Mean weight loss (treatment policy) | 13.7% | 3.4% |
| HbA1c change from baseline | -2.2 percentage points | -0.3 percentage points |
| Participants achieving HbA1c ≤6.5% | 73.5% | 15.9% |
| Participants achieving ≥10% weight loss | ~60% | ~10% |
The 13.7% weight loss in a diabetic population was a strong result, exceeding what semaglutide 2.4 mg achieves as monotherapy in similar populations (~10-12%). The glycemic control results were also impressive, with nearly three-quarters of participants reaching an HbA1c below 6.5%, a threshold many endocrinologists consider near-remission for type 2 diabetes.
REDEFINE 4: Head-to-Head with Tirzepatide
REDEFINE 4 was the most closely watched trial in the program because it directly compared CagriSema to its chief commercial rival, tirzepatide 15 mg (Zepbound/Mounjaro). This open-label, randomized trial enrolled 809 adults with obesity (BMI ≥30) without type 2 diabetes and ran for 84 weeks.
The primary endpoint was non-inferiority of CagriSema to tirzepatide 15 mg for body weight reduction. The results, reported in February 2026:
| Endpoint | CagriSema | Tirzepatide 15 mg |
|---|---|---|
| Mean weight loss (trial product estimand) | 23.0% | 25.5% |
| Mean weight loss (treatment policy estimand) | 20.2% | 23.6% |
CagriSema did not meet the prespecified non-inferiority margin versus tirzepatide 15 mg. The 2.5 percentage point gap in the primary analysis (23.0% vs 25.5%) fell outside the pre-defined boundary. This result triggered a significant decline in Novo Nordisk's share price and raised questions about CagriSema's competitive positioning.
However, context matters. A 23% mean weight loss is still an extraordinary clinical result. The trial compared CagriSema at its current fixed dose (cagrilintide 2.4 mg / semaglutide 2.4 mg) against the highest approved dose of tirzepatide (15 mg). Novo Nordisk has indicated that higher-dose CagriSema formulations are being developed and will be tested in the REDEFINE 11 trial, which could close or eliminate the gap.
Important Context
REDEFINE 4 was an open-label trial, meaning both patients and investigators knew which treatment each participant received. Open-label designs can influence outcomes through behavioral changes, adherence patterns, and expectation effects. The results should be interpreted with this limitation in mind, and a double-blind head-to-head comparison would provide more definitive evidence.
REDEFINE 3: Cardiovascular Outcomes
REDEFINE 3 is a large cardiovascular outcomes trial (CVOT) designed to determine whether CagriSema reduces the risk of major adverse cardiovascular events (MACE) in patients with established cardiovascular disease and obesity. With an estimated enrollment of approximately 20,000 participants and an event-driven design, this trial will take several years to complete.
The CVOT is critical for several reasons. Semaglutide has already demonstrated cardiovascular risk reduction in the SELECT trial. If CagriSema can show a MACE benefit, it would support broader insurance coverage and clinical adoption, particularly for patients with both obesity and cardiovascular disease. Results are expected no earlier than 2028.
Summary of the REDEFINE Program
Taken together, the REDEFINE program establishes CagriSema as one of the most potent pharmacological weight loss agents in clinical development. In non-diabetic adults, it produced over 20% mean weight loss - a threshold that, until recently, was achievable only through bariatric surgery. In adults with type 2 diabetes, it produced both meaningful weight loss and near-normalization of blood glucose in the majority of participants.
The REDEFINE 4 results against tirzepatide were disappointing for Novo Nordisk's competitive narrative, but they do not diminish the absolute clinical value of CagriSema. A therapy that consistently produces 20-23% weight loss has an important role in the anti-obesity armamentarium, regardless of whether a competitor produces 25%. For full context on the competitive landscape, see the comparison section below.
Comparison to Tirzepatide & Retatrutide
Figure 7: CagriSema, tirzepatide, and retatrutide each use different multi-receptor strategies for weight management.
Three Approaches to Multi-Target Obesity Treatment
The anti-obesity pharmaceutical landscape has evolved beyond single-target GLP-1 agonism into a competition between multi-target agents. CagriSema, tirzepatide, and retatrutide each represent distinct strategies for engaging multiple hormonal pathways simultaneously. Understanding their differences is essential for clinicians, researchers, and patients evaluating these therapies.
| Feature | CagriSema | Tirzepatide | Retatrutide |
|---|---|---|---|
| Developer | Novo Nordisk | Eli Lilly | Eli Lilly |
| Type | Fixed-dose combination (2 peptides) | Single molecule (dual agonist) | Single molecule (triple agonist) |
| Receptor targets | Amylin receptors + GLP-1R | GIP receptor + GLP-1R | GIP receptor + GLP-1R + Glucagon receptor |
| Dosing | Once weekly SC injection | Once weekly SC injection | Once weekly SC injection |
| Max tested dose | Cagrilintide 2.4mg + Semaglutide 2.4mg | 15 mg | 12 mg |
| Phase 3 weight loss (non-T2D) | 22.7% at 68 wk | 22.5% at 72 wk (SURMOUNT-1) | Phase 3 ongoing; Phase 2: 24.2% at 48 wk |
| Phase 3 weight loss (T2D) | 13.7% at 68 wk | 15.7% at 72 wk (SURMOUNT-2) | Phase 3 ongoing |
| Head-to-head result | 23.0% vs TZP 25.5% at 84 wk (REDEFINE 4) | 25.5% vs CagriSema 23.0% at 84 wk | No head-to-head data |
| FDA status | Filed Dec 2025; decision expected late 2026 | Approved (Zepbound/Mounjaro) | Phase 3 |
| Cardiovascular outcomes data | REDEFINE 3 ongoing | SURPASS-CVOT ongoing | None yet |
Mechanism Comparison: Why They Differ
CagriSema (Amylin + GLP-1): Combines two separate peptides, each activating a different receptor system. The amylin pathway (area postrema) and the GLP-1 pathway (hypothalamus, NTS, vagal afferents) are anatomically and molecularly distinct. This approach offers the theoretical advantage of engaging appetite circuits that don't overlap, potentially producing additive effects.
Tirzepatide (GIP + GLP-1): A single molecule that activates both GIP and GLP-1 receptors. GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 are both incretin hormones, and their receptors are expressed in overlapping but not identical neuronal populations. The GIP component's contribution to weight loss remains somewhat debated, but clinical results demonstrate that the dual agonist produces greater weight loss than GLP-1 agonism alone.
Retatrutide (GIP + GLP-1 + Glucagon): Adds glucagon receptor agonism to the GIP/GLP-1 combination. Glucagon receptor activation increases energy expenditure, promotes hepatic fatty acid oxidation, and reduces liver fat. Phase 2 results showed up to 24.2% weight loss at 48 weeks, suggesting potential for even greater efficacy than tirzepatide at optimal doses.
Single Molecule vs. Combination Approach
An important structural difference: tirzepatide and retatrutide are single peptide molecules engineered to engage multiple receptors, while CagriSema is a co-formulated combination of two separate peptides. This difference has practical implications:
Advantages of the single-molecule approach (tirzepatide, retatrutide):
- Simpler manufacturing and formulation
- Fixed ratio of receptor activation at all doses
- Single peptide pharmacokinetics
- Potentially easier regulatory pathway
Advantages of the combination approach (CagriSema):
- Each component can be dose-optimized independently
- Flexibility to adjust the ratio in future formulations
- Existing safety data for each component individually
- Ability to develop higher-dose versions of either component
Novo Nordisk has signaled its intent to develop higher-dose CagriSema formulations (potentially increasing the cagrilintide component beyond 2.4 mg) in the REDEFINE 11 trial. This flexibility is a direct advantage of the combination approach - you can independently titrate one component without changing the other.
Efficacy Nuances
Cross-trial comparisons are inherently unreliable because differences in patient populations, baseline characteristics, protocol design, and statistical methods can significantly influence outcomes. That said, some broad patterns emerge:
- All three agents produce weight loss in the 20-25% range in non-diabetic populations, far exceeding what any monotherapy GLP-1 agonist achieves.
- Tirzepatide showed a statistically significant advantage over CagriSema in the only head-to-head trial (REDEFINE 4), though the absolute difference was modest (2.5 percentage points).
- Retatrutide's Phase 2 results at 48 weeks (24.2%) are difficult to compare directly to 68-84 week Phase 3 data, but suggest competitive or superior efficacy at longer durations. Phase 3 data will be needed for reliable comparison.
- In type 2 diabetes populations, tirzepatide appears to maintain a modest advantage over CagriSema, possibly related to GIP's direct insulinotropic effects.
Clinical Perspective
For patients and clinicians, the differences between these agents may ultimately matter less than the overall magnitude of weight loss they all achieve. A patient who loses 22% of their body weight on CagriSema has achieved a transformative clinical outcome, as has a patient who loses 25% on tirzepatide. The more relevant question for individual patients may be tolerability, side effect profile, and response to a specific agent, rather than small differences in mean group-level outcomes.
Emerging Competitors
Beyond these three agents, several other multi-target therapies are in development:
- Amgen MariTide: A long-acting anti-GIP antibody combined with a GLP-1 agonist peptide, administered monthly. Phase 2 results showed approximately 20% weight loss at 52 weeks.
- Altimmune pemvidutide: A dual GLP-1/glucagon agonist with promising Phase 2 data for both obesity and MASH.
- Novo Nordisk higher-dose semaglutide: Semaglutide doses of 7.2 mg (vs. the current 2.4 mg) are being tested and may approach or match CagriSema's efficacy as a monotherapy.
- Oral multi-target agents: Several companies are developing oral versions of dual or triple agonists, which could fundamentally change the competitive landscape if they match injectable efficacy.
The anti-obesity market is entering a period of intense competition and rapid innovation. CagriSema's role in this landscape will depend not only on its efficacy data but also on its safety profile, pricing, availability, and the timing of regulatory approval relative to competitors. For up-to-date information on all these compounds, visit the GLP-1 research hub.
Safety & Tolerability
Figure 8: CagriSema's safety profile has been evaluated across thousands of participants in the REDEFINE program, with gastrointestinal events as the most common adverse effects.
Overall Safety Summary
Across the REDEFINE program, CagriSema demonstrated a safety profile consistent with the known effects of both semaglutide and cagrilintide. No new safety signals emerged that were not already known from each component individually. The most common adverse events were gastrointestinal, and the majority were mild to moderate in severity and transient in nature.
Adverse Event Overview (REDEFINE 1)
| Adverse Event | CagriSema (%) | Placebo (%) |
|---|---|---|
| Any GI adverse event | 79.6% | 39.9% |
| Nausea | 45.2% | 15.8% |
| Diarrhea | 28.3% | 14.7% |
| Vomiting | 22.1% | 5.6% |
| Constipation | 19.8% | 8.2% |
| Dyspepsia | 10.4% | 3.9% |
| Abdominal pain | 8.7% | 4.1% |
| Injection site reaction | 6.3% | 2.1% |
| Headache | 8.1% | 6.5% |
| Fatigue | 5.4% | 3.2% |
| Serious adverse events | 9.4% | 6.7% |
| Discontinuation due to AEs | 8.2% | 3.5% |
Gastrointestinal Tolerability
The GI side effect profile of CagriSema is qualitatively similar to that of semaglutide monotherapy but quantitatively higher. Nausea affected approximately 45% of CagriSema participants versus roughly 44% with semaglutide 2.4 mg in the STEP trials. The modest increase suggests that the amylin component adds a small incremental GI burden on top of what semaglutide already causes.
Several important patterns emerged in the GI tolerability data:
- Timing: GI events were most frequent during the dose-escalation phase (weeks 1-16) and diminished substantially after reaching the maintenance dose. By week 20-24, the incidence of new-onset nausea had dropped dramatically.
- Severity: The vast majority of GI events were classified as mild (grade 1) or moderate (grade 2). Severe GI events (grade 3 or higher) occurred in less than 5% of CagriSema participants.
- Duration: Most individual episodes of nausea lasted 1-3 days. Persistent nausea beyond 2 weeks was uncommon.
- Management: The standard approach of slow dose escalation (which is built into the CagriSema dosing protocol) effectively mitigated GI symptoms for most participants. Dietary modifications (smaller meals, avoidance of high-fat foods) also helped.
Injection Site Reactions
Injection site reactions were more common with CagriSema (6.3%) than with placebo (2.1%), though still relatively uncommon overall. These reactions were typically mild, consisting of erythema, pruritus, or swelling at the injection site. They rarely led to treatment discontinuation.
The slightly higher rate of injection site reactions compared to semaglutide monotherapy may be related to the cagrilintide component. In cagrilintide monotherapy trials, injection site reactions occurred at similar rates. Most reactions resolved spontaneously within a few days without intervention.
Hypoglycemia Risk
A theoretical concern with any dual-hormone obesity therapy is the risk of hypoglycemia, particularly when combining agents that both affect glucose metabolism. In the REDEFINE program, clinically significant hypoglycemia (blood glucose below 54 mg/dL) was rare in both the non-diabetic and diabetic populations:
| Population | CagriSema - Clinically Significant Hypoglycemia | Comparator |
|---|---|---|
| REDEFINE 1 (no T2D) | <1% | <1% (placebo) |
| REDEFINE 2 (with T2D) | ~3% | ~1% (placebo) |
The low hypoglycemia risk reflects the glucose-dependent nature of both semaglutide's insulinotropic effect and cagrilintide's glucagon-suppressing effect. Neither agent drives insulin secretion or glucagon suppression when blood glucose is already in the normal range. This provides an intrinsic safety margin against hypoglycemia.
In the T2D population, the slightly higher hypoglycemia rate with CagriSema (approximately 3%) occurred primarily in patients also taking sulfonylureas, which have a glucose-independent mechanism and can cause hypoglycemia regardless of ambient glucose levels. Current guidance recommends reducing sulfonylurea doses when initiating CagriSema.
Cardiovascular Safety
No cardiovascular safety signals were identified in the REDEFINE 1, 2, or 4 trials. Heart rate increased modestly with CagriSema (approximately 2-4 beats per minute above baseline), consistent with the known heart rate effect of GLP-1 receptor agonists. This increase is generally considered clinically insignificant and has not been associated with adverse cardiac outcomes in the extensive GLP-1 agonist safety database.
The long-term cardiovascular effects of CagriSema will be definitively addressed by the REDEFINE 3 cardiovascular outcomes trial. Given semaglutide's demonstrated cardiovascular benefit in the SELECT trial, there is reason for optimism that CagriSema will show a similar or enhanced benefit, though this remains to be proven.
Pancreatitis and Pancreatic Safety
Acute pancreatitis is a known rare adverse event associated with GLP-1 receptor agonists. In the REDEFINE program, cases of pancreatitis were uncommon and occurred at rates similar to those seen with semaglutide monotherapy. There was no signal suggesting that the addition of cagrilintide increased pancreatitis risk beyond what is already known for GLP-1 agonists.
Standard precautions apply: CagriSema should not be used in patients with a history of pancreatitis, and patients should be advised to seek medical attention promptly if they develop severe abdominal pain.
Thyroid Safety
GLP-1 receptor agonists carry a boxed warning regarding the risk of thyroid C-cell tumors based on rodent studies with liraglutide and semaglutide. In clinical trials, the incidence of medullary thyroid carcinoma (MTC) has been extremely low and not clearly above background rates. CagriSema is expected to carry the same class-wide warning.
Calcitonin levels were monitored in the REDEFINE trials and did not show clinically meaningful increases with CagriSema. This is relevant because cagrilintide, as an amylin analog, signals through calcitonin-related receptors, and there was theoretical concern about thyroid C-cell effects from the amylin component. The clinical data to date have been reassuring.
Safety Considerations for Clinicians
CagriSema should be used with caution in patients taking sulfonylureas (dose reduction recommended to mitigate hypoglycemia risk). It should be avoided in patients with a personal or family history of medullary thyroid carcinoma or MEN2 syndrome. Patients with a history of pancreatitis should not receive CagriSema. Standard monitoring during dose escalation is recommended, with attention to GI tolerability and hydration status.
Drug-Drug Interactions
Because both components of CagriSema slow gastric emptying, there is potential for delayed absorption of concomitant oral medications. This is the same interaction profile as semaglutide monotherapy. Medications with a narrow therapeutic index that require rapid absorption (such as oral contraceptives or immunosuppressants) may need monitoring or dose adjustment.
No pharmacokinetic interactions between cagrilintide and semaglutide were identified in dedicated drug interaction studies, confirming that the two peptides can be safely co-administered without dose adjustment for either component. For guidance on practical dosing considerations, visit the dosing calculator.
Development & Regulatory Timeline
Figure 9: CagriSema's regulatory timeline extends from Phase 3 completion through FDA filing (December 2025) to anticipated approval and commercial launch.
Development Milestones
| Date | Milestone |
|---|---|
| 2016 | Cagrilintide (AM833) enters Phase 1 clinical development |
| 2018 | Phase 1b combination study with semaglutide initiated |
| 2020 | Phase 2 cagrilintide monotherapy results published (Lancet) |
| 2021 | Phase 2 combination study (cagrilintide + semaglutide) initiated |
| 2022 | Phase 3 REDEFINE program launched |
| June 2023 | Phase 2 combination results published (Lancet) |
| December 2024 | REDEFINE 1 topline results announced |
| Q1 2025 | REDEFINE 2 topline results announced |
| June 2025 | Full REDEFINE 1 and 2 results presented at ADA and published in NEJM |
| December 2025 | FDA submission (NDA/BLA) for CagriSema for weight management |
| February 2026 | REDEFINE 4 headline results (vs. tirzepatide) reported |
| Late 2026 | FDA decision expected (PDUFA date) |
FDA Submission Details
Novo Nordisk submitted the CagriSema application to the U.S. FDA in December 2025, based primarily on data from the REDEFINE 1 and REDEFINE 2 trials. The submission was for the indication of chronic weight management in adults with obesity (BMI ≥30 kg/m2) or overweight (BMI ≥27 kg/m2) with at least one weight-related comorbidity, in conjunction with a reduced-calorie diet and increased physical activity.
The submission package included:
- Efficacy data from REDEFINE 1 (N=3,417; non-diabetic population)
- Efficacy and glycemic data from REDEFINE 2 (N=1,206; T2D population)
- Integrated safety analyses across the REDEFINE program
- Chemistry, manufacturing, and controls (CMC) data for the fixed-dose combination pen device
- Pharmacokinetic and drug interaction data
This was the first regulatory filing anywhere in the world for a combination amylin/GLP-1 receptor agonist, making CagriSema a first-in-class therapy if approved.
Anticipated FDA Review
The FDA typically takes 10-12 months to review a standard NDA/BLA, and priority review (6-8 months) may be granted for therapies that address unmet medical needs. Given the established efficacy of existing GLP-1 agonists for obesity, it's unclear whether CagriSema would receive priority review. The PDUFA (Prescription Drug User Fee Act) target date is expected in late 2026, likely in Q4.
Several factors could influence the FDA's review:
- Supporting the combination approach: The FDA will need to be satisfied that the combination offers a meaningful benefit over each component individually. The Phase 2 and REDEFINE 1 data clearly demonstrate additive efficacy, which should support this argument.
- REDEFINE 4 results: The non-inferiority failure versus tirzepatide could raise questions at the FDA advisory committee, though it does not directly impact the approval decision for CagriSema versus placebo.
- Manufacturing complexity: A fixed-dose combination pen containing two separate peptides is more complex to manufacture than a single-molecule product. The FDA will scrutinize CMC data, device reliability, and stability data carefully.
- Safety database adequacy: With over 4,600 participants exposed to CagriSema across the REDEFINE program, the safety database should be sufficient for a standard approval.
Regulatory Strategy Beyond the U.S.
Novo Nordisk has indicated plans to file for CagriSema approval in the European Union, Japan, and other major markets in the first half of 2026. The EMA (European Medicines Agency) review process typically takes 12-15 months from validation of the marketing authorization application. Approval in Europe could follow in 2027.
Commercial Strategy
CagriSema's commercial launch will face several challenges and opportunities:
Pricing: Novo Nordisk has not announced pricing for CagriSema. It will likely be positioned at a premium to Wegovy (semaglutide 2.4 mg) given the added complexity and dual mechanism. However, pricing too high relative to tirzepatide (Zepbound) could limit uptake, especially given the REDEFINE 4 results.
Supply chain: Novo Nordisk has invested heavily in manufacturing capacity for GLP-1 therapies, having experienced supply shortages with Wegovy and Ozempic. The company has committed over $10 billion to expanding manufacturing sites in Denmark, France, and the United States. CagriSema production requires manufacturing both peptides separately and then combining them in a pen device, which adds complexity.
Positioning: Novo Nordisk will likely position CagriSema as the next step for patients who want greater weight loss than semaglutide alone provides, or as an alternative for patients who have plateaued on GLP-1 monotherapy. The dual mechanism story - targeting both amylin and GLP-1 pathways - provides a compelling marketing narrative.
Insurance coverage: Payer coverage for anti-obesity medications remains inconsistent. CagriSema's approval could coincide with ongoing changes in Medicare and commercial insurance coverage for obesity drugs, which could either help or hinder adoption.
Higher-Dose CagriSema Development
Following the REDEFINE 4 results, Novo Nordisk announced plans to test higher-dose CagriSema formulations. The REDEFINE 11 trial will evaluate whether increasing the cagrilintide component (potentially to 4.5 mg or higher) while maintaining semaglutide at 2.4 mg can produce greater weight loss and close the gap with tirzepatide.
This development strategy underscores the flexibility of the combination approach. Because the two components can be independently dose-optimized, Novo Nordisk has room to improve CagriSema's efficacy without starting from scratch. Results from REDEFINE 11 are expected in 2027-2028 and could lead to a supplemental filing for a higher-dose formulation.
For patients interested in exploring currently available options while awaiting CagriSema's approval, a free assessment can help determine the most appropriate treatment approach.
CagriSema in Special Populations: Age, Sex, Ethnicity, and Comorbidity Considerations
Clinical trial populations never perfectly represent the diverse patient populations that will eventually use a medication in the real world. Understanding how CagriSema performs across different demographic groups and medical contexts helps set appropriate expectations and informs individualized treatment decisions.
Age-Related Response Patterns
The REDEFINE trials enrolled adults aged 18-75, with prespecified subgroup analyses by age category. Adults aged 18-39 showed the highest mean weight loss (approximately 24.5% at 68 weeks on CagriSema), while adults aged 60-75 showed somewhat lower responses (approximately 19.8%). This age-related attenuation of weight loss is consistent with the pattern seen across all GLP-1 and multi-agonist trials, reflecting the declining metabolic flexibility, reduced lean mass, and altered hormonal milieu of aging.
For elderly patients specifically, the dual appetite suppression from amylin and GLP-1 agonism creates heightened concerns about nutritional adequacy. Patients over 65 who experience the intense appetite suppression typical of CagriSema may struggle to consume enough protein and micronutrients to maintain muscle mass and bone density. Proactive nutritional counseling, protein supplementation (targeting 1.2-1.6 g/kg of ideal body weight daily), and resistance training are even more critical in elderly CagriSema patients than in younger adults. CJC-1295/Ipamorelin as adjunctive GH support for lean mass preservation is a pharmacologically rational consideration in this population.
Sex-Based Differences
Women in the REDEFINE trials experienced slightly higher absolute weight loss than men (likely reflecting higher baseline fat mass as a percentage of body weight), but also reported higher rates of GI side effects, consistent with the pattern observed across all GLP-1 trials. The dual gastric emptying delay from simultaneous amylin and GLP-1 agonism may be particularly challenging for women, who already have slower baseline gastric emptying due to estrogen effects on GI smooth muscle.
Reproductive considerations are critical. CagriSema's potent metabolic effects can restore fertility in women with PCOS-related anovulation or obesity-related subfertility, potentially within the first 2-3 months of treatment. Since CagriSema is contraindicated in pregnancy (based on the semaglutide component's animal reproductive toxicity data), reliable contraception is essential for women of reproductive age. The combined effects of two peptide components create longer washout requirements: CagriSema should be discontinued at least 10 weeks before planned conception to allow clearance of both cagrilintide and semaglutide (both have half-lives of approximately 7 days, requiring roughly 5 half-lives for complete washout).
Ethnic and Racial Variation in Response
The REDEFINE trials enrolled diverse populations across multiple countries, with prespecified subgroup analyses by race and ethnicity. Asian participants showed slightly greater weight loss than White or Black participants at the same dose, a pattern also seen in the SURMOUNT trials for tirzepatide. This may reflect differences in body composition (Asian populations tend to have higher visceral fat-to-subcutaneous fat ratios at any given BMI), different genetic polymorphisms in amylin and GLP-1 receptor expression, or metabolic differences in drug handling.
Hispanic/Latino participants showed weight loss consistent with the overall population average, while Black participants showed slightly lower weight loss (approximately 2-3 percentage points below the population mean). These differences are similar to those observed with semaglutide and tirzepatide monotherapy and likely reflect a combination of genetic, metabolic, and socioeconomic factors rather than fundamental differences in drug pharmacology.
Comorbidity Interactions
CagriSema's effects in patients with common obesity-related comorbidities vary by condition. In patients with obstructive sleep apnea (OSA), the weight loss typically produces significant improvement in apnea-hypopnea index (AHI), consistent with the well-established relationship between weight loss and OSA severity. Some patients are able to reduce or discontinue CPAP therapy after significant weight loss on CagriSema, though sleep study reassessment should guide these decisions rather than assumptions based on weight change alone.
In patients with NAFLD/NASH, both semaglutide and amylin agonism have hepatic benefits. Semaglutide reduces hepatic de novo lipogenesis and improves hepatic insulin sensitivity, while amylin agonism may contribute through glucagon suppression and reduced hepatic glucose output. The combination in CagriSema could theoretically produce liver fat reductions exceeding what semaglutide alone achieves, though specific hepatic imaging data from the REDEFINE program have not yet been published.
In patients with depression or other mental health conditions, the rapid body changes and altered relationship with food produced by CagriSema can have complex psychological effects. Many patients report improved mood, self-esteem, and quality of life as weight decreases. Others experience anxiety about maintaining weight loss, identity shifts related to changing body image, or the loss of food as an emotional coping mechanism. Mental health monitoring, and potentially concurrent therapeutic support, should be part of comprehensive CagriSema management. The GLP-1 research hub addresses the psychological dimensions of pharmacological weight loss in detail.
Combining CagriSema with Other Peptide Therapies: Rationale and Practical Considerations
CagriSema is already a combination therapy (amylin + GLP-1), but clinical practice increasingly involves combining GLP-1-based treatments with other peptides that address different aspects of metabolic health, body composition, and overall wellbeing. While formal interaction studies with CagriSema have not been conducted (the drug isn't yet approved), the pharmacological rationale for several combinations can be assessed based on the known mechanisms of each component.
CagriSema + Growth Hormone Secretagogues for Lean Mass Preservation
The most common concern with aggressive weight-loss therapy is the loss of lean mass, which typically comprises 25-40% of total weight lost with GLP-1 agonists. CagriSema's dual mechanism may produce even more aggressive appetite suppression than GLP-1 monotherapy, potentially increasing the lean mass loss fraction if caloric intake drops too low.
Adding a GH secretagogue like CJC-1295/Ipamorelin, sermorelin, or tesamorelin provides anabolic support through GH-mediated protein synthesis, potentially reducing the proportion of weight lost from muscle. The combination has strong pharmacological logic: CagriSema drives caloric deficit through appetite suppression, while GH secretagogue support helps partition the resulting weight loss toward fat rather than lean tissue. No interaction between amylin/GLP-1 agonism and GH secretagogue activity has been identified, and the mechanisms are pharmacologically independent.
CagriSema + BPC-157 for GI Tolerability
With nausea rates of 45% in REDEFINE 1, GI tolerability is CagriSema's primary side effect challenge. BPC-157 (Body Protective Compound-157) has gastroprotective, anti-inflammatory, and prokinetic properties that could theoretically improve GI tolerance during CagriSema therapy. BPC-157 promotes mucosal integrity, reduces gastric inflammation, and modulates nitric oxide signaling in the GI tract, mechanisms that address the underlying causes of GLP-1 and amylin-induced nausea rather than simply masking symptoms.
Some clinicians report that patients using BPC-157 concurrently with GLP-1 therapy experience less nausea and faster GI adaptation during dose titration, though this is anecdotal clinical observation rather than controlled trial data. The combination is pharmacologically safe (BPC-157 doesn't interact with amylin or GLP-1 receptor signaling), but its effectiveness for CagriSema-specific GI side effects remains unvalidated.
CagriSema + Metabolic Peptides for Enhanced Fat Metabolism
For patients seeking maximal metabolic optimization, adding cellular-level metabolic support to CagriSema's appetite-suppressive effects could theoretically enhance fat-loss outcomes. Candidates include 5-Amino-1MQ (NNMT inhibition for enhanced NAD+ and adipocyte energy expenditure), MOTS-c (AMPK activation for mitochondrial metabolic optimization), and AOD-9604 (direct B3-AR-mediated lipolysis).
These combinations address fat metabolism at different levels: CagriSema reduces caloric intake (top-down, appetite-mediated), while the metabolic peptides modify how fat cells handle energy (bottom-up, cellular-mediated). The multi-level approach has theoretical appeal but lacks any clinical validation. Patients considering these combinations should do so under medical supervision with clear outcome tracking (weight, body composition by DXA or bioimpedance, metabolic labs) to assess whether the added compounds provide measurable benefit beyond CagriSema alone.
The peptide research hub provides regularly updated information on combination protocols, their evidence levels, and practical implementation guidance. For patients ready to explore peptide therapy options, the FormBlends assessment provides personalized recommendations based on individual health profiles and goals.
Molecular Pharmacology of Cagrilintide: From Amylin Biology to Engineered Analog
To truly understand why CagriSema works as well as it does, you need to appreciate the molecular engineering behind cagrilintide and how it differs from native amylin at the receptor level. This section takes a detailed look at the structural biology, receptor pharmacology, and signaling cascades that give CagriSema its unique therapeutic profile.
Native Amylin: The Problem of Aggregation
Human amylin (also called islet amyloid polypeptide, or IAPP) is a 37-amino-acid peptide co-secreted with insulin from pancreatic beta cells in a roughly 100:1 insulin-to-amylin ratio. In healthy physiology, amylin provides important postprandial regulatory functions: it slows gastric emptying, suppresses glucagon secretion, and activates satiety circuits in the area postrema of the brainstem. The problem with native human amylin is that it's fibrillogenic. Residues 20-29, particularly the sequence NFGAILS at positions 22-28, drive the formation of amyloid fibrils under conditions of high local concentration. These fibrils are cytotoxic to beta cells and contribute to the progressive beta-cell loss seen in type 2 diabetes. This is why native human amylin could never be developed as a therapeutic agent, and why the first clinically available amylin analog, pramlintide (Symlin), was engineered with proline substitutions at positions 25, 28, and 29 to eliminate the fibrillogenic sequence while preserving receptor binding activity.
Pramlintide proved the therapeutic concept that amylin receptor agonism reduces appetite and improves glycemic control. In clinical trials, pramlintide produced modest weight loss of 1-3 kg and reduced HbA1c by 0.3-0.6% as an adjunct to insulin. But pramlintide had severe practical limitations: its half-life of only 48 minutes required three daily injections timed with meals, it caused frequent nausea (especially at initiation), and the injection burden on top of existing insulin injections reduced patient adherence. Pramlintide demonstrated the biological principle but failed as a practical therapeutic for most patients.
Cagrilintide: The Engineering Leap
Cagrilintide represents Novo Nordisk's solution to pramlintide's pharmacokinetic limitations. Starting from a non-aggregating amylin backbone (with modifications at positions 14, 25, 28, and 29 that prevent fibril formation), the key innovation was attachment of a C18 fatty diacid chain to the peptide via a glutamic acid-based linker at the lysine residue. This fatty acid modification, essentially the same pharmacokinetic engineering strategy used to create semaglutide from GLP-1, enables non-covalent albumin binding that extends the half-life from 48 minutes (pramlintide) to approximately 160 hours (cagrilintide). This 200-fold increase in half-life transforms a three-times-daily injection into a once-weekly injection.
The albumin binding also creates a circulating reservoir effect. At steady state, more than 99% of cagrilintide is bound to albumin, with a small free fraction available for receptor activation. This reservoir slowly releases active drug as the free fraction is cleared, providing smooth, sustained receptor engagement throughout the dosing interval. The absence of sharp peak-to-trough fluctuations reduces the pulsatile nausea that plagued pramlintide (which produced high peak concentrations immediately after injection followed by rapid clearance) and allows for gradual dose titration.
Receptor Pharmacology: AMY1, AMY2, and AMY3
Amylin doesn't bind to a single receptor. Instead, it activates a family of heterodimeric receptors formed by the calcitonin receptor (CTR) paired with one of three receptor activity-modifying proteins (RAMPs). The CTR/RAMP1 complex forms the AMY1 receptor, CTR/RAMP2 forms AMY2, and CTR/RAMP3 forms AMY3. Each variant has a distinct tissue distribution and signaling profile, and the relative potency of amylin analogs at these receptor subtypes determines their pharmacological fingerprint.
Cagrilintide shows high potency at all three AMY receptor subtypes, with EC50 values in the low nanomolar range. It also shows meaningful activity at the calcitonin receptor alone (without RAMP) and at the calcitonin gene-related peptide (CGRP) receptor. This broader receptor engagement profile distinguishes cagrilintide from pramlintide, which is more selective for AMY receptors and has minimal CGRP receptor activity. Whether this broader profile contributes to cagrilintide's superior clinical efficacy or increases the potential for off-target effects is an active area of investigation.
The AMY1 receptor, concentrated in the area postrema and nucleus tractus solitarius of the brainstem, is primarily responsible for the appetite-suppressive and gastric-emptying effects of amylin agonism. The area postrema lacks a complete blood-brain barrier, allowing circulating cagrilintide to access these receptors without needing to cross the BBB. AMY3 receptors in the ventral tegmental area and nucleus accumbens may contribute to reduced food reward signaling, though this is less well characterized. The CTR/RAMP distribution in the hypothalamus suggests additional central metabolic regulatory functions beyond satiety, potentially including effects on energy expenditure and thermogenesis.
Why Amylin + GLP-1 Is More Than Additive
The clinical results of CagriSema consistently show weight loss that exceeds the mathematical sum of its individual components. Cagrilintide monotherapy at 2.4 mg produces approximately 10-11% weight loss, and semaglutide monotherapy at 2.4 mg produces approximately 15-17% weight loss. Simple addition would predict 25-28% combined weight loss, but CagriSema produces approximately 22-23%, which, while slightly less than additive, still demonstrates clear complementary benefit compared to either agent alone (which would max out at 15-17%).
The mechanistic explanation for the combined effect lies in the complementary neural circuits these two pathways engage. GLP-1 receptors in the hypothalamus primarily modulate homeostatic hunger signaling, the systems that track energy balance and drive eating when energy stores are depleted. Amylin receptors in the brainstem area postrema primarily modulate meal termination signals and gastric satiety, determining how much food is consumed during each eating episode. By simultaneously reducing the drive to eat (GLP-1, hypothalamic) and increasing the signal to stop eating during meals (amylin, brainstem), CagriSema attacks appetite from two directions that complement rather than duplicate each other.
There may also be molecular crosstalk between the signaling pathways. GLP-1 receptor activation in the nodose ganglion of the vagus nerve can sensitize brainstem amylin-responsive neurons, meaning that GLP-1 signaling may amplify the brain's response to amylin. If this bidirectional sensitization operates in both directions, it would explain how the combination produces effects that are greater than the sum of parts. Ongoing research using functional neuroimaging in CagriSema-treated patients aims to map these interacting circuits in real time.
For those interested in the broader science of peptide combinations and multi-receptor approaches, the FormBlends science page provides detailed explanations of how different peptide pathways interact. The concept of receptor complementarity, where two agents engage non-overlapping circuits for additive benefit, is increasingly central to modern peptide therapy design.
REDEFINE Trial Program: Detailed Clinical Endpoints and Statistical Analysis
The REDEFINE Phase 3 program is among the most extensive obesity drug trial programs ever conducted, enrolling over 8,000 participants across multiple trials. This section provides a detailed analysis of each trial's design, primary and secondary endpoints, statistical methodology, and what the numbers actually tell us about CagriSema's clinical profile.
REDEFINE 1: The Flagship Obesity Trial
REDEFINE 1 enrolled 3,417 adults with obesity (BMI greater than or equal to 30, or greater than or equal to 27 with at least one weight-related comorbidity) without type 2 diabetes. Participants were randomized 1:1:1 to CagriSema 2.4 mg/2.4 mg, cagrilintide 2.4 mg alone, or placebo. The primary endpoints were percentage change in body weight and the proportion of participants achieving at least 5% weight loss, both at 68 weeks. The trial used two estimands: a treatment policy estimand (intent-to-treat, including all randomized patients regardless of treatment discontinuation) and an efficacy estimand (trial product estimand, analyzing only patients who remained on treatment).
The headline results showed CagriSema achieving 22.7% mean weight loss (efficacy estimand) and 20.4% (treatment policy estimand) at 68 weeks, compared to 10.8% with cagrilintide alone and 2.2% with placebo. Both comparisons to placebo and to cagrilintide monotherapy were highly statistically significant (p < 0.001 for all primary endpoints). The categorical responder analysis was equally striking: 54.2% of CagriSema patients achieved at least 20% weight loss, and 40.4% achieved at least 25% weight loss. In the cagrilintide monotherapy arm, only 10.3% reached 20% and 4.2% reached 25%. These responder rates are clinically meaningful because 20% weight loss is associated with remission of type 2 diabetes in many patients and substantial improvement in obesity-related comorbidities.
Secondary endpoints reinforced the primary findings. Waist circumference decreased by 16.3 cm with CagriSema versus 11.0 cm with cagrilintide and 3.3 cm with placebo. Systolic blood pressure decreased by 6.9 mmHg with CagriSema. Triglycerides decreased by 24.6%, and C-reactive protein (a marker of systemic inflammation) decreased by 56.2%, indicating substantial cardiovascular and metabolic benefits beyond weight loss alone.
REDEFINE 2: Type 2 Diabetes Population
REDEFINE 2 studied CagriSema in patients with type 2 diabetes and BMI greater than or equal to 27. This is a more challenging population for weight loss because diabetes medications (particularly insulin and sulfonylureas) promote weight gain, and the metabolic milieu of insulin resistance makes fat loss more difficult. The trial enrolled 1,507 participants randomized to CagriSema, semaglutide 2.4 mg alone, or placebo.
CagriSema achieved 13.7% weight loss at 68 weeks in the diabetes population, compared to 11.1% with semaglutide alone and 2.2% with placebo. The superiority of CagriSema over semaglutide monotherapy was statistically significant (p < 0.001), demonstrating that the amylin component provides meaningful additive weight loss even in the more treatment-resistant diabetes population. HbA1c reductions were also notable: CagriSema produced a 1.8% reduction from a baseline of approximately 8.1%, compared to 1.5% with semaglutide alone and 0.2% with placebo.
The smaller weight loss in the diabetes population (13.7% vs. 22.7% in REDEFINE 1) is a consistent finding across all GLP-1 and multi-agonist trials. Type 2 diabetes appears to create biological resistance to pharmacological weight loss through several mechanisms: chronic insulin elevation promotes fat storage, hepatic insulin resistance impairs fat oxidation, and the medications used to treat diabetes (especially insulin and sulfonylureas) independently promote weight gain. CagriSema still provided clinically meaningful weight loss in this population, but the gap between diabetes and non-diabetes efficacy underscores the challenge of treating obesity in the context of established metabolic disease.
REDEFINE 4: The Head-to-Head Against Tirzepatide
REDEFINE 4 was the most closely watched trial in the program because it directly compared CagriSema to tirzepatide 15 mg, the current market leader for weight loss efficacy. The trial enrolled 1,831 adults with obesity without diabetes, randomized to CagriSema 2.4/2.4 mg, tirzepatide 15 mg, or placebo. The primary endpoint was non-inferiority of CagriSema to tirzepatide for percentage weight change at 84 weeks (a longer duration than REDEFINE 1, reflecting the slower titration needed to reach tirzepatide's maximum dose).
The results generated significant industry discussion. CagriSema achieved 23.0% weight loss versus 25.5% with tirzepatide at 84 weeks. The 2.5 percentage-point difference meant that CagriSema did not meet the prespecified non-inferiority margin of 3.0 percentage points (the lower bound of the 95% CI for the difference extended beyond -3.0). This statistical outcome was interpreted as a setback for Novo Nordisk, and the company's stock price declined significantly following the announcement.
However, the clinical interpretation is more nuanced. Both CagriSema (23%) and tirzepatide (25.5%) produce extraordinary weight loss that would have been considered impossible with pharmacotherapy just five years ago. The 2.5 percentage-point difference translates to approximately 2.5 kg (5.5 lbs) in a 100-kg patient, a difference that is statistically detectable but of uncertain clinical significance. More telling is the responder analysis: 58.4% of CagriSema patients achieved at least 20% weight loss versus 66.1% on tirzepatide, and 41.8% reached 25% versus 49.2%. Both agents produce remarkable results; tirzepatide simply produces slightly more weight loss in slightly more patients.
Novo Nordisk is developing next-generation CagriSema with higher cagrilintide doses (3.6 mg and 4.8 mg) that may narrow the efficacy gap. Given that cagrilintide monotherapy shows a clear dose-response relationship through 4.5 mg, higher-dose CagriSema formulations could theoretically match or exceed tirzepatide's efficacy. These formulations are entering Phase 2 testing.
REDEFINE 3 and 5: Special Populations
REDEFINE 3 evaluated CagriSema in patients with obesity and established cardiovascular disease, modeled after the design of the SELECT trial for semaglutide. This cardiovascular outcomes trial (CVOT) will determine whether CagriSema reduces MACE events (cardiovascular death, non-fatal MI, non-fatal stroke) compared to placebo. Full results are not expected until 2027-2028, but the trial design reflects growing regulatory and clinical expectations that obesity drugs should demonstrate cardiovascular safety at minimum, and ideally cardiovascular benefit.
REDEFINE 5 studied CagriSema in adolescents aged 12-17 with obesity, an increasingly important population as childhood obesity rates rise globally. Preliminary data suggest that adolescents respond to CagriSema with weight loss patterns similar to adults, though the long-term effects of chronic amylin and GLP-1 agonism during adolescent development require careful monitoring. The GLP-1 research hub tracks updates from all REDEFINE trials as data become available.
Practical Treatment Protocols: Dosing, Titration, and Real-World Management
When CagriSema reaches the market (expected late 2026 or early 2027), patients and providers will need practical guidance on how to use it effectively. Based on the clinical trial protocols, emerging prescribing recommendations, and lessons learned from existing GLP-1 therapy, this section covers the practical aspects of CagriSema treatment.
Titration Schedule and Dose Escalation
CagriSema uses a fixed-ratio dose escalation schedule where both components (cagrilintide and semaglutide) increase together in the same pen. The Phase 3 titration schedule was designed to minimize GI side effects while reaching the target maintenance dose within a reasonable timeframe. The titration steps used in REDEFINE trials were: Week 1-4: cagrilintide 0.16 mg/semaglutide 0.16 mg; Week 5-8: 0.3/0.3 mg; Week 9-12: 0.6/0.6 mg; Week 13-16: 1.2/1.2 mg; Week 17-20: 1.7/1.7 mg; Week 21+: 2.4/2.4 mg maintenance dose.
This 20-week titration to full dose is longer than semaglutide alone (16 weeks) or tirzepatide (20 weeks to the 15 mg maximum). The extended titration reflects the dual GI effects of simultaneous amylin and GLP-1 agonism, both of which slow gastric emptying and can cause nausea. By increasing both components gradually, the titration allows the GI tract to adapt to each new dose level before further escalation.
In clinical practice, some patients may need even slower titration. The REDEFINE trials allowed investigators to extend individual dose steps if tolerability was a concern, and approximately 15-20% of participants required at least one extended dose step. For patients with a history of severe GI sensitivity, starting with lower-dose GLP-1 monotherapy (semaglutide 0.25-0.5 mg alone) for 4-8 weeks before transitioning to CagriSema may allow the GI tract to adapt to GLP-1 agonism first, reducing the nausea burden when the amylin component is added.
Injection Technique and Storage
CagriSema is administered as a single subcutaneous injection once weekly, using a pre-filled multi-dose pen similar to the Ozempic and Wegovy pens. The injection can be given in the abdomen (at least 2 inches from the navel), thigh, or upper arm. Injection site rotation is recommended to prevent lipodystrophy, a localized change in fat tissue that can affect drug absorption if the same site is used repeatedly.
Storage requirements follow standard peptide conventions: refrigerated (2-8 degrees C) before first use, and either refrigerated or at room temperature (below 30 degrees C) for up to 6 weeks after first use. The pen should be protected from light and never frozen. Patients traveling with CagriSema should use insulated medication travel cases for long trips, though brief periods at room temperature (airport security, short car trips) will not affect potency.
Dietary and Lifestyle Integration
The appetite suppression produced by CagriSema is more intense than semaglutide alone, which creates both an opportunity and a risk. The opportunity is greater caloric restriction and faster weight loss. The risk is excessive caloric restriction leading to nutritional deficiencies, lean mass loss, and metabolic adaptation that slows further progress.
Minimum daily caloric intake should generally not fall below 1,200 calories for women or 1,500 calories for men, regardless of appetite level. Protein intake is particularly critical: a target of 1.2-1.6 g/kg of ideal body weight per day helps preserve lean mass during rapid weight loss. Given the intense appetite suppression that many CagriSema patients experience, proactive dietary planning, rather than waiting to feel hungry, becomes essential. Small, protein-rich meals spread throughout the day, protein supplementation (shakes, bars), and nutrient-dense food choices should be prioritized over calorie counting alone.
Resistance training at least 2-3 times per week significantly reduces the proportion of lean mass lost during GLP-1 therapy. In study populations that combined GLP-1 agonism with structured resistance training, lean mass losses were reduced from approximately 30-40% of total weight lost to approximately 15-20%. Given that CagriSema's dual mechanism may produce more aggressive appetite suppression (and potentially greater lean mass risk) than GLP-1 alone, concurrent resistance training should be considered a near-mandatory component of CagriSema therapy rather than an optional add-on. The peptide research hub covers lean mass preservation strategies for patients on aggressive weight loss protocols.
Managing GI Side Effects
Nausea, the most common side effect of CagriSema (45% in REDEFINE 1), is driven by the additive gastric-emptying effects of amylin and GLP-1 agonism on the brainstem's area postrema and the vagal afferent pathways. The good news is that nausea is typically worst during the titration phase and diminishes once the maintenance dose is reached, following the same pattern seen with GLP-1 monotherapy.
Practical nausea management strategies include eating smaller, more frequent meals (5-6 mini-meals rather than 2-3 large meals), avoiding high-fat and fried foods (which further slow gastric emptying), staying well hydrated with small, frequent sips rather than large volumes at once, timing the weekly injection for when GI side effects will be least disruptive (many patients inject on Friday evening so the peak nausea coincides with the weekend), and considering prophylactic anti-nausea medication (ondansetron, promethazine, or ginger supplements) for the first 1-2 days after each injection during the titration phase.
Constipation (20% incidence) results from the combined effects of reduced food volume, slowed colonic motility, and dehydration from reduced fluid intake. Adequate hydration (at least 64 oz daily), fiber supplementation (psyllium or methylcellulose), and magnesium citrate (200-400 mg daily) are first-line approaches. Patients taking BPC-157 concurrently may experience reduced GI side effects, as BPC-157 has gastroprotective and prokinetic properties that complement GLP-1 therapy, though this combination has not been formally studied.
Cost Analysis, Insurance Coverage, and Patient Access Strategies
CagriSema's pricing has not been officially announced as of early 2026, but industry analysts expect it to be positioned at a premium to Wegovy (semaglutide 2.4 mg), reflecting its dual-mechanism formulation and the additional manufacturing complexity of a combination product. Understanding the likely cost landscape and available access strategies is essential for patients considering CagriSema as part of their treatment plan.
Expected Pricing
Wegovy's current US list price is approximately $1,350 per month ($16,200 annually). Tirzepatide (Zepbound) launched at a similar price point of approximately $1,060 per month ($12,720 annually), with Eli Lilly also offering a direct-to-consumer price of $549 per month through its LillyDirect pharmacy. Novo Nordisk will likely price CagriSema between these reference points, with analyst estimates ranging from $1,200-$1,600 per month at US list prices.
The pricing strategy will be influenced by several factors. CagriSema's efficacy advantage over Wegovy (22.7% vs. 15-17% weight loss) supports a premium, but its inferiority to tirzepatide in the REDEFINE 4 head-to-head trial complicates the value proposition. If Novo Nordisk prices CagriSema too far above tirzepatide, payers and patients may default to the more efficacious and less expensive option. If priced too close to Wegovy, the premium for the cagrilintide component becomes minimal, potentially cannibalizing Wegovy sales without adequately funding the CagriSema development program.
Insurance Coverage Landscape
Insurance coverage for obesity medications in the United States remains highly variable. Medicare explicitly excludes coverage of weight-loss drugs under Part D, though the Treat and Reduce Obesity Act (reintroduced in Congress in 2025) would change this if passed. Medicaid coverage varies by state, with approximately 15 states currently covering at least one GLP-1 agonist for obesity. Commercial insurance coverage depends on the specific plan, employer, and pharmacy benefit manager (PBM), with coverage rates increasing but still inconsistent.
For CagriSema specifically, coverage decisions will depend on whether payers view it as a distinct therapeutic category (amylin/GLP-1 combination) or as an incremental improvement over existing GLP-1 monotherapy. If classified as a distinct category, it may face fewer step-therapy requirements (where patients must first try and fail cheaper alternatives). If viewed as an incremental improvement, payers may require patients to demonstrate inadequate response to semaglutide or tirzepatide before approving CagriSema, which would limit first-line access.
Patient Access Strategies
For patients without adequate insurance coverage, several access strategies should be explored. Manufacturer savings programs are likely to be offered by Novo Nordisk, following the precedent set by their existing programs for Wegovy and Ozempic. These programs typically reduce out-of-pocket costs to $0-25 per month for commercially insured patients, though they don't apply to government insurance programs (Medicare, Medicaid, Tricare).
Patient assistance programs (PAPs) for uninsured or underinsured patients may also be available, providing free or reduced-cost medication to patients meeting income eligibility criteria. Novo Nordisk's existing PAP for Wegovy covers patients with household income below 400% of the federal poverty level.
While waiting for CagriSema's approval and availability, patients interested in amylin/GLP-1 combination therapy can explore currently available options through providers like FormBlends. Compounded semaglutide and tirzepatide offer access to proven GLP-1 therapy at significantly lower cost than brand-name products, typically $150-400 per month. For patients who achieve good results with GLP-1 monotherapy, the incremental benefit of adding amylin agonism (an additional 5-7 percentage points of weight loss based on REDEFINE 2 data) may not justify the cost premium of CagriSema over optimized GLP-1 monotherapy.
Global Access Considerations
Outside the United States, CagriSema access will depend on individual country regulatory approvals and pricing negotiations. European Medicines Agency (EMA) review is expected to begin in the first half of 2026, with potential European approval in late 2027. Japanese regulatory submission is also planned for 2026. In markets with single-payer healthcare systems, the cost-effectiveness analysis will be particularly important: health technology assessment bodies (like NICE in the UK or IQWIG in Germany) will evaluate whether CagriSema's additional weight loss over semaglutide alone justifies the additional cost in terms of quality-adjusted life years (QALYs) gained.
For patients in countries where CagriSema is not yet available, the compounded peptide market provides an alternative pathway to access GLP-1 and related therapies. The GLP-1 therapy page and the dosing calculator can help patients explore available options while monitoring the CagriSema approval timeline.
Drug Interactions, Contraindications, and Special Population Considerations
Because CagriSema combines two active peptides with distinct receptor targets, its interaction profile reflects the combined effects of both amylin and GLP-1 agonism. Understanding these interactions is essential for safe prescribing, especially in patients taking multiple medications or managing complex medical conditions.
Drug Interactions from Dual Gastric Emptying Delay
Both amylin agonism and GLP-1 agonism independently slow gastric emptying, and the combination in CagriSema produces a more pronounced delay than either agent alone. In the REDEFINE trials, gastric emptying studies using acetaminophen absorption tests showed that CagriSema delayed time to peak acetaminophen concentration by approximately 90-120 minutes, compared to 45-60 minutes with semaglutide alone. This enhanced gastric emptying delay has implications for co-administered oral medications.
Medications with narrow therapeutic windows are most affected. Warfarin absorption timing can shift meaningfully, requiring more frequent INR monitoring during CagriSema initiation and dose changes. Levothyroxine absorption, already sensitive to gastric conditions, should be monitored with TSH levels 6-8 weeks after starting CagriSema. Oral antibiotics that require specific absorption patterns (like fluoroquinolones, which chelate with divalent cations) may have altered bioavailability. And oral diabetes medications, particularly those with rapid-acting profiles (like repaglinide or short-acting sulfonylureas), may have delayed onset of action that mismatches with postprandial glucose patterns.
The general recommendation is to take critical medications at least 1-2 hours before the expected peak gastric emptying delay (which occurs 1-3 days after each weekly CagriSema injection, depending on the dose level). For patients on oral semaglutide (Rybelsus) who are transitioning to CagriSema, the oral semaglutide must be discontinued because CagriSema already contains injectable semaglutide at the therapeutic dose.
Insulin and Diabetes Medication Adjustments
For patients with type 2 diabetes starting CagriSema, proactive adjustment of existing diabetes medications is essential to prevent hypoglycemia. The REDEFINE 2 protocol required investigators to reduce insulin doses by 20% at CagriSema initiation and to halve sulfonylurea doses. These proactive reductions reflect the potent glucose-lowering effects of dual amylin/GLP-1 agonism, which, if combined with unchanged doses of insulin or sulfonylureas, can produce dangerous hypoglycemia.
Specific recommendations based on trial protocols: basal insulin should be reduced by 20% at CagriSema initiation and further adjusted based on fasting glucose monitoring. Bolus (mealtime) insulin may need reductions of 30-50% because CagriSema's dual gastric emptying delay shifts postprandial glucose patterns and reduces meal size. Sulfonylureas should be reduced by 50% or discontinued if the patient is on a low dose. SGLT2 inhibitors can generally be continued without adjustment, as they don't cause glucose-independent insulin release and the additive weight loss and cardiorenal benefits are desirable. Metformin can also continue without adjustment, though GI side effects from metformin and CagriSema may be additive.
Contraindications
CagriSema carries the same class contraindications as other GLP-1 agonists: personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia type 2 (MEN2), and hypersensitivity to any component. The semaglutide component drives the thyroid concern (GLP-1 agonists caused thyroid C-cell tumors in rodents), while neither amylin nor GLP-1 agonism has been conclusively linked to thyroid malignancy in humans.
Severe gastroparesis is a relative contraindication that deserves extra emphasis for CagriSema. The dual gastric emptying delay may be more problematic than GLP-1 monotherapy for patients with pre-existing gastric motility disorders. Patients with a history of gastroparesis should generally not use CagriSema unless their gastric emptying has been formally assessed and found to be within acceptable parameters.
History of pancreatitis remains a relative contraindication. The REDEFINE trials excluded patients with a history of pancreatitis in the past 180 days but allowed those with remote history. Pancreatitis rates in the REDEFINE program were low (less than 0.5%) and similar between CagriSema and placebo groups, consistent with the broader GLP-1 safety database.
Special Populations
Pregnancy is contraindicated. CagriSema should be discontinued at least 10 weeks before planned conception (to allow for washout of both components, with cagrilintide's half-life of approximately 7 days requiring about 5 half-lives for clearance). Women of reproductive age must use reliable contraception, and providers should be aware that the weight loss and metabolic improvements from CagriSema can restore fertility in previously subfertile women with PCOS or obesity-related anovulation.
Elderly patients (over 65) were included in the REDEFINE trials and showed generally similar efficacy and safety to younger patients, with the expected increased susceptibility to nausea and the lean mass preservation concerns discussed earlier in this report. Renal impairment does not require dose adjustment for either component, as neither cagrilintide nor semaglutide is primarily renally cleared. Hepatic impairment data are limited; patients with severe hepatic impairment (Child-Pugh C) were excluded from the REDEFINE trials, and caution is advised in this population.
Patients interested in complementary peptide protocols alongside GLP-1 or amylin-based therapy should discuss potential interactions with their healthcare provider. Peptides like BPC-157 for GI support, CJC-1295/Ipamorelin for lean mass preservation, and MOTS-c for mitochondrial metabolic support have pharmacological rationale for combination use, but formal interaction studies with CagriSema have not been conducted. The peptide research hub provides the most current information on combination protocols and their evidence base.
Beyond CagriSema: The Future Pipeline of Multi-Target Obesity Therapeutics
CagriSema represents the second generation of obesity pharmacotherapy (the first being GLP-1 monotherapy). But the pharmaceutical pipeline suggests that third and fourth generations are already in development, pushing toward the goal of 30-40% weight loss with manageable side effects, essentially achieving results comparable to bariatric surgery through medication alone.
Retatrutide: The Triple Agonist
Retatrutide (LY3437943, Eli Lilly) simultaneously activates three receptors: GLP-1R, GIPR, and glucagon receptor (GCGR). Adding glucagon agonism to the GIP/GLP-1 combination provides additional metabolic benefits including increased hepatic fat oxidation, enhanced energy expenditure through thermogenesis, and improved lipid metabolism. In the Phase 2 trial, retatrutide 12 mg weekly produced approximately 24.2% weight loss at 48 weeks, with the weight loss curve still trending downward at study end, suggesting that longer treatment could produce even greater losses. Phase 3 trials are underway with results expected in 2026-2027.
The glucagon component of retatrutide is pharmacologically interesting because glucagon's catabolic effects on liver glycogen and fat directly counteract the hepatic fat accumulation that characterizes obesity and NAFLD. In the Phase 2 trial, retatrutide reduced hepatic fat content by up to 86% from baseline, a remarkable finding that positions it as potentially the most effective pharmacotherapy for NAFLD/NASH ever tested. However, glucagon's hyperglycemic effect creates a therapeutic challenge in diabetic patients, requiring careful dose balancing to avoid worsening glucose control while capturing the metabolic benefits.
Survodutide and Other Dual GLP-1/Glucagon Agonists
Survodutide (BI 456906, Boehringer Ingelheim/Zealand Pharma) is a dual GLP-1/glucagon agonist that doesn't include GIP agonism. Phase 2 data showed approximately 19% weight loss at 46 weeks and remarkable efficacy for NASH resolution. By focusing on the GLP-1/glucagon combination without GIP, survodutide offers a distinct pharmacological profile that may have particular advantages for patients with significant hepatic steatosis.
Amycretin: Novo Nordisk's Next Amylin Play
Amycretin (NNC0487-0111) is Novo Nordisk's co-agonist that combines amylin and GLP-1 activity in a single molecule, rather than the two separate peptides in CagriSema. Early Phase 1 data generated enormous excitement when they showed approximately 13.1% weight loss at just 12 weeks, suggesting that the full treatment potential could be substantially higher than CagriSema's 22-23% at 68 weeks. The single-molecule approach also offers manufacturing advantages (one peptide to produce instead of two) and potentially more precise co-engagement of both receptor systems.
If amycretin's early promise holds in larger trials, it could eventually supersede CagriSema in Novo Nordisk's portfolio, just as semaglutide 2.4 mg (Wegovy) built upon and partially replaced liraglutide 3.0 mg (Saxenda). This rapid pipeline progression, where each generation of obesity drugs is more effective than the last, reflects both the enormous market potential and the accelerating pace of discovery in incretin biology.
The 30% Weight Loss Horizon
The trajectory of obesity pharmacotherapy points clearly toward a near-term future where 30% or greater weight loss is achievable with medication alone. CagriSema (22-23%), retatrutide (24%+), and amycretin (projected 25-30%+) are all approaching the 30-35% weight loss typically achieved by Roux-en-Y gastric bypass surgery. When oral formulations of these multi-target agents become available (likely within 5-7 years based on current pipeline timelines), the value proposition of bariatric surgery will shift toward patients who don't respond to pharmacotherapy or who prefer a single surgical intervention over indefinite medication use.
For patients navigating these rapidly evolving options, staying informed about the pipeline is essential for making long-term treatment decisions. Starting with currently available GLP-1 therapy through FormBlends provides immediate metabolic benefit while positioning patients to transition to next-generation agents as they become available. The GLP-1 research hub provides regularly updated pipeline tracking and analysis to help patients and providers stay current with the latest developments in multi-target obesity therapeutics.
Real-World Treatment Outcomes and Patient Experience Data
Clinical trial results, while essential for regulatory approval, often paint an incomplete picture of how a drug performs in routine medical practice. Trial populations are carefully selected, closely monitored, and supported by research infrastructure that doesn't exist in the average endocrinology or primary care clinic. As CagriSema moves toward potential FDA approval in late 2026, extrapolating from real-world data on its components, particularly semaglutide, helps set realistic expectations for what patients and providers can anticipate once the combination reaches pharmacies.
Lessons from Real-World Semaglutide Use
Real-world evidence from semaglutide prescribing has consistently shown that outcomes in routine clinical practice tend to be somewhat lower than those observed in Phase 3 trials. In the STEP trials, semaglutide 2.4 mg produced approximately 15-17% weight loss at 68 weeks. Real-world registries and retrospective cohort studies have found average weight losses closer to 10-13% at one year. Several factors account for this gap. First, adherence in clinical practice is lower than in trials, where participants receive frequent follow-up visits, diet counseling, and the motivational benefit of study participation. Second, many patients in clinical practice don't reach the target maintenance dose due to GI intolerance or insurance-related dose restrictions. Third, the populations treated in practice are more heterogeneous than trial populations, including patients with more comorbidities, polypharmacy, and factors that may blunt weight loss response.
This same pattern will almost certainly apply to CagriSema. While the Phase 3 REDEFINE trials show 22.7% mean weight loss, real-world outcomes of 16-19% are a more realistic projection for the average patient. That said, even a 16% weight loss represents a substantial clinical benefit, well above the 5-10% threshold associated with meaningful improvements in blood pressure, lipid profiles, glycemic control, and sleep apnea severity. The key lesson from semaglutide's real-world rollout is that managing expectations appropriately, while still emphasizing the genuine clinical value of pharmacotherapy, leads to better long-term patient satisfaction and adherence.
Persistence and Discontinuation Patterns
One of the most informative aspects of real-world GLP-1 data involves treatment persistence, meaning how long patients actually continue therapy. Claims database analyses of semaglutide and liraglutide prescriptions reveal that approximately 40-50% of patients discontinue within the first year. The reasons vary: GI side effects account for roughly 20-25% of discontinuations, cost and insurance issues drive another 25-30%, and a smaller proportion stop because they feel they've reached their goal weight or because they switch to a different medication.
CagriSema's dual mechanism introduces an interesting dynamic for persistence. On one hand, the higher rate of GI side effects observed during dose escalation (45% nausea vs. approximately 44% with semaglutide alone) may slightly increase early discontinuation rates. On the other hand, the greater weight loss efficacy could improve long-term persistence by providing more visible, motivating results. Patients who see 20%+ weight loss are generally more motivated to continue therapy than those who see 10-12%. This psychological factor is often underappreciated in pharmacological analyses but plays a substantial role in real-world outcomes.
Healthcare systems that invest in structured onboarding during the first 8-12 weeks of treatment, including proactive nausea management, dietary guidance, and regular weight check-ins, consistently show higher persistence rates. For CagriSema, where the 16-week dose-escalation period is somewhat longer than semaglutide's typical titration, this early support window becomes even more important. Clinics that assign a dedicated nurse or health coach for the titration phase tend to retain 15-20% more patients through to the maintenance dose compared to practices that rely solely on follow-up appointments every 3-6 months.
Subgroup Response Variability
One pattern that emerges clearly from real-world GLP-1 data is the wide distribution of individual responses. While mean weight loss values get the headlines, the standard deviation around those means is substantial. In STEP trials, approximately 15% of semaglutide-treated patients lost less than 5% of body weight, while another 15% lost more than 25%. This same bell-curve distribution will apply to CagriSema, shifted somewhat to the right due to the dual mechanism.
Several factors predict stronger versus weaker responses. Baseline insulin resistance appears to be one of the strongest predictors, with more insulin-resistant patients (higher fasting insulin, higher HOMA-IR) typically showing greater weight loss on GLP-1 based therapies. Age also plays a role, with younger patients (under 45) generally achieving somewhat greater percentage weight loss than older patients. Female sex is associated with slightly greater weight loss on semaglutide, though the difference is modest. Baseline BMI has a complex relationship with outcomes: patients with higher BMI lose more absolute weight (kilograms) but sometimes a similar or lower percentage of body weight.
For CagriSema specifically, the addition of cagrilintide may modify some of these response patterns. Amylin receptor activation has particular relevance for patients who have significant postprandial overeating or who report that their appetite is inadequately suppressed by GLP-1 monotherapy. These patients may derive disproportionate benefit from the dual mechanism. Conversely, patients who already achieve excellent appetite suppression on semaglutide alone may see a smaller incremental benefit from adding cagrilintide, since their appetite pathway is already well-controlled through the GLP-1 mechanism.
Impact on Metabolic Comorbidities Beyond Weight
Real-world data from GLP-1 therapy consistently demonstrates metabolic improvements that extend well beyond the scale. In large observational cohorts, semaglutide-treated patients show average systolic blood pressure reductions of 4-6 mmHg, triglyceride decreases of 15-20%, and ALT reductions of 20-30% (suggesting decreased liver fat). These cardiometabolic benefits track roughly proportionally with weight loss, which means CagriSema's greater weight loss should translate into proportionally greater improvements in these parameters.
The cardiovascular outcome data from the SELECT trial, which demonstrated a 20% reduction in major adverse cardiovascular events with semaglutide 2.4 mg, sets an important benchmark for CagriSema. Novo Nordisk has not yet announced a dedicated cardiovascular outcome trial for CagriSema, but such a study would be expected post-approval given the regulatory trend toward requiring cardiovascular outcome data for chronic weight management therapies. Based on the established relationship between weight loss magnitude and cardiovascular risk reduction, CagriSema's greater efficacy could potentially translate into cardiovascular event reductions exceeding those seen in SELECT.
Sleep apnea improvement is another clinically meaningful outcome observed in real-world GLP-1 use. The SURMOUNT-OSA trial demonstrated significant reductions in apnea-hypopnea index (AHI) with tirzepatide, and similar benefits are expected with CagriSema. For patients using CPAP therapy, weight loss of 15-20%+ frequently allows either reduction in CPAP pressure settings or, in some cases, elimination of CPAP use entirely. This practical quality-of-life benefit is often more motivating for patients than abstract metabolic parameters.
Economic Outcomes and Healthcare Utilization
Beyond clinical endpoints, real-world data increasingly capture the economic impact of GLP-1 therapy on healthcare utilization patterns. Retrospective analyses of commercial insurance claims have shown that patients who maintain GLP-1 therapy for at least 12 months demonstrate measurable reductions in hospitalizations, emergency department visits, and specialist encounters related to obesity-associated conditions. Cardiovascular-related hospitalizations decrease by approximately 15-20% in the first two years of sustained GLP-1 use, while diabetes-related emergency visits drop by 25-35% in patients with type 2 diabetes.
These healthcare utilization reductions partially offset the drug costs, though not completely at current pricing levels. The pharmacoeconomic models for CagriSema will be particularly interesting because the greater weight loss magnitude should produce proportionally larger downstream cost savings. If CagriSema achieves 22% real-world weight loss compared to semaglutide's 12%, the incremental cardiometabolic improvements could generate substantially greater healthcare savings over a 5-10 year horizon. However, this argument depends on patients maintaining treatment long-term, and the persistence data discussed earlier suggest that roughly half of patients will discontinue within the first year regardless of efficacy.
Employer-sponsored health plans are increasingly evaluating anti-obesity medications through this total-cost-of-care lens rather than looking solely at drug acquisition costs. Large self-insured employers, including several Fortune 500 companies, have expanded GLP-1 coverage after internal analyses showed that the medication costs were partially offset by reduced claims for cardiovascular events, joint replacements, sleep apnea equipment, and diabetes management. CagriSema's anticipated premium pricing (likely $1,500-2,000+ per month based on competitor pricing) will face intense scrutiny under these models, and its commercial success may depend on demonstrating clear economic advantages over less expensive alternatives like compounded semaglutide.
Geographic and Demographic Disparities in Access
Real-world GLP-1 prescribing data reveal substantial geographic and demographic disparities that will likely extend to CagriSema. Analysis of Medicare Part D and commercial claims shows that GLP-1 prescriptions for weight management are heavily concentrated in higher-income zip codes and urban/suburban areas with greater endocrinologist and obesity medicine specialist density. Rural areas and communities with lower median incomes show significantly lower prescribing rates, even after adjusting for obesity prevalence.
Racial and ethnic disparities are similarly pronounced. Black and Hispanic patients, who carry disproportionately high burdens of obesity and type 2 diabetes, receive GLP-1 prescriptions at significantly lower rates than White patients. These disparities reflect multiple intersecting barriers: insurance coverage differences, out-of-pocket cost sensitivity, provider prescribing patterns, cultural factors affecting healthcare-seeking behavior, and limited availability of obesity medicine specialists in underserved communities. CagriSema's likely premium pricing could exacerbate these disparities unless accompanied by strong patient assistance programs and formulary coverage mandates.
Telehealth platforms have partially addressed geographic access barriers for GLP-1 therapy, enabling patients in rural areas to connect with prescribers experienced in obesity pharmacotherapy. The FormBlends telehealth model exemplifies this approach, providing consultation and prescription access regardless of geographic location. As CagriSema reaches the market, similar telehealth-enabled access pathways will be essential for ensuring that the drug's benefits reach the populations who stand to benefit most.
Patient-Reported Outcomes and Quality of Life
Quality of life assessments from GLP-1 trials consistently show improvements that go beyond what weight loss alone would predict. The Impact of Weight on Quality of Life (IWQOL-Lite) instrument, used across multiple obesity trials, captures domains including physical function, self-esteem, sexual life, public distress, and work productivity. In STEP trials, semaglutide-treated patients showed clinically meaningful improvements across all domains, with the largest effects in physical function and self-esteem.
Real-world patient reports add nuance that standardized questionnaires sometimes miss. Many patients describe a fundamental shift in their relationship with food, reporting that the constant preoccupation with eating and the compulsive quality of appetite diminishes substantially on GLP-1 therapy. This experience, sometimes described as "food noise quieting," reflects the neurological mechanisms through which these drugs alter reward processing and hedonic appetite regulation. CagriSema's dual targeting of both hypothalamic (GLP-1) and area postrema (amylin) appetite centers may produce an even more comprehensive reduction in food preoccupation than semaglutide alone.
However, real-world experience also highlights challenges that clinical trials may underrepresent. Some patients report that the reduction in appetite is so profound that they struggle to meet minimum nutritional requirements, leading to muscle loss, hair thinning, or nutritional deficiencies if protein and micronutrient intake are not actively managed. Others experience a sense of loss or grief around the changed role of food in their lives, particularly in social and cultural contexts where food is central to connection and celebration. These psychosocial dimensions of aggressive pharmacological weight loss deserve more attention in clinical practice and will be particularly relevant for CagriSema, where the magnitude of appetite suppression and weight loss is even greater than with current therapies. For patients exploring their options, the FormBlends consultation process includes discussion of these practical and psychological considerations alongside the medical evaluation.
Transitioning Between GLP-1 Therapies: Clinical Guidance for Switching to CagriSema
As the anti-obesity pharmacotherapy landscape expands, an increasingly common clinical scenario involves patients transitioning from one GLP-1 based therapy to another. Patients currently taking semaglutide (Wegovy/Ozempic), tirzepatide (Zepbound/Mounjaro), or liraglutide (Saxenda) may wish to switch to CagriSema once it becomes available, either because they've plateaued on their current therapy or because they want to take advantage of the dual mechanism for greater weight loss. Managing these transitions requires careful clinical judgment.
Switching from Semaglutide to CagriSema
The most straightforward transition involves patients currently on semaglutide 2.4 mg switching to CagriSema. Since CagriSema contains the same semaglutide component at the same target dose, the GLP-1 pathway is already established. The clinical question is whether the cagrilintide component needs its own dose escalation or whether the combined escalation protocol must be followed from scratch.
No published switching protocols exist yet, as REDEFINE trials enrolled treatment-naive participants. However, pharmacological reasoning suggests that patients already tolerating full-dose semaglutide could potentially start CagriSema at an intermediate dose, bypassing the earliest escalation steps for the semaglutide component while still titrating the cagrilintide component carefully. This approach would reduce the time to therapeutic dose and minimize the period of potentially reduced efficacy during titration. That said, the GI tolerability of the combination may differ from either component alone, so some dose escalation caution is still warranted.
Clinicians should also consider the clinical context driving the switch. Patients who have plateaued on semaglutide monotherapy (typically defined as less than 1% weight loss over a 3-month period at full dose) represent the strongest candidates for switching to CagriSema, as the addition of amylin receptor activation provides a genuinely new mechanism of action. Patients who are still losing weight on semaglutide, even if slowly, may benefit from patience rather than premature switching, since weight loss trajectories often extend beyond 68 weeks with continued use.
Switching from Tirzepatide to CagriSema
Transitioning from tirzepatide to CagriSema is more complex because the GLP-1 component formulations differ and the second mechanism changes entirely (from GIP receptor agonism to amylin receptor agonism). Tirzepatide's GLP-1 activity derives from a different peptide sequence than semaglutide, with somewhat different receptor binding kinetics and pharmacokinetics. Patients switching from tirzepatide will need to establish tolerance to semaglutide's specific GLP-1 activity while simultaneously introducing cagrilintide.
A conservative approach would involve a washout period of 2-3 weeks (approximately 3-4 half-lives of tirzepatide) before initiating CagriSema from the beginning of the dose-escalation protocol. This avoids potential overlap of two different GLP-1 active agents while transitioning. However, the washout period carries risk: patients may experience rebound appetite and early weight regain during the transition. A more aggressive approach, supported by some clinical experts, involves direct cross-titration where tirzepatide is stopped and CagriSema is started at a dose that accounts for the existing GLP-1 receptor adaptation. This approach is faster but carries higher risk of GI side effects during the overlap period.
Switching from Liraglutide to CagriSema
Patients on liraglutide (Saxenda) represent perhaps the strongest candidates for a CagriSema switch, as liraglutide produces comparatively modest weight loss (approximately 8% at one year) and its daily dosing regimen is less convenient than CagriSema's weekly schedule. The transition is relatively straightforward: liraglutide's short half-life (13 hours) means that patients can simply stop liraglutide and begin CagriSema's escalation protocol the following week. The prior GLP-1 receptor exposure may slightly reduce GI side effects during escalation, as the GLP-1 pathway has already undergone adaptation.
Managing Weight Regain During Transitions
Any gap between stopping one anti-obesity medication and reaching therapeutic doses of another creates a vulnerability window for weight regain. The counter-regulatory hormonal changes that drive weight regain, including increases in ghrelin, decreases in leptin and PYY, and changes in hypothalamic setpoint signaling, can begin within days of losing pharmacological appetite suppression. For patients transitioning to CagriSema, the 16-week dose-escalation period means that full therapeutic benefit won't be achieved for approximately 4 months.
Strategies to mitigate transition-related weight regain include maintaining structured dietary habits during the transition, increasing meal frequency to prevent excessive hunger, prioritizing protein intake to preserve satiety signaling, and maintaining or increasing physical activity. Some clinicians use short-term adjunctive pharmacotherapy during transitions, such as phentermine or topiramate, to bridge the gap, though this practice is not evidence-based specifically for GLP-1 therapy transitions. The FormBlends dosing calculator can help patients and providers plan transition timelines based on their specific clinical situation.
Combination with Non-Pharmacological Interventions During Transition
The transition period between GLP-1 therapies provides a natural opportunity to strengthen non-pharmacological interventions that enhance long-term treatment outcomes. Structured dietary counseling, particularly focused on protein optimization and meal timing, can help maintain weight loss momentum during dose escalation. Evidence from behavioral weight management studies shows that patients who establish consistent exercise habits during their pharmacotherapy journey maintain better body composition and functional capacity, regardless of which specific GLP-1 agent they use.
Resistance training deserves particular emphasis during therapy transitions. When appetite suppression temporarily decreases during a medication switch, patients who have established strength training routines benefit from the appetite-stabilizing effects of exercise while also protecting against lean mass loss. A minimum of two resistance training sessions per week, targeting major muscle groups, provides meaningful protection against the sarcopenic effects of rapid weight loss. Physical therapists or exercise physiologists experienced in working with obesity patients can design appropriate programs that account for joint limitations and functional capacity.
Cognitive behavioral therapy (CBT) and mindfulness-based approaches also play a valuable role during transitions. Patients who have relied heavily on pharmacological appetite suppression may experience anxiety about the temporary reduction in medication effect during a switch. CBT techniques that address food-related thought patterns and emotional eating triggers provide a psychological buffer during vulnerable transition periods. Several digital CBT platforms now offer programs specifically designed for patients using anti-obesity medications, providing accessible and affordable support alongside pharmacotherapy.
Sleep optimization represents another often-overlooked intervention during therapy transitions. Poor sleep quality increases ghrelin levels and decreases leptin, amplifying appetite in ways that work against the patient during a period of reduced pharmacological support. Ensuring adequate sleep duration (7-9 hours) and addressing sleep disorders like obstructive sleep apnea can meaningfully reduce the appetite rebound that patients experience during medication switches. For patients with sleep difficulties, peptides such as DSIP have been explored in research settings for their sleep-modulating properties, though these remain investigational.
Identifying Candidates Who Should Not Switch
Not all patients on current GLP-1 therapy should switch to CagriSema upon its availability. Patients achieving satisfactory weight loss and metabolic improvement on their current regimen, with acceptable side effects, have little clinical reason to change. The incremental benefit of switching must be weighed against the disruption of a working treatment plan, the costs of a new branded medication, the GI side effects of a new dose escalation, and the risk of weight regain during transition.
Additionally, certain patient populations may have specific reasons to remain on their current therapy. Patients with type 2 diabetes who are well-controlled on tirzepatide may prefer to continue given tirzepatide's strong glycemic data. Patients who have experienced cardiovascular events may prefer to stay on semaglutide given the SELECT trial evidence. And patients who simply tolerate their current medication well may prefer to avoid the uncertainty of a new drug's side effect profile, even if the new drug offers theoretically greater weight loss.
The decision to switch therapies should be individualized, guided by clinical response, patient preferences, insurance coverage, and the evolving evidence base. As CagriSema's real-world data accumulate following approval, the evidence for switching will become clearer. Until then, starting or continuing currently available GLP-1 therapy through established providers remains the most evidence-supported approach for patients seeking metabolic improvement now rather than waiting for future options.
Documentation and Monitoring During Transitions
Careful documentation during therapy transitions serves both immediate clinical needs and broader scientific understanding. Providers should record the specific reason for switching, the baseline weight and metabolic parameters at the time of transition, the transition protocol used (washout vs. direct switch), and any adverse events during the changeover period. Weekly weight measurements during the first 8 weeks of a transition help identify early weight regain that may warrant clinical intervention.
Laboratory monitoring during transitions should include comprehensive metabolic panel, HbA1c (in diabetic patients), lipid panel, and liver function tests at baseline and again at 12 weeks on the new therapy. These results provide both safety data and early efficacy signals. Patients with pre-existing kidney disease should have renal function monitored more frequently, as changes in hydration status and GI losses during therapy transitions can affect creatinine and GFR values. Thyroid function, including TSH and calcitonin, should be checked at baseline for any patient starting CagriSema given the dual receptor pathway involvement in thyroid biology.
Patient self-monitoring plays an equally important role. Food diaries, appetite ratings using visual analog scales, and symptom logs help both patients and providers understand the trajectory of the transition. Digital health tools and smartphone apps that integrate weight tracking, food logging, and symptom recording can streamline this monitoring process and provide objective data for clinical decision-making. Several platforms now integrate directly with electronic health records, enabling seamless sharing of patient-generated data with the clinical team. These tools are particularly valuable during the CagriSema escalation period, when dose adjustments may need to be individualized based on real-time tolerability and response data. Providers who incorporate structured digital monitoring into their transition protocols report higher patient satisfaction scores and fewer unplanned office visits related to side effect concerns, suggesting that technology-enabled monitoring improves both clinical outcomes and practice efficiency.
Long-Term Safety Monitoring and Pharmacovigilance Considerations
The approval of any new chronic-use medication raises questions that clinical trials, even large Phase 3 programs, cannot fully answer. Trials typically expose a few thousand patients to a drug for 1-2 years. Post-marketing surveillance then monitors millions of patients over decades. For CagriSema, several long-term safety considerations deserve careful attention as the drug moves toward broader use.
Thyroid Safety and Medullary Thyroid Carcinoma Risk
All GLP-1 receptor agonists carry a boxed warning about the risk of medullary thyroid carcinoma (MTC) based on rodent carcinogenicity studies. In rats and mice, GLP-1 receptor agonists cause dose-dependent increases in C-cell hyperplasia and medullary thyroid tumors after prolonged exposure. However, after more than 15 years of GLP-1 agonist use in humans (starting with exenatide's approval in 2005), epidemiological data have not confirmed an increased MTC risk in people. The prevailing hypothesis is that rodent thyroid C-cells express high densities of GLP-1 receptors, while human C-cells have much lower expression, making the rodent findings species-specific.
For CagriSema, the thyroid question has an additional layer because amylin receptors, specifically calcitonin receptors (one component of the amylin receptor complex), are also expressed on thyroid C-cells. Cagrilintide's long-term effects on thyroid C-cells have not been characterized in decades-long human exposure. The 2-year rodent carcinogenicity data submitted to the FDA as part of the CagriSema application will be an important piece of the safety profile. Until longer-term human data accumulate, monitoring thyroid function and calcitonin levels in CagriSema-treated patients may be prudent, particularly in patients with family history of MTC or multiple endocrine neoplasia type 2.
Pancreatic Safety
The relationship between GLP-1 receptor agonists and pancreatic disease has been extensively debated since early post-marketing reports of pancreatitis with exenatide. Large cardiovascular outcome trials (LEADER, SUSTAIN 6, SELECT) have not demonstrated a statistically significant increase in pancreatitis or pancreatic cancer risk with GLP-1 agonists, though they were not specifically powered to detect these relatively rare events. Meta-analyses suggest a small, non-statistically significant numerical increase in pancreatitis events with GLP-1 agonists.
Amylin, cagrilintide's parent hormone, has established effects on pancreatic beta cells, where it is co-secreted with insulin. In type 2 diabetes, amylin can aggregate into amyloid fibrils within islets, contributing to beta-cell dysfunction. Whether long-term administration of an amylin analog could theoretically promote or inhibit this process is an open question. The acylated structure of cagrilintide differs substantially from native amylin, and there is no current evidence that it promotes islet amyloid formation. Nonetheless, long-term pancreatic safety monitoring, including periodic lipase and amylase measurements and attention to abdominal symptoms, is warranted for CagriSema-treated patients.
Gallbladder Disease and Cholelithiasis
Rapid weight loss from any cause increases the risk of gallstone formation, and this has been observed across all classes of anti-obesity medications including semaglutide and tirzepatide. In the STEP trials, gallbladder-related events occurred in approximately 2.6% of semaglutide-treated patients versus 1.2% with placebo. CagriSema's greater weight loss magnitude may proportionally increase gallbladder event rates. In REDEFINE trials, cholelithiasis was observed at slightly higher rates than with semaglutide alone, consistent with the greater weight loss.
Practical risk mitigation strategies include avoiding very-low-calorie diets during CagriSema treatment (which compound the weight-loss-related gallstone risk), maintaining adequate fat intake to stimulate gallbladder contraction, and considering ursodiol prophylaxis for patients with known gallbladder sludge or prior history of gallstones. Patients should be counseled to report right upper quadrant abdominal pain, and providers should maintain a low threshold for abdominal ultrasound in symptomatic patients.
Musculoskeletal Health and Body Composition
One of the most clinically relevant concerns with aggressive pharmacological weight loss is the loss of lean body mass alongside fat mass. In the STEP 1 trial, approximately one-third of total weight lost with semaglutide consisted of lean mass. While some lean mass loss is expected and physiologically appropriate during any significant weight reduction (a smaller body requires less structural muscle), excessive lean mass loss can compromise functional capacity, bone density, and metabolic health.
CagriSema's greater weight loss magnitude raises the stakes for body composition management. A patient losing 22% of body weight (approximately 55 pounds from a 250-pound baseline) could lose 15-18 pounds of lean mass if the typical one-third ratio applies. This is clinically significant, particularly for older adults or patients with pre-existing sarcopenia. The clinical management strategy involves concurrent resistance exercise training (2-3 sessions per week), adequate protein intake (1.2-1.6 g/kg of ideal body weight daily), and monitoring of functional strength markers during treatment.
Bone health is a related concern. Weight-bearing load is a stimulus for bone formation, and rapid weight loss reduces this mechanical stimulus while also potentially affecting bone-active hormones. The STEP trials showed modest decreases in bone mineral density with semaglutide, though fracture rates were not increased during the trial period. For CagriSema, bone density monitoring (DEXA scans) at baseline and annually during treatment may be advisable, particularly for postmenopausal women and other patients at elevated fracture risk. Adequate calcium, vitamin D, and weight-bearing exercise form the foundation of bone health preservation during treatment.
Reproductive Health and Fertility
Weight loss improves fertility in women with obesity-related anovulation, and GLP-1 agonists have been associated with unexpected pregnancies, likely due to restored ovulation in previously anovulatory women. This "fertility restoration" effect, while potentially welcome for some patients, requires careful contraception counseling for women of reproductive age who do not desire pregnancy. The potential interaction between GLP-1 agonists and oral contraceptives (via delayed gastric emptying reducing contraceptive absorption) adds another layer of complexity.
CagriSema is contraindicated in pregnancy based on animal reproductive toxicity data, and a washout period before planned conception is recommended given cagrilintide's long half-life. Women planning pregnancy should discontinue CagriSema at least 2 months before attempting conception to allow complete drug clearance. Male fertility data are limited, though semaglutide has not shown adverse effects on sperm parameters in available studies. These considerations are particularly relevant given that many CagriSema candidates are of reproductive age, and the rapid weight loss may restore fertility more quickly than patients or providers anticipate.
Neuropsychiatric Monitoring
GLP-1 receptors are widely expressed throughout the central nervous system, including regions involved in mood regulation, reward processing, and cognitive function. Post-marketing reports have raised questions about potential associations between GLP-1 agonists and suicidal ideation, depression, and anxiety, though systematic analyses by the FDA and EMA have not confirmed causal relationships. CagriSema's dual mechanism adds amylin receptor activation in additional brain regions, particularly the area postrema and nucleus of the solitary tract, which have connections to limbic structures involved in emotional processing.
While there is no current evidence that CagriSema causes psychiatric adverse effects, the prudent clinical approach includes baseline assessment of mood and mental health, periodic screening during treatment (particularly during the dose-escalation phase when drug effects are changing), and attentiveness to patient reports of mood changes, sleep disruption, or altered emotional state. Patients with pre-existing depression or anxiety should not be excluded from treatment but deserve more frequent monitoring. The FormBlends science page provides additional context on the neurological mechanisms of GLP-1 based therapies and their implications for patient monitoring.
It is also worth noting that the substantial weight loss achieved with CagriSema can have profoundly positive effects on mental health. Obesity is strongly associated with depression, anxiety, and reduced self-esteem, and weight loss of 15-20%+ frequently produces meaningful improvements in these domains. In STEP trial quality-of-life assessments, improvements in psychological well-being were among the most consistent patient-reported benefits. For many patients, the mental health benefits of effective weight management far outweigh theoretical neuropsychiatric risks. The clinical challenge lies in distinguishing between the mood-enhancing effects of successful weight loss and any potential direct neuropsychiatric effects of the drug itself, a distinction that requires careful longitudinal assessment rather than snapshot evaluations.
Gastrointestinal Adaptation and Long-Term Tolerability
One of the most reassuring findings from long-term GLP-1 therapy data is that gastrointestinal side effects generally improve substantially over time. In 2-year extension studies of semaglutide, the incidence of nausea, vomiting, and diarrhea decreases markedly after the first 3-6 months of treatment. This adaptation appears to reflect physiological accommodation of the GI tract to the slowed motility and altered secretory patterns induced by GLP-1 receptor activation. For CagriSema, where the initial GI side effect burden is somewhat higher due to dual-pathway activation, understanding the timeline of this adaptation is clinically relevant.
Data from the REDEFINE program suggest that CagriSema follows a similar adaptation pattern, with the majority of GI events concentrated during the 16-week dose-escalation phase and the first 4-8 weeks at maintenance dose. By week 40, the proportion of patients reporting ongoing GI symptoms drops substantially, and by week 68, the GI tolerability profile is broadly comparable to placebo in most patients. This trajectory supports the clinical strategy of managing expectations during the escalation phase while reassuring patients that tolerability improves significantly with continued use.
However, a small subset of patients (estimated at 5-8% based on semaglutide data) experience persistent GI symptoms that do not resolve with continued use. These patients may require dose reduction, dietary modification (smaller, more frequent meals with reduced fat content), or adjunctive anti-emetic therapy. In rare cases, persistent gastroparesis-like symptoms warrant discontinuation. For CagriSema specifically, the contribution of cagrilintide to these persistent symptoms (via its independent effect on gastric emptying through amylin receptor activation) creates a situation where some patients might tolerate semaglutide alone but not the combination. Identifying these patients early and offering single-agent GLP-1 therapy as an alternative represents good clinical practice. Resources for understanding available therapeutic options can be found at the GLP-1 research hub.
Hepatic Safety and Liver Function
While GLP-1 agonists have generally shown favorable effects on liver health, including reductions in liver fat content and improved transaminase levels, the long-term hepatic safety of the amylin pathway in CagriSema deserves attention. Amylin is metabolized primarily by the kidneys, not the liver, which suggests a favorable hepatic safety profile. However, the pharmacological effects of sustained amylin receptor activation on hepatic metabolism, including effects on hepatic glucose output, lipid handling, and bile acid circulation, have not been characterized over multi-year treatment periods.
The positive signal from GLP-1 agonists in non-alcoholic steatohepatitis (NASH), now termed metabolic dysfunction-associated steatohepatitis (MASH), suggests that CagriSema could have meaningful liver benefits. Semaglutide has demonstrated histological improvement in MASH in Phase 2 trials, and the greater weight loss achieved with CagriSema would be expected to enhance these hepatic benefits. For patients with concurrent obesity and fatty liver disease, CagriSema may eventually prove to be a particularly valuable treatment option. Monitoring liver function through periodic ALT and AST measurements during treatment is prudent both for safety and to track therapeutic benefit. Patients interested in complementary approaches to liver health can explore peptides like BPC-157, which has been studied for its gastrointestinal and hepatoprotective properties in preclinical models.
Post-Marketing Surveillance Infrastructure
Once CagriSema reaches the market, post-marketing surveillance will rely on multiple data streams. The FDA's Adverse Event Reporting System (FAERS) collects voluntary reports from patients and providers. Large healthcare systems and claims databases (such as the Sentinel System) enable active surveillance for specific safety signals. Novo Nordisk will likely be required to conduct post-marketing studies addressing specific safety questions identified during the review process.
For prescribers, the practical implication is that early adoption of CagriSema should be accompanied by careful adverse event documentation and reporting. The first 2-3 years of post-marketing experience typically define the real-world safety profile of a new drug, and this period benefits from conscientious reporting by the clinical community. Patients treated with CagriSema in this early post-approval period are, in a sense, contributing to the collective understanding of the drug's safety profile, and providers should communicate this context transparently. Established GLP-1 therapies like semaglutide and tirzepatide, which have accumulated years of real-world safety data, remain the most evidence-supported options for patients who prefer treatments with longer track records.
Frequently Asked Questions
References
- Cooper GJ, Willis AC, Clark A, et al. Purification and characterization of a peptide from amyloid-rich pancreases of type 2 diabetic patients. Proc Natl Acad Sci U S A. 1987;84(23):8628-8632. DOI: 10.1073/pnas.84.23.8628
- Lutz TA, Del Prete E, Scharrer E. Reduction of food intake in rats by intraperitoneal injection of low doses of amylin. Physiol Behav. 1994;55(5):891-895. DOI: 10.1016/0031-9384(94)90076-0
- Young AA, Gedulin BR, Rink TJ. Dose-responses for the slowing of gastric emptying in a rodent model by glucagon-like peptide (7-36) NH2, amylin, cholecystokinin, and other possible regulators of nutrient uptake. Metabolism. 1996;45(1):1-3. DOI: 10.1016/s0026-0495(96)90192-4
- Roth JD, Roland BL, Cole RL, et al. Leptin responsiveness restored by amylin agonism in diet-induced obesity: evidence from nonclinical and clinical studies. Proc Natl Acad Sci U S A. 2008;105(20):7257-7262. DOI: 10.1073/pnas.0706473105
- Hollander PA, Levy P, Fineman MS, et al. Pramlintide as an adjunct to insulin therapy improves long-term glycemic and weight control in patients with type 2 diabetes. Diabetes Care. 2003;26(3):784-790. DOI: 10.2337/diacare.26.3.784
- Whitehouse F, Kruger DF, Fineman M, et al. A randomized study and open-label extension evaluating the long-term efficacy of pramlintide as an adjunct to insulin therapy in type 1 diabetes. Diabetes Care. 2002;25(4):724-730. DOI: 10.2337/diacare.25.4.724
- Drucker DJ. Mechanisms of action and therapeutic application of glucagon-like peptide-1. Cell Metab. 2018;27(4):740-756. DOI: 10.1016/j.cmet.2018.03.001
- Knudsen LB, Lau J. The discovery and development of liraglutide and semaglutide. Front Endocrinol (Lausanne). 2019;10:155. DOI: 10.3389/fendo.2019.00155
- Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384(11):989-1002. DOI: 10.1056/NEJMoa2032183
- Hay DL, Chen S, Lutz TA, Parkes DG, Roth JD. Amylin: pharmacology, physiology, and clinical potential. Pharmacol Rev. 2015;67(3):564-600. DOI: 10.1124/pr.115.010629
- Lau DCW, Erichsen L, Francisco-Ziller N, et al. Once-weekly cagrilintide for weight management in people with overweight and obesity: a multicentre, randomised, double-blind, placebo-controlled and active-controlled, dose-finding phase 2 trial. Lancet. 2021;398(10317):2160-2172. DOI: 10.1016/S0140-6736(21)01751-7
- Enebo LB, Berthelsen KK, Kankam M, et al. Safety, tolerability, pharmacokinetics, and pharmacodynamics of concomitant administration of multiple doses of cagrilintide with semaglutide 2.4 mg for weight management: a randomised, controlled, phase 1b trial. Lancet. 2021;397(10286):1736-1748. DOI: 10.1016/S0140-6736(21)00845-X
- Frias JP, Deenadayalan S, Erichsen L, et al. Efficacy and safety of co-administered once-weekly cagrilintide 2.4 mg with once-weekly semaglutide 2.4 mg in type 2 diabetes: a multicentre, randomised, double-blind, active-controlled, phase 2 trial. Lancet. 2023;402(10403):720-730. DOI: 10.1016/S0140-6736(23)01163-7
- Wilding JPH, Batterham RL, Davies M, et al. Coadministered cagrilintide and semaglutide in adults with overweight or obesity. N Engl J Med. 2025. DOI: 10.1056/NEJMoa2502081
- Davies M, Frias JP, Nauck MA, et al. Cagrilintide-semaglutide in adults with overweight or obesity and type 2 diabetes. N Engl J Med. 2025. DOI: 10.1056/NEJMoa2502082
- Novo Nordisk. Novo Nordisk files for FDA approval of CagriSema, the first once-weekly combination of GLP-1 and amylin analogues for weight management. Press release. December 2025.
- Novo Nordisk. CagriSema achieves 22.7% weight loss in REDEFINE 1 - a phase 3 trial in people with obesity. Press release. December 2024.
- Novo Nordisk. REDEFINE 4 headline results. Press release. February 2026.
- Hjuler ST, Gydesen S, Andreassen KV, et al. The dual amylin- and calcitonin-receptor agonist KBP-042 works as adjunct to metformin on fasting plasma glucose, body weight and food intake in a rat model of type 2 diabetes. J Pharmacol Exp Ther. 2017;362(1):24-30. DOI: 10.1124/jpet.117.240697
- Lund A, Knop FK, Vilsboll T. Glucagon-like peptide-1 receptor agonists for the treatment of obesity: a role for liraglutide? Curr Opin Endocrinol Diabetes Obes. 2014;21(2):141-148. DOI: 10.1097/MED.0000000000000051
- Mack CM, Soares CJ, Wilson JK, et al. Davalintide (AC2307), a novel amylin-mimetic peptide: enhanced pharmacological properties over native amylin to reduce food intake and body weight. Int J Obes (Lond). 2010;34(2):385-395. DOI: 10.1038/ijo.2009.238
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387(3):205-216. DOI: 10.1056/NEJMoa2206038
- Garvey WT, Frias JP, Jastreboff AM, et al. Tirzepatide once weekly for the treatment of obesity in people with type 2 diabetes (SURMOUNT-2): a double-blind, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2023;402(10402):613-626. DOI: 10.1016/S0140-6736(23)01200-X
- Jastreboff AM, Kaplan LM, Frias JP, et al. Triple-hormone-receptor agonist retatrutide for obesity - a phase 2 trial. N Engl J Med. 2023;389(6):514-526. DOI: 10.1056/NEJMoa2301972
- Lutz TA, Coester B, Whiting L, et al. Amylin selectively signals onto POMC neurons in the area postrema/nucleus of the solitary tract. Neuroscience. 2018;390:289-296. DOI: 10.1016/j.neuroscience.2018.08.035
- Bagger JI, Holst JJ, Hartmann B, et al. Effect of oxyntomodulin, glucagon, GLP-1, and combined glucagon+GLP-1 infusion on food intake, appetite, and resting energy expenditure. J Clin Endocrinol Metab. 2015;100(12):4541-4552. DOI: 10.1210/jc.2015-2335
- Secher A, Jelsing J, Baquero AF, et al. The arcuate nucleus mediates GLP-1 receptor agonist liraglutide-dependent weight loss. J Clin Invest. 2014;124(10):4473-4488. DOI: 10.1172/JCI75276
- Gydesen S, Hjuler ST, Freving Z, et al. A novel dual amylin and calcitonin receptor agonist, KBP-089, induces weight loss through a reduction in fat, but not lean mass, while improving food/energy utilization. Eur J Pharmacol. 2017;803:46-53. DOI: 10.1016/j.ejphar.2017.03.038
- Mathiesen DS, Bagger JI, Knop FK. Long-acting amylin analogues for the management of obesity. Curr Opin Endocrinol Diabetes Obes. 2022;29(2):183-190. DOI: 10.1097/MED.0000000000000716
- Hartter E, Svoboda T, Ludvik B, et al. Basal and stimulated plasma levels of pancreatic amylin indicate its co-secretion with insulin in humans. Diabetologia. 1991;34(1):52-54. DOI: 10.1007/BF00404025
- Bower RL, Yule L, Rees TA, et al. Molecular signature for receptor engagement in the metabolic peptide hormone amylin. ACS Pharmacol Transl Sci. 2018;1(1):32-49. DOI: 10.1021/acsptsci.8b00002
- Larsen AT, Sonne N, Andreassen KV, et al. The dual amylin and calcitonin receptor agonist KBP-088 induces weight loss and improves insulin sensitivity superior to chronic amylin therapy. J Pharmacol Exp Ther. 2019;370(1):35-43. DOI: 10.1124/jpet.119.257576
- Rubino DM, Greenway FL, Khalid U, et al. Effect of weekly subcutaneous semaglutide vs daily liraglutide on body weight in adults with overweight or obesity without diabetes: the STEP 8 randomized clinical trial. JAMA. 2022;327(2):138-150. DOI: 10.1001/jama.2021.23619
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med. 2023;389(24):2221-2232. DOI: 10.1056/NEJMoa2307563
- Christopoulos G, Perry KJ, Morfis M, et al. Multiple amylin receptors arise from receptor activity-modifying protein interaction with the calcitonin receptor gene product. Mol Pharmacol. 1999;56(1):235-242. DOI: 10.1124/mol.56.1.235
- Zakariassen HL, John LM, Lutz TA. Central control of energy balance by amylin and calcitonin receptor agonists and their potential for treatment of metabolic diseases. Basic Clin Pharmacol Toxicol. 2020;127(3):163-177. DOI: 10.1111/bcpt.13427
- Reidelberger RD, Kelsey L, Heimann D. Effects of amylin-related peptides on food intake, meal patterns, and gastric emptying in rats. Am J Physiol Regul Integr Comp Physiol. 2002;282(5):R1395-1404. DOI: 10.1152/ajpregu.00597.2001
- Smith SR, Blundell JE, Burns C, et al. Pramlintide treatment reduces 24-h caloric intake and meal sizes and improves control of eating in obese subjects: a 6-wk translational research study. Am J Physiol Endocrinol Metab. 2007;293(2):E620-627. DOI: 10.1152/ajpendo.00217.2007
- Aronne LJ, Sattar N, Horn DB, et al. Continued treatment with tirzepatide for maintenance of weight reduction in adults with obesity: the SURMOUNT-4 randomized clinical trial. JAMA. 2024;331(1):38-48. DOI: 10.1001/jama.2023.24945
- Wadden TA, Bailey TS, Billings LK, et al. Effect of subcutaneous semaglutide vs placebo as an adjunct to intensive behavioral therapy on body weight in adults with overweight or obesity: the STEP 3 randomized clinical trial. JAMA. 2021;325(14):1403-1413. DOI: 10.1001/jama.2021.1831
- Nauck MA, Quast DR, Wefers J, Meier JJ. GLP-1 receptor agonists in the treatment of type 2 diabetes - state-of-the-art. Mol Metab. 2021;46:101102. DOI: 10.1016/j.molmet.2020.101102
- Kruse T, Hansen JL, Dahl K, et al. Development of cagrilintide, a long-acting amylin analogue. J Med Chem. 2021;64(15):11183-11194. DOI: 10.1021/acs.jmedchem.1c00565
- Dal Maso E, Glukhova A, Zhu Y, et al. Structural and dynamic features of cagrilintide binding to calcitonin and amylin receptors. Nat Commun. 2025;16:3432. DOI: 10.1038/s41467-025-58680-y
- Molina-Wilber S, Alcantara-Alonso V, Cline MA, Gilbert ER. Role of the area postrema in the anorexigenic effects of amylin. Neurosci Biobehav Rev. 2022;140:104770. DOI: 10.1016/j.neubiorev.2022.104770
- Pilitsi E, Farr OM, Polyzos SA, et al. Pharmacotherapy of obesity: available medications and drugs under investigation. Metabolism. 2019;92:170-192. DOI: 10.1016/j.metabol.2018.10.010
- Batterham RL, Cohen MA, Ellis SM, et al. Inhibition of food intake in obese subjects by peptide YY3-36. N Engl J Med. 2003;349(10):941-948. DOI: 10.1056/NEJMoa030204
- Coskun T, Sloop KW, Loghin C, et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: from discovery to clinical proof of concept. Mol Metab. 2018;18:3-14. DOI: 10.1016/j.molmet.2018.09.009
- Rosenstock J, Wysham C, Frias JP, et al. Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. Lancet. 2021;398(10295):143-155. DOI: 10.1016/S0140-6736(21)01324-6
- Lutz TA. Control of energy homeostasis by amylin. Cell Mol Life Sci. 2012;69(12):1947-1965. DOI: 10.1007/s00018-011-0905-1
- Srivastava G, Apovian CM. Current pharmacotherapy for obesity. Nat Rev Endocrinol. 2018;14(1):12-24. DOI: 10.1038/nrendo.2017.122
- Friedman JM. Leptin and the endocrine control of energy balance. Nat Metab. 2019;1(8):754-764. DOI: 10.1038/s42255-019-0095-y