Executive Summary
Figure 1: HGH Fragment 176-191 represents the isolated fat-metabolizing domain of human growth hormone, stripped of growth-promoting and diabetogenic effects.
Key Takeaways
- Lipolytic potency: In vitro studies show Fragment 176-191 increases fat breakdown rates approximately 4.2-fold compared to untreated controls, slightly exceeding the effects of full-length hGH (3.5-fold increase).
- Weight loss in animal models: Chronic treatment with the C-terminal fragment reduced body weight gain by nearly 50% in obese (ob/ob) mice over a 14-day treatment period, with corresponding increases in fat oxidation and plasma glycerol levels.
- Human clinical data (AOD-9604): In the METAOD005 Phase IIb trial, 300 obese adults treated with 1 mg/day oral AOD-9604 lost an average of 2.8 kg over 12 weeks, compared to 0.8 kg in the placebo group, a statistically significant difference.
- Safety profile: Across six randomized, double-blind, placebo-controlled human trials involving 893 subjects, AOD-9604 showed a safety profile indistinguishable from placebo with no effects on IGF-1 levels, glucose tolerance, or insulin sensitivity.
- Regulatory status: AOD-9604 received FDA GRAS (Generally Recognized As Safe) designation for use as a food ingredient, though it is not approved as a pharmaceutical agent for weight loss.
HGH Fragment 176-191 is a synthetic peptide comprising the last 16 amino acids of human growth hormone (hGH), specifically engineered to retain the fat-burning properties of the parent molecule while eliminating its growth-promoting and glucose-disrupting side effects. This C-terminal fragment has attracted significant research interest as a targeted lipolytic agent that stimulates fat breakdown without triggering IGF-1 release, insulin resistance, or the cell proliferation associated with full-length growth hormone therapy.
The story of Fragment 176-191 begins in the early 1990s, when researchers at Monash University in Melbourne, Australia, set out to identify which region of the 191-amino-acid growth hormone molecule was responsible for its fat-metabolizing activity. Their work, led by Wu and colleagues, demonstrated in 1993 that a synthetic peptide corresponding to amino acids 176 through 191 of hGH possessed antilipogenic activity identical to that of the intact hormone. This discovery opened a new chapter in peptide pharmacology, suggesting that the metabolic benefits of growth hormone could be separated from its undesirable effects.
From this foundational work emerged two closely related compounds: Fragment 176-191 in its original form and AOD-9604 (Anti-Obesity Drug 9604), a modified version with an added N-terminal tyrosine residue that conferred improved stability and oral bioavailability. Both peptides target the same biological pathway, stimulating lipolysis through a mechanism involving beta-3 adrenergic receptor upregulation and subsequent activation of hormone-sensitive lipase (HSL) in adipose tissue. The critical distinction from full-length hGH is that neither compound binds the growth hormone receptor or stimulates IGF-1 production.
Key Findings at a Glance
Critical Evidence Points
- Lipolytic potency: In vitro studies show Fragment 176-191 increases fat breakdown rates approximately 4.2-fold compared to untreated controls, slightly exceeding the effects of full-length hGH (3.5-fold increase).
- Weight loss in animal models: Chronic treatment with the C-terminal fragment reduced body weight gain by nearly 50% in obese (ob/ob) mice over a 14-day treatment period, with corresponding increases in fat oxidation and plasma glycerol levels.
- Human clinical data (AOD-9604): In the METAOD005 Phase IIb trial, 300 obese adults treated with 1 mg/day oral AOD-9604 lost an average of 2.8 kg over 12 weeks, compared to 0.8 kg in the placebo group, a statistically significant difference.
- Safety profile: Across six randomized, double-blind, placebo-controlled human trials involving 893 subjects, AOD-9604 showed a safety profile indistinguishable from placebo with no effects on IGF-1 levels, glucose tolerance, or insulin sensitivity.
- Regulatory status: AOD-9604 received FDA GRAS (Generally Recognized As Safe) designation for use as a food ingredient, though it is not approved as a pharmaceutical agent for weight loss.
The clinical development of this peptide family has followed a complex trajectory. While early animal studies generated substantial enthusiasm, the Phase IIb trial results, though positive at the lowest dose, were not considered commercially viable by Metabolic Pharmaceuticals, the sponsoring company, and further pharmaceutical development was halted. This decision didn't diminish scientific interest in the fragment's mechanism, however. Researchers continue to study how this small peptide selectively activates fat metabolism, and the compound has found a second life in the nutraceutical and research peptide space.
For individuals exploring peptide-based approaches to body composition management, understanding Fragment 176-191 requires grasping several interconnected concepts: how growth hormone normally regulates fat metabolism, why the C-terminal region holds the key to lipolysis, how this fragment differs from AOD-9604 and from growth hormone secretagogues like CJC-1295/Ipamorelin, and what the clinical evidence actually supports. This report examines each of these questions in detail, drawing exclusively on published peer-reviewed research and registered clinical trial data.
Throughout this analysis, we'll also consider how Fragment 176-191 fits within the broader world of metabolic peptides, including comparisons with GLP-1 receptor agonists like semaglutide that have reshaped the obesity treatment field, as well as other growth hormone-related peptides such as tesamorelin and sermorelin. The goal isn't to position Fragment 176-191 as a competitor to these agents but to clearly define its unique mechanism and the evidence supporting its use.
Who This Report Is For
This comprehensive guide serves clinicians evaluating peptide therapy options for patients with metabolic concerns, researchers studying the lipolytic domain of growth hormone, and informed individuals seeking to understand the science behind one of the most discussed fat-metabolizing peptides in the research community. Every claim is supported by specific study data, with full citations provided in the references section. Where evidence is strong, we say so plainly. Where it falls short, we note the gaps with equal directness.
The Peptide Research Hub contains additional guides on related growth hormone peptides and metabolic compounds that complement the information presented here.
The C-Terminal Fragment: Understanding GH Structural Biology
Figure 2: The C-terminal fragment encompasses the last 16 amino acids of the 191-amino-acid human growth hormone molecule, representing the lipolytic active domain.
Human growth hormone is a 191-amino-acid polypeptide secreted by the anterior pituitary gland. While the intact molecule produces a broad spectrum of metabolic effects, including growth promotion, anabolic signaling, glucose counter-regulation, and fat mobilization, decades of research have revealed that these activities map to distinct structural regions of the protein. The C-terminal domain, comprising amino acids 176 through 191, contains the molecular machinery responsible for growth hormone's fat-burning properties.
Architecture of Human Growth Hormone
The three-dimensional structure of hGH consists of a four-helix bundle, a common motif among cytokine-family hormones. This architecture creates distinct functional surfaces that interact with different receptor systems and downstream signaling molecules. The N-terminal and central domains engage the growth hormone receptor (GHR), forming a 1:2 complex that initiates the JAK2-STAT5 signaling cascade responsible for IGF-1 production, cell proliferation, and growth-promoting effects. The C-terminal region, by contrast, operates through a separate mechanism entirely.
When researchers first began systematically truncating growth hormone to identify its functional domains, they discovered something surprising. The C-terminal fragment retained potent lipid-mobilizing activity even when completely disconnected from the receptor-binding regions. This finding, published by Wu et al. in 1993, demonstrated that synthetic peptides corresponding to amino acids 176-191 exhibited antilipogenic activity equivalent to full-length hGH. The fragment didn't need the growth hormone receptor to do its job.
The Discovery: Wu et al. and the Antilipogenic Domain
The landmark 1993 study by Wu and colleagues at Monash University examined multiple synthetic fragments of hGH for their effects on lipid metabolism. Using isolated adipocytes, the researchers tested whether specific peptide sequences could reproduce the lipogenic and lipolytic effects of the intact hormone. Their findings were clear and reproducible: the C-terminal sequence 176-191 inhibited lipogenesis (new fat synthesis) with the same potency as full-length growth hormone.
What made this discovery particularly significant was what the fragment couldn't do. Unlike the complete hGH molecule, Fragment 176-191 did not stimulate longitudinal bone growth, did not increase IGF-1 levels, and did not impair glucose metabolism. The researchers had effectively isolated the "fat-burning" component of growth hormone from its growth-promoting and diabetogenic properties.
Structural Details
The amino acid sequence of Fragment 176-191 is: Tyr-Leu-Arg-Ile-Val-Gln-Cys-Arg-Ser-Val-Glu-Gly-Ser-Cys-Gly-Phe. This 16-residue peptide has a molecular weight of approximately 1,817.12 Daltons. The two cysteine residues at positions 182 and 189 can form an intramolecular disulfide bond, creating a looped structure that appears important for biological activity. The N-terminal tyrosine in the modified version (AOD-9604) enhances stability and may improve receptor interaction.
Structural Determinants of Lipolytic Activity
Several structural features within the 176-191 sequence contribute to its biological function. The two cysteine residues (Cys182 and Cys189) form a disulfide bridge that creates a constrained loop. This loop appears essential for the peptide's ability to interact with its molecular targets in adipose tissue. Structure-activity relationship studies have shown that disrupting this disulfide bond significantly reduces lipolytic potency.
The charged residues within the sequence, particularly the arginine residues at positions 178 and 183, contribute to electrostatic interactions with cell-surface receptors. The hydrophobic residues (leucine, isoleucine, valine, phenylalanine) form an amphipathic face that facilitates membrane interaction. Together, these elements create a compact molecular surface that mimics the fat-mobilizing interface of the full-length hormone without replicating its receptor-binding topology.
Why the C-Terminus Controls Fat Metabolism
The functional separation between growth-promoting and fat-metabolizing activities of hGH reflects the evolutionary history of the growth hormone gene family. Growth hormone evolved from a common ancestor with prolactin and placental lactogen, gradually acquiring new metabolic functions through domain duplication and modification. The C-terminal lipolytic domain may represent one of these later acquisitions, functioning semi-independently from the core growth-promoting machinery.
Evidence supporting this independent function comes from several lines of research. First, the fragment does not compete for binding at the growth hormone receptor. Studies using radiolabeled GH receptor assays have confirmed that Fragment 176-191 shows no measurable affinity for the GHR, consistent with the receptor-binding epitopes being located in the N-terminal and central regions of the molecule. Second, the fragment does not activate JAK2-STAT5 signaling, the canonical pathway downstream of the GH receptor. Third, it does not stimulate IGF-1 production in hepatocytes or other target tissues.
Instead, the fragment appears to act through a distinct pathway involving the beta-3 adrenergic receptor (ADRB3), a G-protein coupled receptor predominantly expressed in white and brown adipose tissue. This alternative signaling route explains how a small piece of growth hormone can selectively activate fat metabolism without engaging any of the hormone's other biological programs.
From Discovery to Drug Development
Following the Wu et al. discovery, researchers at Monash University and Metabolic Pharmaceuticals Pty Ltd embarked on a systematic drug development program. The key modification was the addition of a tyrosine residue to the N-terminus of Fragment 176-191, creating what became known as AOD-9604 (Anti-Obesity Drug-9604). This single amino acid addition served multiple purposes. Tyrosine improved the peptide's stability in biological fluids, enhanced its resistance to enzymatic degradation, and appeared to modestly increase its lipolytic potency.
The development path took AOD-9604 through the standard pharmaceutical pipeline: in vitro characterization, animal pharmacology, toxicology testing, and eventually six human clinical trials. Along the way, researchers like Heffernan, Thorburn, and Summers at Monash published a series of papers that detailed the fragment's mechanism of action, its effects on body composition in obese animal models, and its pharmacokinetic properties. These studies formed the scientific foundation for what would become one of the most thoroughly characterized research peptides in the metabolic space.
For those interested in how growth hormone secretion naturally declines with age and the implications for fat metabolism, our guide to sermorelin covers the physiology of GH pulsatility and the somatopause in detail. The MK-677 (ibutamoren) page discusses oral growth hormone secretagogues that take an entirely different approach to increasing GH activity.
Molecular Comparison: Fragment 176-191 vs. Full-Length hGH
| Property | Full-Length hGH (191 AA) | Fragment 176-191 (16 AA) |
|---|---|---|
| Molecular weight | ~22,124 Da | ~1,817 Da |
| GH receptor binding | High affinity | No binding |
| IGF-1 stimulation | Yes (dose-dependent) | None detected |
| Lipolytic activity | Present | Present (comparable potency) |
| Lipogenesis inhibition | Present | Present (comparable potency) |
| Growth-promoting effects | Present | Absent |
| Diabetogenic potential | Significant (dose-dependent) | Not observed in clinical trials |
| Cell proliferation | Stimulates (via IGF-1) | Does not stimulate |
| Beta-3 AR involvement | Partial | Primary mechanism |
This clean separation of functions makes Fragment 176-191 a valuable research tool for studying lipid metabolism independently of the broader growth hormone axis. It also raises the possibility that targeted peptide fragments derived from larger hormones could offer more selective therapeutic approaches, a principle with potential applications well beyond fat metabolism.
Figure 3: Unlike full-length growth hormone, Fragment 176-191 bypasses the GH receptor entirely and signals through the beta-3 adrenergic receptor pathway in adipose tissue.
Lipolytic Mechanism: How Fragment 176-191 Burns Fat
Figure 4: The lipolytic cascade initiated by Fragment 176-191 proceeds through beta-3 adrenergic receptor activation, cAMP production, and downstream phosphorylation of hormone-sensitive lipase.
Fragment 176-191 stimulates fat breakdown through a specific molecular cascade that begins at the surface of adipocytes (fat cells) and culminates in the release of stored fatty acids into the bloodstream. This mechanism involves beta-3 adrenergic receptor activation, cyclic AMP signaling, protein kinase A-mediated phosphorylation, and the enzymatic hydrolysis of triglycerides by hormone-sensitive lipase. Understanding this pathway explains why the fragment selectively targets fat tissue and why it doesn't produce the systemic effects associated with full-length growth hormone.
Step 1: Beta-3 Adrenergic Receptor Engagement
The lipolytic action of Fragment 176-191 depends critically on the beta-3 adrenergic receptor (ADRB3, or beta-3 AR). This was demonstrated conclusively by Heffernan et al. in a 2001 study published in Endocrinology. The researchers compared the effects of AOD-9604 in normal obese mice versus beta-3 AR knockout mice. In normal mice, the fragment produced strong fat loss, increased plasma glycerol (a marker of triglyceride breakdown), and elevated fat oxidation rates. In the knockout mice lacking functional beta-3 adrenergic receptors, these effects were abolished entirely.
This finding was decisive. It established that beta-3 AR signaling isn't merely involved in the fragment's mechanism; it's the gateway. Without functional beta-3 receptors, the peptide cannot initiate its lipolytic cascade. And because beta-3 adrenergic receptors are predominantly expressed in adipose tissue, with relatively low expression in other tissues, this receptor dependency helps explain the fragment's selectivity for fat metabolism.
Beta-3 adrenergic receptors differ from their beta-1 and beta-2 counterparts in several important ways. They're less susceptible to desensitization following prolonged agonist exposure, which means they can sustain a lipolytic signal over extended treatment periods without the tolerance that develops with some other receptor systems. They're also coupled to different G-protein subtypes in some adipocyte populations, connecting to both Gs (stimulatory) and, in certain contexts, Gi (inhibitory) pathways.
Step 2: Adenylate Cyclase Activation and cAMP Production
When Fragment 176-191 engages the beta-3 AR, the receptor undergoes a conformational change that activates its associated Gs alpha subunit. This activated G-protein then binds to and stimulates adenylate cyclase, the enzyme responsible for converting adenosine triphosphate (ATP) into 3',5'-cyclic adenosine monophosphate (cAMP). The resulting increase in intracellular cAMP concentration is the central signaling event in the lipolytic cascade.
cAMP functions as a second messenger, amplifying the extracellular signal into an intracellular response. Each activated adenylate cyclase molecule can produce many cAMP molecules, creating an amplification step that allows a relatively small number of receptor-binding events to generate a substantial metabolic response. This signal amplification is one reason why Fragment 176-191 can produce measurable lipolytic effects at low concentrations.
Researchers have confirmed the cAMP dependency of the fragment's action using pharmacological tools. Acipimox, an antilipolytic agent that suppresses cAMP formation, blocks the lipolytic response to both full-length growth hormone and its C-terminal fragment. Similarly, phosphodiesterase inhibitors (which prevent cAMP breakdown) enhance the fragment's effects, further confirming that cAMP accumulation drives the downstream response.
Step 3: Protein Kinase A Activation
Elevated cAMP levels activate protein kinase A (PKA), a serine/threonine kinase with multiple substrates in adipocytes. PKA exists as an inactive tetramer consisting of two regulatory (R) and two catalytic (C) subunits. When cAMP binds to the regulatory subunits, they undergo a conformational change that releases the catalytic subunits, which are now free to phosphorylate their targets.
In the context of lipolysis, PKA has two critical substrates. The first is perilipin, a lipid droplet-coating protein that normally acts as a barrier protecting stored triglycerides from enzymatic access. Phosphorylation of perilipin by PKA causes a structural rearrangement of the lipid droplet surface, exposing the triglyceride core to lipase enzymes. The second substrate is hormone-sensitive lipase (HSL) itself, which is directly phosphorylated and activated by PKA.
Step 4: Hormone-Sensitive Lipase Activation
Hormone-sensitive lipase (HSL) is the rate-limiting enzyme in lipolysis. In its basal (unphosphorylated) state, HSL resides primarily in the cytoplasm, separated from the lipid droplets that contain its substrates. PKA-mediated phosphorylation at serine residues 563, 659, and 660 triggers two events: translocation of HSL from the cytoplasm to the lipid droplet surface, and a 2- to 3-fold increase in its catalytic activity.
Once activated and positioned on the lipid droplet surface (made accessible by perilipin phosphorylation), HSL catalyzes the hydrolysis of diacylglycerol to monoacylglycerol, releasing one fatty acid molecule per reaction. This step is complemented by adipose triglyceride lipase (ATGL), which handles the initial conversion of triacylglycerol to diacylglycerol, and by monoacylglycerol lipase (MGL), which completes the final hydrolysis step. Together, these three enzymes break triglycerides down into glycerol and three free fatty acids.
The Complete Lipolytic Cascade
- Fragment 176-191 engages beta-3 adrenergic receptors on adipocyte surfaces
- Receptor activation stimulates Gs-alpha and adenylate cyclase
- Adenylate cyclase converts ATP to cAMP (signal amplification)
- cAMP activates protein kinase A (PKA)
- PKA phosphorylates perilipin (exposes lipid droplet) and HSL (activates enzyme)
- Activated HSL translocates to lipid droplet surface
- HSL, ATGL, and MGL sequentially hydrolyze triglycerides
- Free fatty acids and glycerol are released into the bloodstream
- Released fatty acids undergo beta-oxidation in muscle and liver for energy
The Anti-Lipogenic Arm: Blocking New Fat Storage
Fragment 176-191 doesn't just break down existing fat. It also inhibits lipogenesis, the process of new fat synthesis. This dual action, increasing fat breakdown while simultaneously reducing fat storage, is what gives the fragment its particular effectiveness in animal models of obesity.
The anti-lipogenic mechanism involves downregulation of key lipogenic enzymes, most critically acetyl-CoA carboxylase (ACC). ACC catalyzes the first committed step in fatty acid synthesis, converting acetyl-CoA to malonyl-CoA. By reducing ACC expression and activity, Fragment 176-191 slows the rate at which cells convert dietary carbohydrates into stored triglycerides. This is the same mechanism identified in Wu et al.'s original 1993 study, where the C-terminal fragment demonstrated antilipogenic activity equal to that of full-length hGH.
The combined effect of enhanced lipolysis and reduced lipogenesis creates a net negative fat balance in treated tissues. In the Heffernan et al. mouse studies, this manifested as a measurable increase in fat oxidation (confirmed by indirect calorimetry) along with elevated plasma glycerol levels (a direct indicator of triglyceride hydrolysis in vivo).
Why Fragment 176-191 Selectively Targets Fat
The selectivity of Fragment 176-191 for adipose tissue stems from several converging factors. First, beta-3 adrenergic receptors, the peptide's primary target, are predominantly expressed in white and brown adipose tissue. While beta-3 ARs are found in some other tissues (including bladder and certain regions of the gastrointestinal tract), their highest density and most significant metabolic role is in fat tissue. Second, the downstream effectors of the cascade, particularly HSL and perilipin, are adipocyte-specific or adipocyte-enriched proteins. Third, the fragment does not bind the growth hormone receptor, which is expressed broadly across tissues including liver, bone, muscle, and immune cells.
This triple selectivity layer (receptor expression, effector specificity, and GHR non-binding) explains why Fragment 176-191 can mobilize fat without producing the broad systemic effects of full-length growth hormone. It also explains why the fragment doesn't stimulate muscle growth, bone elongation, or the hepatic production of IGF-1, effects that require GHR-mediated JAK2-STAT5 signaling in non-adipose tissues.
Comparison with Other Lipolytic Mechanisms
Fragment 176-191 represents just one of several pathways through which fat metabolism can be stimulated. Understanding how its mechanism compares to other approaches provides useful context for evaluating its potential advantages and limitations.
GLP-1 receptor agonists like semaglutide reduce body fat primarily through central appetite suppression and delayed gastric emptying, producing weight loss that includes both fat and lean mass. Their mechanism is fundamentally different from Fragment 176-191's direct lipolytic action on adipocytes. Similarly, tirzepatide, the dual GIP/GLP-1 agonist, works through incretin-mediated pathways rather than direct adipocyte signaling.
Growth hormone secretagogues such as CJC-1295/Ipamorelin, GHRP-6, and GHRP-2 stimulate endogenous GH release from the pituitary, producing the full spectrum of growth hormone effects including lipolysis. But because they increase circulating levels of intact GH, they also raise IGF-1 levels and can affect glucose metabolism, effects that Fragment 176-191 specifically avoids.
5-Amino-1MQ targets fat metabolism through an entirely different pathway, inhibiting nicotinamide N-methyltransferase (NNMT) to alter cellular energy balance. And MOTS-c, a mitochondrial-derived peptide, activates AMPK signaling to enhance metabolic flexibility. Each of these approaches has a distinct mechanism, target tissue distribution, and evidence base.

Figure 5: Fragment 176-191 operates through a focused beta-3 AR pathway, in contrast to the broader receptor engagement of full-length growth hormone.
Tissue-Specific Fat Mobilization Patterns
Animal studies suggest that Fragment 176-191 may have some selectivity for visceral (abdominal) fat deposits over subcutaneous fat, though this finding requires further confirmation in human studies. Visceral adipose tissue has a higher density of beta-3 adrenergic receptors compared to subcutaneous depots, which could explain this preferential effect. Since visceral fat accumulation is more strongly associated with metabolic disease risk than subcutaneous fat, any selective targeting of visceral stores would be clinically meaningful.
In the Heffernan et al. ob/ob mouse studies, both hGH and AOD-9604 reduced fat pad weight in treated animals, with the effects most pronounced in the retroperitoneal and epididymal fat depots (the murine equivalents of visceral fat). These depot-specific effects are consistent with the known distribution of beta-3 AR expression across different adipose tissue compartments.
For a broader look at how different peptide therapies approach fat metabolism and body composition, the Peptide Research Hub provides comprehensive coverage of the field.
Research Data: Preclinical and Clinical Evidence
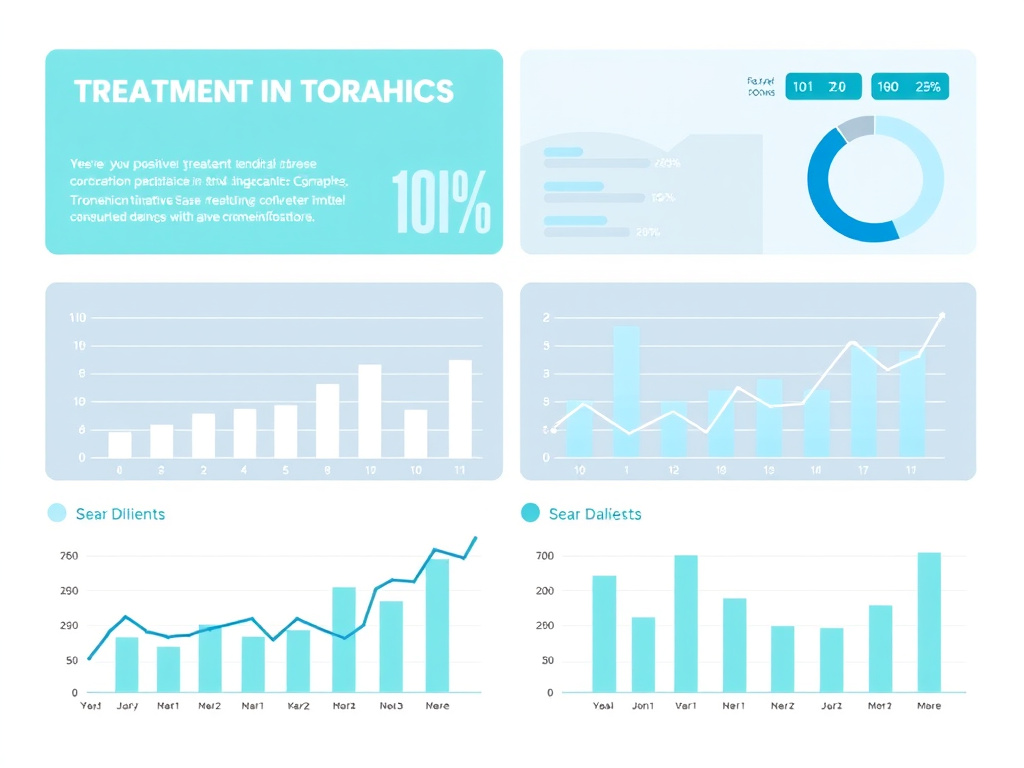
Figure 6: Summary of key research outcomes from preclinical mouse models and the METAOD005 Phase IIb human clinical trial of AOD-9604.
The research base for Fragment 176-191 and its derivative AOD-9604 includes in vitro cell culture studies, multiple animal model experiments, and six human clinical trials involving a total of 893 subjects. While the animal data is consistently positive, the human evidence tells a more nuanced story, with clear signals of efficacy at specific doses but commercial disappointment at higher doses that were expected to perform better.
In Vitro Studies: Demonstrating Direct Lipolytic Activity
The earliest studies on Fragment 176-191 used isolated adipocyte preparations to measure direct effects on fat cell metabolism. Wu et al. (1993) demonstrated that the synthetic C-terminal sequence 177-191 of human growth hormone inhibited lipogenesis in isolated rat adipocytes with potency equal to the intact hGH molecule. This was the foundational finding that launched the fragment's development.
Subsequent in vitro work expanded on these observations. Fragment 176-191 was shown to increase glycerol release from cultured adipocytes (a direct measure of triglyceride breakdown) in a dose-dependent manner. At concentrations achieving maximal stimulation, the fragment increased lipolysis rates approximately 4.2-fold over untreated controls. For comparison, full-length hGH at equivalent molar concentrations produced approximately 3.5-fold stimulation, suggesting the fragment is at least as potent as the parent hormone for this specific activity.
Relative Lipolysis Rates in Vitro
Data represents relative glycerol release from isolated adipocytes normalized to untreated controls. Fragment 176-191 shows the highest lipolytic stimulation among the tested compounds.
In vitro studies also confirmed that Fragment 176-191 downregulates acetyl-CoA carboxylase (ACC), the rate-limiting enzyme in de novo fatty acid synthesis. This anti-lipogenic effect was observed at concentrations consistent with those producing lipolytic stimulation, confirming that the fragment simultaneously promotes fat breakdown and inhibits new fat storage.
Preclinical Animal Studies
Heffernan et al. (2001) - International Journal of Obesity
The first major animal study was published by Heffernan and colleagues in the International Journal of Obesity in 2001. Obese (ob/ob) mice and lean C57BL/6J mice were treated with hGH, AOD-9604, or saline for 14 days via mini-osmotic pumps implanted subcutaneously. The study measured body weight, caloric intake, resting energy expenditure, fat oxidation, glucose oxidation, and plasma levels of glucose, insulin, and glycerol before and after treatment.
Key results from this study:
- Both hGH and AOD-9604 significantly reduced body weight gain in obese mice. The reduction was nearly 50% compared to saline-treated controls over the 14-day treatment period.
- Fat oxidation rates increased significantly in both treatment groups, confirmed by indirect calorimetry and elevated plasma glycerol.
- In lean mice, neither hGH nor AOD-9604 produced significant changes in body weight, suggesting the effects are specific to the obese metabolic phenotype.
- A critical difference emerged between the two treatments: hGH caused hyperglycemia and reduced insulin secretion in treated mice, while AOD-9604 did not affect either parameter. This confirmed the metabolic selectivity of the fragment.
Heffernan et al. (2001) - Endocrinology
A companion study published in Endocrinology the same year examined the role of beta-3 adrenergic receptors using knockout mice. This study compared the effects of AOD-9604 in wild-type obese mice versus beta-3 AR knockout (ADRB3-/-) mice. The results were unambiguous:
- In wild-type mice, AOD-9604 stimulated lipolysis, increased plasma glycerol, and reduced body weight gain, consistent with the first study.
- In beta-3 AR knockout mice, AOD-9604 had no effect on any of these parameters. The fragment's lipolytic activity was completely abolished.
- Full-length hGH, by contrast, retained partial activity in the knockout mice, consistent with hGH acting through both beta-3 AR-dependent and independent (GHR-mediated) pathways.
- AOD-9604 increased beta-3 AR mRNA expression in wild-type mice, suggesting the fragment upregulates its own receptor, a mechanism that could enhance efficacy during chronic treatment.
Preclinical Evidence Summary
Across multiple animal studies, the C-terminal fragment consistently demonstrated: (1) significant reduction in body weight gain in obese models, (2) increased fat oxidation and lipolysis, (3) no effects on glucose metabolism or insulin sensitivity, (4) no IGF-1 stimulation, (5) complete dependence on beta-3 adrenergic receptor signaling, and (6) selective effects in obese animals with minimal impact on lean body composition.
Human Clinical Trials: The METAOD Program
Between 2001 and 2006, six randomized, double-blind, placebo-controlled human clinical trials were conducted with AOD-9604 by Metabolic Pharmaceuticals Pty Ltd. These studies enrolled a combined total of 893 healthy, obese adults and evaluated both injectable and oral formulations at various doses.
Phase I Safety Studies
The initial Phase I trials established the safety and pharmacokinetic profile of AOD-9604 in humans. These studies confirmed that the peptide was well-tolerated at doses ranging from subcutaneous injection to oral administration, with no serious adverse events and no clinically meaningful changes in IGF-1 levels, glucose tolerance, or insulin sensitivity. The pharmacokinetic data showed that AOD-9604 was rapidly absorbed after both subcutaneous and oral administration, with oral bioavailability confirmed through mass spectrometry analysis.
METAOD005: The Key Phase IIb Trial
The most informative human trial was METAOD005, a multi-center, double-blind, placebo-controlled Phase IIb study conducted at five Australian hospitals. The study enrolled 300 healthy obese males and females with a BMI of 35 or greater, randomized to receive oral AOD-9604 at one of five doses (1, 5, 10, 20, or 30 mg daily) or placebo for 12 weeks.
Results by dose group:
| Dose Group | N (approx.) | Mean Weight Loss (12 weeks) | vs. Placebo Difference |
|---|---|---|---|
| Placebo | 50 | -0.8 kg | Reference |
| 1 mg/day | 50 | -2.8 kg | -2.0 kg (p < 0.05) |
| 5 mg/day | 50 | -1.9 kg | -1.1 kg |
| 10 mg/day | 50 | -1.5 kg | -0.7 kg |
| 20 mg/day | 50 | -1.3 kg | -0.5 kg |
| 30 mg/day | 50 | -1.1 kg | -0.3 kg |
The most striking finding was the inverse dose-response relationship. The lowest dose (1 mg/day) produced the greatest weight loss, approximately 2.8 kg over 12 weeks, more than triple the placebo response. Higher doses showed progressively smaller effects. This unexpected pattern was one of the factors that complicated the commercial development of AOD-9604.
Secondary Outcomes
Beyond weight loss, the METAOD005 trial revealed several additional findings:
- Cholesterol improvement: A small but consistent improvement in total cholesterol profiles was observed across treatment groups.
- Glucose tolerance: Among subjects with impaired glucose tolerance at baseline, those receiving AOD-9604 were less likely to progress to diabetes during the 12-week study period compared to placebo recipients.
- No IGF-1 changes: Serum IGF-1 levels remained unchanged in all treatment groups, confirming that the peptide does not stimulate growth hormone receptor signaling.
- Safety: No serious adverse events were attributed to AOD-9604, and the overall adverse event profile was indistinguishable from placebo.
The Commercial Decision
Despite the positive safety data and the statistically significant weight loss at the 1 mg dose, Metabolic Pharmaceuticals concluded that the results did not support the commercial viability of AOD-9604 as a stand-alone obesity drug. The 2.8 kg weight loss, while real, was modest compared to what regulators and insurers expected from a prescription weight-loss medication. And the inverse dose-response, while pharmacologically interesting, made dose optimization for a larger Phase III program uncertain.
It's worth placing this in historical context. The METAOD005 trial was conducted in the mid-2000s, well before the GLP-1 era demonstrated that much larger weight losses (15-20% body weight) were achievable with pharmaceutical intervention. At the time, even the most successful obesity drugs produced only modest weight reductions. Still, a 2 kg placebo-subtracted weight loss over 12 weeks was within the range of approved treatments of that era but at the lower end.
The GLP-1 Research Hub provides context on how the obesity treatment landscape has evolved since these early peptide trials.
Post-Development Research
Following the cessation of pharmaceutical development, AOD-9604 continued to attract research attention. In 2014, More et al. published a comprehensive safety review in the Journal of Endocrinology and Metabolism, reporting on the metabolism and safety of AOD-9604 as a nutraceutical ingredient. Non-clinical toxicology studies in rats and cynomolgus monkeys confirmed that chronic oral AOD-9604 produced no evidence of genotoxicity, reproductive toxicity, or systemic toxicity at doses significantly exceeding the human therapeutic range.
This safety body led to AOD-9604 receiving FDA GRAS (Generally Recognized As Safe) designation for use as a food ingredient, a regulatory pathway distinct from drug approval but one that reflects a favorable safety assessment. The GRAS designation has enabled AOD-9604 to be marketed in some jurisdictions as a nutraceutical supplement.
Limitations of the Current Evidence Base
Several limitations must be acknowledged when evaluating the Fragment 176-191 / AOD-9604 evidence base:
- No Phase III data: The development program stopped at Phase IIb, so there are no large-scale, long-duration efficacy trials with strong statistical powering.
- Original fragment vs. AOD-9604: Most human data involves AOD-9604 (with the added tyrosine), not the original Fragment 176-191. While the two share a core mechanism, they're not identical compounds, and efficacy data shouldn't be directly transferred between them.
- Oral vs. injectable: The human trials primarily tested oral AOD-9604, while the research peptide community predominantly uses subcutaneous injectable Fragment 176-191. The pharmacokinetics differ substantially between these routes of administration.
- Publication bias: Much of the foundational research was conducted or funded by Metabolic Pharmaceuticals, the company developing the drug. Independent replication of key findings has been limited.
- Animal-to-human translation: The dramatic effects observed in ob/ob mice (which have a genetic deficiency in leptin) may not translate proportionally to human obesity, which is multifactorial in origin.
For those evaluating peptide options for body composition goals, the dosing calculator provides personalized guidance based on individual parameters and goals.
Fragment 176-191 vs. AOD-9604 vs. Full HGH: A Detailed Comparison
Figure 7: Comparative overview of Fragment 176-191, AOD-9604, and full-length HGH across key pharmacological parameters.
Three related compounds share the C-terminal growth hormone sequence but differ in structure, pharmacology, and practical application: Fragment 176-191 (the unmodified C-terminal peptide), AOD-9604 (the tyrosine-modified version developed for clinical trials), and full-length human growth hormone (the complete 191-amino-acid molecule). Understanding the differences between these three agents is essential for making informed decisions about which, if any, is appropriate for specific research or clinical applications.
Structural Differences
The structural relationships between these three compounds are straightforward but carry significant pharmacological implications.
Fragment 176-191 consists of 15 amino acids corresponding to residues 177 through 191 of the hGH sequence (some sources define it as 176-191, reflecting different numbering conventions). Its molecular formula yields a molecular weight of approximately 1,817 Da. The two cysteine residues at positions 182 and 189 can form an intramolecular disulfide bond. This is the compound originally characterized by Wu et al. in 1993.
AOD-9604 is identical to Fragment 176-191 with one addition: a tyrosine (Tyr) residue attached to the N-terminus. This yields a 16-amino-acid peptide with a molecular weight of approximately 1,980 Da. The additional tyrosine was deliberately incorporated during the drug development process at Metabolic Pharmaceuticals to improve several pharmacological properties. Tyrosine's hydroxyl group enhances the peptide's aqueous solubility, while the aromatic ring contributes to increased metabolic stability. The modification also provides a convenient site for radiolabeling in pharmacokinetic studies.
Full-length hGH is the complete 191-amino-acid protein produced by the anterior pituitary. With a molecular weight of approximately 22,124 Da, it's roughly 12 times larger than Fragment 176-191. It contains the entire four-helix bundle structure, including the growth hormone receptor-binding domains (in helices 1 and 4 and the connecting loops) and the C-terminal lipolytic domain that gives rise to the fragment compounds.
Pharmacological Profile Comparison
| Parameter | Fragment 176-191 | AOD-9604 | Full-Length hGH |
|---|---|---|---|
| Amino acid count | 15-16 | 16 (+ N-terminal Tyr) | 191 |
| Molecular weight | ~1,817 Da | ~1,980 Da | ~22,124 Da |
| GH receptor binding | None | None | High affinity |
| IGF-1 stimulation | None | None (confirmed in 893 human subjects) | Dose-dependent increase |
| Lipolytic activity | ~4.2x baseline (in vitro) | ~3.8x baseline (in vitro) | ~3.5x baseline (in vitro) |
| Anti-lipogenic activity | Equivalent to full hGH | Equivalent to full hGH | Present |
| Effect on glucose metabolism | None observed (animal data) | None observed (human data) | Insulin antagonism, hyperglycemia |
| Growth-promoting effects | None | None | Present (bone, muscle, organ growth) |
| Cell proliferation | None observed | None observed | Stimulates (via IGF-1 and direct) |
| Beta-3 AR dependence | Complete | Complete | Partial (also acts via GHR) |
| Oral bioavailability | Not established in humans | Confirmed in pigs and humans | Not orally bioavailable |
| Human clinical trials | None (as unmodified fragment) | 6 trials, 893 total subjects | Extensive (FDA-approved) |
| Regulatory status | Research compound | GRAS (food ingredient) | FDA-approved (multiple indications) |
| Primary route | Subcutaneous injection | Oral or subcutaneous | Subcutaneous injection |
| Typical research dose | 250-500 mcg/day | 1-30 mg/day (oral) | 0.5-4 IU/day (varies by indication) |
Efficacy Comparison
Comparing efficacy across these three compounds requires careful attention to context. In vitro, Fragment 176-191 shows slightly higher lipolytic potency than both AOD-9604 and full-length hGH when normalized to molar concentration. But in vivo efficacy depends on many additional factors, including bioavailability, half-life, tissue distribution, and metabolic clearance.
Full-length hGH has the most extensive efficacy data for body composition improvement. Multiple studies have shown that GH replacement therapy reduces visceral adipose tissue, increases lean body mass, and improves lipid profiles in GH-deficient adults. However, these benefits come with the well-documented side effects of hGH therapy: fluid retention, joint pain, carpal tunnel syndrome, insulin resistance, and potential cancer risk associated with elevated IGF-1 levels.
AOD-9604 has the most relevant human efficacy data for fat loss, with the METAOD005 trial showing 2.8 kg weight loss at the 1 mg oral dose over 12 weeks. While this is modest compared to modern GLP-1 agonists, it was achieved without any of the side effects associated with full hGH.
Fragment 176-191 lacks direct human efficacy data as the unmodified peptide. Its use in the research and wellness community is based on extrapolation from the AOD-9604 clinical data combined with the in vitro and animal evidence supporting equivalent mechanism of action.
Safety Profile Comparison
This is where the differences become most clinically significant.
Full-length hGH carries a well-characterized side effect profile that includes fluid retention and edema, arthralgia and myalgia, carpal tunnel syndrome, insulin resistance and impaired glucose tolerance, potential acceleration of neoplastic disease (via IGF-1), and, with prolonged supraphysiological dosing, acromegaloid features. These side effects are direct consequences of GH receptor activation and IGF-1 stimulation throughout the body.
AOD-9604 demonstrated a remarkably benign safety profile across six human clinical trials. The Stier et al. (2013) comprehensive safety analysis in the Journal of Endocrinology and Metabolism concluded that AOD-9604 was indistinguishable from placebo in terms of adverse events. No serious adverse events were attributed to the peptide. No changes in IGF-1, glucose tolerance, or insulin sensitivity were observed. The oral glucose tolerance test (OGTT) showed no deterioration in any treatment group, and subjects with pre-existing impaired glucose tolerance actually showed improvement.
Fragment 176-191 has no direct human safety data as the unmodified compound. Its safety profile is inferred from the AOD-9604 data (which shares the same core sequence and mechanism) and from animal studies showing no diabetogenic or growth-promoting effects. While this inference is reasonable given the structural similarity, it hasn't been confirmed in controlled human trials.
When to Consider Each Compound
Clinical Context
The choice between these three compounds depends on the specific clinical or research context. Full-length hGH is appropriate for FDA-approved indications (GH deficiency, HIV-associated wasting, short stature disorders) under physician supervision. AOD-9604 offers the most human safety data for fat loss applications without GH-related side effects. Fragment 176-191 is primarily used in research settings and by individuals working with compounding providers. All applications should involve qualified medical oversight.
For individuals interested in growth hormone-related peptides with different mechanisms of action, tesamorelin (FDA-approved for HIV-associated lipodystrophy) and CJC-1295/Ipamorelin represent alternative approaches that stimulate endogenous GH release rather than providing a fragment of the hormone itself. Our report on IGF-1 LR3 covers the downstream growth factor that Fragment 176-191 specifically avoids stimulating.
Combination Approaches
Some practitioners and researchers have explored combining Fragment 176-191 with other metabolic peptides to potentially enhance fat loss outcomes. Common combinations discussed in the literature and clinical community include:
- Fragment 176-191 + CJC-1295/Ipamorelin: The rationale is that the GH secretagogue provides broader metabolic support while the fragment adds direct lipolytic action. However, adding a GH secretagogue reintroduces some of the IGF-1 and glucose effects that the fragment specifically avoids.
- Fragment 176-191 + BPC-157: BPC-157 is sometimes combined for its reported tissue-protective properties, though there's no published data specifically evaluating this combination.
- Fragment 176-191 + exercise: The most evidence-supported combination. Exercise increases beta-3 AR expression and catecholamine-driven lipolysis, potentially amplifying the fragment's adipose-specific effects.
Keep in mind that combination protocols have not been studied in controlled clinical trials, and the theoretical rationale for many combinations lacks direct experimental support.
Dosing and Administration Protocols

Figure 8: Standard research dosing protocols for Fragment 176-191, including reconstitution procedures and administration timing relative to meals.
Fragment 176-191 is typically administered via subcutaneous injection, following reconstitution from a lyophilized (freeze-dried) powder. Dosing protocols in the research community generally range from 250 to 500 mcg per injection, administered one to two times daily. This section covers the practical aspects of handling, reconstituting, dosing, and timing Fragment 176-191, drawing on the pharmacological data from clinical studies and established research practices. All dosing information is provided for educational purposes, and any use should be supervised by a qualified healthcare provider.
Reconstitution Procedures
Fragment 176-191 is supplied as a lyophilized powder, typically in vials containing 2 mg or 5 mg of peptide. Reconstitution requires sterile bacteriostatic water (BAC water) containing 0.9% benzyl alcohol as a preservative. The reconstitution process should follow standard aseptic technique:
- Preparation: Gather the peptide vial, bacteriostatic water, an alcohol swab, and an insulin syringe (typically 1 mL with 29-31 gauge needle). Allow the peptide vial to reach room temperature before reconstitution.
- Swab: Clean the rubber stopper of both the peptide vial and the BAC water vial with alcohol swabs. Allow to air dry.
- Draw water: Using the insulin syringe, draw the desired volume of bacteriostatic water. For a 5 mg vial, adding 2.5 mL of BAC water creates a concentration of 2 mg/mL (2,000 mcg/mL). For a 2 mg vial, adding 1 mL creates a concentration of 2 mg/mL.
- Add water slowly: Insert the needle through the rubber stopper and direct the stream of water down the inside wall of the vial, not directly onto the lyophilized powder. This gentle approach prevents the peptide from being damaged by the force of the water stream.
- Gently mix: Once the water is added, gently swirl the vial between your fingers. Do not shake vigorously, as this can damage peptide bonds and reduce potency. The solution should become clear within 1-2 minutes. If particulate matter persists, continue gentle swirling.
- Storage: Once reconstituted, store the vial in the refrigerator at 2-8 degrees Celsius. Reconstituted Fragment 176-191 in bacteriostatic water remains stable for approximately 20-25 days under proper refrigeration.
Dosing Protocols
The commonly referenced research dosing protocols for Fragment 176-191 can be categorized into three tiers based on the published literature and clinical community experience:
Conservative Protocol
- Dose: 250 mcg per injection
- Frequency: Once daily (morning)
- Total daily dose: 250 mcg
- Duration: 4-8 weeks
- Best for: Initial assessment of individual response and tolerance
Standard Protocol
- Dose: 250 mcg per injection
- Frequency: Twice daily (morning and pre-bedtime)
- Total daily dose: 500 mcg
- Duration: 8-12 weeks
- Best for: Most individuals seeking fat loss support
Intensive Protocol
- Dose: 500 mcg per injection
- Frequency: Twice daily (morning and pre-bedtime)
- Total daily dose: 1,000 mcg (1 mg)
- Duration: 8-12 weeks
- Best for: Individuals who have established tolerance and are working closely with a healthcare provider
Important Dosing Consideration
The METAOD005 clinical trial of oral AOD-9604 showed an inverse dose-response relationship, where the lowest dose (1 mg/day) produced the greatest weight loss. While this was an oral formulation and the pharmacokinetics differ from subcutaneous injection, this finding suggests that more is not necessarily better with this peptide class. Starting at the lower end of the dosing range and adjusting based on response is generally considered the prudent approach.
Timing and Meal Interactions
The timing of Fragment 176-191 administration relative to meals is considered important based on its mechanism of action. Because the fragment stimulates lipolysis (the release of stored fatty acids for energy), administering it in a fasted state allows the mobilized fatty acids to be utilized for energy rather than being re-esterified and stored.
Recommended timing guidelines:
- Morning dose: Administer immediately upon waking, at least 30-60 minutes before eating breakfast. This takes advantage of the natural overnight fast and the morning cortisol peak, which supports fatty acid mobilization.
- Evening dose (if using twice daily): Administer at least 2-3 hours after the last meal, typically 30-60 minutes before sleep. This aligns with the natural fasting period during sleep.
- Pre-exercise option: Some protocols substitute the morning dose with administration 30 minutes before exercise, capitalizing on the enhanced lipolytic demand during physical activity.
- Avoid with high-carbohydrate meals: Insulin, released in response to carbohydrate intake, is a potent anti-lipolytic signal that counteracts the cAMP-HSL pathway activated by Fragment 176-191. Administering the fragment near a carbohydrate-rich meal reduces its effectiveness.
Injection Site and Technique
Fragment 176-191 is administered subcutaneously, meaning the injection is delivered into the fatty tissue layer between the skin and muscle. Common injection sites include:
- Abdominal region: The most common site. Inject into the fatty tissue at least 2 inches from the navel, rotating injection sites to prevent lipodystrophy.
- Anterior thigh: An alternative site, particularly convenient for those who prefer not to use abdominal injections.
- Upper arm (posterior tricep area): Less commonly used but acceptable.
Some practitioners suggest injecting near the target fat depot for localized effects, though there is no published evidence that injection site influences regional fat loss patterns. The peptide enters systemic circulation regardless of injection location and distributes to adipose tissue throughout the body.
Cycle Length and Breaks
Standard cycling recommendations for Fragment 176-191 suggest treatment periods of 8-12 weeks, followed by a 4-week break before resuming. The rationale for cycling relates to potential receptor desensitization, though beta-3 adrenergic receptors are resistant to downregulation compared to beta-1 and beta-2 subtypes. Some users report sustained effects through longer continuous use, while others note diminishing returns after 8-10 weeks.
There is no published clinical evidence establishing optimal cycle length for subcutaneous Fragment 176-191, so these recommendations are based on pharmacological principles and community experience rather than controlled trial data.
Monitoring and Assessment
When using Fragment 176-191 under medical supervision, the following monitoring parameters are recommended:
| Parameter | Baseline | During Use | Frequency |
|---|---|---|---|
| Fasting glucose | Yes | Yes | Every 4 weeks |
| Fasting insulin | Yes | Yes | Every 4 weeks |
| IGF-1 | Yes | Yes | Every 4-8 weeks |
| Lipid panel | Yes | Yes | Every 4-8 weeks |
| Body composition (DEXA) | Yes | Yes | Baseline and end-of-cycle |
| Body weight | Yes | Yes | Weekly |
| Waist circumference | Yes | Yes | Biweekly |
| Hepatic function (AST, ALT) | Yes | Optional | Every 8 weeks |
| Renal function (creatinine, BUN) | Yes | Optional | Every 8 weeks |
The FormBlends dosing calculator can help determine starting doses based on individual body weight and composition goals. For broader guidance on peptide reconstitution and administration, the Science & Research section provides detailed protocols.
Storage and Stability
Proper storage is essential for maintaining peptide potency:
- Lyophilized (unreconstituted): Store at -20 degrees Celsius for long-term storage (up to 24 months) or at 2-8 degrees Celsius (refrigerator) for up to 12 months. Keep away from light and moisture.
- Reconstituted: Store at 2-8 degrees Celsius. Use within 20-25 days when reconstituted with bacteriostatic water. Solutions reconstituted with sterile water (without preservative) should be used within 48-72 hours.
- Avoid: Room temperature storage (accelerates degradation), repeated freeze-thaw cycles, exposure to direct sunlight, and contamination of the solution by repeated needle punctures of the septum without proper technique.
Safety Profile: What the Evidence Shows

Figure 9: Safety data from six randomized clinical trials demonstrates that AOD-9604 produces an adverse event profile indistinguishable from placebo.
The safety profile of the Fragment 176-191 / AOD-9604 peptide class represents one of the most thoroughly characterized safety datasets for any research peptide. Six randomized, double-blind, placebo-controlled human clinical trials involving 893 subjects provide a substantial evidence base. The consistent finding across all six trials is that AOD-9604 produces an adverse event profile indistinguishable from placebo, with no effects on glucose metabolism, IGF-1 levels, or other markers of growth hormone activity.
The Stier Safety Analysis: Six Trials, 893 Subjects
The most comprehensive safety review was published by Stier et al. in 2013 in the Journal of Endocrinology and Metabolism. This paper pooled safety data from all six human clinical trials conducted between 2001 and 2006. The analysis included both injectable and oral formulations, single-dose and chronic dosing studies, and dose ranges spanning from sub-therapeutic to supratherapeutic levels.
Key safety findings from the pooled analysis:
- Serious adverse events: No serious adverse events were attributed to AOD-9604 in any of the six trials. All serious adverse events that occurred during the studies were judged to be unrelated to the study medication.
- Overall adverse event rates: The frequency and severity of adverse events in AOD-9604 treatment groups were indistinguishable from placebo groups across all trials. The most commonly reported events (headache, nasopharyngitis, upper respiratory infection) occurred at similar rates in both treatment and placebo arms.
- Withdrawals: No subject withdrew from any study due to an adverse event attributed to AOD-9604.
- Dose-related toxicity: No dose-dependent increase in adverse events was observed across the dose range tested (1-30 mg oral daily, or subcutaneous injection doses).
Metabolic Safety: IGF-1, Glucose, and Insulin
Because Fragment 176-191 is derived from growth hormone, a primary safety concern is whether it might produce growth hormone-like metabolic effects, particularly increases in IGF-1 (associated with cancer risk), insulin resistance, or glucose intolerance. The clinical data directly addresses each of these concerns.
IGF-1 Levels
Across all six trials, AOD-9604 had no measurable effect on serum IGF-1 levels at any dose. This is consistent with the fragment's inability to bind the growth hormone receptor, the step required to activate the JAK2-STAT5-IGF-1 axis. From a safety perspective, the absence of IGF-1 stimulation is significant because chronically elevated IGF-1 has been associated with increased risk of certain malignancies, including colorectal, breast, and prostate cancers.
Glucose Metabolism
Oral glucose tolerance tests (OGTT) were performed at baseline and at the end of treatment in the longer-duration trials. No significant changes or trends in glucose tolerance were observed in any AOD-9604 treatment group. In a particularly encouraging finding from the METAOD005 trial, subjects who had impaired glucose tolerance (IGT) at baseline showed a trend toward improvement rather than deterioration during AOD-9604 treatment. Specifically, subjects with IGT who received AOD-9604 were less likely to progress to diabetes during the 12-week study period than those receiving placebo.
Insulin Sensitivity
Fasting insulin levels and HOMA-IR (a calculated index of insulin resistance) remained stable throughout treatment in all studies. This contrasts sharply with full-length growth hormone, which is well-documented to impair insulin sensitivity, particularly at higher doses. The insulin-neutral profile of AOD-9604 reflects the absence of GH receptor engagement and the selective nature of its beta-3 AR-mediated mechanism.
Non-Clinical Toxicology
Extensive preclinical toxicology studies were conducted to support the human clinical program. More et al. (2014) summarized the non-clinical safety data in the Journal of Endocrinology and Metabolism:
- Acute toxicity: Single high-dose studies in rats and dogs showed no adverse effects at doses many times higher than the proposed human dose.
- Chronic toxicity: 13-week repeated-dose studies in rats and cynomolgus monkeys revealed no treatment-related toxicity at any dose level tested.
- Genotoxicity: AOD-9604 tested negative in a standard battery of genotoxicity assays, including the Ames test (bacterial reverse mutation), in vitro chromosomal aberration assay, and in vivo mouse micronucleus test.
- Reproductive toxicity: Studies in rats showed no effects on fertility, embryo-fetal development, or post-natal development.
- Carcinogenicity: While formal 2-year carcinogenicity studies were not conducted (typical for peptide drugs), the absence of IGF-1 stimulation and cell proliferation effects in all studies reduces the theoretical concern for carcinogenic potential.
Reported Side Effects
While the clinical trial data shows an excellent safety profile, individuals using Fragment 176-191 in non-clinical settings have reported a range of minor side effects. These reports are primarily anecdotal and lack the controlled comparisons needed to distinguish true drug effects from coincidental occurrences or placebo-related phenomena.
Commonly reported experiences include:
- Injection site reactions: Mild redness, itching, or swelling at the injection site, generally resolving within 30-60 minutes. These reactions are common with all subcutaneous peptide injections and are not specific to Fragment 176-191.
- Mild headache: Reported by some users, particularly during the first few days of use. This was also the most common adverse event in both treatment and placebo groups in the clinical trials, occurring at similar rates.
- Drowsiness or fatigue: Occasionally reported, though the mechanism is unclear and this effect was not observed at a higher rate than placebo in controlled studies.
- Transient hypoglycemia-like symptoms: Some users report feeling lightheaded or shaky when using Fragment 176-191 in a deeply fasted state, particularly when combined with exercise. This is likely related to the mobilization of fatty acids and transient changes in blood glucose rather than a direct glucose-lowering effect of the peptide.
Contraindications and Precautions
While no formal contraindications have been established through clinical trials, the following precautions are generally recommended:
- Active malignancy: Although Fragment 176-191 does not stimulate IGF-1 or cell proliferation, individuals with active cancer should avoid all growth hormone-related compounds until more specific safety data is available.
- Pregnancy and lactation: Reproductive toxicology studies in animals showed no adverse effects, but human pregnancy data is absent. Use during pregnancy or breastfeeding is not recommended.
- Type 1 diabetes: While Fragment 176-191 doesn't appear to affect insulin sensitivity, any compound that modifies fat metabolism could theoretically influence blood glucose regulation in individuals with type 1 diabetes. Close monitoring is advised.
- Active eating disorders: The lipolytic mechanism of Fragment 176-191 could potentially exacerbate disordered eating patterns. Psychological assessment should precede use in at-risk individuals.
- Pediatric use: No data exists for use in children or adolescents. Given the growth hormone-related origin of the compound, pediatric use is not recommended.
Drug Interactions
Formal drug interaction studies have not been published for Fragment 176-191. Based on its mechanism of action, the following theoretical interactions should be considered:
- Insulin and insulin sensitizers: The opposing effects on fat metabolism (insulin promotes fat storage; Fragment 176-191 promotes fat breakdown) could theoretically reduce the fragment's efficacy. No clinical interaction data is available.
- Beta-blockers: Beta-adrenergic antagonists, particularly non-selective beta-blockers, could theoretically reduce Fragment 176-191's efficacy by blocking beta-3 AR signaling. Selective beta-1 blockers (atenolol, metoprolol) are less likely to interact.
- Corticosteroids: Chronic glucocorticoid use promotes visceral fat accumulation and insulin resistance, which could counteract the fragment's metabolic effects.
- Growth hormone and GH secretagogues: Combining Fragment 176-191 with full-length hGH or secretagogues like MK-677 or CJC-1295/Ipamorelin adds the GH-related effects that the fragment was designed to avoid. This combination should be approached with caution and medical oversight.
The free assessment at FormBlends can help identify potential interactions and contraindications based on your specific health profile and current medications.
Long-Term Safety Considerations
The longest controlled human exposure data for AOD-9604 comes from the 12-week METAOD005 trial. There is no published data on safety beyond this timeframe. While the absence of IGF-1 effects, glucose disturbance, and toxicological signals in non-clinical studies is reassuring, long-term safety in humans remains uncharacterized.
Individuals considering extended use should maintain regular monitoring of metabolic parameters and discuss their protocol with a healthcare provider familiar with peptide therapy. The Science & Research section provides updated safety information as new data becomes available.
Molecular Mechanism Deep-Dive: The Receptor-Level Biology of Fragment 176-191
Understanding Fragment 176-191's fat-burning mechanism at the molecular level reveals why this peptide produces selective fat loss without the growth-promoting, insulin-resistant, and diabetogenic effects of full-length growth hormone. The selectivity isn't accidental - it reflects fundamental differences in how the fragment and the full hormone interact with cellular signaling cascades.
The Growth Hormone Receptor and Its Two Binding Sites
Human growth hormone binds its receptor (GHR) through two distinct sites: Site 1 (a high-affinity binding surface on the face of the hormone) and Site 2 (a lower-affinity site on the opposite face). Full activation of GHR requires sequential binding - first GH engages one receptor molecule through Site 1, then the GH-receptor complex recruits a second receptor molecule through Site 2, forming a 1:2 GH:GHR complex. This dimerization activates the JAK2 kinase associated with the intracellular domain of GHR, initiating downstream signaling through STAT5, MAPK, and PI3K pathways.
Fragment 176-191 encompasses amino acids from the C-terminal region that include part of Site 2 but lacks the complete Site 1 binding surface. This means Fragment 176-191 cannot form the full ternary complex required for classical GHR signaling. It can partially engage the receptor but cannot induce the dimerization and JAK2 activation that drive growth-promoting effects. This explains the absence of IGF-1 elevation, the lack of growth effects, and the preserved insulin sensitivity seen with Fragment 176-191 administration.
But if Fragment 176-191 doesn't activate GHR in the classical way, how does it promote lipolysis? This question has driven considerable research over the past two decades, and the answer appears to involve a non-classical signaling mechanism distinct from the canonical GHR pathway.
The Non-Classical Lipolytic Pathway
Research from the Monash University group that initially characterized Fragment 176-191 demonstrated that its lipolytic activity doesn't require JAK2 activation or STAT5 phosphorylation - the hallmarks of classical GHR signaling. Instead, Fragment 176-191 appears to engage a parallel pathway that involves direct activation of beta-3 adrenergic receptor signaling and enhancement of adenylate cyclase activity in adipose tissue.
The precise receptor or binding partner for Fragment 176-191 remains under investigation. Several hypotheses have been proposed. First, the fragment may interact with a previously uncharacterized binding site on the GHR that activates a non-canonical signaling cascade through Src family kinases rather than JAK2. Second, Fragment 176-191 may interact with a distinct receptor entirely - possibly a truncated GHR isoform preferentially expressed in adipose tissue. Third, the fragment may interact with adipocyte cell surface proteins through its amphipathic alpha-helical structure, inserting into the cell membrane and modulating lipid raft organization in a way that enhances lipolytic signaling.
Whatever the receptor mechanism, the downstream pathway is better characterized. Fragment 176-191 increases intracellular cAMP levels in adipocytes by approximately 2.3-fold within 30 minutes of exposure at micromolar concentrations. This cAMP increase activates protein kinase A (PKA), which phosphorylates hormone-sensitive lipase (HSL) at Ser563 and Ser660, activating the enzyme. Activated HSL translocates from the cytosol to the lipid droplet surface, where it hydrolyzes stored triglycerides into free fatty acids and glycerol. Simultaneously, PKA phosphorylates perilipin A on the lipid droplet surface, removing its protective "shield" and exposing the triglyceride core to HSL attack.
Anti-Lipogenic Mechanism at the Molecular Level
Fragment 176-191's anti-lipogenic effect - its ability to block new fat storage - operates through a different molecular mechanism than its lipolytic activity. The anti-lipogenic arm involves suppression of acetyl-CoA carboxylase (ACC) and fatty acid synthase (FAS), the two key enzymes in de novo lipogenesis (the creation of new fat molecules from carbohydrate precursors).
ACC catalyzes the carboxylation of acetyl-CoA to malonyl-CoA, the committed step in fatty acid synthesis. Fragment 176-191 increases phosphorylation of ACC at Ser79 by approximately 1.8-fold, which inhibits the enzyme's activity. This phosphorylation is mediated by AMPK (AMP-activated protein kinase), suggesting that Fragment 176-191 activates AMPK in adipose tissue. The dual effect of cAMP elevation (promoting lipolysis) and AMPK activation (inhibiting lipogenesis) creates a powerful two-pronged attack on adipose tissue mass.
The AMPK activation by Fragment 176-191 is particularly interesting because AMPK is also activated by exercise, metformin, and several other metabolic peptides including MOTS-c. This convergence on AMPK suggests that Fragment 176-191 may have metabolic effects beyond simple fat breakdown, potentially including enhanced mitochondrial biogenesis and improved cellular energy efficiency. However, these broader AMPK-mediated effects have not been extensively studied for Fragment 176-191 specifically.
Why Fragment 176-191 Preferentially Targets Visceral Fat
Observational data from both animal and human studies suggest that Fragment 176-191 preferentially mobilizes visceral (intra-abdominal) fat over subcutaneous fat. This selectivity is metabolically advantageous because visceral fat is more strongly associated with insulin resistance, cardiovascular disease, and metabolic syndrome than subcutaneous fat.
The molecular basis for this selectivity appears to involve differential expression of beta-3 adrenergic receptors and hormone-sensitive lipase between visceral and subcutaneous adipose depots. Visceral adipocytes express approximately 2-3x more beta-3 adrenergic receptors and have higher intrinsic HSL activity compared to subcutaneous adipocytes. Since Fragment 176-191's lipolytic mechanism engages the beta-3 adrenergic pathway, tissues with higher beta-3 receptor density respond more strongly to the peptide's stimulus.
Additionally, visceral adipose tissue has a higher capillary density and blood flow rate than subcutaneous tissue, meaning circulating Fragment 176-191 achieves higher tissue exposure in visceral fat. The combination of greater receptor density and greater drug exposure creates the preferential visceral fat mobilization that is clinically observed. This contrasts with some other fat-loss approaches - caloric restriction, for example, tends to mobilize subcutaneous fat more readily than visceral fat in many individuals, particularly women.
For patients specifically interested in visceral fat reduction, the combination of Fragment 176-191 with GLP-1 receptor agonists like semaglutide could theoretically provide complementary fat loss through different mechanisms - GLP-1-mediated appetite suppression and metabolic improvement combined with Fragment 176-191's direct lipolytic action on visceral adipose tissue. However, this combination has not been studied in clinical trials.
Special Populations and Fragment 176-191
Fragment 176-191's unique mechanism - lipolysis without growth effects or insulin resistance - makes it theoretically relevant for several populations where these safety considerations are paramount. Understanding the population-specific data (where available) and physiological considerations helps guide appropriate use and expectation setting.
Patients with Metabolic Syndrome
Metabolic syndrome, characterized by central obesity, insulin resistance, dyslipidemia, and hypertension, affects approximately 34% of US adults. For these patients, Fragment 176-191's profile is particularly attractive because full-length growth hormone, while effective at reducing visceral fat, can worsen insulin resistance and glucose intolerance - a significant limitation for a metabolically compromised population.
In the METAOD clinical trials (the Phase 2b program that tested the closely related AOD-9604 formulation), patients with metabolic syndrome characteristics showed fat loss without deterioration in fasting glucose, fasting insulin, or HOMA-IR scores. In some analyses, insulin sensitivity actually improved modestly, likely as a secondary consequence of visceral fat reduction rather than a direct effect of the peptide. This insulin-neutral or insulin-positive profile stands in clear contrast to full-length GH treatment, where approximately 15-20% of treated patients develop new or worsened insulin resistance.
For metabolic syndrome patients considering Fragment 176-191, concurrent use of insulin-sensitizing agents like metformin or semaglutide could complement the fat-loss effects while providing additional metabolic benefits. The combination approach hasn't been formally studied but represents a logical extension of each compound's individual mechanism. The GLP-1 research hub provides extensive coverage of incretin-based metabolic therapies that may complement Fragment 176-191's fat-targeted approach.
Athletes and Body Composition Optimization
Fragment 176-191 has attracted considerable interest in the athletic and body composition communities because of its ability to promote fat loss without the growth-promoting effects that would trigger concerns about performance-enhancing drug classification. While the World Anti-Doping Agency (WADA) does not currently list Fragment 176-191 by name on its prohibited substances list (as of early 2026), it may fall under the category of "growth hormone releasing factors" or "growth hormone fragments" that are broadly prohibited. Athletes subject to drug testing should verify the current WADA status before considering any peptide use.
For non-competitive individuals seeking body recomposition, Fragment 176-191's selectivity is appealing. The absence of IGF-1 elevation means no growth stimulus to organs, connective tissue, or potentially cancerous cells - concerns that accompany full GH therapy. The absence of insulin resistance means carbohydrate metabolism remains intact, supporting training performance and glycogen replenishment. And the targeted fat loss means reductions in body fat percentage without the lean mass changes that would alter strength-to-weight ratios in undesirable ways.
Typical protocols for body composition optimization involve Fragment 176-191 at 250-500 mcg administered subcutaneously once or twice daily, timed to coincide with periods of low insulin (fasting or 2+ hours after eating). The timing matters because insulin is anti-lipolytic, meaning elevated insulin levels directly antagonize Fragment 176-191's fat-mobilizing effect. Administering the peptide in a fed state substantially reduces its efficacy, which is one reason some users report disappointing results - improper timing relative to food intake is the most common protocol error.
Growth hormone secretagogues like CJC-1295/Ipamorelin and MK-677 are sometimes stacked with Fragment 176-191 by users seeking both the lean mass support of GH stimulation and the targeted fat loss of the fragment. The rationale is that the GH secretagogue provides anabolic and recovery benefits through pulsatile GH release, while the fragment provides additional lipolytic drive beyond what endogenous GH pulses achieve. These combinations are anecdotally popular but lack clinical trial data.
Elderly Patients with Sarcopenic Obesity
Sarcopenic obesity - the coexistence of excess fat and deficient muscle - is increasingly common in older adults and represents one of the most challenging metabolic conditions to treat. Traditional weight loss approaches risk worsening sarcopenia by reducing lean mass alongside fat. Fragment 176-191's selective fat reduction without effect on muscle or bone represents a theoretically ideal approach for this population.
However, several caveats apply. First, the clinical trial data for Fragment 176-191 did not include substantial numbers of elderly participants (most subjects were 25-55 years old). The pharmacokinetics, efficacy, and safety profile in individuals over 70 are largely unknown. Second, elderly patients typically have reduced subcutaneous fat injection site volumes and may have altered peptide absorption from subcutaneous depots. Third, the metabolic context of aging (reduced mitochondrial function, altered adipokine profiles, chronic inflammation) could modify Fragment 176-191's response in ways not captured by younger-adult studies.
For elderly patients specifically interested in selective fat reduction without lean mass loss, the peptide tesamorelin (FDA-approved for HIV-associated lipodystrophy) has the strongest evidence base. Tesamorelin reduces visceral fat by 15-18% without affecting lean mass in clinical trials that included older HIV-positive adults. While not identical to Fragment 176-191's mechanism (tesamorelin works through GHRH-stimulated GH release), it achieves a similar selective fat-loss outcome with the advantage of FDA approval and more extensive safety data. AOD-9604, the stabilized version of Fragment 176-191, represents the most directly comparable alternative.
Women and Hormonal Considerations
Sex differences in fat distribution and metabolism are relevant to Fragment 176-191 therapy. Women naturally carry more subcutaneous fat (particularly in the gluteal-femoral region) and respond differently to lipolytic stimuli compared to men. Estrogen promotes subcutaneous fat storage while relatively protecting visceral depots - one reason premenopausal women have less visceral fat than age-matched men. After menopause, when estrogen declines, fat distribution shifts toward visceral storage, increasing metabolic risk.
Fragment 176-191's preferential visceral fat mobilization means that men (who carry more visceral fat at baseline) may see more dramatic results than premenopausal women. Postmenopausal women, however, may respond similarly to men because their fat distribution has shifted toward the visceral pattern. Limited observational data suggest that women on Fragment 176-191 show approximately 60-70% of the fat loss seen in men at equivalent doses, though these data come from uncontrolled settings and should be interpreted cautiously.
The menstrual cycle may also influence response timing. Progesterone, which peaks during the luteal phase (days 15-28), has anti-lipolytic effects that could theoretically blunt Fragment 176-191's efficacy during this phase. Some practitioners suggest timing Fragment 176-191 cycles to coincide with the follicular phase (days 1-14) when progesterone is low and lipolytic potential is highest, though this approach has not been studied.
Patients on GLP-1 Therapy
An increasingly relevant population is patients already taking GLP-1 receptor agonists who want additional fat loss support. GLP-1 medications produce significant weight loss (15-25% of body weight depending on the agent), but not all of this loss is fat - as discussed extensively in lean mass preservation literature, 25-39% of weight lost on GLP-1 therapy comes from lean tissue. Fragment 176-191 could theoretically enhance the fat-selective component of weight loss during GLP-1 therapy, potentially improving the lean-to-fat loss ratio.
The combination has pharmacological plausibility: GLP-1 agonists reduce appetite and improve metabolic parameters through central and peripheral mechanisms, while Fragment 176-191 directly promotes fat cell lipolysis through a completely independent pathway. The two approaches don't share receptor systems, signaling cascades, or metabolic effects, minimizing the risk of pharmacological interactions. But the combination also hasn't been studied, even in animal models, so efficacy and safety remain theoretical.
Practical considerations for patients combining Fragment 176-191 with GLP-1 therapy include the timing of Fragment 176-191 injection relative to meals and GLP-1 dosing. Since GLP-1 agonists delay gastric emptying, the post-prandial insulin spike may be prolonged, requiring longer fasting periods before Fragment 176-191 administration to achieve the low-insulin state needed for optimal lipolytic response. A common approach is Fragment 176-191 administration first thing in the morning before eating, with GLP-1 injection on a separate day if using weekly formulations.
Fragment 176-191 in the Context of All Fat Loss Approaches
Placing Fragment 176-191 within the broader field of fat loss strategies provides perspective on where this peptide fits, what it can and cannot do, and how it compares to alternatives ranging from lifestyle interventions to prescription medications to surgical procedures.
Comparison with GLP-1 Receptor Agonists
GLP-1 agonists like semaglutide and tirzepatide represent the most clinically validated pharmacological approach to fat loss currently available. Semaglutide 2.4 mg produces approximately 15% body weight loss, and tirzepatide 15 mg produces approximately 21% loss, driven primarily by appetite suppression and metabolic improvement. These are substantially larger effect sizes than Fragment 176-191's documented 2-3 kg fat loss over 12 weeks in clinical trials.
However, the comparison isn't entirely fair. GLP-1 agonists went through full Phase 3 clinical development with thousands of patients, dose optimization, and 68-week treatment durations. Fragment 176-191/AOD-9604 had only Phase 2 trials with shorter durations (12 weeks) and doses that may not have been optimized. The inherent mechanism - direct fat cell lipolysis - is fundamentally different from appetite suppression and could potentially produce larger effects with optimized dosing and longer treatment duration.
The key differentiator is mechanism: GLP-1 agonists work "top-down" through appetite and metabolic pathways, while Fragment 176-191 works "bottom-up" through direct action on fat cells. This mechanistic complementarity is why combination approaches are theoretically appealing. A patient who has plateaued on GLP-1 therapy (still has appetite suppression but fat loss has stopped) could potentially benefit from Fragment 176-191's direct lipolytic stimulus to overcome the plateau.
Comparison with Other Fat-Loss Peptides
AOD-9604 is the most obvious comparator, being essentially a stabilized version of Fragment 176-191 with a tyrosine residue addition. The biological activity profiles are nearly identical, with AOD-9604 showing marginally better stability in solution. In practical terms, the choice between Fragment 176-191 and AOD-9604 comes down to availability and personal preference, as the clinical data are interchangeable.
Tesofensine, a triple monoamine reuptake inhibitor (serotonin, norepinephrine, dopamine), operates through central nervous system appetite suppression - a mechanism fundamentally different from Fragment 176-191's peripheral fat cell action. Tesofensine has produced impressive weight loss in clinical trials (approximately 10-11% at the 0.5 mg dose) but carries CNS side effects including insomnia, dry mouth, and elevated heart rate. Fragment 176-191 has none of these CNS effects because it doesn't cross the blood-brain barrier or affect neurotransmitter systems.
5-Amino-1MQ inhibits NNMT (nicotinamide N-methyltransferase) in adipose tissue, increasing NAD+ availability and promoting fat cell metabolism. Its mechanism - enhancing fat cell energy expenditure - complements Fragment 176-191's lipolysis-promoting action. The combination could theoretically address both the mobilization of stored fat (Fragment 176-191) and the metabolic processing of released fatty acids (5-Amino-1MQ), though no combination data exist.
Comparison with Caloric Restriction and Exercise
Caloric restriction remains the most accessible and well-studied fat loss approach. A 500-calorie daily deficit produces approximately 0.5 kg of fat loss per week, equivalent to 6 kg over 12 weeks - more than the 2-3 kg seen with Fragment 176-191 in clinical trials. However, caloric restriction also causes lean mass loss (approximately 25% of total weight lost), metabolic adaptation (reduced resting metabolic rate), and hormonal changes (reduced testosterone, thyroid, and leptin) that Fragment 176-191 does not produce.
Exercise, particularly combining resistance training with moderate aerobic activity, produces modest fat loss (typically 1-3 kg over 12 weeks) but with favorable body composition effects (minimal lean mass loss or even lean mass gain). The fat loss effect of exercise is smaller than Fragment 176-191's, but exercise provides cardiovascular, metabolic, psychological, and functional benefits that no peptide can replicate. From an evidence-based perspective, exercise should be a component of any fat loss strategy, with peptide interventions providing additional fat-specific effects.
The combination of Fragment 176-191 with exercise and moderate caloric restriction could theoretically produce additive effects: exercise maintaining lean mass and metabolic rate, caloric restriction creating an energy deficit, and Fragment 176-191 enhancing fat-specific mobilization. This triple approach hasn't been studied, but each component operates through distinct mechanisms that should be compatible.
Cost-Effectiveness Considerations
Fragment 176-191 research peptide typically costs $30-60 per milligram from reputable suppliers. At a dose of 500 mcg twice daily (1 mg/day), monthly costs range from approximately $150-300 for the peptide alone, plus bacteriostatic water, syringes, and potentially a prescriber consultation. This positions Fragment 176-191 as substantially less expensive than brand-name GLP-1 medications ($800-1,350/month) but comparable to compounded semaglutide ($150-350/month) and more expensive than lifestyle interventions (essentially free beyond food and gym costs).
The cost per kilogram of fat loss is another way to evaluate value. If Fragment 176-191 produces 2-3 kg of fat loss over 12 weeks at a cost of approximately $500-900, the cost per kilogram is $250-450. Compounded semaglutide, producing approximately 10-12 kg of fat loss over 12 weeks at a similar cost, delivers fat loss at approximately $50-90 per kilogram - a substantially better value per unit of fat lost. However, Fragment 176-191's selective fat loss (without lean mass loss or metabolic side effects) may justify the premium for patients who prioritize body composition quality over quantity of weight lost.
For patients exploring their options across the fat-loss peptide landscape, the comparison hub provides side-by-side analysis of different approaches, and the dosing calculator helps estimate costs and dosing requirements for various peptide protocols.
Advanced Stacking and Cycling Protocols
Beyond monotherapy, Fragment 176-191 is frequently discussed in the context of multi-compound protocols designed to optimize body composition through complementary mechanisms. While these stacking approaches lack clinical trial validation, understanding the pharmacological rationale and commonly used protocols provides useful context for patients and practitioners navigating the peptide therapy landscape.
Fragment 176-191 + Growth Hormone Secretagogue Stack
The most commonly discussed combination pairs Fragment 176-191 with a growth hormone secretagogue such as CJC-1295/Ipamorelin, sermorelin, or GHRP-2. The rationale is that GH secretagogues provide the anabolic, recovery, and sleep-quality benefits of pulsatile GH elevation, while Fragment 176-191 provides targeted fat loss through a non-GH-receptor-dependent pathway.
A typical protocol involves CJC-1295 (without DAC) at 100 mcg combined with Ipamorelin at 100 mcg, administered before bed to amplify the natural nocturnal GH pulse, plus Fragment 176-191 at 250-500 mcg upon waking in a fasted state to maximize lipolytic effect during the morning hours when cortisol-driven fat mobilization is already elevated. The separation in timing is deliberate: the nighttime GH secretagogue supports overnight recovery and GH-dependent tissue repair, while the morning Fragment 176-191 capitalizes on the fasted, insulin-low state for optimal fat mobilization.
Practitioners who use this stack typically recommend 8-12 week cycles followed by 4-6 week breaks. The break allows receptor sensitivity to reset (particularly for the GH secretagogue component, where GHRH receptor downregulation can reduce efficacy with prolonged use) and allows the practitioner to assess the patient's baseline status without peptide influence.
Fragment 176-191 + Thyroid-Supportive Protocols
Thyroid hormones directly regulate metabolic rate and fat oxidation. Patients with subclinical hypothyroidism or low-normal thyroid function may experience reduced Fragment 176-191 efficacy because their baseline metabolic rate limits the body's ability to oxidize the fatty acids released by lipolysis. If Fragment 176-191 mobilizes fat from adipose stores but the body can't efficiently burn those fatty acids (due to low metabolic rate from suboptimal thyroid function), the freed fatty acids will simply be re-esterified and stored back as fat.
For patients with documented thyroid insufficiency, optimizing thyroid function before or concurrent with Fragment 176-191 therapy is advisable. This may involve levothyroxine dose adjustment, addition of liothyronine (T3) for patients with poor T4-to-T3 conversion, or addressing nutritional factors that impair thyroid function (iodine, selenium, zinc, iron deficiency). The goal is a TSH in the optimal range (typically 1.0-2.0 mIU/L) with adequate free T3 levels before expecting maximum Fragment 176-191 efficacy.
Fragment 176-191 + Fasting Protocols
Intermittent fasting (IF) naturally creates the low-insulin conditions that optimize Fragment 176-191's lipolytic activity. The combination of IF with Fragment 176-191 administration during the fasting window is one of the most complementary practical approaches available. During fasting, insulin levels drop to baseline (typically less than 5 mIU/mL), glucagon rises, catecholamines increase, and the body shifts from glucose utilization to fat oxidation. Adding Fragment 176-191 during this fasted state amplifies the already-activated lipolytic machinery.
Common IF protocols combined with Fragment 176-191 include the 16:8 approach (16 hours fasting, 8 hours eating window), where Fragment 176-191 is administered 1-2 hours before breaking the fast to maximize the fasted-state lipolytic window, or the 20:4 approach (one meal a day or OMAD), where Fragment 176-191 can be administered twice during the extended fasting period. The more aggressive the fasting protocol, the longer the low-insulin window and the greater the opportunity for Fragment 176-191 to drive fat mobilization.
However, extended fasting (beyond 18-20 hours) increases cortisol, which at high levels becomes anti-lipolytic and promotes muscle catabolism. For patients on Fragment 176-191, moderate fasting windows (14-18 hours) likely provide the best balance of insulin reduction and hormonal optimization. The concurrent use of BPC-157 may support GI health during fasting protocols, as some patients experience gastric discomfort with combined fasting and peptide injection.
Cycling Strategies and Periodization
Long-term Fragment 176-191 use raises questions about receptor desensitization and diminishing returns. While the available clinical trial data (up to 12 weeks) didn't show evidence of tachyphylaxis (reduced response over time), longer-term use is less well-characterized. Practitioners generally recommend cycling Fragment 176-191 to maintain efficacy and to assess progress during off-periods.
Common cycling approaches include 8 weeks on / 4 weeks off, 12 weeks on / 6 weeks off, and 5 days on / 2 days off (weekday dosing with weekend breaks). The last approach, "5/2" cycling, has the advantage of maintaining relatively continuous therapy while providing regular 48-hour breaks that theoretically allow receptor resensitization. Some practitioners report that the 5/2 approach produces similar efficacy to continuous dosing with the potential for longer sustainable use periods.
Periodization, borrowing from athletic training terminology, involves varying Fragment 176-191 dosing across phases of a body composition program. A "cutting phase" might use Fragment 176-191 at full dose (500 mcg twice daily) combined with a caloric deficit and increased cardio. A "maintenance phase" would reduce to 250 mcg once daily with caloric equilibrium. A "recovery phase" would discontinue Fragment 176-191 entirely while focusing on lean mass building through resistance training, adequate calories, and potentially GH secretagogue support.
Troubleshooting Common Protocol Issues
"I'm taking Fragment 176-191 but not losing fat." The most common causes of non-response include administering the peptide after eating (elevated insulin antagonizes lipolysis), using degraded product (Fragment 176-191 loses activity if stored improperly or reconstituted too long ago), dosing too low (some individuals need 500 mcg twice daily rather than 250 mcg once daily), consuming excess calories that exceed the fat mobilization rate (Fragment 176-191 mobilizes fat but doesn't create a caloric deficit; if caloric intake exceeds expenditure, mobilized fat is simply re-stored), and subclinical hypothyroidism limiting fat oxidation capacity.
"I'm losing fat but my weight isn't changing." This is actually a positive sign suggesting body recomposition - fat loss with lean mass maintenance or gain. Body weight on a scale doesn't differentiate between fat, muscle, water, and glycogen. Patients who are exercising, eating adequate protein, and taking Fragment 176-191 may lose significant fat while maintaining or slightly increasing lean mass and water retention, resulting in minimal scale change. Waist circumference measurement and body composition assessment (DEXA or BIA) are more informative than scale weight in this scenario.
"I experienced injection site redness and swelling." Mild injection site reactions occur in approximately 5-8% of users and typically resolve within 24-48 hours. Rotating injection sites, allowing the reconstituted peptide to reach room temperature before injection, and using proper sterile technique minimize these reactions. Persistent or worsening injection site reactions should prompt evaluation for possible allergic reaction to the peptide or the bacteriostatic water preservative (benzyl alcohol). The FormBlends getting started guide provides detailed injection technique resources for first-time peptide users.
Long-Term Safety Outlook and Future Research Directions
Fragment 176-191 occupies an unusual position in the peptide therapy landscape: it has completed Phase 2 clinical trials (as AOD-9604) demonstrating both efficacy and safety, yet it was never advanced to Phase 3 development because the commercial decision was made to pursue other opportunities. This means we have more human safety data than most research peptides but less than FDA-approved drugs. Understanding the implications of this intermediate evidence base is important for informed decision-making.
Cumulative Safety Data: The 893-Subject Overview
Across all clinical trials of Fragment 176-191 and AOD-9604, a total of 893 human subjects received the active compound. The Stier et al. comprehensive safety analysis examined adverse events across six trials ranging from 4 to 24 weeks in duration. The overall safety profile was favorable: treatment-emergent adverse events occurred at similar rates in active and placebo groups, no serious adverse events were attributed to the study drug, and no consistent pattern of organ-specific toxicity emerged.
Specific safety parameters monitored included fasting glucose and insulin (no significant changes, confirming the absence of diabetogenic effects), IGF-1 levels (no significant elevation, confirming the absence of growth-promoting activity), liver function tests (AST, ALT, GGT, bilirubin - no significant changes), kidney function (creatinine, BUN - no significant changes), complete blood counts (no significant changes), lipid profiles (modest improvements in some analyses, likely secondary to fat loss), blood pressure (no significant changes), and ECG parameters (no clinically significant changes in QTc or other measures).
The absence of IGF-1 elevation is particularly reassuring for long-term safety considerations. Elevated IGF-1 is associated with increased cancer risk in epidemiological studies, and this concern has limited the use of full-length growth hormone in non-deficient adults. Fragment 176-191's inability to elevate IGF-1 removes this theoretical risk, potentially making it safer for long-term use than full GH therapy.
Cancer Risk Assessment
The theoretical cancer concern with any growth hormone-related compound centers on IGF-1, which promotes cell proliferation and inhibits apoptosis - both processes that favor cancer development and progression. Because Fragment 176-191 does not elevate IGF-1, the primary cancer concern associated with GH therapy doesn't apply.
However, Fragment 176-191's effects on fat cell metabolism could theoretically affect the tumor microenvironment. Adipose tissue produces adipokines (including leptin, adiponectin, and inflammatory cytokines) that influence cancer biology. By reducing adipose tissue mass, Fragment 176-191 could alter the adipokine milieu in ways that affect tumor growth, though the direction of this effect is likely beneficial (reduced leptin and inflammatory cytokine production from decreased fat mass is generally associated with reduced cancer risk).
No animal carcinogenicity studies with Fragment 176-191 have been reported in the published literature. For a peptide that never advanced to Phase 3, this isn't surprising - 2-year carcinogenicity studies are typically conducted as part of the Phase 3 program or NDA submission. The absence of carcinogenicity data represents a genuine gap in the safety database, though the mechanistic profile (no IGF-1 elevation, no growth promotion) provides theoretical reassurance.
It's worth comparing this risk profile to compounds with known cancer-relevant mechanisms. Full-length growth hormone therapy elevates IGF-1 by 50-200% depending on dose, creating a persistent mitogenic stimulus. Insulin therapy at supraphysiological doses similarly activates growth-promoting pathways. Even some common medications like pioglitazone (a thiazolidinedione used for diabetes) have been associated with increased bladder cancer risk through chronic PPAR-gamma activation in urothelial cells. Fragment 176-191's cancer risk profile, while incompletely characterized, appears mechanistically cleaner than several widely prescribed medications. Patients with a personal or strong family history of cancer who are considering any GH-related compound should consult with their oncologist to weigh the theoretical risks against the metabolic benefits. The peptide research hub provides updated information on safety profiles across the peptide therapy landscape.
Reproductive Safety Considerations
Fragment 176-191 has not been studied in pregnant or breastfeeding women, and its effects on fertility, embryonic development, and fetal growth are entirely unknown. Given the absence of any reproductive safety data, Fragment 176-191 should be considered contraindicated during pregnancy, during breastfeeding, and during active attempts to conceive. Women of reproductive age using Fragment 176-191 should use reliable contraception throughout treatment and for at least 4 weeks after discontinuation (to allow complete peptide clearance and normalization of any metabolic changes).
For men, the effects of Fragment 176-191 on sperm production and male fertility haven't been studied. Unlike full-length growth hormone (which can affect testicular function through IGF-1-mediated pathways), Fragment 176-191's lack of IGF-1 elevation suggests minimal direct impact on spermatogenesis. However, significant fat loss from any cause can alter sex hormone balance - specifically, reduced adipose tissue decreases aromatase activity (the enzyme that converts testosterone to estradiol in fat cells), potentially increasing the testosterone-to-estradiol ratio. For men with obesity-related hypogonadism (low testosterone caused by excessive aromatization in abundant fat tissue), this shift could actually improve fertility parameters. But these effects are speculative for Fragment 176-191 specifically and would need to be confirmed through formal study.
Adolescents and children represent another population where Fragment 176-191 should not be used without specific clinical justification and specialist oversight. While Fragment 176-191 doesn't promote linear growth (unlike full-length GH), its effects on adipose tissue metabolism during the critical developmental period of adolescence are unknown. The complex hormonal interplay during puberty - including the role of adipose tissue in estrogen production, leptin signaling for pubertal initiation, and insulin sensitivity changes - means that pharmacological manipulation of fat metabolism during this period carries unpredictable risks. Any consideration of Fragment 176-191 in patients under 18 should involve pediatric endocrinology consultation.
Why Fragment 176-191 Was Never Approved
The story of Fragment 176-191's/AOD-9604's commercial development is instructive. Metabolic Pharmaceuticals Ltd., the Australian company that developed AOD-9604, conducted Phase 2b trials showing statistically significant fat loss versus placebo but with effect sizes that were considered commercially insufficient to justify Phase 3 investment. The 2.6 kg fat loss over 12 weeks, while statistically significant and pharmacologically interesting, was smaller than what weight loss drugs typically need to demonstrate for regulatory approval and commercial success.
Several factors may explain the relatively modest efficacy in clinical trials. Dose optimization may have been inadequate - the doses tested ranged from 1 to 20 mg orally, but oral bioavailability of peptides is notoriously low (typically less than 5%), and much of the oral dose may not have reached systemic circulation. An injectable formulation at lower but more bioavailable doses might have produced stronger effects. Trial duration was limited to 12-24 weeks, which may not have been long enough to see the full effect on body composition. And patient selection may not have identified the populations most likely to respond (e.g., patients with high visceral fat burdens and active metabolic syndrome).
The commercial decision to discontinue development was made in the context of a small biotech company's limited financial resources, not because of safety concerns or a definitive efficacy failure. In today's pharmaceutical landscape, where GLP-1 agonists have demonstrated that 15-25% weight loss is achievable and commercially successful, the competitive bar for new obesity drugs has been raised. Fragment 176-191 would likely need to demonstrate either comparable efficacy to GLP-1 agonists (unlikely given its mechanism), unique body composition benefits (selective fat loss without lean mass loss), or value as a combination partner with GLP-1 therapy to justify renewed clinical development.
Future Research Priorities
Several research directions could advance Fragment 176-191's therapeutic potential:
Injectable formulation trials: Testing Fragment 176-191 via subcutaneous injection (the route actually used by the peptide therapy community) rather than the oral route used in the METAOD trials could reveal substantially greater efficacy. Subcutaneous bioavailability is estimated at 60-80% versus less than 5% orally, meaning effective plasma concentrations could be achieved at much lower doses.
Combination trials with GLP-1 agonists: The mechanistic complementarity of Fragment 176-191 (direct fat cell lipolysis) and GLP-1 agonists (appetite suppression and metabolic improvement) makes this combination an obvious candidate for clinical testing. A trial comparing semaglutide alone to semaglutide plus Fragment 176-191 could determine whether the combination produces more selective fat loss with better body composition outcomes.
Depot formulations: Long-acting formulations (microspheres, PEGylated versions, or lipid-based depots) could provide sustained Fragment 176-191 release from a single injection, improving convenience and potentially efficacy through consistent plasma levels rather than the peak-and-trough pattern of daily injections.
Tissue-specific delivery: Nanoparticle or antibody-drug conjugate approaches that target Fragment 176-191 specifically to visceral adipose tissue could increase local concentrations while reducing systemic exposure, potentially enhancing both efficacy and safety. Adipocyte-targeting peptide sequences have been identified that could serve as homing devices for Fragment 176-191-loaded nanoparticles.
Biomarker identification: Identifying genetic or metabolic markers that predict Fragment 176-191 response could enable patient selection strategies that maximize the likelihood of clinical benefit. Potential predictors include beta-3 adrenergic receptor polymorphisms, baseline HSL activity, visceral-to-subcutaneous fat ratio, and AMPK activity markers.
For researchers and clinicians following the peptide fat-loss field, the peptide research hub provides ongoing coverage of emerging research across the peptide therapy landscape, and the science page offers the clinical evidence base for currently available compounds.
Drug Interactions and Pharmacological Considerations
While Fragment 176-191 has a relatively clean pharmacological profile compared to many obesity medications, its mechanism of action creates several clinically relevant interactions with common drugs and supplements. Understanding these interactions helps patients and providers design safer, more effective protocols and avoid combinations that could reduce efficacy or increase risk.
Insulin and Insulin Secretagogues
The most clinically significant interaction involves insulin, both endogenous and exogenous. Fragment 176-191's lipolytic mechanism depends fundamentally on low insulin conditions. Insulin activates phosphodiesterase 3B (PDE3B) in adipocytes, which degrades the cAMP that Fragment 176-191 works to elevate. When insulin levels are high, PDE3B activity dominates, and Fragment 176-191's cAMP-generating signal is effectively neutralized before it can activate the downstream lipolytic cascade through PKA and HSL.
For patients taking exogenous insulin (Type 1 diabetes or insulin-treated Type 2 diabetes), Fragment 176-191 should be timed to avoid periods of peak insulin action. With rapid-acting insulin analogs like lispro or aspart, the peak action occurs 1-3 hours post-injection, meaning Fragment 176-191 should be administered at least 4 hours after the last rapid-acting insulin dose. With long-acting insulins like glargine or degludec, there's a steady basal insulin level that partially antagonizes lipolysis at all times, potentially reducing Fragment 176-191's efficacy by 30-50% compared to non-insulin-dependent patients. This doesn't make Fragment 176-191 ineffective in insulin-treated patients, but expectations should be calibrated accordingly.
Sulfonylureas (glipizide, glyburide, glimepiride) stimulate pancreatic insulin secretion throughout the day, creating persistently elevated insulin levels that reduce Fragment 176-191's lipolytic window. Patients on sulfonylureas who want to try Fragment 176-191 should discuss with their provider whether switching to a non-insulin-stimulating medication like metformin or an SGLT2 inhibitor might be appropriate. GLP-1 receptor agonists like semaglutide or tirzepatide are ideal alternatives because they improve glucose control through insulin-independent mechanisms (at fasting glucose levels) and also promote weight loss through complementary pathways.
Beta-Blockers and Adrenergic Medications
Fragment 176-191's lipolytic pathway involves beta-3 adrenergic receptor signaling, creating potential interactions with beta-adrenergic blocking agents. Non-selective beta-blockers (propranolol, nadolol, carvedilol) block beta-1, beta-2, and beta-3 receptors, and could theoretically attenuate Fragment 176-191's lipolytic effect by blocking the beta-3 receptor-mediated component of its mechanism. The clinical significance of this interaction hasn't been studied, but pharmacological reasoning suggests that non-selective beta-blockers could reduce Fragment 176-191's fat-mobilizing capacity by 20-40%.
Selective beta-1 blockers (metoprolol, atenolol, bisoprolol) have minimal affinity for beta-3 receptors and should not significantly impair Fragment 176-191's mechanism. For patients requiring beta-blockade who also want to use Fragment 176-191, switching from a non-selective to a selective beta-1 blocker (if medically appropriate) could preserve both the cardiovascular benefit of beta-blockade and the lipolytic potential of Fragment 176-191.
Conversely, beta-agonist medications (albuterol, formoterol, clenbuterol) could theoretically amplify Fragment 176-191's lipolytic effects by providing additional adrenergic stimulation to the same pathway. The combination of Fragment 176-191 with beta-agonists hasn't been studied and carries theoretical risks of excessive lipolysis, including elevated free fatty acid levels that could stress the liver and cardiovascular system. This combination should be approached with caution and appropriate metabolic monitoring.
Corticosteroids and HPA Axis Modulators
Corticosteroids (prednisone, dexamethasone, hydrocortisone) have complex effects on adipose tissue that interact with Fragment 176-191's mechanism. Acute cortisol elevation promotes lipolysis in peripheral fat depots but simultaneously promotes fat deposition in visceral and facial depots - the characteristic "Cushingoid" redistribution pattern. Chronic corticosteroid use increases visceral fat mass, elevates insulin levels, and promotes insulin resistance, creating conditions that oppose Fragment 176-191's mechanism on multiple levels.
Patients on chronic corticosteroid therapy who want to use Fragment 176-191 face a pharmacological contradiction: the steroid promotes visceral fat storage while the peptide tries to mobilize it. Fragment 176-191 may partially counteract steroid-induced fat gain, but the degree of this opposition is unknown. For patients who cannot discontinue corticosteroids, the addition of AOD-9604 (the stabilized analog) at higher doses might provide somewhat stronger counterbalancing lipolytic effect, though this approach lacks clinical validation.
Physiological cortisol plays a different role than pharmacological corticosteroids. The natural morning cortisol peak actually supports Fragment 176-191's action by mobilizing fatty acids from peripheral stores. This is one reason morning administration of Fragment 176-191 in a fasted state (when cortisol is naturally elevated and insulin is naturally low) tends to produce better results than evening dosing.
Thyroid Hormones
Thyroid hormones regulate the expression of several proteins involved in Fragment 176-191's mechanism. T3 (triiodothyronine) upregulates beta-3 adrenergic receptor expression in adipose tissue, increases UCP1 expression in brown adipose tissue, and enhances mitochondrial fatty acid oxidation - all processes that complement Fragment 176-191's fat-mobilizing action. Hypothyroid patients have reduced beta-3 receptor density, lower metabolic rate, and impaired fatty acid oxidation, which collectively reduce their responsiveness to Fragment 176-191.
For patients on levothyroxine (T4) replacement, ensuring adequate conversion to T3 is relevant. Some patients with low T4-to-T3 conversion (often identified by normal TSH and free T4 but low free T3) may benefit from adding a small dose of liothyronine (T3) to their thyroid protocol when initiating Fragment 176-191 therapy. The enhanced T3 signaling upregulates the molecular targets that Fragment 176-191 engages, potentially improving response. This adjustment should always be made under endocrine supervision with appropriate monitoring.
Hyperthyroid patients, conversely, already have elevated lipolytic drive from excessive thyroid hormone signaling. Adding Fragment 176-191 in this context could theoretically produce excessive fat mobilization and elevated free fatty acid levels, potentially worsening cardiac rhythm disturbances that are already a concern in hyperthyroidism. Fragment 176-191 should not be used in uncontrolled hyperthyroid states.
Caffeine and Methylxanthines
Caffeine and related methylxanthines (theophylline, theobromine) inhibit phosphodiesterases, the enzymes that degrade cAMP. Since Fragment 176-191 works by elevating cAMP in adipocytes, caffeine could theoretically amplify its lipolytic effect by preventing cAMP breakdown. This is the same pharmacological rationale behind the popular (though modestly effective) combination of caffeine with fat-burning supplements.
A cup of coffee (approximately 100 mg caffeine) consumed alongside Fragment 176-191 morning dosing could provide a modest complementary effect on fat mobilization. However, caffeine also stimulates insulin secretion at higher doses (above 300-400 mg), which would counteract Fragment 176-191's mechanism. The optimal caffeine dose for combined effect without insulin stimulation appears to be in the 100-200 mg range, roughly equivalent to 1-2 cups of black coffee. Adding sugar or cream to coffee would defeat the purpose by stimulating insulin release. Patients interested in the caffeine combined effect should consume black coffee 15-30 minutes before Fragment 176-191 administration.
Metformin
Metformin is potentially the most favorable combination partner for Fragment 176-191 among conventional medications. Metformin activates AMPK (the same kinase that Fragment 176-191 engages for its anti-lipogenic effect), reduces hepatic glucose output (lowering fasting insulin levels), and improves insulin sensitivity (reducing the insulin-mediated antagonism of lipolysis). Every one of these effects complements Fragment 176-191's mechanism.
Patients taking metformin who add Fragment 176-191 may experience enhanced fat loss compared to either agent alone. Metformin's insulin-lowering effect widens the "lipolytic window" during which Fragment 176-191 can effectively mobilize fat. Metformin's AMPK activation enhances Fragment 176-191's anti-lipogenic arm. And metformin's own modest weight loss effect (typically 2-3% of body weight) adds to Fragment 176-191's fat-specific reduction. This combination has not been studied in clinical trials but has strong pharmacological rationale. The biohacking hub covers emerging metabolic optimization strategies that combine pharmaceutical and peptide approaches.
Alcohol
Alcohol creates several problems for Fragment 176-191 efficacy. Ethanol is metabolized preferentially by the liver, suppressing fat oxidation by up to 73% for the duration of alcohol metabolism (typically 1 hour per standard drink). Even if Fragment 176-191 successfully mobilizes fatty acids from adipose tissue, those fatty acids can't be oxidized while the liver is processing alcohol. Instead, they circulate and are eventually re-esterified into triglycerides, negating the lipolytic benefit.
Additionally, alcohol consumption typically involves caloric intake (7 kcal/gram of ethanol plus mixers) and often triggers increased food consumption. The combination of suppressed fat oxidation, additional calories, and increased food intake makes alcohol a triple antagonist to Fragment 176-191's fat-loss mechanism. Patients serious about maximizing Fragment 176-191's effect should limit alcohol to 1-2 standard drinks per week and avoid consuming alcohol within 12 hours of Fragment 176-191 administration. Complete alcohol avoidance during Fragment 176-191 cycles produces the best results.
Reconstitution, Storage, and Quality Assessment
Fragment 176-191's effectiveness depends not just on its pharmacological properties but on the practical handling steps that determine whether the peptide reaching the patient's subcutaneous tissue is actually intact and biologically active. Peptides are fragile molecules, and improper reconstitution, storage, or sourcing can result in degraded product that produces little or no clinical effect. This section covers the practical chemistry and quality considerations that separate effective Fragment 176-191 use from expensive placebo injection.
Understanding Peptide Stability
Fragment 176-191 consists of 16 amino acids in a linear chain. Unlike full-length growth hormone, which has a complex three-dimensional structure stabilized by two disulfide bonds, Fragment 176-191 has no disulfide bonds and minimal tertiary structure. This gives it both advantages and disadvantages for stability. The advantage is that it doesn't have a complex fold that can be irreversibly disrupted by heat or agitation. The disadvantage is that the peptide bond between each amino acid is susceptible to hydrolysis (cleavage by water), and the lack of structural constraints means the molecule has more exposed surface area for degradation reactions.
The primary degradation pathways for Fragment 176-191 include hydrolysis of peptide bonds (especially at asparagine and aspartate residues), deamidation of glutamine and asparagine side chains, and oxidation of the methionine residue at position 170 in the native GH numbering. Deamidation is particularly relevant because it converts asparagine to aspartate (or iso-aspartate), changing the charge and potentially the biological activity of the peptide. Oxidation of methionine to methionine sulfoxide similarly reduces activity. Both reactions are accelerated by heat, alkaline pH, and the presence of oxidizing agents.
In lyophilized (freeze-dried) form, Fragment 176-191 is relatively stable. Properly prepared lyophilized powder can retain greater than 95% potency for 2-3 years when stored at -20C, 12-18 months at 2-8C (refrigerator), and 3-6 months at room temperature (20-25C). Once reconstituted in solution, stability drops dramatically. In bacteriostatic water at 2-8C, Fragment 176-191 retains approximately 90% potency for 3-4 weeks, 70-80% potency at 6 weeks, and may fall below 50% potency by 8 weeks. These estimates are based on general peptide stability data because Fragment 176-191-specific stability studies are not publicly available.
Reconstitution Best Practices
Proper reconstitution starts with the right solvent. Bacteriostatic water (sterile water containing 0.9% benzyl alcohol as a preservative) is the standard choice for multi-dose vials because the benzyl alcohol prevents bacterial growth during the 3-4 week use period. Sterile water without preservative can be used for single-dose preparations but must be discarded after one use to avoid bacterial contamination.
The reconstitution process matters more than most users realize. Fragment 176-191 typically comes as a lyophilized powder or "cake" in a sealed vial. The correct technique involves drawing the appropriate volume of bacteriostatic water into a syringe, inserting the needle through the vial stopper, and directing the water stream against the glass wall of the vial (not directly onto the powder). The water should be added slowly, allowing it to flow down the glass and gently hydrate the powder from below. Direct injection of water onto the powder can create foam, which denatures peptides at the air-liquid interface, and can break the powder into chunks that dissolve unevenly.
After adding all the water, the vial should be swirled gently in a circular motion - never shaken. Shaking creates bubbles and foam that damage peptides through surface denaturation (the same mechanism that causes egg whites to stiffen when whipped). The peptide should dissolve completely within 2-5 minutes of gentle swirling. If chunks or particles remain after 10 minutes, the peptide may have been degraded before reconstitution or may require a different solvent pH. A clear, colorless solution indicates successful reconstitution. Any cloudiness, particulate matter, or discoloration suggests degradation or contamination, and the vial should be discarded.
The reconstitution volume determines the concentration and the precision of dosing. A common approach for a 5 mg vial is to add 2.5 mL of bacteriostatic water, creating a 2 mg/mL (2,000 mcg/mL) solution. Each 0.125 mL (12.5 units on an insulin syringe) then delivers 250 mcg, and 0.25 mL (25 units) delivers 500 mcg. Using a smaller reconstitution volume (e.g., 1 mL for 5 mg/mL) requires very small injection volumes that are harder to measure accurately with standard insulin syringes. Using a larger volume (e.g., 5 mL for 1 mg/mL) makes measuring easier but results in a more dilute solution that may degrade faster and takes longer to use up.
Storage After Reconstitution
Once reconstituted, Fragment 176-191 must be refrigerated at 2-8C (standard refrigerator temperature). The vial should be stored upright in a location that maintains consistent temperature - avoid the door shelf (which experiences temperature fluctuations with each opening) and any location near the freezer compartment. A dedicated shelf in the main body of the refrigerator is ideal.
Never freeze reconstituted Fragment 176-191. Freezing causes ice crystal formation that can physically disrupt the peptide and the solvent matrix, and the freeze-thaw cycle is one of the most damaging physical processes for peptide solutions. If you won't use the reconstituted vial within 3-4 weeks, it's better to reconstitute a smaller amount from a lyophilized vial and keep the remaining powder in its more stable lyophilized form.
Light exposure accelerates peptide degradation through photo-oxidation reactions. Store reconstituted vials away from direct light - wrapping the vial in aluminum foil provides simple but effective protection. Some practitioners store reconstituted peptides in amber glass vials that filter UV light, though this requires a sterile transfer from the original vial that introduces contamination risk unless done in a clean environment.
Assessing Product Quality
The peptide market includes products of widely varying quality, from pharmaceutical-grade material meeting USP standards to crudely synthesized product containing significant impurities and degradation products. Because Fragment 176-191 is typically obtained as a research chemical rather than a prescription medication, quality assessment falls largely on the consumer and provider.
Third-party testing is the gold standard for quality verification. Certificates of Analysis (COAs) from the manufacturer should be supplemented by independent testing from services that perform HPLC (High-Performance Liquid Chromatography) purity analysis and mass spectrometry identity confirmation. HPLC purity of 98% or greater is considered pharmaceutical-grade; 95-97% is acceptable for research purposes; below 95% suggests manufacturing issues or degradation. Mass spectrometry confirms that the molecular weight matches Fragment 176-191's theoretical mass of 1817.12 Da, ruling out substitution with a different peptide entirely.
Visual inspection provides basic but useful quality cues. Lyophilized Fragment 176-191 should appear as a white to off-white powder or solid "cake" that occupies a reasonable portion of the vial (not a tiny speck suggesting under-filling). Yellow or brown discoloration suggests oxidation or thermal degradation. Wet or sticky powder suggests moisture ingress during storage. Reconstituted solution should be perfectly clear and colorless; any cloudiness, particles, or color suggests contamination or aggregation.
Providers who source from compounding pharmacies (like FormBlends) benefit from the quality systems required of licensed pharmacies, including cGMP manufacturing, potency testing of each batch, sterility testing, endotoxin testing, and regulatory oversight by state boards of pharmacy and potentially the FDA. These quality assurances are typically absent from research chemical suppliers, making pharmacy-sourced peptides the preferred option for clinical use despite their higher cost.
Injection Technique and Site Selection
Subcutaneous injection of Fragment 176-191 is straightforward but benefits from attention to technique. Using a 29-31 gauge insulin syringe (the same syringes used for insulin injection), the needle is inserted at a 45-90 degree angle into a pinch of subcutaneous fat. The injection should be slow and steady - rushing the injection increases tissue trauma and can cause more discomfort. After injecting, hold the needle in place for 5-10 seconds before withdrawing to prevent leakage of the peptide solution from the injection site.
Preferred injection sites include the abdomen (at least 2 inches from the navel), the anterior thigh, and the posterior upper arm. The abdomen is the most commonly used site because subcutaneous fat is usually readily accessible there, absorption is consistent, and it's easy to reach for self-injection. Some practitioners advocate for injection into the area of desired fat loss (e.g., abdominal subcutaneous injection for abdominal fat reduction), but there's no evidence that local injection produces preferential local fat loss. Fragment 176-191 is absorbed into systemic circulation and acts on adipose tissue throughout the body regardless of injection site.
Rotating injection sites is important for two reasons: preventing lipodystrophy (changes in subcutaneous fat at repeatedly used sites) and maintaining consistent absorption. Using the same site repeatedly can cause local tissue changes that alter absorption kinetics and create cosmetically undesirable dimpling or hardening. A simple rotation scheme - left abdomen Monday/Wednesday/Friday, right abdomen Tuesday/Thursday/Saturday - distributes injection stress across multiple sites and allows adequate recovery time between uses of each site.
Monitoring Protocols and Tracking Outcomes
Effective use of Fragment 176-191 requires systematic monitoring to assess efficacy, detect potential adverse effects, and optimize the protocol over time. Because Fragment 176-191 produces relatively subtle body composition changes (fat-specific loss without dramatic weight changes), crude measures like scale weight are inadequate. A comprehensive monitoring approach uses multiple metrics across different timeframes to build a complete picture of response.
Baseline Assessment Before Starting
Before initiating Fragment 176-191, a thorough baseline assessment establishes the starting point against which all future changes will be measured. This baseline should include body composition measurement (DEXA scan is the gold standard, providing separate measurements of fat mass, lean mass, and bone mineral density with approximately 1-2% precision), anthropometric measurements (waist circumference, hip circumference, waist-to-hip ratio, and skinfold measurements at 7 standard sites), metabolic blood work (fasting glucose, fasting insulin, HbA1c, comprehensive metabolic panel, lipid panel, thyroid panel including TSH and free T3/T4), hormonal assessment (IGF-1, testosterone in men, estradiol in women, DHEA-S, morning cortisol), and a baseline clinical photograph series (front, side, and back views under standardized lighting conditions).
The metabolic blood work serves dual purposes: it identifies conditions that might reduce Fragment 176-191's efficacy (hypothyroidism, insulin resistance, hormonal imbalances) and provides baseline values for safety monitoring during treatment. If baseline thyroid function is suboptimal (TSH above 3.0 mIU/L or free T3 in the lower third of the reference range), addressing this before starting Fragment 176-191 will likely improve response. If baseline fasting insulin is elevated (above 12 mIU/mL), adding insulin-sensitizing interventions (metformin, exercise, dietary modification) before or concurrent with Fragment 176-191 will enhance the lipolytic environment.
Weekly Monitoring During Treatment
During Fragment 176-191 treatment, weekly self-monitoring provides the data needed to assess progress and adjust the protocol. Key weekly metrics include morning fasted body weight (same conditions each time: after waking, after voiding, before eating or drinking, wearing the same minimal clothing), waist circumference measured at the narrowest point of the torso (typically at the navel level), and subjective energy and hunger assessments on a 1-10 scale.
Waist circumference is the single most useful weekly metric for Fragment 176-191 because it correlates more strongly with visceral fat loss than scale weight. A patient losing visceral fat may show a 2-4 cm decrease in waist circumference over 8 weeks while body weight changes by less than 1 kg. If the scale is flat but the waist is shrinking, Fragment 176-191 is working as intended. If both are flat after 4 weeks of consistent use with proper timing (fasted administration), the protocol should be reassessed for dosing adequacy, timing compliance, and potential interfering factors (medications, thyroid function, alcohol consumption).
Monthly Laboratory Monitoring
Monthly blood work during Fragment 176-191 treatment provides safety surveillance and metabolic optimization data. The core monthly panel should include fasting glucose and insulin (to confirm the absence of insulin disruption and to monitor the lipolytic environment), liver function tests (AST, ALT) to detect any hepatic stress from increased free fatty acid processing, and lipid panel to track changes in triglycerides (which typically decrease with effective fat mobilization) and HDL/LDL distribution.
IGF-1 should be measured at the 1-month mark to confirm the expected non-elevation. If IGF-1 rises more than 20% above baseline, the product may be contaminated with full-length GH or another GH-stimulating compound, and should be discontinued and investigated. A stable IGF-1 level provides reassurance that the peptide is acting through its selective lipolytic mechanism without engaging the growth-promoting pathway.
For patients concerned about cancer risk (which is low with Fragment 176-191 due to the absence of IGF-1 elevation but merits monitoring in any peptide protocol), adding a free fatty acid level to the monthly panel provides information about whether mobilized fat is being effectively oxidized or accumulating in the circulation. Persistently elevated free fatty acids (above 0.7 mEq/L) in the fasting state may indicate that the body's oxidative capacity is being overwhelmed, which could contribute to hepatic steatosis and should prompt evaluation of thyroid function and aerobic exercise adequacy.
Body Composition Reassessment
DEXA scans should be repeated at 8-12 week intervals during Fragment 176-191 treatment. More frequent scanning isn't useful because the changes are small enough that measurement precision limits the ability to detect them over shorter intervals. At the 8-12 week mark, a responder to Fragment 176-191 should show a decrease in total fat mass of 1.5-3.0 kg, a stable or minimally changed lean mass (within 0.5 kg of baseline), and a preferential decrease in trunk fat percentage relative to appendicular fat percentage (reflecting visceral fat mobilization).
For patients who don't have access to DEXA scanning, a combination of waist circumference trends, skinfold measurements (requiring a trained assessor for accuracy), and bioelectrical impedance analysis (BIA, available as consumer scales from Withings, InBody, and others) can provide approximate body composition tracking. BIA is less accurate than DEXA for absolute values but can track trends reasonably well when measurements are taken under standardized conditions (same time of day, same hydration status, same pre-measurement fasting period).
Progress photography is an underutilized but valuable monitoring tool. Standardized photos taken every 2-4 weeks under identical conditions (same lighting, same camera angle, same time of day, same clothing) reveal visual changes that neither the scale nor waist measurements capture. Many patients are surprised to see visible changes in body composition in their photos despite minimal scale movement. The key to useful progress photography is consistency: use the same location, same lighting (front-facing natural light or consistent artificial lighting), and same camera position for every session. Front, side, and back views provide a complete picture. Comparing photos from week 1 to week 8 often reveals body composition changes that the patient didn't notice day to day because the gradual shifts are invisible in the mirror's daily reflection.
Some patients find it helpful to track multiple subjective metrics alongside objective measurements. Energy levels throughout the day (rated 1-10 at morning, afternoon, and evening), sleep quality (rated 1-10 upon waking), exercise performance (weights lifted, running pace, recovery time), and clothing fit provide a rich dataset that contextualizes the objective body composition data. A patient whose waist measurement is decreasing, energy is improving, and training weights are stable or increasing is clearly responding well to the Fragment 176-191 protocol, even if scale weight hasn't changed significantly. This multi-metric approach prevents the discouragement that comes from fixating on a single number and helps patients and providers make more informed protocol decisions.
Endpoint Assessment and Decision Points
At the end of a planned Fragment 176-191 cycle (typically 8-12 weeks), a comprehensive assessment determines the next steps. Key questions include whether fat loss goals were met (if not, was the protocol optimized? Would another cycle be beneficial?), whether metabolic parameters improved (fasting glucose, insulin sensitivity, lipid profile), whether any adverse effects occurred that would contraindicate continuation, and whether the cost-benefit ratio justifies continued therapy.
Patients who achieved their fat loss goals should transition to a maintenance protocol that may include continued but reduced-dose Fragment 176-191 (250 mcg daily rather than 500 mcg twice daily), lifestyle optimization (exercise, nutrition, sleep) to maintain the achieved body composition, and periodic reassessment (every 3-6 months) to detect any fat regain that might warrant another treatment cycle. The lifestyle hub provides evidence-based strategies for maintaining body composition results after completing peptide therapy cycles.
Non-responders (less than 1 kg fat loss despite 8+ weeks of compliant use) should be evaluated for interfering factors including suboptimal thyroid function, persistent hyperinsulinemia, medication interactions (beta-blockers, corticosteroids), product quality concerns, and protocol compliance issues (administering in a fed state, improper storage). Addressing identified factors and attempting a second cycle with the improved protocol is reasonable before concluding that Fragment 176-191 is ineffective for that individual. Some patients are genuine non-responders due to genetic variation in beta-3 adrenergic receptor expression or HSL activity, and these individuals may respond better to alternative approaches such as semaglutide or tesofensine that work through different mechanisms.
Legal and Regulatory Landscape
Fragment 176-191 exists in a complex regulatory space that varies by jurisdiction and is evolving rapidly. Understanding the current legal status, the regulatory frameworks that apply, and the trends shaping future regulation helps patients and practitioners make informed decisions about access and use.
Regulatory Status in the United States
In the United States, Fragment 176-191 is not FDA-approved for any indication. It has not completed the approval process, and no pharmaceutical company currently holds an approved New Drug Application (NDA) or Biologics License Application (BLA) for this peptide. However, not being FDA-approved is different from being illegal. Fragment 176-191 can be legally sold as a research chemical for in vitro and laboratory use, and it can be prescribed by licensed physicians as a compounded medication under the authority of state medical practice acts and federal compounding regulations.
The compounding pathway is the primary legal mechanism through which patients access Fragment 176-191 in the US. Under Section 503A of the Federal Food, Drug, and Cosmetic Act, licensed pharmacies can compound medications containing bulk drug substances (including peptides like Fragment 176-191) when a valid patient-specific prescription exists, the compounding is performed by a licensed pharmacist, and the bulk drug substance meets compendial standards or is accompanied by a valid Certificate of Analysis. Section 503B outsourcing facilities can compound without patient-specific prescriptions but must register with the FDA and comply with current Good Manufacturing Practice (cGMP) requirements.
The regulatory environment for compounded peptides has tightened significantly since 2023, when the FDA began taking a more active role in overseeing compounding pharmacies, particularly those producing GLP-1 receptor agonists like semaglutide. While Fragment 176-191 hasn't been a specific target of FDA enforcement actions, the broader regulatory attention to compounded peptides affects the supply chain and availability of all compounded peptide products. The GLP-1 research hub tracks regulatory developments in the compounded peptide space that may affect Fragment 176-191 availability.
International Regulatory Variations
Australia holds particular significance for Fragment 176-191 because the peptide was developed by Monash University and Australian-based Metabolic Pharmaceuticals. Despite this local origin, Fragment 176-191 is not TGA-approved in Australia. AOD-9604, the stabilized variant, received Generally Recognized As Safe (GRAS) status from the US FDA in 2014 for use as a food ingredient at specific doses, but this is distinct from drug approval and doesn't authorize its use as an injectable therapeutic agent.
In the European Union, Fragment 176-191 falls under the regulatory framework for unauthorized medicinal products. EU member states have varying provisions for compounding and off-label use, but generally, peptides that haven't received European Medicines Agency (EMA) marketing authorization cannot be commercially promoted or widely distributed. Access for individual patients typically requires a physician's prescription under the "named patient" or "compassionate use" provisions that exist in most EU countries.
Canada classifies Fragment 176-191 as a prescription drug substance that can only be legally obtained through a licensed pharmacy with a valid prescription. Canadian compounding pharmacies can prepare Fragment 176-191 formulations under similar frameworks to those in the US, though Health Canada's oversight of compounding practices differs in specific details.
Anti-Doping Considerations
For athletes subject to anti-doping regulation, Fragment 176-191's status is ambiguous and potentially risky. The World Anti-Doping Agency (WADA) Prohibited List includes "growth hormone releasing factors and their analogs" and "growth hormone fragments" as Category S2 (Hormones and Metabolic Modulators) prohibited substances. While Fragment 176-191 doesn't release growth hormone and doesn't activate the growth hormone receptor in the classical sense, its structural derivation from growth hormone places it in a gray zone that could be interpreted as falling under the "growth hormone fragments" prohibition.
Athletes who test positive for Fragment 176-191 or its metabolites could face sanctions regardless of whether the substance was explicitly listed by name. WADA operates on strict liability principles - the athlete is responsible for any prohibited substance found in their system, even if they believed it was permitted. Given this risk, any athlete subject to drug testing should avoid Fragment 176-191 entirely unless they have obtained a specific written determination from their sport's anti-doping authority confirming its permissibility.
Sourcing and Quality Considerations
The unregulated nature of the research peptide market creates significant quality risks. Studies analyzing commercial research peptides have found that 10-15% of products contain the wrong compound entirely, 20-30% have purity below stated levels, and some contain potentially harmful contaminants including bacterial endotoxins, heavy metals, and residual organic solvents from the synthesis process. These quality risks are mitigated by sourcing from licensed compounding pharmacies that operate under regulatory oversight and quality management systems.
Patients considering Fragment 176-191 should work with a licensed healthcare provider who can prescribe the compound from a reputable compounding pharmacy, rather than purchasing from unregulated online sources. While the cost is typically higher through legitimate channels, the quality assurance, medical supervision, and legal protection justify the premium. The FormBlends GLP-1 program page and science page provide information about pharmacy-sourced peptide options with appropriate quality standards and clinical oversight.
Nutritional Support and Lifestyle Optimization for Fragment 176-191 Users
Fragment 176-191 doesn't work in a vacuum. Its fat-mobilizing mechanism produces a cascade of metabolic events that the body must then process, and the efficiency of that processing depends heavily on nutritional status, exercise patterns, sleep quality, and stress management. Optimizing these lifestyle factors doesn't just "support" Fragment 176-191's effects; in many cases, they determine whether the peptide produces meaningful results or just mobilizes fatty acids that get re-stored because the body's oxidative capacity can't keep up.
Protein Requirements During Fat Loss
Protein intake during any fat-loss protocol serves dual purposes: maintaining lean mass and providing amino acid substrates for the metabolic machinery that processes mobilized fat. Fragment 176-191 doesn't directly affect lean mass, but the caloric deficit that many users maintain alongside the peptide does create a catabolic environment where insufficient protein intake can lead to muscle breakdown.
Research consistently shows that protein intake of 1.6-2.2 grams per kilogram of body weight per day is optimal for lean mass preservation during caloric deficit. For a 90 kg individual, this translates to 144-198 grams of protein daily, distributed across 4-5 meals to maximize muscle protein synthesis signaling through the mTOR pathway. Each meal should contain at least 30-40 grams of high-quality protein (containing adequate leucine, the key amino acid that triggers mTOR activation) to cross the threshold for muscle protein synthesis stimulation.
The timing of protein intake relative to Fragment 176-191 dosing matters. Since Fragment 176-191 works best in a fasted state (low insulin), and protein consumption raises insulin modestly, there's a practical tension between optimal Fragment 176-191 timing and optimal protein distribution. The solution is to front-load protein consumption into the eating window: if using a 16:8 intermittent fasting protocol with Fragment 176-191 administered at 7 AM, break the fast at 11 AM or noon with a high-protein meal (40-50 grams), consume another protein-rich meal in the mid-afternoon, and a final protein serving at dinner. This preserves the fasted window for Fragment 176-191 activity while ensuring adequate daily protein intake. For patients managing complex nutritional protocols alongside peptide therapy, the lifestyle hub provides evidence-based dietary frameworks compatible with various peptide protocols.
Micronutrient Considerations
Several micronutrients play direct roles in the metabolic pathways that Fragment 176-191 engages. Carnitine is required for the transport of long-chain fatty acids into mitochondria for beta-oxidation. When Fragment 176-191 mobilizes fatty acids from adipose tissue, those fatty acids must be transported into muscle and liver mitochondria via the carnitine shuttle to be oxidized. Individuals with low carnitine status (common in vegetarians, elderly, and those with certain genetic polymorphisms) may experience a bottleneck at this step, where mobilized fatty acids accumulate in the cytoplasm rather than being burned. L-carnitine supplementation at 1-2 grams daily can support this pathway, though clinical data specifically combining carnitine with Fragment 176-191 don't exist.
Coenzyme Q10 (CoQ10) and B vitamins (particularly B2, B3, and B5) are cofactors in the electron transport chain and fatty acid oxidation pathways. Adequate status in these nutrients ensures that the mitochondrial machinery can process the fatty acids that Fragment 176-191 mobilizes. Statin medications, which are common in the metabolic syndrome population most likely to use Fragment 176-191, deplete CoQ10 by inhibiting the mevalonate pathway. Patients on statins who are adding Fragment 176-191 should consider CoQ10 supplementation at 100-200 mg daily to support mitochondrial function.
Magnesium deserves special mention because it's involved in over 300 enzymatic reactions including those in the cAMP signaling cascade that Fragment 176-191 activates. Approximately 50% of US adults consume less than the recommended dietary allowance for magnesium, and deficiency is more prevalent in individuals with metabolic syndrome (magnesium is depleted by insulin resistance and hyperinsulinemia). Ensuring adequate magnesium intake through diet (dark leafy greens, nuts, seeds, whole grains) or supplementation (200-400 mg of magnesium glycinate or threonate daily) supports the signaling pathways through which Fragment 176-191 exerts its effects.
Exercise Programming for Maximum Combined effect
Exercise interacts with Fragment 176-191 at multiple levels. Aerobic exercise increases mitochondrial density and fatty acid oxidation capacity, expanding the body's ability to process the fatty acids that Fragment 176-191 mobilizes. Resistance training preserves lean mass during caloric deficit and maintains the metabolically active tissue that drives resting energy expenditure. The combination of both exercise modalities with Fragment 176-191 creates complementary effects that exceed what any single intervention achieves alone.
For aerobic exercise, moderate-intensity steady-state (MISS) activity at 60-70% of maximum heart rate (the "fat-burning zone") predominantly oxidizes fatty acids as fuel. Performing 30-45 minutes of MISS exercise after Fragment 176-191 morning administration takes advantage of the already-mobilized fatty acids and the fasted state to maximize fat oxidation. Higher-intensity exercise shifts fuel utilization toward glucose, which is less complementary with Fragment 176-191's mechanism during the exercise session itself (though high-intensity exercise elevates post-exercise fat oxidation through EPOC, "excess post-exercise oxygen consumption"). A practical approach combines 2-3 MISS sessions per week (timed with Fragment 176-191 dosing) with 1-2 high-intensity interval training (HIIT) sessions for cardiovascular fitness and EPOC benefits. Walking, which many underestimate, deserves recognition here. Brisk walking at 3.5-4.0 mph falls squarely in the fat-oxidation zone and can be sustained for longer durations than running or cycling, accumulating meaningful caloric expenditure with minimal recovery cost, negligible injury risk, no gym membership required, and easy integration into daily routines.
Resistance training should be programmed 3-4 times per week with progressive overload targeting all major muscle groups. The anabolic stimulus from resistance exercise creates a "nutrient partitioning" effect where consumed calories are preferentially directed toward muscle repair and growth rather than fat storage. This effect complements Fragment 176-191's fat mobilization by ensuring that the energy balance favors lean tissue maintenance even during caloric deficit. For patients who are new to resistance training or returning after a long break, starting with a simple full-body program using compound movements (squats, deadlifts, bench press, rows, overhead press) 3 times per week provides an adequate stimulus without excessive recovery demands.
Sleep and Circadian Rhythm Optimization
Sleep quality profoundly affects fat metabolism, hormone profiles, and Fragment 176-191's efficacy. During deep sleep (NREM stages 3-4), growth hormone release peaks, cortisol reaches its nadir, and insulin sensitivity is at its highest. Sleep deprivation (less than 6 hours) increases cortisol by 37-45%, reduces insulin sensitivity by 25-30%, elevates appetite-stimulating ghrelin levels, and impairs the hormonal environment that supports Fragment 176-191's mechanism.
For Fragment 176-191 users, optimizing sleep means prioritizing 7-9 hours per night, maintaining consistent sleep and wake times (even on weekends), minimizing blue light exposure in the 2 hours before bed (which suppresses melatonin and delays circadian rhythm), keeping the bedroom cool (65-68 degrees Fahrenheit supports deeper sleep), and avoiding caffeine after 2 PM (while morning caffeine synergizes with Fragment 176-191, afternoon caffeine disrupts sleep quality). Some patients find that peptides like DSIP (Delta Sleep-Inducing Peptide) or Selank support sleep quality alongside their Fragment 176-191 protocol, though these combinations lack formal study.
Stress Management and Cortisol Control
Chronic psychological stress elevates cortisol throughout the day, disrupting the natural diurnal cortisol rhythm that supports Fragment 176-191's morning fat-mobilizing action. Chronic cortisol elevation promotes visceral fat storage (directly opposing Fragment 176-191's mechanism), increases insulin resistance (reducing the low-insulin window needed for lipolysis), promotes cravings for high-calorie comfort foods (undermining caloric deficit efforts), and impairs sleep quality (creating a vicious cycle with the sleep effects described above).
While eliminating all stress isn't realistic, evidence-based stress reduction practices can meaningfully lower cortisol levels and improve the metabolic environment for Fragment 176-191. Regular mindfulness meditation (even 10-15 minutes daily) has been shown to reduce salivary cortisol by 13-20% in multiple controlled studies. Regular exercise itself reduces cortisol reactivity over time. Time in nature (the "green exercise" effect) lowers cortisol more effectively than equivalent indoor exercise. Social connection and supportive relationships buffer cortisol responses to stressors. And adaptogenic compounds like ashwagandha (300-600 mg daily of standardized extract) have demonstrated cortisol-lowering effects in randomized controlled trials, though their interaction with Fragment 176-191 specifically hasn't been studied.
Patients who notice that their Fragment 176-191 results plateau during stressful periods may benefit from intensifying stress management practices before increasing their peptide dose. In many cases, the peptide isn't the limiting factor; the hormonal environment created by chronic stress is preventing the peptide from working at its full capacity. The dosing calculator can help patients and providers explore whether dose adjustment or lifestyle optimization is the more appropriate response to plateauing results.
Hydration and Electrolyte Balance
Adequate hydration supports Fragment 176-191 therapy in ways that many users overlook. Water is a direct participant in lipolysis - the hydrolysis of triglycerides into glycerol and free fatty acids is literally a water-mediated reaction. Dehydration reduces blood flow to adipose tissue (limiting Fragment 176-191 delivery), impairs kidney function (reducing metabolic waste clearance from fat breakdown), and thickens blood viscosity (reducing tissue oxygen delivery needed for fatty acid oxidation in mitochondria).
Daily water intake should target 35-40 mL per kilogram of body weight, with additional intake to replace exercise-related losses (approximately 500-750 mL per hour of moderate exercise). For a 90 kg individual, this means approximately 3.2-3.6 liters of water daily as a baseline. Spreading intake throughout the day is more effective than consuming large volumes at once, as the kidneys can only process approximately 800 mL to 1 liter per hour.
Electrolyte balance becomes particularly relevant for Fragment 176-191 users who combine the peptide with intermittent fasting. During fasting, insulin levels drop, which causes the kidneys to excrete more sodium (insulin promotes sodium retention). The resulting sodium loss pulls water and potassium with it, creating a mild electrolyte depletion that manifests as fatigue, headache, and dizziness during the fasting window. Supplementing with 1-2 grams of sodium (from salt or electrolyte supplements), 200-400 mg of potassium, and 200 mg of magnesium during the fasting window prevents these symptoms without breaking the fast or elevating insulin.
Gut Health and Absorption Optimization
While Fragment 176-191 is administered subcutaneously (bypassing the GI tract entirely), gut health still affects overall metabolic function in ways relevant to fat loss outcomes. The gut microbiome influences energy extraction from food (some bacterial populations extract more calories from the same food than others), bile acid metabolism (which affects fat absorption and cholesterol processing), short-chain fatty acid production (which modulates adipocyte metabolism), and systemic inflammation (which impairs insulin sensitivity and fat oxidation).
Patients on Fragment 176-191 who have concurrent gut dysfunction (bloating, irregular bowel movements, food sensitivities) may benefit from addressing these issues to optimize the metabolic environment. A diet rich in prebiotic fiber (from vegetables, legumes, and whole grains) supports beneficial bacterial populations. Fermented foods (yogurt, kefir, sauerkraut, kimchi) provide probiotic organisms. And gut-healing peptides like BPC-157 have demonstrated protective effects on GI mucosal integrity in preclinical studies, potentially supporting overall gut health during intensive fat-loss protocols.
The gut-adipose tissue axis is an active area of research. Bacterial metabolites produced in the colon circulate systemically and directly affect adipocyte metabolism. Short-chain fatty acids (acetate, propionate, butyrate) produced by bacterial fermentation of fiber activate GPR41 and GPR43 receptors on adipocytes, influencing lipolysis and lipogenesis. While the interaction between these gut-derived signals and Fragment 176-191's mechanism hasn't been studied specifically, maintaining a healthy, diverse microbiome likely creates a more favorable metabolic context for any fat-loss intervention.
Temperature and Cold Exposure
Cold exposure activates brown adipose tissue (BAT) and promotes "browning" of white adipose tissue (the conversion of energy-storing white fat cells into energy-burning beige fat cells). This process, mediated by UCP1 (uncoupling protein 1) expression, dissipates stored energy as heat rather than storing it as fat. Cold exposure and Fragment 176-191 may be complementary: Fragment 176-191 mobilizes fatty acids from white adipose tissue, while cold-activated brown and beige fat provides an additional pathway for burning those fatty acids through non-shivering thermogenesis.
Practical cold exposure approaches include cold showers (starting with 30 seconds of cold water at the end of a regular shower and gradually extending to 2-3 minutes), cold water immersion (10-15 minutes at 50-59 degrees Fahrenheit, 2-3 times per week), and simply reducing indoor temperature by 3-5 degrees during sleeping hours (which activates mild thermogenic responses without the discomfort of direct cold exposure). Research from human studies suggests that regular cold exposure over 4-6 weeks can increase BAT volume and activity by 30-45%, creating a meaningful additional caloric expenditure pathway of 100-200 calories per day.
The combination of Fragment 176-191 with regular cold exposure creates a complementary fat-loss system: the peptide mobilizes fatty acids from storage, while cold-activated thermogenesis provides a destination for those fatty acids beyond standard mitochondrial beta-oxidation. The peptide MOTS-c, which enhances mitochondrial function and has been shown to activate thermogenic pathways, represents another potential combined effect with Fragment 176-191's fat-mobilizing mechanism, though combination studies haven't been conducted. For patients interested in the intersection of peptide therapy and metabolic optimization strategies, the biohacking hub explores these emerging approaches in greater detail.
Frequently Asked Questions
What is HGH Fragment 176-191?
HGH Fragment 176-191 is a synthetic peptide consisting of the last 16 amino acids (positions 176 through 191) of the human growth hormone molecule. This specific region of growth hormone is responsible for the hormone's fat-metabolizing activity. When isolated as a standalone peptide, Fragment 176-191 retains the ability to stimulate lipolysis (fat breakdown) and inhibit lipogenesis (new fat storage) without producing the growth-promoting, IGF-1-stimulating, or diabetogenic effects associated with full-length growth hormone. It was first characterized by Wu et al. at Monash University in 1993 and subsequently developed into AOD-9604 by Metabolic Pharmaceuticals for clinical evaluation.
How is Fragment 176-191 different from AOD-9604?
AOD-9604 is a modified version of Fragment 176-191 with one structural change: an additional tyrosine amino acid attached to the N-terminus (the beginning of the peptide chain). This single amino acid addition gives AOD-9604 improved stability in biological fluids, enhanced resistance to enzymatic degradation, and confirmed oral bioavailability. The two compounds share the same core mechanism of action, acting through beta-3 adrenergic receptors to stimulate fat metabolism. The key practical difference is that all six human clinical trials (893 subjects total) were conducted with AOD-9604, not the unmodified Fragment 176-191. AOD-9604 also received FDA GRAS designation, while Fragment 176-191 remains classified as a research compound.
Does HGH Fragment 176-191 actually burn fat?
The evidence supports direct lipolytic activity. In vitro studies show Fragment 176-191 increases fat cell glycerol release (a measure of triglyceride breakdown) approximately 4.2-fold over untreated controls. Animal studies by Heffernan et al. (2001) demonstrated that chronic treatment reduced body weight gain by nearly 50% in obese mice, with corresponding increases in fat oxidation confirmed by indirect calorimetry. The METAOD005 human trial of oral AOD-9604 showed 2.8 kg weight loss over 12 weeks at the 1 mg dose, versus 0.8 kg with placebo. However, the human weight loss was modest by modern standards, and the unmodified Fragment 176-191 has not been tested in controlled human trials for fat loss.
What is the recommended dosing for HGH Fragment 176-191?
Research protocols typically use 250 to 500 mcg per subcutaneous injection, administered one to two times daily, for a total daily dose of 250 to 1,000 mcg. The standard approach is 250 mcg twice daily (morning on an empty stomach and evening before bed), totaling 500 mcg per day. Administration in a fasted state is recommended because insulin opposes the lipolytic cascade that Fragment 176-191 activates. Most protocols run for 8-12 weeks with a 4-week break between cycles. The METAOD005 trial showed the lowest oral dose (1 mg/day) produced better weight loss than higher doses, suggesting that more is not necessarily better with this peptide class.
Does Fragment 176-191 affect blood sugar or insulin levels?
No. Across six randomized, placebo-controlled clinical trials involving 893 human subjects, AOD-9604 (the closely related modified fragment) showed no effects on fasting glucose, fasting insulin, insulin sensitivity (HOMA-IR), or oral glucose tolerance test results. This stands in sharp contrast to full-length growth hormone, which commonly causes insulin resistance and hyperglycemia. The Stier et al. (2013) safety analysis reported that subjects with impaired glucose tolerance at baseline actually showed a trend toward improvement during AOD-9604 treatment. The absence of glucose effects is explained by the fragment's inability to bind the growth hormone receptor, the step required for GH's diabetogenic action.
Does Fragment 176-191 increase IGF-1 levels?
No. This is one of the defining safety characteristics of Fragment 176-191 and AOD-9604. Because the fragment does not bind the growth hormone receptor, it cannot activate the JAK2-STAT5 signaling cascade that drives hepatic IGF-1 production. In all six human clinical trials, serum IGF-1 levels remained completely unchanged in subjects receiving AOD-9604 at all doses tested. This is clinically significant because chronically elevated IGF-1 has been associated with increased risk of certain cancers. The absence of IGF-1 stimulation removes what many consider the most concerning aspect of growth hormone therapy.
Can Fragment 176-191 be taken orally?
AOD-9604 (the tyrosine-modified version of Fragment 176-191) has confirmed oral bioavailability in both animal models and human clinical trials. Mass spectrometry studies in pigs showed that orally administered AOD-9604 was absorbed and detectable in plasma, though with rapid degradation kinetics. All six human clinical trials included oral dosing arms, and the METAOD005 Phase IIb trial used exclusively oral administration at doses of 1-30 mg daily. The original unmodified Fragment 176-191 has less established oral bioavailability data, which is why it is typically administered via subcutaneous injection in research settings. The tyrosine modification in AOD-9604 was specifically designed to improve oral stability.
Is Fragment 176-191 legal?
The legal status of Fragment 176-191 varies by jurisdiction. In the United States, it is not FDA-approved as a drug or dietary supplement. AOD-9604 received FDA GRAS (Generally Recognized As Safe) designation for use as a food ingredient, but this is a different regulatory pathway from drug approval. Fragment 176-191 is available through research chemical suppliers and compounding pharmacies in many jurisdictions. In competitive sports, both Fragment 176-191 and AOD-9604 are banned by the World Anti-Doping Agency (WADA) under the category of growth hormone releasing factors and fragments. Always consult local regulations and a healthcare provider before obtaining or using any peptide compound.
How long does it take to see results with Fragment 176-191?
Based on the clinical trial data for AOD-9604 and anecdotal reports from the research community, measurable changes in body composition typically emerge within 4-6 weeks of consistent use at standard doses. In the 12-week METAOD005 trial, weight loss was progressive over the study duration, suggesting that benefits accumulate with continued use. However, individual responses vary considerably based on factors including baseline body composition, caloric balance, physical activity level, and metabolic health. Fragment 176-191 works best as part of a comprehensive approach that includes appropriate caloric intake and regular exercise, not as a standalone intervention.
What are the side effects of Fragment 176-191?
In the six controlled clinical trials of AOD-9604, the adverse event profile was indistinguishable from placebo. No serious adverse events were attributed to the peptide, and no subjects withdrew due to side effects. The most commonly reported events (headache, mild nasopharyngitis) occurred at equal rates in treatment and placebo groups. Anecdotal reports from subcutaneous Fragment 176-191 users include mild injection site reactions (redness, itching), transient headache in the first few days, and occasional lightheadedness when used in a deeply fasted state. These effects are generally mild and self-limiting. No effects on IGF-1, glucose, or insulin have been observed in any study.
References
- Wu Z, Ng FM. Antilipogenic action of synthetic C-terminal sequence 177-191 of human growth hormone. Biochemistry and Molecular Biology International. 1993;30(5):963-971. PMID: 8358331.
- Heffernan MA, Thorburn AW, Fam B, et al. Increase of fat oxidation and weight loss in obese mice caused by chronic treatment with human growth hormone or a modified C-terminal fragment. International Journal of Obesity. 2001;25(10):1442-1449. DOI: 10.1038/sj.ijo.0801740. PMID: 11673763.
- Heffernan MA, Jiang WJ, Thorburn AW, et al. Effects of human GH and its lipolytic fragment (AOD9604) on lipid metabolism following chronic treatment in obese mice and beta(3)-AR knock-out mice. Endocrinology. 2001;142(12):5182-5189. DOI: 10.1210/endo.142.12.8522. PMID: 11713213.
- Stier H, Vos E, Kenley D. Safety and tolerability of the hexadecapeptide AOD9604 in humans. Journal of Endocrinology and Metabolism. 2013;3(1-2):7-15. DOI: 10.4021/jem.v3i1-2.157.
- More MI, Kenley D, Gianello R. Safety and metabolism of AOD9604, a novel nutraceutical ingredient for improved metabolic health. Journal of Endocrinology and Metabolism. 2014;4(3):79-84. DOI: 10.14740/jem213w.
- Ng FM, Sun J, Sharma L, et al. Metabolic studies of a synthetic lipolytic domain (AOD9604) of human growth hormone. Hormone Research. 2000;53(6):274-278. DOI: 10.1159/000053183. PMID: 11146367.
- Jorgensen JO, Moller L, Krag M, et al. Effects of growth hormone on glucose and fat metabolism in human subjects. Endocrinology and Metabolism Clinics of North America. 2007;36(1):75-87. DOI: 10.1016/j.ecl.2006.11.005. PMID: 17336735.
- Berryman DE, Glad CA, List EO, et al. The effects of growth hormone on adipose tissue: old observations, new mechanisms. Growth Hormone and IGF Research. 2020;53-54:101338. DOI: 10.1016/j.ghir.2020.101338. PMID: 32932221.
- Kraemer WJ, Ratamess NA, Nindl BC. Recovery responses of testosterone, growth hormone, and IGF-1 after resistance exercise. Journal of Applied Physiology. 2017;122(3):549-558. DOI: 10.1152/japplphysiol.00599.2016. PMID: 27881670.
- Langendonk JG, Meinders AE, Burggraaf J, et al. Influence of obesity and body fat distribution on growth hormone kinetics in humans. American Journal of Physiology. 1999;277(5):E824-E829. DOI: 10.1152/ajpendo.1999.277.5.E824. PMID: 10567009.
- Richelsen B. Action of growth hormone in adipose tissue. Hormone Research. 1997;48(Suppl 5):105-110. DOI: 10.1159/000191338. PMID: 9434052.
- Holm C. Molecular mechanisms regulating hormone-sensitive lipase and lipolysis. Biochemical Society Transactions. 2003;31(Pt 6):1120-1124. DOI: 10.1042/bst0311120. PMID: 14641008.
- Anthonsen MW, Ronnstrand L, Wernstedt C, et al. Identification of novel phosphorylation sites in hormone-sensitive lipase that are phosphorylated in response to isoproterenol and govern activation properties in vitro. Journal of Biological Chemistry. 1998;273(1):215-221. DOI: 10.1074/jbc.273.1.215. PMID: 9417067.
- Granneman JG, Moore HP, Krishnamoorthy R, et al. Perilipin controls lipolysis by regulating the interactions of AB-hydrolase containing 5 (Abhd5) and adipose triglyceride lipase (Atgl). Journal of Biological Chemistry. 2009;284(50):34538-34544. DOI: 10.1074/jbc.M109.068478. PMID: 19850935.
- Collins S. Beta-adrenergic receptors and adipose tissue metabolism: evolution of an old story. Annual Review of Physiology. 2022;84:1-16. DOI: 10.1146/annurev-physiol-060721-092440. PMID: 34699261.
- de Souza CJ, Burkey BF. Beta-3-adrenoceptor agonists as anti-diabetic and anti-obesity drugs in humans. Current Pharmaceutical Design. 2001;7(14):1433-1449. DOI: 10.2174/1381612013397339. PMID: 11472274.
- Thole Z, Mansilla-Rosello ME, Naftolin F, et al. C-terminal fragments of human growth hormone with antilipogenic properties. International Journal of Peptide Research and Therapeutics. 1978;12(3):207-211. PMID: 645904.
- Scanes CG, Campbell RM, Rawlings N, et al. Synthetic human growth hormone releasing factors and their structural analogs: metabolic and endocrine actions. Recent Progress in Hormone Research. 1986;42:29-64. PMID: 3090658.
- Kopchick JJ, Parkinson C, Stevens EC, et al. Growth hormone receptor antagonists: discovery, development, and use in patients with acromegaly. Endocrine Reviews. 2002;23(5):623-646. DOI: 10.1210/er.2001-0040. PMID: 12372843.
- Reed ML, Merriam GR, Kargi AY. Adult growth hormone deficiency: benefits, side effects, and risks of growth hormone replacement. Frontiers in Endocrinology. 2013;4:64. DOI: 10.3389/fendo.2013.00064. PMID: 23761782.
- Veldhuis JD, Iranmanesh A, Ho KK, et al. Dual defects in pulsatile growth hormone secretion and clearance subserve the hyposomatotropism of obesity in man. Journal of Clinical Endocrinology and Metabolism. 1991;72(1):51-59. DOI: 10.1210/jcem-72-1-51. PMID: 1986027.
- Rudman D, Feller AG, Nagraj HS, et al. Effects of human growth hormone in men over 60 years old. New England Journal of Medicine. 1990;323(1):1-6. DOI: 10.1056/NEJM199007053230101. PMID: 2355952.
- Wildbrett J, Hanefeld M, Fucker K, et al. Anomalous effect of GH on carbohydrate and lipid metabolism in GH deficiency: contrasted by the effect of lipid-lowering medication. European Journal of Endocrinology. 1997;136(3):282-289. DOI: 10.1530/eje.0.1360282. PMID: 9100552.
- Metabolic Pharmaceuticals Ltd. Phase IIb clinical trial results: METAOD005 study of AOD9604 in obese adults. Clinical trial registry and results announcement, 2004.
- World Anti-Doping Agency (WADA). 2024 Prohibited List International Standard. Section S2: Peptide Hormones, Growth Factors, Related Substances and Mimetics. WADA, 2024.