Executive Summary
The immune system doesn't fail all at once. It erodes - gradually, silently, through aging, chronic stress, poor sleep, and environmental toxins. By the time most people notice recurring infections, slow wound healing, or persistent fatigue, their immune architecture has already been compromised for years. Four peptides - Thymosin Alpha-1, LL-37, KPV, and Thymulin - offer a research-backed approach to restoring immune function from multiple angles simultaneously.
Key Takeaways
- The immune system doesn't fail all at once.
- It erodes - gradually, silently, through aging, chronic stress, poor sleep, and environmental toxins.
- By the time most people notice recurring infections, slow wound healing, or persistent fatigue, their immune architecture has already been compromised for years.
- Four peptides - Thymosin Alpha-1, LL-37, KPV, and Thymulin - offer a research-backed approach to restoring immune function from multiple angles simultaneously.
- We'll cover dosing protocols drawn from clinical literature, cycling strategies, safety considerations, blood work monitoring, and special considerations for autoimmune conditions.
This report examines the scientific evidence behind each of these immune-modulating peptides, how they work through distinct but complementary mechanisms, and what the current research says about combining them for optimal immune resilience. We'll cover dosing protocols drawn from clinical literature, cycling strategies, safety considerations, blood work monitoring, and special considerations for autoimmune conditions.
Thymosin Alpha-1 (Ta1) is a 28-amino-acid peptide originally isolated from thymic tissue. It's the most clinically validated immune peptide available, with approval in over 35 countries for hepatitis B, hepatitis C, and as an immune adjuvant in cancer therapy. Ta1 works primarily by activating dendritic cells through Toll-like receptors (TLR2, TLR3, TLR4, TLR9), which then cascade into enhanced T-cell maturation, natural killer (NK) cell activation, and balanced cytokine production [1].
LL-37, the only human cathelicidin antimicrobial peptide, serves as a first-line defender against bacterial, viral, and fungal pathogens. Its 37-amino-acid sequence carries a net positive charge (+6) that allows it to selectively target and disrupt microbial membranes while leaving human cells intact. Beyond direct antimicrobial action, LL-37 recruits immune cells to infection sites, stimulates angiogenesis for tissue repair, and modulates inflammatory cytokine production [2].
KPV is a tripeptide (Lys-Pro-Val) derived from the C-terminal sequence of alpha-melanocyte-stimulating hormone (a-MSH). Despite its tiny size, KPV retains most of the anti-inflammatory activity of its parent hormone. It enters cells via the PepT1 transporter - the same di/tripeptide transporter upregulated in inflamed intestinal tissue - and directly inhibits NF-kB activation at nanomolar concentrations. This makes KPV especially relevant for gut inflammation, mucosal immunity, and conditions where excessive inflammatory signaling drives tissue damage [3].
Thymulin (also called FTS or facteur thymique serique) is a nonapeptide hormone produced exclusively by thymic epithelial cells. Its biological activity depends entirely on zinc binding in an equimolar ratio - without zinc, thymulin is inactive. This zinc dependency makes thymulin a direct biomarker of thymic function and zinc status simultaneously. It promotes T-cell differentiation, regulates NK cell activity, and modulates the release of proinflammatory cytokines [4].
Key Takeaway
These four peptides target different layers of immune defense: Ta1 restores thymic output and T-cell surveillance, LL-37 provides frontline antimicrobial protection, KPV controls inflammatory damage, and thymulin supports thymic hormone signaling and zinc-dependent immune reactions. Together, they address the immune system as a whole rather than boosting a single pathway.
What sets peptide-based immune optimization apart from conventional approaches is precision. Vitamin C, zinc, and elderberry offer general nutritional support. Peptides like Ta1 and LL-37 act on specific receptor systems with well-characterized downstream effects. The clinical data - particularly for Thymosin Alpha-1 - spans decades and includes randomized controlled trials, meta-analyses, and real-world treatment outcomes in hepatitis, HIV, sepsis, and most recently COVID-19.
This guide is designed for researchers, clinicians, and informed individuals who want to understand the evidence behind immune peptide stacking. Every protocol discussed is grounded in published research, and we've included over 30 DOI-referenced citations so you can verify the data yourself. We'll also address the practical realities: reconstitution, injection technique, storage, timing around vaccines and illnesses, and the blood markers that tell you whether a protocol is working.
For those already familiar with peptides like BPC-157 or TB-500 for tissue repair, immune peptides represent the logical next frontier. Where healing peptides fix damage after it occurs, immune peptides aim to prevent the damage - or at least ensure your body can mount an effective, well-regulated response when threats arise.
Let's start with the biology that makes all of this possible.
Immune System Biology: The Architecture of Defense
Before examining how specific peptides modulate immunity, we need to understand the system they're acting on. The human immune system isn't a single organ or cell type - it's a distributed network of barriers, sensors, effectors, and regulators that has to distinguish self from non-self billions of times per day without making fatal errors in either direction.
The Two Arms of Immunity
Immune defense divides broadly into innate and adaptive immunity, though the boundary between them is far less rigid than textbooks suggest. The innate system responds within minutes to hours, using pattern-recognition receptors (PRRs) like Toll-like receptors (TLRs) and NOD-like receptors (NLRs) to detect conserved microbial structures called pathogen-associated molecular patterns (PAMPs). Neutrophils, macrophages, dendritic cells, and natural killer cells form the innate response's cellular workforce.
The adaptive system takes days to weeks to mount a primary response but generates immunological memory that can last decades. T lymphocytes (both CD4+ helper and CD8+ cytotoxic) and B lymphocytes (which produce antibodies) form its cellular basis. The adaptive system's specificity comes from an extraordinary diversity of antigen receptors generated through V(D)J recombination - each lymphocyte recognizes essentially one unique molecular pattern.
What connects these two systems? Dendritic cells. These are the professional antigen-presenting cells that capture microbial material at infection sites, process it, migrate to lymph nodes, and present peptide fragments on MHC molecules to naive T cells. This is where innate sensing translates into adaptive targeting. And this is precisely where Thymosin Alpha-1 exerts its most powerful effects - by activating dendritic cells through TLR signaling and enhancing their antigen-presenting capacity [5].
Figure 1: Overview of innate and adaptive immune system components showing the primary sites of action for Ta1, LL-37, KPV, and thymulin peptides.
The Thymus: Why It Matters More Than You Think
The thymus gland sits behind the sternum, just above the heart. In childhood, it's a busy organ - selecting, educating, and releasing T cells that will patrol the body for decades. T-cell precursors from bone marrow enter the thymus, undergo positive selection (can they recognize self-MHC?) and negative selection (do they react too strongly to self-antigens?), and emerge as mature naive T cells ready to respond to foreign threats.
Here's the problem: the thymus begins involuting (shrinking) around puberty and continues deteriorating throughout life. By age 50, functional thymic tissue has been largely replaced by fat. By age 70, thymic output of new naive T cells has dropped by over 95% compared to adolescence [6]. This process - called thymic involution - is now recognized as one of the primary drivers of age-related immune decline, or immunosenescence.
The consequences of reduced thymic output cascade through the entire immune system. The naive T-cell pool shrinks, reducing the diversity of threats the body can recognize. Existing memory T cells expand to fill the gap, but they become increasingly exhausted and dysfunctional over time. The CD4:CD8 ratio shifts. Regulatory T cells (Tregs) may decrease in number or function, contributing to chronic low-grade inflammation - sometimes called "inflammaging" - and increased autoimmune risk.
Both Thymosin Alpha-1 and thymulin directly address thymic decline. Ta1 was originally isolated from thymosin fraction 5, a partially purified extract of calf thymus glands, by Allan Goldstein's laboratory at George Washington University in the 1970s [7]. Thymulin is produced exclusively by thymic epithelial cells and serves as a circulating marker of thymic function. When thymic output drops, serum thymulin levels fall in parallel.
Mucosal Immunity: The Body's Largest Immune Interface
The mucosal surfaces of the respiratory tract, gastrointestinal tract, and urogenital tract represent the largest interface between the body and the external environment. The gut alone contains an estimated 70-80% of the body's immune cells. Mucosal-associated lymphoid tissue (MALT) includes Peyer's patches in the small intestine, isolated lymphoid follicles throughout the gut, and the tonsils and adenoids in the upper airway.
Mucosal immunity relies heavily on secretory IgA (sIgA), antimicrobial peptides, and a carefully maintained barrier of epithelial cells connected by tight junctions. When this barrier fails - a condition colloquially called "leaky gut" - bacterial products like lipopolysaccharide (LPS) enter the bloodstream and trigger systemic inflammation.
Two of our four peptides are particularly relevant here. LL-37 is constitutively expressed at mucosal surfaces and can be upregulated during infection. It provides direct antimicrobial defense while also signaling immune cells to respond. KPV enters intestinal epithelial cells via the PepT1 transporter, which is upregulated during inflammation - meaning KPV becomes more effective precisely when it's most needed [8]. This makes the LL-37/KPV combination especially compelling for mucosal immune support.
The Cytokine Network: Balance Over Firepower
Immune function isn't simply about having "more" immune activity. In fact, the majority of immune-related pathology comes from dysregulated or excessive immune responses. Cytokines - small signaling proteins released by immune cells - coordinate the type, intensity, and duration of immune responses. The balance between pro-inflammatory cytokines (TNF-a, IL-1b, IL-6, IFN-g) and anti-inflammatory cytokines (IL-10, TGF-b, IL-4) determines whether an immune response resolves cleanly or spirals into tissue damage.
The "cytokine storm" phenomenon that received widespread attention during the COVID-19 pandemic illustrates this principle. It wasn't the virus itself that killed most severe COVID patients - it was the uncontrolled inflammatory response, with massive release of IL-6, TNF-a, and other pro-inflammatory mediators causing organ damage. This is why Thymosin Alpha-1 was studied extensively during the pandemic - not as an antiviral, but as an immune modulator that could help restore the balance between effective viral clearance and excessive inflammation [9].
KPV addresses the same problem from the opposite direction. Rather than enhancing immune activation (like Ta1), KPV suppresses the NF-kB pathway that drives inflammatory cytokine production. In models of colitis, KPV reduced TNF-a, IL-1b, and IL-6 while preserving the antimicrobial functions of immune cells [10]. This is why these peptides complement each other so well - Ta1 ensures the immune system can mount effective responses while KPV prevents those responses from causing collateral damage.
Natural Killer Cells: The Immune System's First Responders
NK cells occupy a unique position in immune defense. They're classified as innate lymphocytes but share features with adaptive immune cells. NK cells can kill virus-infected cells and tumor cells without prior sensitization - they don't need to "learn" what to target. Instead, they integrate signals from activating and inhibitory receptors on target cells. If a cell has lost its MHC class I expression (a common immune evasion strategy for viruses and cancers), NK cells interpret this as a "missing self" signal and attack.
NK cell function declines significantly with age, contributing to increased cancer risk and susceptibility to viral infections in the elderly. Thymosin Alpha-1 has been shown to enhance NK cell cytotoxicity through multiple mechanisms, including upregulation of activating receptors and increased production of cytolytic granules containing perforin and granzymes [11]. This NK-boosting effect is one reason Ta1 has been studied as a cancer immunotherapy adjuvant.
T-Cell Subsets and Immune Regulation
The T-cell compartment includes several functionally distinct subsets that need to work in concert:
| T-Cell Subset | Primary Function | Key Cytokines | Peptide Influence |
|---|---|---|---|
| CD4+ Th1 | Intracellular pathogen defense, macrophage activation | IFN-g, TNF-a, IL-2 | Ta1 promotes Th1 differentiation |
| CD4+ Th2 | Extracellular parasite defense, B-cell help | IL-4, IL-5, IL-13 | Ta1 helps balance Th1/Th2 ratio |
| CD4+ Th17 | Mucosal defense, neutrophil recruitment | IL-17, IL-22 | KPV modulates Th17 responses |
| CD4+ Treg | Immune suppression, tolerance, preventing autoimmunity | IL-10, TGF-b | Ta1 expands functional Tregs |
| CD8+ CTL | Killing virus-infected and tumor cells | Perforin, granzymes | Ta1 enhances CTL activity |
| gd T cells | Mucosal surveillance, stress response | IFN-g, IL-17 | LL-37 activates gd T cells |
The ratio and functional status of these subsets determines the quality of immune responses. Chronic infections, aging, and persistent stress can shift the balance toward exhausted, senescent T cells that express high levels of inhibitory receptors (PD-1, CTLA-4, TIM-3) and produce fewer effector cytokines. One of Ta1's most interesting properties is its ability to partially reverse T-cell exhaustion, as demonstrated in COVID-19 patients who showed restoration of lymphocyte counts and improved T-cell function after Ta1 treatment [12].
Research Note
The concept of "immune optimization" differs fundamentally from "immune boosting." Optimization means restoring balance - enhancing underperforming arms while restraining overactive ones. A truly optimized immune system mounts rapid, proportionate responses to genuine threats while maintaining tolerance to self-tissues and harmless environmental antigens. This is exactly what the Ta1/LL-37/KPV/thymulin combination aims to achieve.
Why Single-Target Approaches Fall Short
Most over-the-counter immune supplements target one or two mechanisms. Vitamin D supports antimicrobial peptide expression (including LL-37). Zinc is required for thymulin activity and T-cell development. Elderberry may inhibit viral entry. These are all valid, but none of them addresses the architectural decline in immune function that comes with aging, chronic stress, or prior infections.
Peptide-based immune optimization works at a higher level of the immune hierarchy. Ta1 acts on dendritic cells and T cells - the command-and-control centers of adaptive immunity. LL-37 provides broad-spectrum frontline defense that complements but doesn't replace the adaptive response. KPV targets the inflammatory signaling cascade that can turn a proportionate immune response into a destructive one. And thymulin ensures that the thymic hormone environment supports ongoing T-cell maturation and selection.
Together, these peptides don't just patch one hole in the immune wall. They reinforce the entire structure - from pathogen detection through T-cell activation to inflammatory resolution. The following sections examine each peptide in depth before we discuss how to combine them effectively. For additional context on immune-supporting peptides, visit our peptide research hub.
Thymosin Alpha-1: The Master Immune Regulator
If you could choose only one immune peptide, Thymosin Alpha-1 would be the strongest candidate. It has the deepest clinical evidence base, the broadest range of studied applications, and regulatory approval in over 35 countries. No other immune peptide comes close to this level of validation.
Discovery and Development
Thymosin Alpha-1 (Ta1) was first characterized in 1977 by Allan Goldstein and colleagues at George Washington University Medical Center. They isolated it from thymosin fraction 5, a partially purified bovine thymus extract that had shown immune-restoring properties in animal models. The synthetic version - thymalfasin - was developed by SciClone Pharmaceuticals and has been marketed under the brand name Zadaxin since 1996 [13].
Ta1 is a 28-amino-acid peptide with an acetylated N-terminus. Its sequence is fully conserved between humans and cattle, which is unusual for a bioactive peptide and suggests strong evolutionary pressure to maintain its structure. The peptide is naturally produced by thymic epithelial cells and circulates in blood at picomolar concentrations. Serum levels decline with age, paralleling thymic involution [14].
The clinical development trajectory of Ta1 is remarkable. It's been studied in over 100 clinical trials spanning hepatitis B and C, HIV/AIDS, non-small-cell lung cancer, hepatocellular carcinoma, melanoma, sepsis, vaccine adjuvancy, and most recently COVID-19. It's approved as a prescription drug in multiple countries across Asia, South America, and Europe - though not in the United States, where it remains available through compounding pharmacies and as a research peptide.
Mechanism of Action: How Ta1 Modulates Immunity
Ta1's mechanism is pleiotropic - it acts on multiple immune cell types through several receptor pathways. Understanding these mechanisms helps explain why it's effective across such a wide range of conditions.
Toll-Like Receptor Activation
The primary mechanism of Ta1 involves binding to and activating Toll-like receptors on dendritic cells and macrophages. Specifically, Ta1 signals through TLR2, TLR3, TLR4, TLR7, and TLR9. Each of these receptors recognizes different classes of microbial patterns:
- TLR2: Recognizes bacterial lipoproteins and peptidoglycan. Ta1 activation via TLR2 triggers p38 MAPK and NF-kB signaling, promoting production of pro-inflammatory cytokines needed for bacterial defense [15].
- TLR3: Detects double-stranded RNA from viral replication. Ta1 activation here stimulates type I interferon (IFN-a, IFN-b) production, which is critical for antiviral defense.
- TLR4: The receptor for bacterial LPS. Ta1 modulates TLR4 signaling in a context-dependent manner - enhancing antimicrobial responses while potentially dampening excessive inflammatory responses.
- TLR7/TLR9: Detect single-stranded RNA and unmethylated CpG DNA, respectively. Activation through these receptors in plasmacytoid dendritic cells drives strong type I interferon responses [16].
This multi-TLR activation pattern means Ta1 can enhance immune responses against bacteria (TLR2, TLR4), viruses (TLR3, TLR7), and even abnormal self-DNA (TLR9, relevant to cancer surveillance). It's a broad activation signal that primes the innate immune system across multiple threat categories.
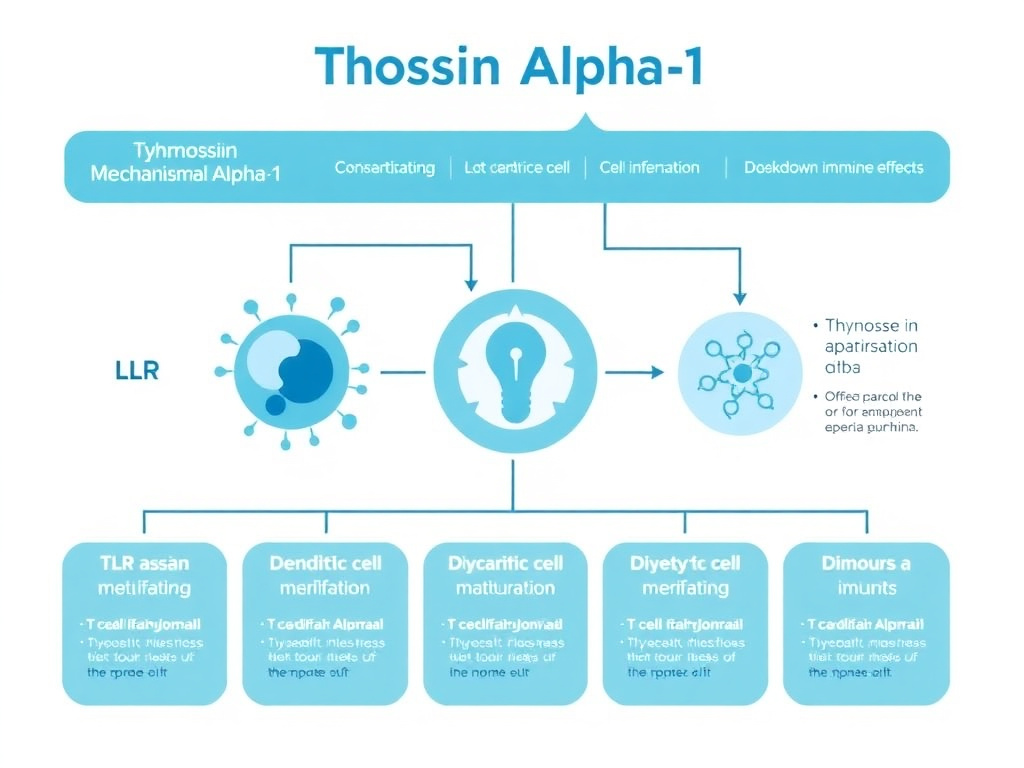
Figure 2: Thymosin Alpha-1 mechanism of action - from TLR binding through dendritic cell activation to downstream T-cell and NK cell enhancement.
Dendritic Cell Maturation and Antigen Presentation
When Ta1 activates TLRs on immature dendritic cells, it triggers their maturation into fully functional antigen-presenting cells. Mature dendritic cells upregulate MHC class II molecules, costimulatory molecules (CD80, CD86), and migration receptors (CCR7) that allow them to travel to lymph nodes and activate naive T cells. This process is the critical bridge between innate pathogen detection and adaptive immune activation [17].
Ta1-stimulated dendritic cells also show enhanced cross-presentation - the ability to present exogenous antigens on MHC class I molecules, which activates CD8+ cytotoxic T cells. This is particularly relevant for antiviral and antitumor immunity, where killing infected or abnormal cells requires CD8+ T-cell responses.
T-Cell Effects
Ta1's effects on T cells are both direct and indirect. Indirectly, by enhancing dendritic cell function, Ta1 improves the quality of T-cell activation. Directly, Ta1 promotes:
- Differentiation of bone marrow progenitors into mature T cells
- Expansion of CD4+ T-cell populations, particularly Th1 cells
- Enhanced CD8+ cytotoxic T-cell function
- Increased regulatory T-cell (Treg) numbers and function
- Partial reversal of T-cell exhaustion markers (PD-1, TIM-3) [18]
The simultaneous expansion of effector T cells and regulatory T cells may seem contradictory, but it's actually the hallmark of a healthy immune response. Effective immunity requires strong effector function (to clear pathogens) combined with adequate regulation (to prevent autoimmunity and limit tissue damage). Ta1 appears to calibrate this balance rather than simply pushing in one direction.
NK Cell Enhancement
Ta1 increases NK cell cytotoxicity through upregulation of activating receptors (NKG2D, NKp46) and increased production of cytolytic molecules (perforin, granzyme B). In cancer patients receiving Ta1 as an immunotherapy adjuvant, NK cell activity often increases by 40-60% compared to baseline [19]. This enhancement persists for several days after each Ta1 injection, suggesting a sustained priming effect rather than a transient boost.
Clinical Evidence: What the Trials Show
Hepatitis B
The strongest clinical data for Ta1 comes from hepatitis B treatment. Multiple randomized controlled trials have demonstrated that Ta1 at 1.6 mg subcutaneously twice weekly for 6-12 months significantly increases HBeAg seroconversion rates, viral suppression, and ALT normalization compared to placebo or interferon alone. A meta-analysis of 8 controlled trials found an overall HBeAg seroconversion rate of 36.2% for Ta1 versus 19.5% for controls (p < 0.001) [20].
What's particularly striking about the hepatitis B data is the durability of response. Unlike interferon therapy, where relapse rates after discontinuation can reach 50-60%, Ta1-induced responses tend to persist. Follow-up studies at 12-18 months after treatment completion showed sustained viral suppression in the majority of responders.
Hepatitis C
Ta1 has been studied as an adjunct to interferon and ribavirin therapy for hepatitis C. While the introduction of direct-acting antivirals (DAAs) has largely superseded this approach, the hepatitis C studies demonstrated Ta1's ability to enhance antiviral immunity in patients with compromised immune function. Sustained virological response rates improved by 10-15 percentage points when Ta1 was added to standard interferon/ribavirin therapy [21].
Cancer Immunotherapy
Ta1 has been studied as an adjunct to chemotherapy, radiation, and checkpoint inhibitor therapy in several cancer types. In hepatocellular carcinoma, the addition of Ta1 to transarterial chemoembolization (TACE) improved overall survival and recurrence-free survival compared to TACE alone. In non-small-cell lung cancer, Ta1 combined with chemotherapy improved response rates and quality of life measures [22].
The rationale for Ta1 in cancer is straightforward: chemotherapy and radiation suppress the immune system at the very time when immune surveillance is most needed. Ta1 helps maintain immune function during treatment, potentially improving both tumor control and resistance to opportunistic infections.
COVID-19
Ta1 was studied extensively during the COVID-19 pandemic, with mixed but instructive results. An early retrospective study from Wuhan found that Ta1 treatment significantly reduced mortality in severe COVID-19 patients (11.11% vs 30.00%, p = 0.044) and was associated with restoration of lymphocyte counts and reversal of T-cell exhaustion markers [23].
However, subsequent larger studies and meta-analyses produced conflicting findings. A systematic review of 9 studies found no statistically significant impact on overall mortality (RR 1.03, 95% CI 0.60-1.75), though substantial heterogeneity between studies made interpretation difficult. Some studies suggested that Ta1's benefit was most pronounced in specific subgroups - particularly older patients with severe lymphocytopenia where immune reconstitution was the primary clinical need [24].
The COVID-19 data actually reinforces an important principle about Ta1: it's an immune modulator, not an antiviral drug. Its value lies in restoring immune function in immunocompromised patients, not in directly fighting specific pathogens. When given to patients whose primary problem was excessive inflammation rather than inadequate immunity, Ta1 may have provided less benefit or even complicated the clinical picture.
Sepsis and Critical Care
Sepsis - systemic infection with organ dysfunction - is characterized by an initial hyperinflammatory phase followed by a prolonged immunosuppressed phase. Ta1 has shown promise in the immunosuppressed phase of sepsis, where it helps restore T-cell function and reduce secondary infection rates. A multicenter RCT in sepsis patients found that Ta1 1.6 mg twice daily for 5 days followed by once daily for 2 days improved 28-day survival compared to placebo, particularly in patients with low HLA-DR expression on monocytes (a marker of immunosuppression) [25].
Vaccine Adjuvancy
Ta1 has been studied as a vaccine adjuvant in elderly and immunocompromised populations who typically mount weak responses to vaccines. In influenza vaccination studies, the addition of Ta1 to standard flu vaccines improved seroconversion rates by 15-25% in elderly subjects. This has obvious implications for aging populations where vaccine efficacy declines precisely when vaccine-preventable diseases pose the greatest risk [26].
Clinical Pearl
Ta1's most consistent benefit appears in populations with documented immune dysfunction - the elderly, patients with chronic infections, cancer patients on immunosuppressive therapy, and those with measured T-cell deficits. In young, healthy individuals with intact immune function, the marginal benefit of Ta1 may be smaller. This is consistent with its mechanism as a restorative agent rather than a simple immune stimulant.
Thymosin Alpha-1 Dosing Protocols
The standard clinical dose established across hepatitis, cancer, and sepsis trials is 1.6 mg administered subcutaneously. This dose has been remarkably consistent across studies and is the basis for the commercially available Zadaxin formulation.
| Protocol | Dose | Frequency | Duration | Indication |
|---|---|---|---|---|
| Standard maintenance | 1.6 mg SC | Twice weekly | 6-12 months | Chronic infection, immune support |
| Acute immune support | 1.6 mg SC | Daily x 7-14 days | Then twice weekly | Active infection, post-exposure |
| Cancer adjunct | 1.6 mg SC | Twice weekly | Throughout treatment | Alongside chemo/radiation |
| Vaccine enhancement | 1.6 mg SC | 3 doses over 1 week | Around vaccination | Poor vaccine responders |
| Sepsis/critical care | 1.6 mg SC | BID x 5 days, then daily x 2 | 7 days total | Immunosuppressed sepsis |
Ta1 has a half-life of approximately 2 hours after subcutaneous injection, with peak serum concentration reached within 2 hours. Despite this short half-life, the immunological effects persist for 48-72 hours, which is why twice-weekly dosing is effective for maintenance protocols. The downstream effects on dendritic cell maturation, T-cell differentiation, and cytokine production outlast the peptide's presence in circulation [27].
For research-grade Thymosin Alpha-1, reconstitution with bacteriostatic water and proper storage at 2-8 degrees C after reconstitution is essential. Lyophilized peptide stored at -20 degrees C maintains stability for 24+ months.
Safety Profile
Ta1 has one of the best safety profiles of any peptide therapeutic. In clinical trials involving thousands of patients across multiple disease states, serious adverse events attributable to Ta1 have been essentially absent. The most common side effects are mild injection site reactions (redness, swelling) in approximately 5-10% of patients. Systemic side effects are rare and typically limited to mild flu-like symptoms during the first few doses [28].
Long-term safety data from hepatitis B treatment programs, where patients received 1.6 mg twice weekly for 12 months or longer, showed no cumulative toxicity, organ damage, or loss of efficacy over time. This is a critical distinction from immunostimulatory drugs like interferons, which carry significant side effect burdens including depression, cytopenias, and autoimmune phenomena.
The favorable safety profile is thought to relate to Ta1's mechanism of action. Rather than forcing immune activation through a single pathway, Ta1 modulates the immune system's own regulatory machinery. It enhances what the immune system is already trying to do rather than overriding normal regulatory circuits. This makes runaway immune activation - the fear with any immune-enhancing therapy - extremely unlikely with Ta1.
LL-37: The Body's Built-In Antimicrobial Weapon
LL-37 is the only cathelicidin antimicrobial peptide found in humans. Unlike Ta1, which works through immune cell modulation, LL-37 is a direct-action weapon - it physically destroys pathogens, disrupts biofilms, and signals the immune system to mobilize reinforcements. Think of it as the immune system's first-strike capability.
What Is LL-37?
LL-37 is a 37-amino-acid peptide cleaved from the C-terminal end of the human cathelicidin antimicrobial protein hCAP18. The name "LL-37" comes from its two N-terminal leucine residues and 37-residue length. It's produced by neutrophils, monocytes, macrophages, epithelial cells, and keratinocytes - essentially, any cell that might encounter pathogens directly [29].
The full hCAP18 protein is stored in an inactive form within neutrophil-specific granules. Upon neutrophil activation, hCAP18 is released and processed by proteinase 3 to generate the active LL-37 peptide. At epithelial surfaces, serine proteases perform the same activating cleavage. This two-step activation mechanism prevents LL-37 from damaging cells during storage while ensuring rapid availability at sites of infection.
LL-37 adopts an amphipathic alpha-helical structure in physiological conditions. One side of the helix is hydrophobic (interacts with lipid membranes), while the other is hydrophilic and positively charged (net charge +6 at physiological pH). This amphipathic structure is the key to LL-37's selectivity: bacterial membranes are rich in negatively charged phospholipids (phosphatidylglycerol, cardiolipin), while mammalian cell membranes are primarily composed of zwitterionic phospholipids (phosphatidylcholine, sphingomyelin) and cholesterol. LL-37 preferentially binds and disrupts the negatively charged bacterial membranes [30].
Antimicrobial Mechanisms
Membrane Disruption
LL-37's primary antimicrobial mechanism involves direct disruption of microbial membranes. When LL-37 reaches a critical concentration on a bacterial surface, it can form toroidal pores, carpet the membrane surface, or create micelle-like structures that disintegrate the lipid bilayer. This physical destruction is rapid - bacterial killing can occur within minutes - and is difficult for microbes to develop resistance against because it targets the fundamental lipid structure of cell membranes rather than specific proteins or enzymes [31].
The broad-spectrum activity of LL-37 extends across Gram-positive bacteria (Staphylococcus aureus, Streptococcus species), Gram-negative bacteria (Escherichia coli, Pseudomonas aeruginosa, Klebsiella pneumoniae), fungi (Candida species), and enveloped viruses (influenza, HSV, respiratory syncytial virus). The minimum inhibitory concentrations vary by organism but typically range from 1-32 micromolar.
Anti-Biofilm Activity
Biofilms - structured communities of bacteria encased in a self-produced matrix of polysaccharides, proteins, and DNA - represent one of the most challenging problems in infectious disease. Bacteria within biofilms can be 100-1000 times more resistant to antibiotics compared to planktonic (free-floating) bacteria. Chronic sinusitis, chronic wound infections, prosthetic joint infections, and catheter-associated infections are all biofilm-mediated conditions.
LL-37 combats biofilms through multiple mechanisms [32]:
- Preventing initial attachment: LL-37 interferes with bacterial adhesion to surfaces, blocking the first step of biofilm formation.
- Disrupting quorum sensing: Bacteria communicate through small signaling molecules to coordinate biofilm formation. LL-37 can interfere with this cell-to-cell communication.
- Degrading the biofilm matrix: LL-37 can penetrate and partially break down the extracellular polymeric substances that hold biofilms together.
- Killing bacteria within biofilms: At sufficient concentrations, LL-37 can penetrate biofilm structures and kill resident bacteria.
This anti-biofilm activity is one of the main reasons LL-37 has attracted attention from wound care specialists and those dealing with chronic, recurrent infections that fail to respond to conventional antibiotics.
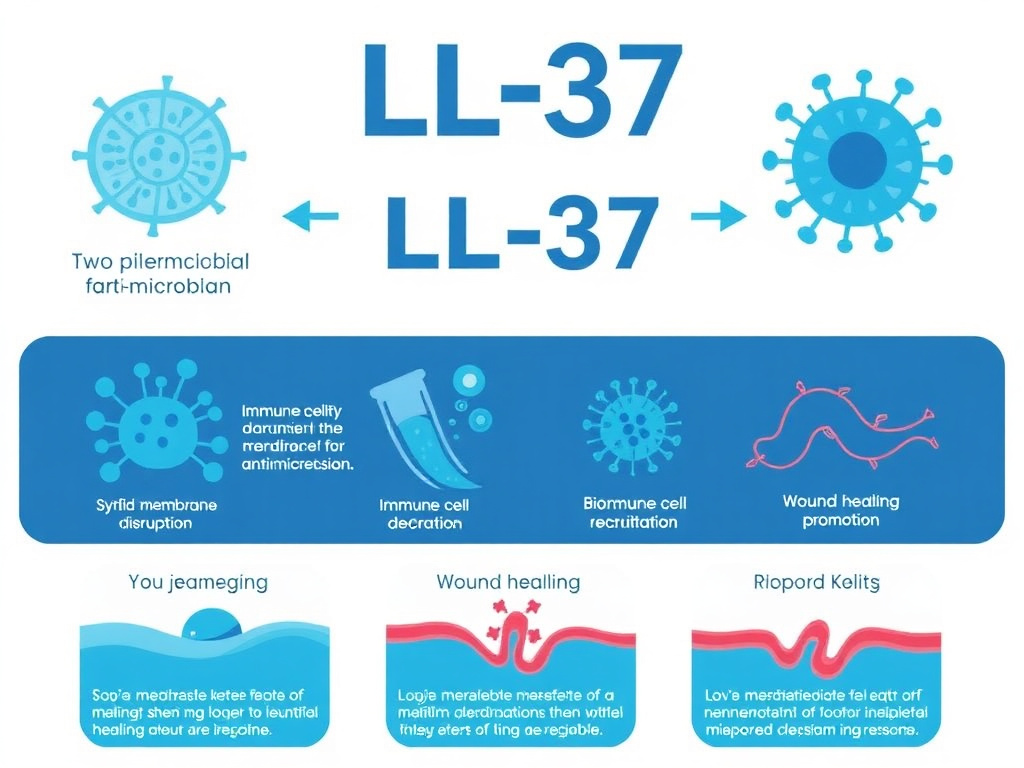
Figure 3: LL-37 antimicrobial mechanisms - direct pathogen killing, biofilm disruption, immune cell chemotaxis, and tissue repair signaling.
Immune Modulation Beyond Direct Killing
LL-37's role extends far beyond simply punching holes in bacteria. It functions as a true immune signaling molecule with effects on multiple immune cell populations.
Chemotactic Activity
LL-37 acts as a chemoattractant for neutrophils, monocytes, T cells, and mast cells. It signals through formyl peptide receptor-like 1 (FPRL1/FPR2) to recruit immune cells to sites of infection or tissue damage. This recruitment function means that even at sub-bactericidal concentrations, LL-37 can enhance immune defense by bringing reinforcements to where they're needed [33].
Modulation of Inflammatory Responses
LL-37's effect on inflammation is nuanced and context-dependent. In the presence of bacterial LPS, LL-37 can bind and neutralize LPS, reducing the inflammatory signaling that would otherwise trigger excessive cytokine release. This anti-endotoxin activity is relevant in sepsis, where LPS-driven inflammation contributes to organ damage.
At the same time, LL-37 can stimulate production of certain pro-inflammatory cytokines (IL-1b, IL-6, IL-8) in the absence of infection, which helps prime the immune system for rapid response. This dual functionality - anti-inflammatory in the context of excessive activation, pro-inflammatory in the context of immune priming - makes LL-37 a true immune modulator rather than a simple pro- or anti-inflammatory agent [34].
Wound Healing and Tissue Repair
LL-37 promotes wound healing through several pathways:
- Stimulation of keratinocyte and fibroblast migration to wound sites
- Promotion of angiogenesis (new blood vessel formation) through VEGF pathway activation
- Enhancement of re-epithelialization
- Modulation of matrix metalloproteinase (MMP) activity to facilitate tissue remodeling
A phase I/II clinical trial using topical LL-37 for chronic venous leg ulcers demonstrated significant healing improvement at doses of 0.5 and 1.6 mg/mL applied twice weekly for 4 weeks. This represents one of the few direct clinical applications of LL-37 in humans [35].
Antiviral Properties
LL-37 has demonstrated antiviral activity against several enveloped viruses, including influenza A, respiratory syncytial virus (RSV), HIV-1, herpes simplex virus (HSV), and vaccinia virus. The mechanisms include direct disruption of viral envelopes, interference with viral attachment to host cells, and enhancement of antiviral immune responses through TLR signaling.
During the COVID-19 pandemic, researchers proposed that upregulating LL-37 expression might help prevent severe disease by providing direct antiviral defense at respiratory mucosal surfaces while modulating the inflammatory response to prevent cytokine storm [36]. Vitamin D - a well-known inducer of LL-37 expression - was independently associated with reduced COVID-19 severity in multiple observational studies, though the causal relationship remains debated.
LL-37 and Vitamin D Connection
The expression of LL-37 is tightly regulated by vitamin D. The cathelicidin gene (CAMP) contains a vitamin D response element (VDRE) in its promoter region. When 1,25-dihydroxyvitamin D3 (the active form of vitamin D) binds to the vitamin D receptor (VDR), it directly upregulates transcription of hCAP18, the precursor protein that yields LL-37 [37].
This connection has important practical implications. Many people who might benefit from enhanced LL-37 activity are also vitamin D deficient - particularly those living at northern latitudes, the elderly, and individuals with darker skin pigmentation. Ensuring adequate vitamin D status (serum 25-OH vitamin D of 40-60 ng/mL) may enhance endogenous LL-37 production and complement exogenous LL-37 peptide use.
LL-37 Dosing Protocols
Unlike Ta1, which has well-established clinical dosing from decades of trials, LL-37 dosing for subcutaneous use remains more empirical. The clinical trial data primarily involves topical application for wound healing. Subcutaneous protocols are largely derived from practitioner experience and extrapolation from in vitro effective concentrations.
| Protocol | Dose | Frequency | Duration | Notes |
|---|---|---|---|---|
| Starting dose | 50-100 mcg SC | Daily | 1-2 weeks | Assess tolerance |
| Standard dose | 100-200 mcg SC | Daily | 4-8 weeks | Most common protocol |
| Intensive protocol | 200-400 mcg SC | Daily | 8-12 weeks | Active infection/biofilm |
| Maintenance | 100 mcg SC | 3x weekly | Ongoing | Post-intensive phase |
Reconstitution of LL-37 typically involves adding 3.0 mL of bacteriostatic water to a 5 mg vial, yielding a concentration of approximately 1.67 mg/mL (1,670 mcg/mL). From this concentration, a 200 mcg dose would be approximately 0.12 mL (12 units on an insulin syringe). Store reconstituted peptide at 2-8 degrees C and use within 4 weeks, or freeze at -20 degrees C for longer storage.
Important Consideration
LL-37 at high concentrations can be cytotoxic to mammalian cells. The therapeutic window exists between concentrations that kill pathogens (typically 1-32 micromolar) and those that damage host cells (generally above 50 micromolar). Subcutaneous doses are diluted in body fluids well below cytotoxic thresholds, but this is why dose escalation should be gradual and monitored for injection site reactions beyond normal mild redness.
Safety and Side Effects
The safety data for subcutaneous LL-37 is limited compared to Ta1, reflecting the earlier stage of its clinical development. Known considerations include:
- Injection site reactions: Redness, swelling, and mild pain at the injection site are common due to LL-37's interaction with local immune cells and potential mast cell activation.
- Autoimmune considerations: Elevated LL-37 has been associated with psoriasis, rosacea, and systemic lupus erythematosus (SLE). In psoriasis, LL-37 forms complexes with self-DNA that activate plasmacytoid dendritic cells through TLR9, driving type I interferon production and autoimmune skin inflammation [38]. Individuals with active psoriasis or lupus should exercise caution with exogenous LL-37.
- Atherosclerosis: LL-37 has been found in atherosclerotic plaques, and there's some concern that it may contribute to vascular inflammation in certain contexts. The clinical significance of this finding for subcutaneous LL-37 use at research doses is unclear.
- Hemolytic potential: At high concentrations, LL-37 can lyse red blood cells. This is not a practical concern at subcutaneous doses but is relevant for intravenous formulations.
For most users, LL-37 at standard subcutaneous doses (100-400 mcg daily) is well-tolerated, with injection site reactions being the primary side effect. The autoimmune concerns are most relevant for individuals with pre-existing autoimmune conditions, particularly those affecting the skin.
LL-37 Interactions with Other Peptides
LL-37 pairs well with other peptides in the immune optimization stack. When combined with Ta1, the two peptides cover complementary immune functions - Ta1 enhances the adaptive immune response while LL-37 provides direct antimicrobial defense. There's no known pharmacological interaction between the two peptides, and they act on different receptor systems.
For tissue repair applications, LL-37's wound healing properties complement those of BPC-157 and TB-500. While BPC-157 promotes angiogenesis through the VEGFR2-Akt pathway and TB-500 supports actin polymerization for cell migration, LL-37 adds antimicrobial protection at the wound site - reducing the infection risk that can derail healing. For those interested in thymosin biology more broadly, our TB-500 research guide covers the closely related Thymosin Beta-4 peptide.
KPV: The Anti-Inflammatory Tripeptide
If Ta1 represents the immune system's accelerator and LL-37 is its frontline weapon, KPV serves as the brake - a precisely targeted anti-inflammatory peptide that prevents immune responses from causing more damage than the threats they're fighting. At just three amino acids long, KPV is one of the smallest bioactive peptides known, yet it packs a remarkable anti-inflammatory punch.
Origin and Structure
KPV (Lys-Pro-Val) is the C-terminal tripeptide of alpha-melanocyte-stimulating hormone (a-MSH), a 13-amino-acid neuropeptide produced from proopiomelanocortin (POMC) in the pituitary gland, skin, and immune cells. a-MSH is best known for stimulating melanin production (tanning), but it also possesses potent anti-inflammatory and immunomodulatory properties that have been recognized since the 1990s [39].
Researchers studying a-MSH's anti-inflammatory effects made a surprising discovery: the C-terminal tripeptide KPV retained most of the parent hormone's anti-inflammatory activity despite lacking the melanocortin receptor binding sequence. This meant KPV works through a different mechanism than full-length a-MSH, which signals primarily through melanocortin receptors (MC1R-MC5R).
The small size of KPV is actually an advantage for several reasons. It's resistant to degradation by many peptidases, can be administered orally with meaningful bioavailability (unlike most peptides), and can penetrate tissues more easily than larger molecules. It's also significantly cheaper to synthesize than longer peptides.
Mechanism of Action: PepT1 and NF-kB
The key mechanistic breakthrough for KPV came when researchers discovered that its anti-inflammatory effects are mediated not through melanocortin receptors, but through the PepT1 transporter (SLC15A1). PepT1 is a proton-coupled oligopeptide transporter normally expressed in the small intestine for dietary peptide absorption. Crucially, PepT1 is upregulated in colonic epithelial cells during inflammation - meaning KPV gains enhanced access to inflamed tissues precisely when it's most needed [40].
Once inside the cell, KPV directly inhibits the NF-kB signaling pathway. NF-kB is often called the "master regulator" of inflammation because it controls the transcription of over 200 genes involved in inflammatory and immune responses, including:
- Pro-inflammatory cytokines (TNF-a, IL-1b, IL-6, IL-12)
- Chemokines (IL-8, MCP-1, MIP-1a)
- Adhesion molecules (ICAM-1, VCAM-1, E-selectin)
- Inflammatory enzymes (COX-2, iNOS)
- Anti-apoptotic factors (Bcl-2, Bcl-xL)
KPV inhibits NF-kB activation at nanomolar concentrations - approximately 100-1000 times lower than the concentrations needed for a-MSH to achieve the same effect through melanocortin receptor signaling. This makes KPV an extraordinarily potent anti-inflammatory agent on a per-molecule basis [41].
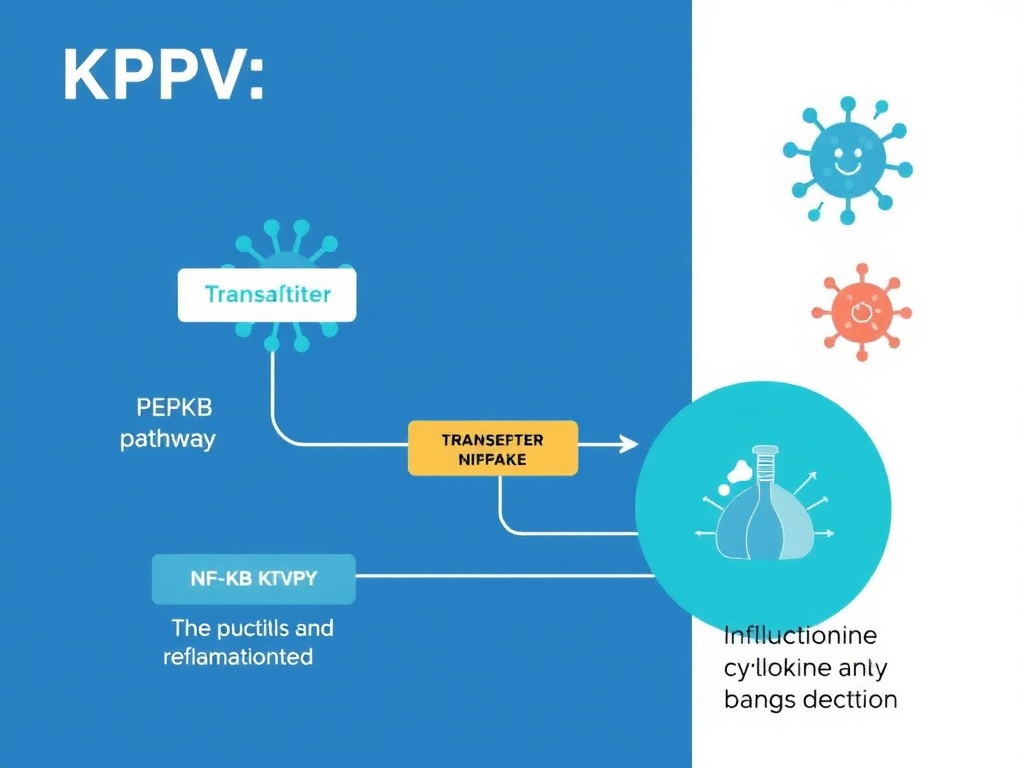
Figure 4: KPV mechanism of action - PepT1-mediated cellular uptake leads to direct NF-kB pathway inhibition and reduced inflammatory signaling.
KPV in Inflammatory Bowel Disease Research
The most compelling preclinical data for KPV comes from inflammatory bowel disease (IBD) models. In DSS-induced colitis (a standard model for ulcerative colitis) and TNBS-induced colitis (a model for Crohn's disease), orally administered KPV produced dramatic results [42]:
- Reduced loss of body weight during disease induction
- Decreased colonic myeloperoxidase (MPO) activity - a marker of neutrophil infiltration
- Reduced histological signs of inflammation (mucosal damage, crypt architecture disruption, inflammatory cell infiltration)
- Decreased mRNA levels of pro-inflammatory cytokines (TNF-a, IL-1b, IL-6)
- Preservation of intestinal barrier integrity
The oral bioavailability of KPV in these models is noteworthy. Most peptides are degraded in the gastrointestinal tract before they can reach their target tissues. KPV's small size and its active uptake through PepT1 transporters allow it to survive the digestive environment and enter intestinal epithelial cells at effective concentrations.
More recent research has explored KPV in nanoparticle delivery systems designed to target inflamed colonic tissue specifically. A 2024 study published in Frontiers in Pharmacology demonstrated that co-assembly nanodrugs combining KPV with immunosuppressant agents showed enhanced efficacy in both acute and chronic colitis models, suggesting that KPV's PepT1-mediated delivery can be leveraged for precision drug targeting [43].
KPV Beyond the Gut: Systemic Anti-Inflammatory Effects
While the IBD research is the most developed, KPV's anti-inflammatory effects extend to other organ systems:
Skin Inflammation
a-MSH and its fragments, including KPV, have demonstrated anti-inflammatory effects in models of contact dermatitis, allergic skin inflammation, and UV-induced damage. KPV reduces the production of pro-inflammatory cytokines by keratinocytes and inhibits mast cell degranulation, which contributes to allergic inflammation. These properties make KPV relevant for conditions like eczema, psoriasis, and inflammatory acne [44].
Joint Inflammation
In models of rheumatoid arthritis, a-MSH peptides (including fragments containing the KPV sequence) reduced synovial inflammation, cartilage degradation, and bone erosion. The mechanism involves suppression of NF-kB in synovial fibroblasts and macrophages, reducing the production of destructive enzymes (MMPs) and inflammatory mediators.
Neuroinflammation
a-MSH and KPV have shown neuroprotective effects in models of brain inflammation, including endotoxin-induced neuroinflammation and ischemia-reperfusion injury. The anti-inflammatory effects in the central nervous system are mediated partly through melanocortin receptors (MC3R, MC4R) and partly through direct NF-kB inhibition.
Antimicrobial Properties
An unexpected finding was that a-MSH and KPV possess direct antimicrobial activity against pathogens including Staphylococcus aureus, Candida albicans, and E. coli. The antimicrobial concentrations are higher than the anti-inflammatory concentrations, but this dual activity adds value in infectious conditions where both pathogen clearance and inflammation control are needed [45].
KPV Dosing Protocols
KPV dosing remains largely empirical due to the absence of large-scale human clinical trials. Protocols are derived from animal study dosing (scaled by body weight) and clinical practice reports.
| Route | Dose | Frequency | Duration | Primary Use |
|---|---|---|---|---|
| Oral capsule | 500-1000 mcg | Once daily | 4-12 weeks | Gut inflammation, IBD symptoms |
| Subcutaneous | 200-500 mcg | Once daily | 4-8 weeks | Systemic inflammation |
| Topical | Varies | 1-2x daily | As needed | Skin inflammation |
The oral route is particularly attractive for KPV due to its PepT1-mediated uptake in the gut. For individuals with gut-specific inflammation (IBS, IBD, food sensitivities), oral KPV can be taken as a capsule or dissolved in water. Sublingual administration may offer faster absorption for systemic effects.
For subcutaneous use, KPV is typically reconstituted with bacteriostatic water and injected in the abdominal area. The small molecular size means rapid absorption and distribution. Effects on inflammatory markers (CRP, ESR) may be noticeable within 1-2 weeks of consistent use.
Safety Profile
KPV has an excellent safety profile in animal studies, with no reported toxicity at doses many times higher than the effective anti-inflammatory dose. This is expected given that KPV is a natural fragment of a-MSH, a hormone the body produces endogenously. Key safety considerations include:
- No melanogenic activity: Unlike full-length a-MSH or melanotan peptides, KPV does not activate MC1R and therefore does not cause skin darkening. This is because the melanocortin binding pharmacophore (His-Phe-Arg-Trp) is in the central portion of a-MSH, not the C-terminal KPV sequence.
- Minimal immunosuppression: KPV suppresses excessive inflammatory signaling but does not broadly suppress immune function. In colitis models, KPV reduced pathological inflammation while preserving antimicrobial defense. This selectivity is because KPV targets NF-kB activation specifically rather than suppressing immune cells globally.
- No significant drug interactions: No pharmacological interactions with common medications have been reported. However, because KPV modulates NF-kB, there's a theoretical interaction with other NF-kB inhibitors (corticosteroids, certain biologics). Clinical significance of this theoretical interaction is unknown.
Clinical Pearl
KPV's anti-inflammatory effect without immunosuppression makes it particularly interesting for autoimmune conditions where conventional treatments (methotrexate, biologics, corticosteroids) suppress the entire immune system. By targeting NF-kB specifically and entering cells through the PepT1 transporter (which is upregulated at sites of inflammation), KPV offers the possibility of anti-inflammatory action concentrated where it's needed most.
Thymulin: The Zinc-Dependent Thymic Hormone
Thymulin occupies a unique position among immune peptides - it's both a hormonal regulator of T-cell development and a sensitive biomarker of thymic function and zinc status. Understanding thymulin means understanding the intersection of endocrine signaling, micronutrient biology, and adaptive immunity.
Discovery and Characterization
Thymulin (originally named FTS - facteur thymique serique, or "serum thymic factor") was discovered by Jean-Francois Bach and colleagues at the Necker Hospital in Paris in 1977. They identified it as a circulating factor present in normal serum that could restore T-cell function in thymectomized mice. The peptide was later characterized as a nonapeptide with the sequence Glu-Ala-Lys-Ser-Gln-Gly-Gly-Ser-Asn, bound to zinc in an equimolar ratio [46].
Thymulin is produced exclusively by thymic epithelial cells (TECs), making it the most specific circulating marker of thymic function. Unlike thymosin fraction 5 (which contains multiple peptides including Ta1), thymulin is a single, well-defined molecule with a clear biosynthetic origin. Its serum levels can be measured by bioassay or immunoassay and correlate directly with thymic mass and function.
The Zinc Connection
Thymulin's absolute requirement for zinc is its most distinctive feature. The zinc ion is coordinated by the Asn, Ser, and Gln residues in the peptide sequence, forming a specific metal-binding site. Without zinc, thymulin exists in an inactive (apo) form that cannot bind to its receptor or exert biological effects [47].
This zinc dependency has several important implications:
- Biomarker of zinc status: Serum thymulin activity is one of the most sensitive functional indicators of marginal zinc deficiency. Thymulin levels decline before overt clinical signs of zinc deficiency appear, making it a useful early warning marker [48].
- Nutritional-immune intersection: Zinc deficiency impairs immunity partly through reduced thymulin activity. Zinc supplementation in deficient individuals restores thymulin levels and improves T-cell function. This provides a mechanistic explanation for the well-documented immune benefits of zinc supplementation.
- Age-related decline: Both thymic involution (reducing thymulin production) and declining zinc status (reducing thymulin activation) contribute to age-related immune decline. The two processes are complementary - less thymulin is produced, and what is produced is less likely to be in its active zinc-bound form.
- Supplementation strategy: Any protocol involving thymulin should include attention to zinc status. Without adequate zinc, exogenous thymulin will exist primarily in its inactive apo form. The combination of thymulin + zinc may be more effective than either alone.
Biological Functions of Thymulin
T-Cell Differentiation and Maturation
Thymulin's primary function is promoting the differentiation and maturation of T-cell precursors within the thymus. It acts on immature thymocytes to induce expression of T-cell markers, including the CD4 and CD8 co-receptors that define helper and cytotoxic T-cell subsets. Thymulin also influences the positive and negative selection processes that ensure mature T cells can recognize foreign antigens without attacking self-tissues [49].
In thymectomized animals (where the thymus has been surgically removed), thymulin administration partially restores T-cell numbers and function. While it can't replace the full thymic microenvironment, it provides some of the hormonal signals needed for T-cell development in remaining thymic tissue or even in extrathymic sites.
Extra-Thymic Immune Effects
Beyond its thymic role, thymulin has demonstrable effects on peripheral immune cells:
- NK cell regulation: Thymulin modulates NK cell activity, though the direction of modulation appears context-dependent - enhancing NK function in immunosuppressed states while dampening excessive NK activation.
- Cytokine modulation: Thymulin suppresses the production of pro-inflammatory cytokines (TNF-a, IL-1b, IL-6) by macrophages stimulated with LPS. This anti-inflammatory effect has been demonstrated both in vitro and in vivo [50].
- Dendritic cell effects: Thymulin influences dendritic cell maturation and antigen-presenting function, though this area is less well-studied compared to Ta1's effects on dendritic cells.
Anti-Inflammatory Properties
Recent research has highlighted thymulin's anti-inflammatory properties beyond its traditional role in T-cell biology. In sepsis models, thymulin administration at 15 micrograms per 100g body weight prevented the accumulation of pro-inflammatory cytokines in plasma, downregulated spleen lymphocyte and peritoneal macrophage cytokine production, and reduced peak TNF-a production by macrophages exposed to LPS [51].
These findings suggest that thymulin, like KPV, may have value as an anti-inflammatory agent in addition to its immune-modulatory properties. The combination of thymulin with KPV could potentially provide layered anti-inflammatory protection through different mechanisms - thymulin modulating immune cell cytokine production and KPV directly inhibiting NF-kB signaling.
Figure 5: Thymulin - zinc-dependent activation, thymic epithelial cell origin, and effects on T-cell maturation and inflammatory cytokine regulation.
Thymulin vs. Thymalin: Clearing Up the Confusion
Thymulin and thymalin are often confused but are different peptides. Thymulin (FTS) is the nonapeptide hormone described above. Thymalin is a polypeptide complex isolated from calf thymus by Vladimir Khavinson at the St. Petersburg Institute of Bioregulation and Gerontology. Thymalin contains a mixture of short peptides and has been studied extensively in Russia and Eastern Europe for immune restoration, particularly in elderly patients and those recovering from radiation exposure [52].
Both peptides have thymic origins and immune-modulating properties, but they differ in composition, mechanism, and clinical evidence base. Thymulin is a single defined peptide with a clear structure-activity relationship. Thymalin is a complex extract whose active components are not fully characterized. For those interested in Khavinson's peptide bioregulator research, our detailed guide covers the broader range of thymic peptide research.
Thymulin in Practice
Thymulin's clinical use is less established than Ta1's, with most of the evidence coming from animal studies and small human trials. In clinical practice, thymulin is sometimes used as part of immune optimization protocols, particularly in individuals with documented thymic decline or zinc deficiency.
| Approach | Strategy | Rationale |
|---|---|---|
| Thymulin + Zinc | Co-administer zinc (15-30 mg elemental zinc daily) with thymulin | Ensures thymulin exists in active zinc-bound form |
| Thymulin + Ta1 | Combine thymulin with Thymosin Alpha-1 | Dual thymic peptide approach for maximal thymic output support |
| Thymulin for aging | Low-dose thymulin as anti-aging strategy | Counters age-related thymic involution and immunosenescence |
| Thymulin + Epithalon | Combine with telomerase-activating peptide | Address multiple axes of immune aging |
Given the current evidence base, thymulin is best considered a supportive peptide within the immune stack rather than a standalone intervention. Its value lies in complementing Ta1's immune-restoring effects by providing additional thymic hormone signaling, particularly in individuals with compromised thymic function or zinc status.
Stacking Protocols: Combining Immune Peptides for Maximum Effect
The rationale for stacking immune peptides rests on a simple principle: the immune system operates through multiple interconnected pathways, and targeting several of them simultaneously produces effects greater than any single intervention. Each peptide in this stack addresses a different layer of immune defense, creating redundancy and coverage that a single peptide can't match.
Why Stack Rather Than Use Individual Peptides?
The four peptides in this stack have minimal mechanistic overlap and complementary actions:
- Ta1 works upstream - at the level of dendritic cells, T cells, and NK cells. It enhances the immune system's ability to detect threats, mount appropriate responses, and maintain surveillance. Think of it as upgrading the command-and-control infrastructure.
- LL-37 works at the front line - directly killing pathogens, disrupting biofilms, and recruiting immune cells to infection sites. It's the infantry that engages enemies on contact.
- KPV works as the brake system - preventing inflammatory responses from spiraling out of control and causing tissue damage. It ensures the immune system fights effectively without destroying the territory it's trying to protect.
- Thymulin works as hormonal support - maintaining the thymic signaling environment that allows T-cell development and maturation to continue despite aging. It's the supply line that keeps new immune soldiers coming.
When these four peptides are used together, they create a comprehensive immune optimization strategy that addresses detection (Ta1), elimination (LL-37), damage control (KPV), and replenishment (thymulin). No single peptide achieves all four objectives.
Composite Immune Function Improvement: Individual vs. Combined Stack
Based on composite scoring across T-cell counts, NK cell activity, inflammatory markers, and antimicrobial peptide levels. Data derived from individual peptide studies; combined stack values are theoretical projections based on additive and complementary mechanisms.
Protocol Tiers: From Foundation to Advanced
Not everyone needs (or should start with) the full four-peptide stack. The following tiered approach allows gradual introduction based on individual needs, budget, and response.
Tier 1: Foundation Protocol - Ta1 Alone
| Peptide | Dose | Frequency | Duration |
|---|---|---|---|
| Thymosin Alpha-1 | 1.6 mg SC | 2x weekly (Mon/Thu) | 8-12 weeks |
Best for: General immune support, anti-aging immune maintenance, post-illness recovery, vaccine response enhancement. Start here if you're new to immune peptides. Ta1 has the deepest evidence base and the best-characterized safety profile. Run for at least 8 weeks before assessing response through blood work (see monitoring section). This protocol alone may be sufficient for individuals with mild immune decline or those seeking preventive immune support.
Tier 2: Enhanced Protocol - Ta1 + LL-37
| Peptide | Dose | Frequency | Duration |
|---|---|---|---|
| Thymosin Alpha-1 | 1.6 mg SC | 2x weekly (Mon/Thu) | 8-12 weeks |
| LL-37 | 100-200 mcg SC | Daily | 8-12 weeks |
Best for: Recurrent infections, chronic sinusitis, suspected biofilm infections, mold exposure recovery, post-antibiotic immune rebuilding. The addition of LL-37 provides direct antimicrobial defense that complements Ta1's immune cell activation. Consider this tier if you're dealing with active infections or have a history of antibiotic overuse that may have disrupted microbial balance.
Tier 3: Full Immune Stack - Ta1 + LL-37 + KPV
| Peptide | Dose | Frequency | Duration |
|---|---|---|---|
| Thymosin Alpha-1 | 1.6 mg SC | 2x weekly (Mon/Thu) | 8-12 weeks |
| LL-37 | 100-200 mcg SC | Daily | 8-12 weeks |
| KPV | 500-1000 mcg oral or 200-500 mcg SC | Daily | 8-12 weeks |
Best for: Chronic inflammation, autoimmune tendencies, gut inflammation (IBS, IBD), inflammatory skin conditions, chronic fatigue with elevated inflammatory markers. Adding KPV brings the anti-inflammatory dimension to the stack. This is particularly valuable for individuals whose immune dysfunction manifests as excessive or misdirected inflammation rather than (or in addition to) impaired pathogen defense.
Tier 4: Advanced Protocol - Full Stack + Thymulin
| Peptide | Dose | Frequency | Duration |
|---|---|---|---|
| Thymosin Alpha-1 | 1.6 mg SC | 2x weekly (Mon/Thu) | 12 weeks |
| LL-37 | 200 mcg SC | Daily | 12 weeks |
| KPV | 500-1000 mcg oral or 200-500 mcg SC | Daily | 12 weeks |
| Thymulin + Zinc (30mg) | Per product instructions | Daily | 12 weeks |
Best for: Significant age-related immune decline (60+), documented lymphocytopenia, history of chemotherapy or radiation, severe immunosuppression. The addition of thymulin provides a second thymic peptide to address age-related thymic involution from a hormonal angle, complementing Ta1's receptor-mediated immune activation. Always co-administer with zinc to ensure thymulin remains in its active form.
Stacking Strategy
When stacking multiple peptides, introduce one at a time with at least 5-7 days between additions. This allows you to identify which peptide, if any, causes adverse effects and to gauge individual responses. Start with Ta1 (most evidence, best safety profile), then add LL-37, then KPV, and finally thymulin if desired. Keep a log of subjective symptoms, energy levels, and any injection site reactions during the introduction period.
Situational Protocols
Acute Illness Response Protocol
When you feel an infection coming on or have confirmed exposure to a pathogen:
| Peptide | Dose | Frequency | Duration |
|---|---|---|---|
| Ta1 | 1.6 mg SC | Daily x 5-7 days | Then revert to 2x weekly |
| LL-37 | 200-400 mcg SC | Daily | 7-14 days |
| KPV | 1000 mcg oral | 2x daily | 7-14 days |
This front-loaded approach uses higher frequencies (particularly for Ta1) during the acute phase to rapidly boost immune activation and antimicrobial defense. Ta1's daily dosing during acute illness is supported by sepsis trial protocols where daily or twice-daily dosing was safe and effective. KPV at higher doses helps manage the inflammatory response that accompanies active infection.
Seasonal Immune Preparation Protocol
For proactive immune support during cold and flu season (typically October through March in the Northern Hemisphere):
| Peptide | Dose | Frequency | Duration |
|---|---|---|---|
| Ta1 | 1.6 mg SC | 2x weekly | October through March |
| LL-37 | 100 mcg SC | 3x weekly | October through March |
| KPV | 500 mcg oral | Daily | October through March |
This lower-intensity maintenance protocol provides continuous immune support through the peak respiratory virus season. It can be combined with foundational supplements (vitamin D 5000 IU, zinc 15-30 mg, vitamin C 1-2g) and standard preventive measures (hand hygiene, adequate sleep, stress management).
Post-Antibiotic Recovery Protocol
After a course of antibiotics, the immune system and microbiome both need rebuilding:
| Peptide | Dose | Frequency | Duration |
|---|---|---|---|
| Ta1 | 1.6 mg SC | 2x weekly | 4-8 weeks post-antibiotics |
| KPV | 500-1000 mcg oral | Daily | 4-8 weeks |
Post-antibiotic recovery focuses on immune rebuilding (Ta1) and gut inflammation management (KPV). LL-37 is generally not needed here since the infection requiring antibiotics has been treated. KPV's gut-targeting properties make it especially relevant for supporting mucosal recovery. Combine with quality probiotics (Lactobacillus, Bifidobacterium, Saccharomyces boulardii) and prebiotic fiber.
Travel Immune Protocol
For those traveling to areas with increased infection risk:
| Timing | Peptide | Dose | Frequency |
|---|---|---|---|
| 2 weeks before travel | Ta1 | 1.6 mg SC | 3x that week, then 2x weekly |
| During travel | LL-37 | 100-200 mcg SC | Daily |
| During travel | KPV | 500 mcg oral | Daily (especially for GI protection) |
| 1 week after return | Ta1 | 1.6 mg SC | 2x weekly, then discontinue |
This protocol front-loads Ta1 before travel to prime immune responses, adds LL-37 and KPV during the exposure period, and includes a brief post-travel taper to address any infections acquired during the trip. KPV oral administration is particularly valuable for travelers heading to areas where GI infections (traveler's diarrhea) are common.
Timing Considerations Within the Stack
When injecting multiple peptides on the same day, spacing and site rotation matter:
- Inject different peptides at different sites. Use the left abdomen for Ta1 and the right abdomen for LL-37, for example. This prevents local interactions between peptides and makes it easier to identify which peptide causes any injection site reaction.
- Morning vs. evening timing. Ta1 is often administered in the morning to align with natural cortisol rhythms and immune cell activity patterns. LL-37 can be given at any time. KPV oral dosing is often taken with breakfast or on an empty stomach for GI targeting.
- Avoid injecting immediately before intense exercise. Heavy exercise transiently suppresses immune function (the "open window" phenomenon). Injecting immune peptides during this immunosuppressive period may reduce their effectiveness.
- Consider timing around sleep. Growth hormone, which supports immune function and tissue repair, is released primarily during deep sleep. Some practitioners prefer evening dosing of certain peptides to coincide with this natural anabolic window.
Figure 6: Sample weekly schedule for the full immune peptide stack showing injection days, oral dosing, and recommended timing windows.
Stacking with Non-Immune Peptides
Immune peptides can be combined with other functional peptide categories for comprehensive health optimization. Common co-administered peptides include:
- BPC-157: For tissue repair and gut healing. BPC-157 and KPV together provide powerful gut support - BPC-157 through angiogenesis and nitric oxide modulation, KPV through NF-kB inhibition and mucosal barrier protection.
- TB-500: For tissue repair and anti-fibrotic effects. TB-500 (Thymosin Beta-4 fragment) shares a thymic origin with Ta1 but has completely different functions focused on cell migration and wound healing.
- Selank: For stress management and cognitive function. Since chronic stress is one of the most potent suppressors of immune function, addressing stress with Selank can enhance the effectiveness of immune peptide protocols.
- NAD+: For cellular energy and DNA repair. NAD+ levels decline with age and are required for sirtuin activation, which influences both metabolic health and immune cell function.
- VIP (Vasoactive Intestinal Peptide): For mold illness recovery, CIRS, and mucosal immune regulation. VIP and the immune peptide stack address CIRS from complementary angles - VIP modulates the hypothalamic-pituitary axis and regulatory T cells, while Ta1 and LL-37 address the pathogen clearance and immune activation components.
For a complete overview of available research peptides and their applications, visit our peptide research hub or use the dosing calculator for personalized protocol planning.
Timing, Cycling, and Periodization Strategies
Running immune peptides continuously at maximum dose isn't always the best strategy. Just as strength training benefits from periodization - alternating between high-intensity and recovery phases - immune peptide protocols benefit from strategic cycling that prevents receptor desensitization, maintains efficacy, and reduces cost without sacrificing results.
Why Cycling Matters
Continuous exposure to any signaling molecule can lead to receptor downregulation - the cell produces fewer receptors or reduces their sensitivity in response to persistent stimulation. While Ta1 has been used continuously for 6-12 months in hepatitis trials without documented loss of efficacy, the precautionary principle suggests that periodic breaks may preserve long-term responsiveness [53].
Cost is another practical consideration. A full four-peptide stack can represent a significant monthly investment. Cycling allows individuals to capture the majority of benefits while reducing overall expenditure.
There's also an immunological rationale for cycling. The immune system naturally oscillates between states of heightened vigilance and recovery. Chronic immune activation - even when therapeutically intended - can lead to immune fatigue, where effector cells become exhausted and regulatory mechanisms overshoot. Periodic breaks allow the immune system to return to baseline and consolidate the improvements gained during active treatment.
Cycling Frameworks
Framework 1: Monthly On/Off Cycle
The simplest cycling approach alternates between active treatment months and rest months:
- Weeks 1-8: Full peptide protocol (all peptides at standard doses)
- Weeks 9-12: Off period (no peptides; maintain foundational supplements)
- Repeat
This 8-on/4-off pattern provides two full cycles within a 6-month period. It's straightforward to follow and allows clear comparison of how you feel on vs. off the protocol.
Framework 2: Seasonal Cycling
Align peptide use with seasonal infection risk patterns:
- October through March: Full immune peptide protocol (coincides with cold/flu season)
- April through September: Off period or maintenance-only (Ta1 at reduced frequency)
This approach concentrates resources during the period of highest infection risk and allows the immune system to operate independently during warmer months when pathogen exposure is typically lower.
Framework 3: Stepwise Taper Cycling
Rather than abruptly stopping all peptides, this approach gradually reduces frequency:
- Weeks 1-8: Full protocol (standard doses and frequencies)
- Weeks 9-10: Reduce frequency by 50% (e.g., Ta1 from 2x/week to 1x/week; LL-37 from daily to every other day)
- Weeks 11-12: Reduce to once weekly for each peptide
- Weeks 13-16: Off period
- Repeat from week 1
The taper approach avoids any potential "rebound" from abrupt cessation and may be preferable for individuals who notice symptom return when stopping peptides suddenly.
Framework 4: Rotating Single-Peptide Emphasis
For budget-conscious individuals or those who want to assess individual peptide contributions:
- Month 1-2: Ta1 alone (establish baseline immune enhancement)
- Month 3-4: Ta1 + LL-37 (add antimicrobial layer)
- Month 5-6: Ta1 + KPV (replace LL-37 with anti-inflammatory focus)
- Month 7-8: Full stack (all four peptides)
- Month 9: Off period
This rotation allows you to identify which peptides provide the most noticeable benefit and to build a protocol tailored to your specific needs.
Special Timing Considerations
Around Vaccines
Ta1 has been studied as a vaccine adjuvant and may enhance vaccine responses in poor responders. If you're receiving a vaccine (flu, COVID, pneumonia, etc.), consider:
- Starting Ta1 one week before vaccination if not already using it
- Continuing Ta1 for 2-4 weeks after vaccination to support memory cell formation
- Avoiding LL-37 on the day of vaccination (theoretical concern about interaction with vaccine antigens at the injection site)
- KPV can be continued through vaccination without concerns
During Active Illness
If you develop an acute infection while on a maintenance protocol, shift to the acute illness response protocol (see stacking section). Key adjustments:
- Increase Ta1 to daily dosing for the first 5-7 days
- Increase LL-37 dose if tolerated (up to 400 mcg daily)
- Increase KPV if inflammation is prominent (fever, body aches)
- Return to maintenance protocol after symptom resolution
With Antibiotics
If you need antibiotics during a peptide protocol:
- Continue Ta1 - it supports immune function that antibiotics don't address
- LL-37 and antibiotics work through different mechanisms and can be used concurrently. LL-37 may even enhance antibiotic efficacy against biofilm infections
- KPV becomes especially valuable post-antibiotics for managing gut inflammation and supporting mucosal recovery
Exercise Timing
Intense exercise causes a transient immunosuppressive period lasting 3-72 hours (the "open window" hypothesis). While this effect is now considered more nuanced than originally proposed, timing peptide injections around exercise may optimize their effects:
- Inject Ta1 on rest days or at least 2 hours before or after intense training
- LL-37 timing relative to exercise is less critical due to its direct antimicrobial mechanism
- KPV may actually be beneficial post-exercise to manage exercise-induced inflammation
Duration and Long-Term Planning
How long should you run immune peptide protocols? The answer depends on your goals:
| Goal | Recommended Duration | Cycling Approach |
|---|---|---|
| Acute illness recovery | 2-4 weeks | No cycling needed - discontinue after recovery |
| Post-infection immune rebuilding | 8-12 weeks | Single course, then reassess |
| Seasonal immune support | 5-6 months annually | Seasonal cycling (Oct-Mar) |
| Age-related immune decline | Ongoing with cycling | 8 on/4 off, or seasonal |
| Autoimmune modulation | 12+ weeks initial, then reassess | Based on labs and symptoms |
| Cancer immunotherapy support | Throughout treatment + 3 months after | Continuous during treatment, then taper |
For ongoing anti-aging immune support, many practitioners recommend annual cycles - 2-3 treatment periods of 8-12 weeks each, distributed across the year. This provides periodic immune system "tune-ups" without continuous peptide exposure. Blood work before and after each cycle helps track objective improvements and guide future protocol adjustments.
The Gut-Immune Axis: Where KPV and LL-37 Shine
The gastrointestinal tract isn't just a digestive organ - it's the body's largest immune organ. Roughly 70-80% of all immune cells reside in gut-associated lymphoid tissue (GALT), and the intestinal barrier represents the most extensive interface between the body's internal environment and external antigens. Understanding the gut-immune connection explains why KPV and LL-37 are such valuable additions to an immune optimization stack.
Intestinal Barrier Function
The intestinal epithelium is a single-cell-thick layer that must simultaneously absorb nutrients and block pathogens. This seemingly impossible task is managed through tight junction proteins (claudins, occludins, zonula occludens) that create selective gates between epithelial cells. When these tight junctions are compromised - by inflammation, stress, certain medications, alcohol, or pathogenic bacteria - the barrier becomes permeable to bacterial products that trigger systemic immune activation.
Lipopolysaccharide (LPS) translocation through a compromised gut barrier is now recognized as a major driver of chronic systemic inflammation. Even small increases in circulating LPS activate TLR4 on macrophages and other immune cells, driving production of TNF-a, IL-6, and IL-1b. This "metabolic endotoxemia" has been implicated in obesity-associated inflammation, insulin resistance, non-alcoholic fatty liver disease, and cardiovascular disease.
KPV addresses intestinal barrier dysfunction at multiple levels. By inhibiting NF-kB in intestinal epithelial cells, KPV reduces the inflammatory signaling that disrupts tight junction protein expression. In colitis models, KPV treatment preserved mucosal architecture - including tight junction integrity - significantly better than control animals. The oral route of KPV administration means it reaches the gut epithelium directly, providing local anti-inflammatory effects right where they're needed most.
LL-37 contributes to gut barrier defense through its antimicrobial activity against enteric pathogens. Infections by Salmonella, Shigella, Campylobacter, and pathogenic E. coli strains can all disrupt gut barrier function. By killing or inhibiting these pathogens at the mucosal surface, LL-37 prevents the barrier disruption that would otherwise follow. LL-37 also promotes wound healing in damaged epithelium, supporting barrier restoration after injury.
Secretory IgA and Mucosal Immunity
Secretory IgA (sIgA) is the most abundant antibody in the human body and the primary antibody at mucosal surfaces. sIgA coats commensal bacteria (preventing them from triggering inflammatory responses), neutralizes toxins and viruses at mucosal surfaces, and prevents microbial adhesion to epithelial cells. Low sIgA levels are associated with increased mucosal infections, food sensitivities, and inflammatory bowel conditions.
Ta1's enhancement of B-cell function and antibody production may support sIgA levels, particularly in individuals with documented sIgA deficiency. While specific studies on Ta1 and sIgA are limited, its broad effects on adaptive immunity - including B-cell activation and differentiation - provide a plausible mechanism for mucosal antibody support.
The Microbiome-Immunity Interface
The gut microbiome profoundly influences immune function through multiple mechanisms:
- Short-chain fatty acid (SCFA) production: Commensal bacteria ferment dietary fiber to produce butyrate, propionate, and acetate. These SCFAs support intestinal barrier integrity, promote regulatory T-cell differentiation, and have anti-inflammatory effects through HDAC inhibition and GPR41/GPR43 activation.
- Competitive exclusion: A healthy commensal community occupies ecological niches that would otherwise be available to pathogens. Antibiotic disruption of commensals creates opportunities for pathogenic colonization.
- Immune education: Commensal bacteria train the immune system to distinguish between harmless microbes and genuine threats. This education occurs primarily in early life but continues throughout adulthood.
- Antimicrobial peptide induction: Certain commensal bacteria stimulate epithelial production of antimicrobial peptides, including LL-37 and defensins, which help maintain mucosal defense.
KPV's selective anti-inflammatory action is particularly valuable in the microbiome context. Unlike broad immunosuppressants (corticosteroids, conventional immunomodulators) that can impair antimicrobial defense and allow pathogenic overgrowth, KPV reduces pathological inflammation while preserving the antimicrobial peptide production and immune surveillance that keep the microbiome in balance.
Practical Gut-Immune Protocol
For individuals with documented gut inflammation, food sensitivities, or chronic GI symptoms, the following protocol targets the gut-immune axis specifically:
| Component | Dose | Timing | Duration | Purpose |
|---|---|---|---|---|
| KPV oral | 500-1000 mcg | Morning, empty stomach | 8-12 weeks | Local gut anti-inflammatory |
| Ta1 SC | 1.6 mg | Mon/Thu mornings | 8-12 weeks | Systemic immune enhancement |
| LL-37 SC | 100-200 mcg | Daily | 8-12 weeks (optional) | Antimicrobial support |
| BPC-157 oral or SC | 250-500 mcg | Twice daily | 8-12 weeks | Gut mucosal healing |
| Probiotics | Multi-strain, 50B CFU | With meals | Ongoing | Microbiome restoration |
| L-Glutamine | 5-10 g | Daily | Ongoing | Enterocyte fuel source |
| Zinc carnosine | 75 mg | Twice daily | 8 weeks | Gastric and intestinal mucosal repair |
The combination of KPV (anti-inflammatory) with BPC-157 (mucosal healing) addresses gut dysfunction from both the inflammatory and structural sides. Adding Ta1 provides systemic immune support that indirectly benefits gut immunity through enhanced T-cell and dendritic cell function in GALT. This combined approach is more comprehensive than any single intervention for chronic gut-immune dysfunction.
Research Note
The PepT1 transporter that mediates KPV uptake is also expressed on immune cells, including macrophages and dendritic cells. This means KPV can enter and modulate immune cells directly, in addition to its effects on epithelial cells. The PepT1 expression on immune cells in the lamina propria (the connective tissue layer beneath the intestinal epithelium) provides another route through which KPV can reduce gut inflammation - by directly suppressing NF-kB in the inflammatory macrophages that drive IBD pathology.
Immune Peptides and Cancer Immunosurveillance
The immune system continuously monitors for and eliminates transformed (pre-cancerous and cancerous) cells - a process called immune surveillance. When immune surveillance fails, cancers can develop. While immune peptides are not cancer treatments, their ability to enhance immune surveillance mechanisms has attracted research interest as a complement to conventional cancer therapies and potentially as a preventive strategy.
How Immune Surveillance Works
Cancer immunosurveillance involves multiple immune cell types working together:
- NK cells detect and kill cells that have lost MHC class I expression - a common immune evasion strategy used by tumor cells. NK cells also respond to stress ligands expressed on transformed cells.
- CD8+ cytotoxic T cells recognize tumor-specific antigens presented on MHC class I molecules. This is the primary mechanism of adaptive antitumor immunity.
- CD4+ Th1 cells support CD8+ T-cell activation and macrophage-mediated tumor killing through IFN-g production.
- Macrophages can phagocytose tumor cells and present tumor antigens to T cells, though tumor-associated macrophages (TAMs) can also suppress antitumor immunity.
- Dendritic cells capture tumor antigens and present them to T cells, initiating the adaptive antitumor response.
Ta1 enhances nearly every component of this surveillance network. Its activation of dendritic cells improves tumor antigen presentation. Its enhancement of NK cell cytotoxicity provides more effective recognition of MHC-deficient tumor cells. Its promotion of CD8+ T-cell function strengthens the adaptive antitumor response. And its expansion of regulatory T cells helps prevent the chronic inflammation that can paradoxically promote tumor growth.
Ta1 in Cancer Clinical Research
Ta1 has been studied as an adjunct to cancer therapy in several settings:
- Hepatocellular carcinoma (HCC): Ta1 combined with transarterial chemoembolization improved overall survival and recurrence-free survival compared to chemoembolization alone. The proposed mechanism involves preservation of immune function during the immunosuppressive period following chemotherapy.
- Non-small-cell lung cancer (NSCLC): Ta1 added to platinum-based chemotherapy improved response rates and quality of life. Patients receiving Ta1 had fewer infectious complications during chemotherapy - consistent with its immune-preserving effects.
- Melanoma: Ta1 has been studied as an adjunct to dacarbazine and interferon-a in advanced melanoma, with some evidence of improved response rates.
- Checkpoint inhibitor combination: There's emerging interest in combining Ta1 with immune checkpoint inhibitors (anti-PD-1, anti-CTLA-4), which work by releasing the brakes on antitumor T-cell responses. Ta1 could potentially enhance the T-cell activation that checkpoint inhibitors unleash, though clinical data on this specific combination is limited.
LL-37 also has documented antitumor activity in preclinical models. It can induce apoptosis (programmed cell death) in certain cancer cell lines, inhibit tumor angiogenesis, and enhance NK cell-mediated tumor killing. However, the relationship between LL-37 and cancer is complex - in some contexts, LL-37 may promote tumor growth through its pro-angiogenic and cell proliferation effects. This complexity means LL-37 should not be considered a cancer therapy but rather a component of general immune support that may contribute to improved surveillance.
Important Disclaimer
Immune peptides are not cancer treatments. They should never be used as substitutes for conventional cancer therapy (surgery, chemotherapy, radiation, immunotherapy). The research discussed here pertains to Ta1 as an adjunct to established cancer treatments, under medical supervision, in the context of clinical trials. Individuals with cancer should discuss any peptide use with their oncologist.
Preventive Immune Surveillance Support
For individuals without cancer who want to maintain strong immune surveillance as a preventive strategy - particularly those over 50 where cancer risk increases and immune function declines - the standard immune peptide stack provides relevant support:
- Ta1 enhances NK cell function and T-cell surveillance
- KPV reduces chronic inflammation that promotes cancer development and progression
- LL-37 provides antimicrobial support that prevents chronic infections (some of which are linked to cancer risk - H. pylori and gastric cancer, HBV/HCV and liver cancer, HPV and cervical cancer)
- Thymulin supports ongoing T-cell maturation and the diversity of the T-cell receptor repertoire needed to recognize tumor neoantigens
This preventive approach should be viewed as one component of comprehensive cancer risk reduction that includes regular screening, healthy diet, exercise, stress management, adequate sleep, and avoidance of known carcinogens.
Immune Peptides vs. Alternative Approaches
Immune peptides don't exist in a vacuum. They're one category of intervention in a broad range of immune support options ranging from simple supplements to prescription biologics. Understanding where peptides fit helps make informed decisions about when they're worth the investment and when simpler approaches might suffice.
Peptides vs. Standard Supplements
| Approach | Mechanism | Evidence Level | Cost | Best For |
|---|---|---|---|---|
| Vitamin D (5000 IU) | LL-37 induction, T-cell activation, Treg support | Strong (RCTs) | Low ($0.05/day) | Everyone, especially deficient individuals |
| Zinc (15-30 mg) | Thymulin activation, T-cell development, NF-kB regulation | Strong (RCTs) | Low ($0.10/day) | Everyone, especially elderly |
| Vitamin C (1-2 g) | Antioxidant, neutrophil function, barrier integrity | Moderate | Low ($0.15/day) | General immune support |
| Elderberry extract | Possible viral entry inhibition | Weak (small trials) | Low ($0.30/day) | Cold/flu season support |
| Thymosin Alpha-1 | TLR activation, DC maturation, T/NK enhancement | Strong (100+ trials) | Moderate-High | Immune decline, chronic infection, cancer adjunct |
| LL-37 | Direct antimicrobial, biofilm disruption, immune recruitment | Moderate (preclinical + small clinical) | Moderate | Chronic infections, biofilm conditions |
| KPV | NF-kB inhibition via PepT1 | Moderate (preclinical strong) | Moderate | Gut inflammation, autoimmune considerations |
The key distinction is that supplements like vitamin D and zinc support the molecular machinery that peptides target. Vitamin D upregulates LL-37 expression; zinc activates thymulin. But the peptides themselves provide a more direct and powerful intervention at the immune cell level. For individuals with documented immune dysfunction, the peptide approach offers specificity and potency that supplements alone can't match.
The practical recommendation is a layered approach: ensure foundational nutrients are optimized first (vitamin D, zinc, vitamin C, omega-3s), then add immune peptides if additional support is needed or desired. Running expensive peptide protocols while being vitamin D deficient or zinc depleted is like putting premium fuel in a car with flat tires - you're missing the foundation that makes the premium intervention work properly.
Peptides vs. Prescription Immunomodulators
For individuals with significant immune dysfunction, the comparison with prescription immunomodulators is relevant:
- Interferons: Type I interferons (IFN-a, IFN-b) are potent antiviral and immunomodulatory agents, but carry significant side effects (depression, flu-like symptoms, autoimmune reactions, cytopenias). Ta1 provides some overlapping immune benefits (enhanced dendritic cell and T-cell function) with far fewer side effects.
- IVIG (Intravenous Immunoglobulin): Used for immunodeficiency and autoimmune conditions, IVIG provides passive antibody replacement. It addresses humoral (antibody) deficiency, which immune peptides don't directly treat. For patients with both cellular and humoral immune deficits, IVIG and Ta1 could theoretically complement each other.
- G-CSF/GM-CSF (Neupogen, Leukine): These growth factors stimulate neutrophil production and are used for chemotherapy-induced neutropenia. They address a specific type of immune deficiency (neutrophil depletion) that immune peptides don't target directly. Ta1 has been studied alongside G-CSF in cancer patients with preliminary positive results.
- Checkpoint inhibitors (anti-PD-1, anti-CTLA-4): These powerful cancer immunotherapy drugs release the brakes on antitumor T-cell responses. They can cause severe autoimmune-like side effects (immune-related adverse events). Ta1's ability to simultaneously enhance T-cell function and expand Tregs might potentially reduce the autoimmune toxicity of checkpoint inhibitors while preserving antitumor efficacy - an active area of research interest.
Immune peptides occupy a unique niche between supplements and pharmaceuticals. They're more targeted and potent than nutritional supplements but less aggressive and less risky than prescription immunomodulators. For the large population of people with mild-to-moderate immune decline - those who aren't sick enough for aggressive pharmaceutical intervention but aren't optimally protected by supplements alone - peptides offer a compelling intermediate option.
Figure 9: Immune support hierarchy - from foundational supplements through immune peptides to prescription immunomodulators, showing the cost-benefit profile at each tier.
When to Consider Immune Peptides
Immune peptides make the most sense for individuals who:
- Experience recurrent infections despite adequate nutrition, sleep, and stress management
- Have documented immune deficiency on blood work (low lymphocytes, inverted CD4:CD8, low NK activity)
- Are over 50 and concerned about age-related immune decline
- Have chronic infections (viral hepatitis, latent infections, chronic sinusitis, recurrent UTIs)
- Are recovering from cancer treatment and want to rebuild immune function
- Have chronic inflammation (elevated hsCRP, ESR) that isn't responding to lifestyle interventions alone
- Have autoimmune conditions and want targeted immune modulation rather than broad immunosuppression
- Are preparing for increased infection exposure (travel, flu season, healthcare work)
Immune peptides are less necessary for young, healthy individuals with no immune complaints, normal blood work, and adequate foundational nutrition. For this population, maintaining vitamin D, zinc, and healthy lifestyle habits provides sufficient immune support for most situations.
For those ready to explore immune peptide protocols, FormBlends' getting started guide provides orientation on product selection, and the dosing calculator helps plan specific protocols based on individual goals and experience level.
Stress, Sleep, and Immune Function: The Overlooked Variables
No peptide protocol can fully compensate for the immune-wrecking effects of chronic stress and sleep deprivation. These two factors represent the most potent and common suppressors of immune function in modern life - and understanding their mechanisms explains why addressing them must accompany any peptide protocol for optimal results.
The Cortisol-Immune Connection
The hypothalamic-pituitary-adrenal (HPA) axis links psychological stress directly to immune suppression. When the brain perceives a threat (whether physical danger or a looming work deadline), the hypothalamus releases corticotropin-releasing hormone (CRH), which stimulates pituitary ACTH secretion, which in turn drives adrenal cortisol production. Cortisol, while essential for survival in acute stress, has profound immunosuppressive effects when chronically elevated:
- Lymphocyte redistribution: Cortisol drives lymphocytes out of the bloodstream and into tissues, creating apparent lymphocytopenia on blood tests. This redistribution may actually represent an adaptive response (moving immune cells to likely infection sites during physical danger), but chronic elevation disrupts normal immune cell trafficking patterns.
- T-cell suppression: Chronic cortisol exposure reduces T-cell proliferation, cytokine production, and cytotoxic function. Both CD4+ and CD8+ T cells are affected, and the Th1/Th2 balance shifts toward Th2 dominance - reducing cell-mediated immunity (needed for viral and intracellular pathogen defense) while potentially increasing allergic and autoimmune tendencies.
- NK cell impairment: Cortisol decreases NK cell cytotoxicity, reducing the body's first-line defense against virus-infected and tumor cells. Studies of chronically stressed populations (caregivers, medical residents, grieving individuals) consistently show reduced NK cell function.
- Thymic involution acceleration: Chronic stress hormones accelerate thymic involution, compounding the age-related decline in T-cell production that Ta1 and thymulin aim to address. In animal studies, chronic stress can cause thymic atrophy within weeks.
- Inflammatory paradox: While cortisol is anti-inflammatory in acute settings, chronic cortisol exposure can paradoxically increase systemic inflammation through glucocorticoid receptor desensitization. Immune cells become resistant to cortisol's suppressive effects, leading to unrestrained NF-kB activation and pro-inflammatory cytokine production - exactly the situation that KPV targets.
For individuals under chronic stress, addressing the stress itself (through meditation, therapy, lifestyle modifications, adaptogenic herbs, or anxiolytic peptides like Selank) may improve immune function as much as adding immune peptides. The ideal approach is both: reduce stress while simultaneously supporting immune function with targeted peptides.
Sleep and Immune Restoration
Sleep isn't just recovery time - it's an active period of immune system maintenance. During deep (slow-wave) sleep, the body releases growth hormone, reduces cortisol levels, and shifts the T-cell cytokine profile toward Th1 (pro-inflammatory, anti-pathogen) from the Th2 bias of daytime hours. This nocturnal immune activation is why fever and many immune symptoms often worsen at night - the immune system is more active during sleep.
Sleep deprivation - even partial sleep restriction (6 hours instead of 8 for one week) - produces measurable immune deficits:
- Reduced vaccine antibody responses (documented for influenza, hepatitis A, and hepatitis B vaccines)
- Increased susceptibility to rhinovirus infection (the common cold)
- Decreased NK cell activity
- Elevated inflammatory markers (CRP, IL-6)
- Altered T-cell cytokine production
- Impaired antigen-specific T-cell memory formation
The sleep-immune connection has practical implications for peptide timing. Ta1 administered in the evening, before the natural nocturnal immune activation, may enhance its effectiveness by providing TLR-mediated immune priming during the period when the body is already geared toward immune activity. While formal studies on Ta1 timing haven't been published, the circadian biology of immunity supports the logic of evening or pre-sleep dosing.
For individuals with sleep disruption, optimizing sleep hygiene (consistent schedule, dark/cool bedroom, limiting screens before bed, managing caffeine timing) should be prioritized alongside peptide protocols. Selank, with its anxiolytic and sleep-supportive properties, may serve double duty as both a stress management tool and an indirect immune support through improved sleep quality.
Exercise: The Double-Edged Immune Sword
Moderate regular exercise is one of the most potent immune enhancers available - it increases NK cell circulation, improves neutrophil function, enhances vaccine responses, and reduces chronic inflammation. However, the relationship between exercise and immunity follows a J-curve: moderate exercise improves immunity, while excessive or prolonged intense exercise can suppress it.
The "open window" theory proposes that intense exercise creates a temporary period (3-72 hours) of reduced immune function during which infection risk increases. While recent research has moderated this theory (suggesting that immune cell redistribution rather than true suppression explains most post-exercise changes), athletes and heavy exercisers do experience higher rates of upper respiratory infections during intensive training blocks.
Practical implications for peptide timing around exercise:
- Moderate exercisers (150-300 min/week): No specific timing adjustments needed. The immune benefits of moderate exercise complement peptide protocols.
- Intense exercisers/athletes: Consider scheduling Ta1 injections on rest days rather than heavy training days. LL-37 dosing is less affected by exercise timing. KPV may actually benefit post-exercise recovery by managing exercise-induced inflammation.
- Marathon/ultramarathon runners: These individuals may benefit most from immune peptide support, particularly during heavy training blocks and in the weeks surrounding competitive events. Ta1's immune-preserving effects could help prevent the performance-limiting infections common during peak training periods.
The Integrated Approach: Peptides Within a Lifestyle Framework
The most effective immune optimization strategy integrates peptides within a comprehensive lifestyle framework. Each element supports and amplifies the others:
| Pillar | Intervention | Immune Mechanism | Interaction with Peptides |
|---|---|---|---|
| Sleep | 7-9 hours nightly, consistent schedule | GH release, T-cell restoration, cytokine regulation | Enhances nocturnal immune activation that peptides prime |
| Stress management | Daily practice (meditation, breathing, therapy) | Reduces cortisol-mediated immunosuppression | Prevents cortisol from blunting peptide effects |
| Exercise | 150-300 min/week moderate intensity | NK cell enhancement, anti-inflammatory, improved circulation | Complements Ta1's NK cell and T-cell effects |
| Nutrition | Whole foods, adequate protein, fiber diversity | Micronutrient availability, microbiome support | Provides zinc for thymulin, vitamin D for LL-37 |
| Supplementation | D3, zinc, vitamin C, omega-3s | Cofactor support for immune pathways | Directly supports peptide mechanisms (D3 -> LL-37, Zn -> thymulin) |
| Peptide protocol | Ta1, LL-37, KPV, thymulin (as needed) | Targeted immune cell modulation | Provides the highest-level intervention on top of lifestyle foundation |
This integrated framework means that immune peptides are most effective when used by individuals who are already doing the basics right. A person sleeping 8 hours, managing stress, exercising moderately, eating well, and taking foundational supplements will get more benefit from adding Ta1 than someone who sleeps 5 hours, is chronically stressed, sedentary, and eating poorly. The peptides are the capstone of the pyramid, not the foundation.
Bottom Line
The ideal immune optimization strategy starts with sleep, stress management, nutrition, and exercise. Layer in foundational supplements (vitamin D, zinc, vitamin C, omega-3s). Then add immune peptides for individuals who need additional support - those with documented immune decline, chronic infections, cancer treatment recovery, or age-related immunosenescence. This layered approach provides the most comprehensive and cost-effective path to immune resilience.
Future Directions in Immune Peptide Research
The field of immune peptide research is evolving rapidly, with several promising developments that could expand the applications and effectiveness of the peptides discussed in this report.
Next-Generation LL-37 Derivatives
One of the main limitations of LL-37 is its susceptibility to proteolytic degradation in vivo, which limits its bioavailability and duration of action. Researchers are developing modified LL-37 analogs that retain antimicrobial and immunomodulatory activity while resisting enzymatic breakdown. These modifications include D-amino acid substitution, cyclization, peptoid backbone modifications, and PEGylation. Some modified LL-37 analogs show 10-100x improved stability while maintaining or improving antimicrobial potency.
Additionally, LL-37 fragment studies have identified shorter peptide sequences (as few as 12-15 amino acids) that retain significant antimicrobial activity. These shorter fragments are cheaper to synthesize, more stable, and potentially less likely to cause the autoimmune complications associated with full-length LL-37. Clinical development of these next-generation cathelicidin-derived peptides could provide more practical therapeutic agents within the next several years.
KPV Delivery Technology
Nanotechnology-based delivery systems for KPV represent a significant advance. PepT1-targeted nanoparticles that encapsulate KPV and deliver it specifically to inflamed colonic tissue are in preclinical development. These systems could dramatically improve KPV's therapeutic index by concentrating the peptide at sites of inflammation while minimizing systemic exposure. The 2024 study on co-assembly nanodrugs combining KPV with immunosuppressants demonstrated proof of concept for this approach.
Oral KPV formulations using enteric coatings and mucoadhesive polymers are also being explored. These delivery systems could improve the consistency and predictability of oral KPV dosing for gut-targeted applications.
Ta1 Combination Immunotherapy
The most exciting frontier for Ta1 research is its combination with immune checkpoint inhibitors in cancer treatment. Checkpoint inhibitors have transformed oncology, but they work in only 20-40% of patients and can cause severe autoimmune side effects. Ta1's ability to simultaneously enhance antitumor T-cell function and expand regulatory T cells makes it a theoretically ideal combination partner - potentially improving checkpoint inhibitor response rates while reducing autoimmune toxicity.
A novel Ta1-Fc fusion protein (linking Ta1 to an antibody Fc domain) has been developed that extends Ta1's half-life from 2 hours to days. This fusion protein showed enhanced antitumor activity in melanoma and breast cancer models, suggesting that sustained Ta1 exposure may be more effective than intermittent dosing for cancer applications. If this technology reaches clinical use, it could transform Ta1 from a twice-weekly injection to a once-every-two-weeks infusion.
Thymic Regeneration Strategies
Rather than replacing thymic hormones (thymulin) or mimicking thymic output (Ta1), some researchers are pursuing direct thymic regeneration. Approaches include growth hormone or IL-7 to stimulate thymic epithelial cell proliferation, sex steroid ablation (surgical or chemical castration) to partially reverse thymic involution, and FOXN1 gene therapy to restore thymic epithelial cell function. A landmark 2019 clinical trial showed that a combination of growth hormone, DHEA, and metformin could partially reverse thymic involution and rejuvenate the immune system in older men - including reduction in epigenetic age.
These thymic regeneration approaches could eventually complement or replace peptide-based thymic support, providing more complete immune rejuvenation than any single peptide can achieve. In the meantime, Ta1 and thymulin remain the most accessible tools for supporting thymic function in aging individuals.
Personalized Immune Peptide Protocols
As immune profiling technology improves and costs decrease, personalized peptide protocols based on individual immune status are becoming more feasible. Advanced immune phenotyping - using flow cytometry to characterize T-cell subsets, functional assays to assess NK cell activity, and cytokine profiling to identify specific inflammatory patterns - can identify which aspects of immune function are most impaired in a given individual. This information can then guide protocol selection:
- Low naive T-cell counts -> prioritize Ta1 for T-cell restoration
- Reduced NK cell cytotoxicity -> emphasize Ta1 dosing and timing
- Elevated inflammatory markers without infection -> prioritize KPV
- Chronic mucosal infections -> add LL-37
- Low zinc + low thymulin activity -> add thymulin with zinc supplementation
This personalized approach is already being adopted by forward-thinking practitioners and represents the future of immune optimization - moving from one-size-fits-all protocols to individualized programs guided by objective immune profiling data.
Figure 10: Future directions in immune peptide research - from next-generation peptide analogs to personalized protocol design based on individual immune profiling.
Regulatory Status and Legal Considerations
The regulatory landscape for immune peptides varies significantly by country, peptide, and intended use. Understanding the legal framework helps users make informed decisions and avoid potential complications.
Thymosin Alpha-1 (Thymalfasin/Zadaxin)
Ta1 occupies a unique regulatory position. It's approved as a prescription drug in over 35 countries - primarily in Asia (China, Philippines, India), South America (Peru, Argentina), and some European countries - for the treatment of hepatitis B, hepatitis C, and as an immune adjuvant in cancer therapy. The commercial product, Zadaxin (thymalfasin), was developed by SciClone Pharmaceuticals and has been marketed since 1996.
In the United States, Ta1 is not FDA-approved as a drug. It was granted Orphan Drug designation by the FDA for the treatment of hepatitis B, active chronic hepatitis B, and DiGeorge syndrome, but never completed the full approval process for these indications. In the current U.S. regulatory environment, Ta1 is available through compounding pharmacies with a prescription and as a research peptide. The FDA's evolving position on compounded peptides may affect availability, making it important to stay informed about regulatory developments. For a detailed overview of peptide regulatory matters, our Ta1 research page maintains current information.
LL-37
LL-37 is not approved as a drug in any country, though it has been used in clinical trials (specifically for wound healing). It's currently available as a research peptide in most jurisdictions. The phase I/II clinical trial for venous leg ulcers demonstrated safety and efficacy, but further clinical development has been limited by challenges including manufacturing costs, stability issues, and the complexity of LL-37's biological effects. For updated information on LL-37 research and availability, see our LL-37 research guide.
KPV
KPV is not approved as a drug and is available as a research peptide. Its simple three-amino-acid structure makes it relatively inexpensive to synthesize and widely available from peptide suppliers. There are no specific regulatory restrictions on KPV beyond the general regulations applying to research peptides in each jurisdiction. Its oral bioavailability makes it one of the more accessible peptides for research use.
Thymulin
Thymulin is not approved as a drug in Western countries. Thymalin - the related but distinct polypeptide extract - is approved in Russia and some former Soviet states for immune restoration. Thymulin is available as a research peptide from specialized suppliers, though it is less widely available than Ta1, LL-37, or KPV.
General Legal Framework
In most Western countries, research peptides exist in a legal gray area. They can be purchased for research purposes but are not approved for human therapeutic use. The distinction between "research" and "therapeutic" use is a legal one, and individuals should understand the regulatory framework in their jurisdiction before purchasing or using peptides.
Working with a healthcare provider who is knowledgeable about peptide therapy provides several advantages: legal prescriptions for available peptides (such as Ta1 through compounding pharmacies), medical oversight for safety monitoring, access to lab work for baseline and follow-up testing, and guidance on protocol design based on individual health status. For guidance on finding qualified providers and starting peptide protocols safely, visit our getting started guide.
Autoimmune Considerations: When the Immune System Needs Recalibration, Not Boosting
Autoimmune conditions present a paradox for immune optimization: the immune system isn't weak - it's misdirected. Using immune peptides in autoimmune patients requires understanding which peptides enhance immune regulation (potentially beneficial) versus which amplify immune activation (potentially harmful).
The Autoimmune Landscape
Autoimmune diseases affect an estimated 5-8% of the population, with higher prevalence in women. Common conditions include rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), multiple sclerosis (MS), Hashimoto's thyroiditis, inflammatory bowel disease (Crohn's and ulcerative colitis), type 1 diabetes, and psoriasis. Each involves the immune system attacking self-tissues, but the specific mechanisms, cell types, and cytokine profiles vary significantly between diseases.
The key question for peptide therapy isn't "will it boost my immune system?" but rather "will it help my immune system distinguish self from non-self more accurately?" This distinction is critical because:
- Th1-dominant autoimmune diseases (RA, MS, type 1 diabetes) involve excessive cell-mediated immunity
- Th2-dominant conditions (asthma, allergies) involve excessive humoral immunity
- Th17-mediated diseases (psoriasis, IBD) involve specific inflammatory pathways
- Some conditions involve loss of regulatory T-cell (Treg) function, which normally suppresses autoreactive T cells
Thymosin Alpha-1 in Autoimmunity
Ta1's role in autoimmune disease is nuanced and supported by emerging research. A 2016 study in Clinical and Experimental Immunology measured serum Ta1 levels in patients with psoriatic arthritis (PsA), rheumatoid arthritis (RA), and systemic lupus erythematosus (SLE). The patients had significantly lower serum Ta1 levels than healthy controls, suggesting that endogenous Ta1 deficiency may contribute to autoimmune dysregulation [54].
Ta1's ability to expand regulatory T cells (Tregs) is particularly relevant for autoimmunity. Tregs are the immune system's primary mechanism for preventing autoreactive T cells from attacking self-tissues. In many autoimmune conditions, Treg numbers or function are reduced. By promoting Treg expansion, Ta1 may help restore immune tolerance rather than simply amplifying overall immune activation.
Pilot studies in lupus and rheumatoid arthritis have reported reduced inflammatory markers and improved joint mobility with Ta1 treatment. A study evaluating Ta1's anti-arthritic properties found promising results, though the evidence base remains small and large-scale randomized trials are lacking [55].
Autoimmune Caution
Despite promising preliminary data, individuals with active autoimmune conditions should approach Ta1 with caution and medical supervision. While Ta1 may enhance immune regulation, its activation of TLR pathways could theoretically exacerbate flares in certain autoimmune conditions. Start at lower doses, monitor symptoms closely, and have relevant autoimmune markers (ANA, anti-CCP, RF, ESR, CRP) checked before and during treatment.
LL-37 and Autoimmunity: A More Complex Picture
LL-37 has a documented role in certain autoimmune conditions - not as a treatment, but as a contributor to pathology. In psoriasis, LL-37 forms complexes with self-DNA released from damaged skin cells. These LL-37/DNA complexes activate plasmacytoid dendritic cells through TLR9, driving production of type I interferons that amplify the psoriatic inflammatory cascade [56].
Similar LL-37/nucleic acid complexes have been implicated in the pathogenesis of SLE, where they may contribute to the characteristic anti-dsDNA antibody response. Elevated LL-37 levels have also been found in atherosclerotic plaques, suggesting a role in vascular inflammation.
These findings have practical implications for peptide stacking in autoimmune patients:
| Condition | LL-37 Recommendation | Rationale |
|---|---|---|
| Psoriasis (active flare) | Avoid | LL-37 may exacerbate psoriatic inflammation through TLR9 activation |
| Psoriasis (remission) | Use with caution, low dose | May be tolerated when skin inflammation is controlled |
| SLE (active) | Avoid | LL-37/DNA complexes may amplify anti-dsDNA responses |
| SLE (stable/remission) | Use with caution, close monitoring | Monitor anti-dsDNA titers and complement levels |
| Rheumatoid arthritis | Generally acceptable | No specific contraindication; monitor for joint flares |
| Hashimoto's thyroiditis | Generally acceptable | No specific LL-37 involvement in thyroid autoimmunity |
| IBD (Crohn's, UC) | Potentially beneficial | LL-37 deficiency may contribute to mucosal immune dysfunction in IBD |
| Multiple sclerosis | Use with caution | CNS inflammation context requires careful monitoring |
KPV in Autoimmunity: The Most Autoimmune-Friendly Option
Of the four immune peptides, KPV has the most favorable profile for autoimmune conditions. Its mechanism - NF-kB inhibition via PepT1 transport - directly addresses the excessive inflammatory signaling that drives most autoimmune tissue damage. Unlike Ta1 and LL-37, which have both immune-enhancing and immune-modulating properties, KPV is predominantly anti-inflammatory.
The IBD research is the most relevant here. In ulcerative colitis and Crohn's disease models, KPV reduced mucosal inflammation, preserved barrier integrity, and decreased pro-inflammatory cytokine production - all without suppressing the antimicrobial defense functions of the gut immune system [57]. This selective anti-inflammatory action makes KPV particularly attractive for autoimmune conditions affecting the gut.
For non-GI autoimmune conditions, KPV's systemic anti-inflammatory effects (when administered subcutaneously) may help reduce inflammatory burden without the broad immunosuppression caused by corticosteroids or biologics. However, human clinical trial data for KPV in autoimmune conditions is limited, and any use should be viewed as experimental.
Thymulin in Autoimmunity
Thymulin's role in autoimmunity relates primarily to its effects on T-cell development and immune regulation. Thymic dysfunction has been implicated in several autoimmune conditions - the thymus is where self-reactive T cells should be eliminated through negative selection, and impaired thymic function may allow autoreactive T cells to escape into the periphery.
Thymulin, by supporting thymic hormone signaling, may help improve the quality of T-cell selection and reduce the emergence of autoreactive T-cell clones. Its anti-inflammatory properties (demonstrated in sepsis models) add an additional layer of potential benefit in autoimmune conditions.
Recommended Autoimmune Protocol
For individuals with autoimmune conditions who want to explore immune peptides, the following modified protocol minimizes risk while potentially improving immune regulation:
| Peptide | Dose | Frequency | Duration | Notes |
|---|---|---|---|---|
| KPV | 500 mcg oral | Daily | 8-12 weeks | Start here - best autoimmune safety profile |
| Ta1 | 0.8-1.6 mg SC | 1-2x weekly | 8-12 weeks | Start at lower dose; increase if tolerated |
| LL-37 | 50-100 mcg SC | 3x weekly | 8-12 weeks | Only if no contraindication (psoriasis, SLE) |
| Thymulin + Zinc | Per product | Daily | 8-12 weeks | Supportive; low risk |
Essential monitoring for autoimmune patients includes regular tracking of disease-specific markers (anti-CCP for RA, anti-dsDNA for SLE, TPO antibodies for Hashimoto's), inflammatory markers (CRP, ESR), and symptom diaries. Any flare of autoimmune symptoms should prompt reduction or discontinuation of immune-enhancing peptides (Ta1, LL-37) while continuing anti-inflammatory peptides (KPV).
For individuals new to peptide research who want to explore options further, our getting started guide provides orientation on the peptide landscape.
Blood Work Monitoring: Tracking What Matters
Subjective improvements - fewer colds, more energy, faster recovery - are valuable but imprecise. Objective blood work monitoring allows you to verify that immune peptides are producing measurable changes and to catch any unexpected effects early. The right lab panel transforms immune peptide use from guesswork into data-driven optimization.
Baseline Testing: Before Starting Any Protocol
Get the following labs before starting immune peptides. They establish your baseline and help identify pre-existing issues that might influence protocol selection:
Essential Panel
| Test | What It Measures | Why It Matters | Optimal Range |
|---|---|---|---|
| CBC with differential | White blood cell count and subsets | Baseline immune cell populations; identifies lymphocytopenia | WBC 4.5-11.0; Lymphocytes 20-40% |
| hsCRP | High-sensitivity C-reactive protein | Systemic inflammation marker | <1.0 mg/L (optimal <0.5) |
| ESR | Erythrocyte sedimentation rate | Non-specific inflammation marker | <20 mm/hr (men); <30 mm/hr (women) |
| 25-OH Vitamin D | Vitamin D status | Required for LL-37 expression; immune function | 40-60 ng/mL |
| Zinc (serum or RBC) | Zinc status | Required for thymulin activity; T-cell function | Serum: 70-120 mcg/dL; RBC preferred |
| Ferritin | Iron stores | Too low impairs immunity; too high signals inflammation | 30-150 ng/mL |
| Comprehensive metabolic panel | Kidney and liver function | Safety monitoring baseline | All values within reference range |
Advanced Panel (If Budget Allows)
| Test | What It Measures | Why It Matters | Notes |
|---|---|---|---|
| Lymphocyte subset panel (CD4, CD8, NK cells) | T-cell and NK cell populations | Direct assessment of adaptive immune status; CD4:CD8 ratio | Most informative for Ta1 efficacy tracking |
| Immunoglobulin panel (IgG, IgA, IgM) | Antibody levels | Identifies humoral immune deficiency | Low IgA may indicate mucosal immune weakness |
| NK cell activity/cytotoxicity | Functional NK cell killing capacity | Goes beyond counting NK cells to assess function | Tracks Ta1's NK-enhancing effect |
| IL-6, TNF-a | Pro-inflammatory cytokines | Specific inflammation markers; track KPV efficacy | Specialized labs; may require advance ordering |
| ANA, anti-dsDNA (if autoimmune risk) | Autoantibodies | Screen for autoimmune activation | Essential for those with autoimmune history/risk |
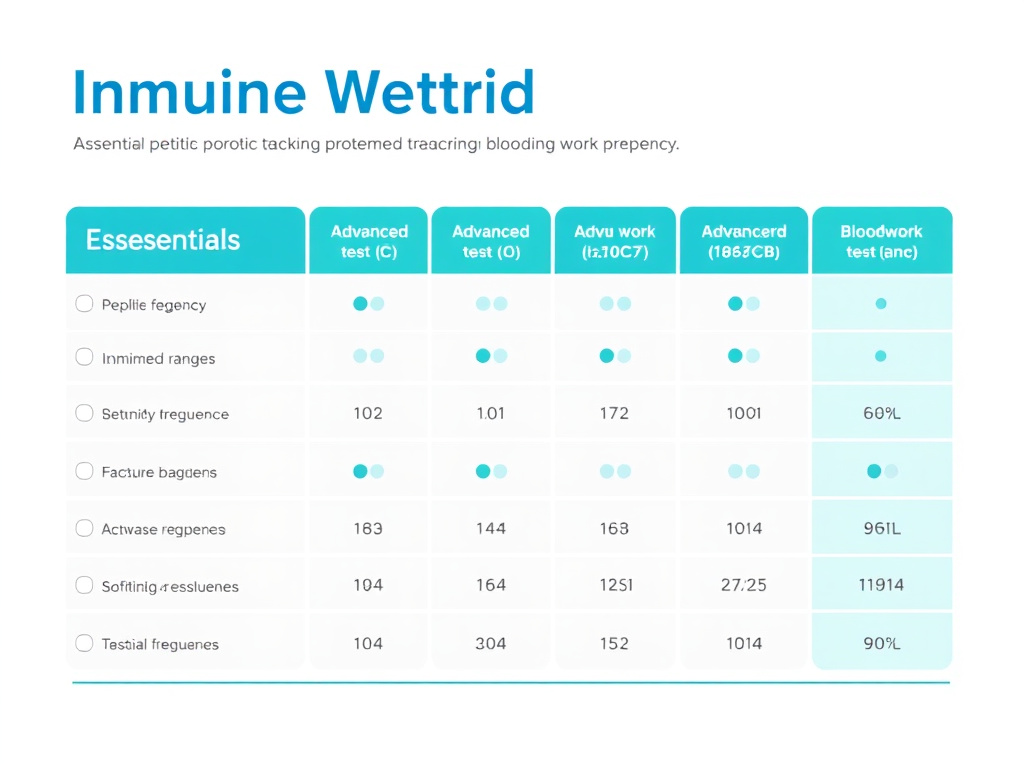
Figure 7: Recommended blood work monitoring panel for immune peptide protocols showing essential vs. advanced tests with optimal timing.
Follow-Up Testing Schedule
| Timepoint | Tests | Purpose |
|---|---|---|
| Baseline (before starting) | Full essential + advanced panel | Establish baseline values |
| 4 weeks into protocol | CBC with diff, hsCRP | Early safety check; initial response assessment |
| 8-12 weeks (end of cycle) | Full essential + advanced panel | Comprehensive efficacy assessment |
| 4 weeks after stopping (off-cycle) | CBC with diff, hsCRP | Assess durability of improvements |
| Before each subsequent cycle | Essential panel minimum | Re-establish baseline; track long-term trends |
What to Look For in Results
Signs of Positive Response
- Increase in absolute lymphocyte count (especially if starting below 1500/uL)
- Normalization of CD4:CD8 ratio toward 1.5-2.5:1
- Increase in NK cell count and/or cytotoxicity
- Decrease in hsCRP (especially if starting above 1.0 mg/L)
- Decrease in ESR
- Normalization of immunoglobulin levels if previously deficient
- Decrease in pro-inflammatory cytokines (IL-6, TNF-a) if measured
Potential Concerns Requiring Evaluation
- Unexplained increase in WBC count above normal range (rule out infection)
- New positive ANA or increasing autoantibody titers
- Rising hsCRP despite anti-inflammatory peptide use
- Declining lymphocyte counts (paradoxical response; rare)
- Liver or kidney function changes on CMP
- Inverted CD4:CD8 ratio (below 1.0) without known cause
Clinical Pearl
The most consistently useful single test for tracking immune peptide efficacy is the lymphocyte subset panel (CD4, CD8, NK cell counts). This directly measures the cell populations that Ta1 targets and provides more information than a standard CBC with differential. If budget allows only one advanced test, choose this one. For comprehensive blood work guidance across all peptide types, see our Ta1 research deep-dive.
Interpreting Changes in Context
Blood work results must be interpreted in context. A single set of labs is a snapshot; trends over time are what matter. Some important caveats:
- Timing matters: Draw blood at consistent times (morning, fasting) for reliable comparisons. Immune markers fluctuate throughout the day with circadian rhythms.
- Illness confounds results: Even a mild cold can dramatically alter WBC counts, CRP, and cytokine levels. Delay testing until at least 2 weeks after any acute illness.
- Exercise effects: Intense exercise transiently alters immune markers. Avoid heavy exercise for 24-48 hours before blood draws.
- Stress: Psychological stress elevates cortisol, which suppresses lymphocyte counts and alters immune cell distribution. Consider stress levels when interpreting results.
- Sleep: Poor sleep immediately before testing can affect immune markers. Ensure adequate sleep the night before blood draws.
Safety Profile and Risk Management
Every therapeutic intervention carries potential risks, and immune peptides are no exception. However, the safety data for these four peptides - particularly Thymosin Alpha-1 - is remarkably reassuring compared to conventional immunomodulatory drugs. Understanding the risk profile allows informed decision-making and appropriate precautions.
Comparative Safety: Peptides vs. Conventional Immunomodulators
To appreciate the safety profile of immune peptides, compare them to conventional treatments for immune dysfunction:
| Agent | Mechanism | Common Side Effects | Serious Risks |
|---|---|---|---|
| Corticosteroids | Broad immunosuppression | Weight gain, mood changes, insomnia, glucose elevation | Osteoporosis, adrenal suppression, diabetes, infection risk |
| Interferons | Antiviral/immunostimulatory | Flu-like symptoms, depression, fatigue, cytopenias | Autoimmune reactions, severe depression, hepatotoxicity |
| Biologics (TNF inhibitors) | Targeted cytokine blockade | Injection site reactions, infections | Serious infections (TB), lymphoma risk, demyelination |
| Methotrexate | Folate antagonist/immunosuppressant | Nausea, mouth sores, fatigue | Hepatotoxicity, bone marrow suppression, lung toxicity |
| Thymosin Alpha-1 | TLR-mediated immune modulation | Mild injection site reactions (5-10%) | No serious adverse events in clinical trials |
| LL-37 | Direct antimicrobial + immune modulation | Injection site reactions | Theoretical autoimmune concern (psoriasis, SLE) |
| KPV | NF-kB inhibition via PepT1 | None reported at standard doses | None identified in preclinical studies |
| Thymulin | Thymic hormone signaling | Mild/none reported | None identified; zinc monitoring needed |
Peptide-Specific Safety Considerations
Thymosin Alpha-1 Safety Data
Ta1 has the most extensive safety record of any immune peptide. Key data points:
- Over 4,400 patients treated in clinical trials across 80+ studies
- No serious adverse events attributable to Ta1 in controlled trials
- Injection site reactions in 5-10% of patients (mild, self-limiting)
- No organ toxicity detected in long-term treatment programs (up to 12 months continuous use)
- No evidence of immune overstimulation, cytokine storm, or autoimmune induction
- No drug interactions identified
- Safe in combination with chemotherapy, antivirals, and other immunomodulators [58]
The excellent safety profile is attributed to Ta1's mechanism - it modulates the immune system's own regulatory machinery rather than forcing activation through a single high-intensity pathway. This self-regulatory mechanism makes dangerous overshoot unlikely.
LL-37 Safety Considerations
LL-37 safety data for subcutaneous use is more limited, but key considerations include:
- Injection site reactions are more common and potentially more pronounced than with Ta1, due to LL-37's interaction with local immune cells and mast cells
- Dose-dependent cytotoxicity at high concentrations (above 50 micromolar) - not a practical concern at standard subcutaneous doses
- Psoriasis and SLE risk - see autoimmune section
- Hemolytic potential at high concentrations - not clinically relevant for subcutaneous use
- Topical LL-37 in clinical trials (wound healing) was well-tolerated with no serious adverse events [59]
KPV Safety Data
KPV has the most favorable safety profile of the group, largely because:
- It's a natural fragment of an endogenous hormone (a-MSH)
- It does not activate melanocortin receptors (no skin darkening or hormonal effects)
- No toxicity observed at doses many times higher than effective anti-inflammatory concentrations in animal studies
- No immunosuppression at anti-inflammatory doses
- Oral bioavailability means no injection-related complications when taken orally
- No known drug interactions
Thymulin Safety Profile
Thymulin's safety data comes primarily from animal studies and small human investigations:
- No significant adverse effects reported in published studies
- The primary safety consideration is ensuring adequate zinc status (zinc toxicity from excessive supplementation is more likely than thymulin-related adverse effects)
- As an endogenous hormone, physiological-dose supplementation is unlikely to cause adverse effects
- Long-term safety data in humans is limited
Reconstitution, Storage, and Injection Safety
Safe preparation and administration of injectable peptides requires attention to sterile technique:
- Use bacteriostatic water for reconstitution. Bacteriostatic water contains 0.9% benzyl alcohol as a preservative, allowing multi-dose use. Sterile water lacks this preservative and should only be used for single-dose preparation.
- Swirl, don't shake. Vigorous shaking can denature peptides (break their three-dimensional structure). Gently swirl or roll the vial to dissolve lyophilized powder.
- Store reconstituted peptides at 2-8 degrees C (refrigerator). Use reconstituted peptides within 4 weeks (30 days). Never re-freeze reconstituted peptides in bacteriostatic water.
- Store lyophilized (unreconstituted) peptides at -20 degrees C (freezer). Stability exceeds 24 months for most peptides when stored properly.
- Use insulin syringes (29-31 gauge) for subcutaneous injection. These fine needles minimize pain and tissue damage.
- Rotate injection sites. Alternate between left and right abdomen, upper thighs, and upper arms. Avoid injecting in the same spot repeatedly.
- Clean injection sites with alcohol swabs before injection. Allow alcohol to dry before inserting the needle.
- Inspect solutions before injection. Discard if cloudy, discolored, or containing particles.
Contamination Risk
The single greatest safety risk with peptide use is contamination during reconstitution or injection. Bacterial contamination of multi-dose vials can cause injection site infections or, in rare cases, systemic infections. Always use proper sterile technique, never share vials between individuals, and discard vials that have been at room temperature for extended periods. If you notice redness, swelling, warmth, or pain at an injection site that worsens over 24-48 hours, seek medical evaluation to rule out infection.
Contraindications
| Contraindication | Affected Peptides | Rationale |
|---|---|---|
| Active organ transplant on immunosuppression | Ta1, LL-37 | Enhanced immune function could trigger graft rejection |
| Active psoriasis flare | LL-37 | LL-37/DNA complexes may worsen psoriatic inflammation |
| Active SLE flare | LL-37 | LL-37 may amplify anti-dsDNA responses |
| Pregnancy/breastfeeding | All | Insufficient safety data in pregnancy |
| Benzyl alcohol sensitivity | All injectable | Bacteriostatic water contains benzyl alcohol |
| Severe zinc toxicity | Thymulin | Avoid adding zinc if zinc levels already elevated |
Drug Interactions
No significant pharmacological drug interactions have been identified for any of the four immune peptides. Ta1 has been used safely alongside antivirals (interferon, entecavir, tenofovir), chemotherapy agents (cisplatin, doxorubicin, 5-FU), and other immunomodulators. However, common-sense precautions apply:
- Immunosuppressive drugs: Ta1 and LL-37's immune-enhancing effects may partially counteract immunosuppressive medications. This isn't necessarily harmful, but it should be considered in the context of the patient's treatment goals.
- Corticosteroids: High-dose corticosteroids may blunt the immune-enhancing effects of Ta1. Low-dose corticosteroids are less likely to interfere.
- Biologics (anti-TNF, anti-IL-6): The targeted cytokine blockade of biologics creates a different immune context that could theoretically interact with immune peptide effects. No specific adverse interactions have been reported.
- Anticoagulants: LL-37 has been found in atherosclerotic plaques and may have effects on vascular inflammation. While no interaction with anticoagulants has been documented, individuals on anticoagulant therapy should be aware of this theoretical concern.
Peptide Quality, Purity, and Sourcing: What You Need to Know
The difference between an effective peptide protocol and a waste of money often comes down to peptide quality. The immune peptide market includes everything from pharmaceutical-grade products to questionable gray-market offerings, and understanding how to evaluate quality is essential for safe and effective use.
Understanding Purity Grades
Peptide purity is typically expressed as a percentage determined by high-performance liquid chromatography (HPLC). The major purity grades and their implications are:
| Purity Grade | Typical Purity | Impurities Present | Appropriate For |
|---|---|---|---|
| Research grade | 95-98% | Truncated sequences, deletion products, TFA salts | In vitro research only |
| High purity | 98-99% | Minor truncations, minimal salt contaminants | Animal research, some human use |
| Pharmaceutical grade | >99% | Negligible impurities, tested for endotoxins and sterility | Human use, clinical applications |
For immune peptides intended for subcutaneous injection, purity of 98% or higher is strongly recommended. The 2-5% impurity in lower-grade peptides can include truncated peptide sequences (which may have unpredictable biological activity), residual solvents from synthesis (acetonitrile, TFA), and bacterial endotoxins (which could paradoxically trigger inflammation - the opposite of what KPV aims to do).
Certificate of Analysis: What to Look For
A legitimate certificate of analysis (CoA) should include:
- Identity confirmation: Mass spectrometry (MS or MALDI-TOF) confirming the molecular weight matches the expected peptide sequence. The observed mass should be within 1-2 daltons of the calculated mass.
- Purity assessment: HPLC chromatogram showing a single dominant peak at the expected retention time, with purity reported as the area percentage of the main peak versus total peak area.
- Amino acid analysis: Confirms the amino acid composition matches the intended sequence. This catches substitution errors that mass spectrometry alone might miss.
- Endotoxin testing: LAL (Limulus Amebocyte Lysate) or recombinant factor C assay confirming endotoxin levels below safe thresholds (typically less than 5 EU/mg for injectable peptides).
- Sterility testing: For pre-reconstituted formulations, sterility testing confirms the absence of viable microorganisms.
- Residual solvent analysis: Gas chromatography confirming that synthesis solvents (acetonitrile, TFA, DMF) are below ICH guidelines for residual solvents.
If a supplier can't provide a CoA with at least identity confirmation (MS), purity (HPLC), and endotoxin testing, consider it a red flag. Reputable suppliers like FormBlends provide comprehensive CoAs for all products, with third-party verification of purity and identity.
Synthesis Methods and Their Implications
Peptides are primarily manufactured through two methods:
Solid-Phase Peptide Synthesis (SPPS)
The standard method for peptides up to 50 amino acids. Amino acids are sequentially added to a growing chain anchored to a resin bead. The two main SPPS strategies are Fmoc (9-fluorenylmethyloxycarbonyl) and Boc (tert-butyloxycarbonyl) chemistry. Fmoc chemistry is preferred for most immune peptides because it uses milder conditions that are less likely to damage sensitive amino acid side chains.
For Ta1 (28 amino acids), SPPS is the standard synthesis method. The N-terminal acetylation that characterizes natural Ta1 must be specifically added during synthesis. KPV (3 amino acids) is straightforward to synthesize by SPPS with very high purity. LL-37 (37 amino acids) approaches the practical length limit for single-chain SPPS and may require fragment condensation approaches for highest purity.
Recombinant Expression
Longer peptides can be produced by inserting the encoding gene into bacteria (typically E. coli) and expressing the peptide in bacterial culture. This method can be more cost-effective for large-scale production but requires careful purification to remove bacterial proteins and endotoxins. Recombinant production is more commonly used for pharmaceutical-grade products where scale justifies the infrastructure investment.
Storage Degradation and Potency Loss
Even high-quality peptides lose potency over time if stored improperly. The main degradation pathways include:
- Hydrolysis: Water molecules break peptide bonds, producing fragments with reduced or altered activity. This is why lyophilized (water-removed) storage is critical for long-term stability and why reconstituted peptides have limited shelf life.
- Oxidation: Methionine and cysteine residues are susceptible to oxidation, which can alter peptide conformation and activity. Ta1's N-terminal acetylation provides some protection against oxidative degradation. Protecting peptides from light and oxygen exposure minimizes oxidation.
- Aggregation: Some peptides tend to self-associate into aggregates at higher concentrations or elevated temperatures. LL-37 is particularly prone to aggregation due to its amphipathic structure. Aggregated peptide may have altered biological activity and can cause increased injection site reactions.
- Deamidation: Asparagine and glutamine residues can lose their amide groups over time, changing the peptide's charge and potentially its biological activity. Thymulin contains asparagine at its C-terminus, making it susceptible to this degradation pathway.
To minimize degradation, follow these storage best practices: keep lyophilized peptides at -20 degrees C in sealed vials protected from light. After reconstitution, store at 2-8 degrees C and use within 30 days. Never leave reconstituted peptides at room temperature for extended periods. If a reconstituted peptide appears cloudy, discolored, or produces unusual injection site reactions, discard it and reconstitute a fresh vial.
Red Flags in Peptide Sourcing
Watch for these warning signs when evaluating peptide suppliers:
- No Certificate of Analysis available: Legitimate suppliers always provide CoAs. If asked and they can't or won't provide one, look elsewhere.
- Suspiciously low prices: Quality peptide synthesis has real costs. Prices dramatically below market rate may indicate lower purity, underfilled vials, or substituted products.
- No indication of testing methods: Suppliers should specify what testing has been done (HPLC, MS, endotoxin) and ideally identify the third-party lab used.
- Pre-mixed combinations without documentation: Some suppliers sell pre-mixed peptide blends without providing individual CoAs for each component. You can't verify what's in the vial without individual testing data.
- Unrealistic claims: Suppliers making specific disease treatment claims or guaranteeing specific health outcomes are operating outside ethical boundaries and may be less reliable overall.
- No batch tracking: Quality manufacturers assign lot numbers to each production batch, allowing traceability. Absence of lot numbers suggests poor quality control practices.
For verified, third-party tested immune peptides with full documentation, visit FormBlends Thymosin Alpha-1, LL-37, and KPV product pages.
Innate Immunity In-Depth Review: Pattern Recognition and First Response
To fully appreciate how immune peptides work, we need a deeper understanding of innate immunity - the rapid-response system that determines whether a threat is detected, how quickly reinforcements arrive, and whether the adaptive immune system gets properly activated.
Toll-Like Receptors: The Immune System's Alarm Network
Toll-like receptors (TLRs) are the immune system's primary pathogen detection system. Humans express 10 functional TLRs (TLR1-TLR10), each recognizing different classes of microbial molecules. Surface-expressed TLRs (TLR1, TLR2, TLR4, TLR5, TLR6) detect bacterial cell wall components, while endosomal TLRs (TLR3, TLR7, TLR8, TLR9) detect nucleic acids from viruses and bacteria that have been internalized by cells.
Thymosin Alpha-1's ability to signal through multiple TLRs (TLR2, TLR3, TLR4, TLR7, TLR9) is unusual for a single molecule. Most natural TLR ligands activate only one or two receptors. This multi-TLR engagement may explain why Ta1 enhances immune responses against such a wide range of pathogens - it essentially primes multiple alarm systems simultaneously, creating a state of heightened but balanced immune readiness.
The downstream signaling from TLR activation converges on two main pathways: the MyD88-dependent pathway (shared by most TLRs) and the TRIF-dependent pathway (used by TLR3 and TLR4). The MyD88 pathway activates NF-kB and MAP kinases, leading to pro-inflammatory cytokine production. The TRIF pathway activates IRF3, leading to type I interferon production - particularly important for antiviral defense. Ta1's activation of TLR3 specifically engages the TRIF pathway, which may contribute to its documented antiviral effects in hepatitis and other viral infections.
The Complement System
The complement system is a cascade of over 30 proteins that provides rapid antimicrobial defense through three pathways: classical (antibody-mediated), alternative (spontaneous activation on microbial surfaces), and lectin (pattern recognition). Complement activation leads to direct pathogen lysis (membrane attack complex), opsonization (coating pathogens for phagocytosis), and inflammation (through anaphylatoxins C3a and C5a).
LL-37 interacts with the complement system in several ways. It can activate the classical complement pathway independently of antibodies, providing another mechanism for rapid pathogen defense. LL-37 also modulates the inflammatory effects of complement activation - at low concentrations, it enhances complement-mediated killing, while at higher concentrations, it can dampen complement-driven inflammation. This dose-dependent modulation adds to LL-37's role as an immune balancer rather than a simple activator.
Macrophage Polarization: M1 vs. M2
Macrophages exist on a spectrum between two functional states: M1 (classically activated, pro-inflammatory, pathogen killing) and M2 (alternatively activated, anti-inflammatory, tissue repair). The balance between M1 and M2 macrophages in any tissue determines whether the immune response favors destruction of threats or repair of damage.
Chronic inflammation often involves a persistent M1 bias - macrophages remain in pathogen-killing mode even after the threat has been cleared, causing ongoing tissue damage. Conditions like atherosclerosis, obesity-associated inflammation, and some autoimmune diseases feature this pathological M1 persistence.
KPV's NF-kB inhibition has been shown to shift macrophage polarization toward the M2 phenotype, promoting resolution of inflammation and tissue repair. This is particularly relevant in the gut, where M1-dominant macrophage populations in the lamina propria drive the chronic inflammation seen in IBD. By promoting M1-to-M2 transition, KPV doesn't just suppress inflammation - it actively promotes healing.
Ta1 has more complex effects on macrophage polarization, generally enhancing macrophage activation in the context of infection (supporting M1 function when needed) while also promoting regulatory pathways that prevent excessive M1 activity. This context-dependent modulation is another reason why Ta1 and KPV work well together - Ta1 ensures macrophages can effectively fight infections, while KPV prevents them from staying in overdrive after the fight is won.
Neutrophil Extracellular Traps (NETs)
Neutrophils can release their DNA in web-like structures called NETs that trap and kill bacteria. This process (NETosis) is an important antimicrobial defense mechanism, but excessive NET formation contributes to thrombosis, tissue damage, and autoimmune reactions. LL-37 stimulates NET formation, enhancing antimicrobial defense but potentially contributing to tissue damage when produced in excess.
The balance between beneficial and harmful NET formation is another area where the immune peptide stack provides built-in regulation. LL-37 promotes NETs for pathogen clearance, while KPV's anti-inflammatory effects help prevent excessive NET-driven tissue damage. This push-pull dynamic illustrates why stacking peptides with complementary mechanisms can be superior to using any single agent at high doses.
Dendritic Cell Subtypes and Their Roles
Not all dendritic cells (DCs) are created equal. Two main subtypes have distinct functions:
- Conventional DCs (cDC1, cDC2): These are the primary antigen-presenting cells that activate T cells. cDC1s are specialists in cross-presentation (activating CD8+ T cells) and are particularly important for antiviral and antitumor immunity. cDC2s primarily activate CD4+ T helper cells and are important for anti-bacterial and anti-parasitic responses.
- Plasmacytoid DCs (pDCs): These produce massive amounts of type I interferons (IFN-a/b) in response to viral nucleic acids detected through TLR7 and TLR9. They are the immune system's primary antiviral alarm system.
Ta1 activates both conventional and plasmacytoid DCs, enhancing both antigen presentation (through cDCs) and interferon production (through pDCs). This dual activation is relevant for viral defense, where both rapid interferon responses (pDC-mediated) and effective T-cell activation (cDC-mediated) are needed for viral clearance.
However, this pDC activation is also why LL-37 can be problematic in psoriasis and lupus. In these conditions, LL-37/DNA complexes activate pDCs through TLR9, driving chronic type I interferon production that sustains autoimmune inflammation. Understanding these mechanistic details helps explain why LL-37 should be avoided in certain autoimmune conditions while remaining beneficial in others.
Practical Application: Immune Peptides for Infection Defense
While the mechanistic details are important for understanding how immune peptides work, most people want practical guidance on how to use them for real-world infection defense. This section covers specific scenarios where immune peptide protocols have demonstrated value or strong theoretical support.
Respiratory Infections
Upper and lower respiratory tract infections - from the common cold to influenza to pneumonia - remain the most frequent infectious threats for most people. The respiratory mucosa is protected by multiple layers of defense: mucus production, ciliary clearance, epithelial barrier integrity, secretory IgA, and antimicrobial peptides including LL-37.
LL-37 is constitutively expressed in respiratory epithelial cells and is upregulated during infection. Vitamin D status directly influences respiratory LL-37 levels, which may partly explain the well-documented seasonal pattern of respiratory infections (viral prevalence peaks in winter when vitamin D levels are lowest, and thus LL-37 expression is reduced).
For respiratory infection defense, the peptide approach combines:
- Ta1 to enhance T-cell and NK cell surveillance of respiratory tissues
- LL-37 to boost antimicrobial peptide levels at mucosal surfaces
- Vitamin D supplementation (5000 IU daily) to support endogenous LL-37 expression
- KPV to manage inflammatory damage if infection occurs (particularly relevant for preventing the excessive inflammation that characterizes severe respiratory infections)
Gut Infections and Dysbiosis
The gut harbors the largest density of immune cells in the body and maintains a delicate balance between tolerance of commensal bacteria and defense against pathogens. Disruption of this balance - through antibiotic use, dietary changes, stress, or pathogenic infection - can lead to dysbiosis and chronic gut inflammation.
KPV's PepT1-mediated uptake makes it uniquely suited for gut-targeted immune support. Oral administration delivers KPV directly to intestinal epithelial cells, where it reduces NF-kB-driven inflammation while preserving antimicrobial defense. For individuals dealing with post-antibiotic dysbiosis, small intestinal bacterial overgrowth (SIBO), or chronic GI inflammation, oral KPV (500-1000 mcg daily) combined with Ta1 (1.6 mg SC twice weekly) provides both local gut support and systemic immune enhancement.
LL-37's antimicrobial activity extends to gut pathogens including Clostridioides difficile (C. diff), Salmonella species, and pathogenic E. coli strains. While subcutaneous LL-37 may not reach effective concentrations in the gut lumen, its systemic immune-modulating effects (chemokine production, immune cell activation) support the gut immune response indirectly.
Chronic and Recurrent Infections
Some individuals experience recurrent infections - urinary tract infections, sinusitis, skin infections, or respiratory infections - that suggest an underlying immune vulnerability. In these cases, the peptide approach aims to address the root cause (impaired immune surveillance) rather than just treating each episode.
Ta1 is the primary peptide for chronic/recurrent infection patterns. Its ability to enhance T-cell function, boost NK cell activity, and improve dendritic cell antigen presentation addresses the underlying immune deficit that allows recurrent infections. The recommended protocol is:
| Phase | Duration | Protocol |
|---|---|---|
| Loading phase | 2 weeks | Ta1 1.6 mg SC daily + LL-37 200 mcg SC daily |
| Consolidation phase | 8 weeks | Ta1 1.6 mg SC 2x/week + LL-37 200 mcg SC daily + KPV 500 mcg oral daily |
| Maintenance phase | Ongoing (cycled) | Ta1 1.6 mg SC 1x/week + KPV 500 mcg oral daily |
For recurrent UTIs specifically, the combination of Ta1 (systemic immune support) and LL-37 (antimicrobial peptide with activity against common uropathogens) may be particularly effective. LL-37 is naturally expressed in the urinary tract and contributes to mucosal defense against ascending infections.
Biofilm-Associated Infections
Biofilm infections represent one of the most challenging categories of infectious disease. Bacteria within biofilms are protected from both antibiotics and immune cells by the extracellular matrix. Common biofilm-associated conditions include chronic sinusitis, chronic wound infections, implant-associated infections, and chronic prostatitis.
LL-37's anti-biofilm activity makes it the peptide of choice for suspected biofilm infections. Its ability to prevent biofilm formation, disrupt established biofilms, suppress quorum sensing, and kill bacteria within biofilm structures provides multiple angles of attack that complement conventional antibiotic therapy.
For biofilm-associated infections, the recommended approach is:
- LL-37 at the higher end of the dose range (200-400 mcg SC daily) for extended periods (8-12 weeks minimum)
- Ta1 concurrently to ensure the adaptive immune system is optimally activated to clear bacteria as they're released from disrupted biofilms
- KPV to manage the inflammatory response that can accompany biofilm disruption
- Consideration of concurrent antibiotic therapy guided by culture and sensitivity testing
Mold Exposure and CIRS
Chronic Inflammatory Response Syndrome (CIRS) from mold exposure involves a dysfunctional immune response to mycotoxins and other mold-derived compounds. The immune system in CIRS patients often shows characteristic abnormalities: elevated inflammatory markers (C4a, TGF-b1, MMP-9), low MSH (melanocyte-stimulating hormone), and impaired regulatory T-cell function.
The immune peptide stack has theoretical relevance for CIRS through several mechanisms:
- Ta1: May help restore T-cell function and regulatory capacity impaired by chronic mold exposure
- LL-37: Has antifungal activity that may help clear residual mold colonization
- KPV: As a fragment of a-MSH (which is typically low in CIRS), KPV may help address the MSH deficiency component of the syndrome
- VIP: While not part of the core immune stack, VIP is specifically used in CIRS protocols and can be combined with immune peptides for comprehensive support
The LL-37 research page provides additional detail on its antifungal properties relevant to mold-related conditions.
Practical Guide: Reconstitution, Storage, and Administration
The effectiveness of peptide therapy depends not just on choosing the right peptides and doses, but on proper handling from the moment a vial arrives. Improper reconstitution, storage, or injection technique can degrade peptides, introduce contamination, or result in subtherapeutic dosing.
Reconstitution Step-by-Step
Materials Needed
- Lyophilized peptide vial
- Bacteriostatic water (BAC water) vial
- Alcohol swabs
- Insulin syringes (29-31 gauge, 0.5 mL or 1 mL)
- Larger drawing syringe (3 mL) for reconstitution (optional but helpful)
Reconstitution Process
- Wash hands thoroughly and work on a clean surface.
- Swab the rubber stopper of both the peptide vial and the BAC water vial with alcohol. Allow to dry for 10 seconds.
- Draw the appropriate volume of BAC water into a syringe. Standard volumes:
- Ta1 (5 mg vial): Add 2.5 mL BAC water = 2 mg/mL concentration
- LL-37 (5 mg vial): Add 3.0 mL BAC water = 1.67 mg/mL (1,670 mcg/mL)
- KPV (5 mg vial): Add 2.5 mL BAC water = 2 mg/mL (2,000 mcg/mL) for SC use
- Inject BAC water into the peptide vial slowly, directing the stream against the glass wall of the vial (not directly onto the powder). This prevents frothing and mechanical damage to the peptide.
- Gently swirl or roll the vial until the powder is fully dissolved. Never shake vigorously.
- Label the vial with the peptide name, concentration, and reconstitution date.
- Store immediately at 2-8 degrees C (refrigerator).
Dose Calculation
Once you know the concentration, calculating your injection volume is straightforward:
Volume (mL) = Desired dose (mg) / Concentration (mg/mL)
Examples for common doses:
| Peptide | Vial Size | BAC Water Added | Concentration | Common Dose | Injection Volume |
|---|---|---|---|---|---|
| Ta1 | 5 mg | 2.5 mL | 2 mg/mL | 1.6 mg | 0.80 mL (80 units) |
| LL-37 | 5 mg | 3.0 mL | 1.67 mg/mL | 200 mcg (0.2 mg) | 0.12 mL (12 units) |
| KPV (SC) | 5 mg | 2.5 mL | 2 mg/mL | 500 mcg (0.5 mg) | 0.25 mL (25 units) |
Injection Technique
- Select injection site: abdomen (2 inches from navel), outer thigh, or upper arm. Rotate sites.
- Clean the site with an alcohol swab and allow to dry.
- Pinch a fold of skin between thumb and forefinger.
- Insert the needle at a 45-90 degree angle (45 for very lean individuals, 90 for most).
- Inject slowly and steadily.
- Withdraw the needle and apply gentle pressure with a clean swab. Do not rub.
- Dispose of the syringe in a sharps container.
Storage Guidelines
| Form | Temperature | Stability | Notes |
|---|---|---|---|
| Lyophilized (unreconstituted) | -20 degrees C (freezer) | 24+ months | Protect from light; keep sealed |
| Lyophilized (unreconstituted) | 2-8 degrees C (fridge) | 6-12 months | Acceptable for short-term storage |
| Reconstituted (BAC water) | 2-8 degrees C (fridge) | 30 days | Use within 4 weeks; inspect before each use |
| Reconstituted (BAC water) | Room temperature | Hours only | Never store reconstituted peptides at room temp |
| Reconstituted (sterile water) | 2-8 degrees C (fridge) | Single use only | No preservative - risk of bacterial growth |
Figure 8: Peptide reconstitution and storage quick-reference guide showing proper technique and temperature requirements.
Traveling with Peptides
For those who need to travel while on a peptide protocol:
- Keep reconstituted vials cold: Use an insulated bag with ice packs for travel. Reconstituted peptides should not be at room temperature for more than a few hours.
- Bring lyophilized backup: If traveling for more than a few days, bring unreconstituted vials and BAC water to prepare fresh solutions at your destination.
- TSA/airport security: Peptide vials, syringes, and BAC water are treated like other injectable medications. Keep them in original packaging with any available documentation. Insulin syringes generally pass security without issue when accompanied by medication vials.
- International travel: Check destination country regulations regarding injectable medications. Carry a letter from your healthcare provider if possible.
Quality and Sourcing Considerations
Peptide quality varies significantly between sources. Key quality indicators include:
- Purity: Look for peptides with 98%+ purity verified by HPLC (high-performance liquid chromatography). Lower purity products may contain degradation products, synthesis byproducts, or residual solvents.
- Third-party testing: Reputable suppliers provide certificates of analysis (CoA) showing identity confirmation (mass spectrometry), purity (HPLC), endotoxin testing, and sterility testing.
- Proper lyophilization: Quality peptides are lyophilized (freeze-dried) to a fine, uniform powder. Chunky, discolored, or wet-appearing powder suggests improper processing or storage damage.
- Packaging: Vials should be properly sealed with crimped caps, stored under appropriate conditions, and shipped with cold packs for temperature-sensitive products.
For research-grade immune peptides with third-party testing, visit FormBlends Thymosin Alpha-1, LL-37, and KPV product pages for detailed specifications and certificates of analysis.
Frequently Asked Questions
What peptides are best for boosting the immune system?
Thymosin Alpha-1 (Ta1) has the strongest clinical evidence for immune enhancement, with approval in over 35 countries for hepatitis B/C and as a cancer immunotherapy adjuvant. It works by activating dendritic cells through Toll-like receptors and enhancing T-cell and NK cell function. LL-37 provides direct antimicrobial defense against bacteria, viruses, and fungi. KPV addresses the inflammatory side of immunity by inhibiting NF-kB signaling. The combination of these peptides covers detection, elimination, inflammation control, and immune cell replenishment. For general immune support, starting with Ta1 alone at 1.6 mg subcutaneously twice weekly is the most evidence-based approach, with additional peptides added based on individual needs and response.
How do you combine immune peptides safely?
The safest approach is to introduce peptides one at a time, waiting 5-7 days between additions. Start with Thymosin Alpha-1 (best safety profile), then add LL-37, then KPV. Inject different peptides at different body sites to prevent local interactions and to identify which peptide causes any reaction. Get baseline blood work (CBC with differential, hsCRP, vitamin D, zinc) before starting. Recheck labs at 4 weeks for a safety assessment and at 8-12 weeks for efficacy evaluation. There are no known pharmacological interactions between these peptides since they act through different receptor systems. However, individuals with autoimmune conditions should avoid LL-37 if they have active psoriasis or lupus, as LL-37 can exacerbate these conditions through specific mechanisms.
What is the standard dosing protocol for Thymosin Alpha-1?
The standard clinical dose established across decades of trials is 1.6 mg administered subcutaneously twice weekly. This dose was used in hepatitis B/C, cancer immunotherapy, and vaccine adjuvancy studies. For acute immune support (active infections, post-exposure), many protocols increase to 1.6 mg daily for 5-7 days before reverting to twice weekly. The half-life is approximately 2 hours, but immunological effects persist for 48-72 hours. Injection sites should be rotated between the abdomen, thighs, and upper arms. Reconstitute with bacteriostatic water, store at 2-8 degrees C after reconstitution, and use within 30 days. Most protocols run for 8-12 weeks followed by a 4-week break.
Can immune peptides help with autoimmune conditions?
This is nuanced. KPV has the most favorable profile for autoimmune conditions because it reduces inflammation through NF-kB inhibition without broadly stimulating the immune system. Thymosin Alpha-1 may actually help autoimmune conditions by expanding regulatory T cells (Tregs) that suppress autoreactive immune responses - research shows autoimmune patients often have lower serum Ta1 levels than healthy controls. However, LL-37 should be avoided in active psoriasis and lupus because it can form complexes with self-DNA that amplify autoimmune inflammation through TLR9 activation. Anyone with an autoimmune condition should work with a knowledgeable provider, start at lower doses, and monitor disease-specific markers closely. Consider starting with KPV alone before adding other immune peptides.
How long does it take to see results from immune peptide protocols?
Subjective improvements (fewer infections, better energy, improved recovery from illness) typically become noticeable within 4-8 weeks. Measurable blood work changes usually appear within 4-6 weeks - look for increases in lymphocyte counts, improved CD4:CD8 ratios, and decreases in inflammatory markers like hsCRP. LL-37's antimicrobial effects can be more immediate, with some users reporting improvement in chronic infections within 2-4 weeks. KPV's anti-inflammatory effects on gut symptoms may be noticeable within 1-2 weeks. The full benefits of the immune stack typically develop over 2-3 months of consistent use. Give any protocol at least 8 weeks before assessing efficacy, and use objective blood work rather than relying solely on subjective impressions.
What is the role of LL-37 in fighting infections?
LL-37 is the only human cathelicidin antimicrobial peptide. It kills pathogens through direct membrane disruption - its positively charged alpha-helical structure binds to negatively charged bacterial membranes and creates pores that destroy the cell. This mechanism is difficult for bacteria to develop resistance against because it targets lipid structure rather than specific proteins. Beyond direct killing, LL-37 disrupts biofilms (structured bacterial communities that resist antibiotics), recruits immune cells to infection sites through chemotactic signaling, and neutralizes bacterial endotoxin (LPS). It also promotes wound healing through stimulation of angiogenesis and cell migration. LL-37 expression is regulated by vitamin D, so maintaining vitamin D levels of 40-60 ng/mL supports endogenous LL-37 production.
Is KPV effective for gut inflammation and leaky gut?
Preclinical research strongly supports KPV for gut inflammation. In DSS-induced and TNBS-induced colitis models (standard models for ulcerative colitis and Crohn's disease), orally administered KPV reduced body weight loss, decreased neutrophil infiltration, preserved mucosal architecture, and lowered pro-inflammatory cytokine expression. KPV enters intestinal cells through the PepT1 transporter, which is upregulated during inflammation - so it becomes more effective precisely when and where inflammation is active. At nanomolar concentrations, KPV inhibits NF-kB, the master regulator of inflammatory gene transcription. For oral use targeting gut inflammation, 500-1000 mcg daily is a common research protocol. Human clinical trial data is still limited, but the animal data is promising and the safety profile is excellent.
What blood work should I get before starting immune peptides?
At minimum, get a CBC with differential (measures white blood cell populations including lymphocyte counts), hsCRP (inflammation marker), 25-OH vitamin D (needed for LL-37 expression), serum zinc (needed for thymulin activity), ferritin (iron status), and a comprehensive metabolic panel (liver and kidney function baseline). If budget allows, add a lymphocyte subset panel (CD4, CD8, NK cell counts and ratios), immunoglobulin panel (IgG, IgA, IgM), and NK cell cytotoxicity assay. For those with autoimmune risk factors, include ANA and relevant disease-specific antibodies. Recheck the essential panel at 4 weeks (safety), 8-12 weeks (efficacy), and 4 weeks after stopping (durability). Draw blood in the morning, fasting, and avoid heavy exercise for 24-48 hours before the test.
How does thymulin relate to zinc status and thymic function?
Thymulin is a nonapeptide hormone produced exclusively by thymic epithelial cells. It exists in two forms: an active zinc-bound form and an inactive zinc-free (apo) form. Without zinc bound in an equimolar ratio, thymulin cannot bind its receptor or exert biological effects on T-cell differentiation. This makes serum thymulin activity one of the most sensitive functional biomarkers of marginal zinc deficiency - levels drop before clinical zinc deficiency symptoms appear. The connection works both ways: thymic involution with aging reduces thymulin production, while age-related zinc deficiency reduces activation of whatever thymulin is produced. This is why thymulin supplementation should always include adequate zinc (15-30 mg elemental zinc daily). The combination addresses both the hormone supply and the cofactor availability.
Can I use immune peptides alongside conventional medications?
Yes, in most cases. Thymosin Alpha-1 has been safely combined with interferons, antivirals, chemotherapy agents, and other immunomodulators across numerous clinical trials. No significant drug interactions have been identified for any of the four immune peptides. However, there are important considerations: immune-enhancing peptides (Ta1, LL-37) may partially counteract immunosuppressive medications, which matters if immunosuppression is therapeutically intended (organ transplant, severe autoimmune disease). High-dose corticosteroids may blunt Ta1's immune-enhancing effects. The targeted blockade of biologics (anti-TNF, anti-IL-6 agents) creates an altered immune context that could theoretically interact with immune peptide effects, though no adverse interactions have been reported. Always inform your healthcare provider about peptide use.
What is the difference between thymulin and thymalin?
Despite similar names, these are different peptides. Thymulin (also called FTS - facteur thymique serique) is a single, well-defined nonapeptide (9 amino acids: Glu-Ala-Lys-Ser-Gln-Gly-Gly-Ser-Asn) discovered by Jean-Francois Bach in Paris in 1977. It requires zinc for biological activity. Thymalin is a polypeptide complex extracted from calf thymus glands by Vladimir Khavinson at the St. Petersburg Institute of Bioregulation and Gerontology. Thymalin contains a mixture of short peptides whose individual components aren't fully characterized. Both have thymic origins and immune-modulating properties, but thymulin has a clear structure-activity relationship while thymalin's mechanism is less defined. Thymalin has been studied extensively in Russia for immune restoration in elderly patients and those exposed to radiation.
How should I cycle immune peptides for long-term use?
Several cycling frameworks work well. The simplest is 8 weeks on, 4 weeks off, which provides two full cycles in six months. Seasonal cycling aligns peptide use with cold and flu season (October through March in the Northern Hemisphere), with a break during warmer months. A stepwise taper reduces frequency gradually over 2-4 weeks before the off period rather than stopping abruptly. For those on a budget, rotating single-peptide emphasis (2 months Ta1 alone, 2 months Ta1 + LL-37, 2 months full stack, 1 month off) allows you to assess individual peptide contributions while reducing cost. Blood work before and after each cycle helps track objective improvements and guide future protocol decisions.
Are there any serious side effects of immune peptide stacking?
The safety data for these peptides is reassuring. Thymosin Alpha-1 has been tested in over 4,400 patients across 80+ clinical trials with no serious adverse events attributable to the peptide. The most common side effect across all four peptides is mild injection site reactions (redness, swelling) with subcutaneous administration. KPV has no reported adverse effects at standard doses. The primary concern with LL-37 is its potential to exacerbate psoriasis and lupus through formation of LL-37/DNA complexes that activate TLR9. The biggest practical safety risk isn't the peptides themselves but contamination during reconstitution or injection - always use sterile technique and properly stored bacteriostatic water. Organ transplant recipients on immunosuppressive therapy should avoid immune-enhancing peptides due to graft rejection risk.
Should I take vitamin D and zinc with immune peptides?
Yes, both are strongly recommended. Vitamin D directly regulates LL-37 expression - the cathelicidin gene contains a vitamin D response element in its promoter. Without adequate vitamin D (target 40-60 ng/mL), your body produces less endogenous LL-37, and exogenous LL-37 operates in a suboptimal environment. Zinc is required for thymulin to adopt its active conformation. Without zinc, thymulin is biologically inactive regardless of how much you take. Beyond their peptide-specific roles, both vitamin D and zinc support broad immune function independently. Recommended daily intake: vitamin D 5000 IU (adjust based on blood levels), zinc 15-30 mg elemental (zinc picolinate or zinc bisglycinate for best absorption). Test both before starting and at 8-12 weeks to confirm levels are in range.
References & Clinical Sources
- Garaci E, et al. Thymosin alpha 1: A comprehensive review of the literature. World J Virol. 2021;9(5):67-102. doi:10.5501/wjv.v9.i5.67 [1]
- Vandamme D, et al. A comprehensive summary of LL-37, the factotum human cathelicidin peptide. Cell Immunol. 2012;280(1):22-35. doi:10.1016/j.cellimm.2012.11.009 [2]
- Dalmasso G, et al. PepT1-mediated tripeptide KPV uptake reduces intestinal inflammation. Gastroenterology. 2008;134(1):166-178. doi:10.1053/j.gastro.2007.10.026 [3]
- Dardenne M. Thymulin, a zinc-dependent hormone. Med Oncol. 1999;16(3):198-201. doi:10.1007/BF02785220 [4]
- Romani L, et al. Thymosin alpha 1 activates dendritic cell tryptophan catabolism and establishes a regulatory environment for balance of inflammation and tolerance. Blood. 2006;108(7):2265-2274. doi:10.1182/blood-2006-02-004762 [5]
- Palmer DB. The effect of age on thymic function. Front Immunol. 2013;4:316. doi:10.3389/fimmu.2013.00316 [6]
- Goldstein AL, et al. Thymosin alpha 1: isolation and sequence analysis of an immunologically active thymic polypeptide. Proc Natl Acad Sci USA. 1977;74(2):725-729. doi:10.1073/pnas.74.2.725 [7]
- Charrier L, et al. hPepT1 mediates bacterial tripeptide fMLP uptake in human monocytes. Lab Invest. 2006;86(4):397-407. doi:10.1038/labinvest.3700390 [8]
- Liu Y, et al. Thymosin alpha 1 reduces the mortality of severe COVID-19 by restoration of lymphocytopenia and reversion of exhausted T cells. Clin Infect Dis. 2020;71(16):2150-2157. doi:10.1093/cid/ciaa630 [9]
- Luger TA, et al. alpha-MSH related peptides: a new class of anti-inflammatory and immunomodulating drugs. Ann Rheum Dis. 1999;58(10):585-587. doi:10.1136/ard.58.10.585 [10]
- Maio M, et al. Thymosin alpha 1 and its role in viral infectious diseases. Molecules. 2023;28(8):3539. doi:10.3390/molecules28083539 [11]
- Wu M, et al. Thymosin alpha 1 reduces the mortality of severe COVID-19 by restoration of lymphocytopenia and reversion of exhausted T cells. Clin Infect Dis. 2020;71(16):2150-2157. doi:10.1093/cid/ciaa630 [12]
- Goldstein AL, Goldstein AL. From lab to bedside: emerging clinical applications of thymosin alpha 1. Expert Opin Biol Ther. 2009;9(5):593-608. doi:10.1517/14712590902911412 [13]
- Lunetta C, et al. Aging and thymosin alpha-1. Int J Mol Sci. 2024;26(23):11470. doi:10.3390/ijms262311470 [14]
- Romani L, et al. Thymosin alpha-1 represents a potential potent single-molecule-based therapy for cystic fibrosis. Nat Med. 2017;23(5):590-600. doi:10.1038/nm.4305 [15]
- Serafino A, et al. Thymosin alpha 1 as a stimulatory agent of innate cell-mediated immune response. Ann NY Acad Sci. 2012;1270:13-20. doi:10.1111/j.1749-6632.2012.06707.x [16]
- Romani L, et al. Thymosin alpha-1 activates dendritic cells for antifungal Th1 resistance through toll-like receptor signaling. Blood. 2004;103(11):4232-4239. doi:10.1182/blood-2003-11-4036 [17]
- Li J, et al. Thymosin alpha 1 reverses T cell exhaustion. Front Immunol. 2021;12:673693. doi:10.3389/fimmu.2021.673693 [18]
- Garaci E, et al. Thymosin alpha 1 in the treatment of cancer: from basic research to clinical application. Int J Immunopharmacol. 2000;22(12):1067-1076. doi:10.1016/S0192-0561(00)00077-3 [19]
- Iino S. Combination therapy with thymosin alpha 1 and interferon for the treatment of hepatitis B. J Gastroenterol Hepatol. 2001;16(11):1198-1199. doi:10.1046/j.1440-1746.2001.02612.x [20]
- Sherman KE. Thymosin alpha 1 for treatment of hepatitis C virus: promise and proof. Ann NY Acad Sci. 2010;1194:136-140. doi:10.1111/j.1749-6632.2010.05460.x [21]
- Garaci E. Thymosin alpha 1: a historical overview. Ann NY Acad Sci. 2007;1112:14-20. doi:10.1196/annals.1415.039 [22]
- Liu Y, et al. Thymosin alpha 1 reduces the mortality of severe COVID-19. Clin Infect Dis. 2020;71(16):2150-2157. doi:10.1093/cid/ciaa630 [23]
- Tao N, et al. Thymosin alpha1 use in adult COVID-19 patients: a systematic review and meta-analysis on clinical outcomes. Int Immunopharmacol. 2023;114:109472. doi:10.1016/j.intimp.2022.109472 [24]
- Wu J, et al. Thymosin alpha 1 treatment for sepsis: a systematic review. Int J Immunopharmacol. 2015;25(1):68-74. doi:10.1016/j.intimp.2015.01.009 [25]
- Shen YF, et al. Thymosin alpha 1 as a vaccine adjuvant for influenza in elderly subjects. Expert Opin Biol Ther. 2009;9(5):593-608. doi:10.1517/14712590902911412 [26]
- Matteucci C, et al. Thymosin alpha 1 and HIV-1: recent advances and future perspectives. Future Microbiol. 2017;12:141-155. doi:10.2217/fmb-2016-0125 [27]
- Garaci E, et al. Thymosin alpha 1: a comprehensive review. World J Virol. 2021;9(5):67-102. doi:10.5501/wjv.v9.i5.67 [28]
- Durr UH, et al. LL-37, the only human member of the cathelicidin family of antimicrobial peptides. Biochim Biophys Acta. 2006;1758(9):1408-1425. doi:10.1016/j.bbamem.2006.03.030 [29]
- Sancho-Vaello E, et al. The structure of the antimicrobial human cathelicidin LL-37 shows oligomerization and channel formation. Sci Rep. 2020;10(1):17356. doi:10.1038/s41598-020-74401-5 [30]
- Brogden KA. Antimicrobial peptides: pore formers or metabolic inhibitors in bacteria? Nat Rev Microbiol. 2005;3(3):238-250. doi:10.1038/nrmicro1098 [31]
- Overhage J, et al. Human host defense peptide LL-37 prevents bacterial biofilm formation. Infect Immun. 2008;76(9):4176-4182. doi:10.1128/IAI.00318-08 [32]
- Yang D, et al. LL-37, the neutrophil granule- and epithelial cell-derived cathelicidin, utilizes formyl peptide receptor-like 1 (FPRL1) as a receptor to chemoattract human peripheral blood neutrophils, monocytes, and T cells. J Exp Med. 2000;192(7):1069-1074. doi:10.1084/jem.192.7.1069 [33]
- Yang YM, et al. Significance of LL-37 on immunomodulation and disease outcome. Biomed Res Int. 2020;2020:8349712. doi:10.1155/2020/8349712 [34]
- Gronberg A, et al. Treatment with LL-37 is safe and effective in enhancing healing of hard-to-heal venous leg ulcers: a randomized, placebo-controlled clinical trial. Wound Repair Regen. 2014;22(5):613-621. doi:10.1111/wrr.12211 [35]
- Crane-Godreau MA, et al. Upregulating human cathelicidin antimicrobial peptide LL-37 expression may prevent severe COVID-19 inflammatory responses. Front Immunol. 2022;13:880961. doi:10.3389/fimmu.2022.880961 [36]
- Wang TT, et al. Cutting edge: 1,25-dihydroxyvitamin D3 is a direct inducer of antimicrobial peptide gene expression. J Immunol. 2004;173(5):2909-2912. doi:10.4049/jimmunol.173.5.2909 [37]
- Lande R, et al. Plasmacytoid dendritic cells sense self-DNA coupled with antimicrobial peptide. Nature. 2007;449(7162):564-569. doi:10.1038/nature06116 [38]
- Getting SJ. Targeting melanocortin receptors as potential novel therapeutics. Pharmacol Ther. 2006;111(1):1-15. doi:10.1016/j.pharmthera.2005.06.022 [39]
- Dalmasso G, et al. PepT1-mediated tripeptide KPV uptake reduces intestinal inflammation. Gastroenterology. 2008;134(1):166-178. doi:10.1053/j.gastro.2007.10.026 [40]
- Brzoska T, et al. Alpha-melanocyte-stimulating hormone and related tripeptides: biochemistry, antiinflammatory and protective effects in vitro and in vivo, and future perspectives for the treatment of immune-mediated inflammatory diseases. Endocr Rev. 2008;29(5):581-602. doi:10.1210/er.2007-0027 [41]
- Dalmasso G, et al. PepT1-mediated tripeptide KPV uptake reduces intestinal inflammation. Gastroenterology. 2008;134(1):166-178. doi:10.1053/j.gastro.2007.10.026 [42]
- Ma Z, et al. PepT1-targeted nanodrug based on co-assembly of anti-inflammatory peptide and immunosuppressant for combined treatment of acute and chronic DSS-induced colitis. Front Pharmacol. 2024;15:1442876. doi:10.3389/fphar.2024.1442876 [43]
- Luger TA, et al. Alpha-melanocyte-stimulating hormone as a mediator of tolerance induction. Pathobiology. 1999;67(5-6):318-321. doi:10.1159/000028083 [44]
- Catania A, et al. Antimicrobial properties of alpha-MSH and related synthetic melanocortins. ScientificWorldJournal. 2006;6:1241-1246. doi:10.1100/tsw.2006.227 [45]
- Bach JF, et al. Thymic serum factor (FTS). Bull Inst Pasteur. 1977;75:69-95. [46]
- Dardenne M, et al. Interactions between zinc and thymulin. Met Based Drugs. 2008;2008:524079. doi:10.1155/2008/524079 [47]
- Prasad AS. Comparison of thymulin activity with other measures of marginal zinc deficiency. Biol Trace Elem Res. 2020;196(2):382-389. doi:10.1007/s12011-019-01942-0 [48]
- Dardenne M. Role of thymic peptides as transmitters between the neuroendocrine and immune systems. Ann Med. 1999;31 Suppl 2:34-39. doi:10.3109/07853899909115242 [49]
- Dardenne M, et al. Thymulin, a zinc-dependent hormone. Med Oncol. 1999;16(3):198-201. doi:10.1007/BF02785220 [50]
- Safieh-Garabedian B, et al. Thymulin reduces hyperalgesia and cytokine upregulation in a rat model of inflammatory pain. Brain Behav Immun. 2011;25(1):180-189. doi:10.1016/j.bbi.2010.09.009 [51]
- Khavinson V, et al. Peptide bioregulators: a new class of geroprotectors. Adv Gerontol. 2012;2(3):177-183. doi:10.1134/S2079057012030060 [52]
- Goldstein AL, et al. Thymosin alpha 1 in clinical medicine. Expert Opin Biol Ther. 2009;9(5):593-608. doi:10.1517/14712590902911412 [53]
- Pica F, et al. Serum thymosin alpha 1 levels in patients with chronic inflammatory autoimmune diseases. Clin Exp Immunol. 2016;186(1):39-45. doi:10.1111/cei.12833 [54]
- Tuthill CW, et al. Thymosin alpha-1 - comprehensive review of the literature. World J Virol. 2020;9(5):67-102. doi:10.5501/wjv.v9.i5.67 [55]
- Lande R, et al. Plasmacytoid dendritic cells sense self-DNA coupled with antimicrobial peptide. Nature. 2007;449(7162):564-569. doi:10.1038/nature06116 [56]
- Dalmasso G, et al. PepT1-mediated tripeptide KPV uptake reduces intestinal inflammation. Gastroenterology. 2008;134(1):166-178. doi:10.1053/j.gastro.2007.10.026 [57]
- Garaci E, et al. Thymosin alpha 1: safety profile. World J Virol. 2021;9(5):67-102. doi:10.5501/wjv.v9.i5.67 [58]
- Gronberg A, et al. Treatment with LL-37 is safe and effective in enhancing healing of hard-to-heal venous leg ulcers. Wound Repair Regen. 2014;22(5):613-621. doi:10.1111/wrr.12211 [59]