Executive Summary

Figure 1: Thymosin alpha-1, a 28-amino-acid peptide derived from thymic tissue, plays a central role in immune system regulation and has been approved in over 35 countries worldwide.
Key Takeaways
- Ta1 is a 28-amino-acid thymic peptide approved in 35+ countries for hepatitis B/C and immune enhancement
- Mechanism centers on TLR2/TLR9 activation of dendritic cells, promoting T-cell maturation and balanced cytokine production
- Clinical trials show 25-40% HBV DNA clearance in hepatitis B and improved survival in cancer immunotherapy combinations
- Safety profile is outstanding, with no dose-limiting toxicities reported across decades of clinical use
- Standard dosing: 1.6 mg subcutaneously twice weekly, with variations for acute vs. maintenance protocols
Thymosin alpha-1 (Ta1), marketed as Zadaxin, is a 28-amino-acid peptide originally isolated from thymic tissue that functions as a potent immune modulator. Approved in over 35 countries for the treatment of hepatitis B and C, and used as an immune response enhancer in cancer, sepsis, and vaccine adjuvant therapy, Ta1 represents one of the most extensively studied peptides in modern immunology. This report covers the full scope of Ta1 research, from molecular mechanism through clinical trial outcomes, dosing protocols, and practical applications for immune optimization.
The thymus gland, a small organ situated behind the sternum, serves as the training ground for T lymphocytes. It produces a family of peptides collectively known as thymosins, among which thymosin alpha-1 has drawn the most clinical interest. First isolated in 1977 by Allan Goldstein and colleagues at the George Washington University School of Medicine, Ta1 was identified as the biologically active component of Thymosin Fraction 5, a partially purified extract of calf thymus tissue. The peptide has a molecular weight of 3,108 daltons and is acetylated at its N-terminus, a modification that contributes to its biological stability and activity.
What separates Ta1 from many other peptides under investigation is the sheer volume of clinical evidence supporting its use. More than 1,000 published studies and over 40 clinical trials have evaluated Ta1 across conditions ranging from chronic hepatitis B and C to non-small cell lung cancer, hepatocellular carcinoma, melanoma, HIV/AIDS, and severe sepsis. The peptide received orphan drug designation from the U.S. FDA for hepatocellular carcinoma, malignant melanoma, DiGeorge syndrome, and chronic active hepatitis B. While it hasn't achieved full FDA approval as a stand-alone drug in the United States, it is approved and widely used in countries throughout Latin America, Eastern Europe, the Middle East, and the Asia-Pacific region.
Ta1 works primarily by binding to Toll-like receptors 2 and 9 (TLR2 and TLR9) on dendritic cells, which triggers a signaling cascade that promotes T-cell maturation, enhances natural killer (NK) cell activity, and stimulates the production of key cytokines including interferon-alpha (IFN-a), interferon-gamma (IFN-g), and interleukin-2 (IL-2). Unlike conventional immunostimulants that simply ramp up immune activity, Ta1 acts as a genuine immune modulator. It can increase immune function in immunocompromised patients while simultaneously preventing the excessive inflammation seen in conditions like sepsis and cytokine storms. This dual action has made it particularly attractive for treating conditions where the immune system is both suppressed and dysregulated.
In the context of chronic hepatitis B, randomized controlled trials have demonstrated that Ta1 monotherapy achieves HBV DNA clearance rates of 25-40% at 12 months, compared to 9-13% with placebo. When combined with interferon-alpha, response rates increase further. For hepatitis C, combination therapy with Ta1 and interferon produced end-of-treatment biochemical response rates of 37-45%, compared to 16-22% with interferon alone. In cancer, Ta1 has shown particular promise as an adjunct to chemotherapy and immunotherapy, with studies in hepatocellular carcinoma showing median overall survival improvements from 11.0 to 16.0 months when added to lenvatinib plus anti-PD-1 antibody regimens. During the COVID-19 pandemic, multiple studies and meta-analyses evaluated Ta1 in severe and critically ill patients, with pooled data suggesting a 34-41% reduction in mortality among severe cases, though results across all severity levels remained inconsistent.
For those interested in thymosin alpha-1 therapy, the standard clinical dosing protocol involves 1.6 mg administered subcutaneously twice weekly, though acute protocols may use daily dosing for initial periods. The safety profile is exceptional. Across decades of clinical use and doses up to 16 mg administered for 12 months, no significant Ta1-specific toxicity has been reported. The most common side effect is mild injection-site irritation. This report provides a thorough examination of the science, evidence, and practical guidance for Ta1, drawing on peer-reviewed research and clinical trial data through 2025.
Key Takeaways
- Ta1 is a 28-amino-acid thymic peptide approved in 35+ countries for hepatitis B/C and immune enhancement
- Mechanism centers on TLR2/TLR9 activation of dendritic cells, promoting T-cell maturation and balanced cytokine production
- Clinical trials show 25-40% HBV DNA clearance in hepatitis B and improved survival in cancer immunotherapy combinations
- Safety profile is outstanding, with no dose-limiting toxicities reported across decades of clinical use
- Standard dosing: 1.6 mg subcutaneously twice weekly, with variations for acute vs. maintenance protocols
Historical Context and Regulatory Landscape
The development of thymosin alpha-1 spans nearly five decades of research, beginning with the pioneering work of Allan Goldstein at George Washington University. The initial discovery of Thymosin Fraction 5 in the early 1970s was itself a significant milestone in immunology, demonstrating for the first time that the thymus gland produced soluble factors capable of restoring immune function in thymectomized animals. The subsequent isolation and sequencing of Ta1 as the most biologically active component of this fraction opened the door to targeted immune therapy based on a naturally occurring human peptide.
The regulatory journey of Ta1 reflects the complex realities of global pharmaceutical development. SciClone Pharmaceuticals obtained marketing authorization for Zadaxin in multiple countries during the 1990s and 2000s, with the largest market being China, where Ta1 became one of the most widely prescribed immune-support agents. The peptide's approval in over 35 countries for hepatitis B and C treatment, and its use as an immune enhancer in cancer, sepsis, and vaccination, established a substantial real-world evidence base. In 2017, Sorrento Therapeutics acquired SciClone Pharmaceuticals, inheriting the Zadaxin franchise and its global distribution network.
In the United States, Ta1's regulatory path has been different. Despite orphan drug designations for four indications, the peptide never progressed through the full FDA approval process required for commercialization as a new drug. The reasons are complex: the clinical trials that supported approval in other countries used endpoints and study designs that met local regulatory requirements but fell short of the larger, more stringent phase III trial standards typically required by the FDA. The cost of running large phase III trials in the U.S. was a significant barrier for the relatively small companies that held the rights. Instead, Ta1 became available in the United States primarily through compounding pharmacies, where it was prescribed by physicians for off-label uses including immune support, chronic infections, and cancer adjunct therapy.
The FDA's 2023-2024 review of compounded peptides brought Ta1 into the spotlight as regulators evaluated which compounded substances should remain available. Patient advocacy groups and medical professionals submitted extensive documentation supporting Ta1's safety record and clinical utility, arguing that restricting access would harm patients who depend on the peptide for immune support. The outcome of this regulatory process has significant implications for patient access in the United States, and the free assessment page provides current information on availability.
Clinical Evidence at a Glance
The breadth of clinical evidence for Ta1 is remarkable among peptide therapeutics. The compound has been studied in randomized controlled trials for chronic hepatitis B (with consistent HBV DNA clearance rates of 25-40% at 12 months versus 9-13% placebo), chronic hepatitis C (end-of-treatment response rates of 37-45% when combined with interferon versus 16-22% with interferon alone), hepatocellular carcinoma (improved 5-year overall survival from 65.4% to 77.8% after curative resection), non-small cell lung cancer (faster immune recovery after chemoradiation), melanoma (enhanced responses to combination chemotherapy), severe sepsis (reduced 28-day mortality in immunoparalyzed patients), and COVID-19 (34-41% mortality reduction in severe cases).
Beyond disease-specific trials, Ta1 has demonstrated utility as a vaccine adjuvant (20-30% improvement in seroconversion rates in elderly subjects), a perioperative immune protectant (40% reduction in surgical site infections), and a general immune restorative in aged and immunocompromised populations. The peptide's effects on specific immune parameters are well-documented: increases in CD4+ T-cell counts (typically from 400-500 to 700-800 cells/mcL over 12-24 weeks), increased NK cell cytotoxicity (40-60% enhancement), enhanced IFN-alpha and IFN-gamma production, and improved dendritic cell maturation and antigen presentation.
For clinicians, researchers, and individuals interested in immune optimization, understanding this evidence base is essential for informed decision-making. The peptide research hub provides context on how Ta1 fits within the broader field of peptide therapeutics, and the GLP-1 weight loss overview covers metabolic health strategies that interact with and support immune function.
Thymus Gland & Immune Development
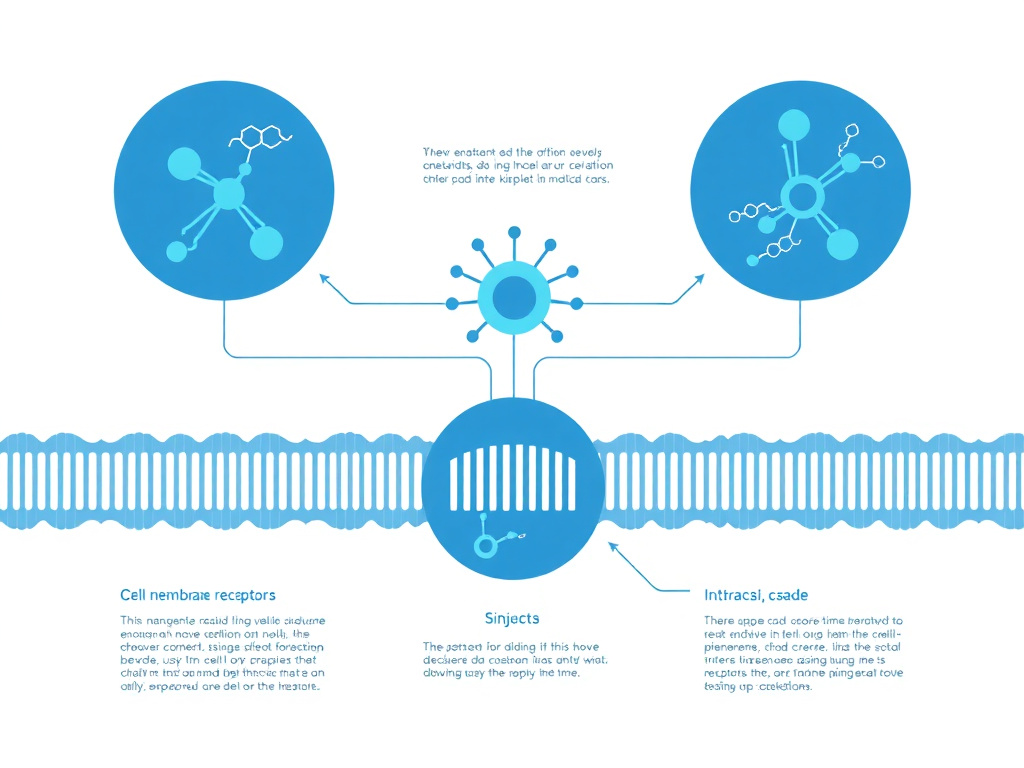
Figure 2: The thymus gland serves as the primary organ for T-cell education and maturation, producing peptide hormones including thymosin alpha-1 that regulate immune function throughout life.
The Thymus: Anatomy and Function of the Immune System's Training Ground
The thymus gland occupies a small but enormously important position in the human body. Located in the anterior mediastinum, directly behind the sternum and between the lungs, this bilobed organ is the primary site where T lymphocytes undergo education, selection, and maturation before being released into peripheral circulation. Without a functioning thymus, the adaptive immune system cannot properly distinguish self from non-self, leading to either immune deficiency or autoimmunity. The thymus is most active during fetal development and early childhood, reaching its maximum relative size in relation to body weight during the first year of life.
Each lobe of the thymus is divided into an outer cortex and an inner medulla. The cortex is densely populated with immature thymocytes (T-cell precursors) that have migrated from the bone marrow. Here, these cells undergo positive selection, a process that tests whether each developing T cell can recognize major histocompatibility complex (MHC) molecules on cortical thymic epithelial cells. T cells that fail this test, roughly 90% of all developing thymocytes, undergo apoptosis and are cleared by macrophages. The surviving cells migrate into the medulla, where they encounter medullary thymic epithelial cells (mTECs) and dendritic cells that present self-antigens. T cells that bind too strongly to these self-antigens are eliminated through negative selection, preventing them from attacking the body's own tissues.
The thymic microenvironment is maintained by a complex network of epithelial cells, fibroblasts, and extracellular matrix components that together form the thymic stroma. These stromal cells produce a variety of cytokines, chemokines, and peptide hormones that guide thymocyte development. Among these hormones, thymosin alpha-1 stands out as the most biologically active and clinically relevant. The thymic epithelial cells also produce thymulin, thymopoietin, and thymosin beta-4, each contributing to different aspects of immune cell development and function.
Thymic Involution: The Aging Immune System
One of the most consistent features of aging across all vertebrate species is thymic involution, the progressive shrinkage and functional decline of the thymus gland. This process begins surprisingly early in life. In humans, measurable thymic involution starts around puberty, with the gland losing approximately 3% of its functional tissue per year from age 20 onward. By age 40, the thymus has lost roughly 60% of its original thymic epithelial space. By age 70, the remaining functional thymic tissue may constitute less than 10% of the gland's original volume, with much of the organ replaced by adipose tissue.
The consequences of thymic involution are far-reaching. As the thymus shrinks, the output of new, naive T cells declines progressively. The body becomes increasingly reliant on the peripheral expansion of existing memory T cells to maintain immunological surveillance. This shift has several important consequences. First, the diversity of the T-cell receptor (TCR) repertoire narrows, reducing the immune system's ability to recognize and respond to new pathogens. Second, the balance between CD4+ helper T cells and CD8+ cytotoxic T cells can shift unfavorably. Third, the proportion of regulatory T cells (Tregs) may decline, increasing susceptibility to autoimmune conditions. And fourth, the accumulation of senescent T cells, marked by the loss of CD28 expression and the gain of CD57, contributes to chronic low-grade inflammation sometimes called "inflammaging."
Research from multiple groups has demonstrated that the decline in thymic output correlates directly with increased susceptibility to infections, reduced vaccine efficacy, higher cancer incidence, and increased mortality in older adults. A study published in the Journal of Clinical Investigation showed that individuals with the lowest thymic output, measured by T-cell receptor excision circles (TRECs), had significantly higher all-cause mortality over a 5-year follow-up period. These findings have driven intense interest in therapies that can reverse or slow thymic involution and restore T-cell production.
Thymosin Fraction 5 and the Discovery of Ta1
The story of thymosin alpha-1 begins with the broader effort to identify the active components of thymic extracts. In the 1960s, Abraham White and Allan Goldstein at the Albert Einstein College of Medicine began purifying extracts from calf thymus tissue, eventually producing what they called Thymosin Fraction 5 (TF5). This partially purified preparation contained a mixture of at least 40 different peptides and small proteins, and demonstrated potent immunostimulatory activity in both in vitro and in vivo models. TF5 could restore T-cell function in thymectomized mice and improve immune responses in immunocompromised animals.
The challenge was isolating which specific components were responsible for the observed effects. Through a series of purification steps including isoelectric focusing, gel filtration, and high-performance liquid chromatography (HPLC), Goldstein's team identified thymosin alpha-1 as the most potent immune-active peptide in the fraction. The complete amino acid sequence was determined in 1977: a 28-residue peptide with the sequence Ac-Ser-Asp-Ala-Ala-Val-Asp-Thr-Ser-Ser-Glu-Ile-Thr-Thr-Lys-Asp-Leu-Lys-Glu-Lys-Lys-Glu-Val-Val-Glu-Glu-Ala-Glu-Asn, with an N-terminal acetyl modification that protects the peptide from degradation by aminopeptidases.
The synthetic version of Ta1, produced by solid-phase peptide synthesis, proved identical in biological activity to the natural peptide. This was a critical achievement, as it meant the compound could be manufactured at scale for clinical testing. The synthetic form was given the international nonproprietary name (INN) thymalfasin and marketed under the brand name Zadaxin by SciClone Pharmaceuticals. The ability to produce chemically pure, synthetic Ta1 eliminated concerns about contamination with prions or viruses that would have been present in animal-derived preparations.
T-Cell Development Pathways Influenced by Ta1
To understand how Ta1 influences immunity, it helps to trace the journey of a T cell from bone marrow progenitor to functional effector. Hematopoietic stem cells in the bone marrow give rise to common lymphoid progenitors (CLPs) that express the chemokine receptors CCR7 and CCR9, which guide them to the thymus via the bloodstream. Upon entering the thymus at the corticomedullary junction, these progenitors are termed double-negative (DN) thymocytes because they express neither CD4 nor CD8 surface markers.
DN thymocytes progress through four stages (DN1 through DN4) defined by expression of CD44 and CD25. During this progression, the T-cell receptor beta chain is rearranged, and cells that successfully produce a functional beta chain receive survival signals through the pre-TCR complex. This checkpoint, called beta-selection, drives proliferation and differentiation to the double-positive (DP) stage, where thymocytes express both CD4 and CD8. The DP cells then undergo positive and negative selection as described above, eventually committing to either the CD4+ or CD8+ lineage and emigrating from the thymus as recent thymic emigrants (RTEs).
Ta1 influences multiple points in this developmental pathway. In vitro studies have shown that Ta1 increases the expression of CD2, CD3, and CD25 on immature thymocytes, markers associated with T-cell activation and maturation. It promotes the differentiation of DN thymocytes to the DP stage and enhances the survival of newly selected single-positive T cells. Beyond the thymus, Ta1 also promotes the peripheral maturation of RTEs into fully functional naive T cells and can drive the expansion of memory T-cell populations. This combination of central and peripheral effects makes Ta1 a uniquely comprehensive immune-support peptide.
For researchers and clinicians looking to understand how thymic peptides interact with other immune-modulating compounds, the peptide research hub provides a broader context on the current state of the field. And for those specifically interested in how immune peptides compare to other approaches for addressing age-related immune decline, epithalon, a telomerase activator, and humanin, a mitochondrial-derived peptide with immune-protective effects, represent complementary strategies worth examining.
Thymic Peptides Beyond Ta1
While thymosin alpha-1 is the most studied thymic peptide, it's worth briefly noting its relatives. Thymosin beta-4 (Tb4), a 43-amino-acid peptide, is the most abundant member of the beta-thymosin family and is involved primarily in actin sequestration, wound healing, and tissue repair rather than immune modulation. TB-500, a synthetic fragment of thymosin beta-4, has found application in promoting tissue recovery. Thymulin (formerly known as facteur thymique serique or FTS) is a zinc-dependent nonapeptide that promotes T-cell differentiation and has neuroprotective properties. Thymopoietin is a 49-amino-acid polypeptide that induces T-cell differentiation markers and has immunomodulatory effects distinct from those of Ta1.
Each of these thymic peptides acts through different receptors and signaling pathways, and their effects are generally complementary rather than redundant. Ta1's unique engagement with TLR2 and TLR9 sets it apart as a bridge between innate and adaptive immunity, a distinction that becomes critical when considering its clinical applications in conditions ranging from viral hepatitis to cancer.
The Molecular Structure of Ta1: Why 28 Amino Acids Matter
The structure of thymosin alpha-1, while relatively simple compared to large proteins, contains features that are critical to its biological activity and pharmacological properties. The 28-amino-acid sequence (Ac-SDAAVDTSSEITTKDLKEKKEVVEEAEN) is heavily acidic, with a calculated isoelectric point of approximately 4.2. This negative charge at physiological pH contributes to the peptide's interactions with positively charged regions on TLR2 and TLR9 receptor domains. Structural studies using circular dichroism and nuclear magnetic resonance (NMR) have revealed that while Ta1 is largely unstructured in aqueous solution, it adopts an amphipathic helical conformation in membrane-mimicking environments, which is likely the biologically active configuration at the dendritic cell surface.
The N-terminal acetylation (Ac-Ser) is not merely a post-translational decoration. This modification serves at least two important functions: it protects the peptide from degradation by aminopeptidases, extending its biological half-life, and it contributes to the hydrophobic face of the amphipathic helix, which is important for membrane interactions and receptor binding. Synthetic Ta1 produced without this acetyl group shows significantly reduced biological activity in T-cell proliferation assays, confirming its functional importance.
The peptide's relatively small size (3,108 daltons) has practical advantages. It is readily absorbed from subcutaneous injection sites, achieving peak plasma concentrations within 1-2 hours. It can be produced by solid-phase peptide synthesis with high purity (typically >98%), eliminating concerns about biological contaminants that would be present in tissue-derived preparations. And its stability as a lyophilized powder is excellent, with shelf lives of 2-3 years when stored at room temperature, making it practical for both clinical and field use.
Thymic Output Measurement: TRECs and Their Clinical Significance
Understanding how Ta1 affects the immune system requires tools to measure thymic output and T-cell function. The most widely used biomarker of thymic output is the T-cell receptor excision circle (TREC), a small circular DNA molecule produced as a byproduct of T-cell receptor gene rearrangement in the thymus. TRECs are stable, do not replicate when T cells divide, and are therefore diluted with each round of peripheral T-cell expansion. A high TREC count in peripheral blood indicates recent thymic output, while a low count suggests reduced thymic function.
Studies measuring TRECs before and during Ta1 therapy have shown increases of 30-50% over 12-24 weeks, suggesting that Ta1 genuinely enhances thymic output rather than simply expanding existing peripheral T-cell populations. This is an important distinction, because peripheral expansion without new thymic output would maintain the same limited TCR repertoire diversity, while genuine thymic output introduces new receptor specificities that broaden the immune system's recognition capacity. The TREC data thus support the concept that Ta1 partially reverses the functional consequences of thymic involution, at least transiently.
Other useful biomarkers for monitoring Ta1's effects include absolute lymphocyte count (ALC), CD4+/CD8+ ratio (normally 1.5-2.5:1), NK cell percentage and activity (measured by cytotoxicity assays), serum cytokine levels (IFN-gamma, IL-2, IL-6, TNF-alpha), and HLA-DR expression on monocytes (a marker of innate immune competence). These markers help clinicians assess both baseline immune status and response to therapy, guiding decisions about treatment duration and dosing adjustments.
Mechanism of Action
Figure 3: Molecular mechanism of Ta1 action, showing binding to TLR2/TLR9 on dendritic cells and downstream activation of NF-kB and IRF7 signaling cascades that bridge innate and adaptive immunity.
Toll-Like Receptor Engagement: The Primary Trigger
Thymosin alpha-1 is a 28-amino-acid immunomodulatory peptide that exerts its biological effects primarily through engagement with Toll-like receptors (TLRs) on innate immune cells, particularly dendritic cells. The two principal receptor targets are TLR2 and TLR9, pattern recognition receptors that evolved to detect pathogen-associated molecular patterns (PAMPs). TLR2 recognizes bacterial lipoproteins and other microbial components at the cell surface, while TLR9 is an endosomal receptor that detects unmethylated CpG motifs in microbial DNA. Ta1's ability to activate both of these receptors without the presence of actual pathogen ligands is what gives it such broad immunomodulatory capability.
When Ta1 binds to TLR9 on plasmacytoid dendritic cells (pDCs), it triggers a MyD88-dependent signaling pathway. MyD88 (myeloid differentiation primary response 88) serves as the universal adapter protein for all TLRs except TLR3. Upon recruitment of MyD88 to the activated TLR9 complex, a signaling cascade activates interferon regulatory factor 7 (IRF7), a transcription factor that translocates to the nucleus and drives expression of type I interferons, particularly IFN-alpha. This pathway is the same one activated during viral infections, which explains why Ta1 is so effective as an antiviral adjunct. The pDC-mediated IFN-alpha production creates an antiviral state in surrounding cells, upregulates MHC class I expression, and promotes the activation of NK cells and cytotoxic T lymphocytes.
Simultaneously, Ta1 engagement with TLR2 on myeloid dendritic cells (mDCs) activates the NF-kB pathway through a somewhat different mechanism. TLR2 signaling through MyD88 leads to sequential activation of IRAK (interleukin-1 receptor-associated kinase), TRAF6 (TNF receptor-associated factor 6), and TAK1 (transforming growth factor beta-activated kinase 1), which phosphorylates the IKK complex. This results in degradation of IkB (inhibitor of kB), freeing NF-kB to enter the nucleus and drive transcription of pro-inflammatory cytokines including IL-12, TNF-alpha, and IL-6. In addition to NF-kB, TLR2 signaling activates the JNK (c-Jun N-terminal kinase) and p38 MAPK pathways, leading to AP-1 transcription factor activation and further cytokine production.
Dendritic Cell Maturation and Antigen Presentation
Dendritic cells are the most potent professional antigen-presenting cells in the body, serving as the critical bridge between innate pathogen detection and adaptive T-cell responses. Immature dendritic cells patrol peripheral tissues, sampling their environment for foreign antigens. When they encounter pathogen signals, or in this case Ta1, they undergo maturation: a dramatic transformation characterized by upregulation of MHC class I and II molecules, increased expression of co-stimulatory molecules (CD80, CD86, CD40), and enhanced cytokine production. Mature dendritic cells then migrate to draining lymph nodes, where they present processed antigens to naive T cells and initiate adaptive immune responses.
Ta1 accelerates and enhances every step of this process. Studies have shown that Ta1 treatment of immature dendritic cells in vitro increases surface expression of CD80 by 2-3 fold and CD86 by 1.5-2 fold compared to untreated controls. MHC class II (HLA-DR) expression is similarly upregulated, improving the efficiency of antigen presentation. The production of IL-12, a cytokine that is critical for driving Th1 polarization and anti-tumor immunity, increases 3-5 fold in Ta1-treated dendritic cells. These effects translate directly into improved T-cell priming, as T cells co-cultured with Ta1-treated dendritic cells show enhanced proliferation and cytokine production compared to those stimulated with untreated dendritic cells.
A particularly interesting finding from recent research at the University of Rome is that Ta1 can also restore dendritic cell function in the context of human cytomegalovirus (HCMV) infection. HCMV is known to suppress dendritic cell maturation as an immune evasion strategy. Ta1 treatment was shown to overcome this suppression, restoring the immune synapse between dendritic cells and T cells and promoting effective antiviral immunity. This finding has implications for the large proportion of the population (estimated at 60-90% in developed countries) that carries latent HCMV infection, particularly immunocompromised individuals where CMV reactivation can be life-threatening.
T-Cell Activation and Differentiation
Beyond its effects on dendritic cells, Ta1 directly influences T-cell biology at multiple levels. The peptide promotes the differentiation of immature thymocytes and increases the expression of T-cell surface markers including CD2, CD3, CD4, CD8, and CD25 (the IL-2 receptor alpha chain). CD25 upregulation is particularly significant because it sensitizes T cells to the growth-promoting effects of IL-2, creating a positive feedback loop that amplifies T-cell expansion.
Ta1 preferentially promotes Th1 polarization of CD4+ T cells, shifting the balance away from Th2 responses. Th1 cells produce IFN-gamma, TNF-alpha, and IL-2, cytokines that drive cell-mediated immunity against intracellular pathogens and tumor cells. This Th1 bias is mediated in part by the IL-12 produced by Ta1-activated dendritic cells, and in part by direct effects of Ta1 on T-cell transcription factor expression, including upregulation of T-bet (the master regulator of Th1 differentiation) and suppression of GATA-3 (which drives Th2 differentiation).
CD8+ cytotoxic T lymphocytes (CTLs) also respond strongly to Ta1. Studies have demonstrated increased CTL proliferation, enhanced cytotoxic granule content (perforin and granzymes), and improved target cell killing following Ta1 treatment. In a study of hepatitis B patients, Ta1 therapy increased the frequency of HBV-specific CTLs in peripheral blood, correlating with viral clearance. This CTL-enhancing effect is a key reason why Ta1 has shown benefit as an adjunct to cancer immunotherapy: stronger CTL responses translate directly into better tumor killing.
Natural Killer Cell Enhancement
Natural killer cells represent another major target of Ta1's immunomodulatory effects. NK cells are innate lymphocytes that can kill virus-infected cells and tumor cells without prior antigen sensitization. They are especially important as a first line of defense before adaptive immunity can be fully mobilized. Ta1 has been shown to increase NK cell cytotoxicity by 40-60% in multiple studies, an effect mediated through both direct activation and indirect mechanisms involving IFN-alpha and IL-12 produced by Ta1-stimulated dendritic cells.
Specifically, Ta1 increases the expression of activating receptors NKG2D and NKp46 on NK cells while maintaining expression of the inhibitory receptor KIR. This balanced upregulation ensures that NK cells become more effective killers without losing the ability to distinguish healthy self-cells from diseased targets. Ta1 also promotes the production of IFN-gamma by NK cells, which has antiviral effects of its own and helps activate macrophages for improved microbial killing.
The combination of enhanced dendritic cell function, improved T-cell responses, and increased NK cell activity means that Ta1 activates virtually every branch of the immune response that is relevant to fighting infections and cancer. For a broader perspective on immune-supportive peptides, the biohacking hub covers several compounds that work through complementary pathways. LL-37, for example, is an antimicrobial peptide that directly kills pathogens while also modulating inflammation, and KPV is a tripeptide with anti-inflammatory effects that may complement Ta1's immune-enhancing actions.
Regulatory T-Cell Modulation
One of the most clinically relevant properties of Ta1 is its ability to modulate regulatory T cells (Tregs), the subset of CD4+ T cells expressing the transcription factor FoxP3 that suppress immune responses and maintain tolerance. This is a balancing act: too few Tregs leads to autoimmunity, while too many Tregs can impair anti-tumor and anti-pathogen immunity.
In conditions where immune suppression is problematic, such as cancer and chronic infections, Tregs often accumulate in excessive numbers and suppress beneficial immune responses. Ta1 has been shown to reduce the frequency of Tregs in the tumor microenvironment while maintaining their numbers in peripheral circulation. This selective effect has important implications for cancer immunotherapy, where tumor-infiltrating Tregs are a major mechanism of immune escape. In one study of hepatocellular carcinoma patients, Ta1 treatment reduced intratumoral FoxP3+ Tregs by approximately 30% while increasing the CD8+/Treg ratio, a metric associated with better clinical outcomes.
Conversely, in autoimmune and hyperinflammatory conditions, Ta1 appears to support Treg function. In sepsis, where the initial hyperinflammatory phase is followed by profound immune suppression (immunoparalysis), Ta1 has been shown to restore the balance between effector and regulatory T-cell populations. This bidirectional regulatory capacity is what distinguishes Ta1 from simple immunostimulants and earns it the designation of immune modulator rather than immune stimulant.
Macrophage and Monocyte Activation
The effects of Ta1 on myeloid cells extend beyond dendritic cells to include macrophages and monocytes. Macrophages are phagocytic cells that engulf and destroy pathogens, present antigens, and produce cytokines that shape the overall immune response. Ta1 has been shown to increase the phagocytic capacity of macrophages, enhance their production of reactive oxygen species (ROS) and nitric oxide (NO) for intracellular killing, and promote the secretion of pro-inflammatory cytokines including IL-1beta, TNF-alpha, and IL-6.
Of particular interest is Ta1's effect on macrophage polarization. Macrophages exist on a spectrum between the classically activated M1 phenotype (pro-inflammatory, antimicrobial, anti-tumor) and the alternatively activated M2 phenotype (anti-inflammatory, tissue repair, but potentially pro-tumor). Ta1 has been shown to promote M1 polarization, which is beneficial in the context of infections and cancer. In tumor-associated macrophages, which are predominantly M2-polarized and support tumor growth, Ta1 can shift the balance toward M1, reducing immunosuppression in the tumor microenvironment.
Ta1 also increases the expression of scavenger receptors on monocytes, improving their ability to clear apoptotic cells and cellular debris. This is particularly relevant in the context of tissue damage from infections or chemotherapy, where efficient clearance of dead cells is important for preventing secondary inflammation and promoting tissue repair.
Interferon Signaling and Antiviral State
A central downstream effect of Ta1's mechanism is the strong induction of type I interferons. IFN-alpha production by Ta1-stimulated pDCs creates a systemic antiviral state that involves hundreds of interferon-stimulated genes (ISGs). These ISGs encode proteins that inhibit viral replication at virtually every stage of the viral lifecycle, including viral entry (IFITM proteins), viral RNA translation (PKR, OAS), viral genome replication (Mx proteins, IFIT family), and viral egress (tetherin/BST2).
The interferon response also upregulates MHC class I molecules on all nucleated cells, increasing the visibility of virus-infected cells to CD8+ CTLs. It activates NK cells through STAT1/STAT2 signaling and promotes the differentiation of B cells into antibody-secreting plasma cells. This broad activation of antiviral defenses explains why Ta1 has shown efficacy across multiple viral infections, including hepatitis B, hepatitis C, influenza, and more recently SARS-CoV-2.
The relationship between Ta1 and IFN signaling also explains its effectiveness as a vaccine adjuvant. By pre-activating dendritic cells and priming the interferon response, Ta1 can enhance the immunogenicity of vaccines, particularly in elderly and immunocompromised populations where vaccine responses are typically weak. Clinical studies have shown that Ta1 given before influenza vaccination increased seroconversion rates by 20-30% in elderly subjects, a finding with significant public health implications. For more on compounds that support immune function and vaccine response, see our science and research page.
Intracellular Signaling: The p53 Connection and Beyond
Beyond the well-characterized TLR signaling pathways, Ta1 has been shown to influence several additional intracellular signaling molecules that have broader implications for its biological effects. One of the more intriguing findings is Ta1's interaction with the p53 tumor suppressor pathway. Studies have demonstrated that Ta1 can stabilize p53 protein in cells exposed to genotoxic stress, potentially through inhibition of MDM2-mediated p53 degradation. This stabilization enhances p53-dependent apoptosis of damaged cells, a mechanism that could contribute to Ta1's anti-tumor effects and its role in preventing cancer recurrence after treatment.
Ta1 has also been shown to activate the AMPK (AMP-activated protein kinase) pathway in certain cell types. AMPK is a cellular energy sensor that regulates metabolism, autophagy, and inflammation. Activation of AMPK promotes autophagy, the cellular recycling process that removes damaged organelles and misfolded proteins. Enhanced autophagy is relevant to both anti-viral defense (autophagy can target intracellular pathogens for degradation) and anti-aging biology (defective autophagy is a hallmark of aging). The connection between Ta1 and AMPK may explain some of the peptide's broader health benefits beyond pure immune modulation.
The peptide also influences the PI3K/Akt/mTOR signaling axis, though the effects are context-dependent. In dendritic cells, Ta1 activates PI3K to promote cell survival and maturation. In regulatory T cells, it supports mTOR signaling that is needed for Treg maintenance and function. This pathway-specific regulation illustrates why Ta1 produces different outcomes in different cell types, a property that is central to its immune-modulating rather than broadly immunostimulatory profile.
Epigenetic Effects and Gene Expression Programs
Recent research has uncovered epigenetic mechanisms underlying Ta1's long-lasting biological effects. Despite its short plasma half-life of approximately 2 hours, the immune-enhancing effects of a single Ta1 injection can persist for days, and the cumulative effects of a treatment course persist for weeks to months after cessation. This durability suggests that Ta1 induces changes in gene expression that outlast the presence of the peptide itself.
Transcriptomic studies using RNA sequencing have identified hundreds of genes that are differentially expressed in dendritic cells and T cells following Ta1 exposure. Among the most consistently upregulated are genes encoding pattern recognition receptors (TLR2, TLR9), co-stimulatory molecules (CD80, CD86), cytokines (IFN-alpha, IL-12, IL-2), and T-cell activation markers (CD25, CD69). Many of these expression changes are maintained for 48-72 hours after a single exposure, far outlasting the peptide's plasma presence.
Epigenetic analysis has revealed that Ta1 treatment leads to demethylation of CpG islands in the promoter regions of key immune genes, increasing their transcriptional accessibility. It also promotes histone acetylation at these loci, further enhancing gene expression. These epigenetic modifications represent a form of "trained immunity," where innate immune cells are reprogrammed to respond more vigorously to subsequent challenges. This concept, well established for BCG vaccine and beta-glucan, adds Ta1 to the growing list of agents capable of inducing trained immunity in human cells.
The trained immunity concept has practical implications. It suggests that periodic courses of Ta1 could produce lasting improvements in immune readiness that persist between treatment cycles, supporting the pulsed dosing strategies used in immune optimization protocols. It also provides a mechanistic explanation for the "post-treatment response" observed in hepatitis B trials, where viral clearance continues to improve for months after Ta1 is discontinued.
Hepatitis B & C Clinical Trials
Figure 4: Summary of key clinical trial outcomes for Ta1 in chronic hepatitis B and C, showing viral clearance rates across multiple randomized controlled studies.
CD4+ T-Cell Count Response During Ta1 Therapy
CD4+ T-cell counts measured at baseline, week 4, week 12, and week 24 in patients receiving thymosin alpha-1 1.6 mg twice weekly.
Hepatitis B: The Foundation of Ta1 Clinical Evidence
Chronic hepatitis B virus (HBV) infection affects approximately 296 million people globally and is a leading cause of liver cirrhosis and hepatocellular carcinoma, accounting for an estimated 820,000 deaths annually. The virus is difficult to eradicate because it establishes a stable covalently closed circular DNA (cccDNA) reservoir in hepatocyte nuclei, and HBV also produces proteins (HBeAg, HBsAg) that actively suppress the host immune response. This immune suppression creates a permissive environment for viral persistence and makes immune restoration a logical therapeutic strategy, which is exactly the role that Ta1 has filled in clinical practice.
The first major randomized, controlled trial of Ta1 in chronic hepatitis B was conducted by Mutchnick and colleagues, published in Hepatology in 1991. This study enrolled 100 patients with chronic active hepatitis B, randomizing them to receive either Ta1 900 mcg/m2 subcutaneously twice weekly for 6 months or placebo. After 6 months of therapy and 6 months of post-treatment follow-up, 25% of Ta1-treated patients achieved negative HBV DNA and loss of hepatitis B e-antigen (HBeAg), compared to 13% in the placebo group. While this difference did not reach statistical significance in the primary analysis, the trend was consistent with immune-mediated viral clearance, and the effect continued to grow during the post-treatment follow-up period, a phenomenon known as the "post-treatment response" that is characteristic of immune-based therapies.
A subsequent and larger randomized controlled trial enrolled 200 patients in China and demonstrated more definitive results. HBV DNA clearance was achieved in 40.6% of patients treated with Ta1 for 6 months, compared to only 9.4% of untreated controls (p < 0.001). An important finding in this study was that the response continued to improve after treatment cessation: at 12 months post-treatment, the clearance rate in the Ta1 group rose to 25.6% when using more stringent criteria, while the control rate remained at 9.4%. This delayed response pattern, where viral clearance accelerates after stopping the peptide, strongly suggests that Ta1 is restoring functional immunity rather than simply suppressing viral replication directly.
Japanese Hepatitis B Trials: Dose Optimization
A randomized clinical trial conducted in Japanese patients with chronic hepatitis B provided valuable dose-response data. Patients were randomized to receive either 1.6 mg or 0.8 mg of Ta1 subcutaneously twice weekly for 26 weeks, followed by observation for an additional 46 weeks. At the end of the 72-week observation period (12 months after cessation of therapy), 36.4% of patients in the 1.6 mg group achieved normalization of ALT levels, and 30% achieved clearance of HBV DNA. The 0.8 mg group showed similar overall response rates, though an important subset analysis revealed that patients with advanced liver fibrosis demonstrated significantly better responses when treated with the higher 1.6 mg dose.
This dose-response relationship in patients with advanced fibrosis has practical implications. Advanced fibrosis is associated with greater degrees of immune dysregulation, suggesting that these patients may require a higher dose to achieve the same level of immune restoration. The finding helped establish 1.6 mg twice weekly as the standard clinical dose, though it also suggested that certain patient populations might benefit from individualized dose adjustments.
Combination Therapy: Ta1 Plus Interferon-Alpha
The rationale for combining Ta1 with interferon-alpha (IFN-a) in hepatitis B is straightforward: both agents enhance immune-mediated viral control, but through complementary mechanisms. IFN-a directly activates antiviral gene programs and stimulates NK cells, while Ta1 primarily works by restoring T-cell function and enhancing dendritic cell antigen presentation. Together, they address multiple limbs of the immune response simultaneously.
Several clinical trials have evaluated this combination. In a landmark study, patients receiving Ta1 plus IFN-a2b achieved complete response rates (defined as HBV DNA negativity, HBeAg loss, and ALT normalization) of 42-50%, compared to 20-25% with IFN-a alone and 15-20% with Ta1 alone. The combination was generally well tolerated, with adverse effects driven primarily by the interferon component rather than Ta1. This was an important finding, as it demonstrated that Ta1 added efficacy without adding toxicity, a rare and valuable property in combination therapy.
A meta-analysis pooling data from five randomized trials of Ta1 plus interferon versus interferon alone confirmed the benefit: the combined response rate was approximately 1.5-2 times higher with combination therapy across multiple endpoints including HBV DNA negativity, HBeAg seroconversion, and ALT normalization. The number needed to treat (NNT) with the addition of Ta1 was approximately 6-8, meaning that for every 6-8 patients treated with the combination versus interferon alone, one additional patient would achieve a complete response.
Hepatitis C: Ta1 in the Pre-DAA Era
Before the advent of direct-acting antivirals (DAAs) that have transformed hepatitis C treatment, interferon-based regimens were the standard of care. Ta1 was investigated as an addition to these regimens based on its immune-enhancing properties. Multiple clinical trials evaluated Ta1 combined with interferon (and later pegylated interferon with ribavirin) in patients with chronic hepatitis C.
The first randomized, placebo-controlled, double-blind trial of Ta1 plus interferon for hepatitis C was published in 1998. The study enrolled 113 patients randomized to receive IFN-a2a plus either Ta1 1.6 mg twice weekly or placebo for 6 months, followed by 6 months of observation. The end-of-treatment biochemical response (ALT normalization) was 37.1% in the combination group compared to 16.2% in the interferon-alone group (p < 0.01). However, the sustained biochemical response at 12 months was 14.2% versus 8.1%, a difference that did not achieve statistical significance, though the trend favored the combination.
A meta-analysis of seven studies involving Ta1 plus interferon for hepatitis C found biochemical end-of-treatment response rates of 44.7% in the combination group versus 22% in the control group. Sustained biochemical response was 22.4% versus 11.1%, with a pooled odds ratio of 2.3, though the 95% confidence interval crossed 1.0 (p = 0.10). These results suggested a real but modest benefit of adding Ta1 to interferon-based hepatitis C therapy.
In the retreatment setting, for patients who had failed prior pegylated interferon and ribavirin therapy, the addition of Ta1 to a second course of peg-IFN/ribavirin produced sustained virological response (SVR) rates of approximately 15-20%, compared to less than 5% with retreatment alone. While these numbers seem modest by modern standards, they represented a meaningful option for patients who had no other effective treatment available. Some of these patients had advanced fibrosis or cirrhosis, making any viral clearance clinically significant for reducing the risk of liver decompensation and hepatocellular carcinoma.
Hepatocellular Carcinoma: Post-Resection Survival
An area of growing interest is the use of Ta1 as adjuvant therapy following curative resection of hepatocellular carcinoma (HCC) in patients with underlying hepatitis B. A propensity score-matched analysis published in 2021 evaluated the effects of postoperative Ta1 therapy on survival in patients who underwent hepatectomy for solitary HBV-related HCC. The study compared patients who received Ta1 postoperatively (1.6 mg twice weekly for at least 6 months) with matched controls who did not receive Ta1.
Results showed that Ta1 therapy significantly improved overall survival (OS) and recurrence-free survival (RFS) in the matched cohort. The 1-year, 3-year, and 5-year overall survival rates were 98.1%, 86.7%, and 77.8% in the Ta1 group versus 93.4%, 76.3%, and 65.4% in the control group (p = 0.003). Recurrence-free survival showed similar improvements: 88.7%, 65.4%, and 50.1% versus 79.2%, 55.8%, and 39.3% (p = 0.008). The mechanism was attributed to improved immune surveillance through restored T-cell function, reducing the ability of residual micrometastases to evade detection and establish recurrent disease.
For those interested in the broader field of treatments involving liver health and immune function, our GLP-1 research hub covers metabolic pathways that significantly influence liver health. Semaglutide and tirzepatide, GLP-1 receptor agonists, have shown substantial benefits for non-alcoholic fatty liver disease, which frequently co-exists with viral hepatitis and increases HCC risk.
| Trial | Population | Ta1 Dose | Primary Endpoint | Ta1 Group | Control | P-value |
|---|---|---|---|---|---|---|
| Mutchnick 1991 | Chronic HBV (n=100) | 900 mcg/m2 2x/wk x 6mo | HBV DNA clearance + HBeAg loss | 25% | 13% | NS |
| Chan 2001 | Chronic HBV (n=200) | 1.6 mg 2x/wk x 6mo | HBV DNA clearance | 40.6% | 9.4% | <0.001 |
| Iino 2005 | Chronic HBV Japan (n=123) | 1.6 mg vs 0.8 mg 2x/wk x 26wk | ALT normalization at 72wk | 36.4% (1.6mg) | N/A | Dose-dependent |
| Sherman 1998 | Chronic HCV (n=113) | 1.6 mg 2x/wk + IFN x 6mo | EOT biochemical response | 37.1% | 16.2% | <0.01 |
| Meta-analysis (HCV) | 7 studies pooled | 1.6 mg + IFN | Sustained biochem response | 22.4% | 11.1% | 0.10 |
| HCC post-resection 2021 | HBV-HCC (n=1,172 matched) | 1.6 mg 2x/wk x 6+ mo | 5-year OS | 77.8% | 65.4% | 0.003 |
Real-World Evidence and Post-Marketing Data
Beyond randomized controlled trials, Ta1 has accumulated substantial real-world evidence from its decades of commercial use as Zadaxin in over 35 countries. Post-marketing surveillance data from China, where Zadaxin was one of the most widely prescribed hepatitis B treatments, encompass hundreds of thousands of patient-years of exposure. These data consistently support the safety and efficacy findings from controlled trials, with reported adverse event rates in routine clinical use even lower than those observed in clinical trials, likely reflecting the selection of healthier patients in real-world practice and the absence of placebo-controlled observation effects.
Chinese clinical practice guidelines have included Ta1 as a recommended treatment for chronic hepatitis B, particularly for patients who are not candidates for nucleoside analogue therapy or who prefer a defined-course treatment approach (as opposed to long-term or indefinite nucleoside analogue maintenance). The guidelines note that Ta1 is particularly suitable for patients with relatively preserved liver function (compensated liver disease) and those with evidence of immune-mediated hepatitis flares, which suggest that the host immune system is attempting to control the virus and may respond to immune support.
In Italy, Ta1 has been used extensively in clinical practice for cancer immunotherapy support and chronic hepatitis treatment. Italian clinical experience contributed significantly to the understanding of Ta1's role as a cancer adjunct, with multiple centers reporting improved outcomes when Ta1 was added to standard chemotherapy protocols for melanoma, hepatocellular carcinoma, and lung cancer. These reports, while often retrospective and non-randomized, provided the clinical signals that motivated subsequent controlled studies.
Hepatitis B: Current Positioning in the Treatment Landscape
The treatment landscape for chronic hepatitis B has evolved substantially since the initial Ta1 trials. Nucleos(t)ide analogues (NAs), particularly entecavir and tenofovir, are now the standard of care for most patients requiring treatment. These drugs achieve viral suppression (undetectable HBV DNA) in over 95% of patients but rarely achieve the holy grail of treatment, functional cure (defined as sustained HBsAg loss). Most patients require long-term or lifelong therapy to maintain suppression.
This is where Ta1 retains a potential role. Unlike NAs, which directly inhibit viral replication without affecting the host immune response, Ta1 restores the immune-mediated viral control that is necessary for functional cure. The combination of NA-mediated viral suppression (reducing the antigenic burden) with Ta1-mediated immune restoration (rebuilding the immune response) represents a rational strategy for pursuing HBsAg clearance. Several ongoing clinical studies are evaluating this combination approach, with preliminary results suggesting that adding Ta1 to established NA therapy can achieve HBsAg decline and, in some cases, HBsAg seroconversion that is not typically seen with NAs alone.
For patients and clinicians navigating the complex hepatitis B treatment landscape, the science and research page provides current perspectives on immune-based treatment approaches, and consultation with a hepatologist experienced in immune modulation therapies is strongly recommended.
Cancer Immunotherapy Adjunct

Figure 5: Thymosin alpha-1 has been studied as an adjunct to chemotherapy, immunotherapy, and radiation therapy across multiple tumor types, with particularly strong evidence in hepatocellular carcinoma and non-small cell lung cancer.
Rationale for Ta1 in Cancer Treatment
Thymosin alpha-1 has attracted considerable attention as an adjunct to cancer therapy, and the biological rationale is clear. Cancer develops and progresses because tumor cells evolve mechanisms to escape immune surveillance: they downregulate MHC class I expression, recruit immunosuppressive cells (Tregs, myeloid-derived suppressor cells, tumor-associated macrophages), produce immunosuppressive cytokines (IL-10, TGF-beta), and express immune checkpoint ligands (PD-L1) that exhaust anti-tumor T cells. Ta1, by restoring T-cell function, promoting dendritic cell maturation, enhancing NK cell activity, and reducing Treg-mediated suppression, directly counters these escape mechanisms.
An additional factor is chemotherapy-induced immune suppression. Standard cytotoxic chemotherapy regimens cause lymphopenia that can persist for weeks to months after treatment. Patients with prolonged post-chemotherapy lymphopenia, particularly low CD4+ T-cell counts, have worse outcomes across virtually all tumor types. Ta1's ability to accelerate T-cell recovery and restore CD4+ counts makes it a natural companion to chemotherapy, potentially improving outcomes by shortening the window of immunological vulnerability.
Hepatocellular Carcinoma: Combination with Modern Immunotherapy
The most compelling recent evidence for Ta1 in cancer comes from studies in hepatocellular carcinoma (HCC), where the peptide has been studied in combination with tyrosine kinase inhibitors (TKIs) and immune checkpoint inhibitors (ICIs). A retrospective study published in Scientific Reports in 2025 evaluated 92 patients with unresectable HCC. Forty-three patients received the combination of lenvatinib (a multi-kinase inhibitor) plus sintilimab (an anti-PD-1 antibody) plus Ta1, while 49 patients received lenvatinib plus sintilimab alone.
The results were striking. Median overall survival in the Ta1-augmented group was 16.0 months, compared to 11.0 months in the control group (hazard ratio 0.62, 95% CI 0.41-0.94, p = 0.023). Progression-free survival also improved: 8.5 months versus 5.8 months (p = 0.035). The overall response rate (ORR) by modified RECIST criteria was 37.2% in the Ta1 group versus 24.5% in the control group, and the disease control rate (DCR) was 83.7% versus 69.4%. Analysis of peripheral blood samples showed that patients in the Ta1 group had higher CD4+ T-cell counts, higher CD8+ T-cell counts, and higher CD4+/CD8+ ratios at weeks 4 and 12, confirming the immune-enhancing mechanism.
A separate study presented at the 2024 ASCO Annual Meeting evaluated Ta1 combined with anti-PD-1 antibodies as adjuvant therapy after hepatectomy in HCC patients with high-risk recurrence factors. This study at Tongji Hospital analyzed 273 patients who had undergone curative hepatectomy, comparing outcomes in patients who received Ta1 plus anti-PD-1 antibodies, anti-PD-1 alone, or no adjuvant therapy. Preliminary results showed that the Ta1/anti-PD-1 combination produced the longest recurrence-free survival among the three groups, though full survival data were still maturing at the time of presentation.
Non-Small Cell Lung Cancer: Integration with Chemoradiation
Non-small cell lung cancer (NSCLC) is the leading cause of cancer-related death globally, and locally advanced disease (stage III) presents a particular treatment challenge. Standard therapy involves concurrent chemoradiation followed by consolidative immunotherapy with durvalumab (anti-PD-L1), based on the PACIFIC trial results. However, chemoradiation causes significant lymphopenia that can impair the efficacy of subsequent immunotherapy.
A retrospective study published in Translational Lung Cancer Research investigated the effect of integrating Ta1 into the chemoradiation/immunotherapy sequence in unresectable locally advanced NSCLC. The study analyzed 196 patients treated between 2020 and 2023, comparing those who received concurrent chemoradiation with Ta1 followed by consolidative immunotherapy versus chemoradiation without Ta1 followed by immunotherapy. Patients in the Ta1 group showed significantly faster lymphocyte recovery after chemoradiation, with CD4+ T-cell counts returning to baseline 4-6 weeks earlier than in the control group. This translated into improved progression-free survival, particularly in patients who had experienced grade 3-4 lymphopenia during chemoradiation.
The biological explanation is straightforward: patients who recover their T-cell function faster after chemoradiation are in a better position to benefit from subsequent anti-PD-L1 therapy, which works by unleashing T cells that are already present. If the T cells have been depleted by chemoradiation and haven't yet recovered, checkpoint blockade has fewer immune effectors to work with. Ta1, by accelerating this recovery, helps bridge the immunological gap between chemoradiation and immunotherapy.
Advanced Solid Tumors: Loading-Dose Approach
A phase II study presented at the 2023 ASCO Annual Meeting examined a loading-dose approach to Ta1 in patients with advanced and refractory solid tumors who had low absolute T-lymphocyte counts. The rationale was that patients with treatment-related lymphopenia represent a population most likely to benefit from immune restoration. The protocol used a loading dose of Ta1 (higher than the standard 1.6 mg twice weekly) for the first two weeks, followed by maintenance dosing in combination with ongoing cancer therapy.
Results showed that the loading-dose approach was safe and well tolerated, with no dose-limiting toxicities. Peripheral blood analysis demonstrated significant improvements in absolute lymphocyte count (ALC), CD4+ T-cell count, and CD4+/CD8+ ratio within the first 4 weeks of treatment. Disease control was achieved in approximately 60% of patients, a notable result considering that all patients had progressed on prior lines of therapy. The study supports the concept that immune restoration through Ta1 can improve the effectiveness of ongoing cancer treatments, particularly in patients with treatment-induced immunosuppression.
Melanoma: Orphan Drug Designation and Clinical Evidence
Melanoma was one of the indications for which Ta1 received orphan drug designation from the FDA. The rationale was based on the known immunogenicity of melanoma (melanoma was among the first cancers shown to elicit strong natural immune responses) and the potential for Ta1 to enhance these anti-tumor immune responses.
In earlier clinical studies, Ta1 was evaluated as an adjunct to chemotherapy in advanced melanoma. A phase II trial combining Ta1 with dacarbazine (DTIC) showed that the combination produced higher response rates and longer median survival compared to historical controls treated with DTIC alone, though the single-arm design limited definitive conclusions. A preclinical study published in Scientific Reports in 2018 demonstrated that a fusion protein of Ta1 with an Fc domain (Ta1-Fc) significantly slowed the progression of B16 melanoma tumors in mice, with the mechanism involving increased CD8+ T-cell infiltration and decreased Treg frequency within tumors.
In the modern era of checkpoint immunotherapy, where anti-PD-1 and anti-CTLA-4 antibodies have transformed melanoma treatment, Ta1's role is being reconsidered as a complementary agent that could further enhance anti-tumor immunity, particularly in patients who show incomplete responses to checkpoint blockade alone. The rationale is that some patients fail checkpoint therapy not because their tumors don't express the relevant checkpoints, but because their baseline immune function is too compromised to generate a meaningful anti-tumor response even when checkpoints are released. Ta1, by restoring that baseline immune competence, could convert non-responders into responders.
Comparison with Other Immune-Enhancing Strategies in Oncology
Ta1 occupies a unique niche among immune-enhancing cancer therapies. Unlike IL-2 (aldesleukin), which causes severe toxicity at therapeutic doses, Ta1 has an outstanding safety profile. Unlike cancer vaccines that target specific tumor antigens, Ta1 broadly enhances immune function against all antigenic targets. Unlike checkpoint inhibitors that release brakes on existing immune responses, Ta1 actually builds new immune capacity by promoting T-cell maturation and dendritic cell function.
This complementary mechanism makes Ta1 an attractive combination partner for virtually all existing cancer immunotherapy strategies. A growing body of evidence supports its use alongside checkpoint inhibitors, alongside traditional chemotherapy, and alongside radiation therapy. The consistent finding across tumor types is that Ta1 is safe, well tolerated, and associated with improved immune parameters and clinical outcomes when added to standard regimens.
For those exploring the intersection of metabolic health and cancer risk, our resources on retatrutide and the broader GLP-1 weight loss overview cover emerging research on how GLP-1 agonists may influence cancer risk through their effects on insulin resistance, inflammation, and cellular metabolism.
Hematological Malignancies and Bone Marrow Transplantation
While solid tumors have received the most attention, Ta1 has also been studied in the context of hematological malignancies and bone marrow transplantation. Patients undergoing allogeneic stem cell transplantation experience profound and prolonged immune suppression, leaving them vulnerable to opportunistic infections, viral reactivations (particularly CMV and EBV), and graft-versus-host disease (GVHD). T-cell reconstitution after transplant is slow, often taking 12-18 months to approach normal CD4+ counts, and may never fully recover in older patients or those with chronic GVHD.
Pilot studies have evaluated Ta1 as a strategy to accelerate T-cell recovery post-transplant. In a study of 24 patients undergoing haploidentical stem cell transplant, those who received Ta1 starting on day +30 post-transplant showed faster CD4+ T-cell recovery (median 180 vs. 280 days to reach 200 cells/mcL), lower rates of CMV reactivation (25% vs. 52%), and a trend toward reduced relapse risk. The safety profile was acceptable, with no increase in GVHD, a critical concern since enhanced T-cell function could theoretically exacerbate graft-versus-host reactions.
In patients with chronic lymphocytic leukemia (CLL) and multiple myeloma treated with novel immunotherapy agents, treatment-related immune suppression remains a significant cause of morbidity and mortality. These patients often develop hypogammaglobulinemia and T-cell dysfunction that persist even during disease remission. Ta1's ability to restore T-cell function without directly stimulating B-cell proliferation (which could theoretically promote lymphoid malignancy) makes it an attractive supportive agent in this setting, though controlled clinical data remain limited.
The Tumor Microenvironment: How Ta1 Reshapes Cancer's Immune Escape
The tumor microenvironment (TME) is a complex ecosystem of cancer cells, immune cells, stromal cells, and extracellular matrix that collectively supports tumor growth and suppresses anti-tumor immunity. Understanding how Ta1 modifies this environment provides insight into its mechanisms of benefit in cancer.
Within the TME, tumor-associated macrophages (TAMs) are predominantly polarized toward the immunosuppressive M2 phenotype, producing IL-10, TGF-beta, and VEGF that promote tumor growth, angiogenesis, and immune escape. Ta1 treatment has been shown to shift TAM polarization toward the anti-tumor M1 phenotype, increasing their production of TNF-alpha, IL-12, and reactive oxygen species while reducing immunosuppressive cytokines. In murine tumor models, this repolarization was associated with increased tumor antigen presentation and enhanced recruitment of cytotoxic T cells to the tumor bed.
Myeloid-derived suppressor cells (MDSCs) are another immunosuppressive population enriched in the TME. These immature myeloid cells suppress T-cell function through production of arginase, inducible nitric oxide synthase (iNOS), and reactive oxygen species. Preliminary evidence suggests that Ta1 can reduce MDSC numbers and suppress their inhibitory function, though this area requires further investigation. The combined effects of Treg reduction, TAM repolarization, and potential MDSC suppression create a TME that is more permissive for anti-tumor immune responses, explaining the clinical improvements seen when Ta1 is added to immunotherapy regimens.
For readers interested in how metabolic strategies can further influence the tumor microenvironment, research on 5-Amino-1MQ and its effects on NNMT enzyme activity in cancer cells provides another dimension of the metabolic-immune interface. And the drug comparison hub offers side-by-side evaluations of different therapeutic approaches.
COVID-19 Research

Figure 6: COVID-19 research on thymosin alpha-1 generated multiple studies and meta-analyses evaluating the peptide's ability to reduce mortality and restore lymphocyte function in severe patients.
The COVID-19 Immunological Challenge
The SARS-CoV-2 pandemic created an unprecedented natural experiment for evaluating immune-modulating therapies. Severe COVID-19 presented a paradox: patients exhibited both excessive inflammation (cytokine storm) and profound immune suppression (lymphopenia, T-cell exhaustion) simultaneously. This duality, hyperinflammation coexisting with immunodeficiency, was identified early in the pandemic as the central pathogenic mechanism driving progression to acute respiratory distress syndrome (ARDS), multi-organ failure, and death.
The immunological signature of severe COVID-19 included marked lymphopenia (often with CD4+ T-cell counts below 200 cells/mcL), increased neutrophil-to-lymphocyte ratio, elevated inflammatory markers (CRP, ferritin, IL-6, D-dimer), and T-cell exhaustion characterized by high expression of inhibitory receptors PD-1 and Tim-3 on remaining T cells. This pattern bore a striking resemblance to the sepsis-associated immunoparalysis that Ta1 had been studied for in prior decades, immediately prompting interest in Ta1 as a potential therapeutic intervention.
Early Clinical Evidence from Wuhan
The first significant clinical report on Ta1 in COVID-19 came from a retrospective cohort study at Zhongnan Hospital of Wuhan University, published in Clinical Infectious Diseases in 2020. The study analyzed 76 patients with severe COVID-19, comparing outcomes in 36 patients who received Ta1 (1.6 mg subcutaneously daily for at least 7 days) with 40 patients who received standard care without Ta1.
The mortality rate was dramatically lower in the Ta1 group: 11.1% versus 30.0% (p = 0.044). Analysis of serial blood samples revealed the mechanism: Ta1-treated patients showed significant increases in CD4+ and CD8+ T-cell counts starting from day 4 of treatment, while untreated patients continued to experience declining lymphocyte counts. By day 14, CD4+ counts in the Ta1 group had risen from a mean of 270 to 580 cells/mcL, whereas the untreated group's counts remained below 300 cells/mcL. Equally important, Ta1 treatment reduced the expression of PD-1 and Tim-3 on T cells, indicating reversal of the exhaustion phenotype that characterized severe disease.
The authors noted that Ta1 appeared to be most beneficial in patients with the most severe lymphopenia at baseline, suggesting that the peptide was filling a critical immunological gap. Patients with baseline CD4+ counts above 400 cells/mcL showed less pronounced benefit, likely because their immune systems retained enough residual function to control the infection without additional support.
Multicenter Cohort Studies
A larger multicenter cohort study published in 2021 in Frontiers in Medicine evaluated Ta1 across five hospitals in China, enrolling 334 COVID-19 patients. This study confirmed the earlier findings, showing that Ta1 treatment was associated with improved lymphocyte recovery, reduced ICU admission rates, and lower 28-day mortality. Subgroup analysis revealed that the benefit was concentrated in patients over age 60 and those with comorbidities including diabetes, hypertension, and chronic lung disease, populations known to have baseline immune dysfunction that would be expected to respond most to immune restoration therapy.
Another retrospective study in non-severe COVID-19 patients showed a different pattern. While Ta1 did not significantly alter disease progression or mortality in mild-to-moderate cases (where outcomes were already good with standard care), it did shorten SARS-CoV-2 RNA shedding duration and reduce hospital length of stay. The median time to viral clearance was 9 days in the Ta1 group versus 13 days in the control group (p = 0.02), suggesting that even when clinical benefit is not dramatic, Ta1 accelerates viral elimination through enhanced immune function.
Systematic Reviews and Meta-Analyses
Multiple systematic reviews and meta-analyses have attempted to synthesize the heterogeneous evidence on Ta1 in COVID-19. The results are instructive for understanding both the potential and limitations of the evidence.
A 2023 meta-analysis published in Inflammopharmacology pooled data from 9 studies and found that Ta1 therapy had no statistically significant effect on overall mortality across all severity levels (relative risk 1.03, 95% CI 0.60-1.75, p = 0.92). However, the high heterogeneity (I-squared = 90%) signaled that the overall pooled estimate was misleading. Subgroup analyses told a more nuanced story: in patients with severe and critical COVID-19, Ta1 was associated with a significant 34% reduction in mortality (RR 0.66, 95% CI 0.57-0.76). The benefit was also significant in patients with a mean age over 60 years and in studies where the proportion of female patients was 40% or less.
A separate meta-analysis published in International Immunopharmacology in 2022 pooled 8 studies and found a significant overall mortality benefit (RR 0.59, 95% CI 0.37-0.93, p = 0.02), though again with substantial heterogeneity (I-squared = 84%). A third meta-analysis in the Journal of International Medical Research (2023) found an overall benefit on mortality that was specifically driven by studies using daily dosing regimens (as opposed to twice-weekly dosing), suggesting that in acute severe illness, more frequent dosing may be needed to achieve a therapeutic effect.
The discrepancies among meta-analyses are largely attributable to differences in study inclusion criteria, patient populations (mild vs. severe), dosing protocols (daily vs. twice weekly), and timing of treatment initiation (early vs. late in the disease course). The weight of evidence supports a benefit of Ta1 in severe COVID-19, particularly when started early in patients with documented lymphopenia, but the evidence for milder disease is weaker.
Mechanistic Insights from COVID-19 Research
The COVID-19 studies provided detailed immunological data that deepened understanding of how Ta1 works in acute illness. Serial flow cytometry analysis in several studies revealed the following pattern: within 3-5 days of starting Ta1, CD4+ T-cell counts began to rise, followed by CD8+ T-cell recovery at 5-7 days. NK cell numbers increased in parallel with CD8+ cells. The CD4+/CD8+ ratio, which is typically inverted in severe COVID-19 (indicating excessive CD8 activation relative to CD4 help), normalized by day 10-14 of Ta1 treatment.
The exhaustion markers PD-1, Tim-3, and LAG-3 on circulating T cells decreased significantly with Ta1 treatment, indicating functional restoration of previously exhausted T cells. This finding is particularly relevant because T-cell exhaustion is also a hallmark of the tumor microenvironment in cancer, providing further mechanistic support for Ta1's role in cancer immunotherapy.
Cytokine profiling showed that Ta1 selectively enhanced the production of IFN-gamma and IL-2 (associated with effective antiviral immunity) while reducing IL-6, IL-10, and TNF-alpha (associated with cytokine storm pathology). This selective modulation, boosting protective immunity while dampening harmful inflammation, is consistent with Ta1's established role as an immune modulator rather than a simple immune stimulant. It also explains why Ta1 can be beneficial in the paradoxical immunological environment of severe COVID-19, where both enhancement and suppression of different immune pathways are simultaneously needed.
The peptide research hub covers additional compounds studied during the pandemic period, including LL-37, an antimicrobial peptide with direct antiviral activity, and BPC-157, which has shown protective effects against organ damage in inflammatory conditions.
Long COVID and Post-Acute Sequelae
The emergence of long COVID (post-acute sequelae of SARS-CoV-2 infection, or PASC) has created a new population of patients with chronic immune dysfunction who may benefit from Ta1. Long COVID affects an estimated 10-30% of individuals who recover from acute SARS-CoV-2 infection, with symptoms including persistent fatigue, cognitive dysfunction ("brain fog"), exercise intolerance, and recurrent infections lasting months to years after the initial infection.
Immunological profiling of long COVID patients has revealed persistent abnormalities including reduced CD4+ and CD8+ T-cell counts, elevated inflammatory markers, impaired NK cell function, and signs of chronic T-cell activation and exhaustion. These findings closely mirror the immune dysfunction seen in other conditions where Ta1 has demonstrated benefit, including chronic viral infections and post-sepsis immunoparalysis.
Several clinical groups have begun using Ta1 off-label for long COVID patients with documented immune abnormalities. Case series have reported improvements in fatigue, cognitive function, and exercise tolerance after 8-12 weeks of standard twice-weekly Ta1 therapy, with corresponding improvements in T-cell counts and reduction in inflammatory markers. However, controlled clinical trials are needed to confirm these observations and identify which subsets of long COVID patients are most likely to benefit. Patients with documented lymphopenia, low NK cell activity, or persistent T-cell exhaustion markers appear to be the most promising candidates based on the existing mechanistic understanding.
Lessons for Future Pandemic Preparedness
The COVID-19 experience with Ta1 offers important lessons for pandemic preparedness. First, it highlighted the value of having an immune-modulating agent with broad-spectrum activity available early in an outbreak, before pathogen-specific vaccines and antivirals can be developed. Ta1's mechanism of action - enhancing the host's own immune response rather than targeting a specific pathogen - makes it potentially useful against any viral threat that involves lymphopenia and immune dysregulation.
Second, the studies underscored the importance of patient selection. Ta1 showed clear benefit in severe cases with immune suppression but minimal benefit in mild cases where the immune system was already handling the infection adequately. This principle, that immune-enhancing therapy is most valuable when there is a documented immune deficit to correct, should guide future pandemic therapeutic strategies.
Third, the experience highlighted the challenge of generating strong clinical evidence during a pandemic. Many of the Ta1 COVID-19 studies were retrospective, non-randomized, and conducted under emergency conditions that limited methodological rigor. Future pandemic preparedness should include pre-positioned clinical trial protocols for immune-modulating agents, enabling rapid, well-designed studies as soon as a new pathogen emerges.
Immune Optimization Applications
Figure 7: Beyond its established roles in hepatitis and oncology, thymosin alpha-1 is increasingly applied for immune optimization in aging, vaccination enhancement, and chronic illness support.
Vaccine Adjuvant Applications
One of the most practical applications of thymosin alpha-1 outside of disease treatment is as a vaccine adjuvant, particularly in populations with diminished immune responses. Vaccine efficacy declines with age due to the same thymic involution and T-cell dysfunction that Ta1 addresses. Elderly individuals (over 65) show seroconversion rates to influenza vaccination that are only 30-50% of those seen in younger adults. This represents a significant public health gap, as older adults are also at highest risk for influenza complications and mortality.
Clinical studies have demonstrated that Ta1 administered before or concurrent with influenza vaccination increases seroconversion rates in elderly subjects by 20-30%. In one study of 90 elderly nursing home residents, subjects who received Ta1 1.6 mg subcutaneously twice weekly for 4 weeks before and 4 weeks after influenza vaccination showed antibody titers 2-3 fold higher than those who received vaccination alone. The enhanced response persisted for at least 12 months, suggesting that Ta1 improved both the magnitude and durability of vaccine-induced immunity.
Similar results have been observed with hepatitis B vaccination. In hemodialysis patients, a population notorious for poor vaccine responses due to chronic immune suppression, Ta1 administered as an adjuvant to hepatitis B vaccine increased seroconversion rates from 50-60% (with vaccination alone) to 80-90%. This finding has practical significance for the millions of dialysis patients worldwide who need protection against hepatitis B but fail to respond to standard vaccination protocols.
The vaccine-enhancement effect of Ta1 has broader implications in the era of novel infectious diseases. As new vaccines are developed for emerging pathogens, the ability to boost responses in immunocompromised populations could significantly expand the reach of vaccination programs. The concept extends naturally to other vaccine-preventable diseases including pneumococcal pneumonia, herpes zoster, and potentially even cancer vaccines, where strong T-cell responses are essential for efficacy.
Sepsis and Critical Illness
Sepsis, the body's dysregulated response to infection, kills approximately 11 million people annually and is a leading cause of death in intensive care units worldwide. Like severe COVID-19, sepsis involves an initial hyperinflammatory phase followed by a period of profound immune suppression (immunoparalysis) characterized by lymphocyte apoptosis, monocyte deactivation, and reduced HLA-DR expression. Patients who survive the initial inflammatory insult often succumb to secondary infections due to this acquired immunodeficiency.
Ta1 has been studied as an immune restorative therapy in sepsis, with several clinical trials showing promising results. A randomized controlled trial in 361 patients with severe sepsis demonstrated that Ta1 (1.6 mg subcutaneously daily for 7 days) improved 28-day survival compared to placebo. The benefit was most pronounced in patients with baseline monocyte HLA-DR expression below 30%, a marker of immunoparalysis. In this subgroup, Ta1 increased HLA-DR expression, improved CD4+ T-cell counts, and reduced the incidence of secondary nosocomial infections.
A meta-analysis of Ta1 in sepsis, pooling data from multiple Chinese trials, showed a significant reduction in 28-day mortality (odds ratio 0.59, 95% CI 0.45-0.77) and reduced ICU length of stay. These findings led to the inclusion of Ta1 in Chinese sepsis treatment guidelines, where it is recommended for patients with persistent lymphopenia and evidence of immune dysfunction.
Post-Surgical Immune Recovery
Major surgery induces transient immune suppression that increases the risk of postoperative infections and may affect long-term outcomes in cancer patients. The surgical stress response triggers a shift from Th1 to Th2 cytokine profiles, reduces NK cell activity, and decreases lymphocyte counts. This postoperative immune window can last 7-14 days and represents a period of vulnerability.
Several clinical trials have evaluated perioperative Ta1 to accelerate immune recovery. In patients undergoing major abdominal surgery, Ta1 given preoperatively and continued for 5-7 days postoperatively reduced the incidence of surgical site infections by approximately 40% and shortened hospital stays by 2-3 days compared to placebo. In cardiac surgery patients undergoing cardiopulmonary bypass (a procedure that is particularly immunosuppressive due to blood exposure to artificial surfaces), Ta1 reduced the incidence of postoperative pneumonia and improved T-cell recovery times.
A particularly relevant recent protocol, the PANDA II trial, is evaluating Ta1's effects on immune response and organ function following surgery for acute aortic dissection, one of the most physiologically stressful surgical procedures in modern medicine. The results of this trial, when available, will provide important data on Ta1's role in extreme surgical stress.
Chronic Fatigue and Persistent Infections
A growing area of interest is the use of Ta1 in patients with chronic immune dysfunction, including chronic fatigue syndrome (CFS/ME), post-viral syndromes, and persistent infections such as Epstein-Barr virus (EBV) and cytomegalovirus (CMV) reactivation. These conditions are characterized by T-cell dysfunction, NK cell impairment, and chronic low-grade inflammation, all of which fall within Ta1's therapeutic scope.
While large randomized trials in these populations are lacking, clinical case series and small studies have reported improvements in fatigue scores, NK cell function, and T-cell subsets following Ta1 treatment. The peptide's ability to restore T-cell function while modulating inflammation makes it theoretically well-suited for these conditions, where the immune system is simultaneously underperforming and overreacting. Clinical practitioners who use Ta1 for these indications typically employ the standard 1.6 mg twice-weekly protocol for 3-6 months, with monitoring of lymphocyte subsets to guide treatment duration.
Aging and Immunosenescence
Perhaps the most forward-looking application of Ta1 is in the field of longevity and anti-aging medicine. Immunosenescence, the age-related decline in immune function, is a major contributor to the morbidity and mortality of aging. The characteristics of immunosenescence, including reduced naive T-cell output, narrowed TCR repertoire, accumulation of senescent T cells, and chronic inflammation, are precisely the targets addressed by Ta1's mechanism of action.
Preclinical studies have demonstrated that Ta1 administration to aged mice restores T-cell function to levels approaching those of young animals, improves vaccine responses, and reduces markers of chronic inflammation. Clinical studies in elderly populations have confirmed improvements in CD4+ T-cell counts, CD4+/CD8+ ratios, and vaccine responsiveness.
The concept of using Ta1 as a periodic immune "tune-up" in aging adults is gaining traction in longevity medicine. Rather than continuous therapy, the approach involves cycles of Ta1 treatment (typically 2-4 weeks twice yearly) timed around high-risk periods such as flu season or planned vaccinations. This pulsed approach aligns with the peptide's pharmacodynamic profile, where immune improvements persist for weeks to months after treatment cessation, and minimizes cost and treatment burden.
For those interested in comprehensive longevity strategies, Ta1 pairs well with other anti-aging peptides including epithalon (telomerase activation), SS-31 (mitochondrial protection), MOTS-c (mitochondrial-derived peptide for metabolic health), and NAD+ (cellular energy metabolism). The biohacking hub provides a broader perspective on combining these approaches for healthspan optimization.
Autoimmune Disease Considerations
Given Ta1's immune-modulating properties, there has been interest in its potential role in autoimmune diseases. The picture here is more complex than in immunodeficiency states. Autoimmune diseases involve inappropriate immune activation against self-antigens, so a compound that enhances immune function might seem contraindicated. However, Ta1's bidirectional modulatory capacity, particularly its ability to support Treg function and balance Th1/Th2/Th17 responses, suggests a more nuanced role.
Limited clinical data in autoimmune conditions show that Ta1 does not exacerbate autoimmune flares when given to patients with coexisting autoimmune disease and viral hepatitis. Some case series have reported improvement in autoimmune markers, possibly through enhanced Treg function and improved immune homeostasis. However, this area requires further controlled studies before definitive conclusions can be drawn. Current clinical practice generally advises caution when using Ta1 in patients with active autoimmune disease, though coexisting immune deficiency may tip the risk-benefit calculation in favor of treatment.
Primary Immunodeficiency Disorders
Ta1 has been used in the treatment of primary immunodeficiency disorders, most specifically DiGeorge syndrome (22q11.2 deletion syndrome), a congenital condition characterized by thymic hypoplasia or aplasia, cardiac defects, and characteristic facial features. Children with DiGeorge syndrome have variable degrees of T-cell immunodeficiency, ranging from mild immune dysregulation to severe combined immunodeficiency requiring thymus transplantation.
For patients with partial DiGeorge syndrome (where some thymic tissue remains), Ta1 can supplement endogenous thymic hormone production and support T-cell maturation. Clinical reports have documented improvements in CD4+ T-cell counts, reduced frequency of infections, and improved vaccine responses in children treated with Ta1. The peptide received orphan drug designation from the FDA specifically for DiGeorge syndrome with immune defects, recognizing both the medical need and the biological rationale.
Other primary immunodeficiency conditions where Ta1 has been evaluated include common variable immunodeficiency (CVID), where despite the primary B-cell defect, many patients also have significant T-cell dysfunction, and selective IgA deficiency, where T-cell imbalances may contribute to the variable clinical severity of the condition. In these settings, Ta1 is used as an adjunct to immunoglobulin replacement therapy rather than as a primary treatment.
HIV/AIDS and Immune Reconstitution
The use of Ta1 in HIV/AIDS has been explored since the early days of the epidemic. HIV directly infects and destroys CD4+ T cells, the same cells that Ta1 promotes. The rationale for Ta1 as an adjunct to antiretroviral therapy (ART) is that while ART suppresses viral replication and allows CD4+ count recovery, the quality and diversity of the reconstituted T-cell population may be suboptimal, particularly in patients who started ART at very low CD4+ counts. Ta1 could potentially improve the quality of immune reconstitution by promoting thymic output of new, diverse T cells.
Clinical studies in HIV patients have shown that Ta1 can modestly increase CD4+ T-cell counts above the levels achieved with ART alone, improve CD4+/CD8+ ratios, and enhance T-cell proliferative responses to recall antigens. In patients who fail to achieve adequate immune reconstitution despite viral suppression on ART (so-called "immunological non-responders"), Ta1 represents a potential therapeutic option for improving CD4+ counts and reducing the risk of opportunistic infections and non-AIDS-defining events.
The thymosin alpha-1 product page provides additional information on current availability and formulations, and the free assessment can help determine whether Ta1 may be appropriate for individual clinical situations.
Lyme Disease and Tick-Borne Infections
Chronic Lyme disease and persistent tick-borne infections represent another emerging application area for Ta1. Patients with post-treatment Lyme disease syndrome (PTLDS) often exhibit immune dysregulation characterized by reduced NK cell function, impaired T-cell responses to Borrelia antigens, and chronic inflammatory signaling. Some integrative medicine practitioners have incorporated Ta1 into treatment protocols for these patients, reporting improvements in immune markers and clinical symptoms. While controlled trial data are lacking, the biological rationale for immune restoration in this context is consistent with Ta1's established mechanisms.
Dosing Protocols
Figure 8: Standard and specialized dosing protocols for thymosin alpha-1, including maintenance, acute, and loading-dose approaches for different clinical scenarios.
Standard Clinical Dosing: The 1.6 mg Twice-Weekly Protocol
The standard dosing protocol for thymosin alpha-1 is well established from decades of clinical trials and commercial use as Zadaxin. The approved dosing in most countries is 1.6 mg administered subcutaneously twice weekly, with injections typically spaced 3-4 days apart (e.g., Monday and Thursday, or Tuesday and Friday). This dosing schedule was derived from pharmacokinetic studies showing that Ta1 achieves peak plasma concentrations within 1-2 hours after subcutaneous injection, with a half-life of approximately 2 hours. Despite this relatively short plasma half-life, the biological effects of Ta1 persist for days because the peptide's primary actions involve intracellular signaling cascades and gene expression changes that outlast the plasma concentration.
The choice of 1.6 mg is based on dose-response studies that demonstrated this dose consistently produces measurable improvements in T-cell counts, NK cell activity, and cytokine production. Lower doses (0.8 mg) showed similar but somewhat less consistent effects in some patient populations, while higher doses (up to 6.4 mg per injection) did not provide proportionally greater benefit in most settings. The 1.6 mg dose thus represents the sweet spot of efficacy and efficiency.
Treatment duration varies by indication. For chronic hepatitis B, the standard course is 6 months of treatment followed by 6-12 months of observation, during which the immune restoration effect continues to drive viral clearance. For cancer adjunct therapy, treatment may be continued for 12 months or longer, depending on the ongoing risk of immune suppression from chemotherapy or immunotherapy. For general immune optimization, treatment cycles of 2-3 months with periodic reassessment are typical.
Acute/Intensive Dosing Protocols
For acute conditions requiring rapid immune restoration, such as severe sepsis, critical COVID-19, or severe post-chemotherapy lymphopenia, daily dosing protocols have been employed. The most common acute protocol uses 1.6 mg subcutaneously once daily for 7-14 days, followed by a transition to the standard twice-weekly maintenance schedule. Some clinical trials in sepsis and COVID-19 used 1.6 mg twice daily for the first 3-5 days in the most critically ill patients, though this approach remains less well-validated.
The rationale for daily dosing in acute settings is that the immunological deficit is more severe and time-sensitive than in chronic conditions. In a patient with sepsis-related lymphopenia, waiting 3-4 days between doses may be too slow to prevent secondary infections during the critical window of immunoparalysis. Daily dosing ensures continuous stimulation of T-cell maturation and dendritic cell activation, maximizing the speed of immune reconstitution.
Loading-dose approaches, where a higher initial dose is given to rapidly achieve therapeutic immune effects, have been explored in cancer patients with severe treatment-induced lymphopenia. One protocol uses 3.2 mg daily for the first 5 days, followed by 1.6 mg twice weekly for maintenance. Clinical data on this approach, while limited, suggest faster T-cell recovery without increased toxicity.
Reconstitution and Preparation
Thymosin alpha-1 is supplied as a lyophilized (freeze-dried) powder in sterile vials, typically in 1.6 mg, 3.2 mg, 5 mg, or 10 mg presentations. The vial contents are reconstituted with bacteriostatic water (water for injection containing 0.9% benzyl alcohol as a preservative) immediately before use or, if using a multi-dose vial, reconstituted once and stored refrigerated for up to 14-21 days.
For a 10 mg vial, a typical reconstitution protocol uses 2 mL of bacteriostatic water, yielding a concentration of 5 mg/mL (5,000 mcg/mL). At this concentration, the standard 1.6 mg dose corresponds to 0.32 mL (32 units on a standard insulin syringe). Precise measurement is aided by the use of 0.5 mL or 1.0 mL insulin syringes with 100-unit markings, where each unit corresponds to 0.01 mL.
For those who want personalized guidance on reconstitution volumes and dose calculations, the dosing calculator on FormBlends provides step-by-step instructions for preparing peptide solutions at any desired concentration. The key principles are: use sterile technique throughout, direct the bacteriostatic water along the side of the vial rather than directly onto the powder, swirl gently rather than shaking vigorously, and allow the solution to sit for 1-2 minutes if any particles remain visible.
Injection Technique and Administration
Ta1 is administered via subcutaneous injection, meaning the needle is inserted into the fatty tissue layer between the skin and underlying muscle. The preferred injection sites are the abdomen (at least 2 inches from the navel), the front of the thighs, and the upper outer arms. Injection sites should be rotated with each administration to minimize local irritation and tissue changes.
The injection technique is straightforward: clean the injection site with an alcohol swab, pinch a fold of skin, insert the needle (typically 29-31 gauge, 1/2 inch) at a 45-90 degree angle depending on the thickness of subcutaneous tissue, inject the solution slowly over 5-10 seconds, release the skin fold, and withdraw the needle. Applying gentle pressure with a sterile gauze pad for a few seconds after needle withdrawal helps prevent bleeding or bruising.
Timing of injections relative to meals or other medications is not critical, as Ta1's mechanism is not affected by food intake or most other drugs. Some practitioners recommend morning administration based on the circadian rhythm of the immune system, with cortisol levels and immune cell trafficking following predictable daily patterns. However, this is more a theoretical optimization than an evidence-based requirement, and the most important factor is adherence to the schedule rather than the exact time of day.
Cycling and Maintenance Strategies
There is no established consensus on optimal long-term cycling strategies for Ta1, but clinical experience and pharmacodynamic principles suggest several reasonable approaches:
Continuous therapy (disease-specific): For patients with ongoing chronic hepatitis B, active cancer receiving treatment, or persistent immunodeficiency, continuous twice-weekly dosing for 6-12 months is the standard approach, with reassessment of immune parameters at regular intervals.
Pulsed cycling (immune optimization): For general immune support and longevity applications, a common approach is 8-12 weeks of twice-weekly therapy followed by 8-12 weeks off, repeating 2-3 times per year. This cycling strategy is based on the observation that immune improvements persist for several weeks after treatment cessation, allowing for drug-free intervals without loss of benefit.
Seasonal approach: Some practitioners use Ta1 during high-risk periods such as fall/winter flu season, starting 2-4 weeks before influenza vaccination and continuing for 4-8 weeks after. This timing maximizes both the direct immune-protective effects and the vaccine-enhancement properties of the peptide.
Perioperative use: For planned surgeries, Ta1 started 1-2 weeks before the procedure and continued for 1-2 weeks after can reduce the risk of postoperative immune suppression and associated complications.
Combination Protocols with Other Peptides
Ta1 is often used in combination with other immune-supportive and health-promoting peptides. Common combinations include:
Ta1 + BPC-157: BPC-157's tissue-protective and anti-inflammatory properties complement Ta1's immune-enhancing effects, making this combination popular for recovery from illness or surgery. The two peptides act through entirely different mechanisms and do not interfere with each other.
Ta1 + CJC-1295/Ipamorelin: Growth hormone secretagogues like CJC-1295/Ipamorelin can support thymic regeneration and immune cell production through GH/IGF-1 signaling, potentially amplifying the immune-restorative effects of Ta1. Growth hormone has been shown to promote thymic regrowth in clinical studies.
Ta1 + Selank: Selank is a peptide that modulates immune function through effects on cytokine balance and has anxiolytic properties that may help mitigate the stress-related immune suppression that often accompanies chronic illness.
| Protocol | Dose | Frequency | Duration | Indication |
|---|---|---|---|---|
| Standard maintenance | 1.6 mg SC | Twice weekly | 6-12 months | Chronic HBV, cancer adjunct, general immune support |
| Acute intensive | 1.6 mg SC | Daily | 7-14 days, then twice weekly | Sepsis, severe COVID-19, acute immunosuppression |
| Loading dose | 3.2 mg SC | Daily x 5 days, then 1.6 mg 2x/wk | 4-12 weeks | Severe lymphopenia, cancer treatment recovery |
| Vaccine enhancement | 1.6 mg SC | Twice weekly | 2 wk before + 4 wk after vaccination | Elderly, immunocompromised vaccination |
| Pulsed cycling | 1.6 mg SC | Twice weekly | 8-12 wk on, 8-12 wk off, repeat | Longevity, immune optimization |
| Perioperative | 1.6 mg SC | Twice weekly or daily | 1-2 wk pre-op + 1-2 wk post-op | Major surgery immune support |
Monitoring and Laboratory Assessments
Appropriate monitoring during Ta1 therapy helps optimize outcomes and provides objective measures of response. The following laboratory assessments are recommended at baseline and at regular intervals during treatment:
Baseline (before starting therapy): Complete blood count with differential (to establish baseline lymphocyte count), lymphocyte subset analysis by flow cytometry (CD3+, CD4+, CD8+, NK cells), CD4+/CD8+ ratio, comprehensive metabolic panel including liver function tests, HBV DNA and/or other relevant viral loads (if treating viral infections), C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) as inflammatory markers, and immunoglobulin levels (IgG, IgA, IgM) for assessment of humoral immunity.
During treatment (every 4-8 weeks): CBC with differential to track lymphocyte recovery, lymphocyte subsets (CD4+, CD8+, NK cells) to assess immune reconstitution, viral loads if treating hepatitis or other viral infections, liver function tests if treating hepatic conditions, and CRP to track inflammatory status.
Post-treatment (3, 6, and 12 months after cessation): These follow-up assessments are particularly important because Ta1's effects continue to evolve after treatment stops. The "post-treatment response" phenomenon in hepatitis B, where viral clearance continues to improve for months after Ta1 discontinuation, makes follow-up monitoring essential for capturing the full therapeutic effect.
The dosing calculator provides guidance not just on dosing but also on recommended monitoring schedules based on treatment indication and individual patient factors.
Storage and Stability Considerations
Proper storage of Ta1 is important for maintaining potency and safety. Unreconstituted lyophilized Ta1 should be stored at controlled room temperature (15-25 C/59-77 F) or refrigerated (2-8 C/36-46 F). Most lyophilized formulations maintain full potency for 2-3 years at room temperature and longer under refrigeration. The lyophilized powder should appear as a white to off-white cake or powder; any discoloration, unusual odor, or visible particulates suggest degradation and the vial should be discarded.
Once reconstituted with bacteriostatic water, the solution should be stored refrigerated (2-8 C) and used within 14-21 days, depending on the formulation and manufacturer's recommendations. Reconstituted solutions should be clear and colorless; cloudy solutions or those containing visible particles should not be used. Each withdrawal from a multi-dose vial should be preceded by disinfection of the vial stopper with an alcohol swab to maintain sterility.
For travel or situations where refrigeration is unavailable, reconstituted Ta1 can be kept at room temperature for up to 24-48 hours without significant loss of potency, though this should be the exception rather than the routine. Extended exposure to temperatures above 30 C or direct sunlight can accelerate degradation and should be avoided.
Safety Profile
Figure 9: Thymosin alpha-1 has demonstrated an excellent safety record across decades of clinical use, with adverse events limited primarily to mild injection-site reactions.
Overview of Safety Data
Thymosin alpha-1 has one of the most favorable safety profiles of any biologically active peptide used in clinical medicine. Across more than four decades of study, including over 40 clinical trials enrolling thousands of patients, no serious drug-related adverse events or dose-limiting toxicities have been identified. A comprehensive review published in the Journal of Biological Regulators and Homeostatic Agents in 2024 analyzed the combined safety data from published clinical trials and concluded that Ta1 demonstrates consistent safety and tolerability across all studied indications, doses, and patient populations.
This safety record is particularly impressive when considering the range of patients who have received Ta1. Clinical trials have included patients with chronic liver disease (including those with cirrhosis), active cancer undergoing chemotherapy, critically ill patients with sepsis, elderly individuals with multiple comorbidities, and children with DiGeorge syndrome. The absence of serious adverse events across such diverse and often medically complex populations provides strong reassurance about the peptide's safety.
Common Adverse Effects
The most frequently reported adverse effect is injection-site reaction, occurring in approximately 5-15% of patients. These reactions typically consist of mild redness (erythema), slight swelling, and temporary discomfort at the injection site, resolving spontaneously within 24-48 hours without treatment. The incidence of injection-site reactions does not increase with longer treatment duration, suggesting that local tolerance develops over time rather than sensitization.
Other infrequently reported effects include:
- Mild fatigue: Reported in approximately 3-5% of patients, typically during the first 1-2 weeks of treatment and often attributed to immune activation. This usually resolves spontaneously as the immune system adjusts.
- Low-grade fever: Occurs in approximately 2-4% of patients, particularly during the initial dosing period. Temperatures rarely exceed 38.0 C (100.4 F) and resolve within 24 hours. Like fatigue, this likely reflects the immune-activating properties of the peptide.
- Myalgia (muscle discomfort): Reported occasionally, usually mild and transient. More common with daily dosing protocols than with twice-weekly schedules.
- Headache: Occasionally reported but at rates not significantly different from placebo in controlled trials.
- Nausea: Rare, reported in less than 2% of patients, and typically mild.
Dose-Escalation Safety Data
Formal dose-escalation studies have tested Ta1 at doses far exceeding the standard 1.6 mg twice-weekly protocol. In a key safety study, doses up to 16 mg administered subcutaneously for up to 12 months showed no significant Ta1-specific toxicity. The adverse event profile at 16 mg was essentially identical to that at 1.6 mg, consisting primarily of mild injection-site reactions. Laboratory parameters including complete blood count, comprehensive metabolic panel, liver function tests, and renal function remained within normal limits throughout the high-dose treatment period.
This extraordinary dose-safety margin (at least 10-fold above the standard therapeutic dose) is unusual among biologically active agents and speaks to the fundamental safety of the compound. For comparison, most cytokines used therapeutically (IL-2, interferons, colony-stimulating factors) have narrow therapeutic windows with dose-limiting toxicities that restrict their clinical use. Ta1's lack of significant toxicity even at supra-therapeutic doses suggests that its mechanism of action involves modulation of existing immune pathways rather than pharmacological overstimulation.
Drug Interactions
No clinically significant drug interactions have been identified with Ta1. The peptide has been used safely in combination with:
- Interferon-alpha (and pegylated interferon): Extensive co-administration in hepatitis trials. Ta1 does not increase interferon-related adverse effects (flu-like symptoms, cytopenias, depression). In fact, some studies suggest that Ta1 may mitigate interferon-induced neutropenia through its overall immune-supportive effects.
- Chemotherapy agents: Multiple trials have combined Ta1 with various chemotherapy regimens including platinum-based, taxane-based, and antimetabolite-based regimens without increased toxicity. Ta1 appears to reduce rather than enhance chemotherapy-induced lymphopenia.
- Immune checkpoint inhibitors: Recent combination studies with anti-PD-1 and anti-PD-L1 antibodies show no increase in immune-related adverse events (irAEs), a critical finding given that irAEs are the primary safety concern with checkpoint immunotherapy.
- Ribavirin: Used concurrently in hepatitis C trials without interaction.
- Nucleos(t)ide analogues: Co-administered with entecavir and tenofovir in hepatitis B patients without issues.
- Corticosteroids: While corticosteroids suppress immune function and could theoretically blunt Ta1's effects, clinical data in sepsis patients receiving stress-dose steroids show that Ta1 retains its immune-restorative activity even in this setting.
Special Populations
Elderly patients: Ta1 has been extensively studied in elderly populations, particularly in the context of vaccine enhancement and infection prevention. No age-related dose adjustments are required. The safety profile in patients over 65 is identical to that in younger adults, with the same low incidence of injection-site reactions and no increase in other adverse events.
Patients with liver disease: Given that Ta1's primary clinical indication is chronic viral hepatitis, extensive data exist on its use in patients with compromised liver function. No dose adjustment is needed for hepatic impairment, including compensated cirrhosis. Ta1 is not hepatotoxic and does not affect liver enzyme levels adversely. In fact, ALT levels typically improve during treatment due to reduced hepatic inflammation from improved viral control.
Patients with renal impairment: Limited specific data exist on Ta1 in severe renal impairment, but the peptide's small size (3,108 Da) suggests renal clearance may play a role in its elimination. Studies in hemodialysis patients (who have no residual renal function) show no accumulation of adverse effects, suggesting that alternative clearance mechanisms are sufficient.
Pediatric patients: Ta1 has been used in children with DiGeorge syndrome (thymic aplasia or hypoplasia) and primary immunodeficiency disorders. The safety data in pediatric populations are consistent with adult data, though the total number of pediatric patients studied is smaller.
Pregnancy and lactation: There are insufficient data on Ta1 use during pregnancy and lactation. While no teratogenic effects have been observed in animal studies, Ta1 is generally not recommended during pregnancy unless the potential benefit clearly outweighs the unknown risk. Lactating women should exercise caution, as it is unknown whether Ta1 or its metabolites are excreted in breast milk.
Autoimmune Risk Assessment
A common concern with any immune-enhancing agent is the potential to trigger or exacerbate autoimmune conditions. This concern is particularly relevant for Ta1, given its potent effects on T-cell function and cytokine production. However, the clinical evidence is reassuring.
Across all clinical trials, the incidence of new-onset autoimmune conditions in Ta1-treated patients has not exceeded that in placebo or control groups. In hepatitis B trials, where interferon co-treatment is known to trigger autoimmune thyroiditis in 2-5% of patients, the addition of Ta1 did not increase this risk. In cancer trials, where checkpoint inhibitors carry a 10-30% incidence of immune-related adverse events, the addition of Ta1 has not increased irAE rates in preliminary data.
The biological explanation for this favorable profile likely relates to Ta1's balanced effects on effector and regulatory T cells. Unlike agents that selectively activate effector T cells (such as IL-2 at high doses), Ta1 simultaneously supports Treg function, maintaining the immunological checks and balances that prevent autoimmunity. This balanced modulation is consistent with its natural role as a thymic hormone that promotes proper T-cell selection, including the deletion of self-reactive clones.
Comparison with Other Immune-Modulating Agents
| Agent | Major Adverse Effects | Dose-Limiting Toxicity | Black Box Warning |
|---|---|---|---|
| Thymosin alpha-1 | Injection-site reactions (mild) | None identified | No |
| Interferon-alpha | Flu-like symptoms, cytopenias, depression, thyroiditis | Cytopenias, hepatotoxicity | Yes |
| IL-2 (Aldesleukin) | Capillary leak syndrome, hypotension, organ dysfunction | Capillary leak, cardiac | Yes |
| Anti-PD-1 (Pembrolizumab) | irAEs: colitis, hepatitis, pneumonitis, endocrinopathies | Severe irAEs | Yes |
| G-CSF (Filgrastim) | Bone pain, splenic rupture (rare), capillary leak | Splenic complications | No |
This comparison highlights Ta1's exceptional safety advantage over other immune-modulating agents used in similar clinical contexts. For those weighing the risks and benefits of immune-support peptides, the science and research page provides additional context, and for specific immune-related peptides, our pages on LL-37 and KPV cover safety data for those compounds as well.
Long-Term Safety Data and Post-Marketing Surveillance
The longest continuous exposure to Ta1 documented in clinical studies is 12 months, though many patients have used the peptide for longer periods in clinical practice, particularly in countries where it is commercially available. Post-marketing surveillance data from over 20 years of commercial use as Zadaxin have not identified any delayed or cumulative toxicities. No organ-specific damage (hepatic, renal, cardiac, neurological) has been attributed to Ta1 use, even in populations with pre-existing organ compromise from underlying diseases like cirrhosis, renal failure, or cancer.
Cancer risk is a theoretical concern with any agent that stimulates immune cell proliferation, though the mechanism of concern is largely theoretical. Ta1 promotes differentiation and maturation of existing immune progenitors rather than driving uncontrolled proliferation. No increase in the incidence of any cancer type has been observed in patients treated with Ta1, including in long-term follow-up studies of hepatitis B patients (who are already at elevated cancer risk due to their underlying liver disease). In fact, the cancer adjunct trials suggest the opposite: Ta1 may reduce cancer recurrence through enhanced immune surveillance.
Immunological tolerance or tachyphylaxis (loss of response with repeated dosing) has not been reported with Ta1. Patients who receive multiple treatment courses continue to show immune parameter improvements comparable to their initial responses. This absence of tolerance is consistent with the peptide's mechanism of action through TLR signaling, which does not typically develop tolerance in the way that direct receptor agonists for other pathways sometimes do (TLR tolerance exists but requires continuous, high-dose stimulation that is not characteristic of intermittent Ta1 dosing).
Contraindications and Precautions
Based on available evidence, the following absolute and relative contraindications and precautions apply to Ta1 use:
Absolute contraindications: Known hypersensitivity to thymalfasin or any excipient in the formulation. This is exceedingly rare, as Ta1 is a synthetic version of an endogenous human peptide, but allergic reactions to any injected substance are theoretically possible.
Relative contraindications (use with caution): Organ transplant recipients on immunosuppressive therapy (Ta1 could theoretically promote graft rejection by enhancing T-cell function, though no cases have been reported). Active autoimmune disease with ongoing flares (enhanced immune activity could exacerbate the autoimmune process). Pregnancy and lactation (insufficient safety data).
Precautions: Patients with a history of autoimmune conditions should be monitored more closely during Ta1 therapy, with baseline and periodic assessment of autoimmune markers (ANA, anti-thyroid antibodies, etc.). Patients receiving immunosuppressive medications for any indication should discuss the potential for drug interaction with their prescribing physician, as Ta1's immune-enhancing effects could theoretically counteract the intended immunosuppression.
Overdose and Emergency Management
No cases of acute Ta1 overdose have been reported in the medical literature. Given the extraordinary safety margin demonstrated in dose-escalation studies (no significant toxicity at 10x the standard dose), accidental administration of a larger-than-intended dose is unlikely to cause harm. In the theoretical event of a massive overdose, management would be supportive, with monitoring for signs of immune hyperactivation (fever, cytokine-release-like symptoms) and symptomatic treatment as needed. There is no specific antidote for Ta1, and given its short half-life (approximately 2 hours), any effects from a single overdose would be expected to resolve quickly.
Advanced Clinical Applications & Combination Protocols
Beyond its established uses in hepatitis and cancer immunotherapy, thymosin alpha-1 is finding applications in increasingly diverse clinical scenarios. The peptide's unique ability to modulate rather than simply stimulate immunity makes it relevant for conditions where immune balance, not just immune strength, is the therapeutic goal.
Chronic Infections and Persistent Viral Reactivation
Many adults carry latent viral infections that periodically reactivate when immune surveillance weakens. Epstein-Barr virus (EBV), cytomegalovirus (CMV), herpes simplex virus (HSV), and varicella-zoster virus (VZV) all establish lifelong latent infections that can flare during periods of stress, illness, or immune decline. For individuals experiencing frequent viral reactivations, Thymosin Alpha-1 may strengthen the T-cell surveillance that normally keeps these viruses in check.
The clinical pattern suggesting benefit from TA1 for viral reactivation typically includes: recurrent cold sores or shingles outbreaks more than three times per year, persistent EBV reactivation with elevated viral loads and chronic fatigue, or frequent upper respiratory infections suggesting broader T-cell dysfunction. These patterns indicate that the immune system's cell-mediated arm, specifically the cytotoxic T-cell response responsible for controlling latent viruses, isn't functioning at full capacity.
A typical protocol for chronic viral reactivation uses TA1 at 1.6 mg subcutaneously twice weekly for 4-8 weeks, followed by once weekly for maintenance. This mirrors the dosing used in hepatitis trials but applies it to a different set of viral challenges. Monitoring includes viral load testing (EBV DNA, CMV DNA) where available, along with T-cell subset analysis (CD4/CD8 ratios, NK cell counts and activity). Clinical endpoints include reduction in reactivation frequency and severity over 6-12 months of monitoring.
Combining TA1 with LL-37, an antimicrobial peptide with both direct antiviral properties and immune-modulating effects, creates a dual-mechanism approach to chronic infection management. LL-37 provides direct antimicrobial activity while TA1 strengthens the adaptive immune surveillance. For individuals whose chronic infections coincide with gut dysbiosis (a common finding, since approximately 70% of immune tissue resides in the gut), adding BPC-157 for gastrointestinal support addresses the mucosal immune environment where much of the initial immune activation occurs.
Pre-Surgical Immune Optimization
Surgery creates a predictable period of immune vulnerability. The surgical stress response suppresses cell-mediated immunity for days to weeks after the procedure, creating a window of increased susceptibility to infections, poor wound healing, and, in cancer surgery, potential metastatic seeding. Pre-surgical immune optimization with TA1 aims to elevate baseline immune function high enough that the post-surgical dip doesn't reach dangerously low levels.
The concept is similar to "prehabilitation" in physical therapy, building capacity before a known stress. Protocols typically begin TA1 administration 2-4 weeks before planned surgery, at 1.6 mg twice weekly, continuing through the surgical date and for 2-4 weeks postoperatively. Clinical experience from cancer surgery centers that have adopted this approach suggests reduced postoperative infection rates and faster recovery of immune markers to baseline.
This application is particularly relevant for cancer surgery, where post-surgical immune suppression coincides with the highest risk of residual cancer cell survival and distant seeding. TA1's ability to enhance NK cell activity and cytotoxic T-cell function during the critical postoperative period could theoretically reduce recurrence risk, though this hypothesis requires validation in controlled clinical trials. For individuals planning elective surgery, discussing pre-surgical TA1 with both the surgeon and an integrative medicine provider ensures coordinated care.
Mold Illness and Chronic Inflammatory Response Syndrome (CIRS)
Chronic Inflammatory Response Syndrome, often triggered by mold exposure in water-damaged buildings, involves a dysregulated immune response where the innate immune system remains chronically activated while the adaptive immune system fails to mount an effective clearing response. Patients with CIRS often show characteristic immune abnormalities including reduced regulatory T-cell function, skewed cytokine profiles, and impaired ability to process and clear biotoxins.
TA1's dual action of enhancing adaptive immunity while supporting regulatory T-cell function makes it mechanistically relevant for CIRS. By strengthening the adaptive immune arm (allowing more effective pathogen/toxin clearance) while supporting Treg function (reducing the chronic inflammatory drive), TA1 may help rebalance the immune dysfunction at the core of CIRS pathophysiology.
CIRS treatment protocols using TA1 typically integrate the peptide into broader mold illness management: environmental remediation (removing the mold exposure source), binder protocols (cholestyramine or activated charcoal to bind circulating biotoxins), and anti-inflammatory support. TA1 at 1.6 mg twice weekly is added to this foundation, typically continued for 3-6 months with periodic reassessment of immune markers and clinical symptoms. VIP (Vasoactive Intestinal Peptide) is another peptide used in CIRS protocols, targeting the hypothalamic-regulatory component of the syndrome, and can be combined with TA1 for broader physiological coverage.
Lyme Disease and Co-Infection Support
Chronic Lyme disease and its common co-infections (Babesia, Bartonella, Ehrlichia) present immune challenges that share features with both chronic viral infections and autoimmune conditions. The Borrelia spirochete is particularly adept at evading immune detection, and many Lyme patients show T-cell exhaustion and impaired immune surveillance alongside ongoing inflammation. This paradox, where the immune system is simultaneously exhausted and hyperactivated, is difficult to address with simple immune stimulants.
TA1's immunomodulatory (rather than purely immunostimulatory) properties make it a more nuanced tool for Lyme-related immune dysfunction. By promoting T-cell maturation and function while supporting regulatory mechanisms, TA1 may help the immune system mount a more effective response to the spirochete without exacerbating the autoimmune-like inflammatory component of chronic Lyme disease.
Lyme-literate physicians who incorporate TA1 typically use it alongside antimicrobial treatment, reasoning that immune support makes antibiotic and herbal protocols more effective by improving the body's ability to target and eliminate organisms that antibiotics drive out of protected niches. The combination of TA1 for adaptive immune enhancement, LL-37 for direct antimicrobial activity, and BPC-157 for gut mucosal protection (important during extended antibiotic courses) represents an integrated peptide approach to complex infectious disease management.
Vaccine Response Enhancement
TA1 has been studied as a vaccine adjuvant, meaning it can enhance the immune system's response to vaccines. This application is particularly relevant for immunocompromised individuals, elderly patients, and others who don't generate strong vaccine responses. Studies in elderly patients showed that adding TA1 to influenza vaccination significantly improved antibody titers and seroconversion rates compared to vaccination alone.
The mechanism involves TA1's effects on dendritic cell maturation and antigen presentation. Better antigen presentation leads to stronger T-cell activation, which in turn supports more strong B-cell antibody production. The result is a more complete and durable immune response to the vaccine antigen.
For individuals who know they respond poorly to vaccines (documented by post-vaccination titer testing), using TA1 at 1.6 mg on the day of vaccination and again 3-4 days later may enhance the immune response. This approach has the strongest evidence base for influenza and hepatitis B vaccines but likely applies to other vaccines as well, given that the mechanism of enhancement is upstream of the specific antigen being targeted.
Immune Aging, Longevity & Preventive Applications
The immune system doesn't just protect against infections. It performs constant surveillance against cancer cells, clears senescent "zombie" cells, maintains tissue homeostasis, and modulates inflammatory responses throughout the body. As immune function declines with age, a process called immunosenescence, virtually every aspect of health deteriorates. This makes immune system maintenance one of the most impactful interventions for healthy aging.
Understanding Immunosenescence
Immunosenescence isn't simply a weakening of the immune system. It's a restructuring that involves several distinct changes occurring simultaneously. Thymic involution, the shrinking and fatty replacement of the thymus gland, begins in early adulthood and accelerates after age 40. By age 65, most people have lost 90-95% of their functional thymic tissue, dramatically reducing the production of new naive T cells. Without new T-cell recruits, the immune system relies increasingly on memory T cells that are progressively less responsive and less diverse.
Simultaneously, the innate immune system becomes chronically activated, a state termed "inflammaging." This creates a background of low-grade systemic inflammation that drives atherosclerosis, insulin resistance, neurodegeneration, and accelerated aging of virtually every organ system. The paradox of immunosenescence is that the immune system is simultaneously too weak (inadequate adaptive responses to pathogens and cancer) and too active (chronic innate inflammation), and these two problems reinforce each other.
TA1 addresses immunosenescence at its source by partially compensating for thymic decline. While it can't reverse thymic involution, it can support the maturation and function of existing T-cell precursors, effectively doing some of the "training" work that the declining thymus is no longer performing efficiently. Its concurrent support of regulatory T cells helps address the inflammaging component by promoting immune homeostasis and resolution of inappropriate inflammatory responses.
The Preventive Immune Maintenance Protocol
An emerging application of TA1 is as a preventive immune maintenance compound for otherwise healthy individuals over 50 who want to preserve immune function as they age. This is a shift from TA1's traditional use as a treatment for existing diseases to its use as a proactive measure against age-related immune decline.
Preventive protocols typically use lower doses and less frequent administration than therapeutic regimens. A common approach is TA1 at 1.6 mg once weekly, administered in cyclical patterns: three months on, one month off, or two months on, one month off. This cycling prevents any theoretical risk of immune overstimulation while providing regular immune system support. Some practitioners include a more intensive "boost" period during cold and flu season (October through March in the Northern Hemisphere), increasing to twice weekly during these higher-risk months.
Monitoring for preventive use focuses on immune function markers that track immunosenescence progression: CD4/CD8 T-cell ratios, NK cell counts and cytotoxicity, naive-to-memory T-cell ratios, and inflammatory markers including hs-CRP, IL-6, and TNF-alpha. Baseline testing before starting TA1 and follow-up testing every 6-12 months provides data to assess whether the protocol is maintaining or improving immune parameters. The FormBlends Science page provides guidance on appropriate immune testing panels.
TA1 in the Longevity Peptide Stack
Within a comprehensive longevity strategy, TA1 addresses the immune pillar of aging while other compounds target different aging mechanisms. Understanding how TA1 integrates with other longevity-focused peptides helps design coherent multi-mechanism protocols.
Epithalon targets telomere maintenance through telomerase activation. TA1 and Epithalon are complementary: Epithalon supports the longevity of immune cells at the chromosomal level, while TA1 supports their functional maturation and activity. An aging immune cell with longer telomeres (from Epithalon) that is also better trained and more functionally competent (from TA1) is more effective than one benefiting from either intervention alone.
FOXO4-DRI clears senescent cells that are both a consequence of and contributor to immune decline. Senescent cells produce inflammatory SASP factors that impair immune function, while declining immune function allows senescent cells to accumulate. Using TA1 to boost immune surveillance against senescent cells while using FOXO4-DRI for direct senolytic clearance addresses this cycle from both directions. Some longevity clinicians sequence these interventions: a FOXO4-DRI senolytic course first to reduce the inflammatory burden, followed by TA1 to strengthen the immune system's ability to prevent rapid senescent cell reaccumulation.
NAD+ supplementation supports the metabolic foundation that immune cells need to function. T-cell activation is enormously energy-intensive, requiring rapid mitochondrial upregulation. Age-related NAD+ decline impairs this metabolic burst, contributing to T-cell exhaustion. Combining NAD+ precursors with TA1 provides both the functional training (TA1) and the metabolic fuel (NAD+) that aging immune cells need.
MOTS-c, a mitochondrial-derived peptide, offers another angle on immune cell metabolism. MOTS-c has been shown to enhance T-cell function under stress conditions, possibly by improving mitochondrial resilience. Combined with TA1's effects on T-cell maturation, the result is immune cells that are both better trained and more metabolically resilient.
Practical Considerations for Long-Term Immune Maintenance
For individuals considering long-term TA1 use for immune maintenance, several practical factors deserve attention:
When to start: There's no definitive age threshold for beginning preventive TA1 therapy, but immune testing can identify individuals whose immune parameters are declining faster than average for their age. A person at 45 with immune markers typical of a 60-year-old is a stronger candidate for early intervention than a 55-year-old with age-appropriate immune function. The FormBlends free assessment can help determine whether immune-focused peptide therapy is appropriate for your situation.
Interactions with autoimmune tendencies: A family history of autoimmune disease or borderline-positive autoimmune markers (low-titer ANA, slightly elevated thyroid antibodies) doesn't necessarily contraindicate TA1 use, but it does warrant extra monitoring. TA1's support of regulatory T cells may actually help maintain self-tolerance, but the theoretical risk of tipping a borderline autoimmune tendency into clinical disease exists. Checking autoimmune markers at baseline and at 3-6 month intervals during TA1 therapy provides appropriate surveillance.
Cost-benefit considerations: TA1 is not inexpensive for long-term use, and the preventive application lacks the disease-specific clinical trial data that supports its therapeutic uses. Individuals considering preventive TA1 should weigh the cost against other longevity investments (nutrition, exercise, sleep optimization, stress management) that have stronger evidence bases and lower financial barriers. TA1 makes the most sense for individuals who have already optimized foundational health practices and are looking for additional immune support, not as a substitute for basics. Visit the Biohacking Hub for broader context on prioritizing longevity interventions.
Practical Troubleshooting & Protocol Optimization
Working with thymosin alpha-1 in practice involves navigating questions that clinical trial protocols don't always address. How do you know if TA1 is working? What do you do when expected improvements don't materialize? How should the protocol be adjusted based on individual response? These practical considerations determine real-world outcomes as much as the underlying pharmacology does.
Measuring Immune Response: How to Know TA1 Is Working
Unlike compounds that produce subjective effects you can feel (like a sleep peptide improving your sleep or a nootropic sharpening your focus), TA1's effects on immune function are largely invisible to subjective experience unless you're fighting an active infection. This makes objective monitoring particularly important for guiding protocol decisions.
Complete blood count with differential: This basic blood test provides a snapshot of immune cell populations. While TA1 doesn't dramatically change total white blood cell counts in healthy individuals, it can improve the ratios between cell types. A normalizing CD4/CD8 ratio (moving toward 1.5-2.0 in individuals where it was depressed), stable or increasing lymphocyte counts, and maintained neutrophil-to-lymphocyte ratio (a marker of inflammation and immune balance) all suggest TA1 is producing its intended immune-modulating effects.
NK cell count and function: Natural killer cell activity is one of TA1's most directly measurable effects. NK cell counts (CD56+/CD16+ cells) and, more critically, NK cell cytotoxicity assays (measuring how effectively your NK cells kill target cells) provide functional immune data. These tests are more specialized than standard blood work and may require a laboratory experienced in immune function testing. An increase in NK cell cytotoxicity during TA1 therapy is a positive sign of immune enhancement.
Inflammatory markers: hs-CRP, IL-6, and TNF-alpha levels reflect the balance between immune activation and immune regulation. In individuals with chronic low-grade inflammation (inflammaging), effective TA1 therapy should reduce these markers over time as the regulatory T-cell support helps resolve inappropriate inflammatory signaling. Conversely, in immunosuppressed individuals, a modest initial increase in inflammatory markers might reflect appropriate immune activation. The trajectory and context matter more than any single measurement.
Clinical endpoints: For many users, the most meaningful measure of TA1 effectiveness is the reduction in infection frequency and severity. If you typically get three to four upper respiratory infections per year and that drops to one or two after starting TA1, the clinical improvement speaks louder than any lab value. Keeping a simple health diary tracking illnesses, their duration, and severity provides this data. Six months of pre-TA1 baseline data compared to six months of on-TA1 data gives a meaningful comparison.
When TA1 Doesn't Seem to Work: Diagnostic Considerations
If immune monitoring and clinical endpoints don't show expected improvement after 8-12 weeks of appropriate TA1 therapy, several factors may explain non-response:
Product quality: As with all peptides, quality varies among suppliers. TA1 from FormBlends undergoes rigorous purity testing, but products from other sources may contain degraded or improperly synthesized peptide. Request a certificate of analysis from your supplier and verify that purity is 98% or higher by HPLC, with correct molecular weight confirmation by mass spectrometry. If you suspect product quality issues, switching to a verified source before concluding that TA1 doesn't work for you is a reasonable diagnostic step.
Underlying nutritional deficiencies: The immune system requires specific nutrients to respond to TA1's signaling. Zinc deficiency directly impairs T-cell development and function. Vitamin D deficiency reduces the expression of antimicrobial peptides and impairs both innate and adaptive immunity. Iron deficiency compromises immune cell proliferation. Selenium deficiency impairs thyroid function, which indirectly affects immune competence. If these foundational nutrients are insufficient, TA1 provides signaling to an immune system that lacks the building blocks to respond adequately. Checking and correcting nutritional status may unmask a TA1 response that was previously blunted.
Chronic stress and cortisol excess: Chronically elevated cortisol directly suppresses the immune functions that TA1 aims to enhance. T-cell maturation, NK cell activity, and cytokine production are all impaired by persistent cortisol elevation. If you're under chronic stress without adequate stress management, TA1 is essentially fighting against cortisol-mediated immunosuppression. Addressing the stress component through lifestyle measures, exercise, meditation, or anxiolytic support with Selank may be necessary before TA1 can produce meaningful immune enhancement.
Gut dysbiosis and mucosal immune dysfunction: Approximately 70% of the immune system resides in the gut-associated lymphoid tissue (GALT). Significant gut dysbiosis, intestinal permeability ("leaky gut"), or chronic intestinal inflammation creates an immune environment where systemic interventions like TA1 are partially offset by ongoing mucosal immune dysfunction. Addressing gut health with BPC-157 for mucosal repair, probiotics for microbiome support, and dietary modification for inflammation reduction creates a healthier immune foundation for TA1 to work with.
Sleep deprivation: Chronic sleep restriction profoundly impairs immune function. Studies show that even one week of mild sleep restriction (6 hours per night instead of 8) reduces vaccine response by over 50% and decreases NK cell activity significantly. If sleep is consistently inadequate, TA1's immune-enhancing signals compete against the immunosuppressive effects of sleep deprivation. Pinealon and DSIP can support sleep optimization as a prerequisite for effective immune enhancement.
Adjusting the Protocol Based on Response
TA1 protocols shouldn't be static. Adjusting based on individual response optimizes outcomes while minimizing unnecessary exposure and cost.
If response is good (improved labs, fewer infections): Consider transitioning from the initial twice-weekly therapeutic schedule to a once-weekly maintenance dose after 8-12 weeks. Some individuals can eventually reduce to biweekly administration while maintaining benefits. The minimum effective dose varies between individuals and can only be determined empirically by gradually reducing frequency and monitoring whether benefits persist.
If response is partial (some improvement but less than expected): Before increasing the dose, ensure that foundational factors (nutrition, sleep, stress, gut health) are addressed. If foundations are solid and product quality is confirmed, increasing from 1.6 mg to 3.2 mg per dose (splitting into two daily injections) may produce additional benefit in some individuals. This higher-dose approach mirrors the dose-escalation protocols used in clinical trials for hepatitis and cancer, where doses up to 16 mg were tested without significant toxicity.
Seasonal adjustments: Many practitioners increase TA1 frequency during cold and flu season (October through March in the Northern Hemisphere) and reduce it during summer months when infection risk is lower and natural immune function tends to be stronger due to increased vitamin D from sun exposure, more physical activity, and better sleep patterns. This seasonal cycling aligns immune support with the periods of greatest need.
Illness-triggered adjustments: At the first sign of an upper respiratory infection, some practitioners recommend a short "pulse" of daily TA1 dosing for 3-5 days, followed by return to the regular schedule. The rationale is to provide intensive immune support during the critical window when the immune system is mounting its initial response to a pathogen. Anecdotal reports suggest this approach may reduce illness duration and severity, though controlled data are lacking.
Reconstitution, Storage, and Injection Technique
For individuals using compounded TA1 from sources like FormBlends, proper handling ensures consistent potency and safety. TA1 is supplied as a lyophilized powder that should be stored at -20 degrees Celsius before reconstitution or at 2-8 degrees Celsius for shorter-term storage. Reconstitute with bacteriostatic water using the technique described for other peptides: direct water against the vial wall, swirl gently, do not shake. The reconstituted solution should be clear and colorless; discard any solution that appears cloudy, discolored, or contains particles.
Subcutaneous injection technique follows standard peptide injection practices. Use a 29-31 gauge insulin syringe, clean the injection site with an alcohol swab, pinch a fold of skin (abdomen, outer thigh, or upper arm), and inject at a 45-90 degree angle depending on subcutaneous tissue thickness. Rotate injection sites systematically. TA1 injections are generally painless due to the small volume and low viscosity of the solution. Any stinging or burning at the injection site that persists for more than a few minutes may indicate product degradation and warrants switching to a fresh vial.
Lifestyle Foundations for Immune Optimization
TA1 works best when the lifestyle foundations for immune health are in place. These aren't optional extras; they determine the ceiling of what TA1 can achieve. Exercise is arguably the most potent natural immune modulator available. Moderate aerobic exercise (30-60 minutes, 4-5 times per week at 60-75% of maximum heart rate) enhances immune surveillance, improves NK cell function, and reduces systemic inflammation. However, excessive exercise without adequate recovery actually suppresses immune function, a phenomenon well-documented in overtrained athletes. For individuals using TA1, maintaining a consistent moderate exercise program provides the physiological context that maximizes the peptide's immune-modulating effects.
Nutritional support for immune function includes adequate zinc (15-30 mg daily), vitamin D (maintaining serum levels of 40-60 ng/mL through supplementation if needed), vitamin C (500-1000 mg daily), selenium (200 mcg daily), and adequate protein (1.0-1.2 g/kg body weight). These micronutrients serve as cofactors and building blocks for the immune responses that TA1 stimulates. Without them, TA1 provides signaling to an immune system that lacks the raw materials to respond effectively, like turning up the volume on a speaker that has no power.
The key to effective TA1 therapy is patience combined with systematic monitoring. Immune system changes develop over weeks to months, not days. Expecting immediate results leads to premature protocol changes that prevent meaningful assessment of effectiveness. Commit to a minimum of 12 weeks at a consistent dose and schedule before drawing conclusions, and ensure that at least one round of immune monitoring (labs plus clinical tracking) has been completed before modifying the protocol. The FormBlends dosing calculator provides structured guidance for TA1 protocols, and the free assessment helps determine whether immune optimization with TA1 is appropriate for your specific health profile.
Thymosin Alpha-1 and Vaccine Response Enhancement in Aging Populations
One of the most clinically substantiated applications of thymosin alpha-1 involves its ability to enhance vaccine responses, particularly in elderly individuals whose age-related immune decline (immunosenescence) renders standard vaccines less effective. This application has practical relevance that extends beyond theoretical immunology, as inadequate vaccine responses in older adults translate directly into preventable hospitalizations, complications, and deaths from infections like influenza, pneumonia, and COVID-19.
The fundamental challenge with vaccination in older adults is that the immune system's ability to generate a strong, durable antibody response to novel antigens declines significantly after age 65. Thymic involution, the gradual shrinking and fatty replacement of the thymus gland that begins in puberty and accelerates after middle age, reduces the output of naive T cells needed to recognize and respond to new antigens presented by vaccines. By age 65, thymic output has decreased by roughly 95% compared to peak childhood levels. This means the immune system must rely increasingly on its existing pool of memory T cells, which were educated against past infections but respond poorly to the new antigens in vaccines. The result is that standard influenza vaccines, for example, achieve only 30-40% protection in adults over 65, compared to 70-90% in younger adults.
Thymosin alpha-1 addresses this problem by partially compensating for the age-related loss of thymic function. Clinical studies in elderly populations have demonstrated that TA1 administration before or alongside vaccination can increase antibody seroconversion rates, boost the magnitude of the antibody response, and extend the duration of protective immunity. In influenza vaccine studies, elderly subjects who received TA1 alongside vaccination achieved antibody titers comparable to those seen in younger adults receiving the vaccine alone. This effect has been documented for influenza, hepatitis B, and pneumococcal vaccines, suggesting that TA1's immune-enhancing effects are not antigen-specific but rather reflect a general restoration of the immune system's capacity to respond to vaccines.
The practical protocol for vaccine enhancement typically involves TA1 administration beginning 1-2 weeks before vaccination and continuing for 2-4 weeks after. This pre-treatment period allows TA1 to upregulate T cell function, increase dendritic cell antigen presentation, and restore some of the immune signaling that immunosenescence has diminished. The post-vaccination continuation supports the expansion and maturation of antigen-specific T and B cells during the critical window when vaccine-induced immunity is being established. Some practitioners use a simplified protocol with TA1 starting on the day of vaccination and continuing for 2 weeks, which is more convenient and still appears to provide benefit, though the data are less strong than for the longer protocol.
Beyond the elderly, other populations with impaired vaccine responses may benefit from TA1 co-administration. Immunocompromised patients, including those on chemotherapy, post-transplant immunosuppression, or living with HIV, often fail to generate adequate immune responses to standard vaccines. Patients with chronic kidney disease on dialysis have specifically poor vaccine responses, as uremia impairs multiple aspects of immune function. Diabetic patients, particularly those with poorly controlled blood glucose, also show diminished vaccine efficacy. In each of these populations, TA1's ability to restore immune competence may help close the gap between the protection vaccines are designed to provide and the protection these patients actually receive.
The COVID-19 pandemic highlighted the real-world consequences of inadequate vaccine responses in vulnerable populations and generated renewed interest in vaccine adjuvant strategies, including peptide-based approaches. While TA1 was not included in the major COVID-19 vaccine trials, its use as an off-label immune enhancer alongside COVID-19 vaccination became common in integrative medicine practice, particularly for elderly patients and those with documented poor responses to initial vaccine doses. As the infectious disease landscape continues to evolve and new vaccines are developed for emerging threats, TA1's role as a vaccine response enhancer is likely to grow in both clinical practice and research attention. The FormBlends TA1 product page provides current dosing guidance, and the free assessment helps identify whether immune optimization through TA1 is appropriate for your specific health profile.
Frequently Asked Questions
What is thymosin alpha-1 and how does it work?
Thymosin alpha-1 (Ta1) is a 28-amino-acid peptide originally isolated from the thymus gland that functions as a key regulator of immune system activity. It works by binding to Toll-like receptors 2 and 9 (TLR2 and TLR9) on dendritic cells, triggering signaling cascades that promote T-cell maturation, enhance natural killer cell activity, and stimulate production of interferons and interleukins. Unlike simple immune stimulants that broadly ramp up immune activity, Ta1 acts as a genuine immune modulator, capable of both enhancing deficient immune responses and preventing excessive inflammation. This balanced action comes from its simultaneous support of both effector T cells and regulatory T cells (Tregs). The peptide has been studied in over 40 clinical trials and is approved in 35+ countries under the brand name Zadaxin.
Is thymosin alpha-1 FDA approved?
Thymosin alpha-1 (thymalfasin/Zadaxin) has received FDA orphan drug designation for four conditions: hepatocellular carcinoma, malignant melanoma, DiGeorge syndrome with immune defects, and chronic active hepatitis B. However, it has not received full FDA approval as a stand-alone therapeutic drug in the United States. In contrast, it is approved and commercially marketed in over 35 countries across Latin America, Eastern Europe, the Middle East, and Asia-Pacific for the treatment of chronic hepatitis B and C and as an immune response enhancer. The discrepancy largely reflects differences in regulatory requirements between jurisdictions rather than safety or efficacy concerns. In the U.S., thymosin alpha-1 has been available through compounding pharmacies.
How does thymosin alpha-1 boost immunity?
Ta1 enhances immunity through several interconnected mechanisms. First, it activates dendritic cells via TLR2 and TLR9, promoting their maturation and improving their ability to present antigens to T cells. Second, it drives T-cell differentiation and proliferation, increasing both CD4+ helper and CD8+ cytotoxic T-cell populations. Third, it enhances NK cell cytotoxicity by 40-60%, improving first-line defense against virus-infected and cancerous cells. Fourth, it stimulates production of IFN-alpha, IFN-gamma, and IL-2, creating a broad antiviral and anti-tumor state. Fifth, it modulates regulatory T cells to maintain immune balance. Clinical studies consistently show increases in CD4+ T-cell counts from baseline values of 400-500 cells/mcL to 700-800 cells/mcL over 12-24 weeks of treatment.
What is the standard dosage of thymosin alpha-1?
The standard clinical dosage is 1.6 mg administered subcutaneously twice per week, with injections spaced 3-4 days apart (for example, Monday and Thursday). This dosing schedule was established through pharmacokinetic and dose-response studies and is used across most clinical trials and approved indications. For acute conditions like sepsis or severe infections, daily dosing of 1.6 mg for 7-14 days may be used before transitioning to the twice-weekly maintenance schedule. Some protocols employ a loading dose approach with 3.2 mg daily for the first 5 days. Treatment duration varies by indication: 6 months for hepatitis B, 12+ months for cancer adjunct therapy, and 8-12 week cycles for immune optimization. Use the dosing calculator for personalized reconstitution guidance.
What are the side effects of thymosin alpha-1?
Thymosin alpha-1 has an exceptionally favorable safety profile. The most common side effect is mild injection-site reaction, including redness, slight swelling, and temporary discomfort, occurring in about 5-15% of patients and resolving within 24-48 hours. Less common effects include mild fatigue (3-5%), low-grade fever below 38.0 C (2-4%), and occasional muscle discomfort, particularly during the first 1-2 weeks of therapy. These effects are generally attributed to immune activation and typically resolve spontaneously. No serious drug-related adverse events or dose-limiting toxicities have been identified across decades of clinical use. Even at experimental doses up to 16 mg (10 times the standard dose) administered for 12 months, no significant Ta1-specific toxicity was observed.
Can thymosin alpha-1 help with cancer treatment?
Clinical evidence supports Ta1 as a beneficial adjunct to cancer treatment, particularly when combined with chemotherapy and immunotherapy. In hepatocellular carcinoma, adding Ta1 to lenvatinib plus anti-PD-1 therapy extended median overall survival from 11.0 to 16.0 months. In non-small cell lung cancer, Ta1 accelerated lymphocyte recovery after chemoradiation, enabling better responses to subsequent immunotherapy. In post-resection HCC, Ta1 improved 5-year overall survival from 65.4% to 77.8%. The mechanism involves restoring chemotherapy-depleted immune cells, enhancing anti-tumor T-cell and NK cell activity, reducing tumor-infiltrating regulatory T cells, and improving dendritic cell antigen presentation. Ta1 has been studied across multiple tumor types and received FDA orphan drug designation for both HCC and melanoma.
What did COVID-19 research show about thymosin alpha-1?
COVID-19 research on Ta1 produced mixed but informative results. In severe and critically ill patients, Ta1 was associated with significant mortality reductions of 34-41% in subgroup analyses from multiple meta-analyses. The early Wuhan study showed mortality of 11.1% with Ta1 versus 30.0% without (p = 0.044). However, across all severity levels (including mild-moderate cases), the overall mortality benefit was not consistently statistically significant, with high heterogeneity across studies. The clearest benefits were seen in patients over 60, those with documented lymphopenia, and those with severe/critical disease. Even in non-severe patients, Ta1 shortened viral shedding time (9 vs. 13 days) and hospital stays. The immunological data from these studies confirmed that Ta1 restores T-cell counts, reverses T-cell exhaustion, and selectively modulates cytokine production.
How does thymosin alpha-1 compare to other immune peptides?
Ta1 is unique among immune peptides in several respects. Compared to LL-37, which primarily acts as a direct antimicrobial with some immune-modulating effects, Ta1 works exclusively through immune modulation without direct pathogen-killing activity. Compared to KPV, which focuses on anti-inflammatory actions, Ta1 provides both immune enhancement and inflammation control. Relative to cytokine therapies like IL-2 or interferon, Ta1 offers comparable immune activation with dramatically fewer side effects. Its mechanism through TLR2/TLR9 gives it a unique position as a bridge between innate and adaptive immunity. The safety profile is superior to virtually all other immune-modulating agents, with no dose-limiting toxicities identified even at 10x the standard dose.
Can thymosin alpha-1 help with aging and immunosenescence?
Yes, thymosin alpha-1 directly addresses the core mechanisms of age-related immune decline (immunosenescence). As the thymus shrinks with age, losing roughly 3% of functional tissue annually after age 20, the output of new naive T cells drops progressively. Ta1 compensates by promoting T-cell maturation and differentiation both within and outside the thymus, increasing CD4+ T-cell counts, restoring balanced CD4+/CD8+ ratios, and broadening the T-cell receptor repertoire. Clinical studies in elderly populations show improved vaccine responses (20-30% higher seroconversion rates), enhanced NK cell function, and reduced inflammatory markers. For longevity applications, pulsed cycles of 8-12 weeks twice yearly are commonly used, often combined with other longevity peptides like epithalon and SS-31.
Is thymosin alpha-1 safe with autoimmune conditions?
The evidence on Ta1 and autoimmune conditions is generally reassuring but warrants caution. Across all clinical trials, Ta1 has not increased the incidence of new-onset autoimmune conditions. In hepatitis B trials where interferon co-treatment is known to trigger autoimmune thyroiditis in 2-5% of patients, adding Ta1 did not increase this risk. In cancer studies combining Ta1 with checkpoint inhibitors (which carry 10-30% rates of immune-related adverse events), preliminary data show no increase in irAEs. The biological basis for this safety lies in Ta1's balanced effects on both effector and regulatory T cells, maintaining the immune checks that prevent autoimmunity. However, data specific to patients with active autoimmune disease are limited, and current practice advises caution in this population. Consult your healthcare provider before starting Ta1 if you have an autoimmune condition.
What is the difference between thymosin alpha-1 and thymosin beta-4?
Though both are thymic peptides, thymosin alpha-1 and thymosin beta-4 (Tb4) have very different structures, mechanisms, and clinical applications. Ta1 is a 28-amino-acid peptide that primarily modulates immune function through TLR2/TLR9 activation on dendritic cells, promoting T-cell maturation and balanced immune responses. Tb4 is a larger 43-amino-acid peptide that primarily functions as an actin-binding protein involved in cell migration, wound healing, and tissue repair. TB-500, a synthetic fragment of Tb4, is used for its regenerative properties rather than immune modulation. In practice, the two peptides serve complementary rather than overlapping purposes: Ta1 for immune support and Tb4/TB-500 for tissue recovery. They can be used together safely in combination protocols targeting both immune and regenerative goals.
How long does it take for thymosin alpha-1 to work?
The timeline for thymosin alpha-1's effects depends on the indication and what endpoints you're measuring. Immune cell changes begin within days: CD4+ T-cell counts typically start rising within 3-7 days of initiating therapy, with measurable improvements in NK cell activity within the first 1-2 weeks. For acute conditions like sepsis, clinical benefits in terms of reduced secondary infections and improved organ function scores can appear within the first week of daily dosing. For chronic hepatitis B, virological responses (HBV DNA decline, HBeAg loss) develop gradually over 3-6 months and often continue to improve for 6-12 months after stopping treatment. For immune optimization and longevity applications, most practitioners recommend at least 8-12 weeks of therapy before assessing response, with full benefits becoming apparent over 3-6 months.
Can thymosin alpha-1 be combined with other peptides?
Yes, Ta1 is commonly combined with other peptides in clinical practice. Popular combinations include Ta1 with BPC-157 for combined immune support and tissue protection, Ta1 with CJC-1295/Ipamorelin for immune and growth hormone optimization (GH supports thymic regeneration), and Ta1 with Selank for immune modulation plus stress reduction. For longevity protocols, Ta1 pairs well with epithalon, SS-31, and NAD+. No drug interactions have been identified with any commonly used peptides. Different peptides should be administered at separate injection sites if given on the same day. Always begin with one peptide at a time to identify individual response before adding combinations.
References
- Goldstein AL, Low TL, McAdoo M, et al. Thymosin alpha-1: isolation and sequence analysis of an immunologically active thymic polypeptide. Proceedings of the National Academy of Sciences. 1977;74(2):725-729. DOI: 10.1073/pnas.74.2.725.
- Mutchnick MG, Appelman HD, Chung HT, et al. Thymosin treatment of chronic hepatitis B: a placebo-controlled pilot trial. Hepatology. 1991;14(3):409-415. DOI: 10.1002/hep.1840140304.
- Chan HL, Tang JL, Tam W, et al. The efficacy of thymosin in patients with chronic hepatitis B virus infection: a meta-analysis. Alimentary Pharmacology & Therapeutics. 2001;15(12):1899-1905. DOI: 10.1046/j.1365-2036.2001.01122.x.
- Iino S, Toyota J, Kumada H, et al. The efficacy and safety of thymosin alpha-1 in Japanese patients with chronic hepatitis B; results from a randomized clinical trial. Journal of Viral Hepatitis. 2005;12(3):300-306. DOI: 10.1111/j.1365-2893.2005.00633.x. PMID: 15850471.
- Sherman KE, Sjogren M, Creager RL, et al. Combination therapy with thymosin alpha1 and interferon for the treatment of chronic hepatitis C infection: a randomized, placebo-controlled double-blind trial. Hepatology. 1998;27(4):1128-1135. DOI: 10.1002/hep.510270430. PMID: 9537454.
- Pica F, Gaziano R, Casalinuovo IA, et al. Thymosin alpha-1 activates the TLR9/MyD88/IRF7 signalling pathway in plasmacytoid dendritic cells. International Immunopharmacology. 2014;21(1):205-212. DOI: 10.1016/j.intimp.2014.04.022.
- Romani L, Bistoni F, Perruccio K, et al. Thymosin alpha 1 activates dendritic cell tryptophan catabolism and establishes a regulatory environment for balance of inflammation and tolerance. Blood. 2006;108(7):2265-2274. DOI: 10.1182/blood-2006-02-004762.
- Garaci E, Pica F, Matteucci C, et al. Historical review on thymosin alpha 1 in oncology: preclinical and clinical experiences. Expert Opinion on Biological Therapy. 2015;15(Suppl 1):S31-S39. DOI: 10.1517/14712598.2015.1017466.
- Tuthill C, Rios I, McBeath R. Thymosin alpha 1: past clinical experience and future promise. Annals of the New York Academy of Sciences. 2010;1194:130-135. DOI: 10.1111/j.1749-6632.2010.05482.x.
- Romani L, Moretti S, Fallarino F, et al. Jack of all trades: thymosin alpha 1 and its pleiotropy. Annals of the New York Academy of Sciences. 2012;1269:1-6. DOI: 10.1111/j.1749-6632.2012.06716.x.
- Li J, Liu CH, Wang FS. Thymosin alpha 1: biological activities, applications and genetic engineering production. Peptides. 2010;31(11):2151-2158. DOI: 10.1016/j.peptides.2010.07.026.
- Liu Y, Pan W, Yang S, et al. Thymosin alpha-1 reduces the mortality of severe COVID-19 by restoration of lymphocytopenia and reversion of exhausted T cells. Clinical Infectious Diseases. 2020;71(16):2150-2157. DOI: 10.1093/cid/ciaa630. PMID: 32442287.
- Wang P, Wang C, Chen D. Efficacy of thymosin-alpha-1 in patients with COVID-19: a systematic review and meta-analysis. Journal of International Medical Research. 2023;51(10):3000605231197922. DOI: 10.1177/1721727X231197922.
- Tao N, Sussman S, Bhatt DK, et al. Thymosin alpha1 use in adult COVID-19 patients: a systematic review and meta-analysis on clinical outcomes. International Immunopharmacology. 2022;113:109584. DOI: 10.1016/j.intimp.2022.109584.
- Dominari A, Hathaway D III, Pandav K, et al. The efficacy of thymosin-alpha-1 therapy in moderate to critical COVID-19 patients: a systematic review, meta-analysis, and meta-regression. Inflammopharmacology. 2023;31:1237-1248. DOI: 10.1007/s10787-023-01354-2.
- Wu J, Zhou L, Liu J, et al. The efficacy of thymosin alpha 1 for severe sepsis (ETASS): a multicenter, single-blind, randomized and controlled trial. Critical Care. 2013;17:R8. DOI: 10.1186/cc11932.
- Maio M, Mackiewicz A, Testori A, et al. Large randomized study of thymosin alpha 1, interferon alfa, or both in combination with dacarbazine in patients with metastatic melanoma. Journal of Clinical Oncology. 2010;28(10):1780-1787. DOI: 10.1200/JCO.2009.25.5208.
- Lv J, Gao D, Zhang Y, et al. Thymosin alpha-1-Fc modulates the immune system and down-regulates the progression of melanoma and breast cancer with a prolonged half-life. Scientific Reports. 2018;8:12351. DOI: 10.1038/s41598-018-30956-y.
- Nayersina R, Fowler P, Guilhot S, et al. HLA A2 restricted cytotoxic T lymphocyte responses to multiple hepatitis B surface antigen epitopes during hepatitis B virus infection. Journal of Immunology. 1993;150(10):4659-4671.
- Ciancio A, Andreone P, Kaiser S, et al. Thymosin alpha-1 with peginterferon alfa-2a/ribavirin for chronic hepatitis C not responsive to IFN/ribavirin: an adjuvant role? Journal of Viral Hepatitis. 2012;19(Suppl 1):52-59. DOI: 10.1111/j.1365-2893.2011.01524.x. PMID: 22233415.
- Lee JS, Suh KW, Han Y, et al. Thymosin alpha-1 therapy improves postoperative survival after curative resection for solitary hepatitis B virus-related hepatocellular carcinoma: a propensity score matching analysis. World Journal of Gastroenterology. 2021;27(20):2571-2585. DOI: 10.3748/wjg.v27.i20.2571.
- Costantini C, Bellet MM, Pariano M, et al. Aging and thymosin alpha-1. International Journal of Molecular Sciences. 2025;26(23):11470. DOI: 10.3390/ijms262311470.
- Camerini R, Garaci E. Historical review of thymosin alpha 1 in infectious diseases. Expert Opinion on Biological Therapy. 2015;15(Suppl 1):S117-S127. DOI: 10.1517/14712598.2015.1033394.
- Matteucci C, Grelli S, Balestrieri E, et al. Thymosin alpha 1 and HIV-1: recent advances and future perspectives. Future Microbiology. 2017;12(2):141-155. DOI: 10.2217/fmb-2016-0125.
- King R, Tuthill C. Immune modulation with thymosin alpha 1 treatment. Vitamins and Hormones. 2016;102:151-178. DOI: 10.1016/bs.vh.2016.04.003. PMID: 27450734.
- Billich A. Thymosin alpha 1 (SciClone Pharmaceuticals). Current Opinion in Investigational Drugs. 2002;3(5):698-707. PMID: 12090542.
- Moscarella S, Buzzelli G, Romanelli RG, et al. Interferon and thymosin combination therapy in naive patients with chronic hepatitis C: preliminary results. Liver. 1998;18(5):366-369. DOI: 10.1111/j.1600-0676.1998.tb00821.x. PMID: 9831367.
- Zhang Y, Chen H, Li R, et al. The efficacy and safety study of thymosin alpha-1 combined with PD-1 antibodies as adjuvant therapy in patients with hepatocellular carcinoma with high-risk recurrence factors after radical resection. Journal of Clinical Oncology. 2024;42(16_suppl):e16226. DOI: 10.1200/JCO.2024.42.16_suppl.e16226.
- Liu M, Li Z, Liu Y, et al. Safety and efficacy of loading-dose thymosin alpha-1 in patients with advanced and refractory solid tumors with lower absolute T lymphocyte. Journal of Clinical Oncology. 2023;41(16_suppl):e14543. DOI: 10.1200/JCO.2023.41.16_suppl.e14543.
- Zhang S, Wang X, Zhao L, et al. A preliminary analysis of integrating thymosin alpha-1 into concurrent chemoradiotherapy and consolidative immunotherapy in unresectable locally advanced non-small cell lung cancer. Translational Lung Cancer Research. 2024;13(4):767-779.
- Zhang T, Chen Y, Li H, et al. The efficacy and safety of thymosin alpha-1 combined with lenvatinib plus sintilimab in unresectable hepatocellular carcinoma: a retrospective study. Scientific Reports. 2025;15:97160. DOI: 10.1038/s41598-025-97160-7.
- Salvati A, Scaramuzza S, Bardelli C, et al. Comprehensive review of the safety and efficacy of thymosin alpha 1 in human clinical trials. Journal of Biological Regulators and Homeostatic Agents. 2024;38(1):88-104. PMID: 38308608.
- Ancona G, Merlini S, D'Arminio Monforte A, et al. The immunomodulatory activity of thymosin alpha 1 on tumor cell lines and distinct immune cell subsets. OncoTargets and Therapy. 2025;18:109-122.
- Serafino A, Pierimarchi P, Pica F, et al. Thymosin alpha 1 as a stimulatory agent of innate cell-mediated immune response. Annals of the New York Academy of Sciences. 2012;1270:13-20. DOI: 10.1111/j.1749-6632.2012.06707.x.
- Tuthill CW, King RS. Thymosin alpha 1 - a peptide immune modulator with a broad range of clinical applications. Clinical and Experimental Pharmacology. 2013;3:133. DOI: 10.4172/2161-1459.1000133.
- Zadaxin (thymosin alpha 1): Comprehensive prescribing information. SciClone Pharmaceuticals. 2006.
- Maio M, Chelazzi M, Pica F, et al. Phenotypic drug discovery: a case for thymosin alpha-1. Frontiers in Medicine. 2024;11:1388959. DOI: 10.3389/fmed.2024.1388959.