Executive Summary
The GLP-1 receptor agonist shortage that began in 2022 and peaked through 2024 disrupted treatment for millions of patients managing obesity and type 2 diabetes. While the FDA resolved the semaglutide shortage in February 2025 and tirzepatide came off the shortage list in December 2024, the aftermath continues to reshape how patients access these medications - from compounding restrictions to insurance battles to new switching protocols between agents.
Key Takeaways
- The FDA officially resolved the semaglutide injection shortage on February 21, 2025, after nearly three years of supply disruptions driven by a 442% surge in prescriptions between 2021 and 2023.
- Compounding pharmacies faced strict wind-down deadlines: 503A pharmacies had until April 22, 2025, and 503B outsourcing facilities until May 22, 2025, to stop compounding semaglutide.
- No FDA-approved dose equivalency exists between semaglutide and tirzepatide - all patients switching to tirzepatide must start at the 2.5 mg introductory dose regardless of prior semaglutide dose.
- Brand-name GLP-1 medications now range from $199-$449/month with manufacturer programs, compared to $100-$300/month for compounded versions that were previously available.
- Treatment interruption studies show patients regain approximately two-thirds of lost weight within one year of discontinuing semaglutide, underscoring the need for uninterrupted access.
This guide covers everything patients, clinicians, and researchers need to know about maintaining treatment continuity in a post-shortage environment. We'll walk through what caused the shortage, which alternatives exist, how to safely switch between GLP-1 medications, the current legal status of compounded formulations, and what non-GLP-1 options might fill the gap when preferred medications aren't available.
Key Takeaways
- The FDA officially resolved the semaglutide injection shortage on February 21, 2025, after nearly three years of supply disruptions driven by a 442% surge in prescriptions between 2021 and 2023.
- Compounding pharmacies faced strict wind-down deadlines: 503A pharmacies had until April 22, 2025, and 503B outsourcing facilities until May 22, 2025, to stop compounding semaglutide.
- No FDA-approved dose equivalency exists between semaglutide and tirzepatide - all patients switching to tirzepatide must start at the 2.5 mg introductory dose regardless of prior semaglutide dose.
- Brand-name GLP-1 medications now range from $199-$449/month with manufacturer programs, compared to $100-$300/month for compounded versions that were previously available.
- Treatment interruption studies show patients regain approximately two-thirds of lost weight within one year of discontinuing semaglutide, underscoring the need for uninterrupted access.
- Alternative GLP-1 agents (liraglutide, dulaglutide), non-GLP-1 compounds (tesofensine, 5-amino-1MQ), and emerging pipeline drugs offer options when first-line agents aren't accessible.
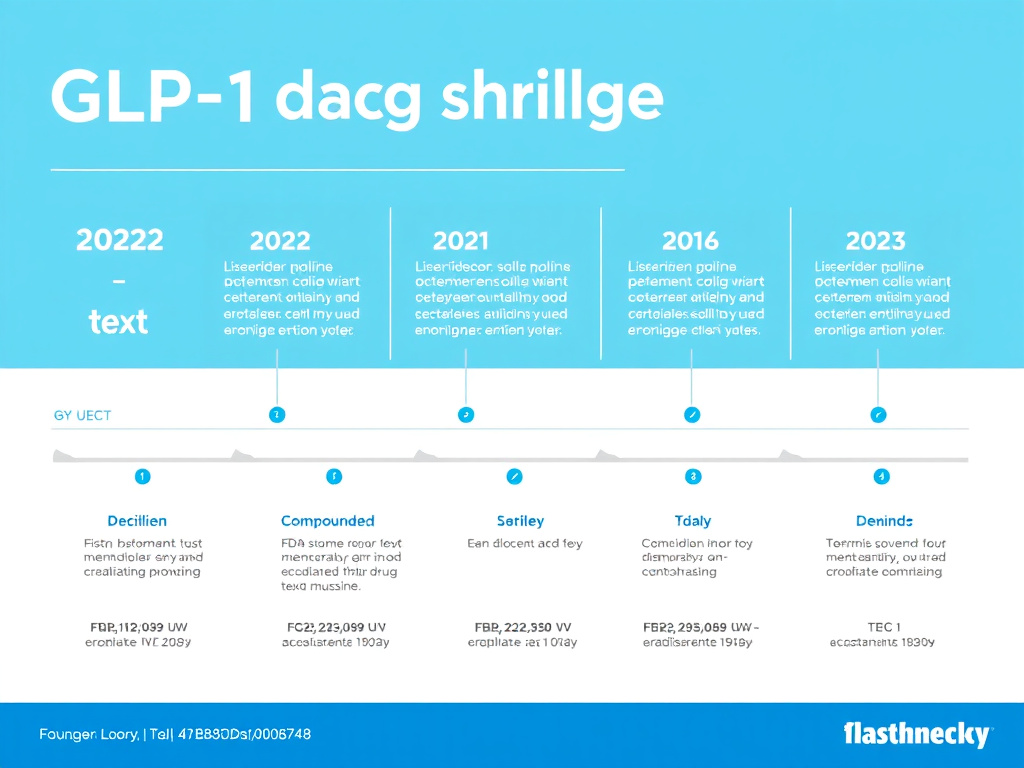
Figure 1: GLP-1 drug shortage timeline highlighting major regulatory milestones from initial shortage declaration through resolution and compounding wind-down periods.
The shortage wasn't just an inconvenience. For patients with type 2 diabetes, gaps in GLP-1 therapy meant deteriorating glycemic control and increased cardiovascular risk. For those using these medications for weight management, treatment interruptions triggered rapid weight regain that often proved difficult to reverse even after resuming therapy. The economic ripple effects touched compounding pharmacies, telehealth startups, insurance companies, and the broader pharmaceutical supply chain.
What makes the current situation particularly complex is the intersection of resolved shortages with ongoing access barriers. Even though semaglutide and tirzepatide are technically back in stock, many patients still can't get them due to insurance denials, prior authorization requirements, or cost. Understanding the full range of alternatives - from FDA-approved substitutes to research compounds to lifestyle interventions - has never been more relevant.
Throughout this guide, we reference peer-reviewed clinical data, FDA regulatory documents, and manufacturer communications to give you the most accurate picture of where things stand and what options are available. Whether you're a clinician helping patients through transitions, a researcher studying the pharmacoeconomics of obesity treatment, or a patient trying to maintain your progress, this resource aims to be the reference you need.
Understanding the Shortage
The GLP-1 shortage wasn't a single event but rather a cascading supply-demand mismatch that evolved over three years. Understanding what happened and why helps contextualize the current regulatory environment and the options available today.
The Demand Explosion: 2021-2024
Between January 2021 and December 2023, semaglutide prescriptions increased by 442% according to pharmacy claims data. This wasn't a gradual uptick - it was an unprecedented surge driven by several converging factors that no pharmaceutical manufacturer could have anticipated at scale.
The catalyst was a shift in public perception. When the STEP trials demonstrated that semaglutide 2.4 mg could produce average weight loss of 14.9% at 68 weeks - roughly 35 pounds for someone weighing 230 pounds - the conversation around obesity treatment changed fundamentally (Wilding et al., 2021; DOI: 10.1056/NEJMoa2032183). Media coverage amplified awareness. Celebrity endorsements, whether intended or not, pushed demand into territory that Novo Nordisk's manufacturing infrastructure simply wasn't built to handle.
Several factors converged simultaneously:
- Expanded indications: Wegovy received FDA approval for chronic weight management in June 2021, opening semaglutide to a vastly larger patient population beyond type 2 diabetes.
- Off-label prescribing: Ozempic, approved for diabetes, was increasingly prescribed off-label for weight loss at lower doses, further straining supply meant for diabetic patients.
- Telehealth expansion: Post-pandemic telehealth infrastructure made it dramatically easier to obtain prescriptions without traditional in-office visits. Dozens of telehealth platforms launched specifically around GLP-1 prescribing.
- Social media amplification: Viral content on TikTok, Instagram, and other platforms created awareness cycles that traditional pharmaceutical marketing never could have achieved.
- Insurance coverage expansion: Major insurers began adding coverage for GLP-1s for weight management, removing the cost barrier for millions of potential patients.
The Supply Side: Manufacturing Realities
Peptide drug manufacturing isn't like producing a small-molecule pill. GLP-1 receptor agonists require complex biological synthesis, extensive purification, sterile fill-and-finish processes, and cold-chain distribution. Scaling up production takes years, not months.
Novo Nordisk invested heavily, but capacity expansion operates on pharmaceutical timescales. The company committed $6 billion to expanding production facilities, including a $4.1 billion investment in a second fill-and-finish manufacturing facility in Clayton, North Carolina. They expanded operations in Denmark, France, Brazil, and China. But construction timelines for pharmaceutical-grade manufacturing facilities typically run 3-5 years from initial construction to GMP-qualified production.
Capital expenditures reached approximately DKK 60 billion (roughly $8.5 billion) in 2025, with DKK 55 billion projected for 2026. The vast majority of this investment went toward increasing active pharmaceutical ingredient (API) capacity for semaglutide production. These figures reflect the scale of the challenge - even billions of dollars in investment couldn't instantly resolve a supply gap of this magnitude.
Figure 2: Supply-demand divergence for semaglutide products, illustrating the gap between exponential prescription growth and gradual manufacturing capacity expansion.
The FDA Shortage List: Timeline and Implications
The FDA maintains a Drug Shortage Database that tracks medications with supply issues. When a drug appears on this list, it triggers specific regulatory consequences - most significantly for compounding pharmacies.
Under the Federal Food, Drug, and Cosmetic Act (FD&C Act), compounding pharmacies generally cannot produce copies of commercially available, FDA-approved drugs. However, Section 503A and 503B provide an exception: when a drug is on the FDA shortage list, compounders can step in to fill the gap. This exception became the legal basis for the entire compounded semaglutide industry.
Here's how the timeline unfolded:
| Date | Event | Impact |
|---|---|---|
| March 2022 | Semaglutide injection first listed as shortage | Compounding pharmacies begin legally producing semaglutide |
| Throughout 2023 | All dose strengths intermittently short | Patients struggle to fill prescriptions; compounding market grows rapidly |
| Early 2024 | Shortage reaches peak severity | Some patients face weeks-long waits; switching between agents increases |
| December 19, 2024 | Tirzepatide removed from shortage list | Compounding pharmacies given wind-down period for tirzepatide |
| February 21, 2025 | Semaglutide injection shortage resolved | 60-day (503A) and 90-day (503B) wind-down periods begin |
| April 22, 2025 | 503A compounding deadline | Traditional pharmacies must stop compounding semaglutide |
| May 22, 2025 | 503B outsourcing facility deadline | Outsourcing facilities must stop compounding semaglutide |
| October 2025 | Last Wegovy dose (0.25 mg) confirmed available | Full supply restoration confirmed across all dose strengths |
Who Was Affected Most?
The shortage hit different patient populations in different ways, and it's worth understanding these distinctions because they inform which alternatives and strategies are most relevant.
Type 2 diabetes patients faced the most medically urgent consequences. For these patients, GLP-1 therapy isn't elective - it's a core component of glycemic management. Treatment gaps meant rising HbA1c levels, increased hypoglycemia risk from compensatory insulin dose adjustments, and potential cardiovascular deterioration. The American Diabetes Association issued specific guidance for clinicians managing patients through shortage-related treatment interruptions (ADA, 2023).
Weight management patients experienced rapid weight regain during gaps. Data from the STEP 1 trial extension showed that participants regained approximately two-thirds of their weight loss within one year of discontinuing semaglutide (Wilding et al., 2022; DOI: 10.1111/dom.14725). This finding has profound implications for patients who lost access during the shortage, as regained weight proved difficult to re-lose even after resuming therapy.
Compounding pharmacy patients - often those without insurance coverage for brand-name medications - faced a double disruption. First, they experienced the same supply challenges as everyone else. Then, as the shortage resolved, they lost access to the more affordable compounded versions entirely. For patients who had been paying $150-$300/month for compounded semaglutide, the transition to $349-$1,000+/month brand-name products represented a dramatic cost increase.
Lessons from the Shortage
The GLP-1 shortage exposed several structural vulnerabilities in pharmaceutical supply chains that are relevant beyond just this drug class:
- Single-source dependency: With Novo Nordisk as the sole manufacturer of semaglutide, there was no backup production capacity when demand outpaced supply. The pharmaceutical industry's consolidation of manufacturing for complex biologics creates inherent fragility.
- Demand forecasting failures: Traditional pharmaceutical demand models based on disease prevalence couldn't account for viral social media dynamics or the expansion of telehealth prescribing platforms.
- Regulatory rigidity: The binary nature of the shortage list (on or off) created cliff-edge transitions for compounding pharmacies rather than allowing graduated wind-down periods proportional to supply recovery.
- Insurance lag: Even as brand-name supply recovered, insurance formulary decisions, prior authorization requirements, and step therapy protocols created persistent access barriers that the shortage list designation had temporarily circumvented.
Understanding these dynamics is essential context for the rest of this guide. The alternatives, switching protocols, and access strategies we'll discuss all exist within this framework of resolved shortages but persistent access challenges.
Brand vs Compounded GLP-1s
The distinction between FDA-approved brand-name GLP-1 medications and compounded versions sits at the center of the post-shortage landscape. Understanding the differences in formulation, regulation, safety, and legal status is critical for informed decision-making.
What Are Brand-Name GLP-1 Medications?
Brand-name GLP-1 receptor agonists have gone through the full FDA approval process, including Phase 1, 2, and 3 clinical trials involving thousands of participants. They're manufactured under current Good Manufacturing Practice (cGMP) regulations, with every batch tested for potency, sterility, purity, and stability. The FDA-approved GLP-1 medications currently on the market include:
| Brand Name | Generic Name | Manufacturer | Approved Indications | Dosing Frequency |
|---|---|---|---|---|
| Ozempic | Semaglutide | Novo Nordisk | Type 2 diabetes | Weekly injection |
| Wegovy | Semaglutide | Novo Nordisk | Chronic weight management; CV risk reduction | Weekly injection |
| Rybelsus | Oral semaglutide | Novo Nordisk | Type 2 diabetes | Daily oral tablet |
| Mounjaro | Tirzepatide | Eli Lilly | Type 2 diabetes | Weekly injection |
| Zepbound | Tirzepatide | Eli Lilly | Chronic weight management | Weekly injection |
| Victoza | Liraglutide | Novo Nordisk | Type 2 diabetes | Daily injection |
| Saxenda | Liraglutide | Novo Nordisk | Chronic weight management | Daily injection |
| Trulicity | Dulaglutide | Eli Lilly | Type 2 diabetes | Weekly injection |
Each of these products uses a specific formulation that has been extensively studied. The delivery devices (auto-injectors, pens) are engineered for accurate dosing. The drug substance itself meets rigorous specifications for the exact molecular form, concentration, and stability profile.
What Are Compounded GLP-1 Medications?
Compounded medications are prepared by licensed pharmacies - either traditional 503A pharmacies or 503B outsourcing facilities - using bulk pharmaceutical ingredients. During the semaglutide shortage, compounding pharmacies legally produced semaglutide injections as an alternative to unavailable brand-name products.
However, compounded versions differ from brand-name products in several important ways:
- No FDA approval: Compounded drugs don't go through the FDA approval process. They're not evaluated for safety and efficacy through clinical trials.
- Variable formulations: Compounders may use different salt forms of semaglutide (such as semaglutide sodium or semaglutide acetate) rather than the base form used in Ozempic and Wegovy. The FDA has noted that it lacks information on whether these salt forms have identical chemical and pharmacologic properties (FDA, 2024).
- Different delivery systems: Instead of pre-filled, pre-dosed injection pens, compounded semaglutide typically comes in multi-dose vials requiring patients to draw up their own doses with syringes - a process that introduces measurement error.
- Manufacturing standards vary: While 503B facilities must follow cGMP regulations, 503A pharmacies operate under less stringent state-level oversight. Quality control testing may be less comprehensive than what FDA-approved manufacturers perform.
FDA Safety Alert
As of July 2025, the FDA received 605 adverse event reports associated with compounded semaglutide and 545 reports for compounded tirzepatide. Reported issues include dosing errors resulting in 5-20x the intended dose, sterility concerns, and products with fraudulent labeling from non-existent pharmacies. These figures likely represent significant underreporting.
The Salt Form Controversy
One of the most technically important distinctions between brand and compounded semaglutide involves the molecular form of the active ingredient. FDA-approved semaglutide products use the base form of the molecule. Many compounding pharmacies, however, sourced semaglutide sodium or semaglutide acetate - salt forms that may behave differently in the body.
The concern isn't trivial. Salt forms can affect solubility, absorption rate, bioavailability, and stability. A semaglutide sodium preparation might deliver a different effective dose than the same milligram amount of base semaglutide. Without bioequivalence studies comparing these salt forms to the approved product, there's genuine scientific uncertainty about whether patients were receiving therapeutically equivalent treatment.
The FDA specifically flagged this issue, stating that some compounded products contain salt forms that represent "different active ingredients" from those in approved drugs - a distinction with regulatory implications under the "essentially a copy" prohibition.
Quality and Safety Comparison
| Parameter | Brand-Name (FDA-Approved) | Compounded (503B) | Compounded (503A) |
|---|---|---|---|
| Clinical trials | Phase 1-3 with thousands of participants | None required | None required |
| FDA oversight | Full pre-market and post-market | FDA registration, inspection, cGMP | Primarily state board oversight |
| Sterility testing | Every batch | Required per cGMP | Varies by state |
| Potency verification | Validated analytical methods | Required but methods vary | Varies significantly |
| Stability data | Extensive (ICH guidelines) | Beyond-use dating, limited | Beyond-use dating, limited |
| Adverse event reporting | Mandatory (MedWatch) | Mandatory | Not required federally |
| Delivery device | Pre-filled, pre-dosed pen | Multi-dose vial + syringe | Multi-dose vial + syringe |
| Dosing accuracy | Pen dial mechanism (high) | Patient-dependent (variable) | Patient-dependent (variable) |
When Compounded GLP-1s May Still Be Legally Available
Even after the shortage resolution and wind-down deadlines, narrow exceptions exist for compounded semaglutide. A compounding pharmacy may still prepare semaglutide if:
- A patient has a documented allergy to an inactive ingredient in the FDA-approved product (such as a specific excipient or preservative).
- A clinically necessary dose strength isn't available in commercial products.
- The compounded formulation is "meaningfully different" from the commercially available product - not merely a lower-cost substitute.
These exceptions are narrow, and both the FDA and brand-name manufacturers (Novo Nordisk and Eli Lilly) have been aggressive in enforcement. Novo Nordisk and Eli Lilly have sent cease-and-desist letters to compounders, and the FDA has issued more than 50 warning letters to GLP-1 compounding operations as of early 2026.
Compounding Legal Framework (503A/503B)
The legal framework governing compounded medications - particularly GLP-1 receptor agonists - involves federal statutes, FDA enforcement discretion, and state-level regulation. Understanding these layers helps clarify what's currently legal and what risks exist for patients and providers.
Section 503A: Traditional Compounding Pharmacies
Section 503A of the FD&C Act covers traditional compounding pharmacies that prepare medications for individual patients based on specific prescriptions. These pharmacies receive several exemptions from federal requirements that apply to drug manufacturers, including:
- Exemption from FDA pre-approval requirements
- Exemption from cGMP manufacturing standards
- Exemption from specific labeling requirements
In exchange for these exemptions, 503A pharmacies must operate within defined boundaries. They must compound based on a valid, patient-specific prescription from a licensed practitioner. They cannot compound drugs that are "essentially copies" of commercially available products (with the shortage exception). They cannot advertise or promote specific compounded products. And they're primarily regulated by their state board of pharmacy rather than the FDA directly.
During the semaglutide shortage, 503A pharmacies operated under the shortage exception, legally preparing semaglutide injections for individual patients with prescriptions. When the FDA resolved the shortage on February 21, 2025, these pharmacies received a 60-day wind-down period ending April 22, 2025.
Section 503B: Outsourcing Facilities
Section 503B outsourcing facilities were created by the Drug Quality and Security Act of 2013, passed in response to the New England Compounding Center (NECC) fungal meningitis outbreak that killed 76 people. These facilities occupy a middle ground between traditional pharmacies and full manufacturers.
503B facilities must:
- Register with the FDA
- Comply with cGMP requirements
- Submit to FDA inspections
- Report adverse events to the FDA
- Provide the FDA with product lists
- Label products with appropriate information
The key advantage of 503B facilities is that they can produce larger batches of compounded drugs without patient-specific prescriptions, distributing them to healthcare providers for office use. This made them particularly important during the GLP-1 shortage, as they could supply telehealth platforms and clinics at scale.
503B facilities received a 90-day wind-down period after the shortage resolution, with their deadline falling on May 22, 2025.

Figure 3: Regulatory framework comparison between 503A traditional compounding pharmacies and 503B outsourcing facilities, highlighting key differences in oversight and capabilities.
The "Essentially a Copy" Doctrine
At the heart of the compounding legal framework is the "essentially a copy" restriction. Under both 503A and 503B, compounding pharmacies generally cannot produce drugs that are identical or nearly identical to FDA-approved products when those products are commercially available.
The FDA defines "essentially a copy" broadly. A compounded drug is essentially a copy if it's identical or nearly identical in active ingredient, route of administration, dosage form, strength, and excipient composition to an approved product. The intent is to prevent compounding pharmacies from functioning as unlicensed manufacturers producing cheaper versions of brand-name drugs.
The shortage exception carved out a temporary pathway. When an FDA-approved drug was listed as being in shortage, compounders could legally produce copies to meet patient needs. Once the shortage resolved, the exception expired - with the wind-down periods providing a transition buffer.
Current Legal Status: Post-Shortage Compounding
As of early 2026, the legal landscape for compounded semaglutide and tirzepatide has largely settled - though litigation continues. The current status:
- Semaglutide: Compounding deadlines have passed. 503A pharmacies technically remain the only entities that can compound semaglutide under narrow clinical-need exceptions, with valid patient-specific prescriptions and documented medical necessity.
- Tirzepatide: The FDA ended compounding discretion for tirzepatide, with deadlines having passed.
- Enforcement: The FDA has issued over 50 warning letters to GLP-1 compounders. Novo Nordisk and Eli Lilly have sent cease-and-desist letters to pharmacies continuing to compound their patented molecules.
- Legal challenges: Several compounding pharmacy associations filed lawsuits challenging the FDA's shortage resolution determination and the resulting compounding restrictions. These cases remain in various stages of litigation.
State-Level Variations
Compounding regulations vary significantly by state. Some states have enacted laws that are more permissive than federal standards, while others impose additional restrictions. Patients and providers should consult their state board of pharmacy for jurisdiction-specific guidance. Several states have introduced legislation specifically addressing GLP-1 compounding access.
FDA Enforcement Actions
The FDA's enforcement posture toward GLP-1 compounders has been aggressive. Beyond warning letters, the agency has taken several significant actions:
- Sterility concerns: Inspections of facilities like Fullerton Wellness raised concerns about sterility of compounded semaglutide and tirzepatide injections, with the FDA noting that non-sterile injectable products can cause life-threatening infections and sepsis.
- Fraudulent products: The FDA identified fraudulent compounded semaglutide and tirzepatide products with labels from pharmacies that don't exist, raising concerns about product integrity across the compounding marketplace.
- Dosing error alerts: Multiple adverse event reports involving patients receiving 5-20 times the intended dose prompted FDA safety communications about the risks of self-measured dosing from multi-dose vials.
- Advertising restrictions: The FDA and HHS have taken action against telehealth platforms advertising compounded GLP-1 drugs, particularly those making therapeutic claims without FDA-approved labeling.
For patients who previously relied on compounded GLP-1 medications, the regulatory shift means transitioning to brand-name products, exploring alternative medications, or working with their healthcare provider to document clinical need for continued compounding under the narrow exceptions that remain.
Alternative GLP-1 Medications
When a patient's preferred GLP-1 medication isn't available or accessible - whether due to residual supply issues, insurance restrictions, or cost - several alternative agents within the GLP-1 class and adjacent drug classes offer viable options. Each comes with its own efficacy profile, side effect spectrum, and practical considerations.
Tirzepatide (Mounjaro/Zepbound): The Dual Agonist Alternative
Tirzepatide represents the most potent alternative to semaglutide for both weight management and glycemic control. As a dual GIP/GLP-1 receptor agonist, it activates two incretin pathways simultaneously, producing weight loss outcomes that consistently exceed semaglutide in head-to-head comparisons.
The SURMOUNT-1 trial demonstrated mean body weight reductions of 15.0%, 19.5%, and 20.9% with tirzepatide 5 mg, 10 mg, and 15 mg respectively at 72 weeks, compared to 3.1% with placebo (Jastreboff et al., 2022; DOI: 10.1056/NEJMoa2206038). These figures surpass the 14.9% achieved with semaglutide 2.4 mg in the STEP 1 trial.
For patients switching from semaglutide due to shortage, cost, or inadequate response, tirzepatide offers several advantages:
- Greater mean weight loss at maximum doses
- Superior HbA1c reduction in diabetic patients (SURPASS-2 showed reductions of 2.01-2.30% across doses)
- Dual mechanism may benefit patients who've plateaued on single-agonist therapy
- Available through Eli Lilly's direct-to-consumer programs at $299-$449/month
The main limitations include the requirement to start at 2.5 mg regardless of prior GLP-1 exposure, a titration period of several months to reach therapeutic doses, and a GI side effect profile that's broadly similar to semaglutide.
Liraglutide (Victoza/Saxenda): The Daily GLP-1 Option
Liraglutide was the first GLP-1 receptor agonist approved for both diabetes (Victoza, up to 1.8 mg daily) and weight management (Saxenda, 3.0 mg daily). While it requires daily rather than weekly injections and produces more modest weight loss than semaglutide, it remains a reliable alternative with extensive long-term safety data.
The SCALE Obesity and Prediabetes trial showed mean weight loss of 8.0% with liraglutide 3.0 mg at 56 weeks compared to 2.6% with placebo (Pi-Sunyer et al., 2015; DOI: 10.1056/NEJMoa1411892). While lower than semaglutide's 14.9%, this still represents clinically meaningful weight reduction for many patients.
An important development for liraglutide access: generic versions of liraglutide became available in July 2025, with Teva and Meitheal producing generic alternatives. This has significantly improved overall availability and reduced costs, making liraglutide a particularly attractive option for cost-sensitive patients.
Clinical Pearl
Liraglutide's daily dosing, while less convenient than weekly injections, actually offers one advantage: more granular dose titration. Patients who experienced intolerable GI side effects on weekly semaglutide may find that the ability to make smaller daily dose adjustments with liraglutide allows better symptom management.
Dulaglutide (Trulicity): Weekly Diabetes-Focused Option
Dulaglutide remains available as a weekly GLP-1 receptor agonist primarily indicated for type 2 diabetes. While it's not FDA-approved for weight management specifically, it does produce meaningful weight loss as a secondary benefit.
The AWARD trial program showed HbA1c reductions of 1.1-1.5% and body weight reductions of 1.5-3.0 kg with dulaglutide across studies. The REWIND cardiovascular outcomes trial demonstrated a 12% reduction in major adverse cardiovascular events (MACE) in patients with type 2 diabetes (Gerstein et al., 2019; DOI: 10.1016/S0140-6736(19)31149-3).
Dulaglutide's primary role as an alternative is for diabetic patients who need a weekly GLP-1 but can't access semaglutide or tirzepatide. Its weight loss effects are more modest, but the cardiovascular benefits and glycemic control are well-established.
Exenatide (Byetta/Bydureon): Historical Context
Exenatide, the first GLP-1 receptor agonist to reach the U.S. market, was available as both a twice-daily injection (Byetta) and an extended-release weekly formulation (Bydureon BCise). However, AstraZeneca discontinued Bydureon BCise effective October 15, 2025, narrowing available options within the class.
Byetta (twice-daily exenatide) may still be available in some markets, but its inconvenient dosing schedule and more modest efficacy compared to newer agents make it a last-resort alternative rather than a preferred switch option.
Oral Semaglutide: A Different Delivery Route
For patients specifically having trouble accessing injectable semaglutide, oral formulations offer an alternative pathway to the same active ingredient. Rybelsus (oral semaglutide) is FDA-approved for type 2 diabetes at doses up to 14 mg daily.
In January 2026, Novo Nordisk launched oral semaglutide 25 mg once daily for obesity, providing a non-injectable option for weight management. This represents a significant access improvement for patients who prefer or require oral administration and may be more readily available than injectable formulations.
Key considerations for oral semaglutide:
- Must be taken on an empty stomach with no more than 4 oz of plain water
- No eating, drinking, or other oral medications for at least 30 minutes after
- Oral bioavailability is approximately 1% - significantly lower than injectable, requiring higher doses to achieve similar effects
- May have a different supply chain than injectable formulations, providing availability when injections are constrained
Comparative Efficacy Summary
| Medication | Max Dose | Frequency | Weight Loss (%) | HbA1c Reduction | CV Benefit Proven |
|---|---|---|---|---|---|
| Semaglutide (Wegovy) | 2.4 mg | Weekly SC | ~14.9% | ~1.5-1.8% | Yes (SELECT) |
| Tirzepatide (Zepbound) | 15 mg | Weekly SC | ~20.9% | ~2.0-2.3% | Pending |
| Liraglutide (Saxenda) | 3.0 mg | Daily SC | ~8.0% | ~1.0-1.5% | Yes (LEADER) |
| Dulaglutide (Trulicity) | 4.5 mg | Weekly SC | ~3.0-5.0% | ~1.1-1.5% | Yes (REWIND) |
| Oral semaglutide (25 mg) | 25 mg | Daily oral | ~12-15% | ~1.5-1.8% | Pending |
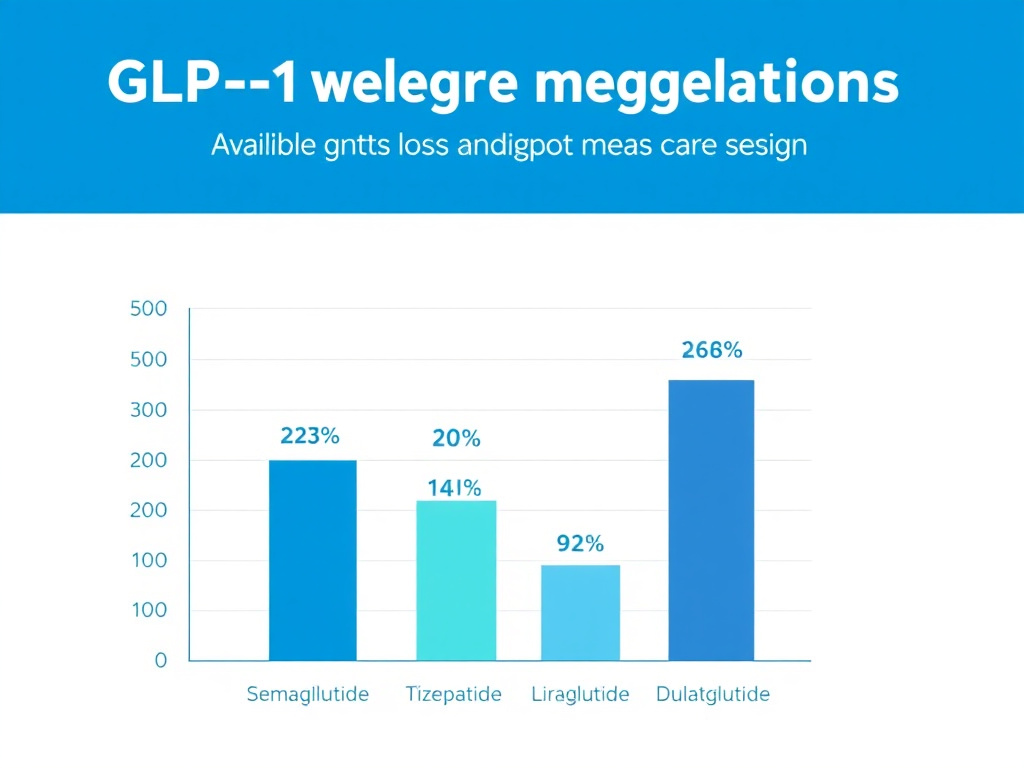
Figure 4: Comparative weight loss efficacy across GLP-1 receptor agonists at maximum approved doses, based on key clinical trial data.
Emerging Pipeline Alternatives
Retatrutide, the triple agonist targeting GLP-1, GIP, and glucagon receptors simultaneously, showed weight loss of up to 24.2% in Phase 2 trials - exceeding even tirzepatide at maximum doses (Jastreboff et al., 2023; DOI: 10.1056/NEJMoa2301972). While still in Phase 3 trials and not yet commercially available, retatrutide represents the next generation of incretin-based therapy and may eventually provide another option for patients who struggle with access to current agents.
Other pipeline candidates include orforglipron (Eli Lilly's oral non-peptide GLP-1 agonist), survodutide (a dual GLP-1/glucagon agonist from Boehringer Ingelheim), and pemvidutide (an investigational dual agonist). For a complete comparison of GLP-1 class agents, see our dedicated research report.
Switching Between GLP-1 Medications
Whether driven by supply issues, insurance requirements, inadequate response, or intolerable side effects, switching between GLP-1 receptor agonists requires careful planning to minimize treatment gaps, manage side effects, and maintain therapeutic progress.
General Principles for Switching
There's no FDA-approved conversion guide between GLP-1 medications, and the existing clinical guidance is based largely on pharmacologic principles, clinical experience, and expert consensus rather than randomized switching trials. That said, several foundational principles guide safe transitions:
- Start low with the new agent. Regardless of the dose a patient was on previously, the new GLP-1 should generally be initiated at the lowest available dose and titrated upward per the standard schedule.
- Time the switch to minimize gaps. The new agent should ideally be started when the next dose of the previous agent would have been due - not sooner, to avoid receptor overstimulation and severe GI effects.
- Expect a transition period. Patients may experience temporary worsening of GI side effects, changes in appetite suppression, and fluctuations in weight or blood glucose during the switch.
- Don't assume equal potency at equal milligrams. A milligram of semaglutide is not equivalent to a milligram of tirzepatide or liraglutide. These are different molecules with different receptor binding affinities and pharmacokinetic profiles.
- Monitor glycemic control closely in diabetic patients. Treatment transitions create periods of variable GLP-1 receptor activation that can affect blood glucose unpredictably.
Semaglutide to Tirzepatide: The Most Common Switch
This is by far the most frequently performed GLP-1 switch, driven by patients seeking greater efficacy, insurance formulary changes, or provider recommendations. The key protocol considerations:
Standard Switching Protocol
All patients switching from semaglutide must start tirzepatide at 2.5 mg weekly regardless of their previous semaglutide dose, including patients on the maximum 2.4 mg dose. This is because tirzepatide's dual GIP/GLP-1 mechanism creates a pharmacologically distinct effect profile that cannot be predicted from single-agonist exposure.
Timing: Start tirzepatide 2.5 mg on the day the next semaglutide injection would have been due. Since both are weekly medications, this creates a seamless weekly injection schedule. Do not overlap - taking both agents simultaneously dramatically increases GI adverse effects.
Titration: Follow the standard tirzepatide titration schedule, increasing by 2.5 mg every 4 weeks as tolerated:
| Week | Tirzepatide Dose | Notes |
|---|---|---|
| Weeks 1-4 | 2.5 mg | Introductory dose; GI effects may be mild since patient has GLP-1 tolerance |
| Weeks 5-8 | 5.0 mg | First maintenance dose; assess tolerability |
| Weeks 9-12 | 7.5 mg | Continue if weight loss or glycemic goals not met |
| Weeks 13-16 | 10.0 mg | Many patients achieve adequate response here |
| Weeks 17-20 | 12.5 mg | Continue upward if needed and tolerated |
| Weeks 21+ | 15.0 mg | Maximum dose |
What to expect: Patients who were well-established on semaglutide often tolerate the early titration steps of tirzepatide better than GLP-1-naive patients, since they've already adapted to GLP-1 receptor activation. However, the GIP component is novel, and some patients experience new or different GI symptoms during the transition. Nausea patterns may differ - some patients report more nausea with tirzepatide's early doses even with prior semaglutide tolerance.
Weight trajectory: Expect a temporary plateau or slight regain during the low-dose titration period. Patients who were losing weight on semaglutide 2.4 mg may not see continued loss at tirzepatide 2.5 mg or even 5 mg. Meaningful additional weight loss typically resumes at 7.5-10 mg and above.
Tirzepatide to Semaglutide: The Reverse Switch
Less common but sometimes necessary - particularly when insurance changes or supply issues affect tirzepatide access. The principles are similar but reversed:
- Start semaglutide at 0.25 mg weekly regardless of prior tirzepatide dose.
- Follow standard titration: 0.25 mg x 4 weeks, then 0.5 mg x 4 weeks, then 1.0 mg, then 1.7 mg, then 2.4 mg for weight management.
- Time the first semaglutide injection for when the next tirzepatide dose would have been due.
- Patients switching from high-dose tirzepatide (10-15 mg) to semaglutide may experience some weight regain during titration, as semaglutide's single-agonist mechanism produces somewhat less weight loss at equivalent time points.
Semaglutide to Liraglutide: Switching to Daily Dosing
This switch involves transitioning from weekly to daily injections - a significant lifestyle change. It's most commonly needed when:
- Insurance formulary changes require liraglutide as a first-line agent
- Patients experience weekly-dosing-related GI peaks that aren't tolerable
- Generic liraglutide availability makes it a more cost-effective option
Protocol: Wait until the next semaglutide dose would have been due (7 days after the last injection). Begin liraglutide at 0.6 mg daily, titrating weekly by 0.6 mg up to the target dose (1.8 mg for diabetes, 3.0 mg for weight management). Some clinicians start at a higher initial dose (1.2 mg) in patients who were established on moderate-to-high dose semaglutide, as these patients already have GLP-1 receptor tolerance.
Between-Agent Dose Comparison (Not Equivalence)
It's critical to understand that these are not dose equivalences - they're rough comparisons of where agents produce similar magnitudes of effect based on clinical trial data. Individual response varies significantly.
| Approximate Effect Level | Semaglutide SC | Tirzepatide SC | Liraglutide SC | Dulaglutide SC |
|---|---|---|---|---|
| Entry/Introductory | 0.25 mg/week | 2.5 mg/week | 0.6 mg/day | 0.75 mg/week |
| Low maintenance | 0.5 mg/week | 5.0 mg/week | 1.2-1.8 mg/day | 1.5 mg/week |
| Moderate effect | 1.0 mg/week | 7.5-10 mg/week | 2.4 mg/day | 3.0 mg/week |
| High/Maximum | 2.4 mg/week | 15 mg/week | 3.0 mg/day | 4.5 mg/week |
Critical Safety Note
Never switch from a submaximal dose of one GLP-1 to the maximum dose of another. The risk of severe gastrointestinal side effects - including persistent vomiting, dehydration, and in rare cases pancreatitis - increases substantially when dose escalation is too aggressive. If a patient experienced significant GI side effects on their previous GLP-1, consider starting even lower than standard or extending the titration intervals.
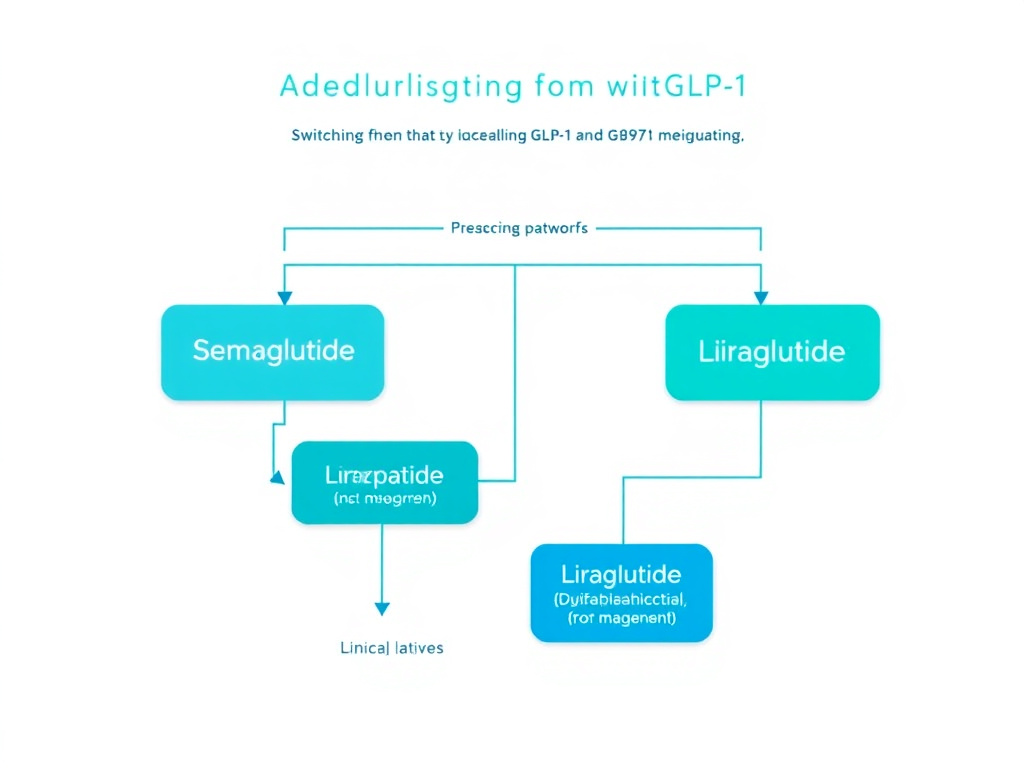
Figure 5: Clinical decision flowchart for GLP-1 switching protocols, including timing, starting doses, and titration considerations.
Managing GI Side Effects During Switches
The gastrointestinal side effects of GLP-1 agonists - nausea, vomiting, diarrhea, constipation, and abdominal discomfort - typically intensify during dose transitions. Several strategies can help patients through the switch period:
- Dietary modifications: Smaller, more frequent meals. Avoid high-fat and fried foods. Eat slowly and stop at the first sign of fullness. Bland, low-residue foods during the first 1-2 weeks at each new dose.
- Hydration: Adequate fluid intake is essential, especially if nausea reduces oral intake. Small sips of water, ginger tea, or electrolyte solutions throughout the day.
- Timing of injection: Some patients find that injecting in the evening (before bed) reduces awareness of peak nausea, which typically occurs 1-3 days post-injection.
- Anti-nausea support: Over-the-counter options like ginger supplements or ondansetron (by prescription) can be used short-term during transitions. Metoclopramide should generally be avoided with GLP-1s due to overlapping effects on gastric motility.
- Extended titration: If standard 4-week intervals produce intolerable symptoms, extending each step to 6-8 weeks is preferable to discontinuation.
Special Considerations for Diabetic Patients
Switching GLP-1 agents in patients with type 2 diabetes carries additional considerations beyond weight management:
- Insulin adjustment: Patients on basal insulin may need temporary dose increases during the low-dose titration period of the new GLP-1, as the glycemic effect will be reduced until therapeutic doses are reached.
- Sulfonylurea risk: The combination of sulfonylureas with GLP-1 agonists carries hypoglycemia risk. During switches, when GLP-1 receptor activation is variable, close glucose monitoring is essential.
- HbA1c monitoring: Check HbA1c before the switch and 3 months after reaching the maintenance dose to assess impact on glycemic control.
- Cardiovascular considerations: For patients on semaglutide specifically for cardiovascular risk reduction (based on the SELECT trial), switching to an agent without equivalent CV outcome data (like tirzepatide) should be discussed carefully with the patient.
Non-GLP-1 Alternatives for Weight Management
When GLP-1 receptor agonists aren't accessible, affordable, or tolerated, several non-GLP-1 compounds and approaches offer alternative pathways for weight management. These range from FDA-approved medications to research-stage peptides with promising preclinical data.
Tesofensine: Triple Monoamine Reuptake Inhibitor
Tesofensine works through an entirely different mechanism than GLP-1 agonists. Originally developed as a treatment for Parkinson's disease and Alzheimer's disease, it inhibits the reuptake of three neurotransmitters - serotonin, norepinephrine, and dopamine - increasing their availability in the brain. This triple reuptake inhibition affects appetite regulation through central nervous system pathways rather than peripheral incretin signaling.
Phase 2 clinical trial data showed dose-dependent weight loss that was substantial for a non-GLP-1 compound:
- 0.25 mg: 4.5% mean body weight loss at 24 weeks
- 0.5 mg: 9.2% mean body weight loss at 24 weeks
- 1.0 mg: 10.6% mean body weight loss at 24 weeks
- Placebo: 2.0% (Astrup et al., 2008; DOI: 10.1016/S0140-6736(08)61525-1)
In the extension phase, patients continuing at 0.5 mg achieved total mean weight loss of 13-14 kg, suggesting sustained efficacy with continued treatment. Recent 2024 research demonstrated that tesofensine silences GABAergic hypothalamic neurons involved in feeding behavior, providing mechanistic insight into its appetite-suppressing effects (2024; DOI: 10.1073/pnas.2318841121).
Research Status
Tesofensine is not FDA-approved for obesity treatment. Its cardiovascular safety profile - specifically effects on heart rate and blood pressure - has been a concern. A 90-day trial combining tesofensine 0.5 mg with metoprolol 100 mg daily (to mitigate cardiac effects) showed 3% weight loss versus 1% for placebo in patients with obesity and type 2 diabetes. Ongoing research explores optimized dosing strategies to balance efficacy with cardiovascular safety.
5-Amino-1MQ: NNMT Inhibitor
5-Amino-1MQ (5-amino-1-methylquinolinium) targets a metabolic pathway distinct from both GLP-1 agonists and traditional appetite suppressants. It works by inhibiting nicotinamide N-methyltransferase (NNMT), an enzyme that plays a significant role in fat cell metabolism and energy expenditure.
The NNMT pathway is particularly interesting because this enzyme is upregulated in adipose tissue of obese individuals. By blocking NNMT, 5-amino-1MQ preserves NAD+ (nicotinamide adenine dinucleotide) - a critical coenzyme for cellular energy production and DNA repair. Higher NAD+ levels in fat cells support more efficient energy utilization and fat oxidation.
Preclinical data has been encouraging:
- Mouse studies showed 7% body mass reduction within 10 days without affecting food intake - suggesting the mechanism operates through metabolic enhancement rather than appetite suppression.
- Treatment of obese mice with potent NNMT inhibition reduced body weight and white adipose tissue mass, decreased adipocyte size, and lowered cholesterol levels.
- The compound appears to selectively affect fat metabolism without the muscle-wasting concerns associated with some weight loss interventions.
5-Amino-1MQ is not FDA-approved and lacks human clinical trial data for weight loss. Its use remains experimental, but the mechanistic rationale - targeting fat metabolism directly rather than appetite - makes it a complementary approach that could theoretically be used alongside GLP-1 therapy or as an alternative when GLP-1s aren't available.
AOD-9604: Growth Hormone Fragment
AOD-9604 is a synthetic peptide fragment corresponding to amino acids 177-191 of human growth hormone. It was designed to isolate the lipolytic (fat-burning) properties of growth hormone without the growth-promoting and diabetogenic effects.
Clinical trial results for AOD-9604 have been mixed:
- A 12-week trial showed 2.6 kg weight loss with AOD-9604 1 mg/day versus 0.8 kg with placebo.
- A 23-week trial showed 2.8 kg weight reduction versus 0.8 kg with placebo.
- However, a larger 24-week trial of 536 subjects failed to demonstrate significant weight loss, leading to development termination in 2007.
Despite the disappointing Phase 2b results, AOD-9604 retains interest in the research community for several reasons: it has an extensive safety database from over 900 clinical trial participants, it doesn't affect IGF-1 levels or glucose metabolism negatively (unlike full-length growth hormone), and it may have applications beyond weight loss, including joint and cartilage health.
FDA-Approved Non-GLP-1 Weight Loss Medications
Several FDA-approved medications outside the GLP-1 class are available for weight management and may serve as alternatives or adjuncts:
| Medication | Mechanism | Typical Weight Loss | Key Considerations |
|---|---|---|---|
| Phentermine-topiramate (Qsymia) | Sympathomimetic + anticonvulsant | ~7-10% | CV stimulant effects; teratogenic; DEA Schedule IV |
| Naltrexone-bupropion (Contrave) | Opioid antagonist + antidepressant | ~5-6% | Seizure risk; not for use with opioids; can improve cravings |
| Orlistat (Xenical/Alli) | Lipase inhibitor | ~3-5% | GI side effects (steatorrhea); OTC at lower dose (Alli) |
| Phentermine (Adipex-P) | Sympathomimetic | ~3-5% | Short-term use only (12 weeks); DEA Schedule IV; CV effects |
| Setmelanotide (Imcivree) | MC4R agonist | Variable | Only for specific genetic obesity disorders (POMC, PCSK1, LEPR deficiency) |
None of these alternatives match the magnitude of weight loss achieved with GLP-1 agonists, but they can be valuable for patients who cannot access or tolerate GLP-1 therapy. Combination approaches - such as phentermine-topiramate plus lifestyle intervention - can produce meaningful results, particularly when the goal is maintaining weight previously lost on a GLP-1 during a treatment gap.
Figure 6: Mechanism of action comparison across non-GLP-1 weight management alternatives, showing distinct pathways targeted by each compound.
Lifestyle Interventions as Bridge Therapy
During treatment gaps, evidence-based lifestyle interventions become critically important for maintaining progress. While they rarely match pharmacotherapy in magnitude of weight loss, they can significantly slow or prevent regain during periods without medication access.
Key components of an effective bridge strategy:
- Structured dietary approach: A protein-sparing modified fast (PSMF) or high-protein, moderate-calorie diet (1.2-1.6 g protein/kg/day) preserves lean mass during weight maintenance or modest regain periods (Westerterp-Plantenga et al., 2012; DOI: 10.3945/ajcn.112.037556).
- Resistance training: Maintaining or increasing resistance exercise during treatment gaps helps preserve metabolic rate and lean body mass - two factors that decline during weight loss and contribute to regain.
- Behavioral support: Structured follow-up with a dietitian or behavioral health specialist during gaps can help patients maintain the eating behavior changes that GLP-1 therapy facilitates.
- Sleep optimization: Sleep deprivation increases ghrelin and decreases leptin, amplifying hunger signals that GLP-1 therapy normally suppresses. Maintaining 7-9 hours of quality sleep becomes more important when pharmacologic appetite suppression is absent.
The Full Impact of the GLP-1 Shortage: Data, Demographics, and Downstream Effects
Beyond supply disruption statistics, the GLP-1 shortage created measurable consequences across clinical outcomes, healthcare system operations, and patient wellbeing. Quantifying these impacts helps justify the urgency of the prevention and mitigation strategies discussed throughout this guide.
Clinical Outcome Data During the Shortage
The discontinuation and reinitiation patterns during the shortage period provided a natural experiment in treatment interruption. A 2025 analysis published in JAMA Network Open examined patterns of GLP-1 discontinuation and reinitiation among U.S. adults with overweight or obesity, revealing several concerning trends (Crasto et al., 2025; DOI: 10.1001/jamanetworkopen.2024.54789).
The data showed that treatment interruptions were common and consequential. Patients who experienced gaps of more than 30 days had measurably worse outcomes upon reinitiation compared to those who maintained continuous therapy. Weight regain during gaps was rapid and substantial - consistent with the STEP 1 extension data showing two-thirds regain within a year (Wilding et al., 2022).
For diabetic patients specifically, the clinical consequences were more immediate. HbA1c levels began rising within 4-6 weeks of treatment discontinuation, with some patients seeing increases of 0.5-1.0% within three months. This glycemic deterioration often triggered compensatory medication changes - increased insulin doses, addition of sulfonylureas, or initiation of SGLT2 inhibitors - each carrying its own side effect profile and monitoring requirements.
The cardiovascular implications were also concerning. The SELECT trial had established semaglutide's cardiovascular benefit with a 20% reduction in major adverse cardiovascular events (MACE) in patients with overweight or obesity without diabetes (Lincoff et al., 2023; DOI: 10.1056/NEJMoa2307563). Treatment interruption meant patients lost this protective effect during the gap period. While the absolute risk increase from a few weeks without treatment was small, for high-risk patients, even brief interruptions carried meaningful implications.
Healthcare System Impacts
The shortage didn't just affect patients - it created substantial operational burden across the healthcare system:
Provider Workload
Endocrinologists and primary care providers reported significant increases in administrative workload during the shortage. Prior authorization calls, pharmacy hunting (contacting multiple pharmacies to locate stock), patient counseling about alternatives, and managing medication switching protocols consumed time that could have been spent on direct patient care.
A survey of obesity medicine specialists conducted during the peak shortage period found that practitioners were spending an average of 45-60 additional minutes per week per patient managing shortage-related issues. For practices with large GLP-1 patient panels, this represented a substantial operational burden.
Emergency Department Utilization
Reports from emergency departments indicated increased visits related to GLP-1 shortage complications:
- Diabetic ketoacidosis (DKA) or hyperglycemic hyperosmolar state (HHS) in patients who abruptly lost access to their GLP-1 medication without adequate alternative glycemic coverage
- Severe GI complications in patients who obtained compounded semaglutide with incorrect concentrations and inadvertently administered overdoses
- Mental health crises related to rapid weight regain and associated psychological distress
- Dehydration requiring IV fluids in patients who restarted GLP-1 therapy at too-high doses after gaps
Insurance System Friction
The shortage exposed and amplified existing friction in pharmaceutical insurance systems. Prior authorization denials increased as insurers struggled to update their formularies in response to rapidly changing availability. Step therapy requirements - mandating that patients try less expensive medications before accessing preferred agents - became nonsensical when the required first-step medications were also in short supply.
Appeals processes, already complex, became more challenging when clinical documentation needed to address both medical necessity and shortage-driven switching rationale. The administrative cost of processing these appeals - estimated at $50-$100 per appeal for both providers and insurers - added to the overall economic impact of the shortage.
Telehealth and Direct-to-Consumer Market Disruption
The GLP-1 shortage period coincided with explosive growth in telehealth-based weight loss services, creating a market that grew to an estimated $10-15 billion annually before the shortage resolution disrupted it dramatically.
Companies like Calibrate, Found, Ro, and Hims/Hers built their business models around prescribing GLP-1 medications - often compounded versions - through virtual visits. When compounding restrictions took effect after the shortage resolved, many of these platforms faced existential challenges:
- Patients who had been paying $150-$300/month for compounded semaglutide through telehealth platforms suddenly faced $299-$449/month for brand-name alternatives through manufacturer direct programs
- Some telehealth companies restructured to partner with brand-name manufacturers, while others pivoted to prescribing non-GLP-1 alternatives
- Patient retention became a significant challenge as cost increases drove discontinuation
- Regulatory scrutiny of telehealth prescribing practices for controlled and high-demand medications intensified
The net effect was a reshaping of the weight management telehealth landscape, with surviving companies generally transitioning toward more comprehensive care models that incorporate behavioral support, nutritional counseling, and exercise programming alongside pharmacotherapy - rather than operating primarily as medication access platforms.
Economic Impact Analysis
The total economic impact of the GLP-1 shortage extended well beyond medication costs:
| Impact Category | Estimated Annual Impact | Notes |
|---|---|---|
| Additional healthcare costs from treatment interruption | $3-5 billion | Worsened comorbidities, ER visits, alternative medications |
| Administrative burden (prior auth, appeals) | $500 million-$1 billion | Provider and payer staff time |
| Lost productivity (patient time spent managing shortage) | $1-2 billion | Phone calls, pharmacy visits, provider appointments |
| Compounding market revenue (during shortage) | $3-5 billion | Now largely displaced by brand-name sales |
| Telehealth market disruption | $2-4 billion | Revenue loss from compounding restrictions |
| Manufacturer expansion investment | $10+ billion (cumulative) | Novo Nordisk and Eli Lilly capex |
These estimates, while rough, illustrate the enormous scale of downstream consequences from a supply disruption in a single drug class. The shortage served as a case study in pharmaceutical supply chain fragility and has informed policy discussions about manufacturing redundancy, supply chain transparency, and shortage prevention strategies.

Figure 9: Estimated economic impact of the GLP-1 drug shortage across healthcare, administrative, and market disruption categories.
Disparate Impact Across Patient Populations
The shortage didn't affect all populations equally. Several groups bore disproportionate burden:
Racial and Ethnic Minorities
Patients from racial and ethnic minority communities - who face higher rates of type 2 diabetes and obesity - were disproportionately affected by the shortage. These populations are also more likely to lack comprehensive insurance coverage, making brand-name medications less accessible when compounded alternatives disappeared. Pre-existing disparities in healthcare access, provider relationships, and pharmaceutical coverage amplified the shortage's impact.
Rural Populations
Patients in rural areas had fewer pharmacy options when local pharmacies ran out of stock. The pharmacy consolidation trend - with rural pharmacies closing at increasing rates - meant that some patients had to travel considerable distances to find available supply. Mail-order pharmacy options partially addressed this, but shipping delays and cold-chain concerns added complexity.
Low-Income Patients
The cost differential between compounded ($100-$300/month) and brand-name ($349-$1,000+/month without assistance) GLP-1 medications was most acutely felt by low-income patients. While manufacturer assistance programs exist, awareness of and access to these programs varies, and the application process itself can be a barrier for patients with limited time, internet access, or health literacy.
Patients with Type 2 Diabetes vs. Weight Management
During the shortage, clinical guidance generally prioritized supply for patients with type 2 diabetes over those using GLP-1s for weight management alone. While this prioritization was medically reasonable - glycemic decompensation carries more immediate health risks than weight regain - it created a two-tier access system that left many weight management patients without medication despite legitimate medical need.
This prioritization framework also exposed philosophical tensions about how the medical system values obesity treatment. The distinction between "treating diabetes" (widely accepted as medical necessity) and "managing weight" (sometimes dismissed as cosmetic or elective) affected insurance coverage, provider prescribing behavior, and ultimately patient access during constrained supply periods.
Compounding Safety: Detailed Analysis of Risks and Quality Indicators
The rapid expansion and subsequent contraction of the compounded GLP-1 market generated substantial data on safety outcomes, quality variations, and regulatory gaps that carry lessons for patients, providers, and policymakers alike.
Adverse Event Analysis
The FDA's MedWatch system captured 605 adverse event reports associated with compounded semaglutide and 545 for compounded tirzepatide as of July 2025. Given the well-documented underreporting of adverse events to FDA passive surveillance systems - estimated at capturing only 1-10% of actual events - the true number of adverse events was likely in the thousands to tens of thousands.
The reported events fell into several categories:
Dosing Errors (Most Frequent)
The single most common category of adverse events involved dosing errors. Unlike brand-name Ozempic and Wegovy pens, which use dial mechanisms that deliver precise predetermined doses, compounded semaglutide typically came in multi-dose vials requiring patients to measure doses using insulin syringes.
The FDA identified cases where patients received 5 to 20 times the intended dose. These overdoses resulted from:
- Confusion between different syringe sizes (0.3 mL, 0.5 mL, 1.0 mL syringes have different unit markings)
- Misunderstanding of concentration versus dose (a vial containing 5 mg/mL semaglutide requires different volumes than a vial containing 2.5 mg/mL)
- Inadequate patient education on measurement technique
- Labeling ambiguities on compounded products (some labels specified dose in mg while others used units or volume)
- Mathematical errors in calculating the correct volume to draw up
Symptoms of semaglutide overdose include severe, prolonged nausea and vomiting (lasting days rather than hours), dangerous dehydration, hypoglycemia (particularly in diabetic patients or those on concurrent glucose-lowering medications), and in severe cases, acute kidney injury from dehydration-induced renal hypoperfusion.
Sterility Failures
FDA inspections of compounding facilities revealed concerning sterility issues at several operations. The case of Fullerton Wellness was particularly notable - the FDA raised concerns about the sterility of semaglutide and tirzepatide injections distributed by this facility, noting that non-sterile injectable products can cause life-threatening infections and sepsis.
Sterility risks in compounded injectables arise from multiple points in the preparation process:
- Inadequate cleanroom conditions during preparation
- Insufficient sterilization of vials, stoppers, and equipment
- Contamination during the filling process
- Breakdown in aseptic technique by compounding personnel
- Inadequate end-product sterility testing (some 503A pharmacies perform limited or no sterility testing)
The historical precedent for compounding sterility failures is sobering. The 2012 New England Compounding Center (NECC) fungal meningitis outbreak, caused by contaminated methylprednisolone injections, killed 76 people and sickened 778 across 20 states. This tragedy directly led to the creation of the 503B outsourcing facility framework under the Drug Quality and Security Act of 2013.
Potency Variations
Independent testing of compounded semaglutide products during the shortage period revealed significant potency variations between batches and between different compounding pharmacies:
- Some products contained less active ingredient than labeled, resulting in sub-therapeutic dosing and inadequate clinical response
- Other products contained more active ingredient than labeled, increasing the risk of adverse effects
- Stability variations meant that some products degraded more rapidly than their labeled beyond-use dates suggested
- The use of different salt forms (semaglutide sodium, semaglutide acetate) introduced additional potency uncertainty, as the molecular weight differences between salt forms affect the actual amount of active semaglutide delivered per milligram of salt
Fraudulent Products
Perhaps most alarming, the FDA identified outright fraudulent compounded semaglutide and tirzepatide products circulating in the U.S. market. These products bore labels from compounding pharmacies that didn't exist. The contents of these fraudulent vials were unknown - they could have contained the correct medication at the wrong concentration, a different medication entirely, or potentially harmful contaminants.
The emergence of fraudulent products was driven by the enormous market demand and the relative ease of creating convincing pharmaceutical labeling. Unlike brand-name medications with sophisticated anti-counterfeiting measures (holographic seals, serialized packaging, tamper-evident closures), compounded medication vials are relatively simple to counterfeit.
How Patients Can Verify Compounded Product Quality
For the limited situations where compounded GLP-1 medications remain legally available, patients should take an active role in verifying product quality:
- Verify pharmacy licensure: Check your state board of pharmacy website to confirm the pharmacy is licensed and in good standing. Look for any disciplinary actions or consent orders.
- Check 503B registration: If the pharmacy claims to be a 503B outsourcing facility, verify on the FDA's registered outsourcing facilities list.
- Request the Certificate of Analysis (COA): Legitimate compounding pharmacies should provide COAs showing potency testing, sterility testing, endotoxin testing, and pH measurement for each batch.
- Verify the API source: Ask where the pharmacy sources its semaglutide active pharmaceutical ingredient. Reputable pharmacies use API from suppliers with Drug Master Files (DMFs) registered with the FDA.
- Check for accreditation: PCAB (Pharmacy Compounding Accreditation Board) accreditation or USP <800> compliance indicates adherence to quality standards above the minimum regulatory requirements.
- Inspect the product: The solution should be clear and colorless. Any cloudiness, particulate matter, or discoloration is a red flag. Check that the vial is properly sealed with an intact cap and crimped aluminum seal.
- Review the label: The label should include the pharmacy name, address, phone number, license number, drug name and concentration, beyond-use date, storage conditions, and lot number.
The Argument for Continued Compounding Access
Despite the safety concerns, legitimate arguments exist for maintaining some level of compounding access for GLP-1 medications:
- Cost access: At $100-$300/month versus $299-$1,000+/month for brand-name products, compounded versions made GLP-1 therapy accessible to patients who otherwise couldn't afford it. The health consequences of untreated obesity - cardiovascular disease, type 2 diabetes, certain cancers, sleep apnea - carry their own costs and risks that should be weighed against compounding risks.
- Clinical flexibility: Compounding allows for dose customization that brand-name products don't offer. Some patients benefit from non-standard doses, combination preparations, or alternative formulations (such as sublingual or different concentrations).
- Patient allergies: Genuine allergies to inactive ingredients in brand-name products (excipients, preservatives, device materials) require compounded alternatives.
- Supply chain resilience: The shortage itself demonstrated that compounding pharmacies provided a critical safety net when brand-name supply failed. Eliminating all compounding capability removes this safety net entirely.
These arguments don't negate the safety concerns - they complicate the policy calculus. The ideal framework would maintain compounding access under stringent quality controls while ensuring that compounded products meet rigorous sterility, potency, and purity standards. Whether the current regulatory framework achieves this balance remains a matter of active debate among pharmacists, physicians, regulators, and patient advocates.
Clinical Pharmacology of GLP-1 Switching: A Provider Reference
For healthcare providers managing patients through GLP-1 transitions, a deeper understanding of the comparative pharmacology of these agents informs more nuanced clinical decision-making. This section provides the pharmacokinetic and pharmacodynamic context underlying the switching protocols described earlier.
Receptor Binding and Activation Profiles
The GLP-1 receptor agonists currently available differ in their receptor binding characteristics, which has direct implications for switching:
| Agent | GLP-1R Affinity | GIP-R Affinity | Glucagon-R Affinity | Half-Life | Bioavailability (SC) |
|---|---|---|---|---|---|
| Semaglutide | High (modified GLP-1) | None | None | ~168 hours (7 days) | ~89% |
| Tirzepatide | Moderate | High | None | ~120 hours (5 days) | ~80% |
| Liraglutide | High (native-like) | None | None | ~13 hours | ~55% |
| Dulaglutide | Moderate (Fc fusion) | None | None | ~120 hours (5 days) | ~47-65% |
| Retatrutide | Moderate | High | Moderate | ~6 days | Data pending |
These differences explain why simple dose conversion between agents is pharmacologically invalid. Tirzepatide's clinical superiority over semaglutide in weight loss doesn't arise from greater GLP-1 receptor activation - in fact, tirzepatide has lower GLP-1R affinity than semaglutide. Instead, the additional GIP receptor activation creates metabolic effects through distinct pathways: enhanced insulinotropic response, direct effects on adipose tissue, and central appetite regulation through GIP-R-expressing hypothalamic neurons.
This mechanistic distinction means that a patient who has achieved receptor tolerance to GLP-1 stimulation through semaglutide therapy hasn't developed tolerance to GIP receptor stimulation. When switching to tirzepatide, the GIP component represents a novel pharmacologic input, explaining both the potential for enhanced efficacy and the possibility of unexpected side effects during transition.
Pharmacokinetic Overlap During Switching
Understanding the pharmacokinetic profiles of each agent is essential for timing switches to minimize both gaps in coverage and excessive receptor stimulation from overlapping drug concentrations:
Semaglutide Washout
Semaglutide's half-life of approximately 7 days means that after the last dose:
- Day 7 (1 half-life): ~50% of steady-state concentration remains
- Day 14 (2 half-lives): ~25% remains
- Day 21 (3 half-lives): ~12.5% remains
- Day 35 (5 half-lives): ~3% remains (generally considered fully cleared)
This means that when a patient takes their last semaglutide dose on Day 0 and starts tirzepatide on Day 7 (the recommended approach), there's meaningful semaglutide still in circulation. The overlapping GLP-1R activation from residual semaglutide plus new tirzepatide explains why some patients experience more pronounced GI effects during the first 1-2 weeks of the switch than they would if starting tirzepatide de novo.
Tirzepatide Washout
Tirzepatide's half-life of approximately 5 days means a somewhat faster washout:
- Day 5 (1 half-life): ~50% remains
- Day 10 (2 half-lives): ~25% remains
- Day 25 (5 half-lives): ~3% remains
When switching from tirzepatide to semaglutide, the 7-day interval between the last tirzepatide dose and first semaglutide dose represents approximately 1.4 half-lives of tirzepatide clearance, meaning roughly 38% of the tirzepatide concentration remains at the time of the first semaglutide injection.
Liraglutide Washout
Liraglutide's much shorter half-life (~13 hours) means rapid clearance:
- 13 hours (1 half-life): ~50% remains
- 26 hours (2 half-lives): ~25% remains
- 65 hours (~3 days, 5 half-lives): ~3% remains
This rapid clearance is both an advantage and disadvantage in switching scenarios. Patients switching from liraglutide to a weekly agent can start the new medication 1-2 days after the last liraglutide dose with minimal overlap concerns. However, the rapid drop in GLP-1R activation means patients may experience a more abrupt return of appetite between the last liraglutide dose and the onset of the new agent's effect.
Practical Provider Decision Framework
When deciding which alternative GLP-1 to switch to, providers should consider multiple factors beyond just efficacy data:
Switching Decision Matrix
- Patient wants greater weight loss: Switch to tirzepatide (dual agonist mechanism provides additional efficacy)
- Patient needs lower cost: Consider generic liraglutide ($200-500/month) or oral semaglutide with insurance coverage
- Patient can't tolerate weekly GI peaks: Switch to daily liraglutide (smoother pharmacokinetic profile with less peak-to-trough fluctuation)
- Patient has significant CV risk: Prioritize agents with CV outcome data - semaglutide (SELECT) or liraglutide (LEADER)
- Patient is needle-averse: Consider oral semaglutide 25 mg or upcoming oral non-peptide options
- Patient needs diabetes indication specifically: Ozempic, Mounjaro, Trulicity, or Victoza (rather than Wegovy/Zepbound/Saxenda which are weight management-labeled)
- Patient plateaued on single agonist: Switch to dual (tirzepatide) or triple (retatrutide, when available) agonist for additional receptor pathways
Monitoring Parameters During and After Switching
A systematic monitoring plan ensures safe transitions and early identification of problems:
| Parameter | Baseline (Before Switch) | During Titration | Maintenance (After Target Dose Reached) |
|---|---|---|---|
| Weight | Record on day of switch | Weekly | Monthly |
| Blood glucose (if diabetic) | Daily or CGM | Daily or CGM for first 4-6 weeks | Per standard diabetes management |
| HbA1c (if diabetic) | Within 4 weeks pre-switch | - | 3 months after reaching target dose |
| Blood pressure | Record on day of switch | At each dose escalation visit | Quarterly |
| GI symptoms | Baseline symptom assessment | Weekly patient-reported assessment | At each follow-up visit |
| Renal function (BMP) | Within 30 days pre-switch | If significant GI symptoms or dehydration | Annually or as indicated |
| Lipid panel | Recent baseline | - | 3-6 months after reaching target dose |
| Thyroid function | Baseline if not recent | - | Annually (GLP-1 class labeling includes thyroid C-cell tumor warning) |
| Mental health screening | Brief assessment | Check for mood changes at each visit | Periodic screening per guidelines |
Documentation Requirements for Insurance Purposes
Thorough documentation of the switching rationale is essential for insurance coverage continuity. A complete switching note should include:
- Medical necessity statement: Why the patient requires GLP-1 therapy (BMI, comorbidities, prior treatment history)
- Reason for switch: Supply issues, insurance formulary change, inadequate response, adverse effects, or cost
- Prior agent documentation: What the patient was taking, at what dose, for how long, and with what response
- New agent selection rationale: Why this particular alternative was chosen over others
- Titration plan: The planned dose escalation schedule with monitoring checkpoints
- Contingency plan: What to do if the new agent isn't tolerated or effective
This documentation serves multiple purposes: clinical record-keeping, prior authorization support, appeals documentation if coverage is denied, and continuity of care if the patient transfers to a different provider.
For patients getting started with GLP-1 therapy or transitioning between agents, working with a knowledgeable provider who understands both the pharmacology and the administrative landscape is essential. The compounding pharmacy peptides guide provides additional context on the broader compounding landscape beyond GLP-1 medications.
Figure 10: Provider decision matrix for GLP-1 switching, incorporating efficacy, safety, cost, and patient preference factors.
Comorbidity-Specific GLP-1 Management During Supply Disruptions
Patients on GLP-1 medications often have multiple comorbidities that complicate treatment decisions during shortages and transitions. This section provides targeted guidance for the most common clinical scenarios where GLP-1 access issues intersect with comorbidity management.
Type 2 Diabetes With Cardiovascular Disease
Patients with both type 2 diabetes and established cardiovascular disease represent the highest-priority group for uninterrupted GLP-1 therapy. These patients derive dual benefit - glycemic control and cardiovascular risk reduction - making treatment gaps potentially consequential from both perspectives.
The SELECT trial demonstrated that semaglutide reduced major adverse cardiovascular events (MACE) by 20% in patients with overweight or obesity without diabetes (Lincoff et al., 2023). The LEADER trial showed liraglutide reduced MACE by 13% in patients with type 2 diabetes at high cardiovascular risk (Marso et al., 2016; DOI: 10.1056/NEJMoa1603827). The REWIND trial demonstrated dulaglutide's 12% MACE reduction (Gerstein et al., 2019).
For these patients, the switching hierarchy should prioritize agents with proven CV benefit:
- First choice: Maintain current GLP-1 if possible (even at lower dose during supply constraints)
- Second choice: Switch to another GLP-1 with CV outcome data (semaglutide to liraglutide, or vice versa)
- Third choice: Dulaglutide (REWIND data, though less commonly used now)
- Fourth choice: Tirzepatide (strong metabolic benefits but dedicated CV outcome trial data still pending at time of writing)
During treatment gaps, these patients require intensified cardiovascular monitoring. Blood pressure checks should increase from quarterly to monthly. Lipid panels should be obtained at the start and end of any treatment gap longer than 4 weeks. Statin therapy should be optimized independently of GLP-1 status. Antiplatelet therapy should be reviewed and maintained per guidelines.
Obstructive Sleep Apnea
Obesity is the strongest modifiable risk factor for obstructive sleep apnea (OSA), and weight loss with GLP-1 agonists has demonstrated meaningful improvements in apnea-hypopnea index (AHI) scores. The STEP 2 trial showed that semaglutide 2.4 mg reduced AHI scores significantly in patients with obesity and moderate-to-severe OSA.
During treatment gaps, patients with OSA should be counseled about the possibility of worsening sleep-disordered breathing as weight rebounds. Specific considerations include:
- Maintain CPAP/BiPAP compliance during treatment gaps, even if previously able to reduce pressure settings during weight loss
- Consider retitration of positive airway pressure settings if significant weight regain occurs (>5% of body weight)
- Monitor daytime sleepiness symptoms (Epworth Sleepiness Scale) as an early indicator of worsening OSA
- Positional therapy (sleeping on side rather than back) becomes more important as weight increases
- Alcohol and sedative medication avoidance is particularly important during weight regain, as these substances worsen OSA independently
Non-Alcoholic Fatty Liver Disease (NAFLD/MASH)
GLP-1 agonists have shown promising effects on hepatic steatosis and inflammation. Semaglutide's Phase 2 NASH trial demonstrated histological improvement in MASH (formerly NASH) without worsening fibrosis. Weight loss of any magnitude improves hepatic fat content, with greater weight loss producing greater benefit.
For patients with NAFLD/MASH experiencing GLP-1 treatment gaps:
- Hepatic fat reaccumulation: Hepatic steatosis can worsen rapidly with weight regain, often more quickly than subcutaneous fat increases. Dietary fat and fructose intake are particularly important modifiers during treatment gaps.
- Liver enzymes: Monitor ALT and AST during gaps longer than 4 weeks. Rising transaminases may indicate hepatic fat reaccumulation and inflammation.
- Alternative agents with hepatic benefit: Pioglitazone has evidence for MASH improvement and could serve as bridge therapy. Vitamin E (800 IU/day) has shown benefit in non-diabetic MASH patients.
- Consider retatrutide: When available, the triple agonist's glucagon receptor activation provides additional hepatic fat mobilization beyond what GLP-1 agonism alone achieves. Phase 2 data showed dramatic liver fat reduction.
Polycystic Ovary Syndrome (PCOS)
GLP-1 agonists have become increasingly used in PCOS management, where their effects on weight loss, insulin resistance, and potentially direct ovarian effects can improve multiple aspects of the syndrome. Treatment gaps raise specific concerns:
- Insulin resistance: The improvement in insulin sensitivity achieved with GLP-1 therapy reverses during treatment gaps, potentially worsening hyperandrogenism and anovulation.
- Fertility implications: Weight loss can restore ovulatory function in women with PCOS. Weight regain during treatment gaps may cause recurrence of anovulation. Patients actively trying to conceive should be aware that fertility may fluctuate during treatment transitions.
- Contraception awareness: GLP-1 discontinuation before conception is recommended (2 months before for semaglutide), but the improved fertility from weight loss means that pregnancies can occur earlier than planned. Ensure appropriate contraception during the GLP-1 treatment period if pregnancy isn't desired.
- Androgen monitoring: Total and free testosterone, DHEA-S, and SHBG levels may shift during treatment gaps. If these values are being tracked for PCOS management, additional monitoring during gaps helps detect early hormonal deterioration.
Chronic Kidney Disease
Emerging evidence suggests GLP-1 agonists have renoprotective effects independent of glucose lowering. The FLOW trial demonstrated that semaglutide reduced the risk of clinically important kidney events in patients with type 2 diabetes and chronic kidney disease (CKD).
For CKD patients during GLP-1 treatment gaps:
- Dehydration risk amplified: CKD patients have reduced renal compensatory capacity. If weight regain increases metabolic demand while GI side effects from medication restarts cause fluid losses, acute kidney injury risk is elevated.
- Blood pressure management: Weight regain often worsens hypertension. In CKD patients, blood pressure control is paramount for preserving remaining kidney function. Consider intensifying antihypertensive therapy during gaps.
- eGFR monitoring: Check serum creatinine and calculate eGFR at the start and end of treatment gaps. Report any significant decline to the nephrology team.
- SGLT2 inhibitors as bridge: For CKD patients who also have type 2 diabetes or heart failure, SGLT2 inhibitors (empagliflozin, dapagliflozin) provide renoprotective and cardiometabolic benefits that can partially compensate for GLP-1 absence.
Psychiatric Conditions and Eating Disorders
Patients with comorbid psychiatric conditions, particularly those involving disordered eating, require specialized attention during GLP-1 treatment transitions:
Depression and Anxiety
Weight regain during treatment gaps can trigger or exacerbate depressive episodes, particularly in patients whose self-esteem and emotional wellbeing are closely tied to weight. Proactive mental health screening (PHQ-9, GAD-7) should be conducted at each contact during treatment gaps. Some psychiatric medications (SSRIs, atypical antipsychotics) can promote weight gain, compounding regain during GLP-1 absence.
Binge Eating Disorder
GLP-1 agonists reduce binge eating frequency through appetite suppression and central reward pathway modulation. When GLP-1 therapy is interrupted, binge eating episodes may resume or intensify. Patients with BED should have behavioral health support intensified during treatment gaps, and consider lisdexamfetamine (Vyvanse, FDA-approved for BED) as bridge therapy if binge eating recurs.
History of Restrictive Eating
Patients with a history of anorexia nervosa or restrictive eating patterns may respond to GLP-1-related appetite suppression in maladaptive ways. Treatment gaps - and the return of normal appetite signals - may paradoxically be distressing for these patients, who may interpret renewed hunger as loss of control. Close monitoring by providers familiar with eating disorder dynamics is essential.
Joint Disease and Mobility Limitations
Patients with osteoarthritis, particularly weight-bearing joint disease, derive significant benefit from the weight reduction achieved with GLP-1 therapy. Every pound of weight lost translates to approximately four pounds of reduced knee joint loading during walking.
During treatment gaps:
- Escalating joint pain from weight regain may limit physical activity, creating a feedback loop that accelerates further weight gain
- Physical therapy referral to maintain mobility and joint function is valuable during treatment interruptions
- Low-impact exercise (swimming, cycling, elliptical) helps maintain weight and joint function without the impact stress of walking or running
- AOD-9604 has been studied for both weight management and joint/cartilage health, potentially addressing both concerns simultaneously for some patients
- If orthopedic surgery is being considered, coordinate the timing of GLP-1 therapy with the surgical team, as these medications should be discontinued before procedures due to aspiration risk from delayed gastric emptying
GLP-1 Receptor Agonist Pharmacology: A Detailed Comparison
Each GLP-1 receptor agonist has a unique structural modification that determines its pharmacokinetic profile, receptor binding characteristics, and clinical properties. Understanding these structural differences provides the scientific foundation for switching decisions.
Structural Modifications and Half-Life Extension
Native GLP-1 has a half-life of approximately 2 minutes - far too short for therapeutic use. Each approved GLP-1 agonist uses a different structural strategy to extend this half-life:
Semaglutide: Fatty Acid Acylation + Amino Acid Substitutions
Semaglutide shares 94% structural homology with native GLP-1 but incorporates two key modifications: an amino acid substitution at position 8 (alanine to alpha-aminobutyric acid, protecting against DPP-4 degradation) and a C-18 fatty acid chain attached via a linker at position 26. The fatty acid chain binds to albumin in the bloodstream, creating a circulating reservoir that slowly releases active semaglutide. This strategy produces a half-life of approximately 168 hours (7 days), enabling weekly dosing.
The high albumin binding affinity (~99.9%) means that only a small fraction of circulating semaglutide is free (unbound) at any time. This creates a very stable plasma concentration profile with minimal peak-to-trough fluctuation - contributing to semaglutide's relatively consistent appetite suppression throughout the dosing interval.
Tirzepatide: Dual Agonist with Fatty Acid Acylation
Tirzepatide is a 39-amino-acid peptide based on the native GIP sequence but engineered to also activate the GLP-1 receptor. It incorporates a C-20 fatty acid chain that binds albumin, producing a half-life of approximately 120 hours (5 days). Despite the shorter half-life compared to semaglutide, weekly dosing provides adequate coverage.
The dual agonist design creates a unique pharmacologic profile. Tirzepatide's GIP receptor affinity is approximately equal to native GIP, while its GLP-1 receptor affinity is roughly five-fold lower than native GLP-1. This imbalanced agonism - strong GIP activation with moderate GLP-1 activation - appears to be therapeutically advantageous, producing greater weight loss than either receptor agonism alone would predict.
Liraglutide: Fatty Acid Acylation (First Generation)
Liraglutide was the first GLP-1 agonist to use fatty acid acylation for half-life extension. It has 97% homology to native GLP-1 with a single amino acid substitution (lysine to arginine at position 34) and a C-16 palmitic acid chain attached at position 26. The shorter fatty acid chain produces less albumin binding affinity than semaglutide, resulting in a half-life of approximately 13 hours - long enough for once-daily dosing but not weekly.
This shorter half-life creates a more pronounced peak-to-trough pharmacokinetic profile than weekly agents. Clinically, this means that the appetite-suppressive effect of liraglutide may wax and wane somewhat throughout the day, with peak effect approximately 8-12 hours after injection. Some patients find this profile less consistent than weekly agents, while others prefer the ability to "feel" when the medication is most active.
Dulaglutide: Fc Fusion Protein
Dulaglutide takes a fundamentally different approach to half-life extension. Rather than using a fatty acid chain, it fuses a modified GLP-1 analog to the Fc (constant) fragment of human IgG4 immunoglobulin. The large Fc fragment slows renal clearance and extends the half-life to approximately 120 hours (5 days), comparable to tirzepatide.
The Fc fusion approach creates a much larger molecular weight (~63 kDa compared to ~4.1 kDa for semaglutide), which affects distribution and receptor interaction kinetics. The larger molecule may have somewhat different CNS penetration characteristics, potentially contributing to dulaglutide's relatively modest weight loss effects compared to semaglutide despite similar GLP-1 receptor activation.
GIP Receptor Biology and Tirzepatide's Dual Mechanism
Understanding GIP (glucose-dependent insulinotropic polypeptide) receptor biology helps explain why tirzepatide outperforms pure GLP-1 agonists and why switching to tirzepatide can produce additional weight loss in patients who've plateaued on semaglutide.
GIP receptor activation produces several effects complementary to GLP-1 agonism:
- Central appetite regulation: GIPR is expressed in hypothalamic neurons involved in appetite regulation. GIP signaling modulates feeding behavior through pathways partially distinct from those activated by GLP-1.
- Adipose tissue effects: GIPR is expressed on adipocytes. GIP signaling in fat tissue affects lipid storage, adipogenesis, and adipocyte insulin sensitivity through mechanisms that GLP-1 doesn't directly influence.
- Enhanced insulinotropic effect: GIP and GLP-1 have complementary effects on pancreatic beta-cell insulin secretion. The combination produces greater glucose-stimulated insulin release than either alone.
- Bone metabolism: GIP has anabolic effects on bone that may partially counteract the bone density reductions sometimes observed with significant weight loss. This is a potential advantage of tirzepatide over pure GLP-1 agonists during major weight loss.
For patients switching from semaglutide to tirzepatide, the implication is clear: the GIP receptor pathway represents an entirely new therapeutic mechanism that the patient hasn't been exposed to. The additional weight loss commonly observed when switching from semaglutide to tirzepatide likely reflects this recruitment of a second receptor system rather than simply greater GLP-1 receptor stimulation.
Glucagon Receptor Agonism: The Next Frontier
Looking ahead, the addition of glucagon receptor agonism in agents like retatrutide introduces a third pharmacologic dimension that further differentiates these compounds from current options.
Glucagon receptor activation contributes to weight loss through:
- Increased energy expenditure: Glucagon stimulates hepatic glucose output and thermogenesis, increasing basal metabolic rate. This addresses one of the main challenges of weight loss - the compensatory decrease in energy expenditure that occurs as body weight drops.
- Hepatic fat mobilization: Glucagon stimulates hepatic lipid oxidation, directly reducing hepatic steatosis. This effect was particularly pronounced in retatrutide Phase 2 data, where liver fat reductions exceeded those seen with GLP-1 agonism alone.
- Amino acid metabolism: Glucagon signaling affects hepatic amino acid catabolism, which may influence whole-body protein metabolism and the ratio of lean mass to fat mass lost during weight reduction.
The trade-off is that glucagon receptor activation also promotes hepatic glucose output, which could worsen glycemic control in diabetic patients. Retatrutide's design balances this concern through the concurrent GLP-1 and GIP receptor activation, which enhance insulin secretion to counteract the glucagon-driven glucose rise. In Phase 2 trials, retatrutide improved glycemic parameters despite the glucagon component, suggesting successful pharmacologic balancing.
For a complete analysis of the GLP-1 class of medications and their comparative pharmacology, our dedicated research report provides additional depth on receptor biology, binding kinetics, and clinical implications.
Telehealth, Digital Health Tools, and Integrated Care During GLP-1 Transitions
The GLP-1 shortage era accelerated the adoption of telehealth and digital health tools for obesity management. Understanding how these platforms work, what they offer, and their limitations helps patients make informed decisions about care delivery - especially during medication transitions.
The Telehealth GLP-1 Landscape: What Changed
Before the shortage, GLP-1 prescribing was dominated by traditional in-office endocrinology and primary care visits. The shortage, combined with expanded telehealth regulations during the COVID-19 public health emergency, catalyzed a massive shift toward virtual prescribing platforms.
At the peak, dozens of telehealth companies offered GLP-1 prescriptions, often bundled with compounded semaglutide supply. The business model was straightforward: virtual consultation, prescription, and medication shipped to the patient's door - typically at a bundled monthly cost of $200-$500 that included both the visit and the compounded medication.
The post-shortage environment has reshaped this landscape dramatically. Companies that built their models exclusively around compounded GLP-1 access faced existential challenges when compounding restrictions took effect. The survivors have generally evolved in one of three directions:
- Brand-name partnership model: Companies like Ro and Hims/Hers transitioned to prescribing FDA-approved brand-name medications, partnering with manufacturers or specialty pharmacies for fulfillment. Monthly costs increased to $299-$599 depending on the medication and dose.
- Comprehensive care model: Some platforms expanded beyond medication prescribing to offer integrated weight management programs combining pharmacotherapy with behavioral health, nutritional counseling, exercise programming, and ongoing clinical monitoring. This higher-touch approach commands premium pricing but may produce more sustainable outcomes.
- Alternative medication focus: A subset of telehealth companies pivoted to prescribing non-GLP-1 weight loss medications (phentermine-topiramate, naltrexone-bupropion, metformin for off-label weight management) or research compounds, creating a lower-cost tier of service.
Evaluating Telehealth GLP-1 Providers
Not all telehealth providers offer equivalent quality of care. Patients considering telehealth for GLP-1 management should evaluate several factors:
Clinical Quality Indicators
- Provider credentials: Who writes the prescription? Is it a board-certified physician, a nurse practitioner, or a physician assistant? What is their specialty training? Providers with board certification in obesity medicine (ABOM), endocrinology, or internal medicine generally offer more nuanced GLP-1 management.
- Follow-up structure: Does the platform require regular follow-up visits for dose titration and monitoring, or is it primarily a one-time prescription service? Ongoing monitoring is essential for safe GLP-1 use, particularly during dose escalation and medication switches.
- Lab monitoring: Does the platform require or facilitate appropriate laboratory monitoring (HbA1c for diabetic patients, metabolic panel for kidney function, lipid panels)? GLP-1 therapy without appropriate monitoring is substandard care.
- Contraindication screening: Does the intake process thoroughly screen for contraindications (personal or family history of medullary thyroid carcinoma, MEN2 syndrome, pancreatitis history, gastroparesis)?
- Medication management: Can the platform manage medication interactions, dose adjustments, and switching between agents? Or does it offer a single medication at fixed dosing?
Business Practice Red Flags
- Guaranteed prescription: Any platform that guarantees a GLP-1 prescription before clinical evaluation is prioritizing revenue over patient safety. Appropriate prescribing requires medical evaluation and may result in a determination that GLP-1 therapy isn't appropriate for a specific patient.
- No mention of side effects or risks: Marketing that emphasizes only benefits without discussing GI side effects, contraindications, and monitoring requirements is misleading.
- Compounded medication claims post-shortage: Platforms still prominently offering compounded semaglutide without clear discussion of the legal restrictions may be operating in a regulatory gray area.
- Pressure to purchase immediately: High-pressure sales tactics, countdown timers, or "limited availability" messaging should raise concerns about the platform's priorities.
- No prescriber-patient relationship: If you can obtain medication without a meaningful clinical interaction - a real conversation with a licensed provider who asks questions, reviews your history, and makes an individualized assessment - the care quality is likely insufficient.
Digital Health Tools for Treatment Gap Management
Several categories of digital health tools can support patients during GLP-1 treatment transitions and gaps:
Continuous Glucose Monitors (CGMs) for Non-Diabetic Patients
CGMs like Dexcom and Abbott FreeStyle Libre have become popular among GLP-1 users for real-time glucose monitoring, even in non-diabetic patients. During treatment gaps, CGM data can provide early warning of glycemic deterioration and help patients understand how dietary choices affect blood glucose without pharmacotherapy support.
While CGM use in non-diabetic patients remains somewhat controversial, the behavioral feedback it provides - seeing real-time glucose spikes after meals - can reinforce dietary adherence during periods without GLP-1 appetite suppression. The cost ($75-$300/month without insurance) and insurance coverage limitations are important considerations.
Smart Scales and Body Composition Monitors
Connected scales that track weight trends over time (rather than single data points) can help patients and providers detect early weight regain during treatment gaps. Apps that display weight as a trend line rather than individual measurements reduce the emotional impact of daily fluctuations while providing clinically useful trajectory data.
Body composition monitors that estimate body fat percentage and lean mass can provide more nuanced tracking than weight alone. During treatment gaps, patients who maintain or increase resistance training may see weight stabilize while body composition shifts toward more lean mass and less fat - a clinically favorable change that simple weight tracking would miss.
Food Tracking and Meal Planning Apps
During GLP-1 therapy, appetite suppression makes caloric restriction feel effortless for many patients. When medication is interrupted, deliberate food tracking becomes a valuable compensatory strategy. Apps like MyFitnessPal, Lose It!, and Cronometer can help patients maintain awareness of caloric intake as appetite returns to pre-treatment levels.
The key is establishing tracking habits while still on medication, when appetite is suppressed and the cognitive burden of tracking is lower. Patients who develop tracking skills during treatment are better equipped to maintain them during gaps, compared to those who only start tracking after medication is interrupted and appetite has already rebounded.
Remote Patient Monitoring Platforms
Integrated remote patient monitoring (RPM) platforms that combine weight tracking, blood pressure monitoring, glucose data, and symptom reporting in a single dashboard allow providers to monitor multiple patients through treatment transitions efficiently. These platforms can generate automated alerts when clinical parameters exceed thresholds - for example, weight gain exceeding 3% from treatment-gap nadir, or blood pressure rising above target.
Medicare and many commercial insurers now reimburse for RPM services under specific CPT codes (99453, 99454, 99457, 99458), making this approach financially viable for practices managing large GLP-1 patient panels. The combination of RPM with periodic telehealth check-ins creates a monitoring framework that catches problems earlier than traditional quarterly office visits.
Behavioral Health Integration
The evidence consistently shows that pharmacotherapy combined with behavioral intervention produces better long-term outcomes than either approach alone. This is particularly true during treatment transitions, when behavioral skills must compensate for reduced pharmacologic support.
Key Behavioral Interventions During Treatment Gaps
Cognitive Behavioral Therapy (CBT) for Weight Management: CBT techniques help patients identify and modify thought patterns that contribute to overeating. Specific CBT skills particularly relevant during GLP-1 gaps include cognitive restructuring (challenging thoughts like "I've lost control" when appetite returns), stimulus control (modifying the food environment to reduce cues), and behavioral activation (maintaining activity levels despite reduced motivation).
Acceptance and Commitment Therapy (ACT): ACT approaches help patients experience hunger and food cravings without acting on them compulsively. This is particularly valuable during treatment gaps when appetite signals return to pre-treatment intensity. ACT's emphasis on values-based behavior (making food choices aligned with health values rather than driven by momentary appetite) can provide psychological resilience during medication absence.
Motivational Interviewing: Provider-delivered motivational interviewing techniques help maintain patient engagement and treatment adherence during challenging periods. Exploring and resolving ambivalence about restarting medication, addressing frustration about weight regain, and reinforcing the chronic disease model of obesity all benefit from MI techniques.
Integrated Care Models for GLP-1 Management
The most effective long-term GLP-1 management occurs within integrated care models that coordinate pharmacotherapy with behavioral health, nutrition, exercise, and primary care. Several models have emerged:
Medical Weight Management Clinics
Dedicated obesity medicine practices staffed by board-certified obesity medicine specialists, registered dietitians, exercise physiologists, and behavioral health providers. These clinics offer the most comprehensive management but are concentrated in urban and suburban areas, limiting access for rural patients.
Primary Care-Based Obesity Management
Primary care practices that have integrated weight management into their service offerings, often using embedded dietitians and behavioral health consultants. This model provides good continuity of care but may lack the specialized expertise of dedicated obesity programs.
Employer-Sponsored Programs
Large employers increasingly offer integrated weight management programs as employee benefits, recognizing the ROI of treating obesity before it drives downstream healthcare costs. These programs may include GLP-1 coverage, behavioral coaching, gym memberships, and nutritional support in a coordinated package.
Health System-Integrated Programs
Large health systems (Kaiser Permanente, Cleveland Clinic, Mayo Clinic) have developed internal weight management pathways that integrate GLP-1 prescribing with their broader care infrastructure - surgical evaluation for qualifying patients, endocrinology referral for complex metabolic cases, and behavioral health support. These programs benefit from shared electronic health records and care coordination infrastructure.
Regardless of the care model, the principle is consistent: GLP-1 medications are most effective when embedded within a comprehensive treatment plan that addresses the behavioral, psychological, nutritional, and physical activity dimensions of weight management. This multi-dimensional approach is especially important during treatment transitions, when pharmacotherapy alone may be insufficient to maintain progress.
The Role of Community and Peer Support
Patient communities - both online and in-person - have become important sources of information, emotional support, and practical advice during the GLP-1 shortage and its aftermath. Online forums, Facebook groups, Reddit communities, and organized support groups provide spaces where patients share experiences with specific medications, switching protocols, side effect management strategies, and insurance navigation tips.
While peer support can be valuable, patients should approach community advice with appropriate caution:
- Individual responses to medications vary significantly - what worked for one person may not work for another
- Dosing and switching decisions should always be made in consultation with a healthcare provider, not based on community advice alone
- Some online communities may include sponsored content or promotional messaging from medication suppliers or telehealth platforms
- Medical misinformation can circulate in peer communities, particularly around topics like compounding legality, supplement interactions, and alternative treatments
- Emotional support from peers who understand the experience is genuinely valuable and should be encouraged alongside professional care
The ideal approach combines professional medical care with community support - using peer networks for emotional solidarity and practical tips while relying on qualified healthcare providers for clinical decisions about medications, doses, and treatment strategies.
Managing Treatment Gaps
Treatment interruptions are one of the most challenging aspects of GLP-1 therapy management. Whether caused by supply shortages, insurance changes, cost barriers, or administrative delays, gaps in treatment can undo months of progress if not managed proactively.
What Happens When You Stop GLP-1 Therapy
The evidence on weight regain after GLP-1 discontinuation is sobering but consistent. The STEP 1 trial extension followed participants for one year after they stopped semaglutide 2.4 mg. The findings paint a clear picture of the biological reality of obesity as a chronic condition:
- Participants regained approximately two-thirds of their prior weight loss within one year of discontinuation (Wilding et al., 2022; DOI: 10.1111/dom.14725).
- Weight regain was accompanied by reversal of cardiometabolic improvements in blood pressure, lipids, and inflammatory markers.
- Those who had lost the most weight during treatment tended to regain the most in absolute terms, but still retained greater net weight loss than those who had lost less initially.
- More rapid and greater weight regain occurred when lifestyle intervention was also withdrawn, compared to trials where behavioral support continued.
A 2025 narrative review examining weight regain across liraglutide, semaglutide, and tirzepatide studies confirmed that rapid regain occurs regardless of treatment duration - whether therapy lasted weeks or years (Milana et al., 2025; DOI: 10.3390/jcm14113791). This finding reinforces that GLP-1 therapy manages rather than cures the underlying neurohormonal dysregulation driving obesity.
The Biological Reality
Weight regain after stopping GLP-1 therapy isn't a failure of willpower. These medications work by modifying the brain's appetite-regulation circuits and gut-brain signaling. When the drug is removed, those circuits revert to their pre-treatment state. Hunger signals increase, satiety signals decrease, and the body's metabolic set point drives recovery of lost weight. This is why obesity is increasingly recognized as a chronic disease requiring ongoing management - similar to how blood pressure rises when antihypertensive medication is stopped.
Strategies for Planned Treatment Gaps
When a treatment gap is anticipated - perhaps due to insurance re-authorization timelines, a planned surgical procedure, or transition between providers - proactive strategies can minimize impact:
1. Gradual Dose Reduction (When Possible)
Rather than abruptly stopping from a high dose, a gradual taper may reduce the severity of appetite rebound. While no formal taper protocol exists in product labeling, some clinicians recommend stepping down through the titration doses in reverse:
- Semaglutide: 2.4 mg to 1.7 mg to 1.0 mg to 0.5 mg over 4-8 weeks
- Tirzepatide: 15 mg to 10 mg to 7.5 mg to 5 mg to 2.5 mg over 5-10 weeks
This approach is theoretical - there's limited clinical evidence supporting tapering versus abrupt discontinuation. However, the pharmacologic rationale (avoiding sudden removal of receptor activation) is sound, and many patients report subjectively better tolerance with gradual reduction.
2. Intensify Lifestyle Interventions Before the Gap
Establish stronger behavioral foundations while pharmacotherapy is still providing appetite suppression. This might include:
- Working with a registered dietitian to solidify meal planning skills
- Establishing a consistent exercise routine (particularly resistance training)
- Implementing structured eating patterns (regular meal times, portion control systems)
- Beginning mindfulness-based eating awareness practices
3. Consider Bridge Medications
If the gap is expected to last weeks to months, a clinician might prescribe an alternative weight management medication as bridge therapy. Options include:
- Phentermine (short-term, for gaps up to 12 weeks)
- Naltrexone-bupropion (Contrave) for longer gaps, particularly in patients with food-reward-driven eating
- Topiramate alone or as phentermine-topiramate (Qsymia) for sustained appetite suppression
4. Increase Monitoring Frequency
More frequent check-ins - whether in-person, telehealth, or via remote monitoring - help catch weight regain early and provide accountability. Weekly weigh-ins with a healthcare team during the gap period allow for rapid intervention if regain accelerates.
Strategies for Unplanned Treatment Gaps
Unplanned gaps - medication not available at the pharmacy, prior authorization denied, sudden insurance change - require more immediate responses:
- Contact the manufacturer: Both Novo Nordisk and Eli Lilly offer patient assistance programs and savings cards. Novo Nordisk's NovoCare program and Eli Lilly's Lilly Cares Foundation can sometimes provide emergency supplies or financial assistance.
- Ask about samples: Physician offices often receive sample medications from pharmaceutical representatives. These can bridge gaps of 1-4 weeks.
- Explore alternative pharmacies: If one pharmacy is out of stock, others may have supply. Specialty pharmacies, mail-order pharmacies, and pharmacies in different geographic areas may have different inventory levels.
- Request expedited prior authorization: If the gap is due to insurance requirements, the prescribing clinician can often request an expedited or urgent prior authorization review, particularly for patients with diabetes or documented medical necessity.
- Consider therapeutic alternatives: If the specific agent isn't available, switching to an available alternative within the class (even if temporarily) is preferable to an unmedicated gap. Liraglutide, with its generic availability, may be the most accessible short-term alternative.
Restarting GLP-1 Therapy After a Gap
When treatment resumes after an interruption, the approach depends on the duration of the gap:
| Gap Duration | Recommended Restart Approach | Rationale |
|---|---|---|
| 1-2 weeks | Resume at previous dose | GLP-1 receptor tolerance is likely maintained; the half-life of semaglutide (~7 days) means the drug hasn't fully cleared |
| 2-4 weeks | Resume at one step below previous dose, then uptitrate | Partial loss of GLP-1 receptor tolerance; resuming full dose may cause significant GI effects |
| 4-8 weeks | Resume at two steps below previous dose | Substantial loss of receptor tolerance; treating as near-naive reduces adverse event risk |
| 8+ weeks | Restart from the beginning of titration | Full loss of tolerance; treat as a new start to minimize side effects and allow safe dose escalation |
These are general guidelines based on pharmacokinetic principles and clinical experience rather than randomized trial data. Individual variation in tolerance retention means close monitoring during restart is always warranted.
Clinical Pearl: Rapid Re-Titration
Some patients who were previously on high-dose GLP-1 therapy and experienced a gap of 4-8 weeks may tolerate a faster-than-standard re-titration schedule. Instead of 4 weeks at each dose step, 2-week intervals may be feasible for patients who previously demonstrated excellent GI tolerance. However, this should be done with close monitoring and clear communication about when to slow down if symptoms emerge.
Psychological Impact of Treatment Gaps
The emotional and psychological effects of treatment interruptions deserve attention. Patients who've experienced significant weight loss on GLP-1 therapy often describe the return of pre-treatment hunger and food preoccupation during gaps as distressing. Weight regain can trigger feelings of failure, shame, and frustration - even when the patient understands the biological basis of regain.
Clinicians should proactively address these psychological dimensions:
- Frame weight regain during treatment gaps as a predictable physiological response, not a personal failure.
- Emphasize that regained weight can typically be re-lost when medication access is restored.
- Consider referral to a psychologist specializing in obesity and eating behavior for patients experiencing significant distress.
- Monitor for signs of disordered eating patterns that may emerge in response to renewed hunger signals.
Finding a Provider and Cost Navigation
Access to GLP-1 medications involves more than just supply availability. Insurance coverage, prior authorization requirements, provider network constraints, and out-of-pocket costs all create barriers that can be as significant as physical drug shortages.
Types of Providers Prescribing GLP-1s
GLP-1 receptor agonists can be prescribed by various types of healthcare providers, and understanding the landscape helps patients find the most efficient path to treatment:
- Primary care physicians (PCPs): Increasingly comfortable prescribing GLP-1s for both diabetes and weight management. PCPs offer the advantage of comprehensive care coordination and established patient relationships.
- Endocrinologists: Specialists in hormonal and metabolic conditions. Often required by insurance as the prescribing provider for weight management indications. May have more experience with complex cases and switching protocols.
- Obesity medicine specialists: Board-certified in obesity medicine (ABOM). These providers specialize in comprehensive weight management and may have the most nuanced understanding of GLP-1 optimization and alternatives.
- Telehealth platforms: Companies like Ro, Hims/Hers, and specialty telehealth services offer GLP-1 prescribing with convenient access. Some include the medication in bundled pricing. However, the regulatory landscape for telehealth GLP-1 prescribing has tightened, particularly regarding compounded formulations.
Insurance and Prior Authorization
Insurance coverage for GLP-1 medications varies dramatically by payer, plan, and indication. Here's the general landscape:
| Coverage Scenario | Typical Requirements | Patient Cost Impact |
|---|---|---|
| Commercial insurance - T2D indication | Prior auth; may require metformin failure first | $25-$100 copay with coverage |
| Commercial insurance - weight management | BMI criteria; prior auth; possible step therapy | Varies widely; many plans exclude |
| Medicare - T2D indication | Part D coverage; formulary dependent | ~$50/month copay (negotiated pricing) |
| Medicare - weight management | Anti-Obesity Medication coverage expanding | ~$50/month with coverage; varies by plan |
| Medicaid | State-dependent; some cover, many don't for weight | Minimal copay if covered |
| No insurance / cash pay | None | $199-$449/month with manufacturer programs |
For weight management specifically, coverage remains inconsistent. Many commercial plans exclude anti-obesity medications entirely, treating them as cosmetic or elective. The Treat and Reduce Obesity Act has been introduced in Congress multiple times to require Medicare coverage of obesity medications, but comprehensive legislative action remains incomplete as of 2026.
Manufacturer Savings Programs
Both Novo Nordisk and Eli Lilly have implemented significant direct-to-consumer pricing programs that have changed the cost landscape:
Novo Nordisk (Ozempic/Wegovy)
- Introductory pricing: $199/month for first two fills of Ozempic or Wegovy injections
- Ongoing pricing: $299-$349/month depending on medication and dose
- Oral Wegovy: $149/month for lower doses
- NovoCare patient assistance: Free medication for qualifying uninsured patients
- Savings cards: Up to specific monthly savings for commercially insured patients
Eli Lilly (Mounjaro/Zepbound)
- LillyDirect: $299-$449/month depending on dose
- Zepbound savings program for commercially insured patients
- Lilly Cares Foundation: Patient assistance for uninsured/underinsured patients
Figure 7: Monthly cost comparison across GLP-1 access pathways, from insurance copays to manufacturer direct programs to historical compounding costs.
Comprehensive Cost Comparison
| Access Pathway | Monthly Cost Range | Pros | Cons |
|---|---|---|---|
| Commercial insurance with coverage | $25-$100 copay | Lowest cost; FDA-approved product | Prior auth delays; formulary restrictions; step therapy |
| Manufacturer direct programs | $199-$449 | No insurance needed; FDA-approved | Higher than insurance copay; prices may change |
| Medicare Part D | ~$50 copay | Negotiated pricing; predictable cost | Limited to specific formulary agents; coverage restrictions |
| Cash pay at pharmacy | $800-$1,400+ | No prior auth needed; immediate access | Very expensive without discounts |
| Compounded (historical) | $100-$300 | Lower cost; available during shortages | No longer legal for most patients; safety concerns |
| Generic liraglutide | $200-$500 | Lower cost than brand; FDA-approved molecule | Daily dosing; lower efficacy than semaglutide |
| International pharmacy | $200-$600 | Potentially lower cost | Legality concerns; quality assurance challenges |
For patients using the GLP-1 cost calculator, these figures provide baseline comparisons. Individual costs vary based on specific dose, geographic location, pharmacy network, and available discount programs.
Tips for Reducing Out-of-Pocket Costs
- Always check GoodRx or similar discount platforms - prices vary significantly between pharmacies, and coupon programs can reduce costs substantially.
- Apply for manufacturer patient assistance programs before assuming you don't qualify. Income thresholds are often more generous than expected.
- Ask about the obesity indication specifically - some insurance plans cover Wegovy or Zepbound for weight management even when they deny Ozempic or Mounjaro for the same purpose (different formulary tier).
- Consider generic liraglutide as a cost-effective entry point, particularly for patients who haven't tried GLP-1 therapy before.
- Request a letter of medical necessity from your provider when fighting insurance denials - these letters should document BMI, comorbidities, prior treatment failures, and specific medical rationale.
- Explore employer advocacy - some employers have added GLP-1 coverage after employee advocacy, particularly when cost-savings from reduced obesity-related healthcare expenses are demonstrated.
- Consider mail-order pharmacies - 90-day supplies through mail order often cost less per month than 30-day retail fills.
Quality Assurance for Compounded Medications
For the limited circumstances where compounded GLP-1 medications remain legally available - documented allergies to branded excipients or clinically necessary non-standard doses - quality assurance becomes a critical safety consideration.
Red Flags to Watch For
The FDA's enforcement actions have revealed several warning signs that a compounded product may be substandard or fraudulent:
- Unusually low pricing: If a compounded semaglutide product is priced dramatically below market rates (under $100/month), this may indicate corner-cutting in API sourcing, sterility testing, or other quality measures.
- No prescription required: Legitimate compounded medications require a valid prescription from a licensed provider. Any source offering semaglutide or tirzepatide without a prescription is operating outside legal boundaries.
- Vague pharmacy information: The label should clearly identify the compounding pharmacy, including name, address, license number, and contact information. The FDA has identified products with labels from non-existent pharmacies.
- No beyond-use dating: All compounded medications should have a clearly stated beyond-use date (BUD). Injectable products without this information represent a safety risk.
- Unclear salt form: Ask specifically whether the product contains semaglutide base, semaglutide sodium, or semaglutide acetate. If the pharmacy can't clearly answer, this is concerning.
- No third-party testing documentation: Reputable compounding pharmacies can provide certificates of analysis (COAs) for their products, including potency and sterility testing results.
Questions to Ask a Compounding Pharmacy
Patients with legitimate clinical need for compounded GLP-1 medications should ask these questions before filling a prescription:
- Are you registered with the FDA as a 503B outsourcing facility, or are you operating as a 503A pharmacy?
- What is the source of your semaglutide/tirzepatide API, and do you have a Drug Master File (DMF) number for the supplier?
- What salt form of semaglutide are you using?
- Can you provide a certificate of analysis for potency and sterility?
- What is the beyond-use date for this preparation?
- Have you received any FDA warning letters or inspections?
- What is your adverse event reporting process?
- Do you carry professional liability insurance that covers compounded preparations?
Verifying Compounding Pharmacy Legitimacy
Several resources can help verify that a compounding pharmacy is operating legitimately:
- FDA 503B Registration Database: The FDA maintains a list of registered outsourcing facilities at fda.gov. If a pharmacy claims 503B status, it should appear in this database.
- State Board of Pharmacy: Every state's board of pharmacy maintains a database of licensed pharmacies. Verify the pharmacy's license status and check for any disciplinary actions.
- PCAB Accreditation: The Pharmacy Compounding Accreditation Board (PCAB), part of the Accreditation Commission for Health Care, accredits compounding pharmacies that meet rigorous quality standards.
- NABP Verification: The National Association of Boards of Pharmacy (NABP) maintains pharmacy verification tools.
International Compounding Sources
Compounded GLP-1 products sourced from international pharmacies carry additional risks. These products may not meet U.S. pharmaceutical standards, may contain incorrect concentrations, and may have been transported without proper cold-chain maintenance. Importing prescription medications from foreign pharmacies is generally illegal under FDA regulations, though enforcement varies. The FDA's Personal Importation Policy provides limited enforcement discretion for small quantities for personal use, but this is not a legal right.
International Considerations and Options
The GLP-1 shortage was a global phenomenon, though its severity and resolution timeline varied by country. Understanding the international landscape provides context for U.S. patients considering cross-border options and for researchers evaluating worldwide treatment access.
Global Supply Status
While the FDA resolved the U.S. semaglutide shortage in February 2025, supply situations vary internationally:
- European Union: The European Medicines Agency (EMA) issued recommendations to manage Ozempic supply constraints, including restricting new prescriptions for off-label use. Supply has largely stabilized across major EU markets as of late 2025.
- United Kingdom: The NHS implemented specific prescribing guidance during the shortage, prioritizing supply for type 2 diabetes patients over weight management use. Diabetes UK issued FAQs addressing patient concerns about treatment continuity.
- Australia: Temporary import restrictions and prescribing limitations were implemented. The Therapeutic Goods Administration (TGA) authorized limited importation of alternative semaglutide products during peak shortage periods.
- Canada: Health Canada reported intermittent supply disruptions through 2024, with stabilization occurring gradually through 2025.
Patent Expiration and Biosimilar Timeline
Semaglutide's patent landscape is complex, with multiple patents protecting different aspects of the molecule, formulation, and manufacturing process. However, patent expirations are approaching in several key markets:
- Initial semaglutide compound patents begin expiring in some countries as early as 2026.
- Formulation and device patents extend protection through the late 2020s and early 2030s in most markets.
- Biosimilar development for peptide drugs like semaglutide is technically feasible but requires significant investment in analytical characterization, manufacturing process development, and clinical bridging studies.
- Several generic pharmaceutical companies have signaled intent to develop semaglutide biosimilars, though none have reached regulatory approval as of early 2026.
The entry of biosimilar semaglutide products would fundamentally change the access landscape, potentially reducing prices by 15-40% based on precedent from other biosimilar markets. However, realistic commercial availability of semaglutide biosimilars in the U.S. is likely still several years away.
Cross-Border Prescription Considerations
Some U.S. patients have explored obtaining GLP-1 medications from international pharmacies, either through personal travel or online ordering. Key considerations include:
- Legal risk: Importing prescription medications into the U.S. from foreign sources is technically illegal under FDA regulations. The FDA's Personal Importation Policy provides enforcement discretion (not a legal right) for small quantities (typically 90-day supply) when certain conditions are met: the product isn't available domestically, it's for personal use, the quantity is reasonable, and the patient has a U.S. prescriber.
- Quality concerns: Medications purchased from international sources may not meet U.S. pharmaceutical standards. Counterfeit GLP-1 products have been identified in international supply chains.
- Cold chain integrity: GLP-1 medications require refrigeration (36-46 degrees F / 2-8 degrees C) until first use. International shipping may compromise cold chain integrity, reducing efficacy or creating safety risks.
- Cost savings: GLP-1 medications are often priced 30-60% lower in countries with price controls (Canada, EU, UK). However, manufacturer direct pricing in the U.S. ($199-$449/month) has narrowed this gap significantly.
Dose Equivalency Guide
Understanding dose relationships between different GLP-1 agents is essential for clinicians managing patients through transitions. While true pharmacologic equivalence doesn't exist between distinct molecules, clinical trial data allows us to map approximate effect-level comparisons.
Why True Dose Equivalence Doesn't Exist
Several pharmacologic factors make direct dose conversion between GLP-1 agents impossible:
- Different receptor binding profiles: Semaglutide is a pure GLP-1 receptor agonist. Tirzepatide activates both GLP-1 and GIP receptors. Retatrutide activates GLP-1, GIP, and glucagon receptors. These different receptor profiles mean that the same "magnitude" of clinical effect may come from fundamentally different biological pathways.
- Different pharmacokinetics: Half-lives range from ~13 hours (liraglutide) to ~5 days (tirzepatide) to ~7 days (semaglutide). These differences affect steady-state concentration, peak-to-trough fluctuation, and the time course of both therapeutic and adverse effects.
- Non-linear dose-response: The relationship between dose and effect isn't proportional for any of these agents. Doubling the dose of semaglutide doesn't double the weight loss. This makes simple mathematical conversion ratios meaningless.
- Individual variation: Patient-level factors - genetics, body composition, baseline insulin resistance, gut microbiome composition, GLP-1 receptor polymorphisms - create wide variation in response to any given dose.
Effect-Level Mapping Based on Clinical Trial Data
Rather than dose equivalence, we can map approximate "effect levels" - doses of different agents that produce roughly similar magnitudes of weight loss based on major trial data:
| Effect Level | Semaglutide SC | Tirzepatide SC | Liraglutide SC | Approx. Weight Loss |
|---|---|---|---|---|
| Minimal | 0.25-0.5 mg/wk | 2.5 mg/wk | 0.6-1.2 mg/day | 3-6% |
| Moderate | 0.5-1.0 mg/wk | 5.0 mg/wk | 1.8-2.4 mg/day | 6-12% |
| Substantial | 1.0-1.7 mg/wk | 7.5-10 mg/wk | 3.0 mg/day | 10-15% |
| Maximum | 2.4 mg/wk | 15 mg/wk | 3.0 mg/day | 14-21% |
These mappings are approximate and based on population-level trial means. Individual patients may respond very differently. A patient achieving 15% weight loss on semaglutide 1.0 mg might need tirzepatide 10 mg or 15 mg to achieve the same result - or might achieve it at 5 mg. There's no way to predict individual cross-agent response without empiric trial.
Figure 8: Approximate effect-level mapping between GLP-1 receptor agonists. These are clinical approximations, not pharmacologic equivalences.
Clinical Application of Effect-Level Mapping
While these mappings can't guide direct dose conversion (which should always start at the introductory dose of the new agent), they serve several practical purposes:
- Setting expectations: A patient switching from semaglutide 2.4 mg to tirzepatide can understand that they may need to reach 10-15 mg before seeing equivalent or greater weight loss effect.
- Planning titration timelines: Knowing that a patient needs to reach approximately tirzepatide 10 mg for equivalent effect helps predict a 3-4 month titration period.
- Insurance documentation: Effect-level comparisons can support prior authorization appeals when switching agents, demonstrating medical rationale for the transition.
- Clinical decision-making: If a patient plateaued at moderate weight loss on semaglutide 1.0 mg, switching to tirzepatide and titrating beyond the equivalent effect level (5 mg) to 10-15 mg offers a pharmacologically rational path to greater efficacy.
Special Dosing Considerations
Renal Impairment
Semaglutide and tirzepatide don't require dose adjustment in mild-to-moderate renal impairment (eGFR 30-89 mL/min). For severe impairment (eGFR 15-29 mL/min), both agents should be used with caution - not due to drug accumulation (they're primarily metabolized by proteolytic degradation) but because GI side effects like nausea, vomiting, and diarrhea can cause dehydration that worsens kidney function.
Hepatic Impairment
Limited data exists on GLP-1 agonist use in severe hepatic impairment. Mild-to-moderate hepatic impairment doesn't require dose adjustment. Interestingly, emerging data suggests GLP-1 agonists may actually benefit patients with non-alcoholic fatty liver disease (NAFLD) and non-alcoholic steatohepatitis (NASH), making them potentially therapeutic rather than concerning in this population.
Elderly Patients (>65 years)
No dose adjustment is needed based solely on age, but elderly patients may be more susceptible to dehydration from GI side effects. Slower titration and more frequent monitoring are prudent, especially during switching protocols.
Pediatric/Adolescent Patients
Semaglutide (Wegovy) is approved for adolescents aged 12 and older with obesity. Liraglutide (Saxenda) is approved for adolescents aged 12 and older. Tirzepatide's pediatric data is still accumulating. Switching protocols in pediatric populations should follow conservative approaches with extended titration intervals and close monitoring.
The Future of GLP-1 Access and Supply
The GLP-1 shortage catalyzed changes across the pharmaceutical supply chain, regulatory framework, and clinical practice that will shape medication access for years to come. Here's what the future landscape looks like based on current trajectories.
Manufacturing Capacity Expansion
Both Novo Nordisk and Eli Lilly have committed massive capital to expanding production capacity, which should provide long-term supply security:
- Novo Nordisk: $6 billion in facility expansions, including $4.1 billion for a second U.S. fill-and-finish facility in Clayton, North Carolina. An additional $500 million investment in an Irish facility for oral GLP-1 manufacturing. Total capex of DKK 55 billion projected for 2026. Construction will finalize gradually from late 2025 through 2029.
- Eli Lilly: Billions invested in expanding manufacturing in the U.S. (Lebanon, Indiana; Research Triangle Park, North Carolina) and internationally (Limerick, Ireland; Alzey, Germany). These investments are designed to meet projected demand growth through the end of the decade.
New Formulations and Delivery Methods
Several innovations in GLP-1 delivery could diversify supply chains and improve access:
- Oral semaglutide 25 mg: Launched in January 2026 for obesity, this provides an alternative supply chain independent of injectable manufacturing. Oral formulations require different APIs (higher quantity due to low bioavailability) but avoid the sterile fill-and-finish bottleneck.
- Wegovy vials: Novo Nordisk has signaled a future launch of Wegovy in vial format rather than pre-filled pens. Vials are less expensive to manufacture and allow more flexible dosing, potentially improving both supply and cost.
- Oral non-peptide GLP-1 agonists: Eli Lilly's orforglipron, a small molecule (non-peptide) oral GLP-1 agonist, could be manufactured using traditional chemical synthesis rather than peptide manufacturing - dramatically simplifying the supply chain. Phase 3 data is expected and regulatory submission is planned.
- Long-acting formulations: Research into monthly or even less frequent dosing formulations could reduce the total number of doses manufactured and distributed, effectively increasing supply capacity.
Regulatory Evolution
The GLP-1 shortage has prompted regulatory discussions about how the FDA manages drug shortages and compounding oversight:
- Shortage prevention: Proposals for earlier manufacturer notification requirements, buffer stock mandates, and more granular demand forecasting data sharing between manufacturers and the FDA.
- Compounding reform: The friction between resolving shortages and removing compounding access has generated calls for a more graduated transition process rather than the current binary on/off shortage list approach.
- Biosimilar pathway: Regulatory clarity around biosimilar peptides could accelerate generic competition, though the technical challenges of peptide biosimilar development are substantial.
- International harmonization: The global nature of the shortage has increased interest in mutual recognition agreements for drug shortage management across regulatory agencies.
Insurance Coverage Evolution
The economics of GLP-1 coverage are evolving rapidly:
- Medicare negotiated prices for Ozempic, Wegovy, Mounjaro, and Zepbound at approximately $245/month, with patient copays around $50/month - a dramatic reduction from list prices.
- An increasing number of commercial insurers are adding anti-obesity medication coverage as the evidence base for long-term health benefits and cost savings grows.
- Employer health plans are increasingly evaluating GLP-1 coverage based on total healthcare cost analyses that factor in reduced spending on obesity-related complications.
- Specialty pharmacy programs and integrated care models are emerging to optimize GLP-1 prescribing, monitoring, and cost management.
For ongoing updates on the GLP-1 medication landscape, including new approvals, supply updates, and access resources, our research hub is regularly updated with the latest developments.
Pipeline Medications That Could Reshape the Landscape
Several next-generation compounds could fundamentally change GLP-1 access dynamics by creating more supply diversity:
| Compound | Developer | Mechanism | Status | Potential Impact |
|---|---|---|---|---|
| Retatrutide | Eli Lilly | Triple agonist (GLP-1/GIP/glucagon) | Phase 3 | Up to 24.2% weight loss; new manufacturing stream |
| Orforglipron | Eli Lilly | Oral non-peptide GLP-1 agonist | Phase 3 | Simplified manufacturing; no peptide supply chain dependency |
| Survodutide | Boehringer Ingelheim/Zealand | Dual GLP-1/glucagon agonist | Phase 3 | Different manufacturer diversifies supply |
| Pemvidutide | Altimmune | Dual GLP-1/glucagon agonist | Phase 2b | Additional supply source; liver fat reduction |
| Ecnoglutide | Sciwind Biosciences | Long-acting GLP-1 agonist | Phase 3 (China) | Monthly dosing; reduced manufacturing burden |
| CagriSema | Novo Nordisk | Semaglutide + cagrilintide | Phase 3 | Enhanced efficacy may mean fewer patients need dose escalation |
The entry of multiple new agents from multiple manufacturers over the next 3-5 years should substantially reduce the risk of future single-agent shortages. For a complete overview of the obesity pharmacotherapy landscape, see our dedicated report.
Patient Action Plan: Step-by-Step Guidance for Every Scenario
Regardless of where you are in your GLP-1 treatment journey, specific actionable steps can help you maintain access, manage transitions, and protect your health outcomes. This section provides scenario-based guidance for the most common situations patients face in the post-shortage environment.
Scenario 1: Your Pharmacy Says Ozempic/Wegovy Is Out of Stock
Even though the FDA has resolved the national shortage, individual pharmacies may still experience temporary stock-outs due to distribution timing, local demand spikes, or inventory management issues. Here's what to do:
- Don't panic about a single stock-out. This is a distribution issue, not a national shortage. Supply is flowing from manufacturers to wholesalers to pharmacies, but the last mile can be inconsistent.
- Call other pharmacies. Different pharmacies receive shipments on different days. Call 3-5 pharmacies in your area, including chain pharmacies (CVS, Walgreens, Rite Aid), independent pharmacies, and specialty pharmacies. Ask specifically about the dose strength you need.
- Ask about expected restock dates. Pharmacies typically know when their next wholesaler delivery is scheduled. If restock is expected within 2-3 days, it may be worth waiting rather than switching pharmacies.
- Consider mail-order pharmacy. Many insurance plans offer mail-order pharmacy options (Express Scripts, OptumRx, Caremark) that maintain larger inventories and can ship 90-day supplies. The initial setup takes 1-2 weeks but provides more reliable long-term supply.
- Contact Novo Nordisk directly. NovoCare (1-888-693-3276) can help locate pharmacies with available stock and may provide bridge supplies in certain circumstances.
- Ask your provider about alternatives. If no pharmacy has your specific medication within a reasonable timeframe, your provider can prescribe an available alternative within the class.
Scenario 2: Your Insurance Denies GLP-1 Coverage
Insurance denials are one of the most common barriers to GLP-1 access, even when supply is plentiful. The appeal process, while frustrating, is often successful with proper documentation.
Understanding Common Denial Reasons
| Denial Reason | What It Means | Response Strategy |
|---|---|---|
| Not medically necessary | Insurer doesn't think the evidence supports coverage for your situation | Letter of medical necessity from provider with BMI, comorbidity documentation, and clinical trial data |
| Step therapy required | Must try cheaper medication first (usually metformin or phentermine) | Document prior failures or clinical contraindications to the required step; expedited exception request |
| Prior authorization needed | Administrative requirement before dispensing | Provider submits prior auth; often approved within 24-72 hours |
| Off-formulary | Your plan doesn't include this specific medication | Formulary exception request with evidence of medical necessity; ask about covered alternatives |
| BMI criteria not met | Your BMI doesn't reach the plan's threshold (typically 30+, or 27+ with comorbidity) | Document all comorbidities; some plans accept lower thresholds with specific conditions |
| Weight management exclusion | Plan excludes anti-obesity medications as a category | If also diabetic, request coverage under diabetes indication instead; appeal through employer if self-insured plan |
The Appeals Process
If your initial claim or prior authorization is denied:
- First-level appeal (internal): File within 30-60 days of denial. Include: letter of medical necessity from your provider, relevant clinical trial data, documentation of your BMI history, list of comorbidities with clinical evidence, prior treatment attempts and outcomes, and a statement explaining why this specific medication is necessary.
- Second-level appeal (external): If the internal appeal is denied, you have the right to an external independent review. An independent physician reviewer examines your case. External review decisions are binding on the insurer in most states.
- State insurance department complaint: Filing a complaint with your state's Department of Insurance creates regulatory pressure and an additional review pathway.
- Employer advocacy (for self-insured plans): If your employer self-insures, they have the power to modify coverage decisions. A well-documented request to HR, especially one that demonstrates cost-effectiveness of GLP-1 treatment versus ongoing obesity-related healthcare costs, can result in plan modifications.
Appeal Success Rates
Data from state insurance departments shows that first-level appeals for GLP-1 medications are successful 40-60% of the time. External reviews succeed at even higher rates, often 50-70%, because independent reviewers tend to weigh clinical evidence more heavily than cost considerations. The effort required for an appeal - typically a few hours of your and your provider's time - is well worth it given the high success rates.
Scenario 3: You Can No Longer Afford Your GLP-1 Medication
Cost is the most common reason for GLP-1 discontinuation after supply is resolved. Multiple strategies exist to reduce your financial burden:
Manufacturer Programs (Apply for These First)
- Novo Nordisk NovoCare: Provides free Ozempic and Wegovy for qualifying uninsured patients. Income thresholds are more generous than you might expect. Application available at NovoCare.com or by calling 1-888-693-3276.
- Eli Lilly Cares Foundation: Similar program for Mounjaro and Zepbound. Income-based eligibility with a straightforward application process.
- Savings cards for commercially insured patients: Both manufacturers offer savings cards that reduce copays for commercially insured patients. These don't apply to government insurance (Medicare, Medicaid, Tricare).
- LillyDirect and NovoDirect: Manufacturer direct programs offering reduced pricing without insurance involvement. Zepbound through LillyDirect: $299-$449/month. Ozempic/Wegovy direct: $199-$349/month.
Alternative Access Strategies
- Generic liraglutide: Available since July 2025, generic Victoza/Saxenda alternatives typically cost $200-$500/month - substantially less than brand-name semaglutide or tirzepatide.
- Oral semaglutide: If your insurance covers Rybelsus (for diabetes) but not Wegovy, the oral formulation may be available at a lower copay tier.
- Clinical trials: ClinicalTrials.gov lists ongoing studies for GLP-1 medications and next-generation agents. Trial participation provides free medication and close medical monitoring, though you may receive placebo.
- Compounding (limited): If you have a documented allergy to a branded excipient or need a non-standard dose, compounding may still be legally available. Discuss with your provider.
- Dose optimization: Some patients achieve adequate response at lower doses than maximum. Discuss with your provider whether a lower dose (e.g., semaglutide 1.0 mg instead of 2.4 mg) provides sufficient benefit - lower doses are often less expensive and may have fewer side effects.
Scenario 4: You're Experiencing Severe Side Effects and Considering Stopping
GI side effects - nausea, vomiting, diarrhea, constipation, and abdominal discomfort - are the most common reason patients discontinue GLP-1 therapy. Before stopping entirely, consider these strategies:
Dose Modification
- Step back to a lower dose: If side effects became intolerable after a dose increase, return to the previous tolerated dose for an extended period (6-8 weeks instead of 4) before attempting the increase again.
- Split timing: For daily agents like liraglutide, some patients tolerate the medication better at a different time of day (evening versus morning).
- Injection site rotation: While absorption shouldn't vary significantly by injection site (abdomen, thigh, upper arm), some patients report different GI tolerability with different sites.
Dietary Modifications
- Eat smaller, more frequent meals: 5-6 small meals rather than 2-3 large ones reduces the gastric distension that triggers nausea with GLP-1 medications.
- Avoid high-fat foods: Fatty meals slow gastric emptying further (already delayed by GLP-1 agonism), amplifying nausea and fullness.
- Stay well hydrated: Small sips of water, ginger tea, or electrolyte solutions throughout the day. Avoid drinking large volumes at once.
- Ginger supplements: Ginger (250 mg capsules or fresh ginger tea) has evidence for anti-nausea effects and may help manage mild-to-moderate GLP-1-related nausea.
- Avoid trigger foods: Commonly reported trigger foods include spicy foods, citrus, carbonated beverages, alcohol, and overly processed foods. Keep a food diary during the first weeks at each dose level to identify your personal triggers.
When to Consider Switching Instead of Stopping
If side effects remain intolerable despite these measures:
- Switch to a different GLP-1 agent - different patients tolerate different agents, and the GI side effect profile varies between molecules
- Switch from injectable to oral semaglutide (or vice versa) - the route of administration affects the GI symptom pattern
- Consider liraglutide's daily dosing as an option for patients who experience severe "peaks" of nausea with weekly injections - the smoother daily profile may be better tolerated
- Discuss non-GLP-1 alternatives like tesofensine or phentermine-topiramate that work through different mechanisms without direct GI effects
Scenario 5: You're Pregnant or Planning Pregnancy
GLP-1 receptor agonists are contraindicated in pregnancy. All agents in the class carry reproductive toxicity warnings based on animal studies, and there isn't sufficient human data to establish safety during pregnancy.
Current guidance recommends:
- Discontinue semaglutide at least 2 months before planned conception (based on the 5-half-life washout period of ~5 weeks, with additional safety margin)
- Discontinue tirzepatide at least 1 month before planned conception
- Discontinue liraglutide at least 2 weeks before planned conception
- Use reliable contraception while on GLP-1 therapy - and be aware that weight loss itself can improve fertility in women with obesity-related anovulation, potentially leading to unplanned pregnancies
- Discuss weight management strategies during preconception and pregnancy with your obstetric provider
Scenario 6: You're a Medicare Beneficiary
Medicare coverage for GLP-1 medications has evolved significantly:
- For type 2 diabetes: Ozempic and Mounjaro are covered under Medicare Part D for the diabetes indication. Negotiated prices bring costs to approximately $245/month with copays around $50/month.
- For weight management: Coverage for Wegovy and Zepbound under Medicare has been expanding, particularly following the demonstration of cardiovascular benefits. The Medicare Anti-Obesity Medication (AOM) coverage landscape continues to evolve. Check your specific Part D plan formulary for current coverage status.
- Low-Income Subsidy (LIS/Extra Help): Medicare beneficiaries who qualify for the LIS program may pay as little as $0-$10 per prescription.
- Medicare Advantage plans: Some MA plans offer more generous GLP-1 coverage than traditional Medicare. Open enrollment periods allow plan switching to access better formulary coverage.
Evidence Quality and Research Methodology Assessment
Understanding the quality and limitations of the evidence underpinning GLP-1 shortage management strategies helps clinicians and patients make appropriately calibrated decisions. Not all recommendations in shortage management carry equal evidentiary weight.
Evidence Hierarchy for GLP-1 Treatment Decisions
The recommendations in this guide draw from evidence of varying quality levels. Understanding these distinctions helps prioritize reliable information:
Strong Evidence (Randomized Controlled Trials)
- Weight loss efficacy: The STEP trials (semaglutide), SURMOUNT trials (tirzepatide), and SCALE trials (liraglutide) provide strong Phase 3 data on the magnitude of weight loss with each agent. These trials involved thousands of participants, lasted 52-72 weeks, and used rigorous double-blind, placebo-controlled designs.
- Cardiovascular outcomes: The SELECT trial (semaglutide), LEADER trial (liraglutide), and REWIND trial (dulaglutide) provide high-quality evidence for cardiovascular benefit. These were large-scale, multicenter, international trials specifically designed and powered to detect cardiovascular event reduction.
- Weight regain after discontinuation: The STEP 1 extension and other controlled withdrawal studies provide strong evidence that weight regain is rapid and substantial after GLP-1 discontinuation.
Moderate Evidence (Observational Studies, Subgroup Analyses)
- Real-world effectiveness: Pharmacy claims data, electronic health record analyses, and registry studies confirm that GLP-1 medications produce meaningful weight loss in real-world populations, though typically somewhat less than in clinical trials (where adherence and monitoring are more intensive).
- Switching outcomes: Limited observational data exists on outcomes when switching between GLP-1 agents. Most switching guidance is extrapolated from pharmacokinetic principles and individual agent trial data rather than head-to-head switching studies.
- Cost-effectiveness: Health economic analyses suggest GLP-1 therapy is cost-effective when considering reduced downstream healthcare costs from obesity-related complications, though these analyses involve assumptions about treatment duration, adherence, and long-term outcomes that introduce uncertainty.
Expert Consensus (Clinical Experience, Guidelines)
- Switching protocols: No randomized trials have studied optimal protocols for switching between GLP-1 agents. Current recommendations (start at low dose, titrate per standard schedule, time switch to next injection date) are based on pharmacologic principles and expert clinical experience.
- Restart protocols after gaps: The recommended approaches for restarting therapy after different gap durations are based on pharmacokinetic reasoning and clinical experience rather than formal studies.
- Bridge therapy strategies: Using non-GLP-1 medications to bridge treatment gaps is a practical approach based on clinical reasoning, not randomized evidence.
Limited/Emerging Evidence
- Non-GLP-1 alternatives: Research compounds like tesofensine, 5-amino-1MQ, and AOD-9604 have preclinical data and limited Phase 2 human data, but lack the large-scale clinical evidence supporting FDA-approved agents. Decisions to use these compounds should be made with full awareness of the evidence limitations.
- Compounded product quality: Systematic data on the quality and safety of compounded GLP-1 products is limited to FDA adverse event reports (with known underreporting) and select inspection findings. No large-scale comparative safety studies exist between compounded and brand-name GLP-1 products.
- Tapering protocols: The suggestion to taper rather than abruptly stop GLP-1 therapy before planned gaps is based on pharmacologic theory, not comparative trial data. No study has demonstrated that tapering produces better outcomes than abrupt discontinuation.
Gaps in Current Research
Several important questions remain inadequately studied:
- Optimal switching protocols: What is the best way to switch between GLP-1 agents? Should patients start from the lowest dose or can certain patients safely start at intermediate doses based on prior GLP-1 tolerance? Randomized studies comparing different switching strategies are needed.
- Long-term outcomes of treatment interruptions: While we know that weight regain occurs after GLP-1 discontinuation, we don't know whether repeated cycles of treatment and interruption have different long-term metabolic consequences than continuous treatment. The concept of "weight cycling" with pharmacotherapy needs further study.
- Comparative effectiveness in real-world switching: When patients switch from semaglutide to tirzepatide (or vice versa), what percentage achieve equivalent or better outcomes? Real-world data on switching outcomes would help set appropriate expectations.
- Patient selection for non-GLP-1 alternatives: Which patients are most likely to benefit from non-GLP-1 weight management approaches when GLP-1s aren't accessible? Biomarkers or clinical characteristics that predict response to different mechanism classes would be valuable.
- Health equity impacts of shortage and cost barriers: How did the shortage differentially affect outcomes across racial, ethnic, socioeconomic, and geographic lines? What interventions could reduce these disparities in future shortage scenarios?
Interpreting Efficacy Data Across Agents
When comparing weight loss numbers across different GLP-1 medications, several methodological considerations are important:
- Trial populations differ: STEP 1 enrolled adults with BMI 30+ (or 27+ with at least one comorbidity). SURMOUNT-1 had similar criteria. But baseline BMI, age distribution, sex ratio, and comorbidity burden vary between trials, affecting the percentage weight loss observed.
- Trial designs differ: Some trials used intensive lifestyle intervention alongside drug therapy (e.g., STEP 3), while others used less intensive behavioral support. The lifestyle intervention component can substantially influence outcomes.
- Duration differs: STEP 1 ran 68 weeks. SURMOUNT-1 ran 72 weeks. SCALE Obesity ran 56 weeks. Weight loss trajectories haven't fully plateaued at any of these timepoints for maximum doses, making direct cross-trial comparisons imprecise.
- Completers vs. ITT: Published headline numbers are typically intention-to-treat (ITT) analyses that include dropouts. On-treatment or completer analyses often show greater weight loss but represent a selected population.
- No head-to-head trials at maximum doses: While SURPASS-2 compared tirzepatide to semaglutide 1.0 mg, no published trial has directly compared tirzepatide 15 mg to semaglutide 2.4 mg. Cross-trial comparisons at maximum doses are inherently approximate.
These methodological considerations don't invalidate the comparative conclusions - tirzepatide is clearly more efficacious than semaglutide, and both are substantially more effective than liraglutide for weight loss. But they do mean that the exact magnitude of differences between agents is somewhat uncertain, and individual patient response will vary more than population-level averages suggest.
Applying Evidence to Individual Patient Decisions
Clinical trial data provides population-level estimates, but individual patients are not population averages. Several factors modulate individual response to GLP-1 therapy:
- Genetic factors: Variations in GLP-1 receptor gene (GLP1R), GIP receptor gene (GIPR), and genes affecting drug metabolism influence individual response. While pharmacogenomic testing for GLP-1 response isn't yet clinically available, research in this area is active.
- Baseline metabolic state: Patients with higher baseline insulin resistance, greater degrees of beta-cell dysfunction, or different adipose tissue distribution patterns may respond differently to specific agents.
- Behavioral factors: Adherence, dietary quality, physical activity level, sleep patterns, and stress management all modulate the clinical response to GLP-1 therapy.
- Concomitant medications: Other medications that affect weight (antipsychotics, antidepressants, beta-blockers, insulin) can attenuate or amplify GLP-1 medication effects.
- Prior weight loss history: Patients with a history of weight cycling may have different metabolic responses than those attempting weight loss for the first time.
The practical implication is that treatment decisions should be individualized based on the patient's complete clinical picture, preferences, and practical constraints - not based solely on population-level efficacy rankings. A medication that produces 8% average weight loss but is affordable, tolerable, and accessible for a specific patient is more effective in practice than a medication producing 20% average weight loss that the patient can't afford, can't tolerate, or can't obtain.
Mental Health and Psychological Impact of GLP-1 Treatment Disruptions
One aspect of the GLP-1 shortage that rarely gets discussed in clinical literature is the psychological toll on patients who suddenly lose access to a medication that was working for them. Weight management carries enormous emotional weight in our culture, and the experience of finding an effective treatment only to have it yanked away by supply chain problems creates a distinctive pattern of distress that providers need to recognize and address.
The Emotional Cycle of Treatment Interruption
Patients who experience forced treatment gaps commonly describe a predictable emotional progression. The initial reaction is anxiety about weight regain, followed by frustration with the healthcare system, then guilt and self-blame as appetite returns and eating patterns shift. Many patients report that the psychological impact of the treatment interruption was worse than the physical weight regain, because it reactivated feelings of helplessness and failure that the medication had helped alleviate.
This emotional pattern is compounded by the pharmacology of GLP-1 agonists themselves. Semaglutide and tirzepatide don't just reduce appetite through peripheral mechanisms. They also act on brain reward circuits, reducing the hedonic drive to eat and dampening food-related cravings. When the medication is withdrawn, these central effects reverse, and patients experience what feels like a return of overwhelming food thoughts and urges. It's not a lack of willpower; it's the reactivation of neurobiological drives that the medication had been suppressing.
Providers should proactively discuss this rebound phenomenon with patients facing treatment gaps. Simply knowing that increased appetite and food preoccupation are expected pharmacological effects of medication withdrawal, not personal failures, can significantly reduce the emotional distress. Setting realistic expectations about temporary weight fluctuations during gaps and establishing clear criteria for when to resume treatment helps patients maintain perspective.
Disordered Eating Considerations
The relationship between GLP-1 medications and disordered eating patterns is complex and clinically important during supply disruptions. Some patients with binge eating disorder (BED) experience significant symptom improvement on GLP-1 therapy, and abrupt discontinuation can trigger binge relapse. For these patients, a treatment gap isn't just about weight, it's about the return of a psychiatric condition that the medication was managing.
Conversely, some patients develop overly restrictive eating patterns while on GLP-1 medications, consuming dangerously few calories because the appetite suppression makes it easy to skip meals. When the medication is interrupted, these patients may swing in the opposite direction, overeating in response to returned hunger signals. Screening for disordered eating patterns during treatment and providing nutritional guidance for medication transitions can help prevent these extremes.
Providers managing patients through GLP-1 treatment gaps should consider temporary supportive interventions including cognitive behavioral therapy strategies for managing food-related anxiety, mindful eating practices that help patients reconnect with hunger and satiety cues, and in some cases, short-term use of adjunctive medications like naltrexone-bupropion to bridge the gap in appetite management.
Building Psychological Resilience for Supply Uncertainty
Given that supply disruptions may recur, helping patients build psychological resilience around their weight management journey is a worthwhile clinical investment. This includes reframing weight management as a long-term process with expected fluctuations rather than a linear trajectory, identifying non-scale measures of health improvement (fitness levels, lab values, energy, sleep quality) that persist even during temporary weight regain, and developing behavioral skills that support weight stability independent of medication.
Support communities, both online and in-person, can also provide significant psychological benefit during treatment disruptions. Connecting with others who understand the specific frustrations of medication access challenges reduces isolation and provides practical tips for managing the transition. Many patients report that peer support during shortage-related gaps was more helpful than clinical advice alone.
For patients exploring complementary approaches during treatment gaps, several non-GLP-1 peptides support metabolic health through different pathways. AOD-9604 targets fat metabolism, MOTS-c enhances cellular energy production and insulin sensitivity, and tesofensine addresses appetite through central monoamine reuptake inhibition. The lifestyle hub covers behavioral strategies that complement pharmacological approaches, and the free assessment can help identify which combination of approaches might best support your goals during a treatment transition.
Insurance Coverage Strategies for GLP-1 Medications: Navigating Authorization and Appeals
One of the most persistent barriers to GLP-1 access, independent of manufacturing supply, is insurance coverage. Many commercial insurance plans and Medicare Part D plans impose strict prior authorization requirements, step therapy protocols, and quantity limits on GLP-1 receptor agonists, particularly when prescribed for weight management rather than type 2 diabetes. Understanding how to navigate these administrative hurdles can mean the difference between affordable access and out-of-pocket costs that exceed $1,000 per month for brand-name medications.
Prior authorization requirements typically mandate documentation of specific clinical criteria before coverage is approved. For diabetes indications, most plans require evidence of inadequate glycemic control on first-line therapy (usually metformin), a recent HbA1c above a threshold value (commonly 7.0% or 7.5%), and documentation that the patient has tried and failed or has contraindications to at least one less expensive diabetes medication. For obesity indications, the requirements are generally more stringent: a BMI above 30 (or above 27 with at least one weight-related comorbidity), documentation of a structured diet and exercise program of specified duration, and often evidence that the patient has failed previous weight loss medications or has contraindications to them.
When initial prior authorization is denied, the appeals process becomes critical. First-level appeals are typically reviewed by the insurance company's own clinical team and succeed in approximately 30-40% of cases when supported by additional clinical documentation. Second-level appeals, which often involve external review by an independent physician, have higher success rates (50-60%) because external reviewers tend to apply medical necessity criteria more liberally. Key elements of a successful appeal include detailed documentation of the patient's medical history, objective evidence of failed alternative therapies, clinical literature supporting the requested medication for the patient's specific condition, and a clear statement of medical necessity from the prescribing physician.
Step therapy requirements deserve special attention. Many plans require patients to try and fail older, less expensive GLP-1 agonists before covering newer agents like semaglutide or tirzepatide. While this may seem reasonable from a cost-containment perspective, it can delay access to more effective therapies by months. Some states have enacted step therapy reform laws that limit how long patients can be required to remain on a step therapy drug before advancing, and that provide exceptions when the prescriber can document clinical reasons why the preferred drug is inappropriate. Patients and prescribers should familiarize themselves with their state's step therapy laws, as these protections vary significantly by jurisdiction.
Manufacturer patient assistance programs represent another avenue for reducing costs. Both Novo Nordisk and Eli Lilly operate savings card programs for commercially insured patients that can reduce copays to as little as $25 per month, though these programs typically exclude patients with government insurance (Medicare, Medicaid, Tricare). For uninsured or underinsured patients, both manufacturers offer free medication programs based on income eligibility, though application processing can take several weeks. The free assessment tool can help patients evaluate their full range of options, including both brand-name and alternative approaches to GLP-1 therapy, while they work through the insurance authorization process.
Employer-sponsored weight management programs represent an increasingly common pathway to GLP-1 coverage. As employers recognize the long-term cost savings associated with treating obesity (reduced hospitalizations, fewer surgical procedures, lower disability claims), many are adding dedicated weight management benefits that include GLP-1 medications. Patients who have been denied coverage through their standard pharmacy benefit should check whether their employer offers a separate weight management program, as these programs sometimes cover medications that the pharmacy benefit excludes. Human resources departments can clarify what obesity treatment benefits are available, and some employers contract with specialized vendors that provide both the medication and the behavioral support components as a bundled benefit.
Frequently Asked Questions
What can I take instead of Ozempic if it's not available or too expensive?
Several alternatives exist depending on your specific needs. Tirzepatide (Mounjaro/Zepbound) is the most effective alternative, producing even greater weight loss than semaglutide in clinical trials - up to 20.9% at the 15 mg dose. Generic liraglutide (previously Victoza/Saxenda) became available in July 2025 and offers a more affordable GLP-1 option, though it requires daily injections and produces more modest weight loss (~8%). Oral semaglutide 25 mg launched in January 2026 for obesity and may have a different supply chain than injectable forms. For non-GLP-1 options, phentermine-topiramate (Qsymia) produces 7-10% weight loss, and naltrexone-bupropion (Contrave) produces 5-6%. Your best choice depends on your insurance coverage, medical history, and treatment goals - discuss all options with your healthcare provider.
Is semaglutide still in shortage?
No. The FDA officially resolved the semaglutide injection shortage on February 21, 2025, after nearly three years of supply disruptions. By October 2025, all dose strengths of both Ozempic and Wegovy were confirmed available, including the 0.25 mg Wegovy starter dose that was the last to resolve. Novo Nordisk is now regularly shipping all doses to U.S. wholesalers. However, individual pharmacies may still experience temporary stock-outs due to distribution logistics, and some patients may struggle to access the medication due to insurance barriers or cost rather than physical supply limitations.
Can I still get compounded semaglutide?
For most patients, no. After the FDA resolved the semaglutide shortage, 503A pharmacies had until April 22, 2025, and 503B outsourcing facilities had until May 22, 2025, to stop compounding semaglutide. After these deadlines, compounding is only permitted in narrow circumstances - such as a documented allergy to an inactive ingredient in the branded product, or a clinically necessary dose strength that isn't commercially available. The compounded formulation must be "meaningfully different" from the commercial product and can't simply be a cheaper substitute. The FDA has issued over 50 warning letters to GLP-1 compounders, and manufacturers have sent cease-and-desist letters. If you were previously using compounded semaglutide, work with your provider to transition to brand-name options or alternative medications.
How do I switch from semaglutide to tirzepatide?
Start tirzepatide at 2.5 mg weekly on the day your next semaglutide injection would have been due - don't overlap the medications. Even if you were on the maximum semaglutide dose (2.4 mg), you must begin tirzepatide at 2.5 mg because its dual GIP/GLP-1 mechanism is pharmacologically distinct. Titrate upward by 2.5 mg every 4 weeks as tolerated: 2.5 mg, then 5 mg, 7.5 mg, 10 mg, 12.5 mg, up to 15 mg maximum. Expect a temporary plateau in weight loss during the early titration weeks. Many patients who tolerated semaglutide well handle the early tirzepatide doses without issue, but the GIP receptor activation is novel and may cause different GI patterns. Don't rush the titration - reaching optimal dose safely takes 3-5 months.
What happens if I have to stop my GLP-1 medication suddenly?
Research from the STEP 1 trial extension showed that patients regained approximately two-thirds of their weight loss within one year of stopping semaglutide. Weight regain is accompanied by reversal of cardiometabolic improvements in blood pressure, lipids, and inflammatory markers. This happens because GLP-1 medications manage, rather than cure, the neurohormonal dysregulation driving obesity. When the drug is removed, appetite signals revert to pre-treatment patterns. To minimize impact, intensify lifestyle interventions (especially protein intake and resistance training), consider bridge medications like phentermine or naltrexone-bupropion, increase monitoring frequency, and work with your provider to restore GLP-1 access as quickly as possible.
Is compounded semaglutide as safe as brand-name Ozempic or Wegovy?
Compounded semaglutide carries additional safety risks compared to FDA-approved products. As of July 2025, the FDA received 605 adverse event reports tied to compounded semaglutide - likely representing significant underreporting. Key concerns include dosing errors (patients measuring 5-20x the intended dose from multi-dose vials), use of different salt forms (semaglutide sodium or acetate rather than the base form in approved products), sterility issues at some compounding facilities, and outright fraudulent products with labels from non-existent pharmacies. FDA-approved products undergo rigorous batch testing for potency, sterility, and stability, and use pre-filled injection pens that eliminate measurement error. While many patients used compounded semaglutide without problems during the shortage, the risk profile is objectively higher than brand-name products.
Are there non-injectable alternatives to GLP-1 drugs for weight loss?
Yes, several options exist. Oral semaglutide 25 mg (launched January 2026) provides the same active ingredient as Wegovy in pill form, though it requires careful timing - taken on an empty stomach with minimal water and no eating for 30 minutes after. Beyond GLP-1s, FDA-approved oral weight loss medications include phentermine-topiramate (Qsymia, ~7-10% weight loss), naltrexone-bupropion (Contrave, ~5-6%), and orlistat (Xenical/Alli, ~3-5%). Research compounds like tesofensine (a triple monoamine reuptake inhibitor showing 9-10% weight loss in trials) and 5-amino-1MQ (an NNMT inhibitor targeting fat metabolism) represent emerging alternatives, though they lack FDA approval. The oral non-peptide GLP-1 agonist orforglipron (Eli Lilly) is in Phase 3 trials and could provide another non-injectable option in the future.
How much do GLP-1 medications cost without insurance?
Without insurance, list prices for GLP-1 medications can exceed $1,000/month. However, manufacturer direct programs have significantly reduced cash-pay costs. Novo Nordisk offers Ozempic and Wegovy injectable at $199/month for the first two fills and $299-$349/month ongoing, with oral Wegovy at $149/month for lower doses. Eli Lilly's LillyDirect offers Zepbound at $299-$449/month depending on dose. Generic liraglutide (the active ingredient in Victoza/Saxenda) is available at $200-$500/month. Discount platforms like GoodRx can further reduce costs at specific pharmacies. Patient assistance programs from both manufacturers provide free medication for qualifying uninsured patients. Medicare negotiated prices bring costs to approximately $245/month with ~$50 copays. The compounded semaglutide market previously offered $100-$300/month pricing, but this option is no longer available for most patients.
Can my doctor prescribe a different GLP-1 if my insurance only covers one specific drug?
Yes, and this is one of the most common reasons for switching between GLP-1 agents. Insurance formularies often prefer one GLP-1 over another based on negotiated pricing. If your plan covers Mounjaro but not Wegovy (or vice versa), your doctor can prescribe the covered medication. All GLP-1 agonists produce meaningful weight loss and glycemic improvement, though the magnitude varies. The switch requires following the standard titration protocol for the new medication - you can't simply convert doses. Your doctor may also file a prior authorization or formulary exception request if there's medical rationale for your preferred medication, such as inadequate response to the formulary-preferred agent or documented intolerance.
What is the difference between 503A and 503B compounding pharmacies?
503A pharmacies are traditional compounding pharmacies that prepare medications for individual patients based on specific prescriptions. They're primarily regulated by state boards of pharmacy, exempt from FDA cGMP requirements, and can't distribute in bulk. 503B outsourcing facilities were created by the 2013 Drug Quality and Security Act after a deadly compounding contamination outbreak. They must register with the FDA, follow cGMP manufacturing standards, undergo FDA inspections, and report adverse events. 503B facilities can produce larger batches without patient-specific prescriptions, distributing to healthcare providers for office use. For GLP-1 medications, 503A pharmacies had a 60-day wind-down period after the shortage resolution, while 503B facilities had 90 days. Currently, 503A pharmacies are the only entities that can compound semaglutide under narrow clinical-need exceptions.
Are there any new GLP-1 drugs expected to come to market soon?
Several promising agents are in late-stage development. Retatrutide (Eli Lilly) is a triple agonist targeting GLP-1, GIP, and glucagon receptors that showed up to 24.2% weight loss in Phase 2 trials - the highest of any anti-obesity drug studied to date. It's currently in Phase 3 trials. Orforglipron (Eli Lilly) is an oral non-peptide GLP-1 agonist that could be manufactured like a traditional small-molecule drug, potentially solving supply chain issues. CagriSema (Novo Nordisk) combines semaglutide with cagrilintide (an amylin analog) for enhanced efficacy. Survodutide (Boehringer Ingelheim) is a dual GLP-1/glucagon agonist in Phase 3. These new entrants should diversify the GLP-1 market, reduce shortage risk, and potentially increase competition that drives down prices.
How can I restart GLP-1 therapy after a treatment gap?
The restart protocol depends on how long you've been off the medication. For gaps of 1-2 weeks, you can generally resume at your previous dose since GLP-1 receptor tolerance is maintained. For 2-4 week gaps, restart one dose step below your previous dose and uptitrate. For 4-8 week gaps, restart two steps below to account for substantial tolerance loss. For gaps over 8 weeks, restart from the beginning of the titration schedule as if you're a new patient. These guidelines are based on pharmacokinetic principles rather than randomized trial data, so individual monitoring is important. Some patients who previously tolerated high doses well may handle faster re-titration (2-week intervals instead of 4), but this should only be done under close clinical supervision.
Will generic semaglutide or tirzepatide become available?
Generic liraglutide (the older GLP-1 agonist) became available in July 2025 from manufacturers including Teva and Meitheal, significantly improving affordability. For semaglutide, initial compound patents begin expiring in some countries around 2026, but formulation and device patents extend protection through the late 2020s and early 2030s. Developing a peptide biosimilar requires extensive analytical characterization and clinical bridging studies, so realistic U.S. availability of generic/biosimilar semaglutide is likely still several years away. Tirzepatide has even longer patent protection. In the meantime, manufacturer direct pricing programs ($199-$449/month) and Medicare negotiated prices (~$245/month) are reducing the cost burden that originally drove demand for compounded alternatives.
References & Clinical Sources
- Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384(11):989-1002. DOI: 10.1056/NEJMoa2032183
- Wilding JPH, Batterham RL, Davies M, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes Obes Metab. 2022;24(8):1553-1564. DOI: 10.1111/dom.14725
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387(4):327-340. DOI: 10.1056/NEJMoa2206038
- Jastreboff AM, Kaplan LM, Frías JP, et al. Triple-hormone-receptor agonist retatrutide for obesity - a phase 2 trial. N Engl J Med. 2023;389(6):514-526. DOI: 10.1056/NEJMoa2301972
- Pi-Sunyer X, Astrup A, Fujioka K, et al. A randomized, controlled trial of 3.0 mg of liraglutide in weight management. N Engl J Med. 2015;372(1):11-22. DOI: 10.1056/NEJMoa1411892
- Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet. 2019;394(10193):121-130. DOI: 10.1016/S0140-6736(19)31149-3
- Astrup A, Breum L, Jensen TJ, et al. Effect of tesofensine on bodyweight loss, body composition, and quality of life in obese patients: a randomised, double-blind, placebo-controlled trial. Lancet. 2008;372(9653):1906-1913. DOI: 10.1016/S0140-6736(08)61525-1
- Lush CW, Grayo L, Bel JS, et al. Tesofensine, a novel antiobesity drug, silences GABAergic hypothalamic neurons. Proc Natl Acad Sci USA. 2024;121(18):e2318841121. DOI: 10.1073/pnas.2318841121
- Neelakantan H, Brightwell CR, Graber TG, et al. Small molecule NNMT inhibitor treats diet-induced obesity in an adipose tissue-specific manner. Biochem J. 2019;476(14):2143-2162. DOI: 10.1042/BCJ20190136
- Heffernan KS, Ranadive SM, Gump BB, et al. The effects of human GH and its lipolytic fragment (AOD9604) on lipid metabolism following chronic treatment in obese mice and beta(3)-AR knock-out mice. Endocrinology. 2001;142(12):5182-5189. DOI: 10.1210/endo.142.12.8522
- Frías JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med. 2021;385(6):503-515. DOI: 10.1056/NEJMoa2107519
- Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375(19):1834-1844. DOI: 10.1056/NEJMoa1607141
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med. 2023;389(24):2221-2232. DOI: 10.1056/NEJMoa2307563
- U.S. Food and Drug Administration. FDA's concerns with unapproved GLP-1 drugs used for weight loss. FDA Drug Safety Communication. Updated 2025.
- U.S. Food and Drug Administration. FDA Drug Shortage Database: Semaglutide injection products. Resolved February 21, 2025.
- Marso SP, Daniels GH, Nissen SE, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes (LEADER trial). N Engl J Med. 2016;375(4):311-322. DOI: 10.1056/NEJMoa1603827
- Westerterp-Plantenga MS, Lemmens SG, Westerterp KR. Dietary protein - its role in satiety, energetics, weight loss and health. Br J Nutr. 2012;108(S2):S105-S112. DOI: 10.1017/S0007114512002589
- Rubino DM, Greenway FL, Khalid U, et al. Effect of weekly subcutaneous semaglutide vs daily liraglutide on body weight in adults with overweight or obesity without diabetes (STEP 8). JAMA. 2022;327(2):138-150. DOI: 10.1001/jama.2021.23619
- Milana F, Mazzeo S, Fazzini P, et al. Weight regain after liraglutide, semaglutide or tirzepatide interruption: a narrative review of randomized studies. J Clin Med. 2025;14(11):3791. DOI: 10.3390/jcm14113791
- Beth Israel Lahey Health Pharmacy Network. GLP-1RA and Dual GLP-1RA/GIP: Conversions and Therapy Gap Management Guide. 2024.
- Garvey WT, Batterham RL, Bhatt DL, et al. Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nat Med. 2022;28(10):2083-2091. DOI: 10.1038/s41591-022-02026-4
- Davies MJ, Bergenstal R, Bode B, et al. Efficacy of liraglutide for weight loss among patients with type 2 diabetes (SCALE Diabetes). JAMA. 2015;314(7):687-699. DOI: 10.1001/jama.2015.9676
- Moré MI, Mischler A, Engel M. Safety and metabolism of AOD9604, a novel nutraceutical ingredient for improved metabolic health. J Endocrinol Metab. 2016;6(2):46-54. DOI: 10.14740/jem340w
- Nauck MA, Quast DR, Wefers J, et al. GLP-1 receptor agonists in the treatment of type 2 diabetes - state-of-the-art. Mol Metab. 2021;46:101102. DOI: 10.1016/j.molmet.2020.101102
- Kadowaki T, Isendahl J, Khalid U, et al. Semaglutide once a week in adults with overweight or obesity, with or without type 2 diabetes in an east Asian population (STEP 6). Lancet Diabetes Endocrinol. 2022;10(3):193-206. DOI: 10.1016/S2213-8587(22)00008-0
- Drug Quality and Security Act (DQSA), Pub. L. 113-54 (2013). Sections 503A and 503B of the Federal Food, Drug, and Cosmetic Act.
- American Diabetes Association. Standards of Care in Diabetes - 2025. Diabetes Care. 2025;48(Suppl 1). DOI: 10.2337/dc25-SINT
- Novo Nordisk. Annual Report 2025: Capital expenditure and manufacturing expansion plans. Copenhagen, Denmark. 2026.
- Dutta D, Surana V, Singla R, et al. Addressing the shortage of GLP-1 RA and dual GIP/GLP-1 RA-based therapies - a systematic review. Endocrines. 2025;6(6):52. DOI: 10.3390/endocrines6060052
- Wadden TA, Bailey TS, Billings LK, et al. Effect of subcutaneous semaglutide vs placebo as an adjunct to intensive behavioral therapy on body weight in adults with overweight or obesity (STEP 3). JAMA. 2021;325(14):1403-1413. DOI: 10.1001/jama.2021.1831
- Rosenstock J, Wysham C, Frías JP, et al. Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1). Lancet. 2021;398(10295):143-155. DOI: 10.1016/S0140-6736(21)01324-6
- FDA. FDA alerts healthcare providers, compounders and patients: dosing errors associated with compounded semaglutide products. FDA Safety Communication. July 2024.
- Crasto W, Khunti K, Davies MJ. Discontinuation and reinitiation of dual-labeled GLP-1 receptor agonists among US adults with overweight or obesity. JAMA Netw Open. 2025;8(1):e2454789. DOI: 10.1001/jamanetworkopen.2024.54789
- Global rise of compounded weight-loss medicines: a worrisome trend. Ann Med. 2025;57(1):2502376. DOI: 10.1080/07853890.2025.2502376
- Navigating access: the future of compounded GLP-1 receptor agonists for weight loss. J Obes Metab Syndr. 2025;34(3):e2025027. PMC12331335
- Frontiers in Endocrinology. A glimpse into the pipeline of anti-obesity medication development: combining multiple receptor pathways. Front Endocrinol. 2025;16:1630199. DOI: 10.3389/fendo.2025.1630199
- Rebound or retention: a meta-analysis of weight regain after the discontinuation of glucagon-like peptide-1 (GLP-1) receptor agonists and other anti-obesity drugs. Cureus. 2025;17(4):e82376. PMC12535773