Executive Summary
Figure 1: GLP-1 receptor agonists and the gastrointestinal system, where the majority of side effects originate
Key Takeaways
- Nausea is the #1 side effect (20-44% of patients), but typically resolves within 4-8 weeks at each dose level
- Slow, protocol-driven dose titration is the single most effective strategy for minimizing GI side effects
- Pancreatitis risk has not been confirmed in large-scale trials - rates are similar to placebo
- The thyroid cancer warning is based on rodent data; human evidence does not support increased MTC risk
- Gallbladder events increase with rapid weight loss, not uniquely with GLP-1 drugs
GLP-1 receptor agonists are among the most effective weight loss and diabetes medications available today, but they come with a well-characterized side effect profile that every patient and prescriber needs to understand. This guide covers everything from the common gastrointestinal complaints to the rare but serious complications, with practical strategies for managing each one.
The GLP-1 drug class - which includes semaglutide (Ozempic, Wegovy), tirzepatide (Mounjaro, Zepbound), liraglutide (Victoza, Saxenda), dulaglutide (Trulicity), and exenatide (Byetta, Bydureon) - has transformed the treatment of type 2 diabetes and obesity. But these drugs work by activating receptors throughout the body, not just in the pancreas. And wherever those receptors exist, there's potential for effects both wanted and unwanted.
Gastrointestinal side effects dominate the picture. Nausea affects between 20% and 44% of patients, depending on the specific agent and dose. Vomiting, diarrhea, and constipation are also common. These symptoms are usually temporary - most patients see significant improvement within the first 4 to 8 weeks at each dose level. But for some, the GI burden is severe enough to warrant dose reduction or even discontinuation.
Beyond the GI tract, there are rarer but more serious concerns to consider. Questions about pancreatitis risk have followed the class since its earliest days. The boxed warning about medullary thyroid carcinoma persists on every label, rooted in rodent data that may not translate to humans. Gallbladder disease rates increase during rapid weight loss. And emerging discussions around gastroparesis, muscle loss, and mental health effects deserve careful analysis.
This report synthesizes data from the major clinical trial programs - SUSTAIN, STEP, SURPASS, SURMOUNT, LEADER, and others - alongside post-marketing surveillance data and real-world evidence. We'll break down the incidence rates for every major side effect, compare them across drugs and doses, and provide actionable management strategies. The GLP-1 research hub provides additional context on the broader clinical evidence base.
Whether you're a patient starting your first GLP-1 prescription, a clinician advising on tolerability, or a researcher evaluating the safety profile of this drug class, this guide aims to give you the most thorough, evidence-based resource available. No side effect is too minor to discuss, and no concern is too rare to address. The goal is informed decision-making - because understanding what might happen and knowing how to handle it makes the treatment journey far more manageable.
Key Takeaways
- Nausea is the #1 side effect (20-44% of patients), but typically resolves within 4-8 weeks at each dose level
- Slow, protocol-driven dose titration is the single most effective strategy for minimizing GI side effects
- Pancreatitis risk has not been confirmed in large-scale trials - rates are similar to placebo
- The thyroid cancer warning is based on rodent data; human evidence does not support increased MTC risk
- Gallbladder events increase with rapid weight loss, not uniquely with GLP-1 drugs
- Most side effects are manageable with proper titration, dietary modifications, and supportive care
This guide is structured to address each side effect category in depth, moving from the most common to the most rare. Each section includes incidence data, proposed mechanisms, risk factors, management strategies, and clear guidance on when to seek medical attention. The dosing calculator can help ensure you're following the recommended titration schedules that minimize side effects.
How This Guide Is Organized
We've structured this report to follow a practical priority order. We start with the GI side effects that nearly every patient encounters, because these dominate the real-world treatment experience. Then we move through the nausea management section - the most actionable part of this guide - before addressing the specific safety concerns that generate the most questions: pancreatitis, thyroid, gallbladder, and gastroparesis. The later sections cover muscle loss, mental health, and rare complications. The FAQ section addresses the questions patients and providers ask most frequently, and the references section provides the primary literature citations for every major claim in this report.
For patients who are currently experiencing side effects and need immediate guidance, skip directly to the Nausea Management Strategies section. For clinicians evaluating whether to initiate or continue GLP-1 therapy in a specific patient, the Side Effect Rates by Drug & Dose section provides the comparative data you need. And for anyone seeking to understand the less common but more serious safety signals, the pancreatitis, thyroid, and gallbladder sections provide thorough evidence reviews.
Throughout this guide, we reference our other reports in the GLP-1 research library, including the comprehensive guides to semaglutide, tirzepatide, liraglutide, and GLP-1 weight loss mechanisms. These resources provide additional depth on specific compounds and pathways that are only summarized here.
GI Side Effects: Nausea, Vomiting, Diarrhea
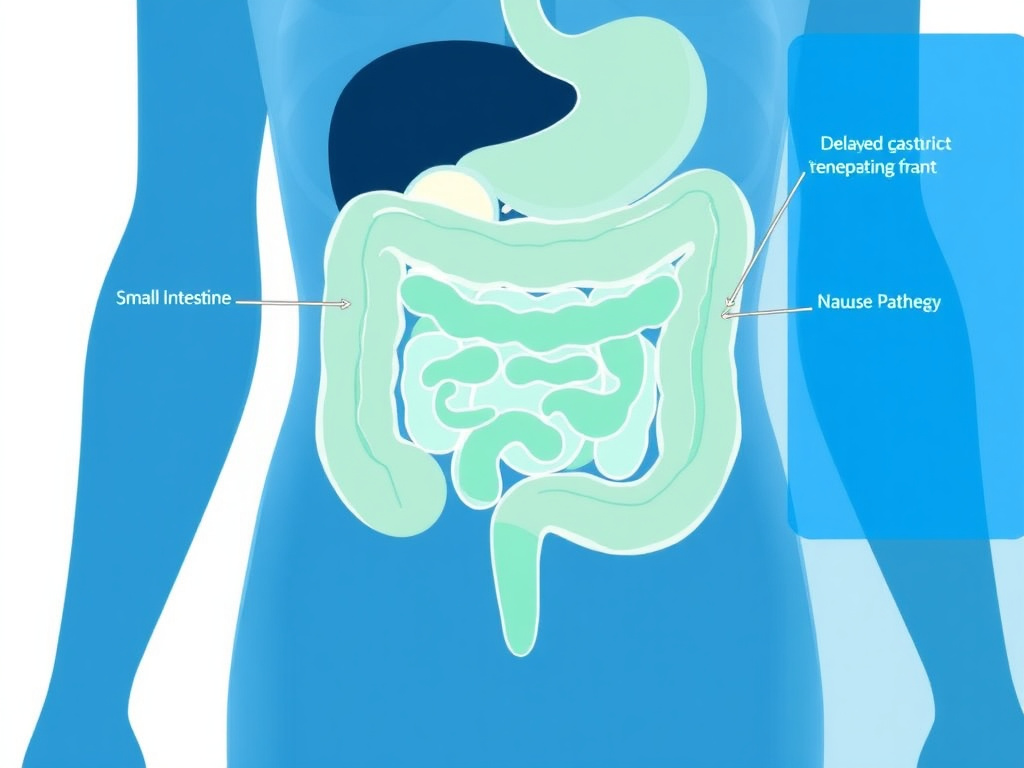
Figure 2: GLP-1 receptor activation affects the entire gastrointestinal tract, from delayed gastric emptying to altered intestinal motility
Gastrointestinal side effects are the hallmark of GLP-1 receptor agonist therapy. They're the most common reason patients consider stopping treatment, the primary driver of dose titration protocols, and the area where practical management strategies make the biggest difference. Understanding why these effects occur and how they evolve over time is essential for anyone taking or prescribing these medications.
Why GLP-1 Drugs Cause GI Symptoms
GLP-1 receptors are densely expressed throughout the gastrointestinal system. When a GLP-1 agonist binds to these receptors, it triggers several physiological responses that contribute to both the drug's therapeutic effects and its side effects. The line between "benefit" and "side effect" is often blurry here - delayed gastric emptying, for instance, is both a key weight loss mechanism and the primary driver of nausea.
The stomach is ground zero. GLP-1 receptor activation relaxes the proximal stomach and inhibits antral contractions, slowing the rate at which food moves from the stomach into the duodenum. This delay can range from 20% to 40% compared to baseline, depending on the dose and agent. The result is a prolonged sense of fullness after meals, which is therapeutically desirable, but it also creates the conditions for nausea and, when severe, vomiting.
The vagus nerve plays a central role. GLP-1 agonists activate vagal afferent neurons that project to the nucleus tractus solitarius (NTS) and the area postrema in the brainstem. The area postrema sits outside the blood-brain barrier, making it particularly sensitive to circulating GLP-1 levels. This is the brain's "vomiting center," and its activation is why nausea can feel so overwhelming during the early weeks of treatment. Our guide on GLP-1 weight loss mechanisms covers these neural pathways in greater depth.
In the small and large intestine, GLP-1 receptor activation alters motility patterns. Some patients experience diarrhea due to changes in fluid secretion and transit time. Others develop constipation, likely because the overall slowing of GI transit predominates. The intestinal effects tend to be less predictable than the gastric ones, varying considerably between individuals and even between different GLP-1 agents.
Nausea: The Most Common Complaint
Nausea is the single most reported adverse event across all GLP-1 receptor agonist trials. It's not a subtle queasiness for many patients - it can be a persistent, debilitating sensation that interferes with daily life, especially during the first weeks at each new dose. In the STEP 1 trial of semaglutide 2.4 mg, 44.2% of participants in the treatment group reported nausea at some point during the 68-week study, compared to 17.4% in the placebo group (Wilding JPH, Batterham RL, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. New England Journal of Medicine. 2021;384(11):989-1002. DOI: 10.1056/NEJMoa2032183).
The temporal pattern of nausea is critical to understand. It typically peaks during the first 1-2 weeks at each new dose level, then gradually diminishes over the next 2-6 weeks. In the STEP trials, the proportion of patients experiencing nausea at any given week was highest during the dose escalation phase and lowest during the maintenance phase. By week 20, the rates of active nausea were substantially lower than during the first month.
What makes some patients more susceptible to nausea? Several factors contribute. Women report nausea more frequently than men across all GLP-1 trials. Patients with pre-existing gastric conditions, including reflux disease and functional dyspepsia, tend to have more GI symptoms. Higher starting BMI is paradoxically associated with somewhat lower nausea rates in some analyses, possibly because the gastric effects are relatively less impactful in larger individuals. And the specific agent matters - as we'll detail in the next section, nausea rates vary from 21% with dulaglutide to 44% with subcutaneous semaglutide at the highest approved weight management doses.
Vomiting: When Nausea Escalates
Vomiting occurs in approximately 5-25% of GLP-1 users, depending on the drug, dose, and study design. It follows the same temporal pattern as nausea, peaking during dose titration and declining during maintenance. In STEP 1, 24.8% of semaglutide-treated patients reported vomiting versus 6.1% on placebo. In SURPASS-2 with tirzepatide, the rates ranged from 5.8% at the 5 mg dose to 9.4% at the 15 mg dose (Frias JP, Davies MJ, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. New England Journal of Medicine. 2021;385(6):503-515. DOI: 10.1056/NEJMoa2107519).
Persistent vomiting is a clinical red flag. While occasional vomiting during dose escalation is expected, frequent vomiting (more than 3-4 episodes per week) that doesn't improve after 2 weeks at the same dose warrants medical attention. It can lead to dehydration, electrolyte imbalances, and in rare cases, acute kidney injury - particularly in patients taking diuretics, ACE inhibitors, or NSAIDs concurrently.
Diarrhea and Constipation
Diarrhea affects 8-20% of GLP-1 users across trials. It tends to be mild to moderate, episodic rather than continuous, and most common during the first few weeks at each new dose. The mechanism involves altered intestinal motility and changes in fluid secretion patterns. For most patients, dietary modifications - reducing high-fat and high-fiber foods temporarily - are sufficient to manage it.
Constipation is less discussed but nearly as common, affecting 5-15% of patients. The overall slowing of GI transit can produce hard stools, infrequent bowel movements, and a sense of incomplete evacuation. Adequate hydration, fiber intake (after the initial adjustment period), and gentle osmotic laxatives can help. Patients using semaglutide at higher doses report constipation somewhat more frequently than those on tirzepatide, though head-to-head comparison data are limited.
Other GI Symptoms
Abdominal pain, bloating, dyspepsia, flatulence, and gastroesophageal reflux are all reported at elevated rates in GLP-1 trials. Abdominal pain occurs in roughly 5-10% of users and can range from mild cramping to more substantial discomfort. Bloating and excessive gas are common during the adjustment period, likely related to altered fermentation patterns as food transit times change.
Decreased appetite - which is technically a therapeutic effect - can itself be distressing when it's extreme. Some patients report food aversion, where the sight or smell of food triggers nausea. This usually resolves as the body adapts, but during the acute phase, it can contribute to inadequate nutritional intake and should be monitored.
Clinical Pearl
The GI side effects of GLP-1 drugs are a direct consequence of their mechanism of action. You can't fully eliminate them without eliminating the therapeutic effect. But you can minimize their severity and duration through proper dose titration, dietary modification, and timing strategies. The patients who do best are the ones who prepare for these effects and have a management plan in place before they start treatment.
Discontinuation Due to GI Effects
How often do GI side effects lead patients to stop treatment? In the STEP trial program, the discontinuation rate due to adverse events was approximately 7% for semaglutide versus 3% for placebo, with GI symptoms being the most common reason. In SURPASS/SURMOUNT with tirzepatide, discontinuation rates ranged from 3% to 7% depending on dose. These numbers are lower than many patients expect, suggesting that most people who experience GI symptoms can manage them well enough to continue treatment.
The key factor in whether a patient continues or stops is usually the first 4-6 weeks. If someone can get through the initial adjustment period with adequate support and management strategies, the likelihood of long-term continuation increases dramatically. This is where proper counseling, realistic expectations, and proactive nausea management pay dividends.
The Temporal Pattern: A Week-by-Week Look
Understanding the typical timeline of GI symptoms helps set realistic expectations. Here's what the data show about the natural history of GI side effects during a standard dose escalation with semaglutide:
Weeks 1-4 (0.25 mg): This is the gentlest introduction to the drug. Many patients experience no symptoms at all. Those who do typically report mild nausea, particularly in the 24-48 hours following the injection. Appetite suppression begins, and some patients notice a reduction in portion sizes. Diarrhea or constipation may occur but are usually mild.
Weeks 5-8 (0.5 mg): The first real test for many patients. Nausea becomes more noticeable, often peaking 12-36 hours post-injection. Some patients develop a pattern where day 1 and day 2 after injection are the toughest, with gradual improvement through the rest of the week. Vomiting may occur for the first time. By week 7-8, most patients report that the symptoms are starting to fade at this dose level.
Weeks 9-12 (1.0 mg): Another adjustment period as the dose increases. Patients who tolerated 0.5 mg well may find 1.0 mg brings back the nausea they thought they'd conquered. The good news: they typically adapt faster this time. The body seems to partially "remember" its adaptation, and the adjustment period at 1.0 mg is often shorter than it was at 0.5 mg.
Weeks 13-16 (1.7 mg): For patients on the weight management titration, this step can be challenging. The GI burden is increasing with each dose, and cumulative frustration can set in. This is the stage where proactive nausea management strategies and clinical support matter most. Patients who reach this level successfully usually complete the full titration.
Weeks 17-20+ (2.4 mg maintenance): The final dose increase brings another wave of GI symptoms in some patients. But after weeks to months at the maintenance dose, the body's adaptation is usually substantial. By month 4-6 at 2.4 mg, active nausea rates drop to single digits, though a small percentage of patients continue to experience intermittent symptoms long-term.
GI Side Effects in Special Populations
Older Adults (Age 65+)
Elderly patients may experience more pronounced GI side effects and tolerate them less well. Reduced gastric motility at baseline, decreased renal clearance (leading to higher drug levels), and increased susceptibility to dehydration all contribute. Slower titration schedules and closer monitoring of hydration and nutritional status are recommended. The risk of acute kidney injury from dehydration is also elevated in this population. For patients using peptides like BPC-157 for gut support, coordination with their GLP-1 prescriber is advisable.
Patients with Pre-Existing GI Conditions
Individuals with irritable bowel syndrome (IBS), inflammatory bowel disease (IBD), gastroesophageal reflux disease (GERD), or functional dyspepsia are at higher risk of exacerbated GI symptoms on GLP-1 therapy. These patients should start at the lowest possible dose, use extended titration schedules, and may need concurrent GI-specific therapies. Some patients with GERD report worsening reflux symptoms, likely due to delayed gastric emptying and increased gastric volume.
Post-Bariatric Surgery Patients
Patients who have undergone bariatric surgery (gastric bypass, sleeve gastrectomy) present unique challenges. Their altered GI anatomy may amplify the effects of delayed gastric emptying and altered motility. The smaller gastric pouch after sleeve gastrectomy is particularly susceptible to the volume-expanding effects of slowed emptying. GLP-1 therapy in post-bariatric patients should be approached with caution and close monitoring.
Drug Interactions That Worsen GI Symptoms
Several commonly co-prescribed medications can compound GLP-1-related GI symptoms:
- Metformin: Also causes GI side effects (nausea, diarrhea) in 20-30% of users. The combination can be complementary in terms of GI burden. Consider the extended-release formulation of metformin, which has a better GI profile.
- Orlistat: Causes oily stool, fecal urgency, and flatulence. Combining with a GLP-1 agonist creates a compounded GI symptom load that most patients find intolerable.
- Antibiotics (erythromycin class): Macrolide antibiotics are prokinetic agents that speed gastric emptying, opposite to GLP-1's effect. This can create confusing, fluctuating symptoms.
- Opioids: Slow GI motility and can worsen constipation when combined with GLP-1 drugs.
- NSAIDs: Can irritate the gastric mucosa, adding GI discomfort on top of GLP-1-related symptoms.
Side Effect Rates by Drug & Dose
Figure 3: Nausea incidence varies considerably across GLP-1 agents, from 21% with dulaglutide to 44% with subcutaneous semaglutide
Not all GLP-1 receptor agonists cause side effects at the same rates. The specific drug, the dose, the formulation (subcutaneous vs. oral), and the titration schedule all influence what patients experience. This section presents the comparative data so you can make informed decisions about which agent might offer the best tolerability profile for your situation.
Nausea Incidence (%) Across GLP-1 Agents
Semaglutide (Subcutaneous): The Highest Nausea Rates
Subcutaneous semaglutide has the highest reported nausea rate among the approved GLP-1 agonists. In the STEP 1 trial (2.4 mg weekly for weight management), 44.2% of patients reported nausea. In the SUSTAIN trials (0.5-1.0 mg for diabetes), nausea rates were lower at roughly 15-20%, reflecting the lower doses used. This dose-response relationship is a consistent finding across the entire class - higher doses produce more GI symptoms.
The vomiting rate with semaglutide 2.4 mg was 24.8% in STEP 1, which is considerably higher than other agents at their approved doses. Diarrhea was reported in 30%, constipation in 24%, and abdominal pain in about 7%. These are "any time during the study" rates, meaning a patient who had one episode of mild nausea in week 2 and then no further symptoms counts the same as someone who experienced persistent nausea for months.
Tirzepatide: Dual Agonist, Different Profile
Tirzepatide, a dual GIP/GLP-1 receptor agonist, showed nausea rates of 12-33% across its dose range in the SURPASS and SURMOUNT programs. At the 5 mg dose, nausea was reported in about 12-15% of patients. At 10 mg, rates increased to roughly 18-24%. And at the 15 mg dose, nausea reached 27-33% (Jastreboff AM, Aronne LJ, et al. Tirzepatide Once Weekly for the Treatment of Obesity. New England Journal of Medicine. 2022;387(3):205-216. DOI: 10.1056/NEJMoa2206038).
The GIP receptor component of tirzepatide may actually help mitigate nausea to some degree. GIP receptor activation has been shown in preclinical models to have some anti-emetic properties, which could explain why tirzepatide's nausea rates are generally lower than semaglutide's at doses producing comparable weight loss. For a detailed comparison, see our complete tirzepatide guide.
Liraglutide: The Established Agent
Liraglutide at the 3.0 mg daily dose (Saxenda) showed nausea rates of approximately 39% in the SCALE Obesity and Prediabetes trial. At the lower diabetes dose (1.8 mg, Victoza), nausea was reported in roughly 25-30% of patients. Vomiting occurred in about 15% at the 3.0 mg dose, and diarrhea in approximately 21% (Pi-Sunyer X, Astrup A, et al. A Randomized, Controlled Trial of 3.0 mg of Liraglutide in Weight Management. New England Journal of Medicine. 2015;373(1):11-22. DOI: 10.1056/NEJMoa1411892).
Liraglutide's daily dosing (versus weekly for semaglutide and tirzepatide) means patients experience more frequent but potentially smaller fluctuations in drug levels. Some clinicians believe this leads to a more gradual adaptation, though the overall nausea rates don't clearly support this theory. Our liraglutide guide covers its specific tolerability profile in greater detail.
Dulaglutide: The Most Tolerable?
Dulaglutide (Trulicity) consistently shows the lowest nausea rates in its class. In the AWARD and REWIND trials, nausea was reported in approximately 12-21% of patients depending on the dose. Even at the highest approved dose (4.5 mg weekly), nausea rates stayed below 25%. Vomiting rates were 5-12%, and diarrhea was reported in approximately 8-15%. For more details, refer to our dulaglutide guide.
The lower GI symptom burden with dulaglutide comes at a cost - its weight loss efficacy is generally lower than semaglutide or tirzepatide. This illustrates a broader pattern in the GLP-1 class: there's a rough correlation between GI tolerability and weight loss efficacy. The drugs that produce the most weight loss also tend to cause the most GI symptoms.
Exenatide: The Pioneer
Exenatide, the first approved GLP-1 agonist, comes in two formulations. The twice-daily formulation (Byetta) showed nausea rates of approximately 40-44% in clinical trials, comparable to semaglutide at weight management doses. The extended-release once-weekly formulation (Bydureon) had somewhat lower nausea rates at 20-26%, along with a unique issue: injection site nodules due to the microsphere delivery system. See our exenatide guide for the complete profile.
Comparative Side Effect Table
| Side Effect | Semaglutide 2.4mg | Tirzepatide 15mg | Liraglutide 3.0mg | Dulaglutide 4.5mg | Exenatide ER |
|---|---|---|---|---|---|
| Nausea | 44% | 33% | 39% | 21% | 26% |
| Vomiting | 25% | 10-12% | 15% | 8-12% | 10-11% |
| Diarrhea | 30% | 17-21% | 21% | 8-15% | 13-18% |
| Constipation | 24% | 11-13% | 19% | 5-8% | 6-11% |
| Abdominal Pain | 7% | 5-7% | 5% | 4-6% | 5-8% |
| Discontinuation (GI) | 4.5% | 3-6% | 6.4% | 1.5-3% | 3-5% |
Important Context
Side effect rates from clinical trials represent "any occurrence during the study period" and include mild, transient symptoms. The percentage of patients experiencing persistent or severe symptoms is substantially lower. In STEP 1, for example, while 44% reported nausea at some point, only about 3% reported nausea severe enough to lead to treatment discontinuation.
Emerging Agents: Retatrutide and Orforglipron
Next-generation GLP-1 receptor agonists currently in late-stage trials are reporting broadly similar GI profiles. Retatrutide, a triple agonist (GIP/GLP-1/glucagon), showed nausea rates of 22-47% in the Phase 2 trial, with higher rates at the 12 mg dose. The oral non-peptide GLP-1 agonist orforglipron showed nausea rates of 14-40% depending on the dose, with the advantage that dose titration flexibility may be easier with an oral formulation. Our guides on retatrutide and orforglipron cover these emerging agents in detail.
The Efficacy-Tolerability Trade-Off
A consistent pattern emerges across the GLP-1 class: the drugs that produce the most weight loss also tend to cause the most GI side effects. This isn't coincidental - the GI effects (particularly appetite suppression from central and peripheral mechanisms) are themselves key drivers of weight loss. You can't fully separate the benefit from the burden.
This creates a practical clinical decision. Do you prioritize maximum efficacy (semaglutide 2.4 mg, tirzepatide 15 mg) and accept a higher GI side effect burden, or do you choose a more tolerable agent (dulaglutide, lower-dose tirzepatide) and accept somewhat less weight loss? The answer depends on the individual patient's goals, their tolerance for side effects, and how well they respond to management strategies.
For patients whose primary goal is glycemic control in type 2 diabetes, even the lower-efficacy agents produce excellent A1c reductions with a milder side effect profile. For patients seeking maximum weight loss for severe obesity with comorbidities, the higher-dose agents may be worth the more intense adjustment period. And for patients who can't tolerate one agent, switching to another in the same class often reveals a very different tolerability profile, even when the drugs have similar mechanisms of action.
Oral vs. Injectable Formulations
Oral semaglutide (Rybelsus) provides an interesting comparison point. At the 14 mg daily oral dose, nausea rates are approximately 15-20%, lower than the injectable weight management dose but still significant. The oral formulation requires specific dosing conditions - taken on an empty stomach with no more than 4 oz of plain water, 30 minutes before any other food or drink - that can themselves be challenging for patients with busy morning routines.
The upcoming oral non-peptide GLP-1 agonists like orforglipron may offer improved convenience and potentially different tolerability profiles. Because they're not peptides, they don't require the same fasting conditions as oral semaglutide. Their dose-response curves for both efficacy and side effects are still being characterized in Phase 3 trials. For updates on emerging oral options, check the GLP-1 research hub.
CagriSema: The Next Frontier
CagriSema combines semaglutide with cagrilintide (an amylin analog) in a single injection. The Phase 3 REDEFINE program is evaluating this combination for obesity, with early data showing weight loss of approximately 22-25% - rivaling bariatric surgery. GI side effect rates in Phase 2 were broadly similar to semaglutide alone, though the amylin component adds its own GI effects profile. Our CagriSema guide covers this combination in detail.
Nausea Management Strategies

Figure 4: Practical nausea management combines dietary modifications, timing strategies, and natural remedies
Managing nausea on GLP-1 therapy is part science, part strategy, and part patience. The good news: most patients find that nausea improves significantly within weeks if they take a systematic approach. This section provides a step-by-step framework that draws from clinical guidelines, patient experience data, and practical nutritional science. These aren't vague suggestions - they're specific, actionable tactics that can make a real difference in your day-to-day experience.
Strategy #1: Follow the Titration Schedule Exactly
The single most effective way to reduce nausea is to follow your prescribed dose titration schedule without skipping steps. Every GLP-1 drug has a recommended escalation protocol specifically designed to give your body time to adapt. With semaglutide, the standard titration is: 0.25 mg for 4 weeks, then 0.5 mg for 4 weeks, then 1.0 mg for 4 weeks, then 1.7 mg for 4 weeks, then the target dose of 2.4 mg. Each step matters.
Patients who try to rush the titration - moving up after just 1-2 weeks instead of 4, or skipping doses entirely - experience significantly worse nausea. If you're tolerating a dose poorly, stay at that level for an additional 2-4 weeks rather than moving up on schedule. Your provider can customize the titration based on your response. Some patients do best with even slower escalation: spending 6-8 weeks at each level rather than 4. Use the dosing calculator to track your titration timeline.
Strategy #2: Meal Size and Frequency
Large meals are the enemy when you're adjusting to a GLP-1 drug. Your stomach is now emptying more slowly, so filling it to capacity creates a prolonged sensation of uncomfortable fullness that easily tips into nausea. Switch to 4-6 smaller meals per day instead of 3 large ones. Each meal should be roughly the size of your fist or smaller.
Stop eating before you feel full. This sounds counterintuitive, but on a GLP-1 agonist, the sensation of fullness arrives with a delay, then intensifies as your stomach holds food longer than it used to. If you eat until you're satisfied, you'll be uncomfortably full 20 minutes later. Aim to eat until you're about 70% satisfied, then stop.
Strategy #3: Dietary Composition Matters
High-fat foods are the worst offenders for triggering nausea on GLP-1 therapy. Fat naturally slows gastric emptying, and layering that on top of the drug-induced gastric delay creates a double hit that your stomach can't handle. During the first 2-4 weeks at each new dose, focus on lean proteins, simple carbohydrates, and non-greasy preparations.
Foods that tend to be well-tolerated during the adjustment period:
- Lean proteins: grilled chicken, turkey, white fish, egg whites, plain Greek yogurt
- Simple carbs: plain rice, toast, saltine crackers, pretzels, bananas, applesauce
- Vegetables: steamed or roasted, with minimal added oil or butter
- Broth-based soups: chicken broth, miso soup, clear vegetable soup
Foods to minimize or avoid during adjustment:
- Fried and greasy foods: french fries, fried chicken, pizza, burgers
- Heavy cream sauces, cheese-heavy dishes, buttery preparations
- Very spicy foods (individual tolerance varies)
- Large amounts of raw vegetables (can cause bloating)
- Carbonated beverages (contribute to bloating and gas)
- Alcohol (worsens nausea and delays gastric emptying further)
Strategy #4: Injection Timing
When you take your injection can influence when nausea peaks. Many patients find that injecting in the evening - ideally a few hours before bedtime - allows the initial nausea surge to occur during sleep. By morning, the worst of the acute reaction has passed. Others prefer morning injections so they can manage symptoms with food and activity throughout the day. There's no single right answer - experiment during your first month to find your optimal timing.
For weekly injections (semaglutide, tirzepatide, dulaglutide), pick the same day each week. Friday evening works well for many people, as mild symptoms over the weekend are easier to manage than during a demanding workweek. For daily liraglutide, the timing flexibility is even greater.
Strategy #5: Natural Anti-Nausea Remedies
Ginger is the most evidence-supported natural anti-nausea agent. Multiple randomized trials support its efficacy for nausea of various causes, including chemotherapy-induced and pregnancy-related nausea. For GLP-1-related nausea, try:
- Ginger tea: steep fresh ginger slices in hot water for 5-10 minutes
- Ginger chews or candies (look for products with real ginger, not just ginger flavoring)
- Ginger supplements: 250 mg capsules, 1-2 capsules up to 4 times daily
- Pickled ginger (the kind served with sushi)
Peppermint is another helpful option. Peppermint tea, peppermint oil capsules (enteric-coated), or simply inhaling peppermint essential oil can reduce nausea. The menthol in peppermint has a direct relaxant effect on the gastric smooth muscle. Smelling rubbing alcohol (isopropyl alcohol) on a cotton pad has also been shown in emergency department studies to reduce nausea rapidly - it's a surprisingly effective trick.
Strategy #6: Hydration, Hydration, Hydration
Dehydration makes nausea worse, and nausea makes dehydration more likely. Break this cycle by sipping fluids constantly throughout the day. Don't gulp large amounts at once - that will contribute to gastric distension. Small, frequent sips work best. Aim for at least 64 ounces daily. Cold or room-temperature water is generally better tolerated than hot beverages during active nausea. Adding a small amount of electrolyte powder or drinking diluted sports drinks can help maintain electrolyte balance, especially if vomiting has occurred.
Strategy #7: When Lifestyle Measures Aren't Enough
If dietary modifications, timing adjustments, and natural remedies don't adequately control nausea, your provider has pharmaceutical options:
- Ondansetron (Zofran): A 5-HT3 receptor antagonist originally developed for chemotherapy-induced nausea. Highly effective for GLP-1-related nausea. Available in oral, sublingual (dissolving), and IV formulations. Typical dose is 4-8 mg as needed. The dissolving tablet is ideal because it doesn't require swallowing with water.
- Promethazine (Phenergan): An antihistamine with anti-emetic properties. Causes drowsiness, so it's best for evening use. 12.5-25 mg as needed.
- Metoclopramide (Reglan): A prokinetic that speeds gastric emptying - the opposite of what the GLP-1 drug does. This seems counterintuitive, but it can be useful for patients with very severe gastric stasis symptoms. Use should be short-term due to the risk of tardive dyskinesia.
- Meclizine (Antivert): An antihistamine useful when nausea has a vestibular component (dizziness-associated nausea). Available over-the-counter.
When to Call Your Doctor
Seek medical attention if you experience: vomiting more than 3 times in 24 hours, inability to keep any fluids down for more than 12 hours, signs of dehydration (dark urine, dizziness, rapid heartbeat), severe abdominal pain that doesn't resolve, or nausea so severe that you can't perform daily activities after more than 2 weeks at the same dose. These may indicate the need for dose reduction, temporary drug holiday, or evaluation for other causes.
Strategy #8: The Extended Slow Titration
For patients who are particularly sensitive to GI effects, an extended slow titration protocol can make all the difference. Instead of the standard 4-week intervals, spend 6-8 weeks at each dose level. Some providers even use intermediate doses - for example, alternating between 0.25 mg and 0.5 mg weekly for 2-4 weeks before moving fully to 0.5 mg. While this approach means reaching the target dose takes longer, the trade-off is dramatically better tolerability.
Another option for patients struggling with the jump from one dose to the next: dose back down to the previous well-tolerated dose for 2 weeks, then try the higher dose again. Sometimes the second attempt at a particular dose level is much better tolerated than the first, as the body has had additional time to adapt to the drug's presence.
Strategy #9: Position and Activity After Eating
Don't lie down immediately after eating. Stay upright for at least 30-60 minutes after meals. Gentle walking (10-15 minutes) after eating can help stimulate gastric motility and reduce the sensation of fullness. Avoid vigorous exercise within an hour of meals, however, as intense physical activity can worsen nausea when the stomach is full and emptying slowly.
Some patients find that wearing loose clothing around the waist helps, as tight waistbands can increase intra-abdominal pressure and contribute to both nausea and reflux symptoms. Small changes in posture and positioning can make a surprisingly large difference in comfort level.
Strategy #10: Acupressure and Mind-Body Techniques
The P6 (Neiguan) acupressure point on the inner wrist has been studied for nausea relief across multiple contexts, including post-operative nausea, pregnancy nausea, and motion sickness. Sea-Band wristbands, which apply continuous pressure to this point, are inexpensive and drug-free. While the evidence for GLP-1-specific nausea is limited, many patients report subjective benefit, and there's essentially no downside to trying.
Deep breathing exercises can help during acute nausea episodes. Slow, controlled diaphragmatic breathing - inhaling for 4 counts, holding for 4, exhaling for 8 - activates the parasympathetic nervous system and can reduce the intensity of nausea. Guided meditation apps focused on nausea relief are another tool some patients find helpful, particularly for the anxiety component that often accompanies persistent nausea.
Strategy #11: The "Nausea Toolkit" Approach
The most successful patients prepare a literal nausea toolkit before starting their GLP-1 medication. Having supplies on hand when nausea strikes eliminates the miserable experience of trying to shop or prepare things while feeling terrible. Your toolkit should include:
- Ginger tea bags or fresh ginger root
- Ginger chews or candies (Gin Gins, Reed's, or similar)
- Peppermint tea bags
- Saltine or plain crackers
- Electrolyte packets (Liquid IV, Pedialyte, or similar)
- Sea-Bands or acupressure wristbands
- A prescription for ondansetron (ask your prescriber proactively)
- Small Tupperware containers for pre-portioned mini meals
- A water bottle you can sip from throughout the day
A Sample Day During Dose Escalation
Here's what a well-managed day might look like during the first week at a new dose level:
7:00 AM - Wake up, sip warm ginger tea slowly. Eat a few crackers if tolerated.
8:00 AM - Small breakfast: 2 scrambled egg whites on a piece of dry toast, small glass of water.
10:30 AM - Mini snack: 1/2 cup plain Greek yogurt or a handful of pretzels. Continue sipping water.
12:30 PM - Small lunch: 3-4 oz grilled chicken breast, 1/2 cup plain rice, steamed vegetables. Eat slowly, stop before full.
3:00 PM - Mini snack: banana, small protein shake (20g protein).
5:30 PM - Small dinner: baked white fish, small portion of mashed potato (minimal butter), steamed green beans.
7:30 PM - Light evening snack if hungry: applesauce, a few crackers, or peppermint tea.
8:00 PM - If this is injection day (weekly medications), take injection now.
9:00 PM - Peppermint tea or ginger tea before bed.
This pattern emphasizes small volumes, lean proteins, simple carbohydrates, and continuous hydration. As your body adapts to each dose level, you can gradually reintroduce more varied foods and larger portions. The goal isn't to eat this way forever - it's to get through the adjustment period comfortably. The lifestyle hub offers additional meal planning guidance for patients on GLP-1 therapy.
When to Consider Switching Agents
If nausea remains intolerable despite maximum management efforts, switching to a different GLP-1 agonist is a reasonable strategy. Individual responses vary significantly between agents, and a patient who can't tolerate semaglutide may do well on tirzepatide or vice versa. The GIP receptor activation component of tirzepatide may provide better tolerability for some patients. Conversely, some patients find daily liraglutide more manageable than weekly injections because they can adjust timing and doses more flexibly.
Discuss switching options with your prescriber if nausea has persisted for more than 6-8 weeks at the same dose despite implementing all management strategies. Some clinicians also consider lower target doses - not everyone needs to reach the maximum approved dose to achieve meaningful clinical benefit. A moderate dose with good tolerability may produce better long-term outcomes than a maximum dose that the patient abandons after 3 months.
Pancreatitis: Risk Assessment
Figure 5: Pancreatitis involves inflammation of the pancreas, and its relationship to GLP-1 therapy has been extensively studied
The question of whether GLP-1 receptor agonists increase pancreatitis risk has generated more debate than almost any other safety concern in this drug class. Early post-marketing reports of acute pancreatitis in patients taking exenatide triggered FDA safety communications, Congressional hearings, and years of additional research. The current consensus, based on extensive clinical trial and real-world data, is reassuring but nuanced.
What the Clinical Trials Show
The largest and most definitive dataset comes from the cardiovascular outcomes trials (CVOTs), which were specifically designed with the statistical power and follow-up duration to detect safety signals. In the LEADER trial, which followed 9,340 patients on liraglutide or placebo for a median of 3.84 years, pancreatitis occurred in 18 patients in the liraglutide group (0.4%) and 23 patients in the placebo group (0.5%) - no significant difference (Marso SP, Daniels GH, et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. New England Journal of Medicine. 2016;375(4):311-322. DOI: 10.1056/NEJMoa1603827).
The SUSTAIN-6 trial with semaglutide showed adjudicated pancreatitis in 9 patients (0.5%) in the semaglutide group versus 12 patients (0.7%) in the placebo group. The PIONEER trials with oral semaglutide produced similarly low and non-differential rates. The REWIND trial with dulaglutide in over 9,900 patients showed no increased pancreatitis signal over 5.4 years of median follow-up (Gerstein HC, Colhoun HM, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. The Lancet. 2019;394(10193):121-130. DOI: 10.1016/S0140-6736(19)31149-3).
Real-World Data and Meta-Analyses
Multiple meta-analyses have pooled data across GLP-1 trials. A 2017 meta-analysis of 33 randomized controlled trials (N = 22,814) found no significant increase in pancreatitis risk with GLP-1 receptor agonists (OR 0.93, 95% CI 0.65-1.34). A 2019 meta-analysis of the cardiovascular outcomes trials specifically confirmed this finding. Large real-world database studies from Scandinavia, the UK, and the US have produced consistent results: GLP-1 users don't have higher pancreatitis rates than matched controls on other diabetes medications.
That said, it's worth understanding why the concern exists and who should be cautious. Patients with type 2 diabetes already have approximately 2-3 times the background risk of pancreatitis compared to the general population, independent of any medication. Elevated triglycerides, gallstone disease, and alcohol use - all common in the diabetic population - are well-established pancreatitis risk factors. Disentangling drug effects from disease-related risk has been the central challenge in studying this question.
Who Should Be Cautious
While the overall data are reassuring, certain patient populations warrant closer monitoring:
- History of pancreatitis: Patients with a prior episode of acute or chronic pancreatitis should use GLP-1 drugs cautiously. Most prescribing guidelines recommend against initiating GLP-1 therapy in patients with a history of pancreatitis, or at minimum, a careful risk-benefit discussion.
- Severe hypertriglyceridemia: Triglyceride levels above 500 mg/dL significantly increase pancreatitis risk. GLP-1 drugs actually lower triglycerides, which is a protective effect, but the baseline risk still warrants monitoring.
- Heavy alcohol use: Alcohol is a leading cause of pancreatitis. Patients who drink heavily should understand this compounding risk factor.
- Gallstone disease: Gallstones can cause pancreatitis by obstructing the pancreatic duct. Since rapid weight loss (on or off GLP-1 therapy) increases gallstone formation, this connection deserves attention.
Recognizing the Symptoms
Acute pancreatitis typically presents with severe upper abdominal pain that often radiates to the back, accompanied by nausea and vomiting. The pain is persistent - it doesn't come and go like typical GI discomfort - and is usually described as a deep, boring, or "belt-like" sensation. Patients often can't find a comfortable position and may instinctively lean forward.
The critical distinction is between this pattern and the common GI side effects of GLP-1 drugs. Mild, fluctuating abdominal discomfort associated with nausea during dose titration is normal. Severe, unrelenting upper abdominal pain radiating to the back is not, and requires immediate medical evaluation. A serum lipase level more than 3 times the upper limit of normal, combined with characteristic symptoms, confirms the diagnosis.
Action Steps
If you experience severe abdominal pain that radiates to your back and persists for more than a few hours, stop your GLP-1 medication and go to the emergency department. This presentation needs urgent evaluation for pancreatitis regardless of what medication you're taking. Don't wait to see if it improves on its own - pancreatitis can become a serious, life-threatening condition if not treated promptly.
Amylase and Lipase Monitoring
GLP-1 receptor agonists cause modest increases in amylase and lipase levels in many patients - typically 10-15% above baseline. This is a pharmacological effect of GLP-1 receptor activation in the pancreas and does not indicate pancreatic inflammation. Routine monitoring of pancreatic enzymes is not recommended, as it would lead to unnecessary anxiety and false-positive pancreatitis workups. Lipase testing should be reserved for patients with symptoms suggestive of pancreatitis.
The Biological Plausibility Question
Why was pancreatitis even suspected with GLP-1 drugs? There's a biological rationale that makes the concern at least theoretically plausible. GLP-1 receptors are expressed on pancreatic acinar cells. Stimulation of these receptors promotes exocrine secretion - the release of digestive enzymes into the pancreatic ducts. The theory was that chronic, sustained GLP-1 receptor stimulation could lead to excessive enzyme secretion, duct obstruction, and ultimately pancreatitis.
Preclinical studies added fuel to this theory. Some rodent studies showed pancreatic duct hyperplasia and focal pancreatitis-like changes in animals treated with GLP-1 agonists at high doses. However, these findings were not consistently replicated across species or studies. And the human clinical data - from trials involving over 60,000 patients across the major programs - have not confirmed a meaningful signal.
A reasonable conclusion based on the totality of evidence: GLP-1 receptor agonists don't cause pancreatitis in the general population, but patients with pre-existing pancreatic vulnerability (prior pancreatitis, gallstone disease, heavy alcohol use, severe hypertriglyceridemia) should be monitored more carefully. The benefits of GLP-1 therapy usually outweigh this theoretical risk, but the discussion should be individualized. Gut-supportive peptides like BPC-157 and larazotide are being studied for their potential to support GI and pancreatic mucosal health, though data specific to GLP-1 co-administration are not yet available.
Pancreatitis vs. Common GI Symptoms: A Comparison Table
| Feature | Common GLP-1 GI Side Effects | Acute Pancreatitis |
|---|---|---|
| Pain location | Diffuse, mild-moderate, periumbilical or lower abdomen | Severe, upper abdomen, radiates to back |
| Pain character | Cramping, intermittent, fluctuating | Deep, boring, persistent, unrelenting |
| Duration | Minutes to hours, comes and goes | Hours to days, does not resolve |
| Nausea/vomiting | Related to meals, improves with fasting | Severe, not relieved by vomiting |
| Fever | Absent | Often present |
| Position effect | No specific pattern | Worse lying flat, better leaning forward |
| Lipase level | Normal or slightly elevated (1-1.5x ULN) | >3x upper limit of normal |
| Response to time | Improves over weeks with adaptation | Worsens without treatment |
Thyroid C-Cell Tumors: Evidence Review
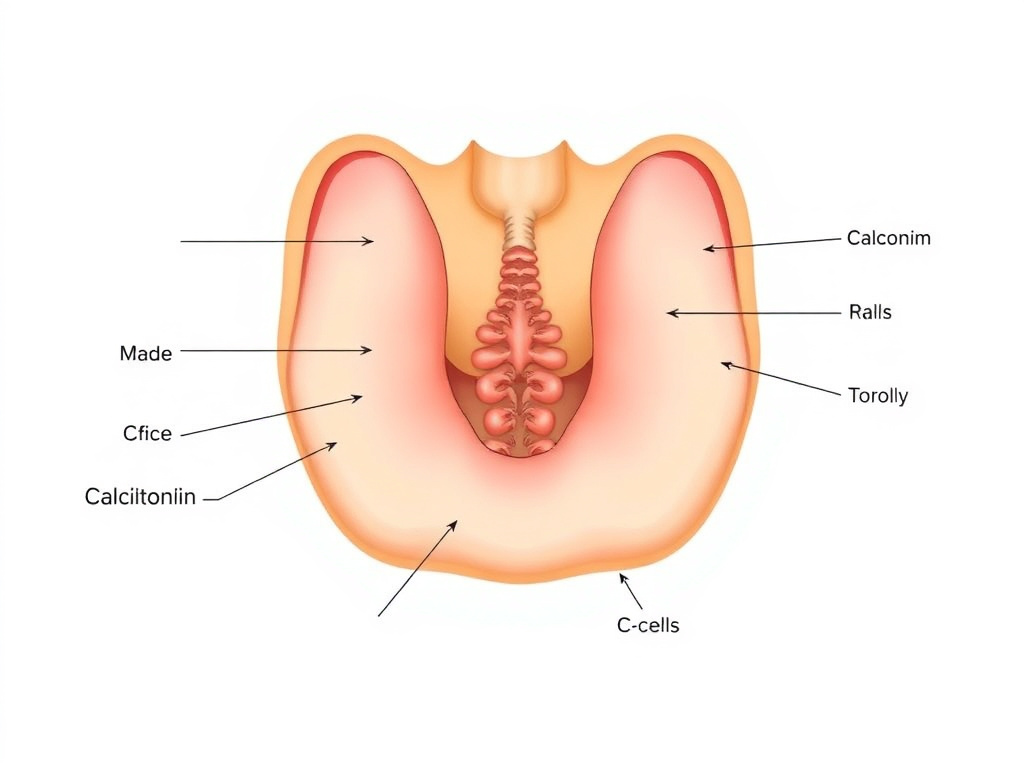
Figure 6: Thyroid C-cells produce calcitonin and have GLP-1 receptors in rodents, but human C-cells express these receptors at much lower levels
Every GLP-1 receptor agonist on the market carries a boxed warning - the FDA's most serious safety label - about the risk of thyroid C-cell tumors, including medullary thyroid carcinoma (MTC). This warning is based on findings in rodent studies, and understanding the biology behind it is essential for evaluating the actual risk to human patients.
The Rodent Data
In preclinical studies, lifelong exposure to GLP-1 receptor agonists in rats and mice produced dose-dependent increases in thyroid C-cell hyperplasia, C-cell adenomas, and C-cell carcinomas (medullary thyroid carcinoma). These findings were consistent across liraglutide, exenatide, semaglutide, and dulaglutide. The doses used in these studies were multiples of the human therapeutic dose, but the signal was clear and consistent enough that the FDA required the boxed warning for the entire class.
The mechanism is straightforward in rodents. Rat thyroid C-cells express GLP-1 receptors at high density. When activated chronically, these receptors stimulate calcitonin release and promote C-cell proliferation. Over time, this proliferative stimulus can progress from hyperplasia to adenoma to carcinoma - a classic tumor progression sequence.
Why Humans Are Different
The critical question is whether this rodent finding translates to humans. The current evidence strongly suggests it does not, for several reasons:
First, human thyroid C-cells express GLP-1 receptors at dramatically lower levels than rodent C-cells. Studies using immunohistochemistry and gene expression analysis have shown that human C-cells have roughly 10-100 times fewer GLP-1 receptors than rat C-cells. This means the same circulating drug level produces a vastly different level of receptor activation in the thyroid between species (Bjerre Knudsen L, Madsen LW, et al. Glucagon-like peptide-1 receptor agonists activate rodent thyroid C-cells causing calcitonin release and C-cell proliferation. Endocrinology. 2010;151(4):1473-1486. DOI: 10.1210/en.2009-1272).
Second, calcitonin levels - the best biomarker for C-cell activation - do not increase in humans on GLP-1 therapy. If GLP-1 drugs were stimulating human C-cells the way they do in rats, you'd expect to see rising calcitonin levels. Across all major clinical trials, calcitonin levels in GLP-1-treated patients have remained within the normal range and have not differed from placebo groups.
What the Human Data Show
The LEADER trial provides the most comprehensive human safety data. Over 9,340 patients were followed for a median of 3.84 years, with adjudicated thyroid events. Thyroid neoplasms were reported in 20 patients in the liraglutide group and 21 in the placebo group. Specifically looking at C-cell pathology, there was no signal of concern. The SUSTAIN-6 trial with semaglutide and the REWIND trial with dulaglutide produced similar findings: no excess thyroid cancer events in the treatment groups.
A 2023 meta-analysis combining data from all GLP-1 cardiovascular outcomes trials found no increased risk of thyroid malignancy (RR 1.01, 95% CI 0.60-1.70). Post-marketing surveillance databases covering millions of patient-years of GLP-1 exposure have not identified a thyroid cancer signal.
Contraindications and Monitoring
Despite the reassuring human data, GLP-1 receptor agonists remain contraindicated in:
- Patients with a personal history of medullary thyroid carcinoma
- Patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN2)
- Patients with a family history of MTC (first-degree relatives)
These contraindications are maintained as a precautionary measure. MTC is extremely rare in the general population (about 500-1,000 new cases per year in the US), but in patients with genetic predisposition (MEN2 or RET proto-oncogene mutations), the risk is substantially elevated. Adding any theoretical additional stimulus - however small - isn't justified in these patients.
Routine thyroid ultrasound or calcitonin monitoring is not recommended for GLP-1 users without risk factors. However, if a patient develops a thyroid nodule or has symptoms suggestive of thyroid disease (neck mass, difficulty swallowing, hoarseness), standard thyroid evaluation should be performed regardless of GLP-1 use.
Putting the Risk in Perspective
Medullary thyroid carcinoma is extremely rare, accounting for only 3-4% of all thyroid cancers. The incidence in the general population is about 0.1-0.2 per 100,000 people per year. To put this in context: even if GLP-1 drugs doubled the risk of MTC (which the data do not suggest), the absolute risk would still be vanishingly small - roughly 0.2-0.4 per 100,000 per year.
Compare this to the well-documented benefits of GLP-1 therapy. In the SELECT trial, semaglutide reduced major adverse cardiovascular events by 20% in patients with obesity but without diabetes. Given that cardiovascular disease kills over 600,000 Americans annually, the cardiovascular risk reduction from GLP-1 therapy dwarfs any theoretical thyroid risk by several orders of magnitude.
This doesn't mean the boxed warning should be ignored - it serves as a useful screen for high-risk patients. But for the vast majority of GLP-1 users, the thyroid cancer concern should not be a barrier to initiating therapy. A simple pre-treatment family history screen for MTC and MEN2 is sufficient. The free assessment at FormBlends includes relevant screening questions.
Differentiated Thyroid Cancers: A Separate Question
Some analyses have looked at whether GLP-1 drugs affect the risk of papillary or follicular thyroid cancer (the more common types, which arise from follicular cells rather than C-cells). These cancers operate through completely different biology than MTC, and there's no mechanistic reason to expect GLP-1 drugs to influence them. Reassuringly, the clinical trial data show no signal for increased differentiated thyroid cancer either.
A 2023 observational study from France raised some concern about overall thyroid cancer risk with GLP-1 use, but the analysis had significant methodological limitations, including immortal time bias and inability to account for detection bias (GLP-1 users may undergo more medical testing, leading to incidental thyroid cancer detection). Several subsequent analyses with better methodology have not confirmed this signal. The consensus view remains that GLP-1 drugs don't increase thyroid cancer risk in humans.
Gallbladder Disease
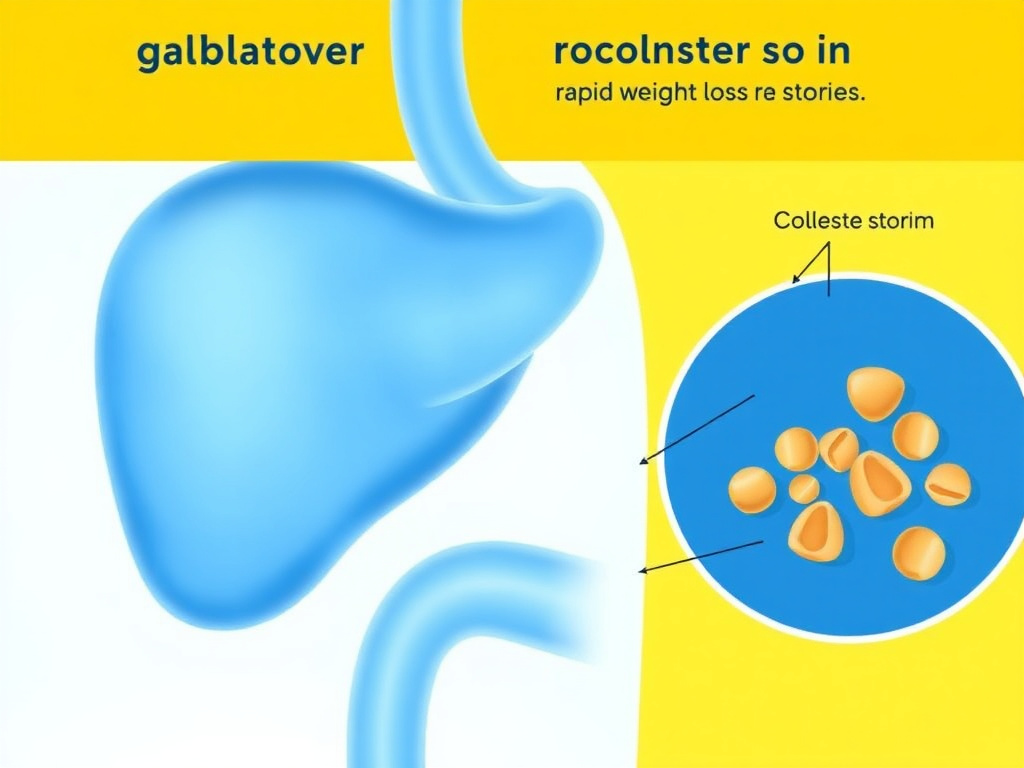
Figure 7: Rapid weight loss increases cholesterol saturation in bile, promoting gallstone formation regardless of the weight loss method used
Gallbladder-related events, including cholelithiasis (gallstones), cholecystitis (gallbladder inflammation), and biliary colic, occur at elevated rates in patients on GLP-1 receptor agonist therapy. But the story is more about rapid weight loss than about the drugs themselves. This distinction matters for understanding the risk and knowing how to mitigate it.
The Weight Loss Connection
Rapid weight loss is one of the most well-established risk factors for gallstone formation. When the body mobilizes stored fat quickly, the liver secretes excess cholesterol into bile, supersaturating it. Simultaneously, gallbladder motility often decreases during caloric restriction, allowing bile to sit in the gallbladder longer and form crystals. These crystals can aggregate into stones. This mechanism operates identically whether weight loss comes from GLP-1 drugs, bariatric surgery, very-low-calorie diets, or any other method.
The risk is proportional to the rate and magnitude of weight loss. Losing more than 1.5 kg per week (about 3.3 pounds) increases gallstone risk significantly. And patients losing more than 25% of their body weight - as is now achievable with higher-dose semaglutide and tirzepatide - are at particularly elevated risk.
Clinical Trial Data
In the STEP 1 trial, gallbladder-related events were reported in 2.6% of semaglutide-treated patients versus 1.2% on placebo. In the STEP 5 (2-year) trial, the rate was 3.7% with semaglutide versus 0.5% with placebo - the higher rate likely reflecting the longer duration of substantial weight loss. Cholecystectomy (surgical gallbladder removal) was required in approximately 1% of semaglutide-treated patients in STEP 5.
The SURMOUNT program with tirzepatide showed similar patterns. In SURMOUNT-1, cholelithiasis was reported in 0.6-1.7% of tirzepatide-treated patients across dose groups versus 0.3% on placebo. The rates were highest in the 15 mg group, which also produced the most weight loss. In the SURPASS trials for diabetes - where weight loss was somewhat less - gallbladder event rates were lower.
Risk Factors for Gallbladder Events
Several factors increase gallstone risk beyond the rate of weight loss:
- Female sex: Women are 2-3 times more likely to develop gallstones than men, an effect mediated by estrogen's influence on cholesterol metabolism
- Age over 40: Gallstone prevalence increases progressively with age
- Obesity itself: Paradoxically, the population most likely to use GLP-1 drugs already has elevated baseline gallstone risk
- Rapid weight cycling: Repeated cycles of weight loss and regain increase cumulative gallstone risk
- Family history: There is a genetic component to gallstone susceptibility
- Native American and Hispanic ethnicity: Higher baseline rates of cholesterol gallstones
Prevention and Monitoring
There's no proven pharmacological prevention strategy for GLP-1-associated gallstones. Ursodeoxycholic acid (UDCA, brand name Actigall) has been shown to reduce gallstone formation during rapid weight loss after bariatric surgery, and some clinicians prescribe it off-label for high-risk GLP-1 patients losing weight rapidly. The typical preventive dose is 300 mg twice daily. However, this is not a standard guideline recommendation, and the decision should be individualized.
Monitoring involves being alert to symptoms. Biliary colic - the hallmark of symptomatic gallstones - presents as intense right upper quadrant or epigastric pain, often after fatty meals, lasting 30 minutes to several hours. It may radiate to the right shoulder or back. Nausea and vomiting frequently accompany the pain. If fever, persistent pain, or jaundice develop, this suggests cholecystitis or common bile duct obstruction, which require urgent medical attention.
Practical Tip
If you're losing weight rapidly on a GLP-1 drug (more than 2-3 pounds per week consistently), discuss gallbladder risk with your provider. A baseline abdominal ultrasound can establish whether you have pre-existing gallstones. Some clinicians recommend periodic ultrasound monitoring for patients with multiple gallstone risk factors who are losing weight quickly. At minimum, know the symptoms of biliary colic so you can seek care promptly if they develop.
Is There a Direct Drug Effect?
Beyond the weight-loss-mediated mechanism, some researchers have hypothesized that GLP-1 receptor agonists might directly affect gallbladder motility. GLP-1 receptors have been identified in gallbladder smooth muscle, and some data suggest that GLP-1 agonists may reduce gallbladder emptying (increase gallbladder stasis). Reduced gallbladder contraction would allow bile to concentrate and form crystals more readily, independent of weight loss.
The evidence for this direct effect is mixed. A small study found that exenatide reduced gallbladder ejection fraction by approximately 20%, but the clinical significance of this finding is unclear. In the clinical trials, gallbladder event rates correlate more strongly with the magnitude of weight loss than with the specific GLP-1 drug or dose, which argues against a major direct drug effect. The most likely explanation is that rapid weight loss is the dominant driver, with a possible minor contribution from direct gallbladder effects.
Comparison to Bariatric Surgery Gallstone Rates
For perspective, the gallstone formation rate after bariatric surgery - particularly gastric bypass and sleeve gastrectomy - is 30-40% within the first year. This is dramatically higher than the 2-4% rate seen in GLP-1 trials, despite the weight loss magnitude being comparable in some cases. The likely explanation is that bariatric surgery produces faster weight loss (particularly in the first 3 months) and more dramatic changes in bile composition. This comparison reinforces the concept that it's the rate of weight loss, not the method, that drives gallstone risk.
Prophylactic ursodeoxycholic acid (UDCA) at 500-600 mg daily is standard practice after bariatric surgery based on randomized trial evidence showing a roughly 50% reduction in gallstone formation (Stokes CS, Gluud LL, et al. Ursodeoxycholic acid and diets higher in fat prevent gallbladder stones during weight loss: a meta-analysis of randomized controlled trials. Clinical Gastroenterology and Hepatology. 2014;12(7):1090-1100. DOI: 10.1016/j.cgh.2013.11.031). Whether the same approach should be adopted for high-risk GLP-1 patients losing weight rapidly is an open question that warrants formal study.
Gastroparesis Concerns
Reports linking GLP-1 receptor agonists to gastroparesis have attracted substantial media attention. Social media accounts and lawsuits have described severe cases of "stomach paralysis" attributed to drugs like semaglutide and tirzepatide. But the clinical picture is more nuanced than these headlines suggest, and it's important to separate the expected pharmacological effect of gastric slowing from true gastroparesis.
Delayed Gastric Emptying vs. Gastroparesis
GLP-1 receptor agonists slow gastric emptying by 20-40% - this is a feature, not a bug. It's one of the primary mechanisms through which they produce weight loss and improve postprandial glucose control. The slowing is dose-dependent, partially reversible with continued use (tachyphylaxis), and fully reversible upon drug discontinuation.
Clinical gastroparesis, by contrast, is a chronic disorder involving a 50% or greater delay in gastric emptying, often associated with structural or functional damage to the gastric nerves or smooth muscle. It causes persistent symptoms including chronic nausea, vomiting, early satiety, bloating, and abdominal pain. Diabetic gastroparesis, the most common form, typically develops after years of poorly controlled diabetes and reflects autonomic neuropathy.
The distinction matters clinically. A patient on semaglutide who feels uncomfortably full after eating, has some nausea during dose escalation, and retains food in the stomach longer than normal is experiencing the expected pharmacological effect of the drug. A patient who develops intractable vomiting, cannot maintain adequate nutrition, and shows a gastric emptying study with near-complete gastric stasis has a more serious problem that requires investigation.
What the Data Actually Show
A 2023 study published in JAMA analyzed insurance claims data from 16 million patients and found that GLP-1 use was associated with a slightly increased rate of gastroparesis diagnoses (hazard ratio 3.67, 95% CI 1.15-11.90) compared to bupropion-naltrexone use. However, the absolute risk was very low - approximately 10 per 10,000 person-years versus 4.6 per 10,000 person-years (Sodhi M, Rezaeianzadeh R, et al. Risk of Gastrointestinal Adverse Events Associated With Glucagon-Like Peptide-1 Receptor Agonists for Weight Loss. JAMA. 2023;330(18):1795-1797. DOI: 10.1001/jama.2023.19574).
There are important caveats with this data. The diagnosis of "gastroparesis" in claims databases often reflects symptom coding rather than formal gastric emptying studies. A patient presenting with GLP-1-related nausea and delayed gastric emptying symptoms might receive a gastroparesis diagnosis code even though the condition is expected, dose-dependent, and reversible. Differentiating true drug-induced gastroparesis from the expected pharmacological effect of gastric slowing requires more detailed clinical assessment than claims data can provide.
Pre-Existing Gastroparesis and GLP-1 Therapy
Patients with pre-existing gastroparesis - particularly diabetic gastroparesis - should approach GLP-1 therapy with caution. Adding a drug that further slows gastric emptying on top of an already compromised gastric motility can produce severe symptoms. Some of the most dramatic adverse event reports involve patients who had undiagnosed or subclinical gastroparesis that became clinically apparent only after starting a GLP-1 agonist.
If you have a history of early satiety, chronic bloating, frequent nausea, or unexplained vomiting before starting GLP-1 therapy, discuss these symptoms with your provider. A baseline gastric emptying study (gastric emptying scintigraphy) may be warranted to assess your gastric motility before adding a drug that will slow it further. This is the approach recommended by the clinical science team at FormBlends.
Management
For patients experiencing severe gastric emptying delay on GLP-1 therapy, the primary management strategy is dose reduction or temporary discontinuation. In most cases, reducing the dose by one step alleviates the worst symptoms while maintaining some therapeutic benefit. If symptoms persist at the lowest available dose, discontinuation with a planned washout period is appropriate.
Dietary modifications that help include eating very small, frequent meals; prioritizing liquids and semi-solid foods that empty from the stomach more easily; avoiding high-fat and high-fiber foods that slow gastric emptying; and staying upright after eating. Prokinetic medications like metoclopramide or domperidone (available outside the US) can be considered for short-term use but have their own significant side effect profiles.
The Legal Landscape
Multiple lawsuits have been filed alleging that GLP-1 manufacturers failed to adequately warn about gastroparesis risk. These lawsuits are ongoing as of early 2026, and their outcomes will likely influence future prescribing information and risk communication. Regardless of the legal outcomes, the scientific question is separate: can GLP-1 drugs cause permanent gastroparesis?
The current answer, based on available evidence, is that GLP-1-induced gastric emptying delay is reversible upon discontinuation in the vast majority of cases. The half-life of weekly semaglutide is approximately 7 days, meaning that drug-related gastric effects should resolve within 4-6 weeks of stopping. If symptoms persist beyond this washout period, other causes of gastroparesis should be investigated - particularly undiagnosed diabetic autonomic neuropathy, which may have been unmasked rather than caused by the GLP-1 drug.
Practical Pre-Treatment Screening
Before starting GLP-1 therapy, your provider should ask about symptoms that might suggest pre-existing gastroparesis or impaired gastric motility:
- Do you frequently feel full after eating very small amounts?
- Do you experience persistent bloating or abdominal distension?
- Do you have frequent nausea or vomiting unrelated to meals?
- Have you been told you have a "slow stomach" or delayed gastric emptying?
- Do you have longstanding diabetes with neuropathy symptoms (numbness, tingling)?
Positive answers to multiple questions warrant further evaluation before initiating a medication that will further slow gastric emptying. A baseline gastric emptying study can clarify whether delayed gastric motility pre-exists and help set appropriate expectations for GI tolerability. Our GLP-1 mechanisms guide provides additional detail on how gastric emptying delay contributes to both the benefits and side effects of these medications.
The Anesthesia Concern
In 2023, the American Society of Anesthesiologists (ASA) issued guidance recommending that patients on GLP-1 receptor agonists consider holding their medication before elective surgery requiring general anesthesia. The concern is that delayed gastric emptying increases the risk of pulmonary aspiration during intubation, because food may still be in the stomach even after the standard pre-operative fasting period.
The current ASA recommendation suggests holding daily GLP-1 drugs (like liraglutide) on the day of surgery, and weekly GLP-1 drugs (like semaglutide and tirzepatide) for at least one week before elective procedures. Patients with active GI symptoms (nausea, vomiting, bloating) should be treated as if they have a full stomach regardless of fasting duration. If surgery is urgent and the GLP-1 drug can't be held, rapid-sequence intubation is recommended.
If you're scheduled for any procedure requiring anesthesia or sedation, inform your anesthesiologist that you're taking a GLP-1 medication, even if the surgical team hasn't asked. This is an area of active guideline development, and not all anesthesia providers may be aware of the gastric emptying implications.
Muscle Loss & Nutritional Deficiencies
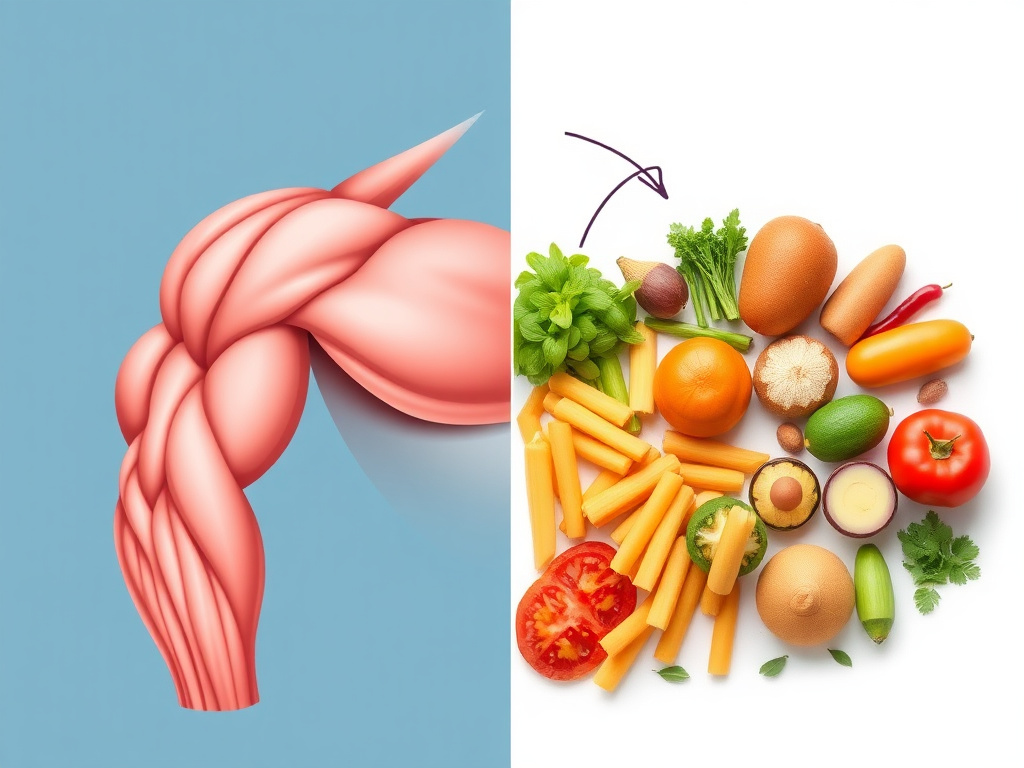
Figure 8: Adequate protein intake and resistance training are essential for preserving lean mass during GLP-1-mediated weight loss
One of the most significant long-term concerns with GLP-1-mediated weight loss is the loss of lean body mass - primarily skeletal muscle. While all weight loss methods result in some lean mass loss along with fat mass, the magnitude matters for metabolic health, physical function, and the sustainability of weight loss itself. And the dramatic caloric reductions that GLP-1 drugs produce can, if unmanaged, lead to nutritional deficiencies that compound the problem.
How Much Muscle Are People Losing?
In any caloric deficit, the body loses a mixture of fat and lean tissue. The ratio depends on the size of the deficit, the protein content of the diet, physical activity levels, and individual factors like age and baseline body composition. In typical non-surgical weight loss, about 25% of total weight lost comes from lean mass.
With GLP-1 receptor agonists, the lean mass loss fraction has been reported at 25-40% in different trials. In STEP 1, DEXA body composition data showed that approximately 39% of weight lost with semaglutide 2.4 mg was lean mass (compared to 25% with placebo, reflecting the much larger magnitude of weight loss). The SURMOUNT-1 data with tirzepatide showed a more favorable ratio at the highest dose: approximately 27% lean mass loss with the 15 mg dose.
The absolute numbers put this in perspective. A patient who loses 20 kg on semaglutide might lose approximately 12-13 kg of fat and 7-8 kg of lean mass. That 7-8 kg of lean mass includes not just skeletal muscle but also organ mass, bone mineral content, and body water associated with lean tissue. The actual skeletal muscle loss is somewhat less than the total lean mass loss, but it's still clinically meaningful.
Why Muscle Loss Matters
Skeletal muscle isn't just for aesthetics. It's the body's primary glucose disposal tissue, responsible for about 80% of insulin-mediated glucose uptake. Muscle loss reduces resting metabolic rate, making weight regain more likely. It impairs physical function, especially in older adults, where the combination of obesity and muscle loss - termed "sarcopenic obesity" - is associated with falls, frailty, and loss of independence.
The metabolic consequence is a lower caloric set point. If you lose significant muscle mass during your weight loss phase, your body burns fewer calories at rest, and maintaining the lower weight becomes harder. This contributes to the weight regain that many patients experience after discontinuing GLP-1 therapy.
Strategies to Preserve Lean Mass
Protein Intake
Adequate protein consumption is the single most important dietary factor for preserving muscle during weight loss. Current evidence supports a protein intake of 1.2-1.6 g per kg of body weight per day during GLP-1-mediated weight loss. For a 100 kg patient, that's 120-160 g of protein daily. This is substantially more than the general population RDA of 0.8 g/kg and can be challenging to achieve when appetite is severely suppressed.
Practical strategies for hitting protein targets on a reduced appetite:
- Prioritize protein at every meal - eat the protein portion first before you fill up
- Use protein supplements: whey protein isolate, casein, or plant-based protein powders
- Choose protein-dense foods: Greek yogurt, cottage cheese, lean meats, fish, eggs
- Consider ready-to-drink protein shakes for days when solid food intake is very low
- Spread protein intake across 3-4 meals rather than concentrating it in one sitting
Resistance Training
Resistance exercise provides the mechanical stimulus that signals muscles to maintain or build mass, even during a caloric deficit. Studies of resistance training combined with caloric restriction consistently show better lean mass preservation compared to caloric restriction alone. The combination of GLP-1 therapy plus resistance training has been examined in smaller studies and shows promising results.
A minimum effective resistance training program during GLP-1 therapy would include 2-3 sessions per week, targeting all major muscle groups, using progressive overload (gradually increasing weight or repetitions). Even moderate-intensity resistance bands or bodyweight exercises provide benefit. The lifestyle hub provides additional exercise guidance for GLP-1 users.
Nutritional Deficiencies
When caloric intake drops substantially, getting adequate micronutrients becomes challenging. GLP-1-mediated appetite suppression can reduce daily intake to 800-1,200 calories in some patients, which is below the threshold where most people can meet their micronutrient needs through food alone. Common deficiencies reported in GLP-1 users include:
- Iron: Reduced meat consumption and lower overall food intake can lead to iron deficiency, particularly in menstruating women
- Vitamin B12: GLP-1 drugs may reduce intrinsic factor secretion, and lower food intake further reduces B12 intake
- Vitamin D: Already widely deficient in obese populations, and reduced caloric intake makes it harder to get from food
- Calcium: Particularly concerning in combination with vitamin D deficiency, as it may accelerate bone mineral density loss during weight loss
- Folate, zinc, and magnesium: Reported at lower-than-optimal levels in some GLP-1-using populations
A daily multivitamin is a reasonable baseline recommendation for anyone on GLP-1 therapy. Additional supplementation with vitamin D (2,000-4,000 IU daily), calcium (500-1,000 mg daily if dietary intake is low), and possibly B12 (especially for patients over 50 or on metformin concurrently) should be individualized based on lab monitoring. Peptides like BPC-157 have been studied for GI mucosal health, and some clinicians incorporate gut-supportive compounds alongside GLP-1 therapy.
Clinical Recommendation
Every patient on GLP-1 therapy should have a nutritional assessment that includes protein intake calculation, micronutrient screening (CBC, iron panel, B12, vitamin D, metabolic panel), and a discussion about resistance training. These conversations should happen at treatment initiation and be revisited at every dose escalation visit. The patients who preserve the most muscle and maintain the best nutritional status are the ones with proactive, structured support.
The Role of Adjunctive Therapies
Several adjunctive approaches are being explored to preserve lean mass during GLP-1-mediated weight loss:
Creatine monohydrate (3-5 g daily) is one of the most well-studied performance supplements and may help support muscle maintenance during caloric restriction. It works by increasing intramuscular phosphocreatine stores, which supports higher-intensity resistance training performance. The additional water retention associated with creatine may slightly offset the scale weight loss, which patients should understand.
Growth hormone secretagogues like CJC-1295/Ipamorelin, MK-677, and sermorelin are used by some patients and clinicians to support lean mass preservation during weight loss. Growth hormone promotes lipolysis (fat burning) and protein synthesis (muscle preservation), making the fat-to-lean mass ratio of weight loss potentially more favorable. However, controlled data on these combinations with GLP-1 therapy are limited, and the approach remains off-label.
Omega-3 fatty acids at doses of 2-4 g daily may support muscle protein synthesis and reduce inflammation. Some studies suggest omega-3 supplementation augments the anabolic response to resistance exercise, though the effect sizes are modest.
Vitamin D optimization deserves special emphasis. Vitamin D deficiency is associated with reduced muscle strength and increased risk of sarcopenia. Given that many obese individuals are vitamin D deficient at baseline, and that caloric restriction further reduces vitamin D intake, optimizing vitamin D status (target 25-OH vitamin D of 40-60 ng/mL) should be a priority. Supplementation with 2,000-5,000 IU daily is typically needed.
Bone Health Considerations
Weight loss is associated with bone mineral density (BMD) reduction, regardless of the method used. Mechanical unloading (less body weight pressing down on bones) reduces the stimulus for bone remodeling, and caloric restriction can impair calcium absorption. In the STEP 1 extension data, total hip BMD decreased by approximately 2.3% over 68 weeks in the semaglutide group versus 0.3% in the placebo group.
This degree of BMD loss is clinically meaningful, particularly for post-menopausal women and older adults already at risk for osteoporosis. Weight-bearing exercise (walking, jogging, stair climbing) and resistance training help maintain bone density. Adequate calcium (1,000-1,200 mg daily from food and supplements) and vitamin D are essential. Patients with risk factors for osteoporosis should have baseline DEXA scanning and periodic monitoring during treatment. Peptides like MOTS-c are being investigated for metabolic and mitochondrial benefits that could theoretically support tissue health during weight loss, though clinical data remain preliminary.
Mental Health Considerations
The potential impact of GLP-1 receptor agonists on mental health has become an area of growing attention. Reports of depression, anxiety, and suicidal ideation in patients taking these medications have prompted regulatory reviews in multiple countries. The evidence is still evolving, but the topic deserves careful examination rather than either dismissal or panic.
The Regulatory Landscape
In 2023, the European Medicines Agency (EMA) initiated a review of GLP-1 receptor agonists and suicidal ideation following reports in its EudraVigilance database. Iceland's national medicines agency had flagged the signal after approximately 150 reports of self-harm or suicidal thoughts in patients taking GLP-1 drugs. The FDA subsequently stated it was evaluating the reports but had not found sufficient evidence to establish a causal link.
By mid-2024, the EMA concluded its review, finding no causal association between GLP-1 receptor agonists and suicidal or self-harm events. However, they recommended continued monitoring. The FDA reached a similar interim conclusion but added language to prescribing information recommending monitoring for changes in mood or behavior.
What the Clinical Trials Show
The cardiovascular outcomes trials and the STEP/SURPASS/SURMOUNT weight management trials all collected data on psychiatric adverse events. In the STEP programs, rates of depression and anxiety were low and similar between semaglutide and placebo groups. The SELECT trial - which randomized over 17,000 patients to semaglutide or placebo with up to 5 years of follow-up - found no increased risk of suicidality (Lincoff AM, Brown-Frandsen K, et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. New England Journal of Medicine. 2023;389(24):2221-2232. DOI: 10.1056/NEJMoa2307563).
A 2024 large cohort study using Scandinavian health registries actually found lower rates of depression and suicidal behavior in GLP-1 users compared to matched controls, suggesting a possible protective rather than harmful effect. This is biologically plausible - GLP-1 receptors are expressed in brain regions involved in mood regulation, and preclinical studies suggest GLP-1 agonists may have antidepressant and anxiolytic properties.
The Complexity of Weight Loss and Mental Health
Rapid body changes, dietary restriction, hormonal shifts, and altered brain reward signaling all have the potential to affect mood. Patients who lose significant weight sometimes experience unexpected emotional responses - a sense of loss, identity confusion, or relationship strain that doesn't fit the expected narrative of "weight loss equals happiness." The relationship between food, emotions, and self-image is complex, and GLP-1 drugs can disrupt patterns that have been entrenched for years.
Some patients describe a feeling of disconnection from food-related pleasure that extends beyond simple appetite reduction. Food has deep social, cultural, and emotional significance, and when a drug dramatically alters your relationship with eating, the psychological effects can be surprising. This isn't a "side effect" in the traditional pharmacological sense, but it's a real experience that deserves acknowledgment and support.
For patients with a history of eating disorders, GLP-1 therapy requires especially careful management. The dramatic appetite suppression these drugs produce can trigger restrictive eating patterns in vulnerable individuals. Patients with a history of anorexia, bulimia, or binge eating disorder should work closely with a mental health provider alongside their prescribing clinician. The free assessment at FormBlends includes mental health screening questions to help identify patients who may need additional support.
Recommendations
Current evidence does not support withholding GLP-1 therapy due to mental health concerns in the general population. However, the following precautions are reasonable:
- Screen for depression and suicidal ideation at baseline and at follow-up visits
- Ask about changes in mood, sleep, energy, interest in activities, and relationship with food
- Maintain a low threshold for mental health referral, especially in patients with psychiatric history
- Educate patients that emotional changes during rapid weight loss are common and manageable
- Consider the PHQ-9 (depression screen) and GAD-7 (anxiety screen) as part of routine monitoring
The Alcohol Connection
An emerging area of interest is the effect of GLP-1 receptor agonists on alcohol consumption and cravings. Multiple preclinical studies have shown that GLP-1 agonists reduce alcohol intake in animal models, and a growing body of observational data suggests that patients on GLP-1 drugs often spontaneously reduce their alcohol consumption. Some patients report that alcoholic beverages simply become less appealing, similar to their altered relationship with food.
This has both positive and potentially concerning implications. For patients who previously drank excessively, reduced alcohol desire could be a significant health benefit. But for some patients, the loss of a coping mechanism or social lubricant can contribute to psychological distress, particularly if they haven't developed alternative coping strategies. Clinicians should ask about changes in alcohol consumption during GLP-1 therapy and frame the conversation positively while remaining alert to patients who may be struggling with the change.
Clinical trials are now underway studying semaglutide specifically for alcohol use disorder, which may provide more definitive data on this relationship. In the meantime, the anecdotal and observational evidence is intriguing enough that it warrants discussion with patients, especially those who use alcohol regularly. The biohacking hub explores the neuroscience behind GLP-1's effects on reward circuitry.
Body Image and Identity
Rapid, significant weight loss changes how people look, how they're perceived by others, and how they perceive themselves. These changes can be profoundly positive - increased confidence, reduced self-consciousness, improved social interactions. But they can also trigger unexpected psychological responses. Some patients describe feeling like they're in someone else's body. Others struggle with increased attention or unwanted comments about their appearance.
Relationship dynamics can shift when one partner loses significant weight. Feelings of jealousy, insecurity, or power imbalance may surface. Long-term friendships built around shared food experiences may change. These are real, common experiences that deserve acknowledgment in the clinical setting, even though they're not "side effects" in the pharmacological sense.
Body dysmorphia - a distorted perception of one's body - can persist or even worsen during rapid weight loss. A patient who has been overweight for decades may continue to see themselves as overweight even after losing 50+ pounds. Conversely, some patients develop an unhealthy fixation on achieving lower and lower weights, which can edge into disordered eating territory. The role of the prescriber extends beyond writing the prescription: it includes monitoring the psychological dimensions of a profoundly life-altering treatment.
Rare Complications
Beyond the common GI side effects and the well-studied safety concerns discussed above, GLP-1 receptor agonists have been associated with several rare but clinically significant complications. These events are infrequent enough that they may not reach statistical significance in individual clinical trials, but awareness among prescribers and patients is still warranted.
Acute Kidney Injury
Cases of acute kidney injury (AKI) have been reported in post-marketing surveillance, primarily in the context of severe dehydration from persistent vomiting and diarrhea. The mechanism is prerenal - volume depletion from GI fluid losses leads to reduced renal perfusion. Patients on concurrent nephrotoxic medications (NSAIDs), diuretics, or renin-angiotensin system blockers (ACE inhibitors, ARBs) are at higher risk.
In the SUSTAIN and STEP programs, renal events were not increased with semaglutide compared to placebo. In fact, several GLP-1 trials have shown renal protective effects, including reductions in albuminuria and slower eGFR decline. The FLOW trial with semaglutide specifically demonstrated renal benefit in patients with chronic kidney disease and type 2 diabetes. But these population-level benefits don't eliminate the risk of AKI in individual patients experiencing severe dehydration. The lesson: if you can't keep fluids down, seek medical attention rather than waiting it out.
Intestinal Obstruction
Rare cases of intestinal obstruction, including ileus (functional obstruction without mechanical blockage) and small bowel obstruction, have been reported in GLP-1 users. The proposed mechanism involves extreme slowing of intestinal motility leading to functional ileus. Patients with prior abdominal surgery, adhesions, or inflammatory bowel conditions may be at higher risk. The JAMA study mentioned in the gastroparesis section found a hazard ratio of 4.22 for intestinal obstruction with GLP-1 use compared to bupropion-naltrexone, though absolute rates were very low (3.1 vs. 0.7 per 10,000 person-years).
Injection Site Reactions
Local reactions at the injection site - including redness, swelling, itching, and nodule formation - occur in 1-5% of patients on subcutaneous GLP-1 formulations. The extended-release exenatide formulation (Bydureon) has the highest rates due to its microsphere delivery system, which can cause palpable nodules that take weeks to resolve. Semaglutide and tirzepatide generally produce milder injection site reactions. Rotating injection sites (abdomen, thigh, upper arm) helps minimize these issues.
Anaphylaxis and Hypersensitivity
Serious allergic reactions to GLP-1 agonists are extremely rare but have been documented. Symptoms can include angioedema (swelling of the face, lips, tongue, or throat), urticaria (hives), and anaphylaxis. These reactions are drug-specific, so a patient who has a hypersensitivity reaction to one GLP-1 agonist may tolerate a different one. If you experience swelling of the face or throat, difficulty breathing, or widespread hives after a GLP-1 injection, seek emergency medical care immediately and do not take another dose.
Hypoglycemia
GLP-1 receptor agonists alone carry a low risk of hypoglycemia because their insulin-stimulating effect is glucose-dependent - they increase insulin secretion only when blood glucose is elevated. However, when combined with sulfonylureas (glipizide, glyburide, glimepiride) or insulin, the hypoglycemia risk increases substantially. In the SUSTAIN trials, hypoglycemia rates were 2-5% with semaglutide monotherapy but increased to 15-25% when combined with a sulfonylurea.
Patients starting a GLP-1 agonist while on insulin or a sulfonylurea should discuss proactive dose reduction of the other medication with their provider. Many clinicians reduce the sulfonylurea dose by 50% when initiating a GLP-1 drug and titrate down further as needed based on glucose monitoring.
Diabetic Retinopathy Worsening
Early worsening of diabetic retinopathy was observed in the SUSTAIN-6 trial with semaglutide, particularly in patients with pre-existing retinopathy who experienced rapid improvements in glycemic control. The proposed mechanism involves retinal "metabolic reperfusion injury" - when chronically high glucose levels drop rapidly, the retinal vasculature undergoes stress. This is not unique to GLP-1 drugs; it has been observed with insulin initiation and other therapies that produce rapid A1c improvements.
The risk is highest in patients with baseline proliferative retinopathy or severe non-proliferative retinopathy. An eye exam before starting GLP-1 therapy and at regular intervals during treatment is recommended for all patients with diabetes. Slow titration that produces gradual rather than abrupt glycemic improvement may reduce this risk. Our semaglutide guide discusses this topic in additional detail.
Hair Loss
Telogen effluvium - temporary, diffuse hair shedding triggered by physiological stress - has been reported by some GLP-1 users, particularly those experiencing rapid weight loss. This is a well-known consequence of significant caloric restriction and rapid weight change, not specific to GLP-1 drugs. It typically begins 2-4 months after the onset of significant weight loss and resolves within 6-12 months even without treatment. Ensuring adequate protein intake, iron levels, and overall nutrition can help minimize the severity and duration. Peptides like GHK-Cu have been studied for their role in hair and skin health, though data specific to weight-loss-related hair shedding is limited.
Facial Aging ("Ozempic Face")
The colloquial term "Ozempic face" refers to the facial volume loss and skin laxity that can accompany significant weight loss. When subcutaneous fat pads in the face deflate rapidly, the result can be a gaunt, aged appearance - hollowed cheeks, more prominent wrinkles, and sagging skin. This is a cosmetic concern rather than a medical one, and it occurs with any form of rapid weight loss, not uniquely with GLP-1 drugs.
Dermal fillers and skin-tightening procedures are the primary cosmetic treatments. From a prevention standpoint, slower weight loss (targeting 0.5-1 kg per week rather than 2+ kg per week) gives the skin more time to contract and adapt. Skincare peptides such as GHK-Cu topical and SNAP-8 are used by some patients for skin quality support during weight loss, though controlled data for this specific application are limited. Other skin-supportive peptides including Matrixyl may support collagen production during periods of significant body composition change.
Fatigue and Asthenia
Generalized fatigue is reported by 5-10% of GLP-1 users in clinical trials. The causes are likely multifactorial: reduced caloric intake, potential dehydration, altered sleep patterns from GI discomfort, and the metabolic adjustments that accompany weight loss all contribute. For most patients, fatigue is mild and improves as the body adapts. Persistent fatigue warrants evaluation for other causes, including thyroid dysfunction, anemia, vitamin B12 deficiency, and depression.
Some patients describe a specific pattern of fatigue on injection day and the day following, which resolves by mid-week. This temporal pattern suggests a direct pharmacological effect rather than a secondary consequence of reduced intake. Adjusting injection timing (trying different days or times of day) can sometimes help. Ensuring adequate hydration, maintaining some level of physical activity (even gentle walking), and avoiding excessive caloric restriction all help manage treatment-related fatigue.
Headache
Headache is reported in approximately 10-15% of GLP-1 users across trials, making it one of the more common non-GI side effects. It's most frequent during the first 1-2 weeks at each dose level and typically resolves with continued treatment. Dehydration, reduced caffeine intake (if the patient has cut back on coffee or tea due to nausea), and blood sugar fluctuations are the most likely contributors. Standard analgesics (acetaminophen preferred over NSAIDs during the GI adjustment period) and adequate hydration are usually sufficient.
Dizziness and Lightheadedness
Approximately 5-8% of patients report dizziness or lightheadedness on GLP-1 therapy. This is often related to dehydration, orthostatic changes during weight loss, or mild hypoglycemia in patients on concurrent glucose-lowering medications. Ensuring adequate fluid intake, rising slowly from seated or lying positions, and monitoring blood glucose if on insulin or sulfonylureas are the primary management strategies.
Summary of Rare Complication Risk
| Complication | Estimated Incidence | Severity | Reversible? |
|---|---|---|---|
| Acute kidney injury | Rare (case reports) | Potentially serious | Yes, with rehydration |
| Intestinal obstruction | ~3 per 10,000 person-years | Serious | Usually, with treatment |
| Anaphylaxis | Very rare | Life-threatening | Yes, with emergency treatment |
| Retinopathy worsening | ~3% (in patients with baseline retinopathy) | Moderate | Variable |
| Hair loss (telogen effluvium) | 5-10% | Mild (cosmetic) | Yes, resolves in 6-12 months |
| Facial volume loss | Common with significant weight loss | Mild (cosmetic) | Partially, with weight stability |
| Headache | 10-15% | Mild | Yes |
| Fatigue | 5-10% | Mild to moderate | Yes, improves with adaptation |
Drug Interactions and Medication Absorption Effects
GLP-1 receptor agonists' effects on gastric emptying don't just change how quickly food moves through your stomach. They also alter how your body absorbs other medications. For the millions of people taking GLP-1 drugs alongside other prescriptions, understanding these pharmacokinetic interactions is essential for safe, effective treatment.
The Gastric Emptying Effect on Oral Drug Absorption
GLP-1 receptor agonists delay gastric emptying by 20-40%, which directly affects the absorption kinetics of co-administered oral medications. The primary pharmacokinetic consequences are a delayed time to peak concentration (Tmax), a potentially reduced peak concentration (Cmax), and, in most cases, a preserved total drug exposure (AUC). For most medications, this means the drug still gets fully absorbed but takes longer to reach its peak blood level.
For medications where the speed of absorption matters - pain medications that need to work quickly, antibiotics that rely on high peak concentrations for bacterial killing, or emergency medications like sublingual nitroglycerin - this delayed absorption can have clinical consequences. For medications where the total amount absorbed matters more than the speed (most chronic medications like statins, ACE inhibitors, and antidepressants), the impact is typically negligible.
Specific Medication Categories of Concern
Oral contraceptives: This interaction deserves special attention because of its implications for unintended pregnancy. Delayed gastric emptying can alter the absorption of ethinyl estradiol and progestins, potentially reducing their peak levels below the threshold needed for reliable ovulation suppression. The clinical significance of this interaction remains debated, with some studies showing minimal impact on contraceptive hormone levels and others showing measurable reductions in Cmax. Prudent practice is to recommend non-oral contraceptive methods (IUDs, implants, depot medroxyprogesterone) during GLP-1 therapy, or to advise patients to take oral contraceptives at least 1 hour before or 11 hours after their GLP-1 injection.
Compounding this concern is the fact that significant weight loss can restore ovulatory cycles in women who were previously anovulatory due to obesity. Women who have relied on obesity-related infertility as de facto contraception may become fertile during GLP-1 treatment without realizing it. Clinicians should proactively discuss contraceptive planning at the initiation of GLP-1 therapy for all premenopausal women.
Thyroid medications: Levothyroxine is particularly sensitive to absorption conditions. It requires an empty stomach, specific gastric pH levels, and consistent transit time for reliable absorption. GLP-1-induced changes in gastric emptying and potentially gastric pH can alter levothyroxine absorption, leading to either under- or over-replacement of thyroid hormone. TSH levels should be checked every 6-8 weeks during GLP-1 initiation and dose escalation, and levothyroxine dose adjustments should be guided by TSH rather than assumed to be stable.
Anticoagulants: Warfarin has a narrow therapeutic index where small changes in blood levels can mean the difference between effective anticoagulation and either bleeding risk or clot risk. While formal interaction studies with semaglutide showed no clinically significant changes in warfarin pharmacokinetics, the FDA labeling for all GLP-1R agonists recommends increased INR monitoring during initiation and dose changes. Direct oral anticoagulants (DOACs like apixaban and rivarelbam) may also have delayed absorption, though these drugs have wider therapeutic margins and are generally less concerning.
Diabetes medications: Sulfonylureas and insulin carry additive hypoglycemia risk when combined with GLP-1R agonists. Insulin doses should be proactively reduced by 15-20% at GLP-1 initiation to prevent hypoglycemia, with further reductions as weight loss progresses and insulin sensitivity improves. Sulfonylureas should ideally be reduced or discontinued before starting GLP-1 therapy. Metformin is generally safe to continue and may provide complementary metabolic benefits. Tirzepatide has particularly potent glucose-lowering effects, and patients on insulin should be counseled about the higher risk of hypoglycemia when initiating this agent.
Psychiatric medications: Many psychiatric drugs (antipsychotics, anticonvulsants, lithium) require consistent blood levels for efficacy and safety. Delayed gastric emptying can alter absorption kinetics, potentially affecting peak-to-trough ratios and steady-state levels. For medications with narrow therapeutic windows (lithium, valproic acid, carbamazepine), therapeutic drug monitoring should be intensified during GLP-1 therapy initiation. For SSRIs and SNRIs, the impact is generally less concerning because these medications have broader therapeutic ranges and gradual onset of action.
Timing Strategies for Co-Administered Medications
General guidance for medication timing with GLP-1R agonists:
- Take time-sensitive medications (levothyroxine, oral contraceptives, quick-onset pain medications) at least 1 hour before eating and at least 2-3 hours before or after GLP-1 injection (for daily formulations).
- For weekly injectable GLP-1R agonists (semaglutide, tirzepatide), the gastric emptying effect is continuous throughout the dosing interval, so timing relative to injection is less relevant. Focus instead on taking critical medications on an empty stomach.
- Consider switching to non-oral formulations of critical medications during active GLP-1 dose escalation when gastric emptying effects are most variable. Transdermal patches, subcutaneous injections, or sublingual formulations bypass the GI tract entirely.
- If a medication's absorption depends on acidic gastric pH (like certain antifungals or protease inhibitors), be aware that GLP-1R agonists can modestly increase gastric pH in some patients, adding another variable to absorption.
Perioperative and Procedural Safety Considerations
The 2023 American Society of Anesthesiologists (ASA) guidance on GLP-1 agonists before elective procedures brought this issue into mainstream medical awareness. For anyone on GLP-1 therapy who may need surgery, endoscopy, or other procedures requiring sedation, understanding these considerations is not optional.
The Aspiration Risk
The core concern is pulmonary aspiration of gastric contents during anesthesia. When patients are sedated or under general anesthesia, the protective airway reflexes (cough, gag, swallowing) are suppressed. If the stomach contains food or liquid, it can reflux into the esophagus and be aspirated into the lungs, causing aspiration pneumonia or, in severe cases, acute respiratory distress syndrome. Aspiration is a rare but serious complication of anesthesia with mortality rates of 3-5% when it occurs.
Standard preoperative fasting guidelines (NPO after midnight, or 6-8 hours for solids and 2 hours for clear liquids) are designed to ensure an empty stomach at the time of anesthesia induction. GLP-1R agonists complicate these guidelines because their gastric emptying delay means that standard fasting periods may not be sufficient for complete gastric emptying. Case reports and small case series have documented retained gastric contents in patients on GLP-1 therapy despite appropriate fasting duration.
Current Recommendations by Procedure Type
Elective procedures under general anesthesia: The ASA consensus guidance recommends holding daily GLP-1R agonists (liraglutide, oral semaglutide) for at least 24 hours before the procedure, and weekly formulations (semaglutide injectable, tirzepatide) for at least 7 days before the procedure. If the patient reports GI symptoms (nausea, vomiting, bloating, abdominal distension) at the time of their preoperative assessment, the procedure should be postponed until symptoms resolve, regardless of the holding period.
Some anesthesiology groups recommend a more cautious approach: holding weekly GLP-1R agonists for 2-3 weeks before elective procedures to allow more complete resolution of gastric motility effects. The evidence base for choosing between 7 days and 21 days is thin, and practice varies between institutions.
Urgent but non-emergent procedures: If a procedure can't wait for the full washout period but isn't a life-threatening emergency, options include proceeding with the assumption of a "full stomach." This means using rapid-sequence induction (RSI) with cricoid pressure and endotracheal intubation rather than supraglottic airway devices (like laryngeal mask airways), and positioning the patient in a slight reverse Trendelenburg position to reduce reflux risk.
Emergency procedures: For true emergencies, the procedure cannot be delayed for GLP-1 washout. All patients on GLP-1 therapy presenting for emergency surgery should be treated as having a full stomach. RSI with cricoid pressure is standard. Postoperative monitoring for aspiration should be heightened.
Endoscopic procedures: Upper endoscopy (EGD) and colonoscopy both carry aspiration risk with sedation. For upper endoscopy, the delayed gastric emptying is directly relevant because retained food in the stomach both increases aspiration risk and impairs visualization. For colonoscopy, the main concern is that delayed gastric emptying can interfere with bowel preparation efficacy, leading to inadequate prep and potentially requiring repeat procedures. Some gastroenterologists recommend extended bowel preparation protocols for patients on GLP-1 therapy.
Practical Preoperative Checklist
- Notify your surgeon and anesthesiologist that you are on GLP-1 therapy at the time of surgical scheduling, not at the preoperative visit.
- Follow the recommended holding period for your specific medication (daily vs. weekly formulation).
- Report any GI symptoms (nausea, vomiting, bloating, early satiety) at your preoperative evaluation, as these may indicate retained gastric contents.
- Consider point-of-care gastric ultrasound on the day of surgery to assess gastric volume. This bedside test takes less than 5 minutes and can identify patients with significant retained contents who need modified anesthesia techniques.
- Resume GLP-1 therapy only after you've resumed normal oral intake postoperatively, typically 24-48 hours after uncomplicated surgery.
Emerging Side Effects from Post-Marketing Surveillance
Clinical trials, even large ones, cannot detect rare adverse events that occur in fewer than 1 in 1,000 patients. As millions of patients use GLP-1 receptor agonists in real-world settings, new safety signals are emerging that weren't apparent in the controlled trial environment. Some of these signals are confirmed concerns; others are still under investigation.
Intestinal Obstruction and Ileus
Post-marketing reports have identified a potential association between GLP-1R agonists and intestinal obstruction or ileus (temporary cessation of intestinal motility). The FDA's Adverse Event Reporting System (FAERS) database contains a higher-than-expected number of intestinal obstruction reports for semaglutide and tirzepatide. However, interpreting FAERS data is complicated by reporting bias: media coverage of GLP-1 side effects has increased reporting rates, and patients with obesity have higher baseline rates of intestinal obstruction due to surgical adhesions, hernias, and other risk factors.
The biological plausibility is clear: GLP-1R agonists slow GI motility throughout the entire gastrointestinal tract, not just the stomach. In patients with pre-existing conditions that narrow the intestinal lumen (adhesions from prior surgery, hernias, strictures from inflammatory bowel disease), further slowing of motility could precipitate obstruction. Patients with a history of abdominal surgery or GI structural abnormalities should be counseled about this potential risk and instructed to seek immediate medical attention for severe abdominal pain, distension, vomiting, or inability to pass gas or stool.
Suicidal Ideation Reports
The European Medicines Agency (EMA) initiated a review in 2023 of reports of suicidal ideation and self-harm associated with GLP-1R agonists. As of early 2026, both the EMA and FDA investigations have not found a causal association between GLP-1R agonists and suicidality. Large epidemiological studies using health system databases have shown no increased risk of depression, anxiety, or suicidal behavior with GLP-1R agonist use, and some studies suggest improved mental health outcomes associated with weight loss.
However, the psychological dimensions of rapid, substantial weight loss deserve ongoing attention. Changes in body image, altered social dynamics, reduced food-related coping mechanisms, and the stress of managing a chronic medication regimen can all contribute to psychological disturbance in vulnerable individuals. Patients with pre-existing mood disorders should be monitored for changes in mental health during GLP-1 therapy, with proactive referral to behavioral health support when needed.
Alopecia (Hair Loss)
Hair thinning and hair loss have been reported by patients on GLP-1 therapy at rates exceeding placebo in some trials. In the SURMOUNT-1 trial, alopecia was reported by 5.7% of patients on tirzepatide 15 mg compared to 1.0% on placebo. For semaglutide, hair loss rates were less well-documented in the STEP trials but have been frequently reported in post-marketing experience.
The mechanism is likely telogen effluvium, a form of diffuse hair loss triggered by physiological stress, including rapid weight loss, nutritional deficiency, and hormonal changes. Telogen effluvium typically occurs 2-4 months after the triggering event and resolves over 6-12 months even without treatment. Nutritional factors may contribute: protein deficiency (from reduced food intake), iron deficiency, zinc deficiency, and biotin deficiency can all exacerbate hair loss during weight loss.
Management strategies include ensuring adequate protein intake (at least 60-80 g/day, ideally 1.2-1.6 g/kg/day), supplementing iron if ferritin is below 40 ng/mL, checking and correcting vitamin D and zinc levels, and potentially slowing the rate of weight loss by reducing the GLP-1R agonist dose if hair loss is severe. Minoxidil (topical or oral) may help if hair loss is persistent, and dermatology referral is appropriate for severe or prolonged cases.
Peptides like GHK-Cu have been studied for their hair growth-stimulating properties, working through increased Wnt signaling and enhanced follicular blood flow. While GHK-Cu is primarily studied for skin applications, its hair follicle effects make it an interesting complementary consideration for GLP-1 users experiencing hair thinning. Visit our peptide research hub for more on GHK-Cu's mechanisms.
Injection Site Reactions and Skin Effects
Injection site reactions occur in approximately 3-7% of patients using injectable GLP-1R agonists. These typically manifest as erythema (redness), induration (hardening), pruritis (itching), or mild pain at the injection site. Most reactions are mild and self-limiting, resolving within 24-48 hours. Rotating injection sites between the abdomen, thigh, and upper arm reduces the frequency and severity of local reactions.
A less common but more persistent issue is the development of injection site lipodystrophy, which can manifest as either lipohypertrophy (localized fat accumulation) or lipoatrophy (localized fat loss) at frequently used injection sites. This is the same phenomenon seen with chronic insulin injection and is prevented by consistent site rotation. Patients should use each injection site no more than once per month and should not inject into areas of existing lipodystrophy.
Post-Marketing Monitoring Recommendations
Given the evolving safety landscape, the following monitoring recommendations supplement the standard care provided during GLP-1 therapy:
- Ask about GI symptoms at every visit, not just nausea but also constipation, bloating, and abdominal pain, which could indicate more serious complications.
- Screen for mood changes using validated instruments (PHQ-9 for depression, GAD-7 for anxiety) at baseline and every 3-6 months.
- Monitor for hair loss and address nutritional deficiencies proactively.
- Review all concurrent medications at each visit, adjusting doses and monitoring as needed based on evolving drug interaction data.
- Stay current with FDA and EMA safety communications, as the post-marketing safety profile is still being defined.
For a thorough review of GLP-1 therapy management, visit our GLP-1 research hub and GLP-1 information center. The personalized assessment can help match you with the approach most suited to your individual health profile and risk factors.
Managing GLP-1 Side Effects with Complementary Approaches
While dose titration and lifestyle adjustments remain the primary strategies for managing GLP-1 side effects, a growing number of clinicians are exploring complementary approaches that target specific side effect mechanisms. Some of these approaches involve peptides that address gut health, inflammation, or nutritional status through pathways independent of GLP-1 receptor signaling.
Gut Health Support During GLP-1 Therapy
The gastrointestinal side effects of GLP-1 therapy (nausea, constipation, diarrhea, bloating) reflect the altered gut motility and digestive function produced by GLP-1R activation. While these effects typically improve with dose stabilization, some patients experience persistent GI discomfort that limits adherence and quality of life.
BPC-157 (Body Protection Compound-157) is a pentadecapeptide originally isolated from human gastric juice that has demonstrated cytoprotective and healing effects throughout the gastrointestinal tract. In preclinical models, BPC-157 accelerates healing of gastric and intestinal mucosal lesions, reduces inflammation in the gut wall, and modulates the NO system and prostaglandin pathways. For patients experiencing GLP-1-related GI irritation, BPC-157's gut-protective effects could theoretically provide symptomatic relief by supporting mucosal integrity and reducing inflammatory signaling in the gut wall.
KPV (Lys-Pro-Val), a tripeptide fragment of alpha-melanocyte-stimulating hormone, has potent anti-inflammatory effects in the intestinal mucosa. KPV inhibits NF-kB activation in intestinal epithelial cells and lamina propria immune cells, reducing the production of pro-inflammatory cytokines that contribute to GI symptoms. For patients with GLP-1-related GI inflammation, KPV could address the inflammatory component of their symptoms through a mechanism entirely distinct from GLP-1R modulation.
Gut microbiome support through targeted probiotics may also help manage GI side effects. Strains like Lactobacillus rhamnosus GG, Bifidobacterium lactis, and Saccharomyces boulardii have evidence for reducing medication-associated GI symptoms. Their benefits include maintaining beneficial bacterial populations during the dietary changes associated with GLP-1 therapy, supporting normal bowel function, and reducing gas production and bloating.
Lean Mass Preservation Strategies
The 25-40% lean mass component of weight loss on GLP-1 therapy is one of the most significant concerns for long-term health outcomes. While resistance training and adequate protein intake are the primary interventions, peptide-based approaches to muscle preservation are gaining clinical interest.
CJC-1295/Ipamorelin stimulates endogenous growth hormone release, which promotes protein synthesis in skeletal muscle and preferential fat oxidation. Growth hormone's anabolic effects could theoretically counterbalance the muscle-wasting tendency of caloric restriction during GLP-1 therapy. The combination provides appetite suppression and metabolic improvement (GLP-1) alongside anabolic muscle support (GH secretagogues), addressing both sides of the body composition equation.
MK-677 (ibutamoren) is an oral growth hormone secretagogue that increases GH and IGF-1 levels through ghrelin receptor agonism. Its oral administration and once-daily dosing make it convenient to combine with GLP-1 therapy. However, MK-677 also stimulates appetite through its ghrelin receptor effects, which could partially counteract GLP-1-mediated appetite suppression. This pharmacological tension between the two mechanisms requires careful dose finding and monitoring. Some clinicians use lower MK-677 doses (10-15 mg rather than 25 mg) when combining with GLP-1 therapy to capture the GH-mediated anabolic effects while minimizing appetite stimulation.
TB-500 (Thymosin Beta-4) promotes tissue repair through enhanced cell migration, angiogenesis, and anti-inflammatory effects. While not directly anabolic for skeletal muscle, TB-500's tissue repair properties could support recovery from resistance training during GLP-1 therapy, allowing more effective training sessions and better lean mass preservation indirectly.
Skin and Hair Health During Weight Loss
Rapid weight loss can produce visible changes in skin and hair that, while not medically dangerous, significantly affect quality of life and treatment satisfaction. Skin laxity from rapid fat loss, hair thinning from nutritional stress, and changes in skin texture from altered hormonal profiles are common concerns among patients achieving 15-25% weight loss.
GHK-Cu (copper peptide) stimulates collagen and elastin synthesis, promotes wound healing, and upregulates thousands of genes involved in tissue repair and regeneration. For patients experiencing skin laxity during GLP-1-mediated weight loss, GHK-Cu's matrix-rebuilding effects could support skin contraction and firmness. Both topical and injectable GHK-Cu formulations are available, with the injectable form providing more systemic exposure and potentially greater effects on deep dermal structures.
For hair thinning, which affects 5-10% of GLP-1 therapy patients (likely through telogen effluvium from rapid weight loss and nutritional stress), GHK-Cu's hair growth-stimulating properties (through Wnt signaling activation in hair follicles) and topical application to the scalp may provide support. Ensuring adequate iron, zinc, biotin, and protein intake is the foundation of hair health management, with peptide approaches serving as complementary support.
The peptide research hub provides detailed information on all of these complementary peptide approaches, and the dosage calculator can help explore appropriate dosing parameters.
Detailed Side Effect Comparison: Drug-by-Drug Clinical Trial Data
Side effect rates vary meaningfully across different GLP-1 agonists, and these differences should factor into agent selection, especially for patients who have experienced side effects on one agent and are considering switching to another. This section provides a detailed, drug-by-drug analysis of side effect data from the major clinical trial programs.
Semaglutide (Ozempic/Wegovy) Side Effect Profile
Semaglutide has the most extensive side effect database among GLP-1 agonists, drawn from the SUSTAIN, PIONEER, STEP, and SELECT trial programs enrolling over 30,000 patients. At the highest approved dose for weight management (2.4 mg weekly), the STEP 1 trial reported: nausea 44.2% (vs. 17.4% placebo), diarrhea 30.0% (vs. 15.8%), vomiting 24.8% (vs. 6.2%), constipation 24.2% (vs. 10.1%), abdominal pain 7.3% (vs. 5.2%), dyspepsia 8.0% (vs. 2.0%), and flatulence 5.1% (vs. 2.1%). Treatment discontinuation due to adverse events was 7.0% (vs. 3.1% placebo).
At the lower doses used for diabetes (0.5-1.0 mg weekly), GI side effect rates are substantially lower: nausea approximately 20-25%, diarrhea 12-18%, vomiting 5-10%, and constipation 5-10%. This dose-dependence of side effects is important because it means that patients who cannot tolerate the 2.4 mg weight loss dose may still benefit from lower doses with significantly reduced GI burden, accepting somewhat less weight loss in exchange for better tolerability.
Semaglutide has a unique side effect consideration in its oral formulation (Rybelsus). Oral semaglutide causes slightly different GI effects than injectable: nausea rates are similar, but the oral formulation can also cause gastric discomfort and eructation (belching) related to the SNAC absorption enhancer. The strict fasting requirement (30 minutes before food, limited water) also creates practical side effects including morning routine disruption and interactions with other morning medications.
Tirzepatide (Mounjaro/Zepbound) Side Effect Profile
Tirzepatide's dual GLP-1/GIP mechanism might be expected to produce more GI side effects than GLP-1 alone, but the clinical data tell a more nuanced story. In the SURMOUNT-1 trial at the 15 mg dose: nausea 26.6% (vs. 4.1% placebo), diarrhea 21.2% (vs. 8.9%), vomiting 11.8% (vs. 1.8%), constipation 11.0% (vs. 4.1%), and dyspepsia 5.7% (vs. 2.5%). Treatment discontinuation due to adverse events was 6.2% (vs. 2.6% placebo).
Comparing directly: tirzepatide 15 mg produces less nausea (26.6% vs. 44.2%) and less vomiting (11.8% vs. 24.8%) than semaglutide 2.4 mg, despite producing more weight loss (20.9% vs. 14.9%). This favorable side effect-to-efficacy ratio is one of tirzepatide's competitive advantages. The GIP receptor component may actually attenuate some of the GI effects of GLP-1 agonism, as GIP signaling can modulate gastric acid secretion and motility through mechanisms that partially counterbalance GLP-1's gastric-slowing effects.
Liraglutide (Saxenda/Victoza) Side Effect Profile
Liraglutide at the 3.0 mg daily dose for obesity (Saxenda) produces: nausea 39.3% (vs. 14.2% placebo), diarrhea 20.9% (vs. 15.1%), vomiting 15.7% (vs. 3.9%), constipation 19.4% (vs. 8.5%), and dyspepsia 9.5% (vs. 2.7%). The daily dosing of liraglutide means that GI side effects occur at a steady-state frequency rather than the weekly peak-and-trough pattern seen with semaglutide and tirzepatide. Some patients prefer this consistent pattern, while others find the relentless daily nausea more challenging than the periodic peaks of weekly agents.
Liraglutide at the lower diabetes dose (1.8 mg daily, Victoza) has substantially lower GI side effect rates: nausea approximately 18-25%, diarrhea 10-15%, vomiting 5-10%. For patients who are very sensitive to GI effects, starting with liraglutide at low doses (0.6-1.2 mg) provides a gentle introduction to GLP-1 therapy that can be used as a stepping stone before transitioning to more potent agents. The dosing calculator can help providers plan liraglutide-to-semaglutide or liraglutide-to-tirzepatide transitions.
Dulaglutide (Trulicity) Side Effect Profile
Dulaglutide consistently shows the lowest GI side effect rates among weekly GLP-1 agonists. At the highest approved dose (4.5 mg weekly): nausea 12.4% (vs. 5.2% placebo), diarrhea 10.6% (vs. 7.0%), vomiting 6.5% (vs. 2.1%), and constipation 5.1% (vs. 3.4%). These rates are roughly half those of semaglutide at the equivalent diabetes dose, making dulaglutide the most GI-friendly option for patients who are particularly sensitive to nausea and vomiting.
The tradeoff is efficacy: dulaglutide produces less weight loss (approximately 3-5% at the 4.5 mg dose) and less HbA1c reduction (approximately 1.1-1.5%) than semaglutide or tirzepatide. For patients whose primary goal is glucose control with minimal GI disruption, dulaglutide is an excellent choice. For patients seeking significant weight loss, dulaglutide's GI advantage comes at too great an efficacy cost. Understanding these tradeoffs is essential for informed agent selection. The GLP-1 research hub provides detailed comparison tools to help patients and providers weigh efficacy against tolerability for each available agent.
Side Effect Variations Across Special Populations
The side effect profiles reported in clinical trials represent population averages, but individual experiences vary enormously based on age, sex, comorbidities, concurrent medications, and genetic factors. Understanding how different patient populations experience GLP-1 side effects helps providers personalize management strategies and set realistic expectations for each patient.
Women vs. Men: Sex-Based Differences in GI Tolerance
Across all GLP-1 clinical trial programs, women consistently report higher rates of GI side effects than men. In the STEP trials, women experienced nausea at rates approximately 30% higher than men at the same semaglutide doses, and vomiting rates were roughly 40% higher. This sex-based difference persists after controlling for body weight, BMI, and dose on a mg/kg basis, suggesting a genuine biological difference rather than a dosing artifact.
Several factors likely contribute to this disparity. Estrogen modulates gastric motility through effects on smooth muscle contraction and vagal tone, and women already have slower baseline gastric emptying than men. GLP-1-induced gastric slowing on top of an already slower baseline may push women past the threshold for nausea and discomfort more readily. Progesterone also slows gastric motility, which means that premenopausal women may experience fluctuating GI tolerance throughout the menstrual cycle, with worse symptoms during the luteal phase when progesterone is highest.
Practical implications for women include: consider starting at the lowest possible dose with extended titration intervals (6-8 weeks per step instead of 4), schedule dose escalation during the follicular phase (when progesterone is low) rather than the luteal phase, be more aggressive with prophylactic anti-nausea strategies from the outset, and track symptoms relative to the menstrual cycle to identify patterns that can inform timing adjustments. Women on hormonal contraceptives or hormone replacement therapy may experience additional gastric motility effects that compound GLP-1 side effects.
Elderly Patients: Dehydration and Fall Risk
Patients over 65 face a different side effect calculus than younger adults. The GI side effects themselves (nausea, vomiting, diarrhea) may be less frequent in elderly patients (some trials suggest slightly lower GI side effect rates in older subgroups), but the consequences of these side effects are more severe. Dehydration from vomiting or diarrhea in a 75-year-old with baseline renal impairment and on antihypertensive medications can rapidly progress to acute kidney injury, orthostatic hypotension, and falls.
Falls are the leading cause of injury-related death in adults over 65, and any medication that increases fall risk demands careful consideration. GLP-1 agonists can increase fall risk through several mechanisms: dehydration-induced orthostatic hypotension, rapid weight loss leading to sarcopenia and balance impairment, hypoglycemia when combined with insulin or sulfonylureas, and general malaise from GI side effects reducing physical activity and muscle conditioning. Providers managing elderly patients on GLP-1 therapy should proactively address hydration, monitor orthostatic blood pressure, ensure adequate protein intake and resistance exercise, and consider lower target doses that provide metabolic benefit with reduced side effect burden.
The lean mass loss concern deserves special emphasis in elderly patients. While younger patients can tolerate the 25-30% lean mass fraction of total weight lost during GLP-1 therapy, elderly patients with already-depleted muscle reserves cannot afford the same proportional loss. Some clinicians use CJC-1295/Ipamorelin or sermorelin as adjunctive therapy to support GH-mediated lean mass preservation in elderly GLP-1 users, though this combination has not been formally studied. The GLP-1 research hub provides detailed guidance on lean mass preservation strategies during GLP-1 therapy.
Patients with Pre-Existing GI Conditions
Patients with irritable bowel syndrome (IBS), inflammatory bowel disease (IBD), gastroparesis, or gastroesophageal reflux disease (GERD) present particular challenges for GLP-1 therapy. These conditions involve baseline abnormalities in GI motility, sensitivity, and inflammation that GLP-1 agonists can exacerbate.
IBS patients may experience worsened symptoms regardless of their IBS subtype. IBS-D (diarrhea-predominant) patients might paradoxically benefit from GLP-1-induced slowed transit, but they may also experience more nausea and bloating. IBS-C (constipation-predominant) patients are at higher risk for severe constipation and should have proactive bowel management protocols (fiber, magnesium, polyethylene glycol) in place before starting GLP-1 therapy.
Gastroparesis patients are generally considered poor candidates for GLP-1 therapy because the further slowing of already-impaired gastric emptying can precipitate gastric outlet obstruction, bezoar formation, and severe nutrient malabsorption. If GLP-1 therapy is deemed necessary in a patient with mild gastroparesis, very low doses with close monitoring and prokinetic support (metoclopramide, domperidone where available) may be attempted, but patients should be counseled about the increased risk.
GERD can paradoxically improve or worsen with GLP-1 therapy. Slowed gastric emptying reduces gastric volume and acid production, which can decrease reflux episodes. However, the nausea and retching associated with GLP-1 initiation can transiently worsen GERD symptoms. Some practitioners report that BPC-157, with its gastroprotective and anti-inflammatory GI effects, helps manage GERD symptoms during GLP-1 therapy, though this combination lacks formal clinical evidence.
Long-Term Side Effects: What the Extended Data Show
As GLP-1 agonists have been on the market for over a decade (liraglutide since 2010, semaglutide since 2017), the accumulating long-term safety data provide important insights that weren't available from the initial approval trials. This extended surveillance reveals both reassuring patterns and emerging concerns that inform the ongoing risk-benefit assessment.
Thyroid Safety: A Decade of Surveillance
The rodent thyroid C-cell tumor signal that prompted the boxed warning on all GLP-1 agonists has now been monitored in human populations for over 15 years. The accumulated evidence is reassuring: large pharmacoepidemiologic studies using claims databases covering millions of patient-years of GLP-1 exposure have not detected an increased incidence of medullary thyroid carcinoma (MTC) or any thyroid cancer subtype. The SUSTAIN 6 and PIONEER 6 trials for semaglutide, the LEADER trial for liraglutide, and the SELECT trial (5 years of follow-up) all showed thyroid cancer rates consistent with background population rates.
The mechanistic explanation for the species difference is now well understood: rodent thyroid C-cells express high densities of GLP-1 receptors and are exquisitely sensitive to GLP-1-mediated mitogenic signaling, while human C-cells express GLP-1 receptors at much lower density and show minimal proliferative response to GLP-1 agonism. The contraindication for MTC/MEN2 family history remains in place as a regulatory precaution, but the biological evidence strongly suggests that GLP-1 agonists do not cause thyroid cancer in humans.
Pancreatitis and Pancreatic Cancer: Updated Evidence
The pancreatitis concern was one of the earliest safety questions raised about GLP-1 therapy, based on post-marketing reports of acute pancreatitis in patients on exenatide and liraglutide. After more than a decade of large-scale clinical trial data and pharmacoepidemiologic studies, the evidence is now fairly clear: GLP-1 agonists do not meaningfully increase pancreatitis risk above baseline rates in the at-risk population (patients with diabetes and obesity already have 2-3x higher pancreatitis rates than the general population, confounding early post-marketing signals).
The SELECT trial, with over 17,000 patients followed for a median of 3.4 years, showed pancreatitis rates of 0.2% in both the semaglutide and placebo groups, providing the most definitive evidence to date that semaglutide does not increase pancreatitis risk. Similarly, pancreatic cancer rates were not elevated in any large GLP-1 trial, and a meta-analysis of all GLP-1 cardiovascular outcome trials found no signal for increased pancreatic malignancy (HR 0.94, 95% CI 0.52-1.68).
Despite this reassuring data, clinical vigilance remains appropriate. Patients should be counseled to report persistent severe abdominal pain (especially with radiation to the back), and providers should check lipase levels if pancreatitis is clinically suspected. Patients with a history of pancreatitis can use GLP-1 agonists with appropriate monitoring, though those with recurrent pancreatitis or known pancreatic duct abnormalities warrant extra caution.
Emerging Concerns: Suicidal Ideation and Facial Aging
Two newer post-marketing concerns have received significant media attention. The first is a potential association between GLP-1 agonists and suicidal ideation. The EMA initiated a safety review in 2023 after receiving reports of suicidal thoughts and self-harm in patients on GLP-1 therapy. However, the evidence to date does not support a causal relationship. The SELECT trial included standardized psychiatric assessments (PHQ-9 and C-SSRS) and found no difference in depression or suicidal ideation scores between semaglutide and placebo groups. The reported cases may reflect the high baseline rates of depression in the obese population, the psychological effects of rapid body changes, or confounding by other psychiatric medications.
The second emerging concern is "Ozempic face," the colloquial term for facial volume loss that accompanies significant weight loss on GLP-1 therapy. This isn't a side effect of the drug per se but rather a consequence of rapid fat loss from a fat depot (buccal fat pad, periorbital fat) that is disproportionately visible. Facial volume loss occurs with any weight loss method that produces 10%+ body weight reduction, including bariatric surgery, and it's more pronounced in older patients who have less skin elasticity to accommodate the reduced facial volume. Dermal fillers and fat grafting can address the cosmetic concern, but the best prevention is a moderate rate of weight loss (0.5-1.0 kg per week rather than 1.5-2.0 kg per week) that allows the skin to gradually adapt. The GHK-Cu peptide has been explored by some practitioners for its collagen-stimulating properties to support skin quality during weight loss, though evidence for this specific application is limited.
For comprehensive, regularly updated information on GLP-1 side effect management, the GLP-1 research hub tracks post-marketing safety data and provides evidence-based management strategies as new information becomes available. The dosing calculator can help patients and providers optimize dose titration to minimize side effect risk while maintaining therapeutic efficacy.
Practical Side Effect Management Protocols: Evidence-Based Strategies for Common Complaints
While understanding the mechanisms behind GLP-1 side effects is valuable, patients and providers need actionable management protocols that can be implemented immediately when symptoms arise. This section provides specific, step-by-step approaches to the most commonly encountered side effects, organized by symptom and severity level.
Nausea Management: A Tiered Approach
Nausea is the most common reason patients contact their provider during GLP-1 dose escalation, and a structured management approach prevents unnecessary discontinuation. The first tier of management involves dietary modifications: eating smaller, more frequent meals (5-6 per day rather than 2-3 large meals), avoiding high-fat and fried foods that slow gastric emptying further, choosing bland, room-temperature or cool foods over hot meals (as heat can intensify nausea), and eating slowly with thorough chewing. Ginger in various forms, including ginger tea, ginger chews, and ginger capsules (250 mg four times daily), has modest evidence for anti-emetic effects and carries minimal risk.
The second tier introduces timing modifications. Some patients find that taking their weekly semaglutide or tirzepatide injection in the evening before bed, rather than in the morning, allows them to sleep through the peak nausea window. Others benefit from injecting on a day when they have lighter schedules and can rest if needed. Staying upright for at least 30 minutes after eating and avoiding lying flat immediately after meals helps prevent nausea triggered by gastroesophageal reflux.
The third tier involves pharmacological management. Over-the-counter options include vitamin B6 (pyridoxine) 25 mg three times daily, which has established anti-emetic properties, particularly for hormonal nausea. For more persistent symptoms, prescription anti-emetics may be warranted: ondansetron (Zofran) 4-8 mg as needed is effective for most patients, while promethazine (Phenergan) 12.5-25 mg can be used for refractory cases with the understanding that it causes drowsiness. Metoclopramide should generally be avoided in GLP-1-treated patients because it accelerates gastric emptying, which partially counteracts the GLP-1 mechanism and may increase GI cramping.
If nausea remains intolerable despite these interventions, the dose-escalation schedule should be modified. Extending each dose step by 2-4 additional weeks allows more gradual receptor adaptation and significantly reduces nausea severity. Some clinicians use half-step dosing, for example going from 0.25 mg to 0.375 mg semaglutide before moving to 0.5 mg, by having compounding pharmacies prepare intermediate-dose formulations. This approach is particularly valuable for patients who report severe nausea at each dose increase.
Constipation Management
Constipation affects 10-20% of GLP-1 users and tends to be more persistent than nausea, often continuing beyond the dose-escalation phase. The underlying mechanism involves slowed colonic transit secondary to GLP-1's effects on gut motility. First-line management includes adequate hydration (targeting at least 64 oz of water daily, which many GLP-1 users underachieve due to reduced thirst alongside reduced appetite), increased fiber intake (targeting 25-35 grams daily through food or supplementation), and regular physical activity (which stimulates colonic motility independently).
For fiber supplementation, psyllium husk (Metamucil) or methylcellulose (Citrucel) are preferred over stimulant laxatives because they add bulk and draw water into the stool without creating dependence. Patients should increase fiber gradually (adding 5 grams per week) to avoid bloating and gas, which can compound the GI discomfort already present from GLP-1 therapy. Magnesium supplementation, particularly magnesium oxide 400-800 mg or magnesium citrate 200-400 mg daily, serves double duty as both an osmotic laxative and a mineral supplement that many GLP-1 users need anyway.
For refractory constipation, polyethylene glycol (MiraLAX) 17 grams daily can be used safely on a longer-term basis. Stimulant laxatives like bisacodyl or senna should be reserved for occasional use only, as chronic use can lead to colonic dependence and electrolyte imbalances. Patients who develop constipation alternating with diarrhea, or who experience severe bloating and abdominal distension, should be evaluated for gastroparesis or small intestinal bacterial overgrowth (SIBO), which can occur or worsen with altered GI motility.
Fatigue and Energy Management
Fatigue is reported by 10-15% of GLP-1 users and is often underrecognized because it isn't tracked as prominently as GI symptoms in clinical trials. The causes are frequently multifactorial: caloric deficit from reduced food intake, dehydration from inadequate fluid consumption, nutritional deficiencies (particularly iron, B12, and vitamin D), and sometimes excessive weight loss causing metabolic adaptation. Unlike nausea, which tends to improve with time, fatigue can persist or worsen if the underlying causes aren't addressed.
The first step in managing GLP-1-related fatigue is ensuring adequate caloric intake. Some patients reduce their intake so dramatically on GLP-1 therapy that they develop symptoms of frank caloric insufficiency. A minimum of 1,200 calories daily for women and 1,500 for men is generally recommended, even during active weight loss. Protein-forward meal planning helps maintain energy levels while supporting lean mass preservation. If caloric intake falls below these minimums consistently, dose reduction may be necessary.
Hydration assessment is equally important. Many patients on GLP-1 therapy report reduced thirst alongside reduced appetite, leading to inadvertent dehydration that manifests as fatigue, lightheadedness, and difficulty concentrating. Setting specific hydration goals and using reminder apps or water tracking bottles can help ensure adequate fluid intake. For patients who struggle with plain water, herbal teas, sugar-free electrolyte beverages, and water flavored with citrus or cucumber provide variety while maintaining hydration.
Laboratory evaluation for fatigue should include CBC (checking for anemia), iron studies, vitamin B12 and folate, vitamin D, thyroid function, and comprehensive metabolic panel (checking electrolytes and kidney function). Addressing identified deficiencies often resolves fatigue symptoms more effectively than any single intervention. For patients whose fatigue persists despite nutritional optimization, evaluation for sleep disorders is warranted, particularly obstructive sleep apnea, which is common in the obesity patient population and may improve but not fully resolve with weight loss. Complementary approaches to energy optimization include peptides like NAD+, which supports cellular energy metabolism, and MOTS-c, which has been studied for its effects on mitochondrial function and exercise capacity.
Hair Loss: Prevention and Treatment
Telogen effluvium, a temporary pattern of diffuse hair shedding, affects approximately 3-6% of patients on GLP-1 therapy and typically begins 2-4 months after starting treatment, correlating with the period of most rapid weight loss. The mechanism is metabolic stress-induced disruption of the hair growth cycle, the same pattern seen after any significant physiological stress including surgery, pregnancy, or crash dieting.
Preventive strategies focus on nutritional adequacy. Ensuring sufficient protein intake (at least 60-80 grams daily minimum), adequate iron stores (target ferritin above 40 ng/mL), zinc supplementation (30 mg daily), and biotin supplementation (5,000 mcg daily) provides the nutritional foundation for hair follicle health. These interventions work best when started proactively at GLP-1 initiation rather than reactively after hair loss begins, since the hair growth cycle operates on a 3-4 month delay between nutritional insult and visible shedding.
For patients who develop noticeable hair shedding, reassurance that the condition is typically self-limiting (resolving within 6-12 months even with continued therapy) helps prevent panic-driven discontinuation. If shedding is severe or prolonged, dermatological evaluation can rule out concurrent alopecia areata, hormonal contributions (thyroid dysfunction, androgen excess), or nutritional deficiency that may be compounding the telogen effluvium. Topical minoxidil 5% can accelerate recovery of hair density and is safe to use alongside GLP-1 therapy. Peptides with skin and tissue repair properties, such as GHK-Cu, have been studied for their potential effects on hair follicle health, though clinical evidence for this specific application remains preliminary. The FormBlends science page provides additional context on supporting overall tissue health during weight loss therapy.
Injection Site Reactions: Best Practices
Injection site reactions, including redness, swelling, itching, and pain at the injection location, affect approximately 3-8% of GLP-1 users. While rarely serious, they can be uncomfortable and, if poorly managed, can lead to patient reluctance to continue treatment. Proper injection technique is the single most effective prevention strategy. The injection should be administered into the subcutaneous fat layer (not intramuscularly), at a 90-degree angle for patients with adequate subcutaneous tissue or a 45-degree angle for leaner patients. Rotation of injection sites among the abdomen, upper thigh, and upper arm prevents local tissue irritation from repeated injections in the same area.
For patients who experience consistent injection site reactions, allowing the medication to warm to room temperature before injection (removing it from the refrigerator 30-60 minutes beforehand) can reduce discomfort. Applying a cold pack to the injection site for 5 minutes before and after injection numbs the area and reduces local inflammatory response. Patients who develop persistent nodules or lumps at injection sites should avoid that area for at least 4-6 weeks and report the finding to their provider, as lipodystrophy (localized changes in fat tissue) can occur with repeated subcutaneous injections in the same location.
Headache and Dizziness Management
Headaches occur in approximately 10-15% of patients during the first few weeks of GLP-1 therapy and are most commonly attributable to dehydration, blood sugar fluctuations, or caffeine reduction (as patients tend to decrease coffee intake alongside food reduction). Ensuring adequate hydration, maintaining stable blood glucose through regular small meals, and avoiding abrupt caffeine changes address most headache triggers. For persistent headaches, standard analgesics like acetaminophen are appropriate. NSAIDs (ibuprofen, naproxen) should be used cautiously in patients with GI symptoms, as they can exacerbate gastrointestinal irritation.
Dizziness and lightheadedness are typically related to orthostatic hypotension from dehydration or blood pressure reduction. Patients on antihypertensive medications may need dose adjustments as weight loss lowers their blood pressure naturally. A practical approach involves checking blood pressure at each follow-up visit and reducing antihypertensive doses proactively when systolic blood pressure falls below 110 mmHg in previously hypertensive patients. Rising slowly from seated or lying positions, wearing compression stockings, and maintaining fluid intake help manage positional dizziness. Patients who experience recurrent dizziness despite these measures should have a comprehensive cardiovascular evaluation to rule out underlying cardiac rhythm disturbances or structural heart disease that may have been masked by their previous weight.
Acid Reflux and GERD Exacerbation
Gastroesophageal reflux can worsen during GLP-1 therapy due to slowed gastric emptying, which increases gastric volume and pressure. Patients with pre-existing GERD may find their symptoms temporarily worsen during the dose-escalation phase. Management strategies include elevating the head of the bed 6-8 inches, avoiding eating within 3 hours of bedtime, reducing portion sizes, avoiding trigger foods (citrus, tomato, spice, chocolate, coffee, alcohol), and considering proton pump inhibitor (PPI) therapy for patients not already taking one. Patients already on PPIs who develop worsening reflux may benefit from switching to a twice-daily PPI regimen temporarily during the escalation phase. The relationship between GLP-1 therapy and Barrett's esophagus in patients with chronic reflux has not been studied and represents an area where clinical vigilance is appropriate. Patients interested in gastrointestinal mucosal support may explore peptides like BPC-157, which has preclinical evidence for gastroprotective effects, though human clinical trial data are limited.
The overarching principle for managing GLP-1 side effects is proactive, rather than reactive, intervention. Patients who receive thorough counseling about expected side effects before starting therapy, who have a clear management protocol to follow when symptoms arise, and who maintain regular communication with their healthcare team during the dose-escalation phase consistently show higher persistence rates and better outcomes than patients who are simply handed a prescription and told to call if problems develop. The investment in upfront education and structured follow-up pays dividends throughout the treatment course, transforming manageable side effects from potential treatment barriers into expected, temporary inconveniences that resolve with appropriate intervention. Patients beginning their GLP-1 therapy journey through FormBlends receive this structured support framework as a standard component of care, ensuring that side effect management is integrated into the treatment plan from day one rather than addressed only when problems become unmanageable.
Emergency Warning Signs: When GLP-1 Side Effects Require Immediate Medical Attention
While the vast majority of GLP-1 side effects are mild to moderate and manageable in the outpatient setting, certain symptoms require prompt medical evaluation to rule out serious complications. Both patients and providers benefit from a clear framework for distinguishing between expected side effects that can be managed at home and warning signs that warrant emergency assessment.
Pancreatitis Warning Signs
Acute pancreatitis, though rare (occurring in fewer than 0.5% of GLP-1 users in clinical trials), is the most serious GI complication associated with this drug class. The hallmark symptom is severe, persistent epigastric pain that radiates to the back, often accompanied by nausea and vomiting that is qualitatively different from the typical GLP-1 nausea, being more intense and unrelenting. Patients describe this pain as "boring through" to the back, and it typically worsens after eating and when lying flat. Any patient on GLP-1 therapy who develops severe abdominal pain lasting more than several hours should seek emergency evaluation, which will include serum lipase measurement (the most specific marker for pancreatitis), abdominal imaging, and clinical assessment. GLP-1 therapy should be discontinued immediately if pancreatitis is confirmed, and should not be restarted after a confirmed episode.
Gallbladder Disease Symptoms
Gallstone formation and cholecystitis are more common in patients undergoing rapid weight loss from any cause, including GLP-1 therapy. Right upper quadrant abdominal pain, especially if it occurs after fatty meals, intensifies over several hours, and is associated with fever, chills, or jaundice (yellowing of the skin or eyes), suggests possible acute cholecystitis or choledocholithiasis (bile duct stone). These conditions require emergency evaluation, typically including right upper quadrant ultrasound and blood work (CBC, liver function tests, bilirubin). While gallbladder disease does not necessarily require permanent GLP-1 discontinuation (the drug can often be resumed after cholecystectomy), the acute episode needs urgent management. Patients with known gallstones should discuss prophylactic strategies with their provider, including ursodiol therapy, adequate dietary fat intake to maintain gallbladder motility, and more frequent monitoring.
Severe Dehydration and Electrolyte Disturbances
Persistent vomiting or severe diarrhea on GLP-1 therapy can lead to clinically significant dehydration and electrolyte imbalances, particularly hypokalemia (low potassium) and hyponatremia (low sodium). Patients taking diuretics or ACE inhibitors alongside GLP-1 therapy are at heightened risk for electrolyte disturbances. Warning signs include dizziness upon standing, rapid heartbeat, muscle weakness or cramping, dark-colored urine, and confusion or disorientation. Any patient who is unable to keep fluids down for more than 24 hours should contact their provider, and those showing signs of severe dehydration should seek emergency care. IV fluid resuscitation and electrolyte correction may be necessary in severe cases.
Acute kidney injury (AKI) can occur secondary to severe dehydration from GLP-1-related GI side effects. Post-marketing reports have documented cases of AKI in patients on semaglutide and other GLP-1 agonists, primarily in the setting of profound volume depletion from vomiting or diarrhea. Patients with pre-existing chronic kidney disease are at highest risk. Monitoring creatinine levels during episodes of significant GI illness, maintaining hydration, and temporarily reducing or holding GLP-1 doses during acute illness episodes represent prudent risk mitigation strategies.
Allergic and Hypersensitivity Reactions
While rare, allergic reactions to GLP-1 agonists have been reported in post-marketing surveillance. Symptoms ranging from localized injection site reactions (redness, swelling, itching) to systemic anaphylaxis (difficulty breathing, facial swelling, widespread urticaria, rapid pulse) have been documented. Localized injection site reactions are common (occurring in 3-8% of users) and are typically mild and self-limiting, not requiring treatment beyond rotating injection sites and applying cool compresses. Systemic allergic reactions are extremely rare but constitute a medical emergency requiring immediate treatment with epinephrine and emergency medical services. Any patient who develops difficulty breathing, throat tightness, or widespread hives after a GLP-1 injection should use an epinephrine auto-injector if available and call emergency services immediately.
Hypoglycemia in Diabetic Patients
For patients with type 2 diabetes taking GLP-1 agents alongside sulfonylureas or insulin, severe hypoglycemia represents a potentially dangerous complication. While GLP-1 agonists alone rarely cause hypoglycemia (because their insulin-stimulating effects are glucose-dependent), the combination with sulfonylureas or exogenous insulin can produce blood glucose levels low enough to cause confusion, seizures, loss of consciousness, or cardiac arrhythmias. Patients on these combination regimens should have access to glucose tablets or gel, understand the symptoms of hypoglycemia (shakiness, sweating, confusion, rapid heartbeat, hunger), and have a plan for treatment. Proactive reduction of sulfonylurea or insulin doses at GLP-1 initiation, as discussed earlier, is the most effective prevention strategy.
Intestinal Obstruction
Very rarely, the profound slowing of GI motility caused by GLP-1 agents can contribute to intestinal obstruction, particularly in patients with pre-existing adhesions from prior abdominal surgery, hernias, or inflammatory bowel disease. Symptoms of intestinal obstruction include severe cramping abdominal pain, complete inability to pass gas or stool, progressive abdominal distension, and bilious (green) vomiting. This is a surgical emergency requiring immediate evaluation with abdominal imaging and possible operative intervention. GLP-1 therapy should be discontinued immediately, and patients with a history of abdominal surgery or known adhesive disease should be counseled about this risk at treatment initiation.
While these emergency scenarios are individually uncommon, their collective possibility underscores the importance of patient education at the start of GLP-1 therapy. Providers should review warning signs during the initial consultation and ensure that patients have a clear action plan for seeking care if concerning symptoms develop. Written or digital reference materials listing warning signs and provider contact information help patients make appropriate decisions during acute episodes. The GLP-1 research hub provides ongoing updates on safety monitoring recommendations as post-marketing surveillance data continue to accumulate across the class. For patients who prefer a step-by-step reference, printing or bookmarking the warning signs listed in this section creates a ready reference that can be consulted quickly during acute symptom episodes, reducing anxiety and enabling appropriate clinical decision-making when it matters most. Communication with prescribers should never be delayed when genuine warning signs are present, as early intervention consistently produces better outcomes than watchful waiting in the context of potentially serious GLP-1 complications.
Frequently Asked Questions
What are the most common GLP-1 side effects?
The most common side effects of GLP-1 receptor agonists are gastrointestinal in nature. Nausea affects 20-44% of patients depending on the specific drug and dose, making it the single most frequent complaint. Vomiting occurs in 5-25% of users, diarrhea in 8-20%, and constipation in 5-15%. These GI effects are typically most intense during the first 4-8 weeks of treatment and during dose escalation periods. Most patients find that symptoms diminish substantially after the body adjusts to each new dose level. Less common side effects include injection site reactions, headache, fatigue, and dizziness. Serious but rare complications include pancreatitis, gallbladder disease, and potential thyroid concerns.
How do you manage nausea on GLP-1 drugs?
Managing GLP-1-related nausea involves several practical strategies. Eat smaller, more frequent meals rather than large portions. Avoid high-fat and greasy foods, which slow gastric emptying further. Stay well hydrated with small sips throughout the day. Ginger tea, ginger chews, or ginger supplements can provide natural relief. Take your injection in the evening so the peak nausea window occurs during sleep. Follow the recommended dose titration schedule strictly and never skip dose steps. If nausea persists beyond 2-3 weeks at the same dose, your provider may prescribe ondansetron or suggest a slower titration. Peppermint tea and bland foods like crackers or toast can also help.
Do GLP-1 drugs cause pancreatitis?
The relationship between GLP-1 receptor agonists and pancreatitis has been extensively studied. Early case reports raised concern, but large-scale clinical trials and real-world database analyses have found no statistically significant increase in pancreatitis risk. The LEADER trial with liraglutide showed pancreatitis rates of 0.4% in the treatment group versus 0.5% in the placebo group. The SUSTAIN and STEP programs with semaglutide showed similarly low rates. However, patients with a history of pancreatitis should use GLP-1 drugs with caution. If you develop severe, persistent abdominal pain that radiates to the back, stop the medication and contact your doctor immediately.
Is there a thyroid cancer risk with GLP-1 drugs?
GLP-1 receptor agonists carry a boxed warning about medullary thyroid carcinoma (MTC) based on rodent studies that showed C-cell tumors in rats and mice at high doses. However, human thyroid C-cells have far fewer GLP-1 receptors than rodent C-cells, and the clinical relevance of these animal findings remains uncertain. Large-scale human studies including the LEADER trial (over 9,000 patients followed for 3.8 years) have not shown an increased rate of thyroid cancer. GLP-1 drugs are contraindicated in patients with a personal or family history of MTC or Multiple Endocrine Neoplasia syndrome type 2 (MEN2).
Can GLP-1 drugs cause gastroparesis?
GLP-1 receptor agonists slow gastric emptying as part of their mechanism of action, but this is not the same as causing gastroparesis. Clinical gastroparesis involves a chronic, severe delay in gastric emptying with structural or functional impairment. The delayed gastric emptying from GLP-1 drugs is dose-dependent and reversible upon discontinuation. Some patients with pre-existing slow gastric motility may experience more pronounced symptoms. True gastroparesis cases in GLP-1 users are rare and typically resolve after stopping the medication. If you experience persistent vomiting, severe bloating, or an inability to keep food down, consult your healthcare provider.
Do GLP-1 side effects go away over time?
Yes, the majority of GLP-1 side effects diminish over time through a process called tachyphylaxis, where the body adapts to the medication. Nausea typically peaks during the first 1-2 weeks at each new dose level and fades within 4-8 weeks. Data from the STEP trials show that nausea was most common during dose escalation and decreased once patients reached their maintenance dose. About 80% of patients who experience initial nausea report significant improvement by week 8-12. Vomiting and diarrhea follow similar patterns. This is precisely why slow, gradual dose titration is so valuable: it gives your body time to adjust at each level before increasing the dose.
References
- Wilding JPH, Batterham RL, Calanna S, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. New England Journal of Medicine. 2021;384(11):989-1002. DOI: 10.1056/NEJMoa2032183.
- Frias JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. New England Journal of Medicine. 2021;385(6):503-515. DOI: 10.1056/NEJMoa2107519.
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide Once Weekly for the Treatment of Obesity. New England Journal of Medicine. 2022;387(3):205-216. DOI: 10.1056/NEJMoa2206038.
- Pi-Sunyer X, Astrup A, Fujioka K, et al. A Randomized, Controlled Trial of 3.0 mg of Liraglutide in Weight Management. New England Journal of Medicine. 2015;373(1):11-22. DOI: 10.1056/NEJMoa1411892.
- Marso SP, Daniels GH, Tanaka K, et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. New England Journal of Medicine. 2016;375(4):311-322. DOI: 10.1056/NEJMoa1603827.
- Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. The Lancet. 2019;394(10193):121-130. DOI: 10.1016/S0140-6736(19)31149-3.
- Marso SP, Bain SC, Consoli A, et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. New England Journal of Medicine. 2016;375(19):1834-1844. DOI: 10.1056/NEJMoa1607141.
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. New England Journal of Medicine. 2023;389(24):2221-2232. DOI: 10.1056/NEJMoa2307563.
- Sodhi M, Rezaeianzadeh R, Kezouh A, et al. Risk of Gastrointestinal Adverse Events Associated With Glucagon-Like Peptide-1 Receptor Agonists for Weight Loss. JAMA. 2023;330(18):1795-1797. DOI: 10.1001/jama.2023.19574.
- Bjerre Knudsen L, Madsen LW, Andersen S, et al. Glucagon-like peptide-1 receptor agonists activate rodent thyroid C-cells causing calcitonin release and C-cell proliferation. Endocrinology. 2010;151(4):1473-1486. DOI: 10.1210/en.2009-1272.
- Stokes CS, Gluud LL, Casper M, et al. Ursodeoxycholic acid and diets higher in fat prevent gallbladder stones during weight loss: a meta-analysis of randomized controlled trials. Clinical Gastroenterology and Hepatology. 2014;12(7):1090-1100. DOI: 10.1016/j.cgh.2013.11.031.
- Davies MJ, Bergenstal R, Bode B, et al. Efficacy of Liraglutide for Weight Loss Among Patients With Type 2 Diabetes: The SCALE Diabetes Randomized Clinical Trial. JAMA. 2015;314(7):687-699. DOI: 10.1001/jama.2015.9676.
- Rubino D, Abrahamsson N, Davies M, et al. Effect of Continued Weekly Subcutaneous Semaglutide vs Placebo on Weight Loss Maintenance in Adults With Overweight or Obesity: The STEP 4 Randomized Clinical Trial. JAMA. 2021;325(14):1414-1425. DOI: 10.1001/jama.2021.3224.
- Garvey WT, Batterham RL, Bhatt DL, et al. Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nature Medicine. 2022;28(10):2083-2091. DOI: 10.1038/s41591-022-02026-4.
- Jastreboff AM, Kaplan LM, Frias JP, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity - A Phase 2 Trial. New England Journal of Medicine. 2023;389(6):514-526. DOI: 10.1056/NEJMoa2301972.
- Drucker DJ. Mechanisms of Action and Therapeutic Application of Glucagon-like Peptide-1. Cell Metabolism. 2018;27(4):740-756. DOI: 10.1016/j.cmet.2018.03.001.
- Nauck MA, Quast DR, Wefers J, et al. GLP-1 receptor agonists in the treatment of type 2 diabetes - state-of-the-art. Molecular Metabolism. 2021;46:101102. DOI: 10.1016/j.molmet.2020.101102.
- Filippatos TD, Panagiotopoulou TV, Elisaf MS. Adverse Effects of GLP-1 Receptor Agonists. The Review of Diabetic Studies. 2014;11(3-4):202-230. DOI: 10.1900/RDS.2014.11.202.
- Perkovic V, Tuttle KR, Rossing P, et al. Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes. New England Journal of Medicine. 2024;391(2):109-121. DOI: 10.1056/NEJMoa2403347.
- Wadden TA, Bailey TS, Billings LK, et al. Effect of Subcutaneous Semaglutide vs Placebo as an Adjunct to Intensive Behavioral Therapy on Body Weight in Adults With Overweight or Obesity: The STEP 3 Randomized Clinical Trial. JAMA. 2021;325(14):1403-1413. DOI: 10.1001/jama.2021.1831.
- Dungan KM, Povedano ST, Forst T, et al. Once-weekly dulaglutide versus once-daily liraglutide in metformin-treated patients with type 2 diabetes (AWARD-6): a randomised, open-label, phase 3, non-inferiority trial. The Lancet. 2014;384(9951):1349-1357. DOI: 10.1016/S0140-6736(14)60976-4.
- Vilsboll T, Bain SC, Leiter LA, et al. Semaglutide, reduction in glycated haemoglobin and the risk of diabetic retinopathy. Diabetes, Obesity and Metabolism. 2018;20(4):889-897. DOI: 10.1111/dom.13172.
- Friedrichsen M, Breitschaft A, Tadayon S, et al. The effect of semaglutide 2.4 mg once weekly on energy intake, appetite, control of eating, and gastric emptying in adults with obesity. Diabetes, Obesity and Metabolism. 2021;23(3):754-762. DOI: 10.1111/dom.14280.
- Smits MM, Van Raalte DH. Safety of Semaglutide. Frontiers in Endocrinology. 2021;12:645563. DOI: 10.3389/fendo.2021.645563.
- Bettge K, Kahle M, Abd El Aziz MS, et al. Occurrence of nausea, vomiting and diarrhoea reported as adverse events in clinical trials studying glucagon-like peptide-1 receptor agonists: A systematic analysis of published clinical trials. Diabetes, Obesity and Metabolism. 2017;19(3):336-347. DOI: 10.1111/dom.12824.