Executive Summary
The treatment of obesity has entered a new era. What was once dismissed as a lifestyle problem now sits at the center of one of the largest pharmacological arms races in modern medicine. GLP-1 receptor agonists, dual and triple hormone agonists, oral small molecules, and muscle-sparing antibodies are reshaping how clinicians think about excess weight and its consequences.
Key Takeaways
- Six FDA-approved anti-obesity medications are currently available, with injectable GLP-1 agonists showing the strongest efficacy
- Tirzepatide (Zepbound) produces the highest weight loss among approved drugs at approximately 20.9% of body weight
- Pipeline agents including retatrutide (28.7% weight loss), CagriSema (20.4%), and oral orforglipron are approaching regulatory submission
- Bimagrumab combined with semaglutide preserves lean muscle mass while achieving 22.1% fat-dominant weight loss
- The first oral GLP-1 for weight management (semaglutide pill, Wegovy 25mg daily) was FDA-approved in December 2025
This guide covers every FDA-approved anti-obesity medication on the market as of early 2026, along with the most promising pipeline candidates expected to reach patients within the next two to three years. We examine mechanisms of action, clinical trial data, head-to-head comparisons, safety profiles, costs, and practical considerations for selecting the right therapy.
The numbers tell a striking story. Semaglutide (Wegovy) delivers roughly 15% body weight loss. Tirzepatide (Zepbound) pushes that to 21%. Retatrutide, a triple agonist still in Phase 3 trials, has shown weight reductions approaching 29%. Meanwhile, oral agents like orforglipron promise to eliminate the need for injections entirely, and combination strategies pairing GLP-1s with muscle-preserving agents like bimagrumab are redefining what "quality weight loss" means.
Yet these advances come with real challenges. Gastrointestinal side effects remain common. Costs often exceed $1,000 per month without insurance. Weight regain after discontinuation is well-documented. And the sheer number of options now available can make choosing the right medication feel overwhelming for both patients and providers.
Whether you're a researcher tracking the latest trial data, a clinician comparing treatment options, or someone exploring what's available, this guide provides the evidence you need. Every claim is backed by peer-reviewed data, and we've included DOI-linked references throughout so you can verify the source material yourself.
Key Takeaways
- Six FDA-approved anti-obesity medications are currently available, with injectable GLP-1 agonists showing the strongest efficacy
- Tirzepatide (Zepbound) produces the highest weight loss among approved drugs at approximately 20.9% of body weight
- Pipeline agents including retatrutide (28.7% weight loss), CagriSema (20.4%), and oral orforglipron are approaching regulatory submission
- Bimagrumab combined with semaglutide preserves lean muscle mass while achieving 22.1% fat-dominant weight loss
- The first oral GLP-1 for weight management (semaglutide pill, Wegovy 25mg daily) was FDA-approved in December 2025
- Non-GLP-1 approaches like tesofensine, 5-amino-1MQ, and AOD-9604 target distinct metabolic pathways
Figure 1: Overview of FDA-approved and pipeline anti-obesity medications with comparative efficacy data (2024-2025)
The Obesity Epidemic: Scope, Costs, and Comorbidities
Prevalence in the United States
Obesity remains one of the most pressing public health challenges worldwide, and the United States sits at the epicenter. According to CDC data covering August 2021 through August 2023, the prevalence of obesity among U.S. adults stands at 40.3%, with rates of 39.2% in men and 41.3% in women. Severe obesity (BMI of 40 or higher) affects 9.4% of the adult population and has been climbing steadily, rising from 7.7% in 2013-2014 to 9.7% in recent surveys (Ogden et al., 2024).
The trends among children and adolescents are equally concerning. Childhood obesity rates have tripled since the 1970s, with approximately 19.7% of children aged 2-19 now classified as obese. A Lancet analysis forecasts that if current trajectories continue, more than 250 million Americans will have overweight or obesity by 2050 (Ward et al., 2024).
Disparities persist across racial and ethnic groups. Non-Hispanic Black adults have the highest age-adjusted obesity prevalence (49.9%), followed by Hispanic adults (45.6%), non-Hispanic white adults (42.4%), and non-Hispanic Asian adults (16.7%). Geographic variation is also substantial, with Mississippi, West Virginia, and Louisiana consistently reporting the highest state-level rates.
Global Burden
The World Health Organization reports that approximately 16% of adults worldwide were living with obesity in 2022, a figure that has more than doubled since 1990. Over 390 million children and adolescents aged 5 to 19 were overweight in 2022, including 160 million living with obesity. The World Obesity Federation projects that 51% of the global population will have overweight or obesity by 2035.
The fastest rates of increase are occurring in low- and middle-income countries, where the dual burden of malnutrition and obesity creates unique public health challenges. Countries across the Middle East, North Africa, and the Pacific Islands now report adult obesity rates exceeding 30%.
Economic Impact
The financial toll of obesity is staggering. The World Obesity Federation estimates that the economic impact of overweight and obesity will surpass $4 trillion annually by 2035, accounting for nearly 3% of global GDP. In the United States alone, obesity-related medical costs exceed $173 billion per year, with obese individuals paying an average of $1,861 more annually in medical expenses compared to those at healthy weight (Cawley et al., 2024).
Indirect costs compound the economic burden. Lost productivity from absenteeism, presenteeism (reduced on-the-job performance), disability, and premature mortality adds another $66 billion annually. Employers bear a significant share of these costs through higher health insurance premiums and disability claims.
Comorbidities and Health Consequences
Obesity is a risk factor for more than 200 medical conditions. The most significant include:
| Comorbidity Category | Specific Conditions | Risk Increase |
|---|---|---|
| Cardiovascular | Heart disease, stroke, hypertension, heart failure | 2-4x higher risk |
| Metabolic | Type 2 diabetes, metabolic syndrome, dyslipidemia | 5-10x higher for T2D |
| Cancer | Breast, colon, endometrial, kidney, liver, pancreatic | 1.5-3x higher risk |
| Respiratory | Obstructive sleep apnea, asthma, obesity hypoventilation | 3-7x higher for OSA |
| Musculoskeletal | Osteoarthritis, chronic back pain, gout | 2-4x higher risk |
| Hepatic | MASLD/MASH (formerly NAFLD/NASH), cirrhosis | 3-5x higher risk |
| Psychological | Depression, anxiety, eating disorders, reduced quality of life | 1.5-2x higher risk |
| Reproductive | Infertility, PCOS, pregnancy complications | 2-3x higher risk |
The relationship between obesity and mortality follows a J-shaped curve, with both the highest and lowest BMI categories associated with increased death rates. A BMI above 35 is associated with a roughly 29% increase in all-cause mortality, while a BMI above 40 raises that risk by approximately 100% (Global BMI Mortality Collaboration, 2016).
These health consequences create a compelling case for pharmacological intervention. Lifestyle modifications alone produce modest weight loss averaging 3-5% of body weight, often insufficient to meaningfully reduce comorbidity risk. This gap between what lifestyle changes can achieve and what patients need has driven the development of the medications we'll examine in the following sections.
Figure 2: Global and U.S. obesity prevalence trends with economic impact projections
History of Anti-Obesity Pharmacotherapy
The history of weight loss drugs is marked by both remarkable innovation and sobering failures. Understanding this trajectory helps explain why today's medications are held to rigorous safety standards, and why the current generation of GLP-1 agonists represents such a meaningful departure from past approaches.
The Early Era: 1890s-1960s
Physicians first attempted pharmacological weight loss in the late 1800s using thyroid hormone extracts. While thyroid hormones did accelerate metabolism, they came with dangerous side effects including heart palpitations, bone loss, and thyrotoxicosis. Despite these risks, thyroid-based weight loss preparations persisted well into the mid-20th century.
The 1930s introduced 2,4-dinitrophenol (DNP), an industrial chemical that uncoupled oxidative phosphorylation to increase metabolic rate. DNP caused dramatic weight loss but also produced cataracts, skin lesions, and fatal hyperthermia. After several deaths, it was pulled from use by 1938, though it has periodically resurfaced in unregulated supplements.
Amphetamines emerged as weight loss agents in the 1940s. Drugs like Benzedrine and Dexedrine suppressed appetite effectively, but their addictive potential and cardiovascular risks soon became apparent. The FDA approved phentermine in 1959, a milder sympathomimetic amine that remains in use today for short-term weight management.
The Fen-Phen Disaster: 1990s
The combination of fenfluramine and phentermine, nicknamed "fen-phen," became one of the most prescribed weight loss regimens of the 1990s. By 1996, physicians were writing 18 million prescriptions annually for the combination. The drugs worked well together: phentermine suppressed appetite via norepinephrine release while fenfluramine boosted serotonin to promote satiety.
Then came the cardiac findings. In 1997, researchers at the Mayo Clinic reported 24 cases of unusual heart valve disease in women taking fen-phen. The FDA moved quickly, and fenfluramine (along with its derivative dexfenfluramine, marketed as Redux) was withdrawn from the market in September 1997. The manufacturer, Wyeth-Ayerst, ultimately agreed to a $3.75 billion settlement to compensate thousands of affected patients (Connolly et al., 1997).
The fen-phen episode cast a long shadow over obesity pharmacotherapy. It made the FDA far more cautious about approving weight loss drugs, required cardiovascular safety data for new approvals, and created public distrust that persists to this day.
Post-Fen-Phen Setbacks: 2000-2012
The two decades following fen-phen saw several more safety-related withdrawals:
- Sibutramine (Meridia) - Approved in 1997, this serotonin-norepinephrine reuptake inhibitor was withdrawn in 2010 after the SCOUT trial revealed a 16% increase in cardiovascular events compared to placebo (James et al., 2010).
- Rimonabant (Acomplia) - This cannabinoid CB1 receptor antagonist was approved in Europe in 2006 but never in the U.S. It was withdrawn in 2008 due to doubled rates of depression and suicidal ideation.
- Lorcaserin (Belviq) - Approved by the FDA in 2012, this selective serotonin 5-HT2C agonist was voluntarily withdrawn in 2020 after a post-marketing safety trial indicated an increased occurrence of cancer (Bohula et al., 2020).
The Modern Toolkit: 2012-2023
Despite these setbacks, several medications gained and retained FDA approval during this period:
- 2012: Phentermine-topiramate ER (Qsymia) - Combines a sympathomimetic amine with an anticonvulsant to suppress appetite and increase satiety. Produces roughly 9.8% weight loss.
- 2014: Naltrexone-bupropion ER (Contrave) - Pairs an opioid antagonist with an antidepressant to target reward-based eating. Achieves approximately 5.4% weight loss.
- 2014: Liraglutide 3.0mg (Saxenda) - The first GLP-1 receptor agonist approved specifically for weight management, delivering about 8% weight loss in clinical trials.
The GLP-1 Revolution: 2021-Present
The approval of semaglutide 2.4mg (Wegovy) in June 2021 marked a turning point. For the first time, a medication consistently produced double-digit percentage weight loss in clinical trials, approaching what had previously been achievable only through bariatric surgery. The STEP 1 trial demonstrated 14.9% body weight reduction at 68 weeks, dwarfing the efficacy of all prior medications (Wilding et al., 2021).
Tirzepatide (Zepbound), approved for chronic weight management in November 2023, pushed the bar even higher. As the first dual GIP/GLP-1 receptor agonist, it achieved 20.9% weight loss at 72 weeks in the SURMOUNT-1 trial, with some dose groups showing that over a third of participants lost 25% or more of their body weight (Jastreboff et al., 2022).
These medications didn't just reduce weight. The SELECT trial showed semaglutide reduced major adverse cardiovascular events by 20% in patients with established cardiovascular disease, leading to a landmark indication expansion in March 2024. Tirzepatide became the first drug approved for obesity-related obstructive sleep apnea in December 2024.
The GLP-1 class had arrived, and with it, a complete transformation of how the medical community views obesity treatment.

Figure 3: Timeline of anti-obesity pharmacotherapy from early thyroid extracts through the GLP-1 era
Currently FDA-Approved Anti-Obesity Medications
As of early 2026, six medications hold FDA approval for long-term chronic weight management in adults with obesity (BMI 30+) or overweight (BMI 27+) with at least one weight-related comorbidity. Their efficacy, mechanisms, and side effect profiles vary widely.
Comparative Efficacy: All FDA-Approved Anti-Obesity Agents
1. Orlistat (Xenical / Alli)
Mechanism of Action
Orlistat is a gastrointestinal lipase inhibitor. It works in the gut lumen by binding to gastric and pancreatic lipases, preventing these enzymes from breaking down dietary triglycerides into absorbable free fatty acids and monoglycerides. The result: approximately 30% of ingested dietary fat passes through the GI tract unabsorbed.
Unlike every other medication on this list, orlistat has no systemic activity. Less than 1% is absorbed into the bloodstream, which means it has virtually no drug-drug interactions through hepatic metabolism. However, it does reduce absorption of fat-soluble vitamins (A, D, E, K), necessitating supplementation.
Clinical Efficacy
Orlistat produces modest weight loss. In the landmark XENDOS trial, 120mg three times daily (with meals) resulted in 5.8% weight loss at 4 years compared to 3.0% with placebo, a net treatment effect of about 2.8 percentage points. In the same trial, orlistat reduced the incidence of type 2 diabetes by 37% compared to lifestyle changes alone (Torgerson et al., 2004).
A lower-dose version (60mg) is available over the counter as Alli, making it the only non-prescription obesity medication on the market. Its efficacy is correspondingly lower, typically producing 3-4% total body weight loss.
Side Effects and Tolerability
The side effects of orlistat are almost entirely gastrointestinal and directly related to its mechanism. Oily spotting, flatus with discharge, fecal urgency, fatty or oily stools, and increased defecation are common, especially when patients consume high-fat meals. These effects diminish with reduced dietary fat intake, which itself contributes to weight loss.
Rare cases of severe liver injury have been reported, prompting an FDA safety review, though the causal relationship remains uncertain. Orlistat can also cause oxalate nephropathy in susceptible individuals.
Practical Considerations
Orlistat is best suited for patients who prefer a non-systemic medication, those with contraindications to other options, or as an adjunct therapy. Its low efficacy relative to newer agents has reduced its clinical use significantly, but its long safety track record (approved since 1999) and OTC availability keep it relevant for certain populations.
2. Phentermine-Topiramate ER (Qsymia)
Mechanism of Action
This combination medication pairs two drugs with complementary mechanisms. Phentermine is a sympathomimetic amine that stimulates norepinephrine release in the hypothalamus, suppressing appetite through central nervous system activation. Topiramate, originally an anticonvulsant, enhances satiety through multiple pathways including GABA-A receptor modulation, glutamate antagonism, and carbonic anhydrase inhibition. The exact mechanism by which topiramate promotes weight loss isn't fully understood, but its effects on appetite regulation, taste perception, and energy expenditure are well-documented.
The combination allows lower doses of each component than would be needed individually, reducing side effects while maintaining efficacy.
Clinical Efficacy
Qsymia is the most effective non-injectable anti-obesity medication currently approved. In the CONQUER trial, the recommended dose (7.5mg/46mg) produced 7.8% weight loss at 56 weeks, while the top dose (15mg/92mg) achieved 9.8%. In the EQUIP trial studying patients with higher BMIs (mean BMI 42), the top dose produced 10.9% weight loss (Gadde et al., 2011).
Beyond weight loss, Qsymia improves cardiometabolic parameters including blood pressure, triglycerides, HDL cholesterol, waist circumference, and fasting insulin levels. In the SEQUEL extension study, weight loss was maintained at 2 years with continued improvement in metabolic markers.
Side Effects and Safety Concerns
Key side effects include paresthesia (tingling in hands and feet, attributed to topiramate), dry mouth, constipation, insomnia, dizziness, and dysgeusia (altered taste). Cognitive effects such as difficulty with concentration and word-finding can occur, particularly at higher doses.
Qsymia carries important safety warnings. Topiramate is teratogenic, associated with increased risk of cleft lip/palate, making pregnancy testing mandatory before and during treatment. It can also increase resting heart rate by 1-2 beats per minute and cause metabolic acidosis. The medication is available only through a certified REMS pharmacy program.
A generic version of phentermine-topiramate ER became available in 2025, improving affordability.
3. Naltrexone-Bupropion ER (Contrave)
Mechanism of Action
Contrave targets the reward and motivation pathways involved in eating behavior. Bupropion, a dopamine and norepinephrine reuptake inhibitor used as an antidepressant and smoking cessation aid, stimulates pro-opiomelanocortin (POMC) neurons in the hypothalamic arcuate nucleus. These neurons release alpha-MSH, which suppresses appetite. However, POMC neurons also release beta-endorphin, which creates a feedback loop that normally dampens their own activity.
This is where naltrexone comes in. As an opioid receptor antagonist, naltrexone blocks the autoinhibitory effect of beta-endorphin on POMC neurons, allowing sustained appetite suppression. The combination produces greater weight loss than either component alone (Greenway et al., 2010).
Clinical Efficacy
The COR (Contrave Obesity Research) trial program demonstrated modest but consistent efficacy. In COR-I, the standard dose (naltrexone 32mg/bupropion 360mg) produced 6.1% weight loss at 56 weeks versus 1.3% with placebo. COR-II showed 6.4% weight loss. COR-BMOD, which combined the medication with intensive behavioral modification, achieved 9.3% weight loss.
Contrave is particularly useful for patients with comorbid depression or those trying to quit smoking, as bupropion addresses both conditions. It may also help patients whose weight gain is driven primarily by hedonic (reward-based) eating rather than homeostatic hunger.
Side Effects
Nausea is the most common side effect, affecting approximately 33% of patients in the first few weeks but typically resolving. Other side effects include headache, constipation, dizziness, insomnia, vomiting, and dry mouth.
Contrave carries a boxed warning regarding suicidal thoughts and behaviors associated with bupropion (shared with all antidepressants). It should not be used in patients with uncontrolled hypertension, seizure disorders, chronic opioid use, or eating disorders (bulimia or anorexia nervosa).
4. Liraglutide 3.0mg (Saxenda)
Mechanism of Action
Liraglutide is a GLP-1 receptor agonist with 97% amino acid homology to native human GLP-1. It activates GLP-1 receptors in multiple tissues, including the hypothalamus (reducing appetite), the pancreas (enhancing glucose-dependent insulin secretion), and the gastrointestinal tract (slowing gastric emptying). An albumin-binding fatty acid side chain extends its half-life to approximately 13 hours, enabling once-daily subcutaneous injection.
For a deeper exploration of GLP-1 mechanisms, see our GLP-1 weight loss mechanisms guide.
Clinical Efficacy
The SCALE Obesity and Prediabetes trial demonstrated 8.0% weight loss with liraglutide 3.0mg at 56 weeks versus 2.6% with placebo. Roughly 63% of patients achieved at least 5% weight loss. In participants with prediabetes, liraglutide reduced the risk of developing type 2 diabetes by 79% over 3 years (Pi-Sunyer et al., 2015).
The LEADER cardiovascular outcomes trial, conducted at the diabetes dose (1.8mg), showed a 13% reduction in major adverse cardiovascular events, establishing cardiovascular benefit for the GLP-1 class.
In August 2025, the FDA approved the first generic version of liraglutide (Saxenda) from Teva Pharmaceuticals, making it the first generic GLP-1 available for weight loss.
Practical Position
Liraglutide has been largely superseded by semaglutide and tirzepatide for weight management, given their superior efficacy and less frequent dosing. However, it remains relevant as a lower-cost option (especially the generic version), for patients who don't tolerate weekly injections, and for adolescents aged 12+ (Saxenda has pediatric approval).
5. Semaglutide 2.4mg (Wegovy)
Mechanism of Action
Semaglutide is an acylated GLP-1 analogue with 94% structural homology to native GLP-1. Its mechanism mirrors liraglutide's but with crucial pharmacokinetic advantages. A C18 fatty diacid chain enables tight albumin binding, extending the half-life to approximately 7 days and allowing once-weekly dosing. Semaglutide also demonstrates greater receptor binding affinity and more potent activation of GLP-1 receptor signaling compared to liraglutide.
Semaglutide acts on multiple sites to reduce body weight. In the hypothalamus, it activates POMC/CART neurons (promoting satiety) and inhibits NPY/AgRP neurons (reducing hunger). In the brainstem, it enhances satiation signals from the gut. It slows gastric emptying, reducing the rate at which nutrients reach the small intestine. And emerging evidence suggests it may alter food preferences, reducing cravings for high-fat and high-sugar foods. Read more in our GLP-1 hub.
Clinical Efficacy
The STEP clinical trial program established semaglutide as a best-in-class obesity treatment:
| Trial | Population | Weight Loss (semaglutide) | Weight Loss (placebo) | Duration |
|---|---|---|---|---|
| STEP 1 | Adults with obesity, no T2D | 14.9% | 2.4% | 68 weeks |
| STEP 2 | Adults with obesity + T2D | 9.6% | 3.4% | 68 weeks |
| STEP 3 | With intensive behavioral therapy | 16.0% | 5.7% | 68 weeks |
| STEP 4 | Withdrawal/continuation design | -7.9% (continued) vs +6.9% (switched to placebo) | - | 68 weeks |
| STEP 5 | Long-term (2-year) | 15.2% | 2.6% | 104 weeks |
| STEP 8 | Head-to-head vs liraglutide | 15.8% | 6.4% (liraglutide) | 68 weeks |
The SELECT cardiovascular outcomes trial was transformative. In 17,604 patients with established cardiovascular disease and BMI 27 or higher (without diabetes), semaglutide 2.4mg reduced the risk of MACE (cardiovascular death, non-fatal myocardial infarction, or non-fatal stroke) by 20% compared to placebo. This led to an expanded FDA indication for cardiovascular risk reduction in March 2024 (Lincoff et al., 2023).
In August 2025, the FDA granted accelerated approval to Wegovy for treating noncirrhotic MASH with moderate to advanced liver fibrosis, adding yet another indication.
Oral Semaglutide for Weight Management
On December 22, 2025, the FDA approved oral semaglutide 25mg daily (Wegovy pill) for chronic weight management and cardiovascular risk reduction. This marks the first oral GLP-1 receptor agonist approved for obesity, eliminating the need for weekly injections. The oral formulation uses an absorption enhancer (SNAC) to protect semaglutide from gastric degradation and promote transcellular absorption in the stomach.
Clinical data for oral semaglutide 25mg in obesity showed weight loss of approximately 13-15% at 68 weeks, somewhat less than the injectable 2.4mg formulation but still superior to most other oral medications.
Side Effects
GI side effects are class effects of GLP-1 agonists. With semaglutide 2.4mg, nausea affects approximately 44% of patients (vs 18% placebo), diarrhea 30% (vs 16%), vomiting 24% (vs 6%), and constipation 24% (vs 11%). These effects are typically mild-to-moderate, occur primarily during dose escalation, and diminish over time. Approximately 7% of patients discontinue due to GI side effects.
Other considerations include the risk of pancreatitis (rare), gallbladder disease (increased with rapid weight loss), and a potential signal for thyroid C-cell tumors seen in rodents (clinical relevance in humans uncertain). Semaglutide is contraindicated in patients with a personal or family history of medullary thyroid carcinoma or MEN2 syndrome.
6. Tirzepatide (Zepbound)
Mechanism of Action
Tirzepatide represents a fundamentally different approach. As the first dual GIP/GLP-1 receptor agonist, it activates two incretin receptors simultaneously. The GLP-1 component works through the same pathways as semaglutide. The GIP (glucose-dependent insulinotropic polypeptide) component adds distinct metabolic effects including enhanced insulin sensitivity in adipose tissue, improved lipid metabolism, and potentially direct effects on energy expenditure through brown adipose tissue activation.
The molecule is built on a modified GIP backbone with GLP-1 activity engineered into the structure. A C20 fatty diacid moiety enables albumin binding for once-weekly dosing with a half-life of approximately 5 days. For a complete exploration, see our tirzepatide complete guide.
Clinical Efficacy
Tirzepatide has produced the highest weight loss of any approved anti-obesity medication:
| Trial | Population | 5mg | 10mg | 15mg | Placebo | Duration |
|---|---|---|---|---|---|---|
| SURMOUNT-1 | Obesity, no T2D | 15.0% | 19.5% | 20.9% | 3.1% | 72 weeks |
| SURMOUNT-2 | Obesity + T2D | - | 12.8% | 14.7% | 3.2% | 72 weeks |
| SURMOUNT-3 | After lifestyle run-in | - | - | 18.4% (beyond run-in) | - | 72 weeks |
| SURMOUNT-4 | Withdrawal design | Maintained vs +14% regain | - | 88 weeks | ||
In SURMOUNT-1, 36% of participants on tirzepatide 15mg achieved 25% or greater weight loss, an outcome historically associated only with bariatric surgery (Jastreboff et al., 2022).
In December 2024, the FDA approved tirzepatide (Zepbound) for moderate-to-severe obstructive sleep apnea in adults with obesity. The SURMOUNT-OSA trial showed significant reductions in the apnea-hypopnea index alongside weight loss.
Head-to-Head vs Semaglutide
The SURPASS-2 trial in type 2 diabetes compared tirzepatide directly to semaglutide 1mg (the diabetes dose, not the 2.4mg obesity dose). Tirzepatide demonstrated superior A1C reduction and weight loss at all three doses. While no head-to-head trial has compared tirzepatide to semaglutide 2.4mg, indirect comparisons suggest tirzepatide 15mg produces roughly 5-6 percentage points more weight loss.
Side Effects
The side effect profile closely mirrors semaglutide. Nausea (up to 31%), diarrhea (23%), decreased appetite (20%), vomiting (12%), constipation (11%), and dyspepsia (9%) are the most common adverse events. Discontinuation rates due to adverse events range from 4-7% across dose groups, comparable to semaglutide.
Hypoglycemia risk is low when used without insulin or sulfonylureas. The same warnings regarding pancreatitis, gallbladder disease, and thyroid C-cell tumors apply to tirzepatide as to other GLP-1 agonists.
Clinical Pearl
When comparing approved agents, the magnitude of weight loss correlates with the breadth of receptor engagement. Orlistat (peripheral lipase inhibition) produces roughly 3.5%. Centrally-acting combinations (Qsymia, Contrave) achieve 5-10%. Single-incretin agonists (semaglutide) reach 15%. Dual-incretin agonists (tirzepatide) hit 21%. This pattern strongly suggests that the pipeline drugs targeting three or more pathways will push efficacy even higher.
Figure 4: Comparative efficacy of all FDA-approved anti-obesity medications by mean percent body weight loss
Pipeline Anti-Obesity Medications: The Next Wave
The pipeline for obesity pharmacotherapy is more active than at any point in pharmaceutical history. At least a dozen candidates are in Phase 2 or Phase 3 trials, with several likely to reach the market by 2027-2028. These agents push beyond single-receptor targeting, combining multiple hormonal pathways, offering oral delivery, or addressing the muscle loss that accompanies significant weight reduction.
Retatrutide (LY3437943) - Triple Agonist
The Triple Receptor Approach
Retatrutide is the first triple incretin receptor agonist, simultaneously targeting GLP-1, GIP, and glucagon receptors. The addition of glucagon receptor agonism represents the key innovation. While it might seem counterintuitive to activate a receptor associated with raising blood sugar, glucagon agonism contributes to weight loss through several mechanisms: it increases hepatic energy expenditure (thermogenesis), stimulates lipolysis in adipose tissue, and may enhance amino acid oxidation. These effects complement the appetite-suppressing and insulin-sensitizing actions of GLP-1 and GIP agonism.
The molecule is a single peptide chain with engineered activity at all three receptors, combined with a fatty acid modification for once-weekly subcutaneous dosing. For detailed coverage, see our retatrutide triple agonist guide.
Phase 2 Results
The Phase 2 trial in 338 adults with obesity (no diabetes) produced striking results. At 48 weeks, mean body weight loss was:
- Placebo: 2.1%
- 1mg: 8.7%
- 4mg (escalated): 17.1%
- 4mg (maintained): 18.2%
- 8mg: 22.1%
- 12mg: 24.2%
The 24.2% weight loss at the highest dose exceeded anything previously reported for any anti-obesity medication at any stage of development (Jastreboff et al., 2023). More than 25% of participants in the 12mg group lost at least 30% of their body weight.
Phase 3 Results (TRIUMPH Program)
In December 2025, Eli Lilly announced topline results from TRIUMPH-4, the first completed Phase 3 trial for retatrutide. The trial enrolled 445 adults with obesity or overweight plus knee osteoarthritis. Results were remarkable:
- 9mg dose: 26.4% mean body weight loss at 68 weeks
- 12mg dose: 28.7% mean body weight loss (average of 71.2 lbs)
- 58.6% of participants on 12mg achieved 25% or greater weight loss
- 39.4% achieved 30% or greater weight loss
Beyond weight, retatrutide reduced osteoarthritis pain by up to 75.8% on the WOMAC scale, lowered systolic blood pressure by 14 mmHg, and improved non-HDL cholesterol, triglycerides, and high-sensitivity C-reactive protein.
Safety Signals
GI side effects remained common but were generally manageable. However, a new safety signal emerged: dysesthesia (abnormal skin sensations like tingling, burning, or numbness) was reported in 8.8% of participants on 9mg and 20.9% on 12mg. This finding has not been seen with other GLP-1 or dual agonists and will require careful evaluation in ongoing trials.
Treatment discontinuation due to adverse events was 12.2% for 9mg and 18.2% for 12mg, specifically higher than semaglutide or tirzepatide discontinuation rates. Additional TRIUMPH trials are ongoing, with regulatory submission expected in 2026-2027.
CagriSema (Cagrilintide + Semaglutide)
Combining Amylin and GLP-1
Cagrilintide is a long-acting amylin analogue that, when combined with semaglutide, creates a complementary dual-mechanism approach. Amylin is a peptide hormone co-secreted with insulin from pancreatic beta cells. It slows gastric emptying, suppresses glucagon secretion, and promotes satiety through direct hypothalamic action. Native amylin has a very short half-life, but cagrilintide is engineered with an albumin-binding fatty diacid chain for once-weekly dosing.
The rationale for combining amylin and GLP-1 agonism is that they target overlapping but distinct neuronal populations in the brain's appetite centers. Together, they produce greater satiety than either alone.
REDEFINE Phase 3 Results
The REDEFINE 1 trial (3,417 participants without type 2 diabetes, 68 weeks) published in the New England Journal of Medicine in June 2025 showed:
- CagriSema: 20.4% body weight loss
- Cagrilintide alone: 11.5%
- Semaglutide alone: 14.9%
- Placebo: 3.0%
Approximately 60% of CagriSema participants achieved at least 20% weight loss, and 23% lost 30% or more (Novo Nordisk, REDEFINE 1, 2025).
The REDEFINE 2 trial (1,206 participants with type 2 diabetes) showed CagriSema achieved 13.7% weight loss versus 3.4% with placebo. 73.5% of CagriSema participants achieved HbA1c of 6.5% or lower compared to 15.9% with placebo (Novo Nordisk, REDEFINE 2, 2025).
Competitive Position
CagriSema's 20.4% weight loss is essentially comparable to tirzepatide's 20.9% (within statistical noise), which was somewhat disappointing for Novo Nordisk investors who had hoped for a clear advantage. The initial announcement noted that CagriSema "fell short of the 25% weight loss target." However, the combination still represents a meaningful advance over semaglutide alone. GI side effects were common (79.6% of participants) but mostly mild to moderate and transient.
Orforglipron - Oral Small-Molecule GLP-1
Why an Oral GLP-1 Matters
Orforglipron is a non-peptide, small-molecule GLP-1 receptor agonist taken as a daily oral pill. Unlike oral semaglutide (which is still a peptide requiring the SNAC absorption enhancer and fasting conditions), orforglipron is a true small molecule that can be taken without food restrictions and manufactured using conventional pharmaceutical processes rather than complex biologic production.
This distinction has enormous implications for cost and accessibility. Small molecules are far cheaper to manufacture than peptides, potentially bringing GLP-1 therapy within reach of the hundreds of millions of people worldwide who currently can't afford injectable treatments.
Phase 3 ATTAIN Results
The ATTAIN-1 trial (adults with obesity, no diabetes, 72 weeks) showed:
- Orforglipron 6mg: 7.5% weight loss
- Orforglipron 12mg: 8.4% weight loss
- Orforglipron 36mg: 11.2% weight loss
- Placebo: 2.1%
While these results are clinically meaningful, they fall short of injectable semaglutide (14.9%) and tirzepatide (20.9%). At the highest dose, 59.6% of participants lost at least 10% of body weight and 39.6% lost at least 15%.
ATTAIN-2 (adults with obesity + type 2 diabetes) showed the 36mg dose produced 10.5% weight loss with 1.8% HbA1c reduction. The ATTAIN-MAINTAIN trial (December 2025) demonstrated that patients could successfully transition from injectable therapies (Wegovy or Zepbound) to oral orforglipron while maintaining their weight loss.
Regulatory Path
Eli Lilly has received National Priority Review Voucher status for orforglipron and is advancing global regulatory submissions, with potential approval expected in 2026. If approved, it would become the first non-peptide oral GLP-1 for weight management.
Side Effects
The side effect profile is consistent with the GLP-1 class. GI events were the most common adverse effects, with treatment discontinuation rates of 5.3% (6mg), 7.9% (12mg), and 10.3% (36mg) vs 2.7% with placebo. These rates are broadly comparable to or slightly better than injectable GLP-1s.
Survodutide (BI 456906) - GLP-1/Glucagon Dual Agonist
Mechanism
Survodutide is a dual GLP-1/glucagon receptor agonist developed by Boehringer Ingelheim and Zealand Pharma. Unlike tirzepatide (GIP/GLP-1), survodutide pairs GLP-1 agonism with glucagon agonism, using the energy expenditure-boosting and lipolytic properties of glucagon receptor activation. The glucagon component particularly targets hepatic fat metabolism, making survodutide especially promising for patients with MASLD/MASH.
Phase 2 Data
A Phase 2 trial across 43 centers involving 386 participants (BMI 27+ without diabetes) showed the 4.8mg dose produced 14.9% body weight loss at 46 weeks compared to 2.8% with placebo. Multiple Phase 3 SYNCHRONIZE trials are now underway evaluating survodutide for obesity and MASH.
In the Phase 2 MASH trial, survodutide achieved histological resolution of steatohepatitis without worsening fibrosis in up to 83% of participants at the highest dose, a figure that significantly exceeds the results seen with other anti-obesity agents in MASH studies.
Differentiation
Survodutide's key differentiator is its hepatic focus. The glucagon receptor is highly expressed in the liver, and glucagon agonism directly stimulates hepatic fatty acid oxidation, potentially making it the treatment of choice for patients with obesity complicated by fatty liver disease.
Amycretin - Oral GLP-1/Amylin Dual Agonist
A Standout Oral Candidate
Amycretin, developed by Novo Nordisk, is an oral dual GLP-1 and amylin receptor agonist taken once daily. Early-phase data showed approximately 22% weight loss in just 36 weeks, an extraordinary result for an oral medication. This would put amycretin's efficacy on par with the best injectable agents currently available.
Amycretin is advancing into Phase 3 studies. If Phase 3 results confirm the early data, it could become the most effective oral obesity treatment in development, potentially disrupting the market for injectable therapies.
Pemvidutide (ALT-801) - GLP-1/Glucagon Agonist
Phase 2 Results
Pemvidutide is a once-weekly subcutaneous GLP-1/glucagon receptor dual agonist developed by Altimmune. The Phase 2 MOMENTUM-1 trial reported mean weight loss up to 15.6% (placebo-adjusted 13.4%) at 48 weeks with the 2.4mg dose.
Like survodutide, pemvidutide shows particular promise for liver fat reduction. In patients with metabolic dysfunction-associated steatotic liver disease (MASLD), 24 weeks of pemvidutide resulted in a relative reduction in liver fat content of up to 76% with the 2.4mg dose versus 14% with placebo. This makes it a potentially attractive option for the large population of obese patients with concomitant fatty liver disease.
Bimagrumab - Muscle-Sparing Antibody
Addressing the Muscle Loss Problem
Bimagrumab represents an entirely different approach. It's a monoclonal antibody targeting activin type II receptors (ActRII), which are part of the myostatin/activin signaling pathway that negatively regulates muscle growth. By blocking these receptors, bimagrumab promotes muscle preservation and growth, directly addressing one of the most significant concerns with pharmacological weight loss: loss of lean body mass.
Typically, 20-40% of weight lost with GLP-1 agonists comes from lean mass rather than fat. This can reduce metabolic rate, impair physical function, and contribute to sarcopenic obesity, a condition where low muscle mass coexists with excess fat.
BELIEVE Phase 2b Trial
The BELIEVE trial (507 participants) evaluated bimagrumab alone and in combination with semaglutide. Results presented at ADA 2025 were impressive:
| Treatment Group | Total Weight Loss | % from Fat Mass | Visceral Fat Reduction | Lean Mass Change |
|---|---|---|---|---|
| Bimagrumab alone | 10.8% | 100% | - | +2.5% |
| Semaglutide alone | 15.7% | 71.8% | 36% | Decreased |
| Bimagrumab + Semaglutide | 22.1% | 92.8% | 58% | Preserved |
| Placebo | ~2% | - | - | No change |
The combination is especially notable for its body composition effects. With bimagrumab alone, 100% of weight loss came from fat with an actual increase in lean mass. When combined with semaglutide, 92.8% of weight loss was fat mass compared to only 71.8% with semaglutide alone. Visceral fat reduction nearly doubled (58% vs 36%).
Eli Lilly acquired bimagrumab from Versanis in December 2023 for up to $1.93 billion, signaling its strategic importance. Phase 3 trials are expected to begin.
Pipeline Summary Table
| Drug | Mechanism | Route | Phase | Best Weight Loss | Developer |
|---|---|---|---|---|---|
| Retatrutide | GLP-1/GIP/Glucagon | SC weekly | Phase 3 | 28.7% | Eli Lilly |
| CagriSema | Amylin + GLP-1 | SC weekly | Phase 3 | 20.4% | Novo Nordisk |
| Orforglipron | GLP-1 (small molecule) | Oral daily | Phase 3 | 11.2% | Eli Lilly |
| Survodutide | GLP-1/Glucagon | SC weekly | Phase 3 | 14.9% | Boehringer/Zealand |
| Amycretin | GLP-1/Amylin | Oral daily | Phase 3 (entering) | ~22% | Novo Nordisk |
| Pemvidutide | GLP-1/Glucagon | SC weekly | Phase 2 | 15.6% | Altimmune |
| Bimagrumab | Anti-ActRII antibody | IV infusion | Phase 2b | 22.1% (combo) | Eli Lilly |

Figure 5: Anti-obesity drug pipeline landscape showing development stages and anticipated regulatory timelines
Mechanism of Action Comparison: How Each Approach Works
Understanding how these drugs work at the molecular level helps explain their different efficacy profiles, side effect patterns, and ideal patient populations. The evolution from single-target to multi-target approaches has been the primary driver of improved outcomes.
GLP-1 Receptor Agonism: The Foundation
Glucagon-like peptide-1 (GLP-1) is an incretin hormone secreted by L-cells in the small intestine in response to food intake. Under normal physiology, native GLP-1 has a half-life of just 2-3 minutes before degradation by dipeptidyl peptidase-4 (DPP-4). All GLP-1 receptor agonists are engineered to resist DPP-4 degradation, extending their duration of action from minutes to hours or days.
GLP-1 receptor activation produces weight loss through multiple convergent pathways:
- Hypothalamic appetite suppression: GLP-1 receptors in the arcuate nucleus and paraventricular nucleus of the hypothalamus activate anorexigenic POMC/CART neurons and inhibit orexigenic NPY/AgRP neurons, reducing hunger drive
- Brainstem satiation signaling: GLP-1 receptors in the nucleus tractus solitarius (NTS) and area postrema enhance meal-induced satiation, making patients feel full sooner
- Delayed gastric emptying: Slowing the rate at which food leaves the stomach prolongs satiety signals and reduces postprandial glucose spikes
- Reward pathway modulation: Emerging neuroimaging evidence suggests GLP-1 agonists reduce activation in mesolimbic dopamine pathways in response to food cues, particularly for high-calorie, palatable foods
- Pancreatic effects: Glucose-dependent insulin secretion (reducing hyperglycemia without causing hypoglycemia) and glucagon suppression (reducing hepatic glucose output)
The mechanisms underlying GLP-1-mediated weight loss are covered in depth in our dedicated guide.
GIP Receptor Agonism: The Dual Incretin Advantage
Glucose-dependent insulinotropic polypeptide (GIP) is the other major incretin hormone, secreted by K-cells in the duodenum and jejunum. For years, GIP was thought to promote weight gain rather than loss, as it enhances lipogenesis in adipose tissue. The discovery that GIP receptor agonism could be combined with GLP-1 agonism to enhance weight loss was therefore surprising.
The proposed mechanisms by which GIP contributes to weight loss in the context of dual agonism include:
- Central appetite regulation: GIP receptors are expressed in the hypothalamus and may enhance the satiety signal when co-activated with GLP-1 receptors
- Improved insulin sensitivity: GIP enhances insulin sensitivity in adipose tissue, potentially improving the metabolic health of remaining fat tissue
- Brown adipose tissue activation: GIP receptor signaling may promote thermogenesis through brown and beige adipose tissue activation
- Lipid metabolism: GIP influences triglyceride clearance and lipoprotein lipase activity, potentially redirecting lipid metabolism away from storage and toward utilization
The combined effect between GIP and GLP-1 agonism likely explains why tirzepatide produces substantially more weight loss than semaglutide, despite semaglutide being a more potent GLP-1 receptor agonist on a molar basis.
Glucagon Receptor Agonism: The Energy Expenditure Component
Glucagon is often viewed solely as the counter-regulatory hormone to insulin, raising blood glucose through glycogenolysis and gluconeogenesis. But glucagon also has significant effects on energy expenditure that make it valuable in the context of obesity treatment:
- Hepatic thermogenesis: Glucagon increases energy expenditure in the liver through futile cycling and enhanced fatty acid oxidation, potentially raising resting metabolic rate
- Lipolysis: Glucagon stimulates breakdown of triglycerides in adipose tissue, mobilizing fatty acids for oxidation
- Hepatic lipid metabolism: Glucagon reduces hepatic lipogenesis and promotes fatty acid oxidation in the liver, directly addressing hepatic steatosis (fatty liver)
- Amino acid metabolism: Glucagon promotes hepatic amino acid catabolism, which may contribute to overall energy expenditure
The challenge of glucagon agonism is its hyperglycemic effect. In retatrutide and survodutide, the concurrent GLP-1 agonism counterbalances glucagon's glucose-raising action, allowing the energy expenditure benefits without worsening glycemic control. This "built-in safeguard" is why triple and dual agonists including glucagon activity can exist as viable therapeutic agents.
Amylin Receptor Agonism: Complementary Satiety
Amylin is a 37-amino-acid peptide co-secreted with insulin from pancreatic beta cells. It produces satiety through mechanisms partially overlapping with, but distinct from, GLP-1:
- Area postrema signaling: Amylin activates neurons in the area postrema (a circumventricular organ with a leaky blood-brain barrier) to promote meal termination
- Gastric emptying: Amylin slows gastric emptying through vagal afferent pathways, complementing GLP-1's similar effect
- Glucagon suppression: Amylin inhibits postprandial glucagon secretion, reducing hepatic glucose output
- Hedonic suppression: Amylin may reduce the rewarding properties of food through effects on mesolimbic pathways
Pramlintide (Symlin) is an existing synthetic amylin analogue approved for diabetes, but its short half-life and modest weight loss limited its utility for obesity. Cagrilintide's long-acting formulation (used in CagriSema) overcomes this limitation.
Myostatin/Activin Pathway Blockade: Muscle Preservation
The activin/myostatin signaling pathway is a key negative regulator of muscle growth. Myostatin (also called GDF-8) signals through activin type II receptors (ActRIIA and ActRIIB) to suppress muscle protein synthesis and promote muscle atrophy. Bimagrumab blocks these receptors, effectively removing the "brake" on muscle growth.
This mechanism is particularly valuable in the obesity context because:
- It directly counteracts the lean mass loss that accompanies significant weight reduction
- Increased muscle mass raises resting metabolic rate, potentially reducing weight regain
- Preserved functional capacity improves quality of life and exercise tolerance during weight loss
- The combination of fat loss and muscle preservation produces a more favorable body composition outcome than weight loss alone
NNMT Inhibition: The 5-Amino-1MQ Approach
5-Amino-1MQ targets nicotinamide N-methyltransferase (NNMT), an enzyme that is overexpressed in adipose tissue of obese individuals. NNMT metabolizes nicotinamide (vitamin B3) by converting it to 1-methylnicotinamide, depleting the substrate pool needed for NAD+ biosynthesis. By inhibiting NNMT, 5-amino-1MQ:
- Increases intracellular NAD+ levels, enhancing mitochondrial function and energy metabolism
- Reduces lipogenesis (new fat formation) in adipocytes
- Increases GLUT4 transporter expression, improving glucose uptake
- Promotes energy expenditure without affecting food intake (in preclinical models)
In animal studies, 5-amino-1MQ-treated mice showed 35% body mass reduction, 30% decrease in adipocyte size, and improved cholesterol profiles without changes in food intake (Neelakantan et al., 2018). However, human clinical data are still lacking.
HGH Fragment (AOD-9604): Targeted Lipolysis
AOD-9604 is a modified fragment of human growth hormone (amino acids 177-191) that selectively activates lipolysis without the growth-promoting or diabetogenic effects of full-length HGH. It stimulates fat breakdown while simultaneously inhibiting lipogenesis, creating a dual anti-fat mechanism.
AOD-9604's activity involves upregulation of beta-3 adrenergic receptors in adipose tissue, though the exact signaling cascade is not fully elucidated. Despite showing promise in animal models, the peptide failed to achieve statistical significance in its largest Phase IIb human clinical trial, and clinical development was discontinued in 2007.
Triple Monoamine Reuptake Inhibition: Tesofensine
Tesofensine operates through an entirely different mechanism. It inhibits the reuptake of three monoamine neurotransmitters: serotonin, norepinephrine, and dopamine. By increasing the availability of all three in the hypothalamus and reward circuits, tesofensine suppresses appetite through both homeostatic (hypothalamic) and hedonic (reward) pathways.
Recent 2024 research published in PLOS One demonstrated that tesofensine silences a subset of GABAergic neurons in the lateral hypothalamus, reducing their ability to promote feeding behavior (Bhatt et al., 2024). Additionally, tesofensine increases thermogenesis, contributing to negative energy balance through both reduced intake and increased expenditure.
Phase 2 trials showed weight loss of 4.5%, 9.2%, and 10.6% at the 0.25mg, 0.5mg, and 1.0mg doses respectively, compared to 2.2% with placebo. At the highest dose, the average weight loss was 12.8 kg over 24 weeks (Astrup et al., 2008). For a full review, see our tesofensine weight loss guide.
Figure 6: Molecular mechanisms of action for major anti-obesity drug classes showing receptor targets and downstream effects
Non-GLP-1 Approaches to Obesity Treatment
While GLP-1 receptor agonists dominate current obesity pharmacotherapy, several non-incretin-based compounds target entirely different metabolic pathways. These agents are of particular interest for patients who don't respond to or can't tolerate GLP-1-based therapies, or as complementary treatments in combination strategies.
Tesofensine: Triple Monoamine Reuptake Inhibitor
Tesofensine was originally developed as a treatment for Alzheimer's and Parkinson's disease. When those trials failed to show neurological benefit, researchers noticed significant weight loss as a side effect, redirecting development toward obesity. This accidental discovery mirrors the trajectory of several other weight loss medications, including bupropion and topiramate.
How It Differs from GLP-1 Agonists
Where GLP-1 agonists work primarily through gut-brain signaling and satiety enhancement, tesofensine targets the central reward and motivation circuits directly. It increases dopamine availability in the nucleus accumbens and prefrontal cortex, potentially reducing the hedonic drive to overeat. The serotonin component promotes satiety, while norepinephrine enhances sympathetic tone and energy expenditure.
This makes tesofensine potentially complementary to GLP-1 agonists rather than redundant. A GLP-1 agonist addresses homeostatic hunger (eating because you're hungry), while tesofensine may better address hedonic eating (eating for pleasure or emotional reasons). Though no combination trials have been conducted, the theoretical basis for pairing these mechanisms is sound.
Current Status
Tesofensine's development has been complicated by cardiovascular concerns. It increases heart rate by approximately 7-8 beats per minute and raises blood pressure in some patients, effects driven by its norepinephrine reuptake inhibition. Saniona (formerly NeuroSearch) continues development under specific regulatory conditions. The compound is not FDA-approved but is available through research and compounding channels.
5-Amino-1MQ: NNMT Inhibitor
5-Amino-1MQ represents one of the most mechanistically distinct approaches in obesity research. By targeting NNMT, it addresses a metabolic root cause of obesity rather than simply suppressing appetite.
The NNMT-Obesity Connection
NNMT expression is significantly elevated in white adipose tissue of obese individuals. This enzyme consumes nicotinamide and S-adenosylmethionine (SAM), depleting two key metabolites needed for cellular energy production and epigenetic regulation. NNMT overactivity creates a metabolic state favoring fat storage over fat oxidation.
By inhibiting NNMT, 5-amino-1MQ restores normal NAD+ and SAM levels, effectively reprogramming adipocyte metabolism from a fat-storing to a fat-burning phenotype. Preclinical data show this occurs without changes in food intake - the weight loss is purely metabolic rather than appetite-driven.
Preclinical Evidence
In diet-induced obese mice, 5-amino-1MQ treatment produced:
- 35% reduction in body mass
- 30% decrease in adipocyte size
- 40% decrease in adipocyte volume
- Normalized cholesterol levels comparable to lean controls
- No changes in food intake or observable adverse effects
The compound also showed anti-inflammatory effects in adipose tissue and improved insulin sensitivity in preclinical models. However, it's critical to note that no human clinical trials have been completed, and the translation from mouse to human metabolic effects is uncertain.
AOD-9604: Growth Hormone Fragment
AOD-9604 takes a targeted approach to lipolysis. By using only the fat-mobilizing portion of human growth hormone (the C-terminal fragment), it avoids the growth-promoting, diabetogenic, and potentially tumorigenic effects of full-length HGH.
Preclinical Promise vs Clinical Reality
In obese Zucker rats, AOD-9604 produced significant fat mass reduction without affecting lean body mass or food intake. The peptide was particularly effective at reducing visceral fat, the metabolically active depot most strongly associated with cardiovascular disease and insulin resistance.
Despite these encouraging animal data, AOD-9604's largest human clinical trial (Phase IIb) failed to achieve its primary endpoint. The disconnect between preclinical and clinical results may relate to species differences in growth hormone receptor signaling, inadequate dosing, or the limitations of the specific patient population studied.
AOD-9604 currently lacks FDA approval for any indication. However, it has received Generally Recognized as Safe (GRAS) status from the FDA for use as a food ingredient, and ongoing research continues to explore its effects on joint health and cartilage repair in addition to metabolic applications.
MOTS-c: Mitochondrial-Derived Peptide
MOTS-c is a mitochondrial-derived peptide encoded within the mitochondrial genome (12S rRNA gene). It functions as a retrograde signaling molecule, with the mitochondria communicating metabolic status to the nuclear genome. MOTS-c activates AMPK (AMP-activated protein kinase), the master metabolic sensor, and promotes glucose uptake, fatty acid oxidation, and mitochondrial biogenesis.
In preclinical studies, MOTS-c prevented diet-induced obesity and improved insulin sensitivity. It also enhanced exercise capacity and has shown potential as an exercise mimetic. However, like 5-amino-1MQ, human clinical data are limited, and MOTS-c is currently available only as a research compound.
Research Status Disclaimer
Several compounds discussed in this section (tesofensine, 5-amino-1MQ, AOD-9604, MOTS-c) are not FDA-approved for obesity treatment. Their inclusion here reflects ongoing research interest and is for informational purposes. Always consult with a qualified healthcare provider before considering any weight loss intervention. Research compounds carry additional risks related to purity, dosing uncertainty, and lack of long-term safety data.
Efficacy Comparison: Head-to-Head and Cross-Trial Analysis
Comparing anti-obesity medications requires caution. Direct head-to-head trials are rare, and cross-trial comparisons are complicated by differences in study populations, baseline characteristics, lifestyle interventions, and outcome definitions. Nonetheless, the available data paint a clear picture of relative efficacy across drug classes.
Approved Agents: Direct Comparison
| Medication | Mean % Weight Loss | % Achieving 5%+ Loss | % Achieving 10%+ Loss | % Achieving 15%+ Loss | Trial Duration |
|---|---|---|---|---|---|
| Orlistat 120mg | 3.5% | ~35% | ~15% | <5% | 52-208 weeks |
| Phentermine-Topiramate 15/92 | 9.8% | 70% | 48% | ~25% | 56 weeks |
| Naltrexone-Bupropion 32/360 | 5.4% | ~50% | ~25% | ~10% | 56 weeks |
| Liraglutide 3.0mg | 8.0% | 63% | 33% | 14% | 56 weeks |
| Semaglutide 2.4mg | 14.9% | 86% | 69% | 50% | 68 weeks |
| Tirzepatide 15mg | 20.9% | 91% | 79% | 56% | 72 weeks |
Only two direct comparisons among approved agents exist. STEP 8 compared semaglutide 2.4mg to liraglutide 3.0mg, with semaglutide producing 15.8% vs 6.4% weight loss. SURPASS-2 compared tirzepatide to semaglutide 1.0mg (the diabetes dose), with all three tirzepatide doses outperforming semaglutide (Frias et al., 2021).
Pipeline Agents vs Approved Standards
Cross-trial comparisons of pipeline agents to approved medications, keeping in mind the limitations of indirect comparisons:
| Agent | Best Weight Loss | Trial Phase | vs Semaglutide 2.4mg | vs Tirzepatide 15mg |
|---|---|---|---|---|
| Retatrutide 12mg | 28.7% | Phase 3 | +13.8 pp | +7.8 pp |
| CagriSema | 20.4% | Phase 3 | +5.5 pp | -0.5 pp |
| Bimagrumab + Semaglutide | 22.1% | Phase 2b | +7.2 pp | +1.2 pp |
| Amycretin (oral) | ~22% | Phase 2 | +7.1 pp | +1.1 pp |
| Survodutide 4.8mg | 14.9% | Phase 2 | 0.0 pp | -6.0 pp |
| Pemvidutide 2.4mg | 15.6% | Phase 2 | +0.7 pp | -5.3 pp |
| Orforglipron 36mg | 11.2% | Phase 3 | -3.7 pp | -9.7 pp |
pp = percentage points. Cross-trial comparisons are approximate and should be interpreted with caution.
Key Comparative Insights
Retatrutide stands apart. Even accounting for cross-trial variability, a 28.7% mean weight loss at 68 weeks significantly exceeds anything else in the pipeline. The triple agonist approach combining energy expenditure-boosting glucagon activity with GLP-1 and GIP effects appears genuinely complementary.
CagriSema lands at parity with tirzepatide. The 20.4% weight loss achieved by adding amylin to semaglutide is meaningful but doesn't clearly leap ahead of tirzepatide 15mg. This creates a competitive challenge for Novo Nordisk, as the REDEFINE results essentially show that their combination matches Lilly's already-approved monotherapy.
Orforglipron trades efficacy for convenience. At 11.2%, oral orforglipron delivers meaningfully less weight loss than injectable semaglutide (14.9%) or tirzepatide (20.9%). However, the oral route and potentially lower cost could make it the preferred first-line agent for patients who decline or can't access injections.
Bimagrumab redefines quality of weight loss. While its total weight loss (22.1% in combination) is impressive, the real story is body composition. Having 92.8% of weight loss come from fat rather than the typical 70% fundamentally changes the health impact of treatment.
Weight Loss vs Body Composition: An Evolving Paradigm
The field is increasingly recognizing that total body weight loss is an incomplete metric. Two patients who each lose 15% of their body weight may have very different outcomes if one loses primarily fat while the other loses substantial lean mass. Metrics like fat-free mass index, visceral adipose tissue volume, and body composition percentage may prove more clinically relevant than scale weight alone.
This shift favors combination approaches like bimagrumab + semaglutide, which optimize body composition rather than simply minimizing the number on the scale. It also highlights the potential value of resistance exercise and adequate protein intake alongside pharmacotherapy, strategies that preserve lean mass regardless of which medication is used.
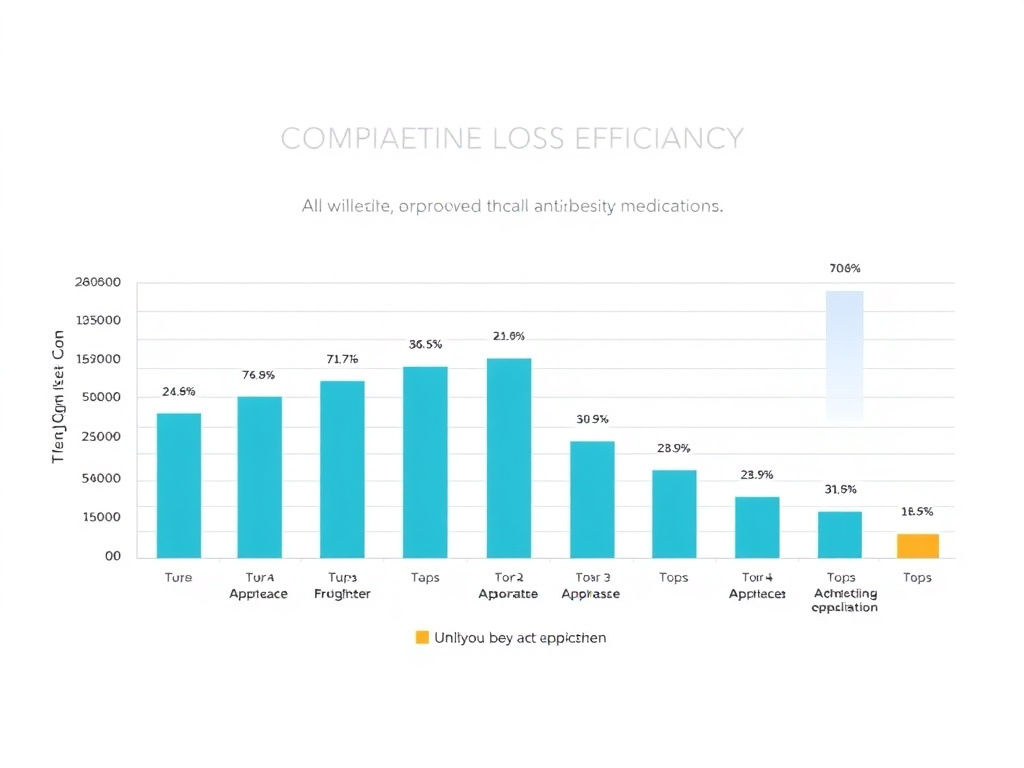
Figure 7: Comparative efficacy of approved and pipeline anti-obesity agents by mean percent body weight loss
Choosing the Right Anti-Obesity Medication
Selecting an anti-obesity medication involves balancing efficacy, tolerability, route of administration, cost, comorbidities, and patient preference. There is no single "best" drug for every patient. Here's a framework for decision-making.
Patient Factors to Consider
Severity of Obesity
For patients with BMI 27-30 and one comorbidity, any approved agent may be reasonable, with the choice driven by cost, tolerability, and preference. For patients with BMI 35+ or significant comorbidities, the higher-efficacy agents (semaglutide, tirzepatide) are generally preferred, as greater weight loss translates to more meaningful risk reduction.
Patients with BMI 40+ or BMI 35+ with severe comorbidities should also be evaluated for bariatric surgery, which remains the most effective long-term intervention for severe obesity. Pharmacotherapy and surgery aren't mutually exclusive; some patients may benefit from pre-surgical medication to reduce operative risk, or post-surgical medication to prevent weight regain.
Type 2 Diabetes
For patients with comorbid type 2 diabetes, tirzepatide and semaglutide offer dual benefits of weight loss and glycemic control. Tirzepatide has shown superior HbA1c reduction in head-to-head trials (SURPASS program). Liraglutide and oral semaglutide (at diabetes doses) are also options. Qsymia and Contrave have modest metabolic benefits but are not first-line for patients with diabetes.
Cardiovascular Disease
Semaglutide 2.4mg (Wegovy) is the only anti-obesity medication with demonstrated cardiovascular outcomes benefit (SELECT trial, 20% MACE reduction). For patients with established CVD or high CV risk, semaglutide should be strongly considered. Liraglutide (at diabetes dose) showed cardiovascular benefit in LEADER, but this hasn't been replicated at the obesity dose. Tirzepatide cardiovascular outcomes trials are ongoing.
Avoid phentermine-based medications in patients with uncontrolled hypertension or significant cardiovascular disease.
MASH/Fatty Liver Disease
Semaglutide received an accelerated approval for MASH in August 2025. Survodutide and pemvidutide (both GLP-1/glucagon agonists) show particular promise for liver fat reduction due to the glucagon component's hepatic effects. Retatrutide (which also includes glucagon agonism) would be expected to provide similar hepatic benefits.
Obstructive Sleep Apnea
Tirzepatide (Zepbound) is the only anti-obesity medication specifically approved for obesity-related OSA (December 2024). Weight loss from any effective anti-obesity medication would be expected to improve OSA, but tirzepatide has the strongest evidence in this population.
Injection Aversion
For patients who refuse or can't manage injections, oral options include:
- Oral semaglutide 25mg (Wegovy pill) - approved December 2025
- Orforglipron (pending approval, expected 2026)
- Phentermine-topiramate ER (Qsymia)
- Naltrexone-bupropion ER (Contrave)
- Orlistat (Xenical/Alli)
Muscle Preservation Concerns
Patients at risk for sarcopenia (older adults, those with limited mobility, patients with large amounts of weight to lose) should consider strategies to preserve lean mass. Bimagrumab (if approved) would be the first pharmacological option. In the interim, resistance training and adequate protein intake (1.2-1.6g/kg ideal body weight per day) are recommended alongside any anti-obesity medication.
Treatment Algorithm
Suggested Treatment Approach
- Foundation: All patients should receive counseling on diet, physical activity, sleep, and behavioral strategies. These are not replacements for medication but essential complements.
- First-line (if injectable acceptable): Semaglutide 2.4mg or tirzepatide, based on individual comorbidity profile and insurance coverage. Tirzepatide preferred for maximum weight loss; semaglutide preferred for cardiovascular risk reduction.
- First-line (if oral preferred): Oral semaglutide 25mg or Qsymia. Orforglipron when available.
- Second-line/Add-on: Consider combination approaches (when available), switch between agents, or add non-GLP-1 adjunct.
- Third-line: Consider bariatric surgery referral for patients with BMI 40+ or BMI 35+ with comorbidities who have not achieved adequate response to pharmacotherapy.
- Long-term: Obesity is a chronic disease. Medication should generally be continued long-term, as weight regain after discontinuation is the rule rather than the exception. STEP 4 and SURMOUNT-4 both demonstrated significant weight regain when active treatment was replaced with placebo.
Visit our dosing calculator for help determining appropriate starting doses, and our getting started guide for practical information on beginning treatment.
Cost, Insurance, and Access Considerations
Cost remains the single largest barrier to obesity pharmacotherapy. The most effective medications are also the most expensive, creating a two-tiered system where access often depends more on insurance coverage and socioeconomic status than on clinical need.
Current Pricing Landscape
| Medication | List Price (Monthly) | With Savings Programs | Generic Available |
|---|---|---|---|
| Orlistat (Xenical) | $200-$400 | Generic: $30-$80 | Yes |
| Orlistat (Alli OTC) | $40-$60 | N/A | OTC |
| Phentermine-Topiramate (Qsymia) | $200-$300 | Generic available (2025) | Yes |
| Naltrexone-Bupropion (Contrave) | $300-$500 | $99+ with coupons | Generic expected |
| Liraglutide (Saxenda) | $1,200-$1,400 | Generic: $400-$600 (Teva) | Yes (Aug 2025) |
| Semaglutide (Wegovy) | $1,300-$1,500 | $0-$25 with some insurers | No |
| Tirzepatide (Zepbound) | $1,000-$1,100 | $0-$25 with some insurers | No |
At list prices, a year of semaglutide or tirzepatide costs $12,000-$18,000. Even with manufacturer savings cards (which typically cover a portion of the cost for insured patients), out-of-pocket expenses can be substantial.
Insurance Coverage Trends
Insurance coverage for obesity medications has been gradually expanding but remains inconsistent:
- Medicare: Medicare Part D explicitly excluded coverage for anti-obesity medications until the Treat and Reduce Obesity Act gained support. As of early 2026, Medicare covers semaglutide and tirzepatide for patients with established cardiovascular disease (under the CV risk reduction indication) but still doesn't cover weight management indications alone for most beneficiaries.
- Medicaid: Coverage varies by state, with some states covering GLP-1 agonists for obesity and others excluding weight loss medications entirely.
- Commercial insurance: Coverage has been improving, particularly for semaglutide and tirzepatide. Many plans require prior authorization, documentation of BMI and comorbidities, and proof of failed lifestyle interventions. Step therapy requirements (trying cheaper medications first) are common.
- Self-insured employers: Some large employers have added GLP-1 coverage, recognizing that the cost of medication may be offset by reduced healthcare utilization, disability claims, and productivity losses.
Compounding and Alternative Access
During periods of FDA-declared shortage (which affected both semaglutide and tirzepatide through 2024-2025), compounding pharmacies were permitted to produce copies of these medications. Compounded versions typically cost $200-$500 per month, significantly less than brand-name products.
However, the FDA has been tightening regulations around compounded GLP-1 agonists. As brand-name supply stabilized, the FDA moved to restrict compounding of semaglutide and tirzepatide. This created significant controversy, with patients who had been using affordable compounded versions facing sudden loss of access.
The regulatory landscape around compounded peptides continues to evolve. Patients considering compounded medications should verify that their pharmacy holds appropriate state and federal licenses and follows current Good Manufacturing Practice (cGMP) standards.
Cost-Effectiveness Analysis
The Institute for Clinical and Economic Review (ICER) has published cost-effectiveness analyses for several anti-obesity medications. Their findings suggest that at list prices, GLP-1 agonists generally exceed conventional cost-effectiveness thresholds ($100,000-$150,000 per quality-adjusted life year). However, when discounted prices are considered (reflecting negotiated insurance rates), cost-effectiveness improves substantially.
Critically, cost-effectiveness calculations for obesity medications should account for downstream savings: reduced incidence of type 2 diabetes, fewer cardiovascular events, less joint replacement surgery, fewer cancer diagnoses, and reduced healthcare utilization for obesity-related comorbidities. When these longer-term savings are included, the economic case for pharmacotherapy becomes considerably stronger.
Future Cost Dynamics
Several factors could reduce costs over the coming years:
- Generic entry: Liraglutide's generic (Teva, August 2025) is the first. Semaglutide and tirzepatide patents won't expire for years, but biosimilar development is underway.
- Small molecule oral agents: Orforglipron and amycretin, as non-biologic compounds, should be cheaper to manufacture than injectable peptides. If approved, they could significantly reduce the cost of effective obesity treatment.
- Market competition: As more agents enter the market, competitive pressure should drive down pricing. Eli Lilly priced Zepbound below Wegovy as a competitive strategy, and further entrants should amplify this effect.
- Legislative pressure: Growing public awareness and political will regarding drug pricing may result in policies that improve access to obesity medications.

Figure 8: Monthly cost comparison of anti-obesity medications at list price versus typical out-of-pocket with insurance
Safety Comparison Across Drug Classes
Every anti-obesity medication involves trade-offs between efficacy and potential adverse effects. Understanding the safety profiles of each class helps guide prescribing decisions and allows for proactive side effect management.
Comparative Safety Overview
| Side Effect | Orlistat | Qsymia | Contrave | GLP-1s | Tirzepatide |
|---|---|---|---|---|---|
| Nausea | Low | Moderate | High (33%) | High (44%) | High (31%) |
| Vomiting | Low | Low | Moderate | High (24%) | Moderate (12%) |
| Diarrhea | Low | Low | Low | High (30%) | Moderate (23%) |
| Constipation | Low | Moderate | Moderate | Moderate (24%) | Moderate (11%) |
| Oily/Fatty Stools | Very High | None | None | None | None |
| Heart Rate Increase | None | Mild | None | Mild (1-4 bpm) | Mild (1-4 bpm) |
| CNS Effects | None | Moderate | Moderate | Low | Low |
| Pancreatitis Risk | None | None | None | Rare | Rare |
| Gallbladder Events | Low | Low | Low | Moderate | Moderate |
| Teratogenicity | No | Yes (topiramate) | No | Uncertain | Uncertain |
| Suicidal Ideation | No | Possible | Boxed Warning | Under review | Under review |
| Discontinuation Rate (AE) | ~4% | ~5% | ~10% | ~7% | ~5% |
GLP-1 Class Safety Considerations
Gastrointestinal Effects
Nausea, vomiting, diarrhea, and constipation are the hallmark side effects of all GLP-1-based therapies. These occur primarily during dose escalation and typically diminish over 4-8 weeks at a stable dose. Slow, stepwise dose titration is the key management strategy. Dietary modifications (smaller meals, avoiding high-fat foods, eating slowly) also help.
Approximately 5-10% of patients find GI side effects intolerable and discontinue treatment. Some patients experience persistent nausea even after dose stabilization, which may limit their maximum tolerated dose and consequently their weight loss.
Pancreatitis
Acute pancreatitis has been reported with all GLP-1 agonists at rates slightly above placebo. In the STEP trials, pancreatitis occurred in 0.2% of semaglutide-treated patients versus 0.1% on placebo. The absolute risk is low, but patients with a history of pancreatitis should use GLP-1 agonists cautiously. GLP-1 therapy should be discontinued immediately if pancreatitis is suspected.
Gallbladder Disease
Rapid weight loss from any cause increases the risk of gallstone formation. GLP-1 agonists may also directly affect gallbladder motility. In the STEP trials, cholelithiasis occurred in approximately 1.6% of semaglutide patients vs 0.6% with placebo. Patients should be counseled about symptoms of gallbladder disease (right upper quadrant pain, nausea after fatty meals).
Thyroid C-Cell Tumors
All GLP-1 agonists carry a boxed warning about the risk of thyroid C-cell tumors based on rodent studies showing medullary thyroid carcinoma (MTC) in rats and mice. The relevance to humans is uncertain, as humans have far fewer GLP-1 receptors on thyroid C-cells than rodents. Large-scale human data have not shown increased MTC risk, but long-term surveillance continues. GLP-1 agonists are contraindicated in patients with personal or family history of MTC or MEN2.
Mental Health
The European Medicines Agency initiated a review of suicidal thoughts and self-harm events with GLP-1 agonists in 2023. Subsequent analyses of clinical trial databases and real-world data have not identified a clear causal association, but monitoring continues. Some patients report mood changes, reduced interest in pleasurable activities (potentially related to the reward pathway effects that suppress food cravings), or depression during treatment.
Weight Regain After Discontinuation
While not a traditional "side effect," weight regain is a predictable consequence of stopping anti-obesity medications. In STEP 4, patients who switched from semaglutide to placebo regained approximately two-thirds of their lost weight over 48 weeks. In SURMOUNT-4, tirzepatide discontinuation led to an average regain of 14 percentage points over 36 weeks. This underscores that obesity is a chronic disease requiring ongoing treatment, much like hypertension or diabetes.
Pipeline Agent Safety Considerations
Retatrutide: The emergence of dysesthesia (abnormal skin sensations) at rates of 9-21% is a novel safety signal not seen with other incretin-based agents. This may be related to the glucagon receptor component and will need careful evaluation. Higher discontinuation rates (12-18%) compared to approved agents are also noteworthy.
Orforglipron: As a small molecule, orforglipron may have different off-target effects than peptide-based GLP-1 agonists. Its safety profile appears broadly consistent with the GLP-1 class based on Phase 3 data, but post-marketing surveillance will be essential.
Bimagrumab: As an anti-myostatin pathway antibody, theoretical concerns include unintended effects on other tissues where activin signaling plays a role (reproductive, cardiovascular). The BELIEVE trial didn't identify major safety signals, but larger, longer trials will be needed.
Psychological and Behavioral Dimensions of Pharmacological Weight Management
The conversation around anti-obesity medications often focuses almost exclusively on weight loss percentages, metabolic biomarkers, and side effect profiles. While these clinical metrics matter enormously, they tell an incomplete story. The psychological and behavioral dimensions of pharmacological weight management profoundly influence treatment outcomes, patient satisfaction, and long-term success. Understanding these dimensions helps clinicians support patients more effectively and helps patients set realistic expectations for their own journeys.
The Psychology of Appetite Suppression
For many patients, the experience of reduced appetite on GLP-1-based medications is psychologically profound in ways that go well beyond simply eating less. Patients who have struggled with persistent food thoughts, intense cravings, and a constant sense of deprivation during previous dieting attempts often describe GLP-1 therapy as the first time they've felt "normal" around food. The mental bandwidth previously consumed by thoughts about eating, resisting cravings, planning meals to avoid temptation, and managing the emotional aftermath of perceived dietary failures suddenly becomes available for other aspects of life. This psychological liberation can be as transformative as the weight loss itself.
However, the sudden removal of food-related preoccupation can also create an unexpected void. For patients who used food as a primary coping mechanism for stress, boredom, loneliness, or emotional distress, appetite suppression removes the coping tool without automatically providing a replacement. This can manifest as increased anxiety, a sense of emptiness or loss, or the emergence of other compensatory behaviors, including increased alcohol consumption, compulsive shopping, or excessive exercise. Mental health screening before initiating anti-obesity pharmacotherapy, and ongoing psychological support during treatment, helps identify patients who may need additional support as their relationship with food changes dramatically.
Body Image and Identity After Significant Weight Loss
Rapid, significant weight loss, particularly the 15-25% body weight reductions now achievable with newer medications, creates a physical transformation that can outpace psychological adaptation. Patients who have been obese for decades may struggle with an updated body image, continuing to perceive themselves as larger than they are (sometimes called "phantom fat"). Social dynamics shift as well: friends and family members may react with jealousy, concern, or unsolicited commentary. Romantic relationships can be destabilized when one partner's appearance changes dramatically while the other's remains the same.
The excess skin that frequently accompanies significant weight loss presents both practical and psychological challenges. While not a medical emergency, loose skin can cause chafing, hygiene difficulties, and profound self-consciousness that undermines the confidence that weight loss was expected to bring. Patients should be counseled early in treatment that excess skin is a common and expected outcome of major weight loss, that it improves somewhat over 12-24 months as skin remodeling occurs, and that surgical body contouring may eventually be desired for certain areas. Peptides like GHK-Cu support collagen remodeling and may modestly improve skin quality during the weight loss process, though they cannot substitute for surgical intervention in cases of severe skin laxity.
The "Food Noise" Phenomenon
The term "food noise," while not a formal clinical designation, has entered the popular and medical vocabulary to describe the persistent, intrusive thoughts about food that many people with obesity experience. This phenomenon appears to involve dysregulation of the mesolimbic dopamine reward system, where food stimuli activate reward pathways with disproportionate intensity, creating a near-constant awareness of food and eating opportunities. GLP-1 receptor agonists, particularly those that cross the blood-brain barrier effectively (like semaglutide), appear to normalize this reward pathway signaling, reducing the intensity and frequency of food-related thoughts.
The reduction in food noise is one of the most consistently reported subjective benefits of GLP-1 therapy, and patients frequently rank it as more valuable than the weight loss itself. This speaks to the significant psychological burden that constant food preoccupation creates, a burden that is often invisible to outside observers and may be minimized or dismissed by healthcare providers who haven't experienced it. Validating the food noise experience and explaining its neurobiological basis helps patients understand that their difficulty with dietary restraint was not simply a matter of willpower but reflected a real neurochemical condition that medication can address.
Behavioral Adaptation and Habit Formation
Pharmacological appetite suppression creates a window of opportunity for behavioral change that many patients have never experienced. When the biological drive to overeat is reduced, patients can more easily establish new eating patterns, develop meal preparation skills, experiment with different foods, and learn to recognize genuine hunger versus emotional eating. The critical question for long-term success is whether patients use this window to build habits that persist beyond medication use, or whether they simply eat less without fundamentally changing their relationship with food.
Structured behavioral support during pharmacological treatment, whether through formal cognitive behavioral therapy (CBT), registered dietitian consultations, health coaching, or structured digital programs, significantly improves both weight loss magnitude and weight loss maintenance. The combination of pharmacological appetite suppression with behavioral skill-building creates a reinforcing loop where each intervention amplifies the other: medication makes behavioral changes easier to implement, and behavioral changes make the medication's effects more durable. Patients who receive both pharmacological and behavioral treatment consistently outperform those who receive either alone, both during active treatment and after medication discontinuation.
Depression, Anxiety, and Mood Changes During Treatment
The relationship between obesity pharmacotherapy and mental health is bidirectional and complex. Depression and anxiety are approximately twice as common in individuals with obesity compared to normal-weight populations, and successful weight loss often improves mood and reduces anxiety. However, the European Medicines Agency initiated a review in 2023 examining reports of suicidal ideation and self-harm with GLP-1 receptor agonists, highlighting that the mental health effects of these medications may not be uniformly positive.
Several mechanisms could mediate mood changes during GLP-1 therapy. First, GLP-1 receptors are expressed in brain regions involved in emotional processing, including the amygdala and prefrontal cortex, and sustained activation of these receptors could influence mood through direct neurochemical effects. Second, the reduction in pleasurable eating can remove a significant source of dopamine-mediated reward, potentially contributing to anhedonia (inability to experience pleasure) in some patients. Third, the physical effects of nausea, fatigue, and GI discomfort during dose titration can independently worsen mood. Fourth, rapid changes in body composition can alter the pharmacokinetics of psychiatric medications that are distributed in body fat, potentially causing breakthrough psychiatric symptoms if doses are not adjusted.
Baseline mental health assessment, ongoing mood monitoring, and proactive psychiatric medication adjustment during weight loss are essential components of comprehensive anti-obesity pharmacotherapy. Patients with pre-existing depression or anxiety should not be excluded from GLP-1 therapy, as the overall balance of evidence suggests net psychological benefit for most patients, but they should receive closer mental health surveillance than patients without psychiatric history.
Obesity Pharmacotherapy in Special Populations
The clinical trial populations that establish drug efficacy and safety represent a narrow slice of the real-world patient population that ultimately uses anti-obesity medications. Special populations, including elderly patients, adolescents, pregnant or breastfeeding women, patients with chronic kidney or liver disease, and individuals from diverse ethnic backgrounds, have unique considerations that generic prescribing guidelines may not adequately address. Understanding these population-specific factors enables more personalized and effective treatment.
Elderly Patients (Age 65 and Older)
Obesity in older adults presents a unique clinical paradox. While excess body fat increases the risk of cardiometabolic disease, falls, functional disability, and joint disease, some degree of adiposity appears protective against all-cause mortality in the elderly, a phenomenon sometimes called the "obesity paradox." This makes the treatment goals for elderly patients fundamentally different from those for younger adults. Rather than targeting maximum weight loss, the emphasis for older patients should be on visceral fat reduction, preservation of lean mass and functional capacity, and improvement of obesity-related comorbidities.
The risk of sarcopenic obesity, where excess fat coexists with inadequate muscle mass and strength, is particularly relevant for older adults on anti-obesity medications. All weight loss, regardless of mechanism, involves some loss of lean tissue alongside fat tissue. In younger adults with adequate muscle reserves, this lean mass loss is usually clinically insignificant. In older adults already near the threshold of functional independence, even modest additional muscle loss can mean the difference between living independently and requiring assisted care. Semaglutide and tirzepatide both produce significant lean mass loss alongside fat loss, and for elderly patients, aggressive resistance training, high-protein diets (1.2-1.6 g/kg/day), and consideration of muscle-protective agents like tesamorelin for GH axis support become essential rather than optional components of the treatment plan.
GLP-1 medications also carry age-specific safety considerations. Older adults have reduced renal clearance, which can affect the elimination of certain metabolites. The nausea and appetite suppression that are tolerable inconveniences in younger patients can cause clinically significant dehydration and malnutrition in elderly patients who may already be eating marginally. Slower dose titration, more frequent monitoring, and lower target doses may be appropriate for patients over 75. Gallbladder disease risk, already elevated with GLP-1 therapy, is further increased by age, and older patients should be counseled about gallstone symptoms and may warrant ursodiol prophylaxis during active weight loss.
Adolescents and Young Adults
The approval of semaglutide 2.4mg (Wegovy) for adolescents aged 12 and older marked a turning point in pediatric obesity management, acknowledging that severe obesity in young people is a medical condition deserving pharmacological treatment rather than simply a behavioral problem requiring more discipline. The STEP TEENS trial demonstrated 16.1% weight loss with semaglutide versus 0.6% weight gain with placebo in adolescents aged 12-17, with a safety profile broadly consistent with adult experience.
However, prescribing anti-obesity medications to developing bodies raises considerations absent from adult treatment. Linear growth continues into the late teens, and caloric restriction during growth periods could theoretically affect final adult height, though the STEP TEENS trial did not show evidence of growth impairment over its 68-week duration. Bone density accrual, which peaks in the early twenties, requires adequate caloric intake and calcium availability; sustained appetite suppression during this critical window warrants monitoring. The hormonal environment of puberty interacts with weight management medications in ways that are not fully characterized, and menstrual irregularity, already common in obese adolescent females, may be influenced by GLP-1 therapy through both weight-dependent and weight-independent mechanisms.
Perhaps most significantly, the psychological implications of pharmacological weight management during adolescence require careful attention. Body image, self-identity, peer relationships, and the development of healthy attitudes toward food and exercise are all in flux during this developmental period. Messaging around medication use should emphasize health improvement rather than appearance, and adolescent patients should have access to counseling that addresses the psychological dimensions of weight management. The fear of weight regain after stopping medication can create medication dependency that, while pharmacologically appropriate (obesity is a chronic condition), may be psychologically burdensome for young patients who see themselves as requiring a drug to be "normal."
Patients with Chronic Kidney Disease
Obesity is both a cause and consequence of chronic kidney disease (CKD), and the intersection of these conditions creates treatment challenges. Semaglutide has shown kidney-protective effects in the FLOW trial, demonstrating a 24% reduction in the composite kidney outcome, making it the first GLP-1 receptor agonist with a dedicated kidney indication. For CKD patients with obesity, this dual benefit of weight reduction and direct renal protection makes semaglutide a particularly attractive option. Tirzepatide's kidney effects are under investigation in the SURPASS-KIDNEY trial, and the FLOW-like benefits may extend to the dual agonist class as well.
Dosing considerations for CKD patients depend on the stage of kidney disease and whether the patient is on dialysis. For most GLP-1 receptor agonists, no dose adjustment is formally required for mild to moderate CKD, as these peptides are primarily degraded by ubiquitous proteolytic enzymes rather than renally cleared. However, the GI side effects of GLP-1 therapy can exacerbate the dehydration and electrolyte imbalances that CKD patients are already prone to, requiring more vigilant monitoring of renal function, electrolytes, and fluid status during dose titration. Patients on dialysis represent a special case where limited clinical trial data exists and where the fluid and nutritional management demands of dialysis complicate the appetite-suppressing effects of GLP-1 therapy.
Ethnic and Genetic Variability in Drug Response
The clinical trials establishing the efficacy of anti-obesity medications have disproportionately enrolled White participants, with variable representation of Black, Hispanic, Asian, and other racial and ethnic groups. This matters because both obesity phenotype and drug response vary by ethnicity. Asian populations, for example, experience metabolic complications at lower BMI thresholds than European-descent populations, which led the WHO to define different BMI cutoffs for obesity in Asian populations. South Asian individuals show a greater propensity for visceral fat accumulation at any given BMI, potentially making visceral fat-targeting agents like tesamorelin more relevant for this population.
Pharmacogenomic factors also influence drug response. Variations in the GLP-1 receptor gene, drug-metabolizing enzymes, and downstream signaling pathways create individual variability in both efficacy and side effect susceptibility. While pharmacogenomic testing for anti-obesity medications is not yet standard practice, the field is moving toward more personalized prescribing that accounts for genetic factors alongside clinical characteristics. In the interim, clinicians should be aware that published trial results may not perfectly predict outcomes for patients from underrepresented backgrounds, and dose titration should be guided by individual response rather than rigid adherence to protocol-derived schedules.
Combining Anti-Obesity Medications with Complementary Peptide Therapies
The emergence of highly effective GLP-1-based obesity treatments has not diminished the relevance of other peptide therapies; if anything, it has increased the strategic value of complementary compounds that address the limitations and side effects of GLP-1-mediated weight loss. A thoughtful combination approach recognizes that weight management is only one dimension of metabolic health and that optimal outcomes require addressing body composition, tissue quality, energy production, sleep, recovery, and cellular aging alongside the primary weight loss intervention.
Growth Hormone Secretagogues for Lean Mass Protection
The most significant limitation of current GLP-1 and dual-agonist therapies is lean mass loss. In the STEP 1 trial, approximately 40% of the weight lost with semaglutide was lean tissue. While some lean mass loss is inevitable during any caloric deficit, the magnitude and rate of loss with potent appetite-suppressing medications raises concerns about long-term muscle function, metabolic rate, and physical capacity, particularly for patients who may use these medications for years or decades.
Growth hormone secretagogues like ipamorelin, CJC-1295/ipamorelin, and tesamorelin offer a physiologically rational approach to lean mass preservation during GLP-1 therapy. By restoring more youthful growth hormone pulsatility, these compounds promote protein synthesis, enhance fat oxidation, and support tissue repair through anabolic pathways that are independent of and complementary to GLP-1's appetite-suppressing mechanism. The logic is straightforward: GLP-1 therapy creates the caloric deficit needed for fat loss, while GH secretagogues help ensure that the body preferentially draws on fat stores rather than muscle tissue to meet its energy needs.
Tesamorelin has particular relevance in this context due to its targeted effects on visceral fat. While GLP-1 agonists reduce both visceral and subcutaneous fat, tesamorelin preferentially mobilizes visceral fat through GH-mediated lipolysis. Combining a GLP-1 agonist with tesamorelin may produce a more favorable pattern of fat loss, where the metabolically dangerous visceral depot is reduced disproportionately, potentially improving cardiometabolic outcomes beyond what either agent achieves alone. Additionally, tesamorelin's hepatic fat-reducing effects address the NAFLD that commonly accompanies obesity, providing liver-specific benefits that GLP-1 therapy alone may not fully address.
Tissue Repair and Recovery Peptides
Patients on anti-obesity medications who are simultaneously increasing their physical activity, as clinical guidelines recommend, often encounter musculoskeletal challenges. Joints that have supported excess weight for years may develop pain and inflammation when subjected to new exercise demands. Tendons and ligaments adapt more slowly than muscles to increased loading, creating a window of vulnerability during the early months of an exercise program. The caloric deficit imposed by GLP-1 therapy can further impair tissue repair by reducing the availability of amino acids and energy for healing processes.
BPC-157 and TB-500 address these tissue repair challenges through complementary mechanisms. BPC-157 promotes healing through the nitric oxide system, growth factor modulation, and angiogenesis, with particular affinity for tendon and GI tissue. TB-500 enhances cell migration, reduces inflammation, and promotes actin polymerization needed for tissue remodeling. For patients beginning exercise programs alongside GLP-1 therapy, these tissue-support peptides can help maintain training consistency by reducing injury-related interruptions and accelerating recovery from the microtrauma that accompanies progressive exercise loading.
Mitochondrial and Cellular Energy Support
Caloric restriction, while metabolically beneficial in many ways, can create an energy availability challenge at the cellular level. Mitochondrial function, which determines how efficiently cells convert nutrients into usable energy, can decline during sustained caloric deficit if the cofactors and substrates needed for optimal mitochondrial function are not adequately supplied. Patients on GLP-1 therapy frequently report fatigue, reduced exercise tolerance, and difficulty maintaining energy levels throughout the day, symptoms that may reflect inadequate mitochondrial support during the caloric deficit.
MOTS-c, a mitochondrial-derived peptide that enhances metabolic flexibility and exercise capacity, offers support for the energy production side of the equation. By promoting AMPK activation and improving the cell's ability to switch between fat and glucose oxidation, MOTS-c can help maintain cellular energy production even during the caloric deficit that GLP-1 therapy creates. NAD+ precursors like NAD+ supplementation support the electron transport chain and sirtuin enzymes that regulate mitochondrial biogenesis, potentially counteracting the mitochondrial stress that caloric restriction can impose. Together, these mitochondrial support compounds help patients maintain the energy levels needed for exercise adherence, daily function, and quality of life during active weight loss.
Longevity and Senolytic Considerations
Obesity accelerates biological aging through multiple mechanisms, including increased oxidative stress, chronic inflammation, telomere shortening, and accumulation of senescent cells. Visceral adipose tissue is a particularly active source of senescent cells, and the inflammatory signals (SASP factors) these cells produce contribute to the systemic inflammation that links obesity to cardiovascular disease, diabetes, cancer, and neurodegeneration. Effective weight loss, particularly visceral fat reduction, directly reduces the senescent cell burden by shrinking the tissue that generates the most senescent cells.
For patients interested in longevity optimization alongside weight management, senolytic peptides like FOXO4-DRI can complement the anti-aging effects of weight loss by actively clearing the senescent cells that have accumulated during years of obesity. The sequential logic is appealing: GLP-1 therapy reduces ongoing senescent cell generation by shrinking visceral fat, while periodic senolytic courses clear the existing backlog of senescent cells that have already accumulated. Humanin and other mitochondrial-derived peptides support the mitochondrial health that underlies cellular resilience against future senescence triggers. This layered approach addresses aging at multiple biological levels, from the organ-level fat reduction that GLP-1 provides to the cellular-level senescent cell clearance that senolytics achieve.
The FormBlends dosing calculator helps structure multi-peptide protocols that integrate weight management with complementary health optimization, and the free assessment provides personalized recommendations based on individual health profiles, goals, and current medications.
Long-Term Management: Maintenance, Discontinuation, and Weight Regain Prevention
Perhaps the single most important concept in obesity pharmacotherapy is that obesity is a chronic, relapsing condition. This understanding, which has taken decades to fully permeate medical practice and public consciousness, fundamentally shapes how anti-obesity medications should be used. Just as hypertension requires ongoing treatment and diabetes requires lifelong management, obesity in most patients requires sustained pharmacological intervention to maintain weight loss. The expectation that a patient can take a medication for 12-18 months, lose weight, stop the medication, and maintain that loss indefinitely is, for most patients, no more realistic than expecting a diabetic to stop insulin and maintain normal blood sugar.
The Biology of Weight Regain
Weight regain after discontinuation of anti-obesity medications is not a failure of willpower. It reflects powerful biological adaptations that evolved to protect against starvation but are maladaptive in an environment of caloric abundance. When body weight decreases substantially, multiple compensatory mechanisms activate simultaneously. Leptin levels drop in proportion to fat mass loss, reducing the satiety signal that suppresses appetite. Ghrelin levels increase, amplifying the hunger signal. Resting metabolic rate decreases beyond what would be predicted from the change in body mass alone, a phenomenon called adaptive thermogenesis or metabolic adaptation. Reward pathway sensitivity to food cues increases, making high-calorie foods more psychologically compelling. These changes can persist for months to years after weight loss and are largely independent of the method used to achieve that loss.
The STEP 4 trial provided the most dramatic illustration of this phenomenon in the GLP-1 era. Patients who had lost an average of 17.3% body weight on semaglutide over 20 weeks were randomized to continue semaglutide or switch to placebo. Over the following 48 weeks, patients who switched to placebo regained approximately two-thirds of their lost weight, while patients who continued semaglutide maintained their weight loss and continued to lose additional weight. The SURMOUNT-4 trial showed a similar pattern with tirzepatide, with patients regaining an average of 14 percentage points of body weight after switching from active treatment to placebo.
Strategies for Sustained Treatment
Given the biology of weight regain, the default approach for most patients should be indefinite continuation of anti-obesity medication at the lowest effective maintenance dose. This doesn't necessarily mean taking the maximum dose forever. Many patients can step down to a lower maintenance dose after achieving their initial weight loss, as the biological set-point mechanisms are less aggressive when weight is being maintained versus when it was being actively lost. For semaglutide, some patients maintain their weight loss on the 1.0mg or 1.7mg dose after reaching their target on 2.4mg. For tirzepatide, stepping down from the 15mg maximum to 10mg or even 7.5mg may maintain results for some patients.
Cost is the primary barrier to indefinite treatment. At current pricing of $1,000-$1,500 per month without insurance, indefinite GLP-1 therapy is financially unsustainable for most patients. Insurance coverage is expanding but remains inconsistent, and many patients experience coverage gaps, formulary changes, or lifetime benefit limits that disrupt treatment continuity. Compounding pharmacies offer more affordable access to certain GLP-1 peptides, particularly semaglutide, and may provide a more sustainable long-term option for patients who cannot maintain access to branded products. The FormBlends semaglutide page provides current information on compounded GLP-1 access.
Planned Discontinuation and Transition Strategies
When medication discontinuation is necessary, whether due to cost, side effects, pregnancy planning, or patient preference, gradual tapering rather than abrupt cessation may reduce the severity of weight regain. While no large trials have specifically studied optimal tapering protocols, the general principle of allowing the body time to adjust to decreasing appetite suppression is physiologically sound. A reasonable approach for semaglutide might involve stepping down from 2.4mg to 1.7mg for 4 weeks, then 1.0mg for 4 weeks, then 0.5mg for 4 weeks, then 0.25mg for 4 weeks before complete discontinuation.
During and after the taper, intensifying behavioral support, increasing protein intake (to maintain satiety through a different mechanism), continuing resistance training (to preserve metabolic rate through lean mass maintenance), and considering complementary approaches like growth hormone optimization can help mitigate weight regain. Some patients transition to less expensive medications, such as naltrexone-bupropion or phentermine-topiramate, as "bridge" therapy when GLP-1 access is lost. These medications are less effective than GLP-1 agonists but may be sufficient to prevent complete weight regain when combined with strong behavioral habits established during GLP-1 therapy.
Monitoring and Follow-Up During Maintenance
Long-term monitoring during obesity pharmacotherapy should include regular assessment of weight and body composition (ideally through DEXA or BIA rather than scale weight alone), metabolic parameters (fasting glucose, HbA1c, lipid panel), liver function, kidney function, and thyroid function. Screening for depression and monitoring mental health should be ongoing, as mood changes can occur at any point during treatment. For patients on GLP-1 therapy, periodic assessment of pancreatic enzymes (lipase, amylase) and gallbladder symptoms helps identify complications early. For patients on combination protocols that include growth hormone secretagogues, IGF-1 monitoring ensures that GH levels remain within the physiological range.
Body composition monitoring deserves particular emphasis during long-term treatment. Patients who are maintaining their weight on medication may be gradually losing lean mass while gaining fat, a phenomenon called weight cycling that worsens body composition over time. Periodic DEXA scans (annually or semi-annually) provide objective data on lean mass and fat mass trends that scale weight alone cannot reveal. If lean mass is declining despite stable scale weight, increasing protein intake, adjusting the exercise program, or adding lean-mass-protective peptides should be considered before the decline becomes functionally significant.
The Future of Obesity Treatment: Personalized Medicine and Emerging Paradigms
The obesity treatment landscape in 2026 is dramatically different from what existed just five years ago, and the pace of innovation shows no signs of slowing. Several converging trends, including pharmacogenomics, digital health technology, combination therapy, and a growing understanding of the heterogeneity of obesity, are pushing the field toward a more personalized, precise, and effective approach to weight management.
Pharmacogenomic-Guided Prescribing
Not all patients respond equally to any given anti-obesity medication, and the reasons for this variability are beginning to be understood at the genetic level. Variations in the GLP-1 receptor gene (GLP1R) affect receptor expression, ligand binding affinity, and downstream signaling efficiency, potentially explaining why some patients lose 20% or more of their body weight on semaglutide while others lose only 5%. Variants in the melanocortin-4 receptor gene (MC4R), which is the most common genetic contributor to monogenic obesity, may predict response to centrally-acting agents that modulate the melanocortin pathway. Polymorphisms in drug-metabolizing enzymes (CYP2D6, CYP3A4) can affect the pharmacokinetics of oral small-molecule agents like orforglipron, influencing both efficacy and side effect profiles.
While routine pharmacogenomic testing for anti-obesity medications is not yet standard, several research groups are developing panels that could guide prescribing decisions. A future scenario in which a patient provides a saliva sample, receives a genetic analysis, and gets a personalized recommendation for the agent most likely to produce optimal results with minimal side effects is technically feasible and commercially inevitable. In the interim, the clinical trial-and-error approach, starting with a first-line agent and switching if results are inadequate, remains the practical standard, but genetic information can increasingly inform these decisions for patients who have access to testing.
Digital Health Integration
The integration of digital health tools with pharmacological obesity treatment is creating treatment paradigms that would have been science fiction a decade ago. Continuous glucose monitors (CGMs) provide real-time feedback on metabolic responses to food, exercise, and medication, allowing patients and clinicians to optimize medication timing and dietary choices based on individual metabolic patterns. Smart scales that track weight, body composition, and hydration status over time provide trend data that is more clinically useful than individual measurements. Mobile apps that use artificial intelligence to track food intake through photo recognition eliminate the tedious food logging that reduces dietary adherence.
Wearable devices that monitor physical activity, sleep quality, heart rate variability, and stress levels provide objective data on the lifestyle factors that influence medication effectiveness. When integrated with electronic health records and prescription data, these digital inputs could enable clinical decision support systems that recommend dose adjustments, flag concerning trends, and identify patients at risk for weight regain before it occurs. The vision of a continuously monitored, algorithmically optimized obesity treatment program is rapidly moving from concept to implementation.
Beyond Weight Loss: The Metabolic Health Paradigm
The definition of treatment success in obesity management is evolving beyond weight loss percentage to encompass a broader concept of metabolic health. A patient who loses 15% of body weight but maintains an unfavorable waist-to-hip ratio, elevated inflammatory markers, and poor glucose regulation has achieved a different outcome than a patient who loses 10% but normalizes their metabolic parameters. This distinction is becoming more clinically relevant as medications with different mechanisms produce different patterns of metabolic improvement.
Tirzepatide, for example, produces greater improvements in insulin sensitivity than would be predicted by weight loss alone, suggesting direct metabolic benefits independent of fat reduction. Retatrutide's glucagon receptor activation may enhance energy expenditure and liver fat clearance in ways that single-agonist GLP-1 therapies cannot. Bimagrumab's muscle-preserving effects maintain metabolic rate and functional capacity even as body weight decreases. Choosing among these agents based on the specific metabolic abnormalities a patient presents, rather than simply targeting maximum weight loss, represents a more sophisticated and ultimately more effective treatment approach.
Combination Pharmacotherapy
The future of obesity treatment almost certainly involves rational combination therapy, where agents with complementary mechanisms are used together to target multiple biological pathways simultaneously. This approach mirrors the evolution of treatment in hypertension, HIV, and oncology, where multi-drug regimens have consistently proven more effective than single agents. For obesity, several combination strategies are under investigation or already being used off-label.
GLP-1 agonist plus growth hormone secretagogue addresses both appetite suppression and lean mass preservation, potentially producing a more favorable body composition outcome than either alone. GLP-1 agonist plus amylin analog (as in the CagriSema combination) targets multiple satiety pathways simultaneously, producing additive appetite suppression. GLP-1 agonist plus SGLT2 inhibitor combines appetite suppression with caloric wasting through urinary glucose excretion, an approach showing promise in diabetic patients with obesity. GLP-1 agonist plus NNMT inhibitor could address both appetite and cellular metabolism, though this combination is theoretical at this point.
The challenge with combination therapy lies in managing complexity: more medications mean more potential interactions, more side effects to monitor, and higher costs. Careful pharmacovigilance, standardized combination protocols, and clinical decision support tools will be needed as combination approaches move from isolated clinical practice into mainstream treatment guidelines.
The Equity Challenge
As obesity pharmacotherapy becomes increasingly effective, the equity challenge becomes increasingly urgent. The most effective medications are also the most expensive, creating a scenario where the patients with the greatest need, often those from lower socioeconomic backgrounds who face higher obesity rates due to food environment, stress, and access factors, have the least access to treatment. The arrival of oral small-molecule agents like orforglipron, which should be cheaper to manufacture than peptide-based injectables, may help narrow this gap, but only if pricing reflects manufacturing costs rather than what-the-market-will-bear strategies.
Insurance coverage decisions, compounding pharmacy access, patient assistance programs, and public health policy will all shape how equitably the benefits of the obesity pharmacotherapy revolution are distributed. The medical community has a responsibility to advocate for access, and patients have a responsibility to demand that effective treatments not be rationed based on ability to pay. The FormBlends GLP-1 Research Hub tracks access developments alongside scientific advances, recognizing that a treatment that exists but isn't accessible is only partially a treatment at all.
For patients navigating the current range of obesity treatment options, the FormBlends free assessment provides personalized guidance on which combination of pharmacological, peptide, and lifestyle interventions best matches their metabolic profile, goals, and access situation.
Nutrition, Exercise, and Lifestyle Integration with Pharmacotherapy
Anti-obesity medications work best when embedded within a comprehensive lifestyle framework. The common misconception that pharmacotherapy eliminates the need for dietary and exercise modifications is contradicted by every major clinical trial, where participants in both active and placebo arms receive lifestyle counseling. The medications create a more favorable physiological environment for lifestyle changes, reducing the biological drive to overeat and, in some cases, directly enhancing metabolic responses to exercise. But the lifestyle component remains essential for maximizing weight loss, preserving lean mass, preventing nutritional deficiencies, and establishing the habits that determine long-term success.
Protein Optimization During Pharmacological Weight Loss
Protein intake becomes a critical variable during GLP-1-mediated weight loss for several reasons. First, the appetite suppression produced by these medications often reduces total food intake by 25-40%, and if protein intake decreases proportionally, the resulting protein deficit accelerates lean mass loss. Second, protein has the highest thermic effect of any macronutrient, meaning that maintaining or increasing protein intake partially offsets the metabolic rate reduction that accompanies weight loss. Third, protein produces greater satiety per calorie than carbohydrates or fat, synergizing with the medication's appetite-suppressing effects to produce a more comfortable caloric deficit.
Current evidence supports a protein intake of 1.2-1.6 grams per kilogram of body weight per day during pharmacological weight loss, with the higher end of this range appropriate for patients engaging in resistance training. For a 100 kg patient, this translates to 120-160 grams of protein daily, which can be challenging to achieve when overall appetite is suppressed. Practical strategies include prioritizing protein at each meal (eating protein foods first before filling up on lower-priority foods), using protein supplements (whey, casein, or plant-based protein powders) to reach daily targets when whole food intake is insufficient, and distributing protein intake across 3-4 meals rather than concentrating it in one or two meals, as muscle protein synthesis has a per-meal ceiling of approximately 40-50 grams.
The type of protein also matters. Animal proteins (meat, fish, eggs, dairy) provide all essential amino acids in proportions that closely match human requirements and are generally more bioavailable than plant proteins. Leucine, the amino acid that most potently stimulates muscle protein synthesis through mTOR activation, is present in higher concentrations in animal proteins, particularly dairy (whey protein is approximately 11% leucine by weight). For patients following plant-based diets, combining complementary protein sources and supplementing with leucine (2.5-3 grams per meal) can compensate for the lower leucine content of plant proteins. Essential amino acid (EAA) supplements provide another option for patients who struggle to meet protein targets through whole foods alone.
Micronutrient Considerations
Reduced food intake during GLP-1 therapy creates a risk of micronutrient deficiencies that is often overlooked in clinical practice. When total caloric intake decreases by 25-40%, intake of vitamins and minerals decreases proportionally unless dietary quality improves dramatically. The micronutrients most likely to become deficient during pharmacological weight loss include iron (particularly in premenopausal women), vitamin B12, folate, zinc, magnesium, calcium, and vitamin D. A high-quality multivitamin provides baseline insurance against these deficiencies, and targeted supplementation based on periodic lab testing addresses individual needs.
Vitamin D deserves particular attention. Vitamin D is stored in adipose tissue, and paradoxically, patients with obesity often have low circulating vitamin D levels because the vitamin is sequestered in fat stores rather than being available for metabolic use. As fat mass decreases during treatment, stored vitamin D may be released into circulation, but this release is unpredictable and may not maintain adequate levels. Vitamin D supports bone health (critical during weight loss, when reduced mechanical loading threatens bone density), immune function, mood regulation, and muscle function. Maintaining serum 25-hydroxyvitamin D levels above 40 ng/mL through supplementation (typically 2,000-5,000 IU daily, adjusted based on blood levels) supports all of these functions during the metabolic stress of weight loss.
Omega-3 fatty acid intake often decreases during GLP-1 therapy as patients reduce consumption of fatty foods. The EPA and DHA in omega-3s have anti-inflammatory effects, support cardiovascular health, improve insulin sensitivity, and may directly enhance the response to GLP-1 therapy by reducing the systemic inflammation that contributes to metabolic dysfunction. Supplementing with 2-3 grams of combined EPA and DHA daily provides these benefits independently of dietary fat intake.
Exercise Programming for Pharmacologically Treated Patients
Exercise during anti-obesity pharmacotherapy serves several purposes beyond caloric expenditure: preserving lean mass, maintaining metabolic rate, improving cardiovascular fitness, supporting bone density, enhancing mood, and improving body composition quality (the ratio of lean mass to fat mass). The type, intensity, and frequency of exercise all influence which of these benefits are realized.
Resistance training (weightlifting, bodyweight exercises, resistance band work) is the single most effective exercise modality for lean mass preservation during weight loss. A minimum of 2-3 sessions per week targeting all major muscle groups, using loads heavy enough to reach near-failure within 8-15 repetitions, provides the mechanical and metabolic stimulus needed to signal the body that muscle tissue must be preserved. Patients new to resistance training benefit from professional instruction (personal training, group fitness classes, or online coaching programs) to learn proper form and develop sustainable training habits. The first 4-8 weeks of resistance training produce rapid strength gains that are primarily neurological (improved muscle recruitment efficiency) rather than hypertrophic (increased muscle size), meaning that patients should not be discouraged by initial lack of visible muscle development.
Cardiovascular exercise (walking, cycling, swimming, jogging) complements resistance training by improving cardiovascular fitness, enhancing fat oxidation, and supporting mental health. However, excessive cardiovascular exercise without adequate resistance training can actually accelerate lean mass loss during caloric deficit, particularly if the cardiovascular exercise is prolonged and moderate-intensity (the type that preferentially uses mixed fuel including amino acids from muscle protein). A balanced program that prioritizes resistance training (3-4 sessions per week) with moderate cardiovascular exercise (150-200 minutes per week at moderate intensity) provides the best body composition outcomes.
High-intensity interval training (HIIT) offers time-efficient cardiovascular benefits with less risk of excessive muscle catabolism than prolonged steady-state cardio. Short bursts of high-intensity effort (20-30 seconds) alternating with recovery periods (60-90 seconds) for 15-20 minutes can produce cardiovascular adaptations comparable to 45-60 minutes of moderate continuous exercise. HIIT also produces a greater EPOC (excess post-exercise oxygen consumption) effect, increasing metabolic rate for hours after the session. For patients on GLP-1 therapy who struggle with exercise motivation due to reduced energy intake, HIIT's time efficiency can make the difference between maintaining and abandoning an exercise program.
Sleep, Stress, and Circadian Alignment
The lifestyle factors that surround pharmacotherapy influence its effectiveness in ways that are often underappreciated. Sleep duration and quality affect hormone levels (cortisol, growth hormone, leptin, ghrelin), metabolic rate, insulin sensitivity, food choices, and exercise motivation. Patients sleeping fewer than 7 hours per night consistently show reduced weight loss on identical caloric deficits compared to adequate sleepers, and the composition of their weight loss shifts unfavorably toward lean mass loss. For patients on GLP-1 therapy, optimizing sleep may be as impactful as adding a second medication.
Chronic psychological stress elevates cortisol levels, which promote visceral fat accumulation, increase appetite for calorie-dense foods, impair insulin sensitivity, and accelerate muscle protein breakdown. The stress-obesity cycle is self-reinforcing: obesity increases stress (through health concerns, social stigma, and reduced physical capacity), and stress promotes weight gain and fat redistribution. Breaking this cycle often requires deliberate stress management practices, including regular physical activity, adequate sleep, social connection, and for some patients, formal stress reduction techniques like meditation, breathwork, or cognitive behavioral strategies. Adaptogenic supplements and anxiolytic peptides like Selank can provide additional support for patients whose stress levels are undermining their weight management efforts.
Meal timing and circadian alignment are emerging as additional variables that influence obesity treatment outcomes. Late-night eating, independent of total caloric intake, appears to promote fat storage and impair metabolic health, likely because insulin sensitivity follows a circadian rhythm that peaks in the morning and declines in the evening. For patients on GLP-1 therapy, confining eating to a 10-12 hour window aligned with daylight hours (for example, 7 AM to 7 PM) may enhance the metabolic benefits of reduced caloric intake. This approach, sometimes called time-restricted eating, doesn't require additional caloric restriction beyond what the medication produces but optimizes when calories are consumed for metabolic benefit.
Real-World Outcomes, Clinical Observations, and Practical Expectations
Clinical trial results, while essential for establishing drug efficacy and safety, represent an idealized treatment scenario that often differs from real-world clinical practice. Trial participants are carefully selected, closely monitored, receive free medication, and benefit from the structure and accountability of a clinical study. Real-world outcomes, while still impressive for modern anti-obesity medications, typically fall somewhat below clinical trial benchmarks. Understanding this gap helps patients and clinicians set appropriate expectations.
Real-World Efficacy Data
Several retrospective analyses and observational studies have examined GLP-1 agonist outcomes outside of clinical trials. Data from electronic health records show that real-world weight loss with semaglutide 2.4mg averages approximately 10-12% at one year, compared to the 14.9% seen in STEP 1. For tirzepatide, real-world data are more limited due to its more recent availability, but early reports suggest weight loss of 13-17% at one year, compared to the 19-21% seen in SURMOUNT-1 at the highest dose. These gaps reflect several real-world factors: less consistent dose titration (patients often reach maximum doses more slowly due to side effects), lower adherence rates (missed doses, treatment interruptions), less intensive lifestyle support, and the inclusion of patients with more complex medical histories than those enrolled in trials.
Adherence is perhaps the most significant real-world factor. In clinical trials, where medication is free, visits are scheduled, and participants are motivated by the study experience, adherence rates typically exceed 85%. In real-world practice, where patients pay for medication, manage competing priorities, and may experience "treatment fatigue" over time, 12-month adherence rates for GLP-1 agonists range from 50-70%. Each missed dose represents a period of reduced appetite suppression during which compensatory eating can occur. For patients using weekly injectables, a single missed dose means a full week without medication effect. Improving adherence through simplified dosing, reduced side effects (areas where oral agents like orforglipron may have advantages), and digital reminders can narrow the gap between trial and real-world outcomes.
Predictors of Treatment Success
Not every patient responds equally to anti-obesity medications, and identifying predictors of success helps clinicians select the right medication for the right patient. Early weight loss response is the most reliable predictor of long-term outcomes: patients who achieve at least 5% weight loss within the first 12-16 weeks of treatment consistently show the best long-term results. This observation has led to the recommendation that patients who don't achieve a 5% weight loss threshold after adequate dose titration should be considered for medication switching rather than indefinite continuation of an ineffective therapy.
Patient factors that predict better GLP-1 response include higher baseline BMI (patients with BMI above 40 tend to lose a larger absolute amount of weight, though similar or slightly lower percentage weight loss compared to those with BMI 30-40), younger age, female sex, higher baseline fasting insulin levels (suggesting greater insulin resistance that GLP-1 therapy can address), and absence of psychiatric comorbidities that complicate behavioral adherence. Genetic factors, particularly variations in the GLP-1 receptor gene and melanocortin pathway genes, likely influence response but are not yet routinely tested.
Lifestyle engagement is a powerful effect modifier. Patients who combine GLP-1 therapy with structured exercise programs and dietary optimization consistently outperform patients who rely on medication alone. In the STEP 3 trial, which combined semaglutide with intensive behavioral therapy (30 individual counseling sessions plus a 1,000-1,200 calorie liquid diet for the first 8 weeks), participants achieved 16% weight loss compared to the 14.9% in STEP 1 with standard lifestyle support. This difference, while modest in percentage terms, may translate into clinically meaningful additional benefits for patients at the margin of achieving diabetes remission, clinically significant cardiovascular risk reduction, or resolution of other obesity-related comorbidities.
Managing Expectations
Setting realistic expectations is one of the most important clinical tasks when initiating anti-obesity pharmacotherapy. Patients who expect their medication to produce complete appetite suppression, effortless weight loss, and permanent results without ongoing treatment are set up for disappointment. A more realistic framing positions the medication as a powerful tool that reduces biological barriers to weight management but doesn't eliminate the need for conscious food choices, regular physical activity, and ongoing adherence.
The timeline of results also matters. GLP-1 agonists require a dose titration period of 4-16 weeks (depending on the medication and the individual's tolerance of GI side effects) before reaching the effective dose. Weight loss during this titration period is typically modest, and patients who expect dramatic results immediately may become discouraged and discontinue before the medication has a fair chance to work. Educating patients that meaningful weight loss begins 4-8 weeks after reaching the target dose, with maximum effects typically observed at 9-12 months, helps maintain motivation during the slow early phase.
The social dimension of weight loss also deserves attention. Patients often find that weight loss changes their social relationships in unexpected ways. Friends and family members who were comfortable with the patient's larger size may feel threatened by the change, sometimes expressing concern about "losing too much weight" or questioning whether the patient is "really healthy." Workplace dynamics can shift as well, with some patients reporting increased professional opportunities and social attention, which, while positive, can create its own stresses. Support groups, whether in-person or online, that connect patients going through similar experiences provide valuable peer support and normalization of the complex emotions that accompany significant physical transformation. Some patients find that individual therapy focused specifically on identity transition during weight loss helps them process the psychological changes that accompany the physical ones. Weight loss plateaus are a normal and expected part of treatment, not a sign of medication failure. As body weight decreases, metabolic rate decreases (both from loss of metabolically active tissue and from adaptive thermogenesis), creating a new energy balance point at a lower weight. Patients may experience months where scale weight doesn't change despite continued medication adherence and lifestyle compliance. During these plateaus, body composition often continues to improve (fat loss offset by lean mass stability or gain from exercise), but this improvement isn't visible on the scale. Regular body composition assessment and measurement of waist circumference provide more complete progress information during plateau periods.
When to Switch or Augment Therapy
Treatment algorithms for anti-obesity pharmacotherapy are evolving but generally follow a stepped approach. If the first-line agent (typically semaglutide or tirzepatide, based on insurance coverage and provider preference) produces inadequate results after 16-24 weeks at the maximum tolerated dose, several options exist. Switching from a single GLP-1 agonist to a dual agonist (semaglutide to tirzepatide) often produces additional weight loss, as the GIP receptor activation adds a complementary mechanism. Switching from an injectable to an oral formulation (or vice versa) may improve adherence for patients whose primary barrier is the route of administration rather than the pharmacological mechanism.
Augmentation, adding a second agent rather than switching, is gaining clinical traction. Adding naltrexone-bupropion to a GLP-1 agonist targets the reward pathway from a different angle, potentially addressing the "food noise" that persists in some patients despite GLP-1 therapy. Adding a growth hormone secretagogue addresses lean mass loss without interfering with the GLP-1 mechanism. Adding an SGLT2 inhibitor (in diabetic patients) creates caloric wasting through urinary glucose excretion, providing an additive caloric deficit that is independent of appetite. These augmentation strategies are increasingly used in clinical practice despite limited randomized controlled trial data for specific combinations, reflecting the pragmatic reality that many patients need multi-mechanism approaches to achieve and maintain clinically meaningful weight loss. The concept of "combination optimization" is gaining traction in obesity medicine circles, where practitioners recognize that the era of single-agent therapy, while producing unprecedented results compared to historical standards, may still fall short of what multi-agent protocols can achieve. Just as oncologists learned decades ago that drug combinations produce better outcomes than sequential monotherapy, obesity medicine specialists are discovering that thoughtfully constructed combination regimens addressing appetite, metabolism, body composition, and cellular health simultaneously produce outcomes that no single agent can match. Documenting these combination protocols through registries and prospective observational studies will be essential for building the evidence base that supports their eventual incorporation into treatment guidelines.
The FormBlends free assessment evaluates individual health profiles and provides personalized recommendations that account for the multi-dimensional nature of weight management, from primary pharmacological approaches to complementary peptide support and lifestyle optimization strategies.
Obesity Pharmacotherapy and Comorbidity Resolution
While weight loss is the primary endpoint in most anti-obesity medication trials, the resolution or improvement of obesity-related comorbidities is arguably more clinically meaningful. A patient who loses 12% of body weight and achieves remission of type 2 diabetes, resolution of sleep apnea, and normalization of blood pressure has gained far more health benefit than a patient who loses 18% but sees no change in their comorbidity profile. Understanding how different anti-obesity medications affect specific comorbidities helps clinicians select the most appropriate agent for individual patients and helps patients understand the full scope of benefits they can expect.
Type 2 Diabetes Remission and Glycemic Control
GLP-1 receptor agonists were originally developed as diabetes medications before their weight loss effects were recognized, and their glycemic benefits remain among their most significant clinical advantages. Semaglutide 2.4mg in the STEP 2 trial (which enrolled patients with both obesity and type 2 diabetes) produced HbA1c reductions of 1.6 percentage points alongside 9.6% weight loss. Tirzepatide's glycemic effects are even more pronounced; in the SURPASS trials for diabetes, the 15mg dose achieved HbA1c reductions of 2.1-2.3 percentage points, with 62% of patients reaching an HbA1c below 5.7% (the normal range), effectively achieving diabetes remission through pharmacological means.
The mechanism of glycemic improvement extends beyond weight loss. GLP-1 directly stimulates glucose-dependent insulin secretion, suppresses glucagon release, and slows gastric emptying (which reduces postprandial glucose excursions). GIP, the second target of tirzepatide, enhances beta-cell function and may improve insulin sensitivity through adipose tissue effects. For patients with type 2 diabetes and obesity, these direct metabolic benefits mean that glycemic improvement often occurs rapidly, within the first few weeks of treatment, before significant weight loss has occurred. This early metabolic benefit can be highly motivating for patients and provides immediate clinical value even during the slow dose titration phase.
The question of whether pharmacological diabetes remission is durable after medication discontinuation remains open. Some data suggest that patients who achieve remission on GLP-1 therapy and maintain it for 12 or more months may have a period of sustained remission after stopping the medication, particularly if they've made substantial lifestyle changes during treatment. However, most experts recommend continued therapy to maintain glycemic control, just as they recommend continued treatment to maintain weight loss. The chronic disease model applies: type 2 diabetes, like obesity, tends to recur when the treatment that produced remission is withdrawn.
Cardiovascular Risk Reduction
The cardiovascular benefits of GLP-1 receptor agonists are among the most well-established findings in modern cardiometabolic medicine. The SELECT trial demonstrated that semaglutide 2.4mg reduced major adverse cardiovascular events (heart attack, stroke, or cardiovascular death) by 20% in patients with overweight or obesity and established cardiovascular disease, even in patients without diabetes. This finding established semaglutide as the first anti-obesity medication with proven cardiovascular benefit, moving the field beyond the historical concern that obesity drugs might increase cardiovascular risk (as fenfluramine famously did).
The cardiovascular benefits appear to extend beyond what weight loss alone would predict. GLP-1 receptors are expressed on cardiomyocytes, vascular endothelial cells, and smooth muscle cells, and GLP-1 signaling has direct cardioprotective effects including reduced myocardial inflammation, improved endothelial function, decreased atherogenesis, and enhanced cardiac glucose uptake. These direct cardiovascular effects, combined with improvements in traditional risk factors (blood pressure, lipids, glucose, waist circumference), produce a multi-level cardiovascular protection that is difficult to achieve with any single lifestyle or pharmacological intervention.
For patients with established cardiovascular disease and obesity, semaglutide has become a first-line consideration, not just for weight management but for cardiovascular risk reduction. The overlap between cardiology and obesity medicine is growing, and cardiologists are increasingly prescribing GLP-1 agonists for their cardiovascular benefits with weight loss as a welcome secondary effect. Tirzepatide's cardiovascular outcomes data (from the SURPASS-CVOT trial) are anticipated and will determine whether the dual agonist approach provides similar or superior cardiovascular protection.
Non-Alcoholic Fatty Liver Disease
NAFLD affects the majority of patients with obesity and progresses to NASH (with inflammation and fibrosis) in approximately 20-30% of cases. Both semaglutide and tirzepatide reduce hepatic fat content, with tirzepatide showing particularly impressive results in the Combined effect-NASH trial, where the 15mg dose achieved NASH resolution in 74% of participants and fibrosis improvement in 59%. These results suggest that incretin-based therapies may become first-line treatments for NASH, a condition that currently has only one FDA-approved medication (resmetirom, approved in 2024 for NASH with moderate to advanced fibrosis).
The mechanism of hepatic benefit involves both direct metabolic effects (improved hepatic insulin sensitivity, enhanced fat oxidation, reduced de novo lipogenesis) and indirect effects mediated through weight loss and reduced visceral fat (which decreases portal free fatty acid delivery to the liver). For patients with documented NAFLD or NASH, selecting an anti-obesity medication with strong hepatic evidence makes clinical sense. Combining GLP-1 therapy with tesamorelin, which has independent evidence for hepatic fat reduction through GH-mediated pathways, creates a dual-mechanism approach to liver fat that may be more effective than either agent alone.
Obstructive Sleep Apnea
The SURMOUNT-OSA trial demonstrated that tirzepatide reduced the apnea-hypopnea index (AHI) by approximately 50-60% in patients with moderate-to-severe obstructive sleep apnea (OSA) and obesity, with 42-52% of patients achieving disease resolution (AHI below 5). These results establish anti-obesity pharmacotherapy as a viable treatment for OSA, potentially reducing dependence on CPAP therapy for many patients. The mechanism is straightforward: weight loss reduces the pharyngeal fat deposition and tissue bulk that contribute to airway obstruction during sleep. However, the relationship between weight loss and OSA improvement is not perfectly linear, and some patients experience significant OSA improvement with relatively modest weight loss, suggesting that the metabolic effects of GLP-1 therapy may influence upper airway function through mechanisms beyond simple fat reduction.
For patients with both obesity and OSA, the improvement in sleep quality that accompanies OSA resolution creates a positive feedback loop. Better sleep improves growth hormone secretion, insulin sensitivity, appetite regulation, exercise tolerance, and mood, all of which support further weight loss and metabolic improvement. Breaking the OSA-obesity cycle through pharmacological weight management can produce benefits that exceed the sum of the individual improvements in each condition.
Osteoarthritis and Joint Health
Excess body weight places enormous mechanical stress on weight-bearing joints, with every kilogram of body weight translating to approximately 4 kilograms of force across the knee joint during walking. Weight loss of 10% or more consistently improves osteoarthritis symptoms, reduces pain medication requirements, and delays the need for joint replacement surgery. For patients with severe osteoarthritis who are not surgical candidates (either due to surgical risk from obesity or because their BMI exceeds the threshold for joint replacement surgery, which many orthopedic surgeons set at BMI 40), pharmacological weight loss can be a pathway to surgical candidacy.
Beyond mechanical offloading, GLP-1 therapy may have direct anti-inflammatory effects on joint tissue. GLP-1 receptors have been identified on synovial cells and chondrocytes, and preclinical data suggest that GLP-1 signaling reduces inflammatory cytokine production in joint tissue. Whether these direct effects are clinically meaningful in humans remains to be determined, but the consistent reports of disproportionate joint symptom improvement relative to weight loss suggest that something beyond simple mechanical relief is occurring.
Patients pursuing weight loss for joint health should be counseled that exercise, despite the short-term joint discomfort it may cause, is essential for long-term joint function. Low-impact activities like cycling, swimming, elliptical training, and walking on flat surfaces provide cardiovascular and metabolic benefits without excessive joint stress. Tissue-support peptides like BPC-157 and TB-500 may support joint tissue healing during the transition to a more active lifestyle, helping patients maintain the exercise consistency that optimizes both weight loss and joint outcomes.
Mental Health and Cognitive Function
The relationship between obesity pharmacotherapy and mental health outcomes encompasses both opportunities and concerns. Weight loss in patients with obesity consistently improves measures of depression, anxiety, quality of life, and self-esteem in most studies, reflecting the psychological burden that obesity imposes and the relief that successful treatment provides. The SELECT trial showed improvements in patient-reported quality of life measures alongside cardiovascular benefits, and the STEP trials consistently demonstrated improvements in physical functioning scores.
Cognitive function is an emerging area of interest. Obesity is associated with accelerated cognitive decline and increased dementia risk through mechanisms including insulin resistance (the brain is highly insulin-dependent), neuroinflammation, cerebrovascular disease, and altered brain-derived neurotrophic factor (BDNF) levels. GLP-1 receptors are expressed throughout the brain, and preclinical evidence suggests neuroprotective effects of GLP-1 signaling independent of weight loss. Several clinical trials are investigating semaglutide and tirzepatide for Alzheimer's disease prevention and treatment, based on the hypothesis that restoring brain GLP-1 and GIP signaling may slow neurodegeneration. While results are pending, the potential for anti-obesity medications to provide cognitive protection alongside metabolic benefits represents one of the most exciting frontiers in the field.
The FormBlends GLP-1 Research Hub provides ongoing coverage of comorbidity-specific outcomes data as new trial results emerge, and the dosing calculator helps structure treatment protocols that account for the full spectrum of metabolic health goals beyond weight loss alone.
Regulatory Landscape, Drug Shortages, and Access Challenges
The practical reality of obesity pharmacotherapy in 2026 is shaped as much by regulatory decisions, supply chain dynamics, and insurance policies as by the science itself. Patients and clinicians navigate a complex environment where the most effective medications may not be available, affordable, or covered, and where the regulatory framework is struggling to keep pace with rapid pharmaceutical innovation and unprecedented demand.
The Supply Crisis and Its Aftermath
The explosive demand for semaglutide and tirzepatide beginning in 2023 created medication shortages that persisted through much of 2024 and into 2025. These shortages affected both the obesity indications (Wegovy, Zepbound) and the diabetes indications (Ozempic, Mounjaro), forcing difficult allocation decisions where pharmacies rationed supplies and new patients were unable to start therapy. The shortages highlighted the fundamental supply chain vulnerability of biologic peptide medications, which require specialized manufacturing facilities, complex quality control processes, and long lead times to scale production.
The shortage period had several lasting effects on the obesity treatment landscape. First, it accelerated interest in compounding pharmacy alternatives, as patients who couldn't access branded products turned to compounded semaglutide as a substitute. This compounding surge prompted both regulatory scrutiny (the FDA issued multiple warnings about compounded semaglutide products) and legitimate access expansion, as established 503B outsourcing facilities increased their GLP-1 production capacity. The availability of compounded semaglutide through pharmacies like FormBlends provided continuity of care for patients who would otherwise have been forced to discontinue therapy during the shortage.
Second, the shortage accelerated pharmaceutical company investment in manufacturing capacity. Novo Nordisk committed billions of dollars to new manufacturing facilities specifically for semaglutide production, while Eli Lilly expanded its tirzepatide manufacturing infrastructure. These investments, while necessary, take years to translate into increased supply, and the lag between demand growth and manufacturing capacity expansion may create future shortage episodes as new indications (cardiovascular risk reduction, NASH, sleep apnea, kidney disease) expand the eligible patient population far beyond the obesity and diabetes populations originally anticipated.
The FDA 503A/503B Compounding Framework
The FDA's regulation of compounded medications operates under a framework established by the Drug Quality and Security Act of 2013, which distinguishes between two types of compounding operations. Section 503A compounding pharmacies prepare medications for individually identified patients based on prescriptions, operating under state pharmacy board oversight. Section 503B outsourcing facilities register with the FDA and can produce larger batches of compounded medications without patient-specific prescriptions, subject to FDA inspection and current good manufacturing practice (cGMP) requirements.
For GLP-1 medications, the regulatory question hinges on whether the branded products are on the FDA Drug Shortage List. When a drug is in shortage, compounding pharmacies have broader latitude to produce copies of the branded product. When the shortage is resolved, the regulatory basis for compounding narrows, though it doesn't necessarily disappear, as compounding for individual patients with specific clinical needs (such as those who require different concentrations, combination formulations, or alternative delivery methods) remains permitted under 503A rules. This regulatory nuance creates uncertainty for patients who have established effective treatment protocols with compounded products and face the possibility that supply could be disrupted by regulatory decisions.
The quality variation between compounding operations is a legitimate concern. While 503B outsourcing facilities undergo FDA inspection and must follow cGMP standards, 503A pharmacies are primarily regulated at the state level, and the rigor of state oversight varies considerably. Reports of subpotent, superpotent, or contaminated compounded semaglutide have emerged, underscoring the importance of sourcing from reputable operations that provide certificates of analysis, conduct sterility testing, and maintain transparent quality documentation. Patients should verify that their compounding pharmacy conducts third-party potency and sterility testing and that the results are available for review upon request.
Insurance Coverage Evolution
Insurance coverage for anti-obesity medications has been one of the most contentious areas in health policy, reflecting a historical reluctance to classify obesity as a disease deserving pharmacological treatment. As recently as 2022, the majority of commercial insurance plans and most state Medicaid programs excluded anti-obesity medications from coverage. The situation has been evolving rapidly since then, driven by several converging factors.
The cardiovascular outcomes data from the SELECT trial, which demonstrated a 20% reduction in major adverse cardiovascular events with semaglutide, shifted the clinical framing of these medications from "weight loss drugs" to "cardiovascular risk reduction agents." This reframing is strategically significant because insurers who exclude weight loss medications from coverage often cover cardiovascular disease prevention. The approval of semaglutide for cardiovascular risk reduction in patients with obesity and established cardiovascular disease provided a coverage pathway that bypasses the "weight loss exclusion" in many insurance formularies.
Medicare's treatment of anti-obesity medications deserves particular attention. Historically, Medicare Part D has been prohibited by statute from covering medications prescribed for weight loss, a restriction that affects tens of millions of elderly Americans with obesity. Legislative efforts to remove this exclusion have gained momentum, with the Treat and Reduce Obesity Act reintroduced in Congress with bipartisan support. If passed, this legislation would open Medicare coverage for anti-obesity medications, dramatically expanding access for the population that bears the highest burden of obesity-related comorbidities.
Employer-sponsored insurance has shown the most rapid coverage expansion. Large self-insured employers, who directly bear the healthcare costs of their employees, have increasingly recognized that the cost of GLP-1 therapy (approximately $12,000-$18,000 per year at list price) may be offset by reduced costs for diabetes management, cardiovascular events, joint replacements, sleep apnea treatment, and disability claims. Some employers have implemented tiered coverage that requires documentation of lifestyle modification efforts before pharmacological coverage is approved, a compromise between universal access and cost containment that reflects the emerging consensus that medication works best as part of a comprehensive treatment approach.
Prior Authorization and Step Therapy Barriers
Even when insurance nominally covers anti-obesity medications, prior authorization requirements and step therapy mandates create practical barriers that delay or prevent access for many patients. Prior authorization typically requires documentation of BMI above 30 (or above 27 with comorbidities), evidence of failed lifestyle modification attempts, and sometimes documentation of failed treatment with less expensive medications before approving GLP-1 therapy. The administrative burden of prior authorization falls disproportionately on smaller practices without dedicated staff to manage the paperwork, potentially creating access disparities based on practice size and resources rather than clinical need.
Step therapy requirements, which mandate that patients try and fail less expensive medications before accessing GLP-1 agonists, have been criticized as clinically inappropriate for several reasons. First, the efficacy gap between older agents (orlistat produces 3-5% weight loss; naltrexone-bupropion produces 5-9%) and GLP-1 agonists (14-21%) is so large that requiring failure on inferior agents delays effective treatment for months while patients continue to experience the health consequences of obesity. Second, the concept of "failure" on a less effective medication is poorly defined, as a patient who loses 5% on naltrexone-bupropion has technically "responded" but may still carry substantial excess weight and metabolic risk. Several states have enacted step therapy reform legislation that gives physicians the ability to override step therapy requirements when clinically indicated, and federal legislation addressing step therapy in employer-sponsored plans is under active consideration by both chambers of Congress, with growing bipartisan support driven by constituent pressure from patients who have experienced firsthand the frustration of bureaucratic barriers delaying access to effective treatment.
Patient assistance programs offered by pharmaceutical manufacturers provide another access pathway for patients who cannot afford out-of-pocket costs. Novo Nordisk's savings card program reduces the cost of Wegovy to $0-$25 per month for commercially insured patients, while Eli Lilly offers similar programs for Zepbound. These programs, while valuable, typically exclude patients on government insurance (Medicare, Medicaid, Tricare) and may have annual caps on total savings. For patients who fall through the cracks of insurance coverage and manufacturer assistance, compounding pharmacies offer the most cost-effective access to GLP-1 therapy, with compounded semaglutide typically priced at $200-$500 per month, representing a 70-85% cost reduction compared to branded products.
International patients face additional access challenges that vary dramatically by country. In the United Kingdom, the National Health Service (NHS) has approved semaglutide for obesity treatment through specialist weight management services, but the strict eligibility criteria and limited clinic capacity create waiting lists that can stretch to months or years. In Australia, tirzepatide received TGA approval for obesity in 2024, but PBS (pharmaceutical benefits scheme) listing, which determines whether the government subsidizes the medication's cost, follows a separate process that can take an additional 12-18 months. In many middle-income countries, GLP-1 agonists are available only at full international pricing, making them accessible only to wealthy patients or those with employer-provided insurance that covers international medications. The global access disparity mirrors the broader challenge of making effective medical treatments available to the populations that need them most. The emergence of biosimilar GLP-1 products represents a potential long-term solution to the cost and access challenges. Semaglutide's core patent expires in the late 2020s in most jurisdictions, and several biosimilar manufacturers have announced development programs. Unlike generic small-molecule drugs, biosimilar biologics require their own clinical trials (though abbreviated compared to the original), and the regulatory pathway for biosimilar peptides is still being refined by regulatory agencies. The first semaglutide biosimilars may reach the market by 2028-2030, potentially reducing costs by 30-50% based on historical biosimilar pricing patterns. Until then, the combination of branded products, compounded formulations, and emerging oral alternatives provides the range of options that patients and clinicians must work within. The evolving field of cost, coverage, and access underscores the importance of working with knowledgeable healthcare providers who can help patients navigate the available options. The FormBlends free assessment provides guidance on treatment access alongside clinical recommendations, and the GLP-1 Research Hub maintains current information on insurance coverage trends, compounding pharmacy availability, and policy developments that affect patient access to anti-obesity medications.
Frequently Asked Questions
What is the most effective FDA-approved weight loss medication currently available?
As of early 2026, tirzepatide (Zepbound) is the most effective FDA-approved anti-obesity medication. In the SURMOUNT-1 clinical trial, the 15mg dose produced an average weight loss of 20.9% over 72 weeks. Semaglutide 2.4mg (Wegovy) is the second most effective, achieving approximately 14.9% weight loss in the STEP 1 trial. Both require weekly subcutaneous injections. For patients preferring oral medication, the oral semaglutide 25mg pill (approved December 2025) produces roughly 13-15% weight loss, making it the most effective oral option among approved drugs.
How do GLP-1 receptor agonists work for weight loss?
GLP-1 receptor agonists mimic the action of glucagon-like peptide-1, a hormone naturally released by the gut after eating. They reduce appetite by activating satiety centers in the hypothalamus and brainstem, slow gastric emptying so you feel full longer, and appear to reduce food cravings by modulating reward pathways in the brain. They also enhance glucose-dependent insulin secretion, which is why they're effective for both weight loss and type 2 diabetes management. The weight loss effect is primarily driven by reduced caloric intake, not increased metabolism.
What new weight loss drugs are expected to be approved in 2026-2027?
Several promising candidates are advancing toward regulatory approval. Orforglipron, an oral small-molecule GLP-1 agonist from Eli Lilly, has completed Phase 3 trials and may receive approval as early as 2026. CagriSema (cagrilintide plus semaglutide) from Novo Nordisk has completed Phase 3 REDEFINE trials. Retatrutide, a triple GLP-1/GIP/glucagon agonist, is in Phase 3 trials with data expected through 2026-2027. Amycretin, an oral GLP-1/amylin agonist showing approximately 22% weight loss, is entering Phase 3 studies.
What is the difference between semaglutide and tirzepatide?
Semaglutide (Wegovy/Ozempic) activates only the GLP-1 receptor, while tirzepatide (Zepbound/Mounjaro) activates both GLP-1 and GIP receptors simultaneously. This dual mechanism gives tirzepatide an efficacy advantage, producing approximately 21% weight loss versus 15% for semaglutide. Both are administered as weekly subcutaneous injections. Semaglutide has stronger cardiovascular outcomes data (SELECT trial showing 20% MACE reduction), while tirzepatide has an additional indication for obstructive sleep apnea. Cost is comparable, though Zepbound was priced slightly lower at launch.
Do you gain weight back when you stop taking weight loss medications?
Yes, weight regain after discontinuing anti-obesity medications is well-documented. In the STEP 4 trial, patients switching from semaglutide to placebo regained about two-thirds of their lost weight over 48 weeks. Similar results were seen in SURMOUNT-4 with tirzepatide. This happens because obesity involves persistent biological changes (altered appetite hormones, reduced metabolic rate, increased hunger signals) that reassert themselves when medication is stopped. For this reason, most guidelines now recommend treating obesity as a chronic disease requiring long-term medication, similar to how hypertension or diabetes is managed.
What are the common side effects of GLP-1 weight loss drugs?
The most common side effects are gastrointestinal: nausea (30-44% of patients), diarrhea (23-30%), vomiting (12-24%), constipation (11-24%), and abdominal pain. These effects typically peak during dose escalation and improve over 4-8 weeks at a stable dose. Slow dose titration reduces severity. Less common but important side effects include gallbladder disease (increased with rapid weight loss), pancreatitis (rare), and injection site reactions. About 5-10% of patients discontinue treatment due to persistent GI effects.
How much do weight loss medications cost without insurance?
At list prices, semaglutide (Wegovy) costs approximately $1,300-$1,500 per month and tirzepatide (Zepbound) costs $1,000-$1,100 per month. Older medications are less expensive: phentermine-topiramate (Qsymia) generic runs $50-$150/month, naltrexone-bupropion (Contrave) $300-$500/month, and generic liraglutide (available since August 2025) approximately $400-$600/month. Over-the-counter orlistat (Alli) costs $40-$60/month. Manufacturer savings programs and insurance coverage can significantly reduce out-of-pocket costs for branded products.
What is retatrutide and why is it generating so much interest?
Retatrutide is the first triple hormone receptor agonist, targeting GLP-1, GIP, and glucagon receptors simultaneously. It has generated excitement because its Phase 3 TRIUMPH-4 trial showed 28.7% average body weight loss at 68 weeks with the 12mg dose, the highest reported for any anti-obesity medication. Nearly 40% of participants lost 30% or more of their body weight. The glucagon receptor component adds energy expenditure effects that go beyond appetite suppression alone. However, it also showed higher rates of side effects, including a new signal of dysesthesia (abnormal skin sensations) in up to 21% of patients.
Can weight loss medications replace bariatric surgery?
Not entirely, but the gap is narrowing. Bariatric surgery (gastric bypass, sleeve gastrectomy) typically produces 25-35% sustained weight loss and has decades of long-term safety and efficacy data. Retatrutide's 28.7% weight loss now overlaps with the lower end of surgical outcomes. However, surgery provides durable effects without ongoing medication costs, while pharmacotherapy requires continued use to maintain results. The choice depends on individual factors including BMI severity, comorbidities, surgical risk, patient preference, and access. Many experts envision a future where pharmacotherapy is first-line for most patients, with surgery reserved for those who don't respond adequately or prefer a one-time intervention.
Is there a weight loss medication that preserves muscle mass?
Bimagrumab is the most promising candidate for muscle preservation during weight loss. This monoclonal antibody blocks activin type II receptors, promoting muscle growth while enhancing fat loss. In the Phase 2b BELIEVE trial, bimagrumab combined with semaglutide produced 22.1% weight loss, with 92.8% coming from fat mass and actual preservation of lean body mass. When used alone, 100% of bimagrumab's weight loss came from fat, with a 2.5% increase in lean mass. It's not yet approved but is expected to advance to Phase 3. In the meantime, resistance exercise and adequate protein intake (1.2-1.6g/kg per day) help preserve muscle during pharmacological weight loss.
Are there oral alternatives to injectable GLP-1 medications?
Yes. In December 2025, the FDA approved oral semaglutide 25mg (Wegovy pill) for weight management, making it the first oral GLP-1 for obesity. Orforglipron, a non-peptide oral GLP-1 from Eli Lilly, showed 11.2% weight loss in Phase 3 and is expected to seek approval in 2026. Amycretin (oral GLP-1/amylin agonist) showed approximately 22% weight loss in early studies. Older oral options include phentermine-topiramate (Qsymia) and naltrexone-bupropion (Contrave), which work through different mechanisms and provide more modest weight loss.
How do non-GLP-1 approaches like tesofensine and 5-amino-1MQ compare?
Tesofensine is a triple monoamine reuptake inhibitor that showed 10.6% weight loss at the 1.0mg dose in Phase 2 trials. It works by suppressing appetite through dopamine, serotonin, and norepinephrine pathways rather than gut hormone signaling. 5-Amino-1MQ inhibits the enzyme NNMT, which in preclinical studies reduced body mass by 35% in obese mice without changing food intake. However, neither is FDA-approved, and 5-amino-1MQ lacks human clinical trial data. These compounds target different pathways than GLP-1 agonists and could theoretically complement them, but combination data don't exist yet.
What is CagriSema and how effective is it?
CagriSema is a combination of cagrilintide (a long-acting amylin analogue) and semaglutide, developed by Novo Nordisk. The REDEFINE 1 Phase 3 trial showed 20.4% weight loss at 68 weeks, with 60% of participants losing at least 20% of body weight. This exceeded semaglutide alone (14.9%) but was roughly comparable to tirzepatide (20.9%), which was somewhat disappointing for Novo Nordisk. The combination targets both GLP-1 and amylin receptors, providing complementary satiety signals. In patients with type 2 diabetes (REDEFINE 2), it achieved 13.7% weight loss and significant HbA1c improvement.
How long do you need to take weight loss medications?
Current evidence and guidelines support long-term, potentially indefinite use. Obesity is a chronic condition with underlying biological drivers (hormonal, neurological, genetic) that persist even after weight loss. Clinical trials consistently show that weight rebounds when medication is stopped. The American Association of Clinical Endocrinology, the Obesity Medicine Association, and other professional bodies recommend continued pharmacotherapy as long as the patient is benefiting and tolerating the medication. Periodic reassessment of risk-benefit is appropriate, but "treatment courses" with planned discontinuation generally lead to weight regain.
References & Clinical Sources
- Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384(11):989-1002. doi:10.1056/NEJMoa2032183
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387(3):205-216. doi:10.1056/NEJMoa2206038
- Jastreboff AM, Kaplan LM, Frias JP, et al. Triple-hormone-receptor agonist retatrutide for obesity - a phase 2 trial. N Engl J Med. 2023;389(6):514-526. doi:10.1056/NEJMoa2301972
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med. 2023;389(24):2221-2232. doi:10.1056/NEJMoa2307563
- Frias JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med. 2021;385(6):503-515. doi:10.1056/NEJMoa2107519
- Pi-Sunyer X, Astrup A, Fujioka K, et al. A randomized, controlled trial of 3.0 mg of liraglutide in weight management. N Engl J Med. 2015;373(1):11-22. doi:10.1056/NEJMoa1411892
- Greenway FL, Fujioka K, Plodkowski RA, et al. Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR-I). Lancet. 2010;376(9741):595-605. doi:10.1016/S0140-6736(10)60888-4
- Gadde KM, Allison DB, Ryan DH, et al. Effects of low-dose, controlled-release, phentermine plus topiramate combination on weight and associated comorbidities in overweight and obese adults (CONQUER). Lancet. 2011;377(9774):1341-1352. doi:10.1016/S0140-6736(11)60205-5
- Torgerson JS, Hauptman J, Boldrin MN, Sjostrom L. XENical in the prevention of diabetes in obese subjects (XENDOS) study. Diabetes Care. 2004;27(1):155-161. doi:10.2337/diacare.27.1.155
- Connolly HM, Crary JL, McGoon MD, et al. Valvular heart disease associated with fenfluramine-phentermine. N Engl J Med. 1997;337(24):581-588. doi:10.1056/NEJM199711133372006
- James WPT, Caterson ID, Coutinho W, et al. Effect of sibutramine on cardiovascular outcomes in overweight and obese subjects. N Engl J Med. 2010;363(10):905-917. doi:10.1056/NEJMoa1003114
- Bohula EA, Wiviott SD, McGuire DK, et al. Cardiovascular safety of lorcaserin in overweight or obese patients. N Engl J Med. 2018;379(12):1107-1117. doi:10.1056/NEJMoa1909510
- Global BMI Mortality Collaboration. Body-mass index and all-cause mortality: individual-participant-data meta-analysis of 239 prospective studies. Lancet. 2016;388(10046):776-786. doi:10.1016/S0140-6736(16)30175-1
- Ward ZJ, Bleich SN, Long MW, et al. Projected U.S. state-level prevalence of adult obesity and severe obesity. Lancet. 2024. doi:10.1016/S0140-6736(24)01548-4
- Cawley J, Biener A, Meyerhoefer C, et al. The economic burden of obesity in 2024: a cost analysis. Critical Public Health. 2024. doi:10.1080/09581596.2024.2333407
- Ogden CL, Fakhouri TH, Carroll MD, et al. Prevalence of obesity among adults and youth: United States, 2021-2023. NCHS Data Brief. 2024;508. doi:10.15620/cdc/148908
- Neelakantan H, Wang HY, Vance V, et al. Selective and membrane-permeable small molecule inhibitors of nicotinamide N-methyltransferase reverse high fat diet-induced obesity in mice. Biochem Pharmacol. 2018;147:141-152. doi:10.1016/j.bmc.2018.01.020
- Astrup A, Madsbad S, Breum L, et al. Effect of tesofensine on bodyweight loss, body composition, and quality of life in obese patients. Lancet. 2008;372(9653):1906-1913. doi:10.1016/S0140-6736(08)61525-1
- Bhatt DK, et al. Tesofensine, a novel antiobesity drug, silences GABAergic hypothalamic neurons. PLOS One. 2024;19(4):e0300544. doi:10.1371/journal.pone.0300544
- Coadministered cagrilintide and semaglutide in adults with overweight or obesity. N Engl J Med. 2025. doi:10.1056/NEJMoa2502081
- Cagrilintide-semaglutide in adults with overweight or obesity and type 2 diabetes. N Engl J Med. 2025. doi:10.1056/NEJMoa2502082
- Orforglipron, an oral small-molecule GLP-1 receptor agonist for obesity treatment. N Engl J Med. 2025. doi:10.1056/NEJMoa2511774
- Rubino D, Abrahamsson N, Davies M, et al. Effect of continued weekly subcutaneous semaglutide vs placebo on weight loss maintenance in adults with overweight or obesity: the STEP 4 randomized clinical trial. JAMA. 2021;325(14):1414-1425. doi:10.1001/jama.2021.3224
- Aronne LJ, Sattar N, Horn DB, et al. Continued treatment with tirzepatide for maintenance of weight reduction in adults with obesity: the SURMOUNT-4 randomized clinical trial. JAMA. 2024;331(1):38-48. doi:10.1001/jama.2023.24945
- Garvey WT, Batterham RL, Bhatt DL, et al. Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nat Med. 2022;28:2083-2091. doi:10.1038/s41591-022-02026-4
- Wadden TA, Bailey TS, Billings LK, et al. Effect of subcutaneous semaglutide vs placebo as an adjunct to intensive behavioral therapy on body weight in adults with overweight or obesity: the STEP 3 randomized clinical trial. JAMA. 2021;325(14):1403-1413. doi:10.1001/jama.2021.1831
- Davies M, Frias JP, Jastreboff AM, et al. Semaglutide 2.4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2). Lancet. 2021;397(10278):971-984. doi:10.1016/S0140-6736(21)00213-0
- Rubino DM, Greenway FL, Khalid U, et al. Effect of weekly subcutaneous semaglutide vs daily liraglutide on body weight in adults with overweight or obesity without diabetes: the STEP 8 randomized clinical trial. JAMA. 2022;327(2):138-150. doi:10.1001/jama.2021.23619
- Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375(19):1834-1844. doi:10.1056/NEJMoa1607141
- Malhotra A, Grunstein RR, Gao W, et al. Tirzepatide for the treatment of obstructive sleep apnea and obesity. N Engl J Med. 2024;391:1919-1930. doi:10.1056/NEJMoa2404881
- Jastreboff AM, Auerbach P, Engstrom T, et al. Efficacy and safety of retatrutide in adults with obesity or overweight and knee osteoarthritis: TRIUMPH-4 phase 3 trial. 2025. Eli Lilly press release. Accessed January 2026.
- Heymsfield SB, Coleman LA, Miller R, et al. Effect of bimagrumab vs placebo on body fat mass among adults with type 2 diabetes and obesity: a phase 2 randomized clinical trial. JAMA Netw Open. 2021;4(1):e2033457. doi:10.1001/jamanetworkopen.2020.33457
- Nauck MA, Quast DR, Wefers J, Meier JJ. GLP-1 receptor agonists in the treatment of type 2 diabetes - state-of-the-art. Mol Metab. 2021;46:101102. doi:10.1016/j.molmet.2020.101102
- Muller TD, Bluher M, Tschop MH, DiMarchi RD. Anti-obesity drug discovery: advances and challenges. Nat Rev Drug Discov. 2022;21(3):201-223. doi:10.1038/s41573-021-00337-8
- World Health Organization. Obesity and overweight fact sheet. Updated March 2024. Accessed January 2026.
- World Obesity Federation. Economic impact of overweight and obesity to surpass $4 trillion by 2035. Published March 2023. Accessed January 2026.
- Apovian CM, Aronne LJ, Bessesen DH, et al. Pharmacological management of obesity: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2015;100(2):342-362. doi:10.1210/jc.2014-3415
- Yanovski SZ, Yanovski JA. Long-term drug treatment for obesity: a systematic and clinical review. JAMA. 2014;311(1):74-86. doi:10.1001/jama.2013.281361
- Tak YJ, Lee SY. Anti-obesity drugs: long-term efficacy and safety - an updated review. World J Mens Health. 2021;39(2):208-221. doi:10.5534/wjmh.200010
- Khera R, Murad MH, Chandar AK, et al. Association of pharmacological treatments for obesity with weight loss and adverse events: a systematic review and meta-analysis. JAMA. 2016;315(22):2424-2434. doi:10.1001/jama.2016.7602