Executive Summary
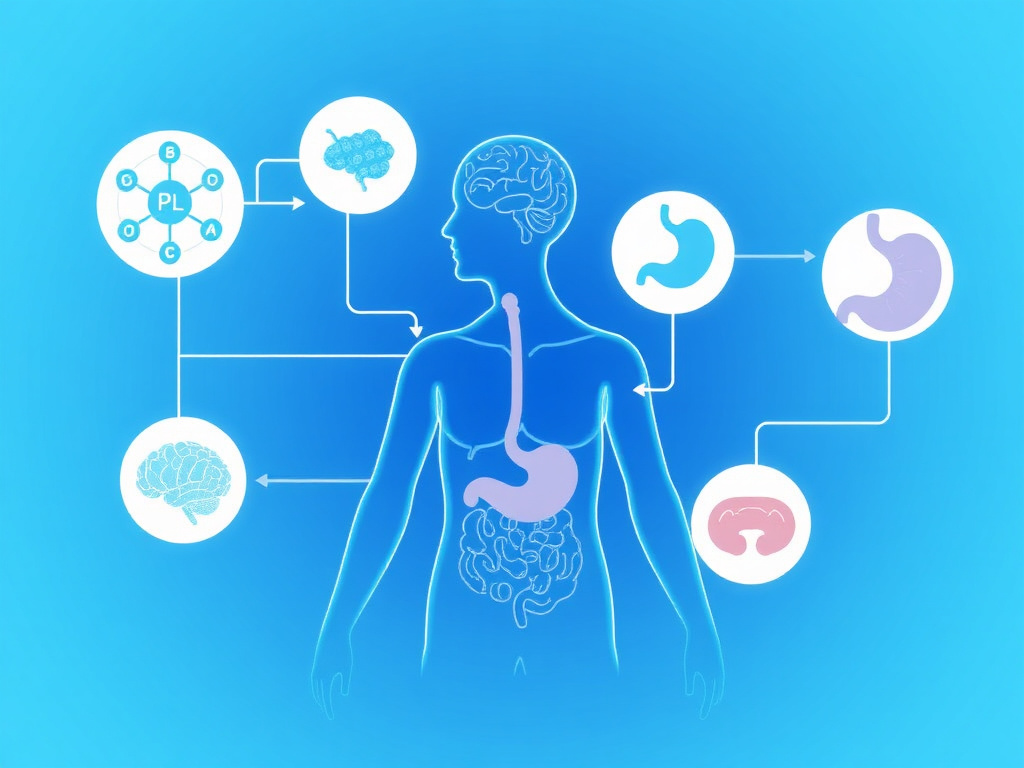
Figure 1: GLP-1 receptor agonists produce weight loss through at least five distinct but overlapping biological pathways.
Key Takeaways
- Central appetite suppression: GLP-1R activation in brainstem and hypothalamus reduces hunger drive
- Delayed gastric emptying: Food stays in the stomach longer, creating early and prolonged satiety
- Reduced food reward: Mesolimbic dopamine modulation decreases cravings for palatable foods
- Improved metabolic signaling: Enhanced insulin secretion, glucagon suppression, improved lipid handling
- Energy expenditure effects: Possible increases in thermogenesis, though data remain mixed
GLP-1 receptor agonists don't cause weight loss through a single trick. They work across at least five interconnected biological systems - from the brainstem to the gut wall to the dopamine-rich reward centers that make you crave a second slice of pizza. Understanding these mechanisms isn't just academic; it explains why these drugs outperform everything that came before them, and it points to strategies for maximizing results while preserving muscle mass.
When you inject semaglutide, tirzepatide, or liraglutide, you're activating the GLP-1 receptor, a protein found on cells throughout your body. The native GLP-1 hormone is produced by L-cells in your small intestine after meals, but it breaks down within two to three minutes. Pharmaceutical GLP-1 receptor agonists are engineered to resist degradation by the enzyme dipeptidyl peptidase-4 (DPP-4), extending their half-life from minutes to days or even weeks. Semaglutide, for example, has a half-life of roughly 165 hours, meaning a single weekly injection maintains continuous receptor activation.
That continuous activation sets off a cascade of effects. In the central nervous system, GLP-1 receptor agonists cross the blood-brain barrier and bind to receptors in the nucleus tractus solitarius (NTS), area postrema, and hypothalamus. These brain regions serve as the body's hunger command center. Activation of GLP-1 receptors here reduces appetite through both homeostatic pathways (the calorie-counting system) and hedonic pathways (the pleasure-seeking system). Patients consistently report that food simply becomes less interesting, that cravings diminish, and that they feel satisfied with smaller portions.
Below the neck, GLP-1 receptor agonists slow gastric emptying, meaning food stays in the stomach longer. This creates a physical sensation of fullness that reinforces the central appetite suppression. They also enhance glucose-dependent insulin secretion from pancreatic beta cells and suppress glucagon release from alpha cells, improving metabolic efficiency and reducing the glucose spikes that can trigger rebound hunger.
Perhaps the most fascinating mechanism involves the brain's reward circuitry. GLP-1 receptors exist on neurons in the ventral tegmental area (VTA) and nucleus accumbens, the same pathways implicated in addiction. Activation of these receptors appears to reduce the rewarding properties of food, particularly highly palatable, calorie-dense foods. This helps explain why patients on GLP-1 therapy often report reduced cravings for sweets, fried food, and alcohol, as well as why early research is investigating these drugs for substance use disorders.
The clinical results reflect this multi-pathway approach. In the STEP 1 trial, semaglutide 2.4 mg produced an average weight loss of 14.9% over 68 weeks. Tirzepatide, which activates both GLP-1 and GIP receptors, achieved up to 22.5% weight loss in the SURMOUNT-1 trial at the highest dose. And retatrutide, a triple agonist hitting GLP-1, GIP, and glucagon receptors, showed up to 24.2% weight loss in phase 2 data. Each new generation builds on additional receptor targets, but GLP-1 receptor activation remains the foundational mechanism.
This report breaks down every known mechanism in detail. We'll walk through the neuroscience of appetite regulation, the gut physiology of delayed gastric emptying, the metabolic effects on insulin and lipid metabolism, and the clinical data on body composition. We'll also address the practical concern that matters most to many patients: how to preserve lean muscle mass during treatment. For a broader overview of the GLP-1 drug class, see our GLP-1 class overview.
Key Mechanisms at a Glance
- Central appetite suppression: GLP-1R activation in brainstem and hypothalamus reduces hunger drive
- Delayed gastric emptying: Food stays in the stomach longer, creating early and prolonged satiety
- Reduced food reward: Mesolimbic dopamine modulation decreases cravings for palatable foods
- Improved metabolic signaling: Enhanced insulin secretion, glucagon suppression, improved lipid handling
- Energy expenditure effects: Possible increases in thermogenesis, though data remain mixed
The weight loss produced by GLP-1 receptor agonists is not evenly distributed between fat and muscle. Clinical data suggest that roughly 25-40% of total weight lost may come from lean mass, a proportion similar to diet-induced weight loss but concerning given the absolute magnitude of weight reduction these drugs produce. Strategies to mitigate muscle loss, including resistance training and adequate protein intake, are essential companions to pharmacotherapy. We cover these in depth in the muscle preservation section.
Weight regain after discontinuation is another practical reality. The STEP 4 trial demonstrated that participants who stopped semaglutide after 20 weeks regained roughly two-thirds of their lost weight over the subsequent 48 weeks. This underscores that GLP-1 receptor agonists treat obesity as a chronic condition, much like statins treat high cholesterol. The biological drives toward weight regain don't disappear when the drug is withdrawn. We address maintenance strategies and the emerging concept of dose reduction (rather than full discontinuation) in section 9.
If you're considering GLP-1 therapy, our free assessment can help determine which compound and dose might be appropriate. For dosing specifics, the dosing calculator provides evidence-based titration schedules.
Central Nervous System Signaling
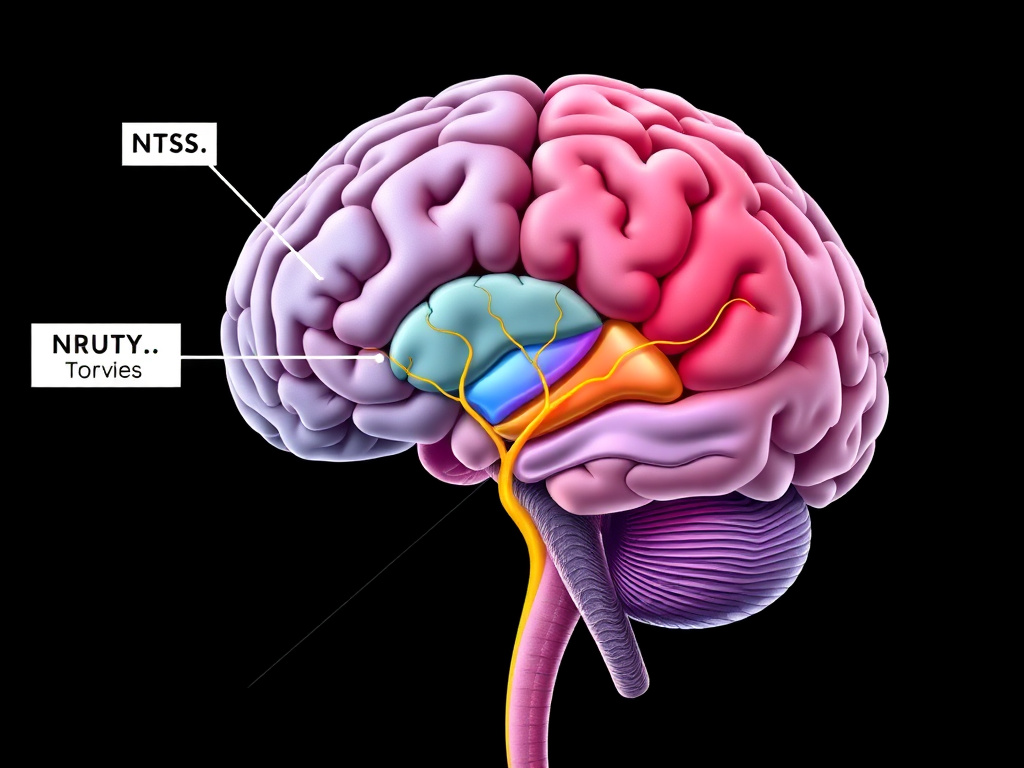
Figure 2: GLP-1 receptor agonists access the brainstem through multiple routes, including vagal afferents and direct blood-brain barrier penetration at circumventricular organs.
The brain is where the weight loss story really begins. GLP-1 receptors are expressed throughout the central nervous system, with particularly dense concentrations in brainstem nuclei that serve as the first relay station for satiety signals traveling from the gut to the brain. Understanding this neurocircuitry explains why GLP-1 drugs produce such consistent and powerful appetite reduction.
The Brainstem: First Point of Contact
The nucleus tractus solitarius (NTS), located in the dorsal medulla of the brainstem, is the primary entry point for visceral sensory information reaching the brain. Vagal afferent fibers from the gastrointestinal tract terminate here, carrying information about stomach distension, nutrient content, and local hormone release. The NTS contains a high density of GLP-1 receptors, and a distinct population of NTS neurons actually produces GLP-1 by expressing preproglucagon (PPG). These PPG neurons are separate from the gut-derived GLP-1 system and constitute a dedicated central GLP-1 signaling network.
When pharmaceutical GLP-1 receptor agonists reach the NTS, they activate neurons that project to multiple higher brain regions involved in appetite regulation. NTS GLP-1 neurons send axons to the hypothalamic paraventricular nucleus (PVN), the lateral hypothalamus, the bed nucleus of the stria terminalis, and the ventral tegmental area. Each of these projection targets mediates a different aspect of feeding behavior - from homeostatic calorie balance to stress-related eating to food reward.
Studies using c-Fos immunohistochemistry (a marker of neuronal activation) have shown that peripheral administration of GLP-1 receptor agonists produces strong activation of NTS neurons within hours. Lesion studies in rodents confirm that destroying NTS GLP-1 receptor-expressing neurons attenuates the anorectic effects of both endogenous GLP-1 and exogenous agonists. The NTS is not just a passive relay; it's an active integration center where peripheral signals are processed and amplified before being transmitted to higher circuits.
The Area Postrema: A Window Without a Wall
Adjacent to the NTS sits the area postrema (AP), one of the brain's circumventricular organs. Unlike most brain regions, the AP lacks a complete blood-brain barrier. Blood vessels here are fenestrated, meaning circulating molecules - including injected GLP-1 receptor agonists - can directly access neurons without needing specialized transport mechanisms. This anatomical feature makes the AP a critical site for the detection of blood-borne signals, including toxins, hormones, and drugs.
GLP-1 receptors in the AP mediate some of the drug class's most recognizable effects, both therapeutic and adverse. AP activation contributes to nausea, which is the most common side effect during GLP-1 therapy initiation and dose escalation. But this same activation contributes to appetite suppression. The AP projects heavily to the NTS and to the lateral parabrachial nucleus (lPBN), a region involved in taste aversion and meal termination. Activation of AP-to-lPBN projections generates a visceral sense that eating should stop, a sensation patients often describe as simply not wanting more food rather than feeling physically full.
The clinical relevance of AP signaling is visible in the dose-titration protocols used for all GLP-1 receptor agonists. Slow dose escalation allows partial desensitization of AP neurons, reducing nausea while maintaining the appetite-suppressive effects mediated by other brain regions. This differential desensitization explains why nausea typically fades within weeks while appetite suppression persists for as long as the drug is continued.
Vagal Afferent Pathways
The vagus nerve provides the most direct communication line between the gut and the brain. Its afferent (sensory) fibers carry information about meal-related events - stomach stretch, nutrient sensing, and local hormone release - from the GI tract to the brainstem. GLP-1 receptors are expressed on vagal afferent neuron cell bodies in the nodose ganglion, and activation of these receptors enhances the transmission of satiety signals.
Native GLP-1 released from intestinal L-cells likely acts primarily through this vagal route. The hormone's half-life is so short (about 2 minutes) that circulating levels may not reach the brain in meaningful concentrations. Instead, GLP-1 activates receptors on nearby vagal afferent terminals in the intestinal wall, and the resulting nerve impulses travel to the NTS. This paracrine signaling pathway was demonstrated in studies showing that vagotomy (cutting the vagus nerve) partially blocks the acute satiety effects of endogenous GLP-1 in animal models.
Pharmaceutical GLP-1 receptor agonists, with their extended half-lives, use a different strategy. They reach sufficient circulating concentrations to directly activate both vagal afferents and central receptors. Semaglutide's fatty acid modification allows it to bind albumin in the blood, creating a circulating reservoir that provides sustained receptor occupancy. This dual mechanism, both vagal and direct central, likely explains why long-acting agonists produce stronger appetite suppression than the body's own short-lived GLP-1.
Blood-Brain Barrier Penetration
Whether GLP-1 receptor agonists fully cross the blood-brain barrier has been debated, but evidence now confirms that at least some of these molecules access deep brain structures. Fluorescently labeled semaglutide has been detected in the arcuate nucleus of the hypothalamus, a region protected by the blood-brain barrier, after peripheral administration in mice. The mechanism likely involves transport via tanycytes, specialized glial cells lining the third ventricle that can shuttle molecules from the blood into the hypothalamic parenchyma.
Tanycytes express GLP-1 receptors and may actively transport GLP-1 receptor agonists across the barrier. This is significant because hypothalamic nuclei contain the body's most precise calorie-sensing machinery. Direct access to these neurons means that GLP-1 drugs can modulate appetite at its most fundamental level, not just through indirect vagal or brainstem signaling but by acting on the very neurons that calculate energy balance.
The albumin-binding properties of semaglutide and other long-acting agonists also facilitate this process. Albumin itself uses receptor-mediated transcytosis to cross the blood-brain barrier, and GLP-1 agonists bound to albumin may hitchhike on this transport system. This pharmacokinetic feature likely contributes to semaglutide's particularly strong weight loss effects compared to earlier GLP-1 agonists with less brain penetration.
Central GLP-1 Production: The Endogenous Brain System
Beyond the signals arriving from the periphery, the brain produces its own GLP-1. A small cluster of neurons in the caudal NTS expresses preproglucagon and processes it into GLP-1 using the enzyme prohormone convertase 1/3. These neurons number only about 200-300 in rats (estimated to be several thousand in humans), yet they project widely throughout the brain, including to the hypothalamus, amygdala, and midbrain reward areas.
Central GLP-1 neurons respond to visceral signals arriving via the vagus nerve, to circulating satiety hormones like cholecystokinin (CCK) and leptin, and to stress-related inputs from the amygdala. They serve as integrators, combining multiple streams of information about the body's nutritional and emotional state into a single output signal that modulates feeding behavior. Activation of these neurons suppresses food intake; their inhibition or destruction increases it.
Pharmaceutical GLP-1 agonists likely amplify the actions of this endogenous system. By providing sustained receptor activation at the same targets where central GLP-1 neurons project, these drugs create a persistent satiety signal that overrides the meal-to-meal fluctuations of normal GLP-1 release. This helps explain the continuous appetite suppression patients experience, even between meals and overnight, which differs from the temporary fullness produced by a large meal activating endogenous GLP-1.
Clinical Relevance
The multi-site central signaling of GLP-1 receptor agonists explains several clinical observations: (1) appetite suppression is more consistent and powerful than with any previous obesity drug; (2) nausea during initiation fades while anorexia persists, reflecting differential adaptation in different brain regions; (3) patients report qualitative changes in their relationship with food, not just reduced hunger, suggesting engagement of higher cognitive and reward circuits beyond simple brainstem reflexes.
Downstream Neurotransmitter Effects
GLP-1 receptor activation in the brain doesn't just suppress one signal. It reshapes the neurochemical environment governing appetite regulation. In the NTS and hypothalamus, GLP-1 signaling enhances the release of brain-derived neurotrophic factor (BDNF), a molecule involved in synaptic plasticity and energy homeostasis. BDNF signaling through its TrkB receptor suppresses food intake independently of GLP-1, creating an additive effect.
GLP-1 receptor activation also modulates serotonergic transmission. Serotonin (5-HT) neurons in the dorsal raphe project to the hypothalamus and influence feeding behavior, and GLP-1 enhances serotonin release at these synapses. This interaction may contribute to the mood-stabilizing effects some patients report on GLP-1 therapy, and it provides a mechanistic link to earlier obesity drugs like fenfluramine that worked primarily through serotonin (but with unacceptable cardiovascular side effects).
GABAergic and glutamatergic transmission in the hypothalamus are also affected. GLP-1 receptor activation reduces GABA release onto anorexigenic POMC neurons (disinhibiting them) and enhances glutamate signaling onto the same cells (exciting them). This dual modulation pushes the hypothalamic circuit toward appetite suppression through multiple synaptic mechanisms simultaneously. We'll explore the specific hypothalamic circuitry in the next section.
For more detailed information on how specific GLP-1 compounds differ in their CNS effects, see our semaglutide guide and our tirzepatide guide.
Hypothalamic Appetite Regulation
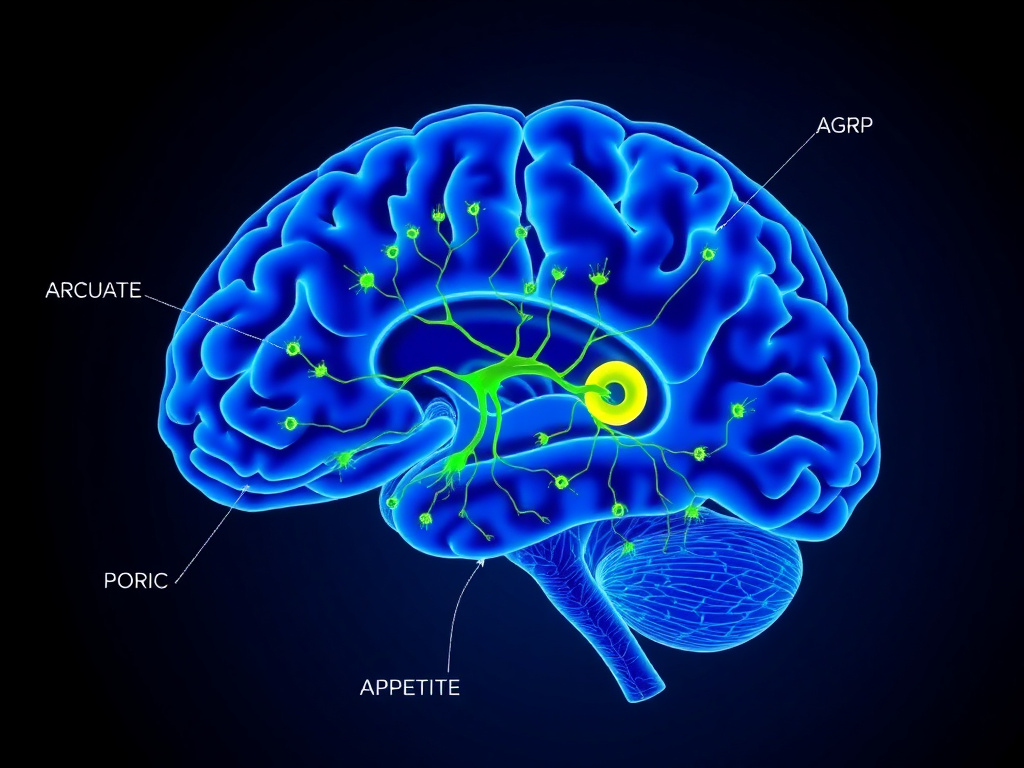
Figure 3: The arcuate nucleus contains two opposing neuronal populations - POMC/CART (anorexigenic) and NPY/AgRP (orexigenic) - that together determine hunger drive. GLP-1 receptor agonists shift this balance toward appetite suppression.
The hypothalamus is the brain's metabolic thermostat. Tucked at the base of the brain, surrounding the third ventricle, it contains specialized neuronal circuits that monitor energy stores, sense circulating nutrients and hormones, and generate the drive to eat or stop eating. GLP-1 receptor agonists act on multiple nodes within this circuit, producing a coordinated shift toward reduced food intake that persists for as long as treatment continues.
The Arcuate Nucleus: Where Hunger is Calculated
The arcuate nucleus (ARC) sits at the base of the hypothalamus, adjacent to the median eminence, another circumventricular organ with a partially permeable blood-brain barrier. This location gives ARC neurons privileged access to circulating metabolic signals - glucose, insulin, leptin, ghrelin, and pharmaceutical GLP-1 receptor agonists among them.
Two genetically distinct neuron populations in the ARC play opposing roles in appetite regulation. The first population expresses pro-opiomelanocortin (POMC) and cocaine- and amphetamine-regulated transcript (CART). These POMC/CART neurons are anorexigenic, meaning their activation suppresses appetite. When POMC is cleaved by prohormone convertases, it produces alpha-melanocyte-stimulating hormone (alpha-MSH), which binds to melanocortin 4 receptors (MC4R) on downstream neurons in the paraventricular nucleus. MC4R activation is one of the most powerful appetite-suppressing signals in the brain. Humans with loss-of-function MC4R mutations develop severe early-onset obesity, confirming the pathway's critical importance.
The second population expresses neuropeptide Y (NPY) and agouti-related peptide (AgRP). These NPY/AgRP neurons are orexigenic - their activation drives hunger and food-seeking behavior. AgRP acts as an inverse agonist at MC4R, blocking the appetite-suppressing effects of alpha-MSH. NPY, released from the same neurons, acts on Y1 and Y5 receptors to stimulate food intake through a separate pathway. When these neurons fire, you feel hungry. When they're silenced, hunger fades.
The two populations also directly inhibit each other. NPY/AgRP neurons send GABAergic projections to POMC neurons, suppressing their activity. POMC neurons, via beta-endorphin release, modulate AgRP neuron firing. This reciprocal inhibition creates a toggle switch: the ARC is either in a hunger-promoting or a satiety-promoting state, with the balance determined by incoming metabolic signals.
How GLP-1 Receptor Agonists Shift the Balance
GLP-1 receptors are expressed on both ARC neuronal populations, but the net effect of their activation strongly favors appetite suppression. Electrophysiological recordings in brain slices show that GLP-1 directly depolarizes POMC neurons, increasing their firing rate and alpha-MSH release. Simultaneously, GLP-1 receptor activation on presynaptic GABAergic terminals (from NPY/AgRP neurons) reduces GABA release onto POMC cells. The result is a double hit: POMC neurons are both directly excited and released from inhibition.
On the other side of the circuit, GLP-1 receptor activation hyperpolarizes NPY/AgRP neurons, reducing their firing rate and decreasing the release of both NPY and AgRP. With less AgRP available to block MC4R, and more alpha-MSH available to activate it, the melanocortin system swings decisively toward appetite suppression.
Studies using optogenetic and chemogenetic tools have provided causal evidence for these pathways. Selectively activating GLP-1 receptors on ARC neurons using designer receptors exclusively activated by designer drugs (DREADDs) reproduces the appetite-suppressive effects of systemic GLP-1 agonist administration. Conversely, deleting GLP-1 receptors specifically from ARC POMC neurons attenuates, though doesn't fully eliminate, the weight loss effects of semaglutide in mice.
The Paraventricular Nucleus: Integrating the Signal
The paraventricular nucleus (PVN) of the hypothalamus receives dense projections from both ARC neuron populations and from NTS GLP-1 neurons. It serves as a major integration center where appetite signals are combined with information about stress, circadian rhythms, and autonomic function. PVN neurons express MC4R and respond to alpha-MSH from ARC POMC neurons, but they also receive direct GLP-1 innervation from the brainstem.
GLP-1 receptor activation in the PVN suppresses food intake through at least two mechanisms. First, it enhances the sensitivity of MC4R-expressing neurons to alpha-MSH, amplifying the melanocortin appetite-suppression signal. Second, it activates PVN neurons that project to the brainstem and spinal cord, engaging autonomic outputs that slow gastric motility and increase energy expenditure through brown adipose tissue activation and sympathetic nervous system drive.
The PVN also produces corticotropin-releasing hormone (CRH) and oxytocin, both of which suppress food intake. GLP-1 receptor activation stimulates CRH release, which may contribute to the mild cortisol elevations sometimes observed during GLP-1 therapy. Oxytocin neurons in the PVN are also activated by GLP-1, and oxytocin itself reduces food intake when administered centrally. This PVN-mediated circuit represents yet another parallel pathway through which GLP-1 agonists reduce appetite.
The Lateral Hypothalamus: Appetite Drive and Arousal
While the ARC calculates energy balance and the PVN integrates metabolic signals with autonomic outputs, the lateral hypothalamus (LH) generates the motivational drive to eat. LH neurons produce two orexigenic neuropeptides: orexin (also called hypocretin) and melanin-concentrating hormone (MCH). These neurons project widely to the cerebral cortex, hippocampus, and reward centers, linking metabolic need to conscious desire for food and the behavioral activation required to obtain it.
GLP-1 receptors are expressed in the LH, and their activation inhibits orexin neuron firing. This likely contributes to the reduced food-seeking motivation patients report on GLP-1 therapy. It may also explain why some patients report mild fatigue or reduced motivation during the early weeks of treatment, as orexin neurons also regulate wakefulness and general arousal. However, this effect is typically transient, possibly because compensatory mechanisms maintain orexin signaling for non-food-related arousal while food-directed motivation remains suppressed.
MCH neurons in the LH are similarly inhibited by GLP-1 signaling. MCH promotes food intake, sleep, and energy conservation. Rodents lacking MCH are lean and hyperactive, while those overexpressing it become obese. By suppressing MCH neuron activity, GLP-1 receptor agonists remove another pro-feeding signal from the hypothalamic circuit.
Leptin and Insulin Resistance in the Hypothalamus
One reason obesity is so difficult to treat is that the hypothalamus becomes resistant to its own satiety signals. Leptin, produced by fat cells in proportion to body fat stores, normally activates POMC neurons and inhibits NPY/AgRP neurons, keeping body weight stable. But chronic overnutrition causes hypothalamic inflammation and endoplasmic reticulum stress, which impair leptin receptor signaling. The brain stops "hearing" the leptin signal, even as circulating levels rise. This hypothalamic leptin resistance is considered a core mechanism maintaining the obese state.
GLP-1 receptor agonists may partially overcome this resistance. Several lines of evidence suggest that GLP-1 signaling enhances leptin sensitivity in ARC neurons. In rodent models, chronic GLP-1 agonist treatment reduces hypothalamic inflammation markers (IL-6, TNF-alpha, NF-kB activation) and restores STAT3 phosphorylation downstream of the leptin receptor. STAT3 signaling is essential for leptin's appetite-suppressive effects, and its restoration could amplify the natural satiety system alongside the direct GLP-1 receptor-mediated effects.
Similarly, central insulin signaling, which also suppresses appetite through ARC POMC neuron activation, is impaired in obesity. GLP-1 receptor activation improves insulin signaling in hypothalamic neurons by enhancing PI3K/Akt pathway activation. This suggests that GLP-1 drugs don't just add a new appetite-suppressing signal; they also restore the brain's responsiveness to existing signals that obesity has muted.
Synaptic Plasticity and Long-Term Remodeling
Beyond acute neuronal activation, GLP-1 receptor agonists appear to induce structural changes in hypothalamic circuits. Chronic semaglutide treatment in rodents alters the ratio of excitatory to inhibitory synapses on POMC neurons, shifting the balance toward greater excitatory input. This synaptic remodeling occurs over weeks, matching the time course of the progressive appetite suppression observed clinically as patients titrate to maintenance doses.
GLP-1 also stimulates neurogenesis and neuronal survival in the hypothalamus, effects mediated through CREB (cAMP response element-binding protein) activation and BDNF upregulation. While the functional significance of hypothalamic neurogenesis in adult humans remains uncertain, these trophic effects may contribute to the sustained appetite suppression observed during long-term treatment and could have implications for neurodegenerative diseases, a separate area of active investigation.
Clinical Correlation
The hypothalamic mechanisms described above explain why GLP-1 receptor agonists produce dose-dependent weight loss that increases with treatment duration. As the dose escalates and hypothalamic remodeling progresses, the appetite-suppressive effect deepens. This matches clinical data from STEP 1, where weight loss continued to accrue through 68 weeks on semaglutide 2.4 mg, and from SURMOUNT-1, where tirzepatide at 15 mg produced progressively greater weight loss over 72 weeks. The hypothalamus is not merely being silenced; its circuitry is being reprogrammed toward a lower weight set point.
The Melanocortin System: The Final Common Pathway
Almost all appetite-regulating signals in the hypothalamus converge on the melanocortin system. POMC-derived alpha-MSH activating MC4R represents the most potent known endogenous appetite suppressant. Loss-of-function mutations in MC4R cause the most common genetic form of severe obesity, affecting roughly 5-6% of people with early-onset obesity. Conversely, MC4R gain-of-function variants are associated with lower BMI in population studies.
GLP-1 receptor agonists effectively amplify melanocortin signaling from multiple angles: increasing POMC neuron firing, increasing alpha-MSH release, decreasing AgRP (the endogenous MC4R antagonist), and enhancing MC4R-expressing neuron sensitivity. This multi-pronged amplification of a single downstream pathway may explain why GLP-1 drugs are effective across diverse patient populations, regardless of the specific upstream cause of their obesity.
There is an important exception. Patients with biallelic loss-of-function MC4R mutations show reduced response to GLP-1 receptor agonists, though they still lose some weight because GLP-1 also works through melanocortin-independent pathways (gastric emptying, reward circuits, metabolic effects). This observation provides a natural experiment confirming that MC4R signaling is a major, but not the sole, mediator of GLP-1-induced weight loss. For more on how different compounds use these pathways, visit the GLP-1 research hub.
Gastric Emptying & Gut Motility
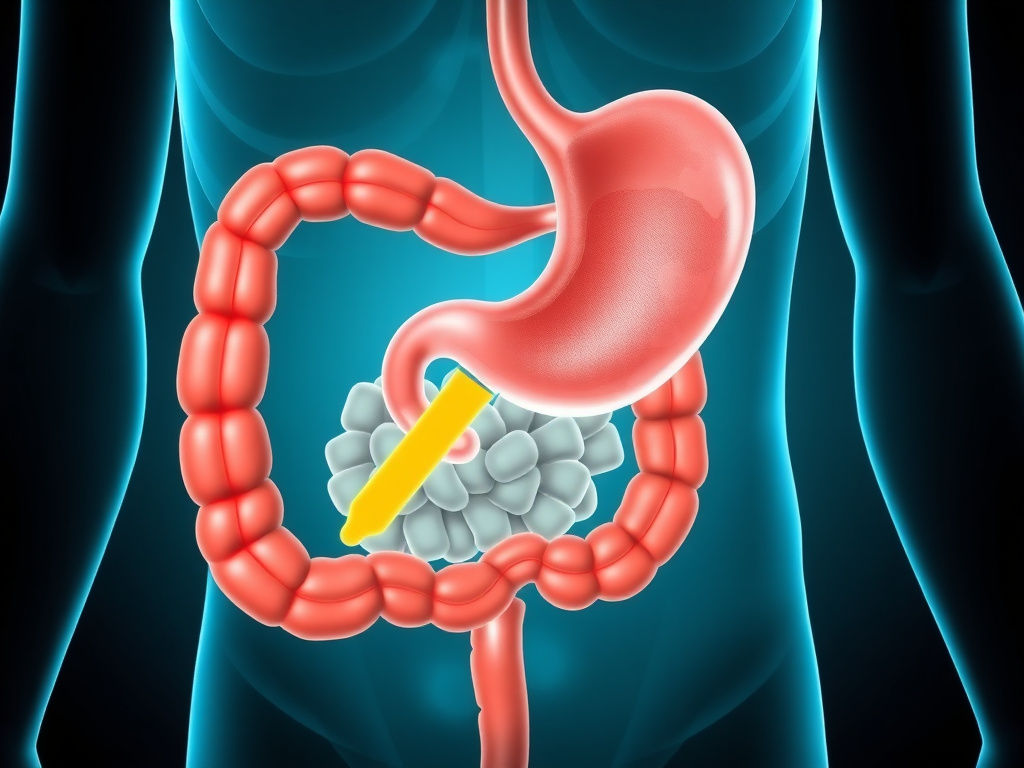
Figure 4: GLP-1 receptor agonists delay gastric emptying by 20-40%, prolonging the presence of food in the stomach and contributing to early satiety.
Ask someone on semaglutide why they eat less, and many will point to their stomach rather than their brain. The sensation of prolonged fullness after small meals is one of the most immediately noticeable effects of GLP-1 therapy. This isn't psychological. GLP-1 receptor agonists physically slow the rate at which the stomach empties its contents into the small intestine, a process with significant implications for satiety, glycemic control, and some of the GI side effects patients experience.
Normal Gastric Emptying Physiology
After a meal, the stomach serves as both a storage organ and a processing plant. Gastric peristalsis breaks food into smaller particles, mixes it with acid and pepsin, and gradually releases a controlled stream of semi-liquid chyme through the pyloric sphincter into the duodenum. The rate of gastric emptying is regulated by a network of neural and hormonal signals, including vagal tone, local enteric nervous system reflexes, and hormones released by the small intestine in response to arriving nutrients.
A typical mixed meal empties from the stomach in roughly 3-5 hours, though this varies with meal composition. Liquids empty faster than solids. Fats slow emptying more than proteins, which slow it more than carbohydrates. The duodenum provides negative feedback through the release of cholecystokinin (CCK), peptide YY (PYY), and GLP-1 itself, slowing gastric emptying when nutrients are arriving faster than the small intestine can absorb them. This feedback system, called the ileal brake, ensures efficient nutrient absorption and prevents overwhelming the intestinal absorptive capacity.
How GLP-1 Receptor Agonists Slow Gastric Emptying
GLP-1 receptors are expressed on vagal afferent neurons innervating the stomach, on neurons in the myenteric plexus of the gastric wall, and on smooth muscle cells in the gastric antrum and pylorus. Activation of these receptors produces three coordinated effects: relaxation of the gastric fundus (the upper stomach, which serves as the food reservoir), reduction of antral contractions (the peristaltic waves that push food toward the pylorus), and tonic contraction of the pyloric sphincter.
The net result is a marked slowing of gastric emptying. Studies using scintigraphy (a technique where patients eat meals labeled with a radioactive tracer) have shown that semaglutide delays gastric emptying of solid meals by approximately 20-40% during the first few hours after a meal. The half-time of gastric emptying, normally around 90-120 minutes for a mixed meal, extends to 140-180 minutes or longer in patients on GLP-1 therapy.
This delayed emptying has direct implications for satiety. Gastric distension activates mechanoreceptors in the stomach wall, which signal fullness to the brainstem via vagal afferents. When food stays in the stomach longer, this distension signal persists longer, reinforcing the central appetite suppression mediated by GLP-1 receptors in the brain. The peripheral and central mechanisms thus operate in concert: the brain reduces the desire to eat, while the stomach provides a physical reason not to.
Tachyphylaxis: Does the Gastric Effect Fade?
One of the more interesting aspects of GLP-1-mediated gastric slowing is that it shows tachyphylaxis, a fancy term for the fading of a drug effect with chronic exposure. Multiple studies have demonstrated that the degree of gastric emptying delay diminishes over weeks to months of continuous GLP-1 receptor agonist therapy. An initial 40% delay may attenuate to a 15-20% delay after 12-16 weeks.
This tachyphylaxis occurs because of desensitization of GLP-1 receptors on vagal afferents and enteric neurons. With sustained receptor activation, the downstream signaling cascade becomes less responsive. Receptor internalization (pulling receptors off the cell surface) and uncoupling from G-proteins both contribute. The stomach "adapts" to the drug's presence and partially restores its normal emptying rate.
Here's what makes this clinically relevant: weight loss continues even as gastric emptying partially normalizes. This proves that delayed gastric emptying, while contributing to early satiety (especially during the first weeks of therapy), is not the primary mechanism driving sustained weight loss. The central nervous system effects on appetite and food reward persist without significant tachyphylaxis, maintaining their appetite-suppressive effects over months and years of treatment.
The partial attenuation of gastric slowing is actually beneficial. Severe gastroparesis (extremely delayed gastric emptying) can cause nausea, vomiting, and nutritional deficiencies. The fact that the body partially compensates for GLP-1-induced gastric slowing helps prevent these extremes while allowing the more sustained central mechanisms to drive ongoing weight loss.
Implications for GI Side Effects
Most gastrointestinal side effects of GLP-1 receptor agonists - nausea, vomiting, constipation, diarrhea, and abdominal discomfort - relate directly or indirectly to altered gut motility. Nausea, the most common complaint, peaks during the first 4-8 weeks of treatment and during dose escalation, corresponding to the period of maximum gastric emptying delay. As tachyphylaxis develops, nausea typically improves.
Constipation results from slowed transit throughout the entire GI tract, not just the stomach. GLP-1 receptors in the colonic myenteric plexus reduce propulsive motility, increasing water absorption and hardening stool. This effect also shows some attenuation but can persist in a subset of patients.
The rare but serious concern of bowel obstruction in patients with pre-existing anatomical risk factors (previous abdominal surgery, diverticular disease) has prompted caution. Slow dose titration, as recommended in the prescribing information for semaglutide and tirzepatide, minimizes these risks by allowing gradual adaptation of the GI tract.
Implications for Glycemic Control
Delayed gastric emptying has a separate and significant benefit for blood glucose management. By slowing the rate at which carbohydrates reach the small intestine for absorption, GLP-1 agonists flatten postprandial glucose excursions. This is one reason these drugs were developed initially for type 2 diabetes, and it remains a meaningful benefit for patients using them for weight loss, many of whom have insulin resistance or prediabetes.
The glycemic benefit of delayed gastric emptying is additive with the drug's direct pancreatic effects (enhanced insulin secretion, glucagon suppression). Together, these mechanisms can reduce postprandial glucose peaks by 30-50%, a magnitude comparable to rapid-acting insulin in some patients. For patients with type 2 diabetes on GLP-1 therapy who also take insulin or sulfonylureas, this raises the risk of hypoglycemia, necessitating dose adjustments of the other medications.
Drug Interactions and Procedural Considerations
Delayed gastric emptying can affect the absorption of oral medications. Drugs that require rapid absorption (such as acetaminophen, which is used as a pharmacokinetic marker for gastric emptying studies) may have delayed peak levels. However, clinical studies with semaglutide have shown that total absorption (area under the curve) of most oral medications is not significantly affected; only the timing of peak levels shifts.
Anesthesiologists have raised concerns about aspiration risk in patients on GLP-1 agonists undergoing procedures requiring sedation or general anesthesia. If the stomach hasn't emptied despite standard fasting protocols, residual gastric contents could be aspirated during intubation. The American Society of Anesthesiologists updated its guidelines in 2023, recommending that patients on GLP-1 therapy consider holding their dose before elective procedures and may need extended fasting periods or gastric ultrasound assessment. Our dosing calculator includes perioperative timing guidance.
Perioperative Consideration
If you're scheduled for surgery or an endoscopic procedure requiring sedation, discuss your GLP-1 medication schedule with both your prescribing clinician and your anesthesiologist. Current guidance suggests holding weekly formulations for at least 7 days before elective procedures, though protocols vary by institution. A liquid-only diet for 24 hours before the procedure may also be recommended.
Food Reward & Hedonic Pathways
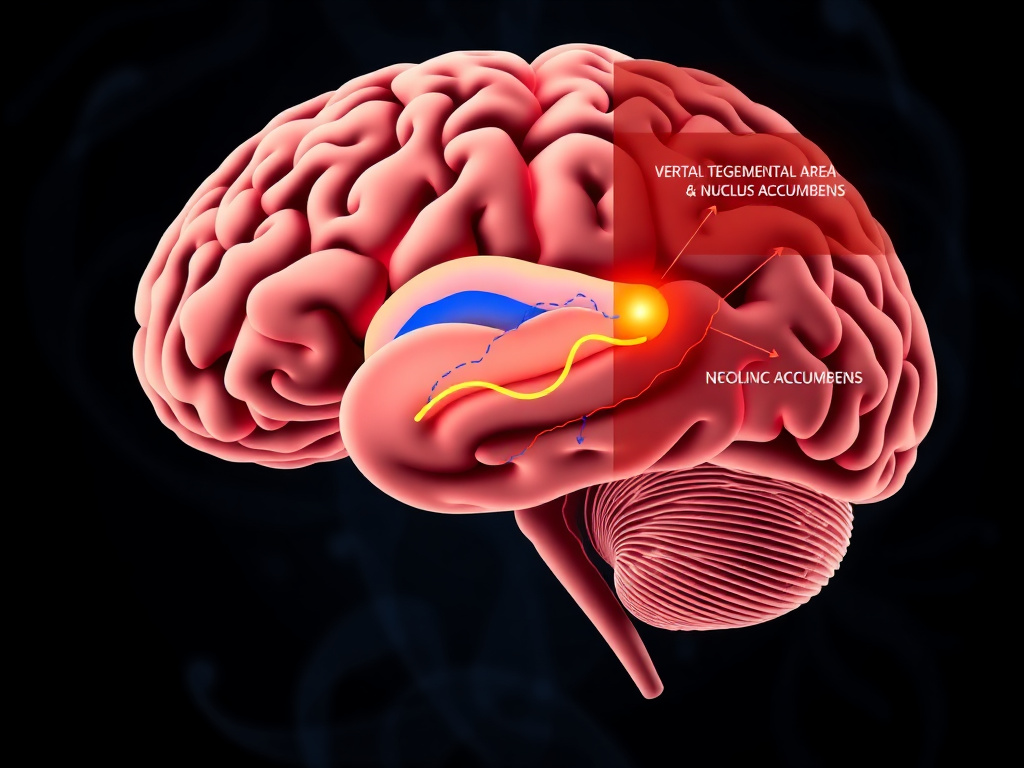
Figure 5: GLP-1 receptors in the mesolimbic dopamine pathway modulate the rewarding properties of food, reducing cravings for highly palatable, calorie-dense items.
Here's what makes GLP-1 drugs different from every previous weight loss medication: they don't just make you less hungry. They make food less rewarding. Patients describe a fundamental shift in their relationship with food - the second helping loses its appeal, the late-night snack doesn't call out from the kitchen, and the dessert menu becomes something they can genuinely skip without willpower. This change traces to GLP-1 receptor activation in the brain's mesolimbic dopamine system, the same circuitry that drives addiction.
The Mesolimbic Reward Circuit
The mesolimbic dopamine pathway runs from the ventral tegmental area (VTA) in the midbrain to the nucleus accumbens (NAc) in the ventral striatum. Dopaminergic neurons in the VTA fire in response to rewarding stimuli - food, sex, social connection, and unfortunately, drugs of abuse. The dopamine they release into the NAc generates the subjective experience of "wanting" and drives motivated behavior to obtain the reward.
This system evolved to promote survival by reinforcing behaviors that provide calories. A calorie-dense food triggers a larger dopamine response than a low-calorie one, creating a learned preference for energy-rich items. In the modern food environment, where ultra-processed foods are engineered to maximize palatability, this ancient reward system drives overconsumption. People don't overeat because they lack willpower; they overeat because their reward circuits are responding exactly as evolution designed them to.
GLP-1 receptors are expressed on neurons in both the VTA and the NAc. Their activation directly modulates the dopamine signaling that makes food rewarding. This isn't a subtle effect. Functional MRI studies in humans have demonstrated that semaglutide treatment reduces neural activation in the reward centers when subjects view images of highly palatable foods. The brain simply responds less to food cues.
Preclinical Evidence: How GLP-1 Dampens Food Reward
The preclinical data on GLP-1 and food reward are extensive and compelling. Direct injection of GLP-1 agonists into the VTA of rodents reduces food intake, with the strongest effect on high-fat, high-sugar foods. This site-specific effect demonstrates that VTA GLP-1 receptors independently contribute to appetite control, separate from the hypothalamic and brainstem mechanisms discussed earlier.
Microdialysis studies show that GLP-1 receptor activation in the VTA reduces dopamine release in the NAc in response to palatable food. The mechanism involves both direct inhibition of VTA dopamine neurons and enhancement of local inhibitory interneuron activity. The result is a dampened dopamine signal for food - the food is still there, but the brain's "want" response to it is weaker.
Conditioned place preference (CPP) experiments provide behavioral confirmation. In CPP studies, animals learn to associate a specific location with a reward (like high-fat food). GLP-1 agonist treatment reduces the preference for food-paired locations, indicating that the rewarding properties of food are genuinely diminished rather than simply being overridden by nausea or malaise. This is an important distinction: the animals don't avoid food because they feel sick; they approach it less because it's less appealing.
Human Neuroimaging Studies
Functional MRI data from human trials have confirmed the preclinical findings. In the STEP 1 substudy, participants treated with semaglutide 2.4 mg showed significantly reduced blood-oxygen-level-dependent (BOLD) signal in the caudate nucleus, putamen, and insula when viewing images of highly palatable foods, compared to both baseline and placebo. These brain regions are core components of the reward and interoceptive processing networks.
A separate crossover study using liraglutide found similar results: reduced activation in the insula, amygdala, and orbitofrontal cortex (OFC) in response to food images after two weeks of treatment. The OFC is particularly significant because it encodes the subjective value of food - how much you'd be willing to "pay" (in effort, time, or opportunity cost) to obtain a particular item. Reduced OFC activation suggests that GLP-1 agonists lower the perceived value of food at a computational level in the brain.
Behavioral data match the neuroimaging. In controlled feeding studies, patients on semaglutide eat less at ad libitum buffets, but they specifically reduce their intake of high-fat and high-sugar items while maintaining relatively normal intake of lean proteins and vegetables. This selective reduction in palatable food consumption is consistent with a reward-based mechanism rather than a general suppression of all eating.
Implications for Food Cravings and Preferences
Survey data from GLP-1 clinical trials consistently report significant reductions in food cravings. In the STEP 5 trial (two-year semaglutide data), patients reported reduced cravings for sweets, fatty foods, and fast food. Many also reported that their taste preferences shifted: foods they previously found irresistible became merely acceptable, and they developed greater appreciation for simpler, less processed foods.
These changes are not simply due to nausea avoidance. While nausea can create temporary food aversions, the cravings reduction persists long after nausea resolves, follows a different time course (deepening over weeks rather than fading), and is specific to palatable foods rather than applying to all food equally. The pattern is consistent with reward circuit modulation rather than conditioned taste aversion.
Anecdotally, clinicians report that patients on GLP-1 therapy describe what amounts to a normalization of their relationship with food. They can keep snacks in the house without eating them. They can attend social events without fixating on the food table. They can eat a portion-controlled meal and feel genuinely satisfied. For people who have struggled with food preoccupation for years or decades, this psychological shift is often described as the most transformative aspect of treatment.
Alcohol and Substance Use: The Addiction Connection
Because the mesolimbic dopamine system mediates reward for all reinforcing stimuli (not just food), GLP-1 receptor agonists may also affect alcohol consumption and other addictive behaviors. Preclinical studies have consistently shown that GLP-1 agonists reduce alcohol intake in rodent models. Exendin-4 and liraglutide both decrease voluntary alcohol drinking, alcohol-seeking behavior, and the dopamine surge in the NAc triggered by alcohol.
Human observational data support this finding. A large pharmacovigilance analysis of FDA adverse event reports found lower-than-expected rates of alcohol use disorder diagnoses among patients prescribed semaglutide. Survey studies report that a substantial minority of patients on GLP-1 therapy spontaneously reduce their alcohol intake, often describing the same change in "wanting" they experience with food: they can have one drink and simply don't want another.
Clinical trials specifically testing GLP-1 agonists for alcohol use disorder are underway. If positive, these drugs would represent a fundamentally new approach to addiction treatment, based on modulating the reward circuit's response to the substance rather than blocking its direct pharmacological effects. Similar early-stage investigations are examining GLP-1 agonists for nicotine dependence and even compulsive gambling, though these remain speculative.
The Dopamine Hypothesis of Obesity
The reward-based mechanism of GLP-1 drugs supports a broader theory that obesity is, in part, a disorder of the dopamine reward system. Research by Nora Volkow and others at the National Institute on Drug Abuse has shown that individuals with obesity have reduced D2 dopamine receptor availability in the striatum, a pattern also seen in people with substance addictions. This "reward deficiency" may drive overconsumption as the brain seeks more stimulation to achieve the same dopamine response.
GLP-1 receptor agonists appear to partially normalize this system. Rather than simply suppressing dopamine (which could cause anhedonia and depression), they modulate the response specifically to food and potentially to other overconsumption behaviors, while preserving normal dopamine function for non-food rewards. How this selectivity is achieved at the receptor level remains an active area of research, but it may involve the specific patterns of GLP-1 receptor expression on different VTA neuron subpopulations.
For comprehensive product information on the GLP-1 compounds discussed here, see the GLP-1 class overview and our individual guides for semaglutide and tirzepatide.
Pancreatic & Metabolic Effects
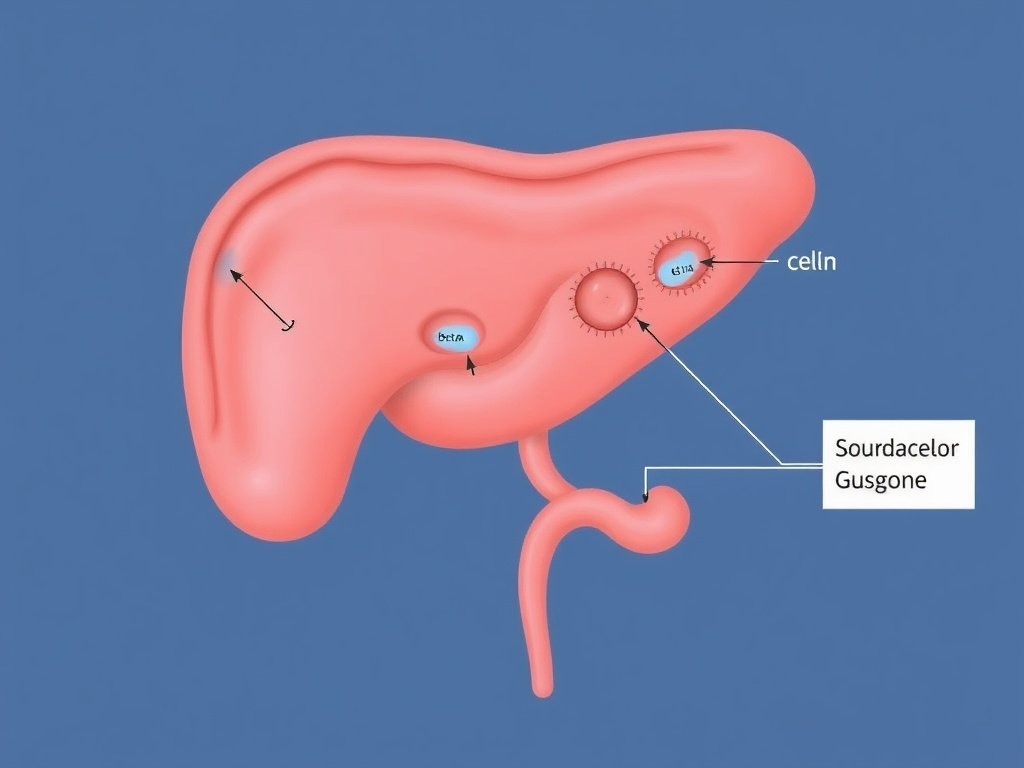
Figure 6: GLP-1 receptor agonists enhance glucose-dependent insulin secretion from beta cells, suppress glucagon from alpha cells, and improve hepatic and lipid metabolism.
Before GLP-1 receptor agonists became weight loss drugs, they were diabetes drugs. And their metabolic effects - particularly on insulin, glucagon, and hepatic glucose production - remain central to their overall physiological impact. These metabolic actions don't just improve blood sugar. They create a metabolic environment that favors fat mobilization and utilization over storage, contributing to the weight loss effect through mechanisms distinct from appetite suppression.
Glucose-Dependent Insulin Secretion
GLP-1 receptors on pancreatic beta cells are the original therapeutic target. When GLP-1 binds these receptors, it activates a cAMP/PKA signaling cascade that sensitizes the beta cell's glucose-sensing machinery. The cell becomes more responsive to ambient glucose: when blood sugar rises after a meal, the GLP-1-primed beta cell releases more insulin than it would without GLP-1 stimulation.
The glucose-dependent nature of this effect is crucial for safety. Unlike sulfonylureas (which stimulate insulin release regardless of blood glucose) or exogenous insulin (which lowers glucose whether it's high or normal), GLP-1 receptor agonists only enhance insulin secretion when glucose is elevated. As blood glucose falls toward normal, the stimulatory effect wanes. This built-in safety mechanism makes hypoglycemia uncommon with GLP-1 monotherapy, a major advantage over older diabetes medications.
At the molecular level, GLP-1 receptor activation increases intracellular cAMP, which activates both protein kinase A (PKA) and exchange protein directly activated by cAMP (Epac2). PKA phosphorylates L-type calcium channels and KATP channels, enhancing calcium influx and insulin granule exocytosis. Epac2 directly interacts with the exocytic machinery (Rab3, Rim2) to increase the readily releasable pool of insulin-containing granules. The result is not just more insulin per secretory event but also faster first-phase insulin release, restoring a pattern lost in type 2 diabetes.
Glucagon Suppression
Alpha cells in the pancreatic islets produce glucagon, the counter-regulatory hormone that stimulates hepatic glucose production. In type 2 diabetes, glucagon secretion is paradoxically elevated, contributing to fasting and postprandial hyperglycemia. GLP-1 receptor agonists suppress glucagon release, though the mechanism is surprisingly complex.
Alpha cells express GLP-1 receptors, but direct receptor activation actually stimulates rather than inhibits them. The suppressive effect on glucagon appears to be indirect, mediated through paracrine signaling. GLP-1-stimulated insulin release from adjacent beta cells activates insulin receptors on alpha cells, which suppresses glucagon secretion. Additionally, GLP-1 stimulates delta cell secretion of somatostatin, another potent glucagon suppressant. This paracrine network within the islet means the glucagon-suppressive effect depends on intact islet architecture and functional beta cells.
The suppression of glucagon has metabolic consequences beyond glucose control. Glucagon promotes hepatic glycogenolysis and gluconeogenesis, but it also stimulates hepatic lipid oxidation and ketogenesis. By suppressing glucagon, GLP-1 agonists shift hepatic metabolism away from glucose production and toward lipid storage. This might seem counterproductive for weight loss, but the net effect is favorable because the reduced glucose output lowers insulin requirements, reducing the lipogenic (fat-storing) signal in peripheral tissues.
Hepatic Effects
The liver doesn't express GLP-1 receptors at meaningful levels, so the hepatic effects of GLP-1 agonists are largely indirect, mediated through changes in insulin, glucagon, and substrate delivery. Reduced glucagon and enhanced insulin shift the liver toward glycogen synthesis and away from glucose production. This reduces fasting glucose and decreases the liver's contribution to postprandial hyperglycemia.
GLP-1 receptor agonists also reduce hepatic steatosis (fatty liver). Clinical trials with semaglutide and liraglutide have shown significant reductions in liver fat content measured by MRI-proton density fat fraction. In the LEAN trial, liraglutide resolved non-alcoholic steatohepatitis (NASH) in 39% of patients versus 9% with placebo. Semaglutide showed even stronger effects in subsequent NASH-focused trials, reducing liver inflammation and fibrosis scores.
The mechanism involves reduced de novo lipogenesis (new fat production) from excess carbohydrate, increased hepatic fatty acid oxidation driven by the overall negative energy balance, and reduced delivery of dietary fat to the liver due to lower food intake. Weight loss itself improves liver fat through these pathways, but GLP-1 agonists may have additional hepatoprotective effects through anti-inflammatory signaling and reduced oxidative stress, though direct hepatocyte GLP-1R signaling remains debated.
Lipid Metabolism
GLP-1 receptor agonist therapy consistently improves the lipid profile. Triglycerides decrease by 15-25%, with the effect attributable to both reduced hepatic VLDL production and enhanced peripheral lipoprotein lipase activity. Total cholesterol and LDL cholesterol show modest reductions (5-10%), while HDL cholesterol may increase slightly. The triglyceride reduction is the most clinically significant change and likely contributes to the cardiovascular benefits observed in outcome trials like SUSTAIN-6 and SELECT.
Free fatty acid levels decrease during GLP-1 therapy, reflecting improved insulin-mediated suppression of lipolysis in adipose tissue. Lower circulating free fatty acids reduce lipotoxicity to beta cells, muscle, and liver, creating a positive feedback loop: better metabolic function leads to further improvements in glucose and lipid handling.
Effects on Energy Expenditure
Whether GLP-1 receptor agonists affect energy expenditure (calorie burning) is one of the more debated topics in the field. Weight loss from any cause typically reduces resting metabolic rate through a process called metabolic adaptation. The body defends against weight loss by becoming more efficient, burning fewer calories per kilogram of body weight. This adaptation is a major driver of weight regain after dieting.
GLP-1 agonists don't fully prevent metabolic adaptation, but some evidence suggests they may partially mitigate it. Rodent studies show that GLP-1 receptor activation increases sympathetic nervous system output to brown adipose tissue, enhancing non-shivering thermogenesis. In humans, indirect calorimetry studies have produced mixed results: some show preservation of resting metabolic rate beyond what would be expected for the degree of weight lost, while others show the typical metabolic slowdown.
Retatrutide, which adds glucagon receptor activation to GLP-1 and GIP, may have stronger energy expenditure effects because glucagon directly stimulates hepatic thermogenesis and brown fat activation. This triple-agonist approach could theoretically produce weight loss from both reduced intake and increased expenditure, a combination that would be particularly powerful for overcoming metabolic adaptation. Early phase 2 data showing up to 24.2% weight loss are consistent with this hypothesis.
Metabolic Summary
The pancreatic and metabolic effects of GLP-1 receptor agonists contribute to weight loss primarily by creating a metabolic environment that favors fat mobilization over storage. Enhanced insulin sensitivity, reduced glucagon-driven glucose production, improved lipid handling, and possible preservation of energy expenditure all work alongside appetite suppression and delayed gastric emptying. For patients with type 2 diabetes or metabolic syndrome, these effects represent independent therapeutic benefits beyond weight loss. Learn more about specific compound profiles at GLP-1 weight loss.
Body Composition Changes
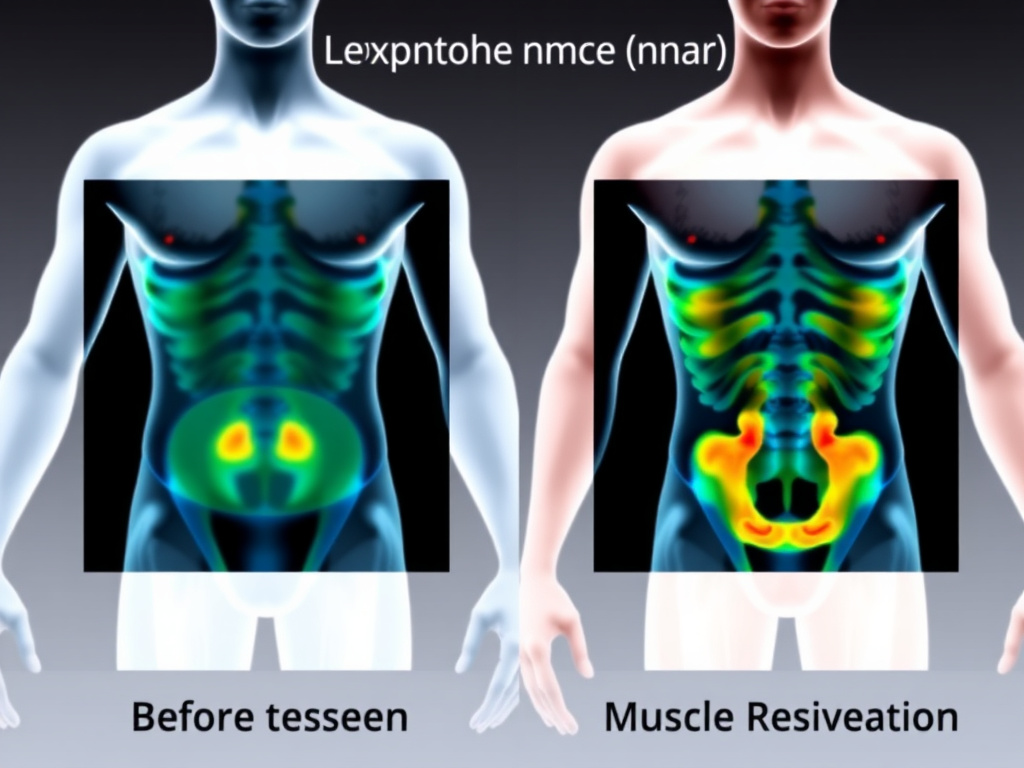
Figure 7: DEXA scanning and other body composition methods reveal that GLP-1 therapy produces predominantly fat loss, though lean mass losses of 25-40% of total weight lost warrant attention.
Weight loss is the headline number, but what you lose matters as much as how much you lose. A kilogram of fat lost and a kilogram of muscle lost have very different health implications. GLP-1 receptor agonist trials have consistently measured body composition using dual-energy X-ray absorptiometry (DEXA), providing detailed data on how these drugs partition weight loss between fat mass and lean mass. The picture is mostly encouraging, with one significant caveat.
The Fat-to-Lean Ratio in GLP-1 Trials
Across the major clinical programs, GLP-1 receptor agonists produce weight loss that is approximately 60-75% fat mass and 25-40% lean mass. This ratio is broadly similar to what's observed with caloric restriction alone, though some analyses suggest GLP-1 drugs may slightly favor fat loss compared to equivalent weight loss from diet alone.
Body Composition Changes on GLP-1 Therapy (% of Total Loss)
In the STEP 1 trial, body composition was assessed in a subset of participants using DEXA. Semaglutide 2.4 mg produced a total body weight loss of approximately 15%, with fat mass accounting for about 68% of the loss and lean mass about 32%. The fat mass reduction was particularly pronounced in visceral adipose tissue (the metabolically dangerous fat surrounding internal organs), which decreased by roughly 20-25%.
The SURMOUNT-1 trial with tirzepatide showed similar proportions. At the 15 mg dose, total weight loss averaged 22.5%, with DEXA-measured fat mass loss accounting for about 73% of total loss and lean mass 27%. The slightly more favorable ratio compared to semaglutide may reflect tirzepatide's GIP receptor co-activation, as GIP signaling has been linked to adipocyte lipid handling and may preferentially mobilize fat stores.
| Trial | Drug | Total Weight Loss (%) | Fat Mass Lost (%) | Lean Mass Lost (%) |
|---|---|---|---|---|
| STEP 1 | Semaglutide 2.4 mg | 14.9% | ~68% | ~32% |
| SURMOUNT-1 | Tirzepatide 15 mg | 22.5% | ~73% | ~27% |
| SURMOUNT-1 | Tirzepatide 10 mg | 19.5% | ~71% | ~29% |
| STEP 1 (Placebo) | Lifestyle alone | 2.4% | ~55% | ~45% |
Why Lean Mass Loss Matters
Lean mass includes skeletal muscle, bone, organs, and water. Of these, skeletal muscle is the component most affected by weight loss and the most relevant to health outcomes. Muscle mass is a major determinant of resting metabolic rate, insulin sensitivity, functional capacity, and fall risk (especially in older adults). Losing excessive muscle during weight loss can undermine long-term metabolic health and physical function.
The concern is amplified by the magnitude of weight loss GLP-1 agonists produce. A 15% total body weight loss with 30% of that from lean mass means roughly 4.5% of starting lean mass is lost. At 22.5% total weight loss (tirzepatide 15 mg), the lean mass loss could approach 6-7% of starting lean mass. For a 100 kg person, that's 6-7 kg of lean tissue, a substantial amount that could affect physical performance and metabolic rate.
Sarcopenic obesity, a condition characterized by both excess fat and inadequate muscle mass, is already a concern in the population most likely to use GLP-1 drugs. Older adults with obesity are at particular risk, as age-related muscle loss (sarcopenia) compounds the lean mass depletion from pharmacological weight loss. This population may benefit most from concurrent exercise interventions.
Visceral Fat: The Primary Target
The news on visceral fat is strongly positive. GLP-1 receptor agonists reduce visceral adipose tissue disproportionately compared to subcutaneous fat. In the STEP 1 DEXA substudy, visceral fat decreased by roughly 20-25%, a larger relative reduction than the 15% total body weight loss. This preferential visceral fat reduction is clinically meaningful because visceral fat is the depot most strongly associated with insulin resistance, systemic inflammation, and cardiovascular risk.
The mechanism likely involves the improved insulin signaling produced by GLP-1 therapy. Insulin normally suppresses lipolysis (fat breakdown), but visceral adipocytes are more resistant to insulin's antilipolytic effect than subcutaneous adipocytes. As insulin sensitivity improves with GLP-1 treatment and overall energy balance becomes negative, visceral fat is mobilized more readily than subcutaneous fat.
AOD-9604, a peptide fragment of human growth hormone, has been investigated for its potential to enhance fat mobilization specifically. Some clinicians have explored combining AOD-9604 with GLP-1 therapy to further target adipose tissue while sparing lean mass, though large controlled trials of this combination are lacking.
Comparison to Surgical Weight Loss
Bariatric surgery, the previous gold standard for substantial weight loss, produces similar body composition changes. Roux-en-Y gastric bypass typically results in 65-70% fat loss and 30-35% lean mass loss as a proportion of total weight lost. Sleeve gastrectomy shows comparable ratios. The fact that GLP-1 agonists produce similar or slightly better fat-to-lean ratios, without the risks of surgery, is a significant advantage.
One difference is that bariatric surgery patients often have more severe protein malabsorption, particularly after Roux-en-Y, which can exacerbate lean mass loss. GLP-1 agonist patients maintain normal absorptive capacity, meaning that dietary protein intake is fully available for muscle protein synthesis. This creates an opportunity: with adequate protein intake and resistance training, the lean mass losses seen in clinical trials (which did not mandate exercise) could likely be reduced substantially. We cover these strategies in the next section.
For those exploring cagrilintide combined with semaglutide (CagriSema), early data suggest body composition outcomes that are at least as favorable as semaglutide alone, with the amylin pathway potentially enhancing fat-specific weight loss. This combination is covered in depth in our GLP-1 class overview.
Muscle Mass Preservation Strategies

Figure 8: Resistance training combined with adequate protein intake is the most evidence-based strategy for preserving lean mass during GLP-1-mediated weight loss.
If GLP-1 receptor agonists are the engine of weight loss, resistance training and protein intake are the steering wheel that directs where that loss comes from. The lean mass losses observed in clinical trials occurred in the absence of structured exercise programs. With the right interventions, you can shift the ratio decisively toward fat loss while maintaining or even building muscle. Here's the evidence-based playbook.
Resistance Training: The Non-Negotiable
Resistance training is the single most effective intervention for preserving muscle mass during caloric deficit, whether that deficit comes from diet, drugs, or both. The mechanical stimulus of lifting weights activates the mTOR (mechanistic target of rapamycin) signaling pathway in muscle fibers, promoting muscle protein synthesis even when overall energy balance is negative. This signal tells the body that muscle is being used and should be preserved, redirecting weight loss toward fat stores.
The data are unequivocal. A meta-analysis of 66 studies found that combining caloric restriction with resistance training preserved an average of 93% of lean mass, compared to 78% preservation with caloric restriction alone. Applied to GLP-1 therapy, this would mean that a patient losing 15 kg on semaglutide might lose only 1 kg of lean mass with resistance training versus 4-5 kg without it.
The recommended protocol is straightforward:
- Frequency: 2-4 sessions per week, targeting all major muscle groups
- Volume: 3-4 sets of 8-12 repetitions per exercise, working to within 2-3 repetitions of failure
- Progressive overload: Gradually increase weight, sets, or repetitions over time
- Exercise selection: Prioritize compound movements (squats, deadlifts, bench press, rows, overhead press) that recruit multiple muscle groups simultaneously
- Recovery: Allow 48-72 hours between sessions targeting the same muscle group
Patients new to resistance training should start conservatively and progress gradually. Working with a qualified trainer for the first few sessions to learn proper form reduces injury risk and improves adherence. The appetite suppression from GLP-1 therapy can sometimes reduce exercise motivation due to lower energy intake; scheduling workouts at consistent times and focusing on habit formation helps maintain consistency.
Protein Intake: The Muscle-Sparing Macronutrient
Protein is the dietary component most directly linked to muscle preservation during weight loss. Each gram of protein provides amino acids for muscle protein synthesis and generates a stronger thermic effect (calorie burn during digestion) than either carbohydrates or fats. During caloric deficit, higher protein intake shifts the body toward preferential fat oxidation while maintaining muscle protein turnover.
Current evidence supports a protein target of 1.2-1.6 grams per kilogram of body weight per day for adults losing weight on GLP-1 therapy. For a 90 kg (200 lb) individual, this translates to 108-144 grams of protein daily. Some exercise scientists recommend even higher intakes (up to 2.0 g/kg) during combined caloric deficit and resistance training, though the incremental benefit above 1.6 g/kg is modest.
This presents a practical challenge. GLP-1 agonists reduce appetite and food volume, making it difficult for some patients to consume enough total food to hit protein targets. Strategies to address this include:
- Protein prioritization: Eat protein-rich foods first at each meal before filling up on lower-protein items
- Protein supplementation: Whey, casein, or plant-based protein shakes can efficiently deliver 25-40 grams of protein in a small volume
- Leucine-rich sources: Leucine is the amino acid that most potently activates mTOR. Whey protein, eggs, chicken, and fish are particularly rich sources
- Meal timing: Distribute protein intake across 3-4 meals/snacks, aiming for at least 25-30 grams per eating occasion to maximize muscle protein synthesis at each meal
Creatine Monohydrate
Creatine is the most extensively studied ergogenic supplement in sports science, with over 500 peer-reviewed papers supporting its safety and efficacy. Creatine monohydrate supplementation (3-5 grams daily) increases intramuscular phosphocreatine stores, enhancing performance during high-intensity exercise (like resistance training) and supporting muscle recovery.
During weight loss, creatine's benefits extend beyond performance. Creatine draws water into muscle cells, creating a cellular hydration effect that may attenuate protein breakdown signals. Multiple studies have shown that combining creatine with resistance training during caloric deficit results in greater lean mass preservation compared to resistance training alone. A reasonable starting protocol is 5 grams of creatine monohydrate daily, taken at any time (timing relative to workouts has minimal impact).
Creatine does increase body weight by 1-2 kg due to intracellular water retention. Patients tracking scale weight should be aware of this and not interpret the initial weight plateau or slight increase as a failure of their GLP-1 therapy. Body composition measurements (DEXA, bioimpedance, or even waist circumference) provide more meaningful data than scale weight alone.
Additional Supportive Strategies
Vitamin D and calcium: Adequate vitamin D (2,000-4,000 IU daily) and calcium (1,000-1,200 mg daily) support both muscle function and bone health during weight loss. GLP-1-mediated weight loss, like any substantial weight reduction, can decrease bone mineral density. Ensuring vitamin D sufficiency (serum 25-OH vitamin D above 30 ng/mL) is a simple protective measure.
Omega-3 fatty acids: Fish oil supplementation (2-3 grams EPA+DHA daily) has shown modest benefits for muscle protein synthesis in older adults and may reduce the inflammatory response to exercise, potentially supporting recovery during the caloric deficit imposed by GLP-1 therapy.
BPC-157: This peptide, derived from a protein found in gastric juice, has been investigated for its potential to support tissue healing and recovery. Some practitioners include BPC-157 in protocols for patients on GLP-1 therapy who are engaged in resistance training, based on preclinical data suggesting enhanced tendon and ligament healing. Human clinical data remain limited, and its use in this context is considered experimental.
Aerobic exercise: While resistance training is the priority for muscle preservation, moderate aerobic activity (150 minutes per week of walking, cycling, or swimming) supports cardiovascular health, insulin sensitivity, and mental wellbeing during weight loss. The key is not to substitute aerobic exercise for resistance training, as aerobic exercise alone does little to prevent lean mass loss.
Practical Protocol Summary
For patients on GLP-1 therapy seeking to maximize fat loss and minimize muscle loss: (1) Perform resistance training 2-4 times per week with progressive overload; (2) Consume 1.2-1.6 g protein per kg body weight daily, distributed across meals; (3) Consider creatine monohydrate 5 g/day; (4) Maintain vitamin D sufficiency; (5) Track body composition rather than relying solely on scale weight. Use our dosing calculator for GLP-1 titration schedules that complement your exercise program.
Weight Regain After Discontinuation
What happens when you stop? It's the question every GLP-1 patient asks eventually, and the data provide a clear, if uncomfortable, answer: most of the weight comes back. Understanding why this happens, how quickly it occurs, and what strategies might mitigate it is essential for anyone using or considering GLP-1 therapy for weight management.
The STEP 4 Withdrawal Study
The most definitive data on weight regain come from STEP 4, a trial specifically designed to answer the discontinuation question. In this study, all participants received semaglutide 2.4 mg for an initial 20-week run-in period, losing an average of 10.6% of their body weight. At week 20, participants were randomized to either continue semaglutide or switch to placebo for the remaining 48 weeks.
Those who continued semaglutide lost an additional 7.9% (total 17.4% from baseline). Those switched to placebo regained an average of 6.9% from their week-20 weight, ending the trial at roughly 5% below their original baseline. In other words, they regained about two-thirds of the weight they had lost during the initial 20 weeks. The regain began within weeks of discontinuation and continued throughout the 48-week off-treatment period.
Cardiometabolic improvements followed the same pattern. Improvements in waist circumference, blood pressure, HbA1c, and lipid levels that had been achieved during the semaglutide treatment period partially reversed during the withdrawal phase. This demonstrates that the metabolic benefits of GLP-1 therapy, like the weight loss itself, are sustained by ongoing treatment rather than by permanent physiological changes.
STEP 1 Extension Data
Follow-up data from STEP 1 participants who discontinued semaglutide after the initial 68-week trial showed similar results. Over the year following treatment cessation, participants regained approximately two-thirds of their lost weight. Appetite scores, which had been significantly reduced during semaglutide treatment, returned to baseline levels within 4-8 weeks of stopping the drug. This rapid return of hunger confirms that the appetite-suppressive effects are pharmacologically maintained rather than reflecting a learned behavioral change.
Why Weight Regain Occurs: The Biology of Defense
Weight regain after stopping GLP-1 therapy is not a failure of willpower. It's the predictable result of deeply conserved biological defense systems. The body treats weight loss as a threat to survival and activates multiple compensatory mechanisms to restore the previous weight. These mechanisms include:
- Increased hunger hormones: Ghrelin (the "hunger hormone") levels rise after weight loss, increasing appetite above pre-diet levels. GLP-1 therapy suppresses ghrelin, but this suppression disappears when the drug is stopped.
- Decreased satiety hormones: Leptin, PYY, and cholecystokinin levels fall in proportion to fat mass loss, reducing the strength of satiety signals reaching the brain.
- Metabolic adaptation: Resting metabolic rate decreases beyond what would be predicted from the loss of metabolically active tissue, meaning the body burns fewer calories at rest. This adaptation can persist for years after weight loss.
- Neural circuit remodeling: The hypothalamic and reward circuit changes induced by GLP-1 therapy likely reverse after discontinuation. The synaptic plasticity and receptor sensitivity changes that contributed to appetite suppression fade as receptor occupancy drops.
- Behavioral return: Without the pharmacological reduction in appetite and food reward, previously established eating patterns and food preferences tend to reassert themselves.
These compensatory mechanisms are not unique to GLP-1 therapy. They are the same forces that drive weight regain after any form of weight loss, including diet, exercise, and even bariatric surgery (where some regain is typical after 2-3 years). The difference is that GLP-1 therapy, while active, directly counteracts many of these mechanisms. Removing the drug removes the counterforce, and biology does what biology does.
Maintenance Strategies
Given the biology of regain, several approaches are being explored to maintain weight loss:
Continued therapy at maintenance doses: The most straightforward approach is continued GLP-1 therapy, potentially at reduced doses. Some clinicians titrate down from the maximum therapeutic dose to the lowest effective dose that maintains weight stability, reducing cost and side effect burden. This approach is supported by data showing dose-dependent effects: even lower doses of semaglutide produce meaningful appetite suppression and weight maintenance, even if they wouldn't produce the same initial weight loss trajectory.
Intermittent dosing: Some practitioners have explored intermittent dosing strategies, such as every-other-week injections at maintenance phase. While formal clinical trial data on this approach are limited, the long half-life of semaglutide (165 hours) means that even extended dosing intervals maintain some receptor occupancy. This remains an area where more research is needed.
Combination with lifestyle changes: Patients who establish strong exercise habits and dietary patterns during GLP-1 therapy may retain some behavioral benefits after discontinuation. While the pharmacological appetite suppression disappears, the dietary knowledge, cooking habits, and exercise routines built during treatment can provide a partial buffer against regain. The key word is "partial" - lifestyle changes alone typically cannot fully compensate for the loss of pharmacological appetite control.
Transition to oral formulations: Oral semaglutide is available, and higher-dose oral formulations (25 mg and 50 mg) have shown weight loss approaching that of the injectable form. Transitioning from injectable to oral semaglutide could provide a lower-burden maintenance option for patients who achieve their weight goals on the injectable form.
Anti-obesity medication combinations: For patients who cannot continue GLP-1 therapy (due to cost, side effects, or preference), transitioning to another anti-obesity medication may partially mitigate regain. Phentermine-topiramate, naltrexone-bupropion, or even lower-cost options might maintain some weight loss, though none approaches the efficacy of GLP-1 agonists.
The Chronic Disease Model
Obesity is increasingly recognized as a chronic, relapsing disease with strong biological underpinnings. Just as hypertension returns when antihypertensive medication is stopped, and blood glucose rises when diabetes medication is discontinued, body weight increases when anti-obesity medication is withdrawn. This framing helps patients understand that long-term treatment is not a personal failure but a medical necessity for a chronic condition. Our free assessment can help you evaluate long-term treatment options.
GLP-1 Receptor Structural Biology and Downstream Signaling
To truly understand how GLP-1 drugs cause weight loss, you need to look beyond the broad strokes of "appetite suppression" and examine the receptor itself. The GLP-1 receptor (GLP-1R) is a class B1 G protein-coupled receptor with a remarkably complex signaling profile, and the way different agonists interact with it determines everything from efficacy to side effect profiles.
Receptor Structure and Ligand Binding
The GLP-1R consists of 463 amino acids organized into two major structural domains: an extracellular domain (ECD) that acts as the initial docking site for peptide ligands, and a transmembrane domain (TMD) composed of seven alpha-helical segments that span the cell membrane. When native GLP-1(7-36) binds, the C-terminal portion of the peptide first contacts the ECD, which then positions the N-terminal region to engage the TMD's orthosteric binding pocket. This two-step binding mechanism is characteristic of class B GPCRs and has important implications for drug design.
Cryo-EM structures published between 2020 and 2024 have revealed that different agonists stabilize distinct receptor conformations. Native GLP-1 produces a receptor conformation that favors balanced signaling through both G-protein and beta-arrestin pathways. Semaglutide, with its C-18 fatty diacid modification at position 26 (replacing the original lysine), creates additional contacts with the receptor's extracellular loops that subtly alter the TMD conformation. This structural difference partly explains why semaglutide produces stronger anorectic effects than native GLP-1 at equivalent receptor occupancy levels.
The fatty acid side chain of semaglutide also binds to serum albumin with high affinity (Kd approximately 2.1 micromolar), which serves two purposes: it shields the peptide from DPP-4 enzymatic degradation, and it creates a circulating reservoir that extends the half-life to approximately 165 hours. This albumin binding is reversible, allowing a steady-state release of free semaglutide that maintains receptor activation throughout the weekly dosing interval. By contrast, native GLP-1 has a half-life of just 2-3 minutes, making it therapeutically impractical without structural modification.
Biased Agonism and Signal Selectivity
One of the most significant discoveries in GLP-1 receptor pharmacology over the past five years is the concept of biased agonism. Not all GLP-1R agonists activate the same intracellular pathways to the same degree. The receptor can signal through at least four major downstream cascades:
- Gas/cAMP/PKA pathway: This is the classical signaling cascade. GLP-1R activation triggers the Gas subunit of the heterotrimeric G-protein complex, which stimulates adenylyl cyclase to produce cyclic AMP (cAMP). Rising cAMP levels activate protein kinase A (PKA), which phosphorylates downstream targets including CREB (cAMP response element-binding protein). In pancreatic beta cells, this pathway drives insulin secretion. In hypothalamic neurons, it modulates synaptic plasticity and neuropeptide expression. In the brainstem, it enhances the processing of vagal satiety signals.
- Beta-arrestin recruitment: Following G-protein activation, GLP-1R kinases phosphorylate the receptor's intracellular loops and C-terminal tail, creating docking sites for beta-arrestin-1 and beta-arrestin-2. Beta-arrestin binding initiates receptor internalization through clathrin-coated pits and can activate MAPK/ERK signaling independently of G-proteins. The ratio of G-protein to beta-arrestin signaling varies between agonists and appears to influence both efficacy and GI side effects.
- Gaq/PLC/calcium pathway: Some GLP-1R agonists can weakly activate the Gaq pathway, leading to phospholipase C activation, IP3 production, and intracellular calcium release. This pathway appears more relevant in certain neuronal populations and may contribute to the nausea response seen with rapid dose escalation.
- EPAC pathway: cAMP can also activate exchange proteins directly activated by cAMP (EPACs), which are guanine nucleotide exchange factors for Rap1. This pathway regulates cell adhesion, cytoskeletal dynamics, and exocytosis. In beta cells, EPAC2 contributes to insulin granule priming and is thought to work cooperatively with PKA to potentiate glucose-stimulated insulin secretion.
Different therapeutic GLP-1R agonists show varying degrees of bias toward these pathways. Exenatide, derived from the Gila monster peptide exendin-4, shows stronger beta-arrestin recruitment relative to G-protein signaling compared to semaglutide. This difference may partly explain why exenatide produces more nausea at comparable levels of appetite suppression. Researchers at Novo Nordisk and other companies are now actively designing "G-protein biased" agonists that maximize the appetite-suppressive and metabolic effects while minimizing GI side effects linked to beta-arrestin signaling and receptor internalization.
Receptor Trafficking and Desensitization
After activation and beta-arrestin recruitment, the GLP-1R undergoes internalization into endosomal compartments. But here's where things get interesting: unlike many GPCRs that are simply recycled or degraded after internalization, the GLP-1R continues to signal from within endosomes. This "endosomal signaling" produces a sustained cAMP response that differs qualitatively from the rapid, transient signal generated at the plasma membrane.
Studies using fluorescent biosensors have shown that endosomal GLP-1R signaling accounts for a significant proportion of the total cAMP response in both beta cells and neurons. The duration and magnitude of this endosomal signal varies between agonists: semaglutide and liraglutide, with their fatty acid modifications and albumin binding, produce more prolonged endosomal signaling than exenatide. This sustained intracellular signaling may contribute to the superior weight loss efficacy of semaglutide compared to shorter-acting agonists.
Receptor desensitization and downregulation also play roles in the clinical response to GLP-1 therapy. During the first weeks of treatment, particularly during dose escalation, GLP-1R surface expression decreases as internalization rates exceed recycling rates. This partly explains why GI side effects like nausea tend to be worst during the titration phase and improve over time. As the receptor population reaches a new steady state between internalization and recycling, the side effect profile stabilizes, while the more centrally-mediated appetite effects persist because brainstem and hypothalamic neurons maintain sufficient receptor expression for continued signaling.
Tissue-Specific Signaling Differences
The GLP-1R is expressed across a surprisingly wide range of tissues, and its signaling consequences differ substantially depending on the cellular context. This tissue-specific signaling is driven by differences in G-protein subunit expression, scaffold protein availability, and downstream effector coupling.
In pancreatic beta cells, GLP-1R signaling is tightly coupled to glucose-stimulated insulin secretion (GSIS). The cAMP/PKA pathway enhances the sensitivity of the secretory machinery to glucose by promoting calcium influx through L-type calcium channels and by directly priming insulin granules for exocytosis. Critically, this effect is glucose-dependent: GLP-1R agonists only potentiate insulin secretion when blood glucose is above approximately 4.5 mmol/L (81 mg/dL). This glucose-dependent mechanism provides a built-in safety margin against hypoglycemia, which distinguishes GLP-1 therapy from sulfonylureas and exogenous insulin.
In hypothalamic neurons, GLP-1R signaling primarily modulates neuronal excitability and neuropeptide gene expression. In POMC neurons of the arcuate nucleus, cAMP/PKA signaling increases neuronal firing rate and upregulates POMC transcription, leading to greater production of alpha-MSH, the primary anorectic neuropeptide in this circuit. Simultaneously, GLP-1R activation on neighboring NPY/AgRP neurons decreases their firing rate and reduces NPY and AgRP expression. The net effect is a sustained shift in the hypothalamic "setpoint" toward reduced food intake.
In the area postrema and nucleus tractus solitarius (NTS) of the brainstem, GLP-1R signaling integrates peripheral satiety signals from the vagus nerve with central appetite regulatory circuits. This region sits outside the blood-brain barrier, allowing circulating GLP-1R agonists direct access to these neurons. The brainstem GLP-1R-expressing neurons project to the hypothalamus, parabrachial nucleus, and central amygdala, creating a distributed network that influences both the homeostatic and emotional aspects of feeding behavior.
In cardiac tissue, GLP-1R signaling activates cardioprotective pathways including PI3K/Akt, which promotes cardiomyocyte survival under ischemic stress. Clinical trial data showing cardiovascular mortality reductions with liraglutide and semaglutide are consistent with direct cardiac GLP-1R activation, though improvements in metabolic risk factors (weight, blood pressure, lipids) also contribute significantly. For more on this topic, see our GLP-1 research hub.
Comparative Pharmacology of GLP-1 Receptor Agonists for Weight Loss
Not all GLP-1 receptor agonists are created equal when it comes to weight loss. The differences in molecular structure, pharmacokinetics, receptor binding, and signaling bias between available agents translate into clinically meaningful differences in weight reduction, side effect profiles, and patient experience.
First-Generation Agents: Exenatide and Liraglutide
Exenatide (Byetta/Bydureon) was the first GLP-1R agonist approved for clinical use, initially for type 2 diabetes in 2005 and later in an extended-release formulation. Based on exendin-4 from Gila monster saliva, exenatide shares approximately 53% sequence homology with human GLP-1. The twice-daily formulation produced modest weight loss of 2-3 kg over 24-30 weeks in diabetes trials, while the once-weekly extended-release formulation (Bydureon) achieved slightly better results of 2-4 kg. These weight loss magnitudes, while statistically significant, were not considered clinically transformative for obesity treatment.
Liraglutide (Victoza for diabetes at 1.8 mg; Saxenda for obesity at 3.0 mg) represented a step forward. As a modified human GLP-1 analog with a C-16 fatty acid chain attached via a glutamic acid spacer at position 26, liraglutide has 97% sequence homology with native GLP-1 and a half-life of 13 hours, enabling once-daily dosing. In the SCALE obesity trials, liraglutide 3.0 mg produced mean weight loss of 8.0% at 56 weeks, with 33.1% of participants losing at least 10% of body weight. While a significant improvement over exenatide, these results would later be eclipsed by next-generation agents.
Second-Generation: Semaglutide's Structural Advantages
Semaglutide introduced three key structural modifications over liraglutide that collectively transformed its pharmacological profile. First, an alpha-aminoisobutyric acid (Aib) substitution at position 8 provides resistance to DPP-4 cleavage. Second, an arginine-to-alanine substitution at position 34 eliminates a secondary fatty acid attachment site. Third, and most consequentially, the C-18 fatty diacid chain at position 26 (replacing liraglutide's C-16 monoacid) dramatically increases albumin binding affinity and extends the half-life to 165 hours.
These structural changes produce pharmacological consequences beyond simple half-life extension. Semaglutide's enhanced brain penetration, demonstrated through radiolabeled tracer studies, means greater activation of hypothalamic and mesolimbic GLP-1 receptors compared to liraglutide at equivalent peripheral receptor occupancy. The STEP 1 trial demonstrated 14.9% mean weight loss with semaglutide 2.4 mg at 68 weeks, with 32% of participants achieving at least 20% weight loss. This nearly doubled liraglutide's efficacy and established a new benchmark for pharmacological obesity treatment.
Oral semaglutide (Rybelsus) uses a co-formulation with the absorption enhancer SNAC (sodium N-[8-(2-hydroxybenzoyl)amino]caprylate) to facilitate transcellular absorption across the gastric mucosa. The original 14 mg oral dose produced approximately 4.4 kg weight loss in the PIONEER trials. Higher-dose oral formulations of 25 mg and 50 mg, tested in the OASIS trials, have shown weight loss approaching the injectable form: the OASIS-1 trial reported 15.1% weight loss with 50 mg oral semaglutide at 68 weeks. This creates the possibility of achieving injectable-level efficacy through a daily pill, which could dramatically improve patient acceptance and adherence.
Multi-Receptor Agonists: Tirzepatide, Survodutide, and Retatrutide
The most significant recent advance in GLP-1-based weight loss therapy is the development of multi-receptor agonists that activate GLP-1R alongside one or two additional metabolic receptors. These combinations produce additive or complementary weight loss through complementary mechanisms.
Tirzepatide (Mounjaro/Zepbound) is a dual GIP/GLP-1 receptor agonist built on a 39-amino-acid backbone. It activates both the GLP-1R and the glucose-dependent insulinotropic polypeptide (GIP) receptor. While GIP was traditionally viewed as an "obesity hormone" because of its role in fat storage, sustained pharmacological GIP receptor activation paradoxically promotes weight loss through mechanisms that include enhanced central appetite suppression, increased energy expenditure in adipose tissue, and improved insulin sensitivity. The SURMOUNT-1 trial demonstrated 22.5% mean weight loss with tirzepatide 15 mg at 72 weeks, with 36% of participants losing at least 25% of body weight, results approaching those of bariatric surgery.
Survodutide takes a different dual-agonist approach, combining GLP-1R agonism with glucagon receptor agonism. While glucagon traditionally opposes insulin's effects and raises blood glucose, its activation also increases hepatic energy expenditure, promotes fat oxidation, and drives thermogenesis. In Phase 2 trials, survodutide produced up to 18.7% weight loss at 46 weeks, with Phase 3 data expected in 2026.
Retatrutide pushes this multi-receptor approach further by simultaneously activating three receptors: GLP-1R, GIPR, and the glucagon receptor. Early Phase 2 data from the trial showed up to 24.2% mean weight loss at 48 weeks, with the highest dose group showing a trajectory suggesting even greater weight loss with longer treatment. The triple mechanism combines appetite suppression (GLP-1), enhanced insulin sensitivity and central satiety (GIP), and increased energy expenditure (glucagon). Phase 3 trials are currently ongoing.
Head-to-Head Efficacy Comparisons
Direct comparison trials are beginning to emerge, though most efficacy comparisons still rely on cross-trial analysis (which has limitations due to different patient populations and trial designs). Based on available data, the weight loss hierarchy at maximum approved or tested doses over comparable timeframes is approximately:
- Exenatide extended-release: 2-4% total body weight loss
- Liraglutide 3.0 mg: 7-8% total body weight loss
- Semaglutide 2.4 mg (injectable): 15-17% total body weight loss
- Oral semaglutide 50 mg: 15-17% total body weight loss
- Survodutide (Phase 2 optimal dose): 16-19% total body weight loss
- Tirzepatide 15 mg: 20-23% total body weight loss
- Retatrutide (highest Phase 2 dose): 22-24% total body weight loss
These numbers represent mean values from clinical trials with structured lifestyle counseling. Real-world weight loss tends to be somewhat lower due to inconsistent adherence, less intensive behavioral support, and population heterogeneity. Use our dosage calculator to explore what these agents might look like in your specific situation.
Peptide Combinations and Complementary Approaches
Beyond multi-receptor agonists, researchers and clinicians are exploring the combination of GLP-1R agonists with other peptide therapies that target complementary pathways. While these approaches are still primarily investigational, the mechanistic rationale is sound.
The combination of GLP-1R agonists with AOD-9604 or Fragment 176-191 targets fat metabolism through the growth hormone pathway alongside GLP-1-mediated appetite suppression. AOD-9604, a modified fragment of human growth hormone, stimulates lipolysis and inhibits lipogenesis without the insulin resistance and growth-promoting effects of full-length growth hormone. The theoretical advantage of this combination is enhanced fat-specific weight loss with better preservation of lean mass.
Another approach under investigation pairs GLP-1R agonists with tesofensine, a triple monoamine reuptake inhibitor that enhances norepinephrine, dopamine, and serotonin signaling. Tesofensine addresses weight loss through increased thermogenesis and central appetite suppression via monoamine pathways that are distinct from, and potentially complementary to, GLP-1R signaling. Early clinical data with tesofensine monotherapy showed 10-13% weight loss at 24 weeks, and the combination with GLP-1R agonism could theoretically produce additive effects. However, cardiovascular safety (specifically heart rate and blood pressure effects) needs careful monitoring with this combination.
For individuals concerned about lean mass preservation during GLP-1-mediated weight loss, the addition of CJC-1295/Ipamorelin or other growth hormone secretagogues like MK-677 is being explored. Growth hormone promotes protein synthesis and fat oxidation, which could theoretically shift body composition changes during weight loss toward greater fat loss and better muscle preservation. Clinical data specifically on these combinations with GLP-1 therapy remain limited, and the metabolic interactions (particularly regarding glucose homeostasis) require careful consideration.
The mitochondrial peptide MOTS-c represents another complementary approach. MOTS-c activates AMPK signaling and enhances cellular energy metabolism, which could augment the metabolic improvements seen with GLP-1R agonists. By improving mitochondrial function and insulin sensitivity through a pathway independent of GLP-1R activation, MOTS-c may address aspects of metabolic dysfunction that GLP-1 therapy alone doesn't fully correct. Visit the peptide research hub for more on these emerging combination strategies.
GLP-1 Weight Loss Mechanisms in Special Populations
GLP-1 receptor agonists don't work identically in everyone. Age, sex, baseline metabolic status, genetic background, and concurrent medical conditions all influence how these drugs produce weight loss. Understanding these population-specific differences helps clinicians optimize treatment and helps patients set realistic expectations.
Adolescents and Young Adults
The STEP TEENS trial evaluated semaglutide 2.4 mg in adolescents aged 12-17 with obesity (BMI at the 95th percentile or above). After 68 weeks, semaglutide produced a mean BMI reduction of 16.1% compared to a 0.6% increase with placebo. This is a striking result, and the magnitude of BMI reduction actually exceeded what was seen in adult trials when expressed as percentage change.
The enhanced response in adolescents likely reflects several physiological factors. First, adolescents have greater neural plasticity, meaning the reward circuit and hypothalamic setpoint modifications induced by GLP-1R activation may be more pronounced and more rapidly established. Second, adolescents typically have higher baseline metabolic rates relative to body size, which means the caloric deficit produced by appetite suppression translates to a proportionally larger energy imbalance. Third, adolescent obesity has a shorter duration, and there's evidence suggesting that the hypothalamic inflammation and gliosis that contribute to "defended" body weight setpoints become more entrenched with longer obesity duration.
However, there are unique considerations for this age group. Adolescents are still growing, and the effects of sustained energy deficit on linear growth, bone mineral accrual, and pubertal development need long-term monitoring. Lean mass preservation is particularly critical during this developmental period, making resistance training and adequate protein intake (1.6-2.0 g/kg/day of ideal body weight) non-negotiable adjuncts to GLP-1 therapy in this population.
Older Adults (65+)
Older adults with obesity present a different challenge. The phenomenon of "sarcopenic obesity," where excess fat mass coexists with depleted muscle mass, is common in this population and raises specific concerns about lean mass loss during GLP-1 therapy. In the STEP trials, the proportion of weight lost as lean mass was similar across age subgroups (roughly 25-35%), but the consequences of lean mass loss are more severe in older adults who already have reduced muscle reserves.
Falls risk is a particular concern. Muscle weakness from lean mass loss, combined with the potential for orthostatic hypotension (from weight loss-related blood pressure reductions and possible mild dehydration from nausea), can increase fall risk in older adults. Data from semaglutide trials suggest that the fall risk increase is small and offset by the mobility benefits of weight loss, but clinicians should monitor functional capacity and balance throughout treatment.
Pharmacokinetically, GLP-1R agonist exposure in older adults is generally similar to younger adults, though reduced renal clearance (common in aging) can modestly increase drug exposure. Dose adjustments are typically not required based on age alone, but slower titration schedules may improve tolerability. Starting at lower doses and extending the interval between dose increases from 4 weeks to 6-8 weeks is a common clinical practice for patients over 65.
The cardiovascular benefits of GLP-1 therapy are particularly relevant in older adults, who carry higher baseline cardiovascular risk. The SELECT trial demonstrated a 20% reduction in major adverse cardiovascular events with semaglutide 2.4 mg in adults with established cardiovascular disease or high cardiovascular risk, and subgroup analyses showed consistent benefit across age groups. For a detailed discussion of cardiovascular mechanisms, see our GLP-1 cardiovascular research.
Sex-Based Differences in Response
Women and men show subtly different responses to GLP-1 therapy, driven by interactions between sex hormones and GLP-1R signaling. In pooled analyses of the STEP trials, women tended to achieve slightly higher percentage weight loss than men (approximately 1-2 percentage points higher), though men lost more absolute weight due to higher baseline body weights.
Several mechanisms contribute to these sex differences. Estrogen upregulates GLP-1R expression in certain brain regions, particularly the hypothalamus and brainstem, which may explain why premenopausal women show enhanced central appetite suppression compared to men. Conversely, testosterone promotes lean mass preservation during caloric restriction, which may explain why men lose proportionally less lean mass during GLP-1 therapy.
Premenopausal women using GLP-1 therapy should be aware that significant weight loss can restore ovulatory cycles in women with obesity-related anovulation. This has contraceptive implications: women who were previously relying on obesity-related infertility as contraception may become fertile during GLP-1 treatment. Additionally, oral contraceptive absorption may be affected by the delayed gastric emptying induced by GLP-1R agonists. Clinicians typically recommend non-oral contraceptive methods (IUDs, implants, injections) or advise patients to take oral contraceptives at least 1 hour before or 4 hours after GLP-1 agonist injection.
Postmenopausal women lose the estrogen-mediated enhancement of GLP-1R expression, which may slightly reduce the magnitude of appetite suppression compared to premenopausal women. However, postmenopausal women also lose estrogen's protective effects on body composition, making them more prone to visceral fat accumulation and sarcopenia, both of which GLP-1 therapy can help address.
Patients with Type 2 Diabetes
People with type 2 diabetes consistently achieve less weight loss on GLP-1 therapy compared to those without diabetes. In the STEP 2 trial (semaglutide in type 2 diabetes), mean weight loss was 9.6% at 68 weeks, compared to 14.9% in the non-diabetic STEP 1 population. This "diabetes attenuation" of weight loss is seen across all GLP-1R agonists and appears to be approximately 30-40% relative to non-diabetic populations.
The mechanisms behind this reduced weight loss in diabetes are multiple. Insulin resistance alters GLP-1R signaling efficiency in both peripheral and central tissues. Chronic hyperglycemia induces changes in hypothalamic neuron function, including increased inflammatory signaling and altered neuropeptide expression, that may blunt the appetite-suppressive effects of GLP-1R activation. Additionally, many diabetes medications (particularly insulin and sulfonylureas) promote weight gain, creating a pharmacological counterforce to GLP-1-mediated weight loss.
Despite the lower magnitude of weight loss, the metabolic benefits of GLP-1 therapy may be proportionally greater in diabetic patients. Even modest weight loss of 5-10% can produce clinically meaningful improvements in HbA1c (typically 1.0-1.5% reduction), insulin sensitivity, and hepatic steatosis. Tirzepatide has shown particularly strong glycemic efficacy, with over 50% of type 2 diabetes patients achieving HbA1c below 5.7% (normal range) in the SURPASS trials.
Patients on Concurrent Psychiatric Medications
Many antipsychotic and antidepressant medications promote significant weight gain through mechanisms that include histamine H1 receptor blockade, serotonin 5-HT2C receptor antagonism, and increased appetite drive. Patients taking olanzapine, clozapine, quetiapine, mirtazapine, or certain tricyclic antidepressants often gain 5-15 kg within the first year of treatment, and this psychiatric medication-induced weight gain is notoriously resistant to lifestyle intervention.
GLP-1R agonists can counteract some of this medication-induced weight gain, though the magnitude of weight loss is typically less than in patients not taking weight-promoting psychotropics. A study of semaglutide in patients with schizophrenia on antipsychotics showed approximately 7% mean weight loss at 16 weeks, which is clinically meaningful but roughly half of what would be expected in patients without antipsychotic exposure.
An important consideration is the potential for pharmacokinetic interactions. Delayed gastric emptying from GLP-1R agonists can alter the absorption kinetics of orally administered psychiatric medications, potentially affecting peak drug levels and time to peak concentration. For medications with narrow therapeutic windows (lithium, clozapine, certain anticonvulsants), this altered absorption may necessitate more frequent therapeutic drug monitoring during GLP-1 initiation and dose escalation.
Genetic Predictors of GLP-1 Response
Pharmacogenomic research is beginning to identify genetic variants that influence GLP-1 therapy response, though this field remains in its early stages. Several candidate genes have been associated with weight loss variability:
Variants in the GLP1R gene itself can alter receptor expression levels, ligand binding affinity, and signaling efficiency. The rs6923761 polymorphism, a nonsynonymous variant causing an arginine-to-glutamine substitution at position 131 of the receptor's extracellular domain, has been associated with reduced weight loss response to GLP-1R agonist therapy in some (but not all) studies. The effect size is modest, approximately 1-2 kg difference in weight loss over 6 months, and likely interacts with other genetic and environmental factors.
Variants in appetite-regulating genes such as MC4R (melanocortin 4 receptor), FTO (fat mass and obesity-associated gene), and BDNF (brain-derived neurotrophic factor) also influence weight loss response. MC4R loss-of-function variants, which are the most common monogenic cause of severe obesity, appear to attenuate the response to GLP-1 therapy by reducing the downstream signaling efficiency of the melanocortin pathway through which hypothalamic GLP-1R signaling partly operates.
The gut microbiome adds another layer of variability. Composition of the intestinal bacterial community influences endogenous GLP-1 secretion from L-cells, bile acid metabolism (which modulates GLP-1R sensitivity through FXR signaling), and intestinal permeability. Certain bacterial profiles, particularly those enriched in Akkermansia muciniphila and specific Bacteroides species, are associated with better metabolic responses to GLP-1 therapy. This has led to investigational approaches combining GLP-1R agonists with targeted probiotics or prebiotics, though clinical evidence for this strategy remains preliminary.
If you're interested in how your individual factors might influence treatment response, our personalized assessment can help identify the approach most likely to work for your specific situation.
Future Directions in GLP-1 Weight Loss Science
The current generation of GLP-1 receptor agonists represents a genuine inflection point in obesity treatment, but the field is evolving rapidly. The next five to ten years will likely bring agents that are more effective, better tolerated, and available in more convenient formulations.
Oral GLP-1 Agonists: Beyond Semaglutide
Oral delivery of GLP-1R agonists has been a pharmacological challenge because peptides are rapidly degraded in the stomach's acidic environment and poorly absorbed across the intestinal epithelium. Oral semaglutide (Rybelsus) solved this partially through co-formulation with the SNAC absorption enhancer, but bioavailability remains low (approximately 1%) and absorption is highly variable depending on gastric pH, meal timing, and water volume.
Next-generation oral approaches aim to improve bioavailability and reduce dosing constraints. Novo Nordisk is developing oral semaglutide at higher doses (25 mg and 50 mg), which have shown weight loss rivaling the injectable form in the OASIS trials. But more transformative are small molecule GLP-1R agonists, non-peptide compounds that activate the GLP-1R through allosteric or orthosteric mechanisms and can be manufactured through traditional small molecule chemistry rather than peptide synthesis.
Several pharmaceutical companies have small molecule GLP-1R agonists in clinical development. Pfizer's danuglipron, a small molecule GLP-1R agonist, showed modest weight loss in Phase 2 trials but was limited by twice-daily dosing and GI tolerability. The company discontinued the twice-daily version but is developing a once-daily formulation. Roche/Genentech's CT-996, another oral small molecule GLP-1R agonist, showed promising Phase 1 data with weight loss of approximately 6.1% over 28 days. AstraZeneca, Amgen, and other companies also have oral programs in early to mid-stage development.
The potential advantages of small molecule oral GLP-1R agonists are significant: they could be taken with food (eliminating the fasting requirement of oral semaglutide), they would be cheaper to manufacture than peptides, and they could potentially be combined with other oral medications in fixed-dose combination tablets. The main challenge is achieving the same receptor activation profile and weight loss efficacy as peptide agonists, since small molecules interact with the receptor differently than peptides and may produce distinct signaling bias profiles.
Amylin-Based Combination Approaches
Amylin is a 37-amino-acid peptide co-secreted with insulin from pancreatic beta cells. It acts on the area postrema to suppress appetite, slow gastric emptying, and suppress postprandial glucagon secretion, making it mechanistically complementary to GLP-1. The combination of GLP-1R agonism with amylin receptor agonism is being explored as a way to achieve even greater weight loss than GLP-1R agonism alone.
Novo Nordisk's CagriSema combines semaglutide 2.4 mg with cagrilintide, a long-acting amylin analog. The Phase 2 REDEFINE-1 trial showed CagriSema producing approximately 15.6% weight loss at 32 weeks, and Phase 3 trials (REDEFINE program) are ongoing with results expected in 2025-2026. The hypothesis is that amylin receptor activation enhances the brainstem-mediated satiety signaling of GLP-1, producing a more complete suppression of appetite than either mechanism alone.
Targeting Muscle Mass Preservation
One of the most important areas of GLP-1 research is finding ways to shift the weight loss composition more decisively toward fat loss while preserving or even building lean mass. Current GLP-1 monotherapy produces weight loss that is approximately 25-40% lean mass, which, while better than some dietary approaches, still represents a significant concern, particularly for older adults and those at risk of sarcopenia.
Several strategies are under investigation. Bimagrumab, an anti-activin receptor type II antibody that blocks myostatin signaling, has been tested in combination with semaglutide. In a Phase 2 trial, the combination produced similar total weight loss to semaglutide alone but with dramatically improved body composition: participants lost more fat mass and actually gained lean mass during the treatment period. This "recomposition" effect, losing fat while gaining muscle without exercise, represents a potential step forward in obesity pharmacotherapy.
The growth hormone secretagogue pathway offers another route to muscle preservation. Peptides like sermorelin and tesamorelin stimulate endogenous growth hormone release, which promotes protein synthesis and fat oxidation. CJC-1295/Ipamorelin provides a more sustained growth hormone pulse profile that could complement the metabolic effects of GLP-1 therapy. While formal clinical trials of these combinations with GLP-1R agonists are limited, the mechanistic rationale is strong, and the combination is increasingly being used in clinical practice. See our peptide research hub for more on growth hormone secretagogue research.
Long-Acting Depot Formulations
Current GLP-1R agonists require weekly (semaglutide, tirzepatide) or daily (liraglutide, oral semaglutide) dosing. For a chronic disease requiring lifelong treatment, longer dosing intervals would improve adherence and patient convenience. Several companies are developing depot formulations that could provide GLP-1R agonism for weeks to months from a single administration.
Approaches include biodegradable polymer microspheres that slowly release the peptide as they degrade, implantable osmotic pumps that deliver a steady dose over months, and peptide-polymer conjugates designed for extended circulation. Roche's Phase 1 data on a long-acting subcutaneous GLP-1R agonist showed sustained drug levels over 4 weeks from a single injection. If depot formulations can match the efficacy and tolerability of weekly injectables while reducing injection frequency to monthly or quarterly, patient adherence rates could improve substantially.
Central Nervous System-Penetrant Formulations
Given that much of GLP-1's weight loss efficacy depends on central nervous system actions, enhancing brain penetration could improve efficacy without increasing peripheral side effects. Current GLP-1R agonists access the brain primarily through the area postrema (which lacks a blood-brain barrier) and through limited transport across the blood-brain barrier (BBB) at the median eminence and other circumventricular organs.
Research into BBB-permeable GLP-1R agonists, potentially through conjugation with transferrin receptor-targeting antibodies or nanoparticle encapsulation, could enable greater hypothalamic and mesolimbic receptor activation at lower systemic doses. This approach could theoretically increase appetite suppression while reducing the GI side effects that are largely driven by peripheral GLP-1R activation in the gut. However, enhanced brain penetration also raises concerns about neuropsychiatric effects, and careful safety evaluation would be essential.
Precision Obesity Medicine
As pharmacogenomic research advances and multiple effective anti-obesity agents become available, the field is moving toward a precision medicine approach. Rather than treating all patients with the same first-line agent, clinicians may eventually select specific GLP-1R agonists (or combination therapies) based on a patient's genetic profile, metabolic phenotype, and comorbidity pattern.
For example, a patient with prominent hedonic eating behaviors (strong food cravings, emotional eating, binge eating tendencies) might benefit most from an agent with strong reward circuit effects, while a patient with primarily metabolic dysfunction (insulin resistance, fatty liver, dyslipidemia) might respond best to a dual GLP-1/glucagon agonist like survodutide that directly targets hepatic metabolism. A patient with both obesity and type 2 diabetes might do best with tirzepatide, which has the strongest glycemic efficacy among current multi-receptor agonists.
Digital biomarkers and wearable technology may also play a growing role. Continuous glucose monitors can identify patients with marked postprandial glucose excursions who might benefit most from the glucose-regulatory effects of GLP-1 therapy. Activity trackers can monitor exercise adherence, which influences body composition outcomes. And app-based dietary tracking can help identify specific eating patterns that predict treatment response.
The integration of pharmacotherapy with technology-enabled behavioral support represents the future of obesity treatment. GLP-1R agonists address the biological drivers of obesity, while digital tools and behavioral coaching address the environmental and psychological factors. This combined approach has the potential to produce more complete, more sustainable weight management than either strategy alone. To explore how these emerging approaches might fit your needs, start with our personalized assessment.
Monitoring, Safety, and Long-Term Considerations During GLP-1 Weight Loss Therapy
Losing 15-25% of body weight is a significant physiological event, regardless of how it's achieved. GLP-1 receptor agonist therapy requires thoughtful monitoring to ensure the weight loss is progressing safely and that metabolic improvements are tracking alongside the scale changes.
Baseline and Ongoing Laboratory Monitoring
Before starting GLP-1 therapy for weight loss, a comprehensive metabolic panel establishes the baseline against which improvements (and potential issues) will be measured. Essential baseline labs include:
- Complete metabolic panel: Fasting glucose, HbA1c, lipid panel (total cholesterol, LDL, HDL, triglycerides), liver function tests (ALT, AST, GGT, alkaline phosphatase), kidney function (creatinine, eGFR, BUN), and electrolytes. These should be repeated at 3-month intervals during active weight loss and at 6-month intervals during maintenance.
- Thyroid function: TSH and free T4 at baseline. GLP-1R agonists carry a boxed warning regarding medullary thyroid carcinoma based on rodent studies (though no increased risk has been confirmed in humans). Additionally, significant weight loss can alter thyroid hormone binding and require dose adjustments in patients on levothyroxine.
- Pancreatic enzymes: Lipase and amylase at baseline. While pancreatitis is rare with GLP-1 therapy (incidence approximately 0.1-0.3%), monitoring these enzymes provides a reference point for evaluation of any abdominal pain during treatment.
- Nutritional markers: Vitamin D, B12, iron studies (ferritin, iron, TIBC), and folate. Reduced food intake during GLP-1 therapy can expose or worsen pre-existing nutritional deficiencies. Vitamin D deficiency is particularly common in obesity, and levels should be checked and supplemented as needed throughout treatment.
- Body composition: While not technically a lab test, DEXA scans at baseline and 6-12 month intervals provide the most accurate assessment of fat mass and lean mass changes. This is particularly valuable for detecting excessive lean mass loss that might warrant intervention (increased protein intake, resistance training modifications, or addition of muscle-preserving agents).
Gastrointestinal Safety and Management
Nausea, vomiting, diarrhea, and constipation are the most common side effects of GLP-1 therapy, affecting 20-45% of patients depending on the specific agent and dose. While these are typically mild to moderate and self-limiting, severe or persistent GI symptoms require careful management.
Nausea management follows a stepwise approach. First-line measures include eating smaller, more frequent meals; avoiding high-fat foods that delay gastric emptying further; staying upright for 30-60 minutes after eating; and staying well-hydrated. If nausea persists, slowing the dose escalation schedule (extending each step from 4 to 6-8 weeks) often helps. For moderate-severe nausea refractory to these measures, ondansetron (4-8 mg before meals) or ginger supplementation (250 mg four times daily) can provide additional relief. Our comprehensive GLP-1 resource center covers detailed side effect management strategies.
Gastroparesis is a concern that has received significant media attention. GLP-1R agonists delay gastric emptying by 20-40% in most patients, and in rare cases, this delay can be more severe, leading to symptoms of gastroparesis including severe nausea, vomiting, early satiety, and abdominal distension. Pre-existing gastroparesis (from diabetes or other causes) is generally considered a relative contraindication to GLP-1 therapy. For patients who develop gastroparesis symptoms during treatment, dose reduction or discontinuation is typically necessary, with symptoms resolving within 2-4 weeks of stopping the medication.
Gallbladder Disease Risk
Rapid weight loss from any cause increases the risk of gallstone formation because cholesterol supersaturation of bile increases as fat stores are mobilized. GLP-1 therapy compounds this risk through its direct effects on gallbladder motility: GLP-1R activation reduces gallbladder contractility, potentially promoting bile stasis and stone formation.
In the STEP trials, cholelithiasis (gallstones) occurred in 1.5-2.6% of semaglutide-treated participants versus 0.7-1.0% of placebo participants, with cholecystitis (gallbladder inflammation) occurring in 0.4-0.6% versus 0.2%. These rates are comparable to the gallstone risk seen with other forms of rapid weight loss, including very low-calorie diets and bariatric surgery.
Risk mitigation strategies include ensuring adequate fat intake (at least 7-10 grams per meal) to stimulate regular gallbladder contraction, maintaining adequate hydration, and avoiding extremely rapid weight loss (more than 1.5 kg per week consistently). Patients with a history of gallstones or cholecystitis should have baseline gallbladder imaging and may benefit from prophylactic ursodeoxycholic acid (300 mg twice daily) during the active weight loss phase.
Psychological and Behavioral Monitoring
The psychological dimensions of rapid, substantial weight loss are often underappreciated. While most patients experience improved mood, self-esteem, and quality of life during GLP-1 therapy, some individuals develop challenging psychological responses that warrant monitoring and support.
"Identity disruption" is increasingly reported by patients who lose 20% or more of their body weight. Rapid body changes can challenge self-image, alter social dynamics, and create anxiety about maintaining the new weight. Patients who have been overweight since childhood may have built their identity, relationships, and coping mechanisms around their body size, and sudden change can feel destabilizing even when the change is desired.
The reduction in food reward signaling can also be psychologically complex. For patients who have used eating as a primary coping mechanism for stress, anxiety, or depression, the pharmacological dampening of food's emotional reward can leave a void in their coping toolkit. This "transfer" phenomenon, well-documented after bariatric surgery, can manifest as increased alcohol consumption, compulsive shopping, or other substitute behaviors. Screening for these behaviors during follow-up visits and proactive referral to behavioral health support when needed is an important aspect of comprehensive GLP-1 therapy management.
For patients interested in peptide-based support for mood and cognitive function during weight loss, compounds like Semax and Selank have been studied for their nootropic and anxiolytic properties, respectively, and may provide complementary support, though clinical evidence for their use alongside GLP-1 therapy specifically is limited.
Drug Interactions and Absorption Considerations
GLP-1R agonists' effects on gastric emptying can alter the absorption kinetics of concurrently administered oral medications. This is particularly relevant for medications with narrow therapeutic indices or time-sensitive absorption requirements.
Oral contraceptives, as mentioned previously, may have altered absorption patterns. Levothyroxine, which requires an empty stomach and is sensitive to gastric pH and transit time, may need dose adjustment during GLP-1 therapy; TSH should be monitored more frequently (every 6-8 weeks) during the initial months of treatment and after dose changes. Warfarin, with its narrow therapeutic window, requires increased INR monitoring during GLP-1 initiation and dose escalation. Certain antibiotics with time-dependent killing (like amoxicillin) may have altered peak concentrations, though this is unlikely to be clinically significant for most infections.
Medications for diabetes require specific attention. Insulin doses often need to be reduced by 20-30% at the initiation of GLP-1 therapy to prevent hypoglycemia, with further reductions as weight loss progresses and insulin sensitivity improves. Sulfonylureas should ideally be discontinued or reduced before starting GLP-1 therapy due to the additive hypoglycemia risk. Metformin is generally safe to continue and may provide complementary metabolic benefits.
Patients taking multiple medications should have their drug regimen reviewed by a pharmacist at GLP-1 therapy initiation, with particular attention to medications requiring precise absorption timing. In some cases, switching from oral to parenteral formulations of critical medications may be warranted during the active dose-escalation phase of GLP-1 therapy when gastric emptying effects are most pronounced.
The Gut Microbiome and GLP-1 Weight Loss Mechanisms
The trillions of bacteria inhabiting the human gastrointestinal tract don't just passively observe GLP-1 therapy. They actively influence how GLP-1 receptor agonists work, and GLP-1 therapy, in turn, reshapes the microbiome in ways that may contribute to both the metabolic benefits and side effects of treatment.
Microbiome Influences on Endogenous GLP-1 Secretion
Before considering how exogenous GLP-1R agonists interact with the microbiome, it's worth understanding that gut bacteria are already major regulators of endogenous GLP-1 production. Intestinal L-cells, the specialized enteroendocrine cells that produce native GLP-1, are equipped with receptors that sense bacterial metabolites including short-chain fatty acids (SCFAs), secondary bile acids, and indole derivatives.
SCFAs, particularly butyrate and propionate produced by bacterial fermentation of dietary fiber, bind to free fatty acid receptors (FFAR2 and FFAR3) on L-cells, directly stimulating GLP-1 secretion. This is one reason why high-fiber diets improve glucose tolerance and satiety: the fiber provides substrate for SCFA-producing bacteria, which in turn stimulate GLP-1 release. Conversely, the reduced microbial diversity and lower SCFA production characteristic of obesity may contribute to impaired endogenous GLP-1 secretion, creating a metabolic vicious cycle.
Secondary bile acids, produced by bacterial modification of primary bile acids, activate the bile acid receptor TGR5 on L-cells, providing another microbiome-dependent stimulus for GLP-1 secretion. The bile acid-TGR5 axis is increasingly recognized as a key mediator of the metabolic benefits of bariatric surgery, where surgical rearrangement of the GI tract dramatically alters bile acid circulation and microbial exposure.
How GLP-1 Therapy Changes the Microbiome
GLP-1R agonist treatment produces measurable changes in gut microbiome composition within weeks of starting therapy. Several studies using 16S rRNA sequencing and metagenomic analysis have identified consistent shifts:
- Increased Akkermansia muciniphila: This mucin-degrading bacterium is consistently associated with metabolic health and leanness. Its relative abundance increases 2-4 fold during semaglutide and liraglutide treatment. Akkermansia strengthens the intestinal mucus layer, reduces bacterial translocation, and may independently improve insulin sensitivity.
- Increased Bacteroidetes-to-Firmicutes ratio: Obesity is associated with a relatively lower proportion of Bacteroidetes and higher proportion of Firmicutes. GLP-1 therapy tends to shift this ratio toward the "lean" pattern, likely through a combination of altered dietary intake (reduced calorie consumption), changed gut transit time, and altered luminal pH.
- Reduced Ruminococcus and Clostridium: Certain species within these genera are associated with increased energy harvest from food (extracting more calories from dietary fiber and resistant starch). Their reduction during GLP-1 therapy may contribute to a modest reduction in caloric extraction efficiency, complementing the appetite-suppressive effects.
- Increased microbial diversity: Alpha diversity (the variety of species within an individual's microbiome) tends to increase during GLP-1 therapy and weight loss. Greater microbial diversity is generally associated with better metabolic health and greater resilience to perturbation.
These microbiome changes are likely driven by the combination of reduced food intake, altered gastric emptying and intestinal transit time, and changes in bile acid composition during GLP-1 therapy. Some changes may also be mediated by direct GLP-1R activation on intestinal epithelial cells and enteric neurons, which influence the secretion of antimicrobial peptides and mucins that shape the microbial environment.
Microbiome Contributions to GLP-1 Side Effects
The GI side effects of GLP-1 therapy (nausea, bloating, constipation, diarrhea) may be partly microbiome-mediated. Altered gastric emptying and intestinal transit change the microenvironment in ways that affect bacterial fermentation patterns. Slower transit allows more complete bacterial fermentation of residual food, potentially increasing gas production and bloating. Changes in SCFA ratios (particularly increased propionate and butyrate production from prolonged fermentation time) may contribute to altered bowel habits.
Individual differences in baseline microbiome composition may partly explain the variability in GI side effect severity. Patients with higher baseline counts of gas-producing bacteria (Methanobrevibacter smithii, certain Clostridium species) may be more susceptible to bloating and flatulence during GLP-1 therapy. This hypothesis has not been formally tested in clinical trials but represents an active area of investigation.
Probiotics targeting specific bacterial populations may help mitigate GI side effects. While clinical evidence for probiotic use specifically alongside GLP-1 therapy is limited, general evidence supports strains like Lactobacillus rhamnosus GG and Saccharomyces boulardii for reducing antibiotic-associated and medication-associated GI symptoms. The rationale for their use during GLP-1 therapy is to maintain beneficial bacterial populations that support normal bowel function during the transition to a new dietary pattern and altered gut transit.
Microbiome as a Predictor of Treatment Response
An exciting frontier is the use of baseline microbiome profiling to predict individual responses to GLP-1 therapy. Preliminary data suggest that patients with higher baseline Prevotella and lower Bacteroides abundance tend to achieve greater weight loss with GLP-1R agonists, possibly because these microbial profiles are associated with greater sensitivity to the appetite-suppressive effects of SCFAs produced during weight loss.
If validated in larger studies, microbiome profiling could become a component of the precision medicine approach to obesity treatment. A patient's microbial profile might help predict not only the magnitude of weight loss but also the likelihood and severity of GI side effects, allowing for preemptive strategies (probiotic supplementation, dietary fiber adjustment, slower dose titration) to improve tolerability and adherence.
The intersection of microbiome science with GLP-1 pharmacology represents one of the most promising areas for improving the real-world effectiveness of obesity treatment. For more on personalized treatment approaches, see our free assessment and lifestyle research hub.
Metabolic Adaptation During GLP-1 Weight Loss: What Actually Happens to Your Metabolism
One of the most feared consequences of weight loss is metabolic adaptation - the disproportionate decline in energy expenditure that makes continued weight loss progressively harder and weight regain almost inevitable. Understanding how GLP-1 therapy interacts with metabolic adaptation is critical for long-term success.
The Biology of Metabolic Adaptation
Metabolic adaptation (sometimes called "adaptive thermogenesis") refers to the reduction in energy expenditure that exceeds what would be predicted from the loss of metabolically active tissue. When you lose weight through any means, your resting metabolic rate (RMR) declines partly because you have less tissue to maintain (a smaller body burns fewer calories). But RMR often declines more than the tissue loss would predict, sometimes by 100-300 kcal/day beyond what body composition changes explain.
This "extra" metabolic decline is driven by hormonal and neural adaptations: thyroid hormone (T3) levels decrease, reducing the metabolic rate of individual cells; sympathetic nervous system activity declines, reducing basal catecholamine-driven thermogenesis; and leptin levels fall in proportion to fat mass loss, which reduces both RMR and physical activity-related energy expenditure through central mechanisms. These adaptations are deeply conserved evolutionary responses designed to defend against starvation and are remarkably resistant to behavioral override.
The famous "Biggest Loser" study (Fothergill et al., 2016) demonstrated that participants who lost an average of 58 kg through aggressive diet and exercise showed metabolic adaptation of approximately 500 kcal/day that persisted for at least 6 years after the competition. This persistent metabolic penalty, combined with the hormone-driven appetite increase, explains why nearly all participants eventually regained most of their lost weight.
Does GLP-1 Therapy Mitigate Metabolic Adaptation?
The evidence on whether GLP-1R agonists protect against metabolic adaptation is nuanced and still evolving. Several lines of evidence suggest a partial protective effect:
Indirect calorimetry data from GLP-1R agonist trials consistently show that RMR declines during treatment, confirming that metabolic adaptation occurs. However, the magnitude of adaptation appears smaller than expected from the degree of weight loss, particularly when compared to diet-only weight loss producing similar amounts of weight loss. In a substudy of the STEP 1 trial, RMR declined by approximately 200 kcal/day with 15% weight loss on semaglutide, whereas diet-only studies producing 15% weight loss typically show RMR declines of 300-400 kcal/day.
This partial protection may be mediated through GLP-1R effects on thyroid hormone metabolism and sympathetic nervous system activity. Some data suggest that GLP-1R agonists attenuate the decline in T3 levels during weight loss, though not completely. The mechanism could involve GLP-1R activation on hypothalamic TRH (thyrotropin-releasing hormone) neurons, which regulate the hypothalamic-pituitary-thyroid axis.
Multi-agonist approaches may offer better protection against metabolic adaptation. Tirzepatide's GIP receptor agonism has been associated with less metabolic adaptation in preclinical models, possibly through GIP-mediated effects on adipose tissue thermogenesis. Glucagon-containing agonists like survodutide and retatrutide may provide the strongest protection through glucagon-mediated increases in hepatic energy expenditure and thermogenesis, directly counteracting the metabolic slowdown.
For individuals seeking to further counteract metabolic adaptation during GLP-1 therapy, complementary approaches include resistance training (which preserves metabolically active lean mass), adequate protein intake (1.2-1.6 g/kg/day to support muscle maintenance), and potentially metabolically activating peptides like MOTS-c (which enhances mitochondrial function through AMPK activation) or 5-Amino-1MQ (which increases cellular energy expenditure through NNMT inhibition). These complementary approaches target the metabolic side of the energy balance equation that GLP-1 therapy primarily addresses through the intake side. Visit our peptide research hub for more on metabolic optimization strategies.
Psychological and Behavioral Dimensions of GLP-1-Mediated Weight Loss
The biological mechanisms of GLP-1 weight loss, from hypothalamic appetite regulation to gastric emptying modulation, operate within a psychological and behavioral context that profoundly shapes treatment outcomes. Understanding how GLP-1 agonists interact with the psychology of eating behavior, body image, and long-term behavioral change adds a dimension that purely mechanistic analyses often miss.
Food Noise and Cognitive Liberation
One of the most consistently reported subjective experiences among patients starting GLP-1 agonist therapy is the reduction in what patients colloquially call "food noise," the persistent, intrusive thoughts about food that dominate the mental field of many people with obesity. This phenomenon reflects the tonic activation of reward-anticipation circuits in the brain that drive food-seeking behavior even in the absence of physiological hunger.
GLP-1 agonists appear to quiet this neural chatter by modulating dopaminergic signaling in the mesolimbic reward system. Patients describe the experience not as willpower or deprivation but as a fundamental shift in their relationship with food. The constant background hum of food desire, the planning of the next meal while still eating the current one, the inability to pass a restaurant or bakery without an internal negotiation, these patterns simply diminish or disappear for many patients on GLP-1 therapy.
This cognitive liberation has effects that extend well beyond caloric intake. Patients report improved concentration at work, reduced anxiety, better sleep (not lying awake thinking about food), and a sense of mental freedom that many describe as the most valuable aspect of treatment. For patients who have spent years or decades locked in a adversarial relationship with food, the sudden absence of food-related cognitive intrusion can be genuinely life-changing.
The mechanism behind this subjective experience maps to the reward circuit modulation discussed in earlier sections. GLP-1 receptors in the ventral tegmental area (VTA), nucleus accumbens, and prefrontal cortex modulate the dopaminergic projections that encode reward anticipation and motivational salience. By reducing the dopaminergic response to food cues, GLP-1 agonists lower the motivational priority that the brain assigns to food-seeking, freeing cognitive resources for other activities. This is not suppression of pleasure (patients still enjoy food when they eat) but reduction of the anticipatory drive that makes food dominate attention even when the person is not eating.
Emotional Eating and Stress Response
Emotional eating, the use of food to manage negative emotions like stress, anxiety, boredom, or sadness, is a major driver of weight gain and a significant barrier to weight loss maintenance. GLP-1 agonists affect emotional eating through multiple pathways. The direct modulation of reward circuits reduces the reinforcing value of comfort food. The reduction in food noise diminishes the conditioned association between negative emotions and eating. And some evidence suggests that GLP-1 receptor activation in the amygdala and prefrontal cortex may modulate the emotional response itself, not just the food-seeking behavior it triggers.
However, some patients experience a paradoxical challenge when GLP-1 therapy removes their primary coping mechanism. Individuals who have relied on food for emotional regulation for years may find themselves without a familiar coping strategy, leading to increased anxiety, irritability, or depressed mood in the early weeks of treatment. This phenomenon is analogous to the emotional upheaval sometimes seen in early sobriety from alcohol or other substances, and it highlights the importance of psychological support during GLP-1 therapy initiation.
Cognitive behavioral therapy (CBT) and acceptance and commitment therapy (ACT) can help patients develop alternative emotional regulation strategies that replace the food-based coping they are losing. Mindfulness-based eating awareness training (MB-EAT) is particularly well-suited as an adjunct to GLP-1 therapy because it helps patients develop a more conscious, intentional relationship with eating that takes advantage of the reduced food noise and altered reward signaling that GLP-1 agonists provide.
Body Image, Identity, and the Psychology of Transformation
Rapid weight loss, whether from semaglutide, tirzepatide, or any other intervention, creates a psychological challenge that is often underappreciated: the disconnect between the changing body and the established self-concept. Patients who have been obese for decades have built their identity, social relationships, clothing habits, movement patterns, and self-image around a larger body. Losing 15-20% of body weight over 12-18 months requires a fundamental reorganization of self-concept that can be disorienting even when the weight loss is desired and medically beneficial.
Some patients experience what psychologists call "phantom fat," the persistent perception of being larger than they actually are, similar to the phantom limb phenomenon after amputation. This perceptual lag can persist for months or years after weight loss and may contribute to body dysmorphia, continued avoidance of activities associated with thinness (swimming, form-fitting clothing, intimate relationships), and a persistent sense that the new body doesn't belong to them.
Others face social complications. Relationships built around shared eating experiences may become strained. Partners who were comfortable with the patient's larger body may feel threatened by the transformation. Friends and family members who are themselves struggling with weight may react with jealousy, skepticism, or passive-aggressive commentary about the "easy way out" of pharmaceutical weight loss. These social dynamics can undermine treatment adherence if not anticipated and addressed.
The most psychologically successful patients tend to be those who use GLP-1 therapy as a platform for comprehensive lifestyle transformation rather than as a standalone weight loss tool. When the reduced appetite and food noise provide cognitive and motivational space for developing new habits, trying new activities, building new social connections, and rebuilding self-image, the psychological benefits compound on top of the metabolic benefits. Complementary approaches like MOTS-c for exercise performance and BPC-157 for recovery from new physical activities can support this broader lifestyle transformation. The GLP-1 research hub covers integrated approaches to weight management that address both the biological and psychological dimensions of this process.
Exercise Optimization During GLP-1 Weight Loss: Maximizing Results and Preserving Lean Mass
The interaction between GLP-1 agonist therapy and exercise is bidirectional and clinically significant. Exercise amplifies the weight loss and metabolic benefits of GLP-1 therapy, while GLP-1 therapy can alter the physiological response to exercise in ways that matter for exercise programming and performance. Understanding this interaction helps patients and clinicians design exercise protocols that maximize the therapeutic benefit of combined pharmacological and physical intervention.
Resistance Training: The Non-Negotiable Component
Resistance training during GLP-1-mediated weight loss is not optional; it is the single most effective intervention for preserving lean body mass during caloric deficit. Without structured resistance exercise, approximately 25-35% of total weight loss consists of lean mass (primarily skeletal muscle), a ratio that worsens with greater weight loss magnitude and faster weight loss rate. With regular progressive resistance training, this ratio can be shifted to 10-20% lean mass loss, dramatically improving the metabolic and functional quality of the weight loss achieved.
The physiological basis for this protective effect involves mechanotransduction, the process by which mechanical loading of muscle tissue activates cellular signaling pathways (primarily mTOR) that stimulate muscle protein synthesis and inhibit protein breakdown. During caloric deficit, the body's default catabolic state favors muscle protein breakdown for gluconeogenesis and energy production. Resistance exercise provides a powerful counter-signal that overrides this catabolic default in exercised muscles, directing the body to preferentially break down fat tissue rather than muscle for energy.
A practical minimum effective dose for lean mass preservation is 2-3 resistance training sessions per week, targeting all major muscle groups, using loads heavy enough to approach muscular failure within 8-15 repetitions per set. Progressive overload, the systematic increase in training volume or intensity over time, maintains the mechanical stimulus as the body adapts. Patients new to resistance training should work with a qualified trainer to establish proper form and develop a sustainable progressive program.
Cardiovascular Exercise Considerations
Cardiovascular exercise (walking, cycling, swimming, jogging) contributes to the caloric deficit that drives weight loss and provides independent cardiovascular health benefits. However, excessive cardiovascular exercise during GLP-1-mediated weight loss can exacerbate lean mass loss, particularly if protein intake is inadequate. The catabolic demands of prolonged endurance exercise, combined with the caloric deficit from both reduced intake and increased expenditure, can create a protein-catabolic environment that overwhelms the body's ability to preserve muscle mass.
The optimal approach balances cardiovascular exercise for health benefits with appropriate limits to prevent excessive catabolic stress. Current guidelines recommend 150-300 minutes per week of moderate-intensity cardiovascular exercise (equivalent to brisk walking) or 75-150 minutes of vigorous-intensity exercise. During active GLP-1-mediated weight loss, staying toward the lower end of these ranges and prioritizing resistance training for the remaining exercise sessions is generally advisable.
For patients whose exercise tolerance is limited by body habitus or joint pain, low-impact options like swimming, cycling, elliptical training, or water aerobics provide cardiovascular benefits with reduced musculoskeletal stress. As weight loss progresses and load-bearing becomes more comfortable, gradual transition to weight-bearing exercise (walking, hiking, jogging) is beneficial for bone health and functional capacity.
Protein Timing and Exercise Recovery
Protein intake of 1.2-1.6 g/kg of ideal body weight per day provides the amino acid substrate needed for muscle protein synthesis in response to resistance training. The timing of protein intake relative to exercise matters: consuming 20-40 g of high-quality protein (containing at least 2-3 g of leucine) within 2 hours of resistance training maximizes the muscle protein synthesis response. This post-exercise protein bolus becomes particularly important during GLP-1 therapy because the appetite suppression may cause patients to skip meals or reduce protein intake below optimal levels.
For patients struggling to meet protein requirements due to GLP-1-induced appetite suppression, liquid protein sources (protein shakes, Greek yogurt, milk) are often better tolerated than solid protein foods. The reduced gastric emptying from GLP-1 therapy can make large solid meals uncomfortable, but liquid calories generally pass through the stomach more easily and can be consumed in smaller volumes with adequate protein content.
Complementary peptides can support the exercise-weight loss program through independent mechanisms. CJC-1295/Ipamorelin enhances post-exercise growth hormone release, supporting both muscle recovery and fat metabolism. BPC-157 accelerates recovery from exercise-induced tissue stress, particularly in tendons and joints that may be adapting to new exercise demands. TB-500 promotes tissue repair and reduces post-exercise inflammation through thymosin beta-4-mediated mechanisms. These supportive peptides operate through pathways independent of the GLP-1 system and can be used alongside semaglutide or tirzepatide without pharmacological interaction concerns. The dosing calculator helps determine appropriate protocols for these complementary agents.
Sleep Architecture Changes During GLP-1-Mediated Weight Loss
An emerging area of research concerns how GLP-1 receptor agonists affect sleep quality and sleep architecture, and how these changes in turn influence the weight loss trajectory. Sleep and metabolism are bidirectionally linked in ways that make this question clinically significant. Poor sleep quality promotes weight gain through elevated ghrelin, reduced leptin, increased cortisol, and impaired glucose tolerance. Conversely, weight loss, particularly the reduction of visceral adipose tissue, tends to improve sleep quality by reducing obstructive sleep apnea severity and decreasing systemic inflammation that disrupts sleep continuity.
Patients on semaglutide and tirzepatide frequently report subjective improvements in sleep quality within the first few weeks of treatment, often before significant weight loss has occurred. This observation suggests that GLP-1 receptor agonists may have direct effects on sleep-regulating circuits independent of weight change. GLP-1 receptors are expressed in brain regions involved in sleep-wake regulation, including the lateral hypothalamus (which contains orexin neurons critical for wakefulness) and the parabrachial nucleus (which modulates arousal). Preclinical studies in rodents have shown that central GLP-1 receptor activation reduces wakefulness and increases non-REM sleep duration, potentially by modulating orexin neuron activity.
The clinical implications are substantial. Patients who sleep better during GLP-1 therapy may experience amplified weight loss benefits because adequate sleep supports the metabolic and behavioral conditions favorable to fat loss. Well-rested individuals make better food choices, have more energy for physical activity, and show improved insulin sensitivity compared to sleep-deprived counterparts. This creates a potential positive feedback loop where GLP-1-mediated sleep improvement enhances the weight loss that further improves sleep, compounding the therapeutic benefit over time. Clinicians should consider asking about sleep quality at follow-up visits, as improvements in sleep can serve as an early indicator of treatment response and patient adherence.
Frequently Asked Questions
How exactly do GLP-1 drugs suppress appetite?
GLP-1 receptor agonists suppress appetite through multiple overlapping mechanisms. They activate receptors in the brainstem's nucleus tractus solitarius and area postrema, which receive and process satiety signals from the gut. They also act on the hypothalamus, where they stimulate appetite-suppressing POMC neurons and inhibit hunger-promoting NPY/AgRP neurons. Additionally, they modulate the brain's reward circuitry, reducing the pleasure response to food. This multi-level approach produces more consistent and powerful appetite suppression than drugs targeting a single pathway. Patients typically notice reduced hunger within days of starting treatment, with effects deepening over the first few weeks as dose escalation progresses.
Do GLP-1 drugs slow down digestion?
Yes. GLP-1 receptor agonists delay gastric emptying by 20-40%, meaning food stays in the stomach longer after meals. This produces a prolonged feeling of fullness that reinforces the central appetite suppression. The gastric slowing effect is strongest during the first few weeks of treatment and partially attenuates over time through a process called tachyphylaxis. This partial adaptation is actually beneficial because it reduces GI side effects (nausea, bloating) while the more sustained brain-mediated appetite effects continue. If you're having a medical procedure requiring anesthesia, discuss your GLP-1 medication timing with your anesthesiologist, as retained stomach contents can pose aspiration risk.
Why do people on GLP-1 drugs stop craving junk food?
GLP-1 receptors are present in the brain's reward circuitry, specifically the ventral tegmental area and nucleus accumbens. These areas use dopamine to assign "reward value" to experiences, including eating highly palatable foods. When GLP-1 agonists activate receptors here, they dampen the dopamine response to food cues, making calorie-dense and ultra-processed foods less appealing. Brain imaging studies confirm this: patients on semaglutide show reduced neural activation in reward centers when viewing images of pizza, cake, or fried food. This isn't nausea-driven avoidance; it's a genuine reduction in how rewarding these foods feel.
How much of the weight lost on GLP-1 therapy is muscle?
Clinical trial data show that approximately 25-40% of weight lost on GLP-1 therapy comes from lean mass (which includes muscle, bone, and water). In the STEP 1 trial, about 32% of weight lost with semaglutide was lean mass; in SURMOUNT-1, about 27% with tirzepatide 15 mg. These ratios are similar to what's seen with diet-induced weight loss. The absolute amount of lean mass lost can be significant given the large total weight reductions these drugs produce. Resistance training and adequate protein intake (1.2-1.6 g/kg/day) can substantially reduce lean mass losses. If preserving muscle is a priority for you, a structured exercise program alongside your GLP-1 therapy is strongly recommended.
Will I regain weight if I stop taking a GLP-1 drug?
Most likely, yes. The STEP 4 trial showed that participants who discontinued semaglutide after 20 weeks regained approximately two-thirds of their lost weight over the following year. STEP 1 follow-up data showed similar regain patterns. This occurs because the biological drives promoting weight regain (increased hunger hormones, decreased satiety signals, metabolic adaptation) reassert themselves once the drug is no longer providing counter-regulation. This doesn't mean the treatment was worthless; it means obesity is a chronic condition that typically requires ongoing management, similar to hypertension requiring continued blood pressure medication.
Do GLP-1 drugs affect metabolism or calorie burning?
The effect on energy expenditure is modest and somewhat debated. Weight loss from any cause reduces resting metabolic rate through metabolic adaptation. GLP-1 agonists don't fully prevent this, but some evidence suggests they may partially mitigate it. Rodent data show increased brown fat thermogenesis with GLP-1 receptor activation. Human data are mixed, with some studies showing better-than-expected metabolic rate preservation and others showing typical adaptation. The primary mechanism of weight loss remains reduced caloric intake, not increased calorie burning. Newer triple agonists like retatrutide may have stronger energy expenditure effects through their glucagon receptor activity.
How do GLP-1 drugs differ from older weight loss medications?
Previous weight loss drugs targeted one or two mechanisms. Phentermine stimulates norepinephrine to suppress appetite. Orlistat blocks fat absorption. Naltrexone-bupropion modulates opioid and dopamine signaling. GLP-1 receptor agonists simultaneously engage at least five distinct pathways: central appetite suppression via brainstem and hypothalamus, gastric emptying delay, food reward reduction, metabolic improvement, and possibly enhanced energy expenditure. This multi-pathway approach produces 2-3 times greater weight loss than any previous medication. It's the difference between turning down one radio station and turning down the entire sound system.
Is the weight loss from GLP-1 drugs mostly from fat or from water?
Predominantly from fat. DEXA body composition scans in clinical trials confirm that 60-75% of weight lost is fat mass, with visceral fat (the metabolically dangerous deep belly fat) showing particularly large reductions of 20-25%. The initial weight loss in the first 1-2 weeks may include some water loss due to reduced glycogen stores (glycogen holds water in a roughly 1:3 ratio). After this initial phase, ongoing weight loss is primarily from adipose tissue. Some lean mass is also lost (25-40% of total), which includes water, muscle, and connective tissue. This is why body composition tracking with DEXA or bioimpedance is more informative than scale weight alone.
Can GLP-1 drugs help with alcohol cravings?
Emerging evidence suggests yes, though clinical trials specifically for alcohol use disorder are still ongoing. GLP-1 receptors in the brain's reward circuit (VTA and nucleus accumbens) modulate dopamine responses to all rewarding stimuli, not just food. Preclinical studies consistently show that GLP-1 agonists reduce alcohol consumption in animal models. Human observational data from FDA adverse event databases show lower-than-expected rates of alcohol-related diagnoses among GLP-1 users. Many patients report spontaneously drinking less, describing the same reduction in "wanting" they experience with food. Formal clinical trials are underway, and results are expected in 2026-2027.
How long does it take for GLP-1 drugs to start working for weight loss?
Appetite suppression typically begins within 1-3 days of the first injection, though the effect strengthens as the dose is titrated upward over 4-16 weeks (depending on the specific drug). Measurable weight loss usually appears within the first 1-2 weeks. The rate of weight loss is typically fastest during the first 3-6 months, then gradually slows as a new equilibrium is approached. Maximum weight loss in clinical trials was reached around 60-72 weeks for semaglutide and tirzepatide. The slow dose escalation schedule used for all GLP-1 agonists is designed to minimize nausea while achieving effective appetite suppression.
Do GLP-1 drugs work for everyone?
Not equally. While the average weight loss in clinical trials is 15-22%, individual responses vary widely. Some patients lose 25-30% of their body weight; others lose less than 5%. Factors affecting response include genetic variation in GLP-1 receptor sensitivity, baseline metabolic health, adherence to the medication, lifestyle factors (diet quality, exercise), and possibly gut microbiome composition. Patients with certain genetic variants (like MC4R loss-of-function mutations) may show reduced but not absent response. If one GLP-1 compound doesn't produce adequate results after reaching the maximum dose, switching to a different compound or a dual-agonist like tirzepatide may help.
What's the difference between how semaglutide and tirzepatide cause weight loss?
Semaglutide activates the GLP-1 receptor alone. Tirzepatide activates both GLP-1 and GIP (glucose-dependent insulinotropic polypeptide) receptors. The GIP receptor adds an additional signaling pathway that enhances insulin sensitivity, may improve fat cell metabolism, and produces stronger suppression of glucagon. In clinical trials, tirzepatide produces roughly 5-7 percentage points more weight loss than semaglutide (22.5% vs 14.9-16.9% at maximum doses). The dual-receptor approach appears to create a more favorable body composition ratio, with slightly less lean mass loss relative to fat mass loss. Both drugs share the core GLP-1 mechanisms discussed in this report.
How much protein should I eat while on a GLP-1 drug?
Aim for 1.2-1.6 grams of protein per kilogram of your current body weight per day. For a 200-pound (91 kg) person, that's 109-146 grams daily. This amount supports muscle protein synthesis and helps preserve lean mass during the caloric deficit created by GLP-1 therapy. Distribute protein intake across 3-4 meals, targeting at least 25-30 grams per eating occasion. Since GLP-1 drugs reduce appetite and total food volume, you may need to prioritize protein-rich foods first at each meal and consider protein supplements (whey, casein, or plant-based shakes) to hit your targets. Combining adequate protein with resistance training provides the strongest protection against muscle loss.
Can GLP-1 drugs change my "set point" weight permanently?
Current evidence suggests they do not permanently reset the body's weight set point. The STEP 4 trial demonstrated that weight regain begins quickly after discontinuation, and appetite returns to pre-treatment levels within weeks. The body's weight defense system (driven by leptin, ghrelin, metabolic adaptation, and hypothalamic circuitry) appears to remember the higher weight and works to restore it once the pharmacological opposition is removed. However, some researchers hypothesize that very long-term treatment (5-10+ years) might eventually allow some degree of set point adjustment through sustained synaptic remodeling in the hypothalamus. This remains unproven. For now, GLP-1 therapy is best understood as ongoing management of a chronic condition.
References
- Drucker DJ. GLP-1 physiology informs the pharmacotherapy of obesity. Molecular Metabolism. 2022;57:101351. DOI: 10.1016/j.molmet.2021.101351
- Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity (STEP 1). New England Journal of Medicine. 2021;384(11):989-1002. DOI: 10.1056/NEJMoa2032183
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity (SURMOUNT-1). New England Journal of Medicine. 2022;387(3):205-216. DOI: 10.1056/NEJMoa2206038
- Turton MD, O'Shea D, Gunn I, et al. A role for glucagon-like peptide-1 in the central regulation of feeding. Nature. 1996;379(6560):69-72. DOI: 10.1038/379069a0
- Holt MK, Richards JE, Cook DR, et al. Preproglucagon neurons in the nucleus of the solitary tract are the main source of brain GLP-1. Molecular Metabolism. 2019;25:131-141. DOI: 10.1016/j.molmet.2019.04.010
- Secher A, Jelsing J, Baquero AF, et al. The arcuate nucleus mediates GLP-1 receptor agonist liraglutide-dependent weight loss. Journal of Clinical Investigation. 2014;124(10):4473-4488. DOI: 10.1172/JCI75276
- Gabery S, Salinas CG, Paulsen SJ, et al. Semaglutide lowers body weight in rodents via distributed neural pathways. JCI Insight. 2020;5(6):e133429. DOI: 10.1172/jci.insight.133429
- Kanoski SE, Fortin SM, Arnold M, et al. Peripheral and central GLP-1 receptor populations mediate the anorectic effects of peripherally administered GLP-1 receptor agonists, liraglutide and exendin-4. Endocrinology. 2011;152(8):3103-3112. DOI: 10.1210/en.2011-0174
- Sisley S, Gutierrez-Aguilar R, Scott M, et al. Neuronal GLP1R mediates liraglutide's anorectic but not glucose-lowering effect. Journal of Clinical Investigation. 2014;124(6):2456-2463. DOI: 10.1172/JCI72434
- Hayes MR, De Jonghe BC, Kanoski SE. Role of the glucagon-like-peptide-1 receptor in the control of energy balance. Physiology & Behavior. 2010;100(5):503-510. DOI: 10.1016/j.physbeh.2010.02.029
- Alhadeff AL, Rupprecht LE, Hayes MR. GLP-1 neurons in the nucleus of the solitary tract project directly to the ventral tegmental area and nucleus accumbens to control for food intake. Endocrinology. 2012;153(2):647-658. DOI: 10.1210/en.2011-1443
- Mietlicki-Baase EG, Ortinski PI, Rupprecht LE, et al. The food intake-suppressive effects of glucagon-like peptide-1 receptor signaling in the ventral tegmental area are mediated by AMPA/kainate receptors. American Journal of Physiology-Endocrinology and Metabolism. 2013;305(11):E1367-E1374. DOI: 10.1152/ajpendo.00413.2013
- Dossat AM, Lilly N, Kay K, Williams DL. Glucagon-like peptide 1 receptors in nucleus accumbens affect food intake. Journal of Neuroscience. 2011;31(41):14453-14457. DOI: 10.1523/JNEUROSCI.3262-11.2011
- van Bloemendaal L, IJzerman RG, Ten Kulve JS, et al. GLP-1 receptor activation modulates appetite- and reward-related brain areas in humans. Diabetes. 2014;63(12):4186-4196. DOI: 10.2337/db14-0849
- Ten Kulve JS, Veltman DJ, van Bloemendaal L, et al. Endogenous GLP-1 and GLP-1 analogue alter CNS responses to palatable food consumption. Journal of Endocrinology. 2016;229(1):1-12. DOI: 10.1530/JOE-15-0461
- Friedrichsen BN, Neubauer N, Lee YC, et al. Stimulation of pancreatic beta-cell replication by incretins involves transcriptional induction of cyclin D1 via multiple signalling pathways. Journal of Endocrinology. 2006;188(3):481-492. DOI: 10.1677/joe.1.06160
- Campbell JE, Drucker DJ. Pharmacology, physiology, and mechanisms of incretin hormone action. Cell Metabolism. 2013;17(6):819-837. DOI: 10.1016/j.cmet.2013.04.008
- Nauck MA, Meier JJ. Incretin hormones: their role in health and disease. Diabetes, Obesity and Metabolism. 2018;20(Suppl 1):5-21. DOI: 10.1111/dom.13129
- Rubino DM, Greenway FL, Khalid U, et al. Effect of continued weekly subcutaneous semaglutide vs placebo on weight loss maintenance (STEP 4). JAMA. 2021;325(14):1414-1425. DOI: 10.1001/jama.2021.3224
- Wilding JPH, Batterham RL, Davies M, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide (STEP 1 extension). Diabetes, Obesity and Metabolism. 2022;24(8):1553-1564. DOI: 10.1111/dom.14725
- Jastreboff AM, Kaplan LM, Frias JP, et al. Triple-hormone-receptor agonist retatrutide for obesity. New England Journal of Medicine. 2023;389(6):514-526. DOI: 10.1056/NEJMoa2301972
- Batterham RL, Cowley MA, Small CJ, et al. Gut hormone PYY3-36 physiologically inhibits food intake. Nature. 2002;418(6898):650-654. DOI: 10.1038/nature00887
- Cowley MA, Smart JL, Rubinstein M, et al. Leptin activates anorexigenic POMC neurons through a neural network in the arcuate nucleus. Nature. 2001;411(6836):480-484. DOI: 10.1038/35078085
- Farooqi IS, Keogh JM, Yeo GS, et al. Clinical spectrum of obesity and mutations in the melanocortin 4 receptor gene. New England Journal of Medicine. 2003;348(12):1085-1095. DOI: 10.1056/NEJMoa022050
- Volkow ND, Wang GJ, Tomasi D, Baler RD. The addictive dimensionality of obesity. Biological Psychiatry. 2013;73(9):811-818. DOI: 10.1016/j.biopsych.2012.12.020
- Erreger K, Davis AR, Poe AM, et al. Exendin-4 decreases amphetamine-induced locomotor activity. Physiology & Behavior. 2012;106(4):574-578. DOI: 10.1016/j.physbeh.2012.03.014
- Egecioglu E, Steensland P, Fredriksson I, et al. The glucagon-like peptide 1 analogue Exendin-4 attenuates alcohol mediated behaviors in rodents. Psychoneuroendocrinology. 2013;38(8):1259-1270. DOI: 10.1016/j.psyneuen.2012.11.009
- Shirazi RH, Dickson SL, Skibicka KP. Gut peptide GLP-1 and its analogue, Exendin-4, decrease alcohol intake and reward. PLoS ONE. 2013;8(4):e61965. DOI: 10.1371/journal.pone.0061965
- Nauck MA, Quast DR, Wefers J, Meier JJ. GLP-1 receptor agonists in the treatment of type 2 diabetes. Diabetes Research and Clinical Practice. 2021;175:108811. DOI: 10.1016/j.diabres.2021.108811
- Armstrong MJ, Gaunt P, Aithal GP, et al. Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN). Lancet. 2016;387(10019):679-690. DOI: 10.1016/S0140-6736(15)00803-X
- Newsome PN, Buchholtz K, Cusi K, et al. A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. New England Journal of Medicine. 2021;384(12):1113-1124. DOI: 10.1056/NEJMoa2028395
- Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes (SUSTAIN-6). New England Journal of Medicine. 2016;375(19):1834-1844. DOI: 10.1056/NEJMoa1607141
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes (SELECT). New England Journal of Medicine. 2023;389(24):2221-2232. DOI: 10.1056/NEJMoa2307563
- Mullally JA, Chung WK, LeDuc CA, et al. Response to melanocortin agonist therapy in patients with MC4R mutations. Obesity. 2022;30(5):1044-1052. DOI: 10.1002/oby.23400
- Leibel RL, Rosenbaum M, Hirsch J. Changes in energy expenditure resulting from altered body weight. New England Journal of Medicine. 1995;332(10):621-628. DOI: 10.1056/NEJM199503093321001
- Sumithran P, Prendergast LA, Delbridge E, et al. Long-term persistence of hormonal adaptations to weight loss. New England Journal of Medicine. 2011;365(17):1597-1604. DOI: 10.1056/NEJMoa1105816
- Rosenbaum M, Hirsch J, Gallagher DA, Leibel RL. Long-term persistence of adaptive thermogenesis in subjects who have maintained a reduced body weight. American Journal of Clinical Nutrition. 2008;88(4):906-912. DOI: 10.1093/ajcn/88.4.906
- Heymsfield SB, Coleman LA, Miller R, et al. Effect of bimagrumab vs placebo on body fat mass among adults with type 2 diabetes and obesity. JAMA Network Open. 2021;4(1):e2033457. DOI: 10.1001/jamanetworkopen.2020.33457
- Cava E, Yeat NC, Mittendorfer B. Preserving healthy muscle during weight loss. Advances in Nutrition. 2017;8(3):511-519. DOI: 10.3945/an.116.014506
- Sardeli AV, Komatsu TR, Mori MA, et al. Resistance training prevents muscle loss induced by caloric restriction in obese elderly individuals. Frontiers in Physiology. 2018;9:423. DOI: 10.3389/fphys.2018.00423
- Morton RW, Murphy KT, McKellar SR, et al. A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength. British Journal of Sports Medicine. 2018;52(6):376-384. DOI: 10.1136/bjsports-2017-097608
- Kreider RB, Kalman DS, Antonio J, et al. International Society of Sports Nutrition position stand: safety and efficacy of creatine supplementation. Journal of the International Society of Sports Nutrition. 2017;14:18. DOI: 10.1186/s12970-017-0173-z
- Hallberg SJ, Gershuni VM, Hazbun TL, Athinarayanan SJ. Reversing type 2 diabetes: a narrative review of the evidence. Nutrients. 2019;11(4):766. DOI: 10.3390/nu11040766
- Holst JJ. The physiology of glucagon-like peptide 1. Physiological Reviews. 2007;87(4):1409-1439. DOI: 10.1152/physrev.00034.2006
- Muller TD, Finan B, Bloom SR, et al. Glucagon-like peptide 1 (GLP-1). Molecular Metabolism. 2019;30:72-130. DOI: 10.1016/j.molmet.2019.09.010
- Krieger JP, Langhans W, Lee SJ. Novel role of GLP-1 receptor signaling in energy expenditure during chronic high fat diet feeding in rats. Physiology & Behavior. 2015;151:142-148. DOI: 10.1016/j.physbeh.2015.07.021
- Wadden TA, Bailey TS, Billings LK, et al. Effect of subcutaneous semaglutide vs placebo as an adjunct to intensive behavioral therapy on body weight (STEP 3). JAMA. 2021;325(14):1403-1413. DOI: 10.1001/jama.2021.1831
- Davies M, Faerch L, Jeppesen OK, et al. Semaglutide 2.4 mg once a week in adults with overweight or obesity and type 2 diabetes (STEP 2). Lancet. 2021;397(10278):971-984. DOI: 10.1016/S0140-6736(21)00213-0
- Garvey WT, Batterham RL, Bhatt DL, et al. Two-year effects of semaglutide in adults with overweight or obesity (STEP 5). Nature Medicine. 2022;28(10):2083-2091. DOI: 10.1038/s41591-022-02026-4
- Kadouh HC, Acosta A. Current paradigms in the etiology of obesity. Techniques in Gastrointestinal Endoscopy. 2017;19(1):2-11. DOI: 10.1016/j.tgie.2016.12.001
- Drucker DJ, Holst JJ. The expanding incretin universe: from basic biology to clinical translation. Diabetologia. 2023;66(10):1765-1779. DOI: 10.1007/s00125-023-05906-7
- Blundell J, Finlayson G, Axelsen M, et al. Effects of once-weekly semaglutide on appetite, energy intake, control of eating, food preference and body weight in subjects with obesity. Diabetes, Obesity and Metabolism. 2017;19(9):1242-1251. DOI: 10.1111/dom.12932
- Tschop MH, Finan B, Clemmensen C, et al. Unimolecular polypharmacy for treatment of diabetes and obesity. Cell Metabolism. 2016;24(1):51-62. DOI: 10.1016/j.cmet.2016.06.021
- le Roux CW, Astrup A, Fujioka K, et al. 3 years of liraglutide versus placebo for type 2 diabetes risk reduction and weight management in individuals with prediabetes (SCALE). Lancet. 2017;389(10077):1399-1409. DOI: 10.1016/S0140-6736(17)30069-7
- Coskun T, Sloop KW, Loghin C, et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus. Molecular Metabolism. 2018;18:3-14. DOI: 10.1016/j.molmet.2018.09.001
- Knudsen LB, Lau J. The discovery and development of liraglutide and semaglutide. Frontiers in Endocrinology. 2019;10:155. DOI: 10.3389/fendo.2019.00155
- Adams TD, Davidson LE, Litwin SE, et al. Weight and metabolic outcomes 12 years after gastric bypass. New England Journal of Medicine. 2017;377(12):1143-1155. DOI: 10.1056/NEJMoa1700459
- Sjoholm K, Pajunen P, Jacobson P, et al. Incidence and remission of type 2 diabetes in relation to degree of obesity at baseline and 2 year weight change. Diabetologia. 2015;58(7):1448-1453. DOI: 10.1007/s00125-015-3591-y
- American Society of Anesthesiologists Task Force. Consensus-based guidance on preoperative management of patients on GLP-1 receptor agonists. ASA Practice Advisory. 2023. Available at: asahq.org
- Tchang BG, Tarazi MS, Aras M, Aronne LJ. An update on pharmacotherapeutic strategies for obesity. Expert Opinion on Pharmacotherapy. 2022;23(10):1123-1140. DOI: 10.1080/14656566.2022.2082871
- Wang GJ, Volkow ND, Logan J, et al. Brain dopamine and obesity. Lancet. 2001;357(9253):354-357. DOI: 10.1016/S0140-6736(00)03643-6
- Smith SR, Prosser WA, Donahue DJ, et al. Lorcaserin (APD356), a selective 5-HT2C agonist, reduces body weight in obese men and women. Obesity. 2009;17(3):494-503. DOI: 10.1038/oby.2008.537
- Grill HJ, Hayes MR. Hindbrain neurons as an essential hub in the neuroanatomically distributed control of energy balance. Cell Metabolism. 2012;16(3):296-309. DOI: 10.1016/j.cmet.2012.06.015