Executive Summary
Blood work is the single most valuable tool you have for ensuring peptide therapy is working safely and effectively. Without lab testing, you're flying blind, guessing at doses, and missing early warning signs that could prevent serious complications.
Key Takeaways
- Blood work is the single most valuable tool you have for ensuring peptide therapy is working safely and effectively.
- Without lab testing, you're flying blind, guessing at doses, and missing early warning signs that could prevent serious complications.
- A patient might feel great while their fasting glucose quietly climbs into prediabetic territory.
- Another might assume a peptide isn't working when their IGF-1 levels show it's actually doing exactly what it should.
- The goal isn't to turn you into an endocrinologist.
Whether you're using growth hormone secretagogues like CJC-1295/Ipamorelin, GLP-1 receptor agonists like semaglutide, healing peptides like BPC-157, or immune-modulating compounds like Thymosin Alpha-1, your blood tells a story that symptoms alone can't reveal. A patient might feel great while their fasting glucose quietly climbs into prediabetic territory. Another might assume a peptide isn't working when their IGF-1 levels show it's actually doing exactly what it should.
This guide covers every lab test relevant to peptide therapy monitoring. We'll walk through baseline panels you should order before starting any peptide protocol, compound-specific markers for each major peptide category, optimal timing for draws, how to interpret results beyond standard reference ranges, red flags that require immediate attention, and practical strategies for affordable testing. The goal isn't to turn you into an endocrinologist. It's to give you the knowledge to have informed conversations with your provider and take an active role in monitoring your own health.
Key Principle
Every peptide protocol should begin with baseline blood work and include regular follow-up testing. The specific tests vary by compound, but the principle is universal: measure before you start, monitor while you're on, and recheck after you stop.
Figure 1: The peptide therapy monitoring cycle - baseline labs establish your starting point, follow-up testing tracks response and safety, and results guide protocol adjustments.
Why Blood Work Matters for Peptide Therapy
Peptides aren't supplements you can take casually and hope for the best. They're bioactive compounds that interact with hormonal axes, metabolic pathways, immune signaling, and tissue repair mechanisms. Each of these systems has measurable biomarkers, and those biomarkers tell you things that subjective experience simply cannot.
The Limits of Symptom-Based Monitoring
Many people rely entirely on how they feel to judge whether a peptide is working. While subjective feedback matters, it has serious blind spots. Consider these scenarios:
- False reassurance: A patient on MK-677 feels more energetic and sleeps better, but their fasting glucose has risen from 92 to 118 mg/dL over three months. Without blood work, they'd never know they're trending toward insulin resistance.
- Premature discontinuation: Someone starts CJC-1295/Ipamorelin and feels nothing different after four weeks. They quit, not realizing their IGF-1 has risen from 145 to 235 ng/mL, indicating the peptide is working exactly as intended and body composition changes are coming.
- Missed interactions: A patient stacking multiple peptides develops mildly elevated liver enzymes. Without periodic testing, this goes unnoticed until symptoms appear, at which point the damage may be more significant.
- Dose optimization: Two patients on the same dose of semaglutide can have very different metabolic responses. Blood work lets you personalize dosing rather than relying on one-size-fits-all protocols.
What Blood Work Actually Tells You
Lab testing serves four distinct purposes in peptide therapy:
| Purpose | What It Reveals | Example |
|---|---|---|
| Efficacy | Whether the peptide is producing its intended biological effect | IGF-1 rise on GH peptides, HbA1c reduction on GLP-1s |
| Safety | Whether the peptide is causing unintended harm to organs or metabolic pathways | Liver enzyme elevation, kidney function decline, glucose dysregulation |
| Dose calibration | Whether the current dose is too low, appropriate, or excessive | IGF-1 above 400 ng/mL suggests GH peptide dose may be too high |
| Baseline comparison | How current values compare to your pre-therapy starting point | Knowing your ALT was 28 before starting helps contextualize a reading of 45 during therapy |
The Cost of Skipping Blood Work
Some people skip testing to save money or avoid the inconvenience. This is a false economy. A basic metabolic panel costs $15-35 through direct-to-consumer labs. An IGF-1 test runs $50-80. Compare that to the cost of managing a medical complication that could have been caught early - or the wasted money on peptides that aren't producing measurable results.
The Endocrine Society, the American Association of Clinical Endocrinologists, and clinical practice guidelines for growth hormone therapy all recommend regular laboratory monitoring. These aren't arbitrary suggestions. They're based on decades of clinical data showing that monitored patients have better outcomes and fewer complications [1].
Warning
Using peptides without any blood work monitoring is like driving without a dashboard. You might get where you're going, but you won't know if the engine is overheating until it's too late. At minimum, get baseline labs before starting and follow-up labs at 6-8 weeks.
Baseline Panel: What to Order Before Starting Any Peptide
Before you take your first dose of any peptide, you need a snapshot of where your body stands right now. This baseline serves as your personal reference point for every future lab draw.
Think of it this way: if your ALT comes back at 42 U/L after eight weeks on a peptide, is that a problem? If your baseline was 22, then yes, that's a meaningful increase worth investigating. If your baseline was 39, it's a minor fluctuation that's probably not concerning. Without baseline data, you can't make that distinction.
The Universal Baseline Panel
Regardless of which peptide you're planning to use, every person should get the following tests before starting therapy. This is your foundation.
Comprehensive Metabolic Panel (CMP-14)
The CMP is the workhorse of baseline testing. It covers 14 analytes in a single blood draw and gives you a broad picture of metabolic, liver, and kidney health. Here's what's included and why each marker matters for peptide therapy:
| Marker | Normal Range | Why It Matters for Peptides |
|---|---|---|
| Glucose (fasting) | 70-99 mg/dL | GH peptides and MK-677 can raise fasting glucose; GLP-1s lower it |
| BUN | 7-20 mg/dL | Kidney function baseline; dehydration from GLP-1 side effects can elevate |
| Creatinine | 0.7-1.3 mg/dL (men), 0.6-1.1 (women) | Kidney function; important for any injectable therapy |
| eGFR | >60 mL/min/1.73m2 | Estimated kidney filtration rate; low values may require dose adjustments |
| Sodium | 136-145 mEq/L | Electrolyte balance; GLP-1 GI side effects can disrupt |
| Potassium | 3.5-5.0 mEq/L | Electrolyte balance; vomiting/diarrhea from GLP-1s can deplete |
| Chloride | 98-106 mEq/L | Acid-base balance marker |
| CO2 (Bicarbonate) | 23-29 mEq/L | Acid-base balance |
| Calcium | 8.5-10.5 mg/dL | Bone metabolism; relevant for GH axis peptides |
| Total Protein | 6.0-8.3 g/dL | Nutritional status; important during weight loss on GLP-1s |
| Albumin | 3.5-5.5 g/dL | Liver synthetic function and nutritional status |
| Bilirubin | 0.1-1.2 mg/dL | Liver function baseline |
| ALP | 44-147 U/L | Liver/bone marker |
| ALT | 7-56 U/L | Liver-specific enzyme; most sensitive marker for hepatocyte injury |
Cost through direct-to-consumer labs: typically $15-35 without insurance.
Complete Blood Count (CBC) with Differential
The CBC measures your red blood cells, white blood cells, platelets, and hemoglobin. It's essential baseline data for several reasons:
- Red blood cells and hemoglobin: GH peptides can stimulate erythropoiesis (red blood cell production). You need to know your starting point to detect polycythemia.
- White blood cell count and differential: Immune-modulating peptides like Thymosin Alpha-1 affect immune cell populations. A baseline WBC with differential lets you track changes in lymphocyte, neutrophil, and monocyte counts.
- Platelets: General safety marker; some healing peptides affect platelet function.
Lipid Panel
A standard lipid panel measures total cholesterol, LDL, HDL, and triglycerides. GLP-1 receptor agonists like semaglutide and tirzepatide often improve lipid profiles significantly, so you want a baseline to document those improvements. GH peptides can also affect lipid metabolism.
HbA1c (Glycated Hemoglobin)
HbA1c reflects your average blood glucose over the past 2-3 months. It's more reliable than a single fasting glucose measurement because it isn't affected by what you ate yesterday or how stressed you were during the blood draw. Normal is below 5.7%, prediabetic is 5.7-6.4%, and diabetic is 6.5% or higher.
This test is especially critical if you're planning to use any GH-axis peptide or MK-677, as these compounds can impair glucose tolerance. It's equally important for GLP-1 users, where you expect to see HbA1c decrease [2].
Fasting Insulin
Fasting glucose alone doesn't tell the full story. You can have normal fasting glucose while your pancreas is working overtime to keep it there, pumping out excessive insulin. Fasting insulin reveals this hidden insulin resistance before it progresses to prediabetes or diabetes.
Optimal fasting insulin is generally considered to be 2-6 uIU/mL, though standard lab ranges go up to 24.9. A reading of 15 might be "normal" by lab standards but indicates meaningful insulin resistance that could worsen on GH-axis peptides.
Thyroid Panel (TSH, Free T4, Free T3)
Growth hormone and thyroid function are deeply interconnected. GH-axis peptides can unmask subclinical hypothyroidism or alter thyroid hormone conversion. A baseline thyroid panel ensures you can detect these changes. TSH alone is often insufficient; free T4 and free T3 give a more complete picture of thyroid function [3].

Figure 2: Complete baseline panel checklist for peptide therapy - order these tests before your first dose of any peptide compound.
Additional Baseline Tests by Peptide Category
Beyond the universal panel, certain peptide categories require additional baseline markers. Here's a quick reference:
| Peptide Category | Additional Baseline Tests | Why |
|---|---|---|
| GH Secretagogues (CJC-1295, Ipamorelin, Sermorelin, GHRP-2/6) | IGF-1, fasting insulin, HOMA-IR | Track GH axis stimulation and glucose effects |
| MK-677 (Ibutamoren) | IGF-1, fasting insulin, HOMA-IR, prolactin | Higher risk of glucose dysregulation and prolactin elevation |
| GLP-1 Agonists (Semaglutide, Tirzepatide) | Lipase, amylase, thyroid panel | Pancreatitis risk markers; thyroid C-cell concern (preclinical) |
| Healing Peptides (BPC-157, TB-500) | CRP, ESR | Inflammation baseline to track healing response |
| Immune Peptides (Thymosin Alpha-1) | Lymphocyte subsets (CD4/CD8), NK cells | Immune function baseline |
| Anti-Aging Peptides (Epithalon, NAD+) | Homocysteine, hsCRP, vitamin D | Methylation and inflammation markers |
| Hormonal Peptides (Kisspeptin, PT-141, Gonadorelin) | Total/free testosterone, estradiol, LH, FSH, prolactin | HPG axis baseline |
Clinical Pearl
Always get baseline labs in the morning, fasting (8-12 hours, water only). Hormones like testosterone, cortisol, and insulin follow circadian rhythms, and fasting ensures glucose and lipid values are accurate. Aim for a blood draw between 7:00 and 10:00 AM for the most reliable results.
How to Order Baseline Labs
If you're working with a prescribing physician or clinic, they'll typically order these tests for you. If you want to order them yourself through a direct-to-consumer lab, here's a cost-effective approach:
- CMP + CBC + Lipid Panel bundle: Most DTC labs offer this as a wellness package for $35-65.
- HbA1c: Add-on test, usually $15-30.
- Fasting insulin: Add-on, $20-35.
- IGF-1: Add-on, $50-80.
- Thyroid panel (TSH, Free T4, Free T3): $30-55.
Total cost for a comprehensive baseline: approximately $150-265 through direct-to-consumer labs, compared to $500-1,500+ through hospital-based labs billed to insurance with high deductibles. We'll cover more cost-saving strategies in the Affordable Testing Options section.
GH Peptide Monitoring: IGF-1, Glucose, Insulin, and Beyond
Growth hormone secretagogues are among the most popular peptides in clinical and wellness settings, and they demand the most rigorous lab monitoring. The GH axis affects virtually every metabolic system, and getting it right requires regular, targeted blood work.
This section covers monitoring for all GH-axis peptides, including CJC-1295/Ipamorelin, sermorelin, tesamorelin, GHRP-2, GHRP-6, hexarelin, and MK-677 (ibutamoren).
IGF-1: The Primary Efficacy Marker
Insulin-like growth factor 1 (IGF-1) is the gold standard biomarker for assessing GH-axis peptide therapy. Here's why: growth hormone itself is released in pulses throughout the day, making a single GH blood test unreliable. IGF-1, on the other hand, remains stable throughout the day because it's produced by the liver in response to cumulative GH exposure. A single morning IGF-1 draw gives you an accurate picture of your overall GH status [4].
What IGF-1 Levels Tell You
IGF-1 Reference Ranges by Clinical Significance
Standard laboratory reference ranges for IGF-1 are age-adjusted, and they're quite broad. For a 40-year-old male, the reference range might be 83-316 ng/mL. That's a huge spread. For peptide therapy monitoring, we need to think more precisely:
| IGF-1 Level (ng/mL) | Clinical Interpretation | Action |
|---|---|---|
| Below 100 | Deficient for most adults under 60 | Consider GH-axis peptide therapy; investigate pituitary function |
| 100-150 | Low-normal; suboptimal for therapeutic goals | Peptide therapy may be beneficial; dose may need increasing |
| 150-250 | Mid-range; the sweet spot many clinicians target | Ideal maintenance range for most patients |
| 250-300 | Upper-optimal; good response to therapy | Monitor closely; consider maintaining current dose |
| 300-350 | High-normal; may be acceptable for younger adults | Monitor for side effects; may need dose reduction if symptomatic |
| Above 350-400 | Elevated; increased risk of side effects | Reduce dose; recheck in 4-6 weeks; assess for symptoms of GH excess |
The target range most functional medicine and anti-aging clinicians aim for is 200-300 ng/mL, positioned in the upper half of age-adjusted reference ranges but not beyond. Going above this range rarely provides additional benefit and increases the risk of side effects including joint pain, water retention, carpal tunnel symptoms, and long-term concerns about IGF-1-driven cell proliferation [5].
IGF-1 Testing Protocol
- Timing: Morning fasting draw, preferably between 7:00-10:00 AM
- Preparation: Fast for 8-12 hours (water only). Stop biotin supplements at least 72 hours before testing, as biotin can interfere with immunoassay results.
- Frequency: Baseline, then at 6-8 weeks after starting therapy, then every 3 months during active therapy, and annually during maintenance.
- Sample handling: Serum sample without anticoagulants. Serum should be separated within 2 hours of collection. Avoid repeated freeze-thaw cycles if samples need to be stored [6].
Fasting Glucose and Insulin: The Metabolic Safety Net
Every GH-axis peptide has the potential to affect glucose metabolism. Growth hormone is a counter-regulatory hormone to insulin, meaning it works in opposition to insulin's blood sugar-lowering effects. When you stimulate GH release through peptides, you may see:
- Increased hepatic glucose output (the liver releases more glucose)
- Decreased peripheral glucose uptake (muscles become slightly insulin resistant)
- Increased lipolysis (fat breakdown), which releases free fatty acids that further impair insulin sensitivity
For most people on moderate-dose GH peptide therapy, these effects are mild and clinically insignificant. But for individuals with pre-existing insulin resistance, metabolic syndrome, or a family history of type 2 diabetes, the effects can be meaningful.
MK-677: A Special Case for Glucose Monitoring
MK-677 (ibutamoren) deserves extra attention when it comes to glucose monitoring. Unlike injectable GH secretagogues that produce pulsatile GH release, MK-677 causes a sustained elevation of GH and IGF-1 over 24 hours due to its oral dosing and long half-life. This sustained GH exposure creates a greater risk of glucose dysregulation compared to compounds like ipamorelin or sermorelin [7].
Clinical trials of MK-677 have consistently shown increases in fasting blood glucose, reductions in insulin sensitivity, and elevations in HbA1c. In the Nass et al. study of healthy older adults, two participants developed diabetes-like glucose elevations requiring discontinuation. Case reports have documented overt new-onset diabetes triggered by MK-677 use [8].
MK-677 Glucose Monitoring Protocol
If you're using MK-677, test fasting glucose and fasting insulin at baseline, 4 weeks, 8 weeks, and then every 3 months. Also get HbA1c at baseline and every 3 months. If fasting glucose exceeds 110 mg/dL, fasting insulin exceeds 15 uIU/mL, or HbA1c rises above 5.7%, discuss dose reduction or discontinuation with your provider immediately. Do not wait for symptoms.
HOMA-IR: Calculating Insulin Resistance
If you're getting both fasting glucose and fasting insulin (which you should), you can calculate your HOMA-IR (Homeostatic Model Assessment of Insulin Resistance). The formula is simple:
HOMA-IR = (Fasting Glucose mg/dL x Fasting Insulin uIU/mL) / 405
| HOMA-IR Value | Interpretation |
|---|---|
| Below 1.0 | Optimal insulin sensitivity |
| 1.0-1.9 | Normal; early insulin resistance unlikely |
| 2.0-2.9 | Borderline; emerging insulin resistance |
| 3.0 and above | Significant insulin resistance; requires intervention |
Track your HOMA-IR over time on GH peptide therapy. A rising HOMA-IR is an early warning signal, often appearing before fasting glucose or HbA1c become abnormal. This gives you a window to adjust your protocol before problems develop.
Additional GH Peptide Monitoring Markers
Prolactin
Certain GH secretagogues, particularly GHRP-6 and to a lesser extent GHRP-2, can stimulate prolactin release. MK-677, as a ghrelin mimetic, can also elevate prolactin. Elevated prolactin can suppress testosterone production, reduce libido, and cause breast tissue sensitivity in men. Check prolactin at baseline and at 8-12 weeks on these compounds. Normal range is 4-15 ng/mL for men and 4-23 ng/mL for non-pregnant women.
Cortisol
GHRP-6 and MK-677 can transiently increase cortisol levels. While this effect typically normalizes with continued use, checking morning cortisol at baseline and during early therapy helps ensure you're not developing sustained cortisol elevation, which would counteract many of the benefits you're seeking from GH peptide therapy.
Thyroid Function
Growth hormone increases the peripheral conversion of T4 to T3 and can unmask latent hypothyroidism. If you develop symptoms like fatigue, cold intolerance, or weight gain despite adequate GH-axis stimulation, recheck TSH and free T4. Some patients on GH peptide therapy require thyroid hormone supplementation they didn't need before [9].
PSA (Prostate-Specific Antigen) - Men Over 40
While GH peptides haven't been shown to directly increase prostate cancer risk, IGF-1 is a growth factor that promotes cell proliferation. Men over 40 should have a baseline PSA before starting GH-axis peptide therapy and recheck annually. Any significant rise warrants further evaluation.
Monitoring Schedule Summary - GH Peptides
Baseline: IGF-1, CMP, CBC, fasting insulin, HbA1c, thyroid panel, prolactin (if using GHRP-6 or MK-677), PSA (men over 40).
6-8 weeks: IGF-1, fasting glucose, fasting insulin.
3 months: IGF-1, CMP, fasting insulin, HbA1c. Add prolactin and thyroid if initial values were borderline.
Every 3-6 months ongoing: IGF-1, fasting glucose/insulin, and CMP. Annual comprehensive panel including thyroid and CBC.
GLP-1 Therapy Monitoring: HbA1c, Lipids, Liver, and Kidney Panels
GLP-1 receptor agonists like semaglutide and tirzepatide produce some of the most dramatic and measurable changes in blood work of any peptide class. Monitoring these changes helps you track therapeutic progress, catch potential complications early, and document improvements that validate your treatment.
For a deeper understanding of these compounds, see our GLP-1 research hub and our beginner's guide to peptide therapy.
HbA1c and Glycemic Markers
HbA1c is the single most valuable lab test for tracking GLP-1 therapy effectiveness. It reflects average blood glucose over 2-3 months, smoothing out the daily fluctuations that make single glucose readings unreliable.
Expected HbA1c Changes on GLP-1 Therapy
| Compound | Typical HbA1c Reduction | Timeline |
|---|---|---|
| Semaglutide 2.4 mg (Wegovy) | 0.5-1.5% in diabetic patients | 12-16 weeks at maintenance dose |
| Tirzepatide (Mounjaro/Zepbound) | 1.0-2.4% in diabetic patients | 12-20 weeks at target dose |
| Semaglutide (non-diabetic, weight loss) | 0.2-0.5% reduction (from already normal levels) | 8-12 weeks |
| Compounded semaglutide | Variable; depends on purity and dose accuracy | Similar timeline if properly compounded |
For non-diabetic patients using GLP-1s primarily for weight loss, HbA1c changes will be modest since they're starting from a normal baseline. The more valuable data point for these patients is fasting insulin and HOMA-IR, which often show meaningful improvements in insulin sensitivity even when HbA1c doesn't change much [10].
Fasting Glucose Monitoring
Check fasting glucose at each lab draw. In diabetic patients on GLP-1 therapy, there's a theoretical risk of hypoglycemia, especially when combined with sulfonylureas or insulin. Fasting glucose below 70 mg/dL or symptoms of hypoglycemia (shakiness, sweating, confusion) should prompt a medication review.
For non-diabetic patients, GLP-1s very rarely cause clinically significant hypoglycemia. A fasting glucose of 75-95 mg/dL is typical and healthy during treatment.
Lipid Panel Changes
GLP-1 receptor agonists frequently improve lipid profiles, sometimes dramatically. Expected changes include:
- Triglycerides: Reductions of 15-35% are common, particularly with tirzepatide, which contains a GIP component that specifically improves triglyceride metabolism.
- LDL cholesterol: Modest reductions of 5-15% are typical. Some patients see larger drops, especially those with high baseline LDL.
- HDL cholesterol: Small increases of 3-8% are seen with sustained weight loss.
- Total cholesterol: Generally decreases in proportion to LDL and triglyceride changes.
Get a lipid panel at baseline, at 3 months, and then every 6 months during ongoing therapy. These results can be valuable for discussions with your primary care provider about potentially reducing statin dosing or other lipid medications [11].
Liver Function Tests
Liver monitoring on GLP-1 therapy serves two purposes: tracking improvement in fatty liver disease (which is very common in overweight patients) and watching for the rare possibility of drug-induced liver injury.
The Good News: NAFLD Improvement
Many patients on GLP-1 therapy see significant improvements in liver enzymes. Elevated ALT and AST, which are markers of liver inflammation often associated with non-alcoholic fatty liver disease (NAFLD), frequently normalize as patients lose weight and improve insulin sensitivity. Studies have shown 30-50% reductions in ALT levels after 6-12 months of GLP-1 therapy, with corresponding reductions in liver fat content visible on imaging [12].
The Caution: Drug-Induced Liver Injury
Rare cases of acute liver injury have been reported with tirzepatide. According to the LiverTox database maintained by the NIH, tirzepatide should be at least temporarily discontinued if serum aminotransferase levels rise above 5 times the upper limit of normal (ULN), or if any ALT elevation is accompanied by symptoms of liver disease such as jaundice, dark urine, or right upper quadrant pain [13].
| Liver Marker | Normal Range | When to Be Concerned | When to Stop and Call Provider |
|---|---|---|---|
| ALT | 7-56 U/L | Rise >2x baseline value | Rise >5x ULN or any elevation with symptoms |
| AST | 10-40 U/L | Rise >2x baseline value | Rise >5x ULN or any elevation with symptoms |
| ALP | 44-147 U/L | Rise above reference range | Rise >3x ULN |
| Total Bilirubin | 0.1-1.2 mg/dL | Rise above reference range | Rise >2x ULN, especially with elevated ALT |
Kidney Function Monitoring
Kidney function monitoring is essential during GLP-1 therapy for two reasons. First, GLP-1 medications can cause nausea, vomiting, and diarrhea that lead to dehydration, which stresses the kidneys. Second, many patients starting GLP-1 therapy have pre-existing kidney disease related to diabetes or hypertension.
The key markers are creatinine and eGFR, both included in the standard CMP. Patients with an eGFR below 30 mL/min/1.73m2 require careful dose adjustment and more frequent monitoring.
Hydration Alert
If you're experiencing GI side effects from GLP-1 therapy (nausea, vomiting, diarrhea), make a conscious effort to maintain adequate fluid intake. Dehydration can rapidly worsen kidney function, especially in patients with pre-existing kidney disease or those taking other medications like ACE inhibitors, ARBs, or NSAIDs. If you can't keep fluids down for more than 24 hours, contact your provider.
Pancreatic Markers
GLP-1 receptor agonists carry a labeled warning for pancreatitis risk, though large-scale studies suggest the absolute risk is very low. Baseline lipase and amylase values help contextualize any future elevations.
- Lipase (normal: 0-160 U/L): More specific for pancreatitis than amylase. An elevation >3x ULN with abdominal pain radiating to the back is concerning for pancreatitis.
- Amylase (normal: 28-100 U/L): Less specific; can be elevated from salivary glands and other sources.
You don't need to check these at every visit, but baseline values are important, and any episode of severe abdominal pain during GLP-1 therapy should prompt urgent lipase testing [14].
Thyroid Monitoring
GLP-1 receptor agonists carry a boxed warning regarding thyroid C-cell tumors based on rodent studies. While this risk has not been confirmed in humans even after years of widespread use and pharmacovigilance data, thyroid monitoring remains a standard recommendation:
- Baseline TSH and free T4 before starting therapy
- Annual thyroid function testing during therapy
- GLP-1 therapy is contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Report any neck mass, dysphagia, dysphonia, or persistent hoarseness to your provider immediately
Nutritional Markers During Weight Loss
Significant weight loss on GLP-1 therapy can create nutritional deficiencies, particularly when appetite is substantially reduced. Consider adding these tests at 3-6 month intervals:
| Nutrient | Test | Why It Matters |
|---|---|---|
| Vitamin B12 | Serum B12 or methylmalonic acid | GLP-1s slow gastric emptying, potentially reducing B12 absorption |
| Vitamin D | 25-hydroxyvitamin D | Rapid weight loss can deplete vitamin D stores; also relevant for bone health |
| Iron | Ferritin, iron, TIBC | Reduced food intake can lead to iron deficiency, especially in menstruating women |
| Folate | Serum folate | Reduced food variety can lower folate intake |
| Magnesium | RBC magnesium (not serum) | GI side effects and reduced intake can deplete magnesium |
Monitoring Schedule Summary - GLP-1 Therapy
Baseline: CMP, CBC, HbA1c, fasting insulin, lipid panel, lipase, thyroid panel (TSH, fT4).
4-6 weeks (during titration): Fasting glucose, CMP (kidney/liver check), electrolytes if GI symptoms are present.
3 months: HbA1c, CMP, lipid panel, fasting insulin.
6 months: Comprehensive panel - HbA1c, CMP, CBC, lipids, nutritional markers (B12, D, iron, folate), thyroid.
Annually: Full panel repeat including all baseline tests.
Figure 3: GLP-1 therapy monitoring timeline showing recommended tests at each interval from baseline through the first year of treatment.
Healing Peptide Monitoring: BPC-157, TB-500, and Tissue Repair Compounds
Healing peptides like BPC-157 and TB-500 (Thymosin Beta-4) are used primarily for tissue repair, gut healing, and injury recovery. Their monitoring requirements are less intensive than GH or GLP-1 peptides, but baseline labs and periodic safety checks are still important.
Why Monitoring is Different for Healing Peptides
Healing peptides don't primarily work through hormonal axes. BPC-157 modulates growth factor expression, angiogenesis, and inflammatory pathways at a local tissue level. TB-500 promotes cell migration, blood vessel formation, and reduces inflammation. Neither compound produces the systemic hormonal shifts that require close lab tracking the way GH peptides or GLP-1s do.
That said, they're still bioactive compounds being injected or ingested, and basic safety monitoring is appropriate, especially for protocols lasting longer than 4-6 weeks [15].
Recommended Labs for Healing Peptide Protocols
Baseline
- CMP: Liver and kidney function baseline (especially if using injectable formulations)
- CBC: General health baseline
- CRP (C-Reactive Protein): Quantitative inflammation marker. BPC-157 and TB-500 are used to reduce inflammation; having a baseline CRP lets you objectively track whether they're working.
- ESR (Erythrocyte Sedimentation Rate): Another inflammation marker; useful alongside CRP for a more complete picture.
During Therapy (4-8 week check)
- CMP: Confirm liver and kidney function remain stable
- CRP: Compare to baseline; expect a decrease if the peptide is effectively reducing inflammation
Optional Advanced Markers
- IL-6, TNF-alpha: Specific inflammatory cytokines that BPC-157 is shown to modulate in animal studies. These are more expensive tests ($50-100 each) but can provide granular data on anti-inflammatory effects.
- VEGF (Vascular Endothelial Growth Factor): BPC-157 stimulates angiogenesis partly through VEGF pathways. Elevated VEGF could theoretically be a concern in patients with a history of certain cancers. This test is rarely ordered in practice but may be relevant for patients with oncologic history.
Special Considerations
Gut Healing Protocols (Oral BPC-157)
When using oral BPC-157 for gut healing (IBS, leaky gut, gastric ulcers), consider adding:
- Fecal calprotectin: Marker of intestinal inflammation; can track improvement objectively
- Lactulose/mannitol test: Measures intestinal permeability ("leaky gut"); can document barrier function improvement
- Food sensitivity panels: Before and after therapy to track whether reduced gut inflammation leads to fewer food reactions
Injectable BPC-157 and TB-500
For subcutaneous or intramuscular injection protocols, liver and kidney monitoring through a standard CMP is sufficient for safety. No specific organ toxicity has been identified with these compounds in the limited human data available, but the precautionary principle applies: if you're injecting a compound regularly, check your organ function periodically.
Clinical Pearl
The best objective evidence that BPC-157 or TB-500 is working isn't always a blood test. For musculoskeletal injuries, functional assessments (range of motion, pain scales, imaging) may be more informative than lab work. Blood tests for healing peptides serve primarily as safety monitoring, with CRP/ESR providing some efficacy data for inflammation-related conditions.
Immune Peptide Monitoring: Thymosin Alpha-1, LL-37, and Immune Modulators
Immune-modulating peptides like Thymosin Alpha-1 directly affect immune cell populations and inflammatory signaling. Monitoring requires specific immune markers that you wouldn't typically order for other peptide categories.
Thymosin Alpha-1 Monitoring
Thymosin Alpha-1 (Ta1) enhances T-cell maturation, increases NK cell activity, and modulates dendritic cell function. It's used for chronic infections, immunodeficiency, cancer adjunctive therapy, and autoimmune conditions. Proper monitoring helps confirm immune activation and ensures the immune system isn't being overstimulated [16].
Recommended Labs
| Test | Baseline | 4-6 Weeks | 3 Months | Purpose |
|---|---|---|---|---|
| CBC with Differential | Yes | Yes | Yes | Track WBC, lymphocyte, and neutrophil counts |
| Lymphocyte Subset Panel (CD4, CD8, CD4/CD8 ratio) | Yes | Yes | Yes | Monitor T-cell population changes |
| NK Cell Count (CD56+) | Yes | Optional | Yes | Track natural killer cell activation |
| CRP / hsCRP | Yes | Yes | Yes | Track systemic inflammation |
| CMP | Yes | Optional | Yes | Liver/kidney safety monitoring |
| ESR | Yes | Optional | Yes | Inflammation tracking |
| Immunoglobulins (IgG, IgA, IgM) | Yes | No | Optional | Humoral immune function |
What to Look For
Thymosin Alpha-1 should produce a measurable increase in CD4 T-cell counts and NK cell activity over 4-8 weeks. The CD4/CD8 ratio often normalizes toward the healthy range of 1.0-3.0. hsCRP should trend downward if the peptide is effectively modulating immune-mediated inflammation.
If you see a dramatic spike in WBC count (above 12,000/uL), excessive lymphocyte elevation, or new autoimmune symptoms (joint pain, rash, unexplained fatigue), this could indicate immune overstimulation. Discuss dose reduction or temporary discontinuation with your provider.
Monitoring for Other Immune Peptides
LL-37 (Antimicrobial Peptide)
LL-37 has direct antimicrobial properties and modulates innate immunity. Monitoring is primarily safety-focused:
- CMP at baseline and 4-6 weeks (liver/kidney safety)
- CBC with differential (track immune cell response)
- hsCRP (inflammation tracking)
VIP (Vasoactive Intestinal Peptide)
VIP is used for chronic inflammatory response syndrome (CIRS) and mold illness. Specific monitoring includes:
- MSH (Melanocyte-Stimulating Hormone)
- TGF-beta 1
- MMP-9 (Matrix Metalloproteinase-9)
- VEGF
- These are specialized CIRS markers that should be ordered and interpreted by a provider familiar with the Shoemaker protocol
Key Consideration for Autoimmune Patients
If you have an autoimmune condition and are using immune-modulating peptides, additional monitoring is warranted. Track disease-specific autoantibodies (ANA, anti-dsDNA, anti-CCP, etc.) at baseline and periodically during therapy. Immune stimulation could theoretically exacerbate autoimmune conditions, though Thymosin Alpha-1 is generally considered immunomodulatory rather than purely immunostimulatory.
Hormone Panels: Testosterone, Estradiol, and Endocrine Markers
Several peptide categories directly or indirectly affect the hypothalamic-pituitary-gonadal (HPG) axis and other endocrine pathways. Comprehensive hormone testing helps you understand these effects and optimize your protocol.
When Hormone Panels Are Needed
Not every peptide user needs a full hormone panel. Here's when to include hormone testing:
| Situation | Recommended Hormone Tests |
|---|---|
| Using GH secretagogues (any) | IGF-1 (covered above), thyroid panel, fasting insulin |
| Using MK-677 or GHRPs that raise prolactin | Prolactin, total testosterone (men), estradiol |
| Using Kisspeptin or Gonadorelin | LH, FSH, total/free testosterone, estradiol, SHBG |
| Using PT-141 (Bremelanotide) | Baseline hormone panel; no specific ongoing requirements |
| Stacking peptides with TRT | Total/free testosterone, estradiol, SHBG, hematocrit, PSA |
| Women on any peptide therapy | Estradiol, progesterone, DHEA-S, testosterone (total), thyroid panel |
Key Hormone Markers and Their Interpretation
Total and Free Testosterone (Men)
Total testosterone measures all testosterone in your blood, both bound to proteins and free-floating. Free testosterone measures only the unbound, biologically active fraction. For peptide therapy monitoring:
- Normal total testosterone: 300-1,000 ng/dL (most clinicians consider 400+ as adequate and 600-900 as optimal)
- Normal free testosterone: 9-30 pg/mL (varies significantly by assay)
- Why it matters: GH peptides can indirectly support testosterone production by improving sleep quality and reducing body fat. MK-677-induced prolactin elevation can suppress testosterone. Kisspeptin and Gonadorelin directly stimulate LH/FSH and can raise testosterone.
Estradiol (E2)
Estradiol monitoring is particularly relevant for men using peptides that affect testosterone, as increased testosterone often leads to increased aromatization and higher estradiol. The sensitive estradiol assay (LC/MS-MS) is preferred over standard immunoassay for male patients.
- Optimal range for men: 20-35 pg/mL (by sensitive assay)
- Too low (<15 pg/mL): Joint pain, low libido, poor mood, bone density concerns
- Too high (>50 pg/mL): Water retention, mood changes, gynecomastia risk
LH and FSH
Luteinizing hormone (LH) and follicle-stimulating hormone (FSH) are the pituitary hormones that drive gonadal function. These are essential tests when using peptides that target the HPG axis:
- Kisspeptin: Should increase LH pulsatility. Check LH and FSH at baseline and 4-6 weeks.
- Gonadorelin (GnRH analog): Acute administration increases LH/FSH; chronic pulsatile administration maintains them. Testing confirms the desired response.
DHEA-S
DHEA-S is a precursor hormone produced by the adrenal glands. It serves as a marker of adrenal function and overall hormonal vitality. While no peptide directly targets DHEA-S production, it's a useful baseline marker for assessing overall endocrine health before starting any hormone-influencing peptide protocol.
SHBG (Sex Hormone-Binding Globulin)
SHBG binds testosterone and makes it inactive. High SHBG can make total testosterone look normal while free testosterone is actually low. GH peptides can lower SHBG, which is one mechanism by which they indirectly improve androgen status. Track SHBG alongside testosterone for a complete picture.
Clinical Pearl
Always draw hormone panels in the morning, ideally between 7:00-10:00 AM. Testosterone peaks in early morning and can drop 20-30% by afternoon. An afternoon draw could yield a misleadingly low result, potentially leading to unnecessary intervention. Fasting is preferred but less critical for hormone levels than for metabolic markers.
Figure 4: How different peptide categories interact with the hormonal axis, showing which hormone markers to monitor for each compound class.
Inflammatory Markers: CRP, ESR, Cytokines, and Beyond
Chronic inflammation underlies many of the conditions that peptide therapy aims to address, from metabolic syndrome and autoimmune disease to slow-healing injuries and accelerated aging. Tracking inflammatory markers gives you objective data on whether your protocol is actually reducing the inflammatory burden in your body.
High-Sensitivity C-Reactive Protein (hsCRP)
hsCRP is the most widely available and clinically validated marker of systemic inflammation. It's produced by the liver in response to inflammatory cytokines, primarily IL-6. The "high-sensitivity" version of the test can detect low levels of chronic inflammation that the standard CRP test would miss.
| hsCRP Level | Risk Category | Interpretation for Peptide Users |
|---|---|---|
| Below 1.0 mg/L | Low cardiovascular risk | Optimal; indicates good baseline inflammatory status |
| 1.0-3.0 mg/L | Average risk | Room for improvement; peptide therapy may help reduce |
| 3.0-10.0 mg/L | High risk | Significant chronic inflammation; monitor closely during therapy |
| Above 10.0 mg/L | Acute inflammation | Usually indicates acute infection or injury, not chronic inflammation; retest after acute cause resolves |
For peptide therapy monitoring, hsCRP is valuable because many compounds, from BPC-157 and Thymosin Alpha-1 to GLP-1 agonists and Epithalon, have anti-inflammatory properties. A declining hsCRP over 3-6 months provides objective evidence that your protocol is working at the inflammatory level [17].
Erythrocyte Sedimentation Rate (ESR)
ESR measures how quickly red blood cells settle in a test tube over one hour. Faster settling indicates more inflammation. It's less specific than hsCRP but captures a different aspect of the inflammatory response and can stay elevated for longer after acute events resolve.
- Normal: 0-22 mm/hr (men), 0-29 mm/hr (women) - ranges increase slightly with age
- Best used alongside hsCRP: When both are elevated, there's strong evidence of systemic inflammation. When they diverge, further investigation may be needed.
Advanced Inflammatory Markers
For patients with significant inflammatory conditions or those who want deeper insight into their inflammatory profile, these additional tests can be informative:
Cytokine Panels
| Marker | What It Measures | Relevant Peptides | Typical Cost |
|---|---|---|---|
| IL-6 (Interleukin-6) | Pro-inflammatory cytokine; driver of CRP production | BPC-157, Thymosin Alpha-1, GLP-1 agonists | $50-100 |
| TNF-alpha | Major pro-inflammatory cytokine | BPC-157, Thymosin Alpha-1 | $50-100 |
| IL-10 | Anti-inflammatory cytokine | Thymosin Alpha-1 (increases IL-10) | $50-100 |
| TGF-beta 1 | Fibrosis and immune regulation marker | VIP, CIRS protocols | $75-150 |
Cytokine panels are expensive and not necessary for routine monitoring. They're most useful for patients with chronic inflammatory conditions who want detailed data on which specific inflammatory pathways are being affected by their peptide protocol.
Homocysteine
Homocysteine is an amino acid that, when elevated, indicates impaired methylation and increased cardiovascular and neurological risk. It's relevant for anti-aging peptide protocols involving NAD+ therapy, as NAD+ metabolism increases methylation demand. Elevated homocysteine suggests a need for B-vitamin support (B12, folate, B6).
- Optimal: Below 8 umol/L
- Acceptable: 8-12 umol/L
- Elevated: Above 12 umol/L (requires B-vitamin supplementation and recheck)
Ferritin
Ferritin is both an iron storage marker and an acute-phase reactant that rises with inflammation. In the context of peptide therapy:
- Low ferritin (below 30 ng/mL) indicates iron deficiency, common during weight loss on GLP-1 therapy
- High ferritin (above 300 ng/mL in men, 200 ng/mL in women) with normal iron may indicate inflammation rather than iron overload
- Track alongside hsCRP to distinguish between iron storage and inflammatory elevation
Practical Tip
You don't need to order every inflammatory marker listed here. For most peptide users, hsCRP alone is sufficient. Add ESR if you want a second perspective. Reserve cytokine panels for specific clinical situations where detailed inflammatory profiling will change your treatment approach. A single hsCRP test costs $15-30 through direct-to-consumer labs, making it one of the most cost-effective monitoring tools available.
Optimal Ranges vs. Standard Reference Ranges: What the Numbers Really Mean
One of the most common sources of confusion in peptide therapy monitoring is the difference between a lab's reference range and what's actually optimal for your health and therapeutic goals. Understanding this distinction is essential for getting the most from your blood work.
Why Lab Reference Ranges Can Be Misleading
Standard laboratory reference ranges are typically derived from the central 95% of the general population tested at that lab. This means the "normal" range includes people who are overweight, sedentary, chronically stressed, sleep-deprived, and in various states of subclinical disease. Being within the reference range means you're similar to 95% of people tested - not that you're in optimal health.
Consider this example: the standard reference range for fasting insulin might be listed as 2.6-24.9 uIU/mL. A reading of 18 falls within the "normal" range, but it clearly indicates insulin resistance. An optimal fasting insulin is 2-6 uIU/mL. The lab wouldn't flag 18 as abnormal, but your clinician should.
Optimal vs. Standard Ranges for Key Peptide Monitoring Markers
| Marker | Standard Lab Range | Optimal/Functional Range | Notes |
|---|---|---|---|
| Fasting Glucose | 65-99 mg/dL | 75-90 mg/dL | Below 75 may indicate reactive hypoglycemia; above 90 shows early glucose dysregulation |
| Fasting Insulin | 2.6-24.9 uIU/mL | 2-6 uIU/mL | Values above 8-10 indicate meaningful insulin resistance |
| HbA1c | Below 5.7% (non-diabetic) | 4.8-5.3% | Optimal metabolic health target |
| HOMA-IR | Below 2.5 (varies) | Below 1.5 | Calculated from fasting glucose and insulin |
| IGF-1 (age 30-50) | 83-316 ng/mL | 200-280 ng/mL | Upper-middle of age-adjusted range; not maximum |
| hsCRP | Below 3.0 mg/L | Below 1.0 mg/L | Cardiovascular and longevity optimal target |
| TSH | 0.45-4.5 mIU/L | 1.0-2.5 mIU/L | Many patients feel best in this narrower range |
| Free T4 | 0.82-1.77 ng/dL | 1.1-1.5 ng/dL | Mid to upper range |
| Total Testosterone (men) | 264-916 ng/dL | 600-900 ng/dL | Upper-middle range for vitality and well-being |
| Vitamin D (25-OH) | 30-100 ng/mL | 50-80 ng/mL | Many patients are suboptimal despite being "in range" |
| Ferritin | 12-300 ng/mL (men) | 50-150 ng/mL | Both deficiency and excess are problematic |
| Homocysteine | Below 15 umol/L | Below 8 umol/L | Cardiovascular and neurological optimal target |
| ALT | 7-56 U/L | Below 25 U/L | Truly healthy liver function is at the lower end |
How to Use This Table
The "optimal" ranges listed above reflect functional medicine targets used by clinicians specializing in preventive and anti-aging medicine. They represent where most people feel and function their best. Your individual optimal range may differ based on age, genetics, medication use, and other factors. These ranges should inform your discussions with your provider, not replace medical judgment.
Figure 5: Standard lab reference ranges vs. optimal functional ranges for key biomarkers in peptide therapy monitoring. The optimal range is typically a narrower window within the broader lab range.
Age-Adjusted IGF-1 Reference Ranges
IGF-1 naturally declines with age, so the same number means different things at different ages. Here's an approximate breakdown of age-adjusted reference ranges used by most clinical laboratories:
| Age Group | Reference Range (ng/mL) | Optimal Target on GH Peptides (ng/mL) |
|---|---|---|
| 18-25 | 116-358 | 250-320 |
| 26-35 | 109-324 | 230-300 |
| 36-45 | 101-297 | 210-280 |
| 46-55 | 94-269 | 200-260 |
| 56-65 | 87-238 | 180-240 |
| 66-75 | 75-212 | 160-220 |
| 76+ | 54-188 | 140-200 |
The optimal target on GH peptides generally corresponds to the upper 40-60th percentile of the age-adjusted range. Going above this doesn't provide meaningful additional benefit for most people and may increase side effects and long-term risks [18].
Interpreting Results: What Changes Mean and When to Act
Getting blood work is only half the equation. Knowing how to read the results, what changes are expected, what changes are concerning, and what to do about abnormal findings is where the real value lies.
Expected vs. Unexpected Changes by Peptide Category
GH Peptide Expected Changes
| Marker | Expected Change | Concerning Change |
|---|---|---|
| IGF-1 | Increase of 50-150% from baseline over 6-12 weeks | IGF-1 above 400 ng/mL or no change after 8 weeks (suggests compound isn't working) |
| Fasting Glucose | May rise 5-15 mg/dL (usually transient) | Rise above 110 mg/dL sustained; above 126 mg/dL is diagnostic for diabetes |
| Fasting Insulin | May increase slightly | Rise above 15 uIU/mL; HOMA-IR above 3.0 |
| Prolactin | May rise modestly on GHRPs/MK-677 | Doubling of baseline value; values above 25 ng/mL in men |
| Thyroid (TSH) | Usually stable | TSH rising above 4.5 mIU/L; new hypothyroid symptoms |
GLP-1 Expected Changes
| Marker | Expected Change | Concerning Change |
|---|---|---|
| HbA1c | Decrease of 0.5-2.0% (diabetics); 0.1-0.5% (non-diabetics) | No change after 3 months at adequate dose; unexpected increase |
| Fasting Glucose | Decrease toward 80-95 mg/dL | Below 70 mg/dL (hypoglycemia); no improvement from elevated baseline |
| ALT/AST | Usually improve (especially if NAFLD was present) | Rise >2x baseline; rise >5x ULN requires urgent evaluation |
| Triglycerides | Decrease of 15-35% | Unexpected rise; lipase elevation with abdominal pain |
| Creatinine/eGFR | Usually stable or slightly improved | Rising creatinine; falling eGFR; especially with dehydration symptoms |
| Albumin | Should remain stable | Falling albumin suggests inadequate protein intake during weight loss |
The Trend Matters More Than a Single Value
One of the most important principles in lab interpretation is that trends are more informative than single readings. A fasting glucose of 102 mg/dL in isolation might not be alarming, but if it was 88 at baseline, 95 at the 6-week check, and 102 at 3 months, that's a clear upward trend that demands attention.
This is exactly why baseline testing is so essential. Without knowing where you started, you can't identify trends. Keep a personal record or spreadsheet of all your lab results over time, organized chronologically. Most direct-to-consumer lab companies provide online portals where you can view historical results and see trends graphed automatically.
Common Interpretation Mistakes
Mistake 1: Panicking Over One Abnormal Value
A single slightly out-of-range result is often meaningless. Lab tests have inherent variability. You could draw blood on two consecutive days and get slightly different results. A fasting glucose of 101 when the range says "below 100" is not cause for alarm. It's the pattern over time that matters. Recheck in a few weeks before making protocol changes.
Mistake 2: Ignoring Values That Are "In Range" But Trending Wrong
As discussed above, being within the reference range doesn't mean everything is fine. An ALT of 48 is "within range" (7-56 U/L) but if your baseline was 22, that's more than a 100% increase and deserves investigation.
Mistake 3: Comparing Results From Different Labs
Different laboratories use different assays and may report different reference ranges. If your IGF-1 was measured at Quest last time and LabCorp this time, the results may not be directly comparable. For the most accurate trend tracking, use the same laboratory for all your testing.
Mistake 4: Not Controlling for Variables
Blood work results can be significantly affected by factors unrelated to your peptide therapy. Heavy exercise within 24-48 hours can elevate liver enzymes (AST, ALT, CK). Alcohol consumption affects liver markers. Dehydration concentrates blood and raises BUN, creatinine, and other values. Acute illness or stress elevates CRP, WBC, and cortisol. Poor sleep the night before affects cortisol and glucose. Always try to control these variables and note them when reviewing results.
Clinical Pearl
When reviewing results with your provider, bring all previous lab results for comparison. Ask three specific questions: (1) Has anything changed significantly from baseline? (2) Are any trends developing that concern you? (3) Do these results suggest any dose adjustments? This framework keeps the conversation focused and productive.
Testing Frequency: When and How Often to Get Blood Work
Timing matters. Testing too infrequently means missing important changes. Testing too often wastes money and can create unnecessary anxiety from normal physiological fluctuations. Here's how to find the right balance for each phase of peptide therapy.
The Three Phases of Monitoring
Phase 1: Initiation (Weeks 0-12)
This is the most intensive monitoring period. You're establishing baseline, confirming the peptide is producing its expected biological effects, and watching for early adverse responses.
| Timepoint | Tests | Purpose |
|---|---|---|
| Before starting (Week 0) | Full baseline panel | Establish personal reference values |
| Week 4-6 | Targeted check (primary efficacy/safety markers) | Early response confirmation; catch acute adverse effects |
| Week 8-12 | Comprehensive follow-up | Assess full response at steady-state; make dose adjustments |
Phase 2: Optimization (Months 3-12)
Once you've confirmed the peptide is working and safe, monitoring frequency decreases. Testing every 3 months during the first year allows you to track ongoing response and detect gradual changes.
Phase 3: Maintenance (After Year 1)
For long-term peptide therapy, testing every 6 months is generally adequate if values have been stable. Annual comprehensive panels should include everything from the baseline panel to catch any slow-developing changes.
Compound-Specific Testing Schedules

Figure 6: Recommended blood work testing schedule organized by peptide category, showing the testing cadence from baseline through one year.
| Peptide Category | Initiation Testing | Optimization Testing | Maintenance Testing |
|---|---|---|---|
| GH Secretagogues | Baseline, 6 weeks, 12 weeks | Every 3 months | Every 6 months |
| MK-677 | Baseline, 4 weeks, 8 weeks | Every 3 months (glucose focus) | Every 3-6 months |
| GLP-1 Agonists | Baseline, 4-6 weeks (during titration), 12 weeks | Every 3 months | Every 6 months |
| Healing Peptides (BPC-157, TB-500) | Baseline, 4-8 weeks (if protocol >4 weeks) | Not typically needed for short protocols | CMP every 6 months if ongoing |
| Immune Peptides (Ta1) | Baseline, 4-6 weeks, 12 weeks | Every 3 months | Every 6 months |
| Anti-Aging (Epithalon, NAD+) | Baseline, 8-12 weeks | Every 6 months | Annually |
Special Timing Considerations
When to Test Relative to Dosing
For GH peptides, the best time to test IGF-1 is any morning fasting draw, since IGF-1 levels are stable throughout the day. It doesn't matter when you took your last dose. However, don't draw blood within 2-3 hours of an acute GH secretagogue injection, as this could transiently affect glucose and insulin values.
For GLP-1 agonists (weekly injections like semaglutide), blood work can be drawn on any day of the weekly cycle. There's no need to time it relative to your injection day, since these drugs have steady-state pharmacokinetics with weekly dosing [19].
When to Add Unscheduled Testing
Beyond your routine schedule, get blood work promptly if you experience:
- New onset joint pain or swelling (on GH peptides) - check IGF-1
- Excessive thirst and urination (any peptide) - check glucose, HbA1c
- Severe or persistent GI symptoms on GLP-1s - check CMP, electrolytes
- Yellowing of skin or eyes - check liver panel urgently
- Significant fatigue or cold intolerance (GH peptides) - check thyroid panel
- Breast tenderness or gynecomastia (men) - check prolactin, estradiol
- New onset edema or rapid weight gain - check kidney function, albumin
Red Flags: When Blood Work Demands Immediate Action
Most abnormal lab findings are manageable with dose adjustments or simple interventions. But some results require immediate action, meaning same-day contact with your healthcare provider or, in rare cases, emergency care.
Critical Lab Values Requiring Urgent Action
| Finding | Threshold | Possible Cause | Action Required |
|---|---|---|---|
| Fasting Glucose | >200 mg/dL | Severe glucose dysregulation; possible new-onset diabetes | Stop all GH peptides/MK-677 immediately; seek medical evaluation same day |
| ALT or AST | >5x Upper Limit of Normal (~280 U/L) | Significant hepatocellular injury | Stop the suspected peptide immediately; seek hepatology evaluation |
| eGFR | Rapid decline below 30 mL/min | Acute kidney injury from dehydration or drug interaction | Stop peptide; aggressive hydration; urgent nephrology referral |
| Potassium | <3.0 or >5.5 mEq/L | GI losses (vomiting/diarrhea) or renal dysfunction | Same-day medical evaluation; cardiac risk |
| Sodium | <130 or >150 mEq/L | Severe dehydration or water intoxication | Same-day medical evaluation |
| Lipase | >3x ULN with abdominal pain | Acute pancreatitis | Emergency evaluation; stop GLP-1 immediately |
| Hematocrit | >54% (men) or >48% (women) | Polycythemia from GH peptide therapy | Stop peptide; hematology evaluation; may need therapeutic phlebotomy |
| WBC | >20,000/uL or <2,000/uL | Immune overstimulation or bone marrow suppression | Stop immune peptides; urgent hematology evaluation |
Serious But Non-Emergency Findings
These results warrant prompt but not emergency attention. Contact your prescribing provider within 1-2 business days:
- Fasting glucose 126-200 mg/dL: Prediabetic/diabetic range. Reduce or stop GH-axis peptide/MK-677; get HbA1c for confirmation.
- HbA1c above 6.5%: Diabetic threshold. Requires formal diabetes workup and management.
- ALT/AST 2-5x ULN: Moderate liver enzyme elevation. Reduce or hold suspected compound. Recheck in 2-4 weeks. Add GGT and direct bilirubin for more specificity.
- IGF-1 above 400 ng/mL: Excessive GH-axis stimulation. Reduce GH peptide dose by 25-50%. Recheck in 4-6 weeks.
- Prolactin above 50 ng/mL: Significantly elevated. Consider MRI of pituitary to rule out prolactinoma, especially if not explained by medication effect.
- TSH above 10 mIU/L: Overt hypothyroidism. Requires thyroid hormone replacement.
- Albumin below 3.0 g/dL: Significant malnutrition or liver dysfunction. Review dietary intake on GLP-1 therapy; evaluate liver function.
Findings That Require Monitoring But Not Panic
These are common findings during peptide therapy that usually resolve with simple interventions:
- Fasting glucose 100-125 mg/dL: Impaired fasting glucose. Adjust diet, exercise, and consider reducing GH-axis peptide dose. Recheck in 4-6 weeks.
- Mild ALT elevation (1.5-2x baseline but still within ULN): Often resolves spontaneously. Recheck in 4-6 weeks. Ensure no alcohol use or recent intense exercise, which can transiently elevate liver enzymes.
- Prolactin mildly elevated (16-30 ng/mL in men): Common on GHRP-6 or MK-677. Consider switching to ipamorelin, which doesn't significantly raise prolactin. Or add vitamin B6 (P5P form), which is a dopamine cofactor that helps suppress prolactin.
- Slightly elevated hsCRP (3-10 mg/L): Could be peptide-related or unrelated (recent illness, poor sleep, stress). Recheck in 4-6 weeks after controlling variables.
The 5x Rule for Liver Enzymes
The hepatology community generally uses the "5x rule" as a threshold for concern. If ALT or AST rises above 5 times the upper limit of normal (approximately 280 U/L for ALT, 200 U/L for AST), the suspected compound should be stopped immediately and a formal hepatology evaluation initiated. This rule applies regardless of symptoms. Below 5x ULN, clinical judgment guides whether to reduce the dose, switch compounds, or continue with closer monitoring [20].
Working With Your Provider: Getting the Most From Your Lab Reviews
The most sophisticated blood work is worthless if it isn't properly reviewed and acted upon. Your relationship with your healthcare provider is the bridge between raw data and meaningful clinical decisions.
Finding the Right Provider
Not all providers are equally equipped to manage peptide therapy monitoring. The ideal provider:
- Has experience prescribing or managing patients on peptide therapy
- Understands functional/optimal ranges, not just standard lab reference ranges
- Is willing to order and review the specific tests relevant to your peptide protocol
- Takes a preventive/proactive approach rather than waiting for overt disease
- Communicates clearly about what your results mean and what actions to take
Common provider types who work with peptide therapy include: anti-aging medicine physicians, functional medicine practitioners, integrative medicine doctors, endocrinologists (for complex hormonal cases), and increasingly, telemedicine peptide clinics that include lab monitoring in their protocols.
If you're just starting to explore peptide therapy, our getting started page and beginner's guide can help orient you.
Preparing for Your Lab Review
Make your appointment productive by preparing in advance:
- Bring all previous results: Either printed or on your phone/laptop. Trends can't be assessed without historical data.
- List your current protocol: Write down every peptide, dose, frequency, and duration. Include any other supplements or medications.
- Note any symptoms: Document any new symptoms since your last lab draw, even if they seem unrelated. Correlating symptoms with lab changes is where clinical insight happens.
- Prepare specific questions: Rather than "how are my labs?" ask targeted questions: "Has my IGF-1 reached the target range?" "Is the trend in my fasting glucose concerning?" "Should we adjust my MK-677 dose based on these insulin levels?"
- Know what you want: Are you looking for permission to increase a dose? Reassurance that your liver is fine? A recommendation to add or drop a compound? Having clarity about your goals makes the conversation more productive.
When Your Provider Doesn't Know Peptides
If your primary care provider isn't familiar with peptide therapy, you can still involve them in your monitoring. Focus the conversation on the lab values and clinical significance rather than the specific peptide. For example, instead of saying "I'm taking CJC-1295/Ipamorelin and need IGF-1 monitoring," you could say "I'd like to check my IGF-1 level as part of a growth hormone screening." Most providers will order standard lab tests when asked, even if they're not the ones prescribing the peptides.
For compound-specific guidance, consider working with a specialized peptide clinic or telemedicine provider while keeping your PCP informed of your results. The peptide calculator can also help you understand dosing parameters to discuss with your provider.
Telehealth and Remote Monitoring
Telehealth has made peptide therapy monitoring more accessible than ever. Many providers now offer:
- Virtual lab review appointments
- Electronic ordering of lab tests at nationwide draw sites
- Automated flagging of abnormal results
- Secure messaging for questions between appointments
- Integration with direct-to-consumer lab platforms
This combination of DTC labs and telehealth can reduce the total cost of monitoring significantly compared to traditional office visits with hospital lab testing.
Cost and Access: Affordable Testing Options
Cost is one of the most commonly cited reasons people skip blood work monitoring. The good news is that lab testing has never been more affordable or accessible, thanks to the growth of direct-to-consumer lab testing platforms.
Direct-to-Consumer Lab Testing
DTC lab companies let you order blood tests online without a doctor's order (in most states), visit a local draw site (Quest, LabCorp, or independent labs), and receive results electronically. Prices are dramatically lower than hospital-based testing, often 50-85% less [21].
Cost Comparison: Hospital vs. DTC Lab Testing
| Test | Hospital/Insurance Billing | DTC Lab Price | Savings |
|---|---|---|---|
| CMP (Comprehensive Metabolic Panel) | $100-250 | $15-35 | 75-90% |
| CBC with Differential | $75-200 | $15-30 | 80-85% |
| Lipid Panel | $75-180 | $15-30 | 80-85% |
| HbA1c | $50-150 | $15-30 | 70-80% |
| Fasting Insulin | $75-200 | $20-35 | 75-85% |
| IGF-1 | $150-350 | $50-80 | 65-80% |
| Thyroid Panel (TSH, fT4, fT3) | $100-300 | $30-55 | 70-82% |
| Total/Free Testosterone | $100-250 | $30-50 | 70-80% |
| hsCRP | $50-150 | $15-30 | 70-80% |
| Comprehensive Peptide Panel (all above) | $800-2,000+ | $150-300 | 75-85% |
Popular DTC Lab Platforms
Several well-established platforms serve the peptide therapy community. All use CLIA-certified and CAP-accredited laboratories, the same labs used by hospitals and physicians:
- Walk-In Lab: Wide test selection including specialized peptide panels. Uses Quest and LabCorp draw sites nationwide. Often has sales and bundle discounts.
- Ulta Lab Tests: Competitive pricing with a large test menu. Detailed result explanations and reference information included.
- Life Extension: Popular among the anti-aging community. Annual blood test sale (usually in spring) offers significant additional discounts.
- Private MD Labs: Good bundle options for hormone and metabolic panels.
- Request A Test: Straightforward ordering with Quest and LabCorp locations.
- Marek Diagnostics: Specializes in panels designed for performance optimization and peptide/hormone therapy monitoring.
Maximizing Value: Building Your Own Panel
Rather than ordering a pre-made "wellness panel" that may include tests you don't need, build a custom panel based on your specific peptide protocol. Here are example panels by use case:
GH Peptide Monitoring Panel (estimated DTC cost: $120-180)
- CMP ($20) + CBC ($20) + IGF-1 ($65) + Fasting Insulin ($25) + HbA1c ($20) + Thyroid Panel ($35)
GLP-1 Monitoring Panel (estimated DTC cost: $80-130)
- CMP ($20) + CBC ($20) + HbA1c ($20) + Lipid Panel ($20) + Fasting Insulin ($25) + Vitamin D ($30)
General Peptide Safety Panel (estimated DTC cost: $50-80)
- CMP ($20) + CBC ($20) + hsCRP ($20) + Lipid Panel ($20)
Insurance Considerations
If your peptide therapy is prescribed by a physician, some or all of the monitoring labs may be covered by insurance, especially if they're ordered for a recognized diagnosis (e.g., metabolic syndrome, insulin resistance, growth hormone deficiency). However:
- Coverage varies widely between plans and may require prior authorization
- High-deductible plans may mean you're paying out of pocket regardless
- Hospital lab prices billed to insurance are often higher than DTC cash prices, even before insurance discounts
- For many patients, DTC testing is both cheaper and more convenient than going through insurance
State Restrictions
A few states restrict or limit direct-to-consumer lab testing. As of 2025-2026, states with restrictions include New York, New Jersey, Rhode Island, and Maryland (though regulations are evolving). In these states, you may need a provider's order to access lab testing. Telehealth providers can often bridge this gap by providing orders for testing.
Money-Saving Tips
Watch for annual sales from DTC lab companies (Life Extension's spring sale is legendary in the optimization community). Bundle tests rather than ordering individually. Some companies offer subscription plans with discounted pricing for regular testing. If you're doing quarterly monitoring, ask about multi-test discount codes. Many peptide clinics include lab work in their service packages, which can be more cost-effective than ordering separately.
Figure 7: Cost comparison between hospital-billed lab testing and direct-to-consumer lab pricing for common peptide monitoring tests. DTC labs can save 65-90% on most tests.
Deep Biomarker Guide: Understanding Every Test in Your Panel
Knowing what tests to order is only the first step. Understanding what each biomarker actually measures, how it's produced in the body, what affects it beyond peptide therapy, and how to interpret subtle changes gives you real mastery over your lab results. This section breaks down every major biomarker relevant to peptide therapy in detail.
Metabolic Biomarkers
Fasting Glucose: More Than Just a Number
Fasting glucose measures the concentration of glucose in your blood after an overnight fast. The liver maintains blood glucose levels during fasting through a process called gluconeogenesis (making new glucose from amino acids and glycerol) and glycogenolysis (breaking down stored glycogen). When you see a fasting glucose result, you're essentially measuring how well your liver is regulating glucose output and how effectively your tissues are clearing glucose from the blood.
For peptide therapy, fasting glucose is particularly informative because growth hormone is a counter-regulatory hormone. It opposes insulin's action by stimulating hepatic glucose output and reducing peripheral glucose uptake. When you use GH-axis peptides, you're deliberately increasing GH levels, and this comes with an inherent risk of elevated glucose. The magnitude depends on the dose, the specific compound, your baseline insulin sensitivity, and your body composition.
A single fasting glucose reading can be misleading. Stress the morning of the draw, poor sleep the night before, or a carb-heavy meal the previous evening can all transiently elevate fasting glucose. This is why trends matter more than isolated values. A pattern of fasting glucose readings slowly creeping upward, from 88 to 93 to 99 to 104 over several months, tells a much clearer story than any single number.
The dawn phenomenon is another factor to be aware of. Some individuals naturally have higher fasting glucose first thing in the morning due to a cortisol-driven spike in hepatic glucose output around 4-6 AM. This can produce fasting glucose readings of 100-115 mg/dL in metabolically healthy people. If your fasting glucose is elevated but your HbA1c and fasting insulin are both optimal, the dawn phenomenon may be the explanation.
Fasting Insulin: The Hidden Story Behind Glucose
Fasting insulin is arguably the most underappreciated test in metabolic health. Most providers focus on glucose, but glucose is a lagging indicator. Your body can maintain normal glucose levels for years while pumping out progressively more insulin to compensate for growing insulin resistance. By the time fasting glucose becomes abnormal, the underlying insulin resistance has been developing for potentially a decade or more.
This is why fasting insulin is so valuable for peptide therapy monitoring. It detects metabolic deterioration years before glucose-based tests would flag a problem. On GH-axis peptides, you might see fasting glucose remain in the normal range while fasting insulin quietly climbs from 5 to 8 to 12 to 16 uIU/mL. Without testing insulin, you'd think everything was fine. With insulin data, you can see the developing resistance and intervene before it progresses.
Insulin also provides context for interpreting glucose readings. A fasting glucose of 95 with a fasting insulin of 4 is very different from a fasting glucose of 95 with a fasting insulin of 18. The first scenario represents excellent metabolic health. The second represents significant insulin resistance that happens to be compensated by high insulin output. The glucose looks the same; the clinical reality is entirely different.
The HOMA-IR calculation (glucose x insulin / 405) integrates both values into a single insulin resistance score. Track HOMA-IR over time as your primary metabolic health metric on GH-axis peptide therapy. It's the most sensitive early warning system you have for metabolic deterioration.
HbA1c: The Three-Month Average
Hemoglobin A1c measures the percentage of hemoglobin molecules in your blood that have glucose attached to them (glycated hemoglobin). Since red blood cells live approximately 90-120 days, HbA1c reflects your average blood glucose over that period. It's like a report card that covers the whole semester rather than a single exam.
For peptide therapy monitoring, HbA1c provides a perspective that no single glucose reading can match. It smooths out the daily fluctuations from meals, exercise, stress, and sleep quality. An HbA1c of 5.4% means your average blood glucose has been approximately 108 mg/dL over the past 2-3 months, regardless of what any individual fasting glucose test showed.
There are some important caveats with HbA1c interpretation. Conditions that affect red blood cell lifespan can skew results. Iron deficiency anemia (common during weight loss on GLP-1 therapy) can falsely elevate HbA1c because red blood cells live longer and accumulate more glycated hemoglobin. Conversely, conditions with increased red blood cell turnover (recent blood loss, hemolytic anemia) can falsely lower HbA1c. If your HbA1c doesn't match your glucose trends, discuss potential confounders with your provider [2].
On GLP-1 therapy, HbA1c serves as both a safety marker and a progress tracker. Diabetic patients can often document dramatic HbA1c improvements that support continued therapy. Non-diabetic patients on GLP-1s for weight loss may see modest reductions that confirm improved metabolic function. On GH-axis peptides, a rising HbA1c is an early warning that glucose management is deteriorating and protocol adjustments are needed.
Liver Biomarkers in Detail
ALT (Alanine Aminotransferase)
ALT is the most liver-specific of the common liver enzymes. While AST is found in liver, muscle, heart, kidney, and brain tissue, ALT is predominantly found in hepatocytes (liver cells). When hepatocytes are damaged or inflamed, they leak ALT into the bloodstream. The degree of elevation roughly correlates with the extent of liver injury, though this isn't a perfect relationship.
For peptide therapy monitoring, ALT serves as your primary liver safety sentinel. The standard reference range goes up to 56 U/L for most labs, but research suggests that truly healthy ALT levels are considerably lower. Studies of large healthy populations have found that the upper limit of normal should probably be around 30-35 U/L for men and 19-25 U/L for women. Many functional medicine practitioners use 25 U/L as their upper optimal target for both sexes.
What does an elevated ALT actually mean? There are many possible causes beyond peptide-related liver injury:
- NAFLD/NASH: Non-alcoholic fatty liver disease is the most common cause of mildly elevated ALT in the general population. Obesity, insulin resistance, and high triglycerides are the primary drivers. GLP-1 therapy typically improves NAFLD, so if you're on semaglutide or tirzepatide, expect ALT to trend downward.
- Exercise: Intense resistance training or endurance exercise can elevate ALT for 24-72 hours. This is especially true for AST, which is abundant in muscle, but ALT can also rise modestly. Always note your exercise history when reviewing liver results.
- Alcohol: Even moderate alcohol consumption (2-3 drinks) in the 48 hours before testing can raise ALT. Chronic alcohol use obviously has larger effects.
- Supplements: Many over-the-counter supplements can stress the liver. Green tea extract in high doses, kava, and excessive vitamin A are common culprits. Review your supplement stack if ALT is unexpectedly elevated.
- Medications: Statins, acetaminophen (Tylenol), and certain antibiotics can elevate liver enzymes. Check for drug interactions.
- Viral hepatitis: Hepatitis B and C cause chronic ALT elevation. Baseline screening is appropriate if you have risk factors.
AST (Aspartate Aminotransferase)
AST is less liver-specific than ALT because it's found in multiple tissues. However, it provides additional information when interpreted alongside ALT. The AST/ALT ratio can help distinguish between different causes of liver enzyme elevation:
- AST/ALT ratio below 1: Typical of NAFLD and most hepatic causes. This is the pattern you'll most commonly see in peptide users.
- AST/ALT ratio above 2: Suggests alcoholic liver disease. If you're not drinking significantly, this pattern in a peptide user warrants further evaluation.
- Isolated AST elevation with normal ALT: Almost always muscular in origin. Check CK (creatine kinase) to confirm. Very common in people who exercise regularly.
GGT (Gamma-Glutamyl Transferase)
GGT isn't included in the standard CMP but is a valuable add-on test. It's elevated in bile duct disease, alcohol use, and oxidative stress. In the context of peptide therapy monitoring, GGT serves two purposes:
- Differentiating liver enzyme elevations: If ALT is elevated, adding GGT helps determine whether the cause is hepatocellular (GGT often normal) or cholestatic/biliary (GGT elevated alongside ALP).
- Oxidative stress marker: GGT is independently associated with oxidative stress and cardiovascular risk, even at levels within the standard reference range. Optimal GGT is below 25 U/L. Anti-aging peptide protocols that target oxidative stress (NAD+, MOTS-c, Epithalon) should produce a declining GGT trend.
Kidney Biomarkers in Detail
Creatinine and eGFR
Serum creatinine is a waste product of muscle metabolism. Your kidneys filter it from the blood at a relatively constant rate, so rising creatinine suggests declining kidney filtration capacity. The eGFR (estimated glomerular filtration rate) is a calculation that uses creatinine, age, sex, and sometimes race to estimate how many milliliters of blood your kidneys filter per minute.
For peptide therapy, kidney monitoring is most relevant for GLP-1 users because dehydration from GI side effects can acutely impair kidney function. It's also important for anyone on multiple medications, as drug-drug interactions can affect renal perfusion.
Important confounders for creatinine/eGFR:
- Muscle mass: Muscular individuals naturally produce more creatinine. A bodybuilder with a creatinine of 1.3 mg/dL may have perfectly normal kidney function. The eGFR calculation doesn't fully account for extreme muscle mass.
- Creatine supplementation: Supplemental creatine is partially converted to creatinine in the body. If you take creatine, your serum creatinine will be higher than it would otherwise be, and your eGFR will appear falsely low. Inform your provider if you supplement with creatine.
- Dehydration: Even mild dehydration can transiently raise creatinine and lower eGFR. Always hydrate normally before testing.
- High-protein diet: Acute protein loading can transiently raise BUN and, to a lesser extent, creatinine. Fasting helps standardize this.
Cystatin C is an alternative kidney function marker that isn't affected by muscle mass or creatine supplementation. If your creatinine/eGFR results are questionable due to these confounders, asking for cystatin C provides a more accurate assessment [24].
BUN (Blood Urea Nitrogen)
BUN measures the nitrogen content of urea, a waste product of protein metabolism. The kidneys excrete urea, so rising BUN can indicate declining kidney function. However, BUN is also affected by protein intake, dehydration, GI bleeding, and catabolic states (where the body is breaking down protein rapidly).
The BUN/creatinine ratio can provide additional diagnostic information. A ratio above 20:1 suggests dehydration, upper GI bleeding, or high protein intake. A ratio below 10:1 might indicate liver disease (reduced urea production) or low protein intake. For GLP-1 users experiencing nausea and reduced food intake, a low BUN with a low BUN/creatinine ratio might indicate inadequate protein intake, which is worth addressing to prevent muscle loss.
Thyroid Biomarkers in Detail
TSH (Thyroid-Stimulating Hormone)
TSH is produced by the pituitary gland and stimulates the thyroid to produce thyroid hormones. It operates on a negative feedback loop: when thyroid hormone levels drop, TSH rises (stimulating more production), and when thyroid hormone levels rise, TSH drops. This makes TSH a sensitive screening test for thyroid dysfunction.
The standard reference range for TSH (0.45-4.5 mIU/L) is controversially broad. Many endocrinologists and functional medicine practitioners consider TSH above 2.5 mIU/L as suboptimal, even though it falls within the standard range. For peptide therapy monitoring, the TSH trend is more informative than any single value. A TSH climbing from 1.8 to 2.5 to 3.4 over six months on GH peptide therapy suggests emerging hypothyroidism that should be addressed.
Free T4 and Free T3
TSH alone doesn't tell the full thyroid story. Free T4 (the storage form of thyroid hormone) and free T3 (the active form) provide direct measurement of circulating thyroid hormones. The ratio between them reveals whether your body is efficiently converting T4 to T3.
GH-axis peptides can alter thyroid hormone metabolism by increasing the conversion of T4 to T3 in peripheral tissues. This can unmask subclinical hypothyroidism in patients who had borderline thyroid function before starting therapy. The mechanism is straightforward: increased T4-to-T3 conversion depletes T4 stores, which triggers a TSH rise. If the thyroid can't increase production enough to compensate, hypothyroid symptoms develop.
If you notice symptoms like fatigue, cold intolerance, constipation, weight gain, or dry skin after starting GH peptide therapy, check a full thyroid panel (TSH, free T4, free T3, and thyroid antibodies). Don't assume these symptoms are unrelated to your peptide protocol [9].
Growth Hormone Axis Biomarkers
IGF-1: The Complete Picture
We covered IGF-1 targets earlier, but understanding the biology helps you interpret results more effectively. IGF-1 is produced primarily by the liver in response to growth hormone stimulation. After GH binds to receptors on hepatocytes, the liver synthesizes and releases IGF-1 into the bloodstream. IGF-1 then acts on tissues throughout the body to promote growth, repair, and anabolic processes.
Several factors beyond GH peptide therapy affect IGF-1 levels:
- Nutritional status: IGF-1 is profoundly affected by caloric and protein intake. During caloric restriction (common on GLP-1 therapy), IGF-1 naturally declines even without any change in GH secretion. A patient on semaglutide who is also taking CJC-1295/Ipamorelin might see a blunted IGF-1 response because their reduced caloric intake is suppressing hepatic IGF-1 production.
- Liver health: Since the liver produces IGF-1, liver disease can impair IGF-1 production. If IGF-1 doesn't respond to GH peptide therapy and liver enzymes are elevated, the liver's synthetic capacity may be compromised.
- Sleep: GH is primarily released during deep sleep (stages 3 and 4 of NREM sleep). Poor sleep quality blunts GH secretion and can limit IGF-1 response to peptide therapy. Many clinicians find that addressing sleep quality is as important as the peptide itself for optimizing IGF-1 levels.
- Age: IGF-1 naturally declines with age. A 60-year-old may not achieve the same absolute IGF-1 levels as a 30-year-old on the same peptide dose. Use age-adjusted targets rather than absolute numbers.
- Body composition: Obesity and excess visceral fat are associated with lower IGF-1 levels due to hepatic insulin resistance affecting GH signaling. As body composition improves, IGF-1 response to peptides often improves as well.
- Insulin levels: Insulin and GH have a complex relationship. High insulin levels can suppress GH receptor expression in the liver, blunting IGF-1 production. This is another reason why insulin-resistant patients may show a weaker IGF-1 response to GH peptides.
IGFBP-3 (IGF Binding Protein 3)
IGFBP-3 is the primary carrier protein for IGF-1 in the blood. It binds approximately 80% of circulating IGF-1 and extends its half-life. In clinical practice, IGFBP-3 is sometimes measured alongside IGF-1 for a more complete picture of the GH axis, particularly in growth hormone deficiency assessment.
For peptide therapy monitoring, IGFBP-3 is optional. It's most useful when IGF-1 results are discordant with clinical response (e.g., IGF-1 is low but the patient is showing signs of adequate GH action). An elevated IGFBP-3 with a low IGF-1 might suggest that IGF-1 is being bound and sequestered rather than being freely available to tissues.
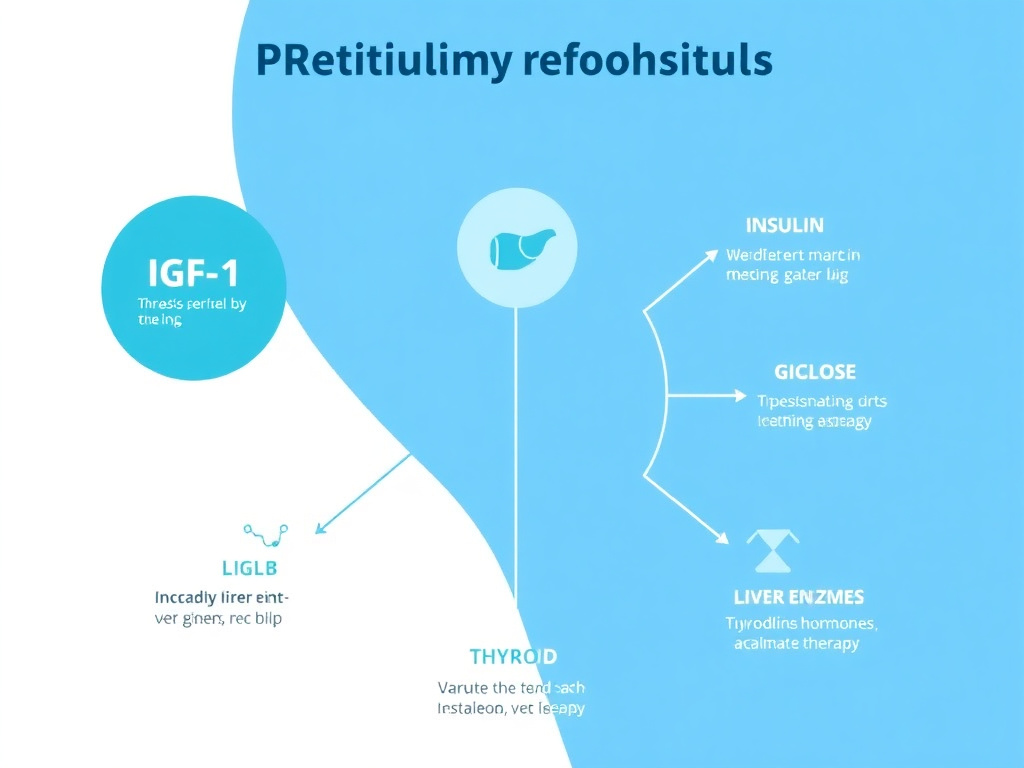
Figure 8: Biological relationships between major peptide therapy biomarkers. Understanding how these markers interact helps you interpret results in context rather than in isolation.
Practical Scenarios: Blood Work Interpretation in Real-World Contexts
Understanding biomarkers in theory is one thing. Seeing how they play out in real clinical scenarios brings the knowledge to life. These composite case scenarios illustrate common patterns you might encounter when monitoring peptide therapy blood work.
Scenario 1: The GH Peptide User With Rising Glucose
Background: A 45-year-old male starts CJC-1295/Ipamorelin (100 mcg each, nightly) for anti-aging and body composition goals. He has no history of diabetes, BMI is 27, and he exercises 4x per week.
| Marker | Baseline | 6 Weeks | 3 Months |
|---|---|---|---|
| IGF-1 | 158 ng/mL | 247 ng/mL | 268 ng/mL |
| Fasting Glucose | 91 mg/dL | 97 mg/dL | 108 mg/dL |
| Fasting Insulin | 5.2 uIU/mL | 7.8 uIU/mL | 11.4 uIU/mL |
| HOMA-IR | 1.17 | 1.87 | 3.04 |
| HbA1c | 5.3% | - | 5.5% |
Interpretation: The peptide is clearly working - IGF-1 has risen from the low-normal range (158) to the optimal range (268). However, there's a concerning trend in metabolic markers. Fasting glucose has gone from 91 (excellent) to 108 (impaired fasting glucose). More tellingly, fasting insulin has more than doubled, and HOMA-IR has crossed the 3.0 threshold, indicating significant insulin resistance. HbA1c has risen modestly but is still normal.
Action Plan: The IGF-1 response is excellent, so the peptide dose doesn't need to increase. The metabolic deterioration needs addressing. Options include: reducing the CJC-1295/Ipamorelin dose by 25-50% and rechecking glucose/insulin in 4-6 weeks; optimizing diet (lower glycemic load, time-restricted eating); increasing cardiovascular exercise; or adding berberine or metformin if lifestyle changes alone aren't sufficient. If HOMA-IR doesn't improve within 6-8 weeks of intervention, consider discontinuing or switching to a peptide with less glucose impact like sermorelin.
Scenario 2: The GLP-1 Patient With Electrolyte Concerns
Background: A 52-year-old female starts semaglutide 0.25 mg weekly for weight loss (BMI 34). She titrates to 1.0 mg weekly over 2 months. She experiences moderate nausea and occasional vomiting during titration.
| Marker | Baseline | 6 Weeks | 3 Months |
|---|---|---|---|
| Weight | 198 lbs | 189 lbs | 179 lbs |
| HbA1c | 5.8% | - | 5.4% |
| Fasting Glucose | 104 mg/dL | 92 mg/dL | 87 mg/dL |
| ALT | 42 U/L | 35 U/L | 24 U/L |
| Triglycerides | 188 mg/dL | 142 mg/dL | 118 mg/dL |
| Potassium | 4.2 mEq/L | 3.6 mEq/L | 3.4 mEq/L |
| Sodium | 140 mEq/L | 138 mEq/L | 136 mEq/L |
| Creatinine | 0.8 mg/dL | 0.9 mg/dL | 0.85 mg/dL |
| Albumin | 4.2 g/dL | 4.0 g/dL | 3.8 g/dL |
Interpretation: The metabolic response is excellent across the board. HbA1c improved from prediabetic (5.8%) to normal (5.4%). Fasting glucose normalized. ALT dropped dramatically, likely reflecting improvement in fatty liver as weight decreased. Triglycerides improved by 37%. However, potassium is trending downward (4.2 to 3.4), likely from GI losses due to nausea and vomiting. Sodium is also drifting lower. Albumin has declined modestly, suggesting possible inadequate protein intake with reduced appetite. Creatinine bumped slightly at 6 weeks, likely from dehydration during the worst of GI symptoms, then recovered.
Action Plan: Continue semaglutide; the metabolic improvements are substantial. Address the declining potassium by supplementing and ensuring adequate dietary intake (bananas, avocados, sweet potatoes). If potassium drops below 3.0 mEq/L, this becomes a serious concern requiring immediate medical attention (cardiac arrhythmia risk). The albumin decline suggests she needs to prioritize protein intake (aim for at least 0.7 grams per pound of body weight) to prevent muscle loss during weight loss. Consider adding a protein supplement if appetite is severely suppressed. Monitor electrolytes more frequently (monthly) until GI symptoms resolve and potassium stabilizes.
Scenario 3: The MK-677 User With Multiple Red Flags
Background: A 38-year-old male uses MK-677 25 mg daily for 4 months for muscle building. He has a family history of type 2 diabetes (both parents diabetic). He didn't get baseline labs.
| Marker | 4 Months (First Test) | Notes |
|---|---|---|
| IGF-1 | 388 ng/mL | High; suggests strong GH-axis stimulation |
| Fasting Glucose | 132 mg/dL | Diagnostic for impaired fasting glucose/possible diabetes |
| Fasting Insulin | 24.2 uIU/mL | At the very top of the standard reference range; clearly insulin resistant |
| HOMA-IR | 7.88 | Severely insulin resistant |
| HbA1c | 6.2% | Prediabetic range |
| Prolactin | 22.4 ng/mL | Mildly elevated for a male (normal below 15) |
| ALT | 58 U/L | Mildly above reference range |
Interpretation: This is a troubling picture. Without baseline labs, we can't know how much of this is from MK-677 versus pre-existing metabolic issues. However, the combination of a fasting glucose of 132, HbA1c of 6.2%, fasting insulin of 24.2, and HOMA-IR of 7.88 represents severe metabolic deterioration. Given his family history of diabetes, MK-677 may have pushed an already vulnerable metabolic system over the edge. The elevated prolactin and mildly high ALT add further concerns. The IGF-1 of 388 is higher than most clinicians would target, suggesting the dose may be excessive.
Action Plan: Stop MK-677 immediately. This level of metabolic disruption requires medical management, not just protocol adjustment. The patient needs a formal diabetes workup including an oral glucose tolerance test. Recheck fasting glucose, insulin, and HbA1c in 6-8 weeks after discontinuation to see how much improvement occurs. Many of MK-677's metabolic effects are reversible after discontinuation, but this patient may have unmasked an underlying predisposition to diabetes that will require ongoing management. This scenario powerfully illustrates why baseline labs are essential and why patients with diabetes risk factors need extra caution with GH-axis stimulating compounds.
Scenario 4: The Healing Peptide Protocol With Tracking Data
Background: A 35-year-old female uses BPC-157 250 mcg twice daily (SubQ near the injury) for a partial hamstring tear, combined with TB-500 2 mg twice weekly, for 6 weeks.
| Marker | Baseline | 6 Weeks |
|---|---|---|
| hsCRP | 4.8 mg/L | 1.2 mg/L |
| ESR | 22 mm/hr | 8 mm/hr |
| ALT | 19 U/L | 21 U/L |
| AST | 22 U/L | 24 U/L |
| Creatinine | 0.7 mg/dL | 0.7 mg/dL |
| CBC | All normal | All normal |
Interpretation: Textbook results for a healing peptide protocol. The hsCRP dropped from 4.8 (indicating significant inflammation, consistent with an acute injury) to 1.2 (low risk range). ESR also normalized. Liver and kidney markers are stable, showing no adverse effects from the injectable peptides. CBC is unchanged. The inflammatory marker improvements correlate with objective clinical improvement in her hamstring function (which she should also be tracking through range-of-motion and strength testing).
Action Plan: Complete the 6-week protocol as planned. The safety labs are reassuring, and the inflammatory marker improvements provide objective evidence that the protocol is working. No dose adjustments needed. If she wants to extend the protocol beyond 6 weeks, recheck CMP and CRP at 8-10 weeks.
Scenario 5: The Comprehensive Stack With Complex Monitoring
Background: A 50-year-old male on a comprehensive peptide stack for anti-aging: CJC-1295/Ipamorelin (nightly), semaglutide 1.0 mg weekly (weight management), BPC-157 250 mcg daily (gut health), and Epithalon 10mg cycling (anti-aging). He started compounds sequentially over 3 months. Visit our stacking guide for more on combination protocols.
| Marker | Baseline | 3 Months | 6 Months |
|---|---|---|---|
| IGF-1 | 162 ng/mL | 238 ng/mL | 255 ng/mL |
| HbA1c | 5.7% | 5.3% | 5.1% |
| Fasting Glucose | 102 mg/dL | 94 mg/dL | 88 mg/dL |
| Fasting Insulin | 8.4 uIU/mL | 5.8 uIU/mL | 4.2 uIU/mL |
| HOMA-IR | 2.11 | 1.35 | 0.91 |
| Triglycerides | 172 mg/dL | 128 mg/dL | 104 mg/dL |
| ALT | 38 U/L | 28 U/L | 22 U/L |
| hsCRP | 3.2 mg/L | 1.8 mg/L | 0.9 mg/L |
| Weight | 218 lbs | 201 lbs | 192 lbs |
Interpretation: This represents an ideal multi-peptide response. IGF-1 has risen to the optimal range (255) from the CJC-1295/Ipamorelin. The potentially glucose-raising effects of the GH peptide are more than offset by semaglutide's glucose-lowering effects, resulting in dramatically improved metabolic markers: HbA1c down from prediabetic (5.7%) to optimal (5.1%), fasting glucose normalized, insulin and HOMA-IR improved to excellent levels. The semaglutide is also driving weight loss, lipid improvements, and liver enzyme normalization. hsCRP has dropped into the optimal range, likely reflecting the combined anti-inflammatory effects of weight loss, improved metabolic health, BPC-157's gut healing effects, and Epithalon's anti-inflammatory properties.
Action Plan: Continue the current protocol. This is the best-case scenario for a comprehensive stack. Schedule next labs at 9 months and 12 months. At the annual mark, do a complete comprehensive panel including thyroid, hormone panel (testosterone, estradiol for a 50-year-old male), nutritional markers (B12, vitamin D, iron), and homocysteine. Use the peptide calculator to verify dosing remains appropriate for his reduced body weight.
How to Read Your Lab Report: A Visual Guide
Lab reports can look intimidating with their columns of numbers, reference ranges, and flags. Here's a practical guide to reading your results like a clinician.
Understanding Lab Report Format
Most lab reports follow a standard format with these columns:
- Test Name: The specific biomarker being measured
- Result: Your measured value
- Units: The unit of measurement (mg/dL, ng/mL, U/L, etc.)
- Reference Range: The lab's "normal" range
- Flag: H (high), L (low), or blank (within range)
What the Flags Mean (and Don't Mean)
A flag of "H" or "L" means your value falls outside the lab's reference range. This does not automatically mean something is wrong. As we've discussed, reference ranges are based on the general population and may not reflect optimal health. Conversely, the absence of a flag doesn't guarantee everything is optimal. A fasting insulin of 18 uIU/mL won't be flagged as high (reference range goes to ~25), but it represents meaningful insulin resistance.
This is why relying solely on flags is insufficient for peptide therapy monitoring. You need to compare results against both the standard reference range and the functional/optimal ranges discussed earlier in this guide. And most critically, you need to compare against your own baseline values to identify trends.
Creating Your Personal Tracking System
Most DTC lab companies provide online dashboards where you can view historical results. However, creating your own tracking spreadsheet gives you more flexibility and better visualization. Here's what to include:
- Date of draw
- Which lab processed the sample (important for consistency)
- Fasting status (yes/no, hours fasted)
- Current peptide protocol (compounds, doses, duration at time of draw)
- All result values
- Notes column (any variables that might affect results: exercise, illness, alcohol, poor sleep, stress, supplement changes)
- Your personal optimal range for each marker (so you can quickly see if you're in target)
Over time, this tracking document becomes your most valuable health asset. It reveals patterns and trends that no single lab report can show. Many patients find that graphing their key metrics (IGF-1, fasting glucose, HOMA-IR, hsCRP) over time provides powerful visual feedback on their protocol's effectiveness.
Figure 9: Example personal tracking spreadsheet for peptide therapy blood work. Graphing key markers over time reveals trends that individual lab reports miss.
When Results From Different Labs Don't Match
If you switch lab companies or draw sites, you may notice differences in results for the same biomarker. This doesn't necessarily mean either result is wrong. Different laboratories use different analytical methods (immunoassay vs. mass spectrometry, different reagent manufacturers), and these can produce systematically different values.
The most common discrepancies occur with:
- IGF-1: Immunoassay results can vary by 15-25% between different platforms. If you switch labs, expect some variability and establish a new trend rather than comparing absolute values to your old lab [6].
- Testosterone (free): Calculated free testosterone (from total T and SHBG) often differs from directly measured free testosterone (equilibrium dialysis or ultrafiltration). Know which method your lab uses.
- Estradiol: The standard immunoassay is unreliable at low levels (typical in men). The sensitive estradiol test (LC-MS/MS) is more accurate for male patients.
The solution is simple: use the same laboratory for all your serial testing whenever possible. If you must switch, note it in your tracking spreadsheet and start a fresh trend line.
Special Population Considerations
Blood work monitoring isn't one-size-fits-all. Certain populations require additional tests, different reference ranges, or modified monitoring frequency. Here's what to keep in mind for specific groups.
Women of Reproductive Age
Menstrual cycle phase affects several biomarkers. Testosterone, estradiol, progesterone, LH, and FSH all fluctuate significantly throughout the cycle. For the most consistent results:
- Hormone panels should be drawn during the early follicular phase (days 2-5 of the menstrual cycle) for baseline values
- Progesterone should be drawn at day 21 (mid-luteal phase) if assessing ovulation
- Iron and ferritin are particularly important to monitor during peptide therapy for women with heavy periods, as blood loss combined with reduced appetite on GLP-1 therapy can quickly deplete iron stores
- Pregnancy testing should be performed before starting any peptide therapy, as safety data in pregnancy is limited or absent for most peptides
Adults Over 65
Older adults on peptide therapy require additional considerations:
- IGF-1 targets should be lower: Age-adjusted ranges for a 70-year-old are significantly lower than for a 40-year-old. Pushing IGF-1 to levels appropriate for a younger person in an older adult may increase cardiovascular and oncologic risks.
- Kidney function declines naturally with age: An eGFR of 55 mL/min in a 75-year-old may reflect normal age-related decline rather than kidney disease. However, GLP-1 therapy and dehydration risk are higher in older adults, so more frequent monitoring is appropriate.
- Glucose tolerance naturally worsens with age: The threshold for concern about fasting glucose and HbA1c on GH-axis peptides should be lower in older adults, as their metabolic reserve is diminished.
- Fall risk from hypoglycemia: Older adults on GLP-1 therapy who also take sulfonylureas or insulin need careful glucose monitoring, as hypoglycemia-related falls are a significant source of morbidity.
- Cancer screening: Before starting any GH-axis peptide, ensure age-appropriate cancer screening is up to date (colonoscopy, mammography, PSA, etc.), given IGF-1's role as a growth factor [25].
Patients With Pre-Existing Conditions
Type 2 Diabetes
Diabetic patients on GLP-1 therapy need more frequent glucose monitoring, especially if they're also on insulin or sulfonylureas (hypoglycemia risk). HbA1c should be checked every 3 months until stable. Consider continuous glucose monitoring (CGM) for real-time data that lab testing can't provide. GH-axis peptides require extreme caution in diabetic patients due to their glucose-raising effects.
Chronic Kidney Disease
Patients with CKD (eGFR below 60) need monthly kidney function monitoring during the first 3 months of any peptide therapy. Dose adjustments may be needed for compounds that affect renal perfusion. GLP-1 agonists can actually benefit kidney function in some CKD patients, but careful monitoring is essential.
Liver Disease
Pre-existing liver disease requires more frequent liver enzyme monitoring (monthly initially). Advanced liver disease (cirrhosis) affects IGF-1 production and may alter the metabolism of peptide compounds. Consult with a hepatologist before starting peptide therapy in patients with significant liver disease.
Autoimmune Conditions
Patients with autoimmune disease using immune-modulating peptides (Thymosin Alpha-1, LL-37, VIP) need tracking of disease-specific autoantibodies alongside standard immune panels. The goal is to confirm that immune modulation is producing beneficial effects without triggering autoimmune flares. Consult with a rheumatologist or immunologist familiar with peptide therapy for complex autoimmune cases.
Athletes and Highly Active Individuals
Athletic individuals present unique monitoring challenges:
- Liver enzymes (AST, ALT): Regularly elevated from exercise-induced muscle breakdown, not liver injury. CK testing helps distinguish the source. Consider a 72-hour exercise washout period before lab draws if you want clean liver values.
- Creatinine/eGFR: Elevated creatinine from muscle mass and creatine supplementation can give falsely low eGFR. Cystatin C is a better kidney marker for muscular individuals.
- Testosterone: Overtraining syndrome can suppress testosterone, which may confound interpretation of GH peptide effects on hormonal balance.
- Iron: Endurance athletes, especially runners, are prone to exercise-induced hemolysis and iron loss through sweat and GI microbleeding. Monitor ferritin closely.
- Hematocrit: Already elevated in many endurance athletes from training adaptation. GH peptides can further increase red blood cell production. Monitor hematocrit and maintain adequate hydration.
The Future of Peptide Therapy Monitoring
Lab testing technology is evolving rapidly, and several emerging tools promise to make peptide therapy monitoring more convenient, more frequent, and more informative than traditional blood draws alone.
Continuous Glucose Monitoring (CGM)
CGM devices like the Dexcom G7, Abbott Libre 3, and Stelo provide real-time glucose data every 1-5 minutes via a small sensor worn on the skin. For peptide therapy monitoring, CGM offers unparalleled insight into glucose dynamics that lab-based fasting glucose simply can't match.
With CGM, you can see:
- How MK-677 or CJC-1295 affects your glucose overnight and around meals
- The real-time impact of GLP-1 therapy on postprandial glucose spikes
- The dawn phenomenon and its contribution to elevated morning fasting glucose
- How exercise, sleep, and stress interact with your peptide protocol to affect glucose
- Time-in-range data (percentage of time glucose stays between 70-140 mg/dL), which is arguably more useful than any single fasting glucose or HbA1c value
CGM doesn't replace blood work, but it provides a real-time layer of data between lab draws that dramatically improves your ability to optimize protocols. Many anti-aging clinics and functional medicine providers now recommend 2-week CGM trials at the start of GH-axis peptide therapy and periodically during treatment [20].
At-Home Blood Testing
Several companies now offer at-home blood testing kits that use finger-prick capillary blood samples rather than venous draws at a lab. While the test menu is more limited and some analytes (like IGF-1) aren't available via finger prick, basic metabolic panels, HbA1c, lipids, and hsCRP can be measured from home. This lowers the barrier to frequent testing, especially for patients who can't easily visit a draw site.
Advanced Biomarkers on the Horizon
Several emerging biomarkers may become standard components of peptide therapy monitoring panels in the coming years:
- ApoB (Apolipoprotein B): A more accurate measure of atherogenic lipoprotein particles than LDL cholesterol. May better capture the cardiovascular risk reduction from GLP-1 therapy.
- GDF-15 (Growth Differentiation Factor 15): A stress-response biomarker associated with GLP-1 receptor agonist activity. May serve as a pharmacodynamic marker for GLP-1 dose optimization.
- Telomere length and epigenetic clocks: Direct measures of biological aging that could objectively assess the impact of anti-aging peptide protocols over months to years.
- Gut microbiome testing: As the connection between peptide therapy (especially oral BPC-157 and GLP-1 agonists) and the microbiome becomes clearer, microbiome analysis may become a standard monitoring tool.
- Metabolomics panels: Comprehensive metabolic profiling that captures hundreds of metabolites in a single test, providing a systems-level view of how peptide therapy is affecting your metabolism.
For now, these advanced tools are optional and often expensive. The core monitoring panels described in this guide, built around the CMP, CBC, lipid panel, HbA1c, fasting insulin, IGF-1, hsCRP, and thyroid function, remain the gold standard for safe and effective peptide therapy monitoring. Master these fundamentals first, and add advanced testing as your protocol evolves and your comfort with lab interpretation grows.
For the latest on peptide therapy research and protocols, explore our complete peptide research hub and the full beginner's guide to peptide therapy.
Figure 10: Emerging technologies for peptide therapy monitoring, from continuous glucose monitors to at-home blood testing kits and advanced biomarker panels.
Advanced Monitoring Strategies for Experienced Peptide Users
Once you've mastered the fundamentals of peptide blood work monitoring, you may want to go deeper. Advanced strategies involve more sophisticated biomarkers, more granular timing, and a systems-thinking approach that considers how multiple organ systems interact under peptide therapy.
The Cardiometabolic Deep Panel
Standard lipid panels (total cholesterol, LDL, HDL, triglycerides) provide a useful but incomplete picture of cardiovascular risk. For peptide users pursuing comprehensive health optimization, an advanced cardiometabolic panel adds several powerful markers:
ApoB (Apolipoprotein B)
ApoB is increasingly recognized as the best single measure of atherogenic lipoprotein burden. Every LDL particle, VLDL particle, IDL particle, and Lp(a) particle carries exactly one ApoB molecule. Counting ApoB gives you the total number of artery-damaging particles circulating in your blood, regardless of how much cholesterol each one carries.
This matters for peptide users because GLP-1 therapy can change LDL particle size and density without always changing the LDL cholesterol number. You might see your LDL cholesterol stay at 120 mg/dL while your ApoB drops from 110 to 85 mg/dL, indicating a real reduction in atherogenic particles that the standard LDL test missed.
- Optimal: Below 80 mg/dL (below 60 mg/dL for high-risk patients)
- Testing frequency: Every 6-12 months
- Cost: $25-40 through DTC labs
Lp(a) - Lipoprotein(a)
Lp(a) is a genetically determined lipoprotein that significantly increases cardiovascular risk. It's largely unaffected by lifestyle or most medications, but it's worth knowing your level because it affects how aggressively you should pursue other risk factor modifications. Lp(a) only needs to be tested once since it's genetically fixed, unless you're on a therapy specifically targeting it.
- Desirable: Below 30 mg/dL (or below 75 nmol/L, depending on the assay)
- Testing frequency: Once (lifetime screening)
Oxidized LDL
Oxidized LDL (oxLDL) is a more atherogenic form of LDL that directly damages arterial walls. Anti-aging peptides that reduce oxidative stress (NAD+, MOTS-c, GHK-Cu) may lower oxLDL levels. Testing before and after anti-aging peptide protocols can provide evidence of reduced oxidative vascular damage.
Insulin and HOMA-IR in Greater Detail
We've discussed HOMA-IR as a single calculation, but there are more sophisticated ways to assess insulin resistance that may be relevant for advanced monitoring:
The Triglyceride/HDL ratio: This simple calculation from your standard lipid panel correlates strongly with insulin resistance. A TG/HDL ratio above 3.0 suggests insulin resistance; below 1.0 is optimal. It's a free data point from a test you're already running.
The Kraft insulin assay: Also known as the 4-hour insulin response test, this extended oral glucose tolerance test with concurrent insulin measurements at 0, 30, 60, 120, 180, and 240 minutes reveals insulin response patterns that fasting insulin alone misses. It can detect hyperinsulinemia decades before fasting glucose or HbA1c become abnormal. While not practical for routine monitoring, a single Kraft test at baseline provides unmatched insight into your metabolic health.
Comprehensive Hormone Assessment
The Complete Male Hormone Panel
For men on GH-axis peptides, hormonal peptides, or comprehensive anti-aging protocols, a thorough hormone assessment includes:
| Marker | Optimal Range | Why It Matters |
|---|---|---|
| Total Testosterone | 600-900 ng/dL | GH peptides indirectly support testosterone; MK-677 prolactin can suppress it |
| Free Testosterone | 15-25 pg/mL | Biologically active fraction; affected by SHBG changes on GH therapy |
| SHBG | 20-50 nmol/L | GH peptides can lower SHBG, freeing up testosterone |
| Estradiol (sensitive) | 20-35 pg/mL | Aromatization marker; rises with testosterone |
| DHT (Dihydrotestosterone) | 30-85 ng/dL | 5-alpha-reductase activity; relevant for prostate and hair concerns |
| Prolactin | 4-15 ng/mL | Elevated by GHRPs, MK-677; suppresses gonadal function |
| LH | 1.8-8.6 mIU/mL | Pituitary signaling to testes |
| FSH | 1.5-12.4 mIU/mL | Spermatogenesis and gonadal function |
| DHEA-S | 280-640 ug/dL (age 30-40) | Adrenal function and hormonal aging |
| Cortisol (AM) | 10-20 ug/dL | Adrenal stress response; can be affected by GHRPs |
| Pregnenolone | 10-200 ng/dL | Master precursor hormone; marker of hormonal vitality |
The Complete Female Hormone Panel
For women on peptide therapy, hormonal assessment requires attention to menstrual cycle timing and includes different reference values:
| Marker | When to Draw | Key Considerations |
|---|---|---|
| Estradiol | Day 3 of cycle (follicular phase baseline) | Affected by body composition changes on GLP-1 therapy |
| Progesterone | Day 21 of cycle (mid-luteal) | Confirms ovulation; important for fertility-age women on peptides |
| Total Testosterone | Day 3 of cycle | Normal range much lower than men (15-70 ng/dL) |
| DHEA-S | Any time | Adrenal androgen production |
| FSH | Day 3 of cycle | Elevated FSH suggests diminished ovarian reserve |
| LH | Day 3 of cycle | LH/FSH ratio elevated in PCOS |
| TSH, Free T4, Free T3 | Any time | Thyroid dysfunction more common in women; GH peptides can unmask it |
| Prolactin | Any time (avoid breast stimulation before draw) | Elevated by certain peptides; affects menstrual regularity |
| Sex Hormone Binding Globulin (SHBG) | Any time | Low in insulin resistance/PCOS; may change with metabolic improvement on GLP-1 |
Women using peptide therapy for weight loss should be aware that rapid weight loss can affect hormone levels. Fat tissue produces estrogen through aromatization. As body fat decreases on GLP-1 therapy, estrogen levels may change, potentially affecting menstrual regularity. Track menstrual cycle changes alongside blood work for a complete picture.
Oxidative Stress and Cellular Health Markers
For patients on anti-aging peptide protocols, markers of oxidative stress and cellular health provide a direct window into the fundamental aging processes these peptides target:
8-OHdG (8-Hydroxy-2'-Deoxyguanosine)
8-OHdG is a marker of oxidative DNA damage. It's measured in urine and reflects the rate at which reactive oxygen species are damaging your DNA. Anti-aging peptides like Epithalon, NAD+, and MOTS-c that target oxidative stress should produce a declining 8-OHdG trend. Available through specialized labs for $50-100.
F2-Isoprostanes
F2-isoprostanes are considered the gold standard biomarker for oxidative stress. They're produced when free radicals attack arachidonic acid in cell membranes. Declining F2-isoprostanes provide objective evidence that an anti-aging protocol is reducing lipid peroxidation at the cellular level.
Glutathione (Reduced/Oxidized Ratio)
Glutathione is the body's master antioxidant. The ratio of reduced (active) to oxidized (depleted) glutathione reflects your overall antioxidant capacity. NAD+ therapy supports glutathione recycling, and several peptides indirectly support glutathione synthesis. Track the GSH/GSSG ratio at 6-month intervals on anti-aging protocols.
Gut Health Markers for Oral Peptide Protocols
For patients using oral BPC-157 for gut healing or experiencing GI effects from GLP-1 therapy, specialized gut health markers can provide valuable data:
Zonulin
Zonulin regulates intestinal permeability (the "tight junctions" between gut cells). Elevated zonulin indicates increased intestinal permeability ("leaky gut"). Oral BPC-157 protocols targeting gut healing should produce a declining zonulin level over 4-8 weeks.
Secretory IgA (sIgA)
sIgA is the primary immunoglobulin protecting mucosal surfaces, including the gut lining. Low sIgA indicates weakened mucosal immunity. BPC-157 and Thymosin Alpha-1 may support sIgA production.
Fecal Calprotectin
Calprotectin is released by neutrophils during intestinal inflammation. It's a validated marker for inflammatory bowel disease but is also useful for tracking subclinical gut inflammation. A declining calprotectin level on oral BPC-157 therapy provides objective evidence of reduced gut inflammation.
Short-Chain Fatty Acids (SCFAs)
Butyrate, propionate, and acetate are produced by beneficial gut bacteria and support gut barrier function, immune regulation, and metabolic health. Comprehensive stool testing can measure SCFA levels. GLP-1 therapy may affect gut transit time and microbiome composition, potentially altering SCFA production.
Building a Tiered Monitoring Approach
Not everyone needs advanced testing. Here's a practical tiered approach:
Tier 1: Essential (Everyone)
CMP, CBC, lipid panel, HbA1c, fasting insulin. Add IGF-1 for GH peptides. This covers safety and basic efficacy for less than $150.
Tier 2: Recommended (Most Peptide Users)
Everything in Tier 1 plus thyroid panel, hsCRP, vitamin D, and compound-specific markers (prolactin, lymphocyte subsets, etc.). Approximately $200-300.
Tier 3: Comprehensive (Optimization-Focused Users)
Everything in Tiers 1-2 plus ApoB, homocysteine, full hormone panel, GGT, uric acid, and iron studies. Approximately $350-500.
Tier 4: Advanced (Anti-Aging Protocol Users)
Everything in Tiers 1-3 plus oxidative stress markers, telomere length, gut health markers, and advanced inflammatory cytokines. Approximately $600-1,200+.
Start with Tier 1 for your first peptide protocol. As you gain experience and refine your goals, add Tier 2 and eventually Tier 3 markers. Tier 4 is for dedicated biohackers and patients on comprehensive anti-aging protocols where the investment in advanced testing is justified by the scope and cost of the overall program.
Cost-Effective Advanced Testing
Many advanced tests are only needed once or twice a year, not quarterly. Lp(a) only needs to be tested once. Telomere length is meaningful only annually. Hormone panels for stable patients can be done every 6-12 months. Build your advanced panel into annual or semi-annual comprehensive assessments rather than adding every test to every quarterly draw. This keeps the cost manageable while providing the depth of data you need for long-term optimization.
How Common Medications Affect Your Lab Results
Peptide therapy doesn't exist in a vacuum. Most patients are taking other medications or supplements that can affect lab results. Understanding these interactions prevents misinterpretation and unnecessary protocol changes.
Medications That Affect Glucose/Insulin Markers
| Medication | Effect on Glucose Labs | Implications for Peptide Monitoring |
|---|---|---|
| Metformin | Lowers fasting glucose, HbA1c, fasting insulin | May mask glucose-raising effects of GH peptides; can create a false sense of metabolic security on MK-677 |
| Corticosteroids (prednisone, etc.) | Raises fasting glucose, insulin; can push HbA1c into diabetic range | Confounds metabolic monitoring on any peptide; account for steroid effects when interpreting results |
| Beta-blockers | Can slightly raise fasting glucose and impair insulin sensitivity | May compound glucose-raising effects of GH-axis peptides |
| Thiazide diuretics | Can raise fasting glucose | Another potential confounder for glucose monitoring on GH peptides |
| Statins | May slightly increase HbA1c (0.1-0.3%) and fasting glucose | Account for this when interpreting metabolic improvement on GLP-1 therapy |
Medications That Affect Liver Markers
| Medication | Effect on Liver Labs | Implications |
|---|---|---|
| Statins | Can elevate ALT, AST (usually mild, dose-dependent) | Don't automatically attribute liver enzyme elevation to peptides if patient is on a statin |
| Acetaminophen (Tylenol) | Elevates ALT at therapeutic doses; toxic at high doses | Common OTC medication that can confound liver monitoring; ask about use |
| Antibiotics (many classes) | Can cause transient liver enzyme elevation | Note any recent antibiotic courses when reviewing liver labs |
| Antifungals (ketoconazole, fluconazole) | Can significantly elevate liver enzymes | Important confounder; azole antifungals are hepatotoxic |
| NSAIDs (ibuprofen, naproxen) | Rarely elevate liver enzymes; more commonly affect kidney markers | Can affect BUN, creatinine, and mask inflammation markers (CRP) |
Medications That Affect Thyroid Labs
| Medication | Effect on Thyroid Labs | Implications |
|---|---|---|
| Levothyroxine | Lowers TSH, raises free T4/T3 | Must be dosed appropriately; GH peptides may increase levothyroxine requirements |
| Biotin supplements | Can cause falsely low TSH and falsely high free T4 on immunoassay platforms | ALWAYS stop biotin 72 hours before thyroid testing; this is a common cause of "abnormal" thyroid results |
| Lithium | Can cause hypothyroidism (elevated TSH) | Important confounder if patient is on lithium for bipolar disorder |
| Amiodarone | Can cause both hypo- and hyperthyroidism | Complex thyroid effects; requires specialist monitoring |
Medications That Affect Hormone Labs
| Medication | Effect | Implications |
|---|---|---|
| Testosterone replacement therapy (TRT) | Elevates total/free testosterone, may raise estradiol, suppresses LH/FSH | If on TRT, LH/FSH will be suppressed; this is expected, not a peptide effect |
| Aromatase inhibitors (anastrozole) | Dramatically lower estradiol | Can cause estradiol to drop too low (joint pain, poor mood, bone risk) |
| Oral contraceptives (women) | Raise SHBG, lower free testosterone, alter estradiol/progesterone | Significantly affects hormone interpretation; note OC use when reviewing panels |
| SSRIs/SNRIs | Can elevate prolactin | Don't attribute prolactin elevation to peptides if patient is on an SSRI |
| Opioids | Suppress testosterone, LH, FSH | Major confounder for hormonal assessment; chronic opioid use causes hypogonadism |
Supplements That Affect Lab Results
- Fish oil (high dose): Can lower triglycerides and thin blood (affecting PT/INR). May make GLP-1 lipid improvements appear larger than they actually are.
- Berberine: Lowers fasting glucose and HbA1c similarly to metformin. Can mask glucose-raising effects of GH peptides.
- Iron supplements: Can elevate ferritin and liver enzymes temporarily. Take iron at a different time of day from other supplements and note supplementation when reviewing labs.
- Vitamin D (high dose): Can raise serum calcium. Important to monitor calcium alongside vitamin D on high-dose supplementation.
- NAC (N-Acetyl Cysteine): Supports liver health and may lower liver enzymes. Can make liver monitoring during peptide therapy appear more favorable than it would be without NAC support.
- Creatine: As discussed, raises serum creatinine and can give a falsely low eGFR. Does not indicate kidney dysfunction.
- Biotin: Interferes with many immunoassay tests. The number one supplement to stop before blood work. Affects thyroid, testosterone, cortisol, and potentially IGF-1 results depending on the assay platform.
Practical Recommendation
Create a comprehensive medication and supplement list before every lab draw. Share this list with whoever is interpreting your results. Many apparently abnormal findings are easily explained by concurrent medications or supplements. A provider who doesn't know you're taking creatine might order an unnecessary kidney workup based on elevated creatinine. A provider who doesn't know you're on berberine might conclude your GH peptide isn't affecting glucose when it actually is, but the berberine is counteracting it. Transparency about your full protocol leads to accurate interpretation.
Step-by-Step: Building Your Personal Monitoring Protocol
Now that you understand the individual biomarkers, testing schedules, and interpretation principles, here's how to put it all together into a personalized monitoring plan that you can execute and maintain over the long term.
Step 1: Define Your Peptide Protocol
Write down every peptide you're using or planning to use, including compound name, dose, frequency, and route of administration. If you're stacking, note the start date for each compound. This document becomes the foundation of your monitoring plan.
Step 2: Identify Required Tests by Compound
Using the tables earlier in this guide, list out the specific tests required for each compound in your protocol. Then combine them into a single panel, removing duplicates. This is your monitoring panel.
Step 3: Establish Your Budget
Determine how much you're willing to spend on monitoring per quarter. If budget is limited, prioritize Tier 1 tests (CMP, CBC, lipid panel, HbA1c, fasting insulin) and add compound-specific tests (IGF-1 for GH peptides, etc.) as the most important add-ons. A minimum viable monitoring budget is approximately $150-200 per quarter for most peptide protocols.
Step 4: Choose Your Lab Provider
Select a DTC lab company and stick with it for consistency. Consider proximity to draw sites, pricing, test menu breadth, and result turnaround time. Create an account and familiarize yourself with the ordering process before your first draw.
Step 5: Schedule Your Draws
Plot your testing schedule on a calendar. For most protocols, this means:
- Week 0: Baseline (before first dose)
- Weeks 4-8: First follow-up (compound-specific)
- Week 12: Comprehensive follow-up
- Every 3 months thereafter for the first year
- Every 6 months after year one if stable
Set calendar reminders 1-2 weeks before each draw to order your tests and prepare (fasting instructions, supplement holds, exercise avoidance).
Step 6: Create Your Tracking Document
Build a spreadsheet or use a health tracking app to log all results. Include columns for date, lab provider, fasting status, current protocol, each biomarker value, reference range, your optimal range, and notes. Update this within 24 hours of receiving each set of results.
Step 7: Review and Act
After each lab draw, review your results using the interpretation framework in this guide:
- Are all values within your optimal range?
- Have any values changed significantly from your previous draw?
- Are there any trends developing (up or down) over multiple draws?
- Do the efficacy markers show the peptide is working?
- Do any red flags require immediate action?
Schedule a follow-up with your provider if any results are concerning or if you want guidance on protocol adjustments. Bring your tracking document to every provider appointment.
Step 8: Adjust and Iterate
Based on your results and provider guidance, make protocol adjustments (dose changes, compound additions or removals, timing modifications) and continue monitoring. Peptide therapy is an iterative process. The data from each lab draw informs the next decision, and over time, you develop a highly personalized, data-driven protocol that produces optimal results with minimal risk.
This iterative, data-driven approach is what separates responsible peptide therapy from guesswork. It takes effort, costs money, and requires discipline. But the payoff, in safety, efficacy, and confidence in your protocol, is well worth it.
Ready to get started? Visit FormBlends.com/start to explore peptide options and begin building your protocol. Our dosing calculator can help you determine appropriate starting doses, and our research library provides detailed compound guides for every peptide mentioned in this article.
Complete Lab Profiles for Every Major Peptide Category
This section provides ready-to-use lab ordering guides for each major peptide compound. Think of these as your cheat sheets - print them out or save them on your phone for quick reference when ordering blood work.
CJC-1295 / Ipamorelin Lab Profile
CJC-1295/Ipamorelin is the most commonly prescribed GH secretagogue combination. CJC-1295 (with or without DAC) provides sustained GHRH-like stimulation, while Ipamorelin acts as a selective ghrelin receptor agonist. Together, they produce amplified, pulsatile GH release that closely mimics natural physiology.
The monitoring requirements reflect both the efficacy targets (IGF-1 response) and safety concerns (glucose metabolism, thyroid function). Because Ipamorelin is one of the "cleaner" GH secretagogues with minimal prolactin and cortisol stimulation, the hormone monitoring requirements are lighter than for GHRP-6 or MK-677.
| Test | Baseline | 6 Weeks | 3 Months | 6 Months | Annual |
|---|---|---|---|---|---|
| IGF-1 | Yes | Yes | Yes | Yes | Yes |
| CMP (includes glucose, liver, kidney) | Yes | Yes | Yes | Yes | Yes |
| Fasting Insulin | Yes | Yes | Yes | Yes | Yes |
| HbA1c | Yes | No | Yes | Yes | Yes |
| CBC | Yes | No | Yes | No | Yes |
| Thyroid (TSH, fT4) | Yes | No | If symptomatic | Yes | Yes |
| Lipid Panel | Yes | No | Yes | No | Yes |
| PSA (men >40) | Yes | No | No | No | Yes |
Target IGF-1: 200-280 ng/mL (age-adjusted upper-middle range)
Key safety thresholds: Fasting glucose >110 mg/dL (flag), >126 (stop and evaluate). IGF-1 >350 (reduce dose).
Estimated quarterly DTC lab cost: $130-180
Sermorelin Lab Profile
Sermorelin is a truncated GHRH analog (the first 29 amino acids of the 44 amino acid GHRH molecule). It stimulates GH release through the GHRH receptor exclusively, making it one of the most physiological GH secretagogues available. It doesn't stimulate prolactin or cortisol and has minimal glucose impact compared to MK-677 or GHRP-6.
Monitoring follows the standard GH peptide profile. Due to its shorter half-life compared to CJC-1295 with DAC, the IGF-1 response may be more modest and slower to develop. Some patients need 8-12 weeks to see meaningful IGF-1 elevation on sermorelin.
Target IGF-1: 180-260 ng/mL (slightly lower targets are appropriate given the more physiological GH stimulation pattern)
Key consideration: If IGF-1 hasn't risen meaningfully after 12 weeks, consider switching to the CJC-1295/Ipamorelin combination for more potent stimulation.
Tesamorelin Lab Profile
Tesamorelin is an FDA-approved GHRH analog specifically indicated for HIV-associated lipodystrophy. It's also used off-label for visceral fat reduction and GH optimization. It produces strong, sustained IGF-1 elevation and has documented effects on liver fat reduction.
Monitoring is similar to other GH peptides, with additional attention to liver imaging if the primary goal is visceral fat/NAFLD reduction. Tesamorelin's clinical trial data provides some of the best reference points for expected lab changes on GH secretagogue therapy.
| Expected Lab Changes on Tesamorelin (from clinical trials) | Typical Magnitude |
|---|---|
| IGF-1 increase | 80-120% from baseline |
| Trunk fat reduction (by DEXA) | 10-18% decrease |
| Liver fat reduction (by imaging) | 30-40% decrease |
| Triglyceride change | Modest decrease (10-15%) |
| Fasting glucose change | Small increase (3-8 mg/dL) or stable |
| HbA1c change | Usually stable or minimal increase |
MK-677 (Ibutamoren) Lab Profile
MK-677 requires the most intensive metabolic monitoring of any GH-axis compound. As an oral ghrelin mimetic with a 24-hour duration of action, it produces sustained GH and IGF-1 elevation along with significant appetite stimulation, water retention, and glucose dysregulation risk.
| Test | Baseline | 4 Weeks | 8 Weeks | 3 Months | 6 Months |
|---|---|---|---|---|---|
| IGF-1 | Yes | Yes | Yes | Yes | Yes |
| Fasting Glucose | Yes | Yes | Yes | Yes | Yes |
| Fasting Insulin | Yes | Yes | Yes | Yes | Yes |
| HbA1c | Yes | No | Yes | Yes | Yes |
| CMP | Yes | Yes | Yes | Yes | Yes |
| Prolactin | Yes | No | Yes | If elevated | If elevated |
| Cortisol (AM) | Yes | No | Optional | If symptomatic | No |
| CBC | Yes | No | No | Yes | Yes |
| Thyroid | Yes | No | No | If symptomatic | Yes |
Target IGF-1: 220-300 ng/mL
Key safety thresholds: Fasting glucose >110 mg/dL (caution), >126 (consider discontinuation). HOMA-IR >3.0 (requires intervention). HbA1c >5.7% (serious concern in non-diabetics). Prolactin >25 ng/mL in men (consider switching compounds).
Absolute contraindications: Pre-existing type 2 diabetes or HbA1c >6.5%. Active malignancy. History of significant glucose intolerance.
Estimated quarterly DTC lab cost: $160-220
Semaglutide Lab Profile
Semaglutide monitoring focuses on tracking metabolic improvement and catching the rare adverse effects associated with GLP-1 receptor agonism. Most lab changes are positive, making this one of the more rewarding peptide categories to monitor.
| Test | Baseline | 4-6 Weeks | 3 Months | 6 Months | Annual |
|---|---|---|---|---|---|
| HbA1c | Yes | No | Yes | Yes | Yes |
| Fasting Glucose | Yes | Yes | Yes | Yes | Yes |
| Fasting Insulin | Yes | No | Yes | Yes | Yes |
| CMP | Yes | Yes | Yes | Yes | Yes |
| Lipid Panel | Yes | No | Yes | Yes | Yes |
| CBC | Yes | No | No | Yes | Yes |
| Thyroid (TSH) | Yes | No | No | No | Yes |
| Lipase | Yes | If abdominal pain | No | No | No |
| B12, Iron, Vitamin D | Yes | No | No | Yes | Yes |
Key efficacy markers: Declining HbA1c, fasting glucose, fasting insulin, triglycerides, ALT. Improving lipid ratios.
Key safety thresholds: ALT >5x ULN (stop immediately). eGFR declining >25% from baseline (evaluate hydration and kidney function). Potassium <3.5 mEq/L (supplement and monitor). Lipase >3x ULN with pain (evaluate for pancreatitis).
Estimated quarterly DTC lab cost: $100-150
Tirzepatide Lab Profile
Tirzepatide is a dual GIP/GLP-1 receptor agonist that generally produces larger metabolic improvements than semaglutide alone. The monitoring profile is similar to semaglutide with slightly more attention to liver markers given the rare reports of hepatotoxicity.
Additional monitoring consideration: Tirzepatide produces more potent weight loss than semaglutide in head-to-head studies. This means greater vigilance is needed for nutritional deficiencies, muscle loss markers (track albumin, prealbumin, and consider DEXA scans), and electrolyte disturbances from more aggressive appetite suppression.
BPC-157 Lab Profile
BPC-157 has the lightest monitoring requirements of any peptide in common use, reflecting both its favorable safety profile in available data and its mechanism of action (local tissue repair rather than systemic hormonal effects).
| Test | Baseline | End of Course (4-8 weeks) | Ongoing (if >8 weeks) |
|---|---|---|---|
| CMP | Yes | Yes | Every 8 weeks |
| CBC | Optional | Optional | Optional |
| hsCRP | Yes | Yes | Every 8 weeks |
| ESR | Optional | Optional | Optional |
Key efficacy marker: Declining hsCRP and ESR (for inflammation-related indications). Functional improvement (range of motion, pain scores) may be more meaningful than lab markers.
Estimated DTC lab cost per assessment: $40-70
Thymosin Alpha-1 Lab Profile
Thymosin Alpha-1 requires immune-specific monitoring that goes beyond standard metabolic panels. The lymphocyte subset panel is the key efficacy marker, documenting the immune activation that Ta1 is intended to produce.
Target responses: CD4 count increase of 20-40% from baseline. CD4/CD8 ratio normalization toward 1.5-2.5. NK cell count increase. hsCRP decline.
Safety concerns: Immune overstimulation (WBC >12,000, new autoimmune symptoms). Monitor autoantibodies in patients with autoimmune conditions.
Estimated quarterly DTC lab cost: $180-280 (lymphocyte subsets are more expensive than standard tests)
NAD+ Therapy Lab Profile
NAD+ therapy (IV, SubQ, or oral NMN/NR) requires attention to methylation markers and metabolic parameters. The unique consideration is homocysteine monitoring, as NAD+ metabolism increases methylation demand and can deplete methyl donors (B12, folate, SAMe) if supplementation is inadequate.
| Test | Baseline | 8-12 Weeks | 6 Months | Annual |
|---|---|---|---|---|
| CMP | Yes | Yes | Yes | Yes |
| Homocysteine | Yes | Yes | Yes | Yes |
| Uric Acid | Yes | Yes | Yes | Yes |
| hsCRP | Yes | No | Yes | Yes |
| Fasting Glucose/Insulin | Yes | No | Yes | Yes |
| CBC | Yes | No | No | Yes |
| B12, Folate | Yes | Yes | Yes | Yes |
Key safety thresholds: Homocysteine >12 umol/L (increase B12/folate supplementation). Uric acid >8 mg/dL (risk of gout; may need dose reduction). hsCRP rising (evaluate for unexpected inflammatory process).
Estimated quarterly DTC lab cost: $120-180
Epithalon Lab Profile
Epithalon is typically used in 10-20 day cycles with months between courses. Monitoring is focused on anti-aging biomarkers and safety during each cycle.
Per-cycle monitoring: CMP and CBC before each cycle (safety baseline). hsCRP before and 4 weeks after cycle (track anti-inflammatory response). Annual comprehensive panel including vitamin D, homocysteine, hormone panel, and advanced lipids.
Optional advanced markers: Telomere length testing (annually). 8-OHdG (oxidative DNA damage). Epigenetic clock testing (experimental but emerging).
Estimated cost per cycle monitoring: $60-100 (basic). $300-600 (with advanced markers).
Quick Reference Summary: Your Blood Work Monitoring Cheat Sheet
For quick reference, here are the essential monitoring guidelines distilled into action-oriented summaries you can use at a glance.
The Universal Rules
- Always get baseline labs before starting any peptide. No exceptions. You can't track changes without knowing where you started.
- Fast for 8-12 hours before blood draws. Water only. No coffee, no supplements. Early morning draws (7-10 AM) are optimal.
- Stop biotin 72 hours before testing. It interferes with many immunoassay tests.
- Avoid intense exercise for 48 hours before draws. Exercise can elevate liver enzymes and skew other markers.
- Use the same lab for serial testing. Different labs use different assays that may not be directly comparable.
- Track trends, not single values. A spreadsheet or tracking app is your most valuable monitoring tool.
- Know your red flags. Fasting glucose >200, ALT >5x ULN, potassium <3.0 or >5.5, eGFR dropping rapidly. These require same-day medical attention.
- Communicate fully with your provider. Share all peptides, medications, and supplements. Complete information leads to accurate interpretation.
The Minimum Viable Panel for Any Peptide
If budget or access is limited and you can only order a few tests, these are the non-negotiable minimums:
- CMP (Comprehensive Metabolic Panel): Covers glucose, liver, kidney, and electrolytes in a single test. Cost: $15-35.
- CBC (Complete Blood Count): General health screening. Cost: $15-30.
- hsCRP: Inflammation marker. Cost: $15-30.
Total minimum viable panel cost: $45-95 through DTC labs. This won't give you efficacy data for specific peptides, but it covers basic safety. Add compound-specific markers (IGF-1 for GH peptides, HbA1c for GLP-1s, etc.) as your budget allows.
Blood work isn't glamorous, but it's the difference between hope and evidence. It transforms peptide therapy from an experiment into a measurable, adjustable, optimizable protocol. Take the time to get tested. Track your numbers. Learn what they mean. Your future self will thank you.
Troubleshooting: Common Problems and Solutions in Peptide Lab Monitoring
Even with the best intentions, things go wrong with lab testing. Samples get mishandled, results don't make sense, and confounding variables create confusion. Here's how to troubleshoot the most common problems you'll encounter.
Problem: IGF-1 Didn't Increase After 8 Weeks on GH Peptides
This is one of the most frustrating findings for GH peptide users. You've been injecting nightly for two months and your IGF-1 hasn't budged from baseline. Before assuming the peptide doesn't work, consider these possibilities:
- Product quality: Unfortunately, the compounded peptide market has quality control issues. Some products contain less active ingredient than labeled, are degraded from improper storage, or in rare cases contain no active compound at all. If multiple peptide markers (IGF-1, sleep quality, body composition) show no response, product quality is a legitimate concern. Consider switching to a different, reputable source.
- Caloric restriction: If you're simultaneously restricting calories significantly (especially on concurrent GLP-1 therapy), the liver may not produce IGF-1 efficiently despite adequate GH stimulation. IGF-1 production requires adequate caloric and protein intake. Ensure you're consuming at least 0.7g of protein per pound of body weight and not in an extreme caloric deficit.
- Injection technique: Subcutaneous injection technique matters. If the peptide is being injected intramuscularly by mistake, or if the injection site is highly fibrosed from repeated use in the same location, absorption may be impaired. Rotate injection sites regularly and ensure proper SubQ depth.
- Storage degradation: Reconstituted peptides are fragile. If they've been stored at room temperature, exposed to direct light, or shaken vigorously, the active compound may have degraded. Reconstituted peptides should be refrigerated at 2-8 degrees Celsius, protected from light, and handled gently.
- Dosing too low: Some individuals require higher doses to achieve meaningful IGF-1 elevation. If the product is legitimate and other confounders are controlled, discuss a dose increase with your provider.
- Insulin resistance: High insulin levels can impair hepatic GH receptor signaling, blunting IGF-1 production. If you have significant insulin resistance (HOMA-IR above 3), addressing insulin sensitivity may improve IGF-1 response to peptides.
- Lab assay variability: As discussed, different IGF-1 assays can produce different results. If you switched labs between baseline and follow-up, the values may not be directly comparable.
Problem: Liver Enzymes Elevated But You Feel Fine
Mildly elevated liver enzymes (ALT 60-100 U/L) with no symptoms is a common finding that causes anxiety but often has benign explanations. Before attributing it to your peptide protocol:
- Check your exercise history: Did you do intense resistance training or a long endurance workout within 48-72 hours of the draw? This is the most common cause of "unexplained" liver enzyme elevation in active peptide users. Order a CK (creatine kinase) to confirm muscle versus liver origin. CK above 300-500 U/L after exercise confirms muscular contribution to AST elevation.
- Review supplements: High-dose vitamin A, green tea extract, kava, and certain herbal supplements are hepatotoxic. NAC (N-acetyl cysteine) can paradoxically lower liver enzymes, masking underlying issues while you're taking it and revealing them if you stop.
- Consider alcohol: Even social drinking (2-3 drinks) within 48 hours can measurably elevate liver enzymes in some individuals.
- Think about body composition: If you're overweight and haven't been tested before, the elevation might represent pre-existing NAFLD/NASH that was already present before peptide therapy began. This is where baseline labs would have been invaluable.
- Retest after controlling variables: Avoid exercise for 72 hours, avoid alcohol for 5 days, stop potentially hepatotoxic supplements for 2 weeks, and retest. If values normalize, the cause was likely non-hepatic.
Problem: Lab Results Don't Match How You Feel
Sometimes patients feel great but have concerning lab results, or feel terrible with perfectly normal labs. This disconnect is more common than you might think, and there are several explanations:
Feeling Great With Bad Labs
Possible explanations include: the subjective benefits of the peptide (better sleep, mood, energy from GH secretagogues) are masking the metabolic effects (rising glucose and insulin). The benefits are real and the metabolic effects are also real; they're not mutually exclusive. This is why blood work is indispensable. You cannot feel insulin resistance developing. You cannot feel your ALT rising. By the time you feel these things, significant damage may have occurred.
Feeling Terrible With Good Labs
Possible explanations: the dose isn't high enough to produce measurable changes yet but is high enough to cause side effects. Or the timing is wrong - GH peptide side effects (water retention, joint stiffness, fatigue) can precede the positive IGF-1 response. Or there are non-peptide factors (stress, sleep deprivation, dietary issues) causing symptoms that aren't captured by standard lab panels. Consider whether your symptoms are truly related to the peptide protocol or whether other life factors are responsible.
Problem: Can't Get the Tests You Need
Several practical obstacles can prevent patients from getting adequate monitoring:
State Restrictions on DTC Testing
If you're in New York, New Jersey, Rhode Island, or Maryland where DTC lab testing is restricted, work with a telehealth provider who can order labs for you. Many telemedicine peptide clinics include lab ordering as part of their services. Some DTC lab companies have found workarounds in restricted states by partnering with licensed physicians who review and approve orders.
Insurance Won't Cover Tests
If you're going through insurance and the tests are being denied, have your provider use appropriate diagnostic codes that justify the testing. For example, "metabolic syndrome" (ICD-10: E88.81) can justify CMP, lipid panel, fasting insulin, and HbA1c. "Concern for growth hormone deficiency" can justify IGF-1 testing. Many providers have experience working with insurance systems to get tests covered. Alternatively, bypass insurance entirely and use DTC labs where pricing is often lower than insured copays anyway.
Can't Find a Provider Who Understands Peptide Monitoring
If your local providers aren't familiar with peptide therapy, consider telemedicine options. Many clinics now operate entirely remotely, providing consultations, prescriptions, lab orders, and lab reviews through video visits. This has dramatically expanded access to knowledgeable providers regardless of geographic location. Our getting started page can help connect you with appropriate clinical resources.
Problem: Results Vary Significantly Between Draws
You get blood work every 3 months and your IGF-1 goes from 245 to 198 to 272 to 215. Is the peptide not working consistently? Is something wrong? Probably not. Biological variability is normal. Even with perfect control of pre-analytical variables, your body's biomarker levels fluctuate from day to day and week to week.
Studies on IGF-1 variability show that intra-individual coefficient of variation (how much one person's results vary over repeated testing) can be 10-20%. That means an "actual" IGF-1 of 230 might test anywhere from 185 to 275 on any given draw. This is normal analytical and biological variability, not a problem with your protocol.
The solution is to focus on rolling averages and long-term trends rather than individual data points. If your last four IGF-1 results are 245, 198, 272, and 215, your average is 232.5 - solidly in the optimal range. The variability between draws is noise; the average is the signal.
For markers with less analytical variability (HbA1c, fasting glucose), changes of 0.1-0.2% or 3-5 mg/dL between draws are within normal variability and shouldn't prompt protocol changes. Meaningful trends typically require at least three consecutive measurements moving in the same direction.
Problem: You Missed Your Baseline Labs
If you've already started peptide therapy without getting baseline labs (a common scenario), it's not too late to start monitoring. Get comprehensive labs now. While you won't have your own baseline for comparison, you can compare against standard reference ranges and begin tracking trends from this point forward.
For some markers, you can estimate what your baseline likely was. If your IGF-1 is now 265 and you're 42 years old on CJC-1295/Ipamorelin, your pre-therapy IGF-1 was probably somewhere between 120-200 based on population averages for your age. This isn't as good as having your own measured baseline, but it gives you an approximate frame of reference.
The key lesson: establish your tracking system now and maintain it going forward. Having 80% of the data is infinitely better than having 0%. Don't let the absence of perfect baseline data discourage you from monitoring going forward.
The Return on Investment: Why Blood Work Pays for Itself
Some people see lab testing as an unnecessary expense on top of already-expensive peptide therapy. Let's reframe the economics.
The Math of Monitoring
A typical GH peptide protocol (CJC-1295/Ipamorelin) costs approximately $200-400 per month. Over a year, that's $2,400-4,800. Quarterly blood work monitoring through DTC labs costs approximately $500-800 per year. That's roughly 10-20% of your peptide investment, and it serves three economic functions:
Function 1: Preventing Waste
Without monitoring, you have no way to know if your peptide is working. You could spend $300/month on a product that's degraded, underdosed, or simply not producing the intended biological response. An IGF-1 test costing $65 confirms whether your $300/month investment is actually doing something. If it's not, you save the cost of continued useless therapy and redirect to a product that works. A single IGF-1 test that identifies a non-functional product saves you hundreds of dollars per month.
Function 2: Preventing Medical Costs
Catching insulin resistance early on MK-677 and intervening (dose reduction, dietary changes, addition of metformin) costs virtually nothing beyond the lab test that detected it. Letting insulin resistance progress to type 2 diabetes costs, on average, $10,000-16,000 per year in medical expenses. Catching elevated liver enzymes early and stopping the causative compound prevents the cost of advanced liver workups ($1,000-5,000+), specialist consultations, and potential hospitalization for severe drug-induced liver injury.
Function 3: Optimizing Results
Blood work allows dose optimization that maximizes your results per dollar spent. Without IGF-1 monitoring, you might stay on a dose that's too low (underspending for inadequate results) or too high (overspending and increasing side effects). Dialing in the optimal dose based on lab data ensures every dollar spent on peptides is producing measurable, documented benefit.
The Psychological Value
Beyond economics, regular blood work provides something invaluable: confidence. When your labs confirm that your peptide protocol is working safely and effectively, you have peace of mind that supports adherence and reduces the anxiety that comes with injecting compounds with limited long-term human safety data. Conversely, if something goes wrong, early detection through blood work converts a potentially serious problem into a manageable adjustment.
The bottom line: blood work monitoring isn't an expense to be minimized. It's an integral part of safe, effective peptide therapy that pays for itself many times over through waste prevention, complication avoidance, and protocol optimization. Any comprehensive peptide protocol that doesn't include regular monitoring is incomplete.
Seasonal and Lifestyle Factors That Affect Your Lab Results
Your blood work isn't just influenced by the peptides you take. Season, stress, sleep, exercise patterns, and dietary changes all create natural fluctuations that you need to account for when interpreting results.
Seasonal Variations
Several biomarkers show predictable seasonal patterns that have nothing to do with your peptide protocol:
Vitamin D
Vitamin D levels peak in late summer/early fall and trough in late winter/early spring in most temperate climates. A vitamin D reading of 42 ng/mL in August and 28 ng/mL in February doesn't indicate a problem with your protocol. It reflects normal seasonal variation in sun exposure. If you're tracking vitamin D as part of a longevity panel, compare results from the same season each year, or maintain consistent supplementation year-round to stabilize levels.
Testosterone
Testosterone follows a subtle seasonal pattern, with levels typically peaking in late fall/early winter and dipping in spring/summer. The magnitude of seasonal variation is usually 10-15%, which can move borderline values across reference range thresholds. If your total testosterone is 520 ng/dL in November and 445 ng/dL in June, the difference is likely seasonal rather than a sign that your peptide protocol stopped supporting testosterone production.
Cholesterol and Lipids
Total cholesterol and LDL tend to be 3-5% higher in winter months, potentially due to dietary changes, reduced physical activity, and altered melatonin-mediated lipid metabolism. Don't make medication changes based on a single winter lipid panel. Recheck in warmer months for a more representative reading.
HbA1c
HbA1c can show seasonal variation of 0.1-0.3% in some populations, with higher values in colder months. This is partly driven by dietary patterns (holiday eating) and reduced physical activity. Account for this when tracking HbA1c trends on GLP-1 therapy. A winter reading that's 0.1% higher than your fall reading may simply reflect seasonal effects rather than worsening glucose control.
Sleep Quality Effects
Sleep profoundly affects multiple biomarkers. Poor sleep the night before a blood draw can acutely alter:
- Cortisol: Elevated after sleep deprivation, sometimes by 30-50%
- Fasting glucose: Acutely elevated by 5-15 mg/dL after a bad night of sleep
- Insulin sensitivity: Reduced after even one night of poor sleep, raising fasting insulin
- CRP: Can be elevated by chronic sleep deprivation
- Testosterone: Reduced by 10-15% after a poor night of sleep
- TSH: Can be affected by sleep timing and circadian disruption
- GH/IGF-1: GH is primarily released during deep sleep. Chronic sleep deprivation blunts GH release and may reduce IGF-1 over time, potentially counteracting the effects of GH peptide therapy
For the most accurate results, prioritize good sleep for the 2-3 nights before a blood draw. If you had a particularly poor night, consider rescheduling your draw rather than getting potentially misleading results.
Stress and Psychological Factors
Acute psychological stress on the morning of your blood draw (traffic, running late, anxiety about the needle) can elevate cortisol, glucose, and blood pressure. Chronic stress has broader effects on inflammatory markers, hormones, and metabolic function.
Practical tip: arrive at the draw site with time to spare. Sit quietly for 5-10 minutes before the draw. Practice slow breathing if you're anxious about needles. These simple measures can reduce stress-related interference with your results.
Dietary Patterns
Beyond the fasting requirement, what you eat in the days before a blood draw can affect results:
- Very high-fat meal the evening before: Can elevate triglycerides even after an overnight fast, as some postprandial lipemia persists beyond 12 hours in susceptible individuals
- High-sodium meal: Can affect sodium levels and blood pressure readings
- Extremely low-carb diet (keto): Produces physiological insulin resistance (adaptive response to carb restriction) that can elevate fasting glucose and worsen HOMA-IR. This doesn't indicate pathological insulin resistance. If you're on a ketogenic diet, note this when interpreting glucose and insulin values.
- Alcohol: Elevates triglycerides, liver enzymes, and uric acid. Suppress these effects by avoiding alcohol for 72 hours before testing.
- Caffeine: Can acutely elevate cortisol and slightly raise glucose. While black coffee doesn't technically break a fast for most markers, avoiding it before blood work gives the cleanest results.
Travel and Time Zone Changes
If you've recently traveled across time zones, your circadian rhythm is disrupted. This affects cortisol patterns, testosterone rhythms, TSH fluctuations, and glucose regulation. If possible, wait 3-5 days after crossing two or more time zones before getting blood work, to allow your circadian system to re-synchronize.
Illness and Recovery
Even a mild cold or upper respiratory infection can significantly affect lab results for 2-4 weeks after recovery. CRP and ESR spike during acute illness and may take weeks to return to baseline. WBC counts fluctuate. Liver enzymes can be transiently elevated by viral infections. If you've been sick within the past 2-3 weeks, consider postponing non-urgent lab testing or noting the illness when interpreting results.
The overarching principle: standardize your pre-draw conditions as much as possible. Same time of day, same fasting duration, similar sleep quality, similar exercise pattern, same lab company. The more consistent your testing conditions, the more meaningful your trends. Variables are the enemy of accurate interpretation, and most variables are controllable with a little planning.
Anti-Aging and Longevity Peptide Monitoring
Anti-aging peptides like Epithalon, NAD+, MOTS-c, and GHK-Cu target fundamental aging mechanisms including telomere maintenance, mitochondrial function, and cellular senescence. Monitoring these protocols requires a slightly different approach than acute therapeutic peptides.
Epithalon Monitoring
Epithalon works primarily through telomerase activation and pineal gland modulation. Monitoring focuses on:
- Melatonin levels: Epithalon may restore more youthful melatonin production. Salivary or serum melatonin can be measured, though this test is not widely available through standard labs.
- hsCRP: Anti-inflammatory effects should produce a declining trend over 3-6 months
- CMP and CBC: Standard safety monitoring at baseline and after each treatment cycle
- Telomere length testing: Available through specialized labs; meaningful changes require 6-12+ months to manifest. Expensive ($200-500) and not necessary for routine monitoring, but provides the most direct measure of Epithalon's purported mechanism.
NAD+ Therapy Monitoring
NAD+ therapy (IV, SubQ, or oral precursors like NMN/NR) requires attention to methylation demand and metabolic status:
- Homocysteine: NAD+ metabolism increases methylation demand. Rising homocysteine suggests inadequate B-vitamin support (B12, folate, B6). Check at baseline and 8-12 weeks.
- CMP: Standard organ function monitoring
- Uric acid: NAD+ metabolism can increase uric acid levels. Baseline and periodic monitoring recommended, especially in patients with gout history.
- hsCRP: Track anti-inflammatory effects
- Fasting glucose and insulin: NAD+ precursors have shown improvements in insulin sensitivity in clinical trials; track to document benefit
MOTS-c and Mitochondrial Peptides
MOTS-c, a mitochondrial-derived peptide, targets metabolic function and exercise capacity. Relevant monitoring includes:
- Fasting glucose and HbA1c (MOTS-c improves glucose metabolism)
- Lactate levels (may improve exercise-related lactate clearance)
- hsCRP (anti-inflammatory effects)
- CMP for safety monitoring
General Longevity Panel
For patients on comprehensive anti-aging peptide protocols, consider a broader longevity-focused panel that captures the multisystem effects of these compounds:
| Marker | Purpose | Frequency |
|---|---|---|
| hsCRP | Systemic inflammation | Every 3-6 months |
| Homocysteine | Methylation status | Every 6 months |
| Fasting insulin + glucose + HbA1c | Metabolic health | Every 3-6 months |
| Lipid panel (advanced with ApoB, Lp(a)) | Cardiovascular risk | Every 6-12 months |
| Vitamin D (25-OH) | Immune and bone health | Every 6 months |
| CBC with differential | Immune cell populations | Every 6 months |
| CMP | Organ function | Every 6 months |
| GGT (Gamma-Glutamyl Transferase) | Liver and oxidative stress | Every 6-12 months |
| Uric acid | Metabolic and inflammatory marker | Every 6-12 months |
| DHEA-S | Adrenal and hormonal aging | Annually |
Monitoring Peptide Stacks and Combined Protocols
Many peptide users don't take a single compound in isolation. Stacking multiple peptides creates compound interactions that require thoughtful monitoring strategies. For a detailed overview of common stacking protocols, see our peptide stacking and combinations guide.
Common Stacking Scenarios and Their Monitoring Requirements
GH Peptide + GLP-1 Agonist Stack
This is one of the most popular combinations, using CJC-1295/Ipamorelin for body composition alongside semaglutide or tirzepatide for weight loss. The monitoring challenge is that GH peptides can raise glucose while GLP-1s lower it. You need to track both effects simultaneously.
Required monitoring: IGF-1, fasting glucose, fasting insulin, HbA1c, CMP (liver/kidney), lipid panel. Testing every 6-8 weeks during the first 3 months, then quarterly.
GH Peptide + Healing Peptide Stack
Combining CJC-1295/Ipamorelin with BPC-157 for injury recovery plus systemic benefits. Monitoring follows the GH peptide schedule, with CRP added to track healing peptide efficacy.
Multiple GH-Axis Compounds
Stacking MK-677 with injectable GH secretagogues creates additive IGF-1 elevation and compounded glucose effects. This combination requires the most vigilant monitoring:
- IGF-1 every 4-6 weeks until stable
- Fasting glucose and insulin every 4 weeks for the first 3 months
- HbA1c monthly for the first 3 months (this is unusually frequent, but justified by the compounded glucose risk)
- Prolactin at baseline and 6 weeks
Immune + Healing Peptide Stack
Thymosin Alpha-1 with BPC-157 for combined immune modulation and tissue repair. Monitor immune panels (lymphocyte subsets, hsCRP) alongside standard CMP safety labs.
The Sequencing Principle
When starting multiple peptides, begin them one at a time with at least 2-4 weeks between introductions. This allows you to attribute any lab changes to the specific compound that caused them. If you start three peptides on the same day and your labs change, you won't know which one is responsible. Sequential introduction combined with lab testing at each introduction creates a clear cause-and-effect record [22].
Stacking Monitoring Rule
When monitoring a peptide stack, test for the union of all individual compound requirements. If Compound A requires IGF-1 and glucose monitoring, and Compound B requires liver enzymes and inflammatory markers, your stack monitoring panel includes all four. The testing frequency should match the most demanding compound in your stack.
Pre-Analytical Variables: How to Get Accurate Results
Even the most sophisticated lab test is only as good as the conditions under which the blood was drawn. Pre-analytical variables, meaning everything that happens before the lab analyzes your sample, can significantly affect results and lead to false conclusions.
Fasting Requirements
| Test | Fasting Required? | Fasting Duration | Notes |
|---|---|---|---|
| Glucose | Yes | 8-12 hours | Water only. Even black coffee can slightly raise cortisol and glucose. |
| Insulin | Yes | 8-12 hours | Any caloric intake invalidates the result |
| Lipid Panel | Preferred | 9-12 hours | Non-fasting is acceptable for screening but fasting gives more accurate LDL and triglycerides |
| IGF-1 | Preferred | 8 hours minimum | Food intake can modestly affect levels |
| CMP | Yes (for glucose component) | 8-12 hours | BUN can be affected by protein intake |
| Thyroid Panel | Preferred | Not strict | TSH is highest in early morning fasting |
| Testosterone | Preferred | Not strict | Morning draw (7-10 AM) is more important than fasting |
| hsCRP | No | N/A | Not significantly affected by food |
| CBC | No | N/A | Hydration status can affect concentration |
Exercise and Physical Activity
Intense exercise within 24-48 hours of a blood draw can affect multiple markers:
- AST and ALT: Can rise 2-3x from muscle breakdown, not liver injury. AST is found in muscle tissue and skeletal muscle, so heavy lifting or endurance exercise will elevate it. This is the most common cause of "abnormal liver enzymes" in otherwise healthy, active peptide users.
- CK (Creatine Kinase): Dramatic elevations from muscle damage. If your AST is elevated, ordering CK can help distinguish muscle versus liver origin.
- Cortisol: Acutely elevated after intense exercise
- Glucose: Can be transiently lower or higher depending on exercise type and timing
- WBC: Transient leukocytosis (elevated WBC) after hard training is normal
Recommendation: avoid intense exercise for 48 hours before lab draws. Light walking or gentle activity is fine.
Supplements and Medications
- Biotin: Can interfere with many immunoassay-based tests, including thyroid, testosterone, and IGF-1. Stop biotin supplements at least 72 hours before testing [23].
- NSAIDs (ibuprofen, naproxen): Can affect kidney function markers (creatinine, BUN) and suppress inflammatory markers (CRP)
- Alcohol: Elevates liver enzymes, triglycerides, and uric acid. Avoid for 48-72 hours before testing.
- Creatine: Can raise serum creatinine, potentially giving a falsely low eGFR. Inform your provider if you take creatine.
Hydration Status
Dehydration concentrates the blood and can artifactually elevate BUN, creatinine, hematocrit, hemoglobin, albumin, and electrolytes. Over-hydration dilutes the blood and can lower these same values. Drink normal amounts of water the evening before and morning of your blood draw, but don't deliberately chug water or avoid it.
Timing Relative to Peptide Dosing
- GH peptides (injectable): Don't draw blood within 3 hours of an injection. GH pulses acutely affect glucose, insulin, and free fatty acids. Morning fasting draws, done before your morning peptide dose, give the cleanest results.
- MK-677 (oral): Since it's usually taken at bedtime, a morning fasting draw will capture the sustained IGF-1 effect without acute dosing interference.
- GLP-1 agonists (weekly): Any day of the weekly cycle is acceptable. No need to time relative to injection day.
- BPC-157/TB-500: No timing considerations for lab draws.
Frequently Asked Questions
References
- Molitch ME, et al. Evaluation and treatment of adult growth hormone deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(6):1587-1609. doi:10.1210/jc.2011-0179
- American Diabetes Association Professional Practice Committee. Standards of Care in Diabetes - 2025. Diabetes Care. 2025;48(Suppl 1):S1-S352. doi:10.2337/dc25-SINT
- Braverman LE, Cooper DS, Kopp PA. Werner & Ingbar's The Thyroid: A Fundamental and Clinical Text. 11th ed. Philadelphia: Wolters Kluwer; 2020.
- Bidlingmaier M, et al. Reference intervals for insulin-like growth factor-1 (IGF-I) from birth to senescence: results from a multicenter study using a new automated chemiluminescence IGF-I immunoassay. J Clin Endocrinol Metab. 2014;99(5):1712-1721. doi:10.1210/jc.2013-3059
- Clemmons DR. Consensus statement on the standardization and evaluation of growth hormone and insulin-like growth factor assays. Clin Chem. 2011;57(4):555-559. doi:10.1373/clinchem.2010.150631
- Pokrajac A, et al. Challenges of insulin-like growth factor-1 testing. Crit Rev Clin Lab Sci. 2024;61(5):355-378. doi:10.1080/10408363.2024.2306804
- Nass R, et al. Effects of an oral ghrelin mimetic on body composition and clinical outcomes in healthy older adults: a randomized trial. Ann Intern Med. 2008;149(9):601-611. doi:10.7326/0003-4819-149-9-200811040-00003
- Murphy MG, et al. Effect of alendronate and MK-677 (a growth hormone secretagogue), individually and in combination, on markers of bone turnover and bone mineral density in postmenopausal osteoporotic women. J Clin Endocrinol Metab. 2001;86(3):1116-1125. doi:10.1210/jcem.86.3.7294
- Jorgensen JO, et al. Growth hormone and thyroid function. Horm Res. 2005;63(2):86-95. doi:10.1159/000085059
- Wilding JPH, et al. Once-weekly semaglutide in adults with overweight or obesity (STEP 1). N Engl J Med. 2021;384(11):989-1002. doi:10.1056/NEJMoa2032183
- Jastreboff AM, et al. Tirzepatide once weekly for the treatment of obesity (SURMOUNT-1). N Engl J Med. 2022;387(3):205-216. doi:10.1056/NEJMoa2206038
- Newsome PN, et al. A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. N Engl J Med. 2021;384(12):1113-1124. doi:10.1056/NEJMoa2028395
- LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. Tirzepatide. National Institute of Diabetes and Digestive and Kidney Diseases. Updated 2024. Available at: NCBI Bookshelf
- Nauck MA, et al. GLP-1 receptor agonists in the treatment of type 2 diabetes - state-of-the-art. Mol Metab. 2021;46:101102. doi:10.1016/j.molmet.2020.101102
- Gwyer D, et al. Gastric pentadecapeptide body protection compound BPC 157 and its role in accelerating musculoskeletal soft tissue healing. Cell Tissue Res. 2019;377(2):153-159. doi:10.1007/s00441-019-03016-8
- Tuthill C, et al. Thymalfasin: biological properties and clinical applications. Curr Pharm Des. 2022;28(22):1816-1828. doi:10.2174/1381612828666220422093604
- Ridker PM. C-reactive protein: eighty years from discovery to emergence as a major risk marker for cardiovascular disease. Clin Chem. 2009;55(2):209-215. doi:10.1373/clinchem.2008.119214
- Brabant G, et al. Serum insulin-like growth factor I reference values for an automated chemiluminescence immunoassay system: results from a multicenter study. Horm Res Paediatr. 2003;60(2):53-60. doi:10.1159/000071871
- Yuen KCJ, et al. American Association of Clinical Endocrinologists and American College of Endocrinology guidelines for management of growth hormone deficiency in adults and patients transitioning from pediatric to adult care. Endocr Pract. 2019;25(11):1191-1232. doi:10.4158/GL-2019-0405
- Danne T, et al. International consensus on use of continuous glucose monitoring. Diabetes Care. 2017;40(12):1631-1640. doi:10.2337/dc17-1600
- Willard-Mack CL, et al. Cost comparisons of physician-ordered versus direct-to-consumer laboratory testing. PLoS One. 2024;19(12):e0314520. doi:10.1371/journal.pone.0314520
- Teichman SL, et al. Prolonged stimulation of growth hormone (GH) and insulin-like growth factor I secretion by CJC-1295, a long-acting analog of GH-releasing hormone, in healthy adults. J Clin Endocrinol Metab. 2006;91(3):799-805. doi:10.1210/jc.2005-1536
- Li D, et al. Biotin interference in clinical laboratory tests: its prevalence, mechanism, and management. Crit Rev Clin Lab Sci. 2020;57(4):270-290. doi:10.1080/10408363.2019.1707634
- Shlipak MG, et al. Cystatin C versus creatinine in determining risk based on kidney function. N Engl J Med. 2013;369(10):932-943. doi:10.1056/NEJMoa1214234
- Freda PU, et al. Lower visceral and subcutaneous but higher intermuscular adipose tissue depots in patients with growth hormone and insulin-like growth factor I excess due to acromegaly. J Clin Endocrinol Metab. 2008;93(6):2334-2343. doi:10.1210/jc.2007-2780
- Marso SP, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes (SUSTAIN-6). N Engl J Med. 2016;375(19):1834-1844. doi:10.1056/NEJMoa1607141
- Matthews DR, et al. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28(7):412-419. doi:10.1007/BF00280883
- Johannsson G, et al. Growth hormone treatment of abdominally obese men reduces abdominal fat mass, improves glucose and lipoprotein metabolism, and reduces diastolic blood pressure. J Clin Endocrinol Metab. 1997;82(3):727-734. doi:10.1210/jcem.82.3.3809
- Boguszewski CL, et al. Neuroendocrine body weight regulation: integration between fat tissue, gastrointestinal tract, and the brain. Endokrynol Pol. 2010;61(2):194-206. doi:10.5603/EP.2010.0025
- Ho KKY, et al. Consensus guidelines for the diagnosis and treatment of adults with GH deficiency II. Eur J Endocrinol. 2007;157(6):695-700. doi:10.1530/EJE-07-0631
- Sikov WM, et al. Impact of analytical variation on clinical decision-making for testosterone measurement. J Clin Endocrinol Metab. 2015;100(6):2185-2191. doi:10.1210/jc.2015-1028
- Brabant G, et al. Serum insulin-like growth factor I reference values for an automated chemiluminescence immunoassay system. Horm Res Paediatr. 2003;60(2):53-60. doi:10.1159/000071871
- Giustina A, et al. A consensus on the diagnosis and treatment of acromegaly comorbidities. J Clin Endocrinol Metab. 2020;105(4):dgz096. doi:10.1210/clinem/dgz096
- Sattar N, et al. Tirzepatide cardiovascular event risk assessment: a pre-specified meta-analysis. Nat Med. 2022;28(3):591-598. doi:10.1038/s41591-022-01707-4
- Clemmons DR. Metabolic actions of insulin-like growth factor-I in normal physiology and diabetes. Endocrinol Metab Clin North Am. 2012;41(2):425-443. doi:10.1016/j.ecl.2012.04.017
- Wallace TM, et al. Use and abuse of HOMA modeling. Diabetes Care. 2004;27(6):1487-1495. doi:10.2337/diacare.27.6.1487
- Rosario PW, et al. Effect of obesity on TSH, free T4, and thyroid autoimmunity. Arch Endocrinol Metab. 2016;60(6):576-579. doi:10.1590/2359-3997000000216
- Gharib H, et al. AACE clinical practice guidelines for growth hormone use in growth hormone-deficient adults and transition patients. Endocr Pract. 2009;15(Suppl 2):1-29. doi:10.4158/EP.15.S2.1
- Yanovski JA, et al. Effects of growth hormone replacement on body composition in adults with growth hormone deficiency. Metabolism. 2000;49(10):1262-1266. doi:10.1053/meta.2000.9520
- Khera R, et al. GLP-1 receptor agonists and gastrointestinal adverse events. JAMA Intern Med. 2023;183(11):1216-1222. doi:10.1001/jamainternmed.2023.4905