Executive Summary
Nicotinamide adenine dinucleotide (NAD+) sits at the crossroads of cellular energy production, DNA repair, and longevity signaling. As research accelerates into how we can restore age-related NAD+ decline, clinicians and patients face a practical question: what's the best way to get NAD+ into the body? This report examines the evidence behind intravenous (IV) infusions, subcutaneous (SubQ) injections, nasal sprays, and oral precursors like NMN and NR.
Key Takeaways
- IV NAD+ achieves the highest peak blood levels (up to 380% of baseline) but requires 2-4 hour clinical sessions and costs $500-1,500 per infusion
- Subcutaneous NAD+ injections offer convenient self-administration with estimated 210% peak levels and growing clinical adoption
- Intranasal NAD+ delivery may bypass the blood-brain barrier directly, though human data remain limited
- Oral NMN at 1g daily raises blood NAD+ approximately 40-60% above baseline in clinical trials
- The optimal strategy likely involves matching the delivery route to specific clinical goals and patient circumstances
NAD+ levels drop by as much as 50% between the ages of 40 and 60, driven largely by rising CD38 enzyme activity on immune cells and sustained PARP activation from accumulated DNA damage. This decline correlates with mitochondrial dysfunction, impaired sirtuin signaling, and the metabolic deterioration we associate with aging. The desire to reverse this trajectory has fueled a multi-billion-dollar supplement industry and a rapidly growing clinical IV therapy market.
Yet the science is more nuanced than marketing materials suggest. While IV NAD+ delivers 100% of the compound directly to the bloodstream, a 2019 pilot study found that plasma NAD+ levels didn't begin rising until two hours into a six-hour infusion, with significant urinary excretion observed shortly after (Grant et al., 2019). Subcutaneous injection offers a practical middle ground with slower, steadier absorption. Oral precursors like NMN and NR take an indirect path, requiring enzymatic conversion before they can raise tissue NAD+ levels.
This report synthesizes findings from clinical trials, pharmacokinetic studies, and real-world clinical protocols across all administration routes. We'll cover the biology of NAD+ metabolism, compare bioavailability data, examine clinical applications from anti-aging to addiction treatment, and provide practical protocol guidance. Whether you're a clinician designing treatment plans or a patient evaluating options, the goal here is clarity grounded in evidence.
Key Takeaways
- IV NAD+ achieves the highest peak blood levels (up to 380% of baseline) but requires 2-4 hour clinical sessions and costs $500-1,500 per infusion
- Subcutaneous NAD+ injections offer convenient self-administration with estimated 210% peak levels and growing clinical adoption
- Intranasal NAD+ delivery may bypass the blood-brain barrier directly, though human data remain limited
- Oral NMN at 1g daily raises blood NAD+ approximately 40-60% above baseline in clinical trials
- The optimal strategy likely involves matching the delivery route to specific clinical goals and patient circumstances
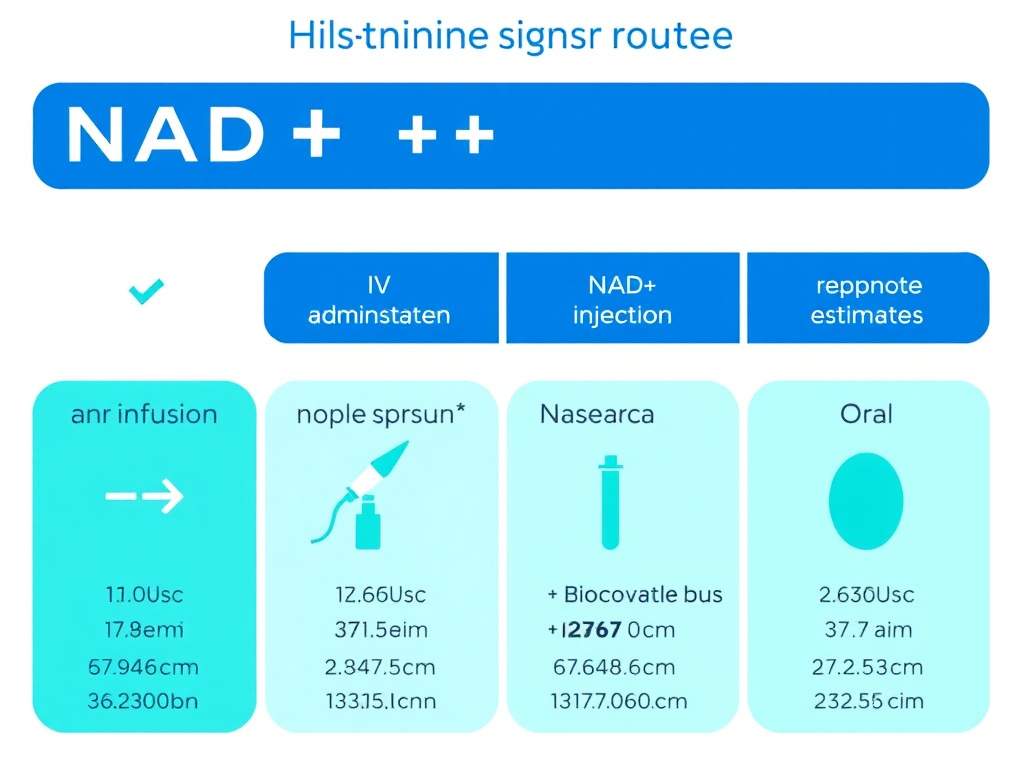
Figure 1: Comparison of NAD+ administration routes and their relative bioavailability profiles based on available clinical data.
Understanding NAD+: What Every Patient Should Know
If you're considering NAD+ therapy, whether through IV infusion, subcutaneous injection, nasal spray, or oral supplements, you deserve a clear understanding of what this molecule does, why it matters, and what realistic expectations look like. This section distills the complex biochemistry into practical knowledge that helps you make informed decisions.
NAD+ in Plain Language
Think of NAD+ as a molecular currency that your cells spend to perform their most essential functions. Every time a cell repairs damaged DNA, generates energy from food, or activates protective stress-response programs, it spends a molecule of NAD+. Your body recycles most of this spent NAD+ through a process called the salvage pathway, but the recycling system becomes less efficient as you age. Meanwhile, the demand for NAD+ increases because aging cells accumulate more damage that needs repairing.
The result is a widening gap between NAD+ supply and demand that begins around age 30-40 and accelerates through middle age and beyond. By the time most people reach their 50s and 60s, their NAD+ levels may be only half of what they were in their 20s. This shortfall doesn't cause a single disease, but it undermines the cellular maintenance programs that keep you healthy. It's like reducing the maintenance budget for a building - nothing breaks immediately, but over time, the deferred maintenance accumulates and systems start failing.
Why Your Doctor May Not Know About NAD+
NAD+ therapy exists at the intersection of mainstream medicine and functional/integrative medicine. Most conventional physicians receive minimal training in cellular metabolism during medical school and none in NAD+ supplementation strategies. This doesn't mean NAD+ therapy is unscientific; the basic biology is well-established and published in top-tier scientific journals. It means that clinical application has advanced faster in functional medicine practices than in conventional medical settings.
The research supporting NAD+ therapy is growing rapidly, with new human clinical trials published regularly. The challenge is that most studies to date have been small pilot trials or observational series rather than the large, multi-center, randomized controlled trials that conventional medicine requires for formal treatment guidelines. This gap is closing as larger trials are underway, but for now, NAD+ therapy remains an evidence-informed rather than evidence-established intervention for most applications.
What NAD+ Therapy Can and Cannot Do
What NAD+ therapy can realistically offer:
- Restoration of cellular energy production capacity in tissues with depleted NAD+ stores
- Reactivation of sirtuin-mediated cellular maintenance and repair programs
- Improvement in subjective energy, mental clarity, and physical vitality (reported by the majority of patients)
- Support for metabolic health parameters including insulin sensitivity and blood pressure
- Acceleration of withdrawal symptom resolution in addiction treatment settings
- Potential neuroprotective benefits, particularly through intranasal or IV delivery
What NAD+ therapy cannot do:
- Reverse aging (it can address one component of aging biology, but aging is a multi-factorial process)
- Cure specific diseases (NAD+ is a metabolic support, not a pharmaceutical treatment for any diagnosed condition)
- Replace healthy lifestyle habits (exercise, nutrition, sleep, and stress management remain the foundation of health)
- Produce permanent results without continued administration (NAD+ levels will return toward baseline if supplementation stops)
- Work equally well for everyone (individual response varies based on baseline NAD+ status, genetics, and health conditions)
Questions to Ask Your Provider
If you're considering NAD+ therapy, here are questions to discuss with your healthcare provider:
- What's the clinical rationale for NAD+ therapy in my specific case?
- Which delivery route do you recommend and why?
- What baseline labs should I have before starting?
- What's the expected timeline for noticing benefits?
- How will we monitor whether the therapy is working?
- What are the expected side effects and how are they managed?
- What's the total expected cost, including labs and follow-up visits?
- How long should I plan to continue therapy?
- Are there any interactions with my current medications?
- Where is the NAD+ sourced from, and what quality standards does the pharmacy follow?
A knowledgeable provider should be able to answer these questions clearly and honestly, including acknowledging the limitations of current evidence where appropriate. Be cautious of providers who make extravagant claims or dismiss the limitations of the current research base.
The History of NAD+ Therapy
Understanding the history of NAD+ therapy provides important context for evaluating current clinical practices and research directions. NAD+ wasn't discovered yesterday. Its therapeutic application spans over six decades, though the science supporting it has only recently caught up to the clinical intuition of early pioneers.
Early Discovery and Biochemistry (1906-1960s)
NAD+ was first identified in 1906 by Arthur Harden and William John Young during their investigation of fermentation in yeast extracts. They noticed that a heat-stable, low-molecular-weight factor was required for fermentation to proceed, which they called "cozymase." The complete structure of NAD+ was elucidated in the 1930s, and its role as an electron carrier in metabolism was established through the work of Otto Warburg, who received the Nobel Prize in 1931 for related work on cellular respiration.
The connection between NAD+ and human disease was established early through the study of pellagra, a disease caused by severe niacin (vitamin B3) deficiency. Pellagra, characterized by dermatitis, diarrhea, dementia, and death (the "4 Ds"), was endemic in populations with corn-based diets lacking adequate tryptophan and niacin. The recognition that niacin supplementation could cure pellagra was one of the great public health achievements of the early 20th century, and it established the principle that NAD+ precursors could treat disease.
The O'Hollaren Era: NAD+ for Addiction (1960s-1980s)
Dr. Paul O'Hollaren, a South African physician practicing in the United States, published the first reports of using intravenous NAD+ (then called "diphosphopyridine nucleotide" or DPN) for the treatment of addiction in the early 1960s. O'Hollaren observed that patients receiving IV NAD+ during alcohol and drug withdrawal experienced dramatically reduced symptoms and improved outcomes compared to standard detoxification protocols.
His 1961 publication in the Western Journal of Surgery described using IV NAD+ for the prevention, diagnosis, and treatment of drug addiction. While his work was ahead of its time and lacked the controlled trial methodology that modern evidence-based medicine demands, his clinical observations were remarkably consistent with what contemporary practitioners report over half a century later.
O'Hollaren's work was largely forgotten by mainstream medicine but was kept alive by a small community of practitioners who continued to use IV NAD+ for addiction treatment. This clinical tradition formed the basis for the modern resurgence of interest in NAD+ therapy.
The Sirtuin Revolution (2000s)
The modern era of NAD+ research began with the discovery of sirtuins and their role in aging. Leonard Guarente's laboratory at MIT identified Sir2 (the yeast homolog of mammalian SIRT1) as a longevity gene that required NAD+ for its activity. This finding, published in 2000, established the first mechanistic link between NAD+ metabolism and the biology of aging.
David Sinclair's group at Harvard subsequently demonstrated that resveratrol, a natural compound found in red wine, could activate SIRT1 and extend lifespan in model organisms. While the direct SIRT1-activating properties of resveratrol were later debated, the research generated enormous interest in the sirtuin-NAD+ axis as a therapeutic target. Sinclair's later work focusing directly on NAD+ precursors (particularly NMN) as more reliable sirtuin activators brought NAD+ supplementation into the mainstream longevity discussion.
The Precursor Supplement Era (2010s-Present)
The commercialization of NR as a dietary supplement (branded as Niagen by ChromaDex) beginning around 2013 made NAD+ precursor supplementation available to the general public for the first time. NR received FDA GRAS (Generally Recognized as Safe) status in 2016 and New Dietary Ingredient (NDI) notification acceptance, providing a regulatory pathway for commercial sales.
NMN entered the supplement market shortly after, initially through international (primarily Japanese) suppliers. The regulatory status of NMN has been more complex, with the FDA initially accepting NMN as a dietary supplement before later questioning its status due to its investigation as a pharmaceutical drug. As of the current date, NMN remains widely available through supplement retailers despite ongoing regulatory uncertainty in some markets.
The IV NAD+ Renaissance (2015-Present)
The convergence of mainstream longevity research with the clinical tradition of IV NAD+ therapy has produced a renaissance in NAD+ infusion clinics. Dr. Richard Mestayer at Springfield Wellness Center in Louisiana was instrumental in reviving clinical IV NAD+ therapy for addiction, building on O'Hollaren's legacy with modern protocols and published case series.
Today, IV NAD+ therapy is offered by hundreds of clinics across the United States and internationally. The treatment has expanded beyond addiction to include anti-aging, cognitive enhancement, chronic fatigue, and general wellness applications. The rapid growth of this market has outpaced the development of standardized protocols and rigorous clinical evidence, creating both opportunity and risk for patients.
The current moment represents a critical juncture for NAD+ therapy. Foundational science is strong, clinical observations are consistently favorable, and regulatory pathways exist for both pharmaceutical and supplement delivery. What's needed now is the large-scale clinical trial data that will either validate or challenge the assumptions underlying current clinical practice.
NAD+ Biology Review
Before comparing delivery routes and protocols, it's essential to understand what NAD+ actually does in the body, why it declines with age, and which enzymes compete for this finite resource. NAD+ isn't just another supplement target. It's a coenzyme present in every living cell, required for hundreds of metabolic reactions that keep you alive.
The Central Role of NAD+ in Cellular Metabolism
NAD+ functions as an electron carrier in mitochondrial oxidative phosphorylation, shuttling electrons through the electron transport chain to generate ATP. Without adequate NAD+, cells simply cannot produce energy efficiently. The ratio of NAD+ to its reduced form NADH acts as a metabolic rheostat, influencing everything from glycolysis to fatty acid oxidation.
But NAD+ does far more than ferry electrons. It serves as an obligate substrate for three major families of signaling enzymes: sirtuins, PARPs, and CD38/CD157 ectoenzymes. Each of these families consumes NAD+ when they function, breaking it apart rather than simply borrowing it. This consumption creates a constant demand for NAD+ replenishment, a demand that becomes harder to meet as we age.
The Sirtuin Family: NAD+-Dependent Longevity Regulators
Sirtuins (SIRT1-7) are NAD+-dependent deacylases and ADP-ribosyltransferases that regulate gene expression, mitochondrial biogenesis, stress resistance, and inflammation. When NAD+ levels are abundant, sirtuin activity increases, promoting cellular maintenance programs associated with longevity. When NAD+ drops, sirtuin activity declines, and cells shift from protective maintenance toward survival mode.
SIRT1, the most studied family member, deacetylates targets including p53, PGC-1alpha, and NF-kB, promoting DNA repair, mitochondrial biogenesis, and anti-inflammatory signaling respectively. SIRT3 operates within mitochondria, where it regulates the electron transport chain and protects against oxidative stress. SIRT6 maintains genomic stability and telomere integrity. Each of these enzymes requires one molecule of NAD+ per deacetylation reaction, producing nicotinamide (NAM) as a byproduct.
The connection between sirtuins and longevity has been demonstrated across species from yeast to mammals. Overexpression of SIRT1 extends lifespan in model organisms, while genetic deletion accelerates aging phenotypes. Restoring NAD+ levels in aged mice reactivates sirtuin-dependent pathways and reverses aspects of mitochondrial decline (Imai & Guarente, 2014).
PARPs: The DNA Repair Responders
Poly(ADP-ribose) polymerases, particularly PARP1 and PARP2, are DNA damage sensors that consume NAD+ to build poly(ADP-ribose) chains at sites of DNA breaks. This process, called PARylation, recruits repair machinery to damaged DNA. Under normal conditions, PARP activity is moderate and well-balanced with NAD+ supply.
The problem emerges with chronic DNA damage. As we age, cumulative oxidative stress, environmental exposures, and replication errors increase the burden of DNA damage. PARP1 activity escalates in response, consuming ever-larger quantities of NAD+. Sustained hyperactivation of PARP1 can deplete cellular NAD+ pools to the point where other NAD+-dependent enzymes, especially sirtuins, can no longer function properly. In extreme cases, NAD+ depletion through PARP hyperactivation triggers a form of cell death called parthanatos.
This creates a tragic competition: the more DNA damage a cell accumulates, the more NAD+ gets diverted to repair, leaving less available for the sirtuin-mediated maintenance programs that could have prevented some of that damage in the first place.
CD38: The Primary NAD+ Consumer in Aging
CD38 is a transmembrane glycoprotein expressed on immune cells that functions as an NADase, breaking down NAD+ to generate cyclic ADP-ribose (cADPR) and other calcium-mobilizing second messengers. What makes CD38 particularly significant for aging is that its expression increases dramatically with age, and it appears to be the single largest contributor to age-related NAD+ decline in multiple tissues.
Camacho-Pereira et al. (2016) demonstrated in Cell Metabolism that CD38 dictates age-related NAD+ decline and mitochondrial dysfunction through a SIRT3-dependent mechanism. CD38 knockout mice maintain youthful NAD+ levels well into old age and show preserved mitochondrial function. Conversely, CD38 overexpression in young mice mimics the NAD+ decline seen in aging.
The story becomes more complex with the discovery that senescent cells promote tissue NAD+ decline by activating CD38+ macrophages (Chini et al., 2020). As senescent cells accumulate with age, they secrete inflammatory factors that recruit and activate macrophages, which in turn upregulate CD38 expression. These CD38-high macrophages then consume local NAD+ stores, creating a feed-forward cycle of NAD+ depletion and cellular dysfunction.
The NAD+ Competition
Three major enzyme families compete for the same limited NAD+ pool: sirtuins (longevity regulation), PARPs (DNA repair), and CD38 (immune signaling). As CD38 expression rises with age, it consumes an increasing share of available NAD+, starving sirtuins and compromising cellular maintenance. This competition is a primary driver of age-related metabolic decline and a key rationale for NAD+ restoration therapy.
The NAD+ Salvage Pathway
Cells don't synthesize most of their NAD+ from scratch. Instead, they recycle it through the salvage pathway, which recovers the nicotinamide (NAM) released when sirtuins, PARPs, and CD38 consume NAD+. The salvage pathway is the dominant source of intracellular NAD+ in most tissues.
The rate-limiting enzyme in this pathway is nicotinamide phosphoribosyltransferase (NAMPT), which converts NAM to nicotinamide mononucleotide (NMN). NMN is then converted to NAD+ by nicotinamide mononucleotide adenylyltransferases (NMNATs), of which three isoforms exist in different cellular compartments: NMNAT1 (nucleus), NMNAT2 (cytoplasm and Golgi), and NMNAT3 (mitochondria).
NAMPT expression and activity decline with aging, reducing the efficiency of NAD+ recycling precisely when demand is highest. This is one reason why supplementation with NAD+ precursors like NMN (which enters the pathway downstream of NAMPT) and NR (which is phosphorylated to NMN by NR kinases) can bypass the age-related bottleneck.
The de novo synthesis pathway, which generates NAD+ from tryptophan through the kynurenine pathway, contributes a smaller share of total NAD+ production but becomes increasingly relevant in inflammatory states, where indoleamine 2,3-dioxygenase (IDO) is upregulated.
Why NAD+ Declines With Age
The age-related decline in NAD+ is now understood to result from multiple converging factors:
- Increased CD38 expression: Rising CD38 on senescent cell-activated macrophages is the largest single driver of tissue NAD+ depletion
- Chronic PARP activation: Accumulated DNA damage leads to sustained PARP1 activity and NAD+ consumption
- Decreased NAMPT: Lower expression of the rate-limiting salvage pathway enzyme reduces NAD+ recycling efficiency
- Reduced NAD+ biosynthesis: De novo synthesis from tryptophan may become less efficient with age
- Inflammatory feedback loops: Chronic low-grade inflammation (inflammaging) promotes CD38 expression and PARP activation simultaneously
Human studies have documented a roughly 50% decline in tissue NAD+ levels between ages 40 and 60, with continued decline thereafter. This timeline correlates with the acceleration of age-related diseases and the onset of measurable mitochondrial dysfunction in most individuals.
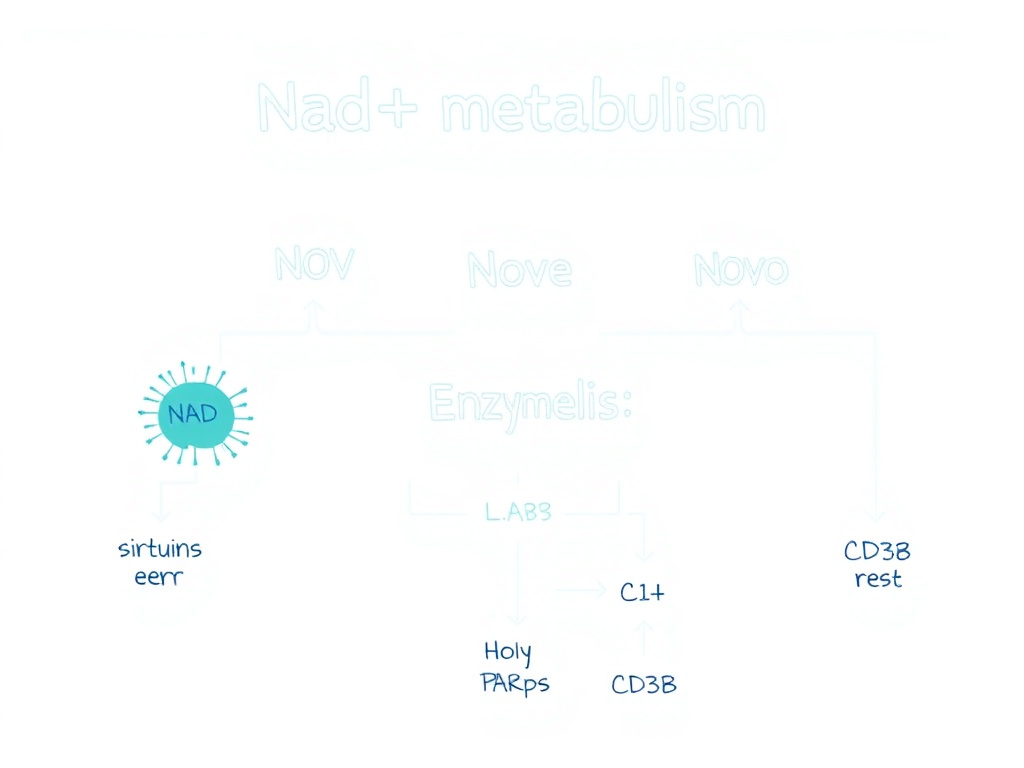
Figure 2: NAD+ metabolic pathways including the salvage pathway (NAMPT/NMNAT), de novo synthesis from tryptophan, and the three major NAD+-consuming enzyme families.
Therapeutic Rationale for NAD+ Restoration
The convergence of declining NAD+ supply and increasing NAD+ demand creates a widening metabolic deficit that many researchers believe underlies multiple hallmarks of aging. Restoring NAD+ levels in aged mice has been shown to improve mitochondrial function, enhance sirtuin activity, reduce inflammation, improve insulin sensitivity, and even extend lifespan in some models.
Complementary peptide therapies like Epithalon (which targets telomere maintenance) and MOTS-c (a mitochondrial-derived peptide) work through parallel longevity pathways that intersect with NAD+ signaling. MOTS-c, for example, activates AMPK and enhances mitochondrial function, pathways that are also supported by adequate NAD+ and sirtuin activity. The combination of NAD+ restoration with these targeted peptides represents an emerging area of biohacking research.
The key question for clinical practice isn't whether NAD+ restoration matters. The evidence strongly suggests it does. The question is how to most effectively deliver NAD+ or its precursors to achieve meaningful tissue-level increases. That's what the remainder of this report addresses.
NAD+ and the Hallmarks of Aging Framework
The "Hallmarks of Aging" framework, first proposed by López-Otín et al. in 2013 and updated in 2023, identifies twelve interconnected biological processes that drive aging. NAD+ decline intersects with a remarkable number of these hallmarks, making it one of the broadest single therapeutic targets in aging biology.
Genomic instability: NAD+ is required for PARP-mediated DNA repair and SIRT6-dependent maintenance of genomic stability. NAD+ depletion directly compromises the cell's ability to repair DNA damage, accelerating the accumulation of mutations.
Telomere attrition: SIRT6, an NAD+-dependent deacetylase, maintains telomeric chromatin structure. Loss of SIRT6 activity due to NAD+ decline contributes to telomere instability and dysfunction.
Epigenetic alterations: SIRT1, SIRT6, and SIRT7 are NAD+-dependent histone deacetylases that maintain epigenetic patterns. NAD+ decline leads to epigenetic drift - progressive changes in gene expression that characterize aging cells.
Loss of proteostasis: Sirtuins regulate autophagy, the cellular recycling system that clears damaged proteins and organelles. NAD+ depletion impairs autophagic flux, leading to accumulation of damaged proteins.
Disabled macroautophagy: SIRT1 directly activates autophagy-related proteins (ATGs), and adequate NAD+ levels are required for efficient autophagic clearance of damaged cellular components.
Deregulated nutrient sensing: NAD+/SIRT1 signaling intersects with insulin/IGF-1, mTOR, and AMPK pathways. NAD+ decline disrupts the coordinated nutrient sensing that maintains metabolic homeostasis.
Mitochondrial dysfunction: NAD+ is directly required for mitochondrial electron transport and indirectly supports mitochondrial quality control through SIRT3 and mitophagy regulation.
Cellular senescence: Senescent cells promote NAD+ decline through CD38 upregulation on local macrophages, while NAD+ depletion may also promote senescence through impaired DNA repair and SIRT1 deficiency.
Altered intercellular communication: NAD+/SIRT1-mediated suppression of NF-kB modulates the inflammatory signaling that drives age-related changes in tissue communication.
Chronic inflammation: The bidirectional relationship between NAD+ depletion and chronic inflammation creates a feed-forward cycle that accelerates inflammaging.
This extensive intersection with the hallmarks of aging explains why NAD+ restoration has shown benefits across so many different organ systems and disease models. Few other single interventions touch as many aging mechanisms simultaneously, which is both the promise and the complexity of NAD+ therapy.
Routes of Administration
NAD+ can reach the bloodstream and tissues through several different routes, each with distinct pharmacokinetic profiles, practical considerations, and trade-offs. Understanding these differences is critical for clinicians designing protocols and patients making informed decisions about their care.
Intravenous (IV) Infusion
IV infusion delivers NAD+ directly into the venous circulation, bypassing all absorption barriers. This route achieves 100% bioavailability in the strict pharmacokinetic sense, meaning every milligram administered reaches the bloodstream. Standard IV NAD+ protocols typically involve infusing 250-1,000 mg of NAD+ dissolved in normal saline over 2-4 hours.
The infusion rate matters considerably. Rapid infusion causes uncomfortable side effects including chest tightness, nausea, headache, and the characteristic "NAD flush," which results from conversion of some NAD+ to nicotinic acid, triggering prostaglandin-mediated vasodilation. Most clinics start at a slow drip rate and gradually increase as tolerated, which is why sessions often last 2-4 hours even for moderate doses.
Grant et al. (2019) conducted the first published pilot study examining the pharmacokinetics of IV NAD+ infusion in humans. Eight healthy participants received a continuous 6-hour infusion of 3 mcg/min/kg NAD+. Plasma NAD+ levels did not begin to rise until approximately 2 hours into the infusion, peaked toward the end of infusion, and increased urinary excretion of NAD+ metabolites was observed at 6 hours. This suggests that a substantial portion of infused NAD+ is rapidly metabolized or excreted rather than being taken up by tissues.
A 2024 randomized, placebo-controlled pilot study compared 500 mg IV NAD+, 500 mg IV nicotinamide riboside (NR), 500 mg oral NR, and saline placebo. The results revealed that IV NR actually outperformed IV NAD+ for raising whole-blood NAD+ levels, with NR IV promoting a 20.7% increase in peak NAD+ concentration relative to baseline. This finding challenges the assumption that direct NAD+ infusion is necessarily superior to precursor delivery.
Clinical Setting Requirements
IV NAD+ requires administration in a medical setting with appropriate monitoring. Patients need IV access, vital sign monitoring during infusion, and supervision by trained clinical staff. These requirements limit accessibility and increase cost but provide the highest degree of clinical oversight.
Subcutaneous (SubQ) Injection
Subcutaneous NAD+ injection has gained significant traction as a practical alternative to IV infusion. The compound is injected into the fatty tissue beneath the skin, typically in the abdomen, thigh, or upper arm. From there, NAD+ is absorbed gradually into the bloodstream through the capillary network within subcutaneous tissue.
SubQ injection bypasses gastrointestinal degradation and hepatic first-pass metabolism, offering substantially higher bioavailability than oral routes. The absorption kinetics are slower and more sustained compared to IV delivery, producing a lower but more prolonged peak in blood NAD+ levels. Clinical estimates suggest subcutaneous NAD+ produces peak blood levels approximately 210% of baseline, compared to up to 380% with IV infusion.
The practical advantages of SubQ injection are considerable. Patients can self-administer at home after proper training, sessions take 1-2 minutes rather than 2-4 hours, and the cost per dose is substantially lower than IV infusion. The primary drawback is injection site discomfort; NAD+ solutions can cause a stinging or burning sensation at the injection site that lasts several minutes.
Typical SubQ dosing ranges from 50-200 mg per injection, administered daily or several times per week. Some protocols use SubQ injections as maintenance therapy following an initial IV loading phase.
Intramuscular (IM) Injection
Intramuscular NAD+ injection delivers the compound into muscle tissue, where it's absorbed somewhat faster than subcutaneous injection due to the higher blood flow in skeletal muscle. IM injection offers similar bioavailability to SubQ but with a slightly faster absorption profile and potentially more discomfort due to the larger volume of tissue displacement.
IM injection is less commonly used than SubQ for NAD+ administration because the SubQ route is easier for self-administration and produces adequate absorption. Some clinics offer IM injections as a rapid-administration option for patients who prefer fewer, higher-dose injections.
Intranasal (Nasal Spray)
NAD+ nasal spray represents an emerging delivery route with a unique potential advantage: direct nose-to-brain delivery. The olfactory and trigeminal nerves provide pathways from the nasal mucosa directly to the central nervous system, potentially bypassing the blood-brain barrier. This makes intranasal delivery particularly interesting for neurological applications of NAD+.
Animal studies have demonstrated that intranasal NAD+ administration significantly increases brain NAD+ content. Wei et al. (2007) showed that intranasal NAD+ at 10 mg/kg increased brain NAD+ levels and reduced brain damage in a rat model of transient focal ischemia. The compound reached brain tissue within minutes of administration.
Estimated bioavailability of intranasal NAD+ is 15-25%, substantially lower than injectable routes but potentially higher than oral NAD+ itself. The key distinction is that intranasal delivery may achieve higher brain concentrations relative to systemic levels compared to other routes. However, no human pharmacokinetic studies on intranasal NAD+ have been published to date, making this the least evidence-supported route from a clinical standpoint.
Typical nasal spray formulations deliver 50-100 mg per dose, with protocols calling for 1-3 daily administrations. The approach is non-invasive, requires no clinical supervision, and can be combined with other delivery routes.
Oral NAD+ Precursors: NMN and NR
Direct oral NAD+ supplementation is ineffective because the NAD+ molecule is too large and charged to cross intestinal epithelial membranes intact, and it's degraded by digestive enzymes. Instead, oral supplementation strategies focus on NAD+ precursors, primarily nicotinamide mononucleotide (NMN) and nicotinamide riboside (NR), which can be absorbed and converted to NAD+ intracellularly.
Nicotinamide Riboside (NR): NR enters cells via equilibrative nucleoside transporters (ENTs) and is phosphorylated to NMN by NR kinases (NRK1 and NRK2), then converted to NAD+ by NMNATs. Clinical trials have shown that oral NR at 1,000 mg/day can increase blood NAD+ levels by approximately 60% after six weeks of supplementation (Martens et al., 2018). NR has the most extensive human clinical trial data of any NAD+ precursor, with multiple published randomized controlled trials.
Nicotinamide Mononucleotide (NMN): The mechanism by which NMN enters cells has been debated. Current evidence suggests that NMN is first dephosphorylated to NR extracellularly, transported into cells as NR, then re-phosphorylated back to NMN before conversion to NAD+. However, a dedicated NMN transporter (Slc12a8) has been identified in some tissues. Clinical trials with oral NMN at 250-900 mg/day have demonstrated significant increases in blood NAD+ concentrations at both 30 and 60 days of supplementation.
The oral precursor route is the most accessible and affordable option, but it involves multiple enzymatic conversion steps, hepatic first-pass metabolism, and variable absorption. Typical oral bioavailability estimates for NR and NMN range from 30-50% for the precursor molecule itself, with subsequent conversion to NAD+ adding another variable layer.
Sublingual Administration
Some NAD+ and NMN products are formulated for sublingual administration, where the compound dissolves under the tongue and absorbs through the oral mucosa directly into the bloodstream. This route avoids gastrointestinal degradation and first-pass liver metabolism, theoretically improving bioavailability compared to swallowed oral supplements.
Clinical data on sublingual NAD+ or NMN bioavailability are very limited. Anecdotal clinical reports suggest improved tolerance and potentially faster onset of effects compared to swallowed formulations, but controlled pharmacokinetic comparisons are lacking.
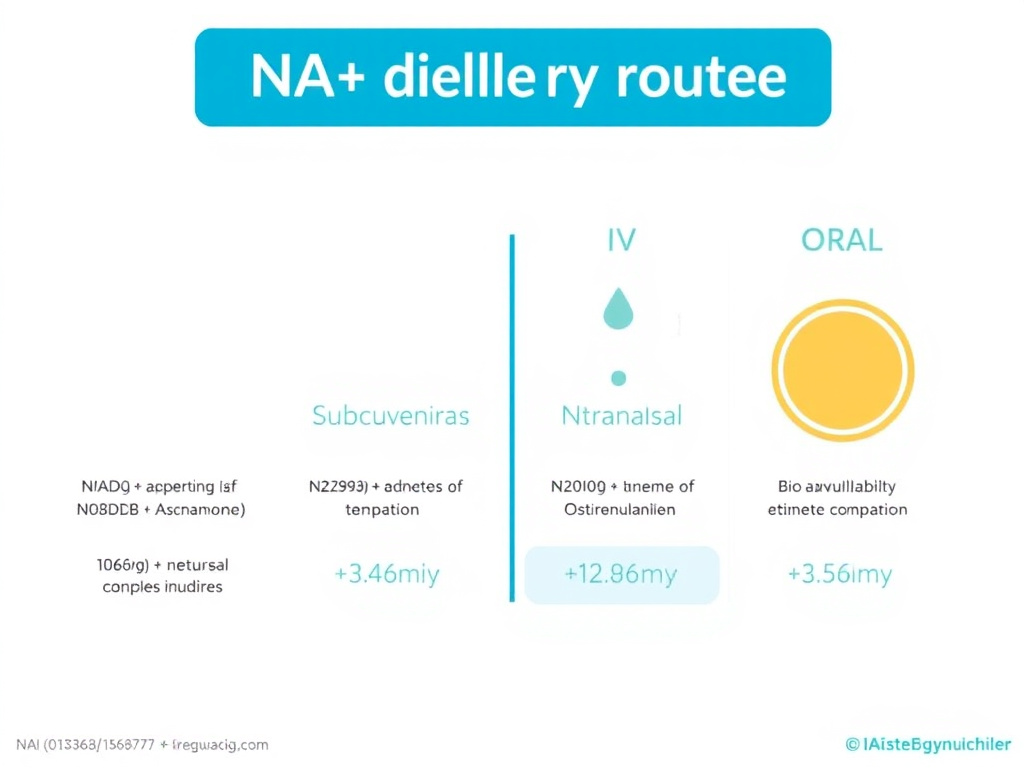
Figure 3: NAD+ delivery route comparison with approximate bioavailability, onset time, and practical considerations for each administration method.
Bioavailability Comparison
Bioavailability is the central question in NAD+ therapy. Delivering NAD+ or its precursors to the bloodstream is only half the battle; the compound must ultimately reach intracellular compartments where sirtuins, PARPs, and other NAD+-dependent enzymes operate. Let's examine what the data actually show for each route.
Defining Bioavailability in the NAD+ Context
Traditional pharmacokinetic bioavailability measures the fraction of an administered compound that reaches systemic circulation unchanged. For IV administration, this is by definition 100%. But for NAD+ therapy, this metric can be misleading. NAD+ that reaches the bloodstream may be rapidly degraded by extracellular CD38, dephosphorylated by CD73, or excreted renally before it can enter cells.
A more meaningful metric would be "functional bioavailability," which reflects how much the administered compound ultimately increases intracellular NAD+ levels in target tissues. Unfortunately, measuring intracellular NAD+ in human tissues is technically challenging, so most clinical studies rely on whole-blood or plasma NAD+ measurements as surrogates.
IV NAD+ Pharmacokinetics
The Grant et al. (2019) study published in Frontiers in Aging Neuroscience provides the most detailed pharmacokinetic data for IV NAD+. Key findings from this 6-hour continuous infusion study include:
| Parameter | Finding |
|---|---|
| Onset of plasma NAD+ rise | ~2 hours after start of infusion |
| Peak plasma NAD+ increase | Up to 398% above baseline at end of infusion |
| Urinary NAD+ metabolites | Significantly elevated at 6 and 8 hours |
| Return to baseline | Within 24 hours post-infusion |
| Primary metabolites detected | MeNAM, Me2PY, Me4PY (methylated nicotinamide derivatives) |
The 2-hour lag before plasma levels rise suggests that infused NAD+ is initially being consumed by extracellular enzymes and distributed to tissues before measurable accumulation occurs in plasma. The significant urinary excretion indicates that a meaningful portion of administered NAD+ is being metabolized and cleared rather than retained in tissues.
Subcutaneous NAD+ Pharmacokinetics
Published pharmacokinetic data specifically for subcutaneous NAD+ injection are limited. Clinical observations suggest the following profile compared to IV:
| Parameter | IV NAD+ | SubQ NAD+ |
|---|---|---|
| Peak blood NAD+ (% baseline) | ~380% | ~210% |
| Time to peak | End of infusion (2-4 hrs) | 1-2 hours post-injection |
| Duration of elevation | 8-12 hours | 6-10 hours |
| Total exposure (AUC) | Higher | Lower but more sustained |
| Administration time | 2-4 hours | 1-2 minutes |
The lower peak but more sustained elevation with SubQ may actually be advantageous in some respects. Rapid, high-peak NAD+ delivery by IV may overwhelm cellular uptake mechanisms, leading to greater metabolic waste and renal excretion. A slower, more sustained delivery could theoretically allow more efficient cellular uptake, though this remains speculative without head-to-head tissue-level comparisons.
NAD+ Blood Levels by Administration Route
Oral Precursor Pharmacokinetics
Oral NR and NMN have the most extensive published pharmacokinetic data, though they measure precursor-to-NAD+ conversion rather than direct NAD+ delivery:
NR (Niagen) Human Data:
- Single dose of 1,000 mg NR increases whole-blood NAD+ by approximately 100-142% within 24 hours (Airhart et al., 2017)
- Chronic supplementation at 1,000 mg/day produces sustained ~60% elevation in blood NAD+ over 6 weeks (Martens et al., 2018)
- Peak plasma NR levels occur 2-3 hours post-dose, with NAD+ elevation following 4-8 hours later
- Steady-state NAD+ elevation is maintained with daily dosing
NMN Human Data:
- 250 mg/day increases blood NAD+ levels significantly by day 30, with continued elevation through day 60 (Yi et al., 2023)
- Doses up to 900 mg/day have been studied with no significant adverse effects
- Peak plasma NMN levels occur approximately 1-2 hours post-dose
- The time course of NMN-to-NAD+ conversion varies by tissue
Intranasal NAD+ Bioavailability
Systemic bioavailability of intranasal NAD+ is estimated at 15-25% based on extrapolation from animal data and comparison with other intranasally delivered molecules of similar size. The more relevant metric for nasal delivery may be brain bioavailability, which is expected to be proportionally higher relative to systemic exposure compared to other routes due to direct nose-to-brain transport.
Wei et al. (2007) demonstrated in rats that intranasal NAD+ significantly increased brain NAD+ content and reduced ischemic brain injury, confirming that the compound does reach brain tissue through this route. But translating rodent nasal anatomy data to humans requires caution, as the ratio of olfactory epithelium to total nasal surface area differs substantially between species.
Route Comparison: What the Data Actually Tell Us
Looking across all available data, several conclusions emerge:
Evidence-Based Route Selection
- For maximum acute NAD+ elevation: IV infusion produces the highest peak blood levels, though much of this may be metabolized and excreted rather than retained
- For sustained daily maintenance: Oral NR or NMN at adequate doses produces meaningful, sustained NAD+ elevation with the best long-term compliance
- For practical injectable therapy: SubQ NAD+ balances efficacy with convenience, offering higher bioavailability than oral routes without the time commitment of IV
- For neurological targets: Intranasal delivery offers the theoretical advantage of direct CNS access, though human evidence remains preliminary
- For combination approaches: Many clinicians use IV or SubQ loading followed by oral maintenance, with nasal spray as an adjunct for cognitive focus
Keep in mind that no large-scale, head-to-head clinical trial has directly compared all four routes using standardized outcome measures. The comparative data presented here represent a synthesis of separate studies with different designs, populations, and measurement methods. This is a significant limitation of the current evidence base.
Anti-Aging Applications
The anti-aging applications of NAD+ restoration therapy represent the largest area of interest and investment, driven by compelling preclinical data showing that boosting NAD+ levels can reverse multiple hallmarks of aging in animal models. Human evidence is still catching up to the animal data, but early clinical findings are encouraging.
Mitochondrial Function and Energy Production
Age-related mitochondrial dysfunction is one of the most well-documented consequences of NAD+ decline. As NAD+ levels fall, the efficiency of oxidative phosphorylation decreases, reactive oxygen species (ROS) production increases, and cellular energy output drops. This manifests as the fatigue, reduced exercise capacity, and metabolic dysfunction that characterize aging.
Gomes et al. (2013) published a landmark study in Cell showing that raising NAD+ levels in aged mice restored mitochondrial function to levels comparable to young mice within just one week of treatment. The improvement was mediated by SIRT1 activation of PGC-1alpha, the master regulator of mitochondrial biogenesis. These findings have been replicated in multiple laboratories using different NAD+ precursors.
Human data on mitochondrial function are more limited but supportive. The NADPARK study of NR supplementation in Parkinson's disease patients demonstrated improved cerebral NAD+ metabolism as measured by phosphorus-31 MRS (Brakedal et al., 2022). While this was a disease-specific study, it confirmed that oral NAD+ precursors can influence mitochondrial metabolism in human brain tissue.
The mitochondrial peptide MOTS-c works through complementary pathways, activating AMPK and enhancing mitochondrial function independently of NAD+. Some practitioners combine NAD+ restoration with MOTS-c to address mitochondrial dysfunction through multiple mechanisms simultaneously. Similarly, Humanin, another mitochondrial-derived peptide, provides cytoprotective effects that complement NAD+-mediated mitochondrial support.
DNA Repair and Genomic Stability
NAD+ is essential for PARP-mediated DNA repair and SIRT1/SIRT6-mediated maintenance of genomic stability. When NAD+ levels decline, DNA repair capacity decreases, leading to accelerated accumulation of mutations and genomic instability. This creates a vicious cycle: DNA damage increases PARP activity (consuming more NAD+), while reduced sirtuin activity impairs the cell's ability to prevent and respond to damage.
Li et al. (2017) demonstrated in Science that NMN supplementation improved DNA repair capacity in aged mice, reducing radiation-induced DNA damage. The effect was mediated by PARP1 activation (which requires NAD+ as a substrate) and SIRT1-dependent signaling. This finding generated substantial interest in NAD+ restoration as a radioprotective and DNA-protective strategy.
The peptide Epithalon targets genomic stability through a different mechanism, activating telomerase to maintain telomere length. The combination of NAD+-supported DNA repair with Epithalon's telomere protection represents a multi-pronged approach to genomic maintenance during aging. You can learn more about this strategy in our Epithalon research report.
Skin Aging and Cellular Senescence
NAD+ decline contributes to skin aging through multiple mechanisms. Reduced sirtuin activity impairs the skin's antioxidant defenses and repair capacity. Increased cellular senescence in the dermis leads to collagen degradation and reduced elasticity. The inflammatory secretome of senescent cells (SASP) further promotes tissue deterioration.
Preclinical studies have shown that topical and systemic NAD+ precursors can reduce UV-induced skin damage, promote keratinocyte survival, and reduce markers of cellular senescence in skin tissue. Clinical observations from NAD+ IV therapy providers consistently report that patients notice improvements in skin quality, hydration, and clarity within the first 2-4 weeks of treatment, though controlled clinical trials specifically measuring skin outcomes are limited.
The copper peptide GHK-Cu provides complementary skin-rejuvenation benefits through stimulation of collagen synthesis, decorin production, and tissue remodeling. Combining NAD+ restoration with GHK-Cu addresses skin aging from both the metabolic/cellular level and the structural/extracellular matrix level.
Metabolic Health and Insulin Sensitivity
NAD+ and sirtuin signaling play central roles in metabolic regulation. SIRT1 deacetylates and activates PGC-1alpha (promoting fatty acid oxidation), modulates gluconeogenesis through FOXO1 and HNF4alpha, and enhances insulin signaling. SIRT3 regulates mitochondrial metabolism and protects against metabolic stress. When NAD+ levels fall and sirtuin activity declines, metabolic flexibility decreases, contributing to insulin resistance and metabolic syndrome.
Yoshino et al. (2021) published a randomized controlled trial in Science showing that 250 mg/day NMN supplementation improved muscle insulin sensitivity in prediabetic postmenopausal women. The treatment enhanced insulin-stimulated glucose disposal and improved the expression of genes involved in muscle remodeling. While this was a relatively small study (25 participants), it provided the first rigorous human evidence linking NAD+ precursor supplementation to measurable metabolic improvement.
Martens et al. (2018) found that 6 weeks of NR supplementation (1,000 mg/day) tended to reduce blood pressure in adults with elevated baseline levels, though the study was not powered to detect this as a primary outcome. Supplementation also reduced aortic stiffness, a marker of vascular aging.
Inflammation and Immune Function
The relationship between NAD+ and inflammation is bidirectional. Chronic inflammation drives NAD+ decline (through CD38 upregulation), while NAD+ decline promotes inflammation (through reduced SIRT1-mediated inhibition of NF-kB). Breaking this cycle through NAD+ restoration may reduce the chronic low-grade inflammation, sometimes called "inflammaging," that underlies many age-related diseases.
SIRT1 directly deacetylates the p65 subunit of NF-kB, reducing its transcriptional activity and suppressing production of pro-inflammatory cytokines including TNF-alpha, IL-1beta, and IL-6. When NAD+ levels are sufficient to support strong SIRT1 activity, this anti-inflammatory brake functions properly. When NAD+ declines, SIRT1 activity falls, and inflammatory signaling increases unchecked.
The tissue-healing peptide BPC-157 modulates inflammatory pathways through distinct mechanisms, including promotion of growth factor expression and acceleration of tissue repair. Some clinical protocols combine NAD+ therapy with BPC-157 to address both the metabolic root causes of inflammation and the downstream tissue damage it produces.
Anti-Aging Summary
NAD+ restoration therapy addresses multiple hallmarks of aging simultaneously: mitochondrial dysfunction, genomic instability, cellular senescence, metabolic derangement, and chronic inflammation. The strongest human evidence supports improvements in metabolic parameters, while effects on mitochondrial function and DNA repair are well-established in animal models with emerging human confirmation. Most anti-aging NAD+ protocols target sustained elevation of tissue NAD+ levels through daily or regular administration rather than intermittent high-dose infusions.
Figure 4: Mechanisms linking NAD+ decline to the hallmarks of aging, showing how reduced sirtuin and PARP activity propagate cellular dysfunction.
Addiction & Recovery Research
NAD+ IV therapy for addiction treatment has one of the longest clinical histories of any NAD+ application, dating back to the work of Dr. Paul O'Hollaren in the 1960s and later refined by Dr. Richard Mestayer at Springfield Wellness Center. While the evidence base remains limited to observational studies and small trials, the clinical reports are consistently favorable, and interest from the addiction medicine community is growing.
Neurobiological Rationale
Substance abuse disorders cause profound disruption of NAD+-dependent pathways in the brain. Chronic alcohol exposure depletes hepatic and cerebral NAD+ stores by diverting the NAD+ precursor niacin toward alcohol metabolism and by directly damaging mitochondria. Opioid use disrupts dopaminergic signaling pathways that depend on NAD+-regulated enzymes. Stimulant abuse generates oxidative stress that activates PARPs and depletes NAD+.
The theory behind NAD+ infusion for addiction is straightforward: by rapidly restoring NAD+ levels in the brain, you can accelerate the repair of damaged neural circuits, reduce neuroinflammation, and support the restoration of normal neurotransmitter function. Higher NAD+ levels support sirtuin-mediated neuroprotection, PARP-dependent DNA repair in neurons, and improved mitochondrial function in energy-hungry brain tissue.
Clinical Evidence
Mestayer et al. (2022) published the largest case series to date, examining 50 patients with substance use disorder (SUD) who received NAD+ and enkephalinase inhibition (IV1114589NAD) infusions. Using validated psychiatric assessment scales, the researchers found statistically significant reductions in:
- Craving scores: P = 1.063 x 10^-9
- Anxiety scores: P = 5.487 x 10^-7
- Depression scores: P = 1.763 x 10^-4
These reductions followed a dose-dependent linear trend, meaning higher cumulative doses of NAD+ correlated with greater symptom improvement. The study included patients with alcohol, opioid, stimulant, and polysubstance use disorders.
An earlier study examining the Brain Restoration Plus (BR+NAD) protocol found that patients completing at least five IV NAD+ treatments over seven days, followed by oral treatments for 30+ days, showed strong long-term outcomes. At six-month follow-up, 91% of 23 evaluated subjects remained sober. At the study endpoint, 82% had experienced no relapse.
Withdrawal Symptom Management
One of the most consistently reported benefits of IV NAD+ in addiction settings is the significant attenuation of acute withdrawal symptoms. Clinical reports describe patients withdrawing from opioids without experiencing the severe physical symptoms (nausea, vomiting, muscle pain, insomnia, agitation) that typically characterize opioid cessation.
The mechanism likely involves rapid restoration of depleted cellular NAD+ in brainstem nuclei and limbic structures that mediate withdrawal symptoms. By supporting mitochondrial function and reducing neuroinflammation during the acute withdrawal period, NAD+ infusion may allow the brain to restabilize more quickly and with less distress.
It's critical to note that NAD+ IV therapy is not a standalone addiction treatment. The best outcomes are achieved when NAD+ infusion is integrated into a comprehensive treatment plan that includes behavioral therapy, counseling, peer support, and addressing underlying psychological and social factors. No responsible practitioner uses NAD+ as a replacement for evidence-based addiction treatment modalities.
Alcohol Use Disorder
Alcohol use disorder has a particularly strong rationale for NAD+ intervention. Chronic alcohol metabolism through alcohol dehydrogenase and aldehyde dehydrogenase consumes large quantities of NAD+, converting it to NADH. This shifts the NAD+/NADH ratio dramatically toward NADH, disrupting metabolic regulation throughout the liver and brain.
The resulting NAD+ depletion contributes to alcoholic liver disease, Wernicke-Korsakoff syndrome, and the cognitive deficits associated with chronic alcoholism. NAD+ restoration may address the metabolic foundation of alcohol-related organ damage while supporting neurological recovery during early sobriety.
Limitations of Current Evidence
Despite encouraging results, the addiction treatment evidence for NAD+ has significant limitations:
- No large-scale, randomized, double-blind, placebo-controlled trials have been completed
- Most studies are observational case series or retrospective analyses with inherent selection bias
- Placebo effects are difficult to control when patients are receiving prolonged IV infusions in a clinical setting
- Long-term follow-up data are limited, and reported sobriety rates may reflect self-selection
- The precise contribution of NAD+ versus other components of treatment protocols (counseling, environment change, supplement regimens) cannot be isolated
These limitations don't invalidate the clinical observations, but they do mean that NAD+ IV therapy for addiction should be considered an emerging intervention rather than an established evidence-based treatment.
Mechanisms of NAD+ in Addiction Recovery
Beyond general metabolic restoration, several specific mechanisms may explain NAD+'s apparent benefits in addiction treatment:
Dopaminergic circuit repair: Substance use disorders are fundamentally disorders of the brain's reward system, centered on dopaminergic signaling in the mesolimbic pathway. Chronic drug use depletes dopamine receptors, reduces dopamine synthesis capacity, and damages dopaminergic neurons. NAD+/SIRT1 signaling promotes the expression of tyrosine hydroxylase (the rate-limiting enzyme in dopamine synthesis) and protects dopaminergic neurons from oxidative damage. Restoring NAD+ may accelerate the recovery of normal dopaminergic function, reducing cravings and anhedonia.
Endorphin system modulation: The BR+NAD protocol used in many addiction treatment centers includes amino acid supplementation alongside NAD+ infusions. The rationale is that NAD+ supports the enzymatic pathways involved in endorphin and enkephalin synthesis, while specific amino acids provide precursors for these endogenous opioid peptides. By restoring the brain's natural opioid system, the protocol aims to reduce the perceived need for exogenous opioids.
Neuroinflammation reduction: Chronic substance abuse creates a state of neuroinflammation, with activated microglia producing pro-inflammatory cytokines that damage neurons and impair synaptic function. This neuroinflammation contributes to the cognitive deficits, emotional dysregulation, and persistent cravings seen in early recovery. NAD+/SIRT1-mediated suppression of NF-kB signaling may reduce neuroinflammation, creating a more favorable neurochemical environment for recovery.
Epigenetic reprogramming: Substance abuse creates lasting epigenetic changes that predispose to relapse. These include alterations in histone acetylation patterns at genes involved in reward processing, stress response, and impulse control. SIRT1, an NAD+-dependent histone deacetylase, may help remodel these epigenetic marks toward more favorable patterns, potentially reducing the biological drive toward relapse.
Mitochondrial restoration in brain tissue: The brain consumes approximately 20% of total body oxygen and is exquisitely sensitive to mitochondrial dysfunction. Substance abuse impairs brain mitochondrial function through oxidative stress, direct toxic effects, and nutrient depletion. Restoring brain NAD+ levels supports mitochondrial recovery, improving cellular energy production and reducing the fatigue and cognitive impairment that characterize early recovery.
Integration with Conventional Addiction Treatment
The most successful NAD+ addiction programs integrate infusion therapy within a comprehensive treatment model that includes:
- Medical detoxification supervision with appropriate withdrawal management
- Individual and group psychotherapy (cognitive-behavioral therapy, motivational interviewing)
- Peer support and 12-step or alternative recovery programming
- Nutritional rehabilitation and supplement support
- Exercise and physical rehabilitation
- Stress management and mindfulness training
- Family therapy and social support development
- Aftercare planning and relapse prevention strategies
NAD+ infusion serves as a biological accelerant within this framework, potentially reducing the severity and duration of acute withdrawal while supporting the neurobiological recovery that underlies successful long-term sobriety. It does not replace behavioral and psychological interventions, which address the cognitive, emotional, and social dimensions of addiction.
Specific Substance Considerations
Alcohol: Perhaps the strongest rationale for NAD+ therapy, given that alcohol metabolism directly depletes NAD+. Patients withdrawing from alcohol may benefit from both the metabolic restoration and the hepatoprotective effects of NAD+. Loading protocols for alcohol detox typically use 500-750 mg IV NAD+ daily for 7-10 days.
Opioids: NAD+ may support endogenous opioid system recovery and reduce the severity of withdrawal symptoms. Some protocols combine NAD+ with conventional opioid withdrawal management (buprenorphine taper, clonidine) to improve patient comfort. The goal is not to replace medication-assisted treatment but to enhance the neurobiological recovery process.
Stimulants (cocaine, methamphetamine): Stimulant abuse causes severe oxidative stress and dopaminergic neurotoxicity. NAD+'s antioxidant-supporting properties (through SIRT1-mediated upregulation of SOD2 and catalase) and its support of dopaminergic neuron repair make it theoretically valuable for stimulant addiction, though published clinical data are primarily from polysubstance use cohorts rather than stimulant-specific studies.
Benzodiazepines: Benzodiazepine withdrawal can be medically dangerous and requires careful medical management. NAD+ may support the neurological recovery process but should never be used as a substitute for medically supervised benzodiazepine tapering. The GABA system restoration during benzodiazepine withdrawal is a distinct process from NAD+-mediated metabolic recovery.
Figure 5: Typical NAD+ IV therapy protocol for addiction treatment showing the multi-day infusion schedule and reported clinical response pattern.
Neurodegenerative Disease
The connection between NAD+ depletion and neurodegeneration is among the most active areas of research in aging biology. The brain is the most metabolically demanding organ in the body, consuming roughly 20% of total oxygen and calories despite comprising only 2% of body weight. This extreme metabolic demand makes neural tissue particularly vulnerable to NAD+ decline.
Alzheimer's Disease
NAD+ levels are significantly reduced in the brains of Alzheimer's disease (AD) patients and in animal models of AD. This depletion appears to be both a consequence and a driver of AD pathology, creating a feedback loop that accelerates disease progression.
A 2025 study published in Cell Reports Medicine demonstrated that pharmacologic NAD+ restoration reversed advanced Alzheimer's phenotypes in mice, including cognitive deficits, amyloid pathology, and neuroinflammation. The treatment, using the neuroprotective compound P7C3-A20 to enhance NAD+ synthesis through the NAMPT-dependent salvage pathway, reversed cognitive deficits and neuropathology in advanced amyloid- and tau-driven AD models.
Separately, research published in 2025 found that NAD+ supplementation corrected aberrant RNA splicing caused by toxic tau protein. In mice with tau-related mutations, NAD+ supplements improved RNA splicing, restored brain function, and enhanced memory performance through regulation of differential alternative RNA splicing of EVA1C.
Several clinical trials are investigating NAD+ precursors in human AD patients. The NAVIGATOR trial is examining oral NR supplementation in adults with mild cognitive impairment, with outcomes including cognitive testing, AD biomarkers in cerebrospinal fluid, and brain imaging. Results are expected in the coming years.
Parkinson's Disease
The NADPARK study, a randomized phase I clinical trial of NR supplementation in Parkinson's disease, demonstrated that 1,000 mg/day NR for 30 days was well-tolerated and increased cerebral NAD+ levels as measured by phosphorus-31 magnetic resonance spectroscopy (Brakedal et al., 2022). While the study was designed primarily to assess safety and pharmacodynamics, secondary analyses suggested trends toward clinical improvement.
The neuroprotective mechanism in PD likely involves SIRT3-mediated mitochondrial protection. Dopaminergic neurons in the substantia nigra are particularly vulnerable to mitochondrial dysfunction and oxidative stress. By restoring NAD+ and activating SIRT3, NAD+ precursor therapy may protect these vulnerable neurons from degeneration.
NR has also shown promise in zebrafish models of PD, improving survival time and motor dysfunction in MPTP-induced models through effects on glucose metabolism and endoplasmic reticulum stress reduction.
Traumatic Brain Injury
Traumatic brain injury (TBI) causes acute NAD+ depletion through massive PARP activation in response to DNA damage and excitotoxicity. This depletion contributes to secondary brain injury, the delayed cascade of inflammation, edema, and cell death that often causes more damage than the initial trauma.
The intranasal route is particularly interesting for TBI applications because it can deliver NAD+ directly to brain tissue without requiring systemic administration, which may be complicated by cardiovascular instability in acute trauma settings. Intranasal NAD+ has shown neuroprotective effects in animal models of ischemic brain injury, and its application to TBI is an active area of investigation.
Screening tools available through FormBlends' free assessment can help identify individuals who may benefit from neuroprotective protocols that include NAD+ therapy alongside complementary peptides.
Long COVID and Post-Viral Neurological Syndromes
Emerging research has linked persistent neurological symptoms following COVID-19 to NAD+ depletion and mitochondrial dysfunction. A randomized controlled trial published in eClinicalMedicine (The Lancet) examined NR supplementation for Long COVID patients and evaluated effects on NAD+ levels, cognition, and symptom recovery.
The rationale is straightforward: viral infection activates innate immune pathways that consume large quantities of NAD+ through CD38 upregulation and PARP activation. In individuals with pre-existing NAD+ decline due to age or metabolic disease, this acute depletion may push NAD+ levels below a critical threshold from which recovery is slow, contributing to persistent symptoms including brain fog, fatigue, and exercise intolerance.
NAD+ in Neuroprotection: Current State
The preclinical evidence for NAD+-mediated neuroprotection is strong and consistent across multiple disease models. Human data are emerging, with the NADPARK study providing proof of concept that oral NR can increase brain NAD+ levels in patients with neurodegenerative disease. However, large-scale efficacy trials demonstrating clinical benefit in human neurological disorders have not yet been completed. Intranasal NAD+ delivery is particularly promising for neurological applications but lacks human pharmacokinetic validation. Visit the Peptide Research Hub for related reports on neuroprotective compounds.
Protocols & Dosing
NAD+ dosing protocols vary considerably across clinical settings, reflecting the absence of standardized guidelines and the evolving nature of the evidence base. The protocols described here represent common clinical practices compiled from published literature, clinical consensus documents, and practitioner experience. They should not be considered prescriptive medical recommendations.
IV NAD+ Protocols
Standard Anti-Aging/Wellness Protocol
| Phase | Dose | Frequency | Duration |
|---|---|---|---|
| Loading | 250-500 mg IV | 2-3x per week | 2-4 weeks |
| Maintenance | 250-500 mg IV | Monthly | Ongoing |
| Boost | 500-750 mg IV | As needed | 1-2 sessions |
Loading protocols typically begin with lower doses (250 mg) to assess tolerance, then increase based on patient response. Each infusion session lasts 2-4 hours depending on dose and infusion rate tolerance. Some clinics start at a rate of 1 mL/min and increase by 0.5 mL/min every 15 minutes as tolerated.
Addiction/Detox Protocol
| Phase | Dose | Frequency | Duration |
|---|---|---|---|
| Intensive detox | 500-1,000 mg IV | Daily | 7-10 days |
| Transition | 250-500 mg IV | 3x per week | 2 weeks |
| Maintenance | 250 mg IV or SubQ transition | Weekly-monthly | 3-6 months |
Addiction protocols use higher daily doses over extended periods. The Brain Restoration Plus (BR+NAD) protocol involves at least 1,500 mg of cumulative NAD+ infused over 7-10 days, beginning with a 250 mg infusion, followed by two 500 mg infusions, and finishing with another 250 mg infusion. Patients are monitored continuously during infusions, and adjunctive therapies (amino acid supplementation, counseling, nutritional support) are integrated throughout.
Neuroprotection/Cognitive Protocol
| Phase | Dose | Frequency | Duration |
|---|---|---|---|
| Initial series | 250-500 mg IV | 2x per week | 4 weeks |
| Maintenance | 250-500 mg IV + nasal adjunct | Biweekly-monthly | Ongoing |
Neuroprotection protocols may combine IV NAD+ with intranasal delivery to maximize CNS exposure. The NAD+ nasal spray is used between IV sessions to maintain brain NAD+ levels.
Subcutaneous NAD+ Protocols
Standard Maintenance Protocol
| Phase | Dose | Frequency | Injection Site |
|---|---|---|---|
| Initiation | 50 mg SubQ | Daily x 3 days | Abdomen, rotating |
| Titration | 100 mg SubQ | Daily x 7 days | Abdomen or thigh |
| Maintenance | 100-200 mg SubQ | Daily or 5x/week | Rotating sites |
SubQ protocols start with lower doses to assess tolerance, as the injection can cause localized stinging or burning. Patients learn to inject slowly (over 30-60 seconds) to reduce discomfort. Ice applied to the injection site before and after can reduce stinging. Injecting at room temperature rather than from refrigeration also improves tolerance.
Hybrid IV + SubQ Protocol
Many clinics now use a hybrid approach: an initial IV loading series to rapidly elevate NAD+ levels, followed by at-home SubQ maintenance injections to sustain the elevation. This combines the acute benefit of IV with the convenience and lower cost of SubQ for long-term therapy.
| Phase | Route | Dose | Frequency | Duration |
|---|---|---|---|---|
| Loading | IV | 500 mg | 2x/week | 2 weeks (4 sessions) |
| Transition | SubQ | 100 mg | Daily | 2 weeks |
| Maintenance | SubQ | 100-200 mg | 3-5x/week | Ongoing |
| Boost | IV | 500 mg | Monthly | Optional |
Oral Precursor Protocols
NR (Nicotinamide Riboside) Protocol
| Phase | Dose | Timing | Duration |
|---|---|---|---|
| Starting | 300 mg/day | Morning with food | 2 weeks |
| Standard | 600-1,000 mg/day | Split AM/PM with food | Ongoing |
| High-dose | 1,000-2,000 mg/day | Split 2-3 doses with food | As directed |
NMN (Nicotinamide Mononucleotide) Protocol
| Phase | Dose | Timing | Duration |
|---|---|---|---|
| Starting | 250 mg/day | Morning, sublingual or oral | 2 weeks |
| Standard | 500-1,000 mg/day | Morning with food | Ongoing |
| High-dose | 1,000-1,500 mg/day | Split AM/PM doses | As directed |
Nasal Spray Protocols
Intranasal NAD+ is typically dosed at 50-100 mg per application, with 1-3 daily administrations. The spray is applied alternating nostrils, with the head tilted slightly forward to direct the spray toward the olfactory region. Protocols vary, but a common approach is:
- Morning dose: 50-100 mg (1-2 sprays per nostril) upon waking
- Optional midday dose: 50 mg for cognitive support
- No evening dosing (may interfere with sleep in some individuals)
Combination Strategies
Increasingly, clinicians are combining multiple NAD+ delivery routes with complementary peptide therapies for comprehensive anti-aging protocols. A typical integrated approach might include:
- NAD+ SubQ injections (100-200 mg, 3-5x/week) for systemic NAD+ elevation
- NAD+ nasal spray (50-100 mg, 1-2x/day) for cognitive/neurological support
- Oral NR or NMN (500-1,000 mg/day) for baseline support
- Epithalon cycling for telomere maintenance
- MOTS-c for mitochondrial optimization
- CJC-1295/Ipamorelin for growth hormone optimization
The FormBlends Dosing Calculator can help determine appropriate starting doses based on individual parameters. Always work with a qualified healthcare provider when designing multi-compound protocols.
Figure 6: Summary of NAD+ dosing protocols across administration routes, showing typical loading and maintenance phases.
Cost Analysis
The cost of NAD+ therapy varies dramatically by administration route, dosing frequency, and clinical setting. Understanding these costs is essential for patients making treatment decisions and clinicians designing sustainable long-term protocols.
IV NAD+ Therapy Costs
| Dose | Typical Cost Per Session | Session Duration | Monthly Cost (Maintenance) |
|---|---|---|---|
| 250 mg | $500-$750 | 2-3 hours | $500-$750 (monthly) |
| 500 mg | $750-$1,200 | 3-4 hours | $750-$1,200 (monthly) |
| 750 mg | $1,000-$1,500 | 4-5 hours | $1,000-$1,500 (monthly) |
| 1,000 mg | $1,200-$2,000 | 4-6 hours | $1,200-$2,000 (monthly) |
IV loading phases can cost $3,000-$8,000 over 2-4 weeks depending on the protocol. Addiction/detox programs involving 7-10 consecutive daily infusions typically run $10,000-$25,000 for the complete initial treatment course.
Costs include nursing time, IV supplies, clinical space, and the NAD+ compound itself. Geographic variation is substantial, with prices in major metropolitan areas (New York, Los Angeles, Miami) running 20-50% higher than smaller markets.
Subcutaneous NAD+ Costs
| Supply | Typical Cost | Supply Duration | Monthly Cost |
|---|---|---|---|
| NAD+ 100 mg/mL, 10 mL vial | $150-$300 | 5-10 injections | $200-$600 |
| NAD+ 200 mg/mL, 10 mL vial | $250-$450 | 10-20 injections | $150-$400 |
| Syringes and supplies | $15-$30 | Monthly supply | $15-$30 |
SubQ NAD+ is substantially more cost-effective than IV, particularly for maintenance therapy. The elimination of clinical supervision time and facility costs reduces the per-dose expense by 70-85%. Most patients sourcing SubQ NAD+ from compounding pharmacies spend $200-$500 per month for daily or near-daily injections.
Nasal Spray Costs
NAD+ nasal spray typically costs $75-$200 per bottle, with each bottle providing 30-60 days of use depending on dosing frequency. Monthly costs range from $75-$200, making this the most affordable non-oral route.
Oral Precursor Costs
| Product | Monthly Cost | Dose |
|---|---|---|
| NR (Niagen/TruNiagen) | $40-$80 | 300-600 mg/day |
| NR (high-dose protocol) | $80-$160 | 1,000-2,000 mg/day |
| NMN (standard) | $50-$120 | 500-1,000 mg/day |
| NMN (pharmaceutical-grade) | $100-$250 | 500-1,000 mg/day |
Oral precursors are the most affordable option by a wide margin, costing $40-$250 per month depending on product quality and dosing. However, lower bioavailability means that a larger fraction of each dose is wasted compared to injectable routes.
Cost-Effectiveness Considerations
The "best value" depends on what you're optimizing for. If the goal is maximum acute NAD+ elevation for a specific clinical purpose (addiction detox, acute recovery), IV is the clear choice despite higher cost. For long-term maintenance and general anti-aging, SubQ injections or oral precursors offer more sustainable economics.
A hybrid approach, using IV loading followed by SubQ or oral maintenance, may offer the best balance of efficacy and cost for most patients. The initial IV investment establishes elevated NAD+ levels rapidly, while the lower-cost maintenance routes sustain them over time.
Insurance Coverage
NAD+ therapy is not covered by insurance in most cases. It is considered an elective, off-label treatment by most insurers. Patients should budget for out-of-pocket expenses and discuss payment plans or package pricing with their clinician. Some clinics offer multi-session packages that reduce the per-session cost by 10-25%.
Safety
The safety profile of NAD+ therapy across all routes is generally favorable based on available data, but the evidence base has significant gaps, particularly regarding long-term use. Understanding both the documented safety data and the limitations of that data is essential for informed decision-making.
IV NAD+ Safety Profile
A systematic review by Radenkovic et al. (2023) evaluating safety and effectiveness of NAD+ across different clinical conditions found that adverse events cataloged by the studies did not present a serious risk to the health of participants. Common side effects of IV infusion are dose- and rate-dependent:
| Side Effect | Frequency | Severity | Management |
|---|---|---|---|
| Nausea | 30-50% | Mild to moderate | Slow infusion rate, antiemetic if needed |
| Chest/abdominal pressure | 20-40% | Mild | Slow infusion rate, usually self-resolving |
| Headache | 15-30% | Mild | Hydration, slow rate |
| Flushing (niacin flush) | 10-25% | Mild | Slow rate; resolves spontaneously |
| Dizziness/lightheadedness | 10-20% | Mild | Slow rate, supine position |
| Muscle cramping | 5-15% | Mild | Electrolyte supplementation |
| Heart palpitations | 5-10% | Mild to moderate | Slow or pause infusion |
Most side effects resolve by slowing the infusion rate or temporarily pausing the infusion. Serious adverse events are rare in published reports. However, keep in mind that cases of significant inflammation marker elevation have been observed in up to 70% of patients in some clinical settings, though the clinical significance of this transient elevation remains unclear.
SubQ NAD+ Safety Profile
Subcutaneous injection side effects are generally milder than IV, primarily because the slower absorption rate avoids the rapid systemic spikes that trigger many IV-related symptoms:
- Injection site reactions: Stinging, burning, and redness at the injection site are common (60-80% of patients) and typically last 5-15 minutes
- Local swelling: Mild swelling or induration at the injection site is occasionally reported and resolves within hours
- Systemic effects: Mild nausea, headache, and flushing can occur but are less frequent and less severe than with IV administration
- Bruising: Minor bruising at injection sites occurs with normal frequency for subcutaneous injections
Oral Precursor Safety Profile
NR and NMN have the most extensive safety data from controlled clinical trials:
NR: A randomized, double-blind, placebo-controlled trial using 1,000 mg/day NR for six weeks reported no significant adverse events. The FDA granted GRAS (Generally Recognized as Safe) status to NR (as Niagen) in 2016. Long-term safety studies extending to 12 months have confirmed the favorable safety profile.
NMN: Clinical trials using 250-900 mg/day have demonstrated no significant adverse effects. A 12-week study at 250 mg/day and separate studies at doses up to 900 mg/day both confirmed absence of serious side effects. NMN has been available as a dietary supplement in many markets, with millions of users providing extensive post-market safety data.
Theoretical Safety Concerns
Several theoretical safety concerns have been raised about long-term NAD+ restoration therapy, though none have been confirmed in clinical studies to date:
- Cancer promotion: Because NAD+ supports cellular energy production and survival signaling, there's a theoretical concern that boosting NAD+ could support the growth of existing cancers. However, the relationship is complex, as SIRT1 activation by NAD+ also has tumor-suppressive effects through p53 deacetylation. Current evidence does not support an increased cancer risk from NAD+ supplementation at standard doses, but patients with active malignancy should consult their oncologist.
- High-dose nicotinamide effects: NAD+ metabolism generates nicotinamide (NAM), which at high concentrations can inhibit sirtuins and PARPs, potentially counteracting some benefits of NAD+ elevation. This creates a theoretical U-shaped dose-response curve where very high doses might be less effective or counterproductive.
- Liver effects: High-dose niacin (nicotinic acid) is known to cause hepatotoxicity, but this effect appears specific to the nicotinic acid form rather than to NAD+, NR, or NMN. Nevertheless, monitoring liver function during high-dose or prolonged NAD+ therapy is prudent.
- Methylation demand: NAD+ metabolism consumes methyl groups (through conversion of NAM to methylated metabolites), potentially increasing demand on the methylation cycle. Patients with MTHFR polymorphisms or existing methylation insufficiency should be aware of this and may benefit from supplemental methyl donors (methylfolate, methylcobalamin, TMG).
Contraindications and Precautions
- Active cancer (theoretical concern; discuss with oncologist)
- Severe renal impairment (reduced clearance of NAD+ metabolites)
- Pregnancy and lactation (insufficient safety data)
- Active acute infection (NAD+ may influence immune cell metabolism in unpredictable ways)
- Concurrent use of medications that affect NAD+ metabolism (discuss with prescribing physician)
- History of severe niacin sensitivity (higher risk of flush reactions)
Blood Work Monitoring
Recommended laboratory monitoring for patients undergoing NAD+ therapy includes:
| Test | Frequency | Rationale |
|---|---|---|
| Complete metabolic panel (CMP) | Baseline, then every 3-6 months | Liver and kidney function monitoring |
| Complete blood count (CBC) | Baseline, then every 6 months | General health monitoring |
| Fasting glucose and HbA1c | Baseline, then every 3-6 months | Metabolic monitoring |
| Lipid panel | Baseline, then every 6 months | NAD+ can influence lipid metabolism |
| Whole blood NAD+ levels | Baseline, 4-8 weeks, then every 3-6 months | Efficacy monitoring |
| Homocysteine | Baseline, then every 3-6 months | Methylation cycle monitoring |
| hs-CRP | Baseline, then every 3-6 months | Inflammation monitoring |
| Uric acid | Baseline, then annually | Purine metabolism monitoring |
NAD+ blood testing is increasingly available through specialty laboratories. Jinfiniti Precision Medicine offers a validated intracellular NAD+ test that provides a more functionally relevant measurement than plasma NAD+ alone. Testing at baseline and 4-8 weeks into therapy helps confirm that the chosen protocol is achieving meaningful NAD+ elevation.
Figure 7: Recommended safety monitoring schedule for patients undergoing NAD+ therapy, including laboratory testing intervals and key parameters.
NMN vs NR vs Direct NAD+: A Detailed Comparison
One of the most common questions in the NAD+ space is whether it's better to supplement with direct NAD+ (via injection or IV) or to take oral precursors like NMN or NR. Each approach has distinct advantages and limitations that make it better suited to different goals and circumstances.
Molecular Differences
NAD+ (Nicotinamide Adenine Dinucleotide): Molecular weight 663.4 g/mol. The full, active coenzyme. Too large and too charged to cross cell membranes or be absorbed orally. Must be delivered parenterally (IV, SubQ, IM) or intranasally for meaningful systemic exposure. Once in the bloodstream, NAD+ is rapidly metabolized by extracellular CD38 and other ectoenzymes.
NMN (Nicotinamide Mononucleotide): Molecular weight 334.2 g/mol. A direct precursor to NAD+ in the salvage pathway. Current evidence suggests that NMN is largely dephosphorylated to NR before cellular uptake, though a dedicated NMN transporter (Slc12a8) has been identified in some tissues. Orally bioavailable, though hepatic first-pass metabolism reduces systemic availability.
NR (Nicotinamide Riboside): Molecular weight 255.2 g/mol. A form of vitamin B3 that enters cells through equilibrative nucleoside transporters. Inside the cell, NR is phosphorylated to NMN by NR kinases, then converted to NAD+ by NMNATs. The smallest of the three molecules and the most readily absorbed orally. Has the most extensive published human clinical trial data.
Head-to-Head Comparison
| Parameter | Direct NAD+ (IV/SubQ) | NMN (Oral) | NR (Oral) |
|---|---|---|---|
| Peak blood NAD+ increase | 210-380% (route-dependent) | 40-80% | 40-142% |
| Time to peak effect | During/shortly after administration | 4-8 hours | 4-8 hours |
| Duration of elevation | 6-24 hours | Sustained with daily dosing | Sustained with daily dosing |
| Route of administration | IV, SubQ, IM, intranasal | Oral, sublingual | Oral |
| Convenience | Low (IV) to moderate (SubQ) | High | High |
| Cost per month | $200-$1,500 | $50-$250 | $40-$160 |
| Published human RCTs | Limited (2-3 pilot studies) | Several (5+) | Extensive (10+) |
| FDA/regulatory status | Compounding pharmacy | Dietary supplement (varies by country) | GRAS, dietary supplement |
| Cellular uptake mechanism | Unclear; may require degradation to NR first | Converted to NR extracellularly, then uptake via ENTs | Direct uptake via ENTs |
| Bypasses NAMPT bottleneck | Yes | Yes | Yes (enters as NR, converted to NMN by NRK) |
| Brain penetration | Limited (BBB); enhanced by nasal route | Crosses BBB after conversion | Crosses BBB after conversion |
When to Choose Each Option
Choose direct NAD+ (IV/SubQ) when:
- Rapid, high-magnitude NAD+ elevation is needed (addiction detox, acute recovery)
- The patient cannot effectively absorb oral supplements (GI conditions, malabsorption)
- Targeting maximum acute effect for specific clinical situations
- Budget allows and the patient prefers clinical-grade delivery
Choose NMN when:
- Long-term daily maintenance is the goal
- The patient prefers oral supplementation
- Cost is a significant consideration
- The patient wants to bypass the NAMPT rate-limiting step
Choose NR when:
- Maximum published evidence is desired (most clinical trial data)
- GRAS status and regulatory confidence are priorities
- Oral convenience is preferred
- The patient responds well to the NR form specifically
Choose a combination when:
- Optimizing across multiple goals (systemic elevation + brain penetration + daily maintenance)
- A comprehensive biohacking protocol is desired
- Initial loading with injectable NAD+ followed by oral maintenance for long-term cost efficiency
The Surprising IV NR Data
A 2024 randomized pilot study comparing IV NAD+ directly with IV NR found that IV NR actually outperformed IV NAD+ for raising whole-blood NAD+ levels. This challenges the intuitive assumption that giving the body the finished product (NAD+) is better than giving it a building block (NR). The finding suggests that cellular uptake and intracellular conversion pathways may be more efficient when working with NR than with exogenous NAD+, which faces rapid extracellular degradation.
This result needs replication in larger studies, but it raises important questions about whether direct NAD+ delivery is truly the optimal approach, or whether providing precursors that cells can efficiently import and convert may be more effective for raising intracellular (not just plasma) NAD+ levels.
For a deeper analysis of NAD+ precursors in the context of longevity peptides, see our NAD+ Longevity Research Report and the MOTS-c Mitochondrial Peptide Report.
Figure 8: Detailed comparison of direct NAD+, NMN, and NR across key parameters including bioavailability, cost, and strength of clinical evidence.
NAD+ and Sleep Quality
One of the most consistently reported subjective benefits of NAD+ therapy is improved sleep quality. Patients across all delivery routes frequently describe falling asleep faster, sleeping more deeply, and waking feeling more refreshed. While controlled clinical data specifically measuring sleep outcomes from NAD+ therapy are limited, the mechanistic connections between NAD+ metabolism and sleep regulation are well-established.
Circadian Regulation of NAD+
As discussed in the metabolism section, NAMPT expression follows a circadian rhythm regulated by the CLOCK/BMAL1 transcription complex. NAD+ levels oscillate with the circadian cycle, peaking during active waking periods and declining during rest. This oscillation is not merely a byproduct of metabolic activity; it actively participates in circadian timekeeping.
SIRT1 deacetylates BMAL1, modulating the amplitude and timing of circadian gene expression. When NAD+ levels are insufficient, SIRT1 activity declines, and the molecular clock becomes less precise. This manifests clinically as the disrupted circadian rhythms, delayed sleep onset, fragmented sleep, and reduced sleep quality that characterize aging.
By restoring NAD+ levels, supplementation may strengthen the molecular clock's amplitude, improving circadian rhythm strength and consequently sleep quality. This effect may explain why many patients report improved sleep as one of the first noticeable benefits of NAD+ therapy, often within the first week of treatment.
NAD+ and Melatonin Synthesis
Melatonin, the primary sleep-promoting hormone, is synthesized from serotonin through a pathway that involves N-acetyltransferase and hydroxyindole O-methyltransferase. The methylation step requires S-adenosylmethionine (SAMe), which is generated through the methionine cycle. NAD+ metabolism generates methylated metabolites (methylnicotinamide, Me2PY, Me4PY) that consume methyl groups from the same methionine/folate cycle.
In theory, high-dose NAD+ supplementation could transiently compete with melatonin synthesis for methyl donors, which may explain why some patients report difficulty sleeping when they take NAD+ supplements too close to bedtime. This observation supports the common clinical recommendation to take NAD+ supplements in the morning and to ensure adequate methyl donor support (methylfolate, methylcobalamin, TMG) alongside NAD+ therapy.
Growth Hormone Secretion and Deep Sleep
Growth hormone (GH) is primarily secreted during deep (slow-wave) sleep. The connection between NAD+, sirtuins, and GH secretion is emerging. SIRT1 has been shown to influence hypothalamic-pituitary signaling pathways, and adequate NAD+ levels may support the hormonal conditions needed for optimal GH release during sleep.
Patients using NAD+ therapy in combination with GH-releasing peptides like CJC-1295/Ipamorelin sometimes report that the addition of NAD+ enhances sleep-related GH secretion effects, though this observation is purely anecdotal and lacks controlled study support.
Practical Sleep Optimization with NAD+
Based on the current understanding of NAD+-sleep interactions, the following practices may optimize sleep benefits from NAD+ therapy:
- Take NAD+ supplements or perform SubQ injections in the morning or early afternoon, not before bed
- If using NAD+ nasal spray, avoid evening dosing unless specifically tolerated
- Support methylation with B vitamins (methylfolate, methylcobalamin) and TMG
- Maintain consistent sleep-wake timing to support circadian NAD+ oscillation
- Limit evening blue light exposure to protect the circadian clock that NAD+ supports
- Track sleep quality using a wearable device to objectively monitor the impact of NAD+ therapy
Athletic Performance and Recovery
NAD+ plays a fundamental role in exercise metabolism, serving as an electron carrier in energy production and as a substrate for sirtuins that regulate mitochondrial biogenesis and metabolic adaptation. However, the translation from basic biochemistry to measurable performance benefits has been more complex than initially expected.
The Exercise-NAD+ Connection
During exercise, the NAD+/NADH ratio shifts dramatically as metabolic flux increases through glycolysis and oxidative phosphorylation. This perturbation in NAD+ availability activates SIRT1 and SIRT3, which in turn promote mitochondrial biogenesis, fatty acid oxidation, and cellular stress resistance. Exercise itself is one of the most potent natural stimulators of the NAD+ salvage pathway through upregulation of NAMPT expression.
The question is whether exogenous NAD+ supplementation can further enhance these exercise-induced adaptations or accelerate recovery between training sessions.
What the Research Shows
The evidence here is surprisingly mixed. A comprehensive review of animal and human NR supplementation studies concluded that NAD+ therapeutics do not appear to alter skeletal muscle metabolism or improve athletic performance in healthy humans. Despite promising data in cell and rodent studies, the potential of various NAD+ therapeutics has yet to translate in humans unless clear NAD+ deficiency is present.
A striking 2025 study published in Cell Metabolism found that skeletal muscle function remains intact despite an 85% reduction in NAD+ content. While mice with severely depleted muscle NAD+ showed increased exercise-induced energetic stress and modest oxidative metabolism alterations, muscle mass, tissue integrity, contractility, and exercise performance were unaffected. This suggests that skeletal muscle may have remarkably resilient mechanisms for maintaining function even with dramatically reduced NAD+ levels.
However, several important caveats apply:
- Most studies used oral NR at moderate doses; injectable NAD+ at higher tissue concentrations may produce different results
- Healthy, young athletes likely have adequate NAD+ levels already, leaving little room for improvement through supplementation
- Older athletes or those with metabolic stress may benefit more, as they're more likely to have clinically meaningful NAD+ deficiency
- Recovery benefits may be more relevant than acute performance enhancement, as PARP-mediated DNA repair and SIRT1-mediated anti-inflammatory signaling are activated during the recovery period
Clinical Observations in Athletes
Despite the equivocal research data, many athletes and their support teams report subjective benefits from NAD+ therapy, including faster perceived recovery, reduced soreness, improved sleep quality (which profoundly affects recovery), and sustained energy levels during training blocks. Whether these effects reflect genuine NAD+-mediated physiological improvements or placebo response is difficult to determine without controlled trials in athletic populations.
For athletes interested in performance and recovery optimization, NAD+ therapy is often combined with growth hormone-stimulating peptides like CJC-1295/Ipamorelin and tissue-repair peptides like BPC-157. The rationale is that NAD+ supports cellular energy and repair at the metabolic level, while GH-releasing peptides and tissue repair peptides work at the systemic and structural levels.
Chronic Fatigue and Energy Applications
Chronic fatigue, whether from chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME), post-viral conditions, or unexplained persistent fatigue, represents one of the most common clinical indications for NAD+ therapy in practice. The rationale centers on mitochondrial dysfunction, which is increasingly recognized as a contributor to fatigue states.
The Mitochondrial Fatigue Hypothesis
If cells cannot produce adequate ATP due to mitochondrial dysfunction, the subjective experience is fatigue, exercise intolerance, and post-exertional malaise. NAD+ is a rate-limiting factor in mitochondrial oxidative phosphorylation, so restoring NAD+ levels could theoretically improve energy production in patients with depleted stores.
CFS/ME patients have been shown to have altered NAD+/NADH ratios, mitochondrial dysfunction, and impaired energy metabolism in multiple studies. These findings provide a biochemical rationale for NAD+ restoration, though the relationship between NAD+ levels and symptom severity has not been definitively established.
Clinical Evidence for Fatigue
A 2023 systematic review found insufficient evidence that IV NAD+ improves fatigue or energy in healthy adults. However, this finding doesn't necessarily apply to patients with documented mitochondrial dysfunction or NAD+ deficiency, who may represent a different clinical population.
Anecdotal clinical reports from NAD+ therapy providers describe significant improvement in fatigue symptoms in a subset of patients, particularly those with post-viral fatigue or documented mitochondrial dysfunction. The response appears to be variable, with some patients reporting dramatic improvement and others experiencing minimal benefit. This variability may reflect differences in underlying pathophysiology. Patients whose fatigue is driven primarily by NAD+ depletion and mitochondrial dysfunction would be expected to respond, while those with fatigue from other causes (hormonal, psychological, structural) may not.
For patients experiencing persistent fatigue, our free assessment tool can help identify whether NAD+ therapy and complementary peptide approaches may be appropriate based on individual symptom profiles.
IV NAD+ Protocol Details: Clinical Implementation
Implementing IV NAD+ therapy requires attention to clinical logistics, patient preparation, infusion management, and post-treatment monitoring that go beyond basic dosing guidelines. This section provides detailed practical information for clinicians and patients navigating the IV NAD+ experience.
Pre-Infusion Preparation
Proper patient preparation significantly impacts the tolerability and effectiveness of IV NAD+ infusions. Patients should arrive well-hydrated, having consumed at least 32-48 ounces of water in the hours before their appointment. Dehydration increases the risk of nausea, headache, and discomfort during infusion. A light meal consumed 1-2 hours before the session helps stabilize blood sugar without causing the GI discomfort that can occur when NAD+ is infused on a completely full stomach.
Pre-treatment vital signs should be documented, including blood pressure, heart rate, respiratory rate, and oxygen saturation. These baseline measurements serve as reference points during the infusion and help identify patients who may need closer monitoring. Patients with resting heart rates above 100 bpm or blood pressure outside normal ranges should be evaluated before proceeding.
First-time patients benefit from a thorough discussion of expected sensations during infusion. The "NAD flush" and feelings of chest tightness or abdominal warmth are common and can be alarming if unexpected. Setting appropriate expectations reduces anxiety and improves the overall treatment experience. Some clinics provide anti-nausea medication (ondansetron 4 mg) prophylactically, though most prefer to manage symptoms reactively through rate adjustment.
Compounding and Preparation
NAD+ for IV infusion is typically sourced from compounding pharmacies as a sterile, pyrogen-free solution. Common concentrations include 100 mg/mL and 200 mg/mL in bacteriostatic normal saline or sterile water. The solution should be clear and colorless; any discoloration or particulate matter warrants discarding the vial.
Storage requirements vary by formulation but generally include refrigeration at 2-8 degrees Celsius with protection from light. Reconstituted solutions should be used within the timeframe specified by the compounding pharmacy, typically 28-90 days depending on preservative content. Multi-dose vials require aseptic technique at each access to prevent contamination.
For infusion preparation, the prescribed dose is drawn from the vial and diluted in 250-500 mL of normal saline (0.9% NaCl). Some protocols use lactated Ringer's solution instead. The diluted solution is administered through a standard IV infusion set with an inline filter. An infusion pump is strongly recommended for precise rate control, as gravity drip can lead to inadvertent rate increases and associated side effects.
Infusion Rate Management
The infusion rate is the single most important factor in determining patient tolerance. Starting rates typically range from 0.5-1.0 mL/min for a 500 mL total volume. The rate is increased by 0.25-0.5 mL/min increments every 15-20 minutes as tolerated. Maximum rates rarely exceed 3-4 mL/min, as faster rates almost invariably produce discomfort.
Clinical experience suggests that patients develop tolerance over multiple sessions. A patient who requires 4 hours for a 500 mg infusion during their first session may tolerate the same dose in 2.5-3 hours by the third or fourth session. This tolerance development should be documented and used to optimize scheduling for subsequent treatments.
Rate-limiting signs that indicate the need to slow or pause infusion include:
- Nausea or dry heaving (most common rate-limiting symptom)
- Chest tightness or sensation of pressure (second most common)
- Abdominal cramping or discomfort
- Headache developing during infusion
- Significant heart rate increase (greater than 20 bpm above baseline)
- Blood pressure changes (greater than 20 mmHg systolic change)
- Skin flushing accompanied by discomfort
- Dizziness or lightheadedness
If symptoms develop, the infusion should be slowed by 50% or paused for 5-10 minutes. Symptoms typically resolve within minutes of rate reduction. If symptoms persist despite rate reduction, consider pausing the infusion entirely for 15-20 minutes before resuming at a lower rate.
Adjunctive Treatments During Infusion
Many clinics add supportive nutrients to the IV bag or administer them as pre-infusion boluses to enhance tolerability and complement NAD+ therapy:
| Adjunct | Dose | Rationale |
|---|---|---|
| Magnesium chloride | 1-2 g IV | Reduces cramping, supports enzymatic reactions |
| Vitamin C | 1-5 g IV | Antioxidant support, may reduce flushing |
| B-complex vitamins | 1 mL IV push | Cofactor support for NAD+ metabolism |
| Glutathione | 200-600 mg IV push (post-NAD+) | Master antioxidant, detoxification support |
| Zinc | 5-10 mg IV | Sirtuin cofactor, immune support |
| Selenium | 200 mcg IV | Antioxidant enzyme support |
Glutathione is typically administered as a separate push after the NAD+ infusion is complete rather than mixed with the NAD+ solution, as compatibility data are limited and some practitioners report that simultaneous administration may reduce the effectiveness of both compounds.
Post-Infusion Care and Response Assessment
After the infusion, patients should remain in the clinical setting for 15-30 minutes of observation. Post-infusion vital signs should be documented and compared to baseline. Most patients feel well immediately after the infusion, though some report mild fatigue or a "spaced out" feeling for 30-60 minutes that transitions to increased alertness and energy.
Common post-infusion experiences reported by patients include:
- Same day: Improved mental clarity, increased energy, enhanced mood (reported by 60-80% of patients after initial sessions)
- Days 1-3: Improved sleep quality, sustained energy improvements, reduced brain fog
- Week 1-2: Cumulative improvements in energy, cognition, and overall well-being with repeated sessions
- Week 2-4: Maximum benefit from loading phase, transition to maintenance appropriate
Patients should be advised to maintain adequate hydration for 24-48 hours after infusion, as NAD+ metabolites are renally excreted and dehydration can concentrate these metabolites. Alcohol consumption should be avoided for at least 24 hours post-infusion, as alcohol metabolism directly competes with NAD+ pathways.
Clinic Setup and Equipment Requirements
Clinics offering IV NAD+ should have the following:
- Comfortable recliners or treatment chairs allowing patients to recline for 2-4+ hours
- IV infusion pumps with precise rate control (gravity drip is suboptimal)
- Vital sign monitoring equipment (BP cuff, pulse oximeter, cardiac monitor for higher-risk patients)
- Emergency supplies including epinephrine, antihistamines, and basic airway management
- Trained nursing staff experienced with IV administration and NAD+ side effect management
- Access to physician consultation for adverse event management
- Appropriate storage facilities for NAD+ solutions (refrigeration, light protection)
- Entertainment options (WiFi, streaming capability, charging stations) for extended infusion times
SubQ NAD+ Injection: Self-Administration Guide
Subcutaneous NAD+ injection has become the preferred maintenance route for many patients who initially receive IV loading. The ability to self-administer at home eliminates the time commitment, travel, and cost of clinical IV sessions while maintaining meaningful NAD+ elevation. This section covers the practical aspects of SubQ NAD+ self-injection.
Patient Training Requirements
Before beginning home SubQ NAD+ injections, patients should receive hands-on training from a qualified healthcare professional covering:
- Proper hand hygiene and aseptic technique
- Vial preparation and dose drawing (including air bubble management)
- Injection site selection and rotation
- Proper injection technique (angle, depth, speed)
- Sharps disposal procedures
- Storage and handling of NAD+ solution
- Recognition of injection site complications (infection, sterile abscess, allergic reaction)
- When to contact their healthcare provider
Most patients become proficient after 2-3 supervised practice injections. Written instructions and video resources should be provided for reference. Patients with needle anxiety may benefit from auto-injector devices, though these are not widely available for NAD+ formulations at current common concentrations.
Injection Technique
The standard SubQ injection technique for NAD+ involves the following steps:
- Wash hands thoroughly with soap and water for at least 20 seconds
- Gather supplies: NAD+ vial, appropriate syringe (typically insulin syringe, 29-31 gauge, 0.5-1.0 mL), alcohol swabs, sharps container
- Allow the NAD+ vial to warm to room temperature if refrigerated (5-10 minutes; injecting cold solution increases discomfort)
- Clean the vial stopper with an alcohol swab and allow to dry
- Draw up the prescribed volume, removing air bubbles by tapping the syringe barrel and pushing small amounts of air out
- Select the injection site (see below) and clean with an alcohol swab in a circular motion from center outward
- Pinch a fold of skin at the injection site
- Insert the needle at a 45-90 degree angle (depending on body composition)
- Inject slowly over 30-60 seconds (faster injection increases stinging)
- Withdraw the needle and apply gentle pressure with a clean gauze pad
- Dispose of the needle immediately in a sharps container
Injection Site Selection and Rotation
Recommended SubQ injection sites for NAD+ include:
| Site | Advantages | Considerations |
|---|---|---|
| Abdomen (2+ inches from navel) | Large area for rotation, easy access, good absorption | Most commonly used; avoid areas with stretch marks or scars |
| Anterior thigh (middle third) | Easy self-access, good tissue volume | May be slightly more painful than abdomen; avoid outer thigh |
| Upper arm (posterior) | Well-absorbed, discreet | May require assistance for injection; limited rotation area |
| Love handles (flank area) | Good tissue volume, less sensitive | Less common; adequate only in patients with sufficient subcutaneous tissue |
Rotation between sites is essential to prevent lipodystrophy (changes in subcutaneous fat distribution) and injection site reactions. A systematic rotation pattern - for example, right abdomen Monday, left abdomen Wednesday, right thigh Friday - helps ensure adequate site rest between injections.
Managing Injection Discomfort
NAD+ solutions cause a stinging or burning sensation at the injection site that typically lasts 5-15 minutes. This is the most common complaint from patients using SubQ NAD+ and the primary reason some patients discontinue this route. Strategies to minimize discomfort include:
- Warm the solution: Room temperature NAD+ stings less than refrigerated solution
- Inject slowly: 30-60 seconds for the full injection volume reduces peak tissue irritation
- Use a smaller gauge needle: 31-gauge needles cause less tissue disruption than 27-gauge
- Apply ice before injection: 2-3 minutes of ice application numbs the skin and reduces pain sensation
- Apply ice after injection: Ice to the site immediately after injection reduces inflammation and stinging
- Consider topical lidocaine: Over-the-counter lidocaine cream applied 30-60 minutes before injection can significantly reduce pain
- Split the dose: Two smaller-volume injections at different sites may be better tolerated than one larger injection
- Distraction: Listening to music or watching a video during and after injection shifts attention from discomfort
Most patients report that injection discomfort decreases over time as they refine their technique and develop a routine. By the second or third week of daily injections, many patients describe the process as routine and minimally bothersome.
Storage and Handling
NAD+ solutions from compounding pharmacies should be stored according to the pharmacy's instructions, typically refrigerated at 2-8 degrees Celsius (36-46 degrees Fahrenheit). The vial should be protected from light and checked for particulate matter or discoloration before each use. Most multi-dose vials have a beyond-use date of 28-90 days after compounding, depending on the formulation and preservative system.
When traveling with NAD+, an insulated travel case with ice packs maintains appropriate temperature. TSA regulations allow prescription medications including injectable solutions through security checkpoints; carrying the pharmacy label and a letter from the prescribing provider is recommended for international travel.
Monitoring Response to SubQ NAD+
Patients self-administering SubQ NAD+ should track their response using both subjective and objective measures:
Subjective tracking:
- Daily energy levels (1-10 scale)
- Sleep quality and duration
- Mental clarity and focus
- Exercise performance and recovery
- Mood and stress resilience
- Any side effects or injection site issues
Objective monitoring:
- Intracellular NAD+ levels at baseline, 4-8 weeks, and every 3-6 months
- Comprehensive metabolic panel every 3-6 months
- Body composition changes (weight, body fat percentage)
- Exercise performance metrics (strength, endurance, recovery time)
- Sleep tracking data (wearable devices)
- Heart rate variability (HRV) trends
NAD+ Nasal Spray: The Brain-Targeted Approach
Of all NAD+ delivery routes, the intranasal approach holds the most intrigue for neuroscience researchers and clinicians focused on brain health. The ability to bypass the blood-brain barrier and deliver NAD+ directly to brain tissue through the olfactory pathway represents a fundamentally different pharmacokinetic strategy than systemic delivery routes.
Nose-to-Brain Delivery Mechanism
The nasal cavity provides two distinct pathways for drug delivery to the brain:
Olfactory Pathway: The olfactory epithelium in the upper nasal cavity contains neurons whose axons project directly through the cribriform plate into the olfactory bulb. Compounds deposited on these neurons can be transported along the olfactory nerve into the CNS, bypassing the blood-brain barrier entirely. This pathway can deliver compounds to the olfactory bulb, hippocampus, and cerebral cortex within minutes of nasal administration.
Trigeminal Pathway: The trigeminal nerve (cranial nerve V) has branches throughout the nasal cavity. Its ophthalmic and maxillary divisions provide a second route for nose-to-brain transport, delivering compounds to the brainstem and other CNS structures through the trigeminal nerve pathway.
Both pathways operate through a combination of intraneuronal transport (molecules traveling within nerve fibers) and perineural transport (molecules traveling along the spaces surrounding nerves). The relative contribution of each mechanism varies by compound and formulation.
Animal Evidence for Intranasal NAD+
The foundational study by Wei et al. (2007) demonstrated that intranasal NAD+ at 10 mg/kg significantly increased brain NAD+ content and reduced infarct volume by approximately 86% in a rat model of transient focal ischemia. This study established proof of concept that intranasal NAD+ reaches brain tissue in pharmacologically meaningful concentrations and exerts biological effects.
Subsequent research in the context of traumatic brain injury screening identified NAD+ and related compounds as candidates for intranasal delivery due to their neuroprotective properties, appropriate molecular characteristics for nasal absorption, and favorable safety profile. The mitochondrial targeting of NAD+ makes it particularly relevant for TBI, where mitochondrial dysfunction drives secondary brain injury.
Translational Considerations
Translating nasal delivery data from rodents to humans requires caution for several reasons. The ratio of olfactory epithelium to total nasal surface area is approximately 50% in rats but only 3-8% in humans. This means that a smaller proportion of nasally administered compound contacts the olfactory region in humans compared to rodents, potentially reducing nose-to-brain delivery efficiency.
However, the absolute surface area of the human nasal cavity is much larger than in rodents, and specialized delivery devices can target spray deposition toward the olfactory cleft. Formulation strategies including mucoadhesive polymers, absorption enhancers, and nanoparticle encapsulation can also improve nasal residence time and epithelial penetration.
Clinical Use of NAD+ Nasal Spray
Despite the limited human evidence, NAD+ nasal spray has gained significant clinical adoption, particularly among practitioners and patients focused on cognitive optimization. The appeal is understandable: non-invasive application, rapid onset of perceived effects (many users report improved mental clarity within 15-30 minutes), and the theoretical advantage of direct brain delivery.
Typical clinical protocols for NAD+ nasal spray include:
| Application | Dose | Frequency | Notes |
|---|---|---|---|
| Cognitive maintenance | 50-100 mg | Once daily, AM | Combine with oral NR/NMN for systemic support |
| Cognitive performance | 50-100 mg | Twice daily, AM and midday | Before demanding cognitive tasks |
| Neuroprotection protocol | 100 mg | Twice daily | Combined with IV or SubQ NAD+ for systemic support |
| Post-concussion support | 50-100 mg | 2-3 times daily | Under medical supervision; start low |
Administration Technique for Optimal Brain Delivery
Proper administration technique can significantly impact the fraction of the spray that reaches the olfactory region versus being swallowed or deposited in the lower nasal passages. Recommended technique includes:
- Clear the nasal passages by gently blowing your nose before application
- Tilt the head slightly forward (not backward, which directs spray toward the throat)
- Insert the spray nozzle just inside the nostril, angling slightly upward and outward
- Breathe in gently through the nose while actuating the spray (avoid forceful sniffing)
- Hold breath for 5-10 seconds after application to allow mucosal contact
- Avoid blowing nose for at least 15-20 minutes after application
- Alternate nostrils with each application to reduce mucosal irritation
Some patients report mild nasal irritation or a bitter taste in the throat after application. These effects are typically transient and diminish with continued use. If significant nasal irritation develops, reducing frequency or dose and ensuring proper technique usually resolves the issue.
Cellular NAD+ Metabolism: Advanced Considerations
For clinicians and researchers seeking a deeper understanding of NAD+ metabolism, several advanced concepts influence how we think about NAD+ therapy design and optimization.
Compartmentalization of NAD+ Pools
NAD+ exists in distinct pools within the cell: nuclear, cytoplasmic, and mitochondrial. These pools are not freely interchangeable, as NAD+ cannot cross the inner mitochondrial membrane. Each compartment maintains its own NAD+ concentration through locally expressed NMNAT isoforms (NMNAT1 in the nucleus, NMNAT2 in the cytoplasm, NMNAT3 in the mitochondria).
This compartmentalization has important therapeutic implications. Raising total cellular NAD+ may not proportionally raise mitochondrial NAD+ if the limiting factor is mitochondrial NMNAT3 activity or NMN transport into the mitochondrial matrix. Similarly, nuclear and cytoplasmic NAD+ pools serve different functions (nuclear NAD+ primarily supports PARPs and SIRT1/6/7; cytoplasmic NAD+ supports SIRT2 and glycolysis; mitochondrial NAD+ supports SIRT3/4/5 and the TCA cycle/electron transport chain).
The practical implication is that different NAD+ precursors may differentially affect specific compartmental pools. NR, for example, appears to be efficiently converted to NAD+ in both the cytoplasm and nucleus through NMNAT1 and NMNAT2, but its impact on mitochondrial NAD+ depends on NMN or NAD+ transport mechanisms that remain under investigation.
The NAD+/NADH Ratio
The ratio of oxidized NAD+ to reduced NADH is a critical metabolic parameter that influences cellular function at multiple levels. This ratio reflects the overall redox state of the cell and directly impacts the activity of NAD+-dependent enzymes.
In healthy, well-oxygenated cells, the cytoplasmic NAD+/NADH ratio is approximately 700:1 in favor of NAD+, while the mitochondrial ratio is approximately 7-8:1. These ratios are maintained by the malate-aspartate shuttle and glycerol-3-phosphate shuttle, which transfer reducing equivalents between compartments.
Conditions that shift the ratio toward NADH (a more reduced state) include hypoxia, alcohol metabolism, high-fat diets, and aging. This shift toward reduction impairs sirtuin activity (which requires NAD+ as a substrate) and can contribute to metabolic dysfunction. NAD+ supplementation may help restore the ratio toward the oxidized state, reactivating sirtuin-dependent pathways.
NAMPT Regulation and the Circadian Clock
NAMPT, the rate-limiting enzyme in the NAD+ salvage pathway, is under circadian regulation through the CLOCK/BMAL1 transcription factor complex. This means that NAD+ synthesis fluctuates throughout the day, with higher production during active periods and lower production during rest. SIRT1 participates in this regulation by deacetylating BMAL1, creating a feedback loop that connects NAD+ metabolism to circadian biology.
The circadian regulation of NAD+ has several practical implications for therapy design:
- Morning dosing of NAD+ or precursors may align with the natural peak in NAMPT activity, potentially enhancing conversion efficiency
- Evening dosing could theoretically interfere with the natural circadian decline in NAD+ that may play a role in sleep initiation
- Shift workers and individuals with disrupted circadian rhythms may have dysregulated NAMPT expression, potentially increasing their need for NAD+ supplementation
- The interaction between NAD+ therapy and circadian biology suggests that timing of administration may matter as much as dose
NAD+ and the Epigenome
NAD+-dependent enzymes, particularly sirtuins, are major regulators of the epigenome. SIRT1 deacetylates histones (H3K9, H4K16), promoting chromatin compaction and gene silencing at specific loci. SIRT6 deacetylates H3K9 and H3K56, maintaining heterochromatin and preventing aberrant gene expression associated with aging.
As NAD+ declines with age and sirtuin activity decreases, epigenetic marks accumulate that progressively alter gene expression patterns toward a pro-inflammatory, pro-senescent profile. This "epigenetic drift" is now recognized as a hallmark of aging and may be partially reversible through NAD+ restoration.
The connection between NAD+ and the epigenome suggests that NAD+ therapy may have effects that persist beyond the period of active treatment, particularly if sustained NAD+ elevation allows sirtuins to remodel chromatin toward more youthful patterns. This is an active area of investigation with significant implications for longevity medicine.
SARM1 and NAD+ in Axonal Degeneration
SARM1 (sterile alpha and TIR motif-containing protein 1) is a NADase that plays a critical role in axonal degeneration. When neurons are injured, SARM1 activation rapidly depletes axonal NAD+, triggering Wallerian degeneration - the programmed self-destruction of the damaged axon. This process is distinct from apoptosis and serves an important physiological role in clearing damaged neural connections.
However, in neurodegenerative diseases, inappropriate SARM1 activation can cause premature axonal loss. Maintaining adequate NAD+ levels may delay SARM1-mediated degeneration by keeping the NAD+ supply above the threshold at which SARM1 becomes active. This mechanism provides additional rationale for NAD+ therapy in neurodegenerative conditions and after traumatic brain or nerve injury.
CD38 Inhibition as a Complementary Strategy
Rather than simply adding more NAD+ to overwhelm CD38-mediated consumption, an alternative strategy is to inhibit CD38 activity directly. Several natural and synthetic CD38 inhibitors have been identified:
- Apigenin: A flavonoid found in parsley, celery, and chamomile tea that inhibits CD38 in vitro. Doses used in research are substantially higher than typical dietary intake.
- Quercetin: Another flavonoid with CD38 inhibitory properties, found in onions, apples, and berries. Also has senolytic properties that may reduce the senescent cell burden driving CD38 upregulation.
- Luteolinidin: A flavonoid derivative showing potent CD38 inhibition in preclinical models.
- 78c: A synthetic thiazoloquin(az)olin(on)e compound that reverses age-related NAD+ decline in mice through specific CD38 inhibition (Tarragó et al., 2018, Cell Metabolism).
Combining NAD+ supplementation with CD38 inhibition addresses both sides of the equation: increasing NAD+ supply while reducing its destruction. Some practitioners recommend quercetin or apigenin supplementation alongside NAD+ therapy for this reason, though clinical evidence for this combination strategy in humans is limited to theoretical rationale and preclinical data.
Clinical Response Patterns and Patient Selection
While controlled clinical trial data for NAD+ therapy remain limited, decades of clinical use have generated consistent patterns in patient response that can guide treatment decisions. These observations, drawn from published case series and clinical practice reports, help identify which patients are most likely to benefit from different NAD+ delivery approaches.
Patient Populations with Strongest Clinical Response
Middle-aged adults (40-65) with fatigue and cognitive decline: This population typically shows the most dramatic response to NAD+ therapy, likely because they have experienced significant NAD+ decline but have not yet developed irreversible end-organ damage. Clinical improvements in energy, mental clarity, and exercise tolerance are commonly reported within the first 1-2 weeks of treatment. This is the "sweet spot" population where the gap between current NAD+ levels and physiological need is largest.
Post-viral fatigue (including Long COVID): Patients with persistent fatigue, brain fog, and exercise intolerance following viral infections represent an emerging indication for NAD+ therapy. The rationale is strong: viral infection activates innate immune pathways that consume NAD+ through CD38 and PARP activation. Restoring depleted NAD+ stores may accelerate recovery of mitochondrial function and reduce persistent neuroinflammation.
Chronic alcohol use: Patients with a history of heavy alcohol consumption often have the most profoundly depleted NAD+ levels due to the direct consumption of NAD+ in alcohol metabolism. Restoration of NAD+ in this population may improve hepatic function, cognitive clarity, and energy levels while supporting sustained sobriety.
Athletes and high-performers over 35: While NAD+ supplementation may not improve performance in healthy young athletes, individuals over 35 who notice declining recovery capacity, reduced training tolerance, or plateaued performance may benefit from NAD+ restoration. The effect is likely mediated through improved mitochondrial function and enhanced cellular repair during recovery periods.
Patient Populations with More Variable Response
Young, healthy adults: Individuals under 35 with no specific health complaints typically have relatively preserved NAD+ levels and may notice minimal benefit from supplementation. The ceiling effect limits improvement when baseline levels are already adequate. This population may derive more benefit from lifestyle optimization (exercise, sleep, diet) than from exogenous NAD+.
Elderly patients (75+) with advanced disease: While NAD+ restoration is theoretically beneficial at any age, patients with advanced organ damage or severe comorbidities may not respond as effectively because downstream targets of NAD+ signaling may be too impaired to respond even when NAD+ levels are restored. Treatment in this population should be approached with appropriate expectations.
Patients with primary psychiatric disorders: Depression, anxiety, and cognitive complaints without underlying metabolic or mitochondrial dysfunction are unlikely to respond specifically to NAD+ therapy, though the general improvements in energy and well-being may have indirect benefits.
Predictors of Treatment Response
Clinical experience suggests several factors that predict stronger response to NAD+ therapy:
- Low baseline intracellular NAD+ levels: Patients with documented NAD+ depletion (measurable through specialized testing) tend to respond more dramatically than those with levels in the normal range
- Symptoms consistent with mitochondrial dysfunction: Fatigue, exercise intolerance, brain fog, cold intolerance, and post-exertional malaise are all consistent with mitochondrial energy deficit
- History of NAD+-depleting exposures: Chronic alcohol use, viral infection, chemotherapy, chronic stress, or poor sleep increase the likelihood of significant NAD+ depletion
- Age 40-65: This age range represents the steepest phase of natural NAD+ decline, where intervention may have the greatest impact
- Absence of confounding conditions: Patients whose symptoms are primarily attributable to NAD+ depletion (rather than other causes) show the clearest response
Non-Responders and Troubleshooting
An estimated 15-25% of patients report minimal benefit from NAD+ therapy. Common reasons for non-response include:
- Symptoms driven by causes other than NAD+ depletion (hormonal imbalance, sleep apnea, autoimmune disease, undiagnosed conditions)
- Insufficient dose or duration of treatment (some patients require 4-6 weeks before noticing benefits)
- Ongoing NAD+-depleting behaviors (heavy alcohol use, severe sleep deprivation, extreme physiological stress) that outpace supplementation
- Genetic variants affecting NAD+ metabolism enzymes (NAMPT, NRK, NMNAT polymorphisms) that may reduce conversion efficiency
- Severely impaired downstream targets (sirtuins, PARPs) that cannot respond even with restored NAD+ levels
For non-responders, the following adjustments may be considered: switching delivery routes (from oral to injectable, or adding nasal spray), increasing dose, extending treatment duration, adding CD38 inhibitors (quercetin, apigenin), addressing confounding conditions, and ensuring adequate cofactor support (B vitamins, magnesium, zinc).
NAD+ and Peptide Therapy: Combination Approaches
NAD+ restoration does not operate in isolation. It intersects with multiple longevity and regenerative pathways that are targets of peptide therapy. Understanding these intersections allows for the design of rational combination protocols that may produce additive benefits greater than either compound alone.
NAD+ and Epithalon: Telomere-NAD+ Crosstalk
Epithalon (epitalon, epithalamin) is a tetrapeptide that activates telomerase, the enzyme responsible for maintaining telomere length. The connection to NAD+ is through SIRT1 and SIRT6, which both play roles in telomere maintenance and chromatin stability at telomeric regions.
SIRT6 deacetylates histone H3K9 at telomeric chromatin, maintaining the heterochromatic state that protects telomere integrity. When NAD+ levels decline and SIRT6 activity falls, telomeric chromatin becomes more open (euchromatic), leading to telomere instability and increased DNA damage at chromosome ends. By restoring NAD+ levels and reactivating SIRT6, NAD+ therapy may complement Epithalon's direct telomerase activation with improved telomere chromatin maintenance.
Our comprehensive Epithalon report covers telomere biology and Epithalon protocols in detail.
NAD+ and MOTS-c: Dual Mitochondrial Support
MOTS-c is a mitochondrial-derived peptide (MDP) encoded by the mitochondrial genome's 12S rRNA gene. It activates AMPK, enhances glucose metabolism, improves insulin sensitivity, and promotes mitochondrial biogenesis through pathways that overlap with but are distinct from NAD+-sirtuin signaling.
The complementarity between NAD+ and MOTS-c operates at multiple levels:
- Energy sensing: NAD+ activates SIRT1, which activates AMPK; MOTS-c directly activates AMPK. Both pathways converge on PGC-1alpha to promote mitochondrial biogenesis.
- Mitochondrial quality control: NAD+/SIRT3 promotes mitochondrial deacetylation and stress resistance; MOTS-c promotes mitochondrial biogenesis and metabolic flexibility.
- Metabolic regulation: NAD+/SIRT1 enhances fatty acid oxidation and insulin signaling; MOTS-c improves glucose uptake and metabolic homeostasis.
- Anti-inflammatory effects: NAD+/SIRT1 suppresses NF-kB; MOTS-c reduces inflammatory cytokine production through AMPK-dependent mechanisms.
This dual approach to mitochondrial support may be particularly valuable for patients with significant mitochondrial dysfunction, such as those with age-related fatigue, metabolic syndrome, or post-viral conditions. See the MOTS-c research report for detailed protocol information.
NAD+ and GHK-Cu: Regenerative Combination
The copper peptide GHK-Cu promotes tissue repair, collagen synthesis, and wound healing through upregulation of growth factors and extracellular matrix remodeling. The connection with NAD+ operates through the cellular energy supply: tissue repair is an energy-intensive process that requires strong mitochondrial function. By ensuring adequate cellular energy production through NAD+ restoration, the repair signals generated by GHK-Cu can be more effectively executed.
Additionally, SIRT1 activation by NAD+ promotes the expression of antioxidant enzymes (SOD2, catalase) that protect newly synthesized collagen and tissue from oxidative damage. This creates conditions under which GHK-Cu's regenerative signals produce more durable results.
NAD+ and BPC-157: Inflammatory Resolution
BPC-157 (Body Protection Compound-157) is a pentadecapeptide with broad tissue-protective and healing properties. It promotes angiogenesis, modulates growth factor expression, and accelerates the resolution of inflammatory processes. Combining it with NAD+ addresses inflammation from both directions: NAD+/SIRT1 suppresses the initiation of inflammatory signaling through NF-kB deacetylation, while BPC-157 promotes the resolution and repair of existing inflammatory damage.
This combination may be particularly relevant for patients with chronic inflammatory conditions, post-surgical recovery, or connective tissue injuries where both metabolic support and tissue repair are needed.
NAD+ and CJC-1295/Ipamorelin: Growth Hormone Optimization
Growth hormone (GH) and NAD+ pathways interact through SIRT1, which influences GH secretion and IGF-1 signaling. CJC-1295/Ipamorelin stimulates pulsatile GH release from the pituitary, promoting lean body composition, tissue repair, and recovery. The combination with NAD+ may enhance the downstream effects of GH by ensuring that cells have adequate metabolic capacity to respond to GH/IGF-1 signaling.
Some practitioners report that patients who add NAD+ therapy to an existing GH peptide protocol experience enhanced body composition changes, improved recovery from training, and better sleep quality compared to GH peptides alone. While this observation lacks controlled trial support, the mechanistic rationale is sound.
Designing Multi-Peptide Protocols
When combining NAD+ with multiple peptides, a sequential introduction approach is recommended. Start with one compound, establish baseline response over 2-4 weeks, then add the next compound. This allows identification of individual effects and any unexpected interactions. A typical phased introduction might look like:
| Phase | Week | Compound(s) | Monitoring |
|---|---|---|---|
| 1 | Weeks 1-4 | NAD+ (SubQ or oral precursor) | Energy, sleep, cognition, baseline labs |
| 2 | Weeks 5-8 | Add MOTS-c or Epithalon | Exercise tolerance, metabolic markers |
| 3 | Weeks 9-12 | Add CJC/Ipamorelin or GHK-Cu | Body composition, recovery, skin quality |
| 4 | Week 12+ | Full protocol maintenance | Comprehensive labs, adjust as needed |
Visit the Peptide Research Hub for detailed information on each peptide mentioned above, and use the FormBlends Dosing Calculator for individualized dosing guidance. A free assessment can help determine which combination approach may be most appropriate for your specific goals.
Figure 9: Pathway convergence between NAD+ signaling and complementary peptide therapies, showing how multiple compounds target overlapping and distinct mechanisms for comprehensive anti-aging support.
NAD+ and Cardiovascular Health
Cardiovascular disease remains the leading cause of death globally, and NAD+ depletion has emerged as a significant contributor to age-related cardiovascular decline. The heart is one of the most metabolically active organs in the body, beating approximately 100,000 times per day and requiring a constant supply of ATP generated primarily through mitochondrial oxidative phosphorylation. This extreme metabolic demand makes cardiac tissue particularly vulnerable to NAD+ deficiency.
NAD+ in Cardiac Energy Metabolism
The adult heart derives approximately 70% of its ATP from fatty acid oxidation and 20-30% from glucose oxidation, both of which require NAD+ as an essential cofactor. The cardiac mitochondrial NAD+/NADH ratio directly influences the efficiency of the electron transport chain and the rate of ATP production. When cardiac NAD+ levels decline, the heart's ability to generate ATP becomes compromised, potentially contributing to diastolic dysfunction, reduced exercise capacity, and heart failure with preserved ejection fraction (HFpEF).
SIRT3, the primary mitochondrial sirtuin, plays an essential role in cardiac metabolism by deacetylating and activating key enzymes in fatty acid oxidation, the tricarboxylic acid (TCA) cycle, and the electron transport chain. In aged hearts, declining NAD+ levels lead to reduced SIRT3 activity, hyperacetylation of mitochondrial proteins, and impaired energy production. This creates a vicious cycle where reduced energy production leads to cellular stress, which further depletes NAD+ through PARP activation.
Vascular Function and Arterial Stiffness
One of the most promising clinical findings for NAD+ therapy comes from cardiovascular research. Martens et al. (2018) demonstrated that chronic NR supplementation tended to reduce systolic blood pressure and aortic stiffness in healthy middle-aged and older adults. Aortic pulse wave velocity, a gold-standard measure of arterial stiffness and a strong independent predictor of cardiovascular events, showed improvement with NR treatment.
The mechanism involves SIRT1-mediated activation of endothelial nitric oxide synthase (eNOS), which produces nitric oxide (NO), the primary endogenous vasodilator. When NAD+ levels are sufficient, SIRT1 deacetylates eNOS at lysine residues 496 and 506, increasing its enzymatic activity and enhancing NO production. This improves endothelium-dependent vasodilation and reduces arterial stiffness.
Additionally, NAD+/SIRT1 signaling suppresses the expression of adhesion molecules (VCAM-1, ICAM-1) on endothelial cells, reducing leukocyte adhesion and the initiation of atherosclerotic plaque formation. SIRT1 also promotes cholesterol efflux from macrophages through upregulation of the liver X receptor (LXR) pathway, potentially slowing atherosclerotic progression.
Heart Failure and Cardiac Remodeling
Preclinical research has demonstrated that NAD+ supplementation can prevent or reverse pathological cardiac remodeling in animal models of heart failure. Diguet et al. (2018) showed that NR supplementation in mice prevented the development of heart failure following pressure overload by maintaining mitochondrial function and reducing protein acetylation in cardiac tissue.
NAD+ restoration in failing hearts improves calcium handling, reduces fibrosis, and normalizes the expression of genes involved in cardiac contraction. These effects are mediated primarily through SIRT1 and SIRT3, which regulate mitochondrial quality control (through mitophagy and biogenesis) and protect against oxidative stress.
While clinical trials of NAD+ precursors specifically for heart failure patients have not been completed, the preclinical evidence is compelling enough to support ongoing investigation. Patients with cardiovascular disease or significant cardiovascular risk factors may represent a population with particular potential to benefit from NAD+ restoration, though any therapy should be coordinated with their cardiologist.
Ischemia-Reperfusion Injury
When blood flow is temporarily interrupted and then restored (as occurs during heart attack, stroke, or cardiac surgery), the restoration of blood flow paradoxically causes additional tissue damage known as ischemia-reperfusion (I/R) injury. This injury involves massive PARP activation in response to DNA damage from oxidative stress, which rapidly depletes NAD+ and triggers cell death.
NAD+ pretreatment or rapid post-ischemic NAD+ restoration has shown significant cardioprotective and neuroprotective effects in animal models. By maintaining the NAD+ pool during and after ischemic events, cells can sustain PARP-mediated DNA repair without depleting NAD+ to lethal levels. This finding has implications for surgical settings where planned ischemia occurs (cardiac bypass, organ transplantation) and for acute stroke management.
NAD+ and Cardiac Rhythm
Emerging evidence suggests that NAD+ levels influence cardiac electrophysiology. SIRT1 regulates the expression of sodium channel genes and potassium channel genes that determine cardiac action potential duration and conduction velocity. NAD+ decline may contribute to age-related changes in cardiac rhythm, including the increased susceptibility to atrial fibrillation seen in older adults.
Animal studies have shown that NAD+ supplementation reduces the incidence of arrhythmias following ischemia-reperfusion injury by maintaining ion channel expression and reducing oxidative damage to the conduction system. Clinical investigation of NAD+ for arrhythmia prevention is in early stages.
NAD+ and Cardiac Stem Cell Function
The heart contains resident cardiac progenitor cells that contribute to tissue homeostasis and limited regenerative capacity. These cells require adequate NAD+ for proliferation, differentiation, and survival. Age-related NAD+ decline may impair cardiac progenitor cell function, reducing the heart's capacity for self-repair and contributing to the progressive loss of cardiomyocytes that characterizes cardiac aging.
Preclinical evidence suggests that NAD+ supplementation can improve cardiac progenitor cell function in aged hearts, potentially enhancing the heart's intrinsic repair capacity. While the clinical significance of this finding for human cardiac regeneration remains to be established, it adds another mechanism through which NAD+ may support cardiovascular health during aging.
Practical Cardiovascular Monitoring During NAD+ Therapy
Patients with cardiovascular disease or risk factors who undergo NAD+ therapy should have the following cardiovascular monitoring integrated into their treatment plan:
- Blood pressure measurement at each clinical visit and home monitoring between visits
- Heart rate and rhythm assessment, including consideration of ambulatory ECG monitoring for patients with arrhythmia risk factors
- Lipid panel at baseline and every 6 months (NAD+ may influence lipid metabolism through SIRT1/SIRT3 pathways)
- hs-CRP and other inflammatory markers to track cardiovascular inflammation
- Echocardiography at baseline for patients with known cardiac disease, with follow-up as clinically indicated
- Exercise stress testing for patients whose primary goal is improved exercise capacity
- Pulse wave velocity or other arterial stiffness measurement if available (most sensitive to NAD+ effects based on published data)
During IV infusions, patients with cardiovascular disease should have continuous cardiac monitoring (at minimum, pulse oximetry and periodic blood pressure checks) and infusion rates should be advanced more conservatively than in healthy patients.
NAD+ and Metabolic Syndrome: Insulin Resistance, Obesity, and Fatty Liver
Metabolic syndrome, the constellation of insulin resistance, central obesity, dyslipidemia, and hypertension, affects approximately 35% of American adults and is a major driver of cardiovascular disease, type 2 diabetes, and non-alcoholic fatty liver disease (NAFLD). NAD+ depletion both results from and contributes to metabolic dysfunction, creating a feed-forward cycle that accelerates disease progression.
NAD+ and Insulin Sensitivity
The relationship between NAD+ and insulin sensitivity operates through multiple mechanisms. SIRT1 directly modulates insulin signaling by deacetylating insulin receptor substrate-2 (IRS-2) in hepatocytes, enhancing insulin sensitivity. In skeletal muscle, SIRT1 activation of PGC-1alpha promotes mitochondrial biogenesis and fatty acid oxidation, improving metabolic flexibility and glucose disposal.
The clinical trial by Yoshino et al. (2021) in Science provided the first rigorous human evidence that NAD+ precursor supplementation improves insulin sensitivity. Postmenopausal women with prediabetes who received 250 mg/day NMN for 10 weeks showed enhanced muscle insulin-stimulated glucose disposal compared to placebo. The effect was mediated through improvements in muscle insulin signaling, platelet-derived growth factor receptor signaling, and remodeling pathways.
This finding is particularly significant because it demonstrates a direct metabolic benefit from NAD+ precursor supplementation in a population at high risk for type 2 diabetes. If confirmed in larger trials, NMN or NR supplementation could become an adjunctive strategy for diabetes prevention alongside lifestyle modification.
NAD+ and Obesity
NAD+/SIRT1 signaling regulates adipogenesis (fat cell formation), lipolysis (fat breakdown), and adipokine secretion (hormones produced by fat tissue). In white adipose tissue, SIRT1 represses PPARgamma, the master regulator of adipogenesis, helping to restrain fat cell expansion. SIRT1 also promotes browning of white adipose tissue, increasing energy expenditure through uncoupled respiration.
Chronic overnutrition and obesity create a state of relative NAD+ depletion in adipose tissue through inflammatory signaling (TNF-alpha, IL-6) that upregulates CD38 and PARP activity. This creates a metabolic environment that favors continued fat storage and resists weight loss, even with caloric restriction.
Remie et al. (2020) conducted a randomized controlled trial of NR supplementation in healthy obese humans and found that NR altered body composition and skeletal muscle acetylcarnitine concentrations, suggesting changes in fatty acid metabolism. While the study was small and the primary outcomes were metabolic rather than weight-related, the findings support the concept that NAD+ modulates metabolic pathways relevant to obesity.
NAD+ therapy is not a weight loss treatment per se, but restoring NAD+ levels in metabolically compromised patients may improve the metabolic flexibility needed for effective weight management and exercise response.
NAD+ and Non-Alcoholic Fatty Liver Disease
NAFLD is characterized by excessive hepatic triglyceride accumulation in the absence of significant alcohol consumption. The progression from simple steatosis to non-alcoholic steatohepatitis (NASH) involves mitochondrial dysfunction, oxidative stress, and inflammatory signaling, all of which are influenced by NAD+ levels.
In the liver, SIRT1 and SIRT3 regulate fatty acid oxidation, ketogenesis, and gluconeogenesis. When hepatic NAD+ levels decline, these metabolic programs become impaired, favoring triglyceride accumulation and lipotoxicity. SIRT1 activation through NAD+ restoration promotes fatty acid oxidation through PGC-1alpha and reduces lipogenesis through SREBP-1c deacetylation.
Animal studies have consistently shown that NAD+ precursor supplementation reduces hepatic steatosis and improves liver function markers in models of NAFLD. The effect is particularly pronounced when combined with exercise, which independently activates NAMPT and promotes hepatic NAD+ synthesis.
Patients with NAFLD or NASH may represent a high-priority population for NAD+ therapy, as their hepatic NAD+ levels are likely depleted and the downstream consequences of this depletion directly contribute to disease progression. However, liver function monitoring is important during therapy, as the liver is a primary site of NAD+ metabolism and high-dose supplementation increases hepatic metabolic demand.
The Metabolic Syndrome Treatment Integration
NAD+ therapy for metabolic syndrome works best as part of an integrated approach:
- Exercise: The most potent natural activator of NAMPT and the NAD+ salvage pathway. Exercise and NAD+ supplementation may have compounding effects on mitochondrial biogenesis and metabolic health.
- Caloric restriction or time-restricted eating: Fasting states increase the NAD+/NADH ratio and activate SIRT1, complementing exogenous NAD+ supplementation.
- NAD+ supplementation: Oral NR or NMN at 500-1,000 mg/day, or SubQ NAD+ for more rapid effect, to restore depleted NAD+ pools.
- CD38 inhibition: Quercetin, fisetin, or apigenin supplementation may reduce CD38-mediated NAD+ consumption in metabolically inflamed tissues.
- Methyl donor support: Methylfolate, methylcobalamin, and TMG to support the methylation demands of NAD+ metabolism.
NAD+ and Immune Function
The role of NAD+ in immune cell function adds another dimension to our understanding of NAD+ therapy. Immune cells are highly metabolically active, particularly during activation, and their function is profoundly influenced by NAD+ availability.
Innate Immunity and NAD+
Macrophages, the primary effectors of innate immunity, undergo dramatic metabolic reprogramming upon activation. M1-polarized (pro-inflammatory) macrophages shift from oxidative phosphorylation to aerobic glycolysis, a process that requires NAD+ for glycolytic enzymes. Simultaneously, activated macrophages upregulate CD38, which consumes large quantities of NAD+.
This creates a paradox in aging: the CD38+ macrophages that drive tissue NAD+ depletion are themselves dependent on NAD+ for their inflammatory function. Restoring NAD+ levels may modulate macrophage polarization, potentially shifting the balance from pro-inflammatory M1 toward anti-inflammatory M2 phenotypes through SIRT1-mediated signaling.
SIRT1 deacetylates and inactivates the RelA/p65 subunit of NF-kB in macrophages, suppressing pro-inflammatory cytokine production. By restoring NAD+ and enhancing SIRT1 activity, NAD+ therapy may dampen the chronic, low-grade inflammation (inflammaging) that characterizes immune aging without compromising acute immune responses when they're needed.
T Cell Function and NAD+
T cells require metabolic reprogramming upon activation, shifting from oxidative metabolism in their quiescent state to a combination of glycolysis and oxidative phosphorylation during effector function. NAD+ availability influences this metabolic switch and consequently affects T cell proliferation, cytokine production, and differentiation.
SIRT1 promotes the differentiation of regulatory T cells (Tregs) while suppressing pro-inflammatory Th17 cells. This balance is crucial for immune homeostasis, and its disruption contributes to autoimmune disease and chronic inflammation. NAD+ restoration may help restore the Treg/Th17 balance in aging individuals, though clinical evidence for this application is limited to preclinical studies.
CD38 is also expressed on T cells, where it serves as an activation marker and contributes to calcium signaling. The relationship between CD38 on T cells and NAD+ metabolism is complex, as CD38-mediated NAD+ consumption may serve important regulatory functions during T cell activation that should not be indiscriminately suppressed.
Immunosenescence and NAD+
Age-related decline in immune function (immunosenescence) is characterized by reduced T cell diversity, impaired vaccine responses, increased susceptibility to infections, and chronic low-grade inflammation. These changes correlate temporally with NAD+ decline and may be partially driven by it.
The senescence-associated secretory phenotype (SASP) of aged and damaged cells promotes CD38 upregulation on tissue-resident macrophages, depleting local NAD+ stores and impairing immune cell function. This creates a self-reinforcing cycle where immune aging drives metabolic decline and metabolic decline accelerates immune aging.
NAD+ restoration in the context of immunosenescence may improve vaccine responses in older adults, reduce susceptibility to infections, dampen chronic inflammation without compromising acute immune defenses, and improve recovery from acute illness. These potential benefits are particularly relevant in the post-COVID era, where immune function optimization has gained widespread interest.
Practical Implications for Immune Health
For patients seeking to optimize immune function, NAD+ therapy can be combined with other evidence-based immune support strategies:
- Regular moderate exercise (enhances NAMPT expression and immune cell function)
- Adequate sleep (circadian NAD+ regulation intersects with immune rhythm regulation)
- Vitamin D optimization (vitamin D receptor signaling intersects with SIRT1 pathways)
- Zinc supplementation (zinc is a cofactor for over 300 enzymes, including several involved in NAD+ metabolism)
- Stress management (chronic stress depletes NAD+ through cortisol-mediated PARP activation and HPA axis dysregulation)
The intersection of NAD+ metabolism with immune function represents an active research area where future discoveries may yield significant clinical applications, particularly for aging populations and individuals with immune-mediated diseases.
NAD+ and Autoimmune Conditions
The balance between immune activation and immune tolerance is central to autoimmune disease. NAD+/SIRT1 signaling promotes regulatory T cell (Treg) differentiation and function, which suppresses inappropriate immune responses against self-tissues. When NAD+ levels decline and SIRT1 activity falls, the Treg/Th17 balance may shift toward pro-inflammatory Th17 cells, potentially contributing to autoimmune flares.
Preclinical models of autoimmune diseases including experimental autoimmune encephalomyelitis (EAE, a model for multiple sclerosis), collagen-induced arthritis (a model for rheumatoid arthritis), and experimental autoimmune hepatitis have shown improvement with NAD+ precursor supplementation. The mechanism involves SIRT1-mediated enhancement of Treg function, suppression of Th17 differentiation, and reduction of pro-inflammatory cytokine production.
Clinical application of NAD+ therapy for autoimmune conditions is in its infancy. Some functional medicine practitioners report improvement in autoimmune markers and symptoms with NAD+ supplementation as part of comprehensive autoimmune protocols, but controlled clinical data are not available. Patients with autoimmune diseases should discuss NAD+ therapy with their treating physician, as the effects on immune modulation could theoretically affect disease activity in either direction.
NAD+ and Infection Response
During acute infection, the innate immune system requires a rapid metabolic shift to mount an effective response. Macrophages and neutrophils dramatically increase glycolysis and NADPH oxidase activity, both of which consume NAD+ or its derivatives. This acute NAD+ consumption is physiologically appropriate and necessary for pathogen clearance.
However, in patients with pre-existing NAD+ depletion (older adults, those with chronic disease), the acute immune response may be compromised because the NAD+ reserve is insufficient to support the metabolic demands of immune activation. This may partly explain the increased susceptibility to infection and reduced vaccine responses seen in older adults.
Maintaining adequate NAD+ levels before immune challenge, through ongoing supplementation or prophylactic treatment, may improve the capacity for an effective immune response. This concept has gained traction in the context of pandemic preparedness, where optimizing baseline immune metabolic capacity could improve population resilience to novel infectious threats.
Conversely, beginning high-dose NAD+ supplementation during an acute infection is more complex. While supporting cellular metabolism during infection is theoretically beneficial, some pathogens may also benefit from increased cellular NAD+ levels. The timing and dosing of NAD+ therapy relative to acute infection requires further investigation.
Practical Considerations for Patients and Clinicians
Beyond the science and protocols, several practical considerations influence the real-world implementation of NAD+ therapy. This section addresses common questions and concerns that arise in clinical practice.
Choosing a Provider
The quality of NAD+ therapy depends heavily on the provider and their protocols. Patients should look for:
- Medical oversight: A licensed physician should supervise all NAD+ therapy, even for SubQ self-injection programs. The physician should review medical history, order appropriate lab work, prescribe therapy, and provide ongoing monitoring.
- Compounding pharmacy quality: NAD+ should be sourced from a 503A or 503B compounding pharmacy that follows Good Manufacturing Practices (GMP). The pharmacy should provide certificates of analysis confirming purity, sterility, and potency.
- Experienced staff: For IV therapy, nursing staff should be experienced with NAD+ infusions specifically, as rate management and side effect handling differ from standard IV therapies.
- Appropriate monitoring: The provider should order baseline labs, follow-up labs, and adjust protocols based on individual response rather than using a one-size-fits-all approach.
- Transparent pricing: Costs should be clearly communicated upfront, including any additional charges for adjunctive treatments, lab work, or supplies.
Quality Concerns with NAD+ Products
The NAD+ supplement and therapy market has grown rapidly, and product quality varies significantly. Key quality considerations include:
Injectable NAD+: Must be pharmaceutical-grade, sterile, and pyrogen-free. Products from unregulated or overseas sources carry risks of contamination, incorrect concentration, and degradation. Only compounding pharmacies regulated by state boards of pharmacy should be used for injectable NAD+.
Oral NR and NMN: Third-party testing for purity and potency is essential, as independent analyses have found that some commercial products contain significantly less active ingredient than claimed. Look for products with certificates of analysis from independent laboratories and Good Manufacturing Practice (GMP) certification.
Nasal spray formulations: Should be prepared by compounding pharmacies using pharmaceutical-grade NAD+ in an appropriate nasal delivery vehicle. Over-the-counter nasal spray products vary widely in quality and may not contain the claimed NAD+ concentration.
Lifestyle Factors That Support NAD+ Therapy
NAD+ supplementation works best in the context of lifestyle practices that support NAD+ metabolism:
Exercise: Aerobic and resistance exercise both increase NAMPT expression and boost the NAD+ salvage pathway. Regular exercise may reduce the dose of exogenous NAD+ needed to maintain optimal levels. Even moderate-intensity exercise (30 minutes of brisk walking) can meaningfully increase NAMPT expression.
Caloric restriction and fasting: Caloric restriction increases the NAD+/NADH ratio and activates SIRT1 through both NAD+-dependent and NAD+-independent mechanisms. Time-restricted eating (confining food intake to an 8-10 hour window) may similarly benefit NAD+ metabolism.
Sleep optimization: NAMPT expression follows a circadian rhythm, with higher activity during wakeful periods. Disrupted sleep disrupts NAD+ cycling and may impair NAD+ homeostasis. Prioritizing 7-9 hours of quality sleep supports natural NAD+ regulation.
Alcohol reduction: Alcohol metabolism directly consumes NAD+, converting it to NADH through alcohol dehydrogenase and aldehyde dehydrogenase. Even moderate alcohol consumption can partially counteract NAD+ supplementation. Patients taking NAD+ therapy should be advised to minimize alcohol intake.
Sun exposure management: UV radiation causes DNA damage that activates PARPs and consumes NAD+. While moderate sun exposure is important for vitamin D synthesis, excessive UV exposure depletes NAD+ and counteracts supplementation. Appropriate sun protection supports NAD+ conservation.
Dietary support: Foods rich in NAD+ precursors include dairy milk (contains NR), edamame, broccoli, avocado, and mushrooms (contain NMN). Tryptophan-rich foods (turkey, chicken, fish, eggs) support de novo NAD+ synthesis through the kynurenine pathway. While dietary sources alone are unlikely to meaningfully raise NAD+ levels compared to supplementation, they contribute to overall NAD+ metabolism support.
When to Expect Results
Patient expectations should be set appropriately based on the delivery route and clinical indication:
| Route | Subjective Response Onset | Objective Marker Changes | Maximum Benefit Timeline |
|---|---|---|---|
| IV NAD+ | Same day to 48 hours | Blood NAD+ elevation within hours | After loading series (2-4 weeks) |
| SubQ NAD+ | 3-7 days | Blood NAD+ elevation within days | 4-8 weeks of daily use |
| Nasal spray | 15-30 minutes (cognitive effects) | Limited data | Ongoing; best as adjunct |
| Oral NR/NMN | 1-3 weeks | Blood NAD+ elevation by 2-4 weeks | 6-12 weeks |
Patients who don't notice any subjective improvement after 6-8 weeks of consistent therapy at adequate doses should be re-evaluated. Consider checking intracellular NAD+ levels to confirm the chosen protocol is achieving meaningful elevation, reviewing potential confounding factors (sleep, alcohol, stress, underlying conditions), and potentially switching delivery routes or adding complementary strategies.
Cost Optimization Strategies
Given that NAD+ therapy is an out-of-pocket expense for most patients, cost optimization is a legitimate consideration:
- Start with oral precursors: NR or NMN at 500-1,000 mg/day provides meaningful NAD+ elevation at the lowest cost. If oral precursors achieve satisfactory results, injectable therapy may not be necessary.
- Use IV loading with oral/SubQ maintenance: The hybrid approach uses costly IV sessions only for initial loading, then transitions to less expensive maintenance routes.
- Buy in bulk when possible: Multi-month supplies of oral supplements and multi-vial purchases of injectable NAD+ often come at reduced per-unit pricing.
- Package deals for IV therapy: Many clinics offer package pricing for multiple IV sessions that reduces the per-session cost by 10-25%.
- Optimize lifestyle factors: Maximizing exercise, sleep, and dietary NAD+ support may allow lower supplement doses to achieve the same effect, reducing ongoing costs.
- Monitor and adjust: Periodic NAD+ testing allows dose optimization - some patients may find they can maintain adequate levels at lower doses than their starting protocol.
Future Directions in NAD+ Research
The NAD+ research field is evolving rapidly, with several emerging areas that may significantly change how we approach NAD+ therapy in the coming years.
Next-Generation NAD+ Precursors
Researchers are developing new NAD+ precursors designed to overcome limitations of current options. Dihydronicotinamide riboside (NRH) is reduced form of NR that raises NAD+ levels more potently than NR in preclinical models. Nicotinic acid riboside (NAR) enters the Preiss-Handler pathway rather than the salvage pathway, potentially reaching compartments that NR and NMN do not access efficiently. These next-generation precursors are in early development but may offer improved potency and tissue targeting.
CD38 Inhibitors as Adjunctive Therapy
Pharmaceutical development of specific CD38 inhibitors for anti-aging applications is underway. Unlike the broadly cytotoxic anti-CD38 antibodies used in multiple myeloma (daratumumab, isatuximab), these new agents are designed to specifically inhibit CD38's NADase activity without depleting CD38-expressing cells. If successful, these compounds could dramatically reduce NAD+ destruction in aging tissues and transform the dosing requirements for NAD+ replacement therapy.
Tissue-Targeted NAD+ Delivery
Current NAD+ delivery methods provide systemic elevation without targeting specific tissues. Future approaches may include nanoparticle-encapsulated NAD+ precursors that accumulate preferentially in specific organs (liver, brain, muscle), implantable slow-release devices that provide continuous NAD+ or precursor delivery, and tissue-specific NAMPT activators that boost endogenous NAD+ production in targeted tissues.
NAD+ Biomarkers and Personalized Dosing
The development of accessible, affordable NAD+ testing methods will enable personalized dosing based on individual NAD+ levels rather than standardized protocols. Point-of-care NAD+ testing, analogous to glucose monitoring, could allow patients to adjust their supplementation in real time based on measured levels. Genetic testing for NAD+ metabolism enzyme variants (NAMPT, NRK, NMNAT, CD38 polymorphisms) may further refine individualized treatment approaches.
Clinical Trials in Progress
Several important clinical trials are currently underway or recently completed that will significantly expand the evidence base for NAD+ therapy:
- NR supplementation in Alzheimer's disease (NAVIGATOR trial)
- NMN supplementation for cardiovascular function in older adults
- NAD+ IV therapy for addiction (randomized controlled trial design)
- NR for Long COVID fatigue and cognitive symptoms
- Combination NAD+ and exercise interventions for aging
- Head-to-head comparisons of NAD+ delivery routes
The results of these trials, expected over the next 2-5 years, will provide the rigorous evidence needed to move NAD+ therapy from an emerging intervention to a standard of care in aging medicine.
The Role of Artificial Intelligence in NAD+ Research
Emerging applications of artificial intelligence and machine learning in NAD+ research include predictive modeling of treatment response based on patient demographics, genetic profiles, and baseline biomarkers. AI-driven analysis of metabolomics data could identify patterns in the NAD+ metabolome that predict which patients will respond best to specific delivery routes and precursors. Drug discovery platforms are using AI to identify novel NAD+-boosting compounds and CD38 inhibitors with improved selectivity and potency.
Large-scale data aggregation from NAD+ therapy providers, combined with patient-reported outcomes, could create real-world evidence databases that supplement traditional clinical trial data. These databases could help establish best practices for dosing, frequency, and route selection across different patient populations. The integration of wearable device data (HRV, sleep metrics, activity levels) with NAD+ level monitoring creates opportunities for precision dosing algorithms that adjust therapy in real time based on physiological feedback.
Regulatory Landscape and Future Access
The regulatory environment for NAD+ therapy is evolving and varies significantly by jurisdiction. In the United States, NAD+ for injection is available through compounding pharmacies under physician prescription. Oral NR has FDA GRAS status and is sold as a dietary supplement. NMN's regulatory status has been uncertain, with the FDA questioning its classification as a dietary supplement due to its investigation as a pharmaceutical drug by Metro International Biotech.
In other countries, regulatory approaches vary from fully permissive to restrictive. Australia has placed NMN on the prescription-only medicine schedule. The European Union regulates NMN and NR under novel food frameworks. Japan has been among the most permissive markets for NMN supplements, with widespread availability and strong consumer adoption.
The future regulatory landscape will likely be shaped by the outcomes of ongoing clinical trials. If NAD+ precursors demonstrate clear clinical benefits in randomized controlled trials, regulatory pathways may expand to include approved pharmaceutical indications. Conversely, if safety signals emerge or efficacy is not confirmed, regulations could tighten. Patients and clinicians should stay informed about regulatory changes that may affect product availability and quality standards.
Figure 10: Emerging research directions in NAD+ therapy, including next-generation precursors, tissue targeting strategies, and key ongoing clinical trials.
NAD+ Testing: Understanding Your Results
One of the most practical questions patients and clinicians face is how to measure whether NAD+ therapy is actually working. Unlike many supplements where the benefits are purely subjective, NAD+ levels can be objectively measured through several testing methods. Understanding these tests, their strengths, and their limitations is essential for optimizing therapy.
Types of NAD+ Tests
Whole blood NAD+: This measures the total NAD+ content in a whole blood sample, including NAD+ within red blood cells, white blood cells, and plasma. Because red blood cells are the predominant cell type in blood, whole blood NAD+ primarily reflects erythrocyte NAD+ content. This test provides a useful overall indicator of NAD+ status but doesn't directly measure NAD+ levels in target tissues like the brain, heart, or liver.
Intracellular NAD+: The Jinfiniti intracellular NAD+ test measures NAD+ within blood cells (primarily lymphocytes and other white blood cells), providing a more functionally relevant measurement than total plasma NAD+. This test has gained significant adoption in the functional medicine community because intracellular NAD+ levels are more likely to reflect the cellular NAD+ status that drives sirtuin activity and metabolic function.
Plasma NAD+: Plasma NAD+ levels represent the extracellular NAD+ pool, which is rapidly metabolized by extracellular CD38 and other ectoenzymes. Plasma NAD+ is highly variable and may not correlate well with intracellular levels, making it a less reliable indicator of functional NAD+ status. However, it's useful for tracking acute changes during and immediately after IV or SubQ administration.
NAD+ metabolome: Comprehensive testing of the NAD+ metabolome measures not just NAD+ but also its metabolites including NMN, NR, nicotinamide (NAM), methylnicotinamide (MeNAM), and others. This provides a more complete picture of NAD+ metabolism, including salvage pathway activity (reflected by NMN and NAM levels) and methylation status (reflected by methylated metabolites).
Interpreting Your Results
Reference ranges for NAD+ testing vary by laboratory and methodology. As a general guide for the Jinfiniti intracellular NAD+ test:
| NAD+ Level (micromolar) | Interpretation | Clinical Action |
|---|---|---|
| Below 20 | Significantly depleted | Aggressive repletion recommended; consider IV loading |
| 20-30 | Below optimal | Supplementation strongly recommended; SubQ or oral |
| 30-40 | Suboptimal | Supplementation recommended; oral may be sufficient |
| 40-50 | Adequate | Maintenance supplementation optional; monitor periodically |
| Above 50 | Optimal | Continue current protocol or reduce dose |
These ranges are approximate and should be interpreted in the context of age, health status, symptoms, and treatment goals. A 65-year-old with chronic fatigue and a level of 25 may benefit more from aggressive repletion than a healthy 40-year-old with the same level but no symptoms.
Tracking Response Over Time
The most valuable aspect of NAD+ testing isn't a single measurement but tracking changes over time. A recommended testing schedule includes:
- Baseline: Before starting any NAD+ therapy, to establish your starting point and confirm the need for supplementation
- 4-8 weeks: After starting therapy, to confirm that your chosen protocol is achieving meaningful NAD+ elevation
- 3 months: To assess sustained response and adjust dosing if needed
- Every 6 months: Ongoing monitoring for long-term therapy, adjusting protocol based on levels and clinical response
- After protocol changes: Retest 4-6 weeks after switching delivery routes, adjusting doses, or adding/removing compounds
When Testing Reveals No Improvement
If NAD+ levels don't improve despite consistent supplementation, consider the following possibilities:
- Insufficient dose: The current dose may be too low to overcome NAD+ consumption. Consider increasing dose or switching to a higher-bioavailability route.
- Excessive CD38 activity: High CD38 expression may be consuming supplemented NAD+ as fast as it's delivered. Consider adding CD38 inhibitors (quercetin, apigenin) to the protocol.
- Ongoing NAD+-depleting factors: Alcohol use, chronic stress, poor sleep, or active inflammation may be counteracting supplementation. Address these factors alongside NAD+ therapy.
- Absorption issues: For oral supplements, GI conditions, dysbiosis, or malabsorption may reduce precursor uptake. Consider switching to injectable or sublingual delivery.
- Genetic factors: Polymorphisms in NAMPT, NRK, NMNAT, or CD38 genes may affect NAD+ metabolism efficiency. Pharmacogenomic testing may reveal relevant variants.
- Testing variability: Ensure testing is performed under consistent conditions (same time of day, fasting status, and time relative to last NAD+ dose) to minimize measurement variability.
The Role of Functional Biomarkers
Beyond direct NAD+ measurement, several functional biomarkers can provide indirect evidence of NAD+ status and sirtuin activity:
- HRV (Heart Rate Variability): Higher HRV correlates with better autonomic function and may improve with NAD+ supplementation. Many patients track HRV using wearable devices as a convenient daily indicator of treatment response.
- Glucose/insulin metrics: Improvements in fasting glucose, HbA1c, and HOMA-IR may reflect enhanced SIRT1-mediated insulin signaling.
- Inflammatory markers: Reductions in hs-CRP, IL-6, and TNF-alpha may indicate SIRT1-mediated NF-kB suppression.
- Homocysteine: Stable or declining homocysteine levels confirm adequate methylation support alongside NAD+ therapy.
- Uric acid: Monitoring for elevation, as purine metabolism intersects with NAD+ pathways.
- GlycanAge or other biological age markers: Some patients use epigenetic or glycan-based biological age testing to track whether NAD+ therapy is influencing aging markers over time. While individual test results should be interpreted cautiously, trends over multiple measurements may be informative.
The combination of direct NAD+ measurement with functional biomarker tracking provides the most comprehensive picture of treatment response and allows for data-driven protocol optimization. Patients who invest in this level of monitoring tend to achieve better outcomes because their protocols can be precisely tailored and adjusted based on objective data rather than subjective impressions alone.
Summary and Evidence Assessment
After reviewing the full scope of NAD+ therapy research, from basic biochemistry through clinical applications and practical protocols, it's useful to step back and assess the overall strength of evidence for each major application area. This helps clinicians and patients make appropriately calibrated decisions about when and how to use NAD+ therapy.
Evidence Strength by Application
| Application | Evidence Level | Confidence | Key Evidence |
|---|---|---|---|
| NAD+ decline with aging | Strong | High | Multiple human studies confirming 30-50% decline by age 50-60 |
| Oral NR/NMN raises blood NAD+ | Strong | High | Multiple randomized controlled trials with consistent results |
| IV NAD+ raises blood NAD+ | Moderate | High | Pilot studies confirm acute elevation; rapid clearance documented |
| Metabolic health improvement | Moderate | Moderate-High | RCT showing insulin sensitivity improvement with NMN |
| Vascular function improvement | Moderate | Moderate | Clinical trial showing trends in BP and arterial stiffness reduction |
| Addiction/withdrawal treatment | Low-Moderate | Moderate | Case series with consistent results; no RCTs completed |
| Anti-aging benefits | Moderate (preclinical), Low (clinical) | Moderate | Strong animal data; limited human aging-specific outcomes |
| Neuroprotection | Moderate (preclinical), Low (clinical) | Moderate | Consistent animal data; NADPARK trial shows brain NAD+ increase |
| Cognitive enhancement (healthy adults) | Low | Low-Moderate | Anecdotal clinical reports; no controlled cognitive outcome studies |
| Athletic performance | Low | Low | Studies show no improvement in healthy athletes; age-related benefit possible |
| Intranasal brain delivery | Low (animal only) | Low-Moderate | Strong animal data; no human PK studies |
Gaps in the Evidence
Several critical questions remain unanswered by current research:
- Head-to-head route comparisons: No study has directly compared IV, SubQ, nasal, and oral NAD+ delivery using standardized tissue-level outcome measures in the same population. The comparative data in this report are synthesized from separate studies.
- Tissue-level NAD+ measurement: Most studies measure blood or plasma NAD+, which may not accurately reflect intracellular or tissue-specific NAD+ levels. The relationship between blood NAD+ and functional NAD+ in target tissues (brain, heart, liver, muscle) is not well characterized in humans.
- Long-term safety: Most safety data come from studies lasting weeks to months. Multi-year safety data on continuous NAD+ supplementation at therapeutic doses do not exist.
- Optimal dosing: No dose-response studies have established the minimum effective dose or the dose ceiling for NAD+ therapy across different populations and indications.
- Individual variation: The role of genetic polymorphisms in NAD+ metabolism enzymes (NAMPT, NRK, NMNAT, CD38) in determining treatment response has not been systematically studied.
- Combination protocols: While combination approaches with peptides are increasingly used, no controlled trials have evaluated multi-compound protocols.
The Bottom Line
NAD+ therapy rests on a strong scientific foundation. The biology of NAD+ decline in aging is well-established, and the consequences of that decline for cellular function are well-characterized. The ability of various delivery routes to raise blood and tissue NAD+ levels is confirmed by clinical data. Where the evidence becomes less certain is in translating those biochemical changes into defined clinical benefits for specific patient populations.
For patients considering NAD+ therapy, the decision should be informed by realistic expectations, appropriate monitoring, and integration with a comprehensive health optimization strategy. NAD+ is not a magic bullet, but it addresses a real and measurable biological deficit that worsens with age. For many patients, particularly those in the 40-65 age range with symptoms consistent with mitochondrial dysfunction and metabolic decline, NAD+ therapy offers a rational, low-risk intervention with the potential for meaningful benefit.
The next few years will bring results from larger clinical trials that should sharpen our understanding of which patients benefit most, which delivery routes are optimal for specific applications, and what the long-term risk-benefit profile looks like. In the meantime, clinical practice continues to advance based on the convergence of strong basic science, favorable safety data, and consistent clinical observations.
For more information about NAD+ and complementary peptide therapies, explore the Peptide Research Hub, review the NAD+ Precursors and Longevity Report, or take the free assessment to explore personalized options.
Frequently Asked Questions
References
- Imai S, Guarente L. NAD+ and sirtuins in aging and disease. Trends Cell Biol. 2014;24(8):464-471. doi:10.1016/j.tcb.2014.04.002
- Camacho-Pereira J, Tarragó MG, Chini CCS, et al. CD38 Dictates Age-Related NAD Decline and Mitochondrial Dysfunction through an SIRT3-Dependent Mechanism. Cell Metab. 2016;23(6):1127-1139. doi:10.1016/j.cmet.2016.05.006
- Chini CCS, Peclat TR, Warner GM, et al. CD38 ecto-enzyme in immune cells is induced during aging and regulates NAD+ and NMN levels. Nat Metab. 2020;2(11):1284-1304. doi:10.1038/s42255-020-00298-z
- Grant R, Berg J, Mestayer R, et al. A Pilot Study Investigating Changes in the Human Plasma and Urine NAD+ Metabolome During a 6 Hour Intravenous Infusion of NAD+. Front Aging Neurosci. 2019;11:257. doi:10.3389/fnagi.2019.00257
- Martens CR, Denman BA, Mazzo MR, et al. Chronic nicotinamide riboside supplementation is well-tolerated and elevates NAD+ in healthy middle-aged and older adults. Nat Commun. 2018;9(1):1286. doi:10.1038/s41467-018-03421-7
- Yoshino M, Yoshino J, Kayser BD, et al. Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women. Science. 2021;372(6547):1224-1229. doi:10.1126/science.abe9985
- Gomes AP, Price NL, Ling AJY, et al. Declining NAD+ induces a pseudohypoxic state disrupting nuclear-mitochondrial communication during aging. Cell. 2013;155(7):1624-1638. doi:10.1016/j.cell.2013.11.037
- Li J, Bonkowski MS, Moniot S, et al. A conserved NAD+ binding pocket that regulates protein-protein interactions during aging. Science. 2017;355(6331):1312-1317. doi:10.1126/science.aad8242
- Airhart SE, Shireman LM, Risler LJ, et al. An open-label, non-randomized study of the pharmacokinetics of the nutritional supplement nicotinamide riboside (NR) and its effects on blood NAD+ levels in healthy volunteers. PLoS One. 2017;12(12):e0186459. doi:10.1371/journal.pone.0186459
- Brakedal B, Dölle C, Riber F, et al. The NADPARK study: A randomized phase I trial of nicotinamide riboside supplementation in Parkinson's disease. Cell Metab. 2022;34(3):396-407.e6. doi:10.1016/j.cmet.2022.02.001
- Mestayer R, Bhatt P, Gould GG, et al. Nicotinamide Adenine Dinucleotide (NAD+) and Enkephalinase Inhibition (IV1114589NAD) Infusions Significantly Attenuate Psychiatric Burden Sequalae in Substance Use Disorder (SUD) in Fifty Cases. J Pers Med. 2022;12(9):1472. doi:10.3390/jpm12091472
- Wei CC, Kong YY, Li GQ, Guan YF, Wang P, Miao CY. Intranasal administration with NAD+ profoundly decreases brain injury in a rat model of transient focal ischemia. Front Biosci. 2007;12:2728-2734. doi:10.2741/2267
- Radenkovic D, Reason, Engelen MPKJ. Evaluation of safety and effectiveness of NAD in different clinical conditions: a systematic review. Am J Physiol Endocrinol Metab. 2023;325(6):E703-E719. doi:10.1152/ajpendo.00225.2023
- Verdin E. NAD+ in aging, metabolism, and neurodegeneration. Science. 2015;350(6265):1208-1213. doi:10.1126/science.aac4854
- Rajman L, Chwalek K, Sinclair DA. Therapeutic Potential of NAD-Boosting Molecules: The In Vivo Evidence. Cell Metab. 2018;27(3):529-547. doi:10.1016/j.cmet.2018.02.011
- Cantó C, Menzies KJ, Auwerx J. NAD+ Metabolism and the Control of Energy Homeostasis: A Balancing Act between Mitochondria and the Nucleus. Cell Metab. 2015;22(1):31-53. doi:10.1016/j.cmet.2015.05.023
- Katsyuba E, Romani M, Hober D, Auwerx J. NAD+ homeostasis in health and disease. Nat Metab. 2020;2(1):9-31. doi:10.1038/s42255-019-0161-5
- Chini EN, Chini CCS, Espindola Netto JM, de Oliveira GC, van Schooten W. The Pharmacology of CD38/NADase: An Emerging Target in Cancer and Diseases of Aging. Trends Pharmacol Sci. 2018;39(4):424-436. doi:10.1016/j.tips.2018.02.001
- Covarrubias AJ, Perrone R, Grozio A, Verdin E. NAD+ metabolism and its roles in cellular processes during ageing. Nat Rev Mol Cell Biol. 2021;22(2):119-141. doi:10.1038/s41580-020-00313-x
- Shade C. The Science Behind NMN - A Stable, Reliable NAD+Activator and Anti-Aging Molecule. Integr Med (Encinitas). 2020;19(1):12-14.
- Yi L, Maier AB, Tao R, et al. The efficacy and safety of beta-nicotinamide mononucleotide (NMN) supplementation in healthy middle-aged adults: a randomized, multicenter, double-blind, placebo-controlled, parallel-group, dose-dependent clinical trial. Geroscience. 2023;45(1):29-43. doi:10.1007/s11357-022-00705-1
- Trammell SAJ, Schmidt MS, Weidemann BJ, et al. Nicotinamide riboside is uniquely and orally bioavailable in mice and humans. Nat Commun. 2016;7:12948. doi:10.1038/ncomms12948
- Lautrup S, Sinclair DA, Mattson MP, Fang EF. NAD+ in Brain Aging and Neurodegenerative Disorders. Cell Metab. 2019;30(4):630-655. doi:10.1016/j.cmet.2019.09.001
- Elhassan YS, Kluckova K, Fletcher RS, et al. Nicotinamide Riboside Augments the Aged Human Skeletal Muscle NAD+ Metabolome and Induces Transcriptomic and Anti-inflammatory Signatures. Cell Rep. 2019;28(7):1717-1728.e6. doi:10.1016/j.celrep.2019.07.043
- Clement J, Wong M, Poljak A, Sachdev P, Braidy N. The Plasma NAD+ Metabolome Is Dysregulated in "Normal" Aging. Rejuvenation Res. 2019;22(2):121-130. doi:10.1089/rej.2018.2077
- Zapata-Pérez R, Wanders RJA, van Karnebeek CDM, Houtkooper RH. NAD+ homeostasis in human health and disease. EMBO Mol Med. 2021;13(7):e13943. doi:10.15252/emmm.202113943
- O'Hollaren P. Diphosphopyridine nucleotide in the prevention, diagnosis, and treatment of drug addiction. A preliminary report. West J Surg Obstet Gynecol. 1961;69:213-215.
- Conlon N, Ford D. A systems-approach to NAD+ restoration. Biochem Pharmacol. 2022;198:114946. doi:10.1016/j.bcp.2022.114946
- Mehmel M, Jovanovic N, Spitz U. Nicotinamide Riboside - The Current State of Research and Therapeutic Uses. Nutrients. 2020;12(6):1616. doi:10.3390/nu12061616
- Yang Y, Zhang N, Zhang G, Sauve AA. NRH:quinone oxidoreductase 2 (NQO2) and the regulation of NAD+ metabolism. Free Radic Biol Med. 2019;142:100-117. doi:10.1016/j.freeradbiomed.2019.02.008
- Chini CCS, Zeidler JD, Kashyap S, Warner G, Chini EN. Evolving concepts in NAD+ metabolism. Cell Metab. 2021;33(6):1076-1087. doi:10.1016/j.cmet.2021.04.003
- Bonkowski MS, Sinclair DA. Slowing ageing by design: the rise of NAD+ and sirtuin-activating compounds. Nat Rev Mol Cell Biol. 2016;17(11):679-690. doi:10.1038/nrm.2016.93
- Remie CME, Roumans KHM, Moonen MPB, et al. Nicotinamide riboside supplementation alters body composition and skeletal muscle acetylcarnitine concentrations in healthy obese humans. Am J Clin Nutr. 2020;112(2):413-426. doi:10.1093/ajcn/nqaa072
- Nadeeshani H, Li J, Ying T, Shen B, Lu J. Nicotinamide mononucleotide (NMN) as an anti-aging health product - Promises and safety concerns. J Adv Res. 2022;37:267-278. doi:10.1016/j.jare.2021.08.003
- Hou Y, Lautrup S, Cordonnier S, et al. NAD+ supplementation normalizes key Alzheimer's features and DNA damage responses in a new AD mouse model with introduced DNA repair deficiency. Proc Natl Acad Sci U S A. 2018;115(8):E1876-E1885. doi:10.1073/pnas.1718819115
- Braidy N, Berg J, Clement J, et al. Role of Nicotinamide Adenine Dinucleotide and Related Precursors as Therapeutic Targets for Age-Related Degenerative Diseases: Rationale, Biochemistry, Pharmacokinetics, and Outcomes. Antioxid Redox Signal. 2019;30(2):251-294. doi:10.1089/ars.2017.7269
- Chini EN, Tarragó MG, Chini CCS. NAD and the aging process: Role in life, death and everything in between. Mol Cell Endocrinol. 2017;455:62-74. doi:10.1016/j.mce.2016.11.030
Explore More Research
This report is part of FormBlends' comprehensive peptide research library. For related topics, visit the Peptide Research Hub or explore specific compound reports including Epithalon and Telomere Biology, MOTS-c and Mitochondrial Function, and our comprehensive NAD+ Precursors and Longevity Peptides report.