Executive Summary
The legal status of peptides in the United States sits at the intersection of FDA drug approval law, pharmacy compounding regulation, state licensing requirements, and international trade controls. Whether a particular peptide is "legal" depends entirely on who's selling it, how it's being marketed, and what regulatory pathway - if any - it follows.
Key Takeaways
- The legal status of peptides in the United States sits at the intersection of FDA drug approval law, pharmacy compounding regulation, state licensing requirements, and international trade controls.
- Whether a particular peptide is "legal" depends entirely on who's selling it, how it's being marketed, and what regulatory pathway - if any - it follows.
- Peptides aren't a single legal category.
- The confusion surrounding peptide legality is understandable: the regulatory framework wasn't designed with this class of therapeutics in mind, and enforcement has been inconsistent for years.
- What changed dramatically in 2023 and 2024 was the FDA's willingness to crack down.
Peptides aren't a single legal category. They're a diverse class of molecules that can be FDA-approved drugs, legally compounded medications, research chemicals sold with disclaimers, dietary supplement ingredients, or outright controlled substances depending on the specific compound and context. The confusion surrounding peptide legality is understandable: the regulatory framework wasn't designed with this class of therapeutics in mind, and enforcement has been inconsistent for years.
What changed dramatically in 2023 and 2024 was the FDA's willingness to crack down. The agency placed 17 popular peptides into Category 2 of the interim bulk drug substance list, effectively banning them from pharmacy compounding. It then resolved the shortages for both semaglutide and tirzepatide, pulling the rug out from under compounding pharmacies that had been producing these GLP-1 receptor agonists legally during shortage periods. And it sent more than 50 warning letters to companies involved in GLP-1 compounding and manufacturing.
KEY TAKEAWAY
Peptide legality depends on three factors: (1) whether the specific peptide has FDA approval or appears on the bulk drug substance compounding list, (2) whether it's being sold through a licensed pharmacy with a valid prescription, and (3) how it's being marketed and labeled. The "research chemical" loophole is narrowing as the FDA increases enforcement.
This guide walks through every layer of the regulatory framework. We'll cover the FDA's drug approval process, the research chemical gray area, 503A and 503B compounding pharmacy laws, bulk drug substance categories, state-level variations, international regulations, WADA prohibitions for athletes, telehealth prescribing rules, and the most recent FDA enforcement actions. Whether you're a researcher, clinician, patient, or someone trying to understand where the legal boundaries actually fall, this report provides the factual foundation you need.
For those looking to understand specific peptide products and their research profiles, the FormBlends Peptide Research Hub provides detailed compound-by-compound analysis. And for information about compounding pharmacy regulations specifically, the Compounding Pharmacy Peptides Guide offers additional context.
Figure 1: The regulatory landscape for peptides involves overlapping federal and state jurisdictions, each with distinct rules governing access and legality.
FDA Regulatory Framework
The Food and Drug Administration's authority over peptides stems from the Federal Food, Drug, and Cosmetic Act (FDCA), which defines a "drug" broadly enough to encompass virtually any peptide marketed with therapeutic claims.
How the FDCA Defines a Drug
Under 21 U.S.C. Section 321(g)(1), a "drug" includes articles intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease, as well as articles (other than food) intended to affect the structure or any function of the body. This definition doesn't require a substance to be approved by the FDA to be classified as a drug. It only requires that it be intended for drug-like use.
This "intended use" doctrine is where most peptide sellers run into trouble. The FDA determines intended use not just from product labels but from the totality of evidence, including website marketing, social media posts, testimonials, blog content, product names, and even customer reviews that sellers allow to remain on their platforms. A peptide labeled "for research use only" but sold alongside dosing guides, injection instructions, and testimonials about muscle growth will be treated as an unapproved drug regardless of its disclaimer.
The New Drug Application (NDA) Process
For a peptide to be legally marketed as a drug in the United States, it must go through the FDA's formal approval process. This typically involves:
- Investigational New Drug (IND) Application: Before clinical trials can begin, the sponsor must file an IND with the FDA containing preclinical data, manufacturing information, and a clinical trial protocol
- Phase I Trials: Small-scale safety studies in healthy volunteers (typically 20-100 subjects)
- Phase II Trials: Efficacy and dosing studies in patients with the target condition (typically 100-500 subjects)
- Phase III Trials: Large-scale confirmatory trials demonstrating safety and efficacy (typically 1,000-5,000+ subjects)
- New Drug Application (NDA): The formal submission containing all clinical trial data, manufacturing processes, labeling, and proposed uses
- FDA Review: A 10-12 month standard review or 6-month priority review period
This process costs an estimated $1-3 billion per drug and takes 10-15 years on average. The cost barrier explains why many peptides with promising research profiles never reach FDA approval; the commercial market for a specific peptide may not justify the investment required for a full NDA submission.[1]
FDA-Approved Peptide Drugs
Despite the high barriers, numerous peptides have achieved FDA approval. These include some of the most commercially successful drugs in history:
| Peptide Drug | Brand Name(s) | Approved For | Year Approved |
|---|---|---|---|
| Semaglutide | Ozempic, Wegovy, Rybelsus | Type 2 diabetes, obesity | 2017/2021/2024 |
| Tirzepatide | Mounjaro, Zepbound | Type 2 diabetes, obesity | 2022/2023 |
| Liraglutide | Victoza, Saxenda | Type 2 diabetes, obesity | 2010/2014 |
| Exenatide | Byetta, Bydureon | Type 2 diabetes | 2005/2012 |
| Tesamorelin | Egrifta | HIV lipodystrophy | 2010 |
| Octreotide | Sandostatin | Acromegaly, carcinoid tumors | 1988 |
| Vasopressin | Vasostrict | Vasodilatory shock | 2014 |
| Leuprolide | Lupron | Prostate cancer, endometriosis | 1985 |
| Gonadorelin | Factrel | Diagnostic agent (LH/FSH) | 1973 |
| Oxytocin | Pitocin | Labor induction | 1980 |
For detailed research on GLP-1 peptides specifically, including semaglutide and tirzepatide, FormBlends maintains updated compound profiles with clinical trial data and mechanism-of-action information.
The Investigational New Drug (IND) Pathway in Detail
For peptides that haven't yet been approved by the FDA, the IND process represents the formal pathway toward eventual approval. Understanding this process helps explain why so many peptides with promising research remain unavailable through conventional medical channels.
Filing an IND application is the first formal step toward FDA approval. The application must contain three essential components: animal pharmacology and toxicology studies sufficient to demonstrate safety for initial human testing, manufacturing information demonstrating that the compound can be consistently produced with adequate quality, and clinical protocols describing the proposed human trials.
The pre-IND meeting with the FDA is often a critical milestone. During this meeting, sponsors discuss their development plans with FDA reviewers and receive feedback on study design, endpoints, and regulatory strategy. For peptides, common discussion topics include the appropriate reference standard for analytical testing, the justification for starting dose in humans based on animal pharmacology data, the duration of toxicology studies needed to support the proposed clinical trial duration, and the specific populations to be studied in initial trials.
After IND filing, the FDA has 30 days to review the application. If the agency doesn't place a "clinical hold" within that period, the sponsor can proceed with clinical trials. Clinical holds are relatively uncommon but can occur if the FDA identifies safety concerns, inadequate manufacturing controls, or deficient clinical protocols.
The Phase I-III clinical trial process for peptides follows the same general framework as for other drugs, but peptides present unique considerations. Peptide stability can be challenging, requiring careful temperature control and limited shelf life that complicates multi-site clinical trials. Immunogenicity is a concern because the body may develop antibodies against synthetic peptides, potentially reducing efficacy or causing adverse reactions over time. Bioavailability for non-injectable formulations (oral, nasal, transdermal) is often poor, requiring specialized delivery technologies. And peptide-specific analytical methods must be validated for each compound, adding time and cost to the development process.
The end-of-Phase II meeting with the FDA is another critical juncture. At this meeting, sponsors present their Phase II efficacy and safety data and propose Phase III trial designs. The FDA provides feedback on whether the Phase III plans are likely to support approval. For peptide developers, this feedback is especially important because Phase III trials represent the largest investment in the development program.
After successful Phase III trials, the sponsor submits an NDA containing the complete body of evidence supporting the peptide's safety and efficacy. The FDA's review process typically takes 10-12 months for a standard review or 6 months for a priority review. Priority review is granted for drugs that offer significant improvements over existing treatments, and some peptides - particularly those addressing unmet medical needs - may qualify.
The entire IND-to-approval process typically costs $1-3 billion and takes 10-15 years. These numbers explain why many peptides with genuine therapeutic potential never reach FDA approval. The commercial return must justify the investment, and for peptides with limited market size or complex patent situations, the economics simply don't work. This is one of the core tensions in peptide regulation: the regulatory framework demands a level of investment that is economically feasible only for a subset of potentially beneficial compounds.[1]
Orphan Drug and Fast Track Designations
The FDA offers several expedited review pathways that could be relevant to peptide development:
Orphan Drug Designation: Available for drugs targeting rare diseases affecting fewer than 200,000 people in the US. Orphan designation provides tax credits for clinical trials, fee waivers, and seven years of marketing exclusivity. Some peptides could potentially qualify for orphan designation if targeted to specific rare conditions.
Fast Track Designation: Available for drugs intended to treat serious conditions that address an unmet medical need. Fast Track designation provides more frequent meetings with the FDA, rolling review of the NDA (allowing the FDA to review completed sections before the full application is submitted), and potential eligibility for accelerated approval.
Breakthrough Therapy Designation: Available for drugs intended to treat serious conditions where preliminary clinical evidence demonstrates substantial improvement over existing treatments. Breakthrough designation provides all Fast Track features plus intensive FDA guidance on drug development.
Accelerated Approval: Allows approval based on a surrogate endpoint that is reasonably likely to predict clinical benefit, with post-marketing confirmatory trials required. This pathway has been used for some peptide drugs.
These expedited pathways could potentially reduce the time and cost of peptide development, but they don't eliminate the fundamental requirement for clinical evidence of safety and efficacy. They also require proactive engagement from peptide developers, which may not occur for compounds without strong commercial sponsors.
Biologics vs. Small-Molecule Drugs
An important regulatory distinction exists between peptides classified as "drugs" under the FDCA and those classified as "biological products" under the Public Health Service Act (PHSA). Generally, peptides with 40 or fewer amino acids are regulated as drugs through the NDA pathway, while larger proteins follow the Biologics License Application (BLA) pathway. This distinction matters because the BLA pathway has different exclusivity provisions and different rules for generic competition (biosimilars vs. generic drugs).
Most of the peptides popular in wellness and anti-aging communities - BPC-157, TB-500, CJC-1295, ipamorelin, and others - are small enough to fall under the drug pathway rather than the biologics pathway. But since none of them have FDA approval through either route, the practical distinction is largely academic for these compounds.
The Dietary Supplement Exception
Under the Dietary Supplement Health and Education Act (DSHEA) of 1994, substances that qualify as "dietary supplements" face much lighter regulation than drugs. However, peptides rarely qualify for this exception. To be a lawful dietary supplement, a substance must either have been marketed as a dietary supplement before October 15, 1994, or the manufacturer must file a New Dietary Ingredient (NDI) notification with the FDA at least 75 days before marketing.
Most synthetic peptides don't meet either criterion. The FDA has repeatedly stated that injectable products cannot be dietary supplements regardless of their composition, since supplements must be intended for ingestion (oral consumption). This means that any injectable peptide product marketed as a "supplement" is automatically misbranded under federal law.[2]
REGULATORY NOTE
Certain amino acid chains and naturally occurring peptides found in food (like collagen peptides or casein-derived bioactive peptides) can be lawfully sold as dietary supplements when marketed for oral consumption without drug claims. The line between a "dietary supplement" peptide and an "unapproved drug" peptide depends on specific claims, form of administration, and the peptide's regulatory history.
FDA's Enforcement Authority
When the FDA determines that a peptide is being marketed as an unapproved drug, it has several enforcement options:
- Warning Letters: The most common first step, giving the company notice of violations and a deadline to correct them (typically 15 business days)
- Import Alerts: Allow for automatic detention without physical examination of imported products from specific companies or countries
- Seizure: The FDA can ask the Department of Justice to file a seizure action in federal court to remove products from the market
- Injunctions: Court orders prohibiting a company from continuing to manufacture, distribute, or sell violative products
- Criminal Prosecution: In egregious cases, the FDA can refer matters for criminal prosecution, which can result in fines and imprisonment
Through 2023 and 2024, the FDA dramatically escalated its enforcement activity related to peptides. The agency sent warning letters to more than 50 companies involved in GLP-1 compounding alone, and issued additional warnings to companies selling research peptides that were clearly intended for human use.[3]
The Role of the FTC in Peptide Advertising
While the FDA focuses on product safety and labeling, the Federal Trade Commission (FTC) has jurisdiction over advertising claims. The FTC Act prohibits unfair or deceptive practices, including false advertising of health products. For the peptide market, FTC enforcement can target:
- Unsubstantiated efficacy claims: Companies that advertise specific health benefits of peptides without adequate scientific support face FTC enforcement. The FTC requires "competent and reliable scientific evidence" to support health claims, which generally means at least one well-designed clinical trial
- Testimonials and endorsements: Customer testimonials must reflect typical results, and material connections between endorsers and sellers must be disclosed. The peptide market's heavy reliance on influencer marketing and user testimonials creates FTC exposure
- Social media advertising: The FTC's Endorsement Guides apply to social media posts about peptides. Influencers who receive free products or payment to promote peptides must disclose these relationships
- Native advertising: Content that appears to be editorial but is actually advertising must be clearly identified as such. Many peptide companies create "educational" blog content that is actually product promotion, which can violate FTC native advertising guidelines
The FTC has brought enforcement actions against companies in related markets (dietary supplements, weight loss products) and could extend this enforcement to the peptide market, particularly as the market grows more visible.
State Consumer Protection Laws
In addition to federal regulatory authority, all 50 states have consumer protection laws that can be applied to peptide sales. State attorneys general and, in some states, individual consumers can bring actions under these laws. Key state-level provisions include:
- Unfair and deceptive practices: State UDAP laws prohibit misleading marketing and sales practices. Claims about peptide benefits that aren't adequately supported can violate these laws
- False advertising: State false advertising statutes provide additional enforcement mechanisms beyond federal law
- Unauthorized practice of medicine: Companies that provide personalized health advice about peptide use without medical licensing may be engaging in unauthorized practice of medicine under state law
- Class action exposure: State consumer protection laws often provide for class action lawsuits, which can result in substantial damages for companies engaged in deceptive peptide marketing practices
Figure 2: FDA regulatory decision tree for peptide products. The pathway depends on marketing claims, form of administration, and whether the compound has prior approval or compounding authorization.
Understanding Peptide Categories Under Federal Law
Before examining specific regulatory pathways, it's helpful to understand how federal law categorizes different types of peptides. The classification determines everything from who can sell them to what penalties apply for violations.
Natural vs. Synthetic Peptides
Not all peptides are treated equally under federal law. The distinction between naturally occurring peptides and synthetic analogs has regulatory implications that affect legality, marketing claims, and enforcement priorities.
Naturally occurring peptides found in food sources - collagen peptides from bone broth, casein-derived bioactive peptides from dairy, and glutathione from various foods - generally face lighter regulation when sold for oral consumption without drug claims. These products can sometimes qualify as dietary supplements under DSHEA, provided they were marketed before the 1994 cutoff date or have appropriate New Dietary Ingredient notifications.
Synthetic peptides, by contrast, are almost always treated as drugs when intended for therapeutic use. Even when a synthetic peptide has the exact same amino acid sequence as a naturally occurring one, the manufacturing process and intended use determine its regulatory classification. A BPC-157 molecule produced by solid-phase peptide synthesis in a laboratory is legally different from a peptide fragment that happens to be present in gastric juice, even though the amino acid sequence is identical.
This distinction creates some counterintuitive situations. Collagen peptides sold as a supplement for skin health are perfectly legal and widely available in grocery stores. But a synthetic peptide with documented wound-healing properties sold for injection is an unapproved drug. The molecular complexity doesn't determine the legal classification - the intended use and marketing context do.
Endogenous Peptide Hormones
Peptides that are identical to hormones naturally produced by the human body present unique regulatory considerations. Growth hormone (somatotropin), for instance, is produced by the pituitary gland but is also a controlled substance when obtained without a prescription. The Anabolic Steroids Control Act of 1990, as amended, includes human growth hormone in its regulatory framework, making unauthorized distribution a criminal offense.
Other endogenous peptide hormones, like oxytocin and vasopressin, have FDA-approved pharmaceutical versions but can also be compounded by pharmacies under appropriate conditions. The legal status depends on the source and the regulatory pathway through which the product reaches the patient.
Synthetic analogs of endogenous peptides - molecules designed to mimic or enhance the effects of naturally occurring hormones - face the strictest regulatory scrutiny. GLP-1 receptor agonists like semaglutide and tirzepatide are synthetic analogs of the naturally occurring incretin hormone GLP-1, but they've gone through the full NDA process and achieved FDA approval. Other synthetic analogs, like CJC-1295 (a synthetic analog of GHRH) and ipamorelin (a synthetic growth hormone secretagogue), have not been approved and face compounding restrictions.
Peptide Length and Regulatory Implications
The size of a peptide molecule can influence its regulatory pathway. The FDA generally draws a line at 40 amino acids: peptides with 40 or fewer amino acids are typically regulated as drugs through the NDA pathway, while larger proteins follow the Biologics License Application (BLA) pathway under the Public Health Service Act.
This distinction matters for several reasons:
- Generic competition: Drugs approved through the NDA pathway face generic competition through Abbreviated New Drug Applications (ANDAs). Biologics approved through the BLA pathway face biosimilar competition, which has different evidentiary requirements
- Data exclusivity: The exclusivity periods differ between NDA and BLA approvals, affecting when competitors can enter the market
- Manufacturing standards: While both pathways require cGMP compliance, the specific manufacturing requirements differ for small molecules versus biological products
- Analytical characterization: Smaller peptides are generally easier to characterize analytically, which affects quality control and stability testing requirements
Most peptides popular in the research and wellness communities fall under the drug pathway. BPC-157 is a 15-amino-acid peptide. CJC-1295 has 29 amino acids (or 30 with DAC). Ipamorelin is just 5 amino acids. Thymosin alpha-1 has 28 amino acids. All of these are well under the 40-amino-acid threshold.
Controlled Substance Classification
The Controlled Substances Act (CSA) classifies drugs into five schedules based on their potential for abuse, accepted medical use, and safety profile. Most peptides are not controlled substances, which means they don't carry the same criminal penalties for possession as drugs like opioids or anabolic steroids.
However, certain peptide-related substances do have controlled substance implications:
| Substance | CSA Schedule | Key Implications |
|---|---|---|
| Human Growth Hormone (HGH) | Not scheduled, but distribution restricted under 21 USC 333(e) | Distribution for non-approved uses is a felony; up to 10 years imprisonment |
| Anabolic Steroids (including SARMs if scheduled) | Schedule III | Possession without prescription is a federal crime; distribution carries enhanced penalties |
| Insulin | Not scheduled | Prescription required but no criminal penalties for possession; available OTC in some states |
| Most research peptides | Not scheduled | No criminal penalties for possession; sale for human use without FDA approval is prohibited |
The non-scheduled status of most peptides means that individual possession typically doesn't carry criminal penalties. This is different from anabolic steroids, where simple possession without a prescription is a federal crime under Schedule III. However, the lack of scheduling doesn't make peptides "legal" for human use - it just means the penalties for individual users are civil rather than criminal in nature.
The Supplement vs. Drug Boundary
Understanding where the dietary supplement boundary falls is critical for anyone involved in the peptide market. The FDCA and DSHEA create a framework where the same molecule can be either a supplement or a drug depending on how it's marketed:
- Structure/function claims: A supplement can claim to "support joint health" but cannot claim to "treat arthritis." The former is a permissible structure/function claim; the latter is a drug claim that triggers FDA regulation
- Form of administration: Supplements must be intended for oral ingestion. Injectable, transdermal (with penetration enhancers), and inhaled forms are automatically classified as drugs regardless of the substance
- New Dietary Ingredient status: Substances not marketed as supplements before 1994 require NDI notifications, and many peptides would not meet the safety standard for NDI acceptance
- Good Manufacturing Practice: Supplements must be manufactured under 21 CFR Part 111 (supplement GMP), while drugs require 21 CFR Part 211 (drug GMP). The drug GMP requirements are substantially more stringent
In practice, very few synthetic peptides can legally be sold as supplements. The main exceptions are certain amino acid combinations and naturally derived peptides (like collagen peptides) sold for oral consumption with appropriate structure/function claims. Any company selling synthetic peptides as "dietary supplements" is almost certainly violating federal law.
CLASSIFICATION SUMMARY
The legal classification of any peptide depends on: (1) whether it's natural or synthetic, (2) whether it's marketed for therapeutic or nutritional purposes, (3) the form of administration (oral vs. injectable), (4) whether it has FDA approval or appears on the bulk drug substance list, and (5) whether it's classified as a controlled substance. Each of these factors independently affects the legal analysis, and all must be considered together.
Research Chemical Classification
The "research chemical" designation is not an official FDA category. It's a market-created label that sellers use to sidestep drug approval requirements by claiming their products are intended only for in-vitro laboratory research.
What "For Research Use Only" Actually Means
When a peptide is sold labeled "for research use only" (RUO) or "not for human consumption," the seller is attempting to position the product outside the FDA's drug regulation framework. The theory is simple: if a substance isn't intended for human use, it doesn't meet the FDCA's definition of a drug and doesn't require FDA approval.
In legitimate scientific research, RUO reagents serve a real purpose. Researchers studying cell biology, pharmacology, and molecular signaling need access to peptides and other bioactive compounds without going through the drug approval process. Academic labs, biotech companies, and pharmaceutical developers all rely on research-grade chemicals that haven't been manufactured under current Good Manufacturing Practice (cGMP) conditions required for human-use products.
The problem is that the vast majority of "research chemical" peptides sold online aren't going to research laboratories. They're being purchased by individuals for personal use, and everyone involved in the transaction understands this. The disclaimers serve as legal cover, not honest descriptions of intended use.
The FDA's Position on Research Chemical Peptides
The FDA has made its position clear through enforcement actions: a "research use only" label does not protect sellers from regulatory action when evidence shows the products are intended for human consumption. The agency considers the following factors when determining intended use:[4]
- Marketing materials: Website content discussing human benefits, dosing for humans, or clinical results in humans
- Product presentation: Packaging in single-dose vials, inclusion of bacteriostatic water, provision of syringes or injection supplies
- Customer communications: Emails, chat logs, or phone conversations providing dosing guidance
- Pricing and quantities: Products sold in quantities and concentrations consistent with personal use rather than laboratory research
- Customer demographics: Marketing to fitness enthusiasts, anti-aging consumers, or wellness communities rather than research institutions
- Website context: Blog posts about peptide benefits for humans, forums discussing personal experiences, or links to clinical studies describing human outcomes
In December 2024, the FDA issued a warning letter to Summit Research Peptides, finding that despite "research" branding, the company's products were marketed as unapproved new drugs based on website content, product descriptions, and overall marketing context. This letter followed a pattern of similar enforcement actions throughout 2024.[5]
Legal Risks for Buyers and Sellers
The legal exposure differs significantly between buyers and sellers of research chemical peptides:
For Sellers
Companies selling peptides labeled as research chemicals face substantial legal risk if the FDA determines their products are intended for human use. Violations of the FDCA can result in:
- Warning letters requiring corrective action within 15 business days
- Product seizure and destruction
- Injunctions prohibiting future sales
- Civil monetary penalties
- Criminal prosecution with potential imprisonment (up to 10 years for repeat offenders)
State attorneys general can also pursue enforcement under state consumer protection laws, adding another layer of legal exposure.
For Buyers
Individual purchasers of research chemical peptides face a more nuanced legal situation. The FDA has historically focused its enforcement on sellers rather than end users. However, purchasing a substance marketed for research and using it for self-administration carries several risks:
- No quality assurance: Research chemicals aren't manufactured under cGMP conditions. Purity, sterility, and potency are not guaranteed
- No legal recourse: If a research chemical causes harm, the buyer has limited legal remedies since they used the product contrary to its labeled purpose
- Import seizure: Customs and Border Protection can seize imported peptides that appear to be unapproved drugs
- Professional consequences: Healthcare professionals who purchase and administer research chemicals to patients face licensing board action
SAFETY CONCERN
Research chemical peptides are not manufactured under pharmaceutical-grade conditions. Independent testing has found that some products contain incorrect peptides, wrong concentrations, bacterial contamination, or harmful impurities. Without cGMP oversight, there's no guarantee that what's on the label matches what's in the vial. For information about evaluating peptide suppliers, see the Peptide Sciences and Research Suppliers Guide.
The "Research Chemical" Market in Practice
Despite the legal ambiguity, the research chemical peptide market has grown substantially over the past decade. Online sellers operate openly, often with sophisticated e-commerce platforms, customer service departments, and loyalty programs. Many accept credit card payments, offer subscription models, and provide detailed "educational" content about each peptide's effects on human physiology.
The market's persistence reflects several realities:
- FDA resource constraints: The agency lacks the staffing and budget to pursue every seller, so enforcement tends to target the most visible or egregious offenders
- Consumer demand: Many peptides offer genuine therapeutic potential that isn't accessible through traditional medical channels, creating strong market demand
- International sourcing: Many research peptides are manufactured overseas (particularly in China), making supply chain enforcement more difficult
- Legal gray area: The combination of RUO labeling and the FDA's focus on sellers creates a perceived (if not actual) safe harbor for individual buyers
SARMs: A Parallel Cautionary Tale
Selective Androgen Receptor Modulators (SARMs) provide an instructive parallel to the peptide market. Like peptides, SARMs were initially sold as research chemicals with "not for human consumption" labels. Like peptides, they were widely understood to be purchased for personal use. And like peptides, they've attracted increasing FDA enforcement attention.
Multiple versions of the SARMs Control Act have been introduced in Congress since 2018, most recently in 2019 as S.2895. If passed, the legislation would classify SARMs as Schedule III controlled substances alongside anabolic steroids, making their possession without a prescription a federal crime. While the SARMs Control Act hasn't passed as of early 2026, its repeated introduction signals Congressional interest in closing the research chemical loophole for performance-enhancing substances.[6]
No equivalent legislation has been introduced specifically targeting peptides, but the FDA's increased enforcement activity suggests the agency doesn't need new laws to act. It can already classify research peptides as unapproved drugs under existing authority.
Figure 3: The legality spectrum for peptide products. Research chemicals occupy a gray area that depends heavily on marketing, intended use, and FDA enforcement priorities.
Compounding Pharmacy Laws
Pharmacy compounding represents the only legal pathway for patients to access peptides that aren't FDA-approved as finished drug products. The rules governing compounding are complex, and recent FDA actions have significantly narrowed which peptides can be compounded.
What Is Pharmacy Compounding?
Compounding is the practice of creating customized medications tailored to individual patient needs. A compounding pharmacy might combine active ingredients, change a drug's dosage form (converting a tablet to a liquid, for example), remove allergens, or prepare a medication in a strength not commercially available. Compounding has been a fundamental part of pharmacy practice for centuries and serves patients whose medical needs can't be met by commercially manufactured drugs.
For peptides, compounding allows licensed pharmacies to prepare specific peptide formulations when a licensed prescriber writes a prescription for an individual patient. This is the mechanism through which patients have legally accessed peptides like BPC-157, CJC-1295, ipamorelin, thymosin alpha-1, and - until recently - compounded versions of semaglutide and tirzepatide.
503A vs. 503B: Two Distinct Legal Frameworks
The Drug Quality and Security Act (DQSA) of 2013, passed in response to the deadly New England Compounding Center meningitis outbreak, established two distinct categories of compounding facilities under the Federal Food, Drug, and Cosmetic Act:[7]
503A Traditional Compounding Pharmacies
Section 503A pharmacies are traditional compounding pharmacies that prepare medications based on individual patient prescriptions. Key requirements include:
- Must hold a valid state pharmacy license
- Must compound based on receipt of a valid prescription for an individually identified patient
- Can only use bulk drug substances that are components of FDA-approved drugs, have USP monographs, or appear on the FDA's 503A bulk drug substance list
- Cannot compound drugs that are "essentially copies" of commercially available products (with limited exceptions during drug shortages)
- Cannot advertise or promote specific compounded drugs
- Are primarily regulated by state boards of pharmacy
- Are exempt from FDA cGMP requirements and premarket approval
503B Outsourcing Facilities
Section 503B outsourcing facilities are a newer category created by the DQSA. These facilities can produce larger quantities of compounded drugs without patient-specific prescriptions, but face more stringent requirements:
- Must register with the FDA as an outsourcing facility
- Must comply with FDA cGMP requirements
- Subject to FDA inspection on a risk-based schedule
- Can distribute compounded drugs without patient-specific prescriptions (to hospitals, clinics, and healthcare facilities)
- Can only use bulk drug substances on the FDA's 503B bulk drug substance list or active ingredients in FDA-approved drugs
- Must report adverse events to the FDA
- Must label products with specific information including the compounding facility's name and the statement "This is a compounded drug"
| Feature | 503A Pharmacy | 503B Outsourcing Facility |
|---|---|---|
| Prescription required? | Yes, patient-specific | No (can compound in anticipation of orders) |
| FDA registered? | Not required | Required |
| cGMP compliance? | Exempt | Required |
| FDA inspection? | Not routine (state-regulated) | Risk-based schedule |
| Batch compounding? | Limited | Permitted |
| Distribution scope | Individual patients | Healthcare facilities and patients |
| Adverse event reporting | Not required federally | Required |
| Primary regulator | State board of pharmacy | FDA |
The Bulk Drug Substance Categories
Central to peptide compounding legality is whether a specific peptide appears on the FDA's bulk drug substance list. The FDA uses a category system to classify nominated bulk drug substances:
Category 1: Can Be Compounded
Substances in Category 1 have been evaluated and determined not to raise significant safety concerns. Both 503A and 503B pharmacies can use these substances in compounding. Examples of peptides meeting these criteria include NAD+ and sermorelin.[8]
Category 2: Previously Restricted (Now Eliminated)
Category 2 previously designated substances with "safety concerns" that could not be used in compounding. In October 2023, the FDA placed 17 peptides into this category, including BPC-157, TB-500 (thymosin beta-4 fragment), AOD-9604, and several others. This was highly controversial because the safety concerns cited were largely theoretical rather than based on documented adverse events.
Following legal challenges and a settlement, the FDA agreed to remove several peptides from Category 2 and submit them for review by the Pharmacy Compounding Advisory Committee (PCAC). The FDA subsequently eliminated Category 2 and Category 3 entirely in its revised guidance, though this doesn't mean all previously restricted peptides are now available for compounding.[9]
The Current Framework
Under the FDA's revised interim guidance released in January 2025, the system has been simplified. 503A pharmacies can compound using substances that:
- Are active ingredients in FDA-approved drugs
- Have a United States Pharmacopeia (USP) monograph
- Appear on the 503A bulk drug substance list
- Are placed in Category 1 of the interim bulk drug substance list
Substances without USP monographs, like most peptides of interest in the wellness and anti-aging spaces, cannot be compounded until the FDA completes its review process. This effectively creates a bottleneck: peptides must go through PCAC review and FDA rulemaking before they can be legally compounded.[10]
CLINICAL CONTEXT
For patients currently using compounded peptides, the regulatory changes mean access depends on timing, location, and the specific compound. Some peptides that were previously available through compounding pharmacies are no longer legally compoundable. Others remain in a gray area pending PCAC review. Patients should consult with their prescribing physicians about the current status of their specific peptide medications. For more on available compounds, visit the FormBlends Science Portal.
The PCAC Review Process
The Pharmacy Compounding Advisory Committee (PCAC) is a federal advisory committee that evaluates bulk drug substances for potential inclusion on the 503A compounding list. In 2024, the PCAC held two significant meetings to review popular peptides:
October 29, 2024 Meeting
The PCAC reviewed ipamorelin acetate, ipamorelin (free base), ibutamoren mesylate, L-theanine, and kisspeptin-10 for potential inclusion on the 503A bulk drug substance list. The committee voted against ipamorelin-related substances, finding insufficient evidence of safety for compounding use.[11]
December 4, 2024 Meeting
The PCAC reviewed AOD-9604, CJC-1295 (in multiple salt forms), CJC-1295 with Drug Affinity Complex (DAC), thymosin alpha-1 acetate, and thymosin alpha-1 (free base). The committee voted against all of these substances. This was a significant blow to the compounding peptide industry, as CJC-1295, thymosin alpha-1, and AOD-9604 were among the most widely compounded peptides.[12]
For research on CJC-1295 and ipamorelin, as well as thymosin alpha-1, FormBlends provides detailed compound profiles covering the available scientific literature.
The GLP-1 Compounding Crisis
The most dramatic compounding story of 2024-2025 involved GLP-1 receptor agonists. During shortage periods, compounding pharmacies were legally permitted to produce versions of semaglutide and tirzepatide. This created a booming market as demand for these weight-loss medications far outstripped the supply of branded products.
The timeline of events:
- 2022-2023: FDA places semaglutide and tirzepatide on the drug shortage list, legally enabling compounding
- October 2, 2024: FDA resolves the tirzepatide shortage, starting a 60-day wind-down for 503A pharmacies and 90-day wind-down for 503B facilities
- February 21, 2025: FDA resolves the semaglutide shortage, triggering similar wind-down periods
- September 2025: FDA sends more than 50 warning letters to GLP-1 compounders and manufacturers
- Early 2026: Novo Nordisk and Eli Lilly send cease-and-desist letters to compounding pharmacies, weight loss clinics, medical spas, and prescribing physicians
The enforcement crackdown has been aggressive. Warning letters targeted not just pharmacies but also clinics, telehealth platforms, and individual practitioners prescribing compounded GLP-1 medications. The FDA specifically objected to marketing language describing compounded products as "generic versions" or claiming they contain the "same active ingredient" as approved brands.[13]
For current information on GLP-1 peptide access and research, the FormBlends GLP-1 Research Center provides updated analysis.
Figure 4: The dual compounding framework under the DQSA. 503A pharmacies serve individual patients with prescriptions, while 503B outsourcing facilities can produce larger batches under direct FDA oversight.
Recent FDA Enforcement Actions
FDA enforcement against peptide sellers has escalated dramatically since 2022, with warning letters, import alerts, and seizure actions increasing year over year. The trend reflects a deliberate policy shift toward greater oversight of the peptide market.
Warning Letter Trends
The chart below illustrates the sharp increase in FDA warning letters directed at peptide sellers between 2019 and 2024:
FDA Warning Letters to Peptide Sellers by Year
Several factors drove this acceleration:
- The GLP-1 boom: Massive consumer demand for semaglutide and tirzepatide brought unprecedented attention to the compounding industry, drawing FDA scrutiny
- Social media visibility: Peptide sellers became increasingly brazen with marketing on platforms like Instagram, TikTok, and YouTube, making enforcement easier to justify
- Safety incidents: Reports of adverse events from contaminated or improperly formulated compounded peptides provided justification for increased oversight
- Manufacturer pressure: Brand-name drug manufacturers (particularly Novo Nordisk and Eli Lilly) aggressively lobbied for enforcement to protect their market share
Major Enforcement Actions: 2023-2025
The Category 2 Ban (October 2023)
In October 2023, the FDA placed 17 peptides into Category 2 of the interim bulk drug substance list, designating them as substances with "safety concerns" that could not be used in compounding. The affected peptides included:[14]
- BPC-157 (Body Protection Compound-157)
- AOD-9604 (Advanced Obesity Drug)
- Thymosin Beta-4 (TB-500)
- Dihexa
- Epitalon
- GHK-Cu (when nominated for injection)
- Ibutamoren (MK-677)
- KPV
- MOTS-c
- Pentadecapeptide BPC-157
- PT-141 (Bremelanotide, for compounding - the FDA-approved version Vyleesi remains available)
- Selank
- Semax
- SS-31 (Elamipretide)
- Thymalin
- Thymosin Alpha-1 (when not for approved indications)
- VIP (Vasoactive Intestinal Peptide)
This action was highly controversial. Industry groups, practitioners, and patient advocates argued that the FDA had unilaterally banned compounds with extensive use histories and limited documented safety concerns. The Alliance for Pharmacy Compounding and other organizations challenged the FDA's authority to create Category 2 without the full rulemaking process, including PCAC review and public comment.
For research profiles of several affected peptides, including BPC-157, FormBlends maintains detailed compound pages.
The Settlement (September 2024)
Following legal challenges, the FDA reached a settlement in September 2024. The agency agreed to:
- Remove five peptides (AOD-9604, CJC-1295, ipamorelin acetate, thymosin alpha-1, and Selank acetate) from Category 2 based on the nominators' withdrawal
- Submit these peptides for formal PCAC review rather than banning them unilaterally
- Schedule public PCAC meetings to evaluate the substances
However, as described in the previous section, the PCAC subsequently voted against all of these peptides at its October and December 2024 meetings, largely affirming the FDA's initial safety concerns.[15]
GLP-1 Warning Letters (September 2025)
In September 2025, the FDA issued more than 50 warning letters to companies involved in compounding or manufacturing GLP-1 medications. These letters targeted:
- Compounding pharmacies still producing semaglutide or tirzepatide after the shortage resolution
- Companies using unapproved salt forms (like semaglutide sodium or tirzepatide sodium) that the FDA considers different drugs from the approved products
- Companies making false or misleading claims about compounded GLP-1 products, such as calling them "generic" versions of Ozempic or Mounjaro
- Companies selling GLP-1 products without prescriptions or through direct-to-consumer channels without appropriate medical oversight
Research Chemical Enforcement (2024-2025)
Beyond compounding, the FDA also increased enforcement against research chemical sellers. The warning letter to Summit Research Peptides in December 2024 was representative of a broader campaign targeting companies that sold peptides online with research-only disclaimers while clearly marketing to consumers seeking human-use products.[16]
State-Level Enforcement
Federal enforcement has been complemented by state actions. The Ohio Board of Pharmacy, for example, issued summary suspension orders and settlement agreements in 2024 involving clinics and distributors where peptide products - including AOD-9604, BPC-157, and ipamorelin - were identified. Other state boards have taken similar actions, though the intensity and focus varies significantly by state.[17]
Cease-and-Desist Campaign by Manufacturers
In addition to government enforcement, pharmaceutical manufacturers have pursued their own legal actions. In early 2026, Novo Nordisk and Eli Lilly escalated enforcement efforts by sending cease-and-desist letters targeting the entire supply chain for compounded GLP-1 medications. These letters went to:
- Compounding pharmacies
- Weight loss clinics
- Medical spas (medspas)
- Individual prescribing physicians
- Telehealth platforms
The letters demanded immediate cessation of all compounding, distribution, and dispensing of GLP-1 medications, threatening patent infringement litigation and referral to the FDA for regulatory action.[18]
ENFORCEMENT TREND
The combination of federal enforcement, PCAC negative votes, shortage resolution, and manufacturer cease-and-desist campaigns represents a coordinated tightening of the regulatory environment for peptides. Practitioners and patients should stay informed about the current status of specific compounds. Court filings indicate the FDA plans to publish a final rule on several reviewed peptides no later than March 14, 2027.
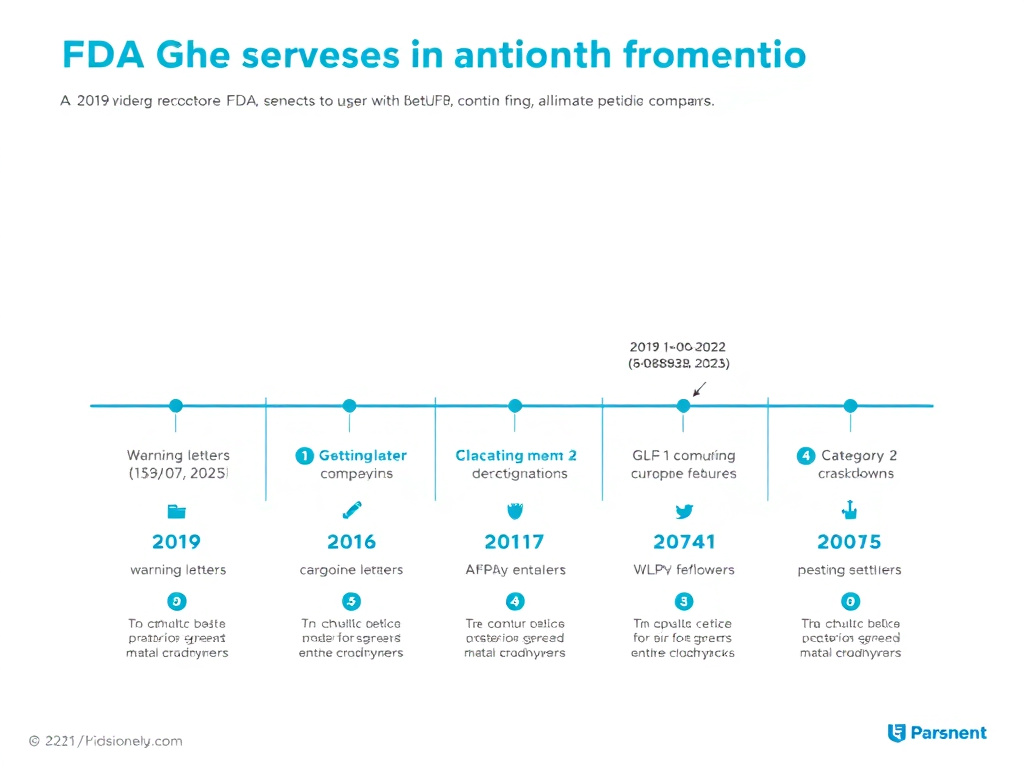
Figure 5: Timeline of major FDA enforcement actions affecting the peptide market, 2019-2025. The pace of enforcement has accelerated dramatically since 2023.
State-by-State Variations
While federal law sets the floor for peptide regulation, individual states can and do impose additional requirements through their boards of pharmacy, medical boards, and state legislatures. These variations create a patchwork of rules that differ based on geography.
How State Regulation Overlaps with Federal Law
The relationship between state and federal peptide regulation follows a general principle: states can be more restrictive than federal law but generally cannot be less restrictive. The FDA sets minimum standards, and state boards of pharmacy, medical boards, and legislatures can add requirements on top.
Key areas where states exercise independent regulatory authority include:
- Pharmacy licensing: Every state has its own board of pharmacy with authority to license and discipline pharmacies, including compounding pharmacies
- Prescriber requirements: State medical boards determine which types of providers can prescribe peptides (physicians, nurse practitioners, physician assistants, naturopathic doctors)
- Compounding oversight: Some states impose additional requirements beyond federal 503A/503B rules
- Telehealth prescribing: States vary widely on whether peptides can be prescribed via telehealth and under what conditions
- Controlled substance scheduling: States can schedule substances that aren't federally scheduled
- Consumer protection: State attorneys general can pursue enforcement under state unfair and deceptive practices laws
State Regulatory Profiles
While a comprehensive 50-state analysis is beyond the scope of any single report, the following profiles highlight states with distinctive approaches to peptide regulation:
Florida
Florida has become a hub for peptide therapy clinics, partly due to its relatively permissive regulatory environment for wellness and anti-aging practices. The state has a large concentration of compounding pharmacies and has historically taken a hands-off approach to peptide-related enforcement. However, the Florida Board of Medicine has increased scrutiny of clinics offering peptide therapies without appropriate medical oversight, and the state follows federal restrictions on compounding specific peptides.
Texas
Texas has been at the center of legal battles over GLP-1 compounding. The Northern District of Texas was the venue for challenges to the FDA's shortage resolution determination for semaglutide and tirzepatide. The Texas State Board of Pharmacy follows federal compounding guidelines but has its own inspection and enforcement protocols. In April and June 2025, the U.S. District Court for the Northern District of Texas denied attempts to reverse the FDA's shortage determination and dismissed the compounding pharmacy case with prejudice.[19]
California
California tends toward stricter regulation. The California State Board of Pharmacy has additional compounding requirements beyond federal minimums, including more stringent sterile compounding standards. California law also gives broader enforcement authority to the state attorney general for consumer protection actions related to health products. Research peptide sales face greater scrutiny in California compared to many other states.
New York
New York has strict pharmacy and supplement laws. The New York State Education Department's Office of the Professions oversees pharmacy licensing and has taken enforcement actions against compounding pharmacies operating outside regulatory bounds. New York also has strong consumer protection laws that can be applied to peptide sales.
Ohio
Ohio stands out for aggressive enforcement by its Board of Pharmacy. In 2024, the board issued summary suspension orders targeting clinics and distributors involved with peptide products including AOD-9604, BPC-157, and ipamorelin. Ohio's approach signals that state boards can act quickly against peptide-related violations even without waiting for federal enforcement.[20]
Arizona
Arizona has a more permissive environment for integrative and naturopathic medicine, with naturopathic doctors having broader prescribing authority than in many states. This creates additional pathways for patients to access peptide therapies through licensed practitioners. However, Arizona still follows federal compounding restrictions.
Tennessee
Tennessee's approach balances access with oversight. The state allows compounding pharmacies to operate with standard federal compliance but has increased inspection frequency for facilities producing injectable compounded products, including peptides.
| State | Regulatory Approach | Compounding Stance | Telehealth Peptide Rx | Notable Actions |
|---|---|---|---|---|
| Florida | Moderate | Follows federal | Permitted with restrictions | High clinic density; increased medical board scrutiny |
| Texas | Moderate | Follows federal | Permitted | Court battles over GLP-1 compounding |
| California | Strict | Above federal minimums | Strict requirements | Enhanced sterile compounding standards |
| New York | Strict | Above federal minimums | Strict requirements | Active consumer protection enforcement |
| Ohio | Aggressive enforcement | Follows federal | Permitted | Summary suspensions of clinics/distributors |
| Arizona | Permissive | Follows federal | Broadly permitted | Naturopathic prescribing authority |
| Colorado | Moderate | Follows federal | Permitted | Active integrative medicine community |
| Minnesota | Moderate to strict | Additional state requirements | Permitted with restrictions | State-level compounding oversight framework |
Prescriber Scope of Practice Variations
Who can prescribe peptides varies significantly by state. In most states, MDs and DOs have unrestricted prescribing authority for legally available medications, including compounded peptides. But the picture gets more complicated for other provider types:
- Nurse Practitioners (NPs): In about 25 states, NPs have full practice authority and can prescribe peptides independently. In other states, they need collaborative agreements with physicians
- Physician Assistants (PAs): Most states allow PAs to prescribe under physician supervision, but the degree of required supervision varies
- Naturopathic Doctors (NDs): Prescribing authority varies dramatically. Some states (like Arizona, Oregon, and Washington) grant NDs significant prescribing authority. Others don't license NDs at all
- Chiropractors: Generally cannot prescribe medications, though some states allow limited supplement recommendations
PRACTICAL GUIDANCE
Patients seeking peptide therapy should verify that their practitioner is licensed in their state and has appropriate prescribing authority for the specific type of peptide being prescribed. State medical board and pharmacy board websites typically provide license verification tools and scope of practice information. For help getting started with peptide research, visit FormBlends' Getting Started Guide.
Detailed State Regulatory Analysis
While the previous section provided an overview of state variations, certain states merit deeper examination because of their outsized impact on the national peptide market, their precedent-setting enforcement actions, or their unique regulatory frameworks.
Florida: The Peptide Therapy Capital
Florida has emerged as the de facto center of the peptide therapy industry in the United States, and this isn't coincidental. Several factors contribute to Florida's prominence:
Regulatory environment: Florida's Board of Medicine has historically taken a lighter regulatory touch on aesthetic and wellness medicine compared to states like California or New York. The state's medical board focuses its enforcement resources primarily on clear patient harm rather than proactively regulating emerging therapies. This has created an environment where practitioners feel more comfortable offering peptide therapy without fear of board action.
Practitioner density: Florida has a high concentration of anti-aging and integrative medicine practitioners, many of whom trained at organizations like the American Academy of Anti-Aging Medicine (A4M) headquartered in Boca Raton. These practitioners have been early adopters of peptide therapy and have built practices around it.
Compounding pharmacy infrastructure: Several major compounding pharmacies specializing in peptides are based in Florida, creating a convenient supply chain for local practitioners. The state's pharmacy board has licensed numerous compounding facilities that serve both local and national markets.
Patient demographics: Florida's large retiree population creates strong demand for anti-aging and wellness therapies, including peptide treatments for age-related conditions.
However, Florida's regulatory approach is evolving. The Florida Board of Medicine has increased scrutiny of clinics offering peptide therapies, particularly those operating without adequate physician oversight or those prescribing peptides that have been removed from the compounding list. The state attorney general's office has also pursued consumer protection actions against companies making unsubstantiated health claims about peptide products.
Texas: Legal Battleground
Texas has been at the center of the most significant legal battles over peptide compounding in the nation. The Northern District of Texas became the primary venue for challenges to the FDA's peptide compounding restrictions and shortage determinations.
Key legal proceedings:
- Outsourcing Facilities Association v. FDA: Compounding industry groups challenged the FDA's authority to restrict compounding of specific peptides without following proper administrative procedures
- GLP-1 shortage challenges: Multiple lawsuits filed in the Northern District of Texas attempted to reverse the FDA's determination that semaglutide and tirzepatide shortages had been resolved. In April and June 2025, the court denied these challenges and dismissed the cases with prejudice
- Constitutional challenges: Some litigants have argued that the FDA's compounding restrictions violate the Commerce Clause, Due Process Clause, or exceed the agency's statutory authority
The Texas State Board of Pharmacy follows federal compounding guidelines but maintains its own inspection and discipline processes. Texas law requires compounding pharmacies to be licensed by the state board and to comply with Texas Administrative Code provisions that, in some areas, exceed federal requirements for sterile compounding.
Texas also has a relatively permissive scope of practice framework. The state allows naturopathic doctors limited prescribing authority, and nurse practitioners with appropriate collaborative agreements can prescribe peptide therapies. The Texas Medical Board has not issued specific guidance on peptide therapy but evaluates complaints on a case-by-case basis.
California: Strictest in the Nation
California consistently ranks among the most restrictive states for compounding and peptide access. The California State Board of Pharmacy imposes additional requirements beyond federal minimums in several areas:
Sterile compounding: California's sterile compounding regulations, codified in the California Business and Professions Code and implementing regulations, impose requirements for environmental monitoring, personnel qualifications, and quality assurance that exceed both federal standards and USP chapter 797 (the pharmacopeial standard for sterile compounding). Pharmacies compounding sterile peptide preparations must meet these enhanced standards.
Inspection frequency: The California Board of Pharmacy conducts more frequent inspections of compounding pharmacies than most other states, and its inspection protocols are more detailed. Pharmacies that compound sterile preparations, including injectable peptides, face the most intensive oversight.
Consumer protection: California's Unfair Competition Law (Business and Professions Code Section 17200) gives the state attorney general and district attorneys broad authority to pursue companies selling peptide products with misleading claims. California courts have been receptive to these cases, and settlements can include substantial civil penalties.
Practitioner restrictions: California's Medical Practice Act and associated regulations impose specific requirements for prescribing and administering medications, including compounded peptides. The Medical Board of California has investigated practitioners whose prescribing patterns suggest commercial motivation rather than patient-centered care.
Research chemical enforcement: California has been more aggressive than most states in pursuing research chemical sellers. The state's Department of Justice has participated in joint enforcement actions with the FDA targeting companies that sell research peptides to California consumers.
New York: Parallel Strictness with Different Emphasis
New York matches California in regulatory strictness but emphasizes different aspects of enforcement:
Professional licensing: New York's Education Law places pharmacy licensing under the State Education Department's Office of the Professions rather than an independent pharmacy board. This creates a different bureaucratic structure that can be slower to act but more thorough when it does.
Compounding oversight: New York follows USP compounding chapters closely and has supplemented them with state-specific requirements, particularly for sterile compounding. The state conducted a major enforcement sweep of compounding pharmacies following the New England Compounding Center meningitis outbreak in 2012, and has maintained heightened oversight since.
Online sales: New York's Internet System for Tracking Over-Prescribing (I-STOP) primarily targets controlled substances but creates a surveillance infrastructure that could be extended to other drug categories. The state has also pursued enforcement against online sellers shipping peptide products to New York consumers without proper pharmaceutical licensing.
Emerging State Trends
Several trends are emerging at the state level that could significantly affect peptide access:
- Naturopathic licensing expansion: More states are considering licensing naturopathic doctors with prescribing authority, which could expand practitioner access to peptide therapy. States currently considering naturopathic licensing include Connecticut, Massachusetts, and Michigan
- Telehealth-specific legislation: States are enacting laws specifically addressing telehealth prescribing, some of which create more permissive frameworks for non-controlled substance prescribing (including most peptides)
- Compounding pharmacy bill of rights: Some states are considering legislation that would explicitly protect patient access to compounded medications, potentially including peptides, from FDA restrictions
- Interstate compounding compacts: Discussions are underway in several states about creating interstate compacts for compounding pharmacy licensing, which would simplify the process for pharmacies that serve patients in multiple states
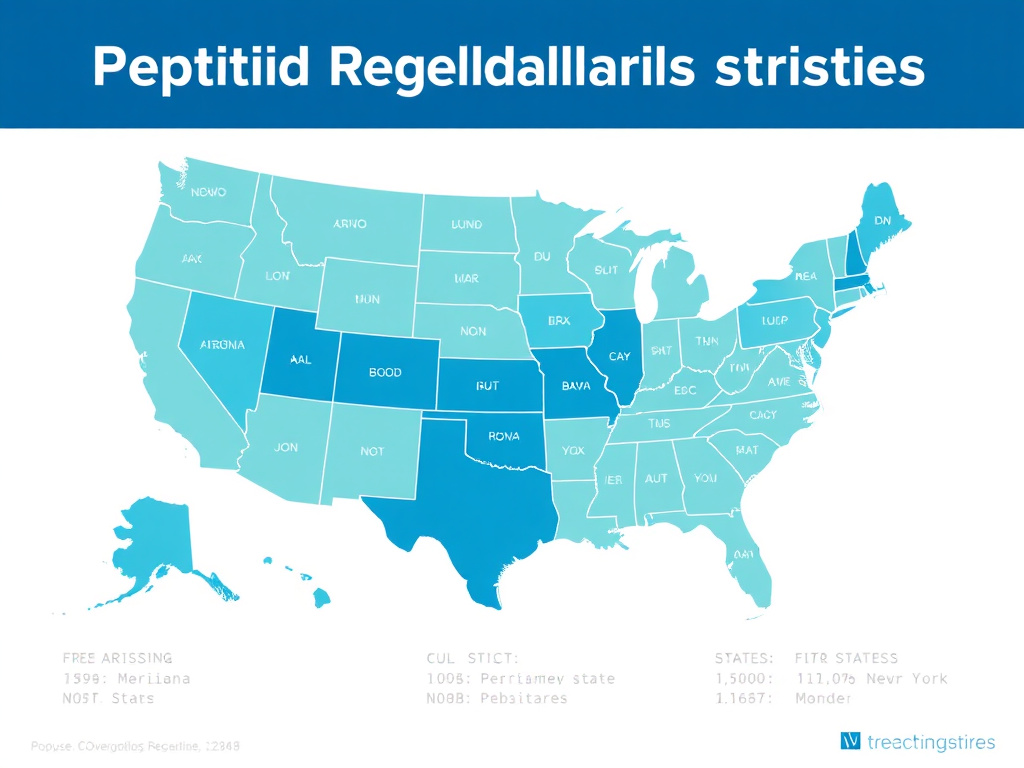
Figure 9: State regulatory environment for peptide therapy. Colors range from green (more permissive) to red (more restrictive), based on compounding oversight, prescriber scope of practice, and enforcement activity.
Telehealth and Prescribing
Telehealth has transformed how patients access peptide therapies, enabling consultations with specialized providers regardless of geography. But the legal framework for prescribing peptides via telehealth involves an evolving patchwork of federal and state rules.
The Ryan Haight Act and Controlled Substances
The Ryan Haight Online Pharmacy Consumer Protection Act of 2008 is the primary federal law governing online prescribing. It requires an in-person medical evaluation before a practitioner can prescribe controlled substances via the internet. This law was designed to combat "pill mills" that prescribed opioids and other controlled substances without proper medical evaluation.[21]
For peptide prescribing, the Ryan Haight Act is relevant primarily when dealing with substances that are federally scheduled. Most peptides are not controlled substances under federal law (with exceptions like certain growth hormone formulations). This means that for most peptide prescriptions, the Ryan Haight Act's in-person examination requirement doesn't apply.
However, the Act still matters for the peptide market because:
- Some states have enacted their own telehealth prescribing restrictions that go beyond the Ryan Haight Act and apply to non-controlled substances
- The DEA's COVID-era telemedicine flexibilities have influenced the broader telehealth prescribing culture, including for non-controlled substances
- If the SARMs Control Act or similar legislation passes and schedules certain peptides, the Ryan Haight Act's in-person requirement would apply to those compounds
COVID-Era Telemedicine Flexibilities
During the COVID-19 public health emergency, the DEA suspended the Ryan Haight Act's in-person requirement, allowing practitioners to prescribe controlled substances via telemedicine without a prior in-person visit. These flexibilities have been extended multiple times:
- March 2020: Initial suspension of in-person requirement during the public health emergency
- November 2024: Third temporary extension through December 31, 2025
- December 2025: Fourth temporary extension through December 31, 2026
The Biden administration also proposed rules in January 2025 for a Special Registration for Telemedicine that would create a permanent pathway for telehealth prescribing of controlled substances, replacing the temporary COVID flexibilities.[22]
How Telehealth Peptide Prescribing Works in Practice
The typical telehealth peptide prescribing process follows this general flow:
- Patient intake: Online questionnaires collecting medical history, current medications, symptoms, and goals
- Provider consultation: Video or audio call (sometimes asynchronous review) with a licensed prescriber
- Lab work: Many legitimate telehealth peptide providers require baseline lab work before prescribing, including hormone panels, metabolic panels, and relevant biomarkers
- Prescription: If appropriate, the provider writes a prescription to a compounding pharmacy
- Dispensing: The compounding pharmacy fills the prescription and ships it to the patient
- Follow-up: Periodic check-ins, lab reviews, and dose adjustments
Legal Requirements for Telehealth Peptide Prescribing
Legitimate telehealth peptide prescribing requires compliance with several overlapping requirements:
- Provider licensing: The prescriber must be licensed in the state where the patient is located at the time of the consultation (not the provider's home state)
- Standard of care: The telehealth consultation must meet the same standard of care as an in-person visit. This means adequate medical history, appropriate examination (even if limited by the telehealth format), informed consent, and proper documentation
- State telehealth requirements: Each state has its own telehealth laws governing provider-patient relationships, consent, and prescribing authority
- Pharmacy compliance: The compounding pharmacy must be licensed in the state where it ships medications and must comply with all applicable 503A or 503B requirements
- Medical necessity: Prescribing peptides without a legitimate medical indication can constitute unprofessional conduct and subject the prescriber to board discipline
Red Flags in Telehealth Peptide Prescribing
Not all telehealth peptide services operate within legal bounds. Warning signs that a telehealth service may be operating outside the law include:
- No real medical consultation (questionnaire-only prescribing without provider interaction)
- No lab work requirements before or during treatment
- Prescribing to patients in states where the provider isn't licensed
- Selling peptides directly rather than sending prescriptions to licensed pharmacies
- Advertising specific peptides as "available" before any medical evaluation
- No follow-up or monitoring protocols
- Prescribing compounded versions of peptides that are no longer legally compoundable
KEY DISTINCTION
There's a meaningful difference between a telehealth service that connects patients with licensed providers who evaluate their medical history and prescribe appropriate peptide therapy, and a website that essentially sells peptides with a rubber-stamp "prescription" attached. The former can be fully legal. The latter exposes everyone involved - the platform, the prescriber, and potentially the patient - to regulatory and legal consequences.
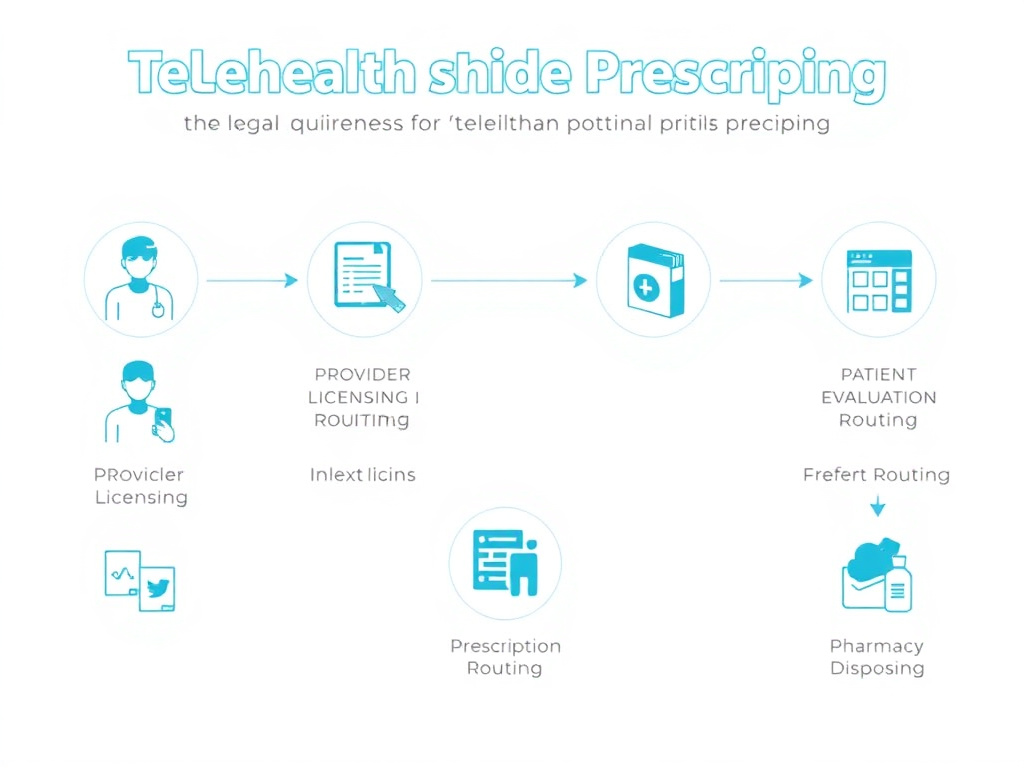
Figure 6: Legal telehealth peptide prescribing involves multiple compliance checkpoints. Each step has specific regulatory requirements that vary by state.
Import and International Law
International peptide regulation varies dramatically by country. What's legally available over the counter in one nation may be a controlled substance in another. And importing peptides across borders adds another layer of regulatory complexity.
United States Import Rules
U.S. Customs and Border Protection (CBP) works with the FDA to enforce drug import restrictions. Under FDA regulations, it is generally illegal to import unapproved drugs into the United States, even for personal use. However, the FDA exercises enforcement discretion under its Personal Importation Policy, which allows limited quantities of unapproved drugs to be imported under certain conditions:[23]
- The product is for the individual's own use (typically a 90-day supply or less)
- The product is not commercialized or promoted in the U.S.
- The product does not represent an unreasonable health risk
- The individual can affirm in writing that the product is for personal use
- The product is not available domestically, or the individual is continuing treatment started in a foreign country
This policy is discretionary, not a legal right. CBP and FDA agents can seize imported peptides at their discretion. Enforcement tends to focus on larger quantities, commercial shipments, and known controlled substances, but personal-use quantities are not immune from seizure.
International Regulatory Frameworks
United Kingdom
In the UK, peptides are regulated under the Medicines and Healthcare products Regulatory Agency (MHRA). Peptides marketed for therapeutic use must be authorized as medicines, similar to FDA approval in the US. The UK's Misuse of Drugs Act 1971 classifies certain hormones and their synthetic equivalents as controlled substances. Human growth hormone (HGH), for example, is a Class C controlled substance under UK law.
Research peptides can be purchased in the UK, but selling them for human consumption without proper authorization violates the Human Medicines Regulations 2012. The UK generally follows a similar enforcement pattern to the US: focusing on sellers rather than individual buyers, but with the same theoretical legal exposure for both parties.[24]
Australia
Australia has some of the strictest peptide regulations in the world. The Therapeutic Goods Administration (TGA) classifies many peptides as follows:
- Schedule 4 (Prescription Only): Many therapeutic peptides require a prescription and can only be supplied by a registered pharmacist
- Schedule 8 (Controlled Drug): Growth hormone and some other peptide hormones are classified as controlled drugs with strict prescribing requirements
- Schedule 9 (Prohibited): Certain substances are completely prohibited
Importing peptides into Australia without proper authorization can result in seizure at customs and potential criminal penalties. Under the Customs Act 1901 and Therapeutic Goods Act 1989, importing Schedule 4 substances without authorization can result in fines exceeding AUD $200,000 for individuals. Criminal prosecution is possible for large-scale or repeated importation.
Australia does maintain a Personal Importation Scheme that allows individuals to import some therapeutic goods for personal use under certain conditions, including having a valid prescription. However, this scheme has limitations, and not all peptides are eligible.[25]
Canada
Health Canada restricts peptide sales for human use unless the products are approved for specific therapeutic purposes through the Canadian drug approval process. Research peptides can be purchased for legitimate laboratory use, but the regulatory framework mirrors the US approach: the "research use only" disclaimer doesn't protect sellers when products are clearly marketed for human consumption.
Canada's Food and Drugs Act and associated regulations govern the import, manufacture, and sale of drugs, including peptides intended for therapeutic use. Health Canada has issued warnings about specific peptide products sold online without proper authorization.
European Union
EU member states regulate peptides through the European Medicines Agency (EMA) for centralized approvals and through national regulatory agencies for country-specific authorizations. The EU's pharmaceutical framework requires marketing authorization for any product presented as having therapeutic properties or intended to treat or prevent disease.
Research chemical sales exist within the EU, but enforcement varies by country. Germany tends toward stricter enforcement, while some Eastern European countries have more permissive environments for research chemical sales.
China
China is a major source of bulk peptide manufacturing for the global research chemical market. Chinese regulations nominally require proper licensing for pharmaceutical production, but enforcement has been inconsistent for companies selling research-grade peptides for export. The quality of Chinese-manufactured research peptides varies widely, and buyers face risks related to purity, contamination, and accurate peptide identification.[26]
WADA Prohibited List and Sports
The World Anti-Doping Agency (WADA) maintains a list of prohibited substances that applies to athletes in competition and, for some substances, out of competition as well. The 2025 WADA Prohibited List, which took effect January 1, 2025, includes multiple categories relevant to peptides:
S2: Peptide Hormones, Growth Factors, Related Substances and Mimetics
This category prohibits:
- Growth hormone (GH) and its releasing factors: Including GH-releasing hormone (GHRH) and its analogs (CJC-1295, sermorelin, tesamorelin), GH secretagogues (ghrelin and ghrelin mimetics like ipamorelin, ibutamoren/MK-677, anamorelin, macimorelin), and GH-releasing peptides (GHRPs including GHRP-1, GHRP-2, GHRP-6, alexamorelin, hexarelin)
- Insulin-like growth factors (IGFs): Including IGF-1 and its analogs
- Erythropoietins (EPOs) and agents affecting erythropoiesis
- Other peptide hormones and related substances: Including BPC-157, thymosin beta-4, AOD-9604, and others
S1: Anabolic Agents
Selective Androgen Receptor Modulators (SARMs) fall under this category, including andarine, ostarine (enobosarm), LGD-4033 (ligandrol), RAD140, S-23, and YK-11.[27]
Consequences for Athletes
Athletes who test positive for prohibited peptides face:
- Potential ban from competition (typically 2-4 years for a first offense)
- Disqualification of results
- Return of prizes and medals
- Public disclosure of the violation
- Possible criminal charges in countries where doping is criminalized
Athletes should be aware that many "supplement" products contain undisclosed peptides or SARMs. USADA (the United States Anti-Doping Agency) and WADA both maintain resources for athletes to check whether specific substances are prohibited.
ATHLETE WARNING
Athletes subject to anti-doping testing should avoid all peptide products unless they have been specifically cleared by their sport's anti-doping authority. Even peptides marketed as supplements or "natural" products may contain prohibited substances. The consequences of a positive test can be career-ending, and strict liability applies - meaning the athlete is responsible regardless of whether the positive test was intentional or accidental.
The Drug Supply Chain Security Act (DSCSA)
The DSCSA, enacted as part of the same 2013 legislation that created the 503B outsourcing facility framework, establishes requirements for electronically tracking and tracing prescription drugs throughout the U.S. supply chain. While primarily affecting commercial pharmaceutical distribution rather than compounding pharmacies, the DSCSA has implications for peptide regulation:
- Manufacturers, wholesalers, and dispensers must maintain and share transaction information for prescription drugs
- By 2025-2026, full electronic interoperability for package-level tracking is required
- The DSCSA helps identify and prevent the distribution of counterfeit, stolen, or contaminated drugs in the legitimate supply chain
Compounding pharmacies are generally exempt from DSCSA requirements for compounded drugs. However, 503B outsourcing facilities that distribute large quantities of compounded drugs may face some DSCSA requirements depending on how their distribution model is structured.[28]
Figure 7: International comparison of peptide regulation. Australia and certain EU countries have the strictest frameworks, while enforcement varies significantly in other regions.
Patient Rights and Access
The tightening regulatory environment for peptides raises real questions about patient access. Many patients have benefited from compounded peptide therapies, and the FDA's actions have disrupted treatment for some individuals with legitimate medical needs.
Right to Try and Compounding Access
The federal Right to Try Act, signed into law in 2018, allows terminally ill patients to access investigational drugs that have completed Phase I clinical trials. This law has limited applicability to peptides because most research peptides haven't entered the formal IND/clinical trial process at all. However, it establishes an important principle: patients have rights to access treatments when traditional pathways are insufficient.
For compounded peptides, patient access depends on:
- Whether the specific peptide remains legally compoundable (appears on the 503A or 503B bulk drug substance list or is a component of an FDA-approved drug)
- Whether a licensed prescriber is willing to write a prescription based on medical necessity
- Whether a licensed compounding pharmacy is willing and able to fill the prescription
- Whether the patient's state allows the specific type of prescribing (in-person vs. telehealth)
What Patients Can Do
Patients interested in peptide therapy who want to stay within legal bounds should consider the following steps:
- Work with a licensed provider: Find a physician, nurse practitioner, or other authorized prescriber who is knowledgeable about peptide therapy and licensed in your state
- Use licensed pharmacies: Ensure prescriptions are filled by a pharmacy that holds appropriate state and federal licenses for compounding
- Verify compound legality: Ask your prescriber to confirm that the specific peptide they're prescribing is currently legal to compound under federal and state law
- Maintain documentation: Keep copies of prescriptions, lab results, and provider communications in case of regulatory inquiries
- Stay informed: The regulatory landscape changes frequently. What's legal today may not be tomorrow, and vice versa
Advocacy and Policy Change
Several organizations are actively advocating for patient access to compounded peptides:
- The Alliance for Pharmacy Compounding has been a vocal opponent of the FDA's Category 2 designations and has participated in legal challenges
- The American Association of Naturopathic Physicians has advocated for continued access to compounded peptides
- Patient advocacy groups have organized around specific peptides, particularly BPC-157, thymosin alpha-1, and GLP-1 compounds
- The RFK Jr. appointment as HHS Secretary has introduced potential policy shifts, with indications that the new administration may take a different approach to peptide regulation than the Biden-era FDA[29]
PROVIDER CONSIDERATIONS
Clinicians prescribing peptide therapy should document their clinical rationale, obtain informed consent that includes discussion of the regulatory status of the prescribed compound, and maintain awareness of current FDA and state board positions. Board complaints and investigations related to peptide prescribing have increased alongside the broader enforcement trend. For comprehensive research resources, visit the FormBlends Peptide Research Hub.
The Compounding Pharmacy as Access Point
For patients with valid prescriptions, compounding pharmacies remain the primary legal access point for non-FDA-approved peptides. When evaluating a compounding pharmacy, patients and providers should consider:
- Accreditation: PCAB (Pharmacy Compounding Accreditation Board) accreditation indicates compliance with quality standards above regulatory minimums
- Testing: Quality pharmacies test finished preparations for potency, sterility, endotoxin levels, and particulate matter
- Transparency: Willingness to provide certificates of analysis (COAs) for bulk drug substances and finished products
- Licensing: Current state pharmacy license and, for 503B facilities, FDA registration
- Beyond-use dating: Appropriate assignment of expiration dates based on stability testing
For guidance on evaluating compounding pharmacies and understanding the compounding process, see the FormBlends Compounding Pharmacy Peptides Guide.
Figure 8: Patient access pathways for peptide therapy. The legal route involves a licensed prescriber, a valid prescription, and a licensed compounding pharmacy.
Specific Peptide Legal Status Reference
The legal status of individual peptides varies based on FDA approval status, bulk drug substance classification, controlled substance scheduling, and WADA prohibition status. This reference table summarizes the current status of the most commonly discussed peptides.
| Peptide | FDA Approved? | Compoundable? | WADA Prohibited? | Current Status Summary |
|---|---|---|---|---|
| Semaglutide | Yes (Ozempic/Wegovy/Rybelsus) | No (shortage resolved Feb 2025) | No | Available by prescription as branded product only |
| Tirzepatide | Yes (Mounjaro/Zepbound) | No (shortage resolved Oct 2024) | No | Available by prescription as branded product only |
| BPC-157 | No | No (Category 2 designation) | Yes | Available only as research chemical; not legally compoundable |
| TB-500 (Thymosin Beta-4) | No | No (Category 2 designation) | Yes | Available only as research chemical; not legally compoundable |
| CJC-1295 | No | No (PCAC voted against, Dec 2024) | Yes | PCAC rejected; awaiting final FDA rule (expected by March 2027) |
| Ipamorelin | No | No (PCAC voted against, Oct 2024) | Yes | PCAC rejected; awaiting final FDA rule |
| Thymosin Alpha-1 | No (approved in some countries) | No (PCAC voted against, Dec 2024) | No | PCAC rejected; not available through legal compounding |
| AOD-9604 | No | No (PCAC voted against, Dec 2024) | Yes | PCAC rejected; awaiting final FDA rule |
| Sermorelin | Previously (withdrawn from market) | Yes (Category 1) | Yes | Legally compoundable with valid prescription |
| Gonadorelin | Yes (Factrel) | Yes (active ingredient in approved drug) | Yes | Available as approved drug and compounded formulations |
| PT-141 (Bremelanotide) | Yes (Vyleesi) | Limited | No | Available as branded product; compounding restricted |
| GHK-Cu | No | Topical may be permissible; injectable restricted | No | Topical cosmetic use generally permitted; injectable restricted |
| NAD+ | No (as drug) | Yes (Category 1) | No | Legally compoundable with valid prescription |
| Selank | No | Pending (removed from Cat 2; under review) | No | Regulatory status in flux; previously Category 2 |
| Semax | No | No (Category 2 designation) | No | Not legally compoundable; available as research chemical |
| DSIP | No | No | No | Available only as research chemical |
| Epithalon | No | No (Category 2 designation) | No | Not legally compoundable; available as research chemical |
| MK-677 (Ibutamoren) | No | No (PCAC voted against, Oct 2024) | Yes | PCAC rejected; WADA prohibited; research chemical only |
| Kisspeptin-10 | No | No (PCAC voted against, Oct 2024) | No | PCAC rejected; available as research chemical |
| VIP | No | No (Category 2 designation) | No | Not legally compoundable; available as research chemical |
STATUS NOTE
This table reflects regulatory status as of March 2026 and is subject to change. The FDA's final rule on several PCAC-reviewed peptides is expected no later than March 2027. Ongoing litigation, administrative proceedings, and potential policy shifts under the current administration could alter the status of individual compounds. Always verify current status with legal counsel or regulatory authorities before making decisions based on this information.
Understanding the Legal Framework: A Deeper Analysis
Beyond the basic categories of approved, compoundable, and research chemical, the legal framework for peptides involves several nuanced concepts that practitioners, patients, and researchers should understand.
The "Essentially a Copy" Doctrine
Under 503A, compounding pharmacies cannot produce medications that are "essentially a copy" of a commercially available drug product. This rule exists to prevent compounders from competing directly with FDA-approved products. For peptide compounding, this means:
- When an FDA-approved version of a peptide is commercially available and not in shortage, pharmacies generally cannot compound that same peptide
- Minor formulation differences (different concentration, different route of administration, different inactive ingredients for a medically necessary reason) may allow compounding even when a commercial product exists
- During a drug shortage, the "essentially a copy" restriction is relaxed, which is why semaglutide and tirzepatide could be legally compounded during their shortage periods
The "essentially a copy" question becomes particularly complex with peptide salt forms. The FDA has taken the position that semaglutide sodium (a salt form used by some compounders) is a different drug from semaglutide (the base form in Ozempic), meaning compounders using the sodium salt aren't making a "copy" of the approved drug but rather an entirely different unapproved drug. This interpretation has been contested in litigation.[30]
USP Monographs and Their Significance
The United States Pharmacopeia (USP) publishes monographs that establish quality standards for drug substances and products. A USP monograph provides specifications for identity, strength, quality, purity, packaging, and labeling. For compounding pharmacies, the existence of a USP monograph for a bulk drug substance is one pathway to legal compounding under 503A.
Most research peptides lack USP monographs. This is because USP monograph development requires:
- A sponsoring organization or manufacturer willing to submit the monograph proposal
- Validated analytical methods for testing identity and purity
- Reference standards
- Stability data
- Public review and comment
The lack of USP monographs for most peptides of interest is a significant barrier to legal compounding. Even if the FDA were to take a more permissive approach to peptide compounding, the absence of pharmacopeial standards means there are no established quality benchmarks for compounded preparations.
The Role of the Prescriber
A valid prescription is essential for legal compounded peptide access. But what constitutes a "valid" prescription in the peptide context involves several considerations:
- Legitimate medical purpose: The prescription must be for a legitimate medical purpose and issued in the course of a bona fide prescriber-patient relationship
- Medical necessity: The prescriber should be able to articulate why the compounded peptide is medically necessary for the specific patient (as opposed to available commercial alternatives)
- Scope of practice: The prescriber must be authorized to prescribe the type of medication in question under their state license
- Documentation: Medical records should support the prescribing decision with appropriate history, examination, diagnosis, and treatment rationale
Prescribers who write peptide prescriptions without adequate medical justification face risks including medical board investigation, loss of prescribing privileges, and potential criminal liability if the prescribing pattern suggests commercial motivation rather than patient care.
Insurance Coverage and Cost Implications
Compounded peptides are rarely covered by insurance. Most health insurance plans exclude compounded medications, and even when they're technically covered, prior authorization requirements and formulary restrictions often prevent coverage. This means patients typically pay out of pocket for compounded peptide therapy.
The lack of insurance coverage creates a two-tier system where access to peptide therapy depends in part on the patient's ability to pay. A month's supply of compounded peptide therapy can range from $100 to $1,000+ depending on the specific peptide, dose, and pharmacy.
FDA-approved peptide drugs like semaglutide (Wegovy) and tirzepatide (Zepbound) may be covered by insurance for approved indications, but coverage is inconsistent and often requires prior authorization. The substantial price difference between branded and compounded versions of these drugs was a major driver of the compounding market during shortage periods.
Liability and Malpractice Considerations
Healthcare providers who incorporate peptide therapy into their practice should consider liability implications:
- Off-label use: Prescribing FDA-approved peptides for non-approved indications is legal but creates additional documentation requirements. Off-label prescribing is considered standard medical practice when supported by evidence, but providers should document their rationale and obtain informed consent
- Compounded medications: Using compounded peptides introduces additional liability considerations because these products haven't gone through FDA safety and efficacy review. Informed consent should address the regulatory status of the medication
- Research chemicals: Providers who administer or prescribe research chemical peptides (i.e., products not from licensed compounding pharmacies) face significant malpractice exposure and potential criminal liability
- Malpractice insurance: Some malpractice carriers exclude coverage for claims arising from the use of non-FDA-approved or non-compounded medications. Providers should verify their coverage before offering peptide therapy
PRACTICE TIP
Practitioners should develop a written informed consent specific to peptide therapy that addresses: (1) the FDA approval status of the specific peptide, (2) the regulatory status of the compounding pharmacy, (3) the off-label nature of the use (if applicable), (4) known risks and potential side effects, (5) the lack of long-term safety data for many peptides, and (6) available alternatives including FDA-approved options.
Detailed Analysis of Key Peptide Regulatory Categories
Different classes of peptides face distinct regulatory challenges. This section provides in-depth analysis of the regulatory status and legal considerations for the most commonly discussed peptide categories.
Growth Hormone Releasing Peptides (GHRPs) and Secretagogues
Growth hormone releasing peptides represent one of the most heavily regulated peptide categories. This group includes compounds like GHRP-2, GHRP-6, hexarelin, ipamorelin, and the non-peptide secretagogue MK-677 (ibutamoren). Their regulatory status reflects concerns about both safety and the potential for misuse.
FDA regulatory position: None of the GHRPs have FDA approval. Ipamorelin and MK-677 were both submitted for PCAC review in October 2024, and the committee voted against both. The FDA's primary concerns center on the potential for uncontrolled growth hormone elevation, effects on glucose metabolism, and the absence of long-term safety data from controlled clinical trials.
Compounding status: Following the PCAC votes, ipamorelin and MK-677 cannot be legally compounded. Other GHRPs (GHRP-2, GHRP-6, hexarelin) were not submitted for PCAC review and remain effectively prohibited from compounding because they lack USP monographs and don't appear on the bulk drug substance list.
Research chemical availability: GHRPs remain widely available as research chemicals from online suppliers. They're among the most popular peptides in the fitness and anti-aging communities, with extensive user experience and community knowledge despite limited formal clinical data.
WADA status: All GHRPs and growth hormone secretagogues are prohibited by WADA under category S2. This includes both peptide-based secretagogues (ipamorelin, GHRP-6) and non-peptide secretagogues (MK-677). Athletes using any of these compounds risk multi-year bans from competition.
Legal considerations: While GHRPs are not controlled substances, their regulatory trajectory suggests increasing restrictions. The PCAC votes effectively close the compounding pathway, and the FDA's enforcement against research chemical sellers signals less tolerance for the "research use only" market. Practitioners should be particularly cautious about prescribing or recommending GHRPs given this regulatory environment.
For detailed research on the CJC-1295/ipamorelin combination, one of the most commonly discussed GHRP protocols, visit the FormBlends CJC-1295/Ipamorelin research page.
GLP-1 Receptor Agonists
GLP-1 receptor agonists represent a unique situation in peptide regulation because they include both FDA-approved drugs and compounded versions whose legal status has changed dramatically.
FDA-approved GLP-1 drugs:
- Semaglutide: Approved as Ozempic (type 2 diabetes, 2017), Rybelsus (oral formulation, 2019), and Wegovy (obesity, 2021). Multiple additional approvals for cardiovascular risk reduction (2024) and other indications
- Tirzepatide: Approved as Mounjaro (type 2 diabetes, 2022) and Zepbound (obesity, 2023). Tirzepatide is a dual GIP/GLP-1 receptor agonist
- Liraglutide: Approved as Victoza (type 2 diabetes, 2010) and Saxenda (obesity, 2014)
- Dulaglutide: Approved as Trulicity (type 2 diabetes, 2014)
- Exenatide: Approved as Byetta (2005) and Bydureon (2012), both for type 2 diabetes
Compounding status: The legal basis for compounding GLP-1 medications existed during drug shortages under the "essentially a copy" exception. With both semaglutide and tirzepatide shortages resolved (October 2024 and February 2025 respectively), compounding is no longer legally permitted for these drugs.
The salt form controversy: Some compounding pharmacies have attempted to continue producing GLP-1 medications by using different salt forms, particularly semaglutide sodium. The FDA has taken the position that semaglutide sodium is a different drug from semaglutide base, meaning it's not covered by the approved product's regulatory framework and is therefore an unapproved new drug. This position has been contested in litigation, but as of March 2026, the FDA's view has been upheld by courts.
Access implications: With compounding no longer available, patients seeking GLP-1 therapy must use branded products. At list prices of $1,000-1,500+ per month, cost is a significant barrier. Insurance coverage is improving but remains inconsistent, with many payers imposing strict prior authorization requirements and limiting coverage to specific indications (typically type 2 diabetes or obesity with BMI above a certain threshold).
For comprehensive GLP-1 research information, including clinical trial data and mechanism of action, visit the FormBlends GLP-1 Research Center. Individual compound profiles are available for semaglutide and tirzepatide.
Healing and Regenerative Peptides
This category includes peptides primarily used for tissue repair, wound healing, and recovery from injury. The two most prominent compounds are BPC-157 and TB-500 (thymosin beta-4 fragment).
BPC-157:
Body Protection Compound-157 is a 15-amino-acid peptide derived from a larger protein found in human gastric juice. It has been the subject of extensive preclinical research, with studies demonstrating wound-healing, anti-inflammatory, and tissue-protective effects in animal models.
Regulatory status: BPC-157 was placed in Category 2 of the FDA's interim bulk drug substance list in October 2023, designating it as a substance with "safety concerns" that cannot be compounded. The FDA's concerns centered on the absence of human clinical trial data establishing safety, the potential for unknown adverse effects with systemic administration, and the lack of established dosing guidelines for human use.
The BPC-157 restriction was one of the most controversial aspects of the Category 2 designations. Proponents argue that BPC-157 has an extensive use history through compounding pharmacies with minimal documented adverse events, and that the preclinical safety profile is favorable. Critics of the ban point to the inconsistency of allowing compounding of substances with known toxicities while restricting a peptide with limited documented harm.
BPC-157 remains available as a research chemical. It's also prohibited by WADA, making it unavailable to competitive athletes through any channel. For current research on BPC-157, including preclinical data and mechanism of action, visit the FormBlends BPC-157 research page.
TB-500 (Thymosin Beta-4 Fragment):
Thymosin beta-4 is a 43-amino-acid peptide that plays a role in cell migration, angiogenesis, and wound healing. TB-500 is a synthetic fragment of thymosin beta-4 that retains the active region of the molecule. Like BPC-157, it was placed in Category 2 in October 2023 and is not legally compoundable.
RegeneRx Biopharmaceuticals has pursued FDA approval for thymosin beta-4-related products, but the development timeline has been protracted. The company's clinical programs have focused on specific wound-healing applications rather than the broad-spectrum regenerative use common in the peptide therapy community.
Immune-Modulating Peptides
Immune-modulating peptides, particularly thymosin alpha-1, have a complex regulatory history that illustrates the challenges of peptide regulation.
Thymosin Alpha-1:
Thymosin alpha-1 is a 28-amino-acid peptide that modulates immune function by enhancing T-cell maturation and function. It's approved in over 35 countries (marketed as Zadaxin) for treatment of hepatitis B and C, but has never been approved by the FDA. The disparity between its international availability and its US regulatory status is a common point of frustration for patients and practitioners.
The FDA initially placed thymosin alpha-1 in Category 2 in October 2023. Following the settlement, it was removed from Category 2 and submitted for PCAC review. At the December 4, 2024 PCAC meeting, the committee voted against thymosin alpha-1, finding insufficient evidence of safety for compounding use in the US market.
The PCAC's decision has been criticized on several grounds: the peptide has been approved and used in dozens of countries for over two decades, it has a substantial published safety record, and the PCAC appeared to apply a standard closer to NDA approval rather than the lower threshold appropriate for compounding authorization.
For research on thymosin alpha-1, including its international approval history and available clinical data, visit the FormBlends Thymosin Alpha-1 research page.
Antimicrobial Peptides
Antimicrobial peptides (AMPs) represent an emerging category that sits at the intersection of pharmaceutical development and peptide regulation. As antibiotic resistance becomes a growing global health concern, AMPs have attracted significant research interest for their ability to kill bacteria, viruses, and fungi through mechanisms that are less susceptible to resistance development than conventional antibiotics.
The regulatory status of AMPs varies depending on their intended application. AMPs being developed as pharmaceutical drugs (such as nisin analogs for bacterial infections or LL-37 for wound healing) follow the standard NDA pathway. AMPs used in food preservation may be regulated as food additives under 21 CFR Part 172 rather than as drugs. And AMPs incorporated into medical devices (wound dressings, catheter coatings) may fall under the medical device regulatory framework.
For the peptide therapy community, AMPs like LL-37 (a human cathelicidin) have attracted interest for their potential immunomodulatory effects beyond direct antimicrobial activity. LL-37 is available as a research chemical but is not on the FDA's bulk drug substance list for compounding. Its regulatory trajectory will depend on whether clinical data supports specific therapeutic applications and whether pharmaceutical companies pursue formal development.
The connection between antimicrobial peptide research and the broader peptide regulatory framework matters because AMPs could be among the next category of peptides to attract FDA enforcement attention as their therapeutic potential gains wider recognition and commercial interest grows.
Metabolic and Weight Management Peptides Beyond GLP-1
While GLP-1 receptor agonists dominate the weight management peptide conversation, several other peptide categories are relevant to metabolic health:
AOD-9604: Originally developed as an anti-obesity drug, AOD-9604 is a modified fragment of human growth hormone (amino acids 177-191) that appears to stimulate lipolysis without the growth-promoting effects of full-length GH. Despite early clinical trials that suggested modest weight loss effects, AOD-9604 never achieved FDA approval as a drug. It was submitted for PCAC review in December 2024 and the committee voted against it. AOD-9604 is also prohibited by WADA, which added it to the prohibited list in 2013 after Australian football players tested positive for it.
Tesamorelin: FDA-approved as Egrifta for reduction of excess abdominal fat in HIV patients with lipodystrophy, tesamorelin is a GHRH analog that stimulates growth hormone release. It's one of the few FDA-approved peptides used primarily for body composition purposes. As an approved drug, it can be prescribed for its approved indication or off-label by licensed practitioners. Compounded versions may be available when the commercial product is in shortage.
Peptide YY analogs: Peptide YY (PYY) is a naturally occurring gut hormone that suppresses appetite. Synthetic analogs are in clinical development as potential obesity treatments. While none are currently approved, they represent an active area of pharmaceutical research that could produce new approved peptide therapies in the coming years.
Amylin analogs: Pramlintide (Symlin) is an FDA-approved synthetic analog of amylin, a peptide hormone co-secreted with insulin from pancreatic beta cells. It's approved for type 1 and type 2 diabetes as an adjunct to insulin therapy. Newer amylin analogs are in clinical development, and the combination of amylin signaling with GLP-1 signaling is an active area of pharmaceutical research.
Neuroprotective and Cognitive Peptides
Peptides marketed for cognitive enhancement and neuroprotection occupy a particularly uncertain regulatory position:
- Selank: A synthetic analog of the immunomodulatory peptide tuftsin, developed in Russia and approved there for anxiety and cognitive enhancement. In the US, Selank was placed in Category 2, then removed as part of the September 2024 settlement. Its current status is in flux pending further regulatory review
- Semax: Another Russian-developed peptide, Semax is a synthetic analog of ACTH(4-10). It remains in Category 2 in the US and is not legally compoundable. It's available as a research chemical
- Dihexa: An angiotensin IV analog with preclinical evidence of cognitive-enhancing effects. It was placed in Category 2 and is not legally compoundable. Its research chemical availability has attracted attention from the nootropic community
- Epithalon (Epitalon): A synthetic tetrapeptide analog of the pineal gland peptide epithalamin, studied primarily in Russian research programs. Category 2 designation; not legally compoundable
The neuroprotective peptide category highlights a broader challenge: many of these compounds were developed and studied primarily in Russia, where regulatory standards and clinical trial methodologies differ from US requirements. The FDA has been skeptical of foreign clinical data for these compounds, contributing to their restrictive classification.
Anti-Aging and Longevity Peptides
The anti-aging peptide market has grown substantially, driven by consumer interest in compounds that may slow biological aging:
- NAD+ and its precursors: NAD+ (nicotinamide adenine dinucleotide) appears in Category 1 of the bulk drug substance list and can be legally compounded. Its precursors (NMN, NR) are more commonly sold as dietary supplements. The regulatory status of NAD+ has been relatively stable, making it one of the more accessible compounds in the anti-aging category
- GHK-Cu: Copper peptide GHK-Cu has a complex regulatory status. Topical formulations for cosmetic use are generally permissible and widely available. Injectable GHK-Cu, however, faces compounding restrictions under the Category 2 designation. The distinction between cosmetic and drug use is critical: the same molecule is legal in a skin cream but restricted as an injection
- MOTS-c: A mitochondria-derived peptide studied for metabolic effects. Category 2 designation; not legally compoundable
- Humanin: Another mitochondria-derived peptide with neuroprotective research. Not widely available even as a research chemical; minimal regulatory attention to date
The anti-aging peptide category is particularly susceptible to marketing claims that cross the line into drug claims. Companies selling "anti-aging" peptide products must be careful to make only structure/function claims (if selling oral supplements) or to operate through prescription channels (if selling compounded preparations). Claims about treating, preventing, or curing age-related diseases are drug claims that trigger FDA jurisdiction.
Sexual Health Peptides
Peptides marketed for sexual health have a mixed regulatory profile:
- PT-141 (Bremelanotide): FDA-approved as Vyleesi for hypoactive sexual desire disorder in premenopausal women. The approved product is available by prescription. Compounded versions face restrictions because an FDA-approved version exists (the "essentially a copy" doctrine)
- Kisspeptin-10: Submitted for PCAC review in October 2024; the committee voted against it. Research chemical availability continues
- Melanotan II: Not FDA-approved and not on the compounding list. Widely available as a research chemical, primarily marketed for tanning effects but also associated with sexual function effects. The FDA has issued warnings about melanotan II products
International Regulatory Analysis
Different countries take markedly different approaches to peptide regulation. Understanding these differences is essential for anyone involved in international peptide commerce, travel with peptide medications, or considering overseas treatment options.
European Union: The EMA Framework
The European Medicines Agency (EMA) administers a centralized authorization procedure for medicines marketed across the EU. For peptides:
Centralized procedure: Certain categories of medicines, including those derived from biotechnology processes, must go through the centralized procedure for EU-wide marketing authorization. This includes most therapeutic peptides. The centralized procedure results in a single marketing authorization valid in all EU member states.
National procedures: Some peptides can be authorized through national procedures in individual member states. This means a peptide might be legally available in Germany but not in France, creating within-EU variations.
Magistral and officinal formulas: The EU's equivalent of compounding falls under the magistral (individual patient) and officinal (pharmacopoeial) formula provisions. Each member state implements these provisions differently. Germany's Arzneimittelgesetz (Medicines Act) allows magistral preparation under strict conditions. France's pharmaceutical code similarly permits preparation magistrale. The UK, post-Brexit, follows its own MHRA regulations.
Research chemical sales: The EU's regulatory approach to research chemicals varies by country. Generally, EU member states follow the principle that substances marketed for therapeutic use are medicines regardless of labeling. Some countries, particularly in Eastern Europe, have less rigorous enforcement, creating a patchwork of availability.
Country-specific notes:
- Germany: Known for strict pharmaceutical regulation. The German Federal Institute for Drugs and Medical Devices (BfArM) actively monitors the peptide market and has taken enforcement action against sellers marketing peptides for human use without proper authorization
- Netherlands: Has historically been more permissive toward research chemical sales, though enforcement has increased in recent years
- Poland and Czech Republic: Have emerging markets for research peptides with varying levels of regulatory enforcement
- Spain: Follows EU-wide regulations but has been less aggressive in enforcement against online peptide sellers
Australia: The TGA's Strict Approach
Australia's Therapeutic Goods Administration (TGA) operates one of the world's strictest pharmaceutical regulatory systems. For peptides, this means:
Scheduling system: The TGA classifies substances into schedules under the Standard for the Uniform Scheduling of Medicines and Poisons (SUSMP). Relevant schedules for peptides include:
- Schedule 4 (Prescription Only Medicine): Includes most therapeutic peptides. Requires a prescription from a registered medical practitioner
- Schedule 8 (Controlled Drug): Includes growth hormone and some peptide hormones. Subject to additional prescribing restrictions, record-keeping requirements, and storage controls
- Schedule 9 (Prohibited Substance): Substances that may be abused or misused, available only for approved research under strict controls
Import restrictions: Australia has particularly strict customs enforcement for pharmaceutical imports. The Australian Border Force (ABF) routinely screens incoming packages for pharmaceutical products, and peptides are frequently seized. Penalties under the Customs Act 1901 include:
- Fines up to AUD $222,000 for individuals importing Schedule 4 substances without authorization
- Criminal prosecution for commercial-scale importation
- Seizure and destruction of imported products
The Personal Importation Scheme: The TGA does allow limited personal importation of therapeutic goods under certain conditions. For peptides, this typically requires a valid prescription from an Australian registered medical practitioner, importation of no more than a 3-month supply, and the product must be for the individual's personal use only. Even with these conditions met, border agents retain discretion to seize products.
Compounding in Australia: Australian compounding pharmacies operate under state and territory pharmacy legislation. The TGA's approach to compounding is more restrictive than the US system, with fewer compounding pharmacies, more limited compounding scope, and closer oversight. The concept of 503B outsourcing facilities doesn't exist in Australian pharmacy law.
Canada: Health Canada's Framework
Health Canada regulates peptides under the Food and Drugs Act and associated regulations:
Drug identification: Therapeutic products require a Drug Identification Number (DIN) or Natural Product Number (NPN) before they can be legally sold in Canada. Peptides marketed for therapeutic use without a DIN are considered unauthorized health products.
Compounding: Canadian pharmacy compounding is governed by provincial pharmacy legislation and the National Association of Pharmacy Regulatory Authorities (NAPRA) Model Standards for Pharmacy Compounding of Non-Sterile Preparations and Sterile Preparations. Canadian compounding regulations are generally comparable to US 503A requirements.
Online sales: Health Canada has issued warnings about specific peptide products sold online, particularly those marketed for weight loss or muscle building. The agency's enforcement has focused on products making health claims rather than those marketed purely as research chemicals.
Import regulations: Canadians can import medications for personal use under certain conditions, but the Canada Border Services Agency (CBSA) can seize products that appear to be unauthorized health products. The enforcement approach is somewhat less aggressive than Australia's but more active than the US FDA's personal importation policy.
Asia-Pacific Regional Approaches
Japan: Japan's Pharmaceuticals and Medical Devices Agency (PMDA) has a rigorous approval process similar to the FDA. Most therapeutic peptides require manufacturing and marketing approval. Japan does allow importation of personal-use medications but limits quantities and requires some products to be declared at customs.
South Korea: The Ministry of Food and Drug Safety (MFDS) regulates peptides similarly to conventional pharmaceuticals. South Korea has become an emerging market for peptide therapy, with several Korean pharmaceutical companies developing peptide products for both domestic and export markets.
Thailand: Thailand has a less restrictive pharmaceutical regulatory environment than most developed nations, making it a destination for "medical tourism" including peptide therapy. Thai clinics openly offer peptide treatments that would be legally complicated in the US or EU. However, product quality and medical oversight vary significantly.
India: India is a growing source of bulk peptide manufacturing, with several companies producing research-grade and pharmaceutical-grade peptides for export. Indian regulatory oversight of peptide manufacturing varies by state and facility, and quality can be inconsistent for products targeting the research chemical market.
Traveling with Peptides
Patients who use prescription peptide medications may need to travel with them. Key considerations include:
- Documentation: Always carry a copy of the prescription, a letter from the prescribing physician, and if possible, the original pharmacy label on the medication
- Syringes and needles: Carrying injection supplies can trigger additional scrutiny at security checkpoints. A physician's letter explaining the medical necessity can prevent problems
- Temperature control: Many peptides require refrigeration. Travel coolers with ice packs are essential for maintaining product stability
- Country-specific restrictions: Research destination countries' import rules before traveling. What's legal with a prescription in the US may be a prohibited substance in Australia, the UAE, or other countries with strict pharmaceutical import controls
- Quantity limitations: Most countries limit personal importation to a 90-day supply. Carrying larger quantities may be treated as commercial importation
TRAVEL ADVISORY
Athletes subject to anti-doping testing should be especially cautious about traveling with peptide medications. Even prescribed medications can trigger positive doping tests, and Therapeutic Use Exemptions (TUEs) are not automatically granted for peptides prohibited by WADA. Consult with your sport's anti-doping authority before traveling with any peptide medication.
The Future of Peptide Regulation
The peptide regulatory landscape is actively evolving. Several pending developments could significantly change what's legal, what's compoundable, and how patients access peptide therapies in the coming years.
Pending FDA Rulemaking
Court filings indicate that the FDA plans to publish a final rule regarding several PCAC-reviewed peptides no later than March 14, 2027. This rule will formally determine whether substances like CJC-1295, ipamorelin, thymosin alpha-1, and AOD-9604 can be used in compounding. Given that the PCAC voted against all of these substances, the final rule is expected to formally prohibit their compounding, though the rulemaking process includes a public comment period that could influence the outcome.[31]
Political and Administrative Changes
The regulatory direction could shift under the current administration. Robert F. Kennedy Jr.'s appointment as HHS Secretary has generated speculation about potential changes to FDA peptide policy. Kennedy has publicly expressed skepticism about FDA regulatory overreach and support for patient access to alternative therapies. While it's premature to predict specific policy changes, the possibility of a more permissive regulatory approach exists.
Any substantive changes would likely require either:
- New FDA guidance documents taking a different approach to peptide compounding
- Executive orders directing the FDA to reconsider its position
- Congressional legislation explicitly authorizing compounding of specific peptides
- Reversal of PCAC recommendations through the administrative process
Legislative Possibilities
Several legislative developments could affect peptide regulation:
- SARMs Control Act: If passed, this would create the first federal controlled substance scheduling specifically targeting performance-enhancing peptide-like compounds
- Compounding reform legislation: Bills have been proposed to expand access to compounded medications and limit the FDA's ability to restrict compounding of specific substances
- Telehealth prescribing laws: Permanent telehealth prescribing frameworks could affect how patients access peptide therapy remotely
- Right to Try expansions: Proposals to expand Right to Try beyond terminally ill patients could create new access pathways for peptide therapy
Industry Adaptation
The peptide industry is adapting to the changing regulatory environment in several ways:
- New drug applications: Some companies are pursuing formal FDA approval for popular peptides, recognizing that regulatory approval - while expensive - provides a clear legal pathway
- Clinical trials: Increased clinical trial activity for peptides like BPC-157 and thymosin beta-4 could eventually produce the safety and efficacy data needed for FDA approval or compounding authorization
- Alternative delivery methods: Oral, topical, and nasal peptide formulations are being developed that may face different regulatory requirements than injectables
- International markets: Some companies are pursuing regulatory approval in countries with less stringent requirements as a stepping stone to US market entry
What to Watch
Key developments to monitor include:
- The FDA's final rule on PCAC-reviewed peptides (expected by March 2027)
- Any HHS or FDA policy changes under the current administration
- Congressional action on the SARMs Control Act or compounding reform
- Ongoing litigation between compounding pharmacies and the FDA/brand-name manufacturers
- Clinical trial results for peptides currently in the research pipeline
- State-level regulatory changes that could expand or restrict access
The Potential Impact of AI on Peptide Regulation
An emerging factor in peptide regulation is the growing use of artificial intelligence in pharmaceutical development and regulatory compliance. AI could affect the peptide landscape in several ways:
Drug discovery acceleration: AI-driven drug discovery platforms are identifying novel peptide candidates with therapeutic potential at unprecedented speed. Machine learning models can predict peptide-target interactions, optimize amino acid sequences for stability and bioavailability, and identify potential toxicity issues before synthesis. If AI dramatically reduces the time and cost of peptide development, it could make FDA approval economically feasible for compounds that currently can't justify the investment.
Regulatory review acceleration: The FDA has signaled interest in using AI tools to accelerate regulatory review processes. AI-assisted review of clinical trial data, manufacturing documentation, and post-market surveillance could reduce review timelines and costs, potentially making the approval pathway more accessible for peptide developers.
Quality control and surveillance: AI systems could enhance post-market surveillance of compounded peptides by analyzing adverse event reports, identifying quality trends across compounding pharmacies, and flagging potential safety signals earlier than traditional monitoring methods. This could address some of the FDA's safety concerns about compounded peptides by providing better real-world safety data.
Enforcement targeting: AI tools could also help regulatory agencies identify non-compliant peptide sellers more efficiently. Natural language processing of websites, social media, and marketplace listings could automate the identification of products being marketed for human use despite research-only labeling.
The Role of Patient Advocacy in Regulatory Change
Patient advocacy has played an increasingly significant role in peptide regulation. The FDA's actions on compounded peptides have energized patient communities who feel their access to beneficial therapies is being restricted without adequate justification.
Organized advocacy efforts: Organizations like the Alliance for Pharmacy Compounding, patient groups focused on specific conditions treated with peptide therapy, and grassroots campaigns through social media have applied political pressure on Congress and the FDA. These advocacy efforts contributed to the settlement that forced the FDA to submit peptides for PCAC review rather than banning them unilaterally.
Congressional engagement: Patient advocates have engaged with members of Congress, resulting in Congressional inquiries to the FDA about its peptide compounding policies. Some members of Congress have expressed concern about the FDA's approach and have supported legislation to protect patient access to compounded medications.
Public comment participation: The FDA's rulemaking process includes public comment periods where patients, practitioners, and industry participants can submit feedback. The volume and quality of public comments on peptide-related rules has been substantial, and patient stories included in these comments have helped humanize the regulatory debate.
Legal support: Patient advocacy organizations have provided funding and organizational support for legal challenges to FDA peptide restrictions. These challenges have forced procedural changes even when they haven't changed the substantive outcome.
International Harmonization Efforts
The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) works to harmonize pharmaceutical regulatory standards across the US, EU, Japan, and other regions. ICH guidelines affect peptide regulation in several ways:
- ICH Q1A/Q1B: Stability testing guidelines that affect peptide shelf life assignments
- ICH Q5C: Quality of biotechnological products, relevant to peptide characterization
- ICH Q6B: Specifications for biotechnological/biological products, including peptides
- ICH M7: Assessment and control of mutagenic impurities, which can affect peptide synthesis process controls
As regulatory harmonization progresses, it could create more uniform international standards for peptide quality and regulation. This could simplify the regulatory pathway for companies seeking approval in multiple markets, potentially reducing development costs and expanding access to approved peptide therapeutics.
The Compounding Pharmacy Industry's Strategic Response
The compounding industry has responded to regulatory pressure through several strategic initiatives:
Quality improvement programs: Industry organizations have developed voluntary quality standards and best practices guidelines for peptide compounding, aiming to demonstrate that compounding pharmacies can produce safe, effective products without FDA-level manufacturing requirements.
Clinical evidence generation: Some compounding pharmacies and industry groups have begun sponsoring clinical research on compounded peptides, hoping to build the evidence base that the PCAC found lacking when it voted against several peptides. While pharmacy-sponsored research has limitations (including potential conflicts of interest), it could contribute to the safety and efficacy evidence needed for future regulatory submissions.
Technology investment: Compounding pharmacies have invested in advanced manufacturing and quality testing technologies, including HPLC systems, mass spectrometry, and environmental monitoring systems. These investments help demonstrate the industry's capacity to produce high-quality products and position pharmacies favorably for regulatory reviews.
Political engagement: The compounding industry has increased its political spending and lobbying activity, particularly around peptide-related issues. Industry PACs and trade associations have contributed to candidates sympathetic to compounding access and have hired lobbyists to advocate for favorable regulatory treatment.
Legal strategy development: Industry associations have developed comprehensive legal strategies for challenging FDA actions they view as overreaching. These strategies include both administrative challenges (through the FDA's own processes) and litigation (in federal courts). The settlement over Category 2 designations demonstrated that legal challenges can force procedural changes, even if they don't always change outcomes.
STAYING CURRENT
The FormBlends Peptide Research Hub maintains updated information on the regulatory status of individual peptides, along with the latest research findings. Bookmark this resource and check back regularly for updates as the regulatory landscape continues to evolve.
Quality, Safety, and Consumer Protection
The legal framework for peptides exists, at least in theory, to protect consumers from unsafe products. Understanding the quality and safety dimensions of peptide regulation helps explain why the FDA takes the positions it does - and where legitimate criticisms of those positions arise.
Manufacturing Quality: The cGMP Divide
One of the most significant differences between FDA-approved peptide drugs, compounded peptide preparations, and research chemical peptides is the manufacturing standard applied to each.
FDA-Approved Drug Manufacturing
Peptides manufactured as FDA-approved drugs must comply with current Good Manufacturing Practice (cGMP) requirements under 21 CFR Parts 210 and 211. These regulations cover every aspect of manufacturing:
- Facility design: Manufacturing areas must be designed to prevent contamination, with appropriate clean room classifications, HVAC systems, and environmental monitoring
- Personnel qualifications: Employees must be trained and qualified for their specific roles, with ongoing competency assessments
- Raw material testing: All incoming materials must be tested for identity, purity, and quality before use
- In-process controls: Manufacturing processes must include defined checkpoints and quality tests at critical stages
- Finished product testing: Each batch must be tested for identity, strength, quality, purity, and sterility (for injectable products) before release
- Stability testing: Ongoing stability programs must verify that products maintain their quality throughout their shelf life
- Documentation: Every step of manufacturing must be documented in batch records, and records must be retained for specified periods
- Validation: Manufacturing processes, equipment, cleaning procedures, and analytical methods must be validated to demonstrate they consistently produce the expected results
The cost of maintaining cGMP compliance is substantial, with estimates ranging from $50 million to $200 million for a new manufacturing facility. This cost is one reason why many peptides never reach FDA approval - the market simply doesn't justify the investment.
Compounding Pharmacy Standards
503A compounding pharmacies are exempt from FDA cGMP requirements but must comply with state pharmacy regulations and USP compounding standards:
- USP Chapter 795: Standards for non-sterile compounding, including ingredient quality, documentation, and beyond-use dating
- USP Chapter 797: Standards for sterile compounding (applicable to injectable peptides), covering environmental controls, personnel training, process validation, and testing requirements
- USP Chapter 800: Standards for handling hazardous drugs (applicable if the peptide is classified as hazardous)
- State-specific requirements: Many states impose additional requirements beyond USP standards
503B outsourcing facilities must comply with FDA cGMP requirements, bringing their manufacturing standards closer to those of conventional drug manufacturers. They face regular FDA inspections and must report adverse events.
The quality gap between cGMP-manufactured approved drugs and pharmacy-compounded preparations is a real concern. While many compounding pharmacies produce high-quality products, the absence of FDA oversight for 503A pharmacies means that quality can vary significantly. Studies have found that compounded sterile preparations sometimes fail potency tests, sterility tests, or contain particles that shouldn't be present.
Research Chemical Quality
Research chemical peptides are manufactured without any regulatory quality standard. There is no requirement for cGMP compliance, USP compliance, or any other quality framework. The quality of research peptides depends entirely on the manufacturer's own standards and the buyer's trust.
Independent testing of research chemical peptides has revealed significant quality concerns:
- Identity failures: Some products contain a different peptide than what's labeled, or contain a mixture of peptides
- Purity issues: Synthesis byproducts, incomplete peptide sequences, and chemical contaminants have been identified in research peptides
- Sterility concerns: Many research peptides are sold as lyophilized powders intended for reconstitution and injection, but the products may not have been manufactured or filled under sterile conditions
- Potency variation: The actual amount of active peptide may differ significantly from what's stated on the label, sometimes by 20-50% or more
- Endotoxin contamination: Bacterial endotoxins (pyrogens) are a serious concern for injectable products and can cause fever, hypotension, and organ failure. Research chemicals are typically not tested for endotoxin content
Adverse Event Reporting
The peptide regulatory framework creates different adverse event reporting obligations depending on the product type:
| Product Type | Adverse Event Reporting Required? | Reporting Mechanism |
|---|---|---|
| FDA-approved peptide drug | Yes (manufacturer and prescriber obligations) | FDA MedWatch; manufacturer reporting under 21 CFR Part 314 |
| 503B compounded peptide | Yes (facility obligation) | FDA MedWatch; direct reporting by outsourcing facility |
| 503A compounded peptide | Varies by state | State board of pharmacy; voluntary MedWatch |
| Research chemical peptide | No formal obligation | No established mechanism; voluntary MedWatch theoretically available |
The absence of adverse event reporting for research chemical peptides means there's no systematic way to track safety problems. If a batch of research BPC-157 causes infections or adverse reactions in users, there's no mechanism to identify the problem, alert other users, or trigger a recall. This is a genuine public health concern that the FDA has cited in its enforcement justifications.
Counterfeit and Adulterated Products
The peptide market - particularly the research chemical segment - faces a significant counterfeit problem. Common issues include:
- Complete fakes: Products containing no active peptide at all, just filler materials
- Substitution: Products containing a different (usually cheaper) peptide than what's labeled
- Dilution: Products containing much less active peptide than labeled, mixed with additional solvent or filler
- Contamination: Products containing active pharmaceutical ingredients (APIs) not listed on the label, sometimes including substances like sildenafil or other drugs
- Expired or degraded products: Products where the peptide has degraded due to improper storage or manufacturing, but are still sold as full-potency
Third-party testing services like Janoshik Analytical, ChromaDex, and others offer peptide analysis for consumers, but testing costs ($50-200 per sample) add to the already significant expense of research peptides. And even testing a single sample doesn't guarantee that the next vial from the same supplier will have the same quality.
The FDA's Safety Argument
The FDA's position on restricting peptide compounding rests primarily on safety concerns. The agency's arguments include:
- Insufficient safety data: Most compounded peptides have not undergone the rigorous safety evaluation required for FDA approval. While some have extensive literature support, the clinical trial evidence typically doesn't meet the standard required for an NDA
- Manufacturing risks: Compounded sterile preparations carry inherent risks that are mitigated by cGMP compliance but not eliminated. The 2012 NECC meningitis outbreak demonstrated what can happen when compounding quality fails
- Lack of standardization: Without USP monographs or approved drug specifications, there's no pharmacopeial standard against which to evaluate compounded peptide preparations
- Unknown drug interactions: Many compounded peptides lack formal drug interaction studies, creating potential risks for patients taking multiple medications
- Inadequate patient monitoring: Some peptide therapy protocols don't include appropriate monitoring for potential adverse effects, particularly when prescribed through telehealth platforms with minimal follow-up
The Counter-Argument
Critics of the FDA's restrictive approach make several compelling points:
- Extensive use history: Many compounded peptides have been used by thousands of patients over years or decades with minimal documented adverse events. The absence of reported problems, while not proof of safety, suggests a favorable risk profile
- Regulatory inconsistency: The FDA allows compounding of many substances with known toxicities (chemotherapy agents, for example) while restricting peptides with limited toxicity evidence. Critics argue this inconsistency suggests the FDA's motivations aren't purely safety-driven
- Access barriers: Restricting compounding doesn't eliminate demand. Patients who can't access compounded peptides through legal channels may turn to research chemicals with no quality controls at all, potentially creating greater safety risks than regulated compounding
- Economic considerations: Brand-name drug manufacturers have lobbied aggressively for compounding restrictions, leading critics to argue that economic interests, not safety concerns, drive enforcement priorities
- Patient autonomy: Some argue that informed patients should have the right to access therapies of their choosing, particularly when those therapies are prescribed by licensed physicians based on clinical judgment
BALANCED PERSPECTIVE
Both sides of the peptide regulation debate have legitimate points. The FDA's safety concerns are real - compounded products do carry risks that approved drugs don't. But the blanket approach of banning compounding of specific peptides, particularly when the safety concerns are largely theoretical, raises questions about proportionality. The ideal regulatory framework would allow informed access through qualified practitioners while maintaining meaningful quality standards. Whether the current framework achieves this balance is a matter of active debate.
The Economics of Peptide Regulation
Regulatory decisions about peptides don't happen in a vacuum. Economic forces - from pharmaceutical company revenues to compounding pharmacy margins to patient out-of-pocket costs - shape the regulatory landscape in ways that aren't always visible.
The GLP-1 Revenue Imperative
The FDA's aggressive enforcement against GLP-1 compounding can't be understood without considering the economics involved. In 2024, Novo Nordisk's Ozempic and Wegovy generated over $18 billion in U.S. revenue combined. Eli Lilly's Mounjaro and Zepbound added billions more. Compounded GLP-1 medications were capturing a meaningful share of this revenue at a fraction of the branded product cost.
A month's supply of branded semaglutide (Wegovy) carries a list price of approximately $1,350. Compounded semaglutide was available from compounding pharmacies for $200-400 per month. This price differential drove enormous consumer demand for compounded versions and created a financial incentive for compounding pharmacies to expand their GLP-1 operations.
The pharmaceutical manufacturers' cease-and-desist campaigns and their lobbying for FDA enforcement are consistent with standard business practices for protecting revenue. Whether this commercial motivation inappropriately influences FDA regulatory decisions is a contested question. The FDA maintains that its enforcement decisions are based on safety and legal authority, not commercial considerations. Critics point to the timing and intensity of enforcement as evidence that manufacturer lobbying plays a role.
The Compounding Industry Economics
The compounding pharmacy industry has grown significantly over the past decade, driven in part by demand for peptide preparations. Key economic factors include:
- Market size: The U.S. compounding pharmacy market is estimated at $12-15 billion annually, with sterile compounding (including peptides) representing a growing share
- Margin structure: Compounded medications typically offer higher profit margins than dispensing commercial products, making compounding an attractive business model for pharmacies
- Investment in infrastructure: Many compounding pharmacies have invested millions in clean rooms, equipment, and personnel to meet sterile compounding requirements. Regulatory changes that restrict compounding can strand these investments
- Employment: The compounding sector employs tens of thousands of pharmacists, pharmacy technicians, and support staff. Restrictions on peptide compounding affect livelihoods as well as patient access
Patient Cost Impact
Regulatory restrictions on peptide compounding directly affect patient costs. When a peptide can no longer be compounded:
- Patients with prescriptions for FDA-approved versions may face costs of $500-1,500+ per month depending on the specific drug and insurance coverage
- Patients using peptides without FDA-approved equivalents (like BPC-157, CJC-1295, or thymosin alpha-1) lose legal access entirely
- Patients who turn to research chemical alternatives sacrifice quality assurance and medical oversight
- Insurance coverage for compounded medications is rare, and coverage for FDA-approved peptide drugs is inconsistent
The cost dimension creates equity concerns. Wealthier patients can afford branded peptide drugs or out-of-pocket compounding costs. Less affluent patients may be priced out of legal access entirely, pushing them toward research chemicals or foregoing treatment.
The Research Pipeline Economics
Several companies are pursuing formal FDA approval for peptides that are currently available only as research chemicals or through compounding. These efforts face significant economic challenges:
- BPC-157: No company has publicly announced an IND filing for BPC-157 in the United States, despite extensive preclinical research. The challenge is defining a sufficiently large and specific indication to justify the $1-3 billion development cost
- Thymosin alpha-1: Approved in several countries outside the US (as Zadaxin), thymosin alpha-1 has been evaluated in US clinical trials but hasn't achieved FDA approval. The economic case for pursuing approval is complicated by the compound's patent status
- Thymosin beta-4: RegeneRx Biopharmaceuticals has pursued FDA approval for thymosin beta-4-related products, but the development timeline has been extended and the commercial pathway remains uncertain
The economic reality is that many peptides with genuine therapeutic potential may never achieve FDA approval because the investment required exceeds the expected return. This creates a tension between the regulatory framework's demand for formal approval and the practical impossibility of achieving it for every potentially beneficial compound.
THE CENTRAL TENSION
Peptide regulation reflects a fundamental tension in healthcare policy: the desire for safety through rigorous evaluation versus the desire for access to potentially beneficial treatments. The FDA's mandate prioritizes safety, which means some effective treatments will remain legally unavailable. Whether this tradeoff is appropriate depends on one's assessment of the relative risks of under-regulation versus over-regulation - a question that doesn't have a single correct answer.
Practical Compliance Guide for Practitioners
Healthcare practitioners who incorporate peptide therapy into their practice need practical guidance on staying within legal bounds. This section provides actionable compliance recommendations based on the current regulatory framework.
Step 1: Verify the Peptide's Legal Status
Before prescribing any peptide, practitioners should verify its current regulatory status:
- Check whether the peptide has FDA approval for any indication
- Check the FDA's interim bulk drug substance list to see if the peptide is in Category 1 (compoundable) or has been reviewed and rejected by the PCAC
- If the peptide was previously in Category 2, verify whether it has been removed and what its current classification is
- Check whether any drug shortage exception currently applies that would enable compounding of an otherwise restricted substance
- Verify state-specific restrictions that may go beyond federal rules
Step 2: Establish a Legitimate Prescriber-Patient Relationship
A valid prescription requires a legitimate prescriber-patient relationship, which generally means:
- The practitioner has conducted an adequate medical evaluation (in-person or via telehealth, depending on state requirements)
- The practitioner has reviewed the patient's medical history, current medications, and relevant lab work
- The practitioner has identified a legitimate medical indication for the peptide therapy
- The practitioner has obtained informed consent that addresses the regulatory status, known risks, and alternatives
- The practitioner has documented all of the above in the medical record
Step 3: Use Licensed Compounding Pharmacies
Practitioners should only send prescriptions to compounding pharmacies that:
- Hold current state pharmacy licenses in all states where they ship medications
- Are registered as 503B outsourcing facilities (if applicable)
- Can provide certificates of analysis (COAs) for their products
- Test finished preparations for potency, sterility, and endotoxins
- Have PCAB accreditation (preferred but not required)
- Are willing to answer questions about their quality control processes
Practitioners should never direct patients to purchase research chemical peptides and should never administer research chemicals in their practices. Doing so exposes the practitioner to malpractice liability, board discipline, and potential criminal prosecution.
Step 4: Implement Appropriate Monitoring
Peptide therapy should include monitoring protocols appropriate to the specific compound being prescribed:
- Baseline labs: Comprehensive metabolic panel, complete blood count, hormone panels relevant to the specific therapy, and any compound-specific biomarkers
- Follow-up labs: At defined intervals (typically 4-12 weeks depending on the compound) to assess efficacy and safety
- Clinical assessments: Regular check-ins (in-person or telehealth) to evaluate response, side effects, and continued medical necessity
- Documentation: Record all lab results, clinical observations, and treatment modifications in the patient chart
Step 5: Maintain Regulatory Awareness
Practitioners should stay current on regulatory changes by:
- Monitoring FDA announcements and guidance documents related to compounding and peptides
- Following state board of pharmacy and medical board communications
- Subscribing to legal updates from healthcare law firms that specialize in compounding regulation
- Participating in professional organizations that track peptide therapy regulation
- Consulting with healthcare attorneys when uncertain about the legality of specific prescribing practices
Step 6: Document Everything
Thorough documentation is both a legal protection and a professional obligation. Practitioners should document:
- The clinical rationale for prescribing each peptide
- The informed consent process, including discussion of the compound's regulatory status
- All lab orders, results, and clinical assessments
- The compounding pharmacy used and its licensing/accreditation status
- Any adverse events or unexpected outcomes
- The decision to continue, modify, or discontinue therapy at each follow-up
MALPRACTICE PROTECTION
Practitioners should review their malpractice insurance policies to verify coverage for peptide therapy claims. Some policies exclude claims arising from use of non-FDA-approved medications, compounded preparations, or off-label prescribing. If your policy has such exclusions, consider supplemental coverage or adjusting your practice accordingly. Discuss specific concerns with your malpractice insurance carrier or a healthcare attorney.
Red Lines: What Practitioners Should Never Do
Certain practices create unacceptable legal and professional risk:
- Never purchase research chemicals for patient use. This is practicing medicine with unregulated, unverified substances and exposes both the practitioner and the patient to serious risk
- Never prescribe peptides that are no longer legally compoundable. Once a peptide is removed from the bulk drug substance list or the PCAC votes against it, prescribing it for compounding creates liability
- Never prescribe without a legitimate medical evaluation. Rubber-stamp prescribing exposes the practitioner to board discipline and potential criminal charges
- Never make therapeutic claims about unapproved peptides in marketing materials. Marketing claims about disease treatment are drug claims that can trigger FDA enforcement
- Never ignore cease-and-desist letters. Whether from the FDA, a state board, or a pharmaceutical manufacturer, these letters require a considered legal response, not dismissal
For additional resources on peptide research and responsible clinical integration, practitioners can visit FormBlends Science Portal for peer-reviewed research summaries and compound profiles.
The Compounding Pharmacy Selection Guide
For patients and practitioners who decide to pursue peptide therapy through legally compoundable peptides, selecting the right compounding pharmacy is a critical decision that affects both safety and legal compliance.
Accreditation and Licensing Verification
The first step in evaluating a compounding pharmacy is verifying its credentials. Every compounding pharmacy should have:
State pharmacy license: This is the most basic requirement. The pharmacy must hold a current license from the state board of pharmacy in the state where it operates. For pharmacies that ship medications to patients in other states, they may also need non-resident pharmacy licenses in those states. State board websites typically provide online license verification tools.
DEA registration: If the pharmacy compounds controlled substances (which most peptide pharmacies don't, since most peptides aren't controlled substances), it needs DEA registration. Even pharmacies that don't compound controlled substances typically maintain DEA registration for their overall operations.
503B registration (if applicable): Outsourcing facilities must register with the FDA. The FDA maintains a searchable list of registered outsourcing facilities on its website. Registration indicates the facility is subject to FDA inspection and must comply with cGMP requirements.
PCAB accreditation: The Pharmacy Compounding Accreditation Board (PCAB) is a voluntary accreditation program that establishes quality standards above regulatory minimums. PCAB accreditation involves detailed evaluation of facilities, processes, personnel, and quality systems. While not required, PCAB accreditation is a strong indicator of quality commitment. As of 2026, fewer than 200 pharmacies nationwide hold PCAB accreditation.
Quality Testing Programs
High-quality compounding pharmacies implement testing programs that go beyond minimum regulatory requirements:
Potency testing: Verification that the finished product contains the correct amount of active ingredient. High-performance liquid chromatography (HPLC) is the standard analytical method for peptide potency testing. Pharmacies should be able to provide potency test results for specific batches upon request.
Sterility testing: For injectable peptide preparations, sterility testing is essential. This typically involves membrane filtration or direct inoculation methods per USP Chapter 71. Testing should be performed on a representative sample from each batch.
Endotoxin testing: Bacterial endotoxins (lipopolysaccharides from gram-negative bacteria) are a particular concern for injectable products. Even products that pass sterility testing can contain endotoxins from the manufacturing process. The Limulus Amebocyte Lysate (LAL) test or recombinant Factor C assay are standard methods for endotoxin detection.
Particulate matter testing: Injectable products must be free of visible particles and meet limits for sub-visible particulate matter. Light obscuration or microscopic methods per USP Chapter 788 are used for this testing.
Beyond-use dating: Quality pharmacies assign beyond-use dates (BUDs) based on stability testing data rather than defaulting to the minimum periods specified in USP Chapter 797. Extended BUDs require extended stability studies demonstrating the product maintains its quality throughout the assigned period.
Questions to Ask a Compounding Pharmacy
Practitioners and informed patients should feel comfortable asking compounding pharmacies the following questions:
- What is your source for bulk drug substances? Quality pharmacies source raw materials from FDA-registered suppliers with Drug Master Files. They should be able to provide certificates of analysis (COAs) for their raw materials
- Do you perform finished product testing? Ask specifically about potency, sterility, endotoxin, and particulate matter testing. Request copies of test results for specific batches if possible
- What are your clean room specifications? For sterile compounding, pharmacies should operate in ISO-classified clean rooms with appropriate environmental monitoring programs
- How do you assign beyond-use dating? Ask whether BUDs are based on stability testing data or default USP periods
- What is your adverse event reporting process? Even 503A pharmacies should have a process for documenting and investigating adverse events, even if federal reporting isn't required
- Are you willing to provide a tour of your facilities? Reputable pharmacies are typically willing to show practitioners their compounding operations. Reluctance to allow facility visits can be a red flag
- What happens if testing reveals an out-of-specification result? Quality pharmacies have written investigation and corrective action procedures for handling failed test results
Red Flags to Watch For
Certain characteristics should raise concerns about a compounding pharmacy:
- Unusually low prices: While compounding pharmacies compete on price, significantly below-market pricing may indicate cut corners on quality testing, raw material sourcing, or manufacturing processes
- No testing documentation: Pharmacies that can't or won't provide COAs or test results are not maintaining adequate quality assurance
- Direct-to-consumer marketing: 503A pharmacies are prohibited from advertising specific compounded drugs. Pharmacies that aggressively market specific peptide preparations may be operating outside their legal framework
- Compounding restricted peptides: If a pharmacy offers to compound peptides that are no longer on the bulk drug substance list (like BPC-157 or CJC-1295), it may be operating illegally
- No prescription requirement: Legitimate 503A pharmacies require valid prescriptions. Any pharmacy offering to dispense peptides without a prescription is operating outside the law
- Shipping from unexpected locations: If a pharmacy ships from a different state or address than its listed location, this may indicate unlicensed operations
For a comprehensive guide to evaluating compounding pharmacies, including additional quality indicators and selection criteria, visit the FormBlends Compounding Pharmacy Peptides Guide.
The Role of the Prescriber in Pharmacy Selection
Prescribers have both an ethical and practical interest in the quality of the pharmacies they use. From a malpractice perspective, prescribers who send prescriptions to pharmacies with known quality issues may face liability for adverse outcomes. From a patient care perspective, the prescriber's choice of pharmacy directly affects the quality of the medication the patient receives.
Best practices for prescribers include:
- Establishing relationships with two or three quality compounding pharmacies rather than relying on a single source
- Periodically reviewing pharmacy credentials, accreditation status, and inspection history
- Requesting COAs for products dispensed to their patients
- Documenting the pharmacy selection rationale in patient charts
- Promptly investigating and reporting any quality concerns or adverse events
Emerging Legal Issues and Unresolved Questions
The peptide regulatory landscape is evolving rapidly, and several legal questions remain unresolved. These emerging issues will shape the future of peptide access and regulation.
The First Amendment and Marketing Restrictions
Some legal scholars and industry advocates have raised First Amendment challenges to FDA restrictions on how compounding pharmacies can market their services. The argument draws on commercial free speech protections established in cases like Sorrell v. IMS Health (2011) and Thompson v. Western States Medical Center (2002).
In Thompson, the Supreme Court struck down a federal provision prohibiting compounding pharmacies from advertising their services, finding it violated the First Amendment. The decision established that restrictions on truthful, non-misleading commercial speech about compounding services must pass intermediate scrutiny under the Central Hudson test.
Current First Amendment arguments in the peptide context focus on:
- Whether the FDA's restrictions on marketing compounded peptides constitute impermissible content-based speech restrictions
- Whether pharmacies have a right to inform patients and practitioners about available compounded medications
- Whether the FDA's "intended use" doctrine, which determines product classification based partly on marketing statements, creates an unconstitutional chilling effect on commercial speech
These arguments haven't been definitively resolved by courts, and their outcome could significantly affect how peptides are marketed and accessed.
Interstate Commerce and the Dormant Commerce Clause
State-level variations in peptide regulation create potential Dormant Commerce Clause issues. When one state effectively restricts the shipment of legally compounded peptides from pharmacies in other states, it may be impermissibly burdening interstate commerce.
This issue is particularly relevant for:
- States that impose additional licensing requirements on out-of-state pharmacies shipping to their residents
- States that restrict which peptides can be compounded or dispensed more strictly than federal law requires
- The practical challenge of determining which state's laws apply when a telehealth provider in one state prescribes a peptide that's compounded in a second state and shipped to a patient in a third state
Patent Issues and Compounding
The intersection of patent law and compounding regulation creates additional complications. When Novo Nordisk and Eli Lilly send cease-and-desist letters to compounding pharmacies, they're partly relying on patent protection in addition to FDA regulatory arguments.
Key patent considerations include:
- Compound patents: If a specific peptide is covered by a composition-of-matter patent, compounding that peptide may constitute patent infringement even during a drug shortage
- Method patents: Patents on methods of treatment using specific peptides can be infringed by practitioners who prescribe and patients who use the compounded versions
- Formulation patents: Patents on specific formulations, delivery mechanisms, or dosage forms can be infringed by pharmacies that reproduce those formulations
- The Hatch-Waxman safe harbor: 35 USC Section 271(e)(1) provides a safe harbor for activities reasonably related to developing information for FDA submission, but this likely doesn't protect commercial compounding for patient use
The patent dimension adds financial risk for compounding pharmacies beyond the FDA regulatory risk. Patent litigation can be extremely expensive, and the threat of patent infringement suits has a chilling effect on compounding activity even when the regulatory pathway might otherwise permit it.
Data Privacy in Telehealth Peptide Prescribing
Telehealth peptide prescribing raises data privacy issues that haven't been fully addressed by existing regulations:
- HIPAA compliance: Telehealth platforms must comply with HIPAA privacy and security requirements. However, the proliferation of direct-to-consumer telehealth peptide services has created questions about whether all platforms maintain adequate HIPAA compliance
- State privacy laws: Some states have health privacy laws that go beyond HIPAA, creating additional compliance burdens for telehealth platforms operating across state lines
- Marketing data use: Some telehealth peptide platforms use patient data for marketing purposes in ways that may not be fully transparent to patients
- Third-party disclosure: The sharing of patient health information between telehealth platforms, compounding pharmacies, and payment processors creates potential privacy gaps
Environmental and Disposal Considerations
An often-overlooked legal issue involves the disposal of unused peptide medications. Federal and state environmental regulations govern pharmaceutical waste disposal, and improper disposal of injectable peptide products can violate these regulations. Healthcare facilities, pharmacies, and even individual patients have obligations regarding pharmaceutical waste that vary by jurisdiction.
Professional Liability Insurance Gaps
The evolving peptide regulatory landscape has created gaps in professional liability insurance coverage that practitioners should be aware of:
- Some malpractice policies exclude claims arising from "experimental" or "investigational" treatments, which could include compounded peptide therapy depending on the policy language
- Policies may exclude coverage for prescribing medications for off-label uses that aren't supported by peer-reviewed literature
- The use of research chemical peptides in clinical practice is almost certainly excluded from standard malpractice coverage
- Practitioners who own or have financial relationships with compounding pharmacies may face additional liability exposure and insurance considerations
Practitioners should review their malpractice policies carefully and consider supplemental coverage if their peptide therapy practice exposes them to risks not covered by their standard policy.
Case Studies in Peptide Enforcement
Examining specific enforcement cases provides concrete illustrations of how the regulatory framework operates in practice. These cases highlight the types of conduct that trigger enforcement and the consequences that follow.
Case Study 1: Summit Research Peptides Warning Letter (December 2024)
In December 2024, the FDA issued a warning letter to Summit Research Peptides (Warning Letter 695607), finding that the company's products were unapproved new drugs and misbranded despite being labeled "for research use only."
What triggered enforcement: The FDA examined Summit's website and found evidence that products were intended for human use, including product descriptions referencing human physiological effects, dosing information consistent with human administration, and marketing context suggesting consumer rather than laboratory use.
Key findings: The FDA determined that the company's "research use only" disclaimer did not overcome the totality of evidence indicating the products were intended as drugs for human use. The warning letter cited specific website content, product names, and contextual marketing materials.
Consequences: The company was given 15 business days to respond with a corrective action plan. Failure to comply could result in product seizure, injunctions, or criminal prosecution.
Lessons: This case demonstrates that the FDA looks beyond labels and disclaimers to determine intended use. Companies cannot insulate themselves from enforcement simply by adding a "not for human consumption" sticker to products that are clearly marketed for human use.
Case Study 2: The GLP-1 Compounding Crackdown (2024-2025)
The FDA's enforcement campaign against GLP-1 compounding represents the largest single regulatory action in the history of the peptide market.
Timeline of events:
- 2022-2023: Drug shortages of semaglutide and tirzepatide create legal basis for compounding
- Late 2023-2024: Compounding market for GLP-1 medications explodes, with hundreds of pharmacies entering the market
- October 2024: FDA resolves tirzepatide shortage; 60/90-day wind-down begins
- February 2025: FDA resolves semaglutide shortage; similar wind-down begins
- April-June 2025: Texas federal court dismisses challenges to shortage resolution
- September 2025: FDA issues 50+ warning letters to GLP-1 compounders
- Early 2026: Novo Nordisk and Eli Lilly send cease-and-desist letters throughout the supply chain
Scope of enforcement: The warning letters targeted not just pharmacies but also telehealth platforms, weight loss clinics, and individual practitioners. The FDA specifically objected to claims that compounded products were "generic" versions or contained the "same active ingredient" as approved drugs.
Lessons: This case illustrates how rapidly the regulatory landscape can shift. Pharmacies that invested heavily in GLP-1 compounding infrastructure found their legal basis evaporated when the shortages were resolved. It also demonstrates the power of branded pharmaceutical manufacturers to complement government enforcement with private legal action.
Case Study 3: Ohio Board of Pharmacy Peptide Actions (2024)
The Ohio Board of Pharmacy's enforcement actions in 2024 provide an example of state-level regulation in action.
What happened: The board issued summary suspension orders and settlement agreements targeting clinics and distributors where peptide products were identified, including AOD-9604, BPC-157, and ipamorelin.
Basis for action: Ohio's enforcement was based on state pharmacy law violations, including operating without proper licensure, dispensing prescription drugs without valid prescriptions, and distributing unapproved drug products.
Significance: Ohio's actions demonstrate that state boards can act independently of federal enforcement. Even if the FDA doesn't pursue a particular company, state regulators can impose their own penalties, including license suspension, fines, and practice restrictions.
Lessons: Compliance with federal law alone isn't sufficient. Practitioners and pharmacies must also comply with state-specific requirements, and state boards may be more aggressive than the FDA in pursuing certain types of violations.
Case Study 4: The Category 2 Settlement (September 2024)
The settlement between the FDA and industry groups over Category 2 peptide designations illustrates how legal challenges can shape regulatory outcomes.
Background: When the FDA unilaterally placed 17 peptides in Category 2 in October 2023, industry groups challenged the action as exceeding the FDA's authority. They argued that the agency was required to follow the PCAC review process before banning specific substances from compounding.
Settlement terms: The FDA agreed to remove five peptides from Category 2 and submit them for PCAC review. This was seen as a partial victory for the compounding industry because it forced the FDA to follow established procedures rather than acting unilaterally.
Outcome: However, the PCAC subsequently voted against all five substances, largely validating the FDA's initial safety concerns. The settlement process may have delayed the final regulatory determination by several years (the final rule isn't expected until March 2027), but it appears unlikely to change the substantive outcome.
Lessons: Legal challenges to FDA actions can force procedural changes and buy time, but they don't necessarily change the underlying regulatory trajectory. The PCAC review process gave industry a formal hearing but ultimately supported the FDA's position.
Case Study 5: International Peptide Import Seizures
While less publicized than domestic enforcement actions, U.S. Customs and Border Protection has conducted numerous seizures of imported peptide products. These seizures illustrate the import dimension of peptide regulation.
Common seizure scenarios:
- Packages from Chinese peptide manufacturers intercepted at international mail facilities and express consignment hubs
- Peptide shipments flagged by automated risk assessment systems based on package weight, declared value, origin country, and customs declarations
- Bulk peptide imports from countries with known research chemical manufacturing operations
- Individual packages containing multiple vials of lyophilized peptides with labels indicating pharmaceutical-grade quality
What happens after seizure: When CBP seizes a peptide shipment, the importer receives a Notice of Seizure and can either abandon the goods or contest the seizure through administrative proceedings. Contesting a seizure requires demonstrating that the products are legally importable, which is difficult for unapproved peptide drugs. Most individual importers choose to abandon the goods rather than incur legal costs contesting the seizure.
Import alert implications: The FDA can issue import alerts that allow for automatic detention of products from specific companies, countries, or product categories without physical examination. Import alerts related to unapproved drug products effectively create a blacklist that prevents specific importers from bringing peptide products into the country.
Trends: Seizure activity has increased in parallel with domestic enforcement. CBP has enhanced its targeting of pharmaceutical shipments from countries with significant research chemical manufacturing operations, and interdiction technology (including machine learning-based risk assessment) has improved the agency's ability to identify suspect shipments.
Case Study 6: The Telehealth Platform Enforcement Wave
Beginning in late 2024 and accelerating through 2025, the FDA and state regulators began targeting telehealth platforms that facilitated peptide prescribing without adequate medical oversight.
What triggered enforcement: Several telehealth platforms had adopted a model where patients could complete a brief online questionnaire, receive an automated or minimal physician review, and obtain prescriptions for compounded peptides with little genuine medical evaluation. This "prescribing mill" model drew regulatory attention for several reasons: the lack of meaningful physician-patient relationships, the absence of required lab work and monitoring, the prescribing of peptides for conditions that weren't medically documented, and the platforms' financial incentives to prescribe rather than decline patients.
Enforcement actions: State medical boards investigated prescribers associated with high-volume telehealth peptide platforms. The FTC examined advertising practices of platforms that guaranteed prescriptions. State attorneys general pursued consumer protection actions against platforms that charged consultation fees without providing genuine medical evaluation. And the FDA issued warning letters to platforms that sold compounded peptides directly (rather than routing prescriptions to independent pharmacies).
Lessons: Telehealth peptide prescribing can be legally compliant, but it must involve genuine medical evaluation, appropriate prescriber-patient relationships, and adequate monitoring. Platforms that reduce these requirements to increase volume and revenue face escalating regulatory risk. The enforcement trend suggests that regulators are increasingly willing to hold telehealth platforms accountable for the quality of care delivered through their systems, not just the technical legality of the prescriptions generated.
Figure 10: Key enforcement case studies illustrate how federal and state regulatory actions interact with industry legal challenges and manufacturer enforcement campaigns.
Frequently Asked Questions
References
- DiMasi JA, Grabowski HG, Hansen RW. Innovation in the pharmaceutical industry: new estimates of R&D costs. J Health Econ. 2016;47:20-33. doi:10.1016/j.jhealeco.2016.01.012
- U.S. Food and Drug Administration. Dietary Supplement Health and Education Act of 1994, Public Law 103-417. FDA. 1994. Available at: https://www.fda.gov/regulatory-information/laws-enforced-fda/dietary-supplement-health-and-education-act-1994
- U.S. Food and Drug Administration. Warning Letters - Compounding. FDA Inspections, Compliance, Enforcement, and Criminal Investigations. 2025. Available at: https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/compliance-actions-and-activities/warning-letters
- U.S. Food and Drug Administration. Intended Use - Questions and Answers. FDA Regulatory Information. 2024. Available at: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/intended-uses
- U.S. Food and Drug Administration. Summit Research Peptides - 695607 - 12/10/2024. Warning Letters. 2024. Available at: https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/warning-letters/summit-research-peptides-695607-12102024
- U.S. Congress. S.2895 - SARMs Control Act of 2019. Congress.gov. 2019. Available at: https://www.congress.gov/bill/116th-congress/senate-bill/2895
- U.S. Congress. Drug Quality and Security Act, Public Law 113-54. 113th Congress. 2013. doi:10.1016/j.clinthera.2014.05.063
- U.S. Food and Drug Administration. Interim Policy on Compounding Using Bulk Drug Substances Under Section 503A. FDA Guidance. 2025. Available at: https://www.fda.gov/media/174456/download
- Alliance for Pharmacy Compounding. Settlement Agreement - FDA Peptide Compounding. APC. 2024. Available at: https://www.a4pc.org
- National Community Pharmacists Association. FDA Releases Guidance for Compounding Pharmacies. NCPA Newsroom. 2025. Available at: https://ncpa.org/newsroom/qam/2025/01/13/fda-releases-guidance-compounding-pharmacies
- U.S. Food and Drug Administration. Pharmacy Compounding Advisory Committee Meeting Minutes - October 29, 2024. FDA Advisory Committees. 2024. Available at: https://www.fda.gov/advisory-committees/pharmacy-compounding-advisory-committee
- U.S. Food and Drug Administration. FDA PCAC Topic 1 December 4, 2024. FDA Media. 2024. Available at: https://www.fda.gov/media/185641/download
- Wilson Sonsini Goodrich & Rosati. FDA Sends Warning Letters to More Than 50 GLP-1 Compounders and Manufacturers. WSGR Insights. 2025. Available at: https://www.wsgr.com/en/insights/fda-sends-warning-letters-to-more-than-50-glp-1-compounders-and-manufacturers.html
- Frier Levitt LLC. Regulatory Status of Peptide Compounding in 2025. Frier Levitt Articles. 2025. Available at: https://www.frierlevitt.com/articles/regulatory-status-of-peptide-compounding-in-2025/
- Holt Law. Detailed look: Regulatory Status of Popular Compounded Peptides. DJ Holt Law. 2024. Available at: https://djholtlaw.com/deep-dive-regulatory-status-of-popular-compounded-peptides/
- Frier Levitt LLC. Peptides Under the Microscope: Recent FDA and State Enforcement Trends. Frier Levitt Articles. 2024. Available at: https://www.frierlevitt.com/articles/injectable-peptides-fda-state-enforcement-compounding/
- Ohio Board of Pharmacy. Enforcement Orders and Settlement Agreements. Ohio BOP. 2024. Available at: https://www.pharmacy.ohio.gov
- HCH Lawyers. Novo Nordisk, Eli Lilly Sending Cease-and-Desist Letters Over Compounded Semaglutide. HCH Law Blog. 2026. Available at: https://www.hchlawyers.com/blog/2026/february/novo-nordisk-eli-lilly-sending-cease-and-desist-/
- Pharmacy Times. Navigating FDA Compounding Regulations: Tirzepatide's Removal From the Shortage List. Pharmacy Times. 2024. Available at: https://www.pharmacytimes.com/view/navigating-fda-compounding-regulations-tirzepatide-s-removal-from-the-shortage-list
- SafeHG. FDA's Overreach on Compounded Peptides: Legal Battles and How Clinics Can Push Back. SafeHG. 2024. Available at: https://www.safehg.com/fdas-overreach-on-compounded-peptides-legal-battles-and-how-clinics-can-push-back/
- U.S. Congress. Ryan Haight Online Pharmacy Consumer Protection Act of 2008, Public Law 110-425. 110th Congress. 2008. Available at: https://www.congress.gov/110/plaws/publ425/PLAW-110publ425.htm
- U.S. Drug Enforcement Administration. Fourth Temporary Extension of COVID-19 Telemedicine Flexibilities. Federal Register. 2025. doi:10.1016/2025-24123
- U.S. Food and Drug Administration. Personal Importation Policy. FDA Import Program. Available at: https://www.fda.gov/industry/import-basics/personal-importation
- UK Medicines and Healthcare products Regulatory Agency. Human Medicines Regulations 2012. MHRA. 2012. Available at: https://www.legislation.gov.uk/uksi/2012/1916
- Australian Therapeutic Goods Administration. Personal Importation Scheme. TGA. 2024. Available at: https://www.tga.gov.au/products/unapproved-therapeutic-goods/access-pathways/personal-importation-scheme
- Cohen PA, Travis JC, Keizers PH, et al. Four experimental stimulants found in sports and weight loss supplements. Clin Toxicol. 2019;57(10):873-878. doi:10.1080/15563650.2019.1579396
- World Anti-Doping Agency. The 2025 Prohibited List. WADA. 2024. Available at: https://www.wada-ama.org/en/prohibited-list
- U.S. Food and Drug Administration. Drug Supply Chain Security Act (DSCSA). FDA. 2024. Available at: https://www.fda.gov/drugs/drug-supply-chain-integrity/drug-supply-chain-security-act-dscsa
- Frier Levitt LLC. FDA Peptide Regulation May Shift: What RFK Jr.'s Announcement Means for Compounding Pharmacies. Frier Levitt Articles. 2025. Available at: https://www.frierlevitt.com/articles/fda-peptide-regulation-rfk-announcement-compounding-pharmacies/
- Sheppard Health Law. FDA's Semaglutide Shortage Resolution: Legal Implications and Risks for Compounding Pharmacies. Healthcare Law Blog. 2025. Available at: https://www.sheppardhealthlaw.com/2025/03/articles/fda/fdas-semaglutide-shortage-resolution-legal-implications-and-risks-for-compounding-pharmacies/
- Lexology. FDA Removes Certain Peptide Bulk Drug Substances from Category 2. Lexology. 2024. Available at: https://www.lexology.com/library/detail.aspx?g=2e55b76a-3173-4e04-beda-bf021202f18d
- Holt Law. The Unregulated World of Peptides: What You Need to Know Before You Inject. DJ Holt Law. 2024. Available at: https://djholtlaw.com/the-unregulated-world-of-peptides-what-you-need-to-know-before-you-inject/
- Florida Healthcare Law Firm. Are Peptides Legal in the U.S.? Complete 2025 Legal Guide. Florida Healthcare Law Firm. 2025. Available at: https://floridahealthcarelawfirm.com/are-peptides-legal/
- Kim YH. Legal Peptide Compounding Guide: 503A & 503B Regulatory Compliance. Yoonhang Kim Law. 2024. Available at: https://www.yoonhangkim.com/post/legal-peptide-compounding-guide503a-503b-regulatory-compliance
- Harris Beach Murtha. GLP-1 Weight-Loss Drugs Off Shortage List; Deadlines to Stop Compounding. Harris Beach Murtha Insights. 2025. Available at: https://www.harrisbeachmurtha.com/insights/glp-1-weight-loss-drugs-off-shortage-list-deadlines-to-stop-compounding/
- Spencer Fane. Beyond the Headlines: FDA Warning Letters to GLP-1 Compounders. Spencer Fane Insights. 2025. Available at: https://www.spencerfane.com/insight/beyond-the-headlines-fda-warning-letters-to-glp-1-compounders/
- Burr & Forman LLP. The FDA Removes Semaglutide from the Drug Shortage List. Burr Newsroom. 2025. Available at: https://www.burr.com/newsroom/articles/the-fda-removes-semaglutide-from-the-drug-shortage-list
- McDermott Will & Emery. Semaglutide Shortage Resolved. MWE Insights. 2025. Available at: https://www.mwe.com/insights/semaglutide-shortage-resolved/
- U.S. Drug Enforcement Administration. DEA and HHS Extend Telemedicine Flexibilities through 2025. DEA Documents. 2024. Available at: https://www.dea.gov/documents/2024/2024-11/2024-11-15/dea-and-hhs-extend-telemedicine-flexibilities-through-2025
- HHS Telehealth. Prescribing Controlled Substances via Telehealth. Telehealth.HHS.gov. 2025. Available at: https://telehealth.hhs.gov/providers/telehealth-policy/prescribing-controlled-substances-via-telehealth