Executive Summary
Peptides are among the most promising molecules in modern biomedical research, but their therapeutic potential hinges on one often-overlooked factor: proper storage. A peptide that's been mishandled, left at room temperature too long, or exposed to light can lose its biological activity entirely. This guide covers everything researchers need to know about keeping peptides stable, potent, and ready for use.
Key Takeaways
- Lyophilized peptides stored at -20 degrees C remain stable for 2 to 5 years. At -80 degrees C, stability can extend beyond a decade. Room temperature storage is acceptable only for short periods of days to weeks.
- Reconstituted peptides in bacteriostatic water should be refrigerated at 2 to 8 degrees C and used within 28 to 30 days. Freezing reconstituted solutions in single-use aliquots can extend this to several months.
- Light exposure degrades tryptophan, tyrosine, and phenylalanine residues. Amber vials or aluminum foil wrapping provide effective protection.
- Oxidation targets methionine and cysteine residues and is accelerated by dissolved oxygen and trace metals. Nitrogen purging and chelating agents help.
- Deamidation of asparagine residues is pH-dependent, generally faster at neutral to basic pH, and accelerated by heat.
Whether you're working with BPC-157 for tissue repair studies, semaglutide for metabolic research, or GHK-Cu for skin and longevity experiments, proper storage isn't optional. It's the difference between valid results and wasted time. Peptide degradation happens through well-characterized chemical pathways, and each one can be slowed or prevented with the right environmental controls.
The core message of this guide is straightforward. Lyophilized (freeze-dried) peptides are remarkably stable when kept cold, dry, and dark. Once reconstituted in solution, that stability window shrinks dramatically, from years down to weeks. Temperature is the single most influential factor, with every 10 degrees Celsius of warming roughly doubling most chemical degradation rates. Light, oxygen, pH, and moisture each play supporting roles that can accelerate or slow these processes.
Here are the key takeaways you'll find throughout this report:
- Lyophilized peptides stored at -20 degrees C remain stable for 2 to 5 years. At -80 degrees C, stability can extend beyond a decade. Room temperature storage is acceptable only for short periods of days to weeks.
- Reconstituted peptides in bacteriostatic water should be refrigerated at 2 to 8 degrees C and used within 28 to 30 days. Freezing reconstituted solutions in single-use aliquots can extend this to several months.
- Light exposure degrades tryptophan, tyrosine, and phenylalanine residues. Amber vials or aluminum foil wrapping provide effective protection.
- Oxidation targets methionine and cysteine residues and is accelerated by dissolved oxygen and trace metals. Nitrogen purging and chelating agents help.
- Deamidation of asparagine residues is pH-dependent, generally faster at neutral to basic pH, and accelerated by heat.
- Aggregation affects hydrophobic peptides at high concentrations and is worsened by agitation, freeze-thaw cycling, and temperature fluctuations.
This guide is organized to take you from the fundamental chemistry of degradation through practical, peptide-specific storage recommendations. We'll cover temperature requirements in detail, explain why light protection matters, compare lyophilized and reconstituted stability profiles, and provide protocols for freezing, travel, and long-term archival storage. A troubleshooting section helps you identify degraded peptides before they compromise your research. And a detailed FAQ addresses the questions researchers ask most often.
For researchers new to peptide handling, we recommend starting with our peptide reconstitution guide for the basics of preparing peptides for use, then returning here for the deeper storage science. For a broader overview of the peptide landscape, visit the FormBlends Peptide Hub.
Key Principle
The single most effective thing you can do for peptide stability is minimize the time any peptide spends in solution at elevated temperatures. Keep lyophilized stocks frozen, reconstitute only what you need, and refrigerate reconstituted solutions immediately. This simple discipline prevents the vast majority of peptide degradation problems.

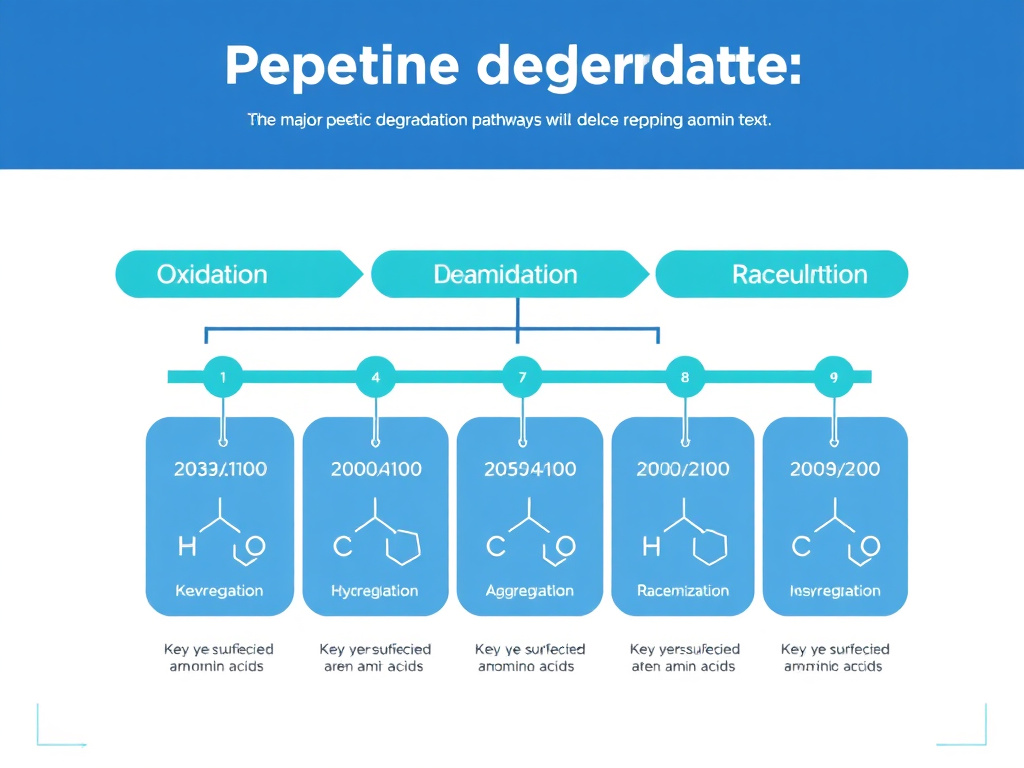
Peptide Degradation Chemistry
Understanding how and why peptides degrade is the foundation for preventing it. Peptide degradation isn't random. It follows specific, well-characterized chemical pathways that target particular amino acid residues and structural features. Each pathway has its own triggers, kinetics, and environmental dependencies.
Peptide degradation falls into two broad categories: chemical degradation, which alters the covalent structure of the molecule, and physical degradation, which changes its higher-order structure or distribution without breaking bonds. Both reduce biological activity, but they do so through different mechanisms and respond to different preventive strategies. Let's walk through each major pathway in detail.
2.1 Oxidation
Oxidation is one of the most common and consequential degradation pathways for therapeutic peptides. It involves the addition of oxygen atoms to susceptible amino acid side chains, altering their chemical properties and often destroying biological function. The amino acids most vulnerable to oxidation are methionine (Met), cysteine (Cys), tryptophan (Trp), tyrosine (Tyr), and histidine (His), with methionine and cysteine being by far the most reactive (Manning et al., 1989).
Methionine oxidation is particularly well-studied. The thioether group in methionine's side chain readily accepts an oxygen atom to form methionine sulfoxide. This reaction can occur through multiple mechanisms. Reactive oxygen species (ROS) such as hydrogen peroxide, superoxide, and hydroxyl radicals are potent oxidants. Trace metal ions, particularly iron and copper, catalyze the generation of ROS through Fenton chemistry. Even dissolved atmospheric oxygen can slowly oxidize methionine residues over time, especially in the presence of light (Li et al., 1995).
The consequences of methionine oxidation depend on where the affected residue sits within the peptide. If the methionine is part of the active site or a receptor-binding region, oxidation can dramatically reduce or eliminate biological activity. Even methionine residues distant from the active site can affect stability by altering the peptide's folding or aggregation propensity. For peptides like BPC-157, which contains methionine at position 14, this is a practical concern that directly influences storage recommendations.
Cysteine oxidation follows a different trajectory. Free cysteine thiols (-SH) can oxidize to form disulfide bonds (-S-S-) with other cysteine residues, sulfenic acid (-SOH), sulfinic acid (-SO2H), or sulfonic acid (-SO3H). The initial formation of disulfide bonds is sometimes reversible, but further oxidation to sulfinic and sulfonic acid is irreversible. For peptides that rely on specific disulfide bonding patterns for their activity, such as oxytocin and some growth hormone-releasing peptides, oxidation can scramble these critical structural elements.
Tryptophan oxidation produces a range of products including N-formylkynurenine, kynurenine, and various hydroxylated derivatives. These reactions are particularly relevant in the context of photodegradation (discussed in Section 4) because tryptophan strongly absorbs UV light in the 270 to 290 nm range. The photoproducts of tryptophan oxidation are often colored, giving degraded peptide solutions a yellow or brown tint that serves as a visible warning sign.
Preventing Oxidation
Several strategies effectively reduce oxidative degradation:
- Nitrogen or argon purging: Displacing dissolved oxygen from vials and headspace dramatically reduces oxidation rates. One study found that replacing air with nitrogen reduced photodegradation of an IgG formulation by more than 50% even without light protection (Qi et al., 2009).
- Antioxidant excipients: Methionine itself can be added as a sacrificial antioxidant at millimolar concentrations. Ascorbic acid and sodium thiosulfate also scavenge ROS effectively.
- Metal ion chelators: EDTA and DTPA bind trace metals that catalyze oxidation. Concentrations of 0.01 to 0.1 mM are typically sufficient.
- Temperature reduction: Oxidation rates follow Arrhenius kinetics. Reducing storage temperature from 25 degrees C to 4 degrees C typically reduces oxidation rates by 3 to 5 fold.
- Light protection: Amber glass vials or aluminum foil wrapping blocks the UV wavelengths that drive photooxidation.
2.2 Deamidation
Deamidation is the hydrolytic removal of an amide group from asparagine (Asn) or glutamine (Gln) residues, converting them to aspartate (Asp) or glutamate (Glu), respectively. This introduces a negative charge at physiological pH and can significantly alter peptide structure and function. Deamidation of asparagine is far more common and rapid than deamidation of glutamine, making Asn residues the primary concern (Geiger & Clarke, 1987).
The mechanism of Asn deamidation has been thoroughly characterized. At pH values above 5, the reaction proceeds predominantly through a cyclic imide (succinimide) intermediate. The backbone nitrogen of the residue following Asn attacks the side chain amide carbonyl, forming a five-membered ring. This succinimide intermediate then hydrolyzes to yield either Asp or iso-Asp (also called beta-Asp) in an approximately 3:1 ratio of iso-Asp to Asp. The iso-Asp product introduces a kink in the peptide backbone that can disrupt secondary structure and receptor binding.
The rate of deamidation is strongly influenced by the amino acid following the Asn residue. The sequence Asn-Gly is the fastest deamidating dipeptide motif, with half-lives as short as 1 to 2 days under physiological conditions. This is because glycine's small side chain offers minimal steric hindrance to succinimide formation. Other small or flexible residues (Ser, Ala, His) in the n+1 position also accelerate deamidation, while bulky residues (Val, Leu, Ile) slow it considerably (Robinson & Robinson, 2001).
pH has a profound effect on deamidation rates. The reaction is generally slowest in the pH range of 3 to 5 and accelerates at both lower and higher pH values, though through different mechanisms. Below pH 3, direct acid-catalyzed hydrolysis of the side chain amide predominates. Above pH 6, the succinimide pathway dominates and accelerates with increasing pH due to the requirement for deprotonation of the backbone nitrogen.
Temperature is another strong driver. Deamidation follows Arrhenius kinetics with activation energies typically in the range of 80 to 100 kJ/mol. This means that a 10 degree C increase in temperature roughly doubles the deamidation rate. For a peptide with an Asn-Gly motif stored at 37 degrees C, the half-life for deamidation might be 1 to 2 days. At 4 degrees C, that same peptide might have a deamidation half-life of several months (Patel & Borchardt, 1990).
For researchers working with peptides that contain susceptible Asn residues, buffer selection matters. Phosphate buffers tend to catalyze deamidation at neutral pH, while histidine and citrate buffers are generally less problematic. Reducing the pH of reconstitution solutions to 4.5 to 5.5 can significantly slow deamidation without affecting most peptide activities, though this must be balanced against other stability considerations and solubility requirements.
2.3 Hydrolysis
Hydrolysis is the cleavage of peptide bonds by water molecules. While the peptide bond is thermodynamically unstable relative to its hydrolysis products, the reaction has a very high activation energy and is kinetically slow under mild conditions. However, certain sequences and conditions make specific peptide bonds much more susceptible to hydrolysis.
Asp-Pro bonds are the most labile peptide bonds in proteins and peptides. The aspartate side chain carboxyl group can catalyze cleavage of the adjacent peptide bond through intramolecular acid catalysis. Under acidic conditions (pH 1 to 3), Asp-Pro cleavage can proceed with half-lives measured in days to weeks, compared to years for most other peptide bonds. This is relevant for peptides that contain Asp-Pro sequences in their primary structure.
More broadly, any peptide bond adjacent to Asp can be susceptible to acid-catalyzed hydrolysis. The mechanism involves protonation of the carbonyl oxygen of the peptide bond, making it more electrophilic and susceptible to nucleophilic attack by water. The Asp side chain participates as an intramolecular acid catalyst, providing a proton donor in close proximity to the reaction site.
Hydrolysis can also be catalyzed by metal ions and trace levels of proteolytic enzymes that may be introduced during handling. Using high-purity water for reconstitution and working in clean conditions minimizes enzyme-mediated hydrolysis. Metal ion-catalyzed hydrolysis is less common but can be reduced by the same chelating agents (EDTA) used to prevent oxidation.
The practical implication is straightforward: peptides in solution are always undergoing slow hydrolysis. This background rate is negligible at cold temperatures over short time periods but becomes meaningful at room temperature over weeks to months. It's yet another reason why reconstituted peptides have limited shelf lives compared to their lyophilized counterparts.
2.4 Aggregation
Aggregation is the association of peptide molecules into larger, often non-functional complexes. Unlike the chemical degradation pathways described above, aggregation is primarily a physical process, though it can involve covalent cross-linking in later stages. Aggregation is particularly concerning because even small amounts of aggregate can dramatically reduce the effective concentration of active peptide and, in therapeutic contexts, can trigger immune responses (Moussa et al., 2016).
Peptide aggregation typically begins with partial unfolding or conformational change that exposes hydrophobic regions normally buried in the interior of the molecule. These exposed hydrophobic surfaces then associate with similar surfaces on other peptide molecules, forming soluble oligomers that can progress to larger insoluble aggregates, visible particulates, or gel-like networks.
Several factors promote aggregation:
- Temperature: Elevated temperatures increase molecular motion and promote conformational changes that expose hydrophobic surfaces. However, extremely low temperatures during freezing can also cause aggregation through ice-interface effects and freeze-concentration.
- Concentration: Higher peptide concentrations increase the probability of intermolecular encounters. There's often a critical concentration above which aggregation accelerates dramatically.
- Agitation: Shaking, stirring, or pumping creates air-liquid interfaces where peptides can adsorb and partially unfold. Shipping and handling vibrations can trigger aggregation in concentrated peptide solutions.
- pH: The isoelectric point (pI) of a peptide is the pH at which it carries zero net charge. Near the pI, electrostatic repulsion between molecules is minimized, making aggregation more favorable. Storage pH should generally be 1 to 2 units away from the pI.
- Ionic strength: High salt concentrations screen electrostatic repulsion between charged peptide molecules, promoting aggregation. However, very low ionic strength can also destabilize some peptides by removing favorable electrostatic interactions.
- Freeze-thaw cycling: Repeated freezing and thawing is a well-known cause of protein and peptide aggregation. Ice crystal formation creates high local concentrations at ice-liquid interfaces, and the physical stress of crystal growth can damage peptide structures. This is why single-use aliquoting is so strongly recommended.
Aggregates can be characterized by their size, reversibility, and mechanism of formation. Reversible self-association produces oligomers that dissociate upon dilution, change in pH, or temperature shift. Irreversible aggregation produces stable complexes that cannot be easily broken apart. Covalent aggregation involves the formation of new chemical bonds between molecules, often through disulfide exchange, dityrosine cross-linking, or formaldehyde-mediated cross-linking from degradation of certain excipients.
For practical storage purposes, the most effective anti-aggregation strategies are: maintaining cold storage temperatures, avoiding freeze-thaw cycling through proper aliquoting, keeping concentrations at or below recommended levels, minimizing agitation during handling, and using appropriate surfactants (such as polysorbate 20 or 80 at 0.01 to 0.1%) to prevent surface adsorption.
2.5 Racemization
Racemization is the conversion of L-amino acid residues to their D-enantiomers within the peptide chain. Since virtually all naturally occurring peptides are composed of L-amino acids, racemization represents an unnatural modification that can alter receptor binding, enzymatic recognition, and biological activity. Racemization of even a single residue can reduce binding affinity by orders of magnitude for peptides that interact with stereospecific receptors.
The mechanism of racemization involves abstraction of the alpha-hydrogen from the amino acid carbon by a base, forming a planar carbanion intermediate that can be reprotonated from either face, yielding either the L or D configuration. This reaction is facilitated by electron-withdrawing groups adjacent to the alpha-carbon, which stabilize the carbanion intermediate. For this reason, residues adjacent to Asp, Asn, and the C-terminal carboxyl group are particularly susceptible (Geiger & Clarke, 1987).
Racemization rates are highest for residues at the N-terminus and the C-terminus of peptides, where the adjacent amino or carboxyl groups provide electron-withdrawing character. Internal residues racemize more slowly due to the electron-donating nature of the peptide bonds flanking them. Serine, aspartate, and asparagine are among the fastest-racemizing amino acids in peptides.
Temperature and pH both influence racemization rates. Like deamidation, racemization follows Arrhenius kinetics and approximately doubles in rate for each 10 degree C increase in temperature. Basic pH promotes racemization by facilitating alpha-hydrogen abstraction, while acidic conditions slow it. The practical impact is that racemization is rarely a concern for peptides stored properly at cold temperatures and mildly acidic pH, but it can become significant during prolonged storage at elevated temperatures.
2.6 Disulfide Scrambling
Many biologically active peptides contain disulfide bonds that are critical for maintaining their three-dimensional structure and function. Disulfide scrambling occurs when existing disulfide bonds break and reform with different cysteine partners, creating non-native disulfide pairings. This process, also called disulfide exchange or disulfide shuffling, can produce misfolded peptides with dramatically reduced or absent biological activity.
Disulfide exchange is catalyzed by free thiol groups. Even a small amount of reduced cysteine (either from partial reduction of the peptide or from exogenous thiol-containing compounds) can initiate a chain reaction of disulfide interchange. The reaction is fastest at alkaline pH (above 7.5) and is promoted by elevated temperatures.
For peptides containing multiple disulfide bonds, the number of possible non-native disulfide arrangements grows rapidly. A peptide with two disulfide bonds has three possible pairings, while one with three disulfide bonds has 15 possible arrangements, and one with four has 105. The probability of maintaining the single native arrangement decreases accordingly.
Preventing disulfide scrambling requires minimizing exposure to thiol-containing compounds, maintaining slightly acidic pH (5.0 to 6.5), keeping storage temperatures cold, and avoiding metal ions that can catalyze disulfide reduction. For research peptides with disulfide bonds, the use of nitrogen-purged, deoxygenated storage conditions is especially valuable because dissolved oxygen can generate reactive intermediates that initiate disulfide exchange.
2.7 Pyroglutamate Formation and Diketopiperazine Formation
Two additional degradation pathways deserve mention for their relevance to specific peptide structures.
Pyroglutamate formation occurs when an N-terminal glutamine (Gln) or glutamate (Glu) residue cyclizes to form pyroglutamate (pGlu) with loss of water or ammonia, respectively. This reaction removes a charged group from the N-terminus and can affect receptor binding or immunogenicity. The reaction is accelerated by heat and acidic pH for Gln cyclization and by heat and basic pH for Glu cyclization. Several neuropeptides and growth hormone-releasing peptides have N-terminal Gln residues that are susceptible to this modification.
Diketopiperazine (DKP) formation occurs when the N-terminal amino group attacks the carbonyl of the second peptide bond, cleaving off the first two amino acids as a cyclic dipeptide. This reaction is fastest when the second residue is proline or glycine and is promoted by elevated pH and temperature. DKP formation can be a significant degradation pathway for short peptides (fewer than 10 residues) where the first two residues represent a large fraction of the total sequence.
Summary of Degradation Pathways
All major peptide degradation pathways share three common accelerants: heat, alkaline pH, and oxidative stress. Cold storage, mildly acidic pH, and protection from oxygen and light address the root causes of virtually every degradation mechanism simultaneously. This is why the basic storage recommendations of "cold, dry, dark" are so universally effective, even though the underlying chemistry varies from pathway to pathway.
| Degradation Pathway | Susceptible Residues | Primary Triggers | pH Effect | Temperature Sensitivity |
|---|---|---|---|---|
| Oxidation | Met, Cys, Trp, Tyr, His | O2, light, metal ions, ROS | Variable, often faster at basic pH | High - Arrhenius kinetics |
| Deamidation | Asn (esp. Asn-Gly), Gln | Water, heat | Slowest at pH 3-5; faster at neutral/basic pH | Very high - doubles per 10 degrees C |
| Hydrolysis | Asp-Pro, Asp-X bonds | Water, acid/base, metals | Acid-catalyzed at pH 1-3 | High |
| Aggregation | Hydrophobic sequences | Heat, agitation, concentration, freeze-thaw | Fastest near isoelectric point | High, but freezing also problematic |
| Racemization | Ser, Asp, Asn, terminal residues | Base, heat | Faster at alkaline pH | High - doubles per 10 degrees C |
| Disulfide scrambling | Cys (disulfide-bonded) | Free thiols, base, metals | Faster above pH 7.5 | Moderate to high |
Temperature Requirements
Temperature is the single most powerful variable you can control to preserve peptide integrity. The relationship between temperature and degradation rate follows well-established thermodynamic principles, and the practical implications are dramatic: a peptide that lasts years in a freezer might degrade in days on a benchtop.
The Arrhenius equation governs the temperature dependence of chemical reaction rates, including peptide degradation. In simplified terms, for most degradation reactions, every 10 degree C increase in temperature approximately doubles the reaction rate. This means that a peptide stored at 25 degrees C (room temperature) degrades roughly 4 to 8 times faster than the same peptide at 4 degrees C (refrigerator), and roughly 30 to 60 times faster than at -20 degrees C (standard freezer). These aren't theoretical numbers. They've been confirmed experimentally across dozens of peptide systems (Manning et al., 1989; Cleland et al., 1993).
But the relationship between temperature and stability isn't perfectly linear across all ranges. Freezing introduces unique stresses that don't follow simple Arrhenius kinetics, and different degradation pathways have different activation energies, meaning the relative importance of each pathway shifts with temperature. Let's look at each temperature range in detail.
3.1 Ultra-Cold Storage: -80 Degrees C
Storage at -80 degrees C in an ultra-low temperature freezer represents the gold standard for long-term peptide preservation. At this temperature, virtually all chemical degradation reactions are effectively halted. Water is fully immobilized as ice, preventing hydrolysis and deamidation. Molecular motion is so limited that oxidation proceeds at negligible rates even in the presence of dissolved oxygen. Aggregation driven by conformational flexibility is essentially impossible.
Research on lyophilized peptide vaccines has demonstrated stability exceeding 5 years at -80 degrees C with no detectable loss of purity, sequence integrity, or biological activity (Slingluff et al., 2001). For reconstituted peptide solutions stored as frozen aliquots at -80 degrees C, stability of 1 to 2 years is commonly reported, though this depends on the specific peptide and formulation.
The main limitations of -80 degrees C storage are practical rather than scientific. Ultra-low freezers are expensive to purchase and maintain, consume significant electricity, and require backup power systems to protect against temperature excursions during power outages. They also have limited capacity compared to standard freezers. For most research applications, -80 degrees C storage is reserved for master stocks and long-term archives rather than working supplies.
Best practices for -80 degrees C storage:
- Aliquot reconstituted peptides into single-use volumes before freezing
- Use cryovials rated for ultra-low temperatures (polypropylene is standard)
- Label everything clearly, as frost can obscure writing on tubes
- Minimize door openings to prevent temperature fluctuations
- Use a temperature monitoring system with alarms
- Maintain a detailed inventory to reduce search time with the door open
3.2 Standard Freezer: -20 Degrees C
The standard laboratory freezer at -20 degrees C is the most commonly recommended storage temperature for lyophilized peptides. It offers an excellent balance of preservation and practicality. Most lyophilized peptides maintain greater than 90% purity after 2 to 5 years at -20 degrees C, provided they remain sealed and protected from moisture (GenScript, 2024).
At -20 degrees C, water is frozen but some molecular mobility persists, particularly in the amorphous (non-crystalline) regions of lyophilized cakes. This residual mobility means that degradation doesn't stop entirely. It just slows to rates that are negligible over typical research timescales. Deamidation, for example, still occurs but at rates roughly 100-fold slower than at room temperature.
One important consideration with -20 degrees C storage is the behavior of frost-free (auto-defrost) freezers. These freezers cycle their temperature to prevent ice buildup, typically warming to around -10 degrees C during defrost cycles before cooling back down. This cycling can subject peptides to repeated temperature fluctuations that accelerate aggregation and other degradation pathways. Manual-defrost freezers maintain a more consistent temperature and are preferable for peptide storage. If a frost-free freezer is the only option, store peptides in insulated containers (styrofoam boxes or insulated bags) within the freezer to dampen temperature swings.
For reconstituted peptide solutions stored at -20 degrees C, stability depends heavily on the specific formulation and whether protective excipients (cryoprotectants) are present. Simple aqueous solutions frozen at -20 degrees C may undergo freeze-concentration effects where the peptide becomes highly concentrated in unfrozen liquid channels between ice crystals. This concentration effect can accelerate aggregation and other concentration-dependent degradation reactions. Adding cryoprotectants such as trehalose, sucrose, or glycerol at 5 to 10% (w/v) protects against these effects.
3.3 Refrigerated Storage: 2 to 8 Degrees C
Refrigerator temperature (2 to 8 degrees C) is the standard recommendation for reconstituted peptides in active use. At this temperature range, degradation rates are substantially slower than at room temperature but not fully arrested. The practical shelf life of most reconstituted peptides at 2 to 8 degrees C ranges from 2 to 6 weeks, depending on the peptide, solvent, and formulation.
Bacteriostatic water (sterile water containing 0.9% benzyl alcohol) is the most common reconstitution solvent for research peptides. The benzyl alcohol serves as a preservative that inhibits bacterial growth, addressing the microbial contamination risk that becomes relevant once peptides are in solution. However, the preservative effect of benzyl alcohol diminishes over time. During weeks 1 through 4, bacterial inhibition is strong and contamination risk is minimal with proper aseptic technique. From weeks 4 through 6, the preservative effect weakens and contamination becomes possible, especially with poor handling. Beyond 6 weeks, the preservative is largely ineffective and bacterial growth is likely if any contamination occurred during reconstitution or dose drawing.
This timeline aligns with the chemical stability data for most peptides. While some particularly stable peptides might retain chemical integrity for 2 to 3 months in the refrigerator, the microbial risk from the diminishing preservative effect generally limits practical shelf life to about 4 weeks. This is why the standard recommendation is to use reconstituted peptides within 28 to 30 days.
For peptides reconstituted in sterile water without preservative, the shelf life at 2 to 8 degrees C is much shorter, typically 1 to 7 days, because bacterial contamination can occur rapidly once the sterile barrier is breached by needle puncture. Sterile water without preservative should only be used when the entire vial will be consumed in a single session.
Some specific storage notes for refrigerated peptides:
- Store vials upright to minimize contact between the solution and the rubber stopper, which can leach extractables
- Keep vials in the interior of the refrigerator, not in the door, where temperature fluctuates most during opening and closing
- Wrap vials in aluminum foil or store in opaque containers if amber glass isn't used
- Write the reconstitution date on each vial and discard after 28 to 30 days
- Never return a vial to frozen storage after it has been at refrigerator temperature for an extended period. The freeze-thaw cycle can trigger aggregation and accelerate other degradation
3.4 Room Temperature: 20 to 25 Degrees C
Room temperature storage should be considered a short-term holding condition, not a long-term storage solution. For lyophilized peptides, room temperature exposure during shipping or brief handling periods (days to a few weeks) is generally acceptable. Studies on lyophilized peptide vaccines have shown that 17 out of 18 tested peptides remained stable at room temperature for up to 3 months (Slingluff et al., 2001). However, this isn't a recommendation for routine room temperature storage. It's evidence that brief excursions aren't catastrophic for well-formulated lyophilized peptides.
For reconstituted peptides, room temperature is problematic. All chemical degradation pathways accelerate substantially at 20 to 25 degrees C compared to refrigerator temperature. Deamidation rates increase roughly 4-fold, oxidation rates 3 to 5-fold, and hydrolysis rates 2 to 4-fold. Microbial growth also accelerates dramatically, with common contaminants doubling in number every 20 to 30 minutes at room temperature compared to virtually no growth at 4 degrees C.
The practical shelf life of reconstituted peptides at room temperature is measured in days, not weeks. Most manufacturers and researchers recommend limiting room temperature exposure of reconstituted peptides to the minimum time needed for dose preparation, typically minutes rather than hours. If a reconstituted peptide will not be returned to the refrigerator within 30 to 60 minutes, it should be placed on ice or in a cooler.
Some commercially formulated peptide products have been validated for longer room temperature storage. For example, semaglutide (Ozempic) pens in use can be stored at up to 30 degrees C for 56 days, and tirzepatide (Mounjaro) pens can be stored at up to 30 degrees C for 21 days. These extended room temperature stabilities are achieved through careful formulation with stabilizing excipients and do not apply to research-grade reconstituted peptides in bacteriostatic water.
3.5 Elevated Temperatures: Above 30 Degrees C
Temperatures above 30 degrees C represent a danger zone for nearly all peptides, whether lyophilized or in solution. At 37 degrees C (body temperature), degradation rates are roughly 2 to 3 times faster than at 25 degrees C. At 40 degrees C, which is easily reached in vehicles, garages, or poorly climate-controlled shipping environments, rates increase further.
Accelerated stability studies conducted at 40 degrees C are commonly used in pharmaceutical development to predict long-term stability at lower temperatures. These studies consistently show that peptides degrade much faster than simple Arrhenius predictions would suggest, because elevated temperatures not only speed up existing degradation pathways but can also activate new pathways that are negligible at lower temperatures. For example, aggregation that might never occur at 4 degrees C can become the dominant degradation pathway at 40 degrees C due to thermally induced conformational changes.
Exposure to temperatures above 50 degrees C, even briefly, can cause irreversible damage to most peptides. For lyophilized peptides, such temperatures can cause the lyophilized cake to collapse (melt back), dramatically increasing moisture absorption and subsequent degradation. For solutions, temperatures above 50 degrees C can trigger rapid aggregation, precipitation, and widespread chemical modification.
During summer months, interior temperatures of vehicles and shipping containers can exceed 60 degrees C. This is why cold chain shipping (with ice packs or dry ice) is essential for peptide transport, and why peptides should never be left in vehicles, near heating vents, or in direct sunlight.
Critical Warning: Heat Exposure
Even a single episode of heat exposure can permanently damage peptides. A vial left in a hot car for a few hours, sitting on a sunny windowsill, or placed near laboratory heating equipment may look unchanged but could have lost significant activity. When in doubt about temperature history, analytical testing (HPLC, mass spectrometry) is the only way to confirm integrity. If testing isn't available, it's safer to discard and use a fresh vial.
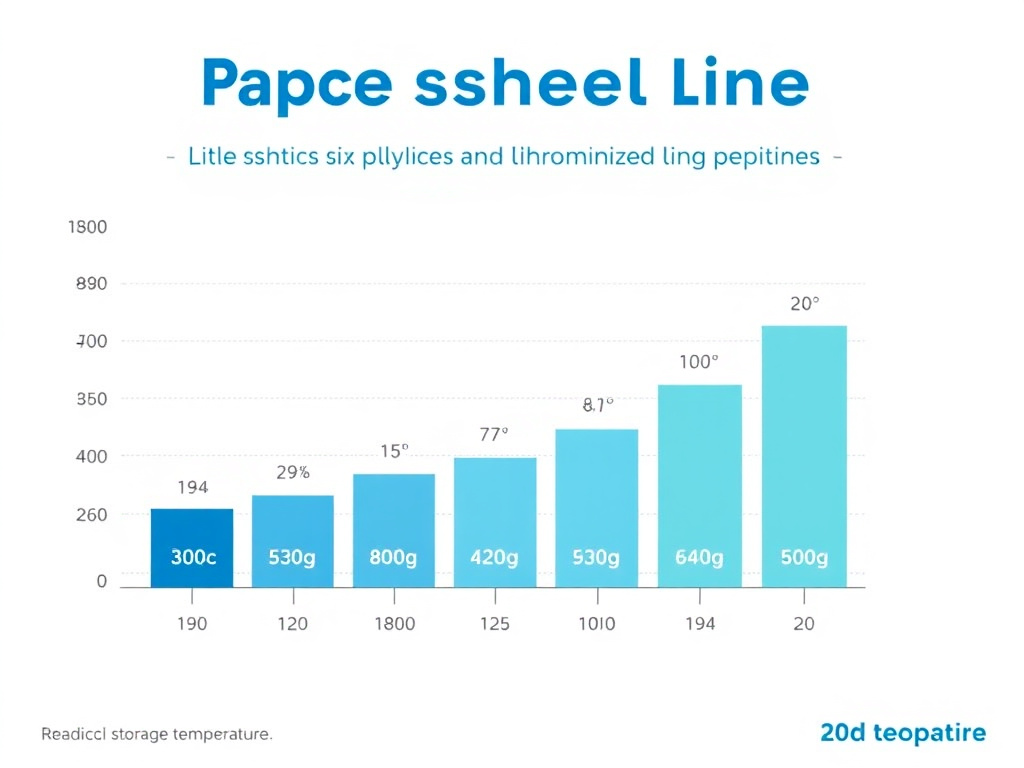
Reconstituted Peptide Stability by Storage Temperature
Data represents approximate stability windows for typical research peptides reconstituted in bacteriostatic water. Actual stability varies by peptide sequence, formulation, and handling conditions.
| Storage Temperature | Lyophilized Stability | Reconstituted Stability | Primary Use Case | Equipment Needed |
|---|---|---|---|---|
| -80 degrees C | 10+ years | 1-2 years (aliquoted) | Master stocks, long-term archive | Ultra-low freezer |
| -20 degrees C | 2-5 years | 3-6 months (aliquoted) | Working stocks, standard storage | Manual-defrost freezer |
| 2-8 degrees C | 6-12 months | 28-30 days | Active use reconstituted vials | Laboratory refrigerator |
| 20-25 degrees C (RT) | Weeks to months | Days | Brief handling only | None (avoid) |
| 30+ degrees C | Days to weeks | Hours | Never recommended | N/A - avoid exposure |
Light and Oxidation Sensitivity
Light is a silent peptide killer. Unlike temperature problems, which at least sometimes produce visible changes, photodegradation can destroy peptide activity without any obvious signs. And it starts faster than most researchers realize, with significant damage possible within hours of unprotected exposure.
Photodegradation of peptides occurs when amino acid residues absorb photons in the ultraviolet (UV) and visible light spectrum, entering excited electronic states that drive chemical reactions. The primary chromophores (light-absorbing groups) in peptides are the aromatic amino acids tryptophan (Trp), tyrosine (Tyr), and phenylalanine (Phe), along with disulfide bonds and histidine (His) to a lesser extent. Each of these absorbs light at characteristic wavelengths and produces distinct degradation products (Kerwin & Remmele, 2007).
4.1 Tryptophan Photodegradation
Tryptophan is the most light-sensitive amino acid in peptides. It absorbs UV light strongly in the 270 to 290 nm range and can also be excited by near-UV light (300 to 320 nm) and even visible light above 320 nm through its indole ring system. When tryptophan absorbs a photon, it enters a singlet excited state that can undergo intersystem crossing to a longer-lived triplet state. From this triplet state, tryptophan can participate in a variety of photochemical reactions.
The primary photoproducts of tryptophan degradation include N-formylkynurenine (NFK), kynurenine, 5-hydroxytryptophan, and various ring-opened products. These reactions can proceed through both Type I (electron transfer) and Type II (energy transfer to oxygen, generating singlet oxygen) photochemical mechanisms. Singlet oxygen generated by Type II photosensitization can then go on to oxidize other susceptible residues, creating a cascade of oxidative damage that extends well beyond the initially excited tryptophan.
Research by Qi et al. (2009) demonstrated that photodegradation of tryptophan in monoclonal antibody formulations could be substantially reduced by two simple measures: wrapping containers in aluminum foil and replacing headspace air with nitrogen. The foil prevented light-initiated reactions, while nitrogen displacement reduced the oxygen available for Type II photosensitization. Together, these two measures essentially eliminated measurable photodegradation.
Peptides containing tryptophan residues require extra attention to light protection. These include many neuropeptides, growth hormone secretagogues, and melanocortin receptor agonists. For example, Selank and Semax both contain aromatic residues that benefit from light-protected storage.
4.2 Tyrosine Photodegradation
Tyrosine absorbs UV light in the 270 to 280 nm range through its phenol side chain. The primary photoproduct is dityrosine, formed when two tyrosine radicals combine in a covalent cross-link. Dityrosine formation is a form of covalent aggregation that is completely irreversible and can serve as a sensitive marker of photodegradation. Other tyrosine photoproducts include 3,4-dihydroxyphenylalanine (DOPA) and various phenoxyl radical-derived species.
Dityrosine cross-linking is particularly problematic because it covalently connects peptide molecules, creating aggregates that cannot be dissociated by dilution, pH change, or temperature adjustment. Even low levels of dityrosine formation can significantly reduce the free monomer concentration in a peptide solution.
4.3 Disulfide Bond Photolysis
Disulfide bonds absorb UV light in the 250 to 270 nm range. Photolysis of disulfide bonds generates thiyl radicals that can undergo a cascade of reactions: recombination to form non-native disulfides (scrambling), reaction with oxygen to form sulfenic acid, or abstraction of hydrogen atoms from other residues. For disulfide-containing peptides, light exposure can therefore trigger both disulfide scrambling and oxidative damage simultaneously.
4.4 Practical Light Protection Strategies
Protecting peptides from light is straightforward and inexpensive. Here are the key strategies, ranked by effectiveness:
- Amber glass vials: Amber glass blocks UV light below about 450 nm, providing excellent protection against the wavelengths most damaging to peptides. Most research peptide suppliers ship in amber vials for this reason. If your peptides arrive in clear glass, transfer them to amber vials or wrap in foil immediately.
- Aluminum foil wrapping: For vials not made of amber glass, wrapping in aluminum foil provides complete light blocking across all wavelengths. This is the simplest and most effective light protection measure. Ensure the foil covers the entire vial, including the top and bottom.
- Opaque storage containers: Storing vials inside opaque boxes, bags, or bins within the refrigerator or freezer provides secondary light protection and also helps maintain consistent temperature.
- Minimizing handling time: Every second a peptide solution spends out of its protected container and exposed to ambient light contributes to photodegradation. Develop efficient dose-drawing techniques to minimize exposure time. Work quickly, and return vials to dark, cold storage immediately after use.
- Avoiding fluorescent lighting: Standard fluorescent lights emit small amounts of UV radiation that can contribute to photodegradation over time. While the intensity is much lower than sunlight, chronic exposure (such as leaving vials on an open lab bench under fluorescent lights) can cause measurable damage over days to weeks. LED lighting produces less UV and is generally preferable in areas where peptides are handled.
- Never storing on windowsills or in sunlight: Direct sunlight is the most intense source of UV radiation in typical indoor environments. Even 30 minutes of direct sunlight exposure can cause significant photodegradation of sensitive peptides. Windows in storage areas should have UV-blocking film, or peptides should simply never be placed near windows.
Practical Tip
A good rule of thumb: if you can see a peptide vial clearly with the naked eye, light can reach the peptide inside it. Amber glass reduces the risk, but fully opaque storage (foil wrap plus opaque container) is the most reliable protection. The few seconds it takes to unwrap and rewrap a foil covering are far less costly than replacing a degraded peptide.
4.5 Oxidation Beyond Light-Driven Pathways
While photodegradation is a major oxidation driver, peptide oxidation also occurs in the dark through several non-photochemical mechanisms. Understanding these pathways is critical for researchers storing peptides in dark, cold conditions who might assume their peptides are fully protected from oxidation.
Dissolved oxygen. Even when stored in the dark at cold temperatures, peptides in solution are exposed to dissolved oxygen. At equilibrium with atmospheric air, water at 4 degrees C contains approximately 12 mg/L of dissolved oxygen, enough to drive slow oxidation of susceptible residues over weeks to months. This background oxidation is generally the limiting factor for the shelf life of refrigerated peptide solutions.
Metal ion-catalyzed oxidation. Trace levels of transition metal ions (particularly Fe2+/Fe3+ and Cu+/Cu2+) catalyze the generation of reactive oxygen species through Fenton and Haber-Weiss chemistry. These metals are ubiquitous contaminants in water, buffer salts, glass, and rubber stoppers. Even at parts-per-billion concentrations, they can significantly accelerate peptide oxidation. This is why high-purity water and chelating agents (EDTA) are important for peptide formulations.
Peroxide contaminants. Some common excipients, particularly polysorbate surfactants (Tween 20, Tween 80), can contain or generate peroxide impurities through autooxidation. These peroxides are potent peptide oxidants. If surfactants are used in peptide formulations, they should be of the highest available purity and tested for peroxide content.
Container-related oxidation. Rubber stoppers and plastic containers can contain or release oxidizing compounds. Fluoropolymer-coated stoppers (such as those with PTFE/Teflon liners) reduce leachable oxidants compared to standard butyl rubber stoppers. Glass vials are generally preferable to plastic for oxidation-sensitive peptides because glass is more inert and has lower oxygen permeability.

Lyophilized vs Reconstituted Stability
The difference in stability between lyophilized and reconstituted peptides is so dramatic that it deserves its own dedicated discussion. Going from powder to solution changes the stability equation by orders of magnitude, and understanding why helps researchers make better decisions about when to reconstitute and how much to prepare at once.
5.1 Why Lyophilization Works So Well
Lyophilization (freeze-drying) removes water from a peptide solution under vacuum at low temperature, leaving behind a dry, porous "cake" or powder. This process is so effective at preserving peptides because it eliminates the medium in which most degradation reactions occur. Without liquid water, hydrolysis can't proceed. Deamidation through the succinimide pathway requires water for the final hydrolysis step. Aggregation driven by hydrophobic interactions in solution is eliminated. Microbial growth is impossible without available water.
The residual moisture content of a properly lyophilized peptide is typically between 1% and 3% (w/w). This small amount of water is tightly bound to the peptide matrix and has very limited chemical reactivity. However, if moisture ingress occurs during storage - through a compromised seal, repeated opening of containers, or storage in humid environments without desiccant - the residual moisture can increase to levels where degradation reactions resume.
Research on lyophilized immunoglobulin (IgG) formulations has shown that moisture content is far more consequential at elevated temperatures than at cold temperatures. Samples with varying moisture levels (0.1% to 5% w/w) showed no significant differences in degradation at 20 degrees C over 6 months. But at 45 degrees C, the same samples showed dramatically different degradation rates, with higher moisture correlating to much faster monomer loss and aggregate formation (Chang et al., 2005). This underscores why the combination of cold and dry is so powerful: even if small amounts of moisture seep in, cold temperatures render it harmless.
The physical structure of the lyophilized cake also matters. A well-formed cake has a porous, uniform structure that reconstitutes quickly and completely when solvent is added. A collapsed cake, caused by exceeding the collapse temperature during freeze-drying or by subsequent heat exposure, has a denser structure with reduced surface area. Collapsed cakes reconstitute more slowly and may contain higher levels of degradation products due to the thermal stress that caused the collapse.
5.2 The Reconstitution Transition
When a lyophilized peptide is reconstituted - typically with bacteriostatic water, sterile water, or a buffer solution - it transitions from a highly stable solid state to a much less stable liquid state. This transition is essentially irreversible from a stability standpoint. Once dissolved, the peptide is exposed to all the degradation pathways discussed in Section 2: oxidation by dissolved oxygen, deamidation by water, hydrolysis, and aggregation driven by molecular motion and intermolecular interactions.
The reconstitution process itself introduces several potential risks:
- Mechanical stress: Vigorous shaking or vortexing during reconstitution can create air-liquid interfaces that promote aggregation. The recommended technique is gentle swirling or slow rotation of the vial until the powder dissolves completely. Direct injection of solvent onto the lyophilized cake can also cause localized high concentrations and aggregation. Instead, aim the needle at the vial wall and let the solvent run down slowly.
- pH shock: If the reconstitution solvent has a very different pH than the original lyophilization buffer, the peptide may experience a transient pH extreme that triggers deamidation, hydrolysis, or aggregation. Most peptides are formulated to reconstitute well in bacteriostatic water (pH approximately 5.5 to 7.0), but checking compatibility is worthwhile for pH-sensitive sequences.
- Temperature shock: Reconstituting a frozen vial that hasn't fully thawed, or using cold solvent with a room-temperature cake, can create temperature gradients that promote aggregation. Allow lyophilized vials to equilibrate to room temperature before reconstituting.
- Contamination introduction: Each needle puncture of a vial's septum creates an entry point for microbial contamination. Using aseptic technique, including alcohol swabbing the septum before each puncture, is essential. Bacteriostatic water provides ongoing antimicrobial protection, but it is not a substitute for clean handling.
For detailed reconstitution protocols, see our complete peptide reconstitution guide, which covers solvent selection, volume calculations, and step-by-step technique for all common peptide formats.
5.3 Stability Comparison by the Numbers
The following table summarizes the approximate stability windows for lyophilized versus reconstituted peptides under various conditions. These are general guidelines; specific peptides may be more or less stable than these averages based on their sequence, formulation, and susceptibility to particular degradation pathways.
| Condition | Lyophilized Peptide | Reconstituted Peptide (BAC Water) | Reconstituted (Sterile Water, No Preservative) |
|---|---|---|---|
| -80 degrees C | 10+ years | 1-2 years (aliquoted) | 6-12 months (aliquoted) |
| -20 degrees C | 2-5 years | 3-6 months (aliquoted) | 1-3 months (aliquoted) |
| 2-8 degrees C (fridge) | 6-12 months | 28-30 days | 3-7 days |
| 20-25 degrees C (RT) | 1-3 months | 1-3 days | Hours |
| 30+ degrees C | Days to weeks | Hours | Not recommended |
Several patterns emerge from this data:
- Lyophilized peptides are roughly 10 to 100 times more stable than reconstituted peptides at any given temperature.
- Bacteriostatic water extends reconstituted stability by roughly 4 to 10 times compared to plain sterile water, primarily through antimicrobial protection.
- Freezing reconstituted solutions in aliquots extends their stability by roughly 3 to 6 times compared to refrigerated storage, because freezing reduces chemical degradation rates while aliquoting prevents freeze-thaw damage.
- The stability gap between lyophilized and reconstituted forms widens as temperature increases. At -80 degrees C, the difference might be 5 to 10 fold. At room temperature, it can be 30 to 100 fold or more.
5.4 Strategic Implications: The Reconstitute-on-Demand Approach
Given the enormous stability advantage of the lyophilized form, the most effective storage strategy is what we call "reconstitute-on-demand." Rather than reconstituting an entire vial at once, prepare only the amount needed for the next 2 to 4 weeks of use. Keep the remaining lyophilized powder sealed in the freezer until needed.
If your research protocol requires reconstituting an entire vial at once (for example, because the lyophilized cake can't be easily divided), immediately aliquot the solution into single-use portions and freeze the extras at -20 degrees C or colder. Thaw one aliquot at a time and use it within 1 to 2 days. Never refreeze a thawed aliquot.
This approach maximizes the time each molecule spends in the stable lyophilized state and minimizes exposure to the degradation-promoting aqueous environment. It requires slightly more planning and handling steps, but the payoff in preserved peptide activity and reduced waste is substantial.
For researchers using CJC-1295/Ipamorelin or TB-500 on multi-week protocols, this approach is particularly valuable. Reconstitute only the first 2 to 4 weeks of supply, keep the rest frozen as lyophilized powder, and reconstitute additional supply as needed. The peptide you use in week 8 of a protocol will be just as potent as the peptide you used in week 1, rather than having spent 8 weeks slowly degrading in solution.
Key Takeaway
Think of reconstitution as starting a countdown timer. From the moment peptide powder meets solvent, degradation accelerates dramatically. The goal is to keep that timer as short as possible for each portion of peptide. Reconstitute small amounts, use them promptly, and keep the rest frozen as powder.

Freezing Protocols
Freezing is the most effective way to extend the shelf life of both lyophilized and reconstituted peptides. But freezing done wrong can actually accelerate degradation rather than prevent it. The difference between helpful and harmful freezing comes down to technique, specifically how you freeze, how you thaw, and how many times you do it.
6.1 The Freeze-Thaw Problem
Freeze-thaw cycling is one of the most damaging things you can do to a peptide solution. Each cycle of freezing and thawing subjects the peptide to multiple physical stresses that promote aggregation, surface denaturation, and concentration effects.
When an aqueous peptide solution freezes, ice crystals form first in the pure water fraction. As ice crystals grow, they exclude dissolved solutes, including the peptide, into progressively smaller channels of unfrozen liquid between the crystals. This "freeze-concentration" effect can increase the local peptide concentration by 10 to 100 fold, dramatically accelerating concentration-dependent aggregation. The peptide is also pushed into contact with the surfaces of growing ice crystals, where it can adsorb and undergo surface-induced conformational changes.
Additionally, freeze-concentration affects buffer composition. In phosphate buffers, the disodium phosphate component crystallizes preferentially during freezing, causing the pH of the remaining liquid to drop by 2 to 3 pH units. This acid shift can trigger acid-catalyzed degradation reactions that would not occur at the original formulation pH. Histidine, citrate, and Tris buffers show less pH shift during freezing and are generally preferred for peptide solutions that will be frozen.
During thawing, the process reverses, but not uniformly. The outer portions of a frozen solution thaw first, creating a liquid layer around a still-frozen core. This liquid layer is initially highly concentrated and may be at a different pH than the fully thawed solution, creating transient conditions that promote aggregation and degradation. Slow, uncontrolled thawing (such as leaving a vial on the bench) maximizes the time the peptide spends in these unfavorable transient conditions. Controlled, rapid thawing is generally preferred.
The cumulative damage from repeated freeze-thaw cycles is typically greater than the sum of individual cycles. After 3 to 5 freeze-thaw cycles, many peptides show measurable increases in aggregates, sub-visible particles, and degradation products. After 10 or more cycles, significant loss of biological activity is common. Some particularly sensitive peptides can show detectable damage after even a single freeze-thaw event.
6.2 The Aliquoting Solution
The single most effective strategy to prevent freeze-thaw damage is to never freeze-thaw. This sounds circular, but it's achieved through aliquoting: dividing a reconstituted peptide solution into single-use portions before freezing, so that each portion is frozen once and thawed once.
Here's a step-by-step aliquoting protocol:
- Calculate your aliquot volume. Determine how much reconstituted peptide you'll need per use session. Include a small excess (10 to 20%) to account for dead volume in the syringe and vial.
- Prepare aliquot containers. Use sterile, low-binding microcentrifuge tubes (polypropylene, 0.5 or 1.5 mL). Standard microcentrifuge tubes work for most peptides, but for very hydrophobic peptides or very dilute solutions, low-binding (siliconized or coated) tubes reduce surface adsorption losses.
- Label everything before you start. Write the peptide name, concentration, date, and aliquot number on each tube using a permanent marker or cryo-labels. Regular labels and markers may become illegible in the freezer.
- Reconstitute the peptide following standard protocols (see our reconstitution guide). Ensure complete dissolution before aliquoting.
- Dispense aliquots using a calibrated pipette or syringe. Mix the solution gently between aliquots to ensure uniform concentration. Avoid creating bubbles or foam.
- Add cryoprotectant if desired. For maximum stability, add trehalose or sucrose to a final concentration of 5 to 10% (w/v) before aliquoting. These sugars form a glassy matrix during freezing that protects the peptide from freeze-concentration and ice crystal damage. Glycerol at 10 to 20% (v/v) is an alternative that depresses the freezing point, reducing ice crystal formation.
- Freeze immediately. Place aliquots in the freezer as soon as possible after preparation. Slow freezing (in a standard freezer) is generally acceptable for peptides, but controlled-rate freezing (1 degree C per minute, achieved using an isopropanol-filled freezing container) produces more uniform ice crystal formation and is preferred for sensitive preparations.
- Store at -20 degrees C or colder. Aliquots stored at -80 degrees C have the longest shelf life, followed by -20 degrees C. Record the storage location and number of aliquots in your inventory system.
6.3 Thawing Best Practices
When you need a dose, remove a single aliquot from the freezer and thaw it using one of these methods, listed from most preferred to least:
Method 1: Rapid hand warming. Hold the tube between your fingers for 1 to 2 minutes until the contents are fully liquid. This is the fastest and most controlled method for small aliquots.
Method 2: Room temperature bench. Place the tube on the bench at room temperature for 5 to 10 minutes. This is slightly slower but requires no active attention.
Method 3: Cold water bath. Place the tube in a beaker of room-temperature water for 2 to 3 minutes. The water provides more efficient heat transfer than air, resulting in faster, more uniform thawing.
Avoid: Warm water baths (above 30 degrees C), microwaving, or any rapid heating method. These can create hot spots that locally denature or aggregate the peptide before the bulk solution has thawed.
Once thawed, gently invert the tube several times to ensure uniform mixing. Do not vortex or shake vigorously. Use the aliquot within 24 hours and discard any remainder. Never refreeze a thawed aliquot.
6.4 Freezing Lyophilized Peptides
For lyophilized peptides that are already in powder form, freezing is simpler because there's no liquid to undergo freeze-concentration or ice crystal formation. The main concern is moisture. Ensure that lyophilized vials are tightly sealed before placing them in the freezer, and allow them to equilibrate to room temperature before opening to prevent condensation from forming on the cold powder.
When removing a lyophilized vial from freezer storage for reconstitution:
- Remove the vial from the freezer
- Allow it to sit at room temperature for 15 to 30 minutes until it reaches ambient temperature
- Only then open the seal and reconstitute
If you open a cold vial, warm, humid room air will rush in and condense on the cold powder and vial surfaces. This moisture can initiate degradation reactions and compromise the stability of any remaining unreconstituted powder. Taking the time to let the vial warm up is a small investment that prevents a significant risk.
6.5 Flash Freezing with Liquid Nitrogen
For researchers with access to liquid nitrogen, flash freezing offers theoretical advantages. Plunging aliquot tubes directly into liquid nitrogen produces extremely rapid freezing that creates very small, uniform ice crystals rather than the larger, more damaging crystals formed during slow freezing. This results in less freeze-concentration and less mechanical stress on the peptide.
In practice, the difference between flash freezing and standard -80 degrees C freezing is modest for most peptides. Flash freezing is most beneficial for large proteins and peptides that are particularly aggregation-prone. For standard research peptides like BPC-157 or Epithalon, standard freezer storage is perfectly adequate.
If using liquid nitrogen flash freezing, transfer the frozen aliquots to a -80 degrees C or -20 degrees C freezer for long-term storage. Do not store peptides in the liquid nitrogen dewar itself, as the extreme cold (-196 degrees C) offers no additional stability benefit over -80 degrees C and the constant evaporation of nitrogen creates handling and safety concerns.
Aliquoting Quick Reference
For a 5 mg vial of peptide reconstituted in 2 mL of bacteriostatic water (2.5 mg/mL): if your dose is 250 mcg, you need 0.1 mL per dose. Create 10 aliquots of 0.2 mL each (providing one dose plus dead volume per aliquot), freeze at -20 degrees C or colder, and thaw one at a time. Use the FormBlends dosing calculator to determine your specific aliquot volumes.
Signs of Degradation
Even with the best storage practices, it's valuable to know how to recognize a degraded peptide. Some signs are obvious; others are subtle. Knowing what to look for can prevent you from using a compromised product in research and help you troubleshoot storage problems before they affect your entire inventory.
Peptide degradation can manifest in visual, physical, and functional changes. Let's cover each category in detail.
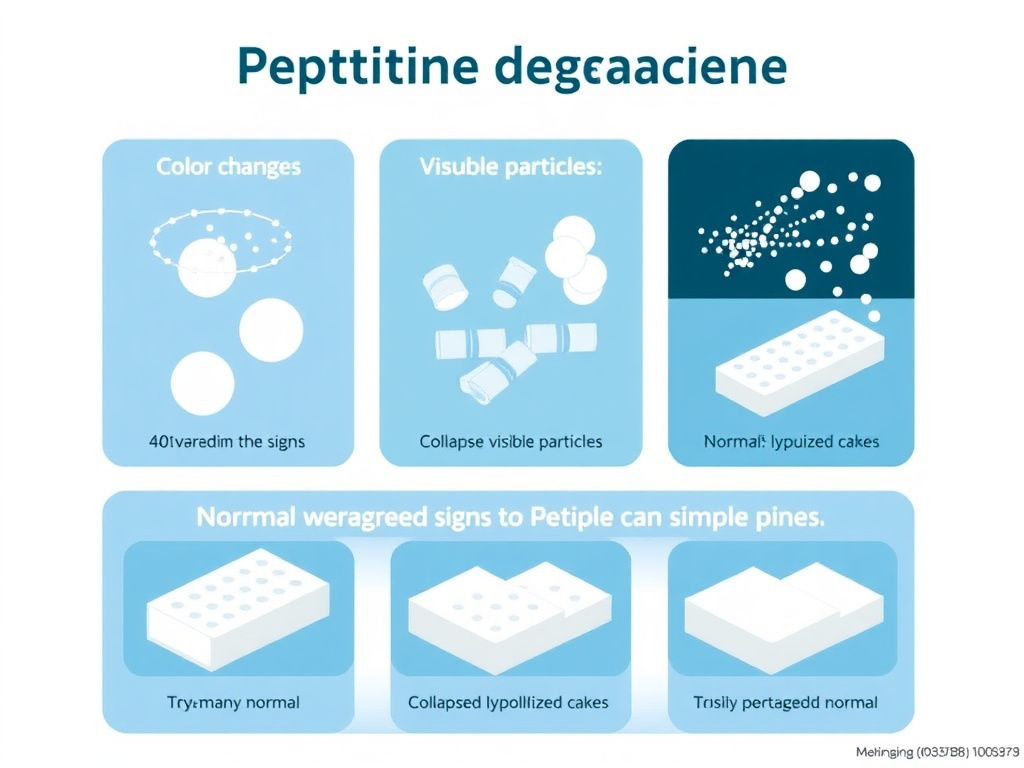
7.1 Visual Indicators
Color changes. Most peptide solutions should be clear and colorless, or at most very slightly yellow. Significant yellowing or browning indicates oxidative degradation, particularly of tryptophan residues (forming kynurenine and N-formylkynurenine, which are yellow-brown compounds). Pink or reddish coloring can indicate oxidation of tyrosine residues. Any noticeable color change from the original appearance at reconstitution is cause for concern.
Cloudiness or turbidity. A clear peptide solution that becomes cloudy or hazy is likely forming aggregates. Small aggregates scatter light, producing turbidity visible to the naked eye. If you hold the vial up to a light source and the solution appears milky, foggy, or opalescent (rather than water-clear), aggregation has probably occurred. Mild turbidity that clears upon gentle warming (to room temperature, not above) may indicate reversible self-association, while persistent turbidity indicates irreversible aggregation.
Visible particles. Flakes, fibers, or visible particles floating in the solution indicate advanced aggregation or precipitation. These particles may appear white, translucent, or colored depending on the type of degradation. Do not attempt to filter or centrifuge out particles and use the remaining solution, as the visible particles represent only the largest fraction of a distribution that also includes smaller, sub-visible particles throughout the solution. If you see particles, the entire solution should be discarded.
Precipitate formation. A solid layer settling at the bottom of the vial indicates that the peptide has precipitated out of solution. This can result from aggregation, pH changes, or exceeding the peptide's solubility limit (which decreases as the peptide degrades). Precipitate that redissolves upon warming may indicate reversible precipitation, but the stress that caused precipitation likely triggered other irreversible degradation reactions as well.
Lyophilized cake changes. For unreconstituted lyophilized peptides, a well-preserved cake should appear as a fluffy, white to off-white powder or cake that retains its original shape within the vial. Warning signs include: cake collapse (the powder has settled or compacted, appearing glassy or rubbery rather than fluffy), color change (yellowing or browning), visible moisture (droplets on the vial walls or wet-looking powder), or structural changes (the cake has shrunk away from the vial walls or developed cracks that weren't originally present).
7.2 Physical Indicators
Reconstitution behavior. A fresh, high-quality lyophilized peptide should reconstitute within 1 to 5 minutes with gentle swirling. If a peptide that previously reconstituted easily now takes much longer, requires vigorous mixing, or doesn't fully dissolve, this suggests structural changes or aggregation in the lyophilized state. Don't force reconstitution by vortexing, heating, or sonicating; these measures may appear to dissolve the powder but can create additional damage.
Foaming or unusual bubbling. Some peptides produce modest foam during reconstitution, which is normal. However, excessive foaming, unusually persistent bubbles, or foam that doesn't dissipate within a few minutes can indicate the presence of aggregates (which act as surfactants) or changes in the peptide's surface activity.
pH changes. If you routinely measure the pH of your reconstituted peptide solutions (a good practice), a shift from the expected pH may indicate degradation. Deamidation converts neutral Asn residues to acidic Asp residues, which can lower the solution pH. Extensive hydrolysis also shifts pH by generating new amino and carboxyl groups.
7.3 Functional Indicators
Reduced efficacy. In research contexts, the most concerning sign of degradation is reduced biological activity. If a peptide that previously produced consistent results in your assays begins showing weaker responses, inconsistent data, or no effect at the usual concentration, degradation should be high on your differential diagnosis list. Before blaming the peptide, rule out other variables (cell health, assay reagents, timing), but recognize that storage-related degradation is a common cause of declining experimental performance over time.
Injection site reactions. For animal research studies involving subcutaneous or intramuscular injection, degraded peptides can cause increased local irritation, redness, or swelling compared to fresh preparations. This is because degradation products, particularly aggregates, can trigger immune and inflammatory responses that the intact peptide does not. If injection site reactions increase over time while using the same peptide vial, degradation is a likely contributor.
Unexpected results. Degradation doesn't always reduce activity uniformly. Some degradation products retain partial activity or have different activity profiles than the parent peptide. If your experimental results shift in character rather than just magnitude, for example, if a peptide that normally produces response A begins producing response B, degradation creating novel active species may be responsible.
7.4 Analytical Confirmation
When visual and functional indicators suggest degradation, analytical testing can confirm and characterize the extent of damage. The most commonly used analytical methods for peptide quality assessment include:
Reversed-phase HPLC (RP-HPLC). The gold standard for assessing peptide purity. Fresh peptide should produce a single sharp peak at its characteristic retention time. Degradation products appear as additional peaks, typically at slightly different retention times from the main peak. A decrease in the main peak area and the appearance of new peaks indicates chemical degradation. Modern HPLC systems can detect degradation levels as low as 0.1 to 0.5% of the total peptide.
Mass spectrometry (MS). Provides molecular weight confirmation and can identify specific degradation products. A mass shift of +16 Da indicates methionine oxidation; -1 Da indicates deamidation; +18 Da indicates hydrolysis; and multiples of the expected molecular weight indicate covalent aggregation. LC-MS (liquid chromatography-mass spectrometry) combines the separation power of HPLC with the identification capability of MS for comprehensive degradation profiling.
Size-exclusion chromatography (SEC). Separates molecules by size and is the method of choice for detecting aggregation. Aggregates appear as peaks at shorter retention times (larger sizes) than the monomer peak. SEC can distinguish between dimers, trimers, and higher-order aggregates.
Visual inspection and turbidity measurement. The simplest "assay" is holding the vial up to a bright light and looking for clarity. Turbidity measurements with a nephelometer provide a quantitative version of this visual check. Increasing turbidity over time, even if the solution still looks clear to the naked eye, can indicate the early stages of aggregation.
When in Doubt, Throw It Out
If you see visible changes, notice reduced efficacy, or have concerns about temperature history, the safest course is to discard the questionable vial and start fresh. Peptides are valuable, but compromised results are far more costly than a replacement vial. If you're unsure whether your peptide supply has been compromised, consider requesting a certificate of analysis or having a sample tested by an analytical laboratory.
| Degradation Sign | Likely Cause | Severity | Action Required |
|---|---|---|---|
| Slight yellowing | Tryptophan oxidation | Moderate | Test potency; discard if reduced |
| Brown or dark color | Advanced oxidation | Severe | Discard immediately |
| Cloudiness | Aggregation | Moderate to severe | Discard; do not filter and use |
| Visible particles | Advanced aggregation/precipitation | Severe | Discard immediately |
| Slow reconstitution | Structural changes in lyophilized cake | Mild to moderate | Assess further; consider testing |
| Collapsed lyophilized cake | Heat exposure, moisture ingress | Moderate | Test before use; replace if possible |
| Reduced efficacy at usual dose | Multiple degradation pathways | Variable | Confirm with fresh vial; discard old |
| Unusual odor | Microbial contamination | Severe | Discard immediately; review technique |
Peptide-Specific Storage Guide
While general storage principles apply broadly across peptide classes, individual peptides have unique structural features that influence their stability profiles. A methionine-rich peptide like BPC-157 has different vulnerabilities than a lipidated GLP-1 agonist like semaglutide or a metal-coordinating peptide like GHK-Cu. This section provides compound-specific guidance for the major peptide categories.
8.1 GLP-1 Receptor Agonists
Semaglutide
Semaglutide is a GLP-1 receptor agonist modified with a C18 fatty diacid side chain that enables albumin binding and extends its half-life. This lipid modification also influences storage stability in several ways.
The fatty acid chain makes semaglutide amphiphilic (having both hydrophilic and hydrophobic regions), which increases its tendency to adsorb at air-water interfaces and on container surfaces. This surface adsorption can reduce the effective concentration in solution and promote aggregation at interfaces. Using low-binding containers and minimizing headspace in vials helps reduce these effects.
Recent preformulation studies have examined semaglutide stability across a range of conditions. The peptide showed reasonable thermal stability, remaining intact for 3 hours at 80 degrees C in short-term stress testing. Under normal storage conditions, lyophilized semaglutide is stable at room temperature for approximately 3 weeks, but optimal storage is below -18 degrees C in a desiccated environment. Once reconstituted, semaglutide should be refrigerated at 2 to 8 degrees C and used within 2 to 7 days for research applications (Arora et al., 2025).
The commercial formulation of semaglutide (Ozempic, Wegovy) uses a phosphate buffer at pH 7.4, which has been optimized for the specific stability requirements of the finished product. Research on the relationship between pH and semaglutide stability found that a higher extent of degradation occurred between pH 4.5 and 5.5, near the peptide's isoelectric point. The optimal finished product pH appears to be above 7.0 (Arora et al., 2025). This is unusual, as most peptides are more stable at mildly acidic pH. It reflects the unique structural features of semaglutide's lipidated structure.
Storage summary for semaglutide:
- Lyophilized: store at -20 degrees C or colder, desiccated, protected from light
- Reconstituted: refrigerate at 2-8 degrees C, use within 7 days
- Avoid freeze-thaw cycling of reconstituted solutions
- Use low-binding containers to minimize surface adsorption
- Commercial pen products (in use) can tolerate up to 30 degrees C for 28-56 days depending on brand
Tirzepatide
Tirzepatide is a dual GIP/GLP-1 receptor agonist, also modified with a C20 fatty diacid chain. Its storage profile is broadly similar to semaglutide, with a few distinctions.
Tirzepatide's longer fatty acid chain increases its hydrophobicity compared to semaglutide, which can make it slightly more aggregation-prone at higher concentrations. At research concentrations, this is rarely a practical concern, but it does mean that tirzepatide solutions benefit from surfactant additives (such as polysorbate 20 at 0.01 to 0.05%) to prevent surface-mediated aggregation.
The commercial product (Mounjaro) can be stored at room temperature (up to 30 degrees C) for 21 days, which is shorter than semaglutide's allowance. This likely reflects tirzepatide's greater susceptibility to thermally accelerated degradation due to its larger size and more complex structure.
Storage summary for tirzepatide:
- Lyophilized: store at -20 degrees C or colder, desiccated, protected from light
- Reconstituted: refrigerate at 2-8 degrees C, use within 7 days
- Particularly sensitive to surface adsorption; use low-binding containers
- Consider adding trace surfactant for concentrated solutions
- Commercial pen products can tolerate up to 30 degrees C for 21 days
8.2 Growth Hormone Secretagogues and Related Peptides
CJC-1295 and Ipamorelin
CJC-1295/Ipamorelin is a popular combination used in growth hormone research. Each peptide has distinct stability characteristics.
CJC-1295 contains a Drug Affinity Complex (DAC) modification that extends its half-life. This modification also influences storage stability. The DAC moiety is susceptible to hydrolysis under acidic conditions, making neutral to slightly alkaline reconstitution solutions preferable. CJC-1295 also contains methionine residues that are oxidation-susceptible, requiring light protection and ideally nitrogen-purged storage.
Ipamorelin is a shorter pentapeptide (5 amino acids) with a relatively simple structure. Short peptides are generally more stable than large ones because they have fewer susceptible residues and less complex three-dimensional structures to maintain. Ipamorelin is reasonably stable in reconstituted form, typically maintaining activity for 3 to 4 weeks at 2 to 8 degrees C.
When used in combination, the two peptides can be reconstituted together in the same vial without significant interaction effects. Standard bacteriostatic water is an appropriate solvent. The mixed solution should be refrigerated and used within 3 to 4 weeks, with the timeline driven primarily by the less stable CJC-1295 component.
Storage summary for CJC-1295/Ipamorelin:
- Lyophilized: store at -20 degrees C or colder, protected from light
- Reconstituted: refrigerate at 2-8 degrees C, use within 3-4 weeks
- Protect from light (methionine oxidation risk)
- Avoid acidic reconstitution solvents (pH below 5)
- Can be reconstituted as a combination without stability concerns
8.3 Healing and Tissue Repair Peptides
BPC-157
BPC-157 (Body Protection Compound-157) is a 15-amino acid peptide derived from a gastric juice protein. It's one of the most widely studied tissue repair peptides and has a generally favorable stability profile with a few important considerations.
BPC-157 contains a methionine residue at position 14 that is the primary site of oxidative degradation. Light exposure, dissolved oxygen, and trace metals all accelerate methionine oxidation, converting the thioether to methionine sulfoxide and reducing biological activity. Amber vials and refrigerated, light-protected storage are standard recommendations.
In the lyophilized state, BPC-157 is highly stable, maintaining integrity for years at -20 degrees C. The peptide's relatively small size and lack of disulfide bonds make it resistant to aggregation and disulfide scrambling, two common challenges with larger peptides.
Once reconstituted in bacteriostatic water, BPC-157 is stable for approximately 4 to 6 weeks at 2 to 8 degrees C under optimal conditions (sealed vial, amber glass, minimal septum punctures). For longer storage of reconstituted solutions, aliquoting and freezing at -20 degrees C extends stability to 3 to 6 months.
BPC-157 is also available in an "arginine salt" form (BPC-157-arginate) that has improved aqueous solubility compared to the acetate salt. The arginine salt form may show slightly different stability characteristics, particularly regarding pH sensitivity, but the general storage recommendations remain the same.
Storage summary for BPC-157:
- Lyophilized: store at -20 degrees C or colder, protected from light and moisture
- Reconstituted: refrigerate at 2-8 degrees C, use within 4-6 weeks
- Primary vulnerability: methionine oxidation (position 14)
- Amber vials strongly recommended
- Good candidate for aliquoting due to multi-week protocols
TB-500 (Thymosin Beta-4)
TB-500 is a 43-amino acid peptide fragment of thymosin beta-4. Its larger size compared to BPC-157 gives it a somewhat more complex stability profile.
TB-500 does not contain disulfide bonds or particularly oxidation-sensitive residues, which gives it reasonable chemical stability. However, its larger size and the presence of hydrophobic segments make it more susceptible to aggregation than smaller peptides. Reconstituted TB-500 solutions should be handled gently (no vortexing) and stored at consistent temperatures without freeze-thaw cycling.
The peptide's solubility is generally good in aqueous solutions, but concentrated solutions (above 5 mg/mL) may show increased tendency to form visible or sub-visible particles. Reconstituting at lower concentrations (1 to 2 mg/mL) and using appropriate aliquot volumes helps maintain solution clarity and stability.
Storage summary for TB-500:
- Lyophilized: store at -20 degrees C or colder
- Reconstituted: refrigerate at 2-8 degrees C, use within 3-4 weeks
- Primary vulnerability: aggregation at high concentrations
- Avoid vortexing and vigorous agitation
- Reconstitute at moderate concentrations (1-2 mg/mL)
8.4 Neuropeptides and Nootropics
Selank
Selank is a synthetic heptapeptide analog of the immunomodulatory peptide tuftsin. It contains a tryptophan residue that makes it susceptible to photodegradation. Light protection is particularly critical for Selank, and its relatively small size means that damage to even one residue represents a significant fraction of the total molecular weight.
Selank is generally stable in the lyophilized form for extended periods at -20 degrees C. Once reconstituted, it should be refrigerated and protected from light. The peptide's small size makes it resistant to aggregation but potentially more susceptible to hydrolysis and deamidation on a per-residue basis.
Intranasal formulations of Selank present additional stability challenges related to the spray mechanism, preservatives, and contact with nasal tissues. For research purposes, standard bacteriostatic water reconstitution with refrigerated storage is appropriate.
Storage summary for Selank:
- Lyophilized: store at -20 degrees C or colder, protected from light
- Reconstituted: refrigerate at 2-8 degrees C, use within 2-3 weeks
- Primary vulnerability: tryptophan photodegradation
- Mandatory light protection (amber vials or foil wrapping)
- Keep reconstituted solutions at lower concentrations if possible
Semax
Semax is a synthetic heptapeptide derived from ACTH (4-10). It has a relatively stable structure without disulfide bonds or particularly sensitive residues. Semax shows solid stability in both lyophilized and reconstituted forms.
The primary storage concern for Semax is standard degradation from heat and moisture exposure. Like Selank, intranasal formulations have additional stability considerations beyond what applies to standard reconstituted solutions.
Storage summary for Semax:
- Lyophilized: store at -20 degrees C or colder
- Reconstituted: refrigerate at 2-8 degrees C, use within 3-4 weeks
- Relatively stable peptide with no unusual vulnerabilities
- Standard light and temperature protection sufficient
8.5 Longevity and Cellular Health Peptides
Epithalon
Epithalon (Epitalon) is a tetrapeptide (Ala-Glu-Asp-Gly) studied for its effects on telomerase activity. Its very small size (4 amino acids) gives it some unique stability characteristics.
The good news is that Epithalon is exceptionally stable in the lyophilized state because of its small size and simple structure. It lacks oxidation-sensitive residues (no Met, Cys, or Trp), disulfide bonds, and complex folding requirements. The main degradation concern is deamidation, as the Asp-Gly sequence at positions 3-4 is a classic deamidation hotspot.
However, the practical impact of Asp-Gly deamidation in Epithalon is somewhat mitigated by the fact that the Asp residue is already present (not Asn). Direct Asp degradation through the succinimide pathway is slower than Asn deamidation, though it can still produce iso-Asp. At cold storage temperatures, this reaction is extremely slow.
Storage summary for Epithalon:
- Lyophilized: store at -20 degrees C or colder (exceptionally stable)
- Reconstituted: refrigerate at 2-8 degrees C, use within 4-6 weeks
- Very stable peptide; standard storage conditions are adequate
- Minor risk of Asp-Gly isomerization at elevated temperatures
GHK-Cu
GHK-Cu is a tripeptide-copper complex studied for skin regeneration and wound healing. The copper coordination adds a layer of complexity to its storage requirements that other peptides don't share.
The copper(II) ion in GHK-Cu is coordinated to the peptide through the histidine imidazole nitrogen, the N-terminal amine, and the deprotonated amide nitrogen of the Gly-His bond. This coordination is pH-dependent, with optimal stability at pH 5 to 7. At pH values below 4, the copper can dissociate from the peptide, producing free copper ions that are potent oxidation catalysts. At pH values above 8, copper hydroxide precipitation can occur.
The presence of copper introduces a dual risk. On one hand, the copper-peptide complex is the biologically active form and must be maintained. On the other hand, any free copper released from the complex can catalyze oxidative degradation of the remaining peptide. This means that conditions that destabilize the copper coordination (pH extremes, high temperatures, competing metal chelators like EDTA) should be avoided.
GHK-Cu solutions typically have a blue-green color from the copper complex. Fading or loss of this color indicates copper dissociation and peptide degradation. Deepening or darkening of the color can indicate copper-catalyzed oxidation reactions.
Storage summary for GHK-Cu:
- Lyophilized: store at -20 degrees C or colder, protected from moisture
- Reconstituted: refrigerate at 2-8 degrees C, use within 2-3 weeks
- Critical: maintain pH between 5 and 7 to preserve copper coordination
- Do NOT add EDTA or other metal chelators
- Monitor solution color as an indicator of complex integrity
- Blue-green color = normal; color fading = degradation
NAD+
NAD+ (nicotinamide adenine dinucleotide) is technically a dinucleotide rather than a peptide, but it's commonly stored and handled alongside peptides in research settings. NAD+ has unique stability challenges related to its dinucleotide structure.
NAD+ is susceptible to hydrolysis of the nicotinamide-ribose bond, which is the biologically active component. This hydrolysis is accelerated by both acidic (below pH 4) and alkaline (above pH 8) conditions, with maximum stability around pH 5 to 7. Temperature is also a strong factor, with hydrolysis rates roughly doubling for each 10 degree C increase.
In the lyophilized state, NAD+ is reasonably stable when stored cold and dry. Once in solution, its stability window is narrower than most peptides, typically 1 to 2 weeks at 2 to 8 degrees C. For longer-term storage of NAD+ solutions, freezing in single-use aliquots at -20 degrees C or colder is strongly recommended.
Light sensitivity is moderate; NAD+ absorbs UV light at 260 nm (adenine ring) and 340 nm (reduced NADH form). Light protection is recommended but less critical than for tryptophan-containing peptides.
Storage summary for NAD+:
- Lyophilized: store at -20 degrees C or colder, desiccated
- Reconstituted: refrigerate at 2-8 degrees C, use within 1-2 weeks
- Sensitive to pH extremes; maintain pH 5-7
- Shorter reconstituted shelf life than most peptides
- Aliquot and freeze for any longer-term storage
| Peptide | Key Vulnerability | Lyophilized Temp | Reconstituted Shelf Life (Fridge) | Special Requirements |
|---|---|---|---|---|
| Semaglutide | Surface adsorption, aggregation | -20 degrees C or colder | 2-7 days | Low-binding containers; pH above 7 |
| Tirzepatide | Aggregation, surface adsorption | -20 degrees C or colder | 2-7 days | Surfactant may help; low-binding containers |
| CJC-1295/Ipamorelin | DAC hydrolysis, Met oxidation | -20 degrees C or colder | 3-4 weeks | Neutral pH; light protection |
| BPC-157 | Met-14 oxidation | -20 degrees C or colder | 4-6 weeks | Amber vials; light protection essential |
| TB-500 | Aggregation | -20 degrees C or colder | 3-4 weeks | Gentle handling; moderate concentration |
| Selank | Trp photodegradation | -20 degrees C or colder | 2-3 weeks | Mandatory light protection |
| Semax | Standard heat/moisture | -20 degrees C or colder | 3-4 weeks | Standard precautions sufficient |
| Epithalon | Asp-Gly isomerization (minor) | -20 degrees C or colder | 4-6 weeks | Very stable; minimal special needs |
| GHK-Cu | Copper dissociation, oxidation | -20 degrees C or colder | 2-3 weeks | No EDTA; pH 5-7; monitor color |
| NAD+ | Nicotinamide bond hydrolysis | -20 degrees C or colder | 1-2 weeks | pH 5-7; aliquot promptly |
pH and Buffer Effects on Peptide Stability
The pH of a peptide solution influences nearly every degradation pathway discussed in this guide. Choosing the right pH and buffer system is one of the most impactful decisions a researcher can make for maximizing peptide shelf life in solution.
9.1 pH Effects on Major Degradation Pathways
Each degradation pathway has its own pH-rate profile, creating a complex optimization landscape where the ideal pH depends on which degradation pathways pose the greatest risk for a given peptide.
Deamidation is perhaps the most pH-sensitive degradation reaction. The succinimide-mediated pathway that dominates above pH 5 accelerates dramatically with increasing pH. At pH 4, the rate might be 10 to 50 times slower than at pH 8 for a typical Asn-Gly sequence. However, below pH 3, direct acid-catalyzed deamidation begins to contribute, creating a U-shaped pH-rate profile with a minimum around pH 3 to 5 (Patel & Borchardt, 1990).
Oxidation shows a more nuanced pH dependence. Metal-catalyzed oxidation (Fenton chemistry) is generally faster at acidic pH where iron is more soluble. However, autoxidation of methionine and the generation of reactive oxygen species from dissolved oxygen is often faster at alkaline pH. The net effect varies with the specific oxidation mechanism and the presence of metal ions and other catalysts.
Hydrolysis is accelerated by both acids and bases. Acid-catalyzed hydrolysis (primarily of Asp-X bonds) is fastest below pH 3. Base-catalyzed hydrolysis accelerates above pH 8. The minimum hydrolysis rate typically occurs in the pH 4 to 6 range.
Aggregation is most likely near the peptide's isoelectric point (pI), where net charge is zero and electrostatic repulsion between molecules is minimized. For most research peptides, the pI falls between pH 4 and 7. Formulating at least 1 to 2 pH units away from the pI maximizes charge-based repulsion and minimizes aggregation risk.
Racemization is base-catalyzed and accelerates with increasing pH. It is generally minimal below pH 6 and becomes significant above pH 8.
Disulfide exchange is promoted by alkaline conditions because thiolate anions (the reactive form of cysteine) are more abundant at higher pH. Maintaining pH below 7 to 7.5 minimizes disulfide scrambling risk.
For the majority of peptides, the pH "sweet spot" that minimizes the aggregate risk across all pathways falls in the range of pH 4 to 6. This is why many pharmaceutical peptide formulations are buffered in this range. However, specific peptides may have unique requirements. As discussed above, semaglutide is more stable above pH 7, which is atypical. Always consider the specific sequence and structural features of your peptide when choosing pH.
9.2 Buffer Selection
The choice of buffer impacts peptide stability beyond simply controlling pH. Different buffers can interact with peptides, catalyze degradation reactions, or provide protective effects.
Acetate buffers (pH 3.5-5.5) are widely used for acidic peptide formulations. Acetate is generally non-reactive with peptides and provides good buffering capacity in the target pH range. It's a good default choice for peptides that are stable at mildly acidic pH.
Histidine buffers (pH 5.5-7.0) have become increasingly popular for peptide and protein formulations because they provide buffering in the near-neutral range without some of the drawbacks of phosphate buffers. Histidine can also act as a mild antioxidant, scavenging reactive oxygen species and potentially protecting oxidation-sensitive peptides.
Phosphate buffers (pH 6.0-8.0) are among the most commonly used in biological research. However, they have several characteristics that can be problematic for peptide storage. Phosphate can catalyze deamidation at neutral pH. During freezing, selective crystallization of disodium phosphate causes the pH of the remaining liquid to drop by 2 to 3 units, creating transient acidic conditions that can damage pH-sensitive peptides. Despite these drawbacks, phosphate buffers remain widely used due to their excellent buffering capacity and biocompatibility.
Citrate buffers (pH 3.0-6.5) provide good buffering in the acidic range and can act as mild metal chelators, offering some protection against metal-catalyzed oxidation. They show less pH shift during freezing compared to phosphate buffers. However, citrate can sometimes promote aggregation of certain peptides through salting-out effects at higher concentrations.
Tris buffers (pH 7.0-9.0) are commonly used in molecular biology but are less ideal for peptide storage because their buffering range is above the optimal pH for many degradation-prone peptides. Tris also has a strong temperature dependence of pKa (its pH decreases by approximately 0.03 units per degree C of warming), meaning that a buffer prepared at room temperature will have a different pH when stored in the refrigerator.
For most research peptide applications, simple bacteriostatic water (without added buffer) is adequate because the peptide concentrations are typically low enough that pH doesn't shift dramatically. If pH control is needed, acetate buffer at pH 4.5 to 5.5 or histidine buffer at pH 6.0 to 6.5 are generally the best starting points.
9.3 Ionic Strength and Excipient Effects
Beyond pH and buffer identity, several other formulation factors influence peptide stability in solution:
Ionic strength. Moderate ionic strength (50 to 150 mM NaCl or equivalent) can stabilize peptides by screening unfavorable electrostatic interactions. However, high ionic strength (above 300 mM) can promote aggregation through salting-out effects, particularly for peptides near their pI. Very low ionic strength can destabilize peptides that depend on favorable electrostatic interactions for their structure.
Sugars and polyols. Trehalose, sucrose, mannitol, and sorbitol stabilize peptides in both liquid and lyophilized formulations. In solution, they preferentially exclude from the peptide surface, thermodynamically favoring the more compact native state over the unfolded state. In the lyophilized state, they form an amorphous glassy matrix that immobilizes the peptide and prevents degradation reactions. Trehalose at 5 to 10% (w/v) is the most commonly recommended stabilizer for peptide formulations (Ohtake & Wang, 2011).
Surfactants. Polysorbate 20 (Tween 20) and polysorbate 80 (Tween 80) at low concentrations (0.001 to 0.1% w/v) prevent surface adsorption and agitation-induced aggregation. They are particularly useful for hydrophobic peptides and for solutions that undergo mechanical stress during handling or shipping. However, polysorbate surfactants can generate peroxide impurities through autoxidation, so pharmaceutical-grade surfactants should be used and checked periodically for peroxide content.
Amino acid excipients. Methionine at 5 to 10 mM can serve as a sacrificial antioxidant, preferentially oxidizing before the peptide's own methionine residues. Arginine at 50 to 200 mM can reduce aggregation by interacting with hydrophobic surfaces on the peptide. Glycine is a common tonicity adjuster that can also help stabilize lyophilized cakes.
Formulation Quick Reference
For researchers who want to optimize the formulation of their peptide solutions beyond simple bacteriostatic water reconstitution, a good starting point is: 20 mM histidine buffer at pH 6.0, 150 mM NaCl, 5% (w/v) trehalose, 0.02% (w/v) polysorbate 20, and 5 mM methionine. This covers the major stability bases: pH control, ionic strength optimization, cryoprotection, surface protection, and antioxidant protection. Adjust based on your specific peptide's requirements.
Travel and Transport
Moving peptides from one location to another, whether across a building or across the country, introduces temperature excursion risks that can undermine even the most careful storage practices. Understanding cold chain logistics helps you protect your peptides during their most vulnerable moments.
10.1 Cold Chain Fundamentals
The "cold chain" refers to the continuous temperature-controlled supply chain from manufacturer to end user. For peptides, this means maintaining appropriate temperatures (typically 2 to 8 degrees C for reconstituted peptides or -20 degrees C for lyophilized peptides) throughout shipping, receiving, and internal transport.
Breaking the cold chain, even briefly, can have consequences that aren't immediately apparent. A lyophilized peptide that sits at room temperature on a loading dock for a few hours is probably fine. But the same peptide left in a delivery truck during summer, where internal temperatures can reach 60 degrees C or higher, may suffer significant degradation. The challenge is that degraded peptides often look identical to intact ones, so the damage goes unnoticed until the peptide fails to perform in an experiment.
10.2 Shipping Best Practices
For lyophilized peptides (manufacturer to researcher):
Most reputable peptide suppliers ship lyophilized peptides with cold packs or gel packs to maintain temperatures below 25 degrees C during transit. For standard domestic shipments (1 to 3 days), this is generally adequate because lyophilized peptides can tolerate brief room temperature exposure without significant degradation.
For international shipments, extended transit times, or shipping during hot weather, stronger cold chain protection is needed. Dry ice shipping maintains temperatures around -78 degrees C but requires special packaging, hazardous materials documentation, and carrier acceptance. Gel packs rated for 48 to 72 hours provide a more practical alternative for most situations.
When receiving peptide shipments, inspect the packaging immediately upon arrival. Check whether cold packs are still cool or have fully thawed. If the peptide arrives warm (ice packs fully melted, insulation warm to the touch), contact the supplier to discuss whether the product may be compromised. Reputable suppliers will replace shipments that experienced obvious cold chain failures.
For reconstituted peptides (lab to travel):
If you need to transport reconstituted peptides, for example, bringing research materials to a conference or collaborating institution, use an insulated cooler with refrigerant gel packs. Pack the vials upright in a secondary container to prevent breakage and label the cooler clearly.
Key transport guidelines for reconstituted peptides:
- Pre-chill the cooler and gel packs before loading peptides
- Wrap vials in bubble wrap or foam to prevent breakage
- Place vials away from direct contact with frozen gel packs (which can freeze the solution and cause damage). Use a towel or foam layer as a buffer.
- Include a temperature indicator strip or min/max thermometer to verify that temperatures stayed in range during transport
- Minimize transport time; ship overnight when possible
- For air travel, never check peptides in luggage. Carry them in a cooler as a carry-on item. The cargo hold of aircraft can reach temperatures below -40 degrees C, which will freeze and potentially damage reconstituted solutions.
10.3 Internal Transport
Even within a building, peptides can experience temperature stress during transfers between the freezer and the bench, or between laboratories. Some practical tips:
- Use an insulated container (even a simple styrofoam box) when moving peptides between rooms
- Don't leave peptides on the bench while preparing other materials; keep them in the cooler until ready to use
- Minimize the number of times a vial is removed from and returned to the freezer
- When pulling multiple vials from a freezer, use a pre-chilled rack or ice bucket rather than setting them on the bench
10.4 Temperature Monitoring
For critical peptide shipments and storage, temperature monitoring provides documentation that the cold chain was maintained. Options include:
Chemical temperature indicators: Single-use strips or labels that change color irreversibly when a threshold temperature is exceeded. Available for various threshold temperatures (25 degrees C, 30 degrees C, 37 degrees C). Inexpensive and easy to include in shipping packages.
Electronic data loggers: Small devices that continuously record temperature at set intervals (every 1 to 15 minutes) throughout transport. After delivery, the data can be downloaded and reviewed to identify any temperature excursions, including their duration and magnitude. More expensive than chemical indicators but provide much more detailed information.
Smart labels: NFC or RFID-enabled temperature monitors that can be read with a smartphone. These provide a middle ground between simple chemical indicators and full data loggers.
For routine peptide storage, a simple min/max thermometer in your refrigerator and freezer is sufficient. Check and record the readings weekly to identify drift or temperature excursions from power outages or equipment malfunction.
Travel Checklist
Before transporting peptides: (1) Pre-chill your cooler and gel packs. (2) Wrap vials in protective padding. (3) Include a temperature indicator. (4) Label the cooler with contents and temperature requirements. (5) Minimize transit time. (6) Check temperature indicator on arrival. (7) Transfer peptides to proper storage immediately upon arrival at the destination.
Storage Equipment and Setup
Having the right equipment doesn't need to be expensive or complicated. A basic peptide storage setup can be assembled for under $200, and even a minimal arrangement is vastly better than leaving peptides on the bench or in an unmonitored refrigerator.
11.1 Essential Equipment
Dedicated laboratory refrigerator (2-8 degrees C). A small, dedicated refrigerator for peptide and reagent storage is ideal. Kitchen-style refrigerators work fine, but choose one without a freezer compartment (which creates temperature fluctuations) or at minimum a model with separate temperature controls. Avoid mini-fridges, which often have poor temperature control and large temperature gradients between zones.
Key features to look for:
- Consistent temperature control with minimal cycling
- Door alarm for when the door is left open
- Interior light (LED preferred over fluorescent to minimize UV exposure)
- Adjustable shelves for organizing vials and boxes
- Temperature display visible without opening the door
Manual-defrost freezer (-20 degrees C). For lyophilized peptide storage, a standard chest or upright freezer set to -20 degrees C is sufficient. Manual-defrost models maintain more consistent temperatures than frost-free (auto-defrost) models, which periodically warm their interiors to melt ice buildup. If a frost-free freezer is the only option, store peptides in insulated containers within the freezer to buffer against defrost cycles.
Ultra-low freezer (-80 degrees C), optional but recommended. For long-term archival storage or master stock preservation, a -80 degrees C ultra-low freezer provides the ultimate in stability. These are expensive (typically $5,000 to $15,000) and consume significant electricity, making them a justified investment primarily for labs with large peptide inventories or long-term research programs.
11.2 Storage Containers and Vials
Amber glass vials. The standard container for reconstituted peptide storage. Amber glass blocks UV light, is chemically inert, and has low oxygen permeability. Available in common sizes (2 mL, 5 mL, 10 mL) with rubber or fluoropolymer-lined stoppers. When purchasing amber vials separately from the peptide supplier, ensure they are sterile or can be sterilized before use.
Cryovials (polypropylene). For frozen aliquots of reconstituted peptides. Standard 0.5 mL or 1.5 mL polypropylene cryovials are suitable for most applications. Ensure they are rated for the storage temperature you'll be using (-80 degrees C rated for ultra-low freezers). Screw-cap designs are preferable to snap-cap for preventing contamination and evaporation.
Desiccant packets. Small silica gel packets placed in the storage container with lyophilized peptide vials absorb ambient moisture and maintain a dry microenvironment. This is particularly useful for peptides stored in the refrigerator (where condensation can occur) or in freezers that are opened frequently.
Aluminum foil. The simplest and most effective light barrier. Wrap individual vials or entire storage boxes in foil to block all light wavelengths. Use heavy-duty foil for durability.
Storage boxes and racks. Organizing vials in labeled storage boxes prevents disorganization, reduces search time (and therefore door-open time for freezers), and provides secondary containment in case of vial breakage. Cardboard cryo-boxes with dividers are inexpensive and effective for freezer storage. Plastic or metal racks work well for refrigerator storage.
11.3 Temperature Monitoring
Min/max thermometer. A simple digital thermometer with min/max memory is the minimum requirement for any peptide storage refrigerator or freezer. Check and record the readings at least weekly. A significant change in the minimum or maximum reading can indicate equipment malfunction, power outages, or door-seal failures.
Continuous temperature logger. For more reliable monitoring, a continuous data logger records temperature at set intervals (typically every 15 to 60 minutes) and stores the data for later review. WiFi-enabled loggers can send alerts by email or text message if the temperature leaves the acceptable range, providing early warning of equipment failures.
Backup power considerations. For critical peptide inventories, consider the consequences of a power outage. A chest freezer at -20 degrees C will maintain safe temperatures for 24 to 48 hours if kept closed during the outage. An ultra-low freezer at -80 degrees C may maintain safe temperatures for only 6 to 12 hours without power. An uninterruptible power supply (UPS) or backup generator can protect against extended outages.
11.4 Minimal Setup vs. Optimal Setup
| Item | Minimal Setup | Optimal Setup | Approximate Cost |
|---|---|---|---|
| Refrigerator | Any reliable fridge with consistent temp | Dedicated lab fridge with alarm and display | $100-800 |
| Freezer | Manual defrost freezer at -20 degrees C | -80 degrees C ultra-low plus -20 degrees C | $200-15,000 |
| Vials | Original supplier vials (usually amber) | Amber glass with PTFE-lined stoppers | $20-100 |
| Aliquot tubes | Standard 1.5 mL microcentrifuge tubes | Sterile low-binding cryovials | $15-50 |
| Light protection | Aluminum foil | Amber vials plus opaque storage boxes | $5-30 |
| Temperature monitoring | Min/max thermometer | WiFi-enabled continuous logger with alerts | $15-300 |
| Desiccants | Silica gel packets | Indicating silica gel (color change when saturated) | $5-15 |
| Organization | Labeled bags or boxes | Cryo-boxes with grid dividers and database | $10-50 |
Even the minimal setup, costing under $200 total, provides dramatically better peptide preservation than leaving peptides at room temperature or in uncontrolled conditions. The optimal setup adds layers of protection and monitoring that justify the additional investment for researchers with larger inventories or higher-value peptides.
Best Practices Checklist
This section distills everything in this guide into actionable checklists you can post in your storage area or include in your standard operating procedures. Print these out. Laminate them. Tape them to your freezer door.
12.1 Receiving Peptides
- Inspect packaging immediately upon arrival
- Check temperature indicators or cold pack status
- Verify vial integrity (no cracks, intact seals, proper labeling)
- Record the lot number, receipt date, and storage location
- Transfer lyophilized peptides to -20 degrees C or colder within 30 minutes of receipt
- Contact supplier if cold chain appears compromised
- Store certificate of analysis (COA) with your records for the lot
12.2 Storing Lyophilized Peptides
- Store at -20 degrees C or colder (preferably -80 degrees C for long-term)
- Ensure vials are tightly sealed
- Include desiccant packets in the storage container
- Protect from light (amber vials, foil wrap, or opaque boxes)
- Organize in labeled boxes with an inventory system
- Record storage date and expected expiration
- Check freezer temperature weekly
- Allow vials to reach room temperature before opening (15-30 min)
- Never open a cold vial (condensation will damage the powder)
12.3 Reconstituting Peptides
- Allow lyophilized vial to reach room temperature before opening
- Use bacteriostatic water for multi-dose vials
- Use sterile water only for single-use preparations
- Inject solvent slowly along the vial wall, not directly onto the powder
- Swirl gently; never shake or vortex
- Confirm complete dissolution before use
- Label the vial with reconstitution date, concentration, and expiration date
- Refrigerate immediately at 2-8 degrees C
- See the FormBlends reconstitution guide for detailed protocols
12.4 Using Reconstituted Peptides
- Swab the vial septum with alcohol before each needle insertion
- Use a new sterile needle for each withdrawal
- Minimize the time the vial spends out of the refrigerator
- Use the FormBlends dosing calculator for accurate volume measurements
- Return the vial to the refrigerator immediately after withdrawing your dose
- Never leave a reconstituted vial at room temperature for more than 30-60 minutes
- Discard reconstituted peptides after 28-30 days (bacteriostatic water) or 3-7 days (sterile water)
- Discard if you see any visual changes (color, cloudiness, particles)
12.5 Aliquoting and Freezing
- Calculate aliquot volumes based on your per-use requirements
- Use sterile, labeled cryovials or microcentrifuge tubes
- Consider adding cryoprotectant (5-10% trehalose or sucrose)
- Freeze aliquots promptly at -20 degrees C or colder
- Record the number of aliquots, concentration, and storage location
- Thaw one aliquot at a time using gentle warming
- Never refreeze a thawed aliquot
- Use thawed aliquots within 24 hours
12.6 Ongoing Maintenance
- Check refrigerator and freezer temperatures weekly and record the readings
- Replace desiccant packets every 3-6 months or when they indicate saturation
- Audit your peptide inventory quarterly; discard expired items
- Test backup power systems regularly if applicable
- Clean storage equipment periodically to prevent microbial contamination
- Review and update your storage SOPs annually
- Train new team members on peptide handling procedures
Not sure where to start with your peptide research journey? Take the FormBlends free assessment to get personalized recommendations based on your research goals.
Frequently Asked Questions
How long do lyophilized peptides last at room temperature?
Most lyophilized peptides remain stable at room temperature (20-25 degrees C) for weeks to a few months, depending on their specific sequence and formulation. Research on lyophilized peptide vaccines showed that 17 out of 18 tested peptides were stable at room temperature for up to 3 months. However, room temperature storage should be considered temporary. For any storage period longer than a few days, move lyophilized peptides to -20 degrees C or colder. The stability gap between room temperature and frozen storage is enormous: months at best versus years at -20 degrees C. Don't gamble with your peptide inventory by leaving it on the bench when a freezer is available.
How long do reconstituted peptides last in the fridge?
Reconstituted peptides stored at 2-8 degrees C in bacteriostatic water typically remain stable for 28 to 30 days. This timeline accounts for both chemical degradation of the peptide and the diminishing antimicrobial effectiveness of the benzyl alcohol preservative in bacteriostatic water. If you used sterile water without preservative, the shelf life drops to 3-7 days due to contamination risk. Some particularly stable peptides may retain activity longer, but the preservative degradation creates a hard ceiling regardless of chemical stability. When in doubt, follow the 28-day rule and discard any reconstituted peptide older than one month.
Can you freeze reconstituted peptides?
Yes, but only if you aliquot them into single-use portions first. Freezing reconstituted peptides in single-use aliquots at -20 degrees C extends shelf life to 3-6 months, and at -80 degrees C to 1-2 years. The critical requirement is avoiding freeze-thaw cycling, which damages peptides through ice crystal formation, freeze-concentration, and pH shifts. Never freeze an entire multi-dose vial and then thaw and refreeze it repeatedly. Instead, divide the solution into individual portions, freeze them all, and thaw only what you need for each use session. Adding 5-10% trehalose or sucrose as a cryoprotectant improves stability during freezing.
How do you know if a peptide has degraded?
Visual signs include color changes (yellowing or browning from oxidation), cloudiness (aggregation), visible particles or precipitate, and collapsed or discolored lyophilized cake. Functional signs include reduced efficacy at the usual dose, inconsistent experimental results, or increased injection site reactions in animal studies. However, many forms of degradation produce no visible changes, which is why proper storage is so much more reliable than visual inspection. For definitive confirmation, analytical methods like HPLC and mass spectrometry can quantify degradation. As a practical rule, if you have concerns about storage history or the peptide is past its recommended use-by date, discard and replace.
Does bacteriostatic water expire?
Unopened bacteriostatic water has a shelf life typically listed as 2-5 years from manufacturing. Once the vial is punctured, the preservative (0.9% benzyl alcohol) begins to degrade, and the sterile barrier is compromised. An opened vial of bacteriostatic water should be used within 28 days and then discarded. During weeks 1-4, bacterial inhibition remains strong with minimal contamination risk when proper aseptic technique is used. From weeks 4-6, the preservative weakens and contamination becomes possible. Beyond 6 weeks, the preservative is largely ineffective. Mark the date of first puncture on every bacteriostatic water vial and adhere to the 28-day limit.
What happens if peptides get too warm during shipping?
The impact depends on how warm and for how long. Lyophilized peptides can generally tolerate room temperature (20-25 degrees C) for several days without measurable degradation. Brief exposure to 30-40 degrees C (a few hours) is typically tolerable for lyophilized peptides, though not ideal. Temperatures above 50 degrees C can cause lyophilized cake collapse, moisture absorption, and accelerated degradation. For reconstituted peptides, the window is much narrower; room temperature exposure should be limited to minutes or hours. If your shipment arrives with fully thawed ice packs and warm insulation, contact the supplier. Many reputable companies will replace shipments that experienced obvious cold chain failures.
Should peptides be stored in amber vials or does aluminum foil work?
Both provide effective light protection, and either is far better than leaving peptides in clear glass under ambient lighting. Amber glass vials block UV light below approximately 450 nm, covering the wavelengths most damaging to peptides (the absorption ranges of tryptophan, tyrosine, and disulfide bonds). Aluminum foil provides complete light blocking across all wavelengths and is therefore technically more effective. In practice, peptides shipped in amber vials from the supplier are adequately protected. If your peptides arrive in clear glass, wrap them in aluminum foil immediately. For maximum protection, use both: store amber vials inside foil-wrapped or opaque storage containers.
Can I use the same syringe to draw from a peptide vial multiple times?
No. Use a new, sterile needle and syringe for each withdrawal from a peptide vial. Reusing needles introduces contamination risk (bacteria from your skin or the environment can be carried into the vial on the needle), and repeated punctures of the septum can compromise the seal, allowing air and moisture to enter. Each puncture also increases the cumulative risk of coring (cutting a small piece of rubber from the septum that falls into the solution). Use an insulin syringe or tuberculin syringe with a fine-gauge needle (27G to 30G) and penetrate the septum at a slight angle to minimize coring. Swab the septum with an alcohol pad before each puncture.
What's the difference between bacteriostatic water and sterile water for reconstitution?
Bacteriostatic water is sterile water that contains 0.9% benzyl alcohol as a preservative. This preservative inhibits bacterial growth, making bacteriostatic water suitable for multi-dose vials that will be accessed multiple times over several weeks. Sterile water for injection contains no preservative. It's used for single-use preparations where the entire solution will be consumed at once. For research peptides that will be dosed over multiple sessions, bacteriostatic water is the standard choice because it extends the usable life of the reconstituted solution from days to approximately 4 weeks. See our reconstitution guide for complete details on solvent selection.
Is it safe to use a peptide past its expiration date?
Expiration dates on peptide products reflect the manufacturer's stability data and represent the last date at which the product is guaranteed to meet its stated purity and potency specifications. Using a peptide past this date doesn't mean it's suddenly toxic or completely inactive, but it means the manufacturer can no longer guarantee its quality. Lyophilized peptides stored at -20 degrees C or colder often remain viable well past their stated expiration, while reconstituted peptides should always be discarded on or before their use-by date. If you choose to use an expired lyophilized peptide, consider having it tested by HPLC to confirm its purity before incorporating it into research.
How should I store peptides during a power outage?
The most important action is keeping your freezer and refrigerator doors closed. A full chest freezer at -20 degrees C can maintain safe temperatures for 24-48 hours without power if kept sealed. An upright freezer maintains temperatures for 12-24 hours. A refrigerator will stay cool for approximately 4 hours with the door closed. If you have advance warning of an outage (planned maintenance, approaching storm), transfer your most valuable peptides to a colleague's freezer, pack them on dry ice in a well-insulated cooler, or add extra ice packs to your storage containers. For labs in outage-prone areas, a backup generator or UPS for the peptide freezer is a worthwhile investment.
Do different peptides need different storage conditions?
The general principles are the same for all peptides: cold, dark, dry for lyophilized; cold, dark, and used promptly for reconstituted. However, specific peptides have unique vulnerabilities that warrant extra attention. Metal-containing peptides like GHK-Cu require pH control (5-7) and should not be stored with chelating agents. Tryptophan-containing peptides like Selank need rigorous light protection. GLP-1 agonists like semaglutide are prone to surface adsorption and benefit from low-binding containers. See the Peptide-Specific Storage Guide section of this report for detailed compound-by-compound recommendations.
Can I travel with peptides on an airplane?
Yes, with proper planning. Carry peptides in your carry-on luggage, never in checked bags, because cargo holds experience extreme temperature swings (as low as -40 degrees C). Use a small insulated cooler with gel packs to maintain 2-8 degrees C for reconstituted peptides. Lyophilized peptides in sealed vials can tolerate room temperature for the duration of most flights without concern. If carrying needles and syringes, bring documentation (a letter from your research institution) as they may be questioned at security. Many countries have specific regulations about importing biological materials, so check destination country rules if traveling internationally. TSA and most security agencies allow small coolers with gel packs through carry-on screening.
How many times can I freeze and thaw a peptide solution?
Ideally zero, which is why aliquoting into single-use portions is so strongly recommended. Each freeze-thaw cycle subjects the peptide to ice crystal damage, freeze-concentration stress, pH shifts, and surface-induced aggregation. After 3-5 cycles, most peptides show measurable degradation. After 10 or more cycles, significant activity loss is common. If you must freeze-thaw (because you forgot to aliquot), limit it to 2-3 cycles maximum, thaw rapidly (hand warming or room temp water bath), and add a cryoprotectant like 5-10% trehalose before the first freeze. But the real solution is to always aliquot before the first freeze.
References
- Manning MC, Patel K, Borchardt RT. Stability of protein pharmaceuticals. Pharm Res. 1989;6(11):903-918. doi:10.1023/A:1015929109894
- Patel K, Borchardt RT. Chemical pathways of peptide degradation. II. Kinetics of deamidation of an asparaginyl residue in a model hexapeptide. Pharm Res. 1990;7(7):703-711. doi:10.1023/A:1015807303766
- Geiger T, Clarke S. Deamidation, isomerization, and racemization at asparaginyl and aspartyl residues in peptides. Succinimide-linked reactions that contribute to protein degradation. J Biol Chem. 1987;262(2):785-794. doi:10.1016/S0021-9258(19)75855-4
- Li S, Schoneich C, Borchardt RT. Chemical instability of protein pharmaceuticals: Mechanisms of oxidation and strategies for stabilization. Biotechnol Bioeng. 1995;48(5):490-500. doi:10.1002/bit.260480511
- Robinson NE, Robinson AB. Molecular clocks: deamidation of asparaginyl and glutaminyl residues in peptides and proteins. Cave Junction, OR: Althouse Press; 2004. doi:10.1016/S0021-9258(19)36821-6
- Cleland JL, Powell MF, Shire SJ. The development of stable protein formulations: a close look at protein aggregation, deamidation, and oxidation. Crit Rev Ther Drug Carrier Syst. 1993;10(4):307-377. PMID:8124728
- Kerwin BA, Remmele RL Jr. Protect from light: photodegradation and protein biologics. J Pharm Sci. 2007;96(6):1468-1479. doi:10.1002/jps.20815
- Qi P, Volkin DB, Zhao H, et al. Characterization of the photodegradation of a human IgG1 monoclonal antibody formulated as a high-concentration liquid dosage form. J Pharm Sci. 2009;98(9):3117-3130. doi:10.1002/jps.21617
- Moussa EM, Panchal JP, Moorthy BS, et al. Immunogenicity of therapeutic protein aggregates. J Pharm Sci. 2016;105(2):417-430. doi:10.1016/j.xphs.2015.11.002
- Ohtake S, Wang YJ. Trehalose: current use and future applications. J Pharm Sci. 2011;100(6):2020-2053. doi:10.1002/jps.22458
- Chang BS, Beauvais RM, Dong A, Carpenter JF. Physical factors affecting the storage stability of freeze-dried interleukin-1 receptor antagonist: glass transition and protein conformation. Arch Biochem Biophys. 2005;331(2):249-258. doi:10.1006/abbi.1996.0305
- Slingluff CL Jr, Yamshchikov G, Neese P, et al. Phase I trial of a melanoma vaccine with gp100(280-288) peptide and tetanus helper peptide in adjuvant: immunologic and clinical outcomes. Clin Cancer Res. 2001;7(10):3012-3024. PMID:11595689
- Zapadka KL, Becher FJ, Gomes Dos Santos AL, Jackson SE. Factors affecting the physical stability (aggregation) of peptide therapeutics. Interface Focus. 2017;7(6):20170030. doi:10.1098/rsfs.2017.0030
- Goolcharran C, Khossravi M, Borchardt RT. Chemical pathways of peptide and protein degradation. In: Frokjaer S, Hovgaard L, eds. Pharmaceutical Formulation Development of Peptides and Proteins. London: Taylor & Francis; 2000:70-108. doi:10.1201/9780367801069
- Hawe A, Wiggenhorn M, van de Weert M, Garbe JHO, Mahler HC, Jiskoot W. Forced degradation of therapeutic proteins. J Pharm Sci. 2012;101(3):895-913. doi:10.1002/jps.22812
- Bak A, Leung D, Barrett SE, et al. Physicochemical and formulation developability assessment for therapeutic peptide delivery: a primer. AAPS J. 2015;17(1):144-155. doi:10.1208/s12248-014-9688-2
- Gervasi V, Dall Agnol R, Cullen S, McCoy T, Vucen S, Crean A. Parenteral protein formulations: an overview of approved products within the European Union. Eur J Pharm Biopharm. 2018;131:8-24. doi:10.1016/j.ejpb.2018.07.011
- Kamerzell TJ, Esfandiary R, Joshi SB, Middaugh CR, Volkin DB. Protein-excipient interactions: mechanisms and biophysical characterization applied to protein formulation development. Adv Drug Deliv Rev. 2011;63(13):1118-1159. doi:10.1016/j.addr.2011.07.006
- Wang W. Instability, stabilization, and formulation of liquid protein pharmaceuticals. Int J Pharm. 1999;185(2):129-188. doi:10.1016/S0378-5173(99)00152-0
- Carpenter JF, Pikal MJ, Chang BS, Randolph TW. Rational design of stable lyophilized protein formulations: some practical advice. Pharm Res. 1997;14(8):969-975. doi:10.1023/A:1012180707283
- Anik ST, Hwang J. Adsorption of D-Nal(2)6LHRHa, a decapeptide, onto glass and other surfaces. Int J Pharm. 1983;16(2):181-190. doi:10.1016/0378-5173(83)90055-7
- Fransson J, Florin-Robertsson E, Axelsson K, Nylander C. Oxidation of human insulin-like growth factor I in formulation studies: kinetics of methionine oxidation in aqueous solution and in solid state. Pharm Res. 1996;13(8):1252-1257. doi:10.1023/A:1016032808039
- Torosantucci R, Schoneich C, Jiskoot W. Oxidation of therapeutic proteins and peptides: structural and biological consequences. Pharm Res. 2014;31(3):541-553. doi:10.1007/s11095-013-1199-9
- Arora S, et al. Influence of buffering capacity, pH, and temperature on the stability of semaglutide: a preformulation study. J Pharm Sci. 2025. doi:10.1016/j.xphs.2025.01.012
- Davies MJ. Photo-degradation of therapeutic proteins: mechanistic aspects. Pharm Res. 2020;37(3):44. doi:10.1007/s11095-020-2763-8
- Strickley RG, Lambert WJ. A review of formulations of commercially available antibodies. J Pharm Sci. 2021;110(7):2590-2608. doi:10.1016/j.xphs.2021.03.017
- Shire SJ. Formulation and manufacturability of biologics. Curr Opin Biotechnol. 2009;20(6):708-714. doi:10.1016/j.copbio.2009.10.006
- Jiskoot W, Randolph TW, Volkin DB, et al. Protein instability and immunogenicity: roadblocks to clinical application of injectable protein delivery systems for sustained release. J Pharm Sci. 2012;101(3):946-954. doi:10.1002/jps.23018
- Ugwu SO, Apte SP. The effect of buffers on protein conformational stability. Pharm Technol. 2004;28(3):86-108.
- Kolhe P, Amend E, Singh SK. Impact of freezing on pH of buffered solutions and consequences for monoclonal antibody aggregation. Biotechnol Prog. 2010;26(3):727-733. doi:10.1002/btpr.377
- Piedmonte DM, Summers C, McAuley A, Kaber L, Bhatt A, de Vries T. Sorbitol crystallization can lead to protein aggregation in frozen protein formulations. Pharm Res. 2007;24(1):136-146. doi:10.1007/s11095-006-9131-1
- Murase N, Franks F. Salt precipitation during the freeze-concentration of phosphate buffer solutions. Biophys Chem. 1989;34(3):293-300. doi:10.1016/0301-4622(89)80066-3
- Stadtman ER. Oxidation of free amino acids and amino acid residues in proteins by radiolysis and by metal-catalyzed reactions. Annu Rev Biochem. 1993;62:797-821. doi:10.1146/annurev.bi.62.070193.004053
- Wakankar AA, Borchardt RT. Formulation considerations for proteins susceptible to asparagine deamidation and aspartate isomerization. J Pharm Sci. 2006;95(11):2321-2336. doi:10.1002/jps.20740
- Zbacnik TJ, Holcomb RE, Katayama DS, et al. Role of buffers in protein formulations. J Pharm Sci. 2017;106(3):713-733. doi:10.1016/j.xphs.2016.11.014
- Tscheliessnig R, Konrath J, Bates R, Jungbauer A. Host cell protein analysis in therapeutic protein bioprocessing: methods and applications. Biotechnol J. 2013;8(6):655-670. doi:10.1002/biot.201200018
- Akers MJ. Excipient-drug interactions in parenteral formulations. J Pharm Sci. 2002;91(11):2283-2300. doi:10.1002/jps.10154
- Crommelin DJA, Storm G, Jiskoot W, Stenekes R, Mastrobattista E, Hennink WE. Nanotechnological approaches for the delivery of macromolecules. J Control Release. 2003;87(1-3):81-88. doi:10.1016/S0168-3659(03)00014-2
- Pikal MJ. Freeze-drying of proteins. Part I: process design. BioPharm. 1990;3(8):18-27.
- Tang X, Pikal MJ. Design of freeze-drying processes for pharmaceuticals: practical advice. Pharm Res. 2004;21(2):191-200. doi:10.1023/B:PHAM.0000016234.73023.75
The Science of Peptide Lyophilization
Lyophilization is so central to peptide stability that it deserves a deeper exploration. Understanding how freeze-drying works at a molecular level helps researchers appreciate why lyophilized peptides are so stable, what can go wrong during the process, and how to evaluate the quality of lyophilized products they receive.
15.1 The Lyophilization Process
Lyophilization is a three-stage process: freezing, primary drying, and secondary drying. Each stage has specific requirements that influence the quality and stability of the final product.
Freezing. The peptide solution is cooled below its eutectic temperature (the temperature at which all components of the solution are frozen). For most peptide formulations, this requires cooling to -40 to -50 degrees C. The freezing rate influences the size of ice crystals that form. Slow freezing produces large ice crystals with large pores in the dried cake, while fast freezing produces small crystals with small pores. Larger pores generally facilitate faster primary drying and faster reconstitution. Some lyophilization processes include an "annealing" step, where the frozen product is briefly warmed and then refrozen to increase ice crystal size and improve pore structure.
During freezing, the same freeze-concentration effects discussed in Section 6 occur. The peptide, buffer salts, and other solutes are concentrated into channels between ice crystals. If the formulation contains crystallizable excipients (such as mannitol or glycine), they may crystallize out of this concentrated phase, potentially leaving the peptide less protected. Amorphous excipients (such as trehalose and sucrose) remain in the concentrated liquid phase with the peptide, providing better protection.
Primary drying (sublimation). After freezing is complete, the chamber pressure is reduced to below the vapor pressure of ice (typically 50 to 200 mTorr), and the shelf temperature is raised slightly (to -10 to -30 degrees C). Under these conditions, ice sublimes directly from solid to vapor without passing through the liquid phase. This is the key advantage of lyophilization over simple evaporative drying: the peptide never experiences the high concentrations and extended solution exposure that would occur during conventional drying.
Primary drying removes approximately 95% of the water in the sample, corresponding to the ice that formed during freezing. The drying front progresses from the surface of the cake inward, as water vapor escapes through the porous structure left behind by the sublimed ice. Primary drying is typically the longest stage, lasting 24 to 72 hours depending on the fill volume and cake thickness.
The critical parameter during primary drying is the product temperature at the sublimation front. If this temperature exceeds the collapse temperature of the formulation (typically -20 to -35 degrees C for amorphous formulations), the dried cake above the sublimation front loses its structure and collapses. A collapsed cake has reduced surface area, higher residual moisture, longer reconstitution time, and potentially compromised peptide stability. Product temperature is controlled indirectly through the shelf temperature and chamber pressure.
Secondary drying (desorption). After all ice has been removed by sublimation, the shelf temperature is raised further (to 20 to 40 degrees C) and maintained for several hours to remove the remaining "bound" or "unfrozen" water that is associated with the solute phase. This typically reduces the residual moisture content from approximately 5 to 10% (after primary drying) to 1 to 3% (after secondary drying). The target residual moisture depends on the specific peptide and formulation. Some peptides are more stable at slightly higher moisture levels because a small amount of water can plasticize the glassy matrix and reduce mechanical stress on the peptide.
15.2 The Glass Transition and Molecular Mobility
The concept of glass transition temperature (Tg) is central to understanding lyophilized peptide stability. When a solution is frozen and dried, the remaining solute phase forms an amorphous solid, or "glass." This glass has the structural disorder of a liquid but the mechanical rigidity of a solid. The glass transition temperature is the temperature at which this amorphous solid transitions from a rigid, glassy state to a more fluid, rubbery state.
Below Tg, molecular mobility is extremely restricted. Chemical reactions require molecular movement to bring reactants together, so reactions in the glassy state proceed at negligible rates. Above Tg, molecular mobility increases rapidly with temperature, and degradation reactions can proceed at appreciable rates. For optimal stability, lyophilized peptides should be stored well below their Tg.
The Tg of a lyophilized peptide formulation depends on its composition. Pure trehalose has a Tg of approximately 115 degrees C, pure sucrose about 65 degrees C, and pure peptide typically 100 to 200 degrees C depending on size. Residual water acts as a potent plasticizer, reducing Tg by approximately 10 degrees C for each 1% increase in moisture content. This is why moisture control is so critical: a formulation with a Tg of 80 degrees C when properly dried (1% moisture) might have a Tg of only 30 degrees C at 5% moisture, meaning that room temperature storage would place it above its glass transition.
For most well-formulated, properly dried lyophilized peptides, the Tg is well above room temperature (often 50 to 100 degrees C), providing a comfortable stability margin for storage at -20 degrees C or even at room temperature for moderate periods. However, formulations with high sugar content, high residual moisture, or exposure to humid conditions may have lower Tg values that make room temperature storage risky.
15.3 Evaluating Lyophilized Cake Quality
The visual appearance of a lyophilized cake provides useful information about the quality of the freeze-drying process and the likely stability of the product:
Ideal cake: Uniform, white to off-white, fluffy texture. The cake fills the vial to approximately the same level as the original solution. The surface is smooth or slightly textured. The cake may have small pores visible to the naked eye. It dissolves rapidly and completely when solvent is added.
Partially collapsed cake: The cake has shrunk from the vial walls, forming a smaller plug that may be tilted or displaced. The surface may appear glassy, translucent, or rough rather than uniformly opaque. Partial collapse suggests that the product temperature exceeded the collapse temperature during primary drying. The peptide may still be usable, but residual moisture may be higher than optimal, and reconstitution may take longer.
Fully collapsed cake: The original cake structure is completely lost. The material appears as a thin film, dense plug, or amorphous residue at the bottom of the vial. This indicates significant process failure or post-lyophilization heat exposure. Residual moisture is likely elevated, and degradation may have occurred. Test before use if possible; replace if available.
Back-splash or skin: A thin, glassy layer on the top surface of the cake, caused by splashing of the solution during loading or melting of the top layer during primary drying. The underlying cake may be normal. Back-splash usually doesn't significantly affect product quality but can slow reconstitution.
Meltback: The entire cake has melted and refrozen during storage, producing a solid, ice-like mass. This indicates severe temperature excursion (storage temperature exceeded the eutectic or collapse temperature). The product should be discarded.
15.4 Excipients and Their Roles in Lyophilized Formulations
Lyophilized peptide formulations typically contain several excipients beyond the peptide itself. Understanding what each component does helps researchers make informed decisions about storage and handling.
Bulking agents (mannitol, glycine) provide physical structure to the lyophilized cake. Without a bulking agent, small quantities of peptide might dry as a thin, barely visible film that's difficult to handle and reconstitute. Bulking agents typically crystallize during freezing, forming a matrix that gives the cake its characteristic appearance and mechanical strength. They have minimal direct stabilizing effect on the peptide.
Lyoprotectants (trehalose, sucrose) stabilize the peptide during and after lyophilization. They work through two mechanisms. During freezing, they preferentially exclude from the peptide surface, thermodynamically favoring the native conformation (the "preferential exclusion" mechanism). After drying, they form an amorphous glass that hydrogen-bonds to the peptide surface, substituting for the water molecules that were removed. This "water replacement" hypothesis explains why lyoprotectants that form stable glasses with high Tg values (like trehalose) are more effective stabilizers than those with lower Tg values.
Buffer salts (phosphate, histidine, citrate) maintain pH during reconstitution and, in some formulations, during the freezing process itself. As discussed in Section 9, different buffers behave differently during freezing, with phosphate being particularly problematic due to selective crystallization of the dibasic form.
Tonicity adjusters (NaCl, sorbitol) ensure that the reconstituted solution is isotonic for injection applications. NaCl is the most common, but it can reduce the Tg of the lyophilized formulation by remaining amorphous in the solute phase. Sorbitol is sometimes preferred because it can crystallize during freezing, contributing to cake structure rather than reducing Tg.
Surfactants (polysorbate 20, polysorbate 80) prevent surface adsorption and aggregation during reconstitution. They are particularly useful for hydrophobic peptides that tend to adsorb to glass surfaces and air-water interfaces. However, as mentioned earlier, polysorbate surfactants can degrade over time to produce peroxides that oxidize the peptide, so they should be used at the minimum effective concentration.
Advanced Stability Considerations
Beyond the fundamental principles covered earlier, several advanced topics influence peptide stability in ways that become relevant for large-scale research programs, long-term studies, and situations where standard protocols need to be adapted to unusual circumstances.
16.1 Container Closure Integrity
The container closure system, the combination of vial, stopper, and seal, is a critical barrier against environmental threats to peptide stability. A compromised container allows moisture, oxygen, and microbial contaminants to reach the peptide, accelerating degradation regardless of how carefully the storage environment is controlled.
Glass vials. Borosilicate glass (Type I) is the standard material for pharmaceutical vials. It has low extractable content, excellent chemical resistance, and minimal interaction with peptide solutions. However, glass surfaces can adsorb peptides (particularly hydrophobic ones), reducing the effective concentration in solution. Silicone-coated or siliconized vials reduce surface adsorption but introduce the possibility of silicone oil particles appearing in the solution, which can be confused with protein aggregates.
Delamination, the flaking of the inner glass surface, can occur in certain types of glass vials, particularly those subjected to aggressive sterilization or long-term storage of alkaline solutions. Glass flakes in the solution are a serious quality concern. Using high-quality Type I borosilicate glass from reputable suppliers minimizes this risk.
Rubber stoppers. The stopper is often the weakest link in the container closure system. Standard butyl rubber stoppers can leach extractable compounds (metals, organic compounds, sulfur-containing species) into the peptide solution, potentially catalyzing oxidation and other degradation reactions. Stoppers also have finite permeability to oxygen and moisture vapor, allowing slow gas exchange between the vial contents and the environment.
Fluoropolymer-coated stoppers (with PTFE or fluoroelastomer linings on the product-contact surface) dramatically reduce extractables and leachables. They also provide a better barrier against moisture and oxygen permeation. For high-value or long-term stored peptides, coated stoppers are strongly recommended.
Stopper swell and coring are additional concerns. If a stopper swells from contact with the solution, the seal can be compromised. Coring occurs when a needle cuts a small piece of rubber from the stopper during puncture, which then falls into the solution. Using proper needle insertion technique (angled insertion with the bevel up) and limiting the number of punctures reduces coring risk.
Crimped aluminum seals. The aluminum crimp seal secures the stopper in place and provides a tamper-evident closure. Improperly crimped seals can allow the stopper to shift, breaking the seal and exposing the contents to the atmosphere. When receiving peptide vials, check that the crimp seal is intact and properly seated. Any vials with loose, damaged, or missing crimp seals should be rejected.
16.2 Moisture Control in Detail
Moisture is the most insidious threat to lyophilized peptide stability because its effects are invisible until significant damage has occurred. Even small amounts of absorbed moisture can dramatically accelerate degradation by mobilizing reactants and reducing the glass transition temperature of the lyophilized matrix.
Sources of moisture ingress include:
- Stopper moisture transmission: Rubber stoppers are not completely impermeable to water vapor. Over months to years, moisture from the ambient atmosphere permeates through the stopper into the vial. The rate depends on the stopper material, thickness, temperature, and humidity gradient. Fluoropolymer-coated stoppers reduce but don't eliminate moisture transmission.
- Headspace air: The air trapped in the vial headspace during sealing contains moisture at the ambient relative humidity. If the vial was sealed in a humid environment (for example, without humidity control on the lyophilizer), the headspace moisture can be significant. Nitrogen or argon purging of the vial headspace before sealing reduces both moisture and oxygen content.
- Opening and closing: Each time a lyophilized vial is opened, ambient air (with its moisture content) enters. If only a portion of the lyophilized powder is used, the remainder is exposed to this moisture. This is one reason why lyophilized peptides should ideally be reconstituted entirely in a single session rather than repeatedly opened for partial use.
- Condensation: Removing a cold vial from the freezer and opening it before it has reached room temperature causes warm, humid air to condense on the cold surfaces inside the vial, including the lyophilized powder. This is why allowing vials to equilibrate to room temperature before opening is so strongly emphasized throughout this guide.
Desiccant use provides a secondary moisture barrier. Silica gel packets placed in the outer container (zip-lock bag, screw-cap jar, or storage box) surrounding the peptide vials absorb moisture from the microenvironment, creating a drier local atmosphere. Indicating silica gel changes color (typically from orange to dark green or from blue to pink) when it has absorbed its capacity of moisture, signaling the need for replacement.
For critical applications where moisture control is paramount, sealed aluminum pouches with desiccant provide the best protection. The aluminum is completely impermeable to moisture and oxygen, and the desiccant absorbs any residual moisture inside the pouch. Some peptide suppliers ship lyophilized products in this format, and it's the preferred packaging for long-term archival storage.
16.3 Accelerated Stability Testing: Predicting Long-Term Stability
Accelerated stability testing is a pharmaceutical development technique that uses elevated temperatures to predict long-term stability at lower storage temperatures. While primarily a tool for product development rather than routine research, understanding the principles helps researchers interpret the stability data provided by suppliers and make informed judgments about their own peptide inventories.
The basic approach involves storing peptide samples at multiple elevated temperatures (typically 25, 40, and 60 degrees C) and measuring degradation at regular intervals (weekly to monthly). The degradation rate at each temperature is then plotted against inverse temperature (an Arrhenius plot), and the resulting line is extrapolated to predict the degradation rate at the intended storage temperature (for example, 5 degrees C for refrigerated products or -20 degrees C for frozen products).
This approach works well for simple, single-pathway degradation but has limitations. If the dominant degradation pathway changes with temperature (for example, if aggregation dominates at 40 degrees C but deamidation dominates at 5 degrees C), the Arrhenius extrapolation may overestimate or underestimate the actual degradation rate at the target temperature. For this reason, accelerated stability data should be viewed as a useful screening tool rather than a definitive prediction.
ICH (International Council for Harmonisation) guidelines define standard accelerated stability conditions for pharmaceutical products. For products intended for refrigerated storage (2 to 8 degrees C), accelerated testing at 25 degrees C and 60% relative humidity is standard, with stressed testing at 40 degrees C and 75% relative humidity. For products intended for freezer storage, accelerated testing at 5 degrees C is common.
For researchers evaluating their own peptide inventory, a simple version of this approach can be useful. If you're concerned about a peptide that may have experienced temperature excursions, you can compare its analytical profile (HPLC purity, visual appearance) to a known-good sample. Any difference beyond the analytical method's variability suggests degradation has occurred.
16.4 Humidity Considerations for Different Climates
Researchers in different geographic locations face different humidity challenges. A laboratory in a dry desert climate (10 to 20% relative humidity) has less moisture ingress risk than a laboratory in a tropical or coastal environment (60 to 90% relative humidity). This humidity difference affects the practical shelf life of lyophilized peptides, particularly those stored at room temperature or in non-hermetically sealed containers.
In high-humidity environments:
- Use hermetically sealed containers (aluminum pouches or tightly sealed vials with crimp seals) for all lyophilized storage
- Include desiccant in every storage container and replace more frequently (every 1 to 2 months)
- Minimize time that vials spend open to the atmosphere during reconstitution
- Consider reconstituting in a low-humidity environment (air-conditioned lab with dehumidifier) if ambient humidity is very high
- Store all opened bacteriostatic water vials tightly sealed with parafilm over the septum to reduce moisture exchange
In low-humidity environments, the main risk shifts from moisture ingress to static electricity. Very dry air can generate static charges on vial surfaces that attract fine lyophilized powder, causing it to cling to the vial walls and stopper rather than settling to the bottom. This can lead to product loss and difficulty reconstituting. Anti-static mats and ionizing air blowers in the preparation area help manage this issue.
16.5 Peptide Stability in Multi-Compound Protocols
Many research protocols use multiple peptides simultaneously, raising questions about compatibility and co-storage. Can different peptides be stored in the same vial? Can they be reconstituted together? Will they interact and affect each other's stability?
In general, different peptides should be stored separately in the lyophilized state. Mixing lyophilized powders from different vials introduces contamination risk and makes it impossible to accurately dose individual components. Each peptide should maintain its own dedicated vial with clear labeling.
For reconstituted peptides, some combinations are well-established and known to be compatible. For example, CJC-1295 and Ipamorelin are commonly reconstituted together without stability issues. BPC-157 and TB-500 are also frequently combined. However, mixing peptides without known compatibility data is risky because interactions between different peptide molecules can promote aggregation, coprecipitation, or chemical cross-linking.
When using multiple peptides in a research protocol, the safest approach is to store and reconstitute each peptide separately and combine them only immediately before use, at the point of administration. This eliminates the extended solution contact time during which unfavorable interactions might occur.
Some specific compatibility concerns to be aware of:
- Peptides with very different optimal pH ranges should not be mixed in solution, as the compromise pH may be suboptimal for both
- Metal-containing peptides (like GHK-Cu) should not be mixed with peptides that contain free cysteine residues, as the metal can catalyze disulfide formation or thiol oxidation
- Highly concentrated mixed solutions are more likely to form aggregates than dilute mixtures
- If mixing peptides, use the shorter of the two individual shelf lives as the expiration for the combined solution
Real-World Scenarios and Troubleshooting
Theory is essential, but practice is where problems actually occur. This section addresses the most common real-world situations researchers encounter and provides concrete guidance for each scenario.
Scenario 1: "My peptide was left at room temperature overnight."
Lyophilized: Almost certainly fine. Lyophilized peptides tolerate room temperature for weeks. Return it to the freezer and use normally. No action needed beyond documenting the excursion for your records.
Reconstituted: Probably okay for a single overnight event, but use it promptly. One night (8 to 12 hours) at 20 to 25 degrees C exposes the peptide to additional degradation equivalent to roughly 2 to 4 days of additional aging at refrigerator temperature. If the reconstituted peptide was already near its 28-day limit, consider discarding it. If it was freshly reconstituted, return it to the refrigerator and continue using it with the understanding that its remaining shelf life has been somewhat reduced. Check for visual changes (cloudiness, color) before the next use.
Scenario 2: "My freezer died and the peptides thawed."
For lyophilized peptides: Assess how warm they got and for how long. If the freezer simply failed and temperatures rose to room temperature over several hours, lyophilized peptides are very likely fine. Return them to a working freezer or pack on dry ice until a replacement freezer is available. If temperatures exceeded 40 degrees C (possible in a sealed, powered-off freezer in a warm room over several days), evaluate cake quality visually and consider analytical testing.
For frozen reconstituted aliquots: These are more concerning because they experienced a full freeze-thaw cycle. If they thawed slowly (over hours) and remained cold (below 8 degrees C), the damage is likely moderate. If they warmed to room temperature, the combination of freeze-thaw stress plus extended ambient temperature exposure may have caused significant degradation. Use the aliquots promptly (within 1 to 2 days) rather than refreezing, or discard them if you have replacement stock available.
Scenario 3: "I accidentally shook/vortexed my reconstituted peptide."
Agitation creates air-liquid interfaces that can promote aggregation. The severity depends on how vigorously and for how long the peptide was agitated. Brief shaking (a few seconds) during handling is generally inconsequential. Vortexing at high speed for 30 seconds or more can cause measurable aggregation in sensitive peptides.
After accidental agitation, let the vial sit undisturbed in the refrigerator for 30 to 60 minutes, then inspect for cloudiness or foam. If the solution remains clear and any foam dissipates, the peptide is likely fine. If persistent cloudiness or particulates appear, the solution may need to be discarded. For future reference, always mix reconstituted peptides by gentle swirling or slow inversion, never by shaking or vortexing.
Scenario 4: "I reconstituted too much and need to store the excess."
This is one of the most common situations. You reconstituted a full vial but only need a fraction of it in the next few weeks. The best approach is immediate aliquoting and freezing:
- Calculate how much you'll use in the next 28 days and keep that portion in the original vial, refrigerated
- Divide the remainder into single-use aliquots in labeled cryovials
- Freeze the aliquots immediately at -20 degrees C or colder
- Thaw one aliquot at a time as needed
If you don't have aliquoting supplies available immediately, refrigerate the entire vial and aliquot within 24 hours. The key is to minimize the time the excess peptide spends in solution at refrigerator temperature before being frozen.
Scenario 5: "My peptide solution turned slightly yellow."
Yellowing indicates oxidative degradation, most commonly of tryptophan residues. The degree of yellowing correlates roughly with the extent of degradation, but not precisely. Slight yellowing (barely noticeable compared to fresh solution) may represent only 2 to 5% degradation, which might be acceptable for some applications. Pronounced yellowing (clearly visible golden or amber color) suggests more extensive damage, possibly 10 to 30% or more degradation.
If slight yellowing is observed, consider the following: How old is the reconstituted solution? Has it been properly protected from light? How much of the total supply does this vial represent? If you have replacement stock, discarding the yellowed vial is the safest option. If replacement isn't immediately available and the yellowing is slight, the peptide may still retain sufficient activity for research purposes, but be aware that results may be attenuated compared to fresh peptide.
To prevent recurrence, review your light protection measures. Ensure vials are stored in amber glass, wrapped in foil, or kept in opaque containers. Check that your refrigerator doesn't have a continuously illuminated interior light.
Scenario 6: "I need to ship peptides to a collaborator."
For lyophilized peptides, shipping is straightforward. Pack the vials with sufficient insulation (styrofoam box, insulated pouch) and gel packs to maintain temperatures below 25 degrees C for the expected transit time. Overnight shipping is preferred. Include a temperature indicator strip to document that the cold chain was maintained. Ship early in the week (Monday or Tuesday) to avoid weekend delays at shipping hubs.
For reconstituted peptides, the logistics are more challenging. Use frozen gel packs or wet ice (double-bagged to prevent leakage) to maintain 2 to 8 degrees C. Insulated shipping containers rated for 48 to 72 hour cold maintenance are available from laboratory supply vendors. Ship overnight and include detailed storage instructions for the recipient. Consider whether it might be simpler and more reliable to ship the lyophilized peptide and have the collaborator reconstitute on-site.
Scenario 7: "I'm starting a long-term research protocol (6+ months). How do I plan my peptide supply?"
For protocols lasting several months, supply planning directly impacts peptide quality and research reliability. Here's a recommended approach:
- Calculate total supply needed. Determine your dose per session, sessions per week, and total protocol duration. Add 20% excess for wastage (dead volume, dropped vials, etc.). Use the FormBlends dosing calculator for precise calculations.
- Order all supply upfront. Receiving all peptide from the same manufacturing lot ensures batch-to-batch consistency throughout your protocol. This eliminates a potential variable that could confound your results.
- Store all lyophilized vials at -20 degrees C or colder. This preserves the full inventory in optimal condition for the entire protocol duration.
- Reconstitute in monthly batches. Every 3 to 4 weeks, reconstitute one vial (or the amount needed for the next 3 to 4 weeks). Aliquot and freeze any excess.
- Maintain a running inventory. Track how many vials remain, when each was reconstituted, and when each should be discarded. This prevents both waste and the use of expired material.
This approach ensures that every dose throughout a 6-month or 12-month protocol is as close to freshly reconstituted as possible, maximizing consistency and reliability of your research results.
Regulatory and Quality Considerations
While most readers of this guide are conducting research rather than manufacturing pharmaceuticals, understanding the regulatory framework around peptide storage helps contextualize best practices and evaluate the quality of peptide products you receive.
18.1 USP and ICH Guidelines
The United States Pharmacopeia (USP) and International Council for Harmonisation (ICH) establish standards for pharmaceutical storage and stability testing that inform best practices for research peptides.
USP General Chapter 659 (Packaging and Storage Requirements) defines storage temperature ranges used throughout pharmaceutical labeling:
- Freezer: -25 to -10 degrees C
- Cold: 2 to 8 degrees C (refrigerator)
- Cool: 8 to 15 degrees C
- Room temperature: 20 to 25 degrees C
- Controlled room temperature: 20 to 25 degrees C, with excursions permitted between 15 and 30 degrees C
- Warm: 30 to 40 degrees C
- Excessive heat: above 40 degrees C
These definitions are useful references when interpreting storage instructions from peptide suppliers.
ICH Q1A (Stability Testing of New Drug Substances and Products) defines the conditions for long-term, accelerated, and stressed stability testing. Key conditions include:
- Long-term storage: 25 degrees C / 60% RH (room temperature products) or 5 degrees C (refrigerated products)
- Accelerated storage: 40 degrees C / 75% RH (room temperature products) or 25 degrees C / 60% RH (refrigerated products)
- Testing frequency: at 0, 3, 6, 9, 12, 18, 24, and 36 months for long-term studies
When peptide suppliers provide stability data, it's often generated following these ICH guidelines. This means the stated shelf life reflects performance under defined, controlled conditions. Real-world storage, with its temperature fluctuations, variable humidity, and occasional mishandling, may result in somewhat shorter practical stability.
18.2 Certificate of Analysis (COA)
A Certificate of Analysis is a document provided by the peptide manufacturer that details the quality testing performed on a specific lot of peptide. A thorough COA for research peptides typically includes:
- Purity by HPLC: The percentage of the total peak area that corresponds to the target peptide. Research-grade peptides are typically 95% or higher; pharmaceutical-grade peptides are 98% or higher.
- Molecular weight by mass spectrometry: Confirmation that the measured molecular weight matches the theoretical mass for the target sequence. Mass accuracy within 1 Da of the expected value confirms sequence identity.
- Amino acid analysis: Quantitative measurement of the amino acid composition after hydrolysis, confirming that the correct amino acids are present in the expected ratios.
- Peptide content: The mass fraction of the lyophilized powder that is actually peptide (as opposed to counter-ions, residual solvents, moisture, and salts). Peptide content is typically 70 to 85% for TFA salt forms and 80 to 95% for acetate salt forms.
- Residual solvents: Levels of solvents used during synthesis (typically acetonitrile and TFA) that remain in the lyophilized product.
- Endotoxin testing: For peptides intended for injection studies, endotoxin levels must be below defined limits.
- Sterility testing: For sterile products, confirmation that no microbial contamination is present.
Request and retain COAs for every lot of peptide you receive. They serve as the baseline against which any subsequent degradation can be measured. If you suspect that a peptide has degraded, comparing a fresh analytical profile to the original COA is the most definitive way to confirm and quantify the degradation.
18.3 Good Documentation Practices for Peptide Storage
Even in a research setting where formal GMP (Good Manufacturing Practice) compliance isn't required, maintaining good documentation of peptide storage conditions supports research reproducibility and troubleshooting.
Recommended documentation practices:
- Maintain a peptide inventory log with lot numbers, receipt dates, storage locations, and expiration dates
- Record the date each vial is reconstituted and the solvent used
- Record weekly temperature checks of refrigerators and freezers
- Document any temperature excursions (power outages, equipment failures, transit events)
- Retain COAs for each lot received
- Note any visual observations (color changes, cloudiness, particulates) when using reconstituted peptides
- Record the date each reconstituted vial is discarded and the reason (expired, degraded, empty)
This documentation creates a traceable history that helps explain unexpected experimental results and supports quality improvement over time. If a peptide performs poorly in an experiment, the storage records can help determine whether degradation was a contributing factor.
Start Your Research Journey
Proper storage is just one piece of the peptide research puzzle. For personalized guidance on selecting the right peptides for your research goals, take the FormBlends free assessment. Our team can help you navigate the full spectrum of available compounds and develop a research protocol tailored to your specific objectives. Visit the Peptide Hub for our complete library of research resources.
Peptide Stability Myths and Misconceptions
The peptide research community is full of anecdotal advice, forum wisdom, and rules of thumb that don't always align with the science. Let's address the most common myths and replace them with evidence-based reality.
Myth 1: "Lyophilized peptides are indestructible."
Reality: Lyophilized peptides are dramatically more stable than reconstituted ones, but they are not immune to degradation. Heat, moisture, and light can all degrade lyophilized peptides, just much more slowly than they degrade solutions. A lyophilized peptide left in a hot car for a week, exposed to direct sunlight, or stored in a humid environment without desiccant can absolutely degrade. The lyophilized form buys you time. It doesn't make the peptide invincible.
Furthermore, the stability of a lyophilized peptide depends heavily on the quality of the lyophilization process. A poorly lyophilized product with high residual moisture, collapsed cake structure, or suboptimal excipient composition may be considerably less stable than its well-made counterpart. Not all lyophilized peptides are created equal.
Myth 2: "If a peptide looks fine, it is fine."
Reality: Visual inspection catches only the most advanced stages of degradation. Deamidation, methionine oxidation, racemization, and early-stage aggregation produce no visible changes. A peptide solution can look perfectly clear, colorless, and normal while having lost 30 to 50% or more of its biological activity. Visual inspection is a useful screening tool for gross problems, but it should never be the sole basis for judging peptide quality.
The only way to truly know whether a peptide has degraded is through analytical testing (HPLC, mass spectrometry) or functional testing (biological activity assays). These methods are more expensive and time-consuming than visual inspection, which is exactly why prevention-focused storage practices are so valuable. It's far cheaper to store peptides properly than to test every vial for degradation.
Myth 3: "Refrigerator storage is good enough for everything."
Reality: Refrigerator temperature (2 to 8 degrees C) is appropriate for reconstituted peptides in active use (with a 28-day shelf life), but it is suboptimal for long-term storage of lyophilized peptides. At refrigerator temperature, chemical degradation reactions still proceed at measurable rates. A lyophilized peptide stored at 4 degrees C for two years will have undergone more degradation than the same peptide stored at -20 degrees C for the same period.
The refrigerator also presents moisture risks for lyophilized peptides. Refrigerators cycle their compressors on and off, creating temperature fluctuations. When the compressor is off and the interior warms slightly, the relative humidity inside increases. When the compressor kicks back on and the temperature drops, condensation can form on cold surfaces, including peptide vials. This moisture cycling can contribute to moisture ingress over months of storage.
For any lyophilized peptide that won't be used within the next month, freezer storage at -20 degrees C is strongly preferred over refrigerator storage.
Myth 4: "Colder is always better."
Reality: For lyophilized peptides, this is essentially true. Colder temperatures always slow chemical degradation, and there are no freeze-thaw risks with dry powders. However, for reconstituted peptide solutions, "colder is always better" has an important exception: the freezing process itself can damage peptides.
As discussed in Section 6, freezing creates ice crystals that can mechanically stress peptide molecules, concentrate solutes to damaging levels, and shift pH by selective buffer crystallization. A properly aliquoted and frozen peptide solution is more stable than a refrigerated one, but an improperly frozen solution (without aliquoting, without cryoprotectant, with repeated freeze-thaw cycles) can actually be worse off than if it had simply been refrigerated and used within 28 days.
The takeaway: freezing is better than refrigeration for long-term storage, but only if done properly (aliquoted, with cryoprotectant, in single-use volumes, with no repeated freeze-thaw cycling).
Myth 5: "Bacteriostatic water keeps peptides stable indefinitely."
Reality: Bacteriostatic water's benzyl alcohol preservative inhibits bacterial growth. It does nothing to prevent chemical degradation. A peptide reconstituted in bacteriostatic water undergoes the same chemical degradation (oxidation, deamidation, hydrolysis, aggregation) as one reconstituted in plain sterile water. The preservative simply prevents the additional problem of microbial contamination, buying about 28 days of microbiological safety.
The 28-day shelf life for reconstituted peptides in bacteriostatic water is driven by two overlapping timelines: the chemical stability of the peptide (which varies by compound but generally allows weeks to months at refrigerator temperature) and the antimicrobial effectiveness of the preservative (which diminishes after about 4 weeks). The shorter of these two timelines determines the practical shelf life. For most peptides, the preservative timeline is the limiting factor at refrigerator temperature.
Myth 6: "You can refreeze a thawed peptide solution if you do it quickly."
Reality: Refreezing a thawed peptide solution subjects it to a second freeze-thaw cycle, with all the associated stresses: ice crystal formation, freeze-concentration, pH shifts, and surface-induced aggregation. Doing it quickly doesn't prevent these stresses. It might limit the time the peptide spends at elevated temperature between the thaw and the refreeze, which reduces some additional chemical degradation, but it doesn't eliminate the freeze-thaw damage itself.
Each freeze-thaw cycle is independently damaging. A peptide that has been frozen, thawed, refrozen, and thawed again has experienced twice the freeze-thaw stress of one that was frozen and thawed only once. The damage is cumulative and, for aggregation in particular, often irreversible. This is why single-use aliquoting is the standard recommendation, and why the mantra "never refreeze" applies regardless of how quickly the refreezing is done.
Myth 7: "Generic plastic storage containers are fine for peptides."
Reality: Not all plastics are suitable for peptide storage. Some plastics (particularly polystyrene and certain polyethylene formulations) can leach plasticizers, stabilizers, or monomers that interact with peptides. These extractables can catalyze oxidation, promote aggregation, or interfere with analytical measurements. Additionally, many common plastics are permeable to oxygen and moisture, which can slowly degrade the peptide even through the container wall.
Polypropylene (PP) is the preferred plastic for peptide and protein storage. It has low extractable content, good chemical resistance, and relatively low permeability to gases. Medical-grade or laboratory-grade polypropylene microcentrifuge tubes and cryovials are appropriate for peptide aliquots. Glass vials remain the gold standard for reconstituted peptide storage because glass is chemically inert and impermeable to gases.
If you must use a plastic container for a reconstituted peptide (for example, a polypropylene microcentrifuge tube for a frozen aliquot), ensure it is laboratory grade, certified free of DNase/RNase and human DNA (which indicates clean manufacturing), and rated for the storage temperature you'll be using.
Myth 8: "All peptides have the same storage requirements."
Reality: While the general principles of cold, dark, and dry storage apply broadly, specific peptides have unique vulnerabilities that warrant different levels of precaution. As detailed in Section 8, metal-coordinated peptides like GHK-Cu require specific pH management. Light-sensitive peptides like Selank need rigorous photoprotection. Lipidated peptides like semaglutide are prone to surface adsorption. Short peptides like Epithalon are exceptionally stable. Large, hydrophobic peptides like TB-500 are aggregation-prone.
A one-size-fits-all approach to storage will work for most peptides most of the time, but researchers working with multiple compounds should familiarize themselves with the specific vulnerabilities of each one. The few minutes spent understanding your peptide's weak points can prevent days of troubleshooting degradation-related experimental failures.
Myth 9: "Room temperature shipping means the peptide is degraded."
Reality: This concern is understandable but usually unfounded for lyophilized peptides. As discussed throughout this guide, lyophilized peptides can tolerate room temperature exposure for days to weeks without significant degradation. Standard domestic shipping (1 to 3 days) at ambient temperature is generally safe for lyophilized products, though cold chain shipping is always preferable.
The real shipping risk is not average room temperature exposure but extreme temperature exposure: a package sitting in a mail truck in summer heat (60+ degrees C) or a delivery left on a doorstep in direct sunlight. These extreme conditions can damage even lyophilized peptides. If you're ordering peptides during summer months or to a location with extreme temperatures, request cold chain shipping with insulated packaging and ice packs.
For reconstituted peptides, the risk calculation is very different. Reconstituted solutions should never be shipped at room temperature. Cold chain shipping with gel packs or wet ice is mandatory for any reconstituted peptide in transit.
Future Directions in Peptide Stabilization
Peptide stabilization technology continues to evolve, driven by the growing therapeutic peptide market and the need for more practical, real-world storage solutions. Here's a look at emerging approaches that may change how peptides are stored and handled in the coming years.
20.1 Room-Temperature Stable Formulations
One of the most active areas of pharmaceutical research is developing peptide formulations that are stable at room temperature for months or years, eliminating the need for cold chain storage and distribution. This would dramatically expand access to peptide therapeutics in regions with limited refrigeration infrastructure and simplify the logistics of peptide-based research globally.
Approaches being explored include advanced drying technologies (spray drying, supercritical fluid processing), microencapsulation in stabilizing polymers, incorporation into silk fibroin matrices, and sugar glass stabilization systems that maintain peptide integrity at temperatures up to 45 degrees C for extended periods. While these technologies are still largely in the research and early development stages, several have shown promising results in preclinical studies.
For researchers, the practical implication is that room-temperature stable peptide formulations may become commercially available for some compounds in the near future. However, until validated room-temperature formulations are specifically offered by your supplier, the standard cold storage recommendations in this guide remain the appropriate standard of care.
20.2 Improved Container Materials
Advances in container materials are addressing several long-standing challenges in peptide storage. Cyclic olefin copolymer (COC) and cyclic olefin polymer (COP) vials offer the transparency of glass with the break-resistance of plastic, plus lower extractable profiles than traditional glass. These materials also have lower surface energy, which reduces peptide adsorption on container walls, a particular advantage for hydrophobic peptides.
Surface-modified containers, with coatings designed to minimize peptide-surface interactions, are becoming more widely available. These include hydrophilic coatings that reduce hydrophobic peptide adsorption and positively charged coatings that repel positively charged peptides. As these technologies mature and become more affordable, they may replace traditional glass and plastic for peptide storage applications where surface adsorption is a significant concern.
20.3 Real-Time Stability Monitoring
Traditional stability assessment relies on periodic sampling and testing, which is resource-intensive and provides only snapshots of stability at discrete time points. Emerging technologies aim to provide continuous or on-demand stability monitoring without disturbing the stored peptide.
Smart packaging systems that incorporate colorimetric or fluorimetric indicators can detect pH changes, oxygen ingress, or specific degradation products within a sealed vial. A color change visible through the vial wall could alert the researcher to degradation without the need to open the container or perform analytical testing.
IoT-enabled temperature and humidity sensors that continuously monitor storage conditions and alert researchers to deviations via smartphone apps are already commercially available and becoming increasingly affordable. These systems provide documented temperature history for every storage location, replacing manual thermometer checks and providing much more detailed excursion data.
20.4 Engineered Peptide Stability
Beyond formulation and storage improvements, peptide engineering approaches can improve the intrinsic stability of peptide molecules themselves. These include:
- Substitution of labile residues: Replacing deamidation-prone Asn-Gly sequences with more stable alternatives (Asn-Ala, Asp-Gly, or non-natural amino acids) without affecting biological activity
- D-amino acid substitution: Replacing L-amino acids at non-critical positions with their D-enantiomers, which are resistant to enzymatic degradation and may also improve chemical stability
- Cyclization: Converting linear peptides to cyclic structures, which constrains conformational flexibility and reduces aggregation and proteolytic susceptibility
- PEGylation and lipidation: Attaching polyethylene glycol chains or lipid moieties to improve solubility, reduce aggregation, and extend circulation half-life. Semaglutide's C18 fatty diacid modification is an example of this approach
- Stapled peptides: Introducing chemical cross-links (staples) that lock the peptide into its bioactive conformation, improving both stability and potency
These engineering approaches don't replace proper storage practices, but they can expand the stability window for peptides that are particularly challenging to store or handle. Semaglutide's C18 fatty acid modification is a perfect example of how lipidation improves pharmacokinetics while creating new formulation challenges around surface adsorption and aggregation. Similarly, telomere-targeting peptides like Epithalon benefit from inherent stability due to their small size, while larger growth hormone-releasing peptides require more careful handling. As more engineered peptides enter the research pipeline, their specific storage requirements may differ from those of their unmodified counterparts, making it increasingly relevant to consult compound-specific storage guidance. Researchers exploring the full range of available compounds can visit the FormBlends Peptide Hub for our complete catalog of research resources covering everything from NAD+ cellular health applications to tirzepatide metabolic studies.