Executive Summary
Gonadotropin-releasing hormone (GnRH) analogs represent one of the most clinically significant classes of peptide therapeutics developed in the past half-century. From fertility preservation and cancer treatment to managing endometriosis and supporting gender-affirming care, these compounds have transformed how clinicians approach hormone-dependent conditions across every stage of life.
Key Takeaways
- GnRH agonists cause an initial hormonal flare (1-2 weeks) followed by sustained suppression through receptor downregulation
- GnRH antagonists provide immediate suppression without flare, making them preferred in certain clinical scenarios
- Gonadorelin (synthetic GnRH) is used at physiological doses to maintain fertility during testosterone replacement therapy (TRT)
- Triptorelin achieves castrate testosterone levels in over 95% of prostate cancer patients within 4 weeks
- Nafarelin delivered via nasal spray reduces endometriosis lesion scores by 40-50% over six months
The story of GnRH analogs begins with a ten-amino-acid peptide released in tiny, rhythmic pulses from the hypothalamus. This small molecule, known as gonadorelin in its synthetic form, orchestrates the entire reproductive cascade. By modifying even a single amino acid in its structure, researchers have created analogs with dramatically different pharmacological profiles - some that supercharge hormone production before shutting it down, others that block receptors immediately and completely.
Today's clinicians can choose from two broad categories of GnRH-based therapeutics. GnRH agonists like gonadorelin, triptorelin, leuprolide, nafarelin, and goserelin initially stimulate the pituitary before causing receptor downregulation and a profound suppression of sex hormones. GnRH antagonists like cetrorelix, ganirelix, and degarelix bypass the initial flare entirely, offering immediate suppression through direct receptor blockade. The newer oral antagonists - elagolix, relugolix, and linzagolix - have further expanded the therapeutic toolkit by allowing dose-dependent estrogen modulation without injections.
This guide provides a thorough examination of each major GnRH analog, covering their molecular pharmacology, clinical evidence base, approved indications, dosing protocols, safety profiles, and emerging applications. Whether you are a clinician selecting the right agent for a patient with advanced prostate cancer, a fertility specialist designing an IVF protocol, or a researcher exploring the latest developments in reproductive endocrinology, the information here draws from peer-reviewed literature and current clinical guidelines to support evidence-based decision-making.
KEY TAKEAWAYS
- GnRH agonists cause an initial hormonal flare (1-2 weeks) followed by sustained suppression through receptor downregulation
- GnRH antagonists provide immediate suppression without flare, making them preferred in certain clinical scenarios
- Gonadorelin (synthetic GnRH) is used at physiological doses to maintain fertility during testosterone replacement therapy (TRT)
- Triptorelin achieves castrate testosterone levels in over 95% of prostate cancer patients within 4 weeks
- Nafarelin delivered via nasal spray reduces endometriosis lesion scores by 40-50% over six months
- Oral GnRH antagonists (elagolix, relugolix) now offer non-injectable options with dose-dependent hormonal control
- All GnRH analogs are considered reversible; normal hormonal function typically returns within weeks to months after discontinuation
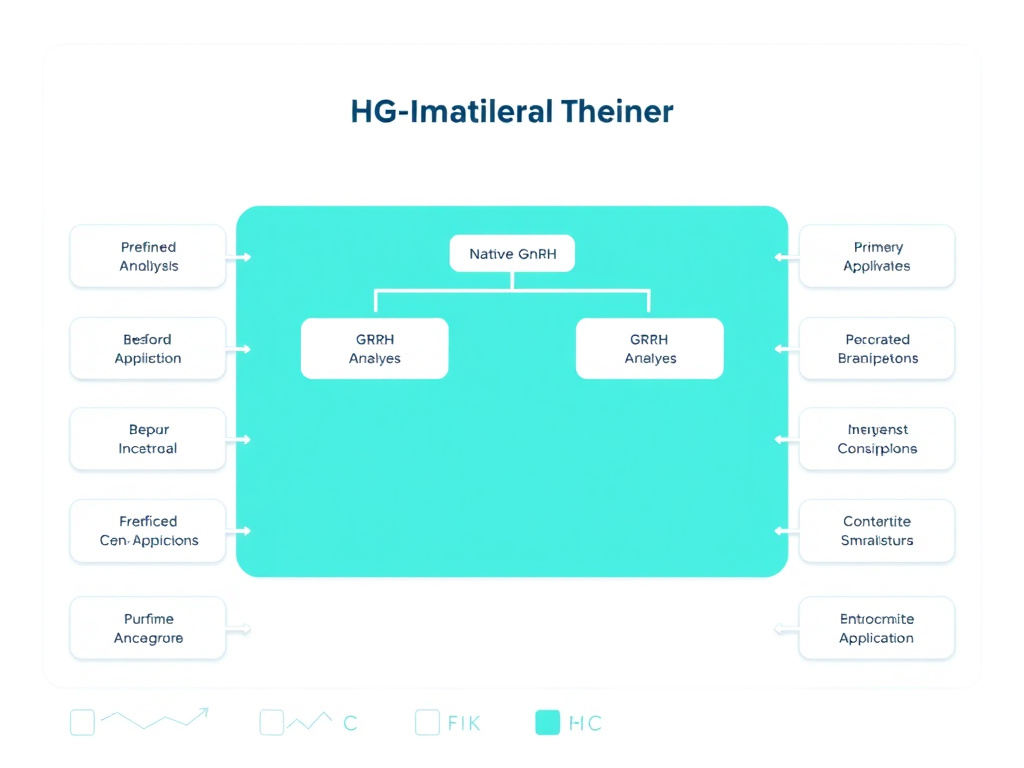
Figure 1: The GnRH analog family tree - agonists and antagonists derived from the native decapeptide, with their primary clinical applications mapped across reproductive health, oncology, and endocrinology.
For those new to peptide therapeutics, our Peptide Hub provides foundational context, while the Free Assessment tool can help determine which peptide protocols may align with your health goals. The Science page offers additional background on the evidence standards we apply across our research library.
GnRH System Biology: The Master Regulator of Reproduction
The Hypothalamic-Pituitary-Gonadal Axis
The hypothalamic-pituitary-gonadal (HPG) axis is the central command system for mammalian reproduction. Understanding how this axis works is essential for grasping why GnRH analogs are so therapeutically versatile. Every clinical application discussed in this report - from IVF protocols to prostate cancer treatment - ultimately traces back to the manipulation of this single neuroendocrine cascade.
At the top of the hierarchy sits the hypothalamus, a small region at the base of the brain that integrates signals from across the nervous system. Specialized neurons in the preoptic area and arcuate nucleus produce GnRH, a decapeptide with the amino acid sequence pyroGlu-His-Trp-Ser-Tyr-Gly-Leu-Arg-Pro-Gly-NH2. These neurons release GnRH in a pulsatile fashion into the hypophyseal portal blood system, which carries it directly to the anterior pituitary gland.
The pulsatile nature of GnRH release is not merely incidental; it is the defining feature of normal reproductive signaling. Pulse frequency and amplitude carry distinct information:
- Fast pulses (approximately every 60-90 minutes) preferentially stimulate luteinizing hormone (LH) secretion
- Slow pulses (approximately every 2-4 hours) preferentially stimulate follicle-stimulating hormone (FSH) secretion
- Continuous (non-pulsatile) exposure paradoxically shuts down gonadotropin production through receptor desensitization
This last point is the pharmacological basis for GnRH agonist therapy. When you deliver a long-acting GnRH agonist that provides constant receptor stimulation, you convert a pulsatile signal into a tonic one, and the pituitary responds by downregulating its GnRH receptors and ceasing LH/FSH production.
GnRH Receptor Signaling
The GnRH receptor (GnRH-R, also called GnRHR1) is a seven-transmembrane G protein-coupled receptor expressed primarily on pituitary gonadotrope cells. Several features make this receptor unusual among GPCRs:
- It lacks a typical intracellular C-terminal tail, which affects its internalization and desensitization kinetics
- It couples primarily to Gq/11 proteins, activating phospholipase C and the inositol phosphate/diacylglycerol signaling cascade
- Receptor density on gonadotrope membranes is itself regulated by GnRH exposure - a critical feature for understanding agonist-mediated downregulation
- A second receptor subtype (GnRH-R2) exists in some species but is a pseudogene in humans
When GnRH binds its receptor, the signaling cascade proceeds through several steps. Phospholipase C cleaves phosphatidylinositol bisphosphate (PIP2) into inositol trisphosphate (IP3) and diacylglycerol (DAG). IP3 triggers calcium release from intracellular stores, while DAG activates protein kinase C (PKC). Together, these second messengers drive the synthesis and secretion of LH and FSH from gonadotrope secretory granules.
Under pulsatile stimulation, this system resets between pulses, maintaining receptor sensitivity. Under continuous stimulation by GnRH agonists, however, several things happen:
- Receptor uncoupling: Sustained signaling leads to phosphorylation of intracellular domains, reducing G-protein coupling efficiency
- Receptor internalization: GnRH-R complexes are endocytosed, reducing surface receptor density
- Transcriptional downregulation: GnRH-R gene expression decreases, further reducing the number of available receptors
- Gonadotrope desensitization: The cell's capacity to synthesize and release gonadotropins diminishes progressively
This four-step desensitization process explains the characteristic biphasic response to GnRH agonist therapy: an initial "flare" of gonadotropin and sex steroid release (days 1-10), followed by profound suppression (weeks 2-4 onward).
The KNDy Neuron Network and Kisspeptin
One of the most significant advances in reproductive neuroendocrinology has been the discovery of the KNDy (Kisspeptin-Neurokinin B-Dynorphin) neuron network. These specialized neurons in the arcuate nucleus serve as the "pulse generator" for GnRH secretion and represent the primary upstream regulator of reproductive function.
Kisspeptin is the most potent known activator of GnRH neurons. Acting through the KISS1R (GPR54) receptor on GnRH neuron surfaces, kisspeptin drives GnRH release into the portal circulation. The relationship between kisspeptin and GnRH is so tight that loss-of-function mutations in either kisspeptin or its receptor cause hypogonadotropic hypogonadism - a complete failure of pubertal development and reproductive function.
Within KNDy neurons, three neuropeptides work in concert:
- Kisspeptin: Stimulates GnRH release (the "accelerator")
- Neurokinin B (NKB): Acts in an autocrine/paracrine manner to stimulate kisspeptin secretion from neighboring KNDy neurons, helping synchronize pulse generation
- Dynorphin: An endogenous opioid peptide that inhibits kisspeptin secretion (the "brake"), terminating each GnRH pulse
This model explains how sex steroids feed back to regulate GnRH pulsatility. Estrogen and testosterone receptors are expressed on KNDy neurons (not on GnRH neurons themselves), allowing circulating sex hormones to modulate pulse frequency and amplitude through effects on kisspeptin, NKB, and dynorphin expression. For deeper coverage of kisspeptin's role, see our dedicated Kisspeptin Research Report.
Downstream Effects: LH, FSH, and Gonadal Function
The gonadotropins released in response to GnRH signaling exert distinct effects on the gonads:
In Males
- LH acts on Leydig cells in the testes to stimulate testosterone production
- FSH acts on Sertoli cells to support spermatogenesis, sperm maturation, and inhibin B secretion
- Testosterone provides negative feedback to the hypothalamus and pituitary, modulating GnRH pulse frequency
- Inhibin B from Sertoli cells selectively suppresses FSH secretion
In Females
- FSH drives follicular recruitment and growth in the ovaries, stimulating estradiol production from granulosa cells
- LH stimulates theca cells to produce androgens (which granulosa cells convert to estrogens), triggers ovulation at mid-cycle through the LH surge, and supports the corpus luteum
- Estradiol exerts complex feedback: negative feedback at low levels (suppressing GnRH/gonadotropins) and positive feedback at high, sustained levels (triggering the pre-ovulatory LH surge)
- Progesterone from the corpus luteum provides negative feedback during the luteal phase
These physiological relationships form the basis for every therapeutic intervention discussed in this report. GnRH agonists exploit the continuous-exposure paradox to shut down this axis. GnRH antagonists block the receptor directly. And gonadorelin at low, pulsatile doses can actually support axis function - a principle used in fertility preservation during testosterone therapy.
CLINICAL RELEVANCE
The pulsatile nature of GnRH secretion means that the same molecule can have opposite effects depending on how it's delivered. Pulsatile gonadorelin restores fertility. Continuous GnRH agonist exposure suppresses it. This duality makes GnRH analogs uniquely versatile in clinical medicine, capable of both activating and silencing the reproductive axis depending on dose, formulation, and administration schedule.
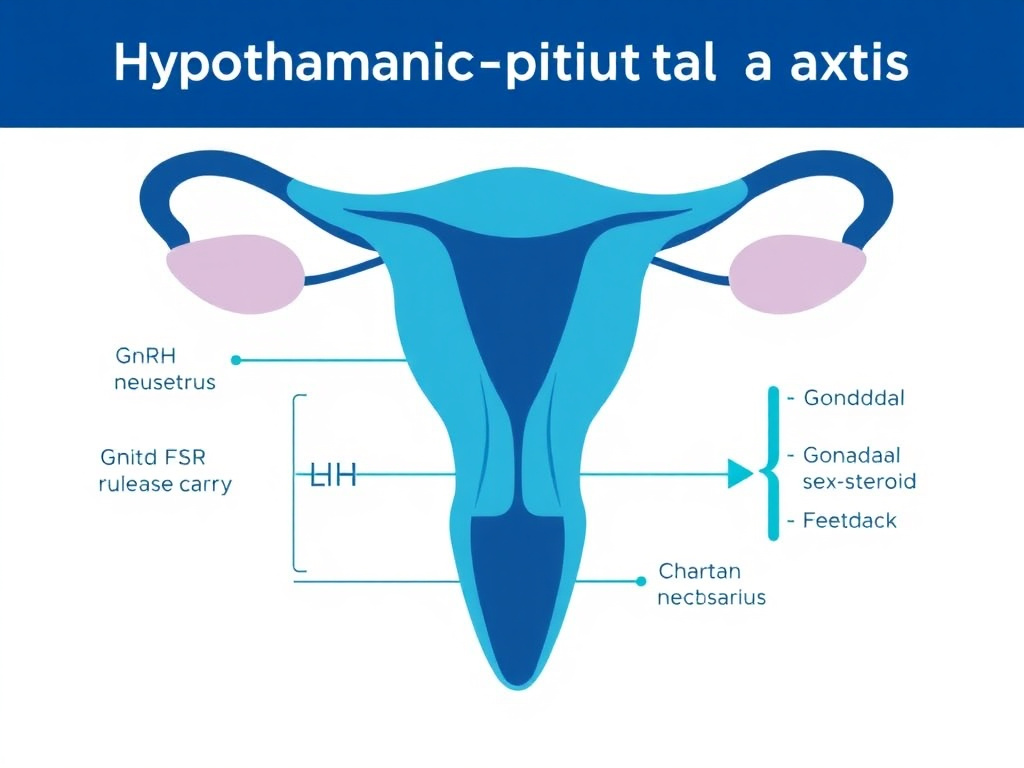
Figure 2: The hypothalamic-pituitary-gonadal (HPG) axis. Pulsatile GnRH from hypothalamic neurons drives LH and FSH release from the anterior pituitary, which in turn stimulates gonadal steroid production. Sex steroids feed back to modulate GnRH pulsatility through KNDy neurons.
GnRH Beyond Reproduction
While the HPG axis is the primary target of GnRH analog therapy, GnRH receptors have been identified in several extra-pituitary tissues, raising the possibility of direct peripheral effects:
- Prostate tissue: GnRH-R expression in prostate cancer cells suggests direct antiproliferative effects of GnRH analogs beyond simple androgen deprivation
- Breast tissue: Similar receptor expression in breast cancer cells has prompted investigation of GnRH analogs in breast cancer treatment
- Immune cells: T lymphocytes and other immune cells express GnRH receptors, suggesting immunomodulatory roles
- Placenta: GnRH and its receptor are expressed in placental tissue, where they may regulate trophoblast function
- Brain: GnRH receptors outside the pituitary may mediate cognitive and behavioral effects of GnRH signaling
These extra-pituitary actions are an active area of research and may partially explain why different GnRH analogs sometimes show clinical effects that cannot be fully attributed to sex steroid suppression alone.
Gonadorelin: The Native GnRH Peptide
Molecular Profile and Pharmacology
Gonadorelin is the synthetic form of endogenous gonadotropin-releasing hormone, identical in structure to the naturally occurring decapeptide. Its amino acid sequence - pyroGlu-His-Trp-Ser-Tyr-Gly-Leu-Arg-Pro-Gly-NH2 - has a molecular weight of 1,182.3 Da. Because it mirrors the body's own GnRH exactly, gonadorelin's pharmacological behavior depends entirely on how it's administered rather than on any structural modification.
Several commercial preparations of gonadorelin exist:
| Brand Name | Generic | Route | Primary Use |
|---|---|---|---|
| Factrel | Gonadorelin hydrochloride | IV/SC | Diagnostic testing |
| Lutrepulse | Gonadorelin acetate | IV (pulsatile pump) | Hypothalamic amenorrhea |
| Compounded | Gonadorelin acetate | SC injection | Fertility preservation during TRT |
Gonadorelin has a very short plasma half-life of approximately 2-4 minutes following intravenous administration and roughly 10-40 minutes after subcutaneous injection. This rapid clearance is both a limitation and an advantage. It means that each dose of gonadorelin mimics a natural GnRH pulse - stimulating a burst of LH and FSH release without causing the sustained receptor activation that leads to desensitization. This property makes it fundamentally different from long-acting GnRH agonists like triptorelin and leuprolide.
Diagnostic Applications
The GnRH stimulation test (also called the gonadorelin test) remains a standard diagnostic tool in endocrinology. By administering a single 100 mcg dose of gonadorelin intravenously and measuring the LH and FSH response at 15, 30, 45, 60, and 120 minutes, clinicians can assess the functional reserve of the anterior pituitary gonadotropes.
Diagnostic indications include:
- Distinguishing hypothalamic vs. pituitary causes of hypogonadism: In hypothalamic hypogonadotropic hypogonadism, the pituitary retains its capacity to respond to GnRH, so gonadorelin elicits a normal or exaggerated LH/FSH response. In pituitary disease, the response is blunted or absent.
- Evaluating precocious puberty: An exaggerated LH response to gonadorelin (particularly a peak LH greater than 5 IU/L or LH/FSH ratio greater than 1) confirms central (GnRH-dependent) precocious puberty, differentiating it from peripheral causes.
- Assessing pubertal status: The LH response pattern helps determine whether a child has entered puberty, which guides decisions about growth hormone therapy, puberty suppression, and other interventions.
- Monitoring GnRH agonist therapy: A suppressed response to gonadorelin confirms adequate pituitary downregulation during GnRH agonist treatment.
Therapeutic Use: Pulsatile Gonadorelin for Hypothalamic Amenorrhea
Perhaps the most elegant application of gonadorelin is pulsatile intravenous administration for women with hypothalamic amenorrhea. This condition, often caused by low body weight, excessive exercise, or psychological stress, results from inadequate endogenous GnRH pulsatility. By delivering gonadorelin through a programmable pump at doses of 5-20 mcg every 60-120 minutes, clinicians can restore normal pituitary-gonadal function and achieve ovulation rates of 80-90% per treatment cycle.
Pulsatile gonadorelin therapy offers several advantages over gonadotropin injections for this patient population:
- Lower risk of ovarian hyperstimulation syndrome (OHSS) because the pituitary self-regulates gonadotropin release through normal feedback mechanisms
- Lower multiple pregnancy rates (approximately 5-10% vs. 15-30% with gonadotropin therapy)
- More physiological follicular development, typically resulting in monofollicular ovulation
- No need for intensive ultrasound monitoring during treatment cycles
Despite these advantages, pulsatile gonadorelin therapy has become less widely available due to supply issues and the logistical challenges of wearing a portable infusion pump. Many fertility centers have shifted to gonadotropin protocols as a result, though some specialists argue that pulsatile GnRH remains the gold standard for hypothalamic amenorrhea when available.
Gonadorelin in Testosterone Replacement Therapy
One of the fastest-growing applications for gonadorelin is co-administration with testosterone replacement therapy (TRT) to preserve testicular function and fertility. This use case has become especially relevant since the FDA's 2020 classification of human chorionic gonadotropin (hCG) as a biologic, which removed it from compounding pharmacies and limited its availability.
The rationale is straightforward. Exogenous testosterone suppresses the HPG axis through negative feedback, reducing endogenous LH and FSH production. Without LH stimulation, Leydig cells decrease testosterone production and testicular volume shrinks. Without FSH, Sertoli cell function declines and spermatogenesis can cease entirely. For men who want the benefits of TRT while maintaining fertility potential, this presents a real problem.
Gonadorelin addresses this by stimulating the pituitary to release LH and FSH, partially overriding the suppressive effect of exogenous testosterone. When administered subcutaneously at typical doses of 100-200 mcg two to three times weekly, gonadorelin can:
- Help maintain testicular volume and prevent the testicular atrophy commonly seen with TRT
- Support ongoing intratesticular testosterone production
- Maintain some degree of spermatogenesis through FSH stimulation
- Preserve the HPG axis responsiveness, potentially making TRT discontinuation easier
CLINICAL REALITY CHECK
While gonadorelin is widely prescribed for TRT adjunctive use, the evidence base for this specific application remains limited compared to hCG. Most published data on fertility preservation during TRT involves hCG rather than gonadorelin. The short half-life of gonadorelin means that intermittent subcutaneous dosing may not replicate the sustained gonadotropin stimulation achieved by hCG (which has a half-life of 24-36 hours). Some clinicians report that gonadorelin is less effective than hCG for maintaining sperm parameters during TRT. Patients should discuss these nuances with their prescribing physician and consider baseline semen analysis and periodic monitoring. Use the Dosing Calculator to review standard protocols.
Gonadorelin Dosing Protocols
| Indication | Dose | Route | Frequency | Duration |
|---|---|---|---|---|
| Diagnostic GnRH test | 100 mcg | IV | Single dose | One-time |
| Hypothalamic amenorrhea | 5-20 mcg/pulse | IV pump | Every 60-120 min | Until ovulation |
| TRT adjunct (fertility) | 100-200 mcg | SC | 2-3x weekly | Ongoing with TRT |
| TRT adjunct (testicular) | 100 mcg | SC | 2x weekly | Ongoing with TRT |
| Post-cycle therapy | 100-200 mcg | SC | Daily x 2 weeks, then 3x/week | 4-8 weeks |
Safety Profile
Gonadorelin has an excellent safety profile at therapeutic doses, consistent with what you'd expect from a peptide identical to the body's own hormone. Commonly reported side effects include:
- Injection site reactions: redness, swelling, mild pain (the most frequent complaint)
- Headache (reported in approximately 5-10% of users)
- Nausea (uncommon)
- Flushing (uncommon)
- Abdominal discomfort (rare)
- Hypersensitivity reactions (very rare, but anaphylaxis has been reported)
Because gonadorelin is cleared rapidly and doesn't cause sustained receptor stimulation at standard therapeutic doses, it does not produce the hormonal flare associated with long-acting GnRH agonists. It also does not cause the bone density loss or menopausal symptoms seen with chronic GnRH agonist use, since it's not intended to suppress gonadal function.
Contraindications include known hypersensitivity to GnRH or its analogs, hormone-dependent tumors where gonadotropin stimulation could be harmful, and pregnancy (Category X).
Triptorelin: The Prostate Cancer Workhorse
Molecular Profile
Triptorelin (D-Trp6-GnRH) is a synthetic GnRH agonist created by substituting D-tryptophan for the native glycine at position 6 of the GnRH decapeptide. This single amino acid change has profound pharmacological consequences. The D-amino acid substitution confers resistance to enzymatic degradation by endopeptidases, extending the peptide's half-life from minutes (for native GnRH) to approximately 3-4 hours. More significantly, triptorelin binds to GnRH receptors with roughly 100-fold greater affinity than native GnRH.
The combination of increased receptor affinity and metabolic stability means that triptorelin provides sustained, non-pulsatile GnRH receptor activation. This continuous stimulation triggers the desensitization cascade described earlier - initial gonadotropin flare, followed by receptor downregulation and profound hormonal suppression.
Triptorelin is marketed under several brand names worldwide, including Decapeptyl, Trelstar, and Diphereline, and is available in multiple depot formulations.
Formulations and Pharmacokinetics
| Formulation | Dose | Route | Dosing Interval | Brand Names |
|---|---|---|---|---|
| Immediate release | 0.1 mg | SC | Daily | Decapeptyl (daily) |
| 1-month depot | 3.75 mg | IM | Every 28 days | Trelstar, Decapeptyl |
| 3-month depot | 11.25 mg | IM | Every 84 days | Trelstar LA |
| 6-month depot | 22.5 mg | IM | Every 168 days | Trelstar |
The depot formulations use biodegradable microsphere technology (poly-lactic-co-glycolic acid, or PLGA) to provide sustained release over one, three, or six months. After intramuscular injection, the microspheres slowly degrade, releasing triptorelin at a controlled rate. Peak plasma concentrations occur within hours of injection (representing the immediate release from the microsphere surface), followed by a sustained low-level release as the polymer matrix erodes.
Hormonal Response: The Flare and Suppression Phases
The hormonal response to triptorelin follows a characteristic and well-documented pattern:
Phase 1 - The Flare (Days 1-10): Triptorelin's initial binding to GnRH receptors causes a surge of LH and FSH release. In men, this drives testosterone levels up significantly, often rising from a baseline of around 300-500 ng/dL to 600-900 ng/dL. This flare can temporarily worsen symptoms in hormone-sensitive conditions, particularly prostate cancer, where it may cause "tumor flare" with increased bone pain, urinary obstruction, or spinal cord compression.
Phase 2 - Transition (Days 10-21): As GnRH receptors begin to downregulate, gonadotropin levels start declining. Testosterone levels fall progressively through this period.
Phase 3 - Castration (Day 21 onward): By 2-4 weeks after the first injection, testosterone levels typically reach castrate levels (defined as less than 50 ng/dL by traditional criteria, or less than 20 ng/dL by the more stringent modern threshold). LH and FSH are profoundly suppressed.
Testosterone Response During Triptorelin Treatment
Data from Phase III clinical trials of triptorelin pamoate in advanced prostate cancer. Baseline testosterone is suppressed to castrate levels by Week 4, with the initial flare visible at Week 1.
Triptorelin in Prostate Cancer
Androgen deprivation therapy (ADT) remains the cornerstone of treatment for advanced and metastatic prostate cancer, and triptorelin is one of the most widely used agents for this purpose. The rationale is simple: prostate cancer cells are typically androgen-dependent, requiring testosterone for growth and survival. By eliminating testosterone through medical castration, ADT causes tumor regression and symptomatic improvement in the majority of patients.
Clinical Trial Evidence
Multiple Phase III trials have established triptorelin's efficacy in prostate cancer:
Triptorelin vs. Leuprolide Equivalence Trial: A landmark non-inferiority study compared triptorelin pamoate (3.75 mg monthly) with leuprolide acetate (7.5 mg monthly) in 284 patients with advanced prostate cancer. Both agents demonstrated clinical equivalence in maintaining castrate testosterone levels (50 ng/dL or less), with 98.8% of triptorelin patients and 97.3% of leuprolide patients achieving this threshold between months 2 and 9. The study confirmed triptorelin as a safe and effective alternative to the more established leuprolide (Crawford et al., 2011).
Testosterone Nadir and Survival: A pooled post-hoc analysis of three Phase III triptorelin studies, published in BJUI Compass in 2024, examined whether deeper testosterone suppression improves outcomes. The analysis found that patients achieving a very low testosterone nadir (less than 0.35 nmol/L, approximately 10 ng/dL) during triptorelin monotherapy had significantly improved overall survival and disease-specific survival compared to those with nadirs between 0.35 and 0.7 nmol/L. This finding supports the trend toward targeting testosterone levels well below the traditional 50 ng/dL castration threshold (Klotz et al., 2024).
Extended Depot Formulations: Studies evaluating the 6-month triptorelin depot (22.5 mg) have shown that 91-97% of patients maintain testosterone levels below 50 ng/dL throughout the treatment period. The 6-month formulation offers improved convenience without sacrificing efficacy, reducing the number of clinic visits from 12 to 2 per year (Lundstrom et al., 2009).
Managing the Testosterone Flare
The initial testosterone flare with triptorelin can cause clinical complications in men with advanced prostate cancer, particularly those with:
- High-volume bone metastases (risk of pathological fractures and bone pain exacerbation)
- Vertebral metastases near the spinal cord (risk of spinal cord compression)
- Obstructive uropathy (risk of acute urinary retention)
To mitigate flare-related complications, clinicians typically co-administer an anti-androgen (such as bicalutamide 50 mg daily or flutamide 250 mg three times daily) beginning 1-2 weeks before the first triptorelin injection and continuing for 2-4 weeks afterward. The anti-androgen blocks the testosterone surge's effects at the receptor level without interfering with the GnRH agonist's mechanism.
Alternatively, clinicians can avoid the flare entirely by using a GnRH antagonist like degarelix for the initial month of treatment before transitioning to triptorelin for maintenance therapy. This "switch" strategy has gained popularity as a way to combine the immediate suppression of an antagonist with the convenience and cost-effectiveness of depot agonist formulations.
Triptorelin in Fertility Preservation
An important emerging application for triptorelin is ovarian function preservation during chemotherapy in premenopausal women with cancer. GnRH agonists like triptorelin are administered before and during chemotherapy to suppress ovarian activity, theoretically protecting quiescent follicles from cytotoxic damage.
The POEMS (Prevention of Early Menopause Study) and PROMISE-GIM6 trials provided key evidence for this approach. The PROMISE-GIM6 trial, a randomized Phase III study in premenopausal women with early breast cancer receiving chemotherapy, found that concurrent triptorelin administration significantly reduced the incidence of premature ovarian failure (8.9% vs. 25.9%) and increased rates of spontaneous pregnancy after treatment (Lambertini et al., 2018).
Current ASCO (American Society of Clinical Oncology) guidelines recommend discussing GnRH agonist co-treatment with all premenopausal women undergoing gonadotoxic chemotherapy, while noting that it should not be used as a substitute for established fertility preservation methods like oocyte or embryo cryopreservation. For more on fertility-related peptide therapies, see our Gonadorelin Research Report.
Triptorelin Safety Profile
Triptorelin's adverse effects are primarily consequences of the sex steroid deprivation it produces, rather than direct drug toxicity:
| Adverse Effect | Frequency | Mechanism |
|---|---|---|
| Hot flashes | 50-80% | Estrogen/testosterone deprivation |
| Sexual dysfunction | 30-60% | Testosterone suppression |
| Fatigue | 20-40% | Hypogonadism |
| Injection site reactions | 10-20% | Local inflammatory response |
| Bone mineral density loss | Progressive with duration | Estrogen/testosterone deprivation |
| Metabolic syndrome features | 15-30% | Hypogonadism-related metabolic changes |
| Mood changes/depression | 10-20% | Hormonal fluctuation |
| Gynecomastia | 5-10% | Altered androgen/estrogen ratio |
| Cardiovascular events | Small increased risk | Metabolic effects of ADT |
Long-term ADT with triptorelin raises particular concerns about bone health and cardiovascular risk. Prospective monitoring should include periodic DEXA scans (every 1-2 years), metabolic panels (glucose, lipids, HbA1c), and cardiovascular risk assessment. Bone-protective agents such as bisphosphonates or denosumab are commonly co-prescribed for men receiving prolonged ADT.
Nafarelin: The Intranasal GnRH Agonist
Molecular Profile
Nafarelin (D-Nal(2)6-GnRH) is a synthetic GnRH agonist distinguished by the substitution of 3-(2-naphthyl)-D-alanine at position 6 of the native GnRH decapeptide. This modification produces a compound approximately 200 times more potent than natural GnRH, with enhanced resistance to enzymatic degradation and a plasma half-life of roughly 3 hours. Nafarelin's defining clinical feature is its formulation as a nasal spray (brand name Synarel), making it the only GnRH agonist routinely administered intranasally.
The intranasal route offers approximately 2-3% bioavailability compared to subcutaneous injection. While this may seem inefficient, nafarelin's high potency compensates, and the convenience of nasal administration makes it especially attractive for conditions requiring daily dosing over extended periods, such as endometriosis and central precocious puberty.
Nafarelin in Endometriosis
Endometriosis affects an estimated 6-10% of reproductive-age women and causes chronic pelvic pain, dysmenorrhea, dyspareunia, and infertility. Because endometriotic implants are estrogen-dependent, creating a hypoestrogenic state through GnRH agonist therapy causes implant regression and symptom relief.
Landmark Clinical Trials
Nafarelin vs. Danazol (NEJM, 1988): The landmark multicenter double-blind trial published in the New England Journal of Medicine compared nafarelin nasal spray (400 and 800 mcg daily) with oral danazol (600 and 800 mg daily) in 236 women with laparoscopically confirmed endometriosis. Key findings included:
- Both nafarelin doses were as effective as danazol in reducing disease severity assessed by repeat laparoscopy after 6 months of treatment
- Mean revised American Fertility Society (rAFS) laparoscopic scores decreased from 21.9 to 12.6 with nafarelin 800 mcg/day (a 42% reduction) and from 20.4 to 11.7 with nafarelin 400 mcg/day (a 43% reduction)
- Severe dysmenorrhea prevalence dropped from approximately 40% to 5-10% during treatment
- The proportion of women reporting no or minimal pain increased from 25% to approximately 70%
- Nafarelin had a more favorable side effect profile than danazol, with fewer androgenic effects (acne, weight gain, deepened voice) but more hypoestrogenic symptoms (hot flashes, vaginal dryness)
Fertility After Treatment: Among 149 patients who attempted conception after completing nafarelin therapy, 58 (39%) achieved pregnancy. This rate compared favorably with historical controls receiving danazol or no treatment (Henzl et al., 1988).
Three vs. Six Months of Treatment: A randomized double-blind trial by Henzl and colleagues examined whether 3 months of nafarelin therapy could provide comparable symptom relief to the standard 6-month course. Both durations produced significant pain reduction. However, symptoms recurred in both groups during follow-up, with pain scores gradually increasing over 12 months but remaining below pre-treatment baseline values. This finding suggested that shorter courses might be sufficient for symptom management while reducing cumulative hypoestrogenic exposure (Henzl et al., 1995).
Add-Back Therapy: A double-blind randomized study evaluated nafarelin alone versus nafarelin combined with norethisterone (an add-back progestogen). The combination maintained efficacy for endometriosis symptoms while significantly reducing bone mineral density loss and vasomotor symptoms. This approach extended the safe treatment window beyond 6 months and anticipated the add-back strategies now standard with oral GnRH antagonists (Hornstein et al., 1997).
Nafarelin in Central Precocious Puberty
Central precocious puberty (CPP) occurs when the HPG axis activates prematurely, typically before age 8 in girls and age 9 in boys. Nafarelin nasal spray was one of the first GnRH agonists approved for this indication and remains in clinical use, particularly in settings where injectable depot formulations are difficult for young patients to tolerate.
Standard dosing for CPP is 1600 mcg daily (800 mcg in each nostril, twice daily). At this dose, nafarelin reliably suppresses pubertal progression, as evidenced by:
- Reduction of LH and FSH to prepubertal levels
- Regression or stabilization of secondary sexual characteristics
- Deceleration of skeletal maturation (bone age advancement slows to match or fall below chronological age)
- Improved predicted adult height by allowing more time for growth before epiphyseal fusion
Treatment continues until the normal age of puberty, at which point nafarelin is discontinued and spontaneous puberty resumes. Studies have shown that reproductive function, including menstruation and fertility, recovers normally after treatment cessation.
Nafarelin in IVF Protocols
Nafarelin has been used in IVF protocols as part of the "long protocol" for pituitary downregulation. Starting nafarelin in the mid-luteal phase of the cycle preceding stimulation allows pituitary desensitization by the time gonadotropin stimulation begins. This prevents premature LH surges that could trigger spontaneous ovulation and cancel the IVF cycle.
However, nafarelin's role in IVF has diminished with the widespread adoption of GnRH antagonist protocols (using cetrorelix or ganirelix), which achieve similar LH suppression with shorter treatment duration, fewer injections, and lower OHSS risk. Nafarelin retains a niche in patients who prefer intranasal administration and in specific protocol variations where gradual pituitary suppression is desired.
Nafarelin Dosing Summary
| Indication | Daily Dose | Administration | Duration |
|---|---|---|---|
| Endometriosis | 400 mcg (200 mcg per nostril, once daily) | Intranasal | 6 months |
| Endometriosis (higher dose) | 800 mcg (400 mcg twice daily) | Intranasal | 6 months |
| Central precocious puberty | 1600 mcg (800 mcg twice daily) | Intranasal | Until normal pubertal age |
| IVF downregulation | 400-600 mcg | Intranasal | 2-4 weeks pre-stimulation |
Safety and Tolerability
Nafarelin's side effect profile reflects its mechanism of action - estrogen deprivation symptoms dominate. In the major endometriosis trials, the most common adverse effects were:
- Hot flashes: 90% of patients (the most frequently reported complaint)
- Nasal irritation: 10-15% (specific to the intranasal route)
- Decreased libido: 10-22%
- Vaginal dryness: 15-19%
- Headache: 15-19%
- Emotional lability: 10-15%
- Acne: 10-13% (related to the initial androgen flare)
- Bone mineral density loss: Average 2-6% decrease over 6 months at the lumbar spine (largely reversible after discontinuation)
The nasal route introduces unique considerations. Concurrent use of nasal decongestants can alter nafarelin absorption and should be avoided or timed at least 2 hours apart from nafarelin administration. Upper respiratory infections may also affect bioavailability, though clinically significant treatment failures from this cause are rare.
CLINICAL PEARL
Nafarelin's intranasal delivery makes it the most patient-friendly GnRH agonist for adolescents with central precocious puberty who are needle-averse. However, compliance can be challenging with the twice-daily dosing schedule, particularly in younger children. Depot injectable formulations (such as leuprolide or triptorelin) offer the advantage of monthly or quarterly administration under medical supervision, ensuring adherence. The choice between nasal and injectable GnRH agonists for CPP should consider the child's age, needle tolerance, family reliability with daily dosing, and clinical response.
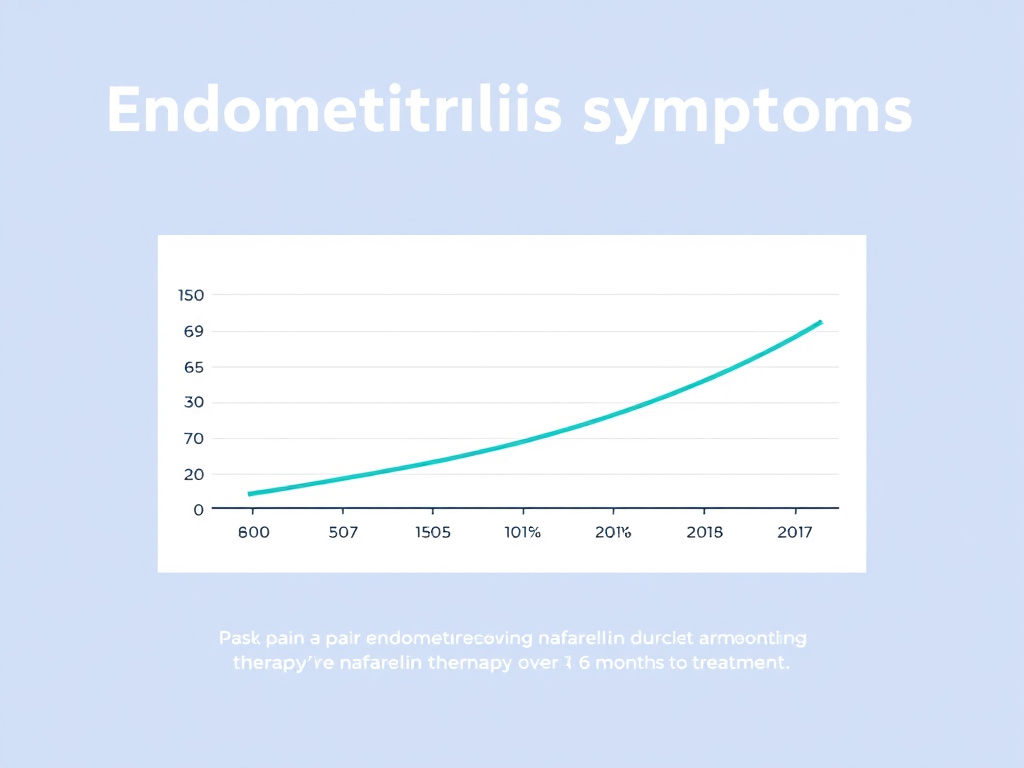
Figure 3: Symptom reduction during 6-month nafarelin therapy for endometriosis. Severe dysmenorrhea prevalence drops from approximately 40% to under 10%, with corresponding improvements in pelvic pain and dyspareunia scores.
Other GnRH Agonists: Leuprolide, Goserelin, Histrelin, and Buserelin
Leuprolide (Lupron)
Leuprolide acetate is the most widely prescribed GnRH agonist worldwide and the reference standard against which newer agents are often compared. Its amino acid substitution (D-Leu6) provides approximately 15-fold greater potency than native GnRH with a half-life of roughly 3 hours after subcutaneous injection.
Leuprolide is available in an extensive range of formulations:
| Product | Formulation | Dose | Interval |
|---|---|---|---|
| Lupron Depot | IM microsphere | 3.75, 7.5, 11.25, 22.5, 30, 45 mg | 1, 1, 3, 3, 4, 6 months |
| Eligard | SC depot | 7.5, 22.5, 30, 45 mg | 1, 3, 4, 6 months |
| Lupron Depot-Ped | IM microsphere | 7.5, 11.25, 15 mg | 1, 3, 1 months |
| Fensolvi | SC depot | 45 mg | 6 months |
FDA-approved indications for leuprolide include advanced prostate cancer, endometriosis, uterine fibroids (preoperative), central precocious puberty, and (in combination with norethindrone acetate) management of endometriosis with add-back therapy. Its clinical efficacy profile is largely interchangeable with triptorelin, with choice between the two often driven by formulary availability, cost, and prescriber familiarity.
Goserelin (Zoladex)
Goserelin is a GnRH agonist with a unique delivery system - a small, solid biodegradable implant inserted subcutaneously in the anterior abdominal wall using a pre-loaded syringe with a 14- or 16-gauge needle. While the needle size can be intimidating, the implant provides sustained drug release for either 28 days (3.6 mg) or 12 weeks (10.8 mg).
Comparative studies suggest goserelin may have a slight edge over leuprolide in maintaining testosterone suppression. A systematic review of GnRH agonists in prostate cancer found that goserelin showed better efficacy in maintaining testosterone levels at or below 50 ng/dL and superior PSA suppression compared to leuprolide, though the clinical significance of these differences remains debated.
Goserelin has broad regulatory approval for prostate cancer, breast cancer (in premenopausal women), endometriosis, endometrial thinning prior to ablation, and IVF pituitary suppression. Its subcutaneous implant route is sometimes considered the most convenient administration method among injectable GnRH agonists, as it doesn't require reconstitution.
Histrelin (Vantas, Supprelin LA)
Histrelin stands apart from other GnRH agonists through its unique subcutaneous implant technology. The Supprelin LA implant is a small, flexible rod inserted in the inner upper arm that provides continuous histrelin release for 12 months. This year-long duration is the longest of any GnRH agonist formulation and offers maximum convenience for chronic conditions.
Supprelin LA is FDA-approved specifically for central precocious puberty, where its annual dosing interval is particularly advantageous for pediatric patients and families. The Vantas implant (50 mg histrelin, 12-month duration) is approved for advanced prostate cancer. Both implants require a minor surgical procedure for insertion and removal.
Buserelin (Suprefact)
Buserelin is available in both intranasal (like nafarelin) and subcutaneous injection formulations. It's widely used in Europe and Canada but is not marketed in the United States. Its clinical applications overlap with the other GnRH agonists: prostate cancer, endometriosis, uterine fibroids, and IVF protocols.
Buserelin's intranasal formulation requires three times daily administration (compared to nafarelin's twice daily), which has limited its adoption relative to nafarelin in the intranasal GnRH agonist space.
Comparative Overview: GnRH Agonists
| Agent | Relative Potency (vs GnRH) | Routes Available | Longest Depot Duration | Key Distinguishing Feature |
|---|---|---|---|---|
| Gonadorelin | 1x (native) | IV, SC | N/A (short-acting) | Diagnostic use; TRT adjunct |
| Leuprolide | 15-50x | IM, SC | 6 months | Most widely prescribed; broadest formulation range |
| Triptorelin | 100x | IM | 6 months | Strong evidence for deep testosterone suppression |
| Goserelin | 100x | SC implant | 3 months | Pre-loaded implant; no reconstitution |
| Nafarelin | 200x | Intranasal | N/A (daily dosing) | Only nasal GnRH agonist in wide use |
| Histrelin | 100x | SC implant (rod) | 12 months | Longest-acting formulation; annual implant |
| Buserelin | 20-40x | Intranasal, SC | 3 months | Available in Europe/Canada; not in US |
GnRH Antagonists: Immediate Suppression Without the Flare
Mechanism: How Antagonists Differ from Agonists
GnRH antagonists represent a fundamentally different pharmacological approach to HPG axis suppression. Rather than stimulating GnRH receptors to the point of desensitization (the agonist strategy), antagonists competitively block the receptor, preventing native GnRH from binding and immediately halting LH and FSH secretion.
This direct blockade produces several clinically significant differences from agonist therapy:
- No hormonal flare: Because antagonists never activate the receptor, there is no initial surge of gonadotropins or sex steroids. Testosterone and estrogen levels begin declining within hours of the first dose.
- Rapid onset: Gonadotropin suppression is measurable within 4-8 hours and reaches clinically significant levels within 24-72 hours. Compare this with GnRH agonists, which require 2-4 weeks to achieve full suppression.
- Dose-dependent suppression: The degree of hormonal suppression is proportional to the antagonist dose. Lower doses partially suppress the axis, while higher doses achieve complete castration. This allows for nuanced hormonal control - a feature exploited by the newer oral antagonists.
- Rapid reversibility: Because antagonists don't cause receptor downregulation, hormonal recovery after discontinuation is faster than with agonists. This is particularly advantageous in IVF protocols.
Injectable GnRH Antagonists
Cetrorelix (Cetrotide)
Cetrorelix is a synthetic decapeptide GnRH antagonist approved for preventing premature LH surges during controlled ovarian stimulation (COS) for IVF. It's administered subcutaneously in either a single-dose protocol (3 mg on stimulation day 7) or a multiple-dose protocol (0.25 mg daily starting on stimulation day 5-6).
The multiple-dose protocol is more commonly used because it allows flexible timing of hCG trigger administration. Cetrorelix at 0.25 mg daily reliably suppresses the LH surge within 4-8 hours of the first injection, maintaining suppression as long as daily dosing continues. Ovulation trigger with hCG or GnRH agonist can be administered when follicles reach appropriate size, and cetrorelix is simply discontinued.
Key pharmacokinetic data for cetrorelix:
- Bioavailability: approximately 85% after subcutaneous injection
- Half-life: approximately 30 hours (single 3 mg dose) or 12 hours (0.25 mg daily dose)
- Time to peak suppression: 4-8 hours
- Duration of suppression after single 3 mg dose: approximately 4 days
Ganirelix (Orgalutran/Antagon)
Ganirelix is functionally interchangeable with cetrorelix for IVF protocols, with comparable efficacy in preventing premature LH surges and similar pregnancy outcomes. It's available as 0.25 mg pre-filled syringes for daily subcutaneous injection, typically starting on stimulation day 5-6.
Both cetrorelix and ganirelix have been extensively compared in clinical trials. A Cochrane systematic review encompassing over 7,000 women found no significant differences between the two agents in pregnancy rates, number of oocytes retrieved, or OHSS incidence. The choice between them is typically driven by availability, cost, and prescriber preference rather than clinical superiority (Al-Inany et al., 2016).
One difference worth noting relates to histamine release. Early GnRH antagonists were associated with histamine-mediated side effects (skin reactions, bronchospasm) due to structural similarities with mast cell-activating peptides. Among the marketed antagonists, degarelix shows the lowest propensity for histamine release, followed by ganirelix, then cetrorelix. In clinical practice, however, significant histamine reactions are rare with all current agents.
Degarelix (Firmagon)
Degarelix occupies a unique position among GnRH antagonists: it's the only injectable antagonist approved specifically for prostate cancer. This indication sets it apart from cetrorelix and ganirelix, which are used exclusively in reproductive medicine.
Degarelix is administered as a starting dose of 240 mg (two 120 mg subcutaneous injections) followed by monthly maintenance doses of 80 mg. Key clinical advantages include:
- No testosterone flare: Testosterone reaches castrate levels within 3 days in 96% of patients, eliminating the need for anti-androgen "flare protection" required with GnRH agonists
- Rapid PSA decline: PSA levels decrease faster with degarelix than with leuprolide during the first 28 days of treatment
- Potential cardiovascular benefit: Post-hoc analyses of the CS21 trial suggested a lower rate of cardiovascular events with degarelix compared to leuprolide, though this finding requires confirmation in prospective studies
- No microsurges: Unlike depot GnRH agonists, which can produce minor testosterone microsurges during re-injection periods, degarelix maintains stable suppression
The primary disadvantage of degarelix is its monthly injection requirement and injection site reactions. Approximately 40% of patients experience injection site pain, erythema, or swelling, compared to less than 5% with GnRH agonist depot formulations. This has limited degarelix's adoption as a first-line ADT agent in many practices, despite its pharmacological advantages.

Figure 4: Hormonal response comparison between GnRH agonists and antagonists. Agonists produce an initial testosterone/estrogen flare before achieving suppression at 2-4 weeks. Antagonists achieve immediate suppression without flare, reaching castrate levels within 1-3 days.
Oral GnRH Antagonists: A New Era
The development of orally bioavailable, non-peptide GnRH antagonists represents one of the most significant advances in reproductive endocrinology pharmacology in the past decade. Three oral antagonists have received regulatory approval, transforming the treatment landscape for endometriosis and uterine fibroids.
Elagolix (Orilissa)
Elagolix was the first oral GnRH antagonist approved by the FDA (July 2018) for the management of moderate-to-severe pain associated with endometriosis. It was later approved in combination with estradiol and norethindrone acetate (marketed as Oriahnn) for heavy menstrual bleeding associated with uterine fibroids (May 2020).
Elagolix's dose-dependent mechanism allows clinicians to titrate estrogen suppression:
- 150 mg once daily: Partial estrogen suppression (estradiol levels approximately 40-60 pg/mL). This dose reduces endometriosis pain while maintaining sufficient estrogen to protect bone density and minimize menopausal symptoms. Approved for up to 24 months of use.
- 200 mg twice daily: Near-complete estrogen suppression (estradiol levels less than 12 pg/mL). This dose provides maximum pain relief but carries a higher risk of bone density loss. Approved for up to 6 months without add-back therapy.
Phase III trials (ELARIS EM-I and EM-II) demonstrated significant reductions in dysmenorrhea and non-menstrual pelvic pain compared to placebo at both dose levels. The 200 mg twice daily dose showed greater efficacy but more hypoestrogenic side effects. The lower 150 mg once daily dose offered a favorable benefit-risk profile for long-term management.
Relugolix (Relumina/Orgovyx)
Relugolix has a dual identity in clinical practice. As a standalone oral tablet (Orgovyx, 120 mg daily), it's approved for advanced prostate cancer. In combination with estradiol and norethindrone acetate (Myfembree), it's approved for heavy menstrual bleeding associated with uterine fibroids and pain associated with endometriosis.
The HERO Phase III trial for prostate cancer demonstrated that oral relugolix achieved sustained castration (testosterone less than 50 ng/dL) in 96.7% of patients at 48 weeks, compared to 88.8% with leuprolide depot. Relugolix also showed faster testosterone recovery after treatment discontinuation and a 54% lower risk of major adverse cardiovascular events compared to leuprolide, though this cardiovascular benefit requires further study (Shore et al., 2020).
For the fibroid and endometriosis indications, the combination product Myfembree includes add-back therapy (1 mg estradiol + 0.5 mg norethindrone acetate) to mitigate bone density loss and vasomotor symptoms while maintaining efficacy against target symptoms.
Linzagolix (Yselty)
Linzagolix is the newest oral GnRH antagonist, approved in Europe (2022) for the treatment of moderate-to-severe symptoms of uterine fibroids. It offers flexible dosing with or without add-back therapy:
- 100 mg daily without add-back: Partial estrogen suppression, suitable for long-term use
- 200 mg daily with add-back (1 mg estradiol + 0.5 mg NETA): Full suppression with hormonal protection, also suitable for long-term use
Phase III PRIMROSE trials demonstrated that both dosing strategies significantly reduced heavy menstrual bleeding compared to placebo, with the higher dose plus add-back showing the greatest efficacy.
Agonist vs. Antagonist: Head-to-Head Comparison
| Feature | GnRH Agonists | GnRH Antagonists (Injectable) | GnRH Antagonists (Oral) |
|---|---|---|---|
| Mechanism | Receptor overstimulation leading to downregulation | Direct competitive receptor blockade | Direct competitive receptor blockade |
| Onset of suppression | 2-4 weeks | Hours to days | Hours to days |
| Hormonal flare | Yes (1-2 weeks) | No | No |
| Anti-androgen co-therapy needed | Often (for prostate cancer) | No | No |
| Depot formulations | 1, 3, 6, 12 months | Monthly only (degarelix) | N/A (daily oral) |
| Dose-dependent suppression | Limited (on/off) | Limited at clinical doses | Yes (partial to complete) |
| Reversibility speed | Weeks to months | Days to weeks | Days |
| Cost (relative) | Moderate | Higher | Highest |
| Patient convenience | Good (depot formulations) | Moderate (monthly injections) | Best (daily oral pill) |
| OHSS risk (IVF context) | Higher | Lower | Not typically used for IVF |
WHEN TO CHOOSE WHICH
- Choose a GnRH agonist when: long depot intervals are desired (3-12 months), the initial flare is clinically acceptable (or can be managed with anti-androgens), cost is a major factor, or the patient prefers fewer clinic visits
- Choose an injectable GnRH antagonist when: flare must be avoided (advanced prostate cancer with spinal metastases, IVF protocols), rapid hormonal suppression is needed, or the patient has cardiovascular risk factors (degarelix may offer benefit)
- Choose an oral GnRH antagonist when: dose-dependent estrogen control is desired (endometriosis, fibroids), the patient prefers oral medication over injections, long-term therapy is anticipated with add-back, or the goal is partial rather than complete hormonal suppression
Clinical Applications Across Medical Specialties
Fertility Medicine and Assisted Reproduction
IVF Protocols: The Role of GnRH Analogs
In vitro fertilization requires precise control over the reproductive hormonal cascade. Left unmanaged, the pituitary can produce a premature LH surge that triggers spontaneous ovulation before oocyte retrieval, canceling the IVF cycle. GnRH analogs solve this problem by controlling when (and if) the LH surge occurs.
Three major IVF protocol categories employ GnRH analogs:
1. The Long Agonist Protocol ("Long Down-Regulation")
This was the dominant IVF protocol for decades and remains widely used. A GnRH agonist (typically leuprolide 0.5-1 mg SC daily, or nafarelin intranasally) is started in the mid-luteal phase of the preceding cycle. After 10-14 days of agonist-induced pituitary downregulation, gonadotropin stimulation (FSH with or without LH) begins while continuing the agonist at a reduced dose. The agonist keeps the pituitary suppressed, preventing premature LH surges throughout the stimulation phase.
Advantages of the long agonist protocol include consistent pituitary suppression, synchronization of follicular cohort development (potentially yielding more uniform follicles), extensive clinical experience and established protocols, and historically the highest pregnancy rates in good-prognosis patients.
Disadvantages include longer treatment duration (typically 4-6 weeks total), higher gonadotropin requirements (the suppressed pituitary provides no endogenous FSH contribution), greater risk of ovarian hyperstimulation syndrome (OHSS), and more medication side effects from prolonged agonist exposure.
2. The Antagonist Protocol
GnRH antagonist protocols begin gonadotropin stimulation on cycle day 2-3 without prior pituitary suppression. A GnRH antagonist (cetrorelix or ganirelix, 0.25 mg SC daily) is added on stimulation day 5-6 (fixed protocol) or when the lead follicle reaches 14 mm (flexible protocol) to prevent premature LH surges.
The antagonist protocol has become the predominant approach in many IVF centers due to shorter treatment duration (typically 8-12 days of stimulation), lower total gonadotropin dose requirements, significantly reduced OHSS risk (especially important in PCOS patients), and the ability to use a GnRH agonist trigger instead of hCG. This last point deserves emphasis. In antagonist protocols, a single dose of GnRH agonist (such as leuprolide 1-2 mg or triptorelin 0.2 mg) can serve as the ovulation trigger. This agonist trigger exploits the initial LH/FSH flare to induce oocyte maturation while avoiding the sustained luteal support and OHSS risk associated with hCG. It has made antagonist protocols dramatically safer for high-risk patients.
A Cochrane review of GnRH agonist versus antagonist protocols found comparable live birth rates overall, with antagonist protocols associated with a significant reduction in OHSS incidence. In the general IVF population, some studies suggested slightly lower ongoing pregnancy rates with antagonist protocols, but this difference has narrowed in recent analyses and may be eliminated when optimal antagonist protocol techniques are used (Al-Inany et al., 2011).
3. GnRH Agonist Trigger Protocols
As mentioned above, GnRH agonist triggering is a specific technique used within antagonist protocols. Instead of hCG, a single dose of GnRH agonist (leuprolide 1-4 mg SC or triptorelin 0.1-0.2 mg) is administered when follicles are mature. This triggers an endogenous LH and FSH surge that lasts 24-36 hours - sufficient for oocyte maturation but short enough to prevent OHSS.
GnRH agonist trigger is particularly valuable for oocyte donors, patients with PCOS at high risk for OHSS, "freeze-all" cycles where fresh embryo transfer is not planned, and any patient who has experienced or is at risk for severe OHSS.
IVF PROTOCOL SELECTION SUMMARY
Long agonist protocol: Consider for normal responders, patients with prior antagonist protocol failures, and when maximal follicular synchronization is desired.
Antagonist protocol: Preferred for PCOS patients, poor responders (shorter exposure), patients at high OHSS risk, and when an agonist trigger is planned. This is now the default protocol in many programs.
For a personalized assessment of peptide and hormonal protocols, visit the Free Assessment page.
Fertility Preservation with GnRH Analogs
Beyond IVF, GnRH analogs serve important roles in fertility preservation for cancer patients. Two distinct strategies exist:
Ovarian suppression during chemotherapy: GnRH agonists (typically triptorelin or leuprolide) administered before and during gonadotoxic chemotherapy reduce the incidence of premature ovarian failure. The POEMS and PROMISE-GIM6 trials showed that this approach roughly halves the risk of early menopause after chemotherapy (from approximately 26% to 9%) and increases post-treatment pregnancy rates.
Emergency IVF/oocyte cryopreservation: When cancer diagnosis allows 2-3 weeks before chemotherapy initiation, random-start antagonist protocols allow rapid ovarian stimulation for oocyte or embryo cryopreservation. GnRH antagonists enable stimulation to begin at any point in the menstrual cycle (not just the early follicular phase), minimizing delays.
Prostate Cancer
Androgen deprivation therapy (ADT) with GnRH analogs is the mainstay of treatment for advanced prostate cancer and an established adjuvant therapy for intermediate- and high-risk localized disease. The clinical context dictates which analog type is most appropriate.
Indications for ADT in Prostate Cancer
| Clinical Scenario | ADT Role | Typical Duration |
|---|---|---|
| Metastatic hormone-sensitive prostate cancer (mHSPC) | Backbone of combination therapy (with docetaxel, abiraterone, enzalutamide, or darolutamide) | Indefinite |
| Non-metastatic castration-resistant prostate cancer (nmCRPC) | Continued suppression combined with novel antiandrogens | Indefinite |
| High-risk localized (with radiation) | Neoadjuvant + concurrent + adjuvant | 18-36 months |
| Intermediate-risk localized (with radiation) | Neoadjuvant + concurrent | 4-6 months |
| Biochemical recurrence after primary therapy | Salvage or intermittent ADT | Variable |
Intermittent vs. Continuous ADT
For patients on long-term ADT, intermittent therapy - cycling between treatment and off-treatment phases based on PSA levels - offers potential quality-of-life benefits. During off-treatment phases, testosterone recovers partially, restoring sexual function, energy, and bone density. The landmark SWOG 9346 trial found that intermittent ADT was non-inferior to continuous ADT for overall survival in men with metastatic disease who achieved a PSA of 4 ng/mL or less after 7 months of induction therapy.
Intermittent ADT is typically implemented by initiating GnRH agonist treatment for 6-9 months, monitoring PSA decline, discontinuing therapy when PSA reaches a nadir (typically less than 4 ng/mL), monitoring PSA during the off-treatment phase, and restarting therapy when PSA rises above a predefined threshold (commonly 10-20 ng/mL). GnRH agonists are preferred over antagonists for intermittent therapy because the depot formulations' defined duration aligns naturally with the cycling approach.
Endometriosis
Endometriosis management has evolved significantly with the introduction of oral GnRH antagonists, but injectable GnRH agonists remain important tools, particularly for more severe disease and as pre-surgical therapy.
Medical Management Strategy
GnRH analogs are typically second-line therapy for endometriosis-associated pain, used when first-line treatments (combined oral contraceptives, progestins, NSAIDs) are insufficient. The treatment algorithm generally follows this progression:
- Empirical treatment with hormonal contraceptives and/or progestins
- If inadequate relief: GnRH analog therapy (with add-back therapy for treatment beyond 6 months)
- If medical therapy fails or fertility is desired: surgical excision/ablation of endometriotic implants
- Post-surgical hormonal suppression to prevent recurrence
The oral GnRH antagonists (elagolix, relugolix combination) have shifted this paradigm by offering a more patient-friendly, dose-adjustable option that can be used earlier in the treatment algorithm. Many clinicians now use oral antagonists as second-line therapy, reserving injectable GnRH agonists for refractory cases or pre-surgical preparation.
Pre-Surgical GnRH Agonist Therapy
A 3-month course of GnRH agonist before planned endometriosis surgery offers several benefits: reducing implant size and vascularity (making surgery technically easier), decreasing intraoperative blood loss, thinning the eutopic endometrium, and reducing inflammation in the peritoneal cavity. However, GnRH agonist pre-treatment can also make superficial implants harder to identify at surgery, so opinions on this approach vary among surgeons.
Uterine Fibroids
GnRH analogs are used in fibroid management primarily for two purposes:
Pre-surgical volume reduction: A 3-6 month course of GnRH agonist typically reduces fibroid volume by 30-60%, decreases uterine vascularity, and corrects pre-operative anemia by stopping menstrual bleeding. This can convert a planned abdominal hysterectomy into a less invasive vaginal approach or reduce the complexity of myomectomy.
Long-term symptom management: The oral GnRH antagonists with add-back therapy now provide the first approved long-term medical management option for fibroid-related heavy menstrual bleeding, allowing women to defer or avoid surgery entirely.
Central Precocious Puberty
GnRH agonist therapy is the standard of care for central precocious puberty (CPP), with treatment goals focused on halting premature secondary sexual development, slowing skeletal maturation to improve predicted adult height, addressing psychosocial challenges of early puberty, and preserving future fertility potential.
Available agents for CPP include leuprolide depot (monthly or 3-month), triptorelin depot (monthly), nafarelin nasal spray (twice daily), and histrelin implant (annual). Treatment is typically initiated when puberty begins before age 8 in girls or 9 in boys, with documented progression and bone age advancement. It continues until the patient reaches an age-appropriate pubertal stage (typically age 11-12 in girls, 12-13 in boys), at which point the GnRH agonist is discontinued.
Long-term outcome studies have consistently shown that GnRH agonist treatment for CPP is safe, effective, and fully reversible. Puberty resumes within 3-12 months of discontinuation. Adult height is improved by an estimated 3-10 cm compared to untreated CPP. Reproductive function, including menstruation, ovulation, and fertility, recovers normally.
Gender-Affirming Care
GnRH agonists play an established role in gender-affirming medical care for transgender and gender-diverse adolescents. Puberty suppression with GnRH agonists serves several purposes in this context:
- Pausing the development of unwanted secondary sex characteristics associated with the birth-assigned sex
- Providing additional time for psychological assessment and exploration of gender identity
- Reducing gender dysphoria by preventing the distressing physical changes of endogenous puberty
- Potentially improving outcomes if the individual subsequently pursues gender-affirming hormone therapy (e.g., less need for surgical interventions to reverse pubertal changes)
The Endocrine Society guidelines recommend consideration of GnRH agonist therapy when the adolescent has reached Tanner stage 2 of puberty, has a documented and persistent pattern of gender dysphoria, has undergone appropriate psychological assessment, and has provided informed consent (with parental consent for minors).
A systematic review published in Frontiers in Endocrinology (2025) synthesized evidence from 19 studies on GnRH agonist use in transgender and gender-diverse youth, finding effective suppression of pubertal development with acceptable safety profiles. Studies have reported improvements in psychological well-being, quality of life, and reductions in depression and suicidal ideation, though the strength of evidence varies across outcomes and longer-term data continues to accumulate.
GnRH agonists used in this context are the same agents and formulations used for CPP. Leuprolide depot and triptorelin depot are the most commonly prescribed. The histrelin annual implant is also used. Treatment continues until the individual reaches an appropriate age and readiness for gender-affirming hormones (estradiol for transfeminine individuals, testosterone for transmasculine individuals) or decides to discontinue puberty suppression and allow endogenous puberty to resume.
EVOLVING REGULATORY LANDSCAPE
The use of GnRH agonists for puberty suppression in gender-diverse youth has become a subject of intense medical, ethical, and political debate in several countries. Some jurisdictions have restricted or paused their use outside of clinical research settings (e.g., England's NHS following the Cass Review in 2024), while others have reaffirmed their place in clinical practice based on existing evidence (e.g., the Endocrine Society, WPATH). Clinicians should follow current guidelines from relevant professional organizations and be aware of local regulatory requirements when considering this intervention. Independent reviews have yielded varying conclusions about the evidence base, reflecting the challenges of conducting long-term randomized trials in this population.
Figure 5: Clinical applications of GnRH analogs span multiple medical specialties, from fertility medicine and oncology to pediatric endocrinology and gender-affirming care.
Breast Cancer
In premenopausal women with hormone receptor-positive (HR+) breast cancer, ovarian function suppression (OFS) with GnRH agonists is an established treatment strategy. The SOFT (Suppression of Ovarian Function Trial) and TEXT (Tamoxifen and Exemestane Trial) studies demonstrated that adding OFS with triptorelin to endocrine therapy improved disease-free survival in premenopausal women with HR+ early breast cancer, particularly in higher-risk patients who received chemotherapy.
The standard approach uses triptorelin 3.75 mg monthly or goserelin 3.6 mg monthly for 5 years, combined with either tamoxifen or an aromatase inhibitor (the latter requiring OFS because aromatase inhibitors alone do not suppress ovarian estrogen production in premenopausal women).
Other Applications
Less common but established applications of GnRH analogs include:
- Premenstrual dysphoric disorder (PMDD): GnRH agonists can serve as a diagnostic and therapeutic tool for severe PMDD, confirming the hormonal basis of symptoms and providing relief. They may also be used as a "test run" before considering bilateral oophorectomy.
- Menstrual suppression before surgery: Short courses of GnRH agonists can correct anemia caused by heavy menstrual bleeding before planned surgery.
- Ovarian cyst management: GnRH agonists can help resolve functional ovarian cysts by suppressing gonadotropin stimulation.
- Severe hyperandrogenism: In combination with estrogen-progestin replacement, GnRH agonists can treat severe hirsutism and acne unresponsive to conventional therapy.
Comprehensive Dosing Reference
The following tables consolidate dosing information across all GnRH analogs discussed in this report. These are general guidelines drawn from FDA-approved prescribing information and clinical practice; individual dosing should always be determined by the prescribing physician based on the specific clinical context. Use the FormBlends Dosing Calculator for additional guidance on peptide dosing.
GnRH Agonist Dosing by Indication
| Indication | Agent | Dose | Route | Schedule |
|---|---|---|---|---|
| Prostate cancer (ADT) | Leuprolide (Lupron Depot) | 7.5 / 22.5 / 30 / 45 mg | IM | q1 / q3 / q4 / q6 months |
| Triptorelin (Trelstar) | 3.75 / 11.25 / 22.5 mg | IM | q1 / q3 / q6 months | |
| Goserelin (Zoladex) | 3.6 / 10.8 mg | SC implant | q1 / q3 months | |
| Histrelin (Vantas) | 50 mg implant | SC implant | q12 months | |
| Endometriosis | Leuprolide (Lupron Depot) | 3.75 / 11.25 mg | IM | q1 / q3 months (max 6 months) |
| Nafarelin (Synarel) | 200 mcg per spray | Intranasal | 400-800 mcg/day (6 months) | |
| Goserelin (Zoladex) | 3.6 mg | SC implant | q1 month (max 6 months) | |
| Central precocious puberty | Leuprolide (Lupron Depot-Ped) | 7.5-15 mg | IM | q1 month |
| Nafarelin (Synarel) | 200 mcg per spray | Intranasal | 1600 mcg/day (2x daily) | |
| Histrelin (Supprelin LA) | 50 mg implant | SC implant | q12 months | |
| TRT fertility preservation | Gonadorelin | 100-200 mcg | SC | 2-3x weekly |
| IVF pituitary suppression | Leuprolide | 0.5-1.0 mg daily, then 0.25-0.5 mg daily | SC | Daily starting mid-luteal |
GnRH Antagonist Dosing by Indication
| Indication | Agent | Dose | Route | Schedule |
|---|---|---|---|---|
| IVF (LH surge prevention) | Cetrorelix (Cetrotide) | 0.25 mg or 3 mg | SC | 0.25 mg daily or single 3 mg dose |
| Ganirelix (Orgalutran) | 0.25 mg | SC | Daily from stimulation day 5-6 | |
| Prostate cancer | Degarelix (Firmagon) | 240 mg loading, 80 mg maintenance | SC | Loading: 2x120 mg; then 80 mg q1 month |
| Relugolix (Orgovyx) | 120 mg (after 360 mg loading) | Oral | Daily | |
| Endometriosis pain | Elagolix (Orilissa) | 150 mg or 200 mg | Oral | 150 mg QD (24 mo) or 200 mg BID (6 mo) |
| Relugolix combo (Myfembree) | 40 mg + E2/NETA | Oral | Daily | |
| Uterine fibroids (HMB) | Elagolix combo (Oriahnn) | 300 mg BID + E2/NETA | Oral | Daily (max 24 months) |
| Relugolix combo (Myfembree) | 40 mg + E2/NETA | Oral | Daily | |
| Linzagolix (Yselty) | 100 mg or 200 mg + E2/NETA | Oral | Daily |
Safety, Side Effects, and Monitoring
Universal Side Effects of GnRH-Mediated Sex Steroid Suppression
Because all GnRH analogs (agonists and antagonists alike) ultimately work by reducing sex steroid production, they share a common set of hypoestrogenic/hypoandrogenic side effects. The severity and clinical significance depend on the degree and duration of suppression.
Short-Term Effects (Present During Active Treatment)
- Vasomotor symptoms: Hot flashes and night sweats are the most common complaint, affecting 50-90% of patients. They result from estrogen withdrawal effects on hypothalamic thermoregulatory centers.
- Sexual dysfunction: Decreased libido, erectile dysfunction (men), and vaginal dryness/dyspareunia (women) are common and directly related to sex steroid suppression.
- Mood changes: Depression, anxiety, emotional lability, and cognitive complaints ("brain fog") are reported by 10-30% of patients. These may reflect direct hormonal effects on neurotransmitter systems and/or psychological responses to other symptoms.
- Fatigue and reduced energy: Reported by 20-40% of patients, particularly men on ADT.
- Weight gain and metabolic changes: Fat redistribution (increased visceral adiposity), insulin resistance, and dyslipidemia can develop during prolonged suppression.
- Musculoskeletal symptoms: Joint pain, muscle weakness, and general musculoskeletal discomfort are common, especially with complete hormonal suppression.
Long-Term Risks (With Prolonged Treatment)
- Bone mineral density (BMD) loss: This is perhaps the most clinically significant long-term risk. GnRH agonists used for endometriosis cause 2-8% BMD loss at the lumbar spine over 6 months, which is largely reversible after discontinuation. However, with prolonged ADT for prostate cancer (years), cumulative BMD loss can reach 5-10% per year initially, significantly increasing fracture risk. Monitoring with periodic DEXA scans and consideration of bone-protective therapy (bisphosphonates, denosumab) is essential for patients on long-term GnRH analog therapy.
- Cardiovascular risk: ADT in men has been associated with increased risks of coronary artery disease, myocardial infarction, stroke, and metabolic syndrome. The magnitude of this risk remains debated, but current guidelines recommend cardiovascular risk assessment before initiating ADT and ongoing monitoring of metabolic parameters. Some data suggest GnRH antagonists may carry lower cardiovascular risk than agonists, but this requires further confirmation.
- Diabetes risk: Insulin resistance and hyperglycemia associated with testosterone deprivation may increase the risk of type 2 diabetes, particularly in men with pre-existing metabolic risk factors.
- Cognitive effects: Some studies have suggested an association between long-term ADT and cognitive decline, including possible increased risk of Alzheimer's disease, though this remains controversial and is not yet established as causal.
Agent-Specific Safety Considerations
| Agent/Class | Unique Safety Concern | Management |
|---|---|---|
| GnRH agonists (all) | Testosterone/estrogen flare (days 1-10) | Anti-androgen co-therapy for prostate cancer patients |
| Nafarelin | Nasal irritation; altered absorption with concurrent nasal medications | Avoid nasal decongestants; space other nasal meds by 2+ hours |
| Degarelix | Injection site reactions (40%); pain, erythema, swelling | Rotate injection sites; proper subcutaneous technique |
| Elagolix | Hepatotoxicity (rare; elevated ALT/AST) | Monitor liver function; contraindicated in severe hepatic impairment |
| Relugolix (oral) | P-glycoprotein drug interactions (reduced efficacy with P-gp inducers) | Avoid co-administration with strong P-gp inducers |
| All GnRH analogs | Bone density loss with prolonged use | Add-back therapy; DEXA monitoring; bisphosphonates if needed |
Monitoring Recommendations
Appropriate monitoring during GnRH analog therapy depends on the indication and expected treatment duration:
Prostate Cancer (ADT)
- Testosterone levels: at 1, 3, and 6 months, then every 6-12 months (target: less than 50 ng/dL, preferably less than 20 ng/dL)
- PSA: every 3-6 months
- DEXA scan: baseline, then every 1-2 years
- Metabolic panel (glucose, HbA1c, lipids): baseline, 3 months, then annually
- Blood pressure and body weight: each visit
- Cardiovascular risk assessment: baseline and ongoing
Endometriosis/Fibroids
- Symptom assessment: monthly during initial therapy, then every 3 months
- DEXA scan: baseline if risk factors present; after 6 months of injectable GnRH agonist use
- Liver function (elagolix): baseline, then periodically
- Estradiol levels: if there is concern about treatment adequacy or excessive suppression
Central Precocious Puberty
- Growth velocity and height measurements: every 3-6 months
- Tanner stage assessment: every 6 months
- Bone age X-ray: every 6-12 months
- LH (stimulated or random): to confirm adequate suppression
- Estradiol or testosterone: to confirm prepubertal levels
TRT Adjunctive Gonadorelin
- Semen analysis: baseline, then every 6-12 months if fertility preservation is a goal
- Testicular volume (by exam or ultrasound): every 6-12 months
- LH, FSH, total testosterone, free testosterone: every 3-6 months initially, then every 6-12 months
COMPLEMENTARY PEPTIDE STRATEGIES
Patients using GnRH analogs for various conditions may benefit from complementary peptide therapies. For example, men on ADT experiencing joint pain and tissue healing concerns may consider BPC-157 for its tissue repair properties. Those dealing with muscle loss and metabolic changes might explore growth hormone secretagogues like CJC-1295/Ipamorelin or MK-677 for body composition support (under appropriate medical supervision). The relationship between oxytocin and reproductive health is explored in our Oxytocin Research Report. Always consult with a healthcare provider before combining peptide therapies.
Future Directions and Emerging Research
Novel GnRH Analog Development
The pharmaceutical pipeline for GnRH-targeted therapeutics continues to expand, with several promising avenues under investigation:
Long-Acting Injectable Antagonists: One major limitation of current injectable GnRH antagonists is the requirement for monthly injections (degarelix). Depot formulations of GnRH antagonists providing 3- to 6-month suppression would combine the pharmacological advantages of antagonists (no flare, immediate onset, potential cardiovascular benefit) with the convenience of depot agonist formulations. Several candidates are in preclinical and early clinical development.
Next-Generation Oral Antagonists: Following the success of elagolix, relugolix, and linzagolix, pharmaceutical companies are developing oral GnRH antagonists with improved pharmacokinetic profiles. Goals include once-daily dosing with more consistent plasma levels, reduced food-effect variability, fewer drug-drug interactions, and expanded indications.
Kisspeptin-Based Therapeutics: As the upstream regulator of GnRH secretion, kisspeptin offers alternative approaches to HPG axis modulation. Kisspeptin receptor agonists and antagonists could provide more physiological regulation of GnRH pulsatility, potentially offering therapeutic benefits with fewer side effects than direct GnRH receptor manipulation. Early clinical studies of kisspeptin administration for IVF triggering, hypothalamic amenorrhea, and diagnostic testing have shown promising results. For details, see our Kisspeptin Research Report.
Targeted GnRH-Drug Conjugates: Researchers are exploring the use of GnRH peptides as targeting vectors for drug delivery to GnRH receptor-expressing tumors. By conjugating cytotoxic agents to GnRH analogs, it may be possible to deliver chemotherapy selectively to cancer cells that express GnRH receptors (including certain prostate, breast, ovarian, and endometrial cancers) while sparing normal tissues.
Expanded Clinical Applications
Alzheimer's Disease: Preclinical research suggests that GnRH may play a role in cognitive function through extra-pituitary receptors. Some researchers have proposed that modulating GnRH signaling could influence amyloid processing and neurodegeneration. A Phase II clinical trial (the LUCINDA trial) is investigating whether leuprolide plus add-back estrogen therapy can slow cognitive decline in postmenopausal women with Alzheimer's disease. Results are anticipated but not yet published.
Polycystic Ovary Syndrome (PCOS): Women with PCOS often have abnormally high GnRH pulse frequency, driving excessive LH secretion and ovarian androgen production. GnRH antagonists at low doses could potentially normalize LH pulsatility in PCOS, though this remains experimental. Kisspeptin antagonists represent another approach, as kisspeptin may mediate the accelerated GnRH pulsatility characteristic of PCOS.
Combination Immunotherapy for Prostate Cancer: Emerging evidence suggests that ADT with GnRH analogs may enhance the efficacy of immune checkpoint inhibitors in prostate cancer by modulating the tumor immune microenvironment. Clinical trials combining ADT with anti-PD-1/PD-L1 antibodies and other immunotherapies are ongoing.
Male Contraception: GnRH antagonists, in combination with exogenous testosterone, have been investigated as a male hormonal contraceptive approach. The antagonist suppresses FSH-driven spermatogenesis, while exogenous testosterone maintains androgenic function. This approach has shown efficacy in clinical trials but has not yet achieved regulatory approval for contraceptive use.
Precision Medicine Approaches
The future of GnRH analog therapy may increasingly involve personalized approaches:
- Pharmacogenomics: Genetic variants in the GnRH receptor gene, enzymes involved in drug metabolism, and hormone receptor genes may predict individual responses to specific GnRH analogs. Research in this area is still early but could eventually guide agent selection.
- Biomarker-Guided Dosing: Rather than fixed dosing schedules, real-time hormone monitoring could enable personalized dose adjustments to achieve optimal suppression with minimal side effects.
- Circulating Tumor DNA (ctDNA) Monitoring: In prostate cancer, ctDNA analysis could help determine the optimal timing for starting, stopping, and restarting ADT in intermittent therapy protocols.
Figure 6: Evolution and future pipeline of GnRH-targeted therapeutics, from first-generation injectable agonists to oral antagonists and emerging kisspeptin-based approaches.
Drug Interactions and Special Populations
Significant Drug Interactions
Most injectable GnRH analogs (agonists and antagonists) have relatively few clinically significant pharmacokinetic drug interactions because they are peptides cleared through enzymatic degradation rather than hepatic CYP metabolism. However, the oral GnRH antagonists are small molecules with more complex interaction profiles.
| GnRH Analog | Interacting Drug/Class | Effect | Clinical Action |
|---|---|---|---|
| Elagolix | Strong CYP3A4 inhibitors (ketoconazole, itraconazole) | Increased elagolix exposure | Limit elagolix to 150 mg QD for max 6 months |
| Elagolix | Strong CYP3A4 inducers (rifampin, phenytoin) | Decreased elagolix exposure | Avoid co-administration |
| Elagolix | OATP1B1 substrates (rosuvastatin) | Increased substrate exposure | Monitor; may need substrate dose adjustment |
| Relugolix (oral) | P-glycoprotein inducers (rifampin, St. John's Wort) | Decreased relugolix exposure; risk of testosterone escape | Avoid co-administration; if unavoidable, increase relugolix dose to 240 mg daily temporarily |
| Relugolix (oral) | P-glycoprotein inhibitors (ketoconazole, erythromycin) | Increased relugolix exposure | Avoid co-administration if possible |
| All GnRH agonists | QT-prolonging medications | Additive QT prolongation risk (due to hypogonadism-related cardiac effects) | ECG monitoring; avoid combinations when possible |
| Nafarelin | Nasal decongestants (oxymetazoline) | Altered nafarelin absorption | Space administration by at least 2 hours |
| All GnRH analogs | Drugs affecting bone density (corticosteroids, anticonvulsants) | Additive bone loss risk | Enhanced BMD monitoring; consider bone-protective agents |
Special Populations
Pediatric Patients
GnRH agonists have an extensive safety record in children with central precocious puberty and are considered the standard of care. Key considerations for pediatric use include the following. Weight-based dosing is not typically used; depot formulations are chosen based on the desired injection interval and available dose strengths. Adherence monitoring is important, especially with daily nasal formulations. Bone age and growth velocity should be tracked every 6-12 months to confirm treatment adequacy. Long-term follow-up studies spanning 20+ years have not identified significant adverse effects on reproductive function, bone density, or general health outcomes following treatment discontinuation.
Hepatic Impairment
Injectable GnRH agonists and antagonists generally do not require dose adjustment for hepatic impairment, as they undergo enzymatic degradation rather than hepatic metabolism. However, oral GnRH antagonists are an exception. Elagolix is contraindicated in severe hepatic impairment (Child-Pugh C) and requires dose limitation in moderate impairment (Child-Pugh B: maximum 150 mg daily for 6 months). Relugolix and linzagolix also require caution in severe hepatic disease due to their hepatic metabolism.
Renal Impairment
Dose adjustment is generally not required for GnRH agonists in patients with renal impairment. The oral antagonists have limited data in severe renal impairment, and cautious use is advised. Degarelix requires no dose adjustment for mild to moderate renal impairment; data in severe impairment is limited.
Elderly Patients
Given that prostate cancer predominantly affects older men, the majority of ADT recipients are elderly. Age-related considerations include increased baseline osteoporosis risk (enhanced by ADT-related bone loss), higher cardiovascular disease prevalence (potentially exacerbated by ADT), sarcopenia risk (accelerated by testosterone suppression), and falls and fracture risk (compounded by bone loss and muscle weakness). These factors reinforce the importance of comprehensive geriatric assessment before initiating long-term ADT and proactive management of cardiovascular and skeletal health throughout treatment.
Pregnancy and Lactation
All GnRH analogs are contraindicated in pregnancy (FDA Category X). GnRH agonists and antagonists can cause fetal harm through disruption of hormonal signaling essential for pregnancy maintenance. Women of reproductive potential must use non-hormonal contraception during treatment (hormonal contraceptives may be less effective during GnRH analog therapy due to altered HPG axis dynamics). Treatment should be discontinued immediately if pregnancy is suspected or confirmed.
Practical Guidance for Clinicians and Patients
Choosing the Right GnRH Analog
Selecting the optimal GnRH analog for a given patient requires weighing several factors:
| Factor | Considerations | Recommendation |
|---|---|---|
| Indication | What condition is being treated? | Match agent to FDA-approved indication when possible |
| Flare tolerance | Can the patient safely undergo the initial hormonal flare? | Use antagonist if flare is contraindicated (spinal mets, high-burden disease) |
| Treatment duration | Short-term (months) vs. long-term (years)? | Depot agonists for long-term; oral antagonists for flexible shorter courses |
| Route preference | Injection vs. oral vs. nasal vs. implant | Oral antagonists for injection-averse; implants for maximum convenience |
| Dose flexibility | Need for partial vs. complete suppression? | Oral antagonists allow dose-dependent control |
| Cost/insurance | Coverage varies substantially between agents | Generic leuprolide/goserelin often most cost-effective |
| Cardiovascular risk | Patient's baseline CV risk profile | Consider degarelix or relugolix if high CV risk (emerging evidence) |
| Fertility goals | Current or future fertility desired? | Gonadorelin for TRT adjunct; GnRH agonist for chemo-protection |
Patient Education Points
When counseling patients starting GnRH analog therapy, the following points should be addressed:
- Onset expectations: GnRH agonists require 2-4 weeks to reach full effect, with a possible worsening of symptoms during the initial flare period. Antagonists and oral agents work within days.
- Side effect management: Hot flashes can be managed with lifestyle measures (layered clothing, cool environments, avoiding triggers like caffeine and alcohol), and in some cases with medications like low-dose venlafaxine or gabapentin.
- Bone health: Weight-bearing exercise, adequate calcium (1000-1200 mg/day) and vitamin D (800-1000 IU/day) supplementation, and avoidance of tobacco and excessive alcohol help mitigate bone density loss.
- Sexual health: Open discussion of sexual side effects, referral to sexual health specialists when appropriate, and awareness that effects are reversible after treatment discontinuation.
- Adherence: For daily medications (oral antagonists, nasal sprays), consistent timing is important for maintaining therapeutic effect. Missed doses of GnRH agonist injections should be administered as soon as possible to avoid hormone escape.
- Reversibility: In most cases, normal hormonal function returns within weeks to months after discontinuing GnRH analog therapy. However, recovery time varies by agent, duration of use, and individual patient factors.
Storage and Handling
| Formulation Type | Storage | Key Handling Notes |
|---|---|---|
| Reconstituted injectable (gonadorelin, daily triptorelin) | Refrigerate (2-8C); use within stated stability period after reconstitution | Protect from light; do not freeze |
| Depot microspheres (leuprolide, triptorelin) | Room temperature (most); some require refrigeration | Reconstitute immediately before injection; do not re-use if settling occurs |
| SC implants (goserelin, histrelin) | Room temperature | Use pre-loaded syringes as packaged; check expiration date |
| Nasal spray (nafarelin) | Room temperature, upright position | Prime before first use; protect from light; discard after stated uses |
| Oral tablets (elagolix, relugolix, linzagolix) | Room temperature | No special handling; store in original packaging |
Figure 7: Administration routes for GnRH analogs range from daily nasal spray and oral tablets to long-acting depot injections and annual subcutaneous implants, offering flexibility to match patient preferences and clinical needs.
Integration with Broader Peptide Therapy Protocols
GnRH analogs do not exist in isolation within the peptide therapy landscape. Understanding how they relate to and can be combined with other peptide therapeutics provides a more complete clinical picture.
GnRH Analogs and Growth Hormone Secretagogues
Men receiving long-term ADT with GnRH agonists often experience adverse body composition changes - increased fat mass, decreased lean mass, and reduced bone density. Growth hormone secretagogues such as CJC-1295/Ipamorelin and MK-677 (Ibutamoren) are sometimes explored to address these changes, though their use in the setting of hormone-sensitive cancers requires careful consideration and oncologist oversight.
In non-oncological settings (such as TRT with adjunctive gonadorelin), growth hormone secretagogues may complement the protocol by supporting lean mass maintenance and metabolic health. However, formal clinical data on these combinations remains limited, and decisions should be made on a case-by-case basis with appropriate medical supervision.
Kisspeptin and GnRH: Upstream and Downstream
Kisspeptin-10, the truncated active fragment of the kisspeptin peptide, acts upstream of GnRH to modulate its release from hypothalamic neurons. The relationship between these two peptides offers interesting therapeutic possibilities. Kisspeptin administration can serve as an alternative IVF trigger, inducing a more physiological LH surge than either hCG or GnRH agonist triggers. In combination with GnRH axis assessment, kisspeptin testing can help localize the level of reproductive axis dysfunction (hypothalamic vs. pituitary). Future therapeutic development may yield kisspeptin receptor modulators that offer more nuanced control of GnRH pulsatility than current direct-acting GnRH analogs.
Tissue Repair Peptides During Hormonal Therapy
Patients undergoing GnRH-mediated hormonal suppression may experience musculoskeletal symptoms, joint pain, and slower tissue healing. Peptides like BPC-157, with its well-documented tissue repair properties, are sometimes considered as supportive therapy. While BPC-157 acts through mechanisms unrelated to the HPG axis (primarily angiogenic, anti-inflammatory, and cytoprotective pathways), its use should be discussed with the treating physician, particularly in oncology patients where any growth-promoting agent requires careful evaluation.
Oxytocin and Reproductive Health
The relationship between oxytocin and the reproductive system intersects with GnRH biology at several points. Oxytocin influences uterine contractility, sexual function, and social bonding, all of which can be affected by GnRH analog therapy. Patients experiencing sexual dysfunction or relationship stress during GnRH analog treatment may benefit from exploring oxytocin's role in these domains, as detailed in our Oxytocin Research Report.
For a broader overview of how various peptide therapies interconnect, visit the Peptide Hub.
Detailed Comparison Tables
Complete GnRH Analog Comparison
| Property | Gonadorelin | Leuprolide | Triptorelin | Goserelin | Nafarelin | Histrelin |
|---|---|---|---|---|---|---|
| Type | Native GnRH | Agonist | Agonist | Agonist | Agonist | Agonist |
| Position 6 Modification | None (Gly) | D-Leu | D-Trp | D-Ser(tBu) | D-Nal(2) | D-His(Bzl) |
| Relative Potency | 1x | 15-50x | 100x | 100x | 200x | 100x |
| Half-Life | 2-4 min (IV) | ~3 hours | ~3-4 hours | ~4 hours | ~3 hours | ~4 hours |
| Routes | IV, SC | IM, SC | IM | SC implant | Intranasal | SC implant |
| Max Depot Duration | N/A | 6 months | 6 months | 3 months | N/A (daily) | 12 months |
| FDA-Approved Indications | Diagnostic; hypothalamic amenorrhea | Prostate cancer; endometriosis; fibroids; CPP | Prostate cancer | Prostate cancer; breast cancer; endometriosis | Endometriosis; CPP | Prostate cancer; CPP |
| Causes Hormonal Flare | No (at pulsatile doses) | Yes | Yes | Yes | Yes | Yes (mild with implant) |
| Approximate Monthly Cost (US) | $15-50 (compounded) | $400-1,200 | $500-1,500 | $500-1,200 | $600-800 | $3,000-5,000 (annualized) |
GnRH Antagonist Comparison
| Property | Cetrorelix | Ganirelix | Degarelix | Elagolix | Relugolix | Linzagolix |
|---|---|---|---|---|---|---|
| Molecule Type | Peptide | Peptide | Peptide | Non-peptide | Non-peptide | Non-peptide |
| Route | SC injection | SC injection | SC injection | Oral | Oral | Oral |
| Primary Indication | IVF (COS) | IVF (COS) | Prostate cancer | Endometriosis; fibroids | Prostate cancer; endometriosis; fibroids | Fibroids |
| Half-Life | 12-30 hours | 12-16 hours | 28-43 days | 4-6 hours | 25 hours | 15-24 hours |
| Time to Suppression | 4-8 hours | 4-8 hours | 1-3 days | Hours | 1-2 days | Hours |
| Histamine Release Risk | Moderate | Low-moderate | Low | None | None | None |
| Dose-Dependent Control | Limited | Limited | Limited | Yes | Yes | Yes |
| Add-Back Combination Available | No | No | No | Yes (Oriahnn) | Yes (Myfembree) | Yes (Yselty) |
| Regulatory Status (US) | FDA approved | FDA approved | FDA approved | FDA approved | FDA approved | Not yet (EU approved) |
GnRH Analogs by Clinical Application
| Clinical Application | First-Line Agent(s) | Alternative(s) | Key Consideration |
|---|---|---|---|
| Advanced prostate cancer (initial ADT) | Leuprolide or triptorelin depot | Degarelix; relugolix (oral) | Anti-androgen flare protection with agonists |
| Prostate cancer with spinal metastases | Degarelix or relugolix | GnRH agonist + anti-androgen | Avoid flare at all costs |
| Prostate cancer with CV risk | Relugolix (oral) or degarelix | GnRH agonist with CV monitoring | Emerging evidence favors antagonists |
| Endometriosis (moderate-severe pain) | Elagolix 150 mg daily or relugolix combo | Leuprolide/nafarelin + add-back | Oral antagonists allow dose titration |
| Uterine fibroids (HMB) | Relugolix combo or elagolix combo | Leuprolide depot (pre-surgical) | Oral options allow long-term management |
| Central precocious puberty | Leuprolide depot or histrelin implant | Triptorelin depot; nafarelin nasal | Annual implant maximizes adherence |
| IVF pituitary suppression | Cetrorelix or ganirelix (antagonist protocol) | Leuprolide (long agonist protocol) | Antagonist protocol is now standard in many centers |
| IVF ovulation trigger | GnRH agonist (leuprolide or triptorelin) in antagonist cycles | hCG trigger | Agonist trigger nearly eliminates OHSS risk |
| Fertility preservation during TRT | Gonadorelin 100-200 mcg SC 2-3x/week | hCG (if available) | hCG generally more effective; gonadorelin is alternative |
| Breast cancer (premenopausal OFS) | Goserelin or triptorelin monthly | Leuprolide depot | Combined with tamoxifen or AI for 5 years |
| Gender-affirming puberty suppression | Leuprolide depot or triptorelin depot | Histrelin implant | Same agents/doses as for CPP |
Figure 8: Comprehensive comparison of GnRH agonists and antagonists across key pharmacological and clinical parameters, designed to support evidence-based agent selection.
Regulatory Status and Cost Considerations
Global Regulatory Landscape
The regulatory status of GnRH analogs varies significantly across jurisdictions. While the core agents (leuprolide, triptorelin, goserelin) are approved in most major markets, newer oral antagonists have different approval timelines across regions.
| Agent | FDA (US) | EMA (EU) | Notable Regional Differences |
|---|---|---|---|
| Leuprolide | Approved (multiple indications) | Approved | Broadest approved indication set globally |
| Triptorelin | Approved (prostate cancer) | Approved (broader indications) | More indications in EU than US |
| Goserelin | Approved | Approved | Available worldwide |
| Nafarelin | Approved (endometriosis, CPP) | Approved | Limited availability in some markets |
| Histrelin | Approved (CPP, prostate cancer) | Limited approval | Primarily US market |
| Cetrorelix | Approved (IVF) | Approved | Widely available globally |
| Ganirelix | Approved (IVF) | Approved | Widely available globally |
| Degarelix | Approved (prostate cancer) | Approved | Available in most major markets |
| Elagolix | Approved | Not approved | US-only as of 2026 |
| Relugolix | Approved (all 3 indications) | Approved (some indications) | Different formulations in US vs. Japan |
| Linzagolix | Under FDA review | Approved (fibroids) | EU approved; US pending |
Cost and Access Considerations
The cost of GnRH analog therapy varies enormously depending on the specific agent, formulation, duration of treatment, and insurance coverage. Some general patterns are worth noting.
Generic availability has significantly reduced costs for older GnRH agonists. Generic leuprolide depot and goserelin are available in many markets at substantially lower cost than branded products. Triptorelin generic availability varies by region.
The newer oral GnRH antagonists (elagolix, relugolix, linzagolix) are currently available only as branded products and carry higher costs, typically ranging from $800-2,000 per month in the United States without insurance. However, manufacturer patient assistance programs, specialty pharmacy discounts, and insurance formulary placement can significantly reduce out-of-pocket costs.
Compounded gonadorelin for TRT adjunctive use is typically the most affordable option in the GnRH analog class, with costs of approximately $15-50 per month from compounding pharmacies. This cost advantage, combined with the unavailability of compounded hCG since the biologics reclassification, has driven substantial growth in gonadorelin prescriptions for this indication.
For patients exploring gonadorelin and other peptide options, the Free Assessment can help identify appropriate options based on individual health goals and circumstances.
Frequently Asked Questions
1. What are GnRH analogs, and how do they work?
GnRH analogs are synthetic versions of gonadotropin-releasing hormone, the body's master reproductive hormone. They come in two types. GnRH agonists (like triptorelin, leuprolide, and nafarelin) initially overstimulate the pituitary gland, causing a short-term hormone surge. After 2-4 weeks, this continuous stimulation exhausts the pituitary's GnRH receptors, shutting down production of LH, FSH, and downstream sex hormones like testosterone and estrogen. GnRH antagonists (like degarelix, cetrorelix, and the oral agents elagolix and relugolix) block GnRH receptors directly, producing immediate suppression without any initial flare. Both approaches are used to treat hormone-dependent conditions including prostate cancer, endometriosis, fibroids, precocious puberty, and infertility.
2. How does gonadorelin help preserve fertility during testosterone replacement therapy (TRT)?
Gonadorelin is a synthetic version of the body's own GnRH. When injected subcutaneously at doses of 100-200 mcg two to three times weekly, it stimulates the pituitary to release LH and FSH, partially counteracting the suppressive effect that exogenous testosterone has on these hormones. This helps maintain some degree of intratesticular testosterone production and spermatogenesis, potentially preserving testicular size and fertility potential. It became widely used after hCG was reclassified as a biologic and removed from compounding pharmacies. However, gonadorelin's very short half-life means it may be less effective than hCG for this purpose, and results vary between individuals.
3. What is the difference between a GnRH agonist and a GnRH antagonist?
The fundamental difference is how they achieve hormonal suppression. GnRH agonists activate pituitary GnRH receptors so intensely and continuously that the receptors downregulate and stop functioning - essentially wearing out the system. This produces an initial 1-2 week hormonal flare before suppression begins. GnRH antagonists physically block the receptor, preventing natural GnRH from binding. This provides immediate suppression without any flare. In clinical practice, this means agonists need 2-4 weeks to reach full effect (and may require anti-androgen cover during the flare), while antagonists work within hours to days. Agonists are available in long-acting depot formulations lasting up to 12 months, while most injectable antagonists require monthly dosing. The oral antagonists offer daily pill convenience.
4. What is triptorelin used for in prostate cancer treatment?
Triptorelin is a GnRH agonist used as androgen deprivation therapy (ADT) for advanced prostate cancer. By causing sustained suppression of testosterone to castrate levels (below 50 ng/dL, and often below 20 ng/dL), triptorelin starves testosterone-dependent prostate cancer cells of their growth signal. It's available in 1-month (3.75 mg), 3-month (11.25 mg), and 6-month (22.5 mg) depot injections. Clinical trials show it achieves castrate testosterone levels in over 95% of patients and is clinically equivalent to leuprolide. Recent analysis suggests that patients achieving deeper testosterone suppression during triptorelin treatment may have improved survival outcomes.
5. How is nafarelin used for endometriosis, and how effective is it?
Nafarelin is a GnRH agonist administered as a nasal spray (brand name Synarel) for endometriosis treatment. The standard dose is 200 mcg sprayed into each nostril once daily (400 mcg total) for six months. By suppressing estrogen production, it causes endometriotic implants to shrink and symptoms to improve. In clinical trials, nafarelin reduced laparoscopic disease scores by 40-50% and decreased the prevalence of severe pain from approximately 40% to under 10%. About 39% of women who tried to conceive after treatment achieved pregnancy. Side effects include hot flashes (90%), vaginal dryness, and modest bone density loss that typically reverses after stopping treatment.
6. Are GnRH analogs safe for long-term use?
Safety depends on the specific situation and duration. For short-term use (3-6 months, as in endometriosis), GnRH analogs are generally well-tolerated with reversible side effects. For long-term use (years, as in prostate cancer ADT), more significant concerns arise: progressive bone density loss increasing fracture risk, metabolic changes (weight gain, insulin resistance, dyslipidemia), cardiovascular risk, fatigue, sexual dysfunction, and possible cognitive effects. These risks are managed through regular monitoring (DEXA scans, metabolic panels, cardiovascular assessment), bone-protective medications when needed, exercise programs, and cardiovascular risk factor management. The oral GnRH antagonists with add-back therapy were specifically designed to allow longer treatment durations by including low-dose hormones to protect bone and minimize menopausal symptoms.
7. Can GnRH analogs affect fertility, and is the effect reversible?
GnRH analogs suppress fertility during active treatment by shutting down gonadotropin-driven gamete production. In women, ovulation ceases. In men, spermatogenesis is severely reduced or eliminated. However, these effects are reversible in the vast majority of cases. After discontinuing GnRH agonists, hormonal function typically recovers within 1-6 months, with menstruation resuming in women and testosterone recovering in men. Spermatogenesis may take longer to fully normalize - up to 6-12 months in some cases. Interestingly, GnRH agonists can actually protect fertility when co-administered during chemotherapy, reducing the rate of premature ovarian failure in premenopausal cancer patients. The distinction between gonadorelin (which supports fertility at pulsatile doses) and long-acting GnRH agonists (which suppress it) is worth understanding.
8. What are oral GnRH antagonists, and how do they compare to injectable options?
Oral GnRH antagonists (elagolix, relugolix, and linzagolix) are non-peptide small molecules that block GnRH receptors when taken as daily pills. They offer several advantages over injectable options: no needles required, dose-dependent hormonal suppression allowing partial or complete estrogen control, rapid onset and offset of action, and the ability to combine with low-dose add-back hormones in a single tablet. This makes them especially well-suited for conditions like endometriosis and uterine fibroids, where partial estrogen suppression provides symptom relief while preserving bone health. Relugolix is also approved for prostate cancer as a daily oral alternative to injectable depot agonists. The main disadvantages are higher cost, daily dosing requirements (versus monthly to annual depot formulations), and more potential drug-drug interactions.
9. What is the testosterone flare, and why does it matter?
The testosterone flare is a temporary surge in testosterone levels (typically 50-100% above baseline) that occurs during the first 1-2 weeks of GnRH agonist treatment. It happens because the agonist initially stimulates GnRH receptors before causing their downregulation. For most patients, the flare is clinically insignificant. However, in men with advanced prostate cancer - particularly those with bone metastases near the spinal cord, high-volume skeletal disease, or obstructive uropathy - the flare can temporarily worsen symptoms and cause serious complications like spinal cord compression. This is managed by co-administering an anti-androgen (bicalutamide or flutamide) during the first 2-4 weeks or by using a GnRH antagonist that produces no flare.
10. How are GnRH analogs used in IVF treatment?
In IVF, GnRH analogs prevent premature ovulation during controlled ovarian stimulation. Two main approaches exist. The "long agonist protocol" uses a GnRH agonist (usually leuprolide) starting in the mid-luteal phase to suppress the pituitary before stimulation begins. The "antagonist protocol" adds a GnRH antagonist (cetrorelix or ganirelix) during stimulation to block premature LH surges. The antagonist protocol has become preferred in many centers because it requires fewer days of treatment, uses less medication, and significantly reduces the risk of ovarian hyperstimulation syndrome. Additionally, a single dose of GnRH agonist can serve as the ovulation trigger in antagonist protocols, nearly eliminating OHSS risk - especially valuable for PCOS patients and oocyte donors.
11. Can GnRH analogs be used for conditions other than cancer and reproductive issues?
Yes, GnRH analogs have several additional applications. They are the standard treatment for central precocious puberty, where they halt premature sexual development and protect adult height potential. They are used in gender-affirming care for transgender youth to suppress endogenous puberty. They can help manage severe premenstrual dysphoric disorder (PMDD) by eliminating hormonal cycling. They are employed for menstrual suppression before surgery to correct anemia. Research is exploring potential applications in Alzheimer's disease, male contraception, and as targeting vectors for delivering drugs to GnRH receptor-expressing tumors. The versatility of these agents stems from the central role of the HPG axis in human physiology.
12. What role does kisspeptin play in GnRH regulation?
Kisspeptin is the primary upstream activator of GnRH neurons in the hypothalamus. Produced by specialized KNDy (Kisspeptin-Neurokinin B-Dynorphin) neurons in the arcuate nucleus, kisspeptin signals GnRH neurons to fire through the KISS1R receptor. It essentially serves as the pulse generator for the entire reproductive hormone cascade. Without functional kisspeptin signaling, GnRH release fails and puberty does not occur. This makes kisspeptin a potential therapeutic target for modulating the HPG axis more physiologically than direct GnRH receptor manipulation. Ongoing research is evaluating kisspeptin for IVF triggering, treating hypothalamic amenorrhea, and as a diagnostic tool for reproductive disorders.
13. How do I know which GnRH analog is right for my situation?
The choice depends on several factors: your specific medical condition, whether hormonal flare is acceptable, how long treatment will last, your preference for injections versus oral medication versus nasal spray, insurance coverage and cost considerations, and any cardiovascular or bone health risk factors. For prostate cancer, depot agonists offer convenience while antagonists avoid flare. For endometriosis, oral antagonists with add-back therapy provide the most flexible long-term option. For IVF, antagonist protocols are now standard in most programs. For fertility preservation during TRT, gonadorelin is the primary option. Your healthcare provider can help determine the best choice based on your individual circumstances. The Free Assessment at FormBlends can also provide initial guidance.
References
- Schally AV, Arimura A, Baba Y, et al. Isolation and properties of the FSH and LH-releasing hormone. Biochem Biophys Res Commun. 1971;43(2):393-399. doi:10.1016/0006-291X(71)90766-2
- Conn PM, Crowley WF Jr. Gonadotropin-releasing hormone and its analogs. N Engl J Med. 1991;324(2):93-103. doi:10.1056/NEJM199101103240205
- Millar RP, Lu ZL, Pawson AJ, et al. Gonadotropin-releasing hormone receptors. Endocr Rev. 2004;25(2):235-275. doi:10.1210/er.2003-0002
- Herbison AE. The gonadotropin-releasing hormone pulse generator. Endocrinology. 2018;159(11):3723-3736. doi:10.1210/en.2018-00653
- Navarro VM, Tena-Sempere M. Neuroendocrine control by kisspeptins: role in metabolic regulation of fertility. Nat Rev Endocrinol. 2012;8(1):40-53. doi:10.1038/nrendo.2011.147
- Skorupskaite K, George JT, Anderson RA. The kisspeptin-GnRH pathway in human reproductive health and disease. Hum Reprod Update. 2014;20(4):485-500. doi:10.1093/humupd/dmu009
- Henzl MR, Corson SL, Moghissi K, et al. Administration of nasal nafarelin as compared with oral danazol for endometriosis. N Engl J Med. 1988;318(8):485-489. doi:10.1056/NEJM198802253180805
- Henzl MR, Kwei L. Efficacy and safety of nafarelin in the treatment of endometriosis. Am J Obstet Gynecol. 1990;162(2):570-574. doi:10.1016/0002-9378(90)90432-7
- Hornstein MD, Surrey ES, Weisberg GW, Casino LA. Leuprolide acetate depot and hormonal add-back in endometriosis: a 12-month study. Obstet Gynecol. 1998;91(1):16-24. doi:10.1016/S0029-7844(97)00579-8
- Crawford ED, Tombal B, Miller K, et al. A phase III extension trial with a 1-arm crossover from leuprolide to degarelix: comparison of gonadotropin-releasing hormone agonist and antagonist effect on prostate cancer. J Urol. 2011;186(3):889-897. doi:10.1016/j.juro.2011.04.083
- Klotz L, Nabid A, Engel J, et al. Testosterone nadir and clinical outcomes in patients with advanced prostate cancer: post hoc analysis of triptorelin pamoate Phase III studies. BJUI Compass. 2024;5(4):e318. doi:10.1002/bco2.318
- Lundstrom EA, Rencken RK, van Wyk JH, et al. Triptorelin 6-month formulation in the management of patients with locally advanced and metastatic prostate cancer. Clin Drug Investig. 2009;29(12):757-765. doi:10.2165/11319690-000000000-00000
- Shore ND, Saad F, Cookson MS, et al. Oral relugolix for androgen-deprivation therapy in advanced prostate cancer. N Engl J Med. 2020;382(23):2187-2196. doi:10.1056/NEJMoa2004325
- Tombal B, Miller K, Boccon-Gibod L, et al. Additional analysis of the secondary end point of biochemical recurrence rate in a phase 3 trial (CS21) comparing degarelix 80 mg versus leuprolide in patients with prostate cancer segmented by baseline characteristics. Eur Urol. 2010;57(5):836-842. doi:10.1016/j.eururo.2009.11.029
- Klotz L, Boccon-Gibod L, Shore ND, et al. The efficacy and safety of degarelix: a 12-month, comparative, randomized, open-label, parallel-group phase III study in patients with prostate cancer. BJU Int. 2008;102(11):1531-1538. doi:10.1111/j.1464-410X.2008.08183.x
- Al-Inany HG, Youssef MA, Ayeleke RO, et al. Gonadotrophin-releasing hormone antagonists for assisted reproductive technology. Cochrane Database Syst Rev. 2016;4:CD001750. doi:10.1002/14651858.CD001750.pub4
- Taylor HS, Giudice LC, Lessey BA, et al. Treatment of endometriosis-associated pain with elagolix, an oral GnRH antagonist. N Engl J Med. 2017;377(1):28-40. doi:10.1056/NEJMoa1700089
- Schlaff WD, Ackerman RT, Al-Hendy A, et al. Elagolix with add-back therapy for heavy menstrual bleeding associated with uterine fibroids. Obstet Gynecol. 2020;135(6):1313-1326. doi:10.1097/AOG.0000000000003836
- Donnez J, Taylor HS, Taylor RN, et al. Treatment of endometriosis-associated pain with linzagolix, an oral gonadotropin-releasing hormone-antagonist: a randomized clinical trial. Fertil Steril. 2020;114(1):44-55. doi:10.1016/j.fertnstert.2020.02.114
- Lambertini M, Moore HCF, Leonard RCF, et al. Gonadotropin-releasing hormone agonists during chemotherapy for preservation of ovarian function and fertility in premenopausal patients with early breast cancer: a systematic review and meta-analysis of individual patient-level data. J Clin Oncol. 2018;36(19):1981-1990. doi:10.1200/JCO.2018.78.0858
- Francis PA, Pagani O, Fleming GF, et al. Tailoring adjuvant endocrine therapy for premenopausal breast cancer. N Engl J Med. 2018;379(2):122-137. doi:10.1056/NEJMoa1803164
- Carel JC, Eugster EA, Rogol A, et al. Consensus statement on the use of gonadotropin-releasing hormone analogs in children. Pediatrics. 2009;123(4):e752-e762. doi:10.1542/peds.2008-1783
- Guaraldi F, Beccuti G, Gori D, Ghizzoni L. Management of endocrine disease: long-term outcomes of the treatment of central precocious puberty. Eur J Endocrinol. 2016;174(3):R79-R87. doi:10.1530/EJE-15-0590
- Hembree WC, Cohen-Kettenis PT, Gooren L, et al. Endocrine treatment of gender-dysphoric/gender-incongruent persons: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2017;102(11):3869-3903. doi:10.1210/jc.2017-01658
- de Vries ALC, Steensma TD, Doreleijers TAH, Cohen-Kettenis PT. Puberty suppression in adolescents with gender identity disorder: a prospective follow-up study. J Sex Med. 2011;8(8):2276-2283. doi:10.1111/j.1743-6109.2010.01943.x
- Hussain M, Tangen CM, Berry DL, et al. Intermittent versus continuous androgen deprivation in prostate cancer. N Engl J Med. 2013;368(14):1314-1325. doi:10.1056/NEJMoa1212299
- Nguyen PL, Alibhai SMH, Basaria S, et al. Adverse effects of androgen deprivation therapy and strategies to mitigate them. Eur Urol. 2015;67(5):825-836. doi:10.1016/j.eururo.2014.07.010
- Levine GN, D'Amico AV, Berger P, et al. Androgen-deprivation therapy in prostate cancer and cardiovascular risk. Circulation. 2010;121(6):833-840. doi:10.1161/CIRCULATIONAHA.109.192695
- Periti P, Mazzei T, Mini E. Clinical pharmacokinetics of depot leuprorelin. Clin Pharmacokinet. 2002;41(7):485-504. doi:10.2165/00003088-200241070-00003
- Engel JB, Schally AV. Drug Insight: clinical use of agonists and antagonists of luteinizing-hormone-releasing hormone. Nat Clin Pract Endocrinol Metab. 2007;3(2):157-167. doi:10.1038/ncpendmet0399
- Padula AM. GnRH analogues: agonists and antagonists. Anim Reprod Sci. 2005;88(1-2):115-126. doi:10.1016/j.anireprosci.2005.05.005
- Sciarra A, Busetto GM, Salciccia S, et al. New perspective in the management of neuroendocrine differentiation in prostate cancer with GnRH analogs. Curr Drug Targets. 2019;20(2):195-204. doi:10.2174/1389450119666180924155533
- van Casteren NJ, Boellaard WPA, Dohle GR, et al. Manual versus automated semen analysis in the assessment of male reproductive toxicity after triptorelin treatment: a randomized controlled trial. Int J Mol Sci. 2025;26(14):6566. doi:10.3390/ijms26146566
- Barra F, Laganà AS, Scala C, et al. Review of pharmacotherapy options for endometriosis-associated pain. Expert Opin Pharmacother. 2021;22(16):2157-2167. doi:10.1080/14656566.2021.1941865
- Yang J, Zhang L, Xiao Y, et al. GnRH agonist versus GnRH antagonist in IVF/ICSI: a retrospective cohort study. Ann Med. 2025;57(1):2564279. doi:10.1080/07853890.2025.2564279
- Simon JA, Al-Hendy A, Engel JB, et al. Oral gonadotropin-releasing hormone antagonists in the treatment of endometriosis: advances in research. J Clin Med Res. 2025;17(6):352-368. doi:10.14740/jocmr6236
- Cass H. Independent review of gender identity services for children and young people: final report. NHS England. 2024. Available at: https://cass.independent-review.uk/home/publications/final-report/
- Bangalore Krishna K, Fuqua JS, Rogol AD, et al. Use of gonadotropin-releasing hormone agonists in transgender and gender diverse youth: a systematic review. Front Endocrinol. 2025;16:1555186. doi:10.3389/fendo.2025.1555186

Figure 9: Summary of key evidence supporting GnRH analog use across clinical applications, highlighting data from the most influential clinical trials discussed in this report.
Figure 10: Clinical decision algorithm for GnRH analog selection based on patient condition, flare tolerance, administration preferences, and individualized risk factors.
Molecular Pharmacology: A Deeper Look at Structure-Activity Relationships
The Native GnRH Decapeptide: Why Every Amino Acid Matters
Understanding why GnRH analogs work the way they do requires a closer look at the parent molecule. The native GnRH decapeptide (pyroGlu-His-Trp-Ser-Tyr-Gly-Leu-Arg-Pro-Gly-NH2) has been extensively studied through systematic amino acid substitution experiments that have revealed which residues are essential for receptor binding, which contribute to biological activity, and which are vulnerable to enzymatic cleavage.
The ten amino acids in the GnRH sequence serve different functional roles. Positions 1-3 (pyroGlu-His-Trp) form the receptor binding domain. These residues make critical contacts with the extracellular loops and transmembrane helices of the GnRH receptor. Modifications at these positions typically reduce or abolish receptor binding affinity. The pyroglutamate at position 1 protects the N-terminus from aminopeptidase degradation, while histidine-2 and tryptophan-3 participate in aromatic stacking interactions with receptor residues.
Position 4 (Ser) contributes to the peptide's beta-turn conformation. Serine-4 can be replaced with certain other amino acids without catastrophic loss of activity, but changes here affect the peptide's three-dimensional folding and can alter receptor selectivity.
Position 5 (Tyr) participates in both receptor binding and signal transduction. Tyrosine-5's hydroxyl group forms a hydrogen bond with a key receptor residue, and modifications at this position affect whether the bound peptide acts as an agonist or antagonist.
Position 6 (Gly) is the most pharmacologically important position for analog design. Glycine-6 is the primary target for endopeptidase cleavage between positions 5 and 6, making it the main metabolic vulnerability in the native sequence. Replacing glycine-6 with a D-amino acid (the hallmark modification in virtually all clinically used GnRH agonists) accomplishes two things simultaneously. First, the D-configuration is not recognized by the endopeptidase, dramatically increasing metabolic stability and extending the peptide's half-life from minutes to hours. Second, D-amino acids at position 6 tend to stabilize the bioactive beta-turn conformation of the peptide, increasing receptor binding affinity. This is why a single amino acid change at position 6 can increase potency 15- to 200-fold while also extending duration of action.
The specific D-amino acid used at position 6 determines the potency and pharmacokinetic profile of each agonist analog. D-Leucine (leuprolide) provides moderate potency enhancement. D-Tryptophan (triptorelin) and D-Serine(tBu) (goserelin) provide roughly 100-fold potency increases. D-2-Naphthylalanine (nafarelin) achieves approximately 200-fold potency, making it the most potent position-6-modified agonist in clinical use. D-Histidine(Bzl) (histrelin) provides approximately 100-fold enhancement.
Position 7 (Leu) and position 8 (Arg) contribute to receptor binding through hydrophobic contacts and charge interactions, respectively. These positions are modified in some GnRH antagonists to introduce bulky, hydrophobic residues that maintain receptor binding while preventing receptor activation.
Positions 9-10 (Pro-Gly-NH2) form the C-terminal domain. The proline-9 introduces a rigid kink in the peptide backbone that is important for bioactive conformation. The C-terminal amide (replacing the free carboxyl group) is essential for full biological activity. Replacing the glycine-10 amide with an ethylamide group (as in some antagonist analogs) can modify receptor binding properties.
From Agonist to Antagonist: The Design Challenge
Creating GnRH antagonists was considerably more difficult than creating agonists. While agonists required only the position-6 D-amino acid substitution to achieve clinically useful compounds, antagonists needed multiple simultaneous modifications to maintain receptor binding while preventing receptor activation.
The first-generation GnRH antagonists (developed in the 1970s-1980s) required substitutions at positions 1, 2, 3, 6, and 10 of the native sequence. Many of these early compounds caused unacceptable histamine release from mast cells due to their net positive charge and structural similarity to substance P and other mast cell-activating peptides. This manifested clinically as injection site reactions, urticaria, and rarely anaphylaxis.
The design challenge was to maintain sufficient modifications for antagonist activity while minimizing the structural features responsible for histamine release. This led to the development of second-generation antagonists with neutral or acidic amino acid substitutions that reduced the peptide's net positive charge.
Cetrorelix (Ac-D-Nal(2)-D-Phe(4Cl)-D-Pal(3)-Ser-Tyr-D-Cit-Leu-Arg-Pro-D-Ala-NH2) represents one successful solution. The acetyl group at the N-terminus and D-amino acids at positions 1, 2, 3, and 6 provide antagonist activity, while the D-citrulline at position 6 (an uncharged, bulky residue) helps reduce histamine release compared to earlier compounds.
Degarelix pushed the design further, incorporating a unique pair of modifications. The acetyl-D-2-naphthylalanine at position 1 and D-4-chlorophenylalanine at position 2, combined with a novel N-(2-aminoethyl)carbamoyl linkage at the C-terminus, produced a compound with minimal histamine release potential while maintaining high-affinity receptor binding and effective antagonist activity. Its long half-life (29-43 days) results from its tendency to form a gel-like depot at the injection site, providing sustained release from a single subcutaneous injection.
Non-Peptide GnRH Antagonists: A Different Approach Entirely
The oral GnRH antagonists (elagolix, relugolix, linzagolix) are not modified peptides at all. They are small-molecule, non-peptide compounds identified through high-throughput screening campaigns and subsequent medicinal chemistry optimization. These molecules bind to the same GnRH receptor as the native peptide but do so through a different binding mode, accessing the receptor's transmembrane domain rather than the extracellular binding pocket used by peptide ligands.
This non-peptide nature confers several pharmacological advantages. Oral bioavailability is achievable (peptides are degraded in the gastrointestinal tract). Half-lives are measured in hours rather than minutes (for native GnRH) or days (for depot formulations), allowing daily dosing with rapid offset after discontinuation. The compounds are not substrates for peptidases, eliminating the enzymatic degradation that limits native GnRH's duration of action. They do not cause histamine release because they lack the structural features of peptide antagonists that activate mast cells.
The trade-off is that non-peptide antagonists are subject to hepatic CYP450 metabolism and P-glycoprotein transport, creating drug-drug interaction potential that peptide analogs largely avoid. Elagolix is metabolized primarily by CYP3A4, making it susceptible to interactions with CYP3A4 inhibitors and inducers. Relugolix is a substrate of P-glycoprotein, and co-administration with P-gp inducers (like rifampin) can reduce its plasma levels enough to cause testosterone escape in prostate cancer patients - a potentially dangerous clinical consequence.
Depot Formulation Technology
The development of depot formulations has been essential to the clinical success of GnRH agonists, converting compounds with half-lives of a few hours into treatments that last one to twelve months from a single injection. Several technologies are used.
PLGA microspheres: Poly(lactic-co-glycolic acid) microspheres are the most widely used depot technology for leuprolide and triptorelin. The peptide is encapsulated within biodegradable polymer microspheres that are suspended in a diluent and injected intramuscularly. After injection, the microspheres slowly degrade through hydrolysis, releasing the peptide over weeks to months. The degradation rate (and therefore the release profile) is controlled by the lactide-to-glycolide ratio, molecular weight, and end-group chemistry of the polymer.
In situ forming gel (Eligard): Leuprolide's Eligard formulation uses a different approach. The peptide is dissolved in a biocompatible organic solvent (N-methyl-2-pyrrolidone) along with PLGA polymer. After subcutaneous injection, the organic solvent dissipates into surrounding tissue, causing the polymer to precipitate and form a solid depot that encapsulates the peptide. This in situ gel formation provides sustained release for 1-6 months depending on the polymer composition.
Solid implant (Zoladex, Supprelin LA, Vantas): Goserelin's Zoladex implant is a small, pre-formed solid cylinder that is inserted subcutaneously. The goserelin peptide is dispersed throughout a biodegradable D,L-lactide-glycolide copolymer matrix. Histrelin's implants use a different approach - the peptide is contained within a small, flexible hydrogel rod that releases drug through diffusion-controlled mechanisms over 12 months.
Each technology has specific advantages and limitations. Microspheres require reconstitution before injection, adding complexity. In situ forming gels can cause injection site discomfort as the organic solvent dissipates. Solid implants require a minor procedure for both insertion and removal. The choice of delivery system affects burst release characteristics (the initial spike in drug levels from surface-bound peptide), steady-state plasma levels, and end-of-dose trough behavior (whether drug levels remain therapeutic throughout the dosing interval).
Historical Development of GnRH Analog Therapeutics
Discovery of GnRH: The 1977 Nobel Prize
The story of GnRH analogs begins with the isolation and characterization of the native hormone. In 1971, Andrew Schally and Roger Guillemin independently isolated GnRH from hypothalamic extracts - Schally from porcine hypothalami, Guillemin from ovine tissue. Both determined it to be a decapeptide, and its amino acid sequence was published shortly afterward. This discovery earned both scientists (along with Rosalyn Yalow for radioimmunoassay development) the 1977 Nobel Prize in Physiology or Medicine.
The identification of GnRH's structure immediately suggested therapeutic possibilities. If the body's reproductive system was controlled by a single small peptide, then synthetic versions of that peptide could potentially treat any condition involving reproductive hormone dysregulation. The race to create clinically useful analogs began within months of the structure's publication.
The Agonist Era: 1970s-1990s
The first GnRH agonist analogs were synthesized in the early 1970s using the D-amino acid substitution strategy at position 6. The key insight was that these super-potent agonists, while initially stimulating gonadotropin release, would paradoxically suppress the reproductive axis when given continuously. This "medical castration" effect was first demonstrated in animal studies by Schally's group and quickly confirmed in human clinical trials.
Leuprolide was among the first GnRH agonists to reach clinical development, receiving FDA approval for prostate cancer in 1985 (Lupron Depot). This marked the beginning of a new era in prostate cancer treatment, offering an alternative to surgical castration (orchiectomy) that was reversible, less psychologically traumatic, and equally effective.
Throughout the 1980s and 1990s, additional GnRH agonists entered the market. Goserelin (Zoladex, approved 1989), nafarelin (Synarel, approved 1990), and triptorelin (Trelstar, approved 2000 in the US) expanded the available options. Each offered somewhat different pharmacological profiles, routes of administration, and formulation conveniences, but all shared the fundamental agonist mechanism of initial stimulation followed by receptor downregulation.
The development of depot formulations was a critical milestone. Early GnRH agonists required daily injections, limiting patient compliance and clinical utility. The introduction of monthly depot formulations (Lupron Depot, 1989) and subsequent 3-month, 6-month, and 12-month formulations transformed GnRH agonist therapy from a daily burden into an infrequent clinic visit.
The Antagonist Challenge: 1980s-2000s
GnRH antagonist development proved far more challenging than agonist development. While the therapeutic rationale was clear (immediate suppression without flare would be clinically advantageous), the molecular design requirements were demanding. Early antagonist candidates caused excessive histamine release, had poor solubility, required large injection volumes, and had inconsistent pharmacokinetics.
The first clinically viable GnRH antagonist, cetrorelix, received FDA approval in 1999 for IVF use. Ganirelix followed in the same year. These agents filled an important niche in reproductive medicine by preventing premature LH surges during controlled ovarian stimulation without the prolonged pituitary downregulation required by agonist protocols.
Abarelix (Plenaxis) became the first GnRH antagonist approved for prostate cancer in 2003, but histamine-related adverse events and supply issues led to its market withdrawal in 2005. Degarelix (Firmagon), approved in 2008, succeeded where abarelix had struggled, offering effective prostate cancer treatment with an acceptable safety profile.
The Oral Revolution: 2010s-Present
The development of orally bioavailable, non-peptide GnRH antagonists represented a paradigm change in the field. These small molecules were identified through high-throughput screening of chemical libraries against the GnRH receptor, followed by extensive medicinal chemistry optimization.
Elagolix was the first to reach the market (FDA approval 2018 for endometriosis), followed by relugolix (Orgovyx for prostate cancer, 2020; Myfembree for fibroids and endometriosis, 2021). Linzagolix received European approval for fibroids in 2022 and remains under FDA review for the US market.
These oral agents have expanded the patient population that can benefit from GnRH-based therapy by removing the barrier of injectable administration. They have also introduced the concept of dose-dependent estrogen modulation - the ability to partially suppress estrogen to levels that relieve symptoms while maintaining enough estrogenic activity to protect bone and minimize menopausal side effects. This "threshold" approach, combined with add-back therapy, enables long-term treatment durations that were impractical with injectable GnRH agonists due to bone density concerns.
Clinical Scenarios and Decision-Making
To illustrate how GnRH analog selection works in practice, the following clinical scenarios demonstrate the decision-making process. These are simplified examples for educational purposes; real clinical decisions involve many additional patient-specific factors.
Scenario 1: A 68-Year-Old Man with Newly Diagnosed Metastatic Prostate Cancer
Clinical situation: A 68-year-old man presents with a PSA of 85 ng/mL, Gleason 4+4=8 prostate adenocarcinoma, and bone scan showing multiple skeletal metastases including L3 vertebral body involvement (no spinal cord compression currently). He has a history of coronary artery disease with prior stent placement.
Key considerations:
- Vertebral metastasis creates risk of spinal cord compression during testosterone flare
- Cardiovascular disease history raises concern about ADT-related cardiovascular risk
- Long-term (indefinite) ADT is indicated for metastatic disease
- Current guidelines recommend combination therapy (ADT plus novel hormonal agent or chemotherapy)
GnRH analog selection rationale:
- Option A - Degarelix: No flare (protects against spinal cord compression), potential cardiovascular benefit based on CS21 post-hoc analysis. However, monthly injections with 40% injection site reaction rate limit long-term acceptability.
- Option B - Relugolix (oral): No flare, 54% lower cardiovascular event rate versus leuprolide in the HERO trial. Daily oral dosing offers convenience but requires strict adherence. P-glycoprotein interactions need review given likely cardiac medications.
- Option C - Degarelix loading followed by agonist depot: Use degarelix for the first month to avoid flare, then transition to triptorelin or leuprolide 3- or 6-month depot for maintenance. This "switch" strategy is increasingly popular, combining flare avoidance with depot convenience.
Likely recommendation: Given the cardiovascular history and vertebral metastasis, relugolix (oral) would be a strong first choice if drug interactions are acceptable. Degarelix followed by depot agonist switch would be an alternative if oral adherence is a concern. In either case, ADT would be combined with a next-generation hormonal agent (abiraterone, enzalutamide, darolutamide, or apalutamide) per current NCCN guidelines.
Scenario 2: A 32-Year-Old Woman with Severe Endometriosis-Associated Pain
Clinical situation: A 32-year-old woman with laparoscopically confirmed stage III endometriosis presents with debilitating pelvic pain and dysmenorrhea despite 18 months of combined oral contraceptive therapy and naproxen. She desires future fertility (not currently) and is concerned about the effects of surgery on her ovarian reserve.
Key considerations:
- First-line hormonal therapy has failed
- Fertility preservation is important but not immediately needed
- She needs effective long-term pain management
- Bone health must be protected given young age
GnRH analog selection rationale:
- Option A - Elagolix 150 mg daily: Partial estrogen suppression provides meaningful pain relief while maintaining estrogen levels sufficient to protect bone. FDA-approved for up to 24 months. Oral administration, no injections. Can be discontinued quickly when fertility is desired.
- Option B - Relugolix combination (Myfembree): Full-dose GnRH antagonist with built-in add-back therapy (estradiol + norethindrone acetate). Provides more complete pain suppression than low-dose elagolix while protecting bone through add-back hormones.
- Option C - Leuprolide depot 3.75 mg monthly with add-back: Injectable GnRH agonist with progestin add-back. Effective but involves monthly injections, has an initial flare period, and is typically limited to 6-12 months of use.
Likely recommendation: Either elagolix 150 mg daily or relugolix combination (Myfembree) would be reasonable first choices. The oral route, absence of flare, and dose adjustability make oral antagonists preferable to injectable agonists for this patient. If she plans pregnancy within 6-12 months, the rapid reversibility of oral antagonists is advantageous. Baseline DEXA scan would be prudent given anticipated treatment duration.
Scenario 3: A 7-Year-Old Girl with Central Precocious Puberty
Clinical situation: A 7-year-old girl presents with breast development (Tanner stage 2-3), pubic hair, and accelerated growth velocity. Her bone age is 9.5 years (chronological age 7.2 years). Stimulated LH after gonadorelin injection is 12 IU/L, confirming central activation of the HPG axis. Brain MRI shows no intracranial pathology.
Key considerations:
- Treatment will continue for several years (until approximately age 11-12)
- Needle tolerance varies in this age group
- Adherence with daily medications may be challenging
- Family lives 2 hours from the nearest pediatric endocrinology clinic
GnRH analog selection rationale:
- Option A - Histrelin implant (Supprelin LA): Annual implant placement provides the longest dosing interval. Eliminates daily medication compliance issues. Requires a minor procedure for insertion and removal. Ideal for families with travel barriers to frequent clinic visits.
- Option B - Leuprolide depot (Lupron Depot-Ped) 11.25 mg every 3 months: Quarterly injections balance frequency with depot convenience. Well-established pediatric dosing and safety data.
- Option C - Nafarelin nasal spray: Non-invasive, needle-free administration. However, twice-daily dosing requires consistent compliance from a 7-year-old, which can be challenging.
Likely recommendation: The histrelin implant (Supprelin LA) would be an excellent choice given the family's distance from the clinic, the expected multi-year treatment duration, and the elimination of compliance concerns. If the child and family prefer to avoid an implant procedure, quarterly leuprolide depot would be the next best option. Nafarelin would be reserved for families confident in maintaining twice-daily nasal dosing adherence.
Scenario 4: A Man on TRT Wanting to Preserve Fertility
Clinical situation: A 35-year-old man has been on testosterone cypionate 200 mg every two weeks for hypogonadism diagnosed 2 years ago. He and his wife plan to conceive within the next 12-18 months. His most recent semen analysis shows oligospermia (5 million sperm/mL, down from 45 million pre-TRT). He does not want to discontinue testosterone entirely due to significant improvement in energy, mood, and quality of life.
Key considerations:
- Active spermatogenesis suppression from exogenous testosterone
- Patient strongly prefers to continue TRT
- Fertility timeline is 12-18 months
- hCG is unavailable from his compounding pharmacy
GnRH analog selection rationale:
- Option A - Add gonadorelin 200 mcg SC three times weekly: Stimulates pulsatile LH and FSH release to support testicular function alongside continued TRT. Less effective than hCG for this purpose but may partially restore spermatogenesis.
- Option B - Discontinue TRT and use gonadorelin during recovery: Allows the HPG axis to recover fully, with gonadorelin supporting the transition. Expect 3-6 months for spermatogenesis to normalize.
- Option C - Reduce TRT dose and add gonadorelin: Lower testosterone dose reduces HPG axis suppression while gonadorelin provides additional gonadotropin stimulation. A compromise approach.
Likely recommendation: For the best chance of conception, temporarily discontinuing or substantially reducing TRT while using gonadorelin and/or clomiphene to accelerate HPG axis recovery would be ideal. If the patient insists on continuing full-dose TRT, adding gonadorelin may help but outcomes are uncertain. A repeat semen analysis 3-4 months after starting gonadorelin will indicate whether the approach is working. If fertility is the priority, our Gonadorelin Research Report provides additional protocol details.
Pharmacovigilance and Post-Marketing Safety Data
Cardiovascular Safety: The Ongoing Debate
The relationship between GnRH analogs and cardiovascular risk has been one of the most actively debated topics in prostate cancer treatment over the past two decades. The concern first emerged from retrospective analyses suggesting that ADT with GnRH agonists was associated with increased rates of myocardial infarction, stroke, sudden cardiac death, and diabetes.
An FDA safety communication in 2010, prompted by these observational studies, required updated labeling for all GnRH agonists to include warnings about increased risk of diabetes, heart attack, stroke, and sudden death. However, the quality of evidence supporting these associations was initially low, based primarily on retrospective cohort studies and database analyses with significant confounding variables.
Subsequent prospective data has provided more nuanced understanding. The cardiovascular risk associated with ADT appears to be modest in absolute terms, concentrated in the first 6-12 months of therapy, and primarily affecting men with pre-existing cardiovascular disease. The mechanisms likely involve ADT-induced metabolic changes (insulin resistance, dyslipidemia, visceral adiposity, increased fat mass) and possibly direct vascular effects of testosterone deprivation.
The HERO trial's finding of 54% lower major adverse cardiovascular events (MACE) with relugolix compared to leuprolide was provocative but requires cautious interpretation. The MACE rate difference (2.9% relugolix vs. 6.2% leuprolide) was a secondary endpoint in a trial not powered for cardiovascular outcomes, and the leuprolide arm had a higher baseline cardiovascular risk profile. Nonetheless, this finding, combined with mechanistic data suggesting that GnRH agonists may have direct adverse effects on the cardiovascular system through GnRH receptors expressed on cardiomyocytes and vascular endothelial cells (effects that antagonists would not share), has generated substantial interest in the cardiovascular advantages of antagonist-based ADT.
A dedicated, prospective, randomized cardiovascular outcomes trial comparing GnRH agonists and antagonists would definitively resolve this question, but no such trial has been completed. In the meantime, current consensus recommends cardiovascular risk assessment before initiating any form of ADT, optimization of modifiable risk factors (hypertension, dyslipidemia, diabetes, smoking, obesity), regular monitoring during therapy, and consideration of GnRH antagonists for patients with significant pre-existing cardiovascular disease.
Bone Health: Quantifying the Risk
The skeletal effects of GnRH-mediated sex steroid deprivation are well-characterized. In men receiving ADT for prostate cancer, bone mineral density decreases at a rate of approximately 2-4% per year at the lumbar spine and 1-3% per year at the femoral neck during the first 2-3 years of treatment. The rate of loss may slow somewhat with longer treatment duration as a new, lower steady state is approached.
The clinical consequence is a significantly elevated fracture risk. A large population-based study found that men receiving GnRH agonist therapy for prostate cancer had a 21% increase in fracture risk compared to age-matched controls not receiving ADT. The risk was cumulative, increasing with longer treatment duration.
In women receiving GnRH agonists for endometriosis, bone density loss is similar in magnitude (2-8% at the lumbar spine over 6 months) but is generally more reversible because treatment durations are shorter. Studies with 12-24 months of post-treatment follow-up have shown substantial recovery of lost bone density, though complete restoration to baseline may not occur in all patients.
Bone-protective strategies during GnRH analog therapy include weight-bearing and resistance exercise, calcium supplementation (1000-1200 mg daily from diet and supplements combined), vitamin D supplementation (800-2000 IU daily, targeting serum 25-OH vitamin D levels of 30+ ng/mL), bisphosphonate therapy (zoledronic acid 4 mg IV annually, or oral alendronate 70 mg weekly) for men on prolonged ADT, denosumab (60 mg SC every 6 months) as an alternative to bisphosphonates, and add-back hormone therapy (for women on GnRH analogs for benign conditions).
Rare but Serious Adverse Events
Several rare adverse events have been identified through post-marketing surveillance and deserve mention:
Pituitary apoplexy: Very rare cases of pituitary hemorrhage or infarction have been reported after the first dose of GnRH agonist, typically in patients with undiagnosed pituitary adenomas. The mechanism is thought to involve rapid swelling of gonadotrope adenoma cells in response to the initial GnRH agonist stimulation, causing hemorrhage within the confined space of the sella turcica. This presents with sudden severe headache, visual disturbances, and altered consciousness - a medical emergency requiring urgent neurosurgical evaluation.
QT prolongation: ADT has been associated with mild QT interval prolongation on electrocardiography. While clinically significant arrhythmias attributable to this effect are extremely rare, caution is advised when combining GnRH analogs with other QT-prolonging medications (certain antibiotics, antipsychotics, antiarrhythmics).
Anaphylaxis: Hypersensitivity reactions to GnRH analogs are rare but have been reported with both agonist and antagonist peptides. Patients should be observed for at least 30 minutes after the first injection of any GnRH analog. Subsequent injections can typically be administered without observation if the first dose was tolerated.
Interstitial lung disease: Extremely rare cases of interstitial pneumonitis have been reported with leuprolide and possibly other GnRH agonists. The mechanism is unclear and may represent an idiosyncratic hypersensitivity reaction. Patients presenting with new dyspnea and cough during GnRH agonist therapy should be evaluated for this possibility.
Hepatotoxicity with elagolix: Elagolix carries a specific warning for liver injury, with cases of elevated transaminases (ALT/AST greater than 3 times the upper limit of normal) reported in clinical trials. Liver function testing is recommended before starting elagolix and periodically during treatment. The drug is contraindicated in patients with severe hepatic impairment (Child-Pugh C).
IVF Protocols: Detailed Protocol Comparisons and Optimization
The Long GnRH Agonist Protocol: Step-by-Step
The long GnRH agonist protocol remains the most extensively studied IVF stimulation protocol, with decades of accumulated clinical data supporting its use. Understanding its implementation in detail helps contextualize why it was the dominant protocol for so long and why the antagonist protocol has progressively replaced it in many settings.
Cycle Day 21 (Mid-Luteal Phase Start): The protocol begins in the luteal phase of the menstrual cycle preceding the planned stimulation cycle. Leuprolide acetate is typically initiated at 0.5 mg or 1.0 mg subcutaneous injection daily. Some protocols use nafarelin nasal spray (400-600 mcg daily) as an alternative. The timing exploits the natural luteal phase progesterone elevation, which cooperates with the GnRH agonist to suppress the hypothalamic-pituitary axis.
Days 1-14 of Agonist (Downregulation Phase): During the first 3-5 days of GnRH agonist administration, the initial flare occurs - FSH and LH levels rise, and estradiol may increase transiently. By days 7-10, receptor downregulation begins, and gonadotropin levels start declining. By day 14, most patients achieve a state of pituitary suppression characterized by estradiol levels below 50 pg/mL, LH levels below 5 IU/L, and no ovarian follicles greater than 10 mm on ultrasound. This is confirmed with a baseline ultrasound and blood work before proceeding to stimulation.
Stimulation Phase (10-14 Days): Once pituitary suppression is confirmed, gonadotropin stimulation begins. Recombinant FSH (follitropin alfa or beta, typically 150-300 IU daily) is administered subcutaneously. Some protocols add recombinant LH or hMG (human menopausal gonadotropin) to provide LH activity, particularly in patients who may be "over-suppressed" by the GnRH agonist. The leuprolide dose is typically reduced to 0.25 mg daily during stimulation to maintain suppression without excessive LH deprivation.
Monitoring during stimulation involves transvaginal ultrasound every 2-3 days to assess follicular growth and endometrial thickness, along with serum estradiol measurements to track follicular steroidogenic activity. The starting gonadotropin dose is adjusted based on follicular response - increased if response is slow, decreased if too rapid.
Trigger and Retrieval: When at least 2-3 follicles reach 17-18 mm diameter, final oocyte maturation is triggered with hCG injection (5,000-10,000 IU intramuscular, or 250 mcg recombinant hCG subcutaneous). Oocyte retrieval is performed 34-36 hours after the trigger. In long agonist protocols, a GnRH agonist trigger cannot be used because the pituitary is already desensitized and cannot produce the required LH surge.
The GnRH Antagonist Protocol: Step-by-Step
The antagonist protocol has become the standard approach in many IVF centers due to its shorter duration, lower medication burden, reduced OHSS risk, and comparable pregnancy outcomes. Two variations exist: the fixed protocol and the flexible protocol.
Fixed Protocol:
Cycle Day 2-3 (Stimulation Start): Gonadotropin stimulation begins directly in the early follicular phase without prior pituitary suppression. Starting doses are similar to the long protocol (FSH 150-300 IU daily). No GnRH analog is given at this point.
Stimulation Day 6 (Antagonist Start - Fixed): Regardless of follicular size, a GnRH antagonist (cetrorelix 0.25 mg or ganirelix 0.25 mg subcutaneous daily) is started on stimulation day 6. This timing is based on population pharmacodynamic data showing that premature LH surges rarely occur before stimulation day 6 in most patients.
Continued Daily Antagonist: The antagonist is administered daily until the trigger day (inclusive). Each injection provides suppression for approximately 24 hours, so consistent daily timing is important.
Flexible Protocol:
The flexible protocol delays antagonist initiation until the lead follicle reaches 14 mm diameter on ultrasound monitoring. This approach uses fewer antagonist injections (typically 3-5 versus 5-7 in the fixed protocol) and may result in a more diverse follicular cohort. The flexible protocol requires more frequent monitoring to identify the appropriate start point.
Trigger Options in Antagonist Protocols:
This is where antagonist protocols offer a unique advantage. Three trigger options are available:
- hCG trigger (standard): Same as in agonist protocols. Provides strong luteal support but carries OHSS risk in high-responders.
- GnRH agonist trigger (leuprolide 1-4 mg or triptorelin 0.1-0.2 mg): Because the pituitary is not desensitized (only blocked by the antagonist), a single dose of GnRH agonist can "break through" the antagonist blockade and induce an endogenous LH/FSH surge. This surge is shorter-lived than hCG-induced stimulation, nearly eliminating OHSS risk. However, the brief luteal support requires aggressive supplementation (progesterone + estradiol) if a fresh embryo transfer is planned, or the cycle can be converted to a freeze-all strategy.
- Dual trigger (low-dose hCG + GnRH agonist): Combines the OHSS risk reduction of the agonist trigger with improved luteal support from low-dose hCG (1,000-1,500 IU). This approach has gained popularity as a middle ground for patients at moderate OHSS risk who prefer fresh embryo transfer.
OHSS Prevention: The GnRH Agonist Trigger Advantage
Ovarian hyperstimulation syndrome remains the most serious iatrogenic complication of IVF. Severe OHSS, characterized by massive ovarian enlargement, ascites, pleural effusion, hemoconcentration, and thromboembolic events, occurs in approximately 1-5% of IVF cycles using conventional hCG triggers. It can be life-threatening and occasionally fatal.
The GnRH agonist trigger in antagonist protocols has dramatically reduced OHSS incidence. The mechanism involves the characteristics of the endogenous LH surge induced by the agonist. The LH surge lasts only 24-36 hours (versus 6-7 days of hCG activity), limiting the post-trigger ovarian stimulation that drives OHSS pathophysiology. The FSH component of the agonist-triggered surge may actually benefit oocyte maturation by mimicking the natural mid-cycle FSH peak. The rapid clearance of the LH surge allows the corpus luteum to involute quickly, reducing vascular endothelial growth factor (VEGF) production - the key mediator of OHSS-related vascular permeability.
Clinical studies have shown that the GnRH agonist trigger reduces the incidence of moderate-to-severe OHSS from 5-8% (with hCG trigger) to less than 1%, with virtual elimination of severe OHSS when combined with a freeze-all strategy. This has been transformative for high-risk patients, particularly women with PCOS who produce large numbers of follicles during stimulation.
Special IVF Populations and GnRH Analog Considerations
Poor Ovarian Responders
Women with diminished ovarian reserve (low AMH, high FSH, low antral follicle count) often respond poorly to standard IVF stimulation. For these patients, the choice of GnRH analog protocol has important implications. GnRH antagonist protocols are generally preferred because they avoid the prolonged pituitary suppression of the long agonist protocol. This matters because poor responders have already diminished follicular pools, and the deeper suppression of the long protocol can further reduce the number of recruitable follicles. The antagonist protocol preserves endogenous FSH contribution during the early stimulation phase, potentially improving follicular recruitment. Shorter overall treatment time reduces the psychological and financial burden for patients who may need multiple IVF cycles.
Some clinicians use a "mini-stimulation" or "mild IVF" approach for poor responders, using minimal gonadotropin doses with an antagonist protocol and accepting fewer oocytes in exchange for potentially higher oocyte quality and lower treatment burden.
PCOS and High Responders
Women with polycystic ovary syndrome represent the highest-risk group for OHSS and benefit most from the GnRH antagonist protocol with agonist trigger strategy. The typical approach involves the antagonist protocol with low-starting-dose gonadotropins (75-150 IU FSH daily), careful monitoring with a willingness to coast or cancel if response is excessive, GnRH agonist trigger (eliminating OHSS risk), freeze-all cycle with segmented embryo transfer in a subsequent natural or hormone-replacement cycle, and in some programs, in vitro maturation (IVM) of immature oocytes as an alternative to conventional stimulation.
A Cochrane review specifically examining antagonist protocols in PCOS patients confirmed that GnRH antagonist protocols produced similar pregnancy rates to long agonist protocols while significantly reducing OHSS incidence, supporting the antagonist protocol as the preferred approach for this population.
Oocyte Donors
Oocyte donors are young, healthy women at increased OHSS risk (particularly due to high ovarian reserve and aggressive stimulation to maximize oocyte yield). GnRH antagonist protocols with agonist trigger are now standard for oocyte donation in most programs, as the donor's luteal phase is irrelevant (she is not carrying the pregnancy). This allows aggressive stimulation with maximal safety through the agonist trigger approach.
Fertility Preservation for Cancer Patients
Cancer patients requiring urgent fertility preservation before chemotherapy face unique timing pressures. GnRH antagonist protocols offer critical advantages in this setting because they can be initiated at any point in the menstrual cycle ("random-start" protocol), rather than waiting for the early follicular phase. This reduces the delay to stimulation start from potentially 2-4 weeks (waiting for the next cycle) to 1-2 days. Time is often of the essence, as oncologists want to begin chemotherapy as quickly as possible.
The random-start antagonist protocol involves beginning gonadotropin stimulation immediately regardless of cycle day, adding GnRH antagonist when follicles reach 13-14 mm, and proceeding to trigger and retrieval when follicles are mature. Studies have shown that random-start antagonist protocols produce comparable oocyte yields and fertilization rates to conventional early-follicular-start protocols, validating this approach for urgent fertility preservation.
Economic Impact and Access to GnRH Analog Therapy
Global Market Overview
The global GnRH analog market represents a substantial segment of the pharmaceutical industry. The market encompasses both well-established generic agents (leuprolide, goserelin) and newer branded products (elagolix, relugolix, linzagolix), with the total market value estimated at over $6 billion annually as of 2025.
Market dynamics are shaped by several factors. Patent expirations on older GnRH agonists have introduced generic competition, reducing costs for these established agents. Leuprolide depot generics are now available in many markets, and generic goserelin has been approved in the US and Europe. Triptorelin generics are available in some countries but not yet in the US.
The oral GnRH antagonists, protected by patents through the early-to-mid 2030s, command premium pricing. In the US market, elagolix (Orilissa) lists at approximately $850-900 per month, relugolix (Orgovyx) at approximately $2,300 per month, and Myfembree at approximately $1,000 per month. Manufacturer copay assistance programs, specialty pharmacy discounts, and insurance negotiated rates substantially reduce patient out-of-pocket costs in many cases, but access disparities remain significant.
Insurance Coverage Patterns
Insurance coverage for GnRH analogs varies considerably by indication, payer, and geographic region.
Prostate cancer ADT with GnRH agonists is typically covered by all major insurance plans under oncology benefits, with established prior authorization pathways. GnRH antagonists for prostate cancer (degarelix, relugolix) face more variable coverage, with some payers requiring documentation of cardiovascular risk factors or GnRH agonist contraindications before authorizing antagonist therapy.
Coverage for endometriosis and fibroid indications has improved with the approval of oral GnRH antagonists but remains uneven. Some commercial plans limit coverage to 6-12 months, and cost-sharing can be substantial without manufacturer assistance. Medicaid coverage varies by state. Prior authorization requirements are common and may require documentation of failed first-line therapies.
IVF-related GnRH analog costs are typically covered as part of the overall IVF cycle in states with IVF mandates but may be out-of-pocket expenses in states without mandated coverage. The cost of cetrorelix or ganirelix for an IVF cycle is relatively modest ($300-600 total) compared to the overall IVF cycle cost ($15,000-25,000+).
Compounded gonadorelin for TRT adjunctive use is typically not covered by insurance and represents an out-of-pocket expense for patients. At $15-50 per month, it remains one of the most affordable peptide therapies available.
Biosimilar and Generic Pipeline
The coming decade will see increased generic and biosimilar competition in the GnRH analog market. Key anticipated developments include additional generic leuprolide depot formulations (technical challenges with PLGA microsphere manufacturing have limited generic entrants), potential generic triptorelin products in the US market, and biosimilar or follow-on peptide GnRH antagonists.
For the oral antagonists, generic competition will depend on patent expiration timelines. Elagolix's composition-of-matter patents extend through the early 2030s. Relugolix's patent protection varies by indication and formulation. These timelines suggest that oral GnRH antagonists will remain branded, higher-cost products for the near future.
Patient Resources and Support
Understanding Your Treatment
Patients starting GnRH analog therapy often have questions and concerns about what to expect. The following resources can support informed decision-making and treatment adherence.
FormBlends Research Library: This report is part of a comprehensive peptide research library covering all major therapeutic peptides. Related reports that may be relevant to GnRH analog users include the Gonadorelin Research Report (detailed coverage of gonadorelin specifically), the Kisspeptin Research Report (the upstream regulator of GnRH), and the Oxytocin Research Report (reproductive health and bonding).
Free Assessment: The FormBlends Free Assessment tool helps patients explore which peptide protocols may be appropriate for their health goals. It provides personalized initial guidance based on individual health profiles, goals, and circumstances.
Dosing Calculator: For patients prescribed gonadorelin or other peptides, the Dosing Calculator can assist with reconstitution calculations and dosing schedules. Always follow your prescriber's specific instructions, as they may differ from general guidelines.
Science Page: Visit FormBlends Science for information about the evidence standards and quality assurance processes that underpin the research presented in this library.
Questions to Ask Your Healthcare Provider
Before starting GnRH analog therapy, consider discussing the following questions with your healthcare provider:
- What specific GnRH analog are you recommending, and why this one rather than alternatives?
- What should I expect during the first few weeks of treatment (particularly regarding the hormonal flare with agonists)?
- How long will I need to continue treatment?
- What monitoring tests will be needed, and how often?
- What side effects should I watch for, and which ones warrant immediate medical attention?
- How will this treatment affect my fertility, and is the effect fully reversible?
- Are there lifestyle modifications (exercise, diet, supplements) that can help mitigate side effects?
- Should I have a baseline DEXA scan before starting treatment?
- Are there any medications I'm taking that could interact with the GnRH analog?
- What is the plan if this treatment doesn't work as expected?
Being an informed and active participant in your treatment decisions leads to better outcomes. The information in this report is designed to support that goal, but it does not replace the personalized guidance of your healthcare provider who knows your individual medical history, risk factors, and treatment goals.
Endometriosis Management with GnRH Analogs: Comprehensive Clinical Guide
Understanding Endometriosis Pathophysiology
To appreciate why GnRH analogs are so effective for endometriosis, a brief review of the disease's pathophysiology is helpful. Endometriosis is defined by the presence of endometrial-like tissue outside the uterine cavity, most commonly on the peritoneal surfaces, ovaries, fallopian tubes, and uterosacral ligaments. These ectopic implants respond to the same hormonal signals as the eutopic endometrium, undergoing cyclical proliferation, secretion, and breakdown with each menstrual cycle.
The disease process involves chronic inflammation driven by cyclical bleeding from ectopic implants. Peritoneal irritation and adhesion formation cause pelvic pain. Ovarian endometriomas (chocolate cysts) can compromise ovarian reserve. Distortion of pelvic anatomy can impair tubal function and fertility. Neurotropism, meaning nerve infiltration by endometriotic implants, contributes to neuropathic pain. Altered peritoneal fluid composition creates a hostile environment for fertilization and implantation.
Estrogen is the primary driver of endometriotic tissue growth. Unlike normal endometrium, endometriotic implants produce their own estrogen through local aromatase expression, creating an autocrine loop that sustains the disease even in the setting of reduced systemic estrogen. This local estrogen production is one reason why complete estrogen suppression (as achieved by GnRH agonists) may be more effective than partial suppression (as provided by low-dose oral antagonists) for more advanced disease.
Staging Endometriosis and Matching Treatment Intensity
The revised American Society for Reproductive Medicine (rASRM) classification stages endometriosis from I (minimal) through IV (severe) based on the extent and depth of implants, adhesions, and ovarian involvement. While staging correlates imperfectly with symptom severity (some women with Stage I disease have debilitating pain, while some with Stage IV are asymptomatic), it helps guide the intensity of medical and surgical intervention.
| Stage | Description | Typical First-Line Medical Therapy | GnRH Analog Role |
|---|---|---|---|
| I (Minimal) | Isolated superficial implants, no adhesions | NSAIDs, combined OC pills, progestins | Second-line if hormonal contraceptives fail |
| II (Mild) | More extensive superficial implants, minimal adhesions | Combined OC pills, progestins, oral antagonists | Second-line; oral antagonists preferred |
| III (Moderate) | Deep implants, small endometriomas, moderate adhesions | Oral GnRH antagonists with add-back; surgical excision | Primary medical therapy option |
| IV (Severe) | Large endometriomas, dense adhesions, extensive deep disease | Surgery typically required; GnRH agonist pre-/post-surgical | Pre-surgical preparation; post-surgical suppression |
The Oral GnRH Antagonist Revolution in Endometriosis
Before 2018, medical management of endometriosis beyond first-line therapies was limited to injectable GnRH agonists (with their attendant side effects, injection burden, and 6-month treatment limitation) and progestins (which some patients cannot tolerate). The introduction of oral GnRH antagonists fundamentally changed the treatment paradigm for several reasons.
Dose-dependent estrogen control: The ability to partially suppress estrogen - reducing it enough to shrink endometriotic implants and relieve pain but maintaining it high enough to prevent bone loss and severe menopausal symptoms - was not possible with injectable GnRH agonists. These older agents produce near-complete estrogen suppression (less than 20 pg/mL), requiring add-back therapy as a separate medication to prevent bone and menopausal complications. Oral antagonists like elagolix at 150 mg daily achieve estradiol levels of approximately 40-60 pg/mL - a range that provides therapeutic benefit while limiting side effects.
Extended treatment duration: The traditional 6-month limit on GnRH agonist therapy for endometriosis (imposed by bone density concerns) was a major limitation, as endometriosis is a chronic disease that frequently recurs after treatment cessation. Elagolix 150 mg daily is approved for up to 24 months, and relugolix combination therapy has no pre-specified maximum duration. This allows truly long-term medical management for the first time with a GnRH-based agent.
Patient autonomy: Oral daily dosing gives patients direct control over their treatment. If side effects become intolerable, the medication can be stopped immediately, with rapid return to normal hormonal cycling within days. This stands in contrast to depot GnRH agonist injections, which cannot be "undone" and continue to exert effects for weeks to months after the last injection.
Clinical Trial Evidence for Oral Antagonists in Endometriosis
The ELARIS EM-I and EM-II trials were the registration studies for elagolix in endometriosis. These were two identical Phase III, randomized, double-blind, placebo-controlled trials enrolling a total of 1,686 women with laparoscopically confirmed endometriosis and moderate-to-severe pain.
In these trials, elagolix demonstrated significant improvements compared to placebo in the co-primary endpoints of dysmenorrhea response (proportion of patients with reduced menstrual pain and stable or reduced analgesic use) and non-menstrual pelvic pain response. At 3 months, the dysmenorrhea response rate was 46% for elagolix 150 mg versus 20% for placebo, and 76% for elagolix 200 mg BID versus 20% for placebo. Non-menstrual pelvic pain response rates were 50% for elagolix 150 mg versus 37% for placebo, and 55% for elagolix 200 mg BID versus 37% for placebo.
The relugolix combination (40 mg relugolix + 1 mg estradiol + 0.5 mg norethindrone acetate, marketed as Myfembree) was studied in the SPIRIT 1 and SPIRIT 2 trials for endometriosis-associated pain. These trials demonstrated significant pain reduction compared to placebo for both dysmenorrhea and non-menstrual pelvic pain, with the built-in add-back therapy limiting bone density loss to clinically insignificant levels over 52 weeks of treatment.
Surgical Considerations and GnRH Analog Timing
The relationship between GnRH analog therapy and surgical management of endometriosis is complex and deserves careful consideration.
Pre-surgical GnRH agonist therapy (3 months before surgery): Benefits include reduction of implant size, vascularity, and inflammation, potentially simplifying the surgical procedure. The eutopic endometrium becomes atrophic, reducing postoperative bleeding. Hemoglobin levels may improve in patients with anemia from heavy menstrual bleeding. Potential drawbacks include that some superficial implants may become less visible against the peritoneal background after hormonal treatment, making complete surgical excision more difficult. Deeper implants are less affected by hormonal suppression and remain identifiable. The 3-month treatment delay may be unacceptable for patients in significant pain or those with fertility timelines.
Post-surgical GnRH analog therapy: Following surgical excision or ablation of endometriotic implants, hormonal therapy helps prevent or delay disease recurrence. Options include oral GnRH antagonists for long-term post-surgical suppression, injectable GnRH agonists for 3-6 months followed by transition to low-maintenance hormonal contraception, continuous oral contraceptives or progestins as lower-cost alternatives, and LNG-IUD (Mirena) for combined contraception and endometriosis suppression.
The optimal post-surgical hormonal strategy depends on the patient's fertility plans, surgical findings, and prior treatment history. Women planning immediate conception typically forego post-surgical hormonal suppression to take advantage of the "window of opportunity" for improved fertility in the 6-12 months following endometriosis surgery.
Uterine Fibroids and GnRH Analog Therapy: Clinical Nuances
Fibroid Biology and Hormonal Sensitivity
Uterine fibroids (leiomyomas) are benign smooth muscle tumors of the myometrium, affecting up to 70-80% of women by age 50, with symptomatic disease in approximately 25-30%. Like endometriosis, fibroids are estrogen- and progesterone-dependent, but with an important distinction: progesterone plays a more prominent role in fibroid growth than in endometriosis, which is primarily estrogen-driven.
This difference in hormonal sensitivity has implications for GnRH analog therapy. While GnRH agonists and antagonists effectively suppress both estrogen and progesterone production (by suppressing gonadotropins), the add-back therapy used with oral GnRH antagonists typically includes a progestogen component (norethindrone acetate). Theoretically, this progestogenic add-back could partially antagonize the anti-fibroid effect. In practice, the low doses of progestogen used in add-back regimens (0.5 mg norethindrone acetate) appear insufficient to stimulate fibroid growth, and clinical trials have demonstrated sustained efficacy with combination therapy.
Fibroid Volume Reduction with GnRH Analogs
GnRH agonists are the most potent medical agents for fibroid volume reduction, typically achieving 30-60% decreases in fibroid volume and 35-65% decreases in uterine volume over 3-6 months of treatment. The maximum volume reduction is usually achieved by 3 months, with minimal additional shrinkage beyond that point.
This volume reduction is the primary reason GnRH agonists are used pre-surgically for fibroids. In women planning hysterectomy, pre-treatment can convert an abdominal procedure to a vaginal or laparoscopic approach, reduce operative time and blood loss, and correct pre-operative anemia by eliminating menstrual bleeding. In women planning myomectomy, pre-treatment reduces fibroid size (potentially allowing a laparoscopic rather than open approach), decreases vascularity (reducing intraoperative bleeding), and improves the surgical field by reducing adjacent tissue edema.
An important caveat: fibroid regrowth to pre-treatment size typically occurs within 3-6 months after GnRH agonist discontinuation. This means that agonist therapy is a temporizing measure for surgical planning rather than a definitive treatment. The oral GnRH antagonists with add-back therapy change this calculus by enabling long-term treatment, making them the first GnRH-based option for chronic fibroid management without surgery.
Heavy Menstrual Bleeding: The Primary Treatment Target
The oral GnRH antagonist combination products (Oriahnn, Myfembree, Yselty) are specifically approved for heavy menstrual bleeding (HMB) associated with uterine fibroids. The registration trials used menstrual blood loss (MBL) measured by alkaline hematin method as the primary endpoint, with response defined as MBL less than 80 mL per cycle and at least 50% reduction from baseline.
In the LIBERTY 1 and LIBERTY 2 trials (elagolix combination), approximately 69-77% of women in the treatment group achieved the primary endpoint versus 9-10% with placebo. The VENUS 1 and VENUS 2 trials (relugolix combination) showed similar results, with approximately 73% of treatment group patients responding versus 19-22% with placebo. The PRIMROSE 1 and 2 trials (linzagolix) demonstrated 56-77% response rates depending on dose, versus 29-35% with placebo.
Beyond MBL reduction, these treatments also improved hemoglobin levels in anemic patients, reduced pain associated with fibroids, and decreased uterine and fibroid volumes, though volume reduction with oral antagonists is generally less than with injectable GnRH agonists due to the partial estrogen suppression approach.
GnRH Analogs in Gender-Affirming Care: Detailed Clinical Considerations
Rationale for Puberty Suppression
The use of GnRH agonists for puberty suppression in transgender and gender-diverse adolescents is based on several clinical considerations that distinguish this application from other uses of GnRH analog therapy.
The primary therapeutic goal is to pause the development of secondary sex characteristics that are incongruent with the adolescent's gender identity. For transgender girls (assigned male at birth, female gender identity), this means preventing deepening of the voice, facial hair growth, Adams apple development, masculine skeletal changes, and genital maturation. For transgender boys (assigned female at birth, male gender identity), this means preventing breast development, menstruation, widening of the hips, and feminine fat distribution.
Many of these pubertal changes are irreversible or only partially reversible without surgical intervention. Voice deepening, for example, cannot be fully reversed by hormonal means. Breast development, once it occurs, typically requires surgical mastectomy if masculinization is subsequently desired. By pausing puberty with GnRH agonists, these irreversible changes are prevented, preserving future options for the adolescent as they mature in their understanding of their gender identity.
Treatment Protocols
GnRH agonist protocols for puberty suppression in gender-diverse youth mirror those used for central precocious puberty, as the pharmacological goal is identical: suppression of the HPG axis to prevent pubertal progression.
Commonly used agents include leuprolide depot 3.75 mg intramuscular monthly or 11.25 mg intramuscular every 3 months, triptorelin depot 3.75 mg intramuscular monthly or 11.25 mg every 3 months, and histrelin 50 mg subcutaneous implant annually. The choice between agents follows the same considerations as for CPP - depot interval preference, needle tolerance, and adherence factors.
Treatment is typically initiated after the onset of puberty (Tanner stage 2 or later), following comprehensive evaluation including psychological assessment of gender dysphoria, medical evaluation to rule out other causes of gender-related distress, informed consent discussion with the adolescent and parents/guardians covering benefits, risks, unknowns, and alternatives, and assessment of decision-making capacity appropriate to the adolescent's age and maturity.
Monitoring During Puberty Suppression
Monitoring protocols for transgender youth on GnRH agonists include regular assessment of pubertal status through Tanner staging (every 3-6 months), growth velocity and height measurements (every 3-6 months), bone age assessment via wrist X-ray (every 12 months), bone mineral density via DEXA scan (every 1-2 years, particularly important given the extended duration of treatment), laboratory monitoring including LH, FSH, and sex steroids to confirm adequate suppression, mental health assessment and psychosocial support throughout treatment, and body mass index monitoring and metabolic assessment.
Transition to Gender-Affirming Hormones
When an adolescent on puberty suppression reaches an appropriate age and demonstrates persistent, well-supported gender identity, gender-affirming hormone therapy (GAHT) may be initiated. The transition typically involves continuing the GnRH agonist while starting cross-sex hormones at low doses, gradually increasing cross-sex hormone doses over 1-2 years to adult levels, and eventually discontinuing the GnRH agonist once cross-sex hormones achieve adequate suppression of endogenous sex steroids.
For transgender girls, estradiol is initiated alongside continued GnRH agonist therapy. For transgender boys, testosterone is started. The GnRH agonist provides a "clean hormonal slate" upon which the desired hormonal environment can be built, allowing more predictable and consistent feminization or masculinization.
Long-Term Outcome Data
Long-term follow-up data on individuals who underwent puberty suppression for gender dysphoria, while growing, remains limited in scope compared to the extensive decades-long follow-up data available for CPP treatment. Available studies have reported favorable psychological outcomes, including reduced depression and anxiety, improved quality of life, and decreased gender dysphoria scores. Physical safety data, drawn from the CPP experience and from gender-diverse youth studies, show no identified long-term safety concerns unique to this population beyond those expected from any long-term GnRH analog use (primarily bone density considerations).
A systematic review published in Frontiers in Endocrinology in 2025, examining 19 studies of GnRH agonist use in transgender and gender-diverse youth, found consistent evidence for effective pubertal suppression and generally positive psychological outcomes, while acknowledging the need for larger, longer-duration prospective studies to fully characterize long-term effects on bone health, brain development, fertility preservation, and psychosocial outcomes.
Areas of ongoing research include the impact of prolonged pubertal delay on peak bone mass acquisition, effects on neurocognitive development during the pubertal window, long-term fertility implications (particularly when puberty suppression is followed directly by cross-sex hormones without a period of natal puberty), and optimal timing for transition from puberty suppression to gender-affirming hormones.
GnRH Analogs in Modern Prostate Cancer Treatment: Combination Strategies
The Evolving Role of ADT in the Treatment Landscape
The role of GnRH analog-based ADT in prostate cancer has evolved dramatically over the past decade. While ADT remains the backbone of treatment for advanced disease, it is increasingly used in combination with other agents rather than as monotherapy. Understanding these combinations is essential for clinicians prescribing GnRH analogs in the oncology setting.
The modern treatment framework recognizes that prostate cancer cells can adapt to castrate testosterone levels through several mechanisms, including androgen receptor amplification, gain-of-function androgen receptor mutations, constitutively active androgen receptor splice variants (particularly AR-V7), intratumoral androgen synthesis, and activation of alternative survival pathways that bypass androgen receptor signaling. These resistance mechanisms drive the eventual progression from castration-sensitive to castration-resistant prostate cancer (CRPC), typically occurring after a median of 18-36 months of ADT.
ADT Plus Novel Hormonal Agents
Multiple randomized Phase III trials have demonstrated that adding novel hormonal agents to ADT improves survival outcomes in metastatic hormone-sensitive prostate cancer (mHSPC):
ADT + Abiraterone: The LATITUDE and STAMPEDE trials showed that adding abiraterone acetate (a CYP17A1 inhibitor that blocks intratumoral androgen synthesis) to ADT in men with newly diagnosed mHSPC improved overall survival by approximately 18 months compared to ADT alone. Abiraterone is administered orally at 1000 mg daily with prednisone 5 mg daily, in combination with a GnRH agonist or antagonist.
ADT + Enzalutamide: The ENZAMET and ARCHES trials demonstrated that adding enzalutamide (a second-generation androgen receptor antagonist) to ADT improved radiographic progression-free survival and overall survival in mHSPC. Enzalutamide 160 mg daily is taken orally alongside GnRH analog therapy.
ADT + Apalutamide: The TITAN trial showed improved overall survival with the addition of apalutamide (another second-generation androgen receptor antagonist) to ADT in mHSPC. Apalutamide 240 mg daily is combined with standard GnRH analog-based ADT.
ADT + Darolutamide + Docetaxel: The ARASENS trial demonstrated that the triplet combination of ADT, darolutamide (a structurally distinct androgen receptor antagonist), and docetaxel chemotherapy improved overall survival compared to ADT plus docetaxel in men with mHSPC. This represents the most intensive upfront combination currently in use.
In all of these combination regimens, the GnRH analog (whether agonist or antagonist) serves as the foundation of hormonal therapy, with the additional agents intensifying androgen pathway suppression or adding cytotoxic activity. The choice of GnRH analog itself (leuprolide, triptorelin, goserelin, degarelix, or relugolix) does not typically affect the selection of the combination partner, as all achieve comparable testosterone suppression.
Intermittent ADT: Detailed Protocol Considerations
For men with lower-volume metastatic disease or biochemical recurrence, intermittent ADT offers a way to balance disease control with quality of life. The practical implementation involves several decision points that clinicians must navigate.
Induction Phase: ADT is initiated with a GnRH agonist depot formulation (typically a 3- or 6-month preparation) for an induction period of 6-9 months. The choice of 6 versus 9 months depends on how quickly PSA reaches a satisfactory nadir. Most clinicians require a PSA nadir of less than 4 ng/mL (and ideally less than 0.2 ng/mL) before considering an off-treatment interval.
Off-Treatment Phase: Once PSA reaches nadir, the GnRH agonist is discontinued. During this phase, testosterone gradually recovers over 2-6 months (faster with shorter induction periods, slower with longer ones). As testosterone recovers, patients typically experience improvement in energy, sexual function, mood, and musculoskeletal symptoms. PSA monitoring continues monthly during the off-treatment phase.
Re-Initiation Triggers: ADT is restarted when PSA rises above a predefined threshold, most commonly 10-20 ng/mL for metastatic disease or 4-10 ng/mL for biochemical recurrence. Some clinicians also use testosterone recovery above a certain level or PSA doubling time as re-initiation criteria. The SWOG 9346 trial used a PSA threshold of 20 ng/mL for re-initiation.
Cycle Duration: With each subsequent cycle, the off-treatment interval tends to shorten as the disease becomes less responsive to hormonal manipulation. Most men complete 2-5 intermittent cycles before transitioning to continuous ADT when the off-treatment interval becomes impractically short or disease progression occurs.
ADT Side Effect Mitigation: A Structured Approach
Given that many men will receive ADT for years, a structured approach to side effect mitigation is essential for maintaining quality of life and preventing long-term complications. The following framework addresses the major domains of ADT-related morbidity.
Body Composition and Metabolic Health:
- Structured resistance exercise program (2-3 sessions per week): reduces muscle loss, improves insulin sensitivity, and helps maintain bone density. Studies show that supervised exercise during ADT significantly reduces fat gain and preserves lean mass.
- Aerobic exercise (150+ minutes per week of moderate-intensity activity): improves cardiovascular fitness, reduces fatigue, and supports metabolic health
- Dietary counseling emphasizing adequate protein intake (1.2-1.5 g/kg/day), calcium-rich foods, and Mediterranean-style dietary patterns
- Metabolic monitoring (fasting glucose, HbA1c, lipid panel) every 3-6 months during the first year, then annually
- Early intervention for metabolic syndrome components (metformin for prediabetes, statins for dyslipidemia, antihypertensive therapy as needed)
Bone Health:
- Baseline DEXA scan before or within 3 months of initiating ADT
- Calcium supplementation (1000-1200 mg daily from diet and supplements combined)
- Vitamin D supplementation (1000-2000 IU daily, targeting serum 25-OH vitamin D levels of 30+ ng/mL)
- Consider bone-protective agent for all men planned for greater than 12 months of ADT: denosumab 60 mg SC every 6 months or zoledronic acid 4 mg IV annually
- Fall prevention assessment in elderly patients
- DEXA monitoring every 1-2 years during ADT
Cardiovascular Protection:
- Comprehensive cardiovascular risk assessment at baseline (Framingham, ASCVD risk calculator)
- Optimization of modifiable risk factors: blood pressure control (target less than 130/80 mmHg), lipid management (consider statin therapy for 10-year ASCVD risk greater than 7.5%), glycemic control, smoking cessation, weight management
- Consideration of GnRH antagonist (degarelix or relugolix) for men with established cardiovascular disease or significant risk factors
- Cardiology referral for men with pre-existing cardiac conditions starting ADT
Sexual Health:
- Proactive discussion of sexual side effects before starting ADT
- Referral to sexual health or urology specialist for men who desire active intervention
- PDE5 inhibitors (sildenafil, tadalafil) for erectile dysfunction, though efficacy is limited in the setting of castrate testosterone
- Couples counseling when relationship stress is present
- Acknowledgment that sexual side effects are reversible in men on intermittent ADT during off-treatment phases
Cognitive and Psychological Well-Being:
- Baseline cognitive assessment for men over 65 starting ADT
- Screening for depression and anxiety at each visit (PHQ-9, GAD-7)
- Cognitive stimulation activities (reading, puzzles, social engagement, learning new skills)
- Referral for psychological support or counseling when indicated
- Physical exercise as a dual-purpose intervention (improves both cognitive function and mood)
Hot Flash Management:
- Lifestyle modifications: layered clothing, cool environments, avoiding triggers (caffeine, alcohol, spicy food, hot beverages)
- Pharmacological options if lifestyle measures insufficient: venlafaxine 37.5-75 mg daily (most studied), gabapentin 300-900 mg daily, megestrol acetate 20-40 mg daily (use with caution due to progestogenic effects), oxybutynin 2.5-5 mg twice daily (emerging evidence)
- Acupuncture (limited evidence but low risk, some RCTs showing modest benefit)
This structured mitigation approach should be implemented proactively at the time ADT is initiated, not reactively after complications develop. Many of these interventions (particularly exercise and bone protection) are most effective when started early. Peptide therapies like BPC-157 for musculoskeletal healing or CJC-1295/Ipamorelin for body composition support may be considered as adjuncts under appropriate medical supervision, though these should always be discussed with the treating oncologist given the hormone-sensitive nature of the underlying cancer.
GnRH Analogs in Male Factor Infertility
Hypogonadotropic Hypogonadism: Where GnRH Analogs Restore Fertility
While most clinical applications of GnRH analogs involve suppressing the reproductive axis, one important application does the opposite: pulsatile GnRH therapy for male hypogonadotropic hypogonadism (HH).
Men with HH have inadequate GnRH signaling from the hypothalamus, resulting in low LH and FSH levels and consequently low testosterone and absent spermatogenesis. Causes include congenital conditions such as Kallmann syndrome (anosmia + HH due to failed GnRH neuron migration) and isolated HH (functional GnRH neurons that fail to activate at puberty), as well as acquired conditions such as pituitary tumors, hemochromatosis, hyperprolactinemia, anabolic steroid use, opioid-induced hypogonadism, and obesity-related HH.
For men with hypothalamic HH (as opposed to pituitary disease), pulsatile gonadorelin therapy can restore the entire reproductive axis by mimicking normal GnRH pulsatility. Administered via a portable pump delivering 5-25 mcg per pulse every 90-120 minutes, pulsatile GnRH therapy stimulates endogenous LH and FSH secretion, which in turn drives testicular testosterone production and spermatogenesis.
Success rates for fertility with pulsatile GnRH therapy in male HH are encouraging but dependent on baseline gonadal function. Men with congenital HH who have never undergone puberty may require extended treatment (12-24 months) before spermatogenesis is established. Those with acquired HH (e.g., post-anabolic steroid use) typically respond more quickly, as the testes have previously functioned normally.
Post-Anabolic Steroid Recovery
The use of anabolic-androgenic steroids (AAS) is a common cause of acquired hypogonadotropic hypogonadism in men, with complete suppression of LH, FSH, and endogenous testosterone production during AAS use. Recovery of the HPG axis after AAS discontinuation is variable and can take months to years, depending on the duration and intensity of steroid use.
Gonadorelin has been proposed as a component of post-cycle therapy (PCT) following AAS use, with the theoretical advantage of stimulating the HPG axis from the top down (hypothalamic-pituitary level) rather than at the gonadal level (as hCG does) or at the pituitary feedback level (as SERMs like clomiphene and tamoxifen do).
However, gonadorelin's role in PCT remains debated. Its very short half-life (2-4 minutes IV, 10-40 minutes SC) means that intermittent subcutaneous injections may not provide sufficient sustained gonadotropin stimulation to meaningfully accelerate HPG axis recovery. In contrast, SERMs (which block estrogen negative feedback at the hypothalamus and pituitary) and hCG (which directly stimulates Leydig cells) have more established evidence for PCT efficacy.
A practical approach for men recovering from AAS-induced hypogonadism might include initial SERM therapy (clomiphene 25-50 mg daily or enclomiphene 12.5-25 mg daily) as the primary axis stimulant, adjunctive gonadorelin 100-200 mcg SC 2-3 times weekly to provide additional GnRH-level stimulation, serial monitoring of LH, FSH, total testosterone, and semen parameters every 4-6 weeks, and gradual tapering as endogenous function recovers.
This area lacks randomized controlled trial data, and protocols are based primarily on clinical experience and physiological reasoning. Patients should be counseled about the limited evidence base and the importance of monitoring.
GnRH Pump Therapy: Technical Considerations
For the small number of men who receive pulsatile GnRH pump therapy, several practical considerations affect treatment success. The pump device is similar to an insulin pump, worn externally with a subcutaneous infusion set. Pulse parameters must be carefully programmed. Standard settings for male HH involve pulse doses of 5-25 mcg of gonadorelin acetate, pulse intervals of 90-120 minutes, continuous 24-hour operation (GnRH pulsatility occurs during sleep as well as waking hours), and dose adjustment based on LH/FSH response and semen analysis results.
Patient training is essential for infusion site care and rotation, pump programming and troubleshooting, recognition of infusion site infections or occlusion, and medication reconstitution and cartridge changes. Treatment duration for fertility induction in congenital HH is typically 12-24 months. Success rates for sperm appearance in the ejaculate are approximately 70-90%, with pregnancy rates of 50-70% in couples without female factor infertility.
Despite its physiological elegance, pulsatile GnRH pump therapy for male HH has been largely replaced by combination gonadotropin therapy (hCG + recombinant FSH) in most clinical settings, primarily due to the logistical complexity of pump wear and the limited availability of pump-compatible gonadorelin formulations. However, it remains the most physiological approach to fertility induction in HH and may offer advantages in specific clinical scenarios.