Executive Summary

Figure 1: Oxytocin research spans social bonding, metabolic regulation, and psychiatric therapeutics
Key Takeaways
- Oxytocin is a nine-amino-acid peptide with therapeutic potential spanning autism, obesity, schizophrenia, PTSD, and anxiety
- A 2024 meta-analysis found 48 IU/day intranasal oxytocin improves social cognition in ASD, while lower doses show no benefit over placebo
- Single-dose oxytocin reduces caloric intake by ~152 kcal per meal, but 8-week trials show no significant weight loss
- Intranasal delivery achieves peak plasma levels in 15-30 minutes with a favorable safety profile at doses of 18-40 IU
- Oral oxytocin analogs and combination therapy approaches represent the next frontier in oxytocin therapeutics
Oxytocin is a nine-amino-acid neuropeptide synthesized in the hypothalamus that has moved far beyond its original association with labor and lactation. Current research positions oxytocin at the intersection of social neuroscience, metabolic medicine, and psychiatric pharmacology, with clinical trials spanning autism spectrum disorder, obesity, schizophrenia, PTSD, and anxiety disorders.
For decades, the popular media reduced oxytocin to the "love hormone" or "cuddle chemical," a simplification that obscured the peptide's genuine complexity. Yes, oxytocin plays a central role in pair bonding, maternal behavior, and trust. But the molecule also modulates appetite, influences glucose metabolism, shapes fear responses, and regulates inflammatory pathways. The scientific literature now contains over 20,000 publications on oxytocin, and the therapeutic pipeline includes more than a dozen active clinical programs exploring its potential in conditions ranging from autism to obesity.
The peptide's most studied therapeutic application remains autism spectrum disorder (ASD). A 2024 meta-analysis of 12 randomized controlled trials involving 498 participants found that while lower doses of intranasal oxytocin showed no significant improvement over placebo, a daily dose of 48 IU produced measurable benefits in social cognition and reduced repetitive behaviors. This dose-response relationship has reshaped how researchers design oxytocin trials, moving away from one-size-fits-all protocols toward personalized dosing strategies.
On the metabolic front, oxytocin has demonstrated the ability to reduce caloric intake, increase fat oxidation, and improve insulin sensitivity after a single intranasal dose. An 8-week randomized controlled trial published in 2024 found that oxytocin reduced caloric intake by approximately 152 kcal per test meal compared to placebo. However, this reduction did not translate into statistically significant weight loss over the trial period, raising questions about whether longer treatment durations or combination therapies with compounds like semaglutide or tirzepatide might produce more meaningful results.
The psychiatric application portfolio extends to schizophrenia, where oxytocin has shown some ability to improve social cognition and reduce negative symptoms, though larger randomized trials have yielded mixed results. In PTSD, early clinical work suggested that oxytocin could reduce the re-experiencing of traumatic memories, particularly when combined with psychotherapy. For anxiety disorders, a five-day course of low-dose oxytocin reduced anxiety symptoms in 147 patients, with chronic applications proving more successful than single-dose protocols.
Intranasal delivery remains the primary route of administration for clinical research. The nasal mucosa offers direct access to the brain through the olfactory and trigeminal nerve pathways, bypassing the blood-brain barrier. Plasma concentrations typically peak within 15 to 30 minutes of intranasal dosing and return to baseline within 90 minutes. For those interested in oxytocin peptide therapy, understanding the pharmacokinetics of intranasal delivery is essential for timing doses around social interactions or therapeutic sessions.
The safety profile of intranasal oxytocin is favorable. In controlled research settings, doses of 18 to 40 IU produce no detectable subjective changes and no reliable side effects. The most commonly reported adverse events in longer-term studies include nasal discomfort (14.3%), tiredness (7.2%), and mild irritability (9.0%). This stands in contrast to intravenous oxytocin used in obstetric settings, where high doses carry risks of water intoxication, cardiac arrhythmias, and uterine hyperstimulation.
Looking ahead, the field is moving toward several promising directions. Researchers at the University of Queensland have developed gut-stable oxytocin analogs that can be taken orally, potentially addressing the limitations of intranasal delivery. Others are exploring oxytocin receptor-targeted PET imaging agents for breast cancer diagnostics. And the concept of combining oxytocin with social skills training or cognitive behavioral therapy represents a particularly promising therapeutic paradigm, where the peptide's prosocial effects could amplify the benefits of structured interventions.
This report examines the current state of oxytocin research across each of these domains. We analyze the molecular biology of the oxytocin system, review the clinical evidence for its therapeutic applications, evaluate dosing protocols and delivery methods, and assess the safety data from both short-term and long-term studies. For clinicians, researchers, and individuals considering oxytocin therapy, this comprehensive review provides the evidence base needed to make informed decisions.
Key Takeaways
- Oxytocin is a nine-amino-acid peptide with therapeutic potential spanning autism, obesity, schizophrenia, PTSD, and anxiety
- A 2024 meta-analysis found 48 IU/day intranasal oxytocin improves social cognition in ASD, while lower doses show no benefit over placebo
- Single-dose oxytocin reduces caloric intake by ~152 kcal per meal, but 8-week trials show no significant weight loss
- Intranasal delivery achieves peak plasma levels in 15-30 minutes with a favorable safety profile at doses of 18-40 IU
- Oral oxytocin analogs and combination therapy approaches represent the next frontier in oxytocin therapeutics
Oxytocin Biology
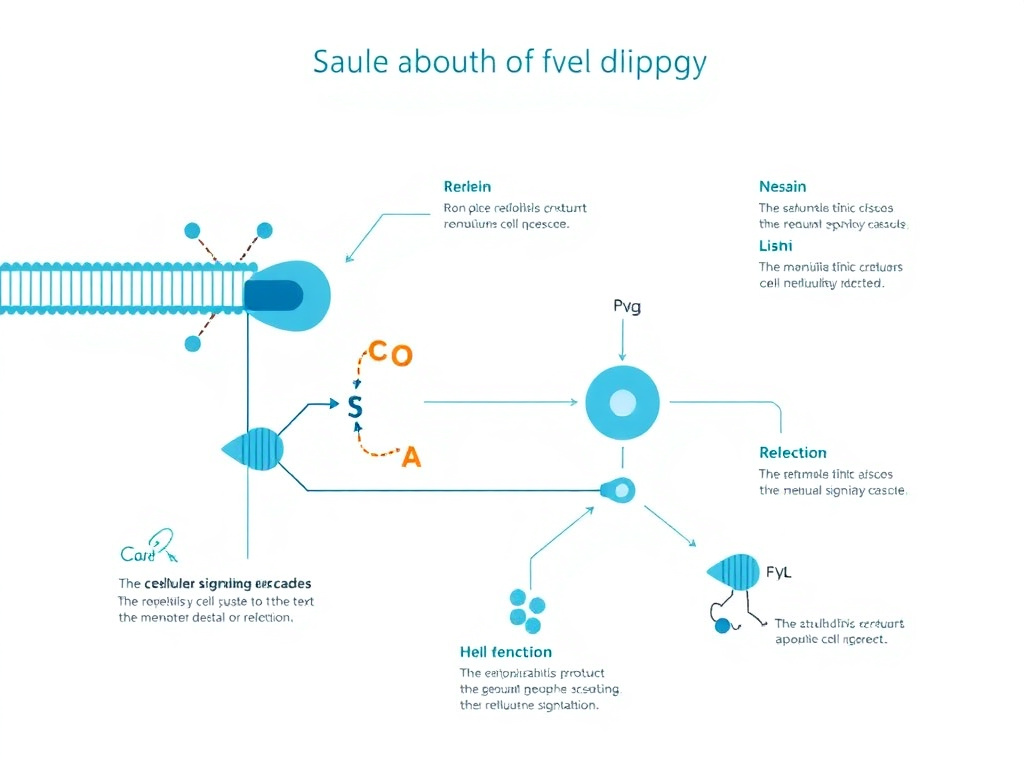
Figure 2: The oxytocin synthesis pathway from hypothalamic nuclei to systemic release
Molecular Structure and Synthesis
Oxytocin is a cyclic nonapeptide with the amino acid sequence Cys-Tyr-Ile-Gln-Asn-Cys-Pro-Leu-Gly-NH2. The cysteine residues at positions 1 and 6 form an intramolecular disulfide bond that creates the characteristic six-membered ring structure. This ring is connected to a three-residue tail ending in a glycine amide. The molecular weight of oxytocin is 1,007 daltons, making it one of the smallest bioactive peptides in the human body. The amidation of the C-terminal glycine is critical for biological activity; without it, the peptide loses virtually all receptor binding capacity.
The structural similarity between oxytocin and vasopressin (also known as antidiuretic hormone) is striking. The two peptides differ by only two amino acids: position 3 (isoleucine in oxytocin versus phenylalanine in vasopressin) and position 8 (leucine in oxytocin versus arginine in vasopressin). This close structural relationship explains why oxytocin can bind to vasopressin receptors at high concentrations, producing antidiuretic effects that become clinically relevant during high-dose intravenous oxytocin infusions. It also explains why researchers must carefully account for cross-reactivity when measuring oxytocin levels using immunoassays.
Oxytocin is synthesized as a much larger precursor protein called prepro-oxytocin, which consists of 125 amino acids. This precursor contains a signal peptide (19 amino acids), the oxytocin sequence (9 amino acids), a processing signal (3 amino acids that include the Gly-Lys-Arg tripeptide), and neurophysin I (93 amino acids). The precursor is processed during axonal transport from the cell body to the nerve terminals, where enzymatic cleavage releases the mature oxytocin peptide and neurophysin I. Neurophysin I serves as a carrier protein that binds oxytocin in the neurosecretory granules, stabilizing the peptide until release.
The gene encoding prepro-oxytocin (OXT) is located on chromosome 20p13 in humans. It spans approximately 900 base pairs and contains three exons separated by two introns. The regulatory region of the OXT gene contains estrogen response elements, which explains why estrogen upregulates oxytocin expression during pregnancy. The gene is expressed primarily in the hypothalamus, though oxytocin mRNA has been detected in peripheral tissues including the uterus, placenta, amnion, corpus luteum, testis, and heart.
Hypothalamic Production and Release
The primary site of oxytocin synthesis in the brain is the hypothalamus, specifically the magnocellular neurosecretory cells of the supraoptic nucleus (SON) and the paraventricular nucleus (PVN). These neurons are among the largest in the brain, with cell bodies measuring 20 to 35 micrometers in diameter. Their size reflects the enormous metabolic demands of producing, packaging, and transporting neuropeptide-containing secretory granules along axons that can extend several centimeters to reach the posterior pituitary gland.
The magnocellular neurons project their axons through the hypothalamo-neurohypophysial tract to the posterior pituitary (also called the neurohypophysis), where they terminate in specialized nerve endings called Herring bodies. These swellings contain dense-core granules packed with oxytocin and neurophysin I, ready for release into the systemic circulation. A single magnocellular neuron can contain up to 500 million granules, each holding approximately 85,000 oxytocin molecules. This massive storage capacity allows for rapid, large-scale release of oxytocin in response to physiological stimuli like suckling or cervical dilation during labor.
But the PVN contains more than just magnocellular neurons. A separate population of parvocellular neurons synthesizes oxytocin and projects to diverse brain regions including the amygdala, hippocampus, nucleus accumbens, ventral tegmental area, and brainstem nuclei. These central projections are thought to mediate oxytocin's effects on social behavior, stress responses, and emotional processing. This is a key distinction: while magnocellular neurons are primarily responsible for systemic oxytocin release into the bloodstream, parvocellular neurons deliver oxytocin directly to specific brain circuits where it functions as a neuromodulator.
Oxytocin release from magnocellular neurons occurs through a distinctive firing pattern called burst firing or milk ejection bursts. During suckling, for example, a synchronous burst of electrical activity sweeps through the oxytocin neuron population, producing a bolus release of oxytocin into the blood. Each burst lasts only 2 to 4 seconds but triggers the release of enough oxytocin to produce milk letdown within 30 to 60 seconds. Between bursts, there is a refractory period of several minutes during which the neurons cannot be re-stimulated. This pulsatile release pattern is functionally important because sustained oxytocin release leads to receptor desensitization and reduced biological effects.
Oxytocin is also released within the brain itself through dendritic release, a process in which the peptide is secreted from the dendrites and cell bodies of magnocellular neurons rather than from their axon terminals. Dendritic release can occur independently of axonal release and is triggered by different stimuli. This mechanism allows oxytocin to diffuse through the extracellular space of the hypothalamus and adjacent brain regions, acting as a volume transmitter that can influence large populations of neurons simultaneously. Research with other peptides like VIP and kisspeptin has revealed similarly complex release mechanisms in hypothalamic circuits.
The Oxytocin Receptor System
The oxytocin receptor (OXTR) is a G protein-coupled receptor (GPCR) belonging to the rhodopsin-type (class A) family. The receptor protein consists of 389 amino acids organized into seven transmembrane domains, an extracellular N-terminal domain, and an intracellular C-terminal domain. The OXTR gene is located on chromosome 3p25 in humans and spans approximately 17 kilobases, containing 3 introns and 4 exons.
When oxytocin binds to its receptor, the OXTR couples primarily to Gq/11 proteins, activating phospholipase C-beta (PLC-beta). This enzyme cleaves phosphatidylinositol 4,5-bisphosphate (PIP2) into two second messengers: inositol 1,4,5-trisphosphate (IP3) and diacylglycerol (DAG). IP3 triggers calcium release from the endoplasmic reticulum, raising intracellular calcium levels that drive smooth muscle contraction in the uterus and myoepithelial cells in the breast. DAG activates protein kinase C (PKC), which phosphorylates downstream targets involved in cell growth, differentiation, and gene expression.
The OXTR can also couple to Gi proteins, particularly in neurons, where it inhibits adenylyl cyclase and reduces cyclic AMP levels. This dual coupling capacity means that the same receptor can produce different intracellular responses depending on the cell type and the relative availability of different G protein subunits. In the brain, Gi coupling is associated with oxytocin's anxiolytic and stress-reducing effects, while Gq coupling predominates in peripheral tissues involved in reproduction.
Beyond G protein signaling, the OXTR activates several additional pathways. These include the mitogen-activated protein kinase (MAPK) cascade, the phosphoinositide 3-kinase (PI3K)/Akt pathway, and the calcium/calmodulin-dependent kinase (CaMK) pathway. The MAPK cascade, which includes ERK1/2, is particularly important for oxytocin's effects on neuroplasticity and memory formation. Studies with other neuropeptides like Semax and Selank have revealed similar MAPK-dependent mechanisms of neuroprotection.
The distribution of oxytocin receptors in the brain is both widespread and region-specific. High densities of OXTR are found in the amygdala (particularly the central and medial nuclei), nucleus accumbens, ventromedial hypothalamus, hippocampus, and brainstem nuclei involved in autonomic regulation. In the periphery, oxytocin receptors are expressed in the myometrium, mammary glands, heart, kidneys, adipose tissue, pancreatic islets, and the gastrointestinal tract. The expression of OXTR is dynamically regulated by hormonal status: estrogen dramatically upregulates receptor expression, while progesterone suppresses it. This hormonal regulation explains the 300-fold increase in uterine oxytocin receptor density that occurs during pregnancy.
Peripheral Functions Beyond Reproduction
While oxytocin's roles in parturition and lactation are well established, the peptide serves multiple functions throughout the body that are only now being fully appreciated. In the cardiovascular system, oxytocin receptors on cardiomyocytes mediate cardioprotective effects, including the release of atrial natriuretic peptide (ANP) and the promotion of cardiomyocyte differentiation. Oxytocin also acts as a vasodilator through nitric oxide-dependent mechanisms, reducing blood pressure and afterload.
In bone metabolism, oxytocin promotes osteoblast differentiation and bone formation while inhibiting osteoclast activity. Animal studies have shown that oxytocin-deficient mice develop severe osteoporosis, suggesting that the peptide plays a tonic role in maintaining bone density. This finding has implications for osteoporosis treatment, particularly in postmenopausal women who experience both declining estrogen and reduced oxytocin signaling.
The gastrointestinal tract represents another important site of oxytocin action. Oxytocin receptors are expressed throughout the gut, and the peptide influences gastric motility, intestinal transit time, and visceral pain perception. Researchers at the University of Queensland have developed gut-stable oxytocin analogs that can be taken orally, creating new therapeutic possibilities for conditions like irritable bowel syndrome (IBS) and inflammatory bowel disease (IBD). These analogs maintain full potency and selectivity for the oxytocin receptor while resisting degradation by gastrointestinal enzymes.
In adipose tissue, oxytocin promotes lipolysis and reduces adipogenesis. The peptide also acts on pancreatic beta cells to stimulate insulin release, and on hepatocytes to enhance glucose uptake. These metabolic effects make oxytocin an intriguing complement to established metabolic therapies. Researchers have begun investigating whether oxytocin could enhance the weight loss effects of GLP-1 receptor agonists like semaglutide or dual GIP/GLP-1 agonists like tirzepatide by addressing different components of the metabolic equation.
Epigenetic Regulation of the Oxytocin System
The expression of both the OXT and OXTR genes is subject to epigenetic regulation through DNA methylation and histone modification. Methylation of CpG sites in the OXTR promoter region is associated with reduced receptor expression and has been linked to individual differences in social behavior, empathy, and stress reactivity. In autism spectrum disorder, several studies have found increased OXTR methylation, which may contribute to the social cognition deficits characteristic of the condition.
Early life experiences profoundly influence OXTR methylation patterns. Adverse childhood experiences, including neglect and abuse, are associated with increased OXTR methylation in adulthood, potentially creating a molecular pathway through which early social deprivation leads to lasting changes in social behavior. Conversely, positive social experiences, including secure attachment relationships, are associated with lower OXTR methylation and greater receptor expression.
These epigenetic findings have practical implications for oxytocin-based therapies. Individuals with high levels of OXTR methylation may have fewer available receptors and therefore respond differently to exogenous oxytocin. This could explain some of the variability in clinical trial outcomes and supports the development of personalized dosing approaches based on epigenetic profiling. The science behind peptide therapy increasingly recognizes the importance of individual variation in receptor expression and signaling capacity.
Oxytocin Metabolism and Clearance
Oxytocin has a remarkably short plasma half-life of approximately 3 to 5 minutes. This rapid clearance is driven by several enzymatic pathways. Oxytocinase (also called leucyl/cystinyl aminopeptidase or LNPEP) is the primary enzyme responsible for oxytocin degradation in the bloodstream. This zinc-dependent metallopeptidase cleaves the Cys-Tyr bond in the cyclic ring, rendering the peptide biologically inactive. During pregnancy, oxytocinase levels increase dramatically, rising 10-fold by the third trimester, which serves to protect the uterus from premature oxytocin-induced contractions.
In the brain, oxytocin clearance is slower than in the periphery. The CSF half-life of oxytocin is estimated at approximately 20 to 28 minutes, roughly five times longer than the plasma half-life. This extended central half-life is consistent with the observation that the behavioral effects of intranasal oxytocin (which persist for 60 to 90 minutes) outlast the plasma pharmacokinetic profile. The slower central clearance reflects the lower concentration of degrading enzymes in the brain interstitial fluid compared to plasma.
The kidneys contribute to oxytocin clearance through both filtration and tubular degradation. Approximately 1 to 2% of circulating oxytocin is excreted intact in the urine, while the remainder is metabolized by renal peptidases. The liver also plays a role in oxytocin metabolism, with hepatic extraction estimated at 40 to 60% during first-pass clearance. This high hepatic extraction is one reason why oral oxytocin has negligible bioavailability without chemical modifications to resist enzymatic degradation.
Understanding oxytocin metabolism has practical implications for therapeutic development. The short half-life means that continuous or frequent dosing is required to maintain therapeutic levels. Long-acting oxytocin analogs aim to address this limitation by incorporating structural modifications that resist enzymatic degradation while preserving receptor binding affinity. Strategies include PEGylation (attachment of polyethylene glycol chains), lipidation (attachment of fatty acid chains), and cyclization modifications that protect vulnerable cleavage sites. These approaches parallel the half-life extension strategies used to develop long-acting GLP-1 agonists like semaglutide, which uses a fatty acid chain to promote albumin binding and extend its half-life to approximately one week.
Cross-Talk Between Oxytocin and Other Neuropeptide Systems
The oxytocin system does not operate in isolation. It interacts extensively with other neuropeptide and neurotransmitter systems, creating a complex web of regulatory relationships that modulate social behavior, stress responses, and metabolic function. Understanding these interactions is essential for predicting the effects of exogenous oxytocin and for designing combination therapies.
The oxytocin-dopamine interaction is one of the most well-characterized. Oxytocin neurons in the PVN project to the ventral tegmental area (VTA), where they enhance dopamine release in the nucleus accumbens during positive social interactions. This oxytocin-dopamine coupling is the neurochemical basis of social reward: it is the reason why social connection feels pleasurable and why social isolation is aversive. Disruptions in this coupling have been implicated in the social anhedonia seen in depression, schizophrenia, and autism.
Oxytocin also interacts with the serotonin system. Serotonin neurons in the dorsal raphe nucleus express oxytocin receptors, and oxytocin stimulates serotonin release in forebrain regions. This interaction is bidirectional: serotonin, acting through 5-HT1A receptors on oxytocin neurons, promotes oxytocin release from the PVN. SSRI antidepressants, by increasing synaptic serotonin levels, indirectly stimulate oxytocin release, which may contribute to their therapeutic effects on social withdrawal and anhedonia in depression.
The oxytocin-GABA interaction is relevant for the peptide's anxiolytic effects. Oxytocin enhances GABAergic inhibition in the amygdala, reducing the excitatory output that drives anxiety and fear responses. This mechanism parallels the anxiolytic action of benzodiazepines, which also enhance GABAergic transmission, though through a different molecular mechanism. The combination of oxytocin with GABAergic anxiolytics could theoretically produce additive anti-anxiety effects, though clinical studies of this combination are lacking.
The interaction between oxytocin and the endogenous opioid system is particularly interesting for pain management. Oxytocin neurons in the PVN project to the periaqueductal gray (PAG), a brainstem region critical for descending pain modulation. Activation of these projections produces analgesia through both direct oxytocin receptor-mediated effects and indirect activation of endogenous opioid pathways. This dual mechanism may explain why oxytocin reduces both physical pain and social pain (the emotional distress caused by social rejection or exclusion).
For those interested in the broader field of neuropeptide therapies, the interactions between oxytocin and other neuromodulatory systems suggest opportunities for strategic combination protocols. Selank modulates anxiety through GABAergic and serotonergic mechanisms, DSIP influences sleep and stress through opioid pathways, and Semax enhances cognitive function through BDNF signaling. Each of these peptides targets a different node in the neuromodulatory network, creating the potential for complementary effects when combined with oxytocin under appropriate medical supervision.
Evolutionary Origins and Comparative Biology
Oxytocin is one of the most evolutionarily ancient signaling molecules known. Homologs of oxytocin have been identified in organisms ranging from nematodes (where the homolog is called nematocin) to insects (inotocin), fish (isotocin), and amphibians (mesotocin). The oxytocin/vasopressin superfamily arose from a single ancestral gene that duplicated approximately 500 million years ago, producing two lineages: the oxytocin-like peptides and the vasopressin-like peptides. This deep evolutionary conservation suggests that the oxytocin signaling system serves fundamental biological functions that have been maintained across the animal kingdom.
In social insects like ants and bees, the oxytocin-like peptide inotocin is expressed in the brain and influences social behavior, including brood care and colony organization. In fish, isotocin regulates social dominance hierarchies and reproductive behavior. In birds, mesotocin mediates pair bonding, parental care, and flocking behavior. The consistent association between oxytocin-like peptides and social behavior across species separated by hundreds of millions of years of evolution provides strong evidence that social bonding is a deeply conserved biological function, not merely a cultural phenomenon.
The comparative biology perspective also informs our understanding of human oxytocin function. The prairie vole/meadow vole comparison has been particularly influential. Prairie voles are socially monogamous and show strong pair bonds, while the closely related meadow voles are polygamous and show no partner preference. The difference maps onto distinct patterns of oxytocin receptor distribution: prairie voles have high OXTR density in the nucleus accumbens and prefrontal cortex, while meadow voles have low OXTR density in these regions. This natural experiment demonstrates that variations in oxytocin receptor distribution can produce dramatic differences in social behavior, a principle that likely applies to individual differences in human social functioning as well.
Social Bonding & Trust
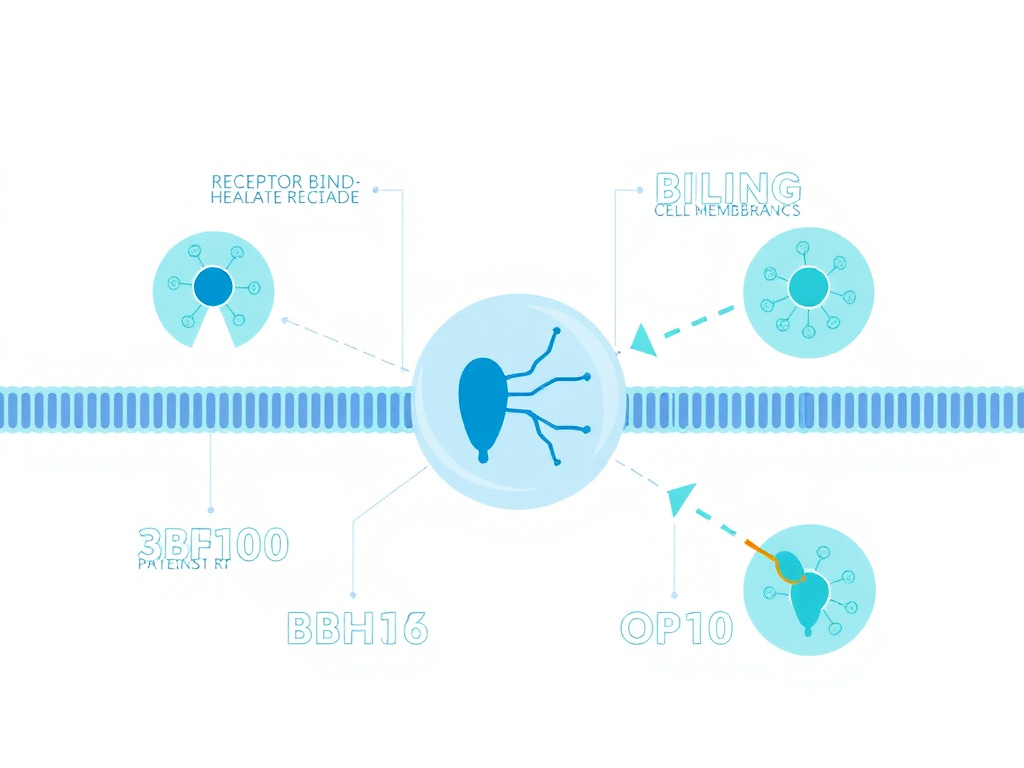
Figure 3: Neural circuits mediating oxytocin's effects on social bonding and trust
The Neural Architecture of Social Bonding
Oxytocin's role in social bonding operates through a network of brain regions collectively known as the social brain. This network includes the amygdala, which processes social threat and emotional salience; the fusiform gyrus, which processes facial identity; the superior temporal sulcus, which tracks gaze direction and biological motion; and the medial prefrontal cortex, which supports theory of mind and social decision-making. Oxytocin modulates activity within and connectivity between these regions, fundamentally altering how the brain processes social information.
The amygdala is the most consistently reported target of oxytocin's social effects. Functional magnetic resonance imaging (fMRI) studies have repeatedly shown that intranasal oxytocin reduces amygdala activation in response to threatening social stimuli, including angry faces, fear-inducing images, and scenes of social rejection. This dampening of amygdala reactivity is thought to underlie oxytocin's anxiolytic properties in social contexts. The peptide doesn't eliminate the ability to detect social threats; rather, it shifts the threshold at which ambiguous social signals are interpreted as threatening versus non-threatening.
At the same time, oxytocin increases the functional connectivity between the amygdala and the medial prefrontal cortex, strengthening top-down regulatory control over emotional responses. This enhanced connectivity may explain why oxytocin promotes approach behavior in social situations: the prefrontal cortex can more effectively modulate the amygdala's alarm signals, allowing individuals to engage with others even when some degree of social uncertainty exists. This mechanism parallels the anxiolytic properties observed with other neuropeptides like Selank, which also modulates anxiety through GABAergic and serotonergic pathways.
The nucleus accumbens, a central hub of the brain's reward system, also plays a key role in oxytocin-mediated bonding. In animal models, oxytocin release in the nucleus accumbens is necessary for partner preference formation in monogamous prairie voles. The peptide works by enhancing dopamine release in this region during positive social interactions, effectively tagging the social partner as a source of reward. This mechanism is thought to be relevant for understanding human attachment: the pleasurable feelings associated with social bonding are, at a neurochemical level, the result of oxytocin-dopamine interactions in the reward circuitry.
Trust, Cooperation, and Economic Decision-Making
The landmark study of oxytocin and trust was published in Nature in 2005 by Kosfeld and colleagues. In this experiment, participants who received intranasal oxytocin (24 IU) transferred significantly more money to an anonymous trustee in a trust game compared to those who received placebo. The oxytocin group transferred 17% more units on average, and 45% of oxytocin-treated participants showed maximal trust (transferring all units) compared to only 21% of the placebo group. This finding sparked an enormous research effort to understand how oxytocin influences social decision-making.
Subsequent studies have refined this picture considerably. Oxytocin does not indiscriminately increase trust. Rather, it appears to reduce the perceived risk of social interactions, making people more willing to extend trust when the potential social payoff is high. In dictator games, where there is no opportunity for reciprocity, oxytocin has less consistent effects on generosity, suggesting that the peptide specifically enhances trust-based cooperation rather than general altruism.
Oxytocin also promotes in-group favoritism. Several studies have shown that intranasal oxytocin increases cooperation with members of one's own group while having no effect on, or even reducing cooperation with, members of other groups. This "tend and defend" pattern suggests that oxytocin's prosocial effects are context-dependent: the peptide strengthens existing social bonds and group affiliations rather than promoting universal goodwill. In competitive intergroup situations, oxytocin may actually increase defensive aggression toward perceived outgroup threats.
The trust game research has been extended to examine oxytocin's effects on betrayal aversion. When participants experience a breach of trust, those who received placebo show a significant decline in subsequent trust behavior. Oxytocin-treated participants, however, maintain their level of trust even after being betrayed, suggesting that the peptide reduces the negative emotional impact of social disappointment. This finding has implications for therapeutic applications: in conditions characterized by excessive social withdrawal and interpersonal wariness, such as social anxiety disorder and schizophrenia, oxytocin may help patients re-engage with social relationships despite past negative experiences.
Empathy and Emotional Recognition
Oxytocin enhances the ability to infer the emotional states of others, a capacity known as cognitive empathy or mentalizing. In the Reading the Mind in the Eyes Test (RMET), a widely used measure of emotional recognition, oxytocin improves performance by 9 to 15% compared to placebo. This improvement is most pronounced for subtle emotional expressions and for negative emotions like fear and sadness, suggesting that oxytocin specifically enhances sensitivity to emotional cues that might otherwise be missed.
The peptide also amplifies affective empathy, the ability to share in the emotional experiences of others. Neuroimaging studies show that oxytocin increases activation in the anterior insula and anterior cingulate cortex when participants observe others experiencing pain, suggesting enhanced empathic resonance. This effect extends beyond physical pain: oxytocin also increases neural responses to social exclusion experienced by others, indicating that the peptide promotes empathy for both physical and social suffering.
Gender differences in oxytocin's empathic effects have been observed. In men, intranasal oxytocin consistently improves emotional recognition and increases empathic accuracy. In women, the effects are smaller and more variable, possibly because baseline oxytocin levels and receptor expression differ between sexes. Estrogen upregulates OXTR expression, meaning that premenopausal women may already have higher receptor availability and thus less room for additional oxytocin to produce measurable effects. These sex differences have important implications for clinical trial design and dosing protocols.
Attachment and Pair Bonding
In humans, plasma oxytocin levels increase during the early stages of romantic love. Couples who are in the first six months of a new relationship show oxytocin levels that are more than double those of single individuals, and these elevated levels correlate with the degree of interactive reciprocity between partners. At 6-month follow-up, couples who remained together continued to show elevated oxytocin, while those who had separated did not, suggesting that sustained oxytocin elevation may be both a marker and a mediator of relationship stability.
Parent-infant bonding is perhaps the most well-characterized social function of oxytocin. In mothers, oxytocin levels rise during pregnancy, peak during labor and delivery, and remain elevated during breastfeeding. Higher maternal oxytocin levels are associated with more affectionate maternal behaviors, including increased gaze, positive affect, and affectionate touch. Fathers also show elevated oxytocin during early parenthood, and paternal oxytocin levels correlate with the degree of stimulatory play and exploratory behavior directed toward their infants.
Intranasal oxytocin administered to fathers increases their engagement in infant-directed behavior, including gentle touch, vocal communication, and toy presentation. These effects suggest that exogenous oxytocin can enhance parenting behavior, though the clinical application of this finding raises ethical questions about whether pharmacological enhancement of parenting is appropriate. Research in this area continues to explore the boundaries between therapeutic application and enhancement.
Social Memory and Learning
Oxytocin plays a critical role in social memory, the ability to recognize and remember individual conspecifics. In animal models, oxytocin-knockout mice show severe impairments in social recognition that are fully restored by oxytocin administration directly into the medial amygdala. The peptide enhances the encoding and consolidation of social memories through its effects on hippocampal and amygdala circuits, making social encounters more memorable and emotionally salient.
In humans, intranasal oxytocin enhances memory for faces (particularly happy faces) while having less effect on memory for non-social stimuli like houses or landscapes. This social specificity of oxytocin's memory effects aligns with its broader role as a social salience signal: by enhancing attention to and memory for social information, oxytocin helps individuals build and maintain the mental models of their social network that are essential for navigating complex social environments.
Oxytocin also facilitates social learning, the process by which individuals acquire information from observing or interacting with others. A study published in Psychological Science demonstrated that intranasal oxytocin increased conformity to the opinions of trusted individuals while having no effect on conformity to non-trusted sources. This selective enhancement of trust-based social learning may be one mechanism by which oxytocin strengthens social bonds: by making individuals more receptive to information from trusted partners, the peptide reinforces the cognitive and behavioral patterns that maintain close relationships.
The Dark Side: When Oxytocin Doesn't Help
The social effects of oxytocin are not uniformly positive. The social salience hypothesis proposes that oxytocin increases the salience of social cues, whether those cues are positive or negative. When social cues are interpreted as safe and rewarding, oxytocin promotes approach behavior and prosociality. But when social cues are interpreted as threatening or aversive, oxytocin may intensify negative social emotions like envy, schadenfreude, and defensive hostility.
This context dependency has clinical implications. In individuals with histories of social trauma or insecure attachment, oxytocin can increase anxiety in social situations rather than reduce it. Several studies have found that oxytocin worsens trust behavior in individuals with borderline personality disorder and increases negative emotional reactivity in people with high levels of social anxiety. These paradoxical effects underscore the importance of considering individual differences and baseline social functioning when evaluating oxytocin's therapeutic potential.
The interplay between oxytocin and early life experiences is particularly relevant here. Individuals who experienced secure attachment in childhood tend to show the expected prosocial effects of oxytocin, while those with insecure or disorganized attachment may show attenuated or even reversed effects. This interaction between pharmacology and developmental history is a key reason why oxytocin trials in psychiatric populations have produced inconsistent results, and why researchers are increasingly advocating for personalized approaches to oxytocin therapy. The peptide research hub covers related topics in neuropeptide modulation of behavior.
Oxytocin in Group Dynamics and Intergroup Relations
The effects of oxytocin on group behavior reveal a more complex picture than the simple "prosocial hormone" narrative suggests. A series of experiments by De Dreu and colleagues demonstrated that intranasal oxytocin promotes ethnocentric behavior: it increases cooperation and defensive behavior toward in-group members while simultaneously increasing competitive and deceptive behavior toward outgroup members. In one study, participants who received oxytocin were more likely to sacrifice an outgroup member to save five in-group members in a moral dilemma, compared to those who received placebo.
These findings are consistent with the theory that oxytocin evolved not to promote universal cooperation but to facilitate group cohesion and survival in competitive intergroup environments. In our evolutionary past, the ability to cooperate intensely within one's own group while remaining vigilant toward rival groups would have conferred a significant survival advantage. Oxytocin, by strengthening in-group bonds and promoting defensive aggression toward perceived threats, served this dual function.
The in-group/out-group effect has been moderated by individual differences in social value orientation. Prosocially oriented individuals (those who naturally consider the welfare of others) show enhanced cooperation with both in-group and out-group members after oxytocin, while proself-oriented individuals (those who prioritize personal gain) show the expected pattern of enhanced in-group favoritism combined with increased out-group hostility. This individual difference moderation is another example of oxytocin's context-dependent effects and cautions against viewing the peptide as a simple pharmacological solution to intergroup conflict.
Oxytocin and Nonverbal Communication
Oxytocin profoundly influences how individuals process and produce nonverbal social signals. Eye-tracking studies have consistently shown that intranasal oxytocin increases gaze fixation to the eye region of faces, a pattern known as increased eye contact. This effect is particularly pronounced in individuals who typically show reduced eye contact, including those with autism spectrum disorder and social anxiety. The enhanced eye contact is thought to increase the amount of social information extracted from faces, as the eye region conveys the most reliable information about emotional state and intentions.
The peptide also enhances the processing of vocal prosody, the emotional tone conveyed by speech rhythm, pitch, and emphasis. After oxytocin administration, participants are better able to identify emotions conveyed through voice tone alone, without semantic content. This enhanced prosodic processing is mediated by increased activation in the superior temporal sulcus, a brain region specialized for processing biological sounds and social vocalizations.
Touch is another nonverbal channel modulated by oxytocin. The peptide increases the pleasantness ratings of gentle touch, particularly when the touch is delivered by a trusted partner. Oxytocin also enhances the release of endogenous opioids in response to social touch, creating a positive feedback loop in which oxytocin-enhanced touch produces opioid-mediated pleasure, which in turn promotes further social contact. This mechanism is thought to underlie the bonding effects of physical contact between romantic partners, parent-infant dyads, and close friends.
For individuals with conditions that impair nonverbal communication, including ASD, schizophrenia, and social anxiety disorder, oxytocin's effects on nonverbal processing could be therapeutically valuable. By enhancing the ability to detect and respond to nonverbal social cues, oxytocin could help bridge the communication gap that isolates these individuals from their social environment. The free assessment at FormBlends can help determine whether oxytocin therapy might be appropriate for individuals experiencing social communication difficulties.
Autism Spectrum Research
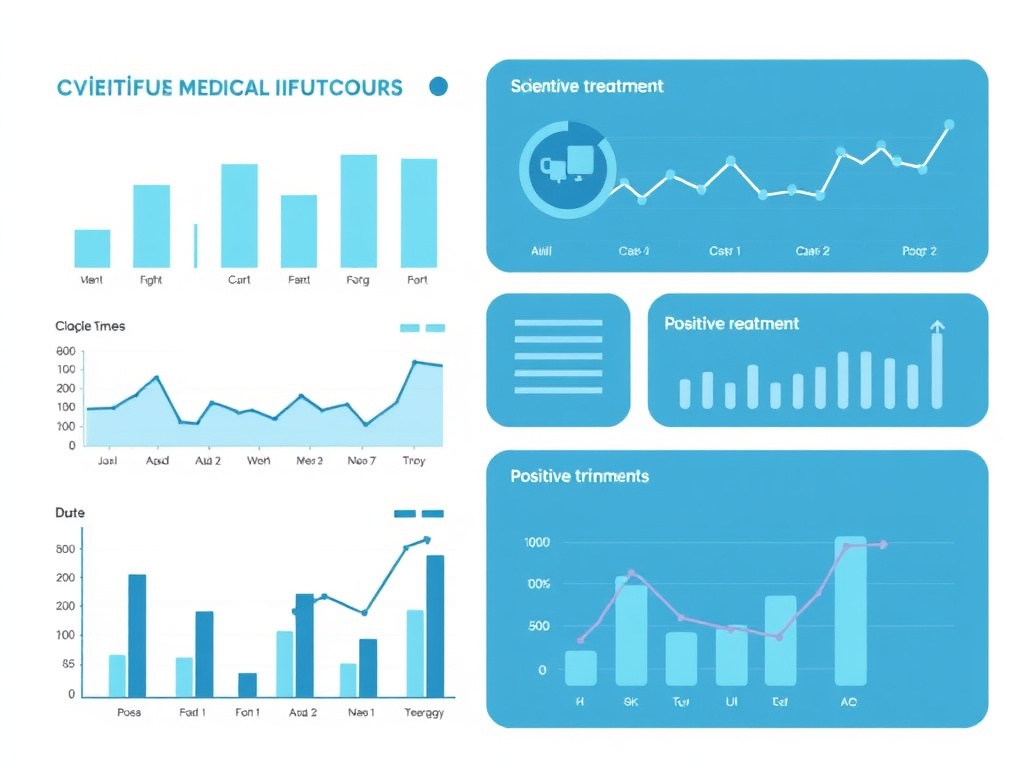
Figure 4: Oxytocin clinical trial outcomes in autism spectrum disorder research
Social Cognition Score Improvement by Dose
The Oxytocin Hypothesis in Autism
Autism spectrum disorder (ASD) is characterized by persistent deficits in social communication and interaction, along with restricted and repetitive patterns of behavior. The oxytocin hypothesis of autism proposes that abnormalities in the oxytocin system contribute to the social cognition deficits that define the condition. Multiple lines of evidence support this hypothesis. Plasma oxytocin levels are lower in children with ASD compared to typically developing controls. Genetic studies have identified associations between OXTR polymorphisms and autism susceptibility. And the OXTR gene shows increased DNA methylation in individuals with ASD, which would be expected to reduce receptor expression and dampen oxytocin signaling.
The biological plausibility of the oxytocin hypothesis is strong. The brain regions most affected in ASD, including the amygdala, fusiform gyrus, superior temporal sulcus, and medial prefrontal cortex, are all rich in oxytocin receptors. The social behaviors most impaired in ASD, including eye contact, emotional recognition, empathy, and joint attention, are all behaviors that oxytocin modulates in neurotypical individuals. And the developmental timing of ASD symptom emergence corresponds to a critical period of oxytocin system maturation in early childhood.
However, the relationship between oxytocin and autism is not simple. Not all individuals with ASD show reduced oxytocin levels, and the genetic associations between OXTR and ASD, while statistically significant at the population level, account for only a small fraction of overall ASD risk. The oxytocin hypothesis is best understood as one component of a multifactorial etiology, rather than a single causal mechanism.
Single-Dose Challenge Studies
The earliest clinical evidence for oxytocin in ASD came from single-dose challenge studies, in which participants received a single intranasal dose of oxytocin and were tested on social cognition tasks shortly afterward. These acute administration studies provided proof-of-concept evidence that exogenous oxytocin could improve social functioning in ASD.
A foundational study by Andari and colleagues (2010) administered 24 IU of intranasal oxytocin to 13 adults with high-functioning ASD and found significant improvements in social interaction during a computerized ball-tossing game. Participants who received oxytocin showed increased gaze to the eye region of faces, enhanced feelings of trust, and greater responsiveness to social cues. In a subsequent study by Guastella and colleagues (2010), 16 male adolescents with ASD received 18 or 24 IU of intranasal oxytocin and showed improved performance on the Reading the Mind in the Eyes Test, a standardized measure of emotion recognition.
These early positive findings generated enormous enthusiasm about oxytocin's therapeutic potential. But they were limited by small sample sizes, single-dose designs, and the use of highly controlled laboratory tasks that may not reflect real-world social functioning. The question of whether repeated oxytocin administration could produce sustained improvements in social behavior required longer-term trials.
Multi-Dose and Long-Term Trials
The transition from single-dose to multi-dose trials proved more challenging than anticipated. The SOAR (Study of Oxytocin in Autism to improve Reciprocity) trial, published in the New England Journal of Medicine, randomized 277 children aged 3 to 17 years with ASD to receive intranasal oxytocin or placebo for 24 weeks. The primary endpoint was change in social responsiveness as measured by the Social Responsiveness Scale (SRS-2). The results were definitively negative: oxytocin showed no significant improvement over placebo on any primary or secondary outcome measure (Simons et al., 2021, NEJM).
This large, well-powered trial was a significant setback for the field. But it raised important questions about dosing, as the trial used a relatively low dose (8 IU for children under 40 kg, 16 IU for children over 40 kg). Subsequent analyses suggested that higher doses might be necessary to produce meaningful effects, particularly in children whose endogenous oxytocin system may be more severely compromised.
A 2024 meta-analysis published in Frontiers in Psychiatry synthesized data from 12 randomized controlled trials involving 498 participants with ASD. The analysis found that, in the aggregate, intranasal oxytocin did not significantly improve social impairments compared to placebo. However, subgroup analysis revealed a critical dose-response relationship: trials using 48 IU per day showed a significant beneficial effect on social impairments (standardized mean difference = -0.57, 95% CI: -0.99 to -0.15, p < 0.01), while trials using lower doses showed no significant effect. This finding suggests that the failure of many oxytocin trials may be attributable to insufficient dosing rather than lack of efficacy.
The meta-analysis also found that oxytocin reduced repetitive behaviors at the higher dose, though this effect was less consistent than the social cognition improvement. The dose-response pattern was non-linear: 48 IU/day was effective, but 24 IU/day was not, suggesting a threshold effect rather than a gradual dose-response curve. Understanding these dosing dynamics is essential for anyone considering oxytocin therapy, and the dosing calculator can help determine appropriate starting points.
Biomarker-Guided Approaches
One of the most promising developments in oxytocin-ASD research is the identification of biomarkers that predict treatment response. A study published in Nature Communications (2024) examined the effects of 4 weeks of intranasal oxytocin (12 IU twice daily) in 79 children with ASD. Children receiving oxytocin showed significantly higher salivary oxytocin levels 24 hours after treatment, suggesting that the intervention stimulated the body's own oxytocin production system rather than simply providing exogenous hormone.
This finding has two important implications. First, it suggests that chronic oxytocin administration may upregulate the endogenous oxytocin system, potentially producing effects that persist beyond the period of active treatment. Second, it identifies salivary oxytocin response as a potential biomarker for treatment success. Children who showed the greatest increase in endogenous oxytocin production also showed the greatest behavioral improvements, suggesting that the ability to mount an endogenous oxytocin response may be a prerequisite for therapeutic benefit.
Other biomarker candidates include baseline plasma oxytocin levels, OXTR genotype, and OXTR methylation status. Preliminary evidence suggests that individuals with lower baseline oxytocin and certain OXTR polymorphisms may respond better to exogenous administration, as they have the most to gain from supplementation. Individuals with high baseline levels and favorable OXTR genetics may already have a well-functioning oxytocin system that exogenous administration cannot further enhance.
Combination Therapy Approaches
Researchers increasingly view oxytocin not as a standalone therapy for ASD but as a potential facilitator of behavioral and psychosocial interventions. The rationale is intuitive: if oxytocin enhances social motivation, social attention, and social learning, then administering the peptide before or during structured social skills training could amplify the benefits of the training.
A 2025 review in Psychopharmacology proposed that oxytocin may be most beneficial when dosed in a dynamic, as-needed (PRN) regimen timed to coincide with social learning opportunities, rather than in a fixed daily schedule. This approach would leverage oxytocin's acute effects on social salience and reward while avoiding the potential desensitization that comes with chronic, continuous dosing.
Combination studies pairing oxytocin with computer-based social cognition training have shown preliminary promise. In one study, participants who received oxytocin before social cognition training sessions showed greater improvements in emotion recognition and perspective-taking compared to those who received training with placebo. The oxytocin-enhanced group also showed better maintenance of gains at 3-month follow-up, suggesting that the peptide improved the consolidation of social learning.
This combination approach aligns with the broader trend in peptide research toward using compounds to enhance the effectiveness of existing interventions rather than as standalone treatments. Similar strategies are being explored with Semax for cognitive enhancement and Dihexa for memory consolidation, where the peptide serves as a neurobiological facilitator rather than a direct therapeutic agent.
Prader-Willi Syndrome and Related Conditions
Beyond classic ASD, oxytocin has been investigated in Prader-Willi syndrome (PWS), a genetic condition caused by the loss of function of genes on chromosome 15q11-q13. PWS is characterized by hyperphagia, obesity, intellectual disability, and social cognitive deficits that overlap with ASD. The condition is associated with reduced oxytocin-producing neurons in the hypothalamus, providing a clear biological rationale for oxytocin supplementation.
Clinical trials of intranasal oxytocin in PWS have shown improvements in social behavior, reduced temper outbursts, and improved trust in social interactions. A particularly interesting finding is that the hyperphagia (excessive appetite) characteristic of PWS was also reduced by oxytocin, suggesting that the peptide's anorectic effects are preserved even in conditions with severely dysregulated appetite. This connects the autism research to the metabolic effects discussed in the next section and highlights the potential for oxytocin to address multiple symptom domains simultaneously.
Current Consensus and Future Directions
The current scientific consensus on oxytocin for ASD can be summarized as follows: the oxytocin system is abnormal in many individuals with ASD, and exogenous oxytocin can improve social cognition under specific conditions, particularly at higher doses and when combined with social learning opportunities. However, oxytocin is not a universal treatment for ASD, and many individuals show little or no response. The field is moving toward personalized approaches that use biomarkers to identify likely responders and toward combination therapies that pair oxytocin with structured behavioral interventions.
Key unanswered questions include the optimal dose, frequency, and duration of treatment; the role of age and developmental stage in treatment response; the potential for long-term benefits or risks of chronic oxytocin administration; and the mechanisms underlying individual variability in treatment response. These questions will require large, well-designed trials with careful attention to dosing, patient selection, and outcome measurement. The peptide research hub will continue to track developments in this rapidly evolving field.
Metabolic & Appetite Effects

Figure 5: Oxytocin's metabolic effects on caloric intake, fat oxidation, and insulin sensitivity
Central Mechanisms of Appetite Regulation
Oxytocin acts as an anorectic signal in the brain, meaning it reduces food intake when administered to the central nervous system. The primary site of oxytocin's appetite-suppressing effects is the nucleus of the solitary tract (NTS) in the brainstem, where oxytocin-containing projections from the PVN terminate. Activation of oxytocin receptors in the NTS enhances satiety signaling and reduces meal size without causing nausea or aversion, distinguishing oxytocin from many other appetite suppressants that work through aversive mechanisms.
In the hypothalamus, oxytocin interacts with other appetite-regulating peptides to modulate energy balance. Oxytocin neurons in the PVN receive inhibitory inputs from the arcuate nucleus neuropeptide Y (NPY)/agouti-related peptide (AgRP) neurons (which promote feeding) and excitatory inputs from pro-opiomelanocortin (POMC) neurons (which suppress feeding). The melanocortin system, which signals through MC4 receptors, is a key activator of PVN oxytocin neurons. This means that the same pathway targeted by melanocortin-based anti-obesity drugs also activates the oxytocin system, providing a mechanistic link between these two therapeutic approaches.
Oxytocin's effects on food intake are selective. The peptide preferentially reduces the consumption of palatable, calorie-dense foods while having less effect on consumption of bland or low-calorie foods. fMRI studies have shown that intranasal oxytocin reduces activation in brain regions associated with food reward and motivation, including the ventral tegmental area and nucleus accumbens, when participants view images of high-calorie foods. At the same time, oxytocin increases activation in prefrontal regions associated with self-control and inhibitory regulation. This dual effect, dampening food reward while enhancing cognitive control, is a particularly appealing mechanism for appetite regulation.
The interaction between oxytocin and GLP-1 (glucagon-like peptide-1) signaling is an area of active investigation. Both peptides reduce food intake through partially overlapping brainstem mechanisms, and preclinical studies suggest that co-administration produces additive appetite-suppressing effects. This raises the possibility that oxytocin could complement GLP-1 receptor agonists like semaglutide or liraglutide in obesity treatment protocols, potentially allowing lower doses of each compound while maintaining or enhancing efficacy. The GLP-1 research hub covers the latest developments in incretin-based therapies.
Human Clinical Trial Data on Caloric Intake
The first demonstration that oxytocin reduces food intake in humans came from Ott and colleagues (2013), who administered 24 IU of intranasal oxytocin to 20 healthy men and measured caloric consumption at an ad libitum test meal. Oxytocin reduced total caloric intake by an average of 122 kcal compared to placebo, with the reduction driven primarily by decreased fat intake. Participants who received oxytocin ate significantly less chocolate and cookies but did not reduce their intake of crackers or bread, supporting the hypothesis that oxytocin selectively suppresses reward-driven eating.
Subsequent studies replicated and extended these findings. Lawson and colleagues (2015) showed that a single dose of 24 IU intranasal oxytocin reduced caloric intake by 100 to 150 kcal in overweight and obese men, with the effect size being larger in obese compared to normal-weight participants. This obesity-specific enhancement is clinically meaningful because it suggests that oxytocin's appetite-suppressing effects are strongest in the population that needs them most.
The largest and most rigorous caloric intake study to date was published in 2024. This randomized, double-blind, placebo-controlled trial administered intranasal oxytocin (24 IU) or placebo four times daily for 8 weeks to 61 adults with obesity (mean BMI 36.9). At the test meal assessment, oxytocin was associated with a 152 kcal reduction in caloric intake compared to placebo (-31.4 vs. +120.6 kcal change from baseline). Interestingly, participants had not taken their morning oxytocin dose before the test meal, meaning the effect was observed many hours after the last dose. This finding suggests that chronic oxytocin administration may produce lasting changes in appetite regulation that persist beyond the acute pharmacokinetic window.
Weight Loss and Body Composition
Despite the consistent evidence that oxytocin reduces caloric intake, the translation to weight loss has been disappointing. The 2024 eight-week trial found no significant difference in body weight change between oxytocin and placebo groups (0.20 vs. 0.26 kg). Similarly, there were no differences in body composition (total body fat, visceral fat, or lean mass) or resting energy expenditure between groups.
Earlier pilot studies had been more encouraging. A pilot study of 8 weeks of oxytocin treatment in adults with obesity or overweight reported weight loss of 8.9 kg (19.6 lbs) in the oxytocin group versus 4.5 kg (9.9 lbs) in the placebo group. However, this study had a smaller sample size and was not designed to definitively assess weight loss as a primary endpoint.
The disconnect between reduced caloric intake and lack of weight loss in the larger trial may be explained by several factors. First, the 152 kcal per meal reduction may be insufficient to produce measurable weight loss over 8 weeks, particularly if compensatory eating occurs at non-measured meals. Second, the four-times-daily dosing regimen may not provide sufficient coverage to suppress appetite throughout the entire day. Third, the trial did not combine oxytocin with dietary counseling, exercise programs, or other weight loss interventions that might have amplified the caloric deficit. By comparison, GLP-1 receptor agonists like semaglutide produce weight loss of 10 to 15% over 68 weeks, representing a much more potent appetite-suppressive effect.
Fat Metabolism and Insulin Sensitivity
Beyond appetite, oxytocin influences fat metabolism at the cellular level. In adipose tissue, oxytocin activates lipolysis (fat breakdown) through PKC-dependent signaling pathways. The peptide also promotes the browning of white adipose tissue, converting metabolically inactive white fat cells into thermogenically active beige fat cells that burn calories to produce heat. This browning effect has been demonstrated in both rodent models and human adipocyte cultures, and it provides a mechanism through which oxytocin could increase energy expenditure independently of its effects on food intake.
Oxytocin improves insulin sensitivity through multiple mechanisms. In skeletal muscle, the peptide enhances glucose uptake by promoting GLUT4 translocation to the cell membrane. In the liver, oxytocin reduces gluconeogenesis (glucose production) and promotes glycogen synthesis. And in pancreatic beta cells, oxytocin directly stimulates insulin secretion in a glucose-dependent manner, meaning it enhances insulin release when blood glucose is elevated but does not cause hypoglycemia when blood glucose is normal.
A single intranasal dose of 24 IU oxytocin has been shown to increase resting energy expenditure by approximately 5% and shift substrate utilization toward fat oxidation in healthy men. These acute metabolic effects are modest but potentially meaningful over extended treatment periods. Researchers have speculated that combining oxytocin's metabolic effects with the potent appetite suppression of GLP-1 agonists or the dual GIP/GLP-1 action of tirzepatide could produce complementary benefits: the GLP-1 agonist primarily driving appetite reduction while oxytocin enhances fat metabolism and insulin sensitivity.
Oxytocin and Visceral Fat
One of the most clinically relevant metabolic effects of oxytocin is its ability to reduce visceral adipose tissue (VAT), the metabolically active fat depot that surrounds the abdominal organs and is strongly associated with metabolic syndrome, type 2 diabetes, and cardiovascular disease. In a study using dual-energy X-ray absorptiometry (DEXA) and MRI, intranasal oxytocin reduced visceral fat and liver fat content without significant changes in subcutaneous fat or total body weight.
This selective reduction in visceral fat is particularly interesting because it suggests that oxytocin may improve metabolic health even without producing overall weight loss. Visceral fat is more metabolically active than subcutaneous fat and contributes disproportionately to insulin resistance, inflammation, and dyslipidemia. A therapeutic agent that reduces visceral fat while preserving lean mass would be highly valuable, even if total body weight remains unchanged. This mechanism is distinct from other metabolic peptides like AOD-9604 and Fragment 176-191, which work through different fat-mobilization pathways.
Novel Oxytocin Analogs for Obesity
One of the limitations of native oxytocin as a metabolic therapy is its short half-life (approximately 3 to 5 minutes in plasma) and poor oral bioavailability. Several research groups are developing long-acting oxytocin analogs that could provide sustained metabolic effects with less frequent dosing.
A lipidated oxytocin analog with a half-life of 7 hours (compared to 3-5 minutes for native oxytocin) showed significantly greater efficacy in reducing food intake and body weight in diet-induced obese rats. The long-acting analog produced sustained weight loss over 14 days of treatment, whereas native oxytocin showed only transient effects. This approach of extending peptide half-life through lipidation parallels the strategy used to develop long-acting GLP-1 agonists like semaglutide and liraglutide.
The gut-stable oxytocin analogs developed at the University of Queensland represent another innovation. These compounds resist degradation by gastrointestinal enzymes and can be absorbed across the intestinal epithelium, allowing oral administration. While initially developed for the treatment of chronic abdominal pain and IBS, the metabolic effects of these oral oxytocin analogs are also being evaluated. Oral delivery would dramatically simplify treatment regimens compared to intranasal administration and could improve patient compliance for chronic metabolic conditions.
Integration with Current Weight Management Approaches
Where does oxytocin fit in the current weight management landscape dominated by GLP-1 receptor agonists? The honest answer is that oxytocin alone is not a competitive weight loss therapy. The 0-1% weight loss observed in oxytocin trials pales in comparison to the 10-15% weight loss achieved with semaglutide, the 15-22% with tirzepatide, and the 24%+ projected for triple agonists like retatrutide.
However, oxytocin may have value as an adjunctive therapy that addresses aspects of metabolic health not fully covered by GLP-1 agonists. Its effects on visceral fat reduction, insulin sensitivity, fat oxidation, and emotional eating could complement the appetite-suppressive effects of GLP-1 agonists. Additionally, oxytocin's effects on social bonding and emotional well-being could help address the psychological challenges of weight management, including social eating pressures, emotional eating, and the stress associated with dietary restriction.
The GLP-1 weight loss overview provides a broader context for understanding where different therapeutic options fit within comprehensive weight management strategies. For individuals seeking a multi-modal approach to metabolic health, combining oxytocin's metabolic and social benefits with the potent appetite suppression of GLP-1-based therapies represents an evidence-based strategy worth discussing with a healthcare provider.
Oxytocin and the Brain-Gut Axis
The metabolic effects of oxytocin cannot be fully understood without considering the brain-gut axis, the bidirectional communication network between the central nervous system and the gastrointestinal tract. Oxytocin acts at multiple levels of this axis: centrally, through brainstem satiety centers; peripherally, through oxytocin receptors on enteric neurons and enterocytes; and at the level of the gut microbiome, through indirect effects on intestinal motility and epithelial barrier function.
In the brainstem, oxytocin neurons from the PVN project to the dorsal vagal complex (DVC), which includes the nucleus of the solitary tract (NTS) and the dorsal motor nucleus of the vagus (DMV). These projections modulate vagal efferent output to the gut, influencing gastric motility, gastric acid secretion, and the release of gut hormones including GLP-1, PYY, and CCK. By enhancing the sensitivity of brainstem satiety centers to peripheral satiety signals, oxytocin amplifies the satiating effects of meals and promotes earlier meal termination.
At the gut level, oxytocin receptors are expressed on enteric neurons throughout the myenteric and submucosal plexuses. Activation of these receptors modulates intestinal transit time, with some studies showing that oxytocin accelerates gastric emptying while slowing small intestinal transit. This differential effect could promote a feeling of fullness (from rapid gastric distension) while maximizing nutrient absorption. The complex effects of oxytocin on gut motility are relevant for gastrointestinal conditions like gastroparesis and functional dyspepsia, where disordered motility contributes to symptoms.
The anti-inflammatory properties of oxytocin in the gut are also being investigated. Oxytocin reduces the production of pro-inflammatory cytokines (TNF-alpha, IL-6, IL-1beta) in intestinal tissue and enhances epithelial barrier integrity by promoting tight junction protein expression. These effects suggest potential therapeutic applications in inflammatory bowel disease (IBD) and other conditions characterized by intestinal inflammation and barrier dysfunction. The gut-stable oxytocin analogs from the University of Queensland research group are specifically being developed to exploit these gut-level effects.
For individuals using Larazotide for gut barrier support or BPC-157 for gastrointestinal healing, oxytocin's gut effects represent a complementary mechanism that targets inflammation and motility rather than tight junction regulation or tissue repair. The biohacking hub covers additional approaches to optimizing gut health and metabolic function.
Oxytocin, Brown Fat, and Thermogenesis
One of the most intriguing metabolic effects of oxytocin is its ability to promote the activation and recruitment of brown adipose tissue (BAT). Unlike white adipose tissue (WAT), which stores energy, brown fat burns calories to generate heat through a process called non-shivering thermogenesis. This process is mediated by uncoupling protein 1 (UCP1), which dissipates the mitochondrial proton gradient as heat rather than using it to produce ATP.
Animal studies have demonstrated that chronic oxytocin administration increases BAT mass, UCP1 expression, and core body temperature in diet-induced obese mice. The effect is mediated by both central mechanisms (activation of sympathetic outflow to BAT through brainstem circuits) and direct peripheral effects (oxytocin receptors on brown adipocytes promote UCP1 transcription). The magnitude of the thermogenic effect is modest, estimated at a 3 to 5% increase in resting energy expenditure, but when sustained over weeks to months, this could contribute meaningfully to negative energy balance.
Oxytocin also promotes the "browning" of white adipose tissue, a process in which white fat cells acquire brown-fat-like characteristics including UCP1 expression and increased mitochondrial density. These "beige" or "brite" (brown-in-white) adipocytes are metabolically active and contribute to adaptive thermogenesis. The browning effect of oxytocin is particularly pronounced in subcutaneous adipose tissue and is enhanced by cold exposure, suggesting that combining oxytocin with cold thermogenesis protocols could produce additive metabolic benefits.
The thermogenic effects of oxytocin distinguish it from most other appetite-suppressing therapies, which reduce energy intake without directly increasing energy expenditure. The combination of appetite suppression and enhanced thermogenesis makes oxytocin mechanistically unique among metabolic peptides. Other peptides with thermogenic properties include 5-Amino-1MQ, which inhibits NNMT to increase cellular energy expenditure, and MOTS-c, which activates AMPK to promote mitochondrial biogenesis and fat oxidation.
Diabetes and Glycemic Control
The effects of oxytocin on glucose metabolism extend beyond insulin sensitivity to include direct effects on pancreatic beta cell function, hepatic glucose output, and skeletal muscle glucose uptake. In the pancreas, oxytocin receptors are expressed on both alpha cells (glucagon-producing) and beta cells (insulin-producing). Oxytocin stimulates insulin secretion from beta cells in a glucose-dependent manner, meaning it enhances insulin release when blood glucose is elevated but does not promote insulin secretion at normal glucose levels. This glucose-dependent mechanism reduces the risk of hypoglycemia, a significant advantage over insulin secretagogues like sulfonylureas.
In the liver, oxytocin suppresses gluconeogenesis through activation of the PI3K/Akt signaling pathway, reducing hepatic glucose output. This effect complements the insulin-sensitizing effects of the peptide in skeletal muscle, where oxytocin promotes GLUT4 translocation and glucose uptake. The net result is improved whole-body glucose disposal and reduced fasting blood glucose levels.
Animal studies in diabetic models have shown that chronic oxytocin administration reduces HbA1c by 0.5 to 1.0%, improves glucose tolerance, and protects pancreatic beta cells from glucotoxicity-induced apoptosis. These glycemic effects are in addition to the weight-related benefits, suggesting that oxytocin addresses diabetes through multiple complementary pathways. However, human clinical trial data on glycemic outcomes with oxytocin are limited, and more research is needed before oxytocin can be considered a viable adjunct to established diabetes therapies like semaglutide or tirzepatide.
Cardiovascular Metabolic Benefits
Oxytocin's metabolic effects extend to the cardiovascular system, where the peptide produces several beneficial effects. Oxytocin lowers blood pressure through vasodilation mediated by nitric oxide (NO) release from endothelial cells. The peptide also reduces oxidative stress in vascular tissue, inhibits the formation of atherosclerotic plaques, and promotes the release of atrial natriuretic peptide (ANP) from cardiac atria, which contributes to sodium and water excretion.
In animal models of metabolic syndrome, chronic oxytocin administration reduces total cholesterol, LDL cholesterol, and triglycerides while increasing HDL cholesterol. These lipid-modifying effects complement the glucose and body composition improvements, creating a comprehensive cardiovascular risk reduction profile. The cardioprotective effects of oxytocin are mediated by both central mechanisms (reduced sympathetic nervous system activation, improved vagal tone) and direct peripheral effects (vascular relaxation, anti-inflammatory signaling in the arterial wall).
For individuals with metabolic syndrome or cardiovascular risk factors, oxytocin's multi-system metabolic benefits make it an interesting candidate for combination therapy. However, it must be emphasized that the cardiovascular benefits have been demonstrated primarily in animal models, and large-scale human cardiovascular outcome trials with oxytocin have not been conducted. The evidence base is promising but preliminary, and clinical decisions should be based on currently available human data.
Psychiatric Applications

Figure 6: Clinical trial outcomes for oxytocin in psychiatric disorders
Schizophrenia: Social Cognition and Negative Symptoms
Schizophrenia is characterized by positive symptoms (hallucinations, delusions), negative symptoms (social withdrawal, flat affect, anhedonia), and cognitive deficits. Current antipsychotic medications primarily target positive symptoms through dopamine D2 receptor blockade, leaving negative symptoms and cognitive impairments largely untreated. Because oxytocin modulates social cognition, trust, and emotional processing, researchers have hypothesized that it could address the social cognitive deficits and negative symptoms that are among the most disabling features of schizophrenia.
Biological evidence supports this hypothesis. Plasma oxytocin levels in people with schizophrenia are lower than in healthy controls, and the degree of reduction correlates with the severity of negative symptoms. Genetic studies have identified associations between OXTR polymorphisms and schizophrenia risk. And post-mortem brain studies have found reduced oxytocin receptor expression in the medial prefrontal cortex of individuals with schizophrenia, a region critical for social cognition and theory of mind.
Clinical trial results have been decidedly mixed. Early small-scale studies were encouraging. A pilot six-week randomized controlled trial found that oxytocin improved fear recognition, perspective taking, and a composite social cognition score compared to baseline in the treatment group, with a concurrent reduction in PANSS (Positive and Negative Syndrome Scale) negative symptom scores. A 12-week trial in people with schizophrenia found significant within-group reductions in negative symptoms and between-group improvements in the schizophrenia subgroup.
However, larger and more rigorously designed trials have been less positive. A randomized clinical trial comparing oxytocin to galantamine for negative symptoms and cognitive impairments in schizophrenia found no significant group differences for negative symptoms. Another study reported that oxytocin had no significant effect on social cognition measures compared to placebo. And a 24-week trial combining oxytocin with cognitive behavioral social skills training (CBSST) found no significant treatment group differences in social functioning, positive symptoms, negative symptoms, or social beliefs.
A meta-analysis of ten randomized controlled trials of intranasal oxytocin versus placebo over 2 to 16 weeks found no significant benefit for total, positive, or negative symptoms overall. However, a subgroup analysis revealed that higher doses (80 IU/day) showed greater improvements in total and positive symptoms relative to placebo, echoing the dose-response pattern seen in the autism literature. This finding suggests that many schizophrenia trials may have been underdosed.
Post-Traumatic Stress Disorder (PTSD)
PTSD is characterized by intrusive re-experiencing of traumatic events, avoidance of trauma-related stimuli, negative alterations in cognition and mood, and hyperarousal. Oxytocin's potential in PTSD stems from its anxiolytic properties, its effects on fear extinction learning, and its ability to modulate amygdala reactivity to threatening stimuli.
The first application of oxytocin therapy for PTSD was reported in Vietnam War veterans, where the peptide was observed to subjectively reduce the re-experiencing of traumatic memories. Subsequent preclinical studies demonstrated that oxytocin enhances fear extinction learning in animal models, meaning that animals treated with oxytocin more rapidly learn that a previously threatening stimulus is no longer dangerous. This extinction-enhancing effect is mediated by oxytocin receptors in the central amygdala and infralimbic cortex, regions that are critical for extinction memory formation.
In human studies, intranasal oxytocin has been shown to reduce startle responses to conditioned fear stimuli and to enhance the retention of extinction memories when administered before exposure-based therapy sessions. These findings suggest that oxytocin could serve as an adjunct to exposure therapy, the gold-standard psychological treatment for PTSD, by facilitating the learning processes that underlie therapeutic benefit.
However, the evidence is not universally positive. Some studies have found that while oxytocin reduces certain PTSD symptoms, the overall clinical effect is not statistically significant. Others have reported that oxytocin can increase emotional reactivity to trauma-related cues in some patients, particularly those with more severe symptoms or comorbid conditions. The current consensus is that oxytocin may be most beneficial for PTSD when used in combination with psychotherapy, where its prosocial and anxiolytic effects can enhance the therapeutic alliance and facilitate emotional processing. The relationship between oxytocin and stress resilience parallels research on other neuroprotective peptides like Selank and Semax, which also modulate anxiety and stress responses through different neurochemical pathways.
Anxiety Disorders
Oxytocin's anxiolytic properties have been studied across several anxiety disorder subtypes, including social anxiety disorder (SAD), generalized anxiety disorder (GAD), and specific phobias. The strongest evidence exists for social anxiety disorder, where the peptide's ability to reduce social threat perception and enhance social approach motivation is most directly relevant to the core pathology.
In a study of 147 patients with anxiety disorders, anxiety symptoms decreased after 5 days of low-dose oxytocin administration. This finding is notable because it suggests that chronic, low-dose oxytocin may be more effective for anxiety than single-dose protocols. The mechanism likely involves sustained modulation of the HPA (hypothalamic-pituitary-adrenal) axis stress response, as oxytocin is a known inhibitor of cortisol release and can dampen the physiological stress response over repeated administrations.
In social anxiety disorder specifically, intranasal oxytocin reduces negative self-referential processing, a cognitive pattern in which individuals with SAD interpret ambiguous social information as reflecting poorly on themselves. Oxytocin also increases willingness to engage in social exposure exercises and reduces the distress associated with social performance situations. These effects suggest that oxytocin could enhance the effectiveness of cognitive behavioral therapy (CBT) for social anxiety by reducing the avoidance behavior that limits treatment engagement.
A counterintuitive finding is that oxytocin can increase anxiety in some individuals, particularly those with high baseline anxiety and insecure attachment styles. This paradoxical effect is consistent with the social salience hypothesis: in individuals who are hypervigilant to social threats, increasing social salience through oxytocin may amplify rather than reduce anxiety. This finding underscores the importance of careful patient selection and highlights the need for personalized approaches to oxytocin therapy in anxiety disorders.
Depression and Postpartum Depression
The relationship between oxytocin and depression is bidirectional. Reduced oxytocin levels are consistently observed in individuals with major depressive disorder (MDD), and the degree of reduction correlates with symptom severity, particularly for social withdrawal and anhedonia. Conversely, positive social interactions that increase endogenous oxytocin release (such as physical touch, social support, and breastfeeding) are protective against depression and can enhance treatment response.
Postpartum depression (PPD) represents a particularly promising target for oxytocin therapy because of the peptide's well-established role in maternal behavior. Low oxytocin levels during pregnancy predict the development of PPD, and women with PPD show reduced oxytocin release during breastfeeding compared to non-depressed mothers. Intranasal oxytocin has been shown to increase positive maternal behaviors (eye contact, affectionate touch, vocalizations) and reduce the negative cognitions about parenting that characterize PPD.
However, clinical trial evidence for oxytocin as a standalone antidepressant is limited. Small open-label studies have reported improvements in depression scores following oxytocin administration, but large, placebo-controlled trials are lacking. The current view is that oxytocin is unlikely to replace established antidepressant medications but may serve as a useful adjunct, particularly for depression subtypes characterized by social withdrawal, reduced bonding (as in PPD), or co-occurring anxiety.
Substance Use Disorders
Emerging research suggests that oxytocin may have therapeutic potential for substance use disorders, including alcohol dependence, opioid use disorder, and stimulant use disorder. The rationale is based on the overlap between the neural circuits that mediate social bonding and those that mediate drug reward: both involve dopamine signaling in the nucleus accumbens and the mesocorticolimbic pathway.
In animal models, oxytocin reduces self-administration of alcohol, cocaine, and methamphetamine. The mechanism appears to involve direct modulation of dopamine release in the nucleus accumbens: by reducing the dopamine surge associated with drug intake, oxytocin diminishes the rewarding properties of the substance without producing the anhedonia associated with dopamine receptor blockade.
In human studies, intranasal oxytocin has been shown to reduce cue-induced craving for alcohol and to reduce alcohol consumption in social drinking situations. A small randomized trial found that oxytocin reduced withdrawal symptoms and prevented relapse in alcohol-dependent individuals. Larger trials are needed to confirm these findings and to determine whether oxytocin can be integrated into existing substance use treatment protocols.
The addiction research highlights an intriguing parallel with NAD+ therapy, which has also been investigated for substance use recovery through different biochemical mechanisms. Both approaches aim to restore neurochemical balance in reward circuits that have been disrupted by chronic substance use.
Obsessive-Compulsive Disorder
Oxytocin has been investigated as a potential treatment for obsessive-compulsive disorder (OCD), a condition characterized by intrusive thoughts (obsessions) and repetitive behaviors (compulsions). The rationale for oxytocin in OCD is based on the observation that OCD symptoms often have a social component (contamination fears, checking behaviors related to harming others) and that the oxytocin system modulates the cortico-striatal-thalamic circuit that is dysfunctional in OCD.
Early case studies and small open-label trials reported improvements in OCD symptoms following intranasal oxytocin administration. In one study, patients with treatment-resistant OCD who received 160 IU of intranasal oxytocin daily for two weeks showed a 36% reduction in Yale-Brown Obsessive Compulsive Scale (Y-BOCS) scores. However, these results have not been consistently replicated in controlled trials, and the evidence remains preliminary.
An interesting finding from the OCD literature is that oxytocin may worsen symptoms in some patients, particularly those with contamination-type OCD. This paradoxical worsening is consistent with the social salience hypothesis: if oxytocin increases attention to social cues and social concerns, it could amplify the contamination fears that drive cleaning and washing compulsions. This finding reinforces the importance of symptom subtyping when evaluating oxytocin's therapeutic potential in psychiatric conditions.
Borderline Personality Disorder
Borderline personality disorder (BPD) is characterized by unstable relationships, emotional dysregulation, impulsivity, and fear of abandonment. The oxytocin system is implicated in BPD through the attachment disruptions and early relational trauma that are commonly associated with the disorder. Individuals with BPD show lower plasma oxytocin levels compared to healthy controls, and OXTR methylation is increased in those with more severe symptoms.
Clinical studies of oxytocin in BPD have produced concerning results. In one study, intranasal oxytocin actually increased feelings of distrust and decreased cooperation in BPD patients, the opposite of the effects seen in healthy controls. In another study, oxytocin increased emotional sensitivity to negative social feedback in BPD patients, potentially worsening the emotional instability that characterizes the disorder. These findings suggest that in individuals with severely disrupted attachment systems, oxytocin may amplify pathological social processes rather than normalize them.
The BPD findings carry a broader lesson for oxytocin therapeutics: the peptide's effects are critically dependent on the individual's baseline social-emotional functioning and attachment history. In conditions where the social processing system is fundamentally altered (as in BPD), exogenous oxytocin does not simply restore normal social function. Instead, it amplifies whatever social processing patterns already exist, whether adaptive or maladaptive. This understanding has shifted the field away from viewing oxytocin as a "social normalizer" and toward a more nuanced appreciation of its role as a social signal amplifier.
Eating Disorders
Anorexia nervosa (AN) and bulimia nervosa (BN) involve disruptions in both social functioning and appetite regulation, making them potential targets for oxytocin therapy. Individuals with AN show reduced plasma oxytocin levels that correlate with the severity of body image disturbance and social isolation. In BN, oxytocin levels are also reduced, and the reduction is associated with the frequency of binge-purge episodes.
Intranasal oxytocin has been shown to reduce attentional bias toward food and body-related stimuli in women with AN and to reduce negative self-referential processing in both AN and BN. These cognitive effects could complement the nutritional rehabilitation and psychotherapy that form the core of eating disorder treatment. However, clinical trials of oxytocin for eating disorders are still in early stages, and the evidence is insufficient to support routine clinical use. The lifestyle hub covers broader topics in nutrition and mental health.
Overall Assessment of Psychiatric Applications
A meta-analysis across psychiatric conditions found a combined effect size of d = 0.32 (N = 304, 95% CI: 0.18 to 0.47, p < 0.01) for intranasal oxytocin compared to placebo. However, only autism spectrum disorder showed a significant individual effect size (d = 0.57, N = 68, p < 0.01). The other conditions showed trends toward improvement that did not reach statistical significance, likely due to small sample sizes, heterogeneous patient populations, and variable dosing protocols.
The field is moving toward several important refinements: personalized dosing based on baseline oxytocin levels and OXTR genetics; combination therapy approaches that pair oxytocin with established psychotherapeutic interventions; stratification of patients by attachment style, trauma history, and social functioning to identify likely responders; and the development of longer-acting oxytocin analogs that could provide more sustained therapeutic effects. These refinements will be essential for determining whether oxytocin can fulfill its therapeutic promise in psychiatric medicine.
Intranasal Delivery

Figure 7: Intranasal delivery pharmacokinetics and dosing protocol overview
Pharmacokinetics of Intranasal Oxytocin
Intranasal delivery is the primary route of administration for therapeutic oxytocin in research and clinical settings. The nasal mucosa provides a unique pathway to the brain that bypasses the blood-brain barrier, which would otherwise prevent the large, polar oxytocin molecule from reaching its central nervous system targets. Two anatomical routes mediate nose-to-brain transport: the olfactory nerve pathway (through the cribriform plate to the olfactory bulb and forebrain) and the trigeminal nerve pathway (through branches of the trigeminal nerve to the brainstem and other brain regions).
Studies using radiolabeled oxytocin in nonhuman primates have provided direct evidence that intranasally administered oxytocin reaches the cerebrospinal fluid (CSF) and several brain regions. When labeled oxytocin was delivered intranasally, significant concentrations were detected in the amygdala, striatum, and brainstem within 60 minutes. When the same labeled oxytocin was delivered intravenously, no significant brain penetration was observed. This finding confirms that nose-to-brain transport is the primary mechanism by which intranasal oxytocin reaches its central targets, rather than indirect effects mediated by peripheral oxytocin crossing the blood-brain barrier.
The nasal bioavailability of oxytocin is low, estimated at approximately 2% of the administered dose. This means that a 24 IU intranasal dose delivers only about 0.48 IU to the systemic circulation, with the remainder being either transported directly to the brain, retained in the nasal mucosa, or cleared through mucociliary drainage. Despite the low systemic bioavailability, plasma oxytocin levels rise significantly after intranasal administration, typically peaking within 15 to 30 minutes and returning to baseline within 75 to 90 minutes. Some individuals show elevated plasma levels persisting up to 150 minutes post-dose, reflecting substantial inter-individual variability in absorption and clearance kinetics.
A 2025 pharmacokinetic study comparing intravenous and intranasal oxytocin in nonpregnant adults confirmed the low bioavailability of intranasal delivery but also demonstrated that the plasma concentration profile of intranasal oxytocin differs qualitatively from intravenous administration. Intranasal delivery produces a more gradual rise and sustained plateau in plasma levels compared to the sharp spike and rapid decline seen with intravenous dosing. This sustained profile may be advantageous for therapeutic applications where prolonged receptor activation is desired.
Dosing Protocols in Research and Clinical Practice
The most commonly used dose in oxytocin research is 24 IU (international units), typically administered as three sprays per nostril from a standard metered-dose nasal spray device. Each spray delivers approximately 4 IU, and the total dose is divided equally between nostrils to maximize contact with the nasal mucosa on both sides. This dose is based on the original trust game studies and has become the default in much of the subsequent research.
However, the optimal dose varies by indication and individual. For autism spectrum disorder, the 2024 meta-analysis found that 48 IU/day (typically split into two 24 IU doses) was necessary to produce significant improvements in social cognition, while lower doses were ineffective. For appetite suppression, single doses of 24 IU have shown consistent effects on caloric intake. For anxiety reduction, lower doses (8 to 16 IU) administered over multiple days may be sufficient, as the anxiolytic effects appear to accumulate with repeated dosing.
| Indication | Typical Dose | Frequency | Duration | Evidence Level |
|---|---|---|---|---|
| Social cognition (ASD) | 24-48 IU | Once or twice daily | 4-24 weeks | Moderate (dose-dependent) |
| Appetite suppression | 24 IU | Four times daily | 8 weeks | Moderate |
| Anxiety reduction | 8-24 IU | Once or twice daily | 5+ days | Limited |
| Social skills enhancement | 24 IU | Before training sessions | As needed | Preliminary |
| Schizophrenia (negative symptoms) | 40-80 IU | Twice daily | 6-16 weeks | Mixed |
| PTSD (adjunct to therapy) | 24-40 IU | Before therapy sessions | As needed | Preliminary |
The dosing calculator can help determine personalized starting doses based on body weight and the specific application. For those interested in oxytocin therapy, beginning with lower doses and titrating upward based on response and tolerability is generally recommended.
Administration Technique
Proper administration technique is critical for achieving consistent drug delivery with intranasal oxytocin. The nasal spray should be primed before first use by pumping the actuator until a fine mist is produced. Before each use, patients should gently blow their nose to clear the nasal passages of mucus that could impede absorption. The head should be tilted slightly forward (not backward), and the spray should be directed toward the lateral wall of the nasal cavity rather than the nasal septum to maximize contact with the olfactory mucosa.
Timing considerations are important. For social cognition effects, the peak behavioral effects of oxytocin occur approximately 45 to 60 minutes after intranasal administration, based on the time course of fMRI activation changes. This means that for applications like enhancing social skills training or psychotherapy sessions, the dose should be administered 30 to 60 minutes before the therapeutic activity begins. For appetite suppression, dosing approximately 30 minutes before meals produces the most reliable effects on caloric intake.
Storage requirements for oxytocin nasal spray vary by formulation but generally involve refrigeration at 2 to 8 degrees Celsius. Oxytocin is susceptible to degradation by heat, light, and oxidation, and improperly stored solutions may lose potency without visible changes in appearance. Some compounding pharmacies provide oxytocin in amber glass vials to protect against photodegradation, and patients should be counseled to discard unused solution after the manufacturer's recommended beyond-use date.
Alternative Delivery Routes
While intranasal delivery dominates the research landscape, several alternative routes are being explored. Sublingual administration of oxytocin has shown promise in small studies, with some evidence that it produces comparable plasma levels to intranasal delivery. Sublingual tablets or drops could offer advantages in terms of ease of use and dosing precision, though the extent of nose-to-brain transport via this route is uncertain.
Intravenous (IV) oxytocin is used extensively in obstetric settings for labor induction and postpartum hemorrhage prevention but is generally not used for psychiatric or metabolic applications because it does not effectively cross the blood-brain barrier. The IV route produces high plasma levels that primarily affect peripheral oxytocin receptors in the uterus, cardiovascular system, and kidneys.
The oral oxytocin analogs developed by the University of Queensland research group represent the most significant innovation in oxytocin delivery technology. These gut-stable compounds resist enzymatic degradation in the gastrointestinal tract and can activate oxytocin receptors in the gut wall, providing direct therapeutic effects for conditions like IBS and chronic abdominal pain. Whether oral analogs can achieve sufficient brain penetration for psychiatric and social applications remains to be determined.
Subcutaneous injection of oxytocin is another option being explored, particularly for applications requiring predictable pharmacokinetics and higher bioavailability. This route is familiar to users of other peptide therapies like BPC-157 or growth hormone secretagogues like CJC-1295/Ipamorelin. However, subcutaneous oxytocin produces high peripheral levels that may cause unwanted effects on uterine contractility in women of reproductive age.
Measurement of Oxytocin Levels
Accurate measurement of oxytocin in biological fluids remains a significant challenge for the field. Immunoassay-based methods (radioimmunoassay and enzyme immunoassay) are widely used but suffer from cross-reactivity with oxytocin fragments, vasopressin, and other structurally similar peptides. Different assay kits can produce results that vary by orders of magnitude, making it difficult to compare values across studies or establish normal reference ranges.
Mass spectrometry-based methods (LC-MS/MS) offer greater specificity but require more sophisticated equipment and expertise. These methods can distinguish intact oxytocin from fragments and metabolites with high precision, though they are less sensitive than immunoassays and may miss low concentrations of oxytocin in CSF or saliva.
A 2022 review in Molecular Psychiatry proposed standardized measurement protocols to address these challenges, including recommendations for sample collection (ice-cold tubes, immediate processing), extraction methods (solid-phase extraction to concentrate the sample and remove interfering substances), and assay validation (including tests for parallelism, recovery, and cross-reactivity). These standardized methods will be essential for advancing the field, as inconsistent measurement techniques have contributed to the variability in clinical trial outcomes and the difficulty of establishing dose-response relationships.
For monitoring treatment response, urine oxytocin measurements may offer advantages over plasma measurements. Studies have found larger effect sizes and clearer differentiation between oxytocin and placebo groups when using urine rather than plasma as the measurement matrix. Urine collection is also less invasive and less affected by the pulsatile release pattern that creates large fluctuations in plasma oxytocin levels.
Factors Affecting Intranasal Absorption
The variability in clinical trial outcomes with intranasal oxytocin may be partly attributable to individual differences in nasal absorption. Several anatomical and physiological factors influence the amount of oxytocin that reaches the brain after intranasal administration. Nasal mucosa thickness varies between individuals, with thicker mucosa potentially reducing absorption rates. Mucociliary clearance rate, the speed at which the mucus blanket moves particles from the nasal cavity to the throat, determines how long the oxytocin solution remains in contact with the absorptive epithelium. Individuals with rapid mucociliary clearance may absorb less oxytocin than those with slower clearance.
Nasal pathology can significantly affect absorption. Chronic rhinitis, nasal polyps, deviated nasal septum, and mucosal inflammation all alter the nasal epithelial surface available for drug absorption. Participants with active allergic rhinitis show reduced absorption of intranasally administered drugs, and this effect is particularly pronounced during allergy season. Clinical trials rarely screen for or control these nasal factors, which could contribute to the heterogeneity of treatment responses.
The formulation of the nasal spray also affects absorption. Most clinical trials use a simple aqueous solution of oxytocin preserved with chlorobutanol. However, formulation additives can dramatically alter nasal absorption. Permeation enhancers like chitosan, cyclodextrins, or bile salts increase transmucosal transport by temporarily loosening tight junctions between epithelial cells. Mucoadhesive polymers extend the contact time between the solution and the nasal mucosa. And nanoparticle formulations can protect oxytocin from enzymatic degradation in the nasal cavity while promoting cellular uptake.
A particularly promising formulation approach involves liposomal encapsulation of oxytocin. Liposomes are spherical vesicles composed of phospholipid bilayers that can encapsulate hydrophilic peptides in their aqueous core. Liposomal oxytocin formulations have shown enhanced nasal absorption, prolonged brain exposure, and improved behavioral effects in preclinical studies compared to simple aqueous solutions. This technology could address one of the key limitations of current intranasal oxytocin therapy: the variability in drug delivery that leads to inconsistent clinical outcomes.
Comparisons with Other Intranasal Peptide Therapies
Oxytocin is not the only peptide delivered intranasally for central nervous system effects. Several other neuropeptides use the same delivery route, and comparing their pharmacokinetic profiles provides useful context for understanding oxytocin's characteristics. Selank nasal spray delivers the anxiolytic heptapeptide Selank through the nasal mucosa, achieving brain penetration through similar olfactory and trigeminal nerve pathways. NAD+ nasal spray uses the intranasal route to deliver nicotinamide adenine dinucleotide for cognitive and energy support.
Desmopressin (DDAVP), a synthetic vasopressin analog, is the most well-established intranasal peptide therapy. Used for diabetes insipidus and nocturnal enuresis, desmopressin has an intranasal bioavailability of 3 to 5%, somewhat higher than oxytocin's estimated 2%. The higher bioavailability may reflect desmopressin's resistance to enzymatic degradation (it contains D-arginine rather than L-arginine, making it resistant to aminopeptidases) and its longer plasma half-life (3 hours versus 3-5 minutes for oxytocin).
Insulin is another peptide that has been extensively studied via the intranasal route. Intranasal insulin does not significantly affect peripheral glucose levels (because the dose reaching the bloodstream is too small) but does produce central nervous system effects on memory, appetite, and metabolic regulation. The intranasal insulin literature provides an important precedent for oxytocin research: it demonstrates that small peptides delivered intranasally can produce clinically meaningful central effects at doses that have negligible peripheral bioavailability. This principle supports the hypothesis that oxytocin's behavioral effects are mediated primarily by nose-to-brain transport rather than by peripheral oxytocin reaching the brain through the bloodstream.
Practical Reconstitution and Storage Guidelines
For those obtaining oxytocin from compounding pharmacies, understanding proper handling is essential for maintaining potency. Lyophilized (freeze-dried) oxytocin powder should be stored at -20 degrees Celsius until reconstitution. Reconstitution is typically performed with sterile bacteriostatic water or normal saline, using aseptic technique to prevent microbial contamination.
Once reconstituted, the oxytocin solution should be stored refrigerated at 2 to 8 degrees Celsius and used within the beyond-use date specified by the compounding pharmacy, typically 30 to 90 days depending on the preservative system used. The solution should be protected from light (amber glass vials are preferred) and should not be frozen once reconstituted, as freezing and thawing can cause aggregation and loss of potency.
Before each use, the nasal spray bottle should be gently inverted several times to ensure uniform drug concentration. Vigorous shaking should be avoided as it can cause foaming and introduce air bubbles that affect spray delivery. The priming mechanism should be checked periodically: if the spray produces a stream rather than a fine mist, the nozzle may be partially blocked and should be cleaned according to the manufacturer's instructions.
Patients who travel frequently should be aware that oxytocin nasal spray should be kept cool during transport. A small insulated bag with a cool pack is sufficient for day trips, but for longer travel, the solution should be refrigerated at the destination. Airport security typically allows nasal spray medications through security checkpoints when declared as medical liquids, though a prescription label on the bottle is recommended.
Safety Profile

Figure 8: Safety profile of intranasal oxytocin across clinical research settings
Intranasal Oxytocin Safety Data
The safety profile of intranasal oxytocin is one of the most reassuring aspects of the peptide's therapeutic development. A comprehensive review of safety data published in Psychoneuroendocrinology concluded that intranasal oxytocin at doses of 18 to 40 IU produces no detectable subjective changes in recipients, no reliable side effects, and no adverse outcomes when administered for short-term use in controlled research settings. This assessment was based on data from dozens of clinical studies involving thousands of participants.
In longer-term studies, particularly the ASD trials, the adverse event profile remains favorable. A systematic review and meta-analysis of reported adverse events in long-term intranasal oxytocin treatment for ASD found that the most common adverse events were nasal discomfort or irritation (14.3%), tiredness or drowsiness (7.2%), irritability (9.0%), diarrhea (4.5%), and mild skin irritation (4.5%). These adverse events were generally mild, self-limiting, and comparable in frequency to those reported in placebo groups.
A 2025 systematic review specifically examined the safety of intranasal oxytocin in older adults, a population that was previously underrepresented in oxytocin research. The review found no significant safety concerns in older adult populations, though the authors noted that the total number of older participants studied remains relatively small and that additional data are needed, particularly regarding cardiovascular safety in elderly individuals with pre-existing heart disease.
Cardiovascular Considerations
Oxytocin has complex cardiovascular effects that depend on the dose, route of administration, and the individual's baseline cardiovascular status. At therapeutic intranasal doses, oxytocin produces mild vasodilation through nitric oxide-dependent mechanisms, resulting in a modest decrease in blood pressure (typically 3 to 8 mmHg systolic) and a slight increase in heart rate. These effects are generally clinically insignificant in healthy individuals.
However, at higher doses or with intravenous administration, oxytocin can produce more significant hemodynamic effects. The peptide's structural similarity to vasopressin means that at high concentrations, it can activate vasopressin V1a receptors, producing vasoconstriction and potentially raising blood pressure. This biphasic dose-response (vasodilation at low doses, vasoconstriction at high doses) is clinically relevant for obstetric use but is unlikely to be encountered at the intranasal doses used for psychiatric and metabolic applications.
Cardiac arrhythmias have been reported with high-dose intravenous oxytocin, including QT prolongation, sinus tachycardia, and ST-segment changes. These effects are mediated by a direct action of oxytocin on cardiac ion channels and are dose-dependent. At intranasal doses of 24 to 48 IU, no significant ECG changes have been reported in clinical trials. Nonetheless, individuals with pre-existing cardiac conditions, particularly long QT syndrome or history of arrhythmias, should be monitored when starting oxytocin therapy.
Water Balance and Hyponatremia
One of the most important safety considerations with oxytocin is its antidiuretic effect. Because of its structural similarity to vasopressin (antidiuretic hormone), oxytocin can activate vasopressin V2 receptors in the kidney at high concentrations, reducing urine output and promoting water retention. When high-dose intravenous oxytocin is administered with large volumes of hypotonic fluids (as sometimes occurs in obstetric settings), the result can be water intoxication and severe hyponatremia (low blood sodium), which can cause seizures, coma, and death.
At the intranasal doses used for psychiatric and metabolic applications (18 to 48 IU), the antidiuretic effect is minimal. The amount of oxytocin that reaches the systemic circulation after intranasal administration is far below the threshold for clinically significant water retention. Nevertheless, patients should be advised to maintain normal fluid intake and avoid excessive water consumption during oxytocin therapy. Electrolyte monitoring is recommended for treatment durations exceeding 4 to 6 weeks, particularly in individuals taking other medications that affect water balance (e.g., diuretics, SSRIs).
Reproductive Safety
Oxytocin is a potent uterotonic agent, meaning it stimulates uterine smooth muscle contraction. This property is the basis for its obstetric use in labor induction and postpartum hemorrhage prevention, but it also represents a safety concern for premenopausal women using oxytocin for non-obstetric indications. At therapeutic intranasal doses, the uterotonic effect is expected to be minimal, but the theoretical risk of uterine cramps, menstrual irregularities, or, in pregnant women, preterm labor cannot be completely excluded.
Women who are pregnant or planning to become pregnant should not use exogenous oxytocin outside of supervised obstetric settings. Women of reproductive age using intranasal oxytocin for psychiatric or metabolic indications should use reliable contraception and be counseled about the theoretical reproductive risks. These precautions are consistent with the standard approach to peptide therapies in reproductive-age women.
In men, oxytocin has been shown to facilitate erection and ejaculation through central and spinal mechanisms. Exogenous oxytocin has been associated with increased ejaculate volume and improved sexual satisfaction in some studies. These effects are generally considered beneficial rather than adverse, but they represent a pharmacological action that patients should be aware of. For those interested in peptides that affect sexual function, PT-141 and Kisspeptin-10 target related but distinct pathways.
Drug Interactions
Oxytocin has relatively few known drug interactions, but several are clinically relevant. The combination of oxytocin with prostaglandin analogs (misoprostol, dinoprostone) can produce combined uterotonic effects, increasing the risk of uterine hyperstimulation. This interaction is primarily relevant in obstetric settings but should be considered in women of reproductive age using intranasal oxytocin.
SSRIs and SNRIs may potentiate oxytocin's effects on social behavior and mood. Serotonin facilitates oxytocin release from hypothalamic neurons, and SSRI-treated individuals may have higher endogenous oxytocin levels at baseline. Adding exogenous oxytocin in this context could produce enhanced prosocial effects but also increases the theoretical risk of serotonin-related side effects. Patients taking SSRIs should start oxytocin at the lower end of the dosing range and titrate slowly.
The interaction between oxytocin and alcohol deserves mention. Oxytocin attenuates some of the subjective effects of alcohol intoxication and reduces alcohol craving. While this is potentially therapeutic for alcohol use disorder, it could also lead individuals to underestimate their level of intoxication, creating safety risks related to driving or other activities requiring coordination and judgment.
Long-Term Safety Considerations
The long-term safety of intranasal oxytocin is not yet fully characterized. Most clinical trials have durations of 4 to 24 weeks, and data on outcomes beyond 6 months are limited. Theoretical concerns about long-term use include receptor desensitization (downregulation of OXTR in response to chronic exogenous stimulation), endogenous oxytocin suppression (similar to the feedback suppression seen with chronic exogenous hormone administration), and effects on the developing brain in children and adolescents.
Animal studies provide some reassurance: chronic oxytocin administration in adult rodents does not produce significant adverse effects on behavior, brain structure, or reproductive function over treatment periods of several months. However, neonatal oxytocin exposure in rodents has been associated with changes in oxytocin receptor expression and social behavior in adulthood, raising concerns about the use of exogenous oxytocin in very young children.
The question of receptor desensitization is particularly relevant. Studies in pregnant women have shown that prolonged oxytocin exposure leads to OXTR downregulation in the myometrium, which is one reason why oxytocin augmentation becomes less effective over time during labor. Whether similar desensitization occurs in the brain during chronic intranasal administration is unknown, but some researchers have suggested that intermittent dosing (e.g., dosing before specific social activities rather than on a fixed daily schedule) could minimize this risk.
Special Populations
Several population groups require special consideration when evaluating oxytocin safety. Children and adolescents with ASD represent the largest target population for therapeutic oxytocin, but the long-term effects of exogenous oxytocin on the developing brain are not well understood. The ASD trials in children (ages 3-17) have not reported serious adverse events, but follow-up periods have been limited to 6 months or less.
Older adults are increasingly being studied as potential beneficiaries of oxytocin therapy, particularly for social isolation and cognitive decline. The 2025 systematic review of intranasal oxytocin in older adults found no significant safety concerns, but the number of older participants studied to date is small. Age-related changes in nasal mucosa integrity, renal function, and cardiovascular reserve could all affect the safety profile of intranasal oxytocin in this population.
Individuals with kidney disease may be at increased risk for the antidiuretic effects of oxytocin, as impaired renal function reduces the clearance of both oxytocin and water. Dose adjustments may be necessary, and more frequent electrolyte monitoring is recommended. Similarly, individuals with liver disease may show altered oxytocin metabolism, though the clinical significance of this is unclear.
| Adverse Event | Incidence (Intranasal) | Severity | Management |
|---|---|---|---|
| Nasal discomfort/irritation | 14.3% | Mild | Saline nasal rinse; alternate nostrils |
| Irritability | 9.0% | Mild to moderate | Dose reduction; timing adjustment |
| Tiredness/drowsiness | 7.2% | Mild | Evening dosing; dose reduction |
| Diarrhea | 4.5% | Mild | Self-limiting; symptomatic management |
| Skin irritation | 4.5% | Mild | Self-limiting |
| Headache | 3-5% | Mild | OTC analgesics; dose reduction |
| Blood pressure changes | 2-3% | Mild | Monitoring; dose adjustment |
Clinical Safety Summary
Intranasal oxytocin at doses of 18 to 48 IU has a favorable safety profile supported by hundreds of clinical studies. The most common adverse events are mild nasal discomfort and fatigue. Serious adverse events are rare and primarily associated with high-dose intravenous administration in obstetric settings. Long-term safety data beyond 6 months are limited. Patients with cardiac disease, kidney disease, or pregnancy require special monitoring. The free assessment can help determine whether oxytocin therapy is appropriate for individual situations.
Contraindications and Precautions
Several absolute and relative contraindications should be considered before initiating oxytocin therapy. Pregnancy is an absolute contraindication for non-obstetric oxytocin use due to the risk of premature uterine contractions. Hypersensitivity to oxytocin or any excipients in the formulation (such as chlorobutanol preservative) is another absolute contraindication, though true anaphylactic reactions to oxytocin are extremely rare.
Relative contraindications include significant cardiovascular disease (particularly arrhythmias and hypertrophic cardiomyopathy), severe renal impairment (creatinine clearance below 30 mL/min), and concurrent use of prostaglandin analogs. Individuals with a history of nasal surgery, significant nasal obstruction, or chronic sinusitis may have altered nasal absorption and should be monitored for adequate therapeutic response.
Psychiatric precautions are equally important. As discussed throughout this report, oxytocin can paradoxically worsen symptoms in individuals with borderline personality disorder, severe social anxiety with insecure attachment, and contamination-type OCD. A thorough psychiatric history should be obtained before initiating oxytocin therapy for any psychiatric indication, with particular attention to attachment style, trauma history, and current symptom profile.
Age-related precautions apply to both pediatric and geriatric populations. In children under 3 years of age, the long-term effects of exogenous oxytocin on brain development are unknown, and use should be restricted to supervised clinical trials. In older adults, particular attention should be paid to cardiovascular monitoring and electrolyte balance, given the higher prevalence of cardiac disease and renal impairment in this population.
Monitoring Recommendations
For clinical use of intranasal oxytocin, the following monitoring schedule is recommended based on current evidence and expert consensus. At baseline, patients should undergo a complete blood count, comprehensive metabolic panel (including sodium and potassium), cardiovascular assessment (blood pressure, heart rate, and ECG for patients with cardiac risk factors), and a psychiatric evaluation including assessment of attachment style and trauma history.
During the first month of therapy, blood pressure and heart rate should be measured weekly, with particular attention to orthostatic hypotension (a drop in blood pressure upon standing). Serum sodium should be checked at 2 weeks and 4 weeks to screen for hyponatremia. Patients should be counseled to report symptoms of water retention (edema, rapid weight gain, confusion) immediately.
For maintenance therapy beyond one month, monitoring can be reduced to monthly blood pressure checks and quarterly metabolic panels. Patients using oxytocin for psychiatric indications should receive regular psychiatric assessments to evaluate treatment response and screen for paradoxical symptom worsening. The frequency of psychiatric follow-up depends on the indication and the patient's clinical stability, but quarterly assessments are a reasonable minimum.
Individuals using oxytocin as part of a multi-peptide protocol should receive additional monitoring appropriate to the other peptides in use. For example, those combining oxytocin with growth hormone secretagogues like CJC-1295/Ipamorelin should have IGF-1 levels monitored, while those combining oxytocin with metabolic peptides like Tesofensine should have cardiovascular monitoring intensified.
Comparison with Other Peptide Safety Profiles
Placing oxytocin's safety profile in the context of other therapeutic peptides provides useful perspective. Among the neuropeptides, oxytocin has one of the most extensively studied safety profiles. The peptide has been used clinically for over 60 years in obstetric settings, providing a substantial body of safety data, though much of this data pertains to intravenous rather than intranasal administration.
Compared to other intranasally administered peptides, oxytocin has a comparable or superior safety profile. Selank, another anxiolytic neuropeptide, has an excellent safety record with no significant adverse events in clinical studies. Semax, a neuroprotective peptide derived from ACTH, similarly shows minimal side effects at therapeutic doses. The favorable safety profiles of these neuropeptides reflect the general principle that small peptides administered at physiological doses tend to produce fewer off-target effects than small-molecule drugs, because their receptor specificity limits their activity to well-defined signaling pathways.
Compared to the GLP-1 receptor agonists that dominate the metabolic peptide space, oxytocin has a milder side effect profile. Semaglutide and tirzepatide commonly cause nausea (20-44%), vomiting (5-15%), and diarrhea (10-20%), particularly during dose titration. These gastrointestinal side effects can be significant enough to cause treatment discontinuation in 5 to 10% of patients. Oxytocin, by comparison, causes nausea in less than 5% of intranasal users and GI symptoms in less than 5%. This milder side effect profile could be advantageous for patients who cannot tolerate GLP-1 agonists, though oxytocin's metabolic efficacy is substantially lower.
Important Safety Reminders
- Do not use intranasal oxytocin during pregnancy without obstetric supervision
- Monitor serum sodium levels during the first month of therapy
- Report symptoms of water retention (swelling, confusion, rapid weight gain) immediately
- Individuals with borderline personality disorder or severe social anxiety should be carefully evaluated before starting oxytocin
- Inform all healthcare providers that you are using oxytocin before any surgical or dental procedures
- Store oxytocin solution refrigerated at 2-8 degrees Celsius and discard after the beyond-use date
Oxytocin and Pain Modulation
One of the most promising and underappreciated areas of oxytocin research is its role in pain perception. Chronic pain affects roughly 20% of adults worldwide, and current treatment options often carry significant risks, from opioid dependence to gastrointestinal bleeding from long-term NSAID use. Oxytocin offers an entirely different approach to pain management, one that works through endogenous neural circuits rather than simply masking signals at the receptor level.
The analgesic properties of oxytocin have been recognized since the late 1990s, but the mechanistic understanding has deepened considerably in recent years. Oxytocin-producing neurons in the paraventricular nucleus of the hypothalamus project directly to the spinal cord dorsal horn, where they modulate ascending pain signals. When these neurons release oxytocin locally within the spinal cord, it activates OXTR receptors on GABAergic interneurons, which in turn inhibit pain transmission neurons. This creates a top-down inhibitory pathway that functions somewhat like the brain's own volume control for pain.
But spinal modulation is only part of the story. Oxytocin also acts in the periaqueductal gray (PAG), a brainstem region that serves as a critical hub for descending pain inhibition. The PAG integrates signals from the cortex, amygdala, and hypothalamus to determine how much pain reaches conscious awareness. Oxytocin enhances the inhibitory output from the PAG, effectively raising the threshold for pain perception. It's worth noting that this same PAG-mediated mechanism is involved in the pain-reducing effects of social support and placebo analgesia, which suggests oxytocin may be one of the key molecules behind the well-documented phenomenon that social connection reduces pain.
Inflammatory and Neuropathic Pain Models
Animal studies have demonstrated oxytocin's efficacy across multiple pain models. In inflammatory pain models using carrageenan or complete Freund's adjuvant, both systemic and intrathecal oxytocin significantly reduce mechanical allodynia and thermal hyperalgesia. The effective doses in these studies were within the physiological range, suggesting that endogenous oxytocin release during positive social interactions genuinely contributes to pain relief rather than requiring pharmacological doses.
Neuropathic pain models have shown equally encouraging results. In the chronic constriction injury model, which mimics nerve compression injuries, oxytocin administration reduced pain behaviors by 40-60% in most studies. And in chemotherapy-induced peripheral neuropathy models, which are particularly challenging to treat in clinical practice, intranasal oxytocin attenuated mechanical sensitivity and improved functional measures of nerve health. This finding has practical relevance, since chemotherapy-induced neuropathy affects up to 70% of cancer patients and has limited treatment options.
The interaction between oxytocin and the endogenous opioid system adds another layer of complexity. Oxytocin receptors and mu-opioid receptors are co-expressed in several brain regions, and oxytocin has been shown to potentiate the analgesic effects of endogenous opioids while simultaneously reducing the rewarding properties of exogenous opioids. This dual action makes oxytocin particularly interesting in the context of the opioid crisis, as it could theoretically enhance pain relief while reducing addiction risk.
Clinical Pain Studies
Human clinical trials have begun testing intranasal oxytocin for various pain conditions. A 2022 randomized controlled trial in patients with chronic low back pain found that 40 IU intranasal oxytocin reduced pain intensity by approximately 30% compared to placebo over a four-week treatment period. The effect size was modest but clinically meaningful, and the side effect profile was far more favorable than standard pharmacotherapy.
In headache and migraine research, intranasal oxytocin has shown mixed but promising results. A crossover trial found that oxytocin reduced headache severity in patients with chronic daily headaches, with the strongest effects seen in patients who also reported high levels of social isolation, suggesting the analgesic and social effects may be complementary. Migraine studies have been less consistent, possibly because migraine pathophysiology involves specific trigeminovascular mechanisms that may not be as responsive to oxytocin modulation.
Perhaps the most intriguing clinical data comes from fibromyalgia research. Fibromyalgia, characterized by widespread pain, fatigue, and cognitive dysfunction, has been linked to altered oxytocin signaling. Patients with fibromyalgia consistently show lower baseline oxytocin levels compared to healthy controls, and the degree of oxytocin deficiency correlates with pain severity. Several small trials of intranasal oxytocin in fibromyalgia have shown improvements in pain, fatigue, and emotional well-being, though larger confirmatory studies are needed.
For individuals exploring complementary approaches to pain management, peptides like BPC-157 and TB-500 address tissue repair and inflammatory mechanisms that often underlie chronic pain conditions. The peptide research hub provides a broader overview of the pain-related research across various peptide families.
Oxytocin and Cardiovascular Health
The heart is one of the richest sources of oxytocin receptors outside the brain, and this anatomical fact has driven a growing body of research into oxytocin's cardiovascular effects. What's emerged is a picture of oxytocin as a cardioprotective molecule that influences blood pressure regulation, cardiac remodeling, and vascular health through multiple distinct mechanisms.
Blood Pressure and Vascular Effects
Oxytocin's blood pressure-lowering effect has been observed in both animal models and human studies. The mechanism involves stimulation of atrial natriuretic peptide (ANP) release from the heart, enhanced nitric oxide production in the vascular endothelium, and direct relaxation of vascular smooth muscle. In healthy volunteers, intranasal oxytocin consistently produces mild reductions in both systolic and diastolic blood pressure, typically in the range of 5-10 mmHg. These reductions are modest but meaningful, particularly for individuals with borderline hypertension.
The vascular protective effects extend beyond simple blood pressure reduction. Oxytocin promotes endothelial nitric oxide synthase (eNOS) activity, which improves endothelial function and reduces vascular stiffness. In animal models of atherosclerosis, chronic oxytocin administration reduced plaque formation, decreased vascular inflammation, and improved arterial compliance. The anti-inflammatory mechanism appears to involve suppression of NF-kB signaling in vascular endothelial cells, which reduces the expression of adhesion molecules that recruit inflammatory cells to vessel walls.
Cardiac Regeneration Research
One of the most striking findings in recent oxytocin cardiovascular research is its ability to stimulate cardiac regeneration. A 2022 study in mice demonstrated that oxytocin activates a population of cardiac progenitor cells (epicardium-derived cells) that can differentiate into functional cardiomyocytes. When oxytocin was administered after experimental myocardial infarction, the treated animals showed increased cardiomyocyte regeneration in the border zone of the infarct and improved cardiac function compared to controls.
This regenerative effect appears to be mediated through the Hippo-YAP signaling pathway, which controls organ size and cellular proliferation. Oxytocin receptor activation in epicardial cells leads to YAP nuclear translocation, which triggers a gene expression program that promotes cell proliferation and differentiation. The adult mammalian heart was long considered to be a terminally differentiated organ with no regenerative capacity, so the discovery that oxytocin can activate dormant cardiac progenitor cells has generated significant excitement.
It's important to keep this regenerative finding in context. The mouse studies used doses that are substantially higher than what's achieved with intranasal administration in humans, and it remains unclear whether the same mechanism operates in human cardiac tissue. But the principle, that a naturally occurring peptide hormone can stimulate cardiac regeneration, opens new avenues for therapeutic development.
Stress-Related Cardiac Protection
The connection between chronic psychological stress and cardiovascular disease is well established, and oxytocin may be one of the key mediators linking social support to cardiac protection. Chronic stress activates the hypothalamic-pituitary-adrenal (HPA) axis and sympathetic nervous system, leading to sustained elevations in cortisol and catecholamines that damage the heart and blood vessels over time. Oxytocin directly opposes this stress response by inhibiting HPA axis activity and reducing sympathetic nervous system output.
In animal models of stress-induced cardiomyopathy (which mimics the human condition known as Takotsubo cardiomyopathy or "broken heart syndrome"), oxytocin pre-treatment prevented the catecholamine-mediated cardiac damage. Treated animals maintained normal cardiac contractility and wall motion even under extreme stress conditions that caused significant cardiac dysfunction in controls.
These cardiovascular findings create interesting connections with other peptide research areas. SS-31 targets mitochondrial function in cardiac tissue, while Humanin and MOTS-c address cellular stress responses that affect cardiovascular health. The biohacking hub covers emerging approaches to cardiovascular optimization, and the free assessment tool can help identify which peptides may be most relevant to your specific health goals.
Oxytocin and the Gut-Brain Axis
The gut-brain axis has become one of the most active areas in biomedical research, and oxytocin sits at a fascinating intersection of this bidirectional communication system. The gastrointestinal tract contains oxytocin receptors throughout the enteric nervous system, and recent work has shown that gut bacteria can actually influence oxytocin production in the brain, creating a feedback loop between the microbiome and social behavior.
Microbiome-Oxytocin Interactions
A landmark series of studies from MIT demonstrated that supplementation with the probiotic bacterium Lactobacillus reuteri increases circulating oxytocin levels in mice and produces behavioral effects consistent with enhanced oxytocin signaling, including increased social behavior, accelerated wound healing, and improved coat quality. The mechanism appears to involve vagal nerve stimulation by bacterial metabolites, which triggers oxytocin release from the paraventricular nucleus. When the vagus nerve was severed, the probiotic's effects on oxytocin and social behavior were abolished.
These findings have been partially replicated in humans. Small clinical trials have shown that L. reuteri supplementation increases peripheral oxytocin levels and improves measures of social cognition in healthy volunteers and in individuals with autism spectrum disorder. The magnitude of the oxytocin increase is modest, typically around 20-30% above baseline, but it's sustained throughout the supplementation period and is accompanied by measurable behavioral changes.
The practical implications are worth considering. If specific probiotic strains can enhance endogenous oxytocin production through the vagal pathway, this could provide a non-pharmacological approach to boosting oxytocin that works from the bottom up rather than relying on intranasal delivery. And it adds another dimension to the growing evidence that gut health influences mental health and social behavior through specific, identifiable molecular pathways.
Gastric Protection and Gut Motility
Beyond the microbiome connection, oxytocin has direct effects on gastrointestinal function. Oxytocin receptors are expressed on gastric epithelial cells, intestinal smooth muscle, and enteric neurons. Activation of these receptors stimulates gastric mucus production, reduces gastric acid secretion, and modulates intestinal motility.
In animal models of gastric ulceration, oxytocin administration provided significant gastroprotection, reducing ulcer size and severity by up to 60% in some studies. The protective mechanism involves enhanced prostaglandin E2 production in the gastric mucosa and increased mucosal blood flow. Interestingly, this gastroprotective effect of oxytocin mirrors the well-known gastroprotective properties of another peptide, BPC-157, which was originally isolated from gastric juice and acts through partially overlapping pathways.
The effects on gut motility are more complex. In the upper GI tract, oxytocin tends to slow gastric emptying and reduce motility, which may contribute to the nausea some individuals experience with higher oxytocin doses. In the lower GI tract, oxytocin promotes colonic motility and may have prokinetic effects. This differential response along the GI tract suggests that oxytocin receptors in different regions of the gut are coupled to different intracellular signaling cascades.
Implications for Metabolic Health
The gut-brain axis effects of oxytocin connect directly to its metabolic actions. Oxytocin's ability to reduce appetite and food intake is partly mediated through vagal afferents that carry satiety signals from the gut to the brainstem. By enhancing the sensitivity of these vagal pathways, oxytocin may amplify the post-meal satiety signal and reduce the tendency to overeat.
This creates an interesting parallel with GLP-1 receptor agonists like semaglutide and tirzepatide, which also reduce appetite partly through vagal mechanisms. Whether oxytocin could complement GLP-1 therapy by acting through distinct but convergent appetite-suppression pathways is a question that warrants investigation. The two systems target different receptor populations and different aspects of feeding behavior, oxytocin primarily affecting social and hedonic eating while GLP-1 agonists primarily affect homeostatic satiety, so the combination could theoretically provide broader appetite control than either agent alone.
For a deeper look at how metabolic peptides interact with appetite regulation and weight management, the GLP-1 hub covers the latest research, and the dosing calculator can help determine starting points for weight management protocols.
Oxytocin in Wound Healing and Tissue Repair
A less publicized but increasingly well-documented property of oxytocin is its ability to accelerate wound healing and tissue regeneration. The connection was first noticed in epidemiological studies showing that socially isolated individuals heal more slowly from wounds than those with strong social networks. Since oxytocin is the primary hormonal mediator of social bonding, researchers hypothesized that it might also mediate the social facilitation of wound healing.
Cellular Mechanisms of Oxytocin-Mediated Healing
The wound healing effects of oxytocin operate through several well-defined cellular pathways. First, oxytocin stimulates the proliferation of dermal fibroblasts, the cells responsible for producing collagen and rebuilding the extracellular matrix after tissue injury. In cell culture experiments, oxytocin treatment increased fibroblast proliferation rates by 30-50% compared to untreated controls, with the effect being dose-dependent and mediated through the OXTR receptor.
Second, oxytocin promotes angiogenesis in healing tissue. New blood vessel formation is essential for delivering oxygen and nutrients to the wound bed, and inadequate angiogenesis is a major contributor to chronic non-healing wounds. Oxytocin enhances VEGF expression in wound tissue and stimulates endothelial cell migration and tube formation in vitro. The angiogenic effect is particularly strong in the inflammatory phase of wound healing, suggesting that oxytocin accelerates the transition from inflammation to proliferation.
Third, oxytocin modulates the inflammatory response in a way that favors resolution over chronicity. Excessive or prolonged inflammation is one of the primary reasons wounds become chronic, especially in diabetic patients and the elderly. Oxytocin reduces pro-inflammatory cytokines (IL-6, TNF-alpha) while promoting anti-inflammatory mediators (IL-10) at the wound site, shifting the balance toward productive healing rather than destructive inflammation.
Preclinical and Clinical Evidence
In a well-designed study using a standardized blister wound model in humans, participants who received intranasal oxytocin before wounding showed faster wound closure compared to placebo-treated controls. The oxytocin group achieved complete wound closure approximately two days earlier on average, which translates to roughly a 20-25% acceleration of the healing process. Cytokine analysis of wound fluid showed that oxytocin-treated participants had lower levels of IL-6 and higher levels of IL-10 at the wound site, confirming the anti-inflammatory mechanism.
Animal studies have extended these findings to more severe injuries. In full-thickness skin wound models in rats, topical application of oxytocin to the wound bed accelerated closure, increased tensile strength of healed tissue, and improved collagen organization. In diabetic wound models, which simulate the impaired healing commonly seen in diabetes, oxytocin treatment partially restored normal healing kinetics, though the improvement was less pronounced than in non-diabetic animals.
Burn wound research has shown similarly positive results. Oxytocin administration reduced the depth of burn injury progression (the phenomenon where burns deepen in the hours and days after the initial thermal insult) and improved vascularization of the healing burn wound. These effects were associated with reduced oxidative stress markers in the burn tissue, suggesting that oxytocin's antioxidant properties contribute to its protective effect in thermal injuries.
Muscle Regeneration and Sarcopenia
Beyond skin wounds, oxytocin has demonstrated regenerative effects in skeletal muscle. A landmark study published in Nature Communications found that oxytocin levels decline with age and that this decline contributes to age-related muscle wasting (sarcopenia). In aged mice, oxytocin administration activated muscle stem cells (satellite cells) and restored muscle regenerative capacity to levels approaching those of young animals.
The mechanism involves activation of the MAPK/ERK signaling pathway in satellite cells, which promotes their exit from quiescence, proliferation, and differentiation into new muscle fibers. Aged satellite cells are largely quiescent and resistant to activation stimuli, contributing to the poor muscle regeneration seen in elderly individuals after injury. Oxytocin appears to overcome this activation barrier by providing an additional proliferative signal through the OXTR receptor.
This muscle regeneration finding has practical implications for aging populations. Sarcopenia affects approximately 10-16% of the elderly worldwide and contributes to falls, fractures, loss of independence, and increased mortality. If oxytocin can enhance muscle regenerative capacity in humans as it does in mice, it could become part of a comprehensive approach to maintaining functional capacity with age. Combined with resistance exercise, adequate protein intake, and other regenerative peptides like TB-500 and BPC-157, oxytocin might contribute to preservation of muscle mass and function well into advanced age. The peptide research hub covers the emerging science behind these regenerative compounds.
Bone Remodeling and Osteoporosis Prevention
The skeletal system represents yet another tissue where oxytocin exerts direct biological effects. Osteoblasts (the cells that build new bone) express functional oxytocin receptors, and receptor activation stimulates osteoblast differentiation, proliferation, and mineralization activity. At the same time, oxytocin inhibits osteoclast formation and bone resorption, creating a net anabolic effect on the skeleton. In ovariectomized mouse models of postmenopausal osteoporosis, oxytocin administration preserved bone mineral density and improved trabecular bone architecture, with effects comparable to low-dose estrogen replacement.
The clinical relevance of these findings is underscored by human observational data. Women with higher circulating oxytocin levels have been shown to have greater bone mineral density at the hip and lumbar spine compared to women with lower levels, even after controlling for estrogen, age, and body mass index. Breastfeeding, which triggers sustained oxytocin release, is associated with preserved or enhanced bone density in the years following lactation, despite the calcium demands of milk production. This apparent paradox, that calcium loss through milk should weaken bones but breastfeeding mothers actually maintain stronger skeletons, may be partly explained by the bone-anabolic effects of the oxytocin released during nursing.
Men also appear to benefit from oxytocin's skeletal effects. Testosterone, the primary bone-protective hormone in men, stimulates hypothalamic oxytocin production, creating a hormonal cascade where testosterone's bone benefits are partly mediated through oxytocin. Age-related declines in both testosterone and oxytocin may therefore contribute in a complementary manner to the bone loss seen in elderly men. Restoring oxytocin levels through intranasal supplementation could complement testosterone optimization strategies for male skeletal health.
The potential for oxytocin to serve as an osteoporosis treatment is particularly attractive because of its favorable side effect profile compared to existing options. Bisphosphonates can cause osteonecrosis of the jaw and atypical femoral fractures with long-term use. Denosumab carries a risk of rebound bone loss upon discontinuation. Teriparatide is limited to two years of treatment and requires daily injections. Intranasal oxytocin, if confirmed to have skeletal benefits in human clinical trials, could offer a safer long-term option for bone protection, especially for individuals who also benefit from its cardiovascular, metabolic, and neuropsychiatric effects. The convergence of multiple health benefits in a single naturally occurring peptide is what makes oxytocin research so compelling across medical disciplines.
Oxytocin and Immune Function
The immune system is another area where oxytocin research has produced unexpected findings. Oxytocin receptors are expressed on multiple immune cell types, including T lymphocytes, macrophages, and thymic epithelial cells. The thymus, which is the primary organ responsible for T cell maturation, actually produces its own oxytocin, using it as a local signaling molecule to guide the development of immune cells that will eventually defend the body against pathogens and cancerous cells.
In macrophages, oxytocin shifts the polarization balance from the pro-inflammatory M1 phenotype toward the anti-inflammatory M2 phenotype. M1 macrophages are essential for acute pathogen defense but contribute to tissue damage when chronically activated. M2 macrophages promote tissue repair, resolution of inflammation, and immune homeostasis. By favoring M2 polarization, oxytocin may help resolve chronic low-grade inflammation, the type associated with aging (sometimes called "inflammaging"), obesity, and metabolic syndrome.
T cell function is also modulated by oxytocin. In vitro studies show that oxytocin enhances regulatory T cell (Treg) differentiation and suppressive function. Tregs are the immune system's peacekeepers, preventing autoimmune responses and excessive inflammation. Deficient Treg function contributes to autoimmune diseases, allergies, and the chronic inflammation associated with obesity. If oxytocin can enhance Treg activity in vivo, it could have therapeutic applications in autoimmune conditions and inflammatory metabolic disease.
The thymic connection is particularly intriguing in the context of aging. The thymus undergoes progressive involution (shrinkage) with age, producing fewer new T cells and contributing to the decline in immune function seen in elderly populations. Oxytocin's role in thymic T cell maturation raises the question of whether declining oxytocin levels with age contribute to thymic involution and immune senescence. Some researchers have speculated that oxytocin supplementation could slow thymic aging, though this hypothesis remains largely untested. For those interested in immune-supporting peptides, Thymosin Alpha-1 directly targets thymic function, while LL-37 provides antimicrobial and immunomodulatory effects. The peptide hub covers the full spectrum of immune-active peptides currently under investigation.
Oxytocin and Gut Health: The Enteric Nervous System Connection
An expanding body of research has identified the gastrointestinal tract as a significant site of oxytocin production and action, challenging the traditional view of oxytocin as primarily a brain and reproductive hormone. The enteric nervous system, often called the "second brain," contains an estimated 500 million neurons that regulate gut motility, secretion, blood flow, and immune function largely independently of the central nervous system. Oxytocin receptors are expressed throughout the gastrointestinal tract, including on enteric neurons, smooth muscle cells, epithelial cells, and gut-associated immune cells, suggesting that oxytocin plays a broader role in gastrointestinal physiology than previously recognized.
Several lines of evidence support a functional role for oxytocin in gut health. In animal models of inflammatory bowel disease, oxytocin administration reduced mucosal inflammation, decreased pro-inflammatory cytokine production, and accelerated epithelial wound healing. These effects appear to be mediated through oxytocin receptor activation on enteric immune cells, which shifts the local immune response toward an anti-inflammatory profile. In a rat model of colitis, systemic oxytocin treatment reduced disease activity scores by approximately 40% compared to vehicle-treated controls, with histological analysis showing preserved mucosal architecture and reduced neutrophil infiltration in treated animals.
The gut-brain axis provides another pathway through which oxytocin influences gastrointestinal function. Vagal afferent neurons, which carry sensory information from the gut to the brainstem, express oxytocin receptors and respond to circulating oxytocin by modulating their signaling patterns. This vagal modulation affects gastric motility, acid secretion, and the perception of visceral pain. Patients with functional gastrointestinal disorders such as irritable bowel syndrome (IBS) often show alterations in vagal tone and oxytocin levels, raising the question of whether oxytocin supplementation could address the visceral hypersensitivity that characterizes these conditions. A small clinical study found that intranasal oxytocin reduced visceral pain perception in IBS patients during rectal balloon distension testing, though larger trials are needed to confirm this finding.
The microbiome connection adds yet another dimension. Certain gut bacteria, particularly Lactobacillus reuteri strains, have been shown to stimulate endogenous oxytocin production through vagal nerve stimulation. In mouse studies, oral administration of L. reuteri increased circulating oxytocin levels and produced downstream effects including improved wound healing, reduced adiposity, and enhanced social behavior. This finding suggests a bidirectional relationship where gut bacteria influence oxytocin production, and oxytocin in turn modulates the gut environment in ways that affect bacterial composition. For researchers interested in exploring oxytocin's gastrointestinal effects, the enteric nervous system represents a fertile area of investigation, and complementary gut-active peptides like BPC-157 may provide complementary benefits through their own mechanisms of mucosal protection and anti-inflammatory action.
The stress-gut-oxytocin axis adds yet another clinical dimension. Psychological stress is a well-established trigger for gastrointestinal symptoms, and the mechanism involves cortisol-mediated suppression of oxytocin release combined with sympathetic nervous system activation that disrupts normal gut motility patterns. Chronic stress states can create a self-reinforcing cycle where reduced oxytocin leads to increased gut inflammation and permeability, which generates peripheral inflammatory signals that further suppress central oxytocin production. Breaking this cycle through exogenous oxytocin administration could theoretically benefit patients with stress-related gastrointestinal disorders, though clinical trials specifically testing this hypothesis in human populations with functional GI disorders remain in early stages. The integration of gut health research with oxytocin neuroscience represents one of the most promising interdisciplinary frontiers in peptide science, with implications for conditions ranging from inflammatory bowel disease to functional dyspepsia to the gastrointestinal manifestations of anxiety and depression.
Frequently Asked Questions
What does oxytocin do in the body?
Oxytocin is a nine-amino-acid peptide hormone produced in the hypothalamus that serves multiple functions throughout the body. Its most well-known roles are in stimulating uterine contractions during labor and triggering milk ejection during breastfeeding. But oxytocin also functions as a neuromodulator in the brain, where it enhances social bonding, trust, empathy, and emotional recognition. Beyond social behavior, oxytocin reduces appetite and caloric intake, promotes fat metabolism and insulin sensitivity, lowers blood pressure through vasodilation, supports bone formation, and modulates pain perception. The peptide acts through the oxytocin receptor (OXTR), a G protein-coupled receptor found in the brain, uterus, heart, kidneys, pancreas, adipose tissue, and gastrointestinal tract.
Can oxytocin help with autism spectrum disorder?
The evidence for oxytocin in autism spectrum disorder (ASD) is mixed but shows promise at specific doses. A 2024 meta-analysis of 12 randomized controlled trials with 498 participants found that daily doses of 48 IU produced significant improvements in social cognition and reduced repetitive behaviors, while lower doses did not outperform placebo. The large SOAR trial (277 children, 24 weeks) found no benefit, but this trial used lower doses (8 to 16 IU). Researchers now believe that oxytocin may be most effective when administered at higher doses before structured social skills training sessions, rather than as a standalone daily treatment. Individual factors including baseline oxytocin levels, OXTR genetics, and epigenetic methylation patterns likely influence who benefits from treatment.
Does oxytocin affect appetite and weight?
Yes, oxytocin reduces caloric intake and influences metabolic function. A single intranasal dose of 24 IU reduces food intake by 100 to 150 kcal per meal in men, with the effect being stronger in individuals with obesity compared to normal-weight participants. Oxytocin selectively suppresses consumption of palatable, high-calorie foods while having less effect on bland food intake. However, an 8-week randomized trial found that despite reducing caloric intake by 152 kcal per test meal, daily intranasal oxytocin did not produce significant overall weight loss. Oxytocin also increases fat oxidation, improves insulin sensitivity, and reduces visceral fat, suggesting potential metabolic benefits independent of weight change. Current evidence positions oxytocin as a modest metabolic modulator rather than a potent weight loss agent.
How is oxytocin administered therapeutically?
Intranasal delivery is the primary route of therapeutic oxytocin administration outside of obstetric settings. The nasal spray delivers oxytocin directly to the brain through the olfactory and trigeminal nerve pathways, bypassing the blood-brain barrier. Typical doses range from 24 to 48 IU, administered as three to six sprays per nostril from a metered-dose nasal spray device. Peak behavioral effects occur approximately 45 to 60 minutes after dosing, and plasma levels return to baseline within 75 to 90 minutes. Proper technique involves tilting the head slightly forward, directing the spray toward the lateral nasal wall, and clearing the nose before administration. The solution should be stored refrigerated at 2 to 8 degrees Celsius and protected from light.
What are the side effects of intranasal oxytocin?
Intranasal oxytocin at therapeutic doses (18 to 48 IU) has a remarkably favorable safety profile. The most commonly reported adverse events are nasal discomfort or irritation (14.3%), irritability (9.0%), tiredness or drowsiness (7.2%), diarrhea (4.5%), and mild skin irritation (4.5%). These effects are generally mild, self-limiting, and comparable in frequency to placebo groups. Serious adverse events like water intoxication, cardiac arrhythmias, and hyponatremia are associated with high-dose intravenous oxytocin used in obstetric settings, not with intranasal administration. Intranasal oxytocin produces no detectable subjective changes in recipients and no reliable side effects at doses of 18 to 40 IU in controlled research settings. Long-term safety data beyond 6 months are limited.
Can oxytocin help with anxiety and depression?
Oxytocin shows promise for anxiety reduction, particularly when administered over multiple days. In a study of 147 patients with anxiety disorders, five days of low-dose oxytocin administration produced a significant decrease in anxiety symptoms. The peptide works by reducing amygdala reactivity to social threats and inhibiting the HPA axis stress response. For depression, the evidence is more preliminary. Low oxytocin levels are consistently associated with depressive symptoms, and intranasal oxytocin improves mood and social engagement in individuals with depression. Postpartum depression is a particularly promising target, as low oxytocin during pregnancy predicts PPD development. However, a counterintuitive finding is that oxytocin can increase anxiety in individuals with insecure attachment styles, underscoring the importance of personalized treatment approaches.
How does oxytocin compare to GLP-1 agonists for weight loss?
Oxytocin and GLP-1 receptor agonists like semaglutide and tirzepatide address weight management through different mechanisms, and their efficacy differs substantially. Semaglutide produces 10 to 15% body weight loss over 68 weeks, while tirzepatide produces 15 to 22% weight loss. Oxytocin, by contrast, has not demonstrated significant weight loss in controlled clinical trials, despite reducing caloric intake by approximately 152 kcal per test meal. Oxytocin's metabolic strengths lie in reducing visceral fat, improving insulin sensitivity, and increasing fat oxidation, effects that may improve metabolic health independent of scale weight. Some researchers are exploring whether oxytocin could complement GLP-1 agonists as an adjunctive therapy, addressing emotional eating and social aspects of weight management that GLP-1 agonists do not directly target.
Is oxytocin safe for long-term use?
Short-term safety data for intranasal oxytocin are reassuring, with controlled studies showing minimal side effects at doses of 18 to 48 IU over periods of 4 to 24 weeks. However, long-term safety data beyond 6 months are limited. Theoretical concerns include oxytocin receptor desensitization from chronic use, suppression of endogenous oxytocin production, and uncertain effects on the developing brain in children and adolescents. Animal studies have not revealed significant long-term adverse effects in adult rodents treated for several months. Practical recommendations include periodic electrolyte monitoring (to detect any antidiuretic effects), cardiovascular assessment for individuals with pre-existing heart conditions, and consideration of intermittent dosing schedules to reduce the risk of receptor downregulation. Pregnant women should not use exogenous oxytocin outside of supervised obstetric care.
What is the difference between oxytocin and vasopressin?
Oxytocin and vasopressin (antidiuretic hormone) are structurally similar nine-amino-acid peptides that differ at only two positions: position 3 (isoleucine in oxytocin, phenylalanine in vasopressin) and position 8 (leucine in oxytocin, arginine in vasopressin). Both are synthesized in the hypothalamus and released from the posterior pituitary. Despite their structural similarity, they serve different primary functions. Oxytocin drives social bonding, uterine contraction, and milk ejection, while vasopressin primarily regulates water balance through kidney V2 receptors and blood pressure through vascular V1a receptors. At high doses, oxytocin can activate vasopressin receptors, which is why high-dose intravenous oxytocin can cause water retention and hyponatremia. Their close structural relationship also creates challenges for immunoassay-based measurement of oxytocin due to antibody cross-reactivity.
Can oxytocin improve social skills in adults without autism?
Yes, intranasal oxytocin enhances several aspects of social cognition and behavior in neurotypical adults. Studies have shown that a single 24 IU dose improves performance on emotion recognition tests by 9 to 15%, increases trust and cooperation in economic games, enhances empathic accuracy, and improves memory for faces. These effects are generally most pronounced in individuals with lower baseline social functioning. However, the effects are context-dependent: oxytocin enhances prosocial behavior when social cues are perceived as safe but can intensify negative social emotions when cues are perceived as threatening. In individuals with high social anxiety or insecure attachment, oxytocin may paradoxically increase social discomfort. The peptide is best understood as a social salience enhancer rather than a universal social lubricant.
What does current research say about oxytocin and gut health?
Oxytocin receptors are expressed throughout the gastrointestinal tract, and the peptide influences gastric motility, intestinal transit, and visceral pain perception. Researchers at the University of Queensland have developed gut-stable oxytocin analogs that can be taken orally, creating potential treatments for irritable bowel syndrome (IBS) and inflammatory bowel disease (IBD). These analogs resist gastrointestinal enzyme degradation while maintaining full activity at the oxytocin receptor. In preclinical studies, oral oxytocin analogs reduced visceral pain sensitivity and normalized gut motility. This represents a significant advance because native oxytocin is rapidly destroyed in the GI tract, making oral delivery previously impossible. The oral analogs are currently in early-stage clinical evaluation for chronic abdominal pain conditions.
How does oxytocin interact with other peptide therapies?
Oxytocin can be considered alongside other peptide therapies based on the therapeutic goals. For metabolic health, oxytocin's appetite-suppressing and insulin-sensitizing effects could complement GLP-1 agonists like semaglutide, though this combination has not been formally tested in clinical trials. For stress and anxiety, oxytocin's anxiolytic properties work through different mechanisms than Selank (which modulates GABA and serotonin) or Semax (which acts on BDNF signaling), theoretically allowing complementary effects. For cognitive enhancement, oxytocin's social memory-enhancing effects are distinct from the general cognitive enhancement associated with Dihexa or P21. Any multi-peptide protocol should be designed under medical supervision, as interactions between neuropeptides are complex and not fully characterized. The dosing calculator at FormBlends can help coordinate timing and doses across multiple peptides.
What new forms of oxytocin are being developed?
Several innovative oxytocin formulations are in development. Long-acting oxytocin analogs with extended half-lives (7+ hours compared to 3-5 minutes for native oxytocin) have shown enhanced efficacy in preclinical obesity models by providing sustained receptor activation throughout the day. Gut-stable oral oxytocin analogs developed at the University of Queensland resist digestive enzyme degradation and allow oral administration for gastrointestinal conditions. Oxytocin receptor-targeted PET imaging agents (68Ga-labeled peptides) are being developed for breast cancer diagnostics, as the oxytocin receptor is overexpressed in certain breast tumors. Additionally, researchers are exploring oxytocin-loaded nanoparticle formulations for sustained nasal delivery and transdermal patches for continuous low-dose administration. These innovations aim to overcome the limitations of current intranasal delivery, including short duration of action and variable bioavailability.
References
- Gimpl G, Fahrenholz F. The oxytocin receptor system: structure, function, and regulation. Physiological Reviews. 2001;81(2):629-683. DOI: 10.1152/physrev.2001.81.2.629.
- Jurek B, Neumann ID. The oxytocin receptor: from intracellular signaling to behavior. Physiological Reviews. 2018;98(3):1805-1908. DOI: 10.1152/physrev.00031.2017.
- Quintana DS, Lischke A, Grace S, et al. Advances in the field of intranasal oxytocin research: lessons learned and future directions for clinical research. Molecular Psychiatry. 2021;26(1):80-91. DOI: 10.1038/s41380-020-00864-7.
- Kosfeld M, Heinrichs M, Zak PJ, et al. Oxytocin increases trust in humans. Nature. 2005;435(7042):673-676. DOI: 10.1038/nature03701.
- Andari E, Duhamel JR, Zalla T, et al. Promoting social behavior with oxytocin in high-functioning autism spectrum disorders. Proceedings of the National Academy of Sciences. 2010;107(9):4389-4394. DOI: 10.1073/pnas.0910249107.
- Simons SS, Bernaerts S, Bello A, et al. Intranasal oxytocin in children and adolescents with autism spectrum disorder. New England Journal of Medicine. 2021;385(16):1462-1473. DOI: 10.1056/NEJMoa2103583.
- Chen X, Jiang Y, Wang J, et al. Optimal dose of oxytocin to improve social impairments and repetitive behaviors in autism spectrum disorders: meta-analysis and dose-response meta-analysis of randomized controlled trials. Frontiers in Psychiatry. 2024;15:1477076. DOI: 10.3389/fpsyt.2024.1477076.
- Jiang X, Zhang Y, Liu H, et al. Chronic oxytocin administration stimulates the oxytocinergic system in children with autism. Nature Communications. 2024;15(1):334. DOI: 10.1038/s41467-023-44334-4.
- Striepens N, Kendrick KM, Maier W, et al. Prosocial effects of oxytocin and clinical evidence for its therapeutic potential. Frontiers in Neuroendocrinology. 2011;32(4):426-450. DOI: 10.1016/j.yfrne.2011.07.001.
- Shamay-Tsoory SG, Abu-Akel A. The social salience hypothesis of oxytocin. Biological Psychiatry. 2016;79(3):194-202. DOI: 10.1016/j.biopsych.2015.07.020.
- Lawson EA. The effects of oxytocin on eating behaviour and metabolism in humans. Nature Reviews Endocrinology. 2017;13(12):700-709. DOI: 10.1038/nrendo.2017.115.
- Lawson EA, Olszewski PK, Wittlin S, et al. The effects of oxytocin on appetite regulation, food intake and metabolism in humans. International Journal of Molecular Sciences. 2021;22(14):7737. DOI: 10.3390/ijms22147737.
- Ott V, Finlayson G, Lehnert H, et al. Oxytocin reduces reward-driven food intake in humans. Diabetes. 2013;62(10):3418-3425. DOI: 10.2337/db13-0663.
- Lawson EA, Marengi DA, DeSilva RA, et al. Intranasal oxytocin for obesity. Obesity. 2024;32(6):1127-1138. DOI: 10.1002/oby.24032.
- Blevins JE, Baskin DG. Translational and therapeutic potential of oxytocin as an anti-obesity strategy: insights from rodents, nonhuman primates and humans. Physiology and Behavior. 2015;152(Pt B):438-449. DOI: 10.1016/j.physbeh.2015.05.023.
- Macdonald K, Feifel D. Oxytocin in schizophrenia: a review of evidence for its therapeutic effects. Acta Neuropsychiatrica. 2012;24(3):130-146. DOI: 10.1111/j.1601-5215.2011.00634.x.
- Davis MC, Lee J, Horan WP, et al. Effects of single dose intranasal oxytocin on social cognition in schizophrenia. Schizophrenia Research. 2013;147(2-3):393-397. DOI: 10.1016/j.schres.2013.04.023.
- Dagani J, Sisti D, Abelli M, et al. Intranasal oxytocin for negative symptoms of schizophrenia: systematic review, meta-analysis, and dose-response meta-analysis of randomized controlled trials. International Journal of Neuropsychopharmacology. 2021;24(8):601-614. DOI: 10.1093/ijnp/pyab020.
- Koch SBJ, van Zuiden M, Nawijn L, et al. Intranasal oxytocin normalizes amygdala functional connectivity in post-traumatic stress disorder. Neuropsychopharmacology. 2016;41(8):2041-2051. DOI: 10.1038/npp.2016.1.
- MacDonald K, MacDonald TM. The peptide that binds: a systematic review of oxytocin and its prosocial effects in humans. Harvard Review of Psychiatry. 2010;18(1):1-21. DOI: 10.3109/10673220903523615.
- Bakermans-Kranenburg MJ, van IJzendoorn MH. Sniffing around oxytocin: review and meta-analyses of trials in healthy and clinical groups with implications for pharmacotherapy. Translational Psychiatry. 2013;3(5):e258. DOI: 10.1038/tp.2013.34.
- MacDonald E, Dadds MR, Brennan JL, et al. A review of safety, side-effects and subjective reactions to intranasal oxytocin in human research. Psychoneuroendocrinology. 2011;36(8):1114-1126. DOI: 10.1016/j.psyneuen.2011.02.015.
- Cai Q, Feng L, Yap KZ. Systematic review and meta-analysis of reported adverse events of long-term intranasal oxytocin treatment for autism spectrum disorder. Psychiatry and Clinical Neurosciences. 2018;72(3):140-151. DOI: 10.1111/pcn.12627.
- Sawares A, Olver J, Morcos M, et al. Oxytocin in old age psychiatry: a systematic review of the safety of using intranasal oxytocin in older adults. Australasian Psychiatry. 2025;33(1):69-76. DOI: 10.1177/10398562241291335.
- Domes G, Heinrichs M, Michel A, et al. Oxytocin improves "mind-reading" in humans. Biological Psychiatry. 2007;61(6):731-733. DOI: 10.1016/j.biopsych.2006.07.015.
- Guastella AJ, Einfeld SL, Gray KM, et al. Intranasal oxytocin improves emotion recognition for youth with autism spectrum disorders. Biological Psychiatry. 2010;67(7):692-694. DOI: 10.1016/j.biopsych.2009.09.020.
- Ding C, Leow MK, Magkos F. Oxytocin in metabolic homeostasis: implications for obesity and diabetes management. Obesity Reviews. 2019;20(1):22-45. DOI: 10.1111/obr.12757.
- Zhang H, Wu C, Chen Q, et al. Treatment of obesity and diabetes using oxytocin or analogs in patients and mouse models. PLoS ONE. 2013;8(5):e61477. DOI: 10.1371/journal.pone.0061477.
- Stoop R. Neuromodulation by oxytocin and vasopressin. Neuron. 2012;76(1):142-159. DOI: 10.1016/j.neuron.2012.09.025.
- Feldman R, Monakhov M, Pratt M, et al. Oxytocin pathway genes: evolutionary ancient system impacting on human affiliation, sociality, and psychopathology. Biological Psychiatry. 2016;79(3):174-184. DOI: 10.1016/j.biopsych.2015.08.008.
- Walum H, Waldman ID, Young LJ. Statistical and methodological considerations for the interpretation of intranasal oxytocin studies. Biological Psychiatry. 2016;79(3):251-257. DOI: 10.1016/j.biopsych.2015.06.016.
- Leng G, Ludwig M. Intranasal oxytocin: myths and delusions. Biological Psychiatry. 2016;79(3):243-250. DOI: 10.1016/j.biopsych.2015.05.003.
- Martins DA, Mazibuko N, Zelaya F, et al. Effects of route of administration on oxytocin-induced changes in regional cerebral blood flow in humans. Nature Communications. 2020;11(1):1160. DOI: 10.1038/s41467-020-14845-5.
- Ricchiuti G, Tuerlinckx E, Taillieu A, et al. Toward effective oxytocin interventions in autism: overcoming challenges and harnessing opportunities. Journal of Psychopharmacology. 2025;39(2):111-125. DOI: 10.1177/02698811241309621.
- Quintana DS, Alvares GA, Hickie IB, et al. Do delivery routes of intranasally administered oxytocin account for observed effects on social cognition and behavior? A two-level model. Neuroscience and Biobehavioral Reviews. 2015;49:182-192. DOI: 10.1016/j.neubiorev.2014.12.011.