Executive Summary

Figure 1: Gonadorelin acts as a synthetic analog of endogenous gonadotropin-releasing hormone, stimulating the pituitary gland to release LH and FSH.
Key Takeaways
- Gonadorelin is structurally identical to endogenous GnRH and acts as a direct stimulator of pituitary LH and FSH release when given in pulsatile fashion.
- Continuous administration causes paradoxical suppression through GnRH receptor desensitization, the basis for GnRH agonist therapy in prostate cancer and endometriosis.
- In the TRT context, gonadorelin serves as an alternative to HCG for maintaining testicular function and fertility, though clinical data supporting this use remain limited compared to HCG.
- The GnRH stimulation test remains a gold-standard diagnostic tool for evaluating pituitary gonadotroph function, hypogonadotropic hypogonadism, and precocious puberty.
- Pulsatile GnRH therapy delivered via programmable pump can induce ovulation in women with hypothalamic amenorrhea, achieving cumulative live birth rates exceeding 80% in experienced centers.
Gonadorelin is a synthetic decapeptide identical to endogenous gonadotropin-releasing hormone (GnRH). It serves as the master regulator of the hypothalamic-pituitary-gonadal (HPG) axis, controlling the release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) from the anterior pituitary. In clinical practice, gonadorelin occupies a unique position as both a diagnostic tool and a therapeutic agent across fertility medicine, hormone replacement therapy, and reproductive endocrinology.
The story of gonadorelin begins with the isolation and characterization of GnRH by Andrew Schally and Roger Guillemin in 1971, work that earned them the Nobel Prize in Physiology or Medicine in 1977. The native GnRH molecule, a decapeptide with the sequence pyroGlu-His-Trp-Ser-Tyr-Gly-Leu-Arg-Pro-Gly-NH2, has a plasma half-life of only 2 to 4 minutes. This rapid degradation by endopeptidases presented both challenges and opportunities for pharmaceutical development. Gonadorelin acetate and gonadorelin hydrochloride were developed as synthetic versions of this peptide for clinical use, while modifications to the native structure yielded the longer-acting GnRH agonists (leuprolide, goserelin, nafarelin) and GnRH antagonists (cetrorelix, ganirelix, degarelix) that dominate modern reproductive pharmacology.
What makes gonadorelin distinct from these analogs is its identical structure to native GnRH. This means it produces physiological effects rather than the pharmacological overstimulation or receptor blockade caused by modified analogs. When administered in pulsatile fashion, gonadorelin can restore normal HPG axis function. When given as a single bolus, it serves as a diagnostic probe to assess pituitary gonadotroph reserve. And when administered continuously, it paradoxically suppresses gonadotropin secretion through receptor desensitization, a principle exploited in the treatment of hormone-dependent cancers and precocious puberty.
The clinical relevance of gonadorelin has expanded considerably in recent years, driven by two developments. First, the FDA's reclassification of human chorionic gonadotropin (HCG) as a biologic in 2020 disrupted the compounding pharmacy market, making HCG less accessible and more expensive for men using it alongside testosterone replacement therapy (TRT) to preserve fertility and testicular function. Gonadorelin emerged as a primary alternative, and its use in men's health clinics has grown substantially. Second, advances in our understanding of KNDy (kisspeptin/neurokinin B/dynorphin) neuron physiology have illuminated the central role of pulsatile GnRH signaling in reproductive health, sparking renewed interest in therapies that work through this natural pathway.
Beyond TRT and fertility, gonadorelin intersects with the broader world of peptide-based therapeutics. Researchers are exploring how GnRH signaling interacts with metabolic pathways, including the incretin system targeted by semaglutide and tirzepatide. The hypothalamic control of reproduction does not operate in complete isolation from energy-sensing pathways, and the relationship between metabolic status and reproductive function is an area of active investigation. Women with hypothalamic amenorrhea, for example, often have underlying energy deficits that simultaneously impair GnRH pulsatility and metabolic regulation. Understanding these connections can inform more holistic approaches to patient care.
The pharmacological development of GnRH-related compounds represents one of the most successful translational stories in endocrinology. From the initial isolation of a 10-amino-acid peptide in the early 1970s to a therapeutic class that generates billions of dollars in annual revenue (primarily through GnRH agonists and antagonists), the GnRH story demonstrates how basic science discovery can transform clinical practice. Gonadorelin itself, as the native peptide, remains the most physiological option in this family and continues to find new clinical applications as our understanding of reproductive neuroendocrinology matures.
This report provides an in-depth analysis of gonadorelin across its key clinical applications. We examine the physiology of GnRH signaling, compare pulsatile and continuous administration paradigms, review evidence for fertility preservation during TRT, evaluate diagnostic applications, detail practical dosing protocols, and address safety considerations. Whether you are a clinician prescribing gonadorelin, a researcher studying reproductive neuroendocrinology, or a patient considering this therapy, this guide consolidates the most current evidence into a practical clinical reference. For broader context on peptide therapies, the Peptide Research Hub covers related compounds and their applications.
Key Takeaways
- Gonadorelin is structurally identical to endogenous GnRH and acts as a direct stimulator of pituitary LH and FSH release when given in pulsatile fashion.
- Continuous administration causes paradoxical suppression through GnRH receptor desensitization, the basis for GnRH agonist therapy in prostate cancer and endometriosis.
- In the TRT context, gonadorelin serves as an alternative to HCG for maintaining testicular function and fertility, though clinical data supporting this use remain limited compared to HCG.
- The GnRH stimulation test remains a gold-standard diagnostic tool for evaluating pituitary gonadotroph function, hypogonadotropic hypogonadism, and precocious puberty.
- Pulsatile GnRH therapy delivered via programmable pump can induce ovulation in women with hypothalamic amenorrhea, achieving cumulative live birth rates exceeding 80% in experienced centers.
GnRH Physiology
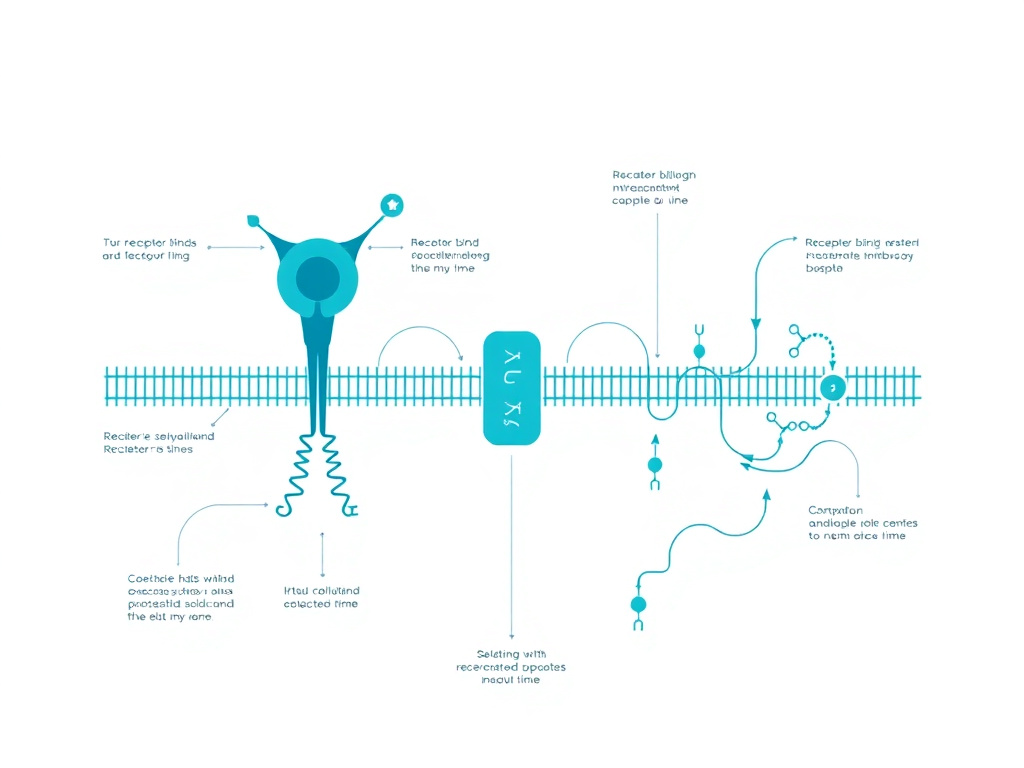
Figure 2: The hypothalamic-pituitary-gonadal axis relies on pulsatile GnRH release from the hypothalamus to drive reproductive hormone production.
Gonadotropin-releasing hormone is the foundational signal of mammalian reproductive physiology. Understanding how this decapeptide controls fertility, sex steroid production, and gonadal function is essential for grasping why gonadorelin therapy works the way it does, and why the pattern of administration matters as much as the dose itself.
The GnRH Neuron Network
GnRH neurons are relatively sparse, numbering approximately 800 to 2,000 in the adult human hypothalamus. Despite their small population, these neurons exert outsized influence over the entire reproductive endocrine system. They originate embryologically from the olfactory placode and migrate into the hypothalamus during fetal development. Failure of this migration causes Kallmann syndrome, a form of congenital hypogonadotropic hypogonadism associated with anosmia. The cell bodies of GnRH neurons are scattered throughout the medial preoptic area and mediobasal hypothalamus, but their axon terminals converge on a common output site: the median eminence, where GnRH is released into the hypophyseal portal blood supply.
The GnRH molecule itself is a decapeptide with the amino acid sequence pyroGlu-His-Trp-Ser-Tyr-Gly-Leu-Arg-Pro-Gly-NH2. Its molecular weight is 1,182 daltons. The pyroglutamate residue at the N-terminus and the amidated glycine at the C-terminus protect the peptide from exopeptidases, but it remains highly susceptible to endopeptidase cleavage, particularly at the Tyr5-Gly6 and Trp3-Ser4 bonds. This gives it a plasma half-life of only 2 to 4 minutes in peripheral blood and approximately 5 to 8 minutes in the portal circulation. The rapid degradation is physiologically important because it allows the pulse pattern to be maintained. If GnRH persisted longer in the portal blood, the sharp on/off pulsatile signal would be blurred.
KNDy Neurons: The Pulse Generator
For decades, the identity of the GnRH pulse generator remained one of the major unsolved problems in reproductive neuroendocrinology. The discovery of KNDy neurons in the arcuate nucleus of the hypothalamus has largely resolved this mystery. KNDy neurons co-express three neuropeptides: kisspeptin, neurokinin B (NKB), and dynorphin A. These three molecules work as an integrated signaling system to generate the episodic GnRH release pattern that drives normal reproductive function.
The model works as follows. Neurokinin B, acting through NK3 receptors on neighboring KNDy neurons, initiates and synchronizes population-wide activation. This synchronized firing triggers the release of kisspeptin from KNDy neuron terminals onto GnRH neurons. Kisspeptin then activates Kiss1 receptors (KISS1R, formerly known as GPR54) on GnRH nerve terminals in the median eminence, causing GnRH release into the portal blood. After a brief delay, dynorphin A, acting through kappa-opioid receptors, inhibits KNDy neuron activity and terminates the pulse. The cycle then resets, with the interval between pulses determined by the time required for the KNDy network to recover from dynorphin-mediated inhibition.
Optogenetic experiments have provided strong evidence for this model. Activation of arcuate kisspeptin neurons in vivo for periods equivalent to the duration of endogenous calcium events elicits LH pulses comparable to those occurring spontaneously. Silencing these neurons decreases LH pulse frequency and amplitude. When silencing is terminated, an immediate LH discharge occurs, followed by a spontaneous pulse after a time-locked interval, confirming that KNDy neurons function as the actual pacemaker rather than a relay of an upstream timing signal (Herbison AE. A simple model of estrous cycle negative and positive feedback regulation of GnRH secretion. Frontiers in Neuroendocrinology. 2020;57:100837. DOI: 10.1016/j.yfrne.2020.100837).
GnRH Pulse Frequency and Its Significance
The frequency of GnRH pulses is not fixed. It varies across the menstrual cycle in women and across the circadian day in both sexes, and these frequency changes carry biological meaning. Faster pulse frequencies (approximately every 60 minutes) favor LH secretion and LH-beta gene transcription. Slower frequencies (approximately every 120 to 180 minutes) favor FSH secretion and FSH-beta transcription. This frequency-dependent differential regulation of LH and FSH from the same cell type is one of the most remarkable features of reproductive endocrinology.
In women, GnRH pulse frequency increases during the late follicular phase, driving the LH surge that triggers ovulation. During the luteal phase, progesterone slows pulse frequency through its action on KNDy neurons (dynorphin expression is progesterone-dependent), shifting the balance toward FSH and supporting follicular recruitment for the next cycle. In men, GnRH pulses occur approximately every 90 to 120 minutes, with higher amplitude pulses during sleep contributing to the early morning testosterone peak.
Sex steroids modulate pulse frequency through feedback at multiple levels. Estradiol exerts both negative feedback (at the arcuate nucleus, suppressing KNDy neuron activity) and positive feedback (at the anteroventral periventricular nucleus, activating a separate population of kisspeptin neurons that drives the LH surge). Testosterone, after local aromatization to estradiol, provides negative feedback through similar mechanisms. The understanding of these feedback loops is critical for appreciating how exogenous testosterone suppresses the HPG axis and why interventions like gonadorelin aim to maintain pituitary stimulation despite this suppression.
The GnRH Receptor
GnRH exerts its effects by binding to the GnRH receptor (GnRHR), a seven-transmembrane G protein-coupled receptor expressed on anterior pituitary gonadotroph cells. The human GnRHR has several unique structural features that distinguish it from most other GPCRs. Most significantly, it lacks an intracellular C-terminal tail, which in other GPCRs mediates rapid receptor desensitization through beta-arrestin recruitment and clathrin-mediated endocytosis. The absence of this tail means that the GnRHR is relatively resistant to rapid homologous desensitization, allowing it to maintain signaling during pulsatile stimulation.
Upon GnRH binding, the receptor couples primarily to Gq/11 proteins, activating phospholipase C-beta. This enzyme cleaves phosphatidylinositol 4,5-bisphosphate (PIP2) into two second messengers: inositol 1,4,5-trisphosphate (IP3) and diacylglycerol (DAG). IP3 triggers calcium release from the endoplasmic reticulum, while DAG activates protein kinase C (PKC) isoforms. The resulting intracellular calcium transient is the immediate trigger for gonadotropin exocytosis. Simultaneously, PKC activation stimulates the mitogen-activated protein kinase (MAPK) cascade, particularly ERK1/2, which drives transcription of the gonadotropin subunit genes (common alpha-subunit, LH-beta, and FSH-beta).
The receptor also couples to Gs proteins under certain conditions, activating adenylyl cyclase and generating cyclic AMP (cAMP). Research published in Frontiers in Endocrinology has shown that pulsatile GnRH stimulation produces matching pulses of cAMP elevation and PKA activation that are rapid and transient, without measurable desensitization from pulse to pulse. In contrast, tonic GnRH stimulation causes a transient increase in cAMP that rapidly returns to baseline despite continued GnRH presence, while the Gq/11 pathway remains active. This differential signaling pattern appears to be part of the molecular mechanism by which gonadotroph cells decode pulse frequency (Armstrong SP, et al. Pulsatile and sustained GnRH receptor signaling. Journal of Biological Chemistry. 2010;285(32):24360-24371. DOI: 10.1074/jbc.M110.155275).
GnRH in the Context of the Complete HPG Axis
GnRH does not operate in isolation. It is part of an integrated feedback system involving the hypothalamus, pituitary, and gonads. LH and FSH released from the pituitary act on the gonads to stimulate sex steroid production and gametogenesis. In men, LH acts on Leydig cells to drive testosterone synthesis, while FSH acts on Sertoli cells to support spermatogenesis. In women, LH and FSH coordinate follicular development, estrogen production, ovulation, and corpus luteum formation.
The sex steroids produced by the gonads (testosterone, estradiol, progesterone) and gonadal peptides (inhibin A, inhibin B, activin, follistatin) feed back to suppress GnRH and gonadotropin secretion, completing the feedback loop. Inhibin B, produced by Sertoli cells in men and granulosa cells in women, selectively suppresses FSH. This entire system represents a classic example of endocrine feedback regulation, and disruption at any level can impair reproductive function.
For men on TRT, exogenous testosterone provides powerful negative feedback at both the hypothalamic and pituitary levels, suppressing GnRH pulse frequency and amplitude. This leads to dramatic reductions in LH and FSH, which in turn cause testicular atrophy and impaired spermatogenesis. The rationale for concurrent gonadorelin therapy is to provide exogenous GnRH stimulation to maintain some degree of pituitary-gonadal signaling despite the suppressive effects of exogenous testosterone. Understanding this physiology is essential for clinicians managing fertility preservation in men receiving testosterone therapy. The GLP-1 Research Hub covers related metabolic and hormonal topics.
Gonadotropin Secretion: The Two-Pool Model
Understanding how gonadotroph cells respond to GnRH requires familiarity with the two-pool model of gonadotropin secretion. Pituitary gonadotrophs contain two distinct pools of gonadotropin-containing secretory vesicles: the readily releasable pool (RRP) and the reserve pool. The RRP consists of vesicles docked at the plasma membrane, ready for immediate exocytosis upon calcium signaling. The reserve pool contains the majority of gonadotropin stores but requires mobilization and transport to the membrane before release can occur.
Each GnRH pulse triggers exocytosis primarily from the RRP, producing the acute LH surge observed within minutes of GnRH exposure. Between pulses, the RRP is replenished from the reserve pool, a process that requires both vesicle trafficking (dependent on the cytoskeleton and motor proteins) and new gonadotropin synthesis (dependent on transcription of the LH-beta, FSH-beta, and alpha-subunit genes). The interpulse interval must be long enough for adequate RRP replenishment; if pulses are too frequent, the RRP becomes depleted and each successive LH response becomes smaller.
This two-pool model explains several clinical observations. The "self-priming" effect of GnRH, where repeated pulses produce progressively larger LH responses in GnRH-deficient patients, reflects the gradual buildup of the RRP in gonadotrophs that have been dormant. The "flare" effect of GnRH agonists represents the massive emptying of both RRP and reserve pools during the initial period of continuous stimulation. And the subsequent suppression reflects the depletion of these pools combined with impaired new synthesis due to signaling pathway desensitization.
GnRH and the Immune System
Emerging research has revealed that GnRH receptors are expressed not only on pituitary gonadotrophs but also on immune cells, including T lymphocytes, B lymphocytes, and thymocytes. GnRH appears to play a role in thymic function and T cell development, and GnRH receptor expression on immune cells raises the possibility of extra-pituitary effects of gonadorelin therapy. While these immunological effects are not the primary clinical target, they represent an active area of research. Some investigators have proposed that GnRH signaling contributes to the sexual dimorphism of the immune system, potentially explaining sex differences in autoimmune disease susceptibility and immune response to infection.
The thymic connection is particularly interesting. The thymus, which produces T cells, expresses GnRH receptors and responds to GnRH stimulation with increased thymocyte proliferation. GnRH agonist-induced castration has been shown to cause thymic regeneration in animal models, an effect that has been explored as a potential adjunct to immune reconstitution after bone marrow transplantation. Thymosin alpha-1, another peptide that targets thymic function, may interact with GnRH-related immune effects, though this relationship has not been formally studied. Compounds like LL-37 and KPV also influence immune function through entirely different pathways.
Species Conservation and Clinical Relevance
The GnRH decapeptide sequence is remarkably conserved across mammalian species, with the same pyroGlu-His-Trp-Ser-Tyr-Gly-Leu-Arg-Pro-Gly-NH2 structure found in humans, primates, rodents, and most other mammals. This conservation extends to the receptor, with the tailless GnRHR present in all mammals studied to date. The high degree of conservation has two practical implications. First, it allowed much of the foundational GnRH physiology research to be conducted in animal models with direct translational relevance. Second, it means that gonadorelin, as a structurally identical synthetic version of the native peptide, interacts with human GnRH receptors in exactly the same way as the endogenous hormone. There is no "foreign" aspect to the molecule, which contributes to its favorable safety profile and low immunogenicity.
Other GnRH forms do exist in the human body. GnRH-II (His5, Trp7, Tyr8-GnRH) is a second GnRH variant found in the midbrain. While GnRH-II has been identified in human tissues, its receptor (GnRH-IIR) appears to be non-functional in humans due to a frameshift mutation and premature stop codon. The physiological role of GnRH-II in humans remains poorly understood, and all current clinical applications of gonadorelin relate exclusively to the GnRH-I pathway described above.
Steroid Feedback Mechanisms in Detail
The negative feedback loop through which sex steroids control GnRH secretion operates at multiple anatomical and molecular levels. Testosterone exerts its feedback effects primarily after local conversion to estradiol by the enzyme aromatase (CYP19A1), which is expressed in hypothalamic neurons including KNDy cells. Estradiol then acts on estrogen receptor alpha (ERalpha) expressed on KNDy neurons to suppress kisspeptin and NKB expression while upregulating dynorphin expression. The net effect is reduced GnRH pulse frequency and amplitude. This is why aromatase inhibitors can increase GnRH pulsatility and gonadotropin secretion, and why men with aromatase deficiency have elevated gonadotropins despite normal or high testosterone levels.
Progesterone provides additional negative feedback in women, primarily through its action on KNDy neurons. The progesterone receptor is expressed on dynorphin-containing neurons in the arcuate nucleus, and progesterone enhances dynorphin signaling, which slows GnRH pulse frequency during the luteal phase. This progesterone-mediated slowing of GnRH pulses shifts the LH:FSH ratio in favor of FSH, supporting the development of a new cohort of follicles for the next menstrual cycle.
Positive feedback, in contrast, operates through a different population of kisspeptin neurons located in the anteroventral periventricular (AVPV) nucleus in rodents and the rostral periventricular area of the third ventricle (RP3V) in primates. These AVPV/RP3V kisspeptin neurons express estrogen receptor alpha but respond to rising estradiol levels with increased kisspeptin expression, a response opposite to that of the arcuate KNDy neurons. This positive feedback circuit generates the preovulatory LH surge that triggers ovulation. The sexually dimorphic nature of this circuit (it is well-developed in females but vestigial in males) explains why men do not experience LH surges despite having circulating estradiol.
Inhibin, Activin, and Follistatin: Fine-Tuning the System
The HPG axis is further refined by gonadal peptide hormones that selectively modulate FSH without affecting LH. Inhibin B, secreted by Sertoli cells in men and granulosa cells in women, selectively suppresses FSH synthesis and secretion at the pituitary level. Inhibin A, produced primarily by the corpus luteum in women, provides additional FSH suppression during the luteal phase. These inhibins act directly on pituitary gonadotrophs, adding a gonadal-level feedback loop that is independent of GnRH signaling.
Activin, structurally related to inhibin, has the opposite effect: it stimulates FSH production. And follistatin, an activin-binding protein, neutralizes activin's FSH-stimulating effect. The interplay between inhibin, activin, and follistatin provides a fine-tuning mechanism that adjusts FSH levels based on gonadal status, supplementing the GnRH-dependent regulation. When interpreting gonadorelin's effects on FSH, clinicians should be aware that the FSH response reflects both direct GnRH stimulation and the prevailing inhibin/activin milieu.
GnRH and Energy Balance: The Metabolic Connection
GnRH neurons are exquisitely sensitive to the body's energy status. This makes biological sense: reproduction is metabolically expensive, and organisms suppress reproductive function when energy is scarce. The molecular links between metabolism and GnRH secretion involve multiple pathways. Leptin, secreted by adipose tissue in proportion to fat stores, provides a permissive signal for GnRH pulsatility. Low leptin levels (as in severe caloric restriction or low body fat) suppress KNDy neuron activity and reduce GnRH pulse frequency. This is the primary mechanism underlying hypothalamic amenorrhea in women with eating disorders or extreme exercise regimens.
Insulin and insulin-like growth factor 1 (IGF-1) also influence GnRH neurons, linking the reproductive axis to glucose metabolism and growth. Ghrelin, a hunger hormone, suppresses GnRH pulsatility, providing another route by which energy deficit impairs reproductive function. These metabolic inputs converge on KNDy neurons and directly on GnRH neurons, integrating nutritional status with reproductive output. The relevance for clinical practice is clear: metabolic optimization may enhance the effectiveness of gonadorelin therapy, while metabolic dysfunction can impair the response. Compounds like MK-677 (ibutamoren) that influence growth hormone and IGF-1 signaling may interact with reproductive axis function through these metabolic pathways.
Circadian Regulation of GnRH Secretion
GnRH pulse amplitude exhibits a circadian pattern, with higher amplitude pulses occurring during sleep, particularly during the early nighttime hours. This sleep-associated increase in GnRH pulsatility drives the well-documented morning peak in testosterone levels in young men. The circadian input to GnRH neurons comes from the suprachiasmatic nucleus (SCN), the brain's master circadian clock, through both direct neural projections and indirect pathways involving vasoactive intestinal peptide (VIP) and other neuropeptides.
The clinical relevance of circadian GnRH regulation extends to the timing of diagnostic tests and therapy. The GnRH stimulation test is typically performed in the morning for standardization, and the higher baseline gonadotropin levels in the morning may influence test results. For therapeutic applications, the rationale for bedtime injection of gonadorelin is partly based on aligning exogenous GnRH stimulation with the natural circadian enhancement of GnRH pulsatility during sleep. Disrupted sleep, shift work, and jet lag can impair GnRH pulsatility and contribute to reproductive dysfunction, a phenomenon increasingly recognized in clinical practice.
GnRH and the Gut-Brain Axis
An emerging area of research explores the connection between gut health, the microbiome, and reproductive endocrine function. The gut-brain axis, which mediates bidirectional communication between the gastrointestinal tract and the central nervous system, can influence hypothalamic function including GnRH secretion. Gut-derived signals, including short-chain fatty acids, gut hormones (GLP-1, peptide YY, ghrelin), and bacterial metabolites, can modulate hypothalamic neuron activity through vagal afferents and systemic circulation.
Gut inflammation has been shown to suppress the HPG axis in animal models, reducing GnRH pulse frequency and causing reproductive dysfunction. This observation has clinical relevance for patients with inflammatory bowel disease, celiac disease, or significant gut dysbiosis, who may experience reproductive impairment partly related to altered gut-brain signaling. Peptides targeting gut health, such as larazotide (which modulates intestinal permeability), BPC-157 (which has gastroprotective properties), and KPV (which has anti-inflammatory effects on gut mucosa), may indirectly support reproductive function by optimizing the gut-brain axis, though these connections are speculative and have not been studied in clinical trials.
The relationship between gut-derived hormones and GnRH is particularly interesting in the context of GLP-1 receptor agonists. GLP-1, originally identified as an incretin hormone from intestinal L-cells, has receptors in the hypothalamus. Whether GLP-1 receptor activation directly influences GnRH neurons is an active area of investigation. Some animal studies suggest that GLP-1 signaling can modulate kisspeptin neuron activity, potentially linking metabolic sensing through the gut-brain axis to reproductive function. This raises intriguing questions about how semaglutide and related GLP-1 therapies might affect reproductive endocrine function beyond their well-documented metabolic effects.
Neuroplasticity of GnRH Neurons
GnRH neurons exhibit remarkable structural plasticity that directly influences their function. The axon terminals of GnRH neurons in the median eminence undergo morphological remodeling that changes their proximity to the portal capillaries and their encapsulation by tanycyte processes. Tanycytes are specialized glial cells that line the floor of the third ventricle and extend processes into the median eminence, where they wrap around GnRH nerve terminals.
During the preovulatory LH surge, tanycyte processes retract, bringing GnRH terminals into closer contact with the portal capillary fenestrations. This structural change facilitates more efficient GnRH release into the portal blood. Outside the surge period, tanycyte processes re-extend and partially insulate GnRH terminals from the portal circulation. This morphological gating mechanism adds yet another level of regulation to GnRH secretion, operating at the interface between the neuroendocrine and vascular compartments.
Estradiol regulates tanycyte morphology, providing a mechanism by which the ovarian steroid environment modulates the efficiency of GnRH release independent of GnRH neuron firing rate. Other factors that influence tanycyte morphology include prostaglandins, nitric oxide, and transforming growth factor beta family members. The tanycyte-GnRH neuron interface represents a relatively new frontier in reproductive neuroendocrinology, and its dysfunction could contribute to forms of reproductive impairment that are not easily explained by alterations in GnRH pulse frequency alone.
Environmental Factors Affecting GnRH Function
Beyond internal physiological regulators, external environmental factors can significantly impact GnRH neuron function and reproductive endocrine output. Endocrine-disrupting chemicals (EDCs) including bisphenol A (BPA), phthalates, parabens, and certain pesticides can interfere with GnRH signaling at multiple levels. Some EDCs act as estrogen mimics, providing inappropriate negative feedback to the hypothalamus and pituitary. Others may directly affect GnRH neuron development, migration, or function.
Temperature is another environmental factor with direct relevance to reproductive function. In men, testicular temperature regulation is critical for spermatogenesis, and elevated scrotal temperature (from tight clothing, hot baths, laptop use, or occupational heat exposure) can impair sperm production independently of gonadotropin levels. While gonadorelin therapy maintains gonadotropin signaling, it cannot overcome the local temperature-dependent suppression of spermatogenesis. Men using gonadorelin for fertility preservation should be counseled on scrotal temperature optimization as part of a comprehensive approach.
Psychological stress operates through the hypothalamic-pituitary-adrenal (HPA) axis to suppress GnRH pulsatility. Cortisol and corticotropin-releasing hormone (CRH) both inhibit GnRH neuron activity, providing a neuroendocrine mechanism for stress-induced reproductive dysfunction. This is the same mechanism underlying stress-related hypothalamic amenorrhea in women. For men on TRT with gonadorelin, managing stress through behavioral interventions, adequate sleep, and potentially stress-modulating peptides like Selank or DSIP may support the effectiveness of gonadorelin therapy by optimizing the neuroendocrine environment in which it operates.
Exercise has a complex relationship with GnRH function. Moderate exercise supports reproductive health by improving insulin sensitivity, reducing body fat, lowering inflammation, and modulating stress hormones. Excessive exercise, particularly in combination with caloric restriction, can suppress GnRH pulsatility and reproductive function. The threshold at which exercise transitions from beneficial to suppressive varies between individuals and depends on energy availability (calories consumed minus calories expended in exercise) rather than exercise volume alone. Men and women using gonadorelin for fertility applications should maintain adequate energy intake to support reproductive function.
Pulsatile vs Continuous Administration
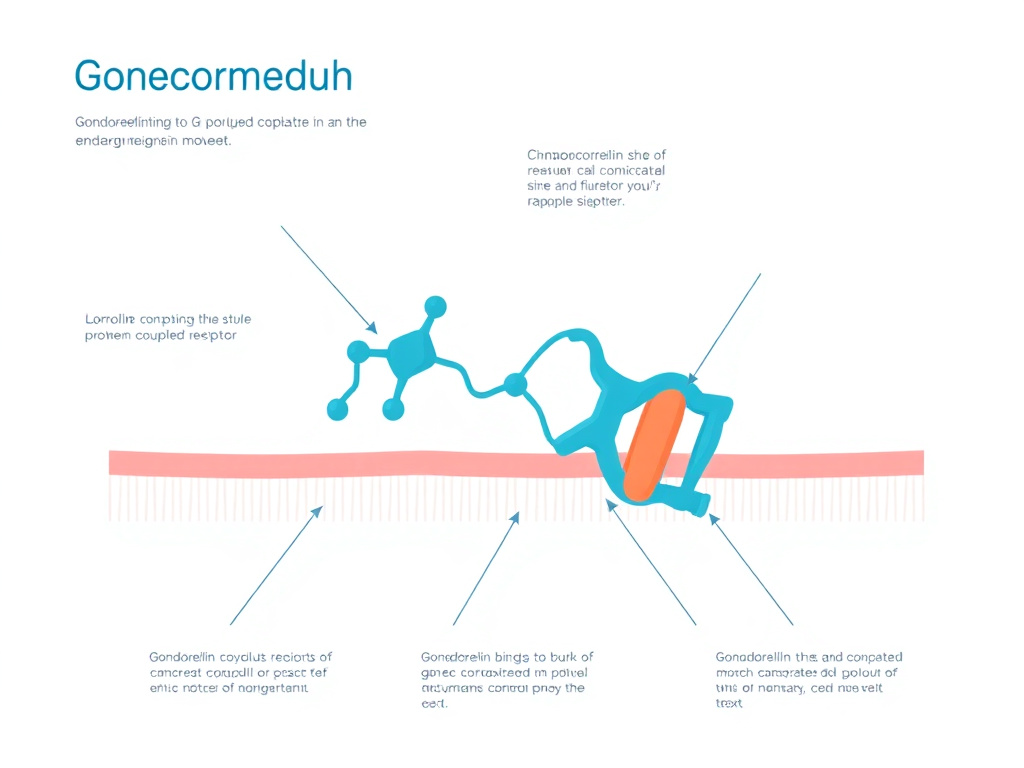
Figure 3: Pulsatile GnRH administration stimulates gonadotropin release, while continuous exposure paradoxically suppresses the HPG axis through receptor desensitization.
The distinction between pulsatile and continuous GnRH administration represents one of the most important pharmacological principles in reproductive endocrinology. The same molecule, delivered in different temporal patterns, produces diametrically opposite biological effects. This section examines the molecular basis for this paradox and its clinical applications.
The Pulsatile Paradigm: Stimulation
Normal GnRH secretion is inherently pulsatile. In healthy men, pulses occur approximately every 90 to 120 minutes. In premenopausal women, pulse frequency varies from approximately 60 minutes in the late follicular phase to 180 to 240 minutes in the mid-luteal phase. Each pulse consists of a brief burst of GnRH release lasting 5 to 10 minutes, followed by a prolonged interpulse interval during which GnRH levels in the portal blood drop to undetectable levels.
When gonadorelin is administered in a pulsatile pattern that mimics this endogenous rhythm, it stimulates the pituitary gonadotroph cells to synthesize and secrete LH and FSH in a physiological manner. Each pulse of GnRH activates the Gq/11 signaling cascade described above, triggering calcium-dependent exocytosis of stored gonadotropin granules. The interpulse interval allows the signaling machinery to reset. Specifically, PKC isoforms return to their basal state, IP3 receptors recover from desensitization, and the gonadotroph cells replenish their pools of readily releasable gonadotropin-containing vesicles.
This pulse-by-pulse replenishment is critical. Each GnRH pulse produces an LH pulse of relatively consistent amplitude (assuming constant GnRH pulse dose and a healthy pituitary). The gonadotroph cell responds faithfully to each pulse because the signaling pathway has fully recovered between pulses. The result is sustained gonadotropin secretion that can maintain gonadal function indefinitely, as demonstrated by decades of clinical experience with pulsatile GnRH pump therapy for hypothalamic amenorrhea.
The Continuous Paradigm: Suppression Through Desensitization
Continuous GnRH exposure produces a strikingly different outcome. When GnRH is delivered without interruption, whether through a continuous infusion or a long-acting GnRH agonist like leuprolide, the initial response is a brief burst of gonadotropin release (the "flare" effect). This is followed by progressive desensitization of the GnRH receptor and downstream signaling pathways, leading to profound suppression of LH and FSH within 1 to 3 weeks.
The molecular mechanisms underlying this desensitization operate at multiple levels. Despite lacking a C-terminal tail for classic beta-arrestin-mediated desensitization, the GnRHR undergoes slow internalization through lipid raft-dependent pathways when continuously occupied by agonist. The rate of receptor internalization exceeds the rate of new receptor synthesis and recycling, reducing cell-surface receptor density. But receptor downregulation alone doesn't fully explain the suppression. Downstream signaling components also become refractory.
Research published in the Journal of Biological Chemistry has demonstrated that under sustained GnRH challenge, the Gq/11 protein coupling efficiency decreases, PKC isoforms become chronically activated and then depleted, and IP3 receptor sensitivity declines. The cAMP/PKA pathway shows rapid adaptation under continuous stimulation, with the initial cAMP surge returning to baseline within minutes despite ongoing receptor occupancy. This contrasts with pulsatile stimulation, where each pulse generates a fresh cAMP response of constant amplitude.
At the transcriptional level, continuous GnRH exposure also suppresses the expression of gonadotropin subunit genes. LH-beta mRNA levels decline, as do levels of the common alpha-subunit. Interestingly, FSH-beta transcription is less affected by continuous GnRH because it depends more on activin signaling and less on GnRH pulse frequency, though FSH protein secretion still declines due to reduced synthesis and secretion of the common alpha-subunit needed to form the complete heterodimeric hormone.
Clinical Applications of the Pulsatile/Continuous Dichotomy
The understanding that pulsatile GnRH stimulates while continuous GnRH suppresses has been exploited clinically in both directions.
Pulsatile Applications (Stimulation)
Pulsatile GnRH therapy using a programmable subcutaneous or intravenous pump has been used since the early 1980s to treat hypothalamic amenorrhea and induce ovulation in women with deficient endogenous GnRH secretion. The standard approach delivers 5 to 20 micrograms of gonadorelin per pulse every 60 to 120 minutes. In experienced centers, this approach achieves ovulation rates of 80 to 95% and cumulative live birth rates exceeding 80% over multiple treatment cycles. The major advantage over gonadotropin injections is a much lower rate of multiple pregnancies, because pulsatile GnRH produces physiological monofollicular ovulation in most cases.
A 25-year cohort study from a Dutch center, published in the Journal of Assisted Reproduction and Genetics, demonstrated that pulsatile GnRH therapy in women with functional hypothalamic amenorrhea resulted in monofollicular ovulation in 89% of ovulatory cycles, with a cumulative live birth rate of 87% after six treatment cycles. The multiple pregnancy rate was only 3.5%, far lower than the 15 to 20% rate typically seen with gonadotropin therapy (Christin-Maitre S, et al. Pulsatile GnRH therapy for hypothalamic amenorrhea. Journal of Assisted Reproduction and Genetics. 2023;40(1):117-125. DOI: 10.1007/s10815-022-02656-0).
In men, pulsatile GnRH therapy has been used to induce spermatogenesis in patients with congenital hypogonadotropic hypogonadism (CHH), including Kallmann syndrome. A comparative study showed spermatogenesis occurred in 90% of men receiving pulsatile gonadorelin versus 83.3% in those receiving HCG plus human menopausal gonadotropin (HMG), with no statistically significant difference between groups. This demonstrates that working through the physiological GnRH pathway can be as effective as direct gonadotropin replacement for restoring fertility in men with hypothalamic causes of hypogonadism.
Continuous Applications (Suppression)
The suppressive effect of continuous GnRH exposure is the pharmacological basis for GnRH agonist therapy. Long-acting GnRH agonists like leuprolide (Lupron), goserelin (Zoladex), and triptorelin (Trelstar) are modified GnRH molecules with amino acid substitutions (typically at positions 6 and/or 10) that resist enzymatic degradation and have half-lives measured in hours rather than minutes. Formulated as depot injections (monthly, 3-monthly, or 6-monthly), they produce continuous GnRH receptor stimulation that suppresses the HPG axis after an initial 1 to 2 week flare.
Clinical applications of GnRH agonist-mediated suppression include treatment of advanced prostate cancer (medical castration), endometriosis, uterine fibroids, precocious puberty, and as part of controlled ovarian hyperstimulation protocols in assisted reproduction. The suppressive effect is fully reversible upon discontinuation, with recovery of gonadotropin secretion and gonadal function typically occurring within 2 to 8 weeks, depending on the formulation used.
The Gray Zone: Intermittent but Non-Pulsatile Administration
Much of the current clinical use of gonadorelin falls into a category that is neither truly pulsatile (every 60 to 120 minutes via pump) nor truly continuous. When clinicians prescribe gonadorelin as once-daily or twice-daily subcutaneous injections for men on TRT, the administration pattern lies in between. A once-daily injection provides a bolus of GnRH that stimulates an acute LH response, followed by approximately 23 hours without stimulation before the next dose.
This pattern is clearly not equivalent to continuous exposure, so full desensitization does not occur. But it also falls short of the physiological pulse frequency. The question of whether once or twice daily injections provide sufficient stimulation to meaningfully preserve pituitary-gonadal function during TRT is addressed in the TRT Support Applications section of this report. The available clinical data suggest that this approach provides partial, but not complete, maintenance of testicular function.
Frequency-Dependent Gene Regulation
Beyond the simple stimulation/suppression dichotomy, GnRH pulse frequency also determines which gonadotropin is preferentially produced. Experiments using perifused pituitary cell cultures and in vivo studies in GnRH-deficient animal models have established that high-frequency GnRH pulses (every 30 to 60 minutes) preferentially drive LH-beta transcription and LH secretion, while low-frequency pulses (every 120 to 240 minutes) preferentially drive FSH-beta transcription and FSH secretion.
The molecular mechanism involves differential activation of MAPK signaling pathways. High-frequency GnRH pulses produce sustained ERK1/2 activation that favors the transcription factors driving LH-beta expression (Egr-1, SF-1). Low-frequency pulses produce transient ERK1/2 activation that favors the transcription factors driving FSH-beta expression (CREB, AP-1). This frequency decoding explains how a single releasing hormone can differentially regulate two distinct gonadotropins from the same cell type.
The clinical relevance is significant. In polycystic ovary syndrome (PCOS), abnormally high GnRH pulse frequency leads to LH excess relative to FSH, contributing to anovulation and hyperandrogenism. Conversely, in hypothalamic amenorrhea, abnormally low or absent GnRH pulses result in insufficient gonadotropin secretion overall. Therapeutic GnRH pulse frequency can be adjusted to optimize the LH:FSH ratio for the desired clinical outcome, whether that is ovulation induction, spermatogenesis, or diagnostic assessment.
Receptor Dynamics: Internalization, Recycling, and Trafficking
Understanding GnRH receptor trafficking provides additional insight into why administration pattern matters so much. Upon agonist binding, the GnRHR undergoes slow internalization through clathrin-independent, lipid raft-dependent endocytic pathways. The rate of internalization is significantly slower than for most GPCRs, which typically use clathrin-mediated endocytosis facilitated by their C-terminal tails. For the tailless GnRHR, the internalization half-time is approximately 20 to 30 minutes, compared to 2 to 5 minutes for typical GPCRs. This slow internalization means that during a pulsatile exposure, the receptor remains on the cell surface and available for signaling throughout the duration of each pulse, supporting efficient signal transduction.
Once internalized, GnRH receptors are primarily routed to lysosomes for degradation rather than recycled to the cell surface. This degradation pathway means that receptor replacement on the cell surface depends on new receptor synthesis from gene transcription and translation, a process that takes hours. Under pulsatile stimulation, the rate of receptor internalization and degradation is balanced by new receptor synthesis during the interpulse intervals, maintaining a stable steady-state level of cell-surface receptor. Under continuous stimulation, receptor internalization and degradation outpace new synthesis, leading to progressive receptor downregulation.
The dynamics of GnRH receptor expression are also influenced by GnRH itself. Low-frequency GnRH pulses actually upregulate GnRHR gene expression, increasing the number of receptors available for the next pulse. This positive autoregulation ensures that the pituitary remains responsive to ongoing pulsatile GnRH stimulation. High-frequency or continuous GnRH exposure, in contrast, can downregulate GnRHR mRNA levels, contributing to the desensitization process. These transcriptional effects, which operate on a timescale of hours to days, add another layer of complexity to the pulsatile/continuous dichotomy.
Implications for Intermittent Injection Protocols
The detailed understanding of receptor signaling dynamics has practical implications for the intermittent injection protocols used in TRT support. A once-daily subcutaneous injection of gonadorelin creates a transient spike of GnRH lasting approximately 10 to 15 minutes (given the 2 to 4 minute half-life, effective concentrations persist for 3 to 5 half-lives). This is followed by approximately 24 hours without GnRH exposure. From the receptor signaling perspective, this provides a single "pulse" per day, well below the physiological frequency of approximately 16 pulses per day (at one pulse every 90 minutes).
However, even a single daily pulse may provide sufficient stimulation to prevent complete gonadotroph dormancy. The pulse activates the Gq/11 and ERK1/2 signaling pathways, triggers gonadotropin release from the readily releasable pool, and stimulates gonadotropin subunit gene transcription. The long interpulse interval (24 hours) allows complete recovery of all signaling components and full replenishment of releasable gonadotropin stores. The question is whether a single daily pulse provides enough cumulative gonadotropin secretion to maintain meaningful testicular function in the face of TRT-induced suppression.
Twice-daily injection protocols, by reducing the interpulse interval to approximately 12 hours, provide a second daily stimulation event. While still far from physiological frequency, this doubles the daily LH output and may provide better testicular support. Some clinicians prescribe three-times-daily injections for men with the most pressing fertility concerns, bringing the protocol closer to (though still far from) physiological pulsatility. The development of wearable subcutaneous micro-pump devices for gonadorelin delivery would represent a significant advance, enabling truly pulsatile therapy in the TRT support context. Such devices are under development but are not yet commercially available for this indication.
GnRH Antagonists: The Alternative Approach to Suppression
While this report focuses on gonadorelin (native GnRH), understanding GnRH antagonists provides important context. Unlike GnRH agonists that initially stimulate before causing desensitization, GnRH antagonists (cetrorelix, ganirelix, degarelix, elagolix, relugolix) directly block the GnRH receptor without any initial flare effect. This provides immediate and reversible suppression of gonadotropin secretion.
GnRH antagonists are increasingly used in prostate cancer treatment (relugolix was FDA-approved in 2020 as the first oral GnRH antagonist), assisted reproduction protocols, and endometriosis management. The absence of the initial testosterone flare that occurs with GnRH agonists is a significant clinical advantage in prostate cancer, where the flare can cause disease progression. For reproductive medicine, GnRH antagonists allow more flexible protocol management in IVF cycles.
The distinction between agonist-mediated desensitization (which gonadorelin can cause if given continuously) and antagonist-mediated blockade (which directly prevents receptor activation) is fundamental to the pharmacology of GnRH-targeted therapies. Both approaches ultimately suppress the HPG axis, but through completely different molecular mechanisms. This pharmacological distinction has implications for the recovery of HPG axis function after treatment cessation: recovery is generally faster after GnRH antagonist therapy than after long-acting GnRH agonist therapy, because antagonist-treated gonadotrophs have not undergone the signaling pathway changes associated with chronic agonist-induced desensitization. Research on kisspeptin-10 provides additional insight into upstream regulators of GnRH signaling.
Fertility Preservation Research
Figure 4: Research data on gonadorelin's effectiveness for preserving fertility parameters during concurrent testosterone replacement therapy.
Fertility preservation during hormone therapy represents one of the most clinically pressing applications of gonadorelin. As testosterone replacement therapy has become increasingly common, with an estimated 2.5 million men in the United States alone using some form of testosterone, the need for strategies to protect reproductive function during treatment has grown proportionally. This section examines the evidence for gonadorelin in this context.
The Fertility Problem with Exogenous Testosterone
Exogenous testosterone, whether delivered by injection, gel, pellet, or patch, profoundly suppresses the HPG axis. Within 2 to 4 weeks of starting TRT, serum LH and FSH levels decline to near-undetectable levels in most men. Without adequate LH stimulation, Leydig cells reduce intratesticular testosterone (ITT) production, and without FSH stimulation, Sertoli cell function deteriorates. Intratesticular testosterone levels, which need to be 50 to 100 times higher than serum levels to support spermatogenesis, drop by approximately 94% in men on TRT who do not receive concurrent gonadotropin support.
The clinical consequences are substantial. Testicular volume decreases by an average of 20 to 40% over the first year of TRT. Sperm counts decline dramatically, with 65 to 90% of men developing severe oligozoospermia (fewer than 5 million sperm/mL) or azoospermia within 6 to 12 months. For younger men who may want biological children in the future, this represents a significant concern. And while these effects are generally reversible upon discontinuation of TRT, recovery of spermatogenesis can take 6 to 24 months, and in a small percentage of men, full recovery may not occur.
HCG: The Historical Standard for Fertility Preservation
Human chorionic gonadotropin has been the mainstay of fertility preservation during TRT for decades. HCG shares structural homology with LH and binds to the LH receptor on Leydig cells, directly stimulating intratesticular testosterone production without requiring pituitary signaling. A landmark study by Coviello et al. showed that co-administration of 250 IU HCG subcutaneously every other day with TRT maintained intratesticular testosterone within 7% of baseline, while 500 IU every other day actually increased ITT by 26% above baseline. Spermatogenesis was preserved in a majority of men at one-year follow-up (Coviello AD, et al. Low-dose human chorionic gonadotropin maintains intratesticular testosterone in normal men with testosterone-induced gonadotropin suppression. Journal of Clinical Endocrinology & Metabolism. 2005;90(5):2595-2602. DOI: 10.1210/jc.2004-0802).
However, the HCG situation changed dramatically in March 2020 when the FDA reclassified HCG as a biologic under the Biologics Price Competition and Innovation Act. This removed HCG from the compounding pharmacy market, limiting availability to brand-name products (Pregnyl, Novarel) and significantly increasing cost. Many men's health clinics and TRT providers began searching for alternatives, and gonadorelin emerged as the primary candidate.
Gonadorelin as an HCG Alternative: Rationale and Mechanism
The rationale for using gonadorelin as an HCG replacement during TRT is straightforward: if HCG works by directly stimulating the LH receptor, gonadorelin works one step upstream by stimulating the pituitary to release endogenous LH (and FSH), which then acts on the testes. This upstream mechanism has a theoretical advantage: it provides both LH and FSH stimulation, whereas HCG provides only LH-like activity. Since FSH is important for Sertoli cell function and spermatogenesis, gonadorelin's ability to stimulate FSH release could theoretically offer superior fertility preservation compared to HCG alone.
The major challenge with this approach, however, is that exogenous testosterone powerfully suppresses the pituitary gonadotroph cells through negative feedback. Even with gonadorelin stimulation, the ability of the pituitary to respond may be blunted when it is under the suppressive influence of supraphysiological exogenous testosterone. This is a fundamentally different situation from treating hypogonadotropic hypogonadism, where the pituitary is intact but simply not receiving adequate GnRH stimulation.
Clinical Evidence for Gonadorelin in TRT Fertility Preservation
It is important to state clearly that large-scale, randomized controlled trials comparing gonadorelin to HCG or placebo for fertility preservation during TRT have not been published as of early 2026. The evidence base consists of clinical experience from TRT clinics, small observational studies, and extrapolation from the broader GnRH physiology literature.
Clinical experience from men's health clinics suggests that gonadorelin administered subcutaneously at doses of 100 to 200 mcg once or twice daily can maintain testicular volume and partially preserve gonadotropin levels in men on concurrent TRT. Clinicians report that approximately 50 to 60% of baseline testicular function is maintained with this approach, compared to roughly 80 to 90% preservation with HCG co-administration. The attenuation of response is attributed to the suppressive effect of exogenous testosterone on pituitary sensitivity to GnRH.
Some practitioners have reported semen analysis results in men using gonadorelin during TRT that show maintenance of some level of spermatogenesis, though typically at lower sperm concentrations than would be seen with HCG co-therapy. The clinical data are not yet sufficient to establish gonadorelin as equivalent to HCG for active fertility preservation in men who are actively trying to conceive. For men on TRT whose primary concern is preventing severe testicular atrophy and maintaining the option of future fertility (but who are not currently trying to conceive), gonadorelin appears to provide a reasonable degree of protection.
Comparison: Gonadorelin vs HCG vs Enclomiphene
Three agents now compete for the fertility preservation niche during TRT: gonadorelin, HCG, and enclomiphene. Each works through a different mechanism and has distinct advantages and limitations.
| Parameter | Gonadorelin | HCG | Enclomiphene |
|---|---|---|---|
| Mechanism | Stimulates pituitary LH + FSH release via GnRH receptor | Directly activates LH receptor on Leydig cells | Blocks estrogen negative feedback at hypothalamus/pituitary (SERM) |
| LH Stimulation | Indirect (requires functional pituitary) | LH-receptor agonist (bypasses pituitary) | Indirect (increases endogenous GnRH via disinhibition) |
| FSH Stimulation | Yes (stimulates both LH and FSH) | No direct FSH effect | Yes (increases endogenous GH and FSH) |
| Route | Subcutaneous injection (daily or BID) | Subcutaneous injection (EOD or 2-3x/week) | Oral (daily) |
| ITT Preservation on TRT | Partial (estimated 50-60%) | Strong (90-100% with 250-500 IU EOD) | Limited data on TRT; better suited as TRT alternative |
| Spermatogenesis | Partial preservation expected | Good preservation documented | Good preservation (when used as TRT alternative) |
| Testicular Size | Partial maintenance | Good maintenance | Good maintenance (when not on concurrent TRT) |
| Availability | Compounding pharmacies (available) | Brand-name only since 2020 (limited, expensive) | Compounding pharmacies (available) |
| Cost (monthly) | $30-80 | $100-300+ | $30-100 |
| Key Limitation | Reduced pituitary response under testosterone suppression | No FSH stimulation; potential LH receptor desensitization at high doses | May conflict with TRT (both trying to raise and suppress T) |
HCG remains the best-studied option for fertility preservation during concurrent TRT, with the strongest evidence for maintaining intratesticular testosterone and spermatogenesis. However, its reduced availability and increased cost since the FDA reclassification have made it less accessible. Gonadorelin offers an accessible alternative that provides partial but meaningful protection of testicular function. Enclomiphene is increasingly popular but is perhaps better suited as a testosterone-boosting monotherapy or TRT alternative rather than an add-on during TRT, since the pharmacological logic of simultaneously giving exogenous testosterone (which suppresses the HPG axis) and a SERM (which tries to activate it) is somewhat contradictory.
Gonadorelin for Female Fertility: Pulsatile GnRH Pump Therapy
While much of the recent attention on gonadorelin focuses on men's health, its longest and best-established fertility application is in women with hypothalamic amenorrhea. Functional hypothalamic amenorrhea (FHA) affects approximately 3 to 5% of women of reproductive age. It is caused by suppression of GnRH pulsatility due to energy deficit, psychological stress, or excessive exercise. These women have intact pituitary glands and functioning ovaries but lack the GnRH signal needed to drive the reproductive cascade.
Pulsatile GnRH pump therapy delivers gonadorelin subcutaneously (or less commonly intravenously) at intervals of 60 to 120 minutes, restoring the missing GnRH pulse signal. The pituitary responds with physiological LH and FSH secretion, which drives normal follicular development, estrogen production, and typically monofollicular ovulation. The approach has been used since the 1980s and has an extensive track record.
A systematic review and meta-analysis by Martin et al. evaluated the efficacy and safety of pulsatile GnRH therapy in patients with idiopathic and functional hypothalamic amenorrhea. The pooled ovulation rate per cycle was 79.3%, the clinical pregnancy rate per ovulatory cycle was 27.8%, and the multiple pregnancy rate was only 5.7%. These outcomes compare favorably with gonadotropin ovulation induction, which has higher multiple pregnancy rates (15 to 20%) and requires more intensive monitoring (Martin KA, et al. Efficacy and safety of pulsatile GnRH therapy in hypothalamic amenorrhea: systematic review and meta-analysis. Fertility and Sterility. 2018;109(5):851-860. DOI: 10.1016/j.fertnstert.2018.01.003).
A more recent comparison from a Chinese center examined pulsatile GnRH therapy versus gonadotropin therapy in both FHA and congenital hypogonadotropic hypogonadism (CHH) patients. Ovulation rates were similar between treatment modalities, but multiple follicular development was significantly more common in the gonadotropin group. In the CHH subgroup, pulsatile GnRH required longer treatment duration to achieve ovulation but produced more physiological hormonal profiles (Liu Y, et al. Pulsatile GnRH therapy: comparison of FHA and CHH. Fertility and Sterility. 2024;122(6):1034-1042. DOI: 10.1016/j.fertnstert.2024.07.009).
Congenital Hypogonadotropic Hypogonadism: Pulsatile GnRH for Male Fertility
Congenital hypogonadotropic hypogonadism (CHH), which includes Kallmann syndrome (CHH with anosmia) and normosmic idiopathic hypogonadotropic hypogonadism (nIHH), affects approximately 1 in 4,000 to 10,000 males and 1 in 40,000 females. These patients have deficient GnRH secretion or action, resulting in absent or incomplete puberty, infertility, and low sex steroid levels. The genetic basis is heterogeneous, with over 50 causative genes identified, including ANOS1 (encoding anosmin-1), FGFR1, GNRHR, KISS1R, TAC3, and TACR3.
For men with CHH who desire fertility, pulsatile GnRH therapy offers a physiological approach to inducing spermatogenesis. By providing the missing GnRH signal, pulsatile gonadorelin allows the intact pituitary to produce LH and FSH, which then stimulate the testes. Clinical studies have demonstrated that pulsatile GnRH therapy induces spermatogenesis in approximately 70 to 90% of men with CHH, with median time to appearance of sperm in the ejaculate of 6 to 12 months. Factors predicting better outcomes include larger testicular volume at treatment initiation (reflecting more Sertoli cell capacity), absence of cryptorchidism history, and prior exposure to gonadotropins (which "primes" the testes).
The European Consensus Statement on CHH recommends either pulsatile GnRH or combined HCG/FSH therapy for fertility induction, noting that both approaches have comparable efficacy. Pulsatile GnRH has the advantage of producing a more physiological gonadotropin profile and allowing endogenous feedback regulation. However, it requires a pump and more frequent dosing, which some patients find burdensome. The choice between approaches often depends on local expertise and patient preference (Boehm U, et al. European Consensus Statement on congenital hypogonadotropic hypogonadism. Nature Reviews Endocrinology. 2015;11(9):547-564. DOI: 10.1038/nrendo.2015.112).
An intriguing phenomenon observed in CHH patients is spontaneous reversal. Approximately 10 to 22% of men initially diagnosed with CHH recover endogenous GnRH pulsatility after treatment with testosterone or pulsatile GnRH, suggesting that the GnRH neuron network retains some plasticity and can be "activated" by appropriate hormonal or maturational signals. This finding challenges the traditional view of CHH as an entirely fixed developmental disorder and suggests that some cases may represent extreme constitutional delay rather than permanent deficiency.
Fertility Recovery After GnRH Agonist Suppression
An additional clinical scenario where gonadorelin may play a role is recovery of fertility after long-term GnRH agonist suppression. Women who have used GnRH agonists for endometriosis or men who have received GnRH agonist therapy for prostate cancer may experience delayed HPG axis recovery. While most patients recover spontaneously within 2 to 6 months of discontinuing GnRH agonist therapy, those with persistent suppression may benefit from pulsatile gonadorelin to "restart" the HPG axis by providing the pituitary with the pulsatile stimulation it needs to resume normal gonadotropin synthesis and secretion.
Spermatogenesis: The Biology of Sperm Production
To understand why fertility preservation during TRT is so challenging, it helps to understand the biology of spermatogenesis itself. Sperm production in the seminiferous tubules depends on the coordinated action of two cell types: Leydig cells (producing intratesticular testosterone under LH stimulation) and Sertoli cells (providing the structural and nutritional support for developing sperm cells under FSH stimulation). Both signals are necessary for complete spermatogenesis.
Intratesticular testosterone concentrations need to be approximately 50 to 100 times higher than serum levels to support spermatogenesis. This huge concentration gradient is maintained by the local production of testosterone by Leydig cells within the testicular interstitium. Exogenous testosterone, which enters the testes from the systemic circulation, cannot achieve these concentrations. Even if serum testosterone is in the high-normal range on TRT, intratesticular testosterone may be insufficient for spermatogenesis because it is being delivered from outside rather than produced locally.
The spermatogenic cycle in humans takes approximately 74 days from spermatogonial stem cell division to mature spermatozoa. Add another 12 to 14 days for epididymal transit, and the total time from initiation of spermatogenesis to appearance of sperm in the ejaculate is approximately 90 days. This timeline has practical implications for monitoring: after starting gonadorelin (or any intervention to restore spermatogenesis), at least 3 to 4 months must pass before a semen analysis can meaningfully assess the response.
Recovery of Spermatogenesis After TRT Discontinuation
For men who have been on TRT without gonadotropin support and wish to conceive, discontinuing TRT and initiating a recovery protocol is often necessary. The recovery timeline varies significantly. Studies have shown that median time to recovery of spermatogenesis after testosterone-based contraceptive protocols is approximately 3 to 6 months, but some men require 12 to 24 months, and a small percentage (approximately 5%) may never fully recover baseline sperm parameters.
Factors that influence recovery include the duration of TRT use (longer duration is associated with slower recovery), the testosterone dose (higher doses cause more profound suppression), patient age (older men recover more slowly), baseline fertility status (men with pre-existing subfertility may not recover as well), and concurrent use of other suppressive agents like nandrolone, which is particularly resistant to recovery. The use of gonadorelin during the recovery period may help accelerate HPG axis reactivation by providing direct pituitary stimulation, though controlled data supporting this specific application are limited.
Emerging Research: Novel Approaches to Male Fertility Preservation
The field of male fertility preservation during hormonal therapy is evolving rapidly. Several emerging approaches deserve mention. First, selective androgen receptor modulators (SARMs) are being investigated as potential alternatives to testosterone that might provide anabolic benefits without the same degree of HPG axis suppression. However, clinical data suggest that SARMs do suppress gonadotropins, albeit sometimes less completely than testosterone. Second, testosterone nasal gel (Natesto) has shown less gonadotropin suppression than injectable testosterone in some studies, potentially because the short peak and rapid clearance of intranasal testosterone mimics pulsatile delivery. Third, combination protocols using gonadorelin with enclomiphene or low-dose HCG represent a multi-target approach to fertility preservation that is being explored in clinical practice.
Special Population: Athletes and Performance-Oriented Men
Athletes and physically active men represent a distinct subpopulation of TRT and gonadorelin users. Some athletes use TRT under medical supervision for documented hypogonadism, particularly common in combat sports, endurance athletes, and formerly obese individuals who have lost significant weight. These men often have heightened concerns about testicular function and fertility due to their younger age demographic and the physical demands of their sport.
For athletes on medically supervised TRT, gonadorelin serves the same testicular preservation function as in non-athletic populations. However, several sport-specific considerations apply. First, athletes should verify gonadorelin's status with their sport's governing body before use, as some athletic organizations may classify it differently. The World Anti-Doping Agency (WADA) does not specifically list gonadorelin as a prohibited substance, but regulations can change and vary by organization. Second, the higher metabolic demands of intense training can affect HPG axis function independently. Overtraining syndrome, relative energy deficiency in sport (RED-S), and exercise-associated hypogonadotropic hypogonadism are recognized conditions that can compound the suppressive effects of TRT on the reproductive axis.
Third, body composition goals common among athletes (low body fat, high muscle mass) interact with reproductive endocrine function. Very low body fat reduces leptin signaling, which can impair GnRH pulsatility. Conversely, adequate body fat and energy intake support reproductive function. Athletes on TRT with gonadorelin should ensure they maintain sufficient caloric intake and body fat percentage to support the HPG axis signaling that gonadorelin is trying to maintain.
The Female Athlete Triad and Pulsatile GnRH
The female athlete triad (now more broadly termed RED-S, relative energy deficiency in sport) consists of low energy availability, menstrual dysfunction (functional hypothalamic amenorrhea), and low bone mineral density. The central feature of the triad relevant to this discussion is functional hypothalamic amenorrhea caused by suppressed GnRH pulsatility due to inadequate energy availability.
Pulsatile GnRH therapy can restore menstrual function and fertility in women with exercise-associated hypothalamic amenorrhea. However, the fundamental intervention is correcting the energy deficit. Treating the GnRH deficiency with exogenous gonadorelin without addressing the underlying energy imbalance is treating the symptom rather than the cause. Best practice involves nutritional rehabilitation to restore energy availability, behavioral modification to reduce exercise intensity or increase caloric intake, psychological support (eating disorders are common in this population), and pulsatile GnRH therapy if fertility is desired and cannot wait for natural recovery of the HPG axis.
This population highlights the importance of viewing gonadorelin therapy within the context of the whole patient. The peptide is a tool, not a complete solution. Addressing the underlying factors that contribute to GnRH deficiency, whether they are metabolic, psychological, pharmacological (TRT), or pathological (pituitary disease), is always part of optimal clinical management.
For detailed information on peptides used in hormonal optimization, explore the Biohacking Hub. The dosing calculator can also help with personalized protocol planning.
TRT Support Applications

Figure 5: Clinical applications of gonadorelin in men using testosterone replacement therapy for testicular function and hormonal support.
Testosterone replacement therapy corrects the symptoms of hypogonadism, including fatigue, reduced libido, decreased muscle mass, depressed mood, and cognitive difficulties. But TRT comes with an inherent trade-off: the exogenous testosterone that resolves these symptoms also suppresses the very system that produces testosterone naturally. This section focuses specifically on how gonadorelin is used in clinical practice to mitigate the downsides of TRT.
Why TRT Suppresses Endogenous Production
The mechanism of TRT-induced HPG axis suppression is direct and predictable. Exogenous testosterone elevates serum testosterone to supraphysiological levels relative to what the hypothalamic-pituitary unit expects. The elevated testosterone (and its aromatization product, estradiol) acts on hypothalamic GnRH neurons and pituitary gonadotrophs to suppress GnRH pulse frequency and amplitude, reduce GnRH receptor expression on gonadotrophs, and directly inhibit LH-beta and FSH-beta gene transcription.
Within 2 to 4 weeks of starting TRT, serum LH falls to less than 1 mIU/mL in most men (normal range: 1.7 to 8.6 mIU/mL). FSH follows a similar trajectory. Without LH stimulation, intratesticular testosterone production drops precipitously. A study by Page et al. demonstrated that intratesticular testosterone levels dropped by 94% in healthy men receiving exogenous testosterone without concurrent gonadotropin support (Page ST, et al. Intratesticular androgens and spermatogenesis during severe gonadotropin suppression induced by male hormonal contraceptive treatment. Journal of Andrology. 2007;28(5):734-741. DOI: 10.2164/jandrol.107.002790).
Practical Gonadorelin Use in TRT Protocols
In current clinical practice, gonadorelin is prescribed as an adjunct to TRT with the goals of maintaining some degree of pituitary LH/FSH response, preventing or reducing testicular atrophy, preserving some level of intratesticular testosterone production, and maintaining the potential for future spermatogenesis recovery. The most common TRT-support protocols involve either subcutaneous injection of 100 to 200 mcg once daily (typically before bed) or 50 to 100 mcg twice daily (morning and evening).
Clinicians who specialize in TRT report several practical observations about gonadorelin co-therapy. First, testicular volume preservation is partial but noticeable. Men using gonadorelin alongside TRT typically report less scrotal shrinkage compared to TRT alone, though the maintenance is not as complete as with HCG co-therapy. Second, some men report subjective improvements in libido, mood, and overall well-being when gonadorelin is added to TRT, potentially due to the endogenous LH-stimulated intratesticular testosterone and the direct effects of FSH on Sertoli cell-derived factors.
Third, there is significant individual variability in response. Some men maintain measurable LH and FSH levels on gonadorelin during TRT, while others show minimal response. Factors that may influence individual response include the dose and type of testosterone used (higher doses cause more profound suppression), the patient's age and baseline pituitary function, genetic variation in GnRH receptor expression and signaling, and the degree of aromatization to estradiol (estradiol is a potent suppressor of pituitary gonadotropin secretion).
Monitoring Gonadorelin Therapy During TRT
Appropriate laboratory monitoring is essential for men using gonadorelin during TRT. The following parameters should be assessed at baseline, then every 3 to 6 months.
| Test | Purpose | Target Range | Interpretation |
|---|---|---|---|
| Serum LH | Assess pituitary response to gonadorelin | Above 1.0 mIU/mL | If undetectable, gonadorelin may not be overcoming TRT suppression |
| Serum FSH | Evaluate FSH component of response | Above 1.0 mIU/mL | FSH is needed for Sertoli cell function and spermatogenesis |
| Total Testosterone | Confirm adequate TRT dosing | 700-1100 ng/dL | Very high levels cause greater HPG suppression |
| Estradiol | Monitor aromatization | 20-40 pg/mL | High estradiol contributes to pituitary suppression |
| Testicular Volume (ultrasound) | Assess structural preservation | Within 20% of baseline | Significant loss suggests inadequate gonadotropin support |
| Semen Analysis (if relevant) | Evaluate spermatogenesis | Above 15 million/mL | May be reduced but ideally not azoospermic |
Gonadorelin vs HCG in TRT: A Practical Comparison
For men and clinicians choosing between gonadorelin and HCG as TRT adjuncts, several practical differences deserve consideration. HCG bypasses the pituitary entirely, acting directly on the LH receptor. This means it works regardless of how suppressed the pituitary is by exogenous testosterone. Gonadorelin, in contrast, requires a functional pituitary response, which is blunted during TRT. This fundamental difference explains why HCG generally provides better testicular function preservation.
However, gonadorelin does have certain practical advantages. It provides FSH stimulation in addition to LH, which HCG does not. It has a lower risk of LH receptor desensitization, since it works through the physiological pathway rather than providing chronic, direct receptor stimulation. The cost is generally lower than brand-name HCG products. And some clinicians report that patients feel that gonadorelin produces a more "natural" hormonal profile, though this is difficult to quantify.
A combined approach is also possible. Some TRT clinics prescribe both gonadorelin (for pituitary maintenance) and low-dose HCG (for direct testicular support), aiming to get the benefits of both mechanisms. While no controlled trials have evaluated this combination, it is used in clinical practice. The addition of growth hormone secretagogues to TRT protocols is another area of growing clinical interest for body composition optimization.
Post-Cycle Therapy (PCT) Applications
Gonadorelin is increasingly used in post-cycle therapy protocols by men discontinuing anabolic-androgenic steroids (AAS) or coming off TRT. The rationale is that pulsatile GnRH stimulation can help "restart" the suppressed HPG axis by directly activating pituitary gonadotrophs that have been dormant during the period of exogenous hormone use.
A typical PCT protocol incorporating gonadorelin might include gonadorelin 100 to 200 mcg subcutaneously twice daily for 2 to 4 weeks during the initial recovery phase, combined with a SERM like clomiphene or enclomiphene to block estrogen negative feedback, and potentially HCG for direct testicular stimulation in the early post-cycle period. This multi-agent approach targets HPG axis recovery at multiple levels simultaneously.
The timing of gonadorelin in PCT is important. It is typically started after the exogenous testosterone has substantially cleared, since giving gonadorelin while serum testosterone is still supraphysiological will produce minimal pituitary response due to ongoing negative feedback. For testosterone cypionate/enanthate, this means starting PCT approximately 2 weeks after the last injection. For testosterone undecanoate, the clearance period is longer (4 to 6 weeks).
The theoretical advantage of including gonadorelin in PCT, compared to using a SERM alone, is that gonadorelin provides direct pituitary stimulation. A SERM works by blocking estrogen negative feedback at the hypothalamus and pituitary, which indirectly increases GnRH pulsatility and gonadotropin secretion. But this mechanism depends on the hypothalamus being capable of increasing GnRH output when estrogen feedback is removed. In men with prolonged AAS use, hypothalamic GnRH neurons may be particularly sluggish to recover, making direct GnRH stimulation with gonadorelin a useful adjunct. The combination of a SERM (removing estrogen brake) plus gonadorelin (providing direct GnRH stimulation) addresses both levels of the suppression simultaneously.
Empirical PCT protocols vary widely among practitioners, and evidence from controlled clinical trials is essentially absent. The following represents a commonly used multi-phase approach for men coming off moderate-dose testosterone or AAS cycles lasting less than 12 months.
| Phase | Duration | Medications | Goal |
|---|---|---|---|
| Phase 1: Bridge | Weeks 1-2 after last T injection | HCG 1500-2000 IU EOD | Maintain testicular stimulation during T clearance |
| Phase 2: Core PCT | Weeks 3-6 | Gonadorelin 200 mcg BID + enclomiphene 25 mg QD | Restart pituitary and remove estrogen brake |
| Phase 3: Taper | Weeks 7-8 | Gonadorelin 100 mcg QD + enclomiphene 12.5 mg QD | Gradual withdrawal while monitoring recovery |
| Phase 4: Assessment | Week 10-12 | None (washout period) | Reassess hormone levels off all medications |
Lab work should be drawn at the beginning of Phase 2 (to establish the suppressed baseline), at the end of Phase 3 (to assess initial recovery), and 4 to 6 weeks after discontinuing all PCT medications (to confirm sustained recovery). The target is LH and FSH in the normal range (not just detectable), total testosterone above 300 ng/dL (ideally above 450), and progressive improvement in symptoms. If recovery is incomplete at the Phase 4 assessment, an extended course of PCT or referral to an endocrinologist may be warranted.
Special Considerations: Age and Pituitary Reserve
The effectiveness of gonadorelin during TRT may decline with age. Older men tend to have reduced pituitary gonadotroph reserve and diminished responsiveness to GnRH stimulation even at baseline. When the additional suppressive burden of exogenous testosterone is added, the pituitary's capacity to respond to gonadorelin may be further compromised. For men over 50 on TRT, clinicians should set realistic expectations about the degree of testicular function preservation achievable with gonadorelin and consider HCG as the primary adjunct if fertility is a priority.
The free assessment at FormBlends can help determine the most appropriate protocol based on individual health profiles and goals.
Estrogen Management During TRT with Gonadorelin
One often-overlooked aspect of using gonadorelin during TRT is its potential impact on estradiol levels. By stimulating endogenous LH release and maintaining some intratesticular testosterone production, gonadorelin adds to the total androgenic load. This additional testosterone production, combined with the exogenous testosterone from TRT, increases the total substrate available for aromatization to estradiol. Some men may experience elevated estradiol levels when gonadorelin is added to their TRT protocol, which can manifest as water retention, gynecomastia, mood changes, or increased blood pressure.
Clinicians managing men on TRT with gonadorelin should monitor estradiol levels and be prepared to adjust the protocol if estradiol rises above the desired range (typically 20 to 40 pg/mL, though optimal ranges are debated). Options for managing elevated estradiol include reducing the TRT dose (since the gonadorelin-stimulated endogenous testosterone partially supplements the exogenous dose), reducing the gonadorelin dose, or in some cases, using a low-dose aromatase inhibitor like anastrozole 0.25 to 0.5 mg twice weekly. However, the routine use of aromatase inhibitors with TRT is controversial, and many experts recommend optimizing TRT dosing before adding an AI. The interplay between exogenous testosterone, gonadorelin-stimulated endogenous testosterone, and aromatization adds a layer of complexity to hormone management that requires careful monitoring and individualization.
Testicular Atrophy: Causes, Consequences, and Prevention
Testicular atrophy is one of the most common and distressing side effects of TRT for many men. The testes shrink primarily because the Leydig cells and Sertoli cells atrophy in the absence of adequate gonadotropin stimulation. Leydig cells normally comprise about 10 to 15% of testicular volume, and their function is entirely dependent on LH stimulation. Without LH, Leydig cells undergo involution, with reduced cell size, decreased steroidogenic enzyme expression, and diminished organelle development. Sertoli cells, while more resistant to atrophy, also decline in function without FSH stimulation, leading to reduced seminiferous tubule diameter and decreased spermatogenic activity.
Beyond the cosmetic concern, testicular atrophy has functional implications. The smaller, less active testes produce less intratesticular testosterone, less inhibin B, less INSL3 (insulin-like factor 3, a Leydig cell marker), and fewer germ cells. INSL3 is particularly interesting as a biomarker: it is produced exclusively by mature Leydig cells, does not respond to HCG or LH acutely, and provides an integrated measure of long-term Leydig cell function. Some clinicians use serum INSL3 levels as an additional marker of testicular health in men on TRT.
Gonadorelin's role in preventing testicular atrophy is to maintain some degree of gonadotropin-driven Leydig and Sertoli cell stimulation. Clinical experience suggests that men using gonadorelin alongside TRT experience less scrotal shrinkage and maintain slightly larger testicular volume compared to men on TRT alone. However, the protection is partial, and men should have realistic expectations. For those who are particularly concerned about testicular size, HCG remains the more reliable option, or a combination of HCG and gonadorelin can be used.
Hormone Optimization: The Complete TRT Ecosystem
In modern men's health practice, TRT is rarely prescribed in isolation. A comprehensive hormone optimization protocol may include testosterone (injection, gel, or pellet), gonadorelin or HCG for testicular support, an aromatase inhibitor if estradiol is elevated, DHEA supplementation if DHEA-S is low, thyroid optimization if thyroid function is suboptimal, growth hormone secretagogues like CJC-1295/ipamorelin or sermorelin for body composition and recovery, and lifestyle interventions including exercise, sleep optimization, and stress management.
Within this ecosystem, gonadorelin serves a specific niche: maintaining the HPG axis connection between the pituitary and the testes. It does not replace testosterone, does not serve as a primary testosterone-boosting agent during TRT, and should not be confused with a standalone treatment for hypogonadism. Its role is adjunctive, supportive, and preservative. When patients and clinicians understand this role clearly, expectations are realistic, and outcomes are more satisfying.
The concept of "total hormone optimization" also extends to metabolic health. The relationship between testosterone levels and metabolic parameters like insulin sensitivity, body composition, and cardiovascular risk is bidirectional. GLP-1 receptor agonists and other metabolic therapies can complement hormonal optimization by addressing the metabolic component of health. The Lifestyle Hub provides additional resources on integrating hormonal and metabolic health strategies.
Diagnostic Use
Figure 6: The GnRH stimulation test protocol with timed blood draws measuring LH and FSH response to a gonadorelin bolus.
The GnRH stimulation test is one of the oldest and most widely used diagnostic applications of gonadorelin. By administering a standardized dose of synthetic GnRH and measuring the subsequent LH and FSH response, clinicians can assess the functional capacity of pituitary gonadotroph cells and help distinguish between hypothalamic and pituitary causes of hypogonadism.
The GnRH Stimulation Test: Standard Protocol
The standard GnRH stimulation test involves the following steps. The patient fasts overnight (though fasting is not strictly required for the test itself, it reduces the variability of baseline gonadotropin levels). A baseline blood sample is drawn for LH and FSH measurement. Gonadorelin 100 mcg is administered as a rapid intravenous bolus (or subcutaneously in some protocols). Additional blood samples are drawn at 15, 30, 45, 60, and sometimes 90 and 120 minutes post-injection. LH and FSH are measured at each time point.
The normal LH response to 100 mcg IV gonadorelin peaks at 15 to 30 minutes and reaches approximately 3 to 5 times the baseline level. FSH response is more modest, typically increasing by 1.5 to 2.5 times baseline, with a delayed peak at 30 to 60 minutes. The chart data for this report illustrates a typical normal LH response: from a baseline of 5.2 mIU/mL, LH rises to 18.5 at 15 minutes, peaks at 25.3 at 30 minutes, declines to 20.1 at 60 minutes, and returns toward baseline at 12.4 by 120 minutes.
LH Response to Gonadorelin 100mcg IV
Diagnostic Interpretation: What the Response Tells You
The pattern and magnitude of the LH response to GnRH stimulation provides valuable diagnostic information across several clinical scenarios.
Normal Response
A brisk LH rise to 3 to 5 times baseline (peak typically above 10 to 15 mIU/mL in adults) indicates intact pituitary gonadotroph function. This response is expected in healthy individuals and in patients with hypothalamic causes of hypogonadism (where the pituitary is intact but not receiving adequate endogenous GnRH stimulation).
Blunted or Absent Response
A minimal or no LH response to GnRH stimulation suggests pituitary gonadotroph damage or dysfunction. This pattern is seen in pituitary adenomas, post-surgical pituitary damage, pituitary apoplexy, hypophysitis, and radiation-induced pituitary injury. In these cases, the gonadotroph cells are either destroyed or unable to respond to GnRH, confirming a pituitary (rather than hypothalamic) cause of hypogonadism.
Exaggerated Response
An excessively high LH response may indicate primed gonadotrophs that have been chronically understimulated and have accumulated large stores of LH ready for release. This pattern can be seen in hypothalamic amenorrhea and certain forms of delayed puberty. It can also be seen after prolonged GnRH agonist desensitization, when the drug is discontinued and the gonadotrophs "rebound."
Response in Central Precocious Puberty
The GnRH stimulation test is the gold standard for diagnosing central precocious puberty (CPP). In prepubertal children, the normal response to GnRH is dominated by FSH, with relatively little LH response. In CPP, the response shifts to a pubertal pattern, with LH predominating. Diagnostic cutoffs vary by assay, but a stimulated LH peak above 5 mIU/mL at 30 to 60 minutes is widely accepted as confirmatory of CPP, with sensitivity of 98% and specificity approaching 100% in most studies (Kim HK, et al. GnRH stimulation test and diagnostic cutoff in precocious puberty. Annals of Pediatric Endocrinology & Metabolism. 2020;25(3):142-149. DOI: 10.6065/apem.2040072.036).
Distinguishing Hypothalamic from Pituitary Disease
One of the primary clinical uses of the GnRH stimulation test is distinguishing hypothalamic hypogonadism (inadequate GnRH production or release) from pituitary hypogonadism (gonadotroph cell dysfunction). This distinction has important implications for treatment, because hypothalamic causes are potentially treatable with pulsatile GnRH therapy, while pituitary causes require direct gonadotropin replacement.
However, interpretation requires caution. In patients with long-standing hypothalamic GnRH deficiency (such as congenital hypogonadotropic hypogonadism), the pituitary gonadotrophs may be "dormant" and respond poorly to a single GnRH bolus, mimicking pituitary disease. In these cases, a priming protocol is used: the patient receives repeated GnRH pulses (typically 100 mcg subcutaneously every 90 minutes for several days) before the formal stimulation test. If the pituitary "wakes up" after priming and responds normally to GnRH, this confirms a hypothalamic cause. Failure to respond after adequate priming suggests intrinsic pituitary pathology.
The GnRH Stimulation Test in Delayed Puberty
Distinguishing constitutional delay of growth and puberty (CDGP) from congenital hypogonadotropic hypogonadism in adolescent boys is one of the most challenging diagnostic dilemmas in pediatric endocrinology. Both conditions present with delayed puberty, prepubertal testosterone levels, and low gonadotropins. The GnRH stimulation test can help differentiate these conditions, though its sensitivity is imperfect.
Boys with CDGP typically show a prepubertal GnRH response (FSH-dominant) that normalizes when puberty eventually begins spontaneously. Boys with CHH show a flat or severely blunted response. However, overlap exists, and a single GnRH stimulation test cannot always distinguish the two conditions. Serial testing over time, combined with other clinical features (family history, anosmia, presence of micropenis or cryptorchidism), provides better diagnostic accuracy (Harrington J, et al. Role of GnRH stimulation testing in diagnosing gonadotropin deficiency in delayed puberty. Indian Journal of Endocrinology and Metabolism. 2016;20(1):95-99. DOI: 10.4103/2230-8210.172245).
Interpretation Nuances: Age and Sex-Specific Considerations
The interpretation of GnRH stimulation test results requires careful consideration of age, sex, and pubertal stage. In prepubertal children (Tanner stage 1), the normal response to GnRH is characterized by a predominant FSH response with relatively little LH rise. As puberty progresses, the response shifts to LH-predominant, reflecting the maturation of the GnRH pulse generator and the development of the positive feedback mechanism. The LH:FSH ratio after GnRH stimulation is therefore a key marker of pubertal activation.
In adult men, the LH response to GnRH is typically brisk and short-lived, with a peak at 15 to 30 minutes. In premenopausal women, the response depends on the menstrual cycle phase. During the early follicular phase, the LH response is moderate. In the late follicular phase, the response is enhanced (due to estradiol priming of the gonadotrophs), and in the luteal phase, the response is suppressed (due to the combined negative feedback of estradiol and progesterone). Testing is therefore standardized to the early follicular phase when possible.
In postmenopausal women, baseline gonadotropin levels are already elevated due to the loss of gonadal negative feedback. The GnRH stimulation test in this context assesses the pituitary's capacity for further gonadotropin release above already-elevated baseline levels. A blunted response in a postmenopausal woman (who should have high gonadotropins) raises concern for pituitary disease. Similarly, in elderly men, baseline gonadotropins may be modestly elevated as part of normal aging, and the GnRH response may be attenuated compared to younger men.
Prolonged GnRH Stimulation Test
The prolonged GnRH stimulation test involves administering gonadorelin repeatedly over 5 to 7 days (typically 100 mcg IV or SC every 90 minutes via pump) and then performing the standard bolus test. This protocol assesses not just acute LH release from stored pools but also the capacity of the gonadotroph to synthesize new LH in response to sustained GnRH stimulation. The prolonged test has better diagnostic accuracy than the single-bolus test for distinguishing hypothalamic from pituitary hypogonadism, particularly in patients with long-standing GnRH deficiency.
Clinical Case Scenarios: Applying the GnRH Stimulation Test
To illustrate the practical utility of the GnRH stimulation test, consider several representative clinical scenarios that demonstrate how test results guide clinical decision-making.
Scenario 1: Young Man with Unexplained Low Testosterone
A 28-year-old man presents with fatigue, decreased libido, and total testosterone of 180 ng/dL (normal: 300 to 1000). LH is 2.1 mIU/mL and FSH is 1.8 mIU/mL, indicating secondary (hypogonadotropic) hypogonadism. The GnRH stimulation test shows a peak LH of 22 mIU/mL at 30 minutes, confirming intact pituitary gonadotroph function. This result suggests a hypothalamic cause of the hypogonadism, prompting further evaluation for hypothalamic pathology (MRI to rule out infiltrative disease, assessment of other pituitary axes, evaluation for energy deficit or excessive exercise). The good pituitary response also confirms that pulsatile GnRH therapy or treatment with a SERM could be effective alternatives to TRT for this patient, potentially allowing him to maintain fertility while treating his hypogonadism.
Scenario 2: Post-Pituitary Surgery Assessment
A 45-year-old man underwent transsphenoidal surgery for a non-functioning pituitary adenoma 6 months ago. Post-surgical testosterone is 220 ng/dL with LH 0.5 mIU/mL. The GnRH stimulation test shows a peak LH of only 3 mIU/mL, a severely blunted response. This confirms post-surgical gonadotroph damage, indicating that pulsatile GnRH therapy would not be effective (the gonadotroph cells are damaged, not simply understimulated). Treatment would require either TRT (with or without gonadorelin) or direct gonadotropin replacement if fertility is desired.
Scenario 3: Adolescent Boy with Delayed Puberty
A 14-year-old boy has no pubertal development (Tanner stage 1), bone age delayed by 2 years, and testosterone of 20 ng/dL. Family history reveals that his father was a "late bloomer" who started puberty at 15. The GnRH stimulation test shows a prepubertal pattern with FSH-dominant response (FSH rises more than LH). This result is consistent with either constitutional delay of growth and puberty (CDGP) or congenital hypogonadotropic hypogonadism (CHH). A prolonged priming protocol may help differentiate: after 5 to 7 days of pulsatile GnRH, if the LH response normalizes, CDGP is more likely. If it remains blunted, CHH is the concern. In practice, many clinicians elect watchful waiting with repeat testing in 6 to 12 months, as a significant proportion of boys with CDGP will begin puberty spontaneously by age 15 to 16.
Scenario 4: Girl with Early Breast Development
A 7-year-old girl presents with breast development (Tanner stage 2) and growth acceleration. Bone age is advanced by 1.5 years. The GnRH stimulation test shows a stimulated LH of 8.5 mIU/mL at 30 minutes, well above the 5 mIU/mL diagnostic threshold for central precocious puberty. This confirms CPP and provides justification for treatment with a GnRH agonist (such as leuprolide depot) to suppress pubertal progression and preserve adult height potential.
GnRH Agonist Stimulation Test: A Modern Alternative
In many clinical settings, the native GnRH stimulation test has been partially replaced by GnRH agonist stimulation tests using agents like triptorelin or leuprolide. These synthetic agonists have several practical advantages: longer half-lives (eliminating the need for precise timing of the injection), the ability to use subcutaneous injection instead of IV, and commercial availability. For diagnosing CPP, a subcutaneous triptorelin stimulation test (with LH measurement at 3 and 6 hours) has been validated as an accurate alternative to the classic gonadorelin test.
However, the native GnRH test using gonadorelin retains advantages in specific situations. Its short half-life makes it better suited for evaluating pulsatile pituitary responses, and it does not cause the prolonged gonadotropin stimulation that GnRH agonists produce. For research applications requiring precise assessment of pituitary responsiveness, the native GnRH test remains the preferred approach.
Limitations and Pitfalls
Several important limitations of the GnRH stimulation test deserve emphasis. The test has limited sensitivity for mild forms of hypogonadotropic hypogonadism. The LH response varies with assay methodology, and results from different immunoassays are not directly comparable. The timing of blood draws matters significantly, as missing the peak LH level can lead to underestimation of pituitary function. In women, the test result is influenced by the phase of the menstrual cycle and estrogen levels. And in men on TRT or anabolic steroids, the test will show a suppressed response due to negative feedback, not pituitary disease.
The GnRH Stimulation Test in Clinical Practice: Step-by-Step Walkthrough
For clinicians performing the GnRH stimulation test, a detailed practical walkthrough is valuable. The test should be performed in the morning (between 8:00 and 10:00 AM) after an overnight fast. The patient should be seated or recumbent for at least 15 minutes before baseline sampling to minimize stress-related gonadotropin fluctuations. An intravenous line should be placed for both gonadorelin administration and serial blood sampling, though some protocols use separate IV access and subcutaneous gonadorelin injection.
The baseline blood sample is drawn and should include LH, FSH, testosterone (in males), estradiol (in females), and prolactin. Prolactin is measured at baseline because hyperprolactinemia is a common cause of hypogonadotropic hypogonadism, and diagnosing it changes the treatment approach entirely (dopamine agonist therapy rather than GnRH or gonadotropin replacement).
Gonadorelin 100 mcg is then administered as a rapid IV push over 15 to 30 seconds. The timing of the injection should be recorded precisely, as all subsequent blood draws are timed relative to this moment. Blood samples are collected at exactly 15, 30, 45, 60, 90, and 120 minutes post-injection. Each sample is processed promptly and sent for LH and FSH measurement. Some protocols include additional time points (5 and 10 minutes) for research purposes, but these are not required for standard clinical interpretation.
Results are plotted as a response curve (LH and FSH versus time), and the peak LH value, time to peak, and LH:FSH ratio are documented. The results are interpreted in the context of the patient's age, sex, clinical presentation, and any concurrent medications. A written report should document the baseline values, the peak response, the pattern of response (normal, blunted, exaggerated, or absent), and the clinical conclusion.
Use in Evaluating Pituitary Recovery After Treatment
The GnRH stimulation test is also useful for assessing pituitary recovery after treatments that suppress or damage the HPG axis. After pituitary surgery (particularly transsphenoidal surgery for pituitary adenomas), the test can evaluate whether gonadotroph function has been preserved. Serial testing at 3, 6, and 12 months post-surgery can track recovery over time. Similarly, after pituitary radiation therapy, which can cause progressive hypopituitarism over months to years, periodic GnRH stimulation testing can detect the development of gonadotropin deficiency before it becomes clinically apparent.
In patients recovering from GnRH agonist therapy (for example, after treatment of prostate cancer or endometriosis), the stimulation test can confirm that the pituitary has recovered from desensitization. The expected pattern is a gradual return of the LH response over weeks to months after agonist discontinuation. Failure to recover pituitary responsiveness after an adequate washout period (typically 2 to 3 months after the last agonist dose) may indicate underlying pituitary pathology rather than residual agonist effect.
The GnRH Stimulation Test in Transgender Medicine
An increasingly relevant application of the GnRH stimulation test is in transgender medicine, where it serves several diagnostic and monitoring functions. For transgender youth being evaluated for puberty suppression with GnRH agonists, the stimulation test can confirm pubertal activation (an elevated LH response indicates Tanner stage 2 or greater) and establish a baseline pituitary response before treatment initiation. During GnRH agonist therapy, periodic stimulation testing can verify adequate suppression (a flat LH response confirms the agonist is achieving the desired effect). And upon discontinuation of GnRH agonists (if the individual chooses to stop treatment), the stimulation test can assess the recovery of pituitary function and predict the timeline for pubertal resumption.
For transgender men (female-to-male) receiving testosterone therapy, the HPG axis is suppressed similarly to cisgender men on TRT. If fertility preservation is a concern, the same considerations about gonadorelin or HCG adjunct therapy apply, though the clinical context and goals may differ. For transgender women (male-to-female) receiving estrogen and anti-androgen therapy, the GnRH stimulation test can assess the degree of HPG axis suppression and guide decisions about the need for GnRH agonist addition to the feminizing protocol.
The use of GnRH stimulation testing in transgender medicine requires sensitivity to the patient's gender identity and awareness that the hormonal reference ranges for test interpretation should be aligned with the patient's affirmed gender and current hormonal milieu rather than their sex assigned at birth.
Alternative Diagnostic Approaches
While the GnRH stimulation test remains valuable, alternative diagnostic approaches have emerged. The clomiphene citrate challenge test (CCCT) evaluates HPG axis integrity by blocking estrogen negative feedback and measuring the subsequent rise in gonadotropins. It is simpler to perform (oral medication, single blood draw) but provides less detailed information about pituitary dynamics. For evaluating male hypogonadism, basal gonadotropin levels alone are often sufficient for diagnosis (elevated LH/FSH with low testosterone indicates primary hypogonadism; low or inappropriately normal LH/FSH with low testosterone indicates secondary hypogonadism). The GnRH stimulation test adds value primarily when the basal results are ambiguous or when distinguishing hypothalamic from pituitary pathology is clinically important.
Anti-Mullerian hormone (AMH) has emerged as an additional biomarker that can complement GnRH stimulation testing. In boys with delayed puberty, AMH levels can help differentiate constitutional delay (where AMH is typically normal) from CHH (where AMH may be elevated due to the absence of testosterone's inhibitory effect on Sertoli cell AMH production). In girls, AMH provides information about ovarian reserve that is independent of gonadotropin status. The combination of GnRH stimulation testing, AMH measurement, and clinical evaluation provides the most accurate diagnostic assessment of reproductive endocrine disorders. For resources on the science behind peptide therapies, FormBlends provides additional educational materials.
Dosing Protocols
Figure 7: Dosing protocols for gonadorelin vary depending on the clinical application, from diagnostic testing to fertility therapy to TRT support.
Gonadorelin dosing varies substantially depending on the clinical application. This section provides detailed practical guidance for each major use case, covering reconstitution, injection technique, dosing schedules, and monitoring parameters. All protocols should be followed under medical supervision. Use the FormBlends dosing calculator for personalized guidance.
Reconstitution and Preparation
Gonadorelin is supplied as a lyophilized (freeze-dried) powder in vials containing 2 mg (2,000 mcg). Proper reconstitution is essential for dosing accuracy and product stability.
Step-by-Step Reconstitution
- Materials needed: Gonadorelin vial (2 mg), bacteriostatic water for injection (containing 0.9% benzyl alcohol), sterile syringe and needle (18G or 20G for reconstitution), alcohol swabs.
- Draw diluent: Using a sterile syringe, draw 2.0 mL of bacteriostatic water. This yields a concentration of 1.0 mg/mL (1,000 mcg/mL). Alternatively, add 1.0 mL for a concentration of 2.0 mg/mL (2,000 mcg/mL).
- Add to vial: Inject the bacteriostatic water slowly down the side of the vial. Do not spray directly onto the powder, as this can cause foaming.
- Mix gently: Swirl or roll the vial gently until the powder is completely dissolved. Do not shake, as this can degrade the peptide and cause aggregation.
- Inspect: The reconstituted solution should be clear and colorless. Do not use if cloudy, discolored, or if particulate matter is visible.
Storage After Reconstitution
| Condition | Lyophilized Powder | Reconstituted Solution |
|---|---|---|
| Refrigerator (2-8C / 36-46F) | Up to 2 years (check expiry date) | 14-28 days |
| Freezer (-20C / -4F) | Up to 3+ years | Do NOT freeze reconstituted solution |
| Room temperature (20-25C) | Acceptable short-term (weeks) | Use within 4-6 hours only |
| Light protection | Store in original packaging | Keep vial in light-protected container |
Dosing Protocol: TRT Support (Men's Health)
This is currently the most common application of gonadorelin in the United States. The goal is to maintain pituitary gonadotroph function and testicular health during concurrent testosterone replacement.
Standard TRT-Support Protocol
- Dose: 100 to 200 mcg per injection
- Frequency: Once daily (before bed preferred) or twice daily (morning and evening)
- Route: Subcutaneous injection (abdomen, thigh, or upper arm)
- Needle: 29G or 30G insulin syringe, 1/2 inch
- Cycle: Continuous use alongside TRT (no cycling needed)
- Starting dose: Begin at 100 mcg daily, assess LH response at 4 weeks, and increase to 200 mcg daily if LH remains undetectable
Enhanced Protocol for Fertility-Focused Men
- Dose: 200 mcg per injection
- Frequency: Twice daily (12 hours apart)
- Monitoring: Monthly semen analysis and LH/FSH every 6-8 weeks
- Adjuncts: Consider adding HCG 500-1000 IU twice weekly if sperm parameters are declining despite gonadorelin
- Duration: At least 3 to 6 months before assessing spermatogenesis outcomes (full sperm production cycle is approximately 74 days)
Combination Protocols: Multi-Agent Approaches
In clinical practice, gonadorelin is rarely the only adjunct added to TRT. Clinicians frequently design multi-agent protocols that target different levels of the HPG axis simultaneously. Understanding the rationale for these combinations helps patients appreciate why their protocol may include multiple medications.
Gonadorelin + Low-Dose HCG
This combination provides both upstream (pituitary) and downstream (testicular) stimulation. Gonadorelin stimulates the pituitary to release LH and FSH, while HCG provides direct LH-receptor activation on Leydig cells. The rationale is that gonadorelin alone may not produce sufficient LH to maintain intratesticular testosterone during TRT, and supplementing with small amounts of HCG ensures a basal level of Leydig cell stimulation. A typical protocol might include gonadorelin 100 mcg daily plus HCG 250 to 500 IU every other day. This protocol uses lower doses of each agent than would be needed as monotherapy, potentially reducing cost and side effects while providing more comprehensive testicular support.
Gonadorelin + Enclomiphene
Combining a direct GnRH stimulus with estrogen receptor blockade targets two different points in the feedback loop. Gonadorelin provides exogenous GnRH signaling, while enclomiphene removes the estrogen-mediated brake on the hypothalamus and pituitary. The theoretical benefit is additive: the pituitary receives more GnRH (from gonadorelin) and is more responsive to it (because estrogen negative feedback is blocked by enclomiphene). This combination may produce higher LH and FSH output than either agent alone. A typical protocol includes gonadorelin 100 to 200 mcg daily plus enclomiphene 12.5 to 25 mg daily.
Gonadorelin + Enclomiphene + Low-Dose HCG (Triple Therapy)
Some fertility-focused TRT clinics employ a triple-agent approach for men who place high priority on fertility preservation while continuing testosterone therapy. This protocol targets three levels: GnRH receptor stimulation (gonadorelin), estrogen feedback blockade (enclomiphene), and direct Leydig cell stimulation (HCG). While comprehensive, this approach requires more medications, more injections, higher cost, and more intensive monitoring. It is typically reserved for men who have shown inadequate response to simpler protocols and who have an active desire for fertility in the near term.
Protocol Adjustments Based on Lab Results
The initial gonadorelin protocol should be viewed as a starting point that will likely need adjustment based on laboratory results. Here is a practical decision framework for protocol optimization.
| Lab Finding | Possible Interpretation | Protocol Adjustment |
|---|---|---|
| LH undetectable, FSH undetectable | TRT suppression overwhelming gonadorelin effect | Increase gonadorelin to 200 mcg BID; consider reducing T dose; consider adding enclomiphene or HCG |
| LH 1-3 mIU/mL, FSH 1-3 mIU/mL | Partial response; some pituitary activation | Current protocol providing meaningful stimulation; maintain or modestly increase dose |
| LH above 4 mIU/mL, FSH above 3 mIU/mL | Good pituitary response | Current protocol effective; maintain dose; consider reducing T dose if levels are very high |
| Estradiol above 50 pg/mL | Excessive aromatization from combined endogenous + exogenous T | Reduce T dose; consider AI (0.25 mg anastrozole twice weekly); monitor closely |
| Testicular volume declining despite gonadorelin | Insufficient gonadotropin support | Escalate to BID dosing; add HCG; re-evaluate fertility goals |
| Semen analysis: azoospermia despite 3+ months | Spermatogenesis not maintained | Consider adding HCG + FSH (or HMG); if fertility is urgent, consider stopping TRT |
This iterative, lab-guided approach ensures that each patient receives an individually optimized protocol. The key principle is that gonadorelin dosing is not one-size-fits-all, and the initial prescription is the beginning of a conversation between patient, clinician, and laboratory data.
Dosing Protocol: Pulsatile GnRH Pump Therapy (Female Fertility)
Pulsatile GnRH therapy requires a programmable microinfusion pump and is managed by reproductive endocrinologists. This protocol is included for reference and educational purposes.
Standard Pulsatile Protocol for Hypothalamic Amenorrhea
- Dose per pulse: 5 to 25 mcg (starting dose typically 5 to 10 mcg subcutaneously or 5 mcg intravenously)
- Pulse interval: Every 90 minutes (can be adjusted from 60 to 120 minutes based on response)
- Delivery: Programmable microinfusion pump (subcutaneous or intravenous via indwelling catheter)
- Monitoring: Serum estradiol and LH every 2-3 days initially, then weekly; transvaginal ultrasound to track follicular development
- Dose adjustment: Increase pulse dose by 5 mcg every 5-7 days if inadequate follicular response; decrease if multiple follicles develop
- Duration per cycle: Typically 10-21 days until ovulation is confirmed by LH surge or ultrasound
- Luteal phase support: Some protocols continue pulsatile GnRH through the luteal phase; others switch to progesterone supplementation
Dosing Considerations for Special Populations
Obese Patients
Obesity significantly affects GnRH physiology and the response to gonadorelin therapy. Obese men have lower testosterone levels due to increased aromatase activity in adipose tissue (converting testosterone to estradiol), elevated estradiol providing excessive negative feedback, increased insulin and insulin resistance affecting GnRH neuron function, and reduced sex hormone binding globulin (SHBG) levels. These factors collectively suppress the HPG axis, making obese men more likely to need TRT and potentially less responsive to gonadorelin as an adjunct.
For obese men on TRT with gonadorelin, dose adjustments may be needed. The higher body mass means that a standard dose may produce lower peak plasma concentrations. Some clinicians use higher starting doses (200 mcg daily rather than 100 mcg) for men with BMI above 30. Weight loss, whether through dietary intervention, exercise, or pharmacological support with tirzepatide or semaglutide, can improve the response to gonadorelin by reducing aromatase activity, lowering estradiol, and improving hypothalamic-pituitary sensitivity to GnRH. This creates a positive feedback loop where weight loss improves hormonal function, which in turn supports further metabolic improvement.
Men with Type 2 Diabetes
Type 2 diabetes is associated with a 2 to 3 times higher prevalence of hypogonadism compared to the general male population. The relationship is bidirectional: diabetes impairs HPG axis function through insulin resistance, chronic inflammation, and metabolic dysfunction, while hypogonadism worsens metabolic syndrome. Men with diabetes who are on TRT may benefit from gonadorelin adjunct therapy, but they should be aware that diabetic neuropathy can affect injection site sensation (making it harder to detect injection site reactions), poor glycemic control can impair GnRH receptor signaling and pituitary function, and metformin, commonly used for diabetes management, may have mild effects on reproductive hormones through AMPK activation in hypothalamic neurons.
Monitoring for diabetic men on TRT with gonadorelin should include more frequent metabolic panels (HbA1c, fasting glucose) in addition to the standard hormone panel, as testosterone levels can influence insulin sensitivity and glycemic control. The addition of liraglutide or other GLP-1 agonists for glycemic control and weight management creates a multi-pronged approach to metabolic and hormonal optimization that some clinics are increasingly adopting.
Men with Depression and Mood Disorders
Depression and hypogonadism share a bidirectional relationship, with each condition increasing the risk of the other. Selective serotonin reuptake inhibitors (SSRIs), commonly prescribed for depression, can affect GnRH pulsatility and gonadotropin secretion through serotonergic modulation of hypothalamic function. Some SSRIs have been associated with mildly reduced testosterone levels and sexual dysfunction, independent of their effects on libido through serotonin pathways.
For men with depression who are on TRT and considering gonadorelin, the potential mood benefits of maintaining testicular function (and the hormonal cascades it supports) may complement the mood benefits of TRT itself. However, clinicians should be aware that mood fluctuations can occur during the initiation of any hormonal therapy, and patients should be monitored for worsening mood symptoms. The anxiolytic peptides Selank and Semax are sometimes used alongside hormonal optimization protocols for their potential mood-supporting effects, though their combination with TRT and gonadorelin has not been studied in controlled settings.
Older Men (Over 60)
Age-related changes in HPG axis function include reduced GnRH pulse amplitude (though frequency may be maintained), decreased pituitary gonadotroph sensitivity to GnRH, reduced Leydig cell sensitivity to LH, and diminished testicular reserve for both testosterone production and spermatogenesis. These changes mean that older men may derive less benefit from gonadorelin therapy, as both the upstream (pituitary) and downstream (testicular) components of the response are attenuated.
For men over 60 on TRT, the primary goal of gonadorelin is typically testicular health maintenance rather than fertility preservation (though exceptions exist, particularly with increasing rates of delayed parenthood). Expectations should be calibrated accordingly. Some clinicians recommend lower gonadorelin doses (50 to 100 mcg daily) for older men, recognizing that the diminished pituitary response means higher doses may not provide proportionally greater benefit. Monitoring testicular volume via ultrasound every 6 to 12 months can provide objective data on whether the gonadorelin is achieving its intended effect.
Dosing Protocol: Diagnostic GnRH Stimulation Test
Standard Single-Bolus Test
- Dose: 100 mcg (adults) or 2.5 mcg/kg (pediatric)
- Route: Rapid intravenous bolus (preferred) or subcutaneous
- Blood draws: Baseline (time 0), then at 15, 30, 45, 60, 90, and 120 minutes
- Assays: LH and FSH at each time point
- Patient preparation: Fasting preferred but not required; morning testing preferred for consistency
- Note: In women, test should be performed in the early follicular phase (days 1-5) for standardized interpretation
Prolonged/Priming Protocol
- Priming phase: Gonadorelin 100 mcg subcutaneously every 90 minutes for 5 to 7 days (via pump or serial injections)
- Test phase: Standard single-bolus test (above) performed on day 7 or 8
- Interpretation: If LH response normalizes after priming, this confirms hypothalamic (not pituitary) cause of hypogonadism
Dosing Protocol: Congenital Hypogonadotropic Hypogonadism (Male Fertility Induction)
For men with confirmed CHH seeking fertility induction through pulsatile GnRH therapy.
Standard CHH Fertility Induction Protocol
- Dose per pulse: 5 to 25 mcg subcutaneously (start at 5-10 mcg, titrate based on response)
- Pulse interval: Every 120 minutes (some protocols use 90 minutes)
- Delivery: Programmable subcutaneous microinfusion pump
- Monitoring: Serum LH, FSH, testosterone every 2 weeks initially, then monthly; semen analysis every 4-8 weeks starting at month 3
- Expected timeline: LH/FSH response within days; testosterone normalization within 2-4 weeks; sperm appearance in ejaculate at 3-12 months (median 6 months)
- Pre-treatment: Some protocols recommend a 3-month course of testosterone or HCG to increase testicular volume before initiating pulsatile GnRH, as larger starting testicular volume predicts better spermatogenic outcomes
- Duration: Continue until pregnancy is achieved or until sperm banking is complete; may require 12-24 months for optimal sperm production
Dosing Protocol: Post-Cycle Therapy
For men recovering HPG axis function after discontinuing exogenous androgens.
PCT Protocol with Gonadorelin
- Start timing: 2 weeks after last testosterone cypionate/enanthate injection; 4-6 weeks after testosterone undecanoate; 1-2 days after discontinuing short-acting oral androgens
- Dose: 100-200 mcg twice daily subcutaneously
- Duration: 4-8 weeks
- Combination agents: Typically combined with a SERM (enclomiphene 12.5-25 mg daily or clomiphene 25-50 mg daily) and potentially short-course HCG (1000-2000 IU twice weekly for 2-3 weeks)
- Monitoring: LH, FSH, and total testosterone at baseline and every 2-4 weeks; semen analysis if fertility is a concern
- Taper: Reduce gonadorelin to once daily for the final 2 weeks before discontinuation
Injection Technique
Proper subcutaneous injection technique maximizes absorption and minimizes injection site reactions.
- Site selection: Rotate among abdomen (2 inches from navel), anterior thigh, and upper outer arm. Avoid areas with scars, bruises, or stretch marks.
- Preparation: Wash hands thoroughly. Clean injection site with alcohol swab and allow to dry completely (wet alcohol stings).
- Drawing the dose: Using a 29G or 30G insulin syringe, draw the calculated dose from the reconstituted vial. For 200 mcg from a 1 mg/mL solution, draw 0.2 mL (20 units on a U-100 insulin syringe).
- Injection: Pinch a fold of skin at the injection site. Insert the needle at a 45-degree angle (or 90 degrees if using a short 1/2-inch needle on adequate subcutaneous tissue). Inject slowly and steadily. Do not aspirate for subcutaneous injections.
- Post-injection: Release the skin fold. Apply gentle pressure with a clean cotton ball or gauze. Do not rub the injection site, as this can affect absorption.
Timing Considerations
The timing of gonadorelin injections can influence their effectiveness. For TRT support, many clinicians recommend evening or bedtime injection based on the physiological observation that GnRH pulse amplitude is naturally higher during sleep and that the nocturnal testosterone surge is driven by sleep-associated LH pulses. However, controlled data specifically comparing morning versus evening gonadorelin injection timing are not available.
For twice-daily protocols, spacing injections approximately 12 hours apart (for example, 7 AM and 7 PM) provides more consistent pituitary stimulation throughout the day. This mimics the endogenous GnRH pattern more closely than a single daily bolus, though it still falls far short of the approximately every-90-minute pulse frequency of normal GnRH secretion.
For fertility-focused protocols, some clinicians recommend injecting at least 30 minutes away from meals, based on the general principle that peptide absorption may be affected by postprandial changes in subcutaneous blood flow. While specific data for gonadorelin are limited, this is a reasonable precaution given the short half-life of the peptide and the importance of reliable absorption.
Dose Calculation Examples
Proper dose calculation prevents under-dosing and over-dosing. Here are common calculation scenarios for a 2 mg vial reconstituted with 2 mL bacteriostatic water (concentration: 1,000 mcg/mL).
| Desired Dose | Volume to Draw | Insulin Syringe Units (U-100) | Application |
|---|---|---|---|
| 50 mcg | 0.05 mL | 5 units | Low-dose starting protocol |
| 100 mcg | 0.10 mL | 10 units | Standard TRT support (QD or BID) |
| 150 mcg | 0.15 mL | 15 units | Intermediate dose |
| 200 mcg | 0.20 mL | 20 units | Standard fertility-focused protocol |
| 250 mcg | 0.25 mL | 25 units | High-dose protocol (under medical supervision) |
For a 2 mg vial reconstituted with 1 mL bacteriostatic water (concentration: 2,000 mcg/mL), all volumes are halved. A 200 mcg dose would require only 0.10 mL (10 insulin syringe units). This higher concentration reduces the injection volume but requires more precise measuring. For patients who prefer smaller injection volumes, the 1 mL reconstitution is practical. For patients new to self-injection who may benefit from easier measurement, the 2 mL reconstitution provides a more forgiving margin.
Vial Usage: How Many Doses Per Vial?
Understanding vial utilization helps patients plan their supply needs and budget.
| Protocol | Dose Per Injection | Injections Per Day | Daily Usage | Doses Per 2mg Vial | Days Per Vial |
|---|---|---|---|---|---|
| Standard TRT (QD) | 100 mcg | 1 | 100 mcg | 20 | 20 days |
| Standard TRT (QD) | 200 mcg | 1 | 200 mcg | 10 | 10 days |
| Enhanced TRT (BID) | 100 mcg | 2 | 200 mcg | 10 | 10 days |
| Fertility-Focused (BID) | 200 mcg | 2 | 400 mcg | 5 | 5 days |
Since reconstituted gonadorelin should be used within 14 to 28 days (depending on pharmacy guidance), patients on lower-dose protocols may have leftover solution at the end of the use-by period. In such cases, it may be more cost-effective to reconstitute with a smaller volume of bacteriostatic water, reducing waste. Always discuss vial management with your prescribing provider and dispensing pharmacy.
Troubleshooting Common Issues
Patients using gonadorelin may encounter several common issues that can be addressed with proper guidance.
Difficulty Drawing Small Volumes
Drawing very small volumes (5 to 10 units on an insulin syringe) can be challenging. Use good lighting, hold the syringe at eye level, and draw slightly past the desired mark before pushing back to the exact volume. Consider using 0.3 mL syringes with 0.5-unit markings for more precise small-volume measurement.
Foaming During Reconstitution
If the solution foams during reconstitution, do not shake the vial. Set it aside for 5 to 10 minutes and allow the foam to settle naturally. The foam is composed of denatured peptide at the air-liquid interface and represents a small loss of active product. Minimizing foam by injecting the bacteriostatic water slowly down the vial wall prevents this issue.
Pain at Injection Site
Injection site discomfort can be minimized by allowing the reconstituted solution to warm to room temperature for 5 to 10 minutes before injection (cold solutions cause more pain), using a fresh needle for each injection (needles dull after puncturing the vial stopper), injecting slowly and steadily, and rotating injection sites systematically. Some patients find that numbing the injection site with an ice cube for 30 seconds before injection helps reduce discomfort.
No Measurable LH Response
If blood work shows no measurable LH response to gonadorelin during TRT, several factors may be responsible. The TRT dose may be causing excessive pituitary suppression (consider reducing the testosterone dose). The gonadorelin dose may be insufficient (consider increasing to 200 mcg BID). The injection technique may be poor, with the peptide being injected intradermally rather than subcutaneously, leading to poor absorption. Or the pituitary gonadotrophs may be genuinely refractory due to prolonged suppression (consider a trial of HCG to bypass the pituitary). Discussing these scenarios with a knowledgeable clinician is essential for optimizing the protocol.
For more information on peptide preparation, the Peptide Research Hub provides detailed guidance on handling and storage.
Safety

Figure 8: The safety profile of gonadorelin is generally favorable, with most adverse effects being mild and transient.
Gonadorelin has a well-established safety profile based on decades of clinical use. As a synthetic version of an endogenous hormone, it interacts with normal physiological pathways and does not introduce foreign pharmacological mechanisms. This section reviews the safety data across different clinical applications.
General Safety Overview
Gonadorelin is classified as FDA Pregnancy Category B (no evidence of fetal harm in animal studies, but no adequate controlled studies in pregnant women). It has been approved by the FDA for diagnostic use (as Factrel, now discontinued) and was historically available for therapeutic use (as Lutrepulse for pulsatile pump therapy). Its safety record spans over four decades of clinical application.
The favorable safety profile of gonadorelin is attributable to several factors. Its structure is identical to the endogenous hormone, minimizing immunogenicity. Its half-life of 2 to 4 minutes means that any adverse effects from a single dose are short-lived. And because it works through the physiological GnRH receptor pathway, its effects are self-limited by the normal feedback mechanisms of the HPG axis.
Common Side Effects
The most frequently reported side effects of gonadorelin are mild and transient.
| Side Effect | Frequency | Severity | Management |
|---|---|---|---|
| Injection site reactions (redness, swelling, mild pain) | 10-20% | Mild | Rotate injection sites; apply ice briefly before injection |
| Headache | 5-15% | Mild-moderate | Usually resolves spontaneously; acetaminophen if needed |
| Nausea | 5-10% | Mild | Typically transient; inject at bedtime if troublesome |
| Flushing | 5-10% | Mild | Brief and self-limiting; no intervention needed |
| Abdominal discomfort | 3-8% | Mild | Usually transient; take with small snack if persistent |
| Dizziness/lightheadedness | 2-5% | Mild | Inject while seated or lying down; resolves within minutes |
| Skin rash (localized or generalized) | 1-3% | Mild-moderate | Antihistamine if needed; discontinue if severe |
Serious Adverse Events
Serious adverse events with gonadorelin are rare. The most significant concern specific to female fertility applications is ovarian hyperstimulation syndrome (OHSS), though this occurs much less frequently with pulsatile GnRH therapy than with gonadotropin injections because the physiological feedback mechanisms remain intact. OHSS is characterized by ovarian enlargement, abdominal pain and distension, nausea and vomiting, rapid weight gain from fluid shifts, and in severe cases, thromboembolism, renal failure, and respiratory compromise.
Anaphylaxis and severe allergic reactions have been reported in isolated cases. Patients with a history of allergic reactions to gonadorelin or any GnRH analog should not receive the medication. Signs of anaphylaxis include urticaria, angioedema, difficulty breathing, and hypotension, and require immediate emergency treatment.
Multi-dose vial use with bacteriostatic water introduces a small risk of injection site infection if proper aseptic technique is not maintained. Always clean the vial stopper with alcohol before each withdrawal and use a new sterile syringe and needle for each injection. Never reuse syringes or needles.
Drug Interactions
Gonadorelin has relatively few clinically significant drug interactions. The following are the most relevant.
Testosterone and Anabolic Steroids
Exogenous testosterone attenuates the pituitary response to gonadorelin through negative feedback. This is not a "drug interaction" in the traditional pharmacokinetic sense but rather a pharmacodynamic interaction at the level of the HPG axis. Men on TRT will have a blunted LH response to gonadorelin, which is expected and does not contraindicate concurrent use.
Estrogens and Progestins
Similarly, estrogen-containing medications (oral contraceptives, hormone replacement therapy) and progestins suppress gonadotropin secretion and will blunt the response to gonadorelin. This should be considered when interpreting GnRH stimulation test results in women taking these medications.
Dopamine Agonists
Medications like cabergoline and bromocriptine, used to treat hyperprolactinemia, may influence gonadotropin secretion and could theoretically interact with gonadorelin's effects. However, no clinically significant interactions have been documented.
GnRH Analogs
Concurrent use of GnRH agonists (leuprolide, goserelin) or GnRH antagonists (cetrorelix, ganirelix, degarelix) with gonadorelin is contraindicated, as these medications occupy or block the GnRH receptor and would negate gonadorelin's effects.
Contraindications
- Absolute contraindications: Known hypersensitivity to gonadorelin or any GnRH analog; active hormone-dependent malignancy (where gonadotropin stimulation could promote tumor growth); pregnancy (particularly after the first trimester, as gonadotropin stimulation could disrupt normal placental hormone balance).
- Relative contraindications: Pituitary tumors (particularly gonadotroph adenomas, where GnRH stimulation could theoretically promote tumor growth, though this has not been convincingly demonstrated); conditions requiring stable hormone levels where GnRH-induced fluctuations could be problematic.
Safety in Special Populations
Pediatric Use
Gonadorelin is well-established for diagnostic use in children being evaluated for precocious puberty or delayed puberty. The safety profile in the pediatric population mirrors that in adults, with injection site reactions and headache being the most common side effects. Dosing is weight-based (typically 2.5 mcg/kg for the stimulation test).
Elderly
Limited data exist on gonadorelin use in elderly patients specifically. The pituitary gonadotroph response to GnRH declines with age, and elderly patients may have comorbidities that affect HPG axis function. The safety profile is not expected to differ substantially from younger adults, but lower doses may be needed due to reduced pituitary reserve.
Renal and Hepatic Impairment
Gonadorelin is primarily degraded by endopeptidases in the blood and tissues, with minimal renal or hepatic metabolism. No dose adjustments are recommended for patients with renal or hepatic impairment. However, patients with severe liver disease may have altered sex hormone binding globulin (SHBG) levels that affect the clinical context in which gonadorelin is used.
OHSS: Detailed Risk Assessment for Pulsatile GnRH Therapy
Ovarian hyperstimulation syndrome deserves specific attention because it is the most serious potential complication of GnRH-based fertility therapy. OHSS is classified as mild (abdominal distension, mild pain, ovarian enlargement less than 8 cm), moderate (nausea, vomiting, ultrasound evidence of ascites, ovarian enlargement 8 to 12 cm), or severe (clinical ascites, oliguria, hematocrit above 45%, hyponatremia, pleural effusion, renal dysfunction, or thromboembolic events). Severe OHSS is life-threatening and requires hospitalization.
The risk of OHSS with pulsatile GnRH therapy is substantially lower than with gonadotropin injections. In the pulsatile GnRH paradigm, the pituitary responds to each GnRH pulse by releasing a physiological amount of FSH, which supports the growth of a dominant follicle. The intact negative feedback loop (rising estradiol from the dominant follicle suppresses FSH via inhibin B) prevents the development of multiple large follicles that characterizes OHSS. In contrast, exogenous gonadotropin injections bypass this feedback mechanism, making multifollicular development and OHSS more likely.
Published rates of OHSS with pulsatile GnRH therapy range from 1 to 3% of treatment cycles, compared to 3 to 8% with gonadotropin ovulation induction. The risk is further minimized by careful monitoring with serial estradiol measurements and transvaginal ultrasound, withholding the HCG trigger if more than 2 to 3 mature follicles develop, using lower GnRH pulse doses in women who show a brisk ovarian response, and canceling the cycle if estradiol rises above 1,500 to 2,000 pg/mL.
Endocrine Disruption Concerns
Some patients and clinicians express concern about the long-term effects of exogenous GnRH stimulation on pituitary function. Could chronic gonadorelin use cause pituitary gonadotroph hyperplasia or interfere with normal HPG axis regulation? The available evidence suggests these concerns are not warranted. Pulsatile GnRH therapy, even when used for extended periods (months to years in CHH patients), does not cause gonadotroph tumors or hyperplasia. The pituitary responds normally to endogenous feedback signals during gonadorelin therapy, indicating that the normal regulatory mechanisms remain intact.
A related question is whether long-term gonadorelin use could cause pituitary "dependence," making the gonadotrophs unable to function without exogenous GnRH stimulation. In patients with functional HPG axes (such as TRT users), discontinuation of gonadorelin has not been associated with any worsening of gonadotroph function beyond what would be expected from the ongoing TRT suppression. In CHH patients, where endogenous GnRH is already deficient, gonadotroph function predictably declines after gonadorelin discontinuation, but this reflects the underlying condition rather than a drug-induced effect.
Long-Term Safety Considerations
Long-term safety data for gonadorelin come primarily from the pulsatile pump therapy literature, where women have used the medication for multiple consecutive ovulation induction cycles. No significant long-term safety signals have been identified. There is no evidence of tachyphylaxis (progressive loss of response) with pulsatile administration at physiological pulse frequencies. The risk of anti-gonadorelin antibody development is very low due to the molecule's identity with the endogenous hormone, though isolated cases have been reported with prolonged use.
For men using gonadorelin as a TRT adjunct on a long-term basis, there are no published safety data beyond clinical experience and extrapolation from the established safety profile. However, given the physiological nature of the molecule and the well-characterized mechanism of action, significant long-term safety concerns are not expected. Ongoing monitoring of LH, FSH, and testicular function is recommended to ensure continued efficacy and detect any unexpected changes.
Gonadorelin and Cardiovascular Safety
Cardiovascular safety is a growing area of attention for all hormonal therapies. The relationship between GnRH signaling and cardiovascular function is complex. GnRH receptors have been identified on cardiac myocytes and vascular endothelial cells, though the functional significance of these extra-pituitary receptors is not fully understood. Long-term GnRH agonist therapy (which suppresses sex steroids) is associated with increased cardiovascular risk, including metabolic syndrome, increased arterial stiffness, and potentially increased cardiovascular events. These risks, however, are attributed to the sex steroid deprivation caused by GnRH agonist suppression rather than to direct effects of GnRH receptor activation.
For gonadorelin used as a TRT adjunct (where sex steroid levels are maintained by the concurrent testosterone therapy), cardiovascular concerns are minimal. The brief, pulsatile nature of the GnRH exposure and the maintenance of normal sex steroid levels distinguish this use case from chronic GnRH agonist therapy. No cardiovascular safety signals have been identified in the available clinical experience with gonadorelin for TRT support. Nonetheless, men on TRT (with or without gonadorelin) should be monitored for cardiovascular risk factors including hematocrit (erythrocytosis), blood pressure, lipid profile, and metabolic parameters, as part of standard TRT management. For related safety information on other peptide therapies, see our guides on BPC-157, TB-500, SS-31, and sermorelin.
Quality and Sourcing Considerations
Quality and Sourcing Considerations
The safety of any peptide therapy depends heavily on product quality. Gonadorelin should be obtained from licensed compounding pharmacies that follow current Good Manufacturing Practice (cGMP) standards and conduct third-party testing for identity, purity, potency, sterility, and endotoxin levels. Peptides obtained from unregulated sources carry risks of contamination, incorrect potency, degradation, and adulteration. FormBlends provides transparency about sourcing and testing standards for their peptide products.
Bacteriostatic Water and Preservative Considerations
Bacteriostatic water for injection (BWFI) contains 0.9% benzyl alcohol as a preservative, which prevents microbial growth in multi-dose vials. This preservative is what allows the reconstituted solution to be used over 14 to 28 days rather than requiring single-use preparation. However, benzyl alcohol can cause local irritation at the injection site in sensitive individuals, and in very rare cases, allergic reactions.
Sterile water for injection (SWFI), which does not contain preservatives, is an alternative for patients sensitive to benzyl alcohol. However, SWFI-reconstituted solutions should be used within 24 hours and must be discarded if not used in a single session, making it impractical for daily dosing protocols. Most patients tolerate BWFI well, and it is the standard recommendation for gonadorelin reconstitution.
Neonates should not receive medications preserved with benzyl alcohol due to the risk of "gasping syndrome," a rare but serious toxicity. This is clinically relevant only for pediatric applications of the GnRH stimulation test in neonatal patients. For pediatric diagnostic testing, SWFI should be used for reconstitution, and the solution should be prepared immediately before administration.
Comparison of Safety Profiles: Gonadorelin vs HCG vs Enclomiphene
When evaluating which adjunctive therapy to use alongside TRT, comparing safety profiles across the three main options provides useful context for clinical decision-making.
| Safety Parameter | Gonadorelin | HCG | Enclomiphene |
|---|---|---|---|
| Route of administration | Subcutaneous injection | Subcutaneous or intramuscular injection | Oral capsule |
| Common side effects | Injection site reaction, headache, nausea | Injection site pain, headache, gynecomastia, water retention | Headache, nausea, hot flashes, visual disturbances (rare) |
| Estradiol impact | Mild increase (via stimulated endogenous T production) | Moderate increase (direct Leydig cell stimulation increases T and E2) | Variable; blocks estrogen feedback but may not increase E2 levels |
| Receptor desensitization risk | Low (with intermittent dosing) | Moderate (chronic LH receptor stimulation at high doses) | Low (indirect mechanism) |
| Long-term safety data | Moderate (decades for pulsatile use; limited for TRT adjunct use) | Extensive (decades of use in fertility and TRT) | Limited (relatively new for this indication) |
| Immunogenicity | Very low (identical to endogenous hormone) | Low to moderate (foreign glycoprotein) | Not applicable (small molecule) |
| Pregnancy category | B | X (in women) | X (in women) |
| Drug interactions | Minimal | Minimal | CYP interactions possible; avoid with tamoxifen |
Gonadorelin stands out for its low immunogenicity (being identical to the endogenous hormone) and low risk of receptor desensitization when used at intermittent dosing frequencies. HCG has the most extensive long-term safety record but carries risks of estradiol elevation and LH receptor desensitization, particularly at higher doses. Enclomiphene offers the convenience of oral dosing but has less established long-term safety data and can cause visual disturbances in rare cases. All three options are generally well-tolerated in the TRT adjunct context, and the choice among them often comes down to availability, cost, patient preference (injection versus oral), and specific clinical goals.
Psychological and Emotional Considerations
An underappreciated aspect of gonadorelin therapy is its psychological impact. For many men on TRT, the addition of gonadorelin provides a sense of maintaining their natural hormonal function and preserving future fertility options. This psychological benefit is real and meaningful, even if the degree of physiological preservation is partial. Conversely, men who are anxious about injection technique or concerned about potential side effects may experience nocebo effects that amplify their perception of adverse reactions.
Clinicians should address psychological aspects of therapy openly. Setting realistic expectations about what gonadorelin can and cannot achieve during TRT prevents disappointment. Explaining that the treatment provides partial rather than complete testicular protection allows patients to make informed decisions about their protocol. Some men, after understanding the limitations, may choose to combine gonadorelin with other interventions (HCG, enclomiphene) for more comprehensive protection, while others may be satisfied with partial preservation and the lower complexity of gonadorelin monotherapy.
Monitoring for Adverse Effects: Practical Checklist
Patients using gonadorelin should be aware of what to watch for and when to contact their healthcare provider. The following checklist covers the key monitoring points.
- At each injection: Inspect the injection site for unusual redness, swelling, warmth, or drainage that could indicate infection. Mild redness and tenderness immediately after injection is normal and resolves within hours.
- Weekly: Note any persistent headaches, mood changes, or unusual symptoms that develop after starting gonadorelin. Keep a brief log to identify patterns.
- At 4-week follow-up: Blood work for LH, FSH, total testosterone, and estradiol. This initial assessment determines whether the gonadorelin dose is producing a measurable pituitary response.
- Every 3 to 6 months: Comprehensive hormone panel including LH, FSH, total and free testosterone, estradiol, SHBG, prolactin, and CBC. Testicular ultrasound if testicular size is a concern. Semen analysis if fertility is a goal.
- Contact provider immediately for: Signs of allergic reaction (hives, swelling, difficulty breathing), severe headache, testicular pain, significant mood disturbances, or injection site infection signs (increasing redness, pus, fever).
This structured monitoring approach ensures that any adverse effects are detected early and that the therapeutic protocol is optimized based on individual response. For related safety information on other peptide therapies, see our guides on BPC-157, TB-500, NAD+, and Epithalon.
Regulatory Status and Future Outlook
The regulatory landscape for gonadorelin continues to evolve. The original brand-name diagnostic product, Factrel (gonadorelin hydrochloride for injection, 100 mcg per vial), was manufactured by Ayerst Laboratories and later by Wyeth. It has been discontinued, leaving compounding pharmacies as the primary source of gonadorelin in the United States. The pulsatile pump product, Lutrepulse (gonadorelin acetate for injection), was manufactured by Ferring Pharmaceuticals and has also been discontinued in the US market, though pulsatile GnRH pump therapy remains available in some European countries.
The availability of gonadorelin through compounding pharmacies has been stable since the HCG reclassification, but the compounding pharmacy regulatory environment is subject to change. The FDA has increased scrutiny of compounding practices in recent years, and potential future regulatory actions could affect gonadorelin availability. Patients and clinicians should stay informed about regulatory developments and maintain relationships with reputable compounding pharmacies that adhere to cGMP standards.
Cost-Effectiveness Analysis
For patients making practical decisions about TRT adjunct therapy, understanding the relative costs of available options is important. Gonadorelin is generally one of the more affordable peptide therapies, particularly when compared to brand-name HCG products post-reclassification.
| Agent | Monthly Cost (Approximate) | Route | Frequency | Notes |
|---|---|---|---|---|
| Gonadorelin (compounded) | $30-80 | Subcutaneous | 1-2x daily | Widely available from compounding pharmacies |
| HCG (brand-name) | $100-300+ | Subcutaneous/IM | 2-3x weekly | More expensive since 2020 reclassification |
| Enclomiphene (compounded) | $30-100 | Oral | Daily | Convenience of oral dosing; variable availability |
| FSH (Gonal-F, Follistim) | $400-1000+ | Subcutaneous | 3x weekly | Expensive; primarily for active fertility treatment |
| Gonadorelin + enclomiphene combo | $60-150 | SC + oral | Daily each | Combination approach; moderate cost |
The cost-effectiveness calculation should consider not just the monthly medication cost but also the cost of syringes and needles (approximately $10 to 20 per month for daily injection), bacteriostatic water ($5 to 15 per vial), monitoring blood work (covered by insurance for most patients, or $50 to 150 per panel out of pocket), and provider visits for protocol management. When all costs are included, gonadorelin-based TRT support typically adds $50 to 150 per month to the base cost of TRT, which most patients consider acceptable given the potential benefits for testicular health and future fertility.
Insurance coverage for gonadorelin varies. When prescribed for a recognized medical indication (such as diagnostic testing or fertility treatment), some insurance plans cover the medication. When prescribed as an adjunct to TRT for testicular maintenance, coverage is less consistent. Patients should check with their insurance provider and consider that compounding pharmacy prices are often competitive even without insurance coverage.
Patient Education and Informed Consent
Proper patient education is critical for successful gonadorelin therapy. Patients should understand what gonadorelin is and how it works (synthetic GnRH that stimulates the pituitary), why it is being prescribed (to maintain testicular function during TRT), what it can realistically achieve (partial, not complete, testicular function preservation), what it cannot do (it is not a replacement for TRT, will not significantly boost testosterone levels during TRT, and may not fully preserve fertility), how to properly reconstitute, store, and inject the medication, what side effects to watch for and when to contact their provider, and the importance of regular monitoring with blood work.
Informed consent should address the off-label nature of gonadorelin use for TRT support (the medication is approved for diagnostic purposes and pulsatile pump therapy, not specifically as a TRT adjunct), the limited clinical trial data supporting this specific application, and the availability of alternative approaches (HCG, enclomiphene, or no adjunct therapy). Documenting this informed consent conversation protects both the patient and the clinician and ensures that treatment decisions are made collaboratively with appropriate expectations.
Looking forward, several developments could reshape the gonadorelin market. The development of longer-acting GnRH formulations that maintain pulsatile-like receptor activation could bridge the gap between once-daily injections and true pulsatile pump therapy. Oral GnRH receptor agonists are another theoretical possibility, though the peptide nature of GnRH makes oral bioavailability a major challenge. And advances in micro-pump technology could make wearable pulsatile gonadorelin delivery practical and affordable for TRT support applications.
Frequently Asked Questions
What is gonadorelin and what does it do?
Gonadorelin is a synthetic peptide that is structurally identical to the body's own gonadotropin-releasing hormone (GnRH). It is a decapeptide produced naturally in the hypothalamus that controls reproductive hormone production. When injected, gonadorelin stimulates the pituitary gland to release two critical hormones: luteinizing hormone (LH) and follicle-stimulating hormone (FSH). In men, LH drives testosterone production in the testes, while FSH supports sperm production. In women, these hormones regulate the menstrual cycle, follicular development, and ovulation. Clinically, gonadorelin is used as a diagnostic tool (GnRH stimulation test), a fertility treatment (pulsatile pump therapy), and an adjunct to testosterone replacement therapy to preserve testicular function. Its effects depend entirely on how it is administered: pulsatile delivery stimulates the reproductive axis, while continuous delivery paradoxically suppresses it.
How does gonadorelin affect testosterone levels?
Gonadorelin affects testosterone levels indirectly by stimulating the pituitary gland to release LH, which then acts on the Leydig cells in the testes to produce testosterone. When administered as a single bolus or in pulsatile fashion, gonadorelin causes a rapid rise in LH within 15 to 30 minutes, followed by a corresponding increase in testosterone over the subsequent 1 to 4 hours. In healthy men with intact HPG axis function, a 100 mcg dose of gonadorelin can increase LH by 3 to 5 times baseline. However, the testosterone-boosting effect is modest and transient because the peptide's half-life is only 2 to 4 minutes. In men on TRT, the ability of gonadorelin to stimulate additional testosterone production is limited because the pituitary is already suppressed by exogenous testosterone. In this context, the primary goal is maintaining some pituitary-testicular signaling rather than significantly increasing testosterone levels.
Can gonadorelin preserve fertility while on TRT?
Gonadorelin can provide partial fertility preservation during TRT, but the evidence is less strong than for HCG. Clinical experience from men's health clinics suggests that gonadorelin maintains approximately 50 to 60% of testicular function when used alongside TRT, compared to roughly 80 to 90% preservation with HCG co-therapy. The challenge is that exogenous testosterone suppresses the pituitary gland, reducing its ability to respond to gonadorelin stimulation. For men actively trying to conceive while on TRT, HCG remains the preferred adjunct due to its direct action on the LH receptor, bypassing the suppressed pituitary. Gonadorelin is more appropriate for men who want to minimize testicular atrophy and maintain the potential for future fertility recovery. If fertility is an immediate priority, the most reliable approach is to temporarily discontinue or reduce TRT and use a combination of HCG, FSH (or HMG), and potentially a SERM to maximize spermatogenesis recovery.
What is the difference between pulsatile and continuous GnRH?
Pulsatile and continuous GnRH administration produce opposite biological effects, despite using the same molecule. Pulsatile delivery, which mimics the body's natural every-60-to-120-minute rhythm, stimulates the pituitary to produce LH and FSH, supporting normal reproductive function. This pattern allows GnRH receptors and downstream signaling pathways to reset between pulses, maintaining responsiveness. Continuous delivery, in contrast, causes an initial burst of gonadotropin release (the "flare" effect) followed by progressive receptor desensitization and downregulation over 1 to 3 weeks, ultimately suppressing LH and FSH to near-undetectable levels. This suppressive effect is the basis for GnRH agonist drugs like leuprolide (Lupron) used in prostate cancer and endometriosis. The molecular basis involves differences in Gq/11 and Gs/cAMP signaling dynamics, PKC activation patterns, and receptor internalization rates under pulsatile versus sustained stimulation conditions.
How do you take gonadorelin injections?
Gonadorelin is administered as a subcutaneous injection using an insulin syringe. The lyophilized powder is first reconstituted with bacteriostatic water (typically 2 mL into a 2 mg vial for a 1 mg/mL concentration). To inject, clean the injection site (abdomen, thigh, or upper arm) with an alcohol swab and let it dry. Pinch a fold of skin at the site, insert the needle at a 45-degree angle, and inject the dose slowly and steadily. Do not aspirate for subcutaneous injections. Release the skin fold, apply gentle pressure with a cotton ball, and do not rub. Rotate injection sites with each dose to prevent lipohypertrophy or localized irritation. For TRT support, the typical dose is 100 to 200 mcg once or twice daily. Store the reconstituted solution refrigerated at 36 to 46 degrees Fahrenheit and use within 14 to 28 days. Always use a new sterile syringe and needle for each injection.
What are the side effects of gonadorelin?
Gonadorelin has a favorable safety profile with mostly mild and transient side effects. The most common side effects include injection site reactions (redness, swelling, mild pain) in 10 to 20% of users, headache in 5 to 15%, nausea in 5 to 10%, flushing in 5 to 10%, and mild abdominal discomfort in 3 to 8%. Dizziness and localized skin rash occur less frequently. Serious adverse events are rare but include allergic reactions (urticaria, angioedema, and in very rare cases, anaphylaxis) and, in women undergoing fertility treatment, ovarian hyperstimulation syndrome (OHSS), though OHSS occurs much less frequently with pulsatile GnRH than with gonadotropin injections. Depression and mood changes have been reported in isolated cases. Because gonadorelin is structurally identical to the body's own GnRH and has a very short half-life (2 to 4 minutes), most side effects resolve quickly. Always report persistent or unusual symptoms to your healthcare provider.
How does gonadorelin compare to HCG?
Gonadorelin and HCG both aim to support testicular function but work through fundamentally different mechanisms. Gonadorelin acts upstream, stimulating the pituitary to release LH and FSH, while HCG acts downstream, directly activating the LH receptor on Leydig cells. This mechanistic difference has several practical implications. HCG bypasses the pituitary entirely, so it works even when the pituitary is suppressed by exogenous testosterone, making it more effective for intratesticular testosterone maintenance during TRT (preserving 90 to 100% of ITT at appropriate doses versus approximately 50 to 60% with gonadorelin). Gonadorelin's advantages include stimulating both LH and FSH (HCG only provides LH-like activity), lower risk of LH receptor desensitization, wider availability since HCG was reclassified as a biologic in 2020, and generally lower cost. For active fertility preservation during TRT, HCG has stronger evidence. For general testicular maintenance and preserving future fertility potential, gonadorelin is a reasonable and accessible alternative.
What is pulsatile GnRH therapy used for?
Pulsatile GnRH therapy is primarily used to induce ovulation in women with hypothalamic amenorrhea, a condition where the hypothalamus fails to produce adequate GnRH pulses, causing absent menstrual periods and infertility. The therapy delivers gonadorelin through a programmable microinfusion pump at intervals of 60 to 120 minutes, mimicking the body's natural GnRH rhythm. The pituitary responds with physiological LH and FSH secretion, driving normal follicular development and typically producing monofollicular ovulation. In experienced centers, cumulative live birth rates exceed 80% over multiple treatment cycles, with a multiple pregnancy rate of only 3 to 5%. This is substantially lower than the multiple pregnancy rate with gonadotropin therapy (15 to 20%). Pulsatile GnRH is also used to induce spermatogenesis in men with congenital hypogonadotropic hypogonadism, achieving comparable outcomes to HCG plus HMG therapy in clinical studies.
What is the GnRH stimulation test?
The GnRH stimulation test is a diagnostic procedure that assesses pituitary gonadotroph function by measuring LH and FSH response to a bolus of synthetic GnRH (gonadorelin). A baseline blood sample is drawn, then 100 mcg of gonadorelin is administered intravenously, followed by additional blood draws at 15, 30, 45, 60, and 120 minutes. A normal response shows LH rising to 3 to 5 times baseline, peaking at 15 to 30 minutes. A blunted response suggests pituitary dysfunction, while an exaggerated response may indicate chronically understimulated gonadotrophs. The test is used to diagnose central precocious puberty (where a stimulated LH above 5 mIU/mL is diagnostic), distinguish hypothalamic from pituitary causes of hypogonadism, evaluate delayed puberty, and assess pituitary recovery after surgery or radiation. When a single test is inconclusive, a prolonged priming protocol with repeated GnRH pulses over 5 to 7 days can improve diagnostic accuracy.
How should gonadorelin be stored?
Gonadorelin should be stored according to its form. Lyophilized (freeze-dried) powder should be kept refrigerated at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius) or frozen at minus 4 degrees Fahrenheit (minus 20 degrees Celsius) for long-term storage. Once reconstituted with bacteriostatic water, the solution must be refrigerated at 36 to 46 degrees Fahrenheit and used within 14 to 28 days (follow the specific guidance from your pharmacy). Never freeze reconstituted solution, as this can cause protein aggregation and loss of potency. Protect both forms from direct light and heat. If the reconstituted solution appears cloudy, discolored, or contains visible particles, discard it and prepare a fresh vial. When traveling, transport the vial in an insulated pouch with a cold pack, keeping it below 46 degrees Fahrenheit but above freezing. Do not leave reconstituted gonadorelin at room temperature for more than 4 to 6 hours.
Can women use gonadorelin?
Yes, gonadorelin has well-established applications in women's health. The primary therapeutic use is pulsatile GnRH pump therapy for ovulation induction in women with hypothalamic amenorrhea, where it has a decades-long track record of safety and efficacy. The GnRH stimulation test is also used diagnostically in women to evaluate pituitary function and help diagnose conditions affecting the HPG axis. In assisted reproduction, GnRH has been used in various protocols for controlled ovarian stimulation. Women with polycystic ovary syndrome (PCOS) may undergo GnRH stimulation testing as part of their diagnostic evaluation. Gonadorelin is classified as FDA Pregnancy Category B, indicating no evidence of fetal harm in animal studies. However, its use during pregnancy is generally limited to early fertility treatment contexts, and it should be discontinued once pregnancy is confirmed unless specifically directed by a reproductive endocrinologist.
What dose of gonadorelin should I use for TRT support?
The standard dose of gonadorelin for TRT support is 100 to 200 mcg per subcutaneous injection, administered once or twice daily. Most clinicians start patients at 100 mcg daily (typically before bed) and assess the LH response with blood work after 4 weeks. If serum LH remains undetectable or very low, the dose can be increased to 200 mcg daily or 100 mcg twice daily. For men with active fertility concerns, the higher-intensity protocol of 200 mcg twice daily (morning and evening) provides more consistent pituitary stimulation. The reconstituted solution from a 2 mg vial in 2 mL of bacteriostatic water yields 1 mg/mL, so a 200 mcg dose equals 0.2 mL (20 units on a U-100 insulin syringe). Individual response varies significantly, and some men on higher-dose TRT regimens may need the upper end of the dosing range. Always work with your healthcare provider to determine the appropriate dose based on your lab results, TRT dose, and clinical goals. The FormBlends dosing calculator can assist with protocol planning.
Is gonadorelin legal and available?
Gonadorelin is a prescription medication in the United States and most other countries. It is legally available through licensed compounding pharmacies with a valid prescription from a licensed healthcare provider. Unlike HCG, which was reclassified as a biologic in 2020 and removed from the compounding pharmacy market, gonadorelin remains available through compounding. This reclassification is one of the primary reasons gonadorelin has become more widely used as a TRT adjunct in recent years. The original brand-name diagnostic product (Factrel) has been discontinued, but compounding pharmacies can legally produce gonadorelin for individual patient use. Gonadorelin is not a controlled substance and is not listed as a banned substance by most athletic organizations, though athletes should always verify with their sport's governing body before use. In research contexts, gonadorelin is also available from peptide research suppliers, though research-grade products are not intended for human administration.
Gonadorelin and Concurrent Peptide Therapy Considerations
Many patients exploring gonadorelin are also interested in or currently using other peptide therapies for different health goals. Understanding potential interactions and complementary uses is valuable for both clinicians and patients managing multi-peptide protocols.
Growth Hormone Secretagogues
Peptides like CJC-1295/ipamorelin, sermorelin, MK-677, hexarelin, and tesamorelin stimulate growth hormone release through the GHRH receptor or ghrelin receptor, pathways entirely distinct from the GnRH receptor. No direct pharmacological interaction exists between gonadorelin and these GH secretagogues. They act on different pituitary cell populations (gonadotrophs versus somatotrophs), use different receptors, and produce different endocrine outputs. Many TRT patients use a GH secretagogue alongside gonadorelin without apparent adverse interactions.
That said, GH and IGF-1 levels can influence reproductive function indirectly. Growth hormone plays a permissive role in gonadal function, and GH-deficient individuals may have impaired reproductive capacity that improves with GH replacement. The addition of a GH secretagogue to a TRT plus gonadorelin protocol could theoretically provide complementary benefits for overall hormonal optimization, body composition, and recovery, though formal studies of this combination are lacking.
Healing and Recovery Peptides
BPC-157, TB-500, and their combined blend act through growth factor modulation, angiogenesis, and anti-inflammatory pathways that are completely independent of the HPG axis. No interactions with gonadorelin are expected or have been reported. Patients using these healing peptides alongside gonadorelin and TRT are essentially targeting different physiological systems simultaneously.
Cognitive and Neuroprotective Peptides
Peptides like Semax, Selank, Dihexa, and Pinealon target neural pathways and neurotrophic factor signaling. While the hypothalamus is a brain structure, the GnRH neurons are a very specific population, and cognitive peptides are not expected to influence GnRH signaling or vice versa. One area of theoretical interest is the relationship between stress, cortisol, and GnRH pulsatility. Stress neuropeptides and anxiolytic peptides like Selank could theoretically support GnRH pulsatility by reducing stress-mediated suppression of the reproductive axis, though this hypothesis has not been tested.
Metabolic Peptides
GLP-1 receptor agonists (semaglutide, tirzepatide, liraglutide, retatrutide) are increasingly used for weight management and metabolic optimization. The relationship between GLP-1 agonists and the reproductive axis is an emerging area of interest. Weight loss from GLP-1 therapy can improve testosterone levels in obese men by reducing aromatase activity in adipose tissue, potentially complementing the effects of TRT and gonadorelin. However, rapid weight loss and caloric restriction can also suppress GnRH pulsatility (as seen in hypothalamic amenorrhea), creating a complex interaction that requires careful monitoring. Men using GLP-1 agonists for weight loss alongside TRT and gonadorelin should have their hormone levels monitored more frequently during the active weight loss phase. Compounds like 5-Amino-1MQ, AOD-9604, and MOTS-c also target metabolic pathways that could interact with reproductive physiology through energy-sensing mechanisms.
Anti-Aging and Longevity Peptides
Epithalon, FOXO4-DRI, Humanin, and NAD+ target aging-related pathways including telomerase activation, senescent cell clearance, mitochondrial function, and cellular energy metabolism. These pathways are largely independent of GnRH signaling, and no direct interactions are expected. However, the decline in GnRH pulsatility that occurs with aging is itself a contributor to age-related hypogonadism, suggesting that maintaining GnRH signaling through gonadorelin therapy could be considered part of a broader anti-aging strategy in appropriate patients.
Patient Selection: Who Benefits Most from Gonadorelin?
Not every man on TRT needs gonadorelin, and not every patient who could benefit from it is a good candidate. Identifying the right patients for gonadorelin therapy optimizes outcomes and avoids unnecessary expense and injection burden.
Ideal Candidates for Gonadorelin During TRT
- Men under 45 on TRT who wish to preserve future fertility potential
- Men concerned about testicular atrophy for cosmetic or psychological reasons
- Men who cannot access or afford HCG since the 2020 reclassification
- Men who prefer a more physiological approach to testicular support (upstream GnRH stimulation versus direct LH receptor agonism)
- Men on moderate-dose TRT (testosterone levels 700 to 1000 ng/dL), where residual pituitary sensitivity to GnRH is more likely
Less Ideal Candidates
- Men on high-dose TRT or multiple androgens (profound pituitary suppression limits response)
- Men over 55 with reduced pituitary reserve (diminished capacity to respond to GnRH)
- Men actively trying to conceive (HCG or treatment discontinuation is more reliable)
- Men with known pituitary pathology (tumors, prior surgery, radiation) that would prevent a gonadotropin response
- Men who are unable or unwilling to adhere to daily subcutaneous injections
Candidates for Pulsatile GnRH Pump Therapy
- Women with confirmed hypothalamic amenorrhea desiring pregnancy
- Men with congenital hypogonadotropic hypogonadism desiring fertility
- Patients with hypothalamic GnRH deficiency and documented intact pituitary function
- Patients who have failed or cannot tolerate gonadotropin injection therapy
Real-World Outcomes: What Patients Report
While controlled clinical trial data for gonadorelin as a TRT adjunct remain limited, real-world patient reports from men's health clinics and online communities provide some insight into practical outcomes. These reports should be interpreted with caution, as they are subject to selection bias, placebo effect, and other confounders. With that caveat, commonly reported experiences include maintained testicular size or reduced atrophy (reported by approximately 60 to 70% of users), subjective improvement in libido or sexual function beyond what TRT alone provided (reported by approximately 30 to 40%), improved sense of well-being (difficult to separate from placebo), and minimal side effects (most users report no significant adverse effects beyond occasional injection site irritation).
Less commonly, some patients report no noticeable benefit from gonadorelin, and a subset report frustration when lab work shows persistently suppressed LH despite consistent use. These non-responders may require dose escalation, protocol modification (twice-daily rather than once-daily), or transition to HCG. Honest discussion of these variable outcomes is important for informed consent and realistic expectation-setting.
The clinical experience with gonadorelin is still accumulating, and the field would benefit greatly from well-designed comparative studies evaluating gonadorelin versus HCG versus placebo as TRT adjuncts, with endpoints including testicular volume, intratesticular testosterone, semen parameters, patient-reported outcomes, and long-term safety. Until such studies are completed, clinical practice will continue to be guided by physiological rationale, extrapolation from related contexts, and accumulated clinical experience. The Drug Comparison Hub provides additional context for comparing different therapeutic options across peptide categories.
Future Directions in GnRH Therapeutics
The field of GnRH-based therapeutics continues to evolve, with several promising research directions that could expand gonadorelin's clinical applications or provide new tools for managing reproductive endocrine disorders.
Kisspeptin as a Therapeutic Agent
Kisspeptin-10, the endogenous activator of GnRH neurons, is being investigated as a potential therapeutic agent for both diagnostic and treatment purposes. Kisspeptin acts upstream of GnRH, stimulating GnRH neurons to release endogenous GnRH. This creates a more physiological stimulation pattern than exogenous GnRH injection, as the kisspeptin-stimulated GnRH release occurs from the actual GnRH nerve terminals in the median eminence with normal spatial and temporal characteristics. Clinical studies have shown that kisspeptin-54 can trigger LH pulses and the LH surge in humans, and it is being evaluated for ovulation triggering in IVF cycles as an alternative to HCG. The advantage of kisspeptin triggering is a much lower risk of OHSS, because the LH surge it produces is shorter in duration and more physiological than the prolonged LH-like stimulation from HCG.
For men's health applications, kisspeptin could theoretically provide a more upstream and physiological stimulus than gonadorelin for maintaining HPG axis function during TRT. However, kisspeptin's even shorter half-life (approximately 1 minute for kisspeptin-10) and the need for very frequent dosing limit its practical utility as a daily-use therapeutic. Future development of longer-acting kisspeptin analogs could change this calculus.
Neurokinin B Receptor Antagonists
Based on the understanding of KNDy neuron physiology, neurokinin B (NKB) receptor (NK3R) antagonists are being developed for reproductive applications. NK3R antagonists reduce GnRH pulse frequency by dampening KNDy neuron synchronization. This approach is being studied for PCOS (where abnormally high GnRH pulse frequency drives LH excess), uterine fibroids, and endometriosis. While these antagonists suppress rather than stimulate GnRH signaling, they represent the therapeutic exploitation of the same KNDy neuron pathway that gonadorelin targets from the downstream side.
Long-Acting GnRH Formulations
Current research is exploring depot formulations of native GnRH or GnRH analogs designed to produce pulsatile-like receptor activation from a single injection. These formulations use controlled-release microsphere technology or biodegradable polymer matrices to deliver GnRH in a pattern that mimics endogenous pulsatility. If successful, such formulations could replace the need for daily gonadorelin injections with weekly or monthly injections, dramatically improving patient compliance and convenience.
Gene Therapy for GnRH Deficiency
For patients with congenital hypogonadotropic hypogonadism, gene therapy approaches are being explored to restore endogenous GnRH production. While still in preclinical stages, the concept of delivering a functional GNRH1 gene or correcting mutations in genes like GNRHR, KISS1R, or ANOS1 offers the possibility of permanent correction of GnRH deficiency. Animal studies have demonstrated proof of concept using adeno-associated virus (AAV) vectors to deliver kisspeptin or GnRH genes to the hypothalamus, resulting in restored reproductive function. Translation to human therapy remains many years away but represents an exciting long-term possibility.
Precision Medicine Approaches
Pharmacogenomic research is beginning to identify genetic variants that influence individual response to GnRH therapy. Polymorphisms in the GNRHR gene, the KISS1R gene, and genes encoding downstream signaling molecules may explain why some patients respond well to gonadorelin while others show minimal response. As genomic profiling becomes more affordable and accessible, personalized dosing and therapy selection based on genetic markers could improve outcomes and reduce the trial-and-error aspect of current protocol development.
These research directions suggest that while gonadorelin has a well-established role in current clinical practice, the future of GnRH-based therapeutics is likely to include more sophisticated approaches that build on the same foundational physiology. For patients and clinicians today, understanding gonadorelin's mechanism and applications provides a strong foundation for evaluating and adopting these emerging therapies as they become available. The Retatrutide Hub covers another area of active therapeutic development in the peptide space.
Summary of Clinical Recommendations
Based on the current evidence and clinical experience reviewed in this report, the following practical recommendations can be offered for clinicians and patients considering gonadorelin therapy.
For men on TRT who want to maintain testicular health, gonadorelin at 100 to 200 mcg subcutaneously once or twice daily provides partial preservation of pituitary-gonadal signaling. This is most appropriate for men who cannot access or afford HCG, prefer a more physiological approach, or are primarily concerned with preventing testicular atrophy rather than actively trying to conceive. Start at 100 mcg daily, check LH at 4 weeks, and escalate to 200 mcg BID if LH remains undetectable. Monitor hormone levels and testicular volume every 3 to 6 months.
For men actively trying to conceive while on TRT, HCG remains the preferred adjunct based on stronger evidence for intratesticular testosterone maintenance and spermatogenesis preservation. If HCG is not available, a combination of gonadorelin 200 mcg BID plus enclomiphene 25 mg daily provides multi-level HPG axis support. Consider temporary TRT discontinuation if semen parameters do not improve within 3 to 6 months on adjunct therapy.
For women with hypothalamic amenorrhea desiring pregnancy, pulsatile GnRH pump therapy (5 to 20 mcg per pulse every 90 minutes) remains an excellent option with high ovulation and live birth rates and low multiple pregnancy risk. This therapy should be managed by a reproductive endocrinologist with experience in pump-based protocols. Address underlying energy deficit or stress factors alongside pharmacological intervention.
For diagnostic testing, the GnRH stimulation test (100 mcg IV with serial LH/FSH sampling at 0, 15, 30, 45, 60, and 120 minutes) provides valuable information about pituitary gonadotroph function. Interpret results in the context of age, sex, pubertal stage, menstrual cycle phase, and concurrent medications. Use the prolonged priming protocol when initial results are ambiguous in patients with suspected hypothalamic pathology.
For post-cycle therapy, gonadorelin 100 to 200 mcg BID combined with a SERM provides a physiologically rational approach to HPG axis recovery after exogenous androgen discontinuation. Start PCT after the exogenous androgen has substantially cleared, and monitor recovery with serial hormone panels over 8 to 12 weeks.
Across all applications, proper reconstitution, storage, and injection technique are essential for consistent results. Obtain gonadorelin only from licensed compounding pharmacies that follow cGMP standards and provide certificates of analysis. Work with knowledgeable healthcare providers who understand the physiology of GnRH signaling and can individualize protocols based on laboratory results and clinical response. The free assessment at FormBlends can help identify the most appropriate starting point for your individual situation.
References
- Schally AV, Arimura A, Baba Y, et al. Isolation and properties of the FSH and LH-releasing hormone. Biochemical and Biophysical Research Communications. 1971;43(2):393-399. DOI: 10.1016/0006-291X(71)90766-2.
- Herbison AE. The gonadotropin-releasing hormone pulse generator. Endocrinology. 2018;159(11):3723-3736. DOI: 10.1210/en.2018-00653.
- Lehman MN, Coolen LM, Goodman RL. Minireview: kisspeptin/neurokinin B/dynorphin (KNDy) cells of the arcuate nucleus: a central node in the control of GnRH secretion. Endocrinology. 2010;151(8):3479-3489. DOI: 10.1210/en.2010-0022.
- Moore AM, Coolen LM, Lehman MN. KNDy neurons of the hypothalamus and their role in GnRH pulse generation: an update. Endocrinology. 2024;165(2):bqad194. DOI: 10.1210/endocr/bqad194.
- Armstrong SP, Caunt CJ, McArdle CA. Pulsatile and sustained GnRH receptor signaling: does the ERK signaling pathway decode GnRH pulse frequency? Journal of Biological Chemistry. 2010;285(32):24360-24371. DOI: 10.1074/jbc.M110.155275.
- Thompson IR, Kaiser UB. GnRH pulse frequency-dependent differential regulation of LH and FSH gene expression. Molecular and Cellular Endocrinology. 2014;385(1-2):28-35. DOI: 10.1016/j.mce.2013.09.012.
- Millar RP, Lu ZL, Pawson AJ, et al. Gonadotropin-releasing hormone receptors. Endocrine Reviews. 2004;25(2):235-275. DOI: 10.1210/er.2003-0002.
- Coviello AD, Matsumoto AM, Bremner WJ, et al. Low-dose human chorionic gonadotropin maintains intratesticular testosterone in normal men with testosterone-induced gonadotropin suppression. Journal of Clinical Endocrinology & Metabolism. 2005;90(5):2595-2602. DOI: 10.1210/jc.2004-0802.
- Page ST, Kalhorn TF, Bremner WJ, et al. Intratesticular androgens and spermatogenesis during severe gonadotropin suppression induced by male hormonal contraceptive treatment. Journal of Andrology. 2007;28(5):734-741. DOI: 10.2164/jandrol.107.002790.
- Dwyer AA, Raivio T, Pitteloud N. Management of endocrine disease: reversible hypogonadotropic hypogonadism. European Journal of Endocrinology. 2016;174(6):R267-R274. DOI: 10.1530/EJE-15-1033.
- Martin KA, Hall JE, Adams JM, et al. Comparison of exogenous gonadotropins and pulsatile gonadotropin-releasing hormone for induction of ovulation in hypogonadotropic amenorrhea. Journal of Clinical Endocrinology & Metabolism. 1993;77(1):125-129. DOI: 10.1210/jcem.77.1.8325935.
- Christin-Maitre S, de Crecy M. Use of pulsatile gonadotropin-releasing hormone in patients with functional hypothalamic amenorrhea results in monofollicular ovulation and high cumulative live birth rates: a 25-year cohort. Journal of Assisted Reproduction and Genetics. 2023;40(1):117-125. DOI: 10.1007/s10815-022-02656-0.
- Sidhoum VF, Chan YM, Lippincott MF, et al. Reversal and relapse of hypogonadotropic hypogonadism: resilience and fragility of the reproductive neuroendocrine system. Journal of Clinical Endocrinology & Metabolism. 2014;99(3):861-870. DOI: 10.1210/jc.2013-2809.
- Kim HK, Kee SJ, Seo JY, et al. Gonadotropin-releasing hormone stimulation test and diagnostic cutoff in precocious puberty: a mini review. Annals of Pediatric Endocrinology & Metabolism. 2020;25(3):142-149. DOI: 10.6065/apem.2040072.036.
- Bhasin S, Brito JP, Cunningham GR, et al. Testosterone therapy in men with hypogonadism: an Endocrine Society clinical practice guideline. Journal of Clinical Endocrinology & Metabolism. 2018;103(5):1715-1744. DOI: 10.1210/jc.2018-00229.
- Harrington J, Palmert MR. Clinical review: distinguishing constitutional delay of growth and puberty from isolated hypogonadotropic hypogonadism: critical appraisal of available diagnostic tests. Journal of Clinical Endocrinology & Metabolism. 2012;97(9):3056-3067. DOI: 10.1210/jc.2012-1598.
- Martin K, Santamaria JD, Toraiwa K, et al. Efficacy and safety of pulsatile gonadotropin-releasing hormone therapy among patients with idiopathic and functional hypothalamic amenorrhea: systematic review and meta-analysis. Fertility and Sterility. 2018;109(5):851-860. DOI: 10.1016/j.fertnstert.2018.01.003.
- Liu Y, Gong Y, Wang Y, et al. Pulsatile gonadotropin-releasing hormone therapy: comparison of efficacy between functional hypothalamic amenorrhea and congenital hypogonadotropic hypogonadism. Fertility and Sterility. 2024;122(6):1034-1042. DOI: 10.1016/j.fertnstert.2024.07.009.
- Saffati A, Anis T. Safety and efficacy of enclomiphene and clomiphene for hypogonadal men. Translational Andrology and Urology. 2024;13(6):1198-1210. DOI: 10.21037/tau-23-647.
- Enclomiphene Citrate Study Group. Enclomiphene citrate for the treatment of secondary male hypogonadism. Expert Opinion on Pharmacotherapy. 2016;17(11):1561-1567. DOI: 10.1080/14656566.2016.1204295.
- Belchetz PE, Plant TM, Nakai Y, et al. Hypophysial responses to continuous and intermittent delivery of gonadotropin-releasing hormone. Science. 1978;202(4368):631-633. DOI: 10.1126/science.100883.
- Filicori M, Santoro N, Merriam GR, et al. Characterization of the physiological pattern of episodic gonadotropin secretion throughout the human menstrual cycle. Journal of Clinical Endocrinology & Metabolism. 1986;62(6):1136-1144. DOI: 10.1210/jcem-62-6-1136.
- Seminara SB, Messager S, Chatzidaki EE, et al. The GPR54 gene as a regulator of puberty. New England Journal of Medicine. 2003;349(17):1614-1627. DOI: 10.1056/NEJMoa035322.
- Topaloglu AK, Reimann F, Guclu M, et al. TAC3 and TACR3 mutations in familial hypogonadotropic hypogonadism reveal a key role for neurokinin B in the central control of reproduction. Nature Genetics. 2009;41(3):354-358. DOI: 10.1038/ng.306.
- McArdle CA, Roberson MS. Gonadotropes and gonadotropin-releasing hormone signaling. In: Plant TM, Zeleznik AJ, eds. Knobil and Neill's Physiology of Reproduction. 4th ed. Academic Press; 2015:335-397.
- Nachtigall LB, Boepple PA, Pralong FP, et al. Adult-onset idiopathic hypogonadotropic hypogonadism: a treatable form of male infertility. New England Journal of Medicine. 1997;336(6):410-415. DOI: 10.1056/NEJM199702063360604.
- Boehm U, Bouloux PM, Dattani MT, et al. European Consensus Statement on congenital hypogonadotropic hypogonadism: pathogenesis, diagnosis and treatment. Nature Reviews Endocrinology. 2015;11(9):547-564. DOI: 10.1038/nrendo.2015.112.
- Conn PM, Crowley WF Jr. Gonadotropin-releasing hormone and its analogues. New England Journal of Medicine. 1991;324(2):93-103. DOI: 10.1056/NEJM199101103240205.
- Wierman ME, Pawlak J, Allen T, et al. Molecular mechanisms of gonadotropin-releasing hormone neuronal migration. Trends in Endocrinology & Metabolism. 2004;15(2):96-102. DOI: 10.1016/j.tem.2004.01.003.
- Kaprara A, Huhtaniemi IT. The hypothalamus-pituitary-gonad axis: tales of mice and men. Metabolism. 2018;86:3-17. DOI: 10.1016/j.metabol.2017.11.018.
- Young J, Xu C, Papadakis GE, et al. Clinical management of congenital hypogonadotropic hypogonadism. Endocrine Reviews. 2019;40(2):669-710. DOI: 10.1210/er.2018-00116.
- Han TS, Bouloux PM. What is the optimal therapy for young males with hypogonadotropic hypogonadism? Clinical Endocrinology. 2010;72(6):731-737. DOI: 10.1111/j.1365-2265.2009.03746.x.
- Drugs.com. Gonadorelin injectable: uses, side effects & warnings. Accessed March 2026. Available at: https://www.drugs.com/mtm/gonadorelin-injectable.html.
- Mayo Clinic. Gonadorelin (intravenous route, injection route): side effects and dosage. Accessed March 2026. Available at: https://www.mayoclinic.org/drugs-supplements/gonadorelin-intravenous-route-injection-route/description/drg-20067426.
- DrugBank. Gonadorelin: uses, interactions, mechanism of action. Accessed March 2026. Available at: https://go.drugbank.com/drugs/DB00644.