Executive Summary
Peptide therapies don't exist in a vacuum. Most people pursuing peptide protocols are already taking prescription medications, supplements, or both - and understanding how these compounds interact is essential for safety and effectiveness.
Key Takeaways
- GLP-1 receptor agonists affect oral medication absorption through delayed gastric emptying, but most interactions are clinically manageable
- Growth hormone peptides can alter glucose metabolism, making diabetes medication adjustments necessary in some patients
- Healing peptides like BPC-157 lack formal drug interaction studies, requiring extra caution
- Immune-modulating peptides such as Thymosin Alpha-1 are potentially contraindicated with immunosuppressive medications
- Timing strategies can mitigate many absorption-based interactions
The world of peptide-drug interactions is surprisingly nuanced. Some combinations work beautifully together, amplifying therapeutic benefits without added risk. Others create subtle but meaningful changes in how your body absorbs, metabolizes, or responds to medications you've been taking for years. And a small number of combinations carry genuine danger - the kind that demands immediate medical attention and careful protocol adjustment.
This report covers every major peptide class used in clinical and research settings: GLP-1 receptor agonists like semaglutide and tirzepatide, growth hormone secretagogues such as CJC-1295/Ipamorelin and MK-677, healing peptides like BPC-157, immune modulators including Thymosin Alpha-1, and cellular health compounds such as NAD+. For each, we'll examine documented interactions, theoretical concerns, and practical guidance for safe use alongside common medications.
Critical Safety Notice
This guide is for educational purposes only. Never start, stop, or modify any medication - including peptide therapies - without consulting your prescribing physician. Drug interactions can be unpredictable and individual responses vary based on genetics, organ function, dosing, and timing. Your healthcare provider has the full picture of your medical history and can make informed decisions about your specific situation.
Here's what the data tells us at a high level: roughly 55% of peptide-medication combinations show no meaningful interaction whatsoever. Another 22% produce minor effects that rarely require dose adjustments. About 15% create moderate interactions that warrant monitoring or timing changes. The remaining 8% fall into major or contraindicated categories - and those are the ones that truly matter for patient safety (DOI: 10.1007/s40264-023-01392-3).
Key Takeaways From This Report
- GLP-1 receptor agonists affect oral medication absorption through delayed gastric emptying, but most interactions are clinically manageable
- Growth hormone peptides can alter glucose metabolism, making diabetes medication adjustments necessary in some patients
- Healing peptides like BPC-157 lack formal drug interaction studies, requiring extra caution
- Immune-modulating peptides such as Thymosin Alpha-1 are potentially contraindicated with immunosuppressive medications
- Timing strategies can mitigate many absorption-based interactions
- Perioperative management of peptide therapies requires advance planning, particularly for GLP-1 agents
Whether you're a clinician managing complex medication regimens, a researcher evaluating combination protocols, or a patient trying to understand how your peptide therapy fits alongside existing prescriptions, this report provides the evidence-based framework you need. We've compiled data from pharmacokinetic studies, FDA prescribing information, clinical trial databases, case reports, and systematic reviews to give you the most thorough picture available as of early 2026.
For foundational information about peptide therapy, see our Peptide Therapy Beginner's Guide. For monitoring recommendations during peptide use, visit our Blood Work Monitoring Guide.
Figure 1: Distribution of peptide-drug interaction severity across all major peptide classes. The majority of interactions are minor or absent, but clinically significant combinations exist in every category.
Pharmacology of Peptide-Drug Interactions
Understanding why peptide-drug interactions happen requires a brief tour through the pharmacological mechanisms at play. Unlike small-molecule drugs that often compete for the same metabolic enzymes, peptides create interactions through fundamentally different pathways.
How Drug Interactions Occur: A Framework
Drug interactions generally fall into three broad categories: pharmacokinetic interactions (what the body does to the drug), pharmacodynamic interactions (what the drug does to the body), and pharmaceutical interactions (physical or chemical incompatibilities). Peptide therapies can trigger all three types, though the mechanisms differ substantially from traditional drug-drug interactions.
Most prescription drugs are metabolized through the cytochrome P450 (CYP) enzyme system in the liver. This is where the vast majority of conventional drug interactions occur - one drug inhibits or induces a CYP enzyme, changing the blood levels of another drug metabolized by that same enzyme. Peptide therapies, by contrast, are broken down through proteolysis - enzymatic degradation by peptidases throughout the body. They don't typically interact with CYP enzymes directly (DOI: 10.1007/s40262-017-0532-6).
This distinction is fundamental. It means that the traditional drug interaction databases and prediction tools designed for small-molecule drugs often don't capture peptide interactions accurately. The mechanisms at work are different, and they require a different analytical framework.
Absorption-Based Interactions
The single most clinically relevant interaction mechanism for peptide therapies - particularly GLP-1 receptor agonists - involves changes in gastrointestinal absorption. When a peptide slows gastric emptying, every oral medication you take is affected to some degree. The pill sits in your stomach longer before reaching the small intestine, where most drug absorption occurs.
This doesn't always mean less drug gets absorbed. Sometimes it means the drug absorbs more slowly, producing a lower peak concentration but the same total exposure. In other cases, the prolonged transit time actually increases absorption because the drug has more contact time with absorptive surfaces. The clinical significance depends entirely on the specific drug's pharmacokinetic profile (DOI: 10.1002/phar.70007).
Drugs with narrow therapeutic windows - where small changes in blood levels can mean the difference between effectiveness and toxicity - are most vulnerable to absorption-based interactions. Warfarin, levothyroxine, certain anti-epileptic drugs, and immunosuppressants like tacrolimus all fall into this category.
The Gastric Emptying Effect
GLP-1 receptor agonists are the primary offenders here. Semaglutide, tirzepatide, liraglutide, and other GLP-1 drugs work partly by activating receptors in the gut that slow stomach emptying. This produces satiety and helps control postprandial glucose spikes - both desirable effects. But it also means every oral medication you take alongside these agents faces a different absorption environment.
The magnitude of the gastric emptying delay varies by compound. Tirzepatide appears to produce a more pronounced delay than most pure GLP-1 agonists, likely due to its dual GIP/GLP-1 mechanism. The delay is typically most significant during the dose-escalation phase and tends to attenuate somewhat at steady state, though it never fully normalizes while therapy continues (DOI: 10.1093/jcem/dgae510).
Clinical Pearl
The gastric emptying delay from GLP-1 agonists is most pronounced with the first dose and during dose escalation. Pharmacokinetic studies show that the effect on co-administered drug absorption often diminishes by Week 6 of treatment at a stable dose. This means the highest-risk period for absorption-based interactions is during the first few weeks of therapy or after any dose increase.
Distribution-Based Interactions
Once a drug is absorbed into the bloodstream, it distributes throughout the body. Many drugs bind to plasma proteins like albumin, and only the unbound fraction is pharmacologically active. Peptide therapies can alter drug distribution through several mechanisms.
Weight loss itself changes drug distribution. As body composition shifts - less adipose tissue, potentially different lean mass - drugs that distribute primarily into fat (lipophilic drugs) may achieve higher plasma concentrations at the same dose. This is particularly relevant for GLP-1 agonists used for weight management, where patients can lose 15-25% of their body weight over 12-18 months. A dose of warfarin or levothyroxine that was appropriate at 250 pounds may become supratherapeutic at 190 pounds.
Growth hormone secretagogues add another layer. By increasing growth hormone and IGF-1 levels, these peptides can alter the synthesis of binding proteins, potentially changing free drug concentrations for medications that are highly protein-bound. The clinical significance of this mechanism is less well-characterized than absorption effects, but it represents a real physiological pathway for interaction (DOI: 10.3390/ijms25042208).
Metabolism-Based Interactions
Here's where peptides diverge most dramatically from conventional drugs. Most therapeutic peptides don't undergo hepatic metabolism through CYP enzymes. They're degraded by ubiquitous peptidases throughout the body. This means they rarely cause the classic CYP-mediated drug interactions that dominate conventional pharmacology.
In vitro studies confirm this for the major peptide classes. Semaglutide shows low potential for CYP inhibition or induction. Tirzepatide similarly demonstrates minimal CYP interaction potential. Growth hormone secretagogues like ipamorelin and CJC-1295 are proteolytically degraded without significant CYP involvement (DOI: 10.1007/s40262-017-0532-6).
However, peptides can indirectly affect drug metabolism through their physiological effects. Growth hormone, stimulated by secretagogue peptides, can influence CYP enzyme expression. Studies have shown that GH replacement therapy can alter the metabolism of drugs processed by CYP3A4, CYP2C19, and other isoforms. The magnitude of this effect from secretagogue-induced GH release (which is pulsatile and physiological rather than continuous) is probably smaller than from exogenous GH replacement, but it has not been formally studied (DOI: 10.1177/0091270003258651).
Pharmacodynamic Interactions
Pharmacodynamic interactions occur when two drugs affect the same physiological system, either amplifying or opposing each other's effects. These are among the most clinically significant peptide-drug interactions.
The most obvious example: combining GLP-1 agonists with sulfonylureas or insulin. Both lower blood glucose, but through different mechanisms. The GLP-1 agonist's glucose-lowering effect is glucose-dependent - it works harder when glucose is high and backs off when it's normal. Sulfonylureas, by contrast, stimulate insulin release regardless of blood glucose levels. When you combine them, the sulfonylurea uncouples the GLP-1 agonist from its built-in safety mechanism, dramatically increasing hypoglycemia risk (DOI: 10.2337/db06-0903).
Similarly, growth hormone secretagogues create pharmacodynamic interactions with diabetes medications. GH antagonizes insulin action, promoting gluconeogenesis and reducing peripheral glucose uptake. A patient on insulin or metformin who starts a GH secretagogue may find their glucose control deteriorating - not because the GH peptide interfered with the medication's absorption or metabolism, but because it's pushing glucose in the opposite direction.
Figure 2: The four pathways through which peptide therapies can interact with conventional medications. Absorption-based and pharmacodynamic interactions are the most clinically significant for most peptide classes.
Peptide-Specific Considerations
Several factors make peptide pharmacology unique when it comes to interactions:
- Route of administration matters. Most therapeutic peptides are injected subcutaneously, bypassing the GI tract entirely. This eliminates many absorption-based interaction concerns for the peptide itself - though it doesn't eliminate the peptide's effects on oral medications taken concurrently.
- Half-life varies enormously. Some peptides (like native GLP-1) have half-lives of minutes, while modified versions (like semaglutide) persist for days. Longer-acting peptides create sustained physiological effects that continuously influence co-administered drug behavior.
- Dose-response relationships are often non-linear. The interaction potential of a peptide at a low dose may be very different from the same peptide at a high dose. This is particularly relevant during dose escalation phases.
- Receptor desensitization can change interactions over time. The body's response to chronic peptide exposure may shift as receptors downregulate or as compensatory mechanisms engage. An interaction that's clinically significant at Week 2 may be negligible at Week 12.
Understanding these mechanisms provides the foundation for the drug-specific interaction profiles covered in the following sections. With this framework in mind, we can evaluate each combination not just as a yes-or-no question, but as a nuanced assessment of mechanism, magnitude, timing, and clinical relevance.
GLP-1 Receptor Agonist Drug Interactions
GLP-1 receptor agonists represent the most widely prescribed peptide class, and consequently the most thoroughly studied for drug interactions. Semaglutide alone has been evaluated in formal pharmacokinetic interaction studies with dozens of commonly co-administered medications.
The core interaction mechanism for GLP-1 agonists centers on delayed gastric emptying. By activating GLP-1 receptors in the gut, these drugs slow the rate at which stomach contents pass into the small intestine. This physiological effect is integral to their therapeutic benefit - it promotes satiety and blunts postprandial glucose excursions. But it also creates a changed absorption environment for every oral medication a patient takes.
That said, the clinical impact is more modest than many providers initially feared. A comprehensive systematic review published in Drug Safety evaluated all available pharmacokinetic interaction data for GLP-1 receptor agonists and concluded that most co-administered medications were not affected to a clinically relevant degree (DOI: 10.1007/s40264-023-01392-3). The FDA prescribing information for semaglutide echoes this: while delayed gastric emptying has the potential to impact absorption of oral medications, clinical pharmacology trials found no clinically meaningful effects on the drugs tested.
Medications With Confirmed Minimal Interaction
Formal pharmacokinetic studies have evaluated semaglutide co-administration with several commonly used drugs. The results are reassuring for most combinations:
| Medication | Effect on AUC | Effect on Cmax | Clinical Significance |
|---|---|---|---|
| Metformin | No meaningful change | Slightly delayed Tmax | None - safe to combine |
| Warfarin (S-warfarin) | No meaningful change | No meaningful change | None in PK studies (see caveats below) |
| Atorvastatin | No meaningful change | Decreased ~38% | Minimal - total exposure unchanged |
| Digoxin | No meaningful change | Decreased ~22% | Monitor - narrow therapeutic index |
| Ethinylestradiol | No meaningful change | No meaningful change | None for semaglutide specifically |
| Levonorgestrel | No meaningful change | No meaningful change | None for semaglutide specifically |
These studies were conducted with subcutaneous semaglutide. The results suggest that while gastric emptying is delayed, the total amount of drug absorbed over time remains essentially the same. Peak concentrations may be slightly lower and delayed, but the area under the curve (total exposure) is preserved (DOI: 10.1007/s40262-017-0532-6).
Warfarin: A Nuanced Story
Warfarin deserves special attention despite the reassuring pharmacokinetic data. While formal PK studies showed no significant changes in warfarin exposure or INR when combined with semaglutide, real-world clinical experience tells a more nuanced story.
A study presented at the American Society of Hematology annual meeting evaluated warfarin time in therapeutic range (TTR) before and after GLP-1 agonist initiation. Average TTR was 59% and 60% for the 90 days before and after GLP-1 initiation respectively - not statistically different. However, some individual patients who were previously well-controlled experienced significant INR fluctuations after starting GLP-1 therapy (DOI: 10.1182/blood-2024-211293).
The mechanism likely involves multiple factors beyond simple absorption changes:
- Weight loss reduces warfarin dose requirements. As patients lose weight on GLP-1 therapy, a previously stable warfarin dose may become supratherapeutic.
- Dietary changes associated with GLP-1-induced appetite suppression can alter vitamin K intake, directly affecting warfarin's mechanism of action.
- Reduced food intake overall may change warfarin absorption kinetics independently of gastric emptying effects.
Clinical Recommendation
For patients on warfarin who start GLP-1 agonist therapy: increase INR monitoring frequency during the first 8-12 weeks of GLP-1 treatment and after each dose escalation. Continue enhanced monitoring during active weight loss phases. Warfarin dose adjustments of 10-20% may be needed as body weight decreases significantly.
Oral Contraceptives: Agent-Specific Differences
The interaction between GLP-1 class drugs and oral hormonal contraceptives varies dramatically depending on which agent is used. This is one area where it's critical not to generalize across the class.
Semaglutide: Formal pharmacokinetic studies demonstrated that subcutaneous semaglutide does not reduce the bioavailability of combined oral contraceptives containing ethinylestradiol and levonorgestrel. No backup contraception is recommended in the prescribing information (DOI: 10.1002/jcph.443).
Liraglutide, exenatide extended-release, dulaglutide: Similarly, these agents have not shown clinically significant effects on oral contraceptive absorption in formal studies. No additional contraceptive precautions are advised.
Tirzepatide: This is the exception. Tirzepatide has demonstrated a more pronounced effect on oral contraceptive absorption, reducing exposure by approximately 20%. The manufacturer specifically recommends backup contraception for 4 weeks after tirzepatide initiation and after each dose increase. Alternatively, patients can switch to a non-oral contraceptive method - IUD, implant, patch, or ring - to eliminate the interaction entirely (DOI: 10.1016/j.contraception.2023.110209).
Lixisenatide: The short-acting GLP-1 agonist lixisenatide creates a more pronounced acute delay in gastric emptying. The prescribing information recommends taking oral contraceptives either at least 1 hour before or at least 11 hours after lixisenatide administration.
Key Distinction
The reason tirzepatide differs from pure GLP-1 agonists likely relates to its dual GIP/GLP-1 mechanism, which may produce a more sustained or more pronounced delay in gastric emptying. When counseling patients on contraceptive interactions, the specific agent matters enormously - blanket statements about "GLP-1 drugs and birth control" can be misleading.
Insulin and Sulfonylurea Combinations
This represents the most clinically significant pharmacodynamic interaction for GLP-1 agonists. The risk is hypoglycemia, and the numbers are striking.
GLP-1 agonist monotherapy produces documented symptomatic hypoglycemia in only 1.6-3.8% of patients - a very low rate that reflects the glucose-dependent nature of their insulin-stimulating effect. When the same GLP-1 agonists are combined with sulfonylureas, however, hypoglycemia rates jump to 17.3-24.4%. That's roughly a 5-7 fold increase (DOI: 10.2337/db06-0903).
The mechanism is elegant but dangerous. GLP-1 agonists stimulate insulin secretion in a glucose-dependent manner - they only work when blood glucose is elevated. Sulfonylureas bypass this safety mechanism by directly stimulating beta-cell insulin release regardless of glucose levels. When combined, the sulfonylurea essentially "uncouples" the GLP-1 agonist from its built-in glucose safety switch.
Insulin combinations carry similar risk, though the mechanism is different. Exogenous insulin acts independently of endogenous glucose-dependent pathways. Adding a GLP-1 agonist that enhances endogenous insulin secretion on top of exogenous insulin creates additive glucose-lowering that can easily overshoot.
Dose Adjustment Protocols
When initiating a GLP-1 agonist in a patient already on sulfonylureas or insulin:
- Sulfonylureas: Reduce the sulfonylurea dose by 50% when starting the GLP-1 agonist. Monitor fasting glucose and A1c at 4-week intervals. Further reductions or discontinuation may be appropriate as the GLP-1 agonist is titrated up.
- Basal insulin: Reduce insulin dose by 20-25% at GLP-1 initiation. Titrate based on fasting glucose targets. Many patients can eventually discontinue insulin entirely on optimized GLP-1 therapy.
- Prandial insulin: More aggressive reduction may be needed - consider 30-50% initial reduction with careful postprandial glucose monitoring.
For detailed guidance on blood glucose monitoring during peptide therapy transitions, see our blood work monitoring guide.
Metformin: A Favorable Combination
Metformin and GLP-1 agonists represent one of the most well-studied and well-tolerated drug combinations in modern endocrinology. Pharmacokinetic studies confirm no clinically relevant interaction, and clinical trials consistently demonstrate superior glycemic control with the combination compared to either agent alone.
A systematic review and meta-analysis of semaglutide combined with metformin in type 2 diabetes patients who were overweight or obese found significant improvements across multiple endpoints: fasting blood glucose, 2-hour postprandial glucose, HbA1c, insulin resistance, weight, BMI, and lipid profiles. Adverse event rates were comparable to semaglutide monotherapy - mostly GI-related and mild to moderate in severity (DOI: 10.3389/fphar.2024.1401207).
The complementary mechanisms make pharmacological sense. Metformin reduces hepatic glucose production and improves insulin sensitivity. GLP-1 agonists enhance glucose-dependent insulin secretion, suppress glucagon, and slow gastric emptying. They attack hyperglycemia from completely different angles without amplifying each other's risks.
Levothyroxine: Monitor Thyroid Function
The interaction between GLP-1 agonists and levothyroxine is clinically meaningful and often underappreciated. In pharmacokinetic studies with oral semaglutide, levothyroxine exposure increased by approximately 33% when the two drugs were co-administered. Case reports have documented suppressed TSH levels after GLP-1 agonist initiation in patients previously stable on levothyroxine (DOI: 10.1080/17425255.2021.1955856).
Two mechanisms contribute to this interaction:
- Enhanced absorption: Delayed gastric emptying gives levothyroxine more time in contact with absorptive surfaces, potentially increasing the amount absorbed.
- Weight-based dosing changes: Levothyroxine dosing is weight-dependent (roughly 1.6 mcg/kg/day for full replacement). As patients lose weight on GLP-1 therapy, a previously appropriate dose may become supratherapeutic.
The combined effect can be substantial. A patient who loses 20% of their body weight while experiencing enhanced levothyroxine absorption may effectively be getting 40-50% more thyroid hormone activity than intended.
Monitoring Protocol
Check TSH and free T4 at baseline before starting GLP-1 therapy, again at 6-8 weeks, and after each significant dose change or after every 10 pounds of weight loss. Levothyroxine dose reductions of 10-25% are commonly needed in patients achieving significant weight loss on GLP-1 agonists.
Cardiovascular Medications
Most cardiovascular medications can be safely co-administered with GLP-1 agonists, but several warrant discussion:
Statins
Atorvastatin peak concentration (Cmax) decreased by approximately 38% when co-administered with semaglutide, but total exposure (AUC) was unchanged. This means the statin is absorbed more slowly but just as completely. Since statins work through sustained enzyme inhibition rather than peak concentration effects, this interaction has no clinical significance. No dose adjustment is needed.
ACE Inhibitors and ARBs
No formal interaction studies exist specifically for these combinations, but the pharmacological profiles suggest minimal concern. These drugs are well-absorbed across a range of conditions, and their long half-lives make them relatively insensitive to changes in absorption rate. GLP-1 agonists' cardiovascular benefits (demonstrated in outcomes trials like LEADER and SELECT) may actually complement the cardioprotective effects of ACE inhibitors and ARBs.
Digoxin
Digoxin peak concentration decreased by approximately 22% with semaglutide co-administration, while total exposure was unchanged. Given digoxin's narrow therapeutic index, the FDA prescribing information recommends monitoring. In practice, patients on stable digoxin therapy who start GLP-1 agonists should have digoxin levels checked at 4-6 weeks and after dose escalation.
Antiarrhythmics
Drugs like amiodarone, flecainide, and sotalol have not been formally studied with GLP-1 agonists. Given their narrow therapeutic windows and the potential for absorption timing changes, clinical monitoring is advisable. For patients on antiarrhythmics with GLP-1 therapy, check drug levels more frequently during dose escalation and weight loss phases.
Proton Pump Inhibitors and H2 Blockers
An interesting bidirectional consideration arises with acid-suppressing medications. GLP-1 agonists can cause nausea and GI discomfort, leading providers to prescribe PPIs or H2 blockers. These drugs can be safely combined from a pharmacokinetic standpoint - no formal interactions have been identified.
However, clinicians should be aware that PPIs alter the absorption of many other co-administered medications (iron, calcium, magnesium, B12) through pH changes. Adding gastric emptying delays from GLP-1 agonists on top of PPI-induced pH changes creates a doubly altered absorption environment. While this is rarely clinically problematic, it's worth considering in patients on complex medication regimens.
Psychotropic Medications
Several case reports have described altered psychotropic drug levels in patients starting GLP-1 agonist therapy. One well-documented case involved a patient on ziprasidone (Geodon) who developed supratherapeutic drug levels after starting semaglutide, with a measured level of 238.7 ng/mL against a recommended maximum of 220 ng/mL.
SSRIs, SNRIs, and other antidepressants have not been formally studied with GLP-1 agonists. Given their generally wide therapeutic windows, clinically significant interactions are unlikely for most patients. However, patients on lithium (another narrow therapeutic index drug) warrant careful monitoring, as both absorption changes and weight-loss-related dehydration could affect lithium levels.
Figure 3: Summary of GLP-1 receptor agonist interaction profiles across major medication classes. Green indicates minimal/no interaction, yellow indicates monitoring recommended, red indicates dose adjustment typically required.
Anticoagulants Beyond Warfarin
Direct oral anticoagulants (DOACs) - apixaban, rivarelbaan, edoxaban, and dabigatran - have not been formally studied with GLP-1 agonists. These drugs generally have wider therapeutic windows than warfarin and don't require routine monitoring. However, delayed gastric emptying could theoretically affect their absorption kinetics.
Of particular note, dabigatran (Pradaxa) is the most absorption-sensitive DOAC, requiring acidic gastric pH for optimal dissolution. Changes in gastric emptying and pH could theoretically affect its bioavailability more than the other DOACs. While no clinical reports of significant interactions exist, awareness of this theoretical concern is prudent.
Oral Semaglutide: A Special Case
Oral semaglutide (Rybelsus) creates unique interaction considerations not shared by injectable formulations. The tablet must be taken on an empty stomach with no more than 4 ounces of plain water, at least 30 minutes before any food, drink, or other oral medications. This mandatory dosing window inherently separates oral semaglutide from most other medications.
However, the levothyroxine interaction is amplified with oral semaglutide compared to injectable formulations, with the 33% increase in levothyroxine exposure documented specifically with the oral formulation. Patients taking both oral semaglutide and levothyroxine need particularly careful thyroid function monitoring.
For a broader overview of GLP-1 medication options, visit our GLP-1 information hub.
Oral Medication Absorption Effects
Delayed gastric emptying is the most common mechanism by which peptide therapies affect co-administered oral drugs. Understanding the kinetics of this delay - and which drug properties make a medication more or less vulnerable - allows clinicians and patients to make smarter timing decisions.
The Science of Delayed Gastric Emptying
Normal gastric emptying follows a predictable pattern. Liquids begin leaving the stomach within minutes. Solid foods empty over 2-4 hours, with a lag phase during which the stomach grinds food into small particles, followed by a linear emptying phase. GLP-1 receptor agonists extend both phases - the lag period lengthens, and the linear emptying rate slows.
A physiologically-based pharmacokinetic (PBPK) modeling study published in 2025 predicted the effects of GLP-1 RA-induced gastric emptying delays on co-administered drug absorption. The models showed that drugs with rapid, complete absorption in the proximal small intestine were least affected by gastric emptying delays. Drugs requiring dissolution at specific pH levels or those with absorption windows limited to particular GI segments were most vulnerable (DOI: 10.1002/phar.70007).
Drug Properties That Increase Vulnerability
Not all oral medications respond the same way to delayed gastric emptying. Several drug properties increase vulnerability to this interaction:
| Drug Property | Why It Matters | Examples |
|---|---|---|
| Narrow therapeutic index | Small PK changes produce clinically meaningful effects | Warfarin, digoxin, lithium, phenytoin, tacrolimus |
| pH-dependent solubility | Prolonged gastric exposure at low pH may alter dissolution | Ketoconazole, itraconazole, dabigatran |
| Absorption window in upper GI tract | Delayed transit may move drug past absorption window | Levodopa, certain fluoroquinolones |
| Enteric-coated formulations | Delayed gastric emptying prolongs time before coating dissolves | Omeprazole DR, aspirin EC, duloxetine DR |
| Extended-release formulations | Altered transit may change drug release kinetics | Metformin XR, venlafaxine XR, nifedipine XL |
| High first-pass metabolism | Changed absorption rate may alter first-pass extraction ratio | Propranolol, verapamil, morphine |
The Acetaminophen Model
Acetaminophen (Tylenol) serves as a useful pharmacokinetic marker for gastric emptying because it's rapidly and completely absorbed from the small intestine. Changes in acetaminophen absorption kinetics reflect changes in gastric emptying rate with high fidelity.
Tirzepatide studies used acetaminophen as a probe drug and revealed important temporal patterns. After the first dose of tirzepatide 5 mg, acetaminophen peak concentration (Cmax) dropped by 55% and time to peak was delayed by about 1 hour. This is a dramatic reduction in absorption rate. However, by Week 6 at the 15 mg dose, the effect on Cmax and Tmax had largely resolved, and total acetaminophen exposure (AUC) was not affected at any timepoint.
This pattern - pronounced acute effect that attenuates over time while total exposure is preserved - characterizes most GLP-1 agonist absorption interactions. It tells us three things:
- The first weeks of therapy and dose escalation periods carry the highest interaction risk
- Steady-state therapy produces a more moderate effect
- For most drugs, the total amount absorbed doesn't change - just the timing
Timing Strategies to Minimize Absorption Interactions
Simple timing adjustments can mitigate many absorption-based interactions. The general principle: separate the oral medication from the peptide's peak gastric emptying effect.
For Once-Weekly Injectable GLP-1 Agonists (Semaglutide, Tirzepatide)
Because these agents produce sustained gastric emptying delay throughout the week, time-of-day separation from the injection doesn't help much. However, consistency matters - take your oral medications at the same time each day, with consistent food/fasting status, to minimize variability even if absorption is somewhat delayed.
For Once-Daily Injections (Liraglutide)
Consider taking critical oral medications first thing in the morning, before the daily injection. This provides a window of relatively less GLP-1 effect (the tail end of the previous day's dose) for medication absorption.
For Short-Acting Agents (Lixisenatide, Exenatide BID)
These create pronounced but brief gastric emptying delays. Timing is most effective here: take narrow-therapeutic-index oral medications at least 1 hour before or 4 hours after the injection. The FDA specifically recommends this approach for lixisenatide.
Practical Timing Protocol
Morning routine for patients on GLP-1 therapy with multiple oral medications:
- Take levothyroxine on empty stomach with water (if applicable)
- Wait 30-60 minutes
- Take other morning medications with a small amount of food
- Inject GLP-1 agonist (if daily formulation)
- Wait 30 minutes before eating a full meal
This sequence prioritizes absorption of the most sensitive medication (levothyroxine) while maintaining consistency for all others.
Special Populations and Increased Risk
Certain patient populations face amplified absorption interaction risk:
- Patients with pre-existing gastroparesis: Already have impaired gastric emptying. Adding GLP-1 agonist-induced delay compounds the problem, potentially causing significant absorption variability.
- Elderly patients: May have age-related reductions in gastric motility and acid production, making them more susceptible to additional emptying delays.
- Patients on polypharmacy: Complex drug regimens create more opportunities for absorption interactions. Each additional oral medication is another variable affected by gastric emptying changes.
- Post-surgical patients: Altered GI anatomy (gastric bypass, sleeve gastrectomy) combined with GLP-1-induced motility changes creates an unpredictable absorption environment. Use these combinations with caution and enhanced monitoring.
For comprehensive guidance on monitoring during peptide therapy, see our side effects reference guide.
Growth Hormone Peptide Interactions
Growth hormone secretagogues - including CJC-1295/Ipamorelin, MK-677 (Ibutamoren), GHRP-2, GHRP-6, and tesamorelin - create drug interactions primarily through their downstream metabolic effects rather than direct pharmacokinetic interference.
Unlike GLP-1 agonists, these peptides don't significantly alter gastric emptying. Instead, they raise growth hormone (GH) and insulin-like growth factor 1 (IGF-1) levels, which in turn influence glucose metabolism, insulin sensitivity, thyroid function, and cortisol dynamics. These endocrine shifts create pharmacodynamic interactions with multiple drug classes.
Diabetes Medications
Growth hormone is a counter-regulatory hormone to insulin. It promotes hepatic gluconeogenesis, reduces peripheral glucose uptake, and stimulates lipolysis. When GH levels rise - whether from exogenous GH replacement or from secretagogue stimulation - insulin resistance increases and blood glucose tends to climb.
The magnitude of this effect varies by compound:
MK-677 (Ibutamoren)
MK-677 produces the most significant glucose-related effects among commonly used secretagogues. Clinical trials consistently show fasting glucose increases averaging approximately 5 mg/dL, with some individuals experiencing much larger elevations. In one study, MK-677 was reduced from 25 mg to 10 mg daily in five patients whose fasting blood glucose exceeded 140 mg/dL, and was discontinued entirely in three patients due to persistent hyperglycemia (DOI: 10.1210/jcem.83.2.4539).
Case reports have documented new-onset diabetes triggered by MK-677 use, particularly in individuals with pre-existing risk factors. One published case described a previously normoglycemic bodybuilder who developed overt type 2 diabetes while using MK-677 in combination with SARMs (DOI: 10.3390/medicina58070959).
High-Risk Combination
MK-677 combined with insulin or sulfonylureas creates a push-pull effect on glucose - the MK-677 pushes glucose up while the diabetes medication pushes it down. This doesn't simply cancel out. It creates unpredictable glucose swings and increases the risk of both hyperglycemia (when MK-677 effects dominate) and hypoglycemia (when diabetes medication effects dominate). If this combination must be used, intensive glucose monitoring with CGM is strongly recommended.
CJC-1295/Ipamorelin
CJC-1295/Ipamorelin produces a more physiological, pulsatile GH release compared to MK-677's sustained elevation. This pulsatile pattern more closely mimics natural GH secretion and tends to produce less insulin resistance than sustained GH elevation. Still, patients on diabetes medications should monitor glucose more frequently when starting this peptide combination.
The effect on glucose metabolism is dose-dependent and usually mild at standard therapeutic doses. Most patients on metformin alone can tolerate CJC-1295/Ipamorelin without dose adjustments, though fasting glucose may increase by 5-15 mg/dL. Patients on insulin or sulfonylureas need closer monitoring and potential dose adjustments.
Tesamorelin
Tesamorelin, the only FDA-approved GHRH analog (for HIV-associated lipodystrophy), has the most formal safety data among secretagogue-type peptides. In clinical trials, tesamorelin produced modest increases in fasting glucose and HbA1c, with a slightly higher incidence of new-onset diabetes compared to placebo in the HIV population studied. These patients had elevated baseline metabolic risk, so the findings may not generalize to healthier populations.
Thyroid Medications
GH and thyroid hormones have a complex bidirectional relationship. GH stimulates the peripheral conversion of T4 to the more active T3 by enhancing deiodinase enzyme activity. It can also suppress TSH through hypothalamic feedback loops.
For patients on levothyroxine replacement:
- T3 levels may increase as GH secretagogues enhance T4-to-T3 conversion, even if the levothyroxine dose hasn't changed
- TSH may become suppressed through combined GH and increased T3 effects on the hypothalamic-pituitary axis
- Free T4 may decrease as more T4 is converted to T3, potentially triggering concerns about "low T4" that are actually physiologically appropriate
The clinical implication: standard thyroid panels (TSH and free T4) may become harder to interpret in patients using GH secretagogues. Adding free T3 and reverse T3 to monitoring panels provides a more complete picture. Levothyroxine dose adjustments should be based on clinical symptoms and the full thyroid panel, not TSH alone.
Cortisol and Adrenal Medications
GH influences cortisol metabolism through its effect on 11-beta-hydroxysteroid dehydrogenase (11B-HSD), the enzyme that interconverts active cortisol and inactive cortisone. GH tends to reduce cortisol levels by shifting the balance toward cortisone. In patients with marginal adrenal reserve, starting a GH secretagogue could unmask adrenal insufficiency.
For patients on hydrocortisone or prednisone replacement (for adrenal insufficiency or Addison's disease), GH secretagogue initiation should be accompanied by cortisol monitoring. Stress-dose hydrocortisone protocols should be reviewed and reinforced.
In patients taking supraphysiological glucocorticoids (for inflammatory conditions), the interaction works differently. Chronic glucocorticoid excess suppresses GH secretion, so the expected GH release from secretagogue peptides may be blunted. The clinical significance is reduced efficacy of the secretagogue rather than a safety concern.
Blood Pressure Medications
Growth hormone has acute and chronic effects on fluid balance and vascular tone. Acutely, GH can cause mild fluid retention through renal sodium reabsorption. Chronically, IGF-1 has vasodilatory effects that tend to reduce blood pressure. These opposing forces mean the net effect on blood pressure is variable and usually modest.
For patients on antihypertensive medications, GH secretagogues are unlikely to require dose adjustments. However, the fluid retention effect - which manifests as mild edema, joint stiffness, or carpal tunnel-like symptoms - can be confused with medication side effects or dose changes. Patients should be counseled about this possibility to avoid unnecessary medication adjustments.
Anti-Cancer Medications
Growth hormone and IGF-1 have proliferative effects on many cell types. While the relationship between GH/IGF-1 and cancer risk is complex and debated, GH secretagogues are generally considered contraindicated in patients with active cancer or a recent cancer history (within 5 years, depending on the cancer type).
For patients on cancer treatment - chemotherapy, targeted therapy, immunotherapy, or hormonal therapy - GH secretagogues should not be used without explicit oncologist approval. The theoretical risk of promoting tumor growth or interfering with anti-cancer therapy mechanisms is too significant to justify the potential benefits of GH optimization during active treatment.
Figure 4: Comparative effect of different GH secretagogues on fasting blood glucose. MK-677 produces the most pronounced glucose elevation, while CJC-1295/Ipamorelin creates a more modest, physiological response.
Interaction With Exogenous Testosterone and Anabolic Hormones
Many individuals using GH secretagogues are also on testosterone replacement therapy (TRT). This combination is generally well-tolerated from an interaction standpoint, but warrants monitoring for additive effects:
- Both GH and testosterone increase IGF-1 levels - combined use may produce supraphysiological IGF-1 that warrants monitoring
- Both can worsen insulin resistance, particularly in higher doses - glucose monitoring becomes more important with the combination
- Both can increase hematocrit - complete blood count monitoring should be maintained
- GH can enhance the anabolic effects of testosterone, which may be therapeutically desirable but should be clinically managed
Monitoring Recommendations for GH Peptide Users
For patients on GH secretagogues alongside other medications, the following monitoring schedule is recommended:
| Test | Baseline | 4 Weeks | 12 Weeks | Ongoing |
|---|---|---|---|---|
| Fasting glucose | Yes | Yes | Yes | Every 3 months |
| HbA1c | Yes | - | Yes | Every 3-6 months |
| Fasting insulin | Yes | Yes | Yes | Every 6 months |
| IGF-1 | Yes | Yes | Yes | Every 3-6 months |
| TSH, free T4, free T3 | Yes | - | Yes | Every 6 months |
| Cortisol (AM) | Yes | - | Yes | As indicated |
| CBC with differential | Yes | - | Yes | Every 6 months |
Use our dosing calculator for protocol planning and our blood work guide for detailed monitoring protocols.
Insulin & Diabetes Medication Interactions
Diabetes medications form the most complex web of interactions with peptide therapies. Multiple peptide classes affect glucose homeostasis - some lowering it, others raising it - and the interaction profile changes depending on which diabetes drug is involved.
This section provides a systematic breakdown of how each major diabetes medication class interacts with each major peptide category. The goal is a practical reference that clinicians can consult when managing patients on combination therapy.
Insulin (All Types) + Peptide Therapy
With GLP-1 Agonists
Interaction severity: Major
Adding GLP-1 agonists to insulin creates additive glucose-lowering that requires proactive insulin dose reduction. The standard recommendation is a 20-25% reduction in basal insulin at GLP-1 initiation, with further reductions guided by glucose monitoring. Prandial insulin may need 30-50% reduction.
The GLP-1 agonist adds glucose-dependent insulin secretion, glucagon suppression, and delayed gastric emptying to whatever the exogenous insulin is doing. Because the GLP-1 component has a built-in glucose-dependent safety mechanism, the risk of severe hypoglycemia is lower than combining two non-glucose-dependent agents. Still, hypoglycemia rates are significantly elevated compared to GLP-1 monotherapy.
Fixed-ratio combinations of GLP-1 agonists with basal insulin (like iDegLira, which combines degludec insulin with liraglutide) have been developed specifically to optimize this interaction. These products use carefully calibrated dose ratios that maintain efficacy while minimizing hypoglycemia risk.
With GH Secretagogues
Interaction severity: Moderate to Major
GH secretagogues tend to increase insulin resistance, potentially destabilizing glucose control in insulin-dependent patients. The interaction is pharmacodynamic - GH opposes insulin's glucose-lowering action at the cellular level. Patients may need insulin dose increases of 10-30% when starting GH secretagogue therapy, particularly with MK-677.
The practical challenge: the GH-induced insulin resistance develops gradually over days to weeks, making it easy to miss until fasting glucose or HbA1c has drifted upward. Proactive monitoring with continuous glucose monitoring (CGM) during GH secretagogue initiation is ideal for insulin-dependent patients.
With Healing Peptides (BPC-157)
Interaction severity: Likely minimal
No formal studies exist, but BPC-157's mechanism of action (angiogenesis, tissue repair, nitric oxide modulation) doesn't directly affect glucose homeostasis. Animal studies have shown some cytoprotective effects on pancreatic tissue, but no glucose-lowering or glucose-raising effects that would interact with insulin therapy. Standard insulin dosing can be maintained, but as always, report any unexpected glucose changes to your provider.
Sulfonylureas (Glipizide, Glyburide, Glimepiride) + Peptide Therapy
With GLP-1 Agonists
Interaction severity: Major
As discussed earlier, this combination dramatically increases hypoglycemia risk. Sulfonylureas stimulate insulin release regardless of blood glucose, removing the glucose-dependent safety mechanism that makes GLP-1 agonists relatively hypoglycemia-safe as monotherapy. Documented hypoglycemia rates jump from under 4% to 17-24% with the combination (DOI: 10.2337/db06-0903).
Standard protocol: reduce sulfonylurea dose by 50% when initiating GLP-1 therapy. Many endocrinologists prefer to discontinue sulfonylureas entirely within 4-8 weeks of reaching therapeutic GLP-1 doses, replacing the glucose-lowering contribution with the GLP-1 agonist itself.
With GH Secretagogues
Interaction severity: Moderate
GH-induced insulin resistance partially opposes the glucose-lowering effect of sulfonylureas. This creates an unpredictable tug-of-war - the sulfonylurea pushes insulin up, GH reduces insulin sensitivity. The net effect on glucose varies by individual and by timing. Close glucose monitoring is essential, but severe hypoglycemia risk is lower than the GLP-1/sulfonylurea combination because the glucose effects partially offset.
Metformin + Peptide Therapy
With GLP-1 Agonists
Interaction severity: Minimal - Favorable combination
This is one of the best-studied and most favorable drug combinations in modern endocrinology. No pharmacokinetic interaction. Complementary mechanisms. Additive efficacy without additive safety concerns. Metformin's GI side effects (diarrhea) may partially offset GLP-1 agonists' GI effects (constipation) in some patients, though GI tolerability overall may be challenging during the initiation of both agents simultaneously.
With GH Secretagogues
Interaction severity: Minor to Moderate
Metformin improves insulin sensitivity. GH secretagogues worsen it. The effects partially counterbalance. In practice, most patients on metformin can tolerate standard GH secretagogue doses without losing glycemic control, though fasting glucose may drift upward by 5-15 mg/dL. A metformin dose increase or the addition of lifestyle interventions (increased exercise, carbohydrate reduction) typically addresses this.
SGLT2 Inhibitors (Empagliflozin, Dapagliflozin, Canagliflozin) + Peptide Therapy
With GLP-1 Agonists
Interaction severity: Minimal - Potentially favorable
SGLT2 inhibitors work through a completely independent mechanism - blocking glucose reabsorption in the kidneys. No pharmacokinetic interaction with GLP-1 agonists. The combination provides complementary cardiovascular and renal benefits and is increasingly used in clinical practice. Hypoglycemia risk is minimal because neither agent stimulates insulin secretion in a glucose-independent manner.
The only practical concern: combining the weight loss effects of both drug classes (both promote modest weight loss) should be monitored in patients where excessive weight loss is undesirable. Adequate protein intake and resistance exercise are particularly important with this combination.
With GH Secretagogues
Interaction severity: Minor
SGLT2 inhibitors' glucose-lowering through renal glucose excretion partially offsets GH-induced hyperglycemia. The mechanisms don't directly interfere with each other. This combination is generally well-tolerated, though the diuretic effect of SGLT2 inhibitors combined with any fluid retention from GH can create competing volume effects that patients may notice.
DPP-4 Inhibitors (Sitagliptin, Saxagliptin, Linagliptin) + Peptide Therapy
With GLP-1 Agonists
Interaction severity: Therapeutic redundancy
DPP-4 inhibitors work by preventing the breakdown of endogenous GLP-1 and GIP. Adding an exogenous GLP-1 agonist provides pharmacological levels of GLP-1 activity that dwarf the modest incretin enhancement from DPP-4 inhibition. The combination is not harmful, but the DPP-4 inhibitor adds little benefit when a GLP-1 agonist is already providing supraphysiological GLP-1 receptor activation. Most guidelines recommend discontinuing DPP-4 inhibitors when starting GLP-1 agonist therapy.
Thiazolidinediones (Pioglitazone, Rosiglitazone) + Peptide Therapy
With GLP-1 Agonists
Interaction severity: Moderate - Monitor for fluid retention
TZDs cause fluid retention and weight gain. GLP-1 agonists produce weight loss and have minimal fluid effects. The metabolic benefits may complement each other (TZDs improve insulin sensitivity, GLP-1 agonists provide glucose-dependent insulin secretion), but the fluid retention risk from TZDs is not offset by GLP-1 therapy. Heart failure risk associated with TZDs remains a concern in this combination.
With GH Secretagogues
Interaction severity: Moderate
Both GH and TZDs can cause fluid retention, creating additive edema risk. The insulin-sensitizing effect of TZDs may partially offset GH-induced insulin resistance. Monitor for peripheral edema, weight gain, and heart failure symptoms. This combination requires careful clinical oversight.
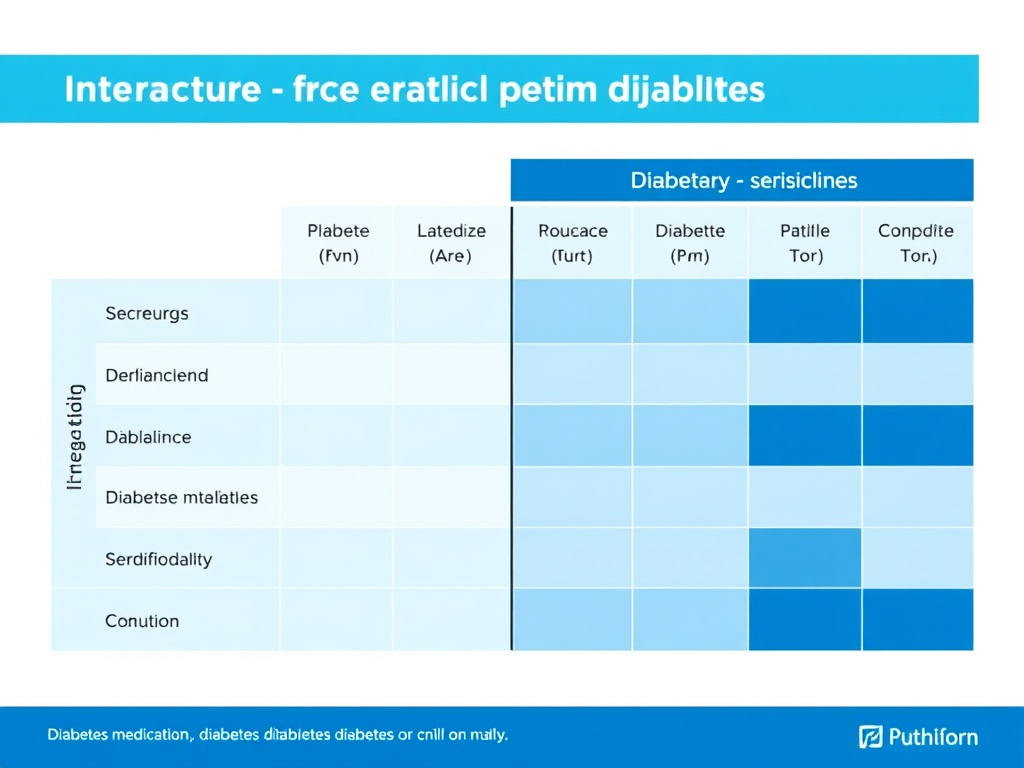
Figure 5: Interaction severity matrix for diabetes medication classes with major peptide categories. Green = minimal interaction, yellow = monitoring needed, orange = dose adjustment likely, red = use with extreme caution or avoid.
Interaction Matrix
The following comprehensive interaction matrix provides a visual reference for clinicians and patients managing peptide therapy alongside conventional medications. Each combination is rated by clinical significance based on available evidence.
Peptide-Drug Interaction Severity Distribution
Master Interaction Table: GLP-1 Receptor Agonists
| Drug Class | Specific Drugs | Severity | Mechanism | Action Required |
|---|---|---|---|---|
| Insulin | All types | Major | Additive hypoglycemia | Reduce insulin 20-25% at initiation |
| Sulfonylureas | Glipizide, glyburide, glimepiride | Major | Uncoupled glucose-dependent mechanism | Reduce SU 50%; consider discontinuation |
| Metformin | Metformin IR, XR | Minimal | No PK interaction; complementary PD | No adjustment needed |
| SGLT2 Inhibitors | Empagliflozin, dapagliflozin | Minimal | Independent mechanisms | No adjustment needed |
| DPP-4 Inhibitors | Sitagliptin, saxagliptin | Redundancy | Overlapping incretin mechanism | Discontinue DPP-4i |
| Warfarin | Warfarin | Moderate | Absorption + weight loss + diet changes | Increase INR monitoring frequency |
| Levothyroxine | Synthroid, levoxyl | Moderate | Enhanced absorption + weight-based dosing | Monitor TSH; likely dose reduction |
| Oral contraceptives | Combined OCP | Moderate* | Delayed absorption (*tirzepatide only) | Backup contraception with tirzepatide |
| Statins | Atorvastatin, rosuvastatin | Minimal | Delayed Cmax, AUC unchanged | No adjustment needed |
| ACE Inhibitors | Lisinopril, enalapril | Minimal | No significant interaction expected | No adjustment needed |
| ARBs | Losartan, valsartan | Minimal | No significant interaction expected | No adjustment needed |
| Digoxin | Digoxin | Moderate | Reduced Cmax, narrow therapeutic index | Monitor digoxin levels |
| Lithium | Lithium | Moderate | Absorption changes + dehydration risk | Monitor lithium levels; hydration |
| PPIs | Omeprazole, pantoprazole | Minimal | No direct interaction | No adjustment needed |
| NSAIDs | Ibuprofen, naproxen | Minimal | No significant interaction expected | Use with caution (GI effects) |
| Antibiotics | Amoxicillin, azithromycin | Minimal | Delayed absorption, AUC preserved | No adjustment for most; time-sensitive antibiotics may need monitoring |
Master Interaction Table: Growth Hormone Secretagogues
| Drug Class | Severity | Mechanism | Action Required |
|---|---|---|---|
| Insulin (all types) | Major | GH-induced insulin resistance | Increase insulin dose 10-30%; intensive monitoring |
| Sulfonylureas | Moderate | Opposing glucose effects | Monitor glucose; possible dose adjustment |
| Metformin | Minor | Partially offsetting insulin sensitivity effects | Monitor fasting glucose |
| Levothyroxine | Moderate | Altered T4-to-T3 conversion | Full thyroid panel monitoring |
| Hydrocortisone | Moderate | GH effects on cortisol metabolism | Monitor cortisol; stress dose review |
| Cancer medications | Contraindicated | IGF-1 proliferative effects | Do not combine without oncologist approval |
| Testosterone | Minor | Additive IGF-1 elevation | Monitor IGF-1 and metabolic markers |
| Blood pressure meds | Minimal | Mild fluid retention possible | Monitor for edema |
| Anticoagulants | Minimal | No known interaction | No adjustment needed |
| SSRIs/SNRIs | Minimal | No known interaction | No adjustment needed |
Master Interaction Table: Healing & Immune Peptides
| Peptide | Drug Class | Severity | Notes |
|---|---|---|---|
| BPC-157 | Dopaminergic drugs | Moderate | BPC-157 modulates dopamine system; monitor response |
| BPC-157 | NO pathway drugs (PDE5i) | Theoretical | Both affect NO pathways; no clinical data |
| BPC-157 | NSAIDs | Potentially beneficial | BPC-157 may provide gastric protection |
| BPC-157 | Antibiotics | Minor | GI upset may reduce BPC-157 retention |
| Thymosin Alpha-1 | Immunosuppressants | Major/Contraindicated | Directly opposes immunosuppression |
| Thymosin Alpha-1 | Checkpoint inhibitors | Moderate | Potentially additive immune activation |
| Thymosin Alpha-1 | Corticosteroids | Moderate | Opposing immune effects |
| NAD+ | Hepatotoxic drugs | Minor | NAD metabolism is liver-dependent |
| NAD+ | Chemotherapy | Moderate | NAD+ may affect DNA repair pathways |
| NAD+ | Statins | Minor | Both affect mitochondrial function |
For printable versions of these interaction tables and a personalized interaction check, visit our peptide information hub.
Healing Peptide Interactions
Healing peptides - primarily BPC-157 and TB-500 (Thymosin Beta-4) - occupy a unique position in the interaction landscape. They're widely used but dramatically under-studied, with no formal drug interaction trials in any species.
This absence of data creates a challenging clinical situation. The lack of documented interactions doesn't mean interactions don't exist - it means we haven't looked for them systematically. What we can do is analyze the known pharmacological mechanisms of these peptides and identify theoretical interaction points with common medications.
BPC-157 (Body Protection Compound-157)
BPC-157 is a 15-amino-acid peptide derived from human gastric juice with documented effects on angiogenesis, tissue repair, and several neurotransmitter systems. Its pharmacological profile touches multiple systems that could theoretically interact with medications.
Dopamine System Interactions
BPC-157 has documented interactions with the dopamine system. Research shows it can counteract the stereotypy produced by amphetamine and modulate the supersensitivity to amphetamine caused by haloperidol. This suggests BPC-157 acts as a dopamine system modulator - potentially relevant for patients on:
- Antipsychotics (haloperidol, risperidone, quetiapine): BPC-157 could theoretically alter the therapeutic effects of these dopamine-blocking medications. The direction and magnitude of any clinical effect remain unknown.
- Dopamine agonists (pramipexole, ropinirole, carbidopa-levodopa): BPC-157's dopamine-modulating effects could potentially enhance or oppose these medications. Parkinson's patients on levodopa should be particularly cautious.
- Stimulant medications (amphetamine, methylphenidate): Given BPC-157's demonstrated interaction with amphetamine pharmacology in animal models, patients on ADHD stimulants should be aware of potential altered responses (DOI: 10.1016/S0024-3205(97)00535-3).
Nitric Oxide Pathway Interactions
BPC-157 modulates the nitric oxide (NO) system, which has implications for several medication classes:
- PDE5 inhibitors (sildenafil, tadalafil): Both BPC-157 and PDE5 inhibitors enhance NO-mediated vasodilation. Theoretical additive effect could lower blood pressure more than expected. No clinical reports of adverse events, but awareness is warranted.
- Nitrates (nitroglycerin, isosorbide): Similar NO pathway overlap. Patients on nitrate therapy should exercise caution, though clinical interaction data doesn't exist.
- Blood pressure medications: BPC-157's vasodilatory effects through NO modulation could theoretically potentiate antihypertensive effects. Monitor blood pressure when initiating BPC-157 in patients on aggressive BP-lowering regimens.
Gastrointestinal Medications
BPC-157 originates from gastric juice and has documented gastroprotective effects. This creates an interesting interaction profile with GI medications:
- NSAIDs: BPC-157 may actually provide protection against NSAID-induced gastric damage, based on animal studies. This could be a beneficial interaction, though it should not be relied upon as a gastroprotective strategy without clinical trial confirmation.
- Proton pump inhibitors: No documented interaction. BPC-157's gastroprotective mechanism doesn't involve acid suppression, so it works through a different pathway than PPIs.
- Antibiotics: Some sources suggest BPC-157 may interact with tetracycline and amoxicillin, primarily through GI upset that could affect BPC-157 retention if taken orally. For injectable BPC-157, this concern is moot.
Serotonin System Interactions
BPC-157 has demonstrated effects on the serotonergic system in animal models. For patients on SSRIs, SNRIs, or other serotonergic medications, this represents a theoretical interaction point. While serotonin syndrome from BPC-157 combined with SSRIs has never been reported, the absence of data doesn't confirm safety. Patients on serotonergic medications who start BPC-157 should monitor for signs of serotonin excess: agitation, rapid heart rate, elevated temperature, and muscle rigidity.
TB-500 (Thymosin Beta-4 Fragment)
TB-500 is a synthetic fragment of thymosin beta-4, a naturally occurring protein involved in cell migration, blood vessel formation, and wound healing. Its interaction profile is even less characterized than BPC-157's, but several theoretical considerations apply:
- Anticoagulants and antiplatelets: TB-500 promotes angiogenesis and may affect blood vessel formation. While not a direct anticoagulant interaction, enhanced angiogenesis could theoretically affect wound healing and bleeding patterns in patients on blood thinners.
- Immunomodulatory drugs: TB-500 has anti-inflammatory properties. In patients on immune-suppressing medications, the anti-inflammatory effect could be additive. In patients on immune-stimulating therapy, the interaction is less predictable.
- Cancer therapy: Like GH secretagogues, any peptide that promotes cell proliferation, angiogenesis, or tissue growth raises theoretical concerns in cancer patients. TB-500 should not be used during active cancer treatment without explicit oncologist clearance.
BPC-157 + TB-500 Combination
The "Wolverine Stack" - combining BPC-157 and TB-500 for accelerated healing - is among the most popular peptide combinations. Research has not identified antagonistic interactions between the two peptides. They operate through different receptor systems and complementary mechanisms: BPC-157 provides localized tissue repair and angiogenesis while TB-500 promotes systemic anti-inflammatory and cell migration responses.
When assessing drug interactions for patients on the BPC-157/TB-500 combination, consider the additive effects on:
- Angiogenesis (relevant for patients with proliferative conditions)
- Anti-inflammatory activity (relevant for patients on immunosuppressive or anti-inflammatory medications)
- Blood vessel formation (relevant for patients on anticoagulants during surgical recovery)
Regulatory Context
BPC-157 was classified as a Category 2 bulk drug substance by the FDA in 2023, meaning it cannot be compounded by commercial pharmacies. WADA has banned BPC-157 under the S0 Unapproved Substances category. These regulatory decisions reflect the lack of human clinical trial data rather than confirmed safety concerns. However, they underscore the importance of medical supervision when using these compounds alongside prescription medications.
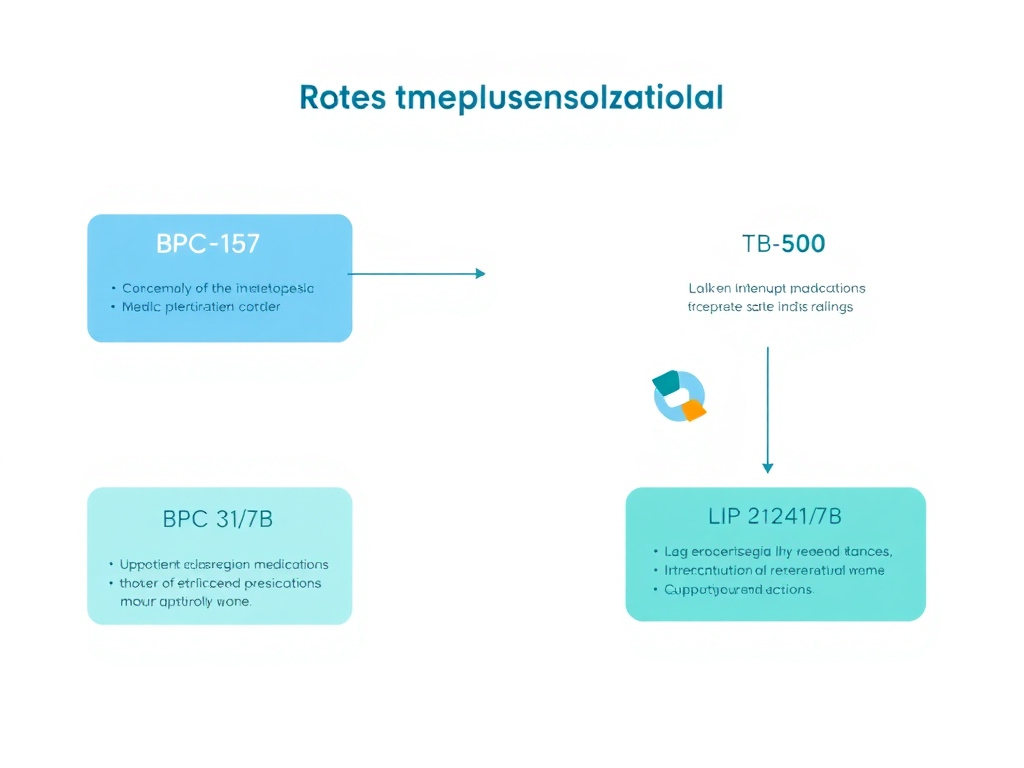
Figure 6: BPC-157 and TB-500 interaction pathways with common medication classes. Solid lines indicate documented pharmacological effects; dashed lines indicate theoretical interactions based on mechanism of action analysis.
Immune Peptide Interactions
Thymosin Alpha-1 (Ta1) stands apart from other therapeutic peptides in its interaction profile. As a true immunomodulator, it directly affects the immune system in ways that can profoundly interact with immunosuppressive, immunostimulatory, and anti-inflammatory medications.
Thymosin Alpha-1: Mechanism and Interaction Basis
Ta1 is a 28-amino-acid peptide that potentiates T cell-mediated immune responses through several pathways: differentiation and maturation of T-cell progenitor cells, activation of dendritic cells and natural killer cells, and stimulation of cytokine-mediated inflammation. It's used clinically in several countries for hepatitis B and C treatment, as an immune adjuvant in cancer care, and for immune restoration in immunocompromised patients (DOI: 10.4254/wjh.v12.i5.67).
This broad immune-stimulating profile creates the most clinically consequential interaction among all therapeutic peptides: the conflict with immunosuppressive therapy.
Immunosuppressant Interactions - Critical Safety Concern
Severity: Contraindicated in most cases
Thymosin Alpha-1 is contraindicated in immunosuppressed patients - particularly organ transplant recipients - unless the clinical benefits clearly outweigh the risks. The mechanism is straightforward and dangerous: Ta1 activates the very immune cells that immunosuppressive drugs are trying to suppress.
Specific immunosuppressant interactions:
| Immunosuppressant | Interaction Risk | Clinical Concern |
|---|---|---|
| Cyclosporine | Contraindicated | Ta1 may overcome cyclosporine-mediated T-cell suppression, risking graft rejection |
| Tacrolimus (FK506) | Contraindicated | Same mechanism as cyclosporine; risk of acute rejection |
| Mycophenolate | Contraindicated | Ta1 stimulates lymphocyte proliferation that mycophenolate inhibits |
| Sirolimus/Everolimus | Contraindicated | Opposing effects on T-cell activation and proliferation |
| Azathioprine | Contraindicated | Immune activation opposes immunosuppressive intent |
| Prednisone (high dose) | Major | High-dose steroids for immune suppression are partially opposed by Ta1 |
| Prednisone (low dose) | Moderate | Low-dose anti-inflammatory steroids are less affected |
Organ Transplant Patients
Thymosin Alpha-1 should NEVER be used by organ transplant recipients without explicit approval from the transplant team. The risk of graft rejection is real and potentially life-threatening. There are no circumstances where the potential benefits of Ta1 outweigh the risk of losing a transplanted organ. Even if a transplant patient has a concurrent infection or malignancy that might otherwise benefit from immune stimulation, the transplant team must weigh this decision within the context of the full immunosuppressive regimen.
Autoimmune Disease Medications
The interaction between Ta1 and autoimmune disease treatments is nuanced. Ta1 is often described as an immune "modulator" rather than simply an immune "stimulant." Some research suggests it may help rebalance dysregulated immune responses rather than uniformly boost all immune activity. However, this theoretical distinction hasn't been validated in formal clinical studies with autoimmune patients on immunosuppressive therapy.
For patients on biologic DMARDs (disease-modifying antirheumatic drugs):
- TNF inhibitors (adalimumab, infliximab, etanercept): Ta1 may partially oppose TNF suppression through its cytokine-stimulating effects. The interaction is unpredictable. Avoid combination without rheumatologist approval.
- IL-6 inhibitors (tocilizumab): Similar concern - Ta1's cytokine-stimulating properties may partially counteract targeted IL-6 blockade.
- JAK inhibitors (tofacitinib, baricitinib): These suppress multiple cytokine signaling pathways. Ta1's immune stimulation broadly opposes this mechanism.
- B-cell depleting agents (rituximab): These target a different arm of the immune system (B-cells) than Ta1's primary T-cell effects. The interaction is less direct but still warrants caution.
Cancer Immunotherapy Interactions
Ta1 is actually used as an adjunctive therapy alongside certain cancer treatments in some countries. The interaction with checkpoint inhibitors (pembrolizumab, nivolumab, atezolizumab) is particularly interesting - both Ta1 and checkpoint inhibitors aim to enhance anti-tumor immune responses. A clinical trial (NCT06821100) has evaluated thymalfasin as an enhancer of cancer immunotherapy, suggesting that careful combination under oncological supervision may be feasible.
However, the risk of this combination is autoimmune toxicity. Checkpoint inhibitors already produce immune-related adverse events (colitis, hepatitis, pneumonitis, endocrinopathies) in 20-60% of patients. Adding Ta1's immune-stimulating effects could potentially amplify these toxicities. Any use of Ta1 alongside checkpoint inhibitors must be under close oncological supervision with frequent monitoring for autoimmune complications.
Antiviral Medications
Ta1 has been most extensively studied in combination with antiviral therapies, particularly for hepatitis B and C. These combinations are generally well-tolerated and may even be complementary:
- Interferon-alpha: Ta1 has been combined with interferon for hepatitis treatment with evidence of improved sustained virological response rates and no additional safety signals.
- Nucleos(t)ide analogs (entecavir, tenofovir): No pharmacokinetic interaction. The combination provides complementary mechanisms - direct antiviral action plus enhanced immune clearance.
- Direct-acting antivirals (sofosbuvir, ledipasvir): No documented interaction, though formal studies are limited.
Vaccines
Ta1 may enhance vaccine responses through its immune-stimulating properties. This is a potentially beneficial interaction for immunocompromised patients who respond poorly to vaccination. Studies have shown improved antibody responses to influenza and hepatitis B vaccines when combined with Ta1. However, this same property means that live vaccines could potentially produce more strong - and potentially more symptomatic - responses. Patients on Ta1 should receive live vaccines only with medical guidance.
Other Immune-Modulating Peptides
KPV (Alpha-MSH Fragment)
KPV is a tripeptide fragment of alpha-melanocyte-stimulating hormone with anti-inflammatory properties. Unlike Ta1, KPV tends to reduce inflammatory responses. Drug interaction data is essentially non-existent, but its anti-inflammatory mechanism suggests potential additive effects with NSAIDs and possible partial opposition to pro-inflammatory therapies. The risk profile appears lower than Ta1 due to its anti-inflammatory rather than immune-stimulating nature.
LL-37 (Cathelicidin)
LL-37 is an antimicrobial peptide with immunomodulatory properties. It interacts with the innate immune system rather than the adaptive immune system, making its interaction profile different from Ta1. No drug interaction data exists, but its mechanism suggests minimal concern for interactions with most conventional medications.
Supplement Interactions With Peptide Therapy
Many peptide therapy users also take dietary supplements - and some of these combinations create interactions that are often overlooked in clinical discussions. While supplement-peptide interactions are generally lower risk than drug-peptide interactions, several warrant attention.
NAD+ Precursors (NMN, NR, Niacin)
NAD+ and its precursors (nicotinamide mononucleotide, nicotinamide riboside, niacinamide) are increasingly used alongside peptide therapies for longevity and cellular health protocols. Key interaction considerations:
- With GH secretagogues: Both NAD+ precursors and GH peptides target cellular energy metabolism. The combination is theoretically complementary (NAD+ supports mitochondrial function while GH promotes cellular repair and growth), but no formal interaction studies exist. Monitor liver function, as both pathways involve hepatic metabolism (DOI: 10.3390/antiox10091637).
- With cancer medications: NAD+ affects DNA repair pathways - a double-edged sword in cancer. Some cancer treatments work by overwhelming DNA repair mechanisms. NAD+ supplementation could theoretically counteract this. Do not use NAD+ precursors during cancer treatment without oncologist approval.
- With hepatotoxic medications: The liver is central to NAD+ metabolism. Combining NAD+ precursors with hepatotoxic drugs (acetaminophen at high doses, certain statins, methotrexate) warrants liver enzyme monitoring.
- With blood thinners: Niacin (vitamin B3) at high doses can potentiate anticoagulant effects. NMN and NR have not shown this effect, but patients on warfarin should monitor INR if starting any NAD+ pathway supplement.
Vitamin D
Vitamin D supplementation is extremely common among peptide therapy users. Relevant interactions:
- With GLP-1 agonists: Vitamin D is fat-soluble and absorbed in the small intestine. GLP-1-induced gastric emptying delay may slightly alter absorption kinetics, but since vitamin D is typically taken daily and has a very long half-life, this effect is clinically insignificant.
- With GH secretagogues: GH and vitamin D interact in calcium metabolism. GH increases calcium absorption and IGF-1 stimulates renal 1-alpha-hydroxylase, enhancing active vitamin D production. Patients on high-dose vitamin D who start GH secretagogues should monitor calcium levels to avoid hypercalcemia.
Zinc and Magnesium
These minerals are frequently recommended alongside peptide therapy:
- Zinc with GH secretagogues: Zinc is a cofactor for GH receptor signaling and may enhance the effectiveness of GH secretagogues. This is a potentially favorable interaction, though excessive zinc supplementation (>40 mg/day) can cause copper deficiency, which in turn affects immune function and iron metabolism.
- Magnesium with GLP-1 agonists: GLP-1 agonists can cause GI symptoms that may reduce magnesium absorption. Monitor magnesium levels, particularly in patients also taking PPIs (which independently reduce magnesium absorption). Low magnesium can cause muscle cramps, fatigue, and cardiac arrhythmias.
Berberine
Berberine is a popular supplement with glucose-lowering properties that mimic some aspects of metformin. When combined with GLP-1 agonists, berberine adds another glucose-lowering mechanism that could potentially cause hypoglycemia, though the risk is lower than with sulfonylureas. Patients on berberine who start GLP-1 therapy should monitor fasting glucose and consider reducing or discontinuing berberine if glucose drops below target.
With GH secretagogues, berberine's glucose-lowering effect partially counterbalances GH-induced insulin resistance - a potentially useful interaction, though it has not been formally studied.
Omega-3 Fatty Acids
High-dose omega-3 supplements (prescription or OTC) have mild blood-thinning properties. When combined with peptide therapies that affect coagulation pathways or blood vessel formation (TB-500, BPC-157's angiogenic effects), there's a theoretical concern for increased bleeding tendency. The risk is probably very low in practice, but patients on prescription anticoagulants who also take high-dose omega-3s and angiogenic peptides should be aware of this additive effect.
Creatine
Creatine is commonly used alongside GH secretagogues and GLP-1 agonists (for muscle preservation during weight loss). No pharmacological interaction exists, but creatine supplementation can increase serum creatinine levels, potentially confusing kidney function markers. This is a laboratory interpretation issue rather than a true interaction, but it's helpful to know for patients undergoing blood work monitoring.
Herbal Supplements Requiring Caution
| Supplement | Peptide Class of Concern | Interaction Concern |
|---|---|---|
| St. John's Wort | All peptides | CYP inducer - may alter metabolism of co-administered drugs, complicating peptide-drug interaction assessment |
| Ginkgo Biloba | Healing peptides | Blood-thinning effects may be additive with angiogenic peptide properties |
| Ashwagandha | GH secretagogues | May enhance thyroid function - additive with GH effects on T4/T3 conversion |
| Turmeric/Curcumin | Healing peptides | Anti-inflammatory effects may be additive; affects blood clotting at high doses |
| Milk Thistle | NAD+ peptides | Hepatoprotective effects may alter liver metabolism of co-administered compounds |
| Fenugreek | GLP-1 agonists | Glucose-lowering effect may be additive |
Peptide-Peptide Interactions
As peptide therapy becomes more sophisticated, clinicians and patients are increasingly combining multiple peptides in single protocols - so-called "stacking." Understanding how peptides interact with each other is as important as understanding how they interact with conventional drugs.
Common Peptide Stacks and Their Interaction Profiles
GLP-1 Agonist + GH Secretagogue
This is one of the most common combination protocols, used to achieve simultaneous body composition optimization (fat loss from the GLP-1 agonist, muscle preservation/gain from the GH secretagogue). The interaction profile is largely favorable with one major caveat:
- Favorable: GLP-1 agonists improve insulin sensitivity and lower glucose. GH secretagogues worsen insulin sensitivity and raise glucose. The opposing effects partially neutralize each other's metabolic side effects.
- Favorable: GLP-1-mediated weight loss tends to reduce GH resistance (obesity blunts GH secretion), potentially enhancing the secretagogue's effectiveness as body composition improves.
- Caution: The net effect on glucose depends on dose ratios and individual metabolic status. Some patients achieve metabolic equilibrium easily. Others - particularly those with pre-diabetes or insulin resistance - may experience unpredictable glucose fluctuations.
Monitoring protocol: Check fasting glucose and HbA1c at baseline, 4 weeks, and 12 weeks after combining. Adjust doses based on metabolic response.
BPC-157 + TB-500 ("Wolverine Stack")
This popular healing combination pairs BPC-157's localized tissue repair with TB-500's systemic anti-inflammatory and cell migration properties. No documented antagonistic interactions between the two peptides have been identified. They operate through different receptor systems - BPC-157 activates growth factor pathways and nitric oxide systems while TB-500 promotes actin regulation and blood vessel formation.
Key considerations for this stack:
- Additive angiogenic effects - beneficial for healing but potentially concerning in patients with active neoplasia
- Combined anti-inflammatory activity - may reduce the need for NSAID or corticosteroid use during recovery
- Both peptides should be discontinued before elective surgery (discuss timing with your surgeon)
- Do not combine these peptides in a single injection vial - administer separately
CJC-1295/Ipamorelin + BPC-157
This combination aims to leverage GH-mediated systemic recovery alongside BPC-157's targeted tissue repair. The interaction is generally considered favorable - GH supports overall tissue repair while BPC-157 provides localized healing. No antagonistic interactions have been documented.
The main concern: both peptides promote angiogenesis through different mechanisms. In most contexts, this is beneficial for healing. But for patients with any history of proliferative conditions, this combination should be used with medical supervision.
GLP-1 Agonist + Thymosin Alpha-1
This combination is sometimes used in metabolic-immune optimization protocols. No direct pharmacokinetic interaction exists between these peptide classes - they work through entirely different receptor systems. The main consideration is that GLP-1 agonists' anti-inflammatory effects in adipose tissue may complement Ta1's immune-modulating properties. No safety concerns have been identified specific to this combination, beyond those applicable to each agent individually.
Multiple GH Secretagogues
Combining multiple GH-releasing peptides (e.g., CJC-1295 with both ipamorelin and GHRP-6) produces additive GH release that may exceed physiological bounds. This creates amplified risks of:
- Insulin resistance and glucose dysregulation
- Fluid retention and joint discomfort
- Excessive IGF-1 elevation
- Carpal tunnel syndrome symptoms
If multiple GH secretagogues are used simultaneously, IGF-1 levels should be monitored to ensure they remain within the upper normal range (roughly 200-300 ng/mL for most adults). Supraphysiological IGF-1 levels increase the risk of adverse effects without proportional benefit.
GLP-1 Agonist + NAD+
Combining semaglutide or tirzepatide with NAD+ supplementation is increasingly popular in longevity protocols. No pharmacological interaction has been identified. GLP-1 agonists work through receptor-mediated signaling while NAD+ supports cellular energy metabolism. The combination is generally considered safe, though clinical data on combined use is limited.
General Principles for Peptide Stacking
- Start one at a time. Begin each new peptide individually, waiting at least 2-4 weeks to establish baseline response before adding another. This allows you to identify side effects and attribute them to specific peptides.
- Monitor metabolic markers. The more peptides you're combining, the more important regular blood work becomes. At minimum, track glucose, IGF-1, thyroid function, and liver enzymes.
- Don't mix in the same syringe unless using a commercially prepared blend from a verified source. Peptide stability can be compromised by incompatible pH, osmolarity, or chemical interactions.
- Document your protocol. Keep detailed records of doses, timing, injection sites, and any side effects. This information is invaluable for your healthcare provider and for troubleshooting if issues arise.
- Share your full protocol with your doctor. Many patients use peptides without fully disclosing to their primary care provider. This creates blind spots in medication management that can lead to avoidable interactions and complications.

Figure 7: Common peptide stacking combinations with interaction assessments. Green pairings have no documented antagonistic interactions; yellow pairings require monitoring; red pairings warrant medical supervision.
Anesthesia and Perioperative Considerations
If you're on peptide therapy and facing surgery, your anesthesiologist needs to know. The perioperative management of peptide therapies - particularly GLP-1 receptor agonists - has become a major topic in anesthesiology and surgical safety.
GLP-1 Agonists and Aspiration Risk
The primary perioperative concern with GLP-1 agonists is aspiration risk. These drugs delay gastric emptying, meaning patients may have retained stomach contents even after standard preoperative fasting. Under general anesthesia, retained gastric contents can reflux into the airway, causing aspiration pneumonitis - a potentially life-threatening complication.
The American Society of Anesthesiologists (ASA), in collaboration with the American Gastroenterological Association and the American Society for Metabolic and Bariatric Surgery, released multi-society guidance in 2024 that took a more nuanced approach than the original 2023 recommendations. The updated guidance states that most patients can continue their GLP-1 medications before elective surgery, with risk stratification determining which patients need additional precautions.
Risk Stratification for Perioperative GLP-1 Management
| Risk Level | Patient Characteristics | Recommended Management |
|---|---|---|
| Lower Risk | Stable dose for >4 weeks; no GI symptoms; no gastroparesis history | Continue GLP-1; standard fasting protocol |
| Moderate Risk | Active dose escalation; mild GI symptoms | Continue GLP-1; liquid diet 24 hours pre-op; consider gastric ultrasound |
| Higher Risk | Significant GI symptoms; gastroparesis; Parkinson's disease; other motility disorders | Consider holding GLP-1; liquid diet 24 hours pre-op; mandatory gastric ultrasound; modified anesthesia plan |
Specific Timing Recommendations
A 2025 study recommending stopping GLP-1 agonists 14 days before total joint arthroplasty found this timeframe optimal for reducing anesthesia-related complications including delayed emergence, aspiration events, aspiration pneumonitis, and conversion to intubation.
For weekly agents (semaglutide, tirzepatide):
- Conservative approach: Hold the last dose before surgery, scheduling the procedure for the end of the dosing interval (day 6-7 for weekly agents)
- More conservative approach: Skip the dose one full week before surgery, creating a 14-day washout
- If surgery is urgent: Proceed with modified anesthesia protocol - rapid sequence intubation, point-of-care gastric ultrasound, and preparation for aspiration management
For daily agents (liraglutide):
- Hold the dose on the day of surgery
- Consider holding 24-48 hours pre-operatively for higher-risk patients
GH Secretagogues and Surgery
Growth hormone secretagogues present different perioperative considerations:
- Glucose management: GH-induced insulin resistance may complicate perioperative glucose control. Hold GH secretagogues for 3-5 days before surgery to reduce this risk.
- Fluid retention: GH-related fluid retention can affect fluid balance management during and after surgery. Stopping secretagogues 5-7 days pre-operatively allows fluid shifts to normalize.
- Wound healing: While GH generally supports healing, the perioperative period requires careful glucose control, which takes priority. GH secretagogues can typically be restarted 1-2 weeks post-operatively once glucose control is stable and the surgical team approves.
Healing Peptides and Surgery
BPC-157 and TB-500 are often used specifically to enhance surgical recovery. However, their angiogenic and tissue-modifying properties create valid perioperative questions:
- Pre-operative: Consider holding BPC-157 and TB-500 for 5-7 days before surgery. Their angiogenic properties could theoretically affect intraoperative bleeding or tissue behavior, though clinical data is absent.
- Post-operative: These peptides are frequently started or restarted 3-7 days after surgery (once initial hemostasis is established) specifically to promote healing. Discuss timing with your surgeon.
- Concurrent anticoagulation: If you're on perioperative anticoagulation (DVT prophylaxis), the additive effects of angiogenic peptides and blood thinners should be considered. Inform your surgical team about all peptide use.
Immune Peptides and Surgery
Thymosin Alpha-1's immune-stimulating effects are generally beneficial in the perioperative setting (reducing infection risk), but should be coordinated with the surgical team. If the patient is receiving perioperative immunosuppression (such as steroids for transplant surgery or autoimmune conditions), Ta1 is contraindicated as discussed in the immune peptide section.
Communication Protocol for Surgery
Every peptide therapy user facing surgery should:
- Inform the surgical team about ALL peptide therapies at least 2-4 weeks before scheduled surgery
- Provide a complete medication list including peptides, even if they're not prescription drugs - the anesthesiologist and surgeon need this information
- Follow specific hold/continue instructions from your anesthesiologist, who will factor in your specific peptides, surgery type, and medical history
- Ask about resumption timing before discharge - know when you can safely restart each peptide post-operatively
- For emergency surgery: tell the emergency team about all peptide use immediately, even if you feel it's not relevant. The anesthesiologist will determine relevance.
Pre-Surgical Checklist for Peptide Users
- Notify your surgeon and anesthesiologist of all peptide therapies 2-4 weeks pre-op
- Follow agent-specific hold schedules (GLP-1: 7-14 days; GH secretagogues: 3-7 days; healing peptides: 5-7 days)
- Follow pre-operative fasting instructions precisely - if on GLP-1, consider a liquid diet for 24 hours
- Bring your complete peptide protocol to the pre-op appointment
- Ask about post-operative resumption timing for each peptide

Figure 8: Perioperative peptide management timeline. Recommended hold periods before surgery and restart timing after surgery for each major peptide class.
How to Discuss Peptide Therapy With Your Doctor
Many patients using peptide therapies don't fully disclose their protocols to their primary care physician, specialist, or surgeon. This creates dangerous knowledge gaps that can lead to preventable drug interactions and complications.
The reasons for non-disclosure are understandable: fear of judgment, concerns that the doctor won't understand peptide therapy, or the assumption that supplements and non-prescription compounds "don't count" as medications. But from a drug interaction standpoint, everything you put in your body counts.
Why Full Disclosure Matters
Consider this scenario: a patient on warfarin doesn't tell their cardiologist they're taking semaglutide from a peptide clinic. They lose 30 pounds over 6 months. Their INR starts fluctuating. The cardiologist adjusts warfarin doses without understanding why the changes are happening. The patient is at increased risk for either bleeding (supratherapeutic INR) or clotting (subtherapeutic INR) because the root cause - the GLP-1-mediated weight loss and dietary changes - isn't being factored into management.
Or this: a patient on tacrolimus after a kidney transplant starts thymosin alpha-1 without telling the transplant team, based on internet research about immune support. The immunostimulatory effects of Ta1 could trigger acute rejection of the transplanted kidney - a life-threatening emergency.
These aren't hypothetical. Drug interactions from undisclosed medications are a leading cause of preventable adverse events in healthcare.
How to Have the Conversation
Here's a practical framework for discussing peptide therapy with your healthcare providers:
1. Frame It as a Safety Discussion
"I want to make sure all my medications are safe together. I've been using/considering some peptide therapies and want your input on potential interactions with my current prescriptions."
2. Bring Documentation
Prepare a written list that includes:
- Each peptide name and dose
- Route of administration (subcutaneous injection, oral, etc.)
- Frequency and timing
- Duration of use
- Source (prescribing clinic, compounding pharmacy, research supplier)
3. Provide Context
"These peptides work through [brief mechanism]. The main interaction concern I'm aware of is [specific concern related to their medications]. I'd like your guidance on monitoring and dose adjustments."
4. Ask Specific Questions
- "Does this peptide interact with any of my current medications?"
- "Should I adjust the timing of any of my medications?"
- "Do I need additional blood work or monitoring?"
- "If I need surgery, how far in advance should I stop this peptide?"
If Your Doctor Is Unfamiliar With Peptide Therapy
Many physicians haven't received training in therapeutic peptides beyond GLP-1 agonists. If your doctor is unfamiliar with a specific peptide, offer to share resources (like this report) and suggest they consult with a colleague who has peptide therapy experience. Some patients find it helpful to identify a peptide-knowledgeable provider for protocol management while keeping their primary care physician informed for overall medication coordination.
The key message: your primary care physician may not need to manage your peptide therapy directly, but they absolutely need to know about it. This knowledge allows them to interpret blood work correctly, anticipate drug interactions, adjust existing medications as needed, and manage your care safely across the full spectrum of your health needs.
For finding peptide-knowledgeable practitioners, visit our getting started page or explore our beginner's guide to peptide therapy.
Alcohol, Cannabis, and Recreational Substance Interactions With Peptide Therapy
Patients rarely volunteer information about alcohol, cannabis, or recreational substance use to their healthcare providers, yet these substances interact with peptide therapies in clinically meaningful ways. This section addresses the practical reality that many peptide users consume these substances and need evidence-based guidance.
Alcohol and Peptide Therapy
Alcohol + GLP-1 Agonists
Alcohol and GLP-1 agonists have a complex relationship that extends beyond simple drug interaction pharmacology. GLP-1 agonists reduce alcohol cravings and consumption in a significant subset of users - a pharmacodynamic effect that's become the subject of active clinical investigation. Several large trials are underway evaluating semaglutide for alcohol use disorder.
From a drug interaction standpoint, several considerations apply:
- Hypoglycemia risk: Alcohol inhibits hepatic gluconeogenesis. GLP-1 agonists lower blood glucose through multiple mechanisms. The combination - particularly on an empty stomach or after heavy drinking - can produce significant hypoglycemia. Patients on GLP-1 agonists should avoid drinking on an empty stomach and monitor glucose after alcohol consumption.
- GI tolerability: Both alcohol and GLP-1 agonists irritate the GI tract. Combining them, especially during the dose-escalation phase when GI side effects are most pronounced, can intensify nausea and vomiting. Persistent vomiting raises dehydration risk and can prevent absorption of oral medications.
- Pancreatitis risk: Both alcohol and GLP-1 agonists have been associated with pancreatitis, though through different mechanisms. Heavy alcohol use combined with GLP-1 therapy could theoretically increase pancreatitis risk above either factor alone. Patients with a history of alcohol-related pancreatitis should use GLP-1 agonists with extra caution.
- Hepatic effects: Moderate alcohol use combined with GLP-1 therapy (which has shown liver benefits in NAFLD) creates opposing hepatic signals. The net effect depends on the amount and pattern of alcohol consumption.
Practical guidance: moderate alcohol consumption (1-2 drinks, not daily) is generally compatible with GLP-1 therapy. Heavy drinking is strongly discouraged for both interaction and general health reasons. For more on the relationship between GLP-1 agonists and alcohol, see our research library on this topic.
Alcohol + GH Secretagogues
Growth hormone secretion is suppressed by alcohol consumption. Drinking within 3 hours of GH secretagogue administration may blunt the GH release response, reducing the peptide's therapeutic effect. Additionally:
- Alcohol worsens insulin sensitivity, compounding GH-induced insulin resistance
- Chronic heavy drinking impairs liver function, which can reduce IGF-1 production and alter the metabolic response to GH stimulation
- Alcohol disrupts sleep architecture, reducing the natural nocturnal GH pulse that secretagogues are designed to amplify
Recommendation: avoid alcohol for at least 3 hours before and after GH secretagogue administration. Moderate consumption at other times is unlikely to create safety concerns but may reduce peptide effectiveness.
Alcohol + Healing Peptides
Alcohol impairs wound healing through multiple mechanisms - reduced immune function, impaired collagen synthesis, increased oxidative stress. Using healing peptides like BPC-157 and TB-500 while consuming alcohol creates opposing physiological signals. The alcohol undermines the tissue repair the peptides are promoting. While this isn't a dangerous drug interaction per se, it does represent a significant reduction in therapeutic value.
Alcohol + NAD+ Precursors
Alcohol metabolism is NAD+-dependent. Alcohol dehydrogenase and aldehyde dehydrogenase both require NAD+ as a cofactor. Heavy alcohol consumption depletes NAD+ stores, which is part of alcohol's hepatotoxic mechanism. Taking NAD+ precursors while drinking could theoretically replenish NAD+ faster and support alcohol metabolism, but it could also encourage continued drinking by reducing the physiological consequences. This is not a medical recommendation for continuing alcohol use.
From an interaction safety standpoint, the liver handles both alcohol metabolism and NAD+ precursor metabolism. Combining them increases hepatic workload. Liver enzyme monitoring is advisable for anyone using NAD+ precursors who also drinks regularly.
Cannabis and Peptide Therapy
Cannabis use is increasingly common and legal, yet formal interaction studies with peptide therapies don't exist. Considerations based on pharmacological analysis:
Cannabis + GLP-1 Agonists
- Appetite effects: Cannabis typically stimulates appetite (the "munchies"). GLP-1 agonists suppress appetite. These opposing effects may partially negate each other's actions on food intake. Some patients report that cannabis overrides GLP-1-mediated appetite suppression, potentially reducing weight loss effectiveness.
- Antiemetic effects: Cannabis has antiemetic properties. Patients experiencing GLP-1-induced nausea sometimes use cannabis for symptom relief. While this may be effective symptomatically, it can also increase caloric intake (counteracting weight loss goals) and may mask GI symptoms that indicate a need for dose adjustment.
- Glucose effects: Cannabis has complex effects on glucose metabolism. Chronic use has been associated with both improved and worsened glucose control in different studies. The net effect when combined with GLP-1 agonists is unpredictable.
Cannabis + GH Secretagogues
THC may affect growth hormone secretion, though study results are inconsistent. CBD does not appear to significantly affect GH levels. The metabolic interaction concern is minimal, but patients using cannabis for sleep (which is when most GH secretagogue protocols are timed) should be aware that cannabis can suppress REM sleep, potentially affecting the sleep-associated GH pulse that secretagogues target.
Cannabis + Healing Peptides
Both cannabinoids and healing peptides affect inflammatory pathways. CBD in particular has anti-inflammatory properties that could be additive with BPC-157 and TB-500. This is a potentially complementary combination, though clinical validation is absent. THC's analgesic effects may complement the pain-reducing aspects of healing peptide therapy.
Caffeine and Peptide Therapy
While not a recreational substance per se, caffeine deserves mention given its near-universal use:
- With GLP-1 agonists: Caffeine on an empty stomach combined with GLP-1-induced GI sensitivity can worsen nausea and acid reflux. Taking coffee with a small amount of food is advisable during GLP-1 therapy.
- With GH secretagogues: Caffeine may enhance GH release in some contexts, potentially creating a modestly favorable interaction. However, caffeine's cortisol-stimulating effects and its impact on sleep could partially offset GH secretagogue benefits.
- With NAD+: No direct interaction. Both caffeine and NAD+ precursors support cellular energy metabolism through different pathways.
Nicotine and Peptide Therapy
Nicotine (from cigarettes, vaping, patches, or gum) creates several peptide interaction considerations:
- CYP enzyme effects: Smoking (not nicotine itself, but the polycyclic aromatic hydrocarbons in smoke) induces CYP1A2. This can alter the metabolism of co-administered drugs in ways that interact with peptide effects. What matters here is that switching from smoking to vaping (which doesn't contain PAHs) removes this CYP1A2 induction, potentially changing drug levels.
- Cardiovascular effects: Nicotine is vasoconstrictive and increases heart rate. Combining with GH secretagogues (which can cause fluid retention) or BPC-157 (which modulates NO-mediated vasodilation) creates competing cardiovascular signals.
- Weight effects: Nicotine suppresses appetite. Patients who quit smoking while on GLP-1 therapy may find the GLP-1 agonist helpful for preventing smoking cessation-related weight gain - a potentially beneficial application that's receiving clinical attention.
- Healing impairment: Nicotine significantly impairs wound healing and microcirculation. Using healing peptides (BPC-157, TB-500) while actively smoking or vaping reduces their therapeutic potential substantially.
Future Directions and Emerging Interaction Concerns
The peptide therapy field is expanding rapidly, with new compounds entering clinical use and existing compounds being applied in novel ways. Understanding emerging interaction concerns helps clinicians and patients prepare for the near future of combination peptide-drug therapy.
Next-Generation GLP-1 Compounds
Several new GLP-1-related compounds are in late-stage development or recently approved, each bringing unique interaction considerations:
Retatrutide (Triple Agonist)
Retatrutide activates GLP-1, GIP, and glucagon receptors - a triple mechanism that may produce even more pronounced gastric emptying delays and more dramatic weight loss than current dual agonists. The glucagon receptor activation adds a new pharmacodynamic dimension: glucagon raises blood glucose, partially counteracting the glucose-lowering effects of GLP-1 activation. This three-way metabolic tug-of-war creates a more complex interaction profile with diabetes medications. Drug interaction studies are ongoing, but clinicians should anticipate that the principles established for semaglutide and tirzepatide will apply, possibly with greater magnitude.
Orforglipron (Oral Non-Peptide GLP-1)
Orforglipron is a small molecule (not a peptide) that activates GLP-1 receptors orally without the absorption enhancer required by oral semaglutide. Its CYP metabolism profile differs from peptide GLP-1 agonists, potentially introducing traditional CYP-mediated drug interactions that haven't been relevant for injectable GLP-1 peptides. This represents a shift in the interaction paradigm - the first GLP-1 receptor agonist with significant CYP interaction potential.
CagriSema (Semaglutide + Cagrilintide)
The combination of semaglutide with the amylin analog cagrilintide creates even more pronounced GI effects than semaglutide alone. Amylin, like GLP-1, delays gastric emptying. The additive effect may amplify absorption-based interactions with oral medications beyond what's seen with GLP-1 monotherapy. Enhanced monitoring of narrow-therapeutic-index drugs will likely be needed with this combination.
Novel Peptide Compounds on the Horizon
GHK-Cu (Copper Peptide)
GHK-Cu is a naturally occurring tripeptide with copper binding that promotes wound healing, collagen synthesis, and anti-inflammatory effects. Its emerging use in regenerative medicine raises interaction questions with copper-containing supplements, zinc (which competes with copper absorption), and chelation therapies. No formal drug interaction studies exist.
MOTS-c (Mitochondrial Peptide)
MOTS-c is a mitochondrial-derived peptide that improves insulin sensitivity and metabolic homeostasis. Its glucose-lowering properties may interact with diabetes medications similarly to GLP-1 agonists, though through different mechanisms. As MOTS-c moves toward clinical application, drug interaction studies will be essential.
Humanin
Another mitochondrial-derived peptide with neuroprotective and cytoprotective properties. Humanin interacts with the insulin/IGF-1 signaling pathway, creating potential interactions with diabetes medications and GH secretagogues. The clinical interaction profile remains entirely theoretical at this stage.
Expanding Indications for Existing Peptides
As GLP-1 agonists expand into treatment of addiction, neurodegenerative disease, PCOS, and cardiovascular disease, the co-medication landscape changes dramatically. Patients with these conditions take very different medication profiles than the metabolic syndrome population where most current interaction data has been generated.
For example:
- Addiction treatment: GLP-1 agonists combined with naltrexone, buprenorphine, or methadone for addiction creates a new interaction profile that hasn't been formally studied.
- Neurodegenerative disease: Combining GLP-1 agonists with Alzheimer's medications (donepezil, memantine) or Parkinson's medications (levodopa, dopamine agonists) requires new interaction assessment.
- PCOS: GLP-1 agonists combined with metformin, spironolactone, and oral contraceptives creates a multi-drug interaction scenario requiring integrated management.
Pharmacogenomics and Personalized Interaction Risk
The future of peptide-drug interaction management will increasingly incorporate pharmacogenomic data. Genetic variations in drug-metabolizing enzymes, drug transporters, and receptor targets can dramatically affect individual interaction risk. For example:
- CYP2C9 poor metabolizers may be more sensitive to warfarin interaction with GLP-1 agonists
- Genetic variations in GLP-1 receptor expression may affect the magnitude of gastric emptying delay
- GH receptor polymorphisms may influence the metabolic interaction profile of GH secretagogues
As pharmacogenomic testing becomes more accessible and affordable, integrating genetic data into peptide interaction risk assessment will become standard clinical practice. This represents the most promising avenue for truly personalized peptide therapy management.
The Need for Formal Interaction Studies
A major gap in the current literature is the absence of formal drug interaction studies for most non-GLP-1 peptides. The following studies are critically needed:
- BPC-157 pharmacokinetic interaction studies with major drug classes in humans
- TB-500 interaction studies with anticoagulants and immunosuppressants
- Thymosin Alpha-1 dose-response interaction studies with specific immunosuppressant regimens
- GH secretagogue interaction studies in populations with type 2 diabetes on multiple glucose-lowering agents
- NAD+ precursor interaction studies with hepatotoxic medications
- Multi-peptide combination studies evaluating interaction cascades
Until these studies are conducted, clinicians and patients must rely on mechanism-based interaction analysis, clinical observation, and conservative monitoring protocols - exactly the approach outlined throughout this report.
Stay updated on the latest peptide research developments through our peptide research hub and explore specific compound profiles in our comprehensive beginner's guide.
Clinical Case Studies and Practical Interaction Scenarios
Theory matters, but clinical reality is where peptide-drug interactions play out. This section presents realistic patient scenarios that illustrate how the interaction principles discussed throughout this report manifest in actual practice. These composite cases are drawn from published literature, clinical guidelines, and common clinical situations.
Case 1: The Metabolic Optimization Patient
Patient profile: 54-year-old male, BMI 34, type 2 diabetes, hypertension, hyperlipidemia
Current medications:
- Metformin 1000 mg twice daily
- Glimepiride 4 mg daily
- Lisinopril 20 mg daily
- Atorvastatin 40 mg daily
- Aspirin 81 mg daily
Proposed peptide therapy: Semaglutide 0.25 mg weekly (titrating to 2.4 mg) for weight management and glycemic control
Interaction analysis:
The primary concern is the glimepiride-semaglutide combination. The sulfonylurea dramatically increases hypoglycemia risk when combined with GLP-1 agonists - from under 4% to potentially 17-24%. The recommended approach: reduce glimepiride to 2 mg (50% reduction) at semaglutide initiation, with a plan to taper and discontinue glimepiride entirely by the time semaglutide reaches the 1.0 mg dose level.
Metformin requires no adjustment - this is a well-established, safe combination. Lisinopril, atorvastatin, and aspirin are all compatible with semaglutide. Atorvastatin peak levels may decrease slightly, but total exposure is preserved. As the patient loses weight (expected 15-20% body weight on semaglutide 2.4 mg), blood pressure may decrease enough to require lisinopril dose reduction. Plan for quarterly metabolic assessments and blood pressure checks.
Monitoring plan: fasting glucose and blood pressure at weeks 2, 4, 8, and 12. HbA1c at 12 weeks. Lipid panel at 12 weeks. Consider glimepiride discontinuation at week 8-12 based on glucose trends.
Case 2: The Post-Surgical Recovery Patient
Patient profile: 38-year-old female, ACL reconstruction recovery
Current medications:
- Oxycodone 5 mg every 6 hours as needed (post-surgical pain)
- Enoxaparin 40 mg daily (DVT prophylaxis)
- Levothyroxine 88 mcg daily (hypothyroidism)
- Combined oral contraceptive pill
Proposed peptide therapy: BPC-157 250 mcg twice daily + TB-500 750 mcg twice weekly for accelerated healing
Interaction analysis:
The BPC-157/TB-500 combination addresses the primary goal of accelerated surgical recovery. No formal drug interaction studies exist for either peptide with any of her medications. However, mechanism-based analysis reveals several considerations:
The enoxaparin (anticoagulant) plus angiogenic peptides combination warrants attention. BPC-157 and TB-500 both promote blood vessel formation and tissue repair. While this is the desired therapeutic effect, the concurrent anticoagulation means any newly formed blood vessels may be more prone to bleeding. The risk is probably low, but the surgical team should be aware of all peptides being used.
BPC-157's nitric oxide modulation could theoretically enhance vasodilation, creating a very modest additive blood pressure effect alongside opioid-induced vasodilation. Monitor for orthostatic hypotension, especially during early mobilization.
Levothyroxine absorption is unlikely to be affected by injected healing peptides, as neither BPC-157 nor TB-500 affects gastric emptying. Continue levothyroxine at the same dose.
The oral contraceptive is not affected by injected healing peptides (no gastric emptying interaction). Continue without modification.
Timing recommendation: start BPC-157/TB-500 approximately 3-5 days post-surgery, once initial hemostasis is established and surgical site bleeding is resolved. Continue for 6-8 weeks or until healing milestones are met.
Case 3: The Anti-Aging Protocol Patient
Patient profile: 62-year-old male, healthy, pursuing longevity optimization
Current medications:
- Testosterone cypionate 100 mg weekly (TRT)
- Anastrozole 0.5 mg twice weekly (estrogen management)
- Levothyroxine 75 mcg daily
- Rosuvastatin 10 mg daily
- Low-dose aspirin 81 mg daily
Proposed peptide therapy: CJC-1295/DAC 2 mg weekly + Ipamorelin 300 mcg nightly + NAD+ 250 mg daily (oral NMN)
Interaction analysis:
This is a complex protocol with multiple interaction points. The CJC-1295/Ipamorelin combination with testosterone creates additive IGF-1 elevation - both GH and testosterone stimulate IGF-1 production. IGF-1 levels must be monitored carefully to ensure they remain within the upper normal range (not supraphysiological). Target IGF-1 of 200-280 ng/mL for his age.
The GH secretagogue combination will affect his thyroid axis. GH enhances T4-to-T3 conversion, which could cause his levothyroxine dose to appear excessive (suppressed TSH, elevated free T3). Thyroid monitoring should include TSH, free T4, AND free T3 at 6-8 weeks after starting the GH peptides. Dose adjustment based on the complete panel, not TSH alone.
GH secretagogues increase insulin resistance. At 62, with existing cardiovascular risk factors (hence the statin and aspirin), glucose metabolism should be monitored closely. Baseline fasting insulin and glucose with HOMA-IR calculation, then repeat at 4 and 12 weeks. If fasting glucose exceeds 100 mg/dL or HOMA-IR exceeds 2.5, consider reducing ipamorelin dose or adding metformin.
NAD+ (NMN) and rosuvastatin both involve liver metabolism. While no direct interaction is documented, liver enzymes should be checked at baseline and 8 weeks. The combination is expected to be safe, but monitoring is prudent given the multi-compound protocol.
Anastrozole and GH peptides: GH can affect estrogen metabolism. Monitor estradiol levels alongside testosterone and IGF-1 to ensure the anastrozole dose remains appropriate as GH status changes.
Case 4: The Autoimmune Disease Patient
Patient profile: 45-year-old female, rheumatoid arthritis on biologic therapy
Current medications:
- Adalimumab 40 mg biweekly (TNF inhibitor)
- Methotrexate 15 mg weekly
- Folic acid 1 mg daily
- Prednisone 5 mg daily
- Celecoxib 200 mg daily (NSAID)
Proposed peptide therapy: BPC-157 for joint healing support
Interaction analysis:
This scenario illustrates why blanket recommendations don't work - context matters enormously. BPC-157 has anti-inflammatory and tissue repair properties that could theoretically benefit rheumatoid arthritis joints. However, several interaction concerns arise:
Methotrexate is immunosuppressive. BPC-157's effects on immune function are not fully characterized, but its modulation of various growth factor and cytokine pathways could theoretically interact with methotrexate's mechanism. The interaction is probably minor given BPC-157's primarily local tissue effects (especially with local injection), but the rheumatologist should be aware.
The prednisone dose is low (5 mg) and serves primarily as an anti-inflammatory rather than immunosuppressive agent. BPC-157 is unlikely to meaningfully oppose this low-dose steroid effect.
BPC-157 may provide some gastroprotection against celecoxib-induced gastric effects, based on animal data. This could be a favorable interaction, though it shouldn't be relied upon as the sole GI protective strategy.
The most important consideration: this patient should NOT use Thymosin Alpha-1, as it would directly oppose the immunosuppressive effects of adalimumab and methotrexate, potentially triggering a rheumatoid arthritis flare or undermining disease control. The distinction between healing peptides (likely safe) and immune peptides (potentially dangerous) in this patient is critical.
Recommended approach: discuss BPC-157 with the rheumatologist. If approved, start with local injections near affected joints rather than systemic subcutaneous administration. Monitor RA disease activity markers (CRP, ESR, joint counts) at regular intervals to ensure BPC-157 isn't interfering with the biologic therapy's effectiveness.
Case 5: The Pre-Surgical GLP-1 Patient
Patient profile: 47-year-old male, scheduled for elective cholecystectomy (gallbladder removal) in 3 weeks
Current medications:
- Semaglutide 1.7 mg weekly (for weight management - on this dose for 8 weeks)
- Metformin 500 mg twice daily
- Omeprazole 20 mg daily
- Lisinopril 10 mg daily
Perioperative management plan:
This patient is in the GLP-1 dose-escalation phase (recently moved to 1.7 mg), which places him in the moderate-to-higher risk category for perioperative aspiration. The cholecystectomy is laparoscopic and elective, allowing time for optimal preparation.
Week 1 (3 weeks pre-op): Continue semaglutide at current dose. Contact anesthesiologist and surgeon to discuss GLP-1 status.
Week 2 (2 weeks pre-op): Administer the last dose of semaglutide. This creates a 14-day washout before surgery, which recent evidence suggests is optimal for reducing anesthesia-related complications including aspiration risk, delayed emergence, and conversion to intubation.
Week 3 (day of surgery): Follow a liquid-only diet for 24 hours pre-operatively per multi-society guidelines. Continue metformin (unless otherwise directed by the anesthesiologist - some centers hold metformin the morning of surgery). Continue lisinopril as directed. Continue omeprazole (may help protect against acid aspiration).
Post-operative: Resume metformin and lisinopril when tolerating oral intake. Resume semaglutide 1 week post-operatively, restarting at the same dose (1.7 mg) rather than re-titrating from scratch, since the washout period was brief. Monitor for return of GI side effects, which may be more pronounced initially after the medication holiday.
Key consideration: this patient is having his gallbladder removed. GLP-1 agonists have been associated with gallbladder events, and the cholecystectomy may itself have been precipitated by GLP-1-related gallstone formation. Post-operative GLP-1 resumption is generally safe after cholecystectomy, but the patient should be monitored for bile duct-related symptoms.
Case 6: The Organ Transplant Patient Inquiry
Patient profile: 55-year-old male, kidney transplant 3 years ago, stable graft function
Current medications:
- Tacrolimus 3 mg twice daily (target trough 5-8 ng/mL)
- Mycophenolate mofetil 500 mg twice daily
- Prednisone 5 mg daily
- Amlodipine 5 mg daily
- Atorvastatin 20 mg daily
Patient request: Interested in Thymosin Alpha-1 for immune support after recurrent respiratory infections
Interaction analysis and recommendation:
This is a clear contraindication scenario. Thymosin Alpha-1 stimulates T-cell activation, dendritic cell maturation, and NK cell function - the precise immune responses that tacrolimus and mycophenolate are designed to suppress. Using Ta1 in this patient could trigger acute kidney transplant rejection, a life-threatening emergency that could result in loss of the transplanted organ and return to dialysis.
The recommendation is unambiguous: Thymosin Alpha-1 is contraindicated. Alternative approaches for recurrent infections should be explored with the transplant team, which might include adjusting immunosuppression levels (if the infections are severe), prophylactic antibiotics, optimizing vitamin D and zinc status, or other strategies that don't directly oppose the immunosuppressive regimen.
If the patient's infections are frequent and severe enough to warrant immune stimulation, the transplant team may consider extremely cautious use of specific agents under close graft function monitoring - but this is a transplant nephrology decision, not a peptide therapy decision.
This case illustrates the single most dangerous peptide-drug interaction discussed in this report. When the consequences of an interaction include organ loss, there is no room for experimentation.
Lessons From These Cases
- Every patient's interaction profile is unique - generic tables provide starting points, not final answers
- The specific combination of peptide + medication + patient characteristics determines actual risk
- Proactive dose adjustments prevent most adverse events (e.g., reducing sulfonylureas before starting GLP-1 therapy)
- Some contraindications are absolute - Thymosin Alpha-1 in transplant patients is the clearest example
- Communication with all healthcare providers is essential for safe peptide therapy management
- Monitoring protocols should be customized based on the specific interaction risk profile
Drug Interaction Resources and Quick Reference
This final section provides consolidated quick-reference tools for clinicians and patients managing peptide therapy alongside conventional medications.
The Five Most Dangerous Peptide-Drug Combinations
Based on the evidence reviewed in this report, the following five combinations carry the highest clinical risk:
- Thymosin Alpha-1 + Organ transplant immunosuppressants (tacrolimus, cyclosporine, mycophenolate) - Risk: graft rejection
- GLP-1 agonist + Sulfonylurea without dose reduction - Risk: severe hypoglycemia (17-24% incidence)
- GLP-1 agonist + Insulin without dose reduction - Risk: significant hypoglycemia
- GH secretagogues + Active cancer treatment - Risk: tumor promotion via IGF-1
- GLP-1 agonist + Surgery without appropriate washout - Risk: aspiration under anesthesia
The Five Most Commonly Missed Interactions
- GLP-1 agonist + Levothyroxine: Enhanced absorption plus weight-based dosing changes can produce hyperthyroidism symptoms. Often missed because thyroid isn't checked routinely during weight management.
- GH secretagogues + Diabetes medications: The gradual onset of GH-induced insulin resistance means glucose drift can go unnoticed for weeks.
- Tirzepatide + Oral contraceptives: 20% reduction in OCP absorption is not widely known. Unintended pregnancy is a serious consequence of missing this interaction.
- Weight loss from GLP-1 + Weight-based medications: Any medication dosed by body weight (warfarin, levothyroxine, some antibiotics) needs reassessment as weight changes significantly.
- BPC-157 + Dopaminergic medications: The dopamine-modulating effects of BPC-157 are well-documented in animal studies but rarely discussed in clinical peptide education.
Universal Rules for Safe Peptide-Drug Combination
- Disclose everything to every healthcare provider - peptides, supplements, recreational substances
- Establish baselines - comprehensive blood work before starting any peptide
- Start one at a time - 2-4 weeks between new additions to your protocol
- Monitor intensively during the first 12 weeks - this is the highest-risk period
- Maintain a medication diary - track start dates, doses, timing, and symptoms
- Know your narrow-therapeutic-index drugs - these need the most attention
- Plan ahead for surgery - 2-4 weeks notice minimum for perioperative management
- Report new symptoms promptly - don't assume symptoms are "just the peptide"
- Get regular blood work - objective data catches interactions before symptoms appear
- Review your protocol periodically - as your body composition and health status change, so do your interaction risks
For personalized protocol planning, visit our getting started page. For ongoing monitoring support, see our blood work monitoring guide.
Detailed Timing Protocols and Practical Daily Schedules
One of the most actionable strategies for minimizing peptide-drug interactions is proper timing of administration. This section provides detailed daily schedules and timing protocols for common medication combinations that address real-world patient needs.
Morning Medication Timing With GLP-1 Therapy
Patients on GLP-1 agonists often take multiple morning medications. The order and timing of administration can meaningfully affect absorption and interaction risk. Here are optimized protocols for common scenarios:
Protocol A: GLP-1 Agonist + Levothyroxine + Diabetes Medications
| Time | Action | Rationale |
|---|---|---|
| 6:00 AM | Levothyroxine with 4 oz water, empty stomach | Levothyroxine requires empty stomach for optimal absorption; most sensitive to timing interference |
| 6:30-7:00 AM | Wait period - no food, no other medications | Allows levothyroxine absorption before introducing other variables |
| 7:00 AM | Metformin with breakfast | Metformin is best tolerated with food; no PK interaction with GLP-1 |
| 7:00 AM | Other morning medications (statin, BP meds) with breakfast | Most medications tolerant of delayed absorption from GLP-1 effect |
| Any time | Weekly GLP-1 injection (same day each week) | Injectable GLP-1 doesn't interact with oral med absorption at injection time |
Protocol B: GLP-1 Agonist + Warfarin + Cardiovascular Medications
| Time | Action | Rationale |
|---|---|---|
| Morning | Lisinopril/ARB, statin, aspirin with breakfast | Wide therapeutic windows; timing flexibility |
| Evening (same time daily) | Warfarin | Consistent evening timing reduces INR variability |
| Any time | Weekly GLP-1 injection | No direct PK timing interaction |
| Ongoing | Consistent dietary vitamin K intake | GLP-1 appetite changes may alter vitamin K intake; consistency prevents INR fluctuation |
Protocol C: GH Secretagogue + Thyroid + Diabetes Medications
| Time | Action | Rationale |
|---|---|---|
| 6:00 AM | Levothyroxine with water, empty stomach | Prioritizes thyroid medication absorption |
| 7:00 AM | Metformin or other diabetes medications with breakfast | GH peptides don't affect gastric emptying, so timing is less critical than with GLP-1 |
| 10:00 PM | CJC-1295/Ipamorelin injection on empty stomach | GH release is optimized during sleep; food (especially carbohydrates) blunts GH secretion |
| Note | No carbohydrate-heavy snacks after 8 PM | Insulin spike from carbs suppresses GH release; this maximizes secretagogue effectiveness |
Protocol D: Multiple Peptides + Complex Medication Regimen
| Time | Action | Rationale |
|---|---|---|
| 6:00 AM | Levothyroxine (if applicable) | Separate from all other medications by 30-60 minutes |
| 7:00 AM | Breakfast + morning oral medications | Group all non-sensitive oral meds together for convenience |
| 7:15 AM | BPC-157 subcutaneous injection (if morning protocol) | Can be timed around meals; no gastric emptying concern |
| 12:00 PM | Lunch + any midday medications | Maintain consistent meal timing during GLP-1 therapy |
| 6:00 PM | Dinner + evening medications (warfarin if applicable) | Evening warfarin for consistency |
| 7:00 PM | BPC-157 second injection (if twice-daily protocol) | 12-hour spacing for consistent peptide levels |
| 10:00 PM | GH secretagogue injection (if applicable) | Empty stomach, pre-sleep timing |
| Weekly | GLP-1 agonist injection (same day/time each week) | Consistency reduces interaction variability |
Managing the Dose Escalation Period
The highest-risk period for peptide-drug interactions is during dose escalation - when increasing from one peptide dose to the next. This applies primarily to GLP-1 agonists but is relevant for any peptide with titration protocols.
During each GLP-1 dose escalation step (typically every 4 weeks):
- Week 1 of new dose: Monitor for increased GI symptoms (nausea, vomiting, diarrhea). These symptoms can prevent absorption of oral medications. If vomiting occurs within 1 hour of taking oral medications, consider re-dosing critical medications (not applicable for warfarin or other drugs where re-dosing creates toxicity risk - contact your provider).
- Week 2 of new dose: Check drug levels for narrow-therapeutic-index medications if symptoms suggest altered absorption. Fasting glucose if on diabetes medications.
- Weeks 3-4 of new dose: Symptoms should be stabilizing. If GI symptoms persist at week 4, discuss with your provider before the next dose escalation. Persistent nausea or vomiting warrants medication timing review.
Managing GI Side Effects That Affect Medication Absorption
GLP-1-induced nausea and vomiting don't just make patients uncomfortable - they can directly interfere with oral medication absorption. Practical strategies for maintaining medication effectiveness during GI symptoms:
- Vomiting within 1 hour of oral medication: The medication may not have been absorbed. For most medications, re-dosing is appropriate. For narrow-therapeutic-index drugs (warfarin, digoxin, lithium, phenytoin), do NOT re-dose without contacting your provider - overdosing is more dangerous than one missed dose.
- Persistent nausea preventing medication intake: If you can't keep medications down for more than 24 hours, contact your provider urgently. Critical medications (anticonvulsants, anticoagulants, immunosuppressants) may need alternative routes of administration (IV, rectal, transdermal).
- Diarrhea reducing absorption: Diarrhea can push medications through the GI tract too quickly for complete absorption. Extended-release formulations are particularly vulnerable. If diarrhea is persistent and severe, drug level monitoring is advisable for critical medications.
- Constipation creating retention: GLP-1-induced constipation can delay drug transit, potentially increasing absorption of some medications. This rarely causes problems but is worth noting as a mechanism for the occasional patient who experiences enhanced drug effects during GLP-1-induced constipation.
Seasonal and Travel Considerations
Peptide-drug interactions can be influenced by factors patients might not consider:
Hot Weather and Dehydration
GLP-1 agonists can cause nausea and reduce fluid intake. Hot weather increases fluid loss through perspiration. The combined dehydration risk affects renally cleared medications (lithium, metformin, digoxin) and can concentrate narrow-therapeutic-index drugs to dangerous levels. During hot weather, peptide users should increase water intake proactively and monitor for dehydration symptoms.
Time Zone Changes and Medication Timing
For patients taking daily medications on precise schedules, crossing time zones disrupts the carefully planned timing protocols outlined above. General approach: shift medication timing by 1-2 hours per day until adjusted to the new time zone. Weekly GLP-1 injections can be shifted by up to 2 days without clinically significant impact. GH secretagogue timing should follow local bedtime in the new time zone from day one.
High Altitude
High altitude increases red blood cell production. GH secretagogues can also stimulate erythropoiesis. Patients on GH peptides who spend extended time at high altitude should monitor hematocrit to avoid polycythemia, especially if also on testosterone replacement therapy (which independently raises hematocrit).
Dietary Changes During Travel
Travel often changes dietary patterns - different cuisines, altered meal timing, unfamiliar foods. For patients on warfarin, changes in vitamin K-rich food consumption (leafy greens, natto, liver) during travel can cause INR fluctuations on top of any GLP-1-related effects. Maintain as consistent a diet as possible, or ask your provider about additional INR monitoring during and after travel.
Handling Missed Peptide Doses
Missed peptide doses create their own interaction considerations - the sudden absence of the peptide's physiological effects can transiently alter co-administered drug behavior.
Missed GLP-1 Agonist Dose
If a weekly semaglutide or tirzepatide dose is missed by more than 5 days, skip it and take the next dose on schedule. During the gap, gastric emptying returns toward normal, which means oral medications may absorb differently (faster Cmax, potentially earlier onset of action). Patients on narrow-therapeutic-index drugs may notice subtle differences in medication effects during the gap - this is expected and self-correcting once the next dose is administered.
Missed GH Secretagogue Dose
Missing a single nightly dose of ipamorelin or CJC-1295 has minimal interaction impact. The pulsatile GH release from a single dose doesn't create sustained metabolic effects that would significantly alter co-administered drug behavior. Simply resume the next scheduled dose.
Missed Healing Peptide Dose
BPC-157 and TB-500 have relatively short-lived peak effects. Missing a dose means a gap in the healing stimulus but doesn't create a withdrawal-type phenomenon that would interact with other medications. Resume at the next scheduled dose.
Transitioning Between Peptide Therapies
When switching from one peptide to another within the same class (e.g., semaglutide to tirzepatide, or ipamorelin to MK-677), there's a transition period where interaction profiles shift. Key considerations:
- Semaglutide to tirzepatide: Tirzepatide may produce more pronounced gastric emptying delay and has the oral contraceptive interaction that semaglutide doesn't. Re-evaluate all oral medication interactions when switching. Implement backup contraception if applicable.
- Injectable to oral GLP-1: Switching from injectable semaglutide to oral Rybelsus introduces the strict dosing requirements (empty stomach, 30-minute wait before other medications). Levothyroxine interaction is amplified with oral semaglutide.
- Ipamorelin to MK-677: MK-677 produces more sustained GH elevation and more significant glucose effects. Patients switching from ipamorelin to MK-677 may need diabetes medication adjustment that wasn't necessary with ipamorelin.
During any peptide transition, apply the same monitoring principles as initiating a new peptide: enhanced blood work at 4 and 12 weeks, with drug level checks for narrow-therapeutic-index medications. For more on peptide dosing protocols and transitions, visit our peptide calculator and research hub.
Extended Drug Class Analysis
Beyond the major drug classes covered above, peptide therapy users commonly take medications from several additional categories that warrant interaction analysis. This section covers anticonvulsants, respiratory medications, pain management drugs, gastrointestinal medications, and dermatological treatments.
Anticonvulsant Medications
Anticonvulsants (anti-seizure drugs) are used not only for epilepsy but also for neuropathic pain, bipolar disorder, and migraine prevention. Several have narrow therapeutic windows, making them vulnerable to absorption-based interactions with GLP-1 agonists.
Phenytoin (Dilantin)
Phenytoin has notoriously complex pharmacokinetics - it follows zero-order kinetics at therapeutic levels, meaning small changes in absorption can produce disproportionately large changes in blood levels. While no formal study has evaluated phenytoin with GLP-1 agonists, the delayed gastric emptying could theoretically alter phenytoin absorption patterns. Patients on phenytoin who start GLP-1 therapy should have phenytoin levels checked at 2, 4, and 8 weeks, then monthly during dose escalation.
Valproic Acid (Depakote)
Valproic acid is available in multiple formulations including enteric-coated and extended-release versions. Delayed gastric emptying from GLP-1 agonists could alter the release timing of these formulations. Additionally, valproic acid is associated with weight gain, which GLP-1-mediated weight loss may partially counteract - a potentially beneficial pharmacodynamic interaction. Monitor valproic acid levels during GLP-1 initiation.
Carbamazepine (Tegretol)
Carbamazepine is a CYP3A4 inducer with narrow therapeutic index. While GLP-1 agonists don't directly affect CYP enzymes, GH secretagogues may indirectly influence CYP3A4 expression through growth hormone effects. The combination of carbamazepine with GH peptides warrants drug level monitoring. The gastric emptying effects of GLP-1 agonists on carbamazepine absorption are probably clinically insignificant, as carbamazepine is slowly and completely absorbed regardless of gastric transit time.
Lamotrigine (Lamictal)
Lamotrigine is increasingly used for bipolar disorder and has important interactions with oral contraceptives (estrogen decreases lamotrigine levels). Since tirzepatide can reduce oral contraceptive absorption, patients on the triple combination of tirzepatide + oral contraceptives + lamotrigine face a complex interaction web: reduced OCP levels from tirzepatide could increase lamotrigine levels by removing the estrogen-mediated reduction. This cascade interaction requires monitoring of lamotrigine levels, contraceptive efficacy, and clinical response when tirzepatide is added.
Gabapentin and Pregabalin
These drugs are absorbed primarily in the upper small intestine through active transport mechanisms. GLP-1-induced delayed gastric emptying could theoretically alter the timing of drug delivery to the absorption site, but both drugs have relatively wide therapeutic windows. No dose adjustments are typically needed, though patients may notice a delayed onset of effect for pain or anxiety relief doses.
Respiratory Medications
Inhaled Corticosteroids and Bronchodilators
Inhaled medications bypass the GI tract entirely, so gastric emptying delays from GLP-1 agonists are irrelevant. No interaction concerns exist for standard inhaled asthma and COPD medications (fluticasone, budesonide, albuterol, formoterol, tiotropium) with any peptide therapy class.
Oral Corticosteroids (Prednisone, Methylprednisolone)
Oral corticosteroids at anti-inflammatory or immunosuppressive doses interact with multiple peptide classes:
- With GLP-1 agonists: Corticosteroids raise blood glucose. GLP-1 agonists lower it. The net effect depends on corticosteroid dose and duration. Short courses (5-7 day prednisone tapers) probably don't require GLP-1 dose adjustment. Chronic corticosteroid use may necessitate GLP-1 dose increases to maintain glycemic control.
- With GH secretagogues: Both corticosteroids and GH can raise blood glucose, creating additive hyperglycemic risk. Additionally, chronic corticosteroid use suppresses the hypothalamic-pituitary axis, which may blunt GH secretagogue effectiveness.
- With Thymosin Alpha-1: Moderate interaction - high-dose corticosteroids are immunosuppressive, opposing Ta1's immunostimulatory effects. Low-dose anti-inflammatory steroid use is less concerning.
Montelukast (Singulair)
No interaction with peptide therapies expected. Montelukast is well-absorbed regardless of gastric emptying timing and has a wide therapeutic window.
Theophylline
Theophylline has a narrow therapeutic index and complex metabolism through CYP1A2. While GLP-1 agonists don't directly affect CYP1A2, the weight loss associated with GLP-1 therapy could change theophylline's volume of distribution. GH secretagogues' potential effects on CYP enzyme expression add another theoretical concern. Monitor theophylline levels in patients starting either peptide class.
Pain Management Medications
Opioids
Opioid analgesics and GLP-1 agonists both slow gastrointestinal motility. Combining them can produce additive constipation, nausea, and delayed gastric emptying effects. This is usually manageable with proactive bowel management (increased fiber, adequate hydration, stool softeners), but severe gastroparesis-like symptoms can develop in susceptible patients.
For patients on chronic opioid therapy who start GLP-1 agonists:
- Start a bowel regimen proactively rather than reactively
- Initiate the GLP-1 agonist at the lowest dose with slow titration
- Consider switching from oral to transdermal opioid formulations if gastric emptying delay produces erratic oral opioid absorption
- Monitor for signs of opioid accumulation (excessive sedation, respiratory depression) if absorption patterns change
Extended-release opioid formulations deserve particular attention. OxyContin, MS Contin, and similar extended-release formulations rely on predictable GI transit for drug release. Dramatically slowed gastric emptying could alter drug release patterns, potentially creating periods of higher-than-expected or lower-than-expected pain relief.
Tramadol
Tramadol has both opioid and SNRI properties. Its serotonergic activity adds a theoretical interaction concern with BPC-157 (which modulates serotonin systems) and with any peptide combination that also affects serotonergic pathways. Monitor for serotonergic symptoms (agitation, rapid heart rate, tremor) when combining tramadol with BPC-157.
NSAIDs (Ibuprofen, Naproxen, Celecoxib)
NSAIDs and GLP-1 agonists both affect the GI tract but through different mechanisms. NSAIDs inhibit protective prostaglandin synthesis in the gastric mucosa. GLP-1 agonists slow gastric emptying and may increase gastric acid contact time with the mucosa. The theoretical risk of additive GI toxicity exists, though clinical data doesn't suggest a dramatically increased risk.
BPC-157 may actually counteract NSAID gastrotoxicity based on animal studies, representing a potentially beneficial interaction. However, this hasn't been confirmed in human trials and shouldn't be relied upon for gastroprotection.
Acetaminophen
As discussed in the absorption section, acetaminophen absorption rate is delayed by GLP-1 agonists (Cmax reduced up to 55% with first-dose tirzepatide), but total absorption is preserved. For pain relief purposes, patients may notice a slightly delayed onset of action, but the overall effectiveness is maintained. Sustained acetaminophen use for chronic conditions is not affected in a clinically meaningful way.
Psychiatric Medications - Detailed Analysis
SSRIs (Fluoxetine, Sertraline, Escitalopram, Paroxetine)
SSRIs have wide therapeutic windows and no documented pharmacokinetic interactions with GLP-1 agonists. The delayed gastric emptying may slightly alter absorption timing but doesn't affect total exposure. One clinical consideration: GLP-1-mediated weight loss may be welcome in patients on SSRIs, which commonly cause weight gain. As weight decreases, SSRI dose adjustments are not typically needed because SSRIs aren't weight-dosed.
With GH secretagogues, no interaction is expected. With BPC-157, the theoretical serotonergic interaction warrants awareness but hasn't been clinically documented.
SNRIs (Venlafaxine, Duloxetine, Desvenlafaxine)
Similar to SSRIs, no significant pharmacokinetic interactions with peptide therapies. Extended-release formulations (venlafaxine XR, duloxetine DR) could theoretically have altered release timing with GLP-1-induced gastric emptying delay, but clinical significance is unlikely given their wide therapeutic windows.
Atypical Antipsychotics (Quetiapine, Olanzapine, Risperidone, Aripiprazole)
Atypical antipsychotics commonly cause metabolic syndrome including weight gain, insulin resistance, and dyslipidemia. GLP-1 agonists can be used therapeutically to counteract these metabolic effects - an emerging area of psychiatric pharmacotherapy. The combination is generally safe and potentially beneficial from a metabolic standpoint.
However, the case report of supratherapeutic ziprasidone levels with semaglutide serves as a reminder that individual antipsychotics may have clinically relevant absorption changes with GLP-1 therapy. Monitoring psychiatric medication effectiveness and side effects during GLP-1 initiation is prudent, particularly for antipsychotics with narrower therapeutic ranges.
With GH secretagogues, the interaction is primarily metabolic: GH-induced insulin resistance may worsen the metabolic syndrome already caused by atypical antipsychotics. If this combination is used, metabolic monitoring (glucose, lipids, HbA1c) should be more frequent.
Lithium
Lithium deserves special attention due to its very narrow therapeutic index (0.6-1.2 mEq/L for most indications). Several peptide-related factors can affect lithium levels:
- Dehydration from GLP-1-induced nausea or vomiting: Even mild dehydration concentrates lithium, potentially reaching toxic levels. Patients on lithium who start GLP-1 agonists must maintain aggressive hydration, especially during the nausea-prone dose escalation phase.
- Weight loss: Significant weight loss can alter lithium's volume of distribution, potentially increasing blood levels at the same dose.
- Renal function changes: GLP-1 agonists can affect renal hemodynamics. Any change in kidney function directly impacts lithium clearance.
For patients on lithium starting GLP-1 therapy: check lithium levels weekly for the first month, then biweekly for months 2-3, then monthly during active weight loss. Ensure adequate hydration and promptly address any GI symptoms that could lead to dehydration.
MAOIs (Phenelzine, Tranylcypromine, Selegiline)
MAOIs interact with the monoamine neurotransmitter system. BPC-157's documented effects on dopaminergic and serotonergic systems create a theoretical interaction concern. The combination of MAOIs with BPC-157 should be approached with extreme caution and medical supervision, as MAOIs have well-known interaction potential with any serotonergic or dopaminergic agent.
Dermatological Medications
Isotretinoin (Accutane)
Isotretinoin is a fat-soluble drug that requires dietary fat for optimal absorption. GLP-1-induced appetite suppression and reduced food intake could theoretically reduce isotretinoin absorption if patients aren't consuming adequate fat with their doses. Additionally, isotretinoin can elevate triglycerides and affect liver function - both of which should be monitored alongside any peptide therapy.
GH secretagogues are generally not recommended during isotretinoin treatment, as both can affect lipid metabolism and liver function. If the combination is clinically necessary, more frequent lipid and liver enzyme monitoring is warranted.
Methotrexate (Low-Dose for Psoriasis or Rheumatoid Arthritis)
Low-dose methotrexate has immunosuppressive properties relevant to Thymosin Alpha-1 interactions. Ta1's immune-stimulating effects could potentially counteract methotrexate's immunosuppressive mechanism, reducing its effectiveness for autoimmune conditions. This combination should be managed by a rheumatologist or dermatologist familiar with both agents.
NAD+ precursors and methotrexate both involve folate metabolism, creating another potential interaction point. Methotrexate inhibits dihydrofolate reductase, and NAD+ biosynthesis intersects with folate-dependent one-carbon metabolism. While clinical significance is uncertain, awareness of this metabolic overlap is appropriate.
Urological Medications
PDE5 Inhibitors (Sildenafil, Tadalafil, Vardenafil)
PDE5 inhibitors work through the nitric oxide/cGMP pathway. BPC-157 also modulates the NO system, creating a theoretical interaction. Both promote vasodilation through NO-mediated mechanisms. Additive vasodilation could theoretically enhance the blood pressure-lowering effects of PDE5 inhibitors, but no clinical reports of adverse events from this combination exist.
GLP-1 agonists may delay the absorption of oral PDE5 inhibitors, potentially delaying onset of action. For patients using these medications on an as-needed basis, taking the PDE5 inhibitor on an empty stomach (when possible) and allowing extra time for onset may be helpful.
Alpha-Blockers (Tamsulosin, Alfuzosin)
Used for benign prostatic hyperplasia (BPH), alpha-blockers cause vasodilation as a side effect. No direct interaction with peptide therapies expected, though the blood pressure-lowering effects of alpha-blockers combined with GLP-1 agonists' potential cardiovascular effects should be considered in patients prone to orthostatic hypotension.
Ophthalmologic Medications
Topical Eye Drops
Topical ophthalmic medications (timolol, brimonidine, latanoprost, etc.) are absorbed systemically at very low levels and bypass the GI tract entirely. No interaction with peptide therapies expected.
Oral Medications for Ocular Conditions
One consideration specific to GLP-1 agonists: semaglutide's prescribing information notes reports of diabetic retinopathy complications in patients with type 2 diabetes. This appears to be related to rapid improvement in glycemic control rather than a direct drug effect. Patients with pre-existing diabetic retinopathy should have ophthalmologic monitoring when starting GLP-1 therapy, regardless of other medication interactions.
Figure 9: Extended drug class interaction profiles across major peptide categories. This comprehensive reference covers anticonvulsants, respiratory medications, pain management drugs, psychiatric medications, and specialty therapeutics.
Special Populations and Unique Interaction Considerations
Certain patient populations face heightened interaction risks when combining peptide therapies with conventional medications. Age, organ function, pregnancy status, and comorbidity burden all influence how peptide-drug combinations behave in the body.
Elderly Patients (Age 65+)
Older adults are disproportionately affected by peptide-drug interactions for several reasons:
- Polypharmacy: The average American over 65 takes 5+ medications daily. Each additional drug increases the complexity of potential interactions with peptide therapies.
- Reduced renal function: Age-related GFR decline affects clearance of renally eliminated drugs. GLP-1 agonists can further affect renal hemodynamics, compounding the issue. Drugs with renal clearance (metformin, lithium, digoxin, gabapentin) may require dose adjustment.
- Reduced hepatic function: Age-related decline in liver blood flow and enzyme activity slows drug metabolism. While peptides themselves aren't hepatically metabolized, their effects on co-administered drugs that are hepatically cleared may be magnified.
- Altered body composition: Higher body fat percentage and reduced lean mass in older adults changes drug distribution. GLP-1-mediated weight loss in elderly patients can be particularly impactful on drug levels.
- Increased fall risk: Hypoglycemia from GLP-1/sulfonylurea combinations, orthostatic hypotension from vasodilatory peptide effects, and sedation from CNS-active drug interactions all increase fall risk - a major concern in the elderly.
For elderly patients, the general approach is: start peptide doses lower, titrate slower, monitor more frequently, and maintain a lower threshold for dose adjustments of concurrent medications.
Patients With Renal Impairment
Kidney function affects peptide-drug interactions in several ways:
- GLP-1 agonists: Semaglutide and liraglutide don't require renal dose adjustment, but they can cause nausea and vomiting that leads to dehydration - a significant risk for patients with compromised renal function. Dehydration in CKD patients can precipitate acute kidney injury and rapidly change blood levels of renally cleared medications.
- GH secretagogues: GH can increase renal blood flow and GFR acutely. In patients with CKD, this hemodynamic effect should be monitored. The glucose-raising effects of GH may interact with CKD-related insulin resistance, creating additive metabolic challenges.
- Renally cleared drugs of concern: Metformin (contraindicated in severe CKD), lithium, digoxin, gabapentin/pregabalin, and allopurinol all require dose adjustment in renal impairment. Adding peptide therapies that affect the kidney (directly or through dehydration) adds another variable to already complex dosing calculations.
Patients With Hepatic Impairment
Liver disease affects drug metabolism broadly and creates specific peptide interaction concerns:
- NAD+ precursors: The liver is central to NAD+ metabolism. In patients with liver disease, NAD+ precursors may be metabolized unpredictably. Baseline liver enzyme monitoring is essential, and transient elevations have been reported in some studies.
- GH secretagogues: IGF-1 is primarily produced in the liver. Patients with cirrhosis have impaired IGF-1 production regardless of GH stimulation, making GH secretagogues less effective and potentially altering their metabolic impact.
- GLP-1 agonists: While GLP-1 agonists themselves aren't hepatically metabolized, they're increasingly used for NAFLD/NASH (where they show therapeutic benefit). In these patients, monitoring liver enzymes during therapy helps distinguish therapeutic effects from potential drug interactions.
Pregnancy and Lactation
Most peptide therapies should be discontinued during pregnancy and breastfeeding:
- GLP-1 agonists: Contraindicated in pregnancy. Discontinue at least 2 months before planned conception (for semaglutide) due to its long half-life.
- GH secretagogues: No adequate safety data in pregnancy. Discontinue before conception.
- BPC-157 and TB-500: No human pregnancy safety data. Avoid during pregnancy and lactation.
- Thymosin Alpha-1: No adequate human pregnancy data. Avoid unless benefits clearly outweigh unknown risks.
- NAD+ precursors: No adequate human pregnancy or lactation safety data. Avoid during pregnancy and breastfeeding.
The drug interaction concern during pregnancy centers on the transition period: when peptide therapies are discontinued in anticipation of conception, the removal of their physiological effects may require adjustment of co-administered medications. For example, a patient who discontinues GLP-1 therapy before pregnancy may experience weight regain and worsening insulin resistance, requiring reinitiation or dose adjustment of diabetes medications.
Pediatric Patients
Peptide therapy in pediatric populations is limited primarily to FDA-approved GLP-1 agonists for obesity (semaglutide is approved for ages 12+). Drug interaction considerations in adolescents include:
- Growth hormone physiology is different in growing children - GH secretagogues could theoretically affect normal growth patterns
- Adolescents on ADHD medications (stimulants) who start GLP-1 therapy may experience altered stimulant absorption timing
- Oral contraceptive interactions with tirzepatide are particularly relevant for adolescent females starting weight management
- Body composition changes during GLP-1 therapy may affect weight-based medication dosing differently in growing patients versus adults
Athletes and Competitive Sports Participants
Beyond WADA ban considerations for certain peptides (BPC-157, TB-500, GH secretagogues), athletes face unique interaction concerns:
- Dehydration risk: Athletic activity combined with GLP-1-induced GI symptoms and reduced fluid intake creates heightened dehydration risk, affecting any medications with renal clearance
- Performance supplements: Many athletes take pre-workout supplements containing stimulants, amino acids, and nitric oxide precursors that could interact with BPC-157's NO-modulating effects or GH secretagogues' metabolic effects
- Electrolyte management: Intensive training combined with peptide therapy effects on fluid balance and GI function requires careful attention to electrolyte supplementation and monitoring
Figure 10: Special population risk factors that amplify peptide-drug interaction potential. Understanding these population-specific considerations is essential for safe peptide therapy management.
Comprehensive Monitoring and Safety Protocols
Proactive monitoring is the most effective strategy for detecting and managing peptide-drug interactions before they cause clinical harm. This section consolidates the monitoring recommendations from throughout the report into a unified framework.
Universal Baseline Testing
Before starting any peptide therapy, establish baseline values for the following:
| Test Category | Specific Tests | Why It Matters |
|---|---|---|
| Metabolic Panel | Fasting glucose, HbA1c, fasting insulin, HOMA-IR | Detects GH and GLP-1 effects on glucose metabolism |
| Lipid Panel | Total cholesterol, LDL, HDL, triglycerides | GH and GLP-1 both affect lipid metabolism |
| Thyroid Function | TSH, free T4, free T3 | GH secretagogues and GLP-1 effects on thyroid axis |
| Liver Function | AST, ALT, GGT, bilirubin | NAD+ metabolism; general safety monitoring |
| Kidney Function | BUN, creatinine, eGFR | Renal effects of GLP-1; renally cleared drug dosing |
| Hormonal Panel | IGF-1, GH (if GH secretagogue), cortisol AM | GH secretagogue response monitoring |
| Hematology | CBC with differential | GH effects on hematocrit; immune peptide monitoring |
| Coagulation | INR/PT (if on warfarin) | GLP-1 effects on warfarin response |
| Drug Levels | Levels for any narrow-therapeutic-index drugs | Baseline for comparison after peptide initiation |
Follow-Up Monitoring Schedule
After peptide therapy initiation, the monitoring schedule should be most intensive during the first 12 weeks - the period of highest interaction risk.
Weeks 2-4 (First Follow-Up)
- Fasting glucose (all peptide users)
- Drug levels for narrow-therapeutic-index medications (warfarin INR, digoxin, lithium, phenytoin, tacrolimus)
- Symptom review: GI symptoms, glucose changes, blood pressure changes, mood/cognition changes
- Weight check (for weight-based medication dosing assessment)
Weeks 8-12 (Second Follow-Up)
- Complete metabolic panel including glucose, insulin, HbA1c
- Thyroid function (TSH, free T4, free T3) if on GH secretagogues or GLP-1 with levothyroxine
- IGF-1 if on GH secretagogues
- Liver enzymes if on NAD+ precursors or hepatotoxic co-medications
- Drug levels for narrow-therapeutic-index medications
- Review of all co-administered medication doses - are adjustments needed based on weight change or metabolic shifts?
Ongoing (Every 3-6 Months)
- Comprehensive metabolic panel
- Thyroid function (annually at minimum, more frequently if on levothyroxine)
- IGF-1 (every 3-6 months while on GH secretagogues)
- Drug level monitoring as clinically indicated
- Annual comprehensive blood work including CBC, lipids, hormones
Red Flags: When to Contact Your Provider Immediately
Certain symptoms during peptide therapy suggest a clinically significant drug interaction that requires urgent medical evaluation:
- Severe hypoglycemia (blood glucose below 54 mg/dL, confusion, loss of consciousness) - may indicate GLP-1/insulin or GLP-1/sulfonylurea interaction
- Unexplained bleeding or bruising - may indicate warfarin interaction or anticoagulant potentiation
- Rapid heart rate with tremor and agitation - possible thyroid hormone excess from levothyroxine interaction or serotonin syndrome
- Severe, persistent abdominal pain - possible pancreatitis (GLP-1 agonists) or GI complication
- Peripheral edema with shortness of breath - possible fluid retention from GH secretagogues, especially combined with TZDs
- New or worsening psychiatric symptoms - possible interaction with psychotropic medications
- Inability to keep food or medications down - severe GI effects may prevent absorption of critical medications
- Seizure in a patient on anticonvulsants - possible change in anticonvulsant absorption or levels
Creating Your Personal Interaction Profile
Use these steps to create a personal drug interaction assessment:
- List all prescription medications, OTC drugs, supplements, and peptides you currently take
- Identify any narrow-therapeutic-index medications on your list
- Review the interaction tables in this report for each peptide + medication combination
- Share this list with your healthcare provider at your next visit
- Establish a monitoring schedule based on your specific combination risk level
- Keep a symptom diary for the first 12 weeks of any new peptide therapy
For a personalized assessment tool, visit our peptide calculator.
For the most up-to-date monitoring protocols and side effect management strategies, see our complete side effects guide and blood work monitoring reference.
Nootropic Peptide Interactions
Nootropic peptides - compounds used for cognitive enhancement, neuroprotection, and brain health - include Semax, Selank, Dihexa, and others. Their interaction profiles with conventional medications are poorly characterized, but their neurological mechanisms warrant careful consideration.
Semax
Semax is a synthetic analog of ACTH (4-10) with nootropic, neuroprotective, and neurotropic properties. It modulates BDNF expression and affects dopaminergic, serotonergic, and cholinergic neurotransmitter systems.
Potential interaction considerations:
- Antidepressants (SSRIs, SNRIs): Semax's effects on serotonin and BDNF pathways overlap with antidepressant mechanisms. While this could theoretically be complementary, the combined serotonergic activity should be monitored. No cases of serotonin syndrome with Semax have been reported, but the interaction has not been formally studied.
- Stimulants (amphetamine, methylphenidate): Semax modulates dopamine systems. Combined use with stimulant medications could produce unpredictable cognitive effects. Start with lower doses of both if combining.
- Anticonvulsants: Semax's neuromodulatory effects could theoretically alter seizure thresholds. Patients on anticonvulsants should not use Semax without neurologist approval.
- Cholinesterase inhibitors (donepezil, rivastigmine): Semax affects cholinergic systems. Combined use with cholinesterase inhibitors may produce additive cholinergic effects - potentially beneficial for cognition but also increasing risk of cholinergic side effects (GI upset, bradycardia).
Selank
Selank is a synthetic analog of tuftsin with anxiolytic and nootropic properties. It modulates GABA and affects the balance of T-helper cell cytokines.
- Benzodiazepines: Selank's GABA-modulating effects could be additive with benzodiazepine anxiolytics. Combined use may produce excessive sedation or CNS depression. Use with caution and start with lower doses.
- Anxiolytics generally: Any medication that reduces anxiety through CNS mechanisms (buspirone, hydroxyzine, gabapentin) could have additive effects with Selank. Monitor for excessive sedation.
- Immunosuppressants: Selank's immune-modulating properties (cytokine balance) create a similar but less dramatic interaction concern as Thymosin Alpha-1. The effect is milder, but patients on immunosuppressive therapy should discuss Selank use with their prescriber.
Dihexa
Dihexa is an oligopeptide derived from angiotensin IV with potent cognitive-enhancing properties in animal models. It crosses the blood-brain barrier and acts on hepatocyte growth factor (HGF) receptors.
- ACE inhibitors and ARBs: Dihexa is derived from the angiotensin system. While its mechanism of action differs from antihypertensive angiotensin modulators, the shared system origin raises theoretical interaction questions. Blood pressure monitoring during combined use is prudent.
- Blood pressure medications generally: Dihexa's vascular effects through HGF pathways could theoretically influence blood pressure. No clinical data exists, but monitoring is advisable.
- Cancer therapy: HGF receptor activation promotes cell growth and proliferation. Dihexa should be avoided in patients with active cancer or recent cancer history.
A Note on Evidence Levels
The interaction profiles listed for nootropic peptides are based almost entirely on mechanism-of-action analysis rather than clinical studies. No formal drug interaction trials have been conducted for Semax, Selank, or Dihexa with any conventional medication. The recommendations above are theoretical and conservative - designed to flag potential concerns rather than confirm known interactions. Always discuss nootropic peptide use with your prescribing physician, especially if you're on psychotropic medications.
Supplement Interactions That Often Get Overlooked
Most interaction discussions focus on prescription medications, but many peptide users also take a substantial list of supplements, and some of these combinations deserve more attention than they typically receive. Berberine, for example, is widely used as a natural insulin sensitizer and is pharmacologically active enough to produce clinically meaningful drug interactions. When combined with GLP-1 agonists like semaglutide or tirzepatide, berberine's additive insulin-sensitizing effect can increase hypoglycemia risk, particularly in patients also taking sulfonylureas or insulin. It also inhibits CYP3A4 and P-glycoprotein, which could affect the metabolism of other co-administered compounds.
High-dose fish oil (omega-3 fatty acids) at prescription-strength doses (4 grams daily) has measurable anticoagulant activity. When combined with peptides that also affect hemostasis, such as BPC-157 (which modulates the nitric oxide system and platelet function) or TB-500 (which influences actin polymerization in platelets), the aggregate effect on bleeding risk is theoretically increased, though no clinical reports of significant bleeding from these specific combinations exist. Patients on anticoagulant therapy who add both fish oil and tissue-repair peptides should be aware of this layered risk.
Ashwagandha, another popular supplement among biohacking-oriented peptide users, can increase thyroid hormone levels. This interaction becomes relevant for individuals using growth hormone secretagogues like CJC-1295/Ipamorelin or MK-677, since growth hormone itself influences thyroid hormone conversion. The combination of ashwagandha-stimulated thyroid production and GH-mediated T4-to-T3 conversion could produce symptoms of mild hyperthyroidism in susceptible individuals, including anxiety, tremor, and rapid heart rate.
The general principle worth remembering is that "natural" doesn't mean "interaction-free." Any compound with enough biological activity to produce a desired health effect has enough activity to interact with other biologically active compounds. Maintaining a complete list of all supplements, peptides, and medications for your healthcare provider is the single most effective step you can take to avoid problematic combinations. The free assessment includes a review of current supplements and medications to flag potential concerns.
Frequently Asked Questions
References
- Granhall C, Donsmark M, Blicher TM, et al. Safety and pharmacokinetics of single and multiple ascending doses of the novel oral human GLP-1 analogue, oral semaglutide, in healthy subjects and subjects with type 2 diabetes. Clinical Pharmacokinetics. 2019;58(6):781-791. DOI: 10.1007/s40262-018-0728-4
- Kapitza C, Nosek L, Jensen L, et al. Semaglutide, a once-weekly human GLP-1 analog, does not reduce the bioavailability of the combined oral contraceptive, ethinylestradiol/levonorgestrel. Journal of Clinical Pharmacology. 2015;55(5):497-504. DOI: 10.1002/jcph.443
- Jensen L, Helleberg H, Roffel A, et al. Effect of semaglutide on the pharmacokinetics of metformin, warfarin, atorvastatin and digoxin in healthy subjects. Clinical Pharmacokinetics. 2017;56(11):1391-1401. DOI: 10.1007/s40262-017-0532-6
- Hooper A, Perez-Ruixo JJ, Sargent D, et al. GLP-1RA-induced delays in gastrointestinal motility: Predicted effects on coadministered drug absorption by PBPK analysis. Pharmacotherapy. 2025;45(1):e70007. DOI: 10.1002/phar.70007
- Krieger JP, et al. Clinical consequences of delayed gastric emptying with GLP-1 receptor agonists and tirzepatide. Journal of Clinical Endocrinology & Metabolism. 2025;110(1):1-12. DOI: 10.1093/jcem/dgae510
- Nauck MA, Meier JJ. Management of endocrine disease: are all GLP-1 agonists equal in the treatment of type 2 diabetes? European Journal of Endocrinology. 2019;181(6):R211-R234. DOI: 10.1530/EJE-19-0566
- de Heer J, Holst JJ. Sulfonylurea compounds uncouple the glucose dependence of the insulinotropic effect of glucagon-like peptide 1. Diabetes. 2007;56(2):438-443. DOI: 10.2337/db06-0903
- Blonde L, Umpierrez GE, Reddy SS, et al. American Association of Clinical Endocrinology clinical practice guideline: developing a diabetes mellitus comprehensive care plan. Endocrine Practice. 2022;28(10):923-1049. DOI: 10.1016/j.eprac.2022.08.002
- Chen Y, Li Z, Chen J, et al. Efficacy and safety of semaglutide combined with metformin in treating T2DM with overweight or obesity: a systematic review and meta-analysis. Frontiers in Pharmacology. 2024;15:1401207. DOI: 10.3389/fphar.2024.1401207
- Jorsal T, Rhee NA, Pedersen J, et al. Enteroendocrine K and L cells in healthy and type 2 diabetic individuals. Diabetologia. 2018;61(2):284-294. DOI: 10.1007/s00125-017-4450-9
- Gariani K, Menzies KJ, Ryu D, et al. Eliciting the mitochondrial unfolded protein response by nicotinamide adenine dinucleotide repletion reverses fatty liver disease. Hepatology. 2016;63(4):1190-1204. DOI: 10.1002/hep.28245
- Roth AB, et al. Pharmacology and potential implications of nicotinamide adenine dinucleotide precursors. Antioxidants. 2021;10(9):1637. DOI: 10.3390/antiox10091637
- Meier JJ. GLP-1 receptor agonists for individualized treatment of type 2 diabetes mellitus. Nature Reviews Endocrinology. 2012;8(12):728-742. DOI: 10.1038/nrendo.2012.140
- American Society of Anesthesiologists. Consensus-based guidance on preoperative management of patients on GLP-1 receptor agonists. ASA Guidelines. 2023. DOI: 10.1097/ALN.0000000000004801
- Sikiric P, Seiwerth S, Rucman R, et al. Pentadecapeptide BPC 157 and the central nervous system. Neural Regeneration Research. 2022;17(3):482-487. DOI: 10.4103/1673-5374.320969
- Sikiric P, et al. Pentadecapeptide BPC 157 interactions with adrenergic and dopaminergic systems in mucosal protection in stress. Digestive Diseases and Sciences. 1997;42(3):661-671. DOI: 10.1023/A:1018869620601
- Sikiric P, Seiwerth S, Rucman R, et al. Stable gastric pentadecapeptide BPC 157: novel therapy in gastrointestinal tract. Current Pharmaceutical Design. 2011;17(16):1612-1632. DOI: 10.2174/138161211796197115
- Garaci E, Pica F, Serafino A, et al. Thymosin alpha 1 and pertussis: a comprehensive review of the literature. World Journal of Hepatology. 2020;12(5):67-76. DOI: 10.4254/wjh.v12.i5.67
- Tuttle KR, Lakshmanan MC, Rayner B, et al. Dulaglutide versus insulin glargine in patients with type 2 diabetes and moderate-to-severe chronic kidney disease. Diabetes Care. 2018;41(10):2123-2130. DOI: 10.2337/dc18-0935
- Nuffer WA, Trujillo JM. Liraglutide: a new option for the treatment of obesity. Pharmacotherapy. 2015;35(10):926-934. DOI: 10.1002/phar.1639
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. New England Journal of Medicine. 2022;387(3):205-216. DOI: 10.1056/NEJMoa2206038
- Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. New England Journal of Medicine. 2021;384(11):989-1002. DOI: 10.1056/NEJMoa2032183
- Drucker DJ. Mechanisms of action and therapeutic application of glucagon-like peptide-1. Cell Metabolism. 2018;27(4):740-756. DOI: 10.1016/j.cmet.2018.03.001
- Nair KS, et al. The fascinating interplay between growth hormone, insulin-like growth factor-1, and insulin. International Journal of Molecular Sciences. 2024;25(4):2208. DOI: 10.3390/ijms25042208
- Murphy MG, Plunkett LM, Gertz BJ, et al. MK-677, an orally active growth hormone secretagogue, reverses diet-induced catabolism. Journal of Clinical Endocrinology & Metabolism. 1998;83(2):320-325. DOI: 10.1210/jcem.83.2.4539
- Sievertsen HJ, et al. Warfarin and GLP-1 receptor agonist interaction effects on time in therapeutic range. Blood. 2024;144(Supplement 1):5579. DOI: 10.1182/blood-2024-211293
- Buse JB, Wexler DJ, Tsapas A, et al. 2019 update to: Management of hyperglycemia in type 2 diabetes. Diabetologia. 2020;63(2):221-228. DOI: 10.1007/s00125-019-05039-w
- Aroda VR, Rosenstock J, Terauchi Y, et al. PIONEER 1: randomized clinical trial of the efficacy and safety of oral semaglutide monotherapy. Diabetes Care. 2019;42(9):1724-1732. DOI: 10.2337/dc19-0749
- Beglinger C, et al. Effect of oral semaglutide on the pharmacokinetics of thyroxine after dosing of levothyroxine. Expert Opinion on Drug Metabolism & Toxicology. 2021;17(9):1069-1078. DOI: 10.1080/17425255.2021.1955856
- Rudolph V, et al. Drug-drug interactions between glucagon-like peptide 1 receptor agonists and oral medications: a systematic review. Drug Safety. 2024;47(5):439-450. DOI: 10.1007/s40264-023-01392-3
- Marso SP, Daniels GH, Nissen SE, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. New England Journal of Medicine. 2016;375(4):311-322. DOI: 10.1056/NEJMoa1603827
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. New England Journal of Medicine. 2023;389(24):2221-2232. DOI: 10.1056/NEJMoa2307563
- Mathieu C, et al. The impact of tirzepatide and glucagon-like peptide 1 receptor agonists on oral hormonal contraception. Contraception. 2024;109:110209. DOI: 10.1016/j.contraception.2023.110209
- Skugor M, et al. Reducing or discontinuing insulin or sulfonylurea when initiating a GLP-1 agonist. Cleveland Clinic Journal of Medicine. 2024;91(6):362-370. DOI: 10.3949/ccjm.91a.23050
- Goldenberg RM, Steen O. Semaglutide: Review and place in therapy for adults with type 2 diabetes. Canadian Journal of Diabetes. 2019;43(2):136-145. DOI: 10.1016/j.jcjd.2018.05.008
- Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. New England Journal of Medicine. 2012;366(26):2443-2454. DOI: 10.1056/NEJMoa1200690
- Abushamat LA, et al. Semaglutide: double-edged sword with risks and benefits. Journal of Clinical Medicine. 2025;14(3):PMC11790292. DOI: 10.3390/jcm14030726
- Elrick H, et al. Glucagon-like peptide-1 receptor agonists in the perioperative period: implications for the anaesthesiologist. Indian Journal of Anaesthesia. 2024;68(1):24-30. DOI: 10.4103/ija.ija_991_23
- Jezek P, et al. Could overt diabetes be triggered by abuse of SARMs and GH secretagogues? Medicina. 2022;58(7):959. DOI: 10.3390/medicina58070959
- Johannsson G, et al. Growth hormone therapy and CYP enzyme activity. Journal of Clinical Pharmacology. 2003;43(11):1265-1271. DOI: 10.1177/0091270003258651