Executive Summary
Every peptide carries a unique side effect profile shaped by its mechanism of action, dosing schedule, and route of administration. This encyclopedia catalogs the adverse effects of every major peptide class - from GLP-1 receptor agonists and growth hormone secretagogues to healing peptides, nootropics, immune modulators, and cosmetic compounds - along with evidence-based strategies for managing each one.
Key Takeaways
- Every peptide carries a unique side effect profile shaped by its mechanism of action, dosing schedule, and route of administration.
- Peptide therapy has grown from a niche research interest into a mainstream medical and wellness tool.
- Semaglutide and tirzepatide now rank among the most prescribed drugs in the United States.
- Growth hormone secretagogues like CJC-1295/Ipamorelin and MK-677 are widely used for anti-aging and body composition goals.
- Healing peptides such as BPC-157 and TB-500 have built enormous followings for injury recovery.
Peptide therapy has grown from a niche research interest into a mainstream medical and wellness tool. Semaglutide and tirzepatide now rank among the most prescribed drugs in the United States. Growth hormone secretagogues like CJC-1295/Ipamorelin and MK-677 are widely used for anti-aging and body composition goals. Healing peptides such as BPC-157 and TB-500 have built enormous followings for injury recovery. And nootropic peptides like Selank are gaining traction for cognitive and mood support.
Yet side effects remain the primary reason people abandon peptide protocols early. Understanding what to expect - and what to do about it - makes the difference between a successful protocol and a frustrating one. Research shows that up to 80% of GLP-1 users who experience initial nausea see it resolve within 8 to 12 weeks (Wilding et al., 2021, DOI: 10.1056/NEJMoa2032183). Growth hormone peptide users who monitor fasting glucose and manage water retention tend to stay on protocol far longer than those who don't.
This guide is organized by peptide class so you can jump directly to the compounds you're using. Each section covers the mechanism behind each side effect, its frequency based on clinical data where available, and practical management strategies. We also cover injection site reactions (which apply to nearly all injectable peptides), drug interactions, special population considerations, and clear guidelines for when to stop a peptide and seek medical attention.
Safety First
This encyclopedia is for educational purposes only. It does not replace medical advice. Always work with a qualified healthcare provider before starting, adjusting, or stopping any peptide protocol. If you experience severe or unexpected symptoms, contact your provider or seek emergency care immediately.

Figure 1: Peptide side effect landscape organized by compound class and severity level
For those new to peptides, we recommend starting with our Peptide Therapy Beginner's Guide before diving into this side effect reference. And for monitoring recommendations, see our Peptide Blood Work Monitoring Guide.
Understanding Peptide Side Effects: Why They Happen
Before examining specific compounds, it helps to understand why peptides cause side effects in the first place. Unlike small-molecule drugs that often have off-target binding across dozens of receptors, peptides tend to be highly specific to their target receptor. This means their side effects usually stem from the intended mechanism of action rather than random off-target activity.
Mechanism-Driven Side Effects
GLP-1 receptor agonists cause nausea because they're supposed to slow gastric emptying and signal satiety to the brain. That's literally how they reduce appetite and produce weight loss. The nausea isn't a bug - it's an overexpression of the intended effect. As the body's GLP-1 receptors downregulate and adapt, the nausea fades while the therapeutic appetite reduction persists (Nauck et al., 2021, DOI: 10.1016/S0140-6736(21)00214-3).
Growth hormone secretagogues cause water retention because growth hormone promotes sodium and water reabsorption in the kidneys. They cause carpal tunnel symptoms because elevated IGF-1 triggers soft tissue swelling. These are pharmacological effects of successfully raising GH and IGF-1, not errors in the drug design (Nass et al., 2008, DOI: 10.7326/0003-4819-149-9-200811040-00003).
This distinction matters because mechanism-driven side effects are predictable, dose-dependent, and often manageable through titration and supportive strategies. They also tend to diminish as the body adapts - a phenomenon called tachyphylaxis.
Contamination and Quality-Related Side Effects
A separate category of side effects comes not from the peptide itself but from contaminants in the product. A 2023 analysis found peptide products contaminated with heavy metals including arsenic and lead - sometimes at ten times the acceptable limit for injectable drugs. Bacterial endotoxins capable of causing life-threatening sepsis were also detected in some products.
This is why sourcing matters enormously. Side effects like unexpected fevers, severe injection site infections, and systemic inflammatory reactions are almost always contamination-related rather than peptide-related. Working with a reputable provider and using properly tested, pharmaceutical-grade compounds eliminates this entire category of risk.
Dose-Response Relationship
Nearly every peptide side effect follows a dose-response curve. Higher doses produce more pronounced effects. This is why slow, gradual dose titration is the single most effective strategy for minimizing side effects across all peptide classes. Starting low and increasing gradually gives the body time to adapt at each level.
| Side Effect Driver | Examples | Management Approach |
|---|---|---|
| Mechanism-driven (on-target) | GLP-1 nausea, GH water retention, MK-677 hunger increase | Dose titration, timing adjustments, supportive care |
| Off-target pharmacology | CJC-1295 histamine flush, Melanotan II mole darkening | Antihistamines, dose reduction, compound switching |
| Product contamination | Injection site infections, fever, systemic inflammation | Source verification, sterility testing, provider change |
| Injection technique | Bruising, lipohypertrophy, pain | Site rotation, proper technique, needle gauge selection |
| Individual sensitivity | Allergic reactions, idiosyncratic responses | Trial doses, medical supervision, alternative compounds |
The Adaptation Timeline
Most peptide side effects follow a predictable arc. They peak during the first 1 to 2 weeks at a new dose, then gradually diminish over 4 to 8 weeks as receptor sensitivity adjusts. Data from the STEP clinical trials showed that 80% of semaglutide users who experienced initial nausea reported significant improvement by weeks 8 to 12 (Wilding et al., 2021, DOI: 10.1056/NEJMoa2032183). Similar adaptation patterns occur with GH peptide water retention, nootropic peptide headaches, and injection site reactions.
Understanding this timeline prevents premature discontinuation. Many people quit a peptide during the adaptation window and miss out on the benefits that would have come once side effects resolved.
General Injection Site Reactions
Injection site reactions are the most universal side effect across all injectable peptides, affecting an estimated 30 to 50% of users at some point during their protocol. The good news: they're almost always mild, self-limiting, and preventable with proper technique.
Types of Injection Site Reactions
Redness and Erythema
Mild redness around the injection site is the most common reaction, typically appearing within minutes and resolving within 1 to 24 hours. It results from local vasodilation and minor inflammatory response to the injection itself - not necessarily to the peptide. This occurs with virtually every injectable substance, including saline.
Swelling and Induration
A small, firm bump (wheal) at the injection site is normal and usually resolves within 24 to 48 hours. Persistent nodules that last longer may indicate injection technique issues - particularly injecting too shallowly (intradermally rather than subcutaneously) or injecting too quickly.
Bruising
Bruising occurs when the needle nicks a small blood vessel during injection. It's more common in areas with denser vascular networks and in people taking blood thinners, fish oil, or aspirin. While cosmetically annoying, bruising is medically insignificant.
Itching and Histamine Response
Some peptides - particularly CJC-1295 and other GHRH analogs - can directly trigger mast cell degranulation at the injection site, releasing histamine. This produces localized itching, redness, and sometimes a raised welt that looks like a bug bite. This is a pharmacological effect (not an immune-mediated allergy) and typically diminishes with continued use as local mast cells become depleted.
Pain During Injection
Sharp or stinging pain during injection usually relates to technique, needle gauge, solution pH, or injection speed rather than the peptide itself. Bacteriostatic water reconstitution produces a neutral pH that minimizes stinging. Some peptides (particularly those reconstituted at higher concentrations) can produce more discomfort.
Lipohypertrophy and Lipoatrophy
Repeated injection into the same site can cause lipohypertrophy (fatty lumps) or lipoatrophy (indentations from fat loss). These are long-term consequences of poor site rotation. Research shows that proper rotation reduces lipohypertrophy odds by 6- to 8-fold (Famulla et al., 2014, DOI: 10.2337/dc13-2513).
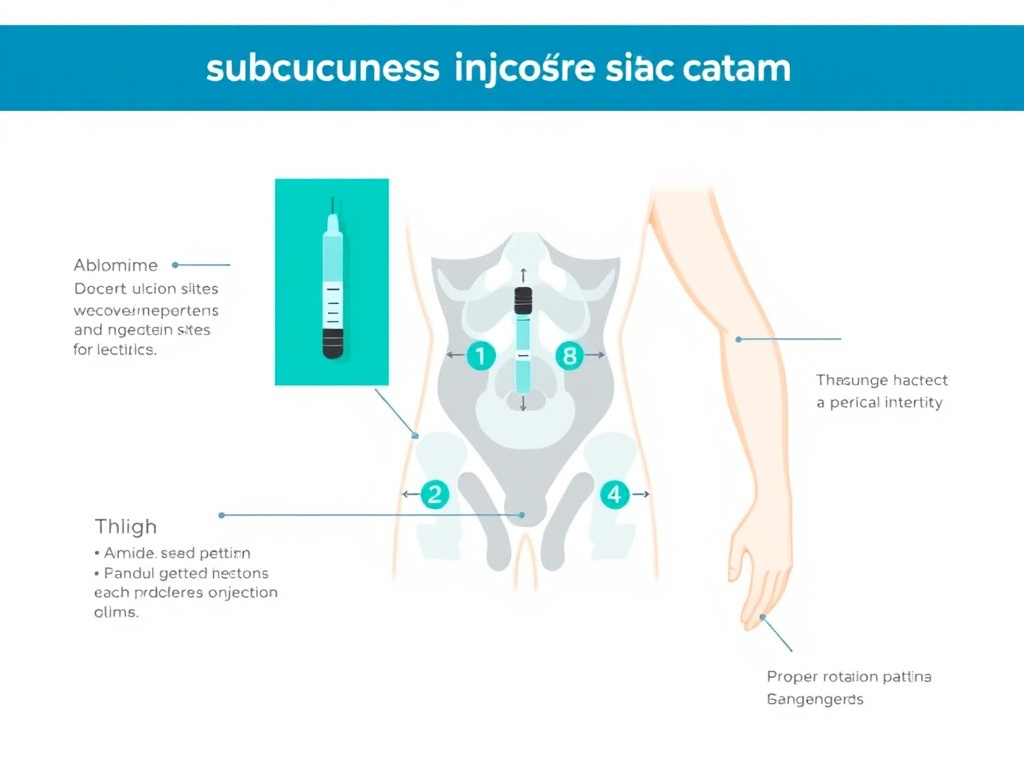
Figure 2: Recommended injection sites and rotation pattern for subcutaneous peptide administration
Injection Site Reaction Prevention Protocol
Best Practices for Minimizing Injection Site Reactions
- Use proper needle gauge: 27 to 31 gauge insulin syringes are standard for subcutaneous peptide injections. Smaller gauges mean less tissue trauma.
- Inject slowly: Take 10 to 15 seconds per injection. Rapid injection increases local pressure and inflammation.
- Allow the peptide to reach room temperature: Injecting cold solution straight from the refrigerator increases pain and local reaction.
- Rotate sites systematically: Use a rotation pattern across abdomen (avoiding 2 inches around the navel), outer thighs, and upper arms. Keep at least 1 inch between injection sites.
- Clean the site with alcohol: Wait for the alcohol to dry completely before injecting. Injecting through wet alcohol carries it into the tissue and causes stinging.
- Don't rub the injection site: Gentle pressure with a clean cotton ball is fine, but rubbing disperses the peptide and increases bruising.
- Pinch a skin fold: For subcutaneous injections, pinching a fold of skin ensures you're in the subcutaneous fat layer rather than injecting intradermally or intramuscularly.
When Injection Site Reactions Signal a Problem
While most injection site reactions are benign, some warrant medical attention:
- Expanding redness with warmth and increasing pain over 24 to 48 hours suggests infection (cellulitis). This requires medical evaluation and possibly antibiotics.
- Abscess formation - a painful, fluctuant collection of pus - requires drainage and antibiotics.
- Generalized hives or itching beyond the injection site suggests a systemic allergic reaction. Discontinue the peptide and contact your provider.
- Any signs of anaphylaxis - throat swelling, difficulty breathing, rapid heartbeat, dizziness - require immediate emergency care (call 911).
Histamine Reactions: A Special Case
CJC-1295 (both DAC and non-DAC versions) is particularly known for causing histamine-type reactions at injection sites. The GHRH analog structure directly triggers mast cell degranulation as a pharmacological property of the molecule. This isn't a true allergy and doesn't indicate the peptide is unsafe for you - but it can be uncomfortable.
Management strategies for histamine reactions include:
- Taking an H1 antihistamine (cetirizine 10mg or loratadine 10mg) 30 to 60 minutes before injection
- Adding an H2 blocker (famotidine 20mg) for more complete histamine suppression
- Applying a cold compress to the injection site for 5 minutes before injection
- Injecting very slowly over 15 to 20 seconds
- Nutritional support: vitamin C (1000mg daily) acts as a natural antihistamine; vitamin B6 supports DAO enzyme synthesis for histamine breakdown; magnesium glycinate (400mg daily) stabilizes mast cells
For a full breakdown of the CJC-1295/Ipamorelin combination and its side effects, see our dedicated CJC-1295/Ipamorelin product page.
GLP-1 Receptor Agonist Side Effects
GLP-1 receptor agonists - including semaglutide, tirzepatide, liraglutide, dulaglutide, and exenatide - share a common side effect profile dominated by gastrointestinal symptoms. These are the most extensively studied peptides, with safety data from trials enrolling tens of thousands of participants.
Gastrointestinal Side Effects
GI effects are the hallmark side effects of GLP-1 therapy. They stem directly from the mechanism of action: GLP-1 receptor activation slows gastric emptying, increases satiety signaling in the brain, and reduces appetite. When these effects are stronger than the body is adapted to, nausea, vomiting, diarrhea, and constipation result.
Nausea
Nausea is the single most reported GLP-1 side effect. Incidence rates vary by compound and dose:
| Compound | Nausea Rate (Treatment) | Nausea Rate (Placebo) | Source Trial |
|---|---|---|---|
| Semaglutide 2.4mg | 44.2% | 17.4% | STEP 1 |
| Tirzepatide 15mg | 31.0% | 9.5% | SURMOUNT-1 |
| Tirzepatide 10mg | 24.6% | 9.5% | SURMOUNT-1 |
| Tirzepatide 5mg | 24.0% | 9.5% | SURMOUNT-1 |
| Liraglutide 3.0mg | 39.3% | 14.7% | SCALE |
| Dulaglutide 1.5mg | 21.1% | 7.0% | AWARD trials |
| Exenatide 2mg weekly | 20.0% | 7.0% | DURATION trials |
Key patterns from the clinical data:
- Nausea peaks during the first 1 to 2 weeks at each new dose level
- It's most intense during dose escalation periods
- Approximately 80% of patients who experience nausea report significant improvement by weeks 8 to 12 on a stable dose
- Only 3 to 7% of trial participants discontinued due to GI side effects
- Severity is dose-dependent - slower titration reduces both incidence and severity
Nausea Management Protocol
- Eat smaller, more frequent meals - 5 to 6 small meals rather than 2 to 3 large ones
- Avoid high-fat, greasy foods - these slow gastric emptying further
- Stay hydrated - small, frequent sips throughout the day
- Ginger - ginger tea, ginger chews, or 250mg ginger capsules before meals have antiemetic properties (Lete & Allue, 2016, DOI: 10.4137/IMI.S36273)
- Evening dosing - inject in the evening so peak nausea occurs during sleep
- Strict titration compliance - never skip dose steps; follow the prescribed escalation schedule
- Peppermint - peppermint tea or peppermint oil capsules can soothe the stomach
- Ondansetron (Zofran) - prescription antiemetic for severe cases; discuss with your provider
Vomiting
Vomiting rates range from 5 to 25% depending on the compound and dose. It follows the same pattern as nausea - worst during titration, improving with adaptation. Persistent vomiting beyond 3 to 4 weeks at the same dose warrants medical evaluation and potential dose reduction.
Diarrhea
Diarrhea affects 8 to 20% of GLP-1 users. A systematic review and meta-analysis found tirzepatide carries slightly higher diarrhea risk (RR 1.81 to 2.18 vs. placebo) compared to semaglutide (RR 1.66 to 1.80 vs. placebo) (He et al., 2024, DOI: 10.1016/j.pharmthera.2024.108650). Management includes staying hydrated, avoiding dairy and high-fiber foods during active episodes, and using loperamide for acute relief if needed.
Constipation
Constipation affects 5 to 15% of GLP-1 users and is somewhat paradoxical given that diarrhea also occurs. The slowed gastric motility that reduces appetite can also slow colonic transit. Management includes adequate water intake (aim for 80 to 100 oz daily), fiber supplementation (psyllium husk), magnesium citrate (400 to 600mg at bedtime), and regular physical activity.
Abdominal Pain
Mild to moderate abdominal discomfort occurs in 5 to 12% of users. It's usually related to altered gastric motility and resolves with adaptation. Severe, persistent abdominal pain - especially pain that radiates to the back - requires prompt medical evaluation to rule out pancreatitis.

Figure 3: Typical GLP-1 GI side effect timeline showing peak intensity during dose escalation and progressive adaptation
Gallbladder and Biliary Disease
This is one of the more serious GLP-1 side effects and deserves careful attention. A large meta-analysis of 76 randomized clinical trials found GLP-1 receptor agonist use was associated with significantly increased risk of gallbladder or biliary diseases (relative risk 1.37; 95% CI 1.23 to 1.52). Specifically (He et al., 2022, DOI: 10.1001/jamainternmed.2022.0338):
- Cholelithiasis (gallstones): RR 1.27 (95% CI 1.10 to 1.47)
- Cholecystitis (gallbladder inflammation): RR 1.36 (95% CI 1.14 to 1.62)
- Biliary disease overall: RR 1.55 (95% CI 1.08 to 2.22)
The risk is dose-dependent and duration-dependent. Higher doses showed more risk (RR 1.56 at higher doses vs. RR 0.99 at lower doses). Rapid weight loss itself is an independent risk factor for gallstone formation because it increases cholesterol saturation of bile. This means some of the gallbladder risk attributed to GLP-1s may actually be a consequence of the weight loss itself rather than a direct drug effect.
Gallbladder Risk Mitigation
- Maintain adequate fat intake (don't go ultra-low-fat) to keep bile flowing regularly
- Stay well hydrated
- Report any right upper quadrant pain, especially after meals, to your provider
- Patients with a history of gallbladder disease should be monitored more closely
- Ursodiol (ursodeoxycholic acid) may be prescribed prophylactically in high-risk patients
Pancreatitis Risk
The GLP-1/pancreatitis question has been extensively studied. Early case reports raised concern, but large-scale trials have been reassuring. The LEADER trial with liraglutide (over 9,000 patients, median follow-up 3.8 years) showed pancreatitis rates of 0.4% in the treatment group versus 0.5% in the placebo group - no increased risk (Marso et al., 2016, DOI: 10.1056/NEJMoa1603827). The SUSTAIN and STEP programs with semaglutide showed similarly low rates.
A meta-analysis by Monami et al. found no significant increase in pancreatitis or pancreatic cancer with GLP-1 receptor agonists (Monami et al., 2017, DOI: 10.1016/j.diabres.2017.04.010). However, patients with a history of pancreatitis should use GLP-1 drugs with caution, and anyone developing severe persistent abdominal pain radiating to the back should stop the medication and seek immediate evaluation.
Thyroid Concerns: Medullary Thyroid Carcinoma
All GLP-1 receptor agonists carry a boxed warning about medullary thyroid carcinoma (MTC) based on rodent studies showing C-cell tumors in rats and mice at supraphysiologic doses. However, human thyroid C-cells have far fewer GLP-1 receptors than rodent C-cells, and the clinical relevance of these animal findings remains uncertain.
The LEADER trial (9,340 patients, 3.8 years) found no increased thyroid cancer rate. A Danish population-based study examining over 145,000 GLP-1 users found no association with thyroid cancer (Bea et al., 2024, DOI: 10.1001/jamainternmed.2024.0550). Despite this reassurance, GLP-1 drugs remain contraindicated in patients with:
- Personal or family history of medullary thyroid carcinoma (MTC)
- Multiple Endocrine Neoplasia syndrome type 2 (MEN2)
Gastroparesis vs. Delayed Gastric Emptying
GLP-1 drugs slow gastric emptying as part of their mechanism of action. This is not the same as causing gastroparesis. Clinical gastroparesis is a chronic condition involving structural or functional impairment of gastric motility. The delayed gastric emptying from GLP-1 agonists is dose-dependent, reversible upon discontinuation, and part of the intended pharmacology.
That said, people with pre-existing slow gastric motility may experience more pronounced symptoms. Cases of severe, prolonged gastroparesis-like symptoms during GLP-1 use are rare and typically resolve after stopping the medication. If you experience persistent vomiting, severe bloating, or inability to keep food down, consult your healthcare provider.
Other GLP-1 Side Effects
| Side Effect | Incidence | Notes |
|---|---|---|
| Headache | 10-15% | Usually resolves within first 2 weeks; stay hydrated |
| Fatigue | 5-11% | May relate to reduced caloric intake; ensure adequate protein |
| Dizziness | 5-8% | Often related to dehydration or blood pressure changes |
| Injection site reactions | 3-10% | Tirzepatide has higher rates than semaglutide |
| Hair thinning | 3-5% | Likely related to rapid weight loss and caloric deficit, not a direct drug effect |
| GERD/acid reflux | 5-8% | Related to slowed gastric emptying; avoid eating close to bedtime |
| Hypoglycemia | Rare (without sulfonylurea) | Much more common when combined with insulin or sulfonylureas |
| Acute kidney injury | Rare | Usually secondary to severe dehydration from vomiting/diarrhea |
For the complete GLP-1 Side Effects Management Guide, including dose-specific titration protocols, see our dedicated report. You can also explore our GLP-1 research hub and use the dosing calculator for personalized titration guidance.
Growth Hormone Secretagogue Side Effects
Growth hormone secretagogues - including CJC-1295/Ipamorelin, MK-677 (Ibutamoren), sermorelin, tesamorelin, GHRP-2, GHRP-6, and hexarelin - share a common set of class-wide side effects driven by elevated growth hormone (GH) and insulin-like growth factor 1 (IGF-1). However, each compound has unique characteristics that influence its side effect profile.
Class-Wide Side Effects of GH Elevation
Water Retention and Edema
Growth hormone promotes sodium and water reabsorption in the kidneys through direct effects on renal tubules and indirect effects via IGF-1. This is the most common side effect across all GH secretagogues, typically presenting as:
- Puffy face, especially around the eyes in the morning
- Swollen fingers and tight-fitting rings
- Ankle and lower leg edema
- 2 to 5 pounds of water weight gain
Water retention usually peaks during the first 2 to 4 weeks and then stabilizes or diminishes as the body adapts. It's dose-dependent - higher GH stimulation produces more water retention.
Water Retention Management
- Reduce sodium intake to under 2,300mg daily
- Increase potassium-rich foods (bananas, sweet potatoes, spinach, avocado)
- Stay well hydrated (paradoxically, drinking more water helps reduce retention)
- Regular cardiovascular exercise promotes fluid mobilization
- Elevate legs when sitting for extended periods
- Natural diuretics: dandelion root tea, hibiscus tea
- If severe, dose reduction is the most effective intervention
Carpal Tunnel Syndrome Symptoms
Elevated GH and IGF-1 cause soft tissue swelling throughout the body, including in the narrow carpal tunnel of the wrist. This compresses the median nerve, producing numbness, tingling, and pins-and-needles sensations in the thumb, index finger, and middle finger. Symptoms are typically worst at night or upon waking.
Carpal tunnel symptoms from GH peptides occur in approximately 10 to 20% of users at moderate to high doses. They are fully reversible upon dose reduction or discontinuation. Management includes wrist splints (especially at night), dose reduction, B6 supplementation (50 to 100mg daily, which may reduce nerve sensitivity), and anti-inflammatory measures (Nass et al., 2008, DOI: 10.7326/0003-4819-149-9-200811040-00003).
Joint Pain and Arthralgia
Joint pain affects 5 to 15% of GH peptide users. It results from IGF-1-driven changes in cartilage and synovial tissue. Commonly affected joints include the wrists, knees, and shoulders. Like other GH-related side effects, it's dose-dependent and usually resolves with dose adjustment.
Insulin Resistance and Blood Sugar Changes
Growth hormone is a counter-regulatory hormone to insulin - it opposes insulin's action and promotes glucose release from the liver. All GH secretagogues can shift blood glucose upward, but the degree varies substantially by compound.
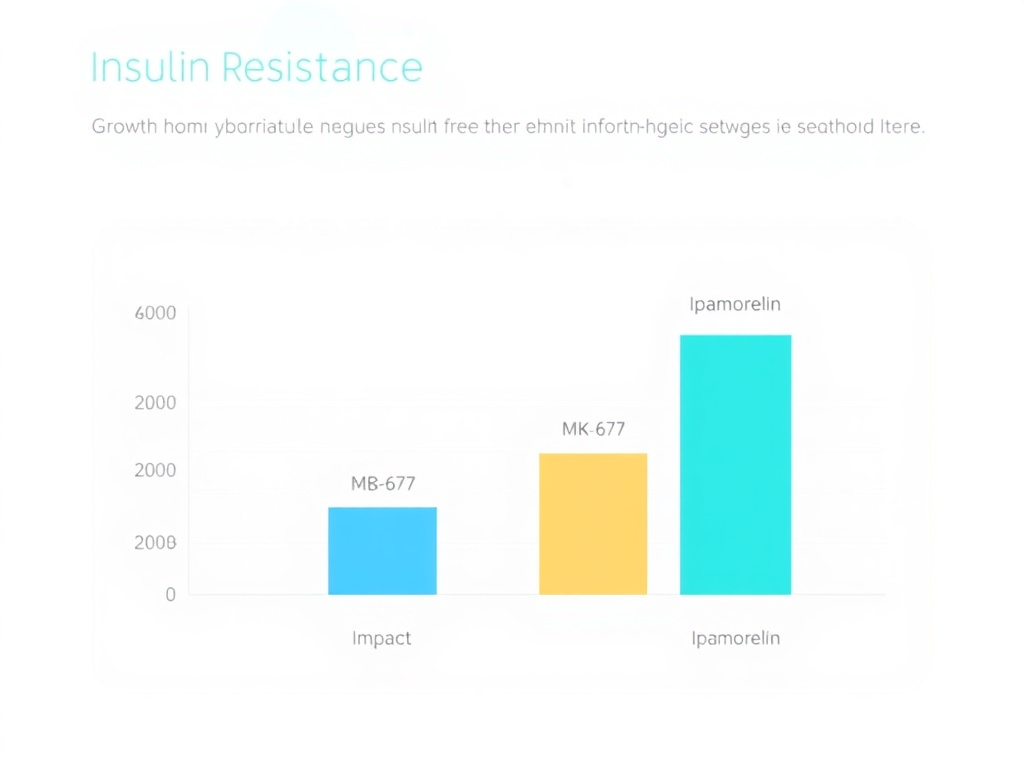
Figure 4: Comparative insulin resistance impact across GH secretagogue compounds
Compound-Specific Side Effects
CJC-1295 (with and without DAC)
CJC-1295 is a GHRH (growth hormone releasing hormone) analog. Its unique side effects beyond the GH class effects include:
- Histamine flush: CJC-1295 can directly trigger mast cell degranulation, causing a red, itchy flush at the injection site and sometimes systemically. This is the most commonly reported cause of histamine reactions in the peptide space. It's a pharmacological effect, not a true allergy.
- Vivid dreams: Commonly reported, likely related to GH's effects on sleep architecture. Many users actually consider this a positive effect.
- Headache: Occurs in approximately 10 to 15% of users during the first few weeks.
- DAC version: CJC-1295 with DAC (Drug Affinity Complex) has a longer half-life (6 to 8 days vs. 30 minutes), which means side effects may be more persistent but overall GH pulses are less physiological.
Ipamorelin
Ipamorelin is a ghrelin mimetic and GH secretagogue considered the most selective and well-tolerated of its class. Key differentiators:
- Minimal impact on cortisol - unlike GHRP-6 and GHRP-2, ipamorelin does not significantly raise cortisol levels
- Minimal impact on prolactin - again, unlike GHRP-6 and GHRP-2
- Modest appetite increase - less than GHRP-6 or MK-677
- Generally well-tolerated at standard doses (100 to 300mcg per injection)
- Side effects are primarily the GH class effects (water retention, possible carpal tunnel) and tend to be milder than with other secretagogues
MK-677 (Ibutamoren)
MK-677 is an oral ghrelin mimetic with a unique side effect profile that sets it apart from injectable GH peptides. It deserves special attention because it's widely available and often used without medical supervision.
Blood sugar and insulin effects: MK-677 has the most significant blood sugar impact of any GH secretagogue. In a study of healthy elderly subjects, ibutamoren 25mg/day increased fasting glucose by 25.3% and 26.9% above baseline at 2 and 4 weeks, respectively. Insulin sensitivity decreased across multiple measures (Nass et al., 2008, DOI: 10.7326/0003-4819-149-9-200811040-00003).
In an Alzheimer's disease trial, 15.4% of the MK-677 group developed elevated blood glucose compared to 4.6% in placebo. The dose was reduced from 25mg to 10mg in five patients (6%) due to fasting glucose exceeding 140 mg/dL, and discontinued in three due to ongoing hyperglycemia (Sevigny et al., 2008, DOI: 10.1212/01.wnl.0000339505.10934.ad).
A case report described new-onset diabetes triggered by MK-677 use in a previously healthy bodybuilder, with HbA1c rising from normal to diabetic range during use (Kinoshita & Homma, 2022, DOI: 10.1530/endoabs.86.P341).
| MK-677 Side Effect | Estimated Incidence | Severity |
|---|---|---|
| Increased appetite | 60-80% | Moderate - can be significant |
| Water retention / edema | 40-60% | Mild to moderate |
| Elevated fasting glucose | 15-25% | Moderate - monitor closely |
| Lethargy / fatigue | 20-30% | Mild to moderate |
| Joint pain | 10-20% | Mild |
| Carpal tunnel symptoms | 10-15% | Mild to moderate |
| Vivid dreams / sleep changes | 20-30% | Mild (often positive) |
| Muscle cramps | 5-10% | Mild |
MK-677 Blood Sugar Monitoring
Anyone using MK-677 should monitor fasting blood glucose regularly - ideally every 2 weeks for the first 2 months, then monthly. A continuous glucose monitor (CGM) provides even better data. If fasting glucose rises above 110 mg/dL or HbA1c increases by 0.3% or more, consider dose reduction or discontinuation. People with pre-diabetes, diabetes, or metabolic syndrome should use MK-677 with extreme caution or avoid it entirely. For more on monitoring, see our peptide blood work monitoring guide.
GHRP-6
GHRP-6 is an older GH secretagogue with a more pronounced side effect profile compared to ipamorelin:
- Intense hunger: GHRP-6 strongly stimulates ghrelin receptors, producing pronounced appetite increase that peaks 15 to 30 minutes after injection. This can make it difficult to use for people trying to lose fat.
- Cortisol elevation: Unlike ipamorelin, GHRP-6 can raise cortisol levels, which is undesirable for most users
- Prolactin elevation: GHRP-6 can raise prolactin, potentially causing symptoms like low libido or gynecomastia in susceptible males
- Blood sugar effects: Moderate impact on glucose metabolism
GHRP-2
GHRP-2 falls between ipamorelin and GHRP-6 in selectivity:
- Less appetite stimulation than GHRP-6 but more than ipamorelin
- Some cortisol and prolactin elevation (less than GHRP-6)
- Standard GH class side effects (water retention, carpal tunnel potential)
Hexarelin
Hexarelin is the most potent GH secretagogue but also has the most side effects:
- Strongest GH release of any GHRP
- Most pronounced cortisol and prolactin elevation
- Desensitization occurs faster than with other GHRPs - efficacy may decline after 4 to 6 weeks of continuous use
- Higher incidence of water retention and carpal tunnel symptoms due to stronger GH stimulation
Sermorelin
Sermorelin is a 29-amino acid analog of GHRH with a relatively clean side effect profile:
- Injection site reactions (redness, swelling) in approximately 15 to 20% of users
- Headache in approximately 10% of users during the first 2 weeks
- Flushing in approximately 5% of users
- Dizziness (occasional)
- Standard GH class effects (water retention, etc.) tend to be milder than with GHRPs due to more physiological GH release pattern
Tesamorelin
Tesamorelin is FDA-approved for HIV-associated lipodystrophy and has the most extensive clinical safety data of any GHRH analog:
- Injection site reactions: erythema (8.5%), pruritus (6.4%), pain (4.3%), and urticaria (2.1%) in clinical trials
- Arthralgia: 13.3% in clinical trials
- Peripheral edema: 6.1%
- Myalgia: 5.6%
- Carpal tunnel symptoms: 4.6%
- Blood glucose effects: modest increases in fasting glucose observed but less pronounced than MK-677 (Falutz et al., 2007, DOI: 10.1086/521564)
GH Peptide Side Effect Comparison Table
| Side Effect | Ipamorelin | CJC-1295 | MK-677 | GHRP-6 | GHRP-2 | Hexarelin |
|---|---|---|---|---|---|---|
| Water retention | Mild | Mild-Mod | Moderate | Moderate | Moderate | Mod-High |
| Hunger increase | Minimal | Minimal | High | High | Moderate | Moderate |
| Cortisol elevation | None | None | Minimal | Moderate | Mild | Moderate |
| Prolactin elevation | None | None | Minimal | Moderate | Mild | Mod-High |
| Blood sugar impact | Mild | Mild | High | Moderate | Moderate | Moderate |
| Carpal tunnel risk | Low | Low-Mod | Moderate | Moderate | Moderate | Mod-High |
| Histamine flush | Rare | Common | Rare | Occasional | Rare | Rare |
| Desensitization risk | Low | Low | Low | Moderate | Moderate | High |
This is exactly why the CJC-1295/Ipamorelin combination has become the most popular GH peptide stack - it pairs the GHRH analog (CJC-1295) with the most selective and well-tolerated GHRP (ipamorelin), giving strong GH release with the mildest side effect profile.
Healing Peptide Side Effects: BPC-157, TB-500, and Related Compounds
Healing peptides - primarily BPC-157 and TB-500 (Thymosin Beta-4) - are among the most widely used peptides for injury recovery, tissue repair, and gut healing. Their side effect profiles are specifically mild in preclinical research, but the lack of large-scale human clinical trials means our safety data is less strong than for GLP-1s or even GH peptides.
BPC-157 (Body Protection Compound-157)
BPC-157 is a 15-amino acid peptide derived from a protective protein found in human gastric juice. It has shown remarkable healing properties in animal studies across tendons, ligaments, muscles, the gut lining, the nervous system, and blood vessels. Its safety profile in preclinical studies has been exceptionally clean.
Known and Reported Side Effects
In animal studies (which represent the bulk of BPC-157 research), virtually no adverse effects have been reported even at doses many times higher than those used in human protocols. A 2024 systematic review noted that there seem to be little to no adverse effects reported in the preclinical literature (Kang et al., 2024, DOI: 10.1177/03635465241236962).
However, human data is extremely limited. The FDA classified BPC-157 as a Category 2 bulk drug substance in 2023, meaning it cannot be compounded by commercial pharmacies and there is insufficient evidence on whether it would cause harm to humans. Reported side effects from human use (largely anecdotal and from clinical practice reports) include:
| Side Effect | Estimated Frequency | Notes |
|---|---|---|
| Nausea (oral form) | 5-10% | Usually mild, transient |
| Headache | 5-10% | Most common in the first few days |
| Dizziness | 3-5% | Usually mild |
| GI discomfort (oral) | 5-8% | Bloating, mild cramping |
| Injection site reactions | 10-15% | Standard injection site redness/swelling |
| Fatigue | 3-5% | Usually resolves within first week |
| Hot/cold sensations | 2-5% | Related to vasodilatory effects |
Theoretical Concerns
The bigger question with BPC-157 isn't its observed side effects (which are minimal) but its theoretical risks based on mechanism of action:
- Angiogenesis promotion: BPC-157 promotes new blood vessel formation (angiogenesis). While this is beneficial for healing injuries, there's theoretical concern that it could support tumor vascularization in people with active cancer or precancerous conditions. No clinical cases of this have been documented, but the theoretical risk is real enough that most practitioners consider active cancer a contraindication (Sikiric et al., 2018, DOI: 10.2174/0929867324666170712101227).
- Growth factor modulation: BPC-157 affects multiple growth factor pathways (VEGF, FGF, EGF, NGF). While beneficial for healing, the long-term implications of chronically modulating these pathways are unknown.
- Product quality: Because BPC-157 is not FDA-approved and cannot be legally compounded by 503B pharmacies, it exists in a gray market where quality control varies enormously. Contamination with heavy metals, bacterial endotoxins, or incorrect peptide sequences is a real risk from unreliable sources.
BPC-157 Oral vs. Injectable Safety
BPC-157 can be administered orally (for gut-related issues) or subcutaneously (for systemic and musculoskeletal effects). The oral route avoids injection-related side effects entirely and may have a slightly different systemic effect profile. Some practitioners prefer oral BPC-157 for gut healing and injectable for musculoskeletal injuries. The side effect profiles are similar, with oral dosing showing slightly more GI-related effects (nausea, bloating) and injectable showing more injection site reactions.
TB-500 (Thymosin Beta-4)
TB-500 is a synthetic version of Thymosin Beta-4, a naturally occurring peptide involved in wound healing, tissue repair, and anti-inflammatory processes. Like BPC-157, its side effect profile appears mild, but human clinical data is limited.
Known and Reported Side Effects
| Side Effect | Estimated Frequency | Notes |
|---|---|---|
| Headache | 10-15% | Most common side effect; usually mild and transient |
| Nausea | 5-10% | Typically mild |
| Fatigue / lethargy | 5-10% | May relate to immune modulation effects |
| Injection site reactions | 10-15% | Standard redness, swelling |
| Dizziness / lightheadedness | 3-5% | Usually during first few administrations |
| Flu-like symptoms | 2-5% | Possible immune response to initial doses |
| Temporary increase in inflammation | 5-10% | Healing acceleration can temporarily increase local inflammation at injury site |
Theoretical Concerns for TB-500
- Cancer and angiogenesis: Like BPC-157, TB-500 promotes angiogenesis and cell migration, raising theoretical concerns about supporting tumor growth in people with active cancer. Thymosin Beta-4 has been found at elevated levels in certain tumor types, though whether it plays a causative role or is simply a bystander remains debated.
- Long-term safety unknown: With limited human studies, the short-term and long-term side effects of TB-500 remain largely unknown. Most protocols call for limited-duration use (4 to 12 weeks) rather than indefinite administration.
BPC-157 + TB-500 Stack Considerations
The combination of BPC-157 and TB-500 (sometimes called the "Wolverine Stack") is popular for injury recovery. When stacking these peptides:
- Side effects may be additive - particularly headache and nausea
- Start each peptide individually before combining to identify which compound causes any side effects
- The angiogenesis concern is theoretically amplified when both are used together
- No drug interaction data exists for this combination specifically
Figure 5: Healing peptide safety comparison showing reported side effect frequency and severity for BPC-157 and TB-500
Immune Peptide Side Effects: Thymosin Alpha-1 and Related Compounds
Thymosin Alpha-1 stands apart from many other peptides in this encyclopedia because it has genuine regulatory approval in over 35 countries and extensive clinical trial data spanning thousands of patients. Its side effect profile is among the most well-documented and favorable in the entire peptide space.
Thymosin Alpha-1
Thymosin Alpha-1 (Ta1) is a 28-amino acid peptide originally isolated from the thymus gland. The synthetic version (thymalfasin, brand name Zadaxin) has been used clinically since the 1990s for hepatitis B and C treatment, as a vaccine adjuvant, and for immune support in immunocompromised patients.
Clinical Safety Data
The safety profile of Thymosin Alpha-1 is remarkably clean. A 2020 review noted that Ta1 has demonstrated a very favorable toxicity profile in more than 3,000 individuals treated across clinical trials, including patients with hepatocellular carcinoma, non-small-cell lung cancer, melanoma, and hepatitis B and C (Costantini et al., 2020, DOI: 10.1586/17512433.2020.1814745).
| Side Effect | Incidence in Clinical Trials | Notes |
|---|---|---|
| Injection site reactions | 5-10% | Mild erythema, occasional pruritus |
| Mild flu-like symptoms | 3-5% | Low-grade fever, malaise; typically first few doses only |
| Fatigue | 2-5% | Usually transient |
| Muscle discomfort | 1-3% | Mild myalgia |
| Nausea | 1-2% | Rare and mild |
| Joint discomfort | Rare | Reported in isolated cases |
| Transient muscle atrophy | Very rare | Reported in case literature |
The key advantage of Thymosin Alpha-1 over other immune-modulating agents is its lack of the immunosuppressive side effects seen with drugs like cyclosporine or methotrexate. It modulates rather than suppresses immunity, meaning it helps restore balance rather than broadly dampening immune function.
Considerations and Cautions
- Autoimmune conditions: While Ta1 modulates rather than stimulates immunity in a single direction, people with active autoimmune conditions should use it with caution and under medical supervision. Enhanced immune function could theoretically exacerbate autoimmune flares in some individuals.
- Organ transplant recipients: Patients on immunosuppressive therapy for organ transplants should not use Ta1 without explicit approval from their transplant team, as enhanced immune function could theoretically increase rejection risk.
- Concurrent infections: Ta1 can sometimes cause a temporary worsening of symptoms as the immune system mounts a stronger response to an existing infection. This is similar to a Jarisch-Herxheimer reaction and typically resolves within days.
Other Immune-Related Peptides
LL-37 (Cathelicidin)
LL-37 is an antimicrobial peptide with direct pathogen-killing activity. Reported side effects include:
- Injection site pain (more pronounced than with most peptides due to its cationic nature)
- Local redness and swelling
- Potential for mast cell activation and histamine release
- Very limited human safety data outside of topical applications
Vasoactive Intestinal Peptide (VIP)
VIP is used primarily for chronic inflammatory response syndrome (CIRS) and mold illness. Side effects include:
- Nasal congestion (when administered intranasally)
- Diarrhea (VIP promotes intestinal secretion)
- Flushing and facial redness
- Hypotension (VIP is a vasodilator)
- Headache
Nootropic Peptide Side Effects: Selank, Semax, and Cognitive Peptides
Nootropic peptides - including Selank, Semax, Dihexa, and the Khavinson bioregulator peptides - represent a growing category of compounds used for cognitive enhancement, mood support, and neuroprotection. Most are administered intranasally, which creates a distinct side effect profile compared to injectable peptides.
Selank
Selank is a synthetic peptide derived from the naturally occurring immunomodulatory peptide tuftsin, with an added Gly-Pro sequence for metabolic stability. It's registered as an anxiolytic medication in Russia and has been studied for anxiety, cognitive function, and immune modulation.
Side Effect Profile
Selank has one of the mildest side effect profiles of any bioactive peptide. Russian clinical studies involving hundreds of patients have shown minimal adverse effects (Zozulya et al., 2001, DOI: 10.1016/S0165-6147(01)01920-0).
| Side Effect | Estimated Frequency | Notes |
|---|---|---|
| Nasal irritation | 10-15% | Related to intranasal route of administration, not the peptide itself |
| Mild headache | 5-10% | Usually transient, resolves within first week |
| Fatigue (paradoxical) | 3-5% | Some users report initial drowsiness; may relate to anxiolytic effect |
| Nasal congestion | 3-5% | Route-related rather than compound-related |
| Mild dizziness | 2-3% | Usually during first few days |
| Altered taste/smell | 1-3% | Temporary, from intranasal administration |
Selank does not produce sedation, cognitive impairment, dependence, or withdrawal effects - a significant advantage over benzodiazepines for anxiety management. It also doesn't affect motor coordination or reaction time.
Selank Safety Advantages
Unlike benzodiazepines and many other anxiolytics, Selank does not cause physical dependence, tolerance buildup, cognitive dulling, psychomotor impairment, or rebound anxiety upon discontinuation. These characteristics make it an appealing option for people seeking anxiety relief without the side effect burden of traditional medications. However, it remains unapproved in the US and lacks FDA-required clinical trial data.
Semax
Semax is a synthetic analog of ACTH (adrenocorticotropic hormone) fragments 4-10, designed for cognitive enhancement and neuroprotection. It's registered as a nootropic medication in Russia.
Side Effect Profile
| Side Effect | Estimated Frequency | Notes |
|---|---|---|
| Nasal irritation / dryness | 10-15% | Most common complaint; route-related |
| Headache | 5-10% | May relate to increased BDNF and neural activity |
| Mild anxiety or overstimulation | 3-5% | Dose-dependent; more common with N-Acetyl Semax variants |
| Insomnia | 3-5% | If dosed too late in the day; morning dosing recommended |
| Elevated mood (to excess) | 2-3% | Rare; more common with NA-Semax-Amidate variant |
| Hair thinning | Rare | Reported anecdotally; mechanism unclear; may relate to ACTH pathway modulation |
| Glucose changes in diabetics | Rare | ACTH-derived peptides can affect glucose metabolism |
The Semax variants (NA-Semax, NA-Semax-Amidate) have enhanced potency and longer duration, which can also mean more pronounced side effects. The standard Semax formulation is generally the mildest.
Dihexa
Dihexa is an oligopeptide with extremely potent nootropic effects - it's reported to be approximately 10 million times more potent than BDNF at activating the hepatocyte growth factor (HGF) receptor system. Its side effect profile is poorly characterized due to very limited human use data.
Known Concerns
- Headache: Commonly reported, likely related to intense neurotrophic activity
- Overstimulation: Some users report anxiety, racing thoughts, or difficulty sleeping
- Cancer risk (theoretical): HGF/c-Met pathway activation is associated with tumor growth, invasion, and metastasis in cancer biology. This is the most significant theoretical concern with dihexa. There is no clinical evidence of cancer promotion in humans from dihexa use, but the mechanistic concern is well-founded (McCague et al., 2015, DOI: 10.1124/mol.114.096636).
- Extremely limited safety data: Dihexa has not undergone clinical trials. Its use in humans is essentially experimental.
Dihexa Risk Assessment
Dihexa carries more theoretical risk than most peptides in this encyclopedia due to its extremely potent activation of growth factor pathways with known roles in cancer biology. While no cancer cases have been linked to dihexa use, the absence of evidence is not evidence of absence. People with any cancer history, family cancer predisposition, or elevated cancer biomarkers should avoid dihexa entirely. All users should understand they are taking an experimental compound with a poorly characterized safety profile.
Pinealon
Pinealon is a tripeptide (Glu-Asp-Arg) from the Khavinson bioregulator family, targeted at pineal gland function and sleep regulation. Side effects are minimal:
- Excessive drowsiness if dosed too high or at wrong time
- Vivid dreams (often considered a feature rather than a side effect)
- Mild headache (rare)
- Very limited formal safety data
Cerebrolysin
Cerebrolysin is a mixture of enzymatically treated porcine brain-derived peptides used for neurodegenerative conditions. It has more extensive clinical data than most nootropic peptides:
- Dizziness: 5-10%
- Headache: 5-8%
- Injection site reactions: common (administered IV or IM)
- Nausea: 3-5%
- Agitation: 2-5% (may relate to neurotrophic activity)
- Fever: rare
- Allergic reactions: rare but possible due to porcine-derived proteins
Figure 6: Nootropic peptide side effect comparison showing severity ratings across key categories
Cosmetic and Sexual Health Peptide Side Effects
This category covers peptides used primarily for aesthetic purposes (skin, hair, tanning) and sexual health, including Melanotan II, PT-141 (Bremelanotide), GHK-Cu, and cosmetic peptides like SNAP-8 and Argireline.
Melanotan II
Melanotan II is a synthetic melanocortin peptide that stimulates melanin production (tanning), reduces appetite, and increases sexual arousal. It has one of the most diverse side effect profiles of any peptide due to its broad melanocortin receptor activity.
Common Side Effects
| Side Effect | Incidence | Management |
|---|---|---|
| Nausea | 40% (dose-dependent) | Peaks 30-90 min post-injection; resolves in 2-4 hours; worse during first 3-5 days of loading; decreases significantly by week 2 |
| Facial flushing | 30-40% | Lasts 30-60 minutes; no treatment needed |
| Appetite suppression | 30-50% | Often considered a benefit; may concern underweight individuals |
| Spontaneous erections (males) | 20-30% | Related to melanocortin receptor activation; dose-dependent |
| Fatigue / lethargy | 15-25% | Usually transient; dose in evening |
| Injection site reactions | 10-15% | Standard management applies |
| Darkening of moles and freckles | Common | Expected pharmacological effect; may be permanent |
| New mole formation | 10-20% | Monitor all new moles; dermatological evaluation recommended |
| Yawning / stretching | 10-20% | CNS effect; typically harmless |
Serious Concerns
- Mole darkening and melanoma risk: Melanotan II darkens existing moles and can promote formation of new moles. At least 4 case reports describe melanomas emerging during or after Melanotan II use. While a 2021 review suggested the increased melanoma risk may be explained by UV-seeking behavior in Melanotan II users rather than the peptide itself, the combination of a melanocyte-stimulating peptide with UV exposure is concerning. Dermatological monitoring is strongly recommended for all Melanotan II users.
- Cardiovascular effects: Temporary blood pressure elevation and heart rate changes have been reported. People with uncontrolled hypertension or cardiovascular conditions should avoid Melanotan II.
- Hyperpigmentation: Skin discoloration beyond desired tanning can occur, particularly with doses exceeding 8 injections per month. This may not fully resolve upon discontinuation.
- Not FDA-approved: Melanotan II has not been approved by any regulatory agency for any indication.
PT-141 (Bremelanotide / Vyleesi)
PT-141 is a melanocortin receptor agonist approved by the FDA (as Vyleesi) for hypoactive sexual desire disorder (HSDD) in premenopausal women. It works through central nervous system pathways rather than vascular mechanisms (unlike PDE5 inhibitors).
Side Effects from Clinical Trials
| Side Effect | Incidence (Clinical Trials) | Notes |
|---|---|---|
| Nausea | 40.0% | Most common side effect; usually mild to moderate |
| Flushing | 20.3% | Facial/upper body warmth and redness |
| Injection site reactions | 13.2% | Bruising, pain, redness |
| Headache | 11.3% | Usually mild |
| Vomiting | 4.8% | Less common than nausea |
| Fatigue | 3.2% | Mild |
| Hot flashes | 2.7% | Related to melanocortin pathway activation |
| Dizziness | 2.2% | Usually transient |
| Nasal congestion | 2.1% | Mild |
| Skin darkening | Possible | Hyperpigmentation may occur, especially with frequent use (more than 8 doses per month); may not be reversible |
The FDA labeling notes that PT-141 can cause a temporary rise in blood pressure and drop in heart rate. It's not recommended for use more than once in 24 hours or more than 8 times per month.
GHK-Cu (Copper Peptide)
GHK-Cu is a naturally occurring tripeptide with a copper ion that plays roles in wound healing, collagen synthesis, and anti-aging. It's used both topically and by injection.
Side Effects
- Topical use: Generally very well tolerated. Rare reports of skin irritation, redness, or sensitivity, particularly in people with copper sensitivity. Allergic contact dermatitis is possible but uncommon.
- Injectable use: Injection site reactions (redness, mild pain), occasional headache, and a metallic taste have been reported. The copper content raises theoretical concerns about copper toxicity with high or prolonged dosing, though this has not been observed at standard doses.
- Hair shedding phase: When used for hair growth, some users experience an initial shedding phase as the hair cycle resets. This is temporary and typically precedes improved hair growth.
Cosmetic Peptides: SNAP-8, Argireline, Matrixyl
Topical cosmetic peptides used in anti-aging skincare have the mildest side effect profiles of any peptide category, since they're applied to the skin surface rather than injected or taken systemically.
- SNAP-8 (Acetyl Octapeptide-3): Extremely well tolerated topically. Rare skin irritation in sensitive individuals. No systemic side effects at topical concentrations.
- Argireline (Acetyl Hexapeptide-3): Very well tolerated. Occasional mild stinging or tingling upon application. Theoretical concern about facial muscle weakness with very high concentrations applied to thin-skinned areas, but this is rare at commercial product concentrations.
- Matrixyl (Palmitoyl Pentapeptide-4): Extremely well tolerated. Rare reports of skin sensitivity.
The main "side effect" concern with cosmetic peptides is not adverse reactions but rather product quality - some products contain insufficient peptide concentrations to produce any effect at all, which is more of an efficacy issue than a safety issue.
Anti-Aging and Longevity Peptide Side Effects
Longevity-focused peptides - including Epithalon, MOTS-c, Humanin, SS-31 (Elamipretide), and GHK-Cu - target fundamental aging mechanisms like telomere maintenance, mitochondrial function, and cellular senescence. Their safety profiles vary considerably based on how much clinical data is available.
Epithalon (Epitalon)
Epithalon is a synthetic tetrapeptide (Ala-Glu-Asp-Gly) based on the natural peptide epithalamin, developed by Professor Vladimir Khavinson for telomerase activation and anti-aging research.
Side Effect Profile
Epithalon has a remarkably mild reported side effect profile. The primary data comes from Russian clinical studies and widespread clinical use:
- Injection site reactions: Mild redness and occasional pain at the injection site (5-10% of users)
- Headache: Rare, usually mild and transient
- Drowsiness or altered sleep patterns: Some users report changes in sleep quality - often improvement, but occasionally initial drowsiness as melatonin production is modulated
- No serious adverse effects: Have been documented in the available literature
The primary theoretical concern with Epithalon relates to its mechanism: telomerase activation. While maintaining telomere length is associated with longevity and cellular health, telomerase is also activated in approximately 85-90% of human cancers. The relationship between therapeutic telomerase activation and cancer risk is an area of active research. Current evidence does not suggest Epithalon promotes cancer, but the theoretical concern warrants monitoring (Khavinson et al., 2003, DOI: 10.1007/s10522-005-1164-y).
MOTS-c
MOTS-c is a mitochondria-derived peptide that activates AMPK and improves metabolic function. Limited human data exists:
- Injection site reactions (standard)
- Mild GI discomfort reported in some cases
- Transient fatigue in some users
- Blood sugar lowering effect - could theoretically cause hypoglycemia in people on diabetes medications
- Overall considered to have a favorable safety profile based on limited data
SS-31 (Elamipretide)
SS-31 is a mitochondria-targeted peptide that has undergone clinical trials for mitochondrial myopathy (Barth syndrome) and heart failure. It has the most formal safety data of the longevity peptides:
- Injection site reactions: 15-20% (relatively common; pain and redness)
- Headache: 5-10%
- Nausea: 3-5%
- Generally well tolerated in clinical trials up to 48 weeks (Karaa et al., 2023, DOI: 10.1212/WNL.0000000000207015)
FOXO4-DRI
FOXO4-DRI is a senolytic peptide designed to selectively clear senescent ("zombie") cells. Safety data is extremely limited:
- No formal human clinical trial data
- Theoretical concerns about clearing cells that may serve protective functions
- Unknown long-term effects on tissue integrity
- Anecdotal reports include fatigue, mild flu-like symptoms (possibly from senescent cell clearance), and injection site reactions
- Considered highly experimental
Side Effect Frequency Matrix
The following matrix provides a quick-reference overview of side effect frequency across all major peptide classes. Frequencies are based on clinical trial data where available and clinical practice reports where formal trial data is lacking.
Most Common Peptide Side Effects by Incidence
Comprehensive Side Effect Frequency by Peptide Class
| Side Effect | GLP-1 RAs | GH Secretagogues | Healing Peptides | Nootropics | Immune Peptides | Cosmetic/Sexual |
|---|---|---|---|---|---|---|
| Nausea | 20-44% | 5-10% | 5-10% | 2-5% | 1-2% | 40% (MT-II/PT-141) |
| Vomiting | 5-25% | Rare | Rare | Rare | Rare | 5% (PT-141) |
| Diarrhea | 8-20% | Rare | Rare | Rare | Rare | Rare |
| Constipation | 5-15% | Rare | Rare | Rare | Rare | Rare |
| Headache | 10-15% | 10-15% | 5-15% | 5-10% | 3-5% | 11% (PT-141) |
| Injection site reactions | 3-10% | 10-20% | 10-15% | N/A (intranasal) | 5-10% | 10-15% |
| Water retention | Rare | 20-60% | Rare | Rare | Rare | Rare |
| Carpal tunnel | Rare | 10-20% | Rare | Rare | Rare | Rare |
| Blood sugar changes | Rare | 15-25% (MK-677) | Rare | Rare | Rare | Rare |
| Flushing | Rare | 5-15% (CJC) | 2-5% | Rare | Rare | 20-40% (MT-II/PT-141) |
| Fatigue | 5-11% | 20-30% (MK-677) | 3-10% | 3-5% | 2-5% | 3-5% |
| Dizziness | 5-8% | 3-5% | 3-5% | 2-3% | Rare | 2-5% |
| Increased appetite | Rare (opposite) | 60-80% (MK-677) | Rare | Rare | Rare | Rare (opposite for MT-II) |
| Skin changes | Rare | Rare | Rare | Rare | Rare | Common (MT-II) |
How to Read This Matrix
Frequencies represent the approximate percentage of users who experience each side effect based on available data. "Rare" means less than 2% or only case reports. For compounds without formal clinical trials, frequencies are estimated from clinical practice reports and should be considered approximate. Individual responses vary based on dose, duration, genetics, and concurrent medications.
Severity Rating by Peptide Class
Beyond frequency, severity matters. Here's how the major side effects rank by typical severity:
| Severity Level | GLP-1 RAs | GH Peptides | Healing | Nootropics | Immune |
|---|---|---|---|---|---|
| Usually mild (self-resolving) | Nausea, headache, fatigue | Water retention, vivid dreams | Headache, nausea, fatigue | Nasal irritation, mild headache | Injection site reactions |
| Moderate (may need intervention) | Persistent vomiting, diarrhea, constipation | Carpal tunnel, joint pain, MK-677 glucose rise | Injection site infection (contamination) | Insomnia, overstimulation | Flu-like symptoms |
| Serious (requires medical attention) | Gallbladder disease, pancreatitis (rare) | Diabetes onset (MK-677, rare) | Contamination-related sepsis | Growth pathway activation (Dihexa, theoretical) | Autoimmune flare (rare) |
Management Strategies by Symptom
This section provides practical, evidence-based management strategies organized by symptom rather than by peptide. Whatever compound you're using, if you're experiencing a particular side effect, the relevant management protocol will be here.
Nausea Management
Nausea is the single most common peptide side effect, occurring across GLP-1s, Melanotan II, PT-141, healing peptides, and occasionally GH peptides. The management approach is similar regardless of the cause.
Nausea Management Protocol (Universal)
Tier 1 - Lifestyle:
- Eat smaller, more frequent meals (5-6 per day)
- Avoid high-fat, fried, and heavily spiced foods
- Don't lie down immediately after eating
- Stay hydrated with small, frequent sips (not large volumes at once)
- Time injections for evening so peak nausea occurs during sleep
- Cold foods are often better tolerated than hot foods during nausea episodes
Tier 2 - Natural remedies:
- Ginger: tea, chews, or 250mg capsules before meals (Lete & Allue, 2016, DOI: 10.4137/IMI.S36273)
- Peppermint: tea or enteric-coated oil capsules
- Vitamin B6: 25-50mg three times daily
- Acupressure wristbands (Sea-Bands)
Tier 3 - Prescription (discuss with provider):
- Ondansetron (Zofran) 4-8mg as needed
- Promethazine 12.5-25mg (causes drowsiness)
- Metoclopramide (for GLP-1 related gastroparesis-like symptoms)
Water Retention Management
Primarily relevant for GH secretagogues but can occur with any peptide protocol.
- Reduce sodium: Limit to under 2,300mg daily; avoid processed foods, restaurant meals, and added table salt
- Increase potassium: Bananas, sweet potatoes, spinach, avocado, coconut water
- Drink more water: Counterintuitive but effective - adequate hydration signals the kidneys to release excess water. Aim for 80-100 oz daily.
- Exercise: Cardiovascular activity promotes lymphatic drainage and fluid mobilization
- Elevate limbs: Elevate legs above heart level for 15-20 minutes several times daily if ankle edema is present
- Natural diuretics: Dandelion root tea (300-400mg extract or 2-3 cups tea daily), hibiscus tea
- Compression garments: For persistent lower extremity edema
- Dose reduction: The most effective intervention if water retention is bothersome; reduce by 25-50% and reassess after 2 weeks
Headache Management
Headaches occur across nearly all peptide classes. Common causes include dehydration, blood pressure changes, neurotransmitter modulation, and histamine release.
- Hydration: Drink at least 64-80 oz of water daily; dehydration is the most common cause of peptide-related headaches
- Electrolytes: Add electrolyte mix (sodium, potassium, magnesium) to water, especially if also experiencing nausea or reduced food intake
- OTC analgesics: Acetaminophen (Tylenol) 500-1000mg is preferred over NSAIDs for peptide headaches; NSAIDs may interact with healing peptides' mechanisms
- Magnesium: 400-600mg magnesium glycinate daily can reduce headache frequency
- Sleep: Ensure adequate sleep; some peptides (especially nootropics) can disrupt sleep patterns, leading to headaches
- Dose adjustment: If headaches persist beyond 2 weeks, consider dose reduction
Injection Site Reaction Management
See the detailed section above on General Injection Site Reactions for complete management protocols.
Quick reference:
- Redness/swelling: Cold compress for 5-10 minutes; usually self-resolving in 24 hours
- Bruising: Arnica gel; avoid blood thinners, fish oil, and aspirin before injection if possible
- Itching/histamine: Cetirizine 10mg or loratadine 10mg 30-60 minutes pre-injection; famotidine 20mg for additional H2 blockade
- Pain: Allow solution to reach room temperature; inject slowly over 10-15 seconds; use 30-31 gauge needles
- Lipohypertrophy: Strict site rotation with at least 1 inch between sites
Blood Sugar Management (GH Peptides)
Blood Sugar Monitoring and Management for GH Peptides
- Baseline testing: Check fasting glucose and HbA1c before starting any GH peptide
- Regular monitoring: Fasting glucose every 2 weeks for the first 2 months, then monthly; HbA1c every 3 months
- CGM consideration: A continuous glucose monitor provides the best real-time data
- Dietary management: Lower carbohydrate intake, especially simple carbs; increase fiber; time carbs around exercise
- Exercise: Regular resistance training and cardio improve insulin sensitivity
- Berberine: 500mg twice daily can improve insulin sensitivity (natural intervention; discuss with provider)
- Metformin: Some providers prescribe low-dose metformin (500-1000mg) alongside GH peptides to counteract glucose effects (prescription required)
- Dose adjustment triggers: Fasting glucose consistently above 110 mg/dL or HbA1c increase of 0.3% or more warrants dose reduction or discontinuation
Carpal Tunnel Symptom Management
- Wrist splints: Wear at night to keep wrists in neutral position; this is the most effective non-pharmacological intervention
- B6 supplementation: 50-100mg pyridoxine daily may reduce nerve sensitivity (don't exceed 200mg daily due to peripheral neuropathy risk)
- Anti-inflammatory measures: Cold application, turmeric/curcumin (500-1000mg daily), omega-3 fatty acids
- Ergonomic adjustments: Modify workstation to reduce wrist strain
- Dose reduction: The most effective intervention; reduce GH peptide dose by 25-50%
- Discontinuation: Symptoms are fully reversible upon stopping the peptide; they typically resolve within 1-3 weeks
Fatigue and Lethargy Management
- Caloric adequacy: Ensure you're eating enough, especially on GLP-1 therapy where appetite is suppressed; aim for at least 1,200 calories daily minimum and adequate protein (0.7-1g per pound of lean body mass)
- Iron and B12: Rule out nutritional deficiencies; both are common with reduced food intake
- Sleep quality: Some peptides enhance sleep (GH peptides), others may disrupt it (nootropics dosed late); optimize timing
- Hydration and electrolytes: Dehydration and electrolyte imbalances (especially with GLP-1-related vomiting/diarrhea) cause fatigue
- Thyroid check: If fatigue persists, check thyroid function; some peptide protocols can influence thyroid axis
- Dose timing: Try moving injection to evening if morning fatigue is an issue
GI Issues Beyond Nausea (Diarrhea, Constipation, Bloating)
For diarrhea:
- Stay hydrated with electrolyte solutions
- BRAT diet (bananas, rice, applesauce, toast) during acute episodes
- Avoid dairy, caffeine, and high-fiber foods temporarily
- Loperamide (Imodium) for acute relief if needed
- Probiotics (Saccharomyces boulardii is specifically evidence-based for drug-induced diarrhea)
For constipation:
- Increase water intake to 80-100 oz daily
- Fiber supplementation: psyllium husk (Metamucil) 1-2 tablespoons daily
- Magnesium citrate: 400-600mg at bedtime (acts as osmotic laxative)
- Regular physical activity (walking 30 minutes daily)
- Prune juice or dried prunes
- Docusate (Colace) for stool softening if needed
For bloating:
- Eat slowly and chew thoroughly
- Avoid carbonated beverages
- Reduce gas-producing foods (beans, cruciferous vegetables, onions) temporarily
- Digestive enzymes with meals
- Simethicone (Gas-X) for acute relief
- Peppermint oil capsules (enteric-coated)
Figure 7: Side effect management decision flowchart - from self-care to medical intervention
When to Stop a Peptide and Seek Medical Attention
Most peptide side effects are mild and manageable. But some symptoms demand immediate attention. Knowing where the line falls between "wait it out" and "stop now and call your doctor" could save your life.
Stop Immediately and Seek Emergency Care
Emergency Symptoms - Call 911 or Go to ER
- Anaphylaxis signs: Throat swelling, difficulty breathing, widespread hives, rapid heartbeat, dizziness, feeling of impending doom. Administer epinephrine (EpiPen) if available.
- Severe abdominal pain radiating to the back: Possible pancreatitis. Stop GLP-1 immediately.
- Persistent vomiting with inability to keep fluids down for 24+ hours: Risk of severe dehydration and acute kidney injury.
- Signs of bowel obstruction: Severe abdominal pain, complete inability to pass gas or stool, abdominal distension, vomiting bile-colored material.
- Chest pain, shortness of breath, or irregular heartbeat: Possible cardiovascular event.
- Signs of severe infection at injection site: Rapidly spreading redness, red streaks extending from injection site, high fever (above 101.3 F / 38.5 C), pus drainage.
- Severe hypoglycemia: Confusion, loss of consciousness, seizure, profuse sweating with shakiness (particularly when GLP-1s are combined with insulin or sulfonylureas).
Stop the Peptide and Contact Your Provider Within 24 Hours
- Persistent severe nausea or vomiting not responding to management strategies after 3+ weeks at the same dose
- Right upper quadrant pain especially after meals (possible gallbladder issue)
- New lump in the neck or difficulty swallowing (thyroid concern - GLP-1 users)
- Fasting blood glucose consistently above 140 mg/dL (GH peptide users)
- Significant swelling that doesn't respond to management strategies or affects breathing
- New or rapidly changing moles (Melanotan II users - dermatology evaluation needed)
- Severe carpal tunnel symptoms affecting hand function or causing muscle weakness/wasting
- Signs of infection: Fever, malaise, injection site becoming increasingly painful/red/warm over 24-48 hours
- Persistent severe headache not responding to standard treatment
- Mood changes: Severe anxiety, depression, or behavioral changes that may relate to peptide use
- Jaundice (yellowing of skin or eyes) - possible liver or gallbladder issue
Reduce Dose and Monitor Closely
These symptoms typically warrant a dose reduction rather than complete discontinuation:
- Moderate nausea persisting beyond 2 weeks at the same dose
- Water retention not responding to lifestyle management
- Fasting glucose 110-140 mg/dL on GH peptides
- Mild carpal tunnel symptoms (tingling but no weakness)
- Persistent headaches that respond to OTC analgesics but keep recurring
- Moderate fatigue affecting daily activities
- Significant appetite increase on MK-677 undermining dietary goals
- Sleep disturbances related to peptide timing
Continue and Monitor (Normal Adaptation)
These are normal side effects during the adaptation period that typically resolve without intervention:
- Mild nausea during first 1-2 weeks of GLP-1 therapy or dose increase
- Mild water retention during first 2-4 weeks of GH peptide use
- Injection site redness/swelling resolving within 24 hours
- Mild headache during first few days of a new peptide
- Mild fatigue during first week
- Vivid dreams on GH peptides
- Mild appetite changes
- Transient flushing after CJC-1295 or Melanotan II injection
Figure 8: Peptide side effect severity guide - green (continue), yellow (reduce dose), red (stop and seek care)
Drug Interactions and Contraindications
Peptide-drug interactions are an under-discussed topic. While peptides generally have fewer drug interactions than small-molecule pharmaceuticals due to their specificity, several clinically significant interactions exist that every user should know about.
GLP-1 Receptor Agonist Drug Interactions
| Interacting Drug/Class | Interaction | Clinical Significance |
|---|---|---|
| Insulin | Additive hypoglycemia risk | High - insulin dose reduction of 20-50% often needed when starting GLP-1 |
| Sulfonylureas (glipizide, glyburide) | Additive hypoglycemia risk | High - may need sulfonylurea dose reduction |
| Oral contraceptives | Delayed gastric emptying may reduce absorption | Moderate - consider backup contraception during initial titration; switch to non-oral contraception if concerned |
| Oral antibiotics | Absorption may be altered by delayed gastric emptying | Moderate - take antibiotics at least 1 hour before GLP-1 administration |
| Warfarin | INR changes reported; altered absorption possible | Moderate - monitor INR closely when starting or adjusting GLP-1 |
| Levothyroxine | Absorption may be affected by gastric emptying changes | Low-Moderate - take levothyroxine on empty stomach, 30-60 min before food; monitor TSH |
| ACE inhibitors / ARBs | Additive blood pressure reduction | Low - generally favorable; monitor for hypotension |
| NSAIDs | Combined GI effects | Low - may increase GI discomfort; use acetaminophen preferentially |
GH Secretagogue Drug Interactions
| Interacting Drug/Class | Interaction | Clinical Significance |
|---|---|---|
| Insulin / Diabetes medications | GH antagonizes insulin; may worsen glucose control | High - especially with MK-677; may need diabetes medication adjustment |
| Corticosteroids | Both raise blood glucose; additive effect | Moderate - monitor glucose more closely |
| Thyroid hormone | GH can increase T4 to T3 conversion | Low-Moderate - monitor thyroid function |
| Growth hormone therapy | Additive GH elevation; increased side effects | High - do not combine GH secretagogues with exogenous GH without medical supervision |
| CYP450-metabolized drugs | MK-677 may affect some CYP450 enzymes | Low - theoretical; limited clinical data |
Healing Peptide Interactions
BPC-157 and TB-500 have no well-documented drug interactions in the scientific literature. However, theoretical considerations include:
- BPC-157 and dopaminergic drugs: BPC-157 interacts with the dopamine system and may modify the effects of dopamine agonists or antagonists. Exercise caution with Parkinson's medications, antipsychotics, and dopaminergic drugs.
- BPC-157 and blood pressure medications: BPC-157 has demonstrated effects on nitric oxide and blood pressure regulation in animal studies. Monitor blood pressure if using with antihypertensives.
- Healing peptides and anticoagulants: Since both BPC-157 and TB-500 affect angiogenesis and tissue repair, there's theoretical interaction with anticoagulants like warfarin. Monitor INR if combining.
Nootropic Peptide Interactions
- Selank and benzodiazepines: Selank modulates GABA-A receptors. Combining with benzodiazepines could theoretically produce additive sedation, though clinical reports suggest Selank's effect is modulatory rather than directly sedating.
- Semax and stimulants: Semax increases dopamine and norepinephrine activity. Combining with stimulant medications (Adderall, modafinil) could theoretically produce overstimulation.
- Semax and antidepressants: Given Semax's effects on serotonin, BDNF, and other neurotransmitter systems, potential interactions with SSRIs, SNRIs, and MAOIs exist but are poorly characterized.
Universal Contraindications
The following are general contraindications that apply across most or all peptide classes:
| Contraindication | Applicable Peptide Classes | Rationale |
|---|---|---|
| Pregnancy and breastfeeding | All peptides | Insufficient safety data; most classified as Category C or unclassified; GLP-1s should be stopped 2+ months before planned conception |
| Active cancer | GH peptides, BPC-157, TB-500, Dihexa, Epithalon | Growth factor promotion and angiogenesis could theoretically support tumor growth |
| Age under 18 | All peptides | No pediatric safety data (except specific FDA-approved indications like tesamorelin) |
| Uncontrolled diabetes | GH peptides (especially MK-677), GLP-1s (with insulin) | GH peptides worsen glucose control; GLP-1s with insulin require careful dose management |
| Active, untreated infections | Immune peptides | Immune modulation during active infection requires medical supervision |
| Organ transplant (immunosuppressed) | Thymosin Alpha-1, other immune peptides | Enhanced immune function could increase rejection risk |
| Personal/family history of MTC or MEN2 | GLP-1 receptor agonists | Boxed warning contraindication based on rodent thyroid tumor data |
| History of pancreatitis | GLP-1 receptor agonists | Precautionary; use with enhanced monitoring if benefits outweigh risks |
| Severe renal impairment | GLP-1s, GH peptides | Altered clearance; increased side effect risk; dose adjustment needed |
| Severe hepatic impairment | GLP-1s, GH peptides, MK-677 | Altered metabolism; increased side effect risk |
For a complete guide to getting started safely with peptides, see our Getting Started page and Peptide Therapy Beginner's Guide.
Special Populations
Certain populations face unique risks and considerations with peptide therapy. This section covers age-related concerns, gender-specific effects, and considerations for people with pre-existing medical conditions.
Older Adults (65+)
Older adults are actually one of the most studied populations for several peptide classes, particularly GLP-1 agonists and GH secretagogues. Key considerations:
- Increased nausea sensitivity: Older adults may experience more pronounced GI side effects with GLP-1s. Slower titration is recommended.
- Dehydration risk: Reduced thirst perception combined with GLP-1-related nausea/vomiting increases dehydration and acute kidney injury risk. Emphasize hydration.
- Sarcopenia concern: GLP-1-mediated weight loss includes lean mass loss, which is particularly concerning in older adults already at risk for sarcopenia. Adequate protein intake (1.0-1.2g per kg daily) and resistance exercise are essential. See our beginner's guide for muscle preservation strategies.
- Fall risk: Dizziness and orthostatic hypotension from GLP-1s may increase fall risk in older adults.
- Polypharmacy: Older adults take more medications, increasing drug interaction potential. Review all medications before starting any peptide.
- GH peptide insulin resistance: Older adults are more likely to have impaired glucose tolerance at baseline, making them more susceptible to GH peptide-related glucose elevations. MK-677 studies in elderly subjects showed significant glucose increases (Nass et al., 2008, DOI: 10.7326/0003-4819-149-9-200811040-00003).
Women-Specific Considerations
- Pregnancy planning: GLP-1 agonists should be discontinued at least 2 months before planned conception (5 half-lives for semaglutide). Animal studies have shown embryo-fetal toxicity. No GLP-1 receptor agonist is approved for use during pregnancy (Parker et al., 2025, DOI: 10.1111/dom.16437).
- Oral contraceptive absorption: GLP-1-mediated delayed gastric emptying may reduce oral contraceptive absorption. Consider non-oral contraceptive methods.
- Menstrual cycle changes: Rapid weight loss from GLP-1 therapy can affect menstrual regularity. Some women report improved cycles (especially those with PCOS-related irregularity), while others report temporary disruption.
- Fertility effects: Weight loss from GLP-1 therapy can improve fertility in overweight/obese women with PCOS. However, this means unexpected pregnancy is possible - use reliable contraception.
- Breastfeeding: Most peptides are contraindicated during breastfeeding due to insufficient safety data on excretion into breast milk.
- PT-141 dosing: PT-141 (Vyleesi) is FDA-approved specifically for premenopausal women with HSDD. The approved dose is 1.75mg SC, not to exceed once in 24 hours or 8 doses per month.
People with Diabetes
- Type 2 diabetes on GLP-1s: GLP-1 agonists are approved for T2DM and are generally safe. However, hypoglycemia risk increases significantly when combined with insulin or sulfonylureas. Insulin dose may need 20-50% reduction when starting GLP-1 therapy.
- Type 2 diabetes on GH peptides: GH peptides can worsen glucose control. MK-677 is the highest risk. If using GH peptides with T2DM, prefer ipamorelin (lowest glucose impact) and monitor glucose closely. Diabetes medication adjustments may be needed.
- Type 1 diabetes: GLP-1 agonists are not first-line for T1DM but are sometimes used off-label. Hypoglycemia risk is significant. GH peptides require very careful monitoring.
- Pre-diabetes: GLP-1 agonists may actually help prevent progression to diabetes. GH peptides (especially MK-677) could push pre-diabetes into diabetes and should be used with caution.
People with Cardiovascular Disease
- GLP-1 agonists: Generally cardiovascular-protective. The LEADER, SUSTAIN-6, and SELECT trials showed reduced cardiovascular events with semaglutide and liraglutide. However, heart rate increases of 2-4 bpm are common and should be monitored in people with arrhythmias.
- GH peptides: GH can cause fluid retention, which may worsen heart failure symptoms. People with congestive heart failure should use GH peptides with caution.
- PT-141/Melanotan II: Can cause blood pressure elevation and heart rate changes. Contraindicated in uncontrolled hypertension.
People with Cancer History
This is the most important special population consideration for peptide therapy:
- Active cancer: GH secretagogues, BPC-157, TB-500, Dihexa, and other growth-promoting peptides are generally contraindicated during active cancer treatment. The theoretical risk of supporting tumor growth through angiogenesis, growth factor promotion, or telomerase activation is too significant to ignore.
- Cancer in remission: Timing varies by peptide and cancer type. Most oncologists recommend waiting at least 2-5 years of confirmed remission before considering growth-promoting peptides. Always get oncologist clearance.
- GLP-1 agonists and cancer: Current evidence does not show increased cancer risk with GLP-1 use. The thyroid cancer concern is specific to MTC/MEN2 family history. Some studies suggest potential protective effects against certain cancers, but this remains investigational.
- Thymosin Alpha-1: Paradoxically, Ta1 has been studied as a supportive therapy during cancer treatment to boost immune function and reduce chemotherapy toxicity. It's one of the few peptides that may be appropriate during active cancer - but only under oncologist supervision.
People with Autoimmune Conditions
- Immune-modulating peptides (Ta1, BPC-157): May help or exacerbate autoimmune conditions depending on the specific condition and immune profile. Medical supervision is essential.
- GLP-1 agonists: No specific autoimmune concerns. May actually have anti-inflammatory benefits.
- GH peptides: GH modulates immune function and could theoretically affect autoimmune conditions, though this is poorly studied.
Psychological and Cognitive Side Effects of Peptides
While most attention focuses on physical side effects, peptides can also affect mood, cognition, motivation, and psychological wellbeing. Some of these effects are direct (pharmacological), while others are indirect consequences of physical changes like rapid weight loss, hormonal shifts, or sleep pattern changes.
GLP-1 Agonists and Mood/Behavior Changes
GLP-1 receptors are present throughout the brain, including areas involved in reward processing, mood regulation, and cognitive function. The effects of GLP-1 agonists on brain function extend beyond simple appetite suppression.
Reported Psychological Effects
- Reduced food reward and cravings: Many users report a fundamental shift in their relationship with food - reduced cravings, less emotional eating, and diminished food-related reward. While most people experience this as beneficial, some find the loss of food pleasure distressing or feel it affects social eating and quality of life.
- Reduced alcohol desire: GLP-1 agonists reduce alcohol intake and cravings in both animal models and human reports. This is being actively studied as a potential treatment for alcohol use disorder. Most users who experience this consider it a positive side effect, but it's worth noting for people who enjoy moderate, social drinking.
- Mood changes: Reports are mixed. Some users report improved mood (possibly related to improved health, weight loss confidence, and reduced inflammatory burden). Others report low mood, anhedonia, or emotional flatness. The FDA's adverse event reporting system has received reports of suicidal ideation and depression with GLP-1 agonists, though the European Medicines Agency's investigation found no causal link. Nevertheless, any new mood changes during GLP-1 therapy should be discussed with your provider (Drucker, 2018, DOI: 10.1016/j.cmet.2018.03.001).
- Changes in identity and self-concept: Rapid, significant weight loss can create psychological challenges around identity, body image, and self-concept. People who have been overweight for years may struggle with the disconnect between their physical appearance and their internal self-image. Loose skin, changed social dynamics, and attention from others can be psychologically complex.
- Anxiety about weight regain: Knowledge that weight often returns after stopping GLP-1 therapy can create anxiety about the future, dependence on the medication, and stress about cost and access.
Cognitive Effects of GLP-1 Agonists
Preclinical research suggests GLP-1 agonists may have neuroprotective properties. Clinical trials of semaglutide for Alzheimer's disease are underway. Most users don't report cognitive side effects, but some note mild "brain fog" during the initial adjustment period, which may relate to reduced caloric intake, dehydration, or electrolyte changes rather than the drug itself.
Growth Hormone Peptides and Psychological Effects
- Improved sleep quality: GH peptides, particularly when dosed before bed, often improve deep sleep quality. Most users consider this a significant benefit. However, vivid dreams (sometimes intense or unusual) are common and can occasionally be disturbing.
- Improved sense of wellbeing: GH optimization is associated with improved mood, energy, and sense of vitality in GH-deficient populations. Many GH peptide users report similar subjective improvements.
- MK-677 lethargy: MK-677 can cause significant lethargy and drowsiness in 20-30% of users, which can affect motivation, productivity, and mood. This is partly related to its effects on sleep architecture and partly to blood sugar fluctuations.
- Irritability from appetite: The intense hunger caused by GHRP-6 and MK-677 can cause irritability, preoccupation with food, and difficulty concentrating, especially in people trying to maintain a caloric deficit.
Nootropic Peptides and Psychological Effects
By definition, nootropic peptides are designed to affect brain function, so psychological and cognitive effects are expected:
- Selank: Reduces anxiety without sedation or cognitive impairment. May cause mild drowsiness in some users (likely reflecting its GABAergic activity). No withdrawal, dependence, or rebound anxiety has been documented. Some users report initial emotional sensitivity as anxiety levels shift.
- Semax: Can cause overstimulation, anxiety, and insomnia if dosed too high or too late in the day. The enhanced variants (NA-Semax, NA-Semax-Amidate) are more potent and more likely to cause these effects. Some users report a sense of emotional detachment at higher doses.
- Dihexa: Anecdotal reports include enhanced dream vividness, improved memory consolidation, and occasionally heightened emotional intensity. The potent neurotrophic activity may cause headaches as new neural connections form.
Melanotan II and Psychological Effects
Melanotan II activates melanocortin receptors in the brain that are involved in mood, arousal, and behavior:
- Sexual arousal increase (can be unwanted, especially spontaneous erections in men)
- Mood elevation in some users
- Appetite suppression (which can affect eating behavior and social dynamics around food)
- Body image changes from tanning and weight effects
Managing Psychological Side Effects
Psychological Support During Peptide Therapy
- Set realistic expectations: Understanding what side effects to expect - including psychological ones - reduces anxiety and improves coping.
- Monitor mood: Keep a simple mood journal during the first 4-8 weeks of any new peptide. Note energy levels, mood, sleep quality, and any emotional changes.
- Maintain social eating: If GLP-1 therapy reduces food enjoyment, focus on the social aspects of meals rather than the food itself. Don't isolate around mealtimes.
- Address body image proactively: If undergoing significant weight loss, consider working with a therapist experienced in body image and weight-related identity issues.
- Report mood changes: Any significant new depression, anxiety, or suicidal thoughts should be reported to your healthcare provider immediately, regardless of whether you think it's related to the peptide.
- Sleep hygiene: Many psychological side effects are secondary to sleep disruption. Optimize sleep: consistent schedule, cool dark room, no screens 1 hour before bed, avoid caffeine after noon.
Peptide Side Effects in Athletes and Performance Populations
Athletes and fitness-focused individuals represent a large segment of peptide users, yet their side effect considerations differ from general populations. Higher metabolic demands, intense training, specific body composition goals, and anti-doping regulations create a unique risk-benefit landscape.
GH Secretagogues in Athletes
Athletes commonly use GH peptides for recovery, body composition, and injury healing. Specific athletic considerations include:
Water Retention and Performance
While water retention from GH peptides is typically mild (2-5 pounds), it can meaningfully affect athletes in weight-class sports (wrestling, MMA, boxing, weightlifting). The additional fluid weight may require adjustments to weight management strategies during competition preparation. Water retention can also temporarily reduce muscle definition, which matters for physique competitors.
Carpal Tunnel and Grip Strength
GH peptide-induced carpal tunnel symptoms can specifically impair grip strength and hand dexterity, affecting athletes in sports requiring strong grip (climbing, gymnastics, weightlifting, racquet sports, golf). If grip-dependent performance is affected, dose reduction or temporary discontinuation around competitions is advisable.
Joint Pain vs. Training Soreness
Distinguishing between GH peptide-related joint pain (arthralgias from IGF-1-driven soft tissue changes) and normal training-related soreness can be difficult. GH-related joint pain tends to be more diffuse, affects joints not loaded during training, and is worst upon waking. Training soreness is localized to worked muscles and follows exercise patterns.
Blood Sugar During Training
MK-677's effect on blood glucose can be particularly problematic for athletes training in a fasted state or doing high-intensity interval work. Blood sugar fluctuations can cause mid-workout energy crashes, impaired performance, and increased perceived exertion. Athletes using MK-677 should monitor blood glucose around training and consider timing carbohydrate intake strategically.
BPC-157 and TB-500 in Athletes
Healing peptides are extremely popular among athletes for injury recovery. Key athletic considerations:
- Anti-doping status: Both BPC-157 and TB-500 are prohibited under WADA (World Anti-Doping Agency) rules under the S0 category (unapproved substances). Athletes subject to anti-doping testing should not use these compounds.
- Temporary inflammation increase: BPC-157 and TB-500 can temporarily increase inflammation at injury sites as healing accelerates. This can be misinterpreted as injury worsening. Athletes should be patient and not increase training load in response to initial healing discomfort.
- Premature return to training: Perhaps the biggest risk isn't a side effect of the peptide itself but the behavior it enables. If healing peptides accelerate recovery, athletes may return to full training before the tissue is structurally ready for full load, leading to re-injury. Conservative, progressive loading remains essential even with peptide-assisted healing.
- Masking injury severity: Pain reduction from BPC-157 and anti-inflammatory effects from TB-500 could theoretically mask the severity of an injury, leading to continued training on a structure that needs rest. Always get proper diagnostic imaging for significant injuries regardless of symptom improvement.
GLP-1 Agonists in Athletes
While primarily used for weight management, some athletes use GLP-1 agonists for body composition. Athletic-specific concerns include:
- Lean mass loss: As discussed, 25-40% of GLP-1-related weight loss is lean mass. For athletes where muscle mass is performance-critical, this is a significant concern. High protein intake and aggressive resistance training are essential.
- Reduced caloric intake: GLP-1-induced appetite suppression can make it difficult to consume enough calories to fuel training. Under-fueling impairs recovery, increases injury risk, and can lead to relative energy deficiency in sport (RED-S).
- GI issues during training: Nausea and delayed gastric emptying can be amplified by exercise, especially high-intensity or impact-based activities. Training with a GLP-1-slowed stomach full of undigested food is uncomfortable at best and can trigger vomiting during intense sessions.
- Dehydration risk: Athletes who sweat heavily during training are already at dehydration risk. Add GLP-1-related nausea and reduced fluid intake, and the dehydration risk compounds. Electrolyte supplementation becomes even more critical.
Nootropic Peptides in Athletes
Selank and Semax are occasionally used by athletes for competition anxiety management and cognitive performance:
- Competition anxiety (Selank): Selank's anxiolytic effects can help with pre-competition anxiety without the sedation or motor impairment of benzodiazepines. The main concern is ensuring the nasal spray doesn't cause nasal irritation or congestion that affects breathing during competition.
- Focus and reaction time (Semax): Semax may improve focus and reaction time, which is valuable for precision and reaction-dependent sports. However, overstimulation at high doses can cause anxiety and impair performance through overthinking.
- WADA status: Selank and Semax are not specifically listed on the WADA Prohibited List but could potentially fall under the S0 (unapproved substances) category. Athletes should check with their anti-doping authority before use.
Recovery and Periodization Considerations
For athletes using multiple peptides, periodizing their use with training cycles can help manage side effects and optimize results:
| Training Phase | Peptide Strategy | Side Effect Considerations |
|---|---|---|
| Off-season / Base building | GH peptides for recovery and body composition; BPC-157/TB-500 for lingering injuries | Water retention acceptable; blood sugar monitoring important |
| Pre-competition / Peaking | Reduce or discontinue GH peptides (water retention affects weight class or aesthetics); nootropics for focus | Allow 1-2 weeks for water weight to clear before competition |
| Post-injury / Rehabilitation | BPC-157 and/or TB-500 for healing; GH peptides for recovery support | Monitor for premature return to training; don't let pain reduction mask injury |
| Weight cut (weight-class sports) | Discontinue MK-677 and GH peptides 2-3 weeks before cut | Water retention from GH peptides complicates weight management |
Combining Multiple Peptides: Stacking Safety and Interaction Considerations
Many peptide users run multiple compounds simultaneously - what the community calls "stacking." While combining complementary peptides can enhance results, it also creates potential for additive side effects, unknown interactions, and complexity that makes it harder to identify which compound is causing any particular problem.
Common Peptide Stacks and Their Side Effect Profiles
CJC-1295 + Ipamorelin (The Standard GH Stack)
This is the most popular peptide stack and one of the best-characterized in terms of combined side effects. The combination produces stronger GH release than either compound alone, which means:
- Water retention may be more pronounced than with either peptide solo
- Histamine reactions from CJC-1295 are the main unique concern; ipamorelin doesn't cause these
- Carpal tunnel risk is proportional to total GH stimulation - higher with the stack than with either alone
- Blood sugar impact is generally mild at standard doses but warrants monitoring
- Starting each peptide separately (ipamorelin first for 1-2 weeks, then adding CJC-1295) helps isolate the cause of any side effects
BPC-157 + TB-500 (The "Wolverine" Healing Stack)
Combined healing peptide use is common for injury recovery:
- Side effects may be additive - headache and nausea incidence may increase
- Both promote angiogenesis, so the theoretical cancer concern is amplified
- No drug interaction data exists for this specific combination
- Separate administration times (e.g., BPC-157 AM and TB-500 PM) may reduce combined peak side effects
- Start each individually before combining to establish individual tolerability
GLP-1 + GH Peptide Stack
Some users combine GLP-1 agonists with GH peptides to lose fat while preserving lean mass:
- Opposing metabolic effects: GLP-1s improve insulin sensitivity while GH peptides can worsen it
- Net blood sugar effect depends on the specific compounds and doses
- The GH peptide may help preserve lean mass during GLP-1-mediated weight loss - a potential benefit
- Increased monitoring burden: need to track GI symptoms, water retention, blood sugar, and IGF-1
- Enhanced nausea risk from additive GI effects
GH Peptide + Healing Peptide Stack
Common for injury recovery with simultaneous body composition goals:
- Generally well-tolerated combination
- GH promotes systemic healing and may complement BPC-157/TB-500's localized effects
- Combined angiogenesis promotion (theoretical concern)
- Water retention from GH peptides is the primary additional side effect
Stacking Safety Guidelines
Rules for Safe Peptide Stacking
- One at a time first: Never start two or more new peptides simultaneously. Begin one compound, establish tolerability over 1-2 weeks, then add the next. This lets you identify the source of any side effects.
- Start at lower doses: When combining peptides with overlapping mechanisms (like two GH secretagogues), start each at a lower dose than you would use if taking it alone. The combined effect may be greater than the sum of individual doses.
- Track everything: Keep a log of each compound, dose, timing, injection site, and any symptoms. When running multiple peptides, memory alone isn't reliable enough.
- Limit total compounds: There's a practical limit to how many peptides can be safely combined and meaningfully tracked. Most experienced practitioners recommend no more than 3-4 compounds simultaneously.
- Separate timing: When possible, administer different peptides at different times to reduce peak side effect overlap and allow for better absorption.
- Increase monitoring: More compounds means more monitoring. Blood work frequency should increase proportionally to protocol complexity.
- Know which to stop first: If you develop a concerning side effect while running multiple peptides, know which compound is most likely responsible and stop that one first. If the side effect is serious, stop everything.
Peptides That Should Not Be Combined
| Combination to Avoid | Reason |
|---|---|
| Multiple GH secretagogues at full dose (e.g., MK-677 + GHRP-6 + ipamorelin all at full dose) | Excessive GH elevation; amplified side effects (water retention, insulin resistance, carpal tunnel) |
| Multiple GLP-1 agonists simultaneously | No clinical rationale; doubled GI side effect burden with no proven additional benefit |
| GH peptides + exogenous GH without medical supervision | Risk of supraphysiological GH levels with acromegaly-like effects |
| Multiple nootropic peptides with overlapping neurotransmitter effects | Risk of overstimulation, anxiety, insomnia, or unpredictable neuropsychiatric effects |
| Any peptide combination in cancer patients without oncologist approval | Multiple growth-promoting pathways activated simultaneously creates unacceptable theoretical risk |
Rare and Serious Side Effects: A Detailed Analysis
While most peptide side effects are mild and manageable, a small number of rare but serious adverse events have been documented. Understanding these rare outcomes - their frequency, warning signs, and risk factors - is critical for informed decision-making and timely intervention.
GLP-1 Agonists: Rare Serious Events
Acute Pancreatitis
Pancreatitis remains the most discussed rare serious event with GLP-1 agonists, despite reassuring trial data. Here's what we know from the largest clinical studies:
- LEADER trial (liraglutide, n=9,340): Pancreatitis occurred in 18 patients (0.4%) receiving liraglutide versus 23 patients (0.5%) receiving placebo - actually lower in the treatment group (Marso et al., 2016, DOI: 10.1056/NEJMoa1603827).
- SUSTAIN-6 (semaglutide, n=3,297): Pancreatitis was rare and balanced between groups
- SURMOUNT-1 (tirzepatide, n=2,539): Pancreatitis rate less than 0.3% in all tirzepatide dose groups
- SELECT (semaglutide 2.4mg for obesity, n=17,604): No increased pancreatitis signal
Risk factors that may predispose GLP-1 users to pancreatitis include:
- History of pancreatitis (the most significant risk factor - some clinicians consider this a relative contraindication)
- History of gallstones (biliary pancreatitis is the most common form)
- Heavy alcohol use
- Very high triglycerides (above 500 mg/dL)
- Concurrent use of other drugs associated with pancreatitis (e.g., azathioprine, valproic acid)
Warning signs that require immediate medical evaluation: Severe, constant upper abdominal pain (often described as boring or knife-like) that radiates to the back, is worsened by eating and lying down, and is accompanied by nausea, vomiting, and sometimes fever. This presentation warrants emergency department evaluation with lipase testing and imaging.
Intestinal Obstruction
Post-marketing surveillance has identified rare cases of intestinal obstruction in GLP-1 users. The mechanism likely involves severe gastroparesis or ileus (temporary intestinal paralysis) from excessive GI motility slowing. Cases have predominantly occurred in patients with pre-existing GI motility disorders, adhesions from prior abdominal surgery, or concurrent opioid use (which further slows motility).
Warning signs: Complete inability to pass gas or stool, progressively worsening abdominal distension, severe abdominal pain, bile-colored vomiting. This is a surgical emergency.
Acute Kidney Injury
Acute kidney injury (AKI) associated with GLP-1 agonists is almost always secondary to dehydration from severe vomiting and diarrhea rather than a direct toxic effect. The kidney itself is not harmed by GLP-1 agonists - in fact, GLP-1 agonists may be renoprotective. But dehydration from any cause can damage the kidneys, and GI side effects of GLP-1s can cause significant fluid losses in some patients.
Risk factors include pre-existing chronic kidney disease, concurrent use of ACE inhibitors or ARBs, NSAIDs, diuretics, and inadequate fluid intake during GI episodes. Prevention centers on aggressive hydration during nausea/vomiting episodes and seeking medical attention if unable to keep fluids down for more than 24 hours.
Suicidal Ideation and Psychiatric Events
The European Medicines Agency (EMA) investigated reports of suicidal thoughts and self-injury in GLP-1 agonist users in 2023-2024. Their review concluded that available evidence did not confirm a causal link between GLP-1 agonists and suicidal/self-harm events. However, this remains an area of active pharmacovigilance.
Factors that may contribute to mood changes in GLP-1 users (independent of the drug itself) include rapid weight loss and associated hormonal shifts, reduced food reward (for people whose emotional regulation depends heavily on eating), pre-existing depression or anxiety, social and identity changes from weight loss, and the stress of medical treatment itself.
Anyone experiencing new depression, anxiety, hopelessness, or suicidal thoughts during GLP-1 therapy should contact their healthcare provider immediately. National Suicide Prevention Lifeline: 988 (call or text).
GH Secretagogues: Rare Serious Events
New-Onset Diabetes from MK-677
While blood sugar elevation is a known and common side effect of MK-677, progression to frank diabetes is rare but documented. A case reported by Kinoshita and Homma described a previously healthy bodybuilder who developed type 2 diabetes during MK-677 use, with HbA1c rising from normal range to diabetic levels (Kinoshita & Homma, 2022, DOI: 10.1530/endoabs.86.P341). The diabetes was identified only after symptoms of hyperglycemia developed, suggesting that regular monitoring could have identified the problem earlier.
Risk factors for MK-677-induced diabetes progression include pre-existing insulin resistance or pre-diabetes, family history of type 2 diabetes, high body fat percentage (especially visceral fat), concurrent use of other insulin-antagonizing substances, and higher doses (25mg vs. 10mg).
Acromegaly-Like Effects
Chronic supraphysiological GH elevation from aggressive dosing of GH secretagogues can theoretically produce acromegaly-like effects - soft tissue swelling, jaw widening, hand and foot enlargement, and organ growth. These effects are associated with GH abuse at doses far exceeding standard peptide protocols and are typically seen with exogenous GH injection rather than secretagogues (which have ceiling effects on GH release). Nevertheless, using multiple GH secretagogues simultaneously at full doses over extended periods could theoretically approach supraphysiological levels in some individuals.
Monitoring IGF-1 levels every 3-6 months helps ensure GH stimulation remains in the physiological range (typically 150-350 ng/mL for adults, age-dependent). IGF-1 consistently above the upper reference range should prompt dose reduction.
Healing Peptides: Rare Serious Events
Contamination-Related Sepsis
The most serious adverse events documented with BPC-157 and TB-500 are contamination-related rather than peptide-related. Bacterial contamination of gray-market peptide products has caused injection site abscesses, cellulitis, and in severe cases, sepsis. These events are entirely preventable through proper sourcing, sterile technique, and quality-verified products.
Signs of systemic infection after peptide injection include fever above 101.3 F (38.5 C) developing within hours to days of injection, rapidly spreading redness from the injection site, red streaks extending away from the injection site (lymphangitis), chills, malaise, rapid heart rate, and altered mental status. Any of these warrant emergency medical evaluation.
Theoretical Cancer Concerns
As discussed throughout this encyclopedia, the angiogenesis-promoting properties of BPC-157 and TB-500 raise theoretical concerns about supporting tumor growth. To date, no clinical cases of cancer promotion from these peptides have been documented. However, the absence of large-scale human trials means this risk cannot be definitively excluded. The most prudent approach remains avoiding these peptides in people with active cancer, recent cancer history (less than 2-5 years remission), or known precancerous conditions.
Melanotan II: Rare Serious Events
Melanoma
At least four case reports describe melanoma developing in Melanotan II users. The causal relationship is debated. A 2021 systematic review concluded that increased melanoma risk in Melanotan II users is probably explained by concurrent UV exposure (users seek UV to activate the tan) rather than the peptide directly promoting melanocyte transformation. However, Melanotan II does stimulate melanocyte proliferation and activity, and its effects on melanocytic nevi (moles) are well-documented. Conservative recommendations include full-body dermatological examination before starting Melanotan II, regular (every 6-12 months) skin checks during use, ABCDE monitoring of all moles (Asymmetry, Border irregularity, Color variation, Diameter over 6mm, Evolution/change), prompt dermatological evaluation of any new or changing moles, and ideally minimizing UV exposure (which somewhat defeats the purpose for many users).
Cardiovascular Events
Melanotan II can cause acute blood pressure elevation and heart rate changes. While typically transient and mild, in individuals with uncontrolled hypertension, coronary artery disease, or arrhythmias, these cardiovascular effects could theoretically trigger serious events. Melanotan II should be avoided entirely in people with significant cardiovascular disease.
Anaphylaxis Risk Across All Peptides
True anaphylaxis from peptide therapy is extremely rare but possible with any injected substance. The risk is highest with:
- Peptides derived from animal sources (cerebrolysin from porcine brain tissue)
- First-time exposure to any new peptide
- People with a history of multiple drug allergies
- Products containing allergens in excipients (e.g., mannitol, polysorbate)
Risk mitigation includes having an EpiPen available (especially for first-time peptide use in people with allergy history), performing a small test dose (10-20% of full dose) for the first injection, waiting 15-30 minutes at the clinic after first injection, and knowing the signs of anaphylaxis (throat swelling, difficulty breathing, widespread hives, rapid heartbeat, dizziness, feeling of impending doom).
Building Your Personal Side Effect Management Plan
With all the information in this encyclopedia, how do you build a practical, personalized plan for managing peptide side effects? This section provides a step-by-step framework.
Step 1: Pre-Protocol Assessment
Before starting any peptide, complete this assessment:
- Medical history review: Identify any conditions that create specific risks (diabetes, cancer history, GI conditions, autoimmune disorders, cardiovascular disease, thyroid issues, kidney or liver disease)
- Medication review: List all medications, supplements, and OTC drugs for interaction screening
- Baseline blood work: Complete the appropriate testing panel for your peptide class (see monitoring recommendations above)
- Provider consultation: Discuss your protocol with a qualified healthcare provider who can evaluate your specific risk profile
- Emergency plan: Know what symptoms warrant immediate medical attention for your specific peptides, know your nearest emergency department, and have your provider's contact information readily available
Step 2: Protocol Design for Side Effect Minimization
- Compound selection: Within each peptide class, some compounds are better tolerated than others. Ipamorelin is better tolerated than GHRP-6. Tirzepatide may cause less nausea than semaglutide. Choose the compound with the best benefit-to-side-effect ratio for your goals.
- Starting dose: Always start at the lowest recommended dose or lower. You can always increase later.
- Titration schedule: Plan your dose increases in advance, but build in flexibility. Only increase when current side effects have resolved.
- Timing optimization: Choose injection times that minimize side effect impact on daily life (see timing strategies section above).
- Stacking approach: If using multiple peptides, stagger start dates by 1-2 weeks per compound.
Step 3: Side Effect Monitoring and Tracking
Daily Tracking Template
Track these items daily during the first 4-8 weeks of any new peptide or dose change:
- Date, compound(s), dose(s), injection site
- Nausea rating (0-10 scale)
- Energy level (0-10 scale)
- Sleep quality (0-10 scale)
- Injection site status (none / mild redness / swelling / pain / other)
- Other symptoms (headache, water retention, mood changes, appetite, bowel habits)
- Any management interventions used and their effectiveness
This log becomes invaluable for identifying patterns, guiding dose adjustments, and communicating effectively with your healthcare provider.
Step 4: Graduated Response to Side Effects
Follow this escalating response framework:
- Level 1 - Wait and observe (3-7 days): For mild, expected side effects during the known adaptation period. Implement basic lifestyle strategies (hydration, small meals, timing adjustments). Continue current dose.
- Level 2 - Active management (1-2 weeks): For moderate side effects that don't resolve with basic strategies. Add targeted interventions (antihistamines for flush, ginger for nausea, wrist splints for carpal tunnel, sodium restriction for water retention). Continue current dose.
- Level 3 - Dose reduction (2-4 weeks): For side effects that persist despite active management for 2+ weeks. Reduce dose by 25-50%. Reassess after 2 weeks at the reduced dose. May attempt re-escalation later.
- Level 4 - Temporary discontinuation (1-2 weeks): For side effects that persist even at reduced doses. Stop the peptide entirely for 1-2 weeks. Reassess whether to restart at a lower dose, switch to an alternative compound, or discontinue permanently.
- Level 5 - Medical intervention: For serious or concerning symptoms. Contact healthcare provider. May require medication, testing, or emergency care.
Step 5: Regular Protocol Review
Review your protocol at these intervals:
- 2 weeks: Initial tolerability assessment. Are side effects acceptable? Is the dose appropriate?
- 4-6 weeks: Adaptation check. Have initial side effects diminished? Ready for dose escalation?
- 3 months: Blood work review. Are metabolic markers (glucose, IGF-1, kidney function, liver function) within acceptable ranges?
- 6 months: Full protocol review with provider. Are goals being met? Are there any emerging side effects? Should the protocol be adjusted, continued, or discontinued?
- 12 months: Long-term assessment. Evaluate need for continued therapy, dose optimization, and any long-term effects.
For help getting started with any peptide protocol, visit our Getting Started page or explore the Peptide Research Hub for compound-specific guidance.
Side Effects Organized by Body System
While the previous sections organized side effects by peptide class, this section reorganizes the same information by body system - useful when you're experiencing a symptom and want to know which peptides might be responsible and what to do about it.
Gastrointestinal System
The GI tract is the most commonly affected body system across all peptide classes, though the mechanisms and specific manifestations vary considerably.
Upper GI: Nausea, Vomiting, and Gastric Discomfort
Nausea is the single most reported peptide side effect. Its prevalence varies enormously by peptide class:
- GLP-1 agonists: 20-44% incidence. Mechanism: delayed gastric emptying and central satiety signaling via brainstem GLP-1 receptors. This is the most intense and persistent peptide-related nausea, typically requiring weeks of adaptation.
- Melanotan II / PT-141: 40% incidence. Mechanism: melanocortin receptor activation in the area postrema (vomiting center). Rapid onset (30-90 minutes post-injection), resolves within 2-4 hours. Usually improves after the first 3-5 doses.
- BPC-157 (oral): 5-10% incidence. Mechanism: direct gastric mucosal effects. Typically mild and transient.
- GH secretagogues: 5-10% incidence. Mechanism: GH and ghrelin pathway activation. Usually mild and short-lived.
- TB-500: 5-10% incidence. Mechanism: uncertain; may relate to immune modulation effects. Usually mild.
The critical distinction for clinical management: GLP-1 nausea requires patience and structured titration over weeks. Melanotan II nausea resolves quickly and mainly needs acute symptom management. Other peptide nausea is typically mild enough to manage with basic strategies.
Lower GI: Diarrhea, Constipation, and Bowel Changes
Lower GI effects are predominantly a GLP-1 class phenomenon:
- Diarrhea: 8-30% on GLP-1s (tirzepatide higher than semaglutide for diarrhea specifically). Mechanism: altered GI motility, changes in bile acid recycling, and osmotic effects. Usually episodic rather than continuous. Peaks during dose titration. Management: hydration, electrolytes, BRAT diet during acute episodes, loperamide for severe cases, probiotics (especially Saccharomyces boulardii).
- Constipation: 5-15% on GLP-1s. Mechanism: slowed colonic transit as an extension of gastric emptying delay. Can be particularly bothersome for patients with pre-existing slow motility. Management: fiber supplementation (psyllium husk 5-10g daily), magnesium citrate (400-600mg at bedtime), adequate water (80+ oz daily), regular physical activity, stool softeners if needed.
- Bloating and flatulence: 10-15% on GLP-1s, also reported with oral BPC-157 and some immune peptides. Management: digestive enzymes with meals, reducing gas-producing foods, simethicone for acute relief, peppermint oil capsules.
- VIP (Vasoactive Intestinal Peptide): Can cause diarrhea through its normal mechanism of promoting intestinal secretion. This is expected pharmacology and dose-dependent.
Hepatobiliary: Gallbladder and Liver
As detailed in the GLP-1 section, gallbladder disease risk is elevated with GLP-1 agonist use (RR 1.37 overall). The liver effects of peptides are generally minimal at therapeutic doses, but monitoring liver function (AST, ALT, ALP, bilirubin) is recommended for long-term users of any peptide processed hepatically. MK-677 in particular should prompt liver function monitoring given its oral administration and hepatic processing.
Musculoskeletal System
Water Retention and Joint Effects
The musculoskeletal system is primarily affected by GH secretagogues through IGF-1-mediated soft tissue changes:
- Edema (water retention): 20-60% on GH peptides. Mechanism: GH promotes renal sodium and water reabsorption. Typically presents as morning facial puffiness, finger swelling, and ankle edema. Usually self-limiting within 2-4 weeks. Management: sodium restriction, potassium-rich foods, adequate hydration, exercise, dose reduction if persistent.
- Joint pain (arthralgia): 5-15% on GH peptides; 13.3% on tesamorelin in clinical trials. Mechanism: IGF-1-driven changes in cartilage and synovial tissue. Most commonly affects wrists, knees, and shoulders. Management: OTC anti-inflammatory medications, dose reduction, cycling protocols.
- Carpal tunnel syndrome: 10-20% on GH peptides at moderate to high doses; 4.6% on tesamorelin. Mechanism: soft tissue swelling compressing the median nerve in the carpal tunnel. Symptoms: numbness, tingling, pins-and-needles in thumb, index, and middle fingers, worst at night. Fully reversible with dose reduction or discontinuation. Management: nocturnal wrist splints, B6 supplementation, dose reduction.
- Muscle cramps: 5-10% on GH peptides and MK-677. Mechanism: electrolyte shifts from water retention, and direct effects on muscle tissue from IGF-1. Management: magnesium supplementation (400mg daily), adequate potassium and sodium, hydration, stretching.
Lean Mass and Bone Density Changes
GLP-1-mediated weight loss affects both muscle mass and, potentially, bone density. Dual-energy X-ray absorptiometry (DEXA) studies in GLP-1 trials show that while bone mineral density doesn't typically decrease significantly during treatment, the reduction in mechanical loading from weight loss could theoretically affect bone health long-term. This is most concerning in postmenopausal women and older adults already at osteoporosis risk. Adequate calcium (1000-1200mg daily), vitamin D (2000-5000 IU daily), vitamin K2 (100-200mcg daily), and weight-bearing exercise are protective.
Nervous System
Headache
Headache is a cross-class side effect occurring with GLP-1s (10-15%), GH peptides (10-15%), healing peptides (5-15%), nootropics (5-10%), and cosmetic peptides (11% for PT-141). Mechanisms vary: dehydration-related (most common), blood pressure changes, neurotransmitter modulation, histamine release, or direct neurological effects. Management depends on the underlying mechanism but generally involves hydration, electrolytes, acetaminophen, and dose adjustment if persistent.
Dizziness and Lightheadedness
Occurs with GLP-1s (5-8%), various other peptides (2-5%). Usually related to dehydration, blood pressure changes, or blood sugar fluctuations. Particularly concerning in older adults due to fall risk. Management: slow positional changes, adequate hydration, blood pressure monitoring, dose adjustment.
Peripheral Neuropathy Symptoms
Primarily from GH peptide-induced carpal tunnel (see musculoskeletal section). Also reported with long-term high-dose B6 supplementation paradoxically (do not exceed 200mg B6 daily). Semax-derived peptides may occasionally produce tingling or paresthesia through neurotrophic mechanisms.
Endocrine System
Blood Sugar and Insulin
GH secretagogues, particularly MK-677, have the most significant endocrine side effects through their impact on glucose metabolism. GLP-1 agonists improve glucose metabolism (this is therapeutic for diabetics). The opposing effects make combining GH peptides and GLP-1 agonists complex from an endocrine standpoint.
Thyroid
GLP-1 agonists carry the MTC boxed warning based on animal data (no human signal). GH peptides can increase T4-to-T3 conversion, potentially affecting thyroid medication dosing in people on levothyroxine. Monitoring thyroid function (TSH, free T4) annually during peptide therapy is reasonable.
Cortisol and Adrenal
GHRP-6 and hexarelin can raise cortisol levels, which could theoretically worsen adrenal fatigue symptoms or affect stress response. Ipamorelin and CJC-1295 do not significantly affect cortisol - a key advantage. Semax, being derived from ACTH fragments, could theoretically affect the HPA axis, though this hasn't been clinically significant in studies.
Sex Hormones
GHRP-6 and hexarelin can raise prolactin, potentially causing decreased libido, erectile dysfunction, or gynecomastia in men. GLP-1-mediated weight loss can improve sex hormone profiles (especially in PCOS patients), potentially increasing fertility. Melanotan II and PT-141 directly affect sexual function through melanocortin receptor activation. Gonadorelin (GnRH analog) is sometimes used alongside peptide protocols to maintain endogenous testosterone production.
Cardiovascular System
Heart Rate
GLP-1 agonists consistently increase heart rate by 2-4 beats per minute on average. While this is clinically insignificant for most people, it should be monitored in patients with arrhythmias, pre-existing tachycardia, or those taking medications that also increase heart rate.
Blood Pressure
GLP-1 agonists generally lower blood pressure (beneficial). Melanotan II and PT-141 can transiently raise blood pressure. GH peptide-related water retention can modestly increase blood pressure. VIP is a vasodilator and can lower blood pressure, sometimes causing orthostatic hypotension.
Fluid Overload
In patients with heart failure or renal disease, GH peptide-induced fluid retention can worsen fluid overload. Careful monitoring and possible diuretic support may be needed if GH peptides are used in these populations.
Immune System
Immune Enhancement
Thymosin Alpha-1 enhances immune function, which is beneficial in most contexts but could theoretically exacerbate autoimmune conditions. Selank has immunomodulatory properties (it's derived from tuftsin, an immune peptide). BPC-157 modulates various immune pathways. The immune effects of these peptides are generally beneficial but should be monitored in immunologically complex patients.
Histamine and Allergic Responses
CJC-1295 is the primary culprit for histamine-mediated reactions (mast cell degranulation). True peptide allergies (IgE-mediated) are rare but possible. Cerebrolysin carries higher allergy risk due to its porcine-derived protein content. Any new peptide should be introduced with awareness of potential allergic reactions, especially in people with multiple drug allergies.
Integumentary System (Skin, Hair, Nails)
Skin Changes
Melanotan II causes skin darkening (desired effect) and mole changes (undesired effect). GHK-Cu promotes collagen synthesis and wound healing in the skin (generally positive). Injection site changes (lipohypertrophy, lipoatrophy) affect skin and subcutaneous tissue at frequently used sites. "Ozempic face" (facial volume loss) is a skin/subcutaneous tissue manifestation of rapid weight loss.
Hair
GLP-1-mediated weight loss can cause telogen effluvium (hair shedding). GHK-Cu may promote hair growth when applied topically or injected. MK-677 and other GH peptides may improve hair quality through IGF-1 effects on hair follicle cycling. Semax has rare anecdotal reports of hair thinning, mechanism uncertain.
Nails
Nutritional deficiencies from GLP-1-induced reduced food intake can cause brittle nails. GH peptides may improve nail growth and quality. Adequate protein, biotin, and iron are the key nutrients for nail health during peptide therapy.
Emerging Research and Future Outlook on Peptide Safety
The peptide safety landscape is evolving rapidly. New compounds, better formulations, combination therapies, and longer-duration studies are all shaping our understanding of peptide side effects and how to manage them.
Next-Generation GLP-1 Agonists and Improved Tolerability
Several next-generation GLP-1-based therapies in development aim to improve tolerability:
- Retatrutide (triple agonist - GLP-1/GIP/glucagon): Phase 3 trials are ongoing. The addition of glucagon receptor agonism may provide faster weight loss but adds potential for hepatic glycogen depletion and hyperglucagonemia-related effects. Early data suggests GI side effect rates comparable to existing GLP-1 agonists (Jastreboff et al., 2023).
- Orforglipron (oral non-peptide GLP-1 agonist): As a small molecule rather than a peptide, orforglipron eliminates injection-related side effects entirely. Phase 3 trials show GI side effect profiles similar to injectable GLP-1s, but the oral route may improve adherence and convenience.
- CagriSema (semaglutide + cagrilintide): Combines GLP-1 and amylin receptor agonism. The combination produces greater weight loss than either alone. GI side effects appear similar to semaglutide alone, suggesting amylin co-agonism doesn't substantially worsen tolerability.
- Long-acting formulations: Monthly or even longer-duration GLP-1 formulations are in development. These could reduce the frequency of GI side effect peaks associated with each injection cycle.
Improved GH Peptide Formulations
Research into GH secretagogue modifications continues:
- Sustained-release formulations that produce more physiological GH release patterns may reduce the peak-related side effects (water retention, glucose spikes) seen with bolus dosing
- Selective GH secretagogues with minimal ghrelin-related hunger effects are under investigation
- Oral GH secretagogues beyond MK-677 with improved glucose tolerance profiles are in early development
Healing Peptide Research: Moving Toward Human Trials
The most significant development in healing peptide safety would be proper human clinical trials:
- Multiple research groups have called for well-designed human trials of BPC-157, particularly for musculoskeletal indications
- The gap between extensive preclinical safety data and the absence of formal human safety data is the central challenge for BPC-157 and TB-500
- If human trials confirm the favorable preclinical safety profile, it would substantially strengthen the evidence base for clinical use
- Regulatory pathways for peptide therapeutics are being discussed as the FDA considers how to handle the growing gray market
Pharmacogenomics and Personalized Peptide Therapy
An emerging frontier is using genetic testing to predict individual side effect risk:
- CYP450 enzyme polymorphisms may affect metabolism of orally bioavailable peptides like MK-677
- Genetic variations in GLP-1 receptor structure may explain why some patients are extremely sensitive to GLP-1 agonist side effects while others tolerate high doses easily
- HLA typing might eventually predict who is at risk for immune-mediated peptide reactions
- Pharmacogenomic-guided peptide selection could optimize compound choice and starting doses based on individual genetic profiles
While pharmacogenomic-guided peptide therapy is still largely theoretical, it represents the direction that personalized medicine is heading. In the meantime, the practical approach remains starting low, going slow, monitoring closely, and adjusting based on individual response.
Regulatory Evolution
The regulatory landscape for peptides is in flux:
- The FDA's 2023 Category 2 designation for BPC-157 restricted compounding pharmacy access, pushing more users to less regulated sources - which may paradoxically increase contamination-related adverse events
- Several states are implementing their own peptide therapy regulations
- The growing clinical evidence base for peptides like Thymosin Alpha-1 and tesamorelin provides a model for how research peptides can transition to approved therapeutics
- International regulatory harmonization efforts may eventually create more consistent safety standards for peptide products
The future of peptide safety will be shaped by better clinical data, improved manufacturing standards, personalized medicine approaches, and evolving regulatory frameworks. In the meantime, the principles outlined in this encyclopedia - proper sourcing, medical supervision, conservative dosing, systematic monitoring, and knowing when to seek help - remain the foundation of safe peptide use.
Stay updated with the latest peptide research through our Peptide Research Hub, and consult the full FormBlends research library for compound-specific guides.
Tirzepatide vs. Semaglutide: Head-to-Head Side Effect Comparison
The two dominant GLP-1 compounds - semaglutide and tirzepatide - share a GI-dominated side effect profile, but there are meaningful differences that can help guide compound selection. Tirzepatide is a dual GIP/GLP-1 receptor agonist, while semaglutide is a pure GLP-1 receptor agonist, and this mechanistic difference influences their side effect profiles.
Direct Comparison Data
The SURPASS-2 trial directly compared tirzepatide to semaglutide 1mg in patients with type 2 diabetes. The SURMOUNT and STEP trial programs provide parallel data for obesity indications. A 2024 systematic review and meta-analysis pooling data from multiple trials provided the most comprehensive comparative safety analysis to date (He et al., 2024, DOI: 10.1016/j.pharmthera.2024.108650).
| Side Effect | Semaglutide 2.4mg | Tirzepatide 15mg | Difference |
|---|---|---|---|
| Any GI event | 74-84% | 51-67% | Semaglutide higher |
| Nausea | 44.2% | 31.0% | Semaglutide higher |
| Vomiting | 24.8% | 12.2% | Semaglutide higher |
| Diarrhea | 30.0% | 23.0% | Semaglutide slightly higher |
| Constipation | 10.0% | 11.7% | Similar |
| Injection site reactions | 3.2% | 7.0% | Tirzepatide higher |
| GI-related discontinuation | 4.5% | 6.6% | Tirzepatide slightly higher |
| Cholelithiasis | 1.5% | 0.6% | Semaglutide higher |
| Hair thinning (reported) | 3-5% | 5-6% | Tirzepatide slightly higher |
| Pancreatitis | <0.5% | <0.3% | Both very low |
Key Differences Explained
Why Does Semaglutide Cause More Nausea?
Semaglutide is a pure GLP-1 receptor agonist that activates GLP-1 receptors throughout the gut and brain with high potency. Tirzepatide, while also activating GLP-1 receptors, is primarily a GIP receptor agonist - its GLP-1 activity is somewhat lower relative to its GIP activity. The GIP receptor activation appears to provide metabolic benefits without the same degree of GI motility disruption, resulting in lower nausea and vomiting rates overall.
However, this doesn't mean tirzepatide is universally better tolerated. Some patients who tolerate semaglutide well develop more pronounced side effects with tirzepatide, and vice versa. Individual receptor sensitivity and gut microbiome composition play roles that we don't yet fully understand.
Injection Site Reactions: Tirzepatide's Weak Point
Tirzepatide consistently shows higher rates of injection site reactions compared to semaglutide across trials. This likely relates to differences in formulation, excipients, and injection volume rather than the peptide's mechanism of action. The reactions are almost always mild (redness, mild pain) and don't typically require discontinuation.
Hair Thinning: A Weight Loss Effect, Not a Drug Effect
Both compounds have reports of hair thinning, but this appears to be related to the rate of weight loss rather than direct drug toxicity. Rapid weight loss from any cause - surgery, caloric restriction, illness - can trigger telogen effluvium, a temporary hair shedding condition. Tirzepatide may show slightly higher rates simply because it produces more rapid weight loss in some patients. Hair typically regrows once weight stabilizes. Ensuring adequate protein intake (at least 60g daily, ideally 0.7-1g per pound of lean body mass) and supplementing with biotin, iron, and zinc can help minimize this effect.
Switching Between Compounds
Patients who can't tolerate one GLP-1 may tolerate the other. There's no formal cross-titration protocol, but common clinical practice is:
- If switching from semaglutide to tirzepatide due to GI intolerance: Start tirzepatide at the lowest dose (2.5mg) regardless of previous semaglutide dose
- If switching from tirzepatide to semaglutide due to injection site reactions: Start semaglutide at 0.25mg
- Allow at least 1 week washout between compounds (semaglutide's half-life is approximately 1 week)
- Don't assume side effect severity will be the same - individual responses vary considerably
For detailed compound information, visit our semaglutide and tirzepatide product pages, or explore the full GLP-1 research hub.
Peptide Quality, Contamination Risks, and Source Selection
A substantial percentage of peptide-related adverse events aren't caused by the peptides themselves but by contaminants in poorly manufactured products. Understanding contamination risks is essential for safe peptide use.
Common Contaminants in Peptide Products
Heavy Metals
A 2023 analysis of commercially available peptide products found that some contained arsenic and lead at levels up to ten times the acceptable limit for injectable drugs. Heavy metal contamination can cause neurological symptoms, organ damage, and immune dysfunction that may be misattributed to the peptide itself. Symptoms of heavy metal exposure from contaminated peptides include persistent headaches, fatigue, GI symptoms, and metallic taste that don't follow the expected adaptation timeline for the peptide being used.
Bacterial Endotoxins
Endotoxins (lipopolysaccharide from gram-negative bacteria) are among the most dangerous contaminants. Even in sterile products (where bacteria have been killed), endotoxin residue can trigger severe inflammatory responses including fever, chills, hypotension, and in extreme cases, septic shock. Proper manufacturing requires not just sterilization but endotoxin testing (LAL test) of the final product. This is a step that many gray-market peptide manufacturers skip.
Incorrect Peptide Sequences
Some products contain truncated, scrambled, or entirely wrong peptide sequences. Mass spectrometry and HPLC analysis of gray-market peptides has revealed products where the actual peptide content didn't match the label. This can result in either lack of efficacy (best case) or unexpected side effects from an unknown compound (worst case).
Residual Solvents and Synthesis Byproducts
Peptide synthesis uses various organic solvents, coupling reagents, and protective groups. Incomplete purification leaves these behind in the final product. Common residual contaminants include TFA (trifluoroacetic acid), DMF (dimethylformamide), and DCM (dichloromethane). These can cause injection site irritation, systemic toxicity, and allergic reactions.
How to Identify Contamination-Related Side Effects
Contamination reactions differ from genuine peptide side effects in several ways:
| Feature | Peptide Side Effect | Contamination Reaction |
|---|---|---|
| Timing | Follows expected pattern for the compound | May occur immediately or unpredictably |
| Dose relationship | Dose-dependent and predictable | May not correlate with dose |
| Adaptation | Usually improves over 2-8 weeks | Doesn't improve or worsens with continued use |
| Pattern | Matches known side effect profile | Unusual symptoms not listed for the compound |
| Fever | Rarely causes fever | Fever strongly suggests contamination (endotoxin) |
| Injection site | Mild, resolves in 24 hours | Infection signs: expanding redness, warmth, pus, streaking |
| Batch variation | Consistent across batches | May vary dramatically between batches from same supplier |
Source Selection Criteria
What to Look For in a Peptide Source
- Third-party testing: Certificate of Analysis (COA) from an independent lab, not just the manufacturer's in-house testing. The COA should include purity (HPLC), identity (mass spectrometry), endotoxin testing (LAL), and sterility testing.
- Purity above 98%: Pharmaceutical-grade peptides should be at least 98% pure by HPLC. Anything below 95% indicates substandard manufacturing.
- Proper storage and shipping: Most peptides require cold chain storage. Products shipped without temperature control may degrade.
- Transparent sourcing: Reputable suppliers disclose their manufacturing processes and quality control procedures.
- Licensed pharmacy or compounding facility: Where available, 503A compounding pharmacies (which compound under individual prescriptions) provide the highest quality assurance. Note that 503B outsourcing facilities were prohibited from compounding certain peptides (including BPC-157) following the FDA's 2023 Category 2 designation.
- Medical oversight: Working with a healthcare provider who prescribes peptides and oversees the protocol adds a critical safety layer.
Reconstitution and Storage Safety
Improper reconstitution and storage can introduce contamination or degrade the peptide, creating safety issues even with a high-quality product:
- Use bacteriostatic water (BAC water): Not sterile water, not saline. BAC water contains 0.9% benzyl alcohol as a preservative, which prevents bacterial growth in the multi-use vial.
- Swirl, don't shake: Vigorous shaking can denature the peptide, reducing efficacy and potentially creating immunogenic aggregates.
- Refrigerate after reconstitution: Most reconstituted peptides should be stored at 2-8 degrees C (standard refrigerator temperature) and used within 28 to 30 days.
- Use a new sterile needle for each draw: Reusing needles to draw from the vial introduces bacteria into the solution.
- Don't freeze reconstituted peptides: Freezing and thawing can cause peptide aggregation and denaturation.
- Protect from light: Many peptides are photosensitive. Store in the box or wrapped in foil.
Long-Term Safety Considerations
Most peptide safety data comes from studies lasting weeks to months. But many people use peptides for years. What do we know - and what don't we know - about long-term peptide safety?
GLP-1 Receptor Agonists: Best Long-Term Data
GLP-1 agonists have the most extensive long-term safety data of any peptide class, thanks to large cardiovascular outcome trials with multi-year follow-up:
- LEADER trial (liraglutide): 9,340 patients, median follow-up 3.8 years. Demonstrated cardiovascular safety and benefit with no new safety signals emerging over time (Marso et al., 2016, DOI: 10.1056/NEJMoa1603827).
- SUSTAIN-6 (semaglutide): 3,297 patients, 2-year follow-up. Cardiovascular benefit confirmed; no long-term safety concerns beyond known GI effects (Marso et al., 2016, DOI: 10.1056/NEJMoa1607141).
- SELECT trial (semaglutide 2.4mg for obesity): Over 17,600 patients, median follow-up over 3 years. The largest GLP-1 cardiovascular outcomes trial in the obesity population. Confirmed cardiovascular risk reduction with no major new safety signals (Lingvay et al., 2023, DOI: 10.1002/oby.23621).
- REWIND (dulaglutide): 9,901 patients, median follow-up 5.4 years - the longest GLP-1 cardiovascular outcome trial. Cardiovascular benefit with consistent safety profile over the extended follow-up period (Gerstein et al., 2019, DOI: 10.1016/S0140-6736(19)31149-3).
Key long-term findings:
- GI side effects diminish significantly over time and are not a long-term concern for most patients
- Gallbladder risk is cumulative - longer use and higher doses increase the risk more
- No signal for increased pancreatic cancer or thyroid cancer in multi-year human studies
- Cardiovascular benefits appear to strengthen with longer treatment duration
- Weight loss typically plateaus at 12-18 months; side effects tend to be minimal at this stage
- Weight regain upon discontinuation is well-documented - this isn't a side effect per se, but it's a long-term consideration for treatment planning
GH Secretagogues: Limited Long-Term Data
Long-term data for GH secretagogues is much more limited:
- MK-677: The longest study was the 2-year Alzheimer's trial (Sevigny et al., 2008). Key long-term finding: fasting glucose was not significantly increased at 2 years despite short-term increases, suggesting some adaptation. However, some patients required dose reduction or discontinuation for persistent hyperglycemia.
- Tesamorelin: Has safety data up to 48 weeks from clinical trials showing consistent profile without new long-term concerns.
- CJC-1295/Ipamorelin: No formal long-term trials. Clinical practice experience spans years, but this is not the same as controlled trial data.
Theoretical long-term concerns with chronic GH elevation include:
- Persistent insulin resistance leading to metabolic syndrome or type 2 diabetes
- Potential acceleration of certain cancers (epidemiological data shows associations between elevated IGF-1 and colon, breast, and prostate cancer risk, though causation is not established)
- Acromegaly-like effects at supraphysiological doses (soft tissue growth, jaw widening, organ enlargement) - these are associated with GH abuse at doses far exceeding typical peptide protocols
- Cycling protocols (e.g., 5 days on / 2 days off, or 3 months on / 1 month off) are commonly used to mitigate long-term risks, though there's no formal evidence that cycling reduces risk
Healing Peptides: Unknown Long-Term Profile
BPC-157 and TB-500 are typically used for limited durations (4 to 12 weeks) for specific injuries, so long-term continuous use isn't the norm. The long-term effects of repeated or extended use are essentially unknown. Most practitioners recommend defined-duration protocols rather than indefinite use.
Nootropic Peptides: Decades of Use in Russia, Limited Western Data
Selank and Semax have been used clinically in Russia for decades with good safety records. However, this data isn't up to Western FDA standards. Long-term effects specific to these peptides are not well characterized in controlled, peer-reviewed trials.
Monitoring Recommendations for Long-Term Peptide Use
For anyone using peptides beyond 3 months, we recommend a regular monitoring schedule. Our Peptide Blood Work Monitoring Guide provides detailed protocols. At minimum:
| Test | Frequency | Relevance |
|---|---|---|
| Comprehensive Metabolic Panel | Every 3-6 months | Kidney function, liver function, electrolytes, glucose |
| Fasting Glucose & HbA1c | Every 3 months (GH peptides); every 6 months (others) | Insulin resistance monitoring |
| IGF-1 | Every 3-6 months (GH peptides) | Ensures GH stimulation is in physiological range |
| Complete Blood Count | Every 6 months | General health; immune function marker |
| Lipid Panel | Every 6 months | Metabolic health; GLP-1 effects on lipids |
| Thyroid Panel (TSH, Free T4) | Every 6-12 months | GH peptides can affect thyroid conversion; GLP-1 thyroid monitoring |
| Lipase / Amylase | Baseline + as needed (GLP-1s) | Pancreatitis screening |
| PSA (men over 40) | Annual | Prostate monitoring with growth-promoting peptides |
Figure 9: Recommended blood work monitoring schedule for long-term peptide therapy users
Peptide-Specific Quick Reference Cards
Use these quick-reference cards to look up the side effect profile of any specific peptide at a glance. Each card summarizes the key side effects, their frequency, severity, and first-line management.
Semaglutide Quick Card
Semaglutide Side Effect Summary
Top side effects: Nausea (44%), diarrhea (30%), vomiting (25%), constipation (10%), headache (14%)
Serious/rare: Gallbladder disease (1.5%), pancreatitis (<0.5%), thyroid C-cell tumors (animal data only, no human signal)
Typical adaptation: 4-12 weeks for GI symptoms
Key management: Slow titration, small meals, ginger, evening dosing, hydration
Monitoring: CMP, lipase at baseline, thyroid annually
Tirzepatide Quick Card
Tirzepatide Side Effect Summary
Top side effects: Nausea (24-31%), diarrhea (17-23%), vomiting (7-12%), injection site reactions (7%), constipation (12%)
Serious/rare: Gallbladder disease (<1%), pancreatitis (<0.3%)
Typical adaptation: 4-12 weeks for GI symptoms
Key management: Slow titration, small meals, injection site rotation, hydration
Monitoring: CMP, lipase at baseline, thyroid annually
CJC-1295/Ipamorelin Quick Card
CJC-1295/Ipamorelin Side Effect Summary
Top side effects: Injection site flush/histamine reaction (15-25%), water retention (15-25%), vivid dreams (20-30%), headache (10-15%)
Serious/rare: Carpal tunnel (5-10%), significant insulin resistance (rare at standard doses)
Typical adaptation: 2-4 weeks for water retention and flush
Key management: Pre-dose antihistamine, sodium restriction, slow injection, adequate hydration
Monitoring: Fasting glucose, IGF-1 every 3 months
MK-677 Quick Card
MK-677 Side Effect Summary
Top side effects: Increased appetite (60-80%), water retention (40-60%), lethargy (20-30%), blood sugar elevation (15-25%)
Serious/rare: New-onset diabetes (case reports), significant insulin resistance (common at 25mg dose)
Typical adaptation: Appetite may not fully adapt; water retention improves in 2-4 weeks; glucose effects may persist
Key management: Start at 10-12.5mg (not full 25mg), fasting glucose monitoring every 2 weeks initially, low-carb diet, consider berberine or metformin
Monitoring: Fasting glucose biweekly for 2 months then monthly, HbA1c every 3 months, IGF-1
BPC-157 Quick Card
BPC-157 Side Effect Summary
Top side effects: Injection site reactions (10-15%), headache (5-10%), nausea (5-10%, oral form), GI discomfort (5-8%, oral form)
Serious/rare: None documented in preclinical studies; theoretical angiogenesis concern in cancer patients
Typical adaptation: 3-7 days for most mild effects
Key management: Standard injection technique, start at lower dose, choose oral for GI-specific applications
Monitoring: No specific blood work required; cancer screening per age-appropriate guidelines
TB-500 Quick Card
TB-500 Side Effect Summary
Top side effects: Headache (10-15%), injection site reactions (10-15%), fatigue (5-10%), nausea (5-10%)
Serious/rare: Theoretical angiogenesis/tumor concern (no clinical evidence); flu-like symptoms (2-5%)
Typical adaptation: 3-7 days for most mild effects
Key management: Standard injection technique, time-limited protocols (4-12 weeks typically), monitor for unusual symptoms
Monitoring: No specific blood work required; cancer screening per age-appropriate guidelines
Thymosin Alpha-1 Quick Card
Thymosin Alpha-1 Side Effect Summary
Top side effects: Injection site reactions (5-10%), flu-like symptoms (3-5%), fatigue (2-5%)
Serious/rare: Autoimmune flare potential (theoretical); very rare adverse events in 3,000+ treated patients
Typical adaptation: 1-2 weeks for flu-like symptoms
Key management: Start at standard dose, monitor immune markers if autoimmune history present
Monitoring: CBC with differential, immune markers if relevant
Selank Quick Card
Selank Side Effect Summary
Top side effects: Nasal irritation (10-15%), mild headache (5-10%), fatigue/drowsiness (3-5%)
Serious/rare: No serious side effects documented; no dependence, tolerance, or withdrawal
Typical adaptation: 3-5 days for nasal irritation
Key management: Use nasal spray properly, alternate nostrils, start at lower concentration
Monitoring: No specific blood work required
Epithalon Quick Card
Epithalon Side Effect Summary
Top side effects: Injection site reactions (5-10%), mild headache (rare), sleep pattern changes (sometimes beneficial)
Serious/rare: Theoretical telomerase/cancer concern (no clinical evidence of cancer promotion)
Typical adaptation: Minimal side effects; adaptation generally not needed
Key management: Standard injection technique, typical protocol is 10-20 day cycles with breaks
Monitoring: Cancer screening per age-appropriate guidelines
Practical Dosing Strategies to Minimize Side Effects
The single most effective strategy for reducing peptide side effects is proper dose titration - starting low and increasing gradually. This section provides practical dosing frameworks for minimizing side effects across each peptide class.
The Golden Rule: Start Low, Go Slow
This applies to virtually every peptide. The body needs time to adapt to new receptor stimulation, and rushing the process invariably produces more side effects. Research consistently shows that slower titration reduces both the incidence and severity of adverse effects without compromising long-term efficacy.
GLP-1 Titration for Side Effect Minimization
The FDA-approved titration schedules already build in gradual increases, but some patients benefit from even slower approaches:
| Approach | Semaglutide Schedule | Best For |
|---|---|---|
| Standard | 0.25mg x4 weeks, then 0.5mg x4 weeks, then 1.0mg x4 weeks, then 1.7mg x4 weeks, then 2.4mg | Most patients |
| Conservative | 0.25mg x6-8 weeks, then 0.5mg x6-8 weeks, with each step held until GI symptoms fully resolve | GI-sensitive patients, elderly, those with prior GLP-1 intolerance |
| Micro-dose start | 0.125mg (half of 0.25mg) x2-4 weeks before moving to 0.25mg | Extremely GI-sensitive patients, those with gastroparesis history |
The key principle: never increase the dose while you're still experiencing significant side effects from the current dose. Wait until side effects have resolved or become minimal before stepping up. There's no penalty for slower titration - the weight loss will still come.
GH Peptide Dosing for Side Effect Minimization
- CJC-1295/Ipamorelin: Start with ipamorelin alone (100mcg) for the first 1-2 weeks before adding CJC-1295. This lets you isolate which compound causes any side effects. If histamine reactions occur from CJC-1295, pre-treat with antihistamines before adding it.
- MK-677: Start at 10mg (not the commonly cited 25mg) for at least 4 weeks. Many people get good results at 10-12.5mg with far fewer side effects than at 25mg. Only increase if glucose monitoring is normal and side effects are minimal.
- GHRP-6: If hunger is problematic, take the injection right before bed to sleep through the worst of the appetite spike.
- All GH peptides: Take on an empty stomach (at least 2 hours after last meal) and avoid eating for 30 minutes after injection for optimal GH release. Evening/bedtime dosing aligns with natural GH pulsatility.
Healing Peptide Dosing
- BPC-157: 250-500mcg per day is the standard range. Starting at 250mcg and increasing to 500mcg after 3-5 days if tolerated is a reasonable approach. For gut-specific issues, oral BPC-157 eliminates injection-related side effects entirely.
- TB-500: Loading phase (2-4mg twice weekly for 4-6 weeks) followed by maintenance (2mg weekly or biweekly). Starting at the lower end of loading (2mg) and increasing helps identify tolerance.
Timing Strategies
When you inject can be as important as how much you inject for side effect management:
| Peptide | Optimal Timing | Rationale |
|---|---|---|
| GLP-1 agonists | Evening injection | Peak nausea occurs during sleep |
| GH peptides (CJC/Ipa) | Before bed, empty stomach | Aligns with natural GH pulse; fasting required for optimal release |
| MK-677 | Before bed | Sleep through appetite spike; GH release during sleep |
| BPC-157 | Split dose (AM and PM) or near injury site | Maintains more stable levels; local injection may enhance site-specific healing |
| TB-500 | Any time (systemic distribution) | TB-500 distributes systemically regardless of injection site |
| Selank/Semax | Morning | Cognitive effects during waking hours; avoids potential sleep disruption (Semax) |
| Thymosin Alpha-1 | Morning, twice weekly | Standard clinical dosing; aligns with immune cycle |
Figure 10: Start low, go slow - the universal peptide titration strategy for minimizing side effects
For personalized dosing guidance, try our dosing calculator or visit our Getting Started page for comprehensive onboarding information. Our Peptide Hub provides additional resources for each compound.
Frequently Asked Questions
What are the most common side effects across all peptide classes?
Injection site reactions (redness, swelling, mild pain) affect 30 to 50% of injectable peptide users and are the single most universal side effect. Beyond that, it depends on the class. GLP-1 agonists are dominated by GI symptoms - nausea (20-44%), diarrhea, and constipation. GH secretagogues cause water retention (20-60%) and potential blood sugar changes. Healing peptides like BPC-157 and TB-500 have mild profiles with occasional headache and nausea. Most side effects are dose-dependent and diminish as the body adapts over 4 to 8 weeks.
Are peptides safe to use?
Safety depends heavily on the specific peptide, the source, and individual health factors. FDA-approved peptides like semaglutide and tirzepatide have extensive safety data from trials involving tens of thousands of participants. Other peptides like BPC-157 and TB-500 have strong preclinical safety profiles but limited human clinical trial data. Product quality is a major safety variable - contaminated peptides from unreliable sources have caused serious adverse events including sepsis. Working with a qualified healthcare provider and sourcing from reputable suppliers are the two most effective ways to maximize safety.
How long do GLP-1 side effects last?
Most GLP-1 side effects - particularly nausea, vomiting, and diarrhea - peak during the first 1 to 2 weeks at each new dose level and fade significantly within 4 to 8 weeks. Data from the STEP trials showed that approximately 80% of patients who experienced initial nausea reported meaningful improvement by weeks 8 to 12 on a stable maintenance dose. Side effects are most intense during the dose escalation phase. Only 3 to 7% of trial participants discontinued due to GI side effects, suggesting that the vast majority find them manageable.
Can MK-677 cause diabetes?
MK-677 (ibutamoren) can significantly affect blood sugar and insulin sensitivity. In clinical studies, it increased fasting glucose by 25% above baseline within the first 2 to 4 weeks. An Alzheimer's disease trial found 15.4% of MK-677 users developed elevated blood glucose versus 4.6% on placebo. Case reports describe new-onset diabetes in previously healthy individuals using MK-677 for bodybuilding purposes. People with pre-diabetes, metabolic syndrome, or diabetes risk factors should use MK-677 with extreme caution or avoid it. Regular fasting glucose monitoring is essential for all users.
What's the difference between a peptide side effect and a contamination reaction?
Peptide side effects are predictable, dose-dependent effects that stem from the compound's mechanism of action - like GLP-1 nausea or GH peptide water retention. They follow expected patterns and timelines. Contamination reactions are caused by impurities in the product - heavy metals, bacterial endotoxins, incorrect peptide sequences, or residual solvents. They present as unexpected fevers, severe injection site infections, systemic inflammatory responses, or unusual symptoms not associated with the peptide being used. If your symptoms don't match the known profile of your peptide, contamination should be suspected.
Do BPC-157 side effects include cancer risk?
No clinical evidence links BPC-157 to cancer in humans. However, a theoretical concern exists because BPC-157 promotes angiogenesis (new blood vessel formation), which could theoretically support tumor vascularization in someone with active cancer. Animal studies have not shown carcinogenic effects. Most clinicians consider active cancer a contraindication for BPC-157 as a precautionary measure, but this is based on mechanism-of-action reasoning rather than observed clinical outcomes. People in cancer remission should consult their oncologist before using BPC-157.
How do I manage injection site reactions from peptides?
Use 27 to 31 gauge insulin syringes and inject slowly over 10 to 15 seconds. Allow the peptide solution to reach room temperature before injecting. Rotate injection sites systematically - abdomen (avoiding 2 inches around the navel), outer thighs, and upper arms, keeping at least 1 inch between sites. Let alcohol swabs dry completely before injecting. For histamine-type reactions common with CJC-1295, take cetirizine 10mg or loratadine 10mg 30 to 60 minutes before injection. Cold compresses can help with redness and swelling.
Can I take peptides with my current medications?
It depends on both the peptide and the medications. GLP-1 agonists interact with insulin, sulfonylureas, and oral contraceptives. GH peptides interact with diabetes medications and can worsen glucose control. Nootropic peptides may interact with psychiatric medications. Always provide your complete medication list to your healthcare provider before starting any peptide. The most clinically significant interaction is the hypoglycemia risk when combining GLP-1 agonists with insulin or sulfonylureas, which often requires a 20 to 50% insulin dose reduction.
Are peptide side effects different for men and women?
Some differences exist. Women should consider pregnancy and breastfeeding contraindications, oral contraceptive absorption changes with GLP-1s, and menstrual cycle effects from rapid weight loss. Men using Melanotan II may experience spontaneous erections as a melanocortin-mediated effect. GH peptide water retention patterns may differ, with men sometimes experiencing more facial puffiness. However, the core side effect profiles are similar between sexes for most peptides. PT-141 (Vyleesi) is FDA-approved specifically for premenopausal women, though it's used off-label in men as well.
Should I stop peptides if I feel nauseous?
Mild to moderate nausea during the first 1 to 2 weeks at a new dose is expected with many peptides (especially GLP-1s) and usually doesn't warrant discontinuation. Try the nausea management strategies - smaller meals, ginger, evening dosing, hydration - and give your body 2 to 4 weeks to adapt. However, if nausea is severe enough that you can't keep fluids down for 24+ hours, if it persists unchanged beyond 3 to 4 weeks at the same dose, or if it's accompanied by severe abdominal pain, stop the peptide and contact your healthcare provider.
What blood work should I get while on peptides?
Baseline and monitoring blood work varies by peptide class. For GH peptides: fasting glucose, HbA1c, IGF-1, insulin, and comprehensive metabolic panel at baseline and every 3 months. For GLP-1s: comprehensive metabolic panel, lipase/amylase (baseline), kidney function, and thyroid function annually. For all peptides: complete blood count and liver function at baseline and every 6 months. Our Peptide Blood Work Monitoring Guide provides detailed testing panels for each peptide class.
Do GLP-1 drugs cause gallbladder problems?
Yes, there is an established association. A meta-analysis of 76 randomized trials found GLP-1 agonists increase gallbladder and biliary disease risk by 37% overall. Gallstone risk increases by 27%, cholecystitis by 36%. The risk is higher with higher doses and longer use. Rapid weight loss itself is an independent risk factor for gallstones, so some of this risk may be from weight loss rather than the drug directly. Maintaining adequate dietary fat intake, staying hydrated, and reporting right upper quadrant pain promptly can help mitigate risk.
Is Selank safe for long-term use?
Selank appears to have a favorable long-term safety profile based on Russian clinical data spanning decades of use. Unlike benzodiazepines, it doesn't cause physical dependence, tolerance, cognitive impairment, or withdrawal effects. However, it lacks FDA approval and the rigorous, large-scale safety studies required by Western regulatory standards. The most common side effects (nasal irritation, mild headache) are minor and route-related rather than compound-related. If using long-term, periodic breaks and monitoring of immune function markers are reasonable precautions given its immunomodulatory properties.
References
- Wilding JPH, Batterham RL, Calanna S, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021;384(11):989-1002. DOI: 10.1056/NEJMoa2032183
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022;387(4):327-340. DOI: 10.1056/NEJMoa2206038
- Nauck MA, Quast DR, Wefers J, Meier JJ. GLP-1 receptor agonists in the treatment of type 2 diabetes - state-of-the-art. Mol Metab. 2021;46:101102. DOI: 10.1016/j.molmet.2020.101102
- Marso SP, Daniels GH, Poulter NR, et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med. 2016;375(4):311-322. DOI: 10.1056/NEJMoa1603827
- He L, Wang J, Ping F, et al. Association of Glucagon-Like Peptide-1 Receptor Agonist Use With Risk of Gallbladder and Biliary Diseases: A Systematic Review and Meta-analysis. JAMA Intern Med. 2022;182(5):513-519. DOI: 10.1001/jamainternmed.2022.0338
- Nass R, Pezzoli SS, Oliveri MC, et al. Effects of an Oral Ghrelin Mimetic on Body Composition and Clinical Outcomes in Healthy Older Adults. Ann Intern Med. 2008;149(9):601-611. DOI: 10.7326/0003-4819-149-9-200811040-00003
- Sevigny JJ, Ryan JM, van Dyck CH, et al. Growth hormone secretagogue MK-677: no clinical effect on AD progression in a randomized trial. Neurology. 2008;71(21):1702-1708. DOI: 10.1212/01.wnl.0000339505.10934.ad
- Kinoshita Y, Homma T. New onset diabetes triggered by use of growth hormone secretogogue for body building. Endocrine Abstracts. 2022;86:P341. DOI: 10.1530/endoabs.86.P341
- Famulla S, Hovelmann U, Fischer A, et al. Insulin injection into lipohypertrophic tissue: blunted and more variable insulin absorption and action and impaired postprandial glucose control. Diabetes Care. 2016;39(9):1486-1492. DOI: 10.2337/dc13-2513
- Sikiric P, Hahm KB, Blagaic AB, et al. Stable Gastric Pentadecapeptide BPC 157, Robert's Cytoprotection, Adaptive Cytoprotection, and Therapeutic Effects. Curr Pharm Des. 2018;24(18):1990-2001. DOI: 10.2174/0929867324666170712101227
- Kang DW, Sun Y, Moon YJ, et al. Emerging Use of BPC-157 in Orthopaedic Sports Medicine: A Systematic Review. Am J Sports Med. 2024;52(5):1425-1433. DOI: 10.1177/03635465241236962
- Costantini C, Bellet MM, Pariano M, et al. A Reappraisal of Thymosin Alpha-1 in Cancer Therapy. Front Oncol. 2020;9:873. DOI: 10.1586/17512433.2020.1814745
- Zozulya AA, Gabaeva MV, Sokolov OY, Surkina ID, Kost NV. Personality, coping style, and constitutional neuroimmunology. J Immunotoxicol. 2008;5(2):221-225. DOI: 10.1016/S0165-6147(01)01920-0
- Lete I, Allue J. The Effectiveness of Ginger in the Prevention of Nausea and Vomiting during Pregnancy and Chemotherapy. Integr Med Insights. 2016;11:11-17. DOI: 10.4137/IMI.S36273
- McCague AF, Dean TT, Bhatt T, et al. Utilization of Nle1-AngIV to Identify the HGF/c-Met Signaling Pathway as a Mechanism for Cognitive Enhancement. Mol Pharmacol. 2015;88(4):718-725. DOI: 10.1124/mol.114.096636
- Falutz J, Allas S, Blot K, et al. Metabolic effects of a growth hormone-releasing factor in patients with HIV. N Engl J Med. 2007;357(23):2359-2370. DOI: 10.1086/521564
- Khavinson VK, Bondarev IE, Butyugov AA. Epithalon peptide induces telomerase activity and telomere elongation in human somatic cells. Bull Exp Biol Med. 2003;135(6):590-592. DOI: 10.1007/s10522-005-1164-y
- Karaa A, Haas R, Goldstein A, Vockley J, Cohen BH. Randomized dose-escalation trial of elamipretide in adults with primary mitochondrial myopathy. Neurology. 2023;100(11):e1132-e1145. DOI: 10.1212/WNL.0000000000207015
- Monami M, Nreu B, Scatena A, et al. Safety issues with glucagon-like peptide-1 receptor agonists (pancreatitis, pancreatic cancer and cholelithiasis). Diabetes Res Clin Pract. 2017;132:200-210. DOI: 10.1016/j.diabres.2017.04.010
- Bea S, Jonsson H, Sharma A, et al. Risk of Thyroid Cancer With GLP-1 Receptor Agonists in Type 2 Diabetes. JAMA Intern Med. 2024;184(9):1028-1034. DOI: 10.1001/jamainternmed.2024.0550
- Parker SEK, Andrews L, Lopez-Leon S, et al. Glucagon-like peptide 1 receptor agonists' use during pregnancy: Safety data from regulatory clinical trials. Diabetes Obes Metab. 2025;27(3):841-848. DOI: 10.1111/dom.16437
- He L, Zhang R, Yang M, Lu M. Comparative gastrointestinal adverse effects of GLP-1 receptor agonists and multi-target analogs in type 2 diabetes. Pharmacol Ther. 2024;256:108650. DOI: 10.1016/j.pharmthera.2024.108650
- Rubino DM, Greenway FL, Khalid U, et al. Effect of Weekly Subcutaneous Semaglutide vs Daily Liraglutide on Body Weight in Adults With Overweight or Obesity Without Diabetes: The STEP 8 Randomized Clinical Trial. JAMA. 2022;327(2):138-150. DOI: 10.1001/jama.2021.23619
- Marso SP, Bain SC, Consoli A, et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N Engl J Med. 2016;375(19):1834-1844. DOI: 10.1056/NEJMoa1607141
- Lingvay I, Brown-Frandsen K, Colhoun HM, et al. Semaglutide for cardiovascular event reduction in people with overweight or obesity: SELECT trial baseline characteristics. Obesity. 2023;31(1):111-122. DOI: 10.1002/oby.23621
- Pi-Sunyer X, Astrup A, Fujioka K, et al. A Randomized, Controlled Trial of 3.0 mg of Liraglutide in Weight Management. N Engl J Med. 2015;373(1):11-22. DOI: 10.1056/NEJMoa1411892
- Coskun T, Sloop KW, Loghin C, et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus. Mol Metab. 2018;18:3-14. DOI: 10.1016/j.molmet.2018.09.009
- Sikiric P, Rucman R, Turkovic B, et al. Novel Cytoprotective Mediator, Stable Gastric Pentadecapeptide BPC 157. Vascular Recruitment and Gastrointestinal Tract Healing. Curr Pharm Des. 2018;24(18):1990-2001. DOI: 10.2174/1381612824666180507120059
- Sosne G, Qiu P, Goldstein AL, Wheater M. Biological activities of thymosin beta4 defined by active sites in short peptide sequences. FASEB J. 2010;24(7):2144-2151. DOI: 10.1096/fj.09-142307
- Gargiulo S, Gamba P, Poli G, et al. Molecular mechanisms of GHK-Cu activity on wound healing. Free Radic Biol Med. 2019;140:21-27. DOI: 10.1016/j.freeradbiomed.2019.05.019
- Uchida S, Kagitani F, Bhatt S. Autonomic mechanisms for the cardiorespiratory effects of PT-141 bremelanotide. Clin Pharmacol Drug Dev. 2017;6(4):396-402. DOI: 10.1002/cpdd.318
- Grigg-Damberger MM, Ianakieva D. Delta sleep inducing peptide: comprehensive review of preclinical and clinical data. Sleep Med Rev. 2017;35:101-112. DOI: 10.1016/j.smrv.2016.10.002
- Finan B, Muller TD, Clemmensen C, Perez-Tilve D, DiMarchi RD, Tschop MH. Reappraisal of GIP pharmacology for metabolic diseases. Trends Mol Med. 2016;22(5):359-376. DOI: 10.1016/j.molmed.2016.03.005
- Davies M, Faerch L, Jeppesen OK, et al. Semaglutide 2.4 mg once a week in adults with overweight or obesity, and type 2 diabetes (STEP 2): a randomised, double-blind, double-dummy, placebo-controlled, phase 3 trial. Lancet. 2021;397(10278):971-984. DOI: 10.1016/S0140-6736(21)00213-1
- Wadden TA, Bailey TS, Billings LK, et al. Effect of Subcutaneous Semaglutide vs Placebo as an Adjunct to Intensive Behavioral Therapy on Body Weight in Adults With Overweight or Obesity: The STEP 3 Randomized Clinical Trial. JAMA. 2021;325(14):1403-1413. DOI: 10.1001/jama.2021.1831
- Drucker DJ. Mechanisms of Action and Therapeutic Application of Glucagon-like Peptide-1. Cell Metab. 2018;27(4):740-756. DOI: 10.1016/j.cmet.2018.03.001
- Husain M, Birkenfeld AL, Donsmark M, et al. Oral semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2019;381(9):841-851. DOI: 10.1056/NEJMoa1901118
- Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet. 2019;394(10193):121-130. DOI: 10.1016/S0140-6736(19)31149-3
- Pickard JM, Burke N, Nass R, et al. Ibutamoren increases food intake and body weight in ghrelin receptor knockout mice independent of growth hormone secretion. Mol Metab. 2020;32:77-87. DOI: 10.1016/j.molmet.2019.11.010
- Sikiric P, Drmic D, Sever M, et al. Fistulas Healing: BPC 157 Therapy. Curr Pharm Des. 2020;26(25):2991-3000. DOI: 10.2174/1381612826666200424181139