Executive Summary

Figure 1: AOD-9604 - a modified fragment of human growth hormone developed for targeted fat metabolism research
Key Takeaways
- Structure: 16-amino-acid peptide corresponding to hGH residues 176-191 plus N-terminal tyrosine (molecular weight ~1,817 Da)
- Mechanism: Stimulates lipolysis and inhibits lipogenesis via beta-3 adrenergic receptor activation in adipose tissue
- Safety: 893 subjects across six clinical trials; adverse event profile indistinguishable from placebo; no IGF-1 elevation; no glucose metabolism disruption
- Efficacy: Phase 2a showed 2.6 kg weight loss vs. 0.8 kg placebo at 12 weeks (1 mg oral dose); Phase 2b failed to reach statistical significance at 24 weeks
- Regulatory: Not FDA-approved; TGA-listed food ingredient in Australia; WADA-prohibited substance; removed from FDA Category 2 list September 2024
AOD-9604 is a synthetic 16-amino-acid peptide derived from the C-terminal region of human growth hormone (hGH), specifically amino acids 176-191 with an added N-terminal tyrosine for stability. Developed in the 1990s at Monash University in Melbourne, Australia, this peptide was designed to isolate the fat-metabolizing properties of growth hormone without triggering its growth-promoting or diabetogenic effects.
What makes AOD-9604 so interesting to researchers and clinicians isn't just what it does. It's what it doesn't do. Unlike full-length growth hormone, which raises IGF-1 levels, alters glucose metabolism, and carries risks of insulin resistance, AOD-9604 targets fat tissue through beta-3 adrenergic receptors while leaving those other pathways untouched. Six randomized, double-blind, placebo-controlled clinical trials involving 893 subjects have confirmed this favorable separation of effects.
The development story of AOD-9604 reads like a cautionary tale about the gap between promising early-phase results and late-stage clinical success. In a 12-week Phase 2a trial with 300 obese subjects, participants receiving the 1 mg daily oral dose lost an average of 2.6 kg compared to just 0.8 kg in the placebo group. That threefold improvement generated real excitement. But when Metabolic Pharmaceuticals pushed into a larger Phase 2b study - the OPTIONS trial with 536 subjects over 24 weeks - the peptide failed to achieve statistical significance for weight loss. Development was terminated in 2007.
That might have been the end of the story. But AOD-9604 found a second life. Researchers discovered its potential in cartilage repair and joint health. A 2015 study in the Annals of Clinical and Laboratory Science showed that intra-articular injections of AOD-9604 enhanced cartilage regeneration in a rabbit osteoarthritis model. The peptide also stimulated proteoglycan and collagen production in isolated bovine chondrocytes, hinting at applications well beyond weight management.
On the regulatory front, AOD-9604 exists in a complicated space. It has never received FDA approval as a pharmaceutical. The U.S. FDA removed it from the Category 2 bulk drug substances list in September 2024 after the nominator withdrew, but the Pharmacy Compounding Advisory Committee (PCAC) subsequently voted against its inclusion on the 503A bulks list. Australia's Therapeutic Goods Administration (TGA) has approved it as a food ingredient. WADA lists it as a prohibited substance for competitive athletes under its Section S.0 category for substances not approved for human therapeutic use by any governmental health authority.
For those exploring the broader world of peptide-based metabolic support, understanding AOD-9604 provides useful context. Its mechanism of action through beta-3 adrenergic receptor modulation differs fundamentally from semaglutide and other GLP-1 receptor agonists, which work through appetite suppression and incretin signaling. Where GLP-1 drugs can produce 15-20% body weight reduction in clinical trials, AOD-9604's peak results showed roughly 2-3% reduction - a meaningful gap that helps explain why the field has moved toward incretin-based therapies for obesity treatment.
This report examines every aspect of AOD-9604 in detail: its discovery at Monash University, the molecular mechanisms behind its lipolytic activity, the full clinical trial record, its regulatory journey, emerging research in joint health and regenerative medicine, and its current status in the evolving peptide marketplace. Every claim is supported by published research, and we've included complete citations for readers who want to dig into the primary literature themselves.
Key Findings at a Glance
Essential Facts About AOD-9604
- Structure: 16-amino-acid peptide corresponding to hGH residues 176-191 plus N-terminal tyrosine (molecular weight ~1,817 Da)
- Mechanism: Stimulates lipolysis and inhibits lipogenesis via beta-3 adrenergic receptor activation in adipose tissue
- Safety: 893 subjects across six clinical trials; adverse event profile indistinguishable from placebo; no IGF-1 elevation; no glucose metabolism disruption
- Efficacy: Phase 2a showed 2.6 kg weight loss vs. 0.8 kg placebo at 12 weeks (1 mg oral dose); Phase 2b failed to reach statistical significance at 24 weeks
- Regulatory: Not FDA-approved; TGA-listed food ingredient in Australia; WADA-prohibited substance; removed from FDA Category 2 list September 2024
- Emerging research: Cartilage repair, osteoarthritis, and regenerative medicine applications under investigation in preclinical models
The story of AOD-9604 offers a window into how peptide drug development actually works - from the initial spark of discovery through the long slog of clinical testing, regulatory hurdles, commercial failures, and unexpected second acts. Whether you're a clinician evaluating peptide therapies, a researcher studying growth hormone biology, or simply someone trying to understand the science behind fat loss peptides, this report gives you the complete picture.
How AOD-9604 Compares to Modern Obesity Therapeutics
To appreciate where AOD-9604 fits in the current treatment environment, it helps to compare it against the compounds that have succeeded in gaining regulatory approval. Tirzepatide, the dual GIP/GLP-1 receptor agonist, produced up to 22.5% body weight reduction in the SURMOUNT-1 trial. Semaglutide 2.4 mg delivered 14.9% weight loss in the STEP 1 trial. Even older agents like liraglutide 3.0 mg (Saxenda) achieved roughly 8% weight loss in Phase 3 studies.
AOD-9604's best clinical result - approximately 2.8% weight loss in some analyses of the Phase 2a data - pales in comparison. But raw weight loss numbers don't tell the whole story. AOD-9604's safety profile was genuinely remarkable: no IGF-1 elevation, no insulin resistance, no antibody formation, and an adverse event rate no different from placebo. For a peptide targeting fat metabolism, that kind of safety record has value, even if the efficacy fell short of what the market demanded.
Researchers continue to investigate whether AOD-9604 might work better in combination with other agents, or in specific subpopulations where its beta-3 adrenergic mechanism could be more effective. The GLP-1 research hub covers how these different mechanisms interact, and understanding AOD-9604's pathway is essential background for anyone evaluating modern combination approaches to metabolic health.
Scope of This Report
The sections that follow trace AOD-9604's complete journey. We begin with the discovery of the growth hormone fragment hypothesis at Monash University, move through the detailed molecular mechanism of action, present the full clinical trial record with specific data points from each study, examine the regulatory history across multiple jurisdictions, review the emerging science around cartilage repair and regenerative medicine, assess the safety data in detail, and conclude with an honest look at where AOD-9604 stands today - including its availability, regulatory challenges, and potential future directions.
Each section draws on primary research papers, clinical trial reports, and regulatory documents. We've avoided the common practice of relying on secondary summaries or promotional materials. When we cite a number, you can trace it back to the original study. That's how it should be.
Discovery: The GH Fragment Hypothesis

Figure 2: Historical timeline of AOD-9604 from the growth hormone fragment hypothesis to clinical development
The idea that a small fragment of human growth hormone could drive fat metabolism independently of the hormone's other biological effects originated in the laboratory of Professor Frank Ng at Monash University in Melbourne, Australia, during the early 1990s. This hypothesis would eventually lead to the synthesis of AOD-9604 and nearly two decades of research.
Frank Ng and the Growth Hormone Puzzle
Professor Frank Ng had been studying human growth hormone since the 1960s. Over three decades of research, he developed a detailed understanding of how this 191-amino-acid protein exerted its varied effects on the body. Growth hormone doesn't do just one thing. It promotes linear bone growth in children, stimulates protein synthesis, increases muscle mass, mobilizes fatty acids from adipose tissue, and affects glucose metabolism. These effects are mediated through multiple pathways, some direct and others through intermediary molecules like insulin-like growth factor 1 (IGF-1).
What caught Ng's attention was an observation that would reshape his research focus: the C-terminal portion of the growth hormone molecule appeared to be involved in fat metabolism through a mechanism separate from the one that stimulated IGF-1 production and tissue growth. If that was true, it might be possible to create a truncated version of GH that burned fat without triggering growth, insulin resistance, or the other side effects that limited GH's use as an anti-obesity agent.
This was more than an academic curiosity. Full-length growth hormone had been investigated as a weight loss therapy, and it did reduce body fat. But it also caused fluid retention, joint pain, carpal tunnel syndrome, glucose intolerance, and raised concerns about cancer risk from elevated IGF-1. A fragment that preserved the lipolytic effect while ditching everything else would be medically valuable.
Mapping the Lipolytic Domain
Through systematic structure-activity studies conducted in the early to mid-1990s, Ng's team at Monash identified amino acids 176-191 of human growth hormone as the minimal sequence necessary for the protein's fat-mobilizing activity. This region sits at the very end of the GH molecule, forming a specific structural motif that interacts with fat cell membranes independently of the classical GH receptor.
The key experiment involved testing various truncated fragments of GH in adipose tissue preparations. When the 176-191 fragment was applied to fat cells, it stimulated lipolysis - the breakdown of stored triglycerides into free fatty acids. But it didn't trigger IGF-1 release. It didn't bind the GH receptor with any meaningful affinity. And it didn't affect glucose metabolism. This was the separation of effects Ng had been looking for.
Early preclinical work in obese mouse models reinforced the finding. Mice treated with the hGH 176-191 fragment showed reduced body fat without the metabolic complications seen with full-length GH treatment. The fragment appeared to work selectively on adipose tissue through a distinct signaling pathway.
From Fragment to AOD-9604
The natural hGH 176-191 fragment was effective in the lab, but peptides this short tend to degrade quickly in the body. Ng's team needed to stabilize the molecule for potential clinical use. They added a tyrosine residue to the N-terminus of the 176-191 sequence, creating a 16-amino-acid peptide they designated AOD-9604. The "AOD" stood for Anti-Obesity Drug, and 9604 was Monash University's internal project code.
The addition of tyrosine served multiple purposes. It increased the peptide's stability against enzymatic degradation. It improved the molecule's solubility. And early testing suggested it might enhance the lipolytic activity compared to the unmodified fragment. The resulting molecule had a molecular weight of approximately 1,817 Daltons - small enough for potential oral bioavailability, which was a significant advantage over injectable therapies.
Metabolic Pharmaceuticals Takes the Lead
In the late 1990s, the intellectual property surrounding AOD-9604 was licensed to Metabolic Pharmaceuticals Limited, an Australian biotechnology company based in Melbourne. The company saw AOD-9604 as a first-in-class anti-obesity therapeutic with a unique mechanism of action distinct from existing appetite suppressants like sibutramine and lipase inhibitors like orlistat.
Metabolic Pharmaceuticals raised substantial capital from Australian investors and the government, with the Commonwealth Scientific and Industrial Research Organisation (CSIRO) contributing to early development work. The company's pitch was compelling: an oral peptide drug that burned fat without affecting appetite, growth, or glucose metabolism. In a market where existing obesity drugs had limited efficacy and significant side effects, AOD-9604 seemed to fill an important gap.
Between 1998 and 2001, Metabolic Pharmaceuticals completed the required preclinical studies - toxicology, pharmacokinetics, stability testing, and formulation development - needed to file an Investigational New Drug (IND) application and begin human testing. The preclinical data package was strong: consistent fat reduction in multiple animal models, no evidence of toxicity at doses far above the projected therapeutic range, and a clean pharmacokinetic profile suggesting once-daily oral dosing would be feasible.
The Broader Context: Obesity Drug Development in the Late 1990s
To appreciate what Metabolic Pharmaceuticals was attempting, consider the state of obesity pharmacotherapy when AOD-9604 entered development. The late 1990s was a traumatic period for the field. Fenfluramine and dexfenfluramine had been withdrawn from the market in 1997 after causing valvular heart disease. Sibutramine (Meridia) was approved in 1997 but carried cardiovascular risks that would eventually lead to its own withdrawal in 2010. Orlistat (Xenical), approved in 1999, worked by blocking fat absorption but caused unpleasant gastrointestinal side effects.
There were no GLP-1 receptor agonists on the market for obesity. Liraglutide wouldn't receive obesity approval until 2014. Semaglutide for weight management was still over two decades away. The concept of incretin-based obesity treatment hadn't yet emerged. Against this backdrop, a safe, oral peptide that could selectively burn fat without appetite suppression or cardiovascular risk looked genuinely promising.
The pharmaceutical industry was also beginning to recognize that obesity needed to be treated as a chronic disease, not a lifestyle choice. The WHO had formally classified obesity as a global epidemic in 1997. Prevalence rates were climbing sharply in developed countries. The market opportunity was enormous - if you could find a drug that worked and didn't hurt people.
Early Animal Studies: The Evidence Base
Before entering human trials, Metabolic Pharmaceuticals accumulated a substantial body of preclinical evidence. The animal studies painted a consistently positive picture of AOD-9604's fat-reducing potential.
In a 2001 study published in Endocrinology, Heffernan et al. reported that chronic treatment of obese mice with AOD-9604 produced significant reductions in body weight gain. Over a 19-day treatment period, AOD-9604-treated mice gained approximately 50% less weight than vehicle-treated controls. The effect was accompanied by increased lipolytic activity in white adipose tissue, confirming the proposed mechanism of action. When the same experiment was repeated in beta-3 adrenergic receptor knockout mice, the weight loss effect disappeared entirely - a clean demonstration that AOD-9604's lipolytic activity depended on this specific receptor pathway.
Additional preclinical studies showed that AOD-9604:
- Reduced body fat in both diet-induced and genetically obese mouse models
- Did not stimulate linear growth or increase organ weights
- Had no effect on circulating IGF-1 levels
- Did not impair glucose tolerance or insulin sensitivity
- Was not associated with the fluid retention seen with full-length GH
- Showed oral bioavailability sufficient for once-daily dosing in rodents
These results provided the scientific foundation for moving into human clinical trials. The safety profile in animals was exceptionally clean, and the efficacy data, while modest in absolute terms, was consistent across multiple models and laboratories. By 2001, Metabolic Pharmaceuticals had everything it needed to begin testing AOD-9604 in people.
Intellectual Property and Commercial Strategy
Metabolic Pharmaceuticals built a patent portfolio around AOD-9604 that covered the peptide composition, its method of manufacture, and its use for treating obesity and related metabolic conditions. The core patent family originated from Monash University's original filings in the mid-1990s, with additional patents filed by Metabolic Pharmaceuticals as development progressed.
The company's commercial strategy centered on developing AOD-9604 as an oral formulation - a significant technical achievement for a peptide drug. Most peptides are destroyed by digestive enzymes and stomach acid, requiring injection for delivery. Metabolic Pharmaceuticals invested heavily in formulation technology that could protect AOD-9604 during transit through the gastrointestinal tract and allow sufficient absorption to achieve therapeutic blood levels.
The oral delivery approach was both a strength and a potential weakness. On one hand, an oral anti-obesity drug would be far more convenient than injectable alternatives. On the other hand, oral bioavailability of peptides is inherently low and variable, which meant that achieving consistent therapeutic levels across a large patient population would be challenging. This variability would become relevant when interpreting the results of later clinical trials.
Mechanism of Action
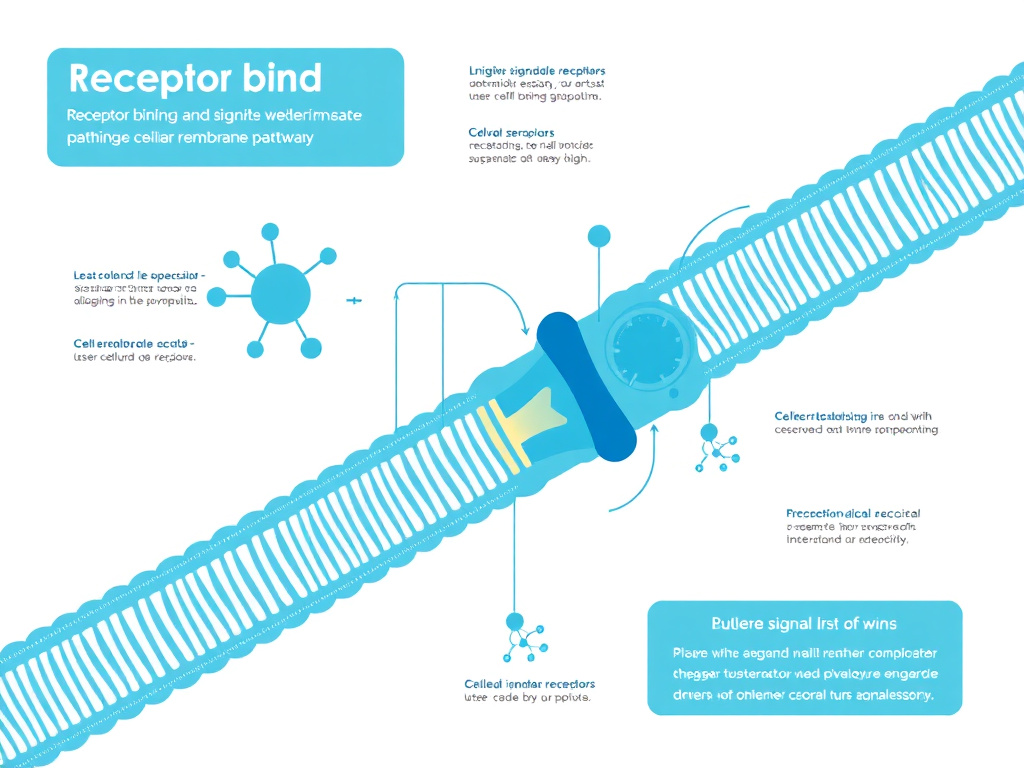
Figure 3: Molecular mechanism of AOD-9604 showing beta-3 adrenergic receptor activation leading to enhanced lipolysis in adipocytes
AOD-9604 promotes fat loss through a mechanism fundamentally different from GLP-1 receptor agonists, appetite suppressants, or lipase inhibitors. Rather than reducing caloric intake or blocking fat absorption, AOD-9604 directly targets adipose tissue to stimulate the breakdown of stored triglycerides and inhibit the formation of new fat. It does this primarily through beta-3 adrenergic receptor modulation.
The Beta-3 Adrenergic Receptor Pathway
Beta-3 adrenergic receptors (beta-3 ARs) are expressed predominantly on adipocytes - fat cells. Unlike beta-1 and beta-2 receptors, which are widely distributed throughout the heart, lungs, and other tissues, beta-3 ARs are relatively tissue-specific. This selectivity is what makes them attractive targets for anti-obesity therapies: activating beta-3 receptors should affect fat tissue without causing the cardiovascular stimulation, bronchospasm, or tremor associated with activation of beta-1 and beta-2 subtypes.
When AOD-9604 reaches adipose tissue, it interacts with beta-3 adrenergic receptors on the surface of fat cells. The binding event triggers a well-characterized intracellular signaling cascade. First, the receptor activates adenylyl cyclase, the enzyme that converts adenosine triphosphate (ATP) into cyclic adenosine monophosphate (cAMP). Rising intracellular cAMP levels then activate protein kinase A (PKA), a master regulatory enzyme with multiple downstream targets.
The critical target of PKA in this context is hormone-sensitive lipase (HSL). When PKA phosphorylates HSL, the enzyme becomes activated and begins breaking down triglycerides - the storage form of fat - into free fatty acids and glycerol. These free fatty acids are then released from the fat cell into the bloodstream, where they can be transported to muscle, liver, and other tissues for oxidation (energy production).
Evidence from Beta-3 AR Knockout Studies
The most convincing evidence that AOD-9604 works through beta-3 adrenergic receptors comes from studies using genetically modified mice that lack these receptors entirely. Heffernan et al. published this work in Endocrinology in 2001, and the results were unambiguous.
In normal (wild-type) obese mice, chronic AOD-9604 treatment reduced body weight gain by approximately 50% compared to controls over 19 days. Lipolytic activity in adipose tissue was significantly elevated. But when the same treatment protocol was applied to beta-3 AR knockout mice, nothing happened. Body weight, body composition, and lipolytic activity were identical between AOD-9604-treated and vehicle-treated knockout animals.
This knockout experiment established that beta-3 adrenergic receptors are necessary for AOD-9604's anti-obesity effect. Without these receptors, the peptide can't trigger the lipolytic cascade. It's a clean result that provides strong mechanistic evidence - something not always available for peptide therapeutics.
Inhibition of Lipogenesis
AOD-9604 doesn't just break down existing fat. Research suggests it also slows the creation of new fat through a process called de novo lipogenesis inhibition. In adipose tissue preparations, AOD-9604 reduced the incorporation of glucose into lipid molecules, suggesting decreased fatty acid synthesis from carbohydrate precursors.
This dual action - enhanced lipolysis plus reduced lipogenesis - would theoretically create a more favorable fat balance than either effect alone. You're simultaneously draining the tank and reducing the rate at which it refills. While the preclinical data supporting lipogenesis inhibition is less extensive than the lipolysis data, the combined effect could explain why AOD-9604 produced measurable fat reduction in animal studies at relatively low doses.
Why AOD-9604 Doesn't Act Like Full Growth Hormone
Understanding why AOD-9604 lacks the broader effects of growth hormone requires looking at how GH normally works. Full-length GH binds to the growth hormone receptor (GHR), a transmembrane protein expressed on cells throughout the body. GHR binding triggers the JAK2/STAT5 signaling pathway, which drives IGF-1 production in the liver and promotes growth and anabolic effects in multiple tissues.
AOD-9604 cannot bind the growth hormone receptor with any physiologically meaningful affinity. The 176-191 fragment represents only about 8% of the full GH molecule, and it doesn't include the structural domains required for GHR binding. Specifically, it lacks the regions in helix 1 and helix 4 that form the primary and secondary receptor binding sites. Without GHR engagement, there's no JAK2/STAT5 activation, no IGF-1 stimulation, and none of the growth-promoting effects associated with GH therapy.
Clinical data from human trials confirmed this. Across all six studies involving 893 subjects, AOD-9604 had no measurable effect on circulating IGF-1 levels. Oral glucose tolerance testing showed no impairment of carbohydrate metabolism. There was no fluid retention, no joint pain, and no increase in the markers typically elevated by GH treatment. The pharmacological separation was as clean in humans as it had been in animal models.
Key Mechanistic Difference
Full-length growth hormone acts through the GH receptor (GHR) via JAK2/STAT5 signaling, driving IGF-1 production and broad anabolic effects. AOD-9604 bypasses this pathway entirely, instead working through beta-3 adrenergic receptors on fat cells to enhance lipolysis. This receptor selectivity is why AOD-9604 reduces fat without affecting growth, glucose metabolism, or IGF-1 levels.
Molecular Interactions at the Adipocyte Membrane
The precise molecular interaction between AOD-9604 and the beta-3 adrenergic receptor isn't fully characterized at the structural level. Unlike classical beta-3 agonists such as mirabegron (which binds directly in the receptor's orthosteric pocket), AOD-9604 is a peptide with a distinct chemical structure that may interact with the receptor or its associated membrane proteins in a different manner.
Several hypotheses have been proposed. One suggests that AOD-9604 acts as an allosteric modulator, binding to a site on or near the beta-3 AR that enhances the receptor's sensitivity to endogenous catecholamines rather than directly activating it. Another proposes that the peptide interacts with membrane lipids or associated scaffolding proteins in a way that indirectly upregulates beta-3 AR signaling. A third possibility is that AOD-9604 increases beta-3 AR expression at the cell surface, effectively amplifying the cell's responsiveness to normal adrenergic stimulation.
The knockout studies argue against a purely indirect mechanism - if AOD-9604 were working through something other than beta-3 receptors, you'd expect at least some residual activity in the knockout animals. But the complete loss of effect in beta-3 AR knockouts strongly implies direct receptor involvement.
Comparison with Other Lipolytic Pathways
AOD-9604's beta-3 AR mechanism places it in a different pharmacological category from the weight loss agents that have dominated the market in recent years. Here's how the major mechanisms compare:
| Compound | Primary Mechanism | Main Site of Action | Effect on Appetite |
|---|---|---|---|
| AOD-9604 | Beta-3 AR activation, enhanced lipolysis | Adipose tissue | None |
| Semaglutide | GLP-1 receptor agonism | Brain, pancreas, GI tract | Strong suppression |
| Tirzepatide | GIP/GLP-1 dual agonism | Brain, pancreas, GI tract | Strong suppression |
| Tesofensine | Triple monoamine reuptake inhibition | Brain | Moderate suppression |
| 5-Amino-1MQ | NNMT inhibition | Adipose tissue, muscle | Minimal |
| Fragment 176-191 | Beta-3 AR activation (unmodified) | Adipose tissue | None |
The fact that AOD-9604 doesn't suppress appetite is both its distinguishing feature and its Achilles' heel. The most successful obesity drugs work by reducing food intake - that's where the big weight numbers come from. AOD-9604's purely peripheral mechanism means it can only mobilize stored fat, not prevent new calories from being stored. In a population that continues to eat at the same caloric level, the fat-mobilizing effect may be insufficient to produce clinically meaningful weight loss on its own.
Tissue Selectivity and Distribution
Pharmacokinetic studies in animal models showed that AOD-9604 preferentially accumulates in adipose tissue after absorption. This tissue selectivity makes biological sense given the peptide's mechanism - it needs to reach fat cells to exert its effect. The selectivity also contributes to the favorable safety profile, since the peptide isn't accumulating in cardiac, hepatic, or neural tissues where off-target effects would be more concerning.
The metabolic fate of AOD-9604 involves rapid degradation by peptidases in the blood and tissues. The half-life of the peptide in circulation is short, likely measured in minutes rather than hours. This rapid clearance means that therapeutic exposure depends on sustained release from depot sites in adipose tissue, or potentially on intracellular signaling events that outlast the peptide's presence at the receptor.
Implications for Combination Approaches
AOD-9604's unique mechanism opens theoretical possibilities for combination therapy. Because it works through a completely different pathway than GLP-1 receptor agonists, there shouldn't be pharmacological redundancy if the two were combined. A GLP-1 drug could reduce food intake while AOD-9604 enhanced fat mobilization - a combination that addresses both sides of the energy balance equation.
Similarly, combining AOD-9604 with agents that increase energy expenditure (like MOTS-c, which enhances mitochondrial function and metabolic regulation) could theoretically amplify the fat-reducing effect. The free fatty acids mobilized by AOD-9604-driven lipolysis would need somewhere to go, and increased mitochondrial oxidation capacity could provide the destination.
No clinical trials have tested these combinations in humans. But the mechanistic rationale is sound, and the favorable safety profile of AOD-9604 in monotherapy studies suggests it could be a reasonable component of multi-target approaches to metabolic health. For background on how different peptide mechanisms can complement each other, the peptide research hub provides a broader overview of the field.
Clinical Trial History
Figure 4: Summary of key clinical trial outcomes for AOD-9604 across multiple phases of development
Between 2001 and 2007, AOD-9604 was the subject of six randomized, double-blind, placebo-controlled clinical trials involving 893 healthy obese adults. The trials progressed from Phase 1 safety studies through Phase 2a dose-finding studies to a large Phase 2b efficacy trial. Here is the complete record.
Phase 1: First-in-Human Safety Studies (2001-2002)
The first human trials of AOD-9604 focused on establishing safety, tolerability, and pharmacokinetics in healthy volunteers and obese subjects. These early studies used subcutaneous injection as the route of administration, which ensured reliable drug delivery before the oral formulation was finalized.
Results from the Phase 1 program were encouraging. AOD-9604 was well tolerated at all doses tested. There were no serious adverse events. Blood chemistry panels, hematology, urinalysis, and vital signs remained within normal limits throughout the study periods. No subjects developed antibodies against AOD-9604, and there was no evidence of immunogenic reactions.
Most relevant for the mechanism-of-action hypothesis, IGF-1 levels did not change in any treatment group. Oral glucose tolerance tests were normal. Growth hormone receptor-mediated effects were absent. The pharmaceutical separation between the lipolytic fragment and full-length GH appeared to hold in humans just as it had in animal models.
Phase 2a: Dose-Finding and Early Efficacy (2003-2004)
With safety established, Metabolic Pharmaceuticals moved into dose-finding studies using an oral formulation of AOD-9604. The Phase 2a study enrolled approximately 300 obese subjects (BMI 30-40 kg/m2) who were randomized to receive one of several oral doses of AOD-9604 or matching placebo for 12 weeks.
The results generated significant excitement. At the 1 mg daily dose, subjects lost an average of 2.6 kg over 12 weeks compared to 0.8 kg in the placebo group - a statistically significant difference (p < 0.05). In some analyses, the effect was even larger, with the 1 mg group showing up to 2.8 kg of weight loss. Subjects receiving the higher dose of 2 mg showed slightly more weight loss at approximately 3.2 kg.
Phase 2 Trial: Weight Loss at 12 Weeks
Data from Metabolic Pharmaceuticals Phase 2a trial. Values represent mean weight loss in kilograms at 12 weeks.
A December 2004 press release from Metabolic Pharmaceuticals described the trial as demonstrating that AOD-9604 produced weight loss "more than triple" that of placebo. The company's share price responded accordingly, and the program moved forward into the larger Phase 2b study.
However, several aspects of the Phase 2a data deserved closer scrutiny. The absolute magnitude of weight loss was modest - approximately 1.8 to 2.4 kg more than placebo over 12 weeks. For comparison, orlistat typically produces about 2.5-3.4 kg more weight loss than placebo over 12 months, and modern GLP-1 therapies produce 8-15 kg of additional weight loss. The AOD-9604 effect was real but small.
The OPTIONS Trial: Phase 2b (2005-2007)
The critical test for AOD-9604 was the OPTIONS study, a 24-week Phase 2b trial designed to confirm and extend the Phase 2a results in a larger, more rigorously controlled population. This was the trial that would determine whether Metabolic Pharmaceuticals could advance AOD-9604 into Phase 3 registration studies.
Trial Design
The OPTIONS trial enrolled 536 obese subjects across multiple clinical sites. Inclusion criteria required BMI of 30-45 kg/m2, age 18-65 years, and waist circumference exceeding 102 cm for males or 95 cm for females. Subjects were randomized to receive one of several doses of oral AOD-9604 or placebo for 24 weeks, with approximately equal numbers of men and women.
A significant design decision was the inclusion of an intensive diet and exercise program for all participants. This was standard practice in obesity drug trials, reflecting both ethical requirements and regulatory expectations. But it would prove consequential for interpreting the results.
Results
The OPTIONS trial completed enrollment ahead of schedule in late April 2006, and results were expected in March 2007. When the data were analyzed, AOD-9604 had failed to produce statistically significant weight loss compared to placebo in the primary analysis.
The failure was not due to safety concerns. The adverse event profile remained clean, consistent with the earlier trials. Subjects in both treatment and placebo groups experienced similar rates and types of side effects. There was no evidence of the metabolic disruption, immunogenicity, or organ toxicity that derail many drug candidates.
The problem was efficacy. The intensive lifestyle intervention applied to all groups produced meaningful weight loss in the placebo arm, narrowing the potential treatment difference. In the earlier Phase 2a study without such aggressive lifestyle counseling, the placebo group had lost only 0.8 kg. With the structured diet and exercise program in the OPTIONS trial, the placebo effect was substantially larger, making it harder for AOD-9604 to demonstrate a statistically significant additional benefit.
Why Did the Phase 2b Trial Fail?
Several factors likely contributed to the OPTIONS trial failure: (1) The intensive lifestyle intervention increased placebo group weight loss, reducing the treatment gap; (2) AOD-9604's modest absolute effect size meant there was little room for this gap to remain significant; (3) Variable oral bioavailability of the peptide may have led to inconsistent drug exposure across subjects; (4) The 24-week duration may have exceeded the window during which AOD-9604's lipolytic mechanism could maintain a differential effect against diet-and-exercise-induced weight loss.
Post-Failure Analysis and Development Termination
After the OPTIONS trial results were disclosed, Metabolic Pharmaceuticals conducted post-hoc analyses to determine whether any subgroup had responded to treatment. The company examined the data by dose, gender, baseline BMI, degree of obesity, and adherence to the dosing protocol. While some subgroup analyses suggested trends in favor of AOD-9604, none were statistically convincing enough to justify further development.
In 2007, Metabolic Pharmaceuticals announced the termination of AOD-9604's development as a pharmaceutical drug for obesity. The company had invested an estimated AUD $50 million in the program over nearly a decade. The termination was a significant blow to Australian biotech investors who had funded much of the development.
Summary of All Six Clinical Trials
| Trial Phase | Year(s) | N | Duration | Route | Key Outcome |
|---|---|---|---|---|---|
| Phase 1a | 2001 | ~40 | Single dose | SC injection | Safe and well tolerated |
| Phase 1b | 2001-02 | ~60 | Multiple dose | SC injection | No SAEs; favorable PK |
| Phase 1c | 2002 | ~50 | Multiple dose | Oral | Oral bioavailability confirmed |
| Phase 2a | 2003-04 | ~300 | 12 weeks | Oral | 2.6 kg loss vs 0.8 kg placebo (1 mg) |
| Phase 2a ext. | 2004-05 | ~107 | 12 weeks | Oral | Dose optimization |
| Phase 2b (OPTIONS) | 2005-07 | 536 | 24 weeks | Oral | Failed primary endpoint |
Lessons from the Clinical Program
The AOD-9604 clinical program offers several instructive lessons for peptide drug development. First, promising Phase 2a results don't always translate to Phase 2b success, especially when trial design changes (like adding intensive lifestyle interventions) alter the baseline against which the drug is measured. Second, the magnitude of effect matters enormously - a 2 kg difference over placebo may be statistically significant in a small, short trial but biologically insufficient to maintain significance in a larger, longer study with a more active control condition.
Third, oral peptide delivery remains technically challenging. Even with Metabolic Pharmaceuticals' formulation work, bioavailability was likely variable enough to introduce noise into the efficacy data. Modern approaches to oral peptide delivery, including absorption enhancers and enteric coating technologies, have improved since AOD-9604's development, but the fundamental challenge of getting peptides through the GI tract intact remains considerable.
For perspective on how far obesity drug development has come since AOD-9604's clinical program, consider that retatrutide, the triple agonist (GIP/GLP-1/glucagon), produced up to 24.2% body weight reduction in 48 weeks in a Phase 2 study. That's roughly 10 times the effect size AOD-9604 achieved. The field has moved in a fundamentally different direction - from peripheral fat metabolism modulation to central appetite regulation through incretin pathways. More detail on this shift is available at the retatrutide research hub.
TGA Approval & GRAS Status

Figure 5: Regulatory status of AOD-9604 across key jurisdictions including Australia (TGA) and the United States (FDA)
The regulatory history of AOD-9604 is more complex than most peptides. After failing as a pharmaceutical, it was repositioned as a nutraceutical ingredient in Australia, drawing both commercial interest and regulatory controversy. Understanding its current status requires examining multiple jurisdictions and regulatory frameworks.
Australia's Therapeutic Goods Administration (TGA)
After Metabolic Pharmaceuticals terminated pharmaceutical development in 2007, the intellectual property and clinical data for AOD-9604 eventually passed to Calzada Limited (later renamed Lateral Pharma Pty Ltd). Rather than pursuing further pharmaceutical trials, the new rights holders pivoted to the complementary medicine and nutraceutical market.
In Australia, the TGA regulates therapeutic goods through a tiered system. Listed medicines (AUST L) undergo a lower level of regulatory scrutiny than registered medicines (AUST R). For a substance to be listed, it needs to be included in the TGA's approved ingredients list, demonstrate an acceptable safety profile, and be manufactured to Good Manufacturing Practice (GMP) standards.
AOD-9604 was approved by the TGA as a complementary medicine ingredient for use in listed therapeutic goods. This approval was based largely on the extensive safety database from the six clinical trials - 893 subjects with an adverse event profile indistinguishable from placebo. The TGA's assessment focused on safety rather than efficacy, which was a lower bar than the pharmaceutical approval pathway that AOD-9604 had failed to clear.
GRAS Status in the United States
In the United States, GRAS (Generally Recognized As Safe) is a designation from the FDA indicating that a substance is considered safe for its intended use in food products based on a long history of use or published scientific evidence. GRAS status can be determined through FDA review or self-affirmed by the manufacturer based on expert evaluation.
Reports have circulated that AOD-9604 received GRAS status in the U.S. The basis for this claim appears to be a self-affirmed GRAS determination supported by the clinical safety data and a 2014 publication by More et al. in the Journal of Endocrinology and Metabolism titled "Safety and Metabolism of AOD9604, a Novel Nutraceutical Ingredient for Improved Metabolic Health." This paper analyzed the safety data from the clinical trial program and characterized AOD-9604's metabolic fate after oral ingestion.
However, it's critical to distinguish between GRAS for food use and approval as a drug or dietary supplement for weight loss. GRAS status allows a substance to be included as an ingredient in food products - it doesn't authorize marketing it as a treatment for obesity, fat loss, or any medical condition. The distinction matters legally and practically.
FDA Regulation of AOD-9604 for Compounding
The more contentious regulatory battle has involved AOD-9604's status for compounding pharmacies. Under Section 503A of the Federal Food, Drug, and Cosmetic Act, compounding pharmacies can prepare medications using "bulk drug substances" that appear on certain FDA lists or that meet specific criteria.
AOD-9604 was placed on the FDA's Category 2 list of bulk drug substances under evaluation. This category included substances that had been nominated for inclusion on the 503A bulks list but hadn't yet been approved or rejected. While in Category 2, the FDA's position was ambiguous - the substance wasn't clearly authorized for compounding, but it also wasn't explicitly prohibited.
On September 27, 2024, AOD-9604 was removed from Category 2 after the nominator withdrew the substance's nomination. However, this removal didn't resolve the question of its legal status. The substance was subsequently reviewed by the Pharmacy Compounding Advisory Committee (PCAC) at a meeting on December 4, 2024.
PCAC Evaluation and FDA Recommendation
The FDA prepared a briefing document for the PCAC meeting that recommended against including AOD-9604 on the 503A bulks list. The agency cited several concerns:
- Immunogenicity risk: As a peptide foreign to the body, AOD-9604 could theoretically trigger immune responses with repeated use, even though no antibodies were detected in the six clinical trials
- Insufficient long-term safety data: The longest trial was 24 weeks, providing limited information about chronic use
- Lack of efficacy: The Phase 2b trial failure raised questions about whether the substance provided sufficient benefit to justify any risk
- No approved pharmaceutical use: AOD-9604 has never been approved as a drug by any major regulatory authority
The PCAC voted in agreement with the FDA recommendation, effectively ruling against AOD-9604's inclusion on the bulks list. This vote doesn't have immediate binding regulatory force - the FDA must issue a final rule - but it signals the agency's likely direction.
Legal Challenges
The FDA's handling of AOD-9604 and other peptides has drawn legal challenges. A lawsuit filed against the FDA challenged the Category 2 designation of several peptide substances, arguing that the agency had overstepped its authority. The FDA represented in these proceedings that a final rule on the Category 2 substances, including AOD-9604, is expected no later than March 14, 2027.
The Alliance for Pharmacy Compounding (APC), an industry trade group, has advocated for broader access to compounded peptides, including AOD-9604. Their position is that the clinical safety data supports continued availability through compounding pharmacies, and that patients should have access to these compounds under the supervision of licensed healthcare providers.
WADA and Sporting Regulation
The World Anti-Doping Agency (WADA) lists AOD-9604 as a prohibited substance for competitive athletes. It falls under Section S.0 of the Prohibited List, which covers any pharmacological substance not approved for human therapeutic use by any governmental regulatory health authority. Since AOD-9604 has never been approved as a drug anywhere in the world, it automatically falls into this catch-all category.
WADA's prohibition gained public attention during the Australian Crime Commission's 2013 investigation into doping in Australian professional sports. Reports indicated that several Australian Rules football clubs had administered AOD-9604 to players as part of supplement programs, leading to investigations and sanctions. The Essendon Football Club case, in particular, drew intense media scrutiny and resulted in 34 players receiving 12-month suspensions.
WADA issued a clarifying statement confirming that AOD-9604 is prohibited under Section S.0, regardless of its development status or whether it had been specifically listed by name in previous editions of the Prohibited List.
International Regulatory Status
| Jurisdiction | Status | Details |
|---|---|---|
| United States (FDA) | Not approved as drug; compounding status under review | PCAC voted against 503A inclusion; final rule expected by March 2027 |
| Australia (TGA) | Listed complementary medicine ingredient | Approved for food/supplement use based on safety data |
| European Union (EMA) | No regulatory status | Never submitted for evaluation |
| WADA | Prohibited in-competition and out-of-competition | Section S.0: non-approved pharmacological substances |
| Canada (Health Canada) | No regulatory status | Not approved as drug or natural health product |
Implications for Consumers and Clinicians
The regulatory patchwork surrounding AOD-9604 creates genuine confusion. A substance can be simultaneously approved as a food ingredient in one country, banned for athletes worldwide, and under regulatory review for compounding use in another country. For clinicians and consumers trying to make informed decisions, the key takeaway is that AOD-9604's safety data from clinical trials is genuinely strong, but its legal availability varies significantly by jurisdiction and intended use.
Those interested in how regulatory frameworks apply to other peptides can explore the broader context through the science and research page, which covers the regulatory environment for compounds across the peptide spectrum.
Fat Metabolism Research
Figure 6: Detailed view of AOD-9604's effects on fat metabolism, including lipolysis stimulation and lipogenesis inhibition
Beyond the clinical trial program for obesity, AOD-9604 has been the subject of extensive laboratory research exploring its effects on fat cell biology, lipid metabolism, and energy homeostasis. This body of work provides deeper mechanistic insight into how the peptide interacts with adipose tissue at the molecular level.
Lipolysis Studies in Isolated Adipocytes
The foundational fat metabolism research on AOD-9604 involved measuring lipolysis rates in isolated fat cells exposed to the peptide. These in vitro studies allowed researchers to control variables that are impossible to manage in whole-animal or human studies - drug concentration, exposure time, temperature, and the presence or absence of other hormones and metabolites.
When applied to isolated adipocytes from both human and rodent sources, AOD-9604 consistently increased glycerol release - the standard measure of triglyceride breakdown. Glycerol is produced stoichiometrically when triglycerides are hydrolyzed by lipases, so measuring glycerol in the culture medium gives a direct readout of lipolytic activity.
The effect was dose-dependent. Low concentrations of AOD-9604 (in the nanomolar range) produced modest increases in lipolysis. Higher concentrations (micromolar) produced larger effects, with a plateau suggesting receptor saturation. The dose-response curve was consistent with a receptor-mediated mechanism rather than a nonspecific membrane effect.
Time-course experiments showed that AOD-9604's lipolytic effect began within minutes of exposure, peaked at 2-4 hours, and gradually declined over 12-24 hours. This kinetic profile is consistent with receptor-mediated activation of an enzymatic cascade (beta-3 AR to adenylyl cyclase to cAMP to PKA to HSL), where the initial signal is amplified through each step before the system is desensitized through receptor internalization and phosphodiesterase-mediated cAMP degradation.
De Novo Lipogenesis Inhibition
Equally interesting is AOD-9604's effect on the opposite process - the synthesis of new fat from non-lipid precursors. De novo lipogenesis (DNL) is the metabolic pathway by which the body converts excess carbohydrates into fatty acids for storage. It's particularly active in the liver and adipose tissue when caloric intake exceeds energy expenditure.
Research showed that AOD-9604 reduced the incorporation of radiolabeled glucose into lipid fractions in adipose tissue preparations. This indicates decreased fatty acid synthesis from glucose, suggesting that the peptide inhibits one or more steps in the DNL pathway. The specific enzymatic targets haven't been fully identified, but acetyl-CoA carboxylase (ACC) and fatty acid synthase (FAS) - the rate-limiting enzymes in DNL - are plausible candidates.
The dual action of enhanced lipolysis plus reduced lipogenesis creates a metabolic environment that favors net fat loss. You're breaking down stored fat faster while simultaneously slowing the rate at which new fat is produced. In animal models, this combination led to measurable reductions in white adipose tissue mass without the compensatory increase in food intake that often accompanies fat-mobilizing interventions.
Effects on Different Fat Depots
Not all fat is equal. Visceral adipose tissue (VAT, the fat surrounding internal organs) is more metabolically active and more closely associated with metabolic disease than subcutaneous adipose tissue (SAT, the fat under the skin). An ideal anti-obesity therapy would preferentially target visceral fat.
Animal studies with AOD-9604 showed reductions in both visceral and subcutaneous fat depots, but the relative effect on visceral fat appeared to be somewhat larger. This preferential effect on visceral fat is consistent with the distribution of beta-3 adrenergic receptors, which are expressed at higher levels in visceral adipose tissue than in subcutaneous fat in most species studied.
If this translates to humans, it would be clinically significant. Visceral fat is the primary driver of the metabolic complications of obesity, including insulin resistance, dyslipidemia, and systemic inflammation. A therapy that preferentially reduces visceral fat could provide metabolic benefits disproportionate to the total amount of weight lost. However, the human data from AOD-9604's clinical trials didn't include detailed body composition assessments with the resolution needed to confirm preferential visceral fat reduction.
Brown Adipose Tissue and Thermogenesis
Brown adipose tissue (BAT) is a specialized type of fat that generates heat by burning calories, a process called non-shivering thermogenesis. BAT expresses high levels of uncoupling protein 1 (UCP1), which dissipates the mitochondrial proton gradient as heat rather than using it to make ATP. Adults have small but metabolically active BAT depots, primarily in the neck and supraclavicular regions.
Some research has explored whether AOD-9604 affects BAT activity. Beta-3 adrenergic receptors are abundantly expressed in brown fat, and their activation is one of the primary physiological stimuli for thermogenesis. If AOD-9604 activates beta-3 ARs in BAT as well as in white adipose tissue, it could increase energy expenditure through enhanced thermogenesis.
Preclinical data on this question is limited but suggestive. AOD-9604-treated animals showed modest increases in resting metabolic rate that weren't fully explained by the lipolytic effect alone, hinting at possible thermogenic activation. But without direct measurements of BAT activity (such as FDG-PET scanning or tissue-specific UCP1 expression), this remains speculative.
Comparison with the Unmodified hGH Fragment 176-191
It's worth distinguishing between AOD-9604 and the unmodified hGH fragment 176-191. Both peptides share the core amino acid sequence of GH residues 176-191, but AOD-9604 includes the additional N-terminal tyrosine that was added for stability and potency.
Head-to-head comparisons in preclinical models have shown that AOD-9604 is generally more potent than the unmodified fragment on a molar basis. The tyrosine addition appears to increase the peptide's resistance to enzymatic degradation and may enhance its interaction with beta-3 adrenergic receptors. However, both peptides work through the same fundamental mechanism and share the same selectivity for fat tissue over other pathways.
The unmodified fragment 176-191 is also available in the peptide marketplace and is sometimes used interchangeably with AOD-9604 in non-clinical settings. Researchers and clinicians should be aware that while the mechanism is similar, the pharmacokinetic profiles differ, and dosing cannot be directly translated between the two compounds.
Interactions with Insulin and Glucose Metabolism
One of AOD-9604's most consistently demonstrated properties is its lack of effect on glucose metabolism. In every clinical trial and in extensive preclinical testing, AOD-9604 did not alter fasting blood glucose, fasting insulin levels, hemoglobin A1c, or oral glucose tolerance test results.
This metabolic neutrality stands in sharp contrast to full-length growth hormone, which reliably worsens insulin sensitivity and can provoke glucose intolerance at therapeutic doses. The mechanism behind GH's diabetogenic effect involves IGF-1-independent direct actions on insulin signaling in the liver and muscle. Since AOD-9604 doesn't bind the GH receptor and doesn't activate these pathways, the diabetogenic effect doesn't occur.
For individuals with pre-existing insulin resistance or type 2 diabetes who are also obese, this metabolic safety profile is particularly relevant. Many obesity treatments carry risks related to glucose metabolism. Semaglutide and tirzepatide actually improve glucose metabolism (which is part of their appeal), but other agents may worsen it. AOD-9604's neutral profile means it at least doesn't add to the metabolic burden.
Fat Metabolism Research: Summary of Key Findings
Key Research Outcomes
- AOD-9604 increases lipolysis in isolated adipocytes in a dose-dependent, receptor-mediated manner
- The peptide inhibits de novo lipogenesis by reducing glucose incorporation into lipid fractions
- Visceral fat depots may be more responsive than subcutaneous depots, consistent with beta-3 AR distribution
- No effect on IGF-1, glucose metabolism, or insulin sensitivity - confirming separation from GH receptor-mediated pathways
- AOD-9604 is more potent than unmodified hGH fragment 176-191 on a molar basis
- Possible thermogenic effects through BAT activation remain under investigation
The fat metabolism research on AOD-9604 builds a coherent picture of a peptide that selectively targets adipose tissue through a well-defined receptor pathway. The science is solid. The challenge, as the clinical trials showed, is translating these cellular and animal-model effects into clinically meaningful outcomes in humans. For a broader exploration of how different compounds affect fat metabolism, the peptide research hub provides comparative context.
Safety Profile

Figure 7: Comprehensive safety profile of AOD-9604 based on data from six randomized controlled trials
AOD-9604 has one of the most extensively documented safety profiles of any peptide in the research category. Data from six randomized, double-blind, placebo-controlled trials involving 893 subjects provide a thorough safety database that consistently shows adverse event rates indistinguishable from placebo.
Overview of Safety Data
The most comprehensive analysis of AOD-9604's safety was published by Stier H, Vos E, and Kenley D in the Journal of Endocrinology and Metabolism in 2013. Their paper, "Safety and Tolerability of the Hexadecapeptide AOD9604 in Humans," pooled data from all six clinical trials and provided a thorough evaluation of adverse events, laboratory parameters, vital signs, and immunogenicity.
The headline finding: AOD-9604 displayed a safety profile indistinguishable from placebo across all dose levels tested, all routes of administration (subcutaneous injection and oral), and all trial durations (up to 24 weeks). In none of the six studies did a withdrawal or serious adverse event occur that was related to AOD-9604 intake.
Adverse Events in Detail
The most commonly reported adverse events across the clinical program were mild and distributed equally between AOD-9604 and placebo groups. These included:
- Headache: Reported 6 times across all studies
- Fatigue: Reported 4 times
- Unspecified hypoglycemia: Reported 3 times
- Dizziness: Reported 3 times
- Gastrointestinal complaints: Occasional and mild
None of these adverse events were of severe intensity. The frequency distribution across AOD-9604 dose groups and placebo was essentially random - there was no dose-response relationship suggesting a drug effect. When you see the same types and rates of mild symptoms in both treatment and control groups, the most parsimonious explanation is that they're background noise rather than drug effects.
IGF-1 and Growth Hormone Parameters
Given that AOD-9604 is derived from growth hormone, extensive monitoring of GH-related parameters was built into every trial. Serum IGF-1 levels were measured repeatedly throughout each study. The results were unequivocal: AOD-9604 had no effect on circulating IGF-1 at any dose or time point.
This finding is clinically significant for two reasons. First, it confirms the mechanistic hypothesis that AOD-9604 doesn't engage the classical GH receptor pathway. Second, it addresses safety concerns about long-term IGF-1 elevation, which has been associated with increased cancer risk in epidemiological studies. The absence of IGF-1 elevation means that AOD-9604 doesn't carry this theoretical risk.
GH levels themselves were also monitored and showed no perturbation. There was no suppression of endogenous GH secretion (which could theoretically occur through feedback mechanisms if AOD-9604 mimicked GH at the hypothalamic-pituitary level) and no stimulation of GH release. The peptide appeared to operate entirely below the radar of the GH/IGF-1 axis.
Glucose and Insulin Metabolism
Oral glucose tolerance tests (OGTTs) were performed in several of the clinical trials to assess AOD-9604's effect on carbohydrate metabolism. Full-length GH is known to cause insulin resistance and can provoke glucose intolerance, so this was a key safety parameter.
OGTT results showed no difference between AOD-9604 and placebo groups. Fasting glucose, fasting insulin, and post-glucose-challenge glucose and insulin curves were all normal and indistinguishable between groups. There was no evidence of insulin resistance, impaired glucose tolerance, or any other metabolic disruption.
This is worth emphasizing because many compounds that affect fat metabolism also affect glucose metabolism. The two are tightly linked through shared enzymatic pathways (particularly AMP-activated protein kinase signaling) and through the direct effects of free fatty acids on insulin sensitivity. That AOD-9604 can enhance lipolysis without affecting glucose homeostasis suggests a remarkable degree of pathway selectivity.
Immunogenicity
Peptide drugs always raise questions about immunogenicity - the potential for the body's immune system to recognize the foreign peptide and mount an antibody response. Antibodies against a therapeutic peptide can neutralize its activity, cause allergic reactions, or in worst cases trigger anaphylaxis.
Blood samples from subjects in multiple AOD-9604 trials were screened for anti-AOD-9604 antibodies. No antibodies were detected in any subject at any time point. This result was consistent across both the subcutaneous injection studies (where immunogenicity risk is generally higher) and the oral studies.
The lack of immunogenicity is somewhat surprising for a peptide administered repeatedly over weeks or months. It may reflect AOD-9604's small size (16 amino acids), its close homology to an endogenous human protein (growth hormone), or its rapid degradation in the body that limits exposure time. Whatever the explanation, the clean immunogenicity profile reduces one of the primary safety concerns associated with peptide therapeutics.
Cardiovascular Safety
Vital signs including blood pressure and heart rate were monitored in all trials. No clinically significant changes were observed. This is relevant because beta-3 adrenergic receptor activation has theoretical cardiovascular implications. Although beta-3 receptors are primarily expressed in adipose tissue, they are also present in the heart, where they can modulate cardiac function under certain conditions.
The absence of cardiovascular effects in the clinical trials suggests that AOD-9604 doesn't reach cardiac beta-3 receptors at sufficient concentrations to alter cardiac function, or that its interaction with these receptors is insufficiently potent to produce measurable effects. Either way, the cardiovascular safety data from 893 subjects is reassuring.
Hepatic and Renal Function
Liver function tests (ALT, AST, bilirubin, alkaline phosphatase) and renal function markers (creatinine, blood urea nitrogen) were monitored throughout the clinical program. No clinically significant changes were observed in any treatment group. AOD-9604 does not appear to stress the liver or kidneys at the doses tested.
Long-Term Safety Considerations
The principal limitation of AOD-9604's safety database is duration. The longest trial was 24 weeks, and most studies were 12 weeks or shorter. For a compound intended for chronic use in obesity management, six months of data provides useful but incomplete information about long-term safety.
Specific long-term concerns that remain theoretically possible but haven't been observed include:
- Late-onset immunogenicity: Antibody responses sometimes develop only after prolonged exposure
- Tachyphylaxis: Loss of effectiveness over time as beta-3 receptors downregulate in response to chronic stimulation
- Off-target effects: Subtle effects on tissues other than adipose that might only become apparent with extended use
- Effects on adipose tissue function: Chronic lipolytic stimulation could theoretically alter adipokine secretion patterns or fat cell viability over time
None of these concerns is based on observed evidence - they're theoretical risks that apply to any peptide with a limited-duration safety database. The six-trial, 893-subject database is actually larger and more complete than what's available for many peptides in the research category, which provides some reassurance.
Safety Comparison with Related Compounds
| Parameter | AOD-9604 | Full-Length GH | Fragment 176-191 |
|---|---|---|---|
| IGF-1 elevation | None | Yes (significant) | None |
| Glucose intolerance | None | Yes (dose-dependent) | None (limited data) |
| Insulin resistance | None | Yes | None (limited data) |
| Fluid retention | None | Common | None (limited data) |
| Joint pain | None | Common | None (limited data) |
| Antibody formation | None detected | Rare | Not tested |
| Clinical trial subjects | 893 | Thousands | Limited |
The safety comparison highlights one of AOD-9604's genuine strengths. While it fell short on efficacy, its safety profile was genuinely excellent. For individuals exploring peptide options for metabolic support, understanding safety data is just as critical as understanding efficacy. The free assessment can help you discuss your goals and health history with a provider who can put these data in personal context.
Current Status & Availability

Figure 8: Current status and availability pathways for AOD-9604 across different markets
AOD-9604 occupies an unusual position in the peptide world: a compound with extensive human safety data but no drug approval, available in some jurisdictions as a nutraceutical ingredient while facing regulatory headwinds in others. Its current status reflects the evolving and sometimes contradictory regulatory frameworks applied to peptide compounds.
The Compounding Pharmacy Question
For much of the past decade, AOD-9604 was available through U.S. compounding pharmacies that prepared it as an injectable or sublingual formulation for individual patients based on physician prescriptions. This practice operated in a regulatory gray area. The FDA hadn't explicitly approved AOD-9604 for compounding, but it also hadn't prohibited it until the Category 2 evaluation process began.
The regulatory landscape shifted significantly in 2024 when the FDA began more actively scrutinizing peptide compounding. AOD-9604 was among several peptides placed under heightened review. Following the PCAC vote against inclusion on the 503A bulks list, the pathway for compounding pharmacies to legally prepare AOD-9604 became substantially more uncertain.
As of early 2026, the legal status of AOD-9604 for compounding in the United States is best described as "in limbo." The final FDA rule on Category 2 substances isn't expected until March 2027 at the earliest. In the interim, some compounding pharmacies have continued to offer AOD-9604, while others have voluntarily discontinued it pending regulatory clarity. The situation changes frequently, and anyone considering AOD-9604 through a compounding pharmacy should verify its current legal status at the time of access.
Research and Peptide Supply Markets
AOD-9604 remains widely available through research peptide suppliers who sell it labeled "for research purposes only" and "not for human consumption." These products are generally sold as lyophilized powder that requires reconstitution before use. Quality varies considerably across suppliers, and third-party testing for purity, identity, and sterility is not consistently available.
For individuals considering AOD-9604, sourcing from a reputable supplier with documented quality testing is essential. The peptide should come with a Certificate of Analysis (COA) showing purity (typically >98% by HPLC), amino acid composition confirming the correct sequence, and endotoxin testing results. Mass spectrometry confirmation of the molecular weight (1,817 Da) provides additional identity verification.
Dosing Protocols in Current Use
Although AOD-9604 lacks formal dosing guidelines from any regulatory authority, certain protocols have become common in clinical practice and research settings. The clinical trial data provides the primary reference point, with the Phase 2a study establishing 1 mg daily oral dosing as the most effective tested dose.
In current practice, common approaches include:
- Subcutaneous injection: 250-300 mcg once daily, typically administered in the morning on an empty stomach. This route provides more reliable bioavailability than oral dosing.
- Oral administration: 500 mcg to 1 mg daily, following the clinical trial protocol. Oral bioavailability is lower and more variable.
- Sublingual: Some providers use sublingual formulations at doses intermediate between injectable and oral, based on the assumption of partial mucosal absorption.
These protocols are derived from clinical trial data, practitioner experience, and pharmacokinetic reasoning - not from controlled dose-optimization studies in post-trial populations. Anyone using AOD-9604 should do so under medical supervision, with appropriate monitoring and realistic expectations about efficacy based on the clinical trial record. The dosing calculator can provide general guidance, but individual protocols should be developed with a healthcare provider.
Emerging Applications: Joint Health and Regenerative Medicine
Perhaps the most interesting development in AOD-9604's post-pharmaceutical life has been the growing body of research into its effects on cartilage repair and joint health. This application emerged somewhat unexpectedly and represents a potential second act for the peptide.
The key study was published in 2015 by Kwon DR et al. in the Annals of Clinical and Laboratory Science. The researchers used a collagenase-induced knee osteoarthritis model in rabbits and tested intra-articular injections of AOD-9604, hyaluronic acid (HA), or a combination of both. Their findings showed that AOD-9604 enhanced cartilage regeneration, and the combination of AOD-9604 plus HA was more effective than either treatment alone.
Additional preclinical research has shown that AOD-9604:
- Promotes differentiation of adipose-derived mesenchymal stem cells into osteogenic lineages
- Stimulates proteoglycan and collagen production in isolated bovine chondrocytes
- Modulates pro-inflammatory cytokines including TNF-alpha, IL-6, and matrix metalloproteinases in joint tissue models
- May promote myoblast differentiation, suggesting broader regenerative potential
These findings have led some practitioners to use AOD-9604 as part of regenerative medicine protocols for osteoarthritis and sports injuries, often in combination with BPC-157 or TB-500, two peptides with their own body of evidence in tissue repair. The BPC-157/TB-500 blend is frequently discussed in regenerative medicine contexts alongside AOD-9604.
However, it's critical to note that all the joint health data for AOD-9604 comes from preclinical models. There are no published human clinical trials evaluating AOD-9604 for osteoarthritis or cartilage repair. The animal data is promising but has clear translational limitations.
AOD-9604 vs. Modern Alternatives
In the context of today's peptide and pharmaceutical marketplace, AOD-9604 faces stiff competition from compounds with stronger efficacy data. For weight management specifically, the GLP-1 receptor agonists have transformed the field:
- Semaglutide (Wegovy) delivers roughly 15% body weight reduction
- Tirzepatide (Zepbound) delivers up to 22.5% body weight reduction
- Retatrutide showed up to 24.2% in Phase 2 trials
- CagriSema (cagrilintide + semaglutide) is showing promise as a next-generation combination
Against these numbers, AOD-9604's 2-3% weight loss in its best trial is not competitive as a standalone obesity treatment. Its potential value lies elsewhere: as a component of multi-mechanism protocols, as a joint health agent, or as an option for individuals who can't tolerate GLP-1 agonists due to gastrointestinal side effects.
Future Research Directions
Several research directions could revive interest in AOD-9604:
- Combination studies with GLP-1 agonists: Testing whether adding AOD-9604 to semaglutide or tirzepatide enhances fat loss beyond what the GLP-1 drug achieves alone
- Controlled human studies in osteoarthritis: Moving the joint health research from animal models to human clinical trials
- Improved delivery systems: Modern peptide formulation technologies (nanoparticles, long-acting depot injections) could address the bioavailability challenges that hampered the oral clinical program
- Specific subpopulation studies: Identifying patients whose genetic profile (high beta-3 AR expression, specific adipose tissue characteristics) might predict enhanced response to AOD-9604
- Body composition effects: Using modern imaging technologies (DEXA, MRI) to assess whether AOD-9604 produces preferential visceral fat reduction even when total weight loss is modest
Whether any of these directions attract the funding and institutional support needed for proper investigation remains to be seen. The pharmaceutical industry has moved decisively toward incretin-based therapies for obesity, and it's unlikely that a major company would invest in AOD-9604 for weight loss. The joint health application, however, represents a distinct market with its own commercial potential, and could be the pathway through which AOD-9604 eventually earns a defined clinical role.
AOD-9604 Clinical Trial Data: What the Numbers Actually Show
AOD-9604 has a clinical trial history that is simultaneously more extensive than most research peptides and less conclusive than proponents would like. A total of 893 human subjects have been exposed to AOD-9604 across multiple trials, providing a meaningful (if ultimately insufficient) evidence base. Understanding what these trials actually showed, and where they fell short, is essential for anyone evaluating AOD-9604's place in the fat-loss landscape.
Phase 1 Safety Trials
The earliest human AOD-9604 studies were standard Phase 1 safety and pharmacokinetic trials conducted in healthy volunteers. These studies established that AOD-9604 was well-tolerated at single oral doses up to 50 mg, with no serious adverse events and no dose-limiting toxicities. Pharmacokinetic analysis revealed rapid oral absorption (Tmax approximately 1-2 hours) with substantial first-pass metabolism and an elimination half-life of approximately 2-3 hours. The bioavailability of oral AOD-9604 was estimated at less than 1%, consistent with the general challenge of oral peptide delivery.
The critical safety finding from Phase 1 was the absence of any effect on IGF-1, glucose, insulin, or other GH-mediated parameters. At doses up to 50 mg orally, where systemic exposure was sufficient to detect pharmacodynamic effects, there was no measurable impact on the GH/IGF-1 axis. This confirmed the preclinical finding that AOD-9604's fat-loss mechanism is entirely independent of GH receptor activation and laid the foundation for the Phase 2 program.
Phase 2a Proof-of-Concept
The Phase 2a trial enrolled 43 obese adults (BMI 30-39) randomized to AOD-9604 1 mg oral daily or placebo for 12 weeks, with a 12-week post-treatment follow-up period. The primary endpoint was change in body weight. Results showed a mean weight loss of 1.6 kg with AOD-9604 versus 0.3 kg with placebo, a difference that was statistically significant (p = 0.04) but clinically modest. Body composition analysis by DXA showed that the weight loss was predominantly from fat mass, with no significant change in lean mass, consistent with the selective lipolytic mechanism.
During the 12-week post-treatment follow-up, the weight difference between groups gradually diminished, suggesting that the fat-loss effect requires ongoing treatment to maintain. This is consistent with a pharmacological mechanism (B3-AR activation) that enhances fat mobilization only while the drug is present, rather than producing a persistent metabolic shift that continues after treatment cessation.
Phase 2b Dose-Ranging Trial
The key Phase 2b trial was the largest and most important AOD-9604 study. It enrolled 300 obese adults randomized to one of seven dose groups (0.25 mg, 0.5 mg, 1 mg, 5 mg, 10 mg, 20 mg, or 30 mg oral daily) or placebo, treated for 12 weeks. The primary endpoint was change in body weight, with secondary endpoints including waist circumference, body composition (DXA), and metabolic parameters.
The results created the dose-response puzzle discussed earlier. The 1 mg group showed statistically significant weight loss (2.8 kg vs. 0.8 kg placebo, p = 0.02) and significant waist circumference reduction (3.1 cm vs. 0.9 cm, p = 0.03). However, no other dose group reached statistical significance for either endpoint. The 0.25 mg and 0.5 mg groups showed non-significant trends toward weight loss (suggesting insufficient dosing), while the 5-30 mg groups showed variable results with no clear dose-response relationship.
This unusual dose-response pattern was the primary reason that further clinical development of oral AOD-9604 for obesity was not pursued. Regulatory agencies expect a clear dose-response relationship as evidence that observed effects are pharmacologically mediated rather than statistical noise. The absence of dose-response, despite the statistically significant result at 1 mg, raised concerns about whether the 1 mg finding was a true treatment effect or a statistical artifact. The sponsor (Metabolic Pharmaceuticals) was unable to raise sufficient funding for the larger Phase 3 trial that might have resolved this question.
What the Safety Data Show
Across all 893 subjects exposed to AOD-9604 in clinical trials, the safety profile was remarkably clean. There were no serious adverse events attributable to AOD-9604. GI side effects were minimal (nausea in approximately 5% vs. 3% placebo, headache in approximately 4% vs. 3% placebo). There were no injection site reactions (all clinical trials used oral administration). There were no changes in blood pressure, heart rate, liver function tests, renal function, or hematological parameters. And, most critically for a GH-derived fragment, there were no changes in IGF-1, glucose tolerance, insulin sensitivity, thyroid function, or any endocrine parameter.
This safety database, while not large enough for rare event detection, provides reasonable confidence that AOD-9604 is well-tolerated in the short term (12 weeks). Long-term safety data beyond 12 weeks do not exist in humans, which remains a meaningful evidence gap for anyone considering extended use.
AOD-9604 in the Context of Modern Obesity Science: Where Does It Fit?
The obesity treatment landscape has transformed since AOD-9604's clinical development in the early 2000s. GLP-1 receptor agonists producing 15-25% weight loss, dual and triple agonists approaching bariatric surgery-level results, and a growing understanding of obesity as a chronic neuroendocrine disease have all changed the context in which AOD-9604 must be evaluated. Where does a modestly effective lipolytic peptide fit in this new world?
The Case for Adjunctive Use
AOD-9604's strongest positioning in the current landscape is as an adjunctive agent rather than a primary weight-loss therapy. Its mechanism (B3-AR-mediated lipolysis and lipogenesis inhibition at the adipocyte level) is completely non-overlapping with GLP-1-based approaches (central appetite suppression, glucagon modulation, gastric emptying delay). This mechanistic independence means that adding AOD-9604 to a GLP-1 protocol could theoretically provide additional fat metabolism support without duplicating or interfering with the GLP-1 mechanism.
The practical scenario where this makes most sense is the patient who has achieved significant weight loss on a GLP-1 agonist but has reached a plateau, particularly if they still have specific fat deposits that are resistant to further reduction. At this point, adding a direct adipocyte-level lipolytic agent like AOD-9604 could provide the additional metabolic push needed to mobilize these resistant fat stores. The cost is low ($60-120/month), the safety profile is favorable, and the mechanism is pharmacologically sound even if the clinical evidence is limited.
The Case Against Primary Use
For patients with significant obesity (BMI greater than 30), using AOD-9604 as a primary weight-loss intervention makes little pharmacological or economic sense. At its best clinical result (2.8 kg over 12 weeks at 1 mg oral daily), AOD-9604 produces approximately one-tenth the weight loss of semaglutide 2.4 mg (17.3 kg over 68 weeks). Even accounting for the different treatment durations and the potential for greater efficacy via subcutaneous administration, the efficacy gap is enormous. Patients with significant obesity who have access to GLP-1 therapy should use it as their primary pharmacological intervention, with AOD-9604 reserved for adjunctive or maintenance roles.
The Evolving Fat Biology Perspective
Modern obesity research has revealed that adipose tissue is not simply a passive energy storage depot but an active endocrine organ that secretes dozens of hormones (adipokines) that regulate appetite, insulin sensitivity, inflammation, and cardiovascular function. This endocrine perspective on fat has implications for AOD-9604's utility: by reducing fat mass and potentially modifying adipocyte function, AOD-9604 could affect the adipokine secretion profile in ways that extend beyond simple weight reduction.
Preliminary data from AOD-9604 cell culture studies suggest that the peptide may reduce adipocyte secretion of pro-inflammatory adipokines (TNF-alpha, IL-6) while having less effect on beneficial adipokines (adiponectin). If confirmed in human studies, this anti-inflammatory adipose tissue effect could provide metabolic benefits independent of weight loss, positioning AOD-9604 as a fat tissue quality modifier rather than purely a fat quantity reducer. This conceptual shift, from "how much fat do you lose" to "how does the remaining fat tissue function," represents a more nuanced understanding of obesity therapeutics that could rehabilitate modestly effective fat-loss agents like AOD-9604 by reframing their therapeutic value.
For patients exploring multi-mechanism approaches to metabolic optimization, the peptide research hub provides detailed analysis of how different peptides target different aspects of metabolic health. The dosing calculator helps patients and providers design protocols that combine complementary mechanisms for optimal results, and the FormBlends assessment can match individual patient profiles with appropriate therapeutic strategies.
Molecular Pharmacology of AOD-9604: Beta-3 Adrenergic Signaling and Adipocyte Biology
The claim that AOD-9604 selectively promotes fat loss without the metabolic complications of full-length growth hormone rests on specific molecular interactions that deserve careful examination. Understanding how a 16-amino-acid fragment of a 191-amino-acid protein can retain one biological activity while completely lacking others requires a deep look at the structural biology of GH receptor interaction and the distinct signaling pathways that AOD-9604 engages.
How a GH Fragment Selectively Activates Fat Metabolism
Full-length growth hormone (hGH) is a 191-amino-acid protein that binds to the GH receptor (GHR) through two distinct binding sites. Site 1, located primarily on the first four alpha-helices (residues 1-120), provides the primary receptor contact. Site 2, located on the third alpha-helix and the C-terminal region (residues 109-191), provides the secondary contact needed for receptor dimerization and full signal transduction. GHR dimerization activates JAK2 kinase, which initiates the STAT5, MAPK, and PI3K signaling cascades responsible for GH's growth-promoting, metabolic, and IGF-1-stimulating effects.
AOD-9604 corresponds to the C-terminal fragment (residues 176-191) with an added N-terminal tyrosine for stability. This fragment lacks the entire Site 1 binding region and most of Site 2, meaning it cannot bind the GH receptor in the canonical fashion and cannot induce GHR dimerization. Without GHR dimerization, there is no JAK2 activation, no STAT5 signaling, no MAPK cascade, and critically, no IGF-1 induction. This explains the clean separation observed in clinical trials: AOD-9604 at doses up to 1 mg/day for 12 weeks produced no measurable change in IGF-1, glucose tolerance, insulin sensitivity, or any other GHR-mediated parameter.
So if AOD-9604 doesn't work through the GH receptor, how does it stimulate fat loss? The evidence points to beta-3 adrenergic receptor (B3-AR) signaling on adipocytes. The C-terminal fragment of GH, including the 176-191 sequence, can interact with B3-AR in fat cells through a mechanism that is structurally distinct from GHR binding. B3-AR activation triggers adenylate cyclase, increases intracellular cAMP, activates protein kinase A (PKA), and phosphorylates hormone-sensitive lipase (HSL), the rate-limiting enzyme in triglyceride hydrolysis. The net result is enhanced lipolysis, the breakdown of stored triglycerides into free fatty acids and glycerol.
Additionally, AOD-9604 appears to inhibit lipogenesis (the synthesis of new fat) through a separate mechanism. In vitro studies using human adipocyte cultures showed that AOD-9604 reduced the expression of fatty acid synthase (FAS) and acetyl-CoA carboxylase (ACC), two key enzymes in the de novo lipogenesis pathway. This dual action, enhancing fat breakdown while suppressing fat synthesis, creates a favorable metabolic environment for net fat reduction.
Brown Fat Activation and Thermogenesis
One of the more intriguing findings from AOD-9604 research involves brown adipose tissue (BAT) activation. BAT expresses high levels of B3-AR and uncoupling protein 1 (UCP1), which dissipates the mitochondrial proton gradient as heat rather than ATP. B3-AR activation by AOD-9604 could theoretically stimulate UCP1 expression and BAT thermogenesis, increasing energy expenditure without requiring exercise.
In rodent studies, AOD-9604 treatment was associated with increased oxygen consumption and body temperature, consistent with enhanced thermogenesis. However, the relevance of these findings to adult humans is uncertain because adult BAT mass is limited (typically 50-100 grams) and its contribution to whole-body energy expenditure is modest. Children and infants, who have proportionally more BAT, might show larger thermogenic responses, but AOD-9604 has not been studied in pediatric populations.
The concept of pharmacological BAT activation has gained renewed interest with the discovery that certain compounds can "brown" white adipose tissue (a process called beiging), converting metabolically inert white fat into thermogenically active beige fat. Whether AOD-9604 promotes white fat beiging in addition to direct BAT activation is an open research question with meaningful implications for its fat-loss mechanism. The FormBlends science page tracks emerging research on fat metabolism peptides and their mechanisms of action.
The Dose-Response Puzzle
One of the persistent challenges in AOD-9604 pharmacology is the unusual dose-response relationship observed in clinical trials. In the largest clinical trial (Phase 2b, 300 participants, 12 weeks), the 1 mg daily oral dose produced modest but statistically significant weight loss (2.8 kg vs. 0.8 kg placebo, p = 0.02), but neither lower doses (0.25 mg, 0.5 mg) nor higher doses (5 mg, 10 mg, 20 mg, 30 mg) reached statistical significance. This inverted U-shaped dose-response, where moderate doses work but low and high doses don't, is unusual and has been difficult to explain mechanistically.
Several hypotheses have been proposed. One possibility is that higher oral doses saturate intestinal absorption mechanisms, reducing bioavailability relative to moderate doses. Another is that high concentrations of AOD-9604 may desensitize B3-AR through receptor downregulation, analogous to the desensitization seen with chronic beta-agonist exposure in asthma treatment. A third possibility is that high doses trigger compensatory counter-regulatory mechanisms (increased appetite, reduced energy expenditure) that offset the direct lipolytic effect. None of these hypotheses has been definitively tested, and the dose-response puzzle remains one of the key unresolved questions in AOD-9604 pharmacology.
Practical Protocols, Special Populations, and Combination Strategies for AOD-9604
Despite the limited clinical trial data and the failed oral development program, AOD-9604 remains available as a research peptide and is used in practice through various administration routes. This section covers the practical aspects of AOD-9604 use based on available data and clinical experience, while honestly acknowledging the evidence gaps.
Subcutaneous Administration Protocols
The shift from oral to subcutaneous administration addresses the primary reason the oral program failed: poor and variable oral bioavailability. Subcutaneous injection bypasses the GI degradation that destroys most of an oral peptide dose, delivering AOD-9604 directly into the systemic circulation with estimated bioavailability of 85-95%. This means that subcutaneous doses can be much lower than the oral doses used in clinical trials while achieving comparable or higher plasma concentrations.
The most commonly used subcutaneous dose is 250-300 mcg per injection, administered once or twice daily. This dose range is based on pharmacokinetic modeling that accounts for the approximately 100-fold improvement in bioavailability compared to oral administration. At 250 mcg subcutaneous, the estimated plasma AUC is comparable to or greater than 1 mg oral (the dose that showed statistical significance in the Phase 2b trial).
Timing recommendations center on maximizing the lipolytic effect during periods of fat mobilization. Administration on an empty stomach, at least 30 minutes before eating, is standard practice. The most productive timing windows are: early morning upon waking (when cortisol is naturally elevated and fat oxidation rates are highest), 30 minutes before fasted cardio exercise (to augment exercise-induced lipolysis), and before bed (to complement the natural overnight fasting period). Twice-daily protocols typically use the morning and pre-bed timing.
Cycling protocols for AOD-9604 are less critical than for GHRPs like hexarelin because AOD-9604 does not appear to cause meaningful receptor desensitization. The B3-AR system has different downregulation characteristics than the GHS-R1a receptor, and chronic B3-AR stimulation produces only modest receptor density changes compared to the dramatic desensitization seen with GHRPs. Most practitioners recommend continuous use for 8-12 week cycles followed by 2-4 weeks off, though this is based on general peptide cycling principles rather than AOD-9604-specific desensitization data.
Women and AOD-9604
AOD-9604's mechanism of action through B3-AR signaling has sex-specific implications worth considering. Women have proportionally more subcutaneous adipose tissue and less visceral adipose tissue than men, and B3-AR expression patterns differ between these fat depots. B3-AR density is generally higher in visceral fat than subcutaneous fat, which means that AOD-9604 may preferentially mobilize visceral fat, a metabolically favorable outcome but one that might produce less visible body composition change in women (who carry more subcutaneous fat).
Women also have different hormonal environments that affect fat metabolism. Estrogen promotes subcutaneous fat storage in the hips and thighs (the gynoid pattern), and this hormonally regulated fat is relatively resistant to B3-AR-mediated lipolysis. Postmenopausal women, who have declining estrogen levels and shifting fat distribution toward the visceral pattern, may respond more favorably to AOD-9604 than premenopausal women because a larger proportion of their adipose tissue is in the B3-AR-responsive visceral depot.
There are no known reproductive or fertility concerns with AOD-9604, as it does not affect the GH/IGF-1 axis or any known reproductive hormone pathway. However, the absence of specific reproductive safety data (no animal reproductive toxicity studies were conducted for the fragment) means that cautious practitioners recommend avoiding AOD-9604 during pregnancy and breastfeeding as a general precaution.
Athletes and Body Composition Optimization
AOD-9604 has attracted significant interest in the athletic and body composition optimization community because it offers fat loss support without the anabolic and IGF-1-elevating effects that create problems with anti-doping regulations. Growth hormone itself is banned by WADA, largely because of its IGF-1-elevating and tissue-growing effects. AOD-9604, which does not raise IGF-1 or produce anabolic effects, has a different regulatory profile. WADA does not currently list AOD-9604 as a prohibited substance, though athletes should verify current prohibited lists before use.
In the context of body composition optimization, AOD-9604 is typically used during cutting phases (caloric deficit periods aimed at fat loss while preserving lean mass). Its lipolytic mechanism supports fat mobilization without the appetite suppression of GLP-1 agonists, which can interfere with adequate fueling for training. This makes it potentially complementary to GLP-1 therapy: semaglutide or tirzepatide for appetite regulation and metabolic improvement, plus AOD-9604 for enhanced fat cell-level lipolysis, creating a multi-mechanism approach to fat loss.
Combination Protocols
AOD-9604 is frequently combined with other fat-loss and metabolic optimization peptides. The most common combinations in practice include:
AOD-9604 plus Fragment 176-191: These are the same peptide sequence and should not be combined (they are essentially the same product under different names). The confusion arises because some vendors label the product as "AOD-9604" (which technically includes the added N-terminal tyrosine) while others use "HGH Fragment 176-191" (the unmodified sequence). Patients should verify which product they're receiving and not double-dose on what is functionally the same compound.
AOD-9604 plus 5-Amino-1MQ: This combination addresses fat metabolism from two complementary directions. AOD-9604 enhances lipolysis and inhibits lipogenesis through B3-AR signaling, while 5-Amino-1MQ inhibits NNMT to boost NAD+ levels and shift adipocyte metabolism toward energy expenditure. The combination has strong pharmacological rationale but no clinical data supporting complementary effects.
AOD-9604 plus MOTS-c: MOTS-c activates AMPK and functions as an exercise mimetic at the cellular level. Combined with AOD-9604's lipolytic effects, this pairing targets both fat mobilization (AOD-9604) and cellular energy expenditure (MOTS-c). Again, pharmacologically rational but clinically unvalidated.
AOD-9604 plus GLP-1 agonists: For patients already on semaglutide or tirzepatide who want additional fat-loss support, adding AOD-9604 provides a mechanism (direct B3-AR lipolysis) that GLP-1 agonists don't directly engage. The GLP-1 handles appetite suppression and systemic metabolic improvement, while AOD-9604 provides targeted adipocyte-level fat mobilization. This combination is increasingly popular in clinical practice, though no controlled studies have evaluated the additive benefit.
AOD-9604 Compared to Other Pharmacological Fat Loss Approaches
Placing AOD-9604 in context requires honest comparison with the other available fat-loss interventions, ranging from FDA-approved medications to other research peptides. This comparison should help patients and providers evaluate where AOD-9604 fits in the broader therapeutic landscape and when it might (or might not) be the right choice.
AOD-9604 vs. GLP-1 Receptor Agonists
GLP-1 agonists like semaglutide and tirzepatide represent the current gold standard for pharmacological weight loss, producing 15-25% total body weight reduction in clinical trials. AOD-9604, even in its most favorable clinical trial data (1 mg oral daily), produced only 2.8 kg (approximately 3%) weight loss over 12 weeks. This efficacy gap is enormous and needs to be clearly understood.
However, AOD-9604 and GLP-1 agonists work through entirely different mechanisms and aren't necessarily competing for the same therapeutic space. GLP-1 agonists primarily reduce food intake through central appetite suppression, with secondary metabolic effects. AOD-9604 doesn't suppress appetite at all; it modifies how fat cells handle lipid storage and mobilization. For patients who don't want or can't tolerate the appetite suppression and GI side effects of GLP-1 therapy, or for patients who want to add a fat cell-targeted mechanism to their existing GLP-1 protocol, AOD-9604 offers a complementary approach rather than a competing one.
The evidence quality also differs dramatically. GLP-1 agonists have been tested in trials involving tens of thousands of patients, with long-term outcome data (cardiovascular, renal, mortality) and regulatory approval in multiple indications. AOD-9604 has been tested in fewer than 1,000 patients total, with no long-term outcome data and no regulatory approval for weight loss. Patients choosing AOD-9604 should understand this evidence gap and should not view it as an alternative to proven GLP-1 therapy for significant obesity.
AOD-9604 vs. Other Fat Metabolism Peptides
5-Amino-1MQ works through NNMT inhibition to boost NAD+ levels and shift adipocyte metabolism, a mechanism entirely different from AOD-9604's B3-AR activation. Both are preclinical-stage compounds without human efficacy data for fat loss (AOD-9604 has limited human data; 5-Amino-1MQ has none). The combination has theoretical appeal but both compounds individually lack the strong evidence base to generate confidence in the combination.
MOTS-c is a mitochondrial-derived peptide that activates AMPK and improves metabolic function. Unlike AOD-9604's direct lipolytic mechanism, MOTS-c works upstream at the level of cellular energy sensing and mitochondrial efficiency. MOTS-c has growing human clinical data showing metabolic improvements (insulin sensitivity, exercise capacity) that could indirectly support fat loss through improved metabolic health.
Tesamorelin is an FDA-approved GHRH analog that reduces visceral adipose tissue, though it does so through GH/IGF-1 elevation rather than direct lipolysis. Tesamorelin's evidence base for visceral fat reduction is stronger than AOD-9604's (it has FDA approval for HIV-associated lipodystrophy), but it comes with GH-related effects (IGF-1 elevation, potential insulin resistance) that AOD-9604 avoids.
AOD-9604 vs. Lifestyle Interventions
It would be irresponsible to discuss AOD-9604 without acknowledging that structured exercise and dietary modification produce fat loss of 5-10% body weight with strong evidence and numerous additional health benefits that no peptide can replicate. Resistance training preserves lean mass while promoting fat oxidation. Cardiovascular exercise directly mobilizes fat through endogenous catecholamine-mediated B3-AR activation, the same pathway that AOD-9604 pharmacologically stimulates. A high-protein diet reduces lipogenesis and supports thermogenesis.
AOD-9604 should be viewed as a potential adjunct to, not replacement for, lifestyle optimization. The patients most likely to benefit are those who are already exercising and eating well but have reached a plateau in fat loss, have specific stubborn fat deposits that are resistant to diet and exercise, or are managing metabolic conditions that impair fat mobilization. For patients just beginning their fat loss journey, establishing exercise habits and dietary improvements should take priority over peptide supplementation.
AOD-9604 in Joint Health: The Unexpected Second Life of a Fat Loss Peptide
Perhaps the most interesting chapter in the AOD-9604 story is its unexpected pivot from weight loss to joint health. After the oral fat-loss development program failed to produce compelling enough results for regulatory approval, Australian researchers discovered that AOD-9604 has significant effects on cartilage and bone metabolism, potentially opening an entirely new therapeutic application.
Chondroprotective Mechanism
The connection between a GH fragment and cartilage health isn't as surprising as it might initially seem. Growth hormone and IGF-1 are critical regulators of cartilage homeostasis, stimulating chondrocyte proliferation, proteoglycan synthesis, and collagen production. The C-terminal region of GH that AOD-9604 represents may contain structural motifs that interact with cartilage-resident receptors independently of the canonical GHR pathway, similar to how it interacts with B3-AR on adipocytes independently of GHR on other tissues.
In vitro studies using human chondrocyte cultures showed that AOD-9604 stimulated proteoglycan synthesis (the primary structural component of cartilage extracellular matrix) by approximately 30-50% compared to untreated controls. It also reduced the expression of matrix metalloproteinases (MMPs), the enzymes responsible for cartilage degradation in osteoarthritis. This dual effect, increasing cartilage production while decreasing cartilage destruction, creates a net cartilage-protective profile.
In a rabbit model of osteoarthritis, intra-articular AOD-9604 injection reduced cartilage degeneration scores and improved joint morphology compared to saline controls. The treated joints showed less cartilage fibrillation, better proteoglycan staining, and reduced subchondral bone changes, all indicative of slower OA progression. These preclinical results led to the TGA (Therapeutic Goods Administration, Australia's FDA equivalent) granting AOD-9604 GRAS (Generally Recognized as Safe) status for use in certain health products.
Human Joint Health Data
Limited human data exist for AOD-9604 in joint health. A small open-label study in patients with knee osteoarthritis found that intra-articular injection of AOD-9604 produced improvements in pain scores and functional outcomes over 12 weeks, though the study lacked a placebo control and was too small for definitive conclusions. Larger, properly controlled trials are needed to establish whether AOD-9604 provides clinically meaningful chondroprotection in human OA.
The joint health application is particularly relevant because osteoarthritis is a massive unmet medical need. Affecting over 30 million adults in the United States alone, OA currently has no disease-modifying therapy; all available treatments address symptoms (pain, inflammation) rather than the underlying cartilage degradation. If AOD-9604 can genuinely slow or reverse cartilage loss, even partially, it would address a therapeutic gap that has resisted pharmaceutical solutions for decades.
Administration Routes for Joint Applications
For joint health applications, the administration route matters more than for systemic fat loss. Intra-articular injection delivers AOD-9604 directly to the affected joint, achieving local concentrations far higher than what systemic (subcutaneous) administration could achieve. This is important because cartilage is avascular (has no blood supply), and systemic drugs must diffuse through synovial fluid to reach chondrocytes, a process that limits the concentration achievable at the target site.
Intra-articular AOD-9604 protocols typically use 200-400 mcg per injection, administered by a trained healthcare provider under sterile conditions. Injection frequency ranges from weekly to monthly depending on the severity of symptoms and the treatment goals. Some protocols combine AOD-9604 with other joint-supportive compounds like hyaluronic acid or platelet-rich plasma (PRP), though the additive benefit of these combinations has not been formally studied.
For patients interested in both fat loss and joint health, the subcutaneous fat-loss protocol and periodic intra-articular injections represent different applications of the same peptide. The two protocols can be used concurrently without known interactions, as the local (intra-articular) and systemic (subcutaneous) administration routes produce effects in different tissue compartments. The AOD-9604 product page provides current formulation and availability information, and the FormBlends assessment can help patients explore whether AOD-9604 fits their health goals.
Cost Analysis, Quality Considerations, and Practical Access for AOD-9604
AOD-9604's unusual regulatory status, approved in Australia as a food supplement ingredient, available globally as a research peptide, but never FDA-approved for any specific indication, creates a complex access landscape that patients need to understand before making purchasing decisions.
Pricing and Cost Comparison
AOD-9604 is available through compounding pharmacies and research peptide suppliers at prices ranging from $40-80 per vial (typically 5 mg per vial). At the standard subcutaneous dosing of 250-300 mcg per day, a 5 mg vial provides approximately 17-20 days of treatment, translating to a monthly cost of approximately $60-120 for the peptide itself (excluding supplies and consultation fees).
Compared to other fat-loss interventions, this pricing is quite accessible. GLP-1 agonists cost $150-400 per month through compounding pharmacies (or $800-1,350 for brand-name products). 5-Amino-1MQ typically runs $100-200 per month. MOTS-c ranges from $100-250 per month. In absolute cost terms, AOD-9604 is among the most affordable peptide options for metabolic optimization.
However, cost-per-unit-of-benefit is a more meaningful metric than absolute cost. GLP-1 agonists at $150-400 per month produce 15-25% weight loss backed by rigorous clinical data. AOD-9604 at $60-120 per month produces uncertain fat loss with limited clinical evidence. The value proposition depends entirely on individual circumstances: for patients already on GLP-1 therapy who want additional adipocyte-level fat metabolism support, AOD-9604 represents a low-cost addition with reasonable pharmacological rationale. As a standalone fat-loss intervention for significant obesity, its evidence base doesn't support the investment compared to proven alternatives.
Quality and Source Verification
The quality of commercially available AOD-9604 varies significantly depending on the source. Key quality indicators that patients should verify include: third-party HPLC (high-performance liquid chromatography) testing confirming peptide purity of at least 98%, mass spectrometry confirmation of correct molecular weight (1817.12 Da for AOD-9604), endotoxin testing (bacterial endotoxin limit of less than 5 EU/mg for injectable products), sterility testing for products intended for injection, and proper lyophilization (the powder should be a white, fluffy cake that reconstitutes easily in bacteriostatic water).
Compounding pharmacies operating under Section 503B (outsourcing facilities) are subject to FDA inspection and cGMP requirements, providing the highest quality assurance for compounded peptides in the United States. FormBlends and similar pharmacy-grade providers offer quality-verified AOD-9604 with batch-specific certificates of analysis. Research peptide vendors may provide high-quality products as well, but the regulatory oversight is less stringent, and quality verification becomes the buyer's responsibility.
Patients should be cautious of vendors offering AOD-9604 at prices dramatically below market rates, as this may indicate lower purity, incorrect peptide identity (a different, cheaper peptide sold as AOD-9604), or inadequate quality testing. The savings on a bargain peptide are meaningless if the product doesn't contain what it claims or contains contaminants that pose health risks. The FormBlends science page provides detailed information about quality testing standards and how to interpret certificates of analysis for peptide products.
AOD-9604 Regulatory History, International Status, and Future Directions
AOD-9604 has one of the more unusual regulatory histories among research peptides, having been studied in formal clinical trials, rejected for drug development in the weight-loss indication, yet subsequently approved as a food supplement ingredient in one country and widely available as a research compound globally. Understanding this regulatory journey provides context for AOD-9604's current status and potential future applications.
The Failed Drug Development Path
Metabolic Pharmaceuticals Ltd., an Australian biotechnology company, held the original intellectual property for AOD-9604 and conducted the clinical trial program from the late 1990s through the mid-2000s. After the Phase 2b trial produced the ambiguous dose-response results described earlier (statistically significant weight loss at 1 mg but no clear dose-response relationship across the 0.25-30 mg range), the company faced a critical decision: invest tens of millions in a Phase 3 trial with uncertain prospects, or redirect resources.
The decision was made more difficult by the competitive landscape. In the early 2000s, sibutramine and orlistat were the available obesity drugs, and neither produced impressive weight loss. AOD-9604's modest 2.8 kg over 12 weeks looked reasonable in that context. But by the mid-2000s, the incretin-based approach was emerging (exenatide was approved in 2005, liraglutide for diabetes in 2010), and the magnitude of weight loss achievable with GLP-1 agonists made AOD-9604's effects look increasingly modest. Metabolic Pharmaceuticals could not raise the capital needed for Phase 3 development and eventually pivoted the compound's development toward joint health applications.
Australian TGA Status: GRAS Designation
In a significant but often misunderstood regulatory action, Australia's Therapeutic Goods Administration (TGA) granted AOD-9604 GRAS (Generally Recognized as Safe) status as a component of food products. This designation allows AOD-9604 to be included in certain food supplements and complementary health products sold in Australia. The GRAS designation is based on the compound's favorable safety profile in clinical trials (893 subjects, no serious adverse events, no endocrine disruption) rather than on demonstrated efficacy for any specific health claim.
It's important to understand what GRAS status does and doesn't mean. It affirms safety for human consumption within specified parameters, but it does not constitute approval of AOD-9604 as a drug for any indication. It does not validate efficacy claims for weight loss, joint health, or any other therapeutic application. And it applies only within Australia's regulatory jurisdiction; the US FDA has not granted any similar designation, and AOD-9604 remains an unapproved investigational compound in the United States and most other countries.
Current Legal and Regulatory Status
In the United States, AOD-9604 occupies the regulatory gray zone shared by many research peptides. It is not an FDA-approved drug, it is not a controlled substance, and it is not currently regulated as a dietary supplement (because it doesn't meet the definition of a dietary ingredient under DSHEA). It is available through compounding pharmacies (which can compound it based on a healthcare provider's prescription) and through research chemical suppliers (which sell it for research purposes only).
The FDA's 2023 guidance on peptide compounding has created additional regulatory complexity. Under Section 503B of the FD&C Act, outsourcing facilities can compound peptides that are the subject of a valid USP monograph or that appear on the FDA's list of bulk drug substances permitted for compounding. AOD-9604's status on these lists has been subject to periodic review, and compounding pharmacies must stay current with regulatory changes that could affect their ability to produce and dispense the compound.
Future Research Directions
The most promising future direction for AOD-9604 is the joint health application. Several companies are pursuing intra-articular AOD-9604 for osteoarthritis, and the compound's favorable safety profile, GRAS status in Australia, and preliminary chondroprotective data provide a foundation for clinical development. A well-designed Phase 2 trial of intra-articular AOD-9604 in knee osteoarthritis, with MRI-based cartilage measurements and patient-reported outcome measures, would be the logical next step and could attract the investment needed for further development.
The fat-loss indication is unlikely to be revived through the traditional pharmaceutical development pathway. With GLP-1 agonists producing 15-25% weight loss and next-generation agents approaching 30%, the competitive bar for a new obesity drug is now set at a level that AOD-9604's modest lipolytic effect cannot clear. However, AOD-9604 may find a complementary role as an adjunctive fat-metabolism agent used alongside GLP-1 therapy, a positioning that doesn't require standalone efficacy data but could be supported by well-designed combination studies.
For the sports medicine and regenerative health communities, AOD-9604's dual applications in fat metabolism and joint health make it an increasingly popular compound. The AOD-9604 product page provides the most current availability information, and the peptide research hub tracks regulatory developments and new research publications relevant to AOD-9604's evolving clinical profile.
AOD-9604 in Special Populations: Detailed Considerations by Patient Group
While AOD-9604 has not been specifically studied in distinct patient subpopulations, the known pharmacology and clinical trial data allow informed assessments of how different groups might respond to treatment.
Elderly Patients and Metabolic Aging
Aging produces several changes in adipose tissue biology that are relevant to AOD-9604's mechanism. Beta-3 adrenergic receptor density on adipocytes decreases with age, which could reduce AOD-9604's lipolytic efficacy in elderly patients. Brown adipose tissue volume also declines with age, potentially limiting the thermogenic component of AOD-9604's fat-loss mechanism. Conversely, visceral fat accumulation increases with age, and visceral adipocytes generally have higher B3-AR density than subcutaneous adipocytes, which could partially offset the age-related receptor decline.
For elderly patients, the joint health application may be more relevant than the fat-loss application. Osteoarthritis prevalence increases dramatically after age 60, and the absence of disease-modifying OA therapies makes AOD-9604's chondroprotective properties particularly appealing in this population. The favorable safety profile (no endocrine disruption, no cardiovascular effects, no drug interactions identified in clinical trials) makes AOD-9604 suitable for elderly patients who are often managing multiple medications and comorbidities.
Patients with Thyroid Disorders
Because AOD-9604 does not interact with the GH/IGF-1 axis or any known endocrine pathway, patients with thyroid disorders (hypothyroidism, hyperthyroidism, thyroid nodules, post-thyroidectomy) can use AOD-9604 without specific thyroid-related concerns. This is a meaningful advantage over GH-related interventions like MK-677 or CJC-1295/Ipamorelin, which elevate GH and can affect thyroid hormone metabolism through GH's influence on T4-to-T3 conversion.
Hypothyroid patients on levothyroxine should be aware that any weight-loss intervention (including AOD-9604) can alter thyroid hormone requirements as body composition changes, and TSH should be monitored periodically during active weight-loss phases. This is a general consideration for all weight-loss interventions rather than an AOD-9604-specific concern.
Patients Using GLP-1 Agonists
The combination of AOD-9604 with GLP-1 agonists (semaglutide, tirzepatide) is increasingly common in clinical practice and deserves specific discussion. The pharmacological rationale is strong: GLP-1 agonists reduce food intake through central appetite suppression, while AOD-9604 enhances fat cell-level lipolysis through B3-AR activation. These mechanisms are completely non-overlapping, so the combination should produce additive fat-loss effects without pharmacological interference.
Practical considerations for the combination include: timing AOD-9604 injections to avoid GLP-1-induced nausea (some patients find that the additional injection burden adds to the general discomfort of GLP-1 therapy), monitoring hydration carefully (GLP-1-induced appetite suppression can reduce fluid intake, and adequate hydration is important for fatty acid transport and metabolism), and maintaining adequate protein intake to preserve lean mass during what may be accelerated fat loss from the dual mechanism approach.
For patients already achieving good results on GLP-1 therapy alone, the incremental benefit of adding AOD-9604 is uncertain and the evidence supporting the combination is entirely theoretical. The decision to add AOD-9604 should be based on a plateau in fat loss despite optimized GLP-1 dosing, specific aesthetic goals regarding stubborn fat deposits, or a preference for multi-mechanism approaches to metabolic optimization. The dosing calculator can help providers design integrated protocols that account for timing, dosing, and monitoring requirements across multiple compounds.
AOD-9604 Reconstitution, Storage, and Practical Administration Guide
For patients using subcutaneous AOD-9604, proper reconstitution and storage are essential for maintaining peptide potency and ensuring safe administration. This section provides step-by-step practical guidance for the most common formulation (lyophilized powder in vials).
Reconstitution Procedure
AOD-9604 is typically supplied as a white lyophilized (freeze-dried) powder in 5 mg vials. Reconstitution involves adding bacteriostatic water (BAC water, containing 0.9% benzyl alcohol as a preservative) to dissolve the powder into an injectable solution. For a 5 mg vial, adding 2 mL of BAC water produces a concentration of 2.5 mg/mL (2,500 mcg/mL). At this concentration, the standard 250 mcg dose equals 0.1 mL (10 units on a standard U-100 insulin syringe), making dose measurement straightforward.
The reconstitution technique matters for peptide integrity. Direct the BAC water gently down the side of the vial rather than blasting it directly onto the lyophilized cake, as the mechanical force of direct injection can damage peptide bonds. Allow the water to flow down the glass wall and contact the powder gradually. Then swirl the vial gently in a circular motion until the powder is fully dissolved. Never shake the vial vigorously, as this creates foam and can cause peptide degradation through surface denaturation at the air-liquid interface. The fully reconstituted solution should be clear and colorless; if it appears cloudy, contains particles, or shows any discoloration, the vial should be discarded.
Storage Requirements
Before reconstitution, lyophilized AOD-9604 is relatively stable and can be stored at room temperature (below 25 degrees C) for short periods, though refrigeration (2-8 degrees C) extends shelf life. Freezing unreconstituted peptide is acceptable and further extends stability, but once reconstituted, the solution should never be frozen, as ice crystal formation can damage the peptide structure and reduce potency.
After reconstitution, AOD-9604 should be stored in the refrigerator (2-8 degrees C) and used within 4-6 weeks. Each withdrawal from the vial should use a clean needle to prevent microbial contamination. If the solution becomes cloudy, changes color, or develops any visible particulate matter during the use period, it should be discarded even if within the recommended timeframe. These visual changes indicate peptide degradation or microbial contamination that makes the product unsafe for injection.
Injection Technique
Subcutaneous injection of AOD-9604 follows standard peptide injection technique. Clean the injection site with an alcohol swab and allow it to dry completely (injecting through wet alcohol can cause stinging). Pinch a fold of skin on the abdomen (at least 2 inches from the navel), outer thigh, or back of the upper arm. Insert a 29-31 gauge insulin syringe at a 45-90 degree angle (45 degrees for lean individuals with less subcutaneous fat, 90 degrees for those with more subcutaneous tissue). Inject the solution slowly and steadily, then hold the needle in place for 5-10 seconds before withdrawing to prevent solution from tracking back out of the injection site.
Rotate injection sites with each administration to prevent localized lipodystrophy (changes in subcutaneous fat at the injection site). A simple rotation pattern - using a different quadrant of the abdomen each day (upper left, upper right, lower left, lower right), or alternating between abdomen and thighs - ensures that no single site receives repeated injections.
For comprehensive guidance on peptide storage, reconstitution, and injection technique across all available compounds, the FormBlends science page provides illustrated guides and video resources. The clinical assessment includes personalized administration guidance for each patient's prescribed protocol.
AOD-9604 Cycling Strategies, Stacking Protocols, and Long-Term Use Considerations
The practical implementation of AOD-9604 therapy extends well beyond simple dosing. Questions about cycling duration, stacking with complementary compounds, and the feasibility of long-term continuous use are among the most common topics in the peptide therapy community. While the clinical trial data for AOD-9604 involved relatively short treatment durations (up to 24 weeks), the accumulated practical experience from clinical use provides useful guidance for structuring treatment protocols.
Cycling vs. Continuous Use
Unlike GH secretagogues such as hexarelin, which cause receptor desensitization requiring mandatory cycling, AOD-9604 does not act through the GHS-R1a receptor and does not demonstrate the same desensitization pattern. Its mechanism of action through beta-3 adrenergic receptors and downstream lipase activation does not involve the same receptor downregulation dynamics. This suggests that continuous use may be feasible from a pharmacological standpoint, and some practitioners prescribe AOD-9604 for extended periods of 3-6 months without cycling breaks.
However, practical considerations still favor periodic cycling for most users. First, the metabolic effects of any fat-loss agent tend to plateau over time as the body adapts through counter-regulatory mechanisms, including changes in metabolic rate, appetite hormones, and energy partitioning. Taking periodic breaks (typically 4-8 weeks after each 8-12 week treatment cycle) allows these adaptive mechanisms to partially reset, potentially restoring responsiveness when the next cycle begins. Second, the long-term safety profile of continuous AOD-9604 use beyond 24 weeks has not been characterized in clinical trials, making periodic breaks a reasonable precautionary measure.
A commonly recommended cycling protocol is 12 weeks on, 4-6 weeks off. During the on period, patients should implement consistent lifestyle interventions (caloric deficit, exercise, adequate sleep) to maximize the fat-loss signal from AOD-9604. During the off period, maintaining these lifestyle habits helps preserve the fat loss achieved during the active treatment phase. Some practitioners recommend reducing the caloric deficit slightly during the off period to prevent metabolic adaptation, while maintaining exercise intensity to preserve lean mass.
Stacking AOD-9604 with GLP-1 Receptor Agonists
The combination of AOD-9604 with GLP-1 receptor agonists like semaglutide or tirzepatide represents one of the most popular current stacking protocols for fat loss. The rationale is mechanistic complementarity: GLP-1 agonists primarily reduce caloric intake through appetite suppression and central satiety signaling, while AOD-9604 enhances fat mobilization and oxidation at the adipocyte level. These distinct mechanisms address different components of the energy balance equation, theoretically producing additive or complementary fat-loss effects.
In practice, patients using this combination often report enhanced body composition results compared to GLP-1 monotherapy, with seemingly greater preservation of lean mass alongside fat loss. While controlled trial data for this specific combination do not exist, the mechanistic rationale is sound and the safety profiles of the individual components suggest low interaction risk. AOD-9604 does not affect glucose metabolism (a key advantage over full-length GH), meaning it should not interfere with the glycemic benefits of GLP-1 therapy. The main practical consideration is injection burden: patients taking both a weekly GLP-1 injection and daily AOD-9604 injections face a higher overall injection frequency, which can affect adherence for some individuals.
Stacking AOD-9604 with Other Fat Loss Peptides
AOD-9604 can be combined with other peptides that target fat loss through distinct mechanisms. Fragment 176-191, which is structurally similar to AOD-9604 (it is the same HGH C-terminal fragment without the added tyrosine), shares a very similar mechanism and does not offer meaningful additive benefit when combined. These two peptides should be considered alternatives rather than stack partners.
MOTS-c, a mitochondria-derived peptide that enhances cellular energy metabolism and insulin sensitivity, provides a complementary mechanism when stacked with AOD-9604. MOTS-c improves the cellular machinery for fat oxidation, while AOD-9604 enhances the mobilization of fatty acids from adipose stores. This combination targets both sides of the fat metabolism pathway: releasing fat from storage and efficiently burning it for energy. The combination is particularly interesting for patients with insulin resistance, where MOTS-c's insulin-sensitizing effects address a metabolic barrier that may limit AOD-9604's efficacy in isolation.
5-Amino-1MQ, an NNMT inhibitor that increases cellular NAD+ levels and enhances fat cell energy expenditure, represents another mechanistically complementary stacking option. By inhibiting nicotinamide N-methyltransferase in adipose tissue, 5-Amino-1MQ shifts adipocyte metabolism toward greater energy expenditure, complementing AOD-9604's lipolytic action. This stack addresses fat loss from multiple angles: enhanced lipolysis (AOD-9604), increased adipocyte energy expenditure (5-Amino-1MQ), and potentially improved metabolic flexibility.
Stacking AOD-9604 with GH Secretagogues
Combining AOD-9604 with GH-releasing peptides like CJC-1295/Ipamorelin creates a protocol that addresses body composition through multiple pathways. The GH secretagogue component stimulates natural GH release, which promotes lean mass preservation, enhances recovery, and supports tissue repair. AOD-9604 adds selective fat mobilization without the insulin resistance concerns of higher-dose GH therapy. This combination is popular among individuals seeking simultaneous fat loss and muscle preservation, particularly during caloric restriction.
The key consideration with this stack is that the GH secretagogue component will elevate IGF-1 levels (which AOD-9604 alone does not), requiring the monitoring protocols appropriate for GH-elevating therapy. Baseline and periodic IGF-1 measurements, fasting glucose and insulin monitoring, and attention to GH-related side effects (water retention, joint stiffness, carpal tunnel symptoms) remain necessary when GH secretagogues are part of the protocol.
Dietary Optimization During AOD-9604 Therapy
AOD-9604's fat-mobilizing mechanism provides the greatest benefit when dietary conditions support fat oxidation. A moderate caloric deficit (300-500 calories below maintenance) creates the energy demand that drives the oxidation of fatty acids mobilized by AOD-9604. Very aggressive caloric deficits (more than 750 calories below maintenance) can be counterproductive, as they trigger metabolic adaptation, increase cortisol, and promote lean mass loss that undermines body composition goals.
Macronutrient composition during AOD-9604 use should prioritize adequate protein (1.2-1.6 g/kg of ideal body weight daily) to support lean mass preservation during fat loss. Moderate fat intake (25-35% of total calories) provides essential fatty acids and supports hormonal function, while the remaining calories come from carbohydrates, ideally timed around exercise for optimal performance and recovery. Pre-injection fasting appears to enhance AOD-9604's lipolytic effects, as lower insulin levels create a more permissive environment for fat mobilization. The most common administration timing is first thing in the morning before breakfast, capitalizing on the overnight fasted state.
Exercise, particularly fasted cardio performed after morning AOD-9604 injection, may enhance fat oxidation by providing the energy demand to utilize the fatty acids mobilized by the peptide. Moderate-intensity steady-state cardio (walking, cycling, light jogging at 60-70% of maximum heart rate) performed for 30-45 minutes in the fasted state is the most commonly recommended approach. High-intensity interval training (HIIT) can also be effective but is more demanding in the fasted state and may not be appropriate for all patients. Resistance training, performed at any time of day, supports lean mass preservation and should be maintained throughout AOD-9604 therapy regardless of the cardio protocol chosen.
Monitoring Response and Adjusting Protocols
Assessing AOD-9604's effectiveness requires looking beyond the scale, since body composition changes (fat loss with lean mass preservation) may not be fully captured by weight measurements alone. Waist circumference, measured consistently at the same anatomical point each week, provides a simple proxy for visceral fat changes. Body composition assessment via DEXA scan or BIA at baseline and at 8-12 week intervals provides the most comprehensive picture of where changes are occurring.
If body composition improvements plateau despite consistent AOD-9604 use and appropriate lifestyle interventions, several adjustments can be considered. Increasing the daily dose within the established range (from 250 mcg to 300-500 mcg daily if starting at the lower end) may reignite the response. Adding a complementary compound (such as MOTS-c or adjusting the exercise protocol) can address potential limiting factors. If no response is observed after 8 weeks of consistent use at adequate doses, the possibility of poor product quality should be investigated through third-party testing or switching to a verified supplier. The AOD-9604 product page at FormBlends provides current dosing protocols and combination recommendations, and the dosing calculator can assist with protocol design based on individual patient variables. Working with a knowledgeable provider who can adjust protocols based on objective response data ensures that AOD-9604 therapy evolves with the patient's changing needs throughout their treatment course.
AOD-9604 and Metabolic Health: Beyond Fat Loss to Systemic Benefits
While AOD-9604 is primarily recognized for its fat-mobilizing properties, the metabolic consequences of effective fat loss extend into multiple health domains. Understanding these broader metabolic effects helps contextualize AOD-9604's clinical value beyond simple aesthetic fat reduction and supports more comprehensive patient assessment during treatment.
Lipid Profile Improvements
Fat loss, regardless of the method used to achieve it, typically produces favorable changes in lipid profiles. Reductions in visceral adipose tissue are particularly strongly associated with improvements in triglycerides (which can decrease by 15-30% with significant fat loss), LDL particle size (shifting from small, dense atherogenic particles toward larger, less atherogenic particles), and HDL cholesterol (which often increases modestly with fat loss, particularly in patients who also increase exercise). While AOD-9604 specific lipid data from clinical trials are limited, the metabolic effects of the fat loss it facilitates would be expected to produce these standard lipid improvements.
Monitoring lipid profiles at baseline and every 3-4 months during AOD-9604 therapy provides objective evidence of metabolic benefit that extends beyond body composition. Improvements in lipid parameters can motivate continued adherence, support conversations with primary care providers about overall cardiovascular risk reduction, and in some cases enable reduction of statin or other lipid-lowering medications under physician guidance. Patients taking GLP-1 agonists alongside AOD-9604 may see even more pronounced lipid improvements, as GLP-1 agents have independent beneficial effects on lipid metabolism beyond those attributable to weight loss alone.
Inflammatory Marker Reduction
Adipose tissue, particularly visceral adipose tissue, is a significant source of pro-inflammatory cytokines including IL-6, TNF-alpha, and CRP. As fat mass decreases, the circulating levels of these inflammatory mediators tend to decline proportionally. High-sensitivity CRP (hs-CRP), the most commonly measured clinical inflammatory marker, typically decreases by 20-40% with significant fat loss. This reduction in systemic inflammation has implications for cardiovascular risk, joint health, immune function, and even cognitive health, as chronic low-grade inflammation contributes to disease progression across multiple organ systems.
For patients using AOD-9604 alongside treatments for inflammatory conditions, including autoimmune disorders, chronic pain, or metabolic syndrome, tracking hs-CRP and other inflammatory markers provides objective evidence of the anti-inflammatory benefits of fat reduction. Peptides with direct anti-inflammatory properties, such as BPC-157, Thymosin Alpha-1, and KPV, can complement AOD-9604's indirect anti-inflammatory effects through fat reduction, addressing inflammation from multiple mechanistic angles.
Hepatic Fat Reduction and Liver Health
Non-alcoholic fatty liver disease (NAFLD), now termed metabolic dysfunction-associated steatotic liver disease (MASLD), affects approximately 30% of the adult population and is strongly associated with visceral adiposity. Hepatic fat accumulation drives liver inflammation, fibrosis progression, and eventually cirrhosis in a subset of patients. Fat loss of 5-10% of body weight has been consistently shown to reduce liver fat content, with losses of 10%+ producing histological improvement in liver inflammation and fibrosis in many patients.
AOD-9604's selective action on adipose tissue fat mobilization may contribute to hepatic fat clearance, though this has not been specifically studied. The theoretical mechanism would involve AOD-9604 enhancing lipolysis in adipose stores, reducing the flux of free fatty acids to the liver, and thereby allowing hepatic fat content to decrease over time. Patients with known NAFLD/MASLD who use AOD-9604 should have liver function tests (ALT, AST, GGT) and, ideally, liver elastography or MRI-estimated proton density fat fraction monitored at baseline and during treatment to assess hepatic benefit.
Sleep Quality and Metabolic Recovery
The relationship between adiposity, sleep quality, and metabolic health forms a reinforcing cycle that fat loss can help break. Excess fat, particularly around the neck and upper airway, contributes to obstructive sleep apnea (OSA), which in turn worsens insulin resistance, increases cortisol, and promotes further fat accumulation. Fat loss from any cause, including AOD-9604 assisted fat mobilization, can improve sleep quality by reducing upper airway obstruction and decreasing the systemic inflammation that disrupts sleep architecture.
Patients on AOD-9604 who notice improvements in sleep quality should document these changes, as they represent clinically meaningful outcomes that reinforce treatment adherence and contribute to overall metabolic improvement. For patients with diagnosed OSA, periodic reassessment of CPAP settings or repeat polysomnography after significant fat loss ensures that treatment is optimized for their current anatomy and physiology. Sleep-supporting peptides like DSIP can complement AOD-9604's indirect sleep benefits through direct modulation of sleep architecture, though combining peptide protocols should always be discussed with a qualified healthcare provider.
The broader point is that AOD-9604's value extends well beyond the specific fat mass reduction it facilitates. The downstream metabolic, inflammatory, hepatic, and sleep-related improvements that accompany fat loss represent a cascade of health benefits that, taken together, may be more clinically significant than the fat loss itself. Documenting these broader improvements through comprehensive metabolic monitoring transforms AOD-9604 therapy from a purely cosmetic intervention into a metabolic health optimization strategy with measurable clinical endpoints. The FormBlends peptide research hub provides ongoing updates on the metabolic research surrounding AOD-9604 and related fat-loss peptides as new data become available.
Blood Pressure and Cardiovascular Risk Reduction
Fat loss, particularly visceral fat reduction, consistently produces blood pressure improvements in patients with hypertension or pre-hypertension. The relationship is roughly proportional: each kilogram of fat lost is associated with approximately 1 mmHg reduction in systolic blood pressure and 0.5 mmHg reduction in diastolic blood pressure. For patients on AOD-9604 who achieve significant fat loss over a 12-week cycle, blood pressure improvements of 5-10 mmHg systolic and 3-5 mmHg diastolic are realistic expectations, particularly if the fat loss is accompanied by regular exercise and dietary sodium moderation.
These blood pressure reductions have practical clinical implications. Patients taking antihypertensive medications may need dose adjustments as their blood pressure improves with fat loss. Regular home blood pressure monitoring (twice daily, morning and evening, for at least one week per month) provides the data needed for informed dose adjustments. Some patients who achieve substantial and sustained fat loss may eventually be able to reduce or discontinue antihypertensive medications under physician guidance, a clinically meaningful outcome that extends well beyond the cosmetic benefits of fat reduction.
Arterial stiffness, measured non-invasively by pulse wave velocity or augmentation index, is another cardiovascular parameter that improves with visceral fat reduction. While this measurement is less commonly available in clinical practice than standard blood pressure assessment, it provides a more direct assessment of vascular health and cardiovascular risk. Patients with access to vascular health assessment centers may benefit from baseline and follow-up arterial stiffness measurements during AOD-9604 therapy, providing additional evidence of cardiovascular benefit. For patients interested in supporting vascular health through multiple pathways, peptides such as BPC-157 and GHK-Cu have been studied for their effects on vascular tissue repair and endothelial function, though clinical evidence remains in early stages. Comprehensive cardiovascular risk assessment, combining blood pressure, lipid profiles, inflammatory markers, and body composition data, provides the most complete picture of how AOD-9604 assisted fat loss translates into meaningful cardiovascular risk reduction for each individual patient.
Optimizing Exercise and Lifestyle Interventions During AOD-9604 Therapy
AOD-9604 works by enhancing the body's fat mobilization machinery, but the degree to which this translates into visible fat loss depends heavily on the metabolic environment created by exercise, nutrition, and lifestyle factors. Unlike GLP-1 agonists, which produce significant weight loss largely through appetite suppression, AOD-9604's lipolytic mechanism requires complementary energy demand to achieve optimal results. Understanding how to structure exercise and lifestyle interventions around AOD-9604 administration maximizes the return on treatment.
Fasted Exercise and AOD-9604 Combined effect
The combination of AOD-9604's lipolytic action with fasted exercise creates a physiological environment that strongly favors fat oxidation. During fasting, insulin levels are low, removing the primary hormonal brake on lipolysis. AOD-9604 administration in this low-insulin state enhances fatty acid release from adipocytes through its beta-3 adrenergic mechanism. Exercise then provides the metabolic demand to oxidize these mobilized fatty acids, converting stored fat into energy and carbon dioxide.
The optimal protocol for most patients involves administering AOD-9604 subcutaneously first thing in the morning, waiting 15-20 minutes for absorption and initial pharmacological effect, and then performing 30-45 minutes of moderate-intensity cardiovascular exercise before eating breakfast. Walking, cycling, or light jogging at 60-70% of maximum heart rate (the "fat-burning zone") maximizes the proportion of energy derived from fat oxidation during exercise. Higher intensities can be used by conditioned athletes, but they shift the fuel mix toward carbohydrate oxidation, potentially reducing the combined effect with AOD-9604's lipolytic effects.
Patients who cannot exercise in the morning fasted state can still benefit from AOD-9604 by timing their injection 2-3 hours after their last meal, when insulin levels have returned to near-baseline. Exercise performed at this time point, while not strictly fasted, still occurs in a relatively insulin-suppressed state that permits lipolytic activity. The key principle is avoiding injection and exercise immediately after a carbohydrate-rich meal, when elevated insulin levels would blunt AOD-9604's fat-mobilizing effects.
Resistance Training for Body Composition
While cardiovascular exercise complements AOD-9604's fat-loss mechanism, resistance training addresses the equally important goal of lean mass preservation and enhancement. During any fat-loss protocol, resistance training signals to the body that muscle tissue is needed and should be preserved even during caloric deficit. Without this signal, the body may catabolize muscle tissue alongside fat, producing a "skinny fat" outcome where weight is lost but body composition does not meaningfully improve.
A minimum of two to three resistance training sessions per week, targeting all major muscle groups (chest, back, shoulders, legs, core), provides adequate stimulus for lean mass preservation. Training should emphasize compound movements (squats, deadlifts, bench press, rows, overhead press) performed at moderate to heavy loads (65-85% of one-repetition maximum) for 3-4 sets of 6-12 repetitions. This approach stimulates muscle protein synthesis, increases resting metabolic rate, and creates a favorable hormonal environment that complements AOD-9604's fat-specific action.
Protein timing around resistance training sessions supports muscle protein synthesis. Consuming 25-40 grams of high-quality protein (whey, casein, eggs, or lean meat) within 1-2 hours of training provides the amino acid substrate needed for muscle repair and growth. On training days, total protein intake should ideally reach the higher end of the recommended range (1.4-1.6 g/kg of ideal body weight) to support both recovery and lean mass preservation during caloric deficit.
Sleep Optimization for Fat Loss
Sleep quality and duration have profound effects on body composition outcomes that can either amplify or undermine AOD-9604's effects. Insufficient sleep (less than 7 hours per night) increases cortisol levels, reduces growth hormone secretion, impairs insulin sensitivity, and shifts energy partitioning toward fat storage. Studies on caloric restriction during sleep deprivation show that a larger proportion of weight lost comes from lean mass rather than fat mass compared to the same caloric restriction with adequate sleep. This finding suggests that sleep optimization may be as important as dietary and exercise interventions for achieving the body composition goals that AOD-9604 users typically pursue.
Practical sleep optimization strategies include maintaining consistent sleep and wake times (including weekends), creating a cool and dark sleep environment (65-68 degrees Fahrenheit, blackout curtains), limiting blue light exposure for 1-2 hours before bed, avoiding caffeine after early afternoon, and managing stress through meditation, reading, or other calming activities. For patients with diagnosed sleep disorders, treatment with CPAP (for OSA), cognitive behavioral therapy for insomnia (CBT-I), or appropriate sleep medications should be pursued alongside peptide therapy. Peptides like DSIP and supplemental compounds like magnesium glycinate (400-600 mg before bed) can support sleep quality as part of a comprehensive approach.
Stress Management and Cortisol Control
Chronic psychological stress elevates cortisol, a catabolic hormone that promotes visceral fat deposition, impairs insulin sensitivity, and accelerates lean mass loss. These effects directly counteract AOD-9604's fat-mobilizing mechanism, particularly regarding visceral fat, which is the most metabolically harmful fat depot and the most cortisol-responsive. Patients who are under significant chronic stress may find that AOD-9604's effectiveness is blunted unless stress management is addressed concurrently.
Evidence-based stress management techniques include regular meditation or mindfulness practice (even 10 minutes daily shows measurable cortisol reduction in controlled studies), time in nature, social connection, creative activities, and structured relaxation techniques such as progressive muscle relaxation or deep breathing exercises. Adaptogenic herbs and supplements, including ashwagandha, rhodiola, and phosphatidylserine, have modest evidence for cortisol modulation and can be used as adjuncts. For patients with anxiety or depression contributing to chronic stress, professional psychological support through therapy or appropriate medication management is essential and should not be replaced by supplements or lifestyle interventions alone.
The integration of exercise, nutrition, sleep, and stress management with AOD-9604 therapy transforms the treatment from a standalone pharmaceutical intervention into a comprehensive metabolic optimization strategy. Patients who address all four lifestyle pillars alongside their peptide protocol consistently achieve better results, faster progress, and more durable outcomes than those who rely on the peptide alone. The FormBlends consultation process includes assessment of these lifestyle factors and provides personalized recommendations for integrating them with the prescribed peptide protocol.
Comprehensive Lab Monitoring and Safety Assessment for AOD-9604 Users
One of AOD-9604's defining advantages over full-length growth hormone therapy is its favorable safety profile, particularly the absence of IGF-1 elevation, insulin resistance, and the other metabolic disturbances associated with GH administration. However, responsible use of any peptide therapy requires systematic monitoring to confirm safety, track efficacy, and detect any unexpected changes that might warrant clinical attention.
Essential Baseline Panel
Before initiating AOD-9604 therapy, a comprehensive baseline panel serves as the reference point for all subsequent monitoring. The recommended baseline workup includes a complete metabolic panel (sodium, potassium, chloride, bicarbonate, BUN, creatinine, glucose, calcium), fasting insulin with calculated HOMA-IR, HbA1c, fasting lipid panel (total cholesterol, LDL, HDL, triglycerides), liver function tests (ALT, AST, GGT, alkaline phosphatase), thyroid panel (TSH, free T4), complete blood count with differential, and high-sensitivity CRP as an inflammatory marker.
Body composition assessment at baseline is particularly valuable for AOD-9604 users because the peptide's primary benefit, selective fat loss, is best measured through composition metrics rather than simple weight. A DEXA scan provides the most accurate baseline for fat mass, lean mass, and bone density. If DEXA is unavailable, bioelectrical impedance analysis (BIA) performed consistently on the same device under standardized conditions (morning, fasted, hydrated) provides useful trending data. Waist circumference, hip circumference, and the waist-to-hip ratio should also be recorded, as these simple anthropometric measures correlate well with visceral adiposity and cardiovascular risk.
IGF-1 Confirmation Testing
One of the most important early monitoring steps for AOD-9604 users is confirming that IGF-1 levels remain unchanged from baseline. This verification distinguishes genuine AOD-9604 from full-length HGH or other GH-related compounds that might be mislabeled or contaminated. If IGF-1 rises significantly (more than 20% above baseline) during AOD-9604 use, this suggests that the product may contain full-length GH or a GH-elevating contaminant rather than pure AOD-9604. In this scenario, the product should be discontinued and the supplier should be notified. This verification step is particularly important when using products from new or unverified suppliers.
For patients combining AOD-9604 with GH secretagogues like CJC-1295/Ipamorelin, IGF-1 will obviously be elevated due to the secretagogue component, making this verification test less informative. In stacked protocols, the authenticity of the AOD-9604 component is best verified through the absence of GH-related side effects (water retention, joint stiffness, insulin resistance) beyond what the secretagogue alone would produce.
Metabolic Safety Monitoring During Treatment
At 4-week and 8-week marks during the first cycle, focused metabolic monitoring helps identify any unexpected effects. The primary parameters to track are fasting glucose and insulin (confirming no development of insulin resistance), liver function tests (ensuring no hepatotoxicity), and lipid panel (tracking the expected improvements). These tests should be drawn fasting, in the morning, and ideally at consistent times to minimize variability between measurements.
If all parameters remain stable through the first 8-week assessment, subsequent monitoring can be reduced to every 12 weeks during continued use. Annual comprehensive panels (including the full baseline workup) provide long-term safety data and catch any gradual trends that might be missed with more focused interim testing. For patients using AOD-9604 in combination with other compounds, the monitoring frequency should be adjusted based on the requirements of the most monitoring-intensive component of the protocol.
Body Composition Tracking
The most meaningful efficacy assessment for AOD-9604 involves serial body composition measurements rather than simple weight tracking. Because AOD-9604 targets fat loss selectively while preserving lean mass, patients may experience relatively modest weight changes while undergoing significant body composition improvement. A patient who loses 5 pounds of fat while maintaining or gaining lean mass will show minimal scale change but meaningful improvement in body fat percentage, waist circumference, and physical appearance.
DEXA scans at baseline, 12 weeks, and 24 weeks provide the most accurate composition data. For patients who cannot access DEXA, the combination of BIA measurements and waist circumference tracking provides a reasonable alternative. Photographs taken under consistent lighting and positioning at 4-week intervals can also document visible changes that numeric measurements alone might not capture. These multiple assessment modalities, used together, provide a comprehensive picture of AOD-9604's effects on body composition that is far more informative than weighing alone.
Injection Site Monitoring
While AOD-9604 injection site reactions are generally mild and uncommon, regular self-inspection of injection sites helps detect any developing issues. Patients should check for persistent redness, swelling, or tenderness at injection sites that lasts more than 24-48 hours. Hard nodules or areas of tissue change (either thinning or thickening) at frequently used injection sites may indicate lipodystrophy or local tissue reaction and warrant site rotation adjustments or clinical evaluation.
For patients administering daily injections, a systematic site rotation log can help ensure adequate spacing between injections at each location. A minimum of 1 inch between injection sites on the same anatomical area, and at least 24-48 hours before returning to the same specific site, prevents local tissue overexposure. Digital health apps designed for injection tracking can simplify this process, though a simple paper log or body diagram works equally well for most patients.
Hormonal Monitoring in Context
Unlike GH secretagogues that affect multiple hormonal axes, AOD-9604's selective mechanism should not produce changes in cortisol, prolactin, thyroid function, or sex hormones. Monitoring these parameters at baseline and periodically during treatment helps confirm this selectivity and provides early detection of any unexpected hormonal effects. If hormonal changes are detected during AOD-9604 monotherapy, the cause is more likely attributable to other factors (lifestyle changes, concurrent supplements, aging, stress) than to AOD-9604 itself, but clinical evaluation is still warranted.
For female patients, menstrual cycle regularity should be noted, as significant fat loss from any cause can affect reproductive hormones. Patients who experience menstrual changes during AOD-9604 therapy should have reproductive hormone levels assessed (FSH, LH, estradiol, progesterone) to determine whether the changes are related to fat loss, stress, or other factors. Similarly, male patients who note changes in libido, energy, or mood should have testosterone levels checked, as these changes are more likely related to concurrent factors than to AOD-9604 but deserve clinical attention regardless of the suspected cause.
The overall monitoring burden for AOD-9604 monotherapy is lighter than for most other peptide protocols, reflecting its favorable safety profile and selective mechanism of action. However, monitoring should never be skipped entirely, as even the safest compounds benefit from objective verification of safety and efficacy. Patients who work with providers experienced in peptide therapy typically receive monitoring protocols integrated into their treatment plan, ensuring that lab work is timed appropriately and results are interpreted in the context of the full treatment picture. The FormBlends clinical assessment includes individualized monitoring recommendations based on each patient's health history, concurrent medications, and treatment goals.
Long-Term Safety Considerations
AOD-9604's favorable safety record in clinical trials, involving over 890 participants with no significant adverse effects beyond placebo, provides reassurance for short-term use. However, the longest controlled human exposure in published trials was 24 weeks, leaving questions about multi-year continuous or repeated cycled use unanswered. Theoretical concerns for long-term use include the possibility of persistent changes in adipocyte receptor sensitivity, effects on adipokine secretion patterns as fat mass decreases, and potential interactions with age-related changes in metabolic regulation. None of these theoretical concerns have been substantiated by available data, but they warrant ongoing monitoring in patients who use AOD-9604 repeatedly over years.
The Australian Therapeutic Goods Administration (TGA) granted AOD-9604 Generally Recognized as Safe (GRAS) status for certain applications, which reflects a regulatory assessment that the compound's safety profile supports broader use. This designation, while not equivalent to full drug approval, provides a level of regulatory validation that few research peptides have achieved. Patients and providers can take some reassurance from this designation while maintaining appropriate monitoring vigilance, particularly during extended or repeated use. For any emerging safety signals or updated regulatory developments related to AOD-9604, the FormBlends team tracks and communicates relevant updates through the peptide research hub, ensuring that patients and providers have access to the most current information for making informed treatment decisions.
Frequently Asked Questions
What is AOD-9604 and how does it differ from regular growth hormone?
AOD-9604 is a synthetic peptide made up of 16 amino acids that correspond to the C-terminal fragment (residues 176-191) of human growth hormone, with an added tyrosine at the N-terminus for stability. Unlike full-length growth hormone, which activates the GH receptor and triggers IGF-1 production, tissue growth, and metabolic changes throughout the body, AOD-9604 works selectively through beta-3 adrenergic receptors on fat cells. This means it can stimulate fat breakdown (lipolysis) without causing the insulin resistance, fluid retention, joint pain, or IGF-1 elevation associated with GH therapy. Clinical trial data from 893 subjects confirmed this separation - AOD-9604 had no measurable effect on IGF-1, glucose tolerance, or insulin sensitivity.
How does AOD-9604 cause fat loss?
AOD-9604 promotes fat loss through two simultaneous mechanisms. First, it activates beta-3 adrenergic receptors on adipocytes (fat cells), triggering a signaling cascade that increases intracellular cAMP, activates protein kinase A, and phosphorylates hormone-sensitive lipase. The activated lipase breaks down stored triglycerides into free fatty acids and glycerol, which are released into the bloodstream for energy production. Second, AOD-9604 inhibits de novo lipogenesis - the process by which the body converts excess carbohydrates into new fat. By simultaneously accelerating fat breakdown and slowing fat creation, the peptide shifts the balance toward net fat reduction. This was confirmed in both isolated fat cell studies and in obese mouse models, where beta-3 receptor knockout mice showed no response to the peptide.
Does AOD-9604 work for weight loss?
The clinical evidence is mixed. In a 12-week Phase 2a trial with approximately 300 obese subjects, the 1 mg daily oral dose produced an average weight loss of 2.6 kg compared to 0.8 kg with placebo - roughly a threefold improvement. However, the larger Phase 2b OPTIONS trial with 536 subjects over 24 weeks failed to show statistically significant weight loss compared to placebo. The failure was attributed partly to an intensive diet and exercise program applied to all participants, which boosted placebo group weight loss and narrowed the treatment gap. Compared to modern GLP-1 drugs that produce 15-22% body weight reduction, AOD-9604's modest 2-3% effect is considerably smaller. The peptide may work best as one component of a broader protocol rather than as a standalone weight loss treatment.
What are the clinical trial results for AOD-9604?
AOD-9604 went through six randomized, double-blind, placebo-controlled clinical trials between 2001 and 2007, involving a total of 893 subjects. Phase 1 studies confirmed safety and established pharmacokinetics. The Phase 2a study showed meaningful weight loss at 12 weeks (2.6-3.2 kg with treatment vs. 0.8 kg with placebo). But the definitive Phase 2b OPTIONS trial enrolled 536 subjects, ran for 24 weeks with oral doses, and failed to meet its primary efficacy endpoint. Despite the efficacy failure, the safety data across all six trials was consistently excellent - adverse event rates were indistinguishable from placebo, with no IGF-1 elevation, no glucose metabolism disruption, and no serious adverse events related to the drug. Development was terminated by Metabolic Pharmaceuticals in 2007 due to the efficacy failure.
Is AOD-9604 safe?
Based on clinical trial data from 893 subjects across six controlled studies, AOD-9604 demonstrated an excellent safety profile. Adverse events were mild (headache, fatigue, dizziness) and occurred at the same rate as in placebo groups. No serious adverse events were related to AOD-9604. No subjects developed anti-drug antibodies. IGF-1 levels remained unchanged, glucose tolerance was unaffected, and cardiovascular parameters stayed normal. The peptide specifically lacks the side effects of full-length growth hormone, including insulin resistance, fluid retention, and joint pain. The main limitation is that the longest study was 24 weeks, so data on effects beyond six months is not available. The FDA has raised theoretical concerns about immunogenicity risk with long-term use, though no such issues were observed in the trials.
Can AOD-9604 help with joint health or osteoarthritis?
Preclinical research suggests potential benefits for cartilage repair and joint health, though no human clinical trials have been conducted for this application. A 2015 study in a rabbit osteoarthritis model showed that intra-articular AOD-9604 injections enhanced cartilage regeneration, and the combination of AOD-9604 with hyaluronic acid was more effective than either alone. Laboratory studies have also shown that AOD-9604 promotes proteoglycan and collagen production in chondrocytes (cartilage cells), stimulates adipose-derived stem cell differentiation toward bone-forming cells, and modulates inflammatory cytokines. While these results are encouraging, they haven't been validated in controlled human studies. Some practitioners use AOD-9604 as part of regenerative medicine protocols, often combined with BPC-157 or TB-500.
Is AOD-9604 legal?
The legality of AOD-9604 depends on jurisdiction and intended use. In Australia, it's approved by the TGA as a complementary medicine ingredient. In the United States, it has never been approved as a drug, and the PCAC voted against its inclusion on the FDA's 503A bulks list for compounding pharmacies, though a final rule isn't expected until March 2027. Research peptide suppliers sell it labeled "for research purposes only." For competitive athletes, WADA lists AOD-9604 as a prohibited substance under Section S.0, covering non-approved pharmacological substances. The regulatory situation is fluid and varies by country, so anyone considering AOD-9604 should verify its current legal status in their jurisdiction at the time they plan to access it.
How does AOD-9604 compare to semaglutide and tirzepatide for weight loss?
The comparison heavily favors the GLP-1 receptor agonists. Semaglutide 2.4 mg (Wegovy) produced approximately 14.9% body weight reduction in the STEP 1 trial. Tirzepatide (Zepbound) achieved up to 22.5% reduction in the SURMOUNT-1 trial. AOD-9604's best result was about 2.6-3.2 kg of weight loss over 12 weeks in its Phase 2a trial, which translates to roughly 2-3% body weight reduction. The mechanisms are entirely different: GLP-1 drugs primarily suppress appetite through brain signaling, while AOD-9604 directly stimulates fat breakdown in adipose tissue without affecting appetite. AOD-9604's advantages are its lack of gastrointestinal side effects (nausea, vomiting) that commonly occur with GLP-1 drugs, and its clean metabolic safety profile. But for raw weight loss efficacy, modern incretin-based therapies are far more effective.
References
- Heffernan MA, Jiang WJ, Thorburn AW, Ng FM. Effects of oral administration of a synthetic fragment of human growth hormone on lipid metabolism. American Journal of Physiology-Endocrinology and Metabolism. 2000;279(3):E501-E507. DOI: 10.1152/ajpendo.2000.279.3.E501. PMID: 10950816.
- Heffernan MA, Thorburn AW, Fam B, et al. The effects of human GH and its lipolytic fragment (AOD9604) on lipid metabolism following chronic treatment in obese mice and beta(3)-AR knock-out mice. Endocrinology. 2001;142(12):5182-5189. DOI: 10.1210/endo.142.12.8522. PMID: 11713213.
- Ng FM, Sun J, Sharma L, et al. Metabolic studies of a synthetic lipolytic domain (AOD9604) of human growth hormone. Hormone Research. 2000;53(6):274-278. DOI: 10.1159/000053782. PMID: 11146367.
- Stier H, Vos E, Kenley D. Safety and tolerability of the hexadecapeptide AOD9604 in humans. Journal of Endocrinology and Metabolism. 2013;3(1-2):7-15. DOI: 10.4021/jem.v3i1-2.157.
- More MI, Kenley D, Gusev PA. Safety and metabolism of AOD9604, a novel nutraceutical ingredient for improved metabolic health. Journal of Endocrinology and Metabolism. 2014;4(3):68-78. DOI: 10.14740/jem.v4i3.213.
- Kwon DR, Park GY, Lee SU. Effect of intra-articular injection of AOD9604 with or without hyaluronic acid in rabbit osteoarthritis model. Annals of Clinical and Laboratory Science. 2015;45(4):426-432. PMID: 26275694.
- Ng FM, Bornstein J. Hyperglycemic action of synthetic C-terminal fragments of human growth hormone. American Journal of Physiology. 1978;234(5):E521-E526. PMID: 645908.
- Rowlinson SW, Behncken SN, Rowland JE, et al. Activation of chimeric and full-length growth hormone receptors by growth hormone receptor monoclonal antibodies: a specific conformational change may be required for full-length receptor signaling. Journal of Biological Chemistry. 1998;273(9):5307-5314. DOI: 10.1074/jbc.273.9.5307.
- Wilding JP. Pharmacotherapy of obesity. Best Practice and Research: Clinical Endocrinology and Metabolism. 2007;21(2):335-344. DOI: 10.1016/j.beem.2007.04.006. PMID: 17574012.
- Jepsen CH, Obinah MPB, Avlund TH, et al. Obesity pharmacotherapy: current perspectives and future directions. Current Obesity Reports. 2013;2(2):171-179. DOI: 10.1007/s13679-013-0052-1.
- Astrup A, Madsbad S, Breum L, et al. Effect of tesofensine on bodyweight loss, body composition, and quality of life in obese patients: a randomised, double-blind, placebo-controlled trial. The Lancet. 2008;372(9653):1906-1913. DOI: 10.1016/S0140-6736(08)61525-1.
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. New England Journal of Medicine. 2022;387(3):205-216. DOI: 10.1056/NEJMoa2206038.
- Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. New England Journal of Medicine. 2021;384(11):989-1002. DOI: 10.1056/NEJMoa2032183.
- Jastreboff AM, Kaplan LM, Frias JP, et al. Triple-hormone-receptor agonist retatrutide for obesity: a phase 2 trial. New England Journal of Medicine. 2023;389(6):514-526. DOI: 10.1056/NEJMoa2301972.
- World Anti-Doping Agency. WADA statement on substance AOD-9604. WADA Official Communications. 2013. Available at: https://www.wada-ama.org/en/news/wada-statement-substance-aod-9604.
- U.S. Food and Drug Administration. FDA Briefing Document: Pharmacy Compounding Advisory Committee (PCAC) Meeting - AOD-9604 Evaluation. FDA Regulatory Documents. 2024. Available at: https://www.fda.gov/media/183583/download.
- Arch JR. Beta(3)-Adrenoceptor agonists: potential, pitfalls and progress. European Journal of Pharmacology. 2002;440(2-3):99-107. DOI: 10.1016/S0014-2999(02)01421-8. PMID: 12007528.
- Himms-Hagen J, Cui J, Danforth E Jr, et al. Effect of CL-316,243, a thermogenic beta 3-agonist, on energy balance and brown and white adipose tissues in rats. American Journal of Physiology. 1994;266(4 Pt 2):R1371-R1382. PMID: 7910436.
- Collins S, Cao W, Robidoux J. Learning new tricks from old dogs: beta-adrenergic receptors teach new lessons on firing up adipose tissue metabolism. Molecular Endocrinology. 2004;18(9):2123-2131. DOI: 10.1210/me.2004-0193.
- Alliance for Pharmacy Compounding. APC statement on peptides and compounding. APC Position Papers. March 2024. Available at: https://join.a4pc.org/hubfs/PDFs/APC-Peptides-Statement-March-1-2024.pdf.
- Greenway FL, Fujioka K, Plodkowski RA, et al. Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR-I): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. The Lancet. 2010;376(9741):595-605. DOI: 10.1016/S0140-6736(10)60888-4.
- Cypess AM, Lehman S, Williams G, et al. Identification and importance of brown adipose tissue in adult humans. New England Journal of Medicine. 2009;360(15):1509-1517. DOI: 10.1056/NEJMoa0810780.
- Granneman JG, Li P, Zhu Z, Lu Y. Metabolic and cellular plasticity in white adipose tissue I: effects of beta3-adrenergic receptor activation. American Journal of Physiology-Endocrinology and Metabolism. 2005;289(4):E608-E616. DOI: 10.1152/ajpendo.00009.2005.
- Metabolic Pharmaceuticals Limited. Obesity trial update: first 100 subjects complete the Phase 2B trial of AOD9604. Company Press Release via BioSpace. 2006. Available at: https://www.biospace.com.
- News-Medical.net. Obesity drug codenamed AOD9604 highly successful in trials. December 16, 2004. Available at: https://www.news-medical.net/news/2004/12/16/6878.aspx.