Executive Summary
The peptide medicine pipeline is experiencing its most productive period in history. Between 2025 and 2030, over 180 peptide-based therapeutics are in active clinical development, spanning metabolic disease, oncology, infectious disease, and neurodegeneration. Oral delivery breakthroughs, multi-receptor agonists, and AI-driven design are rewriting the rules for what peptide drugs can achieve.
Key Takeaways
- 185+ peptide therapeutics in active clinical development across Phases 1-3 as of early 2026
- Oral GLP-1 agents like orforglipron are on track for FDA approval in 2026, potentially transforming patient access and adherence
- Triple agonists such as retatrutide have demonstrated up to 28.7% body weight reduction in Phase 3 trials - exceeding all approved therapies
- Myostatin inhibitors are entering combination trials with GLP-1 drugs to preserve muscle mass during weight loss
- AI-designed peptides have produced validated antimicrobial candidates from libraries of 50,000+ computationally generated sequences
If you've been following the GLP-1 revolution, you already know how a single peptide class can reshape entire therapeutic categories. Semaglutide and tirzepatide turned obesity pharmacotherapy on its head. But what's coming next makes those advances look like the opening act.
This report maps the full scope of the peptide drug pipeline from 2025 through 2030. We'll cover oral non-peptide GLP-1 agents like orforglipron, triple and quad receptor agonists including retatrutide, myostatin and activin inhibitors designed to preserve lean mass, mRNA-encoded peptide therapies, targeted delivery systems, AI-designed peptides, long-acting depot formulations, cell-penetrating peptides, peptide vaccines for cancer immunotherapy, antimicrobial peptides for drug-resistant infections, peptide-drug conjugates for oncology, diagnostics applications, regulatory evolution, manufacturing innovation, and personalized peptide medicine.
The global peptide therapeutics market was valued at approximately $140.9 billion in 2025, and analysts project it will reach $260 billion by 2030 - a compound annual growth rate (CAGR) near 10.8% (Grand View Research, 2025). Several converging forces are fueling this expansion: the commercial success of GLP-1 receptor agonists, advances in solid-phase peptide synthesis and continuous flow manufacturing, AI-accelerated discovery platforms that compress timelines from years to months, and a growing clinical evidence base for peptide-based approaches across therapeutic areas.
Key Takeaways
- 185+ peptide therapeutics in active clinical development across Phases 1-3 as of early 2026
- Oral GLP-1 agents like orforglipron are on track for FDA approval in 2026, potentially transforming patient access and adherence
- Triple agonists such as retatrutide have demonstrated up to 28.7% body weight reduction in Phase 3 trials - exceeding all approved therapies
- Myostatin inhibitors are entering combination trials with GLP-1 drugs to preserve muscle mass during weight loss
- AI-designed peptides have produced validated antimicrobial candidates from libraries of 50,000+ computationally generated sequences
- Peptide-drug conjugates (PDCs) have 96 candidates in development, with six in Phase 3 clinical trials
- Manufacturing investment exceeds $6 billion from Eli Lilly alone for new peptide synthesis facilities
What follows is the most thorough analysis of the peptide drug pipeline available. Whether you're a researcher, clinician, investor, or patient advocate, this report gives you the forward-looking data you need to understand and prepare for the changes ahead.
Current State of Peptide Medicine
Peptide therapeutics have traveled a remarkable path from niche injectables to blockbuster drugs. To understand what's coming, it helps to see where we are right now and how we got here.
The GLP-1 Catalyst
The modern peptide medicine era began in earnest with the approval of exenatide in 2005, but it was semaglutide that proved peptide drugs could become the most commercially successful pharmaceutical class on the planet. Novo Nordisk's Ozempic and Wegovy generated combined revenues exceeding $25 billion in 2024, while Eli Lilly's Mounjaro (tirzepatide) wasn't far behind. For a deeper look at how we got here, see our GLP-1 peptide timeline and history.
These drugs didn't just treat diabetes and obesity. Clinical trials revealed cardiovascular risk reduction, kidney protection, potential benefits in MASH (metabolic dysfunction-associated steatotic liver disease), and even signals for neurodegenerative disease improvement. Each new indication expanded the addressable patient population and validated peptide-based approaches for conditions previously dominated by small molecules.
The Approved Peptide Landscape
As of early 2026, more than 80 peptide drugs hold active FDA approval. They span an impressive range of therapeutic areas:
| Therapeutic Area | Approved Peptide Drugs (Examples) | Market Share (Est.) |
|---|---|---|
| Metabolic/Diabetes/Obesity | Semaglutide, Tirzepatide, Liraglutide, Exenatide | ~52% |
| Oncology | Lutathera (lutetium-177 dotatate), Leuprolide, Degarelix | ~18% |
| Cardiovascular | Bivalirudin, Eptifibatide, Nesiritide | ~8% |
| Infectious Disease | Enfuvirtide, Daptomycin, Telavancin | ~6% |
| Rare/Orphan Disease | Setmelanotide, Teduglutide, Vosoritide | ~5% |
| Endocrine | Octreotide, Lanreotide, Teriparatide | ~5% |
| Cosmetic/Dermatology | Bremelanotide, Afamelanotide | ~2% |
| Other | Ziconotide, Calcitonin, Vasopressin analogs | ~4% |
Why Peptides Are Winning
Peptides occupy a sweet spot between small molecules and large biologics. They offer target specificity similar to antibodies but with better tissue penetration and lower immunogenicity. They're cheaper and faster to manufacture than monoclonal antibodies. And unlike many small molecules, they can engage protein-protein interaction surfaces that were previously considered "undruggable."
Several structural advantages make peptides increasingly attractive to drug developers:
- High potency - Many peptides achieve therapeutic effects at microgram to low milligram doses
- Selectivity - Peptide structure allows precise receptor targeting with minimal off-target effects
- Favorable safety profiles - Peptides typically degrade into natural amino acids, reducing toxicity concerns
- Modular design - Amino acid sequences can be systematically modified to optimize pharmacokinetics
- Scalable synthesis - Modern solid-phase and continuous-flow methods enable commercial-scale production
The remaining challenges - oral bioavailability, serum stability, and manufacturing cost at scale - are precisely the problems that the next generation of peptide technologies is solving. For a broader market perspective, our peptide industry market analysis digs into the commercial and investment landscape.
A Brief History of Peptide Drug Development
Peptide pharmacology has evolved through several distinct eras. Understanding this trajectory helps contextualize the current acceleration.
1920s-1950s: The Extraction Era. The earliest peptide drugs were isolated from animal tissues. Insulin (1922) was extracted from porcine and bovine pancreas. Oxytocin and vasopressin were purified from posterior pituitary glands. ACTH (adrenocorticotropic hormone) was used to stimulate cortisol production. These natural peptide hormones demonstrated that peptides could serve as powerful medicines, but their production was limited by the availability of animal source material and batch-to-batch variability.
1960s-1980s: The Synthesis Era. Robert Bruce Merrifield's invention of solid-phase peptide synthesis (SPPS) in 1963 - for which he won the Nobel Prize in Chemistry in 1984 - transformed peptide drug development. For the first time, peptides could be synthesized chemically with defined sequences and high purity, independent of animal sources. This enabled the development of synthetic analogs of natural hormones with improved properties: longer half-lives, greater potency, and resistance to enzymatic degradation. Examples include desmopressin (a vasopressin analog), goserelin (a GnRH agonist), and octreotide (a somatostatin analog).
1990s-2010s: The Engineering Era. Advances in peptide chemistry, recombinant DNA technology, and delivery systems enabled increasingly sophisticated peptide drug design. PEGylation, lipidation, and Fc-fusion extended peptide half-lives from minutes to days or weeks. Depot formulations (PLGA microspheres, implants) further extended dosing intervals. The approval of exenatide (2005) and liraglutide (2010) demonstrated that engineered peptide drugs could compete effectively in large therapeutic markets.
2020s: The Blockbuster Era. Semaglutide's approval for obesity (2021) and tirzepatide's approval (2022) proved that peptide drugs could achieve commercial scale previously reserved for small molecules and antibodies. Combined annual revenues for GLP-1 agonists are projected to exceed $100 billion by 2030. This commercial validation has attracted unprecedented investment in peptide drug development, manufacturing, and delivery technology.
The Regulatory Evolution of Peptide Drugs
The regulatory treatment of peptides has evolved alongside the science. The FDA initially regulated most peptide drugs as biologics under the Public Health Service Act, requiring Biologics License Applications (BLAs). In 2020, the BPCIA (Biological Product Classification Implementation Act) transferred many shorter synthetic peptides (under 40 amino acids) to the drug pathway under the Federal Food, Drug, and Cosmetic Act, enabling generic (ANDA) approval routes. This regulatory clarification has important implications for generic competition and patient access as key peptide drug patents expire.
The transition of peptides from biologic to drug classification was not without controversy. Some patient advocacy groups argued that the shift could lead to lower quality standards for generic peptide drugs. Industry groups were divided - innovator companies generally opposed easier generic pathways, while generic manufacturers supported them. The FDA ultimately maintained that its quality standards would be equivalent regardless of regulatory pathway, but required that generic peptide manufacturers demonstrate pharmaceutical equivalence through rigorous analytical characterization, including detailed comparison of amino acid sequence, impurity profiles, stereochemistry, and higher-order structure.
This regulatory evolution continues to have practical consequences. For example, the determination of whether a specific peptide drug follows the ANDA or BLA pathway depends on factors including molecular weight, complexity of post-translational modifications, and whether the peptide is produced by chemical synthesis or recombinant technology. These classification decisions can affect the timeline and cost of generic entry by years and tens of millions of dollars, making them commercially significant for both innovator and generic pharmaceutical companies.
Oral Peptide Delivery Breakthroughs
The single biggest barrier to peptide medicine adoption has always been the needle. Oral delivery of peptides - once considered pharmacologically impossible - is now producing Phase 3 clinical data that rivals injectable formulations.
Orforglipron: The Oral GLP-1 Frontrunner
Eli Lilly's orforglipron is the furthest along and the most likely to reach patients first. Unlike oral semaglutide (Rybelsus), which is an actual peptide formulated with an absorption enhancer, orforglipron is a non-peptide small molecule that activates the GLP-1 receptor. That distinction matters enormously for manufacturing, stability, and cost.
Phase 3 results from the ACHIEVE program have been striking. At 72 weeks, orforglipron at the highest tested dose (36 mg) produced mean body weight reductions of 11.2%, compared to 2.1% with placebo (Wharton et al., NEJM, 2025). The 12 mg dose - likely the commercial sweet spot - achieved 8.4% weight loss with a tolerability profile consistent with injectable GLP-1 drugs.
Orforglipron Phase 3 Weight Loss Data (ACHIEVE Program, 72 Weeks)
| Dose | Mean Weight Loss (%) | vs. Placebo | GI Side Effects (%) |
|---|---|---|---|
| 6 mg daily | -7.5% | -5.4% | ~35% |
| 12 mg daily | -8.4% | -6.3% | ~38% |
| 36 mg daily | -11.2% | -9.1% | ~42% |
| Placebo | -2.1% | - | ~18% |
In the ACHIEVE-3 trial comparing orforglipron to oral semaglutide in type 2 diabetes, topline results announced in September 2025 showed competitive glycemic control. Full data published in The Lancet in February 2026 confirmed non-inferiority, with the added advantage of no fasting requirement before dosing - a significant convenience factor that oral semaglutide demands.
Lilly submitted orforglipron for regulatory review for obesity in 2025 and received selection for the FDA Commissioner's National Priority Review Voucher pilot program. A U.S. diabetes submission is planned for 2026, with potential FDA action on the obesity indication expected in Q2 2026.
The Danuglipron Story: A Cautionary Tale
Pfizer's danuglipron was once considered orforglipron's main rival in the oral GLP-1 space. However, Pfizer discontinued development after a single participant in a dose-optimization study experienced potential drug-induced liver injury (DILI). Although the event was asymptomatic and resolved after discontinuation, Pfizer made the decision to exit the program rather than risk further hepatotoxicity signals in larger trials.
This outcome underscores a reality of peptide drug development: the bar for safety in chronic-use obesity medications is extremely high, and a single serious adverse event can end a multi-billion-dollar program.
Other Oral Peptide Approaches in Development
Beyond non-peptide small molecule GLP-1 agonists, several technologies aim to deliver actual peptide molecules orally:
- Permeation enhancers (SNAC technology) - The approach used in oral semaglutide (Rybelsus). Sodium N-[8-(2-hydroxybenzoyl)amino]caprylate (SNAC) protects the peptide from gastric degradation and enhances transcellular absorption. Limitations include low bioavailability (~1%), mandatory fasting, and large tablet size.
- Ionic liquid formulations - Researchers at MIT and Harvard have developed choline and geranic acid (CAGE) ionic liquids that can deliver insulin and other peptides orally with bioavailability up to 50% in preclinical models. Phase 1 studies are underway.
- Intestinal patch systems - Rani Therapeutics' RaniPill is an ingestible capsule that deploys a dissolvable microneedle patch in the small intestine. Phase 2 data for an octreotide formulation showed bioavailability comparable to subcutaneous injection.
- Self-orienting millimeter-scale applicators (SOMA) - Developed by MIT researchers in collaboration with Novo Nordisk, these ingestible devices orient themselves in the stomach and inject peptide drugs directly into the gastric wall. Preclinical data supports delivery of semaglutide, insulin, and other peptides.
- Nanoparticle encapsulation - PLGA and chitosan nanoparticles protect peptides through the GI tract and facilitate absorption through M-cells in Peyer's patches. Multiple candidates are in preclinical development.
Why Oral Delivery Matters So Much
Patient preference data consistently shows that 70-80% of patients would choose an equally effective oral medication over an injectable. Adherence rates for oral medications typically exceed those for injectables by 15-30%. An effective oral GLP-1 could expand the treatable patient population by tens of millions - particularly patients who currently decline injectable therapy despite meeting clinical criteria.
Oral Semaglutide: Lessons from the First Mover
Oral semaglutide (Rybelsus, Novo Nordisk) provides valuable lessons for the oral peptide field. Approved in 2019 for type 2 diabetes, it uses sodium N-[8-(2-hydroxybenzoyl)amino]caprylate (SNAC) as an absorption enhancer to protect semaglutide from gastric acid degradation and facilitate transcellular absorption in the stomach.
The clinical results are meaningful but reveal the limitations of current technology. At the highest approved dose (14 mg), oral semaglutide produces HbA1c reductions comparable to injectable semaglutide 0.5 mg but less than injectable semaglutide 1.0 mg. For weight loss, a higher dose of 50 mg (not yet approved) achieved approximately 15% weight loss in clinical trials - competitive with injectable semaglutide 2.4 mg but requiring a much larger drug dose.
The practical limitations are instructive. Patients must take Rybelsus on an empty stomach with no more than 4 ounces of water, then wait at least 30 minutes before eating, drinking, or taking other medications. These restrictions reduce real-world adherence and patient satisfaction. The bioavailability is approximately 1%, meaning 99% of the ingested peptide is destroyed in the GI tract - an enormous waste of expensive drug substance.
These limitations explain why orforglipron's small molecule approach is so compelling. As a non-peptide molecule, orforglipron doesn't need protection from gastric acid, doesn't require fasting, and has conventional oral bioavailability. The trade-off is that it's a GLP-1 agonist by design rather than by nature - it mimics GLP-1's receptor interaction without being a peptide.
Emerging Oral Peptide Companies and Programs
Several biotechs are pursuing differentiated oral peptide delivery strategies:
Rani Therapeutics has developed the RaniPill, an ingestible capsule containing a dissolvable microneedle array. Once it reaches the small intestine, the capsule's pH-sensitive outer coating dissolves, triggering deployment of micro-needles that inject peptide directly into the intestinal wall. Phase 2 data for an octreotide formulation showed bioavailability comparable to subcutaneous injection, with no serious adverse events. If validated in Phase 3, this platform could be applied to virtually any injectable peptide.
Enteris BioPharma uses its Peptelligence technology - a combination of enteric coating, citric acid pH modifier, and acyl-carnitine absorption enhancer - to protect and deliver peptides through the duodenum. Their oral leuprolide formulation (Ovarest) is in Phase 3 for endometriosis, and the platform is being applied to parathyroid hormone and calcitonin analogs.
Chiasma (now Amryt) achieved FDA approval for Mycapssa, an oral octreotide capsule using their Transient Permeability Enhancer (TPE) technology. While sales have been modest due to the small neuroendocrine tumor patient population, the approval validated the concept of oral delivery for therapeutic peptides and provides a commercial proof point for the field.
Protagonist Therapeutics has developed orally stable cyclic peptides through extensive chemical stabilization. Their approach uses constrained cyclic structures with unnatural amino acids to create peptides that survive the GI tract without permeation enhancers. Rusfertide, a hepcidin mimetic peptide, is in clinical development for polycythemia vera.
Oral Peptide Delivery: The Next Five Years
By 2030, we're likely to see at least 3-4 oral GLP-1 agents on the market. Orforglipron will almost certainly be first. But the real transformation will come from technologies that can deliver larger, more complex peptides orally - opening the door to oral versions of dual and triple agonists, BPC-157, growth hormone secretagogues, and other peptides currently limited to injection.
The economic implications are substantial. Injectable peptide drugs require cold chain storage, sterile manufacturing, pen or vial packaging, and often involve healthcare provider administration or patient injection training. Oral formulations eliminate most of these costs, potentially reducing the total cost of peptide therapy by 40-60%. This cost reduction, combined with improved patient convenience, could dramatically expand the addressable market for peptide therapeutics.
The competitive dynamics are also shifting. Major pharmaceutical companies are investing heavily in oral peptide platforms, either through internal R&D or acquisition of specialized biotechs. The companies that establish leadership in oral peptide delivery will have a durable competitive advantage as the field matures, because the same platform technology can be applied across multiple peptide drug candidates.
Next-Generation Multi-Agonists: Triple, Quad, and Beyond
If semaglutide proved the power of GLP-1 agonism and tirzepatide showed that dual agonism (GLP-1/GIP) could do even better, the next logical step was triple and quadruple receptor agonism. The clinical data arriving in 2025-2026 suggests this logic was sound.
Retatrutide: The Triple Agonist Leader
Retatrutide (Eli Lilly) is a first-in-class GIP/GLP-1/glucagon triple receptor agonist and the most advanced multi-agonist in clinical development. By activating three metabolic hormone pathways simultaneously, retatrutide achieves weight loss efficacy that surpasses all currently approved therapies.
The Phase 2 data published in the New England Journal of Medicine (Jastreboff et al., 2023) showed dose-dependent weight loss reaching 24.2% at the highest dose over 48 weeks. But the Phase 3 TRIUMPH program has pushed those numbers even further.
Retatrutide Phase 3 TRIUMPH-4 Results
At the 12 mg dose, patients achieved an average body weight reduction of 28.7% at 68 weeks - equivalent to approximately 71.2 pounds (32.3 kg) of weight loss. This exceeds the efficacy of tirzepatide's best Phase 3 results (22.5% at the highest dose) and semaglutide 2.4 mg (16.9%).
The glucagon receptor component of retatrutide adds metabolic effects beyond what GLP-1/GIP agonism provides alone. Glucagon increases hepatic energy expenditure, promotes lipolysis in adipose tissue, and may directly improve hepatic steatosis. This makes retatrutide particularly promising for MASH, where Phase 3 trials are underway.
However, the TRIUMPH program also revealed a new safety signal that bears watching. Lilly acknowledged that the data showed signals beyond the typical GI side effects associated with incretin-based therapies, though specific details have not been fully disclosed as of this writing. Seven additional Phase 3 trials evaluating retatrutide in obesity, type 2 diabetes, knee osteoarthritis, obstructive sleep apnea, chronic low back pain, cardiovascular/renal outcomes, and MASH are expected to complete in 2026.
Survodutide: The GLP-1/Glucagon Dual Agonist
Boehringer Ingelheim's survodutide takes a different approach - dual agonism of GLP-1 and glucagon receptors (without GIP). In Phase 2 studies for MASH with liver fibrosis, survodutide produced histologic improvement of MASH without worsening fibrosis in 43-62% of participants. Phase 3 studies for both obesity and MASH are ongoing.
The GLP-1/glucagon combination appears particularly suited to liver disease. Glucagon receptor activation in the liver directly reduces hepatic fat content and inflammation, while GLP-1 provides systemic metabolic improvements. This liver-targeted mechanism differentiates survodutide from pure GLP-1 agonists in the MASH space.
The Multi-Agonist Landscape
| Compound | Targets | Developer | Phase | Key Indication | Max Weight Loss |
|---|---|---|---|---|---|
| Retatrutide | GIP/GLP-1/Glucagon | Eli Lilly | Phase 3 | Obesity, T2D, MASH | 28.7% |
| Survodutide | GLP-1/Glucagon | Boehringer Ingelheim | Phase 3 | MASH, Obesity | ~19% |
| Pemvidutide | GLP-1/Glucagon | Altimmune | Phase 2 | MASH, Obesity | ~15% |
| Mazdutide | GLP-1/Glucagon | Innovent/Lilly | Phase 3 (China) | Obesity, T2D | ~17% |
| CagriSema | GLP-1 + Amylin | Novo Nordisk | Phase 3 | Obesity | ~25% |
| AMG 133 | GIP antagonist/GLP-1 agonist | Amgen | Phase 2 | Obesity | ~14.5% (12 wk) |
CagriSema: The Amylin + GLP-1 Combination
Novo Nordisk's CagriSema takes a different approach to multi-target metabolic therapy. Rather than building multiple agonist activities into a single molecule, CagriSema combines two separate peptides in a fixed-ratio co-formulation: cagrilintide (a long-acting amylin analog) and semaglutide (GLP-1 receptor agonist). The combination is administered as a single weekly injection.
Amylin is a peptide hormone co-secreted with insulin from pancreatic beta cells. It slows gastric emptying, suppresses postprandial glucagon secretion, and promotes satiety through central nervous system pathways that complement but don't overlap with GLP-1 signaling. By engaging two distinct appetite-suppression pathways - GLP-1 through hypothalamic POMC/CART neurons and amylin through area postrema neurons - the combination may produce additive appetite suppression.
Phase 3 data from the REDEFINE program showed that CagriSema achieved approximately 25% body weight loss at 68 weeks - exceeding the ~17% achieved by semaglutide alone and the ~9% by cagrilintide alone. These results place CagriSema among the most effective pharmacological weight loss interventions, approaching the efficacy of bariatric surgery.
CagriSema also showed impressive metabolic improvements beyond weight. HbA1c reductions in diabetic patients were significantly greater with the combination than with either component alone, and cardiovascular risk markers (lipids, blood pressure, inflammatory markers) improved across the board. Novo Nordisk has filed for regulatory approval, with potential market launch in late 2026 or early 2027.
AMG 133 (MariTide): The Contrarian Approach
Amgen's MariTide (AMG 133) challenges a core assumption of the multi-agonist approach. While tirzepatide and retatrutide both activate the GIP receptor as one of their targets, AMG 133 does the opposite - it blocks GIP signaling while activating GLP-1. This bispecific design uses an anti-GIP antibody fused to two GLP-1 agonist peptide sequences.
The rationale is that GIP has complex, context-dependent metabolic effects. In some settings, GIP promotes fat storage in adipose tissue. By blocking GIP's fat-promoting effects while stimulating GLP-1's appetite-suppressing effects, AMG 133 may achieve a distinct metabolic profile. Phase 1 data showed approximately 14.5% weight loss at just 12 weeks - a rapid onset that exceeds other agents at the same timepoint.
Perhaps most intriguing, preliminary data suggests that weight loss with AMG 133 may be more durable after drug discontinuation compared to pure GLP-1 agonists. If confirmed in larger trials, this would address one of the biggest criticisms of incretin-based obesity therapy - that weight returns when the drug stops. The antibody-peptide fusion design gives AMG 133 a very long half-life, enabling monthly dosing from the outset. Phase 2 trials with expanded patient populations are ongoing and expected to read out in 2026.
Amycretin: Novo Nordisk's Next Move
Not content with CagriSema, Novo Nordisk is also developing amycretin - a single molecule that activates both GLP-1 and amylin receptors. Unlike CagriSema (which is two separate molecules combined), amycretin integrates both receptor activities into one peptide sequence. Phase 1 data showed approximately 13% weight loss at just 12 weeks, suggesting the potential for 25%+ weight loss at full treatment duration.
Amycretin's advantage over CagriSema would be manufacturing simplicity (one molecule instead of two) and potentially a more consistent pharmacokinetic profile. It represents Novo Nordisk's hedge against competitive pressure from triple agonists - if amycretin can match retatrutide's efficacy through a different mechanism, it diversifies Novo's metabolic pipeline.
Quad Agonists and Beyond
Multiple companies are exploring quadruple agonist combinations, though most remain in preclinical stages. Conceptual approaches include GIP/GLP-1/glucagon/amylin receptor engagement or GIP/GLP-1/glucagon/FGF21 pathway modulation. The theory is that engaging more metabolic pathways simultaneously could push weight loss efficacy toward surgical outcomes (30-40% body weight reduction) while maintaining pharmaceutical convenience.
There's a natural question of diminishing returns and additive toxicity. Each additional receptor target introduces new pharmacology - and potentially new side effects. The success of triple agonism doesn't guarantee that quad agonism will be better. Clinical data will be the arbiter, and meaningful human results from quad agonist programs likely won't arrive until 2028-2029.
One alternative to quad agonism in a single molecule is polypharmacy - combining separate peptide drugs that each target different pathways. A regimen of semaglutide (GLP-1) + bimagrumab (ActRII/myostatin) + an SGLT2 inhibitor, for example, could address obesity, muscle preservation, and kidney/cardiovascular protection simultaneously. This approach allows individual dose titration and the ability to add or remove components based on patient response, though it adds complexity and cost.
For a full analysis of how these agents compare to currently available therapies, see our complete guide to obesity pharmacotherapy.

Peptide-Drug Conjugates: Precision Oncology's Next Chapter
Peptide-drug conjugates (PDCs) are emerging as the next evolution of targeted cancer therapy. They combine the tumor-targeting precision of homing peptides with the cell-killing power of cytotoxic payloads - and they may solve several problems that limit antibody-drug conjugates (ADCs).
How PDCs Work
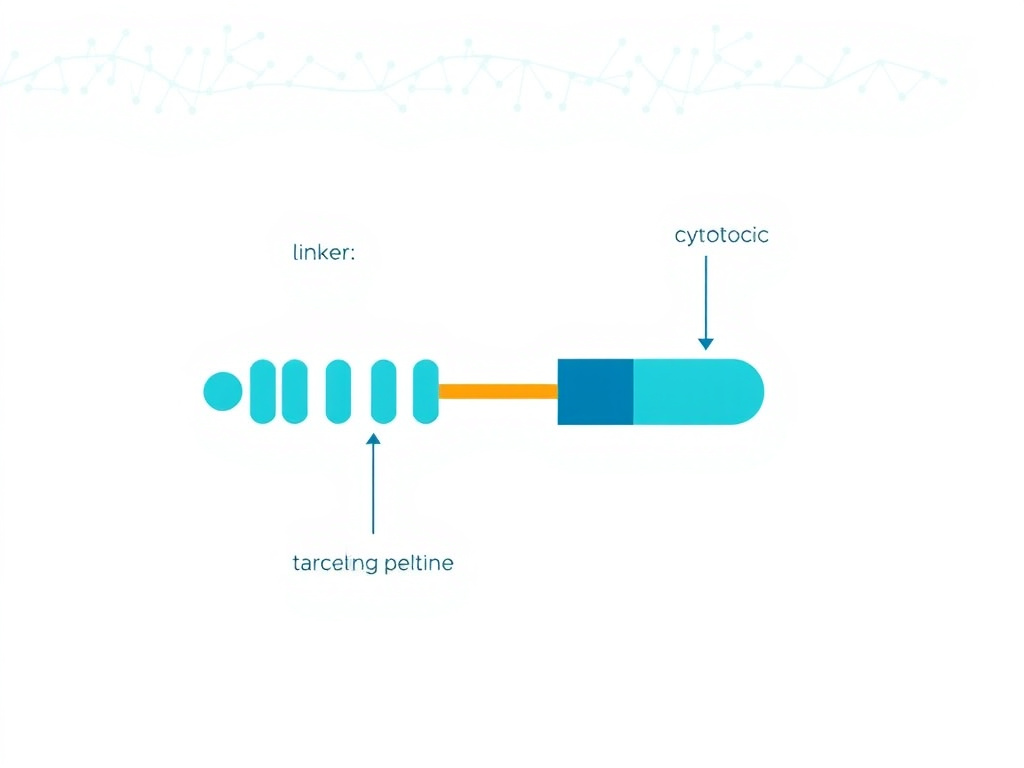
A peptide-drug conjugate consists of three components: a targeting peptide that binds receptors overexpressed on tumor cells, a cytotoxic payload (such as doxorubicin, paclitaxel, or a tubulin inhibitor), and a chemical linker connecting the two. When the PDC reaches a tumor cell, receptor binding triggers internalization, and intracellular conditions (low pH, specific enzymes) cleave the linker to release the payload inside the cancer cell.
Compared to antibody-drug conjugates - which use monoclonal antibodies as the targeting vehicle - PDCs offer several practical advantages (Armstrong et al., J Pept Sci, 2025):
- Better tissue penetration - Peptides (1-5 kDa) are 30-50x smaller than antibodies (~150 kDa), allowing deeper penetration into solid tumors
- Lower immunogenicity - Short peptide sequences rarely trigger anti-drug antibody formation
- Simpler manufacturing - Chemical peptide synthesis is faster and cheaper than antibody production in cell culture
- Rapid clearance - Faster renal elimination means shorter exposure to healthy tissues
- Versatile functionalization - Peptides can be readily modified with unnatural amino acids, cyclization, and stapling
Clinical Pipeline Status
As of 2025, approximately 96 PDCs are in development, with six in Phase 3 clinical trials (Zhou et al., J Nanobiotechnol, 2025). The only FDA-approved PDC is Lutathera (lutetium-177 dotatate), a radiopeptide conjugate for neuroendocrine tumors. Pepaxto (melphalan flufenamide), a peptide-drug conjugate for multiple myeloma, was voluntarily withdrawn from the U.S. market in 2022 after a confirmatory trial missed its primary endpoint, though it retains EMA and MHRA approval in Europe.
| PDC Candidate | Target | Cancer Type | Phase | Notes |
|---|---|---|---|---|
| Lutathera | Somatostatin receptor | Neuroendocrine tumors | Approved (FDA) | Radiopeptide conjugate |
| 177Lu-PSMA-617 | PSMA | Prostate cancer | Approved (Pluvicto) | Radiopeptide conjugate |
| Pepaxto | Aminopeptidase | Multiple myeloma | Withdrawn (US)/Approved (EU) | Peptide-alkylator conjugate |
| MP-0250 | VEGF/HGF | NSCLC | Phase 2 | AI-designed, 34% ORR |
| CBP-1008 | Folate receptor | Ovarian, breast | Phase 2 | PDC with tubulin inhibitor |
| ANG1005 | LRP-1 | Brain metastases | Phase 3 | Crosses blood-brain barrier |
AI-Driven PDC Design
One of the most striking trends is the integration of artificial intelligence into PDC development. Since 2022, 78% of PDCs entering clinical trials have utilized AI-optimized components - up from fewer than 15% before 2020 (Li et al., Front Pharmacol, 2025). AI tools like AlphaFold2 enable precise modeling of peptide-receptor docking interactions, while generative models can propose novel targeting peptide sequences with optimized binding affinity and selectivity.
MP-0250, a VEGF/HGF-targeting PDC designed with AlphaFold2-guided peptide-receptor docking, demonstrated a 34% objective response rate in Phase 2 NSCLC trials - an encouraging result for a targeted approach in a difficult-to-treat cancer type.
Challenges Ahead
PDCs face meaningful hurdles that have limited their clinical success to date. Metabolic instability in serum, premature payload release before reaching the tumor, and rapid renal clearance all reduce the effective dose reaching cancer cells. The short half-life of peptides, while advantageous for reducing systemic toxicity, can also limit tumor accumulation.
Research is addressing these challenges through cyclization to improve metabolic stability, albumin-binding modifications to extend half-life, cleavable linkers optimized for tumor-specific enzymes, and multivalent PDC designs that increase avidity for tumor surface receptors.
Radiopeptide Conjugates: The Theranostic Success Story
Radiopeptide conjugates represent the most clinically validated subclass of PDCs. Lutathera (lutetium-177 dotatate) and Pluvicto (lutetium-177 PSMA-617) have demonstrated that the PDC concept works in clinical practice, producing meaningful survival benefits in neuroendocrine tumors and metastatic castration-resistant prostate cancer, respectively.
The theranostic model used by these drugs - where the same peptide is first labeled with a diagnostic radionuclide (gallium-68 for PET imaging) to confirm target expression, then labeled with a therapeutic radionuclide (lutetium-177) for treatment - represents an elegant application of personalized medicine. Only patients whose tumors express the target receptor (confirmed by diagnostic imaging) receive the therapeutic agent, ensuring that treatment is directed at patients most likely to benefit.
The commercial success of Pluvicto (projected annual sales exceeding $3 billion) has stimulated intense interest in developing additional radiopeptide conjugates for other cancers. Active clinical programs are targeting fibroblast activation protein (FAP) for solid tumors, CXCR4 for hematologic malignancies, and various integrin subtypes for breast and glioblastoma cancers. The radiopharmaceutical production infrastructure - including cyclotrons, radiochemistry labs, and specialized transportation networks for short-lived isotopes - is expanding rapidly to support this growing pipeline.
Next-Generation PDC Design Principles
The field is moving toward second-generation PDCs that incorporate lessons from both the successes and failures of first-generation compounds. Key design principles include:
- Multivalent targeting - Using branched or dendritic peptide structures that display multiple copies of the targeting peptide, increasing binding avidity and improving tumor accumulation by 5-20x compared to monovalent PDCs
- Protease-resistant linkers - New linker chemistries that are stable in serum but cleaved specifically by tumor-associated enzymes (cathepsins, matrix metalloproteinases, prostate-specific antigen) to minimize premature payload release
- Dual-targeted PDCs - Conjugates bearing two different targeting peptides that bind different tumor-associated receptors, improving selectivity for tumor cells that co-express both targets while sparing normal cells that express only one
- Immunostimulatory payloads - Instead of cytotoxic chemotherapy, some next-generation PDCs carry immunostimulatory agents (STING agonists, TLR ligands) that convert immunologically "cold" tumors into "hot" tumors responsive to checkpoint immunotherapy
- Conditional activation - Pro-drug PDCs where the targeting peptide is masked by a cleavable shield in circulation, becoming active only in the tumor microenvironment where specific proteases remove the shield
AI-Designed Peptides: Machine Learning Meets Drug Discovery
Artificial intelligence is compressing peptide drug discovery timelines from years to months. Generative models, protein language models, and diffusion architectures are producing validated peptide candidates at unprecedented speed.
The Technology Stack
Modern AI peptide design draws on several computational approaches (Fernandez-Diaz et al., Drug Discov Today, 2025):
- Protein language models - Large transformer models trained on millions of protein sequences learn the "grammar" of functional peptides, enabling prediction of activity, stability, and toxicity from sequence alone
- Generative adversarial networks (GANs) - These models learn to generate novel peptide sequences that match the statistical properties of known active peptides
- Diffusion models - Adapted from image generation, diffusion models generate peptide sequences by iteratively refining random starting points toward functional sequences
- Graph neural networks - These capture the 3D structural relationships between amino acid residues, improving prediction of folding and binding properties
- AlphaFold2 and derivatives - Structure prediction enables rapid assessment of peptide-target binding before wet-lab validation
Validated Success Stories
The field is moving beyond proof-of-concept. Several AI-designed peptide campaigns have produced experimentally validated results:
In one landmark study, researchers used a latent diffusion model (AMP-diffusion) fine-tuned on antimicrobial peptide sequences using protein language model embeddings. From 50,000 generated candidates, 46 top peptides were synthesized and experimentally tested. The results confirmed broad-spectrum antibacterial activity against multiple drug-resistant pathogens (Wong et al., Nat Microbiol, 2025). This represents a hit rate dramatically higher than traditional screening approaches.
Another team at Gubra, a Danish biotech, has demonstrated end-to-end AI-driven peptide design for metabolic disease targets. Their platform integrates target identification, peptide generation, ADMET prediction, and synthesis planning into a single workflow. The approach has produced GLP-1 receptor agonist candidates with novel sequences and improved pharmacokinetic profiles compared to known compounds.
AI Peptide Design: Key Metrics
- Discovery timeline compression: From 4-6 years (traditional) to 6-18 months (AI-assisted)
- Hit rate improvement: AI-generated libraries show 5-20x higher hit rates than random peptide libraries
- Cost reduction: Estimated 60-80% reduction in pre-clinical discovery costs
- Novel chemical space: AI models can propose sequences with no homology to natural peptides, accessing previously unexplored functional space
Practical Applications Across Therapeutic Areas
AI-designed peptides are entering development across multiple disease areas:
| Application | AI Approach | Stage | Key Result |
|---|---|---|---|
| Antimicrobial peptides | Diffusion models + protein LM | Preclinical validated | 46/46 synthesized peptides showed antibacterial activity |
| GLP-1 receptor agonists | Generative models + AlphaFold | Preclinical | Novel sequences with improved PK profiles |
| PDC targeting peptides | AlphaFold2 docking | Phase 2 (MP-0250) | 34% ORR in NSCLC |
| Anticancer peptides | CNN/LSTM classifiers | Preclinical | Selective cytotoxicity in tumor cell lines |
| Peptide vaccines | Neoantigen prediction models | Phase 1/2 | Strong T-cell responses in melanoma patients |
Remaining Challenges
Despite rapid progress, significant gaps remain between computational prediction and clinical reality. Predicting solubility, aggregation propensity, and immunogenicity from sequence alone remains unreliable. Many AI models are trained on small, biased datasets that don't represent the full diversity of peptide function. And the gap between in silico binding predictions and in vivo efficacy still requires extensive experimental validation.
The most successful programs are those that combine AI-driven design with high-throughput experimental validation in tight feedback loops - using each round of experimental data to improve model predictions for the next cycle.
Company Profiles: AI-First Peptide Discovery
Several companies have built their entire business model around AI-driven peptide discovery:
Nuritas (Dublin, Ireland): Nuritas uses deep learning and genomics to discover bioactive peptides from natural sources - particularly food proteins. Their AI platform analyzes protein databases to predict which fragments will have specific biological activities (anti-inflammatory, antimicrobial, metabolic regulation). The company has partnerships with major food and pharmaceutical companies and has moved several AI-discovered peptides into commercial development for functional food and therapeutic applications.
Peptilogics (Pittsburgh, USA): Peptilogics combines AI-driven peptide design with a focus on antimicrobial applications. Their platform generates novel peptide sequences optimized for antimicrobial potency, minimal mammalian toxicity, and favorable pharmacokinetics. PLG0206, their lead clinical candidate, is an IV-administered antimicrobial peptide for prosthetic joint infections that has completed Phase 1 trials.
Evotec (Hamburg, Germany): Evotec's peptide discovery platform integrates AI-driven design with high-throughput synthesis and screening. They have multiple partnerships with major pharmaceutical companies for AI-assisted peptide drug development across metabolic disease, oncology, and inflammation.
Insilico Medicine (Hong Kong): While primarily focused on small molecules, Insilico Medicine's Chemistry42 generative chemistry platform has been applied to peptide design challenges. Their approach uses reinforcement learning and generative adversarial networks to explore peptide chemical space efficiently. The company has demonstrated the ability to generate and validate novel peptide structures for multiple therapeutic targets in compressed timelines.
The AI Peptide Discovery Workflow
A typical AI-driven peptide discovery campaign now follows a structured workflow:
- Target selection and data curation (Weeks 1-2): Define the therapeutic target and curate existing structure-activity relationship (SAR) data for training the AI model. This includes known active peptides, crystal structures of the target protein, and any available binding affinity data.
- Model training and generation (Weeks 2-4): Train generative models on the curated dataset and generate thousands to millions of candidate peptide sequences. Filter candidates using discriminative models that predict activity, toxicity, solubility, stability, and other drug-like properties.
- In silico validation (Weeks 4-6): Use molecular dynamics simulations, docking studies (often AlphaFold2-assisted), and ADMET prediction models to prioritize the top 50-200 candidates for synthesis. This computational triage eliminates the vast majority of candidates before any wet-lab work.
- Synthesis and screening (Weeks 6-12): Synthesize prioritized peptides using automated SPPS and screen them in biochemical and cell-based assays. Typically, 30-60% of AI-predicted active peptides show confirmed activity - far higher than the 1-5% hit rate of traditional high-throughput screening.
- Optimization cycle (Weeks 12-20): Feed experimental results back into the AI model for iterative optimization. Each cycle improves model accuracy, and 2-3 optimization rounds typically identify lead candidates with drug-like properties.
- Lead characterization (Weeks 20-30): Full pharmacological characterization of 3-5 lead candidates including in vitro ADMET, selectivity profiling, preliminary PK studies, and initial toxicology assessment.
This entire workflow - from target selection to lead identification - can be completed in 6-8 months. Traditional peptide discovery campaigns typically require 2-4 years for the same outcome. The time and cost savings are transformative, particularly for academic labs and smaller biotechs that lack the resources for large-scale traditional screening campaigns.
Limitations and the Validation Gap
It's worth being honest about what AI can and cannot do in peptide design. Current models excel at:
- Generating diverse, novel peptide sequences
- Predicting binding affinity to well-characterized targets
- Filtering for basic drug-like properties (solubility, stability)
- Optimizing known peptide leads through rational mutations
But they struggle with:
- Predicting immunogenicity - whether a peptide will trigger unwanted immune responses in patients
- Modeling complex in vivo pharmacokinetics - how a peptide behaves in a living organism is far more complex than in silico predictions suggest
- Predicting off-target effects - interactions with unintended biological pathways
- Handling data-poor targets - AI models need training data, and novel targets with little existing SAR information remain challenging
- Predicting formulation behavior - how a peptide behaves in a delivery vehicle (stability, aggregation, release kinetics) is difficult to model computationally
The "validation gap" between computational prediction and clinical outcome remains real. An AI can predict that a peptide will bind a target with nanomolar affinity, but it cannot yet reliably predict whether that binding will produce a therapeutic effect in patients, at what dose, with what side effects, and for how long. Closing this gap requires continued investment in both computational methods and the experimental validation infrastructure to test AI-generated predictions at scale.
Myostatin and Activin Inhibitors: Preserving Muscle in the GLP-1 Era
The biggest unresolved problem with GLP-1 receptor agonist therapy is muscle loss. Patients on semaglutide or tirzepatide lose 25-40% of their total weight as lean mass rather than fat. Myostatin and activin pathway inhibitors may provide the solution.
The Muscle Mass Problem
GLP-1 receptor agonists don't discriminate between fat and muscle when driving weight loss. In the STEP trials for semaglutide 2.4 mg, approximately 39% of total weight lost was lean body mass. For tirzepatide, the lean mass fraction was similar at roughly 25-33% depending on the dose and study.
In younger, otherwise healthy patients, this lean mass loss may be tolerable and partially recoverable with resistance training. But in older adults, patients with sarcopenia, or those on long-term GLP-1 therapy, the cumulative loss of skeletal muscle raises real concerns about functional capacity, fall risk, metabolic rate, and long-term outcomes.
Bimagrumab: The Leading Candidate
Bimagrumab is a monoclonal antibody that blocks activin type II receptors (ActRIIA and ActRIIB), preventing the binding of myostatin, activin A, and other TGF-beta superfamily ligands that inhibit muscle growth. By blocking these negative regulators, bimagrumab promotes skeletal muscle hypertrophy while simultaneously reducing fat mass.
In a completed trial (NCT03005288) of obese adults with type 2 diabetes, bimagrumab dosed every four weeks for 48 weeks produced remarkable body composition changes:
- Fat mass: ~20% decrease
- Lean muscle mass: ~4% increase
- Total body weight: ~6.5% decrease
- HbA1c: ~0.76 percentage point decrease
The combination of fat loss with simultaneous muscle gain is unique among pharmacological interventions. No approved weight loss drug has demonstrated this profile. This has generated enormous interest in combining bimagrumab with GLP-1 receptor agonists.
Combination Trials: Bimagrumab + GLP-1 Agonists
Two key trials are testing this combination approach:
- NCT05616013 - Bimagrumab combined with semaglutide in obesity. This Versanis Bio (now acquired by Eli Lilly) trial aims to demonstrate that the combination preserves lean mass while enhancing total weight loss.
- NCT05933499 - Bimagrumab as monotherapy in obesity, providing updated monotherapy data with a broader patient population.
Other Myostatin/Activin Pathway Inhibitors
| Agent | Mechanism | Phase | Indication | Notes |
|---|---|---|---|---|
| Bimagrumab | ActRII blocking antibody | Phase 2 | Obesity (+/- GLP-1) | Simultaneous muscle gain + fat loss |
| Taldefgrobep alfa | Anti-myostatin adnectin | Phase 3 | Spinal muscular atrophy | Selective myostatin binding |
| Trevogrumab | Anti-myostatin antibody | Phase 2 | Obesity (+ GLP-1 combo) | Regeneron; paused, evaluating |
| Garetosmab | Anti-activin A antibody | Phase 2 | Obesity (NCT06299098) | Regeneron |
| SRK-439 | Anti-myostatin antibody | Phase 1 | Obesity (NCT06445075) | Scholar Rock |
Taldefgrobep alfa is an anti-myostatin adnectin - a small protein that binds myostatin with high affinity and specificity without affecting other TGF-beta superfamily ligands. Unlike bimagrumab, which blocks the receptor and thus inhibits multiple ligands, taldefgrobep is selective for myostatin alone. After modest results in Duchenne muscular dystrophy (a 4.9% increase in lean body mass index but no change in motor function), the DMD program was terminated. However, a Phase 3 study in spinal muscular atrophy (NCT05337553) is ongoing, and early-stage evaluation for obesity has begun.
The Bigger Picture for Body Composition
The convergence of GLP-1 agonists (fat loss) with myostatin inhibitors (muscle preservation) represents a potential shift in obesity treatment philosophy - from simply reducing body weight to optimizing body composition. If combination trials succeed, future obesity treatment protocols may routinely include both a metabolic peptide and a muscle-sparing agent.
Myostatin Inhibitors in Aging and Sarcopenia
Beyond obesity, myostatin inhibitors have significant potential in age-related muscle loss (sarcopenia). Sarcopenia affects approximately 10-16% of adults over 60 and is associated with increased fall risk, disability, hospitalization, and mortality. Currently, no FDA-approved drug treats sarcopenia - exercise remains the only proven intervention.
The myostatin pathway is an attractive target for sarcopenia because myostatin expression increases with age while muscle regeneration capacity decreases. Preclinical studies show that myostatin inhibition can partially reverse age-related muscle loss, improve muscle strength, and enhance functional capacity in aged animals. If these findings translate to humans, myostatin inhibitors could become the first pharmacological treatment for one of the most consequential conditions of aging.
Bimagrumab's clinical data in obese adults showing a 4% increase in lean mass is encouraging in this context, though dedicated sarcopenia trials with functional endpoints (gait speed, chair stand time, handgrip strength) are needed. The challenge is that sarcopenia trials require long durations, large sample sizes, and functional outcomes that are harder to measure than body weight - making them expensive and slow. Nevertheless, the enormous potential market (hundreds of millions of older adults globally) makes this a compelling development opportunity.
The Exercise-Mimetic Potential
An intriguing aspect of myostatin pathway inhibition is its overlap with exercise-induced muscle adaptation. Regular resistance exercise naturally suppresses myostatin signaling - this is one mechanism by which exercise promotes muscle growth. Myostatin inhibitors may therefore produce some of the same muscle-building effects as exercise, without the physical activity itself.
This has raised both excitement and concern. Excitement because pharmacological exercise mimetics could help patients who are unable to exercise (bedridden, frail elderly, wheelchair-bound). Concern because they could also reduce motivation for physical activity in patients who are able to exercise - and the cardiovascular, metabolic, and psychological benefits of exercise extend far beyond muscle mass.
The clinical reality will likely be that myostatin inhibitors complement exercise rather than replace it. Patients who combine myostatin inhibition with resistance training would be expected to achieve greater muscle gains than either approach alone. The optimal use case may be in post-surgical rehabilitation, recovery from critical illness, or as an adjunct to structured exercise programs in older adults with sarcopenia.
mRNA-Encoded Peptides: Using the Body as a Peptide Factory
What if instead of manufacturing peptides in a factory and injecting them, you could instruct the body's own cells to produce therapeutic peptides on demand? mRNA technology - validated at massive scale by COVID-19 vaccines - is now being adapted to do exactly that.
The Concept
mRNA-encoded peptide therapy delivers synthetic messenger RNA that encodes the amino acid sequence of a therapeutic peptide or protein. Once the mRNA enters cells (typically via lipid nanoparticle delivery), the cell's ribosomal machinery translates it into the desired peptide. The peptide is then secreted or acts intracellularly, producing a therapeutic effect that lasts as long as the mRNA remains intact - typically days to weeks.
This approach offers several advantages over direct peptide administration:
- Continuous endogenous production - Cells produce peptide over time rather than relying on a single bolus injection
- Correct post-translational modifications - Cell-produced peptides undergo natural folding, glycosylation, and processing
- No manufacturing of the peptide itself - The challenge shifts from peptide synthesis to mRNA production, which is highly scalable
- Programmable duration - Modified nucleosides and delivery formulations can tune expression duration
- Rapid development - New peptide sequences can be encoded in weeks once the platform is established
Current Clinical Programs
Moderna and BioNTech - the two companies that scaled mRNA production for COVID-19 vaccines - are expanding into peptide and protein therapeutics:
- mRNA-1975 and mRNA-1982 (Moderna) - These mRNA therapeutics for Lyme disease are in Phase 1/2 clinical trials (NCT05975099). They encode protein antigens from Borrelia burgdorferi to generate immune responses.
- BNT164a1 and BNT164b1 (BioNTech) - Phase 1/2 clinical trials (NCT05547464) for tuberculosis, encoding mycobacterial antigens.
- Moderna-Immatics collaboration - Applying Moderna's mRNA technology to express half-life extended TCR bispecifics that target cancer-specific HLA-presented peptides. This approach effectively turns the patient's cells into bispecific antibody factories.
mRNA-Encoded Peptides for Metabolic Disease
While most current mRNA programs focus on infectious disease and oncology, the potential for metabolic peptide therapies is considerable. Imagine an mRNA injection that instructs liver cells to produce a long-acting GLP-1/GIP agonist peptide for weeks at a time, eliminating the need for weekly injections entirely. Or an mRNA that encodes a myostatin propeptide inhibitor, producing continuous muscle-protective effects.
These applications remain preclinical, but the foundational technology has been demonstrated. The primary challenges are:
- Controlling expression levels precisely enough for therapeutic peptides with narrow dose-response windows
- Ensuring tissue-specific targeting (e.g., liver vs. muscle vs. adipose tissue)
- Managing immune responses to the mRNA delivery vehicle with repeated dosing
- Achieving expression durations measured in weeks rather than days
Gene-Encoded Peptide Therapies
Looking further ahead, gene therapy approaches using AAV vectors to deliver genes encoding therapeutic peptides could provide permanent or semi-permanent peptide expression. While this moves beyond "peptide medicine" into gene therapy territory, it represents the logical endpoint of using biological systems to produce therapeutic peptides.
Several preclinical programs are exploring AAV-encoded GLP-1 analogs and other metabolic peptides, with the goal of single-dose treatments that produce therapeutic peptide levels for months to years. The University of Pennsylvania has demonstrated AAV-mediated expression of a GLP-1 analog in mice that produced sustained weight loss for over six months after a single intravenous injection. While significant hurdles remain before human translation (including dose-dependent liver toxicity, pre-existing anti-AAV immunity, and the challenge of "turning off" gene expression if adverse effects occur), the concept is scientifically validated.
Circular RNA (circRNA) Approaches
An alternative to linear mRNA, circular RNA molecules lack free ends and are therefore resistant to exonuclease degradation. This results in dramatically longer intracellular persistence - circRNAs can express proteins for days to weeks compared to hours for conventional mRNA. For peptide-encoding applications, circRNA could provide sustained endogenous peptide production from a single dose, bridging the gap between conventional mRNA (hours of expression) and gene therapy (permanent expression).
Orna Therapeutics and several academic groups are developing circRNA platforms for therapeutic protein and peptide expression. The technology is still in early development, but the potential for extended-duration peptide expression without the permanence (and risks) of gene therapy makes circRNA an attractive platform for metabolic and chronic disease peptide applications.
Self-Amplifying RNA (saRNA)
Self-amplifying RNA molecules contain the gene of interest plus the coding sequence for RNA-dependent RNA polymerase (RdRp), enabling intracellular amplification of the mRNA without DNA integration. A small initial dose of saRNA produces much higher levels of protein expression than an equivalent dose of conventional mRNA, and the expression persists longer (weeks rather than days).
For peptide-encoding applications, saRNA could enable therapeutic peptide expression at clinically relevant levels from very small injection volumes. The first saRNA vaccine (ARCT-154 by Arcturus Therapeutics) received regulatory approval in Japan in 2023 for COVID-19, demonstrating the clinical feasibility of the platform. Adaptation for therapeutic peptide expression is a logical next step, with potential applications in metabolic disease, immunotherapy, and rare genetic conditions.
Challenges Specific to mRNA-Encoded Peptides
Several challenges are unique to the mRNA-encoded peptide approach and need resolution before widespread clinical adoption:
- Dose precision - Traditional peptide drugs are administered at precise doses. mRNA-encoded peptides produce variable expression levels depending on the efficiency of cellular uptake, translation, and secretion. For peptides with narrow therapeutic windows (like insulin), this variability could be clinically dangerous. For peptides with wider therapeutic windows (like GLP-1 agonists), variability may be acceptable.
- Tissue targeting - Current LNP delivery systems preferentially target the liver after intravenous administration. For peptides that need to act systemically (GLP-1 agonists) or in specific non-liver tissues (muscle for myostatin inhibitors, brain for neuropeptides), alternative targeting strategies are needed.
- Immune tolerance - Repeated administration of mRNA-LNP formulations can trigger adaptive immune responses against the LNP components, potentially reducing efficacy with repeated dosing. This is less of an issue for vaccines (where immune stimulation is desired) but could be problematic for chronic peptide therapy requiring repeated dosing.
- Post-translational modifications - While cell-produced peptides undergo natural post-translational modifications, the specific modifications depend on the cell type expressing the mRNA. Liver-expressed peptides may have different glycosylation patterns than those produced in muscle or adipose tissue, potentially affecting activity and immunogenicity.
Long-Acting Formulations: From Weekly to Monthly and Beyond
The shift from daily to weekly injections transformed GLP-1 therapy. The next frontier is monthly, quarterly, and even semi-annual dosing through advanced depot technologies.
Current State: Weekly Dosing Standard
Semaglutide, tirzepatide, and most investigational peptide drugs use once-weekly subcutaneous injection. This was a major improvement over daily dosing but still requires 52 injections per year, regular medication management, and cold chain storage. For chronic diseases that require lifelong treatment, further extending dosing intervals would significantly improve adherence, convenience, and patient satisfaction.
Technologies Enabling Monthly+ Dosing
Several platform technologies are in development to extend peptide drug duration of action:
Biodegradable Microspheres (PLGA/PLA)
The most established long-acting injectable platform, used in 11 FDA-approved peptide formulations including leuprolide depot (Lupron Depot) and octreotide LAR (Sandostatin LAR). Peptides are encapsulated in poly(lactic-co-glycolic acid) microspheres that slowly erode over 1-6 months, releasing the drug at a controlled rate. Current research focuses on improving encapsulation efficiency for larger peptides and reducing the initial burst release that can cause dose-related side effects.
In Situ Forming Hydrogels
Injectable solutions that gel upon contact with physiological conditions (temperature, pH, or ion concentration), forming a depot that releases peptide over extended periods. A particularly promising approach uses enzyme-responsive peptoid-peptide hydrogels that form in situ and respond to local tissue enzymes for controlled degradation and drug release (Webber et al., JACS, 2024).
In a preclinical diabetes model, single administrations of hydrogel-based formulations of either semaglutide or tirzepatide maintained therapeutically relevant drug levels for over six weeks (Chen et al., bioRxiv, 2025). If this translates to humans, it could enable monthly or even bimonthly dosing for the most popular weight loss peptides.
Subcutaneous Depot Formulations
Ascletis Pharma's ASC30, a small molecule GLP-1 receptor agonist formulated as an ultra-long-acting subcutaneous depot, produced encouraging Phase 2 results. Three monthly doses achieved a placebo-adjusted mean weight loss of 7.5% at week 16. This demonstrates that monthly dosing intervals are clinically viable for GLP-1-based obesity treatment.
Biodegradable Implants
Solid biodegradable implants placed subcutaneously can release peptide drugs over 3-12 months. Histrelin implant (Vantas, Supprelin LA) already provides 12-month GnRH agonist therapy from a single implant. Similar approaches are being developed for metabolic peptides, though the larger doses required present formulation challenges.
The Future Dosing Landscape
| Dosing Frequency | Technology | Status | Examples |
|---|---|---|---|
| Weekly | Half-life extended peptides | Approved, standard | Semaglutide, Tirzepatide |
| Biweekly | Fc-fusion, albumin binding | Phase 2-3 | Next-gen GLP-1 variants |
| Monthly | PLGA microspheres, depots | Phase 2 | ASC30 depot, hydrogel semaglutide |
| Quarterly | Hydrogel depots, implants | Preclinical | Extended-release GLP-1 depots |
| Semi-annual | Biodegradable implants | Preclinical | GnRH analog precedent exists |
| Annual | Solid implants | Approved (for GnRH) | Histrelin (Supprelin LA) |
Why Dosing Interval Matters
Medication adherence drops significantly as dosing frequency increases. Studies show approximately 80-85% adherence for weekly injections, but real-world data for GLP-1 agonists suggests that only 30-50% of patients remain on therapy at 12 months. Long-acting formulations that reduce injection frequency could dramatically improve persistence and real-world effectiveness of peptide therapies.
Crystal Depot Technology
A novel approach to long-acting peptide delivery uses controlled crystallization to create injectable peptide crystal suspensions. The principle is straightforward: peptide crystals dissolve slowly at the injection site, providing sustained drug release over weeks to months. The dissolution rate depends on crystal size, morphology, and the peptide's intrinsic solubility.
Crystal depot technology has been successfully applied to long-acting insulin formulations (insulin glargine achieves prolonged action partly through crystallization at the injection site). Researchers are now applying similar principles to GLP-1 agonists and other therapeutic peptides, engineering crystal forms optimized for specific release profiles.
The advantages of crystal depots include high drug loading (no polymer carrier needed, so a higher fraction of the injected material is active drug), predictable dissolution kinetics, and the potential for very long duration (3-6 months from a single injection). Challenges include controlling crystal polymorphism (different crystal forms can have different dissolution rates), ensuring consistent resuspension before injection, and managing the injection volume for high-dose peptides.
Implantable Osmotic Pumps
Osmotic pump implants deliver peptide drugs at a constant rate through osmotically-driven flow. The DUROS technology (Intarcia Therapeutics) demonstrated that an implantable osmotic pump could deliver exenatide continuously for 3-6 months from a single subdermal implant. While Intarcia's program faced regulatory challenges, the technology concept is validated and could be applied to next-generation peptide drugs.
The advantages of osmotic pump delivery include zero-order release kinetics (constant drug delivery without peaks and troughs), no patient compliance requirements (the implant works continuously), and very long duration (up to 12 months per implant). The disadvantages include the need for a minor surgical procedure for implantation and removal, the inability to easily adjust dosing, and the risk of local site reactions.
Reversible Implants and On-Demand Control
Looking further ahead, research groups are developing implantable peptide delivery devices that can be wirelessly controlled. Microelectromechanical systems (MEMS) devices containing multiple peptide reservoirs can be triggered by wireless signals to release precise doses on demand. This approach combines the convenience of an implant with the dosing flexibility of on-demand delivery.
MIT researchers have demonstrated a wirelessly controlled, implantable drug delivery device that can deliver precise doses of insulin in response to external signals. Adapting this technology for GLP-1 agonists or other therapeutic peptides could enable truly personalized, responsive peptide therapy - adjusting doses based on real-time metabolic data from wearable sensors.
The regulatory pathway for such integrated delivery systems is complex, requiring approval as both a drug and a device (combination product). But the potential to transform chronic peptide therapy from passive (fixed dosing) to active (responsive dosing) makes this a compelling long-term research direction.
Cell-Penetrating Peptides: Unlocking Intracellular Targets
Most drugs work by binding receptors on cell surfaces. But many disease-relevant targets sit inside cells - in the cytoplasm, nucleus, or mitochondria. Cell-penetrating peptides (CPPs) can carry therapeutic cargo across cell membranes, opening up an entirely new category of intracellular drug targets.
How Cell-Penetrating Peptides Work
CPPs are typically short (5-30 amino acid) peptides that can cross cell membranes through several mechanisms. Cationic CPPs like TAT (derived from HIV-1) interact electrostatically with negatively charged phospholipids and carboxyl groups on the cell surface. Amphipathic CPPs use a combination of charged and hydrophobic residues to insert into and traverse the lipid bilayer. The exact mechanism varies by CPP type, cargo size, cell type, and conditions - and remains an active area of research.
What matters clinically is that CPPs can ferry cargoes into cells that would otherwise be impermeable: small molecules, proteins, nucleic acids, nanoparticles, and even full-length antibodies.
Clinical Programs Using CPPs
Several CPP-based therapeutics are in clinical development:
| Compound | CPP Used | Cargo | Indication | Phase |
|---|---|---|---|---|
| PGN-EDO51 | Proprietary CPP | Exon-skipping oligonucleotide | Duchenne muscular dystrophy | Phase 1 (NCT06079736) |
| Nomlabofusp | TAT | Human frataxin protein | Friedreich's ataxia | Phase 1 (NCT06681766) |
| p28 (azurin fragment) | p28 (self-penetrating) | Self (anticancer activity) | Glioblastoma, solid tumors | Phase 1 (NCT00914914) |
| XG-102 (AM-111) | TAT-derived | JNK inhibitor peptide | Hearing loss, inflammation | Phase 3 |
PGN-EDO51 is particularly noteworthy. In Duchenne muscular dystrophy, the therapeutic goal is to skip a specific exon in the dystrophin mRNA to restore production of a partially functional dystrophin protein. The challenge has always been getting enough oligonucleotide into muscle cells. By conjugating the exon-skipping oligonucleotide to a cell-penetrating peptide, PepGen (the developer) has shown effective exon skipping and dystrophin production in preclinical models and is now testing this in healthy volunteers.
Nomlabofusp combines the TAT CPP with full-length human frataxin to treat Friedreich's ataxia, a genetic disease caused by frataxin deficiency. The CPP enables delivery of the replacement protein directly into cells and mitochondria, where frataxin functions. Phase 1 data has confirmed increased frataxin levels in treated patients.
CPPs in Cancer Immunotherapy
CPPs are finding growing applications in cancer immunotherapy. They can deliver tumor-associated antigens, immune checkpoint inhibitors, and cytokines directly to immune cells or tumor cells, potentially improving efficacy while reducing systemic side effects. Recent research has explored CPP-mediated delivery of STING agonists, TLR ligands, and tumor-specific peptide antigens to dendritic cells for enhanced anti-tumor immune responses.
Limitations and Solutions
The main limitations of CPPs are lack of cell-type specificity (they penetrate most cell types, not just target cells), limited metabolic stability, and rapid clearance. Current research addresses these through:
- Activatable CPPs - Peptides that are masked in circulation and only become cell-penetrating in the tumor microenvironment (low pH, specific proteases)
- Targeted CPP conjugates - Combining a cell-type-selective targeting peptide with a CPP to achieve organ- or cell-type specificity
- D-amino acid substitution - Using mirror-image amino acids to resist protease degradation
- Cyclization - Constraining the peptide structure to improve stability and potentially enhance cell selectivity
CPP-Mediated Gene Editing Delivery
One of the most exciting emerging applications for cell-penetrating peptides is the delivery of gene editing components. CRISPR-Cas9 and base editing systems require delivery of large protein-nucleic acid complexes into target cells - a significant delivery challenge, particularly for in vivo applications where viral vectors raise safety concerns.
CPPs can form non-covalent complexes with Cas9 ribonucleoprotein (RNP) particles, facilitating their entry into cells without the need for viral delivery. This approach offers several advantages over viral vectors: no risk of insertional mutagenesis, no pre-existing immune responses against viral capsids, transient expression that limits off-target editing, and rapid manufacturing without cell culture.
Preclinical studies have demonstrated CPP-mediated delivery of CRISPR-Cas9 to multiple cell types, including T cells (for CAR-T cell engineering), hepatocytes (for metabolic disease gene correction), and hematopoietic stem cells (for sickle cell disease and beta-thalassemia). The efficiency of CPP-mediated CRISPR delivery is typically lower than viral delivery, but optimization of CPP sequences, RNP formulation, and delivery conditions is steadily closing this gap.
If CPP-CRISPR delivery matures to clinical readiness, it could enable a new category of peptide-facilitated gene therapies - using peptide technology not as the drug itself, but as the vehicle that delivers genetic medicines to their intracellular targets. This intersection of peptide delivery science with gene editing therapeutics represents one of the most promising convergence opportunities in the field.
Industrial Applications of CPPs
Beyond therapeutics, CPPs have found applications in several non-pharmaceutical areas that validate the technology and drive continued innovation. Agricultural CPPs deliver pesticides and growth regulators into plant cells with improved efficiency. Cosmetic CPPs deliver anti-aging peptides (argireline, matrixyl) into skin cells more effectively than conventional topical formulations. Research CPPs enable intracellular delivery of molecular probes, imaging agents, and experimental compounds for basic science research. These diverse applications provide revenue streams and optimization data that feed back into therapeutic CPP development.
Peptide Vaccines: Personalized Cancer Immunotherapy
Peptide-based cancer vaccines are entering a new era of personalized medicine. By targeting neoantigens - mutant peptides unique to each patient's tumor - these vaccines train the immune system to recognize and destroy cancer cells with unprecedented specificity.
The Neoantigen Approach
Every tumor accumulates somatic mutations during its evolution. Some of these mutations change the amino acid sequence of cellular proteins, creating neoantigens - peptide fragments that are presented on the cell surface by HLA molecules and can be recognized by T cells as "foreign." Because these neoantigens are unique to cancer cells and absent from healthy tissues, they represent ideal vaccine targets.
The workflow for personalized neoantigen vaccines typically involves:
- Tumor biopsy and sequencing - Whole exome and RNA sequencing to identify somatic mutations
- Neoantigen prediction - AI algorithms predict which mutant peptides will bind the patient's specific HLA type
- Peptide synthesis - Synthetic long peptides (25-30 amino acids) are manufactured for each patient
- Vaccine formulation - Peptides are combined with immune adjuvants (poly-ICLC, GM-CSF) to enhance immune activation
- Administration and monitoring - Vaccination plus immune response assessment via blood tests and imaging
Clinical Pipeline
As of late 2024, 78 personalized cancer vaccine trials were registered on ClinicalTrials.gov. Peptide vaccines are the most common platform with 31 trials, followed by dendritic cell vaccines (15 trials), RNA vaccines (13 trials), and DNA vaccines (8 trials).
Recent clinical highlights include:
- PGV001 (Mount Sinai) - A personalized multi-peptide neoantigen vaccine tested in a Phase 1 trial. At five-year follow-up, 6 of 13 treated patients survived, and 3 of 6 surviving patients were tumor-free. No serious side effects were observed.
- EVX-01 (Evaxion Biotech) - A personalized peptide neoantigen vaccine with the novel adjuvant CAF09b, tested in metastatic melanoma patients. Published results demonstrated strong neoantigen-specific T-cell responses.
- iNATURE trial - A Phase 2 randomized trial combining individualized neoantigen peptide vaccines with radiotherapy in advanced solid tumors (NCT registered as GCOG0028). This study explores whether radiation can synergize with vaccination by releasing additional tumor antigens.
- PNeoVCA (Mayo Clinic) - Phase 1 study combining personalized neoantigen peptide vaccine with pembrolizumab in advanced solid tumors (NCT05269381). This combination approach aims to amplify vaccine-induced immune responses with checkpoint blockade.
Manufacturing Speed Improvements
One of the historical barriers to personalized cancer vaccines has been manufacturing time. Early programs required 3-6 months from biopsy to first vaccination - too slow for patients with aggressive cancers. Current-generation platforms have compressed this to 48-55 days from biopsy to finished vaccine, with treatment initiation within 60 days. This still needs improvement, but it represents dramatic progress.
Combination Strategies
The most promising results are emerging from combinations of peptide vaccines with immune checkpoint inhibitors (anti-PD-1/PD-L1 antibodies). The rationale is straightforward: the vaccine activates tumor-specific T cells, while checkpoint inhibitors remove the brakes that tumors use to suppress those T cells. Early data suggests these combinations produce stronger and more durable immune responses than either approach alone.
Where Peptide Vaccines Have Shown the Most Promise
Clinical data is strongest in melanoma (high mutation burden, well-characterized neoantigens), glioblastoma (limited treatment options, blood-brain barrier challenges favor immunological approaches), urothelial cancer, and pancreatic cancer. Large-scale randomized trials, particularly in adjuvant settings, are needed to confirm survival benefits suggested by these early-phase studies.
Shared Neoantigen Vaccines
While fully personalized vaccines require individual manufacturing for each patient, shared neoantigen vaccines target mutations that occur recurrently across patients with the same cancer type. For example, the KRAS G12D mutation is found in approximately 40% of pancreatic cancers and 12% of colorectal cancers. A peptide vaccine targeting KRAS G12D could serve thousands of patients without individualized manufacturing.
Several companies are developing shared neoantigen vaccines targeting common driver mutations. BioNTech's autogene cevumeran (BNT122) is a personalized mRNA vaccine encoding up to 20 neoantigens, but the company is also developing "off-the-shelf" mRNA vaccines targeting shared cancer antigens. These off-the-shelf approaches sacrifice some specificity but gain manufacturing scalability - they can be produced in large batches and stored for immediate use, eliminating the 48-55 day manufacturing delay of fully personalized vaccines.
The hybrid approach - combining a shared neoantigen backbone with a personalized component - may offer the best balance of speed, scalability, and specificity. A patient could receive an immediate off-the-shelf vaccine targeting shared mutations while their personalized vaccine is being manufactured, then switch to the personalized version when it's ready.
Peptide Vaccine Adjuvant Innovation
The efficacy of peptide vaccines depends heavily on adjuvants - substances that enhance the immune response to the vaccine peptides. Without adjuvants, short peptide fragments are poorly immunogenic and rapidly cleared. Current adjuvant approaches include poly-ICLC (a TLR3 agonist), Montanide ISA-51 (a water-in-oil emulsion), CpG oligonucleotides (TLR9 agonists), and GM-CSF (a cytokine that recruits and activates dendritic cells).
Next-generation adjuvant technologies are being developed specifically for peptide cancer vaccines:
- STING agonists - Stimulator of Interferon Genes pathway activation produces a strong type I interferon response that enhances CD8+ T cell priming - exactly the immune response needed for anti-cancer immunity. Several STING agonist adjuvants are in clinical development paired with peptide vaccines.
- Self-assembling peptide nanofibers - Neoantigen peptides designed to self-assemble into nanofiber structures act as both the antigen and the delivery vehicle, creating a multivalent display that enhances immune recognition. The nanofiber structure also provides depot-like sustained antigen release at the injection site.
- CAF09b (Statens Serum Institut) - A liposome-based adjuvant specifically designed to promote CD8+ T cell responses to peptide antigens. CAF09b has been tested with the EVX-01 personalized neoantigen vaccine in melanoma, showing strong neoantigen-specific T cell responses in clinical studies.
- TLR7/8 agonist conjugates - Covalently linking a TLR7/8 agonist directly to the peptide antigen ensures that the adjuvant signal is delivered to the same antigen-presenting cell that processes the peptide, improving the quality and specificity of the immune response.
The choice of adjuvant can be as important as the choice of neoantigen peptides in determining vaccine efficacy. Future peptide vaccine development will likely involve systematic optimization of antigen-adjuvant combinations for each cancer type and treatment setting.
Antimicrobial Peptides: Fighting Drug-Resistant Infections
Antibiotic resistance kills an estimated 1.27 million people annually and threatens to reverse a century of progress against infectious disease. Antimicrobial peptides (AMPs) - nature's ancient defense molecules - offer a fundamentally different mechanism of action that may overcome resistance.
Why AMPs Resist Resistance
Conventional antibiotics typically target specific bacterial proteins (ribosomes, cell wall enzymes, DNA gyrase). Bacteria evolve resistance by mutating these targets, producing efflux pumps, or acquiring degrading enzymes. AMPs work differently: they target the bacterial cell membrane itself, disrupting its integrity through electrostatic interactions with membrane lipids. Because fundamentally restructuring membrane composition is much harder than mutating a single protein target, bacteria develop resistance to AMPs at significantly lower rates than to conventional antibiotics.
The AMP Pipeline
The APD6 database, updated in March 2025, catalogs 5,188 antimicrobial peptides, including 3,306 natural, 1,380 synthetic, and 239 computationally predicted sequences. Several AMPs are in clinical development:
| AMP Candidate | Source/Type | Target Infection | Phase | Mechanism |
|---|---|---|---|---|
| Pexiganan (MSI-78) | Magainin analog | Diabetic foot ulcer | Phase 3 (completed) | Membrane disruption |
| Omiganan (MBI-226) | Indolicidin analog | Catheter infections, rosacea | Phase 3 | Membrane permeabilization |
| LL-37 (human cathelicidin) | Human endogenous | Venous leg ulcers | Phase 2 | Membrane disruption + immunomodulation |
| IMX942 (SGX942) | IDR-1 analog | Oral mucositis | Phase 3 | Immunomodulatory |
| Iseganan (IB-367) | Protegrin analog | Oral infections | Phase 3 (completed) | Membrane disruption |
| PAC-113 | Histatin 5 derivative | Oral candidiasis | Phase 2 | Antifungal membrane disruption |
AI Acceleration of AMP Discovery
AI is transforming AMP discovery faster than perhaps any other peptide class. The integration of machine learning with high-throughput screening has produced results that would have been impossible through traditional approaches.
In a study published in Nature Microbiology (2025), researchers used a generative AI approach to discover novel antimicrobial peptides effective against multidrug-resistant bacteria. The AI model processed peptide sequences to predict antimicrobial activity, toxicity, and stability, then generated entirely novel sequences optimized for these properties. From 50,000 computationally generated candidates, experimental validation confirmed that the top-ranked peptides showed broad-spectrum activity against resistant pathogens including MRSA, VRE, and carbapenem-resistant Enterobacteriaceae.
Combination Therapy
AMPs combined with conventional antibiotics frequently show additive or amplifying effects. The membrane-disrupting action of AMPs can increase bacterial permeability to conventional antibiotics, effectively re-sensitizing resistant bacteria to drugs they had evolved to resist. This combination approach may be the most practical near-term application of AMPs - not replacing antibiotics entirely, but restoring the efficacy of existing ones.
Challenges to Clinical Translation
Despite decades of research, no AMP has achieved mainstream FDA approval for systemic infection treatment. The challenges are real:
- Serum instability - Many AMPs are rapidly degraded by proteases in blood
- Hemolytic toxicity - Some AMPs damage red blood cells at concentrations near the therapeutic dose
- Manufacturing cost - Peptide synthesis remains expensive compared to small molecule antibiotics
- Salt sensitivity - Physiological salt concentrations can reduce AMP activity
- Narrow therapeutic window - The concentration difference between antimicrobial and cytotoxic effects is sometimes small
Current research addresses these limitations through D-amino acid incorporation, PEGylation, lipidation, cyclization, and the use of peptidomimetics that mimic AMP structure with improved stability.
AMP Applications Beyond Antibiotics
The biological activities of AMPs extend well beyond direct antimicrobial action. Many AMPs have immunomodulatory properties - they can recruit immune cells to infection sites, modulate cytokine production, and bridge innate and adaptive immune responses. This immunomodulatory activity makes AMPs interesting candidates for conditions where infection and inflammation are intertwined.
Specific non-antibiotic AMP applications under investigation include:
- Wound healing - AMPs like LL-37 and human beta-defensins promote wound healing through antimicrobial activity, immune modulation, and direct stimulation of keratinocyte migration and proliferation. LL-37 is in Phase 2 clinical trials for venous leg ulcers.
- Anti-biofilm agents - Bacterial biofilms on medical devices and chronic wounds are inherently resistant to conventional antibiotics. Certain AMPs can disrupt biofilm architecture and kill biofilm-embedded bacteria, making them valuable for treating device-related and chronic wound infections.
- Cancer therapy - Some AMPs show selective toxicity toward cancer cells, which often have altered membrane compositions (increased negative charge, higher phosphatidylserine exposure). Anticancer AMPs are being explored as standalone agents and as components of peptide-drug conjugates.
- Food preservation - Nisin, a lantibiotic AMP produced by Lactococcus lactis, has been used as a food preservative for decades. Newer AMPs are being developed for food safety applications, particularly for controlling antibiotic-resistant foodborne pathogens.
- Antiviral applications - Several AMPs show activity against enveloped viruses (including influenza, HIV, and coronaviruses) by disrupting viral envelopes or blocking viral entry into host cells. The COVID-19 pandemic stimulated significant research into antiviral AMP development.
The Economic Case for AMP Development
Despite the scientific promise, AMP commercial development has been slow. The economics are challenging: AMPs are more expensive to manufacture than small molecule antibiotics, and antibiotic pricing in most markets is too low to support the cost of peptide drug development. The traditional antibiotic business model - high volume, low margin, short treatment courses - doesn't align well with peptide drug economics.
New economic models are needed. Pull incentives (guaranteed revenue regardless of sales volume), transferable exclusivity vouchers, and subscription-based procurement models (like the UK's pilot "Netflix model" for antibiotics) could change the economic calculus for AMP development. Without these incentive reforms, many scientifically promising AMPs may never reach patients, regardless of their therapeutic potential.
The convergence of AI-accelerated discovery (reducing development costs), improved manufacturing technology (reducing production costs), and evolving reimbursement models (improving revenue) may collectively make AMP commercialization viable in the 2025-2030 timeframe. Companies that position themselves at this intersection of technology and market evolution may find significant opportunities.
Targeted Delivery Systems
Getting peptides to the right tissue, in the right concentration, for the right duration remains the central challenge of peptide pharmacology. A new generation of delivery technologies is solving this problem from multiple angles.
Lipid Nanoparticles (LNPs)
Validated at global scale by mRNA COVID-19 vaccines, LNPs are now being applied to peptide delivery. They protect peptide cargo from enzymatic degradation, enhance cellular uptake through endocytosis, and can be engineered with surface ligands for tissue-specific targeting. The transition from mRNA to peptide payloads is relatively straightforward since both are biological macromolecules with similar encapsulation requirements.
Exosome-Based Delivery
Exosomes - natural cell-derived vesicles 30-150 nm in diameter - offer biocompatible peptide delivery with inherent cell-targeting properties. Engineered exosomes can be loaded with therapeutic peptides and decorated with targeting molecules. Their natural origin reduces immunogenicity compared to synthetic nanoparticles. Clinical programs are exploring exosome-delivered peptides for neurological conditions (crossing the blood-brain barrier), cardiac repair, and cancer immunotherapy.
Albumin-Binding Strategies
Human serum albumin has a half-life of approximately 19 days. By engineering peptides to bind albumin non-covalently in circulation, developers can dramatically extend peptide half-life. This is the strategy behind semaglutide's weekly dosing - a fatty acid chain attached to the GLP-1 analog enables albumin binding and slow renal clearance. Newer albumin-binding motifs, including specifically designed albumin-binding peptides and nano-bodies, can achieve even longer circulation times.
Stimuli-Responsive Release Systems
Smart delivery systems that release peptide cargo in response to specific biological triggers are advancing through preclinical development:
- pH-responsive systems - Release payload in acidic tumor microenvironments or inflamed tissues
- Enzyme-responsive systems - Linkers cleaved by disease-specific enzymes (matrix metalloproteinases in tumors, elastase in inflamed joints)
- Glucose-responsive systems - Release insulin or GLP-1 analogs in proportion to blood glucose levels, mimicking natural pancreatic function
- Thermoresponsive systems - Phase-transition polymers that release peptide upon temperature changes, useful for localized hyperthermia-triggered delivery
Microneedle Patches
Dissolving microneedle patches deliver peptides through the skin painlessly. Arrays of microscopic needles (typically 300-800 micrometers long) made of soluble polymer matrices dissolve in the dermis, releasing their peptide payload. This approach eliminates the need for conventional needles, enables self-administration, and may provide more consistent absorption than subcutaneous injection.
Multiple companies are developing microneedle patch formulations for GLP-1 agonists, insulin, and other peptide drugs. If successful, these could combine the convenience of topical application with the bioavailability of injection.
Pulmonary Peptide Delivery
The lungs offer a large surface area (~100 m2), thin epithelial barrier, and rich blood supply - characteristics that make inhalation an attractive alternative to injection for systemic peptide delivery. Inhaled insulin (Afrezza) proved that pulmonary peptide delivery is clinically feasible, though commercial success has been limited by market factors rather than scientific ones.
Advantages of pulmonary delivery for peptides include rapid absorption (peak levels within 10-15 minutes), avoidance of first-pass hepatic metabolism, needle-free administration, and self-administration capability. Challenges include achieving consistent dosing (which depends on inhalation technique, lung function, and respiratory health), formulation stability, and the potential for local pulmonary effects with chronic use.
Several companies are developing inhaled formulations of peptides beyond insulin, including GLP-1 agonists, parathyroid hormone, and antimicrobial peptides for respiratory infections. Dry powder inhalation technology, which eliminates the need for cold chain storage, is particularly attractive for peptide drugs destined for markets with limited cold chain infrastructure.
Transdermal Peptide Delivery Beyond Microneedles
Beyond microneedle patches, several other transdermal peptide delivery technologies are in development:
- Iontophoresis - Using a small electric current to drive charged peptide molecules across the skin barrier. This approach can deliver peptides up to ~10 kDa and has been commercialized for local drug delivery (fentanyl, lidocaine). Adaptation for systemic peptide therapy is under investigation.
- Sonophoresis - Using low-frequency ultrasound to temporarily permeabilize the stratum corneum, allowing peptide transport across the skin. Research has shown effective transdermal delivery of insulin, calcitonin, and other peptides.
- Chemical penetration enhancers - Certain chemicals (fatty acids, surfactants, ionic liquids) can reversibly disrupt skin barrier function to allow peptide passage. The CAGE ionic liquid platform has shown oral delivery capability and is also being explored for transdermal applications.
- Thermal ablation - Brief application of heat creates microscopic channels in the outer skin layer through which peptide molecules can pass. PassPort patch technology uses this approach and has been tested with vaccine peptides and therapeutic proteins.
Oral Mucosa and Sublingual Delivery
The oral mucosa (buccal and sublingual tissues) offers an alternative to GI tract delivery for peptide drugs. The relatively thin mucosa, rich blood supply, and avoidance of hepatic first-pass metabolism make sublingual delivery attractive for peptides that are degraded in the stomach or gut.
Sublingual peptide delivery challenges include the limited absorption area, the continuous washing effect of saliva, and the difficulty of maintaining drug contact with the absorption surface. Mucoadhesive films and wafers that adhere to the oral mucosa and release peptide over time are being developed to address these challenges. Several companies are exploring sublingual GLP-1 agonist formulations, oxytocin wafers, and calcitonin sublingual tablets.
Peptide Diagnostics: Beyond Therapeutics
Peptides aren't just drugs - they're also powerful diagnostic tools. Radiolabeled peptides for PET/SPECT imaging, peptide-based biosensors, and peptide biomarker assays are expanding the diagnostic applications of peptide science.
Radiopeptide Imaging
Radiolabeled peptides that bind specific cell-surface receptors enable visualization of tumors and other disease sites with remarkable sensitivity. The best-established example is gallium-68 DOTATATE PET/CT for neuroendocrine tumors, which has largely replaced older imaging modalities for this cancer type. PSMA-targeted peptides for prostate cancer imaging have similarly transformed diagnostic pathways.
Emerging radiopeptide diagnostics under development include:
- GLP-1 receptor imaging - Radiolabeled exendin-4 derivatives for imaging insulinomas and beta cell mass in diabetes research
- CXCR4-targeted peptides - For imaging hematologic malignancies and bone marrow involvement
- Integrin-targeting RGD peptides - For angiogenesis imaging in tumors and cardiovascular disease
- Amyloid-binding peptides - For early detection of Alzheimer's disease and amyloid cardiomyopathy
Theranostic Peptides
One of the most exciting developments is the theranostic approach - using the same peptide for both diagnosis and therapy by swapping the radionuclide. A diagnostic gamma-emitting isotope (gallium-68) identifies patients with receptor-positive tumors, and the same peptide labeled with a therapeutic beta-emitting isotope (lutetium-177) treats those tumors. Lutathera (lutetium-177 dotatate) and Pluvicto (lutetium-177 PSMA-617) are approved examples of this approach.
Point-of-Care Peptide Biosensors
Peptide aptamers and binding peptides are being integrated into rapid diagnostic devices. Peptide-based lateral flow assays can detect biomarkers for infectious diseases, cardiac events, and cancer with sensitivity approaching laboratory ELISA methods. The advantages of peptide-based biosensors include thermal stability (no cold chain required), low manufacturing cost, and reproducible synthetic production.
Specific applications under development include:
- Cardiac troponin detection - Peptide-based rapid tests for cardiac troponin I and T could enable emergency department rule-out of myocardial infarction within minutes rather than hours
- Sepsis biomarker panels - Multiplex peptide biosensors detecting procalcitonin, presepsin, and other peptide biomarkers for rapid sepsis diagnosis and antibiotic stewardship
- Cancer screening - Peptide-based tests for circulating tumor-derived peptides could enable non-invasive cancer screening from a simple blood draw
- Antimicrobial resistance detection - Peptide probes that detect specific resistance mechanisms (beta-lactamases, carbapenemases) could guide antibiotic selection within hours rather than days
Companion Diagnostics for Peptide Therapies
As peptide therapeutics become more targeted, companion diagnostics become more relevant. The theranostic model - where the same peptide serves as both a diagnostic probe and a therapeutic agent - is expanding beyond nuclear medicine into broader oncology and metabolic disease applications.
For GLP-1 agonist therapy, companion diagnostics may eventually include pharmacogenomic tests that predict individual response to specific GLP-1 agonists, biomarker panels that guide choice between single, dual, and triple agonist therapy, and imaging studies that assess beta cell mass to predict long-term glycemic response.
In oncology, PDC development increasingly integrates companion diagnostics from the earliest stages. Before administering a peptide-drug conjugate targeting a specific tumor receptor, confirming receptor expression in the patient's tumor through biopsy or imaging ensures that the right patients receive the right targeted therapy.
Regulatory Evolution for Peptide Medicines
Regulatory agencies are adapting their frameworks to accommodate the rapid expansion of peptide therapeutics. New guidance documents, expedited pathways, and international harmonization efforts are reshaping how peptide drugs reach patients.
FDA Regulatory Framework Updates
The FDA has increasingly recognized peptides as a distinct pharmaceutical class requiring tailored regulatory approaches. Key developments include:
- National Priority Review Voucher pilot program - Orforglipron was selected for this program in 2025, signaling FDA interest in accelerating oral peptide approval
- Peptide-specific CMC guidance - Updated chemistry, manufacturing, and controls guidance for synthetic peptides addresses characterization, impurity profiles, and stability testing requirements
- Biosimilar vs. generic classification - The FDA's framework for determining whether a peptide drug follows the generic (ANDA) or biosimilar (BLA) approval pathway continues to evolve. Peptides under 40 amino acids generally follow the generic pathway; larger peptides may require the more extensive biosimilar pathway
- Real-world evidence integration - FDA is increasingly accepting real-world data to support label expansion for peptide drugs, particularly for GLP-1 agonists in indications beyond their original approvals
EMA and International Harmonization
The European Medicines Agency (EMA) and other international regulators are working toward harmonized peptide drug evaluation through ICH (International Council for Harmonisation) guidelines. This is particularly relevant for global development programs where sponsors seek simultaneous or near-simultaneous approval in multiple markets.
Compounding Pharmacy Regulation
The explosive demand for GLP-1 agonists has brought peptide compounding under intense regulatory scrutiny. The FDA has tightened oversight of compounding pharmacies producing peptide drugs, particularly semaglutide and tirzepatide during shortage periods. For details on the current regulatory landscape for research peptides, see our science and research section.
Accelerated Approval Considerations
Several peptide therapeutics are pursuing accelerated or conditional approval pathways based on surrogate endpoints. Weight loss percentage, HbA1c reduction, and liver fat fraction reduction are well-established surrogates that enable faster approvals. However, the FDA increasingly requires confirmatory trials demonstrating hard outcomes (cardiovascular events, mortality, liver-related outcomes) post-approval.
The FDA's experience with GLP-1 agonists has shaped its approach to surrogate endpoints for metabolic peptides. The SELECT trial demonstrated that semaglutide 2.4 mg reduced major adverse cardiovascular events (MACE) by 20% in obese/overweight adults without diabetes - providing definitive evidence that weight loss with GLP-1 agonists translates into hard cardiovascular outcomes. This result has important implications for all subsequent metabolic peptide drug approvals: it establishes a benchmark that new agents will be measured against and supports the FDA's position that cardiovascular outcome trials should be required for approval or as post-marketing commitments.
For next-generation multi-agonists like retatrutide and survodutide, the question is whether their superior weight loss (compared to semaglutide) will translate into proportionally greater cardiovascular benefit. The answer isn't guaranteed - weight loss by different mechanisms may not produce identical cardiovascular outcomes. Cardiovascular outcome trials for triple agonists are already incorporated into the TRIUMPH program design for retatrutide, but these trials won't report results until 2028-2029 at the earliest.
Patent and Exclusivity Landscape
The intellectual property landscape for peptide therapeutics is complex and has significant implications for generic competition and patient access. Key considerations include:
Semaglutide patents: Novo Nordisk holds extensive patent protection on semaglutide, including composition of matter patents (core molecule), formulation patents (subcutaneous injection, oral tablet), process patents (manufacturing methods), and use patents (specific therapeutic indications). The core patents begin expiring in the late 2020s, but secondary patents on specific formulations and uses may extend protection into the 2030s. The exact timing of generic/biosimilar entry will depend on patent litigation outcomes and regulatory classification decisions.
Tirzepatide patents: Eli Lilly's tirzepatide patent portfolio includes the novel dual agonist molecule, its specific amino acid sequence modifications, the lipid conjugation strategy, and various formulation and manufacturing process patents. Given that tirzepatide was approved more recently than semaglutide, its core patent protection extends further, likely preventing meaningful generic competition before the early 2030s.
Next-generation compound IP: Retatrutide, orforglipron, survodutide, and other pipeline compounds benefit from fresh patent filing dates, providing 20 years of composition of matter protection from their respective filing dates. Companies are also building extensive patent estates around specific indications, dosing regimens, combination therapies, and delivery technologies to create multiple layers of exclusivity.
Data exclusivity: Beyond patent protection, regulatory data exclusivity provides additional market protection. In the U.S., new chemical entities receive 5 years of data exclusivity (or 12 years for biologics), during which generic/biosimilar applicants cannot reference the innovator's clinical data. The classification of specific peptides as drugs (5 years) versus biologics (12 years) therefore has major commercial implications.
International Regulatory Harmonization
The global nature of peptide drug development makes international regulatory harmonization increasingly important. Companies developing peptide drugs typically seek simultaneous or near-simultaneous approval in the U.S. (FDA), EU (EMA), Japan (PMDA), and China (NMPA). Differences in regulatory requirements across these agencies can increase development costs and delay global access.
ICH (International Council for Harmonisation) guidelines are working toward harmonized approaches for peptide drug quality (Q6B for biotech products, Q3C for impurities), clinical development (E8 for clinical trial design), and safety (S6 for biotechnology-derived pharmaceuticals). These harmonized guidelines enable companies to use a single development program to support regulatory submissions in multiple markets.
However, significant differences remain. The FDA and EMA have different approaches to peptide classification (drug vs. biologic), different requirements for cardiovascular outcome studies, and different standards for biosimilar approval. China's NMPA has its own regulatory framework that sometimes enables faster local development but requires separate clinical trials in Chinese patient populations. Navigating these differences requires dedicated regulatory expertise and careful global development planning.
The WHO prequalification pathway, primarily used for essential medicines in low-income countries, is being considered for GLP-1 agonists as they gain essential medicine status for diabetes management. WHO prequalification would facilitate procurement by international organizations (UNICEF, Global Fund) and enable distribution through established global health channels - potentially extending access to peptide therapies in settings where they are currently unavailable.
Manufacturing Innovation: Scaling Peptide Production
The commercial success of GLP-1 agonists has exposed a critical bottleneck: the world doesn't have enough peptide manufacturing capacity. Billions of dollars are flowing into new facilities and technologies to close the gap.
The Capacity Challenge
Global demand for semaglutide alone consumes a significant fraction of the world's peptide manufacturing capacity. Add tirzepatide, emerging multi-agonists, and the dozens of other peptide drugs in development, and the industry faces a genuine supply challenge. Shortages of semaglutide and tirzepatide in 2023-2025 were directly attributable to manufacturing constraints.
Investment Scale
In December 2025, Eli Lilly announced an investment exceeding $6 billion to build a new active drug ingredient manufacturing facility in Huntsville, Alabama - the company's third such U.S. facility. This site is specifically designed to produce small molecule synthetic and peptide medicines, including orforglipron. Similar billion-dollar manufacturing investments have been announced by Novo Nordisk, Bachem, PolyPeptide Group, and other major peptide manufacturers.
Technology Advances
Continuous-Flow Solid-Phase Peptide Synthesis (CF-SPPS)
Traditional batch SPPS is being replaced by continuous-flow processes that improve reaction efficiency, reduce solvent consumption, and enable real-time quality monitoring. CF-SPPS can reduce synthesis times for complex peptides from days to hours while improving crude peptide purity.
Green Chemistry Approaches
Peptide manufacturing generates significant chemical waste, particularly from coupling reagents and organic solvents. New green chemistry approaches include water-based peptide coupling, recyclable solid supports, and enzymatic ligation methods that reduce the environmental footprint of large-scale production.
Automated Multi-Kilogram Synthesis
Fully automated peptide synthesizers capable of multi-kilogram output per batch are becoming commercially available. These systems integrate synthesis, cleavage, and crude purification into a single automated workflow, reducing labor costs and batch-to-batch variability.
Recombinant Production
For longer peptides (30+ amino acids), microbial fermentation can be more cost-effective than chemical synthesis. Advances in E. coli and yeast expression systems, combined with improved purification methods, are making recombinant production viable for commercial-scale peptide manufacturing.
Enzymatic and Chemoenzymatic Synthesis
Enzymes are increasingly being integrated into peptide manufacturing workflows. Sortase-mediated ligation, butelase-catalyzed cyclization, and other enzymatic approaches can perform specific reactions (such as peptide cyclization or segment condensation) with high selectivity and minimal side products. These enzymatic methods are particularly valuable for reactions that are difficult to achieve with purely chemical approaches.
Chemoenzymatic hybrid strategies combine the efficiency of chemical SPPS for linear precursor synthesis with enzymatic steps for specific modifications. For example, a linear peptide precursor can be synthesized by SPPS, then enzymatically cyclized to form a macrocyclic product. This approach is being used for manufacturing cyclic peptide natural product analogs and constrained peptide libraries.
Supply Chain Resilience
The COVID-19 pandemic exposed vulnerabilities in pharmaceutical supply chains, and the peptide industry is actively working to build resilience. Key strategies include:
- Geographic diversification - Establishing manufacturing capacity in multiple regions (North America, Europe, Asia) to reduce dependence on any single geography
- Raw material sourcing - Qualifying multiple suppliers for critical raw materials including protected amino acids, coupling reagents, and resins
- Strategic inventory - Building buffer stocks of key intermediates and finished drug substance to absorb demand fluctuations
- Vertical integration - Major peptide manufacturers are increasingly producing their own raw materials rather than relying on external suppliers
Novo Nordisk's investment of over $10 billion in manufacturing expansion across Denmark, the U.S., and France reflects the scale of the capacity challenge. The company has acknowledged that demand for semaglutide consistently outpaces manufacturing capacity, and bringing new facilities online takes 3-5 years from announcement to full production.
Quality and Characterization
As manufacturing scales up, analytical methods must keep pace. Advanced characterization techniques including mass spectrometry imaging, hydrogen-deuterium exchange mass spectrometry (HDX-MS), and high-resolution chromatographic methods are enabling tighter quality control. The goal is complete characterization of peptide identity, purity, post-translational modifications, and higher-order structure at commercial scale.
Real-time process analytical technology (PAT) is being integrated into continuous-flow synthesis platforms, enabling in-line monitoring of reaction progress, purity, and yield. This shifts quality control from post-production testing to continuous in-process monitoring, reducing batch failure rates and improving overall manufacturing efficiency.
The regulatory expectations for peptide characterization continue to increase. ICH Q6B guidelines require comprehensive physicochemical characterization, and regulatory agencies are requesting increasingly detailed impurity profiles, stability studies under varied conditions, and demonstration of batch-to-batch consistency at commercial scale. Meeting these standards while scaling production to meet demand for blockbuster peptide drugs is one of the defining operational challenges of the industry.
Personalized Peptide Medicine
The future of peptide medicine isn't one-size-fits-all. Advances in genomics, pharmacogenomics, and rapid peptide synthesis are enabling treatment approaches tailored to individual patients.
Pharmacogenomics and GLP-1 Response
Not all patients respond equally to GLP-1 receptor agonists. Emerging pharmacogenomic data suggests that genetic variants in the GLP-1 receptor gene (GLP1R), as well as variants affecting drug metabolism, adipose tissue biology, and central appetite regulation, influence individual weight loss outcomes. As this field matures, genetic testing may guide selection between GLP-1 agonists, dual agonists, and triple agonists for individual patients.
Personalized Neoantigen Vaccines
As discussed in the peptide vaccines section, neoantigen-based cancer vaccines represent the most advanced form of personalized peptide medicine. Each vaccine is manufactured specifically for one patient based on their tumor's unique mutation profile. The 48-55 day manufacturing timeline, while much improved, still limits applicability. Future advances in automated peptide synthesis and AI-driven neoantigen prediction will continue to reduce this timeline.
Individualized Peptide Combinations
The growing pharmacopeia of peptide therapeutics enables individualized combination protocols. A patient with obesity and MASH might benefit from a GLP-1/glucagon dual agonist like survodutide, while a patient with obesity and sarcopenia might be better served by semaglutide plus bimagrumab. A patient with obesity and cardiovascular risk might receive semaglutide specifically for its proven cardiovascular benefit. As more peptide drugs reach the market, the opportunity for personalized combination strategies grows exponentially.
Peptide Biomarker-Guided Therapy
Circulating peptide biomarkers are increasingly used to guide treatment decisions:
- GLP-1 and GIP levels - Baseline endogenous incretin levels may predict response to incretin-based therapies
- Pro-peptide fragments - Circulating pro-BNP, pro-enkephalin, and other peptide fragments guide cardiovascular and renal treatment decisions
- Tumor-derived peptides - Liquid biopsy approaches can detect tumor-specific peptide fragments in blood, enabling non-invasive monitoring of cancer treatment response
Microbiome-Guided Peptide Selection
An emerging area of personalized medicine involves using gut microbiome analysis to guide peptide therapy selection. The gut microbiome influences peptide drug metabolism, GLP-1 receptor sensitivity, and overall metabolic phenotype. Patients with certain microbiome compositions may respond better to specific GLP-1 agonists or multi-agonists.
Several research groups are correlating baseline microbiome profiles with weight loss outcomes on different GLP-1 agonists. If consistent patterns emerge, microbiome testing could become a standard part of pre-treatment evaluation for obesity pharmacotherapy. The microbiome's role in modulating the efficacy and side effects of orally delivered peptides is particularly relevant, since gut bacteria can directly metabolize peptide drugs before absorption.
Wearable Peptide Delivery Systems
The intersection of wearable technology with peptide delivery is creating opportunities for truly personalized, responsive therapy. Closed-loop insulin delivery systems (artificial pancreas devices) already demonstrate this principle - a continuous glucose monitor detects blood sugar levels and automatically adjusts insulin delivery via a wearable pump.
Future wearable peptide delivery systems could extend this concept to other peptide drugs. A wearable patch that monitors metabolic biomarkers (glucose, ketones, inflammatory markers) in interstitial fluid and automatically adjusts GLP-1 agonist or multi-agonist delivery in real time could optimize both efficacy and tolerability. Rather than fixed weekly dosing, patients would receive precisely calibrated peptide exposure matched to their moment-to-moment metabolic needs.
The technology components for such systems mostly exist: continuous metabolic monitoring (CGM-derived), microfluidic peptide delivery pumps (adapted from insulin pumps), and AI-driven dosing algorithms (already in use for insulin). The regulatory pathway and clinical validation requirements, however, are substantial - and commercial availability of integrated metabolic peptide delivery systems is likely a 2030+ prospect.
Pharmacokinetic Modeling and Digital Twins
Computer-based pharmacokinetic models that simulate how an individual patient will metabolize and respond to a specific peptide drug - sometimes called "digital twin" approaches - are being integrated into clinical decision-making. These models incorporate patient-specific parameters (body weight, body composition, kidney function, liver function, genetic variants) to predict optimal dosing for each individual.
For peptide drugs with narrow therapeutic windows or significant inter-patient variability, model-informed precision dosing could improve outcomes while reducing side effects. The FDA has shown interest in this approach through its Model-Informed Drug Development (MIDD) program, and several pharmaceutical companies are developing digital twin platforms for their peptide drug portfolios.
The Getting Started Framework
For patients and clinicians beginning to explore peptide therapies, understanding the full range of available options is the first step. Our peptide research hub provides comprehensive coverage of currently available compounds, while this report maps the therapies that will become available in the years ahead.
Pipeline by Phase: The Numbers
The peptide drug pipeline is broader and deeper than at any point in pharmaceutical history. Here's what the development landscape looks like by phase, therapeutic area, and expected approval timeline.
Peptide Drug Pipeline by Development Phase (2025)
Pipeline Distribution by Therapeutic Area
| Therapeutic Area | Phase 1 | Phase 2 | Phase 3 | NDA/BLA Filed | Total Active |
|---|---|---|---|---|---|
| Metabolic/Obesity/Diabetes | 22 | 18 | 12 | 3 | 55 |
| Oncology | 28 | 15 | 7 | 2 | 52 |
| Infectious Disease (incl. AMPs) | 12 | 8 | 4 | 1 | 25 |
| Neurology/CNS | 8 | 5 | 2 | 1 | 16 |
| Cardiovascular/Renal | 6 | 3 | 1 | 0 | 10 |
| Rare/Orphan Disease | 5 | 2 | 1 | 1 | 9 |
| Musculoskeletal | 4 | 1 | 1 | 0 | 6 |
| Total | 85 | 52 | 28 | 8 | 173 |
Key Approval Timelines
| Drug | Class | Expected Approval (US) | Notes |
|---|---|---|---|
| Orforglipron | Oral GLP-1 agonist | Q2 2026 (obesity) | Priority review; T2D submission 2026 |
| Retatrutide | Triple agonist (GIP/GLP-1/GCG) | 2027-2028 | Phase 3 completing 2026 |
| Survodutide | Dual agonist (GLP-1/GCG) | 2027-2028 | MASH + obesity Phase 3 |
| CagriSema | GLP-1 + amylin combo | 2026-2027 | Novo Nordisk Phase 3 |
| Bimagrumab + semaglutide | ActRII inhibitor + GLP-1 | 2028+ | Phase 2 ongoing |
What to Watch Through 2030
The next five years will bring several inflection points for peptide medicine:
- 2026: First oral non-peptide GLP-1 agonist approval (orforglipron). First Phase 3 readouts for triple agonists.
- 2027: Triple agonist approvals begin. Monthly dosing formulations enter Phase 3. First AI-designed peptide enters Phase 2.
- 2028: Myostatin inhibitor + GLP-1 combinations reach Phase 3. mRNA-encoded metabolic peptides enter clinical development.
- 2029: Quad agonist clinical data arrives. Personalized neoantigen vaccines complete key Phase 3 trials in melanoma.
- 2030: Monthly/quarterly GLP-1 depot formulations available. 10+ new peptide drugs approved over the five-year period.
Clinical Trials to Watch: 2025-2030
These are the clinical trials most likely to shape the future of peptide medicine. We've selected them based on the novelty of the approach, the size and rigor of the study, and the potential impact on clinical practice.
| Trial | Compound | Phase | NCT Number | Expected Readout | Why It Matters |
|---|---|---|---|---|---|
| TRIUMPH-4 | Retatrutide | 3 | NCT06022029 | 2026 | First triple agonist Phase 3 weight loss data |
| ACHIEVE (multiple) | Orforglipron | 3 | Multiple | 2025-2026 | Oral GLP-1 for obesity and T2D |
| REDEFINE (MASH) | Survodutide | 3 | NCT06417879 | 2026-2027 | GLP-1/glucagon for liver disease |
| Bimagrumab + Sema | Bimagrumab + semaglutide | 2 | NCT05616013 | 2026 | Muscle preservation during GLP-1 weight loss |
| PNeoVCA | Neoantigen vaccine + pembrolizumab | 1 | NCT05269381 | 2025-2026 | Personalized cancer vaccine + checkpoint inhibitor |
| PGN-EDO51 (DMD) | CPP-oligonucleotide conjugate | 1 | NCT06079736 | 2026 | CPP-enabled gene therapy for muscular dystrophy |
| Taldefgrobep (SMA) | Taldefgrobep alfa | 3 | NCT05337553 | 2026-2027 | Anti-myostatin for neuromuscular disease |
| SRK-439 | Anti-myostatin antibody | 1 | NCT06445075 | 2026 | Novel myostatin inhibitor for obesity |
| ASC30 Depot | Long-acting GLP-1 depot | 2 | Ascletis trials | 2026 | Monthly dosing for obesity |
| Nomlabofusp | TAT-frataxin CPP conjugate | 1 | NCT06681766 | 2026 | CPP protein delivery for Friedreich's ataxia |
How to Track These Trials
ClinicalTrials.gov (clinicaltrials.gov) provides real-time updates on enrollment status, protocol amendments, and results postings. For interpreted analysis, our peptide research hub covers major trial readouts as they occur.
Investment Landscape and Industry Dynamics
The peptide medicine market is attracting capital at unprecedented scale. Understanding the investment landscape helps contextualize the pace of innovation and the commercial forces shaping the pipeline.
Market Growth Drivers
Several structural factors are driving capital into peptide therapeutics:
- Proven commercial model: Semaglutide and tirzepatide have demonstrated that peptide drugs can generate $25+ billion in annual revenue per compound
- Expanding indications: GLP-1 agonists alone are being studied for obesity, T2D, MASH, cardiovascular disease, CKD, Alzheimer's, Parkinson's, addiction, and OSA - each indication representing a multi-billion-dollar opportunity
- Patent differentiation: Novel multi-agonists, oral formulations, and delivery technologies offer strong IP positions distinct from existing drugs
- Manufacturing moat: Peptide synthesis expertise and capacity create meaningful barriers to entry, protecting margins for established manufacturers
Key Industry Players
| Company | Peptide Focus | Key Pipeline Assets | Manufacturing Capacity |
|---|---|---|---|
| Eli Lilly | Metabolic peptides | Tirzepatide, Retatrutide, Orforglipron | Expanding ($6B+ investment) |
| Novo Nordisk | GLP-1 agonists | Semaglutide, CagriSema, Amycretin | Expanding ($10B+ investment) |
| Boehringer Ingelheim | Dual agonists | Survodutide | Expanding |
| Bachem | CDMO (peptide synthesis) | Manufacturing partner for multiple programs | Multi-site global capacity |
| PolyPeptide Group | CDMO (peptide synthesis) | Manufacturing partner | EU/US facilities |
| PepGen | CPP conjugates | PGN-EDO51 | Clinical scale |
| Scholar Rock | Myostatin pathway | SRK-439 | Clinical scale |
Acquisition and Licensing Activity
The competitive pressure in peptide medicine has triggered a wave of acquisitions. Eli Lilly's acquisition of Versanis Bio (bimagrumab) for its myostatin pathway inhibitor reflects the strategic importance of addressing the muscle loss problem associated with GLP-1 therapy. Similar acquisitions are likely as larger pharmaceutical companies seek to fill gaps in their metabolic disease pipelines.
CDMOs (contract development and manufacturing organizations) specializing in peptide synthesis are also attracting acquisition interest, as manufacturing capacity becomes a competitive bottleneck. Companies with established multi-kilogram peptide synthesis capabilities command premium valuations.
Risks and Considerations
The peptide medicine sector is not without risk. Late-stage clinical failures (like danuglipron's termination) can destroy significant value. Manufacturing scale-up challenges can delay product launches. Safety signals in the multi-agonist class could affect the entire category. And pricing pressure from payers, governments, and the potential entry of generic/biosimilar peptides will shape long-term margins.
Specific risk categories include:
Clinical risk: Multi-agonist programs are testing novel pharmacology with limited long-term safety data. The TRIUMPH program's undisclosed safety signals for retatrutide illustrate that triple agonism may carry risks not seen with single or dual agonists. Combination therapy programs (GLP-1 + myostatin inhibitor) add another layer of clinical uncertainty. Any serious safety event in a high-profile peptide drug could create negative sentiment across the entire class.
Competitive risk: The obesity drug market is attracting enormous investment, and multiple companies are pursuing similar targets. If three or four triple agonists reach market simultaneously, differentiation becomes challenging and pricing pressure intensifies. The entry of oral formulations could also commoditize the GLP-1 class more quickly than expected.
Regulatory risk: FDA and EMA expectations for peptide drugs continue to evolve. New requirements for cardiovascular outcome studies, cancer monitoring, or longer trial durations could delay approvals and increase development costs. The regulatory treatment of peptide compounding also remains uncertain and could affect market dynamics.
Reimbursement risk: Payer resistance to high-cost obesity medications is significant in both public and private insurance markets. Medicare's 2025 decision to cover anti-obesity medications was positive, but the long-term sustainability of covering $800-1,500/month medications for tens of millions of patients is uncertain. Payer pushback could limit market penetration regardless of clinical efficacy.
Manufacturing risk: New manufacturing facilities take 3-5 years to build and qualify. If demand for next-generation peptides outpaces capacity expansion, shortages could limit commercial potential. Conversely, overbuilding capacity based on optimistic demand projections could create financial strain if clinical programs fail or market penetration is slower than expected.
The CDMO Landscape
Contract development and manufacturing organizations (CDMOs) play a disproportionately important role in peptide drug development because most pharmaceutical companies lack in-house peptide synthesis capabilities. The major peptide CDMOs include:
| CDMO | Headquarters | Peptide Capacity | Specialization |
|---|---|---|---|
| Bachem | Switzerland | Multi-hundred kg/year | Full-service peptide manufacturing |
| PolyPeptide Group | Sweden/Global | Multi-hundred kg/year | Clinical and commercial supply |
| Thermo Fisher (Patheon) | USA/Global | Large scale | Integrated drug product/substance |
| AmbioPharm | USA | Multi-kg to commercial | Complex and modified peptides |
| Corden Pharma | Germany/Global | Multi-hundred kg/year | Peptide APIs and conjugates |
| Piramal Pharma | India | Growing capacity | Cost-effective peptide synthesis |
CDMO capacity for peptide synthesis is one of the most significant bottlenecks in the industry. Bachem, the largest pure-play peptide CDMO, has announced multiple capacity expansions but still has multi-year wait times for new commercial-scale programs. This constraint is driving pharmaceutical companies to build proprietary peptide manufacturing capabilities - Eli Lilly's $6 billion manufacturing investment is partly a response to the difficulty of securing CDMO capacity at the scale needed for blockbuster peptide drugs.
The CDMO landscape is also consolidating. Larger CDMOs are acquiring smaller specialty manufacturers to add capabilities (cyclization, conjugation, formulation) and geographic reach. This consolidation is likely to continue as the peptide drug market grows and manufacturing becomes an increasingly strategic asset.
Workforce and Talent Considerations
The rapid expansion of peptide drug development is creating significant demand for specialized talent - peptide chemists, formulation scientists, regulatory specialists with peptide experience, and manufacturing engineers with SPPS expertise. University training programs have not kept pace with industry demand, creating a talent bottleneck that may constrain the pace of pipeline expansion.
Several initiatives are addressing this gap. Major pharmaceutical companies are establishing internal peptide chemistry training programs. Universities in Switzerland (ETH Zurich, University of Bern), the Netherlands (University of Amsterdam), and the U.S. (MIT, Scripps Research) are expanding peptide science curricula. CDMOs are investing in apprenticeship and on-the-job training programs to develop manufacturing talent. And the increasing use of AI in peptide design is shifting some of the expertise requirements from wet-lab chemistry toward computational biology and data science.
The talent pipeline will be a key factor in determining which companies and which countries capture the most value from the peptide medicine expansion. Regions that invest in training peptide scientists and building manufacturing expertise today will have a durable competitive advantage in the 2030 and beyond timeframe.
Environmental Sustainability in Peptide Manufacturing
Large-scale peptide synthesis consumes significant quantities of organic solvents (DMF, NMP, DCM), coupling reagents, and other chemicals that generate substantial waste. The environmental footprint of peptide manufacturing is receiving increased attention as production volumes scale to meet demand for blockbuster peptide drugs.
Industry responses include:
- Solvent recycling - Implementing closed-loop solvent recovery systems that recapture and purify DMF and other solvents for reuse, reducing both waste and raw material costs
- Greener coupling reagents - Development of coupling reagents with lower toxicity, reduced waste generation, and better atom economy (higher fraction of reagent atoms incorporated into the product)
- Water-based synthesis - Research into peptide coupling reactions that can be performed in aqueous or mixed aqueous-organic solvent systems, dramatically reducing organic solvent consumption
- Enzymatic synthesis - Enzyme-catalyzed peptide bond formation produces minimal waste (water is the only byproduct) and operates under mild conditions. While currently limited to specific peptide sequences and linkages, enzymatic methods are improving and may eventually replace chemical coupling for some applications
- Continuous manufacturing - Continuous-flow processes inherently generate less waste per kilogram of product than batch processes, due to better mixing, temperature control, and reaction efficiency
The pharmaceutical industry's broader commitment to sustainability goals (including Science Based Targets for greenhouse gas reduction) is driving adoption of greener peptide manufacturing methods. Companies that can demonstrate lower environmental impact in their manufacturing processes may also gain a regulatory and commercial advantage as environmental criteria increasingly influence procurement and reimbursement decisions.
Peptide Therapies for Neurodegeneration: Alzheimer's, Parkinson's, and Beyond
Some of the most intriguing signals from the GLP-1 agonist clinical experience have been in neurodegeneration. These observations, combined with purpose-built neuropeptide drug candidates, are creating a growing pipeline of peptide-based neurological therapies.
GLP-1 Agonists and Brain Health
Large-scale observational studies and insurance claims analyses have repeatedly found that patients taking GLP-1 receptor agonists for diabetes have lower rates of Alzheimer's disease, Parkinson's disease, and other neurodegenerative conditions compared to matched controls on other diabetes medications. These associations are consistent enough that multiple prospective clinical trials are now underway.
The biological rationale is plausible. GLP-1 receptors are expressed throughout the brain, particularly in regions affected by neurodegeneration. GLP-1 agonists reduce neuroinflammation, improve cerebral glucose metabolism, protect against mitochondrial dysfunction, and reduce formation of toxic protein aggregates (amyloid-beta and alpha-synuclein) in animal models. Semaglutide crosses the blood-brain barrier at therapeutically relevant concentrations.
Novo Nordisk's EVOKE trial program is studying semaglutide 2.4 mg (the Wegovy dose) specifically for early Alzheimer's disease. The Phase 3 trials (EVOKE and EVOKE+) enrolled approximately 3,700 participants and are expected to read out in 2026. If positive, this would represent the first time a metabolic peptide drug received approval for a neurodegenerative indication.
Liraglutide, the older GLP-1 agonist, completed a Phase 2b trial (ELAD study) in Alzheimer's patients showing that 12 months of treatment reduced the decline in cerebral glucose metabolic rate by 50% compared to placebo - a potential indicator of slower neurodegeneration. Exenatide has also shown promising results in a Phase 2 trial for Parkinson's disease, with treated patients showing sustained improvement on motor function assessments even 12 months after stopping the drug.
Purpose-Built Neuropeptide Therapies
Beyond repurposing metabolic peptides, several neuropeptide drugs are in development for specific neurological conditions:
| Compound | Type | Target Condition | Phase | Mechanism |
|---|---|---|---|---|
| Semaglutide (EVOKE) | GLP-1 agonist | Early Alzheimer's | Phase 3 | Anti-inflammatory, neuroprotective |
| Exenatide (Bydureon) | GLP-1 agonist | Parkinson's disease | Phase 3 | Neuroprotection, mitochondrial support |
| Nomlabofusp | CPP-frataxin fusion | Friedreich's ataxia | Phase 1 | Frataxin protein replacement |
| Cerebrolysin | Neurotrophic peptide mixture | Stroke, TBI, dementia | Approved (non-US) | Neurotrophic factor mimicry |
| PACAP38 analogs | Pituitary adenylate cyclase-activating peptide | Cognitive disorders | Preclinical | Neuroprotection, synaptic plasticity |
| Dihexa | Angiotensin IV analog | Cognitive impairment | Preclinical | HGF/c-Met pathway activation |
Blood-Brain Barrier Challenges
The central challenge for peptide neurotherapeutics is crossing the blood-brain barrier (BBB). Most peptides are too large, too hydrophilic, and too susceptible to enzymatic degradation to cross the BBB efficiently. Several strategies are in development to address this:
- Receptor-mediated transcytosis - Engineering peptides to bind transferrin receptors or insulin receptors on brain endothelial cells, hijacking existing transport mechanisms to shuttle peptides across the BBB
- Lipid modification - Adding fatty acid chains (as in semaglutide) to increase lipophilicity and improve passive BBB penetration
- CPP conjugation - Attaching cell-penetrating peptide sequences to therapeutic peptides to enhance membrane crossing
- Nanoparticle encapsulation - Wrapping peptides in lipid or polymer nanoparticles that can cross the BBB via endocytosis or transcytosis
- Intranasal delivery - Bypassing the BBB entirely by delivering peptides through the olfactory and trigeminal nerve pathways via nasal administration
- Focused ultrasound - Temporarily and locally opening the BBB using focused ultrasound with microbubbles, allowing peptide entry in specific brain regions
Intranasal delivery is particularly promising for neuropeptides. NAD+ and peptide formulations administered intranasally have shown direct nose-to-brain transport in preclinical models, bypassing systemic exposure and first-pass metabolism. Several clinical-stage programs are exploring intranasal delivery of neuroprotective peptides.
Peptide Biomarkers for Neurodegeneration
Beyond therapeutics, peptide-based diagnostics are advancing early detection of neurodegenerative disease. Blood-based biomarkers including phosphorylated tau (p-tau) fragments, neurofilament light chain (NfL), and glial fibrillary acidic protein (GFAP) are enabling earlier diagnosis and treatment monitoring. These peptide and protein biomarkers are increasingly integrated into clinical trial designs as enrollment criteria and pharmacodynamic endpoints.
The convergence of peptide therapeutics (GLP-1 agonists, neuropeptides) with peptide-based diagnostics (blood biomarkers, PET tracers) represents a comprehensive peptide-driven approach to neurodegenerative disease - from early detection through treatment monitoring.
Peptide Therapies for Fibrosis and Organ Protection
Beyond their metabolic effects, peptides are emerging as promising therapeutic agents for fibrotic diseases affecting the liver, kidneys, lungs, and heart. The versatility of peptide-based approaches offers multiple angles of attack against these chronic, progressive conditions.
MASH and Liver Fibrosis
Metabolic dysfunction-associated steatotic liver disease (MASLD, formerly NAFLD) affects roughly 30% of the global adult population. Its progressive form, MASH (metabolic dysfunction-associated steatohepatitis), can advance to liver fibrosis, cirrhosis, and hepatocellular carcinoma. There is currently no FDA-approved therapy for MASH, making this a massive unmet clinical need.
Several peptide-based approaches are targeting MASH from different angles:
GLP-1/Glucagon dual agonists: Survodutide and pemvidutide combine GLP-1 agonism (reducing appetite and insulin resistance) with glucagon agonism (directly reducing hepatic lipid content and increasing hepatic energy expenditure). Survodutide's Phase 2 data showed histologic improvement of MASH without worsening fibrosis in 43-62% of participants - results that compare favorably to the recently approved resmetirom.
Triple agonists for MASH: Retatrutide, with its additional GIP agonism, is also being studied in MASH Phase 3 trials. The triple mechanism may provide even greater hepatic benefits through combined effects on liver fat, inflammation, and fibrosis.
FGF21 analogs: Fibroblast growth factor 21 analogs like efruxifermin (Akero Therapeutics) directly reduce hepatic fat, improve insulin sensitivity, and may have anti-fibrotic properties. While technically a protein rather than a peptide, FGF21 analogs illustrate the broader trend of targeting liver disease with biological molecules that address the underlying metabolic dysfunction.
BPC-157 and organ protection: BPC-157 (Body Protection Compound-157) is a pentadecapeptide derived from human gastric juice that has shown organ-protective effects across multiple preclinical models. Studies have demonstrated protective effects against liver damage, including alcohol-induced liver injury, medication-related hepatotoxicity, and ischemia-reperfusion injury. While clinical trial data in humans is limited, the preclinical evidence base continues to grow and several investigator-initiated studies are underway.
Kidney Disease
GLP-1 agonists have demonstrated kidney-protective effects in large cardiovascular outcome trials. The FLOW trial showed that semaglutide reduced the risk of kidney disease progression by 24% in patients with type 2 diabetes and CKD. These results were so compelling that the trial was stopped early for efficacy.
Beyond GLP-1 agonists, several peptide-based approaches target kidney fibrosis:
- Anti-fibrotic peptides - Peptides that inhibit TGF-beta signaling, the master regulator of fibrosis, are in preclinical development for CKD
- Peptide-based complement inhibitors - Complement activation drives kidney inflammation and fibrosis in many forms of CKD; peptide inhibitors of complement factors C3 and C5 are being explored
- Renal-targeted peptide conjugates - Peptides that home to kidney tubular cells can deliver anti-fibrotic or anti-inflammatory agents directly to the site of disease
Pulmonary Fibrosis
Idiopathic pulmonary fibrosis (IPF) has limited treatment options. Peptide-based approaches include:
- Pentraxin analogs - PRM-151 (Promedior/Roche), a recombinant human pentraxin-2, showed a reduction in lung function decline in Phase 2 trials for IPF
- VIP (vasoactive intestinal peptide) analogs - VIP has anti-inflammatory and anti-fibrotic properties in the lung; stabilized analogs are in preclinical development for IPF and sarcoidosis
- Antimicrobial peptides - AMPs with immunomodulatory properties may reduce the inflammatory component of pulmonary fibrosis without the infection risk of conventional immunosuppressants
Cardiac Fibrosis
Heart failure with preserved ejection fraction (HFpEF) is increasingly recognized as a disease of cardiac fibrosis and metabolic dysfunction. GLP-1 agonists have shown benefits in HFpEF trials, potentially through combined effects on weight loss, inflammation reduction, and direct cardiac effects. Natriuretic peptide analogs (BNP, ANP derivatives) are also being explored for their anti-fibrotic and cardioprotective properties beyond their established role in acute heart failure management.
The convergence of metabolic peptides (GLP-1 agonists, multi-agonists) with organ-specific anti-fibrotic peptides represents a promising multi-pronged approach to the chronic diseases of aging - conditions that share underlying mechanisms of inflammation, metabolic dysfunction, and progressive tissue fibrosis.
Peptide Safety and Pharmacovigilance: Emerging Signals and Long-Term Monitoring
As peptide drugs reach tens of millions of patients, pharmacovigilance data is revealing both the overall safety profile and specific signals that require ongoing monitoring. Understanding these safety considerations is essential for evaluating the risk-benefit profile of next-generation peptide therapies.
GLP-1 Agonist Safety: The Real-World Data
Semaglutide and tirzepatide have accumulated vast real-world safety data through millions of patient-years of exposure. The overall profile confirms what clinical trials showed: gastrointestinal side effects (nausea, vomiting, diarrhea, constipation) are the most common adverse events, occurring in 30-50% of patients but generally decreasing over time. Serious adverse events are rare.
Several specific safety signals have drawn attention:
Pancreatitis
Early concerns about GLP-1 agonists and pancreatitis risk have been largely allayed by large-scale outcome trials (SUSTAIN, SURPASS, SELECT) that showed no significant increase in acute pancreatitis rates. However, pharmacovigilance systems continue to monitor this signal, and GLP-1 agonists remain contraindicated in patients with a history of pancreatitis.
Thyroid Cancer
GLP-1 agonists carry boxed warnings about medullary thyroid carcinoma (MTC) risk based on rodent studies showing thyroid C-cell tumors. Rodents have much higher densities of GLP-1 receptors on thyroid C-cells than humans, and large-scale human epidemiological data has not confirmed an increased MTC risk. Nevertheless, GLP-1 agonists are contraindicated in patients with personal or family history of MTC or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
Gastroparesis and Bowel Obstruction
GLP-1 agonists slow gastric emptying, which is part of their mechanism of action for appetite suppression. In rare cases, this can progress to significant gastroparesis. Case reports of intestinal obstruction in patients on GLP-1 agonists have prompted FDA label updates. Surgeons have also raised concerns about aspiration risk during anesthesia in patients on GLP-1 agonists, leading to updated pre-operative guidelines recommending drug discontinuation 1-3 weeks before elective surgery.
Gallbladder Events
Rapid weight loss from any cause increases gallstone risk. GLP-1 agonists also have direct effects on gallbladder motility via GLP-1 receptors in the biliary tract. Clinical trials have shown modestly increased rates of cholelithiasis and cholecystitis in GLP-1 agonist-treated patients. The risk appears to correlate with the degree and speed of weight loss.
Psychiatric Effects
Anecdotal reports and pharmacovigilance signals have suggested possible associations between GLP-1 agonists and suicidal ideation. The EMA conducted a formal review in 2023-2024 and concluded that available evidence did not support a causal relationship. However, monitoring continues, particularly as these drugs are used in populations with higher baseline rates of depression and anxiety.
Safety Considerations for Next-Generation Peptides
As the pipeline advances, several safety-related questions need answers:
Triple agonist safety: Adding glucagon receptor agonism introduces new pharmacology. Glucagon raises blood glucose, potentially complicating use in diabetic patients. The TRIUMPH program for retatrutide revealed safety signals beyond typical GI effects, though specifics have not been fully disclosed. Hepatic effects, glucose elevations, and potential effects on body composition will need careful monitoring.
Myostatin inhibitor safety: Blocking the myostatin/activin pathway promotes muscle growth but may also affect other tissues where these signaling molecules play roles - including bone, tendon, cardiac muscle, and reproductive organs. Long-term safety data for myostatin inhibitors in chronic use is limited.
Combination therapy safety: As peptide combinations (e.g., semaglutide + bimagrumab) enter clinical testing, potential pharmacokinetic and pharmacodynamic interactions need evaluation. The additive effects of multiple peptide drugs on immune function, wound healing, and metabolic homeostasis are not well characterized.
Long-acting formulation safety: Depot formulations that release drug over weeks to months cannot be "turned off" if adverse effects develop. Unlike daily or weekly formulations where the drug clears within days of discontinuation, depot formulations commit the patient to sustained drug exposure. This makes careful dose selection and safety monitoring during development critically relevant.
Rebound Weight Gain: The Durability Question
One of the most significant challenges for all weight loss peptides is the rebound effect after discontinuation. The STEP 1 extension trial showed that patients who stopped semaglutide regained approximately two-thirds of their lost weight within one year. This suggests that obesity treatment with peptide drugs may need to be lifelong - raising the stakes for long-term safety monitoring and making long-acting formulations that improve adherence even more relevant.
Pharmacovigilance Infrastructure
The scale of GLP-1 agonist use has tested pharmacovigilance systems globally. FDA's FAERS (FDA Adverse Event Reporting System), the EU's EudraVigilance, and WHO's VigiBase are all tracking peptide drug safety signals in real time. Post-marketing requirements for ongoing cardiovascular and cancer outcome studies ensure that long-term safety data continues to accumulate.
For prescribers and patients, the key message is that currently approved peptide drugs have well-characterized safety profiles supported by large clinical trials and extensive real-world experience. Next-generation compounds will undergo the same rigorous evaluation, but some uncertainty is inherent with any new drug class. The FormBlends science section provides ongoing updates on safety data as it emerges.
Drug-Drug Interactions with Peptide Therapies
An underappreciated aspect of peptide drug safety is the potential for drug-drug interactions (DDIs). GLP-1 agonists slow gastric emptying, which can affect the absorption of co-administered oral medications. This is particularly relevant for drugs with narrow therapeutic windows such as warfarin, levothyroxine, and oral contraceptives. Patients on these medications may require closer monitoring and potential dose adjustments when starting GLP-1 agonist therapy.
The effect of delayed gastric emptying on oral medication absorption has been documented clinically. For example, the absorption of oral acetaminophen is delayed by approximately 1 hour when co-administered with semaglutide, and the peak plasma concentration is reduced by about 20%. For most medications, this delay doesn't meaningfully affect overall exposure (area under the curve), but for medications where rapid onset matters or where consistent blood levels are critical, the interaction requires clinical attention.
As oral GLP-1 agonists like orforglipron enter the market, a new category of DDIs may emerge - drug interactions at the level of intestinal absorption between the oral GLP-1 agent and other oral medications. Unlike injectable GLP-1 agonists, which primarily affect other drugs through gastric emptying delay, oral formulations may also compete for intestinal absorption pathways.
For combination peptide therapy (e.g., GLP-1 agonist + myostatin inhibitor), the potential for pharmacodynamic interactions needs careful evaluation. Both drug classes affect body composition, metabolic rate, and insulin sensitivity. The combined effects on glucose homeostasis, bone metabolism, and hormonal axes may not simply be additive and could require dedicated interaction studies before widespread combination use.
Post-Marketing Surveillance for Novel Peptide Classes
The rapid proliferation of peptide drug classes creates challenges for post-marketing surveillance. Each new mechanism of action (triple agonism, myostatin inhibition, cell-penetrating peptide delivery) introduces novel pharmacology for which there is limited long-term safety experience. Regulatory agencies are addressing this through several approaches:
- Risk Evaluation and Mitigation Strategies (REMS) - For peptide drugs with specific safety concerns, FDA may require REMS programs including restricted distribution, prescriber training, or mandatory patient registries
- Post-marketing requirements (PMRs) - FDA routinely requires sponsors to conduct long-term outcome studies after approval, particularly for new mechanisms of action. These studies typically run 5-10 years and evaluate cardiovascular events, cancer incidence, and other long-term outcomes
- Sentinel System - FDA's distributed data system analyzes claims data from over 100 million patients to detect safety signals in near-real-time. This system is actively monitoring GLP-1 agonist safety and will be applied to next-generation peptide drugs as they reach the market
- International collaboration - The Uppsala Monitoring Centre (WHO's drug safety agency) coordinates global pharmacovigilance data sharing, enabling detection of safety signals that might be missed in any single country's data
The ideal outcome of thorough pharmacovigilance is not the absence of safety signals - no drug is entirely free of adverse effects - but rather the rapid detection, characterization, and communication of risks so that prescribers and patients can make fully informed treatment decisions. The peptide medicine field's track record of transparency in reporting safety data, while imperfect, is generally favorable compared to many other drug classes.
Peptide Chemistry Advances: Building Better Molecules
The structural limitations that once constrained peptide drug development - short half-life, poor oral bioavailability, metabolic instability - are being systematically overcome through advances in peptide chemistry. These molecular engineering techniques underpin every category of next-generation peptide therapeutics.
Peptide Stapling
Stapled peptides use a hydrocarbon or other chemical bridge to lock a peptide into its bioactive conformation - typically an alpha-helical structure. This constraint provides multiple benefits simultaneously: improved target binding affinity (since the peptide doesn't need to fold upon binding), dramatically improved protease resistance (proteases struggle to cleave constrained peptides), enhanced cell membrane permeability (the staple increases overall hydrophobicity), and extended half-life.
Stapled peptides have been particularly successful in targeting intracellular protein-protein interactions (PPIs) that are inaccessible to conventional drugs. The BCL-2 family of anti-apoptotic proteins, the MDM2/p53 interaction, and the beta-catenin/TCF interaction have all been targeted by stapled peptides in preclinical and clinical studies.
ALRN-6924 (Aileron Therapeutics) was a dual MDM2/MDMX inhibitor stapled peptide that reached Phase 2 clinical trials for solid tumors and lymphomas. While the clinical program has faced challenges, it demonstrated the clinical feasibility of the stapled peptide approach and generated valuable data on pharmacokinetics, tolerability, and target engagement in humans.
Peptide Cyclization
Cyclic peptides - peptides whose N-terminus and C-terminus are connected to form a ring, or whose side chains are cross-linked - offer many of the same benefits as stapled peptides. Cyclization constrains the structure, improves metabolic stability by 10-100x compared to linear counterparts, and can enhance membrane permeability. Natural cyclic peptides like cyclosporine A demonstrate that cyclic structures can achieve oral bioavailability - cyclosporine is among the very few orally available peptide drugs.
Modern cyclization approaches include:
- Head-to-tail cyclization - Connecting the N- and C-termini to form a macrocycle
- Side-chain-to-side-chain cyclization - Using disulfide bonds, lactam bridges, or triazole linkages between amino acid side chains
- Backbone cyclization - Modifying the peptide backbone to form cyclic structures while preserving side chain functionality
- Bicyclic peptides - Double cyclization creates even more constrained structures with greater metabolic stability and often improved binding selectivity
Bicycle Therapeutics has developed a platform of constrained bicyclic peptides (Bicycles) that combine antibody-like specificity with small molecule-like pharmacokinetics. Their lead compound, BT8009, is a PDC using a bicyclic peptide targeting Nectin-4 conjugated to a cytotoxic payload, currently in Phase 2 clinical trials for solid tumors.
Unnatural Amino Acids
Incorporating amino acids that don't occur naturally in proteins gives peptide designers a vastly expanded toolbox. Key categories include:
- D-amino acids - Mirror-image versions of natural L-amino acids that are invisible to most proteases, dramatically improving stability
- N-methylated amino acids - Blocking the amide nitrogen prevents hydrogen bonding with proteases and can improve oral absorption (this is one reason cyclosporine A is orally available)
- Beta-amino acids - Amino acids with an extra methylene group in the backbone create beta-peptides or mixed alpha/beta-peptides with unique folding properties and excellent protease resistance
- Alpha,alpha-disubstituted amino acids (Aib) - Used in peptide stapling; promote helical structure and improve metabolic stability
- PEGylated amino acids - Side chains bearing polyethylene glycol chains improve solubility and extend half-life
Lipidation
Attaching fatty acid chains to peptides - the strategy that transformed liraglutide into semaglutide - remains one of the most effective approaches for extending peptide half-life. The fatty acid enables non-covalent binding to serum albumin, which provides a circulating reservoir that releases the peptide slowly. Semaglutide's C18 fatty di-acid chain enables once-weekly dosing from a peptide that would otherwise be cleared in hours.
Next-generation lipidation strategies include branched fatty acid chains, optimized linker lengths, and dual lipidation at multiple positions. These modifications can further extend half-life toward biweekly or monthly dosing while maintaining receptor binding potency.
PEGylation and Alternatives
Polyethylene glycol (PEG) conjugation increases peptide hydrodynamic size (reducing renal clearance), shields against proteases, and reduces immunogenicity. However, anti-PEG antibodies have been detected in a significant fraction of the population, potentially reducing the efficacy of PEGylated drugs upon repeated administration.
Alternative polymer conjugation strategies under development include:
- HESylation - Conjugation with hydroxyethyl starch, a natural polymer with no known pre-existing antibodies
- PASylation - Genetic fusion with proline-alanine-serine (PAS) repeat sequences that form an inert, hydrophilic shield
- XTENylation - Fusion with XTEN polypeptide sequences that provide albumin-like half-life extension
- Fc fusion - Fusing peptides to the Fc region of IgG antibodies to leverage FcRn-mediated recycling for extended half-life
Peptide Macrocycles and Beyond
Macrocyclic peptides - larger ring structures typically containing 8-20 amino acids - are attracting increasing attention as a drug class that bridges the gap between traditional small molecules and biologics. They can bind large, flat protein surfaces (like protein-protein interaction interfaces) with high affinity while maintaining sufficient metabolic stability for clinical use.
DNA-encoded libraries (DELs) and mRNA display technologies enable screening of libraries containing billions of macrocyclic peptide structures against therapeutic targets. This combinatorial approach, coupled with AI-driven hit optimization, is accelerating the identification of macrocyclic peptide drug candidates at unprecedented speed.
The Chemistry-to-Clinic Translation
Each of these chemical modifications represents a tool that can be combined and optimized for specific therapeutic applications. A next-generation GLP-1 agonist might combine lipidation (for half-life extension), unnatural amino acids (for protease resistance), and cyclization (for oral bioavailability). The modular nature of peptide chemistry means that improvements in any one area benefit the entire field.
Peptides in Longevity and Anti-Aging Medicine
Peptides are finding growing applications in longevity research, from telomere-modulating compounds to senolytics and metabolic optimization. While much of this field remains early-stage, the convergence of aging biology with peptide pharmacology is producing intriguing results.
Epithalon and Telomere Biology
Epithalon (Epitalon) is a synthetic tetrapeptide (Ala-Glu-Asp-Gly) studied by the Khavinson laboratory in Russia for its effects on telomerase activation. The theory is straightforward: telomere shortening contributes to cellular senescence and aging; if you can maintain telomere length through telomerase activation, you might slow aspects of biological aging.
Preclinical studies in aged rodent models have shown that epithalon treatment increased telomere length in fibroblasts, reduced markers of cellular senescence, and extended lifespan in some experiments. Human studies have been limited primarily to small Russian clinical trials showing improvements in melatonin secretion, immune function, and various biomarkers of aging in elderly patients.
The limitations of the current evidence base are real - most studies are small, not randomized, and published in regional journals. However, the underlying biology of telomerase activation is well-established, and the peptide's simple structure and clean safety profile in existing studies make it a candidate for more rigorous investigation.
GHK-Cu and Tissue Renewal
GHK-Cu (glycyl-L-histidyl-L-lysine copper complex) is a naturally occurring tripeptide-copper complex that declines with age. It was initially discovered for its wound healing properties but has since been found to modulate expression of over 4,000 genes, many of which are involved in tissue repair, antioxidant defense, anti-inflammation, and anti-fibrotic processes.
In skin aging applications, GHK-Cu has demonstrated ability to increase collagen production, improve skin elasticity, reduce fine lines, and enhance wound healing. Systemically, preclinical data suggests anti-fibrotic effects in liver and lung models, neuroprotective properties, and modulation of immune function.
The peptide's gene-modulatory effects are particularly interesting from a longevity perspective. Gene expression analysis shows that GHK-Cu shifts transcriptomic patterns in aged tissues toward a more youthful profile, suppressing genes associated with tissue destruction and chronic inflammation while activating genes involved in tissue repair and regeneration.
NAD+ Peptide Precursors and Cellular Energy
While NAD+ itself is a dinucleotide rather than a peptide, its integration into peptide-based longevity protocols is widespread. NAD+ levels decline with age, and restoring them through IV infusions, subcutaneous injections, or oral precursors (NMN, NR) supports sirtuin activity, mitochondrial function, and DNA repair - three pillars of longevity biology.
The intersection with peptide medicine occurs in several ways. First, GLP-1 agonists have been shown to increase NAD+ biosynthesis in animal models, potentially contributing to their metabolic and neuroprotective effects. Second, peptide carriers are being developed to improve oral delivery of NAD+ precursors. Third, combination protocols pairing NAD+ supplementation with peptide therapies (BPC-157, GHK-Cu, epithalon) are widely used in longevity medicine clinics, though controlled clinical trial data for these combinations is sparse.
Senolytic Peptides
Senescent cells - cells that have stopped dividing but resist programmed cell death - accumulate with age and secrete pro-inflammatory molecules (the senescence-associated secretory phenotype, or SASP) that damage surrounding tissues. Selectively eliminating senescent cells (senolysis) has extended healthspan and lifespan in animal models.
Peptide-based senolytic approaches include:
- FOXO4-DRI - A D-retro-inverso peptide that disrupts the FOXO4-p53 interaction in senescent cells, releasing p53 to trigger apoptosis specifically in senescent cells while sparing normal cells. Preclinical studies showed reduced frailty, improved organ function, and enhanced fur regrowth in aged mice.
- ABT-263 (navitoclax) peptide analogs - BCL-2 family inhibitor peptides that target the anti-apoptotic pathways keeping senescent cells alive. Peptide versions may offer improved selectivity compared to the small molecule parent compound.
- Targeted senolytic peptide conjugates - Peptides that home to senescent cell surface markers (e.g., beta-galactosidase-responsive pro-peptides) coupled with cytotoxic payloads for selective senescent cell elimination.
Growth Hormone Secretagogues
Peptide growth hormone secretagogues (GHS) - including GHRP-2, GHRP-6, ipamorelin, CJC-1295, and tesamorelin - stimulate endogenous growth hormone release rather than directly administering exogenous GH. This approach is used in longevity medicine to support lean mass maintenance, fat metabolism, sleep quality, skin health, and recovery from exercise.
Tesamorelin (Egrifta) is the only GHS with FDA approval, indicated for HIV-associated lipodystrophy. It's a synthetic analog of growth hormone-releasing hormone (GHRH) that stimulates physiological pulsatile GH release. Off-label use in longevity medicine is common but not well-supported by randomized controlled trial data in non-HIV populations.
The next generation of growth hormone secretagogues aims to provide more targeted GH release patterns, oral bioavailability, and reduced tachyphylaxis (loss of response with repeated dosing). Several companies are developing small molecule ghrelin receptor agonists and modified GHRH analogs with improved pharmacokinetic profiles.
Evidence Standards in Longevity Medicine
Many peptides used in longevity medicine have strong preclinical data but limited human clinical trial evidence. Epithalon, GHK-Cu, FOXO4-DRI, and most growth hormone secretagogues (beyond tesamorelin) lack the large-scale randomized controlled trials that support GLP-1 agonist use. Consumers and clinicians should weigh the available evidence carefully and recognize that preclinical promise does not guarantee clinical benefit. For current evidence ratings on specific compounds, see our peptide research hub.
Global Regulatory and Access Landscape
The peptide medicine revolution is unfolding unevenly across the globe. Regulatory frameworks, pricing structures, manufacturing capacity, and healthcare system characteristics all shape how quickly next-generation peptide therapies reach patients in different markets.
United States
The U.S. remains the largest market for peptide therapeutics, accounting for approximately 60% of global revenue. The FDA's regulatory framework is among the most experienced in evaluating peptide drugs, and the agency has shown willingness to use expedited pathways (Priority Review, Breakthrough Therapy, Accelerated Approval) for peptide candidates addressing serious conditions.
However, access remains a major challenge. The cost of branded GLP-1 agonists ($800-1,500/month) limits uptake, and insurance coverage varies widely. Many commercial insurers impose prior authorization requirements, step therapy protocols, and BMI thresholds that restrict access. Medicare's coverage of anti-obesity medications expanded in late 2025, potentially broadening access for millions of older Americans.
The compounding pharmacy sector has played a significant role in U.S. peptide access. During the semaglutide and tirzepatide shortage period (2023-2025), compounding pharmacies produced significant volumes of these peptides at lower cost. FDA oversight of peptide compounding has intensified, with new guidance on quality standards, sterility testing, and the conditions under which compounding is permitted.
European Union
The EMA evaluates peptide drugs through centralized procedures that enable simultaneous approval across all EU member states. European peptide drug pricing is generally 30-60% lower than U.S. prices, reflecting the purchasing power of national healthcare systems and HTA (health technology assessment) bodies.
HTA evaluations in the EU tend to be more stringent regarding cost-effectiveness. NICE (UK), IQWiG (Germany), and HAS (France) have all scrutinized the cost-effectiveness of GLP-1 agonists, sometimes restricting reimbursement to specific patient populations. The arrival of cheaper oral alternatives like orforglipron may shift these analyses favorably.
China
China represents the fastest-growing market for peptide therapeutics. Several Chinese pharmaceutical companies are developing GLP-1 agonists and multi-agonists for the Chinese market, including Innovent Biologics (mazdutide, a GLP-1/glucagon dual agonist developed in partnership with Lilly) and Sciwind Biosciences. China's NMPA (National Medical Products Administration) has approved several peptide drugs for local use that are not yet available in Western markets.
The Chinese peptide manufacturing sector is also expanding rapidly. Companies like Hybio Pharmaceutical and Chinese Peptide Company supply active pharmaceutical ingredients to global peptide drug developers, and Chinese CDMO capacity for peptide synthesis is growing at roughly 25% annually.
Emerging Markets
Access to advanced peptide therapeutics in low- and middle-income countries (LMICs) remains highly constrained. The high cost of branded peptide drugs, limited cold chain infrastructure for injectable peptides, and sparse endocrinology specialist coverage all create barriers. Oral peptide formulations (like orforglipron) could significantly improve access in these markets by eliminating cold chain requirements and enabling distribution through primary care settings.
The WHO has identified GLP-1 agonists as potentially essential medicines for diabetes management, which could accelerate generic development and access programs in LMICs. India, Brazil, and several Middle Eastern countries are developing domestic peptide manufacturing capacity to reduce dependence on imports.
Intellectual Property and Generic Competition
Patent expiration timelines will shape peptide drug access over the next decade. Semaglutide's core patents begin expiring in the late 2020s, potentially opening the door to generic and biosimilar competition. Liraglutide's patents have already expired in many markets, though generic uptake has been slower than expected due to the complexity of peptide drug manufacturing and the regulatory pathway for peptide biosimilars.
The FDA's approach to peptide generics/biosimilars remains in flux. Peptides under 40 amino acids can pursue the generic (ANDA) pathway, which is faster and cheaper than the biosimilar (BLA) pathway but still requires demonstrating pharmaceutical equivalence. For longer peptides, the biosimilar pathway applies, requiring more extensive analytical, preclinical, and clinical comparability studies.
Convergence and Future Outlook: Where Peptide Medicine Is Heading
The individual advances described in this report - oral delivery, multi-agonism, AI design, targeted delivery, mRNA encoding, long-acting formulations - are powerful on their own. But their real transformative potential lies in convergence: combining multiple technologies to create peptide therapies that were inconceivable even five years ago.
Technology Convergence Scenarios
Consider what becomes possible when these technologies are combined:
Oral triple agonist with monthly dosing: An orally available small molecule that activates GLP-1, GIP, and glucagon receptors, formulated in a sustained-release oral depot that maintains therapeutic levels for 4 weeks. This would combine the efficacy of retatrutide with the convenience of a monthly pill. Individual components of this vision (oral GLP-1 agonists, multi-agonism, extended-release oral formulations) already exist - the challenge is integration.
AI-designed personalized peptide therapy: A patient provides a blood sample. AI analyzes their metabolic profile, pharmacogenomic data, and disease characteristics. Within weeks, a personalized peptide or peptide combination is designed computationally, synthesized via automated SPPS, and administered via microneedle patch. The entire workflow from sample to treatment takes less than a month.
mRNA-encoded peptide vaccines with checkpoint inhibitors: A cancer patient's tumor is sequenced. AI predicts optimal neoantigen peptide targets. Instead of synthesizing individual peptides, an mRNA construct encoding multiple neoantigens is delivered via LNP. The patient's own cells produce the vaccine antigens, combined with mRNA-encoded immune costimulatory molecules, in a single injection. Manufacturing time drops from 55 days to under 2 weeks.
Smart delivery systems for organ-specific peptide therapy: A patient with MASH receives an injection of liver-targeted LNPs containing a GLP-1/glucagon dual agonist peptide. The LNPs are decorated with hepatocyte-targeting ligands, concentrating the peptide effect in the liver while minimizing systemic GI side effects. Simultaneously, a separate muscle-targeted formulation delivers a myostatin inhibitor peptide specifically to skeletal muscle.
The 2030 Peptide Medicine Landscape
Based on current pipeline trajectories and technology development, here's what the peptide medicine landscape may look like by 2030:
| Feature | 2025 (Current) | 2030 (Projected) |
|---|---|---|
| Best weight loss efficacy | ~22% (tirzepatide) | ~30-35% (quad agonists, combinations) |
| Dosing frequency | Weekly injection | Monthly injection or daily pill |
| Oral GLP-1 options | Oral semaglutide (limited) | 3-4 oral GLP-1 agents |
| Muscle preservation | None (exercise only) | Myostatin inhibitor combinations |
| Cancer vaccines | Phase 1/2 only | First approvals possible |
| AI-designed drugs | Preclinical validation | Phase 2/3 candidates |
| AMPs approved | None (systemic) | 1-2 topical/local approvals |
| Market size | $141B | $260B+ |
| Approved peptide drugs | ~80 | ~95-100 |
Unresolved Questions
Several fundamental questions will shape the trajectory of peptide medicine over the next five years:
- Weight maintenance after discontinuation: Can any peptide drug produce durable weight loss that persists after stopping treatment? Or is lifelong therapy required? This question has profound implications for healthcare costs, patient autonomy, and long-term safety requirements.
- Optimal body composition: What's the ideal balance between fat loss and muscle preservation? Will combined GLP-1 + myostatin inhibitor therapy produce better long-term health outcomes than weight loss alone?
- Neurodegeneration prevention: Will GLP-1 agonists prove effective for Alzheimer's or Parkinson's disease prevention? The EVOKE trial results in 2026 could reshape our understanding of these diseases.
- Cancer vaccine efficacy: Will personalized neoantigen peptide vaccines show survival benefits in large randomized trials? The answer will determine whether peptide vaccines become standard cancer care or remain a niche approach.
- Manufacturing capacity: Can the industry build enough manufacturing capacity to meet demand? The current shortage of GLP-1 agonists suggests that capacity expansion may lag behind clinical success.
- Pricing and access: Will oral formulations and manufacturing scale drive peptide drug costs down enough for global access? Or will pricing remain a barrier for billions of potential patients?
- Safety of chronic multi-target engagement: What are the long-term consequences of simultaneously activating 3-4 metabolic hormone receptors for years or decades? We simply don't have that data yet.
The Bottom Line
Peptide medicine stands at an inflection point. The foundational science is strong. The commercial incentives are aligned. The manufacturing capacity is expanding. The regulatory pathways are clear. And the unmet medical need - from obesity and diabetes to cancer, neurodegeneration, and antimicrobial resistance - is enormous.
The next five years will determine which of the many promising approaches in this report fulfill their potential and which fall short. What's certain is that peptide drugs will play a central and expanding role in medicine through 2030 and beyond. For researchers, clinicians, patients, and investors, staying informed about this rapidly evolving field isn't optional - it's essential.
Impact on Healthcare Systems
The projected expansion of peptide medicine raises significant questions for healthcare systems worldwide. If even a fraction of the 650 million adults globally with obesity start using peptide-based weight loss therapies, the pharmaceutical expenditure would be staggering. At current prices, treating just 10% of eligible U.S. adults with GLP-1 agonists would cost roughly $200 billion annually - more than the entire current Medicare drug budget.
Several forces may mitigate this cost pressure. First, oral formulations and manufacturing scale should reduce per-patient costs over time. Second, generic and biosimilar competition will emerge as patents expire, potentially reducing prices by 30-80% as has occurred with other biologic classes. Third, if peptide drugs prevent downstream healthcare costs (cardiovascular events, diabetes complications, liver disease progression, joint replacements), the net economic impact may be favorable even at high drug prices.
Healthcare systems are adapting in different ways. The U.S. is slowly expanding insurance coverage for anti-obesity medications. The UK is piloting GLP-1 agonist use in primary care rather than specialist settings. Several European countries are implementing value-based pricing agreements tied to clinical outcomes. And emerging markets are exploring tiered pricing strategies that balance access with sustainability.
The most transformative scenario is one where multiple competing oral GLP-1 agents drive prices down significantly, while manufacturing capacity scales to meet demand. In this scenario, peptide-based obesity therapy could become accessible to a significant fraction of the global population - with profound implications for cardiovascular disease burden, diabetes prevalence, and overall population health. Whether this scenario materializes depends on clinical, regulatory, manufacturing, and economic factors that will play out over the next five years.
Ethical Considerations
The peptide medicine revolution raises ethical questions that the field is only beginning to address. Should obesity be treated primarily with drugs, or should the focus be on addressing environmental and social determinants of weight gain? What are the implications of lifelong medication dependence for weight maintenance? How should access be prioritized when supply is limited - by BMI severity, comorbidity burden, or ability to pay?
The enhancement vs. treatment distinction is also relevant. Some peptide drugs being developed for longevity (epithalon, senolytics), cognitive enhancement (neuropeptides), or body composition optimization (growth hormone secretagogues) blur the line between treating disease and enhancing normal function. Insurance coverage, regulatory oversight, and clinical practice standards will need to evolve as peptide drugs are used for an increasingly broad range of indications beyond traditional disease treatment.
Personalized neoantigen cancer vaccines raise their own ethical considerations around genomic privacy, the cost of truly individualized therapy, and equitable access to treatment that requires expensive sequencing and rapid custom manufacturing. As these therapies mature, ensuring that they benefit diverse patient populations - not just those with access to academic medical centers - will be an important priority.
The Research Pipeline: What We Don't Yet Know
For all the progress documented in this report, enormous gaps in our knowledge remain. Some of the most important unanswered questions include:
- What are the 10-year and 20-year safety outcomes for chronic GLP-1 agonist use? No study has followed patients on these drugs for more than 3-4 years.
- Do multi-agonists produce qualitatively different weight loss (more fat, less muscle) compared to single agonists? Body composition data from large trials is limited.
- Can peptide drugs prevent the development of obesity in at-risk populations, or are they only effective for treating established obesity?
- What happens to patients who stop peptide therapy after many years of use? Is rebound weight gain worse with longer treatment duration?
- Are AI-designed peptides fundamentally safer or more effective than traditionally designed ones, or is AI primarily accelerating the same discovery process?
- Can antimicrobial peptides truly overcome the economic barriers to antibiotic development, or will they remain a scientifically promising but commercially unviable drug class?
- Will personalized neoantigen vaccines produce survival benefits in large randomized trials, or will the responses seen in small Phase 1 studies not scale?
- How will the interaction between peptide drugs and the gut microbiome affect long-term efficacy and safety?
These questions will be answered by the clinical trials, post-marketing studies, and basic science investigations of the next five years. The answers will shape not just peptide medicine but the broader trajectory of pharmaceutical development and healthcare delivery worldwide.
For ongoing coverage of peptide medicine advances, explore our complete peptide research library or begin your research with our getting started guide.
Frequently Asked Questions
References
- Wharton S, et al. Orforglipron, an oral small-molecule GLP-1 receptor agonist for obesity treatment. N Engl J Med. 2025. doi:10.1056/NEJMoa2511774
- Jastreboff AM, et al. Triple-hormone-receptor agonist retatrutide for obesity - a phase 2 trial. N Engl J Med. 2023;389(6):514-526. doi:10.1056/NEJMoa2301972
- Armstrong J, et al. Peptide-drug conjugates: a new hope for cancer. J Pept Sci. 2025;31:e70040. doi:10.1002/psc.70040
- Zhou Y, et al. Current progress and remaining challenges of peptide-drug conjugates: next generation of ADCs? J Nanobiotechnol. 2025;23:123. doi:10.1186/s12951-025-03277-2
- Fernandez-Diaz A, et al. Artificial intelligence in peptide-based drug design. Drug Discov Today. 2025;30(2):104270. doi:10.1016/j.drudis.2025.104270
- Wong F, et al. A generative artificial intelligence approach for the discovery of antimicrobial peptides against multidrug-resistant bacteria. Nat Microbiol. 2025. doi:10.1038/s41564-025-02114-4
- Li M, et al. Trends in the research and development of peptide drug conjugates: artificial intelligence aided design. Front Pharmacol. 2025;16:1553853. doi:10.3389/fphar.2025.1553853
- Ma H, et al. Safety and efficacy of oral small-molecule GLP-1 receptor agonists orforglipron and danuglipron: systematic review and meta-analysis. Eur J Endocrinol. 2024;190(1):S1-S12. doi:10.1093/ejendo/lvad151
- Singh AK, et al. Triple agonism based therapies for obesity. Diabetes Metab Syndr. 2025;19(4):103150. doi:10.1016/j.dsx.2025.103150
- Heymsfield SB, et al. Retatrutide for the treatment of obesity, obstructive sleep apnea and knee osteoarthritis: design of the TRIUMPH trials. Obesity. 2025. doi:10.1002/oby.24217
- Iamukova A, et al. Personalized cancer vaccines in the clinical trial pipeline. Asia Pac J Clin Oncol. 2025;21:e70006. doi:10.1111/ajco.70006
- Chen L, et al. Long-acting hydrogel-based depot formulations of tirzepatide and semaglutide for management of type 2 diabetes and weight. bioRxiv. 2025. doi:10.1101/2025.07.02.662867
- Webber MJ, et al. In situ forming, enzyme-responsive peptoid-peptide hydrogels: an advanced long-acting injectable drug delivery system. J Am Chem Soc. 2024;146(35):24200-24210. doi:10.1021/jacs.4c03751
- Gupta R, et al. Therapeutic applications and challenges in myostatin inhibition for enhanced skeletal muscle mass. Mol Cell Biochem. 2025;480:1847-1862. doi:10.1007/s11010-024-05120-y
- Wei N, et al. Bimagrumab: novel medical therapy for inclusion body myositis, sarcopenia, and medication-induced lean body mass loss. Cardiol Rev. 2025. doi:10.1097/CRD.0000000000000854
- Marwaha M, et al. Emerging role of myostatin inhibitors in the management of GLP-1-associated sarcopenia and metabolic disorders. Front Endocrinol. 2025;16:1503217. doi:10.3389/fendo.2025.1503217
- Stojceski F, et al. Peptide-based drug design using generative AI. Chem Commun. 2026;62:2185-2198. doi:10.1039/D5CC04998A
- Saha D, et al. Peptide-based drug discovery through artificial intelligence. Brief Bioinform. 2024;25(4):bbae275. doi:10.1093/bib/bbae275
- Otvos L, et al. Antimicrobial peptides: from discovery to developmental applications. Appl Environ Microbiol. 2025;91(3):e02115-24. doi:10.1128/aem.02115-24
- Uddin SJ, et al. Antimicrobial peptides: natural templates for next-generation therapeutics against AMR. Front Cell Infect Microbiol. 2025;15:1720027. doi:10.3389/fcimb.2025.1720027
- Ma L, et al. APD6: the antimicrobial peptide database expanded to promote research by deploying an unprecedented information pipeline. Nucleic Acids Res. 2025. doi:10.1093/nar/gkaf534
- Xie J, et al. Applications of cell penetrating peptide-based drug delivery system in immunotherapy. Front Immunol. 2025;16:1540192. doi:10.3389/fimmu.2025.1540192
- Guidotti G, et al. The different cellular entry routes for drug delivery using cell penetrating peptides. Adv Drug Deliv Rev. 2025;210:115483. doi:10.1016/j.addr.2025.115483
- Yang J, et al. Advances and prospects of cell-penetrating peptides in tumor immunotherapy. Sci Rep. 2025;15:3485. doi:10.1038/s41598-025-86130-8
- Zhu G, et al. Advances in neoantigen-based cancer vaccines. Cancer Biol Med. 2025;22(7):1-28. doi:10.20892/j.issn.2095-3941.2025.0267
- Yang T, et al. Neoantigen cancer vaccines: a new star on the horizon. Cancer Biol Med. 2024;21(4):274-297. doi:10.20892/j.issn.2095-3941.2024.0072
- Chen Y, et al. A phase II randomized trial of individualized neoantigen peptide vaccine combined with unusual radiotherapy (iNATURE). Front Immunol. 2025;16:1538032. doi:10.3389/fimmu.2025.1538032
- Grand View Research. Peptide therapeutics market size, share & trends analysis report. 2025. Available at: grandviewresearch.com/industry-analysis/peptide-therapeutics-market
- Eli Lilly and Company. Lilly's oral GLP-1, orforglipron, demonstrated statistically significant efficacy results. Press release, September 2025. Available at: investor.lilly.com
- Eli Lilly and Company. Lilly's triple agonist retatrutide delivered weight loss of up to an average of 71.2 lbs. Press release, 2025. Available at: investor.lilly.com
- Pfizer Inc. Pfizer provides update on oral GLP-1 receptor agonist danuglipron. Press release, 2024. Available at: pfizer.com
- Ascletis Pharma. Positive topline results from U.S. Phase II for ultra-long-acting subcutaneous depot ASC30. Press release, 2025. Available at: prnewswire.com
- Qin W, et al. Progress and prospects of mRNA-based drugs in pre-clinical and clinical applications. Signal Transduct Target Ther. 2024;9:387. doi:10.1038/s41392-024-02002-z
- Xiao Y, et al. mRNA therapeutics: transforming medicine through innovation in design, delivery, and disease treatment. Mol Ther Nucleic Acids. 2025;36(3):102456. doi:10.1016/j.omtn.2025.102456
- Garcia-Sanz P, et al. Recent advances in peptide-drug conjugates as anticancer agents. Eur J Med Chem. 2025;285:117212. doi:10.1016/j.ejmech.2024.117212
- Zhang K, et al. Peptide-drug conjugates: design, chemistry, and drug delivery system as a novel cancer theranostic. ACS Pharmacol Transl Sci. 2024;7(2):274-294. doi:10.1021/acsptsci.3c00269
- Sridharan R, et al. Peptides: potential delivery systems for mRNA. Int J Pharm. 2025;670:125127. doi:10.1016/j.ijpharm.2025.125127
- Torres MDT, et al. Discovery of antimicrobial peptides in the global microbiome with machine learning. Cell. 2024;187(14):3761-3778. doi:10.1016/j.cell.2024.05.013
- Zhang L, et al. Antimicrobial peptide biological activity, delivery systems and clinical translation status and challenges. J Transl Med. 2025;23:258. doi:10.1186/s12967-025-06321-9
- Wagner E, et al. The clinical development of taldefgrobep alfa: an anti-myostatin adnectin for the treatment of DMD. Neurol Ther. 2024;13:93-113. doi:10.1007/s40120-023-00570-w