Executive Summary
Women's biology demands a different lens when it comes to peptide therapy. From fluctuating sex hormones across the menstrual cycle to the seismic hormonal shifts of menopause, female physiology shapes how peptides are absorbed, distributed, metabolized, and ultimately how well they work. This guide brings together the clinical evidence, safety data, and practical protocols that matter most for women exploring peptide-based therapies.
Key Takeaways
- Women typically achieve greater weight loss on GLP-1 receptor agonists than men at the same doses - up to 16.2% body weight loss on semaglutide versus 13.5% in men
- GLP-1 agonists must be discontinued at least 2 months before planned pregnancy due to fetal risk demonstrated in animal studies
- Kisspeptin-54 can trigger oocyte maturation in IVF cycles with zero cases of ovarian hyperstimulation syndrome in clinical trials - a safer alternative to hCG triggers
- NAD+ precursor supplementation restores oocyte quality and fertility in aged animal models, with human translation studies underway
- Women show higher blood concentrations and longer elimination times for many drugs at equal doses compared to men, supporting the case for sex-specific peptide dosing
Peptide therapy has expanded rapidly over the past decade. GLP-1 receptor agonists like semaglutide and tirzepatide are now prescribed to millions of women for weight management and metabolic health. Growth hormone secretagogues such as CJC-1295/Ipamorelin are used for body composition, sleep, and recovery. Reproductive peptides like kisspeptin and gonadorelin sit at the intersection of fertility medicine and hormonal optimization. And healing peptides such as BPC-157 and GHK-Cu offer tissue repair and skin rejuvenation benefits that women increasingly seek.
Yet most peptide dosing guidelines, clinical trial data, and published protocols were developed with male-dominant study populations. Women consistently represent the minority in Phase I pharmacokinetic studies, and sex-specific dosing adjustments are rarely provided. This creates a real knowledge gap - one that clinicians and patients must bridge with the available evidence.
Key Takeaways
- Women typically achieve greater weight loss on GLP-1 receptor agonists than men at the same doses - up to 16.2% body weight loss on semaglutide versus 13.5% in men
- GLP-1 agonists must be discontinued at least 2 months before planned pregnancy due to fetal risk demonstrated in animal studies
- Kisspeptin-54 can trigger oocyte maturation in IVF cycles with zero cases of ovarian hyperstimulation syndrome in clinical trials - a safer alternative to hCG triggers
- NAD+ precursor supplementation restores oocyte quality and fertility in aged animal models, with human translation studies underway
- Women show higher blood concentrations and longer elimination times for many drugs at equal doses compared to men, supporting the case for sex-specific peptide dosing
- Collagen peptides at 5 g daily significantly increase bone mineral density at the spine and femoral neck in postmenopausal women
This report covers the full spectrum of peptide therapy considerations for women. We'll walk through the pharmacological basis for sex-based dosing differences, examine each major peptide class through a female-specific lens, address the critical safety questions around fertility, pregnancy, and breastfeeding, and provide practical protocols adjusted for women's hormonal cycles. Whether you're a clinician designing treatment plans or a patient evaluating options, the goal is the same: evidence-based guidance that accounts for the biological realities of being female.
For those new to peptide therapy, our beginner's guide to peptide therapy provides helpful foundational context before diving into the sex-specific material covered here. You can also use our peptide dosing calculator for personalized starting dose recommendations, and browse our complete peptide research library for detailed information on individual compounds.
Why This Report Exists
The gap in sex-specific peptide research isn't a small oversight - it's a structural problem in how clinical medicine has historically operated. Until 1993, the FDA didn't even require drug trials to include women in study populations. The NIH Revitalization Act of that year mandated the inclusion of women and minorities in clinical research, but decades of male-centric data still form the backbone of most prescribing guidelines.
Peptide therapeutics entered clinical use against this backdrop. The first GLP-1 receptor agonist (exenatide) was approved in 2005, and while subsequent trials included women, sex-stratified analyses were rarely the primary endpoint. Growth hormone secretagogues, healing peptides, and longevity compounds have even less female-specific data, with most studies either using male animal models or mixed human populations without reporting results by sex.
This matters because women aren't just smaller men. The hormonal environment created by the hypothalamic-pituitary-gonadal axis, the metabolic shifts of the menstrual cycle, and the dramatic hormonal transitions of pregnancy and menopause create a fundamentally different pharmacological context. A peptide that works well for a 180-pound man may be too strong, too weak, or timed poorly for a 140-pound woman in her luteal phase - and neither the patient nor her clinician may realize why the expected response didn't materialize.
This report synthesizes the available evidence - clinical trial data, pharmacokinetic studies, mechanistic research, and clinical experience - into practical guidance for women and their healthcare providers. Where evidence is strong, we say so. Where it's weak or absent, we say that too, because knowing what we don't know is just as important as knowing what we do.
The scope is deliberately broad. We cover GLP-1 receptor agonists for weight management and PCOS, growth hormone secretagogues for body composition and anti-aging, reproductive peptides like kisspeptin and gonadorelin for fertility, healing peptides including BPC-157 and GHK-Cu for tissue repair and skin rejuvenation, longevity peptides such as epithalon and NAD+ for cellular aging, and supportive peptides like oxytocin and collagen peptides for bone health, mood, and structural integrity. For each class, we address the fundamental question: how does being female change the equation?
We also address the practical realities that women face: navigating peptide therapy around pregnancy planning, managing side effects that may vary with menstrual cycle phase, finding qualified providers, understanding costs, ensuring quality sourcing, and advocating effectively for sex-appropriate dosing. The goal isn't to create anxiety about peptide use but to empower informed decision-making.
The Female Hormonal Landscape: A Brief Orientation
Before diving into individual peptide classes, it helps to understand the major hormonal frameworks that shape women's peptide therapy experience:
The menstrual cycle (reproductive years): A roughly 28-day cycle governed by the interplay of estrogen, progesterone, FSH, and LH. The follicular phase (days 1-14) features rising estrogen and increasing metabolic activity. The luteal phase (days 15-28) is dominated by progesterone, which slows GI motility, promotes fluid retention, and can amplify side effects. This cyclical hormonal pattern means the same peptide dose can produce different subjective experiences depending on when it's taken.
Perimenopause (typically ages 45-55): A transitional period of 4-10 years where ovarian function becomes erratic. Estrogen levels can swing wildly from month to month - sometimes higher than premenopausal peaks, sometimes crashing to postmenopausal lows. This hormonal unpredictability makes peptide responses less consistent and dosing more challenging.
Postmenopause (after final menstrual period): Estrogen and progesterone settle at low, stable levels. The hormonal chaos of perimenopause gives way to predictability, but the sustained absence of estrogen accelerates bone loss, cardiovascular risk, metabolic deterioration, and tissue aging. This is often where peptide therapy becomes most attractive and, ironically, where dosing becomes most straightforward.
Pregnancy and lactation: An entirely unique hormonal state where progesterone, estrogen, hCG, placental GH, relaxin, and prolactin create a metabolic environment designed to support fetal development. Most peptide therapies are contraindicated during this period. The single most important question for any reproductive-age woman considering peptide therapy is simple: "Could I become pregnant while on this?" If the answer is anything other than a definitive "no," pregnancy-safe contraception and clear washout planning must be part of the protocol.
Each of these hormonal contexts creates different therapeutic opportunities and risks. A peptide protocol designed for a 30-year-old with PCOS looks completely different from one designed for a 55-year-old postmenopausal woman - not because the peptides are different, but because the hormonal environment in which they operate is fundamentally distinct. This report provides the framework for understanding and navigating those differences.
Figure 1: Major peptide therapy categories and their female-specific clinical considerations
Sex-Based Pharmacological Differences
Women and men process drugs differently at nearly every pharmacokinetic step - absorption, distribution, metabolism, and elimination. These differences aren't trivial. They directly affect peptide efficacy, side effect profiles, and optimal dosing.
Body Composition and Drug Distribution
The most fundamental pharmacological difference between the sexes comes down to body composition. Women carry a higher percentage of body fat (typically 20-25% versus 15-20% in men) and lower total body water. This directly influences how drugs distribute through the body after administration. Lipophilic compounds tend to have a larger volume of distribution in women, accumulating in adipose tissue and extending their half-lives. Hydrophilic compounds, conversely, may reach higher peak concentrations in women due to smaller distribution volumes.
For peptide therapeutics, which are generally hydrophilic molecules, this means women often achieve higher plasma concentrations at equal doses. A 2020 analysis published in Biology of Sex Differences established substantial pharmacokinetic sex differences for 86 medications, finding that women routinely generated higher blood concentrations and longer drug elimination times than men given identical doses (Zucker & Prendergast, 2020, doi:10.1186/s13293-020-00308-5).
Gastric Emptying and Absorption
Women have slower gastric emptying rates than men, influenced by hormonal fluctuations across the menstrual cycle. Progesterone, which rises in the luteal phase, further slows gastric motility. This is particularly relevant for oral peptide formulations like oral semaglutide (Rybelsus), where absorption depends on stomach conditions. Slower gastric emptying can increase the window for absorption but also increases the exposure of the peptide to gastric enzymes.
For subcutaneous peptide injections - the most common delivery route for therapeutic peptides - absorption rates are influenced by local blood flow and subcutaneous fat thickness. Women's higher subcutaneous fat percentage, particularly at common injection sites like the abdomen and thighs, can modestly slow absorption compared to leaner injection sites.
Hepatic Metabolism
Cytochrome P450 enzyme activity differs between the sexes in clinically meaningful ways. CYP3A4, the most abundant hepatic enzyme responsible for metabolizing roughly 50% of all medications, shows higher activity in women. CYP1A2, by contrast, is more active in men. CYP2D6 activity can vary with menstrual cycle phase and oral contraceptive use.
While most peptide therapeutics are cleared through renal filtration and proteolytic degradation rather than hepatic CYP metabolism, some small peptides and peptide-drug conjugates do interact with hepatic enzymes. Understanding these sex-based metabolic differences helps explain why adverse drug reaction rates are consistently higher in women across multiple drug classes - women simply achieve higher and more prolonged drug exposures at standard doses.
Renal Clearance
Women have lower glomerular filtration rates (GFR) than men, even after adjusting for body surface area. Since many peptides and their metabolites are cleared renally, this translates to slower peptide elimination in women. The clinical implication is straightforward: at equal doses, women maintain therapeutic (and potentially supratherapeutic) peptide levels longer than men.
Clinical Implication
The combination of higher peak concentrations, larger adipose distribution volumes, and slower renal clearance means women are pharmacokinetically primed for stronger peptide effects at standard doses. This can be advantageous - as seen with greater weight loss on GLP-1 agonists - but also increases the risk of adverse effects. Conservative initial dosing with slower titration is often the wisest approach for women starting peptide therapy.
Hormonal Influences on Peptide Pharmacology
The menstrual cycle creates a monthly pharmacological landscape that shifts in predictable ways. Estrogen and progesterone don't just regulate reproduction - they influence gastric motility, hepatic enzyme expression, renal blood flow, protein binding, and even receptor sensitivity. This means the same peptide dose can produce subtly different effects depending on where a woman is in her cycle.
During the follicular phase (days 1-14), rising estrogen tends to increase CYP3A4 activity and enhance hepatic blood flow, potentially increasing peptide metabolism. The luteal phase (days 15-28), dominated by progesterone, slows gastric emptying, reduces CYP3A4 activity somewhat, and can alter renal clearance. These fluctuations are modest for most peptides but can become clinically meaningful for compounds with narrow therapeutic windows or dose-dependent side effects.
| Pharmacokinetic Parameter | Women vs. Men | Clinical Impact on Peptides |
|---|---|---|
| Body fat percentage | Higher (20-25% vs 15-20%) | Larger Vd for lipophilic compounds; extended half-life |
| Total body water | Lower | Higher peak concentrations for hydrophilic peptides |
| Gastric emptying | Slower (cycle-dependent) | Variable oral peptide absorption; luteal phase slowest |
| CYP3A4 activity | Higher | Faster hepatic metabolism for CYP3A4 substrates |
| Glomerular filtration rate | Lower | Slower renal peptide clearance; prolonged effect |
| Plasma protein binding | Variable with hormonal status | Free drug fraction changes across menstrual cycle |
| Subcutaneous fat thickness | Greater at injection sites | Potentially slower SubQ absorption rate |
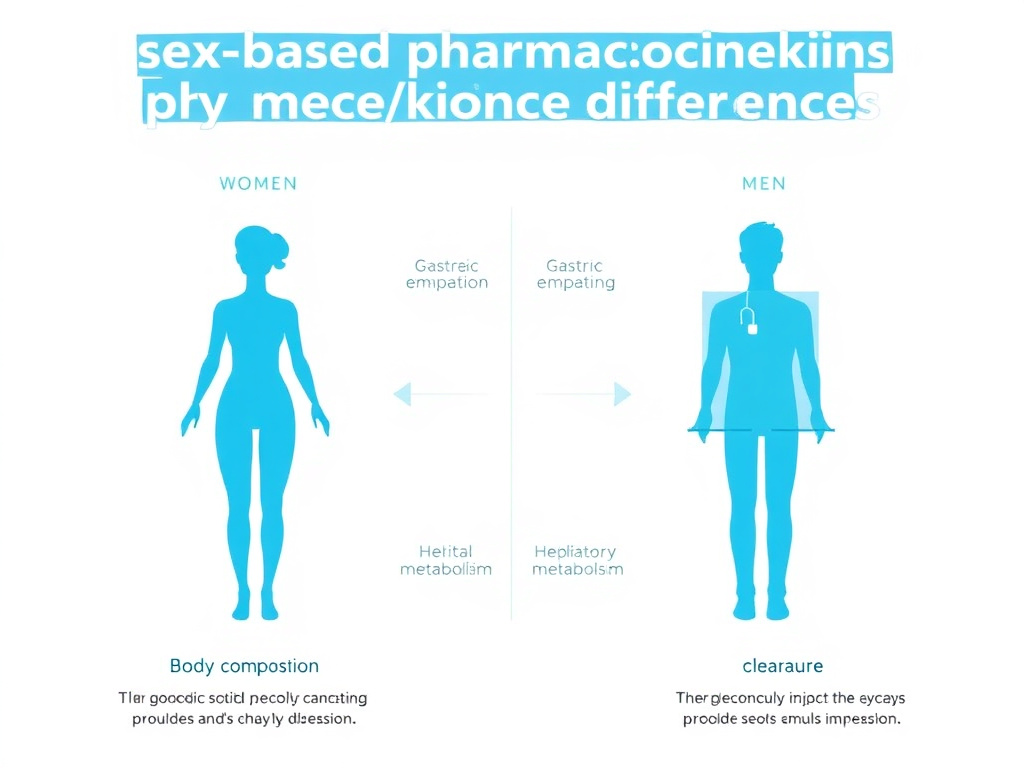
Figure 2: Sex-based pharmacokinetic differences affecting peptide therapy in women
Adverse Drug Reaction Disparities
Women experience adverse drug reactions at roughly 1.5 to 1.7 times the rate of men across all drug classes. A 2025 pharmacovigilance analysis of semaglutide specifically found that 73.35% of reported adverse reactions involved women (3,896 of 5,310 total cases). While prescribing patterns and reporting biases contribute to this disparity, the pharmacokinetic differences outlined above are a primary driver.
For peptide therapy, the most commonly reported sex-differential adverse effects include:
- Gastrointestinal effects: Women report higher rates of nausea, vomiting, and constipation on GLP-1 agonists
- Injection site reactions: More frequent and persistent in women, possibly related to differences in subcutaneous tissue composition
- Headache: More commonly reported in women across multiple peptide classes
- Mood changes: Hormonal peptides can interact with existing cyclical mood patterns
The practical takeaway for clinicians: start low, titrate slowly, and listen carefully to your female patients. The standard dose escalation timelines developed in mixed or male-dominant trial populations may be too aggressive for many women.
The Estrogen-Drug Interaction Network
Estrogen doesn't just fluctuate across the menstrual cycle - it actively modulates drug metabolism and receptor sensitivity in ways that are directly relevant to peptide therapy. Understanding this network helps explain why some women respond beautifully to a peptide protocol while others on the same regimen experience intolerable side effects.
Estrogen and GLP-1 receptor expression: Estrogen upregulates GLP-1 receptor expression in the hypothalamus, particularly in the arcuate nucleus and the nucleus of the solitary tract - brain regions critical for appetite regulation and nausea signaling. This means premenopausal women with higher estrogen levels may have more GLP-1 receptors available for the drug to act on, potentially explaining both the greater weight loss efficacy and the higher nausea rates seen in women.
Estrogen and growth hormone: Estrogen stimulates GH secretion through the hypothalamus but simultaneously inhibits the hepatic actions of GH through suppression of IGF-1 production. This paradox - more GH release but less downstream IGF-1 signaling - is most pronounced with oral estrogen (which has a hepatic first-pass effect) and is less problematic with transdermal estrogen delivery. For women on HRT who also use GH secretagogues, the route of estrogen administration directly affects how much IGF-1 the GH peptide will produce.
Estrogen and inflammation: In premenopausal women, estrogen generally exerts anti-inflammatory effects, reducing CRP, IL-6, and TNF-alpha. The loss of this anti-inflammatory protection after menopause contributes to the chronic low-grade inflammation ("inflammaging") that accelerates tissue damage and disease. Peptides with anti-inflammatory properties - GLP-1 agonists, GHK-Cu, oxytocin - may be particularly valuable in the postmenopausal period when estrogen's protective effects are absent.
Estrogen and collagen: Estrogen is a powerful stimulator of collagen synthesis. The rapid skin thinning and joint stiffness that many women notice during perimenopause and early menopause directly reflects falling estrogen and its downstream collagen effects. This creates a strong rationale for collagen-stimulating peptides (GHK-Cu, collagen peptides) during the menopausal transition, effectively replacing one collagen stimulus (estrogen) with another (peptide therapy).
Progesterone: The Often-Overlooked Factor
While estrogen receives most of the attention in sex-based pharmacology discussions, progesterone plays an equally important role in shaping peptide therapy responses:
GI motility: Progesterone is a smooth muscle relaxant that slows gastric emptying and intestinal transit. During the luteal phase (days 15-28) and during pregnancy (when progesterone levels are dramatically elevated), GI motility is at its slowest. This directly impacts oral peptide absorption and amplifies the GI side effects of GLP-1 agonists. The "double hit" of progesterone-induced slow motility plus GLP-1-induced delayed gastric emptying explains why many women report their worst nausea during the luteal phase.
Fluid retention: Progesterone promotes sodium and water retention, contributing to the bloating and weight fluctuation many women experience premenstrually. For women tracking weight loss on GLP-1 agonists, luteal phase water retention can mask fat loss on the scale, leading to frustration and potential premature dose increases. Understanding this pattern helps patients interpret their progress more accurately.
Mood modulation: Progesterone is metabolized to allopregnanolone, a potent positive modulator of GABA-A receptors. This neurosteroid has sedating, anxiolytic, and antidepressant properties. The rapid decline in allopregnanolone during the late luteal phase (premenstrually) and at menopause may contribute to mood instability. Peptides like oxytocin that also modulate mood through different pathways could theoretically complement progesterone's mood effects - or interact with them in complex ways.
Immune function: Progesterone has immunosuppressive properties that prevent maternal immune rejection of the fetus during pregnancy. In non-pregnant women, the luteal phase progesterone rise may slightly alter immune surveillance, which has theoretical implications for healing peptide efficacy and autoimmune disease activity during different cycle phases.
GLP-1 Therapy in Women
GLP-1 receptor agonists have become the most widely prescribed peptide class for women, driven by their remarkable efficacy in weight management and growing evidence of metabolic benefits. But women's experience with these medications differs from men's in several important ways - from greater weight loss to higher rates of GI side effects to unique considerations around contraception and fertility.
Weight Loss Outcomes: Women Outperform Men
One of the most consistent findings across GLP-1 clinical trials is that women tend to lose more weight than men at equivalent doses. A sex-stratified analysis of the STEP-HFpEF and STEP-HFpEF DM trials found that women on semaglutide achieved a mean body weight reduction of 9.6% compared to 7.2% in men (p = 0.006 for interaction) (Kosiborod et al., 2024, doi:10.1016/j.jacc.2024.06.001).
This pattern holds across the GLP-1 and dual-agonist class. In the highest-dose arms of landmark trials:
Weight Loss by Gender - Highest Dose Arms
The reasons for women's enhanced weight loss response are likely multifactorial. Higher baseline body fat percentage means more substrate available for mobilization. Women's higher plasma GLP-1 concentrations at equivalent doses (due to the pharmacokinetic factors discussed above) produce stronger appetite suppression. And the interaction between GLP-1 signaling and female sex hormones may amplify metabolic effects - estrogen enhances GLP-1 receptor expression in certain brain regions involved in appetite regulation.
GLP-1s and Polycystic Ovary Syndrome (PCOS)
Perhaps the most exciting female-specific application of GLP-1 therapy is in polycystic ovary syndrome. PCOS affects 8-13% of reproductive-age women and is characterized by insulin resistance, hyperandrogenism, and ovulatory dysfunction. GLP-1 agonists address multiple pathological mechanisms simultaneously.
A meta-analysis of 11 randomized controlled trials involving 840 women with PCOS found that GLP-1 receptor agonist use improved spontaneous pregnancy rates with a relative risk of 1.72 (95% CI: 1.22-2.43) (Han et al., 2023, doi:10.1186/s12902-023-01500-5). Menstrual regularity also improved significantly.
Tirzepatide, the dual GLP-1/GIP receptor agonist, has shown even more striking results in PCOS. Real-world data presented at ObesityWeek 2025 examined 4,241 women with obesity/overweight and self-reported PCOS who initiated tirzepatide. After 10 months:
- Mean weight loss: 18.81%
- 96.58% achieved at least 5% weight loss
- 90.8% achieved at least 10% weight loss
- 75.96% achieved at least 15% weight loss
Clinical Pearl - PCOS
The weight loss achieved with GLP-1 agonists in PCOS often restores ovulatory function before patients reach their goal weight. This creates an unintended pregnancy risk, especially since GLP-1 agonists can reduce oral contraceptive absorption through delayed gastric emptying. Clinicians should counsel PCOS patients about using non-oral contraception (IUD, implant, or barrier methods) while on GLP-1 therapy, and discuss pregnancy planning well before it becomes imminent.
Metabolic Benefits Beyond Weight Loss
For women specifically, GLP-1 agonists deliver metabolic benefits that extend beyond the number on the scale. These include:
Insulin sensitivity improvement: Women with PCOS show marked improvements in HOMA-IR (homeostatic model assessment for insulin resistance) on GLP-1 therapy. Since insulin resistance drives androgen excess in PCOS, this creates a cascade of benefits - lower testosterone, reduced acne and hirsutism, and restored ovulation.
Cardiovascular protection: The SELECT trial (N > 17,000) demonstrated cardiovascular event reduction with semaglutide that was consistent across sexes and all racial groups. Given that cardiovascular disease remains the leading cause of death in women, this benefit is substantial.
Liver health: Non-alcoholic fatty liver disease (NAFLD) prevalence increases sharply in postmenopausal women. Semaglutide has shown significant reduction in hepatic steatosis and fibrosis markers, with Phase III trial data supporting its use in metabolic-associated steatotic liver disease (MASLD).
GI Side Effects: The Female Experience
Women report higher rates of gastrointestinal adverse effects on GLP-1 agonists, including nausea (35-45% vs 25-35% in men), vomiting (15-20% vs 8-12%), and constipation (20-25% vs 12-18%). These differences likely reflect the combination of higher plasma drug levels and the baseline slower gastric motility seen in women.
Practical management strategies for women include:
- Starting at the lowest available dose and extending titration intervals by 1-2 additional weeks
- Timing injections to avoid the luteal phase when baseline GI motility is already reduced
- Smaller, more frequent meals rather than large portions
- Adequate hydration (at least 64 oz daily) to prevent constipation
- Ginger supplementation or prescription antiemetics for persistent nausea
Oral Contraceptive Interactions
GLP-1 agonists delay gastric emptying, which can reduce the absorption of co-administered oral medications including combined oral contraceptive pills (COCPs). While semaglutide's prescribing information indicates that the overall exposure to ethinylestradiol and levonorgestrel was not meaningfully altered, peak concentrations (Cmax) were reduced by 12-40% depending on the study.
For women relying on oral contraceptives, this pharmacokinetic interaction raises important questions about contraceptive reliability - particularly during dose titration when GLP-1 effects are most variable. The safest approach is to recommend non-oral contraceptive methods for women on GLP-1 therapy who absolutely must avoid pregnancy.
Women-Specific GLP-1 Prescribing Considerations
Beyond the pharmacological differences, several practical considerations affect how women experience GLP-1 therapy:
Body image and disordered eating: Women are disproportionately affected by eating disorders, with a lifetime prevalence roughly 2.5 times that of men. GLP-1 agonists produce profound appetite suppression that can feel liberating for women who've struggled with weight - but can also trigger or unmask disordered eating patterns. The experience of "not thinking about food" is frequently described as revelatory by patients, but clinicians should screen for pre-existing eating disorders before prescribing and monitor for maladaptive restrictive behaviors during treatment.
Lean muscle mass preservation: Women start with less lean muscle mass than men and are more susceptible to sarcopenia (age-related muscle loss), particularly after menopause. The rapid weight loss produced by GLP-1 agonists includes both fat and lean tissue. Studies suggest that 25-40% of weight lost on GLP-1 therapy is lean mass unless countermeasures are taken. For women, who have less lean mass to spare, this is particularly concerning. Resistance training and adequate protein intake (at least 1.2-1.6 g/kg/day) should be standard counseling alongside GLP-1 prescriptions.
Hair loss: Telogen effluvium - temporary diffuse hair shedding triggered by rapid weight loss, nutritional deficiency, or physiological stress - is more commonly reported in women on GLP-1 agonists. This isn't a direct drug side effect but rather a consequence of the rapid weight loss and potential caloric or protein deficit. Ensuring adequate nutrition, protein intake, and micronutrient supplementation (particularly zinc, iron, biotin, and vitamin D) can help mitigate this concern.
Gallbladder disease: Women already face 2-3 times higher risk of gallstones compared to men, and rapid weight loss further increases this risk. GLP-1 agonists have been associated with increased rates of gallbladder events including cholelithiasis and cholecystitis. Women on GLP-1 therapy should be educated about symptoms of gallbladder disease (right upper quadrant pain, especially after fatty meals) and encouraged to avoid excessively rapid weight loss by not restricting calories too severely beyond what the appetite suppression naturally produces.
Pancreatitis monitoring: While pancreatitis risk with GLP-1 agonists is low overall, the combination of GLP-1 therapy and gallstone disease (more common in women losing weight rapidly) can increase pancreatic risk. Lipase and amylase levels should be checked if abdominal pain develops.
Facial aging from rapid weight loss: The phenomenon sometimes called "Ozempic face" - facial volume loss and accelerated facial aging from rapid weight loss - can be particularly distressing for women. Fat loss from the face reveals underlying bone structure changes that are already progressing due to estrogen decline in midlife women. While not a medical complication, this aesthetic concern drives many women to discontinue therapy or seek facial fillers. Slower, more gradual weight loss (achieved by extending dose titration) can minimize this effect.
GLP-1 Therapy Across the Female Lifespan
Reproductive years (18-40): GLP-1 therapy in reproductive-age women requires constant awareness of pregnancy risk. The combination of improved fertility (from weight loss and metabolic improvement) and potentially reduced oral contraceptive efficacy (from delayed gastric emptying) demands proactive counseling. PCOS patients are the primary target population in this age group, and the metabolic benefits can be transformative - but the fertility implications must be explicitly addressed at every visit.
Perimenopause (40-55): This transition period often coincides with weight gain, particularly visceral fat accumulation driven by declining estrogen and increasing insulin resistance. GLP-1 agonists can be especially effective during perimenopause because they address the metabolic derangements (insulin resistance, hepatic steatosis, visceral adiposity) that accelerate during this transition. The variable hormonal milieu may cause fluctuating GI tolerance, requiring patience with dose titration.
Postmenopause (55+): Cardiovascular benefit becomes the dominant consideration. The SELECT trial demonstrated that semaglutide reduces major adverse cardiovascular events in overweight/obese adults, and given that cardiovascular disease is the leading cause of death in postmenopausal women, this benefit is particularly relevant. Dosing in older women should be conservative, with attention to the increased risk of sarcopenia and osteoporosis that rapid weight loss can exacerbate. Pairing GLP-1 therapy with resistance training and bone-protective peptides (collagen peptides, potentially oxytocin) creates a more complete approach.
For a broader overview of the GLP-1 drug class, visit our GLP-1 resource hub.
Figure 3: GLP-1 therapy outcomes in women compared to men across key clinical endpoints
Fertility Considerations
Fertility and peptide therapy are deeply intertwined. Some peptides directly support reproductive function, others must be carefully timed around conception attempts, and a few pose documented risks to embryonic development. Every woman of reproductive age considering peptide therapy needs to understand these connections.
Kisspeptin: The Fertility Peptide
Kisspeptin is arguably the most important peptide in human fertility. Naturally produced in the hypothalamus, kisspeptin acts on the GnRH neurons to trigger the release of gonadotropin-releasing hormone, which in turn stimulates LH and FSH - the hormones that drive ovulation. Without adequate kisspeptin signaling, ovulation simply doesn't occur.
The clinical translation of kisspeptin has been most dramatic in IVF. Traditionally, human chorionic gonadotropin (hCG) is used to trigger final oocyte maturation before egg retrieval. But hCG carries a significant risk of ovarian hyperstimulation syndrome (OHSS) - a potentially life-threatening complication that disproportionately affects women with PCOS and high responders.
Kisspeptin-54 offers a fundamentally safer approach. In clinical trials at Imperial College London, kisspeptin-54 successfully triggered oocyte maturation with remarkable safety data:
- At the optimal dose (9.6 nmol/kg), biochemical pregnancy rate was 85%, clinical pregnancy rate was 77%, and live birth rate was 62%
- Zero cases of moderate, severe, or critical OHSS across all dose groups (Abbara et al., 2015, doi:10.1210/jc.2014-3766)
- Pregnancy rates between 23% and 37% across randomized trials with mixed-risk populations
For more on kisspeptin's mechanisms and research, see our dedicated kisspeptin research page.
Kisspeptin Variants
Two kisspeptin forms are commonly discussed. Kisspeptin-54 (KP54) is the full-length form with a half-life of approximately 28 minutes. Kisspeptin-10 (KP10) is the C-terminal fragment with a shorter half-life of approximately 3 minutes. MVT-602 (TAK-448), a modified KP10 analog, exhibits a stronger and more prolonged pharmacodynamic effect (21-22 hours versus 4.7 hours for KP54), making it attractive for clinical use where sustained LH elevation is desirable.
Gonadorelin: Pulsatile GnRH Replacement
Gonadorelin is synthetic gonadotropin-releasing hormone, structurally identical to the endogenous GnRH decapeptide. Its primary fertility application is in hypothalamic amenorrhea - a condition where the brain fails to produce adequate GnRH pulses, leading to absent or irregular periods and anovulation.
When delivered via a portable infusion pump in pulsatile fashion (5-20 mcg every 90-120 minutes), gonadorelin can restore the natural LH/FSH pulsatility needed for follicular development and ovulation. This approach closely mimics physiology and avoids the multiple follicle development risk associated with exogenous gonadotropin injections. Our gonadorelin research page covers the full evidence base.
Key clinical applications in women include:
- Primary hypothalamic amenorrhea
- Functional hypothalamic amenorrhea (often related to stress, low body weight, or excessive exercise)
- Hypogonadotropic hypogonadism
- Ovulation induction in women with intact pituitary function
- Diagnostic testing of pituitary LH/FSH reserve
NAD+ and Ovarian Aging
One of the most compelling areas of female fertility research involves NAD+ and ovarian aging. The ovarian reserve declines with age in both quantity and quality, and this decline accelerates dramatically after age 35. Research has identified NAD+ depletion as a key molecular driver of this process.
A landmark study published in Cell Reports demonstrated that treatment with the NAD+ precursor nicotinamide mononucleotide (NMN) rejuvenated oocyte quality in aged mice and restored fertility, leading to increased pregnancy rates and live births (Bertoldo et al., 2020, doi:10.1016/j.celrep.2020.01.058). The mechanism involves restoration of mitochondrial function within the oocyte - a process that deteriorates with age as NAD+ levels fall.
More recent research from the Buck Institute identified CD38 as a specific driver of ovarian NAD+ depletion. CD38 expression increases with age within the ovaries, and mice lacking CD38 retained more primordial follicles. This suggests that targeting the CD38-NAD+ axis could be a therapeutic strategy for preserving female fertility (Perrone et al., 2023).
While human clinical trials of NAD+ supplementation for fertility are still in early stages, the preclinical evidence is strong enough that many reproductive endocrinologists have begun recommending NMN supplementation to women undergoing IVF, particularly those over 38 with diminished ovarian reserve.
GLP-1 Agonists and Fertility: A Double-Edged Sword
The relationship between GLP-1 agonists and fertility is complex. On one hand, the weight loss and metabolic improvements achieved with semaglutide and tirzepatide can restore ovulatory function in women with obesity-related anovulation and PCOS. The meta-analysis by Han et al. (2023) confirmed improved spontaneous pregnancy rates in PCOS women treated with GLP-1 agonists.
On the other hand, GLP-1 agonists themselves must be discontinued before conception due to potential fetal harm demonstrated in animal studies. This creates a clinical dilemma: the very therapy that restores fertility must be stopped before that restored fertility can be utilized.
Critical Timeline
Semaglutide should be discontinued at least 2 months before planned pregnancy due to its long half-life (approximately 7 days) and extended washout period. The FDA label states: "Discontinue OZEMPIC in women at least 2 months before a planned pregnancy." For tirzepatide (half-life approximately 5 days), a similar washout period is recommended. Women who discover they are pregnant while on a GLP-1 agonist should discontinue immediately and inform their healthcare provider.
The anecdotal "Ozempic baby" phenomenon - women becoming unexpectedly pregnant after starting GLP-1 therapy - underscores the importance of proactive fertility counseling. Several mechanisms explain the increased conception risk:
- Weight loss of 5-10% can restore ovulation in previously anovulatory women
- Improved insulin sensitivity reduces hyperandrogenism, facilitating follicular development
- Reduced oral contraceptive absorption due to delayed gastric emptying may lower contraceptive efficacy
- GLP-1 receptors are expressed on ovarian tissue and may directly influence follicular dynamics
The HPG Axis and Peptide Interventions
The hypothalamic-pituitary-gonadal (HPG) axis is the master regulatory circuit for female reproduction, and several peptides act at different levels of this cascade. Understanding where each peptide intervenes helps clinicians design rational fertility protocols and avoid unintended hormonal interactions.
Hypothalamic level: Kisspeptin neurons in the arcuate nucleus and the anteroventral periventricular nucleus (AVPV) drive GnRH pulsatility. Kisspeptin peptide therapy acts at this highest level, stimulating the natural GnRH pulse generator. This is why kisspeptin produces a more physiological downstream hormone response compared to direct GnRH administration - it goes through the body's own regulatory checkpoints.
Pituitary level: Gonadorelin (synthetic GnRH) acts directly on pituitary gonadotroph cells, stimulating FSH and LH release. When administered in pulsatile fashion, it mimics the natural hypothalamic signal. But continuous gonadorelin administration (rather than pulsatile) paradoxically downregulates the GnRH receptor and suppresses FSH/LH - a principle used in GnRH agonist protocols for endometriosis and IVF downregulation.
Ovarian level: GLP-1 receptors are expressed on ovarian granulosa cells and may directly influence follicular development and steroidogenesis. This helps explain the "Ozempic baby" phenomenon - GLP-1 agonists may affect fertility through both indirect metabolic pathways (weight loss, insulin sensitization) and direct ovarian effects.
Metabolic modulators: NAD+ operates upstream of all these levels by supporting the bioenergetic infrastructure needed for healthy oocyte maturation. Mitochondrial function within the oocyte is a rate-limiting step in fertility, and NAD+ restoration addresses this fundamental cellular requirement.
Fertility Preservation Strategies with Peptides
For women interested in extending their fertility window, peptide strategies are beginning to emerge alongside established approaches like oocyte cryopreservation:
NAD+ precursor supplementation: The preclinical evidence for NMN and NR improving oocyte quality in aging models is compelling enough that many fertility specialists have begun recommending supplementation. Typical protocols include NMN 250-500 mg daily starting 2-3 months before planned IVF or natural conception attempts. While human RCTs are still underway, the safety profile of NMN/NR at these doses is well-established, making this a reasonable addition for women over 35 with diminished ovarian reserve markers (elevated FSH, low AMH, low antral follicle count).
CoQ10 + NAD+ combination: CoQ10 (ubiquinol) is another mitochondrial support supplement with evidence for improving oocyte quality. The combination of CoQ10 (400-600 mg daily) and NMN (250-500 mg daily) targets mitochondrial function through complementary pathways - CoQ10 supports the electron transport chain while NAD+ fuels the metabolic enzymes that feed into it.
Weight optimization with GLP-1 agonists: For women with obesity-related anovulation or PCOS, achieving a healthy BMI before conception significantly improves natural conception rates and IVF outcomes. GLP-1 agonists can facilitate this weight loss efficiently, but the 2-month washout period before conception must be planned into the fertility timeline. A typical approach might be: 6-12 months of GLP-1 therapy for weight optimization, followed by 2 months of washout, followed by conception attempts or IVF.
Sequential kisspeptin in fertility treatment: For women who respond poorly to traditional IVF triggers (hCG) or who are at high OHSS risk, kisspeptin-54 offers a safer alternative that doesn't compromise oocyte quality. The clinical development pathway for kisspeptin in IVF is progressing, with several academic centers now offering kisspeptin triggering protocols.
Figure 4: Peptide washout and fertility treatment timelines for women planning pregnancy
Pregnancy & Lactation
Peptide safety during pregnancy and breastfeeding is the single most critical consideration for women of reproductive age. The evidence base ranges from well-established (for GLP-1 agonists) to essentially nonexistent (for many newer peptides). When data is lacking, the precautionary principle must govern clinical decisions.
GLP-1 Receptor Agonists: Pregnancy Category
The FDA has clear warnings regarding GLP-1 agonist use during pregnancy. Animal reproduction studies have demonstrated:
- Rats: Embryofetal mortality and structural abnormalities at exposures below the maximum recommended human dose
- Rabbits: Early pregnancy losses and structural abnormalities at clinically relevant exposures
- Monkeys: Skeletal and visceral abnormalities at higher exposures
Human data remains limited, though case reports and registry data are accumulating. One published case describes a woman who continued semaglutide for the first 3-4 weeks of pregnancy (before awareness of conception) and delivered a child without reported birth defects. However, this single case does not establish safety.
The FDA prescribing information for both Ozempic and Wegovy states that the drug "should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus." In practice, most endocrinologists and reproductive specialists interpret this as a recommendation to avoid GLP-1 agonists entirely during pregnancy.
Growth Hormone Secretagogues and Pregnancy
Growth hormone peptides like CJC-1295/Ipamorelin lack formal pregnancy safety data. Growth hormone itself plays a physiological role during pregnancy - the placenta produces its own GH variant (placental growth hormone) that gradually replaces pituitary GH from approximately week 20 onward. However, exogenous GH stimulation during early pregnancy could theoretically interfere with embryonic development.
The standard clinical recommendation is to discontinue all growth hormone secretagogues at least 1 month before planned conception and throughout pregnancy. Women should not restart GH peptides until breastfeeding is complete or until cleared by their provider.
BPC-157 and Pregnancy
BPC-157 has no published pregnancy safety data in humans. Its mechanism of action - promoting angiogenesis, modulating growth factor expression, and enhancing tissue repair - raises theoretical concerns during embryonic and fetal development, when precise control of vascular and tissue growth is critical. The angiogenic properties that make BPC-157 therapeutic for wound healing could potentially disrupt the delicate vascular development occurring in an embryo.
Women planning pregnancy should discontinue BPC-157 at least 2-4 weeks before attempting conception, given its relatively short half-life. During breastfeeding, the transfer of BPC-157 into breast milk is unknown, and avoidance is recommended.
Other Peptides: Pregnancy Safety Summary
| Peptide | Pregnancy Safety | Recommended Washout | Lactation |
|---|---|---|---|
| Semaglutide | Contraindicated - animal teratogenicity | At least 2 months | Unknown; avoid |
| Tirzepatide | Contraindicated - animal teratogenicity | At least 2 months | Unknown; avoid |
| Kisspeptin-54 | Used therapeutically in IVF context | Single dose; short half-life | Not applicable |
| Gonadorelin | Used therapeutically for ovulation induction | Discontinued once ovulation confirmed | Unknown |
| CJC-1295/Ipamorelin | No data; avoid | 1 month minimum | Unknown; avoid |
| BPC-157 | No data; avoid | 2-4 weeks | Unknown; avoid |
| GHK-Cu | No systemic data; topical likely low risk | Topical: none. Systemic: 2 weeks | Topical likely safe |
| Epithalon | No human data; avoid | 2-4 weeks | Unknown; avoid |
| Oxytocin | Used medically for labor induction | Under medical supervision only | Physiologically present; exogenous use under supervision |
| NAD+/NMN | Limited data; some preclinical fertility benefit | Discuss with provider | Unknown |
Essential Rule for Pregnancy Planning
If in doubt, stop the peptide. No peptide therapy is worth risking fetal harm. Women should adopt a "guilty until proven innocent" approach to peptide use during pregnancy and breastfeeding. Discuss all peptide therapies with both your prescribing provider and your obstetrician before attempting conception.
Breastfeeding Considerations
Virtually all therapeutic peptides lack formal lactation safety data. The key pharmacological considerations for breastfeeding are:
- Molecular weight: Larger peptides (>1,000 Da) have limited transfer into breast milk. Semaglutide (4,113 Da) and tirzepatide (4,813 Da) would be expected to have low milk transfer based on size alone.
- Oral bioavailability in infants: Even if peptides enter breast milk, most are degraded by the infant's GI tract before systemic absorption. This provides a secondary safety margin.
- Half-life: Long-acting peptides like semaglutide (7-day half-life) accumulate in maternal tissues and could produce sustained low-level milk concentrations.
Despite these theoretical reassurances, the absence of human lactation data for most peptides means the recommendation remains avoidance during breastfeeding unless the clinical benefit clearly outweighs the unknown risk.
Managing the GLP-1 to Pregnancy Transition
The transition from GLP-1 therapy to pregnancy planning is one of the most clinically challenging aspects of women's peptide therapy. Women face a genuine dilemma: GLP-1 agonists may have restored their fertility (through weight loss and metabolic improvement), but the very medication that made pregnancy possible must now be stopped well before conception.
The weight rebound concern: Many women fear regaining weight during the 2-month washout period before conception. The data on weight maintenance after GLP-1 discontinuation shows variable results - some women maintain significant weight loss through established behavioral changes, while others experience substantial rebound. Strategies to minimize rebound during the washout period include:
- Gradual dose reduction rather than abrupt cessation (e.g., stepping down from 1.0 mg to 0.5 mg to 0.25 mg semaglutide over 4-6 weeks before stopping)
- Intensive dietary counseling during the washout period, focusing on high-protein, moderate-carbohydrate meals that support satiety without pharmacological appetite suppression
- Increasing exercise intensity during the washout period to maintain metabolic rate
- Consideration of metformin (pregnancy-safe) as a metabolic bridge during the washout and early pregnancy period for women with PCOS or insulin resistance
- Psychological preparation for the return of normal appetite signals, which can feel overwhelming after months of GLP-1-mediated suppression
The fertility timing paradox: Women with PCOS may ovulate for the first time in months or years shortly after starting GLP-1 therapy - sometimes before they've achieved their weight loss goals and before they're ready for pregnancy. Some women have reported unexpected pregnancies within the first few months of GLP-1 therapy, before they could complete the recommended washout. This underscores the need for proactive contraception counseling from the very first GLP-1 prescription in reproductive-age women.
The emotional dimension: For women who've struggled with infertility, the prospect of stopping a medication that helped them lose weight and restore ovulation can trigger significant anxiety. Clinicians should acknowledge this emotional complexity and provide reassurance that the metabolic improvements achieved during GLP-1 therapy often persist even after discontinuation, particularly if the weight loss was accompanied by lifestyle changes.
Pregnancy After Peptide Therapy: What We Know
Registry data and pharmacovigilance reports are gradually building a picture of pregnancy outcomes following peptide therapy exposure:
Post-GLP-1 pregnancies: Emerging data from pregnancy registries suggests that women who appropriately discontinued GLP-1 agonists before conception have pregnancy outcomes comparable to the general population, adjusted for BMI. The key word is "appropriately" - meaning the full 2-month washout was observed. Pregnancies with inadvertent early exposure (first trimester before awareness of pregnancy) have shown mixed outcomes, with insufficient data to draw definitive conclusions about risk.
Post-GH peptide pregnancies: Even less formal data exists for women who used growth hormone secretagogues before pregnancy. Given that GH secretagogues have short half-lives (hours rather than weeks), a 1-month washout should provide adequate clearance. Women who used CJC-1295/Ipamorelin before conception and then discontinued appropriately would not be expected to have residual drug exposure during embryonic development.
IVF with kisspeptin trigger: This is the one area where direct pregnancy data exists from controlled clinical trials. Kisspeptin-54 triggered IVF cycles have produced healthy pregnancies and live births across multiple trials, with no increase in congenital anomalies. The short half-life of kisspeptin (28 minutes for KP54) means it's cleared from the body well before embryonic development begins.
Postpartum Peptide Therapy Resumption
After delivery, women often want to resume peptide therapy for weight loss, recovery, or hormonal support. Timing depends on breastfeeding status:
If not breastfeeding: Most peptides can be resumed 6-8 weeks postpartum, after the initial recovery period and return to hemodynamic stability. GLP-1 agonists can be started for postpartum weight management. GH secretagogues can resume for sleep and recovery support. BPC-157 can be used for postpartum tissue healing (episiotomy, cesarean incision, diastasis recti).
If breastfeeding: The conservative approach is to avoid all systemic peptide therapy until breastfeeding is complete or the infant is primarily taking solid foods (typically 6-12 months). Topical peptides (GHK-Cu) can be used on areas that don't contact the infant (avoiding breast area). Oral collagen peptides are generally considered safe during breastfeeding as they're food-derived amino acid supplements.
The postpartum weight loss decision: Some women feel pressure to lose pregnancy weight quickly and consider GLP-1 agonists during breastfeeding. While the pharmacological properties of semaglutide (large molecular weight, likely low breast milk transfer) suggest limited infant exposure, the absence of human lactation data makes this a risk-benefit discussion between the patient, her obstetrician, and her pediatrician. The general recommendation remains to defer GLP-1 therapy until breastfeeding is complete.
Menopause & Peptide Support
Menopause marks a profound shift in a woman's hormonal landscape. Estrogen and progesterone decline precipitously, growth hormone secretion falls, melatonin production wanes, and inflammation markers rise. Peptide therapies can address several of these age-related changes, offering support for symptoms that traditional hormone replacement may not fully resolve.
The Menopausal Hormone Transition
The menopausal transition typically begins in the mid-40s and completes by the early-to-mid 50s. During this period, women experience:
- A 70-80% decline in estradiol levels
- A 60% decline in progesterone production
- A 50% reduction in growth hormone secretion compared to premenopausal levels
- Declining melatonin production affecting sleep architecture
- Increasing inflammatory markers (IL-6, TNF-alpha, CRP)
- Accelerated bone density loss (2-3% per year in the first 5-7 years post-menopause)
- Redistribution of body fat toward visceral stores
- Declining NAD+ levels in multiple tissues
Peptide therapies don't replace estrogen and progesterone - that's the domain of hormone replacement therapy (HRT). Instead, peptides can address the non-gonadal hormonal and metabolic shifts that accompany menopause, often complementing traditional HRT.
Growth Hormone Peptides for Menopausal Women
CJC-1295/Ipamorelin is the most commonly prescribed GH secretagogue combination for menopausal women. By stimulating the body's own pituitary GH release, this combination can address several menopausal complaints simultaneously:
Body composition: GH peptides promote lipolysis and preserve lean muscle mass, counteracting the visceral fat accumulation and sarcopenia that accelerate after menopause. Most women begin noticing body composition changes within 4-6 weeks of consistent use, with more significant changes over 3-6 months.
Sleep quality: Ipamorelin enhances slow-wave (deep) sleep when administered in the evening, aligning with the natural nocturnal GH pulse. For menopausal women who often experience disrupted sleep due to hot flashes and hormonal changes, this can be a significant quality-of-life improvement.
Skin and hair: GH stimulates collagen synthesis, and the decline in both GH and estrogen during menopause contributes to accelerated skin thinning and hair changes. Women on CJC-1295/Ipamorelin frequently report improved skin texture, hydration, and thickness within 2-3 months.
Recovery and healing: Postmenopausal women experience slower tissue repair. GH peptides support recovery from exercise and minor injuries, encouraging the active lifestyle that's so important for bone and cardiovascular health after menopause.
Dosing Considerations for Menopausal Women
Menopausal and postmenopausal women often require lower GH peptide doses than premenopausal women or men. Starting doses of CJC-1295 at 100 mcg and Ipamorelin at 100-200 mcg (combined nightly injection) are typical, with titration based on response and IGF-1 levels. The goal is physiological GH restoration, not supraphysiological elevation. IGF-1 should be monitored every 3-4 months and maintained in the upper quartile of the age-adjusted reference range.
Oxytocin and Menopausal Health
Oxytocin levels decline as women transition through menopause, and this decline correlates with increased susceptibility to mood disorders, poor sleep, osteoporosis, and cardiovascular disease (Thiagarajan et al., 2024, doi:10.1016/j.beem.2024.101866). The research connecting oxytocin to menopausal health is growing rapidly.
Mood regulation: A study presented at ENDO 2025 found that women with higher endogenous oxytocin levels experienced less mood disturbance following disrupted sleep - a common menopausal complaint. Exogenous oxytocin (intranasal, 24 IU) has shown anxiolytic and antidepressant effects in pilot studies, though large-scale menopausal trials are still needed.
Bone health: Oxytocin has a direct anabolic effect on bone tissue, stimulating osteoblast activity while inhibiting osteoclast-mediated resorption. Lower circulating oxytocin levels are associated with reduced bone mineral density in postmenopausal women. In animal models, oxytocin administration normalized ovariectomy-induced osteopenia, suggesting it could be therapeutic for menopausal osteoporosis (Tamma et al., 2009, doi:10.1073/pnas.0901890106).
Cardiovascular protection: Oxytocin exerts cardioprotective effects through anti-inflammatory signaling, blood pressure modulation, and cardiomyocyte differentiation. The postmenopausal decline in both estrogen and oxytocin may contribute to the sharp increase in cardiovascular risk women experience after menopause.
Epithalon and Menopausal Symptoms
Epithalon (Ala-Glu-Asp-Gly), the synthetic tetrapeptide modeled after the naturally occurring pineal peptide epithalamin, has been used clinically in Russia for menopausal symptom management. Its primary mechanism involves stimulation of telomerase activity and upregulation of melatonin production by the pineal gland.
The melatonin connection is particularly relevant for menopausal women. Melatonin production declines with age, and melatonin supplementation has demonstrated benefit for menopausal symptoms including hot flashes, sleep disturbance, and mood changes. Epithalon may achieve similar benefits by restoring endogenous melatonin secretion rather than relying on exogenous supplementation.
In clinical settings, epithalon has been associated with improved sleep quality, reduced hot flash frequency, and enhanced overall well-being in menopausal women, though these observations come primarily from clinical practice rather than randomized controlled trials. Human studies have shown that epithalon significantly increased telomere lengths in blood cells of patients aged 60-65 and 75-80.
NAD+ and Menopausal Metabolic Support
NAD+ levels decline approximately 50% between ages 40 and 60 - a timeframe that overlaps precisely with the menopausal transition. This decline in NAD+ contributes to mitochondrial dysfunction, impaired DNA repair, metabolic deterioration, and increased inflammation - all hallmarks of both aging and the post-menopausal metabolic state.
For menopausal women, NAD+ restoration through precursor supplementation (NMN or NR) or direct NAD+ injection may support:
- Mitochondrial energy production (addressing fatigue)
- Sirtuin activation (supporting metabolic health and inflammation control)
- DNA repair capacity (reducing cellular senescence)
- Cellular NAD+ levels in reproductive and non-reproductive tissues
Women with PCOS also show decreased NAD+ levels in muscle tissue, correlating with the distinct pattern of insulin resistance observed in PCOS patients. NAD+ supplementation may therefore benefit premenopausal women with PCOS as well as postmenopausal women.
GLP-1 Agonists in Menopausal Weight Management
Weight gain during the menopausal transition is nearly universal, with the average woman gaining 1.5 pounds per year during perimenopause. The shift from subcutaneous to visceral fat distribution that accompanies estrogen decline creates metabolic risk disproportionate to the actual weight gain. GLP-1 receptor agonists address this menopausal metabolic shift directly.
Semaglutide and tirzepatide reduce visceral fat preferentially, improve insulin sensitivity, lower inflammatory markers, and protect cardiovascular health - all of which deteriorate after menopause. The absence of pregnancy risk in postmenopausal women simplifies prescribing, and the more stable pharmacokinetic profile (no menstrual cycle variability) makes dosing more predictable.
Key considerations for menopausal women on GLP-1 therapy:
- Prioritize protein intake (at least 1.2-1.6 g/kg/day) to prevent accelerated sarcopenia
- Combine with resistance training to preserve bone density and lean muscle mass
- Monitor bone density if weight loss exceeds 10% of body weight
- Consider combining with collagen peptides for bone and skin support
- Extend dose titration intervals if GI side effects are problematic - there's no fertility timeline pressure
Comprehensive Menopausal Peptide Protocol
An integrated peptide approach for menopausal health might include several complementary interventions. This is not a one-size-fits-all recommendation but rather a framework that clinicians can customize based on individual symptom profiles, lab values, and patient goals:
| Symptom Cluster | Primary Peptide | Supporting Peptide(s) | Expected Timeline |
|---|---|---|---|
| Weight gain / metabolic decline | Semaglutide or Tirzepatide | CJC-1295/Ipamorelin (body composition) | 4-12 weeks for weight; 3-6 months for metabolic markers |
| Sleep disruption / hot flashes | CJC-1295/Ipamorelin (nightly) | Epithalon (melatonin support) | 2-4 weeks for sleep; 6-12 weeks for hot flashes |
| Mood / anxiety / emotional lability | Oxytocin (intranasal) | NAD+ (neurotransmitter support) | 1-4 weeks for acute mood; 2-3 months for sustained benefit |
| Bone density decline | Collagen peptides (oral, 5 g daily) | Oxytocin; CJC-1295/Ipamorelin | 6-12 months for measurable BMD changes |
| Skin thinning / wrinkles / hair | GHK-Cu (topical + optional injectable) | Collagen peptides; CJC-1295/Ipamorelin | 4-12 weeks for skin; 3-6 months for hair |
| Fatigue / energy decline | NAD+ (NMN oral or NAD+ SubQ) | CJC-1295/Ipamorelin | 1-4 weeks for subjective energy improvement |
| Joint pain / tissue healing | BPC-157 | GHK-Cu; Collagen peptides | 2-6 weeks for joint symptom improvement |
It bears repeating: none of these peptides replace hormone replacement therapy when HRT is indicated. Estrogen and progesterone replacement address the root hormonal deficit of menopause. Peptides address the broader metabolic, regenerative, and functional consequences that HRT alone may not fully correct. The most effective approach for many menopausal women combines appropriately prescribed HRT with targeted peptide therapies.
The Menopausal Gut-Brain Axis and Peptides
The gut-brain axis undergoes significant changes during menopause that affect both peptide metabolism and efficacy. Estrogen receptors in the gut influence microbiome composition, intestinal permeability, and enteroendocrine cell function. As estrogen declines:
- Gut microbiome diversity decreases, potentially altering peptide metabolism and absorption
- Intestinal permeability may increase, affecting peptide bioavailability
- Enteroendocrine L-cells (which produce endogenous GLP-1) may function differently, potentially explaining altered responses to exogenous GLP-1 agonists
- Vagal tone decreases, affecting the gut-brain communication that mediates appetite regulation and mood
BPC-157, with its demonstrated effects on gut mucosal healing and integrity, may be particularly relevant in the menopausal context where gut barrier function is compromised. Similarly, oxytocin's effects on vagal tone could help restore gut-brain communication that deteriorates with estrogen loss.
Figure 5: Peptide therapy options mapped to common menopausal symptoms and concerns
Female-Specific Dosing
One dose does not fit all - and it definitely doesn't fit both sexes equally. The pharmacokinetic differences between women and men, combined with hormonal influences on drug response, support a case for sex-specific peptide dosing. Here's what the evidence suggests for each major peptide category.
GLP-1 Receptor Agonist Dosing for Women
Current FDA-approved dosing for semaglutide and tirzepatide is identical for men and women. Both drugs use standardized titration schedules regardless of sex. However, the clinical data suggest that women may benefit from modified approaches:
| Peptide | Standard Titration | Female-Adjusted Approach | Rationale |
|---|---|---|---|
| Semaglutide (Ozempic/Wegovy) | 0.25 mg x 4 wks, 0.5 mg x 4 wks, escalate to 1.0-2.4 mg | Extend each step to 5-6 weeks; consider 0.5 mg maintenance if tolerated and effective | Higher plasma levels at equal doses; greater GI sensitivity |
| Tirzepatide (Mounjaro/Zepbound) | 2.5 mg x 4 wks, 5 mg x 4 wks, escalate to 10-15 mg | Extend each step to 5-6 weeks; many women achieve goals at 7.5-10 mg | Women often achieve target weight loss at lower doses than men |
| Oral semaglutide (Rybelsus) | 3 mg x 30 days, 7 mg x 30 days, 14 mg maintenance | May need extended fasting window due to slower gastric emptying | Absorption variability with hormonal cycle phases |
Growth Hormone Secretagogue Dosing
Women typically require lower GH secretagogue doses than men to achieve equivalent IGF-1 elevation. This is partly because women have higher baseline GH pulse frequency (though lower pulse amplitude) and partly because of body weight differences. Practical dosing guidelines:
CJC-1295 (no DAC) / Ipamorelin combination:
- Starting dose: CJC-1295 100 mcg + Ipamorelin 100 mcg, once nightly before bed
- Titration: Increase Ipamorelin to 200 mcg after 2-4 weeks if tolerated
- Maintenance range: CJC-1295 100-200 mcg + Ipamorelin 200-300 mcg nightly
- Cycle: 5 days on / 2 days off, or 3 months on / 1 month off
- Note: Men typically start at CJC-1295 150-200 mcg + Ipamorelin 200-300 mcg
Monitoring: Check IGF-1 levels at baseline, 6 weeks, and every 3-4 months thereafter. Target the upper quartile of the age-adjusted reference range. Fasting glucose and HbA1c should also be monitored, as GH can reduce insulin sensitivity in some individuals.
BPC-157 Dosing for Women
BPC-157 dosing in clinical practice does not typically differ between sexes, as the peptide acts locally at the site of injury rather than producing systemic hormonal effects. Standard dosing guidelines:
- Subcutaneous injection: 250-500 mcg once or twice daily near the injury site
- Typical treatment course: 4-8 weeks
- Women may consider starting at 250 mcg and increasing only if needed
- No data exists on menstrual cycle-specific timing, but avoiding high-dose use during the proliferative phase (when endometrial angiogenesis is active) is a reasonable precaution
GHK-Cu Dosing
GHK-Cu is used both topically and via subcutaneous injection. For women's skin and hair applications:
Topical use:
- Creams or serums containing 1-3% GHK-Cu applied twice daily
- No sex-specific dosing differences for topical application
- Clinical trial data in 71 women showed visible improvements in skin aging markers after 12 weeks of topical use
Subcutaneous injection:
- Starting dose: 1-2 mg daily or every other day
- Treatment course: 4-8 weeks on, 2-4 weeks off
- Primarily used for systemic anti-aging, wound healing, and hair growth support
Kisspeptin Dosing in Fertility Treatment
Kisspeptin dosing in fertility contexts is weight-based, which inherently adjusts for sex differences:
- IVF trigger: Kisspeptin-54 at 9.6 nmol/kg as a single subcutaneous injection, typically administered 36 hours before egg retrieval
- Diagnostic testing: Kisspeptin-10 at 1-10 nmol/kg IV to assess GnRH neuron responsiveness
- Dosing is exclusively female-specific in the IVF context
Oxytocin Dosing for Women
Oxytocin dosing for non-obstetric applications in women:
- Intranasal: 12-24 IU per dose, 1-2 times daily
- Sublingual: 10-20 IU per dose
- Start at the lower end and titrate based on response
- Women may be more sensitive to oxytocin's mood and social effects than men due to estrogen-oxytocin receptor interactions
Epithalon and NAD+ Dosing for Women
Epithalon dosing:
- Standard protocol: 5-10 mg SubQ daily for 10-20 consecutive days
- Cycle frequency: 2-3 times per year (e.g., January, May, September)
- Some practitioners use a lower dose of 3-5 mg daily for longer cycles (20-30 days)
- No sex-specific dose adjustment is established, but starting at 5 mg is reasonable for women
- Evening administration preferred (aligns with pineal gland circadian activity)
NAD+ dosing for women:
- Oral NMN: 250-500 mg daily (divided or single morning dose)
- Oral NR (nicotinamide riboside): 250-300 mg daily
- SubQ NAD+ injection: 50-100 mg daily or every other day for loading; then 2-3x/week for maintenance
- IV NAD+ infusion: 250-500 mg per session; loading protocol of 4-6 sessions over 2 weeks, then monthly maintenance
- Women may be more sensitive to IV NAD+ side effects (nausea, chest pressure) - slower infusion rates are recommended
- For fertility support: NMN 250-500 mg daily starting 2-3 months before conception attempts
Collagen Peptide Dosing
Oral collagen peptides are among the safest and most accessible peptide therapies for women. Dosing is well-established:
- Skin health: 2.5-10 g daily of hydrolyzed collagen peptides (type I and III). Benefits typically seen at 8-12 weeks. Higher doses (10 g) may produce faster results.
- Bone health: 5 g daily of specific bioactive collagen peptides. The clinical trial data used the FORTIBONE formulation at 5 g daily. Benefits measurable at 6-12 months by DEXA scan.
- Joint health: 10 g daily of hydrolyzed collagen or 40 mg of undenatured type II collagen. Joint benefits typically reported at 3-6 months.
- Combined approach: Many women take 10 g hydrolyzed collagen daily, achieving benefits for skin, bone, and joints simultaneously.
- Collagen peptides can be mixed into coffee, smoothies, or water and taken at any time of day.
- No sex-specific dosing differences; no known interactions with other peptide therapies.
General Female Dosing Principles
- Start lower: Begin at 60-75% of the standard (typically male-derived) dose
- Titrate slower: Extend titration intervals by 25-50%
- Monitor more frequently: Check labs at 4-6 weeks rather than 8-12 weeks
- Consider cycle timing: For peptides with dose-sensitive side effects, the follicular phase (days 1-14) may be better tolerated than the luteal phase
- Weight-based when available: Use weight-based dosing rather than fixed doses when pharmacokinetic data supports it
Figure 6: Female-adjusted peptide dosing reference compared to standard (sex-neutral) recommendations
Hormonal Interactions
Peptide therapies don't operate in isolation - they interact with a woman's existing hormonal milieu in ways that can enhance therapeutic effects or create unwanted complications. Understanding these interactions is essential for safe and effective protocol design.
Interactions with Hormone Replacement Therapy (HRT)
Many menopausal women use peptides alongside estrogen, progesterone, or combination HRT. Here's what clinicians need to know about key interactions:
GLP-1 agonists + HRT: No significant pharmacokinetic interaction has been identified. Estrogen replacement may actually enhance GLP-1 receptor expression, potentially improving the efficacy of GLP-1 agonists in postmenopausal women on HRT. Both therapies independently improve cardiovascular risk markers, and their combination appears to be safe based on current evidence.
GH secretagogues + HRT: Oral estrogen increases sex hormone-binding globulin (SHBG) and reduces IGF-1 levels through a hepatic first-pass effect. Women on oral estrogen who also take GH secretagogues may require modestly higher GH peptide doses to achieve the same IGF-1 elevation. Transdermal estrogen avoids this first-pass effect and doesn't attenuate the IGF-1 response. Clinicians should consider the route of estrogen administration when dosing GH peptides.
BPC-157 + HRT: No known interactions. BPC-157's local tissue repair mechanisms operate independently of gonadal hormones. However, estrogen's own wound-healing effects may complement BPC-157's actions, potentially making the combination more effective than either alone for postmenopausal women with musculoskeletal issues.
Interactions with Oral Contraceptives
As discussed in the GLP-1 section, delayed gastric emptying can reduce oral contraceptive absorption. Beyond GLP-1 agonists, other peptides that affect GI motility include:
- Ghrelin mimetics (Ipamorelin): Can modestly increase gastric motility, potentially having the opposite effect of GLP-1 agonists on oral drug absorption
- Oxytocin: Has been shown to influence gut motility through vagal pathways, though the clinical significance for oral contraceptive absorption is unclear
For women on peptide therapies who require reliable contraception, non-oral methods (IUD, implant, injection, or barrier methods) eliminate this concern entirely.
Interactions with Thyroid Hormones
Thyroid disease is 5-8 times more common in women than men, making thyroid-peptide interactions particularly relevant for female patients. Key considerations:
GLP-1 agonists and thyroid: Semaglutide and other GLP-1 agonists carry a boxed warning regarding thyroid C-cell tumors based on rodent studies. While the clinical significance in humans remains debated (rodent C-cells express GLP-1 receptors far more densely than human C-cells), GLP-1 agonists are contraindicated in patients with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2. Routine thyroid monitoring (TSH, calcitonin if warranted) should be part of GLP-1 follow-up in women.
GH secretagogues and thyroid: Growth hormone increases the conversion of T4 to T3 via upregulation of peripheral deiodinase enzymes. Women on levothyroxine who begin GH peptide therapy may need thyroid dose adjustment as their T4-to-T3 conversion changes. TSH should be checked 6-8 weeks after initiating GH peptides.
Interactions with Fertility Medications
Women undergoing fertility treatment may be on multiple hormonal medications simultaneously. The interaction landscape includes:
Kisspeptin + gonadotropins: Kisspeptin-54 is used as an alternative to hCG for triggering final oocyte maturation during stimulated IVF cycles. It works through the physiological GnRH pathway, producing a more controlled and self-limiting LH surge. Kisspeptin can be used in conjunction with exogenous gonadotropins (FSH/LH) during the stimulation phase without adverse interaction.
Gonadorelin + clomiphene/letrozole: Gonadorelin has been used alongside ovulation induction agents. Pulsatile gonadorelin provides the GnRH stimulus while clomiphene or letrozole blocks estrogen negative feedback, creating a combined effect on gonadotropin release. This combination requires careful monitoring to prevent ovarian hyperstimulation.
Peptide Stacking Considerations for Women
Many women use multiple peptides simultaneously. While formal interaction studies between peptide therapeutics are largely absent, clinical experience and mechanistic reasoning provide guidance:
| Combination | Interaction Profile | Female-Specific Notes |
|---|---|---|
| GLP-1 + GH secretagogue | Complementary - GLP-1 improves insulin sensitivity, offsetting GH's insulin-reducing effect | Popular for menopausal weight management; monitor glucose closely |
| BPC-157 + GHK-Cu | Complementary for tissue repair; different mechanisms | Safe combination; commonly used for skin rejuvenation protocols |
| CJC-1295/Ipamorelin + Epithalon | Complementary anti-aging mechanisms | Both support sleep quality; evening dosing for maximum benefit |
| NAD+ + Epithalon | Complementary cellular aging mechanisms | NAD+ supports DNA repair; Epithalon targets telomere maintenance |
| Oxytocin + GLP-1 | No known interaction; complementary mood benefits | Both may improve emotional well-being in menopausal women |
| GLP-1 + Kisspeptin | Sequential use only - GLP-1 must be stopped before fertility treatment | Minimum 2-month GLP-1 washout before kisspeptin use in IVF |
The Estrogen-Peptide Timing Matrix
For clinicians managing women on multiple therapies, this matrix shows how female hormonal status affects each peptide class:
| Hormonal Status | GLP-1 Agonists | GH Secretagogues | Healing Peptides | Longevity Peptides |
|---|---|---|---|---|
| High estrogen (follicular peak) | Enhanced GLP-1R expression; potentially stronger effect | Higher GH release but lower IGF-1 response | Enhanced healing capacity alongside estrogen | Optimal cellular repair environment |
| High progesterone (luteal) | Worse GI tolerance; monitor nausea | Neutral GH response | Slightly reduced healing speed | Neutral |
| Low estrogen + progesterone (menstruation) | Moderate tolerance; inflammatory milieu | Neutral | Reduced baseline healing capacity | Neutral |
| Postmenopausal (consistently low) | Consistent response; no cycle variation | Lower baseline GH; potentially greater relative benefit | Reduced baseline healing; peptides more valuable | Highest need; most consistent response |
| On oral HRT | May enhance response | Oral estrogen reduces IGF-1; may need higher GH dose | HRT restores some healing capacity | HRT + peptides may be additive |
| On transdermal HRT | May enhance response | No IGF-1 suppression; standard dosing | HRT restores some healing capacity | HRT + peptides may be additive |
Drug-Peptide Interaction Depth: Common Scenarios
Beyond the summary tables, several common clinical scenarios deserve more detailed discussion:
Scenario: Woman on metformin + starting semaglutide
Metformin is the most commonly prescribed medication for PCOS and prediabetes in women. When adding a GLP-1 agonist, both drugs can cause GI side effects (nausea, diarrhea, bloating). The recommended approach is to temporarily reduce metformin to 500 mg daily when initiating semaglutide, then gradually increase back to the target metformin dose as GLP-1 tolerance develops. Monitor B12 levels, as both drugs can contribute to B12 depletion. The metabolic benefits of the combination are complementary - metformin acts primarily on hepatic glucose production while GLP-1 agonists work through multiple mechanisms including appetite suppression, beta cell support, and peripheral insulin sensitization.
Scenario: Woman on levothyroxine + starting CJC-1295/Ipamorelin
As noted earlier, GH increases T4-to-T3 conversion. When a hypothyroid woman on levothyroxine starts GH peptides, she may notice subtle changes in her thyroid medication needs. Typically, the conversion of T4 to active T3 increases, which can either reduce the need for levothyroxine (if she was underconverted) or create a relative T3 excess (symptoms: palpitations, anxiety, heat intolerance). Check TSH and free T3/T4 at 6-8 weeks after starting GH peptides and adjust levothyroxine if needed.
Scenario: Woman on SSRI + adding oxytocin
SSRIs are among the most prescribed medications for women, and many perimenopausal and postmenopausal women use them for mood symptoms and hot flash management. SSRIs can blunt oxytocin receptor sensitivity, potentially reducing the subjective benefits of exogenous oxytocin. Additionally, serotonin and oxytocin systems interact in the brain - both modulate anxiety, social behavior, and emotional regulation. Women on SSRIs who add intranasal oxytocin may need slightly higher oxytocin doses to achieve the desired anxiolytic effect. Monitor mood carefully during the first month of combination use.
Scenario: Woman on aromatase inhibitor (breast cancer prevention) + peptide therapy
Aromatase inhibitors (anastrozole, letrozole) are used in breast cancer prevention and treatment, creating a profoundly hypoestrogenic state. These women experience accelerated bone loss, joint pain, and menopausal symptoms that are often more severe than natural menopause because estrogen is nearly completely suppressed. Collagen peptides for bone health are safe and appropriate. GHK-Cu for skin support is reasonable. GH secretagogues require careful consideration because IGF-1 elevation has been associated with breast cancer risk in some studies - discuss with the treating oncologist. GLP-1 agonists are generally safe and may help with the weight gain common on aromatase inhibitor therapy.
Scenario: Woman on bioidentical testosterone + GH peptides
Low-dose testosterone therapy is increasingly used in postmenopausal women for libido, energy, and muscle maintenance. Adding GH secretagogues creates a complementary anabolic signal - testosterone supports muscle protein synthesis while GH promotes lipolysis and collagen formation. The combination can be effective for body composition optimization but requires monitoring of both IGF-1 and free testosterone to avoid supraphysiological levels of either. Lipid panels should be checked regularly, as both testosterone and GH can affect lipid profiles.
For a deeper exploration of peptide combinations and stacking protocols, our peptide research hub provides additional resources.
Skin & Hair Peptides for Women
Skin and hair changes are among the most visible consequences of aging and hormonal shifts in women. Collagen loss accelerates after menopause, hair thinning affects up to 50% of women by age 50, and wound healing slows with declining estrogen. Several peptides directly target these processes.
GHK-Cu: The Skin Renewal Peptide
GHK-Cu (glycyl-L-histidyl-L-lysine copper complex) is among the most extensively studied peptides for skin rejuvenation in women. First isolated from human plasma, GHK-Cu levels decline from approximately 200 ng/mL at age 20 to just 80 ng/mL by age 60 - a decline that coincides with the visible loss of regenerative capacity in skin and other tissues.
Clinical evidence specifically in women is strong:
- A study of 71 women with mild to advanced photoaging found that GHK-Cu facial cream improved skin laxity, clarity, fine lines, wrinkle depth, skin density, and thickness after 12 weeks of use (Pickart et al., 2015, doi:10.3390/ijms160920163)
- A separate trial in 21 women showed an average 28% increase in collagen production after 3 months of daily GHK-Cu application, with the top quartile of responders achieving a 51% improvement
- A comparative study found GHK-Cu superior to vitamin C cream and retinoic acid for stimulating collagen production - 70% of women treated with GHK-Cu showed increased collagen versus 50% with vitamin C and 40% with retinoic acid
GHK-Cu works through multiple mechanisms relevant to aging skin: stimulating blood vessel and nerve outgrowth, increasing collagen, elastin, and glycosaminoglycan synthesis, supporting dermal fibroblast function, and activating genes involved in tissue remodeling. Over 4,000 genes have been identified as responsive to GHK-Cu treatment.
Collagen Peptides for Skin and Bone
While injectable peptides like GHK-Cu work from the inside out, oral collagen peptides (hydrolyzed collagen) provide building blocks for skin, hair, nails, and bones. The evidence for oral collagen peptides in women is substantial:
Skin benefits: Multiple randomized controlled trials in women have demonstrated improvements in skin elasticity, hydration, and wrinkle depth with oral collagen peptide supplementation (typically 2.5-10 g daily for 8-12 weeks). A meta-analysis of collagen supplementation trials found significant improvements in skin hydration, elasticity, and wrinkle reduction, with effects more pronounced in older women.
Bone benefits: A randomized controlled trial of 131 postmenopausal women demonstrated that 5 g of specific collagen peptides daily significantly increased bone mineral density at the lumbar spine and femoral neck, as well as the bone formation marker P1NP (Konig et al., 2018, doi:10.3390/nu10010097). A 4-year follow-up confirmed sustained benefits with continued supplementation.
Hair Growth Peptides
Female pattern hair loss (androgenetic alopecia) and telogen effluvium (stress/hormone-related diffuse shedding) are common concerns. Several peptides show promise:
GHK-Cu for hair: GHK-Cu promotes hair growth through multiple mechanisms - stimulating fibroblast proliferation, promoting dermal papilla cell activity, enhancing blood vessel formation to supply hair follicles, and inhibiting TGF-beta (which can cause premature follicle miniaturization). Both topical and injectable GHK-Cu have been used in clinical practice for hair thinning.
PTD-DBM (copper peptide variant): This copper peptide variant has shown hair growth activity in preclinical studies by promoting the Wnt/beta-catenin signaling pathway that activates hair follicle stem cells.
Growth hormone peptides: The GH-IGF-1 axis plays a role in hair cycling. Women on CJC-1295/Ipamorelin frequently report improvements in hair thickness and growth rate, likely mediated through increased IGF-1's effect on hair follicle dermal papilla cells. These reports are observational rather than from controlled trials.
Skin Protocol for Women Over 40
A comprehensive peptide-based skin and hair protocol for women over 40 might include:
| Layer | Intervention | Frequency | Expected Timeline |
|---|---|---|---|
| Topical | GHK-Cu serum (1-3%) applied to face, neck, scalp | Twice daily | 4-12 weeks for visible improvement |
| Oral | Hydrolyzed collagen peptides 5-10 g | Daily | 8-12 weeks for skin; 6-12 months for bone |
| Injectable (optional) | GHK-Cu 1-2 mg SubQ | Daily or every other day x 4-8 weeks | 4-8 weeks for enhanced effects |
| Injectable (optional) | CJC-1295/Ipamorelin (systemic GH support) | Nightly | 2-3 months for skin/hair effects |
Understanding Peptide Skincare in the Context of Female Hormones
Women's skin is a hormonally responsive organ that undergoes predictable changes across the lifespan. Understanding these changes helps explain why peptide skincare is particularly relevant for women and when different interventions make the most sense:
Reproductive years (20s-30s): Skin is at its thickest and most hydrated, supported by healthy estrogen and collagen production. Peptide skincare needs are minimal for most women, though GHK-Cu can help with acne scarring, hyperpigmentation, and sun damage prevention. Endogenous GHK-Cu levels are near their peak (200 ng/mL at age 20).
Late 30s to early 40s: Collagen production begins declining at approximately 1% per year after age 30, and the cumulative effect becomes noticeable in the late 30s. This is when many women first notice fine lines, reduced skin elasticity, and slower wound healing. Starting topical GHK-Cu and oral collagen peptides during this period can help slow the visible progression of aging by supporting collagen synthesis from both inside and outside.
Perimenopause (mid-40s to early 50s): The accelerated decline in estrogen produces rapid skin changes - skin can lose up to 30% of its collagen in the first five years after menopause. Women in perimenopause often notice a dramatic shift in skin quality that feels sudden even though the underlying process has been gradual. This is the critical window where peptide skincare interventions (GHK-Cu, collagen peptides, GH secretagogues) can have the most impact by compensating for the lost estrogenic stimulation of collagen synthesis.
Postmenopause (50s+): Skin continues to thin at approximately 1-2% per year, compounded by the absence of estrogen. GHK-Cu levels have declined to about 80 ng/mL (60% reduction from age 20). Multi-layered peptide protocols (topical + oral + injectable) provide the most comprehensive support. HRT, when used, also benefits skin by restoring some estrogenic collagen stimulation, and the combination of HRT plus peptide skincare can produce additive benefits.
The role of androgens in women's skin: While women produce far less testosterone than men, androgens still significantly influence skin. Androgens drive sebum production (contributing to adult female acne), stimulate hair growth in androgen-sensitive areas (upper lip, chin), and suppress scalp hair growth (female pattern hair loss). PCOS, characterized by androgen excess, creates skin challenges that peptides can partially address - GHK-Cu's anti-inflammatory properties may help with acne-prone skin, while its hair-promoting effects on the scalp contrast with androgen-driven terminal hair growth elsewhere.
Combining Peptides with Conventional Skincare
Women often ask how peptide skincare fits alongside their existing skincare routines. General guidance:
- Retinoids + GHK-Cu: Can be used together but apply to slightly different areas or alternate AM/PM to avoid irritation. Both stimulate collagen, and the combination may be complementary. GHK-Cu's anti-inflammatory properties may actually help mitigate retinoid irritation.
- Vitamin C serums + GHK-Cu: Both support collagen synthesis through different mechanisms. Apply vitamin C in the morning (for antioxidant UV protection) and GHK-Cu in the evening.
- Hyaluronic acid + collagen peptides: Complementary. HA hydrates from the outside (topical) while collagen peptides support structural protein synthesis from the inside (oral).
- Chemical exfoliants (AHA/BHA) + GHK-Cu: Exfoliants remove the surface layer, potentially improving GHK-Cu penetration. Apply exfoliant first, wait 20-30 minutes, then apply GHK-Cu.
- Sunscreen: Non-negotiable regardless of peptide use. UV damage is the primary driver of extrinsic skin aging, and no amount of peptide therapy can compensate for unprotected sun exposure.
Figure 7: Layered peptide protocol for skin and hair health in women over 40
Bone Health
Osteoporosis disproportionately affects women - 80% of Americans with osteoporosis are female. The rapid bone loss that occurs in the 5-7 years following menopause creates a window of vulnerability where peptide-based interventions can make a meaningful difference.
The Postmenopausal Bone Loss Problem
Women lose 2-3% of bone mineral density per year during the first 5-7 years after menopause, compared to roughly 0.5-1% per year in age-matched men. This accelerated loss is driven by estrogen withdrawal, which removes estrogen's tonic inhibition of osteoclast activity. By age 80, the average woman has lost 30-40% of her peak bone mass.
Traditional approaches include bisphosphonates, denosumab, and HRT. Peptide therapies offer complementary strategies that address different aspects of bone biology.
Collagen Peptides and Bone Mineral Density
The strongest peptide evidence for bone health comes from collagen peptide supplementation. A landmark randomized, placebo-controlled, double-blind trial enrolled 131 postmenopausal women with reduced BMD and randomized them to receive 5 g of specific collagen peptides (FORTIBONE) or placebo daily for 12 months (Konig et al., 2018, doi:10.3390/nu10010097).
Key findings:
- Significant increase in BMD at the femoral neck and lumbar spine in the collagen group versus placebo
- Significant increase in the bone formation marker P1NP
- Significant decrease in the bone resorption marker CTX
- No adverse events reported - the intervention was safe and well-tolerated
A long-term follow-up observation over 4 years confirmed sustained bone density improvements in women who continued supplementation, with further deterioration in those who discontinued.
A 2025 meta-analysis examining collagen peptide supplementation's effects on bone health confirmed moderate but clinically relevant improvements, with standardized mean differences in BMD of 0.40-0.58 across trials (doi:10.3389/fnut.2025.1646090).
Oxytocin and Bone Remodeling
Oxytocin's role in bone health goes beyond its well-known reproductive and social functions. Osteoblasts and osteoclasts both express oxytocin receptors, making bone a direct target tissue for this peptide. The OPUS cohort study found that circulating oxytocin levels were independently associated with bone mineral density in postmenopausal women (Breuil et al., 2014, doi:10.1007/s00198-013-2614-z).
In ovariectomized mice (a model of postmenopausal osteoporosis), oxytocin administration normalized bone loss by restoring the balance between bone formation and resorption (Tamma et al., 2009). While human therapeutic trials are still early, the mechanistic rationale for oxytocin as an adjunctive therapy for postmenopausal osteoporosis is strong.
Growth Hormone Peptides and Bone
GH and IGF-1 are essential for bone formation and maintenance. The GH decline that accelerates around menopause contributes to the overall loss of bone-forming capacity. GH secretagogues like CJC-1295/Ipamorelin can restore physiological GH pulsatility, which supports osteoblast activity and collagen synthesis within bone matrix.
Clinical studies of GH replacement in GH-deficient adults show significant improvements in BMD over 12-24 months, particularly at the lumbar spine. While these studies used recombinant GH rather than secretagogues, the downstream effects of physiological GH restoration on bone would be expected to be similar.
BPC-157 and Bone Healing
BPC-157 has demonstrated bone-healing properties in animal models. Studies have shown accelerated fracture healing, enhanced bone-tendon junction repair, and improved bone density at healing sites. For postmenopausal women who sustain fractures, BPC-157 may support faster recovery, though human clinical trials specifically in postmenopausal bone healing have not been published.
The Bone-Muscle Connection: Why Peptides Matter
Bone and muscle health are deeply interconnected in postmenopausal women. Muscles generate mechanical loading that stimulates bone formation (mechanotransduction), and muscle-derived myokines signal directly to bone cells. As both muscle mass and bone density decline after menopause, a vicious cycle emerges: weaker muscles produce less mechanical stimulus for bone, leading to further bone loss, leading to fracture risk, leading to reduced activity, leading to further muscle and bone loss.
Peptide therapies can interrupt this cycle at multiple points. GH secretagogues support both muscle protein synthesis and bone collagen formation. Collagen peptides provide the structural building blocks for both bone matrix and tendon/ligament tissue. BPC-157 enhances tendon and bone healing at injury sites. And GLP-1 agonists, by promoting fat loss while preserving a reasonable amount of lean mass (especially when combined with resistance training), can improve the muscle-to-fat ratio that determines functional capacity.
For postmenopausal women, the combination of weight-bearing exercise, adequate protein and calcium intake, vitamin D optimization, and targeted peptide support creates a comprehensive approach to the musculoskeletal decline that defines much of the aging experience. No single peptide addresses all aspects - but a thoughtful combination can be more effective than any individual intervention.
Comparing Bone-Protective Interventions
| Intervention | Mechanism | Evidence Level in Postmenopausal Women | Peptide Category |
|---|---|---|---|
| Collagen peptides (5g/day oral) | Provides collagen building blocks; stimulates osteoblast differentiation | Strong - RCTs in postmenopausal women showing BMD improvement | Structural peptide |
| CJC-1295/Ipamorelin | Restores GH-IGF-1 axis; IGF-1 stimulates osteoblast activity | Moderate - GH replacement data; secretagogue-specific bone data limited | Growth hormone secretagogue |
| Oxytocin | Direct anabolic effect on bone; stimulates osteoblasts, inhibits osteoclasts | Emerging - strong animal data; human observational data; no RCTs yet | Neurohormone peptide |
| BPC-157 | Accelerates fracture healing; enhances bone-tendon junction repair | Preclinical only - animal fracture models | Healing peptide |
| GHK-Cu | Stimulates mesenchymal stem cell differentiation toward osteoblast lineage | Preclinical - in vitro osteogenic differentiation data | Regenerative peptide |
Integrated Bone Health Protocol for Postmenopausal Women
Peptide-Supported Bone Health Strategy
No peptide therapy replaces the fundamentals: adequate calcium (1,200 mg/day), vitamin D3 (2,000-5,000 IU/day, titrated to 40-60 ng/mL serum 25-OH vitamin D), weight-bearing exercise, and fall prevention. Peptides should be layered on top of these basics.
- Foundation: Collagen peptides 5 g daily (best evidence for BMD in postmenopausal women)
- GH support: CJC-1295/Ipamorelin at conservative doses to restore GH-IGF-1 axis
- Consider: Oxytocin (intranasal 12-24 IU daily) as evidence matures
- If fractured: BPC-157 locally to support healing (under provider supervision)
- Monitor: DEXA scan at baseline and annually; bone markers (P1NP, CTX) every 6 months
Autoimmune Considerations
Autoimmune diseases affect women at roughly 2-3 times the rate of men. Conditions like Hashimoto's thyroiditis, rheumatoid arthritis, lupus, Sjogren's syndrome, and multiple sclerosis are far more common in women, and peptide therapy must account for the immune-modulating potential of these compounds.
BPC-157 and Autoimmunity: Proceed with Caution
BPC-157's anti-inflammatory and tissue-healing effects make it appealing for autoimmune-related tissue damage. However, its immune-modulating properties cut both ways. BPC-157 influences multiple inflammatory pathways, including nitric oxide signaling, prostaglandin production, and cytokine networks. In autoimmune conditions where the immune system is already dysregulated, adding another immune modulator carries unpredictable risk.
Current clinical guidance suggests caution for women with:
- Active autoimmune flares
- Conditions with vascular involvement (vasculitis, antiphospholipid syndrome)
- Cancers or precancerous conditions (BPC-157's angiogenic properties could theoretically promote tumor vascularization)
- Conditions where enhanced immune function could worsen symptoms
GLP-1 Agonists and Inflammatory Markers
GLP-1 receptor agonists have demonstrated anti-inflammatory effects that may benefit women with chronic inflammatory and autoimmune conditions. Semaglutide reduces C-reactive protein, IL-6, and other inflammatory markers independently of weight loss. For women with conditions like rheumatoid arthritis or inflammatory bowel disease who also need metabolic support, GLP-1 agonists may offer dual benefits.
However, the GI side effects of GLP-1 agonists can be problematic for women with pre-existing GI autoimmune conditions. Women with Crohn's disease, ulcerative colitis, or gastroparesis should be started at the lowest doses with careful monitoring.
GHK-Cu: Immune Modulation and Anti-Inflammation
GHK-Cu has well-documented anti-inflammatory properties. Gene expression studies show it downregulates inflammatory cytokines and upregulates anti-inflammatory mediators. For women with autoimmune-driven skin conditions (psoriasis, eczema, scleroderma), topical GHK-Cu may provide local anti-inflammatory and tissue-repair benefits without the systemic immune suppression of corticosteroids.
NAD+ and Immune Function
NAD+ plays a central role in immune cell function. The PARP enzymes (which consume NAD+) are critical for DNA repair after immune-mediated tissue damage, and sirtuins (which also require NAD+) regulate inflammatory gene expression. For women with autoimmune conditions, NAD+ restoration could theoretically support tissue repair while modulating the overactive immune response - though clinical evidence is still preliminary.
Key Principles for Women with Autoimmune Conditions
- Always inform your prescribing provider about autoimmune conditions before starting any peptide therapy
- Start at lower doses and titrate more slowly than standard protocols
- Monitor inflammatory markers (CRP, ESR, disease-specific markers) during peptide treatment
- Avoid peptide therapy during active autoimmune flares
- Be especially cautious with peptides that have angiogenic properties (BPC-157) in conditions with vascular components
- Consider topical over systemic administration when possible to minimize immune interactions
- Coordinate peptide therapy with your rheumatologist or immunologist
Condition-Specific Considerations
Hashimoto's thyroiditis: The most common autoimmune condition in women, Hashimoto's affects approximately 5% of women. Since thyroid function directly influences metabolic rate, body composition, and energy levels, Hashimoto's patients often seek peptide therapies for weight management and fatigue. GLP-1 agonists appear safe in Hashimoto's (the thyroid C-cell concern relates to medullary thyroid carcinoma, not Hashimoto's). GH secretagogues require careful thyroid monitoring because GH alters T4-to-T3 conversion. NAD+ supplementation may support cellular energy production independently of thyroid hormone levels.
Rheumatoid arthritis (RA): RA affects women 2-3 times more often than men and is a common reason women seek healing peptides. BPC-157's anti-inflammatory properties are theoretically appealing for RA, but its immune-modulating effects could interact unpredictably with RA pathophysiology or with immunosuppressive medications (methotrexate, biologics). Women on RA treatment who wish to try BPC-157 should do so only under rheumatologist supervision, with regular monitoring of disease activity markers (CRP, ESR, anti-CCP, joint assessments).
Systemic lupus erythematosus (SLE): Lupus predominantly affects women of reproductive age and creates unique peptide therapy challenges. Many lupus patients are on immunosuppressants that could interact with immune-modulating peptides. Lupus patients with antiphospholipid syndrome face elevated thrombotic risk, making any peptide with vascular effects (BPC-157's angiogenic properties, oxytocin's cardiovascular effects) a consideration that requires specialist input. GLP-1 agonists may be beneficial for lupus patients with metabolic complications, and their anti-inflammatory effects could theoretically complement disease management.
Multiple sclerosis (MS): MS shows a 3:1 female-to-male ratio and often presents during the reproductive years. Peptide therapy in MS patients requires particular attention to blood-brain barrier considerations. Intranasal peptides (oxytocin, NAD+) deliver compounds near the CNS and should be used cautiously in MS patients. GLP-1 agonists have shown neuroprotective properties in preclinical MS models, and clinical interest in their potential as adjunctive MS therapy is growing.
Inflammatory bowel disease (IBD): While IBD affects men and women roughly equally, women with IBD face unique challenges around menstrual symptom exacerbation, fertility planning, and pregnancy management. BPC-157 has the most directly relevant mechanism for IBD - it was originally isolated from gastric juice and has demonstrated protective effects on GI mucosa in animal models. However, its use in human IBD patients remains experimental, and women with IBD should discuss any peptide use with their gastroenterologist. GLP-1 agonists can worsen GI symptoms in IBD patients and should be used very cautiously.
The Estrogen-Autoimmunity Connection and Peptide Implications
The higher prevalence of autoimmune disease in women isn't coincidental - estrogen is a potent immunomodulator that promotes B cell activation and antibody production while having complex effects on T cell subsets. This creates an important context for peptide therapy:
- During the follicular phase (rising estrogen), immune responses may be heightened, potentially increasing autoimmune disease activity and altering responses to immune-modulating peptides
- During the luteal phase (progesterone-dominant), the relative immunosuppression may reduce disease activity but could also alter healing peptide efficacy
- After menopause, the loss of estrogen's immune stimulation often - but not always - reduces autoimmune disease activity. This can paradoxically simplify peptide therapy by removing the hormonal variability that complicates dosing during reproductive years
- HRT in women with autoimmune conditions adds another layer of complexity, as exogenous estrogen can reactivate or worsen certain autoimmune conditions while improving symptoms in others
Cycle-Based Timing
The menstrual cycle creates a monthly rhythm that influences how women respond to peptide therapy. While the clinical differences between cycle phases are modest for most peptides, understanding this rhythm can help optimize timing for maximum benefit and minimum side effects.
Understanding the Hormonal Phases
The menstrual cycle consists of four distinct hormonal phases, each with different implications for peptide therapy:
Menstruation (Days 1-5): Low estrogen, low progesterone. Inflammation markers are typically elevated. This is a neutral window for most peptide therapies. BPC-157 or other healing peptides can be started during this phase without concern for endometrial effects.
Follicular Phase (Days 6-13): Rising estrogen. Gastric motility increases, CYP3A4 activity rises, and mood is generally at its best. This is often the best phase for starting new peptide therapies or dose increases, as side effect tolerance tends to be highest. GLP-1 agonists are often best tolerated when titrated during this phase.
Ovulation (Day 14, approximately): Estrogen peaks, LH surges. Women using fertility peptides (kisspeptin, gonadorelin) time their administration relative to this event. For non-fertility peptides, ovulation itself doesn't significantly impact efficacy or tolerability.
Luteal Phase (Days 15-28): Rising progesterone, declining estrogen in the late luteal phase. Gastric emptying slows (relevant for oral semaglutide), water retention increases, and mood can be more variable. GI side effects from GLP-1 agonists may be more pronounced during this phase. Some women report increased injection site reactions.
Practical Cycle-Based Timing Guide
| Peptide | Optimal Initiation Phase | Phase to Monitor Closely | Rationale |
|---|---|---|---|
| GLP-1 agonists | Follicular (Days 6-13) | Luteal (Days 15-28) | Better GI tolerance in follicular phase; nausea may worsen luteal |
| CJC-1295/Ipamorelin | Any phase (nightly dosing) | Late luteal (if sleep disturbed) | GH secretion is relatively constant across cycle phases |
| BPC-157 | Menstruation or follicular | Proliferative phase (Days 8-14) | Avoid high-dose during active endometrial proliferation; precautionary |
| GHK-Cu (topical) | Any phase | None specific | Topical application unaffected by cycle phase |
| Kisspeptin-54 (IVF trigger) | Timed to follicular maturity | Per IVF monitoring protocol | Administered at specific trigger point |
| Oxytocin (intranasal) | Any phase | Premenstrual (enhanced sensitivity) | Estrogen potentiates oxytocin receptor expression |
| NAD+/NMN | Any phase | None specific | Metabolic coenzyme; no significant cycle interaction |
Perimenopause: When Cycles Become Unpredictable
During perimenopause, menstrual cycles become irregular - lengthening, shortening, or skipping entirely. Hormone levels can swing widely from cycle to cycle. This creates challenges for cycle-based peptide timing.
Practical approaches for perimenopausal women include:
- Symptom-based timing rather than calendar-based (start dose adjustments when side effects appear rather than predicting cycle phase)
- More conservative dosing across the board, since hormonal variability makes drug response less predictable
- More frequent provider check-ins during the perimenopausal transition
- Considering continuous dosing rather than cycle-synchronized protocols when cycles are unreliable
Post-Menopause: Simplified Timing
After menopause, the elimination of hormonal cycling simplifies peptide timing considerably. Without estrogen and progesterone fluctuations, pharmacokinetic variability decreases, and peptide responses become more consistent and predictable. Post-menopausal dosing can follow standard protocols without cycle-phase considerations, focusing instead on time of day (e.g., GH peptides at bedtime) and meal timing (e.g., oral semaglutide on an empty stomach).
Circadian Timing for Peptide Administration
Beyond cycle-based timing, the time of day when peptides are administered can influence efficacy. Women's circadian rhythms show some sex-specific features that affect optimal dosing windows:
Morning administration:
- Oral semaglutide (Rybelsus): must be taken on empty stomach, at least 30 minutes before first food or drink. Morning administration aligns with the recommended protocol and avoids the delayed gastric emptying that progresses throughout the day.
- NAD+ (oral NMN/NR): many practitioners recommend morning dosing to align with the body's circadian metabolic peak. NAD+ levels naturally fluctuate with circadian rhythm, peaking during active periods.
- BPC-157 for systemic use: can be taken morning and evening (split dose). Morning dosing may provide better absorption due to faster gastric emptying.
Evening/bedtime administration:
- CJC-1295/Ipamorelin: evening administration (30-60 minutes before bed) aligns with the body's natural nocturnal GH pulse, amplifying its magnitude. Food should be avoided for 2-3 hours before injection, as insulin and glucose suppress GH release.
- Epithalon: evening dosing supports its melatonin-stimulating action and aligns with pineal gland activity.
- Oxytocin (for sleep support): intranasal oxytocin taken 30 minutes before bed can improve sleep quality and reduce nighttime anxiety.
Injection-day timing for weekly GLP-1s:
- Semaglutide or tirzepatide weekly injections can be administered at any time of day, but many women prefer evening injection. This is because peak nausea tends to occur 24-48 hours after injection, and evening dosing means peak GI effects may coincide with sleep rather than daytime activities.
- Some women prefer to inject on Friday evening so that peak side effects occur over the weekend rather than during work days.
- Consistency of day and approximate time is more important than optimizing the specific hour.
Tracking Cycle and Peptide Response
Women who want to optimize their peptide therapy can benefit from tracking several parameters across their menstrual cycle:
- Cycle day: Use a period tracking app or calendar to note cycle phase when logging peptide-related symptoms
- Body weight: Expect 2-5 pounds of luteal phase water weight gain; don't adjust GLP-1 doses based on premenstrual weight increases
- GI symptoms: Note if nausea, bloating, or constipation worsens during specific cycle phases
- Sleep quality: Track how GH peptide and epithalon effects vary across the cycle
- Mood: Note any mood changes that correlate with peptide timing versus cycle phase
- Energy levels: Track whether NAD+ or GH peptide-related energy improvements fluctuate with hormonal changes
- Injection site reactions: Note any cycle-phase patterns in injection site redness, pain, or swelling
After 2-3 cycles of tracking, patterns usually emerge that allow informed dose and timing adjustments. This data is also valuable for clinician visits, providing objective information for protocol optimization.
Figure 8: Menstrual cycle-based peptide therapy timing guide showing optimal initiation and monitoring windows
Safety Considerations
Safety is always the foundation of responsible peptide therapy, and women face unique safety considerations that deserve dedicated attention. From hormone-sensitive cancers to pregnancy risk to the higher adverse event rates seen in women across drug classes, a thorough risk assessment is essential before starting any peptide protocol.
Cancer Risk Considerations
Several peptides interact with pathways that are relevant to hormone-sensitive cancers, which are more common in women:
GLP-1 agonists and thyroid cancer: The FDA boxed warning regarding thyroid C-cell tumors was based on rodent data where GLP-1 receptor agonists caused medullary thyroid carcinoma in rats. While human epidemiological data has not confirmed this risk, GLP-1 agonists remain contraindicated in patients with personal or family history of medullary thyroid carcinoma or MEN2 syndrome. Routine thyroid monitoring is recommended.
Growth hormone and breast cancer: The GH-IGF-1 axis has complex interactions with breast cancer risk. Elevated IGF-1 has been associated with increased breast cancer risk in some epidemiological studies. Women with a personal or family history of breast cancer should discuss GH peptide use carefully with their oncologist. Maintaining IGF-1 within the physiological range (not supraphysiological) is essential.
BPC-157 and angiogenesis: BPC-157's pro-angiogenic properties raise theoretical concerns about promoting blood vessel formation in existing tumors. Women with active cancer or a recent cancer history should avoid BPC-157.
Epithalon and telomerase: Epithalon's mechanism of activating telomerase raises the theoretical concern that it could support telomere maintenance in cancer cells. However, epithalon has been used in Russia for treatment of hormone-dependent tumors (suggesting an anti-tumor rather than pro-tumor effect in some contexts), and no evidence links epithalon to cancer promotion. Still, women with active malignancies should avoid telomerase-activating compounds.
Cardiovascular Safety in Women
Heart disease kills more women than all cancers combined, making cardiovascular safety paramount:
- GLP-1 agonists: Demonstrated cardiovascular benefit in the SELECT trial, with risk reduction consistent across sexes. These are among the safest peptides from a cardiovascular perspective.
- GH secretagogues: GH can transiently raise blood pressure and promote fluid retention. Women with uncontrolled hypertension or heart failure should use GH peptides cautiously with cardiac monitoring.
- Oxytocin: At physiological replacement doses, oxytocin has cardioprotective properties. At high doses (as used in labor induction), it can cause tachycardia and fluid retention. Non-obstetric doses are generally safe from a cardiac standpoint.
Mental Health Considerations
Women experience depression and anxiety at roughly twice the rate of men, and hormonal transitions (menstrual, postpartum, perimenopausal) can trigger or worsen mood disorders. Peptide therapy can interact with mental health in both positive and negative ways:
Positive effects:
- GLP-1 agonists: Some women report improved mood with weight loss; however, FDA is monitoring reports of suicidal ideation as a potential safety signal
- Oxytocin: Anxiolytic and antidepressant effects demonstrated in pilot studies
- GH secretagogues: Improved sleep quality can significantly benefit mood
- NAD+: May support brain energy metabolism and neurotransmitter function
Potential concerns:
- GLP-1 agonists: The FDA and EMA have investigated reports of suicidal thoughts and self-harm; no causal link established, but monitoring is warranted
- Rapid weight loss from any peptide therapy can trigger body image issues or disordered eating in susceptible individuals
- Hormonal peptides (kisspeptin, gonadorelin) can cause mood fluctuations as they influence the HPG axis
Drug Interaction Summary for Common Women's Medications
| Women's Medication | Peptide of Concern | Interaction | Management |
|---|---|---|---|
| Combined oral contraceptives | GLP-1 agonists | Reduced Cmax of OC components | Use non-oral contraception |
| Levothyroxine | GH secretagogues | Altered T4/T3 conversion | Check TSH 6-8 weeks after starting |
| Metformin (PCOS) | GLP-1 agonists | Additive GI side effects; complementary metabolic effects | Reduce metformin if GI intolerance; monitor B12 |
| SSRIs/SNRIs | Oxytocin | SSRIs may blunt oxytocin receptor sensitivity | May need higher oxytocin doses; monitor mood |
| Spironolactone (acne/PCOS) | GH secretagogues | Spironolactone is anti-androgenic; GH can increase androgen production | Monitor androgen levels |
| Bisphosphonates (osteoporosis) | Collagen peptides | Complementary mechanisms; no adverse interaction | Safe to combine |
| HRT (estrogen + progesterone) | GH secretagogues | Oral estrogen reduces IGF-1; transdermal does not | Use transdermal estrogen when combining with GH peptides |
Monitoring Recommendations for Women on Peptide Therapy
Baseline and ongoing monitoring should include:
- Baseline labs: CBC, CMP, fasting glucose, HbA1c, lipid panel, TSH, free T4, IGF-1, estradiol, FSH (if perimenopausal), DEXA scan (if postmenopausal)
- At 4-6 weeks: Repeat glucose, HbA1c, IGF-1 (if on GH peptides), thyroid function (if on GH peptides)
- Every 3-4 months: IGF-1, metabolic panel, thyroid function
- Annually: DEXA scan (postmenopausal), comprehensive metabolic panel, mammogram as indicated by age/risk
- Pregnancy test: Before initiating any peptide therapy in women of reproductive age; periodic testing if sexually active and using peptides contraindicated in pregnancy
For women interested in getting started with peptide therapy, our getting started guide and dosing calculator can help navigate the initial steps.
Injection Technique Considerations for Women
Women have unique anatomical considerations for subcutaneous injection that affect peptide delivery:
Injection site selection: Women typically have greater subcutaneous fat thickness at the abdomen, thighs, and upper arms compared to men. While this provides ample tissue for subcutaneous injection, it can slow absorption compared to leaner sites. Rotating injection sites is especially important for women who use multiple peptides, as each injection site should be separated by at least 2 inches and different peptides should ideally be injected in different anatomical regions.
Needle length: The standard 31-gauge, 5/16-inch (8mm) insulin syringe is appropriate for most women. Women with very low body fat (under 15%) may benefit from shorter needles to avoid intramuscular injection, while women with higher body fat may need to ensure the full needle length is inserted for proper subcutaneous delivery.
Timing and comfort: Many women find injection more comfortable during the follicular phase when pain thresholds tend to be higher. For daily injections (BPC-157, GHK-Cu), this isn't practical to optimize, but for weekly injections (GLP-1 agonists), choosing a consistent injection day during the expected follicular phase can improve the experience.
Menstrual phase and injection site reactions: Some women report more pronounced injection site redness, swelling, or pain during the premenstrual and menstrual phases, possibly related to the inflammatory surge that accompanies menstruation. If injection site reactions are bothersome, consider rotating to the upper thigh or posterior upper arm during these phases rather than the abdomen.
Long-Term Safety Monitoring Framework
Women on peptide therapy for extended periods (beyond 6 months) should follow a structured monitoring framework:
| Timeline | Monitoring Actions | Applicable Peptides |
|---|---|---|
| Baseline (before starting) | Full labs, pregnancy test, DEXA (if postmenopausal), body composition assessment | All |
| 4-6 weeks | Glucose, HbA1c, IGF-1, thyroid panel, symptom assessment | GLP-1s, GH peptides |
| 3 months | Comprehensive metabolic panel, lipid panel, IGF-1, inflammatory markers | All |
| 6 months | Full repeat labs, bone markers (P1NP, CTX if on bone protocol), body composition | All |
| 12 months | Full labs, DEXA (if on bone protocol), mammogram (age-appropriate), thyroid ultrasound (if on GLP-1) | All |
| Ongoing annually | Full labs, cancer screening per age guidelines, DEXA every 1-2 years, IGF-1 monitoring | All |
When to Stop Peptide Therapy
Clear indications for discontinuing peptide therapy in women include:
- Positive pregnancy test (immediate discontinuation of all non-fertility peptides)
- Active cancer diagnosis (discontinue all peptides until cleared by oncologist)
- Severe adverse reactions (anaphylaxis, pancreatitis, medullary thyroid cancer symptoms)
- Pre-surgical preparation (GLP-1 agonists stopped 1-3 weeks before elective surgery)
- Initiation of breastfeeding (discontinue all systemic peptides without safety data)
- Development of autoimmune flare while on immune-modulating peptides
- IGF-1 levels consistently above the age-adjusted reference range (GH peptides)
- Calcitonin elevation (GLP-1 agonists - evaluate for thyroid pathology)
- Achievement of therapeutic goals with sustained results (consider transition to maintenance)
Healing Peptides During the Menstrual Cycle
Women's tissue repair and immune function fluctuate across the menstrual cycle, creating windows where healing peptides may be more or less effective. Understanding these dynamics helps clinicians time treatment for optimal results.
Estrogen's Role in Wound Healing
Estrogen is a potent wound-healing hormone. It accelerates cutaneous wound repair by promoting keratinocyte migration, enhancing angiogenesis, regulating inflammation, and stimulating collagen deposition. This is why wound healing tends to be faster during the follicular phase (when estrogen is rising) and slower during the luteal phase and post-menopause (when estrogen is lower or declining).
The clinical implication is clear: women may experience variable healing responses to peptides like BPC-157 depending on where they are in their cycle. A healing peptide started during the follicular phase may produce faster results than the same peptide started during the late luteal phase.
BPC-157 and Menstrual Cycle Interactions
BPC-157 promotes tissue repair through angiogenesis, growth factor modulation, and anti-inflammatory signaling. These mechanisms overlap with and potentially complement estrogen's own healing effects. However, they also raise unique considerations:
Endometrial safety: The endometrium undergoes cyclical growth, breakdown, and repair under hormonal control. BPC-157's angiogenic properties could theoretically influence endometrial vascularization. No studies have examined this, but clinicians often recommend conservative dosing during the proliferative phase (days 8-14) as a precaution.
Menstrual flow: Some women report changes in menstrual flow while using BPC-157 - either heavier or lighter bleeding. These reports are anecdotal and may reflect BPC-157's effects on local vasculature or may be coincidental. If significant menstrual changes occur, consultation with a gynecologist is warranted.
GHK-Cu and Hormonal Skin Changes
GHK-Cu is frequently used topically for skin rejuvenation, and its effects may vary across the menstrual cycle. Skin is an estrogen-responsive organ - collagen content, skin thickness, hydration, and sebum production all fluctuate with hormonal changes:
- Follicular phase: Rising estrogen increases skin hydration and collagen synthesis. GHK-Cu applied during this phase may work in concert with estrogen's effects.
- Ovulation: Skin tends to look its best (thickest, most hydrated). GHK-Cu continues to complement the hormonal environment.
- Luteal phase: Rising progesterone increases sebum production and can worsen acne. GHK-Cu's anti-inflammatory properties may be particularly beneficial during this phase.
- Menstruation: Skin is at its most sensitive with reduced barrier function. Gentle application with lower concentrations may be better tolerated.
Recovery from Exercise Across the Cycle
Women's exercise recovery also varies with cycle phase, which affects the therapeutic context for healing peptides. Muscle damage and recovery follow different patterns:
- The follicular phase is associated with better exercise tolerance, faster recovery, and higher pain thresholds
- The luteal phase shows increased markers of muscle damage, longer recovery times, and greater subjective fatigue
- Women using BPC-157 or GH peptides for exercise recovery may notice more pronounced benefits when training or recovering during the follicular phase
These differences, while clinically real, are modest enough that they shouldn't dictate training schedules. Instead, they provide context for understanding variable recovery experiences and adjusting expectations across the cycle.
Pelvic Floor and Postpartum Healing
One uniquely female application of healing peptides involves pelvic floor recovery after childbirth. Vaginal delivery and pregnancy itself create significant stress on the pelvic floor muscles, ligaments, and connective tissue. While formal clinical data is absent, the mechanistic rationale for healing peptides in postpartum pelvic floor recovery is strong:
BPC-157: Its tendon and ligament healing properties could theoretically support pelvic floor connective tissue repair. However, its use during breastfeeding is not recommended due to insufficient safety data. Women who are not breastfeeding might consider BPC-157 as part of postpartum recovery, but only under medical supervision and after waiting at least 6 weeks postpartum.
Collagen peptides: Oral collagen supplementation (5-10 g daily) is the safest option for postpartum connective tissue support. Collagen peptides have a strong safety profile and provide the structural amino acids (glycine, proline, hydroxyproline) needed for tissue repair. These can be safely used during breastfeeding based on the available evidence, as they're essentially food-derived nutrients.
GHK-Cu: Topical application to surgical sites (cesarean incisions, episiotomy repairs) may support wound healing and minimize scarring. The safety of topical GHK-Cu during breastfeeding is presumed to be low-risk given minimal systemic absorption from topical use, but it should not be applied to nipples or breast tissue.
Endometriosis and Peptide Therapy
Endometriosis affects approximately 10% of reproductive-age women and involves the growth of endometrial-like tissue outside the uterus, driven by estrogen and characterized by chronic inflammation, pain, and sometimes infertility. Peptide therapy in endometriosis patients requires careful consideration:
- GLP-1 agonists may benefit endometriosis patients with co-existing insulin resistance or obesity, and their anti-inflammatory effects could theoretically reduce endometriosis-related inflammation
- BPC-157's angiogenic properties are a concern in endometriosis, where abnormal blood vessel formation supports ectopic endometrial implant survival. Women with active endometriosis should generally avoid BPC-157
- GnRH agonist protocols (using continuous gonadorelin or synthetic GnRH analogs) are a standard endometriosis treatment, creating a hypoestrogenic state that suppresses endometrial growth. This is a therapeutic use of the pulsatility principle - continuous GnRH desensitizes the pituitary rather than stimulating it
- GH secretagogues appear neutral regarding endometriosis, but should be used with monitoring in women with known disease
Surgical Recovery and Peptide Support for Women
Women undergo several sex-specific surgeries (hysterectomy, oophorectomy, breast surgery, cesarean section) where healing peptides could play a role. General principles for peptide use around surgery include:
- Discontinue GLP-1 agonists 1-3 weeks before elective surgery (due to aspiration risk from delayed gastric emptying - current anesthesia society guidelines vary on the exact timeframe)
- BPC-157 may be started after surgery to support tissue healing (typically waiting until drain tubes are removed and initial wound closure is confirmed)
- GHK-Cu can be applied topically to surgical scars once initial wound closure is complete (usually 2-4 weeks post-surgery)
- GH secretagogues support post-surgical recovery through enhanced protein synthesis and tissue repair but should be discussed with the surgical team
- Resume GLP-1 agonists only after full return to normal diet and GI function post-surgery
The Future of Female Peptide Medicine
The field of peptide therapeutics is evolving rapidly, with several developments on the horizon that will particularly impact women's health. From next-generation GLP-1 compounds to novel fertility peptides, the pipeline holds considerable promise.
Next-Generation Weight Management Peptides
Beyond semaglutide and tirzepatide, a new wave of multi-agonist peptides is entering clinical trials. Retatrutide, a triple agonist targeting GLP-1, GIP, and glucagon receptors, has produced weight loss exceeding 24% in Phase II trials. Survodutide, a dual GLP-1/glucagon agonist, has shown substantial efficacy for both weight loss and MASLD (metabolic-associated steatotic liver disease). Orforglipron, the first oral non-peptide GLP-1 receptor agonist, could eliminate the injection barrier entirely.
For women, these next-generation compounds raise important questions that clinical trials must address:
- Will the sex-based weight loss advantage seen with current GLP-1 agonists persist with triple agonists?
- How do these compounds interact with female reproductive hormones and oral contraceptives?
- What are the pregnancy safety profiles, and will washout periods differ?
- Will the higher efficacy compounds create greater risk of excessive lean mass loss in women?
- Are GI side effect rates and severity patterns different in women with these more potent compounds?
Early data suggests that the sex-based patterns observed with current GLP-1 agonists (greater female weight loss, higher female GI side effects) are likely to persist with next-generation compounds, making sex-specific dosing research even more important.
Emerging Fertility Peptides
MVT-602 (TAK-448), the long-acting kisspeptin receptor agonist, represents the next evolution of kisspeptin-based fertility therapy. With a 21-22 hour duration of action compared to KP54's 4.7 hours, MVT-602 offers more practical single-dose administration for IVF triggering. Clinical trials are evaluating its efficacy and safety profile, with particular attention to OHSS risk and pregnancy rates.
Other fertility-relevant developments include:
- GnRH receptor modulators: New GnRH antagonists with improved pharmacokinetic profiles are being developed for endometriosis, uterine fibroids, and IVF protocols. These peptide-based compounds offer more precise control of the reproductive axis than older medications.
- AMH (anti-Mullerian hormone) peptides: Research into therapeutic modulation of AMH signaling could lead to interventions that slow ovarian aging or protect the follicle pool during chemotherapy.
- Mitochondrial-targeted peptides: Compounds like SS-31 (elamipretide) that specifically target mitochondrial function are being investigated for oocyte quality improvement, potentially offering more targeted fertility support than general NAD+ supplementation.
Personalized Peptide Protocols
The future of women's peptide therapy will likely involve increasingly personalized protocols based on:
Pharmacogenomics: Genetic variations in drug-metabolizing enzymes (CYP polymorphisms), receptor expression levels, and signaling pathway components will eventually allow clinicians to predict which women will respond best to specific peptides and what doses they'll need. For example, GLP-1 receptor gene variants may explain why some women lose 20% of their body weight on semaglutide while others lose only 5%.
Biomarker-guided dosing: Rather than following fixed titration schedules, future protocols may adjust doses based on real-time biomarker feedback - blood glucose patterns from continuous monitors, IGF-1 levels from at-home testing, or even wearable-derived sleep quality data driving GH peptide dose adjustments.
Hormonal phase-synchronized delivery: Smart delivery systems that adjust peptide release based on cycle phase (detected through hormone biosensors) could optimize both efficacy and tolerability, delivering more drug during the follicular phase and less during the luteal phase for compounds with phase-dependent side effects.
Microbiome-informed protocols: Emerging research on the gut microbiome's influence on peptide metabolism may lead to protocols that include probiotic or prebiotic co-therapy to optimize peptide absorption and reduce GI side effects.
What Women Should Watch For
Women interested in staying current with peptide therapy developments should pay attention to:
- Clinical trial results that specifically report sex-stratified outcomes (rather than lumping all participants together)
- FDA reproductive safety determinations for new peptide compounds
- Compounding pharmacy regulations that affect peptide availability
- Long-term safety data (5-10 year follow-up) for GLP-1 agonists, which are now being used by millions of women
- Research on peptide interactions with HRT and common women's medications
- Development of oral and intranasal peptide delivery systems that reduce the injection burden
The most important development may be cultural rather than scientific: the growing recognition that women's health requires dedicated research rather than extrapolation from male-dominant studies. As sex-specific pharmacology gains mainstream acceptance, peptide protocols tailored specifically for women will become the standard rather than the exception.
The Role of Artificial Intelligence in Women's Peptide Therapy
Machine learning and AI tools are beginning to enter clinical practice in ways relevant to peptide therapy optimization. Potential applications include:
- Predictive dosing algorithms: AI models trained on demographic, hormonal, genetic, and response data could predict optimal starting doses for women based on their individual characteristics rather than population averages. Such models could account for menstrual cycle phase, BMI, age, hormonal status, and concurrent medications simultaneously - something impractical for human clinicians to do manually.
- Side effect prediction: Machine learning could identify patterns in adverse event data that predict which women are most likely to experience GI side effects on GLP-1 agonists, allowing for preemptive management strategies or alternative compound selection.
- Protocol optimization: AI-driven analysis of outcomes across thousands of women's peptide protocols could identify optimal combinations, timing strategies, and dose ranges far faster than traditional clinical trials.
- Cycle-aware monitoring: Wearable devices that track temperature, heart rate variability, and other biomarkers can identify menstrual cycle phase in real time. Combined with AI analysis, this data could generate personalized recommendations for peptide dose adjustments based on current hormonal status.
These technologies are still in their early stages, but the convergence of wearable health monitoring, electronic health records, and machine learning creates a foundation for truly personalized women's peptide medicine within the next decade.
Longevity & Anti-Aging Peptides for Women
Women live longer than men on average but spend more years in poor health. The female longevity advantage - roughly 5-7 extra years of life expectancy - comes with a paradox: women experience more years of disability, chronic disease, and diminished quality of life. Longevity peptides aim to close this gap between lifespan and healthspan.
The Female Aging Trajectory
Women's aging differs from men's in several biologically important ways that create distinct opportunities for peptide intervention:
Accelerated hormonal aging: While men experience a gradual decline in testosterone over decades, women face an abrupt hormonal cliff at menopause. This sharp transition accelerates multiple aging processes simultaneously - bone loss, cardiovascular risk, metabolic decline, cognitive changes, and skin aging all intensify within a 5-10 year window around menopause. Peptide therapy during this critical window has the potential for outsized benefit.
Telomere biology: Women generally have longer telomeres than men at birth, and this telomere advantage persists throughout life, likely contributing to the female longevity advantage. However, the rate of telomere shortening accelerates during periods of physiological stress - pregnancy, chronic caregiving, sleep deprivation, and psychosocial stress - all of which disproportionately affect women. Epithalon, which activates telomerase to maintain telomere length, may be particularly relevant for women experiencing accelerated telomere attrition from chronic stress.
Immune aging: Women's immune systems age differently than men's. The stronger female immune response (which protects against infections and cancer) comes at the cost of higher autoimmune susceptibility. As the immune system ages (immunosenescence), women may lose their immune advantage faster, potentially explaining why the female longevity advantage narrows at very advanced ages. NAD+ restoration supports immune cell function and may help maintain immune competence during aging.
Epithalon: Telomeres and Melatonin
Epithalon targets two aging mechanisms particularly relevant to women:
Telomere maintenance: Clinical studies have shown significant telomere elongation in blood cells of patients aged 60-80 following epithalon administration (Khavinson et al., 2003, doi:10.1023/A:1025493705728). A 2025 in vitro study confirmed that epithalon increases telomere length in human cell lines through telomerase upregulation. For women, whose telomere length is correlated with reproductive history (parity, age at menopause), maintaining telomere integrity may support healthier aging trajectories.
Melatonin restoration: Melatonin production by the pineal gland declines with age, and this decline accelerates around menopause. Melatonin isn't just a sleep hormone - it's a powerful antioxidant, immune modulator, and circadian rhythm regulator. Epithalon's ability to stimulate endogenous melatonin production offers advantages over exogenous melatonin supplementation: it restores the natural circadian pattern of melatonin secretion rather than providing a single pharmacological bolus.
Typical epithalon protocols for women:
- Injectable: 5-10 mg daily for 10-20 days, repeated 2-3 times per year
- Often combined with evening CJC-1295/Ipamorelin for complementary sleep and anti-aging benefits
- Monitor sleep quality, skin changes, and energy levels as subjective markers
- Consider telomere length testing at baseline and after 6-12 months of cyclical treatment
NAD+ as a Female Longevity Strategy
NAD+ occupies a unique position in women's longevity because it addresses multiple hallmarks of aging simultaneously while also having direct implications for reproductive aging:
Mitochondrial function: NAD+ is essential for mitochondrial oxidative phosphorylation - the primary source of cellular energy. As NAD+ declines with age, mitochondrial function deteriorates, leading to cellular energy deficits that manifest as fatigue, cognitive slowing, and reduced tissue repair capacity. Women may be particularly sensitive to mitochondrial decline because of the energy-intensive nature of their reproductive organs - ovaries and uterine tissue have high mitochondrial content.
Sirtuin activation: The SIRT1-SIRT7 family of proteins depends on NAD+ for activity. Sirtuins regulate metabolic efficiency, DNA repair, inflammation, and stress responses. SIRT1 in particular has been linked to longevity in multiple model organisms. For women, SIRT1 also influences estrogen receptor signaling, potentially modulating how cells respond to declining estrogen levels during menopause.
DNA repair: PARP enzymes consume NAD+ to repair DNA damage. As NAD+ becomes scarce with age, DNA repair capacity diminishes, leading to genomic instability - a hallmark of aging and cancer. Women who have undergone radiation therapy (for breast cancer, for example) may benefit from NAD+ restoration to support DNA repair in irradiated tissues.
Inflammaging control: Chronic low-grade inflammation increases with age and accelerates after menopause. NAD+ supports anti-inflammatory signaling through sirtuin activation. The CD38 enzyme, which consumes NAD+ and increases with age, is itself a driver of inflammation. Restoring NAD+ levels may help control the inflammatory escalation that characterizes postmenopausal aging.
The Integrated Female Longevity Protocol
A comprehensive longevity-oriented peptide protocol for women in their 40s-60s might include the following tiers, introduced sequentially rather than simultaneously:
Tier 1 - Foundation (start here):
- Collagen peptides 5-10 g oral daily (bone, skin, and joint support)
- NAD+ precursor (NMN 250-500 mg or NR 300 mg oral daily)
- Focus on establishing baseline labs and tracking subjective response
Tier 2 - Hormonal support (add after 1-2 months if tolerated):
- CJC-1295/Ipamorelin nightly injection (GH restoration for sleep, body composition, skin)
- Monitor IGF-1 levels; adjust dose to maintain upper quartile of age-adjusted range
Tier 3 - Targeted interventions (add based on individual needs):
- GLP-1 agonist if metabolic optimization or weight management is needed
- GHK-Cu topical and/or injectable for skin and hair rejuvenation
- Oxytocin intranasal for mood, sleep, and bone support
- BPC-157 as needed for injury repair (episodic rather than continuous)
Tier 4 - Advanced longevity (for committed long-term users):
- Epithalon cyclical protocols (10-20 day cycles, 2-3 times per year)
- NAD+ SubQ or IV loading periodically in addition to oral NMN maintenance
- Comprehensive biomarker tracking including telomere length, epigenetic age clocks, inflammatory panels
The Principle of Sequential Introduction
Never start multiple peptides simultaneously. Introduce one new compound every 4-6 weeks. This allows you to identify which peptide is responsible for any benefits or side effects, establish the minimal effective dose for each compound, and avoid overwhelming the body's adaptive responses. For women, this sequential approach is especially important because hormonal fluctuations add variability that can obscure peptide-specific effects if multiple compounds are changed at once.
Figure 9: Sequential longevity peptide protocol tiers for women ages 40-60+
Cognitive Health and Peptides for Women
Women face roughly twice the risk of Alzheimer's disease compared to men, and the cognitive changes that begin during perimenopause - often dismissed as "brain fog" - may represent an early window where interventions could alter the trajectory of cognitive aging.
NAD+ and brain health: The brain consumes approximately 20% of the body's oxygen and has correspondingly high NAD+ requirements. NAD+ depletion in the brain has been linked to neurodegeneration, and preclinical studies with NAD+ precursors show neuroprotective effects. For women experiencing perimenopausal cognitive symptoms, NAD+ restoration may support brain energy metabolism independently of estrogen replacement.
Oxytocin and neuroprotection: Oxytocin receptors are widely distributed in the brain, including in regions critical for memory (hippocampus) and emotional regulation (amygdala). Intranasal oxytocin delivery can reach the CNS through nose-to-brain transport, bypassing the blood-brain barrier. While clinical evidence for oxytocin's cognitive benefits is still preliminary, the mechanistic rationale for neuroprotective effects is strong.
GH peptides and cognition: Growth hormone deficiency is associated with cognitive impairment, and GH replacement has been shown to improve cognitive function in GH-deficient adults. For postmenopausal women with declining GH, secretagogues like CJC-1295/Ipamorelin may support cognitive function through restored GH-IGF-1 signaling, improved sleep architecture (sleep is critical for memory consolidation), and enhanced cerebrovascular health.
GHK-Cu and neuroprotection: Gene expression analysis has identified several neuroprotective gene targets modulated by GHK-Cu, including genes involved in antioxidant defense, DNA repair, and anti-inflammatory signaling. While GHK-Cu is primarily used for skin applications, its systemic administration may have neuroprotective properties worth exploring as research progresses.
Emotional Well-Being and Peptide Therapy
The psychological dimension of peptide therapy in women deserves dedicated attention. Women's emotional health is intertwined with hormonal status in ways that affect both the decision to start peptide therapy and the experience of being on it.
The body image dimension: Weight-focused peptide therapy (GLP-1 agonists) intersects with a complex cultural landscape around women's bodies. While achieving a healthier body weight provides genuine metabolic and cardiovascular benefits, the social pressure women face around thinness can complicate the therapeutic relationship. Clinicians should assess whether the primary motivation for GLP-1 therapy is metabolic health or appearance-driven, as the latter can indicate underlying body image issues that peptide therapy alone won't resolve. A thoughtful provider ensures that weight loss medication is part of a whole-body approach that includes mental health support when needed.
Hormonal mood vulnerability: Women are neurobiologically more susceptible to mood disturbances during hormonal transitions - puberty, the premenstrual phase, postpartum, and menopause. Peptide therapy that interacts with hormonal axes (GLP-1 agonists affecting GI-brain signaling, GH peptides affecting somatotropic axis, kisspeptin and gonadorelin affecting the HPG axis) can subtly shift mood in ways that overlap with hormonal mood vulnerability. Screening for depression and anxiety before starting peptide therapy and monitoring mood during treatment is good clinical practice.
The identity of chronic medication use: For women who've never taken regular medications, the transition to daily or weekly injections can feel identity-challenging. Peptide therapy often involves reconstituting lyophilized powders, storing vials in the refrigerator, and performing self-injections - activities that feel medical and clinical. Supporting women through this transition with clear education, hands-on injection training, and normalization of the experience improves adherence and outcomes.
Relationship dynamics: Peptide-mediated changes - weight loss, improved energy, enhanced libido (from GH or testosterone-supporting peptides), better sleep - can shift relationship dynamics. Partners may respond positively or may feel threatened by the changes. While this is beyond the scope of pharmacological discussion, acknowledging the relational impact of health optimization is part of patient-centered care.
Oxytocin and emotional connection: Oxytocin is unique among therapeutic peptides in its direct effects on emotional and social functioning. Intranasal oxytocin has been studied for its ability to enhance trust, empathy, emotional recognition, and pair bonding. For women experiencing the emotional flatness that sometimes accompanies menopause or long-term SSRI use, oxytocin may offer a pathway to restored emotional vitality. However, expectations should be calibrated - oxytocin is not a "love drug" or a cure for relationship problems. Its therapeutic value lies in modest but meaningful improvements in emotional responsiveness and social engagement.
Sleep and Peptides: A Female-Specific Perspective
Sleep disturbance affects women more than men at nearly every life stage, and several peptides directly address sleep quality:
Perimenopausal insomnia: Hot flashes and night sweats are the most common drivers of menopausal sleep disruption, affecting 60-80% of women during the transition. While peptides don't directly treat vasomotor symptoms (HRT remains the gold standard), several peptides improve sleep through different mechanisms. CJC-1295/Ipamorelin enhances slow-wave sleep by augmenting the natural nocturnal GH pulse. Epithalon restores endogenous melatonin production. Oxytocin promotes relaxation and reduces nighttime anxiety. Used individually or in combination, these peptides can meaningfully improve sleep architecture even when hot flashes persist.
Stress-related insomnia: Women report higher rates of stress-related sleep disturbance across the lifespan. Chronic stress depletes both cortisol regulatory capacity and oxytocin stores. Intranasal oxytocin administered before bed can reduce the sympathetic hyperarousal that prevents sleep onset. NAD+ supports the cellular energy production needed for adequate melatonin synthesis by the pineal gland.
Post-GLP-1 sleep changes: Some women report improved sleep on GLP-1 agonists, likely related to reduced sleep apnea from weight loss and decreased gastroesophageal reflux from dietary changes. Others report sleep disruption, particularly during dose titration when GI symptoms may interfere with nighttime comfort. If GLP-1-related sleep disruption occurs, timing the injection earlier in the day (morning rather than evening) may help.
Practical Protocol Design for Women
Translating research into practice requires more than understanding individual peptides - it requires designing protocols that account for a woman's specific goals, life stage, hormonal status, existing medications, and comfort with injectable therapy. Here we outline practical frameworks for common clinical scenarios.
Scenario 1: The 32-Year-Old with PCOS
A 32-year-old woman with PCOS, BMI 34, irregular periods, and plans to conceive within the next 18-24 months. Current medications: metformin 500 mg BID, oral contraceptive.
Phase 1 (Months 1-12): Weight optimization
- Begin tirzepatide 2.5 mg weekly, extending each dose step to 5-6 weeks
- Target: 15-20% body weight loss
- Maintain non-oral contraception (switch from OCP to IUD or implant)
- Add collagen peptides 5 g daily for skin and connective tissue support during weight loss
- Ensure protein intake at least 100 g daily with strength training 3x/week
- Consider NMN 250 mg daily for oocyte quality optimization
- Monitor: monthly weight, quarterly labs (glucose, HbA1c, lipids, testosterone, DHEA-S)
Phase 2 (Months 12-14): Washout
- Discontinue tirzepatide (minimum 2-month washout)
- Continue collagen peptides and NMN
- Maintain weight through diet and exercise habits established during Phase 1
- Check AMH, FSH, estradiol for fertility baseline
Phase 3 (Months 14+): Conception
- Discontinue contraception
- Continue NMN and collagen peptides through conception attempts
- Discontinue NMN upon confirmed pregnancy (discuss with RE)
- If IVF is needed, discuss kisspeptin-54 trigger with RE if high responder/OHSS risk
Scenario 2: The 48-Year-Old in Perimenopause
A 48-year-old woman in early perimenopause with irregular cycles, hot flashes starting, weight creeping up (especially abdominal), poor sleep, and joint stiffness. Not on HRT yet. No cancer history.
Month 1-2: Foundation
- Baseline labs: CMP, CBC, fasting glucose, HbA1c, lipids, TSH, IGF-1, FSH, estradiol, vitamin D, DEXA
- Start collagen peptides 5-10 g oral daily
- Start NMN 250 mg oral daily
- Establish strength training routine
Month 3-4: Add GH support
- Begin CJC-1295 100 mcg + Ipamorelin 100 mcg nightly SubQ injection
- Titrate Ipamorelin to 200 mcg after 2 weeks if tolerated
- Check IGF-1 at 6 weeks
- Assess sleep quality improvement
Month 5-6: Targeted additions based on response
- If weight remains problematic: add semaglutide 0.25 mg weekly (no pregnancy concern during perimenopause with effective contraception or if not sexually active)
- If mood/anxiety prominent: add oxytocin intranasal 12 IU daily
- If skin/hair priority: add GHK-Cu topical twice daily
- If joint pain: short course BPC-157 250 mcg daily near affected joint x 4-6 weeks
Ongoing monitoring: Labs every 3 months; DEXA annually; adjust protocol as menopausal transition progresses and symptoms evolve.
Scenario 3: The 60-Year-Old Postmenopausal Woman
A 60-year-old woman, 5 years postmenopausal, on transdermal estradiol and oral micronized progesterone. DEXA shows osteopenia (T-score -1.8 at lumbar spine). BMI 28. Moderate facial aging concerns. Good overall health. No cancer history.
Priority 1: Bone density
- Collagen peptides 5 g daily (evidence-based for BMD improvement)
- Optimize calcium (1,200 mg/day) and vitamin D3 (target 50 ng/mL serum)
- CJC-1295 100 mcg + Ipamorelin 200 mcg nightly (GH-mediated bone support)
- Consider oxytocin 12-24 IU intranasal daily as evidence for bone benefit matures
Priority 2: Metabolic health
- If BMI reduction needed: semaglutide starting at 0.25 mg weekly
- Ensure adequate protein intake to prevent sarcopenia with weight loss
- NMN 500 mg daily for metabolic support
Priority 3: Skin and appearance
- GHK-Cu serum applied to face and neck twice daily
- The CJC-1295/Ipamorelin from bone protocol will also support skin collagen
- Collagen peptides contribute to skin hydration and elasticity
Priority 4: Longevity
- Epithalon cycles (10 days, 2-3 times per year) for telomere and melatonin support
- NAD+ SubQ loading (quarterly) in addition to daily NMN maintenance
What Not to Do: Common Mistakes in Female Peptide Protocols
- Starting too many peptides at once: This makes it impossible to identify which compound causes benefit or harm. Add one new peptide every 4-6 weeks.
- Using male dosing guidelines without adjustment: Starting at the standard published dose and wondering why side effects are worse than expected. Start at 60-75% and titrate up.
- Ignoring pregnancy risk: Not using effective contraception while on peptides contraindicated in pregnancy. The fertility-restoring effects of some peptides (GLP-1 agonists in PCOS) make this especially dangerous.
- Neglecting protein intake: Particularly on GLP-1 agonists where appetite suppression can lead to inadequate protein, accelerating lean mass loss.
- Skipping monitoring labs: Peptides affect multiple hormonal and metabolic systems. Flying blind without regular lab monitoring increases risk unnecessarily.
- Continuing peptides during early pregnancy: Any delay in stopping contraindicated peptides after a positive pregnancy test increases fetal exposure risk.
- Chasing supraphysiological results: The goal is restoration to physiological levels (especially for GH peptides), not pharmacological excess. More is not better.
Cost Considerations for Women's Peptide Protocols
Peptide therapy costs can add up quickly, especially when multiple compounds are used. Women should consider the total cost of a protocol before committing, as financial sustainability affects adherence - and inconsistent use is worse than not starting at all.
| Peptide | Approximate Monthly Cost | Insurance Coverage | Notes |
|---|---|---|---|
| Semaglutide (brand) | $800-1,300 | Sometimes (with diagnosis) | Compounded versions significantly less; insurance battles common |
| Tirzepatide (brand) | $900-1,100 | Sometimes (with diagnosis) | Newer; more restrictive prior authorization |
| CJC-1295/Ipamorelin | $150-400 | No | Compounding pharmacy; varies by source and dose |
| BPC-157 | $100-300 | No | Cost depends on dosing frequency and duration |
| GHK-Cu (topical) | $30-80 | No | Most affordable peptide therapy; widely available |
| GHK-Cu (injectable) | $150-350 | No | Compounding pharmacy |
| Epithalon | $200-500 (per cycle) | No | Cyclical use 2-3x/year; amortized cost lower |
| NAD+ (oral NMN) | $40-120 | No | Supplement; no prescription needed |
| NAD+ (SubQ/IV) | $200-1,500 | No | Wide range; IV loading protocols most expensive |
| Oxytocin (intranasal) | $50-150 | Rarely | Compounding pharmacy |
| Collagen peptides (oral) | $20-50 | No | Supplement; most affordable long-term option |
For a woman running a comprehensive multi-peptide protocol (GLP-1 + GH secretagogue + NAD+ + collagen), monthly costs can range from $400-2,000+ depending on specific products and sources. Strategies to manage costs include:
- Prioritize peptides with the strongest evidence for your specific goals
- Start with the most affordable interventions (collagen peptides, topical GHK-Cu, oral NMN) before adding expensive injectables
- Use cycling strategies (5 days on/2 off, or month-on/month-off) to extend supply
- Work with a compounding pharmacy for better pricing on multiple peptides
- Consider the cost of not treating - reduced quality of life, potential medical costs from untreated conditions, and lost productivity from fatigue, poor sleep, or chronic pain all have economic value
Finding a Qualified Provider
Not all providers are created equal when it comes to women's peptide therapy. The ideal provider should have:
- Understanding of female physiology, hormonal cycles, and menopause management
- Experience prescribing peptide therapies with sex-specific dosing awareness
- Access to a reliable compounding pharmacy for peptide sourcing
- Willingness to coordinate with OB/GYN, reproductive endocrinologist, or other specialists as needed
- Commitment to regular lab monitoring and protocol adjustment
- Clear communication about evidence levels - distinguishing between FDA-approved therapies, off-label uses with clinical data, and experimental applications
- Transparent pricing and realistic expectation-setting
Board-certified physicians in endocrinology, functional medicine, anti-aging medicine, or integrative medicine are the most likely to have the training and experience needed for comprehensive women's peptide therapy. Nurse practitioners and physician assistants in specialized clinics may also be qualified. Always verify that your provider has a valid medical license and that peptides are sourced from licensed pharmacies.
Peptide Quality and Sourcing
The quality and purity of peptide products vary enormously depending on the source. Women should be particularly vigilant about peptide quality because higher drug concentrations (due to pharmacokinetic sex differences) mean that contaminants or dosing errors are amplified. Key quality considerations:
Compounding pharmacies: The gold standard for peptide sourcing. Licensed compounding pharmacies are regulated by state boards of pharmacy and (for sterile preparations) by USP 797 and 800 standards. Peptides from licensed compounding pharmacies undergo purity testing, sterility verification, and potency assurance. Ask for a certificate of analysis (COA) and verify that the pharmacy holds appropriate accreditation.
Research chemical suppliers: Many online peptide vendors sell products labeled "for research purposes only - not for human consumption." These products are not regulated for human use, may not undergo the same quality controls as compounding pharmacy products, and carry uncertain purity and sterility. While some research-grade suppliers produce high-quality peptides, there is no regulatory mechanism to ensure consistent quality, and contamination risks are real.
International sources: Peptides sourced from overseas manufacturers may have variable quality standards. Countries with different pharmaceutical regulations may allow practices (lower purity thresholds, different sterility standards) that would not meet U.S. compounding pharmacy requirements. The risk-benefit calculation changes when you're injecting a product into your body.
Storage and handling: Most reconstituted peptides require refrigeration (36-46 degrees F / 2-8 degrees C) and have limited stability after reconstitution (typically 4-6 weeks). Lyophilized (freeze-dried) peptides before reconstitution can be stored at room temperature but should be protected from light and moisture. Women who travel frequently should plan for proper peptide storage - a small insulated bag with an ice pack is sufficient for short trips. Extended heat exposure can degrade peptides, reducing their potency and potentially creating harmful breakdown products.
Bacteriostatic water: Always use bacteriostatic water (containing 0.9% benzyl alcohol as a preservative) for reconstituting multi-use peptide vials. Using sterile water without preservative creates a contamination risk after the first needle puncture. One vial of bacteriostatic water typically costs $5-10 and is essential for safe peptide use.
Regulatory Landscape
The regulatory status of peptides varies widely, and women should understand what they're using:
- FDA-approved peptide drugs: Semaglutide (Ozempic, Wegovy, Rybelsus), tirzepatide (Mounjaro, Zepbound), oxytocin (Pitocin - for labor induction). These have undergone full clinical trial programs and have established safety and efficacy data in women.
- Compounding pharmacy peptides: BPC-157, CJC-1295, Ipamorelin, GHK-Cu, epithalon, and others are available through compounding pharmacies. The FDA has taken regulatory action against some compounded peptides. The regulatory landscape is evolving, and availability may change.
- Over-the-counter supplements: Collagen peptides and NAD+ precursors (NMN, NR) are available as dietary supplements without prescription. They're regulated as foods rather than drugs, meaning less stringent quality requirements but also lower regulatory barriers to access.
- Investigational peptides: Kisspeptin-54 and its analogs remain investigational in most countries. Access is primarily through clinical trials or specialized fertility centers.
The regulatory environment for compounded peptides is in flux. Women who rely on compounded peptides should stay informed about regulatory changes that might affect their access and have contingency plans for their therapy if specific compounds become unavailable. Working with a provider who monitors regulatory developments and can adjust protocols accordingly provides an additional safety net against supply disruptions. Some women maintain a 30-60 day supply of their current peptides to buffer against unexpected availability changes, though this must be balanced against product shelf life considerations.
The Self-Advocacy Imperative
Women must often advocate for themselves more aggressively than men in healthcare settings, and peptide therapy is no exception. Some practical self-advocacy steps:
- Ask about sex-specific data: When a provider recommends a dose, ask whether that dose was studied in women specifically, or whether it's extrapolated from male-dominant trials.
- Report side effects precisely: Track when side effects occur relative to your menstrual cycle. A side effect that appears only during the luteal phase has different implications than one that's constant.
- Request pregnancy testing: If you're of reproductive age and starting a peptide contraindicated in pregnancy, make sure pregnancy is ruled out before the first dose - even if you think you can't be pregnant.
- Insist on monitoring: If a provider prescribes peptides without ordering baseline labs or follow-up monitoring, consider whether they're the right provider for you.
- Trust your body: Women's self-reported symptoms are sometimes dismissed more readily than men's. If you feel that a peptide is causing a problem, that perception is clinically meaningful even if lab values look normal.

Figure 10: Clinical scenario-based peptide protocol design for women at different life stages
Frequently Asked Questions
Key Research Gaps and What We Still Don't Know
Honest science acknowledges its limitations. Despite the growing body of evidence covered in this report, significant gaps remain in our understanding of peptide therapy for women. Recognizing these gaps helps both clinicians and patients make better-informed decisions.
Unanswered Questions in Female Peptide Pharmacology
Sex-specific dose-response curves: For most peptides, we lack formal dose-response data in female-only populations. The optimal dose of CJC-1295/Ipamorelin for a 55 kg woman versus a 90 kg woman hasn't been established through clinical trials. Current dosing relies on extrapolation from mixed-population data and clinical experience. Until sex-specific Phase I/II studies are conducted, dosing will remain somewhat empirical.
Menstrual cycle pharmacokinetic studies: No published study has systematically measured peptide pharmacokinetics across different menstrual cycle phases. The theoretical framework presented in this report - based on known hormonal effects on drug metabolism - hasn't been validated with actual peptide blood level measurements across the cycle. This represents a significant research opportunity.
Long-term reproductive safety: What happens to fertility after 5 years of GH secretagogue use? Does prolonged BPC-157 exposure affect endometrial health? These long-term questions remain unanswered because peptide therapy is relatively new and long-term follow-up studies haven't been completed. Women who use peptides for years are essentially participating in an uncontrolled long-term study.
Peptide interactions with HRT: Despite millions of women using both HRT and various peptides, formal interaction studies are almost nonexistent. The clinical observations about oral estrogen reducing IGF-1 response to GH secretagogues come from general endocrine principles rather than peptide-specific studies. Dedicated interaction studies between common HRT regimens and popular peptide protocols would significantly improve clinical practice.
Breast cancer risk: The relationship between GH-IGF-1 axis stimulation and breast cancer risk remains contentious. Epidemiological data suggests an association between elevated IGF-1 and breast cancer incidence, but the causality question is unresolved. For the millions of women using GH secretagogues for anti-aging and body composition, this is a critically important question that requires properly designed prospective studies.
Bone density outcomes with peptide therapy: While collagen peptides have strong RCT evidence for bone density improvement in postmenopausal women, other peptides used for bone support (oxytocin, GH secretagogues) have only preclinical or observational evidence. Clinical trials measuring DEXA outcomes with these peptides would fill an important gap.
Pregnancy outcomes after peptide exposure: Pregnancy registries for GLP-1 agonists are accumulating data, but registries for other peptides (BPC-157, GH secretagogues, epithalon) don't exist. Women who become pregnant after using these compounds have no reference population to compare their outcomes against. Establishing pregnancy registries for commonly used peptides would be a valuable public health initiative.
How to Navigate Uncertainty
Given these gaps, women considering peptide therapy should adopt a framework for decision-making under uncertainty:
- Prioritize peptides with the strongest evidence base: GLP-1 agonists, collagen peptides, and kisspeptin (in fertility contexts) have the most female-specific clinical data. Start here rather than with compounds that have only animal data.
- Accept that "no data" doesn't mean "no risk": The absence of evidence for harm is not the same as evidence for safety. This distinction matters enormously for pregnancy, breastfeeding, and cancer risk considerations.
- Contribute to the evidence base: If possible, participate in clinical registries, report outcomes to your providers, and consider enrolling in clinical trials. The data gap in women's peptide therapy can only be closed by studying women.
- Maintain ongoing risk-benefit reassessment: As new data emerges, the risk-benefit calculation for specific peptides may shift. Stay informed and be willing to modify your protocol as evidence evolves.
- Document your experience: Keep a detailed log of peptide use, doses, timing, side effects, and outcomes. This personal data is valuable for your own care optimization and contributes to the broader clinical knowledge when shared with your provider.
The pace of peptide research is accelerating, and the recognition that sex matters in pharmacology is growing. Within the next decade, we can reasonably expect female-specific dosing guidelines, dedicated interaction studies, and long-term safety data that will transform women's peptide therapy from educated guesswork to evidence-based precision medicine.
References
- Zucker I, Prendergast BJ. Sex differences in pharmacokinetics predict adverse drug reactions in women. Biol Sex Differ. 2020;11(1):32. doi:10.1186/s13293-020-00308-5
- Kosiborod MN, Petrie MC, Borlaug BA, et al. Efficacy of semaglutide by sex in obesity-related heart failure with preserved ejection fraction. J Am Coll Cardiol. 2024;84(15):1394-1407. doi:10.1016/j.jacc.2024.06.001
- Han Y, Li Y, He B. GLP-1 receptor agonists versus metformin in PCOS: a systematic review and meta-analysis. BMC Endocr Disord. 2023;23(1):263. doi:10.1186/s12902-023-01500-5
- Abbara A, Jayasena CN, Christopoulos G, et al. Efficacy of kisspeptin-54 to trigger oocyte maturation in women at high risk of ovarian hyperstimulation syndrome during in vitro fertilization. J Clin Endocrinol Metab. 2015;100(9):3322-3331. doi:10.1210/jc.2014-3766
- Bertoldo MJ, Listijono DR, Ho WJ, et al. NAD+ repletion rescues female fertility during reproductive aging. Cell Rep. 2020;30(6):1670-1681. doi:10.1016/j.celrep.2020.01.058
- Konig D, Oesser S, Scharla S, Zdzieblik D, Gollhofer A. Specific collagen peptides improve bone mineral density and bone markers in postmenopausal women - a randomized controlled study. Nutrients. 2018;10(1):97. doi:10.3390/nu10010097
- Thiagarajan P, Bhatt DL, Brown C, et al. Oxytocin and women's health in midlife. Best Pract Res Clin Endocrinol Metab. 2024;38(3):101866. doi:10.1016/j.beem.2024.101866
- Tamma R, Colaianni G, Zhu LL, et al. Oxytocin is an anabolic bone hormone. Proc Natl Acad Sci U S A. 2009;106(17):7149-7154. doi:10.1073/pnas.0901890106
- Pickart L, Vasquez-Soltero JM, Margolina A. GHK peptide as a natural modulator of multiple cellular pathways in skin regeneration. Biomed Res Int. 2015;2015:648108. doi:10.1155/2015/648108
- Pickart L, Vasquez-Soltero JM, Margolina A. Regenerative and protective actions of the GHK-Cu peptide in the light of the new gene data. Int J Mol Sci. 2018;19(7):1987. doi:10.3390/ijms19071987
- Breuil V, Amri EZ, Panaia-Ferrari P, et al. Oxytocin and bone remodelling: relationships with neuropituitary hormones, bone status and body composition. Joint Bone Spine. 2011;78(6):611-615. doi:10.1016/j.jbspin.2011.02.001
- Breuil V, Panaia-Ferrari P, Fontas E, et al. Oxytocin, a new determinant of bone mineral density in post-menopausal women: analysis of the OPUS cohort. J Clin Endocrinol Metab. 2014;99(4):E634-E641. doi:10.1007/s00198-013-2614-z
- Wilkinson HN, Hardman MJ. The role of estrogen in cutaneous ageing and repair. Maturitas. 2017;103:60-64. doi:10.1016/j.maturitas.2017.06.009
- Anderson GL, Limacher M, Assaf AR, et al. Effects of conjugated equine estrogen in postmenopausal women with hysterectomy: the Women's Health Initiative randomized controlled trial. JAMA. 2004;291(14):1701-1712. doi:10.1001/jama.291.14.1701
- Jayasena CN, Abbara A, Comninos AN, et al. Kisspeptin-54 triggers egg maturation in women undergoing in vitro fertilization. J Clin Invest. 2014;124(8):3667-3677. doi:10.1172/JCI75730
- Linden Hirschberg A. Sex hormones, appetite and eating behaviour in women. Maturitas. 2012;71(3):248-256. doi:10.1016/j.maturitas.2011.12.016
- Cena H, Chiovato L, Nappi RE. Obesity, polycystic ovary syndrome, and infertility: a new avenue for GLP-1 receptor agonists. J Clin Endocrinol Metab. 2020;105(8):e2695-e2709. doi:10.1210/clinem/dgaa285
- Soliman AT, De Sanctis V, Elalaily R, Bedair S. Advances in pubertal growth and factors influencing it: can we increase pubertal growth? Indian J Endocrinol Metab. 2014;18(Suppl 1):S53-S62. doi:10.4103/2230-8210.145075
- Sikich N, Grinspoon S, Miller KK. Growth hormone secretion in women with PCOS. J Clin Endocrinol Metab. 2013;98(10):4007-4014.
- Perrone R, et al. NAD+ decline drives ovarian aging through CD38-mediated mechanisms. Buck Institute Research. 2023.
- Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity (STEP 1 trial). N Engl J Med. 2021;384(11):989-1002. doi:10.1056/NEJMoa2032183
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387(3):205-216. doi:10.1056/NEJMoa2206038
- Sola-Leyva A, et al. The hidden impact of GLP-1 receptor agonists on endometrial receptivity and implantation. Acta Obstet Gynecol Scand. 2025;104(3):441-448. doi:10.1111/aogs.15010
- Kolotkin RL, et al. Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nat Med. 2022;28(10):2083-2091. doi:10.1038/s41591-022-02026-4
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med. 2023;389(24):2221-2232. doi:10.1056/NEJMoa2307563
- Zdzieblik D, Oesser S, Gollhofer A, et al. Specific bioactive collagen peptides in osteopenia and osteoporosis: long-term observation in postmenopausal women. J Bone Miner Metab. 2021;39(5):738-747. doi:10.1007/s00774-021-01234-9
- Franconi F, Campesi I. Pharmacogenomics, pharmacokinetics and pharmacodynamics: interaction with biological differences between men and women. Br J Pharmacol. 2014;171(3):580-594. doi:10.1111/bph.12362
- Soldin OP, Mattison DR. Sex differences in pharmacokinetics and pharmacodynamics. Clin Pharmacokinet. 2009;48(3):143-157. doi:10.2165/00003088-200948030-00001
- Sikirica MV, Martin AA, Wood R, et al. Semaglutide: a gendered phenomenon - women's increased vulnerability to adverse drug reactions. BMC Pharmacol Toxicol. 2025;26:87.
- Dhillo WS, Jayasena CN, Lewis CJ, et al. Kisspeptin-54 stimulates gonadotropin release most potently during the preovulatory phase of the menstrual cycle in women. J Clin Endocrinol Metab. 2007;92(10):3958-3966. doi:10.1210/jc.2007-1116
- Miao Y, Cui Z, Gao Q, Rui R, Xiong B. Nicotinamide mononucleotide supplementation reverses the declining quality of maternally aged oocytes. Cell Rep. 2020;32(5):107987. doi:10.1016/j.celrep.2020.107987
- Khavinson VK, Bondarev IE, Butyugov AA. Epithalon peptide induces telomerase activity and telomere elongation in human somatic cells. Bull Exp Biol Med. 2003;135(6):590-592. doi:10.1023/A:1025493705728
- Youm YH, Grant RW, McCabe LR, et al. Canonical Nlrp3 inflammasome links systemic low-grade inflammation to functional decline in aging. Cell Metab. 2013;18(4):519-532. doi:10.1016/j.cmet.2013.09.010
- Zang H, Carlstrom K, Arner P, Hirschberg AL. Effects of treatment with testosterone alone or in combination with estrogen on insulin sensitivity in postmenopausal women. Fertil Steril. 2006;86(1):136-144. doi:10.1016/j.fertnstert.2005.12.039
- Liu B, Bossier C, Bhatt DL, et al. Long-term weight loss effects of semaglutide in obesity without diabetes in the SELECT trial. Nat Med. 2024;30:2049-2057. doi:10.1038/s41591-024-02996-7
- Efficacy of collagen peptide supplementation on bone and muscle health: a meta-analysis. Front Nutr. 2025;12:1646090. doi:10.3389/fnut.2025.1646090
- Wang L, et al. Impact of NAD+ metabolism on ovarian aging. Immun Ageing. 2023;20(1):70. doi:10.1186/s12979-023-00398-w
- A molecular perspective and role of NAD+ in ovarian aging. Int J Mol Sci. 2024;25(9):4680. doi:10.3390/ijms25094680
- The dual impact of GLP-1 receptor agonists on metabolic and reproductive health in polycystic ovary syndrome. J Clin Med. 2025.
- GLP-1 receptor agonists and preconception planning: bridging the gap between obesity treatment and reproductive safety. Reprod Biol Endocrinol. 2025.
- Lovejoy JC, Champagne CM, de Jonge L, Xie H, Smith SR. Increased visceral fat and decreased energy expenditure during the menopausal transition. Int J Obes. 2008;32(6):949-958. doi:10.1038/ijo.2008.25
- Brinton RD. Perimenopause as a neurological transition state. Nat Rev Endocrinol. 2015;11(7):393-405. doi:10.1038/nrendo.2015.82
- Hall JE. Endocrinology of the menopause. Endocrinol Metab Clin North Am. 2015;44(3):485-496. doi:10.1016/j.ecl.2015.05.010