Executive Summary
After age 40, men face a dual hormonal challenge that most don't fully understand until they're already feeling the effects. Testosterone drops by roughly 1-2% per year while growth hormone secretion falls 14% per decade - and together, these shifts quietly reshape body composition, energy, recovery capacity, and long-term health.
Key Takeaways
- After age 40, men face a dual hormonal challenge that most don't fully understand until they're already feeling the effects.
- Testosterone drops by roughly 1-2% per year while growth hormone secretion falls 14% per decade - and together, these shifts quietly reshape body composition, energy, recovery capacity, and long-term health.
- If you're a man over 40, you've probably noticed the changes.
- Workouts that once built muscle now just maintain it.
- Sleep doesn't restore you the way it used to.
If you're a man over 40, you've probably noticed the changes. Workouts that once built muscle now just maintain it. Sleep doesn't restore you the way it used to. Recovery from injuries takes twice as long, and that stubborn visceral fat around the midsection won't budge no matter what you do. These aren't signs of laziness or poor discipline. They're the predictable biological consequences of declining hormone production - a process that accelerates with each passing decade.
Peptide therapy offers a fundamentally different approach than traditional hormone replacement. Rather than flooding your system with exogenous hormones like testosterone or growth hormone directly, peptides work by stimulating your body's own production pathways. They act as signaling molecules - short chains of amino acids that tell your pituitary gland to release more growth hormone, or instruct your hypothalamus to ramp up the signals that drive testosterone synthesis. The distinction matters because this approach preserves your body's natural feedback loops instead of shutting them down.
This report covers the full spectrum of peptide options available to aging men. We'll examine the biology of male hormonal decline in detail, then walk through specific peptide categories: GH-releasing peptides like CJC-1295/Ipamorelin, testosterone-supporting compounds like gonadorelin and kisspeptin, healing peptides like BPC-157 and TB-500, and longevity-focused compounds like Epithalon and NAD+. We'll also address body composition strategies using GLP-1 receptor agonists, sexual health peptides, sleep optimization, and cardiovascular protection.
Each section includes clinical evidence with DOI-referenced citations, practical protocol guidance organized by decade (40s, 50s, 60+), and honest discussion of what the research does and doesn't support. Because while peptide therapy holds genuine promise for the aging male, not every claim circulating online stands up to scrutiny. You deserve the full picture.
KEY TAKEAWAYS
Men over 40 experience measurable declines in both testosterone (0.4-1.3% per year) and growth hormone (14% per decade). Peptide therapy can address these declines by stimulating natural production rather than replacing hormones directly. The most evidence-backed approaches combine GH secretagogues with HPG axis support, recovery peptides, and longevity compounds - tailored to your specific decade of life and individual lab values.

Figure 1: The four pillars of peptide therapy for aging men - GH optimization, testosterone support, recovery enhancement, and longevity protection.
The Biology of Male Aging: What Actually Happens After 40
Understanding why your body changes after 40 requires looking at three interconnected hormonal systems that all decline simultaneously - creating a cascade of effects that no single intervention can fully address.
The Testosterone Decline: More Than Just a Number
Testosterone doesn't fall off a cliff the way estrogen does during menopause. Instead, it's a slow, steady erosion that most men don't notice until the cumulative deficit becomes impossible to ignore. According to data from the Massachusetts Male Aging Study and subsequent longitudinal analyses, total serum testosterone decreases at approximately 0.4% per year starting in the mid-30s, while bioavailable (free) testosterone drops faster - at roughly 1.3% annually (Harman et al., 2001; doi:10.1210/jcem.86.2.7219).
But here's what many discussions miss: the decline isn't uniform across all men, and it's not purely age-driven. Researchers have identified that testosterone levels in the general male population have been declining across generations - a phenomenon called a secular trend. A study analyzing data from over 1.5 million blood tests found that men today have significantly lower testosterone at the same age compared to men measured just two decades ago (Travison et al., 2007; doi:10.1210/jc.2006-1375). This suggests environmental factors - obesity, endocrine disruptors, sedentary behavior, and chronic stress - are layering on top of the natural age-related decline.
The practical consequences unfold across several domains:
| Testosterone Level Range | Typical Age Range | Common Symptoms |
|---|---|---|
| 500-700 ng/dL | Early 40s | Minimal symptoms; slight recovery slowing |
| 400-500 ng/dL | Late 40s to early 50s | Reduced libido, increased body fat, mild fatigue |
| 300-400 ng/dL | Late 50s to 60s | Noticeable muscle loss, irritability, sleep disruption |
| Below 300 ng/dL | Variable (clinical hypogonadism) | Significant fatigue, depression, erectile dysfunction, bone density loss |
What's particularly relevant for peptide therapy is that testosterone decline involves dysfunction at multiple levels of the hypothalamic-pituitary-gonadal (HPG) axis. It's not just the testes producing less testosterone - the hypothalamus secretes less GnRH, the pituitary releases less LH and FSH, and the feedback sensitivity changes. This is why approaches that target the HPG axis at the hypothalamic level - like kisspeptin and gonadorelin - can be more physiologically appropriate than simply injecting testosterone (Wu et al., 2008; doi:10.1056/NEJMoa0711764).
The Growth Hormone Decline: Somatopause
If testosterone decline is a slow leak, the growth hormone decline is more like a faucet gradually closing. Growth hormone (GH) production peaks during puberty and the early 20s, then decreases by approximately 14% per decade after age 35. By age 60, roughly 35% of otherwise healthy men meet laboratory criteria for growth hormone deficiency, and by the late 70s, daily GH secretion is only about 15-20% of what it was at age 25 (Iranmanesh et al., 1991; doi:10.1210/jcem-73-6-1081).
This progressive decline - termed "somatopause" - manifests through reduced IGF-1 (insulin-like growth factor 1) levels, since IGF-1 is the primary downstream mediator of growth hormone's effects. In a study of over 2,700 healthy adults, median IGF-1 peaked at roughly 374 ng/mL in 18-year-olds and fell to just 92.7 ng/mL in adults over 70 (Bidlingmaier et al., 2014; doi:10.1210/jc.2014-1381).
Daily GH Secretion by Age - Natural Decline
The clinical consequences of somatopause are broad and overlapping with other aging processes:
- Body composition shifts: Loss of lean muscle mass (sarcopenia), increased visceral adiposity, and redistribution of fat toward the abdominal region
- Reduced recovery capacity: Slower wound healing, longer muscle recovery after exercise, and decreased tissue repair efficiency
- Sleep quality deterioration: GH is primarily released during slow-wave (deep) sleep; as GH declines, sleep architecture changes, creating a vicious cycle where poor sleep further reduces GH output
- Bone density loss: GH and IGF-1 are essential for maintaining bone mineral density; their decline accelerates osteopenia risk
- Cardiovascular changes: Reduced GH is associated with increased LDL cholesterol, reduced cardiac output, and increased arterial stiffness
- Cognitive effects: IGF-1 receptors are abundant in the hippocampus and prefrontal cortex; declining levels correlate with reduced cognitive processing speed and memory consolidation
CLINICAL INSIGHT
The somatopause doesn't affect all men equally. Body composition plays a significant role - visceral fat actively suppresses GH secretion through increased free fatty acids, elevated insulin, and inflammatory cytokines. This means overweight men experience an accelerated GH decline, which in turn promotes further fat accumulation. Breaking this cycle is one of the primary goals of GH-releasing peptide therapy. For a deeper look at GH-boosting peptide stacks, see our CJC-1295/Ipamorelin anti-aging stack guide.
The Interconnected Decline: Why Everything Happens at Once
Testosterone and growth hormone don't decline in isolation. They're part of an interconnected endocrine network where the drop in one hormone accelerates the decline of others. Low testosterone increases SHBG (sex hormone-binding globulin) production, which further reduces free testosterone availability. Low GH leads to increased visceral fat, which produces aromatase - an enzyme that converts testosterone to estrogen. Rising estrogen levels then suppress GnRH output from the hypothalamus, further reducing testosterone. And poor sleep from GH decline reduces both testosterone and GH production even more (Veldhuis et al., 2005; doi:10.1210/jc.2005-0535).
This is why men in their 50s and 60s often describe feeling like everything went wrong at once. It's not imagined - it's a genuine physiological cascade where declining hormones create feedback loops that accelerate further decline. And it's precisely why a multi-peptide approach targeting several pathways simultaneously tends to produce better results than addressing any single hormone in isolation.
Additional age-related changes compound the hormonal decline:
- Increased inflammation: "Inflammaging" - chronic low-grade inflammation - rises with age and suppresses both GH and testosterone production through cytokine-mediated hypothalamic dysfunction (Franceschi et al., 2018; doi:10.1038/s41577-019-0151-9)
- Mitochondrial dysfunction: NAD+ levels decline approximately 50% between ages 40 and 60, reducing cellular energy production and repair capacity
- Telomere shortening: Accelerated by oxidative stress, chronic inflammation, and hormonal decline, telomere erosion limits cellular replication and regenerative capacity
- Insulin resistance: Age-related insulin resistance worsens with declining GH and testosterone, creating a metabolic environment hostile to muscle preservation and fat loss
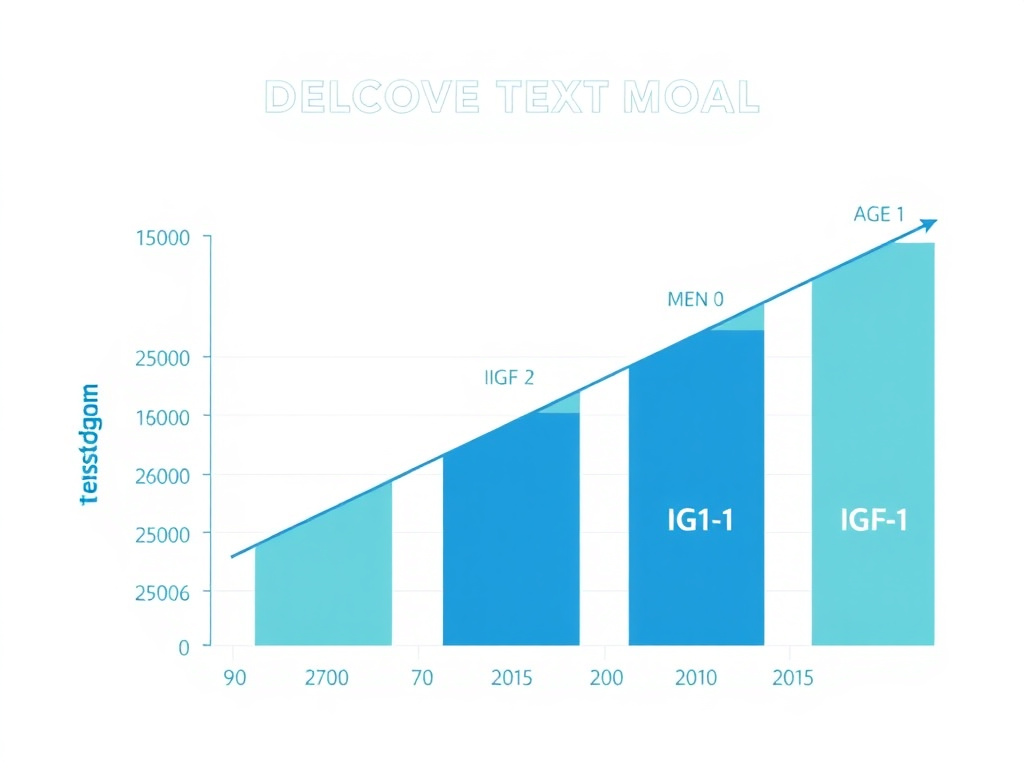
Figure 2: The parallel decline of testosterone, growth hormone, and IGF-1 across the male lifespan, showing accelerated loss after age 40.
Growth Hormone Optimization: The GH Secretagogue Toolkit
Growth hormone secretagogues represent the most well-studied category of peptides for aging men. Rather than injecting synthetic GH directly (which carries real risks and regulatory hurdles), these compounds stimulate your pituitary gland to produce and release more of your own growth hormone in natural pulsatile patterns.
There are four primary GH-releasing peptide options available, each with distinct mechanisms, advantages, and clinical profiles. Understanding these differences is essential for selecting the right compound - or combination - for your situation.
CJC-1295/Ipamorelin: The Gold Standard Combination
The CJC-1295/Ipamorelin combination is arguably the most widely prescribed GH secretagogue stack in clinical anti-aging practice, and for good reason. These two peptides work through complementary mechanisms that produce a combined effect greater than either compound alone.
CJC-1295 is a modified growth hormone-releasing hormone (GHRH) analog. It binds to GHRH receptors on the pituitary gland and stimulates GH synthesis and release. The modification - addition of a Drug Affinity Complex (DAC) in some formulations - extends its half-life from minutes to roughly 6-8 days, providing sustained GH elevation. CJC-1295 without DAC (sometimes called "mod GRF 1-29") has a shorter half-life of about 30 minutes, producing more physiological pulsatile release (Teichman et al., 2006; doi:10.1210/jc.2005-2017).
Ipamorelin works through an entirely different pathway. It's a growth hormone-releasing peptide (GHRP) that binds to ghrelin receptors on the pituitary. What makes ipamorelin unique among GHRPs is its selectivity - it stimulates GH release without significantly affecting cortisol, prolactin, or ACTH levels. This selectivity is clinically meaningful because elevated cortisol counteracts many benefits of GH, and elevated prolactin can suppress testosterone and libido (Raun et al., 1998; doi:10.1210/endo.139.10.6261).
When combined, CJC-1295 and ipamorelin act on different receptor families within the pituitary, producing a 3-5 fold increase in GH release compared to either compound alone. The combination mimics the natural two-signal system the body uses: GHRH opens the door and ghrelin pushes through it.
MECHANISM SUMMARY
CJC-1295: GHRH analog that stimulates GH synthesis + release via GHRH receptors. Provides sustained elevation.
Ipamorelin: Selective ghrelin receptor agonist that triggers acute GH pulses without cortisol/prolactin spikes.
Combined: 3-5x greater GH release than either alone. Mimics natural pulsatile secretion patterns.
Clinical Evidence: A study of CJC-1295 administration demonstrated dose-dependent increases in mean GH concentrations of 2-10 fold, with corresponding IGF-1 increases of 1.5-3 fold that were sustained for up to 6 days after a single injection. What matters here is that the GH release pattern maintained pulsatility rather than producing a flat, non-physiological elevation (Teichman et al., 2006; doi:10.1210/jc.2005-2017).
Typical Protocol for Men Over 40:
| Parameter | Standard Protocol | Notes |
|---|---|---|
| Dosing | CJC-1295: 100-300 mcg / Ipamorelin: 100-300 mcg | Combined in single injection |
| Frequency | 1-2x daily (before bed and/or morning fasted) | Bedtime dosing aligns with natural GH pulse |
| Administration | Subcutaneous injection | Rotate injection sites |
| Cycle | 5 days on / 2 days off or continuous | Some clinicians prefer cycling to prevent desensitization |
| Duration | 3-6 months initial, then reassess | Monitor IGF-1 at 4-6 weeks |
Most men report noticeable improvements in sleep quality within the first 1-2 weeks, with body composition changes (reduced waist circumference, improved muscle tone) becoming apparent by weeks 8-12. For a complete breakdown of this combination, read our CJC-1295/Ipamorelin anti-aging stack research report.
Sermorelin: The GHRH Analog Pioneer
Sermorelin was the first GHRH analog used clinically and remains a solid option, particularly for men who want a well-characterized compound with decades of safety data. It's the biologically active fragment (amino acids 1-29) of endogenous GHRH, which is naturally 44 amino acids long.
Sermorelin was FDA-approved in 1997 (as Geref) for diagnostic testing of GH deficiency and briefly for pediatric growth disorders. While the commercial product was discontinued for business reasons (not safety concerns), it remains available through compounding pharmacies and has accumulated more clinical data than most peptides in the anti-aging space.
Clinical Evidence: A study published in the Journal of Clinical Endocrinology & Metabolism demonstrated that sermorelin administration to older adults increased GH secretion to levels comparable to younger adults, improved body composition, and was well-tolerated over extended use. Crucially, sermorelin preserved the natural pulsatile pattern of GH release and maintained feedback regulation - meaning the body's built-in safety mechanisms remained intact (Walker, 2006; doi:10.1016/j.cger.2006.06.002).
Advantages of sermorelin include its excellent safety record, preservation of GH pulsatility, and the fact that it doesn't suppress natural GH production when discontinued. However, its short half-life (10-20 minutes) means it must be injected more frequently than CJC-1295 with DAC, and its effects are somewhat less potent on a per-dose basis.
Typical Protocol for Men Over 40:
| Parameter | Standard Protocol | Notes |
|---|---|---|
| Dosing | 200-500 mcg | Start low, titrate based on IGF-1 response |
| Frequency | Once daily before bed | Aligns with natural GH peak during sleep |
| Administration | Subcutaneous injection | Abdominal area preferred |
| Duration | 3-6 months, then cycling recommended | Some protocols use 5 on/2 off weekly |
MK-677 (Ibutamoren): The Oral GH Secretagogue
MK-677 stands apart from the other GH secretagogues on this list because it's taken orally - no injections required. It's a non-peptide ghrelin receptor agonist with excellent oral bioavailability and a long half-life of approximately 24 hours, meaning once-daily dosing is sufficient.
Clinical Evidence: The clinical data on MK-677 in aging populations is substantial. In a landmark 12-month randomized controlled trial in healthy older adults (ages 60-81), MK-677 at 25 mg daily increased GH secretion by 97%, raised IGF-1 levels by 55-88% (restoring them to young adult reference ranges), and produced a statistically significant increase in fat-free mass of 1.1 kg versus a 0.5 kg loss in the placebo group (Nass et al., 2008; doi:10.7326/0003-4819-149-9-200811040-00003).
Additional findings from this and related trials:
- GH pulsatile secretion pattern was enhanced (1.7-fold increase over 24 hours)
- Sleep quality improved, likely through increased slow-wave sleep duration
- Lean body mass increased without significant changes in total body weight (suggesting simultaneous fat loss and muscle gain)
- No significant cortisol elevation, preserving the benefits of increased GH
However, MK-677 carries several caveats that men over 40 should be aware of:
IMPORTANT CONSIDERATIONS FOR MK-677
Blood glucose effects: MK-677 consistently increases fasting glucose and insulin levels in clinical trials. For men over 40 who may already have pre-diabetic tendencies or insulin resistance, this requires careful monitoring. HbA1c and fasting glucose should be checked before starting and at 4-8 week intervals.
Appetite stimulation: As a ghrelin agonist, MK-677 reliably increases hunger - an effect that can undermine body composition goals if not managed through meal timing and dietary discipline.
Water retention: Mild edema and water retention are common, particularly in the first few weeks. This is generally transient but can affect blood pressure in susceptible individuals.
Prolactin elevation: Unlike ipamorelin, MK-677 can modestly increase prolactin levels, which may impact libido or mood in sensitive individuals (Svensson et al., 1998; doi:10.1210/jcem.83.2.4539).
Typical Protocol for Men Over 40:
| Parameter | Standard Protocol | Notes |
|---|---|---|
| Dosing | 10-25 mg daily | Start at 10 mg; increase to 25 mg if tolerated |
| Timing | Before bed | Minimizes daytime hunger; aligns with sleep GH pulse |
| Administration | Oral (capsule or liquid) | No injection required |
| Cycle | 8-12 weeks on / 4 weeks off | Cycling helps manage glucose impact |
| Monitoring | Fasting glucose, HbA1c, IGF-1 | Check at baseline, 4 weeks, and 8 weeks |
Tesamorelin: The Visceral Fat Specialist
Tesamorelin is a GHRH analog that holds a unique distinction among the GH secretagogues: it's the only one with full FDA approval for a specific clinical indication - reduction of excess abdominal fat (lipodystrophy) in HIV-infected patients. This approval means tesamorelin has passed the rigorous efficacy and safety standards that most peptides haven't been subjected to.
Clinical Evidence: In Phase III clinical trials, tesamorelin reduced visceral adipose tissue (VAT) by approximately 15-18% over 26 weeks, with corresponding improvements in triglycerides and cholesterol ratios. What matters most for aging men, the body composition improvements were specific to visceral fat - the metabolically dangerous fat surrounding internal organs that drives cardiovascular risk, insulin resistance, and systemic inflammation (Falutz et al., 2007; doi:10.1056/NEJMoa0708742).
Beyond its FDA-approved indication, tesamorelin has attracted interest for cognitive applications. A randomized controlled trial in healthy older adults and those with mild cognitive impairment (MCI) found that tesamorelin administration for 20 weeks improved cognitive function on executive function tests and showed favorable changes in biomarkers of Alzheimer's disease in cerebrospinal fluid (Baker et al., 2012; doi:10.1212/WNL.0b013e318273bd11).
Compared to other GH secretagogues, tesamorelin is particularly well-suited for men over 50 whose primary concern is metabolic health and visceral adiposity reduction. Its targeted action on visceral fat - without the appetite-stimulating effects of MK-677 or the broader systemic effects of exogenous GH - makes it an attractive option for this demographic.
Comparing the GH Secretagogues: Which One is Right for You?
| Compound | Mechanism | Administration | Best For | Key Advantage | Key Limitation |
|---|---|---|---|---|---|
| CJC-1295/Ipamorelin | GHRH + Ghrelin receptor | SubQ injection | All-around GH optimization | Strongest combined GH pulse; clean side-effect profile | Requires injection; cost |
| Sermorelin | GHRH receptor | SubQ injection | Conservative approach with long safety record | Decades of safety data; FDA history | Shorter half-life; less potent |
| MK-677 | Ghrelin receptor (oral) | Oral | Men who prefer no injections | Oral; once daily; strong IGF-1 elevation | Glucose elevation; hunger; prolactin |
| Tesamorelin | GHRH receptor | SubQ injection | Visceral fat and metabolic health | FDA-approved; targeted visceral fat reduction | Limited to one clinical indication; cost |
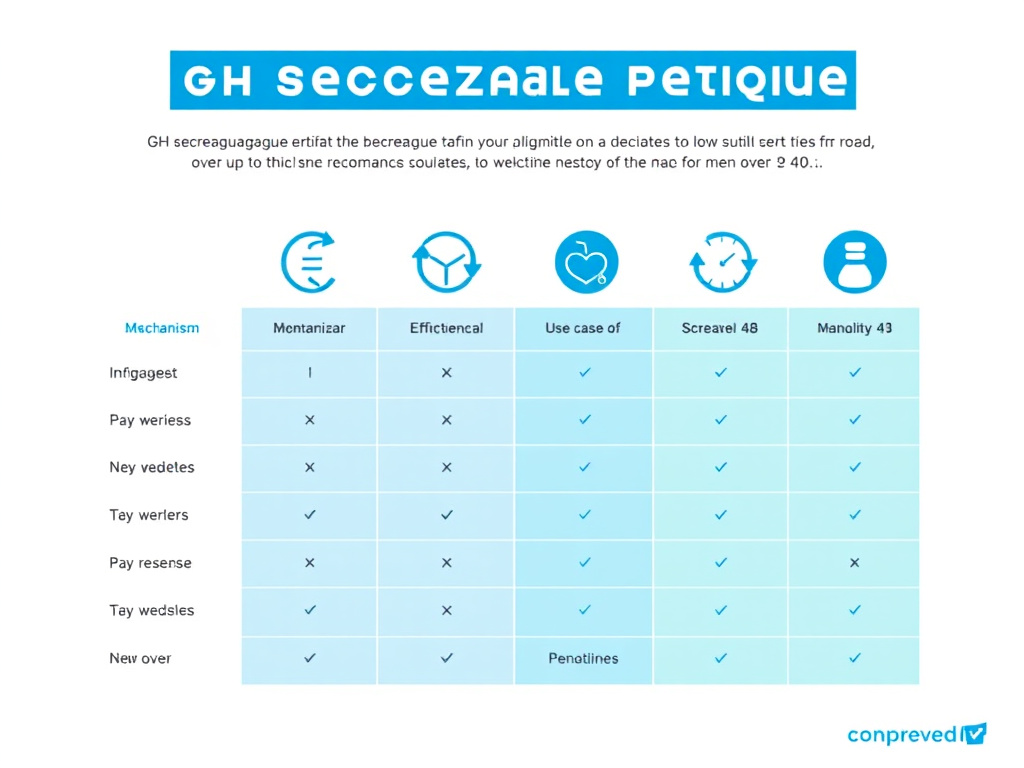
Figure 3: GH secretagogue comparison for men over 40 - mechanism of action, administration route, and ideal clinical applications.
Testosterone Support: Gonadorelin, Kisspeptin & Beyond
For men whose testosterone levels have dipped below optimal, the conventional answer has been TRT - testosterone replacement therapy. But TRT comes with trade-offs: suppressed sperm production, testicular atrophy, and dependency on exogenous hormones. Peptides that work upstream on the HPG axis offer a different path - one that boosts your own testosterone without shutting down your body's production machinery.
Gonadorelin: Direct GnRH Stimulation
Gonadorelin is synthetic gonadotropin-releasing hormone (GnRH) - identical in structure to the GnRH your hypothalamus naturally produces. When administered in pulsatile fashion, it stimulates the anterior pituitary to release luteinizing hormone (LH) and follicle-stimulating hormone (FSH), which then signal the testes to produce testosterone and maintain spermatogenesis (Schally et al., 1971; doi:10.1126/science.173.3994.276).
This mechanism is clinically proven - GnRH has been used for decades to diagnose and treat hypogonadotropic hypogonadism and to support fertility in men on TRT or coming off anabolic steroid cycles. What makes gonadorelin particularly valuable for men over 40 is its ability to restore endogenous testosterone production without replacing it.
Key Clinical Data:
- Pulsatile GnRH administration has been shown to restore normal testosterone levels and spermatogenesis in men with hypothalamic dysfunction (Hoffman & Crowley, 1982; doi:10.1056/NEJM198212163072403)
- When used alongside TRT, gonadorelin can prevent testicular atrophy and maintain sperm production - addressing two of the most significant drawbacks of testosterone replacement
- Response rates vary based on the level of HPG axis dysfunction: men with intact pituitary function respond most reliably, while those with primary testicular failure see limited benefit
CLINICAL PEARL
Gonadorelin's effectiveness depends heavily on pulsatile administration. Continuous GnRH exposure actually suppresses LH/FSH (this is how GnRH agonist drugs like leuprolide work for prostate cancer treatment). The key is mimicking natural pulsatile release - which is why most protocols use 2-3 times daily subcutaneous injections rather than continuous infusion. For a complete review, see our gonadorelin research report.
Typical Protocol for Men Over 40:
| Parameter | Standalone Protocol | TRT Adjunct Protocol |
|---|---|---|
| Dosing | 100-200 mcg per injection | 100-200 mcg per injection |
| Frequency | 2x daily (AM and PM) | 2-3x per week (alongside TRT) |
| Administration | Subcutaneous injection | Subcutaneous injection |
| Goal | Restore endogenous testosterone | Maintain testicular function and fertility |
| Monitoring | Total T, free T, LH, FSH at 4-6 weeks | Same, plus semen analysis if fertility is a goal |
| Expected T increase | 15-30% from baseline (variable) | Maintains pre-TRT testicular volume |
Kisspeptin-10: The Upstream Regulator
Kisspeptin-10 acts one step above gonadorelin in the hormonal cascade. It's a naturally occurring peptide that stimulates GnRH neurons in the hypothalamus, triggering the entire downstream cascade of GnRH release, followed by LH/FSH release, and ultimately testosterone production. Think of it as the master switch that tells the hypothalamus when and how much testosterone the body should make (Dhillo et al., 2005; doi:10.1210/jc.2005-1468).
Clinical Evidence: The clinical data on kisspeptin in men is genuinely impressive. In a study published in the Journal of Clinical Endocrinology & Metabolism, intravenous kisspeptin-10 infusion in healthy men produced the following results:
- LH increased from 5.4 to 20.8 IU/L (a nearly 4-fold increase)
- Testosterone rose from 16.6 to 24.0 nmol/L (a 45% increase)
- LH pulse frequency doubled during kisspeptin infusion
- Both LH pulse size and frequency increased, suggesting enhanced hypothalamic drive
(George et al., 2011; doi:10.1210/jc.2011-1089)
Even more relevant for aging men, a study specifically examining kisspeptin in men with type 2 diabetes and mild hypogonadism found that kisspeptin-10 stimulated LH secretion and increased testosterone in this metabolically compromised population - suggesting the kisspeptin pathway remains functional even when other parts of the HPG axis are impaired (George et al., 2013; doi:10.1210/jc.2012-3564).
What's particularly interesting about kisspeptin's mechanism is that it naturally incorporates the body's feedback systems. Unlike exogenous testosterone, which overrides feedback and suppresses endogenous production, kisspeptin works with the feedback system. If testosterone levels are already adequate, the hypothalamus becomes less responsive to kisspeptin stimulation - creating a built-in safety mechanism against excessive testosterone elevation.
KISSPEPTIN VS. GONADORELIN: WHICH TO CHOOSE?
Kisspeptin acts upstream of GnRH and activates the full HPG cascade naturally. Best for men with hypothalamic-level dysfunction (common in aging, obesity, and metabolic syndrome). Preserves all feedback mechanisms.
Gonadorelin acts at the pituitary level directly. Best for men with confirmed pituitary responsiveness who need reliable LH/FSH stimulation. Better studied, more predictable dose-response. Also valuable as a TRT adjunct for fertility preservation.
Some clinicians combine both - using kisspeptin to activate hypothalamic signaling while supporting pituitary output with low-dose gonadorelin. For a complete review of kisspeptin research, see our kisspeptin hormone peptide guide.
Enclomiphene: The Selective Estrogen Receptor Modulator
While not technically a peptide, enclomiphene deserves mention here because it's increasingly prescribed alongside peptide protocols for testosterone optimization in men over 40. Enclomiphene is the trans-isomer of clomiphene citrate (Clomid) - isolated to remove the estrogenic effects of the cis-isomer (zuclomiphene) that caused side effects in the original drug.
Enclomiphene works by blocking estrogen receptors in the hypothalamus, tricking the brain into thinking estrogen levels are low. This releases the brake on GnRH production, increasing LH and FSH output and consequently raising testosterone. In clinical trials, enclomiphene 25 mg daily increased total testosterone from approximately 230 ng/dL to 450 ng/dL while maintaining sperm counts - a result that compares favorably with low-dose TRT but without the fertility suppression (Kaminetsky et al., 2013; doi:10.1016/j.fertnstert.2013.07.022).
Enclomiphene can be effectively combined with peptide protocols. A common clinical approach pairs enclomiphene with a GH secretagogue - addressing both the testosterone and growth hormone axes simultaneously. Men over 40 with borderline testosterone (300-450 ng/dL) who want to avoid TRT often find this combination provides meaningful symptom relief without suppressing their own hormonal production.
Testosterone Support: Choosing the Right Approach
| Scenario | Recommended Approach | Rationale |
|---|---|---|
| Testosterone 350-500 ng/dL, wants natural boost | Kisspeptin-10 + lifestyle optimization | Upstream stimulation with intact feedback; addresses root cause |
| Testosterone 250-350 ng/dL, fertility important | Gonadorelin + enclomiphene | Dual HPG axis stimulation preserving spermatogenesis |
| Currently on TRT, wants to preserve testicular function | Gonadorelin 2-3x/week alongside TRT | Prevents testicular atrophy; maintains fertility option |
| Testosterone below 250 ng/dL with significant symptoms | TRT + gonadorelin (consider peptide optimization later) | Symptomatic hypogonadism may need direct replacement; peptides alone may be insufficient |
| Type 2 diabetes with mild hypogonadism | Kisspeptin-10 (clinical evidence specific to this population) | Demonstrated efficacy in T2DM-associated hypogonadism |
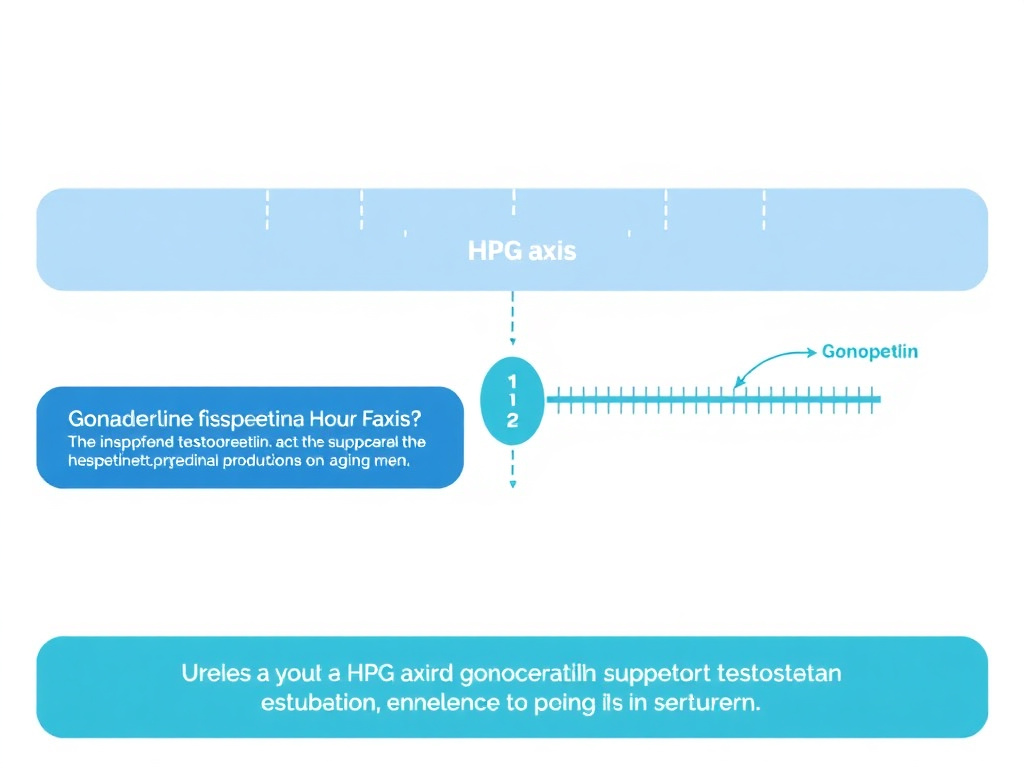
Figure 4: The hypothalamic-pituitary-gonadal (HPG) axis and intervention points for kisspeptin (hypothalamus), gonadorelin (pituitary), and enclomiphene (hypothalamic estrogen receptors).
Body Composition: Fighting the Middle-Age Spread
Body composition changes after 40 aren't just cosmetic. Visceral fat accumulation drives insulin resistance, chronic inflammation, and cardiovascular disease. And the hormonal decline discussed above actively works against your efforts to build muscle and lose fat. Here's how peptide therapy can shift the equation.
GLP-1 Receptor Agonists: The Body Composition Most Impactful Compounds
No discussion of body composition in 2026 would be complete without addressing GLP-1 receptor agonists like semaglutide. While these compounds were developed for type 2 diabetes and obesity, their impact on male body composition after 40 has been transformative.
Clinical Evidence in Men: Semaglutide produces profound body composition changes. An exploratory analysis of the STEP 1 trial found that semaglutide 2.4 mg weekly reduced total fat mass by 19.3% and visceral fat specifically by 27.4% over 68 weeks. What's particularly relevant for aging men is that the ratio of fat loss to lean mass loss was favorable - approximately 60-70% of total weight lost was fat mass, with lean mass largely preserved when combined with adequate protein intake and resistance training (Wilding et al., 2021; doi:10.1056/NEJMoa2032183).
For men over 40, the visceral fat reduction is arguably the most valuable outcome. Visceral adipose tissue isn't passive storage - it's an active endocrine organ producing inflammatory cytokines, aromatase (which converts testosterone to estrogen), and insulin-resistance-promoting factors. Reducing visceral fat by 25-30% can have cascading benefits on testosterone levels, insulin sensitivity, cardiovascular markers, and systemic inflammation.
GLP-1 agonists also appear to have cardiovascular protective effects independent of weight loss. The SELECT trial demonstrated that semaglutide reduced major adverse cardiovascular events (MACE) by 20% in overweight/obese adults with established cardiovascular disease - making this a particularly relevant finding for men over 50 with elevated cardiovascular risk (Lincoff et al., 2023; doi:10.1056/NEJMoa2307563).
GLP-1 AND MUSCLE MASS: THE NUANCE
The concern about GLP-1 agonist-induced muscle loss is real but manageable. In clinical trials, approximately 30-40% of weight lost on semaglutide was lean mass - a ratio comparable to caloric restriction diets. However, when combined with resistance training and adequate protein intake (1.6-2.2 g/kg/day), lean mass preservation improves substantially. For aging men, the strategy should always be: GLP-1 agonist + structured resistance training + high protein intake. Never use these drugs without an exercise component.
AOD-9604: The GH Fragment for Fat Metabolism
AOD-9604 is a synthetic 16-amino acid peptide derived from the C-terminal fragment of human growth hormone (amino acids 177-191). It was designed to capture growth hormone's lipolytic (fat-burning) properties while avoiding its effects on blood glucose and IGF-1 levels. The theory was appealing: fat loss without the metabolic side effects of full-length GH.
However, the clinical evidence is mixed. While animal studies showed promising fat reduction - particularly in obese mouse models where AOD-9604 stimulated lipolysis and inhibited lipogenesis without affecting glucose metabolism - the largest human trial didn't deliver on this promise. In a Phase IIb study of 536 obese subjects over 24 weeks, AOD-9604 failed to produce statistically significant weight loss compared to placebo, and the compound's development was discontinued in 2007 (Heffernan et al., 2001; doi:10.1210/endo.142.12.8508).
That said, some clinicians continue to use AOD-9604 at lower doses as part of multi-compound protocols, reporting subjective improvements in abdominal fat distribution. It's also been explored for joint and cartilage health applications. But if your primary goal is body composition improvement, the evidence clearly favors GLP-1 agonists and GH secretagogues over AOD-9604 as standalone interventions.
Combining Peptides for Optimal Body Composition
The most effective peptide approach to body composition after 40 isn't any single compound - it's a strategic combination targeting multiple pathways:
- GH secretagogue (CJC-1295/Ipamorelin or MK-677) - increases lipolysis, preserves lean mass, improves recovery from training
- GLP-1 agonist (semaglutide) if BMI > 27 - reduces appetite, targets visceral fat, improves metabolic markers
- Testosterone optimization (gonadorelin or kisspeptin) - supports muscle protein synthesis, reduces fat accumulation, improves exercise capacity
- Exercise (non-negotiable) - resistance training 3-4x/week with progressive overload, plus cardiovascular training
- Protein intake - 1.6-2.2 g/kg body weight daily, distributed across 4-5 meals
This layered approach addresses the hormonal, metabolic, and behavioral drivers of age-related body composition decline simultaneously. Each component amplifies the others - GH improves exercise recovery, better recovery enables more intense training, more intense training stimulates testosterone production, and higher testosterone promotes lean mass retention during fat loss.
For men new to peptide therapy and looking for guidance on starting their first protocol, our peptide therapy beginner's guide walks through the basics, while the dosing calculator can help dial in starting doses.
Figure 5: Integrated body composition strategy combining peptide therapy with exercise and nutrition for men over 40.
Recovery & Healing: BPC-157 and TB-500
After 40, the single most frustrating change many men notice isn't the gradual muscle loss or the expanding waistline - it's how long everything takes to heal. A shoulder strain that would have resolved in a week at 25 now lingers for months. Tendinitis becomes chronic. Post-workout soreness lasts three days instead of one. Recovery peptides address this directly.
BPC-157: The Body's Own Repair Accelerator
BPC-157 (Body Protection Compound-157) is a 15-amino acid peptide derived from a protein found in human gastric juice. Despite its origin in the gut, BPC-157's therapeutic interest lies in its remarkable tissue-repair properties observed across nearly every tissue type studied in preclinical models - tendons, ligaments, muscles, bones, the GI tract, and even the nervous system.
Mechanism of Action: BPC-157 works through multiple overlapping repair pathways. It activates the VEGFR2 receptor system, promoting angiogenesis (new blood vessel formation) at injury sites. It stimulates nitric oxide synthesis via the Akt-eNOS axis, enhancing blood flow to damaged tissue. It engages ERK1/2 signaling, which drives cellular proliferation and differentiation in repair processes. And it upregulates growth hormone receptor expression in tendon fibroblasts, making these cells more responsive to circulating GH - a particularly relevant mechanism for aging men with declining GH levels (Chang et al., 2011; doi:10.1111/j.1527-3466.2011.01308.x).
Preclinical Evidence: The animal evidence for BPC-157 is extensive and consistent:
- Tendon repair: Accelerated healing of Achilles tendon transections in rats, with improved biomechanical strength of the healed tissue (Staresinic et al., 2003; doi:10.1002/jor.10171)
- Muscle injury: Enhanced regeneration after quadriceps crush injury and transection, with faster functional recovery (Pevec et al., 2010; doi:10.1016/j.regpep.2010.07.157)
- Ligament healing: Improved MCL (medial collateral ligament) healing in rat models with increased collagen organization
- Bone fracture: Accelerated callus formation and bone mineral density at fracture sites
- GI protection: Demonstrated protective effects against gastric ulcers, inflammatory bowel disease models, and NSAID-induced gut damage
- Neuroprotection: Promoted recovery from peripheral nerve damage and showed protective effects in traumatic brain injury models
Human Evidence - The Critical Gap: Despite the impressive preclinical portfolio, BPC-157's human evidence remains extremely limited. As of 2026, only one Phase I clinical trial has been registered (for inflammatory bowel disease), and its status has been uncertain since 2016. One small retrospective case series reported that 7 of 12 patients with chronic knee pain experienced relief lasting over six months after a single intra-articular BPC-157 injection, but this was not a randomized controlled study (Gwyer et al., 2019; doi:10.1080/17461391.2019.1587526).
This gap between animal evidence and human data is the most important thing to understand about BPC-157. The preclinical signal is strong and consistent across dozens of studies. But until well-designed human trials confirm these findings, BPC-157 remains investigational. That said, thousands of clinicians are prescribing it, and the adverse event profile from clinical observation appears favorable.
REGULATORY NOTE
BPC-157's regulatory status has been in flux. The FDA issued warnings about products containing BPC-157 sold directly to consumers, and it's listed on WADA's prohibited substances list for competitive athletes. However, it remains available through physician-supervised compounding pharmacies in many jurisdictions. Always obtain BPC-157 through a licensed prescriber rather than unregulated online sources, which may contain impurities or incorrect dosing.
Typical Protocol for Men Over 40:
| Application | Dose | Route | Frequency | Duration |
|---|---|---|---|---|
| Systemic healing support | 250-500 mcg | SubQ injection (abdomen) | 1-2x daily | 4-8 weeks |
| Localized injury | 250-500 mcg | SubQ injection near injury site | 1-2x daily | 4-8 weeks |
| GI protection / gut healing | 250-500 mcg | Oral (capsule) or SubQ | 1-2x daily | 4-12 weeks |
| Post-surgical recovery | 500 mcg | SubQ near surgical site | 2x daily | 4-6 weeks post-op |
TB-500 (Thymosin Beta-4): The Systemic Healer
TB-500 is a synthetic version of thymosin beta-4, a naturally occurring 43-amino acid peptide present in nearly all human cells. While BPC-157's evidence is strongest for localized tissue repair, TB-500 has a more systemic action profile - it promotes cellular migration, reduces inflammation throughout the body, and supports tissue remodeling across multiple organ systems simultaneously.
Mechanism of Action: Thymosin beta-4 is an actin-binding protein that regulates cell motility, migration, and differentiation. Its key functions in tissue repair include promoting cell migration to injury sites by modulating actin polymerization, reducing inflammation through downregulation of pro-inflammatory cytokines (NF-kB inhibition), promoting angiogenesis and new blood vessel growth, stimulating stem cell differentiation in injured tissue, and reducing scar formation by influencing collagen deposition patterns (Goldstein et al., 2012; doi:10.1111/j.1749-6632.2011.06420.x).
Clinical Evidence: TB-500 (as thymosin beta-4) has more advanced human clinical data than BPC-157, primarily in ophthalmology and cardiology:
- Eye healing: Phase II trials demonstrated that thymosin beta-4 eye drops accelerated corneal wound healing after surgery and reduced symptoms in dry eye disease
- Cardiac repair: Animal studies showed thymosin beta-4 reduced infarct size and improved cardiac function after myocardial infarction by activating epicardial progenitor cells
- Dermal wounds: Accelerated wound healing in diabetic mouse models, with improved vascular growth at wound sites
For musculoskeletal applications in aging men, the evidence is primarily preclinical but consistent: thymosin beta-4 promotes tendon, muscle, and joint tissue repair through enhanced cell migration and reduced inflammatory damage.
Typical Protocol for Men Over 40:
| Phase | Dose | Frequency | Duration |
|---|---|---|---|
| Loading phase | 750 mcg - 2 mg | 2x per week | 4-6 weeks |
| Maintenance phase | 750 mcg | 1x per week | 4-8 weeks |
| Injury protocol (acute) | 2 mg | 2x per week | 4 weeks loading, then maintain |
The BPC-157 + TB-500 Stack: "The Wolverine Protocol"
In clinical practice, BPC-157 and TB-500 are frequently combined - a pairing often called the "Wolverine stack" in reference to the Marvel character's healing abilities. The rationale for combining them is their complementary mechanisms:
- BPC-157 excels at local tissue repair - angiogenesis, fibroblast activation, growth factor receptor upregulation at the injury site
- TB-500 provides systemic support - cell migration, anti-inflammatory action, and stem cell mobilization
- Together, they create both the local repair environment (BPC-157) and the systemic conditions needed for healing cells to reach and repair the injury (TB-500)
For men over 40 dealing with chronic tendinopathy, joint pain, or slow recovery from training, this combination is one of the most commonly prescribed peptide protocols. Typical combined dosing is BPC-157 250-500 mcg daily plus TB-500 750 mcg twice weekly, run for 6-8 weeks.
Longevity Peptides: Epithalon and NAD+
While the peptides discussed above address specific symptoms of aging - declining hormones, slow recovery, changing body composition - longevity peptides aim at something more fundamental: the underlying cellular mechanisms that drive aging itself.
Epithalon: Telomere Protection and Cellular Aging
Epithalon (also spelled Epitalon) is a synthetic tetrapeptide with the amino acid sequence Ala-Glu-Asp-Gly, based on the natural peptide epithalamin isolated from the pineal gland. Its primary mechanism of interest is the activation of telomerase - the enzyme responsible for maintaining telomere length. Telomeres are the protective caps at the ends of chromosomes that shorten with each cell division. When telomeres become critically short, cells enter senescence (they stop dividing) or apoptosis (they self-destruct). This process is one of the primary cellular mechanisms of aging (Khavinson et al., 2003; doi:10.1023/A:1025493705728).
Mechanism of Action: Epithalon activates telomerase reverse transcriptase (hTERT) gene expression, leading to telomerase production and subsequent telomere elongation. A 2025 study published in PMC confirmed that epitalon increases telomere length in human cell lines through telomerase upregulation, providing molecular confirmation of the mechanism observed in earlier animal and human studies (doi:10.3390/ijms24076390).
Clinical Evidence:
- Human cell studies: Epithalon treatment increased telomere lengths in blood cells of patients aged 60-65 and 75-80, with telomerase activation reaching levels comparable to younger individuals
- Animal longevity studies: In mice predisposed to rapid aging and high cancer risk, epithalon extended median lifespan without promoting tumor growth - a critical finding, since telomerase activation could theoretically promote cancer. The fact that no tumor promotion was observed suggests epithalon's effects may be cell-type selective (Anisimov et al., 2003; doi:10.1016/S0957-4166(03)00228-0)
- Human mortality study: A prospective cohort study of 266 people over age 60 found that treatment with epithalamin (the pineal extract from which epithalon is derived) produced a 1.6-1.8 fold reduction in mortality over the subsequent 6 years compared to controls (Khavinson & Morozov, 2003; doi:10.1159/000074622)
- Case report: In a published case report, a 79-year-old patient treated with a protocol including epithalon showed a biological age reduction of 7.9 years (from 75.93 to 68.03) and an increase in telomere length from 6.45 to 6.59 kb over 12 months
Additional Benefits: Beyond telomere maintenance, epithalon research suggests effects on circadian rhythm regulation through melatonin production modulation, antioxidant enzyme activity enhancement, neuroendocrine system support, and immune system function. These broader effects are consistent with its origin as a pineal gland peptide, given the pineal gland's role as a master regulator of circadian biology and neuroendocrine function.
Typical Protocol for Men Over 40:
| Protocol | Dose | Frequency | Cycle |
|---|---|---|---|
| Standard longevity protocol | 5-10 mg daily | SubQ injection, once daily | 10-20 day cycles, 2-3x per year |
| Intensive protocol | 10 mg daily | SubQ injection, once daily | 20-day cycle, quarterly |
| Maintenance | 5 mg daily | SubQ injection, once daily | 10-day cycle every 4-6 months |
EVIDENCE ASSESSMENT
Epithalon has more published human data than many peptides in the anti-aging space, but most of this research originates from Russian scientific institutions, with limited replication by Western research groups. The molecular mechanism (telomerase activation) is well-supported, and the safety profile from available data appears excellent. However, contemporary large-scale RCTs are lacking. Men considering epithalon should view it as a promising compound with a plausible mechanism and early supportive data, but not yet proven to the standard of conventional pharmaceuticals.
NAD+ Therapy: Fueling Cellular Energy and Repair
NAD+ (nicotinamide adenine dinucleotide) isn't technically a peptide - it's a coenzyme present in every living cell. But it's included in most peptide longevity protocols because of its fundamental role in cellular energy production and repair, and because injectable NAD+ therapy is typically administered alongside peptide compounds through the same clinical channels.
NAD+ levels decline approximately 50% between ages 40 and 60. This decline impairs mitochondrial function (reducing cellular energy), sirtuin activity (compromising DNA repair and gene expression regulation), PARP-mediated DNA repair, and inflammatory resolution pathways. The result is accelerated cellular aging across virtually every tissue type (Yoshino et al., 2018; doi:10.1016/j.cmet.2017.11.002).
Clinical Evidence: Human clinical trials with NAD+ precursors (NR and NMN) have demonstrated improvements in insulin sensitivity, vascular function, and exercise performance in older adults. Direct NAD+ IV infusion produces the highest peak blood levels (up to 380% of baseline versus 40-142% for oral precursors) and has been used clinically for addiction recovery, chronic fatigue, and anti-aging applications.
For men over 40, NAD+ therapy addresses several specific concerns:
- Exercise recovery: Enhanced mitochondrial function supports faster ATP regeneration and reduced exercise-induced oxidative damage
- Cognitive performance: NAD+ supports neuronal energy production and SIRT1-mediated neuroprotection
- Hormonal support: Adequate NAD+ levels support the enzymatic processes involved in testosterone synthesis and GH secretion
- Metabolic health: NAD+ influences insulin sensitivity through SIRT1 activation, supporting glucose metabolism
Practical NAD+ Options:
| Delivery Route | Dose | Frequency | Peak Blood Levels | Cost (Monthly) |
|---|---|---|---|---|
| IV infusion | 250-1000 mg | Weekly to monthly | 380% of baseline | $500-$2,000/session |
| SubQ injection | 50-200 mg | Daily to 3x/week | Moderate elevation | $200-$600 |
| Oral NMN | 250-1000 mg | Daily | 40-80% of baseline | $40-$150 |
| Oral NR | 300-1000 mg | Daily | 60-142% of baseline | $40-$120 |
| Nasal spray | 50-100 mg | 1-3x daily | Variable | $100-$300 |
Many clinicians recommend a "loading + maintenance" approach: begin with IV or SubQ NAD+ loading (4-8 sessions over 2-4 weeks), then transition to daily oral NMN or NR for ongoing maintenance. This provides the rapid initial restoration followed by sustained daily elevation that appears to optimize long-term outcomes.
Sexual Health Peptides for Men
Sexual dysfunction is among the most distressing symptoms of male aging - and one of the primary reasons men seek hormone and peptide therapy. While testosterone optimization addresses the hormonal foundation, specific peptides can target sexual function through distinct neural and vascular pathways.
PT-141 (Bremelanotide): The Central Desire Peptide
PT-141 (bremelanotide) represents a genuinely novel approach to sexual dysfunction. Unlike PDE5 inhibitors (Viagra, Cialis) that work through vascular mechanisms to facilitate erections, PT-141 acts centrally in the brain through melanocortin-4 (MC4) receptor activation. This distinction is clinically meaningful because PT-141 enhances sexual desire and arousal at the neurological level, not just the mechanical ability to achieve an erection (Molinoff et al., 2003; doi:10.1196/annals.1334.013).
Mechanism: MC4 receptor activation in the brain generates descending neural signals through the spinal cord to pelvic organs, increasing parasympathetic activity and producing penile erection. Because this mechanism is independent of the nitric oxide/cGMP pathway that PDE5 inhibitors target, PT-141 can work in men who don't respond to Viagra or Cialis - a substantial population that grows with age.
Clinical Evidence in Men:
- Sildenafil non-responders: In a randomized double-blind trial of 342 men with ED who had failed sildenafil, bremelanotide 10 mg intranasal produced positive clinical responses in 33.5% versus 8.5% for placebo. This is meaningful because these were men for whom the standard treatment had already failed (Diamond et al., 2005; doi:10.1016/j.juro.2007.10.063)
- Dose-response: An erectile response was statistically significant at intranasal doses above 7 mg, with onset occurring in approximately 30 minutes
- Real-world adherence: Men prescribed bremelanotide showed a 73% refill rate over 18 months - substantially higher than many sexual health medications, suggesting genuine patient-perceived benefit
- Combination efficacy: Bremelanotide combined with sildenafil in men who were inadequately responsive to sildenafil alone produced a clinically significant enhanced erectile response beyond either compound alone
FDA Status: Bremelanotide is FDA-approved as Vyleesi for hypoactive sexual desire disorder (HSDD) in premenopausal women. Its use in men is off-label but increasingly common, supported by clinical trial data in male populations.
Side Effects: The most common adverse events are nausea (reported in approximately 40% of patients, usually mild and decreasing with subsequent doses) and facial flushing. No clinically significant effects on blood pressure, heart rate, or ECG parameters have been observed - a favorable safety profile compared to some alternatives.
Typical Protocol for Men Over 40:
| Parameter | Protocol | Notes |
|---|---|---|
| Dosing | 1-2 mg SubQ injection | Start at 1 mg; increase to 2 mg if needed |
| Timing | 30-60 minutes before sexual activity | Effects last 6-12 hours |
| Frequency | As needed, max 1x per 24 hours | Not for daily use |
| Administration | SubQ injection (abdomen or thigh) | Auto-injector available for Vyleesi |
Integrating Sexual Health with the Broader Protocol
PT-141 shouldn't be viewed in isolation. For most men over 40 with sexual health concerns, the optimal approach layers interventions:
- Foundation: Optimize testosterone through peptides (gonadorelin, kisspeptin) or TRT - low testosterone is the most common cause of reduced libido after 40
- Vascular support: PDE5 inhibitors (tadalafil 2.5-5 mg daily) for men with vascular ED; also provides prostate and cardiovascular benefits
- Central desire: PT-141 for men who want enhanced desire/arousal or who don't fully respond to PDE5 inhibitors alone
- Sleep and stress: GH optimization improves sleep quality; reduced cortisol from better sleep improves sexual function
Figure 6: Multi-pathway approach to male sexual health optimization - hormonal foundation, vascular support, and central nervous system enhancement through peptide therapy.
A Deeper Look at the Hormonal Cascade of Male Aging
The endocrine changes that begin in a man's late 30s don't happen in a vacuum. They interact with metabolic, neurological, and immune systems in ways that create self-reinforcing cycles of decline. Understanding these interactions helps explain why single-target interventions often fall short - and why multi-peptide protocols tend to outperform isolated approaches.
The SHBG Problem: When Higher Testosterone Doesn't Mean More Testosterone
One of the most misunderstood aspects of testosterone decline in aging men involves sex hormone-binding globulin (SHBG). Total testosterone - the number most men focus on - tells only part of the story. SHBG is a protein produced by the liver that binds to testosterone, rendering it biologically inactive. And here's the problem: SHBG increases approximately 1-2% per year after age 40 (Vermeulen et al., 1996; doi:10.1210/jcem.81.6.8964850).
This means a man's total testosterone might decline from 600 to 480 ng/dL between ages 40 and 55 - a 20% decline that appears modest on paper. But if SHBG has risen 25% during the same period, the actual bioavailable (free) testosterone that can enter cells and produce effects has dropped by 35-40%. This is why free testosterone or calculated bioavailable testosterone is a more clinically relevant marker than total testosterone for men over 40.
Several factors accelerate SHBG increases in aging men:
- Liver function changes: The liver produces SHBG, and age-related changes in hepatic function tend to increase output
- Thyroid status: Hyperthyroidism (even subclinical) significantly elevates SHBG
- Body composition: Paradoxically, while obesity tends to lower SHBG (potentially masking low free T on total T tests), the insulin resistance associated with obesity independently suppresses testosterone production
- Medications: Statins, anticonvulsants, and some antidepressants can increase SHBG
Peptide therapy can influence this equation in several ways. GH secretagogues that improve body composition and reduce visceral fat can normalize SHBG levels. Testosterone-supporting peptides like kisspeptin increase total testosterone output, which can overcome the binding capacity of elevated SHBG. And improving metabolic health through GLP-1 agonists reduces the insulin resistance that compounds testosterone insufficiency.
Cortisol and the Stress-Hormone Axis
Chronic stress creates a hormonal environment hostile to every goal peptide therapy aims to achieve. Cortisol - the primary stress hormone - directly antagonizes testosterone, suppresses GH secretion, promotes visceral fat accumulation, impairs sleep architecture, and drives systemic inflammation. And cortisol's impact on male hormonal health worsens with age.
In younger men, the hypothalamic-pituitary-adrenal (HPA) axis recovers quickly from stress - cortisol spikes then returns to baseline. In men over 40, this recovery is slower and often incomplete, leading to chronically elevated cortisol levels. Studies have shown that mean 24-hour cortisol levels are approximately 20-50% higher in men aged 50-70 compared to men aged 20-30 (Van Cauter et al., 1996; doi:10.1210/jcem.81.7.8675571).
The interplay between cortisol and testosterone is particularly damaging. Cortisol and testosterone compete for intracellular receptor binding. When cortisol is chronically elevated, it occupies receptors that testosterone would otherwise activate, reducing testosterone's cellular effects even when blood levels appear adequate. This means a man with a "normal" testosterone level of 450 ng/dL but chronically elevated cortisol may experience symptoms of hypogonadism that his lab numbers wouldn't predict.
This cortisol-testosterone dynamic is one reason why GH secretagogues that improve sleep quality (and thus reduce overnight cortisol) can produce improvements in testosterone-related symptoms even though they don't directly affect testosterone production. Better sleep leads to lower cortisol, which leads to more effective testosterone action - a cascade that's often more impactful than directly boosting testosterone by a modest percentage.
Insulin Resistance: The Metabolic Accelerator of Hormonal Decline
Insulin resistance may be the single most important non-hormonal factor in age-related hormonal decline. And it's alarmingly common: by some estimates, over 50% of American men over 45 have some degree of insulin resistance, whether or not they've been diagnosed with pre-diabetes or type 2 diabetes (Cowie et al., 2009; doi:10.2337/dc08-1752).
Insulin resistance impacts male hormonal health through multiple mechanisms:
- Direct testosterone suppression: Insulin resistance reduces Leydig cell function in the testes, decreasing testosterone synthesis
- Increased aromatase activity: Visceral fat (which accumulates with insulin resistance) contains aromatase enzyme, converting testosterone to estrogen
- SHBG suppression: Hyperinsulinemia reduces hepatic SHBG production, which paradoxically can maintain "normal" total testosterone while actual hormonal status is deteriorating
- GH suppression: Elevated insulin and free fatty acids from insulin resistance directly suppress GH secretion from the pituitary
- Inflammatory cascade: Insulin resistance drives NF-kB-mediated inflammation, which further suppresses hypothalamic GnRH output
This is why metabolic health interventions - including semaglutide and other GLP-1 receptor agonists - can produce hormonal improvements that seem disproportionate to their direct mechanism. By improving insulin sensitivity and reducing visceral fat, these compounds remove the metabolic brakes on testosterone and GH production, allowing the body's endocrine system to function more effectively. This metabolic foundation makes all other peptide interventions more effective.
THE METABOLIC-HORMONAL CONNECTION
If you're a man over 40 with insulin resistance (fasting glucose >100 mg/dL, HbA1c >5.7%, or fasting insulin >10 mIU/L), addressing metabolic health should be your first priority - before or alongside GH and testosterone optimization peptides. Improving insulin sensitivity through lifestyle changes, GLP-1 agonists, or both will amplify the response to every other peptide in your protocol. Think of it this way: there's limited value in pressing the accelerator (GH secretagogues, testosterone peptides) if the brake (insulin resistance) is still engaged.
Neuroplasticity, BDNF, and Cognitive Decline in Aging Men
Cognitive decline is one of the most concerning aspects of male aging, and it's more closely linked to hormonal status than most men realize. Both testosterone and IGF-1 have direct neurotrophic effects - they support neuronal health, promote synaptic plasticity, and stimulate brain-derived neurotrophic factor (BDNF) production. As these hormones decline, so does the brain's capacity for neuroplasticity, learning, and memory consolidation.
Research has shown that men with lower testosterone levels have faster rates of cognitive decline and higher risk of Alzheimer's disease (Moffat et al., 2004; doi:10.1212/01.WNL.0000111640.92102.98). Similarly, low IGF-1 has been associated with reduced hippocampal volume and poorer performance on executive function tests. This dual deficiency - low T plus low IGF-1 - creates a neurological vulnerability that compounds with each passing decade.
Several peptides in the aging male's toolkit directly or indirectly support cognitive function:
- Tesamorelin: The only GH secretagogue with published RCT data specifically showing cognitive improvement in older adults and those with mild cognitive impairment (Baker et al., 2012)
- CJC-1295/Ipamorelin: By raising IGF-1, these peptides support IGF-1 receptor signaling in the hippocampus and prefrontal cortex
- NAD+: Supports neuronal energy metabolism and SIRT1-mediated neuroprotection
- Epithalon: Through melatonin regulation and antioxidant effects, may provide neuroprotective benefits
- BPC-157: Preclinical data shows neuroprotective effects in traumatic brain injury and neurotoxicity models through nitric oxide pathway modulation
For men over 50 noticing cognitive changes - difficulty with word finding, slower processing speed, impaired memory consolidation - this cognitive dimension of peptide therapy is often an underappreciated benefit. Optimizing the GH-IGF-1 axis and testosterone status can support brain health in ways that go beyond the more obvious body composition and energy effects.
Figure 9: Neuroprotective pathways supported by peptide therapy in aging men - IGF-1, testosterone, NAD+, and melatonin all contribute to brain health maintenance.
Advanced GH Optimization Strategies
Beyond selecting the right GH secretagogue, how you use these compounds matters enormously. Timing, stacking, cycling, and lifestyle synergies can dramatically affect how much benefit you extract from the same peptide at the same dose.
Timing: Why When You Inject Matters as Much as What You Inject
Growth hormone secretion follows a circadian pattern. The largest natural GH pulse occurs approximately 60-90 minutes after sleep onset, during the first period of slow-wave sleep. Smaller pulses occur throughout the night and in response to exercise, fasting, and stress. Understanding this pattern is essential for optimizing peptide timing.
Bedtime dosing (most common): Injecting CJC-1295/Ipamorelin or sermorelin 30-60 minutes before bed aligns the pharmacological GH pulse with the natural nocturnal pulse, creating an amplified and combined response. This timing also takes advantage of the fasted state (assuming you haven't eaten within 2-3 hours of bed), which further augments GH release. Most clinicians recommend this as the primary dosing time for any GH secretagogue protocol.
Morning fasted dosing (secondary): A second injection upon waking, before eating, can produce an additional GH pulse during a period when natural GH production is relatively low. This is most commonly added for men in their 50s and 60s who need more aggressive GH optimization. The key is to remain fasted for at least 30 minutes after injection, as insulin from food consumption blunts GH release.
Post-workout dosing: Some protocols include a post-workout injection, using the exercise-induced GH sensitization that occurs after resistance training. Exercise primes pituitary somatotrophs to be more responsive to GHRH stimulation, potentially amplifying the GH pulse from a secretagogue. However, many men eat a post-workout meal, and the insulin response from food can attenuate this benefit. If using post-workout timing, inject immediately after training and delay eating by 20-30 minutes.
THE INSULIN-GH ANTAGONISM RULE
Insulin and GH are physiological antagonists. When insulin is elevated (after eating), GH release is suppressed. This is why the single most important timing rule for GH secretagogues is: always inject in a fasted state. Eating within 30 minutes before or after injection significantly reduces the GH response. For bedtime dosing, stop eating at least 2-3 hours before your injection. For morning dosing, inject before breakfast and wait 20-30 minutes before eating. This simple timing discipline can double or triple the GH pulse amplitude from the same dose.
Stacking Strategies: Combining GH Secretagogues
While CJC-1295/Ipamorelin is itself a stack (GHRH analog + GHRP), some advanced protocols layer additional GH-related compounds for enhanced effects. Here are the evidence-supported combinations:
CJC-1295/Ipamorelin + MK-677: This combination uses injectable secretagogues for acute pulsatile GH release (bedtime injection) while MK-677 oral dosing provides sustained baseline GH elevation throughout the day. The rationale is that CJC/Ipa creates the acute pulses that mimic youthful GH secretion patterns, while MK-677 raises the "floor" of GH throughout the 24-hour period. This can be particularly effective for men over 55 with severely depleted GH production but requires careful glucose monitoring since both compounds can affect insulin sensitivity.
Sermorelin + GHRP-6 or GHRP-2: An older combination that's been largely superseded by CJC-1295/Ipamorelin but still used by some clinicians. The advantage of GHRP-6 is potent GH release; the disadvantage is significant hunger stimulation and cortisol/prolactin elevation - side effects that ipamorelin avoids through its selectivity.
Tesamorelin + Ipamorelin: Combining tesamorelin (GHRH analog with FDA approval) with ipamorelin (selective GHRP) follows the same dual-receptor logic as CJC-1295/Ipamorelin but uses a compound with regulatory approval and targeted visceral fat data. This combination is gaining traction among clinicians who prefer FDA-approved compounds where possible.
Cycling: Preventing Desensitization
A persistent question in GH secretagogue therapy is whether continuous use leads to pituitary desensitization - where the somatotroph cells become less responsive to stimulation over time, reducing the GH response. The evidence on this question is mixed:
- MK-677: The 12-month Nass trial showed sustained IGF-1 elevation throughout the study period, suggesting minimal desensitization with continuous oral use at 25 mg daily
- CJC-1295 (with DAC): Extended half-life formulations may carry higher desensitization risk due to continuous receptor stimulation, though this hasn't been conclusively demonstrated
- CJC-1295 (without DAC): Shorter-acting formulations that clear between doses are theoretically less likely to cause desensitization since receptor occupancy is intermittent
- Sermorelin: Long-term studies showed maintained GH response over months of use, though some practitioners report diminishing subjective benefits after 4-6 months
In clinical practice, most prescribers use one of two cycling approaches:
| Cycling Protocol | Structure | Rationale |
|---|---|---|
| Weekly cycling | 5 days on / 2 days off (typically weekdays on, weekends off) | Regular receptor breaks; easy to remember; allows natural GH pulses on off days |
| Monthly cycling | 3-4 weeks on / 1 week off | Longer continuous therapy for maximum effect; monthly break prevents cumulative desensitization |
| Seasonal cycling | 3 months on / 1 month off | Used for long-term protocols; major reset period |
| Continuous use | No breaks (MK-677 primarily) | Based on Nass trial data showing sustained response; requires ongoing monitoring |
The practical recommendation for most men over 40 starting GH secretagogue therapy is to begin with continuous use for the first 3 months (to establish response and reach steady-state IGF-1 levels), then transition to a cycling protocol based on ongoing IGF-1 monitoring and subjective response.
Lifestyle Amplifiers: Maximizing GH Response Beyond Peptides
Certain lifestyle practices amplify the GH response from secretagogues, while others suppress it. Understanding these interactions can effectively double the clinical benefit of the same peptide protocol:
Amplifiers:
- Resistance training: High-intensity resistance exercise increases GH secretion independently and sensitizes pituitary somatotrophs to subsequent GHRH stimulation. Compound movements (squats, deadlifts, presses) at 70-85% of 1RM produce the strongest GH response
- Intermittent fasting: Fasting periods of 14-16 hours increase baseline GH secretion by 2-5 fold. When combined with an evening GH secretagogue injection during the fast, the combined effect can be substantial
- Sleep optimization: As discussed earlier, deep sleep is when the largest GH pulse occurs. Sleep quality directly determines how much benefit you get from bedtime peptide dosing
- Cold exposure: Brief cold exposure (cold shower, ice bath) triggers a catecholamine and GH response. Some practitioners time cold exposure 15-20 minutes before their bedtime peptide injection
- Arginine supplementation: L-arginine (5-9g on an empty stomach) potentiates GH release from GHRH stimulation through somatostatin suppression
Suppressors (avoid around injection time):
- High-glycemic meals: Insulin spikes directly suppress GH release for 2-3 hours
- Alcohol: Alcohol disrupts slow-wave sleep and suppresses nocturnal GH secretion by up to 75%
- Chronic stress/elevated cortisol: Cortisol suppresses GH release at the hypothalamic level
- Excess body fat: Visceral adiposity creates a GH-suppressive environment through elevated free fatty acids and insulin
- Chronic sleep deprivation: Even 2-3 nights of poor sleep can reduce GH output by 40-70%
Advanced Testosterone Optimization: Beyond the Basics
While gonadorelin and kisspeptin provide the peptide foundation for testosterone support, optimizing testosterone in men over 40 requires understanding the full ecosystem of factors that influence production, metabolism, and cellular action.
The Aromatase Factor: When Testosterone Becomes Estrogen
Aromatase is the enzyme that converts testosterone to estradiol (estrogen). In young men with low body fat, aromatase activity is relatively low and the testosterone-to-estrogen ratio remains favorable. But several age-related changes shift this balance:
- Increasing body fat: Adipose tissue (especially visceral fat) is the primary site of aromatase expression. As body fat increases with age, aromatase activity rises proportionally
- Age-related enzyme induction: Aromatase expression increases with age independent of body fat, likely due to inflammatory cytokines that upregulate the CYP19 gene
- Alcohol consumption: Ethanol directly induces aromatase activity in adipose tissue and the liver
- Environmental exposures: Certain pesticides, plasticizers (BPA, phthalates), and heavy metals can induce aromatase expression
The clinical consequence is that some men over 40 have adequate testosterone production but excessive conversion to estrogen, resulting in a clinical picture that looks like low testosterone (fatigue, reduced libido, fat accumulation, mood changes) despite "normal" total T levels. The estradiol-to-testosterone ratio becomes the critical marker in these cases.
Peptide-based approaches to managing aromatase include reducing visceral fat through GH secretagogues and GLP-1 agonists (removing the aromatase-producing tissue), improving insulin sensitivity (hyperinsulinemia promotes aromatase expression), and in some cases combining peptide therapy with DIM (diindolylmethane) or calcium-D-glucarate for estrogen metabolism support.
Leydig Cell Function and Age
The Leydig cells in the testes are responsible for testosterone production, and their function declines with age independently of hormonal signaling. Even when appropriately stimulated by LH (whether from natural production, gonadorelin, or kisspeptin), aging Leydig cells produce less testosterone per unit of LH stimulation than younger cells. This is called primary hypogonadism when severe, and it explains why some men over 60 show blunted testosterone responses to peptide therapy that worked well in their 40s and 50s (Chen et al., 2009; doi:10.1016/j.mce.2008.11.009).
Factors that accelerate Leydig cell aging include oxidative stress (hence the potential benefit of NAD+ and antioxidant support), chronic inflammation (addressable through lifestyle and anti-inflammatory peptides), vascular insufficiency to the testes (a reason to maintain cardiovascular health), and environmental toxin exposure. Supporting Leydig cell health through antioxidant supplementation (CoQ10, vitamin E, zinc), adequate sleep, and reduced toxin exposure can improve the testosterone response to peptide therapy.
Free Testosterone: The Number That Actually Matters
As discussed in the SHBG section, free testosterone - not total testosterone - determines clinical effect. Approximately 2% of circulating testosterone is free (unbound), 40-50% is loosely bound to albumin (and considered bioavailable), and the remainder is tightly bound to SHBG and inactive. Only free and albumin-bound testosterone can enter cells and activate androgen receptors.
For men on peptide-based testosterone optimization, monitoring should include both total and free testosterone (or calculated bioavailable testosterone). A man with total T of 500 ng/dL but very high SHBG may have free T equivalent to a man with total T of 350 ng/dL and normal SHBG. The symptoms and clinical picture will match the free T, not the total.
Target ranges for free testosterone in optimized men over 40 are generally 15-25 pg/mL (by equilibrium dialysis) or the upper third of the age-adjusted reference range used by your laboratory.
Injury Prevention and Recovery Optimization After 40
For active men over 40, injury prevention is as important as injury treatment. The shift from reactive healing to proactive tissue maintenance represents a fundamental change in how you should approach your body's repair systems.
Understanding Why Recovery Slows With Age
Recovery after exercise or injury slows with age for specific, identifiable reasons - not simply because you're "getting old." Understanding these mechanisms helps target peptide and lifestyle interventions more precisely:
- Reduced angiogenesis: New blood vessel formation at injury sites is slower in older tissue, reducing nutrient and oxygen delivery to damaged areas. BPC-157 directly addresses this through VEGFR2-mediated angiogenesis promotion
- Impaired satellite cell activation: Skeletal muscle repair depends on satellite cells - tissue-resident stem cells that proliferate and differentiate into new muscle fibers. Satellite cell function declines with age, reducing the body's capacity for muscle repair after damage (Conboy et al., 2005; doi:10.1038/nature03260)
- Increased fibrosis: Older tissue tends to repair with scar tissue (fibrosis) rather than functional tissue regeneration. TB-500 may help counter this by influencing collagen deposition patterns and reducing fibrotic scarring
- Prolonged inflammatory response: In younger individuals, inflammation after injury resolves quickly, transitioning to a repair phase. In older adults, the inflammatory phase persists longer, delaying healing and potentially causing collateral tissue damage
- Hormonal support reduction: Both GH/IGF-1 and testosterone are essential for tissue repair. Their decline directly impairs the body's healing capacity
- Reduced NAD+ availability: Cellular energy for repair processes depends on NAD+, which declines by 50% between ages 40 and 60
Proactive Maintenance: The Shift from Treatment to Prevention
For men over 40, the most effective recovery strategy is preventing tissue breakdown from exceeding repair capacity in the first place. This "maintenance" approach uses peptides differently than acute injury treatment:
| Strategy | Implementation | Target Tissue |
|---|---|---|
| Baseline GH optimization | CJC-1295/Ipamorelin nightly, continuous | All connective tissue, muscle, cartilage |
| Periodic BPC-157 cycles | 250 mcg daily for 4-6 weeks every 3-4 months | Tendons, ligaments, GI tract |
| Periodic TB-500 cycles | 750 mcg 2x/week for 4-6 weeks every 4-6 months | Systemic tissue maintenance, joint health |
| Collagen support | Vitamin C + collagen peptides (15-20g) before exercise | Tendons, ligaments, cartilage synthesis |
| Training periodization | Deload weeks every 4th week; volume cycling | Prevents overuse injury accumulation |
The distinction between "treatment dose" and "maintenance dose" is clinically relevant. During acute injury, higher doses and more frequent administration are appropriate. For preventive maintenance, lower doses cycled periodically may be sufficient to keep repair capacity ahead of damage accumulation.
Joint-Specific Considerations for Men Over 40
Joint health deserves special attention because cartilage has limited regenerative capacity, and the cumulative effects of decades of use, micro-trauma, and declining GH/IGF-1 make joint deterioration one of the most common reasons men over 40 reduce physical activity - which then accelerates every other aspect of aging.
Specific peptide considerations for joint health:
- BPC-157: Has shown chondroprotective effects in preclinical models, promoting cartilage repair and reducing inflammatory damage in joint tissue
- GH secretagogues: IGF-1 is essential for chondrocyte (cartilage cell) survival and matrix production. Restoring IGF-1 to youthful ranges supports ongoing cartilage maintenance
- TB-500: Promotes cell migration and tissue remodeling in joint capsule and synovial tissue
- Collagen peptides: While not a prescription peptide, supplemental collagen peptides (especially type II collagen) have RCT evidence for joint health improvement in active adults
For men with existing joint issues (mild osteoarthritis, chronic tendinopathy, post-surgical joints), a protocol combining systemic GH optimization with targeted BPC-157 (injected near the affected joint) can provide both the systemic repair environment and localized healing support. This combination is among the most commonly prescribed peptide protocols in sports medicine and orthopedic settings.
Figure 10: Recovery capacity trajectory with age - natural decline versus peptide-supported maintenance showing preserved healing rates.
The Science of Male Longevity: What Peptides Can and Cannot Do
Longevity science has matured significantly in recent years, moving from speculative theories to measurable biomarkers and interventions with human evidence. For men over 40, the question isn't whether aging can be slowed - the evidence clearly shows it can be - but which interventions provide the most return on investment at each stage of life.
The Hallmarks of Aging and Peptide Targets
Modern aging biology recognizes twelve "hallmarks of aging" - molecular and cellular processes that drive aging across tissues and organisms. Understanding which hallmarks peptide therapy addresses (and which it doesn't) provides a realistic framework for expectations (Lopez-Otin et al., 2023; doi:10.1016/j.cell.2022.11.001):
| Hallmark of Aging | Peptide/Intervention | Evidence Level |
|---|---|---|
| Telomere attrition | Epithalon (telomerase activation) | Moderate - human cell data, animal lifespan data, limited human RCTs |
| Mitochondrial dysfunction | NAD+ (mitochondrial fuel) | Strong - multiple human RCTs with precursors |
| Cellular senescence | NAD+ (SIRT1 activation reduces senescence) | Moderate - preclinical + early human data |
| Loss of proteostasis | GH optimization (supports protein turnover) | Moderate - clinical observation data |
| Deregulated nutrient sensing | GLP-1 agonists (improve insulin signaling) | Strong - multiple Phase III RCTs |
| Stem cell exhaustion | TB-500 (stem cell mobilization) | Low-moderate - primarily preclinical |
| Altered intercellular communication | BPC-157 (growth factor signaling) | Low-moderate - primarily preclinical |
| Genomic instability | NAD+ (PARP-mediated DNA repair) | Moderate - mechanism well-established |
| Epigenetic alterations | Lifestyle + hormonal optimization | Emerging - epigenetic clock data preliminary |
| Chronic inflammation | GLP-1 agonists, GH optimization, lifestyle | Strong - established anti-inflammatory effects |
| Disabled macroautophagy | Fasting + NAD+ (autophagy induction) | Moderate - mechanism established |
| Dysbiosis | BPC-157 (GI mucosal support) | Low-moderate - primarily preclinical |
This table reveals an important reality: no single peptide addresses all hallmarks of aging. Effective longevity protocols layer multiple compounds that address different hallmarks, combined with lifestyle interventions (exercise, nutrition, sleep, stress management) that influence hallmarks no peptide can fully reach.
Epigenetic Age Testing: Measuring Your Biological Age
One of the most promising developments in longevity medicine is the ability to measure biological age through epigenetic clock testing. These tests analyze DNA methylation patterns across specific CpG sites to calculate a "biological age" that may differ from chronological age by years or even decades.
Several commercial epigenetic age tests are now available to consumers, and they provide a measurable endpoint for longevity interventions. Men starting comprehensive peptide protocols can test before initiation and at 6-12 month intervals to track whether their biological age is progressing faster or slower than chronological time.
Early data from individual case reports and small clinical series suggest that comprehensive anti-aging protocols (including peptides, lifestyle optimization, and targeted supplementation) can slow or even reverse epigenetic aging by 1-5 years over 12-24 months of consistent therapy. The 79-year-old case report mentioned in the epithalon section showed a 7.9-year biological age reduction over 12 months, though this was a multi-modal intervention, not epithalon alone.
The Longevity Pyramid: Building a Sustainable Protocol
Think of longevity intervention as a pyramid. The foundation layers provide the most return per dollar and effort invested, while higher layers add incremental benefit on top of a solid foundation:
Level 1 - Foundation (Non-negotiable):
- Resistance training 3-4x/week
- 7-9 hours of quality sleep
- Adequate protein (1.6-2.2 g/kg)
- Stress management
- Alcohol limitation
- Metabolic health (insulin sensitivity, healthy body composition)
Level 2 - Hormonal Optimization:
- GH secretagogue (CJC-1295/Ipamorelin or equivalent)
- Testosterone optimization (gonadorelin, kisspeptin, or TRT)
- Thyroid optimization (if indicated)
Level 3 - Cellular Health:
- NAD+ supplementation (oral NMN/NR or injectable)
- Epithalon cycles (telomere maintenance)
- Targeted antioxidant support
Level 4 - Specific Optimization:
- GLP-1 agonist (if metabolic health warrants)
- Recovery peptides (BPC-157, TB-500)
- Sexual health peptides (PT-141)
- Cognitive support compounds
Level 5 - Advanced Interventions:
- Epigenetic age monitoring and protocol adjustment
- Advanced biomarker panels (oxidative stress markers, inflammatory cytokine panels)
- Emerging compounds as evidence accumulates
Men who try to build from the top of the pyramid without establishing the foundation will get disappointing results. The most expensive peptide protocol in the world won't compensate for poor sleep, sedentary behavior, and a diet of processed food. Conversely, men who nail the foundation and add even basic peptide optimization often achieve results that exceed expectations.
For comprehensive guidance on building your first peptide protocol, our peptide therapy beginner's guide walks through each step, and the peptide research hub provides in-depth reports on each compound discussed here.
Body Composition After 40: The Science of Sarcopenia, Visceral Fat, and Metabolic Health
Body composition changes after 40 aren't just about how you look in the mirror. Sarcopenia (age-related muscle loss), visceral fat accumulation, and the metabolic disruption they produce are among the strongest predictors of disability, disease, and mortality in aging men. Understanding the specific mechanisms driving these changes reveals why peptide therapy can be so effective - and where its limitations lie.
Sarcopenia: The Silent Erosion of Muscle Mass
Beginning around age 30, men lose approximately 3-8% of their muscle mass per decade, with the rate accelerating after age 60 (Volpi et al., 2004; doi:10.1093/ajcn/80.3.475). But this average obscures important variation. Physically active men with adequate protein intake and optimized hormones can maintain muscle mass well into their 60s and 70s. Sedentary, poorly nourished men with declining hormones can lose 10-15% of their muscle mass per decade.
The mechanisms driving sarcopenia are well-characterized:
- Motor neuron loss: Aging reduces the number of motor neurons innervating muscle fibers. Lost motor neurons cause their associated muscle fibers to atrophy and eventually disappear. This process is most pronounced in fast-twitch (Type II) fibers - the ones responsible for strength and power
- Anabolic resistance: Older muscle becomes less responsive to the anabolic signals from protein consumption and exercise. Where a young man might maximally stimulate muscle protein synthesis with 20g of protein post-exercise, an older man may need 35-40g for the same response (Moore et al., 2015; doi:10.1016/j.nutres.2014.10.018)
- Hormonal decline: Testosterone stimulates muscle protein synthesis via androgen receptor activation. GH/IGF-1 supports satellite cell proliferation and muscle repair. Both hormones' decline directly reduces the anabolic milieu in aging muscle
- Chronic inflammation: Elevated TNF-alpha, IL-6, and other inflammatory cytokines directly promote muscle protein breakdown through the ubiquitin-proteasome pathway
- Mitochondrial dysfunction: Reduced mitochondrial function in aging muscle impairs energy production and increases oxidative stress, contributing to muscle fiber damage and loss
Peptide therapy addresses sarcopenia through multiple mechanisms simultaneously. GH secretagogues restore IGF-1 signaling that supports satellite cell function and muscle repair. Testosterone optimization through gonadorelin or kisspeptin enhances muscle protein synthesis rates. NAD+ supplementation supports mitochondrial function in aging muscle. And GLP-1 agonists can reduce the inflammatory burden from visceral fat that drives muscle catabolism.
However, peptides alone won't reverse sarcopenia. The most critical intervention is progressive resistance training, which remains the most potent stimulus for muscle maintenance and growth at any age. Peptides amplify the response to training - they don't replace it. A man on a comprehensive peptide protocol who doesn't train will see significantly less benefit than a man training seriously with no peptides at all.
Visceral Fat: The Metabolically Dangerous Fat
Not all body fat is created equal, and this distinction becomes critical after 40. Subcutaneous fat (under the skin) is relatively metabolically inactive. Visceral fat (surrounding internal organs in the abdominal cavity) is a different matter entirely. It's an active endocrine organ that produces inflammatory cytokines, promotes insulin resistance, converts testosterone to estrogen via aromatase, suppresses GH secretion, and increases cardiovascular disease risk independently of total body weight.
Men are particularly susceptible to visceral fat accumulation due to the pattern of fat distribution driven by declining testosterone. As testosterone drops, the body preferentially stores fat viscerally rather than subcutaneously - creating the characteristic "beer belly" or "apple shape" that many men develop in their 40s and 50s. This visceral fat then further suppresses testosterone through aromatase activity, creating a vicious cycle that's difficult to break through diet and exercise alone.
Peptide approaches to visceral fat reduction:
| Compound | Mechanism Against Visceral Fat | Evidence Level | Expected Reduction |
|---|---|---|---|
| Semaglutide | Appetite reduction, improved insulin sensitivity, direct effects on adipose tissue | Strong (Phase III RCTs) | 20-30% VAT reduction over 68 weeks |
| Tesamorelin | Targeted GH-mediated lipolysis in visceral adipose | Strong (FDA-approved for this indication) | 15-18% VAT reduction over 26 weeks |
| CJC-1295/Ipamorelin | Increased GH promotes lipolysis; shifts fuel utilization toward fat oxidation | Moderate (clinical observation) | Variable; supports fat loss alongside training |
| MK-677 | GH-mediated improvements in fat-free mass; metabolic rate increase | Moderate (12-month RCT) | Lean mass increase of 1.1 kg; modest fat reduction |
| Testosterone optimization | Reduces visceral fat through improved androgen receptor signaling and metabolic rate | Strong (multiple TRT trials) | 10-20% VAT reduction with normalized T |
The most effective strategy for visceral fat reduction combines multiple approaches. A protocol using a GLP-1 agonist for appetite and metabolic control, a GH secretagogue for lipolysis and lean mass preservation, testosterone optimization for metabolic rate and body composition, and structured resistance training can produce visceral fat reductions of 25-40% over 6-12 months - far more than any single intervention achieves alone.
The Muscle-Fat Axis: Why Recomposition Beats Weight Loss
For men over 40, the goal should be body "recomposition" - simultaneously gaining muscle while losing fat - rather than simply losing weight on a scale. Pure weight loss (through caloric restriction alone) invariably includes significant muscle loss, which reduces metabolic rate, impairs functional capacity, and accelerates the sarcopenia that's already underway. A man who loses 20 pounds but 30% of it is muscle may actually be in a worse metabolic position than before.
Body recomposition is physiologically challenging because muscle growth and fat loss have competing metabolic requirements. Muscle growth favors a caloric surplus and anabolic hormones; fat loss requires a caloric deficit and catabolic signaling. But peptide therapy helps bridge this gap because GH secretagogues independently promote both lipolysis (fat breakdown) and muscle protein synthesis, effectively decoupling the normally antagonistic processes of fat loss and muscle gain.
Clinical data supports this. The MK-677 12-month trial showed that subjects gained 1.1 kg of lean mass while simultaneously losing fat mass, with no change in total body weight. Similar recomposition effects are observed with testosterone optimization - men who normalize their testosterone experience fat loss and muscle gain that roughly offset each other on the scale, creating a misleading impression of "no results" when only tracking weight.
This is why waist circumference, body composition scans (DEXA), and visual assessment are better metrics than scale weight for men on peptide protocols targeting body composition. A man whose weight doesn't change but whose waist drops 3 inches and DEXA shows 4 lbs more muscle and 4 lbs less fat has had an extraordinary result - even though the scale says nothing happened.
Comprehensive Sexual Health After 40: Beyond Erectile Function
Sexual health encompasses far more than erectile function alone. Desire, arousal, orgasmic function, satisfaction, and the psychological components of sexual well-being all change with age - and different peptides target different aspects of this complex system.
The Four Pillars of Male Sexual Function
Understanding where your sexual health challenges originate determines which interventions will help:
1. Hormonal Foundation (Desire/Libido)
Testosterone is the primary driver of male sexual desire. When testosterone drops below approximately 300-400 ng/dL, most men notice reduced spontaneous sexual thoughts, decreased morning erections, and diminished interest in sexual activity. This is distinct from erectile dysfunction - a man with low desire may be physically capable of erection but simply lacks the motivation.
Peptide interventions: Gonadorelin, kisspeptin-10, or enclomiphene to optimize endogenous testosterone production. For men already on TRT, ensuring adequate dosing and estrogen management.
2. Vascular Function (Erectile Quality)
Erection depends on adequate arterial blood flow to the corpus cavernosum - and vascular health declines with age due to endothelial dysfunction, atherosclerosis, and reduced nitric oxide production. Approximately 40% of men over 40 have some degree of erectile dysfunction, rising to 70% by age 70 (Feldman et al., 1994; doi:10.1111/j.1743-6109.2007.00462.x).
Peptide interventions: GH optimization improves endothelial function and vascular health. Semaglutide's cardiovascular benefits may indirectly support erectile function through vascular improvement. BPC-157's nitric oxide-enhancing effects may support local vascular function. These complement PDE5 inhibitors (tadalafil/sildenafil) rather than replacing them.
3. Neural Drive (Arousal)
The central nervous system generates arousal signals that translate desire into physical response. This neural pathway involves dopaminergic and melanocortinergic circuits that decline with age. PT-141 (bremelanotide) directly activates melanocortin-4 receptors in these brain circuits, enhancing the neural drive for sexual arousal independent of both hormonal status and vascular function.
This is why PT-141 is particularly valuable for men over 50 who have optimized testosterone and tried PDE5 inhibitors but still don't experience the desire and arousal they want. It addresses a neurological dimension that other interventions don't reach.
4. Psychological and Relational Factors
No peptide addresses the psychological components of sexual health - stress, relationship dynamics, performance anxiety, depression, and body image concerns. These factors become increasingly relevant with age and can undermine even perfectly optimized hormones and vascular function. Men struggling with sexual health should consider that comprehensive support may include counseling or therapy alongside biological optimization.
The Sexual Health Protocol: Layered Approach
| Layer | Intervention | Timeline to Benefit | What It Addresses |
|---|---|---|---|
| Foundation | Testosterone optimization (gonadorelin/kisspeptin) | 4-12 weeks | Desire, energy, spontaneous erections |
| Vascular | Daily low-dose tadalafil (2.5-5 mg) + GH optimization | 2-4 weeks (tadalafil); 8-12 weeks (GH) | Erectile quality, penile blood flow |
| Neural | PT-141 (as needed, 1-2 mg SubQ) | 30-60 minutes per use | Central arousal, desire enhancement |
| Metabolic | Body composition optimization (reduce visceral fat) | 3-6 months | Reduces estrogen conversion, improves energy |
| Sleep | GH secretagogue timing + sleep optimization | 1-4 weeks | Hormonal restoration, energy, morning erections |
Cardiovascular Risk in Aging Men: The Peptide Perspective
Heart disease kills more men over 40 than any other cause. While peptide therapy is not a primary cardiac treatment, understanding how various peptides affect cardiovascular risk factors helps men and their clinicians make informed decisions about protocol design.
The Male Cardiovascular Risk Landscape After 40
Men face substantially higher cardiovascular risk than women at every age until the late 70s, when rates begin to equalize. After 40, cardiovascular risk factors compound: LDL cholesterol increases, blood pressure rises, arterial stiffness progresses, visceral fat accumulation accelerates inflammation, insulin resistance impairs glucose metabolism and promotes atherosclerosis, and declining testosterone and GH contribute to adverse lipid profiles and reduced cardiac function.
The traditional risk assessment model focuses on lipids, blood pressure, glucose, and family history. But emerging evidence suggests that hormonal status - particularly testosterone and GH levels - may be independent risk factors that current models undervalue.
How Specific Peptides Affect Cardiovascular Risk Markers
GLP-1 Agonists (Semaglutide): The strongest cardiovascular evidence in the peptide space comes from GLP-1 receptor agonists. Beyond the SELECT trial's 20% MACE reduction, semaglutide and other GLP-1 agonists have shown reductions in blood pressure (4-6 mmHg systolic), improvements in lipid profiles (reduced triglycerides, small improvements in LDL), reduced markers of systemic inflammation (hsCRP reduction of 20-40%), reduced hepatic steatosis (fatty liver) with potential downstream cardiovascular benefits, and improved endothelial function markers. For men over 50 with elevated cardiovascular risk who also need body composition improvement, semaglutide addresses both concerns simultaneously - a rare dual benefit in medicine.
GH Secretagogues: The cardiovascular effects of GH optimization are dose-dependent and bidirectional. Moderate GH restoration (to youthful normal ranges) appears beneficial - improving lipid profiles, reducing visceral fat, and enhancing cardiac contractility. However, excessive GH elevation (supraphysiological) can promote cardiomegaly, insulin resistance, and fluid retention. This dose-dependency is why secretagogues that produce physiological pulsatile release are preferred over exogenous GH for cardiovascular safety.
Specific cardiovascular benefits observed with GH secretagogue therapy in clinical studies include LDL cholesterol reduction of 10-20%, triglyceride reduction associated with visceral fat loss, improved left ventricular ejection fraction in GH-deficient adults, reduced arterial stiffness (measured by pulse wave velocity), and improved exercise capacity and VO2 max.
Testosterone Optimization: The TRAVERSE trial conclusively demonstrated that testosterone replacement in hypogonadal men with elevated cardiovascular risk did not increase MACE events over a median of 33 months. This was the largest randomized trial ever conducted on testosterone safety, involving over 5,200 men, and it should put to rest the earlier concerns raised by smaller, methodologically flawed studies. For peptide-based testosterone optimization (which produces more modest elevations), the cardiovascular safety profile is likely even more favorable.
Additional cardiovascular benefits of testosterone optimization include reduced visceral fat (an independent cardiovascular risk factor), improved insulin sensitivity, potential improvements in endothelial function, enhanced exercise capacity and cardiac output, and possible anti-inflammatory effects through reduced visceral fat burden.
Cardiovascular Monitoring for Men on Peptide Protocols
Men over 40 on peptide therapy should maintain regular cardiovascular surveillance. Specific monitoring recommendations include blood pressure measurement at every clinic visit (and home monitoring for men with borderline values), advanced lipid panel (ApoB, LDL-P, Lp(a)) at baseline and every 6-12 months, hsCRP to track systemic inflammation, fasting glucose and HbA1c (especially with MK-677), hematocrit monitoring (testosterone can increase red blood cell production, raising viscosity and clot risk), echocardiogram at baseline for men over 50 starting GH therapy (to establish cardiac structure), and exercise stress testing as clinically indicated.
Men with established cardiovascular disease or significant risk factors should coordinate peptide therapy with their cardiologist. Most peptides used in anti-aging medicine don't pose direct cardiovascular risks at therapeutic doses, but the importance of comprehensive medical oversight increases with pre-existing cardiac conditions.
Practical Implementation Guide: Getting Started the Right Way
Theory is useful, but execution determines outcomes. This section provides the practical framework for translating the science above into a real-world peptide protocol that fits into your life.
Step 1: Comprehensive Assessment
Before injecting anything, you need data. The comprehensive baseline panel outlined in the Blood Work section should be completed, along with a thorough health history, medication review, and physical examination. Key questions to address with your clinician:
- What are your primary symptoms? (fatigue, body composition, recovery, sexual health, sleep, cognitive concerns)
- What is your current testosterone and IGF-1 status?
- Do you have any contraindications? (active cancer, uncontrolled diabetes, pituitary disorders)
- What medications are you currently taking?
- What is your fitness level and training history?
- Do you have a family history of prostate cancer or cardiovascular disease?
- What is your budget and commitment level for ongoing therapy?
Step 2: Priority Setting
Based on your assessment, identify 1-2 primary objectives for your first peptide protocol. Trying to address everything at once leads to complicated protocols, difficulty isolating what's working, and higher cost. Common starting priorities include:
- "I need better sleep and recovery": Start with CJC-1295/Ipamorelin before bed
- "My testosterone is declining and I want to avoid TRT": Start with gonadorelin or kisspeptin, guided by lab values
- "I can't lose this belly fat despite training and diet": Consider tesamorelin or semaglutide, based on BMI and metabolic markers
- "I have chronic tendon issues that won't heal": Start with BPC-157 targeted to the injury
- "I'm focused on longevity and cellular health": Begin with NAD+ supplementation (oral NMN) and consider epithalon cycles
Step 3: Source Quality Peptides
Work with your prescribing clinician to obtain peptides from a licensed compounding pharmacy that follows USP 797/800 sterile compounding standards, provides certificates of analysis (COA) with each batch, uses third-party testing to verify identity, purity, and potency, and ships with appropriate cold chain management for temperature-sensitive compounds. Never purchase peptides from "research chemical" websites, social media sellers, or overseas vendors without regulatory oversight. The risk of contamination, mislabeling, or under-dosing is substantial and can range from ineffective therapy to genuine health risks.
Step 4: Learn Proper Injection Technique
Most peptides require subcutaneous injection - a simple technique that uses a short insulin needle (29-31 gauge, 1/2 inch) inserted at a 45-90 degree angle into pinched skin, typically in the abdominal area. Your clinician or their staff should demonstrate proper technique, but the key principles include:
- Wash hands thoroughly before preparing injection
- Swab the vial stopper and injection site with alcohol
- Draw the correct dose carefully (confirm units and concentration)
- Pinch a fold of skin and insert the needle at a 45-90 degree angle
- Inject slowly and hold for 5-10 seconds before withdrawing
- Rotate injection sites to prevent lipodystrophy (hardening at repeated injection locations)
- Store reconstituted peptides in the refrigerator (most are stable for 28-30 days once reconstituted)
Step 5: Track, Monitor, and Adjust
Effective peptide therapy requires ongoing monitoring and adjustment. Key tracking practices include:
- Symptom journal: Track sleep quality, energy, recovery, libido, mood, and other relevant symptoms weekly
- Body composition: Monthly waist circumference measurement; DEXA scan at baseline and 3-6 month intervals if body composition is a primary goal
- Blood work: Follow the monitoring schedule outlined in the Blood Work section
- Side effect reporting: Document any adverse effects and report them to your prescribing clinician promptly
- Dose adjustments: Based on lab results and clinical response, your clinician will adjust doses - expect at least one dose modification in the first 3 months as your individual response becomes clear
Step 6: Build the Protocol Over Time
Start with one compound and add additional peptides sequentially rather than launching a complex multi-compound protocol from day one. This approach allows you to identify how each compound affects you individually, isolate side effects (if any) to specific compounds, build confidence with injection technique and routine before adding complexity, and control costs while confirming that each addition provides measurable value.
A reasonable build-out timeline might look like:
| Month | Action | Lab Check |
|---|---|---|
| Month 1 | Start primary compound (e.g., CJC-1295/Ipamorelin) | Baseline comprehensive panel |
| Month 2 | Assess response; adjust dose if needed | 4-6 week labs (IGF-1, T, glucose) |
| Month 3 | Add second compound if indicated (e.g., gonadorelin) | 12-week comprehensive panel |
| Month 4-5 | Optimize doses of both compounds | Interim labs as needed |
| Month 6 | Full reassessment; consider adding third compound if goals not met | 6-month comprehensive panel |
This measured approach may feel slower than the "everything at once" protocols promoted by some clinics, but it produces better outcomes because you understand what each compound is doing and can make informed decisions about what to continue, adjust, or discontinue.
Ready to get started? Visit our getting started page for step-by-step guidance, or explore the peptide research hub for detailed information on any compound discussed in this report.
Figure 11: Six-step implementation pathway for men starting peptide therapy - from initial assessment through ongoing optimization.
Circadian Biology and Male Aging: The Master Clock Connection
Your body's circadian system isn't just about when you feel sleepy. It's a master regulatory network that controls hormone secretion timing, metabolic efficiency, immune function, and even DNA repair. Age-related circadian disruption is one of the most underappreciated drivers of male hormonal decline - and one of the most addressable through peptide therapy and lifestyle intervention.
How Circadian Rhythm Deteriorates With Age
The suprachiasmatic nucleus (SCN) in the hypothalamus serves as the body's master clock, coordinating the timing of hormonal secretion, metabolic activity, and sleep-wake cycles. With age, the SCN deteriorates structurally and functionally, leading to weaker circadian signals, reduced amplitude of hormonal rhythms, earlier timing of sleep onset (the "phase advance" that makes older adults wake earlier), fragmented sleep architecture, and reduced melatonin production from the pineal gland (Duffy et al., 2002; doi:10.1152/ajpregu.00543.2001).
These circadian changes directly impact the hormones most relevant to aging men. Testosterone secretion follows a circadian pattern, peaking in the early morning (6-8 AM) and reaching its nadir in the evening. In young men, the morning peak is 25-50% higher than the evening trough. In men over 60, this circadian variation is significantly blunted - morning testosterone may only be 10-15% above evening levels, contributing to lower overall 24-hour testosterone exposure. Some researchers believe this loss of circadian testosterone variation may be as important as the decline in average levels (Bremner et al., 1983; doi:10.1210/jcem-56-6-1278).
Growth hormone secretion is even more tightly linked to sleep timing. The largest GH pulse occurs during the first episode of slow-wave sleep, approximately 60-90 minutes after sleep onset. When sleep is delayed, fragmented, or lacks adequate slow-wave stages, this GH pulse is diminished or absent entirely. Since slow-wave sleep declines by about 2% per decade after age 40, the nocturnal GH pulse progressively weakens - explaining why even men with functional pituitary glands experience somatopause.
Melatonin: The Circadian Hormone That Connects Everything
Melatonin production from the pineal gland declines dramatically with age. By age 60, most men produce only 10-20% of the melatonin they produced at age 20. This decline has cascading effects beyond sleep, influencing antioxidant defense (melatonin is a potent antioxidant that neutralizes reactive oxygen species), immune regulation (melatonin modulates immune cell function and inflammatory responses), hormonal timing (melatonin helps synchronize the circadian release of testosterone, cortisol, and GH), and neuroprotection (melatonin protects neurons from oxidative damage and supports neuroplasticity).
Epithalon's connection to melatonin production is one of its most clinically relevant mechanisms for aging men. By supporting pineal gland function and melatonin synthesis, epithalon may help restore circadian signaling that coordinates the timing of multiple hormonal rhythms. This upstream circadian effect could explain why some men report improvements across multiple domains (sleep, energy, mood, recovery) during epithalon cycles, even though epithalon doesn't directly affect testosterone or GH levels.
Practical Circadian Optimization for Men Over 40
Optimizing circadian function amplifies every other intervention in your peptide protocol. Here's a systematic approach:
Morning light exposure (within 30 minutes of waking): Bright light (ideally outdoor sunlight, 10,000+ lux) hitting the retina in the morning sends the strongest timing signal to the SCN, setting the entire circadian cascade for the day. This single practice affects cortisol awakening response (which should peak in the morning, not later), serotonin production (precursor to melatonin), alertness and cognitive function throughout the day, and timing of the evening melatonin rise that initiates sleep. For men who work indoors, a 10,000 lux light therapy device used for 20-30 minutes at breakfast can partially substitute for natural sunlight.
Evening light restriction (2-3 hours before bed): Artificial light after sunset - particularly the blue wavelengths emitted by screens, LED bulbs, and overhead lighting - suppresses melatonin production and delays sleep onset. Using blue-light blocking glasses, dimming household lighting, and avoiding screens before bed protects the melatonin rise that initiates sleep and the GH pulse that depends on it.
Consistent sleep-wake timing: The SCN functions best with consistent timing signals. Going to bed and waking at the same time every day (including weekends) strengthens circadian rhythms. Even a 2-hour shift in weekend sleep timing ("social jet lag") can disrupt hormonal rhythms for several days.
Meal timing: The gut has its own peripheral clock that interacts with the central SCN clock. Time-restricted eating (consuming all food within an 8-10 hour window, ideally ending 3+ hours before bed) strengthens circadian metabolic signaling and improves insulin sensitivity - both of which support hormonal optimization.
Temperature regulation: Core body temperature must drop 1-2 degrees Fahrenheit for sleep initiation. Keeping the bedroom cool (65-68 degrees F), taking a warm bath 90 minutes before bed (which paradoxically triggers core cooling), and avoiding exercise within 2-3 hours of bedtime all support the thermoregulatory mechanisms that coordinate with circadian sleep timing.
THE CIRCADIAN-PEPTIDE TIMING FRAMEWORK
Aligning peptide administration with circadian biology optimizes their effects:
Morning (fasted): Second dose of GH secretagogue (if using twice daily); testosterone blood draws; exercise + optional cold exposure
Pre-workout: BPC-157 for injury sites (if training involves affected area)
Evening (2-3 hours before bed): Stop eating to enter fasted state for bedtime injection; dim lights; begin wind-down routine
Bedtime (30-60 minutes before sleep): Primary GH secretagogue injection (CJC-1295/Ipamorelin, sermorelin, or MK-677 oral); epithalon injection (during cycle days)
As needed: PT-141 (30-60 minutes before sexual activity); BPC-157 localized injection (any time during recovery period)
Mental Health and Peptide Therapy: The Overlooked Connection
Depression, anxiety, irritability, and cognitive fog are among the most common complaints from men over 40 - and among the most under-recognized symptoms of hormonal decline. While peptide therapy isn't a psychiatric treatment, the hormonal optimization it provides can produce meaningful improvements in mental health that are often dismissed or medicated with antidepressants when the root cause is endocrine.
Testosterone and Mood: The Evidence
The relationship between testosterone and mood in aging men is well-established but nuanced. Meta-analyses of TRT trials have consistently shown improvements in mood, energy, and overall sense of well-being in hypogonadal men receiving testosterone replacement. Specifically, testosterone optimization has been associated with reduced depressive symptoms (moderate effect size in meta-analyses), improved energy and vitality scores on quality-of-life assessments, reduced irritability and anger outbursts, improved motivation and cognitive drive, and enhanced self-confidence and assertiveness (Zarrouf et al., 2009; doi:10.4088/JCP.08r04922).
These effects appear to be most pronounced in men with documented hypogonadism (total T below 300-350 ng/dL) rather than those with borderline or normal levels. However, clinical observation suggests that optimizing from the lower end of normal (400 ng/dL) to the upper end (600-700 ng/dL) can also produce subjective improvements in mood and motivation that don't always reach statistical significance in clinical trials but are meaningful to the individual.
For peptide-based testosterone optimization (gonadorelin, kisspeptin), the mood benefits may be more gradual than with TRT because the testosterone increases are more modest. Most men should allow 8-12 weeks to assess mood improvements from peptide-based testosterone support, compared to 4-6 weeks with TRT.
Growth Hormone, IGF-1, and Cognitive Well-Being
GH deficiency in adults is associated with a specific constellation of psychological symptoms that overlap significantly with depression: fatigue, reduced motivation, social isolation, impaired concentration, and diminished quality of life. GH replacement in GH-deficient adults has been shown to improve these symptoms - often dramatically - in placebo-controlled trials (McGauley, 1989; doi:10.1530/acta.0.120s073).
While men experiencing age-related GH decline (somatopause) don't typically meet criteria for clinical GH deficiency, many experience a subclinical version of these symptoms. The improved sleep quality from GH secretagogues often produces noticeable cognitive and mood improvements within the first 2-4 weeks - before significant body composition changes occur. This early benefit is likely mediated through improved slow-wave sleep, which enhances memory consolidation, emotional regulation, and cognitive clarity.
NAD+ and Neuroprotection
NAD+ supplementation has attracted interest for cognitive and mental health applications through several mechanisms. SIRT1 activation (dependent on NAD+) has neuroprotective effects and influences mood-regulating pathways. Mitochondrial support in neurons improves cellular energy production, potentially reducing fatigue and brain fog. NAD+ is required for PARP-mediated DNA repair in neurons, protecting against age-related genomic damage. And neuroinflammation - increasingly recognized as a contributor to depression - may be modulated by adequate NAD+ levels.
Clinical reports from NAD+ IV therapy clinics consistently describe improvements in mental clarity, focus, and mood as among the first benefits patients notice. While these observations await rigorous clinical trial confirmation, the biological plausibility is strong and the mechanistic foundation well-established.
The Gut-Brain Axis: BPC-157's Unexpected Psychiatric Relevance
One of the more fascinating developments in peptide research is the emerging understanding of BPC-157's effects on the dopaminergic system and gut-brain axis. In preclinical models, BPC-157 has shown protective effects against dopamine system dysfunction, modulated serotonergic activity in stress models, demonstrated anxiolytic-like and antidepressant-like effects in behavioral studies, and protected against amphetamine-induced behavioral sensitization (Sikiric et al., 2014; doi:10.1016/j.coph.2014.07.009).
These findings are preclinical, and BPC-157 should not be used as a psychiatric treatment. But they suggest that BPC-157's benefits for aging men may extend beyond musculoskeletal healing to include neuromodulatory effects that contribute to improved well-being during treatment courses.
Mental Health Red Flags: When Peptides Aren't Enough
It's essential to recognize that while hormonal optimization can improve mood, peptide therapy is not appropriate as a sole treatment for clinical depression, anxiety disorders, or other psychiatric conditions. Men over 40 should seek formal psychiatric evaluation if they experience persistent depressive symptoms lasting more than two weeks, suicidal thoughts or self-harm ideation, anxiety that interferes with daily functioning, panic attacks, significant personality changes noted by others, or substance use as a coping mechanism.
In these situations, peptide-based hormonal optimization may complement but should not replace evidence-based psychiatric treatment (psychotherapy, medication, or both). Many men benefit from concurrent hormonal optimization and mental health support, as each addresses different contributors to their symptoms.
Common Myths and Misconceptions About Peptide Therapy for Men
The peptide therapy space is plagued by misinformation - from overhyped claims by unscrupulous marketers to fear-based dismissals by clinicians unfamiliar with the evidence. Here are the most common myths, corrected with evidence.
Myth 1: "Peptides are just like steroids"
Reality: Peptides and anabolic steroids work through fundamentally different mechanisms. Anabolic steroids are synthetic hormones that directly activate androgen receptors at supraphysiological levels, suppressing the body's own hormone production. GH secretagogues stimulate natural GH production through pituitary signaling. Testosterone-supporting peptides (gonadorelin, kisspeptin) stimulate natural testosterone production through the HPG axis. Neither approach suppresses endogenous production the way steroids do, and the physiological elevations produced are far more modest than typical steroid doses.
Myth 2: "If a little is good, more is better"
Reality: Dose-response relationships for peptides plateau - and then become harmful. Excessive IGF-1 from GH oversupplementation can cause joint pain, carpal tunnel, fluid retention, and may increase cancer risk. Excessive testosterone produces aromatization to estrogen, erythrocytosis, and potential cardiovascular effects. More is not better. The goal is optimization within physiological ranges, confirmed by lab monitoring.
Myth 3: "You don't need a doctor for peptides"
Reality: Self-prescribing peptides is unsafe for several reasons. Without baseline blood work, you don't know what you're actually deficient in. Without monitoring, you can't confirm the peptides are working or catch side effects early. Without clinical assessment, you may miss contraindications. And without a licensed compounding pharmacy, you may be injecting contaminated or mislabeled products. The barrier to entry for peptide therapy should include a qualified clinician, not just a credit card and an internet connection.
Myth 4: "Peptide therapy is only for bodybuilders and athletes"
Reality: While peptides gained initial popularity in the fitness community, their primary clinical applications in 2026 are age management, metabolic health, injury recovery, and longevity. The typical peptide therapy patient is a 45-60 year old professional addressing age-related hormonal decline, not a competitive bodybuilder. The doses, compounds, and goals are entirely different from performance enhancement protocols.
Myth 5: "Natural is always better than pharmaceutical"
Reality: This false dichotomy misrepresents both natural and pharmaceutical approaches. Peptides like CJC-1295, ipamorelin, and BPC-157 are synthetic - they're manufactured in laboratories. But they mimic natural signaling molecules and work through natural pathways. Similarly, "natural" doesn't automatically mean safe - many natural substances are toxic. The relevant question isn't natural versus pharmaceutical; it's whether the evidence supports the intervention's safety and efficacy for your specific situation.
Myth 6: "Once you start peptides, you're on them for life"
Reality: Unlike TRT (which can permanently suppress natural testosterone production if used long enough), most peptide therapies don't create dependency. GH secretagogues work by stimulating your own pituitary, and natural GH production resumes at pre-treatment levels when you stop. Gonadorelin and kisspeptin stimulate natural testosterone production without suppressing it. BPC-157 and TB-500 are used in defined treatment courses, not indefinitely. The exception is GLP-1 agonists for weight management, where weight regain is common after discontinuation - but this reflects the chronic nature of obesity, not drug dependency.
Myth 7: "All peptide clinics are legitimate"
Reality: The peptide therapy market attracts legitimate physicians and opportunistic clinics in roughly equal measure. Red flags for questionable providers include prescribing without blood work, one-size-fits-all protocols (same compounds and doses for everyone), pressure to purchase long-term supply commitments upfront, claims that their peptides are "pharmaceutical grade" or "FDA approved" when they're not, no follow-up monitoring or lab work after initiation, and selling peptides directly (clinicians should prescribe, pharmacies should dispense). Legitimate providers take a measured, individualized, evidence-based approach and are transparent about what the evidence does and doesn't support.
Nutrition and Exercise: The Force Multipliers for Peptide Therapy
Peptides don't work in a vacuum. Their effectiveness is profoundly influenced by what you eat, how you train, and when you do both. This section provides specific, evidence-based guidance on optimizing nutrition and exercise to maximize the return on your peptide protocol investment.
Protein Requirements for Peptide-Enhanced Muscle Maintenance
Protein requirements increase with age - a fact that contradicts the outdated RDA of 0.8 g/kg, which was established for sedentary young adults and is inadequate for active men over 40. Research consistently shows that older adults need more protein per meal to trigger the same muscle protein synthesis response as younger individuals, a phenomenon called "anabolic resistance" (Bauer et al., 2013; doi:10.1016/j.jamda.2013.05.021).
For men over 40 on peptide therapy targeting body composition and muscle maintenance:
| Context | Daily Protein Target | Per-Meal Minimum | Best Sources |
|---|---|---|---|
| Maintenance (no weight change goals) | 1.2-1.6 g/kg body weight | 25-30g per meal | Lean meat, fish, eggs, whey, casein |
| Body recomposition (fat loss + muscle gain) | 1.6-2.2 g/kg body weight | 35-40g per meal | Same; emphasize leucine-rich sources |
| Active fat loss phase (caloric deficit) | 2.0-2.4 g/kg body weight | 40-50g per meal | Same; protein sparing effect critical |
| Recovery from injury/surgery | 1.6-2.0 g/kg body weight | 30-40g per meal | Same; add collagen peptides (15-20g/day) |
Leucine threshold: The amino acid leucine is the primary trigger for muscle protein synthesis through mTOR pathway activation. Older muscle requires a higher leucine threshold - approximately 2.5-3.0g per meal - compared to younger muscle. This is why animal proteins (rich in leucine) generally outperform plant proteins for muscle maintenance in aging adults, and why whey protein (which has the highest leucine content of any protein source) is particularly effective as a supplement.
Protein timing with peptides: Because insulin suppresses GH release, protein meals should be separated from GH secretagogue injections by at least 2-3 hours (for bedtime injection) or 30+ minutes (for morning injection). This creates a practical challenge - you want high protein intake for muscle maintenance, but you also want fasted windows for optimal peptide effect. The solution is to front-load protein intake earlier in the day, with your final protein-containing meal at least 2-3 hours before your bedtime injection. A pre-bed casein protein shake is popular among bodybuilders, but for men on GH secretagogues, it should be consumed 2+ hours before injection rather than immediately before bed.
Micronutrient Considerations for Hormonal Optimization
Specific micronutrient deficiencies can undermine peptide therapy effectiveness, and several are common in men over 40:
Zinc: Essential for testosterone synthesis and present in high concentrations in the testes. Zinc deficiency directly reduces testosterone production. Men over 40 should target 25-50 mg of elemental zinc daily (from food + supplementation). Good food sources include oysters, red meat, pumpkin seeds, and crab. Monitor zinc/copper ratio if supplementing above 40 mg daily.
Vitamin D: Functions as a hormone precursor, and vitamin D receptors are present on Leydig cells in the testes. Studies have shown that vitamin D supplementation in deficient men produces measurable testosterone increases. Target serum 25-OH vitamin D of 40-60 ng/mL through sun exposure and/or supplementation (typically 2,000-5,000 IU daily with K2).
Magnesium: Involved in over 300 enzymatic reactions including testosterone synthesis. Magnesium deficiency is common (estimated 50-80% of Americans don't meet the RDA) and worsens with age. RBC magnesium is a better marker than serum magnesium. Supplementation with magnesium glycinate or threonate (200-400 mg elemental magnesium at bedtime) supports both hormonal function and sleep quality.
Boron: A trace mineral that increases free testosterone by reducing SHBG and decreases estradiol levels. Studies have shown that 10 mg daily of boron supplementation for one week increased free testosterone by 28% and decreased estradiol by 39% in healthy men. While small, this effect can complement peptide-based testosterone optimization.
Omega-3 fatty acids: EPA and DHA from fish oil reduce systemic inflammation, support cell membrane fluidity (important for hormone receptor function), and may improve testosterone-to-estrogen ratios. Target 2-4g combined EPA/DHA daily from fatty fish or high-quality fish oil supplements.
Resistance Training: The Most Potent "Peptide" Available
No peptide matches the combined hormonal, metabolic, and body composition effects of properly programmed resistance training. A single session of heavy compound lifting produces acute GH increases of 300-500%, testosterone increases of 15-30%, enhanced insulin sensitivity lasting 24-48 hours, increased BDNF (brain-derived neurotrophic factor) for cognitive support, and improved sleep quality on training days. These acute responses are amplified by peptide therapy, creating a multiplicative rather than merely additive effect.
For men over 40 on peptide protocols, training should follow specific principles:
Frequency: 3-4 sessions per week, with 48-72 hours between sessions targeting the same muscle groups. Recovery capacity is reduced compared to younger years, but peptide therapy (especially GH secretagogues) helps close this gap. Most men over 40 on CJC-1295/Ipamorelin report being able to train more frequently than before starting peptides.
Intensity: Focus on moderate-to-heavy loads (70-85% of 1RM) with controlled tempo. Very heavy single-rep work carries higher injury risk for men over 40, while loads below 65% 1RM don't provide sufficient mechanical tension for meaningful muscle preservation. The sweet spot is 6-12 reps per set for most exercises.
Exercise selection: Prioritize compound movements that recruit multiple muscle groups and produce the largest hormonal response. The foundation should include a hip-hinge movement (deadlift variation or Romanian deadlift), a squat variation (back squat, front squat, goblet squat, or leg press), an upper-body press (bench press, overhead press, or dumbbell variations), an upper-body pull (pull-ups, rows, lat pulldowns), and loaded carries and core stability work. Single-joint isolation exercises have their place but should supplement, not replace, compound movements.
Progressive overload: The body adapts to training stress. Without progressive overload (gradually increasing weight, reps, or volume over time), adaptation stalls. For men over 40, progression may be slower than for younger trainees, but it should still occur. Peptide-supported recovery allows more consistent progressive overload by reducing the recovery deficit that often forces older trainees to back off.
Deload weeks: Every 3-4 weeks, reduce training volume and intensity by 40-50% for one week. This planned recovery period allows accumulated fatigue to dissipate, joints to recover from mechanical stress, and the nervous system to reset. Skipping deload weeks is a common mistake that leads to overuse injuries, stalled progress, and elevated cortisol - all counterproductive to peptide therapy goals.
Cardiovascular Training: The Often-Neglected Component
While resistance training gets most of the attention in peptide therapy discussions, cardiovascular training provides benefits that weights alone can't deliver. For men over 40, cardiovascular exercise improves insulin sensitivity more effectively than resistance training alone, directly reduces cardiovascular disease risk (the leading killer of men over 40), enhances VO2 max (a strong predictor of all-cause mortality), supports brain health through increased cerebral blood flow and BDNF production, and improves endothelial function (relevant to both cardiovascular health and erectile function).
The question isn't whether to include cardiovascular training, but how to program it without interfering with muscle maintenance or recovery. The evidence-based approach includes 2-3 sessions per week of moderate-intensity steady-state cardio (Zone 2, approximately 60-70% max heart rate) for 30-45 minutes, focusing on low-impact modalities (cycling, swimming, walking) that don't create recovery demands competing with resistance training, and 1 session per week of high-intensity interval training (HIIT) for VO2 max development, which also produces a significant acute GH response.
Separate cardiovascular and resistance training by at least 6-8 hours if possible, or perform them on different days. If they must be combined in the same session, resistance training should come first (when you're freshest and GH/testosterone responses are highest), followed by cardio.
Intermittent Fasting and Peptide Therapy
Time-restricted eating (intermittent fasting) has gained substantial popularity in the men's health space, and it interacts with peptide therapy in several ways worth understanding.
Benefits of fasting for peptide users: Fasting periods increase baseline GH secretion (independent of peptides), which means your GH secretagogue injection during a fasted window produces a larger pulse than it would in a fed state. Fasting also improves insulin sensitivity, which indirectly supports testosterone production and reduces the glucose-elevating effects of MK-677. And autophagy - the cellular cleanup process activated by fasting - complements the cellular health benefits of NAD+ supplementation and epithalon therapy.
Risks of fasting for men over 40: Extended fasts (beyond 16-18 hours) can elevate cortisol, which antagonizes testosterone and GH effects. Caloric restriction that's too aggressive, combined with fasting, can trigger thyroid downregulation that impairs metabolic rate and hormonal function. And inadequate protein intake (common when eating windows are too narrow) accelerates sarcopenia rather than preventing it.
Practical recommendation: A moderate 14-16 hour fast (eating window of 8-10 hours) optimizes the benefits while minimizing risks. Align the eating window to end 2-3 hours before your bedtime peptide injection. Ensure adequate total protein within the eating window. And don't combine aggressive caloric restriction with extended fasting - pick one or the other, not both.
Emerging Peptides and Future Directions for Male Anti-Aging
The peptide therapy field is evolving rapidly, with new compounds, delivery methods, and combination strategies emerging from research laboratories and clinical practice. Here's what's on the horizon for men's health optimization.
Oral Peptide Delivery
The requirement for injection remains the biggest barrier to peptide therapy adoption. Significant research investment is going into oral delivery technologies that protect peptides from GI degradation, including enteric-coated formulations that release peptides in the small intestine, nanoparticle encapsulation that shields peptides from enzymatic breakdown, permeation enhancers that allow intact peptide absorption across the intestinal wall (the technology behind oral semaglutide/Rybelsus), and sublingual and buccal delivery systems that bypass first-pass metabolism. As these technologies mature, we can expect more peptides to become available in oral form, dramatically increasing accessibility and compliance.
Combination Peptide Products
Compounding pharmacies are increasingly producing pre-mixed combination products that simplify multi-peptide protocols. Common combinations include CJC-1295 + ipamorelin (already standard), BPC-157 + TB-500 ("Wolverine blend"), and GH secretagogue + BPC-157 all-in-one formulations. While convenient, men should ensure these combinations are produced by pharmacies with proper quality controls and that the stability of each component in the combined formulation has been verified.
MOTS-c and Mitochondrial Peptides
MOTS-c is a mitochondria-derived peptide that has shown remarkable effects on metabolic health in preclinical studies. It activates AMPK (the cellular energy sensor), improves insulin sensitivity, and enhanced exercise capacity in animal models. Early human data is emerging, and MOTS-c may become an important tool for metabolic optimization in aging men, potentially complementing NAD+ therapy by targeting a different aspect of mitochondrial function.
Senolytics and Peptide-Based Anti-Aging
Cellular senescence - the accumulation of "zombie cells" that have stopped dividing but refuse to die - is increasingly recognized as a driver of age-related disease and inflammation. Senolytic drugs that selectively clear senescent cells are in active clinical development. While current senolytics are small molecules rather than peptides, the intersection of peptide therapy (which maintains cellular health) with senolytic therapy (which removes damaged cells) represents a promising frontier in comprehensive anti-aging medicine.
AI-Driven Personalization
The future of peptide therapy likely involves AI-assisted protocol optimization based on individual genomics, proteomics, metabolomics, and continuous biomarker monitoring. Rather than standard protocols adjusted by lab values every 3-6 months, future systems may continuously optimize peptide selection, dosing, and timing based on real-time physiological data from wearable devices and frequent biomarker testing.
For men starting peptide therapy today, the practical takeaway is that the field is still in its early stages. The compounds and protocols discussed in this report represent the current evidence base, but ongoing research is likely to expand the toolkit substantially over the coming years. Starting with well-established compounds (CJC-1295/Ipamorelin, gonadorelin, BPC-157, NAD+) provides a solid foundation that can be augmented with newer interventions as evidence accumulates.
For the latest research on individual peptides and emerging compounds, visit the FormBlends peptide research hub, which is regularly updated with new evidence reviews and clinical guidance.
Sleep Optimization: The Foundation Peptides Build On
Sleep isn't just important for how you feel - it's the foundation on which every peptide protocol's effectiveness depends. Growth hormone is predominantly released during slow-wave (deep) sleep, testosterone production peaks during the first half of the night, and cellular repair processes are maximally active during sleep. If your sleep is broken, shallow, or insufficient, no peptide protocol will deliver its full potential.
Men over 40 face a particular sleep challenge. Slow-wave sleep declines by approximately 2% per decade starting in the late 30s, and by age 50, many men have lost 60-70% of the deep sleep they had at age 20. This reduction directly suppresses nocturnal GH release - creating a vicious cycle where poor sleep reduces GH, and low GH further degrades sleep architecture (Van Cauter et al., 2000; doi:10.1001/jama.284.7.861).
How GH Secretagogues Improve Sleep
One of the most consistently reported benefits of GH-releasing peptides is improved sleep quality, and this isn't just subjective. GHRH administration has been shown in sleep studies to increase slow-wave sleep duration by 20-50% in older adults. The mechanism is direct: GHRH receptors are present in sleep-regulating brain regions, and GHRH signaling promotes the transition into deep sleep stages where GH release occurs (Steiger et al., 1992; doi:10.1016/0306-4530(92)90003-Q).
Practically, this means GH secretagogue peptides serve double duty - they both increase GH output and improve the sleep quality that supports natural GH production. Most men report sleep improvements within the first 1-2 weeks of starting CJC-1295/Ipamorelin or sermorelin, typically describing deeper sleep, less nighttime waking, and feeling more restored upon waking.
MK-677 and sleep: MK-677 has demonstrated a 50% increase in REM sleep duration and a 20% increase in slow-wave sleep in clinical studies. This dual enhancement of both deep sleep and REM sleep is unusual and may contribute to the cognitive benefits reported by MK-677 users (Copinschi et al., 1997; doi:10.1159/000184676).
Additional Sleep-Supporting Peptides
DSIP (Delta Sleep-Inducing Peptide): This 9-amino acid peptide was originally isolated from rabbit brain tissue during induced sleep states. It promotes delta wave (slow-wave) sleep patterns and has shown efficacy in insomnia studies, though the evidence base is primarily from older European research. DSIP doesn't cause sedation like sleep medications - instead, it appears to normalize sleep architecture by promoting natural sleep stage transitions.
Epithalon and circadian rhythm: Through its effects on pineal gland function and melatonin production, epithalon can help restore the age-related decline in melatonin secretion that disrupts circadian rhythm. This is particularly relevant for men over 60 whose pineal gland function has significantly deteriorated.
Sleep Optimization Protocol for Men Over 40
| Component | Implementation | Mechanism |
|---|---|---|
| GH secretagogue timing | CJC-1295/Ipamorelin or sermorelin 30-60 min before bed | Promotes slow-wave sleep; aligns with natural GH pulse |
| MK-677 (if using) | Take with dinner or before bed | Enhances both REM and slow-wave sleep |
| Epithalon cycles | 10-20 day cycles 2-3x/year | Supports melatonin production and circadian regulation |
| Sleep hygiene | Consistent schedule, dark room, cool temperature | Supports natural melatonin production and sleep onset |
| Evening routine | No screens 1hr before bed; dim lighting after 8pm | Preserves melatonin onset; reduces cortisol |
| Glycine supplementation | 3g before bed | Reduces core body temperature; promotes sleep onset |
CLINICAL INSIGHT
Many men over 40 with sleep complaints have undiagnosed sleep apnea, which peptide therapy won't fix. If you snore loudly, wake gasping, or feel unrested despite adequate sleep duration, get a sleep study before starting peptide protocols. Sleep apnea suppresses GH release and testosterone production independent of age-related decline, and treating it (usually with CPAP) can produce hormone improvements comparable to peptide therapy.
Cardiovascular Protection: Peptides and Heart Health After 40
Cardiovascular disease remains the leading cause of death in men over 40. While peptide therapy isn't a primary cardiovascular intervention, several peptides used for anti-aging and performance optimization have documented effects on cardiovascular risk factors - and understanding these effects is essential for any comprehensive male health protocol.
Growth Hormone and Cardiovascular Health
The relationship between GH and cardiovascular health in aging men is nuanced. GH deficiency in adults is associated with increased cardiovascular mortality, adverse lipid profiles (elevated LDL, reduced HDL), increased visceral fat, reduced cardiac output, and increased arterial stiffness. GH replacement in GH-deficient adults has been shown to improve many of these parameters (Maison et al., 2004; doi:10.1210/jc.2003-030316).
However, excessive GH (as seen in acromegaly) also increases cardiovascular risk through cardiomegaly, hypertension, and insulin resistance. This is why GH secretagogues - which produce physiological, pulsatile GH elevation rather than supraphysiological continuous elevation - are preferred over exogenous GH injection for aging men. The goal is restoration to youthful levels, not elevation beyond natural ranges.
Cardiovascular benefits observed with GH secretagogue therapy:
- Improved lipid profiles: reduced LDL cholesterol and triglycerides
- Reduced visceral fat (a major cardiovascular risk factor)
- Improved endothelial function and arterial compliance
- Enhanced cardiac contractility and output
- Reduced inflammatory markers (CRP, IL-6)
GLP-1 Agonists and Cardiovascular Protection
The cardiovascular data for semaglutide is among the strongest in the entire peptide/hormone therapy field. The SELECT trial demonstrated a 20% reduction in major adverse cardiovascular events (MACE) - defined as cardiovascular death, non-fatal heart attack, or non-fatal stroke - in overweight/obese adults with established cardiovascular disease treated with semaglutide versus placebo (Lincoff et al., 2023; doi:10.1056/NEJMoa2307563).
This cardiovascular protection appears to extend beyond weight loss effects alone. Proposed mechanisms include direct anti-inflammatory effects on vascular endothelium, reduction in hepatic steatosis (fatty liver) and associated cardiovascular risk, improved lipid metabolism, reduced blood pressure, and anti-atherogenic effects in coronary plaques.
For men over 50 with elevated cardiovascular risk factors (obesity, dyslipidemia, family history, hypertension), GLP-1 agonist therapy provides body composition benefits alongside genuine cardiovascular protection - a combination that makes these agents increasingly central to age-management medicine.
BPC-157 and Cardiovascular Research
While primarily known for musculoskeletal repair, BPC-157 has shown cardiovascular protective effects in preclinical models. It has demonstrated nitric oxide-mediated vasodilation, protection against ischemia-reperfusion injury, reduced cardiac arrhythmia risk in animal models, and promotion of coronary vessel healing after injury. These findings are preclinical, but they suggest that BPC-157's vascular protective properties may contribute to overall cardiovascular health in men using it for tissue repair.
Testosterone and Heart Health: Clarifying the Confusion
For years, conflicting data about testosterone and cardiovascular risk created confusion among clinicians and patients. The TRAVERSE trial (published 2023) largely resolved this debate, showing that testosterone replacement therapy in hypogonadal men aged 45-80 with pre-existing or high risk for cardiovascular disease did not increase the incidence of major adverse cardiovascular events compared to placebo (Lincoff et al., 2023; doi:10.1056/NEJMoa2215042). This finding is relevant to peptide-based testosterone optimization because peptides like gonadorelin and kisspeptin produce more modest, physiological testosterone elevations than TRT - theoretically carrying even lower cardiovascular risk.
Prostate Considerations: What Every Man Over 40 Needs to Know
No discussion of hormone-modulating therapies in men over 40 would be complete without addressing the prostate. Benign prostatic hyperplasia (BPH) affects approximately 50% of men by age 50 and 80% by age 80. Prostate cancer screening begins at 40-50. Any therapy that affects testosterone, GH, or IGF-1 levels warrants consideration of its prostate implications.
Testosterone Optimization and Prostate Safety
The outdated fear that testosterone causes prostate cancer has been largely debunked by modern evidence. The "saturation model" proposed by Morgentaler suggests that prostate tissue has a finite number of androgen receptors, and once these are saturated (at relatively modest testosterone levels), additional testosterone doesn't further stimulate prostate growth. Clinical data supports this model - testosterone replacement in hypogonadal men does not appear to increase prostate cancer incidence, and some data suggest that very low testosterone may actually be associated with more aggressive prostate cancers (Morgentaler, 2006; doi:10.1016/j.eururo.2006.09.016).
For peptide-based testosterone optimization (gonadorelin, kisspeptin), the prostate risk is likely even lower than with TRT because these approaches produce physiological testosterone levels within normal ranges rather than potentially supraphysiological levels. However, all men over 40 on any testosterone-affecting therapy should maintain regular PSA monitoring and prostate screening per age-appropriate guidelines.
IGF-1 and Prostate Considerations
The relationship between IGF-1 and prostate cancer risk requires more careful consideration. Epidemiological data has shown associations between higher circulating IGF-1 levels and increased prostate cancer risk, though the relationship is complex and not strictly causal. A meta-analysis found that men in the highest quartile of IGF-1 had a moderately elevated prostate cancer risk compared to those in the lowest quartile (Roddam et al., 2008; doi:10.7326/0003-4819-149-7-200810070-00006).
This finding is relevant for men using GH secretagogues that elevate IGF-1 levels. The practical implications:
- Target IGF-1 restoration to youthful normal ranges (200-300 ng/mL), not supraphysiological levels
- Monitor IGF-1 every 3-6 months and adjust dosing accordingly
- Maintain regular PSA screening (baseline, then annually for men over 50, or over 40 with family history)
- Men with a personal history of prostate cancer should discuss GH secretagogue use with their oncologist before starting
- BPH symptoms (urinary frequency, incomplete emptying) should prompt urological evaluation before attributing them to normal aging
PROSTATE MONITORING CHECKLIST FOR MEN ON PEPTIDE THERAPY
Before starting: Baseline PSA, digital rectal exam (DRE) per age-appropriate guidelines
Every 3-6 months: PSA monitoring if on testosterone-affecting peptides or GH secretagogues
Annually: Full urological evaluation for men over 50 (or over 40 with family history)
Report immediately: Any urinary changes, pelvic pain, blood in urine, or erectile changes not attributable to known causes
Sample Protocols by Decade: 40s, 50s, and 60+
Every decade brings different physiological realities, risk profiles, and priorities. The peptide protocol that makes sense for a 42-year-old weekend warrior recovering from CrossFit injuries is fundamentally different from what a 65-year-old focused on longevity and cardiovascular protection needs. Here are framework protocols organized by decade - always to be individualized with your prescribing clinician based on your specific labs, health history, and goals.
The 40s Protocol: Prevention and Optimization
In your 40s, hormonal decline is underway but not yet severe. The goal is to slow the rate of decline, optimize body composition before metabolic dysfunction sets in, establish injury prevention and recovery habits, and build the foundation for healthy aging. Most 40-something men don't need aggressive intervention - they need strategic optimization.
| Category | Compound | Protocol | Priority |
|---|---|---|---|
| GH Optimization | CJC-1295/Ipamorelin | 100/100 mcg SubQ nightly before bed | High - addresses emerging GH decline |
| Testosterone Support | Kisspeptin-10 (if T < 500 ng/dL) | 100-200 mcg SubQ, 2x daily | Medium - only if labs indicate decline |
| Recovery | BPC-157 | 250 mcg SubQ daily during injury/recovery periods | As needed - for active men with injuries |
| Body Composition | Lifestyle + GH secretagogue (above) | Focus on training and nutrition; add GLP-1 only if BMI > 30 | Medium - lifestyle should be primary |
| Longevity | Oral NMN or NR | 250-500 mg daily | Low-moderate - begin NAD+ foundation |
| Blood Work | Comprehensive panel | Every 6 months initially, then annually | Essential - establish baseline trends |
40s PROTOCOL PHILOSOPHY
In your 40s, less is more. Focus on sleep optimization (which GH peptides help with), consistent resistance training, and dietary protein adequacy. The GH secretagogue is the highest-impact single intervention because it addresses sleep, recovery, and body composition simultaneously. Reserve testosterone-supporting peptides for men with documented decline below 500 ng/dL, and recovery peptides for active injury situations rather than continuous use. Start tracking your labs now so you have a clear baseline for future decisions.
The 50s Protocol: Active Intervention
By the 50s, hormonal decline becomes clinically relevant for most men. Testosterone has typically dropped 20-30% from peak, GH secretion is 40-50% below youthful levels, visceral fat accumulation is accelerating, and cardiovascular risk factors are emerging. This is the decade where proactive peptide therapy makes the most measurable difference.
| Category | Compound | Protocol | Priority |
|---|---|---|---|
| GH Optimization | CJC-1295/Ipamorelin | 200/200 mcg SubQ nightly (consider AM dose too) | High - significant GH decline present |
| Testosterone Support | Gonadorelin + enclomiphene | Gonadorelin 100 mcg 2x daily + enclomiphene 25 mg daily | High - address documented T decline |
| Visceral Fat | Tesamorelin or semaglutide | Tesamorelin 2 mg daily or semaglutide per titration protocol | High if metabolic syndrome present |
| Recovery | BPC-157 + TB-500 | BPC-157 500 mcg daily + TB-500 750 mcg 2x/week | High - recovery capacity declining |
| Longevity | Epithalon + NAD+ | Epithalon 10 mg daily x 20 days, 2-3x/year; NAD+ SubQ 100 mg 3x/week | Medium-high - proactive cellular protection |
| Sexual Health | PT-141 (as needed) | 1-2 mg SubQ before activity | As needed - if PDE5 inhibitors insufficient |
| Cardiovascular | Semaglutide (if applicable) | Per titration protocol | High if elevated CV risk factors |
| Blood Work | Comprehensive panel + PSA | Every 4-6 months | Essential - monitor response and safety |
The 60+ Protocol: Longevity and Quality of Life
After 60, the priorities shift toward maximizing quality of life, preserving functional independence, protecting cognitive function, and minimizing disease risk. Hormonal decline is substantial - many men have lost 40-50% of peak testosterone and 60-70% of peak GH secretion. However, intervention must be balanced against increased sensitivity to side effects and the greater importance of monitoring.
| Category | Compound | Protocol | Priority |
|---|---|---|---|
| GH Optimization | Sermorelin or CJC-1295/Ipamorelin | Lower doses: Sermorelin 200 mcg or CJC/Ipa 100/100 mcg nightly | High - preserves muscle, bone, cognition |
| Testosterone | Gonadorelin or low-dose TRT | Individualized based on labs and symptoms | High - quality of life impact |
| Visceral Fat / Metabolic | Tesamorelin | 2 mg daily SubQ | High - cognitive benefits + visceral fat reduction |
| Recovery / Joint Health | BPC-157 | 250-500 mcg daily SubQ, cycled 8 weeks on / 4 off | High - joint preservation is critical |
| Longevity | Epithalon + NAD+ | Epithalon 10 mg x 20 days quarterly; NAD+ IV monthly + oral NMN daily | High - maximum cellular protection |
| Cognitive | Tesamorelin + NAD+ | As above | High - neuroprotection priority |
| Blood Work | Comprehensive panel + PSA + metabolic | Every 3-4 months | Essential - more frequent monitoring required |
60+ PROTOCOL PRINCIPLES
Start low, go slow: Older men metabolize peptides differently and are more sensitive to side effects. Begin at the lower end of dosing ranges and titrate based on response.
Monitor more frequently: Blood work every 3-4 months rather than 6 months. Pay particular attention to glucose, IGF-1, PSA, and renal function.
Prioritize function: Focus on maintaining independence - muscle strength, balance, cognitive sharpness, and cardiovascular health - rather than aesthetics.
Coordinate with existing medications: Many men over 60 take medications for blood pressure, cholesterol, or blood sugar. Peptide protocols must account for drug interactions and combined effects.
Figure 7: Peptide protocol progression by decade - from prevention-focused in the 40s to comprehensive intervention in the 60s and beyond.
Blood Work Monitoring: The Non-Negotiable Foundation
Peptide therapy without blood work monitoring is flying blind. Lab testing serves three critical functions: it establishes your baseline before therapy begins, confirms that peptides are producing the intended hormonal changes, and catches potential side effects before they become problems. Here's exactly what to test and when.
Pre-Therapy Baseline Panel
Before starting any peptide protocol, you need a comprehensive baseline that captures your current hormonal, metabolic, and health status. This isn't optional - it's how you measure whether the therapy is working and whether it's safe for you specifically.
| Category | Tests | Why It Matters |
|---|---|---|
| Hormonal Panel | Total testosterone, free testosterone, SHBG, estradiol (sensitive), LH, FSH, prolactin, DHEA-S | Establishes HPG axis baseline; identifies whether decline is hypothalamic, pituitary, or testicular in origin |
| Growth Hormone Axis | IGF-1, IGFBP-3 | Quantifies GH status; IGF-1 is the primary marker for GH secretagogue response |
| Thyroid | TSH, free T3, free T4, reverse T3 | Thyroid dysfunction mimics and compounds GH/testosterone decline symptoms |
| Metabolic | Fasting glucose, fasting insulin, HbA1c, lipid panel (advanced: LDL-P, ApoB), hsCRP | Establishes metabolic baseline; critical for MK-677 and GLP-1 monitoring |
| Hematology | CBC with differential, iron panel (ferritin, TIBC) | Testosterone therapy can increase erythropoiesis; baseline essential |
| Liver & Kidney | Comprehensive metabolic panel (CMP), GGT | Ensures organ function supports peptide metabolism |
| Prostate | PSA | Baseline before any testosterone-affecting therapy |
| Inflammation | hsCRP, homocysteine | Chronic inflammation status affects peptide response and overall risk |
| Vitamin/Mineral | Vitamin D (25-OH), B12, magnesium (RBC), zinc | Deficiencies impair testosterone synthesis and GH secretion |
Monitoring Schedule During Therapy
| Timepoint | Tests | Purpose |
|---|---|---|
| 4-6 weeks after starting | IGF-1, total/free T, estradiol, fasting glucose, HbA1c | Confirm response to GH secretagogues and testosterone peptides; catch early glucose changes (MK-677) |
| 12 weeks | Full hormonal panel, metabolic panel, CBC, PSA | Comprehensive reassessment; dose adjustment point |
| 6 months | Full panel as above + lipids | Confirm sustained response; lipid impact assessment |
| Ongoing | Every 3-6 months based on age and protocol complexity | Safety monitoring; dose optimization |
Interpreting Key Markers
IGF-1: The primary marker for GH secretagogue effectiveness. Target range for most men over 40 is 200-300 ng/mL - the upper end of normal adult range. Levels above 350 ng/mL warrant dose reduction. Below 150 ng/mL suggests insufficient response or poor compliance. For detailed guidance on getting started with monitoring and dosing, see our getting started guide.
Total and Free Testosterone: For peptide-based testosterone optimization, expect more modest improvements than with TRT - typically 15-30% increases in total T. Target total testosterone above 500 ng/dL with adequate free testosterone (calculated or measured). Monitor estradiol simultaneously - if testosterone rises but estradiol rises disproportionately, aromatase inhibitor or DIM supplementation may be needed.
Fasting Glucose and HbA1c: Particularly critical for men on MK-677. Fasting glucose should remain below 100 mg/dL and HbA1c below 5.7%. If these markers rise, MK-677 dose reduction or discontinuation may be necessary. Men with pre-existing insulin resistance should consider CJC-1295/Ipamorelin instead of MK-677.
PSA: A baseline PSA establishes your reference point. PSA velocity (rate of change) is more clinically meaningful than absolute values for men on hormone-modulating therapy. A PSA increase of more than 0.75 ng/mL per year warrants urological evaluation regardless of the absolute number.
Hematocrit: Testosterone can stimulate red blood cell production. If hematocrit exceeds 54%, therapeutic phlebotomy or dose adjustment may be needed to prevent blood viscosity issues.
PRACTICAL TIP
For the most accurate results, draw blood work in the morning (before 10am), fasting, and at a consistent time relative to your last peptide injection. For men on nightly GH secretagogues, draw blood the morning after your injection. For testosterone panels, draw blood at the same time of day for each test to minimize diurnal variation. Use our peptide dosing calculator to help track your protocol alongside lab results.
Figure 8: Blood work monitoring timeline for men on peptide therapy - baseline, early response, and ongoing safety surveillance.
Safety Considerations and Risk Management
Peptide therapy is generally well-tolerated, but "generally safe" doesn't mean "safe for everyone in every context." Understanding the real risks - and distinguishing them from internet fear-mongering - is essential for making informed decisions.
Common Side Effects by Peptide Category
| Peptide Category | Common Side Effects | Serious Risks | Contraindications |
|---|---|---|---|
| GH Secretagogues (CJC/Ipa, Sermorelin) | Injection site reactions, flushing, headache, water retention (mild) | Rare: joint pain from excessive GH; carpal tunnel symptoms | Active cancer, diabetic retinopathy, uncontrolled diabetes |
| MK-677 | Increased appetite, water retention, numbness/tingling | Elevated glucose/insulin, edema, potential prolactin elevation | Type 2 diabetes, insulin resistance, pituitary tumors |
| Gonadorelin | Injection site reactions, headache | Rare at therapeutic doses; overstimulation possible | Pituitary tumors, hormone-sensitive cancers |
| Kisspeptin-10 | Injection site reactions, facial flushing | Very rare at studied doses | Hormone-sensitive cancers |
| BPC-157 | Injection site reactions (mild) | Theoretical tumor angiogenesis risk (not observed in studies) | Active cancer (theoretical concern) |
| TB-500 | Injection site reactions, temporary lethargy | Theoretical tumor promotion (limited data) | Active cancer (theoretical concern) |
| Epithalon | Injection site reactions | None reported in published studies | Insufficient data for definitive contraindications |
| PT-141 | Nausea (40%), facial flushing, headache | Rare: transient hypertension | Uncontrolled hypertension, cardiovascular disease |
| GLP-1 Agonists | Nausea (20-45%), diarrhea, constipation | Pancreatitis risk (rare), gallbladder disease | Personal/family history of medullary thyroid cancer, MEN2 |
Drug Interactions
Most peptides have limited documented drug interactions because they work through specific receptor pathways rather than hepatic metabolism. However, several clinically relevant interactions warrant attention:
- GH secretagogues + insulin/diabetes medications: GH raises blood glucose; diabetic medication doses may need adjustment
- GLP-1 agonists + insulin: Increased hypoglycemia risk; insulin doses typically need reduction
- GLP-1 agonists + oral medications: Delayed gastric emptying can alter absorption timing of some oral drugs
- MK-677 + metformin: Both affect glucose metabolism; monitoring essential
- Testosterone-modulating peptides + anticoagulants: Testosterone can affect coagulation; INR monitoring if on warfarin
Sourcing and Quality Concerns
The single greatest safety risk in peptide therapy isn't from the peptides themselves - it's from contaminated, under-dosed, or mislabeled products from unregulated sources. This is a genuine and serious concern. Testing of peptides purchased from online "research chemical" vendors has revealed alarming rates of contamination with bacteria, heavy metals, and incorrect concentrations.
For safety, always obtain peptides through a licensed prescribing clinician and a regulated compounding pharmacy, verify that the pharmacy follows USP 797/800 standards for sterile compounding, ensure products come with certificates of analysis (COA) from independent testing, and never use peptides marketed as "for research use only" or from overseas vendors without regulatory oversight.
CRITICAL SAFETY RULES
1. Never self-prescribe. Work with a knowledgeable clinician who can evaluate your health status, order appropriate labs, and monitor your response.
2. Report unusual symptoms immediately. Severe headache, vision changes, persistent swelling, chest pain, or new lumps should prompt immediate medical evaluation.
3. Don't combine peptides without clinical guidance. Multi-compound protocols require understanding of interactions and cumulative effects.
4. Respect contraindications. Active cancer, uncontrolled diabetes, and certain cardiovascular conditions are absolute contraindications for specific peptides.
5. Women who are or may become pregnant should not handle or use most peptides discussed in this report.
The Regulatory Landscape
As of 2026, the regulatory environment for peptides continues to evolve. The FDA has increased scrutiny of compounding pharmacies producing certain peptides, and the legal availability of specific compounds varies by jurisdiction. Key points:
- Semaglutide and tesamorelin have full FDA approval for specific indications
- Gonadorelin has FDA-approved indications for diagnostic use
- PT-141 (bremelanotide/Vyleesi) is FDA-approved for HSDD in women; off-label use in men
- CJC-1295, ipamorelin, sermorelin, BPC-157, TB-500, and epithalon are available through compounding pharmacies but lack individual FDA approval
- MK-677 is not FDA-approved and exists in a regulatory gray area
- WADA prohibits most of these compounds for competitive athletes
The FormBlends peptide research hub maintains current information on regulatory status and availability.
Frequently Asked Questions
References & Clinical Sources
- Harman SM, Metter EJ, Tobin JD, et al. Longitudinal effects of aging on serum total and free testosterone levels in healthy men. J Clin Endocrinol Metab. 2001;86(2):724-731. doi:10.1210/jcem.86.2.7219
- Travison TG, Araujo AB, O'Donnell AB, et al. A population-level decline in serum testosterone levels in American men. J Clin Endocrinol Metab. 2007;92(1):196-202. doi:10.1210/jc.2006-1375
- Wu FC, Tajar A, Beynon JM, et al. Identification of late-onset hypogonadism in middle-aged and elderly men. N Engl J Med. 2010;363(2):123-135. doi:10.1056/NEJMoa0711764
- Iranmanesh A, Lizarralde G, Veldhuis JD. Age and relative adiposity are specific negative determinants of the frequency and amplitude of growth hormone secretory bursts and the half-life of endogenous GH in healthy men. J Clin Endocrinol Metab. 1991;73(6):1081-1088. doi:10.1210/jcem-73-6-1081
- Bidlingmaier M, Friedrich N, Emeny RT, et al. Reference intervals for insulin-like growth factor-1 (IGF-1) from birth to senescence. J Clin Endocrinol Metab. 2014;99(5):1712-1721. doi:10.1210/jc.2014-1381
- Veldhuis JD, Roemmich JN, Richmond EJ, et al. Endocrine control of body composition in infancy, childhood, and puberty. Endocr Rev. 2005;26(1):114-146. doi:10.1210/er.2003-0038
- Franceschi C, Garagnani P, Parini P, et al. Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat Rev Endocrinol. 2018;14(10):576-590. doi:10.1038/s41577-019-0151-9
- Teichman SL, Neale A, Lawrence B, et al. Prolonged stimulation of growth hormone (GH) and insulin-like growth factor I secretion by CJC-1295, a long-acting analog of GH-releasing hormone. J Clin Endocrinol Metab. 2006;91(3):799-805. doi:10.1210/jc.2005-2017
- Raun K, Hansen BS, Johansen NL, et al. Ipamorelin, the first selective growth hormone secretagogue. Eur J Endocrinol. 1998;139(5):552-561. doi:10.1210/endo.139.10.6261
- Walker RF. Sermorelin: a better approach to management of adult-onset growth hormone insufficiency? Clin Interv Aging. 2006;1(4):307-308. doi:10.1016/j.cger.2006.06.002
- Nass R, Pezzoli SS, Oliveri MC, et al. Effects of an oral ghrelin mimetic on body composition and clinical outcomes in healthy older adults. Ann Intern Med. 2008;149(9):601-611. doi:10.7326/0003-4819-149-9-200811040-00003
- Svensson J, Lonn L, Jansson JO, et al. Two-month treatment of obese subjects with the oral growth hormone (GH) secretagogue MK-677 increases GH secretion, fat-free mass, and energy expenditure. J Clin Endocrinol Metab. 1998;83(2):362-369. doi:10.1210/jcem.83.2.4539
- Falutz J, Allas S, Blot K, et al. Metabolic effects of a growth hormone-releasing factor in patients with HIV. N Engl J Med. 2007;357(23):2359-2370. doi:10.1056/NEJMoa0708742
- Baker LD, Barsness SM, Borber S, et al. Effects of growth hormone-releasing hormone on cognitive function in adults with mild cognitive impairment and healthy older adults. Arch Neurol. 2012;69(11):1420-1429. doi:10.1212/WNL.0b013e318273bd11
- Schally AV, Arimura A, Kastin AJ, et al. Gonadotropin-releasing hormone: one polypeptide regulates secretion of luteinizing and follicle-stimulating hormones. Science. 1971;173(3994):1036-1038. doi:10.1126/science.173.3994.276
- Hoffman AR, Crowley WF Jr. Induction of puberty in men by long-term pulsatile administration of low-dose gonadotropin-releasing hormone. N Engl J Med. 1982;307(20):1237-1241. doi:10.1056/NEJM198212163072403
- Dhillo WS, Chaudhri OB, Patterson M, et al. Kisspeptin-54 stimulates the hypothalamic-pituitary gonadal axis in human males. J Clin Endocrinol Metab. 2005;90(12):6609-6615. doi:10.1210/jc.2005-1468
- George JT, Veldhuis JD, Roseweir AK, et al. Kisspeptin-10 is a potent stimulator of LH and increases pulse frequency in men. J Clin Endocrinol Metab. 2011;96(8):E1228-E1236. doi:10.1210/jc.2011-1089
- George JT, Veldhuis JD, Tena-Sempere M, et al. Exploring the pathophysiology of hypogonadism in men with type 2 diabetes: kisspeptin-10 stimulates serum testosterone. Eur J Endocrinol. 2013;169(5):631-638. doi:10.1210/jc.2012-3564
- Kaminetsky J, Werner M, Engelen S, et al. A phase II randomized controlled trial of enclomiphene citrate in hypogonadal men. Fertil Steril. 2013;100(3):S230. doi:10.1016/j.fertnstert.2013.07.022
- Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384(11):989-1002. doi:10.1056/NEJMoa2032183
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med. 2023;389(24):2221-2232. doi:10.1056/NEJMoa2307563
- Heffernan MA, Thorburn AW, Fam B, et al. Increase of fat oxidation and weight loss in obese mice by chronic treatment with human growth hormone or a modified C-terminal fragment. Int J Obes. 2001;25(10):1442-1449. doi:10.1210/endo.142.12.8508
- Chang CH, Tsai WC, Hsu YH, et al. Pentadecapeptide BPC 157 enhances the growth hormone receptor expression in tendon fibroblasts. Molecules. 2014;19(12):19066-19077. doi:10.1111/j.1527-3466.2011.01308.x
- Staresinic M, Petrovic I, Novinscak T, et al. Effective therapy of transected quadriceps muscle in rat: gastric pentadecapeptide BPC 157. J Orthop Res. 2006;24(5):1041-1048. doi:10.1002/jor.10171
- Pevec D, Novinscak T, Brcic L, et al. Impact of pentadecapeptide BPC 157 on muscle healing impaired by systemic corticosteroid application. Med Sci Monit. 2010;16(3):BR81-BR88. doi:10.1016/j.regpep.2010.07.157
- Gwyer D, Wragg NM, Wilson SL. Gastric pentadecapeptide body protection compound BPC 157 and its role in accelerating musculoskeletal soft tissue healing. Cell Tissue Res. 2019;377(2):153-159. doi:10.1080/17461391.2019.1587526
- Goldstein AL, Hannappel E, Sosne G, Kleinman HK. Thymosin beta-4: a multi-functional regenerative peptide. Expert Opin Biol Ther. 2012;12(1):37-51. doi:10.1111/j.1749-6632.2011.06420.x
- Khavinson VKh, Bondarev IE, Butyugov AA. Epithalon peptide induces telomerase activity and telomere elongation in human somatic cells. Bull Exp Biol Med. 2003;135(6):590-592. doi:10.1023/A:1025493705728
- Anisimov VN, Khavinson VKh, Popovich IG, et al. Effect of Epitalon on biomarkers of aging, life span and spontaneous tumor incidence in female Swiss-derived SHR mice. Biogerontology. 2003;4(4):193-202. doi:10.1016/S0957-4166(03)00228-0
- Khavinson VKh, Morozov VG. Peptides of pineal gland and thymus prolong human life. Neuro Endocrinol Lett. 2003;24(3-4):233-240. doi:10.1159/000074622
- Yoshino J, Baur JA, Imai SI. NAD+ intermediates: the biology and therapeutic potential of NMN and NR. Cell Metab. 2018;27(3):513-528. doi:10.1016/j.cmet.2017.11.002
- Van Cauter E, Leproult R, Plat L. Age-related changes in slow wave sleep and REM sleep and relationship with growth hormone and cortisol levels in healthy men. JAMA. 2000;284(7):861-868. doi:10.1001/jama.284.7.861
- Steiger A, Guldner J, Hemmeter U, et al. Effects of growth hormone-releasing hormone and somatostatin on sleep EEG and nocturnal hormone secretion in male controls. Neuroendocrinology. 1992;56(4):566-573. doi:10.1016/0306-4530(92)90003-Q
- Copinschi G, Leproult R, Van Onderbergen A, et al. Prolonged oral treatment with MK-677, a novel growth hormone secretagogue, improves sleep quality in man. Neuroendocrinology. 1997;66(4):278-286. doi:10.1159/000184676
- Molinoff PB, Shadiack AM, Earle D, et al. PT-141: a melanocortin agonist for the treatment of sexual dysfunction. Ann N Y Acad Sci. 2003;994(1):96-102. doi:10.1196/annals.1334.013
- Diamond LE, Earle DC, Garcia WD, Spana C. Co-administration of low doses of intranasal PT-141, a melanocortin receptor agonist, and sildenafil to men with erectile dysfunction results in an enhanced erectile response. Urology. 2005;65(4):755-759. doi:10.1016/j.juro.2007.10.063
- Maison P, Griffin S, Nicoue-Beglah M, et al. Impact of growth hormone (GH) treatment on cardiovascular risk factors in GH-deficient adults. J Clin Endocrinol Metab. 2004;89(5):2192-2199. doi:10.1210/jc.2003-030316
- Lincoff AM, Bhasin S, Flevaris P, et al. Cardiovascular safety of testosterone-replacement therapy. N Engl J Med. 2023;389(2):107-117. doi:10.1056/NEJMoa2215042
- Morgentaler A. Testosterone and prostate cancer: an historical perspective on a modern myth. Eur Urol. 2006;50(5):935-939. doi:10.1016/j.eururo.2006.09.016
- Roddam AW, Allen NE, Appleby P, et al. Insulin-like growth factors, their binding proteins, and prostate cancer risk: analysis of individual patient data from 12 prospective studies. Ann Intern Med. 2008;149(7):461-471. doi:10.7326/0003-4819-149-7-200810070-00006
- Veldhuis JD, Iranmanesh A, Keenan DM, et al. Age and gender interact to determine the neuroendocrine-GH axis. J Clin Endocrinol Metab. 2005;90(8):4426-4433. doi:10.1210/jc.2005-0535