Executive Summary
Vasoactive intestinal peptide (VIP) is a 28-amino-acid neuropeptide that has emerged as one of the most clinically relevant molecules in the treatment of chronic inflammatory response syndrome (CIRS), mold-related illness, and a growing list of immune-mediated conditions. First isolated from porcine intestine in 1970 by Sami Said and Viktor Mutt, VIP was initially characterized for its potent vasodilatory properties. In the decades since, researchers have uncovered a far broader biological profile - one that places VIP at the intersection of immune regulation, neuroprotection, pulmonary physiology, gastrointestinal homeostasis, and circadian rhythm control.
Key Takeaways
- VIP is a 28-amino-acid endogenous neuropeptide with broad anti-inflammatory, immunomodulatory, and neuroprotective properties
- It signals through two G protein-coupled receptors (VPAC1 and VPAC2) expressed across immune cells, neurons, pulmonary tissue, and the GI tract
- In the Shoemaker CIRS Protocol, VIP nasal spray (50 mcg four times daily) is the final therapeutic step, used after all preceding interventions have been completed
- Published clinical data show 74% reduction in CIRS symptom scores, with normalization of C4a, TGF-beta 1, VEGF, and MMP-9
- VIP has been used safely in over 10,000 CIRS patients since 2008, with minimal adverse effects when administered as directed
For clinicians and patients dealing with the aftermath of water-damaged building exposure, VIP represents the final step in the Shoemaker Protocol - a structured, evidence-based treatment sequence developed by Dr. Ritchie Shoemaker over more than three decades of clinical practice. In patients who have completed the preceding steps of the protocol, VIP nasal spray has demonstrated the ability to correct persistent inflammatory markers, normalize dysregulated gene expression, and resolve symptoms that other interventions could not address. Published data from Shoemaker's IRB-approved studies show mean symptom score reductions of approximately 74%, with C4a and TGF-beta 1 levels returning to normal ranges in the majority of treated patients.
Beyond CIRS, VIP's therapeutic reach extends into pulmonary arterial hypertension, where early clinical trials demonstrated improvements in hemodynamic parameters and exercise capacity. In the gastrointestinal tract, VIP maintains epithelial barrier integrity, regulates secretory function, and has shown efficacy in preclinical models of inflammatory bowel disease. Its neuroprotective actions - mediated through suppression of microglial activation and promotion of neurotrophic factor release - have attracted attention in Alzheimer's disease research, Parkinson's disease models, and traumatic brain injury studies. VIP also plays an essential role in circadian clock synchronization within the suprachiasmatic nucleus, linking it to sleep-wake regulation and hormonal cycling.
This report provides a thorough examination of VIP's biology, receptor pharmacology, clinical applications, dosing protocols, safety profile, and its place among immune-modulating peptides. Whether you're a clinician considering VIP for a complex CIRS patient, a researcher exploring its anti-inflammatory mechanisms, or a patient trying to understand how this peptide might fit into your recovery plan, the following sections cover the evidence base in detail.
Key Takeaways
- VIP is a 28-amino-acid endogenous neuropeptide with broad anti-inflammatory, immunomodulatory, and neuroprotective properties
- It signals through two G protein-coupled receptors (VPAC1 and VPAC2) expressed across immune cells, neurons, pulmonary tissue, and the GI tract
- In the Shoemaker CIRS Protocol, VIP nasal spray (50 mcg four times daily) is the final therapeutic step, used after all preceding interventions have been completed
- Published clinical data show 74% reduction in CIRS symptom scores, with normalization of C4a, TGF-beta 1, VEGF, and MMP-9
- VIP has been used safely in over 10,000 CIRS patients since 2008, with minimal adverse effects when administered as directed
- Additional research supports VIP's role in pulmonary hypertension, inflammatory bowel disease, autoimmune arthritis, neurodegeneration, and circadian regulation
Figure 1: Overview of VIP's pleiotropic biological activities spanning immune modulation, neuroprotection, pulmonary vasodilation, GI barrier maintenance, and circadian clock regulation.
VIP Biology
Discovery and Structural Characterization
The story of vasoactive intestinal peptide begins in 1970, when Sami Said and Viktor Mutt isolated a novel peptide from porcine duodenal extracts that produced a pronounced and sustained drop in systemic blood pressure upon intravenous administration. They named it vasoactive intestinal peptide, reflecting both its vasodilatory potency and its intestinal origin. The peptide was subsequently sequenced and found to consist of 28 amino acid residues with the sequence: HSDAVFTDNYTRLRKQMAVKKYLNSILN. This placed VIP within the glucagon-secretin superfamily of peptides, which also includes pituitary adenylate cyclase-activating polypeptide (PACAP), secretin, glucagon, and growth hormone-releasing hormone (GHRH).
VIP shares approximately 68% sequence homology with PACAP-27, a relationship that has significant pharmacological implications. Both peptides bind to the same pair of receptors - VPAC1 and VPAC2 - with roughly equal affinity. This overlap means that the biological effects attributed to VIP in many experimental systems may also be partially mediated by PACAP, and vice versa. Understanding this receptor-sharing arrangement is essential for interpreting the research literature accurately.
The human VIP gene is located on chromosome 6q25 and encodes a prepro-VIP precursor that also contains peptide histidine methionine (PHM), another biologically active peptide. Post-translational processing yields the mature 28-amino-acid VIP peptide, which is C-terminally amidated - a modification that protects against enzymatic degradation and is required for full biological activity. Despite this protection, VIP has a relatively short plasma half-life of approximately 1-2 minutes when administered intravenously, owing to rapid degradation by neutral endopeptidases and dipeptidyl peptidase IV (DPP-IV). This short half-life has been both a clinical challenge and a driver of alternative delivery strategies, particularly intranasal administration.
Tissue Distribution and Sources
While VIP was first identified in the gut, subsequent research revealed an remarkably widespread distribution pattern. VIP-containing neurons are found throughout the central and peripheral nervous systems, establishing this molecule as a true neuropeptide rather than merely a gut hormone. In the central nervous system, VIP-expressing neurons are concentrated in the cerebral cortex (particularly layers II-IV), the hippocampus, the hypothalamus (with dense expression in the suprachiasmatic nucleus), the amygdala, and the brainstem. These central VIP neurons participate in cortical processing, memory formation, circadian rhythm generation, and autonomic regulation.
In the peripheral nervous system, VIP is a major neurotransmitter of parasympathetic and sensory neurons. It is found in nerve fibers innervating the respiratory tract (where it serves as a bronchodilator and regulator of mucous secretion), the gastrointestinal tract (where it modulates motility, secretion, and blood flow), the cardiovascular system, the urogenital tract, and exocrine glands including salivary, lacrimal, and sweat glands. VIP is also expressed by immune cells themselves, including T lymphocytes, mast cells, and eosinophils, creating autocrine and paracrine signaling loops within the immune system.
The enteric nervous system deserves special mention. VIP is one of the most abundant neuropeptides in the gut, found in intrinsic neurons of both the myenteric and submucosal plexuses. In the submucosal plexus, VIP-containing secretomotor neurons regulate epithelial ion and water transport. In the myenteric plexus, VIP serves as an inhibitory motor neuron transmitter, mediating relaxation of circular smooth muscle. This dual role makes VIP indispensable for normal digestive function, and disruptions in enteric VIP signaling have been linked to motility disorders, inflammatory bowel disease, and post-infectious irritable bowel syndrome.
VPAC1 and VPAC2 Receptors: Structure, Distribution, and Signaling
VIP exerts its biological effects primarily through two G protein-coupled receptors designated VPAC1 (also known as VIPR1) and VPAC2 (VIPR2). Both receptors belong to the class B (secretin) family of GPCRs, characterized by a large extracellular N-terminal domain that participates in ligand binding and seven transmembrane helices. A third receptor, PAC1, binds PACAP with much higher affinity than VIP and is generally considered a PACAP-preferring receptor, though it may contribute to VIP signaling in tissues where PACAP is absent.
VPAC1 and VPAC2 bind both VIP and PACAP with approximately equal affinity, displaying dissociation constants (Kd) in the range of 0.5-1 nM. Despite this shared ligand profile, the two receptors have distinct tissue distribution patterns that confer tissue-specific responses to VIP signaling.
VPAC1 Distribution and Functions
VPAC1 is widely expressed in the brain (cerebral cortex, hippocampus), lung, liver, intestinal epithelium, and on multiple immune cell types including T lymphocytes, macrophages, and dendritic cells. In the immune system, VPAC1 is considered the predominant receptor mediating VIP's anti-inflammatory effects on innate immune cells. Activation of VPAC1 on macrophages leads to suppression of pro-inflammatory cytokine production (TNF-alpha, IL-6, IL-12) and upregulation of the anti-inflammatory cytokine IL-10. On dendritic cells, VPAC1 signaling shifts antigen presentation toward a tolerogenic phenotype that promotes regulatory T cell differentiation.
Along the length of the intestine, VPAC1 expression follows a gradient pattern, with highest levels in the proximal small intestine and decreasing expression distally. This distribution pattern correlates with VIP's role in regulating intestinal secretion and nutrient absorption in the upper GI tract. In the lungs, VPAC1 is expressed on airway smooth muscle, pulmonary vascular smooth muscle, and alveolar epithelial cells, where it mediates bronchodilation and vasodilation.
VPAC2 Distribution and Functions
VPAC2 shows a somewhat different tissue distribution, with prominent expression in the central nervous system (thalamus, suprachiasmatic nucleus, hippocampus, brainstem), skeletal muscle, pancreas, heart, kidney, adipose tissue, and smooth muscle of the GI and reproductive tracts. In the immune system, VPAC2 is preferentially expressed on Th2 cells and is upregulated on activated T lymphocytes. VPAC2 signaling promotes Th2 differentiation and, through effects on dendritic cells and other antigen-presenting cells, contributes to regulatory T cell induction.
The suprachiasmatic nucleus (SCN) represents a particularly important site of VPAC2 expression. Here, VIP released by retinorecipient neurons signals through VPAC2 on neighboring clock neurons to synchronize circadian oscillations across the SCN network. Loss of either VIP or VPAC2 results in desynchronization of individual neuronal clocks, leading to disrupted circadian behavior, abnormal sleep-wake patterns, and dysregulated cortisol rhythms. This circadian function of VIP has direct clinical relevance for CIRS patients, many of whom exhibit disrupted sleep architecture and cortisol dysregulation.
Intracellular Signaling Cascades
Both VPAC1 and VPAC2 couple primarily to the stimulatory G protein Gs, leading to activation of adenylyl cyclase and elevation of intracellular cyclic AMP (cAMP). This cAMP increase activates protein kinase A (PKA) and exchange proteins directly activated by cAMP (EPACs), which then phosphorylate downstream targets to produce the cellular response. The cAMP-PKA pathway is the canonical signaling mechanism for most VIP effects, including suppression of NF-kB-dependent transcription of pro-inflammatory genes.
Beyond the classical cAMP pathway, VPAC receptors also engage additional signaling mechanisms. VPAC1 can interact with receptor activity-modifying proteins (RAMPs), particularly RAMP2, which shifts signaling toward phospholipase C activation and inositol trisphosphate (IP3) production, leading to calcium mobilization. This dual signaling capability allows VPAC1 to produce context-dependent responses depending on which RAMPs are co-expressed in a given cell type. VPAC2 can also signal through calcium-dependent pathways and activate the ERK1/2 mitogen-activated protein kinase cascade, which is particularly relevant to its circadian clock functions in the SCN.
| Feature | VPAC1 | VPAC2 |
|---|---|---|
| Gene | VIPR1 (chromosome 3p22) | VIPR2 (chromosome 7q36) |
| VIP Binding Affinity | Kd ~1 nM | Kd ~1 nM |
| Primary G Protein | Gs (cAMP elevation) | Gs (cAMP elevation) |
| Secondary Signaling | PLC/IP3/Ca2+ (via RAMPs) | ERK1/2, Ca2+ |
| Key CNS Expression | Cortex, hippocampus | SCN, thalamus, brainstem |
| Key Peripheral Expression | Lung, liver, intestine | Pancreas, muscle, heart |
| Immune Cell Expression | Macrophages, DCs, naive T cells | Th2 cells, activated T cells |
| Primary Immune Function | Innate immune suppression | Treg induction, Th2 promotion |
VIP in the Broader Neuropeptide Network
VIP does not operate in isolation. It functions within a complex network of neuropeptides, neurotransmitters, and immune mediators that collectively regulate the inflammatory response. Understanding VIP's relationships with other signaling molecules helps explain both its therapeutic potential and its limitations.
PACAP, as noted earlier, shares VIP's receptors and many of its biological effects. However, PACAP also signals through the PAC1 receptor, which VIP does not significantly activate. This gives PACAP additional neuronal survival and growth-promoting activities that complement VIP's anti-inflammatory focus. In clinical settings, VIP and PACAP may have partially overlapping but distinct therapeutic profiles.
Alpha-melanocyte stimulating hormone (alpha-MSH) is another anti-inflammatory neuropeptide that shares functional overlap with VIP. The tripeptide KPV, derived from the C-terminal of alpha-MSH, acts through melanocortin receptors to suppress NF-kB activation in a manner analogous to VIP's effects. Clinicians treating complex inflammatory conditions sometimes use both VIP and KPV in combination, reasoning that their distinct receptor pathways may produce additive anti-inflammatory coverage.
The antimicrobial peptide LL-37 represents the other side of the immune equation. While VIP dampens inflammatory responses, LL-37 enhances innate antimicrobial defense. In CIRS treatment, where patients must address both the infectious/toxin burden and the dysregulated immune response, these peptides serve complementary roles. Thymosin Alpha-1 provides yet another mechanism, directly enhancing T cell maturation and natural killer cell activity to support adaptive immune competence.
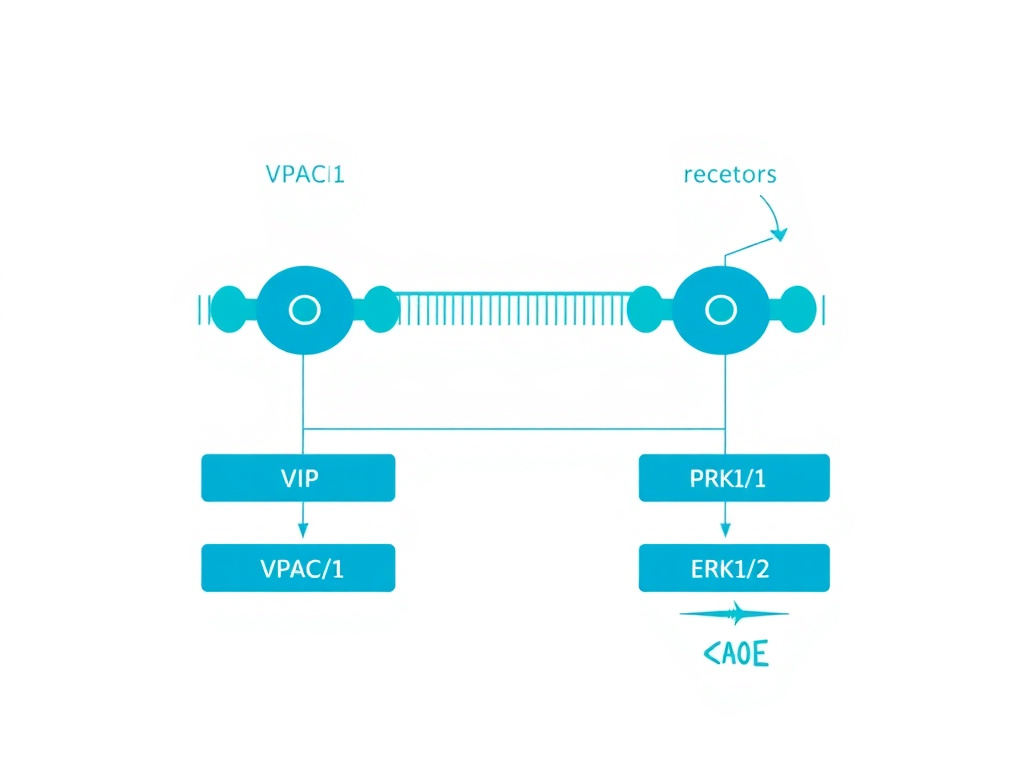
Figure 2: VIP receptor signaling through VPAC1 and VPAC2. Both receptors activate the Gs-cAMP-PKA pathway, with additional signaling through PLC/Ca2+ (VPAC1 via RAMPs) and ERK1/2 (VPAC2). These cascades converge on NF-kB inhibition and CREB activation to produce anti-inflammatory gene expression changes.
Endogenous VIP Regulation and Deficiency States
Under normal physiological conditions, VIP is released from nerve terminals in response to neuronal depolarization and acts locally on nearby target cells before being rapidly degraded by tissue peptidases. Circulating VIP levels in healthy individuals are typically in the low picomolar range (5-25 pg/mL), though local tissue concentrations near VIP nerve terminals are much higher.
Several disease states are associated with abnormally low VIP levels. In CIRS patients, serum VIP levels are frequently suppressed, often falling below the lower limit of the reference range. This VIP deficiency is thought to result from chronic inflammatory stress on VIP-producing neurons and may contribute to the perpetuation of the inflammatory cycle. Low VIP levels have also been documented in pulmonary arterial hypertension, where deficiency of VIP in lung tissue correlates with disease severity. In Alzheimer's disease, loss of VIP-expressing neurons in the cerebral cortex and SCN has been observed, potentially contributing to both the neuroinflammatory component of the disease and the circadian disruption that characterizes its later stages.
The concept of VIP deficiency as a driver of disease pathology provides the rationale for VIP replacement therapy. Rather than introducing a foreign pharmacological agent, VIP nasal spray can be understood as restoring a naturally occurring regulatory peptide to levels sufficient for proper immune and neurological function. This replacement paradigm is central to the Shoemaker Protocol's use of VIP in CIRS treatment.
CIRS and Mold Illness
Understanding Chronic Inflammatory Response Syndrome
Chronic inflammatory response syndrome (CIRS) is a multi-system, multi-symptom illness caused by exposure to biotoxin-producing organisms, most commonly molds found in water-damaged buildings. The condition was first formally described by Dr. Ritchie Shoemaker in the late 1990s, and its diagnostic criteria and treatment protocol have been refined through more than two decades of published clinical research. CIRS affects an estimated 25% of the population who carry HLA-DR genotypes that impair their ability to properly tag and clear biotoxins from the body. In these genetically susceptible individuals, exposure to mycotoxins, endotoxins, beta-glucans, and inflammagens produced by indoor mold colonies triggers a sustained inflammatory cascade that persists long after the exposure source is removed.
The pathophysiology of CIRS involves dysregulation of multiple immune and neuroendocrine pathways simultaneously. Innate immune activation leads to chronically elevated complement split products (C4a, C3a), transforming growth factor beta-1 (TGF-beta 1), matrix metalloproteinase-9 (MMP-9), and various inflammatory cytokines. At the same time, adaptive immune responses become dysregulated, with suppressed melanocyte-stimulating hormone (MSH), suppressed VIP, elevated antidiuretic hormone (ADH) or suppressed ADH, dysregulated sex hormones, and abnormal leptin signaling. The net result is a self-perpetuating cycle of inflammation that produces a characteristic symptom cluster spanning 13 organ systems.
Common CIRS symptoms include persistent fatigue, cognitive impairment ("brain fog"), headaches, joint and muscle pain, respiratory symptoms, light sensitivity, temperature dysregulation, numbness and tingling, mood disturbances, sleep disruption, gastrointestinal complaints, chronic sinusitis, and exercise intolerance. The breadth and variability of these symptoms often leads to misdiagnosis, with patients receiving labels ranging from fibromyalgia to chronic fatigue syndrome to anxiety disorder before the underlying CIRS is identified.
The Role of VIP in CIRS Pathophysiology
Within the complex web of CIRS pathophysiology, VIP occupies a uniquely important position. Multiple studies have documented that CIRS patients consistently show suppressed serum VIP levels, often falling below 20 pg/mL compared to normal ranges of 23-63 pg/mL. This VIP deficiency is not merely a marker of disease - it actively perpetuates the inflammatory cycle through several mechanisms.
First, low VIP removes a key brake on innate immune activation. Without adequate VIP signaling through VPAC1 on macrophages and dendritic cells, these cells remain in a chronically activated state, continuing to produce pro-inflammatory cytokines (TNF-alpha, IL-1beta, IL-6) and chemokines that recruit additional inflammatory cells. This loss of immune regulation contributes directly to the elevated C4a and TGF-beta 1 levels that characterize CIRS.
Second, VIP deficiency impairs regulatory T cell (Treg) generation. VIP signaling through VPAC2 is one of the mechanisms by which the immune system generates CD4+CD25+FoxP3+ Tregs, the cells responsible for maintaining immune tolerance and preventing autoimmune reactions. The loss of VIP-driven Treg induction may explain the autoimmune features (such as anti-gliadin antibodies and anticardiolipin antibodies) that develop in some CIRS patients over time.
Third, VIP deficiency disrupts pulmonary artery regulation. VIP is a potent pulmonary vasodilator, and its absence allows unchecked vasoconstriction and smooth muscle proliferation in the pulmonary vasculature. Shoemaker's research has documented that a subset of CIRS patients develop echocardiographic evidence of elevated pulmonary artery pressures, a finding that often improves with VIP replacement.
Fourth, the loss of VIP in the suprachiasmatic nucleus compromises circadian clock synchronization, contributing to the sleep disruption, cortisol dysregulation, and fatigue that are hallmarks of CIRS. Many CIRS patients report a characteristic pattern of non-restorative sleep, early morning awakening, and inability to maintain normal sleep-wake cycles, which may reflect impaired VIP-VPAC2 signaling in the master clock.
Biomarker Evidence for VIP's Role
The case for VIP's involvement in CIRS rests on multiple lines of biomarker evidence. In his published work, Shoemaker has documented the following patterns in CIRS patients before and after VIP treatment:
Biomarker Changes with VIP Treatment in CIRS
- C4a: Elevated pre-treatment (often >20,000 ng/mL; normal <2,830 ng/mL), decreasing to normal ranges after VIP therapy
- TGF-beta 1: Elevated pre-treatment (often >5,000 pg/mL; normal <2,380 pg/mL), normalizing with VIP administration
- VEGF: Often abnormally low or high in CIRS, tends to normalize with VIP
- MMP-9: Elevated pre-treatment, decreases toward normal with VIP
- VIP itself: Low pre-treatment (<20 pg/mL), increases with exogenous VIP replacement
- MSH: Often remains suppressed even after VIP (addressed by other protocol steps)
- Pulmonary artery systolic pressure: Elevated in a subset of patients, often normalizes with VIP
In Shoemaker's 2013 published study on VIP in CIRS, an IRB-approved clinical trial demonstrated that patients treated with VIP nasal spray showed mean symptom scores decreasing from 12.9 to 3.3, representing a 74% reduction. C4a levels fell significantly, TGF-beta 1 normalized in the majority of patients, and gene expression profiling using transcriptomic analysis revealed correction of previously dysregulated inflammatory gene clusters. The study also documented improvements in pulmonary function and exercise tolerance, as measured by visual contrast sensitivity (VCS) testing and VO2 max measurements.
Gene Expression and Transcriptomic Effects
One of the most compelling aspects of Shoemaker's VIP research is its use of transcriptomic analysis to document gene expression changes. Using differential gene expression profiling on peripheral blood mononuclear cells, Shoemaker's team identified clusters of genes that were abnormally activated in CIRS patients. These gene clusters corresponded to inflammatory pathways, apoptotic signaling, hormonal regulation, and immune cell trafficking.
Treatment with VIP nasal spray produced measurable corrections in these gene expression patterns. Specifically, genes involved in the complement cascade, TGF-beta signaling, and Th17-mediated inflammation showed reduced expression, while genes associated with regulatory T cell function and anti-inflammatory responses showed increased expression. These transcriptomic changes correlated with clinical improvement and biomarker normalization, providing molecular-level evidence that VIP was addressing the underlying pathophysiology rather than merely suppressing symptoms.
This gene expression data is particularly valuable because it helps explain why VIP must be the final step in the Shoemaker Protocol rather than a first-line intervention. The transcriptomic analysis showed that VIP could only produce its full gene expression correction in patients who had already completed the preceding steps (cholestyramine binding, MARCoNS eradication, VCS correction, and other interventions). Patients who attempted VIP before completing these steps showed incomplete gene expression correction and were more likely to experience symptom recurrence.
Figure 3: Biomarker response patterns in CIRS patients treated with VIP nasal spray. C4a, TGF-beta 1, and composite symptom scores show progressive improvement over 30-90 days of treatment.
Mold Illness: The Environmental Trigger
While CIRS can theoretically be triggered by various biotoxin sources (including Lyme disease, dinoflagellate exposure, and certain cyanobacteria), water-damaged building exposure accounts for the vast majority of cases seen in clinical practice. An estimated 50% of buildings in the United States have evidence of water damage, and the National Institute of Environmental Health Sciences (NIEHS) has recognized that mold exposure can contribute to symptoms beyond the traditionally acknowledged respiratory effects, including headaches, fatigue, cognitive difficulties, and mental health effects.
The biotoxin burden from water-damaged buildings is complex and multi-factorial. It includes mycotoxins (toxic metabolites produced by mold species such as Stachybotrys chartarum, Aspergillus, Penicillium, and Chaetomium), endotoxins (lipopolysaccharides from gram-negative bacteria that proliferate in wet building materials), beta-glucans (cell wall components of fungi that activate innate immune receptors), volatile organic compounds (VOCs produced by microbial metabolism), and inflammagens (small molecular weight toxins that directly activate complement and inflammatory cascades). The combined exposure to these multiple biotoxin categories helps explain why CIRS produces such a broad and systemic inflammatory response.
For CIRS patients, removing ongoing mold exposure is an absolute prerequisite for successful treatment. No amount of VIP or other therapeutic intervention can overcome active biotoxin exposure. The Shoemaker Protocol explicitly requires environmental remediation and confirmation of a safe living environment (typically through Environmental Relative Moldiness Index or ERMI testing) before VIP therapy is initiated. Patients who attempt VIP while still living or working in water-damaged buildings will typically show worsening of inflammatory markers rather than improvement, as VIP can unmask ongoing exposure by increasing the sensitivity of the immune system to environmental triggers.
VIP's Relationship to Other CIRS Biomarkers
Understanding how VIP interacts with the broader panel of CIRS biomarkers helps clinicians interpret laboratory results and make treatment decisions. Several key relationships deserve attention:
VIP and C4a: C4a is a complement split product that reflects activation of the innate immune system. In CIRS, C4a levels are often markedly elevated, sometimes exceeding 20,000 ng/mL (normal <2,830). VIP's anti-inflammatory signaling through VPAC1 helps suppress the complement activation cascade, and falling C4a levels are one of the earliest markers of VIP treatment response. However, a paradoxical rise in C4a after the first dose of VIP (the "VIP challenge test") can indicate ongoing mold exposure and should prompt environmental reassessment.
VIP and TGF-beta 1: TGF-beta 1 is a pleiotropic cytokine that drives fibrosis, Th17 differentiation, and tissue remodeling. Chronically elevated TGF-beta 1 in CIRS contributes to the autoimmune tendency and tissue damage that characterize advanced disease. VIP suppresses TGF-beta 1 production through its effects on macrophages and T cells, and normalization of TGF-beta 1 is considered a key treatment endpoint. When TGF-beta 1 remains elevated despite other interventions, it often responds specifically to VIP therapy.
VIP and VEGF: Vascular endothelial growth factor (VEGF) has a complex pattern in CIRS. It may be abnormally low (reflecting impaired angiogenesis and tissue perfusion) or abnormally high (reflecting a compensatory response to tissue hypoxia). VIP helps normalize VEGF levels in both directions, likely through its effects on endothelial cell function and its regulation of hypoxia-inducible factor pathways. Normalization of VEGF with VIP treatment often correlates with improved exercise tolerance and reduced fatigue.
VIP and MSH: Alpha-melanocyte stimulating hormone (MSH) is typically suppressed in CIRS and is one of the most difficult markers to normalize. While VIP can improve many CIRS parameters, MSH may remain low even after successful VIP therapy. This is because MSH production is regulated by multiple factors beyond VIP, including leptin signaling, hypothalamic function, and ongoing inflammatory drive. Persistent MSH suppression after VIP treatment may indicate the need for additional interventions or suggest that some residual inflammatory trigger remains present.
Shoemaker Protocol
Overview of the Multi-Step Treatment Sequence
The Shoemaker Protocol is the most extensively documented treatment approach for CIRS, developed over more than 30 years of clinical practice and refined through multiple published studies. It consists of a sequential series of interventions, each building on the previous step, designed to systematically dismantle the self-perpetuating inflammatory cycle that characterizes CIRS. VIP nasal spray occupies the final position in this sequence - Step 12 in most versions of the protocol - reflecting both its power as an immune modulator and the requirement that earlier steps be completed before VIP can work effectively.
The logic of sequential treatment is grounded in clinical observation and transcriptomic data. Shoemaker has documented that attempting to skip steps or administer VIP prematurely produces inferior results and may actually worsen inflammation. Each preceding step removes a specific barrier to recovery, and VIP's role is to correct the residual immune dysregulation that persists after all other barriers have been addressed. Think of it as fine-tuning an engine - you wouldn't adjust the timing before fixing a blown head gasket.
Steps Preceding VIP Therapy
While a complete discussion of each protocol step is beyond the scope of this VIP-focused report, understanding the preceding steps helps explain why VIP is reserved for the final position. The approximate sequence is as follows:
| Step | Intervention | Purpose |
|---|---|---|
| 1 | Remove from exposure | Eliminate ongoing biotoxin source; confirm safe environment via ERMI or HERTSMI-2 |
| 2 | Cholestyramine (CSM) or Welchol | Bind biotoxins in the GI tract to interrupt enterohepatic recirculation |
| 3 | Treat MARCoNS | Eradicate multiply antibiotic-resistant coagulase-negative staph from nasal passages |
| 4 | Correct antigliadin antibodies | Address gluten sensitivity and intestinal permeability (consider Larazotide for tight junction support) |
| 5 | Correct androgens | Normalize DHEA, testosterone, estradiol imbalances driven by CIRS |
| 6 | Correct ADH/osmolality | Address fluid regulation dysfunction (DDAVP if indicated) |
| 7 | Correct MMP-9 | Reduce matrix metalloproteinase-9 levels with high-dose omega-3 fatty acids |
| 8 | Correct VEGF | Normalize vascular endothelial growth factor levels |
| 9 | Correct C3a | Address complement activation (statin therapy if needed) |
| 10 | Correct C4a | Further complement regulation (often begins responding at this stage) |
| 11 | Correct TGF-beta 1 | Address elevated TGF-beta 1 with losartan if levels remain high |
| 12 | VIP nasal spray | Final immune modulation, gene expression correction, residual marker normalization |
The sequential nature of this protocol means that by the time a patient reaches the VIP step, many of their inflammatory markers will have already improved significantly. VIP's job is to address the residual dysregulation, particularly persistent elevations in C4a and TGF-beta 1, residual gene expression abnormalities, and ongoing symptoms that have not fully resolved with preceding interventions. In some patients, VIP produces dramatic final improvement; in others, the preceding steps have already done most of the heavy lifting, and VIP provides a more subtle but still measurable benefit.
The VIP Challenge Test
Before initiating ongoing VIP therapy, the Shoemaker Protocol calls for a VIP challenge test performed in the clinician's office. This test serves dual purposes: it screens for contraindications to VIP therapy, and it provides valuable diagnostic information about whether ongoing mold exposure is present.
The VIP challenge procedure involves the following steps:
- Draw baseline C4a and TGF-beta 1 levels
- Administer a single spray of VIP (50 mcg) into one nostril in the office
- Monitor the patient for 15-20 minutes for any adverse reactions (particularly hypotension or GI distress)
- Draw follow-up C4a and TGF-beta 1 levels 15 minutes after VIP administration
- Interpret the results based on the magnitude and direction of biomarker changes
The expected response in a patient ready for VIP therapy is a decrease or no significant change in C4a and TGF-beta 1 levels. A paradoxical doubling or greater increase in C4a after VIP administration is a "failed" challenge that typically indicates ongoing mold exposure. This occurs because VIP temporarily enhances immune surveillance, causing the immune system to mount a stronger response to environmental biotoxins that are still present. A failed VIP challenge should prompt the clinician to reassess the patient's living and working environments before proceeding with treatment.
The VIP challenge test also provides an opportunity to assess tolerance. While most patients tolerate VIP well, occasional patients experience nasal irritation, transient headache, or mild dizziness with the first dose. These reactions are generally self-limited and do not preclude ongoing therapy, but they allow the clinician to counsel the patient about what to expect.
Initiating VIP Therapy
Once a patient has passed the VIP challenge test, ongoing therapy begins with a structured titration protocol. The standard initial regimen, as described in Shoemaker's published work, involves:
Standard VIP Dosing Protocol for CIRS
- Month 1: 1 spray (50 mcg) into alternating nostrils, 4 times daily
- Month 2 (if tolerated): May increase to 2 sprays (100 mcg) per administration, 4 times daily in alternating nostrils
- Monitoring at 30 days: Recheck blood pressure, VCS, C4a, TGF-beta 1, and fasting lipase
- If markers improving: Begin taper to 2 doses daily over the next month
- Maintenance: Continue at 2 doses daily for 1 additional month, then discontinue
- 6-month follow-up: Recheck all markers to confirm stability after discontinuation
The total duration of VIP therapy in a typical CIRS case is approximately 2-4 months. This relatively short treatment course reflects VIP's ability to "reset" the immune system rather than merely suppressing symptoms. In most patients who have properly completed the preceding protocol steps, VIP produces lasting changes in gene expression and biomarker levels that persist after the peptide is discontinued. This durability of response distinguishes VIP from symptomatic treatments that require indefinite use.
Some patients, particularly those with severe or long-standing CIRS, may require longer courses of VIP therapy or periodic retreatment. Shoemaker has noted that patients who have been ill for many years before diagnosis may need extended VIP courses to achieve full gene expression correction. The decision to extend therapy is guided by serial biomarker monitoring and symptom assessment rather than by a fixed timeline.
Monitoring During VIP Treatment
Close laboratory and clinical monitoring during VIP therapy is essential for several reasons. First, it confirms that VIP is producing the expected anti-inflammatory effects. Second, it screens for the rare complication of lipase elevation, which would necessitate discontinuation. Third, it provides objective data to guide tapering decisions.
The recommended monitoring schedule includes:
- Baseline (pre-VIP): Complete CIRS panel including C4a, TGF-beta 1, MMP-9, VEGF, MSH, VIP, VCS, fasting lipase, and comprehensive metabolic panel
- 30 days: C4a, TGF-beta 1, fasting lipase, blood pressure, VCS
- 60-90 days (end of treatment): Full CIRS panel repeat
- 6 months post-discontinuation: Full CIRS panel to confirm durability of response
Fasting lipase monitoring deserves special emphasis. VIP receptors are expressed on pancreatic acinar cells, and in rare cases, VIP therapy can cause mild pancreatitis with lipase elevation. If fasting lipase rises above the normal range during treatment, VIP should be discontinued immediately. This complication is uncommon but has been documented, and its prompt identification prevents progression to clinically significant pancreatitis.
Clinical Outcomes and Published Data
The published evidence base for VIP in CIRS centers on Shoemaker's clinical studies, which have been peer-reviewed and published in indexed journals. The key findings from these studies include:
In the 2013 study published in the Health journal (Scientific Research Publishing), Shoemaker and House reported results from an IRB-approved trial of VIP nasal spray in CIRS patients who had completed the preceding protocol steps. Mean symptom scores decreased from 12.9 to 3.3, representing a 74% reduction. C4a levels showed significant decreases, with many patients achieving normal ranges for the first time since diagnosis. TGF-beta 1 followed a similar pattern of normalization. Visual contrast sensitivity testing showed improvement in most patients, indicating resolution of the neurotoxic effects that produce the characteristic visual deficits in CIRS.
Transcriptomic analysis in this study revealed that VIP corrected gene expression abnormalities in 85% of the inflammatory gene clusters that had been identified as dysregulated at baseline. This gene expression correction correlated with clinical improvement and marker normalization, providing mechanistic support for VIP's therapeutic effects. The gene expression data also demonstrated that VIP was not merely suppressing immune activity globally but was selectively correcting the specific pathways that had been dysregulated by biotoxin exposure.
Long-term follow-up data, while less extensively published, suggest that the improvements achieved with VIP are generally durable. Shoemaker has reported that the majority of patients who complete the full protocol including VIP maintain normal biomarker levels at 6-month and 12-month follow-up, provided they remain in clean environments. Relapse is most commonly associated with re-exposure to water-damaged buildings rather than a failure of the VIP-induced immune reset.
Since 2008, VIP nasal spray has been used in over 10,000 CIRS patients with documented safety, making this one of the largest real-world experience databases for any peptide therapy. While much of this experience exists in clinical registries rather than formal publications, it provides substantial reassurance about the safety and tolerability of VIP at the doses used in the Shoemaker Protocol.
Pulmonary Hypertension Research
The VIP-Pulmonary Vascular Axis
The lungs are among the most VIP-rich organs in the body. Dense networks of VIP-containing nerve fibers innervate pulmonary blood vessels, airway smooth muscle, submucosal glands, and alveolar epithelium. In the pulmonary vasculature specifically, VIP acts as a potent vasodilator and inhibitor of smooth muscle cell proliferation, two properties that make it directly relevant to pulmonary arterial hypertension (PAH), a progressive and often fatal condition characterized by elevated pulmonary artery pressures, right ventricular strain, and eventual right heart failure.
The connection between VIP and pulmonary hypertension was first established in 2003 when a group of Austrian researchers, led by Petkov and colleagues, published a landmark study in the Journal of Clinical Investigation demonstrating that VIP was deficient in both the serum and lung tissue of patients with idiopathic pulmonary arterial hypertension (IPAH). This finding was striking because it suggested that VIP deficiency was not just an epiphenomenon but potentially a contributing cause of the disease. The same study showed that VIP replacement produced substantial hemodynamic improvements in these patients.
Early Clinical Trial Results
In the 2003 JCI study, Petkov et al. administered inhaled VIP (aerosolized) to 8 patients with severe IPAH (mean pulmonary artery pressure >50 mmHg). A single dose of inhaled VIP produced:
- Significant reduction in mean pulmonary artery pressure
- Increased cardiac output
- Improved mixed venous oxygen saturation (SvO2), indicating better tissue oxygenation
- No significant change in systemic blood pressure, demonstrating pulmonary selectivity
The pulmonary selectivity of inhaled VIP was a particularly encouraging finding. Unlike systemic vasodilators, which often produce problematic systemic hypotension when used to treat PAH, inhaled VIP appeared to preferentially dilate the pulmonary vasculature. This selectivity likely reflects the high density of VIP receptors in pulmonary vascular smooth muscle combined with the direct delivery of the peptide to the lungs via inhalation, minimizing systemic exposure.
Following the initial acute study, a subsequent 3-month treatment trial was conducted. Patients receiving daily inhaled VIP showed sustained improvements in hemodynamic parameters, increased exercise capacity as measured by the 6-minute walk test, and improvement in mixed venous oxygen saturation. No significant adverse effects were reported during the treatment period. These results generated considerable excitement about VIP as a potential new treatment for PAH.
The Aviptadil Development Program
Aviptadil is the synthetic form of VIP that has been used in most clinical research. The name refers to the pharmaceutical-grade preparation of the 28-amino-acid peptide, identical in sequence to endogenous human VIP. Aviptadil has been formulated for multiple routes of administration, including intravenous infusion, inhalation, and intranasal delivery.
Following the promising early results in PAH, aviptadil was investigated in larger clinical studies. A multicenter trial reported by Leuchte et al. in 2008, published in the European Respiratory Journal, evaluated inhaled aviptadil in 20 patients with PAH. In this study, acute administration of aerosolized VIP produced selective pulmonary vasodilation with improved stroke volume and mixed venous oxygen saturation. Six of the 20 patients (30%) achieved a reduction in pulmonary vascular resistance greater than 20%, meeting the criteria for acute vasodilator responsiveness.
However, the field experienced a setback when subsequent longer-term studies failed to replicate the magnitude of improvement seen in the initial Austrian trials. A randomized controlled trial presented at a major cardiology meeting found no significant reduction in pulmonary vascular resistance or improvement in the 6-minute walk test compared to placebo. These disappointing results were not published in a peer-reviewed journal, making it difficult to evaluate the study design and patient selection criteria. Some researchers have speculated that differences in the VIP formulation, delivery device, dosing regimen, or patient population may account for the discrepant results.
Mechanistic Understanding: Why VIP Should Work in PAH
Despite the mixed clinical results, the mechanistic rationale for VIP in PAH remains strong. VIP acts on pulmonary vascular smooth muscle through multiple complementary mechanisms:
Vasodilation: VIP activates VPAC1 receptors on pulmonary vascular smooth muscle cells, leading to cAMP elevation and activation of protein kinase A. PKA phosphorylates myosin light chain kinase and opens potassium channels, producing smooth muscle relaxation and vasodilation. This mechanism is analogous to the effects of prostacyclin (another PAH treatment) but operates through a distinct receptor pathway.
Anti-proliferation: PAH is characterized not just by vasoconstriction but by pathological remodeling of the pulmonary vasculature, including smooth muscle cell proliferation and neointimal formation. VIP inhibits pulmonary artery smooth muscle cell proliferation through VPAC-mediated cAMP elevation, which suppresses the cell cycle progression required for proliferation. Both VPAC1 and VPAC2 contribute to this anti-proliferative effect.
Anti-inflammation: Inflammation plays an increasingly recognized role in PAH pathogenesis, with perivascular inflammatory infiltrates, elevated circulating cytokines, and autoimmune features identified in many patients. VIP's potent anti-inflammatory properties, including suppression of TNF-alpha, IL-6, and Th17 differentiation, directly address this inflammatory component. In CIRS-associated PAH, this anti-inflammatory mechanism may be particularly relevant.
Anti-fibrotic effects: Pulmonary vascular remodeling includes a fibrotic component driven by TGF-beta signaling and collagen deposition. VIP's ability to suppress TGF-beta 1 and reduce matrix metalloproteinase activity could theoretically counteract the fibrotic remodeling that contributes to fixed pulmonary vascular obstruction.
Combination Therapy Approaches
Recognizing that single-agent therapy may be insufficient for PAH, researchers have explored VIP in combination with established PAH treatments. A preclinical study published by Said et al. in the journal Pulmonary Circulation examined the combination of VIP with bosentan, an endothelin receptor antagonist, in a rat model of pulmonary hypertension. The combination was significantly more effective than either agent alone, with VIP completely preventing disease development in the prophylactic arm and significantly reversing established disease in the treatment arm. When combined with bosentan, the reversal of PAH was more complete than with either agent individually.
This combination approach makes mechanistic sense. Endothelin-1 (ET-1) is one of the most potent pulmonary vasoconstrictors and is elevated in PAH. VIP and ET-1 act on different receptor systems with opposing effects on vascular tone and smooth muscle proliferation. Blocking ET-1 while simultaneously stimulating VIP signaling addresses two pathological pathways simultaneously. Whether this preclinical promise translates to human PAH treatment remains to be determined through properly designed clinical trials.
Relevance to CIRS-Associated Pulmonary Hypertension
The connection between VIP and pulmonary hypertension takes on additional clinical relevance in the context of CIRS. Shoemaker has documented that a significant subset of CIRS patients develop echocardiographic evidence of elevated pulmonary artery pressures, sometimes meeting criteria for PAH. This CIRS-associated PAH appears to result from the combination of VIP deficiency (removing a pulmonary vasodilatory brake), chronic inflammation (promoting vascular remodeling), and endothelin dysregulation. Unlike idiopathic PAH, CIRS-associated elevations in pulmonary pressures often respond to VIP replacement therapy as part of the comprehensive CIRS treatment protocol.
This observation suggests that VIP deficiency-related PAH may be a distinct subtype that responds differently to VIP replacement than does advanced idiopathic PAH with fixed vascular remodeling. Future clinical trials of VIP for PAH might benefit from patient stratification based on VIP levels, inflammatory markers, and the degree of vascular remodeling, rather than treating PAH as a single homogeneous condition.
CIRS Symptom Score Reduction with VIP Nasal Spray
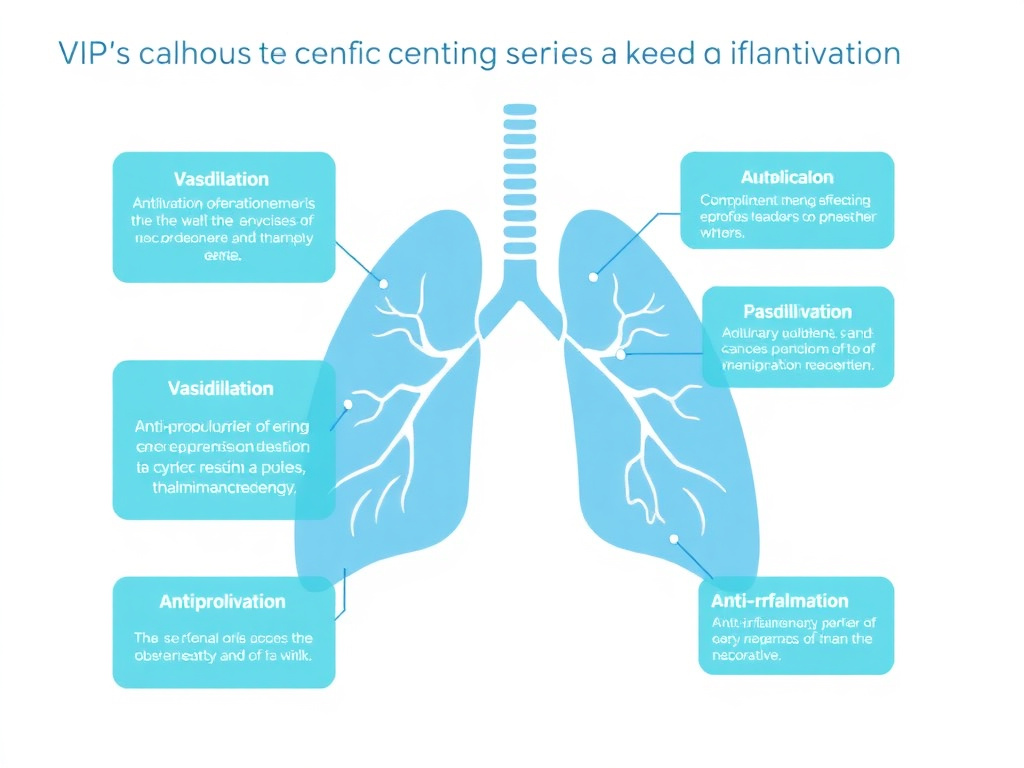
Figure 4: VIP's multi-mechanism approach to pulmonary hypertension. VIP simultaneously promotes vasodilation via cAMP-PKA signaling, inhibits smooth muscle proliferation, suppresses vascular inflammation, and reduces TGF-beta-mediated fibrotic remodeling.
Immune Regulation
VIP as a Master Immune Regulator
If there is a single phrase that captures VIP's role in the immune system, it would be "anti-inflammatory neuropeptide." But that label, while accurate, dramatically undersells the sophistication and breadth of VIP's immune regulatory functions. VIP doesn't simply suppress inflammation like a corticosteroid or NSAID. Instead, it modulates the immune response at multiple levels - from innate immune cell activation through antigen presentation to T cell differentiation - shifting the overall immune tone from a pro-inflammatory, tissue-damaging posture to a tolerogenic, healing-oriented one. This nuanced regulatory profile explains why VIP can address conditions as diverse as CIRS, autoimmune arthritis, inflammatory bowel disease, and transplant rejection without producing the broad immunosuppression and infection vulnerability associated with conventional immunosuppressive drugs.
Effects on Innate Immunity
The innate immune system - comprising macrophages, dendritic cells, neutrophils, and natural killer cells - is the first responder to pathogens and tissue damage. In chronic inflammatory conditions, these cells become hyperactivated, producing excessive amounts of pro-inflammatory mediators that cause collateral tissue damage. VIP modulates innate immune function through several well-characterized mechanisms:
Macrophage Polarization
Macrophages exist on a spectrum from classically activated (M1) pro-inflammatory phenotypes to alternatively activated (M2) anti-inflammatory and tissue-repair phenotypes. VIP promotes the shift from M1 to M2 polarization through VPAC1-mediated cAMP signaling. Specifically, VIP suppresses macrophage production of TNF-alpha, IL-1beta, IL-6, IL-12, CXCL8 (IL-8), and nitric oxide, while simultaneously increasing production of the anti-inflammatory cytokine IL-10. This doesn't render macrophages inactive - they retain their ability to phagocytose pathogens and debris - but it shifts their secretory profile from one that amplifies inflammation to one that promotes resolution and repair.
The mechanism involves VIP-mediated inhibition of NF-kB nuclear translocation. NF-kB is the master transcription factor for pro-inflammatory gene expression in macrophages. VIP, acting through cAMP-PKA, stabilizes the IkB-alpha inhibitor that normally keeps NF-kB sequestered in the cytoplasm. By preventing NF-kB from reaching the nucleus, VIP effectively turns down the transcription of an entire battery of pro-inflammatory genes simultaneously.
Dendritic Cell Tolerogenesis
Dendritic cells (DCs) are the primary antigen-presenting cells that bridge innate and adaptive immunity. The phenotype of DCs at the time of antigen presentation determines whether the adaptive immune response will be inflammatory (immunity) or tolerogenic (tolerance). VIP profoundly influences DC differentiation and function, promoting a tolerogenic phenotype characterized by:
- Reduced expression of co-stimulatory molecules (CD80, CD86, CD40)
- Decreased production of IL-12 (the major Th1-polarizing cytokine)
- Increased production of IL-10
- Enhanced expression of the inhibitory molecules ILT3 and PD-L1
- Preferential induction of regulatory T cells upon antigen presentation
This tolerogenic DC phenotype is particularly relevant to autoimmune conditions where inappropriate immune activation against self-antigens drives disease. By promoting tolerogenic antigen presentation, VIP can help restore immune tolerance to self-tissues without globally suppressing immune function. This mechanism also explains VIP's potential utility in transplant immunology, where tolerance to donor antigens is the therapeutic goal.
Effects on Adaptive Immunity
T Helper Cell Differentiation
CD4+ T helper cells differentiate into several functionally distinct subsets depending on the cytokine milieu present during activation. The major subsets relevant to VIP's effects include:
Th1 cells produce IFN-gamma and drive cellular immunity against intracellular pathogens. Excessive Th1 activity is associated with autoimmune tissue damage in conditions like rheumatoid arthritis, multiple sclerosis, and type 1 diabetes. VIP suppresses Th1 differentiation by reducing IL-12 production by DCs and macrophages, and by directly inhibiting IFN-gamma production by T cells.
Th2 cells produce IL-4, IL-5, and IL-13, driving humoral immunity and allergic responses. VIP promotes Th2 differentiation, shifting the Th1/Th2 balance toward Th2 predominance. This shift is therapeutically beneficial in Th1-driven autoimmune conditions but could theoretically worsen allergic or parasitic disease.
Th17 cells produce IL-17, IL-22, and IL-21, driving neutrophilic inflammation and playing pathological roles in many autoimmune and chronic inflammatory conditions. VIP suppresses Th17 differentiation through its effects on TGF-beta 1 and IL-6 signaling, two cytokines required for Th17 commitment. This anti-Th17 effect is particularly relevant to CIRS, where Th17-driven inflammation is a prominent feature.
Regulatory T cells (Tregs) express FoxP3 and produce IL-10 and TGF-beta, maintaining immune tolerance and suppressing excessive immune activation. VIP is one of the most potent known promoters of Treg differentiation. Through its effects on tolerogenic DCs and through direct VPAC2-mediated signaling on T cells, VIP increases the frequency of CD4+CD25+FoxP3+ Tregs and enhances their suppressive capacity. This Treg-promoting effect is central to VIP's ability to restore immune homeostasis in conditions characterized by immune dysregulation.
VIP's Net Effect on Immune Balance
VIP simultaneously suppresses pro-inflammatory pathways (Th1, Th17, M1 macrophages, inflammatory DCs) while promoting anti-inflammatory and tolerogenic pathways (Th2, Treg, M2 macrophages, tolerogenic DCs). The net result is a rebalancing of the immune system toward tolerance and tissue repair without global immunosuppression. This "immune recalibration" distinguishes VIP from conventional immunosuppressants that simply lower immune function across the board.
Autoimmune Disease Applications
Rheumatoid Arthritis
Rheumatoid arthritis (RA) has been one of the most extensively studied autoimmune conditions in VIP research. In the collagen-induced arthritis (CIA) mouse model, which closely mimics human RA, VIP treatment has consistently demonstrated remarkable efficacy. A study by Delgado and colleagues at Johns Hopkins showed that VIP administration completely prevented the development of CIA when given prophylactically, and significantly reduced disease severity when given therapeutically after disease onset. Treatment with VIP reduced both inflammatory and autoimmune components of the disease, abrogating joint swelling and destruction of cartilage and bone.
The mechanisms underlying VIP's anti-arthritic effects include suppression of pro-inflammatory cytokines in the joints (TNF-alpha, IL-6, IL-17), reduction of inflammatory cell infiltration, inhibition of osteoclastogenesis (bone destruction), and promotion of Treg activity. VIP also reduces the production of matrix metalloproteinases that degrade cartilage and collagen in arthritic joints.
Interestingly, research has found that RA patients show alterations in VIP receptor expression on their immune cells. VPAC1 expression is reduced on immune cells from RA patients compared to healthy controls, and these cells show decreased responsiveness to VIP stimulation. A VPAC1 agonist was highly efficient in treating experimental arthritis, suggesting that restoring VPAC1 signaling could be therapeutically beneficial. This receptor downregulation may represent both a consequence and a perpetuating factor of the chronic inflammatory state in RA, and it highlights the potential for VIP-based therapies that can overcome receptor desensitization.
Sarcoidosis
Sarcoidosis is a systemic granulomatous disease characterized by non-caseating granulomas in multiple organs, most commonly the lungs. It involves excessive Th1/Th17 immune activation and granuloma formation driven by overactive macrophages and dendritic cells. A study published in the American Journal of Respiratory and Critical Care Medicine demonstrated that inhaled VIP exerted immunoregulatory effects in sarcoidosis patients. VIP inhalation was safe, well-tolerated, and produced significant reduction in TNF-alpha production by cells isolated from bronchoalveolar lavage fluids. The study also showed increases in IL-10 production and shifts toward a less inflammatory cytokine profile in the lungs.
These findings position VIP as a potential therapeutic option for pulmonary sarcoidosis, a condition for which treatment options are limited and often involve long-term corticosteroid use with significant side effects. VIP's ability to suppress granulomatous inflammation while preserving antimicrobial immune function could offer a more targeted approach. However, larger clinical trials are needed before VIP can be recommended as a standard sarcoidosis treatment.
Other Autoimmune Conditions
Beyond RA and sarcoidosis, preclinical studies have demonstrated VIP efficacy in animal models of multiple sclerosis (experimental autoimmune encephalomyelitis), type 1 diabetes (NOD mice), Sjogren's syndrome, and systemic lupus erythematosus. In each of these models, VIP reduces disease severity by suppressing pro-inflammatory Th1/Th17 responses and promoting Treg-mediated tolerance. The breadth of these preclinical findings, combined with VIP's favorable safety profile in clinical use for CIRS and other conditions, supports further investigation of VIP in human autoimmune disease.
For patients dealing with immune dysregulation, several peptides available through the Peptide Hub may work in complementary ways. Thymosin Alpha-1 enhances thymic T cell education and NK cell activity, providing immune support that complements VIP's anti-inflammatory effects. Selank offers anxiolytic and immune-modulating properties that may address the neuropsychiatric components of autoimmune conditions. The Peptide Hub at FormBlends provides additional information about how these immune-modulating peptides relate to each other.
Figure 5: VIP's comprehensive immune regulatory network. VIP acts on macrophages (M1 to M2 shift), dendritic cells (tolerogenic phenotype), and T cell subsets (suppressing Th1/Th17, promoting Th2/Treg), producing coordinated immune rebalancing without global immunosuppression.
Comparison with Other Immune-Modulating Peptides
VIP occupies a distinct niche within the growing family of immune-modulating peptides. Understanding how it compares to related compounds helps clinicians make informed decisions about peptide selection and combination strategies.
| Peptide | Primary Mechanism | Immune Effect | Best Application |
|---|---|---|---|
| VIP | VPAC1/VPAC2 - cAMP elevation | Anti-inflammatory, Treg induction, M2 polarization | CIRS, autoimmunity, chronic inflammation |
| Thymosin Alpha-1 | TLR agonism, thymic stimulation | T cell maturation, NK cell activation, DC maturation | Immune deficiency, chronic infections, cancer immunotherapy |
| LL-37 | Membrane disruption, TLR modulation | Antimicrobial, wound healing, immune activation | Infections, biofilm disruption, wound healing |
| KPV | Melanocortin receptors - NF-kB inhibition | Anti-inflammatory, gut barrier support | IBD, colitis, mucosal inflammation |
| BPC-157 | NO system, VEGF modulation | Tissue repair, angiogenesis, anti-inflammatory | GI healing, tendon/ligament repair, organ protection |
| Selank | Tuftin-related, IL-6 modulation | Immune modulation, anxiolytic, cognitive support | Anxiety, immune support, cognitive enhancement |
The key distinction between VIP and immune-activating peptides like Thymosin Alpha-1 and LL-37 is directionality. VIP dampens overactive immune responses, while TA1 and LL-37 enhance immune competence. In complex conditions like CIRS, where the immune system is simultaneously overactive (excessive inflammation) and underperforming (poor pathogen clearance), a combination approach using both categories may be warranted. Clinicians using the Shoemaker Protocol sometimes pair VIP with TA1 to address both arms of immune dysfunction, though published data on this specific combination is limited. For more details on thymosin alpha-1's immune-enhancing properties, see the TA1 research report.
Gastrointestinal and Inflammatory Applications
VIP's Essential Role in Gut Physiology
Given that VIP was first discovered in intestinal tissue, it's fitting that the gastrointestinal tract remains one of the best-characterized sites of VIP action. VIP is one of the most abundant neuropeptides in the enteric nervous system, found in both the myenteric plexus (which controls motility) and the submucosal plexus (which regulates secretion and blood flow). Its GI functions are diverse and include regulation of epithelial water and electrolyte secretion, relaxation of smooth muscle (serving as an inhibitory motor neuron transmitter), dilation of mucosal blood vessels, modulation of mucosal immune responses, and maintenance of epithelial barrier integrity.
In the submucosal plexus, VIP-containing secretomotor neurons release VIP onto the basolateral surface of intestinal epithelial cells. VIP activates VPAC1 receptors on these cells, triggering cAMP-mediated opening of chloride channels (primarily CFTR) and stimulating bicarbonate secretion. This secretory function helps maintain the mucus layer and luminal environment necessary for normal digestion and microbial homeostasis. Excessive VIP release - as occurs in VIPoma tumors - produces profuse watery diarrhea, underscoring the potency of VIP's secretory effects.
In the myenteric plexus, VIP serves as the primary inhibitory neurotransmitter mediating descending relaxation during peristalsis. When a bolus of food triggers a peristaltic reflex, VIP-containing descending interneurons and motor neurons relax the circular smooth muscle ahead of the bolus, allowing it to pass through. Loss of VIP signaling in the myenteric plexus has been associated with motility disorders, including achalasia (failure of lower esophageal sphincter relaxation) and slow-transit constipation.
Intestinal Barrier Function
The intestinal epithelial barrier is a single cell layer that separates the vast microbial and antigenic load of the gut lumen from the body's internal environment. Maintaining the integrity of this barrier while still allowing nutrient absorption is a delicate balancing act, and VIP plays a role in both tight junction regulation and mucosal immune tolerance.
Research published in the journal Peptides demonstrated that VIP promotes intestinal barrier homeostasis and protects against colitis in mice. VIP-deficient mice showed increased intestinal permeability and were more susceptible to chemically induced colitis, while VIP replacement restored barrier function. The mechanism involves VIP-mediated upregulation of tight junction proteins (occludin, claudins, ZO-1) and suppression of inflammatory mediators that would otherwise disrupt the epithelial barrier.
This barrier-protective function has direct clinical relevance for CIRS patients, many of whom show evidence of increased intestinal permeability ("leaky gut"). The connection between CIRS, intestinal permeability, and food sensitivities (particularly gluten sensitivity, as reflected by anti-gliadin antibodies) is well-documented in Shoemaker's work. VIP's ability to support barrier integrity may contribute to the improvement in food sensitivities and GI symptoms that some patients experience during VIP therapy. For additional tight junction support, clinicians may consider Larazotide, a peptide specifically designed to modulate tight junction permeability.
Inflammatory Bowel Disease Research
Inflammatory bowel disease (IBD), encompassing Crohn's disease and ulcerative colitis, has been an active area of VIP research for over two decades. The rationale for VIP in IBD is straightforward: these are chronic inflammatory conditions of the gut driven by dysregulated immune responses (predominantly Th1/Th17 in Crohn's and Th2/NKT-mediated in ulcerative colitis), and VIP is a potent anti-inflammatory peptide with high local concentrations in the gut. However, the reality is more nuanced than this simple framework suggests, and the research has revealed some surprising complexities.
Crohn's Disease
In the trinitrobenzene sulfonic acid (TNBS) mouse model of Crohn's disease, VIP treatment produced striking therapeutic effects. Published in Gastroenterology by Abad et al. in 2003, this study showed that VIP administration reduced the clinical and histopathologic severity of TNBS-induced colitis, eliminating body weight loss, diarrhea, and both macroscopic and microscopic intestinal inflammation. The mechanism involved suppression of Th1 cytokines (IFN-gamma, TNF-alpha, IL-6, IL-12), reduction of inflammatory cell infiltration, and inhibition of NF-kB activation in colonic tissue.
VIP also reduced the production of chemokines (MIP-1alpha, MIP-1beta, RANTES, MCP-1) that recruit inflammatory cells to the gut, addressing the inflammatory cell trafficking component of Crohn's disease. This multi-level anti-inflammatory effect - targeting both the inflammatory mediators themselves and the signals that recruit inflammatory cells - is more comprehensive than most single-agent therapies currently used for Crohn's disease.
Ulcerative Colitis
The story in ulcerative colitis is more complex. Studies using the dextran sodium sulfate (DSS) colitis model, which more closely resembles ulcerative colitis, have revealed receptor-specific effects that highlight the importance of understanding which VIP receptor is mediating the response. Mice lacking VPAC2 developed more severe DSS-induced colitis with greater weight loss and more severe histopathology than wild-type mice, suggesting that VPAC2 signaling is protective. Conversely, VPAC1-knockout mice actually showed milder colitis than wild-type animals, suggesting that VPAC1 signaling may have pro-inflammatory effects in certain colitis contexts.
This paradoxical finding - that one VIP receptor is protective while the other may be harmful in the same disease - illustrates the complexity of VIP pharmacology and cautions against assuming that VIP (which activates both receptors equally) will be uniformly beneficial in all inflammatory bowel conditions. Development of receptor-selective VIP analogs that preferentially activate VPAC2 while sparing VPAC1 could potentially improve the therapeutic profile in ulcerative colitis.
VIP Nanomedicine Approaches
One of the major challenges of VIP therapy for IBD is drug delivery. VIP's short plasma half-life (1-2 minutes) makes systemic administration impractical for achieving sustained local concentrations in the inflamed gut. To address this, researchers have developed VIP-loaded nanoparticle formulations designed for targeted intestinal delivery. A 2018 review in the journal Nano-Micro Letters described sterically stabilized micelles loaded with VIP that could be administered orally and release VIP preferentially at sites of intestinal inflammation. These nanoformulations showed improved stability, extended release kinetics, and enhanced local anti-inflammatory effects compared to free VIP in preclinical IBD models.
The nanomedicine approach to VIP delivery is an active area of research that could eventually enable oral VIP therapy for IBD and other GI inflammatory conditions. For now, intranasal VIP remains the primary clinical route of administration, with potential GI benefits occurring through systemic (absorbed) rather than local (luminal) VIP exposure.
VIP and the Gut Microbiome
An emerging area of VIP research concerns its relationship with the intestinal microbiome. A study published in Frontiers in Microbiology demonstrated that VIP-deficient mice have significantly altered gut microbiota communities compared to wild-type animals. VIP knockout mice showed reduced microbial diversity and shifts in the relative abundance of specific bacterial phyla, including decreased Bacteroidetes and increased Firmicutes. These microbial changes were associated with altered intestinal immune tone and increased susceptibility to inflammatory stimuli.
The mechanism by which VIP influences the microbiome is likely indirect, operating through VIP's effects on intestinal secretion (which shapes the luminal environment), mucosal immunity (which selectively tolerates or eliminates specific microbes), and epithelial barrier function (which controls microbial access to the immune system). These findings suggest that VIP deficiency - whether genetic or acquired (as in CIRS) - may contribute to dysbiosis, adding yet another dimension to VIP's role in maintaining intestinal health.
For patients seeking comprehensive gut support, VIP may be combined with other peptides that address different aspects of GI healing. BPC-157 promotes mucosal repair and angiogenesis, making it a natural complement to VIP's anti-inflammatory and barrier-protective effects. KPV provides additional NF-kB inhibition specifically at mucosal surfaces. Together, these peptides address inflammation, barrier dysfunction, and tissue repair - three pillars of GI recovery.
Neuroprotective Effects
VIP in the Central Nervous System
The brain is one of the richest sources of VIP in the body, with VIP-expressing interneurons distributed throughout the cerebral cortex, hippocampus, amygdala, hypothalamus, and brainstem. These VIP neurons participate in cortical processing, memory consolidation, circadian rhythm generation, and neuroimmune regulation. Given this extensive central presence, it's not surprising that VIP has emerged as a molecule of significant interest in neuroscience research, with particular focus on its neuroprotective and neuroimmune modulatory properties.
Mechanisms of Neuroprotection
VIP protects neurons through both direct and indirect mechanisms. The direct neuroprotective effects involve VIP signaling on neurons themselves, while the indirect effects are mediated through VIP's actions on glial cells - microglia and astrocytes - that form the brain's resident immune and support system.
Direct Neuronal Effects
VIP promotes neuronal survival through VPAC-mediated cAMP elevation, which activates CREB (cAMP response element-binding protein), a transcription factor that drives expression of neuroprotective genes including brain-derived neurotrophic factor (BDNF), nerve growth factor (NGF), and activity-dependent neuroprotective protein (ADNP). ADNP is of particular interest because it was originally identified as a VIP-responsive gene, and a small peptide fragment of ADNP called NAP (davunetide) has been investigated as a neuroprotective therapy in its own right. The VIP-ADNP-NAP pathway represents one of the brain's endogenous neuroprotective circuits.
VIP also supports neuronal survival by maintaining intracellular calcium homeostasis. Excitotoxic neuronal death, which occurs in stroke, traumatic brain injury, and neurodegenerative disease, involves excessive calcium influx through glutamate receptors. VIP helps buffer this calcium overload through cAMP-PKA-mediated modulation of calcium channels and calcium-binding protein expression. This anti-excitotoxic mechanism may contribute to VIP's protective effects in acute neurological injury.
Microglial Modulation
Perhaps the most clinically relevant neuroprotective mechanism of VIP involves its effects on microglia, the brain's resident immune cells. Under normal conditions, microglia survey the brain parenchyma for damage, infection, and abnormal protein aggregates. When activated by inflammatory signals, trauma, or pathological protein deposits (such as beta-amyloid plaques in Alzheimer's disease), microglia shift to a pro-inflammatory phenotype that produces TNF-alpha, IL-1beta, nitric oxide, and reactive oxygen species. While this inflammatory response is initially protective (clearing debris and pathogens), chronic microglial activation produces sustained neuroinflammation that damages bystander neurons and accelerates neurodegenerative processes.
VIP acts as a potent suppressor of microglial activation. Published research by Delgado and colleagues in The FASEB Journal demonstrated that VIP prevents activated microglia-induced neurodegeneration under inflammatory conditions. The mechanism involves VPAC1-mediated suppression of NF-kB activation in microglia, leading to reduced production of TNF-alpha, IL-1beta, nitric oxide, and reactive oxygen species. VIP-treated microglia shift from a neurotoxic to a neuroprotective phenotype, producing neurotrophic factors instead of inflammatory mediators. A 2024 study in the European Journal of Neuroscience further confirmed that microglia cells treated with synthetic VIP or transduced with a VIP-expressing lentiviral vector protected neuronal cells against degeneration in co-culture systems.
This microglial modulation mechanism is directly relevant to brain fog and cognitive impairment in CIRS. Chronic neuroinflammation driven by circulating inflammatory mediators (which cross the disrupted blood-brain barrier in CIRS) activates microglia, producing sustained neurotoxicity that manifests clinically as cognitive dysfunction, memory impairment, difficulty with word finding, and reduced processing speed. VIP's ability to calm microglial activation may explain the cognitive improvements reported by CIRS patients during VIP therapy.
Alzheimer's Disease Research
Alzheimer's disease (AD) involves the progressive accumulation of beta-amyloid plaques and neurofibrillary tau tangles, accompanied by chronic neuroinflammation, synaptic loss, and neuronal death. VIP has been investigated as a potential therapeutic agent in AD through several lines of research:
Beta-amyloid clearance: A study published in PLOS ONE demonstrated that VIP enhances microglial phagocytosis of fibrillar beta-amyloid (Abeta-42) and attenuates amyloid deposition in the brains of APP/PS1 transgenic mice (a standard AD model). Co-incubation with VIP significantly increased microglial uptake of amyloid while simultaneously reducing the inflammatory activation that typically accompanies amyloid clearance. This "clean phagocytosis" - amyloid removal without inflammatory damage - addresses two pathological mechanisms simultaneously.
5xFAD mouse model: Research published by Korkmaz et al. demonstrated that VIP decreases beta-amyloid accumulation and prevents brain atrophy in the 5xFAD mouse model of Alzheimer's disease. Chronic VIP treatment reduced amyloid plaque burden, preserved hippocampal volume, and maintained cognitive function as measured by behavioral testing. These findings suggest that VIP's neuroprotective effects extend beyond symptom management to actual disease modification in the AD context.
VIP neuron loss in AD: Post-mortem studies of Alzheimer's disease brains have documented significant loss of VIP-expressing neurons in the cerebral cortex and the suprachiasmatic nucleus. This neuronal loss creates a VIP-deficient state in the brain that may accelerate neuroinflammation, disrupt circadian function (the "sundowning" phenomenon in AD may reflect SCN VIP neuron loss), and impair the brain's endogenous neuroprotective capacity. VIP replacement therapy could theoretically compensate for this neuronal loss, though the challenge of delivering VIP across the blood-brain barrier remains a significant obstacle.
Parkinson's Disease Research
Parkinson's disease (PD) involves progressive loss of dopaminergic neurons in the substantia nigra, driven by both direct alpha-synuclein toxicity and neuroinflammation. VPAC2 receptor agonists have shown promise in PD models by inducing regulatory T cell neuroprotective activities. A study published by Olson et al. demonstrated that a synthetic VPAC2 agonist increased Treg frequency and function, reduced neuroinflammation, and protected dopaminergic neurons from degeneration in a mouse model of PD. The mechanism involved Treg-mediated suppression of microglial activation and reduction of inflammatory mediators in the substantia nigra.
This Treg-mediated neuroprotection represents an indirect but potentially powerful therapeutic approach. Rather than directly protecting neurons (which would require the drug to cross the blood-brain barrier), the strategy uses peripheral immune modulation (Treg induction) to reduce the neuroinflammatory component of PD. Since Tregs naturally traffic to sites of inflammation, including the brain, VIP-induced Tregs could provide sustained neuroprotection without requiring direct CNS penetration of the peptide.
Circadian Rhythm Regulation
One of VIP's most fascinating and underappreciated roles is its function as the master synchronizer of the circadian clock. The suprachiasmatic nucleus (SCN) of the hypothalamus serves as the body's central circadian pacemaker, generating the ~24-hour rhythms that govern sleep-wake cycles, hormone secretion, body temperature, metabolism, and immune function. Within the SCN, VIP-expressing neurons receive light input from the retina (via the retinohypothalamic tract) and use VIP signaling through VPAC2 receptors to synchronize the molecular clocks of neighboring SCN neurons.
Research published in Nature Communications by Hamnett et al. (2019) demonstrated that VIP controls the suprachiasmatic circadian clock network via ERK1/2 and DUSP4 signaling. The study showed that VIP released by retinorecipient SCN neurons activates VPAC2 on target neurons, triggering ERK1/2 phosphorylation and subsequent activation of Per gene transcription - core components of the molecular clock machinery. DUSP4, a phosphatase that negatively regulates ERK1/2, acts as a tuning mechanism that controls the sensitivity of clock neurons to VIP signaling.
The consequences of disrupted VIP-VPAC2 signaling in the SCN are severe. Mouse models lacking VIP or VPAC2 show fragmented behavioral rhythms, loss of coordinated clock gene oscillation across SCN neurons, disrupted sleep-wake patterns, and abnormal corticosterone (cortisol in humans) secretion. VIP is also critical for circadian regulation of glucocorticoids, with VIP-deficient animals showing blunted cortisol rhythms and impaired adrenal responses to stress.
These circadian findings have direct clinical implications for CIRS and other conditions treated with VIP. CIRS patients frequently exhibit disrupted sleep architecture, abnormal cortisol patterns, and desynchronized circadian rhythms. VIP deficiency in CIRS may contribute directly to these circadian disturbances through loss of SCN synchronizing function. VIP replacement therapy may help restore normal circadian organization, potentially improving sleep quality, cortisol regulation, and the numerous physiological processes that depend on proper circadian timing. This may also be relevant for patients using NAD+, which has its own connections to circadian metabolism and cellular energy regulation.
Figure 6: VIP's neuroprotective mechanisms. The peptide protects neurons through direct neurotrophic signaling (cAMP-CREB-BDNF), microglial modulation (M1 to M2 shift), enhanced amyloid clearance, Treg-mediated neuroinflammation control, and circadian clock synchronization in the SCN.
Traumatic Brain Injury
Traumatic brain injury (TBI) triggers a secondary inflammatory cascade that can be more damaging than the initial mechanical injury. Activated microglia, infiltrating peripheral immune cells, and disrupted blood-brain barrier integrity produce a neuroinflammatory environment that causes ongoing neuronal death for days to weeks after the primary injury. VIP's ability to suppress microglial activation and reduce neuroinflammation positions it as a potential intervention for the secondary injury phase of TBI.
Preclinical research has demonstrated that VIP administration after experimental TBI reduces microglial activation markers, decreases brain edema, preserves blood-brain barrier integrity, and improves functional outcomes in behavioral testing. The therapeutic window for VIP in TBI appears to extend at least several hours after injury, coinciding with the time course of secondary inflammatory activation. While human clinical trials of VIP for TBI have not yet been conducted, the preclinical data supports its investigation in this devastating condition.
Dosing and Administration
Intranasal VIP: The Primary Clinical Route
The intranasal route has become the standard method for clinical VIP administration, particularly in CIRS treatment. This choice reflects several practical and pharmacological considerations. First, nasal delivery bypasses first-pass hepatic metabolism, allowing a greater proportion of the administered dose to reach systemic circulation compared to oral administration (which would largely be destroyed by GI peptidases). Second, the nasal mucosa provides a relatively large absorptive surface area with rich blood supply, enabling reasonable bioavailability for a peptide of VIP's molecular size. Third, a portion of intranasally administered peptides can access the brain via the olfactory and trigeminal nerve pathways, potentially delivering VIP directly to the CNS without requiring blood-brain barrier penetration. Fourth, intranasal delivery is non-invasive, self-administered, and practical for the multi-dose daily regimens required in CIRS treatment.
Pharmacokinetic studies of intranasal VIP have characterized its absorption and distribution profile. A study published by Gressens et al. evaluated the pharmacodynamics and toxicity of VIP for intranasal administration. Peak plasma VIP levels are achieved within approximately 15-30 minutes of intranasal dosing, with an effective half-life significantly longer than intravenous VIP due to the depot effect of the nasal mucosa (VIP is gradually absorbed from the mucosal surface over time). The nasal bioavailability of VIP has been estimated at approximately 1-5% of the administered dose, which, combined with the high potency of VIP at nanomolar receptor concentrations, is sufficient to produce measurable biological effects.
VIP Nasal Spray Formulation
Clinical VIP nasal spray is prepared as a compounded medication, typically by specialty compounding pharmacies. The standard formulation contains VIP at a concentration that delivers 50 mcg per spray actuation in a preserved saline vehicle. Some formulations use benzalkonium chloride as a preservative, while others use alternative preservative systems or are prepared preservative-free with limited shelf life.
Storage requirements for compounded VIP nasal spray are important for maintaining peptide stability. VIP should be refrigerated (2-8 degrees C / 36-46 degrees F) when not in active use. During active use, the spray can be kept at room temperature for short periods (typically up to the duration between refills). Exposure to excessive heat, light, or contamination can degrade the peptide and reduce efficacy. Patients should be counseled to avoid touching the spray tip to nasal tissue (to prevent bacterial contamination) and to prime the spray device before first use and after any extended period of non-use.
Compounding and Regulatory Status
VIP nasal spray is a compounded medication, not an FDA-approved drug product. It has not been evaluated by the FDA for safety, quality, or efficacy in the treatment of any specific disease. Compounded VIP should be obtained only through licensed compounding pharmacies operating under appropriate state and federal regulations. The quality of compounded VIP can vary between pharmacies, and clinicians should work with pharmacies that perform potency testing and stability studies on their VIP formulations.
Dosing Protocols by Indication
CIRS (Shoemaker Protocol - Standard)
The most extensively documented VIP dosing protocol is the one developed by Shoemaker for CIRS treatment. This protocol follows a structured titration and taper schedule:
| Phase | Duration | Dose | Frequency | Notes |
|---|---|---|---|---|
| Challenge | Single dose | 50 mcg (1 spray) | Once, in office | Monitor C4a/TGF-beta 1 at 15 min; screen for adverse reactions |
| Initiation | Month 1 | 50 mcg (1 spray) | 4 times daily | Alternate nostrils; total daily dose 200 mcg |
| Escalation (optional) | Month 2 | 100 mcg (2 sprays) | 4 times daily | Only if tolerated and markers not yet normalized; total daily dose 400 mcg |
| Taper | Month 3 | 50 mcg (1 spray) | 2 times daily | Begin if markers improving; total daily dose 100 mcg |
| Maintenance | Month 4 | 50 mcg (1 spray) | 2 times daily | Continue for 1 month, then discontinue |
| Follow-up | 6 months post-stop | None | N/A | Recheck full CIRS panel to confirm durability |
CIRS - Severe or Refractory Cases
Patients with severe, long-standing CIRS or those who show incomplete biomarker normalization during the standard protocol may benefit from modified approaches:
- Extended duration: Continue VIP at therapeutic doses for 3-6 months rather than the standard 2-4 months, guided by serial biomarker monitoring
- Higher dose: Some clinicians use up to 100 mcg four times daily (400 mcg/day) for the full treatment period in refractory cases, monitoring lipase closely
- Intermittent courses: For patients who relapse after initial VIP therapy, repeated courses of VIP can be administered. Some patients require 2-3 treatment courses to achieve stable remission
- Combination therapy: Pairing VIP with Thymosin Alpha-1 for concurrent immune support, or with BPC-157 for GI healing, may improve outcomes in complex cases, though published data on specific combinations is limited
Pulmonary Applications (Investigational)
For pulmonary indications (PAH, sarcoidosis), VIP has been administered via inhalation rather than intranasal delivery. The inhaled route delivers VIP directly to the pulmonary vasculature and airways, achieving high local concentrations while minimizing systemic exposure. Published pulmonary dosing protocols have used aerosolized VIP at doses ranging from 100-200 mcg per inhalation session, administered 1-4 times daily. These protocols remain investigational and should only be pursued in research settings or under direct specialist supervision.
Timing and Administration Tips
Based on clinical experience and published guidance, the following practical tips apply to VIP nasal spray administration:
- Timing: Space doses approximately evenly throughout waking hours (e.g., morning, midday, afternoon, evening). Some clinicians recommend a dose close to bedtime to support circadian VIP signaling during sleep.
- Technique: Gently blow nose before administration to clear mucus. Tilt head slightly forward, insert spray tip into one nostril, and actuate while inhaling gently. Avoid sniffing forcefully, which may deposit the spray in the pharynx rather than on the nasal mucosa.
- Alternating nostrils: Switch between left and right nostrils with each dose to minimize local irritation and maximize mucosal absorption area.
- Empty stomach: While not strictly required, some practitioners recommend administering VIP away from meals for more consistent absorption.
- Concurrent nasal medications: If using other nasal sprays (corticosteroids, antihistamines), space them at least 15-30 minutes apart from VIP to avoid dilution or interference.
Patients interested in exploring VIP therapy can use the Dosing Calculator at FormBlends for general guidance, and the Free Assessment can help determine whether VIP may be appropriate based on individual health status.
Figure 7: VIP nasal spray administration guide showing proper intranasal technique, the standard CIRS dosing schedule with titration and taper phases, and recommended monitoring timepoints.
Safety
Overall Safety Profile
VIP has an established safety record that spans multiple clinical applications and routes of administration. In the CIRS context, Shoemaker has documented the use of VIP nasal spray in over 10,000 patients since 2008, describing safety as "unmatched" in this population. The most comprehensive safety data comes from CIRS treatment, where the standardized dosing protocol and regular monitoring provide a structured framework for identifying and managing adverse effects.
The favorable safety profile of VIP can be attributed to several factors. First, VIP is an endogenous human peptide - patients are receiving a molecule that their bodies normally produce, at doses designed to restore physiological levels rather than achieve supraphysiological effects. Second, intranasal administration delivers relatively small total systemic doses (compared to intravenous infusion), reducing the risk of systemic side effects. Third, the short half-life of VIP means that any adverse effects tend to be transient, resolving quickly after the peptide is cleared. Fourth, the Shoemaker Protocol's prerequisite of completing earlier treatment steps before VIP initiation ensures that patients begin VIP therapy with many of their inflammatory markers already improved, reducing the risk of inflammatory flares.
Common Side Effects
Based on published reports and clinical experience, the following side effects have been reported with intranasal VIP:
| Side Effect | Frequency | Severity | Management |
|---|---|---|---|
| Nasal irritation or dryness | Common (10-20%) | Mild | Alternate nostrils; saline rinse before VIP; consider preservative-free formulation |
| Transient headache | Occasional (5-10%) | Mild to moderate | Usually resolves within first week; acetaminophen if needed |
| Mild dizziness | Occasional (5-10%) | Mild | Sit or lie down after administration; may reflect mild vasodilation |
| Nasal congestion or rhinorrhea | Occasional | Mild | Usually transient; clear nose before dosing |
| GI symptoms (mild nausea, loose stools) | Uncommon (2-5%) | Mild | Usually self-limited; reduce dose if persistent |
| Mild blood pressure reduction | Uncommon | Mild | Monitor BP, especially in patients on antihypertensives; orthostatic precautions |
| Flatulence | Rare | Mild | Self-limited |
Serious Adverse Effects
Serious adverse effects from intranasal VIP at the doses used in CIRS treatment are rare but have been reported:
Lipase Elevation and Pancreatitis
The most important serious adverse effect to monitor is lipase elevation, which may indicate VIP-induced pancreatic irritation. VIP receptors are expressed on pancreatic acinar cells, and exogenous VIP stimulates pancreatic exocrine secretion. In rare cases, this stimulation can produce clinical or subclinical pancreatitis. Shoemaker's protocol mandates fasting lipase monitoring at baseline and 30 days, with immediate discontinuation of VIP if lipase rises above the normal range. When caught early through monitoring, VIP-related lipase elevation is reversible upon discontinuation. The estimated incidence based on available clinical data is less than 1-2% of treated patients.
Risk factors for VIP-related lipase elevation may include pre-existing pancreatic disease, alcohol use, concurrent medications that affect pancreatic function, and hypertriglyceridemia. Patients with these risk factors should be monitored more closely during VIP therapy.
Hypotension
VIP is a systemic vasodilator, and at higher doses or in sensitive individuals, it can produce clinically significant blood pressure reduction. Cardiovascular studies in healthy volunteers have shown that VIP infusion can decrease total peripheral resistance and mean arterial pressure by 12-30%, depending on the dose and route of administration. Intranasal VIP at CIRS treatment doses produces much more modest hemodynamic effects, but patients who are already hypotensive (which is common in CIRS) or who are taking antihypertensive medications should be monitored for excessive blood pressure reduction, particularly during the initial dosing period.
Diarrhea (High Doses)
VIP is a potent intestinal secretagogue, and at high doses, it can produce significant watery diarrhea. This is the defining feature of VIPoma syndrome, where VIP-secreting tumors produce massive diarrhea. At therapeutic intranasal doses, diarrhea is uncommon and mild. However, if doses are escalated aggressively or if systemic VIP levels become elevated for any reason, secretory diarrhea can develop. Preclinical data from murine studies showed that at higher doses (10 nmol), VIP worsened diarrhea, possibly due to receptor downregulation. This observation reinforces the importance of using VIP at established therapeutic doses rather than escalating empirically.
Contraindications
Based on the mechanism of action and available safety data, the following contraindications and precautions apply to VIP therapy:
Contraindications and Precautions
- Absolute: Active pancreatitis or history of VIP-induced pancreatitis
- Absolute: Known hypersensitivity to VIP or formulation components
- Relative: Ongoing mold or biotoxin exposure (VIP should not be initiated until environment is remediated and preceding protocol steps completed)
- Relative: Elevated baseline lipase
- Relative: Symptomatic hypotension or concurrent high-dose antihypertensive therapy
- Relative: Active VIPoma or history of pancreatic neuroendocrine tumors
- Relative: Severe diarrheal illness
- Caution: Pregnancy and lactation (insufficient safety data)
- Caution: Severe renal impairment (may alter peptide clearance)
Drug Interactions
Formal drug interaction studies for intranasal VIP have not been conducted. Based on its pharmacology, potential interactions include:
- Antihypertensive medications: Additive blood pressure lowering. Monitor BP closely, especially during initiation.
- Phosphodiesterase inhibitors (e.g., sildenafil): VIP produces cAMP, and PDE inhibitors prevent cAMP breakdown. The combination could potentiate VIP effects. Use with caution.
- Insulin and hypoglycemic agents: VIP has modest effects on insulin secretion and glucose metabolism. Monitor glucose more closely in diabetic patients starting VIP.
- Corticosteroids: Both VIP and corticosteroids are anti-inflammatory. No direct antagonism is expected, but the combined immunosuppressive effect should be considered in patients on chronic corticosteroid therapy.
- Cholestyramine: If the patient is still on CSM from earlier in the Shoemaker Protocol, timing VIP administration at least 2 hours away from CSM doses prevents potential binding of absorbed VIP to the resin.
Long-Term Safety Considerations
The long-term safety of VIP is reassuring based on available data, but important caveats apply. Most clinical experience involves treatment courses of 2-6 months, the standard duration for CIRS treatment. Data on continuous long-term VIP use (years) is limited. Theoretical concerns about chronic exogenous VIP include potential receptor desensitization (downregulation of VPAC1/VPAC2 with sustained agonist exposure), effects on pancreatic function with prolonged stimulation, and alterations in gastric acid secretion (VIP inhibits acid secretion). These theoretical concerns have not been documented as clinical problems at the doses and durations used in CIRS treatment, but they underscore the wisdom of using VIP for defined treatment courses rather than indefinite chronic therapy.
Safety in Special Populations
Pediatric patients: VIP has been used in children with CIRS, though published pediatric dosing data is limited. Dose adjustments based on body weight may be appropriate, and closer monitoring is warranted given the lack of formal pediatric pharmacokinetic studies.
Elderly patients: Older adults may be more sensitive to VIP's hypotensive effects and should be started at lower doses with careful BP monitoring. The potential benefits of VIP for age-related neuroinflammation and cognitive decline are intriguing but remain investigational.
Pregnancy: VIP plays physiological roles in pregnancy, including placental function and maternal immune tolerance of the fetus. However, the safety of exogenous VIP in human pregnancy has not been established, and its use during pregnancy should be avoided unless the potential benefit clearly outweighs the risk.
VIP Metabolism, Pharmacokinetics, and Drug Development
Enzymatic Degradation and Half-Life Challenges
One of the most significant obstacles to developing VIP as a therapeutic agent has been its rapid enzymatic degradation in vivo. When administered intravenously, VIP has a plasma half-life of only 1-2 minutes, a duration too short to produce sustained therapeutic effects through conventional systemic delivery. This ultra-short half-life results from the combined action of several proteolytic enzymes that cleave VIP at specific sites along its 28-amino-acid chain.
The primary enzymes responsible for VIP degradation include neutral endopeptidase (NEP, also known as neprilysin or CD10), which cleaves VIP at multiple sites within the peptide chain, generating inactive fragments. Dipeptidyl peptidase IV (DPP-IV, also known as CD26) removes the N-terminal His-Ser dipeptide, producing VIP(3-28), which has markedly reduced biological activity. Mast cell tryptase, aminopeptidases in the lung and kidney, and serum proteases also contribute to VIP catabolism. The combined action of these enzymes ensures that any VIP entering the bloodstream is rapidly cleared, creating a fundamental pharmacokinetic challenge for systemic VIP therapy.
This rapid degradation is not simply a pharmaceutical inconvenience - it also has physiological significance. The short half-life of VIP ensures that its signaling remains tightly localized to the sites of neuronal release. VIP released from nerve terminals acts on nearby target cells within a limited diffusion radius before being degraded, creating the precise spatial control needed for functions like regulating individual segments of intestinal motility or modulating immune responses within specific tissue compartments. This paracrine signaling pattern means that circulating VIP levels may not fully reflect local tissue VIP activity, a consideration that complicates the interpretation of serum VIP measurements in clinical practice.
Strategies to Overcome Short Half-Life
Researchers have pursued multiple strategies to extend VIP's biological availability for therapeutic applications:
Intranasal Delivery
The intranasal route, now standard for CIRS treatment, partially addresses the half-life problem through several mechanisms. The nasal mucosa serves as a depot from which VIP is gradually absorbed, effectively extending the period of VIP input into the systemic circulation beyond what a single bolus injection would provide. Additionally, a fraction of intranasally administered VIP may bypass the systemic circulation entirely, reaching the brain via olfactory nerve transport. This nose-to-brain pathway could deliver VIP directly to CNS targets without requiring it to survive passage through the bloodstream, though the quantitative contribution of this pathway in humans remains debated.
VIP Analogs with Enhanced Stability
Chemical modification of VIP to resist enzymatic degradation has been an active area of medicinal chemistry. Approaches include substitution of specific amino acids at protease cleavage sites with non-natural or D-amino acids, PEGylation (attachment of polyethylene glycol chains to increase molecular size and reduce protease access), cyclization of the peptide backbone to restrict conformational flexibility needed for protease recognition, and development of retro-inverso peptide analogs that maintain receptor binding while resisting proteolysis. Several stabilized VIP analogs have shown extended half-lives in preclinical studies while retaining VPAC receptor binding and biological activity, but none have yet advanced to widespread clinical use.
Nanoparticle and Liposomal Formulations
Encapsulation of VIP within lipid nanoparticles, liposomes, or polymeric microspheres can protect the peptide from enzymatic degradation while providing controlled release over extended periods. Sterically stabilized micelles loaded with VIP have shown particular promise for oral delivery in IBD applications, protecting VIP from gastric acid and intestinal proteases while releasing the peptide at sites of intestinal inflammation. These advanced delivery systems are in preclinical and early clinical development and could eventually enable oral or long-acting injectable VIP therapy.
Gene Therapy Approaches
Rather than delivering the VIP peptide itself, gene therapy strategies aim to introduce the VIP gene into target tissues, enabling sustained local VIP production. Adeno-associated viral (AAV) vectors and lentiviral vectors carrying the VIP gene have been used in preclinical studies to transduce cells in arthritic joints, inflamed intestinal tissue, and brain regions affected by neurodegeneration. These gene therapy approaches produce sustained, localized VIP expression without requiring repeated peptide administration, but they raise the usual safety and regulatory concerns associated with gene therapy and remain far from clinical application.
Aviptadil: The Pharmaceutical VIP
Aviptadil is the International Nonproprietary Name (INN) for synthetic VIP used as a pharmaceutical agent. Chemically identical to endogenous human VIP, aviptadil has been formulated for intravenous, inhaled, and intranasal administration. Its development history reflects the broader challenges and opportunities of VIP therapeutics:
In 2003, aviptadil gained attention when the Austrian PAH studies demonstrated its hemodynamic benefits in pulmonary hypertension. Subsequent development efforts focused on inhaled formulations for pulmonary applications and continued assessment in PAH clinical trials with mixed results.
In 2020, aviptadil attracted renewed interest when it was investigated for COVID-19-associated acute respiratory distress syndrome (ARDS). The rationale was based on VIP's anti-inflammatory and lung-protective properties: in patients with severe COVID-19 pneumonia, VIP could potentially reduce the cytokine storm driving lung damage while supporting pulmonary surfactant production and vascular integrity. Clinical trials of intravenous aviptadil for COVID-19 ARDS were conducted, and while some showed signals of benefit (particularly in oxygenation parameters and survival), the evidence was not strong enough to secure FDA approval. The COVID-19 experience did, however, demonstrate the feasibility of intravenous VIP administration in critically ill patients and added to the safety database for systemic VIP delivery.
Measuring VIP: Laboratory Considerations
Accurate measurement of VIP levels is essential for diagnosing VIP deficiency in CIRS and for monitoring treatment response. However, VIP measurement presents several technical challenges that clinicians should understand:
Sample handling: VIP degrades rapidly at room temperature. Blood samples for VIP measurement must be collected in chilled EDTA tubes containing aprotinin (a protease inhibitor), immediately centrifuged at 4 degrees C, and frozen within 30 minutes of collection. Failure to follow these stringent collection requirements can produce falsely low VIP values, leading to incorrect interpretation.
Assay methodology: VIP is typically measured by radioimmunoassay (RIA) or enzyme-linked immunosorbent assay (ELISA). Different assays may use different antibodies with varying specificities for intact VIP versus degradation fragments, which can produce discrepant results between laboratories. Clinicians should ideally use the same laboratory and assay method for serial VIP measurements to ensure comparability.
Reference ranges: Normal reference ranges for serum VIP vary by laboratory but typically fall in the range of 23-63 pg/mL. CIRS patients often show values below 20 pg/mL, though the clinical significance of borderline values (20-25 pg/mL) depends on the overall clinical context and other biomarker findings.
Timing: VIP levels show some diurnal variation, reflecting circadian patterns of VIP release. For optimal consistency, blood samples for VIP should be drawn at the same time of day across serial measurements, ideally in the morning before the patient has exercised or eaten.
Advanced CIRS Considerations and VIP
Proteomics, Genomics, and the Future of CIRS Diagnostics
The field of CIRS research has evolved significantly from its early focus on individual biomarkers (C4a, TGF-beta 1, MSH, VIP) toward a more systems-level understanding of the disease. Modern "omics" technologies are providing new insights into how CIRS disrupts cellular function at the molecular level and how VIP therapy restores normal physiology.
Transcriptomic Profiling
Transcriptomic analysis - measuring the expression levels of thousands of genes simultaneously using microarray or RNA sequencing technology - has been one of Shoemaker's most significant contributions to CIRS research. By analyzing differential gene expression in peripheral blood mononuclear cells from CIRS patients, his team identified characteristic gene expression signatures that distinguish CIRS patients from healthy controls and from patients with other chronic inflammatory conditions.
These transcriptomic profiles reveal that CIRS involves dysregulation of gene networks spanning inflammation (complement cascade genes, cytokine genes, NF-kB pathway genes), coagulation (tissue factor, thrombin receptor genes), immune cell trafficking (chemokine genes, adhesion molecule genes), apoptosis (pro- and anti-apoptotic gene families), metabolic regulation (nuclear receptor genes, glucose metabolism genes), and hormonal signaling (cortisol receptor genes, thyroid hormone pathway genes).
The therapeutic power of VIP is most evident in its transcriptomic effects. Pre- and post-VIP transcriptomic analysis shows correction of multiple dysregulated gene clusters, with inflammatory and coagulation genes showing decreased expression, immune regulatory genes showing increased expression, and metabolic and hormonal gene networks normalizing. This comprehensive gene expression correction - affecting hundreds of genes simultaneously - helps explain why VIP produces such broad clinical improvement in CIRS patients. No single conventional drug produces this breadth of transcriptomic correction, suggesting that VIP is addressing a fundamental regulatory deficit rather than merely blocking one pathway.
Proteomic Considerations
While transcriptomic data reveals changes in gene expression, proteomics measures the actual protein products of those genes. Proteomic studies in CIRS are still emerging, but preliminary data suggests that the protein-level changes in CIRS patients correlate with the transcriptomic abnormalities. Complement proteins, cytokines, matrix metalloproteinases, and coagulation factors are among the proteins that show altered levels in CIRS plasma, and many of these normalize with treatment including VIP.
Advanced proteomic techniques such as mass spectrometry-based plasma proteomics and proximity extension assays are being applied to CIRS research, offering the potential for more comprehensive biomarker panels that could improve diagnostic accuracy and treatment monitoring. These technologies may eventually replace the current practice of measuring individual biomarkers one at a time with multiplex panels that provide a holistic view of disease activity.
The Exposome and CIRS Complexity
An increasingly recognized challenge in CIRS research and treatment is the complexity of the exposome - the totality of environmental exposures that contribute to disease. While water-damaged buildings are the primary trigger for most CIRS cases, patients are often exposed to multiple concurrent environmental stressors that can complicate diagnosis and treatment response. These include outdoor mold and mycotoxin exposure (especially in humid climates or near agricultural operations), environmental pollutants (volatile organic compounds, heavy metals, pesticides), electromagnetic fields (which some practitioners believe can activate mast cells and worsen CIRS), dietary antigens (particularly gluten and other lectins in genetically susceptible individuals), and chronic infections (tick-borne infections, chronic viral reactivation, gut dysbiosis).
The multi-exposure reality of CIRS patients means that VIP therapy may need to be integrated with additional interventions addressing specific exposure categories. For example, a patient with CIRS and concurrent Lyme disease may need both the Shoemaker Protocol (including VIP) for the mold/biotoxin component and antimicrobial therapy for the tick-borne infection. Similarly, patients with significant gut dysbiosis may benefit from microbiome restoration alongside VIP therapy, since VIP-deficient states are associated with altered gut microbial communities.
VIP and the Blood-Brain Barrier in CIRS
Blood-brain barrier (BBB) disruption is an underappreciated feature of CIRS that has direct implications for both disease pathogenesis and VIP therapy. The BBB normally excludes most circulating proteins, peptides, and immune cells from the brain parenchyma, protecting the CNS from peripheral inflammation. In CIRS, chronic systemic inflammation appears to compromise BBB integrity, allowing inflammatory mediators and immune cells to access the brain.
Several lines of evidence support BBB disruption in CIRS. First, the cognitive symptoms ("brain fog") experienced by most CIRS patients suggest neuroinflammation, which requires inflammatory mediators to reach the brain. Second, visual contrast sensitivity (VCS) abnormalities in CIRS are thought to reflect neurotoxic effects on the optic nerve and visual processing pathways, implying CNS penetration of circulating toxins or inflammatory mediators. Third, neuroimaging studies in some CIRS patients have shown brain structural changes consistent with neuroinflammation.
For VIP therapy, BBB disruption may actually be a double-edged sword. On one hand, a compromised BBB allows peripheral inflammatory mediators to enter the brain and cause neurological damage. On the other hand, it may also allow intranasally administered VIP to reach the brain in greater quantities than would be possible with an intact BBB, enhancing the neuroprotective effects of treatment. As VIP therapy reduces systemic inflammation and neuroinflammation improves, the BBB may begin to restore its normal integrity, potentially reducing both peripheral inflammatory CNS access and VIP CNS penetration simultaneously. This dynamic relationship between BBB integrity and VIP access to the brain is an area that deserves further investigation.
Re-Exposure and Relapse After VIP Therapy
One of the most common clinical challenges in CIRS management is relapse following successful treatment, and understanding the role of re-exposure is critical for long-term outcomes. Shoemaker's data indicate that the most frequent cause of relapse after VIP therapy is re-exposure to water-damaged buildings - either return to an inadequately remediated prior environment or exposure to a new moldy environment.
The question of whether VIP therapy provides lasting protection against re-exposure has important practical implications. Based on available data, VIP appears to produce durable gene expression changes that persist for months to years after treatment discontinuation, but these changes do not make the patient immune to future biotoxin exposure. A patient with susceptible HLA-DR genotypes who is re-exposed to a significant mold burden will likely develop recurrent CIRS, potentially requiring retreatment including repeat VIP therapy.
Several strategies can reduce the risk of relapse:
- Environmental vigilance: Regular ERMI or HERTSMI-2 testing of home and workplace environments to detect water damage before it produces clinical illness
- Rapid intervention: If re-exposure is identified early (through symptom recurrence or rising biomarkers), prompt cholestyramine binding and environmental remediation may prevent the full CIRS cascade from re-establishing
- Periodic monitoring: Annual or biannual CIRS biomarker panels to detect subclinical inflammation before it produces symptoms
- VIP retreatment: Short courses of VIP nasal spray can be used to treat early relapse, typically requiring less extensive retreatment than the initial course
- Immune resilience building: Supporting overall immune competence through adequate nutrition, sleep, stress management, exercise, and appropriate supplementation may reduce vulnerability to re-exposure
Figure 8: The CIRS treatment timeline from diagnosis through VIP therapy and long-term maintenance. Most patients achieve durable remission with one VIP course, while a subset requires extended treatment or periodic retreatment for relapse management.
Emerging Applications and Future Directions
VIP and COVID-19: Lessons from a Pandemic
The COVID-19 pandemic brought unexpected attention to VIP when researchers recognized that the peptide's pharmacological profile aligned well with the pathophysiology of severe COVID-19 pneumonia. The severe acute respiratory syndrome caused by SARS-CoV-2 involves a "cytokine storm" - an overwhelming systemic inflammatory response driven by excessive production of IL-6, TNF-alpha, IL-1beta, and other pro-inflammatory mediators. This cytokine storm causes acute lung injury, surfactant dysfunction, pulmonary edema, and often fatal respiratory failure.
VIP's potent anti-inflammatory properties, combined with its specific actions on lung tissue (surfactant production support, pulmonary vasodilation, airway smooth muscle relaxation, and anti-fibrotic effects), made it a rational candidate for COVID-19 ARDS treatment. Clinical trials of intravenous aviptadil in COVID-19 patients were initiated at multiple centers, with some studies reporting improvements in oxygenation parameters, reduced ICU length of stay, and survival benefit in treated patients compared to controls.
While aviptadil did not ultimately receive FDA approval for COVID-19, the pandemic experience validated several important principles about VIP therapeutics. First, it demonstrated that intravenous VIP could be safely administered to critically ill patients at doses sufficient to produce measurable anti-inflammatory effects. Second, it confirmed VIP's ability to suppress cytokine storm-like inflammatory cascades in a human clinical setting. Third, it generated new pharmacokinetic and safety data that extends the evidence base beyond the CIRS population. These lessons may inform future VIP development for other acute inflammatory conditions.
Systemic Exertional Intolerance Disease (SEID) and ME/CFS
Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS), also known as systemic exertional intolerance disease (SEID), shares numerous clinical features with CIRS, including persistent fatigue, post-exertional malaise, cognitive impairment, sleep disruption, and multi-system symptom involvement. Shoemaker has specifically advocated for VIP use in SEID, arguing that many SEID patients may have undiagnosed or inadequately treated CIRS as an underlying etiology.
Published on the SurvivingMold.com platform, Shoemaker has presented data supporting VIP use in SEID patients who meet CIRS diagnostic criteria and have completed the preceding protocol steps. The rationale is that SEID patients with biotoxin exposure histories, susceptible HLA-DR genotypes, and characteristic biomarker abnormalities are essentially CIRS patients who have been misclassified under the SEID/ME/CFS diagnostic umbrella. In these patients, VIP therapy produces the same pattern of biomarker improvement and symptom resolution seen in properly diagnosed CIRS patients.
Beyond the CIRS-overlap population, VIP may have relevance for ME/CFS through its effects on neuroinflammation, autonomic nervous system function, and circadian regulation - all of which are disrupted in ME/CFS. The VIP-VPAC2 circadian signaling axis in the suprachiasmatic nucleus may be particularly relevant, given that ME/CFS patients frequently exhibit disrupted circadian rhythms, abnormal cortisol patterns, and non-restorative sleep. However, controlled clinical trials of VIP in ME/CFS patients without documented CIRS have not been conducted, and the evidence base remains limited to case series and clinical experience.
Organ Transplantation and Graft Tolerance
VIP's ability to promote regulatory T cells and tolerogenic dendritic cells has attracted attention in transplant immunology, where the central challenge is achieving immune tolerance to donor organs while maintaining immune competence against infections. Preclinical studies have shown that VIP treatment prolongs graft survival in mouse models of heart transplant, skin transplant, and islet transplant. The mechanism involves VIP-mediated induction of tolerogenic DCs that present donor antigens in a way that promotes Treg differentiation rather than effector T cell activation.
In the islet transplant model (relevant to type 1 diabetes), VIP treatment extended islet graft survival and maintained functional insulin production for significantly longer than control animals. This finding has potential implications for improving outcomes in clinical islet transplantation, a procedure that currently requires lifelong immunosuppressive therapy to prevent graft rejection.
While clinical translation of VIP for transplant tolerance is still in early stages, the concept of using a naturally occurring immune-regulatory peptide instead of (or in combination with) conventional immunosuppressive drugs is appealing. Conventional immunosuppressants like tacrolimus and mycophenolate carry significant toxicity risks including increased infection susceptibility, metabolic effects, and increased cancer risk. VIP-based immune tolerance strategies could potentially reduce the immunosuppressive burden while maintaining graft acceptance.
Cancer Immunology: A Complex Relationship
VIP's relationship with cancer biology is complex and, in some respects, paradoxical. On one hand, VIP's anti-inflammatory and immune-suppressive properties could theoretically benefit tumors by reducing the anti-tumor immune response. Indeed, some studies have shown that certain cancers express VPAC receptors and may use VIP signaling to evade immune surveillance. VIPoma tumors, which produce excessive VIP, are themselves a cancer type (typically pancreatic neuroendocrine tumors).
On the other hand, VIP's promotion of Treg cells and its effects on immune cell trafficking may have context-dependent effects on tumor biology. In some experimental systems, VIP-induced immune modulation has been beneficial against certain tumor types, particularly those driven by chronic inflammation. The anti-inflammatory resolution of tumor-promoting inflammation may, in specific contexts, actually enhance anti-tumor immunity by resolving the immunosuppressive tumor microenvironment.
Given this complexity, the use of VIP in cancer patients requires careful consideration. In patients with active malignancy, VIP therapy should be approached cautiously, with thorough discussion between the patient, their oncologist, and the prescribing clinician about potential risks and benefits. In cancer survivors, VIP's anti-inflammatory properties may be beneficial for reducing cancer-related inflammation and supporting immune reconstitution, but again, individual assessment is warranted.
Fertility and Reproductive Medicine
VIP plays physiological roles in reproductive function that are beginning to attract clinical interest. In female reproduction, VIP is involved in ovarian function (follicular development, steroidogenesis), uterine contractility (VIP inhibits uterine smooth muscle contraction, potentially playing a role in implantation and pregnancy maintenance), and placental function (VIP is produced by the placenta and may contribute to maternal immune tolerance of the fetus).
Research has shown that VIP promotes regulatory T cell induction by trophoblast cells, supporting the immune tolerance that is essential for successful pregnancy. Trophoblast-derived VIP signals through VPAC2 on maternal immune cells, increasing the frequency of CD4+CD25+FoxP3+ Tregs and suppressing the maternal alloimmune response against fetal tissues. This finding suggests that VIP deficiency could contribute to pregnancy complications characterized by insufficient maternal immune tolerance, including recurrent pregnancy loss, preeclampsia, and intrauterine growth restriction.
In male reproduction, VIP is found in nerve fibers innervating the vas deferens, prostate, and penile erectile tissue. VIP is one of the key neurotransmitters mediating penile erection through its vasodilatory effects on cavernosal smooth muscle. Intracavernosal VIP injection has been used clinically for erectile dysfunction, though it has been largely superseded by phosphodiesterase inhibitors (sildenafil and related drugs) that are easier to administer.
Future Research Priorities
Based on the current state of VIP research, several priorities for future investigation can be identified:
- Randomized controlled trials in CIRS: While Shoemaker's published data provides compelling evidence, larger randomized, placebo-controlled trials with independent replication would strengthen the evidence base for VIP in CIRS and potentially support regulatory approval
- Biomarker-guided patient selection: Developing predictive biomarkers that identify which patients are most likely to respond to VIP therapy would improve treatment efficiency and reduce unnecessary exposure in non-responders
- Receptor-selective VIP analogs: Creating VPAC1-selective and VPAC2-selective agonists would allow more precise targeting of specific immune or neurological effects without activating both receptor pathways simultaneously
- Long-acting VIP formulations: Developing extended-release VIP preparations (depot injections, implantable devices, or oral nanoformulations) would simplify dosing regimens and improve patient compliance
- Combination therapy protocols: Systematically evaluating VIP in combination with other immune-modulating peptides (Thymosin Alpha-1, KPV, BPC-157) to identify optimal multi-peptide regimens for complex immune conditions
- Pediatric studies: Establishing age-appropriate dosing guidelines and safety data for VIP use in children with CIRS
- Neuroimaging correlation: Using functional MRI, PET, or other neuroimaging modalities to correlate VIP therapy with objective measures of neuroinflammation resolution and cognitive network function
Figure 9: Future directions in VIP research. Current clinical applications in CIRS and pulmonary disease provide the foundation for expanded investigations into novel formulations, receptor-selective compounds, combination therapies, and emerging clinical indications.
Clinical Considerations and Patient Selection
Identifying Appropriate Candidates for VIP Therapy
Not every patient with chronic inflammation or immune dysfunction is an appropriate candidate for VIP therapy. The strongest evidence supports VIP use in specific clinical contexts, and clinicians should consider several factors when evaluating whether VIP is indicated for a particular patient.
Ideal VIP Candidates
The best-supported indication for VIP nasal spray remains CIRS in the context of the Shoemaker Protocol. Ideal candidates for VIP in this setting share the following characteristics:
- Confirmed CIRS diagnosis based on history of biotoxin exposure, susceptible HLA-DR genotype, characteristic symptom cluster (minimum 8 of 13 symptom clusters), and supporting biomarker panel
- Completion of preceding Shoemaker Protocol steps, with documentation that environmental remediation, toxin binding, MARCoNS treatment, and earlier biomarker corrections have been performed
- Persistently abnormal C4a and/or TGF-beta 1 despite preceding interventions, indicating residual immune dysregulation that VIP specifically targets
- No contraindications to VIP (active pancreatitis, known hypersensitivity, ongoing mold exposure)
- Normal baseline fasting lipase
- Willingness and ability to comply with the monitoring schedule (blood draws at baseline, 30 days, end of treatment, and 6-month follow-up)
Patients Who May Benefit from VIP Outside the CIRS Context
While evidence is less extensive outside CIRS, several other patient populations may be considered for VIP therapy based on the available research:
- Post-COVID long-haulers with CIRS-like presentations: Patients who developed chronic inflammatory symptoms after COVID-19 infection, particularly those with elevated inflammatory markers and VIP deficiency
- Refractory autoimmune conditions: Patients with rheumatoid arthritis, sarcoidosis, or other autoimmune conditions who have not responded adequately to conventional therapies, under specialist supervision
- Pulmonary hypertension: Patients with PAH and documented VIP deficiency, particularly those with CIRS-associated PAH
- Chronic neuroinflammatory states: Patients with persistent cognitive impairment linked to documented neuroinflammation, after other treatable causes have been excluded
Patients Unlikely to Benefit from VIP
Conversely, VIP is unlikely to help (and may waste time and money) in the following situations:
- Patients with fatigue or cognitive symptoms unrelated to inflammatory pathophysiology
- Patients with normal VIP levels and normal inflammatory biomarkers
- CIRS patients who have not completed preceding protocol steps (VIP will likely fail or produce paradoxical worsening)
- Patients with ongoing mold exposure who have not remediated their environment
- Patients seeking VIP as a "wellness" supplement without specific medical indication
Working with Clinicians Experienced in VIP Therapy
VIP therapy for CIRS requires specialized knowledge that goes beyond standard medical training. The Shoemaker Protocol is complex, requiring familiarity with environmental medicine, biotoxin illness, HLA genetics, and a specific panel of inflammatory biomarkers that most physicians do not routinely order. Patients considering VIP therapy should seek clinicians who have specific training in the Shoemaker Protocol or equivalent biotoxin illness treatment approaches.
Before seeking a clinician, patients can complete the Free Assessment to evaluate their situation. Key questions to ask a potential prescribing clinician include whether they are familiar with the Shoemaker Protocol and have experience implementing it, whether they perform the VIP challenge test before initiating therapy, what monitoring schedule they follow during VIP treatment, whether they have access to the full CIRS biomarker panel (including C4a, TGF-beta 1, MMP-9, VEGF, MSH, VIP, ADH, osmolality), and which compounding pharmacy they use for VIP nasal spray (ensuring quality and potency testing).
FormBlends offers a Free Assessment that can help patients determine whether their symptoms align with conditions where VIP may be beneficial, and the VIP product page provides additional clinical information. For a broader understanding of how VIP fits within the peptide therapy landscape, the Peptide Hub offers educational resources on immune-modulating peptides, and the LL-37 research report covers complementary antimicrobial peptide approaches that may be used alongside VIP in comprehensive treatment protocols.
Figure 10: Patient selection flowchart for VIP therapy. Appropriate patient identification requires confirmed CIRS diagnosis, completion of preceding protocol steps, documentation of persistent biomarker abnormalities, absence of contraindications, and commitment to the monitoring schedule.
VIP in Clinical Practice: Detailed Treatment Considerations
Pre-Treatment Laboratory Workup
Before initiating VIP therapy, a comprehensive laboratory evaluation establishes the baseline against which treatment response will be measured. This workup serves multiple purposes: confirming the CIRS diagnosis, documenting the severity of immune dysregulation, identifying potential contraindications, and establishing reference values for monitoring. The complete pre-VIP laboratory panel should include the following markers, each with specific interpretive significance.
Complement Split Products
C4a is the single most important inflammatory biomarker in the VIP treatment context. This complement split product reflects activation of the mannose-binding lectin pathway of complement, which is the primary complement pathway activated by biotoxin exposure. Normal C4a levels are below 2,830 ng/mL (Quest Diagnostics reference range), though values above 20,000 ng/mL are not uncommon in actively symptomatic CIRS patients. Markedly elevated C4a levels that have persisted despite cholestyramine treatment and preceding protocol steps provide the strongest indication for VIP therapy, as VIP specifically targets the innate immune activation that drives C4a elevation.
C3a, another complement split product, reflects activation of the classical and alternative complement pathways. While C4a is more specific to biotoxin-driven complement activation, C3a provides complementary information about the overall state of complement system activation. Some clinicians use the C4a/C3a ratio to assess the relative contribution of different complement pathways to the patient's inflammatory burden. Normal C3a levels are below 940 ng/mL. Unlike C4a, which often requires VIP for full normalization, C3a frequently improves with earlier protocol steps including statin therapy.
Transforming Growth Factor Beta-1
TGF-beta 1 is a pleiotropic cytokine with particularly complex biology in CIRS. At normal levels (below 2,380 pg/mL), TGF-beta 1 contributes to immune regulation, wound healing, and tissue homeostasis. In CIRS, TGF-beta 1 is often markedly elevated (sometimes exceeding 10,000 pg/mL), driving pathological effects including Th17 polarization (promoting autoimmune-type inflammation), tissue fibrosis, and immune cell trafficking to sites of inflammation. Chronically elevated TGF-beta 1 in CIRS is associated with autoantibody formation, restrictive lung disease, and skeletal muscle wasting.
VIP's suppression of TGF-beta 1 is one of its most clinically significant effects. Some CIRS patients have TGF-beta 1 levels that remain stubbornly elevated despite losartan therapy (the standard TGF-beta 1 lowering agent in the Shoemaker Protocol), and VIP may be the intervention that finally brings this marker under control. Serial TGF-beta 1 measurement during VIP therapy provides direct feedback on treatment efficacy and guides decisions about dose adjustment and treatment duration.
Matrix Metalloproteinase-9
MMP-9 is a zinc-dependent endopeptidase involved in extracellular matrix remodeling, inflammatory cell migration, and tissue destruction. In CIRS, MMP-9 levels are elevated (normal <332 ng/mL), reflecting ongoing tissue inflammation and matrix degradation. Elevated MMP-9 contributes to blood-brain barrier disruption (MMP-9 degrades tight junction proteins and basement membrane components), vascular inflammation, and tissue damage in multiple organ systems.
While MMP-9 often responds to earlier protocol steps (particularly high-dose omega-3 fatty acid supplementation), persistent MMP-9 elevation may improve further with VIP therapy. VIP's suppression of NF-kB transcriptional activity reduces MMP-9 gene expression in macrophages and other inflammatory cells, contributing to the overall decrease in matrix-degrading activity that accompanies treatment.
Vascular Endothelial Growth Factor
VEGF has a U-shaped relationship with health in CIRS - both abnormally low and abnormally high values are pathological. Low VEGF (below 31 pg/mL) indicates impaired angiogenesis and tissue perfusion, contributing to the tissue hypoxia, fatigue, and exercise intolerance experienced by many CIRS patients. High VEGF (above 86 pg/mL) may represent a compensatory response to tissue hypoxia or, in some cases, may be driven by inflammatory mediators that stimulate VEGF production independently of tissue oxygen demand.
VIP's effects on VEGF tend to be normalizing rather than unidirectionally suppressive. In patients with low VEGF, VIP therapy often produces an increase toward normal ranges, potentially reflecting improved tissue oxygenation and reduced inflammatory suppression of VEGF-producing cells. In patients with elevated VEGF, the improvement in tissue perfusion and reduced inflammatory drive can normalize VEGF downward. This bidirectional normalizing effect is consistent with VIP's role as a physiological regulator rather than a pharmacological agent that pushes parameters in a single direction.
Additional Biomarkers
A complete pre-VIP workup should also include:
- Alpha-MSH: Usually suppressed in CIRS (normal 35-81 pg/mL); may or may not improve with VIP. Persistent MSH suppression after VIP may indicate residual inflammatory drive from an unaddressed source.
- Serum VIP: Documents the VIP-deficient state that provides rationale for replacement therapy. Expected to be low (normal 23-63 pg/mL).
- ADH and serum osmolality: Assess fluid regulation, which is often dysregulated in CIRS and should have been addressed in preceding protocol steps.
- Leptin: Often elevated in CIRS, reflecting leptin resistance that contributes to weight gain and metabolic dysfunction.
- Fasting lipase: Essential baseline for monitoring pancreatic safety during VIP therapy. Must be within normal range before starting VIP.
- Comprehensive metabolic panel: Liver function, kidney function, and electrolytes provide overall health context and identify potential complicating conditions.
- Visual contrast sensitivity (VCS): A functional test that correlates with neurotoxin exposure and neuroinflammation. Serial VCS testing during VIP therapy provides a non-invasive marker of neurological improvement.
Interpreting Treatment Response
The expected trajectory of treatment response during VIP therapy follows a characteristic pattern that clinicians should recognize. In the first 1-2 weeks, many patients experience a "settling in" period where symptoms may fluctuate. Some patients notice improvement almost immediately, while others experience a transient worsening of symptoms (sometimes called a "herx" reaction, though this term is more properly applied to antimicrobial die-off reactions) before improvement begins. The mechanism of early symptom fluctuation is not fully understood but may relate to the immune system's adjustment to a new regulatory set point.
By weeks 2-4, most responding patients begin to notice consistent symptom improvement. Fatigue typically improves first, followed by cognitive function, sleep quality, and pain symptoms. The degree of improvement varies widely among patients, with some experiencing dramatic, life-changing improvement and others noting more subtle but still meaningful gains. Patients who have been ill for shorter periods before diagnosis tend to respond more completely and more quickly than those with long-standing illness.
The 30-day laboratory reassessment provides the first objective confirmation of treatment response. Expected findings include declining C4a (often with the most dramatic decrease seen in the first month), improving TGF-beta 1, stable or improving MMP-9, normalizing VEGF, and - critically - normal fasting lipase (confirming pancreatic safety). If C4a has not decreased or has increased, the clinician should consider whether ongoing mold exposure is present, whether the VIP formulation is potent and properly stored, whether the patient is adhering to the dosing schedule, and whether the patient may have been started on VIP prematurely (with preceding protocol steps incompletely addressed).
Handling Incomplete Responses
Not all CIRS patients achieve complete biomarker normalization and symptom resolution with VIP therapy, even after a full treatment course. In these cases, the clinician should systematically evaluate potential reasons for incomplete response:
Environmental factors: The single most common cause of incomplete VIP response is ongoing or intermittent biotoxin exposure. This may not be from the patient's home (which has presumably been remediated) but from their workplace, vehicle, gym, church, school, or other frequently visited environments. ERMI testing of these locations may reveal unsuspected mold exposure.
Concurrent infections: Tick-borne infections (Borrelia, Babesia, Bartonella, Ehrlichia), chronic viral reactivation (EBV, HHV-6, CMV), and small intestinal bacterial overgrowth (SIBO) can produce overlapping inflammation that VIP alone cannot fully address. Comprehensive infectious disease evaluation may be warranted in patients with incomplete VIP response.
MARCoNS recurrence: Multiply antibiotic-resistant coagulase-negative staphylococci in the nasal passages can interfere with VIP delivery and produce ongoing inflammatory stimulation. Repeat nasal culture should be considered in non-responders, especially if VIP is being delivered intranasally to the same nasal passages where MARCoNS may be colonizing.
Dietary and lifestyle factors: Ongoing dietary triggers (particularly gluten in patients with positive antigliadin antibodies), sleep deprivation, chronic psychological stress, and physical inactivity can each sustain inflammation independently of biotoxin exposure.
Genetic factors: Patients with certain HLA-DR genotypes (particularly "multisusceptible" genotypes) may have more deeply ingrained immune dysregulation that requires longer or more intensive treatment. These patients may benefit from extended VIP courses or combination peptide therapy.
VIP and Mast Cell Activation Syndrome
An increasingly recognized overlap exists between CIRS and mast cell activation syndrome (MCAS). Mast cells are immune cells that release histamine, prostaglandins, leukotrienes, and cytokines in response to various triggers, and MCAS involves inappropriate activation of these cells producing symptoms that can mimic or accompany CIRS. Many CIRS patients meet diagnostic criteria for concurrent MCAS, and the two conditions may share common triggers and pathophysiological mechanisms.
VIP's relationship with mast cells is bidirectional. Mast cells express VPAC receptors and release VIP from their granules, while VIP can modulate mast cell degranulation through both stimulatory and inhibitory pathways depending on the context. In CIRS patients with concurrent MCAS, VIP therapy may help stabilize mast cells through its anti-inflammatory effects on the surrounding tissue environment, reducing the inflammatory signals that trigger mast cell activation. However, some MCAS patients are highly sensitive to any new substance, including peptides, and may need to initiate VIP at lower doses with careful monitoring for mast cell activation reactions.
Clinicians treating CIRS patients with concurrent MCAS often use mast cell stabilizers (cromolyn sodium, ketotifen, quercetin) before and during VIP therapy to minimize the risk of mast cell-mediated adverse reactions. This combination approach addresses both the upstream immune dysregulation (via VIP) and the downstream mast cell hyperreactivity (via mast cell stabilizers) that contribute to the patient's symptom burden.
Integration with Functional Medicine Approaches
VIP therapy for CIRS is frequently practiced within a functional medicine framework that addresses root causes and systemic imbalances rather than simply targeting individual symptoms. This integrative approach recognizes that CIRS patients typically have multiple concurrent health issues that can influence VIP treatment outcomes.
Gut health optimization is a common adjunctive focus. Since VIP deficiency is associated with altered gut microbiota and increased intestinal permeability, supporting gut health through probiotics, prebiotics, dietary modification, and peptides like BPC-157 (for mucosal healing) and Larazotide (for tight junction support) may enhance VIP therapy outcomes. Some practitioners use the combination of VIP (systemic anti-inflammatory effects) with KPV (localized mucosal anti-inflammatory effects) to address both systemic and gut-specific inflammation simultaneously.
Mitochondrial support is another common focus, since chronic inflammation impairs mitochondrial function, contributing to the profound fatigue experienced by CIRS patients. NAD+ supplementation to support mitochondrial electron transport chain function may complement VIP's anti-inflammatory effects by addressing the cellular energy deficit that persists even after inflammation is controlled. Coenzyme Q10, alpha-lipoic acid, and B vitamins are additional mitochondrial support strategies frequently used alongside VIP therapy.
Adrenal and hormonal optimization addresses the endocrine disruptions common in CIRS. VIP's effects on the SCN circadian clock may help normalize cortisol rhythms, but some patients require additional adrenal support through adaptogenic herbs, low-dose cortisol replacement, or DHEA supplementation. Thyroid function should be monitored and optimized, as thyroid hormone production can be impaired by chronic inflammation and by the autoimmune processes that sometimes develop in CIRS.
Detoxification support continues to be important during and after VIP therapy. While the formal cholestyramine binding phase occurs earlier in the Shoemaker Protocol, ongoing low-level biotoxin clearance may benefit from supportive measures including adequate hydration, regular exercise (within the patient's tolerance), sauna therapy for mobilization of lipophilic toxins, and specific binding agents such as activated charcoal or bentonite clay. These detoxification strategies complement VIP's immune-resetting effects by reducing the ongoing toxic burden that the immune system must manage.
Cost Considerations and Access
VIP nasal spray is a compounded medication not covered by most insurance plans. The cost can be a significant barrier for patients, particularly given that the Shoemaker Protocol involves a lengthy sequence of tests, treatments, and office visits before VIP is even initiated. Costs for compounded VIP nasal spray typically range from $100-300 per month, depending on the pharmacy, formulation, and dosing schedule. Laboratory monitoring adds additional cost, with a full CIRS biomarker panel running several hundred dollars per draw.
Patients should factor in the total cost of the VIP treatment phase, including the VIP medication itself (typically 2-4 months of supply), the VIP challenge test (office visit plus stat lab processing), monitoring labs at 30 days and end of treatment, the follow-up labs at 6 months post-discontinuation, and potential additional costs for retreatment if relapse occurs. While these costs are significant, they should be weighed against the ongoing costs of poorly treated CIRS, including lost productivity, disability, repeated doctor visits, and the use of multiple symptomatic medications that treat individual CIRS symptoms without addressing the underlying cause.
Some compounding pharmacies offer VIP at reduced cost through prescription discount programs or patient assistance programs. Patients should inquire about these options and also verify that their pharmacy performs potency testing on VIP formulations, as cost savings should not come at the expense of product quality.
Synthesis and Clinical Perspective
VIP's Unique Therapeutic Position
After reviewing the extensive body of research on vasoactive intestinal peptide, several themes emerge that define VIP's unique position in the therapeutic landscape. First, VIP is one of the few therapeutic agents that addresses inflammation through immune recalibration rather than immune suppression. Unlike corticosteroids, calcineurin inhibitors, or JAK inhibitors that broadly dampen immune function (increasing infection risk and sometimes cancer risk), VIP shifts the immune system's operating point from inflammatory to tolerogenic while preserving - and in some ways enhancing - the ability to respond to genuine threats. This recalibrating effect, mediated through coordinated changes across macrophages, dendritic cells, and T cell subsets, is qualitatively different from the blunt immunosuppression provided by most anti-inflammatory drugs.
Second, VIP's pleiotropic nature - its ability to simultaneously affect immune regulation, pulmonary physiology, GI function, neuroprotection, and circadian rhythms - makes it uniquely suited for multi-system conditions like CIRS that involve dysfunction across multiple organ systems. No other single agent addresses as many of the pathological pathways active in CIRS as VIP does. This breadth of action is reflected in the comprehensive transcriptomic changes that VIP produces, correcting hundreds of dysregulated genes across multiple functional categories simultaneously.
Third, VIP operates as a replacement therapy rather than a pharmacological intervention. In CIRS patients with documented VIP deficiency, nasal VIP spray restores a naturally occurring regulatory peptide to levels that support normal immune and neurological function. This replacement paradigm may explain VIP's favorable safety profile: unlike drugs that introduce foreign chemical entities, VIP simply replenishes a molecule that the patient's body was designed to produce but can no longer maintain at adequate levels due to chronic inflammatory stress.
Limitations and Unanswered Questions
Despite the impressive body of research supporting VIP's therapeutic potential, several important limitations and unanswered questions remain. The clinical evidence base for VIP in CIRS, while compelling, consists primarily of the work of a single research group (Shoemaker and colleagues). Independent replication of the Shoemaker Protocol's VIP results by other research teams would substantially strengthen the evidence. The lack of large, multicenter, randomized, placebo-controlled trials means that VIP's evidence base does not meet the standards typically required for FDA approval, and some mainstream physicians remain skeptical of claims about CIRS and its treatment protocol.
The complexity of the Shoemaker Protocol itself creates challenges for research and clinical implementation. The sequential, multi-step design makes it difficult to isolate VIP's specific contribution from the cumulative effects of all preceding interventions. A patient who improves on VIP has also recently completed cholestyramine binding, MARCoNS treatment, hormonal correction, and multiple biomarker-targeted interventions. Determining how much of the final improvement is attributable to VIP versus the lasting effects of prior steps is methodologically challenging.
Questions also remain about optimal dosing, treatment duration, and the long-term durability of VIP's effects. The current dosing protocol is based on clinical experience and Shoemaker's studies but has not been subjected to formal dose-ranging studies or pharmacokinetic optimization. Some patients may require higher or lower doses, longer or shorter treatment courses, or different administration schedules than the current standard protocol specifies. Individual variation in nasal absorption, VIP receptor expression, and enzymatic degradation rates could all influence optimal dosing for a given patient.
Finally, the regulatory status of VIP as a compounded medication raises quality assurance concerns. Without FDA oversight of manufacturing processes, the potency, purity, and stability of compounded VIP nasal spray can vary between pharmacies. Patients and clinicians should work with compounding pharmacies that follow USP 797 and USP 800 standards, perform potency testing on their formulations, and can provide certificates of analysis confirming that their VIP products meet specifications.
The Broader Significance of VIP Research
Beyond its specific clinical applications, VIP research has contributed fundamental insights to immunology, neuroscience, and chronobiology. The discovery that a single endogenous neuropeptide can coordinate immune regulation, neuronal protection, circadian timing, and tissue homeostasis across multiple organ systems has profound implications for how we understand the integration of the nervous and immune systems. VIP's story illustrates the concept of neuroimmune communication - the bidirectional signaling between the nervous system and the immune system that maintains health and, when disrupted, contributes to disease.
The VIP-VPAC signaling axis represents one of the best-characterized examples of neuroimmune regulation, providing a molecular framework for understanding how stress, circadian disruption, environmental exposures, and neurological conditions can produce immune consequences, and vice versa. As the field of psychoneuroimmunology continues to mature, VIP and its receptors will likely remain central to our understanding of how the body's regulatory systems are integrated and how they can be therapeutically modulated.
For patients navigating complex chronic inflammatory conditions, VIP represents both a specific therapeutic tool and a broader lesson: that the body possesses endogenous regulatory systems capable of restoring balance when properly supported. The challenge for medicine is to identify when these regulatory systems have failed, understand why they have failed, and develop strategies to restore their function. VIP therapy for CIRS exemplifies this approach - identifying a specific regulatory peptide deficiency, understanding its contribution to disease pathology, and replacing it to restore physiological balance. As peptide therapeutics continue to develop, this paradigm of identifying and correcting endogenous regulatory deficits may expand to encompass an increasingly wide range of chronic conditions.
VIP in Specific Clinical Conditions: Detailed Evidence Review
VIP and Chronic Sinusitis in CIRS
Chronic sinusitis is one of the most prevalent symptoms in CIRS patients, affecting an estimated 50-70% of individuals with biotoxin illness. The relationship between VIP and sinus health is complex and clinically relevant, particularly given that VIP nasal spray is delivered directly into the nasal passages where chronic sinusitis pathology resides.
In healthy sinuses, VIP-containing nerve fibers innervate the submucosal glands, blood vessels, and epithelium of the nasal and paranasal sinus mucosa. VIP serves as a vasodilator (regulating mucosal blood flow), a secretagogue (stimulating serous secretion that contributes to the protective mucus blanket), and an anti-inflammatory agent (suppressing local immune cell activation). When VIP levels are deficient, as in CIRS, each of these functions is compromised: mucosal blood flow is reduced, secretory function is impaired, and local inflammation persists, creating an environment that favors chronic bacterial colonization and biofilm formation.
The relationship between VIP deficiency and MARCoNS colonization deserves special attention. MARCoNS (multiply antibiotic-resistant coagulase-negative staphylococci) are bacteria that form biofilms in the nasal passages of CIRS patients. These biofilms are notoriously difficult to eradicate and can persist despite topical antibiotic therapy. The MARCoNS biofilm produces exotoxins that further suppress MSH production and contribute to the inflammatory burden in CIRS. VIP deficiency in the nasal mucosa may create conditions that favor MARCoNS biofilm formation by reducing mucosal immune surveillance and impairing secretory clearance of bacteria.
When VIP nasal spray is administered to the nasal passages, it acts locally on the sinus mucosa in addition to its systemic effects (absorbed through the mucosal vasculature). This local action may help restore mucosal blood flow, improve secretory function, reduce local inflammation, and enhance the mucosal immune environment in ways that make it less hospitable to pathogenic bacterial colonization. Some clinicians have observed improvement in chronic sinus symptoms during VIP therapy that appears disproportionate to the systemic inflammatory improvement, suggesting a meaningful local effect of intranasal VIP delivery.
However, an important clinical caveat applies: if MARCoNS are still present in the nasal passages when VIP is initiated, the nasal spray could potentially spread the bacterial biofilm or create a paradoxical inflammatory response as VIP enhances local immune recognition of the MARCoNS organisms. This is one reason the Shoemaker Protocol requires MARCoNS eradication (typically with BEG spray - bacitracin, EDTA, gentamicin) before VIP therapy begins. Attempting to deliver VIP through MARCoNS-colonized nasal passages is both less effective (impaired mucosal absorption) and potentially counterproductive (enhanced recognition of persistent colonizers).
VIP and Autonomic Nervous System Dysfunction
Autonomic nervous system (ANS) dysfunction, often manifesting as postural orthostatic tachycardia syndrome (POTS), orthostatic hypotension, temperature dysregulation, and abnormal sweating, is common in CIRS patients and in other conditions where VIP therapy may be relevant. VIP's role in autonomic regulation provides a mechanistic framework for understanding and potentially treating these symptoms.
VIP is a co-transmitter in parasympathetic neurons throughout the body. In the cardiovascular system, parasympathetic VIP release contributes to vasodilation, heart rate regulation, and blood pressure control. VIP also modulates sympathetic nervous system activity indirectly through its effects on central autonomic control centers in the brainstem and hypothalamus. When VIP levels are deficient, the balance between sympathetic and parasympathetic tone may shift toward sympathetic predominance, contributing to the tachycardia, vasoconstriction, anxiety, and hyperarousal that many CIRS patients experience.
In the thermoregulatory system, VIP-containing neurons innervate sweat glands and cutaneous blood vessels, participating in the body's temperature control mechanisms. CIRS patients frequently report temperature dysregulation, including inability to maintain normal body temperature, night sweats, and abnormal sensitivity to heat or cold. VIP deficiency in the thermoregulatory neural circuitry may contribute to these symptoms, and VIP replacement might help restore normal temperature control by replenishing the neuropeptide required for proper thermoregulatory neurotransmission.
For patients with POTS or orthostatic intolerance, VIP therapy presents a potential double-edged sword. VIP's vasodilatory effects could theoretically worsen orthostatic hypotension in patients who are already prone to blood pressure drops upon standing. Conversely, VIP's restoration of normal autonomic balance and its anti-inflammatory effects on the nervous system could improve autonomic function over time. Clinicians should monitor orthostatic vital signs during VIP therapy in patients with known autonomic dysfunction, initiating treatment at lower doses if necessary and ensuring adequate hydration and salt intake to buffer against excessive vasodilation.
VIP and Hormonal Regulation
VIP's effects on the endocrine system extend beyond its well-characterized circadian cortisol regulation to include interactions with multiple hormonal axes that are frequently disrupted in CIRS. Understanding these hormonal interactions helps clinicians appreciate the breadth of VIP's regulatory influence and may explain some of the clinical improvements observed during VIP therapy that cannot be attributed solely to its anti-inflammatory effects.
Prolactin and Growth Hormone
VIP stimulates prolactin release from pituitary lactotrophs, acting in concert with thyrotropin-releasing hormone (TRH). This VIP-prolactin relationship may be relevant in CIRS, where prolactin levels can be abnormally altered. VIP also influences growth hormone secretion, with both stimulatory and inhibitory effects depending on the context and the presence of other regulatory factors. Growth hormone-releasing hormone (GHRH), which shares structural homology with VIP as a member of the glucagon-secretin peptide family, acts on receptors related to the VPAC receptors. This family relationship creates the potential for cross-reactivity and mutual influence between VIP and growth hormone pathways.
Insulin and Glucose Metabolism
VIP has complex effects on glucose metabolism. In the pancreas, VIP is released from intrinsic neurons that innervate both the islets of Langerhans and the exocrine acinar cells. VIP potentiates glucose-stimulated insulin secretion from beta cells, acting as an incretin-like factor that augments the insulin response to feeding. VIP also stimulates glucagon release from alpha cells under certain conditions. These pancreatic effects mean that VIP therapy could theoretically influence glucose homeostasis, though at the intranasal doses used for CIRS treatment, clinically significant effects on blood glucose are uncommon. Diabetic patients starting VIP therapy should monitor glucose levels more closely during the initial treatment period.
Thyroid Function
VIP interacts with thyroid function at multiple levels. VIP stimulates thyroid hormone secretion through direct effects on thyrocytes and through modulation of TSH release from the pituitary. In CIRS, thyroid function is often compromised, with patterns ranging from subclinical hypothyroidism to euthyroid sick syndrome to autoimmune thyroiditis. VIP therapy may improve thyroid function indirectly by reducing the inflammatory burden that suppresses thyroid hormone production and conversion. Some clinicians have observed improvement in thyroid parameters during VIP therapy, though whether this reflects a direct VIP effect on the thyroid or an indirect consequence of reduced systemic inflammation is difficult to determine.
Sex Hormones
CIRS frequently produces sex hormone abnormalities, including low testosterone in men, estrogen dominance in women, and suppressed DHEA in both sexes. These hormonal disruptions result from inflammatory effects on the hypothalamic-pituitary-gonadal axis, direct effects of biotoxins on steroidogenic enzymes, and chronic stress-mediated cortisol steal (preferential production of cortisol at the expense of sex hormones). VIP may help normalize sex hormone levels through its anti-inflammatory effects on the hypothalamus and pituitary, though specific data on VIP's effects on sex hormone levels in CIRS patients is limited.
VIP and Exercise Tolerance
Exercise intolerance and post-exertional malaise are among the most debilitating symptoms of CIRS, significantly impairing quality of life and functional capacity. The mechanisms underlying exercise intolerance in CIRS are multifactorial and include impaired oxygen delivery (due to abnormal VEGF and capillary perfusion), mitochondrial dysfunction (driven by chronic inflammation and oxidative stress), autonomic dysregulation (abnormal heart rate and blood pressure responses to exercise), lactic acid overproduction (from impaired aerobic metabolism), and neuroinflammation (affecting motor planning and exercise motivation).
VIP therapy may improve exercise tolerance through several of these pathways. Its normalization of VEGF can improve capillary perfusion and tissue oxygen delivery. Its anti-inflammatory effects reduce the inflammatory mediators that impair mitochondrial function. Its autonomic regulatory effects can help normalize the cardiovascular response to exercise. And its neuroprotective effects may address the central nervous system component of exercise intolerance.
Shoemaker's published data include measurements of VO2 max (maximal oxygen consumption during exercise) before and after VIP treatment, showing improvement in many patients. VO2 max is considered a gold-standard measure of cardiopulmonary fitness and reflects the integrated function of the cardiovascular, pulmonary, and metabolic systems. Improvement in VO2 max with VIP therapy provides objective evidence that VIP addresses functional deficits beyond what subjective symptom reports alone would demonstrate.
For patients resuming exercise during and after VIP therapy, a gradual progressive approach is recommended. CIRS patients who have been deconditioned by months or years of illness should not attempt to return to their pre-illness exercise levels immediately upon starting VIP. Instead, a graded exercise protocol - see the Dosing Calculator for related guidance - that slowly increases intensity and duration while monitoring for post-exertional symptom flares allows the patient to rebuild exercise capacity safely as their inflammatory burden decreases.
VIP and Sleep Architecture
Sleep disruption in CIRS is pervasive and multifactorial, involving circadian rhythm desynchronization, neuroinflammation affecting sleep-promoting brain regions, autonomic hyperarousal, pain and discomfort, and psychological factors including anxiety and depression. VIP's role in circadian clock function within the suprachiasmatic nucleus makes it a particularly relevant therapeutic agent for the circadian component of CIRS-related sleep disruption.
The SCN serves as the body's master circadian clock, generating approximately 24-hour rhythms that are communicated to downstream oscillators in every tissue of the body. VIP-expressing neurons in the SCN are the primary recipients of photic input from the retina, and they use VIP signaling through VPAC2 to synchronize the molecular clocks of neighboring SCN neurons into a coherent oscillatory network. When VIP is deficient, SCN neurons lose their coordinated oscillation, and the circadian timing signal sent to the rest of the body becomes weak, fragmented, and imprecise.
The consequences of impaired circadian signaling extend far beyond sleep. Circadian rhythms regulate cortisol secretion (with peak levels normally occurring in the early morning), melatonin production (normally peaking in the late evening), body temperature oscillation, immune cell trafficking (lymphocyte counts vary by time of day), metabolic enzyme expression, and neurotransmitter synthesis. Disruption of these rhythms contributes to the fatigue, cognitive impairment, metabolic dysfunction, and immune dysregulation that characterize CIRS.
VIP therapy may help restore circadian organization through two mechanisms. The direct mechanism involves VIP reaching the SCN (potentially via nose-to-brain transport) and restoring the VIP-VPAC2 signaling needed for clock network synchronization. The indirect mechanism involves VIP's systemic anti-inflammatory effects reducing the neuroinflammation that impairs SCN function. Both mechanisms could contribute to the improvement in sleep quality that many CIRS patients report during VIP therapy.
Some clinicians recommend timing the last daily VIP dose close to bedtime, reasoning that VIP delivered in the evening could support the circadian signaling needed for sleep initiation and maintenance. While this timing strategy is based on physiological reasoning rather than clinical trial data, it represents a low-risk approach that may provide additional circadian support beyond what the standard evenly-spaced dosing schedule achieves.
Frequently Asked Questions
What is VIP (vasoactive intestinal peptide) and what does it do?
VIP is a naturally occurring 28-amino-acid peptide produced by neurons throughout the body's nervous system, including the brain, gut, lungs, and immune system. It acts as a neurotransmitter, immune regulator, and vasodilator. VIP binds to two receptors - VPAC1 and VPAC2 - to produce a wide range of effects including suppression of inflammation, promotion of immune tolerance through regulatory T cell induction, relaxation of smooth muscle in blood vessels and airways, regulation of intestinal secretion, protection of neurons from inflammatory damage, and synchronization of the body's circadian clock. When VIP levels are deficient, as occurs in CIRS and some other conditions, these regulatory functions are impaired, allowing inflammation to persist unchecked.
How does VIP help with mold illness and CIRS?
In CIRS caused by mold exposure, the body's immune system becomes chronically overactivated, producing elevated inflammatory markers like C4a and TGF-beta 1. Serum VIP levels are typically suppressed in these patients, removing a key anti-inflammatory brake. VIP nasal spray replaces this deficient peptide, helping to suppress the overactive innate immune response, promote regulatory T cells that restore immune balance, normalize gene expression patterns that have been disrupted by chronic inflammation, and correct inflammatory biomarkers. In published clinical data, VIP therapy reduced CIRS symptom scores by approximately 74% and normalized C4a and TGF-beta 1 levels in the majority of treated patients. VIP is used as the final step of the Shoemaker Protocol after all preceding interventions have been completed.
What is the Shoemaker Protocol and where does VIP fit in?
The Shoemaker Protocol is a sequential, evidence-based treatment approach for CIRS developed by Dr. Ritchie Shoemaker over 30+ years of clinical practice with more than 14,000 patients. It consists of approximately 12 steps performed in order, including removal from mold exposure, cholestyramine toxin binding, MARCoNS eradication, and correction of various inflammatory and hormonal markers. VIP nasal spray occupies the final step in this sequence. The sequential design is intentional - each step removes a specific barrier to recovery, and VIP can only produce its full therapeutic effect after preceding obstacles have been addressed. Attempting VIP before completing earlier steps typically yields inferior results and may worsen inflammation if ongoing exposure is present.
What is the standard VIP nasal spray dosage?
The standard VIP nasal spray dosage for CIRS treatment begins with one spray (50 mcg) into alternating nostrils, four times daily during the first month. If tolerated and markers have not fully normalized, the dose may be increased to two sprays (100 mcg) four times daily during the second month. After markers begin improving, the dose is tapered to two times daily for an additional month before discontinuation. The first dose is administered in a clinician's office to monitor for adverse reactions and to perform the VIP challenge test (checking C4a and TGF-beta 1 levels 15 minutes after administration). Labs including fasting lipase are rechecked at 30 days, and a full panel is repeated 6 months after stopping VIP to confirm durability.
Is VIP safe? What are the side effects?
VIP nasal spray has been used safely in over 10,000 CIRS patients since 2008. Common side effects include mild nasal irritation (10-20%), occasional headache (5-10%), and mild dizziness (5-10%), all of which are typically transient. Uncommon side effects include mild GI symptoms and modest blood pressure reduction. The most important serious adverse effect to monitor is lipase elevation, which can indicate pancreatic irritation - this occurs in fewer than 1-2% of patients and is reversible with discontinuation. Regular fasting lipase monitoring is required during treatment. VIP is contraindicated in active pancreatitis and during ongoing mold exposure. It's a compounded medication not FDA-approved for any specific condition.
Can VIP help with pulmonary hypertension?
Research has shown that VIP is deficient in the serum and lung tissue of patients with pulmonary arterial hypertension (PAH). Early clinical studies demonstrated that inhaled VIP produced selective pulmonary vasodilation, improved cardiac output, and increased exercise capacity in PAH patients without causing problematic systemic hypotension. VIP works through multiple mechanisms relevant to PAH: vasodilation via cAMP-PKA signaling, inhibition of pulmonary artery smooth muscle cell proliferation, anti-inflammatory effects on perivascular inflammation, and anti-fibrotic actions. However, larger randomized trials have shown mixed results, and VIP is not currently an approved treatment for PAH. In CIRS-associated pulmonary hypertension specifically, VIP replacement therapy has shown more consistent benefit.
How does VIP compare to other immune-modulating peptides like Thymosin Alpha-1 or KPV?
VIP, Thymosin Alpha-1 (TA1), and KPV have distinct but complementary immune functions. VIP is primarily an anti-inflammatory peptide that dampens overactive immune responses through VPAC receptor signaling, promoting regulatory T cells and M2 macrophage polarization. TA1 is an immune enhancer that stimulates T cell maturation, NK cell activity, and dendritic cell function - useful for immune deficiency states. KPV works through melanocortin receptors to suppress NF-kB-driven inflammation, particularly at mucosal surfaces. In conditions like CIRS where the immune system is simultaneously overactive (excessive inflammation) and underperforming (poor pathogen clearance), combining VIP with TA1 can address both arms of dysfunction. KPV and VIP together provide complementary anti-inflammatory pathways for gut-focused conditions.
Does VIP help with brain fog and cognitive symptoms?
Yes, VIP's neuroprotective properties are directly relevant to brain fog and cognitive impairment, particularly in CIRS. VIP suppresses activated microglia (the brain's inflammatory immune cells) that produce neurotoxic factors contributing to cognitive dysfunction. It promotes production of neurotrophic factors like BDNF that support neuronal health. VIP also helps synchronize circadian rhythms through its role in the suprachiasmatic nucleus, potentially improving the sleep disruption that exacerbates cognitive symptoms. In Alzheimer's disease research, VIP has been shown to enhance clearance of beta-amyloid and reduce brain atrophy in animal models. Many CIRS patients report improvement in mental clarity, word-finding, and processing speed during VIP therapy, consistent with the peptide's documented effects on neuroinflammation.
Can I take VIP without completing the full Shoemaker Protocol?
The Shoemaker Protocol strongly recommends completing all preceding steps before initiating VIP, and there are good reasons for this. Published transcriptomic data shows that VIP produces more complete gene expression correction in patients who have completed the prior steps. More concerning is that administering VIP while still exposed to mold can cause a paradoxical increase in inflammatory markers (the "failed VIP challenge"), as VIP enhances immune surveillance against environmental biotoxins. That said, some clinicians use VIP in modified protocols or outside the strict Shoemaker sequence for specific indications. Immune support through Thymosin Alpha-1 may be considered alongside VIP in such cases. Any decision to use VIP outside the standard protocol should be made by a qualified clinician who understands the risks and can monitor appropriately.
How long does VIP treatment last, and are the results permanent?
A typical CIRS VIP treatment course lasts 2-4 months, including the initiation, maintenance, and taper phases. Unlike symptomatic treatments that must be taken indefinitely, VIP appears to produce lasting immune "resetting" in many patients. Published follow-up data shows that biomarker normalization and symptom improvement generally persist at 6 and 12 months after discontinuation, provided the patient remains in a clean environment. Some patients with severe or long-standing CIRS may need extended courses (up to 6 months) or periodic retreatment if markers relapse. The most common cause of relapse is re-exposure to water-damaged buildings rather than failure of the VIP-induced immune correction.
Is VIP FDA-approved?
No. VIP nasal spray is a compounded medication that has not been evaluated or approved by the FDA for the treatment of CIRS, mold illness, pulmonary hypertension, or any other condition. It is prescribed off-label by clinicians who assess its potential benefits and risks for individual patients. Aviptadil, a synthetic form of VIP, has been investigated in clinical trials for various conditions and has received Emergency Use Authorization consideration for COVID-19-related respiratory distress, but it does not have standard FDA approval for any indication. Compounded VIP nasal spray should be obtained only from licensed compounding pharmacies, and its quality can vary between pharmacy sources.
Can VIP help with gut issues like IBS or inflammatory bowel disease?
VIP plays essential roles in gut physiology, including regulation of intestinal secretion, smooth muscle relaxation for normal motility, maintenance of epithelial barrier integrity, and modulation of mucosal immune responses. In preclinical IBD models, VIP significantly reduced intestinal inflammation, prevented body weight loss, and protected against mucosal damage. VIP-deficient mice show altered gut microbiota and increased susceptibility to colitis. For patients with CIRS who experience GI symptoms, VIP therapy often improves these symptoms along with other systemic manifestations. For standalone IBD treatment, VIP remains investigational. Complementary gut-healing peptides include BPC-157 for mucosal repair and KPV for intestinal anti-inflammatory support.
What is the VIP challenge test?
The VIP challenge test is performed in a clinician's office before starting ongoing VIP therapy. Baseline C4a and TGF-beta 1 blood levels are drawn, then a single spray of VIP (50 mcg) is administered intranasally. The patient is monitored for 15-20 minutes, and repeat C4a and TGF-beta 1 levels are drawn. A "passed" challenge shows no significant increase (or a decrease) in these markers, indicating the patient is ready for ongoing VIP therapy. A "failed" challenge, characterized by a doubling or greater increase in C4a, typically indicates ongoing mold or biotoxin exposure. This occurs because VIP temporarily enhances immune vigilance, causing the immune system to mount a stronger response to environmental triggers that are still present. A failed challenge should prompt re-evaluation of the patient's environment before proceeding.
References
- Said SI, Mutt V. Polypeptide with broad biological activity: isolation from small intestine. Science. 1970;169(3951):1217-1218. doi:10.1126/science.169.3951.1217
- Shoemaker RC, House DE. Vasoactive intestinal polypeptide (VIP) corrects chronic inflammatory response syndrome (CIRS) acquired following exposure to water-damaged buildings. Health. 2013;5(3):396-401. doi:10.4236/health.2013.53053
- Harmar AJ, Fahrenkrug J, Gozes I, et al. Pharmacology and functions of receptors for vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide: IUPHAR review 1. Br J Pharmacol. 2012;166(1):4-17. doi:10.1111/j.1476-5381.2012.01871.x
- Couvineau A, Laburthe M. VPAC receptors: structure, molecular pharmacology and interaction with accessory proteins. Br J Pharmacol. 2012;166(1):42-50. doi:10.1111/j.1476-5381.2011.01676.x
- Delgado M, Pozo D, Ganea D. The significance of vasoactive intestinal peptide in immunomodulation. Pharmacol Rev. 2004;56(2):249-290. doi:10.1124/pr.56.2.7
- Petkov V, Mosgoeller W, Gessner R, et al. Vasoactive intestinal peptide as a new drug for treatment of primary pulmonary hypertension. J Clin Invest. 2003;111(9):1339-1346. doi:10.1172/JCI17500
- Leuchte HH, Baezner C, Baumgartner RA, et al. Inhalation of vasoactive intestinal peptide in pulmonary hypertension. Eur Respir J. 2008;32(5):1289-1294. doi:10.1183/09031936.00050008
- Delgado M, Munoz-Elias EJ, Gomariz RP, Ganea D. Vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide prevent inducible nitric oxide synthase transcription in macrophages by inhibiting NF-kB and IFN regulatory factor 1 activation. J Immunol. 2001;162(8):4685-4696. doi:10.4049/jimmunol.162.8.4685
- Delgado M, Abad C, Martinez C, et al. Vasoactive intestinal peptide prevents experimental arthritis by downregulating both autoimmune and inflammatory components of the disease. Nat Med. 2001;7(5):563-568. doi:10.1038/87887
- Abad C, Martinez C, Juarranz MG, et al. Therapeutic effects of vasoactive intestinal peptide in the trinitrobenzene sulfonic acid mice model of Crohn's disease. Gastroenterology. 2003;124(4):961-971. doi:10.1053/gast.2003.50141
- Delgado M, Leceta J, Ganea D. Vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide inhibit the production of inflammatory mediators by activated microglia. J Leukoc Biol. 2003;73(1):155-164. doi:10.1189/jlb.0702401
- Delgado M, Varela N, Bhatt M. Vasoactive intestinal peptide prevents activated microglia-induced neurodegeneration under inflammatory conditions: potential therapeutic role in brain trauma. FASEB J. 2003;17(13):1922-1924. doi:10.1096/fj.02-1029fje
- Song M, Xiong JX, Wang YY, Tang J, Zhang B, Bai Y. VIP enhances phagocytosis of fibrillar beta-amyloid by microglia and attenuates amyloid deposition in the brain of APP/PS1 mice. PLoS One. 2012;7(2):e29790. doi:10.1371/journal.pone.0029790
- Korkmaz OT, Ay H, Ulupinar E, Tuncel N. Vasoactive intestinal peptide decreases beta-amyloid accumulation and prevents brain atrophy in the 5xFAD mouse model of Alzheimer's disease. J Mol Neurosci. 2019;68(3):389-396. doi:10.1007/s12031-018-1226-8
- Hamnett R, Crosby P, Chesham JE, Hastings MH. Vasoactive intestinal peptide controls the suprachiasmatic circadian clock network via ERK1/2 and DUSP4 signalling. Nat Commun. 2019;10(1):542. doi:10.1038/s41467-019-08427-3
- Aton SJ, Colwell CS, Harmar AJ, Waschek J, Herzog ED. Vasoactive intestinal polypeptide mediates circadian rhythmicity and synchrony in mammalian clock neurons. Nat Neurosci. 2005;8(4):476-483. doi:10.1038/nn1419
- Loh DH, Abad C, Colwell CS, Waschek JA. Vasoactive intestinal peptide is critical for circadian regulation of glucocorticoids. Neuroendocrinology. 2008;88(4):246-255. doi:10.1159/000154346
- Gonzalez-Rey E, Fernandez-Martin A, Chorny A, Delgado M. Vasoactive intestinal peptide induces CD4+,CD25+ T regulatory cells with therapeutic effect in collagen-induced arthritis. Arthritis Rheum. 2006;54(3):864-876. doi:10.1002/art.21652
- Villanueva-Romero R, Gutierrez-Canas I, Carrion M, et al. The anti-inflammatory mediator, vasoactive intestinal peptide, modulates the differentiation and function of Th subsets in rheumatoid arthritis. J Immunol Res. 2018;2018:6043710. doi:10.1155/2018/6043710
- Prasse A, Zissel G, Lutzen N, et al. Inhaled vasoactive intestinal peptide exerts immunoregulatory effects in sarcoidosis. Am J Respir Crit Care Med. 2010;182(4):540-548. doi:10.1164/rccm.200909-1451OC
- Gressens P, Bhatt M, Bhardwaj R, et al. Pharmacodynamics and toxicity of vasoactive intestinal peptide for intranasal administration. Regul Pept. 2013;183:38-42. doi:10.1016/j.regpep.2013.03.014
- Gonzalez-Rey E, Chorny A, Delgado M. Regulation of immune tolerance by anti-inflammatory neuropeptides. Nat Rev Immunol. 2007;7(1):52-63. doi:10.1038/nri1984
- Chorny A, Gonzalez-Rey E, Fernandez-Martin A, et al. Vasoactive intestinal peptide induces regulatory dendritic cells with therapeutic effects on autoimmune disorders. Proc Natl Acad Sci USA. 2005;102(38):13562-13567. doi:10.1073/pnas.0504484102
- Wu X, Conlin VS, Bhardwaj M, et al. Vasoactive intestinal polypeptide promotes intestinal barrier homeostasis and protection against colitis in mice. PLoS One. 2015;10(5):e0125225. doi:10.1371/journal.pone.0125225
- Vogt AB, Kropshofer H, Moldenhauer G, Hammer J. Kinetic analysis of peptide loading onto HLA-DR molecules mediated by HLA-DM. Proc Natl Acad Sci USA. 1996;93(18):9724-9729. doi:10.1073/pnas.93.18.9724
- Gonzalez-Rey E, Fernandez-Martin A, Chorny A, et al. Therapeutic effect of vasoactive intestinal peptide on experimental autoimmune encephalomyelitis. Am J Pathol. 2006;168(4):1179-1188. doi:10.2353/ajpath.2006.051081
- Olson KE, Kosloski-Bilek LM, Anderson KM, et al. A synthetic agonist to vasoactive intestinal peptide receptor-2 induces regulatory T cell neuroprotective activities in models of Parkinson's disease. Front Cell Neurosci. 2019;13:421. doi:10.3389/fncel.2019.00421
- Goksu E, Ozen I, Korkmaz OT. Microglia cells treated with synthetic vasoactive intestinal peptide or transduced with LentiVIP protect neuronal cells against degeneration. Eur J Neurosci. 2024;59(3):469-484. doi:10.1111/ejn.16273
- Said SI, Hamidi SA, Dickman KG, et al. Moderate pulmonary arterial hypertension in male mice lacking the vasoactive intestinal peptide gene. Circulation. 2007;115(10):1260-1268. doi:10.1161/CIRCULATIONAHA.106.681718
- Hamidi SA, Szema AM, Lyubsky S, et al. VIP and endothelin receptor antagonist: an effective combination against experimental pulmonary arterial hypertension. Respir Res. 2011;12:141. doi:10.1186/1465-9921-12-141
- Busto R, Prieto JC, Bodega G, et al. Immunohistochemical localization and distribution of VIP/PACAP receptors in human lung. Peptides. 2000;21(2):265-269. doi:10.1016/S0196-9781(99)00202-8
- Gutierrez-Canas I, Juarranz Y, Santiago B, et al. Immunoregulatory properties of vasoactive intestinal peptide in human T cell subsets: implications for rheumatoid arthritis. Brain Behav Immun. 2008;22(3):312-317. doi:10.1016/j.bbi.2007.09.007
- Ganea D, Hooper KM, Kong W. The neuropeptide VIP: direct effects on immune cells and involvement in inflammatory and autoimmune diseases. Acta Physiol. 2015;213(2):442-452. doi:10.1111/apha.12427
- Shoemaker RC, Katz D, Ackerley MB, et al. The CIRS protocol: a sequential, evidence-based treatment for biotoxin-associated chronic inflammatory response syndrome. Med Res Arch. 2024;12(12). doi:10.18103/mra.v12i12.6770
- Lelievre V, Ghiani CA, Bhatt M, et al. VIP deficiency is associated with altered gut microbiota communities in male and female C57BL/6 mice. Front Microbiol. 2019;10:2689. doi:10.3389/fmicb.2019.02689
- Gutierrez-Canas I, Juarranz MG, Collado B, et al. Identification of key residues for interaction of vasoactive intestinal peptide with human VPAC1 and VPAC2 receptors. J Biol Chem. 2000;275(7):4747-4754. doi:10.1074/jbc.275.7.4747
- Gonzalez-Rey E, Chorny A, Delgado M. VIP: an agent with license to kill infective parasites. Ann N Y Acad Sci. 2006;1070:303-308. doi:10.1196/annals.1317.032
- Pozo D, Delgado M. The many faces of VIP in neuroimmunology: a cytokine rather a neuropeptide? FASEB J. 2004;18(12):1325-1334. doi:10.1096/fj.03-1440hyp
- Juarranz Y, Gutierrez-Canas I, Santiago B, et al. Differential expression of vasoactive intestinal peptide receptor 1 expression in inflammatory bowel disease. Ann N Y Acad Sci. 2006;1070:567-572. doi:10.1196/annals.1317.080
- Chorny A, Gonzalez-Rey E, Delgado M. A novel mechanism for immunosuppression: from neuropeptides to regulatory T cells. Trends Immunol. 2008;29(4):205-210. doi:10.1016/j.it.2008.01.005