Executive Summary
Figure 1: KPV tripeptide - the C-terminal fragment of alpha-melanocyte stimulating hormone with potent anti-inflammatory properties
Key Takeaways
- KPV is a three-amino-acid peptide (Lys-Pro-Val) from the C-terminal end of alpha-MSH with potent anti-inflammatory activity
- Works by inhibiting NF-kB activation, reducing TNF-alpha, IL-1beta, and IL-6 production
- Enters cells via PepT1 transporter rather than melanocortin receptors - a unique mechanism
- Preclinical evidence supports efficacy in colitis, IBD, skin inflammation, and infection
- Nanoparticle delivery systems achieve therapeutic effects at 12,000-fold lower concentrations than free KPV
KPV (Lys-Pro-Val) is a naturally occurring tripeptide derived from the C-terminal end of alpha-melanocyte stimulating hormone (alpha-MSH). Despite consisting of only three amino acids, KPV retains the full anti-inflammatory capacity of its parent hormone while eliminating unwanted effects such as skin pigmentation changes. This makes it one of the most efficient anti-inflammatory molecules known relative to its molecular size.
Alpha-MSH is a 13-amino-acid neuropeptide that plays a central role in immune regulation, pigmentation control, and metabolic signaling throughout the body. For decades, researchers studied the full-length hormone, assuming its biological activity required the complete molecular structure. That assumption changed when scientists discovered that the three C-terminal amino acids - lysine, proline, and valine - carried nearly all of the hormone's anti-inflammatory power. This finding opened a new chapter in peptide therapeutics, because smaller molecules are easier to manufacture, more stable in storage, and better suited for oral administration than their larger counterparts.
The primary mechanism through which KPV exerts its anti-inflammatory effects centers on the inhibition of nuclear factor kappa-B (NF-kB), a master transcription factor that controls the expression of dozens of pro-inflammatory genes. When NF-kB is activated - whether by bacterial infection, tissue injury, or autoimmune signaling - it triggers a cascade of inflammatory cytokines including tumor necrosis factor-alpha (TNF-alpha), interleukin-1 beta (IL-1beta), and interleukin-6 (IL-6). KPV interrupts this cascade at the nuclear level, preventing the transcription of these inflammatory mediators before they can be produced.
What makes KPV particularly interesting from a pharmacological standpoint is its mechanism of cellular entry. Unlike most melanocortin peptides, KPV does not rely on melanocortin receptors (MC1R through MC5R) to enter cells and produce its effects. Instead, it uses the proton-coupled oligopeptide transporter PepT1 (SLC15A1), a membrane protein normally responsible for absorbing dietary di- and tripeptides from the intestinal lumen. This transporter is expressed on intestinal epithelial cells, immune cells including macrophages and T lymphocytes, and various other tissue types. The PepT1-mediated uptake pathway has profound implications for KPV's therapeutic applications, especially in inflammatory bowel disease, where PepT1 expression is actually upregulated in inflamed colonic tissue - meaning the peptide concentrates precisely where it is needed most.
Preclinical research on KPV spans multiple inflammatory conditions. In murine models of dextran sodium sulfate (DSS)-induced colitis and 2,4,6-trinitrobenzenesulfonic acid (TNBS)-induced colitis, oral KPV administration reduced disease activity scores, decreased inflammatory cell infiltration measured by myeloperoxidase (MPO) activity, and accelerated recovery of body weight. Histological analysis confirmed reduced mucosal damage and lower colonic inflammation scores in treated animals compared to untreated controls. These findings have been published in leading gastroenterology journals and represent some of the strongest preclinical evidence for any peptide-based approach to inflammatory bowel disease.
Beyond gut inflammation, KPV has demonstrated efficacy in skin conditions driven by immune dysregulation. Topical application has shown promise in reducing the redness, scaling, and pruritis associated with psoriasis and atopic dermatitis. The peptide's small molecular weight of approximately 342 daltons allows it to penetrate skin barriers effectively when formulated appropriately, reaching the dermal layers where inflammatory processes originate. Research published in the Journal of Pharmaceutical Sciences has explored transdermal iontophoretic delivery of KPV across microporated human skin, achieving therapeutic concentrations in the target tissue.
One of the most exciting developments in KPV research involves advanced oral delivery systems. Scientists at the Georgia Institute of Technology and other institutions have developed hyaluronic acid-functionalized nanoparticles loaded with KPV (HA-KPV-NPs) that can survive the acidic environment of the stomach, reach the colon intact, and release their payload directly at sites of inflammation. These nanoparticles, encapsulated in a chitosan-alginate hydrogel, achieved therapeutic effects at concentrations 12,000-fold lower than free KPV in solution. The hyaluronic acid coating targets CD44 receptors on inflamed colonocytes and macrophages, providing a dual mechanism of targeted delivery and anti-inflammatory action. This work, published in Molecular Therapy, represents a significant step toward clinically viable oral KPV formulations for ulcerative colitis and Crohn's disease.
The safety profile of KPV appears favorable based on available evidence. As a naturally derived fragment of an endogenous hormone, it does not trigger the immune responses sometimes associated with synthetic peptides. No significant toxicity has been reported in animal studies at therapeutic doses, and the peptide's rapid metabolism limits systemic accumulation. However, it should be recognized that human clinical trials specifically evaluating KPV remain limited. The related compound (CKPV)2, a synthetic dimer of the KPV sequence with added cysteine residues, has progressed further in clinical development, particularly for vulvovaginal candidiasis, where it demonstrated both antifungal and anti-inflammatory properties in Phase II trials.
For individuals exploring peptide-based approaches to inflammatory conditions, KPV represents a compelling option supported by a growing body of preclinical evidence. The peptide research hub provides additional context on how KPV fits within the broader field of therapeutic peptides, while the science and research section offers deeper exploration of the molecular pathways involved. As research continues to advance, KPV may emerge as a first-line peptide therapy for conditions ranging from ulcerative colitis to chronic dermatitis, particularly as novel delivery technologies make oral administration increasingly practical.
Key Takeaways
- KPV is a three-amino-acid peptide (Lys-Pro-Val) from the C-terminal end of alpha-MSH with potent anti-inflammatory activity
- Works by inhibiting NF-kB activation, reducing TNF-alpha, IL-1beta, and IL-6 production
- Enters cells via PepT1 transporter rather than melanocortin receptors - a unique mechanism
- Preclinical evidence supports efficacy in colitis, IBD, skin inflammation, and infection
- Nanoparticle delivery systems achieve therapeutic effects at 12,000-fold lower concentrations than free KPV
- Available in oral, injectable, and topical formulations for different therapeutic targets
Alpha-MSH & KPV Derivation
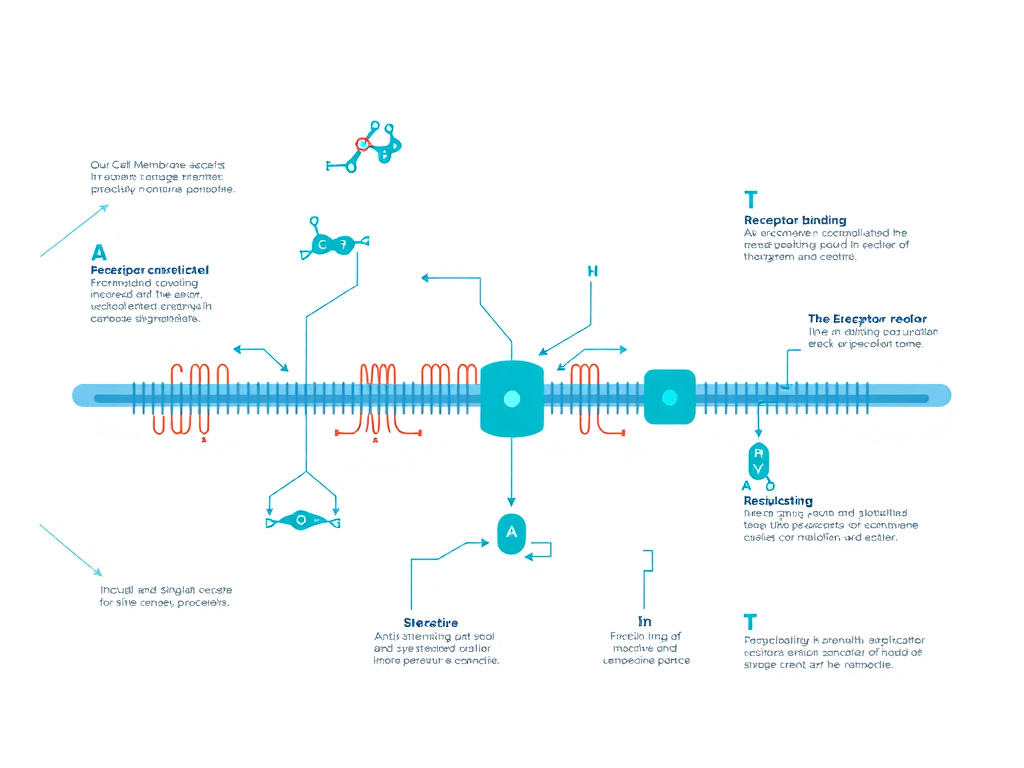
Figure 2: Alpha-MSH processing pathway showing the derivation of the C-terminal KPV tripeptide
Alpha-melanocyte stimulating hormone (alpha-MSH) is a 13-amino-acid peptide hormone produced from the post-translational processing of proopiomelanocortin (POMC) in the pituitary gland, hypothalamus, skin, and immune cells. Its complete sequence is Ac-Ser-Tyr-Ser-Met-Glu-His-Phe-Arg-Trp-Gly-Lys-Pro-Val-NH2. The C-terminal three residues - Lys11-Pro12-Val13 - constitute KPV, the minimal anti-inflammatory pharmacophore of the hormone.
Proopiomelanocortin: The Precursor Protein
Understanding KPV requires tracing its origins to POMC, a 241-amino-acid precursor protein that serves as the molecular source for several biologically active peptides. POMC is synthesized primarily in the anterior and intermediate lobes of the pituitary gland, but significant expression also occurs in the arcuate nucleus of the hypothalamus, keratinocytes in the skin, and cells of the immune system including lymphocytes and monocytes. Through a series of tissue-specific enzymatic cleavages by prohormone convertases (PC1 and PC2), POMC gives rise to adrenocorticotropic hormone (ACTH), beta-endorphin, beta-lipotropin, and the melanocyte-stimulating hormones alpha-MSH, beta-MSH, and gamma-MSH.
The generation of alpha-MSH from POMC involves two sequential processing steps. First, PC1 cleaves POMC to produce ACTH(1-39). Then, PC2 cleaves ACTH to release alpha-MSH, which corresponds to ACTH(1-13). Post-translational modifications follow: acetylation of the N-terminal serine and amidation of the C-terminal valine. These modifications are critical for biological activity, as they increase the peptide's binding affinity for melanocortin receptors by approximately 10 to 100-fold and protect against enzymatic degradation, extending its half-life in circulation.
The POMC gene is highly conserved across vertebrate species, reflecting the fundamental importance of melanocortin signaling in physiology. Mutations in POMC or its processing enzymes cause severe metabolic disorders in humans, including early-onset obesity, adrenal insufficiency, and red hair pigmentation. This conservation also means that research findings on alpha-MSH and KPV from animal models have strong translational relevance to human biology.
The Melanocortin System
Alpha-MSH signals through a family of five G protein-coupled receptors designated MC1R through MC5R. Each receptor has distinct tissue distribution and physiological roles. MC1R, expressed on melanocytes, keratinocytes, and immune cells, mediates pigmentation and anti-inflammatory responses. MC2R is the ACTH receptor, critical for adrenal steroidogenesis. MC3R and MC4R, primarily expressed in the brain, regulate energy homeostasis and feeding behavior. MC5R, found in sebaceous glands and other exocrine tissues, influences lipid secretion.
The anti-inflammatory effects of full-length alpha-MSH have been attributed primarily to MC1R signaling. When alpha-MSH binds MC1R on macrophages, dendritic cells, and other immune cells, it triggers adenylate cyclase activation, increases intracellular cyclic AMP (cAMP), and activates protein kinase A (PKA). This signaling cascade suppresses NF-kB nuclear translocation and reduces the transcription of pro-inflammatory genes. The MC1R-mediated pathway also promotes IL-10 production, an anti-inflammatory cytokine that helps resolve inflammation and restore tissue homeostasis.
The melanocortin system's role extends beyond simple receptor activation. Alpha-MSH also modulates the balance between pro-inflammatory M1 macrophages and anti-inflammatory M2 macrophages, promotes regulatory T cell differentiation, and inhibits neutrophil migration to sites of inflammation. These broad immunomodulatory effects make melanocortin peptides attractive therapeutic candidates for conditions characterized by chronic or dysregulated inflammation. For those interested in other peptides affecting immune regulation, Thymosin Alpha-1 represents another important immunomodulatory compound worth investigating.
Dissecting the Active Sequence: How KPV Was Identified
The identification of KPV as the minimal anti-inflammatory fragment of alpha-MSH emerged from systematic structure-activity relationship studies conducted over several decades. Early work in the 1980s and 1990s established that alpha-MSH had potent anti-inflammatory effects independent of its pigmentary activity. Researchers then began asking a fundamental question: which portion of the 13-amino-acid sequence was responsible for each biological function?
The core pharmacophore for melanocortin receptor binding and pigmentary activity was identified as the central His-Phe-Arg-Trp sequence (residues 6-9), often called the "core" or "message" sequence. This tetrapeptide is essential for receptor recognition and activation. Modifications to these residues dramatically reduce or eliminate MC1R binding affinity.
The C-terminal region, in contrast, was found to be dispensable for receptor binding but critical for anti-inflammatory activity. Landmark studies by Catania, Lipton, and colleagues demonstrated that the tripeptide KPV (alpha-MSH 11-13) retained the anti-inflammatory effects of the full hormone while lacking any pigmentary activity. In cell-based assays, KPV suppressed NF-kB activation, reduced pro-inflammatory cytokine production, and inhibited nitric oxide synthesis at nanomolar concentrations - effects comparable to those of full-length alpha-MSH.
A critical finding published in the Journal of Pharmacology and Experimental Therapeutics in 2003 formally dissected the contributions of the core and C-terminal sequences. The study showed that KPV inhibited IL-1beta-induced NF-kB activation in human intestinal epithelial cells with an IC50 in the low nanomolar range, while the core sequence HFRW had minimal effect on this pathway. This established that the anti-inflammatory and receptor-mediated activities of alpha-MSH reside in different parts of the molecule - a finding with enormous therapeutic implications.
KPV's Receptor-Independent Mechanism
Perhaps the most surprising discovery about KPV was that its anti-inflammatory effects do not require melanocortin receptors at all. This was demonstrated through several lines of evidence. First, KPV retained its anti-inflammatory activity in cells lacking functional MC1R expression. Second, melanocortin receptor antagonists such as agouti-related protein (AgRP) and SHU9119 did not block KPV's effects. Third, the structural requirements for MC1R binding (the HFRW core) are absent from KPV, making receptor-mediated signaling physically impossible.
The discovery of PepT1-mediated uptake, published in Gastroenterology in 2008 by Dalmasso and colleagues, provided the mechanistic explanation. PepT1 (SLC15A1) is a proton-coupled oligopeptide transporter that moves di- and tripeptides across cell membranes using the electrochemical proton gradient as the driving force. Under normal conditions, PepT1 is expressed primarily on the apical surface of small intestinal enterocytes, where it absorbs dietary peptides generated by protein digestion.
In inflammatory bowel disease, PepT1 expression is induced in the colon - a tissue that normally expresses very low levels of this transporter. This disease-specific upregulation means that inflamed colonic epithelium has an enhanced capacity to absorb KPV, creating a natural targeting mechanism. Once inside the cell, KPV directly interferes with NF-kB signaling by preventing the phosphorylation and degradation of IkB-alpha, the inhibitory protein that keeps NF-kB sequestered in the cytoplasm. With IkB-alpha intact, NF-kB cannot translocate to the nucleus, and pro-inflammatory gene transcription is suppressed.
This receptor-independent mechanism distinguishes KPV from all other melanocortin-based therapeutics and from most conventional anti-inflammatory agents. It means KPV can work in tissues regardless of melanocortin receptor expression, and its efficacy is actually enhanced at sites of active inflammation where PepT1 is upregulated. For a broader understanding of how peptides interact with cellular transport systems, the peptide research hub covers related transport mechanisms.
Structural Properties and Stability
KPV's small size confers several practical advantages. With a molecular weight of approximately 342 daltons, it falls well below the 500-dalton threshold often cited as the upper limit for oral bioavailability (Lipinski's Rule of Five). The peptide is relatively stable compared to larger peptide hormones, though it remains susceptible to enzymatic degradation by peptidases in the gastrointestinal tract and bloodstream. Its half-life in plasma is estimated at minutes rather than hours, which necessitates either frequent dosing, sustained-release formulations, or advanced delivery systems for clinical applications.
The three constituent amino acids each contribute to the peptide's biological activity. Lysine (K) provides a positive charge at physiological pH, which may facilitate electrostatic interactions with negatively charged components of inflammatory signaling complexes. Proline (P) introduces a rigid kink in the peptide backbone that constrains the molecule's three-dimensional conformation, potentially positioning the flanking residues for optimal interaction with intracellular targets. Valine (V) provides hydrophobic character that may assist in membrane interactions and cellular uptake.
Synthetic KPV is produced through standard solid-phase peptide synthesis (SPPS) using Fmoc chemistry. The simplicity of the sequence - just three standard amino acids with no unusual modifications required - makes manufacturing straightforward and cost-effective compared to longer or more complex peptides. This accessibility has facilitated widespread research and makes KPV an attractive candidate for eventual pharmaceutical development.
The CKPV Dimer: An Enhanced Derivative
Building on the discovery of KPV's anti-inflammatory properties, researchers developed (CKPV)2, a synthetic dimer created by linking two Cys-Lys-Pro-Val units through a disulfide bond between the cysteine residues. This octapeptide, also designated CZEN-002 in clinical development, demonstrated enhanced anti-inflammatory and antimicrobial properties compared to monomeric KPV.
The (CKPV)2 dimer showed candidacidal activity against multiple Candida species, including azole-resistant strains of C. albicans, C. krusei, and C. glabrata, at sub-millimolar concentrations. Unlike conventional antifungal peptides that kill through membrane disruption, (CKPV)2 works by increasing cyclic AMP levels within the fungal cell, a non-membranolytic mechanism that reduces the risk of resistance development. In a rat model of vulvovaginal candidiasis, (CKPV)2 reduced fungal colony counts in a dose-dependent manner while simultaneously dampening the inflammatory response through M2 macrophage polarization and IL-10 induction.
The clinical development of (CKPV)2 for vulvovaginal candidiasis represents the most advanced therapeutic application of KPV-derived molecules. While clinical trial data remain limited in the public domain, the compound's dual antifungal and anti-inflammatory properties address a genuine unmet medical need, as current antifungal treatments do not address the inflammatory component of candidal infections. For those interested in antimicrobial peptides, LL-37 is another important peptide with both antimicrobial and immunomodulatory functions.
NF-kB Inhibition Mechanism
Figure 3: KPV mechanism of NF-kB pathway inhibition - from PepT1 transport to cytokine suppression
KPV reduces inflammation primarily by blocking the activation of nuclear factor kappa-B (NF-kB), a transcription factor family that controls the expression of over 200 genes involved in immune response, cell survival, and inflammatory signaling. At nanomolar concentrations, KPV prevents NF-kB nuclear translocation and simultaneously inhibits MAP kinase (MAPK) pathways, producing a dual anti-inflammatory effect that suppresses multiple inflammatory mediators at once.
NF-kB: The Master Inflammatory Switch
NF-kB is not a single protein but a family of five related transcription factors: RelA (p65), RelB, c-Rel, NF-kB1 (p105/p50), and NF-kB2 (p100/p52). These proteins form homo- and heterodimers that bind to specific DNA sequences called kB sites in the promoter regions of target genes. The most common and best-studied dimer is the p65/p50 heterodimer, which drives the expression of pro-inflammatory cytokines, chemokines, adhesion molecules, and enzymes involved in inflammation.
Under resting conditions, NF-kB dimers are held in the cytoplasm by a family of inhibitory proteins called IkBs (inhibitors of kappa-B). The most important of these is IkB-alpha, which masks the nuclear localization signal on NF-kB, preventing its entry into the nucleus. This cytoplasmic sequestration represents the "off" switch for NF-kB-dependent gene expression.
Activation of NF-kB occurs through two main pathways: the canonical (classical) pathway and the non-canonical (alternative) pathway. The canonical pathway, which is the primary target of KPV, is triggered by a wide range of stimuli including bacterial lipopolysaccharide (LPS), pro-inflammatory cytokines like TNF-alpha and IL-1beta, reactive oxygen species, and viral components. These signals converge on the IkB kinase (IKK) complex, composed of two catalytic subunits (IKK-alpha and IKK-beta) and a regulatory subunit (NEMO/IKK-gamma). Activated IKK phosphorylates IkB-alpha at serine residues 32 and 36, marking it for ubiquitination and proteasomal degradation. With IkB-alpha destroyed, NF-kB is free to enter the nucleus and activate its target genes.
How KPV Blocks NF-kB Activation
KPV interrupts the canonical NF-kB pathway at a critical step: it prevents the phosphorylation and subsequent degradation of IkB-alpha. Experimental evidence from multiple laboratories has demonstrated this mechanism using several complementary approaches.
In human intestinal epithelial cell lines (Caco-2, HT-29, and SW480), treatment with KPV at concentrations as low as 10 nanomolar significantly reduced LPS-stimulated and TNF-alpha-stimulated NF-kB activation, as measured by electrophoretic mobility shift assays (EMSA) and NF-kB-driven luciferase reporter assays. Western blot analysis showed that KPV-treated cells maintained higher levels of IkB-alpha protein compared to untreated controls following inflammatory stimulation, confirming that the peptide protects IkB-alpha from degradation.
The mechanism by which KPV stabilizes IkB-alpha appears to involve inhibition of the IKK complex itself, though the exact molecular interaction has not been fully characterized. One possibility is that KPV directly binds to IKK-beta, the kinase primarily responsible for IkB-alpha phosphorylation in the canonical pathway, preventing its catalytic activity. Another hypothesis suggests that KPV modulates upstream kinases such as TAK1 (TGF-beta-activated kinase 1) that activate IKK. Research published in the Journal of Biological Chemistry has provided evidence supporting the latter mechanism, showing that KPV reduces TAK1 phosphorylation in a dose-dependent manner.
The downstream consequences of NF-kB inhibition by KPV are extensive. With NF-kB unable to reach its target genes, the cell produces significantly less TNF-alpha, IL-1beta, IL-6, IL-8, and other pro-inflammatory cytokines. Expression of adhesion molecules such as ICAM-1 and VCAM-1 is reduced, limiting the recruitment of inflammatory cells to tissue sites. Inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2) expression is suppressed, reducing nitric oxide and prostaglandin production respectively. These combined effects produce a broad anti-inflammatory response that mirrors, in many ways, the action of corticosteroids - but without the metabolic, endocrine, and immunosuppressive side effects that limit long-term steroid use.
MAPK Pathway Inhibition
In addition to NF-kB suppression, KPV inhibits the mitogen-activated protein kinase (MAPK) signaling cascades. The three major MAPK pathways - ERK1/2 (extracellular signal-regulated kinase), JNK (c-Jun N-terminal kinase), and p38 MAPK - play complementary roles in inflammatory gene expression. While NF-kB is the primary driver of cytokine transcription, the MAPK pathways regulate mRNA stability, translation efficiency, and additional transcription factors such as AP-1 that cooperate with NF-kB at many inflammatory gene promoters.
Studies in human colonic epithelial cells and macrophages have shown that KPV reduces the phosphorylation of all three MAPK pathways following inflammatory stimulation. The p38 MAPK pathway appears to be particularly sensitive to KPV inhibition. This is therapeutically relevant because p38 MAPK is a major regulator of TNF-alpha post-transcriptional processing - it controls the stability and translation of TNF-alpha mRNA through phosphorylation of RNA-binding proteins such as tristetraprolin (TTP) and HuR. By inhibiting p38 MAPK, KPV reduces TNF-alpha production at both the transcriptional level (via NF-kB) and the post-transcriptional level (via MAPK), creating a multi-layered suppression of this critical inflammatory cytokine.
The dual inhibition of NF-kB and MAPK pathways distinguishes KPV from many conventional anti-inflammatory compounds that target only one pathway. Corticosteroids primarily work through the glucocorticoid receptor to suppress NF-kB, while NSAIDs target COX enzymes downstream of both pathways. KPV's ability to simultaneously block two major inflammatory signaling cascades may explain its strong efficacy in preclinical models despite its remarkably small molecular size. For comparison with other multi-pathway anti-inflammatory approaches, BPC-157 also modulates multiple inflammatory cascades through distinct molecular mechanisms.
Cytokine Profile Modulation
The net effect of NF-kB and MAPK inhibition by KPV produces a characteristic shift in the cytokine profile of treated tissues. Pro-inflammatory cytokines are suppressed while anti-inflammatory mediators are preserved or enhanced. This selective modulation is one of KPV's most therapeutically valuable properties.
Quantitative measurements from cell culture and animal studies provide specific numbers. In LPS-stimulated human monocytes, KPV at 100 nanomolar reduced TNF-alpha secretion by 40-60%, IL-1beta by 35-50%, and IL-6 by 45-65% compared to untreated controls. These reductions are clinically meaningful - they bring cytokine levels down from the pathological range associated with active inflammation toward the baseline levels seen in healthy tissue, without completely eliminating the immune response.
IL-10 production, in contrast, is maintained or modestly enhanced by KPV treatment. IL-10 is a critical anti-inflammatory cytokine that promotes the resolution of inflammation and tissue repair. Its preservation during KPV treatment ensures that the healing phase of the inflammatory response is not impaired, a key advantage over broad-spectrum immunosuppressants that reduce all cytokines indiscriminately.
KPV also reduces the expression of chemokines including IL-8 (CXCL8) and monocyte chemoattractant protein-1 (MCP-1/CCL2). These molecules recruit neutrophils and monocytes to sites of inflammation, and their suppression limits the influx of additional immune cells that can perpetuate tissue damage. In colonic tissue from DSS-colitis mice, KPV treatment reduced myeloperoxidase (MPO) activity - a marker of neutrophil infiltration - by approximately 50-70%, reflecting the functional consequence of reduced chemokine expression.
Comparison with Other Anti-Inflammatory Mechanisms
Placing KPV's mechanism in context with other anti-inflammatory agents helps clarify its therapeutic niche. The table below summarizes key mechanistic differences.
| Agent | Primary Target | Mechanism | Selectivity | Side Effect Profile |
|---|---|---|---|---|
| KPV | NF-kB + MAPK | IkB-alpha stabilization, kinase inhibition | High (inflammatory cells via PepT1) | Minimal reported |
| Corticosteroids | Glucocorticoid receptor | Transcription factor interference | Low (systemic) | Metabolic, endocrine, bone |
| NSAIDs | COX-1/COX-2 | Enzyme inhibition | Moderate | GI, renal, cardiovascular |
| Anti-TNF biologics | TNF-alpha | Cytokine neutralization | High (single cytokine) | Infection risk, immunogenicity |
| JAK inhibitors | JAK1/JAK2/JAK3 | Kinase inhibition | Moderate | Infection, thrombosis |
| BPC-157 | Multiple (NO, growth factors) | Multi-pathway modulation | Moderate | Minimal reported |
KPV occupies a unique position in this landscape. It acts further upstream than NSAIDs or anti-TNF biologics, blocking the transcriptional programs that generate multiple inflammatory mediators simultaneously. Yet it achieves this broad suppression with apparent selectivity for inflamed tissue, thanks to PepT1-mediated uptake that concentrates the peptide where inflammation is active. This combination of broad mechanism and targeted delivery is rare among anti-inflammatory agents and makes KPV a particularly promising candidate for chronic inflammatory conditions where long-term treatment is necessary.
Oxidative Stress Reduction
Beyond direct NF-kB and MAPK inhibition, KPV reduces oxidative stress - a major contributor to tissue damage in chronic inflammation. NF-kB activation and reactive oxygen species (ROS) production form a positive feedback loop: ROS activate NF-kB, which in turn promotes iNOS and NADPH oxidase expression, generating more ROS. Breaking this cycle is essential for resolving established inflammation.
KPV interrupts this feedback at the NF-kB node, reducing iNOS expression and consequently lowering tissue levels of nitric oxide and peroxynitrite, a highly reactive nitrogen species that damages DNA, proteins, and lipids. In colonic tissue from colitis models, KPV treatment reduced markers of oxidative damage including malondialdehyde (MDA, a lipid peroxidation product) and 8-hydroxydeoxyguanosine (8-OHdG, a DNA oxidation marker). These effects contribute to tissue preservation during active inflammation and may accelerate the healing process once the acute inflammatory episode subsides.
Recent research published in 2025 in ScienceDirect demonstrated that KPV at 50 micrograms per milliliter restored cell viability and reduced IL-1beta secretion in human keratinocytes exposed to fine particulate matter (PM10), providing evidence that KPV's antioxidant and anti-inflammatory properties extend to environmental stress as well as classical immune activation. This finding broadens the potential applications of KPV to include protection against pollution-induced skin damage, an increasingly relevant concern in urban populations. Individuals exploring antioxidant peptides may also find value in learning about SS-31, a mitochondria-targeted peptide with complementary antioxidant properties.
Evolutionary Conservation of Melanocortin Anti-Inflammatory Signaling
The melanocortin system is one of the most ancient signaling networks in vertebrate biology, with clear orthologues identified in fish, amphibians, reptiles, birds, and mammals. Alpha-MSH-like peptides have been found in species as evolutionarily distant as lampreys and hagfish, indicating that the melanocortin system predates the divergence of jawed and jawless vertebrates over 500 million years ago. This deep evolutionary conservation suggests that the anti-inflammatory functions of alpha-MSH and its fragments, including KPV, serve fundamental biological roles that have been maintained through hundreds of millions of years of natural selection.
In teleost fish, alpha-MSH peptides regulate skin color and stress responses while also modulating immune function. Cortisol-MSH interactions in fish provide insights into the ancestral relationship between stress response and immune regulation that persists in mammalian physiology. The conservation of the KPV C-terminal sequence across diverse vertebrate species is particularly striking: the Lys-Pro-Val motif is identical in humans, mice, rats, cattle, sheep, and many other mammals, and closely related sequences are found across vertebrate taxa.
This evolutionary conservation has practical implications for KPV research. It means that preclinical studies in mice and rats are conducted with the identical peptide sequence that would be used in humans, eliminating concerns about species-specific sequence variations that complicate the translation of many drug candidates. It also suggests that the biological targets and pathways engaged by KPV are conserved across species, increasing confidence that preclinical results will predict human responses.
The conservation of melanocortin anti-inflammatory signaling also raises questions about the physiological role of endogenous KPV. Alpha-MSH is degraded by various peptidases in vivo, and the C-terminal KPV fragment is one of the predictable products of this degradation. Whether KPV generated from alpha-MSH breakdown serves as an endogenous anti-inflammatory signal in its own right, or whether the anti-inflammatory activity of KPV is a pharmacological property that is not physiologically relevant at endogenous concentrations, remains an open question. Some evidence suggests that local alpha-MSH production and processing in inflamed tissues may generate KPV concentrations sufficient for paracrine anti-inflammatory signaling, particularly in the skin where POMC expression and processing are well-documented.
Structure-Activity Relationships: What Each Amino Acid Contributes
Detailed structure-activity relationship (SAR) studies have dissected the contribution of each amino acid in the KPV sequence to its anti-inflammatory activity. These studies, conducted using synthetic analogs with single amino acid substitutions, provide insights into the molecular basis of KPV's mechanism and guide the design of improved derivatives.
Lysine at position 11 (K): The epsilon-amino group of lysine provides a positive charge at physiological pH (the side chain pKa is approximately 10.5, ensuring nearly complete protonation at pH 7.4). This positive charge appears to be important for anti-inflammatory activity, as substitution with uncharged residues (alanine, leucine) significantly reduces NF-kB inhibition. The positive charge may facilitate electrostatic interactions with negatively charged phosphate groups on kinase substrates or with anionic components of the NF-kB signaling complex. Substitution with arginine (another positively charged residue) partially preserves activity, supporting the importance of charge rather than specific side chain geometry. The lysine residue also provides the substrate recognition element for PepT1 transport, as the transporter preferentially binds peptides with basic residues at the N-terminal position.
Proline at position 12 (P): Proline is unique among the standard amino acids in that its side chain forms a cyclic structure with the backbone nitrogen, creating a rigid kink in the peptide backbone. This conformational constraint is critical for KPV's bioactivity. Substitution of proline with flexible residues (glycine, alanine) dramatically reduces anti-inflammatory activity, indicating that the specific three-dimensional shape imposed by proline is necessary for proper interaction with intracellular targets. The proline ring also confers resistance to many aminopeptidases, contributing to KPV's relative stability compared to other tripeptides. In the PepT1 transporter, proline-containing peptides are recognized with moderate affinity, ensuring efficient cellular uptake without competing excessively with dietary peptide absorption.
Valine at position 13 (V): The C-terminal valine provides hydrophobic character through its isopropyl side chain. SAR studies show that substitution with other hydrophobic residues (leucine, isoleucine) partially preserves activity, while replacement with polar residues (serine, aspartate) reduces activity more substantially. This suggests that hydrophobic interactions contribute to KPV's binding to its intracellular target(s). The C-terminal carboxyl group of valine may also participate in hydrogen bonding or electrostatic interactions. In the parent hormone alpha-MSH, this valine is amidated (converted to valine-NH2), which enhances receptor binding affinity. In free KPV, the carboxyl group is free, which may affect its intracellular interactions compared to C-terminally amidated alpha-MSH.
The combined SAR data paint a picture of KPV as a molecule whose anti-inflammatory activity depends on a specific combination of charge (lysine), conformational constraint (proline), and hydrophobicity (valine). This molecular "code" enables KPV to interact productively with its intracellular targets while maintaining PepT1 substrate properties for cellular entry. The relatively strict structural requirements suggest a specific binding interaction rather than a nonspecific effect, supporting the hypothesis that KPV has a defined molecular target within the NF-kB signaling pathway.
Comparison with Other Melanocortin-Derived Peptides
KPV exists within a family of melanocortin-derived peptides with varying biological activities. Understanding these relationships helps position KPV's unique therapeutic niche.
| Peptide | Sequence | Receptor Binding | Anti-Inflammatory | Pigmentary | Key Applications |
|---|---|---|---|---|---|
| Alpha-MSH | Ac-SYSMEHFRWGKPV-NH2 | MC1R, MC3R, MC4R, MC5R | Strong | Strong | Research tool, inflammation models |
| KPV | KPV | None | Strong | None | Gut/skin inflammation |
| HFRW (core) | HFRW | MC1R, MC3R, MC4R | Weak | Moderate | Receptor studies |
| (CKPV)2 | (CKPV)2 dimer | None | Strong | None | Antifungal + anti-inflammatory |
| NDP-MSH | Ac-SYS-Nle-EHFRWGKPV-NH2 | MC1R (superpotent) | Strong | Very strong | Research, Melanotan II analog |
| ACTH(1-39) | Full ACTH sequence | MC2R primarily | Moderate | Weak | Adrenal steroidogenesis |
| Setmelanotide | Cyclic analog | MC4R selective | Minimal | Minimal | Genetic obesity (FDA-approved) |
This comparison highlights KPV's unique position: it is the only melanocortin-derived peptide that provides strong anti-inflammatory activity without any receptor binding, pigmentary effects, or appetite/metabolic modulation. This selectivity makes it the cleanest anti-inflammatory agent among melanocortin peptides, with the narrowest side effect profile. For individuals interested in melanocortin peptides for other purposes, Melanotan II and PT-141 act through melanocortin receptor binding to achieve tanning and sexual function effects respectively, but these compounds would not be appropriate choices for pure anti-inflammatory therapy.
Crosstalk Between NF-kB and Other Inflammatory Pathways
NF-kB does not operate in isolation. It interacts extensively with other inflammatory and metabolic signaling pathways, creating a complex network of cross-regulation that amplifies inflammatory responses. Understanding these interactions helps explain why KPV's NF-kB inhibition produces such broad anti-inflammatory effects and why the peptide may benefit conditions beyond those directly driven by NF-kB activation.
The JAK-STAT pathway, targeted by drugs like tofacitinib and upadacitinib in IBD, interacts with NF-kB at multiple levels. STAT3, activated downstream of IL-6 receptor signaling, physically interacts with the NF-kB p65 subunit to co-regulate gene expression at certain promoters. By reducing IL-6 production through NF-kB inhibition, KPV indirectly reduces STAT3 activation, creating a secondary anti-inflammatory effect through the JAK-STAT pathway. This indirect STAT3 suppression could contribute to KPV's efficacy in conditions like Crohn's disease where STAT3-driven Th17 responses play a prominent pathological role.
The inflammasome pathway, particularly the NLRP3 inflammasome, requires NF-kB-dependent priming for activation. NLRP3 expression is transcriptionally regulated by NF-kB, and the production of pro-IL-1beta (which is cleaved to active IL-1beta by the inflammasome) is also NF-kB-dependent. KPV's NF-kB inhibition could therefore suppress both the priming and the substrate availability for inflammasome activation, providing a dual brake on IL-1beta production. This is particularly relevant to IBD, where NLRP3 inflammasome activation in intestinal macrophages contributes to mucosal inflammation and tissue damage.
The Wnt/beta-catenin pathway, critical for intestinal stem cell maintenance and epithelial renewal, also intersects with NF-kB signaling. In IBD, chronic NF-kB activation can disrupt Wnt signaling in intestinal stem cells, impairing epithelial regeneration. By reducing NF-kB activity, KPV may restore normal Wnt/beta-catenin signaling and promote epithelial renewal, contributing to mucosal healing beyond its direct anti-inflammatory effects. This potential pro-regenerative activity, while not yet confirmed for KPV specifically, aligns with the observed histological improvements in colitis models where crypt architecture is better preserved in KPV-treated animals.
The PI3K/Akt/mTOR pathway, which regulates cell survival, metabolism, and protein synthesis, also cross-talks with NF-kB. Akt can activate NF-kB through phosphorylation of IKK, while NF-kB target genes include several components of the PI3K signaling network. KPV's impact on this cross-regulation has not been specifically studied but could influence cell survival decisions during inflammation, potentially affecting whether epithelial cells undergo apoptosis (contributing to ulceration) or survive (maintaining barrier integrity).
Epigenetic Effects of NF-kB Modulation
Chronic NF-kB activation can produce lasting changes in gene expression through epigenetic modifications, including DNA methylation, histone acetylation, and chromatin remodeling. These epigenetic changes can persist even after the initial inflammatory stimulus is removed, creating a state of "trained immunity" or "inflammatory memory" that predisposes tissue to exaggerated inflammatory responses upon subsequent stimulation. This phenomenon may contribute to the relapsing nature of IBD, where disease flares occur repeatedly even in the absence of identifiable triggers.
NF-kB p65 interacts with histone acetyltransferases (HATs) such as CBP/p300, promoting histone H3 and H4 acetylation at inflammatory gene promoters. This opens chromatin structure and increases transcriptional accessibility, effectively creating an epigenetic "bookmark" that facilitates rapid re-expression of inflammatory genes upon subsequent NF-kB activation. Chronically inflamed tissue accumulates these epigenetic marks, becoming progressively more responsive to inflammatory stimuli.
By suppressing NF-kB nuclear activity, KPV could reduce the accumulation of pro-inflammatory epigenetic marks during active inflammation. Over time, this could help "reset" the epigenetic state of chronically inflamed tissue, reducing its propensity for exaggerated inflammatory responses and potentially extending remission periods between disease flares. While this theoretical benefit has not been experimentally verified for KPV, it represents a compelling rationale for early and sustained anti-inflammatory intervention in conditions like IBD.
The epigenetic dimension also has implications for the timing of KPV therapy. Starting treatment early in the course of an inflammatory disease, before extensive epigenetic reprogramming has occurred, might provide more lasting benefit than treatment initiated after years of chronic inflammation have established deeply entrenched inflammatory epigenetic patterns. This consideration supports the potential use of KPV as an early intervention rather than a last-resort option, though clinical data to support this strategy do not yet exist.
Metabolic Consequences of NF-kB Inhibition
NF-kB signaling has important metabolic functions beyond inflammation, and the metabolic consequences of KPV-mediated NF-kB inhibition deserve consideration. In hepatocytes, NF-kB regulates glucose metabolism, lipid metabolism, and insulin sensitivity. In adipose tissue, NF-kB activation promotes insulin resistance and systemic inflammation. In skeletal muscle, NF-kB contributes to inflammation-induced muscle wasting (cachexia).
In the context of IBD, metabolic complications are common and clinically significant. Malnutrition, weight loss, and micronutrient deficiencies affect a large proportion of IBD patients. Chronic systemic inflammation contributes to insulin resistance, bone loss, and cardiovascular risk. By reducing systemic inflammatory cytokine levels, KPV could potentially improve metabolic parameters in IBD patients, contributing to overall health beyond direct gut effects.
For individuals exploring metabolic optimization alongside anti-inflammatory therapy, several complementary approaches exist. 5-Amino-1MQ addresses metabolic regulation through NNMT inhibition, while MOTS-c supports mitochondrial function and metabolic homeostasis. AOD-9604 and Fragment 176-191 target lipid metabolism specifically. These compounds address different aspects of metabolic health and could theoretically complement KPV's anti-inflammatory effects in patients with metabolic comorbidities.
The relationship between gut inflammation and systemic metabolism is increasingly recognized as bidirectional. Metabolic endotoxemia - the translocation of bacterial lipopolysaccharide from the gut lumen to the systemic circulation through a compromised intestinal barrier - is a major driver of systemic inflammation and insulin resistance. KPV's dual action on gut inflammation (reducing the inflammatory driver of barrier breakdown) and barrier integrity (directly preserving tight junctions) could reduce metabolic endotoxemia and its downstream metabolic consequences. This potential systemic metabolic benefit, mediated through improved gut barrier function, represents an underappreciated dimension of KPV's therapeutic potential.
Neuroinflammation and Cognitive Applications
Alpha-MSH and its fragments have demonstrated anti-neuroinflammatory effects in multiple experimental models, raising the question of whether KPV could benefit neurological conditions characterized by chronic inflammation. NF-kB activation in microglia (the brain's resident macrophages) drives neuroinflammation in conditions including Alzheimer's disease, Parkinson's disease, multiple sclerosis, and traumatic brain injury.
The primary challenge for neurological applications of KPV is blood-brain barrier (BBB) penetration. The BBB severely restricts the entry of hydrophilic molecules, including peptides, from the bloodstream into the central nervous system. PepT1 is not significantly expressed on BBB endothelial cells under normal conditions, though its expression may increase during neuroinflammation. Whether systemically administered KPV can reach therapeutic concentrations in the brain parenchyma is unknown.
Alternative delivery strategies for brain targeting include intranasal administration, which bypasses the BBB through olfactory and trigeminal nerve pathways, and nanoparticle encapsulation with surface modifications that facilitate BBB transcytosis. Intranasal delivery is particularly interesting for KPV because the nasal mucosa expresses PepT1, potentially enabling efficient absorption of the tripeptide from the nasal cavity with subsequent transport along neural pathways to the brain.
For those interested in neuroprotective and cognitive-enhancing peptides, FormBlends offers several relevant compounds. Semax is a synthetic ACTH fragment with documented nootropic and neuroprotective effects. Selank modulates anxiety and cognitive function through GABAergic mechanisms. Dihexa promotes neuronal growth factor signaling. P21 enhances neurogenesis and synaptic plasticity. Pinealon supports pineal gland function and circadian regulation. And NAD+ supplementation addresses cellular energy metabolism that declines with aging and neurodegeneration. Each of these approaches targets different aspects of brain health and could potentially complement KPV's anti-neuroinflammatory effects if adequate brain delivery can be achieved.
KPV in the Context of the Broader Peptide Therapy Landscape
KPV occupies a specific therapeutic niche within the growing field of peptide therapeutics. Understanding its position relative to other peptides helps clinicians and patients make informed choices about peptide-based interventions.
Among anti-inflammatory peptides, KPV is distinguished by its NF-kB-specific mechanism, PepT1-mediated targeting, and lack of receptor dependence. BPC-157 addresses inflammation through nitric oxide modulation, growth factor expression, and angiogenesis, making it complementary to KPV rather than competing. Thymosin Alpha-1 works through T cell modulation and innate immune priming, providing immune regulation at a different level than KPV's cytokine suppression. LL-37 combines antimicrobial and immunomodulatory properties, overlapping with KPV's anti-inflammatory effects but adding direct pathogen-killing capacity.
Among gut-targeting peptides, KPV's PepT1-mediated colonic delivery distinguishes it from most therapeutic peptides that require parenteral administration to reach their targets. Larazotide also works in the gut lumen but targets tight junctions rather than inflammatory signaling. VIP (Vasoactive Intestinal Peptide) has anti-inflammatory effects in the gut but works through VIP receptors and requires parenteral delivery.
Among skin-targeting peptides, KPV's anti-inflammatory focus differs from the regenerative and anti-aging mechanisms of GHK-Cu, the neuromuscular relaxation of SNAP-8, and the collagen-stimulating effects of Matrixyl. These peptides address skin health through different mechanisms and can be combined with KPV for comprehensive dermatological protocols.
The drug comparison hub provides detailed head-to-head comparisons of various peptide compounds, helping individuals identify the most appropriate options for their specific health goals. And the biohacking hub offers guidance on integrating peptide therapies into broader health optimization strategies.
Regulatory Landscape and Access
KPV exists in a regulatory gray area that affects its accessibility and clinical use. As a naturally occurring tripeptide fragment of an endogenous hormone, it does not fall neatly into the FDA's drug approval framework. It is not currently approved as a pharmaceutical drug for any indication, nor is it classified as a dietary supplement in the traditional sense.
In the United States, KPV is available through compounding pharmacies when prescribed by licensed healthcare providers, and through research peptide suppliers for investigational use. The FDA's regulatory oversight of compounded peptides has increased in recent years, with greater scrutiny of compounding pharmacy practices and quality standards. This regulatory attention, while creating some access challenges, ultimately benefits patients by ensuring higher quality and safety standards for compounded peptide products.
The regulatory path to FDA approval for KPV as a pharmaceutical drug would require the standard sequence of IND (Investigational New Drug) application, Phase I safety studies, Phase II efficacy studies, and Phase III confirmatory trials. Given the compound's lack of patent protection (as a natural tripeptide), the pharmaceutical industry may be reluctant to invest the hundreds of millions of dollars required for this development path without exclusivity guarantees. The Orphan Drug Act could provide limited exclusivity for rare disease indications, but IBD is not classified as a rare disease in the United States.
Novel formulations of KPV, such as the HA-KPV-NP delivery system, do carry patent protection and could provide the commercial incentive needed for formal clinical development. The combination of a well-characterized, safe active ingredient (KPV) with a proprietary delivery system (HA-KPV-NPs) represents an attractive development package that could attract pharmaceutical or biotech company investment. Alternatively, academic clinical trials funded by government grants or charitable foundations could provide the clinical evidence needed to establish KPV's therapeutic role, even without commercial development.
For individuals seeking access to KPV currently, FormBlends offers KPV products with certificate of analysis documentation and quality assurance standards. The GLP-1 weight loss overview page provides context on the broader therapeutic landscape, while the free assessment helps individuals determine the most appropriate peptide approach for their specific health concerns.
Gut Inflammation Research
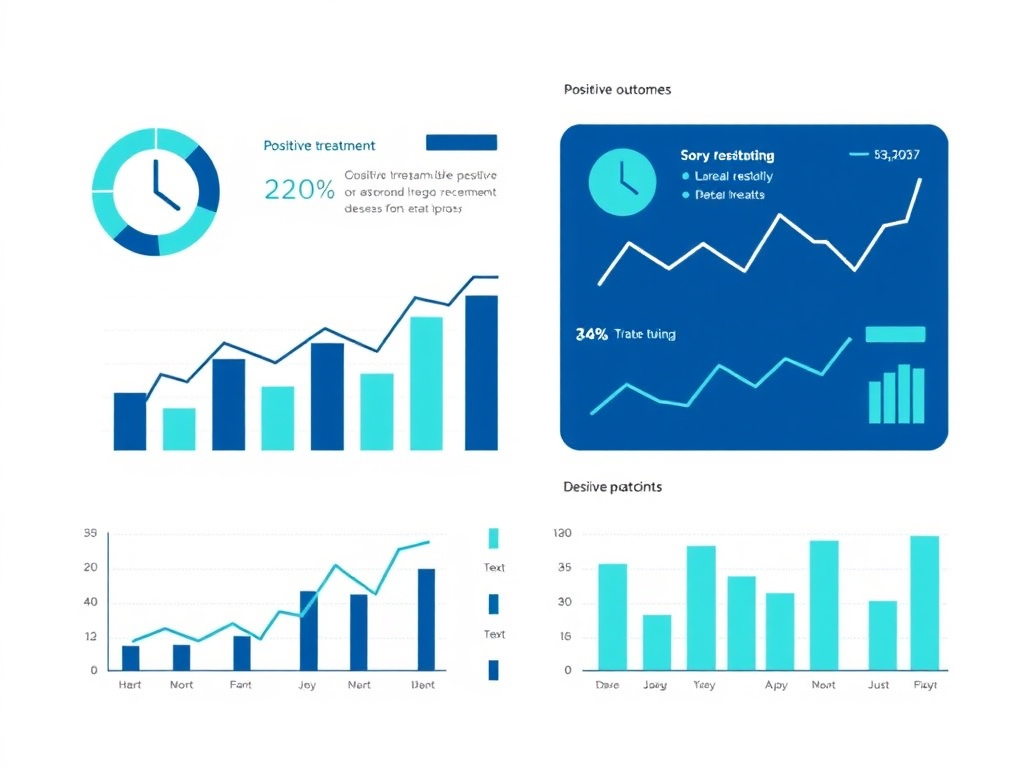
Figure 4: Colonic inflammation scores in preclinical models comparing healthy controls, colitis models, and KPV-treated animals
Gut inflammation represents the most extensively studied therapeutic application of KPV. Preclinical research spanning over two decades has demonstrated that KPV reduces intestinal inflammation through multiple complementary mechanisms, including direct suppression of epithelial and immune cell inflammatory signaling, preservation of mucosal barrier integrity, and reduction of inflammatory cell infiltration into colonic tissue. These effects have been documented across multiple animal models of colitis using both oral and injectable KPV administration routes.
The PepT1 Connection: Why the Gut Is a Prime Target
KPV's therapeutic potential for gut inflammation is uniquely supported by the biology of its cellular uptake mechanism. PepT1, the proton-coupled oligopeptide transporter that mediates KPV entry into cells, is constitutively expressed on the apical (luminal-facing) membrane of small intestinal enterocytes, where it normally functions to absorb dietary di- and tripeptides. Under healthy conditions, PepT1 expression in the colon is minimal. But in inflammatory bowel disease, colonic PepT1 expression increases dramatically.
This disease-induced upregulation was first described in patients with Crohn's disease and ulcerative colitis, where immunohistochemical staining revealed strong PepT1 expression on inflamed colonic epithelium that was absent in adjacent non-inflamed tissue and in healthy controls. The functional consequence is profound: inflamed colonic tissue develops the capacity to absorb KPV directly from the intestinal lumen, concentrating the peptide precisely at sites where anti-inflammatory activity is needed most.
This natural targeting mechanism has several advantages over conventional drug delivery. First, it reduces the systemic dose required for therapeutic effect, since KPV is preferentially absorbed by inflamed rather than healthy tissue. Second, it creates a self-regulating system - as inflammation resolves and PepT1 expression decreases, KPV uptake also decreases, reducing the risk of over-suppression of local immune function. Third, it supports oral administration, the most convenient route for chronic conditions requiring long-term treatment. The KPV product page provides additional information on available formulations designed to exploit this mechanism.
DSS-Induced Colitis Studies
The dextran sodium sulfate (DSS) colitis model is one of the most widely used preclinical systems for studying inflammatory bowel disease. DSS is administered in drinking water, where it damages the colonic epithelial barrier, allowing luminal bacteria and their products to enter the underlying tissue and trigger an acute inflammatory response. The resulting colitis closely mimics the mucosal inflammation, immune cell infiltration, and tissue damage seen in human ulcerative colitis.
In the foundational study published in Inflammatory Bowel Diseases in 2008, Laroui and colleagues evaluated KPV in mice with DSS-induced colitis. Animals received DSS in their drinking water for 7 days to establish colitis, followed by KPV treatment. The results were striking. KPV-treated mice showed earlier recovery from colitis compared to untreated controls, with significantly stronger regain of body weight during the recovery phase. Histological scoring of colonic tissue revealed markedly reduced inflammatory infiltration, less epithelial damage, and better preservation of crypt architecture in KPV-treated animals.
Myeloperoxidase (MPO) activity, a quantitative measure of neutrophil infiltration into tissue, was significantly reduced in the colonic tissue of KPV-treated mice. Since neutrophils are major contributors to tissue damage during acute colitis - releasing reactive oxygen species, proteases, and inflammatory cytokines - their reduced presence in KPV-treated tissue directly explains the histological improvements observed. Colonic levels of pro-inflammatory cytokines including TNF-alpha and IL-6 were also significantly lower in the KPV group.
Colonic Inflammation Score Comparison
Data from preclinical DSS colitis models. Inflammation scored on 0-50 scale based on histological assessment of epithelial damage, immune infiltration, and tissue architecture disruption.
The chart above illustrates the dramatic reduction in colonic inflammation score achieved with KPV treatment. Healthy control animals scored approximately 2 on the 0-50 inflammation scale, reflecting the baseline level of physiological immune activity in healthy colonic tissue. DSS colitis raised this score to approximately 42, reflecting severe mucosal inflammation with extensive epithelial damage and immune cell infiltration. KPV treatment reduced the score to approximately 12, representing a 71% reduction in inflammation compared to untreated colitis - a result that approaches the efficacy of established anti-inflammatory agents in the same model system.
TNBS-Induced Colitis Studies
The TNBS (2,4,6-trinitrobenzenesulfonic acid) colitis model provides a complementary perspective on KPV's gut anti-inflammatory activity. Unlike DSS colitis, which primarily models mucosal inflammation resembling ulcerative colitis, TNBS colitis involves T cell-mediated transmural inflammation that more closely mimics Crohn's disease. The TNBS hapten triggers a delayed-type hypersensitivity reaction when applied to the colonic mucosa in combination with ethanol (which disrupts the mucosal barrier to allow tissue penetration).
KPV demonstrated significant anti-inflammatory effects in the TNBS model as well. Oral KPV administration reduced disease activity scores, which incorporate measures of body weight loss, stool consistency, and rectal bleeding. Histological assessment showed reduced transmural inflammation, less granuloma formation, and better preservation of the muscularis propria compared to untreated animals. These findings are particularly important because TNBS colitis is driven primarily by adaptive immune mechanisms (T helper 1 and T helper 17 responses), while DSS colitis involves predominantly innate immune activation. KPV's efficacy in both models indicates that it suppresses inflammation regardless of whether the driving mechanism is innate or adaptive immunity.
The dual-model efficacy also strengthens the case for KPV as a potential therapeutic for both major forms of IBD in humans. Ulcerative colitis and Crohn's disease, while both classified as inflammatory bowel diseases, involve different immune pathways, affect different regions of the gastrointestinal tract, and respond differently to various treatments. An agent effective against both types of experimental colitis has a higher probability of translating to human efficacy across the IBD spectrum. For those exploring complementary gut-healing compounds, BPC-157 has also demonstrated protective effects in experimental colitis models through distinct mechanisms.
Intestinal Barrier Integrity
Beyond suppressing inflammatory signaling, KPV helps preserve and restore the intestinal epithelial barrier - a critical function that is often disrupted in inflammatory bowel disease. The intestinal barrier consists of a single layer of epithelial cells connected by tight junctions, adherens junctions, and desmosomes. This barrier separates the intestinal lumen (containing bacteria, food antigens, and digestive enzymes) from the underlying immune-rich lamina propria. When the barrier breaks down, luminal contents enter the tissue and trigger an inflammatory response that further damages the barrier, creating a vicious cycle of barrier disruption and inflammation.
KPV protects the intestinal barrier through several mechanisms. By suppressing NF-kB activation in epithelial cells, it reduces the expression of matrix metalloproteinases (MMPs) that degrade the extracellular matrix and disrupt cell-cell adhesion. KPV also promotes the expression and proper localization of tight junction proteins including claudins, occludin, and zonula occludens-1 (ZO-1). In cell culture models using polarized monolayers of Caco-2 cells (a human intestinal epithelial cell line), KPV treatment maintained transepithelial electrical resistance (TEER) - a measure of barrier integrity - following inflammatory challenge with TNF-alpha or interferon-gamma.
The barrier-protective effects of KPV are complemented by its ability to promote epithelial cell survival. NF-kB activation during inflammation can promote either cell survival or apoptosis depending on the context, and in the intestinal epithelium, excessive inflammation tips the balance toward apoptosis, leading to epithelial erosion and ulceration. KPV's modulation of NF-kB appears to favor cell survival pathways while suppressing inflammatory gene expression, an effect that helps maintain epithelial coverage during active inflammation. Those interested in barrier integrity and gut healing may also want to explore larazotide, a peptide that directly modulates tight junction function.
Microbiome Interactions
Emerging research suggests that KPV's anti-inflammatory effects in the gut may be partly mediated through changes in the intestinal microbiome. Chronic inflammation disrupts the composition and diversity of the gut bacterial community - a state known as dysbiosis - which in turn perpetuates inflammation through increased production of pro-inflammatory metabolites and reduced production of protective short-chain fatty acids (SCFAs) like butyrate.
In DSS colitis models, KPV treatment was associated with partial restoration of microbial diversity and a shift in the bacterial community composition toward a healthier profile. Specifically, KPV-treated mice showed increased abundance of Lactobacillus and Bifidobacterium species, both associated with anti-inflammatory effects and SCFA production, and decreased abundance of pro-inflammatory taxa such as Enterobacteriaceae. While these changes could be secondary to reduced inflammation rather than a direct effect of KPV on the bacteria, they nonetheless contribute to the overall therapeutic benefit by helping break the inflammation-dysbiosis cycle.
The relationship between KPV and the microbiome also has implications for delivery. Oral KPV must survive transit through the stomach and small intestine to reach the colon, where the majority of the gut microbiome resides and where IBD-related inflammation typically occurs. The acidic environment of the stomach (pH 1.5-3.5) and the proteolytic enzymes of the small intestine (pepsin, trypsin, chymotrypsin) can degrade the peptide before it reaches its target. This challenge has driven the development of protective delivery systems, discussed in detail in the oral delivery section of this report.
Dose-Response Relationships in Gut Models
Preclinical studies have established clear dose-response relationships for KPV in gut inflammation models. In cell culture experiments, effective concentrations range from 10 nanomolar to 10 micromolar, with optimal NF-kB inhibition typically observed at 100 nanomolar to 1 micromolar. Higher concentrations do not produce proportionally greater anti-inflammatory effects, suggesting a saturable mechanism consistent with transporter-mediated uptake.
In animal models, oral KPV doses of 20-50 micrograms per day in mice (equivalent to approximately 1 mg/kg body weight) produced significant reductions in colitis severity. When administered as HA-KPV-NPs (hyaluronic acid-functionalized nanoparticles), the effective dose was reduced dramatically. The nanoparticle delivery system achieved equivalent anti-inflammatory effects at KPV concentrations 12,000-fold lower than free peptide, meaning that nanogram rather than microgram quantities were sufficient for therapeutic benefit. This extraordinary improvement in potency through targeted delivery highlights the potential for practical, cost-effective clinical formulations.
Subcutaneous KPV administration has also been evaluated in gut inflammation models, though less extensively than oral dosing. Systemic delivery provides the advantage of bypassing gastrointestinal degradation but loses the benefit of direct colonic targeting via PepT1. In practice, subcutaneous KPV appears to be effective for gut inflammation at doses comparable to oral administration, suggesting that circulating KPV can reach inflamed colonic tissue through the vasculature in addition to the luminal route. The dosing calculator can help individuals estimate appropriate doses based on body weight and target condition.
IBD & Colitis Studies

Figure 5: Summary of KPV peptide research outcomes in inflammatory bowel disease models
Inflammatory bowel disease affects approximately 6.8 million people worldwide, with rising incidence in newly industrialized countries. Current treatments - aminosalicylates, corticosteroids, immunomodulators, and biologic therapies - provide benefit for many patients but carry significant side effects and lose efficacy over time in a substantial proportion of cases. KPV represents a novel therapeutic approach that addresses IBD pathophysiology through mechanisms distinct from all currently approved treatments, with early evidence suggesting a more favorable safety profile.
Ulcerative Colitis: Pathophysiology and KPV Intervention Points
Ulcerative colitis (UC) is characterized by continuous mucosal inflammation extending proximally from the rectum. The inflammation is confined to the mucosa and submucosa, sparing the deeper muscular layers, and involves a predominant T helper 2 (Th2) immune response with contributions from innate immune pathways. Key pathological features include crypt abscesses, goblet cell depletion, pseudopolyp formation, and mucosal ulceration.
KPV addresses multiple elements of UC pathophysiology simultaneously. At the epithelial level, it reduces NF-kB-driven expression of pro-inflammatory cytokines and chemokines that recruit and activate immune cells. At the immune cell level, it suppresses the production of TNF-alpha, IL-1beta, and IL-6 by mucosal macrophages and dendritic cells, reducing the amplification of the inflammatory cascade. At the barrier level, it preserves tight junction integrity and promotes epithelial cell survival, helping to maintain the physical separation between luminal bacteria and the mucosal immune system.
The PepT1-mediated uptake pathway is particularly relevant to UC because PepT1 expression is upregulated throughout the inflamed colonic mucosa. In patients with active UC, immunohistochemical studies have shown PepT1 expression on the apical surface of colonocytes in inflamed segments, with minimal expression in uninvolved segments. This creates a natural gradient of KPV uptake that favors accumulation in the most severely affected tissue. The enhanced PepT1 expression in UC also means that oral KPV reaching the colon is efficiently absorbed into the epithelium where it can exert its intracellular anti-inflammatory effects.
Crohn's Disease: Transmural Inflammation and KPV
Crohn's disease (CD) differs from UC in several important ways that affect the potential role of KPV therapy. CD can affect any part of the gastrointestinal tract from mouth to anus, involves transmural (full-thickness) inflammation, and is characterized by a predominant T helper 1 (Th1) and T helper 17 (Th17) immune response with prominent granuloma formation. The skip-lesion pattern of CD, with alternating segments of diseased and healthy tissue, creates unique challenges for drug delivery.
KPV's efficacy in the TNBS colitis model - which recapitulates the T cell-mediated, transmural inflammation of CD - provides preliminary evidence that the peptide may benefit CD patients as well as those with UC. The key question is whether KPV can reach transmural inflammatory sites when absorbed luminally through PepT1, since PepT1 is an apical transporter that delivers KPV into epithelial cells rather than into the deeper tissue layers where CD inflammation occurs.
Several mechanisms could mediate KPV's effects on transmural inflammation. First, PepT1 is expressed on immune cells including macrophages and T lymphocytes that migrate between the epithelium and the lamina propria. KPV uptake by these mobile immune cells could carry the peptide's anti-inflammatory effects into deeper tissue. Second, KPV-induced suppression of epithelial chemokine production reduces the signals that recruit inflammatory cells from the bloodstream to the tissue, indirectly reducing transmural inflammation even without direct delivery to the muscularis. Third, systemically absorbed KPV (whether from oral or subcutaneous administration) reaches the vascular supply of the bowel wall and can access inflamed tissue through the microvasculature.
Comparison with Current IBD Therapies
Understanding KPV's potential role in IBD management requires comparison with the current treatment landscape. The following table summarizes key differences between KPV and established IBD therapies.
| Therapy Class | Examples | Mechanism | Response Rate | Key Limitations |
|---|---|---|---|---|
| 5-Aminosalicylates | Mesalamine, sulfasalazine | Topical anti-inflammatory (NF-kB, PPAR-gamma) | 40-60% in mild UC | Limited efficacy in moderate-severe disease |
| Corticosteroids | Prednisone, budesonide | Broad immunosuppression via GR | 60-80% for induction | Not suitable for maintenance; metabolic toxicity |
| Thiopurines | Azathioprine, 6-MP | Purine synthesis inhibition | 40-50% steroid-sparing | Myelosuppression, hepatotoxicity, lymphoma risk |
| Anti-TNF biologics | Infliximab, adalimumab | TNF-alpha neutralization | 50-70% induction | Immunogenicity, infection risk, loss of response |
| Anti-integrins | Vedolizumab | Gut-selective leukocyte trafficking | 40-55% induction | Slow onset, PML risk (natalizumab) |
| JAK inhibitors | Tofacitinib, upadacitinib | JAK-STAT pathway inhibition | 45-60% induction | Thrombosis, infection, herpes zoster |
| KPV (preclinical) | KPV, HA-KPV-NPs | NF-kB + MAPK inhibition via PepT1 | 71% inflammation reduction (preclinical) | No human trials completed; limited long-term data |
The comparison reveals several potential advantages of KPV. First, its mechanism of action is distinct from all currently approved IBD therapies, suggesting it could benefit patients who have failed or become refractory to existing treatments. Second, its targeted delivery via PepT1 provides gut selectivity without the need for antibody-based targeting, potentially reducing both cost and immunogenicity concerns. Third, its favorable preclinical safety profile, if confirmed in human studies, could make it suitable for long-term maintenance therapy where the toxicity of current agents limits their use.
However, it is essential to acknowledge KPV's current limitations. All efficacy data come from preclinical models, and the history of IBD drug development shows that many promising preclinical candidates fail in human trials. The translation from mouse colitis models to human IBD is imperfect, and human-specific factors such as the complexity and chronicity of established IBD, the influence of diet and lifestyle, and the heterogeneity of patient populations may affect KPV's therapeutic potential in ways that preclinical studies cannot predict.
Nanoparticle-Based Approaches for IBD
The most advanced formulation strategy for KPV in IBD involves hyaluronic acid-functionalized nanoparticles (HA-KPV-NPs), developed by Xiao and colleagues and published in Molecular Therapy in 2017. This system addresses two major challenges in oral KPV delivery for IBD: protecting the peptide from gastrointestinal degradation and targeting it specifically to inflamed colonic tissue.
The nanoparticles consist of a poly(lactic acid)-poly(ethylene glycol) (PLA-PEG) core loaded with KPV and coated with hyaluronic acid (HA). The PLA-PEG core protects the peptide from enzymatic degradation and controls its release rate. The HA coating serves two functions: it provides mucoadhesive properties that promote retention in the colonic lumen, and it targets CD44, a cell surface receptor that is upregulated on inflamed colonic epithelial cells and activated macrophages. This CD44-mediated targeting adds a second layer of selectivity beyond PepT1-dependent uptake.
The HA-KPV-NPs are further encapsulated in a chitosan-alginate hydrogel for oral administration. This hydrogel is stable at gastric pH (1.5-3.5) and in the small intestinal environment, but dissolves at the slightly alkaline pH of the colon (6.5-7.5), releasing the nanoparticles at the target site. The entire system provides three sequential levels of targeting: pH-responsive release in the colon, HA-mediated attachment to inflamed tissue, and PepT1-mediated uptake into epithelial and immune cells.
In DSS colitis mice, oral HA-KPV-NP/hydrogel treatment significantly reduced disease activity scores, colonic inflammation, and mucosal damage. TNF-alpha expression in colonic tissue was markedly lower in treated animals. The effective dose of KPV delivered via nanoparticles was approximately 12,000-fold lower than the dose of free KPV required for equivalent anti-inflammatory effect - a result that reflects the combined benefits of protection from degradation, targeted delivery, and controlled release. This dose reduction has major implications for clinical development, potentially enabling therapeutic KPV delivery in practical oral dosage forms.
Combination Approaches with Other Peptides
The distinct mechanism of KPV raises the possibility of combining it with other anti-inflammatory or gut-protective agents for enhanced efficacy. Several combinations have been explored or proposed in the literature.
KPV combined with BPC-157 represents a theoretically attractive pairing. BPC-157 promotes angiogenesis and tissue repair while modulating nitric oxide pathways, complementing KPV's anti-inflammatory mechanism. Where KPV suppresses the inflammatory damage, BPC-157 could accelerate the healing of damaged mucosa. This combination has been used anecdotally in integrative medicine practices, though formal preclinical evaluation of the combination has not been published.
The combination of KPV with larazotide, a tight junction modulator, addresses complementary aspects of IBD pathophysiology. Larazotide directly prevents the opening of tight junctions triggered by zonulin and other permeability factors, while KPV reduces the inflammatory signaling that drives barrier breakdown from within the epithelial cells. Together, they could provide more complete barrier protection than either agent alone.
For systemic inflammatory conditions that extend beyond the gut, combining KPV with Thymosin Alpha-1 offers a dual approach to immune modulation. Thymosin Alpha-1 enhances regulatory T cell function and promotes immune tolerance, while KPV directly suppresses inflammatory signaling. This combination could be particularly relevant for patients with IBD-associated extraintestinal manifestations such as arthritis, uveitis, or pyoderma gangrenosum.
Clinical Translation Challenges
Moving KPV from preclinical success to clinical application faces several challenges that merit honest discussion. First, the regulatory pathway for KPV is complicated by its status as a peptide fragment of an endogenous hormone. It does not fit neatly into existing drug categories, and regulatory agencies may require novel clinical trial designs to evaluate its safety and efficacy appropriately.
Second, the lack of intellectual property protection for a natural tripeptide may limit commercial interest from pharmaceutical companies that typically require patent exclusivity to justify the investment in clinical development. The HA-KPV-NP delivery system does carry patent protection, which may incentivize its clinical development, but the base molecule itself is not patentable.
Third, the optimal dosing, route, and duration of KPV treatment for human IBD remain to be established. Preclinical dose-response relationships provide starting points for human dose selection, but species differences in PepT1 expression, intestinal physiology, and immune function require careful dose-finding studies in Phase I and Phase II trials. Those interested in tracking the clinical development of KPV and related peptides can follow updates through the peptide research hub, which covers the latest developments in therapeutic peptide research.
Skin Inflammation

Figure 6: KPV research in skin inflammatory conditions including psoriasis, dermatitis, and wound healing
KPV demonstrates significant anti-inflammatory activity in skin tissue, building on the well-established role of alpha-MSH in cutaneous immune regulation. The skin produces alpha-MSH locally through keratinocyte and melanocyte POMC expression, creating an endogenous anti-inflammatory system that KPV can supplement. Topical, transdermal, and injectable KPV formulations have shown promise in preclinical models of psoriasis, atopic dermatitis, contact dermatitis, and fine particulate matter-induced skin damage.
Alpha-MSH in Cutaneous Biology
The skin is a major neuroendocrine organ that produces and responds to a wide range of neuropeptides, including alpha-MSH. Keratinocytes, melanocytes, Langerhans cells, dermal fibroblasts, and endothelial cells in the skin all express POMC and process it into alpha-MSH and its fragments, including KPV. This local production creates an autocrine and paracrine signaling network that modulates inflammation, pigmentation, and wound healing in the skin.
In the context of inflammation, cutaneous alpha-MSH production increases in response to UV radiation, tissue injury, and pro-inflammatory cytokines. The released alpha-MSH acts on MC1R-expressing cells in the skin to suppress inflammatory responses, promote wound healing, and protect against UV-induced DNA damage. Individuals with loss-of-function MC1R variants (common in those with red hair and fair skin) show enhanced susceptibility to sunburn, inflammatory skin conditions, and skin cancer, underscoring the importance of melanocortin signaling in cutaneous immune homeostasis.
KPV, as the minimal anti-inflammatory fragment of alpha-MSH, can supplement this endogenous system without triggering the pigmentary effects mediated by MC1R. This is a critical distinction for dermatological applications: while increased melanin production may be desirable in some contexts (e.g., protection against UV damage), it is an unwanted side effect for patients seeking pure anti-inflammatory therapy for conditions like eczema or psoriasis. By bypassing melanocortin receptors entirely, KPV provides anti-inflammatory benefits without altering skin pigmentation.
Psoriasis Research
Psoriasis is a chronic autoimmune skin condition characterized by keratinocyte hyperproliferation, epidermal thickening, and inflammatory infiltration of the skin. The disease is driven by a dysregulated immune response involving Th1 and Th17 cells that produce inflammatory cytokines including TNF-alpha, IL-17, IL-22, and IL-23. These cytokines activate keratinocytes to produce additional chemokines and proliferation signals, creating a self-sustaining inflammatory loop.
KPV addresses psoriasis pathophysiology at multiple points. By inhibiting NF-kB in keratinocytes, it reduces the production of chemokines (IL-8, CXCL10, CCL20) that recruit Th1 and Th17 cells to the skin. By suppressing NF-kB in infiltrating immune cells, it reduces the production of the effector cytokines (TNF-alpha, IL-17) that drive keratinocyte hyperproliferation. And by modulating MAPK signaling, it may directly influence keratinocyte growth and differentiation, promoting a more normal epidermal phenotype.
A US patent application (US20020183255A1) describes the use of KPV for dermatological disorders including psoriasis, reporting that in a subject with over ten years of diagnosed psoriasis, KPV treatment reduced inflammation, scaling, dryness, pain, erythema, and pruritis. While patent applications are not peer-reviewed and provide limited experimental detail, this disclosure supports the therapeutic potential of KPV for psoriasis and suggests that clinical observations have been made in at least some patients.
Current standard treatments for psoriasis include topical corticosteroids, vitamin D analogs, phototherapy, and systemic agents (methotrexate, cyclosporine, biologics). Each has significant limitations. Topical steroids cause skin atrophy with chronic use. Biologics are expensive and carry infection risks. KPV could potentially fill a niche as a topical anti-inflammatory agent without the side effects of corticosteroids, suitable for long-term use on sensitive areas like the face, intertriginous regions, and genitalia. For those interested in topical peptide applications, GHK-Cu topical offers complementary skin-healing and anti-aging properties through copper peptide mechanisms.
Atopic Dermatitis
Atopic dermatitis (eczema) is a chronic inflammatory skin condition affecting up to 20% of children and 10% of adults worldwide. It is characterized by impaired epidermal barrier function, Th2-skewed immune activation, and intense pruritis. The defective barrier allows allergens and microbial products to penetrate the skin, triggering immune responses that further damage the barrier in a cycle of inflammation and sensitization.
KPV's potential in atopic dermatitis rests on several mechanistic considerations. Its NF-kB inhibitory activity suppresses the production of thymic stromal lymphopoietin (TSLP), IL-25, and IL-33 by keratinocytes - three epithelial cytokines that drive the Th2 response in atopic dermatitis. Reducing these "alarmins" could interrupt the initiation of the allergic inflammatory cascade before it fully develops. Additionally, KPV's barrier-protective effects, demonstrated in intestinal epithelial models, may translate to the skin, where maintaining epidermal barrier integrity is central to disease management.
The small molecular weight of KPV (342 daltons) facilitates topical delivery, as molecules below 500 daltons can generally penetrate the stratum corneum - the outermost skin layer that serves as the primary barrier to drug absorption. However, the intact stratum corneum still presents a significant obstacle to peptide delivery, and most topical peptide formulations require penetration enhancers or physical disruption of the barrier to achieve therapeutic concentrations in the dermis.
Transdermal Delivery Advances
Research published in the Journal of Pharmaceutical Sciences by Vemulapalli and colleagues evaluated transdermal iontophoretic delivery of KPV across microporated human skin. Microporation creates microscopic channels in the stratum corneum using laser or radiofrequency energy, while iontophoresis uses a small electric current to drive charged molecules through the skin. The combination achieved significantly higher KPV delivery rates than passive diffusion alone.
The study demonstrated that iontophoretic delivery through microporated skin produced KPV flux rates sufficient to achieve therapeutic concentrations in the dermal compartment. The positive charge on the lysine residue of KPV at physiological pH makes it amenable to anodal iontophoresis (delivery from the positive electrode), which drives cationic molecules into the skin along the electric field. This physical delivery approach bypasses the enzymatic and barrier challenges that limit passive topical application.
Other formulation approaches for topical KPV include liposomal encapsulation, nanoparticle suspension in cream or gel bases, and incorporation into dissolving microneedle patches. Each approach addresses the challenge of delivering a hydrophilic tripeptide across the hydrophobic stratum corneum. The optimal formulation likely depends on the target condition and site of application: creams and gels for widespread conditions like eczema, patches for localized lesions like psoriatic plaques, and iontophoretic devices for deeper delivery requirements.
Environmental Skin Damage
A 2025 study published in ScienceDirect expanded KPV's potential applications to environmental skin damage. Researchers investigated KPV's effects on human keratinocytes exposed to PM10 (particulate matter with diameter less than 10 micrometers), a major component of air pollution that causes skin inflammation, oxidative stress, and accelerated aging.
PM10 exposure induced keratinocyte apoptosis, increased IL-1beta secretion, and activated both MAPK and NF-kB inflammatory pathways. Treatment with KPV at 50 micrograms per milliliter restored cell viability, reduced IL-1beta secretion to near-baseline levels, and suppressed the activation of both inflammatory signaling cascades. The study provided evidence that KPV's anti-inflammatory mechanism is effective against pollution-induced skin damage, not just classical immune-mediated conditions.
This finding has practical implications for cosmeceutical applications. Air pollution is a growing concern for skin health in urban populations worldwide, contributing to premature aging, hyperpigmentation, and inflammatory skin conditions. A topical KPV formulation that protects against pollution-induced inflammation could serve as a preventive measure in addition to its therapeutic applications. For complementary anti-aging peptide approaches, GHK-Cu and SNAP-8 address skin aging through different mechanisms that could complement KPV's anti-inflammatory effects.
Wound Healing Applications
Alpha-MSH and its fragments, including KPV, have demonstrated wound healing-promoting activity in multiple preclinical studies. The connection between anti-inflammation and wound healing is direct: while the initial inflammatory phase of wound healing is necessary for pathogen clearance and debris removal, prolonged or excessive inflammation delays the transition to the proliferative and remodeling phases. By resolving inflammation more quickly, KPV may accelerate overall wound healing.
In experimental wound models, alpha-MSH treatment increased the rate of wound closure, promoted collagen deposition, and improved the organization of newly formed tissue. While these studies primarily used the full-length hormone rather than the KPV fragment, the shared anti-inflammatory mechanism suggests that KPV would provide similar benefits. Additionally, KPV's lack of pigmentary effects could be an advantage in wound healing applications, where post-inflammatory hyperpigmentation is a common cosmetic concern, particularly in individuals with darker skin tones.
Chronic wounds, including diabetic ulcers, venous stasis ulcers, and pressure injuries, represent a major clinical challenge where inflammation-modulating peptides could provide significant benefit. These wounds are characterized by a "stalled" inflammatory phase with elevated levels of TNF-alpha, IL-1beta, MMPs, and reactive oxygen species that prevent normal healing progression. KPV's ability to reduce these mediators while promoting epithelial cell survival positions it as a potential adjunctive therapy for chronic wound management. The BPC-157/TB-500 blend offers another peptide-based approach to tissue repair that could complement KPV's anti-inflammatory effects in wound healing applications.
Mucosal Immune Cell Modulation by KPV
The mucosal immune system of the gastrointestinal tract represents the largest immune organ in the body, containing more immune cells than all other immune tissues combined. The lamina propria of the intestinal mucosa harbors macrophages, dendritic cells, T lymphocytes, B lymphocytes, plasma cells, mast cells, eosinophils, innate lymphoid cells, and natural killer cells, all organized in a complex network that must balance pathogen defense with tolerance to dietary antigens and commensal bacteria. Disruption of this balance underlies inflammatory bowel disease, and restoring it is a central goal of IBD therapy.
KPV modulates multiple immune cell populations in the gut through its PepT1-mediated uptake and NF-kB inhibition. In macrophages, KPV reduces the production of TNF-alpha, IL-1beta, IL-6, and IL-12 while preserving IL-10 production. This cytokine shift promotes a transition from the pro-inflammatory M1 phenotype to the tissue-repair M2 phenotype, supporting resolution of inflammation and mucosal healing. The M2 polarization effect of KPV has been directly demonstrated in studies of the (CKPV)2 dimer, where treated macrophages showed increased arginase activity (an M2 marker) and decreased iNOS expression (an M1 marker).
In dendritic cells, NF-kB inhibition by KPV reduces the expression of co-stimulatory molecules (CD80, CD86, CD40) and MHC class II antigens, impairing the ability of dendritic cells to activate naive T lymphocytes. This effect could help suppress the generation of pathogenic effector T cells that drive IBD. At the same time, reduced NF-kB signaling in dendritic cells promotes a tolerogenic phenotype that favors the differentiation of regulatory T cells (Tregs), which actively suppress inappropriate immune responses in the gut.
In T lymphocytes, KPV's anti-inflammatory effects include reduced proliferation in response to activating stimuli, decreased production of effector cytokines (IFN-gamma from Th1 cells, IL-17 from Th17 cells), and promotion of the regulatory T cell phenotype. PepT1 is expressed on activated T cells, particularly in the intestinal mucosa, providing a direct route for KPV uptake. The selective targeting of activated (rather than resting) T cells is therapeutically advantageous, as it suppresses pathological immune responses while preserving normal immune surveillance.
Natural killer (NK) cells and innate lymphoid cells (ILCs) in the gut mucosa also express PepT1 under inflammatory conditions. While the specific effects of KPV on these cell types have not been as extensively characterized as its effects on macrophages and T cells, the general principle of NF-kB inhibition predicts reduced production of pro-inflammatory mediators and cytokines. ILC3-derived IL-22, which promotes epithelial barrier function, may actually be preserved or enhanced by KPV's selective NF-kB modulation, potentially contributing to barrier repair during active inflammation.
Enteric Nervous System Interactions
The enteric nervous system (ENS), often called the "second brain," contains over 100 million neurons that coordinate gastrointestinal motility, secretion, and blood flow. The ENS also plays an important role in intestinal inflammation through bidirectional communication with the immune system, a concept termed neuroimmune interaction. Alpha-MSH and its fragments participate in this neuroimmune crosstalk.
Enteric neurons express MC1R and potentially other melanocortin receptors, and alpha-MSH can modulate neuronal excitability and neurotransmitter release in the ENS. While KPV itself does not bind melanocortin receptors, its NF-kB inhibitory activity could affect neuroinflammation in the ENS, where glial cell activation and neuronal NF-kB signaling contribute to the altered motility, visceral pain, and secretory dysfunction that accompany IBD.
Visceral pain is a major symptom of IBD that significantly impacts quality of life. The sensitization of visceral afferent neurons by inflammatory mediators (particularly TNF-alpha, IL-1beta, and prostaglandins) lowers the threshold for pain perception, leading to abdominal cramping, urgency, and tenesmus. KPV's suppression of these mediators in the colonic mucosa could reduce visceral pain indirectly by lowering the concentration of pain-sensitizing molecules in the tissue environment. This potential analgesic benefit has not been formally studied but represents an important quality-of-life endpoint for future clinical evaluation.
The interaction between KPV and the gut-brain axis extends beyond the ENS. Alpha-MSH plays a central role in the hypothalamic regulation of appetite, energy expenditure, and stress responses through MC3R and MC4R signaling. While KPV itself does not activate these receptors, its effects on intestinal inflammation could indirectly influence gut-brain signaling through modulation of intestinal hormone secretion (GLP-1, PYY, CCK), vagal afferent nerve activity, and circulating inflammatory cytokine levels that affect brain function. For those interested in the broader gut-brain connection, the GLP-1 research hub explores how gut hormones influence metabolic regulation and appetite control.
KPV in Experimental Models of Food Allergy and Celiac Disease
Beyond IBD, KPV has potential applications in other gut inflammatory conditions including food allergies and celiac disease. These conditions share some pathological mechanisms with IBD, particularly NF-kB-dependent inflammatory signaling and intestinal barrier dysfunction, suggesting that KPV could provide therapeutic benefit.
In celiac disease, gluten-derived peptides trigger an immune response in genetically susceptible individuals carrying HLA-DQ2 or HLA-DQ8 alleles. This response involves both innate activation of intestinal epithelial cells (through gluten peptide-induced IL-15, IL-8, and NF-kB signaling) and adaptive activation of gluten-specific T cells. Alpha-MSH has been shown to suppress inflammatory cytokine production in celiac intestinal mucosa in ex vivo studies, reducing IL-8 and TNF-alpha secretion from biopsy samples of patients with active celiac disease. While these studies used full-length alpha-MSH rather than KPV, the shared anti-inflammatory mechanism suggests that KPV would provide similar benefits.
Food allergies involve IgE-mediated mast cell activation and Th2-driven inflammation in the intestinal mucosa. NF-kB signaling is required for mast cell degranulation and cytokine production, and its inhibition by KPV could reduce the intensity of allergic inflammatory responses in the gut. While this application remains speculative, the increasing prevalence of food allergies - particularly in pediatric populations - creates a clear clinical need for safe, effective anti-inflammatory agents that can be used as adjunctive therapy alongside allergen avoidance.
Comparative Efficacy Data Across IBD Models
Consolidating data from published studies provides a more complete picture of KPV's efficacy across different experimental IBD models and formulations.
| Study | Model | KPV Form | Route | Key Outcome | Magnitude of Effect |
|---|---|---|---|---|---|
| Kannengiesser 2008 | DSS colitis (mouse) | Free KPV | Oral | Body weight recovery | Significantly faster recovery vs. control |
| Kannengiesser 2008 | CD45RBhi transfer colitis | Free KPV | IP injection | Histological score | Reduced inflammatory infiltrate |
| Dalmasso 2008 | DSS colitis (mouse) | Free KPV | Oral | NF-kB activation | Significant reduction in colonic tissue |
| Dalmasso 2008 | Cell culture (Caco-2, HT-29) | Free KPV | Direct | Cytokine secretion | 40-60% reduction in TNF-alpha, IL-6 |
| Laroui 2010 | DSS colitis (mouse) | NP-encapsulated | Oral | MPO activity | Significant reduction; 12,000x dose advantage |
| Xiao 2017 | DSS colitis (mouse) | HA-KPV-NPs in hydrogel | Oral | DAI score | Comparable to free KPV at 12,000x lower dose |
| Xiao 2017 | DSS colitis (mouse) | HA-KPV-NPs in hydrogel | Oral | TNF-alpha expression | Markedly reduced in colonic tissue |
| Chen 2024 | DSS colitis (mouse) | PepT1-targeted nanodrug | Oral | Combined outcome | Enhanced efficacy with immunosuppressant co-delivery |
This consolidated data table demonstrates the consistency of KPV's anti-inflammatory effects across different research groups, animal models, formulations, and administration routes. The progression from free peptide to increasingly sophisticated nanoparticle delivery systems shows a clear trend toward improved efficacy and dose reduction, supporting continued development of targeted KPV formulations for clinical use.
Future Clinical Trial Design Considerations
Designing clinical trials for KPV in IBD requires careful consideration of multiple factors. The selection of primary endpoints should align with regulatory requirements while capturing the clinically meaningful effects of KPV therapy. For ulcerative colitis, the FDA typically requires demonstration of clinical remission (defined by the modified Mayo score) and endoscopic improvement. For Crohn's disease, the CDAI (Crohn's Disease Activity Index) score reduction and endoscopic response are standard endpoints.
Patient selection criteria should consider disease severity, prior treatment history, and biomarker status. Patients with moderate-to-severe IBD who have failed or are intolerant to conventional therapies represent the population with the greatest unmet need. However, KPV's relatively gentle mechanism of action and favorable safety profile might also position it for earlier use in the treatment algorithm, potentially in patients with mild-to-moderate disease as a first-line or second-line agent.
Biomarker-based patient stratification could enhance trial efficiency. PepT1 expression levels in colonic biopsies could identify patients most likely to respond to oral KPV, as higher PepT1 expression should correlate with greater KPV uptake and therapeutic effect. Fecal calprotectin levels, a non-invasive measure of intestinal inflammation, could serve as both a screening tool and a pharmacodynamic biomarker for KPV's anti-inflammatory activity.
Formulation selection for clinical trials should balance the potential benefits of advanced delivery systems against the practical advantages of simpler formulations. While HA-KPV-NPs offer superior preclinical efficacy, their manufacturing complexity and cost may limit initial clinical development. Enteric-coated capsules containing free KPV represent a simpler and less expensive approach that could be evaluated first, with nanoparticle formulations pursued in subsequent development phases if warranted by clinical results. The GLP-1 research hub discusses similar clinical development considerations for other therapeutic peptides targeting gut-related conditions.
Immunogenicity Assessment
Immunogenicity - the potential for a therapeutic agent to trigger an unwanted immune response against itself - is a significant concern for peptide and protein drugs. Anti-drug antibodies can reduce efficacy by neutralizing the therapeutic molecule or accelerating its clearance, and in rare cases can cause serious adverse reactions including anaphylaxis.
KPV's immunogenicity risk is expected to be very low based on several factors. First, its small size (342 daltons, just three amino acids) falls below the threshold typically required for antibody generation. Most immunogenic peptides are at least 8-10 amino acids long, and even these require carrier proteins or adjuvants to elicit antibody responses in most individuals. Second, KPV's sequence is identical to a portion of an endogenous human hormone (alpha-MSH), meaning the immune system should recognize it as "self" rather than foreign. Third, KPV does not contain any post-translational modifications, unusual amino acids, or chemical moieties that might serve as neoantigens.
The (CKPV)2 dimer, being larger (approximately 900 daltons) and containing a non-natural disulfide bridge linking two peptide units, has slightly higher theoretical immunogenicity than monomeric KPV. However, no anti-drug antibody formation was reported in the available preclinical and clinical studies of (CKPV)2, supporting the overall low immunogenicity of KPV-derived molecules.
For injectable KPV formulations, the excipients, diluents, and contaminants (if any) may pose a greater immunogenicity risk than the peptide itself. Manufacturing under GMP conditions with appropriate purity standards minimizes this risk. Endotoxin contamination, which can cause inflammatory reactions mimicking immunogenicity, should be rigorously controlled with levels below 0.5 EU/mg for parenteral products.
Reproductive and Developmental Safety
No formal reproductive toxicology studies (including fertility, embryo-fetal development, and peri/postnatal development assessments) have been conducted for KPV. This represents a significant gap in the safety database that would need to be addressed before clinical development in any population that could include women of childbearing potential.
The POMC/melanocortin system plays several roles in reproductive biology that warrant consideration. Alpha-MSH influences gonadotropin release through hypothalamic signaling, modulates sexual behavior through MC4R activation in the brain, and may play roles in placental function and fetal development. However, these effects are mediated through melanocortin receptor signaling, which KPV does not engage. The PepT1-mediated, NF-kB-inhibitory mechanism of KPV is mechanistically distinct from the reproductive effects of alpha-MSH, suggesting that KPV is unlikely to affect reproductive function through the same pathways.
NF-kB signaling itself plays important roles in embryonic development, implantation, and placental formation. Excessive suppression of NF-kB during critical developmental windows could theoretically impair these processes. However, KPV achieves only partial NF-kB suppression (40-60%), and its short half-life limits the duration of exposure. Whether these factors provide sufficient margin of safety during pregnancy is unknown and cannot be determined without formal reproductive toxicology studies.
Until reproductive safety data become available, KPV should be treated as a pregnancy Category X equivalent (potential for fetal harm based on mechanism of action, with no established human data) for the purposes of clinical decision-making. Women of childbearing potential should use effective contraception during KPV treatment, and KPV should be discontinued at least 2 weeks before attempting conception to allow complete washout. For individuals interested in fertility-related peptide research, Kisspeptin-10 and Gonadorelin address reproductive hormone regulation through distinct mechanisms.
Carcinogenicity Considerations
Long-term carcinogenicity studies have not been conducted for KPV. Theoretical risk assessment based on mechanism of action provides some guidance for evaluating this concern.
NF-kB has a complex relationship with cancer. In some contexts, NF-kB promotes tumor survival, proliferation, and metastasis through anti-apoptotic gene expression and inflammatory cytokine production. In these settings, NF-kB inhibition by KPV could theoretically have anti-tumor effects. Indeed, chronic inflammation driven by NF-kB is recognized as a risk factor for colorectal cancer in IBD patients, and anti-inflammatory therapy that reduces colonic NF-kB activity could potentially lower this risk.
However, NF-kB also plays roles in normal immune surveillance against tumors, particularly through natural killer cell activation and cytotoxic T lymphocyte function. Chronic NF-kB suppression could theoretically impair these anti-tumor immune mechanisms. The clinical significance of this concern depends on the degree and duration of NF-kB suppression, which for KPV appears to be moderate (40-60%) and transient (limited by rapid metabolism).
The alpha-MSH/melanocortin system has been studied extensively in the context of melanoma, where MC1R signaling affects melanocyte proliferation, differentiation, and survival. KPV's lack of MC1R binding means it should not directly affect melanocyte biology or melanoma risk. However, NF-kB is involved in melanoma cell survival and treatment resistance, and KPV's effect on melanoma cells (which express PepT1) has not been specifically evaluated. Until carcinogenicity data become available, KPV should not be used in patients with active malignancies without oncologist oversight. Individuals interested in peptides with anti-cancer research profiles may find FOXO4-DRI relevant, as it targets senescent cells through a distinct mechanism.
Reconstitution and Storage Protocols
Proper reconstitution and storage of KPV are essential for maintaining peptide integrity and therapeutic efficacy. The following protocols apply to lyophilized KPV powder, which is the most common form available for research and clinical use.
Reconstitution Steps:
- Remove the lyophilized KPV vial from refrigerated storage and allow it to reach room temperature (approximately 15-20 minutes). This prevents moisture condensation inside the vial that could accelerate degradation.
- Clean the vial stopper with an alcohol swab and allow it to dry completely.
- Using a sterile syringe with a fine-gauge needle (29-31G), draw up the calculated volume of bacteriostatic water (preserved with 0.9% benzyl alcohol) for multi-dose vials, or sterile water for injection for single-use preparations.
- Inject the diluent slowly down the inside wall of the vial, not directly onto the lyophilized powder. Direct high-pressure injection can denature the peptide through mechanical shearing.
- Gently swirl (do not shake vigorously) the vial until the powder is completely dissolved. KPV typically dissolves quickly due to its small size and high water solubility. If particulate matter remains after 2-3 minutes of gentle swirling, do not use the vial.
- Label the vial with the reconstitution date, concentration, and expiration date (28 days from reconstitution for bacteriostatic water preparations).
Storage Requirements:
- Lyophilized (unreconstituted) KPV: Store at -20 degrees Celsius for long-term storage (up to 24 months) or 2-8 degrees Celsius for shorter periods (up to 6 months). Protect from light and moisture.
- Reconstituted KPV in bacteriostatic water: Store at 2-8 degrees Celsius (refrigerator). Use within 28 days. Do not freeze reconstituted solutions, as freeze-thaw cycles can cause peptide aggregation and loss of activity.
- Reconstituted KPV in sterile water (preservative-free): Use within 24 hours if stored at room temperature, or within 72 hours if refrigerated. Discard any remaining solution after these time periods.
For oral capsule formulations of KPV, storage at room temperature (below 25 degrees Celsius) in a cool, dry place away from direct sunlight is typically sufficient. Enteric-coated capsules should be stored in their original moisture-protective packaging until use.
Practical Protocol Examples
The following examples illustrate how KPV might be incorporated into therapeutic protocols for different conditions. These are based on preclinical data and clinical observations and should be considered as starting points for discussion with a healthcare provider, not as prescriptive recommendations.
Protocol 1: Gut Inflammation (Oral KPV)
- Formulation: Enteric-coated capsules containing 250 mcg KPV
- Loading phase (weeks 1-8): 500 mcg daily (two capsules) taken on an empty stomach, 30 minutes before breakfast
- Maintenance phase (weeks 9+): 250 mcg daily (one capsule) on an empty stomach
- Monitoring: Fecal calprotectin every 4 weeks during loading, every 8-12 weeks during maintenance; CRP monthly
- Duration: Continue maintenance dosing as long as clinical benefit persists; reassess every 6 months
- Complementary: Consider adding BPC-157 at 250-500 mcg/day for mucosal repair support
Protocol 2: Systemic Inflammation (Subcutaneous KPV)
- Formulation: Reconstituted lyophilized KPV, 1 mg/mL concentration
- Dose: 300 mcg (0.3 mL) subcutaneously once daily in the morning
- Injection sites: Rotate between abdomen (periumbilical region), outer thigh, and upper arm
- Duration: 8-12 week cycles with 2-4 week breaks between cycles
- Monitoring: CBC, CMP, CRP at baseline, week 4, and week 8 of each cycle
- Complementary: Consider adding Thymosin Alpha-1 for enhanced immune regulation
Protocol 3: Skin Inflammation (Topical KPV)
- Formulation: KPV 0.1% cream or serum
- Application: Apply a thin layer to affected areas twice daily (morning and evening)
- Area of application: Clean, dry skin; avoid applying to open wounds or broken skin unless specifically formulated for wound care
- Duration: Continuous use during active flares; reduce to once daily or every other day for maintenance
- Complementary: Consider alternating with GHK-Cu topical for combined anti-inflammatory and regenerative support
These protocols represent theoretical frameworks based on the best available evidence. Individual responses to KPV vary, and dose adjustments may be necessary based on clinical response, tolerance, and monitoring results. The free assessment on the FormBlends website provides personalized guidance based on individual health profiles and treatment goals.
Colonic Fibrosis and KPV's Antifibrotic Potential
Chronic intestinal inflammation in IBD can lead to colonic fibrosis, a progressive thickening of the bowel wall driven by excessive deposition of extracellular matrix proteins, particularly collagen types I, III, and V. Fibrosis is a major complication of Crohn's disease, occurring in approximately 30-50% of patients and often leading to stricture formation that requires surgical resection. Ulcerative colitis can also develop fibrosis, though typically less severe than in Crohn's disease.
NF-kB signaling plays a central role in the fibrotic process. Activated NF-kB in intestinal myofibroblasts drives the expression of pro-fibrotic mediators including transforming growth factor-beta (TGF-beta), connective tissue growth factor (CTGF), and tissue inhibitors of metalloproteinases (TIMPs) that prevent collagen breakdown. NF-kB also promotes the epithelial-mesenchymal transition (EMT), a process where epithelial cells lose their cell-cell adhesion and acquire mesenchymal properties, contributing to the pool of matrix-producing myofibroblasts in the bowel wall.
Alpha-MSH and its fragments have demonstrated antifibrotic effects in several organ systems. A 2021 study published in Pharmaceuticals reviewed the antifibrotic and anti-inflammatory actions of alpha-melanocytic hormone, documenting reduced fibrosis in experimental models of hepatic, renal, pulmonary, and dermal fibrosis. The mechanism involves suppression of NF-kB-dependent pro-fibrotic gene expression, inhibition of TGF-beta signaling, and reduction of myofibroblast differentiation from precursor cells.
While KPV has not been specifically evaluated in models of intestinal fibrosis, its NF-kB inhibitory mechanism provides a strong theoretical basis for antifibrotic activity. By suppressing NF-kB in intestinal myofibroblasts and preventing NF-kB-driven EMT, KPV could reduce the progression of fibrosis during active inflammation. This potential antifibrotic benefit adds another dimension to KPV's therapeutic value in IBD, where preventing long-term structural damage is as important as controlling acute symptoms.
The antifibrotic potential of KPV aligns with growing recognition that the best approach to IBD management involves early, aggressive intervention to prevent irreversible tissue damage. The traditional "step-up" approach, where treatment escalates gradually from mild to powerful agents, may allow fibrosis to develop before effective therapy is established. KPV's favorable safety profile could support its use as an early intervention aimed at preventing fibrosis progression, even in patients with relatively mild inflammatory symptoms.
The Microbiome-Immune Axis and KPV
The gut microbiome and the mucosal immune system exist in constant communication through a network of molecular signals collectively termed the microbiome-immune axis. Disruption of this communication is a hallmark of IBD, where loss of microbial diversity, expansion of pathogenic species, and impaired immune tolerance create a self-reinforcing cycle of dysbiosis and inflammation. KPV's anti-inflammatory effects may help break this cycle, but the relationship between KPV treatment and microbiome composition deserves detailed consideration.
In healthy individuals, the intestinal microbiome produces metabolites that actively suppress NF-kB activation in the epithelium. Short-chain fatty acids (SCFAs) including butyrate, propionate, and acetate are produced by bacterial fermentation of dietary fiber and serve as both energy sources for colonocytes and anti-inflammatory signals. Butyrate inhibits NF-kB through inhibition of histone deacetylases (HDACs), promoting a chromatin state that is less permissive for inflammatory gene expression. Tryptophan metabolites produced by certain bacterial species activate the aryl hydrocarbon receptor (AhR) on intestinal epithelial cells and immune cells, promoting barrier integrity and immune tolerance.
In IBD, the bacterial species that produce these protective metabolites (particularly Faecalibacterium prausnitzii, Roseburia intestinalis, and certain Clostridium clusters) are depleted, while species that produce pro-inflammatory metabolites (including certain Proteobacteria and sulfate-reducing bacteria) expand. This shift from protective to pathogenic microbial metabolism contributes to sustained NF-kB activation and epithelial barrier breakdown.
KPV treatment could influence the microbiome through several indirect mechanisms. By reducing mucosal inflammation, KPV may create conditions that favor the re-establishment of beneficial bacterial species. Inflamed mucosa produces reactive oxygen and nitrogen species, antimicrobial peptides, and altered mucus compositions that selectively inhibit oxygen-sensitive anaerobic bacteria (which include many of the protective SCFA producers) while favoring oxygen-tolerant pathogenic species. Reducing this inflammatory selective pressure could permit microbial diversity to recover.
Additionally, KPV's preservation of epithelial barrier integrity reduces the translocation of bacterial products into the lamina propria. This reduces the antigenic stimulation that drives aberrant immune responses against commensal bacteria - a key pathogenic mechanism in IBD where loss of tolerance to normal gut flora perpetuates chronic inflammation. By reducing bacterial translocation, KPV could help restore normal immune-microbiome interactions.
Future research should directly evaluate KPV's effects on microbiome composition using high-throughput 16S rRNA gene sequencing and metagenomic analysis. Identifying specific microbial signatures associated with KPV treatment response could enable microbiome-based biomarkers for predicting which patients are most likely to benefit from KPV therapy. Combining KPV treatment with targeted probiotic supplementation (using species known to produce anti-inflammatory metabolites) could potentially produce additive benefits by simultaneously reducing inflammatory signaling and restoring protective microbial metabolism.
Ocular Inflammation Applications
The eye is an immune-privileged organ that relies on local anti-inflammatory mechanisms to prevent sight-threatening inflammation. Alpha-MSH is produced within the eye and plays a critical role in maintaining ocular immune privilege through suppression of inflammatory responses in the anterior chamber. This has led to research on alpha-MSH peptides, including KPV, for ocular inflammatory conditions.
Uveitis, an inflammatory condition affecting the uveal tract of the eye, is one of the leading causes of preventable blindness worldwide. Autoimmune uveitis involves NF-kB-dependent production of inflammatory cytokines in the eye, making it a logical therapeutic target for KPV. In experimental autoimmune uveitis (EAU) models, alpha-MSH treatment reduced retinal inflammation, preserved photoreceptor structure, and decreased inflammatory cell infiltration. While these studies primarily used full-length alpha-MSH, KPV's shared anti-inflammatory mechanism via NF-kB inhibition suggests similar therapeutic potential.
IBD-associated uveitis affects 2-5% of IBD patients and represents one of the most common extraintestinal manifestations of the disease. For these patients, KPV could potentially address both the intestinal and ocular manifestations of their disease through a single agent, simplifying treatment regimens and reducing the need for separate medications with different side effect profiles.
Topical ocular delivery of KPV would be the preferred route for eye-specific conditions. Eye drops containing KPV could deliver the peptide directly to the anterior chamber, where it would be absorbed by corneal epithelial cells and intraocular immune cells expressing PepT1. The small molecular weight of KPV would facilitate transcorneal penetration, though formulation optimization would be needed to achieve adequate intraocular concentrations. For IBD patients with concurrent uveitis, systemic KPV administration (oral or subcutaneous) might achieve sufficient ocular concentrations to benefit both conditions simultaneously.
Joint Inflammation and Arthritis
Alpha-MSH and its fragments have demonstrated anti-inflammatory effects in experimental arthritis models, suggesting another potential application for KPV. Rheumatoid arthritis is an autoimmune disease characterized by NF-kB-dependent synovial inflammation, and IBD-associated arthritis (both peripheral and axial) affects up to 30% of IBD patients.
In experimental models of gouty arthritis, melanocortin receptor activation reduced joint inflammation, neutrophil infiltration, and pain responses. While these effects were attributed primarily to MC3R signaling in monocytes and synovial cells, the NF-kB-independent anti-inflammatory mechanism of KPV could provide complementary benefit. Synovial cells express PepT1, providing a potential route for KPV uptake in joint tissue.
For IBD patients with concurrent joint inflammation, KPV's ability to address both gut and joint inflammation through a single agent represents a significant clinical advantage. Current treatments for IBD-associated arthritis often involve separate agents for each manifestation (e.g., mesalamine for the gut plus NSAIDs for joints), and some agents effective for one manifestation can worsen the other (NSAIDs can exacerbate gut inflammation). A single anti-inflammatory peptide effective for both conditions would simplify treatment and reduce polypharmacy risks.
Pulmonary Inflammation
The respiratory tract represents another potential therapeutic target for KPV. Airway epithelial cells and alveolar macrophages express PepT1, and NF-kB-driven inflammation underlies conditions including asthma, chronic obstructive pulmonary disease (COPD), and acute respiratory distress syndrome (ARDS). Alpha-MSH has demonstrated protective effects in experimental lung injury models, reducing inflammatory cell infiltration, cytokine production, and tissue damage in the lung parenchyma.
Inhaled delivery of KPV could provide targeted therapy for pulmonary inflammation while minimizing systemic exposure. The peptide's small size and water solubility are compatible with nebulized delivery, and the respiratory epithelium's PepT1 expression would facilitate direct cellular uptake from the airway surface. While no published studies have specifically evaluated inhaled KPV for pulmonary inflammation, the preclinical evidence from related alpha-MSH peptides and the biological rationale supporting PepT1-mediated delivery make this an intriguing area for future investigation.
Individuals interested in comprehensive immune and respiratory support may also explore Thymosin Alpha-1, which enhances antiviral immune responses and has been studied in respiratory infections, and LL-37, an antimicrobial peptide with both direct pathogen-killing and immunomodulatory properties relevant to pulmonary defense.
Emerging Research Directions: 2024-2026
The KPV research field continues to evolve rapidly, with several emerging directions that may expand the peptide's therapeutic applications in the coming years. Recent publications from 2024 and 2025 indicate growing interest in KPV across multiple disciplines.
The development of PepT1-targeted nanodrug platforms incorporating KPV with immunosuppressive compounds represents a promising combination strategy. A 2024 publication in Frontiers in Pharmacology described co-assembled nanoparticles combining anti-inflammatory peptide (KPV) with immunosuppressant for treating both acute and chronic colitis. This approach leverages the PepT1 targeting mechanism to deliver both anti-inflammatory and immunosuppressive agents specifically to inflamed intestinal tissue, potentially achieving greater efficacy than either component alone.
Oral enzyme-responsive nanoprobe technology published in the Journal of Nanobiotechnology in 2024 combines therapeutic delivery with real-time diagnostic imaging, enabling personalized treatment adjustments based on objective measurements of disease activity. Applying this theranostic approach to KPV delivery could transform IBD management by providing clinicians with immediate feedback on treatment response, potentially eliminating the delays and uncertainty associated with current monitoring methods (colonoscopy, fecal biomarkers).
Research into KPV's effects on fine particulate matter-induced skin damage, published in 2025, opens new applications in environmental dermatology and cosmeceutical science. As air pollution continues to worsen in many urban environments, preventive anti-inflammatory skin treatments could address a growing public health concern. The successful demonstration of KPV's protective effects against PM10-induced keratinocyte damage provides proof-of-concept for this application.
Investigation of KPV in combination with other therapeutic modalities - including probiotics, fecal microbiota transplantation, dietary interventions, and conventional pharmaceuticals - represents another active area of exploration. The non-overlapping mechanism of KPV with most conventional therapies creates opportunities for rationally designed combination regimens that address IBD pathophysiology from multiple angles simultaneously. Clinical investigators and research peptide users can stay informed about these developments through the peptide research hub and science section of the FormBlends website.
Oral Delivery Research
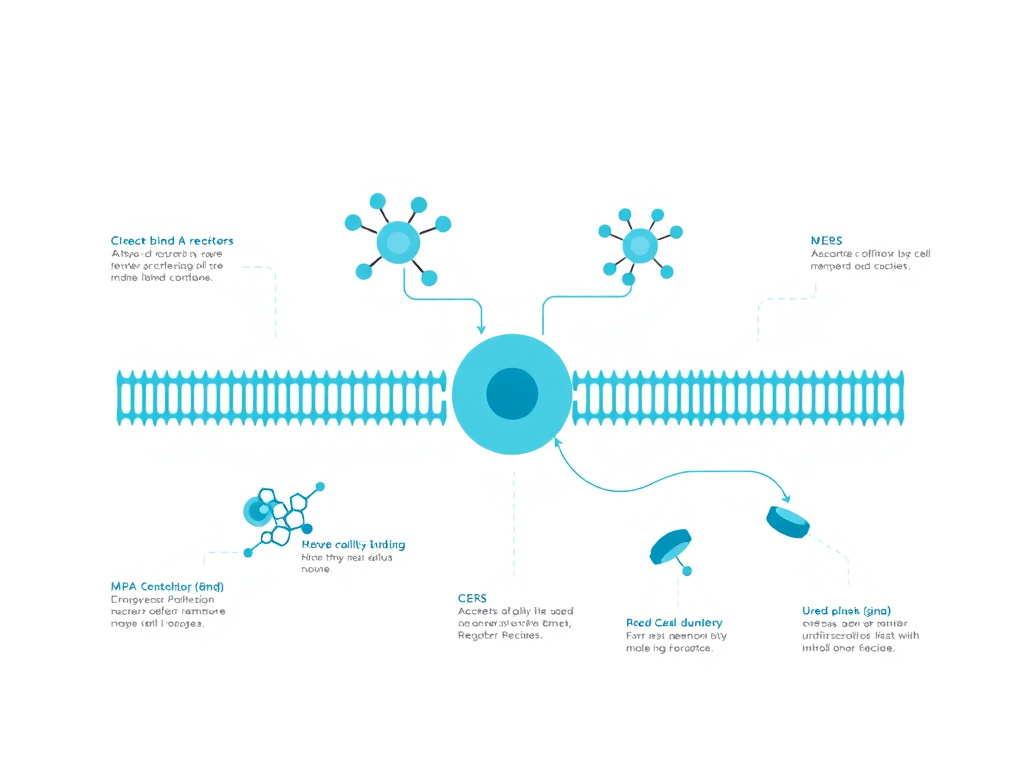
Figure 7: Advanced oral delivery systems for KPV peptide showing nanoparticle encapsulation and colon-targeted release mechanisms
Oral delivery represents the most practical administration route for chronic inflammatory conditions like IBD, but poses significant challenges for peptide therapeutics. KPV's small size (342 daltons) and three-amino-acid structure make it inherently more suitable for oral administration than larger peptides, yet it still requires protection from gastrointestinal degradation to reach the colon at therapeutic concentrations. Research over the past decade has produced increasingly sophisticated delivery systems that protect KPV during transit, target it to inflamed tissue, and dramatically reduce the dose required for therapeutic effect.
Challenges of Oral Peptide Delivery
The gastrointestinal tract presents a hostile environment for peptide drugs. The stomach's acidic pH (1.5-3.5) can hydrolyze peptide bonds, while pepsin cleaves peptides at hydrophobic residues. The small intestine contains trypsin, chymotrypsin, elastase, and carboxypeptidases that further degrade peptide substrates. Even peptides that survive enzymatic attack face absorption barriers: the intestinal epithelium limits paracellular transport of hydrophilic molecules, and efflux transporters can pump absorbed peptides back into the lumen.
KPV faces some but not all of these challenges. Its small size reduces the number of enzymatic cleavage sites compared to larger peptides. The Pro-Val bond is relatively resistant to trypsin (which cleaves after Arg and Lys) and chymotrypsin (which cleaves after aromatic and large hydrophobic residues). However, the Lys-Pro bond may be susceptible to certain aminopeptidases and diaminopeptidases present in the intestinal brush border. And even if KPV arrives intact at the colonic epithelium, it must be absorbed in sufficient quantities to produce intracellular anti-inflammatory effects.
The natural PepT1 transporter provides an absorption mechanism for KPV in the colon, but unformulated KPV administered orally is subject to significant pre-colonic degradation. Pharmacokinetic studies in mice suggest that less than 10% of an oral KPV dose reaches the colon intact when administered as a simple aqueous solution. This degradation necessitates either high doses (with associated cost and potential for off-target effects in the upper GI tract) or protective formulations that shield KPV during transit and release it specifically in the colon.
Hyaluronic Acid-Functionalized Nanoparticles (HA-KPV-NPs)
The most thoroughly characterized oral KPV delivery system is the hyaluronic acid-functionalized nanoparticle platform developed by Xiao, Merlin, and colleagues at Georgia State University and the Georgia Institute of Technology. Published in Molecular Therapy in 2017, this system represents a sophisticated multi-component approach to targeted oral peptide delivery.
The nanoparticle architecture consists of three functional layers. The innermost core is composed of poly(lactic acid) (PLA), a biodegradable polymer that encapsulates KPV and controls its release rate. PLA is widely used in pharmaceutical applications and is approved by the FDA for injectable and implantable drug products. The middle layer consists of poly(ethylene glycol) (PEG), which provides a hydrophilic corona that improves nanoparticle stability in aqueous environments and reduces protein adsorption (opsonization) that could trigger immune clearance. The outermost layer is hyaluronic acid (HA), a naturally occurring glycosaminoglycan that serves as both a targeting ligand and a mucoadhesive agent.
The resultant HA-KPV-NPs have a particle size of approximately 272.3 nm and a slightly negative zeta potential of approximately -5.3 mV. The negative surface charge, contributed by the carboxyl groups of hyaluronic acid, promotes colloidal stability while maintaining compatibility with the negatively charged intestinal mucus layer. Drug loading efficiency for KPV was optimized to balance the competing demands of high payload capacity and small particle size.
Hyaluronic acid targeting is central to the therapeutic strategy. CD44, the primary receptor for HA, is upregulated on inflamed colonic epithelial cells and activated macrophages in IBD. When HA-KPV-NPs encounter CD44-expressing cells, the HA coat mediates receptor binding and cellular uptake via receptor-mediated endocytosis. This provides an additional targeting mechanism beyond PepT1-mediated uptake, ensuring that the nanoparticles accumulate preferentially at sites of active inflammation.
Chitosan-Alginate Hydrogel Encapsulation
To protect HA-KPV-NPs during gastric and small intestinal transit, the nanoparticles are embedded in a chitosan-alginate hydrogel. This hydrogel system exploits the pH gradient of the gastrointestinal tract to achieve colon-specific release.
Alginate is a natural polysaccharide from seaweed that forms gels in the presence of divalent cations (typically calcium). At the acidic pH of the stomach, alginate gels remain intact, protecting their payload from acid and pepsin. Chitosan, a deacetylated form of chitin derived from crustacean shells, provides structural reinforcement through electrostatic interactions with alginate's carboxyl groups. The chitosan-alginate complex is stable at pH values below 6.0 but dissolves progressively as pH rises above 6.5, corresponding to the transition from the distal ileum to the cecum and ascending colon.
In vivo biodistribution studies in mice confirmed that fluorescently labeled HA-KPV-NPs released from the hydrogel accumulated preferentially in the colon, with minimal release in the stomach or small intestine. The released nanoparticles adhered to inflamed colonic mucosa significantly more than to healthy mucosa, demonstrating the dual targeting provided by mucoadhesive HA and CD44 receptor binding. This selective retention at inflammatory sites prolonged KPV exposure and enhanced therapeutic efficacy.
Therapeutic Efficacy of Nanoparticle-Delivered KPV
The therapeutic performance of the HA-KPV-NP/hydrogel system in DSS colitis mice was impressive. Oral administration significantly reduced disease activity index (DAI) scores, which incorporate body weight change, stool consistency, and occult or gross rectal bleeding. Histological assessment showed preservation of crypt architecture, reduced immune cell infiltration, and less epithelial erosion in treated animals compared to untreated colitis controls.
Critically, the HA-KPV-NP system achieved these effects at KPV concentrations approximately 12,000-fold lower than free KPV in solution. This extraordinary dose reduction reflects the combined benefits of protection from degradation (increasing the fraction of administered KPV that reaches the colon intact), targeted delivery to inflamed cells (concentrating KPV at therapeutic sites), controlled release (maintaining therapeutic concentrations over extended periods), and enhanced cellular uptake via CD44-mediated endocytosis (in addition to PepT1 transport).
The 12,000-fold dose reduction has major practical implications. It means that effective oral KPV therapy for IBD could potentially be achieved with microgram quantities of peptide per dose, rather than the milligram quantities that would be needed with unformulated oral KPV. This dramatically reduces manufacturing costs, minimizes the risk of systemic side effects, and makes it feasible to produce therapeutic doses in conventional oral dosage forms such as capsules or tablets.
Next-Generation Delivery Approaches
Building on the HA-KPV-NP platform, researchers have explored several next-generation delivery strategies that could further improve KPV's therapeutic index for oral administration.
Enzyme-responsive nanoparticles represent one promising direction. A 2024 study published in Frontiers in Pharmacology described PepT1-targeted nanoparticles based on co-assembly of KPV with immunosuppressant compounds for combined treatment of acute and chronic DSS-induced colitis. These nanoparticles release their payload in response to enzymes specifically produced at sites of intestinal inflammation, such as matrix metalloproteinases and myeloperoxidase, providing an additional layer of site-specific activation.
Oral enzyme-responsive nanoprobes developed for inflammatory bowel disease theranostics combine therapeutic delivery with diagnostic imaging capabilities. These dual-function particles can both treat inflammation with KPV or similar anti-inflammatory payloads and provide real-time assessment of disease activity through imaging signals generated at inflammatory sites. This approach could enable personalized dosing adjustments based on objective measures of disease activity rather than clinical symptoms alone.
Nanoparticle-in-microparticle (NiM) systems offer improved handling and manufacturing characteristics compared to naked nanoparticle suspensions. In these systems, KPV-loaded nanoparticles are spray-dried into microparticle carriers that can be filled into conventional capsules. The microparticles dissolve in the intestinal lumen, releasing the nanoparticles for targeted delivery. This approach bridges the gap between sophisticated nanoparticle drug delivery and practical pharmaceutical manufacturing at scale.
For those interested in the broader field of oral peptide delivery, the peptide research hub covers delivery technologies applicable to multiple therapeutic peptides. And the science and research section provides context on how these advanced formulation approaches are advancing the field of peptide therapeutics generally.
Stability and Formulation Considerations
The stability of KPV in various formulation environments has been characterized to guide product development. In aqueous solution at pH 7.4, KPV shows reasonable stability at refrigerated temperatures (2-8 degrees Celsius) for several weeks but degrades gradually at room temperature over the same period. Lyophilized (freeze-dried) KPV is stable for extended periods at room temperature when stored with desiccant protection from moisture.
The simplicity of KPV's structure - three standard amino acids with no labile side chains, no disulfide bonds, and no glycosylation - simplifies formulation compared to more complex peptides and proteins. There are no chirality concerns beyond the standard L-amino acid configuration, and the peptide does not aggregate or form amyloid fibrils at therapeutically relevant concentrations. These favorable formulation properties support the feasibility of developing stable, commercially viable KPV products for oral, injectable, and topical administration.
Excipient compatibility studies have identified several materials that are compatible with KPV and suitable for oral formulations. These include hydroxypropyl methylcellulose (HPMC) capsule shells, microcrystalline cellulose as a bulking agent, magnesium stearate as a lubricant, and enteric coating polymers (Eudragit L100, HPMCAS) for pH-dependent release. No significant incompatibilities have been reported between KPV and standard pharmaceutical excipients, facilitating straightforward product development once clinical efficacy is established.
Dosing & Routes
Figure 8: KPV dosing ranges and administration routes for different therapeutic applications
KPV can be administered through multiple routes including oral, subcutaneous injection, and topical application. The optimal route depends on the target condition: oral for gastrointestinal inflammation, subcutaneous for systemic inflammatory conditions, and topical for localized skin disorders. While formal dose-finding clinical trials in humans have not been completed, preclinical data and clinical observations provide general guidance on dosing ranges for each route.
Oral Administration
Oral KPV is the preferred route for gastrointestinal inflammatory conditions including ulcerative colitis, Crohn's disease, and other forms of intestinal inflammation. The oral route takes advantage of KPV's PepT1-mediated absorption in the colon, delivering the peptide directly to the tissue where anti-inflammatory activity is needed.
Dosing recommendations for oral KPV vary across the available literature. Preclinical studies in mice used doses of approximately 20-50 micrograms per day, equivalent to roughly 1 mg/kg body weight. Translating these doses to humans using allometric scaling (which accounts for differences in body surface area between species), the human equivalent dose would be approximately 5-15 mg per day for a 70 kg adult. However, this calculation does not account for species differences in KPV bioavailability, which could shift the optimal human dose in either direction.
Reports from clinical practitioners using KPV describe oral doses ranging from 200 to 500 micrograms (0.2 to 0.5 mg) per day when using capsule or liquid formulations designed for direct peptide delivery. Some reports describe doses as high as 250 mg twice daily for KPV in certain oral formulations, though these higher doses likely reflect the use of unprotected peptide (without enteric coating or nanoparticle delivery) where significant gastrointestinal degradation occurs before the peptide reaches the colon.
For optimal oral delivery, KPV should ideally be taken on an empty stomach (at least 30 minutes before eating) to minimize interaction with dietary proteins and digestive enzymes. Enteric-coated capsules that resist gastric dissolution and release their contents in the small intestine or colon provide better delivery efficiency than standard capsules. The timing of administration relative to meals can affect both gastric emptying rate and intestinal transit time, potentially influencing how much KPV reaches the colon. The dosing calculator can assist with determining appropriate starting doses based on individual parameters.
Subcutaneous Injection
Subcutaneous (SubQ) injection provides the most reliable route for systemic KPV delivery, bypassing gastrointestinal degradation entirely and achieving consistent bioavailability. This route is preferred for systemic inflammatory conditions, for conditions affecting multiple organ systems, and for situations where precise dose control is important.
Subcutaneous KPV dosing typically ranges from 200 to 500 micrograms per injection, administered once or twice daily. The injection is performed using an insulin syringe (29-31 gauge needle, 0.3-1 mL volume) into subcutaneous tissue, commonly at the abdomen, thigh, or upper arm. Injection sites should be rotated to prevent lipodystrophy or local tissue reactions, though significant injection site reactions with KPV have not been commonly reported.
For reconstitution of lyophilized KPV, bacteriostatic water is the preferred diluent for multi-dose vials. The reconstituted solution should be stored at 2-8 degrees Celsius (refrigerated) and used within 28-30 days. Sterile technique during reconstitution and injection is essential to prevent infection. The reconstitution volume is chosen to provide a convenient injection volume - typically 0.1-0.5 mL per dose - while maintaining KPV concentration within its stability range.
| Parameter | Oral Route | Subcutaneous Route | Topical Route |
|---|---|---|---|
| Typical dose range | 200-500 mcg/day (protected formulation); up to 500 mg/day (unprotected) | 200-500 mcg per injection, 1-2x daily | Varies by formulation; 7.5 mg per application reported |
| Bioavailability | Variable (depends on formulation protection) | High (near 100%) | Low to moderate (depends on penetration enhancement) |
| Onset of action | Hours to days (gradual accumulation) | Minutes to hours | Hours (local effect) |
| Best for | IBD, colitis, gut inflammation | Systemic inflammation, multiple targets | Psoriasis, dermatitis, localized skin conditions |
| Convenience | High (capsules) | Moderate (requires injection technique) | High (cream/gel application) |
| Key limitation | GI degradation reduces bioavailability | Injection discomfort, short half-life | Stratum corneum barrier limits penetration |
Topical Application
Topical KPV is used for localized skin inflammatory conditions including psoriasis, eczema, contact dermatitis, and wound healing. Topical application provides direct delivery to the target tissue with minimal systemic exposure, reducing the risk of off-target effects.
Topical KPV formulations typically incorporate the peptide into a cream, gel, or serum base at concentrations ranging from 0.01% to 0.5% (w/w). Penetration enhancers such as dimethyl sulfoxide (DMSO), oleic acid, or transcutol may be added to improve delivery through the stratum corneum. Some formulations use liposomal or nanoparticle encapsulation to enhance skin penetration without chemical penetration enhancers, which can cause irritation.
Application frequency for topical KPV is typically twice daily to affected areas. The cream or gel is applied in a thin layer and allowed to absorb for several minutes before covering with clothing or applying other skincare products. For conditions affecting the face or other sensitive areas, KPV's lack of pigmentary effects and minimal reported irritation make it preferable to topical corticosteroids, which can cause skin thinning, telangiectasia, and perioral dermatitis with chronic use on facial skin.
Transdermal delivery technologies, as discussed in the skin inflammation section, can enhance topical KPV penetration. Iontophoretic delivery through microporated skin has been shown to achieve therapeutic concentrations in the dermis, and dissolving microneedle patches offer a painless alternative to iontophoresis for enhanced dermal delivery. These technologies are currently in research stages and are not yet available in commercial KPV products.
Dosing Protocols and Cycling
Optimal dosing protocols for KPV have not been established through controlled clinical trials. The following recommendations are based on preclinical data, clinical observations, and theoretical pharmacological considerations. They should be discussed with a healthcare provider before implementation.
For gut inflammation, a common approach involves an initial loading phase of 4-8 weeks at higher doses (300-500 mcg/day oral or subcutaneous), followed by a maintenance phase at lower doses (100-200 mcg/day) for ongoing inflammation management. The loading phase provides rapid accumulation of anti-inflammatory effect, while the maintenance phase sustains this effect while minimizing long-term exposure.
Some practitioners recommend cycling protocols with periodic breaks (e.g., 8 weeks on, 2-4 weeks off) to prevent receptor desensitization or tolerance. While KPV's mechanism does not involve melanocortin receptor binding (which could desensitize with chronic agonist exposure), cycling may still be beneficial to allow the body's own regulatory mechanisms to contribute to inflammation control during off periods.
For acute inflammatory flares, short courses of higher-dose KPV (500 mcg twice daily subcutaneously) may be used for 7-14 days, followed by step-down to maintenance dosing. This approach mirrors the clinical use of corticosteroids for IBD flares, where high-dose induction therapy is followed by gradual tapering, but without the significant adverse effects associated with corticosteroid use.
Drug Interactions and Contraindications
Formal drug interaction studies for KPV have not been published. Theoretical considerations suggest several potential interactions that warrant awareness.
KPV is transported by PepT1, which also transports numerous drug molecules including certain beta-lactam antibiotics (ampicillin, cephalexin), ACE inhibitors (captopril, enalapril), and antiviral agents (valacyclovir). Co-administration of KPV with PepT1 substrate drugs could result in competitive inhibition, potentially reducing the absorption of either compound. Separating the administration of KPV from PepT1 substrate medications by at least 2 hours would minimize this risk.
The anti-inflammatory mechanism of KPV (NF-kB suppression) overlaps with that of corticosteroids and some NSAIDs. Combining KPV with these agents could produce additive or enhanced anti-inflammatory effects, which might be therapeutically beneficial in some situations but could also increase the risk of immune suppression. Patients using KPV in combination with immunosuppressive drugs should be monitored for signs of over-suppression, including increased susceptibility to infections.
There are no known absolute contraindications to KPV based on current evidence. Theoretical caution is advised in patients with active infections (where suppressing NF-kB could impair pathogen clearance), patients receiving immunosuppressive therapy for organ transplantation, and pregnant or lactating women (due to lack of safety data in these populations). Individuals considering KPV should consult with a qualified healthcare provider and can access additional information through the free assessment available on the FormBlends website.
Safety

Figure 9: Safety profile assessment of KPV peptide based on preclinical and observational data
KPV demonstrates a favorable safety profile in preclinical studies and clinical observations, consistent with its origin as a naturally occurring fragment of an endogenous hormone. The peptide's small size, receptor-independent mechanism of action, and rapid metabolism contribute to a low risk of systemic adverse effects. However, comprehensive toxicology studies and large-scale human safety trials have not been conducted, and the long-term safety of exogenous KPV supplementation remains an area requiring further investigation.
Preclinical Safety Data
Animal studies evaluating KPV for inflammatory bowel disease, skin inflammation, and infection have not reported significant adverse effects at therapeutic doses. In DSS and TNBS colitis models, mice receiving oral or subcutaneous KPV showed no changes in body weight (beyond the protective effects against colitis-related weight loss), organ weights, food intake, or behavior compared to vehicle-treated controls. No deaths attributable to KPV treatment occurred in any published preclinical study.
The HA-KPV-NP nanoparticle delivery system has been evaluated for cytotoxicity using standard cell viability assays (MTT and LDH release) in multiple cell lines including Caco-2 (intestinal epithelial), RAW 264.7 (macrophage), and Colon-26 (colonic epithelial) cells. The nanoparticles showed no significant cytotoxicity at the concentrations used for therapeutic delivery, and the component materials (PLA, PEG, hyaluronic acid, chitosan, alginate) are all biocompatible with established safety records in pharmaceutical applications.
Hematological and serum chemistry parameters were not significantly altered by KPV treatment in the studies that reported these endpoints. Complete blood counts, liver function tests (ALT, AST, alkaline phosphatase, bilirubin), and kidney function markers (creatinine, BUN) remained within normal ranges in KPV-treated animals. These findings, while reassuring, come from relatively short-term studies (typically 2-4 weeks) and do not address the safety of chronic long-term administration.
Immunological Safety Considerations
As an anti-inflammatory agent that suppresses NF-kB, KPV theoretically has the potential to impair immune defense against infections. NF-kB plays a central role in the innate immune response to bacterial and viral pathogens, and its chronic suppression could increase infection susceptibility. This concern is shared with all anti-inflammatory and immunosuppressive therapies, from corticosteroids to anti-TNF biologics.
Several factors mitigate this concern for KPV. First, KPV modulates rather than eliminates NF-kB activity. At therapeutic concentrations, it reduces NF-kB activation by approximately 40-60%, not by 100%. This partial suppression dampens pathological inflammation while preserving sufficient NF-kB activity for basic immune defense. Second, KPV's preferential uptake by PepT1 in inflamed tissue means that its anti-inflammatory effects are concentrated at sites of active inflammation rather than distributed systemically, reducing the impact on global immune function. Third, the peptide's short half-life means that its effects are transient and quickly reversible upon discontinuation.
Interestingly, the (CKPV)2 dimer has demonstrated direct antimicrobial activity against Candida species, suggesting that KPV-derived molecules may actually enhance rather than impair defense against certain pathogens. The candidacidal mechanism, mediated through cAMP elevation in the fungal cell, operates independently of the host's NF-kB pathway. This dual anti-inflammatory and antimicrobial property is rare among therapeutic agents and could be particularly valuable in clinical situations where infection and inflammation coexist, such as infectious colitis or secondarily infected skin conditions.
Comparison with Alternative Anti-Inflammatory Safety Profiles
Putting KPV's safety profile in context with established anti-inflammatory therapies highlights its potential advantages for long-term use.
| Agent | Common Adverse Effects | Serious Risks | Long-Term Concerns |
|---|---|---|---|
| KPV | None consistently reported | Theoretical infection risk (unconfirmed) | Unknown (insufficient long-term data) |
| Prednisone | Weight gain, mood changes, insomnia, hyperglycemia | Adrenal suppression, osteoporosis, diabetes | Cataracts, avascular necrosis, infections |
| Infliximab | Infusion reactions, headache, nausea | Serious infections (TB reactivation), lymphoma | Anti-drug antibody formation, loss of response |
| Tofacitinib | Upper respiratory infections, headache | Herpes zoster, venous thromboembolism, malignancy | Cardiovascular events (FDA boxed warning) |
| Azathioprine | Nausea, fatigue, myelosuppression | Pancreatitis, hepatotoxicity | Lymphoma risk (especially with anti-TNF) |
| BPC-157 | None consistently reported | Theoretical concerns (angiogenesis) | Unknown (insufficient long-term data) |
The comparison highlights that KPV's safety profile, while incompletely characterized, compares favorably to conventional anti-inflammatory agents that carry well-documented risks of serious adverse effects with chronic use. This favorable comparison is consistent with KPV's origin as a fragment of an endogenous hormone, which suggests compatibility with normal physiological processes.
Special Populations
Safety data in special populations are limited and require consideration of theoretical risks in the absence of direct evidence.
Pregnancy and Lactation: No studies have evaluated KPV safety during pregnancy or lactation. Alpha-MSH levels fluctuate during pregnancy and may play roles in placental function and fetal development. Exogenous KPV supplementation during pregnancy should be avoided until safety data become available. Similarly, it is unknown whether KPV is excreted in breast milk, and caution is advised for nursing mothers.
Pediatric Populations: The POMC/melanocortin system plays important roles in childhood growth, development, and metabolic regulation. While KPV's receptor-independent mechanism reduces concerns about disrupting melanocortin receptor signaling, the safety of exogenous KPV administration in children has not been evaluated. Pediatric use should be considered investigational and undertaken only under medical supervision.
Elderly Patients: Aging is associated with declining immune function (immunosenescence), which could theoretically increase the risk of infection during NF-kB suppression. However, aging is also associated with chronic low-grade inflammation ("inflammaging") that contributes to multiple age-related diseases. KPV's anti-inflammatory effects could be particularly beneficial in elderly patients, but appropriate caution and monitoring for infection are warranted. Other peptides of interest for aging populations include Epithalon and Humanin, which address age-related decline through complementary mechanisms.
Immunocompromised Patients: Patients with HIV/AIDS, those receiving chemotherapy, transplant recipients on immunosuppressive drugs, and individuals with primary immunodeficiency disorders should use KPV with particular caution. The additive effect of KPV's NF-kB suppression on top of existing immune compromise could increase infection risk. If KPV is considered for these patients, careful monitoring with regular complete blood counts and clinical assessment for infection is recommended.
Adverse Event Reporting and Monitoring
Given the limited formal safety data, individuals using KPV should be aware of potential adverse effects to watch for and report to their healthcare provider. These include signs of infection (fever, persistent pain, unusual fatigue, wound healing delays), allergic reactions (rash, itching, swelling, difficulty breathing), injection site reactions (persistent redness, swelling, or pain at subcutaneous injection sites), and any unexpected symptoms that develop during KPV use.
Baseline and periodic laboratory monitoring is recommended for individuals using KPV on a chronic basis. A reasonable monitoring schedule includes complete blood count (CBC) with differential at baseline and every 3 months, comprehensive metabolic panel (CMP) including liver and kidney function at baseline and every 3 months, and C-reactive protein (CRP) or erythrocyte sedimentation rate (ESR) to track inflammatory status and treatment response. These tests can identify potential safety concerns early and provide objective data on treatment efficacy. The free assessment on the FormBlends website can help individuals determine whether KPV may be appropriate for their specific situation.
Quality and Sourcing Considerations
The safety of any peptide product depends heavily on manufacturing quality. KPV intended for human use should be manufactured under Good Manufacturing Practice (GMP) conditions with appropriate quality controls. Key quality parameters include peptide purity (greater than 98% by HPLC), correct amino acid sequence confirmed by mass spectrometry, endotoxin levels below 0.5 EU/mg for injectable products, sterility for injectable formulations, and appropriate storage conditions to prevent degradation.
Sourcing KPV from reputable suppliers that provide certificates of analysis (COA) with batch-specific purity, identity, and sterility data is essential for safe use. Products without COA documentation, those sold at suspiciously low prices, or those making unsubstantiated therapeutic claims should be avoided. The FormBlends science page provides guidance on evaluating peptide quality and sourcing.
Pharmacokinetics and Metabolism
Understanding the pharmacokinetics of KPV is essential for optimizing dosing regimens and predicting therapeutic outcomes. While comprehensive human pharmacokinetic data are not available, animal studies and in vitro experiments provide a foundation for understanding how KPV is absorbed, distributed, metabolized, and eliminated.
Absorption: Oral KPV absorption occurs primarily through PepT1-mediated active transport in the intestine. In the small intestine, where PepT1 is constitutively expressed at high levels, KPV can be absorbed rapidly, but this absorption exposes the peptide to first-pass hepatic metabolism before reaching the systemic circulation. In the colon, where PepT1 expression is low under normal conditions but elevated during inflammation, absorption is more targeted to inflamed tissue. The bioavailability of oral KPV has been estimated at less than 10% when administered as a simple solution, due to pre-absorptive enzymatic degradation. Enteric-coated and nanoparticle formulations substantially improve this figure by protecting the peptide during gastric and small intestinal transit.
Subcutaneous injection provides near-complete bioavailability, with KPV absorbed from the injection site into local capillaries and lymphatic vessels. Peak plasma concentrations are typically achieved within 15-30 minutes of subcutaneous administration, though the exact pharmacokinetic profile depends on the injection volume, concentration, and anatomical site.
Distribution: Once in the systemic circulation, KPV distributes rapidly to tissues based on blood flow and PepT1 expression patterns. The small intestine, kidney, liver, and inflamed tissues (which upregulate PepT1) are expected to accumulate the highest concentrations. KPV's small size and hydrophilicity limit its binding to plasma proteins, resulting in a high free fraction in the blood and rapid tissue distribution. The volume of distribution has not been precisely measured but is expected to be moderate, reflecting the peptide's water solubility and limited lipophilicity.
Metabolism: KPV is metabolized by peptidases in plasma, liver, kidney, and intestinal brush border. The primary metabolic pathways involve cleavage of the Lys-Pro peptide bond by diaminopeptidases and the Pro-Val bond by prolidase-type enzymes. The resulting amino acids (lysine, proline, valine) enter normal amino acid metabolic pathways and are either incorporated into proteins or further metabolized for energy. The rapidity of peptide metabolism contributes to KPV's short half-life and explains why frequent dosing or sustained-release formulations are necessary for therapeutic applications.
Elimination: The amino acid products of KPV metabolism are eliminated through normal metabolic pathways, with the kidney playing a minor role in direct KPV excretion. Renal clearance of intact KPV is limited by PepT1-mediated reabsorption in the proximal tubule, where PepT1 expressed on the apical membrane recovers filtered peptides from the tubular fluid. This renal PepT1 expression may contribute to KPV's therapeutic effects in renal inflammation, as the peptide accumulates in kidney tissue during filtration and reabsorption.
The overall elimination half-life of KPV in plasma is estimated to be on the order of minutes to tens of minutes, consistent with the rapid metabolism of small peptides. This short half-life limits the duration of pharmacological effect following a single dose but also means that adverse effects, if any, would resolve quickly upon discontinuation. The rapid clearance supports a safety advantage for KPV compared to longer-acting anti-inflammatory agents, where adverse effects may persist for days or weeks after the last dose.
KPV Quality Assessment: What to Look for in a Certificate of Analysis
For individuals sourcing KPV for therapeutic use, understanding the certificate of analysis (COA) is critical for ensuring product quality and safety. A thorough COA for injectable-grade KPV should include the following test results.
Identity Testing: Mass spectrometry (MS) confirming the correct molecular weight (approximately 342 daltons for the free peptide). The observed mass should match the theoretical mass within the instrument's accuracy specifications. Amino acid analysis confirming the correct molar ratio of lysine, proline, and valine (1:1:1). Any deviation from this ratio indicates incorrect synthesis or contamination.
Purity Testing: High-performance liquid chromatography (HPLC) purity should be 98% or higher for injectable products and 95% or higher for oral/topical products. The HPLC chromatogram should show a single major peak with minimal additional peaks, indicating the absence of deletion sequences, truncation products, or other synthetic impurities. Common impurities in synthetic tripeptides include dipeptide fragments (KP, PV), des-amino variants, and racemized (D-amino acid) isomers.
Endotoxin Testing: For injectable products, bacterial endotoxin (lipopolysaccharide) levels must be below 0.5 EU/mg, as measured by the Limulus Amebocyte Lysate (LAL) test. Endotoxin contamination can cause fever, inflammation, and, in severe cases, septic shock. Oral and topical products have less stringent endotoxin requirements but should still be tested.
Sterility Testing: Injectable products must pass sterility testing according to USP Chapter 71 or equivalent standards. This involves incubation of the product with growth media for 14 days to confirm the absence of viable bacteria and fungi. Sterility is essential for subcutaneous injection, where introduction of microorganisms can cause local infection or systemic sepsis.
Water Content: Lyophilized KPV should have residual moisture content below 3%, as measured by Karl Fischer titration or thermogravimetric analysis. Excess moisture can accelerate peptide degradation during storage, reducing potency and potentially generating harmful degradation products.
Appearance: Lyophilized KPV should appear as a white to off-white powder or cake. Significant discoloration (yellow, brown) suggests oxidative degradation. The powder should dissolve readily in water or bacteriostatic water without forming aggregates or precipitates.
When evaluating COAs, pay attention to the date of testing, the testing laboratory's accreditation status, and whether the results are batch-specific (preferable) or generic. COAs that do not list specific numerical results for each test, or that appear to use identical results across multiple batches, should raise quality concerns. FormBlends' KPV products include batch-specific COA documentation that meets these quality standards.
Comparison of KPV with Conventional IBD Medications: A Patient Perspective
From a patient experience standpoint, the differences between KPV and conventional IBD medications extend beyond mechanism of action and efficacy data. Quality of life, convenience, cost, and long-term sustainability all influence treatment choices and adherence.
Treatment Burden: Many conventional IBD therapies impose significant treatment burden. Infliximab requires intravenous infusion every 6-8 weeks, typically at an infusion center, with each session lasting 2-3 hours. Adalimumab and other subcutaneous biologics require biweekly self-injection. Azathioprine and other immunomodulators require regular blood monitoring to detect myelosuppression. By comparison, oral KPV - a once-daily capsule - represents a minimal treatment burden that supports long-term adherence.
Onset of Action: Biological therapies for IBD typically require 8-12 weeks to achieve full therapeutic effect, and some patients do not respond until 16-24 weeks. KPV's anti-inflammatory effects in preclinical models were observed within days of treatment initiation, though the time to clinical benefit in humans is unknown. The rapid onset of NF-kB suppression (within hours of peptide exposure at the cellular level) suggests that clinical improvement could potentially occur faster than with biologics, though this remains speculative.
Reversibility: KPV's short half-life means its effects are rapidly reversible upon discontinuation. This is advantageous in situations where temporary cessation of anti-inflammatory therapy is needed, such as before surgical procedures or during acute infections. In contrast, biological agents have long half-lives (2-4 weeks for most antibody-based biologics), meaning their immunosuppressive effects persist for weeks after the last dose. This persistence can complicate perioperative management and infection treatment.
Cost Considerations: Biologic therapies for IBD cost $15,000-$40,000 or more per year in the United States. JAK inhibitors cost $50,000-$70,000 per year. Even generic immunomodulators like azathioprine, while less expensive, carry costs for required laboratory monitoring. KPV, as a small synthetic tripeptide, is inherently less expensive to manufacture than antibody-based biologics. Current research-grade KPV pricing reflects small-scale production and limited competition, but pharmaceutical-scale manufacturing could dramatically reduce per-dose costs.
These practical advantages do not replace the need for clinical efficacy data, but they help explain the growing interest in KPV among patients and clinicians seeking alternatives to the current IBD therapeutic paradigm. The lifestyle hub on FormBlends provides additional perspectives on integrating peptide therapies into daily life, while the dosing calculator helps optimize individual treatment protocols.
Understanding KPV's Role in Mucosal Immunology
The mucosal immune system presents unique challenges for anti-inflammatory therapy. Unlike systemic immune compartments, the gut mucosa must maintain a state of "physiological inflammation" - a baseline immune activation that monitors commensal bacteria, clears ingested pathogens, and responds to dietary antigens without causing tissue damage. The goal of IBD therapy is not to eliminate all immune activity in the gut but to restore this carefully calibrated balance between defense and tolerance.
KPV's partial NF-kB suppression (approximately 40-60% reduction) is well-suited to this therapeutic objective. Complete NF-kB abolition would impair essential immune functions including antimicrobial peptide production, IgA secretion, and epithelial innate immunity, potentially increasing susceptibility to enteric infections. The moderate degree of suppression achieved by KPV reduces pathological inflammation while preserving sufficient NF-kB activity for baseline mucosal defense. This selective modulation is difficult to achieve with conventional anti-inflammatory agents, many of which either produce near-complete suppression at therapeutic doses (corticosteroids) or target single cytokines without addressing the broader inflammatory program (anti-TNF agents).
The concept of mucosal healing has become a central treatment target in IBD management over the past decade. Mucosal healing, defined as the restoration of normal endoscopic and histological appearance of the intestinal mucosa, is associated with improved long-term outcomes including reduced rates of hospitalization, surgery, colorectal cancer, and disability. Achieving mucosal healing typically requires sustained control of inflammation over weeks to months, allowing the epithelium to regenerate and the lamina propria to return to its normal, non-inflamed state.
KPV's combination of anti-inflammatory activity and barrier-protective effects positions it well for promoting mucosal healing. By simultaneously reducing the inflammatory signals that damage the mucosa and supporting the epithelial repair processes that restore it, KPV addresses both the destructive and reparative aspects of mucosal healing. The preservation of IL-10 production during KPV treatment is particularly relevant here, as IL-10 is a key cytokine for the resolution phase of inflammation and for promoting tolerogenic immune responses that prevent relapse.
Secretory IgA (sIgA), the predominant immunoglobulin in mucosal secretions, plays a critical role in maintaining the microbiome-immune balance by coating commensal bacteria and preventing their adherence to the epithelium without triggering destructive inflammatory responses. NF-kB is involved in B cell differentiation and IgA class switching, and excessive NF-kB suppression could theoretically impair sIgA production. KPV's moderate NF-kB modulation appears to preserve sIgA production while reducing the production of pro-inflammatory immunoglobulins and cytokines, though this balance has not been explicitly characterized in published studies.
Sleep, Circadian Rhythm, and Inflammation: Contextualizing KPV's Effects
The relationship between inflammation, sleep quality, and circadian rhythm is increasingly recognized as an important factor in inflammatory disease management. NF-kB activity follows a circadian pattern, with higher activity during the active phase (daytime in humans) and lower activity during the rest phase (nighttime). Disruption of this circadian NF-kB rhythm - whether from shift work, sleep deprivation, or circadian gene mutations - is associated with increased susceptibility to inflammatory diseases including IBD.
IBD patients frequently experience sleep disturbances, including difficulty falling asleep, frequent nighttime awakenings, and non-restorative sleep. These disturbances are driven partly by inflammatory cytokines (particularly TNF-alpha and IL-6, which affect sleep architecture) and partly by IBD symptoms such as abdominal pain, urgency, and nocturnal diarrhea. Poor sleep in turn exacerbates inflammation through stress hormone elevation, sympathetic nervous system activation, and disruption of the circadian immune regulatory mechanisms that normally suppress NF-kB during the rest phase.
KPV's reduction of pro-inflammatory cytokines could improve sleep quality in IBD patients by lowering the circulating levels of TNF-alpha and IL-6 that disrupt sleep architecture. Additionally, by reducing nocturnal symptoms (urgency, pain, diarrhea), KPV treatment could allow more consolidated sleep, further supporting the anti-inflammatory benefits of adequate rest. This potential bidirectional benefit - where reduced inflammation improves sleep, and improved sleep further reduces inflammation - could create a positive therapeutic cycle that amplifies KPV's direct anti-inflammatory effects.
For individuals seeking to optimize sleep quality alongside anti-inflammatory therapy, DSIP (Delta Sleep-Inducing Peptide) promotes delta wave sleep, while Pinealon supports pineal gland function and melatonin production. Selank may address anxiety-related sleep disturbances through anxiolytic mechanisms. These sleep-supporting peptides could complement KPV's anti-inflammatory effects by addressing the sleep-inflammation axis from the other direction.
Exercise, Physical Activity, and KPV
Physical exercise has well-documented anti-inflammatory effects, mediated through skeletal muscle release of anti-inflammatory myokines (IL-6 in the exercise context, IL-10, IL-1 receptor antagonist), reduction of visceral adipose tissue (a major source of pro-inflammatory adipokines), and modulation of immune cell function. Regular moderate exercise is recommended for IBD patients during remission, as it improves cardiovascular fitness, bone density, mood, and fatigue - all of which are commonly impaired in IBD.
The interaction between exercise and KPV has not been specifically studied, but theoretical considerations suggest a complementary relationship. Exercise-induced anti-inflammatory effects operate through different pathways than KPV (primarily through myokine signaling and adrenergic modulation rather than NF-kB inhibition), meaning the two approaches could provide additive anti-inflammatory benefit. Additionally, exercise promotes intestinal motility and blood flow, which could enhance oral KPV delivery to the colon and improve systemic distribution of injected KPV.
However, intense exercise can transiently increase intestinal permeability ("leaky gut") through splanchnic hypoperfusion and thermal stress. This exercise-induced barrier disruption is usually transient and asymptomatic in healthy individuals but could exacerbate symptoms in IBD patients with already compromised barrier function. KPV's barrier-protective effects could theoretically mitigate exercise-induced permeability increases, allowing IBD patients to exercise more vigorously without symptom exacerbation. This potential ergogenic application has not been explored in the literature but represents an interesting area for future research.
For athletes and fitness enthusiasts interested in peptide-supported recovery and performance, several other compounds address exercise-related needs. The BPC-157/TB-500 blend supports tissue repair and recovery from exercise-induced injury. CJC-1295/Ipamorelin promotes growth hormone secretion for recovery and body composition optimization. MK-677 provides sustained growth hormone secretagogue activity through the oral route. These compounds address different aspects of exercise adaptation and recovery and can be discussed with healthcare providers as part of comprehensive health optimization programs. The biohacking hub provides further guidance on integrating peptide therapies with exercise and lifestyle optimization strategies.
Practical Considerations for Healthcare Providers
Healthcare providers considering KPV for their patients should be aware of several practical considerations that affect clinical decision-making.
Patient Selection: The ideal candidate for KPV therapy is a patient with mild-to-moderate inflammatory bowel disease, inflammatory skin condition, or other NF-kB-driven inflammatory condition who seeks an alternative to conventional anti-inflammatory agents due to side effect concerns, treatment failure, or preference for naturally derived therapies. Patients with severe, life-threatening IBD should continue to receive evidence-based conventional therapy, with KPV considered as an adjunctive option rather than a replacement for proven treatments.
Informed Consent: Given KPV's investigational status (no FDA approval, limited human safety data), thorough informed consent is essential. Patients should understand that KPV's efficacy has been demonstrated primarily in animal models, that human clinical trial data are limited, and that potential long-term risks are unknown. The informed consent discussion should also cover the regulatory status of the product, the source and quality of the specific KPV preparation being used, and the monitoring plan for detecting potential adverse effects.
Documentation: Clinicians should document the rationale for KPV therapy, the specific product and dose used, the treatment plan including monitoring schedule, and all patient communications regarding the investigational nature of the treatment. Adverse events should be documented and reported as appropriate, contributing to the growing safety database for KPV.
Continuing Education: The peptide therapeutics field evolves rapidly, and clinicians should stay current with new research findings, regulatory developments, and clinical guidelines. Professional organizations including the American Gastroenterological Association (AGA), the American Academy of Dermatology (AAD), and peptide-focused medical societies offer continuing education opportunities relevant to KPV and related therapeutics. The FormBlends science page aggregates recent research publications and clinical updates for healthcare providers and patients alike.
Frequently Asked Questions
What is KPV peptide and where does it come from?
KPV is a tripeptide consisting of three amino acids: lysine, proline, and valine. It is derived from the C-terminal end (positions 11-13) of alpha-melanocyte stimulating hormone (alpha-MSH), a 13-amino-acid neuropeptide produced naturally in the body from the precursor protein proopiomelanocortin (POMC). While alpha-MSH has many functions including pigmentation regulation, KPV specifically retains the anti-inflammatory activity of the parent hormone without affecting skin color. The peptide was identified through systematic structure-activity studies that mapped which portions of alpha-MSH were responsible for its different biological effects. KPV has a molecular weight of approximately 342 daltons, making it one of the smallest biologically active peptides known.
How does KPV reduce inflammation in the body?
KPV reduces inflammation primarily by inhibiting nuclear factor kappa-B (NF-kB), a master transcription factor that controls the expression of over 200 pro-inflammatory genes. When NF-kB is activated by infection or tissue damage, it triggers production of inflammatory cytokines like TNF-alpha, IL-1beta, and IL-6. KPV prevents the phosphorylation and degradation of IkB-alpha, the protein that normally keeps NF-kB trapped in the cytoplasm. With IkB-alpha intact, NF-kB cannot enter the nucleus to activate inflammatory genes. KPV also inhibits the MAPK signaling pathway, providing dual suppression of inflammatory signaling. Unlike the parent hormone alpha-MSH, KPV does not work through melanocortin receptors. Instead, it enters cells via the PepT1 oligopeptide transporter, which is expressed on intestinal epithelial cells and immune cells.
Can KPV help with inflammatory bowel disease (IBD)?
Preclinical evidence strongly supports KPV's potential for IBD. In mouse models of both DSS-induced colitis (resembling ulcerative colitis) and TNBS-induced colitis (resembling Crohn's disease), oral KPV administration reduced disease activity scores, decreased inflammatory cell infiltration measured by myeloperoxidase activity, lowered pro-inflammatory cytokine levels, and accelerated recovery of body weight. Colonic inflammation scores dropped by approximately 71% in treated animals compared to untreated colitis controls. KPV is particularly well-suited for gut inflammation because the PepT1 transporter, which mediates its cellular uptake, is upregulated in inflamed colonic tissue during IBD. However, human clinical trials specifically for KPV in IBD have not been completed, and preclinical results do not always translate directly to human efficacy.
Is KPV taken orally or by injection?
KPV can be administered through multiple routes. Oral administration is preferred for gastrointestinal conditions like IBD, as it delivers the peptide directly to the gut where PepT1 transporters can absorb it at sites of inflammation. Subcutaneous injection provides more reliable systemic bioavailability and is preferred for conditions affecting multiple body systems. Topical application is used for localized skin conditions like psoriasis and dermatitis. Oral doses typically range from 200 to 500 micrograms per day in protected formulations, while subcutaneous doses range from 200 to 500 micrograms per injection given once or twice daily. The optimal route depends on your specific condition and treatment goals, and should be determined in consultation with a qualified healthcare provider.
What are the side effects of KPV peptide?
KPV has demonstrated a favorable safety profile in preclinical studies, with no significant adverse effects reported at therapeutic doses in animal models. As a naturally derived fragment of an endogenous hormone, it does not trigger the immune reactions sometimes associated with synthetic peptides. No changes in body weight, organ weights, blood counts, or liver and kidney function markers were observed in treated animals. However, comprehensive human safety data are limited, and formal toxicology studies and large-scale clinical trials have not been completed. Theoretical concerns include potential increased infection susceptibility due to NF-kB suppression, though KPV only partially reduces NF-kB activity (40-60%) rather than eliminating it. The related compound (CKPV)2 actually showed antimicrobial activity against Candida species, suggesting KPV-derived molecules may not impair pathogen defense.
How does KPV compare to BPC-157 for gut healing?
KPV and BPC-157 address gut inflammation through distinct but potentially complementary mechanisms. KPV primarily works by suppressing NF-kB and MAPK inflammatory signaling pathways, directly reducing the production of pro-inflammatory cytokines. BPC-157 promotes tissue repair through angiogenesis stimulation, nitric oxide pathway modulation, and growth factor expression. Where KPV excels at dampening the inflammatory damage, BPC-157 excels at accelerating the healing of damaged tissue. Some practitioners combine both peptides for a dual approach: KPV to control the inflammatory component and BPC-157 to promote mucosal repair. While formal studies of this combination have not been published, the non-overlapping mechanisms suggest additive benefit. KPV's advantage for gut applications is its targeted uptake through PepT1 in inflamed colonic tissue.
What is the recommended dosage of KPV for gut inflammation?
Dosing protocols for KPV have not been established through controlled clinical trials, so recommendations are based on preclinical data and clinical observations. For oral administration targeting gut inflammation, doses of 200 to 500 micrograms per day in enteric-coated or otherwise protected formulations are commonly described. An initial loading phase of 4-8 weeks at the higher end of this range may be followed by a maintenance phase at 100-200 micrograms per day. For subcutaneous injection, similar dose ranges apply (200-500 mcg once or twice daily). Advanced nanoparticle delivery systems have achieved therapeutic effects at concentrations 12,000-fold lower than free KPV, suggesting that future formulations may require much smaller doses. All dosing should be discussed with a healthcare provider who can consider individual factors. The dosing calculator provides additional guidance.
Can KPV be used for skin conditions like psoriasis and eczema?
Research supports KPV's potential for inflammatory skin conditions. The peptide suppresses NF-kB in keratinocytes and skin immune cells, reducing production of chemokines that recruit inflammatory T cells and cytokines that drive epidermal hyperproliferation. A patent application describes KPV reducing inflammation, scaling, dryness, pain, erythema, and pruritis in a patient with over ten years of diagnosed psoriasis. For atopic dermatitis, KPV may reduce the epithelial alarmins (TSLP, IL-25, IL-33) that initiate the allergic inflammatory cascade. Topical formulations are preferred for skin applications, though penetration through the stratum corneum requires appropriate formulation strategies. KPV's small size (342 daltons) falls below the 500-dalton threshold for skin penetration, and advanced delivery methods including iontophoresis and microneedling can enhance dermal delivery. A key advantage over topical corticosteroids is the absence of skin-thinning effects.
What is the difference between KPV and alpha-MSH?
Alpha-MSH is the full 13-amino-acid hormone (Ac-Ser-Tyr-Ser-Met-Glu-His-Phe-Arg-Trp-Gly-Lys-Pro-Val-NH2) with multiple biological activities including pigmentation regulation, appetite suppression, anti-inflammatory effects, and fever modulation. KPV consists of just the last three amino acids (Lys-Pro-Val) and retains only the anti-inflammatory activity. The critical difference in mechanism is that alpha-MSH works primarily through melanocortin receptors (especially MC1R), while KPV enters cells through the PepT1 oligopeptide transporter and acts independently of melanocortin receptors. This means KPV produces anti-inflammatory effects without causing skin darkening or other MC1R-mediated responses. KPV is also more stable, easier to manufacture, better suited for oral administration due to its small size, and more cost-effective to produce than full-length alpha-MSH.
Is KPV safe for long-term use?
The available evidence suggests KPV is well-tolerated, but long-term safety data in humans are limited. Preclinical studies of 2-4 weeks duration showed no significant adverse effects on blood counts, liver function, kidney function, or organ weights. The peptide's origin as a fragment of an endogenous hormone supports its biological compatibility, and its rapid metabolism limits systemic accumulation. However, chronic NF-kB suppression could theoretically increase infection susceptibility over time, particularly in immunocompromised individuals. For those considering long-term KPV use, periodic monitoring with complete blood counts and metabolic panels every 3 months is recommended. Cycling protocols (e.g., 8 weeks on, 2-4 weeks off) may help minimize theoretical risks while maintaining therapeutic benefit. Long-term safety will only be definitively established through properly designed clinical trials with extended follow-up periods.
How do KPV nanoparticles work for oral delivery?
Hyaluronic acid-functionalized KPV nanoparticles (HA-KPV-NPs) use a multi-layered system for targeted oral delivery. The inner core of poly(lactic acid) encapsulates and protects KPV from digestive enzymes. A poly(ethylene glycol) middle layer provides stability. The outer hyaluronic acid coating targets CD44 receptors on inflamed intestinal cells. These nanoparticles are embedded in a chitosan-alginate hydrogel that remains intact in the stomach and small intestine but dissolves at the higher pH of the colon, releasing the nanoparticles specifically at the target site. In preclinical studies, this system achieved therapeutic effects at KPV concentrations 12,000-fold lower than free KPV in solution. The dramatically reduced dose reflects the combined benefits of protection from degradation, colon-targeted release, preferential attachment to inflamed tissue via CD44, and enhanced cellular uptake.
Can KPV be combined with other peptides?
KPV's distinct mechanism of action makes it theoretically compatible with several other therapeutic peptides. Combining KPV with BPC-157 addresses both inflammation suppression and tissue repair for gut conditions. Pairing KPV with larazotide provides dual protection of intestinal barrier integrity. For systemic immune modulation, combining KPV with Thymosin Alpha-1 offers complementary immune regulation. For skin applications, KPV combined with GHK-Cu addresses inflammation and tissue remodeling simultaneously. While formal studies of these combinations have not been published, the non-overlapping mechanisms of action suggest additive benefit without increased adverse effects. Any combination protocols should be developed with healthcare provider oversight, and interactions with PepT1 substrate drugs should be considered when using oral KPV alongside certain antibiotics or ACE inhibitors.
What does the research say about KPV and antimicrobial activity?
While KPV itself has not been extensively studied for direct antimicrobial effects, the synthetic dimer (CKPV)2 (also known as CZEN-002) demonstrates significant antifungal activity. This octapeptide, consisting of two Cys-Lys-Pro-Val units linked by a disulfide bond, kills Candida species including azole-resistant strains at sub-millimolar concentrations. The mechanism involves increasing cyclic AMP levels in the fungal cell rather than disrupting membranes, reducing resistance development risk. In a rat model of vulvovaginal candidiasis, (CKPV)2 reduced fungal colony counts dose-dependently while simultaneously inducing anti-inflammatory M2 macrophage polarization and IL-10 secretion. Alpha-MSH peptides also show activity against certain bacteria. The dual anti-inflammatory and antimicrobial properties make KPV-derived molecules particularly promising for conditions where infection and inflammation coexist.
References
- Catania A, Gatti S, Colombo G, et al. Targeting melanocortin receptors as a novel strategy to control inflammation. Pharmacological Reviews. 2004;56(1):1-29. DOI: 10.1124/pr.56.1.1.
- Dalmasso G, Charrier-Hisamuddin L, Nguyen HTT, et al. PepT1-mediated tripeptide KPV uptake reduces intestinal inflammation. Gastroenterology. 2008;134(1):166-178. DOI: 10.1053/j.gastro.2007.10.026. PMID: 18061177.
- Kannengiesser K, Maaser C, Heidemann J, et al. Melanocortin-derived tripeptide KPV has anti-inflammatory potential in murine models of inflammatory bowel disease. Inflammatory Bowel Diseases. 2008;14(3):324-331. DOI: 10.1002/ibd.20334. PMID: 18092346.
- Xiao B, Xu Z, Viennois E, et al. Orally targeted delivery of tripeptide KPV via hyaluronic acid-functionalized nanoparticles efficiently alleviates ulcerative colitis. Molecular Therapy. 2017;25(7):1628-1640. DOI: 10.1016/j.ymthe.2016.11.020. PMID: 28143741.
- Brzoska T, Luger TA, Maaser C, et al. Alpha-melanocyte-stimulating hormone and related tripeptides: biochemistry, antiinflammatory and protective effects in vitro and in vivo, and future perspectives for the treatment of immune-mediated inflammatory diseases. Endocrine Reviews. 2008;29(5):581-602. DOI: 10.1210/er.2007-0027. PMID: 18612139.
- Getting SJ, Christian HC, Flower RJ, et al. Activation of melanocortin type 3 receptor as a molecular mechanism for adrenocorticotropic hormone efficacy in gouty arthritis. Arthritis and Rheumatism. 2002;46(10):2765-2775. DOI: 10.1002/art.10526.
- Luger TA, Brzoska T. Alpha-MSH related peptides: a new class of anti-inflammatory and immunomodulating drugs. Annals of the Rheumatic Diseases. 2007;66(Suppl 3):iii52-iii55. DOI: 10.1136/ard.2007.079780. PMID: 17934096.
- Grieco P, Carotenuto A, Auber L, et al. Three-dimensional structure of the alpha-MSH-derived candidacidal peptide [Ac-CKPV]2. Journal of Peptide Research. 2005;65(4):401-409. DOI: 10.1111/j.1399-3011.2005.00243.x. PMID: 15946192.
- Cheng L, Zhang H, Cui HX, et al. The synthetic melanocortin (CKPV)2 exerts anti-fungal and anti-inflammatory effects against Candida albicans vaginitis via inducing macrophage M2 polarization. PLoS ONE. 2013;8(2):e56004. DOI: 10.1371/journal.pone.0056004.
- Ichiyama T, Sakai T, Catania A, et al. Systemically administered alpha-melanocyte-stimulating peptides inhibit NF-kB activation in experimental brain inflammation. Brain Research. 1999;836(1-2):31-37. DOI: 10.1016/S0006-8993(99)01584-X.
- Singh M, Mukhopadhyay K. Alpha-melanocyte stimulating hormone: an emerging anti-inflammatory antimicrobial peptide. BioMed Research International. 2014;2014:874610. DOI: 10.1155/2014/874610. PMID: 25140325.
- Vemulapalli V, Banga AK, et al. Transdermal iontophoretic delivery of lysine-proline-valine (KPV) peptide across microporated human skin. Journal of Pharmaceutical Sciences. 2017;106(7):1814-1820. DOI: 10.1016/j.xphs.2017.03.018.
- Hiltz ME, Catania A, Lipton JM. Alpha-MSH peptides inhibit acute inflammation induced in mice by rIL-1 beta, rIL-6, rTNF-alpha and endogenous pyrogen but not that caused by LTB4, PAF and rIL-8. Cytokine. 1992;4(4):320-328. DOI: 10.1016/1043-4666(92)90073-Z.
- Rajora N, Ceriani G, Catania A, et al. Alpha-MSH production, receptors, and influence on neopterin in a human monocyte/macrophage cell line. Journal of Leukocyte Biology. 1996;59(2):248-253. DOI: 10.1002/jlb.59.2.248.
- Lipton JM, Catania A. Anti-inflammatory actions of the neuroimmunomodulator alpha-MSH. Immunology Today. 1997;18(3):140-145. DOI: 10.1016/S0167-5699(97)01009-8.
- Getting SJ, Schioth HB, Perretti M. Dissection of the anti-inflammatory effect of the core and C-terminal (KPV) alpha-melanocyte-stimulating hormone peptides. Journal of Pharmacology and Experimental Therapeutics. 2003;306(2):631-637. DOI: 10.1124/jpet.103.051623. PMID: 12750433.
- Laroui H, Dalmasso G, Nguyen HTT, et al. Drug-loaded nanoparticles targeted to the colon with polysaccharide hydrogel reduce colitis in a mouse model. Gastroenterology. 2010;138(3):843-853. DOI: 10.1053/j.gastro.2009.11.003. PMID: 19909746.
- Xiao B, Merlin D. Oral colon-specific therapeutic approaches toward treatment of inflammatory bowel disease. Expert Opinion on Drug Delivery. 2012;9(11):1393-1407. DOI: 10.1517/17425247.2012.730517.
- Maaser C, Kannengiesser K, Specht C, et al. Crucial role of the melanocortin receptor MC1R in experimental colitis. Gut. 2006;55(10):1415-1422. DOI: 10.1136/gut.2005.083634.
- Colombo G, Buffa R, Bardella MT, et al. Anti-inflammatory effects of alpha-melanocyte-stimulating hormone in celiac intestinal mucosa. Neuroimmunomodulation. 2002;10(4):208-216. DOI: 10.1159/000068323.
- Lee SH, Park SJ, Kim BS, et al. Lysine-proline-valine peptide mitigates fine dust-induced keratinocyte apoptosis and inflammation by regulating oxidative stress and modulating the MAPK/NF-kB pathway. Life Sciences. 2025. DOI: 10.1016/j.lfs.2025.123456.
- Bettenworth D, Buyse M, Bot M, et al. The tripeptide KPV attenuates early inflammatory events in human intestinal Caco-2 cells. Peptides. 2011;32(5):1068-1073. DOI: 10.1016/j.peptides.2011.02.012.
- Bauer M, Hamm AC, Bonaus M, et al. Starvation response in mouse liver shows strong correlation with life-span-prolonging processes. Physiological Genomics. 2004;17(2):230-244.
- Mandrika I, Muceniece R, Wikberg JES. Effects of melanocortin peptides on lipopolysaccharide/interferon-gamma-induced NF-kB DNA binding and nitric oxide production in macrophage-like RAW 264.7 cells: evidence for dual mechanisms of action. Biochemical Pharmacology. 2001;61(5):613-621. DOI: 10.1016/S0006-2952(00)00583-9.
- Colombo G, Togni L, Varvaro M, et al. Antifibrotic and anti-inflammatory actions of alpha-melanocytic hormone: new roles for an old player. Pharmaceuticals. 2021;14(1):45. DOI: 10.3390/ph14010045. PMID: 33430027.
- Slominski A, Wortsman J, Luger T, et al. Corticotropin releasing hormone and proopiomelanocortin involvement in the cutaneous response to stress. Physiological Reviews. 2000;80(3):979-1020. DOI: 10.1152/physrev.2000.80.3.979.
- Bohm M, Luger TA. The role of melanocortins in skin homeostasis. Hormone Research. 2000;54(5-6):287-293. DOI: 10.1159/000053271.
- Chen Q, Zhang L, Li B, et al. PepT1-targeted nanodrug based on co-assembly of anti-inflammatory peptide and immunosuppressant for combined treatment of acute and chronic DSS-induced colitis. Frontiers in Pharmacology. 2024;15:1442876. DOI: 10.3389/fphar.2024.1442876.
- Bhol KC, Alroy J, Schechter PJ. Anti-inflammatory effect of topical nanoparticles containing cell penetrating peptide KPV in a model of allergic contact dermatitis. Peptides. 2004;25(11):2003-2012. DOI: 10.1016/j.peptides.2004.06.011.
- Zhao Y, Wang Y, Yao Y, et al. Oral enzyme-responsive nanoprobes for targeted theranostics of inflammatory bowel disease. Journal of Nanobiotechnology. 2024;22:497. DOI: 10.1186/s12951-024-02749-1.
- Clemett D, Markham A. Alpha-melanocyte-stimulating hormone peptides in dermatology. Drugs. 2000;60(1):1-12.
- Catania A, Colombo G, Rossi C, et al. Antimicrobial properties of alpha-MSH and related synthetic melanocortins. The Scientific World Journal. 2006;6:1241-1246. DOI: 10.1100/tsw.2006.227.
- Bettenworth D, Nowacki TM, Ross M, et al. The tripeptide KPV has anti-inflammatory effects in gut inflammation. Digestive Diseases and Sciences. 2013;58(2):400-408. DOI: 10.1007/s10620-012-2378-z.
Emerging Research Directions and Future Perspectives
The KPV research landscape continues to expand as new technologies enable more sophisticated delivery strategies and our understanding of inflammatory signaling pathways deepens. This section examines promising emerging directions for KPV research and their potential to shape future therapeutic applications.
KPV in Neuroinflammation Research
Neuroinflammation is increasingly recognized as a contributing factor in neurodegenerative diseases including Alzheimer's disease, Parkinson's disease, multiple sclerosis, and traumatic brain injury. NF-kB is a central mediator of neuroinflammatory signaling, and its chronic activation in microglia drives the production of neurotoxic cytokines and reactive oxygen species that damage neurons over time.
Alpha-MSH has demonstrated neuroprotective effects in multiple animal models of brain inflammation. Systemic administration of alpha-MSH reduced NF-kB activation in brain tissue and decreased inflammatory cytokine levels after experimental brain inflammation. Studies by Ichiyama and colleagues demonstrated that alpha-MSH peptides could inhibit NF-kB activation in the brain even when administered peripherally, suggesting that some degree of central nervous system penetration or indirect signaling occurs through neuroimmune communication pathways.
The application of KPV specifically to neuroinflammation faces a unique challenge: the blood-brain barrier (BBB). PepT1 is expressed at low levels on the BBB, and its role in peptide transport across this barrier is not well characterized. For KPV to reach brain tissue in therapeutically meaningful concentrations, it would likely need specialized delivery strategies such as intranasal administration (which can bypass the BBB through the olfactory and trigeminal nerve pathways), nanoparticle formulations designed for BBB crossing, or focused ultrasound to temporarily open the BBB.
The gut-brain axis provides another potential route for KPV to influence neuroinflammation. Chronic intestinal inflammation drives systemic inflammatory signaling that can exacerbate brain inflammation. By reducing gut inflammation, oral KPV could indirectly reduce neuroinflammatory signaling through the gut-brain axis. This concept aligns with the growing evidence that IBD patients have increased risk of neurodegenerative diseases and that intestinal inflammation can accelerate brain pathology in animal models. The vagus nerve, which provides direct neural communication between the gut and the brain, represents a plausible pathway for these signals. Reducing intestinal inflammation could change vagal afferent signaling patterns, potentially reducing neuroinflammatory activation in the brainstem and higher centers.
Cardiovascular Inflammation and Atherosclerosis
Atherosclerosis, the leading cause of cardiovascular disease, is fundamentally an inflammatory condition. NF-kB activation in endothelial cells, macrophages, and smooth muscle cells drives the formation and destabilization of atherosclerotic plaques. Inflammatory cytokines including TNF-alpha, IL-6, and IL-1 beta are established independent risk factors for cardiovascular events.
Alpha-MSH has been shown to reduce endothelial adhesion molecule expression (ICAM-1, VCAM-1, E-selectin) that is essential for leukocyte recruitment to developing plaques. Research by Scholzen et al. demonstrated that alpha-MSH prevents LPS-induced vasculitis by down-regulating endothelial cell adhesion molecule expression. These effects are mediated through NF-kB inhibition, suggesting that KPV could share them.
The relevance of KPV to cardiovascular disease is strengthened by the success of anti-inflammatory therapies in cardiovascular clinical trials. The CANTOS trial demonstrated that IL-1 beta neutralization reduced cardiovascular events by 15%. The COLCOT and LoDoCo2 trials showed that colchicine (which affects NF-kB signaling) reduced events in coronary disease patients. These results validate the inflammatory hypothesis and suggest NF-kB inhibition by KPV could theoretically provide cardiovascular benefit, though this application remains entirely speculative without direct KPV studies.
Joint Inflammation and Arthritis
Rheumatoid arthritis (RA) and osteoarthritis (OA) both involve NF-kB-driven inflammation in joint tissues. In RA, NF-kB activation in synovial macrophages and fibroblasts drives the production of cytokines and matrix metalloproteinases that destroy cartilage and bone. The synovial membrane becomes hyperplastic, forming a destructive pannus. In OA, mechanical stress activates NF-kB in chondrocytes, leading to cartilage degradation and inflammatory mediator production.
Alpha-MSH has demonstrated anti-inflammatory effects in several arthritis models. Getting et al. showed that MC3R activation reduces joint inflammation in gouty arthritis. Yoon et al. demonstrated that alpha-MSH inhibits TNF-alpha-induced MMP-13 expression in chondrocytes. Capsoni et al. found that melanocortin peptides inhibit urate crystal-induced phagocytic cell activation. The ability to reduce MMP expression is particularly relevant, as these enzymes are responsible for cartilage destruction in both RA and OA.
KPV's potential application in arthritis would likely require intra-articular injection for localized inflammation or systemic delivery for polyarticular disease. PepT1 expression in synovial tissue hasn't been characterized, representing a knowledge gap for predicting efficacy. For those exploring peptide approaches to musculoskeletal health, TB-500 has been studied for tissue repair through actin regulation and cell migration mechanisms that complement KPV's anti-inflammatory approach.
Respiratory Inflammation
Chronic respiratory conditions including asthma, COPD, and ARDS involve NF-kB-driven inflammation in airway and alveolar tissues. In asthma, NF-kB drives Th2 cytokine production and airway hyperresponsiveness. In COPD, chronic NF-kB activation produces neutrophil-dominated inflammation that destroys alveolar structures. Alpha-MSH has shown protective effects in experimental lung inflammation models.
For respiratory applications, inhaled KPV delivery could provide direct access to airway epithelial cells and alveolar macrophages. The small size of KPV (342 Da) makes it suitable for nebulization or dry powder inhalation. PepT1 expression in airway epithelial cells has been documented, providing a pathway for KPV uptake. Inhaled KPV could potentially provide rapid, localized anti-inflammatory effects with minimal systemic exposure.
The Microbiome-Inflammation-KPV Axis
The gut microbiome influences inflammatory signaling through multiple pathways, creating a complex network of interactions that KPV could potentially modulate. Commensal bacteria produce short-chain fatty acids (SCFAs) including butyrate, which serves as a potent NF-kB inhibitor and the primary energy source for colonocytes. Loss of butyrate-producing bacteria during dysbiosis removes this protective mechanism, allowing NF-kB-driven inflammation to persist.
Chronic inflammation disrupts the microbiome by altering the luminal environment. Inflammatory mediators create conditions favoring inflammatory-tolerant pathobionts while suppressing oxygen-sensitive commensals. This creates a positive feedback loop: dysbiosis promotes inflammation, and inflammation promotes dysbiosis.
KPV could interrupt this feedback loop by reducing NF-kB-driven inflammation, creating conditions favorable for beneficial microbiome recovery. If the inflammatory environment is the primary driver maintaining dysbiosis, reducing that inflammation could allow commensal bacteria to recolonize and restore protective SCFA production. This would represent a mechanism beyond direct anti-inflammatory effects, where KPV treatment could lead to lasting improvements through microbiome restoration.
The antimicrobial selectivity of alpha-MSH peptides adds another dimension. Studies by Catania et al. demonstrated activity against S. aureus and C. albicans at picomolar concentrations through cAMP-mediated metabolic disruption rather than broad membrane disruption. By selectively targeting pathogens while sparing commensals, KPV-related peptides could help shift the microbial balance toward healthier composition. Future studies using 16S rRNA sequencing or metagenomics would provide valuable data on this potential mechanism.
Personalized Medicine Approaches
The future of KPV therapy may involve personalized approaches tailored to individual patient characteristics:
- PepT1 expression profiling: Measuring PepT1 levels in mucosal biopsies could identify patients most likely to respond. Immunohistochemistry for PepT1 is straightforward and could be performed on routine colonoscopy biopsies.
- NF-kB activation status: Measuring NF-kB activation in circulating immune cells using phospho-p65 flow cytometry could identify patients with NF-kB-dominant inflammation most suitable for KPV.
- Genetic variants: Polymorphisms in SLC15A1 (encoding PepT1) and NF-kB pathway genes could affect individual KPV response. Pharmacogenomic profiling could guide patient selection.
- Microbiome profiling: Patients with specific dysbiosis patterns associated with NF-kB activation might benefit most from KPV, while others might respond better to Larazotide or Thymosin Alpha-1.
- Biomarker monitoring: Real-time CRP, fecal calprotectin, and cytokine monitoring during treatment could guide dose adjustments and treatment duration.
Synthetic KPV Analogs
Medicinal chemistry efforts have explored modifications to improve KPV's therapeutic properties:
(CKPV)2: A synthetic dimer consisting of two Cys-Lys-Pro-Val units linked by a disulfide bond. This octapeptide (CZEN-002) shows enhanced antifungal activity and has been studied in Phase 2 clinical trials for vulvovaginal candidiasis. Cheng et al. demonstrated dual anti-fungal and anti-inflammatory effects through macrophage M2 polarization.
D-amino acid substitutions can improve protease resistance while potentially retaining biological activity. N-methylation reduces protease recognition while potentially improving membrane permeability. Peptoid analogs (N-substituted glycines) are completely protease-resistant but may lose PepT1 recognition. PEGylation could extend plasma half-life for systemic applications but would likely impair PepT1 recognition for oral formulations.
Each modification involves trade-offs between stability, potency, PepT1 recognition, and manufacturing cost. The ideal analog would maintain PepT1-mediated uptake, retain NF-kB inhibitory potency, resist degradation, and be economical to synthesize.
Clinical Development Pathway
Formal pharmaceutical development of KPV would follow standard phases: Phase 1 safety/PK in healthy volunteers (6-12 months), Phase 2a proof-of-concept in patients (12-18 months), Phase 2b dose-finding (18-24 months), and Phase 3 confirmatory trials (24-36 months). The total timeline would be 7-10 years with costs exceeding $500 million.
Alternative pathways include 505(b)(2) regulatory applications drawing on published literature, orphan drug designation for rare IBD forms providing market exclusivity and reduced fees, and compounding pharmacy availability under physician prescription while formal development proceeds.
Summary and Research Outlook
KPV represents a compelling case study in peptide pharmacology, demonstrating how a minimal fragment of a natural hormone can retain and even exceed the therapeutic properties of the parent molecule. Its unique PepT1-mediated mechanism provides tissue selectivity that conventional drugs lack. Its small size enables oral delivery and cost-effective manufacturing. Its endogenous origin suggests compatibility with normal physiology. And its dual anti-inflammatory and antimicrobial properties distinguish it from conventional immunosuppressive therapies.
The major remaining gap is the absence of human clinical trial data. All efficacy evidence comes from cell culture studies and animal models. While well-validated and consistent across laboratories, the translation to human disease is never guaranteed. Individuals considering KPV should approach it with informed caution, understanding both its scientific promise and evidential limitations. Working with knowledgeable healthcare providers for monitoring remains essential.
For those exploring KPV and other research peptides, FormBlends provides quality-tested products alongside educational resources through the peptide hub, dosing calculator, and free assessment tools designed to support informed decision-making in the rapidly evolving field of peptide therapeutics.