Executive Summary
Injectable L-carnitine and lipotropic injection compounds have become increasingly popular tools in clinical weight management, athletic performance, and metabolic optimization programs. This report examines the scientific evidence behind these formulations, their pharmacokinetics, and how they compare to oral alternatives.
Key Takeaways
- Injectable L-carnitine achieves roughly 95-100% bioavailability versus 5-18% for oral supplemental doses
- L-carnitine is essential for mitochondrial long-chain fatty acid transport via the CPT1/CPT2 carnitine shuttle system
- MIC (methionine, inositol, choline) lipotropic injections support hepatic fat metabolism through complementary pathways
- Injectable L-carnitine bypasses the gut microbiome, potentially reducing TMAO production linked to cardiovascular concerns with oral supplementation
- Clinical evidence supports L-carnitine for cardiovascular protection, male fertility, and exercise recovery, with more mixed results for direct fat loss
L-carnitine is a naturally occurring amino acid derivative that plays a central role in mitochondrial fatty acid transport. Without adequate carnitine, long-chain fatty acids simply cannot enter the mitochondrial matrix for beta-oxidation. Your body makes some of it from lysine and methionine, and you get the rest from dietary sources like red meat and dairy. But here's the catch: oral L-carnitine has remarkably poor bioavailability, typically between 5% and 18% for supplemental doses. That's where injectable formulations change the equation entirely.
When L-carnitine is administered via intramuscular or subcutaneous injection, bioavailability jumps to near 100%. This isn't a marginal improvement. It's a dramatic pharmacokinetic advantage that eliminates the absorption ceiling imposed by intestinal transporters and avoids first-pass metabolism. For patients pursuing fat loss, enhanced exercise recovery, or metabolic support, this difference can be clinically meaningful.
Lipotropic injections take the concept further by combining multiple compounds that support hepatic fat metabolism. The classic MIC formulation pairs methionine, inositol, and choline, often with added vitamin B12 and sometimes L-carnitine itself. These "lipo shots" have become a staple in weight loss clinics across the country, though the clinical evidence supporting their use as a combined injectable formulation remains limited compared to the research on individual components.
Key Takeaways
- Injectable L-carnitine achieves roughly 95-100% bioavailability versus 5-18% for oral supplemental doses
- L-carnitine is essential for mitochondrial long-chain fatty acid transport via the CPT1/CPT2 carnitine shuttle system
- MIC (methionine, inositol, choline) lipotropic injections support hepatic fat metabolism through complementary pathways
- Injectable L-carnitine bypasses the gut microbiome, potentially reducing TMAO production linked to cardiovascular concerns with oral supplementation
- Clinical evidence supports L-carnitine for cardiovascular protection, male fertility, and exercise recovery, with more mixed results for direct fat loss
- Lipotropic injections are compounded formulations that are not FDA-approved for weight loss
This report covers the biochemistry of L-carnitine and its role in fatty acid oxidation, compares oral and injectable bioavailability data, reviews the evidence for fat metabolism enhancement, athletic performance, cardiovascular protection, and male fertility. We also break down the individual components of lipotropic MIC injections, review dosing protocols used in clinical practice, and examine the safety profile including the TMAO controversy. Throughout, we'll explore how these compounds can complement peptide-based therapies such as AOD-9604, semaglutide, and other metabolic peptides available through research programs.
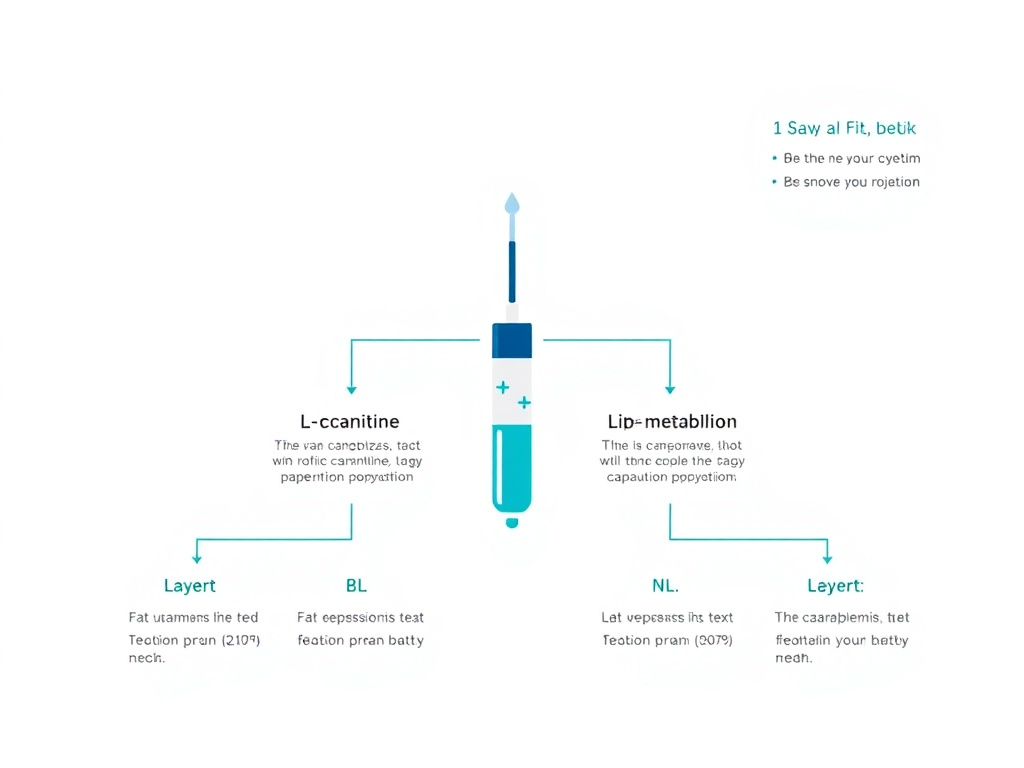
Figure 1: Overview of injectable L-carnitine and lipotropic compound pathways in fat metabolism support.
Historical Context and Clinical Adoption
The clinical use of injectable L-carnitine has a longer history than many people realize. Intravenous levocarnitine was first approved by the FDA in 1999 for the treatment of carnitine deficiency in end-stage renal disease patients on dialysis. In this population, carnitine is continuously lost through dialysis membranes while endogenous synthesis is impaired by kidney dysfunction. The approval was based on well-designed clinical trials demonstrating that IV carnitine corrected deficiency states, improved erythropoiesis, and reduced the need for exogenous erythropoietin in dialysis patients.
The use of injectable L-carnitine in weight management and sports medicine settings developed somewhat independently, driven by clinicians who recognized the bioavailability advantage of parenteral administration and the logical connection between carnitine status and fat oxidation efficiency. While these applications lack the same level of regulatory validation as the dialysis indication, they're built on the same pharmacological foundation and supported by a growing body of clinical experience.
Lipotropic injections have an even longer informal history. The concept of lipotropic factors dates back to the 1930s, when researchers at the University of Toronto discovered that choline and methionine could prevent fatty liver in animals fed high-fat diets. Charles Best, who co-discovered insulin with Frederick Banting, was a pioneer in this research. The transition from these basic science observations to clinical lipotropic injection formulations occurred gradually over subsequent decades, with compounding pharmacies developing standardized MIC formulations by the 1970s and 1980s.
Today, injectable L-carnitine and lipotropic injections represent one of the most widely prescribed categories of compounded injectable products in the weight management sector. The market has expanded significantly as interest in peptide-based therapies and personalized metabolic medicine has grown. Many patients who begin with GLP-1 receptor agonist therapy subsequently explore adjunctive compounds like L-carnitine to optimize their metabolic outcomes.
Who May Benefit Most from Injectable L-Carnitine?
While the general population can synthesize and absorb adequate carnitine from a typical omnivorous diet, several specific populations may derive particular benefit from injectable L-carnitine supplementation:
Individuals on caloric restriction. During aggressive caloric restriction, especially very-low-calorie diets (VLCDs) or programs incorporating GLP-1 agonists that substantially reduce food intake, dietary carnitine intake can drop below maintenance levels. This is particularly relevant because caloric restriction simultaneously increases the metabolic demand for fatty acid oxidation. Ensuring adequate carnitine availability through injectable supplementation helps maintain metabolic efficiency during weight loss.
Vegetarians and vegans. Plant-based diets provide very little dietary carnitine, since the primary food sources are red meat and dairy products. While endogenous synthesis can maintain adequate levels in most healthy vegetarians, some may develop subclinical carnitine insufficiency, particularly if physical activity levels are high or if cofactors for carnitine synthesis (iron, vitamin C, B6, niacin) are also suboptimal. Injectable supplementation offers a reliable way to optimize carnitine status without relying on dietary animal products.
Individuals with genetic variations in carnitine transport. Polymorphisms in the OCTN2 transporter gene (SLC22A5) can reduce carnitine absorption and retention. Severe mutations cause primary carnitine deficiency, a rare but serious condition. More common mild variants may create subclinical insufficiency that becomes clinically relevant under metabolic stress. Injectable administration bypasses the absorption step entirely.
Patients on medications that deplete carnitine. Several commonly prescribed medications reduce carnitine levels, including valproic acid (used for epilepsy and bipolar disorder), pivalic acid-containing antibiotics, and certain chemotherapy agents. For patients on these medications who are also pursuing weight management or athletic performance goals, injectable carnitine supplementation can offset medication-induced depletion.
Older adults. Skeletal muscle carnitine content declines with aging, coinciding with reduced mitochondrial function and increased risk of sarcopenia. Older adults also tend to absorb dietary nutrients less efficiently. Injectable carnitine may help maintain metabolic function and support physical performance in this population.
Athletes with high training volumes. Intense exercise increases carnitine utilization and may temporarily deplete muscle stores. While healthy athletes eating adequate diets generally maintain normal carnitine levels, those undergoing particularly demanding training blocks or those following restricted diets for weight-class sports may benefit from supplementation.
The common thread among these populations is that either carnitine supply is reduced, carnitine demand is increased, or both. Injectable administration ensures that the supplemented carnitine actually reaches the bloodstream in predictable quantities, regardless of individual variations in intestinal absorption.
L-Carnitine Biology
What Is L-Carnitine?
L-carnitine (beta-hydroxy-gamma-N-trimethylaminobutyric acid) is a quaternary ammonium compound synthesized endogenously from the essential amino acids lysine and methionine. The biosynthetic process requires several cofactors including vitamin C (ascorbic acid), iron, niacin, and vitamin B6. Approximately 75% of the body's carnitine comes from dietary intake, with the remaining 25% produced primarily in the liver and kidneys.
The name "carnitine" derives from the Latin word carnis, meaning flesh or meat, reflecting the compound's original isolation from meat extract in 1905 by Russian scientists Gulewitsch and Krimberg. It wasn't until the 1960s, however, that Fritz and Bremer independently established carnitine's essential role in fatty acid oxidation, a discovery that transformed our understanding of lipid metabolism.
Total body carnitine stores in a healthy adult typically range from 20 to 25 grams, with roughly 98% concentrated in skeletal and cardiac muscle tissue. Plasma carnitine levels normally fall between 40 and 60 micromoles per liter. The body maintains carnitine homeostasis through a combination of dietary absorption, endogenous synthesis, and efficient renal reabsorption that recovers over 95% of filtered carnitine.
The Carnitine Shuttle System: CPT1, CACT, and CPT2
The primary biological function of L-carnitine is to facilitate the transport of long-chain fatty acids (those with 14 or more carbon atoms) across the inner mitochondrial membrane for beta-oxidation. This process involves a sophisticated multi-step transport mechanism known as the carnitine shuttle system, comprising three key components.
CPT1 (Carnitine Palmitoyltransferase I) - The Gatekeeper
CPT1 sits on the outer mitochondrial membrane and catalyzes the first and rate-limiting step of long-chain fatty acid entry into the mitochondria. It transfers the acyl group from acyl-CoA to L-carnitine, forming acylcarnitine. This reaction is essentially the "entry ticket" for fatty acids to begin their journey into the mitochondrial matrix.
There are three tissue-specific isoforms of CPT1. CPT1A is expressed predominantly in the liver. CPT1B predominates in skeletal and cardiac muscle. CPT1C is found in the brain and has a less well-characterized role. The existence of tissue-specific isoforms allows for fine-tuned regulation of fatty acid oxidation in different metabolic contexts.
CPT1 is allosterically inhibited by malonyl-CoA, the first committed intermediate of fatty acid synthesis. This regulatory mechanism creates an elegant metabolic switch: when malonyl-CoA levels are high (signaling that the cell is in a fed, anabolic state and synthesizing fat), CPT1 activity drops and fatty acid oxidation slows. When malonyl-CoA levels fall (during fasting, exercise, or caloric restriction), CPT1 becomes active and fatty acid oxidation increases. This is one reason why exercise and caloric restriction enhance fat burning, and it's also why L-carnitine availability can become rate-limiting during periods of high fatty acid flux.
CACT (Carnitine-Acylcarnitine Translocase) - The Transporter
Once CPT1 forms the acylcarnitine conjugate, CACT facilitates its translocation across the inner mitochondrial membrane. This is an antiport mechanism: for every acylcarnitine molecule moved into the matrix, one free carnitine molecule is shuttled back out to the intermembrane space. The inner mitochondrial membrane is impermeable to long-chain acyl-CoA molecules, making this translocase absolutely essential for beta-oxidation.
CPT2 (Carnitine Palmitoyltransferase II) - The Releaser
Located on the inner face of the inner mitochondrial membrane, CPT2 performs the reverse reaction of CPT1. It converts acylcarnitine back to acyl-CoA, releasing free L-carnitine in the process. The regenerated acyl-CoA then enters the beta-oxidation spiral, while the free carnitine is recycled back through CACT to participate in another round of fatty acid transport.
The Complete Carnitine Shuttle in Five Steps
- Activation: Long-chain fatty acids are activated to acyl-CoA on the outer mitochondrial membrane by acyl-CoA synthetase
- Conjugation: CPT1 transfers the acyl group from CoA to carnitine, forming acylcarnitine
- Translocation: CACT moves acylcarnitine across the inner membrane while simultaneously transporting free carnitine outward
- Release: CPT2 reconverts acylcarnitine to acyl-CoA inside the mitochondrial matrix
- Oxidation: Acyl-CoA enters beta-oxidation, generating acetyl-CoA, NADH, and FADH2 for ATP production
Beta-Oxidation: Turning Fat Into Energy
Once long-chain acyl-CoA arrives in the mitochondrial matrix courtesy of the carnitine shuttle, it enters the beta-oxidation pathway. This is a repeating cycle of four enzymatic reactions that sequentially cleave two-carbon units (as acetyl-CoA) from the fatty acid chain. Each cycle produces one FADH2, one NADH, and one acetyl-CoA.
The four steps of each beta-oxidation cycle are: oxidation by acyl-CoA dehydrogenase (producing FADH2), hydration by enoyl-CoA hydratase, a second oxidation by 3-hydroxyacyl-CoA dehydrogenase (producing NADH), and thiolysis by beta-ketothiolase (releasing acetyl-CoA and a shortened acyl-CoA). The acetyl-CoA feeds into the citric acid cycle, while NADH and FADH2 donate electrons to the electron transport chain for ATP synthesis.
Consider a palmitate molecule (C16:0), one of the most abundant fatty acids in the body. Complete beta-oxidation yields 8 acetyl-CoA molecules, 7 FADH2, and 7 NADH. When these products are fully processed through the citric acid cycle and oxidative phosphorylation, a single palmitate molecule generates approximately 106 ATP, compared to about 30-32 ATP from one glucose molecule. Fat is simply a far more energy-dense fuel, and L-carnitine is the key that unlocks access to it.
Beyond Fatty Acid Transport: Additional Roles of L-Carnitine
While fatty acid transport is carnitine's headline function, the compound plays several other metabolic roles that are clinically relevant.
Acetyl group buffering. L-carnitine can accept acetyl groups from acetyl-CoA via the enzyme carnitine acetyltransferase (CrAT), forming acetylcarnitine. This reaction serves as a metabolic buffer that prevents the accumulation of acetyl-CoA, which would inhibit pyruvate dehydrogenase and impair glucose oxidation. By "mopping up" excess acetyl-CoA, carnitine helps maintain metabolic flexibility, the ability to switch between fat and carbohydrate fuels as needed.
CoA homeostasis. By accepting acyl groups from acyl-CoA and releasing free CoA, carnitine helps maintain adequate pools of free CoA (coenzyme A), which is needed for numerous metabolic reactions beyond fatty acid oxidation, including the citric acid cycle and amino acid catabolism.
Branched-chain amino acid metabolism. Carnitine is involved in the metabolism of branched-chain amino acids (leucine, isoleucine, valine), helping to clear their metabolic intermediates from muscle tissue during intense exercise.
Peroxisomal fatty acid processing. Very-long-chain fatty acids (C22 and longer) undergo initial shortening in peroxisomes. Carnitine participates in transferring the partially oxidized products from peroxisomes to mitochondria for completion of beta-oxidation.
Antioxidant and membrane-stabilizing effects. L-carnitine and its acetylated form, acetyl-L-carnitine, have demonstrated antioxidant properties in vitro and in animal models, scavenging reactive oxygen species (ROS) and supporting mitochondrial membrane integrity. These effects are particularly relevant for cardiac and neurological tissues that have high metabolic rates and are vulnerable to oxidative damage.
Understanding these diverse metabolic roles helps explain why carnitine deficiency, whether primary (genetic) or secondary (due to kidney disease, medications, or malnutrition), can manifest as such a wide range of symptoms including muscle weakness, cardiomyopathy, hypoglycemia, and fatigue. It also provides the rationale for therapeutic carnitine supplementation in specific clinical scenarios, and why ensuring adequate delivery through injectable formulations may offer advantages over oral supplementation for patients with significant metabolic demands.
L-Carnitine Biosynthesis: A Multi-Organ Pathway
The endogenous synthesis of L-carnitine is a multi-step process that spans multiple organs and requires the coordinated activity of several enzymes and cofactors. Understanding this pathway helps explain why certain individuals may benefit from supplementation even when they appear to have "normal" diets.
The process begins with the methylation of lysine residues in proteins by specific methyltransferases that use S-adenosylmethionine (SAMe) as the methyl donor. This reaction occurs on lysine residues within specific proteins, not on free lysine. When these proteins are degraded during normal protein turnover, trimethyllysine (TML) is released. TML then undergoes four sequential enzymatic reactions to produce L-carnitine:
- TML hydroxylation: Trimethyllysine hydroxylase (TMLH) converts TML to 3-hydroxy-trimethyllysine (HTML). This reaction requires iron (Fe2+) and alpha-ketoglutarate as cofactors, with ascorbic acid (vitamin C) needed to maintain the iron in its reduced, active state.
- Aldol cleavage: HTML aldolase cleaves HTML to 4-trimethylaminobutyraldehyde (TMABA) and glycine. This is a pyridoxal phosphate (vitamin B6)-dependent reaction.
- Oxidation: TMABA dehydrogenase oxidizes TMABA to gamma-butyrobetaine (GBB) using NAD+ as a cofactor.
- Final hydroxylation: Gamma-butyrobetaine hydroxylase (GBBH) converts GBB to L-carnitine. This reaction also requires iron, alpha-ketoglutarate, and ascorbic acid, and is the final step in carnitine biosynthesis.
The first three steps occur in multiple tissues, including skeletal muscle, liver, kidney, and brain. However, GBBH, the enzyme that catalyzes the final step, is primarily expressed in the liver and kidneys (and in small amounts in the brain). This means that most tissues produce GBB but must export it to the liver or kidneys for the final conversion to carnitine. The finished carnitine is then redistributed to other tissues through the bloodstream.
This multi-organ dependency has practical implications. Patients with liver disease, kidney disease, or deficiencies in any of the required cofactors (vitamin C, iron, B6, niacin) may have impaired carnitine synthesis. Additionally, the fact that SAMe is needed for the initial methylation step connects carnitine biosynthesis to the broader one-carbon metabolism and methylation cycle. This is the same metabolic network that methionine, choline, and B12 (the components of lipotropic injections) all feed into, creating a coherent biochemical rationale for combining these compounds.
Carnitine Forms and Derivatives
L-carnitine exists in several forms and derivatives, each with slightly different properties and clinical applications. Understanding the differences helps clinicians select the appropriate form for a given patient.
L-Carnitine (free carnitine). This is the basic, unmodified form of carnitine. It's the form used in most injectable formulations and the form directly involved in the mitochondrial carnitine shuttle. When we discuss "injectable L-carnitine," this is typically what's being administered. It's the workhorse form for systemic fat metabolism support.
Acetyl-L-Carnitine (ALCAR). This is L-carnitine with an acetyl group attached. ALCAR crosses the blood-brain barrier more readily than free L-carnitine, making it the preferred form for neurological applications. It can donate its acetyl group for acetylcholine synthesis in the brain, supporting cognitive function. ALCAR is commonly used in oral supplement form for cognitive support and peripheral neuropathy, and it has been studied for depression, age-related cognitive decline, and diabetic neuropathy.
Propionyl-L-Carnitine (PLC). This esterified form has shown particular benefits for cardiovascular applications, especially peripheral arterial disease. The propionyl group enters the citric acid cycle as succinyl-CoA, potentially providing additional energy substrate to ischemic tissues. PLC has the strongest evidence base for improving walking distance in intermittent claudication.
L-Carnitine L-Tartrate (LCLT). This is a salt form of L-carnitine that's commonly used in oral supplements and research studies, particularly in exercise performance trials. The tartrate salt improves stability and absorption characteristics compared to pure L-carnitine base. Most oral supplementation research has used this form.
Glycine Propionyl-L-Carnitine (GPLC). This is a combination of glycine and propionyl-L-carnitine. It has been studied for exercise performance and nitric oxide production. Some research suggests it may support blood flow and oxygen delivery during exercise.
| Form | Primary Application | Blood-Brain Barrier | Typical Route |
|---|---|---|---|
| L-Carnitine (free) | Systemic fat metabolism, general supplementation | Limited | Oral, IM, IV, SC |
| Acetyl-L-Carnitine (ALCAR) | Cognitive support, neuropathy, depression | Good | Oral, occasionally IV |
| Propionyl-L-Carnitine (PLC) | Peripheral arterial disease, cardiac | Limited | Oral |
| L-Carnitine L-Tartrate (LCLT) | Exercise performance, recovery | Limited | Oral |
| Glycine Propionyl-L-Carnitine | Blood flow, exercise performance | Limited | Oral |
Carnitine Deficiency: Primary and Secondary Forms
Understanding carnitine deficiency states provides clinical context for when supplementation transitions from "optimization" to "medical necessity."
Primary carnitine deficiency (PCD) is an autosomal recessive genetic disorder caused by mutations in the SLC22A5 gene encoding the OCTN2 carnitine transporter. PCD is rare (estimated 1 in 40,000-120,000 births) but potentially life-threatening. Patients with PCD have severely impaired carnitine absorption from the gut and renal reabsorption, leading to profoundly low plasma and tissue carnitine levels. Clinical manifestations include cardiomyopathy, skeletal myopathy, hypoglycemia, and hepatic dysfunction. PCD is a clear indication for lifelong carnitine supplementation, often using injectable formulations in acute settings.
Secondary carnitine deficiency is much more common and can result from numerous causes: chronic kidney disease (especially in dialysis patients), certain medications (valproic acid, pivalic acid antibiotics, cisplatin), organic acidurias, fatty acid oxidation disorders, liver cirrhosis, malnutrition, vegetarian diets, aging, and high-demand states such as pregnancy, intensive exercise, or critical illness. Secondary deficiency typically presents more subtly than PCD, with symptoms like fatigue, exercise intolerance, and muscle weakness that may be attributed to other causes.
Between these clear-cut deficiency states and normal carnitine status lies a gray zone of "carnitine insufficiency," where levels are technically within the reference range but may not be optimal for the individual's metabolic demands. This is analogous to the concepts of "suboptimal" vitamin D or B12 levels, where frank deficiency criteria aren't met but supplementation may still produce clinical benefit. The increasing recognition of this insufficiency spectrum has driven much of the clinical interest in carnitine supplementation for metabolic optimization.
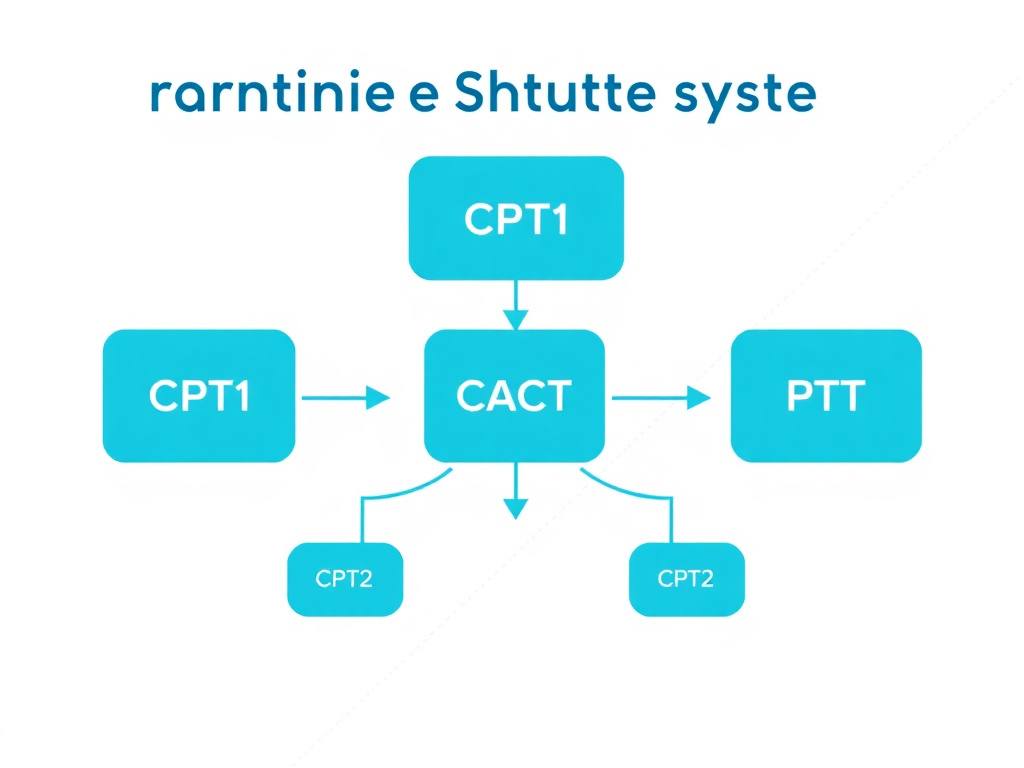
Figure 2: The carnitine shuttle system. CPT1 on the outer mitochondrial membrane conjugates carnitine with acyl groups, CACT translocates acylcarnitine across the inner membrane, and CPT2 regenerates acyl-CoA for beta-oxidation.
Injectable vs Oral Bioavailability
The Oral Absorption Problem
The bioavailability of oral L-carnitine is one of the biggest limitations of traditional supplementation. Study after study has confirmed that intestinal absorption of supplemental carnitine is surprisingly poor and follows a saturable, dose-dependent pattern.
In a landmark pharmacokinetic study published in the European Journal of Clinical Pharmacology, Harper and colleagues administered oral bolus doses of 2 g and 6 g L-carnitine to healthy volunteers. The results were striking: oral bioavailability was approximately 16% for the 2 g dose and dropped to just 5% for the 6 g dose. The data showed that mucosal absorption was already saturated at the 2 g dose, meaning that taking more orally didn't proportionally increase what reached the bloodstream. You could triple the dose and actually end up with a lower percentage absorbed.
The NIH Office of Dietary Supplements reports that typical dietary carnitine (from foods like red meat) is absorbed at a significantly higher rate, roughly 54% to 87% of a small dietary dose, compared to 5% to 18% for pharmacological supplement doses of 1 to 6 grams. This disparity exists because the body's intestinal carnitine transporter, OCTN2 (organic cation transporter, novel, type 2), becomes saturated at relatively low luminal concentrations. Once OCTN2 is maxed out, unabsorbed carnitine passes through the gut and is metabolized by intestinal bacteria.
This bacterial metabolism is more than just a bioavailability problem. Gut microbes convert unabsorbed L-carnitine to trimethylamine (TMA), which the liver then oxidizes to trimethylamine N-oxide (TMAO). As we'll discuss in the safety section, elevated TMAO has been associated with increased cardiovascular risk in observational studies, creating an ironic situation where oral supplementation of a compound intended to support cardiovascular health might potentially work against it at high doses.
Injectable Administration: Near-Complete Delivery
Intravenous and intramuscular L-carnitine administration bypasses the gastrointestinal tract entirely, achieving bioavailability approaching 100%. This isn't just a theoretical advantage. It represents a fundamental shift in how much active compound actually reaches target tissues.
The FDA-approved injectable formulation (levocarnitine injection, brand name Carnitor) delivers L-carnitine directly into systemic circulation. Pharmacokinetic studies in hemodialysis patients demonstrated that IV L-carnitine maintained significantly higher and more sustained plasma carnitine levels compared to oral administration, even when the IV dose was approximately half the oral dose. That's worth repeating: half the injectable dose outperformed double the oral dose in terms of sustained plasma levels.
For intramuscular (IM) and subcutaneous (SC) injections used in clinical weight management settings, absorption is rapid and nearly complete. Peak plasma levels are typically achieved within 1 to 3 hours after IM injection, compared to 3 to 4 hours for oral dosing, and the peak concentration is dramatically higher with injectable routes.
L-Carnitine Bioavailability by Route and Dose
Pharmacokinetic Comparison
| Parameter | Oral (2 g) | Oral (6 g) | IV/IM (500 mg - 1 g) |
|---|---|---|---|
| Bioavailability | ~16% | ~5% | ~95-100% |
| Effective dose delivered | ~320 mg | ~300 mg | 475-1,000 mg |
| Time to peak plasma | 3-4 hours | 3-4 hours | 0.5-2 hours |
| GI side effects | Moderate (nausea, diarrhea) | Common (dose-limiting) | Minimal |
| TMAO production via gut | Yes (dose-dependent) | High | Minimal (bypasses gut) |
| Absorption saturation | Approaching maximum | Fully saturated | Not applicable |
| Half-life (plasma) | ~17 hours (similar regardless of route once in circulation) | ||
Distribution and Elimination
Regardless of the route of administration, once L-carnitine reaches the bloodstream, its distribution and elimination follow similar patterns. Carnitine distributes primarily to skeletal muscle and cardiac tissue. Plasma half-life is approximately 17 hours, though tissue turnover is much slower. The kidneys are the primary route of elimination, with renal tubular reabsorption recovering more than 95% of filtered carnitine under normal circumstances. This efficient renal conservation is why healthy individuals with adequate dietary intake rarely develop carnitine deficiency.
One practical consideration: because skeletal muscle serves as the primary carnitine reservoir and uptake into muscle is relatively slow, achieving meaningful increases in intramuscular carnitine stores requires sustained supplementation over weeks to months, regardless of route. A single injection produces a transient spike in plasma carnitine that supports acute metabolic demand but won't dramatically shift tissue stores overnight. This is why consistent, protocol-based dosing is emphasized in clinical practice.
Clinical Implications of the Bioavailability Gap
The bioavailability advantage of injectable L-carnitine has several clinical implications that go beyond simple math.
Dose precision. With oral supplementation, there's substantial inter-individual variability in absorption. Factors like gut transit time, OCTN2 expression, microbiome composition, and concurrent food intake all influence how much carnitine makes it into the bloodstream. Injectable administration eliminates this variability, allowing clinicians to predict delivered doses with much greater accuracy.
Reduced GI side effects. High-dose oral L-carnitine commonly causes nausea, abdominal cramping, and diarrhea. These side effects limit the practical oral dose ceiling for many patients. Injectable routes bypass the gut entirely, eliminating GI-related tolerability issues.
TMAO avoidance. By skipping intestinal transit, injectable L-carnitine avoids exposure to gut bacteria that convert carnitine to TMA. This is a meaningful advantage for patients concerned about cardiovascular risk, though the clinical significance of supplement-related TMAO elevation remains debated.
Cost-effectiveness at the tissue level. Although injectable L-carnitine is more expensive per dose than oral supplements, the dramatically higher bioavailability means that a smaller injectable dose delivers more active compound to tissues. When comparing cost per milligram of carnitine actually absorbed, the gap narrows considerably.
For researchers and clinicians interested in fat metabolism optimization, the injectable route aligns well with other parenteral therapies. Many patients who receive injectable L-carnitine are already using peptide protocols such as CJC-1295/Ipamorelin for growth hormone support or BPC-157 for tissue repair, making the addition of another subcutaneous injection a minimal incremental burden.
The OCTN2 Transporter: Why Oral Absorption Has a Ceiling
To fully appreciate why injectable L-carnitine provides such a dramatic pharmacokinetic advantage, it helps to understand the molecular biology of intestinal carnitine absorption. The organic cation/carnitine transporter OCTN2, encoded by the SLC22A5 gene, is the primary transporter responsible for absorbing carnitine from the intestinal lumen into enterocytes.
OCTN2 is a sodium-dependent, high-affinity carnitine transporter with a Km (half-maximal saturation concentration) of approximately 4-5 micromolar. This means the transporter works efficiently at the low carnitine concentrations present in the intestinal lumen after a normal meal (which might provide 50-200 mg of carnitine). But when you take a 2-6 gram supplement, the luminal concentration vastly exceeds the transporter's capacity, and most of the carnitine simply passes through the gut unabsorbed.
Think of OCTN2 as a revolving door with a fixed throughput capacity. At normal dietary levels, the door handles the traffic fine. But flood the entrance with 10-50 times the usual crowd, and the bottleneck becomes obvious. There's no way to increase OCTN2 expression acutely, no secondary transport system that kicks in at high concentrations, and no passive diffusion pathway for this polar, charged molecule. The transporter is the sole gateway, and its capacity is finite.
This saturation kinetics explains the paradoxical dose-bioavailability relationship seen in pharmacokinetic studies: doubling the oral dose doesn't double absorption. In fact, tripling the dose from 2g to 6g actually reduced the percentage absorbed from 16% to 5% in the Harper study. The absolute amount absorbed may increase slightly with higher doses, but the efficiency drops precipitously.
OCTN2 is also the transporter responsible for carnitine reabsorption in the kidneys, where it recovers over 95% of filtered carnitine. This renal conservation mechanism is why healthy people rarely develop carnitine deficiency despite the poor oral absorption of supplements; the body simply holds on to the carnitine it already has. But for patients seeking to elevate carnitine levels above their baseline for therapeutic purposes, the oral route is genuinely limited by OCTN2 biology.
Differential Effects of Oral vs. Injectable on the Gut Microbiome
An underappreciated aspect of the oral versus injectable comparison involves differential effects on the gut microbiome. When large doses of L-carnitine pass through the gut unabsorbed (which is the fate of 82-95% of an oral supplement dose), this carnitine becomes a substrate for intestinal bacteria.
Specific bacterial species, particularly members of the Firmicutes phylum and the genus Acinetobacter, can cleave the nitrogen-carbon bond in carnitine to produce trimethylamine (TMA). The extent of this bacterial conversion depends on the individual's gut microbiome composition, which is itself influenced by long-term dietary habits. Studies have shown that regular red meat consumers harbor significantly more carnitine-metabolizing bacteria than vegetarians, leading to greater TMA (and subsequently TMAO) production from the same carnitine dose.
This creates an interesting dynamic: chronic oral L-carnitine supplementation may actually reshape the gut microbiome over time, enriching for carnitine-metabolizing species and potentially increasing TMAO production capacity. A study by Koeth and colleagues published in the Journal of Clinical Investigation confirmed this, showing that L-carnitine supplementation in omnivores induced a gut microbial pathway that enhanced TMA/TMAO production over time.
Injectable L-carnitine avoids this entire microbial interaction. By delivering carnitine directly to the bloodstream, it doesn't provide substrate for intestinal bacteria, doesn't reshape the gut microbiome toward greater TMA production capacity, and doesn't generate the burst of TMAO that follows a high oral dose. While some circulating carnitine does enter the gut through biliary secretion and intestinal mucosal shedding, the quantities are much smaller than those from unabsorbed oral supplements.
This microbiome dimension adds another layer to the oral-versus-injectable comparison that goes beyond simple bioavailability numbers. For patients who are particularly concerned about cardiovascular risk or who already have elevated TMAO levels, the injectable route may offer meaningful safety advantages over oral supplementation.
Practical Considerations: Real-World Absorption Scenarios
To make the bioavailability comparison more tangible, consider these real-world scenarios:
Scenario 1: A patient takes 2 g oral L-carnitine daily. With approximately 16% bioavailability, about 320 mg reaches the bloodstream. The remaining 1,680 mg transits the gut, feeds bacteria that produce TMA, and is eventually excreted. The patient may experience nausea and loose stools from the unabsorbed carnitine, which draws water into the bowel by osmotic effect. If the patient skipped breakfast and took it on an empty stomach, absorption might be even lower. If they took it with a large carbohydrate-rich meal, insulin secretion might improve subsequent muscle uptake of the absorbed fraction, but intestinal absorption itself remains capped by OCTN2.
Scenario 2: The same patient receives a 500 mg intramuscular L-carnitine injection. Essentially all 500 mg enters the bloodstream over the next 1-3 hours. No GI side effects. No bacterial TMA production. No dependence on OCTN2 capacity or meal timing. The delivered dose is actually higher (500 mg vs. 320 mg) despite the supplement dose being four times smaller. And the cost of the "wasted" unabsorbed oral carnitine, both in dollars and in unwanted TMAO production, is eliminated entirely.
Scenario 3: A vegetarian athlete wants to optimize muscle carnitine for a training block. Their baseline dietary carnitine intake is very low (perhaps 10-25 mg/day versus 100-300 mg/day for an omnivore). Oral supplementation at 2 g/day would provide about 320 mg absorbed, but their gut microbiome, adapted to a plant-based diet, would produce less TMAO than an omnivore's (a relative advantage of vegetarian status for oral supplementation). However, the bioavailability ceiling still applies. A 500 mg injection twice weekly provides 1,000 mg per week with certainty, versus approximately 2,240 mg weekly from daily oral dosing (but with only 320 mg daily actually absorbed, totaling the same 2,240 mg nominal but only ~2,240 mg absorbed over the week from 14g oral). Wait - let's recalculate: 2g oral daily x 7 days = 14g oral/week, with 16% bioavailability = ~2,240 mg absorbed. Two 500 mg injections = 1,000 mg absorbed. So the oral route, despite its poor efficiency, can deliver more total carnitine if you're willing to take 2 g every single day and tolerate the GI effects. The injectable route delivers less total but with perfect predictability and no side effects. The practical choice depends on patient preference, compliance likelihood, and GI tolerance.
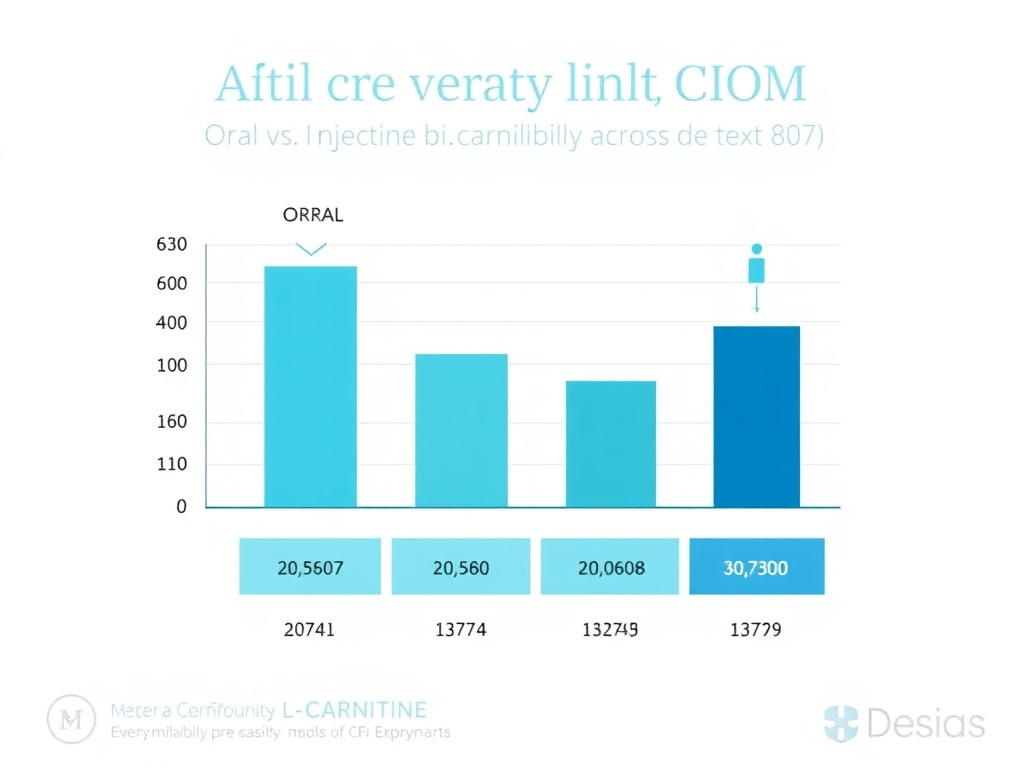
Figure 3: Comparison of L-carnitine bioavailability by administration route. Injectable formulations achieve near-complete systemic delivery, while oral doses face intestinal absorption saturation.
MIC Injection Components
MIC injections - named for their three core ingredients: methionine, inositol, and choline - are the most widely used lipotropic injection formulation in clinical weight management. Understanding each component's biochemistry helps clarify what these injections can and cannot do.
Methionine
Methionine is an essential sulfur-containing amino acid that cannot be synthesized by the body and must be obtained from dietary protein sources. In the context of lipotropic function, methionine serves several metabolic roles relevant to fat metabolism.
Transmethylation and fat export. Methionine is the metabolic precursor to S-adenosylmethionine (SAMe), the body's principal methyl donor. Adequate methylation capacity is required for the synthesis of phosphatidylcholine, the primary phospholipid component of VLDL particles that the liver uses to export triglycerides. When methyl donor status is compromised, hepatic fat export can be impaired, contributing to fatty liver. This connection between methionine deficiency and hepatic steatosis has been well-established in both animal models and human studies.
Glutathione synthesis. Methionine is also a precursor to cysteine via the transsulfuration pathway, and cysteine is the rate-limiting amino acid for glutathione synthesis. Glutathione is the body's most abundant intracellular antioxidant and plays a central role in hepatic detoxification. Supporting glutathione status through methionine supplementation may protect the liver during periods of increased metabolic stress, such as rapid weight loss.
Creatine synthesis. Methionine contributes methyl groups to the synthesis of creatine, which is relevant for exercise performance and lean body mass maintenance during weight loss programs.
In MIC injection formulations, methionine is typically dosed at 25 mg per mL. The injectable route ensures complete bioavailability of this amino acid, though methionine deficiency is uncommon in individuals consuming adequate protein.
Inositol
Inositol (specifically myo-inositol, the most abundant biological form) is a carbocyclic sugar that was once classified as vitamin B8, though this designation has been abandoned since the body can synthesize it from glucose. Despite not being a true vitamin, inositol plays several roles in cellular signaling and lipid metabolism.
Insulin signaling. Inositol derivatives, particularly D-chiro-inositol and myo-inositol, function as second messengers in the insulin signaling pathway. Research in women with polycystic ovary syndrome (PCOS) has demonstrated that myo-inositol supplementation can improve insulin sensitivity, reduce circulating androgen levels, and support metabolic function. A systematic review of randomized controlled trials found that inositol improved insulin resistance markers and lipid profiles in PCOS patients.
Hepatic lipid metabolism. Inositol participates in the formation of phosphatidylinositol, a membrane phospholipid involved in lipid transport and liver function. Animal studies dating back to the 1940s demonstrated that inositol deficiency leads to hepatic fat accumulation, establishing its classification as a lipotropic factor. However, translating these findings to human supplementation in non-deficient individuals is more complex.
Serotonin and neurotransmitter function. Inositol is involved in serotonin receptor signaling in the brain. High-dose oral inositol (12-18 g/day) has been studied for anxiety and depression, conditions that can contribute to emotional eating and weight gain. The mood-supporting potential of inositol may indirectly support weight management efforts in some patients.
Typical MIC injection formulations include 50 mg of inositol per mL. The research supporting inositol's lipotropic effects is most compelling in the context of insulin resistance and PCOS, conditions where semaglutide and tirzepatide are also being studied extensively.
Choline
Choline is an essential nutrient recognized by the Institute of Medicine in 1998. Although it can be synthesized endogenously in small amounts via the phosphatidylethanolamine N-methyltransferase (PEMT) pathway, this production is insufficient to meet physiological needs, making dietary intake essential. Choline deficiency is remarkably common - the National Health and Nutrition Examination Survey (NHANES) data suggests that approximately 90% of Americans don't consume the adequate intake level.
Phospholipid synthesis and hepatic fat export. Choline is the direct precursor to phosphatidylcholine, which comprises roughly 95% of the phospholipid content in VLDL particles. Without adequate phosphatidylcholine, the liver cannot assemble and secrete VLDL, leading to triglyceride accumulation and non-alcoholic fatty liver disease (NAFLD). This is the most well-characterized lipotropic mechanism of choline, and it has been confirmed in controlled human feeding studies where choline deprivation rapidly induces hepatic steatosis and organ dysfunction.
Acetylcholine synthesis. Choline is the precursor to the neurotransmitter acetylcholine, which is involved in muscle contraction, memory, and autonomic nervous system function. Adequate choline status may support cognitive function and physical performance during caloric restriction.
Betaine and methylation. Choline can be oxidized to betaine, which serves as an alternative methyl donor in the methionine cycle. This links choline metabolism directly to methionine status and overall methylation capacity, reinforcing the complementary rationale behind combining all three MIC components.
MIC formulations typically contain choline chloride at 50 mg per mL. Given the widespread prevalence of suboptimal choline intake in the general population, this may be the MIC component most likely to provide genuine biochemical benefit to a broad range of patients.
Common Lipotropic Injection Variations
Compounding pharmacies offer numerous variations on the basic MIC theme, often marketed under different trade names. Understanding the differences helps clinicians and patients make informed choices.
| Formulation | Components | Typical Dose per mL | Notes |
|---|---|---|---|
| MIC (Basic) | Methionine, Inositol, Choline | 25/50/50 mg | Original lipotropic formulation |
| Lipo-B (MIC + B12) | MIC + Cyanocobalamin | 25/50/50 mg + 1 mg B12 | Most popular variation; B12 for energy |
| Lipo-C | MIC + B12 + L-Carnitine | Varies; typically + 125 mg carnitine | Enhanced fat metabolism formulation |
| Lipo Plus / Super Lipo | MIC + B12 + B-Complex | Varies by pharmacy | Added B1, B2, B5, B6 for energy support |
| MICC | MIC + Cyanocobalamin | 25/50/50/1 mg | Alternative naming for Lipo-B |
| Lipotropic + Amino Blend | MIC + B12 + L-Carnitine + Amino acids | Varies widely | Premium formulations from specialty compounders |
Vitamin B12 in Lipotropic Injections
Nearly all commercial lipotropic injection formulations include vitamin B12 (cyanocobalamin or methylcobalamin), and for good reason. B12 deficiency affects an estimated 6% to 20% of older adults and is more common in individuals taking metformin, proton pump inhibitors, or following plant-based diets.
B12 serves as a cofactor for methionine synthase, the enzyme that converts homocysteine back to methionine in the methylation cycle. It also supports the function of methylmalonyl-CoA mutase, an enzyme in the catabolic pathway of certain amino acids and odd-chain fatty acids. Adequate B12 is necessary for normal red blood cell formation, neurological function, and DNA synthesis.
In the context of lipotropic injections, B12 serves a dual purpose. First, it supports the methylation cycle that methionine and choline also feed into, creating a coherent metabolic support package. Second, it addresses a commonly identified deficiency that, when corrected, often produces a noticeable improvement in energy levels. This energy boost, while not a direct fat-burning effect, can improve adherence to exercise programs and active lifestyles that support weight loss.
The injectable route for B12 is particularly advantageous in patients with pernicious anemia, atrophic gastritis, or other conditions that impair intrinsic factor-dependent B12 absorption. Even in individuals without diagnosed malabsorption, injectable B12 achieves higher and more predictable serum levels than oral supplementation.
Clinical Perspective
While the individual ingredients in MIC injections have established biochemical roles in fat metabolism and liver function, keep in mind that no large-scale, randomized, placebo-controlled clinical trial has tested the combined MIC injectable formulation specifically for weight loss. The lipotropic injection concept is built on sound biochemical rationale and decades of clinical use in weight management settings, but the evidence base consists primarily of mechanistic data for individual components rather than outcome studies for the combined product. Patients should understand that MIC injections are best viewed as metabolic support tools within comprehensive weight management programs, not standalone fat-loss treatments.
The Methionine-SAMe-Methylation Axis in Detail
Methionine's role in lipotropic injections becomes clearer when you trace its metabolic fate through the one-carbon metabolism pathway. This pathway is central to dozens of cellular processes, and understanding it explains why methionine, choline, and B12 work together so cohesively.
When methionine enters the methionine cycle, it's first activated by methionine adenosyltransferase (MAT) to form S-adenosylmethionine (SAMe). This is the most important methylation step in human biochemistry. SAMe is the universal methyl donor for over 200 methyltransferase reactions in the body, including DNA methylation (epigenetic regulation), phosphatidylcholine synthesis (PEMT pathway, critical for VLDL assembly), creatine synthesis (the single largest consumer of methyl groups in the body), neurotransmitter synthesis (norepinephrine to epinephrine, and others), and carnitine biosynthesis (the initial methylation of lysine residues).
After donating its methyl group, SAMe becomes S-adenosylhomocysteine (SAH), which is then hydrolyzed to homocysteine. Homocysteine sits at a critical metabolic branch point. It can be remethylated back to methionine (requiring either vitamin B12 and folate via methionine synthase, or betaine via betaine-homocysteine methyltransferase), or it can be irreversibly committed to the transsulfuration pathway (requiring vitamin B6 via cystathionine beta-synthase), which ultimately produces cysteine and glutathione.
This interconnection explains the rationale for combining methionine with B12 in lipotropic injections. B12 is essential for the remethylation of homocysteine to methionine, keeping the methionine cycle turning. Without adequate B12, homocysteine accumulates (raising cardiovascular risk) and the methionine cycle stalls (reducing methylation capacity, including the methylation needed for phosphatidylcholine synthesis and fat export).
Choline enters this same metabolic network through betaine, providing an alternative remethylation route. This redundancy is protective: if folate or B12 status is suboptimal, the choline/betaine pathway can partially compensate for the impaired methionine synthase pathway. Conversely, when choline is depleted, the demand on the folate/B12-dependent pathway increases. The MIC formulation provides support at multiple points in this interconnected network, making it more strong than supplementing any single component alone.
Non-Alcoholic Fatty Liver Disease (NAFLD) and Lipotropic Factors
The relevance of lipotropic injections to liver health extends beyond theoretical biochemistry into a genuine clinical epidemic. Non-alcoholic fatty liver disease (NAFLD), now being reclassified as metabolic dysfunction-associated steatotic liver disease (MASLD), affects an estimated 25-30% of the global adult population. In the United States, prevalence may be even higher, particularly among populations with obesity, insulin resistance, and type 2 diabetes.
NAFLD represents a spectrum from simple hepatic steatosis (fat accumulation in the liver without inflammation) to non-alcoholic steatohepatitis (NASH, featuring inflammation and hepatocyte injury), which can progress to fibrosis, cirrhosis, and hepatocellular carcinoma. The disease burden is enormous, and effective treatments beyond weight loss and lifestyle modification remain limited.
Each MIC component has documented connections to hepatic fat handling. Choline deficiency directly causes hepatic steatosis in controlled human feeding studies - when researchers restrict choline intake in volunteers, liver fat accumulation occurs within weeks and reverses when choline is restored. Methionine deficiency impairs VLDL assembly and export through reduced phosphatidylcholine availability. Inositol deficiency promotes hepatic fat accumulation in animal models.
These observations form the strongest biochemical rationale for lipotropic injections. By ensuring adequate supply of all three lipotropic factors, MIC injections may support the liver's ability to process, package, and export triglycerides, particularly during the increased hepatic fat mobilization that occurs during weight loss programs. When patients lose weight rapidly (as can occur with GLP-1 agonists, aggressive caloric restriction, or bariatric surgery), the liver must handle a surge of fatty acids released from shrinking adipose depots. Lipotropic support during this period may help the liver manage this increased workload without developing transient steatosis.
It's important to acknowledge that while this rationale is sound, it hasn't been validated in large clinical trials specifically testing MIC injections for NAFLD prevention or treatment. The evidence base is largely inferential, drawn from deficiency studies and mechanistic research. Patients with diagnosed NAFLD should be under medical supervision, and MIC injections should not be viewed as a substitute for evidence-based NAFLD management.
The Choline Crisis: Why This MIC Component May Matter Most
Of the three MIC components, choline may be the one most likely to provide genuine clinical benefit in a broad patient population, simply because choline deficiency is so widespread. NHANES data consistently shows that the vast majority of Americans don't meet the adequate intake (AI) for choline (550 mg/day for men, 425 mg/day for women).
Several factors contribute to this widespread inadequacy. Choline is concentrated in a relatively narrow range of foods: eggs (particularly egg yolks), liver, beef, chicken, fish, and soybeans. People who limit egg consumption (due to cholesterol concerns), avoid organ meats, follow plant-based diets, or simply eat a standard processed-food diet are likely to have suboptimal choline intake.
Genetic factors further complicate the picture. The PEMT gene, which encodes the enzyme responsible for endogenous phosphatidylcholine synthesis, has common polymorphisms that increase dietary choline requirements. Premenopausal women have some protection from choline deficiency because estrogen upregulates PEMT expression, but postmenopausal women, men, and women with certain PEMT variants are more vulnerable.
Pregnant and lactating women have substantially increased choline requirements because choline is needed for fetal brain development and is concentrated in breast milk. Many prenatal vitamins contain little or no choline, leaving pregnant women at particular risk of deficiency during a period when the consequences for fetal neural development are most significant.
While the 50 mg of choline in a standard MIC injection is small relative to the daily AI, it provides a guaranteed, 100% bioavailable dose that goes directly to the liver, where it's most needed for phospholipid synthesis and VLDL assembly. For patients who are chronically under-consuming dietary choline, even this modest injectable supplement may help support hepatic function.
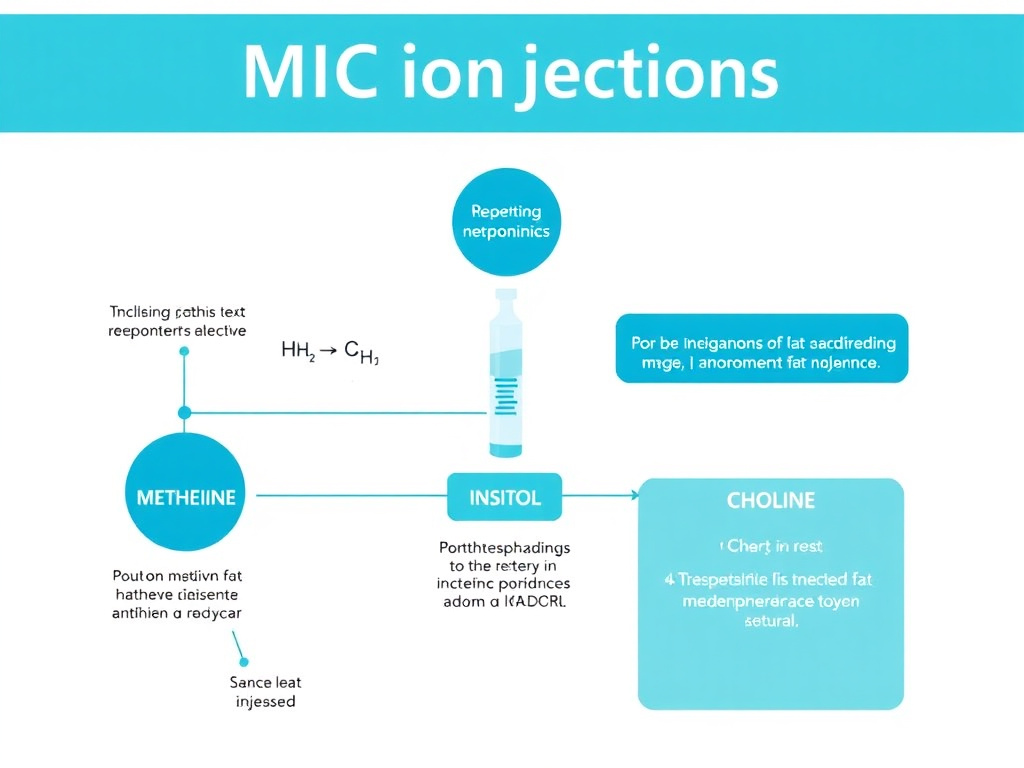
Figure 4: Metabolic pathways of MIC components. Methionine feeds SAMe and glutathione synthesis; inositol supports insulin signaling and phospholipid formation; choline provides phosphatidylcholine for VLDL assembly and hepatic fat export.
Fat Metabolism Enhancement
L-Carnitine and Fat Oxidation: What the Research Shows
The theoretical case for L-carnitine as a fat-burning agent is straightforward: more carnitine should mean more efficient fatty acid transport into mitochondria, leading to increased fat oxidation. The reality, as is often the case in biology, is more nuanced than the theory suggests.
A 2020 systematic review published in the Journal of the International Society of Sports Nutrition by Damian Pons and colleagues examined both the beneficial and adverse aspects of L-carnitine supplementation. The review found that chronic L-carnitine supplementation could result in a greater fatty acid oxidation rate during exercise, particularly when combined with carbohydrate co-ingestion. However, the effects on body composition and direct fat loss were less consistent.
The inconsistency appears related to several factors. First, most studies used oral supplementation, which faces the bioavailability limitations discussed earlier. Second, in healthy individuals with adequate carnitine status and moderate exercise levels, carnitine may not be rate-limiting for fat oxidation; the bottleneck may lie elsewhere in the metabolic chain. Third, increased fat oxidation during exercise doesn't automatically translate to net fat loss if total caloric balance isn't addressed.
The Muscle Carnitine Loading Problem
A key finding from the research group led by Francis Stephens at the University of Nottingham clarified an important barrier to oral L-carnitine's effectiveness. Their work demonstrated that simply raising plasma carnitine through oral supplementation does not reliably increase muscle carnitine content. The OCTN2 transporter that moves carnitine from blood into muscle is, under resting conditions, already operating near capacity.
However, the Stephens group found that insulin dramatically stimulates muscle carnitine uptake. When L-carnitine was taken with a substantial carbohydrate load (sufficient to raise insulin levels to approximately 50 mU/L or higher), muscle carnitine stores did increase over a 24-week protocol. After 24 weeks of carnitine plus carbohydrate supplementation, participants showed a 21% increase in muscle carnitine content, reduced muscle glycogen utilization during low-intensity exercise, and increased muscle glycogen content, reflecting improved metabolic flexibility.
This finding has important implications. It suggests that carnitine's fat-oxidation benefits may be most accessible when combined with conditions that promote insulin release, and that injectable L-carnitine, by achieving higher sustained plasma levels, might facilitate greater muscle uptake even without the need for carbohydrate co-ingestion, though this specific comparison hasn't been studied directly.
L-Carnitine and Specific Fat Loss Outcomes
Several clinical trials have examined body composition changes with L-carnitine supplementation:
A meta-analysis by Talenezhad and colleagues (2020) pooled data from 37 randomized controlled trials and found that L-carnitine supplementation produced statistically significant reductions in body weight (weighted mean difference: -1.21 kg), BMI (-0.47 kg/m2), and fat mass (-2.08 kg). These effects were more pronounced in studies lasting longer than 12 weeks and in overweight or obese participants.
Conversely, a study by Villani and colleagues found that L-carnitine supplementation combined with aerobic training did not promote additional weight loss in moderately obese women beyond what exercise alone achieved, suggesting that carnitine's contribution may be marginal in some contexts.
The balance of evidence suggests that L-carnitine supplementation produces modest but measurable fat loss benefits, particularly in overweight individuals, during longer treatment durations, and when combined with regular exercise and caloric management. It's not a standalone fat-loss solution, but it can be a useful component of a comprehensive approach.
Combining L-Carnitine with Fat-Loss Peptides
Where injectable L-carnitine may shine brightest is as part of a multi-modal metabolic optimization protocol. Several peptide compounds target fat loss through different mechanisms, and adding L-carnitine can theoretically amplify their effects by ensuring the downstream fat oxidation machinery runs at full capacity.
AOD-9604. This modified fragment of human growth hormone (hGH fragment 176-191) stimulates lipolysis, releasing fatty acids from adipose tissue into circulation. Once freed, those fatty acids need to be transported into mitochondria for oxidation. AOD-9604 addresses the "release" step, while L-carnitine supports the "burn" step. Combining them creates a more complete fat mobilization and oxidation pathway. Learn more in our AOD-9604 research report.
GLP-1 receptor agonists. Compounds like semaglutide and tirzepatide reduce appetite and improve insulin sensitivity, creating a caloric deficit that shifts the body toward fat utilization. L-carnitine supports the increased fatty acid oxidation demand that occurs during weight loss. See our GLP-1 overview for a deeper look at these compounds.
5-Amino-1MQ. This small molecule inhibits NNMT (nicotinamide N-methyltransferase), an enzyme that, when overactive, promotes fat storage and reduces cellular energy expenditure. 5-Amino-1MQ shifts metabolic programming away from fat storage, and L-carnitine ensures efficient processing of the mobilized fatty acids. Our 5-Amino-1MQ research report covers this mechanism in detail.
MOTS-c. This mitochondrial-derived peptide activates AMPK and enhances mitochondrial function. MOTS-c essentially upgrades the mitochondrial machinery, while L-carnitine ensures a steady supply of fatty acid fuel to those upgraded mitochondria.
Tesofensine. This triple monoamine reuptake inhibitor reduces appetite while also increasing energy expenditure. During tesofensine-induced caloric deficit, L-carnitine can support the metabolic shift toward increased fat oxidation. More details in the tesofensine weight loss report.
For patients exploring these combinations, the free assessment at FormBlends can help determine which protocols may be appropriate, and the dosing calculator can assist with protocol planning.
Research Summary: L-Carnitine and Fat Metabolism
- Meta-analyses show modest but significant reductions in body weight (-1.21 kg) and fat mass (-2.08 kg) with L-carnitine supplementation
- Muscle carnitine uptake requires elevated insulin, explaining why carbohydrate co-ingestion improves outcomes with oral dosing
- Injectable L-carnitine achieves higher sustained plasma levels that may facilitate better tissue loading
- Effects are most pronounced in overweight/obese individuals with longer supplementation durations (12+ weeks)
- L-carnitine is best understood as a metabolic support tool rather than a primary fat-loss agent
- Combining with lipolytic peptides (AOD-9604, GLP-1 agonists) creates a more complete fat mobilization and oxidation pathway
Understanding Metabolic Flexibility and the Role of Carnitine
Metabolic flexibility is a concept that has gained significant attention in metabolic research over the past decade. It refers to the body's ability to efficiently switch between burning carbohydrates and burning fat as fuel depending on substrate availability, hormonal signaling, and energy demands. A metabolically flexible individual can readily ramp up fat oxidation during fasting, moderate exercise, or low-carbohydrate feeding, and can efficiently switch to carbohydrate oxidation when glucose availability is high (after a meal, during intense exercise).
Metabolic inflexibility, conversely, is a hallmark of insulin resistance, type 2 diabetes, and obesity. In metabolically inflexible individuals, the ability to increase fat oxidation in response to fasting or exercise is blunted, while basal fat oxidation may paradoxically be elevated (because insulin-resistant tissues don't properly suppress fat oxidation after meals). This creates a metabolic log-jam where fat oxidation isn't properly regulated, intermediary lipid metabolites accumulate, and overall energy efficiency suffers.
L-carnitine sits right at the center of metabolic flexibility because it directly controls the flow of fatty acids into mitochondria. When carnitine availability is adequate and the CPT1 regulatory system (malonyl-CoA inhibition in the fed state, malonyl-CoA relief in the fasted state) is functioning properly, the system adapts fluidly to changing metabolic demands. But when carnitine availability is rate-limiting, the fasted-state upregulation of fat oxidation is constrained, and metabolic flexibility is impaired.
This framework helps explain why L-carnitine supplementation may be more effective in certain populations than others. In lean, metabolically healthy individuals who already have adequate carnitine stores and good metabolic flexibility, additional carnitine may not provide a meaningful benefit because the system isn't carnitine-limited. But in overweight or insulin-resistant individuals, where metabolic flexibility is already compromised and where increased fatty acid flux creates greater demand for carnitine-dependent transport, supplementation may make a measurable difference.
The Stephens group's finding that insulin is required for effective muscle carnitine uptake adds another layer. Insulin resistance impairs the ability to load carnitine into muscle cells, creating a vicious cycle: the very patients who most need improved fat oxidation (those with insulin resistance) are the ones least able to benefit from oral carnitine supplementation because their insulin-mediated muscle uptake is impaired. Injectable L-carnitine, by maintaining higher sustained plasma levels, may partially overcome this barrier by providing a stronger concentration gradient driving passive carnitine diffusion into muscle, though this hypothesis hasn't been directly tested.
Carnitine and Adipose Tissue Biology
Most discussions of carnitine focus on its role in skeletal muscle and liver, but adipose tissue itself expresses the carnitine shuttle components and carries out fatty acid oxidation. In recent years, research on brown and beige adipose tissue has revealed that these metabolically active fat depots are highly dependent on mitochondrial fatty acid oxidation for their thermogenic function.
Brown adipose tissue (BAT) generates heat through uncoupled oxidative phosphorylation, with fatty acids serving as both the fuel and the activators of uncoupling protein 1 (UCP1). The carnitine shuttle is essential for delivering fatty acids to BAT mitochondria for this process. Emerging research suggests that carnitine availability may influence BAT activity and the "browning" of white adipose tissue into metabolically active beige adipose tissue.
While this research is still in relatively early stages, it opens an interesting avenue for L-carnitine's potential role in fat metabolism that goes beyond simple "fat burning." If carnitine supplementation can support BAT activity or white-to-beige fat conversion, the thermogenic effects could contribute to energy expenditure in ways that conventional fat oxidation measurements might underestimate.
Several peptides under active research also influence adipose tissue biology. Fragment 176-191 (the active lipolytic fragment of growth hormone) preferentially targets abdominal fat. MOTS-c influences mitochondrial function across multiple tissues including adipose. Understanding how L-carnitine interacts with these compounds at the adipose tissue level represents a frontier of research that could inform more targeted combination protocols.
The Lipolysis-Oxidation Disconnect
A critical concept that practitioners and patients alike need to understand is the distinction between lipolysis (the release of fatty acids from stored triglycerides in adipose tissue) and fatty acid oxidation (the actual burning of those fatty acids for energy in mitochondria). These are two separate metabolic steps, and stimulating one without supporting the other can be counterproductive.
Many popular fat-loss interventions primarily stimulate lipolysis. Caffeine, catecholamines, growth hormone, and fasting all increase the release of free fatty acids from fat cells. But if those released fatty acids aren't efficiently oxidized, they simply recirculate in the bloodstream, get re-esterified in the liver or adipose tissue, or contribute to lipotoxicity in non-adipose tissues like muscle and liver. This is part of why crash diets and extreme stimulant use don't reliably produce lasting fat loss; they create a flood of free fatty acids without proportionally increasing the capacity to burn them.
L-carnitine addresses the oxidation side of this equation. By ensuring efficient transport of fatty acids into mitochondria, it supports the downstream processing of mobilized fat. This is why the combination of a lipolytic stimulus (exercise, fasting, peptides like AOD-9604, or GLP-1-mediated caloric deficit) with carnitine-supported oxidation capacity creates a more physiologically complete approach to fat loss than either strategy alone.
The concept is straightforward: release the fat (lipolysis), transport it to the furnace (carnitine shuttle), and burn it (beta-oxidation). Any bottleneck in this chain limits the overall rate of fat utilization. L-carnitine primarily addresses the transport bottleneck, making it a logical complement to interventions that address the release and burning steps.
Figure 10: The three-step fat utilization pathway. Lipolysis releases fatty acids from adipose tissue, the carnitine shuttle transports them into mitochondria, and beta-oxidation converts them to ATP. L-carnitine primarily supports the transport step.
Exercise Performance
Mechanisms of Performance Enhancement
L-carnitine's potential to enhance exercise performance stems from several interconnected mechanisms that go beyond simple fat burning. During exercise, carnitine serves as a metabolic buffer and fuel-partitioning agent that can influence which energy substrates the body prioritizes and how efficiently they're used.
Glycogen sparing. During low to moderate-intensity exercise (below the lactate threshold), increasing muscle carnitine content promotes greater reliance on fat oxidation, thereby "sparing" finite muscle glycogen stores for high-intensity efforts. This glycogen-sparing effect could theoretically extend endurance capacity, much like the concept behind "fat-adapted" endurance training.
Pyruvate dehydrogenase buffering. During high-intensity exercise, muscle carnitine plays a different role. The enzyme pyruvate dehydrogenase (PDH) converts pyruvate to acetyl-CoA, feeding carbohydrate-derived carbon into the citric acid cycle. When exercise intensity is high and acetyl-CoA production exceeds citric acid cycle capacity, the resulting acetyl-CoA accumulation inhibits PDH and shunts excess pyruvate toward lactate. By accepting acetyl groups via carnitine acetyltransferase, carnitine buffers this acetyl-CoA excess, maintaining PDH activity and reducing lactate accumulation. Less lactate means less acidosis and potentially delayed fatigue.
Reduced muscle damage and oxidative stress. Multiple studies have shown that L-carnitine supplementation reduces biomarkers of exercise-induced muscle damage, including creatine kinase, lactate dehydrogenase, and myoglobin leakage. The antioxidant properties of carnitine may protect muscle cell membranes from oxidative damage during intense training.
Systematic Review Evidence
A 2021 systematic review in Nutrients by Gnoni and colleagues examined the effects of acute and chronic oral L-carnitine supplementation on exercise performance stratified by exercise intensity. The findings revealed an important pattern:
Chronic supplementation (weeks to months) at doses of 1-3 g/day was more consistently associated with performance benefits than acute (single-dose) supplementation. Benefits included improved maximal oxygen consumption (VO2max), reduced lactate accumulation, and enhanced recovery markers.
Acute supplementation (a single dose before exercise) generally failed to improve performance outcomes. This aligns with the pharmacokinetic reality that a single oral dose cannot meaningfully increase muscle carnitine content.
A separate meta-analysis published in Nutrients by Fielding and colleagues examined clinical effects of L-carnitine on physical performance from a rehabilitation perspective. They found that L-carnitine supplementation improved physical performance metrics including peak power output and recovery, with effects more pronounced in less-trained individuals and clinical populations compared to elite athletes.
A randomized, double-blind, placebo-controlled crossover study published in 2025 specifically tested 3 g of L-carnitine L-tartrate before CrossFit training and found no significant performance improvement in recreational athletes. This result, while negative, is consistent with the chronic-versus-acute pattern: a single pre-workout dose doesn't change muscle carnitine stores.
Recovery and Muscle Damage Reduction
Perhaps the most consistently supported benefit of L-carnitine in exercise contexts is its effect on recovery. A 2018 study by Spiering and colleagues at the US Army Research Institute found that 9 weeks of L-carnitine supplementation (2 g/day as L-carnitine L-tartrate) in resistance-trained males significantly reduced markers of exercise-induced muscle damage and oxidative stress compared to placebo.
The proposed mechanism involves multiple pathways. L-carnitine supports blood flow and oxygen delivery to exercising muscle by increasing nitric oxide production and reducing endothelial damage. It reduces the accumulation of hypoxanthine and xanthine (byproducts of ATP degradation that generate free radicals via xanthine oxidase). And it may stabilize muscle cell membranes, reducing mechanical damage during eccentric contractions.
For athletes and active individuals, the recovery benefit may be more practically valuable than any direct performance enhancement. Faster recovery enables higher training volume and frequency, which over time produces greater physiological adaptations and performance gains. This is an indirect but powerful pathway to improved athletic outcomes.
Injectable L-Carnitine for Athletes
While most exercise performance studies have used oral L-carnitine, the bioavailability advantage of injectable formulations suggests they could be particularly useful for athletic applications. Higher and more predictable plasma levels, achieved without GI side effects or the need for carbohydrate co-ingestion, could improve tissue loading efficiency.
Athletes interested in recovery support often stack L-carnitine with other compounds. BPC-157 is commonly used for tissue repair and injury recovery. Growth hormone-releasing peptides like CJC-1295/Ipamorelin or the oral secretagogue MK-677 support recovery through enhanced GH pulsatility. NAD+ supports mitochondrial function and cellular energy production. These combinations, while lacking formal combination trial data, follow a logical biochemical rationale and are widely used in integrative sports medicine settings.
The FormBlends Peptide Hub provides additional information on peptides commonly used in athletic and recovery contexts.
Endurance vs. Strength Training: Different Benefits
The type of exercise performed significantly influences how L-carnitine supplementation manifests its effects. Understanding these distinctions helps clinicians and athletes optimize their supplementation strategy.
Endurance exercise (running, cycling, swimming, rowing). During sustained aerobic activity below the lactate threshold, fat oxidation is a major energy source, supplying 50-70% of ATP production. This is precisely the metabolic context where carnitine's role in fatty acid transport is most relevant. Endurance athletes with elevated muscle carnitine content can theoretically maintain higher fat oxidation rates during long-duration events, sparing glycogen reserves for critical high-intensity moments (surges, sprints, hill climbs). The Stephens/Greenhaff studies from Nottingham demonstrated this glycogen-sparing effect directly: subjects with elevated muscle carnitine performed the same exercise bout while using less glycogen and more fat. For marathon runners, ultramarathon athletes, and Ironman competitors, even small improvements in fat oxidation efficiency over hours of racing could translate to meaningful performance advantages.
Resistance training (weight lifting, CrossFit, HIIT). During high-intensity, glycolytic exercise, carnitine's role shifts from fat transport to metabolic buffering. The acetyl-CoA buffering function, mediated by carnitine acetyltransferase (CrAT), becomes dominant. By accepting acetyl groups from excess acetyl-CoA, carnitine prevents the inhibition of pyruvate dehydrogenase, maintaining carbohydrate flux through the citric acid cycle and reducing lactate accumulation. For strength athletes, this translates to potentially improved work capacity during high-volume training sessions, reduced lactate-related fatigue during multiple-set protocols, and improved inter-set recovery.
The recovery benefits of L-carnitine may be even more valuable for resistance-trained athletes than for endurance athletes. Eccentric muscle contractions (the lowering phase of a lift) cause the greatest mechanical damage to muscle fibers, triggering an inflammatory response that produces soreness and temporary strength loss. L-carnitine's documented ability to reduce creatine kinase and lactate dehydrogenase release (markers of muscle cell membrane damage) suggests protective effects against this exercise-induced damage. Faster recovery between training sessions allows for higher training frequency, which over time produces greater strength and hypertrophy adaptations.
Interval and mixed-modality training. Activities that alternate between high and low intensities (interval training, team sports, CrossFit-style workouts) place unique demands on metabolic flexibility. Athletes must rapidly switch between fat-dominant and carbohydrate-dominant fuel systems. Carnitine supports both of these transitions: facilitating fat oxidation during low-intensity intervals and buffering acetyl-CoA during high-intensity bursts. This makes carnitine supplementation theoretically valuable across a wide range of athletic disciplines, not just pure endurance or pure strength sports.
Timing of L-Carnitine Administration Around Exercise
Practical questions about timing are common among athletes using injectable L-carnitine. The evidence suggests the following considerations:
Pre-exercise administration. For injectable L-carnitine, administration 30-60 minutes before training allows time for plasma levels to rise before exercise begins. The peak plasma concentration after IM injection typically occurs at 1-3 hours, so timing the injection to align with the training window maximizes acute plasma availability. However, a single injection won't significantly alter muscle carnitine stores in the acute time frame; the benefit comes from having elevated plasma carnitine available to support increased metabolic demand during the session.
Post-exercise administration. Some practitioners advocate post-exercise injection, reasoning that the combination of elevated blood flow to exercised muscles and potential insulin sensitivity improvements after training might facilitate better muscle carnitine uptake. While this rationale has some physiological basis, there's no direct evidence comparing pre- versus post-exercise injection timing for L-carnitine specifically.
Non-training day administration. For athletes using L-carnitine 2-3 times per week, some injections will inevitably fall on rest days. This is perfectly fine. The goal of consistent supplementation is to maintain elevated plasma carnitine levels over time, gradually increasing tissue stores. The acute timing relative to exercise is less important than the cumulative effect of sustained, regular supplementation.
Fasted vs. fed state. For oral L-carnitine, co-ingestion with carbohydrates is critical for muscle uptake (via insulin-mediated OCTN2 activation). For injectable L-carnitine, this consideration is less relevant since the absorption step is bypassed. However, some clinicians recommend that patients eat a meal containing carbohydrates within a few hours of injection to provide the insulin stimulus that facilitates muscle carnitine uptake from the bloodstream. The evidence for this recommendation is extrapolated from the oral carbohydrate co-ingestion studies rather than studied directly with injectable administration.
Figure 5: L-carnitine's dual role during exercise. At low intensities, it promotes fat oxidation and glycogen sparing. At high intensities, it buffers acetyl-CoA to reduce lactate production.
| Performance Parameter | Evidence Strength | Typical Protocol | Notes |
|---|---|---|---|
| Recovery / muscle damage reduction | Strong | 2 g/day for 9+ weeks | Most consistently supported benefit |
| Fat oxidation during exercise | Moderate | 2-3 g/day chronic + carbs (oral) | Requires elevated insulin for muscle uptake |
| Endurance capacity | Moderate | 2 g/day for 12+ weeks | More consistent in less-trained individuals |
| Lactate reduction | Moderate | 2-3 g/day chronic | Via acetyl-CoA buffering |
| Acute performance boost | Weak | Single pre-workout dose | Single doses don't change muscle stores |
| VO2max improvement | Mixed | Varies | Some positive results in specific populations |
Cardiovascular Benefits
L-Carnitine and the Heart
The heart is arguably the most carnitine-dependent organ in the body. Cardiac muscle derives approximately 60-70% of its ATP from fatty acid oxidation under normal resting conditions, making the carnitine shuttle system essential for maintaining cardiac energy supply. Carnitine concentrations in cardiac tissue are among the highest of any organ, reflecting this metabolic dependency.
This relationship has driven decades of research into L-carnitine supplementation for various cardiovascular conditions. The results have been encouraging, though the picture is complicated by the TMAO controversy we'll address shortly.
Heart Failure
A 2017 meta-analysis by Song, Qu, and Zhang published in Experimental and Therapeutic Medicine pooled data from 17 randomized controlled trials involving 1,625 patients with chronic heart failure. L-carnitine treatment was associated with statistically significant improvements in several cardiac parameters:
- Left ventricular ejection fraction improved by 4.14% (95% CI: 2.34-5.94, p < 0.001)
- Stroke volume increased by 8.21 mL (95% CI: 1.31-15.11, p = 0.02)
- Cardiac output improved by 0.88 L/min (95% CI: 0.47-1.29, p < 0.001)
- Significant decreases in BNP and NT-proBNP (biomarkers of heart failure severity)
- Reductions in left ventricular end-systolic and end-diastolic dimensions
However, the meta-analysis found no significant difference in all-cause mortality or 6-minute walk distance between L-carnitine and control groups. This dissociation between improved cardiac function markers and hard endpoints may reflect limited statistical power (most included studies were small) or may indicate that carnitine's cardiac benefits, while real, are not sufficient alone to change survival outcomes.
Acute Myocardial Infarction and Secondary Prevention
A 2013 systematic review and meta-analysis by DiNicolantonio and colleagues in Mayo Clinic Proceedings examined L-carnitine's role in secondary cardiovascular prevention. Pooling data from 13 controlled trials with 3,629 patients, they found compelling associations:
- 27% reduction in all-cause mortality (RR: 0.73, 95% CI: 0.54-0.99)
- 65% reduction in ventricular arrhythmias (RR: 0.35, 95% CI: 0.21-0.58, p < 0.0001)
- 40% reduction in angina development (RR: 0.60, 95% CI: 0.50-0.72, p < 0.001)
- No significant reduction in heart failure development or reinfarction
The mortality reduction is substantial and clinically meaningful. The anti-arrhythmic effect is particularly interesting, as it suggests that carnitine supplementation helps stabilize cardiac electrical activity during the vulnerable post-infarction period, possibly by maintaining adequate energy substrate supply to ischemic but viable myocardium.
Peripheral Arterial Disease
L-carnitine, particularly in its propionyl-L-carnitine form, has shown benefits for peripheral arterial disease (PAD), also known as intermittent claudication. Several clinical trials have demonstrated improved walking distance and reduced pain in PAD patients treated with propionyl-L-carnitine. The mechanism likely involves improved energy metabolism in ischemic skeletal muscle of the legs, where carnitine depletion occurs due to impaired blood supply.
Angina Pectoris
Multiple trials have evaluated L-carnitine in patients with stable angina. The consistent finding is that carnitine supplementation increases exercise tolerance and delays the onset of ST-segment depression (an ECG marker of myocardial ischemia) during exercise testing. These results make sense physiologically: during angina, the heart is energy-starved due to reduced blood flow, and optimizing the efficiency of available fatty acid oxidation helps maintain cardiac function with limited oxygen supply.
Mechanisms of Cardiovascular Protection
Beyond its direct role in cardiac energy metabolism, L-carnitine appears to protect the cardiovascular system through several additional mechanisms:
Anti-inflammatory effects. Carnitine supplementation has been shown to reduce circulating levels of C-reactive protein (CRP) and other inflammatory markers. Chronic inflammation is a driver of atherosclerosis, so anti-inflammatory effects may contribute to cardiovascular protection.
Endothelial function. L-carnitine supports nitric oxide bioavailability and endothelial function. Studies have shown improvements in flow-mediated dilation (a measure of endothelial health) with carnitine supplementation, which could improve vascular function and reduce atherosclerotic progression.
Antioxidant protection. By reducing oxidative stress in vascular tissues, carnitine may help prevent the oxidation of LDL cholesterol, a key step in atherogenesis. Carnitine's ability to improve mitochondrial efficiency also reduces electron leakage and superoxide generation from the electron transport chain.
Lipid profile modulation. Some studies have reported modest improvements in lipid profiles with L-carnitine supplementation, including reductions in total cholesterol and triglycerides, though results have been inconsistent across trials.
The Mendelian Randomization Perspective
A 2022 Mendelian randomization study published in BMC Medicine by Xu and colleagues used genetic variants associated with L-carnitine levels to assess causal relationships with cardiovascular outcomes. Mendelian randomization is a powerful epidemiological technique that uses genetic variation as a natural experiment, reducing confounding. Their analysis found evidence that higher genetically predicted L-carnitine levels were associated with reduced cardiovascular disease risk, providing genetic support for the observational and trial data.
However, the same study also flagged the complexity of the carnitine-TMAO axis, noting that the relationship between carnitine, TMAO, and cardiovascular outcomes may be more nuanced than initially proposed. This brings us to one of the most debated topics in carnitine research.
Cardiac Energy Metabolism: Why the Heart Needs Carnitine More Than Any Other Organ
To appreciate why L-carnitine supplementation shows such consistent cardiovascular benefits, it helps to understand the unique energy metabolism of cardiac muscle. The heart is arguably the most metabolically demanding organ in the body. It contracts roughly 100,000 times per day, never takes a rest, and consumes approximately 6-8 kg of ATP daily despite weighing only about 300 grams. To sustain this extraordinary workload, the heart maintains a metabolic rate 10-20 times higher than the average tissue.
Under normal resting conditions, the heart derives 60-70% of its ATP from fatty acid oxidation, with the remainder coming from glucose, lactate, and amino acids. This heavy reliance on fat as fuel makes the carnitine shuttle system absolutely essential for cardiac function. Cardiac tissue maintains some of the highest carnitine concentrations of any organ, typically 3,000-5,000 nmol/g tissue, compared to roughly 1,000-4,000 nmol/g in skeletal muscle and 100-200 nmol/g in liver.
During myocardial ischemia (reduced blood flow, as occurs during a heart attack or during angina), the heart's ability to oxidize fatty acids is impaired due to oxygen limitation. The heart shifts toward anaerobic glucose metabolism, which is less efficient and produces lactate. Meanwhile, fatty acyl-CoA intermediates accumulate, which are toxic to cell membranes and can disrupt cardiac electrical conduction. L-carnitine helps mitigate this ischemic toxicity by buffering excess acyl groups, forming less-toxic acylcarnitine esters that can be exported from the cell. This detoxification function, separate from its transport role, may explain carnitine's anti-arrhythmic effects observed in the post-MI meta-analyses.
Additionally, in the ischemic heart, carnitine's ability to accept acetyl groups from acetyl-CoA via CrAT helps maintain free CoA availability. Free CoA is essential for the citric acid cycle and for the activation of pyruvate dehydrogenase, which becomes critically important when the heart shifts toward glucose oxidation during ischemia. By maintaining CoA homeostasis, carnitine helps the ischemic heart more efficiently use whatever oxygen is available, maximizing ATP production from limited resources.
Diabetic Cardiomyopathy and Metabolic Syndrome
The intersection of metabolic syndrome, diabetes, and cardiovascular disease creates a context where L-carnitine supplementation may be particularly valuable. In diabetic cardiomyopathy, the heart becomes excessively reliant on fatty acid oxidation (up to 90% of ATP from fat) due to insulin resistance that impairs glucose uptake. This excessive fat oxidation increases oxygen consumption per unit of ATP produced (fatty acid oxidation requires more oxygen than glucose oxidation), reduces cardiac efficiency, and generates more reactive oxygen species.
L-carnitine supplementation in diabetic patients has been associated with improvements in cardiac function markers, lipid profiles, and glycemic control in several clinical trials. The mechanism likely involves multiple pathways: improved efficiency of fatty acid oxidation (so that fat is burned more completely with fewer toxic intermediates), better metabolic flexibility through acetyl-CoA buffering, reduced oxidative stress, and improved endothelial function.
For patients managing metabolic syndrome who are also using medications or peptides targeting insulin sensitivity and weight loss, L-carnitine may provide complementary cardiac protection. Patients on tirzepatide or semaglutide are already benefiting from the cardiovascular risk reduction documented in the major GLP-1 RA trials (SUSTAIN-6, PIONEER 6, SELECT, SURPASS-CVOT). Adding L-carnitine could theoretically provide additional metabolic support at the cardiac mitochondrial level, though this specific combination hasn't been studied in controlled trials.
L-Carnitine Versus Other Cardiovascular Supplements
Patients and clinicians often ask how L-carnitine compares to other supplements commonly used for cardiovascular support. Here's a comparative overview:
| Supplement | Primary CV Mechanism | Evidence Level | Complementary with L-Carnitine? |
|---|---|---|---|
| CoQ10 | Electron transport chain support, antioxidant | Strong for heart failure (Q-SYMBIO trial) | Yes - different mitochondrial targets |
| Omega-3 fatty acids | Anti-inflammatory, anti-arrhythmic, lipid lowering | Strong (REDUCE-IT trial) | Yes - omega-3s provide fuel, carnitine facilitates burning |
| Magnesium | Vasodilation, anti-arrhythmic, enzyme cofactor | Moderate | Yes - different mechanisms |
| D-Ribose | ATP precursor, purine salvage pathway | Limited | Yes - D-ribose supports ATP backbone, carnitine supports ATP production |
| Taurine | Antioxidant, calcium regulation, osmoregulation | Moderate for heart failure | Yes - complementary cardioprotective mechanisms |
| NAD+ | Mitochondrial function, sirtuin activation | Emerging | Yes - NAD+ and carnitine both support mitochondrial metabolism |
The take-home message is that L-carnitine's cardiovascular benefits are generally complementary to other evidence-based supplements, as each targets different aspects of cardiac energy metabolism, antioxidant defense, or vascular function. There are no significant antagonistic interactions between these compounds, and some clinicians use multi-targeted "cardioprotective stacks" that include several of these agents alongside standard pharmaceutical therapy.
Figure 6: Summary of cardiovascular outcome data from L-carnitine meta-analyses. Secondary prevention data show significant reductions in mortality, arrhythmias, and angina after myocardial infarction.
L-Carnitine and Male Fertility
Carnitine in Reproductive Biology
L-carnitine and its acetylated derivative, acetyl-L-carnitine, are found in extraordinarily high concentrations in the male reproductive tract, particularly in the epididymis. Epididymal carnitine concentrations can be 2,000 times higher than plasma levels, reflecting the compound's central role in sperm maturation and energy metabolism.
Sperm cells rely heavily on fatty acid oxidation for energy production during the long journey through the female reproductive tract to reach the oocyte. The carnitine shuttle system is active in sperm mitochondria, and adequate carnitine availability is necessary for normal sperm motility. Additionally, carnitine's antioxidant properties protect sperm DNA and cell membranes from oxidative damage, which is a leading cause of idiopathic male infertility.
Clinical Evidence for Sperm Quality Improvement
A 2023 systematic review published in the Journal of Clinical Medicine by Micic and colleagues comprehensively evaluated the evidence for L-carnitine supplementation in male fertility. The review confirmed several consistent findings across multiple randomized controlled trials.
Sperm motility. L-carnitine supplementation at doses of 1-3 g/day for 3-6 months consistently improved both total sperm motility and progressive motility. In a network meta-analysis of 23 randomized controlled trials examining various antioxidant supplements for idiopathic male infertility, L-carnitine was ranked first for improvements in sperm motility.
Sperm morphology. Improvements in normal sperm morphology have been reported across multiple trials. The same network meta-analysis ranked L-carnitine first for morphology improvements as well, suggesting it may be superior to other commonly used antioxidant supplements including CoQ10, vitamin E, and selenium.
Sperm concentration. Results for sperm concentration (count per mL) have been less consistent, with some studies showing improvement and others showing no significant change. The overall meta-analytic effect on concentration is not statistically significant.
DNA fragmentation. L-carnitine supplementation has been associated with reduced sperm DNA fragmentation in several studies, likely mediated by its antioxidant activity against reactive oxygen species in the seminal fluid.
Clinical Outcomes: Pregnancy Rates
Here's where the evidence requires careful interpretation. Despite clear improvements in measurable sperm quality parameters, the available data do not demonstrate a significant improvement in pregnancy rates with L-carnitine supplementation. This disconnect between laboratory parameters and clinical pregnancy outcomes is a recurring theme in male infertility research and may reflect the multifactorial nature of conception, the complexity of sperm function beyond what standard semen analysis measures, or insufficient statistical power in existing trials.
A 2022 review in Reproduction and Fertility by Kowalczyk and colleagues specifically examined L-carnitine and L-acetylcarnitine for idiopathic male infertility and reached similar conclusions: improvements in semen parameters without confirmed improvements in clinical pregnancy rates. The authors called for larger, well-designed trials with pregnancy as the primary endpoint.
Comparison with Other Antioxidant Approaches
A head-to-head comparison trial published in the European Review for Medical and Pharmacological Sciences compared L-carnitine (2 g/day) against a combination of CoQ10 and vitamin E for idiopathic male infertility. L-carnitine produced superior improvements in sperm motility and morphology compared to the CoQ10/vitamin E combination, supporting its position as a first-line antioxidant supplement for male fertility.
Dosing for Fertility Applications
Research protocols for male fertility typically use higher doses and longer durations than general supplementation:
| Form | Typical Fertility Dose | Duration | Key Findings |
|---|---|---|---|
| L-Carnitine | 2-3 g/day oral; 500 mg-1 g injectable | 3-6 months minimum | Best for motility and morphology |
| Acetyl-L-Carnitine (ALC) | 1-3 g/day | 3-6 months | Better blood-testis barrier penetration |
| L-Carnitine + ALC combination | 2 g LC + 1 g ALC/day | 6 months | May provide complementary benefits |
The duration requirement, typically 3-6 months, aligns with the spermatogenesis cycle, which takes approximately 74 days for a new sperm cell to develop from spermatogonium to mature spermatozoon. Interventions aimed at improving sperm quality need at least one full cycle to show effects, and most protocols recommend two full cycles (about 5-6 months) for optimal assessment.
Injectable L-carnitine may offer advantages in fertility applications due to its superior bioavailability and avoidance of GI side effects that can reduce compliance during the lengthy supplementation periods required. However, studies specifically comparing injectable versus oral L-carnitine for fertility outcomes have not been published.
Clinical Perspective on Male Fertility
L-carnitine ranks among the most evidence-supported supplements for improving measurable semen parameters. For men with idiopathic infertility undergoing fertility evaluation, L-carnitine supplementation (alone or combined with acetyl-L-carnitine) represents a low-risk, relatively inexpensive intervention with consistent effects on motility and morphology. However, patients should be counseled that improved semen parameters don't guarantee improved pregnancy rates, and that L-carnitine should be part of a comprehensive fertility optimization approach, not a standalone treatment.
The Oxidative Stress Connection in Male Infertility
To understand why L-carnitine is so effective at improving semen parameters, it's essential to understand the role of oxidative stress in male infertility. Spermatozoa are uniquely vulnerable to oxidative damage for several biological reasons.
First, mature sperm cells have very little cytoplasm and therefore minimal antioxidant enzyme content. Most cells carry strong intracellular antioxidant defenses including superoxide dismutase, catalase, and glutathione peroxidase. Sperm cells shed most of their cytoplasm during spermatogenesis, leaving them with far less of this protective machinery than other cell types.
Second, sperm cell membranes are exceptionally rich in polyunsaturated fatty acids (PUFAs), particularly docosahexaenoic acid (DHA). These PUFAs are essential for membrane fluidity and the acrosome reaction needed for fertilization, but they are also highly susceptible to lipid peroxidation by reactive oxygen species. When ROS attack these membrane PUFAs, the resulting lipid peroxides damage membrane integrity, reduce motility, and impair the sperm's ability to undergo the acrosome reaction and fuse with the oocyte.
Third, sperm mitochondria in the midpiece generate ROS as a byproduct of the intense oxidative phosphorylation needed to power the flagellar motor. A moderate level of ROS is actually required for normal sperm capacitation and hyperactivation, but excessive ROS production overwhelms the limited antioxidant defenses and triggers oxidative damage.
Fourth, sperm DNA is particularly vulnerable because it's tightly condensed with protamines rather than histones, making DNA repair mechanisms less accessible. Oxidative DNA damage in sperm can cause DNA fragmentation, base modifications, and cross-linking, all of which impair fertility potential and may affect embryo development even if fertilization occurs.
L-carnitine addresses this oxidative vulnerability through multiple mechanisms. It directly scavenges ROS in seminal plasma. It enhances the expression and activity of endogenous antioxidant enzymes in the reproductive tract. It supports efficient mitochondrial electron transport, reducing electron leakage and superoxide generation. And by maintaining efficient fatty acid oxidation in sperm mitochondria, it ensures that the energy needs of the flagellar motor are met without excessive metabolic stress.
The combination of these antioxidant effects with carnitine's direct role in sperm energy metabolism explains why it consistently outperforms other antioxidant supplements in head-to-head comparisons. Most antioxidants (vitamin C, vitamin E, selenium) address only the ROS-scavenging aspect. L-carnitine addresses both the oxidative damage and the underlying energy metabolism that drives sperm motility.
The Role of Acetyl-L-Carnitine in Epididymal Maturation
Acetyl-L-carnitine (ALCAR) deserves specific attention in the fertility context because it plays a unique role in sperm maturation that goes beyond its antioxidant properties. As spermatozoa transit through the epididymis (a journey that takes approximately 12 days), they undergo critical maturation changes that confer motility and fertilization capacity. During this transit, sperm cells progressively acquire carnitine and acetylcarnitine from the surrounding epididymal fluid.
The epididymis concentrates L-carnitine to levels approximately 2,000 times higher than plasma, making it one of the most carnitine-enriched tissues in the body. This extraordinary concentration gradient is maintained by active OCTN2-mediated transport. The carnitine and acetylcarnitine accumulated by maturing sperm cells serve as energy reserves for the long journey through the female reproductive tract, where external nutrient availability is limited.
ALCAR specifically provides acetyl groups that can be used for immediate energy production without requiring the full beta-oxidation pathway, giving sperm a rapid-access energy source for the bursts of hyperactivated motility needed during capacitation and oocyte penetration. This is why combination therapy with both L-carnitine (for sustained energy via fat oxidation) and ALCAR (for rapid-access energy and antioxidant protection) has shown superior results in some fertility studies compared to either compound alone.
For men pursuing fertility optimization alongside metabolic health goals, the combination of injectable L-carnitine (for systemic metabolic support and improved bioavailability) with oral ALCAR (for direct reproductive tract effects and blood-brain barrier penetration) represents a rational dual-route approach. Some compounding pharmacies offer ALCAR in injectable form as well, though the oral form has been used in most fertility research.
Dosing Protocols
Injectable L-Carnitine Dosing
Injectable L-carnitine is available through compounding pharmacies as levocarnitine injection, typically supplied in multi-dose vials at concentrations ranging from 200 mg/mL to 500 mg/mL. The FDA-approved form (Carnitor injection) is supplied at 200 mg/mL for intravenous use in dialysis patients, but compounded formulations for subcutaneous and intramuscular use follow different concentration standards.
Weight Management Protocols
Clinical weight management protocols commonly use injectable L-carnitine at the following doses:
| Protocol | Dose | Frequency | Route | Duration |
|---|---|---|---|---|
| Standard fat metabolism support | 500 mg | 2-3x per week | IM or SC | 8-12 weeks |
| Intensive weight loss program | 1,000 mg | 2-3x per week | IM | 12-16 weeks |
| Maintenance/cycling | 500 mg | 1-2x per week | IM or SC | Ongoing with cycling |
| Pre-workout performance | 500-1,000 mg | 30-60 min before training | IM | As needed with cycling |
Lipotropic MIC Injection Protocols
MIC injections are typically administered on a weekly schedule in clinical practice:
| Protocol | Formulation | Volume | Frequency | Notes |
|---|---|---|---|---|
| Basic MIC | MIC 25/50/50 mg/mL | 1 mL | Weekly | Standard lipotropic support |
| Lipo-B | MIC + B12 1 mg/mL | 1 mL | Weekly | Most commonly prescribed |
| Lipo-C Enhanced | MIC + B12 + L-Carnitine | 1-2 mL | Weekly to 2x/week | Combined fat metabolism support |
| Loading phase | Any MIC variant | 1 mL | 2x per week for 4 weeks | Initial period for new patients |
Oral L-Carnitine Dosing (for Comparison)
For patients who prefer oral supplementation, the following doses are commonly used in research protocols:
- General supplementation: 500-2,000 mg/day in divided doses
- Athletic performance: 2-3 g/day, ideally with 60-80 g carbohydrate for insulin-mediated muscle uptake
- Male fertility: 2-3 g/day L-carnitine + 1 g/day acetyl-L-carnitine for 3-6 months
- Cardiovascular support: 1-3 g/day, based on meta-analysis protocols
Important: at oral doses above 2 g, absorption efficiency drops markedly and GI side effects become common. There's limited value in exceeding 2-3 g oral per day, as the excess is simply not absorbed.
Combination Protocol Considerations
When incorporating injectable L-carnitine or lipotropic injections into a peptide-based protocol, timing and coordination matter. Here are common combination approaches used in clinical practice:
L-Carnitine + GLP-1 agonists: Patients using semaglutide or tirzepatide for weight management may add L-carnitine injections to support fat oxidation during the caloric deficit these agents create. L-carnitine is typically administered on different days from GLP-1 injections for simplicity and to avoid multiple injection sites on the same day.
L-Carnitine + AOD-9604: AOD-9604 promotes lipolysis (fat release from adipocytes), and L-carnitine supports the subsequent oxidation of those released fatty acids. Some protocols administer both compounds on the same day, as they target different steps in the fat utilization pathway.
L-Carnitine + GH-releasing peptides: Growth hormone promotes lipolysis, particularly during fasting and overnight. Combining L-carnitine with GH-releasing peptides like CJC-1295/Ipamorelin aims to maximize the body's utilization of GH-mobilized fatty acids.
MIC + L-Carnitine: The Lipo-C formulation combines MIC with L-carnitine in a single injection, simplifying administration while providing both hepatic lipotropic support and mitochondrial fat transport enhancement.
Cycling and Duration
There's no universally agreed-upon cycling protocol for injectable L-carnitine, but clinical practice generally follows these patterns:
- 12 weeks on, 4 weeks off: A common approach that allows periodic assessment of whether the patient still benefits from supplementation
- Continuous use during active weight loss: Some clinicians maintain L-carnitine throughout the weight loss phase and discontinue during maintenance
- Seasonal cycling: Athletes may use L-carnitine during intensive training blocks and reduce or stop during off-season
- Indefinite low-frequency maintenance: Once-weekly injections can be maintained long-term with periodic monitoring
Lab Testing to Guide L-Carnitine Dosing
Informed dosing decisions benefit from appropriate laboratory assessment. Several tests can help clinicians evaluate a patient's carnitine status and guide supplementation protocols:
Total and free carnitine levels. Plasma total carnitine and free (unconjugated) carnitine levels provide the most direct assessment of carnitine status. Normal plasma free carnitine ranges from approximately 25-50 micromoles per liter in adults. Levels below 20 micromol/L suggest deficiency, while levels in the low-normal range (20-30 micromol/L) may indicate suboptimal status in patients with high metabolic demands. Serial testing at baseline and after 8-12 weeks of supplementation can document response to therapy.
Acylcarnitine profile. The ratio of acylcarnitine (esterified carnitine) to free carnitine reflects the balance between fatty acid flux and carnitine availability. An elevated acylcarnitine/free carnitine ratio (greater than 0.4) suggests that carnitine is being consumed faster than it's being replenished, indicating a potential benefit from supplementation. Acylcarnitine profiling can also reveal specific fatty acid oxidation disorders that might be contributing to metabolic symptoms.
TMAO levels. For patients using oral L-carnitine or those concerned about cardiovascular risk, measuring plasma TMAO can provide reassurance or identify patients who should switch to injectable administration. TMAO testing is available through several reference laboratories and specialty metabolic testing services.
Metabolic panels and lipid profiles. Standard metabolic panels and lipid profiles provide context for L-carnitine's effects on glucose metabolism and lipid handling. Improvements in triglycerides, fasting glucose, and liver enzymes may indicate that carnitine supplementation is providing metabolic benefit.
Inflammatory markers. High-sensitivity CRP, interleukin-6, and other inflammatory markers can be tracked to assess L-carnitine's anti-inflammatory effects in patients with elevated baseline inflammation.
Injection Technique for Patients
For patients self-administering injectable L-carnitine or lipotropic injections at home (with a valid prescription and under medical supervision), proper injection technique is essential for safety and efficacy.
Intramuscular (IM) injection technique:
- Wash hands thoroughly with soap and water
- Prepare the injection site (deltoid, vastus lateralis, or ventrogluteal) with an alcohol swab
- Draw the prescribed volume into a sterile syringe using a drawing needle (typically 18-20 gauge)
- Switch to the injection needle (typically 22-25 gauge, 1-1.5 inch length depending on injection site and body composition)
- With the skin taut, insert the needle at a 90-degree angle into the muscle
- Aspirate briefly to ensure you're not in a blood vessel
- Inject the solution slowly and steadily
- Withdraw the needle and apply gentle pressure with a clean gauze pad
- Dispose of the needle and syringe in a sharps container
Subcutaneous (SC) injection technique:
- Wash hands thoroughly
- Prepare the injection site (abdominal fat pad, avoiding a 2-inch radius around the navel; or upper outer thigh)
- Draw the prescribed volume into a sterile syringe
- Use a 27-30 gauge, 0.5-inch needle for subcutaneous injection
- Pinch a fold of skin and fat at the injection site
- Insert the needle at a 45-90 degree angle (depending on body composition; leaner patients use 45 degrees)
- Inject the solution slowly
- Release the skin fold and withdraw the needle
- Apply gentle pressure; do not rub
Patients should rotate injection sites to prevent lipodystrophy (changes in subcutaneous fat at the injection site) and to maintain consistent absorption. A simple rotation pattern, alternating between left and right sides of the body and between upper and lower injection sites, prevents repeated injections at the same location.
Dosing Considerations
All dosing information presented is derived from published research protocols and clinical practice patterns. Individual dosing should be determined by a qualified healthcare provider based on the patient's specific clinical situation, goals, lab work, and concurrent medications. Self-administration of injectable compounds without medical supervision is not recommended.
Figure 7: Example 16-week dosing timeline for combined L-carnitine and lipotropic injection protocols in a clinical weight management setting.
Safety Profile
General Safety of L-Carnitine
L-carnitine has a well-established safety profile supported by decades of clinical use. The FDA-approved injectable formulation (Carnitor) has been used in dialysis patients since the 1990s, and oral L-carnitine supplements have been widely available for even longer. At standard doses (up to 3 g/day oral or up to 1 g injectable), serious adverse effects are uncommon.
The most commonly reported side effects of oral L-carnitine are gastrointestinal in nature: nausea, vomiting, abdominal cramps, and diarrhea. These effects are dose-dependent and typically limit practical oral dosing to 2-3 g/day. An unusual side effect of high-dose oral carnitine is a fishy body odor caused by the excretion of trimethylamine, a metabolite produced by gut bacteria.
Injectable L-carnitine largely avoids GI side effects since it bypasses the digestive tract. Injection-site reactions, including mild pain, redness, or swelling, are the most common adverse effects of intramuscular or subcutaneous administration. These are generally mild and self-limiting.
The TMAO Controversy
The most significant safety concern surrounding L-carnitine supplementation relates to trimethylamine N-oxide (TMAO), a metabolite that has been associated with increased cardiovascular risk in observational studies.
The pathway works like this: intestinal bacteria metabolize L-carnitine to trimethylamine (TMA). TMA is absorbed into the bloodstream and transported to the liver, where the enzyme flavin-containing monooxygenase 3 (FMO3) oxidizes it to TMAO. Elevated plasma TMAO levels have been correlated with increased risk of atherosclerosis, thrombotic events, and cardiovascular mortality in multiple observational cohort studies.
The landmark 2013 paper by Koeth and colleagues in Nature Medicine demonstrated that gut microbiota metabolism of L-carnitine in red meat promotes atherosclerosis in mouse models and that TMAO levels predict cardiovascular events in human cohorts. This finding generated considerable controversy and concern about carnitine supplementation safety.
However, several important nuances merit consideration:
The TMAO-carnitine paradox. If oral L-carnitine supplementation raises TMAO and TMAO causes cardiovascular disease, then carnitine supplementation should worsen cardiovascular outcomes. But the meta-analyses discussed earlier show the opposite: L-carnitine supplementation is associated with improved cardiovascular outcomes, including reduced mortality post-MI. This paradox suggests the relationship between carnitine, TMAO, and cardiovascular risk is more complex than a simple linear pathway.
Mendelian randomization challenges. While observational studies show correlations between TMAO and cardiovascular events, Mendelian randomization studies examining genetic variants that influence TMAO levels have produced mixed results regarding causal relationships. Some suggest TMAO may be a biomarker of gut dysbiosis or dietary patterns rather than a direct causal agent of cardiovascular disease.
Route matters. Injectable L-carnitine bypasses the gut entirely, minimizing bacterial conversion to TMA. This provides a theoretical safety advantage over oral supplementation for patients concerned about TMAO. While some systemic L-carnitine may be excreted back into the intestinal lumen and undergo bacterial conversion, the overall TMAO production from injectable carnitine is expected to be substantially lower than from equivalent oral doses.
Dietary context. Regular red meat consumers harbor more carnitine-metabolizing bacteria and produce more TMAO from carnitine than vegetarians. Supplement-related TMAO production depends heavily on the individual's baseline gut microbiome composition.
Mitigating factors. Research has identified several dietary components that may reduce TMAO production, including resveratrol, berberine, garlic-derived allicin, and dietary fiber. Maintaining a diverse, plant-rich diet may help offset TMAO production from carnitine supplementation.
Drug Interactions
L-carnitine has relatively few clinically significant drug interactions, but several merit awareness:
| Drug/Class | Interaction | Clinical Significance |
|---|---|---|
| Warfarin and other anticoagulants | L-carnitine may potentiate anticoagulant effects | Monitor INR; dose adjustments may be needed |
| Thyroid hormones | L-carnitine may inhibit thyroid hormone entry into cell nuclei | May reduce thyroid hormone efficacy; monitor thyroid function |
| Valproic acid | Valproate depletes carnitine stores | Carnitine supplementation often indicated in VPA-treated patients |
| Pivalic acid-containing antibiotics | These drugs deplete carnitine stores | Carnitine supplementation may be needed during treatment |
| Zidovudine (AZT) | AZT inhibits carnitine-dependent fatty acid oxidation | L-carnitine supplementation may help manage myopathy |
Contraindications and Precautions
Seizure disorders. There are case reports of increased seizure frequency in patients with pre-existing seizure disorders who received high-dose intravenous L-carnitine. While the causal relationship is uncertain, caution is advised in this population.
Hypothyroidism. L-carnitine has been shown to inhibit the entry of thyroid hormones (both T3 and T4) into cell nuclei in vitro. While this effect has been proposed as potentially therapeutic for hyperthyroidism, it raises theoretical concerns about exacerbating hypothyroidism in treated patients. Thyroid function should be monitored in patients on thyroid replacement therapy who begin carnitine supplementation.
Renal impairment. Patients with significantly impaired kidney function may have altered carnitine metabolism and clearance. The FDA-approved IV carnitine product is specifically indicated for carnitine deficiency in dialysis patients, but dosing must be adjusted based on pre- and post-dialysis carnitine levels.
Pregnancy and lactation. There is insufficient data on supplemental L-carnitine use during pregnancy and breastfeeding. While carnitine is a naturally occurring substance and dietary carnitine is considered safe, supplemental doses above normal dietary intake should be used during pregnancy only when the potential benefit justifies the potential risk.
Safety of MIC Injection Components
Each component of MIC injections has its own safety considerations:
Methionine: High-dose methionine supplementation can raise homocysteine levels, a risk factor for cardiovascular disease. However, the doses used in standard MIC injections (25 mg) are too low to meaningfully affect homocysteine in most patients. Patients with homocystinuria or significantly elevated homocysteine should exercise caution.
Inositol: Inositol has an excellent safety profile even at doses far exceeding those used in lipotropic injections. Oral doses up to 18 g/day have been used in psychiatric research with minimal adverse effects (mainly GI upset at high oral doses). The small doses in MIC injections are very unlikely to cause problems.
Choline: The upper tolerable intake level for choline is 3.5 g/day for adults. MIC injections provide 50 mg, well below this threshold. At very high oral doses, choline can cause fishy body odor, sweating, GI distress, and hypotension, but these effects are not relevant to standard injection protocols.
Vitamin B12: B12 has no established upper tolerable intake level because toxicity is essentially unheard of. The body excretes excess B12 through the kidneys. Rare allergic reactions to injectable B12 have been reported but are extremely uncommon.
Monitoring Recommendations
For patients receiving regular injectable L-carnitine or lipotropic injections, the following monitoring schedule is reasonable:
- Baseline: Complete metabolic panel, lipid panel, free and total carnitine levels, thyroid function (if on thyroid medication), homocysteine (if elevated risk), B12 level
- 4-6 weeks: Reassess symptoms, injection site tolerance, basic metabolic panel
- 12 weeks: Repeat carnitine levels, lipid panel, metabolic markers; assess body composition changes
- Ongoing: Periodic monitoring every 3-6 months during continued use; thyroid function if applicable
Safety Summary
Injectable L-carnitine and lipotropic MIC injections are generally well-tolerated with a favorable safety profile at standard doses. The TMAO concern is primarily relevant to high-dose oral supplementation and is largely mitigated by injectable routes that bypass gut bacterial metabolism. Drug interactions are limited but clinically relevant with anticoagulants and thyroid hormones. As with any injectable compound, proper medical supervision and periodic monitoring are recommended.
Figure 8: Recommended safety monitoring schedule for patients receiving injectable L-carnitine and lipotropic injection protocols.
Additional Research Areas
L-Carnitine and Insulin Resistance
Emerging research has highlighted L-carnitine's potential role in improving insulin sensitivity, a finding with implications that extend well beyond weight management. In states of insulin resistance, such as type 2 diabetes and metabolic syndrome, mitochondrial fatty acid oxidation is often impaired, leading to the accumulation of lipotoxic intermediates (ceramides, diacylglycerols) in skeletal muscle and liver. These lipid metabolites interfere with insulin signaling cascades.
By supporting more efficient fatty acid oxidation through the carnitine shuttle, L-carnitine supplementation may help clear these lipotoxic intermediates and restore insulin signaling. Several clinical trials have demonstrated improved glucose disposal rates and reduced fasting glucose with L-carnitine supplementation in insulin-resistant populations.
This mechanism aligns well with the approaches taken by GLP-1 receptor agonists and dual agonists like tirzepatide, which also improve insulin sensitivity through different pathways. The combination of L-carnitine with GLP-1-based therapies offers complementary mechanisms targeting both the demand side (improved insulin signaling) and the supply side (enhanced fatty acid clearance) of metabolic health.
Neuroprotective Properties
Acetyl-L-carnitine (ALCAR), the acetylated form of L-carnitine, crosses the blood-brain barrier more readily than L-carnitine itself and has been studied for neuroprotective effects. ALCAR serves as a source of acetyl groups for acetylcholine synthesis and supports mitochondrial function in neurons.
Clinical research has examined ALCAR for peripheral neuropathy (particularly diabetic neuropathy), cognitive decline in aging, and depression. A meta-analysis of randomized controlled trials found that ALCAR supplementation significantly reduced depression scores compared to placebo, with effect sizes comparable to some antidepressant medications, though with fewer side effects.
For patients managing both metabolic health and cognitive function, the combination of injectable L-carnitine (for systemic fat metabolism support) with oral ALCAR (for central nervous system effects) may provide complementary benefits across multiple organ systems.
L-Carnitine in Chronic Kidney Disease
Carnitine deficiency is common in patients undergoing hemodialysis because carnitine is efficiently removed by dialysis membranes but cannot be adequately replaced through dietary intake alone. The FDA-approved indication for intravenous levocarnitine is specifically the treatment of carnitine deficiency in dialysis patients.
In this population, IV carnitine supplementation has been associated with improved lipid profiles, reduced erythropoietin requirements (suggesting improved red blood cell production), improved exercise tolerance, reduced intradialytic hypotension, and improved quality of life measures. These benefits reflect the correction of true carnitine deficiency rather than supplementation above normal levels.
Cancer-Related Fatigue
L-carnitine deficiency is frequently observed in cancer patients, particularly those receiving chemotherapy agents like cisplatin, ifosfamide, and doxorubicin, which can deplete carnitine stores. Cancer-related fatigue, one of the most debilitating symptoms reported by oncology patients, may be partly attributable to carnitine depletion.
Several clinical trials have evaluated L-carnitine supplementation for cancer-related fatigue with mixed but generally positive results. A systematic review found that carnitine supplementation improved fatigue scores in carnitine-deficient cancer patients, though benefits were less consistent in carnitine-replete patients.
Aging and Sarcopenia
As we age, skeletal muscle carnitine content declines, coinciding with reductions in mitochondrial function and increased susceptibility to sarcopenia (age-related muscle loss). L-carnitine supplementation in older adults has been associated with improvements in physical performance, reduced fatigue, and better preservation of lean body mass in some studies.
The combination of L-carnitine with growth hormone-releasing peptides may be particularly relevant for aging populations. Compounds like CJC-1295/Ipamorelin support GH secretion, which promotes lean mass preservation and fat mobilization, while L-carnitine supports the downstream oxidation of mobilized fatty acids and mitochondrial function in aging muscle.
Polycystic Ovary Syndrome (PCOS)
Women with PCOS frequently exhibit insulin resistance, elevated androgens, and impaired metabolic profiles. L-carnitine supplementation has been studied in this population with encouraging results. Several randomized controlled trials have demonstrated improvements in insulin sensitivity, lipid profiles, and inflammatory markers in women with PCOS receiving L-carnitine.
Combined with inositol (already a component of MIC injections and independently studied for PCOS), L-carnitine may provide complementary metabolic support for this common condition. GLP-1 receptor agonists like semaglutide are also being investigated for PCOS, and the combination of these approaches represents an emerging area of interest in metabolic endocrinology.
L-Carnitine and Type 2 Diabetes
The relationship between L-carnitine and type 2 diabetes deserves detailed exploration because the metabolic disruptions seen in diabetes directly involve carnitine-dependent pathways. In type 2 diabetes, insulin resistance in skeletal muscle impairs glucose uptake, forcing greater reliance on fatty acid oxidation for energy. Simultaneously, elevated circulating free fatty acids (from insulin-resistant adipose tissue that fails to suppress lipolysis appropriately) flood the liver and muscle with lipid substrates.
This creates a metabolic overload scenario: the carnitine shuttle is asked to handle dramatically increased fatty acid flux. If carnitine availability is insufficient, incomplete fatty acid oxidation occurs, generating toxic lipid intermediates like ceramides and diacylglycerols that further worsen insulin resistance. This creates a self-reinforcing cycle that some researchers have termed "lipotoxicity."
Several randomized controlled trials have evaluated L-carnitine supplementation in type 2 diabetes with generally positive results. A 2018 review by Bene and colleagues in Nutrition and Diabetes summarized the evidence, noting improvements in fasting glucose, HbA1c, total cholesterol, LDL cholesterol, and markers of oxidative stress. The magnitude of improvement in glycemic control was modest (typically 0.2-0.5% HbA1c reduction), but clinically relevant when viewed as an adjunct to standard diabetes management.
The mechanism appears to involve improved mitochondrial fatty acid handling (reducing lipotoxic intermediate accumulation), enhanced glucose oxidation through acetyl-CoA buffering (improving PDH activity), and direct insulin-sensitizing effects that may involve activation of insulin signaling pathways. Some preclinical research has also suggested that L-carnitine may protect pancreatic beta cells from lipotoxicity, potentially preserving insulin secretory capacity.
For diabetic patients who are also using GLP-1 receptor agonists or dual GIP/GLP-1 agonists, L-carnitine supplementation may provide complementary metabolic support. While the GLP-1-based medications address insulin secretion, appetite, and central metabolic regulation, L-carnitine supports the peripheral machinery of fatty acid oxidation that handles the increased lipid flux during weight loss.
Thyroid Function and L-Carnitine
An often-overlooked area of L-carnitine research involves its interactions with thyroid hormone action. L-carnitine has been shown to inhibit the entry of both triiodothyronine (T3) and thyroxine (T4) into cell nuclei in vitro, effectively reducing thyroid hormone activity at the cellular level. This inhibitory effect has been proposed as potentially therapeutic for hyperthyroidism.
A randomized, double-blind, placebo-controlled trial by Benvenga and colleagues demonstrated that L-carnitine (2-4 g/day) could ameliorate symptoms of hyperthyroidism, including preventing bone loss associated with thyroid excess. Patients receiving L-carnitine showed improvements in thyrotoxic symptoms including heart rate, nervousness, and tremor.
For hypothyroid patients on thyroid hormone replacement, however, this interaction raises a theoretical concern. If L-carnitine reduces the cellular action of exogenous thyroid hormones, patients on levothyroxine or liothyronine might require dose adjustments when starting carnitine supplementation. The clinical significance of this interaction at standard supplementation doses hasn't been fully characterized, but prudent practice includes monitoring thyroid function (TSH, free T4, free T3) in thyroid patients who begin L-carnitine.
This thyroid-carnitine connection also has implications for weight management. Hypothyroidism is a common cause of weight gain and metabolic slowdown. Patients being treated for hypothyroidism who add L-carnitine for weight management should be aware that carnitine might blunt the metabolic-boosting effect of their thyroid medication, potentially requiring thyroid dose optimization.
L-Carnitine in Pregnancy: Limited Data, Important Considerations
Carnitine plays essential roles during pregnancy. Fetal carnitine synthesis is limited, and the developing fetus relies on placental transfer of maternal carnitine. Maternal plasma carnitine levels naturally decline during pregnancy, likely due to increased fetal demand, expanded plasma volume, and enhanced renal clearance. This physiological decline raises the question of whether carnitine supplementation during pregnancy might be beneficial.
Some observational studies have suggested associations between low maternal carnitine levels and adverse pregnancy outcomes including gestational diabetes, preeclampsia, and intrauterine growth restriction. However, the evidence is insufficient to recommend routine carnitine supplementation during pregnancy, and the safety of supplemental doses above normal dietary intake hasn't been established in pregnant populations.
Women planning pregnancy who have been using injectable L-carnitine or lipotropic injections should discuss discontinuation timing with their healthcare provider. Most clinicians recommend stopping all non-essential supplements before conception or immediately upon discovering pregnancy, then reassessing with the obstetrician.
Exercise-Induced Immune Function and L-Carnitine
Intense, prolonged exercise can transiently suppress immune function, creating a "window of vulnerability" to upper respiratory infections and other opportunistic infections. This phenomenon, often called exercise-induced immunosuppression, is particularly relevant for endurance athletes, military personnel, and others engaged in demanding physical training.
L-carnitine has been shown to modulate immune function in exercising individuals through several mechanisms. It reduces exercise-induced oxidative stress that damages immune cells. It supports lymphocyte proliferation and function. And it may attenuate the cortisol response to intense exercise, which is a primary driver of post-exercise immunosuppression.
Several studies have reported reduced incidence of upper respiratory tract infections in athletes supplementing with L-carnitine during intensive training periods. While the evidence base isn't as strong as for recovery or cardiovascular applications, immune support represents another potential benefit that contributes to L-carnitine's value proposition for athletes and active individuals.
Athletes interested in immune optimization during training often combine L-carnitine with other immune-supporting strategies. NAD+ supports cellular energy metabolism in immune cells, while BPC-157 has demonstrated anti-inflammatory properties that may help modulate the inflammatory response to intense training.
Patient Selection and Practical Integration
Assessment Framework for L-Carnitine and Lipotropic Therapy
Determining which patients are likely to benefit from injectable L-carnitine or lipotropic injections requires a structured assessment that considers metabolic status, lifestyle factors, treatment goals, and concurrent therapies. Rather than offering these injections as a one-size-fits-all weight management adjunct, thoughtful patient selection can improve outcomes and cost-effectiveness.
Step 1: Metabolic baseline assessment. Before initiating carnitine or lipotropic therapy, a comprehensive metabolic baseline should be established. This includes fasting lipid panel (total cholesterol, LDL, HDL, triglycerides), comprehensive metabolic panel (liver enzymes, kidney function, glucose), HbA1c or fasting insulin (to assess insulin resistance), thyroid panel (TSH, free T4; add free T3 if thyroid disease is suspected), vitamin B12 and folate levels, homocysteine (if cardiovascular risk factors are present), and total and free carnitine levels (if available through the laboratory).
Patients with elevated triglycerides, evidence of hepatic steatosis, insulin resistance, low B12, or documented carnitine insufficiency are likely to benefit most from supplementation. Patients with already-normal metabolic parameters may receive less measurable benefit, though subjective improvements in energy and exercise recovery can still justify treatment.
Step 2: Dietary and lifestyle assessment. Understanding the patient's dietary pattern helps identify potential nutritional gaps that lipotropic injections could address. Key questions include: How much red meat and dairy do they consume (primary dietary carnitine sources)? Do they eat eggs regularly (primary dietary choline source)? Are they following a calorie-restricted diet, and if so, how aggressively? What is their exercise frequency, intensity, and type? Are they taking any medications known to deplete carnitine or B vitamins?
Patients on plant-based diets, aggressive caloric restriction, or carnitine-depleting medications have the strongest nutritional rationale for supplementation. Patients consuming a balanced omnivorous diet with adequate calories may have less to gain from a purely nutritional standpoint, though the pharmacological benefits of supraphysiologic carnitine levels during exercise or weight loss may still apply.
Step 3: Treatment goal alignment. Different treatment goals call for different protocols. A patient primarily seeking weight loss support benefits from a different approach than one focused on athletic performance or fertility. Clarifying the primary and secondary goals helps select the appropriate formulation (L-carnitine alone vs. MIC vs. Lipo-C), dosing frequency, and duration, as well as complementary interventions that align with those goals.
Step 4: Integration with existing therapy. Many patients seeking L-carnitine or lipotropic injections are already using other treatments. Common combinations include GLP-1 agonists (semaglutide or tirzepatide), growth hormone-releasing peptides (CJC-1295/Ipamorelin), fat-targeting peptides (AOD-9604, Fragment 176-191), metabolic modulators (5-Amino-1MQ, tesofensine), thyroid hormones, metformin, and other medications. Creating a coordinated injection schedule that minimizes patient burden while maintaining appropriate spacing between compounds is an important practical consideration.
Creating a Practical Injection Schedule
For patients managing multiple injectable compounds, a well-organized weekly schedule reduces confusion and improves compliance. Here's an example framework for a patient using several peptide and lipotropic compounds:
| Day | Morning | Evening | Notes |
|---|---|---|---|
| Monday | L-Carnitine 500 mg IM | CJC/Ipamorelin SC (if applicable) | L-carnitine on training days when possible |
| Tuesday | - | CJC/Ipamorelin SC (if applicable) | Rest from injectable metabolics |
| Wednesday | L-Carnitine 500 mg IM | CJC/Ipamorelin SC (if applicable) | - |
| Thursday | Lipo-B 1 mL IM | CJC/Ipamorelin SC (if applicable) | Weekly lipotropic injection |
| Friday | Semaglutide SC (if applicable) | CJC/Ipamorelin SC (if applicable) | GLP-1 agonist on its designated day |
| Saturday | - | - | Injectable-free day when possible |
| Sunday | - | - | Injectable-free day |
This is illustrative only; actual schedules should be customized by the prescribing provider. The key principles are: space injectable compounds across different days when possible, maintain consistency in timing for each compound, reserve at least one or two injection-free days per week when feasible, and use different injection sites for different compounds to prevent tissue irritation.
Managing Patient Expectations
Clear communication about realistic expectations is essential for patient satisfaction and long-term compliance. Several common misconceptions need to be addressed proactively:
"Lipotropic injections will melt my fat." This is the most common misconception. Lipotropic injections do not directly destroy fat cells or cause dramatic fat loss. They provide metabolic support that may enhance the body's ability to process and utilize fat during a caloric deficit. The effect is supportive, not transformative. Patients who expect dramatic weight loss from lipotropic injections alone will be disappointed.
"I'll feel a dramatic energy boost immediately." While some patients, particularly those with pre-existing B12 deficiency, do notice improved energy within the first week or two, many patients experience a more gradual, subtle improvement that may take several weeks to become apparent. The metabolic effects of L-carnitine build over time as tissue stores increase. Setting the expectation for gradual improvement prevents premature discontinuation.
"These are just 'skinny shots' from a weight loss clinic." While the marketing around lipotropic injections can be overly promotional, the biochemistry behind MIC and L-carnitine is legitimate. Helping patients understand the genuine metabolic roles of these compounds, while maintaining honesty about the limitations of the clinical evidence, builds trust and informed consent.
"I don't need to change my diet or exercise." L-carnitine and lipotropic injections are metabolic support tools, not substitutes for the fundamental energy balance equation. Patients must understand that these injections work within the context of a comprehensive program that includes caloric management, regular physical activity, adequate sleep, and stress management. Without these foundations, the injections alone are unlikely to produce meaningful results.
Cost-Benefit Considerations
Injectable L-carnitine and lipotropic injections represent an out-of-pocket cost for most patients, as they are generally not covered by insurance for weight management or performance applications (though injectable carnitine may be covered for diagnosed primary carnitine deficiency or dialysis-related deficiency).
Typical costs vary by region and provider but generally range from $15-50 per lipotropic injection (Lipo-B or MIC) and $25-75 per L-carnitine injection, depending on dose, formulation, and whether the injection is administered in-office or self-administered at home from a multi-dose vial. Multi-dose vials dispensed for home use are generally more cost-effective per injection than in-office administration.
When evaluating cost-effectiveness, patients should consider the bioavailability comparison: a $25 injectable dose of 500 mg L-carnitine delivers approximately the same usable carnitine as $5-10 worth of oral L-carnitine supplements (2-3 grams/day for several days), minus the GI side effects and TMAO production. The per-milligram cost of absorbed carnitine is not as different between routes as the per-dose cost might suggest.
For patients whose primary motivator is weight loss, the cost of L-carnitine and lipotropic injections should be weighed against the costs of the health consequences of remaining overweight: diabetes medication, cardiovascular risk, joint replacement surgery, and reduced quality of life. In this context, modest expenditure on metabolic support that contributes to successful weight management may represent good value.
Compounding and Formulation Considerations
Regulatory Status
It's important to understand the regulatory landscape for injectable L-carnitine and lipotropic injection products. The FDA-approved levocarnitine injection (Carnitor, manufactured by Leadiant Biosciences) is specifically indicated for the prevention and treatment of carnitine deficiency in patients with end-stage renal disease undergoing dialysis. This is the only FDA-approved injectable L-carnitine product.
All other injectable L-carnitine and lipotropic injection formulations used in weight management and wellness settings are compounded preparations. Compounding pharmacies operate under Section 503A or 503B of the Federal Food, Drug, and Cosmetic Act. Section 503A pharmacies compound on a per-patient basis with a valid prescription, while 503B outsourcing facilities can produce larger batches under cGMP (current Good Manufacturing Practice) conditions and are subject to FDA inspection.
When selecting a compounding pharmacy for injectable L-carnitine or lipotropic products, clinicians and patients should look for:
- PCAB (Pharmacy Compounding Accreditation Board) accreditation or state-equivalent
- 503B registration with the FDA (for outsourcing facilities)
- Third-party potency and sterility testing of products
- Proper beyond-use dating (BUD) based on USP guidelines
- Certificates of analysis available for each batch
Formulation Stability
Injectable L-carnitine is generally stable in aqueous solution, but stability can be affected by pH, temperature, and the presence of other ingredients in multi-component formulations. L-carnitine injection solutions are typically formulated at a pH of 6.0-6.5 and should be stored at controlled room temperature (20-25 degrees C) unless otherwise specified by the compounder.
Multi-component lipotropic formulations require more careful stability considerations. Each ingredient must remain stable and compatible in the presence of the others throughout the assigned beyond-use date. Compounding pharmacies should have stability data supporting the BUD assigned to their specific formulations.
Storage and Handling
Patients receiving injectable L-carnitine or lipotropic injections should be counseled on proper storage:
- Store multi-dose vials as directed, typically at room temperature or refrigerated depending on formulation
- Protect from light if indicated on the label
- Do not use if the solution appears discolored or contains particulate matter
- Follow the beyond-use date strictly; discard any remaining contents after the BUD
- Practice proper aseptic technique when withdrawing doses from multi-dose vials
- Use a new sterile needle and syringe for each injection
Quality Indicators for Compounded Injectables
Not all compounding pharmacies are created equal, and the quality of compounded injectable products can vary significantly. Patients and clinicians should be aware of several quality indicators when selecting a compounding pharmacy for L-carnitine and lipotropic injection products.
USP compliance. The United States Pharmacopeia (USP) publishes chapters that govern compounding practices. USP Chapter 797 specifically covers sterile compounding and sets standards for personnel training, environmental monitoring, equipment, facilities, and quality assurance for sterile preparations like injectable products. Pharmacies that comply with current USP 797 standards follow rigorous protocols for ensuring the sterility, potency, and stability of their products.
Third-party testing. Quality-focused compounding pharmacies submit finished products for independent third-party testing to verify potency (the product contains the labeled amount of each active ingredient), sterility (no microbial contamination), endotoxin levels (pyrogen testing), and particulate matter (ensuring the product is free of visible and sub-visible particles). Certificates of analysis from these tests should be available upon request.
Beyond-use dating. The beyond-use date (BUD) assigned to a compounded product must be based on stability data for that specific formulation, not simply borrowed from the BUD of individual ingredients. Multi-component lipotropic formulations require stability studies confirming that all active ingredients remain potent and compatible throughout the assigned BUD. Conservative BUD assignment (shorter dating based on cautious interpretation of stability data) is generally preferable to overly aggressive dating that risks product degradation.
Source ingredient quality. The quality of active pharmaceutical ingredients (APIs) used in compounding matters. Pharmacies should use USP-grade or equivalent APIs from qualified, audited suppliers. Some pharmacies use lower-grade chemicals that may contain impurities or have uncertain potency. Asking the pharmacy about their API sourcing and whether they maintain supplier qualification files is a reasonable quality check.
Environmental monitoring. Sterile compounding must occur in controlled environments with appropriate ISO-classified clean rooms, laminar airflow workbenches or isolators, and regular environmental monitoring (air sampling, surface sampling). Pharmacies should be able to describe their clean room classifications and monitoring protocols.
Home Storage and Handling Best Practices
For patients receiving multi-dose vials for home self-administration, proper storage and handling directly affect product quality and safety throughout the life of the vial.
Temperature control. Most compounded L-carnitine and lipotropic injection vials should be stored at controlled room temperature (20-25 degrees Celsius, or 68-77 degrees Fahrenheit) unless the label specifies refrigeration. Some formulations, particularly those containing B12 (which is light-sensitive) or preservative-free preparations, may require refrigeration. Always follow the specific storage instructions on the vial label. Avoid exposing vials to extreme heat (such as leaving them in a car during summer) or freezing temperatures.
Light protection. Cyanocobalamin (B12) is photosensitive, meaning it degrades when exposed to light. Lipotropic formulations containing B12 should be stored in the original packaging or wrapped in foil to minimize light exposure. Amber vials provide some light protection, but additional measures are still recommended for products stored at room temperature.
Contamination prevention. Multi-dose vials are designed for multiple uses, but each entry through the rubber stopper creates a potential contamination pathway. Always clean the stopper with an alcohol swab before inserting a needle. Use a new sterile needle for each draw. Never touch the stopper with bare fingers after cleaning. If the vial contents become cloudy, discolored, or show visible particles, discard the vial immediately.
Tracking usage. When you first puncture a multi-dose vial, write the date on the label. Most multi-dose vials with preservatives have a 28-day beyond-use dating once punctured, though some may be longer. Track the number of doses withdrawn to ensure you're not exceeding the prescribed quantity or using the vial past its punctured BUD, whichever comes first.
Sharps disposal. Used needles and syringes must be disposed of in FDA-cleared sharps disposal containers. Never throw needles in household trash, flush them, or place them in recycling bins. When the sharps container is about three-quarters full, seal it and dispose of it according to your local community guidelines. Many pharmacies, hospitals, and public health departments offer sharps disposal programs.
Figure 9: Common compounded lipotropic injection formulations and their component concentrations. Formulations vary between compounding pharmacies.
Clinical Case Considerations
The following hypothetical clinical scenarios illustrate how injectable L-carnitine and lipotropic injections might be incorporated into different patient management contexts. These are not treatment recommendations but rather illustrative frameworks for understanding clinical decision-making.
Scenario 1: Metabolic Syndrome Patient on GLP-1 Therapy
Consider a 48-year-old male with a BMI of 34, prediabetes (fasting glucose 118 mg/dL, HbA1c 6.1%), dyslipidemia (triglycerides 220 mg/dL, HDL 36 mg/dL), and mild hepatic steatosis on ultrasound. He has been started on semaglutide for weight management and is beginning to lose weight at a rate of approximately 1-1.5 kg per week.
In this scenario, injectable L-carnitine could serve several purposes. The rapid weight loss means increased fatty acid flux from adipose tissue to the liver, increasing the demand for hepatic fat processing and mitochondrial fatty acid oxidation. L-carnitine injections (500 mg, 2-3 times per week) could support the liver's capacity to handle this increased lipid load while optimizing fat oxidation in skeletal muscle. Simultaneously, a weekly Lipo-B injection could provide additional hepatic support through MIC components, with the B12 component supporting energy levels that might otherwise decline during caloric restriction.
The combination protocol might look like: semaglutide once weekly (per prescribing guidelines), L-carnitine 500 mg IM twice weekly (on non-semaglutide days), and Lipo-B 1 mL IM once weekly. Lab monitoring would include baseline and 12-week carnitine levels, comprehensive metabolic panel, lipid panel, and liver enzymes.
Scenario 2: Female Patient with PCOS and Insulin Resistance
Consider a 32-year-old female with PCOS, BMI 30, insulin resistance (HOMA-IR 4.2), irregular menstrual cycles, and difficulty conceiving. She is not yet ready for fertility treatments but wants to optimize her metabolic health and improve her chances of natural conception.
This patient has several overlapping indications for L-carnitine and lipotropic support. L-carnitine has been shown to improve insulin sensitivity in PCOS patients. Inositol (a component of MIC injections) has strong evidence for improving insulin resistance and ovulatory function in PCOS. If she decides to pursue fertility treatments in the future, L-carnitine's beneficial effects on oocyte quality (an emerging research area) could be relevant.
A reasonable approach might include: Lipo-C injections (MIC + L-carnitine) weekly, supplemented with oral myo-inositol (2-4 g/day) for additional insulin-sensitizing effects. If she progresses to active fertility efforts, her partner could be started on L-carnitine supplementation for semen quality optimization.
Scenario 3: Competitive Endurance Athlete During Training Block
Consider a 35-year-old male marathon runner preparing for a qualifying race. He's increasing training volume to 80-100 miles per week, wants to optimize fat oxidation efficiency during racing, and reports that recovery from long runs is slower than expected.
For this athlete, the primary goal of L-carnitine supplementation is to support the glycogen-sparing fat oxidation shift during prolonged running and to enhance recovery between training sessions. Injectable L-carnitine (1,000 mg IM, 2-3 times per week) provides reliable tissue loading without the GI issues that oral carnitine can cause during high-volume training (many endurance athletes already have GI sensitivity during intense training blocks).
The protocol might run for 16 weeks leading into the goal race, with carnitine injections administered on easy training days to allow for potential injection-site soreness. BPC-157 might be added for general tissue repair and recovery support. Post-race, the carnitine protocol could be reduced to maintenance dosing (once weekly) or discontinued until the next training block.
Scenario 4: Older Adult Addressing Sarcopenia and Metabolic Health
Consider a 67-year-old female with a BMI of 27, declining muscle mass (confirmed by DEXA), fatigue, borderline high cholesterol, and mild cognitive complaints. She's moderately active (walks daily, does light resistance training twice weekly) but feels her energy and physical capacity have declined significantly over the past few years.
Age-related carnitine decline, reduced mitochondrial function, and sarcopenia all converge in this scenario. Injectable L-carnitine (500 mg twice weekly) could support mitochondrial energy production in both skeletal muscle and cardiac tissue. Lipo-B injections would address potential B12 insufficiency (common in older adults due to reduced intrinsic factor and gastric acid) and provide metabolic methylation support.
If growth hormone secretion is also declining (as is expected with aging), CJC-1295/Ipamorelin could be considered to support GH-mediated fat mobilization and lean mass preservation, with L-carnitine supporting the downstream oxidation of mobilized fatty acids. NAD+ could further support mitochondrial function and cellular energy production.
For the cognitive complaints, oral acetyl-L-carnitine (ALCAR) at 1-2 g/day could be added, as ALCAR crosses the blood-brain barrier and has evidence supporting cognitive function in aging populations. This creates a dual-form approach: injectable L-carnitine for systemic metabolic support, oral ALCAR for cognitive and neurological support.
Comparison with Other Fat-Targeting Compounds
L-carnitine and lipotropic injections exist within a broader category of compounds used for fat metabolism support and weight management. Understanding how they compare to other available options helps clinicians and patients make informed decisions about which approaches to include in a comprehensive protocol.
L-Carnitine vs. GLP-1 Receptor Agonists
GLP-1 receptor agonists like semaglutide and dual GIP/GLP-1 agonists like tirzepatide represent the most evidence-based pharmacological tools currently available for weight management. They produce weight loss through appetite suppression, delayed gastric emptying, improved insulin sensitivity, and central nervous system satiety signaling. Clinical trials have demonstrated weight reductions of 15-20% or more of body weight with these agents.
L-carnitine operates through an entirely different mechanism. It doesn't suppress appetite, delay gastric emptying, or directly influence central satiety pathways. Instead, it supports the downstream metabolic processing of fatty acids that have already been mobilized. This makes L-carnitine genuinely complementary to GLP-1 agonists rather than competitive. GLP-1 agonists create the conditions for fat loss (caloric deficit, improved metabolic signaling); L-carnitine supports the machinery that processes the mobilized fat. Neither substitutes for the other, and they can be used concurrently without pharmacological conflict.
The magnitude of effect is also different. GLP-1 agonists produce large, clinically obvious effects on body weight. L-carnitine's contribution to body weight reduction is modest by comparison (1-2 kg in meta-analyses). However, L-carnitine may contribute benefits that GLP-1 agonists don't directly provide, including improved exercise recovery, cardiovascular protection through cardiac energy metabolism support, and enhanced metabolic flexibility.
L-Carnitine vs. AOD-9604 and Fragment 176-191
AOD-9604 and Fragment 176-191 are modified peptide fragments derived from human growth hormone that specifically stimulate lipolysis (fat release from adipocytes) without the full spectrum of growth hormone effects. They primarily target adipose tissue, promoting the breakdown of stored triglycerides into free fatty acids.
L-carnitine addresses the step that comes after lipolysis: the transport of those released fatty acids into mitochondria for oxidation. In theory, combining a lipolytic agent with L-carnitine creates a more complete fat utilization pathway. Without adequate carnitine-dependent transport capacity, fatty acids released by AOD-9604 or Fragment 176-191 could simply recirculate and be re-esterified rather than being oxidized for energy. Conversely, providing ample carnitine without a lipolytic stimulus may not maximize fat utilization if the rate of fatty acid release from adipose tissue is the limiting factor.
This complementary relationship is the strongest pharmacological rationale for combining L-carnitine with lipolytic peptides. The combination addresses both the "release" and "burn" sides of fat utilization.
L-Carnitine vs. 5-Amino-1MQ
5-Amino-1MQ is a small molecule that inhibits nicotinamide N-methyltransferase (NNMT), an enzyme implicated in adipose tissue energy metabolism. By inhibiting NNMT, 5-Amino-1MQ shifts cellular metabolism away from fat storage and toward energy expenditure. It operates through transcriptional and epigenetic mechanisms, essentially reprogramming fat cell behavior rather than directly transporting or burning fat.
L-carnitine works at a completely different level, the mitochondrial transport of fatty acids. The two compounds target different bottlenecks in fat metabolism. 5-Amino-1MQ addresses the cellular "decision" to store versus burn fat. L-carnitine addresses the physical transport of fat into the mitochondrial furnace. Like the AOD-9604 combination, there's a logical rationale for using these compounds together, though clinical data on the specific combination are not available.
L-Carnitine vs. Tesofensine
Tesofensine is a triple monoamine reuptake inhibitor that reduces appetite while also increasing resting energy expenditure. It works through norepinephrine, dopamine, and serotonin pathways in the central nervous system. Clinical trials have shown weight loss of 10-12% with tesofensine, making it one of the more effective weight loss agents under investigation.
L-carnitine complements tesofensine by supporting the peripheral fat oxidation that increases during tesofensine-induced caloric deficit and enhanced metabolic rate. While tesofensine drives the caloric deficit through appetite suppression and increased expenditure, L-carnitine ensures the mitochondrial machinery can efficiently process the increased fat substrate flux. See the tesofensine research report for more details on this compound.
L-Carnitine vs. MOTS-c
MOTS-c is a mitochondrial-derived peptide that activates AMPK (AMP-activated protein kinase), the cell's master energy sensor. AMPK activation promotes mitochondrial biogenesis, glucose uptake, and fatty acid oxidation while inhibiting fat synthesis and storage. MOTS-c essentially upgrades the cellular energy infrastructure.
L-carnitine and MOTS-c target related but distinct aspects of mitochondrial metabolism. MOTS-c increases the number and quality of mitochondria (the factories). L-carnitine supplies fuel to those factories (fatty acid transport). More mitochondria without adequate fuel supply is like building factories without raw materials. More fuel without adequate factory capacity creates a metabolic bottleneck. Together, they create a more complete mitochondrial optimization strategy.
| Compound | Primary Mechanism | Complementary with L-Carnitine? | Combined Rationale |
|---|---|---|---|
| Semaglutide/Tirzepatide | Appetite suppression, insulin sensitization | Yes, strongly | Creates deficit + supports fat oxidation |
| AOD-9604 / Fragment 176-191 | Lipolysis (fat release) | Yes, strongly | Release fat + transport fat to mitochondria |
| 5-Amino-1MQ | NNMT inhibition (metabolic reprogramming) | Yes | Reprogram cells + supply mitochondrial transport |
| Tesofensine | Triple reuptake inhibitor (appetite + expenditure) | Yes | Increase demand + support supply processing |
| MOTS-c | AMPK activation, mitochondrial biogenesis | Yes, strongly | Build more mitochondria + fuel them with fat |
| CJC-1295/Ipamorelin | GH release (lipolysis, lean mass) | Yes | GH mobilizes fat + carnitine burns it |
Emerging Research and Future Directions
L-Carnitine and the Gut-Heart Axis
The relationship between gut microbiome composition, carnitine metabolism, TMAO production, and cardiovascular outcomes represents one of the most actively researched areas in carnitine science. Recent research has moved beyond the initial alarming TMAO findings to develop a more nuanced understanding of this complex axis.
A 2024 review in Nutrients revisited the role of carnitine in heart disease through the lens of the gut microbiota, noting that the relationship between carnitine, TMAO, and cardiovascular risk is modulated by multiple factors including gut microbiome diversity, dietary context, fish consumption (which also raises TMAO but is cardioprotective), and individual genetic variation in TMAO metabolism.
Research into TMAO-lowering strategies, including specific probiotic strains, prebiotic fibers, and phytochemicals like resveratrol and berberine that can shift the gut microbiome away from TMA-producing species, may eventually allow patients to receive oral L-carnitine supplementation while mitigating TMAO production. This could be relevant for patients who prefer oral over injectable administration or who want to optimize both gut health and carnitine status simultaneously.
The injectable route, of course, largely sidesteps this entire question by avoiding significant gut exposure. But understanding the gut-heart axis helps explain why oral and injectable L-carnitine may have genuinely different risk profiles, rather than simply different absorption profiles.
Carnitine and Epigenetics
An emerging area of interest involves L-carnitine's potential influence on epigenetic regulation. Through its role in acetyl-CoA metabolism (via carnitine acetyltransferase), L-carnitine can influence the availability of acetyl groups for histone acetylation, a key epigenetic modification that controls gene expression. Histone acetylation generally promotes gene transcription, while deacetylation silences genes.
Acetyl-L-carnitine, by providing readily available acetyl groups, has been shown in preclinical studies to influence histone acetylation patterns in neural tissue, potentially contributing to its neuroprotective and antidepressant effects. Whether similar epigenetic effects occur in other tissues (muscle, liver, adipose) with L-carnitine supplementation is an active area of investigation.
If L-carnitine influences epigenetic programming in metabolically relevant tissues, it could affect gene expression patterns related to fat storage, insulin sensitivity, and mitochondrial biogenesis, effects that would persist beyond the immediate pharmacokinetic window of supplementation. This could help explain some of the sustained benefits observed with chronic carnitine supplementation that seem disproportionate to the modest pharmacological effects of acute dosing.
Mitochondrial Biogenesis and Carnitine
Beyond supporting existing mitochondrial function, there's evidence that L-carnitine may promote the creation of new mitochondria, a process called mitochondrial biogenesis. Preclinical studies have shown that L-carnitine supplementation can activate PGC-1alpha, the master regulator of mitochondrial biogenesis, in muscle and liver tissue.
If confirmed in human studies, this would represent a qualitative step beyond carnitine's established role as a fatty acid transporter. Rather than just improving the efficiency of existing mitochondria, carnitine might actually increase the total mitochondrial density in metabolically active tissues. More mitochondria means greater oxidative capacity, improved insulin sensitivity, and enhanced metabolic flexibility, effects that align with the long-term goals of metabolic optimization protocols.
This area of research connects with other compounds being studied for mitochondrial biogenesis, including exercise itself (the most potent physiological stimulus for mitochondrial biogenesis), NAD+ precursors (which activate sirtuins that regulate PGC-1alpha), and the mitochondrial-derived peptide MOTS-c. A comprehensive mitochondrial optimization approach might combine several of these interventions for additive or greater-than-additive effects on mitochondrial density and function.
Precision Dosing: Pharmacogenomics and Individual Variation
As personalized medicine advances, there's growing interest in tailoring L-carnitine supplementation to individual genetic and metabolic profiles. Several areas of genetic variation could influence optimal dosing and route selection:
OCTN2 (SLC22A5) polymorphisms. Variations in the gene encoding the primary carnitine transporter affect both intestinal absorption and renal reabsorption of carnitine. Patients with less-efficient OCTN2 variants may absorb oral carnitine poorly and may benefit more from injectable administration.
FMO3 polymorphisms. The flavin-containing monooxygenase 3 enzyme converts TMA to TMAO in the liver. Genetic variants that increase FMO3 activity could amplify TMAO production from oral carnitine, making injectable administration more appropriate for these individuals. Conversely, patients with reduced FMO3 activity (including those with trimethylaminuria, or "fish odor syndrome") may produce less TMAO from oral carnitine but may experience more TMA-related body odor.
CPT1 and CPT2 variants. Polymorphisms in the genes encoding the carnitine shuttle enzymes can affect the rate of fatty acid oxidation. Patients with CPT variants associated with reduced enzyme activity might benefit more from optimizing carnitine availability, as sufficient substrate (carnitine) can partially compensate for reduced enzyme efficiency.
While pharmacogenomic-guided carnitine dosing isn't yet standard clinical practice, the framework for personalized supplementation is developing. Genetic testing panels that include metabolic pathway variants are becoming more accessible, and integrating these results with clinical response data could eventually allow truly individualized carnitine protocols.
L-Carnitine in Long COVID and Post-Viral Fatigue
The COVID-19 pandemic brought renewed attention to post-viral fatigue syndromes, and L-carnitine has emerged as a supplement of interest for long COVID management. Several observations support this interest: many long COVID patients report profound fatigue and exercise intolerance; mitochondrial dysfunction has been proposed as a contributing mechanism; and some patients show biochemical evidence of impaired fatty acid oxidation.
Early clinical reports and small trials have suggested improvements in fatigue, exercise tolerance, and quality of life in long COVID patients receiving L-carnitine supplementation. The rationale aligns with carnitine's established benefits in other fatigue-related conditions (cancer-related fatigue, chronic fatigue syndrome, dialysis-related fatigue) where mitochondrial energy metabolism is compromised.
While the evidence is still preliminary and larger trials are needed, L-carnitine represents a low-risk intervention that addresses a plausible pathophysiological mechanism in post-viral fatigue. Injectable formulations may be particularly appropriate for long COVID patients who report GI symptoms (common in this population) that might limit oral supplement tolerance.
Frequently Asked Questions
Summary of Evidence by Application
For clinicians and patients reviewing this extensive body of evidence, the following summary table provides a quick-reference guide to the strength of evidence supporting L-carnitine and lipotropic injections across different clinical applications.
| Application | Evidence Strength | Key Findings | Recommended Form |
|---|---|---|---|
| Post-MI cardiovascular protection | Strong (meta-analysis) | 27% mortality reduction, 65% arrhythmia reduction | Oral or injectable L-carnitine |
| Chronic heart failure support | Strong (meta-analysis) | Improved LVEF (+4.14%), cardiac output (+0.88 L/min) | Oral or injectable L-carnitine |
| Male fertility (semen parameters) | Strong (multiple RCTs) | Ranked #1 for motility and morphology improvement | Oral or injectable L-carnitine + oral ALCAR |
| Exercise recovery / muscle damage | Moderate-Strong | Reduced CK, LDH, myoglobin markers | Injectable L-carnitine (avoid GI issues during training) |
| Body weight / fat mass reduction | Moderate (meta-analysis) | -1.21 kg weight, -2.08 kg fat mass; better with >12 weeks | Injectable L-carnitine (superior bioavailability) |
| Insulin sensitivity improvement | Moderate | Improved glucose disposal, reduced fasting glucose | Oral or injectable L-carnitine |
| Peripheral arterial disease | Moderate | Improved walking distance | Propionyl-L-carnitine (oral) |
| Hepatic fat metabolism support | Moderate (individual components) | Choline prevents fatty liver; methionine supports fat export | MIC/Lipo-B injections |
| Depression (ALCAR) | Moderate (meta-analysis) | Effect sizes comparable to some antidepressants | Oral ALCAR |
| Endurance exercise performance | Mixed | Chronic supplementation more effective than acute | Injectable or oral with carbohydrate |
| Cancer-related fatigue | Limited-Moderate | Benefit primarily in carnitine-deficient patients | Injectable L-carnitine |
| MIC injections for weight loss | Limited (no RCTs of combination) | Biochemical rationale strong; clinical outcome data absent | MIC/Lipo-B injection |
| Male fertility (pregnancy rates) | Insufficient | Improved parameters but no demonstrated pregnancy improvement | Oral or injectable L-carnitine |
This evidence hierarchy helps set appropriate clinical expectations. Applications with strong evidence (cardiovascular protection, semen parameter improvement, exercise recovery) have the most compelling rationale for supplementation. Applications with moderate evidence (body composition, insulin sensitivity) may benefit specific patient populations. Applications with limited evidence (MIC combination for weight loss) should be presented honestly, with patients understanding the gap between biochemical rationale and proven clinical outcomes.
The free assessment at FormBlends can help patients and providers determine which compounds and protocols may be most appropriate for their specific metabolic goals. The dosing calculator provides additional guidance on protocol planning for injectable compounds, including L-carnitine and lipotropic formulations.
Across all applications, injectable L-carnitine offers a consistent pharmacokinetic advantage over oral administration: near-complete bioavailability, no GI side effects, minimal TMAO production, and predictable dosing. Whether this pharmacokinetic advantage translates to superior clinical outcomes for a given application depends on whether oral bioavailability limitations are a meaningful bottleneck for that specific use case. For applications where tissue carnitine loading is important (muscle stores for exercise, cardiac reserves for cardiovascular protection), the injectable route's ability to maintain consistently higher plasma levels likely offers a genuine clinical advantage. As research continues to refine our understanding of carnitine metabolism, optimal dosing strategies, and the interplay between the gut microbiome and systemic carnitine levels, injectable formulations are expected to maintain their position as the preferred delivery method for patients seeking reliable and predictable metabolic support from L-carnitine supplementation.
References
- Harper P, Elwin CE, Cederblad G. Pharmacokinetics of bolus intravenous and oral doses of L-carnitine in healthy subjects. Eur J Clin Pharmacol. 1988;35(1):69-75. doi:10.1007/BF00555510
- Rebouche CJ. Pharmacokinetics of L-carnitine. Ann N Y Acad Sci. 2004;1033:30-41. doi:10.1196/annals.1320.003
- Evans AM, Fornasini G. Pharmacokinetics of L-carnitine. Clin Pharmacokinet. 2003;42(11):941-967. doi:10.2165/00003088-200342110-00002
- Koeth RA, Wang Z, Levison BS, et al. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat Med. 2013;19(5):576-585. doi:10.1038/nm.3145
- Flanagan JL, Simmons PA, Vehige J, Willcox MD, Garrett Q. Role of carnitine in disease. Nutr Metab (Lond). 2010;7:30. doi:10.1186/1743-7075-7-30
- Fritz IB. Carnitine and its role in fatty acid metabolism. Adv Lipid Res. 1963;1:285-334. doi:10.1016/B978-1-4831-9937-5.50013-4
- McGarry JD, Brown NF. The mitochondrial carnitine palmitoyltransferase system. From concept to molecular analysis. Eur J Biochem. 1997;244(1):1-14. doi:10.1111/j.1432-1033.1997.00001.x
- Ramsay RR, Gandour RD, van der Leij FR. Molecular enzymology of carnitine transfer and transport. Biochim Biophys Acta. 2001;1546(1):21-43. doi:10.1016/S0167-4838(01)00147-9
- Stephens FB, Constantin-Teodosiu D, Greenhaff PL. New insights concerning the role of carnitine in the regulation of fuel metabolism in skeletal muscle. J Physiol. 2007;581(Pt 2):431-444. doi:10.1113/jphysiol.2006.125799
- Wall BT, Stephens FB, Constantin-Teodosiu D, Marimuthu K, Macdonald IA, Greenhaff PL. Chronic oral ingestion of L-carnitine and carbohydrate increases muscle carnitine content and alters muscle fuel metabolism during exercise in humans. J Physiol. 2011;589(Pt 4):963-973. doi:10.1113/jphysiol.2010.201343
- Talenezhad N, Mohammadi M, Ramezani-Jolfaie N, Mozaffari-Khosravi H, Salehi-Abargouei A. Effects of L-carnitine supplementation on weight loss and body composition: A systematic review and meta-analysis of 37 randomized controlled clinical trials with dose-response analysis. Clin Nutr ESPEN. 2020;37:9-23. doi:10.1016/j.clnesp.2020.03.008
- Villani RG, Gannon J, Self M, Rich PA. L-Carnitine supplementation combined with aerobic training does not promote weight loss in moderately obese women. Int J Sport Nutr Exerc Metab. 2000;10(2):199-207. doi:10.1123/ijsnem.10.2.199
- Gnoni A, Longo S, Gnoni GV, Giudetti AM. Effect of acute and chronic oral L-carnitine supplementation on exercise performance based on the exercise intensity: a systematic review. Nutrients. 2021;12(12):4244. doi:10.3390/nu12124244
- Fielding R, Riede L, Luber JP, Mankowski RT. L-Carnitine supplementation in recovery after exercise. Nutrients. 2018;10(3):349. doi:10.3390/nu10030349
- Spiering BA, Kraemer WJ, Vingren JL, et al. Responses of criterion variables to different supplemental doses of L-carnitine L-tartrate. J Strength Cond Res. 2007;21(1):259-264. doi:10.1519/00124278-200702000-00046
- Pons DG, Moran C, Alorda-Clara M, et al. The bright and the dark sides of L-carnitine supplementation: a systematic review. J Int Soc Sports Nutr. 2020;17(1):49. doi:10.1186/s12970-020-00377-2
- DiNicolantonio JJ, Lavie CJ, Fares H, Menezes AR, O'Keefe JH. L-carnitine in the secondary prevention of cardiovascular disease: systematic review and meta-analysis. Mayo Clin Proc. 2013;88(6):544-551. doi:10.1016/j.mayocp.2013.02.007
- Song X, Qu H, Yang Z, Rong J, Cai W, Zhou H. Efficacy and safety of L-carnitine treatment for chronic heart failure: a meta-analysis of randomized controlled trials. Exp Ther Med. 2017;13(5):2281-2288. doi:10.3892/etm.2017.4263
- Xu M, Zheng J, Hou T, et al. L-carnitine, a friend or foe for cardiovascular disease? A Mendelian randomization study. BMC Med. 2022;20(1):272. doi:10.1186/s12916-022-02477-z
- Micic S, Lalic N, Djordjevic D, et al. L-Carnitine and male fertility: is supplementation beneficial? J Clin Med. 2023;12(18):5796. doi:10.3390/jcm12185796
- Kowalczyk K, Blazejewska-Hyzorek B, Plotek W, Kusz DJ. L-carnitine and L-acetylcarnitine supplementation for idiopathic male infertility. Reprod Fertil. 2022;3(1):R37-R46. doi:10.1530/RAF-20-0037
- Balercia G, Regoli F, Armeni T, Koverech A, Mantero F, Boscaro M. Placebo-controlled double-blind randomized trial on the use of L-carnitine, L-acetylcarnitine, or combined L-carnitine and L-acetylcarnitine in men with idiopathic asthenozoospermia. Fertil Steril. 2005;84(3):662-671. doi:10.1016/j.fertnstert.2005.03.064
- Lenzi A, Sgrò P, Salacone P, et al. A placebo-controlled double-blind randomized trial of the use of combined L-carnitine and L-acetyl-carnitine treatment in men with asthenozoospermia. Fertil Steril. 2004;81(6):1578-1584. doi:10.1016/j.fertnstert.2003.10.034
- Tang WH, Hazen SL. The contributory role of gut microbiota in cardiovascular disease. J Clin Invest. 2014;124(10):4204-4211. doi:10.1172/JCI72331
- Koeth RA, Lam-Galvez BR, Kirsop J, et al. L-Carnitine in omnivorous diets induces an atherogenic gut microbial pathway in humans. J Clin Invest. 2019;129(1):373-387. doi:10.1172/JCI94601
- Zeisel SH, da Costa KA. Choline: an essential nutrient for public health. Nutr Rev. 2009;67(11):615-623. doi:10.1111/j.1753-4887.2009.00246.x
- Fischer LM, daCosta KA, Kwock L, et al. Sex and menopausal status influence human dietary requirements for the nutrient choline. Am J Clin Nutr. 2007;85(5):1275-1285. doi:10.1093/ajcn/85.5.1275
- Unfer V, Facchinetti F, Orrù B, Giordani B, Nestler J. Myo-inositol effects in women with PCOS: a meta-analysis of randomized controlled trials. Endocr Connect. 2017;6(8):647-658. doi:10.1530/EC-17-0243
- Best CH, Huntsman ME. The effects of the components of lecithine upon deposition of fat in the liver. J Physiol. 1932;75(4):405-412. doi:10.1113/jphysiol.1932.sp002899
- Brass EP. Supplemental carnitine and exercise. Am J Clin Nutr. 2000;72(2 Suppl):618S-623S. doi:10.1093/ajcn/72.2.618S
- Bremer J. Carnitine - metabolism and functions. Physiol Rev. 1983;63(4):1420-1480. doi:10.1152/physrev.1983.63.4.1420
- Malaguarnera M. Carnitine derivatives: clinical usefulness. Curr Opin Gastroenterol. 2012;28(2):166-176. doi:10.1097/MOG.0b013e3283505a3b
- Hoppel C. The role of carnitine in normal and altered fatty acid metabolism. Am J Kidney Dis. 2003;41(4 Suppl 4):S4-S12. doi:10.1016/S0272-6386(03)00112-4
- Hiatt WR, Regensteiner JG, Creager MA, et al. Propionyl-L-carnitine improves exercise performance and functional status in patients with claudication. Am J Med. 2001;110(8):616-622. doi:10.1016/S0002-9343(01)00707-2
- Bene J, Hadzsiev K, Melegh B. Role of carnitine and its derivatives in the development and management of type 2 diabetes. Nutr Diabetes. 2018;8(1):8. doi:10.1038/s41387-018-0017-1
- Veronese N, Stubbs B, Solmi M, et al. Acetyl-L-carnitine supplementation and the treatment of depressive symptoms: a systematic review and meta-analysis. Psychosom Med. 2018;80(2):154-159. doi:10.1097/PSY.0000000000000537
- Cruciani RA, Dvorkin E, Homel P, et al. Safety, tolerability and symptom outcomes associated with L-carnitine supplementation in patients with cancer, fatigue, and carnitine deficiency: a phase I/II study. J Pain Symptom Manage. 2006;32(6):551-559. doi:10.1016/j.jpainsymman.2006.09.001
- Samimi M, Jamilian M, Ebrahimi FA, Rahimi M, Tajbakhsh B, Asemi Z. Oral carnitine supplementation reduces body weight and insulin resistance in women with polycystic ovary syndrome: a randomized, double-blind, placebo-controlled trial. Clin Endocrinol (Oxf). 2016;84(6):851-857. doi:10.1111/cen.13003