Executive Summary
Figure 1: MOTS-c is a mitochondrial-derived peptide that functions as an exercise mimetic and metabolic regulator with implications for aging research.
Key Takeaways
- Figure 1: MOTS-c is a mitochondrial-derived peptide that functions as an exercise mimetic and metabolic regulator with implications for aging research.
- MOTS-c (mitochondrial open reading frame of the 12S rRNA type-c) is a 16-amino-acid peptide encoded within the mitochondrial genome that has emerged as one of the most promising metabolic regulators discovered in the past decade.
- The core mechanism through which MOTS-c exerts its effects centers on the activation of AMP-activated protein kinase (AMPK), the cell's master energy sensor.
- MOTS-c achieves this by inhibiting the folate-methionine cycle and its directly tethered de novo purine biosynthesis pathway.
- This inhibition causes a buildup of AICAR (5-aminoimidazole-4-carboxamide ribonucleotide), a well-characterized endogenous AMPK activator.
MOTS-c (mitochondrial open reading frame of the 12S rRNA type-c) is a 16-amino-acid peptide encoded within the mitochondrial genome that has emerged as one of the most promising metabolic regulators discovered in the past decade. Unlike most signaling peptides encoded in nuclear DNA, MOTS-c originates from a short open reading frame within the mitochondrial 12S rRNA gene, making it part of a small but growing family of mitochondrial-derived peptides (MDPs) that include humanin and the small humanin-like peptides (SHLPs). First identified in 2015 by Changhan Lee and colleagues at the University of Southern California, MOTS-c has since attracted intense research interest for its ability to mimic the metabolic benefits of physical exercise, regulate glucose homeostasis, and potentially slow aspects of biological aging.
The core mechanism through which MOTS-c exerts its effects centers on the activation of AMP-activated protein kinase (AMPK), the cell's master energy sensor. MOTS-c achieves this by inhibiting the folate-methionine cycle and its directly tethered de novo purine biosynthesis pathway. This inhibition causes a buildup of AICAR (5-aminoimidazole-4-carboxamide ribonucleotide), a well-characterized endogenous AMPK activator. The downstream consequences are far-reaching: improved insulin sensitivity in skeletal muscle, enhanced glucose uptake via GLUT4 translocation, increased fatty acid oxidation, and broad metabolic reprogramming that mirrors many of the cellular adaptations produced by regular aerobic exercise.
Preclinical research has produced compelling findings. In diet-induced obesity mouse models, MOTS-c treatment prevented insulin resistance and reduced weight gain. In aged mice (22 months old, roughly equivalent to a 70-year-old human), MOTS-c injections doubled running capacity on treadmill tests and improved balance on rotating rod assessments. A 2021 study published in Nature Communications demonstrated that late-life MOTS-c treatment improved physical performance in elderly mice and contributed to a 6.4% increase in median lifespan, suggesting the peptide can enhance both healthspan and lifespan.
Human observational data adds context to these animal findings. Circulating MOTS-c levels decline with age - plasma concentrations in individuals aged 70 to 81 are approximately 21% lower than those measured in 18- to 30-year-olds. People with type 2 diabetes consistently show lower circulating MOTS-c compared to healthy controls, and in obese male children and adolescents, MOTS-c levels are reduced and correlate inversely with markers of insulin resistance. Exercise itself appears to boost endogenous MOTS-c production, with studies showing increased MOTS-c expression in skeletal muscle and circulation following physical activity.
Beyond metabolism, emerging research points to MOTS-c's potential roles in bone health (promoting osteoblast activity while inhibiting osteoclast formation), cardiovascular protection, and even cancer biology. A 2024 study in Advanced Science found that MOTS-c levels are reduced in ovarian cancer patients and that exogenous MOTS-c inhibits cancer cell proliferation and migration. These findings suggest the peptide's influence extends well beyond its initial characterization as a metabolic regulator.
For practitioners and researchers in the peptide therapy space, MOTS-c represents a fundamentally different category of compound. It is not a growth hormone secretagogue like CJC-1295/Ipamorelin, nor a tissue repair peptide like BPC-157. Instead, it functions as a mitochondrial signal molecule - an encoded message from the organelle responsible for cellular energy production that tells the rest of the cell to optimize its metabolic machinery. This positions MOTS-c at the intersection of metabolic medicine, exercise science, and longevity research, making it one of the most intellectually interesting and therapeutically promising peptides currently under investigation.
This report provides an evidence-based examination of MOTS-c across its discovery history, molecular mechanisms, preclinical and human data, practical dosing considerations, and safety profile. All claims are grounded in peer-reviewed research, with citations to primary literature throughout. Whether you are a clinician evaluating MOTS-c for metabolic patients, a researcher studying mitochondrial biology, or an informed individual exploring peptide options through providers like FormBlends, this analysis aims to give you the complete scientific picture.
Discovery of Mitochondrial-Derived Peptides
Figure 2: Timeline of mitochondrial-derived peptide discoveries, from humanin (2001) through SHLPs (2013) to MOTS-c (2015).
The Fundamental change: Mitochondria as Signaling Organelles
For most of the 20th century, mitochondria were understood primarily as the cell's power plants - organelles that converted nutrients into ATP through oxidative phosphorylation. The mitochondrial genome, a compact circular DNA molecule of approximately 16,569 base pairs, was thought to encode only 13 proteins (all components of the electron transport chain), 22 transfer RNAs, and 2 ribosomal RNAs. The prevailing view held that mitochondrial DNA was too small and too functionally specialized to harbor additional protein-coding genes of biological significance.
This understanding began to shift in 2001 with the discovery of humanin, a 24-amino-acid peptide encoded within the 16S ribosomal RNA gene of mitochondrial DNA. Hashimoto and colleagues identified humanin while screening for genes that could protect neurons from amyloid-beta toxicity in Alzheimer's disease models. The finding was initially met with skepticism - how could a ribosomal RNA gene encode a functional peptide? But subsequent research confirmed that humanin was indeed translated from mitochondrial DNA, secreted from cells, and capable of exerting protective effects against cellular stress, apoptosis, and metabolic dysfunction.
Humanin's discovery cracked open a new field of mitochondrial biology. If one short open reading frame (sORF) hidden within a ribosomal RNA gene could produce a biologically active peptide, others might exist as well. Researchers began systematically scanning the mitochondrial genome for additional sORFs, using computational approaches to identify sequences with the hallmarks of protein-coding potential: start codons, reasonable reading frame lengths, and evolutionary conservation across species.
The SHLP Family: Expanding the Mitochondrial Peptidome
In 2013, Cobb and colleagues at the University of Southern California's Leonard Davis School of Gerontology identified six additional peptides encoded within the 16S rRNA gene, the same mitochondrial gene that harbors humanin. These were named the small humanin-like peptides (SHLP1 through SHLP6), ranging from 24 to 38 amino acids in length. Each SHLP showed distinct biological activities despite originating from the same gene region.
SHLP2 and SHLP3 enhanced cell survival and protected against apoptotic stress. SHLP4 promoted cell proliferation. SHLP6, intriguingly, induced apoptosis rather than preventing it. Circulating levels of SHLP2 declined with age, mirroring the pattern seen with humanin and suggesting a broader pattern of age-related decline in mitochondrial-derived peptide signaling. Evolutionary analysis later confirmed that humanin and SHLP6 are subject to natural selection across vertebrate species, reinforcing the idea that these peptides serve genuine biological functions rather than representing translational noise.
The SHLP discoveries established a clear principle: the mitochondrial genome contains multiple functional peptide-coding sequences embedded within genes previously thought to serve only structural roles in mitochondrial ribosome assembly. This set the stage for searching the other major ribosomal RNA gene in the mitochondrial genome - the 12S rRNA.
MOTS-c: Discovery and Initial Characterization (2015)
The breakthrough came in March 2015, when Changhan Lee, Jennifer Zeng, Brian Drew, and colleagues published their landmark paper in Cell Metabolism identifying MOTS-c. Using bioinformatic analysis of the mitochondrial 12S rRNA gene (MT-RNR1), they found a short open reading frame encoding a 16-amino-acid peptide with the sequence MRWQEMGYIFYPRKLR. They named it MOTS-c, for "mitochondrial open reading frame of the 12S rRNA type-c."
Several features of MOTS-c immediately distinguished it from previously identified MDPs. First, it was encoded in the 12S rRNA gene rather than the 16S rRNA gene that housed humanin and the SHLPs. Second, its amino acid sequence was highly conserved across mammalian species, suggesting strong evolutionary pressure to maintain its function. Third, and most striking, MOTS-c appeared to target skeletal muscle specifically, with potent effects on glucose metabolism and insulin sensitivity that the researchers had not seen with other MDPs.
In their initial characterization, Lee et al. demonstrated that MOTS-c treatment in cell culture activated AMPK, increased glucose uptake, and enhanced mitochondrial function. When they injected MOTS-c into mice fed a high-fat diet, the peptide prevented obesity and insulin resistance. Mice on the same high-fat diet but receiving MOTS-c gained significantly less weight and maintained normal glucose tolerance compared to untreated controls. These effects were comparable to what one might expect from a regular exercise program - a finding that would earn MOTS-c its reputation as an "exercise mimetic."
Nuclear Translocation: An Unexpected Finding (2018)
Perhaps the most surprising discovery about MOTS-c came in 2018, when the same research group at USC showed that MOTS-c can translocate from the cytoplasm to the cell nucleus under conditions of metabolic stress. This finding, published in Cell Metabolism, was remarkable because it suggested a form of mito-nuclear communication that had not been previously described for any mitochondrial-derived peptide.
Under normal conditions, MOTS-c circulates in the cytoplasm and bloodstream. But when cells face metabolic challenges - glucose deprivation, oxidative stress, or serum starvation - MOTS-c migrates to the nucleus, where it interacts with transcription factors and regulates the expression of hundreds of genes involved in stress response and metabolic adaptation. This nuclear translocation depends on AMPK activation and involves a specific structural motif within the MOTS-c sequence.
Structural analysis revealed two key domains within the 16-amino-acid peptide. A hydrophobic core consisting of residues 8 through 11 (YIFY) is essential for nuclear entry - mutating these residues to alanines blocks translocation entirely. A cluster of basic residues at positions 13 through 16 (RKLR) resembles a classical nuclear localization signal, though substituting these with alanines does not prevent nuclear entry, suggesting the hydrophobic domain mediates protein-protein interactions necessary for import.
The nuclear translocation discovery fundamentally changed how researchers understood MOTS-c's mechanism. It was no longer simply a circulating metabolic hormone; it was a stress-responsive signal molecule that could directly reprogram gene expression. This placed MOTS-c in a unique category among known peptide hormones and opened new avenues for understanding how mitochondria communicate with the nuclear genome to coordinate cellular responses to metabolic stress.
The MDP Family in Context
Today, the known family of mitochondrial-derived peptides includes humanin, SHLP1 through SHLP6, and MOTS-c - nine peptides in total. Researchers suspect additional MDPs remain undiscovered, as computational scans of the mitochondrial genome have identified numerous candidate sORFs that have not yet been experimentally validated. The field remains young, with the first MDP discovered barely two decades ago and MOTS-c identified less than a decade ago.
| Peptide | Gene Location | Length | Year Discovered | Primary Functions |
|---|---|---|---|---|
| Humanin | 16S rRNA (MT-RNR2) | 24 amino acids | 2001 | Neuroprotection, anti-apoptosis, insulin sensitization |
| SHLP1 | 16S rRNA (MT-RNR2) | 26 amino acids | 2013 | Mitochondrial metabolism |
| SHLP2 | 16S rRNA (MT-RNR2) | 26 amino acids | 2013 | Cell survival, insulin sensitization, chaperone activity |
| SHLP3 | 16S rRNA (MT-RNR2) | 38 amino acids | 2013 | Cell survival, anti-apoptosis |
| SHLP4 | 16S rRNA (MT-RNR2) | 27 amino acids | 2013 | Cell proliferation |
| SHLP5 | 16S rRNA (MT-RNR2) | 24 amino acids | 2013 | Mitochondrial metabolism |
| SHLP6 | 16S rRNA (MT-RNR2) | 24 amino acids | 2013 | Pro-apoptotic signaling |
| MOTS-c | 12S rRNA (MT-RNR1) | 16 amino acids | 2015 | Exercise mimetic, AMPK activation, glucose regulation |
What makes the MDP field particularly exciting is the realization that mitochondria are not passive energy generators but active signaling organelles. They encode and secrete peptides that regulate whole-body metabolism, influence aging, and protect against disease. For researchers working with compounds like SS-31 (which targets mitochondrial cardiolipin) or humanin, MOTS-c adds another dimension to the growing understanding of mitochondrial medicine. The peptide research hub provides additional context on how these compounds fit within the broader peptide landscape.
From Discovery to Therapeutic Exploration
The pace of MOTS-c research has accelerated considerably since 2015. A PubMed search reveals fewer than 10 publications on MOTS-c in 2015 and 2016 combined, growing to over 40 publications per year by 2023 and 2024. Research groups across the United States, China, South Korea, Japan, and Europe are now studying MOTS-c across diverse disease contexts including diabetes, obesity, cardiovascular disease, cancer, osteoporosis, and neurodegeneration.
CohBar, Inc. (now known as Yumanity Therapeutics before its closure) developed a MOTS-c analog called CB4211 that entered Phase 1a clinical testing for non-alcoholic steatohepatitis (NASH) and obesity. While the parent company ultimately shifted focus away from MDP-based therapeutics, the CB4211 trial provided the first human safety data for a MOTS-c-related compound and demonstrated proof of concept for translating MDP biology into clinical medicine.
The transition from basic science discovery to therapeutic development is still in its early stages for MOTS-c. But the biological rationale is strong: a naturally occurring peptide that activates the same metabolic pathways as exercise, with demonstrated benefits in preclinical models of obesity, diabetes, and aging. The challenge now lies in establishing proper human dosing, confirming long-term safety, and determining which patient populations stand to benefit most from MOTS-c supplementation.
MOTS-c Structure & Mitochondrial Origin
Figure 3: Structural features of MOTS-c, highlighting the hydrophobic core (YIFY) and basic residue cluster (RKLR) essential for its biological activity.
Amino Acid Sequence and Functional Domains
MOTS-c is a 16-amino-acid peptide with the primary sequence methionine-arginine-tryptophan-glutamine-glutamic acid-methionine-glycine-tyrosine-isoleucine-phenylalanine-tyrosine-proline-arginine-lysine-leucine-arginine (MRWQEMGYIFYPRKLR). Despite its small size - one of the shortest bioactive peptides known to function as a systemic hormone - MOTS-c packs considerable functional complexity into its compact structure.
Two structural domains within the peptide have been identified as critical for its biological activity. The first is a hydrophobic core spanning residues 8 through 11, consisting of tyrosine-isoleucine-phenylalanine-tyrosine (YIFY). This aromatic-rich motif mediates protein-protein interactions that are essential for MOTS-c's nuclear translocation under metabolic stress. When researchers mutated these four residues to alanines, the modified peptide lost its ability to enter the nucleus entirely, even though it retained some cytoplasmic activity. The YIFY domain likely serves as a recognition element for nuclear import machinery or for chaperone proteins that facilitate MOTS-c's transport across the nuclear envelope.
The second key domain is a cluster of basic (positively charged) amino acids at positions 13 through 16: arginine-lysine-leucine-arginine (RKLR). This motif resembles classical nuclear localization sequences (NLS) found in many nuclear-targeted proteins. However, experiments showed that substituting RKLR with four alanines did not prevent nuclear entry, which was unexpected. The basic residue cluster may instead contribute to DNA binding or interactions with nuclear transcription factors once MOTS-c reaches the nucleus, rather than serving as the primary nuclear import signal.
Between these two domains, a proline residue at position 12 introduces a structural kink that may separate the functional regions and influence the peptide's three-dimensional conformation. The N-terminal portion of the peptide (residues 1 through 7, MRWQEMG) contains the start methionine and a mix of charged, polar, and aromatic residues that likely contribute to the peptide's solubility and receptor interactions in the extracellular space.
Mitochondrial Genomic Origin
MOTS-c is encoded within the mitochondrial 12S ribosomal RNA gene (MT-RNR1), located on the heavy strand of the mitochondrial genome. The human mitochondrial genome is a 16,569-base-pair circular DNA molecule that, until the discovery of MDPs, was believed to encode only 37 genes: 13 messenger RNAs for electron transport chain subunits, 22 transfer RNAs, and 2 ribosomal RNAs (12S and 16S).
The MOTS-c coding sequence represents a short open reading frame (sORF) embedded within the 12S rRNA gene. This arrangement is unusual from a molecular biology perspective. In most organisms, ribosomal RNA genes are transcribed as rRNA, not translated into protein. The existence of a functional peptide-coding sORF within an rRNA gene suggests that mitochondrial genetic information is more densely packed and multi-functional than previously appreciated.
The MOTS-c sORF uses the standard genetic code rather than the mitochondrial genetic code, which differs at several codons. This raises an interesting question about where MOTS-c translation actually occurs. Some evidence suggests that the MOTS-c mRNA may be exported from mitochondria to the cytoplasm for translation on cytoplasmic ribosomes, which use the standard genetic code. This would represent yet another unconventional aspect of MOTS-c biology - a mitochondrial-encoded message translated by the cell's cytoplasmic protein synthesis machinery.
Evolutionary Conservation
One of the strongest arguments for MOTS-c's biological importance is its evolutionary conservation. The MOTS-c amino acid sequence is highly conserved across mammalian species, with the human sequence showing strong homology to sequences found in mice, rats, primates, and other mammals. This degree of conservation over tens of millions of years of evolution implies that MOTS-c serves a function important enough to resist mutational drift.
Conservation is particularly notable within the YIFY hydrophobic core and RKLR basic cluster - the two domains essential for nuclear translocation and transcriptional regulation. Mutations in these regions would presumably be selected against because they would impair MOTS-c's ability to coordinate mito-nuclear communication, a function that appears critical for metabolic adaptation and stress response.
Some population-level variation in the MOTS-c sequence has been identified in humans. A polymorphism in the MT-RNR1 gene affects the MOTS-c coding region in certain populations, and one study has linked specific MOTS-c variants to exceptional longevity in Japanese centenarians. This finding, while preliminary, supports the hypothesis that MOTS-c plays a role in human aging and that genetic variation in its sequence can influence lifespan. Researchers studying longevity peptides like Epithalon and FOXO4-DRI have noted MOTS-c as a complementary target in anti-aging research.
Biosynthesis, Secretion, and Tissue Distribution
MOTS-c is produced in cells throughout the body, as mitochondria are present in virtually all nucleated human cells. However, the level of MOTS-c expression varies across tissues. Skeletal muscle is a major site of MOTS-c production, consistent with the peptide's primary metabolic effects on muscle glucose uptake and energy metabolism. Other tissues with significant MOTS-c expression include the liver, brain, kidney, and adipose tissue.
After synthesis, MOTS-c is secreted into the bloodstream, where it circulates as an endocrine factor capable of acting on distant tissues. Plasma MOTS-c levels in healthy young adults range approximately from 400 to 600 ng/mL, though measurements vary across studies depending on the assay methodology used. The peptide's half-life in circulation has not been precisely established in humans, but preclinical data suggest it is relatively short, consistent with other small peptides. This short half-life is one of the practical challenges for therapeutic development, as it may necessitate frequent dosing or the development of stabilized analogs.
An important finding regarding tissue distribution came from a 2020 study showing that skeletal muscle MOTS-c expression actually increases with age in some contexts, even as circulating levels decline. Older men (70 to 81 years) and middle-aged men (45 to 55 years) showed approximately 1.5-fold higher skeletal muscle MOTS-c expression compared to young men (18 to 30 years). This paradoxical finding may represent a compensatory upregulation - aging muscle produces more MOTS-c locally in an attempt to maintain metabolic function, even though systemic levels are falling. The disconnect between tissue expression and circulating levels highlights the complexity of MOTS-c biology and suggests that both local and systemic MOTS-c signaling matter for metabolic health.
Post-Translational Processing and Stability
As a small peptide, MOTS-c faces the same stability challenges that affect other therapeutic peptides in the research pipeline. It is susceptible to degradation by peptidases in blood and tissues, which limits its bioavailability after injection. The peptide does not appear to undergo significant post-translational modifications (such as glycosylation or phosphorylation) based on current evidence, meaning its activity depends entirely on the primary amino acid sequence.
These stability concerns have driven interest in developing MOTS-c analogs with improved pharmacokinetic properties. The CB4211 analog developed by CohBar incorporated structural modifications designed to resist enzymatic degradation while preserving the core biological activity. For researchers and clinicians working with native MOTS-c, proper storage, reconstitution, and handling are important practical considerations. The MOTS-c product page at FormBlends provides guidance on these handling requirements.
AMPK Activation & Metabolic Effects
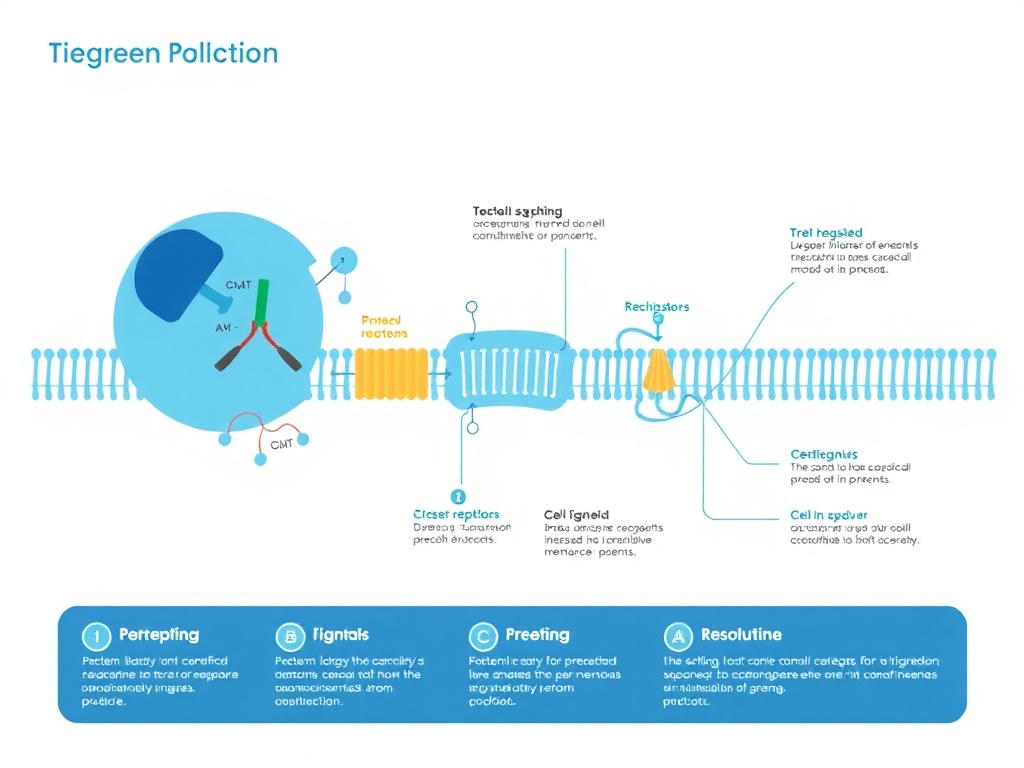
Figure 4: The MOTS-c signaling cascade from folate cycle inhibition through AICAR-mediated AMPK activation to metabolic reprogramming.
The Folate-AICAR-AMPK Pathway
MOTS-c's primary mechanism of action is the activation of AMPK through a unique upstream pathway that distinguishes it from other AMPK activators. While drugs like metformin activate AMPK primarily through inhibition of mitochondrial complex I (which increases the AMP/ATP ratio), MOTS-c takes an entirely different route. It targets the folate-methionine cycle and its directly connected de novo purine biosynthesis pathway, creating a metabolic bottleneck that leads to AMPK activation through AICAR accumulation.
The pathway works as follows. MOTS-c inhibits the folate cycle at the level of 5-methyltetrahydrofolate (5Me-THF), blocking the conversion of tetrahydrofolate intermediates needed for one-carbon metabolism. This one-carbon pool is essential for de novo purine synthesis, the biochemical pathway cells use to build the adenine and guanine nucleotides needed for DNA, RNA, and energy carrier molecules like ATP. When MOTS-c blocks the folate-dependent step in this pathway, the purine synthesis intermediate AICAR accumulates to extraordinary levels - up to 20-fold higher than baseline in MOTS-c-treated cells compared to controls.
AICAR (5-aminoimidazole-4-carboxamide ribonucleotide) is a well-characterized AMPK activator that has been studied for decades as a pharmacological tool. It mimics AMP, binding to the gamma subunit of AMPK and triggering the conformational change that activates the kinase. The fact that MOTS-c generates massive AICAR accumulation endogenously explains its potent AMPK-activating properties without the off-target effects associated with direct AMP analogs or mitochondrial poisons.
This mechanism was established through unbiased global metabolomics combined with gene microarray analysis. Lee et al. treated cells with MOTS-c and measured changes in hundreds of metabolites simultaneously, identifying the folate-methionine cycle as the primary metabolic target. The specificity was notable - MOTS-c did not broadly suppress cellular metabolism but rather created a targeted disruption that channeled metabolic flux in specific directions.
AMPK: The Master Metabolic Switch
Understanding why AMPK activation matters requires appreciating the enzyme's central role in cellular energy management. AMPK (5'-AMP-activated protein kinase) is often described as the cell's fuel gauge. When cellular energy levels drop - reflected by rising AMP-to-ATP ratios - AMPK activates and initiates a coordinated program of metabolic changes designed to restore energy balance.
AMPK activation by MOTS-c triggers several downstream effects that collectively improve metabolic function. In skeletal muscle, AMPK promotes the translocation of GLUT4 glucose transporters to the cell surface, allowing muscle cells to take up glucose from the blood without requiring insulin signaling. This insulin-independent glucose uptake is one of the key mechanisms through which exercise improves blood sugar control, and MOTS-c appears to activate the same pathway.
AMPK also stimulates fatty acid oxidation by phosphorylating and inhibiting acetyl-CoA carboxylase (ACC), which reduces malonyl-CoA levels and relieves the inhibition of carnitine palmitoyltransferase 1 (CPT1). This allows long-chain fatty acids to enter mitochondria for beta-oxidation, increasing fat burning. In MOTS-c-treated mice, increased beta-oxidation prevented fat accumulation in diet-induced obesity models, mirroring the fat-reducing effects of regular exercise.
AMPK activation further inhibits anabolic processes that consume energy. It suppresses mTORC1 signaling (a growth-promoting pathway), reduces lipogenesis (fat synthesis), and decreases gluconeogenesis in the liver. These effects collectively shift cells from a "growth and storage" mode to a "burn and conserve" mode - exactly the metabolic reprogramming that occurs during physical activity. Researchers interested in mTOR pathway modulation may also find relevance in how 5-Amino-1MQ approaches metabolic regulation through complementary mechanisms.
Glucose Metabolism and Metabolic Routing
MOTS-c's effects on glucose metabolism extend beyond simple increases in uptake. Metabolomic analysis revealed that glucose taken up by MOTS-c-treated cells is preferentially routed through the pentose phosphate pathway (PPP) rather than glycolysis. The PPP generates ribose-5-phosphate, a sugar needed for nucleotide synthesis, as well as NADPH, a reducing equivalent used for biosynthetic reactions and antioxidant defense.
This metabolic routing makes biological sense in the context of MOTS-c's mechanism. By blocking de novo purine synthesis at the folate-dependent step, MOTS-c creates a demand for alternative carbon sources for nucleotide production. Directing glucose through the PPP provides those carbon sources while simultaneously generating NADPH to support cellular redox balance. The result is a coordinated metabolic reprogramming that improves glucose disposal, enhances antioxidant capacity, and maintains nucleotide homeostasis simultaneously.
MOTS-c treatment has also been shown to increase cellular NAD+ levels. NAD+ is a critical coenzyme in energy metabolism and serves as a substrate for sirtuins, a family of deacetylase enzymes involved in longevity signaling. The NAD+ increase may contribute to AMPK activation through SIRT1 (which deacetylates and activates the AMPK kinase LKB1) and provides another link between MOTS-c and the molecular pathways associated with caloric restriction and exercise. For individuals interested in NAD+ biology, NAD+ supplementation represents a complementary approach to supporting cellular energy metabolism.
Effects on Lipid Metabolism
MOTS-c's impact on lipid metabolism parallels the fat-reducing effects of aerobic exercise. In diet-induced obesity mouse models, MOTS-c treatment significantly reduced body weight gain, decreased adipose tissue mass, and improved the lipid profile. Specific effects included reduced circulating triglycerides, lower free fatty acid levels, and decreased hepatic fat accumulation - all hallmarks of improved metabolic health.
The molecular basis for these lipid effects traces back to AMPK activation. AMPK phosphorylates and inhibits several lipogenic enzymes, including ACC (reducing malonyl-CoA and promoting fatty acid oxidation), HMG-CoA reductase (reducing cholesterol synthesis), and SREBP-1c (a transcription factor that drives expression of lipogenic genes). The combined effect is a shift from fat storage to fat utilization, reducing the lipotoxicity that contributes to insulin resistance, fatty liver disease, and cardiovascular risk.
These lipid-modifying properties position MOTS-c as a potential complement to other metabolic interventions. While semaglutide and tirzepatide address obesity primarily through appetite suppression via GLP-1 receptor agonism, MOTS-c works at the cellular level to enhance the metabolic machinery that processes and burns fat. The two approaches target different aspects of the obesity problem and could theoretically complement each other, though no clinical studies have tested this combination.
Mitochondrial Function Enhancement
Beyond its effects on cytoplasmic metabolic pathways, MOTS-c also influences mitochondrial function directly. A 2025 study published in Frontiers in Physiology demonstrated that MOTS-c treatment restored mitochondrial respiration in cardiac tissue from type 2 diabetic rats. The treatment improved oxygen consumption rates and enhanced the efficiency of the electron transport chain, suggesting that MOTS-c can rescue mitochondrial dysfunction in metabolically stressed tissues.
This effect on mitochondrial respiration creates a positive feedback loop. MOTS-c, produced by mitochondria, improves mitochondrial function, which in turn enhances cellular energy production and metabolic capacity. This feedback mechanism may explain why MOTS-c's effects are particularly pronounced in metabolically compromised states - such as obesity, diabetes, and aging - where mitochondrial dysfunction is a contributing factor.
The relationship between MOTS-c and mitochondrial health has implications for other mitochondria-targeted therapies. Compounds like SS-31 (elamipretide), which stabilizes mitochondrial membranes, and NAD+, which supports mitochondrial energy metabolism, may work through complementary mechanisms that could enhance MOTS-c's effects. The biohacking hub explores these and other strategies for optimizing mitochondrial function.
Comparison with Other AMPK Activators
MOTS-c is not the only compound that activates AMPK, and comparing it to other AMPK activators helps clarify its unique therapeutic niche.
| AMPK Activator | Mechanism | Route | Key Advantages | Key Limitations |
|---|---|---|---|---|
| MOTS-c | Folate cycle inhibition, AICAR accumulation | Subcutaneous injection | Endogenous peptide, exercise mimetic, nuclear gene regulation | Short half-life, limited human data |
| Metformin | Complex I inhibition, increased AMP/ATP ratio | Oral | Decades of safety data, inexpensive | GI side effects, lactic acidosis risk |
| AICAR (drug) | Direct AMPK activation (AMP mimetic) | IV infusion | Well-studied research tool | Poor bioavailability, cardiac effects |
| Exercise | ATP depletion, calcium signaling, ROS | Physical activity | Broad systemic benefits, no drug costs | Requires physical capacity, adherence challenges |
| Berberine | Complex I inhibition (similar to metformin) | Oral | Natural compound, readily available | GI effects, drug interactions |
MOTS-c's advantage over synthetic AMPK activators lies in its identity as an endogenous signal molecule. The body already produces and uses MOTS-c for metabolic regulation, suggesting that supplemental MOTS-c may work within existing physiological frameworks rather than imposing artificial metabolic changes. Additionally, MOTS-c's ability to translocate to the nucleus and regulate gene expression gives it a dimension of activity that simple AMPK activators lack. These unique properties have generated enthusiasm in the peptide research community, as reflected in the growing body of literature tracked by the FormBlends science page.
Exercise Mimetic Properties
Figure 5: MOTS-c mimics key metabolic adaptations of physical exercise through AMPK-dependent pathways in skeletal muscle.
What Makes MOTS-c an Exercise Mimetic?
An exercise mimetic is a compound that reproduces some of the molecular and physiological adaptations normally triggered by physical exercise, without requiring actual physical activity. MOTS-c qualifies as an exercise mimetic because it activates many of the same signaling pathways and metabolic responses that exercise triggers in skeletal muscle. These include AMPK activation, enhanced glucose uptake, increased fatty acid oxidation, improved mitochondrial function, and upregulation of genes involved in metabolic adaptation and stress resistance.
The concept of exercise mimetics has attracted enormous interest in medicine and aging research. Regular physical exercise is arguably the single most effective intervention for preventing chronic disease, maintaining metabolic health, and slowing biological aging. But many people - particularly the elderly, the physically disabled, or those with severe obesity - cannot exercise at the intensity or duration needed to achieve these benefits. A compound that could replicate even a fraction of exercise's metabolic effects would have profound clinical implications.
MOTS-c is not the first compound to be called an exercise mimetic. AICAR itself was characterized as an exercise mimetic in a landmark 2008 study by Narkar et al. in Cell, which showed that AICAR treatment improved running endurance in sedentary mice. But MOTS-c has a key advantage: it is an endogenous molecule that the body naturally produces in response to exercise. When you run, swim, or cycle, your muscles and other tissues increase MOTS-c production. Supplementing with exogenous MOTS-c may therefore amplify a natural physiological response rather than introducing a foreign pharmacological stimulus.
Exercise-Induced MOTS-c Expression
Multiple studies have documented that physical exercise increases endogenous MOTS-c levels. Reynolds et al. (2021) showed that exercise induces MOTS-c expression in skeletal muscle and increases its presence in circulation. This exercise-induced upregulation occurs across different exercise modalities and intensities, though the specific dose-response relationship between exercise parameters and MOTS-c production is still being characterized.
A study examining professional athletes found elevated serum MOTS-c and humanin levels compared to sedentary controls, and chronic endurance exercise training was associated with sustained increases in circulating MDP levels. This observation supports a model in which regular exercise trains the mitochondrial peptide system much as it trains the cardiovascular and musculoskeletal systems - through repeated stimulus and adaptation.
A 2021 study in Scientific Reports specifically examined the effect of aerobic and resistance exercise on MOTS-c levels in breast cancer survivors, finding that both exercise modalities influenced circulating MOTS-c, though the response varied by ethnicity. Hispanic breast cancer survivors showed different MOTS-c responses to exercise compared to non-Hispanic white survivors, highlighting the importance of considering genetic and demographic factors in exercise-peptide interactions.
The Reynolds 2021 Study: Doubling Physical Capacity in Aged Mice
The most striking demonstration of MOTS-c's exercise mimetic properties came from a 2021 study by Joseph Reynolds and colleagues, published in Nature Communications. This study tested MOTS-c treatment across the mouse lifespan and produced results that captured significant attention in the aging research community.
The researchers tested MOTS-c injections in three age groups of mice: young (2 months), middle-aged (12 months), and old (22 months, roughly equivalent to a 70-year-old human). All three age groups were subjected to physical performance tests including the rotarod (which measures balance and coordination on a rotating cylinder) and treadmill running (which measures endurance capacity).
The results were dramatic. MOTS-c-treated mice at all ages performed significantly better than age-matched controls on both tests. But the most remarkable findings came from the oldest mice. Twenty-two-month-old mice receiving MOTS-c showed physical performance improvements that essentially doubled their running capacity compared to untreated old mice. Their performance levels approached those of much younger animals. On the rotarod, old MOTS-c-treated mice maintained balance significantly longer than untreated old mice, demonstrating improved neuromuscular coordination.
The study also examined the molecular basis for these improvements. MOTS-c treatment in old mice was associated with improved skeletal muscle mitochondrial function, reduced markers of cellular senescence, and changes in muscle gene expression that favored metabolic efficiency and stress resistance. The treated mice showed gene expression profiles that more closely resembled younger animals than untreated elderly mice.
Comparison with Exercise Adaptations
To understand the full scope of MOTS-c as an exercise mimetic, it's helpful to compare its effects directly with the adaptations produced by regular exercise.
| Adaptation | Exercise | MOTS-c | Status |
|---|---|---|---|
| AMPK activation in muscle | Yes | Yes | Confirmed in multiple models |
| Increased glucose uptake (GLUT4) | Yes | Yes | Confirmed via mitofusion-dependent mechanism |
| Enhanced fatty acid oxidation | Yes | Yes | Confirmed in DIO mice |
| Improved insulin sensitivity | Yes | Yes | Confirmed in aged and obese models |
| Mitochondrial biogenesis | Yes | Partial | MOTS-c improves function; biogenesis data limited |
| Improved endurance capacity | Yes | Yes | Confirmed in young, middle-aged, and old mice |
| Cardiovascular conditioning | Yes | Unknown | No direct cardiac exercise studies with MOTS-c |
| Muscle hypertrophy | Yes (resistance) | No evidence | MOTS-c appears metabolic, not anabolic |
| Bone density improvement | Yes (weight-bearing) | Yes | MOTS-c promotes osteoblast, inhibits osteoclast |
| Cognitive benefits | Yes | Unknown | Limited neurological data |
| Mood enhancement | Yes (endorphins) | No evidence | MOTS-c does not appear to affect mood pathways |
This comparison reveals that MOTS-c reproduces many, but not all, of exercise's metabolic benefits. It appears strongest in the areas of glucose metabolism, insulin sensitivity, and endurance capacity. However, it likely cannot replace the cardiovascular conditioning, muscle building, cognitive enhancement, and mood benefits that come from actual physical movement. For most individuals, MOTS-c is best understood as a potential supplement to - not a replacement for - regular exercise.
GLUT4 Translocation and Mitofusion
A 2021 study in Scientific Reports provided mechanistic detail on how MOTS-c promotes glucose uptake in muscle cells. The researchers found that MOTS-c-induced GLUT4 translocation to the cell surface requires mitofusion - the process by which mitochondria fuse together into larger, more interconnected networks. Mitofusion is a normal response to exercise and metabolic stress, and it improves mitochondrial efficiency by allowing the sharing of metabolites, proteins, and genetic material between individual mitochondria.
When the researchers inhibited mitofusion (by knocking down mitofusin-2), MOTS-c's ability to promote GLUT4 translocation was significantly reduced. This finding links MOTS-c's metabolic effects to mitochondrial dynamics and suggests that MOTS-c does not simply bypass mitochondrial function but rather enhances it through promoting mitochondrial networking. This is yet another parallel to exercise, which also promotes mitochondrial fusion and network formation in skeletal muscle.
Implications for Sedentary and Immobile Populations
The exercise mimetic properties of MOTS-c have particular relevance for populations that cannot exercise. Bedridden patients, individuals with severe osteoarthritis or mobility limitations, elderly persons with sarcopenia and frailty, and astronauts experiencing microgravity-induced metabolic changes could all potentially benefit from a compound that activates exercise-like metabolic pathways without requiring physical movement.
The Reynolds 2021 study's finding that MOTS-c improved physical function even in the oldest mice (those nearing end-of-life) suggests a potential application in geriatric medicine for combating frailty. Frailty, characterized by loss of muscle mass, reduced endurance, and metabolic decline, affects up to 15% of adults over 65 and is associated with increased falls, hospitalizations, and mortality. An exercise mimetic that could partially reverse these declines would address a major unmet medical need.
However, it is worth emphasizing that MOTS-c has not been tested for these applications in human clinical trials. The leap from improved treadmill performance in mice to clinical frailty intervention in humans is substantial. Researchers interested in supporting metabolic function through peptide interventions may also consider growth hormone secretagogues like sermorelin or tesamorelin, which address muscle wasting through different mechanisms. The GLP-1 research hub covers metabolic optimization from the incretin angle.
Glucose & Insulin Sensitivity Research
Figure 6: MOTS-c treatment reduces fasting glucose and improves insulin sensitivity across multiple preclinical models.
Preclinical Evidence for Glucose Regulation
MOTS-c's effects on glucose metabolism represent some of the most well-documented aspects of its biology, with data from multiple independent research groups confirming its insulin-sensitizing and glucose-lowering properties in animal models. The original 2015 Cell Metabolism paper by Lee et al. established the foundation: MOTS-c treatment prevented age-dependent and high-fat-diet-induced insulin resistance in mice and significantly improved glucose tolerance.
In the diet-induced obesity (DIO) model - where mice are fed a 60% fat diet to simulate metabolic syndrome - MOTS-c treatment produced striking improvements in glucose handling. Fasting glucose levels dropped from approximately 180 mg/dL in untreated DIO mice to around 120 mg/dL in MOTS-c-treated animals at the highest dose, a reduction comparable to what was seen in exercised mice (approximately 125 mg/dL). Lower doses of MOTS-c produced intermediate effects, with fasting glucose around 145 mg/dL, demonstrating a dose-dependent response.
Fasting Glucose in Diet-Induced Obesity Model
Data derived from preclinical DIO mouse models. MOTS-c treatment showed dose-dependent glucose reduction comparable to exercise.
Glucose tolerance tests (GTTs) provided additional evidence. When DIO mice were given a glucose challenge, MOTS-c-treated mice cleared the glucose load significantly faster than untreated controls, with blood glucose returning to baseline levels in a time course similar to lean, healthy mice. Insulin tolerance tests (ITTs) showed enhanced insulin responsiveness in MOTS-c-treated animals - less insulin was required to achieve the same degree of glucose lowering, indicating improved peripheral insulin sensitivity.
Insulin Sensitization Mechanisms
MOTS-c improves insulin sensitivity through multiple complementary mechanisms, which distinguishes it from single-target insulin-sensitizing drugs. The primary mechanism involves AMPK-mediated GLUT4 translocation in skeletal muscle. By activating AMPK, MOTS-c promotes the movement of GLUT4 glucose transporters from intracellular vesicles to the plasma membrane, allowing muscle cells to take up glucose independently of insulin signaling. This insulin-independent glucose uptake pathway is particularly valuable in insulin-resistant states where the canonical insulin signaling cascade (IRS-1, PI3K, Akt) is impaired.
A second mechanism involves the reduction of lipotoxicity. By promoting fatty acid oxidation and reducing intramyocellular lipid accumulation, MOTS-c removes one of the key drivers of skeletal muscle insulin resistance. Excess intracellular lipids (particularly diacylglycerols and ceramides) interfere with insulin signaling by activating protein kinase C isoforms that phosphorylate insulin receptor substrate 1 on inhibitory serine residues. MOTS-c's ability to clear these lipids through enhanced oxidation restores the normal insulin signaling cascade.
Third, MOTS-c's effects on NAD+ levels may contribute to insulin sensitization through sirtuin activation. SIRT1 in particular has been shown to deacetylate PTP1B and other negative regulators of insulin signaling, improving insulin receptor sensitivity. The increase in NAD+ produced by MOTS-c could potentiate this SIRT1-mediated insulin-sensitizing pathway.
Human Observational Data
While human interventional trials with MOTS-c are still limited, several observational studies have examined the relationship between circulating MOTS-c levels and metabolic health in humans.
Du et al. (2018) measured circulating MOTS-c in obese versus normal-weight male children and adolescents. MOTS-c levels were significantly lower in the obese group (472.61 plus or minus 22.83 ng/mL) compared to normal-weight controls (561.64 plus or minus 19.19 ng/mL, P less than 0.01). MOTS-c levels correlated inversely with HOMA-IR (homeostatic model assessment of insulin resistance), BMI z-score, and waist circumference, suggesting that lower MOTS-c is associated with worse metabolic health in youth. Interestingly, this association was seen only in males, not females, pointing to possible sex-dependent differences in MOTS-c biology.
In adults, the relationship between MOTS-c and obesity is more nuanced. A 2025 study found that plasma MOTS-c concentrations were actually elevated in obese adults compared to lean individuals, positively correlating with BMI and HOMA-IR. This finding appeared to contradict the pediatric data. However, researchers proposed that elevated MOTS-c in obese adults may represent a compensatory response - the body attempting to counteract metabolic dysfunction by increasing MOTS-c production, similar to the hyperinsulinemia seen in early insulin resistance where the pancreas produces more insulin to overcome reduced sensitivity.
In type 2 diabetes, the picture is clearer. Multiple studies consistently show that circulating MOTS-c levels are reduced in people with established type 2 diabetes compared to healthy controls. A 2024 systematic review and meta-analysis examining mitochondrial-derived peptide levels across metabolic conditions confirmed significantly lower MOTS-c in diabetic populations. This reduction may reflect both impaired mitochondrial function (leading to reduced MOTS-c production) and increased metabolic consumption of the peptide in diabetic states.
Pancreatic Beta Cell Protection
A 2025 study published in Experimental and Molecular Medicine added an important dimension to MOTS-c's metabolic profile by demonstrating that the peptide protects pancreatic beta cells from senescence. Pancreatic beta cells are the insulin-producing cells of the islets of Langerhans, and their progressive dysfunction and death is a hallmark of type 2 diabetes progression.
The study showed that MOTS-c prevented pancreatic islet cell senescence through regulation of AMPK and mTOR pathways. Senescent beta cells lose their ability to produce insulin in response to glucose and secrete inflammatory factors (the senescence-associated secretory phenotype, or SASP) that damage neighboring cells. By blocking senescence, MOTS-c helped maintain beta cell function and insulin secretory capacity, potentially delaying the progression from insulin resistance to overt diabetes.
This finding connects MOTS-c to the broader field of senolytic and senostatic therapy. While compounds like FOXO4-DRI work by inducing apoptosis in senescent cells (senolytic action), MOTS-c appears to work by preventing cells from becoming senescent in the first place (senostatic action). Both approaches aim to reduce the burden of cellular senescence that accumulates with aging and contributes to metabolic decline.
Comparison with Other Insulin-Sensitizing Agents
MOTS-c's insulin-sensitizing properties invite comparison with established diabetes medications and other metabolic peptides. The semaglutide and tirzepatide class of GLP-1 receptor agonists improve insulin sensitivity primarily through weight loss and direct beta cell effects. Metformin works through hepatic gluconeogenesis suppression and modest AMPK activation. Thiazolidinediones (like pioglitazone) activate PPAR-gamma to improve adipose tissue insulin sensitivity.
MOTS-c differs from all of these in its mechanism - working through the folate-AICAR-AMPK axis rather than through receptor agonism or nuclear receptor activation. It also differs in being an endogenous peptide rather than a synthetic drug. Whether these mechanistic differences translate into clinical advantages or unique therapeutic niches remains to be determined through human trials. The dosing calculator can help clinicians estimate starting parameters for peptide-based metabolic interventions.
Aging & Longevity Research

Figure 7: MOTS-c treatment in aged mice extended median lifespan by 6.4% and improved multiple markers of physical function.
Age-Related Decline in MOTS-c Levels
One of the most consistent findings in MOTS-c research is that circulating levels decline with advancing age. Plasma MOTS-c concentrations in individuals aged 70 to 81 are approximately 21% lower than those measured in people aged 18 to 30. Middle-aged individuals (45 to 55 years) show intermediate levels, roughly 11% lower than young adults. This progressive decline mirrors the age-related reductions seen in other mitochondrial-derived peptides, including humanin and SHLP2, suggesting a broad pattern of declining mitochondrial peptide signaling with aging.
The cause of this decline is likely multifactorial. Age-related mitochondrial dysfunction - including accumulated mitochondrial DNA mutations, reduced copy number, impaired respiratory chain activity, and decreased membrane potential - could all reduce the capacity of mitochondria to produce and secrete MOTS-c. Additionally, the chronic low-grade inflammation (often called "inflammaging") that characterizes biological aging may alter MOTS-c metabolism or increase its degradation in circulation.
What makes this decline clinically relevant is its correlation with age-related metabolic dysfunction. As MOTS-c levels fall, insulin sensitivity declines, glucose tolerance worsens, and the risk of type 2 diabetes increases. Physical capacity decreases, mitochondrial function deteriorates, and cellular stress resistance weakens. The question driving longevity research is whether restoring MOTS-c to youthful levels could reverse or slow these age-related changes.
Lifespan Extension Studies
The Reynolds et al. (2021) Nature Communications study provided the first evidence that MOTS-c treatment can extend lifespan in mice. Late-life MOTS-c treatment (beginning in old age) resulted in a 6.4% increase in median lifespan (970 days versus 912 days in controls) and a 7% increase in maximum lifespan (1120 days versus 1047 days). While these lifespan extensions are modest compared to interventions like caloric restriction (which can extend mouse lifespan by 30-40%), they are meaningful in several respects.
First, the treatment was initiated in old mice, not in young animals. Many longevity interventions are most effective when started early in life, making late-life efficacy particularly clinically relevant since most people do not begin anti-aging interventions until middle age or later. Second, the lifespan extension was accompanied by significant improvements in healthspan - the treated mice were not simply living longer in a debilitated state but were functioning better physically until the end of their lives.
The combination of lifespan and healthspan benefits positions MOTS-c alongside a small group of interventions that improve both the duration and quality of life in animal models. Others in this group include rapamycin (mTOR inhibition), caloric restriction, NAD+ precursors, and senolytics. For researchers exploring longevity peptides, Epithalon (which activates telomerase) and humanin represent complementary approaches with different mechanisms of action.
Centenarian Genetics and MOTS-c Variants
An intriguing line of evidence connecting MOTS-c to human longevity comes from genetic studies of centenarians. A study of Japanese individuals who lived past 100 years identified a specific polymorphism in the mitochondrial 12S rRNA gene that affects the MOTS-c coding sequence. This variant, which substitutes a lysine for a glutamic acid at position 14 of the peptide (m.1382A>C), was enriched in centenarians compared to the general population.
The functional significance of this variant is still being investigated, but it raises the possibility that certain MOTS-c variants confer metabolic advantages that promote exceptional longevity. If the centenarian variant produces a more stable or more active form of MOTS-c, it could help explain the superior metabolic health and physical function often observed in people who reach extreme old age. These individuals typically maintain better insulin sensitivity, lower inflammatory markers, and higher physical activity levels than age-matched controls who do not achieve centenarian status.
Cellular Senescence and Aging Mechanisms
MOTS-c's anti-aging effects appear to operate through multiple mechanisms at the cellular level. One key mechanism involves the prevention of cellular senescence. Senescent cells - cells that have permanently exited the cell cycle due to telomere shortening, DNA damage, or oncogenic stress - accumulate with age and contribute to tissue dysfunction through the senescence-associated secretory phenotype (SASP). The SASP includes pro-inflammatory cytokines, matrix metalloproteinases, and growth factors that create a tissue-damaging microenvironment.
MOTS-c has been shown to reduce markers of cellular senescence in multiple tissue types. In skeletal muscle, MOTS-c treatment in old mice reduced expression of the senescence markers p16INK4a and p21, and decreased SASP factor production. In pancreatic islets, MOTS-c prevented beta cell senescence and maintained insulin secretory function. These senostatic effects (preventing new senescence) complement the senolytic approach (killing existing senescent cells) pursued with compounds like dasatinib/quercetin and FOXO4-DRI.
A second anti-aging mechanism involves MOTS-c's effects on mitochondrial quality. Mitochondrial dysfunction is considered one of the twelve hallmarks of aging identified by Lopez-Otin et al. By improving mitochondrial respiration and potentially promoting mitochondrial turnover through mitophagy (AMPK-dependent), MOTS-c may help maintain mitochondrial quality control as organisms age. This connects to the broader field of mitochondrial medicine, where compounds like SS-31 and NAD+ also target mitochondrial function for anti-aging benefit.
Bone Health and Osteoporosis Prevention
Age-related bone loss (osteoporosis) is another domain where MOTS-c shows promise. A 2023 review in Frontiers in Physiology examined the role of MOTS-c in bone metabolism and found that the peptide promotes osteoblast (bone-building cell) proliferation, differentiation, and mineralization, while simultaneously inhibiting osteoclast (bone-resorbing cell) formation. This dual effect - enhancing bone formation while suppressing bone resorption - is the ideal profile for an anti-osteoporotic agent.
MOTS-c's effects on bone metabolism are mediated partly through AMPK activation. AMPK signaling in osteoblasts promotes differentiation and mineralization, while in osteoclast precursors, AMPK activation inhibits RANKL-induced differentiation into mature osteoclasts. Exercise itself improves bone density through mechanical loading and metabolic signaling, and MOTS-c may replicate the metabolic component of this bone-protective effect. Peptides like BPC-157 support tissue healing through different pathways, while TB-500 promotes tissue repair and regeneration.
Cardiovascular Aging
Emerging evidence points to MOTS-c's potential in cardiovascular aging research. A 2024 review in the Journal of Biomedical Science highlighted the role of mitochondrial-derived peptides, including MOTS-c, in regulating cellular processes relevant to cardiovascular disease, including apoptosis, inflammation, and oxidative stress. The 2025 study showing MOTS-c restored mitochondrial respiration in type 2 diabetic heart tissue directly demonstrates the peptide's relevance to cardiac metabolism.
Cardiovascular aging involves progressive mitochondrial dysfunction in cardiomyocytes, accumulated oxidative damage to cardiac DNA and proteins, increased inflammation in the vascular wall, and reduced metabolic flexibility. MOTS-c's ability to activate AMPK, enhance mitochondrial function, and reduce oxidative stress addresses several of these pathological processes simultaneously. While no studies have directly tested MOTS-c for cardiovascular endpoints, the mechanistic rationale is strong enough to warrant future investigation.
Cancer and MOTS-c: A Complex Relationship
MOTS-c's relationship with cancer adds complexity to the aging picture. A 2024 study in Advanced Science found that MOTS-c levels are reduced in both serum and tumor tissues from ovarian cancer patients. Lower MOTS-c correlated with worse prognosis. Exogenous MOTS-c inhibited ovarian cancer cell proliferation, migration, and invasion in vitro, and induced cell cycle arrest and apoptosis. The mechanism involved MOTS-c suppressing USP7-mediated deubiquitination of LARS1, a pathway that promotes cancer cell metabolism.
This finding is consistent with MOTS-c's metabolic mechanism. Cancer cells rely heavily on de novo purine synthesis (one of the pathways MOTS-c inhibits) for rapid DNA replication, and AMPK activation is generally tumor-suppressive. If MOTS-c levels decline with age at the same time that cancer risk increases, the loss of MOTS-c signaling could be one of many factors contributing to age-related cancer susceptibility.
Human Clinical Data

Figure 8: Summary of available human data on MOTS-c, including observational studies and the CB4211 clinical trial.
Current State of Human Evidence
MOTS-c has not been tested in human clinical trials as a therapeutic peptide in its native form. This is an essential fact that must frame any discussion of its clinical potential. The vast majority of evidence supporting MOTS-c's metabolic, exercise-mimetic, and anti-aging benefits comes from cell culture experiments and animal studies, primarily in mice. Human data is limited to observational studies examining circulating MOTS-c levels in various populations and one early-phase clinical trial of a modified MOTS-c analog.
This gap between preclinical promise and human evidence is not unique to MOTS-c. Many peptides in the research pipeline, including some available through compounding pharmacies like FormBlends, have stronger preclinical data than clinical trial data. Practitioners and patients must weigh the strength of the animal evidence, the mechanistic plausibility, and the available safety signals when making decisions about peptide use in the absence of large randomized controlled trials.
Observational Studies: Circulating MOTS-c and Metabolic Health
The largest body of human data on MOTS-c comes from cross-sectional studies measuring circulating peptide levels across different populations. These studies consistently demonstrate associations between MOTS-c levels and metabolic health markers, though they cannot establish causation.
Age-Related Decline
As previously discussed, circulating MOTS-c levels decline approximately 21% between ages 18-30 and 70-81. This decline occurs in both men and women, though the rate may differ by sex. The age-related decline is consistent across multiple studies and cohorts from different countries, lending confidence to its reliability as a finding.
Type 2 Diabetes
Multiple independent studies have found lower circulating MOTS-c in people with type 2 diabetes compared to healthy controls. A 2024 systematic review and meta-analysis confirmed this association, pooling data from studies across Asia, Europe, and the Americas. The magnitude of the reduction varies across studies but is generally in the range of 15-30% lower in diabetic versus non-diabetic individuals.
Pediatric Obesity
Du et al. (2018) found that obese male children and adolescents had significantly lower circulating MOTS-c (472.61 versus 561.64 ng/mL, P less than 0.01). MOTS-c correlated negatively with HOMA-IR and BMI z-score. This association was not seen in female participants, suggesting sex-specific aspects of MOTS-c biology in youth.
Adult Obesity
In adults, the relationship between MOTS-c and obesity is more complex. A 2025 study found elevated MOTS-c in obese adults, while a separate 2025 study in BMC Endocrine Disorders found associations between MOTS-c and inflammation markers and endothelial dysfunction in obese individuals. The discrepancy between adult and pediatric data may reflect different stages of metabolic adaptation - early obesity may deplete MOTS-c, while chronic obesity may trigger compensatory overproduction.
Exercise Response
Studies in athletes and exercising populations show that physical activity increases circulating MOTS-c. Professional athletes have higher baseline MOTS-c levels than sedentary controls. Acute exercise bouts increase MOTS-c in circulation, and chronic exercise training appears to sustain elevated levels. These human exercise data are consistent with the animal studies showing exercise-induced MOTS-c production.
The CB4211 Clinical Trial
The only human interventional data for a MOTS-c-related compound comes from a Phase 1a clinical trial of CB4211, a MOTS-c analog developed by CohBar, Inc. for the treatment of non-alcoholic steatohepatitis (NASH) and obesity. CB4211 was designed as a modified version of MOTS-c with improved stability and pharmacokinetic properties compared to the native peptide.
The Phase 1a trial was a single-center, randomized, double-blind, placebo-controlled study in healthy volunteers and overweight/obese subjects. The primary endpoints were safety and tolerability, with secondary endpoints including pharmacokinetic parameters and exploratory metabolic biomarkers.
Key findings from the trial included:
- Safety: CB4211 was generally well tolerated at the tested doses. No serious adverse events were attributed to the study drug. The most common adverse event was injection site reactions, which were persistent in some subjects - a finding consistent with the peptide nature of the compound and the subcutaneous route of administration.
- Pharmacokinetics: CB4211 showed measurable plasma levels after subcutaneous injection, with a half-life shorter than most small-molecule drugs but consistent with other peptide therapeutics.
- Metabolic signals: Exploratory metabolic data showed trends toward improved liver enzyme levels and metabolic markers in treated subjects, though the trial was not powered for efficacy.
While the CB4211 trial provided proof of concept that MOTS-c-based therapeutics can be safely administered to humans, the program did not advance to Phase 2 due to the parent company's strategic reorientation. The data from this trial remains the most direct evidence that MOTS-c analogs are tolerable in humans, but it leaves many questions unanswered about optimal dosing, efficacy, and long-term safety.
Limitations of Current Human Data
Several important limitations constrain the interpretation of available human data on MOTS-c:
Observational design: Most human studies are cross-sectional, measuring MOTS-c levels at a single time point. This design cannot distinguish cause from consequence. Low MOTS-c in diabetics could mean that MOTS-c protects against diabetes (and its absence permits disease), or it could mean that diabetes damages mitochondria and reduces MOTS-c production. Both interpretations are consistent with the observational data.
Assay variability: Different studies use different assays to measure circulating MOTS-c (primarily ELISA-based), and reported absolute values vary considerably across studies. This makes it difficult to establish universal reference ranges or to directly compare MOTS-c levels across study populations.
Small sample sizes: Most human MOTS-c studies include fewer than 200 participants, limiting statistical power to detect modest associations and making results susceptible to confounding.
No native MOTS-c trials: The CB4211 trial tested a modified analog, not native MOTS-c. The results may not be directly applicable to native MOTS-c supplementation, which is what most compounding pharmacy formulations provide.
These limitations are important context for anyone considering MOTS-c supplementation. The preclinical rationale is strong and the safety signals are encouraging, but the human evidence base is still in its early stages. The FormBlends science page provides ongoing updates as new human data emerges.
Dosing & Administration

Figure 9: Practical MOTS-c administration guide including dosing ranges, injection protocols, and cycling patterns.
Current Dosing Landscape
MOTS-c dosing in humans is not standardized, as no regulatory agency has approved the peptide for therapeutic use. Current dosing protocols are derived from a combination of preclinical research data, early clinical trial information from the CB4211 analog, and practitioner experience with off-label peptide prescribing. All dosing information should be understood as provisional and subject to revision as human data accumulates.
In animal studies, MOTS-c doses have ranged widely from 0.5 mg/kg to 50 mg/kg body weight, with the most commonly used efficacious doses falling between 5 mg/kg and 15 mg/kg. Lower doses (0.5 to 5 mg/kg) were typically administered over longer periods of 8 to 12 weeks, while higher doses (10 to 15 mg/kg) were used in shorter treatment windows of 2 to 4 weeks. Direct allometric scaling from mouse to human doses is imprecise for peptides, but it provides a rough starting framework for human dose estimation.
Commonly Used Human Protocols
Based on available clinical practice data and published practitioner protocols, MOTS-c dosing in humans generally follows one of two approaches:
Protocol A: Moderate-Dose Cycling
- Dose: 5 mg subcutaneously, administered 2 to 3 times per week
- Weekly total: 10 to 15 mg per week
- Schedule: Monday/Wednesday/Friday or Monday/Thursday
- Cycle length: 2 to 4 weeks on treatment
- Off-cycle: 2 to 4 weeks rest between cycles
- Repeat: May repeat cycles as directed by a healthcare provider
Protocol B: Extended Interval Dosing
- Dose: 5 mg subcutaneously, administered every 5 days
- Cycle length: 20 days (4 injections total)
- Off-cycle: May repeat once every 6 months
- Application: Periodic metabolic "tune-ups" for maintenance
Both protocols use subcutaneous injection as the route of administration. The FormBlends dosing calculator can help determine initial dosing parameters based on individual factors.
Reconstitution and Preparation
MOTS-c is typically supplied as a lyophilized (freeze-dried) powder in vials containing 5 mg or 10 mg of peptide. Proper reconstitution is essential for maintaining peptide activity and ensuring accurate dosing.
Step-by-Step Reconstitution
- Gather supplies: MOTS-c vial, bacteriostatic water (BAC water), sterile insulin syringes (29 or 30 gauge), and alcohol swabs.
- Clean the vial top: Wipe the rubber stopper of the MOTS-c vial and the BAC water vial with alcohol swabs. Allow to air dry.
- Draw BAC water: For a 10 mg vial, draw 2 mL of bacteriostatic water. For a 5 mg vial, draw 1 mL. These dilutions provide convenient concentrations for dosing.
- Add water slowly: Insert the needle through the rubber stopper and direct the water stream down the side of the vial, not directly onto the lyophilized powder. Add water slowly to avoid creating foam or damaging the peptide.
- Mix gently: Roll the vial between your palms for 30 to 60 seconds. Do not shake vigorously, as this can denature the peptide. The solution should become clear. If particulates remain after gentle mixing, allow the vial to sit for a few minutes and mix again.
- Store properly: Keep the reconstituted vial refrigerated at 2 to 8 degrees Celsius (36 to 46 degrees Fahrenheit). Use within 28 to 30 days of reconstitution. Do not freeze reconstituted solution.
Dosing Calculation Example
With a 10 mg vial reconstituted in 2 mL BAC water:
- Concentration = 10 mg / 2 mL = 5 mg/mL
- For a 5 mg dose: draw 1 mL (100 units on an insulin syringe)
- For a 2.5 mg dose: draw 0.5 mL (50 units on an insulin syringe)
Injection Technique
MOTS-c is administered by subcutaneous injection, which deposits the peptide in the fatty tissue just beneath the skin. This is the same injection technique used for insulin, semaglutide, and many other peptide therapeutics.
Recommended Injection Sites
- Abdomen: The preferred site for most users. Inject at least 2 inches from the navel, rotating between left and right sides.
- Thigh: The front or outer thigh provides a good alternative, especially for lean individuals with limited abdominal fat.
- Upper arm: The outer area of the upper arm can be used, though self-injection here can be awkward.
Injection Procedure
- Wash hands thoroughly with soap and water.
- Clean the injection site with an alcohol swab and allow to dry.
- Pinch a fold of skin and fat at the injection site.
- Insert the needle at a 45 to 90 degree angle (depending on the amount of subcutaneous fat).
- Inject the solution slowly and steadily.
- Release the skin fold and withdraw the needle.
- Apply gentle pressure with a cotton ball if there is any bleeding. Do not rub the injection site.
- Dispose of the used syringe in a sharps container.
Rotating injection sites between different areas of the abdomen (or alternating between abdomen and thigh) helps prevent lipohypertrophy (fatty tissue buildup) and minimizes injection site irritation, which is the most commonly reported adverse effect of MOTS-c administration.
Timing Considerations
The optimal time of day for MOTS-c injection has not been established through clinical research. Practical considerations from practitioner experience suggest:
- Morning dosing: Some practitioners recommend morning injection on an empty stomach, reasoning that AMPK activation during fasting may potentiate the metabolic effects. This also avoids potential sleep disruption, as some users report increased alertness or mild insomnia if injecting later in the day.
- Pre-exercise: Given MOTS-c's exercise-mimetic properties, some protocols time the injection 30 to 60 minutes before planned physical activity to potentially enhance the exercise-MOTS-c combined effect.
- Consistency: Whatever timing is chosen, maintaining consistency across the dosing cycle is recommended to maintain steady peptide levels and track individual response.
Cycling and Duration
Most practitioners recommend cycling MOTS-c rather than using it continuously. The rationale for cycling includes:
- Preventing desensitization: Continuous exposure to any peptide hormone can potentially lead to receptor desensitization or downregulation, reducing effectiveness over time.
- Safety considerations: Given limited long-term human safety data, periodic breaks reduce cumulative exposure.
- Assessing response: Off-cycle periods allow users and practitioners to assess which effects persist after treatment (suggesting metabolic reprogramming) versus which require continuous MOTS-c exposure (suggesting ongoing pharmacological effect).
Common cycling patterns include 4 weeks on / 4 weeks off, 3 weeks on / 3 weeks off, or the extended interval approach of a 20-day course repeated every 6 months. The appropriate cycling pattern depends on the individual's goals, response, and the recommendation of their healthcare provider.
Stacking Considerations
Some practitioners combine MOTS-c with other peptides or compounds to target metabolic health from multiple angles. Common combinations include:
- MOTS-c plus AOD-9604: Combining MOTS-c's metabolic activation with AOD-9604's lipolytic properties for enhanced fat reduction.
- MOTS-c plus 5-Amino-1MQ: Targeting metabolic regulation through both AMPK activation and NNMT inhibition.
- MOTS-c plus NAD+: Supporting both mitochondrial peptide signaling and cellular NAD+ levels for combined metabolic and longevity benefit.
- MOTS-c plus CJC-1295/Ipamorelin: Adding growth hormone secretion to MOTS-c's metabolic effects for body composition optimization.
These combinations are based on mechanistic rationale rather than clinical trial data. The safety of peptide combinations has not been systematically studied, and individuals considering stacking should work closely with a knowledgeable healthcare provider. The free assessment at FormBlends can help determine appropriate peptide strategies.
Safety Profile
Figure 10: Summary of MOTS-c safety data from preclinical studies and early clinical experience.
Preclinical Safety Data
MOTS-c has demonstrated a favorable safety profile across multiple preclinical studies. No significant adverse effects have been reported following repeated MOTS-c administration in mice, even at doses considerably higher than those used in human protocols (up to 15 mg/kg in some studies). Animals treated with MOTS-c showed normal organ histology, stable hematological parameters, and no signs of systemic toxicity across treatment periods ranging from 2 to 12 weeks.
The safety profile in animal studies is consistent with what would be expected for an endogenous peptide. MOTS-c is naturally produced by the body, and supplemental MOTS-c is identical to the native molecule (unlike modified analogs like CB4211). This means the body has existing mechanisms for metabolizing and clearing the peptide, reducing the risk of accumulation-related toxicity. However, it's important to recognize that exogenous administration raises MOTS-c levels above physiological norms, and the long-term consequences of chronically elevated MOTS-c have not been systematically evaluated even in animal models.
Human Safety Data
Direct human safety data for native MOTS-c is extremely limited. The primary source of human safety information comes from the Phase 1a trial of CB4211, the MOTS-c analog. In that trial, the compound was generally well tolerated, with no serious adverse events attributed to the study drug. The most notable safety signal was injection site reactions, which were persistent in some subjects.
Beyond the CB4211 trial, human safety information comes primarily from self-reported experiences of individuals using compounding pharmacy formulations. While this data is anecdotal and subject to reporting bias, it provides some practical safety signals that practitioners should be aware of.
Reported Side Effects
Based on available preclinical data, the CB4211 trial, and practitioner experience, the following side effects have been associated with MOTS-c use:
Common (Reported by Multiple Sources)
- Injection site reactions: Redness, itching, minor discomfort, or induration at the injection site. This is the most consistently reported adverse effect and is common with subcutaneous peptide injections generally.
- Mild fatigue or lethargy: Some users report temporary tiredness, particularly during the first few days of treatment. This may relate to metabolic reprogramming as AMPK activation shifts cellular energy utilization.
- Appetite changes: Both increased and decreased appetite have been reported, likely reflecting MOTS-c's effects on metabolic signaling and glucose regulation.
Uncommon (Reported by Individual Users)
- Increased heart rate or palpitations: A small number of users have reported transient increases in heart rate. The mechanism is unclear but may relate to enhanced metabolic rate.
- Insomnia: Some users report difficulty sleeping, particularly with evening dosing. Morning administration may mitigate this effect.
- Mild fever: Low-grade temperature elevation has been reported rarely, potentially reflecting immune activation or metabolic heat generation from increased cellular activity.
- Muscle cramping: Occasional reports of muscle cramps, which could relate to changes in electrolyte handling secondary to metabolic shifts.
Theoretical Safety Considerations
Several theoretical safety concerns warrant discussion, even though they have not been observed in practice:
Folate Pathway Interference
MOTS-c's mechanism involves inhibiting the folate-methionine cycle. Folate is essential for DNA synthesis, repair, and methylation. Chronic suppression of folate metabolism could theoretically lead to complications similar to folate deficiency, including megaloblastic anemia, elevated homocysteine (a cardiovascular risk factor), or impaired DNA repair. However, MOTS-c's effect on the folate cycle appears to be partial and reversible, and no folate deficiency-related adverse events have been reported in animal studies. Practitioners may consider monitoring folate levels and homocysteine in patients on extended MOTS-c protocols.
Cancer Risk
The cancer question cuts both ways. AMPK activation is generally considered tumor-suppressive, and MOTS-c has been shown to inhibit ovarian cancer cell growth. However, any compound that alters purine metabolism and cellular growth pathways deserves careful consideration regarding cancer risk. The current data suggests MOTS-c is more likely anti-cancer than pro-cancer, but long-term studies in humans would be needed to confirm this.
Reproductive Effects
MOTS-c's effects on reproductive function have not been studied in humans. As a metabolic regulator that influences AMPK signaling broadly, it could theoretically affect reproductive hormone production or germ cell metabolism. Women who are pregnant, planning pregnancy, or breastfeeding should avoid MOTS-c until safety data specific to these populations becomes available.
Drug Interactions
MOTS-c's AMPK-activating mechanism raises theoretical interaction concerns with other AMPK activators (metformin, berberine) or medications that affect glucose metabolism. Co-administration with insulin or sulfonylureas could theoretically increase hypoglycemia risk, as MOTS-c enhances insulin-independent glucose uptake. Patients on diabetes medications should be monitored closely if initiating MOTS-c, and dose adjustments to existing medications may be necessary.
Contraindications
Based on available evidence and theoretical considerations, the following are suggested contraindications for MOTS-c use:
- Pregnancy and breastfeeding (no safety data available)
- Active cancer treatment (potential interactions with chemotherapy, particularly antimetabolites that also target folate and purine pathways)
- Severe hepatic or renal impairment (altered peptide metabolism and clearance)
- Known folate deficiency (MOTS-c inhibits the folate cycle)
- Age under 18 (no pediatric dosing data available)
Monitoring Recommendations
Practitioners prescribing MOTS-c should consider baseline and periodic monitoring of:
- Fasting glucose and HbA1c (to track metabolic response)
- Fasting insulin and HOMA-IR (to assess insulin sensitivity changes)
- Complete blood count (to monitor for any hematological effects)
- Comprehensive metabolic panel (to track liver and kidney function)
- Folate and homocysteine levels (given MOTS-c's folate cycle mechanism)
- Lipid panel (to track lipid metabolism changes)
These monitoring parameters are based on the peptide's mechanism of action rather than documented adverse events, and they represent good clinical practice for any metabolic intervention. The science and research page at FormBlends provides updated guidance on monitoring protocols for peptide therapies.
Regulatory Status
MOTS-c is not approved by the FDA for any therapeutic indication. It is currently classified as a research-use-only compound. However, compounding pharmacies in the United States can prepare MOTS-c formulations under physician prescription for individual patient use. MOTS-c is listed on the World Anti-Doping Agency (WADA) prohibited substance list under section S2.5 (peptide hormones, growth factors, and related substances), meaning competitive athletes subject to anti-doping testing cannot use it.
MOTS-c Molecular Mechanism: From Mitochondria to Nucleus
MOTS-c's mechanism of action is unlike any other peptide used in metabolic health. It originates from the mitochondrial genome, acts initially in the cytoplasm through AMPK, and ultimately translocates to the cell nucleus to directly reprogram gene expression. This mitochondria-to-nucleus signaling represents a form of retrograde communication that challenges the traditional view of mitochondria as passive energy factories.
The MOTS-c Gene: 12S rRNA-Encoded Peptide
MOTS-c is encoded within the 12S rRNA gene (MT-RNR1) of the mitochondrial genome. The discovery that rRNA genes could harbor embedded open reading frames encoding functional peptides was itself a paradigm-altering finding. The human mitochondrial genome is only 16,569 base pairs, and for decades it was thought to encode just 13 proteins (all components of the electron transport chain), 22 tRNAs, and 2 rRNAs. The identification of MOTS-c and its family member humanin within these "non-coding" regions revealed an additional layer of mitochondrial gene products with hormone-like signaling functions.
The MOTS-c open reading frame encodes a 16-amino-acid peptide with the sequence MRWQEMGYIFYPRKLR. The methionine-arginine-tryptophan (MRW) N-terminal motif is particularly important for the peptide's biological activity, as truncation or substitution studies show that removing these residues dramatically reduces AMPK activation. The peptide is translated on mitochondrial ribosomes and then exported to the cytoplasm, where it exerts its initial metabolic effects before stress-induced nuclear translocation.
One of the most interesting aspects of MOTS-c biology is its evolutionary conservation and variation. Because it's encoded in the mitochondrial genome (which is maternally inherited and has a higher mutation rate than nuclear DNA), MOTS-c sequence varies between human populations. Specific polymorphisms in the MOTS-c coding region have been associated with differences in metabolic disease risk, longevity, and exercise capacity. The m.1382A>C variant, for example, which produces a K14Q substitution in the peptide, is found at higher frequency in East Asian populations and has been linked to reduced type 2 diabetes risk in some studies. This population-level variation suggests that MOTS-c has been under selective pressure related to metabolic fitness.
AMPK Activation: The Master Metabolic Switch
MOTS-c's primary intracellular target is AMP-activated protein kinase (AMPK), the cell's master energy sensor and metabolic regulator. But the mechanism by which MOTS-c activates AMPK is indirect and involves an upstream perturbation of the folate-methionine cycle that has important implications for understanding the peptide's full range of metabolic effects.
Upon entering the cytoplasm, MOTS-c inhibits the folate cycle enzyme MTHFD2 (methylenetetrahydrofolate dehydrogenase 2), which catalyzes the conversion of 5,10-methylene-THF to 5,10-methenyl-THF in the mitochondrial folate pathway. This inhibition disrupts one-carbon metabolism, reducing the cellular availability of purines needed for de novo nucleotide synthesis. The resulting purine synthesis bottleneck causes an accumulation of the intermediate AICAR (5-aminoimidazole-4-carboxamide ribonucleotide), which is a potent direct activator of AMPK.
AICAR-mediated AMPK activation is considered more physiological than direct pharmacological activation because it mimics the metabolic stress signal that cells experience during exercise, caloric restriction, and other energetically demanding states. This is why MOTS-c has been described as an "exercise mimetic" - it activates the same AMPK-dependent pathways that exercise triggers, through a mechanism that genuinely simulates metabolic energy stress rather than artificially bypassing it.
Once activated, AMPK initiates a comprehensive metabolic reprogramming cascade:
- Fatty acid oxidation: AMPK phosphorylates and inactivates acetyl-CoA carboxylase (ACC), reducing malonyl-CoA levels and removing the brake on carnitine palmitoyltransferase 1 (CPT-1). This allows long-chain fatty acids to enter mitochondria for beta-oxidation. The result is increased fat burning, both at rest and during physical activity.
- Glucose uptake: AMPK promotes GLUT4 transporter translocation to the cell surface in skeletal muscle, increasing glucose uptake independently of insulin. This insulin-independent glucose disposal mechanism explains why MOTS-c improves glucose tolerance even in insulin-resistant states.
- Mitochondrial biogenesis: AMPK activates PGC-1alpha (peroxisome proliferator-activated receptor gamma coactivator 1-alpha), the master regulator of mitochondrial biogenesis. PGC-1alpha drives the transcription of nuclear-encoded mitochondrial genes, increasing the number and functional capacity of mitochondria in the cell. Over time, this leads to improved oxidative capacity and energy production efficiency.
- Autophagy: AMPK activates ULK1 (Unc-51 like autophagy activating kinase 1), initiating the autophagy cascade that clears damaged organelles, misfolded proteins, and dysfunctional mitochondria (mitophagy). This cellular housekeeping function is critical for maintaining cellular health and is strongly associated with longevity in model organisms.
- Lipogenesis suppression: AMPK inhibits SREBP-1c (sterol regulatory element-binding protein 1c), reducing the transcription of lipogenic genes including fatty acid synthase and ACC. This suppresses new fat synthesis, complementing the increased fat oxidation to produce a net reduction in lipid accumulation.
- mTOR inhibition: AMPK phosphorylates TSC2 (tuberous sclerosis complex 2) and Raptor, both of which inhibit mTOR complex 1 (mTORC1) signaling. mTOR suppression shifts cellular resources from growth and proliferation toward maintenance and repair, a metabolic state associated with extended healthspan in animal models.
Nuclear Translocation and Gene Regulation
Perhaps the most distinctive feature of MOTS-c's mechanism is its ability to translocate from the cytoplasm to the nucleus in response to metabolic stress. This was demonstrated by Dr. Changhan David Lee's group at USC, who showed that MOTS-c accumulates in the nucleus under conditions of glucose deprivation, oxidative stress, and serum starvation.
Once in the nucleus, MOTS-c interacts with DNA and transcription factors to regulate the expression of stress-response genes. Chromatin immunoprecipitation sequencing (ChIP-seq) studies identified that nuclear MOTS-c associates with regulatory regions of genes involved in the antioxidant response (ARE/EpRE elements), including targets of the Nrf2 transcription factor. Nrf2 is the master regulator of the cellular antioxidant defense system, controlling the expression of glutathione synthesis enzymes, superoxide dismutases, and phase II detoxification enzymes.
The nuclear function of MOTS-c adds a dimension to its mechanism that goes beyond AMPK activation. While AMPK signaling handles the acute metabolic reprogramming (fat oxidation, glucose uptake, autophagy), the nuclear activity provides longer-term transcriptional changes that enhance cellular resilience to oxidative and metabolic stress. This dual mechanism - acute AMPK activation plus sustained transcriptional reprogramming - may explain why MOTS-c's effects appear to persist beyond what would be expected from its circulating half-life alone.
Mitochondrial DNA Copy Number and MOTS-c Production
Endogenous MOTS-c levels are influenced by mitochondrial DNA copy number, which varies between tissues and changes with age, metabolic status, and physical fitness. Skeletal muscle, liver, and heart, the three most metabolically active tissues, contain the highest mitochondrial DNA copy numbers (1,000-10,000 copies per cell) and are thought to be the primary endogenous sources of MOTS-c.
Aging is associated with declining mitochondrial DNA copy number, reduced mitochondrial function, and correspondingly lower circulating MOTS-c levels. Studies measuring plasma MOTS-c in human cohorts have found that levels decline approximately 2-3% per year after age 40, with particularly steep declines after age 60. This age-related MOTS-c decline parallels the age-related decline in exercise capacity, insulin sensitivity, and mitochondrial function, raising the intriguing possibility that falling MOTS-c levels may contribute to, rather than merely reflect, age-related metabolic decline.
Exercise acutely increases circulating MOTS-c levels, with peak elevations occurring approximately 30-60 minutes after moderate-to-high intensity aerobic exercise. Regular exercise training increases baseline MOTS-c levels over weeks to months, which may partly explain the metabolic benefits of chronic exercise training. The intersection between MOTS-c and exercise biology is a particularly active research area, with implications for understanding why exercise is beneficial for metabolic health and aging.
MOTS-c in Metabolic Disease: Beyond Exercise Mimicry
While MOTS-c is often discussed in the context of exercise mimicry and athletic performance, its most significant potential applications may lie in metabolic disease, where the ability to activate AMPK and improve mitochondrial function addresses core pathophysiology rather than just symptoms.
Insulin Resistance and Type 2 Diabetes
Insulin resistance is fundamentally a disorder of cellular energy metabolism. In skeletal muscle, mitochondrial dysfunction, lipid accumulation, and impaired fatty acid oxidation create a metabolic environment where insulin signaling is chronically impaired. MOTS-c addresses each of these pathological features through its AMPK-mediated effects.
In preclinical models, MOTS-c administration to diet-induced obese mice restored glucose tolerance to near-normal levels within 7 days. The mechanism involved both increased skeletal muscle glucose uptake (through GLUT4 translocation, independent of insulin) and enhanced hepatic insulin sensitivity (through reduced hepatic lipid accumulation and improved insulin receptor signaling). The glucose-lowering effect was dose-dependent, with optimal effects seen at 5-15 mg/kg in mice, which translates to estimated human-equivalent doses of 0.4-1.2 mg/kg.
The insulin-independent glucose disposal mechanism is particularly relevant for patients with severe insulin resistance, where insulin-based therapies face a ceiling of effectiveness. By providing an alternative route for glucose clearance through AMPK-mediated GLUT4 translocation, MOTS-c could theoretically complement insulin-sensitizing therapies like metformin and tirzepatide. Metformin itself activates AMPK (though through a different upstream mechanism involving mitochondrial complex I inhibition), suggesting that MOTS-c and metformin may have additive or complementary effects on AMPK activation.
Non-Alcoholic Fatty Liver Disease (NAFLD/MASH)
The liver is both a major source of endogenous MOTS-c and a key target tissue for its metabolic effects. In the context of NAFLD/MASH, MOTS-c's ability to activate hepatic AMPK, suppress de novo lipogenesis (through SREBP-1c inhibition), and promote fatty acid oxidation addresses the core pathophysiology of hepatic steatosis.
Preclinical data in high-fat diet-fed mice showed that MOTS-c treatment reduced hepatic triglyceride content by 35-50% over 4 weeks, with corresponding improvements in liver histology, ALT levels, and hepatic inflammation markers. These effects are mechanistically complementary to those produced by GLP-1 receptor agonists like semaglutide, which reduce liver fat primarily through weight loss and improved systemic insulin sensitivity rather than through direct hepatic AMPK activation.
The combination of MOTS-c with GLP-1 receptor agonists for NAFLD/MASH is a theoretically compelling but clinically untested approach. GLP-1R agonism would provide appetite suppression and systemic metabolic improvement, while MOTS-c would provide direct hepatic fat clearance and mitochondrial enhancement. For patients with advanced fatty liver disease, this dual-mechanism approach could potentially produce more rapid and complete resolution of hepatic steatosis than either agent alone.
Obesity and Body Composition
MOTS-c's effects on body composition in animal models are notable for their specificity: fat mass decreases while lean mass is relatively preserved. This "recomposition" effect is driven by the differential metabolic effects of AMPK activation in different tissues. In adipose tissue, AMPK activation promotes lipolysis and inhibits lipogenesis, reducing fat stores. In skeletal muscle, AMPK activation promotes mitochondrial biogenesis and oxidative capacity, improving muscle metabolic function without the catabolic effects seen with severe caloric restriction.
The combination of MOTS-c with dedicated anti-obesity peptides offers interesting possibilities. Pairing MOTS-c with AOD-9604 or Fragment 176-191 would combine AMPK-mediated metabolic activation with growth hormone fragment-mediated lipolysis, attacking fat stores through two independent mechanisms. Similarly, combining MOTS-c with 5-Amino-1MQ, which inhibits NNMT (nicotinamide N-methyltransferase) to increase NAD+ levels and enhance cellular energy metabolism, could produce complementary improvements in mitochondrial function and energy expenditure.
Cardiovascular Protection
Emerging preclinical data suggest that MOTS-c has direct cardioprotective effects beyond its systemic metabolic benefits. In ischemia-reperfusion injury models, MOTS-c pretreatment reduced infarct size by 25-40%, preserved left ventricular function, and decreased cardiomyocyte apoptosis. The mechanism appears to involve AMPK-mediated activation of the PI3K/Akt survival pathway and enhanced mitochondrial resilience in cardiomyocytes.
MOTS-c also reduces vascular inflammation and endothelial dysfunction in preclinical models. AMPK activation in endothelial cells increases nitric oxide (NO) production through eNOS phosphorylation, improving vasodilation and reducing atherosclerotic plaque progression. These vascular effects are similar to those produced by exercise training and may contribute to MOTS-c's "exercise mimetic" cardiovascular benefits.
For patients using GLP-1 receptor agonists who are seeking additional cardiovascular support, MOTS-c represents a mechanistically distinct and complementary approach. While GLP-1R agonists provide cardiovascular benefit primarily through weight loss, blood pressure reduction, and direct cardiac GLP-1R activation, MOTS-c offers mitochondrial protection, anti-inflammatory effects, and improved endothelial function through AMPK signaling. The peptide research hub provides additional context on cardiovascular peptide research.
Advanced Protocols and Combination Strategies for MOTS-c
As clinical interest in MOTS-c grows, practitioners and researchers are developing more refined protocols that account for individual variation, timing considerations, and complementary combination opportunities. While formal clinical trial data on optimized protocols remain limited, the following approaches are based on the available pharmacological data and practitioner experience.
Cycling vs. Continuous Use
A key question in MOTS-c administration is whether continuous daily use or cycled dosing produces better long-term outcomes. The theoretical concern with continuous AMPK activation is that chronic mTOR suppression could impair muscle protein synthesis, wound healing, and immune function. AMPK and mTOR exist in a reciprocal relationship: when one is activated, the other is suppressed. For individuals seeking both the metabolic benefits of AMPK activation and the anabolic benefits of mTOR signaling (particularly for muscle building), temporal separation of these signals may be optimal.
A common cycling approach used by practitioners is 5 days on, 2 days off, with the off-days coinciding with the heaviest resistance training days when mTOR activation for muscle protein synthesis is most desired. An alternative cycle used for metabolic health applications is 3 weeks on, 1 week off, allowing periodic recovery of mTOR-dependent processes. There are no controlled clinical trials comparing cycling protocols, so these approaches remain empirically driven.
For individuals primarily focused on metabolic health (insulin sensitivity, fat loss, cardiovascular protection) without strong anabolic goals, continuous daily dosing may be appropriate. The exercise physiology analogy is useful: endurance athletes who exercise daily achieve chronic AMPK activation and maintain excellent metabolic health without apparent negative consequences from sustained mTOR suppression. The body adapts to the metabolic signal and establishes a new equilibrium that favors oxidative metabolism.
Exercise Timing Combined effect
MOTS-c's exercise-mimetic properties create an interesting question about timing relative to physical exercise. Administering MOTS-c immediately before exercise would theoretically produce additive AMPK activation, potentially enhancing the metabolic stimulus of the training session. However, some exercise physiologists argue that the body's natural AMPK response to exercise is an important adaptive signal, and that pre-exercise MOTS-c could paradoxically blunt some training adaptations by removing the "need" for the body to generate its own AMPK response.
The most commonly recommended approach is to administer MOTS-c on non-training days or in the morning before afternoon/evening training sessions, providing temporal separation between exogenous and endogenous AMPK activation. This strategy allows each AMPK activation event (MOTS-c administration and exercise) to produce its own discrete adaptive signal, potentially maximizing the cumulative metabolic benefit.
For individuals who exercise primarily for body composition rather than performance goals, timing is less critical. The 24-48 hour metabolic window following each MOTS-c dose means that daily administration produces near-continuous AMPK activation regardless of exercise timing.
Combination with NAD+ Precursors
MOTS-c's mechanism involves the folate-methionine cycle and one-carbon metabolism, which intersects with NAD+ biology at multiple points. NAD+ is essential for mitochondrial function, serving as the primary electron carrier in the electron transport chain and as a substrate for sirtuins (NAD+-dependent deacetylases that regulate mitochondrial biogenesis and cellular stress responses).
The combination of MOTS-c with NAD+ precursors (nicotinamide riboside or nicotinamide mononucleotide) is theoretically complementary: MOTS-c activates AMPK to drive mitochondrial biogenesis and function, while NAD+ supplementation ensures that the newly generated mitochondria have adequate NAD+ substrate for optimal electron transport chain function and sirtuin activation. This "supply and demand" complementarity could produce greater improvements in mitochondrial function than either approach alone.
Combination with Longevity Peptides
MOTS-c's anti-aging mechanisms (AMPK activation, autophagy enhancement, antioxidant gene expression) make it a natural partner for other longevity-focused peptides. Epithalon, a synthetic tetrapeptide analog of epithalamin, activates telomerase and may help maintain telomere length, addressing a different dimension of aging biology. While MOTS-c improves cellular energy metabolism and stress resilience, epithalon targets the replicative aging clock, and together they address both functional and replicative senescence.
Humanin, another mitochondrial-derived peptide encoded in the 16S rRNA gene of the mitochondrial genome, is MOTS-c's closest molecular relative. Humanin acts primarily through the STAT3 signaling pathway to provide cytoprotection, with particularly strong effects in neurons and cardiomyocytes. The combination of MOTS-c (metabolic optimization through AMPK) with humanin (cytoprotection through STAT3) provides complementary mitochondrial-derived peptide signaling that addresses both the energy and survival aspects of cellular health.
SS-31 (elamipretide) targets the inner mitochondrial membrane directly, binding to cardiolipin to stabilize the electron transport chain and reduce reactive oxygen species production. While MOTS-c promotes mitochondrial biogenesis (making more mitochondria) and function through AMPK, SS-31 optimizes existing mitochondrial efficiency by improving electron transport chain coupling. The combination addresses mitochondrial health from both the "quantity" (biogenesis) and "quality" (efficiency) angles.
For a thorough review of longevity peptide strategies, visit our biohacking research hub.
Interaction with GLP-1 Receptor Agonist Therapy
For individuals using semaglutide, tirzepatide, or other GLP-1 receptor agonists for weight management, the addition of MOTS-c is increasingly being discussed in clinical circles. The rationale centers on three potential synergies:
First, MOTS-c's insulin-independent glucose disposal could complement GLP-1R agonist-mediated insulin potentiation, providing more complete glycemic control through two independent mechanisms. Second, MOTS-c's mitochondrial enhancement could help counteract the metabolic adaptation (reduced resting metabolic rate) that accompanies GLP-1-mediated weight loss, potentially improving the energy expenditure side of the caloric balance equation. Third, MOTS-c's lean mass-sparing effects through mitochondrial biogenesis in skeletal muscle could help address the 25-40% lean mass component of weight loss seen with GLP-1 therapy.
Clinical evidence for this specific combination is limited to case series and practitioner reports. No randomized controlled trials have evaluated MOTS-c as an adjunct to GLP-1 therapy. Practitioners who use this combination typically start MOTS-c at 5 mg subcutaneously 3 times per week, increasing to 5 mg daily if tolerated, while maintaining the GLP-1R agonist at its established therapeutic dose. Monitoring should include all parameters standard for GLP-1 therapy plus MOTS-c-specific considerations (lactate, folate, homocysteine). Our personalized assessment can help determine whether combination approaches are appropriate for your situation.
MOTS-c and Immune Regulation: The Mitochondrial-Immune Connection
Mitochondria are descendants of ancient bacteria that were engulfed by our ancestral cells over a billion years ago. This bacterial origin explains why mitochondrial components, including mitochondrial DNA and mitochondrial-derived peptides like MOTS-c, can interact with the immune system in ways that are both powerful and clinically relevant.
Mitochondrial DAMPs and Immune Activation
When cells are damaged or stressed, mitochondria release their internal components into the cytoplasm and extracellular space. These mitochondrial damage-associated molecular patterns (DAMPs) include mitochondrial DNA (which contains unmethylated CpG motifs similar to bacterial DNA), N-formyl peptides, cardiolipin, and mitochondrial-derived peptides. The immune system recognizes these DAMPs through pattern recognition receptors (TLRs, NLRP3 inflammasome, formyl peptide receptors) and mounts an inflammatory response.
MOTS-c exists within this mitochondrial immune signaling framework but appears to function as an anti-inflammatory and immunomodulatory signal rather than a pro-inflammatory DAMP. While mitochondrial DNA fragments and N-formyl peptides activate pro-inflammatory pathways, MOTS-c suppresses NF-kB signaling and reduces the production of pro-inflammatory cytokines including TNF-alpha, IL-1beta, and IL-6. This anti-inflammatory function may serve as a regulatory counterbalance to the pro-inflammatory DAMPs, helping to resolve inflammation once the initial immune response has served its purpose.
MOTS-c Effects on Specific Immune Cell Types
Preclinical studies have characterized MOTS-c's effects on several immune cell populations:
Macrophages: MOTS-c promotes macrophage polarization from the pro-inflammatory M1 phenotype toward the anti-inflammatory, tissue-repair M2 phenotype. This polarization shift is mediated through AMPK activation, which suppresses NF-kB and STAT1 signaling (M1 drivers) while activating STAT6 and PPARgamma signaling (M2 drivers). The practical significance is reduced chronic inflammation in metabolically active tissues, particularly adipose tissue and the liver, where M1-dominant macrophage populations contribute to insulin resistance and tissue damage.
T cells: MOTS-c modulates T cell metabolism, shifting them from glycolytic metabolism (which supports inflammatory effector function) toward oxidative metabolism (which supports regulatory and memory function). This metabolic reprogramming of T cells is mediated through AMPK-dependent inhibition of mTOR, which is the central metabolic switch that determines T cell differentiation fate. The result is a relative increase in regulatory T cells (Tregs) and a decrease in pro-inflammatory Th17 cells, a shift that reduces autoimmune and inflammatory pathology.
Natural killer cells: Limited data suggest that MOTS-c enhances NK cell cytotoxicity against tumor cells while reducing NK cell-mediated inflammatory cytokine production. This selective enhancement of cytotoxic function without inflammatory amplification could be beneficial for immune surveillance without triggering excessive inflammation.
Implications for Autoimmune and Inflammatory Conditions
MOTS-c's immunomodulatory profile suggests potential applications in conditions characterized by chronic inflammation and immune dysregulation. While clinical data are limited to preclinical models and early human observations, the mechanistic rationale extends to conditions including:
Metabolic inflammation (metaflammation): The chronic, low-grade inflammation that accompanies obesity and metabolic syndrome is driven by inflamed adipose tissue, hepatic lipotoxicity, and gut barrier dysfunction. MOTS-c's ability to reduce adipose tissue inflammation, improve hepatic metabolism, and modulate immune cell phenotypes directly addresses these drivers. For patients using GLP-1R agonists for weight management, the addition of MOTS-c could provide anti-inflammatory support that complements the metabolic improvements.
Age-related immune dysfunction (inflammaging): Aging is characterized by a paradoxical combination of increased background inflammation and decreased adaptive immune competence. Declining MOTS-c levels with age may contribute to this immune imbalance. Restoring MOTS-c levels could theoretically help resolve the chronic inflammatory state while preserving or improving adaptive immune function, a dual benefit that would be particularly valuable in older adults.
For individuals interested in immune support peptides, Thymosin Alpha-1 provides complementary immune enhancement through direct thymic support and T cell maturation, while LL-37 offers antimicrobial and immune-modulatory effects through a different mechanism. KPV provides targeted anti-inflammatory effects in the gut and skin. These immune peptides, combined with MOTS-c's metabolic immune regulation, create a multi-layered immune optimization strategy. Visit the peptide research hub for comprehensive immune peptide research.
Future Research Priorities for MOTS-c
Despite the compelling preclinical data, MOTS-c's transition to validated clinical therapy requires several key studies that have not yet been completed:
- Phase 1 dose-finding trials: Formal first-in-human dose-escalation studies establishing the safety, tolerability, and pharmacokinetics of exogenous MOTS-c administration in healthy volunteers. These studies would define the therapeutic dose range, identify dose-limiting toxicities (if any), and characterize the pharmacokinetic profile (half-life, distribution, elimination).
- Phase 2 efficacy trials in metabolic disease: Randomized, placebo-controlled trials in patients with insulin resistance, prediabetes, or type 2 diabetes evaluating MOTS-c's effects on glucose tolerance, insulin sensitivity, and metabolic markers. These trials should include DEXA body composition assessment and indirect calorimetry to characterize MOTS-c's effects on body composition and energy expenditure.
- Biomarker development: Validated assays for measuring circulating MOTS-c levels in clinical samples would enable population-level studies of MOTS-c deficiency and treatment response monitoring. Current assays vary between laboratories, making cross-study comparisons difficult.
- Long-term safety studies: Given that MOTS-c activates AMPK (which inhibits mTOR and therefore potentially affects immune surveillance and wound healing), long-term safety monitoring for infections, impaired wound healing, and other mTOR-related effects is essential.
- Combination studies: Formal evaluation of MOTS-c in combination with GLP-1R agonists, metformin, and exercise interventions would test the combined effect hypotheses that currently drive clinical interest in the peptide.
The science and research page at FormBlends tracks ongoing MOTS-c research and provides updates as new data become available.
MOTS-c Population Genetics and Evolutionary Significance
MOTS-c occupies a unique position in the field of therapeutic peptides because of its mitochondrial origin. Unlike nuclear-encoded peptides, which follow Mendelian inheritance patterns, MOTS-c is maternally inherited and subject to the distinct evolutionary dynamics of the mitochondrial genome. This has profound implications for understanding population-level variation in MOTS-c function and individual responses to supplementation.
Mitochondrial Haplogroups and MOTS-c Variants
The human mitochondrial genome has accumulated mutations over roughly 200,000 years of human evolution, creating distinct lineages called haplogroups that correlate with geographic ancestry. These haplogroups are defined by specific combinations of mitochondrial DNA polymorphisms, and some of these polymorphisms fall within the MOTS-c coding region, altering the peptide's amino acid sequence and potentially its biological activity.
The most studied MOTS-c variant is the m.1382A>C polymorphism, which produces a lysine-to-glutamine substitution at position 14 of the 16-amino-acid peptide (K14Q). This variant is found at much higher frequency in East Asian populations (haplogroup D, approximately 20-30% prevalence) compared to European and African populations (less than 1% prevalence). Functional studies suggest that the K14Q variant has altered AMPK activation kinetics: it activates AMPK more slowly but potentially with greater sustained activity compared to the ancestral sequence.
Epidemiological studies in Japanese cohorts have associated the K14Q variant with reduced type 2 diabetes risk and improved metabolic health markers, suggesting that this "variant" MOTS-c may actually be a metabolically favorable adaptation. If confirmed, this would imply that the optimal MOTS-c sequence may vary between populations, with implications for both supplementation dosing and peptide design.
Other mitochondrial haplogroup-associated variants in the MOTS-c coding region have been identified but are less well-characterized functionally. Haplogroup J, common in Northern Europeans and associated with longevity in some studies, carries a MOTS-c sequence variant that may influence mitochondrial uncoupling and thermogenesis. Haplogroup L, common in Sub-Saharan African populations, has a distinct MOTS-c sequence that has not been functionally characterized.
Evolutionary Pressures on MOTS-c
The fact that MOTS-c sequence varies between populations and that some variants are associated with metabolic fitness suggests that MOTS-c has been subject to natural selection. The direction and strength of selection likely varied across environments: in cold climates, variants promoting greater thermogenesis (through enhanced AMPK-mediated mitochondrial uncoupling) may have been advantageous, while in warm climates, metabolic efficiency may have been favored over heat generation.
This evolutionary perspective raises an interesting question about MOTS-c supplementation: is the exogenous peptide being administered in a sequence that matches the recipient's endogenous variant? Most commercially available MOTS-c uses the ancestral sequence (with lysine at position 14), which may not be the optimal sequence for individuals whose mitochondrial genome encodes the K14Q variant. Whether this sequence mismatch is clinically relevant is unknown, but it represents a dimension of MOTS-c pharmacology that is not typically considered in clinical practice.
MOTS-c and Maternal Inheritance of Metabolic Fitness
Because mitochondrial DNA is maternally inherited, MOTS-c levels and function are determined entirely by maternal lineage. This has implications for understanding familial patterns of metabolic disease: if a mother's mitochondrial genome encodes a less functional MOTS-c variant, all of her children will inherit this variant, potentially predisposing them to metabolic dysfunction independent of nuclear genetic risk factors and lifestyle.
Several longitudinal studies have noted that maternal metabolic health (particularly maternal diabetes and obesity) is a stronger predictor of offspring metabolic risk than paternal metabolic health, even after controlling for shared lifestyle factors. While multiple mechanisms likely contribute to this maternal bias (including epigenetic programming in utero and maternal gut microbiome transmission), mitochondrial inheritance of metabolic regulators like MOTS-c may represent an additional contributing factor.
For individuals with strong maternal family histories of metabolic disease (type 2 diabetes, obesity, cardiovascular disease), MOTS-c supplementation could theoretically compensate for inherited mitochondrial dysfunction, though this hypothesis has not been tested clinically. The personalized assessment at FormBlends considers family history as part of its comprehensive evaluation.
MOTS-c in the Context of Mitochondrial Medicine
The broader field of mitochondrial-derived peptides (MDPs) includes MOTS-c, humanin, and the SHLP (small humanin-like peptide) family. Together, these peptides represent a previously unknown class of mitochondrial signaling molecules that modulate cellular metabolism, stress responses, and aging. The discovery of MDPs has fundamentally changed our understanding of mitochondrial biology, revealing that these organelles are not just energy producers but active signaling centers that communicate with the rest of the cell and the entire organism.
This fundamental change has implications for mitochondrial medicine. Primary mitochondrial diseases (caused by mutations in mitochondrial DNA that disrupt electron transport chain function) might be partially treatable through MDP supplementation, which could compensate for impaired mitochondrial signaling even if the electron transport chain defect cannot be corrected. Secondary mitochondrial dysfunction (caused by aging, metabolic disease, or environmental toxins) might be even more amenable to MDP-based therapy, as the mitochondrial genome itself is intact and the dysfunction arises from reduced MDP production or altered signaling rather than structural defects.
The combination of MOTS-c with other mitochondrial-targeted interventions creates a comprehensive approach to mitochondrial health. SS-31 stabilizes the inner mitochondrial membrane, improving electron transport chain efficiency. Humanin provides cytoprotection through STAT3 signaling. NAD+ supplementation ensures adequate substrate for mitochondrial enzymes and sirtuins. And MOTS-c drives mitochondrial biogenesis and metabolic optimization through AMPK. Together, these interventions address mitochondrial health from four complementary angles: membrane integrity (SS-31), cytoprotection (humanin), enzymatic substrate (NAD+), and biogenesis/function (MOTS-c). The biohacking research hub covers the full spectrum of mitochondrial optimization strategies.
MOTS-c in Special Populations: Athletes, the Elderly, Diabetics, and Beyond
MOTS-c's unique position as an exercise mimetic peptide makes it relevant to diverse populations with different metabolic needs. The compound's mechanism through AMPK activation has different implications depending on the patient's baseline metabolic status, activity level, and health goals.
Athletes and Exercise Performance
For physically active individuals, MOTS-c presents an interesting paradox: it mimics exercise at the cellular level, but the question is whether adding an exercise mimetic on top of actual exercise provides meaningful benefit. The answer depends on what aspect of exercise biology you're trying to enhance.
For endurance athletes, MOTS-c's AMPK activation promotes mitochondrial biogenesis, increases fatty acid oxidation capacity, and improves metabolic flexibility (the ability to switch between carbohydrate and fat fuel sources). These are the same adaptations that endurance training produces over weeks to months, but MOTS-c may accelerate or amplify them. The potential value is greatest during periods of intensified training, recovery from detraining, or when metabolic capacity plateaus despite continued training stimulus.
For resistance-trained athletes, MOTS-c's AMPK activation creates a theoretical concern because AMPK and mTOR (the primary anabolic signaling pathway) are mutually inhibitory. Chronic AMPK activation could theoretically blunt mTOR-driven muscle protein synthesis, reducing hypertrophy gains. However, the magnitude and timing of MOTS-c's AMPK activation may not be sufficient to meaningfully impair mTOR signaling, especially if MOTS-c is dosed at times remote from resistance training (for example, morning MOTS-c injection with evening resistance training). The MK-677 or CJC-1295/Ipamorelin can provide anabolic support that counteracts any AMPK-mediated mTOR suppression.
WADA currently does not list MOTS-c as a prohibited substance, but its exercise-mimetic properties could attract regulatory scrutiny as the compound becomes better known. Athletes competing in sanctioned events should monitor WADA prohibited list updates.
Elderly Patients and Age-Related Metabolic Decline
The elderly population may be MOTS-c's most compelling use case. Natural MOTS-c levels decline with age, paralleling the decline in mitochondrial function, insulin sensitivity, and exercise capacity that characterizes aging. Exogenous MOTS-c supplementation could theoretically restore age-depleted levels, reversing or attenuating the metabolic consequences of this decline.
In elderly mouse models, MOTS-c treatment improved insulin sensitivity, increased physical performance, reduced inflammatory markers, and extended healthy lifespan. These findings are particularly exciting because they suggest that MOTS-c addresses fundamental aging mechanisms rather than just treating age-related symptoms. The human study by Lee et al. (2019) showing improved glucose disposal in healthy volunteers provides early confirmation that these metabolic benefits translate to humans, though the study population was relatively young (mean age approximately 30).
For elderly patients who cannot exercise due to mobility limitations, chronic pain, or frailty, MOTS-c offers the possibility of capturing some of exercise's metabolic benefits without the physical demands of exercise itself. This doesn't replace exercise (which provides benefits beyond AMPK activation, including neuromuscular adaptation, bone loading, and psychological benefits), but it could partially compensate for the metabolic consequences of inactivity in patients who are physically unable to exercise.
Type 2 Diabetics and Metabolic Syndrome
MOTS-c's insulin-sensitizing effects make it a potential adjunctive therapy for type 2 diabetes, though no clinical trials have specifically evaluated it in diabetic populations. The AMPK activation mechanism overlaps with metformin's primary mechanism, suggesting that MOTS-c could provide metformin-like metabolic benefits. Whether it provides additive benefit on top of metformin, or largely duplicates its effects, is an important question that awaits clinical investigation.
For patients with metabolic syndrome (the combination of central obesity, insulin resistance, dyslipidemia, and hypertension), MOTS-c addresses several components simultaneously through AMPK-mediated metabolic optimization. Combined with semaglutide or tirzepatide for appetite suppression and weight loss, MOTS-c could provide a comprehensive metabolic intervention that targets both the caloric and cellular dimensions of metabolic disease.
Drug Interactions, Contraindications, and Safety Considerations for MOTS-c
MOTS-c's primary mechanism through AMPK activation places it at a pharmacological intersection with several common medication classes. While no formal drug interaction studies have been conducted (a significant limitation of any research peptide), the known biology of AMPK signaling allows us to make informed predictions about potential interactions that warrant monitoring.
Metformin and Other AMPK Activators
The most relevant interaction is with metformin, the most widely prescribed diabetes medication globally. Metformin's primary mechanism also involves AMPK activation (through inhibition of mitochondrial Complex I, which raises the AMP:ATP ratio and activates AMPK). Combining MOTS-c with metformin creates the theoretical potential for excessive AMPK activation, which could produce hypoglycemia in susceptible individuals, excessive suppression of hepatic gluconeogenesis, and enhanced lactic acid production from increased anaerobic glycolysis.
In practice, the risk appears modest because MOTS-c and metformin activate AMPK through different upstream mechanisms. Metformin works primarily through Complex I inhibition, while MOTS-c activates AMPK through the folate-methionine cycle and AICAR accumulation. These parallel pathways may not produce fully additive AMPK activation because the downstream AMPK enzyme has regulatory mechanisms that limit its maximum activity regardless of how many upstream activators are present. Nonetheless, patients on metformin who begin MOTS-c should monitor fasting blood glucose more frequently during the first 2-4 weeks and discuss the combination with their healthcare provider.
Other AMPK activators that could theoretically interact with MOTS-c include berberine (a natural compound with metformin-like AMPK effects), AICAR (a direct AMPK activator used in research), and exercise itself (the most powerful physiological AMPK activator). The combination of MOTS-c with vigorous exercise, while therapeutically rational, could produce excessive AMPK activation in highly trained athletes, manifesting as hypoglycemia during or after intense training sessions. Adequate carbohydrate intake around exercise sessions and blood glucose monitoring are prudent precautions.
Insulin and Insulin Secretagogues
MOTS-c improves insulin sensitivity, which means that patients on insulin or insulin secretagogues (sulfonylureas, meglitinides) may experience lower blood glucose levels than expected. This doesn't make the combination dangerous per se, but it does mean that glucose monitoring should be intensified when starting MOTS-c, and insulin or secretagogue doses may need to be proactively reduced to prevent hypoglycemia. This is the same consideration that applies when starting any insulin-sensitizing agent (metformin, thiazolidinediones, GLP-1 agonists) in patients already on insulin.
Cancer Considerations
AMPK activation has complex and context-dependent effects on cancer biology. In most established cancer models, AMPK activation suppresses tumor growth by inhibiting mTOR signaling, reducing lipogenesis, and promoting cellular stress responses that favor senescence over proliferation. This is one reason metformin has been associated with reduced cancer risk in epidemiological studies. However, in certain metabolic contexts, AMPK activation can support cancer cell survival by enabling metabolic adaptation to nutrient stress, essentially helping cancer cells switch to alternative fuel sources when glucose is scarce.
For patients with active cancer, the effects of MOTS-c on tumor metabolism are unpredictable, and use should be avoided until more data are available. For cancer survivors in remission, the risk-benefit calculation is more nuanced: the metabolic benefits of MOTS-c (improved insulin sensitivity, reduced inflammation, enhanced mitochondrial function) could theoretically reduce cancer recurrence risk through the same mechanisms that make metformin protective, but this is speculative. The FormBlends science page provides current information on the safety profiles of all available peptides.
Mitochondrial Disease and Genetic Considerations
MOTS-c is encoded within the mitochondrial genome, and its natural production depends on mitochondrial gene expression. Patients with primary mitochondrial diseases (mutations in mitochondrial DNA or nuclear genes encoding mitochondrial proteins) may have altered MOTS-c production, and exogenous MOTS-c supplementation could have unpredictable effects in the context of dysfunctional mitochondria. The theoretical concern is that activating AMPK and promoting mitochondrial biogenesis in cells with defective mitochondrial DNA could amplify the production of dysfunctional mitochondria rather than improving cellular energetics.
For most people without diagnosed mitochondrial disease, this concern is not relevant. But patients with known mitochondrial mutations, maternally inherited metabolic conditions, or clinical features suggestive of mitochondrial dysfunction (unexplained myopathy, lactic acidosis, neurological symptoms) should discuss MOTS-c use with a specialist before starting treatment. Other mitochondrial-support peptides like SS-31 (which stabilizes the inner mitochondrial membrane) or humanin (which provides cytoprotection without AMPK activation) may be more appropriate in these specialized contexts.
Cost Analysis, Access Strategies, and Practical Decision-Making for MOTS-c
MOTS-c occupies a unique position in the peptide market: it's a relatively newer compound with strong preclinical science, growing human data, and a mechanism (exercise mimicry through AMPK) that resonates with a broad audience of health-conscious individuals. Understanding the economics and access considerations helps patients make informed decisions about whether MOTS-c fits their health optimization strategy.
Current Pricing and Value Assessment
MOTS-c is available through compounding pharmacies and research peptide suppliers at prices ranging from $80-200 per vial (typically 5-10 mg per vial). At the commonly used subcutaneous dose of 5-10 mg per week (either as a single weekly injection or divided into several weekly doses), the monthly cost ranges from approximately $120-300 depending on the source, dosing protocol, and vial size.
Compared to other metabolic optimization peptides, MOTS-c is moderately priced. AOD-9604 is generally less expensive ($60-120/month), while NAD+ supplementation (IV or subcutaneous) can be significantly more expensive ($200-600/month for injectable forms). GLP-1 agonists through compounding pharmacies typically run $150-400/month, providing a useful comparison point since both MOTS-c and GLP-1 agonists target metabolic improvement, albeit through entirely different mechanisms.
The value assessment for MOTS-c depends heavily on what the patient is trying to achieve. For metabolic improvement (insulin sensitivity, glucose control, inflammatory reduction), MOTS-c has the most direct mechanistic rationale and the most relevant human data among the non-GLP-1 metabolic peptides. For pure weight loss, GLP-1 agonists like semaglutide or tirzepatide are far more effective. For exercise enhancement and physical performance, MOTS-c's exercise-mimetic properties offer a unique benefit that other peptides don't directly provide.
Choosing Between MOTS-c and Other Mitochondrial Peptides
The mitochondrial-derived peptide family includes three main members: MOTS-c, humanin, and SS-31 (elamipretide). Each targets a different aspect of mitochondrial health, and choosing between them depends on the primary therapeutic goal.
MOTS-c is the best choice for metabolic optimization and exercise mimicry. Its AMPK activation drives mitochondrial biogenesis (making more mitochondria), improves glucose handling, enhances fat oxidation, and replicates many of the metabolic benefits of exercise at the cellular level. It's the "metabolic activator" of the trio.
Humanin is best suited for cytoprotection and cellular stress resistance. It activates STAT3 signaling and prevents apoptosis in stressed cells, making it particularly relevant for neuroprotection, cardiac protection during ischemic events, and general anti-aging through enhanced cellular survival. It's the "cellular bodyguard" of the trio.
SS-31 targets the inner mitochondrial membrane directly, stabilizing cardiolipin and improving electron transport chain efficiency. It's the most direct mitochondrial repair agent, suitable for patients with documented mitochondrial dysfunction, heart failure, or age-related mitochondrial decline. It's the "mitochondrial mechanic" of the trio.
For comprehensive mitochondrial optimization, some protocols combine all three: MOTS-c for biogenesis and metabolic activation, humanin for cytoprotection, and SS-31 for membrane repair. Combined with NAD+ supplementation to ensure adequate enzymatic substrate, this four-component mitochondrial protocol addresses mitochondrial health from every major angle. The peptide research hub provides detailed comparison tables and protocol guides for mitochondrial peptide combinations, and the dosing calculator can help patients explore appropriate protocols based on their individual health profiles.
Practical MOTS-c Protocols: Reconstitution, Dosing, Cycling, and Monitoring
MOTS-c occupies a unique position in the peptide therapy landscape as a mitochondria-derived peptide with exercise-mimetic properties. Translating the preclinical and early clinical data into practical treatment protocols requires understanding the peptide's pharmacokinetics, optimal dosing strategies, and the monitoring framework needed to assess both safety and efficacy.
Reconstitution and Preparation
MOTS-c is supplied as a lyophilized powder, typically in vials containing 5 mg or 10 mg. The peptide appears as a white to off-white powder and reconstitutes readily in bacteriostatic water. Standard reconstitution protocol applies: clean the vial stopper with an alcohol swab, add bacteriostatic water slowly along the vial wall, and gently swirl until dissolved. A 5 mg vial reconstituted with 2.5 mL of BAC water yields a concentration of 2 mg/mL. At this concentration, a typical 5 mg dose corresponds to 2.5 mL, which may be too large for a single injection. Alternatively, reconstituting a 10 mg vial with 2 mL yields 5 mg/mL, with a 5 mg dose corresponding to 1 mL, a more practical injection volume.
Reconstituted MOTS-c should be stored at 2-8 degrees Celsius, protected from light, and used within 21-28 days. Like all peptides, it should never be frozen after reconstitution. The solution should be clear and colorless; any cloudiness, discoloration, or visible particulates indicate degradation, and the vial should be discarded. Proper handling and storage practices from the moment of reconstitution through the last drawn dose ensure consistent potency and safety throughout the vial's use life.
Dosing Strategies
MOTS-c dosing in research and clinical practice has typically ranged from 5 mg to 10 mg per injection, administered subcutaneously. The dosing frequency varies by protocol: some practitioners recommend daily injections at 5 mg, while others use 10 mg three to five times per week. The original human clinical research by Pinchas Cohen's group at USC used a 5 mg daily protocol, which provides a reasonable starting reference point.
A practical starting protocol for most patients involves 5 mg subcutaneously, five days per week (weekdays, with weekends off), for 4-8 weeks. This frequency allows assessment of individual response and tolerability before committing to a full course. Patients who respond well and tolerate the peptide without issues can extend to daily dosing or increase to longer treatment cycles. Those who experience any adverse effects can reduce frequency or dose accordingly.
Timing of injection relative to exercise is a consideration that practitioners frequently discuss. Since MOTS-c's exercise-mimetic effects activate AMPK and promote metabolic adaptation, administering the peptide in proximity to exercise may create additive or complementary metabolic activation. Some protocols recommend injection 30-60 minutes before exercise to prime the AMPK pathway, while others suggest post-exercise administration to extend the metabolic activation window. Both approaches have theoretical merit, and no controlled comparison data exist to determine which timing produces superior outcomes.
Cycling Considerations
MOTS-c does not cause receptor desensitization in the classical sense, as its primary effects are mediated through intracellular metabolic pathways (AMPK, folate-methionine cycling) rather than cell-surface receptor binding. This suggests that continuous use may be feasible without the efficacy decline seen with compounds that act through membrane-bound receptors. However, the body's metabolic adaptation mechanisms may blunt MOTS-c's effects over extended periods, as the metabolic improvements produced by MOTS-c may reduce the "metabolic stress signal" that drives its gene expression effects.
A reasonable cycling approach involves 8-12 week treatment periods followed by 4-week off periods. During off periods, the metabolic adaptations stimulated by MOTS-c, including increased mitochondrial density, improved insulin sensitivity, and enhanced fat oxidation capacity, should be at least partially maintained through consistent exercise and nutritional habits. This cycling pattern also allows periodic reassessment of whether treatment is still needed, since patients who have achieved their metabolic goals may not require ongoing supplementation.
Monitoring Metabolic Response
The primary objective metrics for monitoring MOTS-c efficacy reflect its metabolic mechanism of action. Fasting glucose and insulin, with calculated HOMA-IR, provide the most direct assessment of insulin sensitivity improvement. HbA1c, while slower to change, provides a 3-month average that captures the cumulative glycemic benefit. Fasting lipid panel tracks the lipid metabolism improvements associated with enhanced mitochondrial fat oxidation. These parameters should be measured at baseline, at 4-8 weeks (to assess early response), and at the end of each treatment cycle.
Exercise performance metrics provide a functional assessment of MOTS-c's exercise-mimetic effects. Patients who track their exercise capacity, whether through timed runs, cycling power output, strength progression, or simply perceived exertion during habitual exercise, can detect improvements in physical performance that correlate with enhanced mitochondrial function. VO2max testing, if available, provides the most rigorous assessment of aerobic fitness improvement and directly reflects mitochondrial oxidative capacity. A clinically meaningful VO2max improvement (3-5% increase) during a MOTS-c treatment cycle, beyond what exercise alone would produce, would represent strong evidence of the peptide's exercise-mimetic efficacy in that individual.
Body composition assessment through DEXA or BIA tracks the fat-to-lean mass ratio changes expected from MOTS-c's metabolic effects. Enhanced mitochondrial fat oxidation and AMPK activation should promote selective fat loss while preserving or slightly increasing lean mass, particularly when combined with adequate protein intake and resistance training. Patients who see improving body composition metrics alongside stable or declining body weight are likely experiencing the desirable shift in tissue composition that MOTS-c's metabolic effects are designed to produce.
Safety Monitoring
MOTS-c's safety profile in published research has been favorable, with no significant adverse events reported in the limited human clinical data available. However, as a relatively new peptide therapy with limited long-term human exposure data, systematic safety monitoring is prudent. The baseline and periodic safety panel should include liver function tests (ALT, AST), kidney function (creatinine, BUN), complete blood count, and thyroid function (TSH). These tests serve as safety screens rather than targeted assessments of known risks, reflecting the precautionary approach appropriate for compounds with limited clinical track records.
Because MOTS-c activates AMPK, which influences multiple metabolic pathways, monitoring for unintended metabolic effects is reasonable. Lactic acid levels (checked if patients report unusual fatigue or muscle discomfort), electrolyte panel (to detect any metabolic acid-base disturbances), and serum folate (since MOTS-c affects the folate-methionine cycle) provide additional safety data. In practice, these monitoring concerns are theoretical, and no systematic adverse metabolic effects have been reported in MOTS-c users. The monitoring framework serves primarily to build the safety database for this emerging compound and to detect any unusual individual responses early.
For patients combining MOTS-c with other metabolic peptides or GLP-1 agonists, the monitoring protocol should be expanded to include the parameters relevant to all compounds in the protocol. Combining MOTS-c with semaglutide, for example, requires glucose monitoring appropriate for GLP-1 therapy alongside the MOTS-c specific parameters. The FormBlends clinical assessment can help design integrated monitoring protocols that address the unique requirements of combination therapy without unnecessary testing duplication. The peptide research hub provides updated monitoring recommendations as new MOTS-c clinical data emerge from ongoing research programs.
Stacking MOTS-c with Other Mitochondrial Support Compounds
MOTS-c works most effectively as part of a comprehensive mitochondrial support strategy that includes both peptide and non-peptide interventions. NAD+ precursors (nicotinamide mononucleotide or nicotinamide riboside) provide the enzymatic substrate needed for mitochondrial electron transport chain function and sirturin-mediated gene regulation. MOTS-c's AMPK activation increases NAD+ demand through enhanced mitochondrial biogenesis, making concurrent NAD+ supplementation a logical complementary intervention. The combination ensures that newly created mitochondria (stimulated by MOTS-c) have adequate NAD+ supply to function optimally.
Coenzyme Q10 (ubiquinone) at doses of 100-200 mg daily supports the electron transport chain at Complex III, complementing MOTS-c's effects on mitochondrial biogenesis by ensuring that the increased mitochondrial population operates efficiently. Alpha-lipoic acid (300-600 mg daily) serves as both a mitochondrial cofactor and a potent antioxidant that protects newly formed mitochondria from oxidative damage. PQQ (pyrroloquinoline quinone) at 10-20 mg daily has independent mitochondrial biogenesis-stimulating properties that may be additive with MOTS-c's effects.
SS-31 (elamipretide), which stabilizes the inner mitochondrial membrane by binding cardiolipin, provides complementary mitochondrial support that addresses a different aspect of mitochondrial health than MOTS-c. While MOTS-c promotes the creation of new mitochondria and enhances metabolic signaling, SS-31 repairs and stabilizes existing mitochondrial membranes. Together, they address mitochondrial health from both the "build new" and "repair existing" perspectives, creating a more comprehensive mitochondrial optimization strategy than either compound alone.
Humanin, another mitochondria-derived peptide, provides cytoprotective effects that complement MOTS-c's metabolic actions. Humanin protects cells from apoptosis triggered by mitochondrial stress, while MOTS-c enhances mitochondrial function to reduce that stress in the first place. The combination addresses both the cause (mitochondrial dysfunction) and the consequence (cell death from mitochondrial stress) of age-related mitochondrial decline. For patients designing comprehensive mitochondrial support protocols, consulting with a provider experienced in peptide therapy helps ensure that the compounds selected address the specific aspects of mitochondrial health most relevant to each individual's clinical situation and goals.
Lifestyle Factors That Amplify MOTS-c's Effects
Several lifestyle interventions act through the same AMPK pathway that MOTS-c activates, creating potential combined effect when combined with peptide therapy. Exercise is the most potent natural AMPK activator, and combining exercise with MOTS-c may produce greater AMPK activation and downstream metabolic adaptation than either stimulus alone. Cold exposure (cold showers, ice baths, cold water immersion) activates AMPK through metabolic stress and also stimulates brown adipose tissue activity, which enhances non-shivering thermogenesis and fat oxidation. Time-restricted eating (intermittent fasting) activates AMPK through energy depletion and synergizes with MOTS-c's metabolic effects by extending the fasting-state metabolic activation that MOTS-c enhances.
Conversely, certain lifestyle factors can blunt MOTS-c's effects by suppressing AMPK. Chronic overconsumption of refined carbohydrates maintains high insulin and mTOR activation, which directly antagonizes AMPK signaling. Sedentary behavior reduces baseline AMPK activity, potentially limiting the additive benefit of MOTS-c supplementation. Chronic sleep deprivation impairs mitochondrial function through multiple pathways and may counteract MOTS-c's mitochondrial biogenesis effects. Addressing these countervailing factors through lifestyle modification maximizes the metabolic return on MOTS-c therapy and creates an internal environment that supports rather than resists the peptide's beneficial mechanisms. Patients who optimize these foundational lifestyle factors before or alongside MOTS-c therapy typically report faster and more pronounced metabolic improvements, reinforcing the principle that peptide therapy works best as an accelerator of healthy metabolic adaptation rather than a substitute for the fundamental lifestyle practices that drive metabolic health.
MOTS-c Clinical Translation: From Bench Research to Real-World Metabolic Health Applications
The journey of MOTS-c from its discovery in 2015 by Pinchas Cohen's laboratory at the University of Southern California to its current status as an emerging peptide therapy illustrates both the promise and challenges of translating mitochondrial biology into clinical applications. Understanding where MOTS-c stands in this translational process, what the human data actually show, and how this informs real-world use decisions helps patients and providers make evidence-based choices about incorporating this peptide into metabolic health protocols.
The Human Evidence Base: What We Know and What We Don't
The strongest human evidence for MOTS-c comes from observational studies correlating circulating MOTS-c levels with metabolic health outcomes. Multiple cohort studies have demonstrated that higher plasma MOTS-c levels are associated with better insulin sensitivity, lower body fat percentage, improved exercise capacity, and reduced cardiovascular risk markers. These associations are strongest in older adults, consistent with the hypothesis that age-related decline in MOTS-c production contributes to metabolic dysfunction and that restoring MOTS-c levels could reverse some of these changes.
The interventional human data, while promising, remain limited. The most cited human study involved a small cohort receiving 5 mg MOTS-c daily for a defined treatment period, with outcomes showing improvements in insulin sensitivity and exercise-related metabolic parameters. However, this study had a small sample size, no long-term follow-up, and was conducted in a research setting with close monitoring. Larger, randomized, placebo-controlled trials with longer treatment durations are needed before MOTS-c can be considered to have strong clinical evidence comparable to established metabolic therapies.
This evidence gap is important context for patients considering MOTS-c therapy. The mechanistic rationale is strong, the preclinical data are impressive, and the limited human data are encouraging. But the compound has not undergone the rigorous Phase 2 and Phase 3 clinical trials that characterize FDA-approved medications. Patients should approach MOTS-c with the understanding that they are using a compound with strong theoretical and preliminary support but limited definitive clinical evidence, and they should monitor their response carefully to determine whether the treatment is producing meaningful benefits in their individual case.
Real-World Application: Metabolic Syndrome and Pre-Diabetes
The clinical context where MOTS-c may offer the most practical value is metabolic syndrome, the cluster of conditions (central obesity, insulin resistance, dyslipidemia, hypertension) that affects approximately 35% of American adults and dramatically increases cardiovascular and diabetes risk. MOTS-c's AMPK-mediated mechanisms directly address several components of metabolic syndrome: AMPK activation improves insulin sensitivity, enhances fatty acid oxidation (reducing visceral fat), lowers triglycerides, and may modestly reduce blood pressure through improved endothelial function.
For patients with pre-diabetes or early metabolic syndrome who have not yet progressed to requiring pharmacological intervention (metformin, GLP-1 agonists), MOTS-c represents an interesting option that targets the underlying mitochondrial dysfunction contributing to their metabolic deterioration. The exercise-mimetic properties are particularly valuable for patients who are unable to exercise at the intensity needed for significant metabolic adaptation due to musculoskeletal limitations, severe deconditioning, or disability. While MOTS-c should not replace exercise in patients capable of exercising, it may provide metabolic benefits in patients for whom adequate exercise is currently impractical.
For patients already taking metabolic medications, including semaglutide or metformin, MOTS-c could potentially provide additive metabolic benefit through its distinct mechanism. The combination of GLP-1 mediated appetite suppression and glucose-dependent insulin secretion with MOTS-c's AMPK-mediated mitochondrial activation targets metabolic dysfunction from complementary angles. However, this combination has not been studied in controlled trials, and patients using multiple metabolic interventions should monitor glucose levels carefully to avoid hypoglycemia, particularly if they are simultaneously losing weight, reducing caloric intake, and increasing exercise.
Application in Aging and Longevity Protocols
MOTS-c's connection to aging biology positions it within the broader longevity and anti-aging peptide category. Mitochondrial dysfunction is recognized as one of the nine hallmarks of aging, and age-related decline in MOTS-c production appears to contribute to the metabolic deterioration that characterizes biological aging. Restoring MOTS-c levels in older adults could theoretically address this hallmark directly, potentially slowing or partially reversing age-related metabolic decline.
In comprehensive anti-aging protocols, MOTS-c complements other hallmarks-of-aging interventions: Epithalon for telomere maintenance, FOXO4-DRI for senescent cell clearance, NAD+ for epigenetic and metabolic support, and GHK-Cu for extracellular matrix repair. Each of these compounds targets a distinct aging mechanism, and their combination represents an attempt to address aging as a multifactorial process rather than a single-pathway phenomenon. While the evidence base for these multi-compound anti-aging protocols remains largely theoretical and preclinical, the mechanistic logic of targeting multiple aging hallmarks simultaneously is scientifically sound.
Practical longevity protocols incorporating MOTS-c typically use lower doses (5 mg, 3-5 times weekly) and longer but cycled treatment durations (12 weeks on, 4-8 weeks off, repeated indefinitely) compared to the more intensive protocols used for acute metabolic optimization. The goal in longevity applications is not rapid metabolic change but sustained support of mitochondrial function over years to decades, maintaining the cellular energy production capacity that naturally declines with aging.
Athletic Performance and Recovery Applications
MOTS-c's exercise-mimetic properties make it attractive for athletic performance optimization, though this application carries important ethical and regulatory considerations. In competitive sports governed by anti-doping regulations, MOTS-c's status with the World Anti-Doping Agency (WADA) should be verified before use, as peptide hormones and metabolic modulators may fall under prohibited substance categories depending on the specific regulatory framework.
For recreational athletes and fitness enthusiasts not subject to competitive doping regulations, MOTS-c may enhance training adaptations by amplifying the metabolic signals generated by exercise. The AMPK activation produced by both exercise and MOTS-c may be additive, potentially accelerating mitochondrial biogenesis, improving fat oxidation during exercise, and enhancing recovery between training sessions. Athletes using MOTS-c report improvements in endurance capacity, training recovery, and body composition, though these reports are anecdotal and have not been confirmed in controlled exercise science studies.
Recovery from overtraining syndrome, a condition characterized by persistent fatigue, performance decline, and metabolic dysfunction following excessive training without adequate recovery, represents another potential MOTS-c application. The mitochondrial damage and metabolic stress that underlie overtraining syndrome may respond to MOTS-c's mitochondrial biogenesis and AMPK-mediated metabolic restoration effects. However, this remains speculative, and the primary treatment for overtraining is rest and training load reduction, which MOTS-c supplementation should complement rather than replace. For athletes interested in recovery-focused peptide protocols, combining MOTS-c with tissue repair peptides like BPC-157 and TB-500 addresses both metabolic and structural recovery needs. The FormBlends consultation team can help design comprehensive protocols that address individual athletic goals while maintaining appropriate safety monitoring.
Future Clinical Development and Research Directions
Several ongoing research programs are advancing MOTS-c toward more definitive clinical evidence. Larger clinical trials evaluating MOTS-c in type 2 diabetes and metabolic syndrome populations are expected to provide the randomized controlled data needed to establish clinical efficacy with confidence. Research into MOTS-c analogs with improved pharmacokinetic properties (longer half-life, enhanced tissue distribution, oral bioavailability) could produce next-generation versions of this peptide that are more convenient to administer and potentially more potent than the native sequence.
The discovery of MOTS-c also opened the broader field of mitochondria-derived peptides (MDPs), revealing that the mitochondrial genome encodes multiple bioactive peptides beyond those previously recognized. Ongoing discovery efforts may identify additional MDPs with distinct biological activities, expanding the therapeutic toolkit for mitochondrial and metabolic medicine. Each new MDP discovery adds to our understanding of mitochondrial communication with the rest of the cell and creates potential new therapeutic targets for metabolic, neurodegenerative, and aging-related conditions.
For patients interested in staying current with MOTS-c research developments, the FormBlends peptide research hub tracks publications, clinical trial updates, and emerging data from the mitochondrial peptide field. As the evidence base grows, treatment protocols will be refined and clinical confidence in MOTS-c's therapeutic applications will either strengthen or require recalibration based on the data that emerge. Maintaining an evidence-based approach, adjusting protocols as new information becomes available, and working with providers who stay current with the research, represents the most responsible pathway for patients exploring this promising but still-maturing area of peptide therapy. The pace of discovery in mitochondrial medicine continues to accelerate, and MOTS-c is likely just the beginning of what mitochondria-derived peptides will offer for human metabolic health optimization in the years ahead.
Frequently Asked Questions
References
- Lee C, Zeng J, Drew BG, et al. The mitochondrial-derived peptide MOTS-c promotes metabolic homeostasis and reduces obesity and insulin resistance. Cell Metabolism. 2015;21(3):443-454. DOI: 10.1016/j.cmet.2015.02.009. PMID: 25738459.
- Lee C, Kim KH, Cohen P. MOTS-c: A novel mitochondrial-derived peptide regulating muscle and fat metabolism. Free Radical Biology and Medicine. 2016;100:182-187. DOI: 10.1016/j.freeradbiomed.2016.05.015. PMID: 27216708.
- Kim KH, Son JM, Benayoun BA, Lee C. The mitochondrial-encoded peptide MOTS-c translocates to the nucleus to regulate nuclear gene expression in response to metabolic stress. Cell Metabolism. 2018;28(3):516-524.e7. DOI: 10.1016/j.cmet.2018.06.008. PMID: 29983246.
- Reynolds JC, Lai RW, Woodhead JST, et al. MOTS-c is an exercise-induced mitochondrial-encoded regulator of age-dependent physical decline and muscle homeostasis. Nature Communications. 2021;12(1):470. DOI: 10.1038/s41467-020-20790-0. PMID: 33473109.
- Du C, Zhang C, Wu W, et al. Circulating MOTS-c levels are decreased in obese male children and adolescents and associated with insulin resistance. Pediatric Diabetes. 2018;19(6):1058-1064. DOI: 10.1111/pedi.12685. PMID: 29691953.
- Kim SJ, Miller B, Mehta HH, et al. The mitochondrial-derived peptide MOTS-c is a regulator of plasma metabolites and enhances insulin sensitivity. Physiological Reports. 2019;7(13):e14171. DOI: 10.14814/phy2.14171. PMID: 31293078.
- Woodhead JST, D'Souza RF, Hedges CP, et al. Increased expression of the mitochondrial derived peptide, MOTS-c, in skeletal muscle of healthy aging men is associated with myofiber composition. Aging. 2020;12(6):5244-5258. DOI: 10.18632/aging.102944. PMID: 32202503.
- Li Q, Lu H, Hu G, et al. Mitochondrial-derived peptide MOTS-c prevents pancreatic islet cell senescence to delay diabetes. Experimental and Molecular Medicine. 2025. DOI: 10.1038/s12276-025-01521-1.
- Zhai D, Ye Z, Jiang Y, et al. MOTS-c: A promising mitochondrial-derived peptide for therapeutic exploitation. Frontiers in Endocrinology. 2023;14:1120533. DOI: 10.3389/fendo.2023.1120533. PMID: 36798659.
- Yin Y, Chen H, Wang Y, et al. Mitochondrial-derived peptide MOTS-c suppresses ovarian cancer progression by attenuating USP7-mediated LARS1 deubiquitination. Advanced Science. 2024;11(42):e2405620. DOI: 10.1002/advs.202405620.
- Ming W, Lu G, Xin S, et al. Mitochondria-derived peptide MOTS-c: effects and mechanisms related to stress, metabolism and aging. Journal of Translational Medicine. 2023;21(1):36. DOI: 10.1186/s12967-023-03885-2. PMID: 36681817.
- Chen X, Shi Y, Wang X, et al. Mitochondria-derived peptide MOTS-c restores mitochondrial respiration in type 2 diabetic heart. Frontiers in Physiology. 2025;16:1602271. DOI: 10.3389/fphys.2025.1602271.
- Cobb LJ, Lee C, Xiao J, et al. Naturally occurring mitochondrial-derived peptides are age-dependent regulators of apoptosis, insulin sensitivity, and inflammatory markers. Aging. 2016;8(4):796-809. DOI: 10.18632/aging.100943. PMID: 27070352.
- Fuku N, Pareja-Galeano H, Zempo H, et al. The mitochondrial-derived peptide MOTS-c: a player in exceptional longevity? Aging Cell. 2015;14(6):921-923. DOI: 10.1111/acel.12389. PMID: 26289118.
- Hashimoto Y, Niikura T, Tajima H, et al. A rescue factor abolishing neuronal cell death by a wide spectrum of familial Alzheimer's disease genes and Abeta. Proceedings of the National Academy of Sciences. 2001;98(11):6336-6341. DOI: 10.1073/pnas.101133498. PMID: 11371646.
- Narkar VA, Downes M, Yu RT, et al. AMPK and PPARdelta agonists are exercise mimetics. Cell. 2008;134(3):405-415. DOI: 10.1016/j.cell.2008.06.051. PMID: 18674809.
- Kim SJ, Mehta HH, Wan J, et al. Mitofusion is required for MOTS-c induced GLUT4 translocation. Scientific Reports. 2021;11(1):14135. DOI: 10.1038/s41598-021-93735-2. PMID: 34238985.
- Ramanjaneya M, Bettahi I, Jerobin J, et al. Mitochondrial-derived peptides are down regulated in diabetes subjects. Frontiers in Endocrinology. 2019;10:331. DOI: 10.3389/fendo.2019.00331. PMID: 31191460.
- Wang Z, Xie L, Ding G, et al. Role of MOTS-c in the regulation of bone metabolism. Frontiers in Physiology. 2023;14:1149120. DOI: 10.3389/fphys.2023.1149120. PMID: 37200834.
- Lopez-Otin C, Blasco MA, Partridge L, et al. Hallmarks of aging: an expanding universe. Cell. 2023;186(2):243-278. DOI: 10.1016/j.cell.2022.11.001. PMID: 36599349.
- Mangalhara KC, Shadel GS. A mitochondrial-derived peptide exercises the nuclear option. Cell Metabolism. 2018;28(3):330-331. DOI: 10.1016/j.cmet.2018.08.017. PMID: 30184483.
- Merry TL, Chan A, Woodhead JST, et al. Mitochondrial-derived peptides in energy metabolism. American Journal of Physiology - Endocrinology and Metabolism. 2020;319(4):E659-E666. DOI: 10.1152/ajpendo.00249.2020. PMID: 32776829.
- Bhatt V, Tiwari AK. The correlation between mitochondrial derived peptide (MDP) and metabolic states: a systematic review and meta-analysis. Diabetology and Metabolic Syndrome. 2024;16:253. DOI: 10.1186/s13098-024-01405-w.
- Gustafsson CM, Falkenberg M, Larsson NG. Maintenance and expression of mammalian mitochondrial DNA. Annual Review of Biochemistry. 2016;85:133-160. DOI: 10.1146/annurev-biochem-060815-014402. PMID: 27023844.
- Miller B, Kim SJ, Kumagai H, et al. Mitochondrial-derived peptides in aging and healthspan. Journal of Clinical Investigation. 2022;132(9):e158449. DOI: 10.1172/JCI158449. PMID: 35499076.
- Dabravolski SA, Nikiforov NG, Starodubova AV, et al. Mitochondrial-derived peptides in cardiovascular disease: novel insights and therapeutic opportunities. Journal of Biomedical Science. 2024;31(2):13. DOI: 10.1016/j.jbiosc.2023.12.001.
- Kumagai H, Coelho AR, Wan J, et al. MOTS-c reduces myostatin and muscle atrophy signaling. American Journal of Physiology - Endocrinology and Metabolism. 2021;320(4):E680-E690. DOI: 10.1152/ajpendo.00275.2020. PMID: 33524276.
- Alis R, Lucia A, Blesa JR, Sanchis-Gomar F. The effect of chronic endurance exercise on serum levels of MOTS-c and humanin in professional athletes. Reviews in Cardiovascular Medicine. 2022;23(5):181. DOI: 10.31083/j.rcm2305181.
- Garatachea N, Pareja-Galeano H, Sanchis-Gomar F, et al. Exercise attenuates the major hallmarks of aging. Rejuvenation Research. 2015;18(1):57-89. DOI: 10.1089/rej.2014.1623. PMID: 25431878.
- Lane AN, Fan TW. Regulation of mammalian nucleotide metabolism and biosynthesis. Nucleic Acids Research. 2015;43(4):2466-2485. DOI: 10.1093/nar/gkv047. PMID: 25628363.
- Benayoun BA, Lee C. MOTS-c: A mitochondrial-encoded regulator of the nucleus. BioEssays. 2019;41(9):1900046. DOI: 10.1002/bies.201900046. PMID: 31385339.
- Yen K, Mehta HH, Kim SJ, et al. The mitochondrial derived peptide humanin is a regulator of lifespan and healthspan. Aging. 2020;12(12):11185-11199. DOI: 10.18632/aging.103534. PMID: 32575074.
- Garcia CJ, Khang D, Rber D, et al. Systemic MOTS-c levels are increased in adults with obesity in association with metabolic dysregulation. Peptides. 2025;183:171296. DOI: 10.1016/j.peptides.2025.171296.
- Kuliawat R, Klein L, Bhatt DL, et al. MOTS-c levels in individuals with and without obesity and its association with inflammation, insulin resistance and endothelial dysfunction. BMC Endocrine Disorders. 2025;25:58. DOI: 10.1186/s12902-025-01800-4.