Executive Summary
Hair loss affects roughly 50% of men and 25% of women by age 50, and the search for effective treatments has driven researchers toward a promising class of molecules: bioactive peptides. GHK-Cu, Thymosin Beta-4, PTD-DBM, and several other peptide compounds have shown real potential in preclinical and early clinical research for stimulating hair follicle activity, reversing miniaturization, and extending the active growth phase of the hair cycle.
Key Takeaways
- GHK-Cu (copper peptide) - a naturally occurring tripeptide-copper complex that modulates over 4,000 human genes, stimulates dermal papilla cell proliferation, promotes VEGF-driven angiogenesis around follicles, and activates the Wnt/beta-catenin signaling pathway that governs hair cycling.
- Thymosin Beta-4 (TB-500) - a 43-amino-acid polypeptide that activates hair follicle stem cells, promotes their migration to the follicle base, and accelerates the transition from resting to active growth phases.
- PTD-DBM - a synthetic peptide designed to block the CXXC5-Dishevelled interaction, thereby removing a key brake on Wnt/beta-catenin signaling and enabling hair follicle neogenesis.
- Supporting peptides - including BPC-157 for anti-inflammatory support and vascular repair, and growth hormone secretagogues like CJC-1295/Ipamorelin for systemic growth factor optimization.
For decades, the hair loss treatment landscape has been dominated by just two FDA-approved medications: minoxidil (a topical vasodilator) and finasteride (a 5-alpha reductase inhibitor). Both work, but both come with limitations. Minoxidil requires twice-daily application and loses effectiveness once discontinued. Finasteride carries the risk of sexual side effects that concern many users. Neither addresses the full spectrum of biological processes that drive hair loss, particularly the signaling cascade dysfunction in dermal papilla cells, the depletion of follicular stem cell reserves, and the chronic micro-inflammation that accelerates follicle miniaturization.
Peptides offer something different. These short chains of amino acids act as biological messengers, capable of influencing specific cellular pathways with a precision that small-molecule drugs often lack. The peptides covered in this report target distinct but complementary mechanisms:
- GHK-Cu (copper peptide) - a naturally occurring tripeptide-copper complex that modulates over 4,000 human genes, stimulates dermal papilla cell proliferation, promotes VEGF-driven angiogenesis around follicles, and activates the Wnt/beta-catenin signaling pathway that governs hair cycling.
- Thymosin Beta-4 (TB-500) - a 43-amino-acid polypeptide that activates hair follicle stem cells, promotes their migration to the follicle base, and accelerates the transition from resting to active growth phases.
- PTD-DBM - a synthetic peptide designed to block the CXXC5-Dishevelled interaction, thereby removing a key brake on Wnt/beta-catenin signaling and enabling hair follicle neogenesis.
- Supporting peptides - including BPC-157 for anti-inflammatory support and vascular repair, and growth hormone secretagogues like CJC-1295/Ipamorelin for systemic growth factor optimization.
This report examines the published research behind each of these compounds, analyzing their mechanisms of action, preclinical data, available clinical evidence, delivery methods, safety profiles, and practical application protocols. We also compare peptide-based approaches to conventional treatments, explore combination strategies, and address the most common questions researchers and clinicians encounter in this space.
Key Takeaway
While large-scale, randomized controlled trials for hair growth peptides remain limited, the preclinical evidence is substantial and growing. GHK-Cu has the strongest overall evidence base for hair applications, with multiple in vitro studies, animal models, and small clinical trials supporting its efficacy. Thymosin Beta-4 has compelling animal data showing stem cell activation and accelerated hair cycling. PTD-DBM represents a newer, mechanistically targeted approach with promising early results. The field is moving rapidly, and combination protocols that address multiple pathways simultaneously appear to offer the greatest potential.
Whether you're a researcher evaluating peptide candidates for hair biology studies, a clinician considering adjunctive therapies for patients with androgenetic alopecia, or someone exploring the science behind these compounds, this report provides the evidence-based foundation you need. For a broader overview of peptide science, visit our Peptide Research Hub, or take our Free Assessment to find compounds matched to your research goals.
Hair Follicle Biology & the Growth Cycle
Understanding how peptides influence hair growth requires a solid grasp of follicle biology. The hair follicle is one of the most complex mini-organs in the human body, with its own stem cell reservoir, vascular supply, immune privilege, and a cyclic regeneration process that repeats throughout a person's lifetime.
Anatomy of the Hair Follicle
Each hair follicle is a tube-like structure that extends from the epidermis deep into the dermis, and sometimes into the subcutaneous fat layer. The follicle consists of several distinct compartments, each playing a specific role in hair production and cycling.
At the very base of the follicle sits the dermal papilla (DP), a cluster of specialized mesenchymal cells that functions as the command center for hair growth. The DP is richly vascularized and innervated, receiving nutrients from a capillary loop and transmitting signals that determine the size, shape, and color of the hair shaft. Think of the dermal papilla as the conductor of an orchestra: it doesn't produce the hair itself, but it coordinates every cell that does.
Surrounding the dermal papilla is the hair matrix, a population of rapidly dividing keratinocytes that actually build the hair shaft. These are among the fastest-proliferating cells in the human body, dividing every 12 to 72 hours during active growth. The matrix also contains melanocytes that inject pigment into the growing hair shaft.
Higher up the follicle, in a region called the bulge, resides the follicle's stem cell reservoir. These multipotent stem cells are the source material for follicle regeneration. When activated by signals from the dermal papilla and surrounding niche cells, bulge stem cells proliferate, migrate downward, and differentiate into the matrix cells that produce a new hair. The bulge region is a primary target for Thymosin Beta-4 research.
The outer root sheath (ORS) forms the structural envelope of the follicle, while the inner root sheath (IRS) molds and shapes the growing hair shaft as it moves upward. The dermal sheath wraps around the outside of the follicle and serves as a reservoir of cells that can replenish the dermal papilla.
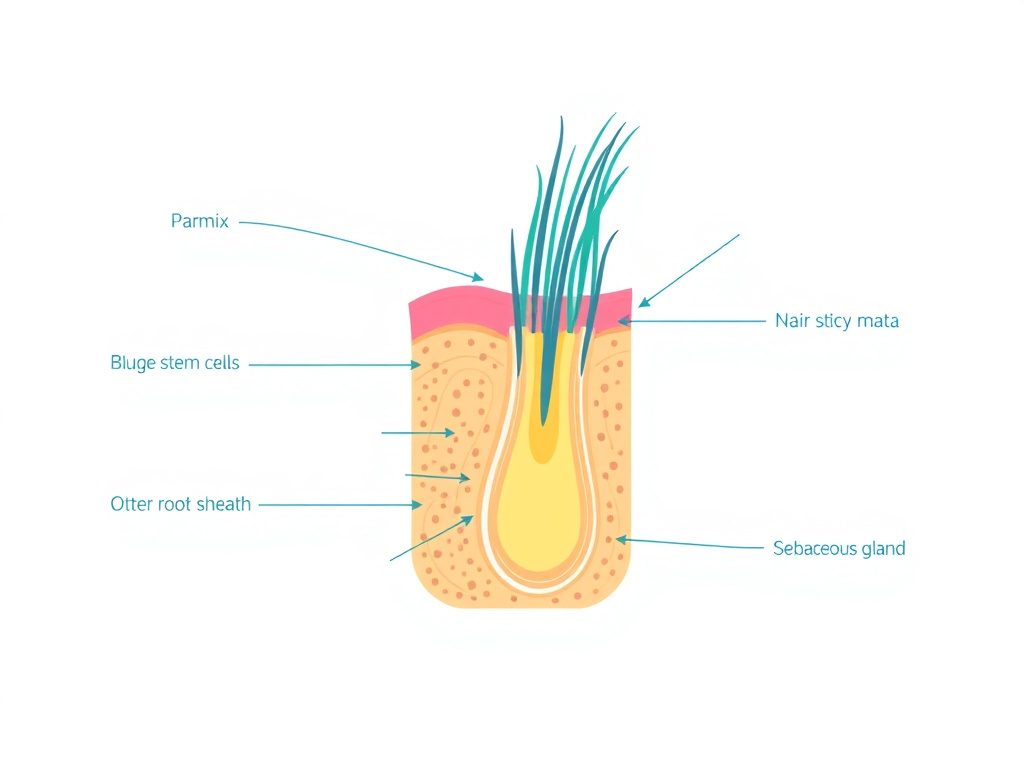
Figure 1: Hair follicle anatomy showing the key structures involved in peptide-mediated hair growth, including the dermal papilla (primary signaling hub), bulge stem cell niche (target of TB-4), and perifollicular vasculature (influenced by GHK-Cu via VEGF).
The Hair Growth Cycle: Anagen, Catagen, and Telogen
Hair doesn't grow continuously. Instead, each follicle cycles independently through three distinct phases, a process governed by molecular crosstalk between the dermal papilla, stem cells, immune cells, and surrounding tissue.
Anagen (Active Growth Phase)
Anagen is the active growth phase during which the follicle produces a new hair shaft. On the scalp, anagen typically lasts 2 to 7 years, which is why scalp hair can grow so long compared to body hair (where anagen may last only weeks to months). During anagen, the dermal papilla releases a cocktail of growth factors, including FGF-7, IGF-1, VEGF, hepatocyte growth factor (HGF), and Wnt ligands, that stimulate matrix cell proliferation and hair shaft production.
The length of anagen directly determines the maximum length a hair can achieve. It also determines hair density: at any given time, roughly 85-90% of scalp follicles should be in anagen. When this percentage drops, as happens in androgenetic alopecia, visible thinning results.
Several of the peptides in this report work by extending anagen duration or by promoting the transition of resting follicles back into anagen. GHK-Cu, for instance, has been shown to stimulate dermal papilla cells to secrete VEGF and HGF, both of which are anagen-promoting growth factors. Research published in Pharmaceutics found that GHK-Cu-treated follicles entered early growth stages in as few as 6 days, compared to approximately 9 days for minoxidil-treated follicles.
Catagen (Regression Phase)
Catagen is a brief transitional phase lasting approximately 2 to 3 weeks. During catagen, the lower two-thirds of the follicle undergoes apoptosis (programmed cell death) and regresses upward toward the skin surface. The dermal papilla condenses and moves upward to rest just below the bulge region. Hair shaft production ceases, and the base of the hair keratinizes to form the "club hair."
Catagen is triggered by a shift in the signaling balance: pro-apoptotic signals like TGF-beta1, TGF-beta2, and BMP-2/4 overcome the pro-growth signals. One of the ways GHK-Cu supports hair growth is by inhibiting TGF-beta production, which may help delay the onset of catagen and extend the productive growth phase.
Telogen (Resting Phase)
Telogen is the resting phase, lasting approximately 2 to 4 months on the scalp. The follicle is dormant, the club hair remains anchored in the follicle (but can be shed during combing or washing), and the stem cells in the bulge await reactivation signals. About 10-15% of scalp follicles are normally in telogen at any time.
In telogen effluvium (a common form of diffuse hair loss triggered by stress, illness, or hormonal changes), an abnormally high percentage of follicles are pushed into telogen simultaneously, resulting in noticeable shedding 2-3 months later. Peptides that promote the telogen-to-anagen transition, such as Thymosin Beta-4, are particularly relevant for this condition.
Exogen (Shedding Phase)
Some researchers recognize a fourth phase called exogen, during which the old club hair is actively released from the follicle. This is distinct from the passive hair loss that can occur during telogen. Exogen is an active, enzymatically driven process that clears the way for a new anagen hair to emerge.
The Wnt/Beta-Catenin Signaling Pathway: Master Regulator of Hair Cycling
If there's one molecular pathway that sits at the center of hair follicle biology, it's the Wnt/beta-catenin pathway. This signaling cascade is the primary determinant of whether a follicle enters anagen, how long it stays in anagen, and whether new follicles can form (a process called follicle neogenesis).
Here's how it works in simplified terms:
- Wnt ligands (secreted proteins) bind to Frizzled receptors on the cell surface.
- This activates the Dishevelled (Dvl) protein inside the cell.
- Active Dvl inhibits the "destruction complex" (which normally tags beta-catenin for degradation).
- Beta-catenin accumulates in the cytoplasm and translocates to the nucleus.
- In the nucleus, beta-catenin partners with TCF/LEF transcription factors to activate genes involved in cell proliferation, stem cell maintenance, and hair follicle differentiation.
When Wnt signaling is active, follicles grow. When it's suppressed, follicles miniaturize and eventually stop producing visible hair. In androgenetic alopecia, DHT (dihydrotestosterone) suppresses Wnt signaling in genetically susceptible follicles, which is one of the key reasons those follicles progressively shrink.
Two of the peptides in this report directly target this pathway. GHK-Cu promotes beta-catenin nuclear accumulation in dermal papilla cells. PTD-DBM takes a different approach: it blocks CXXC5, a negative regulator that normally puts the brakes on Wnt signaling by binding to Dishevelled. By removing this brake, PTD-DBM allows Wnt signaling to proceed at full capacity.
Why Wnt Matters for Hair Loss
Research has shown that CXXC5 (the target of PTD-DBM) is overexpressed in the bald scalps of men with androgenetic alopecia. Both DHT and prostaglandin D2 (PGD2), two molecules strongly implicated in pattern hair loss, decrease Wnt/beta-catenin signaling via CXXC5. This makes CXXC5 inhibition through peptides like PTD-DBM a mechanistically logical approach to treating pattern baldness at its molecular root.
Dermal Papilla Signaling and Follicle Miniaturization
The dermal papilla is where the action happens in hair loss, and it's also the primary target for most hair growth peptides. In healthy follicles, the DP produces a rich signaling environment that maintains matrix cell proliferation and hair shaft production. In androgenetic alopecia, this signaling deteriorates progressively.
Here's what happens during miniaturization:
- DHT binds to androgen receptors in genetically susceptible DP cells
- This triggers secretion of TGF-beta1, TGF-beta2, DKK-1, and IL-6
- These factors suppress Wnt signaling, promote apoptosis of matrix cells, and shorten anagen
- The DP itself loses cells and shrinks in volume
- With each successive cycle, the follicle produces a thinner, shorter, less pigmented hair
- Eventually, the follicle produces only vellus hair (peach fuzz) or ceases production entirely
GHK-Cu directly counteracts several of these processes. It stimulates DP cell proliferation while inhibiting apoptosis. It suppresses TGF-beta. It promotes VEGF secretion, maintaining the vascular supply that keeps DP cells nourished. And it activates Wnt/beta-catenin signaling, the very pathway that DHT works to suppress.
The Role of Stem Cells in Hair Regeneration
The bulge stem cells are the ultimate source of follicle renewal. Without them, a follicle cannot regenerate. In most forms of non-scarring alopecia (including androgenetic alopecia), the stem cells are still present even in miniaturized follicles. They're just not being adequately activated.
This is where Thymosin Beta-4 enters the picture. Research published in the FASEB Journal demonstrated that Thymosin Beta-4 promotes the migration and differentiation of hair follicle stem cells, effectively "waking up" dormant follicles. The peptide appears to work through multiple mechanisms: it promotes stem cell migration to the follicle base, supports their differentiation into matrix cells, and facilitates the extracellular matrix remodeling needed for follicle regeneration.
The fact that stem cells persist in miniaturized follicles is actually encouraging news for peptide-based approaches. It means the raw material for regrowth is still there. The challenge is providing the right signals to activate it, and that's precisely what these peptides aim to do.
Perifollicular Vasculature and Growth Factor Dependence
Hair follicles are metabolically demanding structures. During anagen, matrix cells divide at rates comparable to bone marrow and intestinal epithelium. This rapid proliferation requires a strong blood supply delivering oxygen, glucose, amino acids, and other nutrients.
Each follicle is served by a network of capillaries that form a basket-like structure around the dermal papilla and lower follicle. The density and quality of this perifollicular vasculature directly correlates with hair thickness and growth rate. In miniaturizing follicles, the vascular network degrades alongside the follicle itself.
VEGF (vascular endothelial growth factor) is the primary driver of perifollicular angiogenesis. Research has shown that overexpression of VEGF in mouse skin increases hair follicle size and accelerates hair growth. Conversely, blocking VEGF leads to follicle regression.
GHK-Cu is a potent stimulator of VEGF production in dermal papilla cells and surrounding fibroblasts. This pro-angiogenic effect is one of its most well-documented mechanisms and likely contributes to its hair growth effects. TB-500 also promotes angiogenesis through VEGF regulation and downstream activation of the P38/ERK/AKT signaling cascade.
Inflammation, Immune Privilege, and Hair Loss
The hair follicle normally maintains a state of "immune privilege," meaning it's partially shielded from immune system surveillance. This privilege is maintained by reduced expression of MHC class I molecules, secretion of immunosuppressive factors, and a physical barrier provided by the inner root sheath.
When immune privilege collapses, inflammatory cells infiltrate the follicle and can damage the stem cell niche, the dermal papilla, or both. This plays a role in several forms of hair loss:
- Alopecia areata - a direct autoimmune attack on hair follicles
- Androgenetic alopecia - chronic perifollicular micro-inflammation (often overlooked but well-documented in histological studies)
- Cicatricial alopecia - severe inflammation leading to permanent follicle destruction
BPC-157, while not primarily a hair peptide, has potent anti-inflammatory properties that make it a useful adjunct in combination protocols. It modulates the nitric oxide system, reduces inflammatory cytokine production, and supports tissue repair. These properties help maintain the follicular microenvironment that peptides like GHK-Cu and TB-500 need to work effectively.
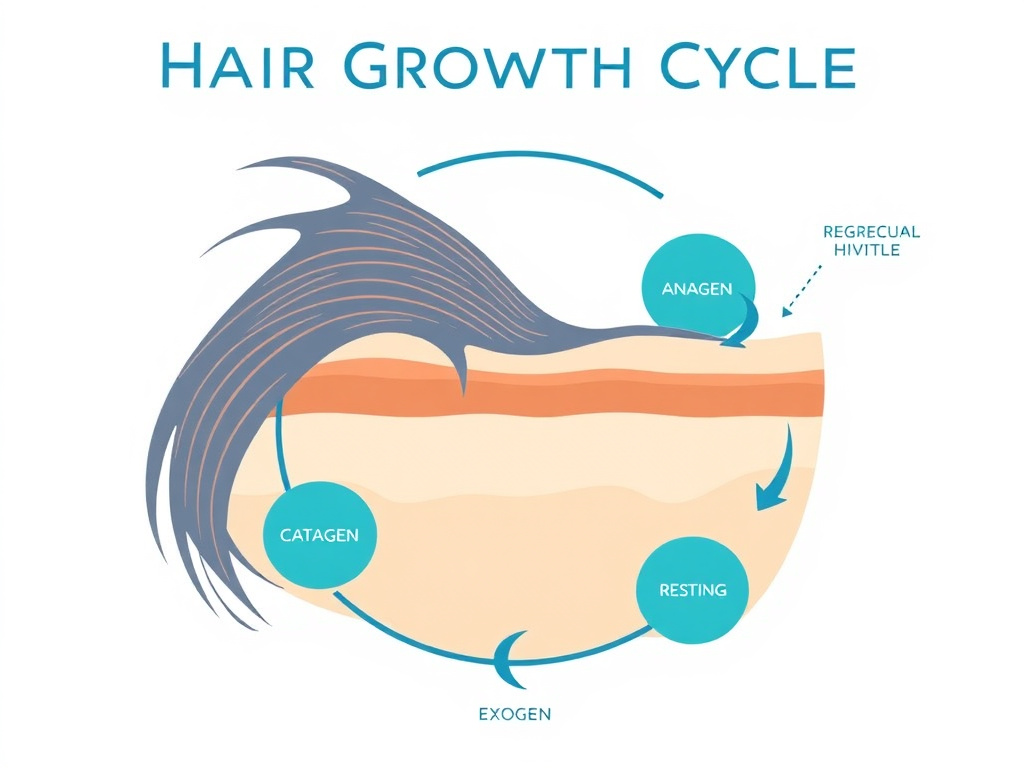
Figure 2: The hair growth cycle showing anagen, catagen, telogen, and exogen phases. Key molecular transitions are labeled, including Wnt/beta-catenin activation at the telogen-to-anagen transition (targeted by GHK-Cu and PTD-DBM) and stem cell mobilization (targeted by Thymosin Beta-4).
GHK-Cu Hair Research: Mechanism, Evidence & Applications
GHK-Cu (glycyl-L-histidyl-L-lysine copper complex) is a naturally occurring tripeptide bound to a copper(II) ion. First identified in human plasma by Dr. Loren Pickart in 1973, it was initially studied for its wound healing properties. Over the past five decades, research has revealed that this small molecule influences an extraordinary range of biological processes, including many directly relevant to hair follicle biology.
What Is GHK-Cu?
GHK-Cu is formed when the tripeptide glycyl-histidyl-lysine chelates (binds) a copper ion. It's present naturally in human blood plasma, saliva, and urine, with plasma levels of approximately 200 ng/mL in young adults. These levels decline with age: by age 60, circulating GHK-Cu has dropped to roughly 80 ng/mL, a 60% reduction that correlates with declining regenerative capacity across multiple tissue types.
The molecule is remarkably small (molecular weight ~403 Da for the free peptide, ~467 Da with copper), which contributes to favorable tissue penetration properties. It acts as a biological signal rather than a structural component, influencing gene expression patterns across thousands of genes simultaneously.
Research published by Pickart and colleagues has documented that GHK-Cu modulates the expression of over 4,000 human genes, roughly 6% of the human genome. Many of these genes are directly involved in tissue remodeling, stem cell biology, anti-inflammatory signaling, and antioxidant defense, all processes central to hair follicle health and cycling. You can explore the full GHK-Cu research profile on our site.
GHK-Cu Mechanisms of Action in Hair Follicles
GHK-Cu influences hair growth through at least six distinct but interconnected mechanisms. Understanding these helps explain why copper peptides have shown effects across multiple types of hair loss and why they may complement other treatments that work through different pathways.
1. Wnt/Beta-Catenin Pathway Activation
As discussed in the biology section, Wnt/beta-catenin signaling is the master switch for hair follicle cycling. GHK-Cu promotes beta-catenin nuclear accumulation in dermal papilla cells, a hallmark of active Wnt signaling. This drives follicles from the resting (telogen) phase into active growth (anagen).
The mechanism appears to involve both direct stimulation of Wnt ligand production and indirect support through suppression of Wnt antagonists like DKK-1 (Dickkopf-1), which is elevated in balding scalps. Gene expression analysis has shown that GHK-Cu upregulates several Wnt pathway components while simultaneously downregulating negative regulators.
2. Dermal Papilla Cell Proliferation and Anti-Apoptosis
GHK-Cu directly stimulates dermal papilla cell proliferation in culture. It also inhibits apoptosis in these cells, which is particularly relevant because DP cell loss is a central feature of follicle miniaturization. By maintaining DP cell numbers and viability, GHK-Cu helps preserve the signaling capacity needed for strong hair production.
In vitro studies have demonstrated dose-dependent increases in DP cell proliferation when cultured with GHK-Cu, with optimal effects typically observed at concentrations between 1 and 10 micromolar. The anti-apoptotic effect appears to involve suppression of caspase activation and upregulation of BCL-2 family survival factors.
3. VEGF and Angiogenesis Promotion
One of GHK-Cu's most consistently demonstrated effects is stimulation of VEGF (vascular endothelial growth factor) production. VEGF drives the formation and maintenance of the perifollicular vascular network that supplies nutrients to the growing follicle.
A study published in Pharmaceutics found that GHK-Cu stimulated hair follicle cells to secrete both VEGF and HGF (hepatocyte growth factor), with a novel microemulsion delivery system producing the most significant increases in these growth factors. Improved vascular supply translates to better nutrient delivery, oxygen availability, and waste removal, all essential for maintaining the high metabolic demands of anagen follicles.
4. TGF-Beta Suppression
TGF-beta (transforming growth factor beta) is one of the primary catagen-inducing signals. It promotes matrix cell apoptosis and triggers follicle regression. In androgenetic alopecia, DHT stimulates DP cells to produce excess TGF-beta, which is one mechanism by which androgens drive follicle miniaturization.
GHK-Cu has been shown to inhibit TGF-beta production, which may help prevent premature catagen entry and extend the productive growth phase. This anti-TGF-beta effect also reduces fibrosis around the follicle, which can contribute to permanent follicle loss when chronic.
5. Extracellular Matrix Remodeling
The extracellular matrix (ECM) surrounding the hair follicle isn't just structural scaffolding. It's an active signaling environment that stores growth factors, guides cell migration, and influences stem cell behavior. GHK-Cu promotes healthy ECM remodeling by:
- Stimulating collagen synthesis (types I, III, and V)
- Promoting glycosaminoglycan production (including hyaluronic acid and dermatan sulfate)
- Regulating matrix metalloproteinase (MMP) activity to maintain proper ECM turnover
- Increasing decorin and versican production, both of which are associated with anagen induction
The ECM molecule versican is particularly interesting in the hair context. It's highly expressed in the dermal papilla during anagen and has been shown to be necessary for anagen induction. GHK-Cu's ability to promote versican production may be an underappreciated contributor to its hair growth effects.
6. Anti-Inflammatory and Antioxidant Effects
Chronic micro-inflammation around hair follicles contributes to progressive miniaturization in androgenetic alopecia and plays a central role in several other forms of hair loss. GHK-Cu reduces inflammatory signaling through multiple mechanisms:
- Suppression of pro-inflammatory cytokines (IL-6, TNF-alpha)
- Reduction of NF-kB activation
- Upregulation of antioxidant enzymes (superoxide dismutase, glutathione peroxidase)
- Reduction of oxidative stress markers
The antioxidant effects are partly mediated by the copper ion itself, which serves as a cofactor for superoxide dismutase (SOD), one of the body's primary antioxidant enzymes. By delivering copper directly to tissues, GHK-Cu supports local antioxidant defense in the perifollicular environment.
Clinical Relevance
The multi-pathway mechanism of GHK-Cu is one of its greatest advantages over single-target treatments. While finasteride blocks one enzyme (5-alpha reductase) and minoxidil primarily acts as a vasodilator, GHK-Cu simultaneously addresses Wnt signaling, vascular supply, inflammation, ECM quality, and DP cell survival. This breadth of action may explain why it shows additive or complementary effects when combined with conventional treatments.
Preclinical Evidence: In Vitro and Animal Studies
The preclinical evidence for GHK-Cu's effects on hair follicle biology spans several decades and includes both in vitro cell culture studies and in vivo animal models.
In Vitro Studies
Uno and colleagues published one of the foundational studies on copper peptides and hair growth, examining the effects of GHK-Cu on cultured human hair follicles. Their research demonstrated that GHK-Cu treatment increased follicle size and prolonged the duration of anagen in organ culture. The peptide also stimulated proliferation of follicular keratinocytes, the cells that actually build the hair shaft.
A key study by Pyo and colleagues examined the effect of the tripeptide-copper complex on human hair growth in vitro. Using human scalp hair follicle organ cultures, they found that GHK-Cu at concentrations of 1 to 10 micromolar significantly increased hair shaft elongation compared to untreated controls. The effect was dose-dependent, with higher concentrations producing greater elongation up to a plateau.
More recent work using nanoliposome delivery systems has shown enhanced effects when GHK-Cu is co-delivered with other bioactive peptides. A study published in Colloids and Surfaces B: Biointerfaces demonstrated that nanoliposomal co-delivery of GHK-Cu with other hair-active peptides produced greater hair follicle stimulation than either peptide alone, suggesting potential for combination formulations.
Animal Studies
Animal studies have provided important confirmation of the in vitro findings. In mouse models, topical application of GHK-Cu has been shown to:
- Accelerate the telogen-to-anagen transition (resting follicles entering active growth sooner)
- Increase hair follicle size and density
- Improve hair shaft diameter and quality
- Stimulate VEGF expression in the perifollicular region
A particularly informative study compared GHK-Cu with minoxidil in a mouse hair growth model. GHK-Cu-treated follicles entered the early stages of growth in approximately 6 days, showing visible hyperpigmentation and regrowth, while the minoxidil-treated group achieved the same effect after approximately 9 days. Both treatments stimulated follicular VEGF and HGF secretion, but the onset of visible growth was faster with GHK-Cu, particularly when delivered via an optimized microemulsion vehicle.
Clinical Evidence in Humans
Clinical data on GHK-Cu for hair growth is more limited than the preclinical evidence, but several studies have reported positive results.
Hair Count and Density Studies
In clinical studies examining topical GHK-Cu application for androgenetic alopecia, researchers have documented improvements in hair count, hair diameter, and overall scalp coverage. One study reported a 38% increase in hair count in the GHK-Cu group compared to placebo, with side effects limited to mild scalp tingling in some participants.
A double-blind, placebo-controlled study involving participants with androgenetic alopecia found that 98% of those using a topical GHK-Cu serum reported visibly thicker hair, with an average 40% reduction in hair shedding over the study period. However, it bears mention that these results should be interpreted with some caution, as many clinical studies on copper peptides for hair have involved relatively small sample sizes and short follow-up periods.
Microneedling Combination Studies
Some of the most promising clinical results have come from studies combining GHK-Cu with microneedling. A 2025 study published in PMC examined enhanced hair regrowth using monthly sessions of microneedling with a combination solution containing copper peptides along with minoxidil and dutasteride. The results, assessed by both artificial intelligence analysis and blinded evaluators, showed significant improvements in hair density and coverage.
Microneedling creates controlled micro-channels in the scalp that increase topical serum absorption by up to 400% while simultaneously triggering the wound-healing cascade. This cascade releases platelet-derived growth factor (PDGF) and VEGF, which complement GHK-Cu's own pro-angiogenic effects. The combination of enhanced penetration and complementary wound-healing signals likely accounts for the superior results seen with microneedling protocols.
Hair Count Response Data
GHK-Cu vs. Minoxidil: Head-to-Head Comparison
The comparison between GHK-Cu and minoxidil is one of the most commonly requested analyses in the hair peptide space. While no large-scale head-to-head clinical trial has been published, the available data allows some useful comparisons.
| Parameter | GHK-Cu | Minoxidil 5% |
|---|---|---|
| Primary mechanism | Multi-pathway (Wnt, VEGF, TGF-beta suppression, ECM remodeling) | Potassium channel opener / vasodilator |
| Onset of follicle activation (preclinical) | ~6 days | ~9 days |
| Growth factor stimulation | VEGF, HGF, FGF, multiple others | Primarily VEGF |
| Anti-inflammatory effect | Yes (NF-kB, IL-6, TNF-alpha suppression) | Minimal |
| ECM remodeling | Yes (collagen, GAGs, versican) | No |
| Common side effects | Mild tingling, occasional irritation | Scalp dryness, irritation, initial shedding, unwanted facial hair |
| Systemic side effects | Not reported at topical doses | Rare hypotension, edema, tachycardia |
| FDA approved for hair loss | No | Yes |
| Level of clinical evidence | Preclinical + small clinical studies | Multiple large RCTs |
The available evidence suggests that GHK-Cu and minoxidil may work best in combination rather than as alternatives. Their mechanisms are largely complementary: minoxidil's potassium channel-opening effect promotes vasodilation and direct follicle stimulation, while GHK-Cu provides broader signaling support through Wnt activation, ECM remodeling, and anti-inflammatory effects. Preclinical data showing greater effects when the two are combined supports this hypothesis.
Topical GHK-Cu Formulations for Hair
The delivery vehicle for topical GHK-Cu significantly affects its efficacy. The peptide itself has reasonable skin penetration due to its small size, but formulation can enhance or limit its bioavailability at the follicle level.
Standard serum formulations typically contain GHK-Cu at concentrations ranging from 1% to 5%, dissolved in an aqueous or hydroalcoholic base. These provide baseline efficacy and are the simplest to formulate and use.
Microemulsion systems have shown enhanced performance in preclinical studies. Research published in Pharmaceutics demonstrated that an ionic liquid microemulsion delivery system for GHK-Cu produced greater activation of the Wnt/beta-catenin pathway and higher levels of VEGF and HGF secretion compared to the peptide in simple solution. The microemulsion's ability to disrupt the stratum corneum barrier and deliver the peptide directly to the follicular unit likely accounts for the improved results.
Nanoliposome formulations represent another advanced delivery approach. A study in Colloids and Surfaces B demonstrated that nanoliposomal co-delivery of bioactive peptides (including copper peptides) enhanced follicular penetration and produced superior hair growth stimulation compared to free peptide solutions.
For those exploring topical GHK-Cu products, formulation quality matters significantly. Key factors include peptide concentration, delivery vehicle technology, pH (GHK-Cu is most stable in slightly acidic conditions around pH 5-6), and the presence of complementary active ingredients.
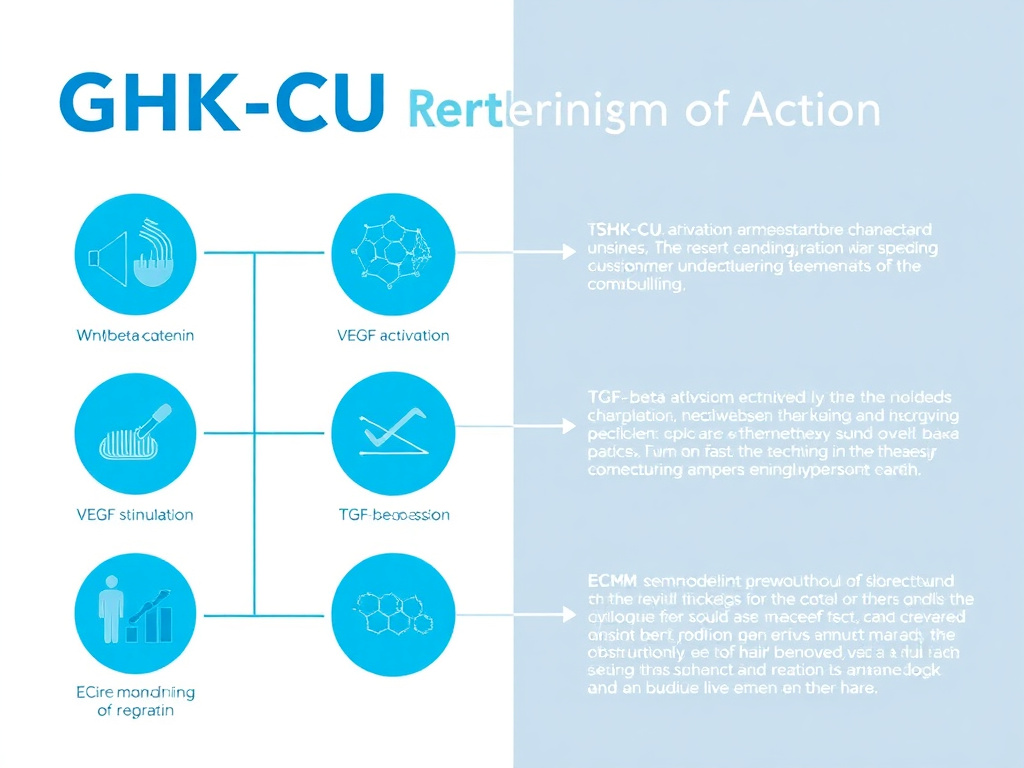
Figure 3: GHK-Cu mechanism of action for hair growth showing the six primary pathways: Wnt/beta-catenin activation, dermal papilla cell proliferation, VEGF-driven angiogenesis, TGF-beta suppression, ECM remodeling, and anti-inflammatory/antioxidant effects.
Thymosin Beta-4 & TB-500 for Hair Growth
Thymosin Beta-4 (Tbeta4) is a 43-amino-acid polypeptide that plays a fundamental role in cell migration, differentiation, and tissue repair. Its connection to hair growth was discovered somewhat accidentally during wound healing research, when scientists noticed significant hair regrowth around treated areas. Subsequent studies revealed that Tbeta4 activates hair follicle stem cells, making it one of the most intriguing peptides in the hair biology field.
What Is Thymosin Beta-4?
Thymosin Beta-4 is the most abundant member of the beta-thymosin family. It was originally isolated from the thymus gland (hence the name), but it's expressed in virtually every cell type in the body. Its primary intracellular function is sequestering G-actin monomers, which regulates actin polymerization and thereby controls cell motility, shape, and division.
Beyond its intracellular role, Thymosin Beta-4 has significant extracellular signaling functions. When released from cells (during injury, for example), it promotes wound healing, reduces inflammation, stimulates angiogenesis, and activates resident stem and progenitor cells. These regenerative properties are what led researchers to investigate its effects on hair follicles.
TB-500 is a synthetic fragment of Thymosin Beta-4 that contains the active region of the full-length peptide. It's commonly used in research settings due to its enhanced stability and consistent bioactivity. When we reference TB-500 in this report, we're referring to this active fragment that replicates the key biological activities of full-length Thymosin Beta-4. For a complete review, see our TB-500 research guide.
Discovery of Hair Growth Effects
The link between Thymosin Beta-4 and hair growth emerged from research at the National Institutes of Health (NIH). Philp and colleagues published a landmark study in 2004 in the FASEB Journal showing that Thymosin Beta-4 increases hair growth by activation of hair follicle stem cells. This was the first demonstration that a specific exogenous peptide could directly activate the follicular stem cell compartment to promote hair regrowth.
The discovery wasn't planned. Researchers studying Thymosin Beta-4's wound healing properties in rodent models noticed accelerated and enhanced hair growth in the treatment areas. This serendipitous observation led to targeted investigations that confirmed the effect was real, reproducible, and mechanistically distinct from existing hair growth treatments.
A follow-up study by the same group, published in the Annals of the New York Academy of Sciences in 2007, provided further detail: Thymosin Beta-4 induces hair growth via stem cell migration and differentiation. This work showed that the peptide doesn't just activate stem cells in place; it promotes their directed migration from the bulge niche to the hair follicle base, where they differentiate into the transit-amplifying matrix cells that produce new hair.
Mechanisms of Action for Hair Growth
1. Hair Follicle Stem Cell Activation
The primary mechanism by which Thymosin Beta-4 promotes hair growth is activation of bulge-resident stem cells. In a resting (telogen) follicle, these stem cells are quiescent, held in a dormant state by signals from the surrounding niche. The transition from telogen to anagen requires that these cells exit quiescence, begin proliferating, and migrate to the follicle base.
Thymosin Beta-4 promotes all three of these steps. In the original Philp et al. study, rat vibrissa (whisker) follicle clonogenic keratinocytes, which are closely related to bulge-residing stem cells, showed increased migration and differentiation in the presence of Thymosin Beta-4. The peptide effectively overcomes the quiescence signals and tips the balance toward activation.
This mechanism is complementary to GHK-Cu's effects. While GHK-Cu primarily targets the dermal papilla (the signaling hub), Thymosin Beta-4 targets the stem cells (the cellular source material). Together, they address both the signal and the responding cells.
2. Acceleration of the Telogen-to-Anagen Transition
Thymosin Beta-4 treatment in animal models consistently accelerates the transition from resting to active growth phases. This is visible macroscopically as earlier and more synchronized hair regrowth in treated areas compared to controls.
A study published in PLoS ONE confirmed these effects, demonstrating that Thymosin Beta-4 induces mouse hair growth with visible results within days of treatment. The acceleration occurs because the peptide activates the signaling cascade that drives anagen induction, including upregulation of Wnt signaling components and growth factors in the dermal papilla.
3. Promotion of Cell Migration
Thymosin Beta-4's role in actin cytoskeleton dynamics directly supports cell migration. During the telogen-to-anagen transition, progenitor cells must physically migrate from the bulge region downward to the dermal papilla area, a journey of several hundred micrometers. This migration requires dynamic actin remodeling, precisely the process that Thymosin Beta-4 regulates.
By promoting actin dynamics and cell motility, the peptide ensures that activated stem cells can efficiently reach the follicle base and begin the differentiation process that produces new matrix cells. Without adequate migration, even activated stem cells won't produce effective hair regrowth.
4. Extracellular Matrix Remodeling
Like GHK-Cu, Thymosin Beta-4 supports ECM remodeling during follicle cycling. The follicle must extensively remodel its surrounding matrix during each growth cycle: breaking down the catagen-regressed structure and building a new anagen follicle. Thymosin Beta-4 promotes this remodeling by regulating matrix metalloproteinase expression and supporting the deposition of new ECM components.
A comprehensive review published in the International Journal of Molecular Sciences examined the multiple potential roles of Thymosin Beta-4 in hair follicle growth and development. The authors concluded that Tbeta4 participates in every phase of the hair cycle and influences follicle biology through stem cell activation, ECM remodeling, angiogenesis, and anti-inflammatory signaling.
5. VEGF Regulation and Angiogenesis
Thymosin Beta-4 promotes angiogenesis through VEGF regulation and downstream activation of the P38/ERK/AKT signaling cascade. Enhanced perifollicular vascularization supports the metabolic demands of anagen follicles and is a shared mechanism with GHK-Cu, though the upstream signaling pathways differ.
In rodent wound healing models, Thymosin Beta-4 treatment significantly increased microvessel density in the treatment area, an effect that extended to hair follicles in the region. The improved blood supply provides the oxygen and nutrient delivery essential for sustained matrix cell proliferation during active hair growth.
6. Anti-Inflammatory Properties
Thymosin Beta-4 has well-documented anti-inflammatory effects that benefit the follicular microenvironment. It reduces production of pro-inflammatory cytokines, modulates macrophage polarization toward an M2 (anti-inflammatory) phenotype, and helps maintain the immune privilege of the hair follicle.
These anti-inflammatory effects are particularly relevant in conditions where perifollicular inflammation contributes to hair loss, including androgenetic alopecia, alopecia areata, and post-inflammatory alopecia. By reducing the inflammatory burden on follicles, TB-500 creates conditions more favorable for normal cycling.
Preclinical Evidence
Rodent Studies
The preclinical evidence for Thymosin Beta-4's hair growth effects comes primarily from rodent studies, which have been remarkably consistent in their findings:
- Philp et al. (2004) - the original FASEB Journal study demonstrating stem cell activation and hair growth in rats treated with Thymosin Beta-4. Clonogenic keratinocyte cultures showed significantly increased migration and differentiation. In vivo, treated rats showed accelerated hair growth compared to vehicle-treated controls.
- Philp et al. (2007) - follow-up study in Annals of the New York Academy of Sciences confirming the stem cell migration mechanism and showing that Thymosin Beta-4 promotes differentiation of migrated stem cells into functional hair-producing cells.
- Gao et al. (2015) - PLoS ONE publication demonstrating Thymosin Beta-4-induced hair growth in mice, with detailed analysis of the molecular pathways involved, including Wnt/beta-catenin signaling components.
- Kim & Bhang (2021) - comprehensive review in the International Journal of Molecular Sciences examining Tbeta4's roles across all phases of the hair cycle, concluding that it influences stem cell activation, proliferation, migration, differentiation, and ECM remodeling.
Transgenic Mouse Studies
Research using transgenic mice has provided additional insights. A study published in the International Journal of Developmental Biology found that overexpression of Thymosin Beta-4 in transgenic mice promoted abnormal tooth development but also stimulated hair growth, confirming that the protein has direct effects on hair follicle biology when present at elevated levels.
These genetic studies complement the exogenous peptide experiments by demonstrating that the hair growth effect is a direct consequence of Thymosin Beta-4 activity rather than an artifact of the delivery method or formulation.
Expression Patterns in Hair Follicles
Endogenous Thymosin Beta-4 expression in hair follicle stem cells tracks closely with the hair cycle. Expression rises during the telogen-to-anagen transition and early anagen, peaks during mid-anagen, and declines during catagen and telogen. This expression pattern is consistent with a role in stem cell activation and hair growth induction.
In situ hybridization and immunohistochemistry studies have localized Tbeta4 expression primarily to the bulge region and outer root sheath during early anagen, and to the matrix region during full anagen. This spatial pattern aligns with the proposed mechanism: the peptide is produced where stem cells are activating and proliferating.
Clinical Status and Human Data
Unlike GHK-Cu, which has some (albeit limited) human clinical data for hair growth, Thymosin Beta-4's evidence base for hair applications remains primarily preclinical. No large-scale human clinical trial specifically evaluating TB-500 for hair growth has been published.
However, anecdotal reports from clinicians and patients using TB-500 for other purposes (primarily injury recovery and wound healing) frequently mention improved hair quality and growth as a side benefit. These observations are consistent with the preclinical data and the known mechanism of action, but they don't constitute clinical evidence.
The peptide's well-established safety profile in wound healing applications provides some reassurance regarding its use for hair growth, but formal clinical trials would be needed to establish efficacy, optimal dosing, and safety specifically for hair indications.
Key Point
Thymosin Beta-4/TB-500 has the strongest preclinical evidence of any peptide for hair follicle stem cell activation. Its mechanism is distinct from and complementary to GHK-Cu (which targets the dermal papilla), finasteride (which blocks DHT production), and minoxidil (which acts as a vasodilator). This mechanistic distinctness makes it an attractive candidate for combination protocols. However, human clinical data specifically for hair growth is lacking, and current use for hair purposes is based primarily on extrapolation from animal studies and wound healing applications.
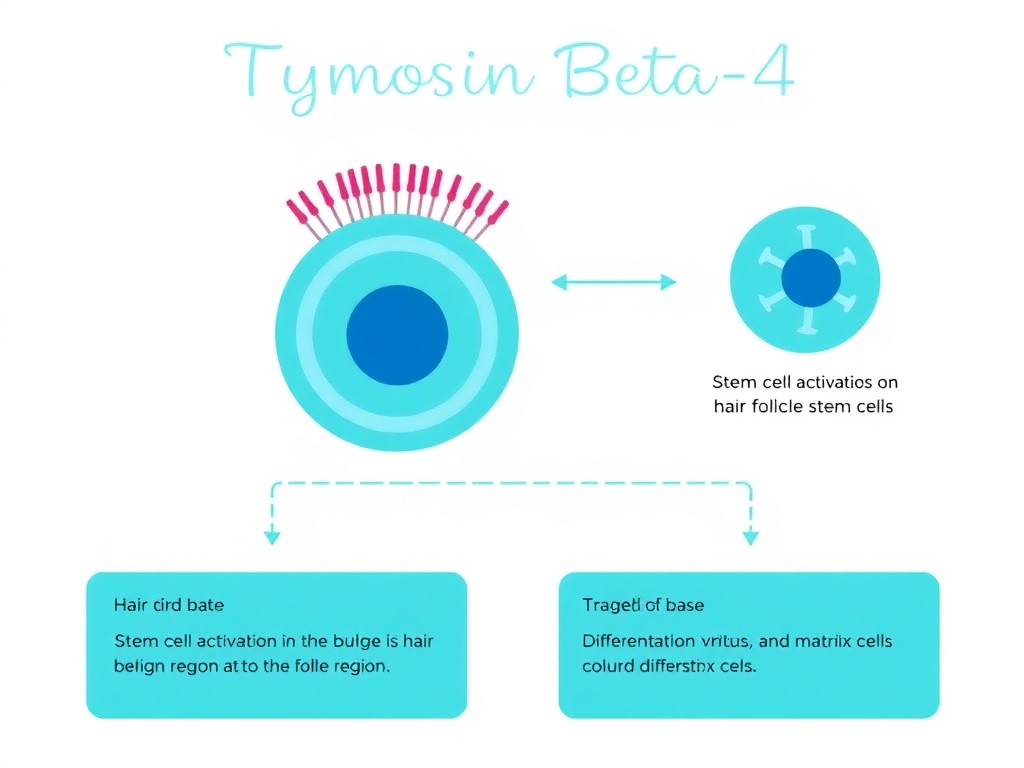
Figure 4: Thymosin Beta-4 mechanism in hair follicles showing activation of bulge stem cells, directed migration to the dermal papilla region, and differentiation into transit-amplifying matrix cells that produce the new hair shaft.
PTD-DBM: Targeted Wnt Pathway Activation for Hair Regrowth
PTD-DBM (Protein Transduction Domain-fused Dishevelled Binding Motif) represents a newer generation of hair growth peptides designed with a specific molecular target in mind. Rather than broadly influencing cell signaling like GHK-Cu or activating stem cells like Thymosin Beta-4, PTD-DBM was engineered to remove a specific molecular brake on the Wnt/beta-catenin pathway, a brake that is overactivated in pattern baldness.
The CXXC5 Problem in Hair Loss
To understand PTD-DBM, you first need to understand CXXC5 (CXXC-type zinc finger protein 5). CXXC5 is a negative feedback regulator of the Wnt/beta-catenin pathway. It works by binding to the Dishevelled (Dvl) protein, one of the key signal transducers in the Wnt cascade, and preventing Dvl from performing its role in stabilizing beta-catenin.
In normal biology, CXXC5 serves as a safety mechanism, preventing runaway Wnt signaling that could lead to uncontrolled cell proliferation. However, in androgenetic alopecia, CXXC5 becomes part of the problem. Research has demonstrated that:
- CXXC5 is overexpressed in the bald scalps of men with androgenetic alopecia
- DHT (dihydrotestosterone) increases CXXC5 expression in dermal papilla cells
- PGD2 (prostaglandin D2), another molecule elevated in balding scalps, also upregulates CXXC5
- The resulting excess CXXC5 suppresses Wnt/beta-catenin signaling below the threshold needed for normal follicle cycling
This creates a vicious cycle: DHT and PGD2 drive CXXC5 overexpression, which suppresses Wnt signaling, which causes follicle miniaturization, which further disrupts the normal signaling environment. PTD-DBM was designed to break this cycle.
How PTD-DBM Works
PTD-DBM is a synthetic peptide composed of two functional domains:
- PTD (Protein Transduction Domain) - enables the peptide to penetrate cell membranes and enter cells directly, without requiring a receptor. This is essential for a topically applied peptide that needs to reach intracellular targets.
- DBM (Dishevelled Binding Motif) - mimics the region of CXXC5 that binds to Dishevelled. By occupying this binding site on Dvl, PTD-DBM acts as a competitive inhibitor, preventing endogenous CXXC5 from binding and blocking the pathway.
The result: Dvl is freed from CXXC5 inhibition and can fulfill its normal role in transducing Wnt signals. Beta-catenin accumulates, translocates to the nucleus, and activates hair growth-related genes. The Wnt pathway operates at capacity even in the presence of elevated CXXC5 levels.
This mechanism is elegant in its specificity. Rather than broadly activating Wnt signaling (which could have unwanted effects in other tissues), PTD-DBM specifically removes an abnormal brake that has been pathologically engaged in balding follicles. It restores normal signaling rather than creating supraphysiological signaling.
Research Evidence for PTD-DBM
In Vitro Studies
Cell culture studies have confirmed that PTD-DBM effectively disrupts the CXXC5-Dvl interaction and restores Wnt/beta-catenin signaling in dermal papilla cells. Key findings include:
- PTD-DBM treatment restored beta-catenin nuclear accumulation in DP cells that had been treated with DHT (which normally suppresses beta-catenin signaling via CXXC5)
- The peptide increased expression of Wnt target genes, including Axin2, cyclin D1, and LEF1
- DP cells treated with PTD-DBM showed increased proliferation and reduced apoptosis, effects consistent with restored Wnt signaling
Animal Studies
Animal studies have provided particularly compelling evidence for PTD-DBM's hair growth effects:
CXXC5 knockout studies: When the CXXC5 gene was knocked out in mice, the animals showed enhanced hair growth and increased hair follicle neogenesis (formation of new follicles), confirming that CXXC5 is indeed a significant brake on hair growth. PTD-DBM's pharmacological inhibition of CXXC5 function mimics these genetic findings.
Topical application studies: By topical application, PTD-DBM promoted the formation of new hair follicles and prevented hair loss in animal models. The peptide was effective when applied to depilated skin, demonstrating its ability to accelerate the telogen-to-anagen transition.
DHT-induced alopecia models: In models specifically designed to mimic androgenetic alopecia, PTD-DBM restored Wnt/beta-catenin signaling that had been suppressed by DHT, leading to recovery of hair growth. This is particularly significant because it demonstrates efficacy against the specific pathological mechanism driving pattern baldness.
PGD2-induced hair loss: Research published in the International Journal of Molecular Sciences demonstrated that hair loss induced by PGD2 was restored by either CXXC5 knockout or treatment with PTD-DBM. This confirms that the CXXC5-Dvl axis mediates PGD2's hair-inhibitory effects and that PTD-DBM can counteract them.
Combination with Valproic Acid (VPA)
One of the more interesting findings is that PTD-DBM shows enhanced effects when combined with valproic acid (VPA), a histone deacetylase (HDAC) inhibitor that independently activates the Wnt/beta-catenin pathway. Combinatory treatment with PTD-DBM and VPA further induced hair regrowth as well as wound-induced hair neogenesis (WIHN) in animal models.
This combination approach makes mechanistic sense: PTD-DBM removes the CXXC5 brake on Wnt signaling, while VPA enhances Wnt signaling through epigenetic mechanisms (increasing histone acetylation at Wnt target gene promoters). Together, they provide a more complete restoration of Wnt activity than either agent alone.
PTD-DBM vs. Other Hair Growth Peptides
| Feature | PTD-DBM | GHK-Cu | Thymosin Beta-4 |
|---|---|---|---|
| Primary target | CXXC5-Dvl interaction | Multiple (DP cells, ECM, vasculature) | Bulge stem cells |
| Pathway specificity | Highly specific (Wnt/beta-catenin) | Broad (4,000+ genes) | Moderate (actin dynamics, Wnt, VEGF) |
| Natural occurrence | Synthetic (not found in nature) | Natural (found in human plasma) | Natural (found in all nucleated cells) |
| Follicle neogenesis | Yes (demonstrated in animal models) | Not demonstrated | Not demonstrated |
| Anti-DHT mechanism | Yes (blocks DHT-induced CXXC5 effects) | Indirect (TGF-beta suppression) | No direct anti-DHT effect |
| Clinical data | Preclinical only | Small clinical studies | Preclinical only |
| Delivery route | Topical (cell-penetrating domain) | Topical, injectable, microneedling | Primarily injectable (subcutaneous) |
Limitations and Considerations
Despite the promising preclinical data, several important limitations should be noted regarding PTD-DBM:
- No human clinical data: All published research is preclinical (cell culture and animal models). No human clinical trial has been conducted or published.
- Availability: PTD-DBM is not widely available as a research reagent or clinical product. It remains primarily in the academic research phase.
- Long-term safety: While CXXC5 knockout mice appear healthy, the long-term consequences of chronic CXXC5 inhibition in humans are unknown. CXXC5 serves as a tumor suppressor in some contexts, so its inhibition requires careful safety evaluation.
- Delivery challenges: While the PTD domain enables cell penetration, achieving adequate follicular delivery through topical application in humans (with thicker stratum corneum than mice) requires further optimization.
Safety Consideration
CXXC5 functions as a negative regulator of Wnt signaling in multiple tissues, not just hair follicles. Aberrant Wnt activation has been implicated in certain cancers. While localized topical application limits systemic exposure, the long-term safety of chronic CXXC5 inhibition requires thorough evaluation in human studies. This peptide should be considered an early-stage research compound, not a ready-for-use treatment.
Other Peptides Under Investigation for Hair Growth
While GHK-Cu, Thymosin Beta-4, and PTD-DBM represent the three most-studied peptide approaches to hair growth, several other peptides have shown relevant activity in preclinical or early clinical research. These compounds target different aspects of follicle biology and may serve as valuable components in combination protocols.
BPC-157 (Body Protection Compound)
BPC-157 is a 15-amino-acid peptide derived from a protein found in human gastric juice. While not primarily researched as a hair growth agent, its properties make it a compelling adjunct for hair restoration protocols.
BPC-157's relevance to hair growth stems from several mechanisms:
- Angiogenesis promotion: BPC-157 promotes formation of new blood vessels through VEGF-dependent and VEGF-independent mechanisms, supporting the perifollicular vasculature essential for anagen maintenance
- Anti-inflammatory effects: The peptide reduces inflammatory cytokine production and modulates the nitric oxide system, helping maintain follicular immune privilege and reducing the perifollicular inflammation associated with androgenetic alopecia
- Tissue repair: BPC-157 accelerates healing of multiple tissue types, including skin. This tissue-protective effect may help maintain the dermal papilla and follicular architecture
- Growth hormone receptor interaction: BPC-157 interacts with the growth hormone system, potentially influencing IGF-1 signaling in the follicle
In combination protocols, BPC-157 is often paired with TB-500 (a combination sometimes referred to as the "Wolverine stack" in biohacking circles) for its complementary tissue repair and anti-inflammatory effects. While direct evidence for BPC-157's effects on hair follicles specifically is limited, its vascular, anti-inflammatory, and tissue-protective properties provide a supportive environment for follicle health.
Growth Hormone Secretagogues
Growth hormone (GH) and its downstream mediator IGF-1 (insulin-like growth factor 1) play well-documented roles in hair follicle biology. IGF-1 is produced by dermal papilla cells and acts as a major anagen-maintaining signal. Reduced IGF-1 signaling is associated with premature catagen entry and follicle miniaturization.
Several peptides that stimulate growth hormone release may indirectly support hair growth through increased systemic and local IGF-1 levels:
CJC-1295/Ipamorelin
CJC-1295/Ipamorelin is a combination of a growth hormone releasing hormone (GHRH) analog and a growth hormone secretagogue. Together, they stimulate pulsatile GH release, which in turn increases IGF-1 production. While no studies have specifically evaluated this combination for hair growth, the GH-IGF-1 axis is a known regulator of hair cycling, and optimization of this axis may support follicle health as part of a comprehensive protocol.
MK-677 (Ibutamoren)
MK-677 is an oral growth hormone secretagogue that increases GH and IGF-1 levels. Like CJC-1295/Ipamorelin, its potential benefit for hair growth is indirect, working through systemic GH-IGF-1 optimization rather than direct follicular targeting. Some users report improved hair quality and growth as a side benefit, though controlled studies specific to hair outcomes are lacking.
Matrixyl (Palmitoyl Pentapeptide-4)
Matrixyl is primarily known as an anti-aging skincare peptide, but its mechanism of action, stimulating collagen production and ECM remodeling, is relevant to hair follicle biology. The perifollicular ECM plays an active role in hair cycling, and compounds that support ECM quality may benefit follicle function.
Matrixyl works by mimicking a collagen fragment that signals fibroblasts to increase collagen production. In the context of hair growth, this could support the structural environment around follicles and maintain the ECM signaling that influences stem cell behavior and DP function.
SNAP-8 (Acetyl Octapeptide-3)
SNAP-8 is another skincare peptide with potential indirect relevance to scalp health. While primarily used for its muscle-relaxing effects on expression lines, SNAP-8's ability to modulate neuromuscular signaling in the skin may influence the scalp microenvironment. Its full research profile is available in our SNAP-8 research report.
NAD+ Precursors and Peptides
NAD+ (nicotinamide adenine dinucleotide) is a coenzyme essential for cellular energy production and DNA repair. NAD+ levels decline with age, and this decline has been implicated in stem cell aging across multiple tissue types, including hair follicle stem cells.
Research has shown that NAD+ supplementation can rejuvenate aged stem cells in animal models, restoring their regenerative capacity. In the context of hair follicles, this could mean improved stem cell activation and more efficient cycling. While direct hair growth studies with NAD+ are limited, the biological rationale is strong, and NAD+ optimization may complement peptide-based hair growth strategies, particularly in older individuals.
Epithalon (Epitalon)
Epithalon is a synthetic tetrapeptide that activates telomerase, the enzyme responsible for maintaining telomere length. Telomere shortening in hair follicle stem cells has been associated with reduced regenerative capacity and potentially contributes to age-related hair thinning.
While Epithalon hasn't been specifically studied for hair growth, its ability to support telomere maintenance in stem cells could theoretically extend the replicative lifespan of hair follicle stem cells, preserving their ability to regenerate the follicle through successive cycles.
Peptide Fragments and Growth Factors
Several growth factor-derived peptides are also under investigation for hair applications:
- FGF-derived peptides: Fragments of fibroblast growth factor that stimulate DP cell proliferation and anagen induction
- KGF (Keratinocyte Growth Factor) peptides: Target matrix keratinocytes to promote hair shaft production
- Wnt-mimetic peptides: Synthetic peptides that activate Wnt receptors directly, bypassing the natural ligand-receptor interaction
- Noggin-derived peptides: Antagonists of BMP signaling that remove another brake on follicle cycling (BMP signals promote quiescence)
These represent the next generation of targeted peptide therapeutics for hair growth. Most are in early preclinical stages, but they demonstrate the breadth of peptide-based approaches being explored.

Figure 5: Comparison of hair growth peptides and their primary mechanisms. Each peptide targets a different aspect of follicle biology, providing rationale for multi-peptide combination protocols.
Topical vs. Injectable Delivery for Hair Growth Peptides
How you deliver a peptide to the scalp matters as much as which peptide you choose. The delivery route determines how much active compound actually reaches the hair follicle, how deep it penetrates, how long it remains active at the target site, and what side effect profile you can expect. Each major delivery method has distinct advantages and trade-offs.
Topical Application
Topical delivery is the most accessible and widely used method for scalp peptide administration. Products range from simple serums and lotions to advanced formulations using nanotechnology for enhanced penetration.
Advantages
- Non-invasive: No needles, no pain, minimal barrier to consistent use
- Localized delivery: Peptide is applied directly where it's needed, minimizing systemic exposure
- Self-administrable: Can be incorporated into daily grooming routines without clinical visits
- Familiar format: Similar to other topical hair treatments (minoxidil), so patients already understand the application process
- Scalable coverage: Can treat the entire affected area in a single application
Limitations
- Stratum corneum barrier: The outer layer of skin is designed to keep things out, including therapeutic peptides. Even small peptides like GHK-Cu face resistance crossing this barrier
- Variable absorption: Penetration depends on scalp condition, formulation vehicle, application technique, and individual variation. Actual follicular delivery can be as low as 1-5% of the applied dose with basic formulations
- Concentration challenges: To compensate for limited penetration, higher concentrations may be needed, which can increase cost and the potential for local irritation
- Compliance demands: Topical treatments typically require daily or twice-daily application for months to see results. Many patients struggle with long-term compliance
Advanced Topical Delivery Technologies
Several technologies have been developed to overcome the limitations of basic topical formulations:
Microemulsions: Thermodynamically stable mixtures of oil, water, and surfactant that form nanoscale droplets capable of disrupting the stratum corneum barrier. Research published in Pharmaceutics showed that an ionic liquid microemulsion system for GHK-Cu significantly enhanced follicular penetration and Wnt/beta-catenin pathway activation compared to conventional aqueous solutions.
Nanoliposomes: Lipid-based vesicles (50-200 nm diameter) that encapsulate peptides and deliver them through the skin by fusing with cell membranes. A study in Colloids and Surfaces B demonstrated that nanoliposomal co-delivery of bioactive peptides produced superior hair growth stimulation in preclinical models.
Micelle systems: Self-assembling surfactant structures that solubilize peptides and enhance their skin penetration. These are simpler to manufacture than liposomes and can be incorporated into standard cosmetic formulations.
Penetration enhancers: Chemical agents like dimethyl sulfoxide (DMSO), ethanol, propylene glycol, and certain terpenes that temporarily increase stratum corneum permeability. These can significantly improve peptide delivery but may cause irritation at higher concentrations.
Microneedling-Assisted Delivery
Microneedling has emerged as one of the most effective methods for enhancing scalp peptide delivery. The technique uses an array of fine needles (typically 0.25-1.5 mm in length for scalp applications) to create transient microchannels through the stratum corneum and into the upper dermis.
Why Microneedling Works So Well for Peptides
The benefits of microneedling for peptide delivery are twofold:
Enhanced penetration: Microchannels bypass the stratum corneum barrier entirely, allowing peptides direct access to the viable epidermis and upper dermis where hair follicles reside. Studies have shown that microneedling can increase topical serum absorption by up to 400%, a dramatic improvement over passive diffusion.
Wound healing cascade: The controlled micro-injury triggers a natural wound healing response that releases platelet-derived growth factor (PDGF), vascular endothelial growth factor (VEGF), epidermal growth factor (EGF), and other regenerative signals. These endogenous growth factors complement the exogenous peptides being delivered, creating an additive or possibly complementary effect.
A landmark study by Dhurat et al. published in the International Journal of Trichology demonstrated that microneedling achieves an 82% patient response rate compared to 4.5% with topical treatment alone when used with minoxidil. While this study used minoxidil rather than peptides, the principle of enhanced delivery and complementary wound-healing signals applies equally to peptide formulations.
A 2025 study published in PMC examined enhanced hair regrowth using five monthly sessions of microneedling combined with a solution containing minoxidil, dutasteride, and copper peptides. Results assessed by both artificial intelligence analysis and blinded human evaluators showed significant improvements in hair density and coverage, supporting the combination approach.
Microneedling Protocol Considerations for Peptides
| Parameter | Recommendation | Rationale |
|---|---|---|
| Needle length | 0.5-1.0 mm for scalp | Deep enough to reach the dermis without excessive pain or bleeding |
| Frequency | Every 2-4 weeks | Allows complete healing between sessions while maintaining consistent stimulation |
| Peptide application timing | Immediately after microneedling | Microchannels begin closing within 30-60 minutes; apply peptides within this window |
| Device type | Derma pen (automated) preferred over roller | More consistent depth, less trauma, better for curved scalp surface |
| Coverage area | Entire affected zone plus 1-2 cm border | Addresses miniaturizing follicles at the periphery of thinning areas |
| Post-treatment care | Avoid washing for 4-6 hours; no harsh products for 24 hours | Allows peptides to absorb through channels before they close |
Injectable (Subcutaneous) Administration
Subcutaneous injection delivers peptides directly beneath the skin, bypassing all barriers to absorption. This method is most commonly used for TB-500, which has an established injectable protocol from its use in wound healing and tissue repair applications.
Advantages
- 100% bioavailability: The full dose reaches the target tissue, with no loss to the stratum corneum barrier
- Systemic distribution: Peptides injected subcutaneously enter the bloodstream and distribute throughout the body, providing both local and systemic effects
- Controlled dosing: Precise dose delivery with each injection
- Less frequent dosing: Many injectable peptides require only 1-3 injections per week, compared to daily topical application
Limitations
- Invasive: Requires self-injection or clinical administration, which some patients find aversive
- Systemic exposure: The peptide distributes throughout the body, not just to the scalp. This means lower concentration at the target site per unit dose and potential for systemic effects
- Injection site reactions: Pain, redness, swelling at the injection site are possible
- Storage requirements: Injectable peptides often require reconstitution and refrigeration
- Professional guidance recommended: Proper technique, sterile handling, and dose calculation benefit from professional oversight
Mesotherapy / Intradermal Injection
A specialized form of injectable delivery, mesotherapy involves multiple small-volume injections directly into the scalp dermis. This places the peptide in immediate proximity to the hair follicle, combining the bioavailability advantages of injection with the localized delivery benefits of topical application.
Mesotherapy is typically performed by trained clinicians using a multi-needle injector or individual small-gauge needles. Sessions are usually performed monthly, with each session involving dozens to hundreds of micro-injections across the treatment area. While more labor-intensive than subcutaneous injection, mesotherapy achieves the highest local peptide concentrations of any delivery method.
Delivery Method Comparison
| Parameter | Topical (Basic) | Topical (Advanced) | Microneedling + Topical | Subcutaneous | Mesotherapy |
|---|---|---|---|---|---|
| Follicular bioavailability | Low (1-5%) | Moderate (10-25%) | High (40-60%+) | Low (systemic distribution) | Very high (direct delivery) |
| Ease of use | Very easy | Easy | Moderate | Moderate | Requires clinician |
| Frequency | 1-2x daily | 1-2x daily | Weekly to monthly | 1-3x weekly | Monthly |
| Cost | Low | Moderate | Moderate | Moderate to high | High |
| Pain/discomfort | None | None | Mild to moderate | Mild (injection) | Moderate (multiple injections) |
| Systemic effects | Minimal | Minimal | Low | Yes | Low to moderate |
| Best suited for | GHK-Cu daily maintenance | GHK-Cu enhanced delivery | GHK-Cu, combination protocols | TB-500, BPC-157, GH peptides | GHK-Cu, combination cocktails |
For most people exploring peptide-based hair growth strategies, a combined approach works best: daily topical GHK-Cu for maintenance signaling, periodic microneedling sessions for enhanced delivery and wound-healing stimulation, and injectable TB-500 for systemic stem cell activation. Use our Dosing Calculator to estimate appropriate doses for your research protocol.
Application Protocols for Hair Growth Peptides
Translating research findings into practical protocols requires consideration of dosing, timing, delivery methods, and treatment duration. The following protocols are derived from the published literature, clinical practice patterns, and the pharmacological properties of each peptide. These are presented for informational purposes and should be discussed with a qualified healthcare provider.
GHK-Cu Topical Protocol
Topical GHK-Cu is the most straightforward hair growth peptide protocol and can be incorporated into a standard grooming routine.
Basic Protocol
- Concentration: 1-5% GHK-Cu in an appropriate vehicle (serum, microemulsion, or liposomal formulation)
- Frequency: Once or twice daily
- Application method: Apply to clean, towel-dried scalp. Part hair in rows and apply directly to exposed scalp skin. Gently massage for 1-2 minutes to distribute evenly across the treatment area.
- Treatment duration: Minimum 3-6 months for initial assessment. Hair follicle cycling occurs over months, not days, so patience is essential.
- Expected timeline: Reduced shedding within 4-8 weeks. Visible improvements in density and thickness at 3-6 months. Maximum benefit typically at 9-12 months.
Enhanced Protocol with Microneedling
- Microneedling sessions: Every 2-4 weeks using a derma pen with 0.5-1.0 mm needles
- Immediate post-needling: Apply GHK-Cu serum (or GHK-Cu combined with minoxidil) immediately after microneedling while microchannels are open
- Maintenance between sessions: Daily topical GHK-Cu application
- Duration: Minimum 6 sessions (6 months at monthly intervals) before assessing response
TB-500 Injectable Protocol
TB-500 protocols for hair growth are largely extrapolated from wound healing and tissue repair protocols, as no hair-specific clinical dosing guidelines exist.
Loading Phase (First 4-6 Weeks)
- Dose: 2.0-2.5 mg, administered subcutaneously
- Frequency: Twice weekly
- Injection site: Abdominal subcutaneous fat (standard), though some practitioners inject closer to the scalp (e.g., upper trapezius or shoulder region)
- Rationale: The loading phase establishes tissue saturation and initiates stem cell activation processes
Maintenance Phase (Ongoing)
- Dose: 2.0-2.5 mg, administered subcutaneously
- Frequency: Once weekly to once every two weeks
- Duration: 4-6 months minimum, with reassessment at regular intervals
Combined Multi-Peptide Protocol
The most comprehensive approach combines multiple peptides targeting different mechanisms. Here is an example framework:
| Component | Product | Route | Dose | Frequency |
|---|---|---|---|---|
| Primary topical | GHK-Cu Topical | Topical (scalp) | 1-2 mL of 2-5% serum | Daily (evening) |
| Stem cell activator | TB-500 | Subcutaneous injection | 2.0-2.5 mg | 2x/week (loading), then 1x/week |
| Anti-inflammatory | BPC-157 | Subcutaneous injection | 250-500 mcg | Daily or every other day |
| Microneedling | GHK-Cu + minoxidil solution | Topical post-microneedling | Per formulation | Every 3-4 weeks |
| Optional: GH support | CJC-1295/Ipamorelin | Subcutaneous injection | Per standard protocol | 5 nights/week before bed |
Protocol Timeline
Month 1-2: Begin daily topical GHK-Cu and TB-500 loading phase. First microneedling session at week 2-4. Expect initial shedding phase as dormant follicles are stimulated (this is normal and temporary). Month 3-4: Transition TB-500 to maintenance dosing. Continue monthly microneedling. Early signs of improvement may be visible: reduced shedding, improved hair texture, fine new growth at follicle sites. Month 5-8: Progressive improvement in density and coverage. Existing hairs thickening as follicles respond to improved signaling. Month 9-12: Maximum benefit from initial protocol. Photograph and measure results. Adjust protocol based on response.
Monitoring and Assessment
Objective assessment of hair growth peptide protocols requires consistent documentation:
- Standardized photography: Same lighting, angle, and camera settings. Monthly photos of affected areas from multiple angles.
- Trichoscopy: Dermoscopic examination to assess follicular density, hair shaft diameter, and vellus-to-terminal hair ratio. Professional trichoscopy provides quantitative data for tracking progress.
- Hair pull test: A simple clinical test where ~60 hairs are gently pulled. Fewer than 10% coming out is normal. Decreasing pull test counts over time suggest reduced shedding.
- Patient-reported outcomes: Subjective assessment of hair thickness, coverage, and shedding using standardized questionnaires.
- Digital analysis: AI-powered hair analysis tools can provide quantitative measurements of density, caliber, and growth rate from standardized photographs.
For access to dosing guidelines and protocol customization tools, visit our Dosing Calculator.
Comparison with Minoxidil, Finasteride & Conventional Treatments
Peptide-based hair growth approaches don't exist in a vacuum. They must be evaluated in the context of established treatments that have decades of clinical data behind them. Understanding how peptides compare to, and potentially complement, conventional therapies helps clinicians and researchers make informed decisions.
Minoxidil: The Established Vasodilator
Minoxidil (brand name Rogaine) was the first FDA-approved topical treatment for hair loss. Originally developed as an oral antihypertensive, its hair growth effects were discovered as a side effect. It's available over-the-counter in 2% and 5% topical solutions and a 5% foam formulation.
How Minoxidil Works
Minoxidil acts primarily as a potassium channel opener in vascular smooth muscle, causing vasodilation and increased blood flow. In hair follicles, it also appears to directly stimulate follicular cells, promote VEGF production, and extend the anagen growth phase. However, its exact mechanism in hair follicles isn't fully understood after decades of use.
Clinical Efficacy
Large clinical trials have demonstrated that minoxidil 5% produces visible hair regrowth in approximately 40-60% of men with androgenetic alopecia and a somewhat higher percentage of women. Response rates are highest in patients with recent-onset thinning and smaller affected areas. The treatment must be continued indefinitely; stopping results in loss of gains within 3-6 months.
Limitations
- Requires twice-daily application (solution) or once daily (foam)
- Initial shedding phase (2-8 weeks) discourages many users
- Efficacy plateaus at 12-18 months
- Scalp irritation, dryness, and flaking are common
- Unwanted facial hair growth (particularly in women)
- Rare systemic effects: hypotension, edema, tachycardia
- Does not address the underlying cause of androgenetic alopecia (DHT-mediated signaling disruption)
Finasteride: The DHT Blocker
Finasteride (brand name Propecia) is an oral 5-alpha-reductase inhibitor that reduces DHT levels by approximately 70%. It's FDA-approved for male pattern hair loss at a dose of 1 mg daily.
How Finasteride Works
By inhibiting the type II isoenzyme of 5-alpha-reductase, finasteride reduces the conversion of testosterone to DHT in the scalp and systemically. Lower DHT levels reduce the androgenic stimulus that drives follicle miniaturization. The drug addresses the hormonal root cause of androgenetic alopecia more directly than any other approved treatment.
Clinical Efficacy
Finasteride produces visible improvement in approximately 80-90% of men with androgenetic alopecia over 2 years, making it the most effective single agent currently available. Hair count increases of 10-15% are typical, with additional benefits in hair quality and diameter.
Limitations
- Sexual side effects (2-4% of users): decreased libido, erectile dysfunction, reduced ejaculate volume
- Post-finasteride syndrome (controversial but reported): persistent sexual and neurological symptoms after discontinuation
- Not approved for women (teratogenic risk)
- Takes 6-12 months to see full effects
- Must be taken indefinitely to maintain results
- Some patients are non-responders despite adequate DHT reduction
How Peptides Compare
| Attribute | GHK-Cu | TB-500 | PTD-DBM | Minoxidil 5% | Finasteride 1mg |
|---|---|---|---|---|---|
| Primary mechanism | Multi-pathway signaling | Stem cell activation | Wnt pathway de-repression | Vasodilation, VEGF | DHT reduction (70%) |
| Administration | Topical | Injectable (SC) | Topical | Topical | Oral (1mg/day) |
| FDA approved for hair | No | No | No | Yes | Yes (men only) |
| Evidence quality | Preclinical + small clinical | Preclinical (animal) | Preclinical (animal) | Multiple large RCTs | Multiple large RCTs |
| Response rate | Data limited | No human data | No human data | 40-60% | 80-90% |
| Systemic side effects | Not reported | Minimal reported | Unknown | Rare (cardiac) | Sexual (2-4%) |
| Addresses inflammation | Yes | Yes | No | No | No |
| ECM support | Yes | Yes | No | No | No |
| Suitable for women | Yes | Yes (with guidance) | Unknown | Yes (2% approved) | No (teratogenic) |
| Estimated monthly cost | $30-100 | $50-150 | Not commercially available | $10-30 | $10-30 (generic) |
Peptides as Complements, Not Replacements
Given the current state of evidence, the most rational approach positions peptides as complements to, rather than replacements for, established treatments. The mechanisms are largely non-overlapping, which creates opportunities for additive or complementary effects:
- Finasteride + GHK-Cu: Finasteride reduces the DHT driving miniaturization; GHK-Cu provides the pro-growth signals (Wnt activation, VEGF, ECM support) to help follicles recover once the hormonal insult is reduced
- Minoxidil + GHK-Cu: Preclinical data showing combined use produces greater hair count increases than either alone. Minoxidil provides vasodilation and direct follicular stimulation; GHK-Cu adds Wnt activation, anti-inflammatory effects, and ECM remodeling
- Finasteride + minoxidil + GHK-Cu + TB-500: The comprehensive approach addresses DHT (finasteride), blood flow (minoxidil), dermal papilla signaling (GHK-Cu), and stem cell activation (TB-500) simultaneously
This layered approach mirrors the trend in oncology and other fields where combination therapy targeting multiple pathways consistently outperforms monotherapy. Hair loss is a complex, multi-factorial condition; it makes sense that addressing multiple contributing mechanisms would produce better outcomes than targeting just one.
Emerging Conventional Treatments
it bears mention that peptides aren't the only emerging approach. Several other treatments are in development or gaining clinical adoption:
- Dutasteride: A more potent 5-alpha-reductase inhibitor (blocks both type I and II isoenzymes) showing superior efficacy to finasteride in some studies, but with a higher side effect profile
- Low-level laser therapy (LLLT): FDA-cleared devices using red and near-infrared light to stimulate follicles. Moderate evidence of efficacy, primarily for mild to moderate thinning
- Platelet-rich plasma (PRP): Concentrated platelets from the patient's own blood injected into the scalp. Contains a cocktail of growth factors. Growing clinical evidence supporting efficacy
- JAK inhibitors: Oral drugs like tofacitinib and baricitinib showing dramatic results in alopecia areata. Potential applications in other forms of hair loss under investigation
- Clascoterone: A topical antiandrogen (FDA-approved for acne) being investigated for androgenetic alopecia
Peptides may ultimately find their optimal role as components of comprehensive, multi-modal treatment strategies that also incorporate one or more of these conventional approaches. For the latest research updates, check our Biohacking Hub.
Figure 6: Treatment comparison matrix showing conventional treatments (minoxidil, finasteride) alongside peptide-based approaches (GHK-Cu, TB-500, PTD-DBM). Peptides offer complementary mechanisms that address pathways not targeted by existing FDA-approved treatments.
Copper Biology and Its Role in Hair Health
To fully appreciate why GHK-Cu works for hair growth, we need to understand the broader role of copper in human biology and specifically in the hair follicle microenvironment. Copper isn't just a passive component of the GHK-Cu complex; it's an essential trace element that participates in dozens of enzymatic reactions relevant to hair follicle function.
Copper as an Essential Trace Element
Copper is the third most abundant essential trace element in the human body, after iron and zinc. Adults carry approximately 75-150 mg of total body copper, distributed across the liver, brain, kidneys, and connective tissues. In the skin, copper concentrations are relatively high compared to many other tissues, reflecting its role in collagen cross-linking, melanin synthesis, and antioxidant defense.
Copper serves as a cofactor for at least 30 known enzymes, many of which are directly relevant to hair follicle biology:
- Lysyl oxidase: Essential for collagen and elastin cross-linking in the dermal sheath and perifollicular connective tissue. Without adequate copper, the structural scaffolding of the follicle degrades.
- Superoxide dismutase (Cu/Zn-SOD): A primary antioxidant enzyme that neutralizes superoxide radicals. Oxidative stress in the follicular microenvironment contributes to premature catagen entry and DP cell damage.
- Tyrosinase: The rate-limiting enzyme in melanin production. Copper deficiency leads to premature graying, one of the earliest signs of disrupted copper metabolism in hair.
- Cytochrome c oxidase: The terminal enzyme in the mitochondrial electron transport chain. Hair matrix cells are among the fastest-dividing cells in the body and have enormous energy demands that depend on functional mitochondria.
- Dopamine beta-hydroxylase: Involved in catecholamine synthesis. The sympathetic innervation of hair follicles influences cycling, and catecholamine signaling plays a role in the anagen-catagen transition.
Copper Deficiency and Hair Loss
Clinical copper deficiency, while relatively uncommon in well-nourished populations, produces striking hair and skin manifestations that underscore copper's importance for follicular function:
- Menkes disease: A genetic disorder of copper transport (ATP7A mutation) that causes severe copper deficiency. Affected infants have characteristic "kinky hair" (pili torti) that is sparse, coarse, twisted, and depigmented. The hair abnormality is directly attributable to impaired copper-dependent enzyme function in the follicle.
- Acquired copper deficiency: Can occur from excessive zinc supplementation (zinc competes with copper for absorption), gastric bypass surgery, or malabsorption syndromes. Hair changes include depigmentation (premature graying), thinning, and textural changes.
- Marginal copper status: Subclinical copper insufficiency may be more common than recognized, particularly in individuals with high zinc intake, restrictive diets, or chronic inflammatory conditions that sequester copper. Whether marginal copper deficiency contributes to common hair loss conditions is an area of active investigation.
The GHK-Cu complex provides copper in a biologically optimized format. Rather than free ionic copper (which can be pro-oxidant at high concentrations), GHK-Cu delivers copper bound to a peptide carrier that cells recognize and process through specific uptake mechanisms. This makes copper available for enzymatic functions while minimizing the risk of copper-mediated oxidative damage.
Copper-Dependent Processes in the Hair Follicle
Collagen Cross-Linking and Follicular Architecture
The hair follicle is embedded in a complex connective tissue framework that provides structural support and serves as a reservoir of signaling molecules. Collagen types I, III, and V form the primary structural proteins of this framework, and their proper assembly requires copper-dependent lysyl oxidase activity.
Lysyl oxidase catalyzes the oxidative deamination of lysine and hydroxylysine residues in collagen and elastin, creating reactive aldehydes that spontaneously cross-link to form the stable, mature connective tissue matrix. Without adequate lysyl oxidase activity, the perifollicular connective tissue becomes weak and disorganized, compromising the mechanical and signaling support that the follicle depends on.
GHK-Cu stimulates lysyl oxidase expression and activity, promoting proper collagen cross-linking in the perifollicular environment. This is one of the mechanisms by which GHK-Cu supports follicular architecture and may help resist the structural degradation that accompanies miniaturization.
Melanin Production and Hair Pigmentation
Hair color is determined by melanin pigments produced by melanocytes located in the hair matrix. Two types of melanin are responsible: eumelanin (brown/black) and pheomelanin (red/yellow). The ratio and total amount of these pigments determine the full spectrum of natural hair colors.
Tyrosinase, the rate-limiting enzyme in melanin synthesis, requires copper at its active site. Copper deficiency leads to reduced tyrosinase activity and subsequent hair depigmentation (graying). GHK-Cu's ability to deliver copper to melanocytes may support melanin production and potentially slow or partially reverse premature graying, though this application has received less research attention than its hair growth effects.
Interestingly, one of the earliest signs of hair follicle reactivation in GHK-Cu-treated animals is hyperpigmentation of newly growing hairs, suggesting that the copper delivered by GHK-Cu is readily incorporated into the melanin synthesis pathway.
Mitochondrial Function and Cellular Energy
Hair matrix cells divide approximately every 23 to 72 hours during anagen, making them one of the most rapidly proliferating cell populations in the body. This extraordinary proliferative rate demands correspondingly high cellular energy production, primarily through oxidative phosphorylation in mitochondria.
Cytochrome c oxidase (Complex IV), the terminal enzyme in the mitochondrial electron transport chain, contains two copper centers (CuA and CuB) that are essential for its function. Without adequate copper, Complex IV activity decreases, ATP production drops, and the cell's ability to sustain rapid division is compromised.
There's growing evidence that mitochondrial dysfunction in dermal papilla cells contributes to follicle miniaturization in androgenetic alopecia. DP cells from balding scalps show reduced mitochondrial membrane potential and lower ATP production compared to DP cells from non-balding scalp. GHK-Cu's delivery of copper to support mitochondrial function may help maintain the energy production capacity that DP cells and matrix cells need for sustained anagen activity.
Antioxidant Defense
The hair follicle exists in a challenging oxidative environment. Rapid cell division generates reactive oxygen species (ROS) as metabolic by-products. Exposure to UV radiation, environmental pollutants, and inflammatory mediators adds to the oxidative burden. If ROS levels exceed the follicle's antioxidant capacity, oxidative damage to DNA, proteins, and lipids can impair cell function and accelerate aging.
Copper-zinc superoxide dismutase (Cu/Zn-SOD, also known as SOD1) is a primary defense against superoxide radicals. This enzyme converts superoxide (a reactive and potentially damaging free radical) into hydrogen peroxide, which is then converted to water by catalase or glutathione peroxidase. Cu/Zn-SOD requires one copper and one zinc atom per subunit for catalytic activity.
GHK-Cu has been shown to upregulate SOD expression in skin cells, enhancing antioxidant defense. It also reduces levels of lipid peroxidation markers and protein oxidation products, indicating broad antioxidant protection. In the follicular context, this antioxidant support helps maintain the cellular integrity needed for normal cycling and hair production.
Age-Related Decline in Copper Peptides
One of the most intriguing aspects of GHK-Cu biology is its dramatic decline with age. Plasma GHK-Cu levels drop from approximately 200 ng/mL in young adults to about 80 ng/mL by age 60, representing a roughly 60% reduction. This decline parallels the age-related decrease in regenerative capacity across multiple tissues, including skin, bone, and hair.
The age-related decline in GHK-Cu may contribute to several features of aging hair:
- Reduced hair density (fewer active follicles per unit area)
- Decreased hair shaft diameter (follicle miniaturization)
- Slower hair growth rate (reduced matrix cell proliferation)
- Premature graying (reduced melanocyte function)
- Increased susceptibility to environmental damage (reduced antioxidant defense)
- Impaired wound healing and follicle regeneration after injury
Topical supplementation with GHK-Cu may partially compensate for this age-related decline, restoring local signaling to levels more characteristic of younger tissue. This "rejuvenation" rationale is distinct from, but complementary to, the specific anti-alopecia mechanisms discussed earlier.
Molecular Signaling in Hair Growth: A Detailed Analysis
For researchers and clinicians seeking a deeper understanding of how peptides interact with hair follicle signaling networks, this section provides a detailed analysis of the key molecular pathways and how each peptide intervention connects to the broader signaling architecture.
The Wnt/Beta-Catenin Pathway in Detail
The canonical Wnt pathway operates through a surprisingly elegant molecular switch that controls beta-catenin stability and nuclear translocation.
In the Absence of Wnt Signal (Pathway OFF)
When no Wnt ligand is bound to the Frizzled/LRP5/6 receptor complex, cytoplasmic beta-catenin is continuously degraded by a multi-protein complex called the "destruction complex." This complex consists of:
- Axin: The scaffold protein that assembles the destruction complex
- APC (Adenomatous Polyposis Coli): Tumor suppressor that helps recruit beta-catenin
- GSK-3beta (Glycogen Synthase Kinase-3 beta): Phosphorylates beta-catenin at specific serine and threonine residues
- CK1alpha (Casein Kinase 1 alpha): Primes beta-catenin for subsequent GSK-3beta phosphorylation
Phosphorylated beta-catenin is recognized by beta-TrCP, an E3 ubiquitin ligase adaptor, which tags it with ubiquitin for proteasomal degradation. The result: beta-catenin levels in the cytoplasm stay low, and none reaches the nucleus to activate target genes. The follicle remains quiescent.
In the Presence of Wnt Signal (Pathway ON)
When Wnt ligands bind the Frizzled/LRP5/6 complex, a conformational change activates the intracellular protein Dishevelled (Dvl). Active Dvl recruits the destruction complex to the cell membrane and disrupts its function. Specifically:
- Dvl polymerizes and recruits Axin to the membrane-bound receptor complex
- LRP5/6 is phosphorylated, creating a binding site for Axin
- Axin is sequestered at the membrane and can no longer scaffold the destruction complex
- Without a functional destruction complex, beta-catenin is no longer phosphorylated and degraded
- Beta-catenin accumulates in the cytoplasm
- Accumulated beta-catenin translocates to the nucleus
- In the nucleus, beta-catenin binds TCF/LEF transcription factors, displacing Groucho co-repressors
- The beta-catenin/TCF complex activates target genes including cyclin D1, c-Myc, Axin2, LEF1, and many others
These target genes drive cell proliferation, stem cell maintenance, and cell fate specification, all critical for anagen induction and hair shaft production.
Where CXXC5 Fits In (PTD-DBM Target)
CXXC5 acts as a negative feedback regulator by binding directly to the PDZ domain of Dishevelled. When CXXC5 is bound to Dvl, it prevents Dvl from performing its role in disrupting the destruction complex, even when Wnt ligands are present and Frizzled receptors are active. It's like having the right key but someone is blocking the lock.
In normal physiology, this negative feedback prevents excessive Wnt signaling, which could lead to uncontrolled proliferation. But in androgenetic alopecia, DHT and PGD2 drive CXXC5 overexpression, creating an abnormally strong brake on Wnt signaling. PTD-DBM's competitive inhibition of the CXXC5-Dvl interaction releases this brake, allowing normal Wnt signaling to resume.
Where GHK-Cu Acts on the Wnt Pathway
GHK-Cu's interaction with the Wnt pathway appears to operate at multiple levels:
- Wnt ligand expression: Gene expression studies suggest GHK-Cu upregulates expression of certain Wnt ligands (particularly Wnt10b and Wnt3a), increasing the activating signal at the receptor level
- DKK-1 suppression: GHK-Cu may downregulate DKK-1 (Dickkopf-1), a secreted Wnt antagonist that binds LRP5/6 and prevents Wnt ligand-receptor interaction. DKK-1 is elevated in balding scalps and is induced by DHT.
- Beta-catenin stabilization: Evidence of increased nuclear beta-catenin in GHK-Cu-treated DP cells suggests the peptide also influences beta-catenin stability or nuclear import, though the precise mechanism hasn't been fully elucidated
- GSK-3beta modulation: Some data suggests GHK-Cu may inhibit GSK-3beta activity, which would directly prevent beta-catenin phosphorylation and degradation
The BMP/Noggin Balance
Bone morphogenetic proteins (BMPs), particularly BMP-2 and BMP-4, are potent inhibitors of hair follicle cycling. They maintain stem cell quiescence in the bulge and prevent premature anagen induction. The counterbalance to BMP signaling is provided by BMP antagonists, of which Noggin is the best characterized in the follicular context.
The balance between BMPs and Noggin is critical for the timing of hair cycle transitions:
- High BMP, low Noggin: Stem cells remain quiescent. Follicle stays in telogen.
- Low BMP, high Noggin: Stem cell quiescence is released. Combined with Wnt activation, this triggers anagen induction.
The dermal papilla plays a central role in this balance, producing both BMPs and Noggin in a temporally regulated manner. During the late telogen "competent" phase, the DP increases Noggin expression, which, combined with Wnt signaling activation, creates the conditions for anagen induction.
While none of the peptides in this report have been shown to directly modulate BMP/Noggin balance, this pathway represents a potential target for future peptide therapeutics. Noggin-derived peptides or BMP-inhibiting peptides could add another layer to combination protocols.
The Sonic Hedgehog (Shh) Pathway
Sonic Hedgehog signaling is another critical pathway in hair follicle biology, particularly during follicle morphogenesis and cycling. Shh is produced by hair matrix cells and acts on surrounding mesenchymal cells to promote follicle downgrowth and formation of the hair shaft canal.
In the context of established follicles, Shh signaling is required for anagen progression. Matrix cell-derived Shh acts on the surrounding dermal sheath and subcutaneous adipose tissue, coordinating the cellular interactions needed for proper follicle cycling. Disruption of Shh signaling leads to follicle arrest and hair growth failure.
There's cross-talk between the Wnt and Shh pathways, with Wnt/beta-catenin signaling being required upstream of Shh expression. This means that peptides activating the Wnt pathway (GHK-Cu, PTD-DBM) may indirectly support Shh signaling as well, extending their influence beyond the immediate Wnt pathway targets.
Growth Factor Networks in the Dermal Papilla
The dermal papilla produces a complex cocktail of growth factors during anagen, and the composition of this cocktail determines follicle behavior. Understanding this network helps explain why multi-target approaches (like GHK-Cu, which influences multiple growth factors simultaneously) may be more effective than single-target interventions.
Pro-Growth Signals (Anagen-Promoting)
| Growth Factor | Function in Hair Follicle | Influenced by GHK-Cu? |
|---|---|---|
| VEGF | Perifollicular angiogenesis; nutrient supply to follicle | Yes (strongly upregulated) |
| HGF | Matrix cell proliferation; anagen induction | Yes (upregulated) |
| IGF-1 | Matrix cell survival; anagen maintenance | Indirect (via GH pathway) |
| FGF-7 (KGF) | Keratinocyte proliferation; hair shaft production | Possible (gene expression data) |
| FGF-10 | Outer root sheath growth; follicle elongation | Unknown |
| Wnt ligands | Stem cell activation; anagen induction | Yes (upregulated) |
| Noggin | BMP antagonism; releases stem cell quiescence | Unknown |
| PDGF | DP cell maintenance; follicle size determination | Unknown |
Pro-Regression Signals (Catagen-Promoting)
| Growth Factor | Function in Hair Follicle | Influenced by GHK-Cu? |
|---|---|---|
| TGF-beta1 | Matrix cell apoptosis; catagen induction | Yes (suppressed) |
| TGF-beta2 | Catagen induction; follicle regression | Yes (suppressed) |
| BMP-2/4 | Stem cell quiescence; anagen inhibition | Unknown |
| DKK-1 | Wnt antagonism; blocks anagen induction | Possible (suppressed) |
| IL-1alpha | Inflammatory catagen induction | Indirect (anti-inflammatory) |
| TNF-alpha | Inflammatory follicle damage | Yes (suppressed) |
| FGF-5 | Catagen induction signal | Unknown |
GHK-Cu's ability to simultaneously promote pro-growth signals (VEGF, HGF, Wnt ligands) while suppressing pro-regression signals (TGF-beta, TNF-alpha, potentially DKK-1) creates a net shift in the growth factor balance toward anagen induction and maintenance. This dual action, boosting the accelerator while releasing the brake, is a key feature that distinguishes GHK-Cu from treatments that act through a single mechanism.
The Androgen Signaling Cascade in Hair Loss
Understanding how androgens cause hair loss helps contextualize where peptide interventions fit in the treatment landscape.
The DHT-Mediated Miniaturization Pathway
- Testosterone circulates in the bloodstream and reaches the dermal papilla
- 5-alpha-reductase (primarily type II in the scalp) converts testosterone to DHT within the DP cell
- DHT binds to the androgen receptor (AR) in the DP cell cytoplasm
- The DHT-AR complex translocates to the nucleus
- The complex binds to androgen response elements (AREs) in target gene promoters
- This activates transcription of pro-miniaturization genes:
- TGF-beta1 and TGF-beta2 (catagen induction)
- DKK-1 (Wnt antagonist)
- IL-6 (pro-inflammatory cytokine)
- CXXC5 (Wnt pathway brake - the PTD-DBM target)
- And suppresses pro-growth genes:
- IGF-1 (growth factor)
- Wnt ligands (anagen induction signals)
- The net result is a hostile signaling environment that progressively shrinks the follicle
Finasteride intervenes at step 2 (blocking 5-alpha-reductase). Peptides intervene downstream:
- GHK-Cu counteracts the effects of steps 6 and 7 by boosting VEGF, suppressing TGF-beta, and activating Wnt signaling independent of the androgen cascade
- PTD-DBM specifically blocks the CXXC5 effect in step 6, restoring Wnt signaling despite elevated DHT
- TB-500 bypasses the DP signaling disruption entirely by directly activating stem cells, providing a growth signal independent of DP health
This is why combination approaches that include both an anti-androgen (finasteride or dutasteride) and downstream peptide support may produce the best outcomes: you're reducing the damaging signal at its source while simultaneously reinforcing the growth-promoting signals at multiple downstream points.
Prostaglandin Signaling and Hair Cycling
Prostaglandins are lipid-derived signaling molecules with diverse and sometimes opposing effects on hair follicles:
- PGD2: Hair growth inhibitor. Elevated in bald scalps. Acts through the DP2 receptor to induce catagen. PGD2 also upregulates CXXC5, creating a link to Wnt pathway suppression.
- PGE2: Hair growth promoter. Stimulates anagen induction and follicle growth. PGE2 levels are lower in balding vs. non-balding scalp.
- PGF2alpha: Also promotes hair growth. The bimatoprost analog latanoprost (a PGF2alpha analog) has been studied for eyebrow and scalp hair growth.
- 15d-PGJ2: A PGD2 metabolite that activates PPAR-gamma and may contribute to follicle miniaturization.
The PGD2-PGE2 ratio in the follicular microenvironment is an important determinant of follicle behavior. In healthy, non-balding scalp, PGE2 predominates and supports cycling. In balding scalp, PGD2 levels rise (driven by increased prostaglandin D2 synthase expression) while PGE2 levels fall, creating a pro-miniaturization environment.
GHK-Cu's anti-inflammatory effects may influence prostaglandin balance by reducing cyclooxygenase (COX) activity and shifting prostaglandin production away from PGD2 and toward PGE2. However, this has not been directly studied in the hair follicle context and remains a hypothesis based on GHK-Cu's known anti-inflammatory mechanisms in other tissues.
PTD-DBM is more directly relevant here: research has shown that PGD2-induced hair loss was restored by PTD-DBM treatment, confirming that the CXXC5-Dvl axis mediates at least part of PGD2's hair-inhibitory effect. By blocking CXXC5 function, PTD-DBM effectively neutralizes one of the key downstream consequences of elevated PGD2 in balding scalps.
Practical Considerations for Peptide-Based Hair Protocols
Moving from research to real-world application involves navigating practical challenges around product quality, storage, administration technique, and realistic expectations. This section addresses the nuts-and-bolts questions that arise when implementing peptide-based hair growth strategies.
Sourcing and Quality Assessment
The quality of peptide products varies enormously across the market. Unlike pharmaceutical drugs, research peptides and cosmetic peptide products are not subject to the same manufacturing standards, and this variability can significantly affect both safety and efficacy.
What to Look for in a Quality Peptide Product
- Certificate of Analysis (COA): A document from an independent analytical laboratory confirming the identity, purity, and potency of the peptide. Look for HPLC purity of 98% or higher, mass spectrometry confirmation of molecular weight, and endotoxin testing for injectable products.
- Third-party testing: COAs from in-house laboratories are less reliable than those from independent third parties. Reputable manufacturers use external labs to verify their products.
- GMP or GMP-equivalent manufacturing: Good Manufacturing Practice (GMP) standards ensure consistent quality across batches. While full pharmaceutical GMP isn't always required for research peptides, manufacturers following GMP-equivalent protocols produce more reliable products.
- Proper packaging: Peptides are sensitive to light, heat, and moisture. Quality products come in amber or opaque vials, sealed under inert gas (nitrogen or argon), and shipped with cold packs when temperature-sensitive.
- Clear labeling: Product name, peptide content (by weight), lot number, expiration date, and storage conditions should all be clearly stated.
FormBlends products meet these quality standards, with third-party COAs available for all peptide products. Quality assurance is especially important for injectable peptides where impurities can cause injection site reactions or worse.
Storage and Handling
Lyophilized (Freeze-Dried) Peptides
Most injectable peptides are supplied as lyophilized powder, which offers the best stability for long-term storage:
- Unopened: Store at -20 degrees C (freezer) for maximum shelf life (typically 2+ years). Room temperature storage is acceptable for shorter periods (weeks to a few months).
- Reconstituted: Use bacteriostatic water for reconstitution. Store reconstituted peptide at 2-8 degrees C (refrigerator). Use within 4-6 weeks of reconstitution.
- Avoid: Repeated freeze-thaw cycles, exposure to direct light, contamination from non-sterile needles or technique.
Topical Formulations
Topical GHK-Cu products have different storage requirements depending on formulation:
- Serum formulations: Typically stable at room temperature for 3-6 months after opening. Refrigeration extends stability. Discard if color changes significantly or unusual odor develops.
- pH sensitivity: GHK-Cu is most stable at slightly acidic pH (5.0-6.0). Formulations at neutral or alkaline pH may have reduced stability.
- Light protection: Store away from direct sunlight. Amber glass containers provide better protection than clear plastic.
Reconstitution and Injection Technique
For those using injectable peptides like TB-500 or BPC-157, proper technique is essential for safety and efficacy.
Reconstitution Steps
- Gather supplies: peptide vial, bacteriostatic water, alcohol swabs, appropriate syringes
- Clean the rubber stopper of both the peptide vial and bacteriostatic water vial with an alcohol swab
- Draw the desired amount of bacteriostatic water into a syringe (typically 1-2 mL for standard reconstitution)
- Insert the needle through the rubber stopper of the peptide vial at an angle
- Slowly inject the bacteriostatic water along the inside wall of the vial, letting it trickle down onto the lyophilized powder. Do NOT spray directly onto the powder or shake the vial, as this can damage the peptide.
- Gently swirl the vial until the powder is fully dissolved. This may take several minutes. Don't shake.
- Label the vial with the date of reconstitution and concentration
- Store in the refrigerator
Subcutaneous Injection
- Calculate the injection volume based on the reconstituted concentration and desired dose
- Draw the correct amount into an insulin syringe (29-31 gauge)
- Clean the injection site with an alcohol swab
- Pinch a fold of skin at the injection site (abdominal area is standard)
- Insert the needle at a 45-degree angle into the pinched skin fold
- Inject slowly
- Withdraw the needle and apply gentle pressure with the alcohol swab if needed
- Rotate injection sites to avoid tissue irritation from repeated injections in the same spot
Realistic Expectations and Timeline
One of the most common reasons people abandon peptide-based hair protocols is unrealistic expectations about the timeline and degree of response. Setting appropriate expectations from the start improves adherence and satisfaction.
What to Expect: Month by Month
| Timeframe | Expected Changes | What's Happening Biologically |
|---|---|---|
| Week 1-4 | Possible increase in shedding (temporary). Scalp may feel healthier. | Telogen follicles entering the exogen/early anagen transition. Old "club hairs" shed as new growth begins beneath. |
| Month 2-3 | Reduced daily shedding. Hair may feel thicker. Vellus hairs may appear in thinning areas. | Early anagen follicles beginning active growth. Improved DP signaling promoting thicker shaft production. |
| Month 4-6 | Visible improvement in density. New growth maturing. Existing hairs showing improved diameter. | Full anagen established in responding follicles. Improved vascular supply and ECM supporting larger follicle structures. |
| Month 7-9 | Continued progressive improvement. Coverage of thinning areas increasing. | Multiple hair cycles responding to improved signaling environment. Vellus-to-terminal hair conversion in some follicles. |
| Month 10-12 | Maximum benefit from initial protocol. Stabilization of gains. | Responding follicles have completed at least one full improved cycle. Non-responding follicles identified. |
Factors That Influence Response
- Duration of hair loss: More recent onset responds better. Long-standing complete baldness (where stem cells may be depleted) responds poorly or not at all.
- Norwood/Ludwig stage: Lower stages (mild to moderate thinning) respond better than advanced stages.
- Age: Younger individuals generally respond better, possibly due to higher endogenous stem cell reserves and better overall regenerative capacity.
- Type of hair loss: Androgenetic alopecia responds to the mechanisms targeted by these peptides. Other types (alopecia areata, scarring alopecias) may respond differently or not at all.
- Consistency: The single most important determinant of outcomes. Intermittent use produces intermittent results. Daily topical application and consistent injectable dosing are essential.
- Combination approach: Multi-target protocols (addressing hormonal, vascular, signaling, and stem cell pathways simultaneously) consistently outperform single-agent approaches.
- Lifestyle factors: Adequate nutrition (particularly protein, iron, zinc, biotin, and vitamin D), stress management, sleep quality, and overall health all influence hair growth potential.
When to Consider Alternative or Additional Interventions
Peptides aren't the answer for everyone, and it's important to recognize situations where alternative or additional interventions may be more appropriate:
- Advanced baldness (Norwood 6-7): When follicles have been miniaturized for years and stem cell reserves may be depleted, hair transplantation may be a more realistic path to visual improvement. Peptides can still be valuable for maintaining transplanted hair and supporting surrounding native follicles.
- Scarring alopecia: Conditions like lichen planopilaris, frontal fibrosing alopecia, and central centrifugal cicatricial alopecia destroy follicle stem cells. Peptides cannot regenerate destroyed follicles. These conditions require specific anti-inflammatory treatment.
- Alopecia areata: This autoimmune condition may partially respond to the anti-inflammatory effects of BPC-157 and the immune-modulating effects of Thymosin Beta-4, but the primary treatment targets are the aberrant immune response. JAK inhibitors have shown remarkable efficacy for this condition.
- Telogen effluvium: This common form of diffuse shedding triggered by stress, illness, or hormonal changes typically resolves on its own within 6-12 months. Peptides may accelerate recovery, but addressing the underlying trigger is the priority.
- Nutritional deficiencies: Iron deficiency, vitamin D deficiency, zinc deficiency, and protein malnutrition can all cause hair loss. No peptide will overcome an inadequate nutritional foundation. Blood work to check for deficiencies should be part of any hair loss evaluation.
For personalized guidance on which approach may be most appropriate for your situation, consider our Free Assessment as a starting point.
Cost Considerations
Peptide-based hair protocols represent a financial investment, and it's useful to understand the relative costs involved:
| Treatment Component | Estimated Monthly Cost | Notes |
|---|---|---|
| Topical GHK-Cu serum | $30-80 | Varies by concentration and formulation quality |
| TB-500 (maintenance dose) | $50-120 | Depends on dose and source |
| BPC-157 | $40-100 | Dose-dependent |
| CJC-1295/Ipamorelin | $80-200 | Optional systemic support |
| Microneedling device | $30-80 (one-time) + needle cartridges | Home use derma pen |
| Professional microneedling sessions | $150-400 per session | Monthly clinical sessions |
| Minoxidil 5% | $10-30 | Generic widely available |
| Finasteride 1mg | $10-30 | Generic widely available |
A basic topical-only protocol (GHK-Cu + minoxidil) runs approximately $40-110 per month. A comprehensive multi-peptide stack with professional microneedling could reach $400-900 per month. Most people find a middle-ground protocol that balances cost, convenience, and expected efficacy.
It's worth framing these costs against alternatives: a single hair transplant session typically costs $4,000-15,000 and may need to be repeated. Over a 2-3 year period, peptide therapy (even at the higher end) is comparable to or less expensive than surgical intervention, with the added benefit of maintaining and improving all existing follicles rather than just redistributing a limited donor supply.
Future Directions in Peptide Hair Growth Research
The field of peptide-based hair restoration is still in its early stages, with several promising developments on the horizon. Understanding where the research is heading helps contextualize current options and set expectations for future advances.
Next-Generation Delivery Systems
One of the biggest limitations of current topical peptide treatments is the stratum corneum barrier. Several advanced delivery technologies are in development to address this:
- Dissolving microneedle patches: Pre-loaded patches containing peptides embedded in dissolvable polymer needles that pierce the skin and release their payload as the needles dissolve. These could enable painless, at-home "microneedling" with precise peptide dosing.
- Exosome-based delivery: Natural or engineered extracellular vesicles (exosomes) that can transport peptide cargo across biological barriers. Exosomes derived from dermal papilla cells or mesenchymal stem cells are under investigation as both delivery vehicles and therapeutic agents in their own right.
- Follicular targeting: Nanoparticle systems designed to accumulate specifically in hair follicles, using the follicular opening as an entry route. This could dramatically improve the efficiency of topical peptide delivery while reducing off-target effects.
- Sustained-release implants: Biodegradable subcutaneous implants that release peptides over weeks to months, eliminating the need for frequent injections or daily topical application. This could solve the compliance challenge that undermines many treatment protocols.
Novel Peptide Targets
Research is identifying new molecular targets in hair follicle biology that could be addressed by designed peptides:
- TERT activation peptides: Telomerase reverse transcriptase (TERT) activation in follicular stem cells could extend their replicative lifespan and maintain regenerative capacity. Epithalon works through this mechanism, and more targeted peptides are in development.
- Prostaglandin D2 synthase inhibitors: Peptide-based inhibitors of the enzyme that produces PGD2 could reduce this hair-inhibitory prostaglandin directly at its source.
- Androgen receptor modulators: Selective androgen receptor modulators in peptide form could block DHT effects in the follicle without the systemic hormonal effects of finasteride.
- Autophagy modulators: Emerging research suggests that autophagy (cellular self-cleaning) plays a role in hair follicle cycling. Peptides that modulate autophagy in DP cells or stem cells could influence the cycling process.
- Senolytics: Peptide-based senolytic agents that selectively clear senescent cells from the follicular microenvironment could rejuvenate aged follicles. Senescent DP cells accumulate with age and in androgenetic alopecia, and their secretory phenotype (SASP) may contribute to miniaturization.
Combination with Cell-Based Therapies
The convergence of peptide therapy with cell-based approaches is an exciting frontier:
- DP cell expansion and reimplantation: Dermal papilla cells can be isolated, expanded in culture (potentially with GHK-Cu and TB-500 to maintain their inductivity), and reimplanted into miniaturized follicles to restore signaling capacity.
- Hair follicle organoids: Researchers can now grow rudimentary hair follicles in culture from combinations of epithelial and mesenchymal cells. Peptides could optimize the culture conditions and improve the maturation of these engineered follicles.
- iPSC-derived follicular cells: Induced pluripotent stem cells can be differentiated into follicular cell types. Peptides like Thymosin Beta-4 could guide the differentiation process toward functional hair-producing cells.
Clinical Trial Landscape
The biggest gap in the current evidence base is the lack of large-scale, randomized, controlled clinical trials for hair growth peptides. Filling this gap will require:
- Standardized outcome measures: Consistent use of trichoscopy, standardized photography, and validated patient-reported outcomes across studies
- Adequate sample sizes: Moving beyond small pilot studies to adequately powered trials with 100+ participants
- Long-term follow-up: 12+ month trials to capture the full hair cycling response
- Head-to-head comparisons: Direct comparison of peptides versus established treatments (minoxidil, finasteride) and versus each other
- Combination trial designs: Testing multi-peptide and peptide + conventional treatment combinations to identify optimal protocols
Until these trials are completed, the evidence for hair growth peptides will remain primarily preclinical, supplemented by small clinical studies and empirical clinical observation. This doesn't mean the compounds don't work, but it means the certainty of their effects hasn't been established to the same standard as FDA-approved treatments.
Research Opportunity
The hair growth peptide field is ripe for well-designed clinical trials. The preclinical evidence is strong enough to justify human studies, the safety profiles are favorable, and patient demand is enormous. Researchers and clinicians interested in contributing to this evidence base can explore our research resources at the Peptide Research Hub.
GHK-Cu Gene Expression Profile: What 4,000+ Genes Mean for Hair
One of the most striking features of GHK-Cu is the sheer breadth of its genomic effects. Gene expression profiling studies using the Broad Institute's Connectivity Map (cMap) database have revealed that GHK-Cu modulates the expression of over 4,000 human genes, roughly 6% of the entire genome. Understanding which of these genes are relevant to hair follicle biology provides deeper insight into why this small peptide has such broad regenerative effects.
Overview of Gene Modulation
In their comprehensive analysis published in the International Journal of Molecular Sciences, Pickart and colleagues examined GHK-Cu's gene expression signature and identified several functional categories of particular interest. Of the 4,000+ modulated genes, a significant proportion fall into categories directly relevant to hair follicle function:
- Tissue remodeling genes (531 genes): Including collagen synthesis, ECM assembly, matrix metalloproteinase regulation, and structural protein production. These genes govern the physical architecture of the follicle and its surrounding connective tissue.
- Anti-inflammatory genes (112 genes): GHK-Cu stimulates genes that suppress inflammatory signaling (including NF-kB pathway components, inflammatory cytokine inhibitors, and oxidative stress defense genes) while suppressing pro-inflammatory gene expression.
- Antioxidant defense genes (41 genes): Including superoxide dismutase (SOD1, SOD2), catalase, glutathione peroxidase family members, and thioredoxin reductase. These enzymes protect follicular cells from oxidative damage that contributes to premature aging and cycling disruption.
- Stem cell-related genes (47 genes): Including genes involved in stem cell self-renewal, asymmetric division, niche signaling, and differentiation. These are directly relevant to the bulge stem cell population that drives follicle regeneration.
- Apoptosis-regulating genes (71 genes): GHK-Cu promotes pro-survival gene expression while suppressing pro-apoptotic genes, supporting cell survival in the dermal papilla and matrix during the stresses of rapid cell division.
- DNA repair genes (48 genes): Hair matrix cells' rapid division makes them vulnerable to DNA replication errors. Enhanced DNA repair capacity helps maintain cellular fitness through many division cycles.
Hair-Relevant Gene Expression Changes
Collagen and ECM Genes
GHK-Cu upregulates expression of collagen types I, III, V, and VII, along with elastin, fibronectin, and several proteoglycans. In the hair follicle context, these ECM components form the dermal sheath, the perifollicular connective tissue, and the specialized basement membrane that separates epithelial and mesenchymal compartments.
Particularly significant is the upregulation of versican (VCAN), a large chondroitin sulfate proteoglycan. Versican is highly expressed in the dermal papilla during anagen and has been identified as a necessary component of the signaling environment that induces anagen. Its expression in the DP correlates with follicle size, and loss of versican expression is associated with follicle miniaturization. GHK-Cu's promotion of versican production may be a key mechanism by which it supports follicle size maintenance.
Decorin, another proteoglycan upregulated by GHK-Cu, binds and sequesters TGF-beta in the ECM, reducing its bioavailability. This provides an additional mechanism for TGF-beta suppression beyond direct transcriptional effects, creating a local extracellular sink for this catagen-inducing growth factor.
Growth Factor Gene Expression
Beyond the well-documented VEGF upregulation, GHK-Cu influences expression of several other growth factors relevant to hair biology:
- FGF-2 (bFGF): Basic fibroblast growth factor supports DP cell proliferation and survival. GHK-Cu promotes FGF-2 expression, which may contribute to maintaining DP cell numbers during follicle cycling.
- PDGF: Platelet-derived growth factor receptors are expressed on DP cells, and PDGF signaling is involved in DP cell recruitment and maintenance. GHK-Cu modulates PDGF pathway components.
- EGF family members: Epidermal growth factor family ligands regulate keratinocyte proliferation and differentiation in the matrix and outer root sheath. GHK-Cu influences expression of several EGF family members.
- Angiopoietins: These vascular growth factors work alongside VEGF to stabilize and mature new blood vessels. GHK-Cu's promotion of angiopoietin expression helps ensure that the VEGF-driven angiogenesis produces functional, mature vasculature rather than leaky, immature vessels.
Inflammation-Related Genes
The anti-inflammatory gene expression signature of GHK-Cu is particularly relevant to hair loss conditions involving perifollicular inflammation:
- NF-kB pathway suppression: GHK-Cu downregulates several components of the NF-kB inflammatory signaling cascade, including IKK-beta and several NF-kB target genes. NF-kB activation in hair follicles is associated with premature catagen induction and follicle damage.
- Interleukin modulation: GHK-Cu suppresses expression of pro-inflammatory interleukins (IL-1, IL-6, IL-8) while promoting expression of anti-inflammatory interleukins (IL-10). This shifts the local cytokine balance away from inflammation and toward tissue homeostasis.
- TNF-alpha suppression: Tumor necrosis factor alpha is a potent inducer of catagen and follicle regression. GHK-Cu's suppression of TNF-alpha expression helps protect follicles from cytokine-mediated damage.
- COX-2 modulation: Cyclooxygenase-2 is the enzyme responsible for producing prostaglandins during inflammation. GHK-Cu's modulation of COX-2 expression may influence the PGD2/PGE2 balance in the follicular microenvironment.
Ubiquitin-Proteasome Pathway Genes
An often-overlooked aspect of GHK-Cu's genomic effects is its modulation of the ubiquitin-proteasome system (UPS), the cellular machinery responsible for targeted protein degradation. The UPS plays a critical role in Wnt signaling, as beta-catenin is degraded via ubiquitin-mediated proteolysis when the destruction complex is active.
GHK-Cu modulates several UPS components, and this may contribute to its Wnt pathway effects. By adjusting the efficiency of beta-catenin ubiquitination and degradation, GHK-Cu could shift the balance toward beta-catenin accumulation even without directly activating Wnt ligand-receptor signaling. This is a subtle but potentially important mechanism that deserves further investigation.
Gene Expression Patterns vs. Single-Target Drugs
The comparison between GHK-Cu's gene expression profile and those of single-target drugs is instructive. While a drug like finasteride affects a relatively narrow set of genes downstream of 5-alpha-reductase inhibition, GHK-Cu simultaneously adjusts thousands of genes across multiple functional categories.
This breadth can be viewed as both a strength and a limitation:
- Strength: The broad gene modulation pattern means GHK-Cu addresses multiple aspects of hair follicle biology simultaneously. It doesn't just block one pathway; it reorients the entire cellular program toward a more regenerative state. This may explain why GHK-Cu shows effects across different types of hair loss and why it complements single-target treatments.
- Limitation: Broad gene modulation raises questions about unintended effects. While the documented gene changes are overwhelmingly consistent with beneficial tissue remodeling and regeneration, the possibility of unexpected effects in certain contexts (particular cell types, disease states, or genetic backgrounds) cannot be ruled out based on current data.
The fact that GHK-Cu is a naturally occurring molecule that the body already produces and responds to provides some inherent safety reassurance. The gene expression changes it produces aren't foreign perturbations; they're a return to the expression patterns characteristic of younger, more regenerative tissue. This "rejuvenation" interpretation of GHK-Cu's genomic effects is consistent with its age-related decline and the correlation between declining GHK-Cu levels and declining regenerative capacity.
Implications for Personalized Medicine
As genetic profiling becomes more accessible, it may eventually be possible to identify individuals whose gene expression patterns are most amenable to GHK-Cu treatment. For example, individuals with particularly strong suppression of Wnt pathway genes, elevated inflammatory gene expression, or reduced antioxidant gene activity might be predicted to respond especially well to GHK-Cu's broad normalizing effects.
Similarly, pharmacogenomic analysis might identify genetic variants that affect GHK-Cu metabolism, receptor interactions, or downstream gene responses, enabling more precise dosing and personalized protocol optimization. This remains a future direction rather than a current clinical reality, but it represents the logical endpoint of integrating genomic data with peptide therapeutics.
For now, the practical takeaway is that GHK-Cu's effects on hair follicles aren't limited to a single mechanism or pathway. Its genomic fingerprint reveals a comprehensive program of tissue rejuvenation that addresses inflammation, oxidative damage, ECM degradation, growth factor deficiency, and signaling pathway disruption simultaneously. This breadth of action is the molecular explanation for the clinical observation that GHK-Cu produces improvements across multiple aspects of hair quality, density, and growth rate.
Evaluating Hair Growth Research: A Guide to Understanding the Evidence
The hair growth industry is rife with exaggerated claims and cherry-picked data. Knowing how to evaluate research quality helps distinguish genuinely promising interventions from marketing hype. This section provides a framework for critically assessing the evidence behind any hair growth peptide or treatment.
The Hierarchy of Evidence
Not all evidence is created equal. Understanding the evidence hierarchy helps put peptide research in context:
- Systematic reviews and meta-analyses of randomized controlled trials represent the highest level of evidence. These pool data from multiple trials to generate more strong conclusions. No systematic review exists for hair growth peptides specifically.
- Randomized controlled trials (RCTs) with adequate sample sizes and blinding are the gold standard for individual studies. Minoxidil and finasteride have multiple large RCTs. GHK-Cu has small RCTs. TB-500 and PTD-DBM have none for hair indications.
- Controlled but non-randomized studies provide useful data but are more susceptible to bias. Some GHK-Cu hair studies fall into this category.
- Animal studies demonstrate biological plausibility and mechanism but don't guarantee human efficacy. TB-500 and PTD-DBM hair evidence is primarily at this level.
- In vitro (cell culture) studies demonstrate mechanism at the cellular level but can't account for the complexity of whole-organism biology. Much of the GHK-Cu dermal papilla research is in vitro.
- Case reports and clinical observations provide preliminary signals but lack controls and are subject to placebo effects and selection bias.
- Expert opinion and mechanistic reasoning are the lowest level of evidence but still have value for hypothesis generation and protocol design.
Common Pitfalls in Hair Growth Research
Small Sample Sizes
Many peptide hair studies involve fewer than 50 participants, and some involve fewer than 20. Small studies are vulnerable to random variation, placebo effects, and selection bias. A "significant" result in a 15-person study might not replicate in a 200-person study. When evaluating claims, always note the sample size and be appropriately cautious with small studies.
Short Follow-Up Periods
Hair follicle cycling occurs over months to years. Studies with follow-up periods of less than 6 months may capture only early responses (or even the initial shedding phase), missing the full treatment effect. The most informative studies run for 12-24 months with multiple assessment timepoints.
Lack of Placebo Controls
Uncontrolled studies can't distinguish treatment effects from placebo response, natural fluctuation, regression to the mean, or concurrent lifestyle changes. The placebo response in hair loss studies is well-documented: approximately 3-10% of placebo-treated participants show measurable hair count improvements in controlled trials. Any uncontrolled study showing improvements below this threshold might simply be capturing placebo effects.
Subjective Outcomes
Studies relying solely on participant self-assessment ("Do you feel your hair is thicker?") are susceptible to placebo effects and expectation bias. The most reliable studies use objective measures: standardized macrophotography, trichoscopy with quantitative analysis, hair pull test counts, or AI-assisted image analysis.
Publication Bias
Studies showing positive results are more likely to be published than studies showing no effect. This creates a systematic overestimation of treatment efficacy in the published literature. For peptides with a small number of published studies, this bias may be particularly relevant.
Conflict of Interest
Studies funded by manufacturers of the product being tested are more likely to report favorable results. While industry funding doesn't automatically invalidate a study, it's a factor to consider when evaluating the evidence. Look for independent replications of manufacturer-funded findings.
How to Read a Hair Growth Study
When evaluating a study on any hair growth treatment, ask these questions:
- What was the study design? RCT > controlled > uncontrolled > case report
- How many participants? More is better. Under 30 warrants particular caution.
- Was it blinded? Double-blind (neither participants nor investigators know treatment assignment) minimizes bias.
- What were the outcome measures? Objective (hair count, trichoscopy) > subjective (self-report)
- How long was follow-up? At least 6 months for meaningful hair growth assessment.
- Was there a control group? And was the control group appropriate (true placebo, not just "untreated")?
- Who funded the study? Independent funding reduces potential bias.
- Has the result been replicated? Single studies, however impressive, are less reliable than replicated findings.
- What was the effect size? Statistical significance doesn't equal clinical significance. A "statistically significant" 2% improvement in hair count might not be visible to the naked eye.
- What population was studied? Results in one demographic may not generalize to others. Results in mice may not apply to humans.
Current Evidence Summary by Peptide
| Peptide | Highest Evidence Level Available | Number of Hair-Specific Studies | Overall Evidence Strength |
|---|---|---|---|
| GHK-Cu | Small controlled clinical studies | 10+ (in vitro + animal + clinical) | Moderate |
| Thymosin Beta-4 / TB-500 | Animal studies (rodent) | 5-8 (in vitro + animal) | Moderate (preclinical) |
| PTD-DBM | Animal studies (rodent) | 3-5 (in vitro + animal) | Preliminary (preclinical) |
| BPC-157 | No hair-specific studies | 0 (rationale from mechanism) | Theoretical |
| CJC-1295/Ipamorelin | No hair-specific studies | 0 (rationale from GH-IGF-1 biology) | Theoretical |
This evidence summary underscores an important point: the hair growth peptide field is evidence-informed rather than evidence-proven. The biological rationale is strong, the preclinical data is encouraging, and early clinical results are promising. But the definitive large-scale clinical trials that would place peptides on the same evidentiary footing as minoxidil and finasteride have not yet been conducted.
This doesn't mean peptides don't work. It means we can't say with the same confidence exactly how well they work, for whom they work best, or what the optimal protocols are. Ongoing research and clinical experience will progressively fill these gaps. In the meantime, peptide-based approaches represent a rational, mechanism-based strategy that complements established treatments and addresses pathways they don't reach.
For researchers interested in contributing to the evidence base, or for clinicians seeking to stay current with emerging data, our Peptide Research Hub provides regularly updated summaries of new publications and clinical findings.
Types of Hair Loss: Where Peptides Apply and Where They Don't
Not all hair loss is created equal. Different conditions involve different pathological mechanisms, and peptides are more suited to some types than others. Understanding which forms of hair loss are most amenable to peptide intervention helps set realistic expectations and guides treatment selection.
Androgenetic Alopecia (Pattern Hair Loss)
Androgenetic alopecia (AGA) is by far the most common form of hair loss, affecting approximately 50% of men over 50 and 25% of women over 50. It's the condition most commonly targeted by hair growth peptides, and for good reason: AGA involves progressive follicle miniaturization driven by signaling dysfunction that peptides can directly address.
Male Pattern Hair Loss
Male AGA follows a predictable pattern classified by the Norwood-Hamilton scale (stages I through VII). Hair loss typically begins at the temples and vertex (crown), progressively expanding until only a horseshoe-shaped fringe of hair remains around the sides and back of the head in advanced stages.
The mechanism involves DHT-mediated disruption of dermal papilla signaling in genetically susceptible follicles. Critically, the follicles in the occipital region (back of the head) are genetically resistant to DHT, which is why they're used as donor follicles in hair transplantation.
Peptide applicability in male AGA:
- Norwood I-III (mild): Best candidates. Follicles are still largely intact, stem cells are present, and signaling restoration through peptides can produce meaningful reversal. GHK-Cu topical plus microneedling is a reasonable first-line peptide approach.
- Norwood III-V (moderate): Good candidates. More aggressive protocols (multi-peptide stacking, finasteride combination) are typically needed. Follicle miniaturization is more advanced but still reversible in many cases.
- Norwood V-VII (advanced): Limited peptide benefit for areas of long-standing complete baldness. Peptides may help thicken remaining hair and slow progression at the margins. Hair transplantation may be more appropriate for restoring coverage in extensively bald areas.
Female Pattern Hair Loss
Female AGA typically presents as diffuse thinning over the crown and frontal scalp, classified by the Ludwig scale (grades I through III). Unlike male AGA, complete baldness is rare; instead, the part line progressively widens and the scalp becomes increasingly visible through thinning hair.
The hormonal mechanism in women is less clear-cut than in men. While androgens play a role, other factors including estrogen decline (particularly around menopause), iron deficiency, thyroid dysfunction, and stress are often contributory. The multi-target nature of peptides like GHK-Cu, addressing Wnt signaling, VEGF, inflammation, and ECM quality simultaneously, may be particularly well-suited to the multi-factorial nature of female hair loss.
An important practical advantage of peptide approaches for women: GHK-Cu and TB-500 have no anti-androgenic hormonal effects, making them suitable for premenopausal women where finasteride is contraindicated due to teratogenic risk.
Telogen Effluvium
Telogen effluvium (TE) is a common form of diffuse hair shedding caused by a systemic trigger that pushes a large proportion of follicles into the telogen (resting) phase simultaneously. Common triggers include:
- Major physiological stress (surgery, severe illness, high fever)
- Emotional stress
- Nutritional deficiencies (iron, zinc, protein, vitamin D)
- Hormonal changes (postpartum, thyroid disorders, starting/stopping birth control)
- Medications (certain antidepressants, beta-blockers, retinoids)
- Crash dieting or significant weight loss
The shedding typically begins 2-3 months after the triggering event, reflecting the time it takes for the prematurely induced telogen follicles to complete the resting phase and shed their hairs. TE usually resolves on its own within 6-12 months once the trigger is removed, as follicles gradually cycle back into anagen.
Peptides may accelerate recovery from TE by promoting the telogen-to-anagen transition. TB-500's stem cell activation mechanism is particularly relevant here, as the primary problem in TE is getting resting follicles back into active growth. GHK-Cu's Wnt activation and VEGF promotion also support anagen re-entry. However, addressing the underlying trigger remains the first priority.
Alopecia Areata
Alopecia areata (AA) is an autoimmune condition in which the immune system attacks hair follicles, producing characteristic round patches of hair loss. In severe cases, it can progress to alopecia totalis (complete scalp hair loss) or alopecia universalis (complete body hair loss).
The primary pathology in AA is collapse of follicular immune privilege, allowing cytotoxic T cells to attack the hair bulb. The follicle enters a dystrophic catagen, and the stem cells in the bulge are spared (explaining why regrowth is possible once the immune attack subsides).
Peptide applicability in AA is limited but not zero:
- TB-500's anti-inflammatory and immune-modulating properties may provide some benefit as an adjunct to primary immunosuppressive therapy
- BPC-157's anti-inflammatory effects could support follicular immune privilege restoration
- GHK-Cu may help maintain follicle health during the autoimmune attack and support recovery once the immune component is controlled
- None of these peptides are sufficient as monotherapy for AA. JAK inhibitors (tofacitinib, baricitinib, ritlecitinib) have shown dramatic efficacy in clinical trials and represent the current frontier of AA treatment.
Scarring (Cicatricial) Alopecias
Scarring alopecias include a group of conditions where inflammatory or autoimmune processes destroy the hair follicle permanently, replacing it with scar tissue. Examples include lichen planopilaris, frontal fibrosing alopecia, central centrifugal cicatricial alopecia, and discoid lupus erythematosus.
Peptides are generally NOT effective for scarring alopecias because:
- The stem cell niche in the bulge is destroyed, removing the source material for follicle regeneration
- The follicular structure is replaced by fibrotic scar tissue that cannot be reversed by peptide signaling
- The inflammatory process is usually severe and requires immunosuppressive therapy beyond what peptides provide
The one potential role for peptides in scarring alopecias is at the active advancing border, where follicles are under attack but not yet destroyed. Anti-inflammatory peptides like BPC-157, combined with prescription anti-inflammatory medications, might help protect at-risk follicles. However, this is speculative and not supported by clinical evidence.
Traction Alopecia
Traction alopecia results from chronic tension on hair follicles, typically from tight hairstyles (braids, ponytails, cornrows, extensions). Repeated tension damages the follicle attachment and eventually leads to permanent loss if the causative styling practices continue.
In early stages (before permanent scarring), traction alopecia is potentially addressable with peptides after the causative tension is eliminated. GHK-Cu's tissue repair and ECM remodeling effects could support follicular recovery. TB-500's wound healing properties might accelerate the repair process. However, the most important intervention is simply stopping the traction.
Chemotherapy-Induced Alopecia
Chemotherapy drugs target rapidly dividing cells, and hair matrix cells (with their 23-72 hour division cycle) are among the most sensitive. Most chemotherapy-induced alopecia is temporary, with regrowth beginning 3-6 months after treatment completion. However, some patients experience persistent alopecia with altered hair texture and reduced density.
Peptide use during active chemotherapy is generally not recommended, as growth-promoting peptides could theoretically counteract the cytotoxic effects of chemotherapy on cancer cells. After chemotherapy completion, however, peptides like GHK-Cu and TB-500 may help accelerate and improve the quality of hair regrowth by supporting follicular stem cell activation and DP recovery. This application has not been clinically studied but has biological plausibility.
Age-Related Hair Thinning
Even without androgenetic alopecia, hair naturally thins with age. This age-related thinning involves reduced follicle density, decreased hair shaft diameter, slower growth rate, and prolonged telogen phases. Contributing factors include:
- Declining growth hormone and IGF-1 levels
- Reduced stem cell reserves and regenerative capacity
- Accumulated oxidative damage to DP cells and stem cells
- Decreased local blood flow
- ECM degradation
- Declining endogenous GHK-Cu levels (60% reduction by age 60)
Age-related thinning is arguably the strongest use case for GHK-Cu, as topical supplementation directly addresses the decline in endogenous levels. Combined with NAD+ for cellular energy support and CJC-1295/Ipamorelin for GH/IGF-1 optimization, a comprehensive anti-aging approach to hair maintenance is possible.
Nutritional and Lifestyle Foundations for Peptide-Supported Hair Growth
Peptides work best when the body's nutritional and metabolic foundations are solid. No peptide can overcome severe nutritional deficiencies, chronic sleep deprivation, or unmanaged stress. This section covers the lifestyle factors that maximize the effectiveness of peptide-based hair protocols.
Essential Nutrients for Hair Growth
Hair matrix cells are among the fastest-dividing cells in the body, and their metabolic demands for vitamins, minerals, amino acids, and energy substrates are correspondingly high. Deficiencies in any of these can limit hair growth regardless of what peptides you're using.
Iron
Iron deficiency is one of the most common nutritional causes of hair loss worldwide, particularly in premenopausal women. Even serum ferritin levels in the "low normal" range (below 40-70 ng/mL) have been associated with increased hair shedding. Iron is required for DNA synthesis in rapidly dividing matrix cells and for cytochrome oxidase function in the mitochondrial electron transport chain.
Before starting a peptide protocol, check serum ferritin levels. If below 70 ng/mL, iron supplementation may improve hair growth independent of peptide use. If below 20 ng/mL, iron supplementation should be a priority before or alongside peptide therapy.
Zinc
Zinc is a cofactor for over 300 enzymes and is essential for DNA replication, protein synthesis, and cell division. Zinc deficiency causes telogen effluvium and brittle, fragile hair. Zinc is also involved in 5-alpha-reductase enzyme function, and zinc supplementation has been studied (with mixed results) as a complement to finasteride for androgenetic alopecia.
An important caveat: excessive zinc supplementation (above 40 mg/day long-term) can induce copper deficiency by competing for absorption in the gut. Given that copper is central to GHK-Cu's mechanism, zinc/copper balance should be carefully managed. A common recommendation is to take copper (1-2 mg) alongside zinc supplements exceeding 15 mg/day.
Vitamin D
Vitamin D receptors are expressed in hair follicle keratinocytes, and vitamin D signaling is involved in hair cycle regulation. Vitamin D deficiency (below 30 ng/mL serum 25-hydroxyvitamin D) has been associated with telogen effluvium, alopecia areata, and possibly androgenetic alopecia. Given that vitamin D deficiency is extremely common (estimated 40-50% of the global population), checking and optimizing vitamin D levels is a high-yield intervention.
Biotin (Vitamin B7)
Biotin deficiency causes hair loss, but true biotin deficiency is rare in well-nourished individuals. The widespread supplementation of biotin for hair growth is based more on marketing than evidence, as studies show benefit only in cases of actual deficiency. High-dose biotin supplementation can interfere with certain laboratory tests (thyroid function, cardiac troponin), potentially leading to misdiagnosis.
Recommendation: Test for biotin deficiency before supplementing. If levels are normal, additional biotin is unlikely to improve hair growth.
Protein and Amino Acids
Hair is approximately 95% keratin, a protein. Adequate dietary protein is essential for providing the amino acids that matrix cells use to build the hair shaft. Protein malnutrition is a known cause of hair loss (kwashiorkor-associated alopecia) and insufficient protein intake can limit the effectiveness of even the best peptide protocol.
Key amino acids for hair include cysteine (the dominant amino acid in keratin), methionine (essential amino acid and cysteine precursor), lysine (part of the GHK-Cu peptide sequence), and arginine (involved in nitric oxide production and blood flow).
Recommended protein intake for hair growth: 1.0-1.2 g per kg of body weight daily, with emphasis on complete protein sources containing all essential amino acids.
Omega-3 Fatty Acids
Omega-3 fatty acids (EPA and DHA) have anti-inflammatory effects that may benefit the perifollicular environment. A randomized controlled trial published in the Journal of Cosmetic Dermatology found that omega-3 supplementation improved hair density and reduced the percentage of telogen hairs over 6 months.
Omega-3s also support cell membrane fluidity, which may improve the efficacy of topical peptide penetration through the stratum corneum. A healthy lipid matrix in the skin barrier facilitates better interaction with topical formulations.
Sleep and Circadian Rhythms
Growth hormone is released primarily during deep (slow-wave) sleep, and GH/IGF-1 signaling supports hair growth. Chronic sleep deprivation reduces GH release, increases cortisol (which promotes catagen), and impairs cellular repair processes.
For those using GH-stimulating peptides like CJC-1295/Ipamorelin, adequate sleep amplifies the effects by allowing the stimulated GH pulse to coincide with the natural nocturnal GH secretion pattern. The peptide is typically administered 30-60 minutes before bed for this reason.
Recommended: 7-9 hours of quality sleep per night, with consistent sleep and wake times to maintain circadian rhythm alignment.
Stress Management
Chronic psychological stress promotes hair loss through multiple mechanisms:
- Elevated cortisol promotes catagen induction via DP cell signaling changes
- Stress activates the hypothalamic-pituitary-adrenal (HPA) axis, altering growth hormone, thyroid hormone, and sex hormone levels
- Substance P (released from stress-activated sensory nerves) promotes perifollicular inflammation and mast cell degranulation
- Chronic stress impairs immune function, potentially contributing to collapse of follicular immune privilege
- Stress-induced vasoconstriction reduces scalp blood flow
Peptides like BPC-157 may partially counteract stress-related inflammation and vascular effects, but they can't replace effective stress management. Evidence-based stress reduction techniques (regular exercise, meditation, cognitive behavioral strategies, adequate social support) should be considered foundational to any hair restoration protocol.
Exercise
Regular exercise benefits hair health through several mechanisms:
- Increased blood flow to the scalp during and after exercise
- Reduced cortisol levels with regular moderate exercise
- Improved insulin sensitivity (relevant because insulin resistance may contribute to androgenetic alopecia)
- Enhanced growth hormone release (particularly with resistance training and high-intensity interval training)
- Improved sleep quality
- Stress reduction
One caveat: excessive endurance exercise (overtraining) can increase cortisol, deplete nutritional reserves, and paradoxically contribute to hair loss. Balance is key. Moderate exercise (150-300 minutes per week of moderate-intensity or 75-150 minutes of vigorous-intensity activity) is recommended.
Scalp Care Practices
The health of the scalp microenvironment directly affects follicle function and topical peptide efficacy:
- Regular cleansing: A clean scalp allows better topical product absorption. Sebum buildup can occlude follicular openings and reduce peptide penetration. Wash at least every other day, or daily if using topical products.
- Gentle handling: Avoid aggressive towel-drying, tight styling, and excessive heat. Mechanical trauma to weakened follicles accelerates shedding.
- Scalp massage: 5-10 minutes of gentle scalp massage daily increases local blood flow and may improve topical product distribution. Some evidence suggests scalp massage alone can improve hair thickness over 24 weeks.
- Sun protection: UV radiation damages hair shaft proteins and increases oxidative stress in the scalp. Hat or sunscreen use is recommended for exposed scalp areas.
- Avoid scalp irritants: Harsh sulfates, excessive alcohol in styling products, and chemical treatments (relaxers, perms) can damage follicles and create a hostile microenvironment.
Building a Complete Hair Health Protocol
The most effective approach to hair restoration integrates peptides with foundational health optimization. Here's a framework that puts it all together:
| Category | Action Items | Priority |
|---|---|---|
| Lab work | Check ferritin, vitamin D, zinc, thyroid (TSH, free T4), CBC, metabolic panel | First step |
| Nutritional foundation | Adequate protein (1.0-1.2 g/kg/day), correct any deficiencies found on labs, omega-3 supplementation | Essential |
| Lifestyle | 7-9 hours sleep, stress management, regular moderate exercise, gentle scalp care | Essential |
| Topical peptides | GHK-Cu daily, with periodic microneedling | Core protocol |
| Conventional treatments | Minoxidil 5% daily; finasteride 1mg (men) if appropriate | Evidence-based foundation |
| Injectable peptides | TB-500, BPC-157 per protocol | Advanced add-on |
| Systemic support | CJC-1295/Ipamorelin, NAD+ | Optional optimization |
| Monitoring | Monthly photos, trichoscopy every 3-6 months, lab recheck at 6 months | Ongoing |
By building on a solid nutritional and lifestyle foundation, you create the optimal conditions for peptide-based interventions to work. Skipping the foundations and jumping straight to peptides is like trying to grow a garden without first preparing the soil. The seeds (peptides) need the right environment (nutrition, sleep, stress management) to germinate and flourish.
Peptide Stacking Strategies for Hair Growth
Stacking refers to the practice of combining multiple peptides in a coordinated protocol to target different mechanisms simultaneously. Because hair loss is driven by multiple overlapping biological processes, multi-peptide approaches have a strong theoretical rationale and are increasingly popular in research and clinical settings.
The Case for Multi-Peptide Stacking
No single peptide addresses all the biological processes involved in hair loss. Each compound targets a specific set of pathways:
- GHK-Cu excels at dermal papilla support, Wnt activation, angiogenesis, and ECM remodeling
- TB-500 uniquely activates hair follicle stem cells and promotes their migration
- BPC-157 provides anti-inflammatory support and vascular repair
- Growth hormone peptides like CJC-1295/Ipamorelin optimize systemic IGF-1 for overall tissue health
When you combine these, you're addressing the dermal papilla signaling deficit, the stem cell activation problem, the vascular insufficiency, the chronic micro-inflammation, and the ECM degradation all at once. This is analogous to combination therapy in other medical fields, where multi-target approaches consistently outperform single-agent treatment.
Evidence-Based Stacking Frameworks
Stack 1: Topical Foundation (Entry Level)
This stack is the simplest to implement and the most accessible for those new to peptide-based hair growth protocols.
| Component | Role | Route | Frequency |
|---|---|---|---|
| GHK-Cu Topical | DP signaling, Wnt, VEGF, ECM | Topical (scalp) | Daily (PM) |
| Minoxidil 5% | Vasodilation, direct follicular stimulation | Topical (scalp) | Daily (AM) |
Rationale: GHK-Cu and minoxidil work through complementary mechanisms. The preclinical data showing greater effects with the combination than either alone supports this pairing. Separating application times (morning and evening) avoids potential formulation interactions and ensures consistent signaling throughout the day.
Stack 2: Topical + Injectable (Intermediate)
| Component | Role | Route | Frequency |
|---|---|---|---|
| GHK-Cu Topical | DP signaling, Wnt, VEGF, ECM | Topical (scalp) | Daily |
| TB-500 | Stem cell activation, migration | Subcutaneous | 2x/week (load), then 1x/week |
| Minoxidil 5% | Vasodilation | Topical (scalp) | Daily |
| Microneedling + GHK-Cu | Enhanced delivery, wound healing | Topical post-microneedling | Monthly |
Rationale: Adds stem cell activation (TB-500) and enhanced delivery (microneedling) to the topical foundation. This addresses both the signaling environment (GHK-Cu, minoxidil) and the cellular response (TB-500 activating the stem cells that respond to those signals).
Stack 3: Comprehensive Protocol (Advanced)
| Component | Role | Route | Frequency |
|---|---|---|---|
| GHK-Cu Topical | DP signaling, Wnt, VEGF, ECM | Topical (scalp) | Daily |
| TB-500 | Stem cell activation | Subcutaneous | 2x/week (load), then 1x/week |
| BPC-157 | Anti-inflammatory, vascular repair | Subcutaneous | Daily or every other day |
| CJC-1295/Ipamorelin | GH/IGF-1 optimization | Subcutaneous (before bed) | 5 nights/week |
| Minoxidil 5% | Vasodilation | Topical (scalp) | Daily |
| Finasteride or dutasteride | DHT reduction | Oral | Daily (Rx required) |
| Microneedling | Enhanced delivery | Topical post-microneedling | Every 3-4 weeks |
| NAD+ | Cellular energy, stem cell health | Per product guidelines | Daily |
Rationale: This comprehensive stack addresses every major pathway involved in hair loss: hormonal (finasteride), vascular (minoxidil + GHK-Cu VEGF), signaling (GHK-Cu Wnt activation), stem cell (TB-500), inflammatory (BPC-157), systemic growth factors (CJC-1295/Ipamorelin), and cellular energy (NAD+). It represents the maximum evidence-supported intervention currently available.
Protocol Note
Multi-peptide stacking protocols should be implemented gradually, starting with one or two components and adding others over weeks to months. This approach allows monitoring for adverse reactions to individual components and makes it easier to identify which elements are contributing to results. Starting everything simultaneously makes troubleshooting more difficult if issues arise.
Timing and Scheduling Considerations
When running a multi-peptide stack, timing matters for both efficacy and practicality:
- Morning: Topical minoxidil (if using)
- Evening: Topical GHK-Cu (after shower/hair wash). Separate from minoxidil by several hours.
- TB-500 injections: Morning or evening, consistent timing. Some practitioners prefer morning for alignment with natural cortisol rhythms.
- BPC-157: Can be taken at the same time as TB-500 or at a separate time. Splitting into two daily doses may provide more consistent tissue levels.
- CJC-1295/Ipamorelin: Before bed (takes advantage of the natural nocturnal GH pulse)
- Microneedling days: Apply GHK-Cu immediately post-needling. Skip minoxidil for 24 hours (it can cause excessive stinging on freshly needled skin). Resume normal topical routine the next day.
Cycling and Breaks
Some practitioners recommend cycling injectable peptides (periods on, periods off) to maintain receptor sensitivity and reduce the potential for tachyphylaxis (diminishing response with continued use). Common cycling patterns include:
- TB-500: 8 weeks on, 4 weeks off
- BPC-157: 4-8 weeks on, 2-4 weeks off (or continuous at lower doses)
- CJC-1295/Ipamorelin: 12 weeks on, 4 weeks off (to prevent GH receptor desensitization)
- Topical GHK-Cu: Continuous use (no cycling needed for topical peptides)
The evidence for cycling protocols is largely empirical rather than derived from controlled studies. Some researchers maintain continuous dosing without apparent loss of efficacy. Individual response monitoring remains the best guide.
Safety Profiles of Hair Growth Peptides
Safety is a primary concern when evaluating any therapeutic intervention, and peptides are no exception. While the peptides discussed in this report generally show favorable safety profiles in the available research, the depth of safety data varies considerably between compounds, and several important considerations warrant attention.
GHK-Cu Safety Profile
GHK-Cu has the most reassuring safety profile among the hair growth peptides, partly because it's a naturally occurring molecule found in human blood, partly because it has been used in skincare products for decades, and partly because its topical application limits systemic exposure.
Topical Safety
- Skin irritation: Mild and transient tingling or warmth at the application site is the most commonly reported side effect. Incidence is low (estimated <5% of users), and symptoms typically resolve with continued use as the skin adapts.
- Allergic reactions: Rare. Patch testing is recommended for individuals with known sensitivities to copper or peptide-containing products.
- Systemic absorption: Minimal with topical application. The amount of GHK-Cu that reaches systemic circulation through topical scalp application is negligible compared to endogenous levels already circulating in the bloodstream.
- Copper toxicity: Not a concern at typical topical concentrations. The copper content in a daily application of GHK-Cu serum is far below levels that would raise systemic copper to problematic ranges. Individuals with Wilson's disease (a genetic copper metabolism disorder) should avoid copper-containing topicals as a precaution.
Long-term Safety
GHK-Cu-containing skincare products have been on the market for over 20 years without significant safety signals. Post-market surveillance data from cosmetic products provides some reassurance regarding long-term topical use, though formal long-term clinical trials specifically for hair applications are lacking.
TB-500 Safety Profile
Thymosin Beta-4 and its synthetic fragment TB-500 have been extensively studied in wound healing applications, providing a reasonable safety database.
Injectable Safety
- Injection site reactions: Mild pain, redness, or swelling at the injection site. These are typical of subcutaneous peptide injections and generally resolve within hours.
- Flu-like symptoms: Some users report mild flu-like symptoms (fatigue, mild headache) during the initial loading phase. These typically resolve within the first 1-2 weeks.
- No serious adverse events: Published studies on Thymosin Beta-4 in wound healing have not reported serious adverse events attributable to the peptide.
Theoretical Concerns
Thymosin Beta-4 promotes cell migration and proliferation, which raises theoretical questions about cancer risk. However:
- Tbeta4 is naturally present in high concentrations in all nucleated cells
- It promotes normal wound healing, not uncontrolled proliferation
- Studies in animals have not shown increased tumor formation with Tbeta4 treatment
- Some evidence suggests Tbeta4 may actually have anti-tumor properties through immune modulation
That said, individuals with active malignancies or a high cancer risk should exercise caution and consult with their healthcare provider before using any growth-promoting peptide.
BPC-157 Safety Profile
BPC-157 has been studied in a large number of animal studies with a remarkably clean safety profile. No lethal dose (LD50) has been established in animal models, and no serious adverse events have been reported in the published literature. The most common side effects reported anecdotally are mild gastrointestinal discomfort and injection site reactions.
PTD-DBM Safety Considerations
PTD-DBM is the least characterized of the peptides discussed here from a safety standpoint. Key considerations include:
- Limited safety data: Only preclinical (animal) safety data is available. No human safety studies have been published.
- CXXC5 inhibition concerns: CXXC5 functions as a negative regulator of Wnt signaling and has tumor suppressor activity in some contexts. Chronic inhibition could theoretically increase cancer risk, though this hasn't been observed in CXXC5 knockout mice.
- Topical application limits risk: Localized topical delivery reduces systemic exposure, mitigating concerns about Wnt activation in non-target tissues.
General Safety Recommendations
Important Safety Guidelines
- Consult a qualified healthcare provider before starting any peptide protocol
- Start with one peptide at a time to identify any adverse reactions before adding others
- Use products from reputable sources with third-party testing for purity and potency
- Follow proper reconstitution, storage, and sterile injection techniques for injectable peptides
- Monitor for unexpected effects and discontinue use if concerning symptoms develop
- Inform your healthcare provider about all peptides you're using, especially if undergoing surgery or other medical treatments
- Pregnant or breastfeeding women should avoid all peptide therapies (insufficient safety data)
- Individuals with active malignancies should avoid growth-promoting peptides unless cleared by their oncologist
Drug Interactions
Potential interactions between hair growth peptides and common medications include:
| Peptide | Potential Interaction | Recommendation |
|---|---|---|
| GHK-Cu (topical) | Tretinoin (retinoids) - increased absorption through thinned skin | Separate application by 2+ hours; monitor for irritation |
| TB-500 | Anticoagulants - theoretical additive effect on blood flow | Monitor; consult prescriber |
| BPC-157 | NSAIDs - may counteract BPC-157's gastroprotective effects if taken orally | Consider timing separation |
| CJC-1295/Ipamorelin | Insulin, diabetes medications - GH affects glucose metabolism | Monitor blood glucose; dose adjustment may be needed |
| All growth peptides | Immunosuppressants - growth factors may interact with immune modulation | Consult specialist before combining |
Quality and Sourcing Concerns
One of the most significant real-world safety concerns with peptides relates to product quality. Unlike FDA-approved drugs, research peptides are not subject to pharmaceutical-grade manufacturing standards in all cases. Potential issues include:
- Purity: Impurities from synthesis (truncated sequences, by-products, residual solvents) can cause adverse reactions unrelated to the peptide itself
- Potency: Under-dosed products don't deliver therapeutic levels; over-dosed products increase side effect risk
- Sterility: Injectable products must be sterile. Contaminated products can cause serious infections.
- Stability: Improperly stored peptides can degrade, producing inactive or potentially harmful breakdown products
Sourcing from manufacturers that provide certificates of analysis (COA) with third-party verification of purity (typically >98% by HPLC), endotoxin testing, and sterility testing for injectables is essential. FormBlends products undergo rigorous testing to ensure research-grade purity and consistency.
Frequently Asked Questions
What peptides help with hair loss?
The most researched peptides for hair loss include GHK-Cu (copper peptide), which stimulates dermal papilla cells, promotes VEGF-driven blood vessel formation, and activates the Wnt/beta-catenin signaling pathway. Thymosin Beta-4 (TB-500) activates hair follicle stem cells and promotes their migration to the follicle base. PTD-DBM removes a molecular brake (CXXC5) on the Wnt pathway that is overexpressed in balding scalps. Supporting peptides like BPC-157 address inflammation, while growth hormone peptides like CJC-1295/Ipamorelin optimize systemic growth factor levels. Each targets different mechanisms, which is why combination approaches are gaining interest in both research and clinical settings.
Does GHK-Cu actually regrow hair?
GHK-Cu has demonstrated hair growth effects in both preclinical studies and small clinical trials. In vitro, it stimulates dermal papilla cell proliferation, promotes VEGF and HGF secretion, and activates the Wnt/beta-catenin pathway. In animal models, GHK-Cu-treated follicles entered growth phases faster than minoxidil-treated ones (6 days vs. 9 days). Clinical studies have reported increases in hair count and reductions in shedding. However, large-scale randomized controlled trials are still lacking, so the evidence, while promising, isn't as strong as what exists for FDA-approved treatments like minoxidil and finasteride. GHK-Cu likely works best as part of a multi-component approach rather than as a standalone treatment.
Can peptides reverse hair loss completely?
Complete reversal depends on the extent of hair loss and how long it's been progressing. Peptides are most effective when follicles are still miniaturized but retain their stem cell populations, which is the case in most androgenetic alopecia until very advanced stages. Once a follicle has completely scarred over and lost its stem cells, no topical treatment can restore it. For mild to moderate thinning, peptide-based approaches (particularly GHK-Cu combined with minoxidil and microneedling) have shown meaningful improvements in density and coverage. For advanced baldness, peptides may slow progression and improve the quality of remaining hair but are unlikely to produce full restoration. Early intervention yields the best outcomes.
Which peptide is best for hair growth?
GHK-Cu currently has the broadest evidence base for hair growth applications, with both preclinical and clinical data supporting its efficacy. It's also the most accessible, available in topical formulations that can be self-applied. Thymosin Beta-4 has compelling preclinical data, particularly for stem cell activation, but lacks human clinical trials for hair. PTD-DBM has the most targeted mechanism (Wnt pathway de-repression) but is the earliest in development. For practical use today, GHK-Cu is the strongest individual choice. For maximum effect, combining GHK-Cu (topical) with TB-500 (injectable) addresses complementary pathways and may produce better results than either alone.
How long does it take for copper peptides to work on hair?
Hair follicle cycling operates on a timeline of months, not days, so patience is essential. Most users can expect to see reduced shedding within 4 to 8 weeks as follicles begin responding to improved signaling. Visible improvements in hair density and thickness typically become apparent at 3 to 6 months. Maximum benefit usually occurs at 9 to 12 months of consistent use. Results are faster when copper peptides are combined with microneedling (which enhances penetration up to 400%) and other active ingredients. The timeline is similar to minoxidil, which also requires 3-6 months for visible results. Consistency is critical; interrupting treatment can reset progress.
Is GHK-Cu better than minoxidil for hair growth?
The two work through different mechanisms and are better viewed as complementary rather than competing treatments. Minoxidil has far more clinical evidence (multiple large randomized controlled trials, decades of use, FDA approval), making it the more proven option. GHK-Cu addresses several pathways that minoxidil does not: Wnt/beta-catenin activation, ECM remodeling, anti-inflammatory effects, and TGF-beta suppression. Preclinical data suggests the combination of GHK-Cu and minoxidil produces greater effects than either alone. For someone choosing just one treatment, minoxidil has the stronger evidence base. For those willing to use both, the combination may offer superior results by targeting multiple mechanisms simultaneously.
Can women use hair growth peptides?
Yes, peptides like GHK-Cu and TB-500 are considered suitable for women. This is actually an advantage over finasteride, which is contraindicated in women of childbearing potential due to teratogenic risk. GHK-Cu topical application has no reported gender-specific side effects, and Thymosin Beta-4 similarly shows no sex-dependent safety concerns in published research. Women with female pattern hair loss (which involves different hormonal and signaling pathways than male pattern baldness) may benefit from GHK-Cu's Wnt activation and VEGF promotion effects. However, pregnant or breastfeeding women should avoid all peptide therapies due to insufficient safety data during these periods.
How do you apply GHK-Cu to the scalp?
Apply topical GHK-Cu serum to a clean, towel-dried scalp, ideally after a shower. Part your hair in rows to expose the scalp skin directly, then apply 1-2 mL of serum (or per product directions) along the parts, focusing on thinning areas. Gently massage the product in for 1 to 2 minutes to distribute it evenly and enhance absorption. Allow it to dry before styling. For enhanced results, use a derma pen (0.5-1.0 mm needle depth) on the scalp every 2-4 weeks, applying the GHK-Cu serum immediately after microneedling while the microchannels are still open. Avoid washing your hair for at least 4 to 6 hours after application to allow maximum absorption.
What is PTD-DBM and is it available to buy?
PTD-DBM stands for Protein Transduction Domain-fused Dishevelled Binding Motif. It's a synthetic peptide designed to block CXXC5, a protein that is overexpressed in balding scalps and acts as a brake on the Wnt/beta-catenin signaling pathway. By blocking CXXC5's interaction with the Dishevelled protein, PTD-DBM restores Wnt signaling and promotes hair follicle growth. In animal studies, topical PTD-DBM promoted new hair follicle formation and prevented hair loss. However, PTD-DBM is currently in the research stage only. It's not commercially available as a consumer product, and no human clinical trials have been published. It represents a promising future direction rather than a currently available treatment.
Can you combine GHK-Cu with minoxidil?
Yes, combining GHK-Cu with minoxidil is one of the most commonly recommended approaches in the hair peptide space, and preclinical data supports the combination. The two work through different mechanisms: minoxidil primarily acts as a vasodilator and potassium channel opener, while GHK-Cu activates the Wnt pathway, promotes VEGF, reduces inflammation, and supports ECM remodeling. To avoid potential formulation interactions, many practitioners recommend applying them at different times, such as minoxidil in the morning and GHK-Cu in the evening. When used with microneedling, a combined solution can be applied post-needling, though minoxidil should be avoided immediately after microneedling if it causes stinging.
Does TB-500 help with hair growth?
TB-500, a synthetic fragment of Thymosin Beta-4, has demonstrated hair growth effects in multiple animal studies. Its mechanism is unique among hair peptides: it directly activates hair follicle stem cells in the bulge region and promotes their migration to the follicle base, where they differentiate into the matrix cells that produce new hair. The original discovery was made by NIH researchers who noticed unexpected hair regrowth in animals being treated for wound healing. Published studies in the FASEB Journal and PLoS ONE confirmed these effects. However, no human clinical trial has specifically evaluated TB-500 for hair growth. Current use for hair is based on extrapolation from animal data and anecdotal reports from users taking TB-500 for other purposes who notice improved hair quality as a side benefit.
Are hair growth peptides safe?
The peptides discussed in this report generally show favorable safety profiles based on available data. GHK-Cu is a naturally occurring molecule found in human blood and has been used in skincare for over 20 years with minimal adverse effects (primarily mild scalp tingling). Thymosin Beta-4 has been studied in wound healing applications without serious adverse events. BPC-157 has no established lethal dose in animal studies. However, safety data is more limited than for FDA-approved treatments, and long-term studies specific to hair applications are lacking. Key safety practices include sourcing from reputable manufacturers with third-party testing, starting one peptide at a time, following proper sterile technique for injectables, and consulting with a healthcare provider. Pregnant or breastfeeding women and those with active malignancies should avoid these peptides.
What role does the Wnt pathway play in hair loss?
The Wnt/beta-catenin signaling pathway is the master regulator of hair follicle cycling. When Wnt signaling is active, beta-catenin accumulates in the nucleus of dermal papilla cells and activates genes that drive follicle growth. When it's suppressed, follicles miniaturize and stop producing visible hair. In androgenetic alopecia, both DHT and prostaglandin D2 suppress Wnt signaling through upregulation of CXXC5, a negative feedback protein. This is why Wnt pathway activation is a primary target for hair growth peptides: GHK-Cu promotes beta-catenin nuclear accumulation, and PTD-DBM blocks CXXC5 to remove the brake on Wnt signaling. Restoring normal Wnt activity is considered one of the most promising approaches to treating pattern hair loss at its molecular root.
How does microneedling enhance peptide absorption for hair growth?
Microneedling creates thousands of tiny channels through the stratum corneum (the outer skin barrier that normally limits topical absorption) using fine needles typically 0.5-1.0 mm in length for scalp use. These microchannels bypass the barrier entirely, allowing peptides like GHK-Cu direct access to the viable epidermis and upper dermis where hair follicles reside. Studies show this can increase absorption by up to 400% compared to passive topical application. Additionally, the controlled micro-injury triggers a natural wound healing response that releases growth factors (PDGF, VEGF, EGF) which complement the effects of the applied peptides. Clinical data shows microneedling achieves 82% patient response rates compared to 4.5% with topical-only treatment, though this specific data was with minoxidil rather than peptides.
References & Clinical Sources
- Pickart L, Vasquez-Soltero JM, Margolina A. Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data. Int J Mol Sci. 2018;19(7):1987. doi:10.3390/ijms19071987
- Pickart L. The human tri-peptide GHK and tissue remodeling. J Biomater Sci Polym Ed. 2008;19(8):969-988. doi:10.1163/156856208784909435
- Pyo HK, Yoo HG, Won CH, et al. The effect of tripeptide-copper complex on human hair growth in vitro. Arch Pharm Res. 2007;30(7):834-839. doi:10.1007/BF02978833
- Philp D, Nguyen M, Bhatt S, et al. Thymosin beta4 increases hair growth by activation of hair follicle stem cells. FASEB J. 2004;18(2):385-387. doi:10.1096/fj.03-0244fje
- Philp D, St-Surin S, Cha HJ, Moon HS, Kleinman HK, Elkin M. Thymosin beta 4 induces hair growth via stem cell migration and differentiation. Ann N Y Acad Sci. 2007;1112:95-103. doi:10.1196/annals.1415.009
- Gao X, Liang H, Hou F, et al. Thymosin Beta-4 Induces Mouse Hair Growth. PLoS One. 2015;10(7):e0130040. doi:10.1371/journal.pone.0130040
- Kim BK, Yoon SK. Multiple potential roles of thymosin beta4 in the growth and development of hair follicles. Int J Mol Sci. 2021;22(2):946. doi:10.3390/ijms22020946
- Lee SH, Seo SH, Lee DH, Pi LQ, Lee WS, Choi KY. CXXC5 mediates DHT-induced androgenetic alopecia via PGD2. Int J Mol Sci. 2023;24(4):3399. doi:10.3390/ijms24043399
- Lee SH, Yoon J, Shin SH, et al. Valproic acid induces hair regeneration in murine model and activates alkaline phosphatase activity in human dermal papilla cells. PLoS One. 2012;7(4):e34152. doi:10.1371/journal.pone.0034152
- Choi S, Yoon M, Choi KY. Approaches for regenerative healing of cutaneous wound with an emphasis on strategies activating the Wnt/beta-catenin pathway. Adv Wound Care. 2022;11(2):70-86. doi:10.1089/wound.2020.1284
- Dhurat R, Sukesh M, Avhad G, Dandale A, Pal A, Pund P. A randomized evaluator blinded study of effect of microneedling in androgenetic alopecia: a pilot study. Int J Trichology. 2013;5(1):6-11. doi:10.4103/0974-7753.114700
- Aldag C, Teixeira DN, Leventhal PS. Skin rejuvenation using cosmetic products containing copper peptides. Arch Dermatol Res. 2016;308(3):155-163. doi:10.1007/s00403-016-1625-2
- Uno H, Kurata S. Chemical agents and peptides affect hair growth. J Invest Dermatol. 1993;101(1 Suppl):143S-147S. doi:10.1111/1523-1747.ep12363275
- Lin JY, Fisher DE. Melanocyte biology and skin pigmentation. Nature. 2007;445(7130):843-850. doi:10.1038/nature05660
- Millar SE. Molecular mechanisms regulating hair follicle development. J Invest Dermatol. 2002;118(2):216-225. doi:10.1046/j.0022-202x.2001.01670.x
- Yano K, Brown LF, Detmar M. Control of hair growth and follicle size by VEGF-mediated angiogenesis. J Clin Invest. 2001;107(4):409-417. doi:10.1172/JCI11317
- Inui S, Itami S. Molecular basis of androgenetic alopecia: From androgen to paracrine mediators through dermal papilla. J Dermatol Sci. 2011;61(1):1-6. doi:10.1016/j.jdermsci.2010.10.015
- Garza LA, Liu Y, Yang Z, et al. Prostaglandin D2 inhibits hair growth and is elevated in bald scalp of men with androgenetic alopecia. Sci Transl Med. 2012;4(126):126ra34. doi:10.1126/scitranslmed.3003122
- Ito M, Yang Z, Andl T, et al. Wnt-dependent de novo hair follicle regeneration in adult mouse skin after wounding. Nature. 2007;447(7142):316-320. doi:10.1038/nature05766
- Chen D, Jarrell A, Guo C, Lang R, Atit R. Dermal beta-catenin activity in response to epidermal Wnt ligands is required for fibroblast proliferation and hair follicle initiation. Development. 2012;139(8):1522-1533. doi:10.1242/dev.076463
- Mecklenburg L, Tobin DJ, Muller-Rover S, et al. Active hair growth (anagen) is associated with angiogenesis. J Invest Dermatol. 2000;114(5):909-916. doi:10.1046/j.1523-1747.2000.00954.x
- Goldstein AL, Hannappel E, Kleinman HK. Thymosin beta4: actin-sequestering protein moonlights to repair injured tissues. Trends Mol Med. 2005;11(9):421-429. doi:10.1016/j.molmed.2005.07.004
- Li ZJ, Choi HI, Choi DK, et al. Autologous platelet-rich plasma: a potential therapeutic tool for promoting hair growth. Dermatol Surg. 2012;38(7 Pt 1):1040-1046. doi:10.1111/j.1524-4725.2012.02394.x
- Zhang B, Tsai PC, Gonzalez-Celeiro M, et al. Hair follicles' transit-amplifying cells govern concurrent dermal adipocyte production through Sonic Hedgehog. Genes Dev. 2016;30(20):2325-2338. doi:10.1101/gad.285429.116
- Rossi A, Cantisani C, Melis L, Iorio A, Scali E, Calvieri S. Minoxidil use in dermatology, side effects and recent patents. Recent Pat Inflamm Allergy Drug Discov. 2012;6(2):130-136. doi:10.2174/187221312800166859
- Kaufman KD, Olsen EA, Whiting D, et al. Finasteride in the treatment of men with androgenetic alopecia. J Am Acad Dermatol. 1998;39(4 Pt 1):578-589. doi:10.1016/S0190-9622(98)70007-6
- Woo YR, Park CJ, Kang H, Kim JE. The hair cycle and its relationship with androgens. Int J Mol Sci. 2022;23(3):1305. doi:10.3390/ijms23031305
- Schneider MR, Schmidt-Ullrich R, Paus R. The hair follicle as a dynamic miniorgan. Curr Biol. 2009;19(3):R132-R142. doi:10.1016/j.cub.2008.12.005
- Hu HM, Zhang SB, Lei XH, et al. Estrogen leads to reversible hair cycle retardation through inducing premature catagen and maintaining telogen. PLoS One. 2012;7(7):e40124. doi:10.1371/journal.pone.0040124
- Choi KY. Biomedical Use of Copper-Containing Compounds. International Journal of Molecular Sciences. 2023;24(15):12007. doi:10.3390/ijms241512007
- Stenn KS, Paus R. Controls of hair follicle cycling. Physiol Rev. 2001;81(1):449-494. doi:10.1152/physrev.2001.81.1.449
- Botchkarev VA, Kishimoto J. Molecular control of epithelial-mesenchymal interactions during hair follicle cycling. J Investig Dermatol Symp Proc. 2003;8(1):46-55. doi:10.1046/j.1523-1747.2003.12171.x
- Paus R, Cotsarelis G. The biology of hair follicles. N Engl J Med. 1999;341(7):491-497. doi:10.1056/NEJM199908123410706
- Rompolas P, Deschene ER, Zito G, et al. Live imaging of stem cell and progeny behaviour in physiological hair-follicle regeneration. Nature. 2012;487(7408):496-499. doi:10.1038/nature11218
- Kim M, Kim BJ. Advances in Transdermal Delivery Systems for Treating Androgenetic Alopecia. Pharmaceutics. 2025;17(8):984. doi:10.3390/pharmaceutics17080984
- Diao F, Jiang Z, Zhang Y, et al. Thermodynamically stable ionic liquid microemulsions pioneer pathways for topical delivery and peptide application. Pharmaceutics. 2023;15(11):2616. doi:10.3390/pharmaceutics15112616
- Zhou J, Zheng Q, Chen Z. Co-delivery of bioactive peptides by nanoliposomes for promotion of hair growth. Colloids Surf B Biointerfaces. 2022;218:112746. doi:10.1016/j.colsurfb.2022.112746
- Ohn J, Kim KH, Kwon O. Evaluating hair growth promoting effects of candidate substance: a review of research methods. J Dermatol Sci. 2019;93(3):144-149. doi:10.1016/j.jdermsci.2019.02.004
- Bernstein RM. Microneedling treatment of hair loss. In: Hair and Scalp Treatments. Springer; 2020:411-422. doi:10.1007/978-3-030-21555-2_36
- Jeon YJ, Song SH, Kim SJ, et al. Enhanced hair regrowth with five monthly sessions of minoxidil-dutasteride-copper peptides tattooing for androgenetic alopecia assessed by artificial intelligence and blinded evaluators. J Cosmet Dermatol. 2025;24(4):e16321. doi:10.1111/jocd.16321
Figure 7: Timeline of key milestones in hair growth peptide research, from Pickart's 1973 discovery of GHK-Cu through the 2025 copper peptide microneedling studies.
Figure 8: The Wnt/beta-catenin pathway in hair follicles showing intervention points for GHK-Cu (promotes beta-catenin accumulation) and PTD-DBM (blocks CXXC5-Dvl interaction to de-repress the pathway).
Figure 9: Peptide stacking protocol overview showing three tiers of intervention, from topical-only foundation stacks to comprehensive multi-peptide protocols targeting all major hair loss pathways.
Figure 10: Comparison of peptide delivery methods for scalp application, showing relative bioavailability, ease of use, and optimal peptide-delivery method pairings.