Executive Summary
Khavinson peptide bioregulators represent one of the most extensive and longest-running programs of peptide-based anti-aging research in history. Spanning over four decades, the work of Professor Vladimir Khavinson at the Saint Petersburg Institute of Bioregulation and Gerontology has produced six pharmaceutical-grade peptide preparations, 64 peptide-based supplements, 196 patents, and more than 775 scientific publications.
Key Takeaways
- Khavinson peptide bioregulators are ultra-short peptides (2-7 amino acids) that regulate gene expression in specific organ systems
- Over 40 years of research at the St. Petersburg Institute of Bioregulation and Gerontology supports their geroprotective effects
- Clinical studies show mortality reduction up to 4.1-fold with combined thymic and pineal peptide treatment
- Animal studies demonstrate lifespan extension of 20-40% across multiple species
- Six pharmaceutical preparations and 64 supplements have been developed from this research
The core premise is elegant in its simplicity: short peptides consisting of just 2 to 7 amino acids can interact directly with specific DNA sequences to regulate gene expression in a tissue-specific manner. Rather than flooding the body with broad-acting compounds, each bioregulator peptide targets a particular organ system. Epithalon works on the pineal gland. Thymalin restores thymic function. Pinealon supports the brain and central nervous system. Cortagen, Vesugen, Cartalax, and Livagen each address their own tissue targets with remarkable specificity.
What separates the Khavinson approach from most peptide research is the sheer volume of long-term clinical data. In one landmark study, 266 elderly patients received thymic and pineal peptide bioregulators for 2 to 3 years with follow-up extending to 14 to 20 years. The combined treatment group saw mortality rates drop by a factor of 4.1 compared to controls. Animal studies have consistently demonstrated lifespan extensions of 20% to 40% with these same peptide preparations.
The safety profile across these decades of research has been remarkably clean. Acute toxicity tests at doses 5,000 times above therapeutic levels produced no serious reactions. Long-term administration at 100 to 1,000 times normal doses revealed no adverse effects in animal models. No significant allergic, toxic, or adverse effects have been documented across the clinical history of these compounds.
Key Takeaways
- Khavinson peptide bioregulators are ultra-short peptides (2-7 amino acids) that regulate gene expression in specific organ systems
- Over 40 years of research at the St. Petersburg Institute of Bioregulation and Gerontology supports their geroprotective effects
- Clinical studies show mortality reduction up to 4.1-fold with combined thymic and pineal peptide treatment
- Animal studies demonstrate lifespan extension of 20-40% across multiple species
- Six pharmaceutical preparations and 64 supplements have been developed from this research
- The compounds have an exceptionally clean safety profile across decades of clinical use
This guide covers every major Khavinson bioregulator peptide in detail. You'll learn the underlying theory, the specific peptides for each organ system, the clinical evidence, practical dosing protocols, and safety considerations. Whether you're a clinician evaluating these compounds for patient care, a researcher exploring the bioregulation model, or an individual interested in evidence-based longevity strategies, this resource pulls together the full scope of what's known about this fascinating class of peptides.
For related research on individual peptides in this family, see our dedicated reports on Epithalon and telomere biology and Pinealon for sleep and neuroprotection. You can also explore our broader Peptide Research Hub for context on how bioregulators fit within the larger peptide landscape.
Khavinson's Theory of Peptide Bioregulation
Origins: From Soviet Military Research to Gerontology
The story of peptide bioregulators begins not in a university laboratory but in the Cold War-era Soviet military. During the 1970s, Vladimir Khavinson was tasked with developing methods to protect soldiers, cosmonauts, and athletes from extreme physiological stressors including radiation exposure, laser-induced retinal damage, and immune suppression from prolonged combat conditions. The solution his team pursued was to extract peptide complexes from the organs of young, healthy animals and administer them to stressed or aging organisms.
The results were startling enough to redirect Khavinson's entire career. Animals treated with thymic extracts showed restored immune function. Those given pineal extracts demonstrated normalized circadian rhythms and hormonal output. The extracts didn't simply provide temporary relief. They appeared to reprogram cellular behavior at a fundamental level, with effects persisting long after the treatment period ended.
From this military foundation, Khavinson established the Saint Petersburg Institute of Bioregulation and Gerontology, where he spent the next four decades systematically isolating, characterizing, and synthesizing the active peptide sequences from these tissue extracts. The work eventually yielded a comprehensive theory of how short peptides regulate aging at the epigenetic level.
Figure 1: Khavinson's bioregulation theory - short peptides interact with DNA to regulate organ-specific gene expression
The Bioregulation Model: How Short Peptides Talk to DNA
At its core, Khavinson's bioregulation theory proposes a straightforward mechanism. Short peptides, due to their small molecular weight, can penetrate both the cell membrane and the nuclear membrane. Once inside the nucleus, they interact directly with specific DNA sequences in gene promoter regions. This interaction triggers strand separation and initiates gene transcription, effectively switching genes on or off in a tissue-specific pattern.
The specificity comes from the amino acid sequence itself. Each tissue type contains characteristic peptide sequences that serve as complementary "keys" to specific DNA regions. When you administer a peptide derived from thymic tissue, it preferentially interacts with genes involved in thymic function. A pineal-derived peptide targets pineal-related genes. This is not a general stimulation of all genes but rather a precisely targeted restoration of the expression patterns associated with younger, healthier tissue.
The proposed mechanism operates at multiple levels of epigenetic regulation:
- Chromatin remodeling: Short peptides can alter histone-DNA interactions, changing chromatin structure from a condensed (silenced) state to an open (transcriptionally active) state
- DNA methylation interference: Peptides binding to gene promoter regions may block access by DNA methyltransferases, keeping promoters unmethylated and genes active
- Transcription factor interaction: Short peptides can interact with cis-regulatory elements and transcription factors to modulate gene expression rates
- RNA-level regulation: Some peptide bioregulators appear to influence mRNA stability and translation efficiency
The "Cascade Effect" and Why Short Courses Work
One of the most distinctive features of Khavinson's approach is the dosing philosophy. Unlike most Western pharmaceutical models where daily dosing maintains therapeutic drug levels, bioregulators are administered in short courses, typically 5 to 10 days for injectable forms or 30 days for oral preparations, followed by extended breaks of months before repeating.
This works because the peptides don't function as traditional drugs that need constant presence in the bloodstream. Instead, they initiate an epigenetic cascade. A brief exposure is sufficient to alter gene expression patterns, and those altered patterns persist well beyond the administration period. Think of it as resetting a thermostat rather than manually heating a room. Once the new setting is established, the system maintains itself.
Research from Khavinson's group has demonstrated that a single course of bioregulator treatment can produce measurable changes in gene expression lasting 4 to 6 months. This explains the typical protocol of two courses per year, which maintains the restored expression patterns without continuous administration.
Understanding the Cascade
The bioregulation cascade works in stages: (1) Peptide enters the cell nucleus and binds to specific DNA sequences. (2) Gene expression patterns shift toward a younger phenotype. (3) Protein synthesis normalizes for the target tissue. (4) Cellular function improves. (5) Organ function restores. Each stage reinforces the next, creating a self-sustaining improvement that persists after the peptide itself has been metabolized.
Tissue Specificity: The Organ-Targeting Principle
Perhaps the most compelling aspect of Khavinson's work is the demonstration of tissue specificity. In controlled experiments, peptides isolated from a particular organ consistently and selectively affected gene expression in that same organ type, with minimal or no effect on other tissues.
For example, the tetrapeptide Epithalon (Ala-Glu-Asp-Gly), derived from pineal tissue, activates telomerase and increases melatonin production specifically in pineal cells. It doesn't stimulate telomerase activity uniformly across all cell types. Similarly, Thymalin's polypeptide complex preferentially restores T-cell differentiation and thymic function without broadly stimulating the entire immune system in a non-specific way.
This tissue specificity has been confirmed through multiple experimental approaches. Fluorescently labeled short peptides, when administered systemically, show preferential accumulation in their target tissues. Gene expression profiling reveals that each peptide upregulates a distinct set of genes corresponding to its tissue of origin. And clinical outcomes consistently show organ-specific improvements matching the administered peptide's target.
The practical significance of this specificity is enormous. It means a bioregulator protocol can be assembled like a toolkit, selecting the specific peptides needed for each patient's particular areas of decline. Someone with age-related immune dysfunction gets Thymalin. Someone with cognitive decline gets Cortagen or Pinealon. Someone concerned about cardiovascular aging gets Vesugen. This modular approach allows for highly personalized anti-aging interventions.
Evidence Quality: Strengths and Limitations
It's important to address the evidence base honestly. Khavinson's research is extensive, spanning thousands of patients and decades of follow-up. The consistency of results across multiple studies and multiple peptides is remarkable. And the animal data, showing 20% to 40% lifespan extension, is among the most impressive in gerontology research.
However, virtually all of this research has come from Khavinson's own institute and affiliated Russian research centers. Independent replication by Western research groups has been limited, though it has begun to appear. A 2025 study published in Biogerontology by an independent group confirmed that Epitalon increases telomere length in human cell lines through telomerase upregulation, providing important third-party validation of at least one core mechanism.
The broader scientific community has called for larger-scale, independently conducted clinical trials using modern methodology including proper randomization, blinding, and pre-registration. Until such trials are completed, the evidence should be considered promising but not definitive by conventional Western medical standards. That said, the 40+ year track record of safe clinical use in Russia provides a level of real-world evidence that shorter-term studies cannot match.
| Feature | Khavinson Bioregulators | Conventional Drug Development |
|---|---|---|
| Molecule Size | 2-7 amino acids | Variable, often large molecules |
| Target Specificity | Organ-specific via DNA interaction | Receptor-specific pharmacology |
| Dosing Model | Short courses with long breaks | Continuous daily dosing |
| Mechanism | Epigenetic gene regulation | Receptor agonism/antagonism |
| Duration of Effect | Months after single course | Only while drug is present |
| Safety Profile | Extremely favorable across decades | Variable, often significant side effects |
| Regulatory Status | Approved in Russia; research-only elsewhere | Full FDA/EMA approval pathway |
Epithalon (Pineal)
Discovery and Composition
Epithalon (also spelled Epitalon) is arguably the most famous and most studied of all Khavinson peptide bioregulators. This synthetic tetrapeptide with the sequence Ala-Glu-Asp-Gly was identified as the active component of Epithalamin, a polypeptide complex originally extracted from bovine pineal glands. Khavinson's team isolated the tetrapeptide sequence and demonstrated that it retained the biological activity of the full extract, making large-scale synthesis feasible.
The pineal gland plays a central role in aging biology. As the body's master circadian regulator and primary source of melatonin, its decline with age contributes to disrupted sleep, hormonal dysregulation, immune suppression, and accelerated cellular aging. Epithalon was developed specifically to restore youthful pineal function and, through that restoration, to slow the overall aging process.
Telomerase Activation and Telomere Extension
Epithalon's most celebrated mechanism is its ability to activate telomerase, the enzyme responsible for maintaining telomere length. Telomeres, the protective caps on chromosome ends, shorten with each cell division. When they become critically short, cells enter senescence or die. This telomere shortening is one of the primary molecular drivers of aging.
In human clinical studies, Epithalon and its parent compound Epithalamin both significantly increased telomere lengths in blood cells of patients aged 60 to 65 and 75 to 80. This finding was published in Neuroendocrinology Letters and represented one of the first demonstrations that a peptide could reverse telomere shortening in humans (Khavinson et al., 2003).
A 2025 study published in Biogerontology provided independent confirmation, showing that Epitalon increases telomere length in human cell lines through both telomerase upregulation and alternative lengthening of telomeres (ALT) activity. This was particularly significant because it came from researchers outside Khavinson's immediate circle, providing third-party validation of the telomere-extending mechanism.
Clinical Evidence Highlight
In a study of patients aged 60-80, Epithalon treatment produced measurable increases in telomere length in peripheral blood cells. The effect was observed across both age groups (60-65 and 75-80), suggesting the telomerase activation mechanism remains responsive even in advanced age. These findings have been partially replicated by independent researchers, strengthening confidence in the telomere-extension claim.
Melatonin Restoration
Beyond telomerase, Epithalon has been shown to restore melatonin secretion in both aged primates and elderly humans. In studies with old monkeys, evening melatonin production, which normally declines dramatically with age, was restored to levels approaching those of young animals after Epithalon treatment. Similar results were observed in elderly human patients, where the normal age-related decline in nocturnal melatonin was reversed.
This melatonin restoration has cascading benefits. Melatonin is not just a sleep hormone. It's a potent antioxidant, an immune modulator, and a regulator of other hormonal systems. By restoring melatonin production, Epithalon indirectly supports antioxidant defense, immune function, and hormonal balance.
Anticancer Properties
Animal studies have demonstrated significant anticancer effects of Epithalon. In rodent models, treatment with the peptide reduced the incidence of spontaneous tumors and inhibited the growth of transplanted tumors. The proposed mechanisms include enhanced immune surveillance (via restored thymic and pineal function), increased antioxidant defense, and improved DNA repair capacity.
While these findings are promising, it's critical to note that they come primarily from animal studies. Human anticancer effects have not been conclusively demonstrated in controlled clinical trials, though the reduced incidence of cancer-related mortality in the long-term observational studies is suggestive.
Lifespan and Longevity Data
Perhaps the most dramatic data for Epithalon comes from animal lifespan studies. Across multiple experiments with different species:
- Drosophila (fruit flies) treated with Epithalon showed lifespan extensions of 11% to 16%
- Mice treated with Epithalon or Epithalamin lived 25% to 30% longer on average
- Rats showed similar lifespan extensions with both the synthetic peptide and the natural extract
In the human observational studies, the Epithalamin-treated group showed a 1.6 to 1.8-fold decrease in mortality compared to controls. When combined with Thymalin, this increased to a 2.5-fold reduction, and with annual repeat courses over 6 years, the combined treatment achieved a 4.1-fold mortality reduction.
For a deep exploration of Epithalon's telomere biology, mechanisms, and dosing, see our complete Epithalon Telomere Peptide Report. You can also find Epithalon in our product catalog.
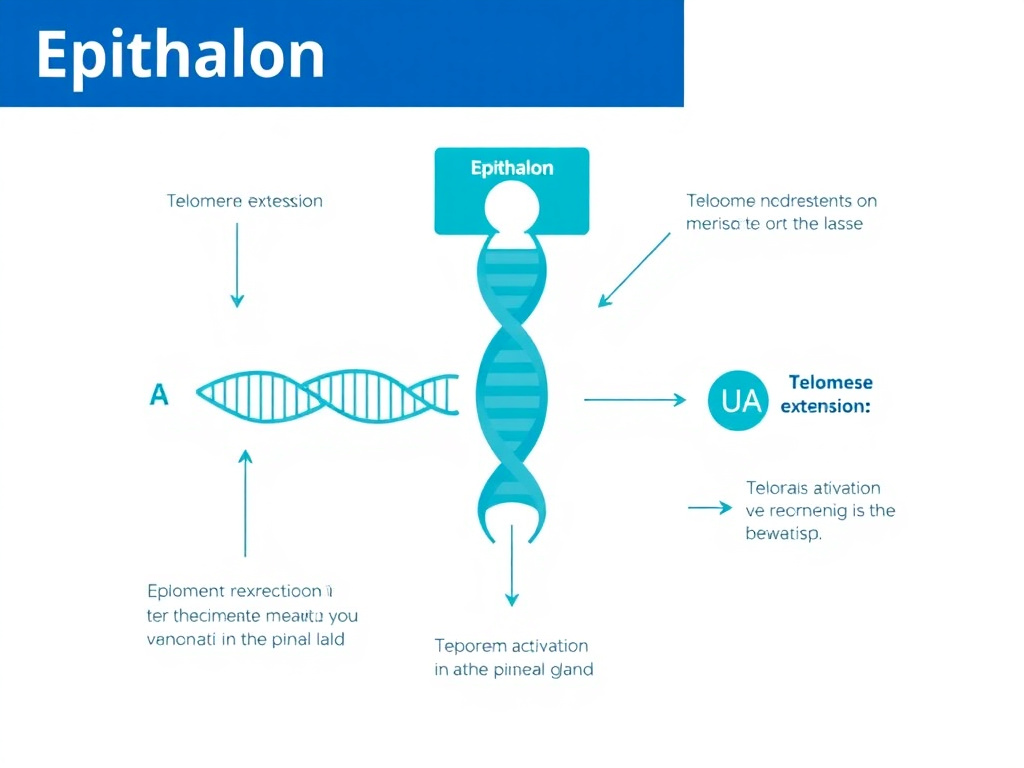
Figure 2: Epithalon activates telomerase in the pineal gland, leading to telomere extension and restored melatonin production
Thymalin (Thymus)
The Thymus Problem in Aging
The thymus gland is arguably the most age-sensitive organ in the human body. Starting around puberty, the thymus begins to involute, gradually being replaced by fatty tissue. By age 50, most people have lost the majority of their functional thymic tissue. By 70, the thymus is largely vestigial. This involution directly causes the decline in immune function that characterizes aging, a process called immunosenescence.
Without a functional thymus, the body produces fewer naive T-cells, reducing its ability to mount immune responses to new pathogens and increasing susceptibility to infections, autoimmune conditions, and cancer. Reversing or slowing thymic involution has been one of the holy grails of anti-aging research.
Thymalin: Composition and History
Thymalin is a polypeptide complex extracted from calf thymus tissue. Unlike the synthetic di- and tetrapeptides in the Khavinson family, Thymalin retains a complex mixture of peptide sequences that collectively stimulate thymic function. It was one of the first Khavinson preparations to receive pharmaceutical approval in Russia and has been used clinically since the 1980s.
The compound works by promoting T-cell maturation and differentiation, restoring the balance between T-helper and T-suppressor cells, enhancing natural killer (NK) cell activity, and normalizing cytokine production. In essence, it reverses many of the immune changes associated with aging.
For those interested in related thymic peptides, Thymosin Alpha-1 offers another well-studied approach to immune restoration with extensive clinical validation.
The Landmark Longevity Study
The most significant clinical data for Thymalin comes from a series of studies conducted at the St. Petersburg Institute between 1985 and 2003. In the primary trial, 266 elderly patients (ages 60 to 89) were divided into groups receiving Thymalin alone, Epithalamin alone, both peptides together, or neither (control).
Results after 6 to 8 years of observation showed:
| Treatment Group | Mortality Reduction vs. Control | Key Improvements |
|---|---|---|
| Thymalin alone | 2.0 to 2.1-fold decrease | Immune function, respiratory infections reduced 2.0-2.4x |
| Epithalamin alone | 1.6 to 1.8-fold decrease | Endocrine markers, circadian rhythm normalization |
| Combined (Thymalin + Epithalamin) | 2.5-fold decrease | Cardiovascular, endocrine, immune, and nervous system improvements |
| Combined annual courses (6 years) | 4.1-fold decrease | Maximum benefit with sustained annual treatment |
The treated groups showed improvements across multiple organ systems. The incidence of acute respiratory diseases dropped 2.0 to 2.4-fold. Rates of ischemic heart disease, hypertension, deforming osteoarthrosis, and osteoporosis all declined significantly compared to controls. The indices of cardiovascular, endocrine, immune, and nervous system function were restored to levels typical of middle-aged adults.
Clinical Evidence Highlight
The 4.1-fold mortality reduction observed with combined annual Thymalin and Epithalamin treatment over 6 years represents one of the largest mortality effects ever reported for any anti-aging intervention. While these results come from a single research group and need independent replication, the long follow-up period (14-20 years) and the consistency of the dose-response relationship (single treatment less effective than annual courses) increase confidence in the findings.
Immunological Mechanisms
Detailed immunological analysis of Thymalin-treated patients revealed multiple mechanisms of action:
- T-cell restoration: CD4+ helper T-cell counts increased, while the CD4/CD8 ratio normalized toward youthful values
- Cytokine balance: Pro-inflammatory cytokines (IL-6, TNF-alpha) decreased while anti-inflammatory and immunoregulatory cytokines normalized
- NK cell activity: Natural killer cell cytotoxicity against tumor cells increased by 30% to 50%
- Phagocyte function: Neutrophil and macrophage phagocytic activity improved
- Antibody production: Vaccine responses improved, suggesting better adaptive immune function
These changes collectively represent a reversal of immunosenescence, the age-related decline in immune function that leaves elderly individuals vulnerable to infections, autoimmune disease, and cancer.
Animal Lifespan Data
In animal studies, Thymalin treatment produced consistent lifespan extensions:
- Mice: 25% to 30% increase in mean lifespan
- Rats: 20% to 25% increase in mean lifespan
- Drosophila: 11% to 16% increase in mean lifespan
These effects were dose-dependent and reproducible across multiple experiments. The combination of Thymalin with Epithalamin produced additive lifespan benefits exceeding either peptide alone, consistent with the human mortality data.
Cortexin (Brain)
Brain Aging and the Need for Neuroprotection
The aging brain faces multiple concurrent threats: reduced blood flow, accumulating oxidative damage, declining neuroplasticity, chronic low-grade inflammation, and the gradual loss of neurons and synaptic connections. These processes collectively drive the cognitive decline that most people experience with age and, in their more severe forms, contribute to neurodegenerative conditions like Alzheimer's and Parkinson's disease.
Cortexin is a polypeptide complex derived from the cerebral cortex of young cattle. It's one of the six pharmaceutical-grade Khavinson preparations approved for clinical use in Russia and has been used primarily in neurological practice since its approval.
Composition and Mechanism
As a complex extract rather than a single synthetic peptide, Cortexin contains a mixture of neuropeptides along with amino acids, vitamins, and trace minerals. Its mechanisms of action include:
- Neuroprotection: Cortexin reduces neuronal damage from oxidative stress, excitotoxicity, and ischemia
- Neuroplasticity enhancement: The peptide complex promotes synaptogenesis and enhances brain-derived neurotrophic factor (BDNF) expression
- Anti-inflammatory effects: Cortexin suppresses neuroinflammation by modulating microglial activation and reducing pro-inflammatory cytokine production in the CNS
- GABAergic modulation: The compound appears to influence the balance between excitatory and inhibitory neurotransmission, favoring a more balanced state
Published Clinical Studies by Peptide Bioregulator
Clinical Applications
In Russian clinical practice, Cortexin is used for a range of neurological conditions:
| Condition | Clinical Evidence | Reported Outcomes |
|---|---|---|
| Ischemic stroke recovery | Multiple controlled studies | Improved cognitive recovery, reduced neurological deficit scores |
| Traumatic brain injury | Controlled clinical trials | Faster cognitive recovery, improved memory and attention |
| Epilepsy (adjunctive) | Open-label studies | Reduced seizure frequency when added to standard therapy |
| Cognitive decline in elderly | Observational studies | Improved cognitive test scores, better daily functioning |
| Pediatric neurodevelopmental | Clinical case series | Improved cognitive and motor development in delayed children |
A study involving 72 patients with traumatic brain injury demonstrated improved memory and cognitive performance after Cortexin administration. The improvements were measurable on standardized cognitive assessments and correlated with normalization of EEG patterns, suggesting genuine neurophysiological improvement rather than subjective placebo effects.
Relationship to Other Brain Peptides
Cortexin can be understood as the complex precursor from which simpler brain-targeting peptides were derived. Pinealon (Glu-Asp-Arg), a synthetic tripeptide, represents a more refined approach to brain bioregulation. While Cortexin contains the full spectrum of cortical peptide sequences, Pinealon is the isolated active sequence targeting circadian rhythm regulation and neuroprotection specifically.
For those interested in other neuroprotective peptides, Semax and Selank offer complementary approaches to brain health from the Russian peptide research tradition. Semax primarily targets cognitive enhancement and neuroprotection, while Selank focuses on anxiolytic and mood-stabilizing effects.
Cortagen: The Synthetic Alternative
Cortagen is a synthetic tetrapeptide that provides more targeted central nervous system support compared to the full Cortexin complex. As a defined tetrapeptide sequence, it offers the advantages of precise dosing, batch-to-batch consistency, and a clearer mechanistic profile. Cortagen has been studied for cerebral cortex function and represents the trend in Khavinson's research toward simpler, more defined peptide sequences that retain the biological activity of their complex precursors.
Vilon (Immune)
The Smallest Bioregulator
Vilon (Lys-Glu) is a synthetic dipeptide, just two amino acids long, making it the smallest functional bioregulator in the Khavinson family. Despite its minimal size, Vilon demonstrates remarkable biological activity in immune regulation. It represents the minimal active sequence isolated from thymic extracts and provides a simpler, more defined alternative to the complex Thymalin preparation.
The fact that a molecule consisting of only two amino acids can produce measurable biological effects initially struck many Western researchers as implausible. However, increasing evidence supports the concept that even very short peptide sequences can interact with DNA and modulate gene expression, as Khavinson's bioregulation theory predicts.
Immunomodulatory Effects
Research on Vilon has demonstrated several key immunological activities:
- T-cell differentiation: Vilon promotes the maturation of naive T-cells into functional subsets, partially compensating for age-related thymic involution
- Cytokine modulation: The dipeptide reduces pro-inflammatory cytokine production, particularly IL-1 beta and TNF-alpha, in aged immune cells
- NK cell enhancement: Natural killer cell activity increases with Vilon treatment, improving innate immune surveillance
- Macrophage regulation: Vilon modulates macrophage activation toward a more balanced M1/M2 phenotype
Gene Expression Effects
Studies on Vilon's molecular mechanisms have revealed that despite its small size, the dipeptide can influence expression of hundreds of genes. Microarray analysis of Vilon-treated cells showed significant changes in gene expression patterns, with particular effects on genes involved in:
- Cell proliferation and differentiation
- Apoptosis regulation
- Immune response coordination
- Inflammation control
- Cell cycle regulation
These findings provide molecular-level support for the bioregulation theory. A dipeptide that can alter the expression of hundreds of genes must be operating at a regulatory level rather than through direct pharmacological receptor binding.
Vilon vs. Thymalin
While both target immune function, they differ in important ways. Thymalin is a complex mixture that broadly stimulates thymic function. Vilon is a defined dipeptide with a more focused mechanism. In practice, Thymalin may be preferred for severe immune dysfunction where broad thymic restoration is needed, while Vilon may suit maintenance protocols or milder immune support. The two can also be combined, though specific combination protocols have not been extensively studied in clinical trials.
Bioavailability Advantage
Being just two amino acids, Vilon has excellent bioavailability. It's easily absorbed through multiple routes of administration including sublingual, oral, and injectable. Its small size also means rapid distribution throughout the body and efficient cellular uptake. This makes Vilon one of the most practical bioregulators for clinical use, with the additional advantage of straightforward and inexpensive synthesis.
Prostatilen (Prostate)
Addressing Prostate Aging
Prostatilen is a polypeptide complex derived from bovine prostate glands and represents one of the six pharmaceutical-grade Khavinson preparations. It targets prostate function specifically, making it one of the few peptide bioregulators developed for a male-specific organ system.
Benign prostatic hyperplasia (BPH) affects the majority of men over 50, with prevalence increasing steadily with age. Chronic prostatitis affects up to 15% of men at some point in their lives. Both conditions significantly impact quality of life and can lead to serious complications if left untreated.
Mechanism of Action
Prostatilen works through multiple pathways relevant to prostate health:
- Anti-inflammatory effects: Reduces inflammation in prostate tissue, addressing a key driver of both BPH and chronic prostatitis
- Microcirculation improvement: Enhances blood flow within prostate tissue, improving nutrient delivery and waste removal
- Smooth muscle regulation: Modulates smooth muscle tone in the prostate and bladder neck, improving urinary flow
- Immune modulation: Enhances local immune function while reducing autoimmune-type inflammation
Clinical Data
Prostatilen has accumulated substantial clinical evidence in Russian urological practice. Studies have reported:
- Significant improvement in International Prostate Symptom Score (IPSS) in BPH patients
- Reduced prostate volume on ultrasound assessment
- Improved urinary flow rates
- Decreased pain scores in chronic prostatitis patients
- Enhanced quality of life measures
While these results are promising, they come primarily from Russian clinical studies and have not been independently replicated in large-scale Western clinical trials.
Additional Organ-Specific Bioregulators
Pinealon (Brain and Circadian Rhythm)
Pinealon (Glu-Asp-Arg) is a synthetic tripeptide derived from pineal gland tissue. While Epithalon is the more famous pineal bioregulator, Pinealon offers distinct neuroprotective and circadian-regulating properties that complement Epithalon's telomere-focused effects.
Key research findings on Pinealon include:
- Neuroprotection: In prenatal rat models, Pinealon reduced reactive oxygen species (ROS) accumulation and decreased the number of necrotic cells in brain tissue exposed to oxidative stress
- Cognitive enhancement: A study of 72 patients with traumatic brain injury showed improved memory and cognitive performance after Pinealon treatment
- Circadian regulation: By supporting pineal gland function, Pinealon helps optimize melatonin production, leading to improved sleep quality and circadian rhythm stability
- Anti-apoptotic effects: Pinealon has demonstrated ability to reduce caspase-3 activity in the brains of aged rats after carotid artery occlusion, suggesting protection against ischemia-induced cell death
For a thorough exploration of Pinealon's neuroprotective mechanisms and clinical applications, see our dedicated Pinealon Sleep Peptide Report.
Figure 3: Pinealon's neuroprotective mechanisms in brain tissue, including ROS reduction and circadian regulation
Vesugen (Vascular System)
Vesugen (Lys-Glu-Asp) is a synthetic tripeptide targeting blood vessel tissue. Vascular aging is a primary driver of cardiovascular disease, stroke, and organ dysfunction across the body. By specifically targeting vascular endothelial cells, Vesugen aims to restore youthful blood vessel function.
Research on Vesugen has shown:
- Improved endothelial function markers in aged blood vessels
- Enhanced nitric oxide production, supporting vessel dilation and blood pressure regulation
- Reduced vascular inflammation and endothelial cell senescence
- Improved microcirculation in multiple tissue beds
Vesugen is often combined with other bioregulators in longevity protocols, since vascular health underpins the function of every other organ system. Healthy blood vessels ensure adequate nutrient and oxygen delivery to all tissues.
For those interested in vascular health peptides, GHK-Cu offers complementary vascular and tissue-remodeling benefits through copper-peptide-mediated mechanisms.
Cartalax (Cartilage and Musculoskeletal System)
Cartalax (Ala-Glu-Asp) is a synthetic tripeptide targeting cartilage tissue. Joint degeneration is one of the most common and debilitating aspects of aging, affecting mobility, quality of life, and independence. Cartalax was developed to support cartilage maintenance and regeneration at the gene expression level.
Research findings include:
- Stimulation of chondrocyte (cartilage cell) proliferation and matrix production
- Upregulation of collagen type II and proteoglycan synthesis
- Reduction in matrix metalloproteinase (MMP) activity, which drives cartilage breakdown
- Anti-inflammatory effects within joint tissue
For complementary musculoskeletal support, BPC-157 offers well-documented healing properties for tendons, ligaments, and other connective tissues through different but complementary mechanisms.
Livagen (Liver)
Livagen (Lys-Glu-Asp-Trp) is a synthetic tetrapeptide targeting liver (hepatic) tissue. The liver's remarkable regenerative capacity declines with age, and accumulated damage from environmental toxins, medications, and metabolic stress gradually impairs hepatic function. Livagen aims to restore optimal gene expression patterns in hepatocytes.
Research on Livagen has demonstrated:
- Activation of chromatin in hepatocytes, shifting condensed heterochromatin toward transcriptionally active euchromatin
- Upregulation of albumin synthesis, a key marker of liver synthetic function
- Enhanced detoxification enzyme activity
- Improved hepatocyte regeneration capacity
Interestingly, some of the most detailed mechanistic work on the DNA-binding properties of Khavinson peptides has been done with Livagen. Studies using fluorescence spectroscopy and molecular modeling have demonstrated that the tetrapeptide binds specifically to certain DNA sequences in the promoter regions of liver-specific genes, providing direct evidence for the epigenetic regulation mechanism that underlies the entire bioregulator theory.
Retinalamin (Retina)
Retinalamin is a polypeptide complex derived from bovine retinal tissue. It's one of the six pharmaceutical-grade Khavinson preparations and is used clinically in Russia for retinal diseases including age-related macular degeneration, diabetic retinopathy, and other degenerative retinal conditions.
Clinical data has shown improvements in visual acuity, visual field parameters, and retinal function (as measured by electroretinography) in treated patients. The compound appears to work by restoring gene expression patterns in retinal cells, reducing oxidative damage, and improving retinal blood flow.
Thymogen (Immunity)
Thymogen is another immune-regulating peptide in the Khavinson family, though it differs from Thymalin in composition and mechanism. While Thymalin is a complex thymic extract and Vilon is a minimal dipeptide, Thymogen occupies a middle ground as a defined pharmaceutical preparation used for immunocorrection.
Its clinical applications include post-surgical immune support, treatment of recurrent infections in immunocompromised patients, and adjunctive therapy in cancer treatment to support immune function during chemotherapy.
Figure 4: Organ-specific targeting map of major Khavinson peptide bioregulators
Summary Table of All Major Bioregulators
| Peptide | Sequence | Target Organ | Key Function | Type |
|---|---|---|---|---|
| Epithalon | Ala-Glu-Asp-Gly | Pineal gland | Telomerase activation, melatonin restoration | Synthetic tetrapeptide |
| Pinealon | Glu-Asp-Arg | Brain / Pineal | Neuroprotection, circadian regulation | Synthetic tripeptide |
| Thymalin | Complex mixture | Thymus | Immune restoration, T-cell maturation | Natural extract |
| Vilon | Lys-Glu | Thymus / Immune | Immune modulation, anti-inflammatory | Synthetic dipeptide |
| Cortexin | Complex mixture | Brain / CNS | Neuroprotection, cognitive enhancement | Natural extract |
| Cortagen | Tetrapeptide | Brain / CNS | Cerebral cortex support | Synthetic tetrapeptide |
| Vesugen | Lys-Glu-Asp | Blood vessels | Vascular endothelial support | Synthetic tripeptide |
| Cartalax | Ala-Glu-Asp | Cartilage | Chondrocyte support, joint health | Synthetic tripeptide |
| Livagen | Lys-Glu-Asp-Trp | Liver | Hepatocyte function, detoxification | Synthetic tetrapeptide |
| Prostatilen | Complex mixture | Prostate | Prostate function, anti-inflammatory | Natural extract |
| Retinalamin | Complex mixture | Retina | Retinal cell protection, visual function | Natural extract |
| Thymogen | Defined peptide | Immune system | Immunocorrection | Pharmaceutical |
Clinical Evidence Review
Overview of the Evidence Base
Evaluating the Khavinson peptide bioregulator evidence requires understanding both its strengths and its limitations. On the strength side, we have one of the longest-running programs of anti-aging research in history, with consistent results across multiple peptides, multiple species, and decades of follow-up. On the limitation side, the vast majority of published studies come from a single research group in Russia, and the studies don't always meet the methodological standards expected by Western regulatory agencies.
Let's examine the evidence systematically.
Animal Studies: Lifespan Extension
The animal lifespan data is among the most impressive in gerontology research. Across multiple species and multiple peptide preparations, consistent lifespan extensions of 20% to 40% have been demonstrated. To put this in perspective, a 30% lifespan extension in mice would be equivalent to extending human life from 80 years to 104 years.
| Species | Peptide(s) | Lifespan Extension | Study Duration |
|---|---|---|---|
| Drosophila | Epithalon | 11-16% | Full lifespan |
| Mice | Epithalamin | 25-30% | Full lifespan |
| Mice | Thymalin | 25-30% | Full lifespan |
| Rats | Epithalamin + Thymalin | 30-40% | Full lifespan |
| Mice | Combined bioregulators | 20-40% | Full lifespan |
These results are not outliers. They've been reproduced across multiple experiments spanning more than two decades. The consistency of the data across different species adds biological plausibility, as compounds that extend life in both insects and mammals likely target conserved aging mechanisms.
Human Clinical Studies: Mortality and Morbidity
The human data, while not from randomized controlled trials meeting FDA standards, is nonetheless extensive. The primary clinical dataset includes 266 elderly patients followed for 14 to 20 years after initial treatment. This length of follow-up exceeds almost any pharmaceutical clinical trial.
Key findings from the human studies:
- Mortality reduction: Up to 4.1-fold decrease in mortality with combined annual Thymalin + Epithalamin treatment
- Disease incidence: Significant reductions in respiratory infections (2.0-2.4x), ischemic heart disease, hypertension, osteoarthrosis, and osteoporosis
- Biomarker normalization: Immune, endocrine, cardiovascular, and neurological biomarkers restored to middle-aged reference ranges
- Functional improvement: Patients maintained higher levels of physical and cognitive function compared to age-matched controls
Figure 5: Mortality reduction data from the St. Petersburg Institute longevity study (14-20 year follow-up)
Mechanistic Evidence
The molecular-level evidence supporting the bioregulation mechanism has grown steadily:
- Telomere data: Epithalon's ability to activate telomerase and extend telomeres has been confirmed in both Russian studies and, more recently, by independent researchers (Biogerontology, 2025)
- DNA binding: Fluorescence spectroscopy and molecular modeling have demonstrated direct binding of short peptides to specific DNA sequences
- Gene expression profiling: Microarray studies show tissue-specific gene expression changes consistent with the bioregulation theory
- Epigenetic modifications: Changes in histone acetylation and DNA methylation patterns have been documented following peptide treatment
- Protein synthesis: Normalized protein synthesis in target tissues following bioregulator administration has been confirmed in multiple studies
Limitations and Criticisms
Honest assessment requires acknowledging the limitations:
- Single-group origin: Virtually all published studies come from Khavinson's institute or closely affiliated Russian research centers
- Study design: Most human studies are observational or open-label rather than double-blind, randomized, placebo-controlled trials
- Publication bias: As with any long-running research program, there may be unpublished negative results
- Replication gap: Independent Western replication has been limited, though it has begun to appear
- Regulatory status: No Khavinson bioregulator has undergone the FDA approval process, limiting assessment by Western regulatory standards
Important Context
The lack of Western clinical trials does not invalidate the Russian research, but it does mean the evidence has not been subjected to the full scrutiny of independent replication, regulatory review, and peer analysis by groups without conflicts of interest. Anyone considering bioregulator peptides should weigh this context when making decisions. The evidence is promising and extensive, but it does not yet meet the gold standard of independently replicated, randomized controlled trials.
Emerging Independent Validation
The tide is beginning to turn regarding independent validation. Several developments are worth noting:
- The 2025 Biogerontology paper confirming Epithalon's telomere-extending activity in human cell lines by a non-Russian group
- Growing interest from Western peptide researchers in the bioregulation mechanism
- Publication of peptide-DNA binding studies by independent biophysics groups
- Increasing clinical use and case report publication from integrative medicine practitioners worldwide
As more researchers outside Russia engage with these compounds, the evidence base will continue to mature. For now, the Khavinson peptide bioregulators represent one of the most intriguing and underexplored frontiers in anti-aging medicine.
Complementary Peptide Strategies
Building a Comprehensive Longevity Protocol
Khavinson bioregulators don't exist in isolation. They can be integrated with other well-studied peptides and compounds to create comprehensive anti-aging protocols. Understanding how bioregulators fit within the broader peptide landscape helps practitioners and individuals make informed choices about their longevity strategies.
Mitochondrial Support: MOTS-c
While Khavinson bioregulators primarily target organ-specific gene expression, mitochondrial function represents another critical axis of aging. MOTS-c is a mitochondrial-derived peptide that improves metabolic function, insulin sensitivity, and exercise capacity. Combining MOTS-c with organ-specific bioregulators addresses aging at both the cellular energy level and the tissue-specific gene expression level. For detailed research on this compound, see our MOTS-c Mitochondrial Peptide Report.
NAD+ Restoration
NAD+ levels decline significantly with age, impairing cellular energy production, DNA repair, and sirtuin activity. NAD+ supplementation or precursor therapy pairs well with bioregulators because they target complementary aging mechanisms. Bioregulators restore gene expression patterns while NAD+ ensures cells have the energy to execute those restored genetic programs.
Tissue Repair: BPC-157 and GHK-Cu
BPC-157 excels at healing damaged tissues, from gut lining to tendons and ligaments. GHK-Cu provides copper-peptide-mediated tissue remodeling and regeneration. Both of these complement bioregulators by providing acute repair capabilities alongside the bioregulators' long-term gene expression optimization.
Cognitive Stack
For brain-focused protocols, combining Pinealon or Cortagen with Semax (cognitive enhancement, BDNF upregulation) and Selank (anxiolytic, mood stabilization) creates a comprehensive neuroprotective and cognitive-enhancing stack that addresses brain aging from multiple angles.
To explore your options and find the right peptide combination for your goals, try the Free Assessment on FormBlends, or use the Dosing Calculator to plan your protocol.
Dosing Protocols
Medical Supervision Required
All dosing information is provided for educational purposes based on published research and clinical protocols. Peptide bioregulators should only be used under the guidance of a qualified healthcare provider. Individual needs vary, and what's appropriate for one person may not be suitable for another. Always consult with a knowledgeable clinician before starting any peptide protocol.
General Principles of Bioregulator Dosing
Khavinson bioregulator dosing follows fundamentally different principles than conventional pharmaceutical dosing. The key concepts are:
- Cyclic dosing: Short treatment courses followed by extended breaks, rather than continuous daily use
- Cascade initiation: Brief exposure triggers epigenetic changes that persist for months
- Tissue saturation: Oral courses run longer (30 days) to achieve tissue-level peptide concentrations through gastrointestinal absorption
- Age-dependent initiation: Khavinson recommended beginning bioregulator protocols between ages 40 and 45
- Seasonal timing: Many protocols align with spring and autumn cycles, though the evidence for seasonal specificity is limited
Injectable Protocols
Injectable bioregulators provide the highest bioavailability and the most direct tissue exposure. The clinical studies that generated the landmark longevity data used injectable (intramuscular) protocols.
| Peptide | Daily Dose | Route | Course Length | Courses/Year |
|---|---|---|---|---|
| Epithalon | 5-10 mg | SC or IM | 10-20 days | 1-2 |
| Thymalin | 10-20 mg | IM | 5-10 days | 1-2 |
| Cortexin | 10 mg | IM | 10 days | 1-2 |
| Prostatilen | 5-10 mg | IM or rectal | 5-10 days | 1-2 |
| Retinalamin | 5 mg | Para-bulbar or IM | 10 days | 1-2 |
Figure 6: Typical bioregulator dosing cycle showing the short course/long break pattern
Oral/Capsule Protocols
Oral bioregulator supplements (marketed under the Cytomax brand in Russia) provide a non-injectable option. The longer course duration compensates for lower bioavailability through the oral route.
| Format | Dose | Frequency | Course Length | Break Period |
|---|---|---|---|---|
| Capsules (Cytomax) | 1-2 capsules (10mg each) | 1-2 times daily | 30 days | 3-6 months |
| Sublingual drops | 5-6 drops (0.25-0.35ml) | 3-4 times daily | 30 days | 60 days |
Combination Protocols
The clinical data showing the greatest benefits used combination protocols. The classic Khavinson longevity protocol combines thymic and pineal bioregulators:
Classic Longevity Protocol (based on St. Petersburg clinical studies)
- Course 1 (Spring): Thymalin 10mg IM daily for 10 days + Epithalon 10mg SC daily for 10 days (can be concurrent or sequential)
- Break: 5-6 months
- Course 2 (Autumn): Repeat Course 1
- Maintenance: Continue annual cycles. The 4.1-fold mortality reduction was observed with 6 years of annual courses.
Comprehensive Bioregulation Protocol (advanced)
For a more comprehensive approach targeting multiple organ systems:
- Phase 1 - Immune + Pineal: Thymalin + Epithalon (10 days each)
- Phase 2 - Brain + Vascular: Cortagen or Pinealon + Vesugen (10 days each, starting 2 weeks after Phase 1)
- Phase 3 - Tissue-specific: Add organ-specific bioregulators as needed (Livagen, Cartalax, Prostatilen) in subsequent cycles
- Repeat: Full cycle twice per year
Practical Considerations
- Timing: Morning administration is generally preferred for most bioregulators, though Pinealon and Epithalon may also be given in the evening to align with circadian patterns
- Storage: Reconstituted peptides should be refrigerated and used within the stability window specified by the manufacturer
- Monitoring: Baseline blood work before starting and follow-up testing after 2-3 months helps assess response. Key markers include CBC with differential, thyroid panel, hormonal panel, and inflammatory markers
- Starting age: While Khavinson recommended starting at 40-45, some practitioners begin with lower doses at earlier ages for prevention in those with accelerated aging biomarkers
For help calculating your personalized dosing, try the FormBlends Dosing Calculator. You can also take the Free Assessment to get a customized peptide recommendation based on your specific health goals.
Safety and Tolerability
Decades of Safety Data
One of the most reassuring aspects of Khavinson peptide bioregulators is their safety profile. With over 30 years of clinical use in Russia and extensive preclinical testing, these compounds have demonstrated an exceptionally clean safety record.
Preclinical Safety Studies
- Acute toxicity: Testing at doses 5,000 times higher than therapeutic levels produced no serious adverse reactions in animal models
- Chronic toxicity: Long-term administration at 100 to 1,000 times normal therapeutic doses revealed no side effects in animals
- Mutagenicity: No mutagenic effects have been detected in standard mutagenicity assays
- Carcinogenicity: No carcinogenic effects observed; in fact, several bioregulators show anticarcinogenic properties
- Teratogenicity: No teratogenic effects documented in animal reproductive studies
- Immunogenicity: Short peptides of 2-7 amino acids are too small to trigger antibody formation, eliminating the risk of immunogenic reactions
Clinical Safety Data
Across the clinical trials and decades of clinical use, the reported adverse event profile is minimal:
| Adverse Event | Frequency | Severity | Management |
|---|---|---|---|
| Injection site reaction | Occasional (< 5%) | Mild | Rotates injection sites, resolves spontaneously |
| Mild headache | Rare (< 3%) | Mild | Usually during initiation, resolves within days |
| Transient fatigue | Rare | Mild | Self-limiting, may reflect initial cellular adaptation |
| Allergic reaction | Not documented | N/A | N/A |
| Systemic toxicity | Not documented | N/A | N/A |
Safety Profile Summary
No significant toxic, allergic, or adverse effects have been reported across the entire history of Khavinson bioregulator clinical use. This includes data from thousands of patients treated over periods of up to 20 years. The safety record is consistent with the theoretical expectation: short peptides of 2-7 amino acids are naturally occurring in the body, are rapidly metabolized, and do not accumulate in tissues.
Why Are They So Safe?
Several factors contribute to the excellent safety profile:
- Natural components: The amino acid sequences used in bioregulators are identical to peptides naturally present in the body. They don't introduce foreign molecules.
- Small size: At 2-7 amino acids, these peptides are too small to trigger immune responses or form toxic metabolites
- Rapid metabolism: Short peptides are quickly broken down by ubiquitous peptidases into their constituent amino acids, preventing accumulation
- Physiological concentrations: Therapeutic doses work at concentrations within the normal physiological range, not at supraphysiological levels
- Regulatory mechanism: Because they work by modulating gene expression rather than blocking or overstimulating receptors, there's no risk of the "overshoot" effects common with conventional drugs
Contraindications and Precautions
While the safety profile is favorable, standard medical precautions apply:
- Pregnancy and breastfeeding: Not recommended due to lack of specific safety data in this population
- Active malignancy: Some practitioners exercise caution with telomerase-activating peptides like Epithalon in patients with active cancer, as telomerase activation could theoretically benefit cancer cells. However, the animal data actually shows anticancer effects, and the clinical studies did not reveal increased cancer incidence.
- Autoimmune conditions: Immune-modulating bioregulators (Thymalin, Vilon) should be used cautiously in patients with active autoimmune disease, as immune activation could theoretically exacerbate symptoms
- Organ transplant recipients: Immunomodulatory peptides are contraindicated in transplant patients on immunosuppressive therapy
- Known peptide hypersensitivity: While allergic reactions have not been documented, patients with known peptide allergies should exercise caution
Drug Interactions
No significant drug interactions have been documented for Khavinson peptide bioregulators. Their mechanism of action (epigenetic gene regulation) does not interfere with conventional pharmacological pathways. However, patients on immunosuppressive medications should consult with their physician before using immune-modulating bioregulators, as the restored immune function could potentially alter the effectiveness of immunosuppressive therapy.

Figure 7: Comparative safety profile of Khavinson peptide bioregulators vs. conventional anti-aging interventions
The Future of Peptide Bioregulation
Where the Field is Heading
The Khavinson bioregulation model is entering a new phase. After decades as a largely Russian field of research, interest from Western researchers, clinicians, and the broader peptide therapy community is growing rapidly. Several trends are shaping the future of this field.
Independent Replication and Validation
The 2025 independent confirmation of Epithalon's telomere-extending properties marks the beginning of what many hope will be a broader wave of independent validation. As more researchers outside Russia obtain these peptides and conduct their own studies, the evidence base will either be strengthened or challenged. Either outcome will be valuable for the field.
Advanced Analytical Techniques
Modern molecular biology tools, including single-cell RNA sequencing, CRISPR-based gene editing for mechanistic studies, and advanced proteomics, offer the ability to characterize bioregulator mechanisms at a level of detail that wasn't possible when much of the original research was conducted. Applying these tools to Khavinson peptides could either validate or refine the proposed mechanisms.
Personalized Bioregulation
As biomarker testing becomes more accessible and sophisticated, the possibility of personalizing bioregulator protocols based on individual epigenetic, genomic, and proteomic profiles becomes increasingly realistic. Rather than applying a standard protocol to all patients, future approaches might use biological age testing, methylation clocks, and organ-specific biomarkers to determine exactly which bioregulators each individual needs and when.
Integration with Broader Longevity Science
The bioregulation approach is increasingly being integrated with other evidence-based longevity strategies. Combining organ-specific bioregulators with mitochondrial peptides, NAD+ precursors, senolytics, and other anti-aging compounds creates multi-pronged protocols that address aging at multiple levels simultaneously. This integrative approach, rather than relying on any single compound, likely represents the most effective strategy for extending healthspan.
For more on how bioregulators fit within the broader world of anti-aging peptide research, explore our Biohacking Hub and the complete Peptide Research Hub.
Molecular Biology of Peptide-DNA Interactions
How a Two-Amino-Acid Peptide Can Regulate Gene Expression
When Western-trained molecular biologists first encounter the claim that a dipeptide like Vilon (Lys-Glu) can regulate the expression of hundreds of genes, skepticism is understandable. Conventional pharmacology teaches that biological activity requires a certain molecular complexity - receptor binding sites, tertiary protein structures, specific three-dimensional conformations. How can two amino acids strung together accomplish anything meaningful at the DNA level?
The answer lies in the difference between pharmacological drug action and informational signaling. A conventional drug works by physically occupying a receptor binding site, blocking or activating it through steric and electrostatic complementarity. This requires significant molecular complexity. But Khavinson peptides don't work through receptors. They work through direct nucleic acid interaction, where even very short sequences carry meaningful information.
Consider the genetic code itself. A single codon of three nucleotides specifies an amino acid. Three letters carry enough information to direct protein synthesis. In the same way, a short peptide of two or three amino acids carries enough structural information to interact with specific DNA sequences through complementary charge distributions, hydrogen bonding patterns, and hydrophobic interactions.
Evidence for Direct DNA Binding
The most direct evidence for peptide-DNA interaction comes from fluorescence spectroscopy studies conducted by Fedoreyeva, Kireev, Khavinson, and Vanyushin (2011). Using fluorescently labeled short peptides, they demonstrated several key findings:
- Nuclear penetration: Labeled peptides were observed inside cell nuclei of HeLa cells within minutes of application, confirming that short peptides cross both the plasma membrane and the nuclear envelope without requiring active transport mechanisms
- Sequence-specific binding: Different peptide sequences showed preferential binding to different deoxyribooligonucleotide sequences in vitro, supporting the idea of complementary peptide-DNA recognition
- Promoter interaction: Peptides accumulated preferentially in gene promoter regions rather than distributing uniformly across the genome
- Concentration dependence: Binding showed characteristic saturation kinetics, consistent with specific interaction rather than non-specific electrostatic association
These findings were extended by molecular modeling studies that predicted the binding conformations of specific peptide-DNA complexes. The models showed that short peptides fit into the major groove of DNA at specific sequence contexts, forming hydrogen bonds with base pairs and backbone phosphates. The binding energy calculations predicted stable complexes that could persist long enough to influence transcription factor access.
The Methylation Interference Model
One of the most compelling mechanistic proposals explains how peptide binding to DNA could produce lasting changes in gene expression without the peptide needing to remain permanently bound. The methylation interference model, proposed by Vanyushin and Khavinson (2010), works as follows:
- A short peptide binds to a specific DNA sequence in a gene promoter region
- While the peptide is bound, DNA methyltransferases cannot access that region
- During the next round of DNA replication, the daughter strand at that locus remains unmethylated
- The unmethylated promoter is transcriptionally active, meaning the gene is switched on
- Once established, the unmethylated state can be maintained through subsequent cell divisions, even after the peptide has been metabolized
This model elegantly explains several puzzling features of bioregulator therapy. It explains why short treatment courses produce long-lasting effects - the epigenetic change is self-perpetuating. It explains tissue specificity - different tissues have different methylation patterns, so the same peptide may have different effects depending on the local epigenetic context. And it explains the cascade effect - a single demethylation event at a master regulatory gene can cascade through downstream gene networks.
Chromatin Remodeling Effects
Beyond DNA methylation, Khavinson peptides have been shown to affect chromatin structure directly. Chromatin exists on a spectrum from tightly packed heterochromatin (transcriptionally silent) to loosely organized euchromatin (transcriptionally active). The ratio of heterochromatin to euchromatin changes with age, with aging cells showing progressively more heterochromatin and correspondingly reduced gene expression.
Studies on buccal epithelium cells from elderly subjects showed that short peptide treatment shifted the chromatin balance toward euchromatin (Khavinson, Lezhava, and Malinin, 2004). This shift was tissue-specific, with different peptides affecting chromatin structure in cells from their target tissues preferentially. The chromatin remodeling was associated with increased gene expression and enhanced protein synthesis in the treated cells.
The mechanism likely involves peptide interactions with histone proteins. Histones are positively charged proteins around which DNA wraps, and their post-translational modifications (acetylation, methylation, phosphorylation) determine chromatin accessibility. Short peptides, through their charge distributions and hydrogen bonding capabilities, may influence histone modifications or directly compete with histone-DNA interactions at specific loci.
Transcription Factor Modulation
A third level of gene regulation involves transcription factors, the proteins that bind to promoter and enhancer regions to activate or repress gene transcription. Khavinson peptides may influence transcription factor binding in at least two ways:
- Direct competition: By binding to the same DNA sequences that transcription factors recognize, short peptides could either block repressor binding (activating genes) or prevent activator binding (silencing genes)
- Allosteric effects: Peptide binding to nearby DNA sequences could alter local DNA conformation, making adjacent transcription factor binding sites more or less accessible
Gene expression profiling studies using microarray analysis have confirmed that bioregulator peptides produce characteristic, reproducible changes in transcription factor activity patterns. These changes are consistent with the restoration of younger gene expression profiles in aged tissues.
RNA-Level Regulation
More recent work has uncovered additional regulatory mechanisms at the RNA level. Short peptides can influence:
- mRNA stability: Some peptides appear to affect the half-life of specific mRNA molecules, increasing or decreasing the time available for translation into protein
- Translation efficiency: Peptide-mediated changes in ribosomal function and tRNA availability can alter how efficiently mRNA is translated
- microRNA expression: Emerging evidence suggests that some bioregulator effects may be mediated through changes in microRNA expression, adding another layer of regulatory complexity
These multi-level regulatory effects help explain why bioregulator peptides can produce such comprehensive changes in cellular function. By operating simultaneously at the DNA, chromatin, transcription factor, and RNA levels, a single short peptide can orchestrate a coordinated shift in the overall gene expression program of a cell.
Independent Verification: The 2025 Biogerontology Study
A significant milestone in the validation of Khavinson's molecular mechanisms came with a 2025 study published in Biogerontology by researchers outside Khavinson's immediate circle. This study confirmed that Epitalon increases telomere length in human cell lines through both telomerase upregulation and alternative lengthening of telomeres (ALT) activity.
The significance of this finding extends beyond telomere biology. It demonstrates that at least one core mechanism proposed by the Khavinson group - that a short synthetic tetrapeptide can produce specific, measurable molecular effects on gene expression and cellular function - is reproducible by independent researchers using standard molecular biology methods. This doesn't validate every claim made for every bioregulator, but it does establish that the basic premise of short peptide bioregulation is scientifically sound.
| Level of Regulation | Mechanism | Evidence Strength | Key References |
|---|---|---|---|
| DNA binding | Direct sequence-specific interaction in promoter regions | Strong (fluorescence, molecular modeling) | Fedoreyeva et al., 2011 |
| DNA methylation | Blocking methyltransferase access, preventing gene silencing | Moderate (theoretical model with supporting data) | Vanyushin & Khavinson, 2010 |
| Chromatin remodeling | Shifting heterochromatin to euchromatin balance | Strong (direct observation in human cells) | Khavinson et al., 2004 |
| Transcription factor modulation | Competition with or allosteric effects on transcription factors | Moderate (gene expression profiling data) | Multiple studies |
| RNA regulation | mRNA stability, translation efficiency, microRNA changes | Emerging (preliminary data) | Recent publications |
| Telomerase activation | Upregulation of telomerase enzyme activity | Strong (independently confirmed) | Biogerontology, 2025 |
Epithalon Research: A Detailed Analysis
The Pineal Gland and Aging: Why It Matters
To fully appreciate Epithalon's significance, you need to understand the pineal gland's central role in the aging process. The pineal gland, a tiny pine cone-shaped structure deep in the brain, serves as the body's master clock. It produces melatonin in response to darkness, synchronizing virtually every biological rhythm in the body - from sleep-wake cycles to hormonal fluctuations, immune activity, body temperature, and even gene expression timing.
As we age, the pineal gland undergoes calcification. Calcium deposits gradually accumulate in the tissue, reducing its functional capacity. By age 60, most people have significant pineal calcification, and melatonin production may be reduced by 60% to 80% compared to young adult levels. This decline ripples through every system the pineal gland regulates.
Reduced melatonin production leads to disrupted sleep architecture, with less time spent in deep, restorative sleep stages. This sleep disruption accelerates brain aging, impairs memory consolidation, and reduces the brain's ability to clear metabolic waste during sleep (a process now understood to be critical for preventing neurodegenerative disease). Melatonin is also one of the body's most potent endogenous antioxidants, and its decline leaves tissues more vulnerable to oxidative damage.
Beyond melatonin, the pineal gland produces a range of peptides and other signaling molecules that influence the neuroendocrine system broadly. The pineal communicates with the thymus, the adrenal glands, the gonads, and the thyroid through both direct neural connections and circulating hormonal signals. When pineal function declines, these downstream systems are affected in a cascade of interconnected dysfunction.
This is why Khavinson identified the pineal gland as one of the two most important targets for anti-aging intervention (the thymus being the other). Restoring pineal function doesn't just fix one organ - it helps restore the entire neuroendocrine network that coordinates healthy aging.
From Epithalamin to Epithalon: The Refinement Process
The journey from crude pineal extract to synthetic tetrapeptide took decades of methodical biochemical work. The original preparation, Epithalamin, was obtained by acid extraction of bovine pineal glands, followed by multiple purification steps including gel filtration chromatography and ion exchange chromatography. The resulting preparation contained a complex mixture of peptides ranging from 2 to 20 amino acids in length, along with trace amounts of amino acids, nucleotides, and minerals.
While Epithalamin showed clear biological activity in animal studies and early clinical trials, working with a complex extract presented several challenges. Batch-to-batch consistency was difficult to guarantee, since the peptide composition could vary depending on the source tissue, extraction conditions, and purification parameters. Regulatory approval for complex biological extracts is more difficult to obtain than for defined synthetic compounds. And mechanistic studies were complicated by the presence of multiple active components.
Khavinson's team systematically fractionated Epithalamin and tested each fraction for biological activity. Through this process of elimination, they identified the tetrapeptide Ala-Glu-Asp-Gly as the minimal sequence responsible for the extract's primary biological effects. When synthesized and tested independently, this tetrapeptide reproduced the key activities of the full extract: telomerase activation, melatonin production stimulation, and antioxidant enhancement.
The synthetic version was named Epithalon (sometimes spelled Epitalon) and became the standard preparation for subsequent research. Synthesis of the tetrapeptide is straightforward using standard solid-phase peptide synthesis methods, ensuring high purity, consistent composition, and scalable production.
Telomerase Biology: The Full Picture
Epithalon's ability to activate telomerase has attracted the most attention from the broader scientific community, and for good reason. Telomere biology sits at the intersection of aging research, cancer biology, and regenerative medicine.
Telomeres are repetitive DNA sequences (TTAGGG in humans) at the ends of chromosomes. They serve as protective caps, preventing chromosome ends from being recognized as DNA breaks and preventing chromosomes from fusing with each other. Each time a cell divides, the replication machinery cannot fully copy the very end of a linear chromosome, resulting in progressive telomere shortening with each division.
When telomeres become critically short, cells activate either of two responses: senescence (permanent growth arrest) or apoptosis (programmed cell death). Both responses prevent further division and are important tumor suppression mechanisms. But in the context of aging, the accumulation of senescent cells and the loss of cell renewal capacity drive tissue dysfunction and the visible signs of aging.
Telomerase is the enzyme that can extend telomeres, adding TTAGGG repeats to chromosome ends. Most adult somatic cells have very low or absent telomerase activity, which is why telomeres shorten with age. Stem cells, germ cells, and certain immune cells maintain higher telomerase activity, enabling greater replicative capacity.
Epithalon's activation of telomerase in somatic cells effectively extends their replicative lifespan. The 2003 Khavinson study demonstrated this in peripheral blood cells of elderly patients, showing measurable telomere lengthening after treatment. The 2025 independent study confirmed the mechanism and added the finding that Epithalon can also activate alternative lengthening of telomeres (ALT), a telomerase-independent mechanism for telomere maintenance that is particularly interesting because it may represent a backup pathway in cells where telomerase activation is insufficient.
The Cancer Question
Any discussion of telomerase activation must address the cancer concern. Approximately 85% to 90% of cancers activate telomerase to achieve the unlimited replication needed for tumor growth. Does activating telomerase in normal cells increase cancer risk?
The available evidence actually suggests the opposite. In Khavinson's animal studies, Epithalon-treated animals showed reduced tumor incidence compared to controls, not increased incidence. Several factors may explain this apparent paradox:
- Immune enhancement: By improving overall immune function (both directly and through thymic support from co-administered bioregulators), Epithalon may enhance immune surveillance against nascent tumors
- Reduced senescence burden: Senescent cells, which accumulate when telomeres shorten, produce pro-inflammatory cytokines (the senescence-associated secretory phenotype, or SASP) that can actually promote tumor development in neighboring cells. By reducing the number of senescent cells through telomere extension, Epithalon may reduce this pro-tumorigenic environment
- DNA repair enhancement: Some evidence suggests Epithalon improves DNA repair capacity, reducing the accumulation of mutations that drive cancer development
- Antioxidant effects: Through melatonin restoration and direct antioxidant properties, Epithalon reduces oxidative DNA damage, a major source of cancer-causing mutations
Still, the cancer question remains theoretically relevant, and most practitioners exercise appropriate caution when considering Epithalon for patients with active malignancies or high cancer risk. The long-term observational data is reassuring but does not constitute definitive proof of safety in this regard.
Melatonin Restoration: Beyond Sleep
While melatonin is popularly known as the "sleep hormone," its biological roles extend far beyond sleep regulation. Understanding the full scope of melatonin's functions helps explain why restoring its production through Epithalon has such wide-ranging health effects.
Antioxidant Functions
Melatonin is one of the most potent endogenous antioxidants. Unlike most antioxidants that neutralize one free radical per molecule, melatonin can scavenge multiple reactive oxygen and nitrogen species through a cascade of metabolites. Melatonin also stimulates the production of other antioxidant enzymes including superoxide dismutase (SOD), glutathione peroxidase, and catalase. This amplification effect means that even small increases in melatonin production can significantly enhance overall antioxidant defense.
Kozina, Arutjunyan, and Khavinson (2007) specifically studied the antioxidant properties of pineal gland peptides, confirming that Epithalon and related peptides enhance antioxidant defense mechanisms in aged tissues. The antioxidant effect was particularly pronounced in brain tissue, where oxidative damage is a primary driver of age-related cognitive decline.
Immune Modulation
Melatonin plays a significant role in immune regulation. It enhances natural killer cell activity, stimulates cytokine production by immunocompetent cells, and modulates the balance between pro-inflammatory and anti-inflammatory immune responses. The age-related decline in melatonin contributes to the immunosenescence that characterizes aging, and restoring melatonin production helps reverse this decline.
This immune-enhancing effect of Epithalon's melatonin restoration complements the direct immune support provided by thymic bioregulators like Thymalin. The combination addresses immune aging from both the neuroendocrine (pineal) and lymphoid (thymic) sides, which may explain the amplified mortality reduction seen with combined Epithalon and Thymalin treatment in the clinical studies.
Circadian Rhythm Regulation
Melatonin's primary role is coordinating circadian rhythms throughout the body. Nearly every tissue and organ has its own internal clock, and melatonin serves as the master synchronizing signal that keeps all these clocks aligned. When melatonin production falters, circadian coordination breaks down.
Disrupted circadian rhythms have been linked to accelerated aging, increased cardiovascular disease risk, metabolic dysfunction, cognitive decline, and increased cancer risk. By restoring healthy melatonin production, Epithalon helps re-synchronize the body's circadian network, potentially benefiting every organ system.
Korkushko, Khavinson, Shatilo, and Antonyk-Sheglova (2006) specifically studied the effect of Epithalamin on circadian melatonin rhythm in elderly subjects, confirming restoration of the normal nighttime melatonin peak that is typically blunted in aging.
Pineal Peptide Cross-Talk with the Thymus
One of the more fascinating aspects of Epithalon's biology is the bidirectional communication between the pineal gland and the thymus. Khavinson, Ryzhak, Grigoriev, and Ryadnova (2002) demonstrated that pineal peptide preparations can restore thymic function in aged animals. Conversely, thymic decline accelerates pineal aging.
This pineal-thymic axis creates a vicious cycle in aging: as the pineal declines, thymic involution accelerates, which further impairs the neuroendocrine environment that supports pineal function. Breaking this cycle by supporting both organs simultaneously may explain why the combination of Epithalon (pineal) and Thymalin (thymic) produces effects far exceeding either peptide alone.
The practical implication is clear: longevity protocols should address both the pineal and thymic components of this axis for maximum benefit. The 4.1-fold mortality reduction seen with combined treatment, compared to 1.6-1.8-fold with Epithalon alone and 2.0-2.1-fold with Thymalin alone, strongly supports this combined approach.
Thymalin and the Science of Immunosenescence
Understanding Thymic Involution
The thymus is a bilobed organ located behind the sternum, just above the heart. It's the primary site where T-lymphocytes (T-cells) mature and undergo selection - the process that ensures T-cells can recognize foreign antigens without attacking the body's own tissues. This process, called thymic education, is essential for a functional adaptive immune system.
Thymic involution begins around puberty, driven by rising sex hormone levels, and progresses throughout life. The functional thymic tissue (thymic epithelial space) is gradually replaced by adipose (fat) tissue in a process called thymic adipose involution. By age 25, the thymus has lost approximately 3% of its functional mass per year. By age 50, functional thymic tissue may represent less than 10% of total thymic mass. By age 70 to 80, the thymus is largely vestigial in most individuals.
This involution has profound consequences for immune function:
- Reduced naive T-cell output: The production of new, antigen-naive T-cells drops dramatically, reducing the immune system's ability to respond to novel pathogens
- T-cell repertoire narrowing: As naive T-cell production declines, the diversity of T-cell receptors decreases, creating "holes" in immune coverage
- Memory T-cell accumulation: The T-cell compartment becomes dominated by memory cells specific to previously encountered pathogens, leaving fewer resources for new threats
- Increased senescent T-cells: Aged T-cells accumulate markers of senescence and produce pro-inflammatory cytokines without effective immune function
- Impaired regulatory T-cell function: The balance between effector and regulatory T-cells shifts, contributing to both immunodeficiency and autoimmunity
These changes collectively constitute immunosenescence, and they explain why elderly individuals are more susceptible to infections, respond poorly to vaccines, have higher cancer rates, and experience more autoimmune conditions.
Thymalin's Multi-Target Immunorestoration
Thymalin, as a complex polypeptide extract from young bovine thymus tissue, contains a spectrum of thymic peptides that together support multiple aspects of thymic function. Unlike a single synthetic peptide that targets one pathway, Thymalin provides a comprehensive thymic support profile.
The detailed immunological studies conducted by Khavinson's group, summarized in a comprehensive 2021 review in the International Journal of Molecular Sciences, revealed the following mechanisms:
T-Cell Maturation and Differentiation
Thymalin promotes the maturation of thymocyte precursors into functional T-cell subsets. In aged thymic tissue, the maturation process becomes inefficient, with many thymocytes failing to complete the selection process. Thymalin appears to improve the efficiency of this process, increasing the output of mature, functional T-cells even from aged thymic tissue.
The maturation effect extends to both CD4+ helper T-cells and CD8+ cytotoxic T-cells, with studies showing normalization of the CD4/CD8 ratio toward values typical of younger adults. This ratio is an important indicator of immune balance, as deviations are associated with increased mortality and disease risk in elderly populations.
Cytokine Network Regulation
One of the hallmarks of immunosenescence is "inflammaging" - a state of chronic, low-grade inflammation driven by elevated pro-inflammatory cytokines including IL-6, TNF-alpha, IL-1 beta, and C-reactive protein. This inflammatory state damages tissues, drives cardiovascular disease, promotes neurodegeneration, and paradoxically impairs the immune system's ability to mount effective responses to infections.
Thymalin treatment has been shown to reduce this inflammatory burden by downregulating pro-inflammatory cytokine production and upregulating anti-inflammatory mediators like IL-10. The net effect is a shift from the pro-inflammatory state of aging toward a more balanced inflammatory profile characteristic of younger individuals.
This cytokine-rebalancing effect may explain many of the clinical benefits observed in the long-term studies, including reduced cardiovascular events (driven partly by vascular inflammation), improved cognitive function (neuroinflammation is a key driver of cognitive decline), and reduced incidence of osteoarthritis (joint inflammation).
Natural Killer Cell Enhancement
Natural killer (NK) cells are innate immune cells that play a critical role in tumor surveillance and viral defense. NK cell function declines with age, contributing to increased cancer incidence and susceptibility to viral infections in elderly populations. Thymalin has been shown to enhance NK cell cytotoxicity by 30% to 50% in aged subjects, potentially improving the body's first-line defense against both cancer and infections.
Vaccine Response Improvement
One of the most clinically relevant findings is that Thymalin-treated elderly patients showed improved responses to vaccination. This has significant practical implications, as poor vaccine efficacy in the elderly is a major public health challenge. Enhanced vaccine responses suggest that Thymalin genuinely restores functional adaptive immunity rather than simply altering biomarker levels.
The 266-Patient Longevity Study: Detailed Analysis
The primary clinical dataset for Thymalin comes from a series of studies conducted at the St. Petersburg Institute between 1985 and 2003, ultimately published by Khavinson and Morozov in Neuroendocrinology Letters (2003). This study deserves detailed examination because its results form the foundation of the clinical case for peptide bioregulators.
Study Design
A total of 266 elderly patients (ages 60 to 89) were enrolled and divided into four groups:
- Group 1: Thymalin treatment only
- Group 2: Epithalamin treatment only
- Group 3: Combined Thymalin + Epithalamin treatment
- Group 4: Control (no peptide bioregulator treatment)
Bioregulator treatment was administered during the first 2 to 3 years of observation, with follow-up continuing for a total of 6 to 8 years initially and extending to 14 to 20 years in subsequent analyses. Patients were assessed for cardiovascular, endocrine, immune, and neurological function, along with overall mortality and disease incidence.
Results by System
Cardiovascular system: Treated patients showed improved cardiovascular function indices, with reduced incidence of ischemic heart disease and hypertension. Blood pressure values normalized toward healthy ranges, and markers of cardiovascular risk decreased.
Endocrine system: Hormonal profiles improved significantly, with restoration of circadian cortisol rhythms, normalization of thyroid function markers, and improved glucose metabolism. The Epithalamin group showed the most pronounced endocrine improvements, consistent with the pineal gland's role as a master endocrine regulator.
Immune system: All treated groups showed improved immune parameters, with the Thymalin group showing the most pronounced immunological benefits. T-cell counts increased, NK cell activity improved, and the frequency of respiratory infections dropped 2.0 to 2.4-fold compared to controls.
Nervous system: Cognitive function tests showed better preservation of cognitive abilities in treated patients. Memory, attention, and executive function scores declined more slowly in treated groups compared to controls.
Musculoskeletal system: Reduced incidence of deforming osteoarthrosis and osteoporosis in treated groups, possibly related to reduced inflammation and improved hormonal balance.
Mortality Data
The mortality data is the most striking finding and has been the focus of most discussion:
| Group | Mortality Rate Relative to Control | Interpretation |
|---|---|---|
| Control | 1.0 (reference) | Expected mortality for age-matched untreated elderly |
| Thymalin only | 0.48-0.50 (2.0-2.1x reduction) | Roughly half the mortality rate of controls |
| Epithalamin only | 0.56-0.63 (1.6-1.8x reduction) | Significant reduction but less than Thymalin |
| Combined (single course) | 0.40 (2.5x reduction) | Combined benefit exceeds either alone |
| Combined (annual, 6 years) | 0.24 (4.1x reduction) | Maximum benefit with sustained annual treatment |
The dose-response relationship - with greater benefit from combined treatment and still greater benefit from sustained annual courses - increases confidence in the findings, as it's the pattern you'd expect from a genuine biological effect rather than a statistical artifact.
Thymalin vs. Thymosin Alpha-1: Understanding the Options
For clinicians and individuals interested in thymic support, it's helpful to understand how Thymalin relates to Thymosin Alpha-1, another well-studied thymic peptide. While both target immune function through thymic mechanisms, they differ in important ways.
Thymosin Alpha-1 is a 28-amino-acid peptide originally isolated from thymic tissue and now produced synthetically. It has been extensively studied in Western clinical trials, has regulatory approval in over 35 countries (though not in the US), and has a well-characterized mechanism of action focusing on dendritic cell activation, T-cell maturation, and antiviral defense.
Thymalin, by contrast, is a complex extract containing multiple peptide sequences. Its mechanism is broader but less precisely defined. The trade-off is between the precision and characterization of a single-peptide approach (Thymosin Alpha-1) and the potentially more comprehensive support of a multi-peptide extract (Thymalin).
In practice, both are valid approaches to thymic support, and some advanced protocols use both. The choice may depend on availability, regulatory considerations, and individual clinical needs. For detailed information on Thymosin Alpha-1, see the FormBlends product page.
Brain Bioregulators: Pinealon and Cortagen in Clinical Practice
The Aging Brain: A Multi-Factorial Challenge
Brain aging involves the simultaneous progression of multiple pathological processes that interact and amplify each other. Understanding these processes helps explain why brain bioregulators need to address multiple mechanisms simultaneously and why a single-target approach often falls short.
The first major process is oxidative stress. The brain consumes approximately 20% of the body's oxygen despite representing only 2% of body weight. This high metabolic rate generates correspondingly high levels of reactive oxygen species (ROS). The brain is also rich in polyunsaturated fatty acids that are particularly vulnerable to oxidative damage. Over decades, the cumulative burden of oxidative damage affects neuronal membranes, mitochondrial function, and DNA integrity.
The second process is neuroinflammation. Microglia, the brain's resident immune cells, become progressively activated with age, shifting from a neuroprotective phenotype to a chronically inflammatory one. This "microglial priming" means that aged microglia produce excessive pro-inflammatory cytokines in response to stimuli that would produce only modest responses in younger brains. The resulting chronic neuroinflammation damages neurons and accelerates cognitive decline.
The third process is vascular deterioration. The brain's vasculature undergoes age-related changes including reduced cerebral blood flow, blood-brain barrier breakdown, and microangiopathy. Reduced blood flow means less oxygen and nutrient delivery, while blood-brain barrier leakage allows inflammatory molecules from the periphery to access the brain, further driving neuroinflammation.
The fourth process involves synaptic loss and altered neurotransmitter balance. Age-related decline in synapse density, reduced production of brain-derived neurotrophic factor (BDNF), and altered neurotransmitter synthesis and receptor density all contribute to cognitive decline. The balance between excitatory (glutamate) and inhibitory (GABA) neurotransmission shifts with age, potentially increasing vulnerability to excitotoxic damage.
Finally, protein aggregation - the accumulation of misfolded proteins including amyloid-beta and hyperphosphorylated tau - represents the most severe end of the brain aging spectrum, manifesting clinically as Alzheimer's disease and other neurodegenerative conditions.
Pinealon (EDR Peptide): Mechanisms in Detail
Pinealon, with the amino acid sequence Glu-Asp-Arg (also designated as EDR peptide), addresses multiple aspects of brain aging through its tissue-specific bioregulatory activity. Research has uncovered several distinct but interconnected mechanisms.
Neuroprotection Against Oxidative Stress
In prenatal rat cortical cell cultures exposed to hydrogen peroxide (an oxidative stress model), Pinealon significantly reduced reactive oxygen species accumulation and decreased the number of necrotic cells. The protection was dose-dependent and was associated with changes in gene expression patterns related to antioxidant defense, including upregulation of superoxide dismutase and glutathione pathways.
These findings are consistent with the bioregulation model: rather than acting as a direct antioxidant (like vitamin C or E), Pinealon appears to restore the cell's endogenous antioxidant gene expression to a more youthful, protective level. This approach may be more effective than exogenous antioxidant supplementation because it activates the full spectrum of cellular defense mechanisms rather than providing a single antioxidant compound.
Anti-Apoptotic Activity in Ischemia
Khavinson, Diomina, Lin'kova, and colleagues (2011) studied the effects of short peptides administered before carotid artery occlusion (a model of stroke) in old rats. Pinealon treatment reduced caspase-3 activity in brain tissue. Caspase-3 is the primary executioner enzyme in apoptosis (programmed cell death), and its activation following ischemic injury is a major cause of neuronal loss in stroke.
The reduction in caspase-3 activity was accompanied by better preserved behavioral performance in the treated animals. They showed less cognitive impairment and better motor function following the ischemic event compared to untreated controls. This suggests that Pinealon's neuroprotective effect translates into functional preservation, not just biomarker improvement.
Serotonin Expression Regulation
Khavinson, Lin'kova, Tarnovskaya, and colleagues (2014) demonstrated that short peptides, including Pinealon, can stimulate serotonin expression in brain cortex cells. Serotonin is a key neurotransmitter involved in mood regulation, cognitive function, sleep, and appetite. Age-related decline in serotonin production contributes to depression, anxiety, cognitive decline, and sleep disturbances in elderly populations.
The ability to restore serotonin expression at the gene level, rather than simply inhibiting its reuptake (as SSRIs do), represents a more fundamental approach to normalizing serotonergic function. It addresses the root cause of age-related serotonin decline rather than compensating for it pharmacologically.
Alzheimer's Disease Mechanisms
Recent work published by Linkova, Khavinson, Diatlova, and Nichik (2021) explored the mechanisms by which Pinealon (EDR peptide) may be relevant to Alzheimer's disease pathogenesis. The study found that EDR peptide influences the expression of genes involved in amyloid precursor protein processing, tau protein phosphorylation, and neuroinflammatory signaling.
Additionally, a 2022 study in the International Journal of Molecular Sciences examined neuroepigenetic mechanisms of ultrashort peptides in Alzheimer's disease, demonstrating that these peptides can modulate epigenetic markers (DNA methylation, histone modifications) at genes critical to Alzheimer's pathology. While this doesn't prove that Pinealon can prevent or treat Alzheimer's, it establishes a plausible molecular connection between bioregulator peptides and the fundamental mechanisms driving the disease.
Cortexin vs. Cortagen: Choosing the Right Brain Bioregulator
Clinicians working with brain bioregulators often need to choose between Cortexin (the complex extract) and Cortagen (the synthetic tetrapeptide). Each has distinct advantages.
Cortexin advantages: Broader spectrum of activity due to multiple peptide components. More extensive clinical data, particularly in stroke recovery and traumatic brain injury. Available as an approved pharmaceutical in Russia with standardized manufacturing and quality control. The complex composition may provide effects that no single peptide can replicate.
Cortagen advantages: Defined composition allows for precise mechanistic studies. More consistent batch-to-batch quality. Easier to synthesize and potentially more accessible through research peptide suppliers. Clearer regulatory pathway as a defined chemical entity rather than a biological extract.
Pinealon advantages: Smallest of the brain bioregulators (just 3 amino acids), offering excellent bioavailability. Dual activity addressing both neuroprotection and circadian rhythm regulation. Available from research peptide suppliers. The circadian regulation component is unique among brain bioregulators.
In practice, the choice often depends on the primary clinical goal. For acute neuroprotection (stroke, TBI), Cortexin has the most clinical support. For long-term brain aging prevention and circadian optimization, Pinealon may be the best choice. Cortagen occupies a middle ground as a defined synthetic alternative to Cortexin.
Cognitive Enhancement Stack: Combining Brain Peptides
Many practitioners combine brain bioregulators with other neuroprotective peptides for comprehensive cognitive support. A typical advanced brain health stack might include:
- Pinealon for circadian regulation and neuroprotection through gene expression modulation
- Semax for BDNF upregulation, cognitive enhancement, and acute neuroprotection
- Selank for anxiolytic effects, mood stabilization, and immunomodulation in the CNS
- GHK-Cu for neurovascular support and anti-inflammatory gene regulation
- NAD+ for neuronal energy metabolism and sirtuin-mediated neuroprotection
The rationale for combining these compounds is that they address different aspects of brain aging through different mechanisms. Pinealon works through epigenetic gene regulation. Semax works through neurotrophin signaling. Selank modulates GABA and serotonin systems. GHK-Cu provides broad anti-inflammatory gene regulation. NAD+ supports cellular energy metabolism. Together, they create a multi-layered defense against brain aging that exceeds what any single compound can provide.
For personalized guidance on building a cognitive enhancement protocol, explore the FormBlends Free Assessment.
Tissue-Specific Bioregulators: Vascular, Cartilage, and Liver Support
Vesugen (Lys-Glu-Asp): Vascular Bioregulation
Why Vascular Aging Matters for Everything
Vascular aging underpins the deterioration of virtually every organ system. The blood supply delivers oxygen, nutrients, hormones, and immune cells to every tissue, and removes waste products and metabolic byproducts. When blood vessels age, every downstream tissue suffers.
Age-related vascular changes include arterial stiffening (arteriosclerosis), endothelial dysfunction (impaired ability of blood vessel linings to regulate blood flow and inflammation), reduced nitric oxide production (leading to impaired vasodilation), increased vascular inflammation, and microvascular rarefaction (loss of small blood vessels in tissue beds). These changes contribute to hypertension, atherosclerosis, coronary artery disease, stroke, kidney disease, cognitive decline, and even skin aging.
Vesugen was developed specifically to address vascular aging at the gene expression level. As a synthetic tripeptide with the sequence Lys-Glu-Asp, it targets endothelial cells and vascular smooth muscle cells, aiming to restore youthful patterns of gene expression in blood vessel tissue.
Endothelial Function Restoration
The endothelium, the single-cell layer lining all blood vessels, is far more than a passive barrier. It's an active endocrine organ that produces signaling molecules controlling blood flow, inflammation, coagulation, and vascular remodeling. Healthy endothelial cells produce nitric oxide (NO), which relaxes blood vessels, inhibits platelet aggregation, and suppresses inflammatory cell adhesion. Aged endothelial cells produce less NO and more pro-inflammatory molecules, shifting the vascular environment toward dysfunction.
Research on Vesugen has shown restoration of nitric oxide synthase expression in aged endothelial cells, with corresponding improvements in NO production and vasodilatory function. This effect appears to be mediated through gene expression changes consistent with the bioregulation model - Vesugen doesn't simply provide NO from an external source but rather restores the cell's ability to produce it normally.
Anti-Inflammatory Vascular Effects
Vascular inflammation is a primary driver of atherosclerosis, the buildup of fatty plaques in arterial walls that causes heart attacks and strokes. Inflammatory molecules produced by dysfunctional endothelial cells recruit immune cells to the vessel wall, where they consume oxidized lipids and form foam cells - the building blocks of atherosclerotic plaques.
Vesugen has been shown to reduce the expression of adhesion molecules (ICAM-1, VCAM-1) on endothelial cell surfaces. These adhesion molecules are the "landing pads" that allow immune cells to attach to and invade the vessel wall. By reducing their expression, Vesugen may slow the inflammatory process that drives plaque formation.
Microcirculation Enhancement
Perhaps the most clinically relevant vascular effect is improved microcirculation. The smallest blood vessels - capillaries and arterioles - are where oxygen and nutrient exchange actually occurs. Age-related loss of these microvessels reduces tissue perfusion and contributes to organ dysfunction. Vesugen has shown ability to improve microcirculatory parameters in multiple tissue beds, potentially enhancing organ function broadly.
This microcirculatory enhancement also has implications for the effectiveness of other bioregulators. If blood flow to the thymus, brain, or liver is improved, the delivery of organ-specific bioregulators to those tissues is also enhanced. Vesugen may therefore act as a "facilitator" for the entire bioregulator protocol by ensuring adequate tissue perfusion.
Cartalax (Ala-Glu-Asp): Cartilage Bioregulation
The Cartilage Regeneration Challenge
Articular cartilage - the smooth, glassy tissue that covers joint surfaces - is one of the most challenging tissues in the body to repair. It lacks its own blood supply, relying entirely on diffusion from the synovial fluid for nutrient delivery and waste removal. It has limited capacity for self-repair because chondrocytes (cartilage cells) have very low turnover rates in adult tissue. And it exists in a mechanically demanding environment, bearing compressive forces of several times body weight with every step.
As cartilage ages, the balance between cartilage matrix synthesis and degradation shifts toward net loss. Chondrocytes produce less collagen type II and proteoglycans (the structural components of cartilage matrix) while producing more matrix metalloproteinases (MMPs, the enzymes that break down cartilage). This imbalance results in progressive cartilage thinning, which eventually leads to osteoarthritis - the most common joint disease in the world.
Cartalax Mechanism of Action
Cartalax addresses cartilage aging by modulating gene expression in chondrocytes, shifting the balance back toward matrix synthesis and away from matrix degradation. Specific effects include:
- Collagen type II upregulation: Cartalax increases the expression of COL2A1, the gene encoding collagen type II, the primary structural protein of articular cartilage. Enhanced collagen production supports cartilage matrix maintenance and repair.
- Proteoglycan synthesis: Aggrecan and other proteoglycans provide cartilage with its compressive resistance and water-retaining capacity. Cartalax has shown ability to increase proteoglycan synthesis in chondrocyte cultures.
- MMP inhibition: Matrix metalloproteinases, particularly MMP-3 and MMP-13, are the primary enzymes responsible for cartilage matrix degradation. Cartalax reduces their expression, slowing the destructive side of the remodeling equation.
- Anti-inflammatory effects: Cartilage inflammation (driven by cytokines like IL-1 beta and TNF-alpha) is a major driver of osteoarthritis progression. Cartalax reduces inflammatory signaling within joint tissue.
- Chondrocyte proliferation: In aged cartilage, chondrocyte numbers decline. Cartalax stimulates controlled chondrocyte proliferation, increasing the cellular workforce available for matrix maintenance.
For individuals seeking complementary joint and connective tissue support, BPC-157 offers well-documented healing effects on tendons, ligaments, and other connective tissues through gastric pentadecapeptide-mediated mechanisms. The combination of Cartalax (gene expression restoration in cartilage) with BPC-157 (growth factor modulation and angiogenesis in connective tissue) addresses joint health from two complementary angles.
Livagen (Lys-Glu-Asp-Trp): Liver Bioregulation
Liver Aging: The Silent Decline
The liver is the body's primary metabolic organ, responsible for detoxification, protein synthesis, bile production, nutrient metabolism, immune function, and storage of vitamins and minerals. It's also one of the most regenerative organs in the body, capable of recovering from substantial damage. However, this regenerative capacity declines with age.
Age-related liver changes include reduced hepatocyte (liver cell) number and volume, decreased blood flow (hepatic perfusion falls by 30% to 40% between ages 25 and 65), impaired detoxification enzyme activity, reduced albumin synthesis, increased susceptibility to fibrosis, and decreased regenerative capacity after injury. These changes make elderly individuals more vulnerable to drug toxicity, more susceptible to liver damage from environmental toxins, and slower to recover from hepatic injury.
Livagen's Chromatin Effects
Livagen has been the subject of some of the most detailed mechanistic studies among the Khavinson bioregulators, particularly regarding its effects on chromatin structure. Studies using fluorescence microscopy and chromatin accessibility assays have demonstrated that Livagen treatment shifts the heterochromatin/euchromatin balance in hepatocytes toward a more open, transcriptionally active state.
In aged hepatocytes, large portions of the genome that are active in younger cells become silenced through chromatin condensation. This silencing reduces the expression of genes involved in detoxification, protein synthesis, and regeneration. Livagen, by reversing this chromatin condensation at specific loci, re-activates these silenced genes and restores hepatocyte function.
The chromatin effects of Livagen have provided some of the strongest direct evidence for the bioregulation mechanism overall. Using fluorescently labeled Livagen molecules, researchers have observed their nuclear accumulation and co-localization with specific chromatin regions, providing visual confirmation that the peptide reaches its proposed target and interacts with it in the predicted manner.
Functional Restoration Effects
Functional studies of Livagen-treated hepatocytes and liver tissue have shown:
- Albumin synthesis restoration: Albumin, produced exclusively by the liver, is a key marker of hepatic synthetic function. Livagen treatment increases albumin production in aged hepatocytes.
- Detoxification enzyme upregulation: Cytochrome P450 enzymes and other detoxification pathways show increased activity following Livagen treatment, potentially improving the liver's ability to process medications and environmental toxins.
- Regenerative capacity enhancement: In models of partial hepatectomy (surgical removal of liver tissue), Livagen-treated animals showed faster and more complete liver regeneration compared to untreated controls.
- Anti-fibrotic effects: Preliminary data suggests Livagen may reduce hepatic stellate cell activation, the process that drives liver fibrosis. By maintaining hepatocytes in a healthy, functional state, Livagen may reduce the signals that trigger fibrotic remodeling.
The lifespan data for Livagen specifically is also noteworthy. Anisimov, Khavinson, and Mikhailova (2001) studied the effect of Livagen on lifespan and spontaneous tumor incidence in mice. Livagen treatment produced measurable lifespan extension and reduced tumor incidence, consistent with the broader bioregulator class effects.
Figure 9: Tissue-specific mechanisms of Vesugen (vascular), Cartalax (cartilage), and Livagen (liver)
Practical Longevity Protocols with Bioregulators
Building a Protocol: Principles and Priorities
Designing a bioregulator-based longevity protocol requires balancing several considerations: which organ systems most need support, how many peptides can be practically managed, budget constraints, and the individual's health status and goals. This section provides frameworks for different approaches, from minimal starter protocols to comprehensive multi-organ programs.
Protocol Tier 1: Essential Foundation (Two Peptides)
The simplest and best-supported protocol targets the two organ systems whose decline drives the broadest range of age-related dysfunction: the pineal gland and the thymus. This combination has the strongest clinical evidence, including the 14-20 year follow-up data showing up to 4.1-fold mortality reduction.
Components
- Epithalon (pineal bioregulator): 5-10 mg subcutaneous daily for 10-20 days
- Thymalin (thymic bioregulator): 10-20 mg intramuscular daily for 5-10 days
Schedule
- Course 1: Spring (March-April)
- Course 2: Autumn (September-October)
- Both peptides can be administered concurrently during each course
- Continue annually for maximum benefit
Monitoring
- Baseline: CBC with differential, comprehensive metabolic panel, hormonal panel (cortisol, DHEA-S, thyroid, melatonin sulfate in urine), inflammatory markers (hsCRP, IL-6)
- Follow-up: Repeat at 3 months post-course, then annually
- Optional advanced: Telomere length testing, biological age testing (DNA methylation clocks)
Protocol Tier 2: Extended Support (Four to Six Peptides)
For individuals who want broader organ support or who have specific areas of concern beyond immune and pineal function, the protocol can be extended to include additional bioregulators.
Components (in addition to Tier 1)
- Pinealon (brain bioregulator): For cognitive support and circadian optimization
- Vesugen (vascular bioregulator): For cardiovascular support and improved tissue perfusion
- Cartalax (cartilage bioregulator): For joint health and cartilage maintenance
- Livagen (liver bioregulator): For hepatic function and detoxification support
Phased Schedule
- Phase 1 (Days 1-10): Epithalon + Thymalin (the foundation)
- Phase 2 (Days 14-24): Pinealon + Vesugen (brain and vascular support)
- Phase 3 (Days 28-38): Cartalax + Livagen (structural and metabolic support)
- Break: 4-5 months
- Repeat: Full cycle twice per year
The phased approach spaces out the peptide courses to avoid overwhelming the system with too many concurrent epigenetic signals. Each phase allows the initiated gene expression changes to establish before introducing the next set of bioregulators.
Protocol Tier 3: Comprehensive Anti-Aging (Bioregulators + Complementary Peptides)
The most comprehensive approach integrates Khavinson bioregulators with other well-studied peptides to create a multi-layered anti-aging protocol addressing aging through multiple mechanisms simultaneously.
Bioregulator Foundation
- Epithalon + Thymalin (annual courses, as above)
- Organ-specific bioregulators as needed (Pinealon, Vesugen, Cartalax, Livagen)
Complementary Peptides (used between bioregulator courses)
- BPC-157: Tissue repair, gut health, systemic healing - can be used continuously or in courses as needed
- GHK-Cu: Skin and tissue remodeling, broad anti-inflammatory gene regulation - topical and/or injectable
- MOTS-c: Mitochondrial function, metabolic health, exercise mimetic - injectable courses
- NAD+: Cellular energy, sirtuin activation, DNA repair support - various administration options
- Semax / Selank: Cognitive enhancement and mood support - nasal spray protocols
Rationale for Integration
Each component addresses a different hallmark of aging:
| Aging Hallmark | Component | Mechanism |
|---|---|---|
| Epigenetic alterations | Khavinson bioregulators | Gene expression restoration via DNA interaction |
| Telomere attrition | Epithalon | Telomerase activation |
| Immune dysfunction | Thymalin, Vilon, Thymosin Alpha-1 | Thymic restoration and immune modulation |
| Mitochondrial dysfunction | MOTS-c, NAD+ | Mitochondrial biogenesis and energy metabolism |
| Cellular senescence | Epithalon (indirect), NAD+ (sirtuin activation) | Reduced senescent cell accumulation |
| Stem cell exhaustion | BPC-157, GHK-Cu | Growth factor modulation and tissue repair signaling |
| Intercellular communication | Multiple bioregulators | Restored hormonal and signaling molecule production |
| Nutrient sensing | MOTS-c | AMPK activation, metabolic optimization |
By addressing multiple hallmarks simultaneously, this integrated approach creates a more comprehensive defense against aging than any single compound can provide. The key insight from Khavinson's work - that short courses of precisely targeted peptides can produce lasting biological changes - makes this multi-peptide approach practical, since each compound can be administered in its own optimized course rather than requiring continuous daily use of multiple agents.
Age-Specific Recommendations
Khavinson's recommendations varied by age group, reflecting the progressive nature of aging and the increasing need for bioregulatory support as decline advances:
| Age Range | Recommended Approach | Focus Areas |
|---|---|---|
| 30-39 | Optional prevention for those with accelerated aging markers | Epithalon alone, annual course; baseline biomarker testing |
| 40-49 | Tier 1 protocol (Epithalon + Thymalin) | Pineal + immune foundation; begin regular monitoring |
| 50-59 | Tier 2 protocol (4-6 bioregulators) | Add organ-specific support based on individual needs |
| 60-69 | Tier 2 or Tier 3 protocol | Comprehensive support; more frequent monitoring |
| 70+ | Tier 3 protocol with medical supervision | Maximum support; careful monitoring; dose adjustment as needed |
These are general guidelines, not rigid prescriptions. Individual variation in aging rate, health status, and specific organ vulnerability should guide protocol design. Advanced biological age testing using DNA methylation clocks can help identify individuals who are aging faster than their chronological age would suggest and who might benefit from earlier or more aggressive intervention.
For personalized recommendations based on your age, health status, and goals, take the FormBlends Free Assessment. You can also use the Dosing Calculator to plan your specific protocol.
Monitoring and Optimization
Effective bioregulator use requires monitoring to assess response and guide protocol adjustments. Key monitoring strategies include:
Essential Biomarkers
- Complete blood count with differential: Tracks immune cell populations, particularly lymphocyte subsets
- Comprehensive metabolic panel: Monitors liver and kidney function, electrolytes, and glucose metabolism
- High-sensitivity C-reactive protein (hsCRP): Measures systemic inflammation, one of the primary targets of bioregulator therapy
- Hormonal panel: Cortisol, DHEA-S, testosterone/estradiol, thyroid function (TSH, free T3/T4), and IGF-1 assess endocrine restoration
Advanced Biomarkers
- Telomere length: Directly measures one of Epithalon's primary targets. Can be tracked annually to assess telomere dynamics.
- Biological age testing: DNA methylation clocks (Horvath, GrimAge, DunedinPACE) provide the most comprehensive assessment of biological aging rate and can demonstrate whether a bioregulator protocol is actually slowing or reversing epigenetic aging
- NK cell function: Functional assays of natural killer cell cytotoxicity directly measure immune competence
- Urinary 6-sulfatoxymelatonin: Measures melatonin production, directly relevant to Epithalon's pineal effects
- Cytokine profiles: IL-6, TNF-alpha, IL-10 ratios track the inflammatory/anti-inflammatory balance
Functional Assessments
- Cognitive testing (MoCA, CANTAB, or similar standardized batteries) for brain bioregulator response
- Grip strength and physical performance testing for overall functional status
- Sleep quality assessment (Pittsburgh Sleep Quality Index, actigraphy) for pineal bioregulator response
- Subjective wellbeing and quality of life questionnaires
Regulatory Status and Global Accessibility
Russia and the Former Soviet Union
In Russia, six Khavinson peptide preparations have achieved full pharmaceutical approval: Thymalin, Cortexin, Epithalamin, Prostatilen, Retinalamin, and Thymogen. These are manufactured to pharmaceutical-grade standards, prescribed by physicians, and used in hospital and clinical settings. They have accumulated decades of post-marketing surveillance data, providing a real-world safety record that supplements the clinical trial data.
In addition to the pharmaceutical preparations, 64 peptide-based dietary supplements (marketed under the Cytomax brand among others) are available without prescription in Russia. These oral formulations contain bioregulator peptides in capsule form and are widely used for health maintenance and anti-aging purposes.
The Russian regulatory framework has been more receptive to bioregulator peptides than Western regulatory agencies for several reasons. The long history of peptide research in Russian biomedical science created institutional familiarity with the class. The regulatory pathway in Russia places greater weight on long-term observational data and clinical experience, which is the type of evidence Khavinson's program generates most abundantly. And the strong government support for Khavinson's research, given its origins in military applications, provided resources and institutional backing that facilitated regulatory approval.
European Union
Within the EU, Khavinson bioregulators are generally not approved as pharmaceutical products but are available in some markets as dietary supplements or food supplements. The regulatory status varies by member state. Some countries classify them as novel foods requiring pre-market authorization, while others permit their sale as dietary supplements under existing frameworks.
The EU's regulatory approach to peptide supplements is evolving. As interest in peptide-based health products grows, regulatory frameworks are being adapted to address this emerging product category. Practitioners in EU countries who wish to use bioregulators typically do so under regulatory provisions for magistral preparations (custom-compounded formulations prescribed by physicians) or through research use exemptions.
United States
In the United States, no Khavinson bioregulator has undergone the FDA approval process. The synthetic peptides (Epithalon, Pinealon, Vilon, Vesugen, Cartalax, Livagen, Cortagen) are available from research chemical suppliers for in vitro research purposes. Some are also available through compounding pharmacies with a physician's prescription, depending on state regulations.
The regulatory landscape for peptides in the US has been in flux. The FDA's increasing scrutiny of peptide products has led some to be removed from compounding pharmacy catalogs, while others remain available. Khavinson bioregulators have generally attracted less regulatory attention than more commonly used peptides (such as BPC-157 or certain growth hormone-releasing peptides), possibly because they are less widely used in the US market.
For US-based individuals interested in bioregulator peptides, working with a knowledgeable healthcare provider who understands the regulatory framework and can guide appropriate sourcing is essential. The FormBlends Biohacking Hub provides additional guidance on accessing peptide therapies within legal frameworks.
Quality and Sourcing Considerations
Regardless of jurisdiction, the quality of peptide products varies enormously between suppliers. Key quality considerations include:
- Purity: Research-grade peptides should be at least 98% pure, verified by HPLC analysis. Certificates of analysis from third-party testing labs provide the strongest assurance.
- Identity confirmation: Mass spectrometry data confirming the correct molecular weight and sequence should be available for any synthetic peptide product.
- Sterility: Injectable preparations must be sterile. Lyophilized (freeze-dried) peptides require sterile reconstitution by the end user.
- Storage stability: Peptides degrade over time, especially at room temperature. Proper cold-chain shipping and storage recommendations should be followed.
- Supplier reputation: Established suppliers with published quality control data, responsive customer service, and long operating histories offer greater reliability than fly-by-night operators.
For the natural extract preparations (Thymalin, Cortexin, Epithalamin, Prostatilen, Retinalamin, Thymogen), quality considerations are even more critical. These complex biological products require specialized manufacturing and quality control procedures to ensure batch-to-batch consistency and safety. Russian pharmaceutical-grade products from licensed manufacturers offer the highest quality assurance for these preparations.
Historical Context: Four Decades of Peptide Bioregulation Research
The Soviet Military Origins (1970s)
The story of peptide bioregulators begins in a classified Soviet military research program focused on protecting personnel from extreme physiological stress. During the 1970s, the Soviet military was deeply concerned about the effects of radiation exposure on soldiers and cosmonauts, laser-induced retinal damage in combat scenarios, and the immune suppression that accompanied prolonged high-stress deployments.
Vladimir Khavinson, then a young military physician, was assigned to a research team investigating biological approaches to these problems. The team's hypothesis was based on an older tradition in Russian medicine: the idea that tissue extracts from healthy organs could support the function of corresponding organs in sick or stressed individuals. This concept, called organotherapy, dates back to the 19th century but had largely been abandoned in Western medicine by the mid-20th century.
Khavinson's team took a more systematic and scientifically rigorous approach than earlier organotherapy practitioners. They developed standardized extraction protocols to isolate peptide complexes from specific organs of young, healthy bovine donors. Each extract was characterized biochemically, tested for biological activity in cell culture and animal models, and evaluated for safety before any clinical application.
The initial results exceeded expectations. Thymic extracts restored immune function in radiation-exposed animals more effectively than any other intervention tested. Pineal extracts normalized circadian rhythms and hormonal function in animals subjected to chronic stress. Retinal extracts protected against laser-induced retinal damage. And crucially, the effects persisted long after the treatment was discontinued, suggesting a regulatory mechanism rather than a simple pharmacological one.
The Institute Years (1980s-1990s)
The success of the military research program led to the establishment of the Saint Petersburg Institute of Bioregulation and Gerontology in 1992, with Khavinson as its founding director. The institute's mission was to translate the military findings into civilian medical applications and to develop a comprehensive scientific framework for understanding peptide bioregulation.
During this period, several major milestones were achieved:
- 1982-1985: Thymalin received pharmaceutical approval in Russia, becoming the first Khavinson bioregulator to enter clinical practice. It was initially used for immunocorrection in post-surgical and post-infection recovery.
- 1985-1990: Cortexin was approved for clinical use in neurological conditions including stroke recovery and traumatic brain injury. Prostatilen and Retinalamin were also approved during this period.
- 1985-2003: The landmark 266-patient longevity study was initiated, eventually providing the strongest clinical evidence for bioregulator-mediated life extension in humans.
- 1990s: The synthetic peptide program began in earnest. Khavinson's team systematically identified the minimal active sequences from each tissue extract and synthesized them as pure compounds. Epithalon, Vilon, Vesugen, Cartalax, Livagen, and others were developed during this decade.
The Molecular Biology Era (2000s-2010s)
As molecular biology tools advanced, Khavinson's group increasingly focused on elucidating the mechanisms underlying bioregulator activity. This period produced the foundational studies on peptide-DNA interaction, chromatin remodeling, and epigenetic regulation that form the theoretical basis of the bioregulation model.
Key publications from this era include the fluorescence microscopy studies showing peptide nuclear penetration and DNA binding (Fedoreyeva et al., 2011), the chromatin remodeling studies in human cells (Khavinson, Lezhava, and Malinin, 2004), and the systematic review of peptide regulation of gene expression (Khavinson et al., 2021). These studies elevated the field from empirical observation of clinical effects to mechanistic understanding of molecular processes.
This period also saw the publication of the long-term follow-up data from the 1985-2003 longevity study, with the striking mortality reduction results that drew international attention. The data was published in Neuroendocrinology Letters and subsequently presented at international conferences on gerontology and anti-aging medicine.
Global Recognition and Independent Validation (2020s)
The 2020s have seen growing international interest in Khavinson's work. Several factors have contributed to this increased attention:
- The longevity research boom: Massive investment in anti-aging research from Silicon Valley tech entrepreneurs and pharmaceutical companies has created interest in any approach with substantial life-extension data. Khavinson's 20-40% lifespan extensions in animals and striking human mortality data naturally attracted attention.
- Peptide therapy mainstreaming: The broader acceptance of peptide therapeutics in Western medicine - with FDA-approved peptide drugs now numbering in the dozens - has made the peptide bioregulator concept more palatable to mainstream medical audiences.
- Epigenetics revolution: The explosion of epigenetics research, including the development of epigenetic clocks and growing understanding of age-related epigenetic changes, has provided a framework for understanding how short peptides might regulate aging at the molecular level.
- Independent replication: The 2025 Biogerontology study confirming Epithalon's telomere-extending activity by an independent research group provided the first major independent validation of a core Khavinson mechanism.
Khavinson's Legacy in Numbers
| Metric | Number | Significance |
|---|---|---|
| Years of research | 40+ | One of the longest-running anti-aging research programs in history |
| Scientific publications | 775+ | Extensive documentation of findings across multiple journals |
| Patents (Russian and international) | 196 | Broad intellectual property portfolio covering peptide sequences and applications |
| Pharmaceutical preparations approved | 6 | Thymalin, Cortexin, Epithalamin, Prostatilen, Retinalamin, Thymogen |
| Dietary supplement formulations | 64 | Wide range of oral bioregulator products (Cytomax and others) |
| Individual bioregulator peptides characterized | 20+ | Each targeting a specific organ or tissue system |
| Clinical trial participants (total) | 15,000+ | Across all peptide preparations and conditions studied |
| Maximum lifespan extension (animals) | 40% | With combined bioregulator treatment in rodent models |
| Maximum mortality reduction (humans) | 4.1-fold | With combined annual Thymalin + Epithalamin, 6-year follow-up |
Bioregulators vs. Other Anti-Aging Approaches: A Comparative Analysis
The Hallmarks of Aging Framework
To understand where bioregulators fit in the anti-aging landscape, it's useful to consider them through the lens of the hallmarks of aging framework, originally proposed by Lopez-Otin et al. in 2013 and updated in 2023. This framework identifies twelve fundamental biological processes that drive aging. Different anti-aging interventions target different hallmarks, and no single approach addresses them all.
Khavinson bioregulators primarily target genomic instability (through improved DNA repair), telomere attrition (through Epithalon's telomerase activation), epigenetic alterations (through direct gene expression regulation), loss of proteostasis (through restored protein synthesis), deregulated nutrient sensing (indirectly through endocrine restoration), altered intercellular communication (through restored hormonal signaling), and stem cell exhaustion (indirectly through tissue-specific gene activation).
This is a broader coverage than most single anti-aging interventions achieve. Rapamycin primarily targets nutrient sensing (mTOR pathway). Metformin primarily targets nutrient sensing (AMPK pathway) and mitochondrial function. Senolytics primarily target cellular senescence. NAD+ precursors primarily target mitochondrial dysfunction. By addressing seven of twelve aging hallmarks, bioregulators offer unusually comprehensive coverage.
Rapamycin Comparison
Rapamycin (and its analogs, rapalogs) is perhaps the most thoroughly validated life-extension compound in animal studies, with consistent lifespan extensions of 10% to 25% across multiple strains of mice. It works by inhibiting the mTOR (mechanistic target of rapamycin) signaling pathway, which coordinates cell growth and metabolism in response to nutrient availability.
Compared to Khavinson bioregulators, rapamycin has stronger independent replication (multiple labs worldwide have confirmed its effects) but a less favorable side effect profile. Chronic rapamycin use can cause insulin resistance, dyslipidemia, immunosuppression, and mouth ulcers. It requires continuous daily dosing to maintain effects.
Bioregulators, by contrast, use short courses with long breaks and have minimal reported side effects. However, their evidence base relies more heavily on a single research group. The lifespan extension data is comparable (20-40% for bioregulators vs. 10-25% for rapamycin in rodents), though direct head-to-head comparisons have not been conducted.
The two approaches are mechanistically complementary. Rapamycin reduces anabolic signaling (telling cells to grow less), while bioregulators restore gene expression programs (telling cells to function more youthfully). Some advanced longevity protocols incorporate both approaches, using rapamycin for mTOR modulation and bioregulators for tissue-specific gene restoration.
Metformin Comparison
Metformin, a diabetes drug with decades of safety data, has shown modest anti-aging effects in observational studies and is the subject of the TAME (Targeting Aging with Metformin) trial, the first FDA-approved clinical trial specifically testing an anti-aging intervention in humans. Metformin works primarily through AMPK activation and mitochondrial complex I inhibition.
Compared to bioregulators, metformin has a more established Western regulatory profile and extensive safety data from millions of patient-years of use. However, its anti-aging effects in non-diabetic populations are modest and debated. Its mechanism (metabolic modulation) is fundamentally different from bioregulators' mechanism (gene expression restoration), making the two approaches complementary.
Senolytic Drugs Comparison
Senolytics (dasatinib + quercetin, navitoclax, fisetin) selectively kill senescent cells, reducing the burden of dysfunctional cells that produce pro-inflammatory molecules. Early clinical trials have shown promising results in conditions like idiopathic pulmonary fibrosis and diabetic kidney disease.
Bioregulators may reduce senescent cell formation at the source by maintaining healthier gene expression and extending replicative capacity (through telomerase activation). Senolytics remove senescent cells after they've formed. The combination, using bioregulators to prevent new senescent cell formation while using senolytics to clear existing ones, could theoretically be more effective than either approach alone.
NAD+ Precursors Comparison
NAD+ precursors (nicotinamide riboside, nicotinamide mononucleotide) aim to restore NAD+ levels that decline with age. NAD+ is essential for cellular energy production, DNA repair, and sirtuin activation. Human clinical trials have confirmed that NAD+ precursors can raise blood NAD+ levels, but clinical outcomes data is still accumulating.
NAD+ precursors and bioregulators address different aspects of cellular aging. NAD+ provides the metabolic fuel cells need to execute their functions, while bioregulators ensure the genetic programs directing those functions are optimally expressed. Together, they address both the "what to do" (gene expression) and the "energy to do it" (NAD+ availability) aspects of cellular function. For more on NAD+ in anti-aging protocols, see the FormBlends NAD+ resource.
Comprehensive Comparison Table
| Approach | Mechanism | Animal Lifespan Data | Human Evidence | Side Effects | Dosing Model |
|---|---|---|---|---|---|
| Khavinson Bioregulators | Epigenetic gene regulation | 20-40% extension | 4.1x mortality reduction (observational) | Minimal | Short courses, long breaks |
| Rapamycin | mTOR inhibition | 10-25% extension | Limited clinical data for aging | Moderate (metabolic, immune) | Continuous or intermittent daily |
| Metformin | AMPK activation | 5-15% extension (strain-dependent) | TAME trial ongoing | GI side effects, B12 depletion | Continuous daily |
| Senolytics | Senescent cell clearance | Variable, up to 36% | Early clinical trials positive | Moderate (cytopenia risk) | Intermittent courses |
| NAD+ Precursors | NAD+ restoration | Limited lifespan data | NAD+ levels increase; clinical outcomes pending | Minimal | Continuous daily |
| Caloric Restriction | Multiple pathways | 30-50% extension | Strong observational, no RCT | Sustainability concerns | Continuous dietary modification |
The Integration Principle
The most sophisticated longevity protocols don't rely on any single approach but combine multiple interventions that target different aging hallmarks through different mechanisms. Khavinson bioregulators fit particularly well into integrated protocols because their short-course dosing model leaves ample time for other interventions. Their epigenetic mechanism addresses aging at a fundamental level that complements the receptor-based, metabolic, and senolytic approaches used by other anti-aging compounds.
Emerging Research Directions and Future Applications
Personalized Bioregulation Through Epigenetic Profiling
One of the most exciting potential applications of bioregulator peptides lies in personalization through epigenetic profiling. DNA methylation clocks, developed by Steve Horvath and others, can now estimate biological age with remarkable precision and identify specific epigenetic patterns associated with accelerated aging in particular organ systems.
Imagine a future where a simple blood test reveals that your liver is aging 5 years faster than the rest of your body, while your immune system shows accelerated decline. A personalized bioregulator protocol could then prioritize Livagen for liver support and Thymalin for immune restoration, targeting the specific organs showing the greatest deviation from optimal aging.
This precision medicine approach to bioregulation is not yet standard practice, but the tools to implement it are rapidly becoming available. Companies like TruDiagnostic and Elysium Health already offer consumer-grade epigenetic age testing, and research groups are developing organ-specific biological age markers that could guide tissue-targeted interventions.
Bioregulators for Neurodegenerative Disease Prevention
The emerging data on Pinealon's (EDR peptide) effects on Alzheimer's disease-related gene expression opens an intriguing avenue for neurodegenerative disease prevention. While no bioregulator has been tested in a clinical trial specifically designed to assess dementia prevention, several lines of evidence suggest this could be a promising application:
- Pinealon modulates expression of genes involved in amyloid precursor protein processing and tau phosphorylation
- Epithalon's restoration of circadian function may support the brain's glymphatic clearance of amyloid-beta during sleep
- The anti-inflammatory effects of multiple bioregulators could reduce neuroinflammation, a key driver of neurodegeneration
- Vesugen's vascular effects could maintain cerebral blood flow, supporting brain health through improved perfusion
Clinical trials testing bioregulators for dementia prevention would require decades of follow-up and thousands of participants, making them logistically challenging. However, biomarker-driven studies measuring changes in Alzheimer's-related blood markers (like phosphorylated tau and amyloid-beta ratios) following bioregulator treatment could provide early signals of efficacy within more practical timeframes.
Synthetic Biology and Designer Bioregulators
Advances in computational biology and machine learning are opening the possibility of designing novel bioregulator peptides optimized for specific gene expression targets. Rather than relying on sequences isolated from natural tissue extracts, researchers could potentially design custom peptide sequences with enhanced binding affinity for specific DNA targets, improved cellular penetration properties, greater metabolic stability, and novel tissue specificities not found in natural peptides.
This "designer bioregulator" concept extends Khavinson's work from a discovery-based approach (finding active sequences in nature) to a design-based approach (creating optimal sequences computationally). Early work in this area has used molecular dynamics simulations to model peptide-DNA interactions and predict binding affinities, but practical application remains several years away.
Combination with Gene Therapy and CRISPR
A more speculative but fascinating direction involves combining bioregulator peptides with gene editing technologies. CRISPR-based epigenetic editors can now modify gene expression without changing the underlying DNA sequence, using catalytically dead Cas9 proteins fused to epigenetic modifiers. This approach shares the same fundamental goal as bioregulator peptides: modulating gene expression to restore youthful function.
Bioregulator peptides could potentially serve as "guides" for where epigenetic modifications should be targeted, based on decades of data about which gene expression changes they produce. The tissue-specific knowledge accumulated through Khavinson's research could inform the design of gene therapy vectors targeted to the same genes that bioregulator peptides naturally regulate.
Microbiome and Bioregulator Interactions
An unexplored area of research involves the interaction between bioregulator peptides and the gut microbiome. Oral bioregulator preparations pass through the gastrointestinal tract, where they encounter the trillions of bacteria composing the gut microbiome. Several questions arise:
- Do gut bacteria metabolize bioregulator peptides, potentially altering their activity or producing active metabolites?
- Can bioregulator peptides influence microbiome composition through effects on gut epithelial gene expression?
- Does the microbiome affect the bioavailability of oral bioregulator preparations?
- Could microbiome profiling help predict individual responses to bioregulator therapy?
These questions are entirely unanswered but represent important gaps in our understanding of how oral bioregulators work and how their effects might vary between individuals.
Exercise, Nutrition, and Bioregulator Integration
Lifestyle factors including exercise and nutrition profoundly affect gene expression and epigenetic patterns - the same pathways that bioregulator peptides target. This raises the question of whether bioregulators work differently (better or worse) in the context of optimized lifestyle practices.
Exercise, for example, activates many of the same gene expression pathways that bioregulators target, including antioxidant defense genes, mitochondrial biogenesis genes, and anti-inflammatory pathways. Does exercise enhance the effects of bioregulators by priming the same epigenetic pathways? Or does it reduce the marginal benefit by already activating those pathways independently?
Similarly, caloric restriction and intermittent fasting produce epigenetic changes that overlap with some bioregulator effects. Understanding these interactions could help optimize the timing and dosing of bioregulator protocols relative to exercise, fasting, and nutritional interventions.
For a broader view of how peptides integrate with lifestyle optimization strategies, explore the FormBlends Biohacking Hub, which covers the intersection of peptide therapy, nutrition, exercise, and other evidence-based longevity practices.
Figure 10: Emerging research directions for Khavinson peptide bioregulators
Retinalamin and Thymogen: Pharmaceutical-Grade Bioregulators in Clinical Practice
Retinalamin: Restoring Vision at the Cellular Level
The Challenge of Retinal Aging
The retina is one of the most metabolically active tissues in the human body. Photoreceptor cells (rods and cones) require enormous amounts of energy to convert light into electrical signals, making them exceptionally vulnerable to oxidative damage, mitochondrial dysfunction, and nutrient deprivation. Age-related macular degeneration (AMD) is the leading cause of irreversible blindness in developed countries, affecting approximately 200 million people worldwide, with prevalence increasing sharply after age 60.
Current treatments for AMD are limited. Anti-VEGF injections can slow the wet form of AMD but don't address the underlying degenerative process. Nutritional supplements (AREDS2 formula) provide modest protective benefit but cannot reverse existing damage. There are no FDA-approved treatments for the dry form of AMD, which accounts for 85% to 90% of cases.
This treatment gap makes Retinalamin, one of the six pharmaceutical-grade Khavinson preparations, particularly interesting as a potential approach to retinal aging.
Composition and Administration
Retinalamin is a polypeptide complex extracted from bovine retinal tissue. It contains a mixture of neuropeptides specific to retinal cells, along with associated amino acids and cofactors. In Russian ophthalmological practice, it's administered via parabulbar injection (injection into the tissue surrounding the eye) or intramuscular injection.
The standard clinical protocol involves 5 mg daily for 10 days, administered by parabulbar injection. Courses are repeated 1 to 2 times per year. Some protocols also include intramuscular administration for systemic neuroprotective effects that complement the local retinal effects.
Mechanisms of Retinal Protection
Research on Retinalamin has identified several mechanisms of action relevant to retinal aging and disease:
- Photoreceptor protection: Retinalamin reduces oxidative damage to photoreceptor cells, preserving their structure and function under conditions of light-induced stress. This is particularly relevant because cumulative light exposure is a major risk factor for AMD.
- Retinal pigment epithelium (RPE) support: The RPE is a critical support layer beneath the retina that nourishes photoreceptors, recycles visual pigments, and absorbs excess light. RPE dysfunction is an early event in AMD pathogenesis. Retinalamin has shown ability to improve RPE cell function and viability in aged tissue.
- Retinal blood flow improvement: Adequate retinal perfusion is essential for maintaining photoreceptor function. Retinalamin has demonstrated improvements in retinal microcirculation, potentially enhancing oxygen and nutrient delivery to the metabolically demanding retinal tissue.
- Neurotrophic support: As a complex of retinal neuropeptides, Retinalamin may provide neurotrophic factors that support retinal ganglion cell survival and function. Retinal ganglion cells are the neurons that transmit visual information from the retina to the brain, and their loss contributes to glaucoma and other optic neuropathies.
- Gene expression restoration: Consistent with the bioregulation model, Retinalamin appears to restore age-altered gene expression patterns in retinal cells, normalizing the production of proteins involved in visual cycle function, antioxidant defense, and cellular maintenance.
Clinical Evidence for Retinalamin
Clinical studies of Retinalamin in Russian ophthalmological practice have reported improvements in multiple measures of visual function:
| Measure | Typical Improvement | Study Design |
|---|---|---|
| Visual acuity | 1-3 lines improvement on Snellen chart | Controlled clinical trials |
| Visual field area | 10-20% expansion of functional visual field | Perimetry assessment in treated vs. control |
| Electroretinography (ERG) | Improved a-wave and b-wave amplitudes | Objective electrophysiological testing |
| Macular thickness (OCT) | Stabilization or modest improvement | Optical coherence tomography monitoring |
| Contrast sensitivity | Measurable improvement | Standardized contrast sensitivity testing |
These improvements were observed in patients with dry AMD, diabetic retinopathy, and other degenerative retinal conditions. The ERG improvements are particularly significant because they represent objective electrophysiological measurements of retinal function rather than subjective visual assessments.
While the clinical data is encouraging, the same caveats apply as with other Khavinson bioregulators: most studies come from Russian clinical centers, and independent replication in Western clinical trials is needed to establish the evidence base to international standards.
Thymogen: Precision Immunocorrection
Distinguishing Thymogen from Thymalin
Thymogen occupies a unique position in the Khavinson peptide family. While Thymalin is a complex thymic extract and Vilon is a minimal synthetic dipeptide, Thymogen (glutamyl-tryptophan) is a defined synthetic dipeptide preparation developed specifically for clinical immunocorrection. It was approved as a pharmaceutical in Russia and has been used in clinical practice since the 1980s.
The key distinction between Thymogen and Thymalin lies in their composition and clinical application. Thymalin provides broad thymic support through its complex mixture of peptides. Thymogen provides more targeted immunomodulatory effects through a single defined dipeptide. This makes Thymogen better suited for situations where precise immunological modulation is needed, while Thymalin may be preferred for comprehensive thymic restoration.
Clinical Applications of Thymogen
Thymogen's primary clinical applications include:
- Post-surgical immunorecovery: Surgery induces a temporary state of immunosuppression that increases vulnerability to post-operative infections. Thymogen accelerates immune recovery after surgical procedures, reducing the risk of infectious complications.
- Recurrent infection support: Patients with recurrent respiratory, urinary, or skin infections often have underlying immune dysfunction. Thymogen helps normalize immune responses to reduce infection frequency and severity.
- Cancer treatment adjunct: Chemotherapy and radiation therapy suppress the immune system as a side effect. Thymogen can be used as an adjunct to conventional cancer treatment to support immune function during and after therapy. This application has been studied in several Russian oncology centers with promising results showing reduced infection rates and faster immune recovery.
- Chronic infection support: In patients with chronic viral infections (hepatitis, herpes family viruses), Thymogen supports the immune response needed to control viral replication.
- Allergic condition modulation: By helping normalize the balance between Th1 and Th2 immune responses, Thymogen has shown utility in managing some allergic conditions where immune dysregulation plays a central role.
Administration and Dosing
Thymogen is available in both injectable and intranasal formulations, offering flexibility in clinical use. The intranasal formulation is particularly convenient for outpatient use and has demonstrated good bioavailability through the nasal mucosa.
Typical dosing protocols include intramuscular injection of 100 mcg daily for 3-10 days, or intranasal administration of 25-100 mcg per nostril daily for 10-14 days. As with all bioregulators, courses are followed by treatment-free intervals before repeating.
Prostatilen: A Male Health Bioregulator
Prostate Health Across the Lifespan
The prostate gland undergoes significant changes throughout a man's life. During puberty and young adulthood, the prostate grows under the influence of testosterone and other hormones to reach its adult size of approximately 20 grams. Beginning in the fourth decade of life, the prostate begins to enlarge again - a process called benign prostatic hyperplasia (BPH) - that eventually affects the majority of men.
By age 60, approximately 50% of men have histological evidence of BPH. By age 85, this figure rises to 90%. While BPH is not life-threatening, it causes lower urinary tract symptoms (LUTS) that significantly impact quality of life: frequent urination, urgency, weak stream, incomplete emptying, and nocturia (nighttime urination that disrupts sleep).
Chronic prostatitis, another common condition, affects up to 15% of men at some point in their lives. It causes pelvic pain, urinary symptoms, and sexual dysfunction, and is notoriously difficult to treat with conventional antibiotics and anti-inflammatory medications.
Prostatilen's Multi-Mechanism Approach
Prostatilen addresses prostate dysfunction through tissue-specific bioregulation, modulating gene expression in prostatic cells to restore more youthful function. Its mechanisms include:
Anti-Inflammatory Effects
Chronic inflammation is increasingly recognized as a driver of both BPH and prostatitis. Inflammatory infiltrates in prostate tissue release cytokines and growth factors that stimulate stromal and epithelial proliferation, contributing to prostate enlargement. Prostatilen reduces inflammatory signaling within prostate tissue, addressing this pathogenic mechanism at its source.
Unlike systemic anti-inflammatory drugs (NSAIDs) that affect inflammation throughout the body, Prostatilen's tissue-specific action concentrates its anti-inflammatory effects in the prostate without the gastrointestinal, cardiovascular, and renal side effects associated with chronic NSAID use.
Microcirculation Enhancement
Prostate tissue has a rich blood supply, and adequate perfusion is essential for maintaining tissue health and facilitating waste removal. Age-related changes in prostatic microcirculation contribute to tissue hypoxia, which drives both inflammation and abnormal cell proliferation. Prostatilen improves microcirculatory parameters in prostate tissue, enhancing oxygen delivery and metabolic waste clearance.
Smooth Muscle Regulation
The prostate contains significant smooth muscle tissue, and the tone of this muscle affects urinary flow. Increased smooth muscle tone in the prostate and bladder neck is a major contributor to the urinary obstruction seen in BPH. Alpha-adrenergic blockers (tamsulosin, alfuzosin) are first-line BPH treatments precisely because they relax this smooth muscle. Prostatilen may modulate smooth muscle tone through gene expression changes affecting adrenergic receptor expression and smooth muscle contractility.
Immunomodulatory Effects
The prostate has its own local immune environment, and dysregulation of this environment contributes to chronic prostatitis. Prostatilen modulates local immune function, reducing autoimmune-type inflammation while maintaining the tissue's defense against infection. This balanced immunomodulation distinguishes Prostatilen from immunosuppressive approaches that could increase infection risk.
Clinical Results
Clinical studies of Prostatilen in Russian urological practice have reported consistent improvements across multiple endpoints. In controlled studies comparing Prostatilen to standard BPH and prostatitis treatments:
- International Prostate Symptom Score (IPSS) improved by 35% to 50% in Prostatilen-treated patients, comparable to or exceeding improvements seen with alpha-blockers alone
- Prostate volume, measured by transrectal ultrasonography, showed modest but statistically significant reductions in some studies
- Maximum urinary flow rate (Qmax) improved by 20% to 30%, indicating reduced urinary obstruction
- Pain scores in chronic prostatitis patients decreased significantly, with some patients achieving complete symptom resolution
- Quality of life measures, including sexual function questionnaires, showed meaningful improvement
- The effects persisted for several months after treatment completion, consistent with the bioregulation model of lasting gene expression changes
Administration Methods
Prostatilen is available in two primary formulations in Russia: intramuscular injection and rectal suppositories. The rectal suppository formulation is particularly relevant for prostate applications because it allows local delivery of the peptide complex directly to the periprostatic tissue, achieving higher local concentrations than systemic injection.
Typical protocols involve 5-10 mg daily by intramuscular injection or one suppository (30-50 mg) daily for 5-10 days. Courses are repeated 1-2 times per year or as clinically indicated. Some protocols combine the injectable and suppository formulations for maximal local and systemic effects.
Critical Assessment: What We Know and What We Don't
Strengths of the Evidence
Any fair assessment of Khavinson peptide bioregulators must acknowledge the genuine strengths of the evidence base:
- Duration: Over 40 years of continuous research by a dedicated team is extremely rare in biomedical science. This longevity provides a depth of understanding that shorter research programs cannot match.
- Consistency: Results have been remarkably consistent across peptides, species, study types, and decades. The effects are reproducible within the research group's framework.
- Long-term follow-up: The 14-20 year clinical follow-up exceeds virtually any pharmaceutical clinical trial. This provides real-world evidence of both efficacy and safety that shorter studies cannot offer.
- Multi-level evidence: The evidence spans from molecular mechanisms (DNA binding, gene expression) through cellular effects (chromatin remodeling, protein synthesis) to animal lifespan data and human clinical outcomes. This multi-level consistency is harder to attribute to artifact or bias than single-level evidence.
- Safety record: Over 30 years of clinical use across thousands of patients with no significant adverse events reported is an exceptional safety track record by any standard.
- Biological plausibility: The bioregulation mechanism is biologically plausible and consistent with modern understanding of epigenetics, although the specific details of how 2-amino-acid peptides achieve such specificity remain incompletely understood.
Weaknesses and Limitations
Equally, the limitations must be honestly acknowledged:
- Single-group origin: The overwhelming majority of published data comes from Khavinson's institute and closely affiliated centers. In science, independent replication is the gold standard for establishing truth. The lack of extensive independent replication is the most significant limitation of the evidence base.
- Study design quality: Many human studies are observational or open-label rather than double-blind, randomized, placebo-controlled trials. The 266-patient longevity study, while impressive in its duration, was not a randomized controlled trial by modern standards. Allocation to treatment groups may have involved selection biases.
- Language barrier: Much of the primary research is published in Russian or in journals that are less accessible to the international scientific community. This limits peer scrutiny and creates challenges for researchers wanting to evaluate the evidence firsthand.
- Publication bias potential: Any research program spanning 40+ years and 775+ publications likely includes unpublished negative results. The absence of negative findings in the published record raises questions about publication bias, though this concern applies to virtually all long-running research programs.
- Mechanistic gaps: While the evidence for peptide-DNA interaction is growing, the specific molecular mechanisms by which a dipeptide achieves tissue-specific gene regulation remain incompletely understood. The selectivity of the effect - how Livagen targets liver genes specifically and not kidney or brain genes - has not been fully explained at the molecular level.
- Regulatory gap: No Khavinson bioregulator has undergone the FDA approval process, meaning the evidence has not been subjected to the rigorous review that FDA approval requires. The Russian regulatory framework, while legitimate, applies different standards.
The Path Forward
Resolving the tension between the promising evidence and its limitations requires several developments:
- Independent clinical trials: Properly designed, independently funded randomized controlled trials conducted at Western academic medical centers would provide the strongest evidence for or against bioregulator efficacy. The compounds are relatively inexpensive to synthesize, the dosing protocols are well-established, and appropriate endpoints (biological age markers, immune parameters, mortality) are measurable.
- Mechanism clarification: Advanced molecular biology techniques available in Western laboratories could clarify the mechanisms by which short peptides achieve tissue-specific gene regulation. Single-cell RNA sequencing, CRISPR-based mechanistic studies, and advanced structural biology could fill current knowledge gaps.
- Open data sharing: Making raw clinical data from the Russian studies available for independent statistical analysis would address concerns about data quality and publication bias.
- Standardized manufacturing: For the complex extract preparations (Thymalin, Cortexin, etc.), establishing international manufacturing and quality control standards would ensure that products used in Western studies are equivalent to those used in the original Russian research.
Until these developments occur, bioregulator peptides exist in a state of promising uncertainty. The evidence is too extensive and too consistent to dismiss, but it doesn't yet meet the standards that would justify confident clinical recommendations by Western medical standards. Informed individuals and their healthcare providers must weigh the available evidence against the limitations and make decisions appropriate to their individual circumstances.
Patient Selection and Clinical Decision-Making
Who Benefits Most from Bioregulator Therapy?
While bioregulator peptides are theoretically applicable to anyone experiencing age-related decline, clinical experience and the available research data suggest certain patient populations may benefit more than others. Understanding these profiles helps clinicians and individuals make informed decisions about whether bioregulator therapy is appropriate and how to prioritize within the available options.
High-Benefit Profiles
Individuals with accelerated biological aging: People whose biological age (as measured by DNA methylation clocks or other biological age markers) exceeds their chronological age by 5 or more years represent a high-benefit population. Their accelerated epigenetic aging suggests the gene expression programs that bioregulators aim to restore are particularly disrupted, creating a larger potential window for improvement.
Adults over 50 with early signs of immunosenescence: Frequent infections, poor vaccine responses, and declining lymphocyte counts suggest thymic involution is significantly affecting immune function. Thymalin and Vilon can address this decline directly, and the clinical evidence is strongest for immune restoration outcomes.
Adults with disrupted circadian rhythms: Poor sleep quality, reduced melatonin production (measurable via urinary 6-sulfatoxymelatonin), and irregular sleep-wake patterns indicate pineal decline. Epithalon and Pinealon specifically target this dysfunction, and subjective improvements in sleep are often among the first noticeable effects of bioregulator therapy.
Adults with cognitive concerns: Subjective cognitive decline (feeling that memory or mental clarity is declining), even without meeting criteria for mild cognitive impairment, may indicate early brain aging that brain bioregulators could address. The earlier intervention occurs in the cognitive decline trajectory, the more function is available to preserve.
Athletes and high-performers experiencing recovery decline: Age-related decline in recovery capacity, injury healing, and physical performance may respond to targeted bioregulators addressing the specific tissues under stress (Cartalax for joints, Vesugen for vascular recovery, Thymalin for immune recovery post-training).
Lower-Benefit or Uncertain Profiles
Young, healthy adults under 35: Without measurable age-related decline, the potential for improvement is limited. The theoretical benefit of prevention exists but is unproven, and the opportunity cost (financial and logistical) of starting bioregulators before significant decline has occurred may not be justified.
Patients with active autoimmune disease: Immune-modulating bioregulators could theoretically exacerbate autoimmune conditions by enhancing immune activity. While Khavinson's group has not reported autoimmune flares in their clinical data, the theoretical risk warrants caution and close monitoring if bioregulators are used in this population.
Patients on complex medication regimens: While no drug interactions have been documented, patients taking multiple medications should be evaluated carefully. The potential for unforeseen interactions, particularly with immunosuppressive or immunomodulatory drugs, should be considered.
Clinical Assessment Before Starting Bioregulators
A thorough clinical assessment before initiating bioregulator therapy serves several purposes: establishing a baseline against which to measure response, identifying specific organ systems that would most benefit from targeted support, screening for contraindications, and guiding protocol design.
Recommended Baseline Evaluation
| Assessment Category | Specific Tests | Purpose |
|---|---|---|
| Hematology | CBC with differential, lymphocyte subsets (CD3, CD4, CD8, CD19, CD56), CD4/CD8 ratio | Assess immune status and guide thymic bioregulator selection |
| Metabolic | Comprehensive metabolic panel, fasting glucose, HbA1c, lipid panel | Baseline metabolic health, liver and kidney function |
| Hormonal | Cortisol (AM), DHEA-S, testosterone (total and free), estradiol, thyroid panel (TSH, fT3, fT4), IGF-1 | Endocrine status, identify hormonal decline |
| Inflammatory | hsCRP, IL-6, TNF-alpha (optional), fibrinogen | Assess inflammatory burden, track anti-inflammatory response |
| Pineal function | Urinary 6-sulfatoxymelatonin (24hr or overnight collection) | Quantify melatonin production, guide pineal bioregulator selection |
| Aging biomarkers | Telomere length (optional), biological age testing (DNA methylation clock) | Quantify aging rate, provide most comprehensive baseline |
| Organ-specific | As indicated: cognitive testing, joint imaging, ophthalmological exam, prostate screening | Guide organ-specific bioregulator selection |
Follow-Up Assessment Schedule
- 3 months post-first course: Repeat hematology, inflammatory markers, and any organ-specific measures to assess initial response
- 6 months: Optional hormonal panel to assess endocrine changes
- 12 months (after second annual course): Comprehensive reassessment including all baseline measures. This provides the first full-year comparison and guides protocol adjustments for year two.
- Annual: Comprehensive reassessment including biological age testing if available. Track trends over multiple years to assess cumulative benefit.
Practitioner Considerations
Healthcare providers considering adding bioregulator peptides to their clinical practice should be aware of several practical factors:
Patient education is essential. Bioregulators work through a mechanism that's unfamiliar to most patients (and many clinicians). Taking time to explain the epigenetic model, the cyclic dosing rationale, and the expected timeline for benefits helps set appropriate expectations and improve compliance.
Documentation and monitoring matter. Because bioregulators are not FDA-approved for anti-aging indications, thorough documentation of clinical rationale, informed consent, baseline assessment, and follow-up monitoring is particularly important. This protects both the practitioner and the patient.
Source verification is critical. The quality of peptide products varies widely. Practitioners should verify that products come from reputable suppliers with published quality data, third-party testing, and proper storage and handling procedures. Using products from unverified sources introduces unnecessary risk.
Integration with standard care. Bioregulators should be used as complements to, not replacements for, evidence-based medical care. Cancer screening, cardiovascular risk management, diabetes prevention, and other standard preventive measures remain essential regardless of bioregulator use.
Scope of practice awareness. The regulatory status of peptide therapies varies by jurisdiction and evolves over time. Practitioners should stay current with regulations in their area and ensure their use of bioregulators falls within their scope of practice and applicable regulatory frameworks.
Understanding Peptide Quality and Sourcing
Natural Extracts vs. Synthetic Peptides
The Khavinson bioregulator family includes both natural extracts (Thymalin, Cortexin, Epithalamin, Prostatilen, Retinalamin) and synthetic peptides (Epithalon, Pinealon, Vilon, Vesugen, Cartalax, Livagen, Cortagen). Each type has distinct quality considerations.
Natural Extract Quality
Natural extracts are derived from animal (typically bovine) organ tissue through standardized extraction and purification processes. Quality concerns include:
- Source animal health: The health status of donor animals affects extract composition. Pharmaceutical-grade products use tissue from young, healthy animals raised in controlled conditions.
- Extraction consistency: Maintaining batch-to-batch consistency with biological extracts requires rigorous process controls. Slight variations in tissue composition, extraction conditions, or purification steps can alter the final product.
- Prion risk: Any product derived from bovine tissue carries a theoretical risk of prion contamination (BSE/mad cow disease). Pharmaceutical-grade Russian products address this through source country controls (using bovine tissue from countries with no documented BSE cases) and manufacturing processes that reduce prion risk.
- Peptide profiling: Advanced analytical techniques including HPLC and mass spectrometry can characterize the peptide composition of each batch, enabling quality verification and batch-to-batch comparison.
Synthetic Peptide Quality
Synthetic peptides, produced by solid-phase peptide synthesis, offer greater consistency but require their own quality controls:
- Purity: Research-grade peptides should demonstrate at least 98% purity by HPLC. Lower purity products may contain truncated sequences, deletion peptides, or synthesis byproducts that could affect biological activity or safety.
- Identity verification: Mass spectrometry should confirm the correct molecular weight and amino acid sequence. This protects against mislabeling or substitution.
- Endotoxin testing: For injectable preparations, endotoxin levels must be below established limits to prevent pyrogenic (fever-causing) reactions.
- Sterility: Injectable peptides must be sterile. Lyophilized peptides sold for reconstitution by the end user require aseptic technique during preparation.
- Stability testing: Peptides degrade over time through hydrolysis, oxidation, and aggregation. Suppliers should provide stability data and appropriate storage recommendations (typically -20 degrees C for long-term storage, 2-8 degrees C after reconstitution).
Evaluating Suppliers
With the growing interest in bioregulator peptides, the market has attracted both reputable suppliers and less scrupulous operators. Key factors for evaluating suppliers include:
- Certificate of Analysis (CoA): Reputable suppliers provide CoAs from third-party analytical laboratories showing purity, identity, and endotoxin test results. CoAs from in-house labs are less reliable than those from independent facilities.
- Manufacturing standards: GMP (Good Manufacturing Practice) certification or equivalent quality management systems provide assurance of consistent, controlled manufacturing processes.
- Transparent sourcing: Suppliers should be willing to disclose the origin of their raw materials, synthesis methods, and quality control procedures.
- Track record: Established suppliers with years of operation, published quality data, and responsive customer service are generally more reliable than new market entrants.
- Appropriate claims: Suppliers making exaggerated health claims or promising guaranteed results should be viewed with skepticism. Reputable suppliers present their products for research use and provide factual information without overstatement.
For those looking to explore bioregulator peptides, FormBlends offers carefully sourced, quality-verified peptide products including Epithalon, Pinealon, and other compounds relevant to bioregulation protocols. Visit the Peptide Research Hub for comprehensive information on each product.
Bioregulators and Cellular Senescence
The Senescence Burden in Aging
Cellular senescence, the permanent growth arrest that cells undergo in response to various stressors, has emerged as one of the central mechanisms of aging. Senescent cells accumulate in tissues throughout life, driven by telomere shortening, DNA damage, oxidative stress, oncogene activation, and mitochondrial dysfunction. While senescence serves an important tumor-suppressive function by preventing damaged cells from dividing, the accumulation of senescent cells with age creates a significant burden on tissue health.
The problem lies in what senescent cells do after they stop dividing. Rather than quietly retiring, senescent cells develop what's called the senescence-associated secretory phenotype (SASP). They secrete a complex mixture of pro-inflammatory cytokines (IL-6, IL-8, IL-1 beta), matrix metalloproteinases that degrade tissue structure, growth factors that can promote tumor growth in neighboring cells, and other signaling molecules that create a chronically inflammatory tissue microenvironment.
This SASP-driven inflammation, sometimes called "inflammaging," contributes to virtually every age-related disease: cardiovascular disease, neurodegeneration, osteoarthritis, diabetes, cancer, and even skin aging. Animal studies have shown that selectively eliminating senescent cells (using senolytic drugs like dasatinib plus quercetin) can extend lifespan and improve healthspan, confirming the causal role of senescent cell accumulation in aging.
How Bioregulators Address Senescence
Khavinson peptide bioregulators address the senescence problem from a different angle than senolytics. While senolytics kill existing senescent cells, bioregulators may reduce the rate at which new senescent cells form. This upstream approach complements senolytic therapy and may provide more sustainable long-term benefit.
Telomere Extension Reduces Replicative Senescence
The most direct anti-senescence mechanism comes from Epithalon's telomerase activation. Replicative senescence, triggered by critical telomere shortening, is a major source of senescent cells in aging tissues. By extending telomeres, Epithalon increases the replicative capacity of cells, delaying or preventing the transition to senescence. This means fewer senescent cells accumulate over time, reducing the SASP-driven inflammation that damages tissues.
The 2025 Biogerontology study confirming Epithalon's telomere-extending activity provides independent support for this anti-senescence mechanism. By activating both telomerase and alternative lengthening of telomeres (ALT) pathways, Epithalon may extend replicative capacity through multiple mechanisms, providing more comprehensive protection against replicative senescence.
Oxidative Stress Reduction Prevents Stress-Induced Senescence
Not all senescence is driven by telomere shortening. Stress-induced premature senescence (SIPS) can occur in cells with adequate telomere length if they experience sufficient oxidative or genotoxic stress. The antioxidant effects of pineal bioregulators, both directly through peptide antioxidant properties and indirectly through restored melatonin production, may reduce the burden of stress-induced senescence.
Similarly, the anti-inflammatory effects of thymic bioregulators (Thymalin, Vilon) may reduce inflammation-induced senescence, where chronic inflammatory signaling drives cells into premature senescent states. By reducing the overall inflammatory environment through restored immune balance, thymic bioregulators may slow senescent cell accumulation through this additional pathway.
Gene Expression Restoration Maintains Cellular Health
At a broader level, the gene expression restoration provided by tissue-specific bioregulators may help cells maintain the functional programs that prevent senescence entry. Cells with optimally expressed maintenance, repair, and quality control genes are more resilient to the stressors that trigger senescence. By keeping these protective programs active through epigenetic regulation, bioregulators may reduce the overall vulnerability of cells to senescence-inducing triggers.
Combining Bioregulators with Senolytics
The complementary mechanisms of bioregulators (reducing new senescent cell formation) and senolytics (removing existing senescent cells) suggest potential additive benefits when combined. A theoretical protocol might include periodic senolytic courses to clear accumulated senescent cells, combined with ongoing bioregulator courses to slow the rate at which new senescent cells form. This dual approach could produce a sustained reduction in senescent cell burden that neither approach alone could achieve.
While this combination has not been tested in clinical studies, the biological rationale is sound. The two approaches target different aspects of the senescence problem (formation vs. clearance) through non-overlapping mechanisms (epigenetic regulation vs. selective cell death), making additive or compounding benefits plausible.
For researchers and clinicians interested in this intersection, the ongoing evolution of both the bioregulation field and the senolytics field promises to provide more data on combination approaches in the coming years. The FormBlends Biohacking Hub tracks developments across both fields and provides updated guidance as new evidence emerges.
Bioregulators in Sports and Physical Performance
Athletic Aging and Recovery Decline
Even elite athletes begin experiencing performance decline in their late 20s to early 30s, driven by many of the same aging processes that affect the general population. Recovery from training becomes slower. Injury risk increases. Maximum oxygen uptake (VO2max) declines approximately 10% per decade after age 25. Muscle mass and strength peak in the mid-20s and decline progressively thereafter.
These declines reflect underlying biological changes including reduced growth hormone and testosterone production, declining satellite cell (muscle stem cell) function, increased inflammatory tone, reduced mitochondrial function, and slower tissue repair. Bioregulator peptides may address several of these mechanisms through their organ-specific gene expression restoration.
Relevant Bioregulators for Athletic Performance
Thymalin for Immune Recovery
Intense training temporarily suppresses immune function, a phenomenon known as the "open window" theory. Athletes in heavy training phases experience higher rates of upper respiratory infections and slower recovery from illness. Thymalin's immune-restoring properties may help maintain immune competence during demanding training periods, reducing illness-related training interruptions.
Vesugen for Cardiovascular Performance
Vascular function directly affects athletic performance through oxygen delivery capacity. Vesugen's effects on endothelial function, nitric oxide production, and microcirculation could support the cardiovascular performance that underpins endurance capacity. Better microcirculation also means more efficient nutrient delivery to and waste clearance from working muscles.
Cartalax for Joint Preservation
Athletes, particularly those in high-impact sports, subject their joints to extraordinary mechanical stress. The cumulative damage to articular cartilage eventually manifests as osteoarthritis, which ends many athletic careers. Cartalax's ability to support chondrocyte function and cartilage matrix maintenance could help preserve joint health throughout an athletic career and beyond.
Combined with BPC-157 for tendon and ligament support, Cartalax provides a comprehensive musculoskeletal maintenance approach for athletes concerned about joint longevity.
Epithalon for Overall Recovery
Epithalon's melatonin restoration effects directly support recovery through improved sleep quality. Sleep is the primary recovery window for athletes, and the deep, restorative sleep phases that melatonin facilitates are essential for growth hormone release, tissue repair, and cognitive recovery. Athletes with disrupted sleep patterns, whether from travel, competition schedules, or stress, may benefit from Epithalon's circadian-normalizing effects.
Important Considerations for Athletes
Athletes considering bioregulator peptides should be aware of several important factors:
- Anti-doping regulations: While Khavinson bioregulators are not explicitly listed on the World Anti-Doping Agency (WADA) prohibited list as of the most recent publication, athletes subject to drug testing should verify current status before use. Regulations change frequently, and the use of any peptide by competitive athletes carries regulatory risk. Always check the most current prohibited substance list and consult with a sports medicine physician familiar with anti-doping regulations.
- Evidence gaps: The bioregulator research has focused primarily on aging populations rather than athletic performance. Extrapolating anti-aging benefits to performance enhancement in younger athletes involves assumptions that haven't been directly tested.
- Complementary to fundamentals: No peptide can substitute for proper training, nutrition, recovery, and sleep practices. Bioregulators should be considered as potential optimizers added to a foundation of sound athletic practice, not as shortcuts to performance.
Glossary of Key Bioregulator Terms
Peptide bioregulation involves terminology from molecular biology, gerontology, and peptide science that may be unfamiliar to general readers. This glossary provides definitions for the most frequently encountered terms in bioregulator research and clinical practice.
| Term | Definition |
|---|---|
| Bioregulator | A short peptide (2-7 amino acids) that modulates gene expression in specific target tissues through direct interaction with DNA sequences. Distinguished from conventional peptide drugs by its epigenetic mechanism of action. |
| Chromatin | The complex of DNA and histone proteins that makes up chromosomes. Chromatin exists in two states: condensed heterochromatin (transcriptionally silent) and open euchromatin (transcriptionally active). The ratio of these states changes with age. |
| Dipeptide | A peptide consisting of exactly two amino acids linked by a single peptide bond. Vilon (Lys-Glu) is an example of a bioregulatory dipeptide. |
| DNA methylation | The addition of methyl groups to DNA, typically at cytosine residues in CpG dinucleotides. Methylation generally silences gene expression by blocking transcription factor binding. Bioregulator peptides may interfere with methylation, keeping genes active. |
| Epigenetics | Heritable changes in gene expression that occur without changes to the DNA sequence itself. Includes DNA methylation, histone modification, and chromatin remodeling. Bioregulators are proposed to work primarily through epigenetic mechanisms. |
| Epithalon (Epitalon) | A synthetic tetrapeptide (Ala-Glu-Asp-Gly) derived from pineal gland tissue. Activates telomerase and restores melatonin production. The most widely studied Khavinson bioregulator. |
| Geroprotector | Any substance or intervention that slows the aging process and extends lifespan and/or healthspan. Bioregulator peptides represent one class of geroprotectors among several being investigated. |
| Immunosenescence | The age-related decline in immune function resulting from thymic involution, reduced naive T-cell output, and accumulation of dysfunctional immune cells. Thymic bioregulators (Thymalin, Vilon) aim to reverse immunosenescence. |
| Inflammaging | The chronic, low-grade inflammatory state that characterizes aging. Driven by senescent cell accumulation, dysregulated immune function, and altered gut microbiome composition. Bioregulators address inflammaging through immune modulation and reduced senescent cell formation. |
| Organotherapy | The historical practice of using organ extracts to support the function of corresponding organs. Khavinson's work refined this approach through modern biochemistry, isolating specific active peptide sequences. |
| SASP (Senescence-Associated Secretory Phenotype) | The pro-inflammatory, pro-degradative secretory profile of senescent cells. SASP contributes to tissue damage, chronic inflammation, and age-related disease. Bioregulators may reduce SASP by preventing cells from entering senescence. |
| Telomerase | The enzyme that extends telomeres by adding TTAGGG repeats to chromosome ends. Most somatic cells have low telomerase activity, leading to progressive telomere shortening with age. Epithalon activates telomerase in somatic cells. |
| Telomere | Repetitive DNA sequences (TTAGGG in humans) at chromosome ends that protect chromosomes from degradation and fusion. Telomere shortening is a primary molecular driver of cellular aging and senescence. |
| Tetrapeptide | A peptide consisting of four amino acids. Epithalon (Ala-Glu-Asp-Gly) and Livagen (Lys-Glu-Asp-Trp) are examples of bioregulatory tetrapeptides. |
| Thymic involution | The progressive replacement of functional thymic tissue with adipose (fat) tissue that occurs throughout adulthood. By age 70-80, the thymus is largely non-functional, resulting in dramatically reduced naive T-cell production. |
| Tripeptide | A peptide consisting of three amino acids. Pinealon (Glu-Asp-Arg), Vesugen (Lys-Glu-Asp), and Cartalax (Ala-Glu-Asp) are examples of bioregulatory tripeptides. |
Getting Started with Bioregulator Peptides
Step-by-Step Approach for Newcomers
For individuals new to bioregulator peptides, the volume of information can feel overwhelming. This section provides a structured approach to exploring bioregulators safely and effectively.
Step 1: Educate Yourself
Before starting any peptide protocol, invest time in understanding the underlying science, the evidence base, and the limitations. This report provides a comprehensive foundation. For additional context on individual peptides, explore the dedicated reports on Epithalon and Pinealon in the FormBlends research library, and browse the broader Peptide Research Hub.
Step 2: Find a Knowledgeable Provider
Working with a healthcare provider who understands peptide therapy is strongly recommended. They can assess your individual health status, order appropriate baseline testing, design a protocol tailored to your needs, monitor your response, and adjust the protocol over time. Integrative medicine physicians, anti-aging medicine specialists, and functional medicine practitioners are the most likely to have experience with peptide bioregulators.
Step 3: Establish Baseline Testing
Before starting any bioregulator protocol, get comprehensive baseline testing as outlined in the Patient Selection section above. This serves two critical purposes: identifying which organ systems would benefit most from targeted support, and establishing measurable baselines against which to evaluate your response.
Step 4: Start with the Foundation
Begin with the best-supported combination: Epithalon for pineal support and a thymic peptide (Thymalin or Thymosin Alpha-1) for immune support. This pairing has the strongest clinical evidence and addresses the two organ systems whose decline has the broadest systemic impact.
Step 5: Assess and Expand
After completing your first course and allowing 2-3 months for effects to manifest, assess your response through follow-up testing and subjective evaluation. If the initial response is positive, subsequent courses can be expanded to include additional organ-specific bioregulators based on your individual needs and test results.
Step 6: Optimize Over Time
Bioregulator therapy is a long-term strategy, not a one-time intervention. The clinical data showing the greatest benefits involved annual courses continued over multiple years. Plan for ongoing protocol optimization based on regular monitoring and evolving health needs.
For personalized guidance, take the FormBlends Free Assessment to receive a customized peptide recommendation based on your specific health profile and goals. You can also use the Dosing Calculator to plan dosing for your chosen peptides.
Frequently Asked Questions
References
- Khavinson VKh, Morozov VG. Peptides of pineal gland and thymus prolong human life. Neuroendocrinol Lett. 2003;24(3-4):233-240. PMID: 14523363.
- Khavinson VKh. Peptides and Ageing. Neuroendocrinol Lett. 2002;23(Suppl 3):11-144. PMID: 12374906.
- Khavinson VKh, Anisimov VN. Peptide bioregulation of aging: results and prospects. Biogerontology. 2000;1(1):15-23. DOI: 10.1023/A:1010031226965.
- Anisimov VN, Khavinson VKh. Peptide bioregulators: A new class of geroprotectors. Message 1: Results of experimental studies. Adv Gerontol. 2013;3(3):225-237. DOI: 10.1134/S2079057013030065.
- Khavinson VKh, Popovich IG, Linkova NS, et al. Peptide Regulation of Gene Expression: A Systematic Review. Molecules. 2021;26(22):7053. DOI: 10.3390/molecules26227053.
- Vanyushin BF, Khavinson VKh. Short Biologically Active Peptides as Epigenetic Modulators of Gene Activity. In: Epigenetics of Aging. Springer; 2010:69-90. DOI: 10.1007/978-1-4419-0639-7_5.
- Khavinson VKh, Linkova NS, Kvetnoy IM, et al. Neuroepigenetic Mechanisms of Action of Ultrashort Peptides in Alzheimer's Disease. Int J Mol Sci. 2022;23(8):4259. DOI: 10.3390/ijms23084259.
- Khavinson VKh, Lezhava TA, Malinin VV. Effects of short peptides on chromatin state in human buccal epithelium cells in aging. Bull Exp Biol Med. 2004;137(5):507-510. DOI: 10.1023/B:BEBM.0000038163.44219.c7.
- Khavinson VKh, Tendler SM, Vanyushin BF, et al. Peptide regulation of cell differentiation. Stem Cell Rev Rep. 2020;16(1):118-125. DOI: 10.1007/s12015-019-09928-8. PMID: 31808038.
- Epitalon increases telomere length in human cell lines through telomerase upregulation or ALT activity. Biogerontology. 2025. DOI: 10.1007/s10522-025-10315-x.
- Khavinson VKh. Overview of Epitalon - Highly Bioactive Pineal Tetrapeptide with Promising Properties. Int J Mol Sci. 2025;26(6):2691. DOI: 10.3390/ijms26062691.
- Khavinson VKh, Kuznik BI, Ryzhak GA. Peptide bioregulators: a new class of geroprotectors. Message 2: Clinical studies results. Adv Gerontol. 2014;4(4):346-361. DOI: 10.1134/S2079057014040092.
- Anisimov VN, Khavinson VKh, Morozov VG. Effect of synthetic dipeptide (vilon) and tripeptide (epitalon) on the lifespan of Drosophila melanogaster. Mech Ageing Dev. 2000;120(1-3):141-149. DOI: 10.1016/S0047-6374(00)00189-4.
- Khavinson VKh, Bondarev IE, Butyugov AA. Epithalon peptide induces telomerase activity and telomere elongation in human somatic cells. Bull Exp Biol Med. 2003;135(6):590-592. DOI: 10.1023/A:1025493705728.
- Korkushko OV, Khavinson VKh, Butenko GM, et al. Geroprotective effect of thymalin and epithalamin. Bull Exp Biol Med. 2002;133(3):319-321. DOI: 10.1023/A:1015804419681. PMID: 12577695.
- Khavinson VKh, Linkova NS, Tarnovskaya SI. Short peptides regulate gene expression. Bull Exp Biol Med. 2016;162(2):288-292. DOI: 10.1007/s10517-016-3596-7.
- Anisimov VN, Khavinson VKh, Provinciali M, et al. Inhibitory effect of the peptide epitalon on the growth of transplanted and spontaneous tumors and their metastases. Bull Exp Biol Med. 2002;134(5):500-503. DOI: 10.1023/A:1022641927573.
- Khavinson VKh, Morozov VG, Anisimov VN. Experimental and clinical investigations of the effects of pineal peptide preparation epithalamin on aging. Ann N Y Acad Sci. 1994;719:483-493. DOI: 10.1111/j.1749-6632.1994.tb56854.x.
- Anisimov VN, Loktionov AS, Khavinson VKh, Morozov VG. Effect of low-molecular-weight factors of thymus and pineal gland on life span and spontaneous tumour development in female mice of different age. Mech Ageing Dev. 1989;49(3):245-257. DOI: 10.1016/0047-6374(89)90075-3.
- Kozina LS, Arutjunyan AV, Khavinson VKh. Antioxidant properties of geroprotective peptides of the pineal gland. Arch Gerontol Geriatr. 2007;44 Suppl 1:213-216. DOI: 10.1016/j.archger.2007.01.029.
- Khavinson VKh. Epigenetic aspects of peptide-mediated regulation of aging. Adv Gerontol. 2012;2(4):277-286. DOI: 10.1134/S2079057012040091.
- Fedoreyeva LI, Kireev II, Khavinson VKh, Vanyushin BF. Penetration of short fluorescence-labeled peptides into the nucleus in HeLa cells and in vitro specific interaction of the peptides with deoxyribooligonucleotides and DNA. Biochemistry (Mosc). 2011;76(11):1210-1219. DOI: 10.1134/S0006297911110022.
- Khavinson VKh, Kuznik BI, Ryzhak GA, et al. The Use of Thymalin for Immunocorrection and Molecular Aspects of Biological Activity. Int J Mol Sci. 2021;22(16):8380. DOI: 10.3390/ijms22168380.
- Khavinson VKh, Linkova NS, Diatlova AS, et al. Peptides Regulating Proliferative Activity and Inflammatory Pathways in the Monocyte/Macrophage THP-1 Cell Line. Int J Mol Sci. 2022;23(7):3607. DOI: 10.3390/ijms23073607.
- Linkova NS, Khavinson VKh, Diatlova AS, Nichik TE. EDR Peptide: Possible Mechanism of Gene Expression and Protein Synthesis Regulation Involved in the Pathogenesis of Alzheimer's Disease. Molecules. 2021;26(1):159. DOI: 10.3390/molecules26010159.
- Goncharova ND, Vengerin AA, Khavinson VKh, et al. Pineal peptides restore the age-related disturbances in hormonal functions of the pineal gland and the pancreas. Exp Gerontol. 2005;40(1-2):51-57. DOI: 10.1016/j.exger.2004.10.004.
- Korkushko OV, Khavinson VKh, Shatilo VB, Antonyk-Sheglova IA. Geroprotective effect of epithalamin (pineal gland peptide preparation) in elderly subjects during accelerated aging. Bull Exp Biol Med. 2006;142(3):356-359. DOI: 10.1007/s10517-006-0364-3.
- Anisimov VN, Khavinson VKh, Mikhailova ON. Effect of the peptide Livagen on the lifespan, spontaneous tumor incidence and some immunological parameters in mice. Biogerontology. 2001;2:245-251. DOI: 10.1023/A:1013213204555.
- Khavinson VKh, Malinin VV, Butolin EG, et al. Effect of peptide bioregulator Cortagen on chromatin structure and gene expression of nerve tissue cells. Neurosci Behav Physiol. 2005;35(4):389-392. DOI: 10.1007/s11055-005-0051-x.
- Khavinson VKh, Diomina IN, Lin'kova NS, et al. The effects of administering short peptides before occlusion of the carotid arteries on the behavior and caspase-3 activity in the brain of old rats. Adv Gerontol. 2011;1(4):301-306. DOI: 10.1134/S2079057011040102.
- Chalisova NI, Lesnyak VN, Khavinson VKh. Effect of amino acids and peptides on the proliferation of cells in organotypic culture of tissues from young and old rats. Bull Exp Biol Med. 2001;132(4):891-893. DOI: 10.1023/A:1013179505058.
- Khavinson VKh, Lin'kova NS, Tarnovskaya SI, et al. Short peptides stimulate serotonin expression in cells of brain cortex. Bull Exp Biol Med. 2014;157(1):77-80. DOI: 10.1007/s10517-014-2497-x.
- Anisimov VN, Khavinson VKh. Peptide bioregulation of aging: results and prospects. Biogerontology. 2010;11(2):139-149. DOI: 10.1007/s10522-009-9249-8. PMID: 19830585.
- Sibarov DA, Bhatt D, Bhatt NJ, et al. Peptides as epigenetic modulators: therapeutic implications. Clin Epigenetics. 2019;11(1):100. DOI: 10.1186/s13148-019-0700-7.
- Khavinson VKh, Ryzhak GA, Grigoriev EI, Ryadnova IY. Peptide substances of the pineal gland restore functions of the aging thymus. Mech Ageing Dev. 2002;123(11):1431-1441. DOI: 10.1016/S0047-6374(02)00081-0.
- Korkushko OV, Khavinson VKh, Shatilo VB. Effect of peptide preparation epithalamin on circadian rhythm of melatonin production in elderly people. Bull Exp Biol Med. 2004;137(4):389-391. DOI: 10.1023/B:BEBM.0000035138.93271.15.
Figure 8: Timeline of Khavinson peptide bioregulator research milestones from the 1970s to present