Executive Summary
The gastrointestinal tract isn't just where you digest food - it's a 30-foot barrier system that decides what enters your bloodstream and what stays out. When that barrier breaks down, the consequences ripple through every organ system. Peptide therapy offers a targeted approach to restoring gut integrity that conventional treatments often miss.
Key Takeaways
- BPC-157 is a gastric pentadecapeptide stable in human stomach acid that promotes mucosal healing, angiogenesis, and cytoprotection throughout the GI tract. Over 100 preclinical studies support its gut-healing properties.
- KPV is a tripeptide (Lys-Pro-Val) from alpha-melanocyte-stimulating hormone that inhibits NF-kB signaling and reduces intestinal inflammation via the PepT1 transporter.
- Larazotide acetate is the first-in-class tight junction regulator derived from zonulin, currently in Phase III trials for celiac disease, that directly stabilizes paracellular permeability.
- VIP regulates gut motility, secretion, and immune responses while maintaining barrier homeostasis - particularly relevant for SIBO and motility disorders.
- LL-37 provides antimicrobial defense at the mucosal surface, modulates gut microbiome composition, and strengthens epithelial barrier integrity.
Gut barrier dysfunction - sometimes called "leaky gut" or increased intestinal permeability - sits at the intersection of dozens of chronic conditions. From inflammatory bowel disease (IBD) and irritable bowel syndrome (IBS) to autoimmune disorders, metabolic syndrome, and even neuropsychiatric conditions, the integrity of your intestinal lining plays a far larger role in overall health than most people realize. And while diet, lifestyle, and conventional medications form the foundation of any gut healing strategy, a growing body of preclinical and early clinical research suggests that specific peptides can accelerate barrier repair, reduce mucosal inflammation, and restore normal gut function in ways that other interventions cannot.
This report examines the science behind the most promising gut-healing peptides: BPC-157 (Body Protection Compound), KPV (the anti-inflammatory tripeptide from alpha-MSH), larazotide acetate (the tight junction regulator), VIP (Vasoactive Intestinal Peptide), and LL-37 (the antimicrobial cathelicidin peptide). We'll cover what each one does at the molecular level, what the research actually shows, and how practitioners are combining them into structured gut healing protocols.
Key Takeaways
- BPC-157 is a gastric pentadecapeptide stable in human stomach acid that promotes mucosal healing, angiogenesis, and cytoprotection throughout the GI tract. Over 100 preclinical studies support its gut-healing properties.
- KPV is a tripeptide (Lys-Pro-Val) from alpha-melanocyte-stimulating hormone that inhibits NF-kB signaling and reduces intestinal inflammation via the PepT1 transporter.
- Larazotide acetate is the first-in-class tight junction regulator derived from zonulin, currently in Phase III trials for celiac disease, that directly stabilizes paracellular permeability.
- VIP regulates gut motility, secretion, and immune responses while maintaining barrier homeostasis - particularly relevant for SIBO and motility disorders.
- LL-37 provides antimicrobial defense at the mucosal surface, modulates gut microbiome composition, and strengthens epithelial barrier integrity.
- Combining peptides in a phased protocol - aligned with the functional medicine 5R framework - may produce outcomes greater than any single agent alone.
Before we examine each peptide individually, it helps to understand the biological system they're working on. The gut barrier is not a simple wall - it's a dynamic, multi-layered defense system with tight junctions, mucus layers, antimicrobial peptides, and immune surveillance all working in concert. When we understand how the barrier works and how it fails, the rationale for each peptide becomes much clearer.
Research Status Disclaimer
Most peptides discussed in this report have strong preclinical evidence but limited human clinical trial data. Larazotide acetate is the exception, with Phase III trial data in celiac disease. BPC-157, KPV, VIP, and LL-37 are research compounds not approved by the FDA for any medical indication. This report is for educational purposes only. Always consult a qualified healthcare provider before starting any peptide protocol.
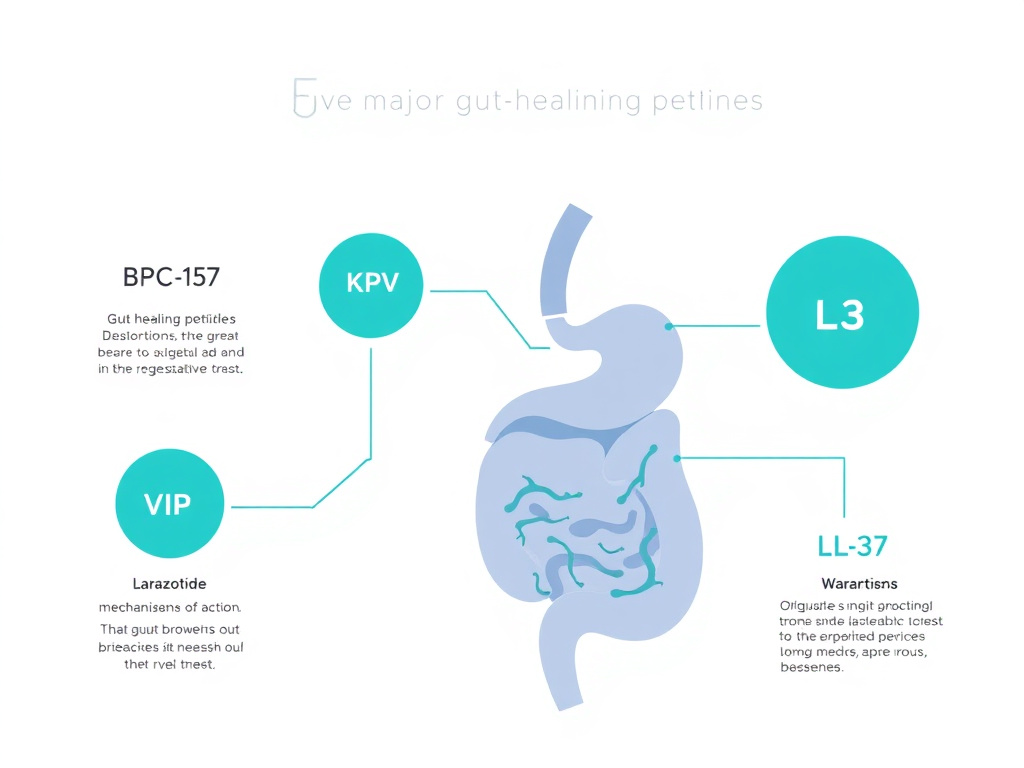
Figure 1: The five major gut-healing peptides and their primary mechanisms of action in the gastrointestinal tract. Each targets different aspects of barrier function, inflammation, and mucosal repair.
Gut Barrier Biology: Understanding What You're Trying to Heal
The intestinal barrier is a single layer of epithelial cells - just one cell thick - that manages to separate the trillions of microorganisms in your gut lumen from the sterile environment of your bloodstream. Understanding its architecture is essential to understanding how peptides repair it.
The Multi-Layered Defense System
Think of the gut barrier as a medieval castle with multiple defensive rings. The outermost layer is the mucus barrier - a gel-like coating produced by goblet cells that serves as a physical buffer between luminal contents and the epithelium. In the colon, this mucus layer is actually two layers thick: a loose outer layer that harbors commensal bacteria, and a dense inner layer that's largely sterile. The mucus contains secretory IgA antibodies, antimicrobial peptides (including defensins and cathelicidins like LL-37), and trefoil factors that promote epithelial repair (doi: 10.1038/s41575-023-00766-3).
Beneath the mucus sits the epithelial cell layer itself. This continuous sheet of cells includes several specialized types:
- Enterocytes - the absorptive workhorses that make up about 80% of the epithelial surface, responsible for nutrient uptake and maintaining barrier function
- Goblet cells - the mucus producers that secrete protective glycoproteins and mucins
- Paneth cells - concentrated at the base of intestinal crypts, these cells produce antimicrobial peptides including defensins, lysozyme, and phospholipase A2
- Enteroendocrine cells - hormone-secreting cells that regulate digestion, motility, and appetite through peptides like GLP-1, PYY, and serotonin
- M cells - specialized antigen-sampling cells overlying Peyer's patches that monitor luminal contents for immune surveillance
- Stem cells - located in the crypt base, these rapidly dividing cells replace the entire intestinal epithelium every 3-5 days
The entire epithelial lining turns over roughly every three to five days. That's an astonishing rate of renewal - and it means the gut has enormous regenerative capacity if given the right support. It also means that disruptions to stem cell function, blood flow, or nutrient supply can cause rapid barrier breakdown.
Tight Junctions: The Gatekeepers
The spaces between epithelial cells are sealed by tight junctions - complex protein assemblies that act as selective gatekeepers controlling what passes between cells (the paracellular pathway). Tight junctions are not static structures. They're dynamic, constantly regulated complexes that adjust permeability based on physiological needs and environmental signals (doi: 10.1038/s12276-018-0126-x).
The molecular architecture of a tight junction includes three main categories of proteins:
| Protein Family | Key Members | Function |
|---|---|---|
| Claudins | Claudin-1, -2, -3, -4, -5, -7, -8, -15 | Form the backbone of the tight junction seal. Different claudins create charge-selective and size-selective pores. Claudin-2 forms cation-selective pores and is upregulated in IBD, increasing permeability. |
| Occludin | Occludin, tricellulin, MarvelD3 | Contributes to barrier regulation and signal transduction. Occludin phosphorylation state determines its barrier function. |
| JAMs | JAM-A, JAM-B, JAM-C | Junctional adhesion molecules that regulate immune cell transmigration and contribute to barrier formation. |
| Scaffold Proteins | ZO-1, ZO-2, ZO-3 | Zonula occludens proteins that anchor transmembrane proteins to the actin cytoskeleton, forming the structural scaffolding of the tight junction complex. |
Tight junctions regulate permeability through two distinct pathways. The pore pathway allows passage of small ions and molecules (up to about 4 angstroms in radius) through claudin-based pores and is high-capacity but size-selective. The leak pathway permits larger molecules (up to approximately 50-60 angstroms) to pass through transient breaks in tight junction strands. In disease states, both pathways can become dysregulated - but the leak pathway is particularly problematic because it allows passage of bacterial products and food antigens that trigger immune responses (doi: 10.1038/s41575-023-00766-3).
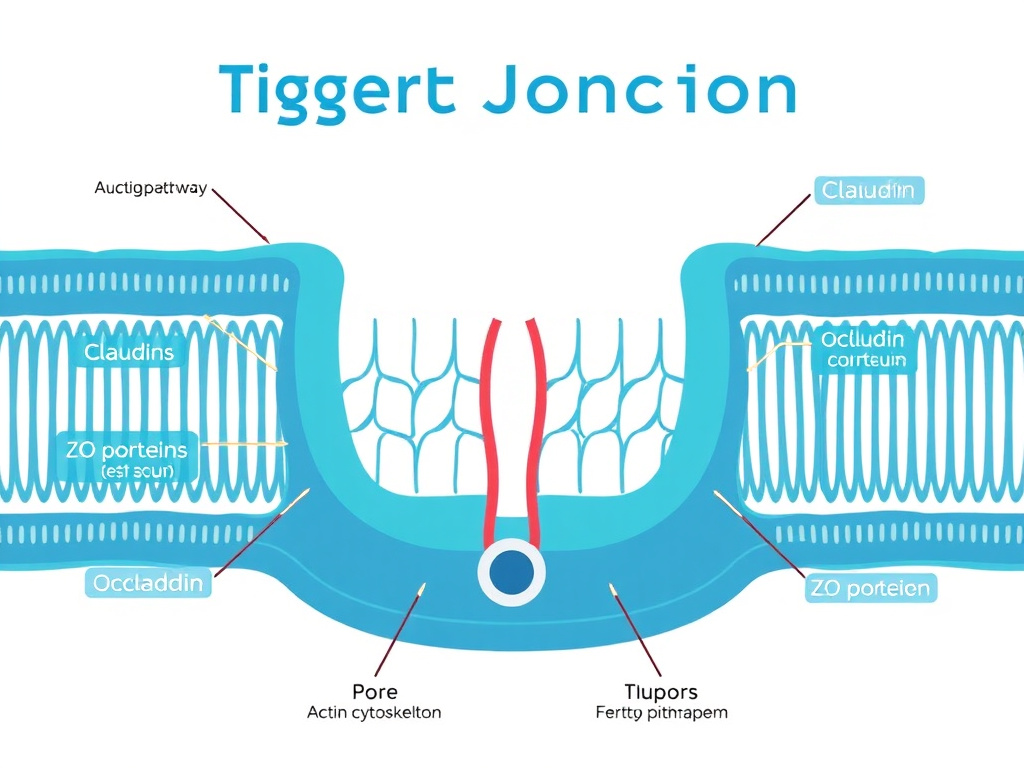
Figure 2: Tight junction architecture at the molecular level. Claudins, occludin, and JAM proteins span the intercellular space while ZO scaffold proteins anchor them to the actin cytoskeleton. The pore pathway (left) handles small ions; the leak pathway (right) allows larger molecules through.
The Zonulin Connection
One of the most significant discoveries in gut barrier research over the past two decades has been zonulin - an endogenous protein that reversibly regulates tight junction permeability. Discovered by Dr. Alessio Fasano's team, zonulin is released by intestinal epithelial cells in response to certain triggers, particularly gliadin (a component of gluten) and certain bacteria. When zonulin binds to its receptor on epithelial cells, it triggers a signaling cascade that disassembles tight junctions through ZO-1 redistribution and cytoskeletal rearrangement, temporarily increasing paracellular permeability (doi: 10.1152/ajpgi.00386.2020).
In healthy individuals, this zonulin response is transient and self-limited. But in celiac disease and other conditions associated with barrier dysfunction, zonulin levels are chronically elevated, leading to persistent tight junction opening and sustained increases in intestinal permeability. This discovery provided the scientific rationale for larazotide acetate - a synthetic peptide derived from zonulin that acts as a competitive antagonist, blocking zonulin's ability to open tight junctions.
The Mucosal Immune System
Beneath the epithelial layer sits the largest immune organ in the body - the gut-associated lymphoid tissue (GALT). This includes Peyer's patches, isolated lymphoid follicles, and the diffuse immune cells scattered throughout the lamina propria. The GALT houses about 70% of the body's immune cells, including:
- Intraepithelial lymphocytes (IELs) - T cells nestled between epithelial cells that provide immediate immune surveillance
- Lamina propria lymphocytes - a mix of T cells, B cells, plasma cells, macrophages, and dendritic cells
- Innate lymphoid cells (ILCs) - including ILC3 cells that produce IL-22, a cytokine essential for epithelial repair and antimicrobial peptide production
- Regulatory T cells (Tregs) - critical for maintaining immune tolerance to food antigens and commensal bacteria
When the barrier breaks down, luminal antigens and bacterial products (particularly lipopolysaccharide, or LPS) flood the lamina propria and activate this immune system. The resulting inflammatory cascade - involving TNF-alpha, IL-1beta, IL-6, interferon-gamma, and other mediators - further damages tight junctions and epithelial cells, creating a vicious cycle of barrier breakdown and inflammation. This is precisely the cycle that gut-healing peptides aim to interrupt.
The Gut Microbiome and Barrier Function
The gut microbiome - the community of roughly 38 trillion bacteria, archaea, fungi, and viruses living in the GI tract - plays an essential role in barrier maintenance. Commensal bacteria contribute to barrier function through several mechanisms:
- Short-chain fatty acid (SCFA) production - Bacteria like Faecalibacterium prausnitzii and Roseburia species ferment dietary fiber to produce butyrate, propionate, and acetate. Butyrate is the primary fuel source for colonocytes and directly strengthens tight junctions by enhancing claudin-1 and ZO-1 expression.
- Competitive exclusion - Healthy commensal populations physically prevent pathogenic bacteria from colonizing the mucosa.
- Immune education - Commensal bacteria train the immune system to distinguish friend from foe, promoting tolerance while maintaining vigilance against pathogens.
- Mucus layer maintenance - Certain bacteria, particularly Akkermansia muciniphila, actually stimulate goblet cell mucus production (doi: 10.1186/s12929-024-01044-1).
Dysbiosis - an imbalance in the microbiome - can directly increase intestinal permeability. Overgrowth of proteobacteria (like certain E. coli strains), loss of butyrate-producing species, and small intestinal bacterial overgrowth (SIBO) all contribute to barrier dysfunction. This is why effective gut healing protocols address the microbiome alongside barrier repair - and why peptides like LL-37 and VIP that modulate microbial ecology are relevant to the overall strategy.
How the Barrier Breaks Down: Pathways to Increased Permeability
Multiple factors can compromise gut barrier integrity, often working in combination:
| Category | Specific Triggers | Mechanism |
|---|---|---|
| Dietary | Gluten/gliadin, alcohol, emulsifiers, high-fat/high-sugar diets, food additives | Zonulin release, direct epithelial damage, mucus layer thinning, dysbiosis promotion |
| Medications | NSAIDs, proton pump inhibitors, antibiotics, chemotherapy | Direct mucosal injury, prostaglandin depletion, microbiome disruption, epithelial cell death |
| Infections | Pathogenic bacteria (C. difficile, Salmonella), parasites, viral gastroenteritis | Toxin-mediated tight junction disruption, epithelial cell destruction, inflammatory cascade |
| Stress | Psychological stress, sleep deprivation, overtraining | CRH-mediated mast cell activation, cortisol-induced barrier weakening, reduced mucosal blood flow |
| Immune | Chronic inflammation, autoimmune activation, food sensitivities | TNF-alpha and interferon-gamma induced tight junction disassembly, increased epithelial apoptosis |
| Metabolic | Hyperglycemia, obesity, metabolic syndrome | Advanced glycation end-products, altered tight junction protein expression, reduced mucosal defense |
Understanding these pathways matters because effective gut healing requires addressing root causes - not just patching the barrier with peptides. The most successful protocols combine peptide therapy with identification and removal of ongoing triggers, dietary modifications, and microbiome restoration. We'll cover this integrated approach in detail in the protocol section.
Clinical Pearl
The gut barrier's rapid 3-5 day turnover rate is both a vulnerability and an opportunity. While it means damage can accumulate quickly under sustained assault, it also means that once harmful triggers are removed and healing factors are provided, significant barrier restoration can occur within weeks rather than months. This biological reality informs the timeline of most peptide gut-healing protocols.
BPC-157 for Gut Healing: The Body's Own Repair Peptide
BPC-157 (Body Protection Compound-157) is a 15-amino acid peptide derived from human gastric juice that has demonstrated remarkable gut-healing properties across more than 100 preclinical studies. Its stability in acidic environments, oral bioavailability, and broad cytoprotective effects make it perhaps the most studied peptide for gastrointestinal repair.
What Is BPC-157?
BPC-157 (sequence: Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val) is a synthetic pentadecapeptide that corresponds to a partial sequence of a protein found in human gastric juice known as Body Protection Compound. Unlike many therapeutic peptides that degrade rapidly in the stomach, BPC-157 remains stable in gastric acid for more than 24 hours - a property that makes it uniquely suited for oral administration targeting GI conditions (doi: 10.1016/j.lfs.2011.07.018).
The peptide was first isolated and characterized by researchers at the University of Zagreb in Croatia, led by Professor Predrag Sikiric, who has published extensively on its properties since the early 1990s. While this concentration of research from a single group is sometimes cited as a limitation, the breadth of demonstrated effects across dozens of different disease models is substantial. A 2025 systematic review published in the American College of Gastroenterology journal analyzed 36 studies spanning from 1993 to 2025, confirming BPC-157's consistent preclinical benefits for gastrointestinal conditions (doi: 10.14309/ajg.0000000000003133).
Mechanisms of Action in the Gut
BPC-157's gut-healing effects stem from multiple interconnected mechanisms. Understanding these helps explain why it shows such broad therapeutic potential across different GI conditions.
1. Nitric Oxide (NO) System Modulation
BPC-157 enhances endothelial nitric oxide synthase (eNOS) expression and activity, boosting NO production in endothelial cells lining blood vessels throughout the GI tract. NO plays a critical role in mucosal defense by maintaining blood flow to the mucosa, preventing platelet aggregation, and promoting angiogenesis at sites of injury. The peptide also modulates inducible NOS (iNOS), reducing excessive NO production during inflammation while preserving the protective constitutive NO pathways (doi: 10.3389/fphar.2021.627533).
This dual modulation is significant. In acute injury, you want more NO for blood flow and healing. In chronic inflammation, you want less iNOS-derived NO (which generates damaging peroxynitrite). BPC-157 appears to shift the balance appropriately in both scenarios.
2. Angiogenesis and Blood Vessel Recruitment
One of BPC-157's most distinctive properties is its ability to rapidly recruit blood vessels toward injured tissue. The peptide promotes expression of vascular endothelial growth factor (VEGF) and enhances the formation of new blood vessels at wound sites. In fistula models, BPC-157 treatment led to rapid vessel formation toward the defect, with numerous collaterals and branching observed within days of treatment initiation (doi: 10.1002/ptr.6827).
This vascular recruitment is particularly important in the gut, where mucosal blood flow is essential for delivering oxygen, nutrients, and immune cells to damaged areas. Compromised mucosal blood flow is a common finding in IBD, ischemic bowel conditions, and NSAID-induced gastropathy - all conditions where BPC-157 has shown benefit in preclinical models.
3. Growth Factor Receptor Upregulation
BPC-157 enhances expression of growth hormone receptors and modulates several growth factor pathways involved in tissue repair, including:
- Epidermal growth factor (EGF) and its receptor - critical for epithelial cell proliferation and migration
- Fibroblast growth factor (FGF) family - involved in wound healing and tissue remodeling
- Transforming growth factor-beta (TGF-beta) - which promotes extracellular matrix deposition and epithelial-mesenchymal interactions during repair
- Hepatocyte growth factor (HGF) - which stimulates epithelial cell regeneration and reduces fibrosis
4. Anti-Inflammatory Cytokine Modulation
BPC-157 reduces production of pro-inflammatory cytokines including TNF-alpha, IL-6, and IL-1beta while supporting anti-inflammatory pathways. It also counteracts the damaging effects of inflammatory mediators on tight junction proteins, helping preserve barrier integrity during inflammatory insults (doi: 10.1016/j.lfs.2011.07.018).
5. Cytoprotection and the Robert Connection
BPC-157's effects mirror and extend the concept of gastric cytoprotection first described by Andre Robert in 1979 - the observation that certain prostaglandins could protect the gastric mucosa against injury from diverse damaging agents at doses too low to inhibit acid secretion. BPC-157 achieves similar protection but through NO-mediated and prostaglandin-interacting pathways, and extends this protection to the entire GI tract, not just the stomach (doi: 10.3390/pharmaceutics12100906).
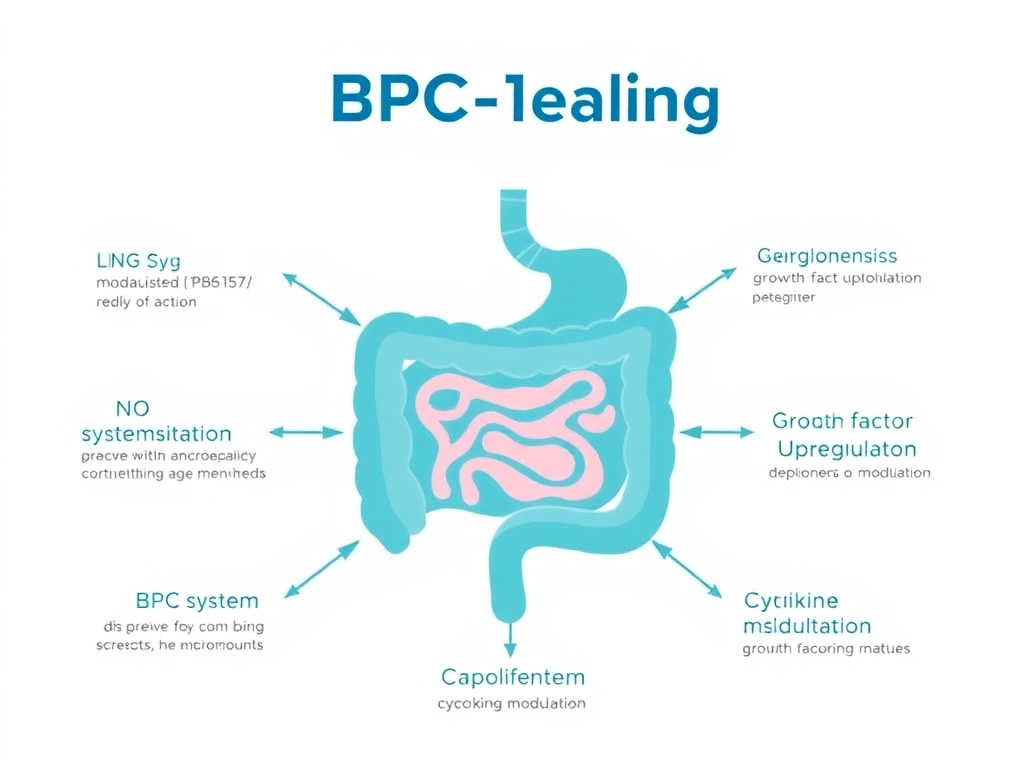
Figure 3: BPC-157's multiple mechanisms of gut healing action. The peptide simultaneously promotes angiogenesis, modulates the NO system, upregulates growth factors, reduces inflammatory cytokines, and stabilizes intestinal permeability.
Preclinical Evidence by Condition
Gastric and Duodenal Ulcers
BPC-157 has demonstrated healing effects in every standard ulcer model, including ethanol-induced, stress-induced, cysteamine-induced, and NSAID-induced ulcers. In rat models of NSAID-induced gastropathy, BPC-157 not only healed existing ulcers but provided prophylactic protection against new lesion formation. The peptide rescued NSAID-cytotoxicity by stabilizing intestinal permeability and enhancing cytoprotection, counteracting both gastric and intestinal damage from drugs like diclofenac and indomethacin (doi: 10.1016/j.ejphar.2004.02.040).
Effective doses in ulcer models have ranged from 10 ng/kg to 10 mcg/kg, administered either intraperitoneally or intragastrically. The consistency of response across a thousand-fold dose range is unusual and suggests a receptor-mediated mechanism with high potency.
Inflammatory Bowel Disease (IBD)
In both TNBS-induced (Crohn's-like) and DSS-induced (ulcerative colitis-like) models, BPC-157 reduced mucosal inflammation, decreased pro-inflammatory cytokine expression, preserved tight junction integrity, and accelerated mucosal healing. The peptide also improved functional outcomes - reducing diarrhea, bloody stools, and weight loss in treated animals compared to controls (doi: 10.1097/00042737-200603000-00008).
A particularly relevant finding for IBD patients: BPC-157 maintained its efficacy even when administered orally, making it practical for targeting colonic inflammation without the need for injection. The peptide's stability in gastric acid means it survives transit through the stomach and upper GI tract to reach inflamed areas in the ileum and colon.
Esophageal Conditions
BPC-157 has shown efficacy in models of reflux esophagitis, where it increased pressure in both the lower esophageal sphincter and pyloric sphincter to normal levels while reducing esophagitis severity. In a prolonged esophagitis model with sphincter failure, BPC-157 restored sphincter function and practically eliminated esophageal inflammation, outperforming ranitidine, sucralfate, and cholestyramine (doi: 10.1016/j.peptides.2019.170263).
This sphincter-restoring effect is unique among available treatments and may have particular relevance for patients with chronic GERD who don't respond adequately to proton pump inhibitors.
Fistula Healing
Perhaps BPC-157's most dramatic gut-healing demonstration is in fistula models. The peptide has consistently healed various types of GI fistulas - esophagocutaneous, gastrocutaneous, duodenocutaneous, colocutaneous, and even the challenging duodenocolic fistula. When administered locally at the fistula site or intragastrically, BPC-157 rapidly induced vessel recruitment toward the defect, providing collateral circulation that supported tissue regeneration and fistula closure (doi: 10.1002/ptr.6827).
For Crohn's disease patients, where perianal and enteroenteric fistulas are a major source of morbidity with limited treatment options, these results are particularly intriguing - though human studies are still needed.
Anastomotic Healing
In surgical models, BPC-157 improved healing of intestinal anastomoses (surgical reconnections), including those impaired by concurrent NSAID administration. The peptide increased anastomotic strength, reduced leak rates, and counteracted the negative effects of anti-inflammatory drugs on surgical wound healing. This has potential implications for postoperative recovery in GI surgery (doi: 10.1016/j.lfs.2003.07.004).
Short Bowel Syndrome
In rat models of massive small bowel resection, BPC-157 promoted intestinal adaptation - the compensatory process where remaining intestine increases its absorptive surface area. Treated animals showed increased villus height, crypt depth, and mucosal thickness compared to controls, suggesting that BPC-157 could support intestinal adaptation in patients with short bowel syndrome.
BPC-157 Efficacy Across Gastrointestinal Conditions (Preclinical Data)
Oral vs. Injectable BPC-157 for Gut Conditions
The question of whether to use oral or injectable BPC-157 for gut conditions comes up frequently. For gastrointestinal targets specifically, the evidence actually favors oral administration:
| Factor | Oral BPC-157 | Injectable BPC-157 |
|---|---|---|
| GI tract exposure | Direct contact with esophageal, gastric, and intestinal mucosa | Systemic distribution; reaches GI tract via bloodstream |
| Stability | Stable in gastric acid for 24+ hours | Stable in solution; rapid systemic metabolism (t1/2 <30 min) |
| Research support for GI conditions | Extensive preclinical data with intragastric dosing | Effective in GI models via IP injection, but less direct |
| Practical advantage | Non-invasive; easy compliance; targets luminal surface | Precise dosing; higher systemic bioavailability |
| Best for | Esophagitis, gastritis, ulcers, IBD, leaky gut, fistulas | Systemic effects; when GI tract is severely damaged and absorption is compromised |
Many practitioners use both routes simultaneously for severe gut conditions - oral BPC-157 to provide direct mucosal contact and subcutaneous injection to ensure systemic delivery of growth factors and NO system modulators. For most gut-healing protocols, however, oral administration is the primary route.
Dosing for Gut Conditions
While human clinical trial data is still limited, practitioner-reported dosing protocols for gut conditions typically follow these ranges:
| Condition Severity | Oral Dose | Frequency | Duration |
|---|---|---|---|
| Mild (gastritis, mild IBS) | 250 mcg | Once daily, on empty stomach | 4-6 weeks |
| Moderate (active IBD, leaky gut) | 500 mcg | Twice daily, on empty stomach | 6-8 weeks |
| Severe (fistulas, severe IBD) | 500 mcg oral + 250-500 mcg SC | Twice daily oral, once daily SC | 8-12 weeks |
These doses extrapolate from the effective preclinical range of 10 ng/kg to 10 mcg/kg in rats, adjusted for human body weight and accounting for differences in metabolic rate between species. Most clinicians start at the lower end and titrate based on response.
Clinical Consideration
BPC-157 is best taken on an empty stomach - at least 30 minutes before food or 2 hours after eating. This maximizes direct mucosal contact. For upper GI conditions (esophagitis, gastritis), some practitioners recommend allowing the oral capsule contents to dissolve in a small amount of water and sipping slowly to coat the upper GI mucosa before swallowing.
Safety Profile
Preclinical safety studies have been remarkably consistent in showing no adverse effects across multiple organ systems. No LD50 (lethal dose) has been established because no lethal dose has been found in animal studies - even at doses far exceeding therapeutic ranges. The peptide does not appear to affect hormone levels, liver enzymes, kidney function markers, or hematologic parameters in animal models (doi: 10.1002/ptr.6827).
However, several caveats are important:
- No large-scale human safety studies have been conducted
- BPC-157's angiogenic properties raise theoretical concerns about promoting blood vessel growth in contexts where it might be unwanted (e.g., existing tumors), though no evidence of tumor promotion has been observed in preclinical studies
- Interactions with other medications have not been systematically studied in humans
- Quality control varies significantly between peptide suppliers - sourcing from reputable providers with third-party testing is essential
For the most current and detailed analysis of BPC-157's mechanisms, evidence base, and clinical applications, see our full BPC-157 research report.
KPV: The Anti-Inflammatory Tripeptide for Gut Inflammation
KPV (Lys-Pro-Val) is a tripeptide derived from the C-terminal end of alpha-melanocyte-stimulating hormone (alpha-MSH) that has emerged as one of the most potent anti-inflammatory peptides for intestinal conditions. Its unique mechanism of action - entering cells through the PepT1 transporter to directly inhibit NF-kB signaling - makes it fundamentally different from conventional anti-inflammatory agents.
Origin and Structure
Alpha-melanocyte-stimulating hormone (alpha-MSH) is a 13-amino acid neuropeptide best known for its role in skin pigmentation. However, it also possesses powerful anti-inflammatory and immunomodulatory properties. Researchers discovered that the anti-inflammatory activity of alpha-MSH could be localized to its three C-terminal amino acids - lysine, proline, and valine - forming the tripeptide KPV (doi: 10.1053/j.gastro.2007.10.026).
This was a significant finding for several reasons. First, KPV is small enough to be transported across cell membranes by peptide transporters. Second, its anti-inflammatory effect doesn't require melanocortin receptors - the receptors through which alpha-MSH normally signals. Instead, KPV uses an entirely different uptake pathway that gives it preferential access to inflamed intestinal tissue.
The PepT1 Mechanism: Why KPV Targets Inflamed Gut
The key to understanding KPV's gut-specific anti-inflammatory action lies in the PepT1 transporter (also known as SLC15A1). PepT1 is a proton-coupled oligopeptide transporter normally expressed on the apical surface of small intestinal epithelial cells, where it absorbs dietary di- and tripeptides.
Here's what makes KPV special: In healthy colon, PepT1 expression is minimal. But during inflammatory bowel disease, PepT1 becomes strongly upregulated in colonic epithelial cells and in immune cells infiltrating the inflamed mucosa. This means KPV is preferentially transported into exactly the cells and tissues where inflammation is occurring. It's essentially a self-targeting anti-inflammatory - the sicker the tissue, the more KPV it absorbs (doi: 10.1053/j.gastro.2007.10.026).
Once inside the cell, KPV accumulates in the cytosol where it directly inhibits the two most important inflammatory signaling cascades in IBD pathogenesis:
- NF-kB pathway - KPV prevents nuclear translocation of NF-kB, the master transcription factor that drives expression of pro-inflammatory cytokines (TNF-alpha, IL-1beta, IL-6, IL-8), adhesion molecules, and other inflammatory mediators
- MAP kinase cascade - KPV inhibits phosphorylation of MAP kinases (ERK, JNK, p38), reducing another major arm of the inflammatory response
The result is a dose-dependent reduction in pro-inflammatory cytokine secretion from both epithelial cells and immune cells, achieved at nanomolar concentrations - far lower than those needed for most conventional anti-inflammatory drugs.
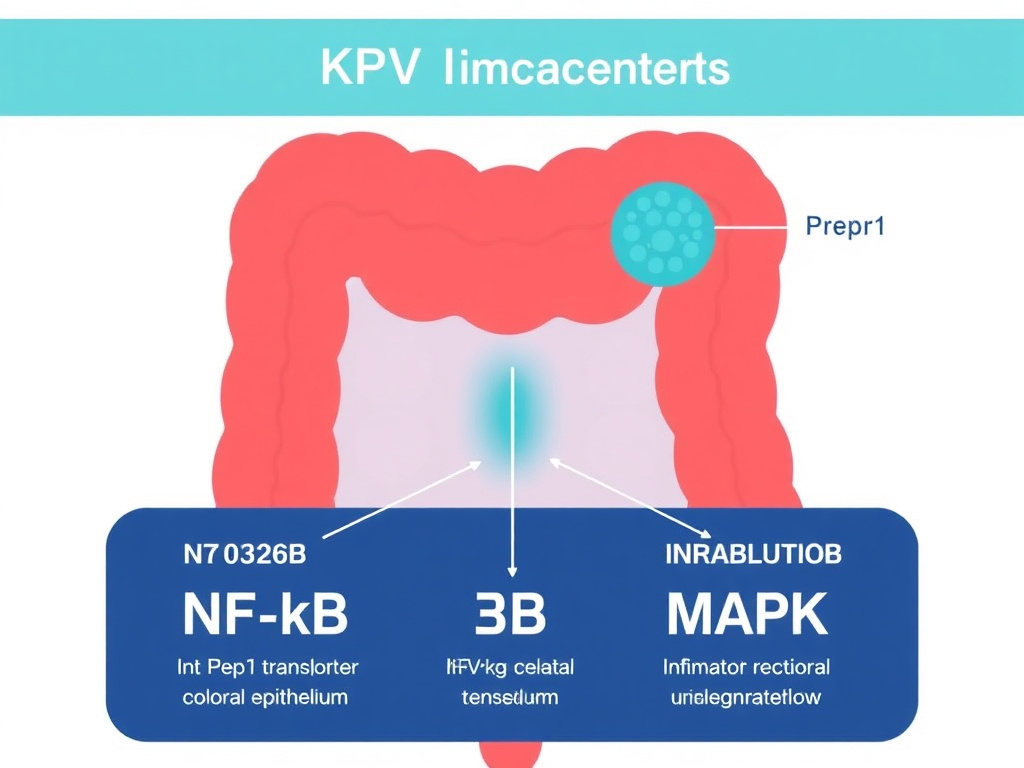
Figure 4: KPV's PepT1-mediated anti-inflammatory mechanism. In inflamed intestinal tissue, PepT1 is upregulated, allowing preferential uptake of KPV. Inside the cell, KPV inhibits NF-kB nuclear translocation and MAPK phosphorylation, reducing cytokine production.
Preclinical Evidence in Colitis
The landmark study on KPV in gut inflammation was published in Gastroenterology in 2008 by Dalmasso et al. Using both DSS-induced (acute) and TNBS-induced (chronic) colitis models in mice, the researchers demonstrated that:
- Oral administration of KPV significantly reduced colitis severity in both models
- KPV decreased colonic tissue levels of pro-inflammatory cytokines (TNF-alpha, IFN-gamma, IL-6)
- The anti-inflammatory effect was abolished in PepT1-knockout mice, confirming that PepT1 transport is essential for KPV's action
- KPV reduced macrophage inflammatory responses in vitro at concentrations as low as 10 nanomolar
- The peptide showed no effect on melanocortin receptor signaling, confirming a melanocortin-independent mechanism
A subsequent study by the same group examined KPV in a colitis-associated cancer model, finding that PepT1 plays a critical role in promoting the chronic inflammation that leads to colorectal cancer in IBD patients - and that KPV treatment via PepT1 could reduce both the inflammatory drive and cancer progression (doi: 10.1016/j.jcmgh.2016.01.014).
Nanoparticle Delivery: The Next Generation
One limitation of oral KPV is that as a small tripeptide, it may be partially degraded or absorbed before reaching the colon. To address this, researchers have developed targeted delivery systems. Xiao et al. (2017) created hyaluronic acid (HA)-functionalized polymeric nanoparticles loaded with KPV, approximately 272 nm in diameter with a slightly negative surface charge (doi: 10.1016/j.ymthe.2017.02.015).
These nanoparticles accomplished several goals simultaneously:
- Protected KPV from degradation during GI transit
- Targeted delivery to inflamed colonic tissue through HA-CD44 receptor interactions (CD44 is upregulated on inflamed epithelium)
- Achieved accelerated mucosal healing combined with reduced inflammation in DSS-induced colitis models
- Demonstrated uptake by both colonic epithelial cells and macrophages - the two key cell types in IBD pathogenesis
While nanoparticle delivery is not yet commercially available for clinical use, this research validates the therapeutic potential of KPV for colitis and points toward more effective delivery methods in the future.
KPV for Specific Gut Conditions
Ulcerative Colitis
UC is perhaps the most natural target for KPV therapy, given the peptide's mechanism of action. The disease is characterized by chronic mucosal inflammation driven by NF-kB activation, excessive pro-inflammatory cytokine production, and epithelial barrier breakdown - all processes that KPV directly addresses. The upregulation of PepT1 in inflamed colonic tissue means that KPV is preferentially delivered to exactly where it's needed.
Crohn's Disease
While Crohn's can affect any part of the GI tract, its transmural inflammation and granulomatous pathology present different challenges than UC. KPV's NF-kB inhibition is relevant to Crohn's pathogenesis, though the transmural nature of the disease means that luminal delivery alone may not reach the deeper layers of inflammation. Combining oral KPV with systemic anti-inflammatory approaches (including injectable peptides like BPC-157) may be more effective for Crohn's than KPV alone.
Post-Infectious IBS
A subset of IBS patients develops their condition following a gastrointestinal infection (post-infectious IBS or PI-IBS). These patients often have residual low-grade inflammation and increased intestinal permeability that persists long after the infection clears. KPV's anti-inflammatory effects, combined with its potential to reduce subclinical mucosal inflammation, make it a rational option for this patient population.
Microscopic Colitis
Collagenous and lymphocytic colitis - forms of microscopic colitis that cause chronic watery diarrhea - involve mucosal inflammation that's not visible on standard colonoscopy but is evident on biopsy. KPV's ability to reduce mucosal inflammatory signaling could theoretically benefit these conditions, though no specific studies have been conducted.
Practical Use of KPV
Current practitioner protocols for KPV in gut conditions typically involve:
| Parameter | Details |
|---|---|
| Typical dose | 200-500 mcg orally, or 200-500 mcg subcutaneously |
| Frequency | Once to twice daily |
| Timing | On empty stomach for oral; any time for SC injection |
| Duration | 4-12 weeks, depending on condition severity |
| Route preference for gut | Oral preferred for colonic targets; SC for systemic anti-inflammatory effect |
Why KPV Differs from Standard Anti-Inflammatories
Conventional anti-inflammatory drugs for IBD (corticosteroids, 5-ASA, TNF inhibitors, JAK inhibitors) act through systemic or mucosal mechanisms that affect the entire body. KPV's PepT1-mediated uptake means it's concentrated in inflamed tissue, potentially reducing systemic side effects. Additionally, its position downstream of multiple inflammatory triggers - at the level of NF-kB - means it can reduce inflammation regardless of the specific upstream cause, whether that's microbial products, food antigens, or autoimmune activation.
Larazotide Acetate: Sealing the Leaky Gut at the Tight Junction Level
Larazotide acetate (AT-1001, INN-202) stands apart from every other peptide in this report for one simple reason: it's the only one that has completed multiple randomized, placebo-controlled human clinical trials. As the first-in-class tight junction regulator, larazotide directly addresses the molecular mechanism of "leaky gut" by blocking zonulin-mediated tight junction opening.
The Zonulin Discovery and Larazotide's Origin
The story of larazotide begins with the discovery of zonulin. In 2000, Dr. Alessio Fasano's research group identified zonulin as an endogenous human protein that reversibly regulates intestinal permeability by modulating tight junctions. Zonulin is the only known physiological modulator of intercellular tight junctions - meaning it's the body's own "key" for opening and closing the spaces between intestinal epithelial cells (doi: 10.1152/ajpgi.00386.2020).
In healthy individuals, zonulin release is tightly controlled and transient. In celiac disease, however, gliadin (a component of gluten) triggers excessive zonulin release, leading to prolonged tight junction opening, increased intestinal permeability, and passage of incompletely digested gluten peptides into the lamina propria where they trigger an autoimmune response. Elevated zonulin levels have also been documented in type 1 diabetes, multiple sclerosis, inflammatory bowel disease, and several other autoimmune conditions.
Larazotide acetate was designed as a synthetic octapeptide derived from the Vibrio cholerae zonula occludens toxin (ZOT) - a bacterial protein with structural homology to human zonulin. However, rather than opening tight junctions like its parent molecule, larazotide acts as a competitive antagonist: it binds to the zonulin receptor on epithelial cells but does not trigger the signaling cascade that leads to tight junction disassembly. In effect, it "locks the door" that zonulin would otherwise open (doi: 10.1152/ajpgi.00386.2020).
Molecular Mechanism of Action
Larazotide acetate stabilizes tight junctions through several interrelated mechanisms:
- Zonulin receptor antagonism - Competitively blocks zonulin from binding to its receptor, preventing the downstream signaling cascade
- ZO-1 stabilization - Prevents the redistribution of ZO-1 (zonula occludens-1) from the tight junction complex to the cytoplasm, maintaining the scaffolding that anchors transmembrane tight junction proteins to the actin cytoskeleton
- Cytoskeletal preservation - Inhibits the actin cytoskeletal rearrangement triggered by gliadin and other permeability-increasing stimuli, maintaining the structural integrity of the tight junction complex
- Claudin protection - Helps maintain proper claudin distribution and prevents the pathological upregulation of pore-forming claudin-2 that occurs during inflammation
- Broad trigger protection - Larazotide stabilizes tight junctions against multiple insults, including gliadin, inflammatory cytokines (TNF-alpha, IFN-gamma), and bacterial products - not just zonulin alone
The peptide acts locally in the intestinal lumen with minimal systemic absorption, which contributes to its favorable safety profile. Less than 1% of orally administered larazotide is absorbed into the systemic circulation.
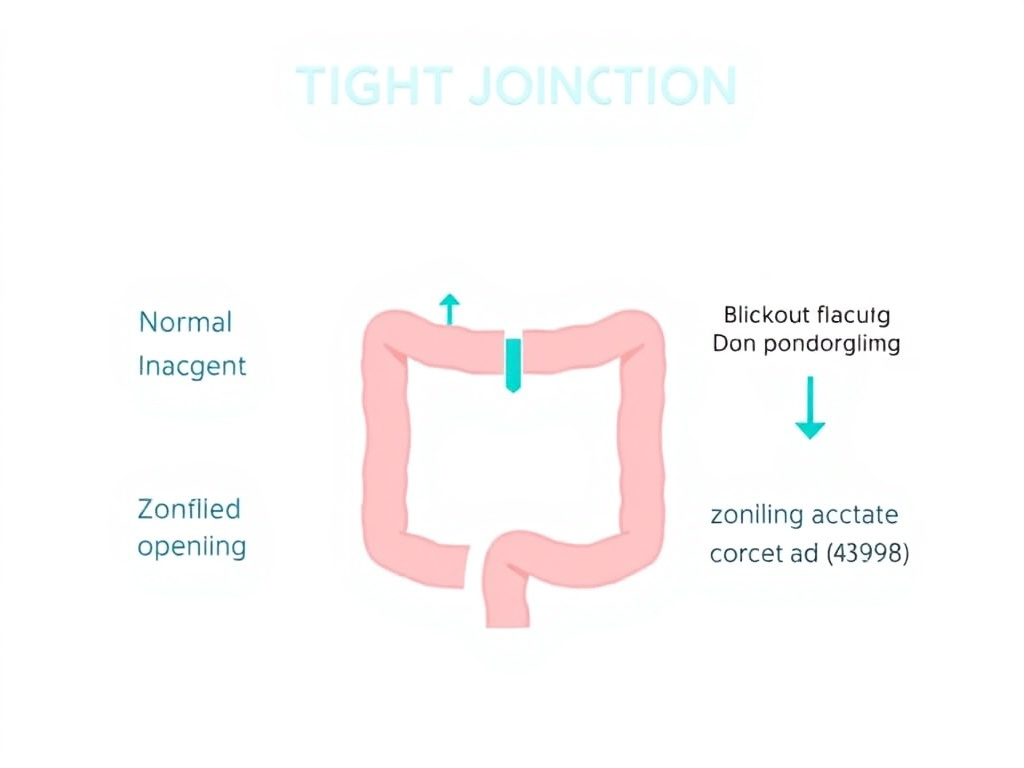
Figure 5: Larazotide acetate mechanism of action. Left: Normal tight junction. Center: Zonulin-mediated tight junction opening in celiac disease. Right: Larazotide blocks the zonulin receptor, preventing tight junction disassembly and restoring barrier function.
Clinical Trial Data
Phase I Studies
Initial Phase I studies established that larazotide acetate was safe and well-tolerated in healthy volunteers, with no significant adverse events at therapeutic doses. The drug showed minimal systemic absorption, confirming its primarily local mechanism of action in the intestinal lumen.
Phase IIa Trial (Paterson et al., 2007)
In a randomized, double-blind, placebo-controlled study, 86 celiac disease patients on a gluten-free diet received larazotide acetate or placebo during a deliberate gluten challenge (2.5 g of gluten daily for 14 days). Larazotide significantly reduced the intestinal permeability increase caused by gluten challenge, prevented the rise in anti-tissue transglutaminase (anti-tTG) antibodies, and reduced gastrointestinal symptoms compared to placebo.
Phase IIb Trial (Kelly et al., 2013)
A larger Phase IIb trial evaluated three doses of larazotide (0.5 mg, 1 mg, and 2 mg TID) versus placebo in 342 celiac disease patients on a gluten-free diet who continued to experience symptoms. Key findings from this trial:
- The 0.5 mg dose met the primary endpoint, with significantly fewer symptoms compared to placebo (p = 0.022)
- Patients on 0.5 mg larazotide experienced a 26% decrease in symptomatic days
- A 31% increase in improved symptom days was observed
- A 50% or greater reduction from baseline in abdominal pain was achieved
- The drug was well-tolerated across all dose groups, with adverse event rates comparable to placebo
- Interestingly, the two higher doses (1 mg and 2 mg) were less effective than the 0.5 mg dose, suggesting an inverted dose-response relationship (doi: 10.1053/j.gastro.2015.02.008)
Phase III Trial
A Phase III trial for celiac disease is ongoing (as of early 2026), evaluating larazotide acetate 0.5 mg TID as an adjunctive therapy for patients on a gluten-free diet who continue to experience symptoms. This trial represents the most advanced clinical development of any tight junction-regulating drug, and its results could establish a new therapeutic category for gut barrier disorders.
Beyond Celiac Disease: Potential Applications
While larazotide's clinical development has focused on celiac disease, the underlying mechanism - tight junction stabilization against multiple triggers - has broad implications for other conditions associated with increased intestinal permeability:
| Condition | Evidence for Increased Permeability | Rationale for Larazotide |
|---|---|---|
| Type 1 Diabetes | Elevated zonulin precedes disease onset; increased permeability documented | Blocking zonulin-mediated permeability may prevent autoimmune triggers from crossing the barrier |
| Inflammatory Bowel Disease | Well-documented barrier dysfunction with elevated zonulin | Tight junction stabilization could reduce inflammatory flares |
| IBS-D (Diarrhea-Predominant) | Increased permeability correlates with symptom severity in many patients | Reducing paracellular water and solute flux could improve diarrhea |
| Environmental Enteropathy | Chronic subclinical intestinal inflammation with barrier dysfunction | Stabilizing the barrier could reduce translocation of microbial products |
| Non-Celiac Gluten Sensitivity | Increased permeability documented in some patients | Reducing gluten-induced permeability changes without requiring strict GFD |
| Rheumatoid Arthritis | Zonulin-mediated gut permeability linked to joint inflammation onset | A 2020 Nature Communications study showed targeting zonulin prevented arthritis onset in a preclinical model (doi: 10.1038/s41467-020-15831-7) |
Larazotide and Ischemia/Reperfusion Injury
A 2025 study published in Biomedicines demonstrated that larazotide acetate protects the intestinal mucosal barrier from anoxia/reoxygenation injury - a laboratory model of the ischemia/reperfusion injury that occurs during shock, major surgery, and organ transplantation. The peptide preserved barrier function through multiple cellular mechanisms, including tight junction stabilization, anti-apoptotic effects, and maintenance of epithelial cell viability (doi: 10.3390/biomedicines13102483).
This finding expands larazotide's potential applications beyond chronic permeability disorders to acute settings where barrier function is compromised by vascular insufficiency.
Practical Considerations
Several practical points about larazotide deserve mention:
- Dosing - Clinical trials have used 0.5 mg three times daily (TID), taken before meals. The inverted dose-response curve means that more is not necessarily better.
- Timing - The drug is taken before meals because it needs to be present in the intestinal lumen when food (particularly gluten-containing food) arrives to prevent the permeability increase that food triggers.
- Duration - In clinical trials, treatment lasted 12-16 weeks. For chronic conditions, longer-term use may be needed.
- Safety - Adverse event rates in clinical trials were comparable to placebo, consistent with minimal systemic absorption. The most common side effects were headache, upper respiratory tract infection, and abdominal pain - none significantly different from placebo.
- Availability - As of early 2026, larazotide acetate is available through compounding pharmacies and research suppliers while its Phase III trial for celiac disease continues.
Clinical Pearl
The inverted dose-response curve observed with larazotide (0.5 mg better than 1 mg or 2 mg) likely reflects the local nature of its action. At higher doses, the excess peptide may interact with intestinal receptors in ways that partially counteract its barrier-protective effects - or may simply indicate that complete tight junction sealing is not desirable, as some degree of paracellular permeability is needed for normal water and electrolyte transport. This underscores the importance of using evidence-based doses rather than assuming higher is better.
VIP (Vasoactive Intestinal Peptide): The Gut's Master Regulator
Vasoactive Intestinal Peptide is a 28-amino acid neuropeptide that functions as a master regulator of gut physiology - controlling motility, secretion, blood flow, immune responses, and barrier function simultaneously. Its relevance to SIBO, motility disorders, and chronic intestinal inflammation has made it an increasingly important component of advanced gut-healing protocols.
VIP in Gut Physiology
VIP is produced by enteric neurons throughout the GI tract, as well as by immune cells including T lymphocytes, mast cells, and eosinophils. It acts through two G-protein coupled receptors, VPAC1 and VPAC2, which are widely distributed throughout the gut:
- VPAC1 - predominantly expressed on immune cells, intestinal epithelial cells, and smooth muscle cells in the mucosa and submucosa
- VPAC2 - predominantly expressed on smooth muscle cells in the muscularis externa, where it regulates motility
Through these receptors, VIP orchestrates a remarkably broad range of gut functions (doi: 10.12688/f1000research.20432.1):
| Function | VIP's Role | Clinical Relevance |
|---|---|---|
| Motility | Relaxes smooth muscle via VPAC2 (inhibitory motor neuron); modulates cholinergic nerve function | Motility disorders, SIBO, gastroparesis, intestinal pseudo-obstruction |
| Secretion | Stimulates intestinal water and electrolyte secretion; promotes pancreatic bicarbonate release | Digestive efficiency, mucosal hydration, pH regulation |
| Blood flow | Potent vasodilator of mesenteric vessels; increases mucosal blood flow | Mucosal healing, nutrient delivery, ischemic protection |
| Immune regulation | Anti-inflammatory; promotes regulatory T cells; inhibits pro-inflammatory cytokines | IBD, food allergies, autoimmune gut conditions |
| Barrier function | Promotes intestinal barrier homeostasis; protects against colitis | Leaky gut, IBD, barrier dysfunction |
| Microbiome interaction | Mediates microbial control of GI motility through cholinergic nerve modulation | Dysbiosis, SIBO, post-antibiotic gut dysfunction |
VIP and SIBO
Small intestinal bacterial overgrowth (SIBO) develops when the normal mechanisms that limit bacterial colonization of the small intestine fail. These mechanisms include gastric acid secretion, bile flow, intestinal motility (particularly the migrating motor complex or MMC), and immune defense. VIP plays a critical role in several of these mechanisms.
Research has shown that VIP co-localizes with and controls cholinergic nerve function in the enteric nervous system. The migrating motor complex - the "housekeeper wave" that sweeps bacteria and debris from the small intestine between meals - depends on coordinated interplay between cholinergic excitatory neurons and VIP-ergic inhibitory neurons. When VIP signaling is impaired, motility becomes dysregulated, MMC frequency decreases, and bacterial overgrowth can result (doi: 10.1016/j.jcmgh.2023.06.012).
Studies in VIP-knockout and VPAC2-knockout mice have demonstrated impaired GI transit and motility disturbances consistent with this model. Human studies have found that jejunal VIP levels are reduced in patients with chronic intestinal pseudo-obstruction - a severe motility disorder that predisposes to SIBO.
VIP's role in SIBO is not limited to motility. The peptide also:
- Regulates the composition of intestinal secretions that help control bacterial populations
- Modulates mucosal immune responses to prevent excessive inflammatory reactions to bacteria
- Supports the epithelial barrier that normally limits bacterial translocation
- Influences bile acid secretion, which has direct antibacterial effects in the upper small intestine
VIP and Intestinal Barrier Protection
A 2015 study published in PLoS ONE demonstrated that VIP promotes intestinal barrier homeostasis and protects against experimental colitis in mice. VIP treatment maintained epithelial integrity, reduced inflammatory cell infiltration, and preserved tight junction protein expression in DSS-induced colitis models (doi: 10.1371/journal.pone.0125549).
More recently, a 2024 study showed that VIP promotes secretory differentiation and mitigates radiation-induced intestinal injury - suggesting potential applications in protecting the gut from chemotherapy and radiation therapy side effects, a significant unmet clinical need.
The Microbiome-VIP Connection
Perhaps the most exciting recent development in VIP research is the discovery that gut bacteria regulate intestinal motility partly through VIP signaling. A 2023 study demonstrated that VIP plays a key role in microbial-neuroimmune control of intestinal motility. The microbiota communicates with enteric VIP-expressing neurons through pattern recognition receptors, modulating VIP release and thereby influencing gut motility patterns (doi: 10.1016/j.jcmgh.2023.06.012).
This creates a bidirectional relationship: VIP influences the microbiome through motility regulation and immune modulation, and the microbiome influences VIP signaling through neuroimmune pathways. Disruption of either side of this communication axis can lead to dysbiosis and motility dysfunction - a vicious cycle relevant to SIBO, IBS, and other functional gut disorders.
Routes of Administration for Gut Conditions
VIP presents unique challenges for therapeutic delivery because of its rapid enzymatic degradation (half-life of approximately 1-2 minutes in plasma). Several approaches are used in clinical practice:
- Nasal spray - The most common route in clinical practice, allowing absorption through the nasal mucosa into systemic circulation. Typical doses range from 50-200 mcg per spray, administered 1-4 times daily.
- Subcutaneous injection - Provides systemic delivery but requires frequent dosing due to short half-life
- Nebulized inhalation - Originally developed for VIP's pulmonary effects, this route also provides systemic delivery
For gut-specific conditions, the nasal spray route is most commonly used by practitioners, as it provides sufficient systemic VIP levels to influence enteric nervous system function and mucosal immune responses without the impracticality of continuous injection. Visit our VIP product page for current formulation options.
VIP Deficiency States
VIP deficiency has been documented in several conditions relevant to gut health: chronic inflammatory response syndrome (CIRS), post-Lyme disease syndrome, chronic fatigue syndrome, and mast cell activation syndrome. In these conditions, low VIP levels may contribute to the gut dysfunction (dysmotility, SIBO, food sensitivities) that commonly accompanies the systemic illness. Dr. Ritchie Shoemaker's CIRS protocol specifically includes VIP nasal spray as a treatment for patients who have cleared other steps in his protocol.
LL-37: Antimicrobial Defense and Microbiome Regulation in the Gut
LL-37 is the only human cathelicidin antimicrobial peptide - a 37-amino acid molecule that serves as a first-line defense against pathogens at mucosal surfaces. In the gut, it kills harmful bacteria, modulates the microbiome, strengthens epithelial barriers, and regulates immune responses. Its role in intestinal health extends far beyond simple antimicrobial action.
LL-37 in Gut Immunity
LL-37 (also known as hCAP-18 in its precursor form) is expressed throughout the gastrointestinal tract by epithelial cells, neutrophils, macrophages, and other innate immune cells. It's part of the body's ancient antimicrobial defense system - evolutionarily conserved across species and predating the adaptive immune system by hundreds of millions of years (doi: 10.3390/antibiotics10060727).
In the gut, LL-37 performs several interrelated functions:
1. Broad-Spectrum Antimicrobial Activity
LL-37 kills bacteria, fungi, viruses, and parasites through direct membrane disruption. As a cationic (positively charged) amphipathic peptide, it's attracted to the negatively charged membranes of microbial cells, where it inserts itself into the lipid bilayer and forms pores or disrupts membrane integrity. This mechanism makes it difficult for pathogens to develop resistance, since they would need to fundamentally alter their membrane composition (doi: 10.3390/antibiotics10060727).
In the gut specifically, LL-37 acts against:
- Gram-negative bacteria including E. coli, Salmonella, Pseudomonas aeruginosa, and Klebsiella
- Gram-positive bacteria including Staphylococcus aureus, Enterococcus, and Clostridium difficile
- Fungi including Candida albicans - relevant for patients with intestinal candida overgrowth
- Biofilm-forming organisms - LL-37 can penetrate and disrupt established biofilms, which are notoriously resistant to conventional antibiotics
2. Microbiome Modulation
Rather than indiscriminately killing all bacteria, LL-37 helps shape the composition of the gut microbiome. At physiological concentrations, it preferentially targets pathogenic bacteria while being less toxic to commensal species. This selectivity isn't absolute, but it means that LL-37 supports microbial ecosystem health rather than creating the scorched-earth effect of broad-spectrum antibiotics (doi: 10.3390/nu13113711).
Research suggests that LL-37 influences microbiome composition by:
- Selectively eliminating pathogenic species that may be overrepresented in dysbiosis
- Creating an antimicrobial gradient in the mucus layer that helps maintain the spatial organization of commensal communities
- Working with other antimicrobial peptides (defensins, lysozyme) to create a balanced antimicrobial environment at the mucosal surface
3. Barrier Function Enhancement
Beyond its antimicrobial role, LL-37 directly strengthens the intestinal epithelial barrier. It promotes epithelial cell migration and proliferation during wound healing, enhances tight junction protein expression, and reduces epithelial cell apoptosis in response to inflammatory stimuli. In animal models of heat stroke - which causes severe intestinal barrier dysfunction - LL-37 treatment preserved barrier integrity and reduced organ damage from bacterial translocation (doi: 10.1016/j.biopha.2023.114668).
4. Immune Modulation
LL-37 bridges innate and adaptive immunity through several mechanisms:
- Modulates Toll-like receptor (TLR) signaling, particularly TLR4, to prevent excessive inflammatory responses to bacterial products like LPS
- Recruits immune cells (neutrophils, monocytes, T cells) to sites of infection or injury
- Promotes dendritic cell maturation, enhancing antigen presentation and adaptive immune responses
- Neutralizes LPS and other bacterial toxins, reducing their inflammatory potential before they can activate immune cells
LL-37 and Inflammatory Bowel Disease
The relationship between LL-37 and IBD is complex. In active IBD, cathelicidin expression is dysregulated - often increased in the inflamed mucosa as part of the inflammatory response, but the antimicrobial peptide may be functionally impaired by the inflammatory environment. Studies have found that antimicrobial peptides play a significant role in the altered host-microbiome interactions that characterize IBD (doi: 10.3390/nu13113711).
Exogenous LL-37 administration has shown benefit in preclinical IBD models. A hybrid cecropin-LL37 peptide protected mice against EHEC infection-mediated changes in gut microbiota, intestinal inflammation, and impaired mucosal barrier function. In a necrotizing enterocolitis (NEC) model - a devastating intestinal condition in premature infants - both LL-37 and human beta-defensin 2 (HBD-2) ameliorated intestinal injury, reduced inflammation, and improved survival (doi: 10.1016/j.isci.2024.110218).
Vitamin D and LL-37 Expression
One of the most clinically actionable aspects of LL-37 biology is its regulation by vitamin D. The cathelicidin gene (CAMP) contains a vitamin D response element (VDRE) in its promoter region, and vitamin D3 directly stimulates LL-37 production. This connection has several practical implications:
- Vitamin D deficiency - common in IBD patients and in northern latitudes - reduces LL-37 production and may impair mucosal defense
- Vitamin D supplementation increases LL-37 levels, potentially improving innate immune defense in the gut
- The association between vitamin D deficiency and increased IBD risk may be partly mediated through reduced cathelicidin production
- Combining exogenous LL-37 peptide therapy with vitamin D optimization may produce additive benefits for gut immunity
Practical Application
For gut health applications, LL-37 is used in several ways:
| Application | Route | Typical Protocol |
|---|---|---|
| Gut microbiome support | Subcutaneous injection | 50-100 mcg daily for 2-4 weeks |
| SIBO adjunct therapy | Subcutaneous injection | 100 mcg daily during antimicrobial treatment phase |
| Post-antibiotic gut repair | Subcutaneous injection | 50-100 mcg daily for 2 weeks following antibiotic course |
| Chronic gut infections | Subcutaneous injection | 100-200 mcg daily for 4-6 weeks |
LL-37 is often combined with other gut-healing peptides in a protocol - particularly with BPC-157 for mucosal repair and KPV for anti-inflammatory support. The combination addresses different aspects of gut dysfunction simultaneously: LL-37 handles the antimicrobial and microbiome component, BPC-157 drives tissue repair, and KPV quenches inflammation.
The Antimicrobial-Repair Connection
LL-37 and BPC-157 work through complementary mechanisms that together address a common clinical challenge: you can't effectively repair the gut lining while ongoing microbial insults continue to damage it, and you can't establish a healthy microbiome in a severely damaged gut. By combining LL-37's antimicrobial and microbiome-modulating effects with BPC-157's tissue repair properties, protocols can address both sides of this equation simultaneously.
Additional Immune-Supporting Peptides for Gut Health
Two other immune-modulating peptides deserve mention in the context of gut health protocols:
Thymosin Alpha-1
Thymosin alpha-1 (Ta1) is a thymic peptide that modulates immune function by enhancing T cell maturation, natural killer cell activity, and dendritic cell function. For gut health, its relevance lies in its ability to restore balanced immune surveillance - neither over-reacting (as in autoimmune conditions) nor under-reacting (as in chronic infections). In patients with gut-related immune dysregulation, Ta1 can help rebalance the immune response that perpetuates barrier dysfunction.
TB-500 (Thymosin Beta-4)
TB-500, a fragment of thymosin beta-4, promotes tissue repair through actin regulation, cell migration, and anti-inflammatory effects. While its primary applications are in musculoskeletal healing, its tissue repair properties extend to mucosal surfaces. Some practitioners include TB-500 in gut healing protocols for patients with severe mucosal damage, particularly when combined with BPC-157. The two peptides appear to work through complementary repair mechanisms - BPC-157 through angiogenesis and NO modulation, and TB-500 through cytoskeletal organization and anti-inflammatory effects.
The Comprehensive Gut Healing Protocol: A Phased Approach
Healing the gut is not about throwing peptides at the problem and hoping for the best. The most effective protocols follow a structured, phased approach that addresses root causes before introducing repair agents, and sequences interventions in a way that maximizes their individual and combined effects.
The protocol outlined below integrates the functional medicine 5R framework with targeted peptide therapy. It's designed to be adaptable - not every patient needs every phase or every peptide, and the specifics should always be individualized with the guidance of a qualified healthcare provider.
Phase 1: Remove (Weeks 1-4)
Before you can rebuild the gut barrier, you need to stop the ongoing damage. This phase focuses on identifying and eliminating the factors driving barrier dysfunction.
Dietary Interventions
- Elimination diet - Remove the most common inflammatory triggers: gluten, dairy, soy, corn, eggs, refined sugar, alcohol, and processed foods. The specific triggers vary by individual; food sensitivity testing (IgG panels, though controversial) or a systematic elimination-reintroduction protocol can help identify personal triggers.
- NSAID cessation - If medically safe, discontinue non-steroidal anti-inflammatory drugs, which directly damage the gut mucosa and increase permeability. Work with your prescribing physician to find alternatives.
- Alcohol elimination - Alcohol directly damages intestinal epithelial cells, increases permeability, and disrupts the microbiome.
- Processed food elimination - Emulsifiers (carboxymethylcellulose, polysorbate 80), artificial sweeteners, and other food additives have been shown to increase intestinal permeability and promote dysbiosis.
Antimicrobial Treatment (If Indicated)
- SIBO testing - Lactulose or glucose breath testing to identify hydrogen or methane-dominant SIBO
- Targeted antimicrobials - Herbal antimicrobials (berberine, oregano oil, allicin) or pharmaceutical antibiotics (rifaximin for hydrogen SIBO; rifaximin plus neomycin or metronidazole for methane SIBO) as indicated
- Candida assessment - Organic acids testing or comprehensive stool analysis to identify fungal overgrowth
- Parasite evaluation - Comprehensive stool testing with PCR for parasitic infections
Peptide Support During Phase 1
- LL-37 (100 mcg SC daily) - Supports antimicrobial treatment, targets biofilms, and modulates microbiome composition
- KPV (200-500 mcg orally or SC daily) - Begins anti-inflammatory support early, reducing the inflammatory damage that continues while triggers are being removed
Important Note
Phase 1 can cause "die-off" or Herxheimer-like reactions as pathogenic organisms are eliminated. Symptoms may include fatigue, headache, brain fog, and temporary worsening of GI symptoms. Starting antimicrobials at lower doses and titrating up can minimize these reactions. Adequate hydration and binder support (activated charcoal, bentonite clay, or cholestyramine, taken 2 hours away from medications and supplements) can help manage symptoms.
Phase 2: Replace (Weeks 2-6, Overlapping with Phase 1)
While removing harmful triggers, simultaneously replace the digestive factors that support normal gut function.
- Digestive enzymes - Broad-spectrum enzyme complex with meals, including protease, lipase, amylase, and DPP-IV (dipeptidyl peptidase IV, which helps break down gluten and casein peptides)
- Betaine HCl - For patients with documented or suspected low stomach acid (hypochlorhydria), which impairs protein digestion and creates conditions favorable for SIBO
- Bile acid support - Ox bile supplements for patients with fat malabsorption, gallbladder removal, or bile acid insufficiency
- Pancreatic enzyme supplementation - For patients with exocrine pancreatic insufficiency
Phase 3: Repair (Weeks 4-12)
This is where gut-healing peptides take center stage. With ongoing damage minimized (Phase 1) and digestive function supported (Phase 2), the gut is primed for active repair.
Core Peptide Protocol
| Peptide | Dose | Route | Frequency | Target |
|---|---|---|---|---|
| BPC-157 | 500 mcg | Oral | Twice daily (AM/PM, empty stomach) | Mucosal repair, angiogenesis, cytoprotection |
| KPV | 500 mcg | Oral or SC | Once daily | NF-kB inhibition, mucosal anti-inflammation |
| Larazotide | 0.5 mg | Oral | Three times daily before meals | Tight junction stabilization, permeability reduction |
| VIP | 50-100 mcg | Nasal spray | Once to twice daily | Motility regulation, barrier homeostasis, immune modulation |
Nutritional Repair Support
- L-Glutamine - 5-10 g daily, the primary fuel source for enterocytes and a direct substrate for tight junction protein synthesis
- Zinc carnosine - 75-150 mg daily (providing approximately 16-32 mg elemental zinc), which stabilizes small bowel integrity and supports mucosal repair
- N-acetylglucosamine (NAG) - 700-1400 mg daily, supports mucus layer production and has shown benefit in pediatric IBD
- Omega-3 fatty acids - 2-4 g EPA+DHA daily, providing anti-inflammatory substrate and supporting epithelial cell membrane integrity
- Vitamin D3 - Optimize to serum 25(OH)D of 50-80 ng/mL (dose varies; typically 4,000-10,000 IU daily), supporting LL-37 production and immune regulation
- Vitamin A - 5,000-10,000 IU daily, essential for mucosal integrity and goblet cell function
- Colostrum - 2-5 g daily, containing growth factors (IGF-1, TGF-beta), immunoglobulins, and lactoferrin that support mucosal repair
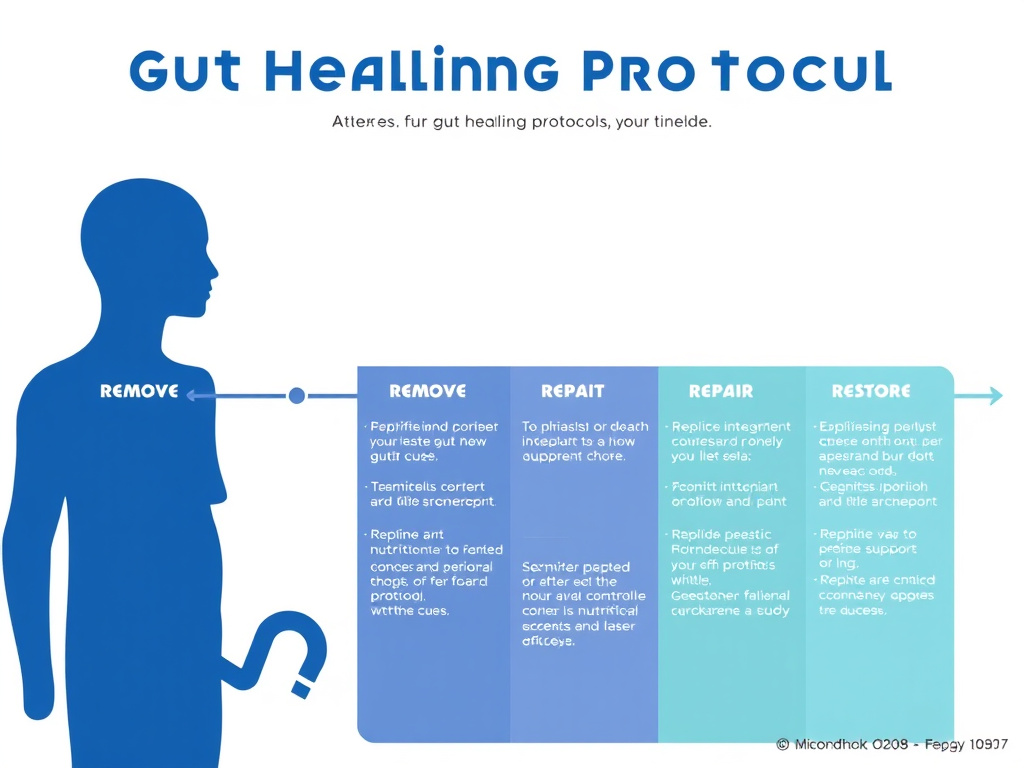
Figure 6: The four-phase gut healing protocol timeline. Peptides are introduced strategically at different phases to maximize their individual contributions - LL-37 and KPV in the Remove phase, BPC-157, larazotide, and VIP in the Repair phase, and probiotics with continued BPC-157 in the Restore phase.
Phase 4: Restore (Weeks 8-16+)
With the barrier repaired and inflammation controlled, the final phase focuses on rebuilding a healthy microbiome and maintaining long-term gut health.
Microbiome Restoration
- Probiotics - Multi-strain probiotic containing Lactobacillus and Bifidobacterium species, plus soil-based organisms (Bacillus species). Specific strain selection should be guided by stool testing when available. Common choices include Saccharomyces boulardii (especially post-antibiotic), Lactobacillus rhamnosus GG, and Bifidobacterium infantis 35624.
- Prebiotic fibers - Gradually reintroduce prebiotic fibers including partially hydrolyzed guar gum (PHGG), acacia fiber, GOS (galacto-oligosaccharides), and FOS (fructo-oligosaccharides). Start with small amounts (2-3 g/day) and increase slowly to avoid gas and bloating. PHGG is particularly well-tolerated and has evidence supporting its use in IBS and SIBO prevention.
- Fermented foods - Introduce small amounts of naturally fermented foods (sauerkraut, kimchi, kefir, yogurt) as tolerated, providing diverse bacterial strains and postbiotics.
- Polyphenol-rich foods - Blueberries, dark chocolate, green tea, and pomegranate provide polyphenols that act as prebiotics, selectively promoting beneficial bacteria like Akkermansia muciniphila and Faecalibacterium prausnitzii.
Maintenance Peptide Support
- BPC-157 - Continue at 250 mcg once daily for an additional 4-8 weeks, then taper to 2-3 times per week as a maintenance dose
- VIP - Continue nasal spray if motility or SIBO tendency remains a concern
- Consider NAD+ - for cellular energy support and mitochondrial function during the recovery phase
Lifestyle Optimization
- Stress management - Chronic psychological stress is one of the most potent drivers of gut barrier dysfunction, operating through the hypothalamic-pituitary-adrenal (HPA) axis and corticotropin-releasing hormone (CRH)-mediated mast cell activation. Implement stress reduction through meditation, breathwork, yoga, or other evidence-based practices.
- Sleep optimization - Poor sleep quality and circadian disruption increase intestinal permeability and alter the microbiome. Aim for 7-9 hours of quality sleep with consistent sleep/wake times.
- Exercise - Moderate exercise supports gut motility and microbiome diversity. Avoid overtraining, which can increase permeability (especially in endurance athletes).
- Meal timing - Allow 4-5 hours between meals to permit the migrating motor complex to function. Avoid constant grazing, which suppresses the MMC and promotes SIBO.
Protocol Customization
Not every patient needs every peptide or every phase. A patient with mild IBS and food sensitivities might only need BPC-157 and dietary changes. A patient with severe Crohn's disease and fistulas might need the full protocol plus conventional medical management. Use the peptide dosing calculator and work with a knowledgeable practitioner to customize the approach. The getting started guide provides additional resources for beginners.
Diet and Lifestyle Integration: The Foundation Peptides Build On
Peptides are powerful tools, but they work best when the biological environment supports healing. Without dietary and lifestyle changes, using gut-healing peptides is like patching a tire while continuing to drive over nails. This section covers the dietary frameworks and lifestyle modifications that create the optimal conditions for peptide-driven gut repair.
Anti-Inflammatory Dietary Frameworks
Several evidence-based dietary approaches support gut barrier repair and complement peptide therapy:
The Autoimmune Protocol (AIP)
The AIP is an elimination diet specifically designed for people with autoimmune conditions and gut barrier dysfunction. It removes all potentially inflammatory foods - grains, legumes, dairy, eggs, nuts, seeds, nightshades, alcohol, refined sugars, food additives, and NSAIDs - for 30-90 days before systematically reintroducing them one at a time. Research published in Inflammatory Bowel Diseases demonstrated that the AIP produced clinical remission in 73% of IBD patients by week 6 of the elimination phase.
The AIP is particularly well-suited as the dietary foundation during Phases 1-3 of the gut healing protocol, as it eliminates the dietary triggers that would undermine peptide-driven repair.
Low-FODMAP Diet
For patients with IBS symptoms or SIBO, the low-FODMAP diet reduces fermentable carbohydrates that feed bacterial overgrowth and cause gas, bloating, and abdominal pain. FODMAPs (Fermentable Oligo-, Di-, Mono-saccharides And Polyols) include fructose, lactose, fructans, galactans, and sugar alcohols found in many common foods.
Important note: The low-FODMAP diet is a diagnostic and therapeutic tool meant for short-term use (2-6 weeks), not a permanent dietary pattern. Prolonged FODMAP restriction can reduce beneficial bacteria like Bifidobacterium species. Once SIBO is treated and symptoms improve, FODMAPs should be gradually reintroduced to support a healthy microbiome.
Specific Carbohydrate Diet (SCD)
Originally developed for celiac disease and later adopted for IBD, the SCD eliminates complex carbohydrates (disaccharides and polysaccharides) while allowing monosaccharides, specific fruits and vegetables, and certain dairy products. The theory is that undigested complex carbohydrates feed pathogenic bacteria, perpetuating dysbiosis and inflammation. Clinical studies have shown improvement in pediatric Crohn's disease with the SCD.
Mediterranean Diet
For long-term maintenance (Phase 4 and beyond), the Mediterranean diet provides an evidence-based framework rich in anti-inflammatory omega-3 fatty acids, polyphenols, fiber, and fermented foods. It's associated with greater microbiome diversity, lower inflammatory markers, and reduced risk of IBD flares. It also happens to be enjoyable and sustainable - qualities that matter for long-term compliance.
Gut-Healing Foods to Emphasize
| Food Category | Examples | Mechanism |
|---|---|---|
| Bone broth | Chicken, beef, or fish bone broth | Provides collagen, glycine, proline, glutamine, and glycosaminoglycans that support mucosal repair |
| Fermented foods | Sauerkraut, kimchi, kefir, miso, natto | Provide diverse probiotic strains and postbiotic metabolites; introduce gradually in Phase 4 |
| Prebiotic-rich foods | Cooked onions, garlic, leeks, asparagus, green bananas, Jerusalem artichoke | Feed beneficial bacteria, promote SCFA production; introduce gradually to avoid bloating |
| Polyphenol sources | Blueberries, pomegranate, dark chocolate, green tea, extra virgin olive oil | Selective prebiotic effect; promote Akkermansia and F. prausnitzii; direct anti-inflammatory effects |
| Anti-inflammatory fats | Wild salmon, sardines, mackerel, avocado, olive oil, coconut oil | EPA/DHA reduce inflammatory prostaglandins; medium-chain triglycerides provide easy energy for healing |
| Glutamine-rich foods | Bone broth, grass-fed beef, poultry, wild fish, eggs (when tolerated) | Provide glutamine - the primary fuel for enterocytes and a building block for tight junction proteins |
| Zinc-rich foods | Oysters, pumpkin seeds, grass-fed beef, lamb | Zinc is essential for enterocyte proliferation, tight junction assembly, and mucosal immune function |
Foods and Substances to Avoid
During active gut healing (Phases 1-3), the following should be strictly avoided:
- Gluten - Triggers zonulin release, increases permeability, and causes immune activation even in non-celiac individuals with gut inflammation. This is where larazotide provides particular value - it blocks gluten-induced permeability changes for unavoidable exposures.
- Alcohol - Directly damages enterocytes, increases intestinal permeability, disrupts the microbiome, and impairs liver function needed for bile acid production and detoxification.
- NSAIDs (ibuprofen, naproxen, aspirin) - Cause direct mucosal injury by inhibiting prostaglandin synthesis. Even low-dose aspirin increases small intestinal permeability. BPC-157 can help counteract NSAID damage when these drugs cannot be discontinued, but avoidance is always preferable.
- Emulsifiers and food additives - Carboxymethylcellulose (CMC) and polysorbate 80, commonly found in processed foods, thin the mucus layer and promote bacterial encroachment toward the epithelium. Even "clean" processed foods may contain these additives.
- Artificial sweeteners - Sucralose, saccharin, and aspartame alter microbiome composition and may increase intestinal permeability. Stevia and monk fruit appear to be safer alternatives, though data is still limited.
- Refined seed oils - High omega-6 vegetable oils (soybean, corn, canola, sunflower) promote inflammatory prostaglandin production when consumed in excess relative to omega-3 intake.
Lifestyle Factors That Affect Gut Healing
Stress Management
The gut-brain axis is not a metaphor - it's a bidirectional communication system involving the vagus nerve, the HPA axis, the enteric nervous system, and the microbiome. Chronic psychological stress increases intestinal permeability through multiple mechanisms:
- CRH (corticotropin-releasing hormone) release in the gut activates mast cells, which degranulate and release histamine, tryptase, and other mediators that directly open tight junctions
- Cortisol suppresses secretory IgA production, reducing mucosal immune defense
- Sympathetic nervous system activation reduces mesenteric blood flow, starving the mucosa of oxygen and nutrients
- Stress alters the microbiome composition, reducing Lactobacillus species and promoting pathogenic bacteria
Evidence-based stress management practices that support gut healing include:
- Meditation - Even 10-15 minutes daily of mindfulness meditation has been shown to reduce inflammatory markers and improve GI symptoms in IBS patients
- Vagal nerve stimulation - Through deep breathing exercises (particularly extended exhalation), cold water face immersion, gargling, and singing/chanting
- Yoga - Particularly restorative yoga, which combines gentle movement with deep breathing and relaxation
- Nature exposure - Forest bathing and nature walks reduce cortisol and improve autonomic nervous system balance
Sleep
Sleep deprivation and circadian disruption directly increase intestinal permeability - even a single night of total sleep deprivation has been shown to increase permeability markers in healthy subjects. The mechanisms include:
- Disruption of circadian clock genes that regulate tight junction protein expression
- Increased cortisol and sympathetic activation that stress the barrier
- Altered microbiome composition - the microbiome has its own circadian rhythms that are disrupted by irregular sleep
- Impaired immune function that compromises mucosal defense
Sleep optimization during gut healing should include consistent sleep/wake times (even on weekends), a dark and cool sleeping environment, avoidance of screens for 1-2 hours before bed, and treatment of any underlying sleep disorders (including sleep apnea, which is independently associated with increased gut permeability).
Exercise
Moderate exercise supports gut health through improved motility, enhanced microbiome diversity, reduced systemic inflammation, and improved stress resilience. However, intense or prolonged exercise - particularly endurance training in heat - can temporarily increase intestinal permeability through splanchnic hypoperfusion (reduced blood flow to the gut).
During active gut healing, the sweet spot is moderate exercise 4-5 times per week: walking, swimming, cycling at conversational pace, yoga, or resistance training. High-intensity interval training and endurance training should be moderated until barrier integrity is restored.
Meal Timing and the Migrating Motor Complex
The migrating motor complex (MMC) is a cyclical pattern of electromechanical activity that occurs in the stomach and small intestine during fasting periods - typically between meals and during overnight fasting. It takes approximately 90-120 minutes to complete one full cycle, with Phase III (the "housekeeper wave") producing the strongest contractions that sweep bacteria, debris, and undigested particles from the small intestine into the colon.
The MMC is inhibited by eating. Every meal or snack resets the clock. This means that constant grazing - eating every 2-3 hours, as commonly recommended for blood sugar management - prevents the MMC from completing its cleaning cycle, potentially contributing to SIBO.
For gut healing, particularly in patients prone to SIBO:
- Eat 3 defined meals per day with 4-5 hours between them
- Avoid snacking between meals whenever possible
- Allow at least 12 hours of overnight fasting (e.g., finish dinner by 7 PM, breakfast at 7 AM)
- VIP nasal spray can support MMC function in patients with impaired motility
Integration with Peptide Timing
The fasting windows needed for MMC function also provide ideal timing for oral peptide administration. BPC-157 and KPV taken on an empty stomach get maximum mucosal contact without food interference. Larazotide should be taken 15 minutes before meals because its job is to be present in the lumen when food arrives. This means a typical daily schedule might look like: wake up, take BPC-157 + KPV, wait 30 minutes, take larazotide, wait 15 minutes, eat breakfast. Repeat larazotide before lunch and dinner.
Figure 7: Sample daily schedule integrating peptide dosing with meals, fasting windows for MMC function, exercise, and stress management. Timing matters - peptides taken on an empty stomach achieve better mucosal contact, while larazotide should precede meals.
Monitoring Progress: How to Know If Your Gut Is Healing
Gut healing is not always visible or immediately obvious from symptoms alone. Objective testing, symptom tracking, and biomarker monitoring provide the feedback needed to adjust protocols and confirm that healing is occurring at the tissue level.
Subjective Symptom Tracking
While subjective, symptom tracking provides the most immediate and practical feedback. Key symptoms to monitor include:
| Symptom | Improvement Indicates | Expected Timeline |
|---|---|---|
| Bloating and gas | Reduced bacterial overgrowth, improved digestion | 2-4 weeks |
| Abdominal pain/cramping | Reduced inflammation, improved motility | 2-6 weeks |
| Stool consistency | Improved absorption, reduced inflammation | 2-4 weeks |
| Stool frequency | Normalized motility, reduced inflammation | 2-6 weeks |
| Food sensitivities | Reduced permeability, barrier restoration | 4-12 weeks |
| Brain fog/fatigue | Reduced LPS translocation, improved gut-brain axis | 4-8 weeks |
| Skin conditions (acne, eczema) | Reduced systemic inflammation from gut origin | 6-12 weeks |
| Joint pain | Reduced immune activation from barrier dysfunction | 6-12 weeks |
Daily symptom logging using a simple 0-10 scale for key symptoms allows pattern recognition and correlation with dietary changes, peptide dosing, and other interventions. Many patients find that symptom improvement is not linear - there may be "good weeks and bad weeks" early in the process, with the overall trend gradually improving.
Objective Testing
Intestinal Permeability Testing
The gold standard for measuring intestinal permeability is the lactulose/mannitol test (also called the dual sugar test). The patient drinks a solution containing lactulose (a large disaccharide that should not cross intact tight junctions) and mannitol (a small monosaccharide that crosses easily). Urine is collected for 6 hours, and the ratio of lactulose to mannitol recovery is calculated. An elevated lactulose/mannitol ratio indicates increased paracellular permeability.
Test timing recommendations:
- Baseline - before starting the protocol
- Midpoint - at 6-8 weeks
- Completion - at 12-16 weeks
Zonulin Testing
Serum or fecal zonulin levels can be measured to assess tight junction regulation. Elevated zonulin indicates ongoing tight junction opening and may predict response to larazotide therapy. However, zonulin assays have significant variability between manufacturers, so it's important to use the same laboratory for serial measurements.
Stool Testing
Comprehensive stool analysis provides multiple relevant markers:
- Calprotectin - A neutrophil-derived protein that reflects intestinal inflammation. Levels above 50-100 mcg/g suggest active mucosal inflammation and are useful for monitoring IBD activity.
- Secretory IgA (sIgA) - Reflects mucosal immune function. Low sIgA may indicate impaired mucosal defense; elevated sIgA may indicate active immune response to a trigger.
- Microbiome analysis - 16S rRNA or metagenomic sequencing to assess microbiome diversity, identify pathogenic overgrowth, and track changes in key species during treatment.
- Short-chain fatty acids - Butyrate, propionate, and acetate levels reflect the metabolic output of the microbiome and the health of the colonic environment.
- Pancreatic elastase - Assesses exocrine pancreatic function; low levels indicate pancreatic insufficiency requiring enzyme supplementation.
Blood Markers
- LPS-binding protein (LBP) and anti-LPS antibodies - Elevated levels indicate bacterial translocation across a compromised barrier
- hs-CRP - High-sensitivity C-reactive protein reflects systemic inflammation, which often has a gut origin
- Vitamin D (25-OH) - Optimize to 50-80 ng/mL to support LL-37 production and immune function
- Anti-tTG and anti-DGP antibodies - For celiac patients, these antibodies should decline as barrier function improves and gluten exposure is minimized
- Food sensitivity panels - While controversial, IgG food antibody levels often decline as barrier function improves, reflecting reduced translocation of food antigens
SIBO Testing
For patients with SIBO as part of their gut dysfunction:
- Lactulose breath test - measures hydrogen and methane gas production; retest at 4-6 weeks after antimicrobial treatment to confirm eradication
- Glucose breath test - more specific for proximal SIBO but misses distal small bowel overgrowth
- Trio-Smart breath test - newer test that also measures hydrogen sulfide, identifying a third subtype of SIBO
Benchmarks for Protocol Progression
Clear benchmarks help determine when to transition between protocol phases:
| Transition | Criteria |
|---|---|
| Phase 1 to Phase 2 | Antimicrobial treatment complete; breath test negative (if SIBO present); major dietary triggers identified and removed |
| Phase 2 to Phase 3 | Digestive symptoms improving; adequate digestive enzyme support established; no ongoing acute infections |
| Phase 3 to Phase 4 | Significant symptom improvement (50%+ reduction in symptom scores); calprotectin declining (if elevated); lactulose/mannitol ratio improving |
| Phase 4 to Maintenance | Symptoms resolved or at acceptable baseline; permeability testing normalized; microbiome diversity improving; food reintroductions tolerating well |
Combining Peptides for Gut Health: Stacking Strategies
Each gut-healing peptide targets a different aspect of barrier dysfunction. Combining them strategically can produce results that exceed what any single peptide achieves alone. But the order, timing, and selection of combinations matters.
Rationale for Combination Therapy
Gut barrier dysfunction is not a single problem - it's a convergence of multiple failures: tight junction breakdown, mucosal inflammation, impaired blood flow, dysbiosis, reduced antimicrobial defense, and motility disturbances. No single peptide addresses all of these simultaneously. Combination therapy targets multiple mechanisms in parallel, potentially accelerating healing and reducing the risk that a single untreated pathway undermines overall progress.
The concept is analogous to combination antiretroviral therapy for HIV - where targeting multiple steps in the viral life cycle produces outcomes far superior to monotherapy. For gut healing, targeting multiple steps in the barrier dysfunction cascade may similarly produce additive or compounding outcomes.
Recommended Combinations by Condition
| Condition | Primary Peptide Stack | Supporting Interventions |
|---|---|---|
| Leaky gut / increased permeability | BPC-157 (oral) + Larazotide (oral) | L-glutamine, zinc carnosine, vitamin D |
| Ulcerative colitis | BPC-157 (oral) + KPV (oral) + Larazotide (oral) | Omega-3, curcumin, Boswellia |
| Crohn's disease | BPC-157 (oral + SC) + KPV (SC) + LL-37 (SC) | AIP diet, vitamin D optimization |
| SIBO | VIP (nasal) + LL-37 (SC) + BPC-157 (oral) | Prokinetics, herbal antimicrobials, meal spacing |
| IBS-D | BPC-157 (oral) + KPV (oral) + Larazotide (oral) | Low-FODMAP diet, probiotics, stress management |
| IBS-C | VIP (nasal) + BPC-157 (oral) | Magnesium, adequate fiber, exercise, hydration |
| Post-antibiotic gut repair | BPC-157 (oral) + LL-37 (SC) | Saccharomyces boulardii, multi-strain probiotic, prebiotic fiber |
| NSAID-induced gut damage | BPC-157 (oral, high priority) | PPI discontinuation if possible, misoprostol if not |
| Celiac disease (adjunct to GFD) | Larazotide (oral) + BPC-157 (oral) | DPP-IV enzymes, strict gluten-free diet |
| Mast cell activation / histamine issues | KPV (SC) + VIP (nasal) + BPC-157 (oral) | Low-histamine diet, DAO enzyme, quercetin |
Timing and Sequencing
Not all peptides should be started simultaneously. A phased introduction allows assessment of individual responses and reduces the risk of confounding variables if adverse reactions occur.
- Week 1-2: Start BPC-157 oral as the foundation. This is the most well-studied gut-healing peptide and provides broad cytoprotective support.
- Week 2-3: Add KPV (oral or SC) if inflammation is a significant component. The anti-inflammatory support complements BPC-157's repair mechanisms.
- Week 3-4: Add larazotide if permeability testing confirms tight junction dysfunction, or if clinical symptoms suggest ongoing leaky gut (food sensitivities, post-meal fatigue, joint pain).
- Week 4+: Add VIP nasal spray if motility issues or SIBO are present. Add LL-37 if dysbiosis or chronic infections are contributing factors.
This staggered approach allows each peptide to be introduced individually, making it easier to identify which components are contributing to improvement and to detect any adverse reactions early.
Peptide Interactions and Safety
No significant adverse interactions between these peptides have been reported in the available literature. Several points are worth noting:
- BPC-157 and KPV have complementary mechanisms (repair + anti-inflammation) with no known antagonistic interactions
- Larazotide acts locally in the gut lumen with minimal systemic absorption, minimizing the potential for interactions with systemically-active peptides
- VIP's short half-life (1-2 minutes) means that timing relative to other peptides is unlikely to create interaction concerns
- LL-37's antimicrobial mechanism (membrane disruption) is directed at microbes, not mammalian cells at therapeutic doses
However, as with any combination therapy, starting with lower doses and titrating up while monitoring for unexpected effects is prudent clinical practice.
Cost Considerations
A full five-peptide stack can be expensive. For patients on a budget, prioritize based on the primary mechanism of their gut dysfunction. BPC-157 oral is typically the highest-priority single peptide for most gut conditions. Adding KPV provides the most additional benefit for inflammatory conditions. Larazotide is highest priority for permeability-driven conditions with food sensitivities. Work with a practitioner to identify which 1-2 peptides will provide the most benefit for your specific situation, and visit the peptide research hub for the latest comparative information.
Special Conditions: SIBO, Crohn's, Ulcerative Colitis, and IBS
While the general gut healing protocol provides a framework, specific gastrointestinal conditions have unique pathophysiology that warrants tailored peptide approaches. This section addresses the four most common conditions where patients seek peptide support.
SIBO (Small Intestinal Bacterial Overgrowth)
SIBO affects an estimated 6-15% of healthy asymptomatic individuals and up to 80% of patients with IBS. It occurs when bacteria that normally inhabit the colon proliferate in the small intestine, producing gas, causing bloating, and damaging the small intestinal mucosa. Three subtypes are recognized based on the predominant gas produced:
- Hydrogen-dominant SIBO - associated with diarrhea; treated with rifaximin or herbal antimicrobials
- Methane-dominant SIBO (now called intestinal methanogen overgrowth or IMO) - associated with constipation; treated with rifaximin plus neomycin or metronidazole
- Hydrogen sulfide-dominant SIBO - associated with diarrhea and foul-smelling gas; treated with bismuth-based protocols
Peptide Strategy for SIBO
The key challenge with SIBO isn't just eliminating the overgrowth - it's preventing recurrence. SIBO recurrence rates are notoriously high (up to 44% within 9 months of treatment) because the underlying motility and immune factors that allowed overgrowth in the first place often persist. This is where peptides can make a meaningful difference.
| Phase | Peptides | Purpose |
|---|---|---|
| Antimicrobial phase (weeks 1-4) | LL-37 (100 mcg SC daily) + BPC-157 (250 mcg oral daily) | LL-37 enhances antimicrobial efficacy and disrupts biofilms; BPC-157 protects the mucosa during treatment |
| Repair phase (weeks 4-10) | BPC-157 (500 mcg oral twice daily) + VIP (50-100 mcg nasal spray daily) | BPC-157 repairs mucosal damage; VIP restores motility and MMC function to prevent recurrence |
| Prevention phase (ongoing) | VIP (50 mcg nasal spray daily or as needed) | Maintains MMC function and healthy motility; reduces SIBO recurrence risk |
Critical adjuncts for SIBO management include:
- Prokinetics - Low-dose erythromycin (50 mg at bedtime), prucalopride, or herbal prokinetics (ginger, 5-HTP, Iberogast) taken at bedtime to support the overnight MMC
- Meal spacing - Minimum 4-5 hours between meals; no snacking; 12+ hour overnight fast
- Partially hydrolyzed guar gum (PHGG) - 5 g daily, shown to reduce SIBO recurrence when used as a prebiotic after treatment
Figure 8: SIBO treatment and prevention strategy incorporating peptide therapy. LL-37 supports the antimicrobial phase, BPC-157 repairs mucosal damage, and VIP restores motility to prevent recurrence - the most challenging aspect of SIBO management.
Crohn's Disease
Crohn's disease is a chronic transmural inflammatory condition that can affect any part of the GI tract, from mouth to anus, though it most commonly involves the terminal ileum and colon. Its pathogenesis involves genetic susceptibility, environmental triggers, dysbiosis, and a dysregulated immune response that produces deep, penetrating inflammation.
Unique Challenges in Crohn's
- Transmural inflammation - Unlike UC, Crohn's inflammation extends through all layers of the bowel wall, meaning luminal peptide delivery may not reach the deepest layers of disease
- Fistula formation - Abnormal connections between intestinal loops, between intestine and skin, or between intestine and other organs occur in up to 50% of Crohn's patients over their lifetime
- Strictures - Chronic inflammation leads to fibrotic narrowing of the bowel lumen, potentially requiring surgical intervention
- Skip lesions - Patchy distribution means healthy and diseased segments alternate, complicating treatment targeting
Peptide Approach for Crohn's
Crohn's disease typically requires a more aggressive and multimodal peptide approach than other gut conditions:
- BPC-157 - Use both oral (500 mcg twice daily for luminal contact) AND subcutaneous injection (250-500 mcg daily for systemic delivery to deeper tissue layers). BPC-157's fistula-healing properties are particularly relevant here - it's the only peptide with extensive preclinical data showing healing of various GI fistula types.
- KPV - Subcutaneous injection (500 mcg daily) rather than oral, to ensure systemic anti-inflammatory delivery that reaches transmural inflammation. The NF-kB inhibition addresses a core pathogenic mechanism in Crohn's.
- LL-37 - Subcutaneous (100 mcg daily) to address the antimicrobial peptide deficiency that has been documented in Crohn's patients, particularly those with ileal disease and NOD2/CARD15 mutations. Crohn's patients often have a deficiency of Paneth cell-derived antimicrobial peptides, and supplemental LL-37 can partially compensate.
- Thymosin alpha-1 - Consider adding for immune modulation, particularly in patients with recurrent infections or those on immunosuppressive therapy. Ta1 can support immune surveillance while reducing the dysfunctional inflammatory response.
Crohn's Disease and Conventional Treatment
Peptide therapy for Crohn's disease should be viewed as complementary to, not a replacement for, evidence-based conventional treatments. Biologics (infliximab, adalimumab, vedolizumab, ustekinumab), immunomodulators, and corticosteroids have strong evidence for inducing and maintaining remission. Peptides may enhance response, support mucosal healing, and address mechanisms not fully covered by conventional therapy. Always work with a gastroenterologist experienced in IBD management when using peptides alongside standard treatment.
Ulcerative Colitis
Ulcerative colitis is a chronic inflammatory condition limited to the colon and rectum, with inflammation confined to the mucosal and submucosal layers. Its continuous, non-skip pattern (starting at the rectum and extending proximally) and superficial inflammation make it more amenable to luminally-delivered peptide therapy than Crohn's disease.
Why UC Is Well-Suited for Peptide Therapy
- Mucosal confinement - UC inflammation is superficial compared to Crohn's, meaning orally-delivered peptides that contact the colonic mucosa can directly reach the inflamed tissue
- PepT1 upregulation - PepT1 is strongly upregulated in the inflamed colonic mucosa of UC patients, making KPV delivery highly efficient in active disease
- Barrier dysfunction as a driver - Increased intestinal permeability is both a cause and consequence of UC, making tight junction stabilization (larazotide) directly relevant
- Continuous inflammation - The non-skip pattern means that orally-delivered peptides that reach the colon will encounter inflamed tissue throughout the affected area
Peptide Protocol for UC
| Disease Activity | Peptide Stack | Notes |
|---|---|---|
| Remission (maintenance) | BPC-157 oral 250 mcg daily + Larazotide 0.5 mg TID | Focus on maintaining barrier integrity and mucosal health |
| Mild flare | BPC-157 oral 500 mcg BID + KPV oral 500 mcg daily + Larazotide 0.5 mg TID | Add anti-inflammatory KPV; increase BPC-157 dose |
| Moderate-severe flare | BPC-157 oral 500 mcg BID + KPV SC 500 mcg daily + Larazotide 0.5 mg TID + VIP nasal 100 mcg BID | Full stack with systemic KPV delivery; VIP for immune modulation |
Irritable Bowel Syndrome (IBS)
IBS is the most common functional gastrointestinal disorder, affecting 10-15% of the global population. While traditionally considered a "functional" condition without structural pathology, modern research has identified measurable abnormalities in many IBS patients: low-grade mucosal inflammation, increased intestinal permeability, visceral hypersensitivity, altered microbiome composition, and gut-brain axis dysfunction.
IBS Subtypes and Peptide Selection
| IBS Subtype | Key Pathophysiology | Primary Peptides | Additional Support |
|---|---|---|---|
| IBS-D (Diarrhea) | Increased permeability, mucosal inflammation, rapid transit | BPC-157 oral + KPV oral + Larazotide | Low-FODMAP diet, probiotics (S. boulardii), soluble fiber |
| IBS-C (Constipation) | Slow transit, possible methane SIBO, reduced VIP signaling | VIP nasal + BPC-157 oral | Magnesium, prokinetics, adequate hydration, exercise |
| IBS-M (Mixed) | Variable motility, dysbiosis, visceral hypersensitivity | BPC-157 oral + VIP nasal + KPV oral | Stress management, dietary modification, peppermint oil |
| Post-Infectious IBS | Residual inflammation, increased permeability, altered microbiome | BPC-157 oral + KPV oral + LL-37 SC | Time-limited; aim for 3-6 month protocol |
The Gut-Brain Axis in IBS
IBS is increasingly understood as a disorder of gut-brain interaction (DGBI). Visceral hypersensitivity - where normal gut sensations are perceived as painful - is driven by sensitization of enteric neurons and altered central pain processing. VIP, as a neuromodulator in the enteric nervous system, may help normalize these aberrant neural signals. BPC-157's neuroprotective properties, including its effects on dopaminergic and serotonergic systems, may also contribute to normalizing gut-brain communication.
Stress management is particularly critical in IBS, as psychological stress directly exacerbates symptoms through CRH-mediated mast cell activation in the gut. Some practitioners combine peptide therapy with targeted stress-reduction interventions (gut-directed hypnotherapy, cognitive behavioral therapy) and find that the combination produces results superior to either approach alone.
When to Consider Peptide Therapy for IBS
Not every IBS patient needs peptide therapy. Consider peptides when: (1) standard dietary interventions (low-FODMAP, elimination diet) provide incomplete relief; (2) testing reveals increased intestinal permeability, elevated calprotectin, or positive SIBO; (3) there is a clear post-infectious onset suggesting structural damage; or (4) the patient has tried multiple conventional approaches without adequate improvement. For mild IBS responsive to dietary changes and stress management alone, peptides may be unnecessary.
Additional Conditions Where Gut-Healing Peptides May Help
Gastroesophageal Reflux Disease (GERD)
BPC-157's ability to restore lower esophageal sphincter pressure while healing esophageal inflammation makes it particularly relevant for GERD, especially in patients trying to discontinue proton pump inhibitors. The peptide addresses the root cause (sphincter dysfunction) rather than just suppressing symptoms (acid secretion).
Celiac Disease
Larazotide acetate is the primary peptide for celiac disease, with Phase III clinical trial data supporting its use as an adjunct to the gluten-free diet. BPC-157 can complement larazotide by supporting mucosal repair of the villous atrophy that characterizes celiac disease.
Gastritis
Chronic gastritis - whether from H. pylori, autoimmune mechanisms, or NSAID use - responds to BPC-157's cytoprotective and mucosal healing properties. For H. pylori-related gastritis, combining LL-37 (for antimicrobial support) with BPC-157 (for mucosal repair) addresses both the infection and the damage it causes.
Diverticular Disease
Recurrent diverticulitis involves mucosal barrier breakdown, bacterial translocation, and localized inflammation in diverticular pockets. BPC-157 (oral) for mucosal protection and KPV (oral or SC) for anti-inflammatory support may reduce the frequency and severity of diverticulitis episodes, though direct evidence is lacking.
Radiation Enteritis/Proctitis
Radiation therapy for pelvic cancers damages the intestinal mucosa, causing acute and chronic enteritis/proctitis. VIP has shown protection against radiation-induced intestinal injury in preclinical models, and BPC-157's tissue repair properties are relevant for radiation-damaged mucosa. For patients undergoing pelvic radiation, prophylactic peptide use during and after treatment could theoretically reduce intestinal side effects.
Oral vs. Injectable Peptides for Gut Conditions: Choosing the Right Route
Route of administration matters enormously for gut-targeting peptides. The right delivery method can mean the difference between a peptide reaching its target tissue at therapeutic concentrations and one that never gets there. Here's how to think about route selection for each peptide in a gut-healing context.
General Principles
For gut conditions, the choice between oral and injectable delivery depends on three factors:
- Where is the target tissue? - Luminal/mucosal targets (ulcers, mucosal inflammation, tight junctions) are best reached by oral delivery. Deeper targets (transmural inflammation, systemic immune dysfunction) require injectable routes.
- Is the peptide stable in the GI tract? - BPC-157 and larazotide are stable in gastric acid; KPV is a small tripeptide that may be partially degraded before reaching the colon; VIP and LL-37 are rapidly degraded if taken orally.
- What's the primary mechanism? - Local/luminal mechanisms (barrier sealing, mucosal cytoprotection, antimicrobial surface defense) favor oral delivery. Systemic mechanisms (immune modulation, neurological effects, growth factor circulation) favor injectable routes.
Route Selection by Peptide
| Peptide | Preferred Route for Gut | Alternative Route | Rationale |
|---|---|---|---|
| BPC-157 | Oral | SC injection (add for severe cases) | Stable in gastric acid (24+ hrs); direct mucosal contact; most preclinical GI data used intragastric dosing |
| KPV | Oral or SC (both valid) | Rectal (for left-sided colitis) | Oral reaches small bowel via PepT1; SC provides systemic anti-inflammatory effect; rectal targets distal colon directly |
| Larazotide | Oral (exclusively) | N/A | Acts locally in intestinal lumen; <1% systemic absorption by design; no benefit from injection |
| VIP | Nasal spray | SC injection | Rapidly degraded orally (t1/2 1-2 min); nasal provides systemic delivery; affects gut via enteric nervous system |
| LL-37 | SC injection | N/A for gut use | Degraded by digestive proteases if taken orally; SC provides systemic antimicrobial and immune effects |
The Case for Dual-Route BPC-157
For severe gut conditions - active Crohn's disease, complex fistulas, or severe pan-colonic UC - some practitioners use BPC-157 via both routes simultaneously. The oral dose provides direct mucosal contact throughout the GI tract, while the subcutaneous injection ensures systemic delivery of the peptide's angiogenic, anti-inflammatory, and growth factor-modulating effects to tissue layers that luminal delivery may not adequately reach.
For a detailed comparison of oral vs. injectable BPC-157 with dosing guidance, see our full oral vs. injectable BPC report.
Rectal Administration: An Underutilized Route
For patients with left-sided ulcerative colitis or proctitis - where inflammation is concentrated in the sigmoid colon and rectum - rectal administration of certain peptides may provide the most direct delivery to the affected tissue. KPV in particular, with its PepT1-mediated uptake mechanism, could theoretically be formulated as a retention enema or suppository for targeted delivery to inflamed distal colon.
This approach mirrors the use of mesalamine (5-ASA) enemas and suppositories in UC management, where topical rectal delivery often outperforms oral dosing for distal disease. While commercial rectal peptide formulations are not widely available, compounding pharmacies can prepare custom formulations.
Practical Route Selection Framework
When in doubt, use this simple decision tree: (1) Is the target in the GI lumen or mucosa? Use oral if the peptide is acid-stable. (2) Is the target below the mucosa or systemic? Use injectable. (3) Is the target specifically the distal colon/rectum? Consider rectal delivery. (4) Is the condition severe with both mucosal and transmural involvement? Use dual routes. Most patients with common gut conditions (IBS, leaky gut, mild IBD, gastritis) will do well with oral peptides alone.
Safety Considerations for Gut-Healing Peptides
Safety must be the foundation of any therapeutic protocol. While gut-healing peptides have generally favorable safety profiles in available data, understanding the limitations of current evidence and the specific cautions for each compound is essential for responsible use.
General Safety Principles
Several overarching safety considerations apply to all peptide protocols for gut health:
1. Evidence Limitations
With the exception of larazotide acetate, none of the peptides discussed in this report have completed Phase III human clinical trials for any gastrointestinal indication. BPC-157 has over 100 preclinical studies but limited human data. KPV, LL-37, and VIP have preclinical and mechanistic evidence but minimal controlled human trial data for gut conditions specifically. This doesn't mean they're unsafe - it means the safety profile is based on animal studies, mechanistic understanding, clinical experience from practitioners, and limited human data rather than the rigorous, large-scale safety databases that accompany FDA-approved drugs.
2. Source Quality
Peptide quality varies dramatically between suppliers. Contaminants, incorrect sequences, degradation products, and inaccurate concentrations are all real risks with substandard sources. For all peptides in this report:
- Use only suppliers that provide third-party certificates of analysis (COA) from independent labs
- Look for High-Performance Liquid Chromatography (HPLC) purity testing showing greater than 98% purity
- Verify peptide identity through mass spectrometry
- Check for endotoxin testing (particularly important for injectable peptides)
- Ensure proper cold-chain storage and handling
3. Drug Interactions
Systematic drug interaction studies have not been conducted for most of these peptides. Known and theoretical interactions include:
| Peptide | Potential Interactions | Precaution |
|---|---|---|
| BPC-157 | May interact with drugs metabolized through NO system; theoretical interaction with blood pressure medications | Monitor blood pressure; start low dose if on antihypertensives |
| KPV | May enhance effects of other immunosuppressive agents through NF-kB inhibition | Monitor for excessive immunosuppression if used with biologics or immunomodulators |
| Larazotide | Minimal systemic absorption reduces interaction potential; may affect absorption of other orally-administered drugs | Take 15 minutes before meals; separate from other medications by 1-2 hours |
| VIP | May enhance hypotensive effects of blood pressure medications; may affect blood glucose in diabetic patients | Monitor blood pressure and blood glucose; start with lowest dose |
| LL-37 | Theoretical additive effects with other antimicrobial agents; may influence vitamin D metabolism | Use cautiously with concurrent antibiotic therapy; optimize vitamin D status |
4. Contraindications and Cautions
| Situation | Caution Level | Guidance |
|---|---|---|
| Active cancer | Caution with BPC-157 | BPC-157's angiogenic properties could theoretically support tumor blood supply. No evidence of tumor promotion exists, but most practitioners avoid BPC-157 in patients with active malignancy until more data is available. |
| Pregnancy/lactation | Avoid all peptides | No safety data exists for any of these peptides in pregnancy or lactation. Avoid until more data is available. |
| Children | Limited data | Pediatric dosing data is essentially nonexistent. Any use in children should be under strict medical supervision with compelling justification. |
| Severe immunosuppression | Caution with LL-37 and KPV | LL-37's immune-modulating effects and KPV's NF-kB inhibition could theoretically worsen immunosuppression. Consult with an immunologist. |
| History of allergic reactions to peptides | Caution with all peptides | Start with micro-dosing to test for allergic response. Have antihistamines available. |
| Autoimmune conditions | Monitor closely | Immune-modulating peptides may influence autoimmune disease activity in either direction. Close monitoring of disease activity markers is essential. |
5. Monitoring During Peptide Therapy
The following monitoring schedule is recommended during gut-healing peptide protocols:
- Baseline (before starting) - Complete blood count, comprehensive metabolic panel, inflammatory markers (CRP, ESR), vitamin D level, intestinal permeability testing, comprehensive stool analysis
- 4-week check - Symptom assessment, repeat CRP/ESR if elevated at baseline, assess for any adverse effects
- 8-week check - Repeat permeability testing, stool calprotectin (if elevated at baseline), assess symptom trajectory
- 12-week review - Comprehensive reassessment including all baseline tests, protocol adjustment based on response
6. Adverse Effects to Watch For
While serious adverse effects are uncommon with these peptides based on available data, patients should be aware of potential reactions:
- Injection site reactions (SC peptides) - Redness, swelling, or itching at injection sites. Usually mild and self-limiting. Rotate injection sites.
- GI symptoms (oral peptides) - Nausea, mild cramping, or changes in bowel habits when starting. Usually transient. Start with lower doses if sensitive.
- Headache - Reported with VIP nasal spray and occasionally with other peptides. Usually dose-related and mild.
- Flushing/warmth - VIP is a vasodilator and may cause transient facial flushing or warmth. This is a pharmacological effect, not an allergy.
- Herxheimer-like reactions - When LL-37 is used alongside antimicrobial treatment, die-off of pathogenic organisms can cause temporary symptom worsening.
- Blood pressure changes - VIP and BPC-157 both affect vascular tone. Patients on blood pressure medications should monitor blood pressure when starting these peptides.
When to Stop and Seek Medical Attention
Discontinue peptides and seek medical attention if you experience: severe allergic reaction (difficulty breathing, facial swelling, hives), significant blood pressure changes, unexpected bleeding, severe or worsening GI symptoms that don't resolve within a few days, signs of infection at injection sites (increasing redness, warmth, swelling, or pus), or any symptom that feels significantly concerning. While serious adverse events are rare, erring on the side of caution is always appropriate with research compounds.
Regulatory Status
As of early 2026, the regulatory landscape for gut-healing peptides includes:
- BPC-157 - Not FDA-approved for any indication. Available as a research compound and through compounding pharmacies. In 2024, the FDA raised concerns about peptides sold for self-administration, though BPC-157 was not specifically prohibited.
- KPV - Not FDA-approved. Available as a research compound.
- Larazotide acetate - In Phase III clinical trials for celiac disease. Fast Track designation from the FDA. If approved, it would be the first tight junction-regulating drug on the market.
- VIP - FDA-approved for diagnostic use only (in the form of secretin-VIP); therapeutic use is off-label. Available through compounding pharmacies.
- LL-37 - Not FDA-approved for any indication. Available as a research compound.
Patients should be aware that using non-FDA-approved peptides carries inherent regulatory and safety uncertainties. Working with a knowledgeable healthcare provider who can supervise use, monitor for adverse effects, and adjust protocols based on response is strongly recommended. Visit our immune peptide research hub for additional regulatory updates.
Emerging Research and Future Directions in Gut Peptide Therapy
The field of peptide-based gut healing is evolving rapidly. Several research frontiers promise to expand both our understanding of how peptides repair the gut and our ability to deliver them effectively. Here's where the science is heading.
Next-Generation Delivery Systems
One of the biggest challenges in gut peptide therapy is getting the right peptide to the right tissue at the right concentration. Current oral delivery works well for acid-stable peptides like BPC-157 and larazotide, but many promising gut-healing peptides are degraded before reaching their target. Research into advanced delivery systems is addressing this limitation.
Nanoparticle Encapsulation
The hyaluronic acid-functionalized nanoparticle system developed for KPV delivery (approximately 272 nm particles with negative surface charge) represents a proof of concept that could be applied to other gut-healing peptides. These nanoparticles accomplish three things simultaneously: they protect the peptide from enzymatic degradation during GI transit, they target delivery to inflamed tissue through receptor-mediated interactions (HA binding to CD44, which is upregulated on inflamed epithelium), and they enable sustained release at the target site (doi: 10.1016/j.ymthe.2017.02.015).
Future nanoparticle systems could encapsulate multiple peptides in a single formulation - for example, a nanoparticle carrying both KPV (for anti-inflammatory effect) and BPC-157 (for mucosal repair) targeted to inflamed colonic mucosa. Such combination nanoparticles are already in early preclinical development for other drug combinations.
pH-Responsive Coatings
Enteric coatings that dissolve only at specific pH ranges can target peptide release to particular segments of the GI tract. A pH 5.5 coating dissolves in the proximal small intestine, while a pH 7.0 coating survives until the terminal ileum or colon. This technology, already used for mesalamine delivery in IBD, could be adapted for peptides that need to reach specific GI segments.
Mucoadhesive Formulations
Mucoadhesive polymers (chitosan, alginate, carbomers) can extend the residence time of peptides on the mucosal surface, increasing the duration of contact between the peptide and its target tissue. For BPC-157, which works through direct mucosal contact, a mucoadhesive oral formulation could significantly enhance efficacy by preventing rapid transit past the target area.
Probiotic-Based Peptide Delivery
Perhaps the most creative approach involves engineering probiotic bacteria to produce and secrete therapeutic peptides directly in the gut lumen. Researchers have successfully engineered Lactococcus lactis strains to produce and secrete anti-inflammatory peptides, including trefoil factors and anti-TNF nanobodies, directly at the site of intestinal inflammation. Applying this approach to gut-healing peptides like KPV or LL-37 fragments could provide continuous, targeted delivery without the need for repeated dosing.
The Gut-Brain Axis and Peptide Therapy
The bidirectional communication between the gut and brain - mediated by the vagus nerve, immune signaling, microbial metabolites, and neuroactive peptides - is increasingly recognized as a central axis in both GI and neuropsychiatric health. Several gut-healing peptides have direct or indirect effects on this axis.
BPC-157 and the Brain-Gut Axis
A 2023 review in Pharmaceuticals specifically addressed BPC-157's potential to recover brain-gut and gut-brain axis function. The peptide's effects on the dopaminergic, serotonergic, GABAergic, and opioid systems - combined with its gut-healing properties - position it as a unique compound that could simultaneously address GI dysfunction and its neuropsychiatric consequences, including depression, anxiety, and cognitive impairment that often accompany chronic gut conditions (doi: 10.3390/ph16050682).
For patients with IBS, where gut-brain axis dysfunction is a central feature, BPC-157's dual gut-brain effects may explain why some patients report improvements in both GI symptoms and mood/cognition during treatment.
VIP as a Neurogastroenterological Agent
VIP is both a neuropeptide and a gut peptide, making it uniquely positioned to modulate gut-brain communication. Its effects on enteric nervous system function, motility regulation, and immune modulation are all mediated through neural pathways that connect to the central nervous system via the vagus nerve. Restoring VIP signaling in the gut may have upstream effects on brain function - a hypothesis supported by observations of cognitive improvement in CIRS patients treated with VIP nasal spray.
Microbiome-Targeted Peptide Approaches
The emerging understanding of how the gut microbiome influences - and is influenced by - peptide signaling opens new therapeutic possibilities.
Antimicrobial Peptides as Microbiome Modulators
Rather than using broad-spectrum antibiotics that devastate the microbiome, antimicrobial peptides like LL-37 offer the possibility of selective microbiome modulation - reducing pathogenic species while preserving or even promoting beneficial commensals. Research is exploring whether designer antimicrobial peptides can be created with even greater selectivity, targeting specific pathogenic species (like adherent-invasive E. coli strains implicated in Crohn's disease) while leaving the rest of the microbiome intact.
Postbiotic Peptides
Bacteria themselves produce peptides that influence gut health. Short peptides produced by Lactobacillus species have been shown to strengthen tight junctions, and bacteriocins (antimicrobial peptides produced by commensal bacteria) contribute to microbiome homeostasis. Understanding these naturally produced peptides could lead to new therapeutic agents derived from the microbiome itself.
Microbiome-Responsive Peptide Release
Future smart delivery systems could use microbiome-specific enzymes as triggers for peptide release. For example, azo-bond coatings that are cleaved by colonic bacterial azoreductases could trigger peptide release specifically in the colon. This would allow precise delivery of anti-inflammatory or barrier-healing peptides to the colonic environment without affecting the upper GI tract.
Personalized Peptide Protocols
As diagnostic tools improve, peptide protocols can become increasingly personalized:
- Permeability phenotyping - Advanced permeability testing can distinguish between pore pathway dysfunction (claudin-mediated) and leak pathway dysfunction (tight junction strand breaks), potentially guiding selection between peptides that target different mechanisms
- Microbiome profiling - Metagenomic analysis can identify specific dysbiosis patterns that guide selection between antimicrobial peptides (LL-37 for bacterial overgrowth) and microbiome-supportive approaches (VIP for motility-driven dysbiosis)
- Genetic testing - Variants in genes like NOD2/CARD15 (associated with Crohn's and reduced Paneth cell antimicrobial peptide production), FUT2 (affecting mucus composition), and HLA-DQ2/DQ8 (celiac susceptibility) can inform which peptides are most relevant for a given patient
- Inflammatory profiling - Cytokine panels can reveal whether a patient's inflammation is primarily Th1-driven (more relevant to KPV's NF-kB inhibition), Th2-driven (potentially responsive to VIP), or Th17-driven (potentially responsive to LL-37's immune modulation)
Figure 9: Personalized peptide selection framework. Diagnostic testing - including permeability phenotyping, microbiome profiling, genetic analysis, and inflammatory markers - guides selection of the most appropriate peptides for each patient's specific pattern of gut dysfunction.
Clinical Trials on the Horizon
Several developments in the clinical trial landscape are worth watching:
- Larazotide Phase III results - Expected to report in 2026-2027, these results will determine whether the first tight junction-regulating drug reaches the market for celiac disease
- BPC-157 first-in-human trials - Multiple groups have expressed interest in conducting formal Phase I/II trials, though none have been registered as of early 2026. Such trials would provide the first rigorous human safety and efficacy data for this extensively-studied peptide
- KPV nanoparticle formulations - The transition from preclinical nanoparticle studies to human trials could establish KPV as a targeted colitis therapy
- VIP for motility disorders - As understanding of VIP's role in the microbiome-motility axis grows, clinical trials for SIBO prevention and functional GI disorders may emerge
Integration with Conventional Medicine
The future of gut healing likely lies not in choosing between conventional medicine and peptide therapy, but in integrating them. Biologic therapies for IBD (anti-TNF, anti-integrin, anti-IL-12/23) are effective but incomplete - many patients achieve clinical remission without full mucosal healing. Adding peptides that specifically target mucosal repair (BPC-157), barrier integrity (larazotide), and antimicrobial defense (LL-37) to biologic regimens could help close the gap between clinical and endoscopic remission.
Similarly, for SIBO, combining conventional antimicrobials with VIP for motility restoration and BPC-157 for mucosal repair could reduce the high recurrence rates that plague current treatment approaches.
The key is moving from an either/or mentality to a both/and approach - using the best of conventional medicine and peptide therapy together for optimal gut healing outcomes.
Staying Current
The research landscape for gut-healing peptides is evolving quickly. For the latest evidence, formulation updates, and protocol refinements, visit the FormBlends peptide research hub, which is updated regularly with new study summaries and clinical insights. Individual peptide research pages for BPC-157, VIP, and immune peptides provide the most detailed and current analysis of each compound.
Clinical Scenarios: Putting It All Together
Theory is useful, but application is what matters. Here are several clinical scenarios that illustrate how the principles, peptides, and protocols discussed in this report come together in practice. These are composite examples for educational purposes and do not represent specific patients.
Scenario 1: The Post-Infectious IBS Patient
Presentation: A 34-year-old woman developed persistent diarrhea, bloating, and food sensitivities 6 months after a severe case of food poisoning from contaminated salad greens. She was diagnosed with post-infectious IBS-D. A low-FODMAP diet helped somewhat but didn't resolve symptoms. Testing revealed elevated fecal calprotectin (85 mcg/g, mildly elevated), positive lactulose breath test for hydrogen-dominant SIBO, and an elevated lactulose/mannitol ratio on permeability testing.
Protocol:
- Phase 1 (Weeks 1-2): Rifaximin 550 mg TID for 14 days + LL-37 100 mcg SC daily + Low-FODMAP diet continues
- Phase 2-3 (Weeks 3-10): BPC-157 500 mcg oral twice daily + KPV 250 mcg oral daily + VIP 50 mcg nasal spray at bedtime (for MMC support) + L-glutamine 5 g daily + Zinc carnosine 75 mg daily
- Phase 4 (Weeks 10-16): Continue BPC-157 250 mcg daily + PHGG 5 g daily + Multi-strain probiotic + Gradual FODMAP reintroduction
Expected trajectory: SIBO breath test negative at 4 weeks. Bloating and diarrhea improving by weeks 4-6. Food sensitivities gradually resolving by weeks 8-12. Calprotectin and permeability testing normalizing by week 12-16.
Scenario 2: The Celiac Patient with Persistent Symptoms
Presentation: A 45-year-old man with confirmed celiac disease has been strictly gluten-free for 3 years but continues to experience intermittent bloating, loose stools, fatigue, and joint pain. Follow-up biopsy shows improved but incomplete villous recovery (Marsh 3a). Anti-tTG levels have decreased but remain mildly positive. He suspects inadvertent gluten cross-contamination at restaurants is contributing.
Protocol:
- Ongoing: Strict gluten-free diet with added vigilance + DPP-IV enzyme with meals outside the home + Larazotide 0.5 mg oral TID before meals (primary peptide for this scenario)
- Repair phase (12 weeks): BPC-157 500 mcg oral twice daily (for villous repair) + Vitamin D optimization to 60+ ng/mL + Zinc carnosine 150 mg daily
- Maintenance: Continue larazotide TID (especially before meals outside the home) + BPC-157 250 mcg daily
Expected trajectory: Larazotide provides immediate protection against gluten cross-contamination. BPC-157 supports ongoing villous repair. Anti-tTG levels should decline further over 3-6 months. Joint pain improvement by weeks 6-12 as barrier function improves and gluten peptide translocation decreases.
Scenario 3: The Mild-to-Moderate Ulcerative Colitis Patient
Presentation: A 28-year-old woman with left-sided UC on mesalamine 4.8 g daily achieves partial remission but continues to have 2-4 bloody stools daily with mild urgency. Calprotectin is 220 mcg/g (elevated). She is reluctant to escalate to biologics and wants to explore adjunctive approaches before considering escalation.
Protocol (adjunctive to ongoing mesalamine):
- Weeks 1-12: BPC-157 500 mcg oral twice daily + KPV 500 mcg oral daily (targeting colonic PepT1) + Larazotide 0.5 mg oral TID before meals
- Nutritional support: Omega-3 (EPA 2 g + DHA 1 g daily) + Curcumin 1 g twice daily (Meriva/Theracurmin formulation for absorption) + Vitamin D to 60+ ng/mL
- Dietary: AIP diet for first 8 weeks, then gradual reintroduction
Expected trajectory: Stool frequency and bleeding should decrease over 4-8 weeks. Calprotectin recheck at 8 weeks. If calprotectin drops below 150 and symptoms improve significantly, continue adjunctive peptide therapy with ongoing monitoring. If insufficient response by 12 weeks, biologic escalation should be reconsidered - peptides are adjunctive, not a substitute for necessary escalation of conventional therapy.
These Scenarios Are Educational
These clinical scenarios are composite examples for educational purposes. They are not meant to replace individualized medical advice. The specific peptide doses, combinations, and durations described here should be adjusted based on individual patient factors, concurrent medications, and response to treatment. Always work with a qualified healthcare provider who can monitor response and adjust the protocol accordingly.
Scenario 4: The SIBO-Recurrence Patient
Presentation: A 52-year-old man has been treated for methane-dominant SIBO three times in the past 18 months. Each time, rifaximin plus neomycin clears the overgrowth on breath testing, but symptoms and positive breath tests recur within 3-4 months. He has a history of an appendectomy and suspected vagal nerve dysfunction. Gastric emptying study shows borderline delayed emptying.
Protocol focus: Prevention of recurrence
- Antimicrobial phase (Weeks 1-2): Rifaximin 550 mg TID + metronidazole 250 mg TID (for methane) + LL-37 100 mcg SC daily
- Repair and motility restoration (Weeks 3-12): BPC-157 500 mcg oral twice daily + VIP 100 mcg nasal spray twice daily (critical for MMC restoration) + Low-dose prucalopride 0.5 mg at bedtime (prokinetic)
- Long-term prevention: VIP 50 mcg nasal spray daily (ongoing) + PHGG 5 g daily + Strict meal spacing (3 meals, no snacking, 5-hour intervals) + Ginger capsule 1 g at bedtime (natural prokinetic)
Expected trajectory: Breath test negative at 4 weeks. The combination of VIP (restoring neurological motility control) with prokinetics and meal spacing addresses the root cause of recurrence rather than just treating each episode. This patient's history suggests enteric nervous system dysfunction, making VIP particularly important. Monitor with breath testing every 3-4 months initially, extending intervals as confidence in sustained clearance grows.
Scenario 5: The Multi-System "Leaky Gut" Patient
Presentation: A 40-year-old woman with progressively worsening food sensitivities (now reactive to 20+ foods), chronic fatigue, brain fog, eczema, and joint pain. No specific GI diagnosis. Testing shows markedly elevated lactulose/mannitol ratio, elevated serum zonulin, multiple elevated IgG food antibodies, low secretory IgA, and reduced microbiome diversity with low Akkermansia and Faecalibacterium.
Protocol:
- Phase 1 (Weeks 1-4): Strict AIP diet + LL-37 50 mcg SC daily (for antimicrobial support and microbiome modulation) + KPV 250 mcg SC daily (beginning anti-inflammatory support early)
- Phase 2-3 (Weeks 4-12): BPC-157 500 mcg oral twice daily + Larazotide 0.5 mg oral TID before meals + KPV 500 mcg oral daily (switch to oral route for gut-specific delivery) + L-glutamine 10 g daily + Vitamin D 8,000 IU daily (targeting 60+ ng/mL) + Colostrum 5 g daily
- Phase 4 (Weeks 12-20): Continue BPC-157 250 mcg daily + Continue larazotide TID + Begin careful food reintroduction (one food every 5 days) + Introduce probiotics (focus on Akkermansia-supporting polyphenols and Faecalibacterium-feeding fiber) + PHGG 5 g daily
- Maintenance: Mediterranean diet + BPC-157 250 mcg 3x/week + Larazotide before high-risk meals + Ongoing prebiotic and probiotic support
Expected trajectory: This is a longer healing journey - expect 4-6 months for significant improvement. Food sensitivities should begin improving by weeks 8-12 as permeability decreases. Extra-intestinal symptoms (fatigue, brain fog, eczema, joint pain) typically improve as the barrier heals and systemic inflammation from translocated antigens decreases - usually 2-4 weeks after GI symptoms begin improving. Repeat permeability testing at 8 and 16 weeks to confirm barrier restoration. IgG food antibodies should decline over 3-6 months as antigen translocation decreases.
Figure 10: Expected healing timeline for different symptom categories during a comprehensive gut-healing peptide protocol. GI symptoms typically improve first (weeks 2-6), followed by permeability markers (weeks 4-8), food sensitivities (weeks 8-12), and extra-intestinal symptoms (weeks 6-16).
Intestinal Permeability Testing: A Practical Guide
If you can't measure it, you can't manage it. Objective testing for gut barrier function has advanced considerably, but the landscape remains confusing for patients and even for some practitioners. Here's a detailed look at the available tests, what they actually measure, and how to interpret results in the context of peptide therapy.
The Lactulose-Mannitol Test: Gold Standard with Caveats
The lactulose-mannitol (L/M) test has been the most widely used clinical measure of intestinal permeability since the 1970s. The principle is straightforward: the patient drinks a solution containing two sugar probes of different molecular sizes. Mannitol (molecular weight 182 Da) is a small molecule that crosses the intestinal barrier easily through the transcellular pathway and tight junction pore pathway. Lactulose (molecular weight 342 Da) is larger and should only cross through the leak pathway - the pathway that becomes excessively permeable in barrier dysfunction (doi: 10.1038/s41575-023-00766-3).
After ingestion, urine is collected for 6 hours and analyzed for both sugars. The results are expressed as a ratio:
- Normal L/M ratio: Less than 0.03 (varies by laboratory)
- Elevated L/M ratio: Greater than 0.03 indicates increased paracellular permeability
- Low mannitol recovery: Suggests villous atrophy or reduced absorptive surface area (as in celiac disease or environmental enteropathy)
- High lactulose recovery: Directly indicates increased leak pathway permeability
The L/M test has several practical limitations that affect its use in clinical practice:
- Primarily measures small intestinal permeability - Lactulose and mannitol are absorbed primarily in the small intestine. Colonic permeability (relevant to UC and colonic inflammation) is less well captured by this test.
- Point-in-time measurement - Permeability fluctuates throughout the day and in response to meals, stress, and other factors. A single test may not capture the overall picture.
- Urine collection compliance - Complete 6-hour urine collection is essential for accurate results. Incomplete collection leads to falsely low recovery of both markers.
- Medication interference - NSAIDs, aspirin, and alcohol can acutely increase permeability, potentially confounding results if taken close to the test.
- Renal function dependence - Since results depend on urinary excretion, impaired kidney function can affect marker recovery.
Multi-Sugar Permeability Tests
To address the limitations of the two-sugar test, researchers have developed multi-sugar tests that simultaneously assess permeability at different GI locations:
| Sugar Probe | Primary Site Assessed | What Elevated Recovery Indicates |
|---|---|---|
| Sucrose | Gastric and duodenal mucosa | Upper GI permeability increase (gastritis, duodenitis, NSAID damage) |
| Lactulose / Mannitol ratio | Small intestine | Small intestinal permeability increase (celiac, SIBO, IBD) |
| Sucralose | Colon (and whole gut) | Colonic and whole-gut permeability (UC, colonic inflammation) |
| Erythritol / Rhamnose | Alternative small intestinal probes | Similar to L/M but with better stability and assay characteristics |
The multi-sugar approach can identify which segment of the GI tract has compromised barrier function, guiding peptide selection more precisely. For example, elevated sucrose recovery (upper GI damage) might prioritize oral BPC-157 for gastric cytoprotection, while elevated sucralose recovery (colonic damage) might prioritize KPV and larazotide for colonic inflammation and permeability.
Blood-Based Permeability Markers
Several blood-based biomarkers can complement sugar permeability testing:
Zonulin
As discussed earlier, zonulin is the endogenous regulator of tight junction permeability. Elevated serum zonulin levels indicate increased tight junction opening and predict response to larazotide therapy. However, zonulin assays have been controversial - a 2019 study found that commercial "zonulin" ELISA kits may actually detect related but different proteins (haptoglobin precursors), leading to questions about assay specificity. Despite these concerns, serial measurements using the same assay from the same laboratory can track trends over time, which is clinically useful even if absolute values vary between assays.
Lipopolysaccharide-Binding Protein (LBP)
LBP is produced by the liver in response to LPS (lipopolysaccharide, also called endotoxin) from gram-negative bacteria. When the gut barrier is compromised, LPS translocates from the lumen into the portal circulation, triggering LBP production. Elevated LBP is an indirect marker of bacterial translocation across a leaky gut barrier. Normal serum LBP levels are typically 5-15 mcg/mL; levels above 20 mcg/mL suggest increased bacterial translocation.
Anti-Endotoxin Antibodies (EndoCAb)
IgM, IgG, and IgA antibodies against LPS core antigens can be measured to assess chronic endotoxin exposure. Elevated IgM anti-endotoxin antibodies suggest acute bacterial translocation, while elevated IgG levels suggest chronic exposure. A pattern of high IgG with low IgM may indicate ongoing chronic translocation with an exhausted acute immune response.
Intestinal Fatty Acid Binding Protein (I-FABP)
I-FABP is a cytoplasmic protein released into the bloodstream when enterocytes are damaged. Elevated serum I-FABP is a direct marker of intestinal epithelial cell injury - not just permeability, but actual cell damage. This makes it useful for assessing the severity of mucosal injury and monitoring response to repair-oriented peptides like BPC-157.
Citrulline
Plasma citrulline is produced almost exclusively by small intestinal enterocytes. Low plasma citrulline levels indicate reduced functional enterocyte mass - as seen in celiac disease, radiation enteritis, short bowel syndrome, and severe small intestinal inflammation. Citrulline can serve as a marker of small intestinal absorptive capacity and mucosal health, tracking recovery during peptide-supported healing.
Stool-Based Markers: Beyond Calprotectin
While fecal calprotectin is the most commonly used stool marker for intestinal inflammation, several additional stool markers provide complementary information:
Fecal Lactoferrin
Like calprotectin, lactoferrin is a neutrophil-derived protein that indicates intestinal inflammation. Some studies suggest it may be more specific than calprotectin for IBD-related inflammation versus other causes of elevated calprotectin (like NSAID use). Levels above 7.25 mcg/g are considered positive.
Fecal Alpha-1 Antitrypsin
Alpha-1 antitrypsin is a serum protein that's normally excluded from the intestinal lumen by intact tight junctions. Elevated fecal levels indicate protein-losing enteropathy - leakage of serum proteins across a compromised barrier. This is a direct functional measure of barrier failure rather than an inflammatory marker.
Fecal Secretory IgA
Secretory IgA (sIgA) is the primary immunoglobulin at mucosal surfaces, providing the first line of adaptive immune defense in the gut lumen. Low fecal sIgA may indicate impaired mucosal immunity and reduced ability to control luminal pathogens and antigens. Chronically low sIgA is associated with increased susceptibility to GI infections and food sensitivities. Peptides like thymosin alpha-1 can support sIgA production through immune modulation.
Beta-Glucuronidase
This bacterial enzyme, produced predominantly by certain gram-negative bacteria, is involved in estrogen recirculation and can indicate dysbiosis when elevated. While not a direct permeability marker, elevated beta-glucuronidase suggests a shift toward a more inflammatory microbiome ecology that often accompanies barrier dysfunction.
Creating a Testing Protocol
For a comprehensive gut healing protocol supported by peptide therapy, the following testing schedule provides optimal feedback:
| Timepoint | Tests | Purpose |
|---|---|---|
| Baseline (before starting) | Multi-sugar permeability test, comprehensive stool analysis (calprotectin, sIgA, microbiome, pancreatic elastase), blood panel (CRP, LBP, vitamin D, citrulline, I-FABP), SIBO breath test if indicated | Establish baseline, identify all contributing factors, guide peptide selection |
| Week 4 | Symptom assessment, SIBO breath test (if treating SIBO), basic blood panel (CRP) | Confirm antimicrobial response, assess early symptom trajectory |
| Week 8 | Repeat permeability test, fecal calprotectin, CRP, I-FABP | Quantify barrier improvement, confirm inflammation reduction |
| Week 12-16 | Full repeat of baseline testing panel | Comprehensive reassessment; guide transition to maintenance phase |
| 6 months | Permeability test, stool analysis, symptom review | Confirm sustained improvement off active protocol; guide long-term management |
Peptide Sourcing and Quality: What to Look For
The gap between a high-quality peptide and a contaminated or degraded one can mean the difference between therapeutic benefit and wasted money - or worse, adverse effects from impurities. Quality assurance is non-negotiable when using research peptides.
Certificate of Analysis (COA)
Every peptide should come with a current Certificate of Analysis from an independent third-party laboratory. The COA should include:
- Identity confirmation - Mass spectrometry (MS) confirming the correct molecular weight matches the target peptide sequence. Electrospray ionization mass spectrometry (ESI-MS) or matrix-assisted laser desorption/ionization (MALDI-TOF) are standard methods.
- Purity assessment - High-Performance Liquid Chromatography (HPLC) showing peptide purity. For therapeutic use, purity should be greater than 98%. Lower purity means higher concentrations of deletion sequences, truncated peptides, and synthesis byproducts.
- Amino acid analysis - Confirms the correct amino acid composition and ratios, verifying that the synthesized peptide matches the intended sequence.
- Endotoxin testing - Particularly critical for injectable peptides. Bacterial endotoxin (LPS) contamination from the manufacturing process can cause fever, inflammation, and immune reactions. Acceptable endotoxin levels for injectables are less than 5 EU/kg body weight.
- Sterility testing - For injectable formulations, sterility must be confirmed. Bacterial contamination in an injectable peptide can cause serious infections.
- Moisture content - Excessive moisture in lyophilized peptides accelerates degradation. Moisture content should typically be less than 5%.
Storage and Handling
Peptide stability depends heavily on proper storage:
| Form | Storage Temperature | Expected Stability | Notes |
|---|---|---|---|
| Lyophilized powder (unopened) | -20C (freezer) | 2+ years | Most stable form; protect from moisture |
| Lyophilized powder (opened) | -20C (freezer), desiccated | 6-12 months | Use desiccant packets; minimize exposure to air |
| Reconstituted solution | 2-8C (refrigerator) | 2-4 weeks (peptide dependent) | Use bacteriostatic water for multi-dose vials; avoid repeated freeze-thaw |
| Oral capsules | Room temperature or refrigerator | Varies by formulation | Enteric coating may be needed for acid-labile peptides |
BPC-157 is notable for its exceptional stability - it remains active in gastric juice for over 24 hours and has good shelf stability in lyophilized form. KPV, as a small tripeptide, is also relatively stable. VIP and LL-37 are more fragile and require careful cold-chain handling.
Red Flags in Peptide Sourcing
Be wary of the following warning signs when evaluating peptide suppliers:
- No third-party COA available, or COA from an in-house laboratory only
- Purity listed below 95% without explanation
- No endotoxin testing for injectable products
- Dramatically lower prices than established suppliers (may indicate lower purity, smaller quantities, or cutting with inactive filler)
- No clear information about manufacturing standards (look for GMP or GMP-equivalent processes)
- Marketing making specific medical claims or guaranteeing outcomes (reputable suppliers sell research compounds, not medicines)
- No proper labeling with batch numbers, expiration dates, and storage instructions
FormBlends provides third-party tested peptides with full COA documentation for all products in the gut-healing category.
Reconstitution Best Practices
For injectable peptides requiring reconstitution from lyophilized powder:
- Use appropriate diluent - Bacteriostatic water (BAC water, containing 0.9% benzyl alcohol as preservative) for multi-dose vials. Sterile water for single-use reconstitution. Normal saline (0.9% NaCl) is also acceptable for most peptides.
- Calculate concentration - Add enough diluent to achieve a practical concentration for your dosing needs. For example, adding 2 mL of BAC water to a 5 mg vial of BPC-157 gives a concentration of 2.5 mg/mL (2,500 mcg/mL). A 500 mcg dose would then be 0.2 mL (20 units on a 1 mL insulin syringe).
- Gentle mixing - Direct the diluent down the side of the vial, not directly onto the lyophilized cake. Swirl gently rather than shaking vigorously, as agitation can damage peptide structure.
- Refrigerate immediately - After reconstitution, store at 2-8C and use within 2-4 weeks.
- Use appropriate syringes - Insulin syringes (29-31 gauge, 1 mL or 0.5 mL) are standard for subcutaneous peptide injections. The thin needles minimize discomfort and tissue damage.
Oral vs. Injectable Preparation
For oral BPC-157, many practitioners use pre-made oral capsules or tablets rather than reconstituted solutions. These formulations often include enteric coating or acid-stable encapsulation to protect the peptide through gastric transit. For patients who prefer to use lyophilized BPC-157 orally, the reconstituted solution can be taken by mouth - simply measure the dose into a small amount of water and drink on an empty stomach. BPC-157's stability in gastric acid means no special acid protection is needed for this particular peptide.
Historical Context: How Gut Peptide Therapy Evolved
The use of peptides for gut healing didn't emerge from a vacuum. It grew from decades of research in gastroenterology, immunology, and peptide science. Understanding this history provides context for where the field stands today and where it's heading.
The Cytoprotection Era (1970s-1990s)
The concept that the gut can be pharmacologically protected from injury - independent of acid suppression - was established by Andre Robert in 1979. Working at Upjohn Laboratories, Robert demonstrated that small doses of prostaglandins could prevent gastric mucosal injury from a wide variety of damaging agents, including ethanol, acids, bases, and boiling water. He termed this phenomenon "gastric cytoprotection."
Robert's work established several principles that remain relevant to peptide therapy today: (1) the gut has endogenous protective mechanisms that can be pharmacologically enhanced, (2) cytoprotection works through multiple mechanisms beyond simple barrier function, and (3) effective cytoprotection can be achieved at doses far below those needed for anti-secretory effects.
BPC-157's discovery in the early 1990s by the Zagreb group built directly on Robert's cytoprotection framework. The peptide was identified as a partial sequence of a protein naturally present in human gastric juice - essentially an endogenous cytoprotective agent that the stomach itself produces. This gave BPC-157 an intellectual pedigree within mainstream gastroenterology, even as its research remained primarily preclinical.
The Leaky Gut Revolution (1990s-2010s)
For decades, "leaky gut" was dismissed by mainstream medicine as a fringe concept. That changed with two major developments. First, the development of reliable permeability testing (lactulose-mannitol and multi-sugar tests) allowed researchers to objectively measure barrier dysfunction. Second, Alessio Fasano's discovery of zonulin in 2000 provided a molecular mechanism for how intestinal permeability is regulated - and how it goes wrong in disease.
Fasano's work showed that increased intestinal permeability precedes the development of autoimmune diseases like type 1 diabetes and celiac disease - suggesting that barrier dysfunction isn't just a consequence of disease but a causal contributor. This shifted the conversation from "leaky gut is a myth" to "intestinal permeability is a measurable, modifiable risk factor for disease."
The development of larazotide acetate grew directly from this research. If zonulin-mediated tight junction opening drives disease, then a zonulin antagonist should prevent or reduce disease activity. The clinical trials in celiac disease validated this hypothesis, establishing the first pharmacological tight junction regulator in clinical development.
The Microbiome-Barrier Connection (2010s-Present)
The explosion of microbiome research in the 2010s revealed the intimate bidirectional relationship between the gut microbiome and barrier function. Studies showed that specific bacterial species (like Akkermansia muciniphila) directly strengthen the barrier, while dysbiosis promotes barrier dysfunction. This connection explained why antibiotic use, dietary changes, and probiotic therapy could all affect intestinal permeability.
For peptide therapy, this research highlighted the importance of antimicrobial peptides like LL-37 in maintaining the microbiome-barrier equilibrium. It also emphasized that barrier repair (BPC-157, larazotide) and microbiome restoration must go hand in hand for lasting results.
The PepT1 Discovery and KPV (2008)
The 2008 demonstration by Dalmasso et al. that KPV is transported into inflamed intestinal cells via PepT1 was a watershed moment. It showed that a naturally-derived anti-inflammatory peptide could be selectively delivered to exactly the tissue that needs it most - without requiring complex drug delivery systems or targeting technologies. The inflamed gut essentially targets itself for treatment by upregulating the very transporter that carries the therapeutic peptide.
This principle - using the disease's own molecular changes as a delivery mechanism - represents an elegant therapeutic strategy that could be applied to other peptides and conditions in the future.
The VIP Renaissance (2015-Present)
Although VIP was discovered in 1970 and has been studied extensively since, its therapeutic applications were limited by its extremely short half-life. The renewed interest in VIP for gut conditions stems from several converging factors: the recognition that motility disorders underlie many chronic GI conditions (particularly SIBO), the discovery of VIP's role in microbiome-motility communication, and the development of practical delivery systems (nasal spray) that make therapeutic VIP administration feasible.
Dr. Ritchie Shoemaker's incorporation of VIP nasal spray into the CIRS (Chronic Inflammatory Response Syndrome) protocol - where it improved not only respiratory but also gastrointestinal symptoms - brought VIP to the attention of a broader clinical community. This cross-pollination between environmental medicine and gastroenterology expanded the range of conditions for which VIP therapy is considered.
Where We Are Now
As of early 2026, gut peptide therapy sits at an interesting inflection point. The preclinical evidence is extensive, particularly for BPC-157. Clinical trial data exists for larazotide (the most advanced). Practitioner experience with peptide protocols is growing rapidly. But large-scale human clinical trials for most compounds remain in the future.
The field is moving from empirical use - based on preclinical data and clinical experience - toward evidence-based protocols supported by human trial data. This transition will likely accelerate over the next 3-5 years as interest from pharmaceutical companies and academic institutions grows. In the meantime, informed practitioners and patients can use the available evidence thoughtfully while contributing to the growing body of real-world clinical experience.
Head-to-Head Peptide Comparison for Gut Healing
With five major gut-healing peptides to choose from, knowing which one (or which combination) is right for a given situation requires understanding their relative strengths, weaknesses, and evidence bases. This section provides a detailed side-by-side comparison to guide clinical decision-making.
Comprehensive Comparison Table
| Parameter | BPC-157 | KPV | Larazotide | VIP | LL-37 |
|---|---|---|---|---|---|
| Size | 15 amino acids | 3 amino acids (tripeptide) | 8 amino acids (octapeptide) | 28 amino acids | 37 amino acids |
| Origin | Human gastric juice protein fragment | C-terminal of alpha-MSH | Derived from V. cholerae ZOT (homolog of human zonulin) | Enteric neurons, immune cells | Human cathelicidin precursor hCAP-18 |
| Primary mechanism | NO modulation, angiogenesis, growth factor upregulation, cytoprotection | NF-kB and MAPK inhibition via PepT1 transport | Zonulin receptor antagonism, tight junction stabilization | VPAC1/VPAC2 receptor agonism, neuromodulation | Membrane disruption of microbes, immune modulation |
| GI stability | Excellent (24+ hours in gastric acid) | Moderate (may be partially degraded) | Good (designed for luminal action) | Poor (rapidly degraded orally) | Poor (degraded by digestive proteases) |
| Preferred route for gut | Oral | Oral or subcutaneous | Oral | Nasal spray | Subcutaneous |
| Human trial data | Very limited (no Phase III) | None (preclinical only) | Phase II/III completed | Limited (approved for diagnostic use only) | None for gut indications |
| Preclinical evidence strength | Very strong (100+ studies) | Moderate (key studies solid) | Strong (plus human trials) | Moderate (mostly mechanistic) | Moderate |
| Best for | Ulcers, IBD, fistulas, general mucosal repair, GERD, anastomotic healing | Active colonic inflammation, UC, NF-kB driven inflammation | Celiac disease, tight junction dysfunction, food sensitivities, leaky gut | SIBO, motility disorders, CIRS-related gut dysfunction | Dysbiosis, gut infections, antimicrobial support, microbiome modulation |
| Systemic absorption | Moderate (oral); full (SC) | Low (oral); full (SC) | Minimal (<1%) | Full (nasal); poor (oral) | Full (SC) |
| Onset of action | Days to weeks | Days to weeks | Hours to days | Minutes to hours | Days |
| Cost range (monthly) | $$ - $$$ | $$ - $$$ | $$ - $$$ | $$$ - $$$$ | $$$ - $$$$ |
Decision Framework: Selecting the Right Peptide
Use this decision framework to identify the highest-priority peptide for your situation:
- If mucosal damage is the primary issue (ulcers, erosions, villous atrophy, post-surgical healing) - start with BPC-157 oral
- If active inflammation is dominant (elevated calprotectin, bloody stools, active IBD flare) - start with KPV
- If permeability/food sensitivities are the primary concern (elevated L/M ratio, zonulin, multiple food reactions) - start with larazotide
- If motility dysfunction is driving symptoms (recurrent SIBO, slow transit, gastroparesis) - start with VIP nasal spray
- If dysbiosis/infection is the root cause (pathogenic overgrowth, biofilm infections, post-antibiotic dysbiosis) - start with LL-37
Once the primary peptide is established and tolerated (usually after 1-2 weeks), add the next highest-priority peptide based on remaining mechanisms of dysfunction. Most patients benefit most from 2-3 peptides rather than all five.
Monotherapy vs. Combination: When Is One Peptide Enough?
Monotherapy with a single peptide may be sufficient for:
- Mild, uncomplicated conditions (mild gastritis, mild IBS with no SIBO)
- Budget-constrained patients who need to prioritize
- Initial assessment - starting with one peptide helps identify individual response before adding complexity
- Maintenance therapy after completing a full combination protocol
Combination therapy is generally recommended for:
- Moderate to severe IBD
- Multi-system dysfunction (gut + autoimmune + neurological symptoms)
- Treatment-resistant conditions that haven't responded to conventional therapy
- Conditions with multiple concurrent mechanisms (e.g., SIBO + leaky gut + inflammation)
- Crohn's disease (transmural inflammation requires multiple approaches)
Athlete Gut Health and Peptide Applications
Endurance athletes represent a unique population for gut health peptide therapy. Exercise-induced gastrointestinal syndrome affects 30-70% of endurance athletes during training and competition, with symptoms ranging from bloating and cramping to bloody diarrhea and hospitalization for ischemic colitis.
Exercise-Induced Gut Damage
During intense or prolonged exercise, blood is redirected from the splanchnic (gut) circulation to working muscles, skin, and heart. This splanchnic hypoperfusion can reduce intestinal blood flow by up to 80% during maximal exercise. The resulting ischemia damages enterocytes, disrupts tight junctions, and increases intestinal permeability - a phenomenon documented in marathon runners, triathletes, cyclists, and even during intense CrossFit-style workouts.
The damage follows a predictable ischemia-reperfusion pattern:
- Ischemic phase (during exercise) - Reduced blood flow causes enterocyte hypoxia, ATP depletion, and cell swelling
- Reperfusion phase (post-exercise) - Restored blood flow generates reactive oxygen species (ROS) that further damage cells and tight junctions, triggering an inflammatory cascade
- Translocation phase - Increased permeability allows LPS and other bacterial products to enter the portal circulation, causing systemic inflammation (exercise-induced endotoxemia)
This exercise-induced barrier damage is exacerbated by heat stress, NSAID use (common among athletes for pain management), dehydration, and high-carbohydrate sports nutrition products that may contain emulsifiers and artificial sweeteners.
Peptide Strategy for Athletes
BPC-157 is the most relevant peptide for athletic gut protection because its mechanisms directly counteract exercise-induced gut damage:
- NO-mediated vasodilation helps maintain mucosal blood flow during exercise
- Angiogenesis promotes development of collateral blood supply, improving the gut's resilience to ischemic stress
- Cytoprotection protects enterocytes against ischemia-reperfusion injury
- Anti-inflammatory effects reduce the post-exercise inflammatory cascade
- NSAID counteraction - for athletes who continue to use NSAIDs, BPC-157 can protect the gut from NSAID-induced damage while the NSAID provides musculoskeletal pain relief
Recommended Protocol for Endurance Athletes
| Timing | Peptide/Supplement | Purpose |
|---|---|---|
| Daily (training blocks) | BPC-157 250-500 mcg oral, morning on empty stomach | Ongoing mucosal protection and repair |
| Pre-race (24 hours before) | BPC-157 500 mcg oral + L-glutamine 10 g | Pre-loading mucosal protection before extreme stress |
| Post-race/post-intense session | BPC-157 500 mcg oral + zinc carnosine 75 mg | Accelerate repair of exercise-induced damage |
| Recovery week | BPC-157 250 mcg oral daily + colostrum 5 g daily | Support mucosal recovery during reduced training load |
Additional strategies for athlete gut health include:
- Gut training - Systematically practicing race-day nutrition during training to adapt the gut to high-carbohydrate intake during exercise
- Avoid NSAIDs before/during exercise - NSAIDs dramatically increase exercise-induced gut permeability. Use acetaminophen as an alternative for pain when needed, or better yet, address pain through BPC-157, TB-500, and physical therapy.
- Heat acclimatization - Gradual heat exposure improves gut resilience and reduces heat-induced barrier dysfunction
- Pre-exercise nutrition timing - Allow 2-3 hours between the last solid meal and intense exercise to ensure adequate gastric emptying
- Hydration - Dehydration exacerbates splanchnic hypoperfusion; maintaining hydration status is critical for gut protection
Anti-Doping Considerations for Athletes
Athletes subject to anti-doping regulations should be aware that the regulatory status of therapeutic peptides in competitive sport varies by governing body and is subject to change. As of early 2026:
- BPC-157 - Listed by WADA (World Anti-Doping Agency) under S0 (non-approved substances) category, which prohibits substances not approved for human therapeutic use by any governmental regulatory authority. Competitive athletes should avoid BPC-157 during competition periods and within any required washout windows.
- VIP, KPV, LL-37, Larazotide - Anti-doping classification may vary. Athletes should consult with their sport's anti-doping authority and a sports medicine physician before using any peptide.
For recreational athletes not subject to anti-doping testing, these considerations are less relevant, but it's worth being aware of the broader regulatory context.
The Athlete's Gut: A Summary
Athletes push their bodies hard, and the gut often bears the consequences. Exercise-induced gut damage is real, measurable, and preventable. BPC-157's combination of cytoprotective, anti-inflammatory, and mucosal repair properties makes it particularly well-suited for athletes dealing with exercise-induced GI symptoms. Combined with smart training practices (gut training, avoiding NSAIDs, proper hydration, heat acclimatization), peptide support can help athletes train harder and race better without being derailed by GI distress.
Gut Healing Peptides in Special Populations
Age, sex, and life stage all influence gut barrier function and the approach to peptide therapy. While the core principles remain the same, several populations require special considerations in protocol design.
Women's Gut Health and Hormonal Influences
The gut barrier is exquisitely sensitive to sex hormones, and many women experience cyclical changes in GI symptoms that correlate with their menstrual cycle. Estrogen generally supports barrier function by enhancing tight junction protein expression and promoting mucosal blood flow. Progesterone modulates gut motility, slowing transit in the luteal phase. These hormonal effects explain several clinical observations:
- Premenstrual GI symptoms - The drop in estrogen and progesterone just before menstruation is associated with increased intestinal permeability, more frequent bowel movements, and worsening of IBS symptoms in many women. BPC-157's cytoprotective effects and larazotide's tight junction stabilization may help buffer against these cyclical permeability changes.
- Perimenopause and menopause - Declining estrogen levels during perimenopause are associated with increased intestinal permeability, altered microbiome composition (particularly reduced Lactobacillus species), and increased systemic inflammation. Many women develop new GI symptoms or worsening of existing conditions during this transition. A gut-healing peptide protocol during perimenopause - particularly BPC-157 for mucosal support and KPV for anti-inflammatory effects - may help manage the GI component of the menopausal transition.
- Endometriosis and gut symptoms - Endometriosis frequently causes GI symptoms that mimic IBS, including bloating, diarrhea, constipation, and abdominal pain. The inflammatory nature of endometriosis can affect gut barrier function through elevated systemic inflammatory cytokines. KPV's anti-inflammatory properties may provide benefit for the GI symptoms associated with endometriosis, though this application has not been specifically studied.
- Polycystic ovary syndrome (PCOS) - PCOS is associated with increased intestinal permeability and altered microbiome composition, which may contribute to the metabolic inflammation that characterizes the condition. Addressing gut barrier function with peptides could theoretically improve the inflammatory and metabolic aspects of PCOS, though direct evidence is lacking.
Contraception and Peptide Interactions
There is no evidence that gut-healing peptides interact with oral contraceptives. However, patients with significantly impaired intestinal absorption (severe IBD, short bowel syndrome) may have reduced absorption of oral contraceptives - and improved absorption during gut healing could theoretically alter contraceptive drug levels. Women in this situation should discuss with their prescribing physician and consider non-oral contraceptive methods during active gut healing.
Gut Health in Older Adults
Aging is associated with progressive changes in gut barrier function that increase vulnerability to GI conditions and systemic inflammation:
- Reduced mucosal regeneration - Intestinal stem cell function declines with age, slowing the 3-5 day epithelial turnover rate and reducing the gut's ability to repair damage. BPC-157's growth factor-enhancing properties may help compensate for this age-related decline.
- Decreased antimicrobial peptide production - Aging Paneth cells produce fewer defensins and cathelicidins, weakening mucosal defense. LL-37 supplementation could address this deficit.
- Microbiome shifts - The aged microbiome typically shows reduced diversity, decreased Bifidobacterium and Faecalibacterium, and increased Proteobacteria - changes associated with increased barrier permeability and systemic inflammation ("inflammaging").
- Polypharmacy - Older adults often take multiple medications, many of which affect gut barrier function (NSAIDs, PPIs, antibiotics, metformin, statins). BPC-157's ability to counteract medication-induced gut damage is particularly relevant in this population.
- Reduced gastric acid - Achlorhydria and hypochlorhydria become more common with aging, predisposing to SIBO and impaired nutrient absorption. VIP and prokinetic support may help maintain motility, while betaine HCl supplementation addresses acid insufficiency.
Dosing Considerations for Older Adults
While no age-specific dosing data exists for gut-healing peptides, general principles for older adults include:
- Start at the lower end of dosing ranges (reduced renal clearance may prolong peptide half-life)
- Titrate upward more slowly (reduced regenerative capacity means the body needs more time to respond)
- Monitor more closely for blood pressure effects (VIP and BPC-157 both affect vascular tone, and older adults are more sensitive to hemodynamic changes)
- Review all medications for potential interactions, particularly anticoagulants (BPC-157's effects on blood vessel formation could theoretically interact), antihypertensives (VIP's vasodilatory effects), and immunosuppressants (KPV's NF-kB inhibition)
- Ensure adequate vitamin D status (essential for LL-37 production; deficiency is extremely common in older adults)
Gut Health in Children and Adolescents
Pediatric gut conditions - including pediatric IBD, celiac disease, food allergies, and functional abdominal pain - are increasing in prevalence. While the biological mechanisms are similar to adult conditions, peptide therapy in children requires extreme caution:
- No pediatric dosing data exists for any of the peptides discussed in this report
- Growth and development considerations - BPC-157's effects on growth factors and angiogenesis could theoretically affect developing tissues in ways that are unpredictable
- Immune system maturation - KPV's NF-kB inhibition and LL-37's immune modulation could affect the developing immune system, potentially interfering with normal immune education and tolerance development
- Long-term safety unknown - The effects of chronic peptide exposure during development have not been studied
For pediatric gut conditions, the following non-peptide approaches are safer and better supported:
- Dietary interventions (specific carbohydrate diet for IBD, gluten-free diet for celiac, elimination diets for food sensitivities)
- Probiotics with pediatric evidence (Lactobacillus rhamnosus GG, Bifidobacterium infantis, VSL#3)
- Glutamine supplementation (generally considered safe in children at 0.3-0.5 g/kg/day)
- Zinc supplementation (within age-appropriate RDA ranges)
- Vitamin D optimization
- Conventional medications with established pediatric safety profiles (mesalamine, corticosteroids, biologics for IBD)
If peptide therapy is considered for a pediatric patient with a severe, treatment-resistant condition, it should only be undertaken by a pediatric gastroenterologist with expertise in peptide therapeutics, with careful documentation and monitoring. Larazotide, which has the most favorable safety profile due to minimal systemic absorption, would theoretically be the safest option if a peptide were to be used.
Gut Health During Chronic Illness
Chronic illnesses frequently involve gut barrier dysfunction as both a cause and consequence of the disease process. Several conditions deserve specific mention:
Chronic Fatigue Syndrome / Myalgic Encephalomyelitis (CFS/ME)
Multiple studies have documented increased intestinal permeability, altered microbiome, and elevated LPS-binding protein in CFS/ME patients. The resulting bacterial translocation and systemic immune activation may contribute to the fatigue, cognitive dysfunction, and post-exertional malaise that characterize the condition. A gut-healing protocol with BPC-157 (mucosal repair), larazotide (permeability reduction), and VIP (if CIRS overlap is suspected) may address this component of CFS/ME pathophysiology.
Mast Cell Activation Syndrome (MCAS)
MCAS frequently causes GI symptoms through mast cell degranulation in the gut mucosa, which releases histamine, tryptase, and other mediators that directly open tight junctions and trigger inflammation. KPV's anti-inflammatory effects (NF-kB inhibition) can help dampen the mast cell-driven inflammatory cascade, while VIP has mast cell-stabilizing properties. Larazotide's tight junction stabilization helps prevent the permeability increase caused by mast cell mediators. Patients with MCAS should start peptides at very low doses, as they may be more sensitive to peptide-induced immune modulation.
Autoimmune Conditions
The connection between intestinal permeability and autoimmune disease - established through zonulin research - suggests that gut barrier repair could help modulate autoimmune disease activity. For conditions like Hashimoto's thyroiditis, rheumatoid arthritis, and lupus, where increased intestinal permeability allows passage of antigens that may drive molecular mimicry or immune activation, a barrier-focused approach (larazotide + BPC-157) combined with immune modulation (KPV for inflammation, thymosin alpha-1 for immune balance) represents a rational strategy. The 2020 Nature Communications study showing that targeting zonulin prevented arthritis onset in a preclinical model provides direct support for this approach in at least one autoimmune condition.
Type 2 Diabetes and Metabolic Syndrome
Hyperglycemia directly impairs intestinal barrier function through advanced glycation end-products and altered tight junction protein expression. The resulting bacterial translocation contributes to the chronic low-grade inflammation ("metabolic endotoxemia") that drives insulin resistance - creating a vicious cycle where metabolic disease damages the gut, and the damaged gut worsens metabolic disease. BPC-157's cytoprotective effects and larazotide's permeability reduction can help break this cycle. Interestingly, NAD+ supplementation may support both metabolic function and cellular energy for gut repair simultaneously.
Post-COVID Gut Dysfunction
SARS-CoV-2 infects intestinal epithelial cells through the ACE2 receptor, which is highly expressed on the brush border of small intestinal enterocytes. Acute COVID-19 can cause direct intestinal damage, increased permeability, and dysbiosis. In long COVID, persistent GI symptoms (bloating, diarrhea, food sensitivities, nausea) affect up to 30% of patients, often accompanied by measurably increased intestinal permeability. A comprehensive gut-healing protocol with BPC-157, KPV, and larazotide - potentially combined with LL-37 for antimicrobial support and immune modulation - is a rational approach for post-COVID gut dysfunction, though specific studies in this population are needed.
Individualization Is Everything
The common thread across all special populations is that one-size-fits-all protocols don't exist. The peptides are tools, and tools need to be used appropriately for the specific job at hand. An endurance athlete with exercise-induced gut damage needs a different approach than a menopausal woman with new-onset food sensitivities, which is different from an elderly patient with NSAID-induced gastropathy. Comprehensive assessment - including testing, medical history, current medications, and individual goals - should always guide peptide selection, dosing, and monitoring. Use the FormBlends peptide calculator as a starting point, and work with a practitioner who can customize your protocol.
Frequently Asked Questions
Which peptide is best for leaky gut?
For intestinal permeability specifically, larazotide acetate has the strongest evidence because it directly stabilizes tight junctions - the molecular structures that seal the spaces between intestinal epithelial cells. It's the only gut-healing peptide with human clinical trial data showing reduced permeability. For a broader healing approach, combining larazotide with oral BPC-157 (which repairs mucosal damage and promotes angiogenesis) covers both barrier sealing and tissue repair. If you're choosing just one peptide, BPC-157 oral is the most versatile single option because it provides cytoprotection, anti-inflammatory effects, and mucosal healing - all contributing to reduced permeability over time.
Can BPC-157 fix leaky gut on its own?
BPC-157 can significantly improve gut barrier function through its cytoprotective, anti-inflammatory, and mucosal healing properties. Preclinical studies consistently show that it reduces intestinal permeability and heals mucosal damage across multiple GI models. However, "fixing" leaky gut requires more than peptides alone. If the root causes of barrier dysfunction persist - poor diet, NSAID use, chronic stress, dysbiosis, or untreated infections - no peptide will produce lasting results. BPC-157 works best as part of a comprehensive protocol that includes dietary changes, stress management, and identification and removal of ongoing triggers. Think of it as a powerful accelerator of healing rather than a standalone fix.
Is oral or injectable BPC-157 better for gut conditions?
For gut-specific conditions, oral BPC-157 is generally preferred. The peptide is stable in gastric acid for over 24 hours, which means it survives transit through the stomach and makes direct contact with the intestinal mucosa - exactly where you want it for gut healing. Most preclinical studies demonstrating GI benefits used intragastric (oral) administration. For severe conditions with deep tissue involvement, like Crohn's disease with fistulas, many practitioners use both oral and subcutaneous routes simultaneously. The oral dose targets the mucosal surface directly, while the injection ensures systemic delivery of growth factors and anti-inflammatory mediators to deeper tissue layers.
How long does it take for gut-healing peptides to work?
Most patients begin noticing symptom improvements within 2-4 weeks of starting a gut-healing peptide protocol, with significant improvement typically occurring by 6-8 weeks. The timeline depends on several factors: the severity of the condition, how many root causes are being addressed simultaneously, which peptides are being used, and individual healing capacity. Mild conditions like gastritis or mild IBS may respond within 2-3 weeks. Moderate conditions like active IBD or significant leaky gut typically require 6-12 weeks. Severe conditions like Crohn's fistulas may take 12 or more weeks. Objective markers like calprotectin and permeability testing can confirm healing is occurring even before symptoms fully resolve. The gut epithelium turns over every 3-5 days, so the biological capacity for rapid healing exists - the key is providing the right conditions.
What is KPV peptide and how does it reduce gut inflammation?
KPV (Lys-Pro-Val) is a three-amino-acid peptide derived from the tail end of alpha-melanocyte-stimulating hormone (alpha-MSH). It reduces gut inflammation through a unique mechanism: it enters inflamed intestinal cells through the PepT1 transporter - a peptide transporter that's normally present in the small intestine but becomes upregulated in inflamed colonic tissue during IBD. Once inside the cell, KPV inhibits NF-kB nuclear translocation and MAP kinase signaling, effectively shutting down two of the most critical inflammatory cascades at their convergence point. This reduces production of TNF-alpha, IL-6, and other pro-inflammatory cytokines. The beauty of this mechanism is that KPV preferentially accumulates in inflamed tissue (where PepT1 is upregulated), creating a self-targeting anti-inflammatory effect.
Can peptides help with SIBO?
Yes, peptides can meaningfully support SIBO treatment and prevention, though they work best as part of a comprehensive approach rather than as standalone treatments. VIP nasal spray helps restore the migrating motor complex (MMC) - the "cleaning wave" that normally sweeps bacteria from the small intestine between meals. Impaired MMC function is one of the primary reasons SIBO develops and recurs. LL-37 provides antimicrobial support that can enhance the effectiveness of herbal or pharmaceutical antimicrobials during the eradication phase. BPC-157 repairs the mucosal damage that SIBO causes in the small intestine. The most impactful peptide for SIBO prevention is arguably VIP, because it addresses the motility dysfunction that is often the root cause of recurrence.
What does larazotide do for celiac disease?
Larazotide acetate is a tight junction-regulating peptide derived from zonulin - the body's own protein that controls intestinal permeability. In celiac disease, gluten triggers excessive zonulin release, which opens tight junctions and allows gluten peptides to cross the intestinal barrier and trigger an immune reaction. Larazotide blocks the zonulin receptor, preventing this tight junction opening. In Phase II clinical trials, the 0.5 mg dose taken three times daily reduced symptoms in celiac patients on a gluten-free diet who continued to experience problems. It showed a 26% decrease in symptomatic days and a 50%+ reduction in abdominal pain. It's currently in Phase III trials. Larazotide is not a substitute for a gluten-free diet - it's an adjunctive therapy that helps protect against inadvertent gluten exposure and residual permeability.
Are gut-healing peptides safe to use with IBD medications?
In general, gut-healing peptides appear to be compatible with standard IBD medications based on their mechanisms of action and available evidence, though formal drug interaction studies are limited. BPC-157 works through NO modulation and growth factor pathways that don't directly conflict with biologics (like infliximab or vedolizumab), immunomodulators (azathioprine, methotrexate), or 5-ASA compounds. KPV's NF-kB inhibition is complementary to, not competitive with, TNF inhibitors. Larazotide acts locally with minimal systemic absorption. However, theoretical concerns exist: KPV might enhance immunosuppression when combined with strong immunosuppressive agents, and BPC-157's angiogenic properties warrant monitoring in patients on certain biologics. Always disclose peptide use to your gastroenterologist and work with both your GI specialist and peptide prescriber as a team.
How does VIP help with gut motility and digestive function?
VIP (Vasoactive Intestinal Peptide) acts as a master regulator of gut function through two receptor types (VPAC1 and VPAC2) distributed throughout the GI tract. For motility, VIP co-localizes with and modulates cholinergic neurons in the enteric nervous system - the "brain of the gut" that controls peristalsis and the migrating motor complex. VIP also regulates intestinal secretion (water and electrolyte flux), stimulates pancreatic enzyme and bicarbonate release, increases mesenteric blood flow, and modulates mucosal immune responses. For patients with SIBO, slow transit, or gastroparesis, VIP's ability to normalize enteric nervous system function can help restore the motility patterns that prevent bacterial overgrowth. It's typically administered as a nasal spray (50-100 mcg, 1-2 times daily) because oral VIP is rapidly degraded.
Can I use gut-healing peptides during pregnancy?
No. There is no safety data for BPC-157, KPV, larazotide, VIP, or LL-37 in pregnancy or lactation. Until human reproductive safety studies are conducted, all gut-healing peptides should be avoided during pregnancy, while trying to conceive, and while breastfeeding. If you're dealing with gut issues during pregnancy, work with your obstetrician and gastroenterologist on pregnancy-safe interventions. Dietary modifications, probiotics with established pregnancy safety profiles (like Lactobacillus rhamnosus GG), and some nutritional supplements (glutamine, zinc, omega-3) can support gut health during pregnancy without the unknowns associated with research peptides.
What's the best diet to combine with gut-healing peptides?
The optimal diet during peptide-based gut healing depends on your specific condition, but a reasonable general approach involves three phases. During active treatment (Phases 1-3), an anti-inflammatory elimination diet - such as the Autoimmune Protocol (AIP) or a modified paleo diet - removes the dietary triggers that undermine peptide-driven repair. This means eliminating gluten, dairy, refined sugar, alcohol, processed foods, and individual triggers identified through testing or elimination. During the restore phase (Phase 4), gradually reintroduce eliminated foods one at a time every 3-5 days while monitoring for reactions. For long-term maintenance, transition to a Mediterranean-style diet rich in omega-3 fats, polyphenol-rich fruits and vegetables, fermented foods, and adequate fiber. Bone broth, glutamine-rich proteins, and zinc-containing foods support ongoing mucosal health. Throughout all phases, avoid NSAIDs, minimize alcohol, and practice meal spacing (4-5 hours between meals) to support MMC function.
How do I know when to stop using gut-healing peptides?
Consider tapering and discontinuing peptides when three criteria are met: (1) Symptoms have resolved or reached an acceptable stable baseline for at least 4-6 weeks, (2) Objective markers have normalized - including intestinal permeability testing, calprotectin (if previously elevated), and SIBO breath testing (if previously positive), and (3) You've successfully reintroduced previously problematic foods without significant reactions. Tapering typically involves reducing the dose by 50% for 2-4 weeks, then moving to every-other-day dosing for 2-4 weeks, then discontinuing. Some patients with chronic conditions (IBD, recurrent SIBO) may benefit from low-dose maintenance therapy long-term. If symptoms recur after discontinuation, a repeat course can be initiated. The gut's rapid epithelial turnover means that you'll typically know within 2-4 weeks of stopping whether the improvements are durable.
Is there evidence that combining multiple gut peptides works better than using just one?
Direct head-to-head comparison studies of peptide combinations vs. monotherapy for gut conditions haven't been published. However, the rationale for combination therapy is strong based on the complementary mechanisms of each peptide. Gut barrier dysfunction involves multiple simultaneous failures - tight junction breakdown (larazotide's target), mucosal damage (BPC-157's target), inflammation (KPV's target), motility dysfunction (VIP's target), and microbial imbalance (LL-37's target). Addressing only one of these while the others persist limits the overall healing response. Clinical experience from practitioners who use combination protocols generally reports better outcomes than single-peptide approaches, particularly for complex conditions. That said, not everyone needs every peptide. A thoughtful assessment of which mechanisms are most relevant to your specific condition should guide selection.
Do gut-healing peptides help with food sensitivities?
Food sensitivities often stem from increased intestinal permeability - when tight junctions open excessively, incompletely digested food proteins cross the barrier and trigger immune responses. By repairing the gut barrier, peptides can reduce the antigenic load that drives food sensitivities. Larazotide directly stabilizes tight junctions. BPC-157 heals the mucosal surface and reduces inflammation-driven permeability. KPV reduces the inflammatory response that both causes and results from barrier dysfunction. Most patients report gradual improvement in food tolerance over 4-12 weeks of peptide therapy combined with an elimination diet. However, true food allergies (IgE-mediated) are immune-mediated responses that won't be resolved by barrier repair alone. The food sensitivities that improve with gut healing are typically IgG-mediated or non-immune reactions related to permeability rather than true allergies.
References
- Sikiric P, et al. Stable gastric pentadecapeptide BPC 157: novel therapy in gastrointestinal tract. Curr Pharm Des. 2011;17(16):1612-1632. doi: 10.2174/138161211796197115
- Seiwerth S, et al. BPC 157 and Blood Vessels. Curr Pharm Des. 2014;20(7):1121-1135. doi: 10.2174/13816128113199990421
- Sikiric P, et al. Stable Gastric Pentadecapeptide BPC 157, Robert's Stomach Cytoprotection/Adaptive Cytoprotection/Organoprotection, and Selye's Stress Coping Response: Progress, Achievements, and the Future. Gut Liver. 2020;14(2):153-167. doi: 10.5009/gnl18490
- Sikiric P, et al. Stable gastric pentadecapeptide BPC 157 and wound healing. Front Pharmacol. 2021;12:627533. doi: 10.3389/fphar.2021.627533
- Sikiric P, et al. Fistulas Healing. Stable Gastric Pentadecapeptide BPC 157 Therapy. Curr Pharm Des. 2020;26(25):2991-3000. doi: 10.2174/1381612826666200424180139
- Sever M, et al. Stable gastric pentadecapeptide BPC 157 in trials for inflammatory bowel disease (PL-10, PLD-116, PL14736, Pliva, Croatia) heals ileoileal anastomosis in the rat. Surg Today. 2009;39(1):57-63. doi: 10.1007/s00595-008-3795-y
- Klicek R, et al. Stable gastric pentadecapeptide BPC 157 heals cysteamine-colitis and colon-colon-anastomosis and counteracts cuprizone brain demyelination and behavioral changes. J Physiol Pharmacol. 2013;64(5):597-612. PMID: 24304573
- Sikiric P, et al. BPC 157 therapy to detriment sphincters failure - esophagitis - Loss of antrum-Loss of fundus in rat. Regul Pept. 2012;178(1-3):33-46. doi: 10.1016/j.regpep.2012.06.009
- Cesarec V, et al. Pentadecapeptide BPC 157 and the esophagocutaneous fistula healing therapy. Eur J Pharmacol. 2013;701(1-3):203-212. doi: 10.1016/j.ejphar.2012.11.055
- Duzel A, et al. Stable gastric pentadecapeptide BPC 157 in the treatment of colitis and ischemia and reperfusion in rats: New insights. World J Gastroenterol. 2017;23(48):8465-8488. doi: 10.3748/wjg.v23.i48.8465
- Sikiric P, et al. An experimental model of prolonged esophagitis with sphincter failure in the rat and the therapeutic potential of gastric pentadecapeptide BPC 157. J Pharmacol Sci. 2020;143(2):56-69. doi: 10.1016/j.jphs.2020.02.001
- Dalmasso G, et al. PepT1-mediated tripeptide KPV uptake reduces intestinal inflammation. Gastroenterology. 2008;134(1):166-178. doi: 10.1053/j.gastro.2007.10.026
- Viennois E, et al. Critical role of PepT1 in promoting colitis-associated cancer and therapeutic benefits of the anti-inflammatory PepT1-mediated tripeptide KPV in a murine model. Cell Mol Gastroenterol Hepatol. 2016;2(3):340-357. doi: 10.1016/j.jcmgh.2016.01.014
- Xiao B, et al. Orally targeted delivery of tripeptide KPV via hyaluronic acid-functionalized nanoparticles efficiently alleviates ulcerative colitis. Mol Ther. 2017;25(7):1628-1640. doi: 10.1016/j.ymthe.2017.02.015
- Chen Y, et al. PepT1-targeted nanodrug based on co-assembly of anti-inflammatory peptide and immunosuppressant for combined treatment of acute and chronic DSS-induced colitis. Front Pharmacol. 2024;15:1442876. doi: 10.3389/fphar.2024.1442876
- Gopalakrishnan S, et al. Larazotide acetate: a pharmacological peptide approach to tight junction regulation. Am J Physiol Gastrointest Liver Physiol. 2021;320(6):G983-G989. doi: 10.1152/ajpgi.00386.2020
- Kelly CP, et al. Larazotide acetate in patients with coeliac disease undergoing a gluten challenge: a randomised placebo-controlled study. Aliment Pharmacol Ther. 2013;37(2):252-262. doi: 10.1111/apt.12147
- Leffler DA, et al. Larazotide acetate for persistent symptoms of celiac disease despite a gluten-free diet: a randomized controlled trial. Gastroenterology. 2015;148(7):1311-1319. doi: 10.1053/j.gastro.2015.02.008
- Tajik N, et al. Targeting zonulin and intestinal epithelial barrier function to prevent onset of arthritis. Nat Commun. 2020;11(1):1995. doi: 10.1038/s41467-020-15831-7
- Gitter AH, et al. Larazotide acetate protects the intestinal mucosal barrier from anoxia/reoxygenation injury via various cellular mechanisms. Biomedicines. 2025;13(10):2483. doi: 10.3390/biomedicines13102483
- Fasano A. Zonulin, regulation of tight junctions, and autoimmune diseases. Ann N Y Acad Sci. 2012;1258(1):25-33. doi: 10.1111/j.1749-6632.2012.06538.x
- Leliefeld PHC, et al. The potential utility of tight junction regulation in celiac disease: focus on larazotide acetate. Therap Adv Gastroenterol. 2015;8(1):37-49. doi: 10.1177/1756283X14551570
- Ganesh BP, et al. Recent advances in vasoactive intestinal peptide physiology and pathophysiology: focus on the gastrointestinal system. F1000Research. 2019;8:1629. doi: 10.12688/f1000research.20432.1
- Rao M, et al. Vasoactive intestinal polypeptide plays a key role in the microbial-neuroimmune control of intestinal motility. Cell Mol Gastroenterol Hepatol. 2023;16(4):545-564. doi: 10.1016/j.jcmgh.2023.06.012
- Wu WKK, et al. Vasoactive intestinal polypeptide promotes intestinal barrier homeostasis and protection against colitis in mice. PLoS ONE. 2015;10(5):e0125549. doi: 10.1371/journal.pone.0125549
- Vandamme D, et al. A comprehensive summary of LL-37, the factotum human cathelicidin peptide. Cell Immunol. 2012;280(1):22-35. doi: 10.1016/j.cellimm.2012.11.009
- Fabisiak A, et al. LL-37: cathelicidin-related antimicrobial peptide with pleiotropic activity. Pharmacol Rep. 2016;68(4):802-808. doi: 10.1016/j.pharep.2016.03.015
- Mookherjee N, et al. Antimicrobial host defence peptides: functions and clinical potential. Nat Rev Drug Discov. 2020;19(5):311-332. doi: 10.1038/s41573-019-0058-8
- Tai EKK, et al. A new role for cathelicidin in ulcerative colitis in mice. Exp Biol Med. 2007;232(6):799-808. doi: 10.3181/00379727-232-2320799
- Ma X, et al. A novel cecropin-LL37 hybrid peptide protects mice against EHEC infection-mediated changes in gut microbiota, intestinal inflammation, and impairment of mucosal barrier functions. Front Immunol. 2020;11:1361. doi: 10.3389/fimmu.2020.01361
- Kai-Larsen Y, Agerberth B. The role of the multifunctional peptide LL-37 in host defense. Front Biosci. 2008;13:3760-3767. doi: 10.2741/2963
- Odenwald MA, Turner JR. The intestinal epithelial barrier: a therapeutic target? Nat Rev Gastroenterol Hepatol. 2017;14(1):9-21. doi: 10.1038/nrgastro.2016.169
- Turner JR. Intestinal mucosal barrier function in health and disease. Nat Rev Immunol. 2009;9(11):799-809. doi: 10.1038/nri2653
- Chelakkot C, et al. Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp Mol Med. 2018;50(8):1-9. doi: 10.1038/s12276-018-0126-x
- Shen L, et al. Tight junctions on the move: molecular mechanisms for epithelial barrier regulation. Ann N Y Acad Sci. 2012;1258(1):9-18. doi: 10.1111/j.1749-6632.2012.06543.x
- Otani T, Bhatt A. Paracellular permeability and tight junction regulation in gut health and disease. Nat Rev Gastroenterol Hepatol. 2023;20:417-432. doi: 10.1038/s41575-023-00766-3
- Konig J, et al. Human intestinal barrier function in health and disease. Clin Transl Gastroenterol. 2016;7(10):e196. doi: 10.1038/ctg.2016.54
- Bischoff SC, et al. Intestinal permeability - a new target for disease prevention and therapy. BMC Gastroenterol. 2014;14:189. doi: 10.1186/s12876-014-0189-7
- Mu Q, et al. Leaky gut as a danger signal for autoimmune diseases. Front Immunol. 2017;8:598. doi: 10.3389/fimmu.2017.00598
- Kelly JR, et al. Breaking down the barriers: the gut microbiome, intestinal permeability and stress-related psychiatric disorders. Front Cell Neurosci. 2015;9:392. doi: 10.3389/fncel.2015.00392