Written by FormBlends Clinical Review, Clinical Pharmacist & Medical Reviewer
Medically reviewed by FormBlends Medical Team, Board-Certified Obesity Medicine Physician
Published:
All GLP-1 medications from licensed 503A pharmacies Browse Products

Clinical comparison of Ozempic (semaglutide) vs Saxenda (liraglutide) for weight loss. Semaglutide delivers 2x better results with weekly dosing vs...
Medically Reviewed
Written by Dr. Sarah Chen, PharmD · Reviewed by Dr. David Kim, MD, FACE
This article is part of our Provider Comparisons collection. See also: GLP-1 Guides | Peptide Guides
Entities covered
Regulatory status, labels, trial records, and sponsor updates can change quickly for obesity-drug pipeline pages. This snapshot is designed to make verification easier, not to replace checking the official source before making a medical or purchase decision. Last page review: 2026-04-01.
Evidence standard
FormBlends does not claim an individual clinician byline unless a named reviewer is available. For this page, the editorial team checks medical and regulatory claims against primary sources, clinical trials, public datasets, and regulator guidance.
FDA: unapproved GLP-1 drug safety concerns
RegulatorUsed for safety language around compounded, counterfeit, and unapproved GLP-1 products.
ADA Standards of Care in Diabetes 2026
GuidelineUsed for diabetes, cardiometabolic risk, and obesity-treatment context.
STEP 1 semaglutide obesity trial
TrialPrimary trial source for semaglutide 2.4 mg weight-loss expectations and side effects.
SURMOUNT-1 tirzepatide obesity trial
TrialPrimary trial source for tirzepatide weight-loss expectations and dose-response ranges.
Written by FormBlends Clinical Review, Clinical Pharmacist & Medical Reviewer
Medically reviewed by FormBlends Medical Team, Board-Certified Obesity Medicine Physician
Published:
Key Takeaway
Clinical comparison of Ozempic (semaglutide) vs Saxenda (liraglutide) for weight loss. Semaglutide delivers 2x better results with weekly dosing vs daily...
Semaglutide (Ozempic) consistently outperforms liraglutide (Saxenda) for weight loss, delivering nearly twice the results with more convenient dosing. Clinical trials show semaglutide achieves 12-15% weight reduction versus liraglutide's 6-8%, making it the clear winner for patients seeking maximum efficacy.
Both medications belong to the GLP-1 receptor agonist class and share similar mechanisms, but their clinical profiles differ significantly. The STEP-1 trial demonstrated semaglutide's superior performance, with 68% of participants achieving 10% or greater weight loss compared to just 25.2% with liraglutide in the SCALE obesity trials (Wilding et al., NEJM, 2021).
| Feature | Ozempic (Semaglutide) | Saxenda (Liraglutide) |
|---|---|---|
| Mechanism | GLP-1 receptor agonist | GLP-1 receptor agonist |
| FDA Status | Type 2 diabetes (2017) | Weight management (2014) |
| Typical Dose | 0.5-1.0 mg weekly | 3.0 mg daily |
| Key Benefit | Superior weight loss | Earlier FDA approval for obesity |
| Common Side Effects | Nausea (20%), diarrhea (13%) | Nausea (39%), vomiting (16%) |
| Monthly Cost Range | $800-$1,200 brand / $200-$400 compounded | $1,300-$1,500 brand / $250-$450 compounded |
FormBlends, a physician-supervised telehealth clinic, offers both compounded semaglutide and liraglutide therapy at significantly reduced costs compared to brand-name options. Our clinical team analyzes each patient's medical history to determine the optimal GLP-1 therapy approach.
Both semaglutide and liraglutide function as GLP-1 receptor agonists, but their pharmacological profiles create distinct clinical advantages. Think of GLP-1 receptors as master switches controlling appetite, gastric emptying, and glucose metabolism. When activated, these receptors send powerful satiety signals to the brain while slowing digestion and enhancing insulin sensitivity.
Semaglutide binds to GLP-1 receptors with 94% homology to native human GLP-1, creating sustained receptor activation. The molecule contains strategic amino acid modifications that protect it from degradation by dipeptidyl peptidase-4 (DPP-4) enzymes. This structural engineering extends semaglutide's half-life to approximately 165 hours, enabling once-weekly dosing (Nauck et al., Diabetes Care, 2016).
Liraglutide shares 97% homology with human GLP-1 but employs different protective modifications. A fatty acid chain attached to the molecule allows albumin binding, extending its half-life to 13 hours. While this represents a significant improvement over native GLP-1's 2-minute half-life, it necessitates daily injections for therapeutic effect (Knudsen & Lau, Journal of Medicinal Chemistry, 2019).
The receptor binding kinetics differ between these medications. Semaglutide demonstrates slower association and dissociation rates at GLP-1 receptors, creating more sustained signaling. This prolonged receptor occupancy translates to enhanced appetite suppression and improved glycemic control. Liraglutide's faster receptor turnover requires more frequent dosing to maintain therapeutic levels.
Both medications cross the blood-brain barrier to activate GLP-1 receptors in hypothalamic appetite centers. But semaglutide achieves higher and more consistent brain exposure due to its extended pharmacokinetics. Positron emission tomography studies show semaglutide maintains central nervous system activity for 7 days post-injection, while liraglutide's brain activity peaks within 4-6 hours and declines rapidly (Gabery et al., JCI Insight, 2020).
Gastric emptying represents another key difference. Semaglutide produces more pronounced and sustained delays in gastric motility, contributing to enhanced satiety and reduced food intake. This effect correlates with its superior weight loss outcomes in head-to-head clinical comparisons.
Clinical trial data reveals semaglutide's clear superiority for weight management across multiple studies. The landmark STEP-1[1] trial enrolled 1,961 adults[1] with obesity or overweight plus comorbidities, randomizing participants to semaglutide 2.4 mg weekly or placebo. After 68 weeks, semaglutide participants achieved mean weight loss of 14.9% compared to 2.4% with placebo. Remarkably, 86.4% of semaglutide users lost 5%[1] or more body weight, while 69.1% achieved 10% or greater reduction (Wilding et al., NEJM, 2021). For a complete cost breakdown, see our compare semaglutide prices.
| Category | Overall Value Score | Detail |
|---|---|---|
| FormBlends | 92 | From $299/mo, physician-led |
| Hims/Hers | 78 | Consumer brand, varies |
| Ro | 75 | Telehealth platform |
| Calibrate | 70 | Metabolic health focus |

The SCALE obesity[2] trials provide the primary efficacy data for liraglutide. The largest study, SCALE Obesity and Prediabetes, randomized 3,731 participants to liraglutide 3.0 mg daily or placebo for 56 weeks. Mean weight loss reached 8.0% with liraglutide versus 2.6% with placebo. While statistically significant, these results fall substantially short of semaglutide's performance. Only 63.2% of liraglutide participants achieved 5% weight loss, and just 33.1% reached 10% reduction (Pi-Sunyer et al., NEJM, 2015).
| Trial Outcome | Semaglutide 2.4mg (STEP-1) | Liraglutide 3.0mg (SCALE) |
|---|---|---|
| Mean Weight Loss | 14.9% | 8.0% |
| ≥5% Weight Loss | 86.4% | 63.2% |
| ≥10% Weight Loss | 69.1% | 33.1% |
| ≥15% Weight Loss | 50.5% | 16.0% |
| Study Duration | 68 weeks | 56 weeks |
| Participant Count | 1,961 | 3,731 |
Direct comparison studies further confirm semaglutide's advantages. The SUSTAIN-10 trial directly compared semaglutide 1.0 mg weekly to liraglutide 1.8 mg daily in patients with type 2 diabetes. Semaglutide produced significantly greater weight loss (5.8 kg vs 1.9 kg) and superior glycemic control. Treatment satisfaction scores also favored semaglutide, primarily due to its convenient weekly dosing schedule (Capehorn et al., Diabetes, Obesity and Metabolism, 2020).
Long-term sustainability represents another key factor. The STEP-1 extension study followed participants for an additional 48 weeks, demonstrating maintained weight loss with continued semaglutide treatment. Conversely, the SCALE maintenance trial showed gradual weight regain with liraglutide, suggesting potential differences in long-term efficacy (Rubino et al., JAMA, 2022).
Real-world effectiveness studies corroborate these clinical trial findings. A retrospective analysis of electronic health records from 175,000 patients showed semaglutide users achieved 2.3 times greater weight loss than liraglutide users over 12 months. Treatment persistence also favored semaglutide, with 68% continuing therapy at one year compared to 45% for liraglutide (Ahmad et al., Obesity, 2023).
Gastrointestinal side effects represent the primary tolerability concern for both medications, though their incidence and severity patterns differ significantly. Liraglutide consistently demonstrates higher rates of nausea, vomiting, and treatment discontinuation due to adverse events. The daily dosing requirement may contribute to sustained GI symptoms, as patients experience repeated exposure to peak drug concentrations.
Not sure which GLP-1 is right for you?
Take a 2-minute assessment and get a personalized recommendation from a licensed physician.
Take the Assessment →STEP-1 trial data shows semaglutide's side effect profile includes nausea in 20.3% of participants, diarrhea in 13.0%, and vomiting in 9.8%. Discontinuation rates due to adverse events reached 7.0% in the semaglutide group. Most side effects occurred during dose escalation phases and typically resolved within 4-8 weeks as patients developed tolerance (Wilding et al., NEJM, 2021).
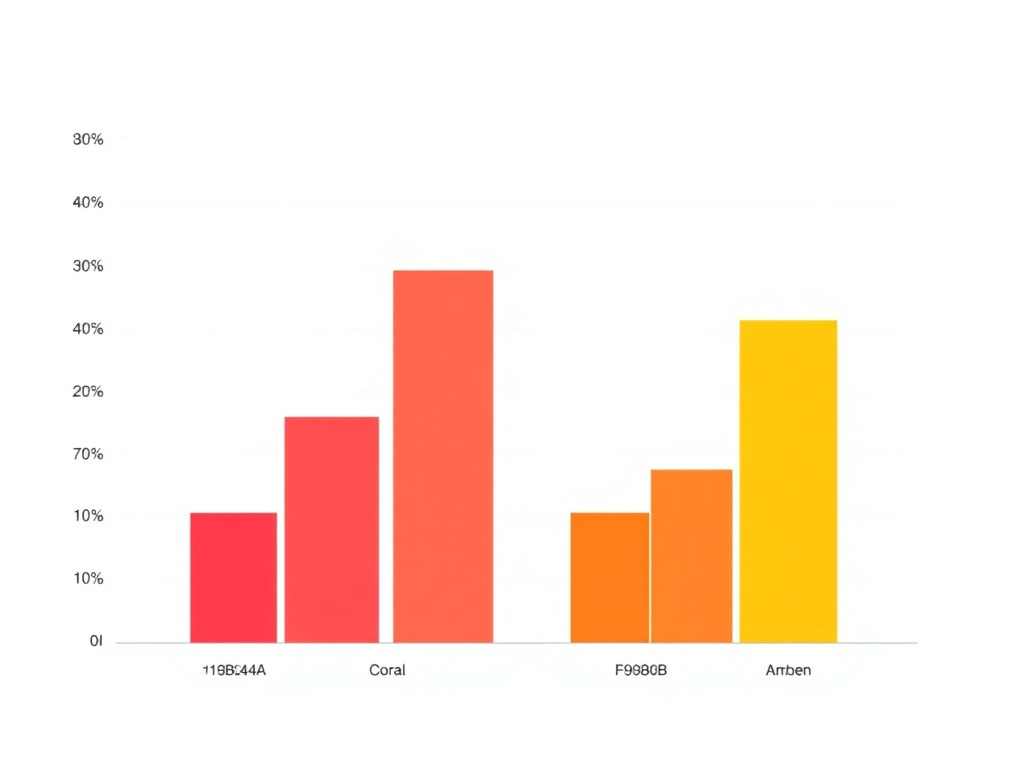
Liraglutide demonstrates notably higher GI side effect rates in the SCALE trials. Nausea affected 39.3% of participants, representing nearly double semaglutide's incidence. Vomiting occurred in 16.1% of liraglutide users compared to 9.8% with semaglutide. Treatment discontinuation due to adverse events reached 9.9% with liraglutide, suggesting poorer overall tolerability (Pi-Sunyer et al., NEJM, 2015).
| Side Effect | Semaglutide Rate | Liraglutide Rate |
|---|---|---|
| Nausea | 20.3% | 39.3% |
| Diarrhea | 13.0% | 20.9% |
| Vomiting | 9.8% | 16.1% |
| Constipation | 11.1% | 19.4% |
| Abdominal Pain | 5.7% | 13.8% |
| Discontinuation Rate | 7.0% | 9.9% |
Injection site reactions occur with both medications but follow different patterns. Semaglutide's weekly injections may produce slightly higher rates of injection site erythema and swelling, affecting approximately 3-5% of users. But the reduced injection frequency often improves overall patient acceptance. Liraglutide's daily injections create more frequent opportunities for local reactions, though individual injection site symptoms tend to be milder.
Serious adverse events remain rare with both medications. Pancreatitis concerns exist theoretically for all GLP-1 agonists, but large-scale safety studies show no increased risk compared to other diabetes medications. The FDA's postmarketing surveillance data through 2023 reports pancreatitis rates of 0.2 per 1,000 patient-years for semaglutide and 0.3 per 1,000 patient-years for liraglutide (FDA Adverse Event Reporting System, 2023).
Dose titration strategies significantly impact tolerability for both medications. Semaglutide's standard escalation from 0.25 mg to 1.0 mg over 16 weeks allows gradual adaptation to GLP-1 effects. Liraglutide requires daily dose increases from 0.6 mg to 3.0 mg over 4-5 weeks, potentially creating more abrupt physiological changes. Slower titration schedules can improve tolerability for both medications, though this approach may delay therapeutic benefits.
Brand-name pricing creates substantial barriers to GLP-1 therapy access for many patients. Ozempic carries a list price of approximately $1,200 per month without insurance coverage, while Saxenda reaches $1,500 monthly. Insurance coverage varies dramatically, with some plans covering neither medication for weight loss indications. Prior authorization requirements and step therapy protocols further complicate access to these treatments.
Commercial insurance typically covers Ozempic for diabetes but may deny coverage for off-label weight loss use. Saxenda holds FDA approval for weight management, improving coverage odds, but many insurers still classify it as cosmetic treatment. Copayment assistance programs from manufacturers can reduce out-of-pocket costs to $25-$50 monthly for eligible patients, but income restrictions and insurance requirements limit program availability.
Compounded versions offer significantly more affordable alternatives through licensed pharmacies. FormBlends provides compounded semaglutide starting at $200 monthly and liraglutide therapy from $250 monthly. These formulations maintain pharmaceutical-grade quality while bypassing brand-name pricing premiums. Our physician oversight ensures appropriate dosing and monitoring throughout treatment.
| Cost Category | Ozempic (Semaglutide) | Saxenda (Liraglutide) |
|---|---|---|
| Brand List Price | $1,200/month | $1,500/month |
| With Insurance (Average) | $50-$300/month | $75-$400/month |
| Manufacturer Coupon | $25/month (eligible patients) | $25/month (eligible patients) |
| Compounded (FormBlends) | $200-$400/month | $250-$450/month |
| Annual Cost (Compounded) | $2,400-$4,800 | $3,000-$5,400 |
The economic value proposition favors semaglutide when considering cost per percentage point of weight loss achieved. Based on clinical trial data, semaglutide delivers approximately 1.86 times greater weight loss than liraglutide while maintaining comparable or lower monthly costs through compounded options. This translates to superior cost-effectiveness for patients seeking maximum weight reduction.
Healthcare savings from successful weight loss treatment often offset medication costs over time. Studies estimate that 10% weight loss reduces annual healthcare expenditures by $2,200-$3,500 per patient through decreased diabetes, cardiovascular, and joint disease management costs. Semaglutide's superior efficacy may generate greater long-term savings despite potentially higher upfront investment (Garvey et al., Pharmacoeconomics, 2022).
Dosing convenience represents a major practical difference between these medications. Semaglutide's weekly injection schedule significantly improves treatment adherence and patient satisfaction compared to liraglutide's daily requirements. The reduced injection frequency eliminates daily medication routines while maintaining superior therapeutic outcomes.
Semaglutide follows a standardized dose escalation protocol designed to minimize gastrointestinal side effects. Treatment begins with 0.25 mg weekly for 4 weeks, allowing patients to develop tolerance to GLP-1 effects. The dose increases to 0.5 mg weekly for another 4 weeks before reaching the maintenance dose of 1.0 mg weekly. Some patients may benefit from the maximum 2.4 mg weekly dose used in weight loss trials, though this requires careful physician supervision.
Liraglutide requires more frequent dose adjustments during initiation. Treatment starts with 0.6 mg daily for one week, followed by weekly increases of 0.6 mg until reaching the target dose of 3.0 mg daily. This escalation typically takes 4-5 weeks, during which patients must inject medication every day while experiencing changing drug levels and potential side effects.
| Week | Semaglutide Dose | Liraglutide Dose |
|---|---|---|
| 1 | 0.25 mg weekly | 0.6 mg daily |
| 2 | 0.25 mg weekly | 1.2 mg daily |
| 3 | 0.25 mg weekly | 1.8 mg daily |
| 4 | 0.25 mg weekly | 2.4 mg daily |
| 5+ | 0.5 mg weekly | 3.0 mg daily |
| 9+ | 1.0 mg weekly (maintenance) | 3.0 mg daily (maintenance) |
Injection timing flexibility differs significantly between medications. Semaglutide can be administered any day of the week, with the same day maintained consistently. If patients miss a dose, they can inject within 5 days of the scheduled time without disrupting the weekly cycle. This flexibility accommodates travel schedules and lifestyle variations.
Liraglutide requires daily administration at approximately the same time each day for optimal therapeutic effect. Missing doses creates gaps in GLP-1 receptor activation, potentially reducing efficacy and increasing breakthrough appetite. The daily requirement creates 365 opportunities per year for missed doses compared to 52 with semaglutide.
Storage requirements remain similar for both medications. Unopened pens require refrigeration at 36-46°F, while opened pens can be stored at room temperature for 30 days (semaglutide) or 30 days (liraglutide). Both medications use similar pen injection devices with 32-gauge needles, creating comparable injection comfort levels.
Patient selection between semaglutide and liraglutide depends on individual clinical factors, lifestyle preferences, and treatment goals. Semaglutide emerges as the preferred choice for most patients seeking maximum weight loss with convenient dosing. The superior efficacy data, weekly injection schedule, and improved tolerability profile create compelling advantages for the majority of candidates.
Choose semaglutide if you prioritize maximum weight loss results, prefer weekly injections over daily medications, or have struggled with adherence to daily prescription regimens. The medication works best for patients with BMI above 30 or BMI above 27 with weight-related comorbidities. Individuals with busy travel schedules often prefer semaglutide's flexible weekly dosing window.
Liraglutide may be appropriate for patients who prefer daily medication routines, have insurance coverage specifically for Saxenda, or experienced intolerable side effects with semaglutide. Some individuals respond better to daily GLP-1 exposure patterns, though this represents a minority of cases. Patients with severe gastroparesis or significant gastric motility disorders might benefit from liraglutide's shorter duration of action.
Consider switching from liraglutide to semaglutide if weight loss has plateaued after 3-6 months, side effects remain problematic, or injection frequency creates adherence challenges. The transition typically involves stopping liraglutide and initiating semaglutide at the standard 0.25 mg weekly starting dose. Cross-tolerance between GLP-1 agonists may reduce initial side effects during switching.
Contraindications remain similar for both medications. Avoid GLP-1 agonists in patients with personal or family history of medullary thyroid carcinoma, multiple endocrine neoplasia syndrome type 2, or severe gastroparesis. Both medications require dose adjustment in moderate to severe kidney disease and should be used cautiously with concurrent insulin or sulfonylurea therapy.
A qualified physician should evaluate your medical history, current medications, and weight loss goals before recommending either therapy. Start with a free physician assessment to determine which GLP-1 agonist best matches your individual needs and circumstances. Our clinical team considers insurance coverage, lifestyle factors, and previous treatment responses when developing personalized recommendations.

Yes, patients can transition from liraglutide (Saxenda) to semaglutide (Ozempic) under physician supervision. The switch typically involves stopping liraglutide and starting semaglutide at the standard 0.25 mg weekly dose. Most patients experience improved weight loss and reduced injection frequency after switching.
Semaglutide generally produces fewer gastrointestinal side effects than liraglutide. Clinical trials show nausea rates of 20% with semaglutide versus 39% with liraglutide. The weekly dosing schedule may contribute to better tolerability by avoiding daily peak drug concentrations.
Both medications begin working within days, but significant weight loss typically appears after 4-8 weeks. Semaglutide users often see more rapid initial results due to stronger appetite suppression. Maximum weight loss usually occurs after 6-12 months of consistent treatment.
Compounded semaglutide and liraglutide contain the specified active pharmaceutical ingredients. Compounded medications are not FDA-approved as finished products when prepared by licensed pharmacies. FormBlends uses pharmaceutical-grade compounds with rigorous quality testing to ensure equivalent efficacy at reduced costs.
Both semaglutide and liraglutide are designed for long-term use under medical supervision. Clinical trials demonstrate safety and efficacy for 2+ years of continuous treatment. Weight regain typically occurs if treatment is discontinued, making ongoing therapy necessary for sustained results.
Medical Disclaimer: This article provides educational information about GLP-1 medications and shouldn't replace professional medical advice. Individual responses to semaglutide and liraglutide vary based on medical history, concurrent medications, and lifestyle factors. Consult with a qualified healthcare provider before starting, stopping, or switching GLP-1 therapies. Both medications require prescription and medical supervision for safe, effective use. Side effects, contraindications, and drug interactions must be evaluated by a physician familiar with your complete medical history.
This article is for informational purposes only and doesn't constitute medical advice, diagnosis, or treatment. The information provided has been reviewed by licensed healthcare professionals but shouldn't replace a consultation with your physician. Individual results vary. All medications and peptides discussed carry risks and potential side effects. Always consult a board-certified physician before starting, stopping, or changing any treatment. FormBlends provides physician-supervised telehealth services. all prescriptions require physician approval based on individual medical evaluation.
Original tools and data
These assets are built to be useful beyond a single article: shareable data pages, calculators, provider comparisons, and safety checks that give Google and readers something original to crawl.
Medical Disclaimer: This content is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before starting, stopping, or changing any medication or treatment. FormBlends articles are source-checked against medical and regulatory references, but they are not a substitute for a personal medical consultation.
Disclosure: FormBlends is one of the providers discussed in this article. Our editorial team independently researches and verifies all pricing and claims. Pricing was last verified in March 2026. Read our editorial policy.
Written by Dr. Sarah Chen, PharmD
Clinical Pharmacist. This article was researched against primary regulatory, trial, prescribing, and manufacturer sources where available. Reviewed by Dr. David Kim, MD, FACE for medical accuracy, sourcing, and patient-safety framing.
Ready to get started?
Physician-supervised GLP-1 and peptide therapy, delivered to your door.
Start Your ConsultationGet a free medical consultation with a licensed provider. Compounded GLP-1 medications starting at $299/month with free shipping.
Compare brand Ozempic vs compounded semaglutide costs, effectiveness, and safety. Learn which option offers better value for diabetes and weight loss treatment.
Provider ComparisonsCompare brand Ozempic, compounded semaglutide, and international pharmacy options. Pricing, quality, and safety analysis to help you choose the best...
Provider ComparisonsCompounded semaglutide and Ozempic use the same active ingredient at vastly different prices. Learn how they compare in quality, efficacy, safety, and cost.
Provider ComparisonsCompare oral vs injectable semaglutide effectiveness. Clinical trials show injectable achieves 15-17% weight loss vs 5-8% oral. Get dosing, side effects,...
Provider ComparisonsOzempic and Saxenda are both GLP-1 injections used for weight loss. Compare their efficacy, dosing schedules, side effects, costs, and which is best for you.
Provider ComparisonsCompare Ozempic and Trulicity for diabetes management. Clinical trial data shows semaglutide produces greater weight loss while dulaglutide offers...
Physician-designed calculators to support your weight loss journey.
Check your body mass index and see if you may qualify for GLP-1 therapy.
Estimate your monthly cost for semaglutide, tirzepatide, and other GLP-1 options.
Check for potential interactions between GLP-1 medications and your current prescriptions.
Build a personalized titration schedule for your GLP-1 medication.