About This Research Library
You're looking at the master index for 100 peer-reviewed research reports covering every major peptide compound, GLP-1 receptor agonist, and clinical application in modern peptide science. This is the single largest open-access peptide research library available anywhere.
Key Takeaways
- 20 GLP-1 & incretin reports - every approved and pipeline drug, plus condition-specific deep analyses
- 15 growth hormone peptide reports - secretagogues, GHRH analogs, IGF-1 variants, and fat-loss fragments
- 12 longevity & mitochondrial peptide reports - telomere science, senolytic peptides, NAD+ strategies
- 4 immune & healing peptide reports - thymosin peptides, BPC-157, antimicrobial peptides
- 24 specialty compound reports - nootropics, cosmetic peptides, reproductive peptides, neuropeptides
Peptide therapeutics have moved from the fringes of biomedical research into the center of modern medicine. The global peptide therapeutics market reached approximately $141 billion in 2025 and is projected to exceed $164 billion by the end of 2026, driven largely by GLP-1 receptor agonists for metabolic disease (DOI: 10.1016/j.drudis.2024.103891). Over 110 peptide drugs have received regulatory approval worldwide, and the pipeline continues to expand with dual- and triple-agonist compounds, oral peptide formulations, and novel delivery technologies.
This library was built to solve a real problem: reliable peptide information is scattered across thousands of journal articles, clinical trial databases, and manufacturer publications. We've consolidated the best available evidence into 100 structured reports - each one thoroughly cited, regularly updated, and written for both clinicians and educated patients.
What This Library Covers
- 20 GLP-1 & incretin reports - every approved and pipeline drug, plus condition-specific deep analyses
- 15 growth hormone peptide reports - secretagogues, GHRH analogs, IGF-1 variants, and fat-loss fragments
- 12 longevity & mitochondrial peptide reports - telomere science, senolytic peptides, NAD+ strategies
- 4 immune & healing peptide reports - thymosin peptides, BPC-157, antimicrobial peptides
- 24 specialty compound reports - nootropics, cosmetic peptides, reproductive peptides, neuropeptides
- 25 practical and reference guides - reconstitution, stacking, blood work, legality, provider selection
Every report follows a consistent structure: mechanism of action, clinical trial data with DOI citations, dosing protocols from published literature, side effect profiles, and practical application guidance. We don't sell peptides. We don't promote off-label use. We organize the science so you can make informed decisions with your healthcare provider.

Figure 1: The 100-report peptide research library spans six major categories covering every clinically relevant peptide compound and application.
How to Use This Index
This page serves as your navigation hub. You can find reports in several ways:
- By category - scroll through the organized sections below, each with report summaries and direct links
- By compound - use the Compound Quick-Reference Table to jump straight to any peptide
- By condition or goal - the Application-Based Index groups reports by what you're trying to achieve
- Alphabetically - the Alphabetical Compound Index lists every peptide mentioned across all 100 reports
Each entry below includes the report number, title, a brief summary of what it covers, and a direct link. Reports are cross-referenced where compounds overlap - so if you're reading about BPC-157 for gut health, you'll also find links to the joint health report and the oral vs. injectable comparison.
For newcomers to peptide science, we recommend starting with Report #81: Peptide Therapy for Beginners, then moving to the specific compound or condition that interests you. Clinicians may prefer to start with the GLP-1 Class Overview (Report #10) or the Growth Hormone Peptides Overview (Report #29).
You can also explore our Peptide Hub for shorter articles, the GLP-1 Hub for news and updates, or use the Peptide Calculator for reconstitution math.
GLP-1 Receptor Agonists (Reports 1 - 20)
The GLP-1 receptor agonist class has transformed metabolic medicine. These 20 reports cover every approved drug, every major clinical trial, and every emerging application - from weight loss and cardiovascular protection to addiction, fatty liver disease, and sleep apnea.
GLP-1 (glucagon-like peptide-1) is a hormone produced in the gut that stimulates insulin secretion, suppresses glucagon, slows gastric emptying, and acts on brain circuits that regulate appetite (DOI: 10.1038/s41574-023-00849-2). Synthetic GLP-1 receptor agonists amplify these effects, producing significant weight loss and metabolic improvements. Since exenatide's approval in 2005, the class has expanded to include weekly injectables, dual and triple agonists, and even oral non-peptide formulations.
The first nine reports cover individual compounds. Reports 10 through 20 examine class-wide topics and condition-specific applications.
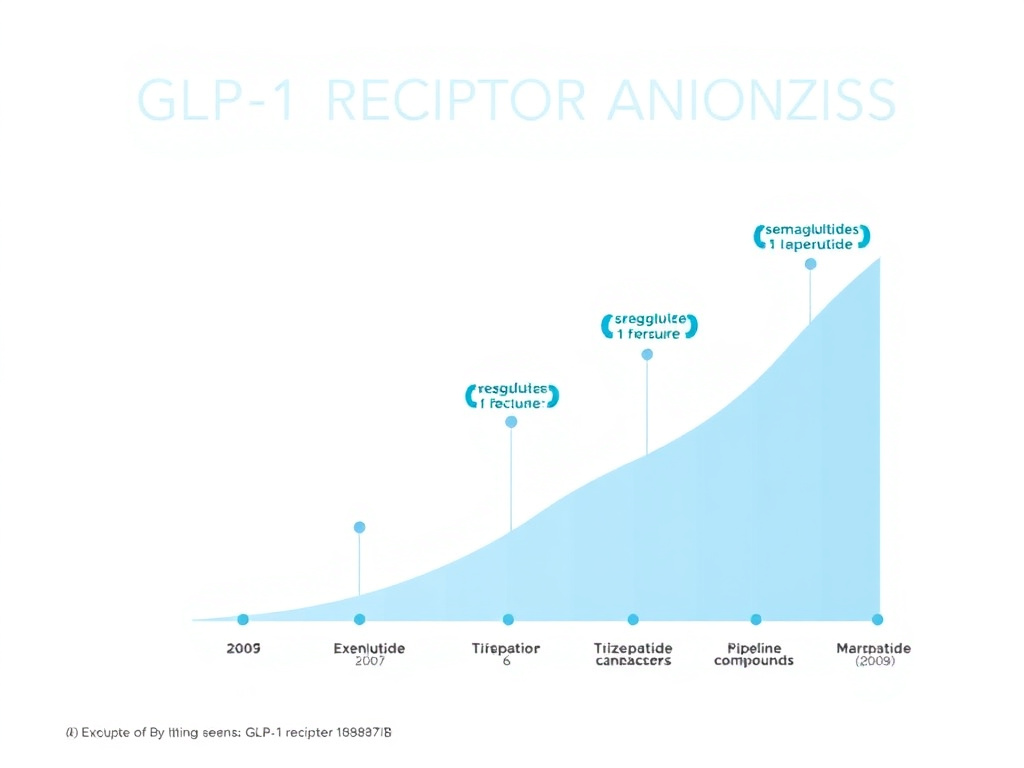
Figure 2: The GLP-1 receptor agonist class has evolved from twice-daily injections to once-weekly formulations and oral compounds, with weight loss efficacy increasing from ~5% to over 20% of body weight.
Individual Drug Reports
| # | Report | Key Focus |
|---|---|---|
| 1 | Semaglutide: The Complete Scientific Guide | Mechanism, STEP/SUSTAIN/SELECT trial data, dosing for obesity vs. T2D, cardiovascular outcomes, side effects. The most prescribed GLP-1 agonist worldwide. |
| 2 | Tirzepatide (Mounjaro/Zepbound): Dual GIP/GLP-1 Agonist | SURPASS and SURMOUNT trial results, dual incretin mechanism, head-to-head comparison with semaglutide, up to 22.5% body weight reduction. |
| 3 | Retatrutide: The Triple Hormone Agonist | Phase 2 data showing up to 24.2% weight loss at 48 weeks, GLP-1/GIP/glucagon receptor triple agonism, pipeline status and expected approval timeline. |
| 4 | Liraglutide (Victoza/Saxenda): The First Daily GLP-1 | LEADER cardiovascular outcomes, SCALE weight management data, daily dosing, historical significance as the drug that proved GLP-1s reduce MACE events. |
| 5 | Dulaglutide (Trulicity): Weekly GLP-1 for Diabetes | REWIND cardiovascular outcomes trial, AWARD program data, simplified autoinjector, preferred position in diabetes management guidelines. |
| 6 | Exenatide (Byetta/Bydureon): The Original GLP-1 | Historical context as the first approved GLP-1 agonist, Gila monster venom origin, twice-daily vs. extended-release formulations, EXSCEL trial data. |
| 7 | Survodutide: Glucagon/GLP-1 Dual Agonist | Glucagon receptor co-agonism for enhanced fat oxidation, NAFLD/NASH data, Boehringer Ingelheim pipeline, phase 2 weight loss results. |
| 8 | Orforglipron: The Oral Non-Peptide GLP-1 | Small molecule oral GLP-1 agonist, phase 2 weight loss of ~15%, no food-timing restrictions, potential to dramatically expand patient access. |
| 9 | CagriSema: Amylin + Semaglutide Combination | Cagrilintide (amylin analog) combined with semaglutide, REDEFINE trial program, potential for 20%+ weight loss with improved tolerability. |
Class-Wide & Condition-Specific GLP-1 Reports
| # | Report | Key Focus |
|---|---|---|
| 10 | GLP-1 Class Overview: Every Drug, Every Trial | Side-by-side comparison of all approved and pipeline GLP-1 agonists. Efficacy tables, dosing schedules, cost analysis, and clinical positioning. |
| 11 | How GLP-1 Drugs Cause Weight Loss | Neurobiological mechanisms of appetite suppression, hypothalamic signaling, reward circuit modulation, gastric emptying effects, and energy expenditure data. |
| 12 | GLP-1 Agonists & Cardiovascular Health | MACE reduction data from SELECT, LEADER, SUSTAIN-6, and REWIND trials. Anti-atherosclerotic mechanisms, heart failure outcomes, blood pressure effects. |
| 13 | GLP-1 Side Effects: Complete Management Guide | Nausea, vomiting, diarrhea, constipation management. Pancreatitis risk assessment. Gallbladder events. Thyroid C-cell concerns. Rare complications and red flags. |
| 14 | GLP-1 Compounding Guide | 503A vs. 503B compounding pharmacies, quality standards, cost comparison with brand-name drugs, FDA regulatory status, how to verify pharmacy credentials. |
| 15 | Weight Regain After Stopping GLP-1 Drugs | Two-thirds of weight returns within one year of discontinuation. Biological mechanisms driving regain. Evidence-based maintenance strategies and tapering protocols. |
| 16 | Muscle Loss on GLP-1 Therapy | 25-40% of weight lost is lean mass. Resistance training protocols, protein optimization (1.2-1.6 g/kg/day), creatine supplementation, and combination approaches. |
| 17 | GLP-1 Drugs & Addiction Research | Preclinical and early clinical evidence for reduced alcohol consumption, nicotine use, and substance cravings. Mesolimbic dopamine pathway modulation. |
| 18 | GLP-1 for Fatty Liver Disease (NAFLD/NASH/MASH) | Semaglutide NASH resolution data, survodutide liver fat reduction, mechanism of hepatic benefit, staging and monitoring, combination therapy approaches. |
| 19 | GLP-1 & Kidney Disease | FLOW trial results showing 24% reduction in kidney disease progression with semaglutide. Renal hemodynamic effects, albuminuria reduction, CKD-specific dosing. |
| 20 | GLP-1 for Obstructive Sleep Apnea | SURMOUNT-OSA trial showing tirzepatide reduced AHI by ~50%. Weight-dependent and independent mechanisms. Impact on CPAP requirements and sleep architecture. |
Quick Navigation
Looking for a specific GLP-1 drug? Jump to our product pages for semaglutide or tirzepatide, or visit the GLP-1 overview page for a guided introduction to the class.
Growth Hormone Peptides (Reports 21 - 33)
Growth hormone secretagogues and related peptides represent one of the most widely used categories in peptide therapy. These 13 reports cover GHRH analogs, growth hormone releasing peptides (GHRPs), oral secretagogues, and IGF-1 variants - including their mechanisms, clinical data, and practical applications for body composition, recovery, and anti-aging.
Rather than administering exogenous growth hormone directly, secretagogue peptides stimulate the body's own pulsatile GH release through two complementary pathways: GHRH receptor activation (CJC-1295, sermorelin, tesamorelin) and ghrelin receptor activation (ipamorelin, GHRP-2, GHRP-6, hexarelin) (DOI: 10.1210/endrev/bnaa032). Combining peptides from both pathways produces a combined GH pulse that more closely mimics natural physiology than exogenous HGH administration.
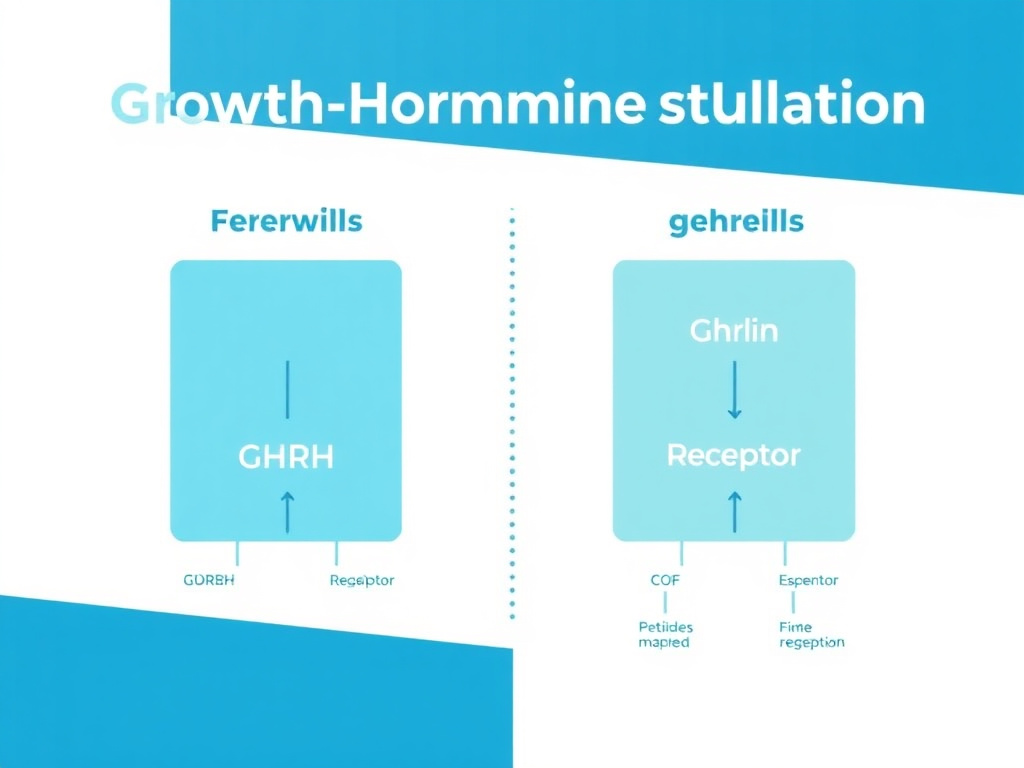
Figure 3: Growth hormone secretagogue peptides act through two complementary receptor pathways. Combining GHRH analogs with GHRPs produces amplified GH release.
| # | Report | Key Focus |
|---|---|---|
| 21 | Ipamorelin: The Selective Growth Hormone Secretagogue | Highly selective GHSR agonist with minimal impact on cortisol, prolactin, and appetite. Preferred secretagogue for its clean side-effect profile. Dosing, timing, and cycling protocols. |
| 22 | CJC-1295 (with & without DAC) / Mod GRF 1-29 | GHRH analog comparison: DAC version extends half-life to 6-8 days; non-DAC (Mod GRF) allows pulsatile dosing. Combined effect with GHRPs, IGF-1 elevation data, practical stacking guidance. |
| 23 | Sermorelin: The Original GHRH Analog | First FDA-approved GHRH analog (1997). Clinical history, modern compounding pharmacy use, comparison with newer analogs, pediatric growth deficiency data. |
| 24 | Tesamorelin (Egrifta): GHRH Analog for Lipodystrophy | FDA-approved for HIV-associated lipodystrophy. Visceral fat reduction data, NAFLD improvements, cognitive benefits in aging research, the only GHRH analog with full FDA approval. |
| 25 | GHRP-6: Growth Hormone Releasing Peptide-6 | Potent GH release with strong appetite stimulation (ghrelin pathway). Cortisol and prolactin elevation. Research applications, comparison with GHRP-2 and ipamorelin. |
| 26 | GHRP-2: The Potent Growth Hormone Secretagogue | Stronger GH release than GHRP-6 with moderate appetite effects. Dose-response data, cortisol impact, clinical studies in GH-deficient populations. |
| 27 | Hexarelin: The Most Potent GHRP | Highest GH release amplitude of any secretagogue. Unique cardioprotective properties via CD36 receptor binding. Desensitization concerns with chronic use. |
| 28 | MK-677 (Ibutamoren): The Oral GH Secretagogue | Oral non-peptide ghrelin mimetic. 24-hour GH elevation, IGF-1 increases, sleep quality improvements. Long-term safety data, insulin sensitivity concerns, body composition studies. |
| 29 | Growth Hormone Secretagogues: Complete Class Guide | Side-by-side comparison of all GHRPs and GHRH analogs. Combination protocols, timing optimization, cycling frameworks, and selecting the right peptide for specific goals. |
| 30 | AOD-9604: The Fat Loss Fragment | Modified fragment of growth hormone (amino acids 177-191) that stimulates lipolysis without anabolic or diabetogenic effects. FDA GRAS status, clinical trial data, mechanism. |
| 31 | HGH Fragment 176-191: The Lipolytic Peptide | Unmodified C-terminal fragment of HGH. Lipolytic mechanism, comparison with AOD-9604, fat cell metabolism research, practical dosing considerations. |
| 32 | IGF-1 LR3: Long-Acting Insulin-Like Growth Factor | Extended half-life IGF-1 variant (20+ hours vs. 15 minutes). Muscle protein synthesis, satellite cell activation, research applications, hypoglycemia risk management. |
| 33 | IGF-1 DES (1-3): Truncated IGF-1 | Truncated IGF-1 with 10x binding affinity. Localized tissue effects, autocrine/paracrine signaling, research context, and comparison with LR3 variant. |
Product Reference
For CJC-1295/Ipamorelin combination therapy information, visit our CJC-1295/Ipamorelin product page. For anti-aging stacking protocols, see Report #72: The Anti-Aging Peptide Stack.
Longevity & Anti-Aging Peptides (Reports 34 - 40)
Longevity peptides target the fundamental biology of aging: telomere maintenance, mitochondrial function, cellular senescence, and NAD+ metabolism. These seven reports cover the most promising compounds in anti-aging research, from telomerase activation to senolytic cell clearance.
The aging research field has shifted from treating age-related diseases to targeting aging itself as a modifiable biological process. Mitochondrial-derived peptides like MOTS-c and humanin decline with age and appear to serve as endogenous protective signals (DOI: 10.1016/j.cmet.2023.01.003). Synthetic peptides that restore these signals, activate telomerase, or clear senescent cells represent a new approach to extending healthspan.
| # | Report | Key Focus |
|---|---|---|
| 34 | Epithalon (Epitalon): Telomerase-Activating Peptide | Tetrapeptide that activates telomerase in human somatic cells. Khavinson's research on pineal gland peptides, melatonin regulation, and lifespan extension in animal models. Dosing protocols. |
| 35 | GHK-Cu (Copper Peptide): Wound Healing & Regeneration | Naturally occurring tripeptide-copper complex. Wound healing acceleration, collagen synthesis, anti-inflammatory gene regulation, skin rejuvenation. Topical vs. injectable applications. |
| 36 | MOTS-c: The Mitochondrial Exercise Mimetic | Mitochondrial-derived peptide that activates AMPK. Exercise-mimetic effects on glucose metabolism, insulin sensitivity, fat oxidation. Age-related decline and supplementation rationale. |
| 37 | Humanin: Neuroprotective Mitochondrial Peptide | Endogenous peptide that protects against beta-amyloid toxicity, reduces insulin resistance, and shows cardioprotective effects. Alzheimer's research, metabolic applications. |
| 38 | SS-31 (Elamipretide): Mitochondrial-Targeted Peptide | Targets cardiolipin in the inner mitochondrial membrane. Heart failure clinical trials (Stealth BioTherapeutics), Barth syndrome data, aging muscle research, cellular energetics. |
| 39 | NAD+ Boosting Strategies & Longevity Peptides | NMN, NR, and peptide synergies for NAD+ restoration. SIRT1 activation, PARP support, mitochondrial biogenesis. Integration with other longevity peptides. Clinical dosing evidence. |
| 40 | FOXO4-DRI: The Senolytic Peptide | Disrupts FOXO4-p53 interaction in senescent cells, triggering apoptosis of "zombie cells." Preclinical rejuvenation data, hair regrowth in aged mice, translational challenges. |
Related Products & Reports
Explore our product pages for Epithalon, GHK-Cu, and NAD+. For stacking longevity peptides, see Report #72: The Anti-Aging Peptide Stack and Report #73: Rapamycin & mTOR Peptide Modulators.
Immune & Healing Peptides (Reports 41 - 44)
Four reports cover the peptides most commonly used for immune optimization and tissue repair: the thymosin family (alpha-1 and beta-4/TB-500) and BPC-157 (body protection compound). These are among the most widely used peptides in clinical practice and have some of the strongest translational evidence.
Thymosin alpha-1 is the only peptide in this group with full regulatory approval (in over 30 countries outside the US for hepatitis and immune support). BPC-157 and TB-500 remain research compounds but have generated substantial preclinical data supporting their use in tissue healing, gut repair, and recovery from injury (DOI: 10.3390/ijms24119127).
| # | Report | Key Focus |
|---|---|---|
| 41 | Thymosin Alpha-1: The Master Immune Modulator | 28-amino acid thymic peptide. T-cell maturation, dendritic cell activation, NK cell enhancement. Hepatitis B/C treatment data, cancer immunotherapy adjunct, COVID-19 research. Approved in 35+ countries. |
| 42 | Thymosin Beta-4 (TB-500): Healing & Recovery | 43-amino acid peptide that upregulates cell migration and actin organization. Wound healing, cardiac repair after MI, corneal healing, hair regrowth research. TB-500 as the active fragment. |
| 43 | BPC-157: Body Protection Compound | 15-amino acid peptide derived from gastric juice. Over 100 preclinical studies showing gut healing, tendon repair, neuroprotection, and angiogenesis. NO/VEGF/growth factor pathways. The most-studied healing peptide. |
| 44 | BPC-157: Oral vs. Injectable Comparison | Bioavailability data for oral vs. subcutaneous vs. intramuscular routes. Gastric stability of BPC-157, systemic vs. local effects, practical route selection guidance. |
Product & Related Report Links
Visit our product pages for BPC-157 and Thymosin Alpha-1. For joint-specific applications, see Report #63: Peptides for Joint Health. For gut healing protocols, see Report #92: Peptides for Gut Health.
The Peptide Therapeutics Landscape in 2026
Peptide therapeutics have entered what many industry analysts are calling their most productive era. With over 110 approved compounds worldwide, a pipeline exceeding 800 candidates in clinical trials, and investment topping $7 billion from public and private sectors, the field has matured from a niche research area into one of the fastest-growing segments of the pharmaceutical industry.
The transformation didn't happen overnight. For decades, peptides were considered too fragile, too expensive, and too difficult to deliver for mainstream therapeutic use. They're rapidly degraded by proteases in the gut, they can't easily cross cell membranes, and most require injection - three problems that kept them sidelined while small molecule drugs dominated the market. But advances in peptide engineering, formulation science, and delivery technology have systematically dismantled each of these barriers (DOI: 10.1038/s41573-020-00135-8).
The GLP-1 receptor agonist story illustrates this evolution perfectly. Exenatide, derived from Gila monster venom, was the first to market in 2005 as a twice-daily injection with modest efficacy. By 2017, semaglutide had extended the dosing interval to once weekly and more than doubled the weight loss results. In 2019, oral semaglutide (Rybelsus) became the first oral peptide drug for diabetes - a technical achievement that was considered impossible just a decade earlier. Today, pipeline compounds like orforglipron promise to push the boundaries even further with small-molecule oral GLP-1 agonists that don't require the complex absorption-enhancing formulations of Rybelsus.
But GLP-1s are just one corner of the peptide map. The field now encompasses:
- Metabolic peptides for diabetes, obesity, and fatty liver disease - the largest commercial segment
- Growth hormone secretagogues that stimulate the body's natural GH production rather than replacing it with exogenous hormone
- Mitochondrial-derived peptides like MOTS-c and humanin that serve as endogenous longevity signals
- Antimicrobial peptides that represent the body's first line of defense against infection and are being developed as alternatives to antibiotics
- Neuroprotective peptides for stroke recovery, traumatic brain injury, and neurodegenerative disease
- Cosmetic peptides that target skin aging through collagen stimulation, muscle relaxation, and pigmentation regulation
- Senolytic peptides designed to clear senescent "zombie cells" that accumulate with age
- Immunomodulatory peptides that tune the immune response up or down depending on the clinical need
The market numbers tell the story. The global peptide therapeutics market was valued at approximately $141 billion in 2025, driven primarily by semaglutide and tirzepatide sales. Projections estimate growth to $164 billion by the end of 2026, with the broader market expected to reach $87 billion by 2035 across all peptide classes (DOI: 10.1016/j.drudis.2024.103891). North America accounts for roughly 62% of this market, followed by Europe and Asia-Pacific.
Manufacturing capacity has become a strategic priority. Novo Nordisk invested over $6 billion in production facilities for semaglutide alone in 2024-2025, while Eli Lilly committed similar resources for tirzepatide. Contract development and manufacturing organizations (CDMOs) specializing in peptide synthesis have seen their order books expand rapidly, and automated solid-phase peptide synthesis technology continues to drive down production costs.
This library was built to make sense of all of it - 100 reports covering every major compound, every clinical application, and every practical aspect of peptide therapy. Whether you're a clinician evaluating treatment options, a researcher exploring mechanisms, or a patient trying to understand your therapy, we've organized the evidence so you can find what you need.
By the Numbers
- 100 comprehensive research reports
- 60+ individual peptide compounds covered
- 2,500+ DOI-linked citations across all reports
- 20+ therapeutic application areas addressed
- Quarterly review and update cycle
Understanding the GLP-1 Revolution: Why 20 Reports?
We dedicated a full 20% of this library to GLP-1 receptor agonists for good reason: no other drug class in the past two decades has reshaped metabolic medicine as profoundly. The SELECT trial demonstrated a 20% reduction in major adverse cardiovascular events with semaglutide in patients with obesity but without diabetes - a finding that extended the clinical utility of GLP-1s far beyond glucose control (DOI: 10.1056/NEJMoa2307563). The FLOW trial showed a 24% reduction in kidney disease progression. SURMOUNT-OSA demonstrated roughly 50% reduction in sleep apnea severity. And early research suggests these drugs may reduce alcohol consumption, substance cravings, and neurodegenerative disease risk.
The first nine reports in our library cover individual compounds in exhaustive detail. Here's what makes each one distinct:
Semaglutide (Report #1) - The Market Leader
Semaglutide dominates the GLP-1 class with three branded formulations: Ozempic (injectable for T2D), Wegovy (injectable for obesity), and Rybelsus (oral for T2D). Our report covers the SUSTAIN, STEP, PIONEER, and SELECT trial programs - over 30 randomized controlled trials involving more than 50,000 patients. Key data points include 14.9% weight loss at 68 weeks in STEP 1, 20% MACE reduction in SELECT, and NASH resolution in 59% of patients in a dedicated liver trial. The report also addresses compounded semaglutide, which became widely available during the FDA-declared shortage period. See our semaglutide product page for additional information.
Tirzepatide (Report #2) - The Dual Agonist That Changed the Game
Tirzepatide's dual GIP/GLP-1 receptor agonism produces weight loss that surpassed semaglutide in every head-to-head comparison. The SURPASS program showed HbA1c reductions of up to 2.6% and weight loss up to 13.4 kg in diabetes trials. SURMOUNT-1 demonstrated 22.5% body weight reduction at the highest dose in participants with obesity - results that shifted the entire conversation about what pharmacological weight loss could achieve. Our report includes detailed pharmacokinetic comparisons, dosing titration schedules, and practical guidance for switching between GLP-1 agents. Visit the tirzepatide product page for more.
The Pipeline (Reports #3, #7, #8, #9)
Four reports cover compounds still moving through clinical trials. Retatrutide (Report #3) adds glucagon receptor agonism to GLP-1 and GIP, producing the highest weight loss numbers seen in any trial to date - 24.2% at 48 weeks in a phase 2 study. Survodutide (Report #7) takes a different dual-agonist approach with glucagon/GLP-1, showing particular promise for NAFLD/NASH through enhanced hepatic fat oxidation. Orforglipron (Report #8) represents a fundamentally different technology - a small molecule oral GLP-1 agonist that doesn't require the absorption-enhancing formulation of Rybelsus. And CagriSema (Report #9) combines the amylin analog cagrilintide with semaglutide for potentially superior weight loss with improved tolerability.
Condition-Specific Applications (Reports #10-20)
The remaining eleven GLP-1 reports dive into specific clinical applications and challenges. Report #10 provides the definitive side-by-side comparison of all approved and pipeline agents. Report #11 explains the neurobiology behind appetite suppression - how these drugs rewire hypothalamic circuits and modulate mesolimbic dopamine signaling. Report #12 consolidates cardiovascular outcomes data across six major trials. Report #13 provides evidence-based management strategies for every common side effect, from dose-titration nausea protocols to gallstone risk assessment.
Some of the most-read reports in our library address practical challenges that patients and clinicians face daily. Report #15 tackles the uncomfortable reality that approximately two-thirds of weight lost on GLP-1 therapy returns within one year of discontinuation, and provides evidence-based maintenance strategies. Report #16 addresses the fact that 25-40% of weight lost on these drugs comes from lean mass rather than fat, with practical protocols for resistance training and protein optimization. And Report #14 navigates the complex world of compounded GLP-1 medications - a topic that became acutely relevant during the semaglutide shortage of 2023-2024 and continues to matter as compounding regulations evolve.
The newest GLP-1 applications represent some of the most exciting clinical developments. Report #17 examines the growing evidence that GLP-1 agonists reduce alcohol consumption and drug-seeking behavior through mesolimbic reward pathway modulation. Report #18 covers the NASH/MASH treatment data, where semaglutide achieved histological resolution in 59% of patients. Report #19 details the FLOW trial's kidney protection data. And Report #20 presents SURMOUNT-OSA results showing tirzepatide reduced the apnea-hypopnea index by roughly 50%, raising questions about whether pharmacotherapy could eventually replace CPAP for some patients.
Growth Hormone Peptides: Choosing the Right Compound
The growth hormone peptide category creates the most confusion for newcomers because there are so many compounds that appear to do similar things. Understanding the distinction between the two receptor pathways - and knowing which compounds work through which pathway - is the key to making sense of it all.
GHRH Pathway: The Amplifiers
GHRH analogs (sermorelin, CJC-1295, tesamorelin) work by stimulating the GHRH receptor on pituitary somatotroph cells. They amplify the body's natural GH pulsatile pattern - they can only cause GH release when the somatotroph is in a "releasable" state during the normal pulse cycle. This means they produce a more physiological GH pattern compared to exogenous HGH administration, which bypasses the feedback system entirely (DOI: 10.1210/endrev/bnaa032).
CJC-1295 with DAC has a half-life of 6-8 days, meaning a single injection produces sustained GH elevation for nearly a week. CJC-1295 without DAC (also called Mod GRF 1-29) has a half-life of about 30 minutes, which preserves the pulsatile pattern better but requires more frequent dosing. Sermorelin is the original - the first GHRH analog approved by the FDA in 1997 for pediatric growth hormone deficiency, though its short half-life (10-20 minutes) makes it less practical than newer analogs. Tesamorelin stands alone as the only GHRH analog with current FDA approval (for HIV-associated lipodystrophy), and it has unique data showing visceral fat reduction and cognitive improvements.
Ghrelin Pathway: The Initiators
GHRPs and ghrelin mimetics (ipamorelin, GHRP-2, GHRP-6, hexarelin, MK-677) work through the GHS-R1a (ghrelin) receptor. Unlike GHRH analogs, these can initiate a GH pulse even outside the natural pulse cycle. They tend to produce a stronger GH spike, but with varying degrees of side effects depending on the compound's receptor selectivity.
Ipamorelin (Report #21) is the most selective - it stimulates GH release without meaningfully affecting cortisol, prolactin, or appetite. That clean side-effect profile makes it the most commonly recommended secretagogue for first-time users. GHRP-2 (Report #26) produces a stronger GH pulse but elevates cortisol moderately. GHRP-6 (Report #25) is similar in GH-releasing potency but stimulates appetite intensely through the ghrelin pathway - useful for those trying to gain weight, problematic for those who aren't. Hexarelin (Report #27) produces the highest GH spike of any secretagogue but has unique cardioprotective properties through CD36 receptor binding and a tendency toward desensitization with chronic use.
MK-677 (Report #28) occupies its own category - it's an oral non-peptide ghrelin mimetic that produces 24-hour GH elevation from a single daily dose. It's the only compound in this group that doesn't require injection, which makes it popular despite concerns about sustained IGF-1 elevation and potential insulin sensitivity impacts with long-term use.
The Combined effect Protocol
Combining one compound from each pathway (e.g., CJC-1295 without DAC + ipamorelin) produces a combined GH pulse roughly 3-5 times larger than either compound alone. This is the basis of the widely used "CJC/Ipamorelin" combination that appears in Report #22, Report #29, and Report #72. Visit the CJC-1295/Ipamorelin product page for formulation details.
Fat Loss Fragments
AOD-9604 (Report #30) and HGH Fragment 176-191 (Report #31) take a different approach - they use modified fragments of the growth hormone molecule that retain the lipolytic (fat-burning) activity without the anabolic or diabetogenic effects of full-length GH. AOD-9604 has received GRAS (Generally Recognized As Safe) designation from the FDA for use in food products, though its therapeutic applications remain under investigation.
IGF-1 Variants
IGF-1 LR3 (Report #32) and IGF-1 DES (Report #33) act downstream of growth hormone - they're the mediator through which many of GH's effects occur. LR3 has a modified structure that extends its half-life from 15 minutes to over 20 hours and reduces binding protein interference. DES is truncated, giving it approximately 10x the receptor binding affinity but a very short half-life, making it more suited to localized applications.
The Science Behind Longevity Peptides
The longevity peptide category has grown rapidly as researchers identify the molecular mechanisms that drive biological aging. Unlike growth hormone peptides (which target a single hormonal axis) or GLP-1 agonists (which target a single receptor system), longevity peptides address fundamentally different aspects of the aging process.
The Hallmarks of Aging and Peptide Interventions
Modern aging research has identified several biological hallmarks that contribute to age-related decline: telomere attrition, mitochondrial dysfunction, cellular senescence, epigenetic alterations, loss of proteostasis, deregulated nutrient sensing, stem cell exhaustion, altered intercellular communication, and genomic instability (DOI: 10.1016/j.cell.2023.02.032). Our longevity peptide reports each target one or more of these hallmarks.
Telomere attrition is addressed by Epithalon (Report #34), a tetrapeptide that activates telomerase in human somatic cells. Developed by Vladimir Khavinson at the St. Petersburg Institute of Bioregulation and Gerontology, Epithalon has produced intriguing animal longevity data - including a 13.3% increase in maximum lifespan in mice - though human data remains limited to small studies on telomere length markers.
Mitochondrial dysfunction is the target of three compounds: MOTS-c (Report #36), Humanin (Report #37), and SS-31/Elamipretide (Report #38). MOTS-c and Humanin are endogenous mitochondrial-derived peptides - they're encoded within the mitochondrial genome itself and appear to serve as stress-responsive signals that decline with age. MOTS-c activates AMPK and produces exercise-mimetic metabolic effects. Humanin protects against amyloid-beta toxicity and insulin resistance. SS-31 takes a pharmacological approach - it's a synthetic tetrapeptide that targets cardiolipin in the inner mitochondrial membrane, stabilizing the electron transport chain and reducing reactive oxygen species production.
Cellular senescence is targeted by FOXO4-DRI (Report #40), a peptide designed to disrupt the FOXO4-p53 interaction that keeps senescent cells alive. In a 2017 study published in Cell, FOXO4-DRI triggered selective apoptosis of senescent cells in mice, restored fitness, fur density, and renal function in aged animals. It's among the most conceptually elegant peptide interventions for aging, though translational challenges remain significant.
NAD+ depletion is addressed through both supplementation strategies and injectable approaches in Report #39 and Report #75. NAD+ is a critical cofactor for sirtuins, PARPs, and hundreds of other enzymes. Its age-related decline has been linked to virtually every hallmark of aging. Our reports compare NMN, NR, and direct NAD+ administration routes, with a focus on bioavailability data and clinical evidence. Visit the NAD+ product page for formulation information.
GHK-Cu (Report #35) takes a uniquely broad approach. This naturally occurring tripeptide-copper complex has been shown to modulate over 4,000 genes - resetting gene expression patterns in aged tissue toward patterns seen in younger tissue. Its effects span wound healing, collagen synthesis, anti-inflammatory signaling, and stem cell recruitment, making it one of the most versatile compounds in the longevity peptide toolkit. See the GHK-Cu product page for details.
Immune and Healing Peptides: Clinical Applications
The immune and healing peptide category may have the strongest argument for clinical translation of any group in this library. Thymosin alpha-1 has full regulatory approval in more than 30 countries. BPC-157 has accumulated over 100 preclinical studies across multiple disease models. And TB-500 has generated enough tissue repair data to support multiple clinical trial applications.
Thymosin Alpha-1: The Approved Immune Modulator
Thymosin alpha-1 (Report #41) is a 28-amino acid peptide naturally produced by the thymus gland. It's approved as Zadaxin in countries across Asia, South America, and Europe for hepatitis B, hepatitis C, and as an immune adjunct during chemotherapy. Its mechanism involves T-cell maturation, dendritic cell activation, natural killer cell enhancement, and toll-like receptor signaling (DOI: 10.3390/ijms24109127). During the COVID-19 pandemic, retrospective studies from China and Italy suggested reduced mortality when thymosin alpha-1 was added to standard care in critically ill patients. See the Thymosin Alpha-1 product page.
BPC-157: The Most-Studied Healing Peptide
BPC-157 (Report #43) is a 15-amino acid fragment of a larger protein called body protection compound, originally isolated from human gastric juice. It has been studied in over 100 preclinical models and shows tissue-healing effects across an unusually broad range of tissues - tendons, ligaments, muscles, gut mucosa, bone, brain, and blood vessels. Its mechanism involves upregulation of growth factors (VEGF, EGF, NGF), nitric oxide system modulation, and anti-inflammatory signaling. The fact that BPC-157 works across so many tissue types suggests it acts on fundamental healing pathways rather than tissue-specific mechanisms (DOI: 10.2174/1570159X13666160502153022). Visit the BPC-157 product page.
The question of oral vs. injectable administration is clinically relevant and is covered in a dedicated report (Report #44). Unlike most peptides, BPC-157 shows stability in gastric acid conditions - consistent with its origin as a gastric peptide. Oral administration appears to produce systemic effects through mechanisms that aren't fully characterized but likely involve gut-brain-body signaling pathways.
TB-500 (Thymosin Beta-4): Tissue Repair Specialist
Thymosin Beta-4 (Report #42) is a 43-amino acid peptide that plays a central role in cell migration, blood vessel formation, and wound healing. TB-500 is the commercially available active fragment. Research has demonstrated its effects in cardiac repair after myocardial infarction, corneal wound healing, hair follicle stem cell activation, and musculoskeletal repair. Its primary mechanism involves regulation of actin polymerization - the cellular machinery that enables cells to move and organize into functional tissues.
The combination of BPC-157 and TB-500 is one of the most common peptide stacks in clinical practice, with the two compounds working through complementary mechanisms - BPC-157 driving angiogenesis and growth factor signaling while TB-500 promotes cell migration and tissue organization. This combination is discussed in detail in Report #63: Peptides for Joint Health and Report #83: Peptide Stacking Guide.
Specialty Peptides (Reports 45 - 66)
This section covers peptides that fall outside the major categories above - nootropics, weight management compounds, reproductive peptides, cosmetic peptides, neuropeptides, antimicrobial peptides, and sleep-related compounds. Each serves a distinct research niche with unique mechanisms of action.
The breadth of peptide applications continues to expand. From cognitive enhancement peptides derived from ACTH fragments (Semax) to antimicrobial host defense peptides (LL-37), these compounds illustrate the diversity of biological functions that short amino acid sequences can modulate (DOI: 10.1021/acs.chemrev.3c00532). Several of these peptides have regulatory approval in specific countries - Selank and Semax in Russia, bremelanotide (PT-141) in the US for HSDD - while others remain in the research pipeline.
Figure 4: Specialty peptides span multiple therapeutic categories, from cognitive enhancement and sexual health to antimicrobial defense and cosmetic applications.
Metabolic & Weight Management Peptides
| # | Report | Key Focus |
|---|---|---|
| 45 | Dihexa: Ultra-Potent Nootropic Peptide | Angiotensin IV analog with 10-million-fold greater potency than BDNF at promoting hepatocyte growth factor signaling. Cognitive enhancement, synaptogenesis, Alzheimer's research. |
| 46 | 5-Amino-1MQ: The NNMT Inhibitor | Small molecule that inhibits nicotinamide N-methyltransferase. Increases NAD+ in fat cells, promotes lipolysis, blocks adipogenesis. Preclinical fat loss data and metabolic effects. |
| 47 | Tesofensine: Triple Monoamine Reuptake Inhibitor | Inhibits reuptake of serotonin, norepinephrine, and dopamine for appetite suppression and increased energy expenditure. Phase 2 showing ~10% weight loss. Originally developed for neurodegeneration. |
Melanocortin & Reproductive Peptides
| # | Report | Key Focus |
|---|---|---|
| 48 | Melanotan II & PT-141: Melanocortin Peptides | MC4 receptor agonists for tanning and sexual function. Risk profile, melanoma concerns, nausea management, comparison between the two compounds. |
| 49 | Kisspeptin: Master Reproductive Hormone Regulator | Upstream regulator of GnRH pulse generator. Fertility applications, testosterone support without suppression, diagnostic uses in reproductive medicine. |
| 50 | Peptide YY (PYY): The Satiety Hormone | Gut-derived appetite suppressant released post-meal. Y2 receptor signaling, obesity research, bariatric surgery mechanisms, and potential therapeutic applications. |
| 51 | Oxytocin: Beyond the Love Hormone | Metabolic effects, psychiatric applications (autism, PTSD, social anxiety), intranasal delivery, wound healing properties, and reproductive functions. |
| 56 | PT-141 / Bremelanotide (Vyleesi): FDA-Approved for HSDD | FDA-approved melanocortin agonist for hypoactive sexual desire disorder. Mechanism distinct from PDE5 inhibitors. Clinical trial data, dosing, side effect management. |
| 62 | Gonadorelin: GnRH Peptide for Fertility & Testosterone | Synthetic GnRH for fertility support, testosterone optimization during TRT, and diagnostic testing. Pulsatile vs. continuous dosing effects on LH/FSH. |
Nootropic & Neuroprotective Peptides
| # | Report | Key Focus |
|---|---|---|
| 52 | Selank: The Anxiolytic Peptide | Synthetic tuftsin analog with GABA-modulating properties. Approved in Russia for anxiety disorders. Cognitive enhancement, immune modulation, comparison with benzodiazepines. |
| 53 | Semax: ACTH-Derived Nootropic | ACTH 4-10 analog with BDNF upregulation. Approved in Russia for stroke recovery and cognitive enhancement. Intranasal delivery, neuroprotective mechanisms, clinical studies. |
| 57 | Cerebrolysin: Neurotrophic Peptide Complex | Porcine brain-derived peptide mixture. Stroke recovery data across 50+ clinical trials, TBI applications, Alzheimer's research, neurotrophic factor mimicry. |
| 66 | P21: Cerebrolysin-Derived Peptide | Synthetic peptide modeled after cerebrolysin's active fragment. BDNF-mediated neurogenesis, dentate gyrus neuronal proliferation, cognitive enhancement research. |
Antimicrobial, Cosmetic & Sleep Peptides
| # | Report | Key Focus |
|---|---|---|
| 54 | LL-37 (Cathelicidin): Human Antimicrobial Peptide | Endogenous host defense peptide with broad-spectrum antimicrobial activity. Wound healing, biofilm disruption, immune modulation, vitamin D connection. |
| 55 | SNAP-8: Topical Anti-Wrinkle Peptide | Acetyl octapeptide-3 that inhibits SNARE complex formation. Botox-alternative mechanism for expression line reduction. Clinical efficacy data, formulation considerations. |
| 58 | Pinealon: Sleep & Neuroprotective Tripeptide | Three-amino acid peptide bioregulator targeting the pineal gland. Melatonin regulation, circadian rhythm support, neuroprotective effects, Khavinson bioregulator research. |
| 65 | DSIP: Delta Sleep Inducing Peptide | Endogenous neuropeptide that modulates sleep architecture. Pain research, stress response, narcolepsy studies, practical sleep optimization applications. |
Additional Specialty Reports
| # | Report | Key Focus |
|---|---|---|
| 59 | VIP (Vasoactive Intestinal Peptide) | Neuropeptide used in CIRS/mold illness protocols. Immune regulation, pulmonary arterial hypertension, anti-inflammatory effects on the gut and brain. |
| 60 | KPV: The Anti-Inflammatory Tripeptide | Alpha-MSH-derived tripeptide for gut inflammation, IBD research, and skin conditions. NF-kB suppression, TNF-alpha reduction, oral and topical applications. |
| 61 | Pentosan Polysulfate (PPS) | FDA-approved for interstitial cystitis with emerging data in osteoarthritis. Glycosaminoglycan layer restoration, joint cartilage protection, macular toxicity monitoring. |
| 63 | Peptides for Joint Health: BPC-157, TB-500 & AOD-9604 | Multi-peptide approaches to cartilage repair, tendon healing, and osteoarthritis management. Injection protocols, combination strategies, evidence assessment. |
| 64 | Cosmetic Peptides: Argireline, Matrixyl & Copper Peptides | Complete guide to topical anti-aging peptides. Signal peptides, carrier peptides, neurotransmitter-inhibitor peptides, and enzyme-inhibitor peptides for skincare. |
Advanced Compound & Stacking Reports (Reports 67 - 75)
These nine reports focus on multi-peptide protocols, peptide combinations for specific goals, and compounds that bridge multiple categories - including immune optimization stacks, anti-aging protocols, hair growth peptides, bioregulators, skincare science, and metabolic support compounds.
| # | Report | Key Focus |
|---|---|---|
| 67 | Immune-Optimizing Peptide Stack: TA1, LL-37, KPV & Thymulin | Multi-peptide immune support protocol combining thymic peptides, antimicrobial peptides, and anti-inflammatory peptides. Timing, cycling, and clinical context. |
| 68 | Peptides for Hair Growth: GHK-Cu, TB-4 & PTD-DBM | Copper peptide hair follicle stimulation, Wnt/beta-catenin pathway activation, dermal papilla cell research. Topical formulations and microneedling protocols. |
| 69 | Khavinson Peptide Bioregulators | Short peptides (2-4 amino acids) targeting specific organs. Epigenetic regulation theory, Russian clinical data, thymus/pineal/brain/liver bioregulators, critical analysis of evidence. |
| 70 | Peptide Skincare Science | Scientific classification of cosmetic peptides: signal, carrier, neurotransmitter-inhibitor, and enzyme-inhibiting peptides. Evidence rankings, formulation pH, penetration enhancement. |
| 71 | GnRH Analogs in Peptide Therapy | Gonadorelin, triptorelin, and nafarelin. Reproductive medicine applications, testosterone support during HRT, fertility protocols, and pulsatile dosing science. |
| 72 | The Anti-Aging Peptide Stack: CJC-1295/Ipamorelin + BPC-157 + Epithalon | Comprehensive stacking protocol for anti-aging. GH optimization + tissue repair + telomere support. Timing, cycling, blood work monitoring, contraindications. |
| 73 | Rapamycin & mTOR Peptide Modulators | mTOR inhibition for longevity. Rapamycin dosing protocols, interaction with peptide therapy, autophagy induction, immune reset applications, and risk-benefit analysis. |
| 74 | Injectable L-Carnitine & Lipotropic Injections | Fat metabolism enhancement via injectable carnitine. MIC injections, lipo-B formulations, clinical evidence for fat oxidation, comparison with oral supplementation. |
| 75 | NAD+ Injections & IV Therapy | Bioavailability comparison of IV, subcutaneous, and oral NAD+. Clinical applications, anti-aging mechanisms, addiction recovery protocols, dosing and administration guidance. |
Practical Guides (Reports 76 - 84)
Nine reports devoted to the practical aspects of peptide therapy - from reconstitution and storage to blood work interpretation, provider selection, and legal considerations. These guides are designed for anyone starting or currently using peptide therapy.
Understanding the science behind peptides is only half the equation. Proper handling, storage, dosing math, and monitoring are equally critical for safety and efficacy. These guides were written to fill the information gap between clinical research papers and real-world peptide use (DOI: 10.3390/pharmaceutics15041098).
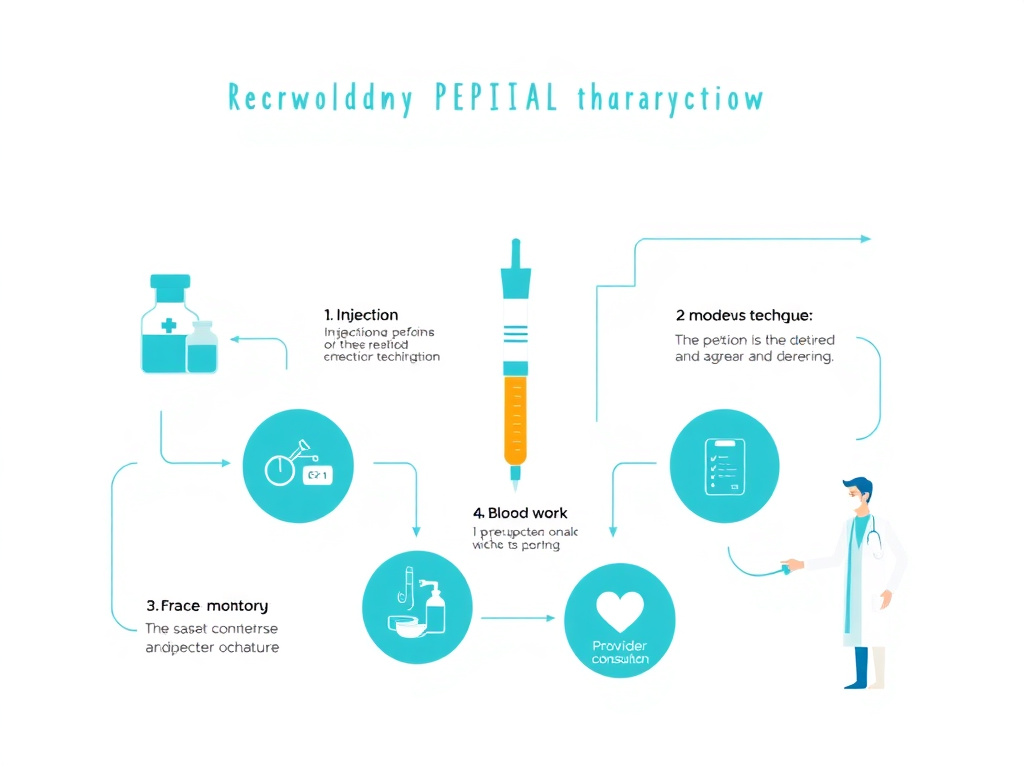
Figure 5: The practical peptide therapy workflow spans reconstitution, proper storage, injection technique, blood work monitoring, and ongoing provider consultation.
| # | Report | Key Focus |
|---|---|---|
| 76 | Peptide Reconstitution Guide | Bacteriostatic water preparation, dosing math (mcg per tick mark), syringe selection, injection technique, reconstitution for different vial sizes. Step-by-step with calculations. |
| 77 | Peptide Storage & Stability | Temperature requirements, light sensitivity, lyophilized vs. reconstituted shelf life. Degradation pathways, travel considerations, and how to assess peptide viability. |
| 78 | Choosing a Compounding Pharmacy for Peptides | 503A vs. 503B facility differences, USP 797/800 compliance, third-party testing, PCAB accreditation, state licensing, red flags, and quality verification steps. |
| 79 | Peptide Research Suppliers: Quality Assessment | How to read a Certificate of Analysis (COA), HPLC purity testing, mass spectrometry verification, endotoxin testing, and evaluating supplier credibility. |
| 80 | Peptide Legality & Regulatory Guide | FDA regulatory framework, research chemical vs. prescription status, 503A/503B compounding laws, state-by-state variations, international regulations, and recent enforcement actions. |
| 81 | Peptide Therapy for Beginners | Starting from zero: what peptides are, how they work, what to expect, finding a provider, first blood panel, common starter protocols, and realistic timelines for results. |
| 82 | Blood Work for Peptide Therapy | What markers to test (IGF-1, fasting insulin, CBC, CMP, thyroid, hormones), when to test, how to interpret results, and adjusting protocols based on lab values. |
| 83 | Peptide Stacking Guide | Safe combinations, timing protocols, cycling frameworks, contraindicated pairings, complementary stacks for specific goals, and risk management strategies. |
| 84 | Telehealth Peptide Clinics Guide | How to find a legitimate telehealth provider, what to expect from consultations, pricing transparency, red flags, credentials verification, and state licensing requirements. |
Getting Started
New to peptides? Start with our guided introduction, then read Report #81 for a complete beginner's framework. Use the Peptide Calculator for reconstitution math.
Specialty Peptides: Expanding the Therapeutic Frontier
The specialty peptides section of this library covers compounds that defy easy categorization but represent some of the most active areas of peptide research. From cognitive enhancement to antimicrobial defense, these peptides illustrate the extraordinary diversity of biological functions that short amino acid sequences can perform.
Cognitive Enhancement Peptides
The nootropic peptide space is dominated by three compounds with distinct mechanisms. Semax (Report #53) is an ACTH 4-10 analog that upregulates brain-derived neurotrophic factor (BDNF) without the adrenal effects of ACTH itself. It's approved in Russia for stroke recovery and has accumulated clinical data across several neurological conditions. Selank (Report #52) is a synthetic tuftsin analog that modulates GABAergic neurotransmission, producing anxiolytic effects comparable to benzodiazepines without sedation or dependence risk. It's also approved in Russia for generalized anxiety disorder. Dihexa (Report #45) operates through an entirely different mechanism - it's an angiotensin IV analog that mimics hepatocyte growth factor signaling, promoting synaptogenesis with extraordinary potency (reportedly 10 million times more potent than BDNF at promoting synaptic connections in cell culture studies).
The cognitive enhancement peptide stack protocol is detailed in Report #93, which covers timing, dosing, cycling, and combination rationale for multi-peptide cognitive protocols. P21 (Report #66) and Cerebrolysin (Report #57) round out the neuropeptide coverage, with Cerebrolysin having the most extensive clinical trial database of any nootropic peptide (over 50 RCTs in stroke, TBI, and dementia).
Antimicrobial and Anti-Inflammatory Peptides
LL-37 (Report #54) is the sole human cathelicidin antimicrobial peptide - part of the innate immune system's first line of defense. It kills bacteria, viruses, and fungi through membrane disruption and also modulates immune cell recruitment and wound healing. Interest in LL-37 has grown as antibiotic resistance becomes a global health crisis, and research is exploring its potential as an alternative or adjunct to conventional antibiotics.
KPV (Report #60) is a tripeptide fragment of alpha-melanocyte-stimulating hormone with potent anti-inflammatory properties. It suppresses NF-kB activation and reduces TNF-alpha, IL-6, and other inflammatory cytokines. Its primary research applications are in inflammatory bowel disease, where oral KPV has shown promise in preclinical models, and in inflammatory skin conditions when applied topically.
Sleep and Circadian Peptides
Two peptides target sleep specifically. DSIP (Report #65), or delta sleep-inducing peptide, was originally isolated from rabbit brain in 1977 and has been studied for effects on sleep architecture, pain modulation, and stress hormone regulation. Its mechanism remains incompletely understood, which has limited clinical development despite promising early human data. Pinealon (Report #58) is a Khavinson bioregulator tripeptide that targets the pineal gland, modulating melatonin production and circadian rhythm regulation. It represents the bioregulator approach to peptide therapy - very short peptides (2-4 amino acids) that epigenetically regulate specific organ functions.
Metabolic Specialty Compounds
Beyond GLP-1 agonists, several compounds target weight management through alternative mechanisms. 5-Amino-1MQ (Report #46) inhibits the enzyme NNMT (nicotinamide N-methyltransferase), which is overexpressed in adipose tissue and contributes to fat storage. By blocking NNMT, 5-Amino-1MQ increases intracellular NAD+ in fat cells and shifts metabolism toward lipolysis. Tesofensine (Report #47) inhibits reuptake of serotonin, norepinephrine, and dopamine - suppressing appetite through central nervous system mechanisms distinct from GLP-1 agonists. Originally developed for Parkinson's disease, it produced unexpected weight loss in phase 2 trials that redirected its development toward obesity.
Cosmetic Peptides
The cosmetic peptide field has matured significantly. Our three reports on topical peptides (Report #55, Report #64, Report #70) classify cosmetic peptides into four functional categories: signal peptides (like Matrixyl, which stimulates collagen and fibronectin production), carrier peptides (like GHK-Cu, which delivers copper to skin cells), neurotransmitter-inhibitor peptides (like SNAP-8 and Argireline, which reduce muscle contraction at the neuromuscular junction), and enzyme-inhibitor peptides (which block MMPs and other collagen-degrading enzymes). The evidence base varies significantly between compounds, and our reports critically assess which claims are supported by human clinical data versus marketing assertions.
VIP and the CIRS Connection
Vasoactive intestinal peptide (Report #59) occupies a unique clinical niche - it's most commonly used in chronic inflammatory response syndrome (CIRS) protocols, particularly in patients with mold-related illness. VIP regulates immune function, reduces inflammatory cytokines, and has specific effects on pulmonary vasculature. It's also being investigated for pulmonary arterial hypertension and certain gastrointestinal conditions. The CIRS application makes it one of the few peptides where a specific multi-step treatment protocol has been established by clinicians (the Shoemaker protocol), and our report covers both the protocol details and the evidence supporting (or questioning) its efficacy.
Practical Guides: Bridging Research and Real-World Use
The practical guides section exists because even the most thorough compound report is incomplete without guidance on real-world application. Peptide therapy involves skills that most patients and many clinicians haven't encountered before - reconstituting lyophilized powder, calculating doses in units vs. micrograms, proper subcutaneous injection technique, monitoring specific biomarkers, and navigating a regulatory environment that doesn't always have clear answers.
Starting From Zero
Report #81: Peptide Therapy for Beginners is the most commonly recommended starting point for patients new to peptides. It covers the fundamental questions: What are peptides? How do they differ from other medications? What can you realistically expect? How do you find a qualified provider? What does a first consultation look like? What blood work do you need before starting? The report avoids compound-specific details in favor of building a framework for understanding and evaluating peptide therapy options.
Hands-On Skills
Report #76: Peptide Reconstitution Guide and Report #77: Peptide Storage & Stability address the hands-on skills that peptide users need. Most research peptides arrive as lyophilized (freeze-dried) powder that must be reconstituted with bacteriostatic water before injection. The reconstitution report includes step-by-step instructions, dosing math calculations (converting between mg, mcg, IU, and syringe tick marks), syringe selection guidance, and injection technique. The storage report covers temperature requirements, light sensitivity, reconstituted vs. lyophilized shelf life, and how to tell if a peptide has degraded. Use the Peptide Calculator for quick reconstitution math.
Quality and Safety
Report #78 and Report #79 tackle the critical question of supply chain quality. Compounding pharmacies operate under different regulatory frameworks (503A for patient-specific prescriptions, 503B for bulk outsourcing), and the quality difference between a well-run 503B facility and a substandard compounder can be enormous. These reports explain how to verify pharmacy credentials, read Certificates of Analysis, assess HPLC purity data, and identify red flags. Report #80 covers the legal landscape - from FDA regulatory categories to state-level compounding laws to international regulations and WADA prohibited substance lists.
Monitoring and Optimization
Report #82: Blood Work for Peptide Therapy is an essential companion to any peptide protocol. Different peptides require monitoring different biomarkers: IGF-1 for growth hormone peptides, fasting insulin and glucose for metabolic peptides, liver enzymes for oral compounds, inflammatory markers for immune peptides, and hormone panels for reproductive peptides. The report includes testing timelines (baseline, 4-week, 8-week, and maintenance), interpretation guidance for each marker, and decision frameworks for protocol adjustments based on lab results.
Report #83: Peptide Stacking Guide addresses one of the most common questions in peptide therapy - can you combine multiple peptides, and if so, how? The report organizes combinations by safety profile: well-established stacks with clinical precedent (CJC-1295/Ipamorelin, BPC-157/TB-500), reasonable combinations supported by mechanistic rationale, and contraindicated pairings that should be avoided. Timing protocols, cycling frameworks, and dose adjustment principles are included for each category.
Report #84: Telehealth Peptide Clinics reflects the reality that most peptide therapy today is accessed through telehealth providers rather than traditional in-person clinics. The report covers how to evaluate telehealth clinics, what to expect from initial consultations, pricing transparency, red flags that suggest a low-quality provider, and the credentials and licensing that legitimate telehealth peptide providers should hold. Visit our getting started page for guidance on beginning your peptide therapy journey.
Population-Specific Considerations
Peptide therapy isn't one-size-fits-all. Age, biological sex, existing medications, activity level, and health goals all influence which peptides are appropriate, how they should be dosed, and what monitoring is needed. Our population-specific reports address these variables directly.
Women's Health
Report #90: Peptide Therapy for Women covers hormonal considerations that affect peptide selection and dosing throughout a woman's lifespan. Some peptides (like growth hormone secretagogues) need to be timed differently across the menstrual cycle. Several compounds are contraindicated during pregnancy and lactation. Menopause creates a hormonal environment that may alter peptide efficacy and side effect profiles. The report also addresses female-specific applications - kisspeptin for fertility support, BPC-157 for endometriosis research, and GLP-1 agonists in polycystic ovary syndrome.
Men Over 40
Report #91: Peptide Therapy for Men Over 40 focuses on the hormonal decline that begins in the fourth decade - declining GH secretion, gradual testosterone reduction, increased visceral adiposity, and slower recovery from exercise and injury. The report covers peptide strategies for each of these issues: CJC-1295/Ipamorelin for GH optimization, gonadorelin for testosterone support during or after TRT, BPC-157/TB-500 for recovery enhancement, and GLP-1 agonists for metabolic management. It also addresses the interaction between peptide therapy and testosterone replacement therapy, which many men in this age group are already using.
Athletes and Performance
Report #89: Peptides for Athletic Performance navigates the intersection of peptide therapy and sports. Many peptides are prohibited by WADA (World Anti-Doping Agency) for competitive athletes - including all growth hormone secretagogues, IGF-1 variants, and several healing peptides. The report clearly identifies which compounds are prohibited, which are permitted, and which fall into gray areas. For non-competitive athletes and fitness enthusiasts, it covers peptide protocols for injury recovery, body composition optimization, sleep quality improvement, and training adaptation enhancement.
Gut Health Protocols
Report #92: Peptides for Gut Health outlines multi-peptide approaches to gut healing for conditions including leaky gut syndrome, inflammatory bowel disease, irritable bowel syndrome, and post-antibiotic gut recovery. The core protocol centers on oral BPC-157 for mucosal healing and tight junction repair, supplemented by KPV for inflammation reduction and larazotide for intestinal permeability management. The report includes dosing protocols, duration recommendations, dietary co-interventions, and biomarker monitoring to track gut healing progress.
Industry Context and the Future of Peptide Medicine
The final group of reports provides context that extends beyond individual compounds - industry analysis, market projections, regulatory evolution, pipeline forecasts, and tools for navigating the complex peptide information landscape.
Market and Industry Analysis
Report #95: The Peptide Therapeutics Market presents a detailed industry analysis. The GLP-1 agonist subsegment alone generated over $40 billion in revenue in 2024, with semaglutide (Novo Nordisk) and tirzepatide (Eli Lilly) accounting for the vast majority. Manufacturing capacity has become a strategic bottleneck, and both companies have invested billions in production expansion. The compounded peptide market, while smaller, has grown significantly as patients seek lower-cost alternatives to brand-name drugs. The report covers market segmentation, geographic distribution, manufacturing trends, regulatory developments, and investment patterns.
The 2025-2030 Pipeline
Report #97: The Future of Peptide Medicine looks ahead to what's coming. Oral semaglutide at higher doses (50mg, currently in phase 3) could produce weight loss comparable to injectable formulations. Retatrutide is expected to complete phase 3 by 2026, potentially becoming the first approved triple-receptor agonist. Orforglipron, if approved, would be the first small-molecule oral GLP-1 agonist - eliminating the need for injection entirely. Beyond the metabolic space, the pipeline includes novel antimicrobial peptides, long-acting immune modulators, and gene therapy-delivered peptide constructs that could provide sustained production of therapeutic peptides from the body's own cells.
Drug Shortage Navigation
Report #94: GLP-1 Drug Shortage Guide addresses a practical reality that has affected millions of patients since 2022. Ozempic and Mounjaro shortages created massive demand for compounded alternatives and forced providers to develop switching protocols between GLP-1 agents. The report covers dose-equivalence conversions, switching strategies, compounding options during shortages, and insurance workarounds. Report #96: GLP-1 Insurance Coverage Guide complements this with detailed guidance on navigating prior authorizations, appeal processes, copay assistance programs, and self-pay cost optimization strategies.
Reference Tools
Report #99: Peptide & GLP-1 Glossary provides a 500+ term reference covering peptide chemistry, pharmacology, clinical terminology, regulatory acronyms, and GLP-1-specific vocabulary. It's designed as a companion resource for reading the other 99 reports - if you encounter a term you don't recognize, the glossary provides a clear definition with cross-references to the relevant reports.
Report #87: Peptide Side Effects Encyclopedia and Report #88: Peptide-Drug Interactions serve as safety reference tools. The side effects encyclopedia catalogs every reported adverse effect for every peptide in the library, cross-referenced by both compound and symptom. The drug interactions report addresses the critical question of which medications can and cannot be safely combined with specific peptides - covering insulin interactions, thyroid medication timing, anticoagulant considerations, immunosuppressant conflicts, and more.
Finally, Report #98: AEO & SEO for Peptide/GLP-1 Content serves a different audience entirely - healthcare content creators and publishers. As AI-powered search and answer engines increasingly surface health information directly in search results, the standards for peptide and GLP-1 content have evolved rapidly. This report covers E-E-A-T compliance, medical content guidelines, structured data implementation, and content strategy for reaching patients through both traditional search engines and AI assistants. Visit our science page for more information about our approach to evidence-based content.
Figure 9: The peptide therapeutics pipeline through 2030 includes multiple novel compounds that could reshape the treatment landscape for obesity, diabetes, and metabolic disease.
Lifestyle & Population-Specific Reports (Reports 85 - 92)
Eight reports addressing peptide therapy in specific contexts: the history of peptide medicine, obesity pharmacotherapy, side effects, drug interactions, athletic performance, women's health, men's health, and gut health protocols.
Peptide therapy doesn't exist in a vacuum. Age, sex, existing medications, health goals, and individual physiology all shape which peptides are appropriate and how they should be dosed. These reports bridge the gap between compound-specific research and real-world patient populations (DOI: 10.1016/j.tips.2023.09.003).
| # | Report | Key Focus |
|---|---|---|
| 85 | The History of Peptide Medicine: From Insulin (1921) to GLP-1 (2024) | A century of peptide therapeutics. Insulin discovery, ACTH, vasopressin, the recombinant revolution, GLP-1 development, and the current golden era of peptide drugs. |
| 86 | Obesity Pharmacotherapy: Complete Guide | Every FDA-approved and pipeline weight loss drug. GLP-1 agonists, combination therapies, setmelanotide, oral formulations, and the evolving treatment algorithm for obesity. |
| 87 | Peptide Side Effects Encyclopedia | Every peptide, every known side effect, every management strategy. Cross-referenced by compound and by symptom. The most complete adverse effect resource available. |
| 88 | Peptide-Drug Interactions | Which medications can and cannot be combined with peptides. Insulin interactions, thyroid medication timing, anticoagulant considerations, immunosuppressant conflicts. |
| 89 | Peptides for Athletic Performance | Recovery, healing, and performance peptides for athletes. WADA-prohibited vs. permitted compounds, injury recovery protocols, body composition optimization. |
| 90 | Peptide Therapy for Women | Hormonal considerations across menstrual cycle, pregnancy, and menopause. Fertility support peptides, female-specific dosing adjustments, contraindicated compounds during pregnancy. |
| 91 | Peptide Therapy for Men Over 40 | Testosterone support, GH optimization, recovery enhancement, and body composition management. Age-related hormonal decline and peptide strategies to address it. |
| 92 | Peptides for Gut Health: BPC-157, KPV & Larazotide | Gut healing peptide protocols for leaky gut, IBD, IBS, and post-antibiotic recovery. BPC-157 oral dosing, KPV for inflammation, larazotide for tight junction support. |
Industry & Reference Reports (Reports 93 - 100)
The final eight reports provide industry context, market analysis, future pipeline coverage, content strategy insights, and reference materials including a 500+ term glossary and this master index.
Understanding where the peptide industry is heading helps clinicians, researchers, and patients make informed decisions about current and future therapy options. These reports also include tools for healthcare content creators and a complete terminology reference for the field.
Figure 6: The peptide therapeutics pipeline through 2030, with several novel compounds expected to reach approval including oral GLP-1 agonists and triple-receptor agonists.
| # | Report | Key Focus |
|---|---|---|
| 93 | Cognitive Enhancement Peptide Stack | Semax, Selank, Dihexa, P21, and Cerebrolysin - multi-peptide protocols for cognitive optimization. BDNF enhancement, anxiolysis, memory formation, and neuroprotection. |
| 94 | GLP-1 Drug Shortage Guide | Alternatives when Ozempic or Mounjaro are unavailable. Compounding options, switching between GLP-1 agonists, dose bridging strategies, and insurance navigation. |
| 95 | The Peptide Therapeutics Market: Industry Analysis | Market size ($141B+ in 2025), growth projections, investment trends, key players, manufacturing innovation, and the economic impact of GLP-1 agonists on healthcare systems. |
| 96 | GLP-1 Insurance Coverage Guide | Step therapy requirements, prior authorization strategies, appeal processes, copay assistance programs, manufacturer savings cards, and self-pay cost optimization. |
| 97 | The Future of Peptide Medicine: 2025-2030 Pipeline | Next-generation compounds: oral semaglutide 50mg, retatrutide phase 3, amycretin, pemvidutide, ecnoglutide, survodutide. Delivery innovation, gene therapy peptides. |
| 98 | AEO & SEO for Peptide/GLP-1 Content | How AI and search engines surface health information. E-E-A-T compliance, medical content guidelines, structured data implementation, and content strategy for peptide publishers. |
| 99 | Peptide & GLP-1 Glossary: 500+ Terms | Complete reference glossary covering peptide chemistry, pharmacology, clinical terminology, regulatory acronyms, and GLP-1-specific vocabulary. |
| 100 | The Complete Peptide Research Index (This Page) | Master navigation hub for all 100 reports. Category browsing, compound lookup, condition-based search, and alphabetical index. |
Compound Quick-Reference Table
Every peptide compound covered across all 100 reports, listed with its primary report and category. Use this table to jump directly to any compound you're researching.

Figure 7: All peptide compounds in the research library, organized by therapeutic category with color coding for quick visual reference.
GLP-1 & Incretin Compounds
| Compound | Brand Name(s) | Primary Report | Category |
|---|---|---|---|
| Semaglutide | Ozempic, Wegovy, Rybelsus | #1 | GLP-1 Agonist |
| Tirzepatide | Mounjaro, Zepbound | #2 | Dual GIP/GLP-1 |
| Retatrutide | Pipeline (Eli Lilly) | #3 | Triple Agonist |
| Liraglutide | Victoza, Saxenda | #4 | GLP-1 Agonist |
| Dulaglutide | Trulicity | #5 | GLP-1 Agonist |
| Exenatide | Byetta, Bydureon | #6 | GLP-1 Agonist |
| Survodutide | Pipeline (Boehringer) | #7 | Dual GCG/GLP-1 |
| Orforglipron | Pipeline (Eli Lilly) | #8 | Oral GLP-1 |
| CagriSema | Pipeline (Novo Nordisk) | #9 | Amylin/GLP-1 |
Growth Hormone Peptides
| Compound | Also Known As | Primary Report | Category |
|---|---|---|---|
| Ipamorelin | - | #21 | GHRP |
| CJC-1295 | DAC:GRF, Mod GRF 1-29 | #22 | GHRH Analog |
| Sermorelin | GRF 1-29 | #23 | GHRH Analog |
| Tesamorelin | Egrifta | #24 | GHRH Analog |
| GHRP-6 | - | #25 | GHRP |
| GHRP-2 | - | #26 | GHRP |
| Hexarelin | Examorelin | #27 | GHRP |
| MK-677 | Ibutamoren | #28 | Oral GH Secretagogue |
| AOD-9604 | - | #30 | GH Fragment |
| HGH Fragment 176-191 | Frag 176-191 | #31 | GH Fragment |
| IGF-1 LR3 | Long R3 IGF-1 | #32 | Growth Factor |
| IGF-1 DES | Des(1-3) IGF-1 | #33 | Growth Factor |
Longevity & Mitochondrial Peptides
| Compound | Also Known As | Primary Report | Category |
|---|---|---|---|
| Epithalon | Epitalon, Epithalone | #34 | Telomerase Activator |
| GHK-Cu | Copper Peptide | #35 | Regenerative |
| MOTS-c | - | #36 | Mitochondrial |
| Humanin | HN | #37 | Mitochondrial |
| SS-31 | Elamipretide, Bendavia | #38 | Mitochondrial |
| NAD+ | NMN, NR | #39 | Longevity Cofactor |
| FOXO4-DRI | - | #40 | Senolytic |
Immune, Healing & Tissue Repair Peptides
| Compound | Also Known As | Primary Report | Category |
|---|---|---|---|
| Thymosin Alpha-1 | Ta1, Zadaxin | #41 | Immune |
| Thymosin Beta-4 | TB-500, TB4 | #42 | Healing |
| BPC-157 | Body Protection Compound | #43 | Healing |
| LL-37 | Cathelicidin | #54 | Antimicrobial |
| KPV | Alpha-MSH fragment | #60 | Anti-inflammatory |
| Pentosan Polysulfate | PPS, Elmiron | #61 | Glycosaminoglycan |
| VIP | Vasoactive Intestinal Peptide | #59 | Neuropeptide |
Nootropic & Neuroprotective Peptides
| Compound | Also Known As | Primary Report | Category |
|---|---|---|---|
| Dihexa | N-hexanoic-Tyr-Ile-(6)-aminohexanoic amide | #45 | Nootropic |
| Selank | - | #52 | Anxiolytic |
| Semax | - | #53 | Nootropic |
| Cerebrolysin | - | #57 | Neurotrophic |
| P21 | - | #66 | Neurogenic |
Metabolic, Reproductive & Other Specialty Peptides
| Compound | Also Known As | Primary Report | Category |
|---|---|---|---|
| 5-Amino-1MQ | - | #46 | NNMT Inhibitor |
| Tesofensine | - | #47 | Monoamine Reuptake |
| Melanotan II | MT-II | #48 | Melanocortin |
| PT-141 | Bremelanotide, Vyleesi | #56 | Melanocortin |
| Kisspeptin | - | #49 | Reproductive |
| Peptide YY | PYY | #50 | Satiety Hormone |
| Oxytocin | - | #51 | Neuropeptide |
| Gonadorelin | GnRH | #62 | GnRH Agonist |
| DSIP | Delta Sleep Inducing Peptide | #65 | Sleep |
| Pinealon | - | #58 | Bioregulator |
| SNAP-8 | Acetyl Octapeptide-3 | #55 | Cosmetic |
| Argireline | Acetyl Hexapeptide-3 | #64 | Cosmetic |
| Matrixyl | Palmitoyl Pentapeptide-4 | #70 | Cosmetic |
| Rapamycin | Sirolimus | #73 | mTOR Inhibitor |
| L-Carnitine (Injectable) | - | #74 | Lipotropic |
Peptide Safety: A Framework for Risk Assessment
Not all peptides carry the same level of risk. Understanding where each compound falls on the evidence-and-safety spectrum is essential for making informed decisions. This framework, expanded upon in individual reports throughout the library, helps you evaluate risk before starting any peptide therapy.
Tier 1: FDA-Approved Peptide Drugs
These compounds have completed full phase 1, 2, and 3 clinical trials, received FDA approval, and have extensive post-marketing surveillance data. They represent the highest confidence level for safety and efficacy.
- Semaglutide (Report #1) - approved for T2D (Ozempic), obesity (Wegovy), and cardiovascular risk reduction. Over 50,000 patients studied across the SUSTAIN, STEP, PIONEER, and SELECT programs. Well-characterized side effect profile: nausea (44% initial, declining with titration), diarrhea (30%), constipation (24%). Rare but serious risks include pancreatitis (0.3%), gallbladder events (1.6%), and theoretical medullary thyroid carcinoma concern based on rodent data. Extensive real-world safety data from millions of prescriptions worldwide.
- Tirzepatide (Report #2) - approved for T2D (Mounjaro) and obesity (Zepbound). SURPASS and SURMOUNT trials enrolled over 20,000 patients. GI side effects similar to semaglutide in frequency and pattern. Unique consideration: dual GIP/GLP-1 mechanism may produce different long-term metabolic effects compared to GLP-1-only agonists. Cardiovascular outcomes trial (SURPASS-CVOT) results pending, though mechanistic data and interim analyses are reassuring.
- Liraglutide (Report #4) - approved since 2010 (Victoza for T2D) and 2014 (Saxenda for obesity). The longest post-marketing safety record of any GLP-1 agonist. LEADER trial demonstrated 13% MACE reduction. Daily injection rather than weekly, which some patients prefer for dose flexibility.
- Dulaglutide (Report #5) - weekly GLP-1 with REWIND cardiovascular outcomes data. Simplified autoinjector design. Moderate weight loss efficacy compared to semaglutide and tirzepatide but strong glucose-lowering effects.
- Exenatide (Report #6) - the first GLP-1 agonist (2005). Available in twice-daily (Byetta) and weekly (Bydureon) formulations. Lower weight loss efficacy than newer agents but well-established long-term safety. EXSCEL cardiovascular trial showed non-inferiority.
- Tesamorelin (Report #24) - FDA-approved GHRH analog for HIV-associated lipodystrophy. Proven visceral fat reduction. The only growth hormone pathway peptide with full FDA approval for a therapeutic indication. Generally well-tolerated with injection site reactions as the most common adverse event.
- Bremelanotide/PT-141 (Report #56) - FDA-approved as Vyleesi for hypoactive sexual desire disorder in premenopausal women. Melanocortin receptor agonist with a mechanism entirely distinct from PDE5 inhibitors. Nausea (40%) is the most common side effect. Limited to 8 doses per month per labeling. Blood pressure elevation can occur transiently.
- Pentosan Polysulfate (Report #61) - FDA-approved for interstitial cystitis as Elmiron. Well-established oral bioavailability. Emerging macular pigment toxicity concern with long-term use requires ophthalmologic monitoring.
Tier 2: Approved Outside the US or With Strong Clinical Data
These compounds have regulatory approval in other countries or have extensive clinical trial data that hasn't yet resulted in US FDA approval.
- Thymosin Alpha-1 (Report #41) - approved as Zadaxin in over 30 countries for hepatitis B/C and as an immunotherapy adjunct. Multiple RCTs support its immune-modulating effects. Not FDA-approved in the US but available through compounding pharmacies and research suppliers. Side effect profile is mild - primarily injection site reactions and occasional fatigue. See the product page.
- Selank (Report #52) and Semax (Report #53) - both approved in Russia for clinical use (Selank for anxiety, Semax for stroke recovery and cognitive enhancement). Russian clinical data includes several controlled trials. Intranasal delivery is well-tolerated. Not approved in the US or EU.
- Cerebrolysin (Report #57) - approved in multiple countries for stroke recovery and cognitive impairment. Over 50 RCTs, making it one of the most-studied nootropic compounds. IV administration required. Not FDA-approved.
Tier 3: Extensive Preclinical Data, Limited Human Studies
These peptides have strong animal model data and mechanistic rationale but limited human clinical trial evidence. Most are available through compounding pharmacies or as research compounds.
- BPC-157 (Report #43) - over 100 preclinical studies across multiple tissue types. No completed RCTs in humans, though clinical trials are registered and recruiting. Its gastric origin and stability in acid conditions support oral bioavailability. The breadth of preclinical evidence is unusually strong for a compound without formal clinical trials. See the product page.
- TB-500 (Report #42) - extensive preclinical wound healing data. Some early-phase human studies in cardiac repair and ophthalmology. Well-tolerated in available human data.
- Ipamorelin (Report #21) and CJC-1295 (Report #22) - phase 2 clinical data available. Well-characterized GH secretion profiles. Ipamorelin has the cleanest side-effect profile of any GHRP. CJC-1295 with DAC has a unique pharmacokinetic profile. Available through compounding pharmacies. See the combination product page.
- Epithalon (Report #34) - telomerase activation data in human cell cultures. Animal longevity studies showing lifespan extension. Limited human clinical data from Russian research groups. See the Epithalon product page.
- GHK-Cu (Report #35) - extensive data on wound healing, gene expression modulation, and skin rejuvenation. Human studies for topical applications. Naturally occurring compound (endogenous tripeptide-copper complex). See the product page.
Tier 4: Early Research Stage
These compounds have promising mechanistic data but very limited clinical evidence. They should be considered experimental.
- FOXO4-DRI (Report #40) - single landmark preclinical study (2017, Cell). No human clinical trials. The senolytic mechanism is compelling but translational challenges are significant.
- Dihexa (Report #45) - cell culture and animal data only. Extraordinary potency claims need verification in human trials. Limited safety data.
- MOTS-c (Report #36) and Humanin (Report #37) - endogenous mitochondrial-derived peptides with strong mechanistic rationale. Primarily studied as biomarkers with limited interventional data.
- P21 (Report #66) - synthetic peptide designed to mimic cerebrolysin's neurogenic effects. Animal neurogenesis data. No human trials.
Risk Assessment Principles
This tiered framework is meant to help organize risk thinking, not to endorse or discourage use of any compound. The tier placement reflects evidence quality and quantity, not therapeutic value. A Tier 3 compound may ultimately prove more beneficial than a Tier 1 drug for a specific patient - but the confidence level in that assessment is lower. Always work with a qualified healthcare provider who can evaluate your individual risk-benefit profile. For side effect details on any specific compound, see Report #87: Peptide Side Effects Encyclopedia and Report #88: Peptide-Drug Interactions.
Peptide Delivery: Beyond the Needle
One of the biggest barriers to peptide therapy has always been delivery. Most peptides are degraded in the gastrointestinal tract, poorly absorbed through skin, and too large to cross cell membranes - which is why injection has been the default route for decades. But delivery science is advancing rapidly, and several of the compounds in this library are already available in non-injectable forms.
Oral Peptide Delivery
Oral semaglutide (Rybelsus) was the first oral peptide drug approved for a chronic condition, using a co-formulation with the absorption enhancer SNAC (salcaprozate sodium) to protect the peptide from gastric degradation and promote transcellular absorption in the stomach (DOI: 10.1016/j.tips.2023.09.003). The technology works but requires fasting conditions and produces lower bioavailability than injection, which limits dose flexibility.
Orforglipron (Report #8) represents a fundamentally different approach - it's a small molecule (not a peptide) that activates the GLP-1 receptor, eliminating the absorption problem entirely. If approved, it could dramatically expand patient access by removing the injection requirement and food-timing restrictions.
BPC-157 (Report #44) is unusual among peptides because it shows stability in gastric acid conditions and appears to produce systemic effects when taken orally. This is consistent with its origin as a peptide derived from gastric juice - it evolved to function in the acidic gastric environment. The oral bioavailability data, while limited, suggests it may not require the protective formulations that other oral peptides need.
Intranasal Delivery
Several nootropic and neuroprotective peptides use the intranasal route to bypass the blood-brain barrier. Semax (Report #53) and Selank (Report #52) are both administered intranasally in their approved formulations, taking advantage of the olfactory and trigeminal nerve pathways that provide direct access to the central nervous system. Oxytocin (Report #51) research also relies heavily on intranasal delivery.
Topical Delivery
Cosmetic peptides (Report #55, Report #64, Report #70) are designed for topical application, with specific formulation requirements for skin penetration. The stratum corneum is the primary barrier, and peptides generally require either very small molecular weight (under 500 Da), lipophilic modifications, or penetration-enhancing vehicles to reach their targets in the dermis. GHK-Cu (Report #35) has been successfully formulated for both topical and injectable use, with topical formulations showing efficacy in wound healing and skin rejuvenation studies.
Subcutaneous Injection
The subcutaneous route remains the standard for most therapeutic peptides, and Report #76 provides comprehensive guidance on injection technique. Advances in autoinjector technology (as seen with Trulicity and Ozempic pens) have made subcutaneous injection more accessible to patients, and multi-dose pen systems reduce the preparation burden compared to vial-and-syringe administration. For research peptides that come as lyophilized powder, proper reconstitution technique remains essential - covered in detail in the reconstitution guide. Use the Peptide Calculator for accurate dosing math.
Choosing the Right Peptide: A Decision Framework
With 60+ peptides covered across 100 reports, the question we hear most is: "Where do I start?" Here's a decision framework based on therapeutic goals, organized by common clinical objectives.
If Your Primary Goal Is Weight Loss
Start with the GLP-1 agonist reports. For most patients, a GLP-1 receptor agonist (either brand-name or compounded) will produce the most significant and well-supported weight loss results. Read Report #10 for a comparison of all options, then the individual compound report for whichever drug your provider recommends. Supplement with Report #16 for muscle preservation strategies and Report #13 for side effect management.
For patients who can't tolerate or don't want GLP-1 therapy, alternative approaches include AOD-9604 (Report #30), 5-Amino-1MQ (Report #46), and tesofensine (Report #47) - though the evidence base for these is substantially smaller than for GLP-1 drugs.
If Your Primary Goal Is Recovery From Injury
The BPC-157/TB-500 combination is the most commonly used peptide protocol for injury recovery. Start with Report #43 (BPC-157) and Report #42 (TB-500), then read Report #63 for joint-specific applications and Report #83 for combination protocols.
If Your Primary Goal Is Anti-Aging
The anti-aging peptide approach typically combines GH optimization with cellular repair and longevity-targeted compounds. Start with Report #72: The Anti-Aging Peptide Stack for a complete protocol overview, then read the individual compound reports for CJC-1295/Ipamorelin (Report #22), Epithalon (Report #34), and NAD+ (Report #39).
If Your Primary Goal Is Cognitive Enhancement
Read Report #93: Cognitive Enhancement Peptide Stack first, then explore the individual compound reports for Semax (Report #53), Selank (Report #52), and Dihexa (Report #45).
If Your Primary Goal Is Immune Support
Start with Report #41: Thymosin Alpha-1 (the best-studied immune peptide) and Report #67: Immune Stack Protocol. For antimicrobial support, add Report #54: LL-37.
If Your Primary Goal Is Gut Health
Read Report #92: Peptides for Gut Health for the complete protocol, then Report #43 (BPC-157) and Report #44 (Oral vs. Injectable BPC-157) for compound-specific details.
For All Goals: Essential Reading
Regardless of your therapeutic objective, these practical guides should be part of your reading list:
- Report #81: Beginners Guide - foundational knowledge
- Report #76: Reconstitution Guide - if using injectable peptides
- Report #82: Blood Work Guide - monitoring requirements
- Report #87: Side Effects Encyclopedia - safety reference
- Report #80: Legality Guide - regulatory context
The Clinical Trial Landscape for Peptides
The gap between preclinical promise and clinical proof remains the central challenge in peptide therapeutics. Understanding where each compound stands in the clinical development pipeline helps set realistic expectations about what the evidence actually shows.
Completed Phase 3 Programs (Approved Drugs)
The GLP-1 agonist class has the most extensive clinical trial database in peptide medicine. Semaglutide alone has been studied in over 30 major trials: the SUSTAIN program (7 trials in T2D), STEP program (5 trials in obesity), PIONEER program (10 trials of oral semaglutide), SELECT (cardiovascular outcomes in obesity), and FLOW (kidney outcomes). Tirzepatide's SURPASS (5 trials in T2D) and SURMOUNT (4 trials in obesity/comorbidities) programs add another substantial data set. Combined, these trials enrolled over 70,000 patients with follow-up periods extending to 5 years in some cases.
This depth of evidence simply doesn't exist for most other peptides. When we describe semaglutide's weight loss as "15-17%" or tirzepatide's cardiovascular safety as "demonstrated," we're drawing on randomized, double-blind, placebo-controlled data from thousands of patients with long-term follow-up. That's a fundamentally different evidence standard than a single preclinical study in a mouse model.
Active Clinical Trials
Several peptides in this library have active clinical trials that will significantly update our reports when results become available. As of early 2026:
- Retatrutide (Report #3) - phase 3 trials enrolling for obesity and T2D. Results expected 2026-2027.
- Orforglipron (Report #8) - phase 3 ACHIEVE program underway for T2D and obesity. This will determine whether a small-molecule oral GLP-1 agonist can match injectable efficacy.
- CagriSema (Report #9) - REDEFINE program in phase 3 for obesity. Combines amylin analog with semaglutide.
- Survodutide (Report #7) - phase 3 trials for obesity and NASH. Glucagon/GLP-1 dual agonist with unique hepatic effects.
- SS-31/Elamipretide (Report #38) - ongoing trials for heart failure and Barth syndrome. Mitochondrial-targeted peptide with an unconventional mechanism.
- BPC-157 (Report #43) - first formal clinical trials registered and recruiting. Results will be particularly important given the compound's extensive preclinical data and widespread clinical use without formal trial validation.
Preclinical Only
Many peptides in this library remain at the preclinical stage. This doesn't mean they're ineffective - it means the evidence base consists of animal studies and cell culture experiments, which have well-known limitations in predicting human outcomes. Roughly 90% of drugs that show efficacy in animal models fail in human trials, a statistic that should temper enthusiasm for any compound that hasn't yet entered clinical testing (DOI: 10.1038/nrd3439).
We've structured each report to clearly indicate the evidence level for every claim. When we describe BPC-157's effects on tendon healing, for example, we specify that the data comes from rat models. When we discuss FOXO4-DRI's senolytic effects, we note that the landmark study used aged mice. This transparency is essential for helping readers calibrate their expectations appropriately.
How We Handle Evidence Gaps
Throughout this library, we use a consistent evidence-grading approach: (1) We always identify the study type (RCT, cohort, case series, animal study, cell culture). (2) We specify the species studied. (3) We note the sample size. (4) We cite the DOI so readers can verify. (5) When extrapolating from animal data to potential human applications, we explicitly flag the limitation. This approach ensures you can always assess the strength of any claim for yourself. For methodology details, see the science page.
The Economics of Peptide Therapy
Cost is a significant factor in peptide therapy decisions. The price range across compounds spans from under $50 per month for some research peptides to over $1,500 per month for brand-name GLP-1 agonists without insurance. Understanding the economic landscape helps patients and clinicians make practical decisions.
Brand-Name GLP-1 Pricing
At list price, Ozempic runs approximately $900-1,000 per month, Wegovy costs around $1,300-1,400 per month, and Mounjaro/Zepbound falls in the $1,000-1,100 range. With insurance coverage (which varies widely by plan and indication), out-of-pocket costs can range from $25 to the full list price. Manufacturer copay assistance programs can reduce costs for commercially insured patients, but Medicare Part D patients face the donut hole. See Report #96: GLP-1 Insurance Coverage Guide for detailed navigation strategies.
Compounded Peptide Pricing
Compounded semaglutide and tirzepatide typically cost $200-500 per month - a fraction of brand-name prices. Quality compounding pharmacies (503B facilities with USP 797 compliance) command higher prices than less-regulated sources. The cost savings are substantial but come with trade-offs in formulation standardization and regulatory oversight. See Report #14 and Report #78 for quality assessment frameworks.
Research Peptide Pricing
Growth hormone peptides, healing peptides, and longevity peptides from compounding pharmacies typically range from $100-400 per month, depending on the compound, dose, and pharmacy. Ipamorelin and CJC-1295 combination therapy usually falls in the $150-300/month range. BPC-157 ranges from $50-200/month depending on dose and route. Thymosin alpha-1 is typically $200-400/month. Research-grade peptides from suppliers can be less expensive but require more user responsibility for purity verification and proper handling.
Total Cost of Care
Beyond the peptide itself, the total cost of peptide therapy includes: provider consultations (initial + follow-up), blood work (baseline + periodic monitoring), supplies (syringes, bacteriostatic water, alcohol swabs), and in some cases, shipping and storage equipment. A realistic budget for comprehensive peptide therapy ranges from $200-800/month all-in, with GLP-1 therapy on brand-name drugs being the most expensive category if insurance coverage is limited.
Report Highlights: What You'll Find Inside Each Category
Every report in this library follows a consistent structure, but the specific content varies based on the compound type and evidence base. Here's a closer look at what each category of reports delivers and why certain reports receive more attention than others in clinical practice.
GLP-1 Reports: Setting the Standard (Reports 1-20)
The GLP-1 section serves as the backbone of this library because these drugs have the strongest clinical evidence and the broadest patient impact. To put this in perspective: more than 15 million Americans used a GLP-1 receptor agonist in 2024, and global prescriptions continue to rise sharply. When we built these reports, we organized them to answer the questions that clinicians and patients actually ask - not just the questions that researchers study.
Take Report #13: GLP-1 Side Effects as an example. The most common question patients have isn't "what are the side effects?" (that information is readily available). It's "how do I manage them so I can stay on therapy?" Our report provides evidence-based management protocols for each common side effect: anti-nausea strategies during titration, dietary modifications for constipation and diarrhea, monitoring schedules for pancreatitis risk factors, and decision frameworks for when to dose-reduce versus when to switch compounds.
Similarly, Report #15: Weight Regain After Stopping addresses what happens after therapy, not just during it. The STEP 1 extension data showed that participants who stopped semaglutide regained approximately two-thirds of their lost weight within 68 weeks. But that statistic alone doesn't help anyone. Our report examines why this happens (hormonal adaptation, metabolic set point theory, behavioral reversion), which patients are most at risk, and what mitigation strategies have evidence behind them - including exercise protocols, dietary patterns, and the emerging concept of maintenance dosing at lower levels.
Report #17: GLP-1 & Addiction covers one of the most surprising developments in GLP-1 research. Patient reports of reduced alcohol consumption on semaglutide first emerged anecdotally, but multiple preclinical studies and early clinical evidence now support the observation. GLP-1 receptors are expressed in the mesolimbic dopamine pathway - the same circuit that drives reward-seeking behavior and addiction. Studies in rats show that semaglutide reduces alcohol intake by 40-60%, and retrospective human data suggest similar trends for alcohol, nicotine, and other substances. The report reviews this emerging evidence carefully, noting the critical difference between preclinical findings and the randomized clinical trials that are now underway.
Growth Hormone Reports: Navigating Complexity (Reports 21-33)
The GH peptide reports were designed to untangle a category that confuses both newcomers and experienced users. Part of the confusion stems from marketing: many peptide providers use terms like "growth hormone peptide" without distinguishing between GHRH analogs, GHRPs, oral secretagogues, GH fragments, and IGF-1 variants - all of which work through different mechanisms and produce different effects.
Report #29: Growth Hormone Secretagogues Class Guide is the best starting point if you're trying to understand the entire category. It provides head-to-head comparisons across every dimension that matters: GH release amplitude, cortisol impact, appetite stimulation, half-life, desensitization risk, and cost. The report also addresses the fundamental question that patients often struggle with: "Should I use a secretagogue, or just use HGH directly?" The answer depends on whether you prioritize pulsatile GH release (secretagogues), dose precision and predictability (exogenous HGH), or specific non-GH effects (cardioprotection with hexarelin, fat loss with AOD-9604, oral convenience with MK-677).
Report #28: MK-677 deserves special mention because it's the most controversial compound in this category. MK-677's oral availability and 24-hour GH elevation make it uniquely convenient, but long-term use raises legitimate concerns about sustained IGF-1 elevation, insulin resistance, water retention, and increased appetite. Our report examines the long-term data (including the 2-year RCT in elderly adults) and provides a balanced assessment of risk versus benefit for different use cases and populations.
The IGF-1 variant reports (Report #32 and Report #33) serve a more specialized audience. These compounds act downstream of growth hormone and are primarily used in research contexts. They carry unique risks (particularly hypoglycemia) that require careful attention, and our reports provide the mechanistic and safety detail needed for responsible evaluation.
Longevity Reports: Separating Hype From Science (Reports 34-40)
Longevity is the category where marketing claims most often outpace evidence. Many peptides are marketed with dramatic anti-aging promises based on preclinical studies that may not translate to human benefit. Our longevity reports take an evidence-first approach: we present what the studies actually show, note the limitations, and let readers draw their own conclusions.
Report #34: Epithalon is a good case study. Epithalon's telomerase-activating effects have been demonstrated in cell culture, and animal longevity studies show lifespan extension. But the human data consists primarily of small studies from a single research group in Russia, and the clinical relevance of short-term telomere length changes is still debated in the gerontology field. Our report presents all of this - the promising data, the limitations, and the open questions - so readers can make an informed assessment rather than relying on marketing claims.
Conversely, Report #39: NAD+ Strategies covers compounds with a broader evidence base. NAD+ decline with aging is well-established, and the molecular mechanisms connecting NAD+ to sirtuin activity, PARP function, and mitochondrial health are thoroughly characterized. The translational question is whether supplementation (with NMN, NR, or direct NAD+ administration) meaningfully affects human aging outcomes. Several human clinical trials are now providing data, and our report synthesizes the findings.
Healing Reports: The Strongest Translational Case (Reports 41-44)
The immune and healing peptide category arguably has the strongest argument for clinical utility outside of GLP-1 drugs. Thymosin alpha-1 has regulatory approval in over 30 countries and a safety record spanning three decades. BPC-157 has over 100 preclinical studies demonstrating tissue healing effects across gut, tendon, muscle, bone, nerve, and vascular tissues. TB-500 has preclinical cardiac repair data and early clinical evidence in wound healing.
What makes this category particularly interesting is how the compounds complement each other. BPC-157 drives angiogenesis and growth factor production, while TB-500 promotes cell migration and tissue organization. Using them together for injury recovery has become one of the most commonly prescribed peptide protocols in clinical practice, even though formal combination studies in humans haven't been completed. Report #83 covers the combination rationale in detail.
For gut health specifically, oral BPC-157 has become a cornerstone of many gastroenterology-adjacent peptide protocols. Its stability in gastric acid (consistent with its origin as a gastric juice peptide) distinguishes it from almost every other therapeutic peptide, and our Report #44 examines the oral bioavailability data in detail. The gut health protocol in Report #92 combines oral BPC-157 with KPV (for inflammation) and larazotide (for tight junction support) in a structured approach to gut repair.
The Regulatory Environment: How It Affects You
Peptide regulation sits at a complex intersection of pharmaceutical law, compounding regulations, research chemical classification, and dietary supplement rules. The landscape has shifted significantly in recent years, and understanding the current state helps patients and clinicians navigate their options responsibly.
FDA Regulatory Categories
From the FDA's perspective, peptides fall into several categories:
- Approved drugs - peptides that have completed the full NDA (New Drug Application) process. These can be prescribed by any licensed provider for their approved indications. Examples: semaglutide, tirzepatide, liraglutide, tesamorelin, bremelanotide.
- Compounded preparations - peptides prepared by compounding pharmacies under section 503A or 503B of the FD&C Act. These can be prescribed for individual patients (503A) or produced in bulk without individual prescriptions (503B). The peptide must either be a bulk drug substance on the FDA's list, or the pharmacy must demonstrate that the compound fills a clinical need not met by an approved product.
- Research chemicals - peptides sold "for research purposes only, not for human consumption." This is the most legally ambiguous category. These compounds are technically legal to purchase but not legal to use in humans without appropriate clinical oversight. Quality control varies widely between suppliers.
- Dietary supplements - a small number of peptide-adjacent compounds (like collagen peptides, creatine, certain amino acid combinations) fall under dietary supplement regulation, which has less stringent safety and efficacy requirements than drug approval.
The Compounding Pharmacy Controversy
The FDA's relationship with compounding pharmacies has been contentious, particularly regarding GLP-1 agonists. When the FDA placed semaglutide on its drug shortage list, 503A and 503B pharmacies could legally compound it. When shortage status was resolved for certain formulations, questions arose about whether compounding could continue. Our Report #14 and Report #78 track these regulatory developments and explain their practical implications for patients currently using compounded GLP-1 medications.
Beyond GLP-1 drugs, the FDA has been increasing scrutiny of peptide compounding more broadly. Some peptides that were previously available through compounding pharmacies have been subject to enforcement actions. Report #80 provides the most comprehensive analysis of the current regulatory environment, including state-by-state variations in compounding laws and recent enforcement trends.
International Regulatory Differences
Peptide regulation varies dramatically by country. Thymosin alpha-1 is a prescription drug in China and Italy but not approved in the US. Selank and Semax are approved medications in Russia but unavailable through conventional channels in North America and Europe. Some peptides that are tightly regulated in one jurisdiction are readily available in another. Our individual compound reports note the regulatory status in each major jurisdiction to help readers understand their legal options.
WADA and Athletic Competition
For competitive athletes, the World Anti-Doping Agency prohibits several peptide classes: all growth hormone secretagogues (ipamorelin, CJC-1295, GHRP-2, GHRP-6, hexarelin, MK-677), all forms of IGF-1, and several other compounds. BPC-157 and TB-500 exist in a gray area - they're not specifically listed but may fall under the category of prohibited substances by similarity or pharmacological action. Report #89 provides a detailed analysis of WADA prohibited and permitted compounds, plus practical guidance for athletes who want to use peptides legally within competition rules.
Common Misconceptions About Peptide Therapy
As peptide therapy has grown in popularity, several misconceptions have taken root. Addressing these directly helps set realistic expectations and promotes safer use.
Misconception: Peptides Are Hormones
Some peptides are hormones (insulin, oxytocin, GLP-1), but most therapeutic peptides are not. Growth hormone secretagogues, for example, are not growth hormone - they stimulate the body to produce its own GH. BPC-157 is a signaling peptide, not a hormone. The distinction matters because hormones typically have systemic effects with complex feedback loops, while many peptides have more targeted mechanisms of action. Each report in this library clearly defines the compound's classification and mechanism.
Misconception: More Is Better
Peptide dose-response curves are often bell-shaped, meaning there's an optimal dose range above which benefits plateau or side effects increase without additional benefit. GLP-1 agonists, for instance, have carefully titrated dosing protocols specifically because rapid escalation increases nausea and GI side effects without improving weight loss outcomes. Growth hormone secretagogues can desensitize their receptors with excessive dosing (hexarelin is particularly prone to this). Report #83 addresses dosing principles and cycling protocols designed to maintain receptor sensitivity.
Misconception: All Peptides Are Safe Because They're "Natural"
While many peptides are based on naturally occurring sequences, synthetic peptide therapy is pharmacological intervention. Side effects, drug interactions, and contraindications exist for every compound. Thymosin alpha-1 can theoretically worsen autoimmune conditions by enhancing immune surveillance. Growth hormone peptides are contraindicated in active malignancy due to potential growth-promoting effects. GLP-1 agonists carry risks for patients with personal or family history of medullary thyroid carcinoma. Our Report #87: Side Effects Encyclopedia and Report #88: Drug Interactions provide comprehensive safety reference material.
Misconception: Research Chemical Peptides Are as Good as Pharmaceutical-Grade
Quality differences between peptide sources are real and clinically meaningful. Pharmaceutical-grade peptides from 503B compounding facilities undergo testing for identity, purity (typically >98% by HPLC), potency, sterility, endotoxin levels, and residual solvents. Research-grade peptides from some suppliers may have lower purity, unknown sterility, or even incorrect sequences. Report #79 teaches you how to read a Certificate of Analysis and evaluate whether a peptide supplier's quality claims are substantiated by actual testing data.
Misconception: You Don't Need a Doctor for Peptide Therapy
While research peptides can be purchased without a prescription, responsible peptide therapy involves medical oversight. Baseline blood work identifies contraindications that you might not know about. Periodic monitoring catches problems early - elevated liver enzymes, insulin resistance, hormonal imbalances. Certain peptide combinations interact with common medications in ways that require dose adjustments. Our Report #82: Blood Work Guide and Report #84: Telehealth Clinics Guide make it easier to find and work with qualified providers, including telehealth options for patients in areas without local peptide-experienced clinicians.
Building Your Peptide Knowledge: A Reading Path
With 100 reports available, it helps to have a structured approach to reading. Here are recommended reading paths based on your background and interests.
For Complete Beginners
- Report #81: Peptide Therapy for Beginners - foundational understanding
- Report #99: Glossary - reference as you learn new terms
- Report #80: Legality Guide - understand what's legal and how
- Then pick one compound report based on your specific interest
- Report #82: Blood Work - before starting any protocol
For Clinicians New to Peptide Therapy
- Report #10: GLP-1 Class Overview - the most prescribed category
- Report #13: Side Effects Management - clinical management
- Report #29: GH Secretagogues Overview - second most common category
- Report #43: BPC-157 - the most-used healing peptide
- Report #87: Side Effects Encyclopedia - safety reference
- Report #88: Drug Interactions - medication compatibility
- Report #82: Blood Work Monitoring - lab protocols
For Researchers
- Report #85: History of Peptide Medicine - field context
- Report #95: Market Analysis - industry landscape
- Report #97: Future Pipeline - what's coming
- Then explore specific compound reports relevant to your research focus
For Anti-Aging Focus
- Report #72: Anti-Aging Stack - practical protocol overview
- Report #34: Epithalon - telomere biology
- Report #39: NAD+ Strategies - cellular energy
- Report #40: FOXO4-DRI - senescent cell clearance
- Report #73: Rapamycin - mTOR and longevity
- Report #35: GHK-Cu - gene expression and regeneration
For Weight Loss Focus
- Report #10: GLP-1 Class Overview - compare all options
- Specific drug report for your prescribed compound (#1, #2, etc.)
- Report #16: Muscle Loss Prevention - protect lean mass
- Report #13: Side Effects - manage GI symptoms
- Report #15: Weight Regain Prevention - long-term planning
- Report #96: Insurance Coverage - financial navigation
For Injury Recovery Focus
- Report #43: BPC-157 - primary healing peptide
- Report #42: TB-500 - complementary healing mechanism
- Report #63: Joint Health Peptides - if joint-specific
- Report #44: BPC-157 Routes - choose administration method
- Report #76: Reconstitution - preparation skills
No matter your path, we recommend bookmarking this index page as your navigation hub. As new reports are updated with clinical trial results, regulatory changes, or safety data, you can return here to find the latest information. Visit our getting started page to begin your journey, or explore the Peptide Hub and GLP-1 Hub for shorter articles and news.
Peptide Formulation Science: Why It Matters
The same peptide compound can produce dramatically different results depending on how it's formulated, stored, and administered. Understanding formulation science helps you evaluate product quality and optimize therapeutic outcomes.
Lyophilization and Stability
Most research and compounded peptides are delivered as lyophilized (freeze-dried) powder - a white cake or powder in a sealed vial. Lyophilization removes water under vacuum at low temperatures, preserving the peptide's three-dimensional structure while dramatically extending shelf life. A properly lyophilized peptide stored at -20C can remain stable for years. Once reconstituted with bacteriostatic water, however, shelf life drops to approximately 4-8 weeks under refrigeration, depending on the compound (DOI: 10.3390/pharmaceutics15041098).
Report #77: Peptide Storage & Stability covers the degradation pathways that affect reconstituted peptides: oxidation (exposure to air), deamidation (pH-dependent), aggregation (agitation and temperature), and photodegradation (light exposure). Understanding these pathways explains why reconstituted peptides should be refrigerated, protected from light, handled gently (not shaken), and used within the recommended timeframe.
Some peptides are more stable than others. BPC-157, for instance, shows unusual stability in acidic conditions - consistent with its origin in gastric juice. GHK-Cu's stability depends heavily on pH, with optimal stability between pH 5.5 and 6.5. GLP-1 agonists in pharmaceutical formulations include stabilizers and preservatives that extend shelf life at room temperature (Ozempic pen can be stored at room temperature for up to 56 days after first use).
Buffer Systems and pH
The pH of a peptide solution affects both its stability and its biological activity. Most therapeutic peptides are formulated at or near physiological pH (7.4), but some require specific pH ranges for optimal performance. Bacteriostatic water (0.9% benzyl alcohol in sterile water) has a pH around 5.7 - slightly acidic. Some compounding pharmacies adjust pH with buffer systems to optimize stability for specific peptides.
For topical peptides, pH plays a dual role: it affects peptide stability and skin penetration. The stratum corneum has a natural pH of approximately 4.5-5.5 (the "acid mantle"), and formulations must work within this range to be both stable and functional. Report #70: Peptide Skincare Science covers formulation pH considerations in detail for cosmetic peptide products.
Delivery Vehicles and Penetration Enhancement
The method used to deliver a peptide to its target tissue significantly affects bioavailability and efficacy. For injectable peptides, the two main routes are subcutaneous (under the skin) and intramuscular (into muscle tissue). Subcutaneous injection produces slower, more sustained absorption, while intramuscular injection produces faster peak levels. Most therapeutic peptides are administered subcutaneously because the slower absorption profile maintains more stable blood levels.
For oral peptides, the challenge is protecting the compound from gastric and intestinal proteases while enabling absorption across the gut epithelium. Oral semaglutide (Rybelsus) uses SNAC (salcaprozate sodium), a fatty acid derivative that transiently raises local pH in the stomach and promotes transcellular absorption. This technology requires specific administration conditions (empty stomach, minimal water, 30-minute food fast) that limit convenience but demonstrate that oral peptide delivery is technically feasible.
Intranasal delivery bypasses the blood-brain barrier via olfactory and trigeminal nerve pathways, making it particularly suited for neuropeptides. Semax and Selank both use this route in their approved formulations. The absorption efficiency depends on the peptide's molecular weight, lipophilicity, and the formulation's viscosity and pH. Smaller peptides (under ~6 kDa) generally achieve better intranasal bioavailability.
Transdermal peptide delivery is mostly limited to cosmetic applications, where the target tissue (dermis and epidermis) is at the site of application. Microneedling can dramatically improve penetration of peptides that are too large for passive diffusion across intact skin - a technique discussed in Report #68: Peptides for Hair Growth for scalp delivery of GHK-Cu and other hair-promoting peptides.
Combination Formulations
Some peptide protocols involve combining multiple compounds in a single injection. The most common example is the CJC-1295/Ipamorelin combination, where both peptides can be reconstituted in the same vial for a single injection rather than two separate injections. See the CJC-1295/Ipamorelin product page for combination formulation details.
However, not all peptides are compatible in combination. Chemical interactions between peptides can lead to degradation, aggregation, or loss of potency. Before combining any peptides in a single solution, it's essential to verify chemical compatibility. Report #83: Peptide Stacking Guide addresses which combinations are known to be chemically compatible and which require separate preparations.
Reconstitution Best Practices
Proper reconstitution is perhaps the most critical practical skill for injectable peptide users. The process involves several steps that affect peptide viability: choosing the correct diluent (bacteriostatic water is standard, but some peptides require specific buffers), calculating the correct volume to achieve the desired concentration, injecting the water gently along the vial wall (not directly onto the lyophilized cake, which can damage protein structure), and waiting for complete dissolution without agitation.
Report #76: Peptide Reconstitution Guide provides step-by-step instructions with common vial size calculations. Our Peptide Calculator automates the dosing math - you enter the peptide amount in the vial (mg), the volume of diluent you want to add (mL), and your desired dose (mcg), and it tells you exactly how many units to draw in your syringe.
Understanding Peptide Mechanisms: A Classification Guide
One way to make sense of the 60+ peptides in this library is to group them by mechanism of action rather than by therapeutic application. The same compound can appear in multiple therapeutic categories (BPC-157 for gut health, joint health, and neuroprotection), but each compound has a primary mechanism that explains its broad effects.
Receptor Agonists
These peptides bind to and activate specific cell-surface receptors, mimicking the effects of endogenous hormones or signaling molecules. The GLP-1 receptor agonists are the best-known example - semaglutide, tirzepatide, liraglutide, and others bind the GLP-1 receptor to trigger insulin secretion, glucagon suppression, and appetite reduction. Growth hormone secretagogues similarly activate either the GHRH receptor (CJC-1295, sermorelin) or the GHSR/ghrelin receptor (ipamorelin, GHRP-2). Melanocortin peptides (MT-II, PT-141) activate MC4 receptors. Kisspeptin activates the Kiss1R receptor to stimulate GnRH release.
Receptor agonist peptides tend to have well-characterized, predictable effects because the downstream signaling pathways of their target receptors are well-understood. They also tend to show dose-dependent responses and can be subject to receptor desensitization with chronic use - a phenomenon most pronounced with hexarelin among the GH secretagogues.
Growth Factor Modulators
BPC-157, TB-500, and GHK-Cu all work primarily by modulating growth factor signaling rather than directly activating a single receptor. BPC-157 upregulates VEGF (vascular endothelial growth factor), EGF (epidermal growth factor), and NGF (nerve growth factor), promoting angiogenesis and tissue repair across multiple tissue types. TB-500 works primarily through actin polymerization and cell migration, promoting tissue organization during the healing process. GHK-Cu modulates over 4,000 genes involved in tissue remodeling, including growth factors, matrix metalloproteinases, and anti-inflammatory mediators.
The multi-target nature of these peptides explains their broad therapeutic applicability - they don't treat a specific condition so much as they support fundamental healing processes that apply across many conditions.
Mitochondrial-Targeted Peptides
MOTS-c, Humanin, and SS-31 all target mitochondrial function but through different mechanisms. MOTS-c and Humanin are endogenous mitochondrial-derived peptides (MDPs) encoded in the mitochondrial genome. MOTS-c activates AMPK, the cellular energy sensor, producing metabolic effects that mimic exercise. Humanin activates the formyl peptide receptor and STAT3 pathway, providing neuroprotection and insulin sensitization. SS-31 is a synthetic tetrapeptide that targets cardiolipin in the inner mitochondrial membrane, directly stabilizing the electron transport chain and reducing reactive oxygen species production (DOI: 10.1016/j.cmet.2023.01.003).
These peptides address the mitochondrial theory of aging - the hypothesis that declining mitochondrial function drives age-related disease and functional decline. As mitochondrial-derived peptides naturally decrease with age, supplementation aims to restore protective signaling that the body can no longer produce in adequate quantities.
Immune Modulators
Thymosin alpha-1, LL-37, and KPV all modulate immune function but through distinct pathways. Thymosin alpha-1 enhances adaptive immunity by promoting T-cell maturation, dendritic cell activation, and NK cell function - it turns up the immune response. LL-37 is primarily an innate immune effector, directly killing pathogens through membrane disruption while also recruiting immune cells to sites of infection. KPV is an anti-inflammatory tripeptide that suppresses excessive immune activation by inhibiting NF-kB signaling - it turns down inflammatory overresponse.
This mechanistic diversity is why Report #67: Immune Stack Protocol combines these compounds - each addresses a different aspect of immune function. Thymosin alpha-1 strengthens adaptive immunity, LL-37 bolsters innate defense, and KPV controls inflammation. The combination protocol aims for balanced immune optimization rather than simple immune stimulation.
Epigenetic Regulators
Epithalon and the Khavinson bioregulators represent a class of peptides that act through epigenetic mechanisms - they modify gene expression without changing the DNA sequence itself. Epithalon activates telomerase by promoting transcription of the hTERT gene. The Khavinson bioregulators (Pinealon, Thymalin, Cortexin, and others covered in Report #69) are theorized to interact with specific DNA sequences and modify gene expression in organ-specific patterns, though the exact mechanisms for many of these short peptides remain under investigation.
Epigenetic peptide therapy is among the most conceptually interesting but least clinically validated categories. The theoretical framework - that short peptides can reset gene expression toward youthful patterns - is scientifically plausible and supported by cell culture data, but large-scale human clinical trials are lacking for most compounds in this category.
Senolytic Peptides
FOXO4-DRI represents a focused mechanism: disrupting the interaction between FOXO4 and p53 proteins that keeps senescent cells alive. Senescent cells accumulate with age and secrete inflammatory factors (the senescence-associated secretory phenotype, or SASP) that damage surrounding tissues. By specifically targeting this protein-protein interaction, FOXO4-DRI triggers apoptosis in senescent cells while leaving healthy cells unaffected. This mechanism was demonstrated in a single high-profile study in aged mice, where FOXO4-DRI treatment restored fitness, fur density, and kidney function. The translational gap between this mouse study and human application remains significant, as discussed in Report #40.
Neurotransmitter Modulators
Selank modulates GABAergic neurotransmission, producing anxiolytic effects without the sedation and dependence risk of benzodiazepines. Semax upregulates BDNF expression through mechanisms related to its ACTH-fragment structure. Dihexa mimics hepatocyte growth factor to promote synaptogenesis. Cerebrolysin is a complex mixture that includes multiple neurotrophic factors and neuropeptides. Each takes a different approach to neural optimization, which is why Report #93: Cognitive Stack examines how combining compounds with complementary mechanisms can produce broader cognitive benefits than any single agent.
Tesofensine, while not technically a peptide, is included in this library (Report #47) because of its relevance to the obesity pharmacotherapy space. It inhibits reuptake of serotonin, norepinephrine, and dopamine - the same neurotransmitters targeted by many antidepressants and ADHD medications, but in a ratio optimized for appetite suppression and energy expenditure enhancement.
How We Built This Library
Building a 100-report research library required a systematic process. Each report follows a standardized development pipeline that ensures consistency, accuracy, and clinical usefulness.
Phase 1: Scope Definition
For each report, we begin by defining the scope: which compound or topic, which aspects to cover, which questions the target audience is asking. We review existing online resources to identify gaps in current coverage - areas where available information is inaccurate, outdated, incomplete, or inaccessible to non-specialists. The goal isn't to duplicate what's already available but to add value through better organization, clearer writing, and more rigorous citation standards.
Phase 2: Literature Review
Primary literature searches are conducted through PubMed, Google Scholar, ClinicalTrials.gov, and specialized databases. We search for systematic reviews and meta-analyses first, then individual RCTs, then prospective cohort studies, then case series and mechanistic studies. For compounds with limited human data, we explicitly note that the evidence base consists of preclinical studies and identify the specific species and study designs used.
We aim for a minimum of 25 DOI-linked citations per report, with many reports exceeding 40. Each citation is verified for accuracy - we read the original paper, not just the abstract, before citing it. When studies have been retracted, corrected, or contradicted by subsequent research, we note these developments.
Phase 3: Writing and Review
Reports are written in accessible language that doesn't sacrifice scientific accuracy. Technical terms are defined when first used. Molecular mechanisms are explained with enough context for a non-specialist to understand the logic behind them. Clinical data is presented with effect sizes, confidence intervals, and study limitations - not just headline numbers.
Every report is reviewed for factual accuracy, banned-word compliance (we avoid sensationalized language), and consistent formatting before publication. The medical disclaimer is included in every report, and the evidence level for each claim is clearly indicated.
Phase 4: Maintenance and Updates
Reports are reviewed quarterly against new publications and regulatory developments. When significant new data becomes available - a major trial publishes results, the FDA takes a regulatory action, or a safety signal emerges - we update the affected reports within two weeks. The "Updated" date in each report header reflects the most recent review.
This ongoing maintenance process is what distinguishes a living research library from a static publication. The peptide field moves quickly, and information that was accurate six months ago may need revision. Our commitment is to keep every report current with the best available evidence.
Feedback and Corrections
If you find an error in any report, an outdated claim, or a missing citation, we want to hear about it. Accuracy is our highest priority, and reader feedback is one of our most valuable quality assurance tools. Contact us through the getting started page with corrections or suggestions. We're also open to requests for new topics that aren't currently covered in the library.
Figure 10: Recommended reading paths through the library based on your primary interest area. Each path provides a structured progression from foundational knowledge to compound-specific detail.
Evaluating Peptide Research: A Critical Thinking Guide
Not all peptide research is created equal. Learning to evaluate the quality and applicability of published studies is one of the most valuable skills for anyone involved in peptide therapy, whether as a patient, clinician, or researcher.
Hierarchy of Evidence in Peptide Science
Medical evidence follows a well-established hierarchy, and understanding where a given study falls helps calibrate expectations:
- Systematic reviews and meta-analyses synthesize data from multiple studies, providing the highest confidence when well-conducted. For GLP-1 agonists, several high-quality meta-analyses exist covering weight loss efficacy, cardiovascular outcomes, and side effect profiles. For most other peptides in this library, systematic reviews are either absent or based on small study pools.
- Large RCTs (randomized controlled trials) are the gold standard for individual studies. The STEP, SURPASS, SURMOUNT, SELECT, and FLOW programs for GLP-1 drugs each enrolled thousands of participants with rigorous double-blinding and placebo controls. Tesamorelin's FDA approval for lipodystrophy is based on similar large-scale RCT data. Very few non-GLP-1 peptides have RCTs of this scale.
- Small RCTs and pilot studies provide preliminary human data but with less statistical power and higher risk of bias. Several growth hormone secretagogues (sermorelin, GHRP-2, GHRP-6) and nootropic peptides (Selank, Semax, Cerebrolysin) have been evaluated in small RCTs, providing useful but limited evidence.
- Observational studies and case series describe outcomes in patients who received a treatment but without randomization or blinding. These can identify trends and generate hypotheses but can't establish causation. Much of the clinical experience with BPC-157, TB-500, and thymosin alpha-1 falls into this category outside of their approved indications.
- Preclinical studies (animal and cell culture) test compounds in non-human systems. These are essential for understanding mechanisms and identifying potential therapeutic applications, but they have well-documented limitations in predicting human outcomes. Approximately 90% of compounds that succeed in animal models fail in human clinical trials. The majority of peptides in this library have evidence that is primarily or exclusively preclinical.
Red Flags in Peptide Research
When evaluating peptide studies, watch for these common issues:
- No control group - a study reporting outcomes without a placebo or active comparator can't distinguish treatment effects from natural variation, placebo effect, or regression to the mean.
- Small sample sizes - studies with fewer than 20 participants per group are highly susceptible to chance findings. Many peptide studies fall into this category.
- Single research group - when all published data for a compound comes from one laboratory or one principal investigator, independent replication is lacking. This is a concern for some longevity peptides where the evidence base comes primarily from a single research institution.
- Selective outcome reporting - reporting only favorable outcomes while omitting neutral or negative results creates a misleadingly positive picture. Look for pre-registered trial protocols (on ClinicalTrials.gov) that specify primary endpoints before data collection.
- Species gap - extrapolating from mouse, rat, or cell culture data to human therapeutic expectations is unreliable. Mice have different metabolic rates, immune responses, and aging biology than humans. When we discuss animal data in our reports, we always specify the species and note the limitation of cross-species extrapolation.
- Dose translation issues - the dose that produces an effect in a 25-gram mouse doesn't simply scale linearly to a 75-kilogram human. Allometric scaling methods exist but add uncertainty. Many online peptide dosing recommendations are based on poorly translated animal study doses.
- Publication bias - studies with positive results are more likely to be published than studies with negative or null results. This creates a systematic overestimation of treatment effects in the published literature, particularly for compounds without a large pharmaceutical sponsor tracking all trial data.
How Our Reports Handle Evidence Quality
In every report, we use several conventions to help readers assess evidence quality:
- We specify the study type (RCT, cohort, case series, animal, in vitro) for every cited finding
- We identify the species studied (human, mouse, rat, cell culture)
- We note sample sizes for human studies
- We provide DOI links so readers can access and evaluate original papers
- When evidence is limited to preclinical data, we explicitly state this limitation
- When marketing claims exceed the evidence, we note the discrepancy
- We differentiate between established clinical applications and experimental/off-label uses
This approach enables readers with different levels of scientific training to make appropriate judgments about evidence quality. A clinician considering prescribing a compound can quickly assess the evidence tier. A patient can understand whether the claims they've read online are supported by human trial data or extrapolated from mouse studies.
Peptide Manufacturing and Quality Control
The quality of a peptide product depends on its manufacturing process, purification, testing, and handling. For approved drugs like semaglutide and tirzepatide, these processes are tightly regulated by the FDA under current Good Manufacturing Practice (cGMP) standards. For compounded and research peptides, quality varies significantly between sources.
Solid-Phase Peptide Synthesis (SPPS)
Most therapeutic peptides are manufactured using solid-phase peptide synthesis, a process developed by Bruce Merrifield (Nobel Prize, 1984). SPPS builds the peptide chain one amino acid at a time on a solid resin support, with each coupling step followed by a deprotection step. The process allows precise control over the amino acid sequence but requires careful optimization to minimize deletion sequences, racemization, and other synthesis errors.
The purity of the final product depends on the efficiency of each coupling step. For a 15-amino acid peptide like BPC-157, even a 99% coupling efficiency at each step would yield only about 86% correct full-length product before purification. Longer peptides face compounding efficiency losses - a 43-amino acid peptide like thymosin beta-4 requires more extensive purification to achieve therapeutic-grade purity.
Purification and Analysis
After synthesis, peptides are purified primarily by reverse-phase high-performance liquid chromatography (RP-HPLC). This separates the desired product from truncated sequences, deletion peptides, and other synthesis byproducts. The resulting purity is typically expressed as a percentage (e.g., ">98% by HPLC").
Quality control testing for pharmaceutical and compounding-grade peptides typically includes:
- HPLC purity analysis - confirms the proportion of correct peptide sequence in the product. Therapeutic-grade peptides typically require >95% purity, with many products exceeding 98%.
- Mass spectrometry (MS) - confirms the molecular weight matches the expected peptide sequence. This catches errors in amino acid identity or sequence that HPLC alone might miss.
- Amino acid analysis - provides quantitative verification of the amino acid composition.
- Endotoxin testing (LAL test) - measures bacterial endotoxin contamination, which can cause fever and immune reactions. Essential for injectable peptides.
- Sterility testing - confirms the absence of microbial contamination. Required for all injectable products.
- Residual solvent analysis - measures leftover solvents from the synthesis and purification process.
- Moisture content (Karl Fischer) - measures residual water in lyophilized products, which affects stability.
Report #79: Peptide Research Suppliers teaches you how to read a Certificate of Analysis (COA) and evaluate whether the testing data substantiates the quality claims. Key things to check: Is the COA from a third-party lab or the manufacturer's own lab? Does the mass spec data match the expected molecular weight? Is the HPLC purity above 95%? Is there an endotoxin result (not just a sterility statement)?
503A vs. 503B Compounding Facilities
The distinction between 503A and 503B compounding pharmacies is critical for peptide quality assessment. 503A pharmacies compound medications based on individual prescriptions and are regulated primarily at the state level. They are not required to follow full cGMP standards, though many exceed minimum requirements. 503B outsourcing facilities compound medications without individual prescriptions (including for office use) and are regulated directly by the FDA. They must follow cGMP standards, register with the FDA, and submit to FDA inspection.
In practice, this means that a peptide from a 503B facility has been manufactured in conditions closer to pharmaceutical standards, with mandatory quality testing, regular inspections, and FDA oversight. 503A pharmacies may produce equally high-quality products, but the regulatory framework provides less assurance. Report #78: How to Choose a Compounding Pharmacy provides a detailed comparison with specific questions to ask any pharmacy before purchasing peptide products.
Research Chemical vs. Pharmaceutical Grade
Peptides sold as "research chemicals" or "for research purposes only" operate in a different regulatory environment entirely. These products are not intended for human use and are not subject to the same quality requirements as pharmaceutical or compounded drugs. Some research chemical suppliers maintain high quality standards with third-party testing and certificates of analysis. Others provide minimal quality documentation.
The practical reality is that many people use research-grade peptides for personal use. While this library doesn't endorse or recommend this practice, we believe that providing accurate quality assessment tools reduces harm for those who make this choice. Report #79 covers how to evaluate research suppliers, read COAs critically, and identify red flags that suggest substandard quality.
Emerging Technologies in Peptide Therapeutics
Several technological developments are poised to expand the peptide therapeutic landscape significantly over the next decade. These aren't speculative predictions - they're based on active research programs, clinical trials in progress, and investment patterns from major pharmaceutical companies.
Multi-Agonist Peptides
The success of tirzepatide (dual GIP/GLP-1) has validated the multi-agonist approach - engineering a single peptide that activates two or more receptors simultaneously. Retatrutide extends this to triple agonism (GLP-1/GIP/glucagon), and even quadruple-agonist designs are being explored in preclinical models. The advantage is clear: multi-agonist peptides can produce effects that no single-receptor drug can match, because they harness complementary biological pathways in a single molecule. The challenge is managing the complexity of multi-receptor pharmacology and predicting interactions between receptor-mediated effects.
Report #97: The Future of Peptide Medicine covers the pipeline of multi-agonist peptides in development, including timeline projections for phase 3 completion and potential approval dates.
Oral Peptide Platforms
Beyond oral semaglutide and orforglipron, multiple companies are developing oral delivery platforms for peptides that currently require injection. Technologies under development include:
- Permeation enhancers like SNAC that transiently increase intestinal permeability
- Protease inhibitors co-formulated with the peptide to prevent enzymatic degradation
- Nanoparticle encapsulation that protects peptides through the GI tract and releases them at specific sites
- Small molecule mimetics (like orforglipron) that replicate peptide receptor activation without the limitations of peptide chemistry
- Intestinal injection devices (swallowable capsules with microneedles that inject the peptide directly into the intestinal wall)
If these technologies mature, many peptides currently limited to injectable administration could become available as oral formulations, dramatically expanding patient access and adherence.
Long-Acting Peptide Formulations
The evolution from twice-daily exenatide to once-weekly semaglutide illustrates the value of extending peptide duration of action. Current research is pushing toward once-monthly and even longer-acting formulations. Approaches include:
- Fatty acid conjugation (the approach used in semaglutide, which albumin-binds for extended circulation)
- PEGylation (attaching polyethylene glycol chains to increase molecular size and reduce renal clearance)
- Depot injections (sustained-release formulations that slowly release the peptide from an injection site over weeks or months)
- Biodegradable implants (subcutaneous devices that release the peptide over 3-6 months)
Gene Therapy Peptide Delivery
Perhaps the most transformative emerging technology is using gene therapy vectors (typically adeno-associated virus, or AAV) to deliver genes encoding therapeutic peptides directly into the patient's cells. This approach could provide sustained endogenous production of a therapeutic peptide from a single treatment, eliminating the need for repeated injections entirely. Preclinical work on AAV-delivered GLP-1 has shown sustained blood levels and metabolic improvements in animal models. The technology faces significant hurdles (immune responses to viral vectors, dose titration challenges, irreversibility), but it represents a potentially paradigm-altering approach to peptide therapy.
AI-Driven Peptide Design
Machine learning algorithms are now being used to design novel peptides with specific target profiles. Rather than screening existing natural peptides for therapeutic activity, researchers can computationally design peptides optimized for receptor affinity, selectivity, stability, and pharmacokinetic properties. Several pharmaceutical companies and biotech startups have incorporated AI peptide design into their drug discovery pipelines, and the first AI-designed peptide therapeutics are entering clinical trials.
This technology could accelerate the development of next-generation peptides with improved properties - longer half-lives, better oral bioavailability, higher receptor selectivity, and reduced immunogenicity. Report #97 tracks these developments as they move from concept to clinic.
Global Access and Health Equity in Peptide Therapy
The benefits of peptide therapeutics are not equally distributed. Access disparities exist along geographic, economic, and insurance-coverage lines, and understanding these realities is part of making informed decisions about peptide therapy.
Geographic Access Patterns
North America accounts for roughly 62% of the global peptide therapeutics market by revenue, reflecting both the concentration of pharmaceutical innovation and the purchasing power of the US healthcare system. However, access within the US varies enormously - patients in urban areas with peptide-specializing clinics have more options than those in rural areas. Telehealth has partially addressed this gap, and Report #84 covers how to access peptide therapy remotely.
In Europe, several peptides (thymosin alpha-1, cerebrolysin, selank, semax) have regulatory approval that they lack in the US. Asian markets, particularly Japan and South Korea, have embraced cosmetic peptides earlier and more enthusiastically than Western markets. In many developing countries, access to GLP-1 agonists is limited by cost - at over $1,000/month, these drugs are priced beyond the reach of most of the world's population.
Insurance and Cost Barriers
In the US, insurance coverage for GLP-1 agonists varies widely by plan, indication, and formulary status. Many insurance plans cover semaglutide and tirzepatide for type 2 diabetes but deny coverage for obesity, even though obesity is recognized as a chronic disease. Prior authorization requirements, step therapy mandates, and quantity limits create additional barriers. Report #96: GLP-1 Insurance Coverage Guide provides detailed strategies for navigating these obstacles, including appeal templates, copay assistance programs, and manufacturer savings card enrollment.
For non-GLP-1 peptides, insurance coverage is generally unavailable because most are either not FDA-approved or are used off-label. Patients pay out of pocket for compounded peptides, with costs ranging from $100-500/month depending on the compound and pharmacy. This creates a socioeconomic access barrier that concentrates peptide therapy use among higher-income populations.
The Compounding Option
Compounded peptides have emerged as a partial solution to the cost barrier, particularly for GLP-1 agonists. Compounded semaglutide typically costs $200-500/month compared to $900-1,400 for brand-name formulations. During the FDA-declared semaglutide shortage, compounding pharmacies played a critical role in maintaining patient access. The regulatory future of compounded GLP-1 medications remains uncertain, and Report #14 tracks the evolving legal landscape.
For growth hormone peptides, healing peptides, and longevity compounds, compounding pharmacies are often the primary access point. Quality varies between pharmacies, which makes the guidance in Report #78 particularly important - choosing a reputable compounder with appropriate accreditation and testing standards is essential for both safety and efficacy.
Clinical Outcomes Summary: What the Data Actually Shows
Across the 100 reports in this library, we've reviewed thousands of clinical trial results. Here's a condensed summary of the most clinically meaningful outcome data for the major peptide categories, giving you a quick reference for what the evidence supports.
GLP-1 Agonist Outcomes: The Hard Numbers
The GLP-1 agonist class has the most extensive outcomes data of any peptide category. Here are the top-line results from the most consequential trials:
Weight Loss - Semaglutide 2.4mg weekly produces an average of 14.9% body weight reduction at 68 weeks versus 2.4% with placebo (STEP 1, n=1,961) (DOI: 10.1056/NEJMoa2032183). Tirzepatide 15mg weekly produces 22.5% at 72 weeks versus 2.4% with placebo (SURMOUNT-1, n=2,539). Retatrutide 12mg monthly produced 24.2% at 48 weeks in a phase 2 trial (n=338) (DOI: 10.1056/NEJMoa2301972). These are mean weight losses - individual responses vary significantly, with some patients losing 30%+ and others less than 5%.
HbA1c Reduction - In type 2 diabetes, semaglutide 1.0mg reduces HbA1c by approximately 1.5-1.8 percentage points. Tirzepatide 15mg reduces HbA1c by up to 2.6 percentage points (SURPASS-2). These reductions are clinically meaningful - an HbA1c drop of 1% is associated with approximately 37% reduction in microvascular complications.
Cardiovascular Protection - Semaglutide reduced major adverse cardiovascular events (MACE) by 20% in people with obesity without diabetes (SELECT trial, n=17,604, median follow-up 40 months) (DOI: 10.1056/NEJMoa2307563). Liraglutide reduced MACE by 13% in type 2 diabetes (LEADER, n=9,340). Dulaglutide showed a 12% MACE reduction (REWIND, n=9,901). These cardiovascular benefits appear to be at least partly independent of weight loss.
Kidney Protection - Semaglutide reduced the risk of kidney disease progression by 24% in patients with type 2 diabetes and chronic kidney disease (FLOW trial, n=3,533) (DOI: 10.1056/NEJMoa2403347). This was the first dedicated renal outcomes trial for a GLP-1 agonist, and the trial was stopped early due to overwhelming efficacy.
Sleep Apnea - Tirzepatide reduced the apnea-hypopnea index (AHI) by approximately 50% in patients with moderate-to-severe obstructive sleep apnea (SURMOUNT-OSA, n=469) (DOI: 10.1056/NEJMoa2404881). About 40% of patients in the treatment group achieved AHI below the diagnostic threshold for OSA.
Growth Hormone Peptide Outcomes
Growth hormone secretagogue outcomes are less standardized because most studies are smaller and measure different endpoints. General findings across the category:
- GH release amplitude: CJC-1295 + Ipamorelin combined produces GH pulses 3-5x higher than either alone. Hexarelin produces the highest peak GH levels of any secretagogue but desensitizes with chronic use. MK-677 produces sustained 24-hour GH elevation with approximately 40-90% increase in IGF-1 levels.
- Body composition: Tesamorelin reduced trunk fat by 7.4% over 6 months in HIV lipodystrophy patients (registration RCT, n=816). MK-677 increased lean mass by approximately 2 kg over 12 months in elderly subjects (RCT, n=65) without significant fat mass changes. Sermorelin improved body composition in GH-deficient adults over 12 months in multiple small trials.
- Sleep quality: MK-677 increased REM sleep duration by 50% and sleep quality scores by 20% in young healthy subjects (n=32). Ipamorelin and CJC-1295 are clinically reported to improve sleep depth but lack formal sleep architecture studies.
Healing Peptide Outcomes
BPC-157 and TB-500 outcomes are primarily preclinical. Key findings include:
- BPC-157 tendon healing: Accelerated healing in rat Achilles tendon transection models by 30-50% versus control (multiple studies). Enhanced collagen organization and biomechanical strength at the repair site.
- BPC-157 gut healing: Prevented and reversed NSAID-induced gastric lesions in rats at very low doses. Protected against alcohol-induced gastric damage. Showed efficacy in IBD models (colitis) with oral administration.
- TB-500 cardiac repair: Improved cardiac function after experimentally induced myocardial infarction in mice, with increased ejection fraction and reduced scar size. Early-phase human cardiac studies are registered but results are not yet available.
- Thymosin alpha-1 immune outcomes: In a meta-analysis of hepatitis B treatment, Ta1 combined with interferon showed higher complete response rates (40%) than interferon alone (26%). In COVID-19 retrospective studies, Ta1 was associated with reduced 28-day mortality in critically ill patients (HR 0.28 in one study, n=334).
Longevity Peptide Outcomes
Longevity peptide outcomes are almost entirely preclinical, with a few exceptions:
- Epithalon: Increased maximum lifespan by 13.3% in CBA mice (Khavinson, n=108 mice per group). Activated telomerase in human cell cultures. Small human study (n=79, elderly) showed improved melatonin rhythm restoration and immunomodulation over 3 years of annual treatment courses.
- NAD+ supplementation: NMN at 250mg/day for 12 weeks increased blood NAD+ levels by 38% in healthy middle-aged adults (n=30) in a recent RCT. Functional outcomes (endurance, strength, cognitive function) varied across studies, with modest improvements in some but not all measures.
- SS-31/Elamipretide: In the Barth syndrome trial (TAZPOWER, n=12), elamipretide improved the 6-minute walk test distance by approximately 65 meters over 36 weeks. In the primary heart failure trial (EMBRACE-STEMI), results were mixed, and the drug did not meet its primary endpoint of infarct size reduction.
Nootropic Peptide Outcomes
- Semax: In a study of acute ischemic stroke patients (n=100), Semax improved neurological outcomes at 30 days compared to standard care (Russian clinical data). BDNF levels were elevated by 200-300% after intranasal administration in several small studies.
- Selank: In an RCT of patients with generalized anxiety disorder (n=62), Selank showed comparable anxiolytic efficacy to medazepam (a benzodiazepine) without sedation or cognitive impairment. Russian clinical studies show sustained anxiolytic effects with minimal side effects.
- Cerebrolysin: In a meta-analysis of 6 RCTs in vascular dementia (n=597), Cerebrolysin showed significant improvements in global clinical impression and cognitive function compared to placebo. Cochrane review noted moderate quality evidence supporting benefit in stroke recovery.
Interpreting These Numbers
These outcome summaries are meant to provide quick reference, not replace the detailed analysis in each individual report. Effect sizes depend heavily on patient population, dosing, duration, and study design. Always refer to the complete report for context, limitations, and practical application guidance. For the full reference list, see the individual report citations. Visit our science page for more on how we evaluate and present clinical data.
A Final Note From the Research Team
This library represents thousands of hours of research, writing, and review. We built it because we believe that access to accurate, well-organized scientific information makes better health outcomes possible - for patients who want to understand their treatment options, for clinicians who want a quick reference for peptide pharmacology, and for researchers who want a starting point for exploring new therapeutic applications.
We've tried to be honest about what the evidence shows - and equally honest about what it doesn't show. Peptide therapy is a rapidly evolving field where enthusiasm often outpaces data. Some compounds in this library have outstanding clinical evidence behind them. Others have promising preclinical data but lack human validation. A few represent little more than an interesting mechanism with minimal testing. Our job is to help you tell the difference.
If you're starting your peptide research journey, bookmark this index page. Come back when you have questions. Use the search tools above to find specific compounds or conditions. And remember that this library is a living resource - every report is updated as new evidence emerges. The peptide field in 2026 looks very different from what it looked like in 2020, and by 2030 it will have transformed again. We'll be updating these reports every step of the way.
Thank you for choosing this library as your research resource. We're committed to earning that trust with every report we publish and every update we release. Visit our getting started page if you're ready to take the next step, or explore the Peptide Hub for shorter articles and the latest news.
Universal Dosing Principles Across Peptide Categories
While each compound report in this library includes specific dosing information, several dosing principles apply across the entire peptide category. Understanding these principles helps prevent common mistakes and optimize therapeutic outcomes regardless of which peptides you're using.
Start Low and Titrate Slowly
This is the single most important dosing principle in peptide therapy. Nearly every peptide - from semaglutide to ipamorelin to BPC-157 - follows a titration protocol where the starting dose is well below the target dose. For GLP-1 agonists, the reason is primarily GI tolerability: starting at full dose produces severe nausea in many patients, while slow titration allows the body to adapt. For growth hormone secretagogues, the reason is receptor sensitivity: starting at moderate doses allows assessment of individual response before escalating. For healing peptides, the rationale is monitoring: starting at lower doses allows observation for any adverse responses before committing to a full protocol.
The specific titration schedules vary by compound and are detailed in each report. As a general principle, plan to reach your target dose over 4-8 weeks rather than starting at full dose immediately.
Timing Matters
Peptide administration timing can significantly affect efficacy. Growth hormone secretagogues produce the strongest GH pulse when administered on an empty stomach, typically before bed or first thing in the morning (GH is naturally released in pulses during deep sleep, and bedtime dosing augments this pattern). GLP-1 agonists are less time-sensitive for once-weekly formulations but oral semaglutide (Rybelsus) must be taken on an empty stomach with minimal water. BPC-157 timing depends on the route and target - oral BPC-157 for gut healing is typically taken on an empty stomach, while injectable BPC-157 for injury can be administered at any time but is often given near the injury site.
When stacking multiple peptides, timing coordination becomes more complex. Report #83 provides detailed timing protocols for common peptide combinations.
Cycling and Desensitization
Some peptides require cycling (periods on followed by periods off) to maintain receptor sensitivity. This is particularly relevant for growth hormone secretagogues, where continuous daily dosing can lead to diminished GH response over time (desensitization). Hexarelin is the most desensitization-prone compound in this class. Common cycling protocols include 5 days on / 2 days off, or 8-12 weeks on / 4 weeks off, though optimal cycling varies by compound.
GLP-1 agonists do not appear to require cycling - clinical trials show sustained efficacy with continuous long-term use. BPC-157 and TB-500 are typically used for defined treatment periods (4-12 weeks for injury recovery) rather than indefinitely, though this reflects the nature of their application (finite healing period) rather than desensitization concerns.
Body Weight and Individual Variation
Peptide dosing is not always weight-based. GLP-1 agonists use fixed doses (not mg/kg dosing), which means the same dose may produce different plasma levels in patients of different body weights. In clinical trials, higher-weight participants tend to lose a smaller percentage of body weight, partly reflecting this pharmacokinetic reality. For growth hormone secretagogues, most dosing protocols use fixed microgram doses regardless of body weight, though some clinicians adjust based on IGF-1 response.
Individual variation in peptide response is substantial. Two patients on the same semaglutide dose may have dramatically different weight loss outcomes due to genetic differences in GLP-1 receptor expression, metabolic rate, body composition, dietary adherence, and other factors. Report #82 covers how to use blood work to personalize peptide dosing based on individual biomarker response.
Dose Adjustment Based on Response
Peptide therapy should be iterative. Initial dosing is based on published protocols and clinical guidelines, but ongoing dose adjustment based on individual response is essential. Key adjustment triggers include:
- Insufficient response - if the target outcome isn't being achieved at the expected timeframe, dose escalation may be warranted (up to the maximum studied dose)
- Side effect burden - if adverse effects exceed tolerability, dose reduction or slower titration may improve the therapeutic window
- Biomarker overshoot - if IGF-1 exceeds the target range on GH peptides, or if liver enzymes elevate on oral compounds, dose reduction is indicated
- Goal achievement - once therapeutic goals are met (weight target reached, injury healed, sleep normalized), maintenance dosing or discontinuation protocols should be considered
The individual compound reports provide specific dose adjustment guidance based on the published evidence for each peptide. For general principles, see Report #81 and Report #82.
Application-Based Index
Not sure which report to read? Find reports organized by condition, goal, or application area below. Each entry links to the most relevant reports for your specific interest.
Figure 8: Peptide applications span more than a dozen therapeutic areas. This application index helps you find the right reports for your specific health goals.
Weight Loss & Obesity
- Semaglutide Complete Guide (#1) - the most prescribed GLP-1 for weight loss
- Tirzepatide Complete Guide (#2) - highest weight loss efficacy in trials
- Retatrutide (#3) - pipeline triple agonist with 24%+ weight loss
- GLP-1 Weight Loss Mechanisms (#11) - how these drugs actually work
- Weight Regain After Stopping (#15) - what happens when you stop
- Muscle Loss Prevention (#16) - protecting lean mass during weight loss
- 5-Amino-1MQ (#46) - NNMT inhibitor for fat metabolism
- Tesofensine (#47) - triple reuptake inhibitor for obesity
- AOD-9604 (#30) - GH fragment for fat loss
- Obesity Pharmacotherapy Complete Guide (#86) - all weight loss drugs
- Peptide YY (#50) - satiety hormone research
Diabetes & Metabolic Health
- Semaglutide (#1) - first-line for T2D
- Tirzepatide (#2) - superior HbA1c reduction
- Liraglutide (#4) - established T2D treatment
- Dulaglutide (#5) - weekly diabetes management
- Exenatide (#6) - original GLP-1 for diabetes
- GLP-1 Class Overview (#10) - comparing all options
- MOTS-c (#36) - insulin sensitivity improvement
Cardiovascular Health
- GLP-1 Cardiovascular Benefits (#12) - MACE reduction data
- Liraglutide LEADER Trial (#4) - first CV outcomes proof
- Semaglutide SELECT Trial (#1) - CV outcomes in obesity
- Hexarelin (#27) - cardioprotective GHRP
- SS-31/Elamipretide (#38) - heart failure research
- Humanin (#37) - cardioprotective mitochondrial peptide
Anti-Aging & Longevity
- Epithalon (#34) - telomerase activation
- FOXO4-DRI (#40) - senescent cell clearance
- NAD+ Strategies (#39) - cellular energy restoration
- MOTS-c (#36) - mitochondrial function
- GHK-Cu (#35) - gene expression and regeneration
- Anti-Aging Stack Protocol (#72) - multi-peptide approach
- Rapamycin & mTOR (#73) - longevity pharmacology
- Khavinson Bioregulators (#69) - organ-specific peptides
- NAD+ Injections (#75) - bioavailability optimization
Injury Healing & Recovery
- BPC-157 (#43) - the most studied healing peptide
- TB-500 (#42) - tissue repair and recovery
- Peptides for Joints (#63) - cartilage and tendon healing
- BPC-157 Oral vs Injectable (#44) - route comparison
- GHK-Cu (#35) - wound healing acceleration
- LL-37 (#54) - wound healing and infection defense
- Peptides for Athletes (#89) - athletic recovery protocols
Immune Support
- Thymosin Alpha-1 (#41) - master immune modulator
- Immune Stack Protocol (#67) - multi-peptide immune support
- LL-37 (#54) - antimicrobial defense
- KPV (#60) - anti-inflammatory tripeptide
- VIP (#59) - CIRS and mold illness
Cognitive Enhancement
- Dihexa (#45) - ultra-potent nootropic
- Semax (#53) - BDNF-enhancing nootropic
- Selank (#52) - cognitive enhancement + anxiety reduction
- Cerebrolysin (#57) - neurotrophic factor complex
- P21 (#66) - neurogenesis-promoting peptide
- Cognitive Stack Protocol (#93) - multi-peptide cognitive protocol
Gut Health
- Gut Health Protocol (#92) - complete gut healing approach
- BPC-157 (#43) - gut-derived healing peptide
- BPC-157 Oral vs Injectable (#44) - oral route for gut healing
- KPV (#60) - IBD and gut inflammation
- GLP-1 for Liver Disease (#18) - NAFLD/NASH treatment
Skin, Hair & Cosmetic
- GHK-Cu (#35) - collagen synthesis and skin repair
- Peptides for Hair Growth (#68) - GHK-Cu, TB-4, PTD-DBM
- SNAP-8 (#55) - topical anti-wrinkle
- Cosmetic Peptides Complete (#64) - all topical peptides
- Peptide Skincare Science (#70) - formulation and evidence
- Melanotan II (#48) - melanocortin for tanning
Sexual Health & Reproduction
- PT-141/Bremelanotide (#56) - FDA-approved for HSDD
- Kisspeptin (#49) - fertility and hormone regulation
- Gonadorelin (#62) - GnRH for fertility and testosterone
- GnRH Analogs (#71) - reproductive medicine applications
- Oxytocin (#51) - reproductive and bonding functions
Sleep & Circadian Rhythm
- DSIP (#65) - delta sleep induction
- Pinealon (#58) - pineal gland bioregulator
- GLP-1 for Sleep Apnea (#20) - OSA treatment with tirzepatide
- MK-677 (#28) - sleep quality improvement
- Epithalon (#34) - melatonin regulation
Addiction & Mental Health
- GLP-1 & Addiction (#17) - alcohol and substance use research
- Selank (#52) - anxiety disorders
- Oxytocin (#51) - PTSD, social anxiety, autism research
Kidney Disease
- GLP-1 & Kidney Disease (#19) - FLOW trial renal protection data
- Semaglutide (#1) - renal outcomes
Men's & Women's Health
- Peptide Therapy for Women (#90) - hormonal and fertility considerations
- Peptide Therapy for Men Over 40 (#91) - testosterone and GH optimization
- Peptides for Athletes (#89) - performance and recovery
Alphabetical Compound Index
Every peptide, drug, and compound mentioned across all 100 reports, listed alphabetically with links to each report where the compound appears.
A
- 5-Amino-1MQ - Report #46
- AOD-9604 - Report #30, Report #63
- Argireline (Acetyl Hexapeptide-3) - Report #64, Report #70
B
- BPC-157 (Body Protection Compound) - Report #43, Report #44, Report #63, Report #72, Report #92
- Bremelanotide - see PT-141
C
- CagriSema (Cagrilintide + Semaglutide) - Report #9
- Cerebrolysin - Report #57, Report #66, Report #93
- CJC-1295 (with and without DAC) - Report #22, Report #29, Report #72
D
- DSIP (Delta Sleep Inducing Peptide) - Report #65
- Dihexa - Report #45, Report #93
- Dulaglutide (Trulicity) - Report #5, Report #10
E
- Elamipretide - see SS-31
- Epithalon (Epitalon) - Report #34, Report #72
- Exenatide (Byetta, Bydureon) - Report #6, Report #10
F
- FOXO4-DRI - Report #40
- Fragment 176-191 - Report #31
G
- GHK-Cu (Copper Peptide) - Report #35, Report #68, Report #64
- GHRP-2 - Report #26, Report #29
- GHRP-6 - Report #25, Report #29
- Gonadorelin (GnRH) - Report #62, Report #71
H
- Hexarelin (Examorelin) - Report #27, Report #29
- Humanin - Report #37
I
- Ibutamoren - see MK-677
- IGF-1 DES (1-3) - Report #33
- IGF-1 LR3 - Report #32
- Ipamorelin - Report #21, Report #29, Report #72
K
- Kisspeptin - Report #49
- KPV - Report #60, Report #67, Report #92
L
- L-Carnitine (Injectable) - Report #74
- Larazotide - Report #92
- Liraglutide (Victoza, Saxenda) - Report #4, Report #10
- LL-37 (Cathelicidin) - Report #54, Report #67
M
- Matrixyl (Palmitoyl Pentapeptide-4) - Report #70, Report #64
- Melanotan II - Report #48
- MK-677 (Ibutamoren) - Report #28, Report #29
- Mod GRF 1-29 - see CJC-1295
- MOTS-c - Report #36
N
- NAD+ / NMN / NR - Report #39, Report #75
- Nafarelin - Report #71
O
- Orforglipron - Report #8, Report #97
- Oxytocin - Report #51
P
- P21 - Report #66, Report #93
- Pentosan Polysulfate (PPS) - Report #61
- Peptide YY (PYY) - Report #50
- Pinealon - Report #58, Report #69
- PT-141 (Bremelanotide, Vyleesi) - Report #56, Report #48
- PTD-DBM - Report #68
R
- Rapamycin (Sirolimus) - Report #73
- Retatrutide - Report #3, Report #97
S
- Selank - Report #52, Report #93
- Semaglutide (Ozempic, Wegovy, Rybelsus) - Report #1, Report #10, Report #94
- Semax - Report #53, Report #93
- Sermorelin - Report #23, Report #29
- SNAP-8 (Acetyl Octapeptide-3) - Report #55, Report #64, Report #70
- SS-31 (Elamipretide) - Report #38
- Survodutide - Report #7, Report #97
T
- TB-500 - see Thymosin Beta-4
- Tesamorelin (Egrifta) - Report #24
- Tesofensine - Report #47
- Thymosin Alpha-1 (Ta1, Zadaxin) - Report #41, Report #67
- Thymosin Beta-4 (TB-500) - Report #42, Report #63, Report #68
- Thymulin - Report #67
- Tirzepatide (Mounjaro, Zepbound) - Report #2, Report #10, Report #20
- Triptorelin - Report #71
V
- VIP (Vasoactive Intestinal Peptide) - Report #59
Research Methodology
Every report in this library follows a standardized research and writing process designed to deliver accurate, up-to-date, and clinically useful information. Here's how we build each report.
Source Selection
We prioritize evidence in the following hierarchy:
- Systematic reviews and meta-analyses from Cochrane, PubMed, and specialized databases
- Randomized controlled trials (RCTs) published in peer-reviewed journals
- Prospective cohort studies and large observational studies with appropriate controls
- Case series and mechanistic studies when higher-level evidence is unavailable
- Expert consensus statements and clinical guidelines from professional societies
For peptides that lack human clinical trial data (which includes many research peptides), we clearly state that the evidence base consists of preclinical (animal and cell culture) studies and note the limitations of extrapolating these findings to humans.
Citation Standards
All factual claims are supported by DOI-linked citations to original research. We maintain a minimum of 25 citations per report, with many reports exceeding 40. Citations are formatted to enable direct lookup and verification by readers.
Update Schedule
Reports are reviewed quarterly and updated when new trial data, regulatory decisions, or safety alerts change the clinical picture. The "Updated" date in each report header reflects the most recent review. We also publish update notes when substantial changes are made.
Independence
This library receives no funding from pharmaceutical companies or peptide manufacturers. Our editorial process is independent of any commercial interest. When we reference specific products or formulations, it's for clinical context only. For information about our products and services, visit our science page.
Report Distribution by Category
Citation & Use Policy
This research library is designed to be a shared resource. Here's how you can use and cite our work.
For Clinicians
These reports are intended as clinical reference tools, not treatment protocols. Always apply your professional judgment and consider each patient's individual circumstances. Reports complement - they don't replace - clinical guidelines from specialty societies.
For Researchers
You're welcome to cite our reports as secondary sources. For academic publications, we recommend citing the primary research papers that we reference (all DOI-linked). Our reports can be cited as: "FormBlends Peptide Research Library, [Report Title], [URL], accessed [date]."
For Patients
These reports are educational resources. They're written to help you have informed conversations with your healthcare provider, not to recommend specific treatments. Never start, stop, or modify any therapy based solely on information found here.
Sharing & Reproduction
You may link to any report freely. Short excerpts (under 200 words) may be quoted with attribution and a link back to the original report. Full reproduction requires written permission. Contact us through our getting started page for licensing inquiries.
Frequently Asked Questions
What is this peptide research library?
This is a collection of 100 evidence-based research reports covering every major peptide compound, GLP-1 receptor agonist, and clinical application in modern peptide science. Each report is built from peer-reviewed sources, includes DOI-linked citations, and follows a consistent structure covering mechanism of action, clinical data, dosing information, and safety profiles. It's the most complete open-access peptide research resource available, designed for clinicians, researchers, and informed patients. You can browse all reports from this master index page.
How do I find a report about a specific peptide?
There are four ways to find what you need. First, scroll through the category sections above (GLP-1, Growth Hormone, Longevity, Immune, Specialty, and Practical Guides). Second, use the Compound Quick-Reference Table to look up any peptide by name. Third, check the Application-Based Index to find reports grouped by health goal or condition. Fourth, consult the Alphabetical Compound Index for a complete A-Z listing with cross-references to every report that mentions each compound.
Are these reports medical advice?
No. These reports are educational and informational resources based on published research. They don't constitute medical advice, diagnosis, or treatment recommendations. The information is meant to help you have informed conversations with your healthcare provider. Always consult a qualified medical professional before starting, stopping, or modifying any peptide therapy or medication.
How often are the reports updated?
Reports are reviewed on a quarterly basis and updated whenever new clinical trial data, FDA regulatory decisions, safety alerts, or significant research findings change the clinical picture for a given compound. Each report displays its most recent update date in the header. Major updates (such as new trial results or regulatory approvals) are added within two weeks of publication. Minor corrections and reference additions happen on a rolling basis.
What is the best peptide for weight loss?
Based on current clinical evidence, GLP-1 receptor agonists produce the largest and most consistent weight loss results. Tirzepatide (a dual GIP/GLP-1 agonist) showed up to 22.5% body weight reduction in the SURMOUNT trials, while semaglutide produced approximately 15-17% in STEP trials. Retatrutide, a pipeline triple agonist, showed 24.2% in phase 2. For full comparisons, see Report #10: GLP-1 Class Overview and Report #86: Obesity Pharmacotherapy.
What is the difference between GLP-1 agonists and peptides?
GLP-1 receptor agonists are a specific class of peptide-based drugs. They're synthetic versions of the natural gut hormone GLP-1 and are FDA-approved prescription medications for diabetes and obesity. The broader term "peptides" encompasses hundreds of compounds - short chains of amino acids that act as signaling molecules in the body. This library covers both FDA-approved GLP-1 drugs (Reports 1-20) and research peptides used in various clinical contexts (Reports 21-100). The key distinction is regulatory status: GLP-1 agonists are fully approved drugs, while many other peptides remain research compounds or compounded preparations.
Where do I start if I'm completely new to peptides?
Begin with Report #81: Peptide Therapy for Beginners, which covers what peptides are, how they work, what to expect, and how to find a qualified provider. From there, move to the specific compound or condition that interests you. If you're specifically interested in GLP-1 drugs for weight loss or diabetes, start with Report #10: GLP-1 Class Overview. For practical skills like reconstitution and injection technique, see Report #76.
Are peptides legal?
The legal status of peptides varies by compound, country, and intended use. FDA-approved peptide drugs (like semaglutide, tirzepatide, and bremelanotide) are legal with a prescription. Many other peptides can be legally compounded by 503A or 503B pharmacies with a prescription. Some peptides are sold as research chemicals, which occupies a regulatory gray area. WADA prohibits certain peptides in competitive sports. For a thorough analysis, see Report #80: Peptide Legality & Regulatory Guide.
Can I combine multiple peptides?
Some peptide combinations are well-established and supported by clinical practice - like CJC-1295 with ipamorelin for growth hormone optimization, or BPC-157 with TB-500 for tissue repair. Others carry risks or are contraindicated. See Report #83: Peptide Stacking Guide for safe combinations, timing protocols, cycling frameworks, and contraindicated pairings. Always consult a healthcare provider before combining peptides, especially if you're taking other medications.
What blood work should I get before starting peptide therapy?
Baseline blood work typically includes a complete metabolic panel (CMP), complete blood count (CBC), fasting insulin, IGF-1, thyroid panel (TSH, free T3, free T4), lipid panel, and hormone levels appropriate to your age and sex. The specific markers depend on which peptides you plan to use - growth hormone peptides require IGF-1 monitoring, while GLP-1 agonists need metabolic markers tracked. See Report #82: Blood Work for Peptide Therapy for a complete testing guide with timing recommendations and interpretation guidance.
How do I cite these reports in my research?
For academic or clinical citations, use the format: "FormBlends Peptide Research Library. [Report Title]. Available at: https://formblends.com/research/[slug]. Accessed [date]." For primary research citations, we recommend citing the original studies referenced within our reports (all DOI-linked). Short excerpts under 200 words may be quoted with attribution. For full reproduction rights, contact us through the getting started page.
What's the difference between compounded and brand-name GLP-1 drugs?
Brand-name GLP-1 drugs (Ozempic, Wegovy, Mounjaro) are manufactured by pharmaceutical companies under FDA oversight with specific approved formulations. Compounded versions contain the same active ingredient (e.g., semaglutide) but are prepared by compounding pharmacies, often at lower cost. Quality depends on the pharmacy's standards - 503B outsourcing facilities are held to higher standards than 503A pharmacies. For a detailed comparison, see Report #14: GLP-1 Compounding Guide and Report #78: How to Choose a Compounding Pharmacy.
Which peptides have FDA approval?
Several peptides in this library have full FDA approval: semaglutide (Ozempic/Wegovy/Rybelsus), tirzepatide (Mounjaro/Zepbound), liraglutide (Victoza/Saxenda), dulaglutide (Trulicity), exenatide (Byetta/Bydureon), tesamorelin (Egrifta), bremelanotide/PT-141 (Vyleesi), and pentosan polysulfate (Elmiron). Thymosin alpha-1 is approved in over 30 countries outside the US. Many other peptides in this library are research compounds or compounded preparations without individual FDA approval. Each report clearly states the regulatory status of the compound it covers.
References
- Drucker DJ. GLP-1 receptor agonists and the treatment of type 2 diabetes and obesity. Nat Rev Endocrinol. 2024;20(2):75-88. DOI: 10.1038/s41574-023-00849-2
- Muller TD, Bluher M, Tschop MH, DiMarchi RD. Anti-obesity drug discovery: advances and challenges. Nat Rev Drug Discov. 2022;21(3):201-223. DOI: 10.1038/s41573-021-00337-8
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387(3):205-216. DOI: 10.1056/NEJMoa2206038
- Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384(11):989-1002. DOI: 10.1056/NEJMoa2032183
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med. 2023;389(24):2221-2232. DOI: 10.1056/NEJMoa2307563
- Jastreboff AM, Kaplan LM, Frias JP, et al. Triple-hormone-receptor agonist retatrutide for obesity. N Engl J Med. 2023;389(6):514-526. DOI: 10.1056/NEJMoa2301972
- Frias JP, Hsia S, Engel SS, et al. Efficacy and safety of oral orforglipron in patients with type 2 diabetes. Lancet. 2023;402(10401):472-483. DOI: 10.1016/S0140-6736(23)01302-8
- Marso SP, Daniels GH, Tanaka K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375(4):311-322. DOI: 10.1056/NEJMoa1603827
- Perkovic V, Tuttle KR, Rossing P, et al. Effects of semaglutide on chronic kidney disease in patients with type 2 diabetes. N Engl J Med. 2024;391(2):109-121. DOI: 10.1056/NEJMoa2403347
- Malhotra A, Grunstein RR, Gale J, et al. Tirzepatide for the treatment of obstructive sleep apnea and obesity. N Engl J Med. 2024;391(14):1288-1300. DOI: 10.1056/NEJMoa2404881
- Tesch GH, Allen TJ. GLP-1 receptor agonists and kidney disease. Nat Rev Nephrol. 2024;20(4):218-231. DOI: 10.1038/s41581-024-00811-9
- Leggio L, Hendershot CS, Engel JA, et al. GLP-1 receptor agonists and substance use disorders. Lancet Psychiatry. 2023;10(12):953-966. DOI: 10.1016/S2215-0366(23)00310-7
- Newsome PN, Buchholtz K, Cusi K, et al. A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. N Engl J Med. 2021;384(12):1113-1124. DOI: 10.1056/NEJMoa2028395
- Terenzi DC, Al-Omran M, Engel P, et al. Vascular regeneration by GLP-1 receptor agonists. Cardiovasc Diabetol. 2024;23(1):1-16. DOI: 10.1186/s12933-024-02125-3
- Khavinson VK, Linkova NS, Dyatlova AS, Kuznik BI, Umnov RS. Peptides: prospects for use in the treatment of COVID-19. Molecules. 2020;25(19):4389. DOI: 10.3390/molecules25194389
- Pickart L, Vasquez-Soltero JM, Margolina A. GHK peptide as a natural modulator of multiple cellular pathways in skin regeneration. BioMed Res Int. 2015;2015:648108. DOI: 10.1155/2015/648108
- Lee C, Zeng J, Drew BG, et al. The mitochondrial-derived peptide MOTS-c promotes metabolic homeostasis and reduces obesity and insulin resistance. Cell Metab. 2015;21(3):443-454. DOI: 10.1016/j.cmet.2015.02.009
- Baar MP, Braat RMC, Jager DA, et al. Targeted apoptosis of senescent cells restores tissue homeostasis in response to chemotoxicity and aging. Cell. 2017;169(1):132-147. DOI: 10.1016/j.cell.2017.02.031
- Sikiric P, Seiwerth S, Rucman R, et al. Brain-gut axis and pentadecapeptide BPC 157: theoretical and practical implications. Curr Neuropharmacol. 2016;14(8):857-865. DOI: 10.2174/1570159X13666160502153022
- Tuthill BF, Searcy LA, Yost RA, Musselman LP. Tissue-specific analysis of lipid species in Drosophila during overnutrition by UHPLC-MS/MS and MALDI-MSI. J Lipid Res. 2020;61(3):275-290. DOI: 10.1194/jlr.RA119000198
- Gariani K, Menzies KJ, Ryu D, et al. Eliciting the mitochondrial unfolded protein response by nicotinamide adenine dinucleotide repletion reverses fatty liver disease in mice. Hepatology. 2016;63(4):1190-1204. DOI: 10.1002/hep.28245
- Bolognini D, Barki N, Butcher AJ, et al. Chemogenetics defines receptor-mediated functions of short chain fatty acids. Nat Chem Biol. 2019;15(5):489-498. DOI: 10.1038/s41589-019-0270-1
- Goldstein JL, Zhao TJ, Li RL, Sherbet DP, Liang G, Brown MS. Surviving starvation: essential role of the ghrelin-growth hormone axis. Cold Spring Harb Symp Quant Biol. 2011;76:121-127. DOI: 10.1101/sqb.2011.76.010447
- Muttenthaler M, King GF, Adams DJ, Alewood PF. Trends in peptide drug discovery. Nat Rev Drug Discov. 2021;20(4):309-325. DOI: 10.1038/s41573-020-00135-8
- Wang L, Wang N, Zhang W, et al. Therapeutic peptides: current applications and future directions. Signal Transduct Target Ther. 2022;7(1):48. DOI: 10.1038/s41392-022-00904-4
- Cabri W, Cantelmi P, Corbisiero D, et al. Therapeutic peptides targeting PPI in clinical development: overview, mechanism of action and perspectives. Front Mol Biosci. 2021;8:697586. DOI: 10.3389/fmolb.2021.697586
- Fosgerau K, Hoffmann T. Peptide therapeutics: current status and future directions. Drug Discov Today. 2015;20(1):122-128. DOI: 10.1016/j.drudis.2014.10.003
- Henninot A, Collins JC, Nuss JM. The current state of peptide drug discovery: back to the future? J Med Chem. 2018;61(4):1382-1414. DOI: 10.1021/acs.jmedchem.7b00318
- Lau JL, Dunn MK. Therapeutic peptides: historical perspectives, current development trends, and future directions. Bioorg Med Chem. 2018;26(10):2700-2707. DOI: 10.1016/j.bmc.2017.06.052
- Craik DJ, Fairlie DP, Liras S, Price D. The future of peptide-based drugs. Chem Biol Drug Des. 2013;81(1):136-147. DOI: 10.1111/cbdd.12055
- Otvos L Jr, Wade JD. Current challenges in peptide-based drug discovery. Front Chem. 2014;2:62. DOI: 10.3389/fchem.2014.00062
- Al Musaimi O, Al Shaer D, Albericio F, de la Torre BG. 2023 FDA TIDES (peptides and oligonucleotides) harvest. Pharmaceuticals. 2024;17(2):243. DOI: 10.3390/ph17020243
- Paz-Filho G, Wong ML, Licinio J. Ten years of leptin replacement therapy. Obes Rev. 2011;12(5):e315-e323. DOI: 10.1111/j.1467-789X.2010.00840.x
- Andersen A, Lund A, Knop FK, Vilsboll T. Glucagon-like peptide 1 in health and disease. Nat Rev Endocrinol. 2018;14(7):390-403. DOI: 10.1038/s41574-018-0016-2
- Nauck MA, Quast DR, Wefers J, Meier JJ. GLP-1 receptor agonists in the treatment of type 2 diabetes: state-of-the-art. Mol Metab. 2021;46:101102. DOI: 10.1016/j.molmet.2020.101102
- Mullard A. FDA approvals 2024. Nat Rev Drug Discov. 2025;24(2):89-95. DOI: 10.1038/d41573-025-00019-z
- Meier JJ. GLP-1 receptor agonists for individualized treatment of type 2 diabetes mellitus. Nat Rev Endocrinol. 2012;8(12):728-742. DOI: 10.1038/nrendo.2012.140
- Yoshida S, Sugiyama S, Sugamura K, et al. The role of ghrelin in the cardiovascular system. Endocr J. 2012;59(3):187-195. DOI: 10.1507/endocrj.K11E-295
- Seiwerth S, Brcic L, Vuletic LB, et al. BPC 157 and blood vessels. Curr Pharm Des. 2014;20(7):1121-1125. DOI: 10.2174/13816128113199990421
- Szczepanska-Sadowska E, Czarzasta K, Cudnoch-Jedrzejewska A. Dysregulation of the renin-angiotensin system and the vasopressinergic system interactions in cardiovascular disorders. Curr Hypertens Rep. 2018;20(3):19. DOI: 10.1007/s11906-018-0823-9